Abstract
Background
Headaches are a common source of pain and suffering. The study’s purpose was to assess beta-blockers efficacy in preventing migraine and tension-type headache.
Methods
Cochrane Register of Controlled Trials; MEDLINE; EMBASE; ISI Web of Science, clinical trial registries, CNKI, Wanfang and CQVIP were searched through 21 August 2018, for randomized trials in which at least one comparison was a beta-blocker for the prevention of migraine or tension-type headache in adults. The primary outcome, headache frequency per month, was extracted in duplicate and pooled using random effects models.
Data synthesis
This study included 108 randomized controlled trials, 50 placebo-controlled and 58 comparative effectiveness trials. Compared to placebo, propranolol reduced episodic migraine headaches by 1.5 headaches/month at 8 weeks (95% CI: -2.3 to -0.65) and was more likely to reduce headaches by 50% (RR: 1.4, 95% CI: 1.1–1.7). Trial Sequential Analysis (TSA) found that these outcomes were unlikely to be due to a Type I error. A network analysis suggested that beta-blocker’s benefit for episodic migraines may be a class effect. Trials comparing beta-blockers to other interventions were largely single, underpowered trials. Propranolol was comparable to other medications known to be effective including flunarizine, topiramate and valproate. For chronic migraine, propranolol was more likely to reduce headaches by at least 50% (RR: 2.0, 95% CI: 1.0–4.3). There was only one trial of beta-blockers for tension-type headache.
Conclusions
There is high quality evidence that propranolol is better than placebo for episodic migraine headache. Other comparisons were underpowered, rated as low-quality based on only including single trials, making definitive conclusions about comparative effectiveness impossible. There were few trials examining beta-blocker effectiveness for chronic migraine or tension-type headache though there was limited evidence of benefit.
Registration
Prospero (ID: CRD42017050335).
Introduction
Headaches are a common problem, world-wide. The two most common types of headaches are migraine and tension-type. Migraines have a prevalence of 6–8% [1–9], and cause significant disability [10–13], even during periods between attacks [14]. Migraines are responsible for $1 billion in medical costs and $16 billion in lost productivity per year [15;16] in the US alone. While episodic migraine is more common than chronic migraine, chronic migraine has greater disability as well as financial and occupational consequences [8;9] and has received much greater research attention [17].
Tension-type headache is more common than migraine; up to 90% of adults experience one at some time in their life [18–22]. In any given month, a tension-type headache occurs in 46% of adults [22]. Most tension-type headaches are managed with over the counter medications, consequently most do not seek medical attention. However, tension-type headache reduces the quality of life [23], results in up to a fifth of all missed work days [24], and costs EUR 21 billion annually in Europe [25].
There are several options available for preventing migraines including alpha antagonists, antiepileptics [26], beta-blockers [27], botulinum toxin-A [28], calcium channel blockers [29], flunarizine [17], pizotifen [17], serotonin agonists [30], serotonin reuptake inhibitors (SSRIs) [31] and tricyclic antidepressants (TCAs) [32]. Nearly half of males and a third of females who are candidates for prophylactic therapy do not receive it [33]. Selection of prophylactic treatment is tailored on individual patient characteristics, costs, perceived efficacy of the intervention and side effects of the available options.
The 2012 American Academy of Neurology guideline recommends beta-blockers, specifically propranolol and metoprolol, as first line therapy for preventing migraines [34]. Specific medications commonly used in prophylaxis has not been well described. In Europe, commonly prescribed prophylactic agents include antiepileptics, beta-blockers, flunarizine, pizotifen and TCAs [35]. Other studies found that specialists are twice as likely to prescribe antiepileptics than primary care providers [36], that treatment persistence is low [37] and that use of prophylactic medications has increased [38], though none of these three characterized the specific medications used.
The purpose of this study is to assess the efficacy of beta-blockers in the prophylaxis of migraine and tension-type headache. Two previous systematic reviews focused on the use of beta-blockers in migraine headaches, both are more than 15 years old [39;40], and included limited outcomes, though both suggest benefit of beta-blockers compared to placebo. There are two more recent comparative effectiveness analyses of headache management that included beta-blockers. Shamliyan reviewed pharmacologic treatment for episodic migraine and reported that beta-blockers were effective; their outcome was 50% reduction in headaches, an outcome recommended by the International Headache Society (IHS) as a secondary outcome. They also excluded beta-blockers not approved for headaches in the U.S [41]. In the other meta-analysis, we found that beta-blockers were beneficial for migraine headaches, but did not differentiate between episodic and chronic migraine headaches, did not include all possible outcomes and did not examine beta-blockers for management of tension-type headache [17].
Methods
This study was conducted in accordance with PRISMA guidelines (S1 Table. Prisma Checklist) [42] and was registered in PROSPERO (ID: CRD42017050335). Databases searched (without language restriction) included the Cochrane Central Register of Controlled Clinical Trials, MEDLINE, EMBASE, ISI Web of Science (SCI, SSCI, CPCI-S & CPCI-SSH), and three Chinese databases (CNKI, Wanfang and CQVIP) trough 21 August 2018 using the search strategies in supporting information (S2 Table. Search Strategy). Randomized controlled studies of adults that were at least four weeks in duration and used a beta-blocker in at least one study arm were included. Articles were assessed for inclusion in duplicate (JLJ, AK). Because the definition of headache has changed over time, articles were reviewed by at least two authors to determine if the headache could be reasonably classified as migraine or tension-type headache and as either frequent episodic or chronic according to the most recent IHS criteria [43]. IHS recommendations were followed by including only patient-reported outcomes [44] and including the monthly headache frequency as the primary outcome. Additional outcomes included headache index, headache days, severity, duration, quality of life, the use of acute analgesic medications, the proportion with at least 50% improvement in headaches, study withdrawal and the occurrence of adverse events. Data were abstracted in duplicate. Because of the large volume of articles, after training on a separate set of pediatric headache articles, the articles were divided among the authors with all authors serving as the primary abstractor for some articles and as the secondary reviewer, assessing for data accuracy in other articles. Disagreements resolved through consensus between the two and if consensus could not be reached, the entire group discussed and made consensual decisions.
Bias was assessed using the Cochrane risk of bias instrument [45] as well as the JADAD scale [46]. Study size was also included as a risk of bias based on sample size calculations. It was estimated that 60 subjects were required for continuous outcomes and 200 for dichotomous ones (S3 Table. Quality Ratings of Included Trials) based on results from our previous review of treatment of migraine headaches [17]. Studies with more than one arm were pooled into a single arm (if the study reported no differences between arms). For crossover trials, several approaches were used, depending on how the data was reported. For trials that provided only pooled data from both time-periods, the sample size was reduced by 50%, to avoid over-weighting the study [45]. For trials that provided data from both time periods separately, if there was no difference between the two-time periods, the average point estimates and variance was used, with reduction of the sample size by 50%.
The preference was to pool study outcomes in their original unit of analysis. Headache frequency was pooled as headache days per month, headache duration as hours per month, and analgesic use as number of doses per month. Since headache severity and headache index metrics varied, these outcomes were pooled using standardized mean differences [47]. Missing outcome variances were imputed from the reported mean, sample size, and P values [48]. Heterogeneity was assessed using Chi2 (Cochrane Q), Galbraith plots [49] and the I2 statistic [50]. Data were pooled at each reported time point using a random-effects model [51] using Stata (V15.1 College Station TX). A priori, the analytic plan was to pool data at 4, 8, 12, 16, 20 and 24 weeks. Studies that reported outcomes at different time points were combined at the closest time point available (for example, 9-week outcomes were pooled with the 8-week group). For comparative effectiveness trials with 2 or fewer studies, outcomes were reported at the last point reported. Small study effects (publication bias) was assessed using the methods of Peters [52] for dichotomous outcomes and Egger [53] for continuous outcomes if there were a sufficient number of studies. Trial sequential analysis was performed for the comparisons of propranolol to placebo for headache frequency (at 8 weeks), using the O’Brien-Fleming method of alpha-spending function to robustness of the pooled estimates against type 1 and type 2 error [54], using TSA software (Copenhagen, Denmark).
A network meta-analysis was performed for beta-blockers that were compared with placebo at 8 and 12 weeks using the residual maximum likelihood with a modification to the coefficients’ estimated variance using the Kapp and Hartung approach [55] that had a minimum of 2 studies. Both 8- and 12-week results were pooled including all beta-blockers using multivariate random-effects meta-analysis using the network package in STATA [56].
Finally, the quality of evidence was assessed using the GRADE (Grades of Recommendation, Assessment, Development and Evaluation) system to rate the quality of the evidence (GRADEPro GDT 2015) following Cochrane guidelines [45]. Grade assesses quality in four levels: High (further research is unlikely to change estimate of effect); Moderate (further research may impact effect estimate); Low (further research is likely to have important impact on estimate); Very low (any estimate of effect is very uncertain).
Results
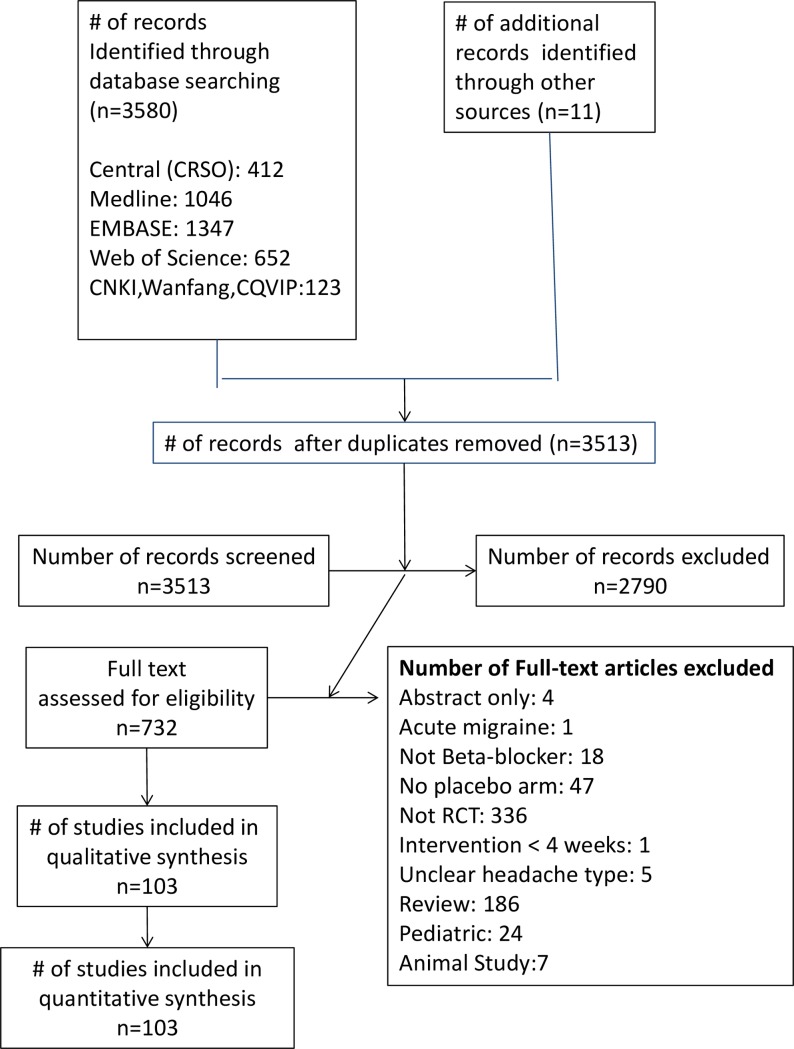
The literature search yielded 3513 unique studies after excluding duplicates. Application of inclusion and exclusion criteria (Fig 1) resulted in 108 randomized controlled trials [57–164], of which 50 had a placebo arm [57–59;62;68;69;71;72;75;77–79;81–83;88;94–97;99;107;108;113;115;116;118;121–124;126;130;133–138;141;142;144;145;147;149;151;152;152;154;156;157;162]. Because some placebo-controlled trials included non-placebo comparisons, there were a total of 86 comparative effectiveness arms. Nearly all trials (n = 106) focused on migraine headaches, most (n = 83) could be classified as episodic. Only 4 trials studied chronic migraines [76;105;129;151] and there was only 1 trial of beta-blockers for chronic tension-type headache [57].
Fig 1. PRISMA flow chart.
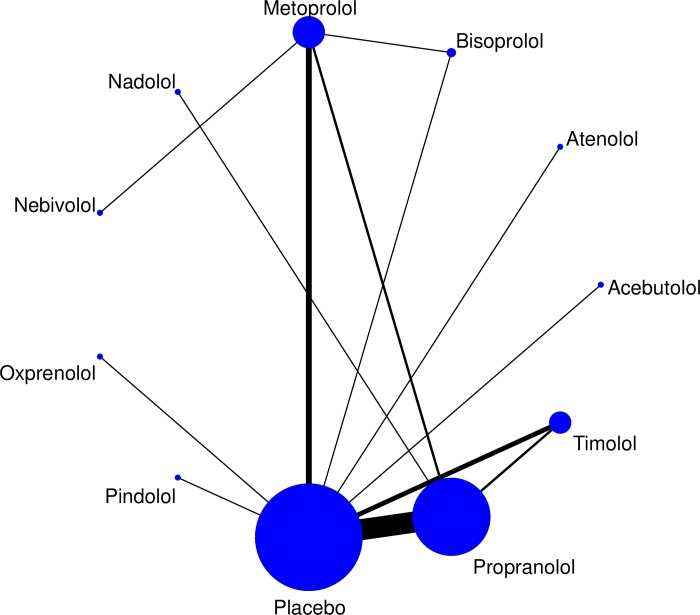
The 108 included studies ranged from 4 to 64 weeks in duration (average: 12.9). Fifty-one were parallel in design and 57 had a crossover design. Among crossover trials, 43 were randomized, with washout periods ranging from zero to four weeks. Twenty-five different countries (Table 1) and four languages (Chinese (n = 15), English (n = 86), German (n = 6), Polish (n = 1) were represented. The average age was 38.6 years and 77% of participants were women. Ten different beta-blockers were studied. Propranolol (n = 74) and metoprolol (n = 21) were the most commonly evaluated beta-blockers. Atenolol, nadolol, pindolol and timolol had two studies each. Several beta-blockers were evaluated in only a single trial (acebutolol, alprenolol, bisoprolol and oxprenolol). Study characteristics for included trials are provided in Table 1 and quality ratings are given in S2 Table.
Table 1. Included trial characteristics.
| Study, Year, Country | Comparison (mg) | Headache Type | Classification | Design | Duration (weeks) | Sample Size (dropout %) | Age | Women | Dose titrated | Rescue Medication Allowed |
|---|---|---|---|---|---|---|---|---|---|---|
| Acebutolol | ||||||||||
| Nanda, 1978, Scotland | Acebutolol (800) v. Placebo | Migraine-episodic | NS | Crossover | 12 | 43 (23%) | NS | 74% | Yes | Yes |
| Alprenolol | ||||||||||
| Ekbom, 1975, Sweden | Alprenolol (200) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 6 | 33 (15) | 41.5 | 82% | No | Yes |
| Atenolol | ||||||||||
| Forssman, 1983, Sweden | Atenolol (100) v. Placebo | Migraine-unspecified | Ad-hoc 1962 | Crossover | 13 | 24 (17) | 40 | 80% | No | Yes |
| Johannsson, 1987, Sweden | Atenolol (100) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 72 (13) | 43 | 70% | No | Yes |
| Stensrud, 1980, Norway | Propranolol (160) v. Atenolol (100) | Migraine-episodic | Ad-hoc 1962 | Crossover | 6 | 35 (20) | NS | 69% | No | Yes |
| Bisoprolol | ||||||||||
| van de Ven, 1997, Europe | Bisoprolol (5) v. Bisoprolol (10) v. Placebo | Migraine-episodic | IHS1988 | Parallel | 12 | 226 (14) | 38.7 | 82% | No | Yes |
| Metoprolol | ||||||||||
| Andersson, 1983, Denmark | Metoprolol (200) v. Placebo | Migraine-episodic | WFNRG 1969 | Parallel | 8 | 71 (13) | 39.7 | 85% | No | Yes |
| Diener, 2001, Europe | Metoprolol (200) v. Aspirin (300) | Migraine-episodic | IHS 1988 | Parallel | 16 | 270 (15) | 39.4 | 81% | Yes | Yes |
| Gong, 2016, China | Metoprolol (25) + Flunarizine (5) v. Flunarizine (5) | Migraine-unspecified | Parallel | 12 | 80 (0) | 47.5 | 40% | No | Yes | |
| Grotemeyer, 1988, Germany | Metoprolol (200) v. Flunarizine (10) | Migraine-episodic | Ad hoc 1962 | Crossover | 7 | 29 (17) | 39 | 79% | No | Yes |
| Grotemeyer, 1990, Germany | Metoprolol (200) v. Acetylsalicyclic Acid (1500) | Migraine-episodic | IHS 1988 | Crossover | 12 | 28 (NS) | 31 | 82% | No | Yes |
| Hesse, 1994, Denmark | Metoprolol (100) v. Acupuncture | Migraine-episodic | IHS 1988 | Parallel | 17 | 85 (10) | 44.7 | 84% | No | Yes |
| Kangasniemi, 1987, Scandinavia | Metoprolol (200) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 8 | 77 (11) | 37.5 | 80% | No | Yes |
| Langohr, 1985, Germany | Metoprolol (100) v. Clomipramine (100) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 4 | 63 (43) | 44.4 | 67% | No | Yes |
| Li, 2006, China | Metoprolol (125) v. Placebo | Migraine-unspecified | IHS 1988 | Parallel | 12 | 60 (0) | 48.5 | 100% | No | Yes |
| Louis 1985, Europe | Metoprolol (100) v. Clonidine (0.1) | Migraine-episodic | WFNRG 1969 | Crossover | 8 | 31 (26) | 35.5 | 81% | Yes | Yes |
| Ma, 2011, China | Metoprolol (50) + Flunarizine (5) v. Flunarizine (5) | Migraine-episodic | HIS 2004 | Parallel | 48 | 56 (0) | 36.3 | 65% | No | Yes |
| Schellenberg, 2008, Germany | Metoprolol (142.5) v. Nebivolol (5) | Migraine-episodic | IHS 2004 | Parallel | 18 | 30 (7) | 39 | 87% | Yes | Yes |
| Siniatchkin, 2007, Germany | Metoprolol (200) v. Placebo | Migraine-unspecified | IHS2004 | Parallel | 12 | 20 (0%) | 37 | 85% | Yes | Yes |
| Sorensen 1991, Denmark | Metoprolol (200) v. Flunarizine (10) | Migraine | IHS 1988 | Parallel | 20 | 149 (1) | 42 | 79% | No | Yes |
| Steiner, 1988, UK | Metoprolol Cr (100) v. Placebo | Migraine-episodic | Vahlquist 1955 | Parallel | 8 | 59 (NS) | 37.4 | 76% | No | Yes |
| Streng 2005, Germany | Metoprolol (200) v. Acupuncture) | Migraine-episodic | IHS 1997 | Parallel | 12 | 114 (17) | 36.6 | 88% | Yes | Yes |
| Vilming, 1985, Sweden | Metoprolol (100) v. Pizotifen (1.5) | Migraine-episodic | WFNRG 1969 | Crossover | 8 | 35 (14) | 37.6 | 83% | Yes | Yes |
| Worz 1992, Germany | Metoprolol (200) v. Bisoprolol (10) | Migraine-episodic | IHS1988 | Crossover | 12 | 125 (38) | 38.5 | 71% | Yes | Yes |
| Yang, 2006, China | Metoprolol (90) v. Placebo | Migraine-episodic | IHS 1988 | Parallel | 12 | 60 (0) | 48.5 | 100% | No | Yes |
| Yang, 2016, China | Metoprolol (95) v. Metoprolol (95) + Fluoxetine | Migraine-episodic | NS | Parallel | 6 | 120 (0) | 38.4 | 64% | No | Yes |
| Zhou, 2015, China | Metoprolol (95) v. Metoprolol (95) + Fluoxetine | Migraine-episodic | NS | Parallel | 6 | 112 (0) | 37.0 | 63% | No | Yes |
| Nadolol | ||||||||||
| Freitag, 1984, USA | Nadolol (80) v. Nadolol (160) v. Placebo | Migraine-unspecified | Ad-hoc 1962 | Parallel | 12 | 32 (20) | 36.7 | 81% | No | Yes |
| Ryan, 1982, USA | Nadolol (80) v. Nadolol (160) v Nadolol (240) v Placebo | Migraine-episodic | NS | Parallel | 12 | 80 (1%) | NS | 78% | No | Yes |
| Oxprenolol | ||||||||||
| Ekbom, 1977, Sweden | Oxprenolol (240) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 34 (12) | 41.8 | 76% | No | Yes |
| Pindolol | ||||||||||
| Ekbom, 1972, Sweden | Pindolol (7.5) v. Pindolol (15) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Parallel | 4 | 30 (NS) | 33.7 | 87% | No | Yes |
| Pindolol + Amitriptyline | ||||||||||
| Agius, 2013, Italy | Pindolol (10)+ Amitriptyline (10) v. Amitriptyline (10) v. Placebo | Tension-chronic | IHS 2004 | Parallel | 8 | 64 (3) | 35.6 | 74% | No | Yes |
| Streng, 2005, Germany | Metoprolol (200) v. Acupuncture | Migraine-episodic | IHS 1997 | Parallel | 12 | 114 (17) | 36.6 | 88% | Yes | Yes |
| Propranolol | ||||||||||
| Ahuja, 1985, India | Propranolol (120) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 8 | 26 (NS) | NS | 46% | No | NS |
| al-Qassab, 1993, UK | Propranolol (80) v. Propranolol (160) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 8 | 45 (33) | 36 | 80% | No | Yes |
| Albers, 1989, USA | Propranolol (180) v. Nifedipine (60) | Migraine-episodic | Ad hoc 1962 | Parallel | 24 | 40 (37) | 35.2 | 89% | Yes | Yes |
| Andersson, 1981, Denmark | Propranolol (160) v. Femoxitine (400) | Migraine | NS | Crossover | 24 | 49 (24) | 38 | 69% | Yes | Yes |
| Ashtari, 2008, Iran | Propranolol (80) vs. Topiramate (50) | Migraine-episodic | IHS 2005 | Parallel | 8 | 62 (3) | 30.8 | 82% | Yes | Yes |
| Baldrati, 1983, Italy | Propranolol (80) v. Aspirin (1.9 mg/kg) | Migraine-not specified | Ad hoc 1962 | Crossover | 12 | 18 (33) | 33.3 | 89% | No | NS |
| Behan, 1980, Scotland | Propranolol (120) v. Methysergide (3) | Migraine-not specified | NS | Crossover | 12 | 56 (36) | NS | 66% | No | No |
| Bonuso, 1998, Italy | Propranolol (80) v. Flunarizine (10) | Migraine-not specified | IHS 1988 | Parallel | 8 | 50 (16) | 32 | 68% | No | NS |
| Bordini, 1997, Brazil | Propranolol (60) v. Flunarizine (10) v. Combo. | Migraine-episodic | IHS 1988 | Parallel | 17 | 52 (13) | 31.2 | 91% | No | Yes |
| Borgesen, 1974, Denmark | Propranolol (120) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 12 (33) | 37.6 | 83% | Yes | Yes |
| Carroll, 1990, UK | Propranolol (80) v. Propranolol (160) | Migraine-episodic | Ad hoc 1962 | Crossover | 12 | 51 (27) | 39 | 69% | No | Yes |
| Chen, 2009, China | Propranolol (60) + Flunarizine (10) v. Topoiramate (100) | Migraine-episodic | IHS 1988 | Parallel | 12 | 82 (0) | 38.2 | 60% | Yes | Yes |
| Dahlof, 1987, Sweden | Propranolol (120) v. Placebo | Migraine-episodic | WFNRG 1969 | Crossover | 4 | 28 (0) | NS | 93% | No | Yes |
| Diener, 1996, German | Propranolol (120) v. Cylcendalate (1200) v. Placebo | Migraine-episodic | IHS 1988 | Parallel | 12 | 214 (17) | 39 | 78% | Yes | Yes |
| Diener, 2002, Germany | Propranolol (160) v. Flunarizine (5) v. Flunarizine (10) | Migraine-episodic | IHS 1988 | Parallel | 16 | 808 (18) | 38.8 | 63% | Yes | Yes |
| Diener, 2004, Europe | Propranolol (160) v. Topiramate (100) v. Topiramate (200) v. Placebo | Migraine-episodic | IHS 1988 | Parallel | 26 | 575 (37) | 41 | 80% | Yes | Yes |
| Domingues, 2009, Brazil | Propranolol (80) v. Nortriptyline (40) v. Combo. | Migraine-chronic | IHS 2004 | Parallel | 12 | 76 (42) | NS | NS | Yes | Yes |
| DongXiang, 2010, China | Propranolol (90) + Amitriptyline (100) v. Amitriptyline (100) | Migraine-episodic | HIS 1988 | Parallel | 12 | 310 (0) | 32.5 | 80% | Yes | Yes |
| Formisano, 1991, Italy | Propranolol (120) v. Nimodipine (120) | Migraine-episodic | IHS 1988 | Parallel | 16 | 22 (14) | 39.2 | 55% | No | Yes |
| Forssman, 1976, Sweden | Propranolol (240) v. Placebo | Migraine-unspecified | NS | Crossover | 10 | 40 (20) | 37.4 | 88% | No | Yes |
| Gawel, 1992, Canada | Propranolol (120) v. Flunarizine (10) | Migraine-episodic | WFNRG 1970 | Parallel | 16 | 94 (19) | 35.9 | 89% | Yes | Yes |
| Gerber, 1991, Germany | Propranolol (120) v. Metoprolol (150) v. Nifedipine (30) | Migraine-episodic | IHS 1988 | Parallel | 12 | 58 (NS) | 42.4 | 73% | Yes | Yes |
| Gerber, 1995, Germany | Propranolol (120/160) v. Cyclandelate (1200/1600) | Migraine-episodic | IHS 1988 | Parallel | 8 | 84 (26) | 40.9 | 90% | No | Yes |
| Ghobadi, 2013, Iran | Propranolol (120) v. Nimodipine (30) | Migraine | IHS 2004 | Parallel | 24 | 102 (2) | 47 | 83% | No | Yes |
| Grotemeyer, 1987, German | Propranolol (120) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 30 (20) | 36 | 73% | No | Yes |
| Havanka-Kannianen, 1988, Finland | Propranolol (80) v. Propranolol (160) | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 48 (13) | 37.7 | 81% | No | Yes |
| Hedman, 1986, Denmark | Propranolol (80) v. Metoprolol (100) | Migraine-episodic | WFNRG 1970 | Crossover | 4 | 12 (0) | 40 | 67% | NS | Yes |
| Holdorff, 1977, Germany | Propranolol (120) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Parallel | 12 | 53 (30) | NS | NS | No | Yes |
| Holroyd, 2010, USA | Propranolol/Nadolol v. Propranolol/Nadolol + Behavior Therapy v. Behavior therapy v. Placebo | Migraine-episodic | IHS 1988 | Parallel | 64 | 232 (51) | 38.2 | 79% | Yes | Yes |
| Jin, 2001, China | Propranolol (30) + Flunarizine (10) v. Diazepam (30) + Nimodipine (60) | Migraine | NS | Parallel | 24 | 84 (0) | NS | 75% | No | Yes |
| Johnson, 1986, New Zealand | Propranolol (240) Mefenamic Acid (1500) v. Placebo | Migraine-episodic | NS | Crossover | 12 | 29 (41) | 42 | 69% | No | Yes |
| Kangasniemi 1983, Finland | Propranolol (160) v. Femoxetine (400) | Migraine-episodic | NS | Crossover | 12 | 29 (11) | 37 | 86% | No | Yes |
| Kangasniemi 1984, Finland | Propranolol (240) v. Metoprolol (200) | Migraine-episodic | WFNRG 1970 | Crossover | 8 | 36 (6) | 33.8 | 89% | No | Yes |
| Kaniecki, 1997, USA | Propranolol (240) v. Divalproex (1500) | Migraine-episodic | IHS 1988 | Crossover | 12 | 37 (14) | NS | 81% | Yes | Yes |
| Kass, 1980, Norway | Propranolol (160) v. Clonidine (0.1) | Migraine-unspecified | WFNRG 1970 | Crossover | 16 | 23 (9) | 39.7 | 30% | No | Yes |
| Kaushik, 2005, India | Propranolol (80) v. Biofeedback | Migraine-episodic | IHS 1988 | Parallel | 24 | 192 (13) | NS | 69% | No | Yes |
| Ke, 2003, China | Propranolol (30) v. Propranolol (30) + Flunarizine (5) v. Flunarizine (5) | Migraine-chronic | IHS 1988 | Parallel | 8 | 121 (0) | 31 | 74% | No | Yes |
| Kjaersgard 1994, Denmark | Propranolol (120) v. Tolfenamic Acid (300) | Migraine-unspecified | IHS 1988 | Crossover | 12 | 76 (26) | 43.3 | 79% | No | Yes |
| Klapper, 1994,USA | Propranolol (140) v. Divalproex (1100) | Migraine-unspecified | IHS 1988 | Crossover | 8 | 24 (50) | NS | NS | Yes | Unclear |
| Kozubski, 1995, Poland | Propranolol (160) Valproaic Acid (1500) | Migraine-unspecified | IHS 1988 | Crossover | 10 | 35 (NS) | NS | 100% | Yes | NS |
| Kuritzky, 1987, Israel | Propranolol (160) v. Placebo | Migraine-episodic | NS | Crossover | 8 | 38 (18) | NS | NS | No | Yes |
| Li, 2002, China | Propranolol (30) v. Flunarizine (5) | Migraine-Episodic | IHS, 1988 | Parallel | 4 | 126 (0) | 38.7 | 60% | No | Yes |
| Li, 2004, China | Propranolol (60) v. Valproate (.45 mg/kg) | Migraine-Episodic | NS | Parallel | 36 | 40 (0) | NS | NS | No | NS |
| Lucking, 1988, Germany | Propranolol (120) v. Flunarizine (10) | Migraine-episodic | NS | Parallel | 16 | 521 (NS) | 42 | 80% | No | Yes |
| Maissen, 1991, Germany | Propranolol (120) v. 5-Hydroxytryptophan (300) | Migraine-episodic | NS | Parallel | 16 | 39 (18) | 39.4 | 67% | Yes | Yes |
| Malvea, 1973, USA | Propranolol (NS) v. Placebo | Migraine-episodic | NS | Crossover | 6 | 31 (6) | NS | 87% | Yes | Yes |
| Mathew, 1980, USA | Propranolol (160) v. Placebo v. Amitriptyline (75) v. Biofedback | Mixed- headaches | NS | Parallel | 24 | 340 (20) | 35.5 | 94% | Yes | Yes |
| Mikkelsen, 1986, Denmark | Propranolol (120) v. Tolfenamic Acid (300) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 31 (21) | 39.4 | 84% | No | Yes |
| Nadelmann, 1986, USA | Propranolol (320) v. Placebo | Migraine-unspecified | Ad-hoc 1962 | Crossover | 12 | 57 (39) | NS | 86% | No | Yes |
| Nair, 1975, India | Propranolol (80) v. Placebo | Migraine-episodic | NS | Crossover | 8 | 20 (0) | 27.3 | 50% | No | No |
| Nambiar, 2011, India | Propranolol (80) v. Riboflavin (100) | Migraine-episodic | IHS 1988 | Parallel | 24 | 100 (NS) | 31 | 55% | Yes | Yes |
| Palferman, 1983, UK | Propranolol (120) v. Placebo | Migraine-unspecified | NS | Crossover | 8 | 10 (38) | 41.4 | 80% | No | Yes |
| Olerud, 1986, Sweden | Propranolol (80) v. Nadolol (80) | Migraine-episodic | NS | Parallel | 24 | 28(NS) | NS | 79% | No | Yes |
| Pita, 1977, Spain | Propranolol (160) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 8 | 9 (0) | 32 | 78% | No | Yes |
| Pradalier, 1989, Norway | Propranolol (160) v. Placebo | Migraine-episodic | IHS 1988 | Parallel | 12 | 74 (26) | 37.4 | 76% | No | NS |
| Ryan, 1984, USA | Propranolol (160) v. Nadolol (80) v. Nadolol (160) | Migraine-episodic | NS | Parallel | 12 | 48 (6) | NS | 73% | No | Yes |
| Sargent, 1985, USA | Propranolol (120) v. Naproxen (1100) v. Placebo | Migraine-episodic | NS | Parallel | 14 | 149 (16) | 30 | 79% | Yes | Yes |
| Shimell, 1990, South Africa | Propranolol (180) v. Flunarizine (10) | Migraine-episodic | Ad hoc 1962 | Parallel | 16 | 58 (2) | 34 | 70% | Yes | NS |
| Silberstein, 2012, USA | Propranolol (240) + Topiramate (100) v. Topiramate (100) | Migraine-chronic | IHS 2006 | Parallel | 24 | 191 (39) | 42 | 90% | Yes | Yes |
| Sjaastad, 1972, Norway | Pindolol (15) v. Placebo | Migraine-episodic | NS | Crossover | 4 | 28 (14) | 35.3 | 79% | Yes | Yes |
| Soyka, 1990, Germany | Propranolol (120) v. Flunarizine (10) | Migraine-unspecified | NS | Parallel | 16 | 434 | 42 | 82% | Yes | Yes |
| Standnes, 1982, Norway | Propranolol (80) v. Timolol (10) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 25 (28) | 41.4 | 80% | Yes | Yes |
| Stensrud, 1976, Norway | Propranolol (160) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 4 | 20 (5) | 43.5 | 70% | No | Yes |
| Stensrud, 1980, Norway | Propranolol (80) v. Atenolol (50) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 6 | 35 (20) | NS | 69% | No | Yes |
| Stovner, 2014, Norway | Propranolol (160) v. Candesartan (16) v. Placebo | Migraine-episodic | NS | Crossover | 12 | 72 (15) | 37 | 82% | Yes | Yes |
| Sudilovsky, 1987, USA | Propranolol (160) v. Nadolol (80) v. Nadolol (160) | Migraine-episodic | Ad hoc 1962 | Parallel | 12 | 140 (30) | 39.3 | 76% | Yes | Yes |
| Tfelt-Hansen, 1984, Scandinavia | Propranolol (160) v. Timolol (20) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 96 (28) | 39.5 | 74% | No | Yes |
| Weber, 1971, USA | Propranolol (20) v. Placebo | Migraine-unspecified | Ad-hoc 1962 | Crossover | 12 | 25 (24) | 40.6 | 52% | No | Yes |
| Wideroe, 1974, Norway | Propranolol (160) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 12 | 30 (13) | 40 | 90% | No | Yes |
| Wen, 2016, China | Propranolol (30) v. Flunarizine (10) | Migraine Episodic | NS | Parallel | 8 | 100 (0) | 25.6 | 65% | No | Yes |
| Yuan, 2005, China | Propranolol (120) v. Topiramate (150) | Migraine-unspecified | IHS 1988 | Parallel | 12 | 67 (0) | 29.9 | 64% | Yes | No |
| Zhu, 2005, China | Propranolol (30) v. Flunarizine (10) | Migraine-unspecified | IHS 1988 | Parallel | 8 | 90 (0) | 28.1 | 73% | No | No |
| Ziegler, 1993, USA | Propranolol (240) v. Amitriptyline (150) v. Placebo | Migraine-episodic | NS | Crossover | 10 | 54 (44) | 38 | 73% | Yes | Yes |
| Timolol | ||||||||||
| Briggs, 1979, UK | Timolol (20) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 6 | 14 (7) | NS | 71% | No | Yes |
| Stellar, 1984, USA | Timolol (30) v. Placebo | Migraine-episodic | Ad-hoc 1962 | Crossover | 8 | 107 (12) | 43 | 72% | No | Yes |
NS: Not Stated
Studies had a number of common quality problems (S3 Table) including high drop-out rates (16.1%, range 0–51%), lack of intention to treat analysis (76%), inadequate sequence generation (83%), lack of evidence of concealed allocation (90%) and inadequate blinding (60%). Twenty-three studies assessed compliance (21%). Fifty-one (47%) studies reported all collected outcomes. Sixteen trials (15%) were sponsored by industry. All comparisons that had only a single study were graded as low-quality evidence.
Episodic migraines
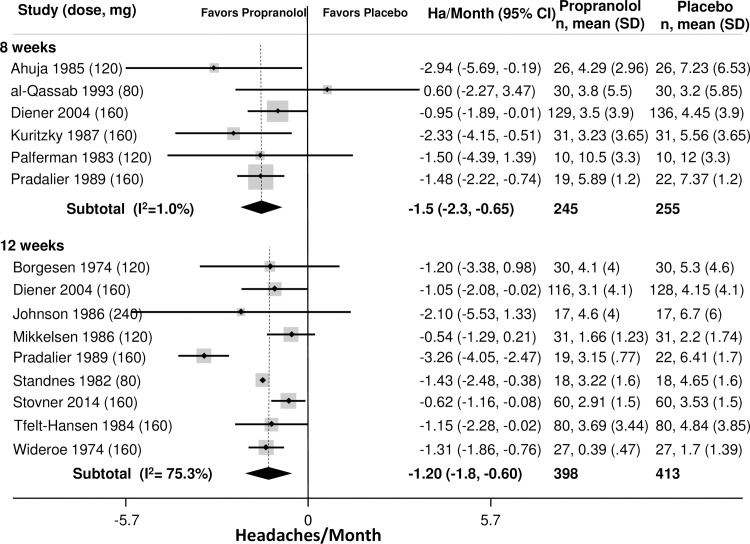
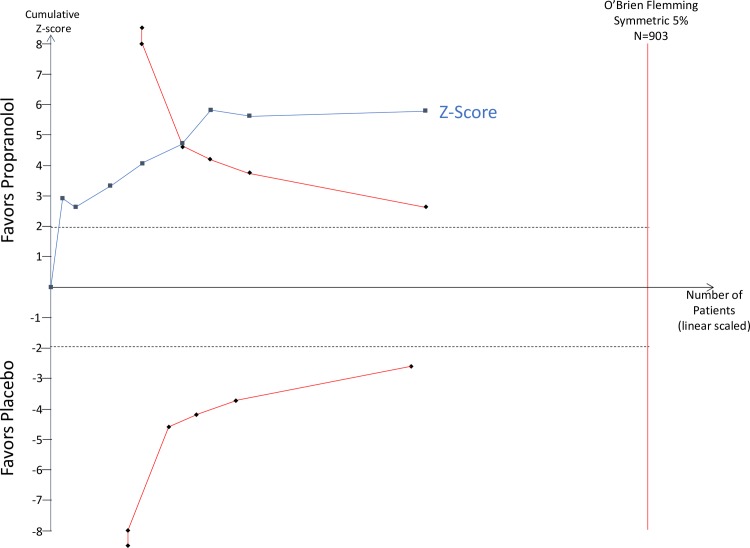
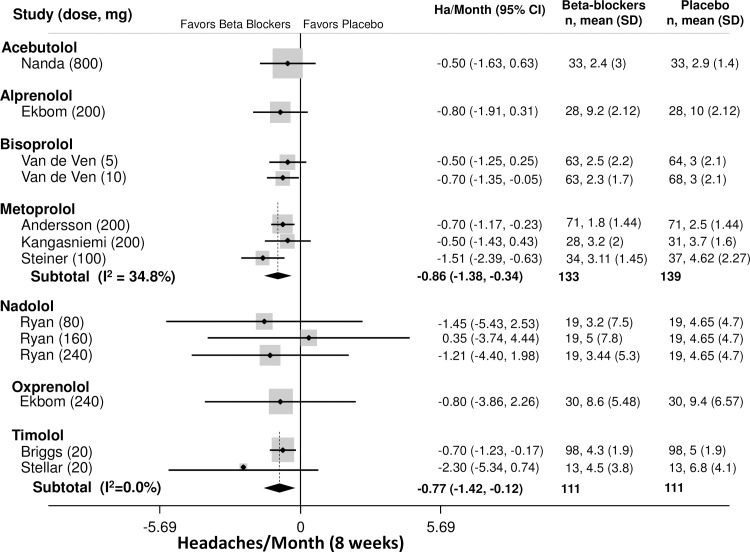
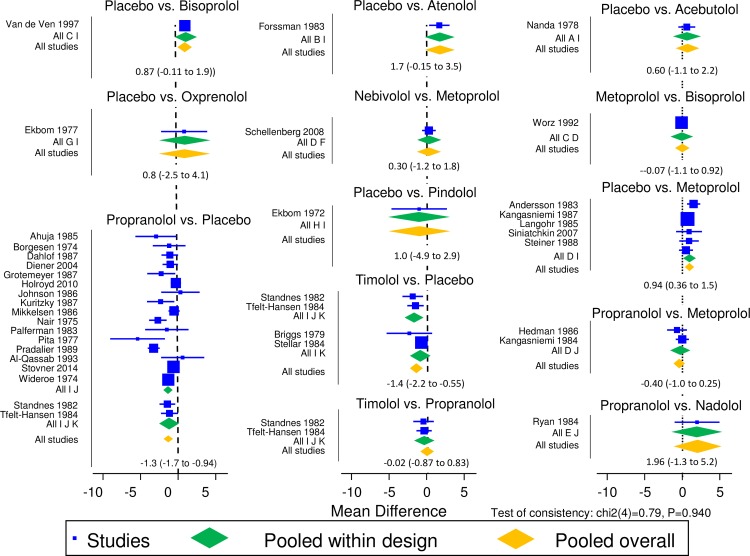
The primary outcome was headaches per month. Outcomes from placebo-controlled trials for all beta-blockers and time-points are provided in Tables 2–3. Outcomes at 8 weeks was the most commonly reported time point. Among patients with episodic migraines (Table 2), the average number of headaches at baseline was 4.9 headaches/month (95% CI: 4.4–5.4). The best studied beta-blocker was propranolol, which was more effective than placebo at 8 and 12 weeks (8 weeks: -1.5 ha/month, 95% CI: -2.3 to -0.65); 12 weeks: -1.2 ha/month, 95% CI: -1.8 to -0.60, Fig 2). Propranolol outcomes at 8 and 12 weeks were both graded as high-quality evidence. TSA analysis of propranolol vs. placebo for headache frequency demonstrated that it is unlikely that these results are due to a Type 1 error (Fig 3). Other beta-blockers that were more effective than placebo at 8 weeks (Fig 4) included bisoprolol (-0.70 ha/month, 95% CI: -1.4 to -0.05, low quality), metoprolol (-0.86 ha/month, 95% CI: -1.4 to -0.34, moderate quality) and timolol (-0.77 ha/month, 95% CI: -1.4 to -0.12, moderate quality). The remaining beta-blockers, in single trials did not significantly reduce headache frequency (Fig 3). There was a similar pattern at twelve weeks.
Table 2. Placebo controlled primary outcome (headache frequency per month).
| Episodic Migraines | |||
|---|---|---|---|
| Time point | Weighted Mean Difference (95% CI) | Heterogeneity | Quality of Evidence (GRADE) |
| Acebutolol (HA frequency at baseline: 4.8 headaches/month (95% CI: -0.64 to 8.9)) | |||
| Baseline Frequency | 0.00 (-0.14 to 0.14) | — | Low |
| 4 weeks | -0.20 (-0.38 to -0.02) | — | |
| 8 weeks | -0.50 (-1.6 to 0.63) | — | |
| 12 weeks | -0.6 (-1.7 to 0.53) | — | |
| Alprenolol | |||
| 8 weeks | -0.80 (-1.9 to 0.31) | — | Low |
| Atenolol | |||
| 12 weeks | -1.7 (-3.0 to -0.32) | — | Low |
| Bisoprolol (HA frequency at baseline: 5.5 headaches/month (95% CI: 2.7 to 8.3)) | |||
| Baseline | Low | ||
| 5 mg | 0.40 (-0.19 to 0.99) | — | |
| 10 mg | 0.20 (-0.40 to 0.80) | — | |
| 4 weeks | |||
| 5 mg | -0.40 (-1.1 to 0.29) | — | |
| 10 mg | -0.40 (-1.1 to 0.34) | — | |
| 8 weeks | |||
| 5 mg | -0.50 (-1.2 to 0.25) | — | |
| 10 mg | -0.70 (-1.4 to -0.05) | — | |
| 12 weeks | |||
| 5 mg | -0.90 (-1.53 to -0.27) | — | |
| 10 mg | -0.90 (-1.6 to -0.24) | — | |
| Metoprolol (HA frequency at baseline: 3.9 headaches/month (3.1 to 4.7)) | |||
| Baseline | -0.04 (-0.49 to 0.41) | Q = 3.68, df = 3, I2 = 18.5% | Low |
| 4 weeks | -0.91 (-2.6 to 0.82) | — | — |
| 8 weeks | -0.86(-1.4 to -0.34) | Q = 3.07, df = 2, I2 = 34.8% | Moderate |
| 12 weeks | -0.90 (-2.2 to 0.41) | — | Low |
| Nadolol (HA frequency at baseline: 6.7 headaches/month (3.4 to 9.9) | |||
| Baseline | 0.22 (-1.8 to 2.3) | — | Low |
| 4 weeks | 1.1 (-0.98 to 3.2) | — | |
| 8 weeks | -0.86 (-2.9 to 1.3) | — | |
| 12 weeks | -0.96 (-3.1 to 1.2) | — | |
| Oxprenolol | |||
| 8 weeks | -0.80 (-3.9 to 2.3) | — | Low |
| Propranolol (HA frequency at baseline: 4.8 headaches/month (4.3 to 5.3))— | |||
| Baseline | -0.04 (-0.28 to 0.20) | Q = 0.83, df = 10, I2 = 0.0% | High |
| 4 weeks | -1.1 (-1.8 to -0.43) | Q = 0.0, df = 1, I2 = 0.0% | Moderate |
| 8 weeks | -1.5 (-2.3 to -0.65) | Q = 11.37, df = 7, I2 = 38.4% | High |
| 12 weeks | -1.2 (-1.8 to-0.60) | Q = 35.29, df = 8, I2 = 77.3% | High |
| 16 weeks | -1.2 (-2.4 to -0.01) | — | Low |
| 20 weeks | -0.9 (-1.8 to -0.02) | — | Low |
| 24 weeks | -0.9 (-1.5 to -0.32) | — | Low |
| 40 weeks | -0.3 (-0.9 to 0.34) | — | Low |
| 64 weeks | -0.3 (-0.98 to 0.38) | — | Low |
| Timolol (HA frequency at baseline: 4.8 headaches/month (95% CI: -0.64 to 8.9)) | |||
| Baseline | (-0.45 to 0.45) | Q = 0.0, df = 2, I2 = 0.0% | Moderate |
| 8 weeks | -0.77 (-1.4 to -0.12) | Q = 1.03, df = 1, I2 = 3.2% | |
| 12 weeks | -1.53 (-2.5 to -0.78) | Q = 0.16, df = 1, I2 = 0.0% | |
| Migraine-Chronic | |||
| Propranolol | |||
| 8 weeks | -2.1 (-5.5 to 1.3) | — | Low |
| Chronic Tension-type HA | |||
| Pindolol+ Amitriptyline (HA frequency at baseline: 20.0 headaches/month (95% CI: 18.5 to 29.5)) | |||
| Baseline | 1.4 (-2.2 to 5.0) | — | Low |
| 4 weeks | -7.8 (-13.9 to -1.5) | — | |
| 8 weeks | -11.2 (-16.7 to -5.5) | — | |
| Propranolol (HA frequency at baseline: 20.0 headaches/month (95% CI: 18.5 to 29.5)) | |||
| Baseline | (-8.1 to 8.1) | — | Low |
| 8 weeks | -4.5 (-8.2 to -0.82) | — | |
Table 3. Secondary outcomes of placebo controlled trials.
| Episodic Migraines | |||
| 50% Improvement in Headaches | |||
| Beta-blocker | RR (95% CI)/NNT | Heterogeneity | Quality of Evidence |
| Atenolol | 1.8 (1.0 to 3.2)/6.3 (3.2–33.3) | — | Low |
| Metoprolol | 1.7 (1.0 to 2.9)/5 (3.5–8.8) | Q = 8.85, df = 3, I2 = 66.1% | Moderate |
| Nadolol | 5.1 (0.32 to 81.3)/3.7 (1.9–90.9) | — | Low |
| Propranolol | 1.4 (1.1 to 1.8)/5.3 (3.4–11.4) | Q = 26.1, df = 10, I2 = 59.5% | High |
| Timolol | 1.8 (1.4 to 2.3)/4.5 (3.1–7.7) | Q = 0.86, df = 2, I2 = 0.0% | Moderate |
| Analgesic Medication Consumption | |||
|
Beta-blocker Time point |
Weighted Mean Difference (95% CI) | Heterogeneity | |
| Metoprolol (Baseline analgesic doses/month: 6.7, 95% CI: 3.4 to 10.0) | |||
| Baseline | -0.17 (-1.4 to 1.1) | Q = 0.5, df = 3, I2 = 0.0% | High |
| 4 weeks | -2.4 (-4.9 to 0.08) | — | Low |
| 8 weeks | -4.0 (-7.5 to -0.48) | Q = 3.49, df = 2, I2 = 42.8% | Moderate |
| Propranolol (Baseline analgesic doses/month 11.1, 95% CI: 4.8–17.4) | |||
| Baseline | 0.0 (-1.9 to 1.9) | Q = 0.26, df = 7, I2 = 0.00 | High |
| 4 weeks | -6.0 (-11.8 to -0.12) | — | Low |
| 8 weeks | -2.9 (-25.9 to 20.2) | — | Low |
| 12 weeks | -2.1 (-3.2 to -0.95) | Q = 33.82, df = 5, I2 = 85.2% | High |
| Headache Index | |||
|
Beta-blocker Time point |
Standardized Mean Difference (95% CI) | Heterogeneity | |
| Alprenolol | |||
| 8 weeks | 0.05 (-0.47 to 0.58) | — | Low |
| Atenolol | |||
| 8 weeks | -0.62 (-1.2 to -0.004) | — | Low |
| 12 weeks | -0.65 (-1.3 to -0.01) | — | |
| Metoprolol | |||
| Baseline | 0.12 (-0.22 to 0.47) | Q = 0.07, df = 1, I2 = 0.0% | Moderate |
| 8 weeks | -0.42 (-0.77 to -0.07) | Q = 0.98, df = 1, I2 = 0.0% | |
| Nadolol | |||
| Baseline | 0.12 (-0.24 to 0.48) | — | Low |
| 4 weeks | 0.16 (-0.20 to 0.52) | — | |
| 8 weeks | -0.14 (-0.50 to 0.23) | — | |
| 12 weeks | -0.27 (-0.64 to 0.10) | — | |
| Oxprenolol | |||
| 4 weeks | -1.7 (-2.3 to -1.1) | — | Low |
| 8 weeks | -0.42 (-0.09 to 0.93) | — | |
| Pindolol | |||
| Baseline | 0.14 (-0.52 to 0.80) | — | Low |
| 4 weeks | 0.27 (-0.40 to 0.93) | — | |
| Propranolol | |||
| 0 weeks | -0.03 (-0.23 to 0.16) | Q = 6.24, df = 6, I2 = 19.9% | High |
| 4 weeks | -0.66 (-1.3 to -0.01) | — | Low |
| 8 weeks | -0.48 (-0.75 to -0.22) | Q = 2.54, df = 4, I2 = 0.0% | High |
| 12 weeks | -0.41 (-0.65 to -0.17) | Q = 2.56, df = 3, I2 = 0.0% | High |
| Timolol | |||
| Baseline | 0.0 (-0.31 to 0.31) | — | Low |
| 12 weeks | -0.53 (-0.84 to -0.21) | — | |
| Headache Severity | |||
| Metoprolol | |||
| Baseline | -0.06 (-0.33 to 0.21) | Q = 1.65, df = 2, I2 = 0.0% | High |
| 8 weeks | -0.53 (-0.71 to -0.14) | Q = 0.15, df = 1, I2 = 0.0% | Moderate |
| Propranolol | |||
| Baseline | (-0.21 to 0.21) | Q = 0.00, df = 2, I2 = 0.0% | High |
| 8 weeks | -0.51 (-0.76 to -0.26) | Q = 1.73, df = 4, I2 = 0.0% | |
| 12 weeks | 0.18 (-0.30 to 0.01) | Q = 3.70, df = 2, I2 = 46.0% | |
| Timolol | |||
| Baseline | 0.00 (-0.31 to 0.31) | — | Low |
| 12 weeks | -0.35 (-0.66 to -0.04) | — | |
| Headache Duration (hours) | |||
|
Beta-blocker Time point |
Weighted Mean Difference (95% CI) | Heterogeneity | |
| Bisoprolol (baseline duration 20.6 hours, 95% CI: 0 to 46.7) | |||
| Baseline | -3.9 (-8.6 to 0.77) | — | Low |
| 12 weeks | -1.9 (-6.5 to 2.5) | — | |
| Metoprolol (baseline duration: 11.6 hours, 95% CI: 2.6 to 20.5) | |||
| Baseline | 0.06 (-1.6 to 1.7) | — | Low |
| 4 weeks | -2.6 (-4.2 to -0.88) | — | |
| 8 weeks | -2.0 (-3.7 to -0.26) | — | |
| Propranolol (baseline duration: 28.9 hours, 95% CI: 0 to 72.3) | |||
| Baseline | 0.22 (-2.4 to 2.8) | Q = 0.99, df = 2, I2 = 0.0% | High |
| 8 weeks | -6.1 (-16.2 to -0.39) | Q = 5.67, df = 2, I2 = 64.7% | |
| 12 weeks | -1.6 (-3.0 to -0.11) | Q = 2.50, df = 4, I2 = 0.0% | |
| Pindolol (baseline duration: 6.6 hours, 95% CI: 0 to 23.7) | |||
| Baseline | -0.93 (-9,7 to 7.8) | Q = 0.01, df = 1, I2 = 0.0% | Moderate |
| 4 weeks | -0.18 (-8.8 to 8.4) | Q = 0.01, df = 1, I2 = 0.0% | |
| Timolol (baseline duration 9.4 hours, 95% CI: 0 to 19.8) | |||
| Baseline | (-1.3 to 1.3) | — | Low |
| 8 weeks | -0.70 (-2.2 to 0.80) | — | |
| 12 weeks | -0.54 (-2.7 to 1.6) | — | |
| Tension-type Headache | |||
| 50% improvement in Headache | |||
| Beta-blocker | RR (95% CI) | Heterogeneity | |
| Pindolol + Amitriptyline | 3.8 (1.5 to 9.3) | — | Low |
| Headache Index | |||
| Propranolol | |||
| 4 weeks | -0.52 (-1.0 to -0.003) | — | Low |
| Severity | |||
| Pindolol + Amitriptyline | |||
| Baseline | -0.29 (-0.34 to 0.91) | — | Low |
| 8 weeks | -0.68 (-1.3 to -0.04) | — | |
Fig 2. Headaches per month, Propranolol v. Placebo.
Fig 3. Trial Sequential Analysis (Propranolol v. Placebo).
Fig 4. Headaches per month, other Beta Blockers v Placebo.
Among secondary outcomes, the majority of trials studied subjects with episodic migraine headaches (Table 3). Propranolol was the most commonly studied beta-blocker. Propranolol was more likely to reduce headaches by 50% than placebo at 12 weeks (RR: 1.4, 95% CI: 1.1–1.8, NNT: 4.5, 95% CI: 2.8–12.9). Other effective beta-blockers included atenolol (RR: 1.8, 95% CI: 1.0–3.2, NNT: 6.3, 95% CI: 3.2–332.4), metoprolol (RR: 1.9, 95% CI: 1.3–2.8, NNT: 4.7, 95% CI:3.0–10.4) and timolol (RR: 1.8, 95% CI: 1.4–2.3, NNT: 4.2, 95% CI: 2.7–8.8,). At 8 weeks, metoprolol reduced analgesic medication use (-4.0 doses/month, 95% CI: -7.5 to -0.48) as did propranolol at 12 weeks (-2.1 doses/month, 95% CI: -3.2 to -0.95). The headache index was modestly reduced by a number of different beta-blockers including atenolol (SMD: -0.62, 95% CI: -1.2 to -0.004), metoprolol (SMD: -0.42, 95% CI: -0.77 to -0.07), propranolol (SMD: -0.48, 95% CI: -0.75 to -0.22) and timolol (SMD: -0.53, 95% CI: -0.84 to -0.21). At 8 weeks, headache severity was modestly reduced by both metoprolol (SMD: -0.53, 95% CI: -0.71 to -0.14) and propranolol (SMD: -0.51, 95% CI: -0.76 to -0.26). Headache duration was reduced by metoprolol (-2.0 hours, 95% CI: -3.7 to -0.26) and propranolol (-6.1 hours, 95% CI: -16.2 to -0.39).
Network meta-analysis
For the primary outcome, headache frequency, the network meta-analysis found no difference at 8 weeks (p = 0.27) in effectiveness in comparisons between propranolol (n = 9) compared to bisoprolol (n = 2), metoprolol (n = 3) and nadolol (n = 3). Similarly, at 12 weeks, there was no difference (p = 0.84) n effectiveness in comparisons between propranolol (n = 9) compared to bisoprolol (n = 2), nadolol (n = 3) and timolol (n = 2). The 8- and 12-week analysis confirmed this lack of difference between all beta-blockers (Fig 5), including those with single trials (Fig 6)
Fig 5. Network meta-analysis map.
Fig 6. Results of network meta-analysis comparisons.
Comparative effectiveness trials
There were 83 randomized trials that included at least one comparison to a non-placebo treatment. Propranolol was the most commonly compared beta-blocker (n = 72, 87%). Propranolol was compared to pharmacologic interventions in 50 trials. Eleven comparisons that were from single trials (5-hydroxytryptophan, aspirin, atenolol, candesartan, clonidine, cyclandelate, mefenamic acid, naproxen, nifedipine, nimodipine, nortriptyline, pindolol, riboflavin). Comparisons with more than one study included: amitriptyline (n = 2), femoxetine (n = 2), flunarizine (n = 11), metoprolol (n = 4), nadolol (n = 3), nimodipine (n = 2), timolol (n = 2) tofenamic acid (n = 2), topiramate (n = 3) and valproate (n = 3). Several combinations were tested including propranolol + amitriptyline vs. amitriptyline (n = 1), and propranolol + flunarizine was compared to flunarizine. Propranolol combined with topiramate was compared with topiramate alone. Nonpharmacologic interventions were compared to propranolol in three trials (acupuncture, behavioral management, biofeedback). Metoprolol was assessed in 14 trials, 8 were comparisons with pharmacologic interventions (aspirin, bisoprolol, clomipramine, clonidine, flunarizine, nebivolol, pizotifen) and one with nonpharmacologic (acupuncture). Metoprolol was compared to a combination of metoprolol and fluoxetine in 2 trials and the combination of metoprolol and flunarizine was compared to flunarizine alone in 2 trials.
The primary outcome comparison (headache frequency) is provided in Table 4, and secondary outcomes are in Table 5. The majority of comparisons were single trials, making definitive conclusions difficult. Metoprolol, but not propranolol, was more effective than aspirin. Metoprolol was more effective than clomipramine, though comparable to acupuncture, bisoprolol, clonidine, flunarizine, nebivolol and pizotifen. Adding fluoxetine to metoprolol or flunarizine to either propranolol or metoprolol did not improve headache frequency. All comparisons were graded as low-quality. Propranolol was more effective than femoxetine, mefenamic acid, naproxen, nifedipine and very low-dose (40mg) nortriptyline, but comparable to 5-hydroxytryptophan, acupuncture, atenolol, behavioral management, biofeedback, candesartan, clonidine, cyclandelate, flunarizine, metoprolol, nadolol, naproxen, nimodipine, riboflavin, timolol, tolfenamic acid, topiramate and valproic acid. All comparisons were single trials and were rated as low-quality evidence with the exception of the comparisons to flunarizine and metoprolol at 8 weeks that were graded as moderate or high-quality. The network meta-analysis confirmed these findings, but suggested that metoprolol was also superior to naproxen (SMD: -1.2, 95% CI: -1.6 to -0.78).
Table 4. Primary outcome (headache frequency) of comparative effectiveness trials.
| Comparison, Time point | Weighted Mean Difference (95% CI) | Heterogeneity | Quality of Evidence | |
|---|---|---|---|---|
| Bisoprolol (5mg) | Bisoprolol (10mg) | |||
| Baseline | 0.20 (-0.43 to 0.83) | — | Low | |
| 4 Weeks | 0.0 (-0.74 to 0.74) | — | ||
| 8 weeks | 0.20 (-0.47 to -0.87) | — | ||
| 12 weeks | 0.0 (-0.67 to -0.67) | — | ||
| Metoprolol | Acupuncture | |||
| 17 weeks | -0.7 (-2.7 to 1.3) | — | Low | |
| Aspirin | ||||
| 8 weeks | -1.6 (-2.8 to -0.46) | — | Low | |
| Bisoprolol | ||||
| Baseline | 0.0 (-0.31 to 0.31) | — | Low | |
| 12 weeks | -0.09 (-0.62 to 0.44) | — | ||
| Clomipramine | ||||
| 4 weeks | -2.0 (-3.9 to -0.16) | — | Low | |
| Clonidine | ||||
| Baseline | 0.00 (-0.51 to 0.51) | — | Low | |
| 8 weeks | -1.40 (-3.3 to 0.44) | — | ||
| Flunarizine | ||||
| Baseline | -0.60 (-1.4 to 0.19) | — | Low | |
| 4 weeks | -0.3 (-1.1 to 0.53) | — | ||
| 8 weeks | -0.9 (-1.7 to -0.10) | — | ||
| 12 weeks | -0.5 (-1.3 to 0.33) | — | ||
| 16 weeks | -0.1 (-0.93 to 0.73) | — | ||
| 20 weeks | -0.36 (-1.5 to 0.75) | — | ||
| Metoprolol + Fluoxetine | ||||
| Baseline | -0.03 (-0.40 to 0.33) | — | Low | |
| 8 weeks | 0.30 (0.19 to 0.40) | — | ||
| Nebivolol | ||||
| Baseline | 0.10 (-0.62 to 0.82) | — | Low | |
| 16 weeks | -0.30 (-1.2 to 0.60) | — | ||
| Pizotifen | ||||
| Baseline | 0.00 (-0.47 to 0.47) | — | Low | |
| 8 weeks | 0.90 (-0.01 to 1.8) | — | ||
| Metoprolol + Flunarizine | Flunarizine | |||
| 12 weeks | -0.9 (-1.6 to -0.22) | — | Low | |
| 24 weeks | -0.8 (-1.6 to -0.04) | — | ||
| 36 weeks | -0.6 (-2.4 to 1.2) | — | ||
| 48 weeks | -0.3 (-1.2 to 0.57) | — | ||
| Nadolol (80mg) | Nadolol (160mg) | |||
| Baseline | 0.57 (-2.1 to 3.3) | — | Low | |
| 4 weeks | -0.25 (-2.9 to 2.4) | — | ||
| 8 weeks | 0.11 (-2.6 to 2.8) | — | ||
| 12 weeks | -0.34 (-3.1 to 2.4) | — | ||
| Pindolol | Pindolol (15mg) | |||
| Baseline | 4.0 (-0.6 to 8.1) | — | Low | |
| 4 weeks | 4.0 (-0.05 to 8.1) | — | ||
| Propranolol | 5-Hydroxy-tryptophan | |||
| Baseline | -2.3 (-5.9 to 1.3) | — | Low | |
| 16 weeks | -2.9 (-6.6 to 0.81) | — | ||
| Acupuncture | ||||
| Baseline | -0.10 (-0.59 to 0.40) | — | Low | |
| 12 weeks | 0.30 (-0.25 to 0.85) | — | ||
| Behavioral Management | ||||
| Baseline | -0.40 (-1.1 to 0.34) | — | Low | |
| 40 weeks | -0.30 (-0.94 to 0.34) | — | ||
| 64 weeks | -0.20 (-0.89 to 0.49) | — | ||
| Propranolol + Behavioral Management | ||||
| Baseline | -0.50 (-1.2 to 0.18) | — | Low | |
| 40 weeks | 0.70 (-.16 to 1.2) | — | ||
| 64 weeks | 0.80 (0.19 to 1.4) | — | ||
| Biofeedback | ||||
| 24 weeks | 0.53 (0.08 to 0.97) | — | Low | |
| Candesartan | ||||
| 12 weeks | -0.04 (-0.59 to 0.51) | — | Low | |
| Cyclandelate (HA days/mo) | ||||
| Baseline | -0.16 (-0.66 to 0.35) | — | Low | |
| 4 weeks | -0.32 (-0.83 to 0.18) | — | ||
| 8 weeks | 0.13 (-0.37 to 0.63) | — | ||
| Femoxetine | ||||
| Baseline | 0.0 (-2.2 to 2.2) | — | Low | |
| 8 weeks | -0.70 (-1.5 to 0.13) | — | ||
| 12 weeks | -1.5 (-3.6 to 0.55) | — | ||
| Flunarizine | ||||
| Baseline | -0.005 (-0.11 to 0.10) | Q = 0.83, df = 5, I2 = 0.0% | High | |
| 4 weeks | 0.40 (-0.34 to 1.1) | Q = 3.1, df = 3, I2 = 3.7% | High | |
| 8 weeks | 0.42 (-0.55 to 1.4) | Q = 1.48, df = 1, I2 = 32.6% | Moderate | |
| 12 weeks | 0.68 (-0.06 to 1.4) | Q = 0.55, df = 2, I2 = 0.0% | High | |
| 16 weeks | -0.04 (-0.19 to 0.12) | Q = 5.44, df = 6, I2 = 7.0% | High | |
| Mefenamic Acid | ||||
| 12 weeks | -2.1 (-5.3 to 1.3) | — | Low | |
| Metoprolol | ||||
| Baseline | 0.00 (-0.38 to 0.38) | — | Low | |
| 4 weeks | -0.70 (-2.0 to 0.62) | — | ||
| 8 weeks | 0.00 (-0.54 to 0.56) | Q = 0.0, df = 1, I2 = 0.0% | ||
| Nadolol | ||||
| Baseline | 0.27 (-2.6 to 3.2) | — | Low | |
| 4 weeks | 1.6 (-0.28 to 3.4) | — | ||
| 8 weeks | 1.7 (-0.23 to 3.6) | — | ||
| 12 weeks | 1.8 (-0.16 to 3.7) | — | ||
| 24 weeks | -4.8 (-8.9 to -0.77) | — | ||
| Naproxen | ||||
| 12 weeks | -1.4 (-1.9 to -0.95) | — | Low | |
| Nifedipine | ||||
| Baseline | 2.5 (1.0 to 3.9) | — | Low | |
| 12 weeks | -0.40 (-2.6 to 1.8) | — | ||
| 24 weeks | 0.70 (-1.5 to 2.9) | — | ||
| Nimodipine | ||||
| Baseline | 0.20 (-0.86 to 1.2) | — | Low | |
| 4 weeks | 1.6 (-0.24 to 3.4) | — | ||
| 8 weeks | -1.5 (-4.1 to 1.1) | — | ||
| 12 weeks | -1.5 (-2.9 to -0.04) | — | ||
| 16 weeks | -0.30 (-1.7 to 1.1) | — | ||
| 24 weeks | -0.01 (-1.3 to 1.3) | — | ||
| Propranolol + Flunarizine | ||||
| Baseline | -0.14 (-1.8 to 1.5) | — | Low | |
| 16 weeks | 0.18 (-1.4 to 1.8) | — | ||
| Riboflavin | ||||
| Baseline | 0.00 (-0.39 to 0.39) | — | Low | |
| 4 weeks | -0.60 (-0.92 to -0.28) | — | ||
| 8 weeks | -0.10 (-0.42 to 0.22) | — | ||
| 12 weeks | 0.0 (-0.27 to 0.27) | — | ||
| 24 weeks | -0.10 (-0.37 to 0.17) | — | ||
| Timolol | ||||
| Baseline | 0.00 (-0.84 to 0.84) | Q = 0.00, df = 1 I2 = 0.0% | Moderate | |
| 12 weeks | 0.37 (-0.45 to 1.2) | Q = 0.01, df = 1, I2 = 0.0% | ||
| Tolfenamic Acid | ||||
| 12 weeks | 0.01 (-0.68 to 0.70) | — | Low | |
| Topiramate | ||||
| Baseline | -0.02 (-0.35 to 0.31) | Q = 1.47, df = 2 I2 = 0.0% | Moderate | |
| 4 weeks | -0.19 (-0.76 to 0.39) | Q = 0.18, df = 1 I2 = 0.0% | ||
| 8 weeks | 0.30 (-0.30 to 0.91) | — | Low | |
| 12 weeks | 0.10 (-0.98 to 1.2) | — | ||
| 16 weeks | 0.30 (-0.94 to 1.5) | — | ||
| 20 weeks | 0.40 (-0.60 to 1.4) | — | ||
| 24 weeks | -0.75 (-1.6 to 0.13) | Q = 4.38, df = 1 I2 = 77.2% | ||
| Valproic Acid | ||||
| Baseline | -0.20 (-1.1 to 0.71) | — | Low | |
| 8 weeks | -0.16 (-1.7 to 0.75) | — | ||
| Propranolol + Flunarizine | Topiramate | |||
| Baseline | -0.06 (-0.82 to 0.70) | — | Low | |
| 12 weeks | 0.70 (0.29 to 1.1) | — | ||
| Propranolol + Topiramate | Topiramate | |||
| 24 weeks | -0.01 (-2.4 to 2.1) | — | Low | |
Table 5. Secondary outcomes of comparative effectiveness trials.
| Drug1 | Comparison, Time Point | Weighted Mean Difference (95% CI) | Heterogeneity | Quality of Evidence |
|---|---|---|---|---|
| Headache Days Weighted Mean Difference (95% CI) | ||||
| Metoprolol | Clonidine | |||
| Baseline | 0.0 (-3.7 to 3.7) | — | Low | |
| 8 Weeks | -1.8 (-5.7 to 2.14) | — | ||
| Metoprolol + Fluoxetine | ||||
| 8 weeks | 0.83 (0.63 to 1.0) | — | Low | |
| Nadolol (80mg) | Nadolol (160mg) | |||
| Baseline | 2.3 (-3.4 to 7.9) | — | Low | |
| 4 weeks | -0.22 (-5.9 to 5.4) | — | ||
| 8 weeks | -0.05 (-5.9 to 5.8) | — | ||
| 12 weeks | 2.2 (-7.9 to 3.6) | — | ||
| Pindolol (7.5mg) | Pindolol (15mg) | |||
| Baseline | 0.05 (-0.94 to 1.0) | — | Low | |
| 4 weeks | 0.28 (-0.72 to 1.3) | — | ||
| Propranolol | Acupuncture | |||
| Baseline | 0.0 (-1.0 to 1.0) | — | Low | |
| 12 weeks | 0.70 (-0.43 to 1.8) | — | ||
| Atenolol | ||||
| 8 weeks | 0.06 (-1.8 to 1.9) | — | Low | |
| Behavioral Management | ||||
| Baseline | 0.50 (-0.88 to 1.9) | — | Low | |
| 40 weeks | -0.10 (-1.3 to 1.1) | — | ||
| 64 weeks | 0.10 (-1.1 to 1.3) | — | ||
| Candesartan | ||||
| 12 weeks | 0.45 (-1.2 to 2.1) | — | Low | |
| Clonidine | ||||
| 16 weeks | 0.10 (-2.1 to 2.3) | — | Low | |
| Cyclandelate | ||||
| Baseline | -0.60 (-2.5 to 1.3) | — | Low | |
| 4 weeks | -1.1 (-2.8 to 0.64) | — | ||
| 8 weeks | 0.40 (-1.2 to 2.0) | — | ||
| Femoxetine | ||||
| 8 weeks | -1.6 (-4.5 to 1.3) | — | Low | |
| Flunarizine | ||||
| Baseline | -0.80 (-2.2 to 0.59) | — | Low | |
| 4 weeks | 1.4 (0.02 to 2.8) | — | ||
| 8 weeks | 0.50 (-0.91 to 1.9) | — | ||
| 12 weeks | -0.5 (-1.9 to 0.96) | — | ||
| 16 weeks | 0.61 (-0.91 to 2.1) | — | ||
| Metoprolol | ||||
| Baseline | -0.31 (-1.3 to 0.68) | — | Low | |
| 4 weeks | -0.60 (-4.3 to 3.1) | — | Low | |
| 8 weeks | -0.17 (-0.96 to 0.51) | Q = 0.73 df = 1, I2 = 0.0% | Moderate | |
| 16 weeks | 0.06 (-1.9 to 2.02) | — | Low | |
| 24 weeks | -2.2 (-4.2 to -0.24) | — | Low | |
| Naproxen | ||||
| 12 weeks | -2.8 (-3.6 to -1.9) | — | Low | |
| Nifedipine | ||||
| Baseline | -0.80 (-3.3 to 1.7) | — | Low | |
| 4 weeks | -3.6 (-7.0 to -0.16) | — | ||
| 16 weeks | -2.2 (-4.0 to -0.32) | — | ||
| 28 weeks | -0.80 (-2.6 to 1.0) | — | ||
| Propranolol (80mg v 160mg) | ||||
| Baseline | 0.0 (-1.7 to 1.7) | — | Low | |
| 12 weeks | -2.7 (04.9 to -0.5) | — | ||
| Tolfenamic Acid | ||||
| Baseline | 0.0 (-1.6 to 1.6) | — | Low | |
| 12 weeks | -0.22 (-2.1 to 1.6) | — | ||
| Topiramate | ||||
| Baseline | 0.12 (-0.3 to 0.55) | — | Low | |
| 24 weeks | -0.25 (01.13 to 0.63) | — | ||
| Headache Index, Standardized Mean Difference (95% CI) | ||||
| Metoprolol | Clonidine | |||
| Baseline | 0.0 (-0.49 to 0.49) | — | Low | |
| 8 Weeks | -0.24 (-0.76 to 0.29) | — | ||
| Pindolol (7.5mg) | Pindolol (15 mg) | |||
| Baseline | 0.05 (-0.94 to 1.04) | — | Low | |
| 4 weeks | 0.27 (-0.72 to 1.3) | — | ||
| Propranolol | Amitriptyline | |||
| Baseline | 0.05 (-0.42 to 0.52) | — | Low | |
| 6 weeks | -0.42 (-0.89 to 0.06) | — | ||
| Amitriptyline + Biofeedback | ||||
| Baseline | 0.08 (-0.37 to 0.53) | — | Low | |
| 6 weeks | -0.24 (-0.69 to 0.21) | — | ||
| Amitriptyline + Propranolol | ||||
| Baseline | 0.01 (-0.44 to 0.46) | — | Low | |
| 6 weeks | 0.06 (-0.39 to 0.51) | — | ||
| Amitriptyline + Propranolol + Biofeedback | ||||
| Baseline | -0.04 (-0.52 to 0.44) | — | Low | |
| 6 weeks | 0.25 (-0.23 to 0.74) | — | ||
| Biofeedback | ||||
| Baseline | 0.17 (-0.31 to 0.64) | — | Low | |
| 6 weeks | -0.35 (-0.83 to 0.13) | — | ||
| Biofeedback + Propranolol | ||||
| Baseline | -0.03 (-0.50 to 0.44) | — | Low | |
| 6 weeks | 0.39 (-0.09 to 0.86) | — | ||
| Aspirin | ||||
| Baseline | 0.00 (-0.65 to 0.65) | — | Low | |
| 6 weeks | 0.25 (-0.56 to 1.05) | — | ||
| Atenolol | ||||
| 6 weeks | 0.01 (-0.59 to 0.62) | — | Low | |
| Femoxetine | ||||
| Baseline | 0.00 (-0.57 to 0.57) | — | Low | |
| 8 weeks | -0.24 (-0.70 to 0.21) | — | ||
| 12 weeks | -0.35 (-0.92 to 0.22) | — | ||
| Flunarizine | ||||
| Baseline | 0.12 (-0.17 to 0.42) | Q = 1.59, df = 2, I2 = 0.0% | Moderate | |
| 4 weeks | -0.18 (-0.82 to 0.46) | Q = 2.45, df = 2, I2 = 59.3% | ||
| 8 weeks | -0.13 (-0.75 to 0.48) | Q = 2.27, df = 1, I2 = 55.9% | ||
| 12 weeks | 0.16 (-1.1 to 1.4) | Q = 8.7, df = 1, I2 = 88.5% | ||
| 16 weeks | -0.08 (-0.48 to 0.32) | Q = 3.13, df = 2, I2 = 49.3% | ||
| Flunarizine + Propranolol | ||||
| Baseline | 0.42 (-0.30 to 1.1) | — | Low | |
| 4 weeks | 0.29 (-0.43 to 1.0) | — | ||
| 8 weeks | 0.17 (-0.54 to 0.89) | — | ||
| 12 weeks | 0.47 (-0.26 to 1.2) | — | ||
| 16 weeks | 0.67 (-0.07 to 1.4) | — | ||
| Metoprolol | ||||
| Baseline | 0.00 (-0.29 to 0.29) | Q = 0.00, df = 1, I2 = 0.0% | Moderate | |
| 8 weeks | 0.06 (-0.24 to 0.35) | Q = 0.23, df = 1, I2 = 0.0% | ||
| Nadolol | ||||
| Baseline | 0.41 (-0.09 to 0.90) | — | Low | |
| 4 weeks | 0.34 (-0.16 to 0.83) | — | ||
| 8 weeks | 0.44 (-0.08 to 0.96) | — | ||
| 12 weeks | 0.29 (-0.22 to 0.81) | — | ||
| Timolol | ||||
| Baseline | 0.00 (-0.31 to 0.31) | — | Low | |
| 12 weeks | 0.17 (-0.14 to 0.48) | — | ||
| Valproic Acid | ||||
| Baseline | -0.09 (-0.56 to 0.38) | |||
| 10 weeks | -0.03 (-0.50 to 0.44) | — | Low | |
| Propranolol + Flunarizine | Topiramate | |||
| Baseline | 0.07 (-0.36 to 0.57) | — | Low | |
| 12 weeks | 0.99 (0.53 to 1.4) | — | ||
| Episodic Migraine, 50% reduction in Headaches | ||||
| Comparison | RR (95% CI) | Heterogeneity | Quality | |
| Metoprolol | ASA | 2.4 (0.88 to 6.7) | — | Low |
| Clonidine | 1.3 (0.62 to 2.9) | — | Low | |
| Flunarizine | 1.1 (0.98 to 1.3) | — | Low | |
| Nebivolol | 1.1 (0.56 to 2.2) | — | Low | |
| Pizotifen | 0.69 (0.36 to 1.3) | — | Low | |
| Metoprolol + Flunarizine | Flunarizine | 1.3 (1.0 to 1.6) | — | Low |
| Propranolol | ASA | 1.3 (0.88 to 1.9) | Q = 2.44, df = 1, I2 = 59.1% | Moderate |
| Acupuncture | 0.88 (0.57 to 1.3) | — | Low | |
| Candesartan | 0.93 (0.61 to 1.4) | — | Low | |
| Clonidine | 1.6 (0.86 to 3.1) | — | Low | |
| Cyclandelate | 1.0 (0.71 to 1.5) | Q = 4.39, df = 2, I2 = 54.4% | Moderate | |
| Femoxetine | 3.5 (0.43 to 29.4) | — | Low | |
| Flunarizine | 1.0 (0.89 to 1.2) | Q = 3.30, df = 5, I2 = 0.0% | High | |
| Flunarizine + Propranolol | 0.86 (0.59 to 1.2) | — | Low | |
| Metoprolol | 0.86 (0.60 to 1.2) | Q = 0.11, df = 1, I2 = 0.0% | Moderate | |
| Nadolol | 0.66 (0.27 to 1.6) | Q = 6.20, df = 2, I2 = 67.8% | Moderate | |
| Nifedipine | 2.2 (1.3 to 3.8) | — | Low | |
| Nortriptyline | 1.5 (0.54 to 4.2) | — | Low | |
| Nortriptyline + Propranolol | 1.1 (0.48 to 2.7) | — | Low | |
| Propranolol (80 vs. 160mg) | 0.94 (0.72 to 1.2) | — | Low | |
| Timolol | 1.1 (0.84 to 1.4) | — | Low | |
| Topiramate | 1.2 (0.98 to 1.4) | Q = 0.05, df = 2 I2 = 0.0% | Moderate | |
| Valproic Acid | 0.96 (0.77 to 1.2) | Q = 0.16, df = 2, I2 = 0.0% | Moderate | |
| Propranolol + Amitriptyline | Amitriptyline | 1.02 (0.92 to 1.1) | — | Low |
| Propranolol + Flunarizine | Nimodipine + Diazepam + Oryzanol | 1.5 (1.2 to 2.0) | — | Low |
| Propranolol + Nadolol | Behavioral Management | 0.98 (0.58 to 1.7) | — | Low |
| Propranolol + Nadolol | Behavioral Management + Propranolol | 0.44 (0.30 to 0.66) | — | Low |
| Medicine Use (doses/month) | ||||
| Comparison | Weighted Mean Difference (95% CI) | Heterogeneity | Quality of evidence | |
| Metoprolol | Bisoprolol | |||
| Baseline | 0.0 (-0.31 to 0.31) | — | Low | |
| 12 weeks | 0.01 (-0.30 to 0.32) | — | ||
| Clomipramine | ||||
| 4 weeks | -0.55 (-1.2 to 0.08) | — | Low | |
| Clonidine | ||||
| Baseline | 0.0 (-0.50 to 0.50) | — | Low | |
| 8 weeks | -0.52 (-1.0 to 0.02) | — | ||
| Propranolol | 5-Hydroxytryptophan | |||
| Baseline | 0.48 (-0.16 to 1.1) | — | Low | |
| 16 weeks | 0.22 (-0.41 to 0.85) | — | ||
| Candesartan | ||||
| 12 weeks | -0.45 (-0.82 to -0.09) | — | Low | |
| Cyclandelate | ||||
| Baseline | -0.49 (-1.0 to 0.02) | — | Low | |
| 4 weeks | -0.70 (-1.2 to -0.19) | — | ||
| 8 weeks | -0.19 (-0.69 to 0.32) | — | ||
| Femoxetine | ||||
| Baseline | 0.00 (-0.57 to -0.57) | — | Low | |
| 12 weeks | -0.83 (-1.4 to -0.24) | — | ||
| Flunarizine | ||||
| Baseline | 0.08 (-0.10 to 0.26) | Q = 1.98, df = 2, I2 = 0.0% | Moderate | |
| 4 weeks | -.44 (0.02 to 0.86) | — | Low | |
| 8 weeks | -0.15 (-0.57 to 0.28) | — | Low | |
| 12 weeks | -0.23 (-0.43 to -0.04) | Q = 0.06, df = 1, I2 = 0.0% | Moderate | |
| 16 weeks | -0.07 (-0.41 to 0.27) | Q = 0.01, df = 1, I2 = 0.0% | Moderate | |
| Metoprolol | ||||
| Baseline | 0.00 (-0.29 to 0.29) | Q = 0.00, df = 1, I2 = 0.0% | Moderate | |
| 8 weeks | -0.19 (-0.49 to 0.11) | Q = 1.00, df = 1, I2 = 0.5% | ||
| Nadolol | ||||
| Baseline | -0.31 (-1.1 to 0.45) | — | Low | |
| 24 weeks | -0.73 (-1.5 to 0.05) | — | ||
| Health Related Quality of Life | ||||
| Comparison | Standardized Mean Difference (95% CI) | Heterogeneity | Quality of evidence | |
| Metoprolol | Nebivolol (MOS SF36) | |||
| Baseline | -0.21 (-0.93 to 0.51) | — | Low | |
| 16 Weeks | -0.46 (-1.2 to 0.27) | — | ||
| Propranolol | Acupuncture (MOS SF36) | |||
| Baseline | -0.19 (-0.56 to 0.19) | — | Low | |
| 12 weeks | -0.47 (-0.84 to -0.10) | — | ||
| Behavioral Management | ||||
| Baseline | 0.14 (-0.24 to 0.52) | — | Low | |
| 40 weeks | 0.48 (0.10 to 0.86) | — | ||
| 64 weeks | 0.23 (-0.15 to 0.60) | — | ||
| Behavioral Management + Propranolol | ||||
| Baseline | 0.07 (-0.28 to 0.43) | — | Low | |
| 40 weeks | 0.68 (0.31 to 1.0) | — | ||
| 64 weeks | 0.61 (-.24 to 0.97) | — | ||
| Candesartan | ||||
| 12 weeks | -0.18 (-0.54 to 0.19) | — | Low | |
| Riboflavin (MIDAS) | ||||
| Baseline | -0.06 (-0.46 to 0.33) | — | Low | |
| 4 weeks | 0.00 (-0.39 to 0.39) | — | ||
| Propranolol + Topiramate | Topiramate (MIDAS) | |||
| 24 weeks | 0.01 (-0.39 to 0.41) | — | Low | |
| Headache Severity | ||||
| Comparison | Standardized Mean Difference (95% CI) | Heterogeneity | Quality of evidence | |
| Metoprolol | Aspirin | |||
| 8 weeks | 0.33 (-0.20 to 0.86) | — | Low | |
| Acupuncture | ||||
| 17 weeks | -0.46 (-0.91 to -0.002) | — | Low | |
| Bisoprolol | ||||
| Baseline | 0.00 (-0.31 to 0.31) | — | Low | |
| 12 weeks | 0.19 (-0.13 to 0.3) | — | ||
| Flunarizine | ||||
| Baseline | 0.46 (0.14 to 0.79) | — | Low | |
| 4 weeks | 0.13 (-0.22 to 0.48) | — | ||
| 8 weeks | 0.38 (0.02 to 0.73) | — | ||
| 12 weeks | 0.13 (-0.22 to 0.48) | — | ||
| 16 weeks | 0.75 (0.39 to 1.1) | — | ||
| 20 weeks | 0.42 (0.07 to 0.77) | — | ||
| Nebivolol | ||||
| 16 weeks | 0.19 (-0.53 to 0.91) | — | Low | |
| Pizotifen | ||||
| Baseline | 0.00 (-0.47 to 0.47) | — | Low | |
| 8 weeks | -0.91 (-1.4 to -0.40) | — | ||
| Metoprolol + Flunarizine | Flunarizine | |||
| 12 weeks | -0.25 (-0.79 to 0.26) | — | Low | |
| 24 weeks | -0.55 (-1.1 to -0.02) | — | ||
| 36 weeks | -0.49 (-1.0 to 0.047) | — | ||
| 48 weeks | -0.54 (-0.72 to -0.19) | — | ||
| Propranolol | Acupuncture | |||
| Baseline | 0.26 (-0.13 to 0.65 | — | Low | |
| 12 weeks | 0.63 (0.23 to 1.03) | — | ||
| 5-hydroxytryptophan | ||||
| Baseline | 0.18 (-0.45 to 0.81) | — | Low | |
| 16 weeks | 0.00 (-0.63 to 0.63) | — | ||
| Biofeedback | ||||
| Baseline | -0.07 (-0.36 to 0.21) | — | Low | |
| 24 weeks | 0.13 (-0.15 to 0.42) | — | ||
| Cyclandelate | ||||
| Baseline | -0.07 (-0.58 to 0.43) | — | Low | |
| 4 weeks | -0.17 (-0.68 to 0.33) | — | ||
| 8 weeks | -0.01 (-0.51 to 0.48) | — | ||
| Flunarizine | ||||
| Baseline | 0.00 (-0.32 to 0.32) | Q = 0.0, df = 1, I2 = 0.0% | Moderate | |
| 4 weeks | -0.24 (-0.66 to 0.17) | — | Low | |
| 8 weeks | -0.57 (-1.0 to -0.13) | — | Low | |
| 12 weeks | -0.25 (-0.69 to 0.19) | — | Low | |
| 16 weeks | 0.17 (-0.52 to 0.85) | Q = 79.7, df = 3, I2 = 92.9% | High | |
| Metoprolol | ||||
| Baseline | 0.0 (-0.37 to 0.37) | — | Low | |
| 8 weeks | 0.0 (-0.38 to 0.38) | — | ||
| Nadolol | ||||
| Baseline | 0.00 (-0.67 to 0.84) | — | Low | |
| 24 Weeks | 0.86 (0.07 to 1.7) | — | ||
| Naproxen | ||||
| 12 weeks | 0.14 (-0.29 to 0.56) | — | Low | |
| Nimodipine | ||||
| 24 weeks | -0.50 (-0.89 to -0.11) | — | Low | |
| Propranolol (80 v 160mg doses) | ||||
| Baseline | 0.00 (-0.40 to 0.40) | — | Low | |
| 8 weeks | 0.00 (-0.51 to 0.51) | — | ||
| 12 weeks | 0.00 (-0.43 to 0.43) | — | ||
| Riboflavin | ||||
| Baseline | 0.32 (-0.08 to 0.71) | — | Low | |
| 4 weeks | 0.33 (-0.07 to 0.72) | — | ||
| 8 weeks | 0.21 (-0.18 to 0.60) | — | ||
| 12 weeks | 0.42 (0.02 to 0.82) | — | ||
| 24 weeks | 0.11 (-0.29 to 0.50) | — | ||
| Timolol | ||||
| Baseline | 0.00 (-0.31 to 0.31) | — | Low | |
| 12 weeks | 0.14 (-0.17 to 0.45) | — | ||
| Tolfenamic Acid | ||||
| Baseline | 0.26 (-0.37 to 0.68) | — | Low | |
| 12 weeks | 0.15 (-0.63 to 0.93) | Q = 4.53, df = 1, I2 = 77.8% | Moderate | |
| Topiramate | ||||
| Baseline | -0.44 (-0.95 to 0.07) | — | Low | |
| 4 weeks | -0.19 (-0.69 to 0.32) | — | ||
| 8 weeks | 0.23 (-0.28 to 0.74) | — | ||
| Headache Duration | ||||
| Comparison | Weighted Mean Difference (95% CI) | Heterogeneity | Quality of evidence | |
| Bisoprolol (5mg) | Bisoprolol (10 mg) | |||
| Baseline | 2.4 (-4.3 to 9.1) | — | Low | |
| 12 weeks | -4.8 (-9.9 to 0.31) | — | ||
| Metoprolol | Acupuncture | |||
| 12 weeks | -2.4 (-6.5 to 1.7) | — | Low | |
| Bisoprolol | ||||
| Baseline | 0.0 (-4.9 to 4.9) | — | Low | |
| 12 weeks | 0.30 (-4.2 to 4.8) | — | ||
| Clomipramine | ||||
| 4 weeks | -2.8 (-4.4 to -1.2) | — | Low | |
| Flunarizine | ||||
| Baseline | -1.3 (-3.7 to 1.1) | — | Low | |
| 4 weeks | -3.2 (-5.7 to -0.69) | — | ||
| 8 weeks | -3.3 (-5.9 to -0.66) | — | ||
| 12 weeks | -2.4 (-4.9 to 0.11) | — | ||
| 16 weeks | -0.40 (-3.2 to 2.4) | — | ||
| 20 weeks | -1.3 (-4.1 to 1.5) | — | ||
| Metoprolol + Fluoxetine | ||||
| Baseline | 0.10 (-0.15 to 0.35) | — | Low | |
| 12 weeks | -0.53 (-0.75 to -0.31) | — | ||
| Nebivolol | ||||
| 16 weeks | 11 (-18.6 to 40.6) | — | Low | |
| Nifedipine | ||||
| Baseline | 1.0 (-14.3 to 16.3) | — | Low | |
| 4 weeks | 15.8 (0.49 to 31.1) | — | ||
| 28 weeks | 12.2 (0.66 to 23.7) | — | ||
| Pindolol (7.5 mg) | Pindolol (15 mg) | |||
| Baseline | -0.70 (-7.9 to 6.6) | — | Low | |
| 4 weeks | -0.80 (-8.2 to 6.5) | — | ||
| Propranolol | 5-hydroxytryptophan | |||
| Baseline | 6.1 (-1.1 to 13.3) | — | Low | |
| 16 weeks | 3.6 (-3.7 to 10.9) | — | ||
| Biofeedback | ||||
| Baseline | 0.04 (-2.5 to 2.6) | — | Low | |
| 24 weeks | 0.33 (-1.3 to 2.0) | — | ||
| Candesartan | ||||
| 12 weeks | 6.3 (-1.9 to 14.5) | — | Low | |
| Cyclandelate | ||||
| Baseline | 6.7 (-22.5 to 9.1) | — | Low | |
| 4 weeks | -12.6 (-37.4 to 12.2) | — | ||
| 8 weeks | 6.6 (-18.2 to 31.4) | — | ||
| 12 weeks | -4.5 (-26.6 to 17.6) | — | ||
| Flunarizine | ||||
| Baseline | 2.7 (-1.2 to 6.6) | — | Low | |
| 4 weeks | -0.55 (-4.8 to 3.7) | Q = 0.06, df = 1, I2 = 0.0% | Moderate | |
| 8 weeks | 0.29 (-3.3 to 3.9) | Q = 0.08, df = 1, I2 = 0.0% | Moderate | |
| 12 weeks | -0.21 (-3.3 to 2.9) | Q = 0.09, df = 1, I2 = 0.0% | Moderate | |
| 16 weeks | 1.4 (0.23 to 2.6) | Q = 4.33, df = 4, I2 = 7.6% | High | |
| Mefenamic Acid | ||||
| 12 weeks | 7.0 (-27.3 to 41.3) | — | Low | |
| Metoprolol | ||||
| Baseline | -24.0 (-40.3 to -7.7) | — | Low | |
| 4 weeks | 4.2 (-12.1 to 20.5) | — | ||
| 16 weeks | 0.0 (-12.3 to 12.3) | — | ||
| 28 weeks | 33.0 (6.3 to 59.7) | — | ||
| Nadolol | ||||
| Baseline | -8.1 (-11.4 to -4.8) | — | Low | |
| 24 weeks | -19.5 (-31.8 to -7.1) | — | ||
| Nifedipine | ||||
| Baseline | -1.0 (-16.3 to 14.3) | — | Low | |
| 4 weeks | -15.8 (-31.1 to -0.49) | — | ||
| 16 weeks | -12.2 (-23.7 to -0.66) | — | ||
| 28 weeks | 20.0 (0.97 to 39.0) | — | ||
| Nimodipine | ||||
| 24 weeks | -4.0 (-7.9 to -0.08) | — | Low | |
| Propranolol (80 mg vs 160mg) | ||||
| Baseline | 0.0 (-1.2 to 1.2) | — | Low | |
| 8 weeks | -0.30 (-6.9 to 6.3) | — | ||
| 12 weeks | 0.20 (-1.8 to 2.2) | — | ||
| Riboflavin | ||||
| Baseline | 0.10 (-0.29 to 0.49) | — | Low | |
| 4 weeks | 0.10 (-0.31 to 0.51) | — | ||
| 8 weeks | 0.00 (-0.26 to 0.26) | — | ||
| 12 weeks | -0.10 (-0.39 to 0.19) | — | ||
| 24 weeks | 0.30 (-0.06 to 6.6) | — | ||
| Timolol | ||||
| Baseline | 0.00 (-2.6 to 2.6) | — | Low | |
| 12 weeks | -0.03 (-2.3 to 2.3) | — | ||
| Tolfenamic Acid | ||||
| Baseline | 6.4 (-21.9 to 34.7) | — | Low | |
| 12 weeks | 1.7 (-4.6 to 8.0) | Q = 0.05, df = 1, I2-0.0% | Moderate | |
| Topiramate | ||||
| Baseline | -1.3 (-4.8 to 2.3) | — | Low | |
| 4 weeks | -1.2 (-4.6 to 2.3) | — | ||
| 8 weeks | 1.0 (-1.9 to 4.0) | — | ||
| Valproic Acid | ||||
| Baseline | -1.4 (-9.9 to 7.2) | — | Low | |
| 10 weeks | -0.70 (-4.0 to 2.6) | — | ||
Chronic migraine
There were four trials that evaluated beta-blockers for chronic migraine headaches, none were placebo controlled. (Table 6). Propranolol was compared to flunarizine [151], nortriptyline [76], valproic acid [105] and to the combination of propranolol and flunarizine [151]. In addition, a combination of propranolol and topiramate was compared to topiramate alone [129]. Propranolol was no better than valproic acid or flunarizine and the combinations (propranolol + topiramate and propranolol + flunarizine) was no better than topiramate and flunarizine alone (Table 6).
Table 6. Non-episodic trials.
| Chronic Migraine | |||||
|---|---|---|---|---|---|
| Comparison | Outcome | Time point | Effect | Heterogeneity | Quality of Evidence |
| Propranolol v. Placebo | 50% reduction in headache (RR) | 42 weeks | 2.0 (0.94 to 4.3) | — | Low |
| Propranolol v. Flunarizine | Headache Frequency (headaches/month) | Baseline | 3.0 (0.79 to 5.2) | — | Low |
| 8 weeks | 1.0 (-1.5 to 3.5) | — | |||
| Propranolol + Flunarizine vs. Flunarizine | Headache Frequency (headaches/month) | Baseline | 2.0 (-0.19 to 4.2) | — | Low |
| 8 weeks | 3.0 (0.56 to 5.4) | — | |||
| 50% reduction in headache (RR) | 8 weeks | 1.3 (0.97 to 1.6) | — | Low | |
| Propranolol v. Nortriptyline | Headache Frequency (Headaches/month) | Baseline | -1.0 (-4.7 to 2.7) | — | Low |
| 8 weeks | -9.0 (-12.7 to -5.3) | — | |||
| Propranolol v. Propranolol + Nortriptyline | Headache Frequency (Headaches/month) | Baseline | -4.0 (-7.8 to -0.24) | — | Low |
| 8 weeks | -7.0 (-10.8 to -3.3) | — | |||
| Propranolol + Topiramate v. Topiramate | Headache Frequency (headaches/month) | Baseline | 0.0 (-0.28 to 0.28) | — | Low |
| 12 weeks | -0.80 (-2.3 to 0.67) | — | |||
| Health Related Quality of Life (MIDAS) | Baseline | 0.00 (-0.28 to 0.28) | — | Low | |
| 12 weeks | 0.09 (-0.26 to 0.44) | — | |||
| 50% reduction in headache (RR) | 12 weeks | 1.05 (0.63 to 1.7) | — | Low | |
| 24 weeks | 1.1 (0.79 to 1.8) | — | |||
| Propranolol v. Valproic Acid | Headache Frequency (headaches/month) | 8 weeks | 4.8 (0.27 to 9.2) | — | Low |
| Chronic Tension-Type Headache | |||||
| Pindolol + Amitriptyline v. Placebo | Headache Frequency (headaches/month) | Baseline | 1.4 (-2.3 to 5.0) | — | Low |
| 4 weeks | 7.8 (-13.9 to -1.5) | — | |||
| 8 weeks | -11.6 (-16.8 to -5.5) | — | |||
| 50% reduction in headache (RR) | 8 weeks | 3.8 (1.5 to 9.3) | — | Low | |
| Headache Severity (SMD) | Baseline | 0.29 (-0.34 to 0.91) | — | Low | |
| 8 weeks | -0.68 (-1.3 to -0.04) | — | |||
| Pindolol + Amitriptyline v. Amitriptyline | Headache Frequency (headaches/month) | Baseline | 1.6 (-2.2 to 5.3) | — | Low |
| 4 weeks | 0.64 (-5.2 to 6.4) | — | |||
| 8 weeks | -1.2 (-6.4 to 4.1) | — | |||
| 50% reduction in headache (RR) | 8 weeks | 1.4 (0.87 to 2.2) | — | Low | |
| Headache Severity (SMD) | Baseline | 3.7 (2.7 to 4.7) | — — |
Low | |
| 8 weeks | -0.05 (-0.65 to 0.56) | — | |||
Tension-type headache
There was only one trial evaluating tension-type headache, comparing the combination of pindolol and amitriptyline to placebo and to amitriptyline alone [57]. The combination of pindolol and amitriptyline was more effective than placebo at reducing headache frequency at 4 and 8 weeks (Table 6) and in reducing headaches by at least 50% (RR: 3.8, 95% CI: 1.5–9.3), but equally effective with amitriptyline.
Adverse events
Participants on beta-blockers were more likely to experience side effects than those on placebo (RR: 1.2, 95% CI: 1.0–1.4), though they were not more likely to withdraw (RR: 0.99, 95% CI: 0.83 to 1.2). Specific side effects more common with beta-blockers included dizziness (RR: 1.5, 95% CI: 1.0–2.3) and fatigue (RR: 1.5, 95% CI: 1.2–2.0). Depression, gastrointestinal problems, paresthesia’s and weight gain were not significantly different than placebo.
Propranolol was the only beta-blocker with sufficient numbers of studies to perform sensitivity analysis. There was no evidence of publication bias for propranolol’s reduction of headache frequency at 8 weeks (Egger’s p = 0.77) or at 12 weeks (p = 0.62). There was no evidence of an effect of quality (p = 0.97), age (p = 0.71), percent women (p = 0.28), percentage of dropouts (p = 0.55), dose (p = 0.61), intention to treat analysis (p = 0.35), concealed allocation (p = 0.38) or appropriateness of blinding (p = 0.98).
Discussion
This review included one hundred and eight randomized controlled trials. Nearly all evaluated the efficacy of beta-blockers for episodic migraine headaches. Compared to placebo, propranolol was effective in reducing episodic migraine frequency. The effect began as early as four weeks. Migraine headache sufferers experienced an average reduction of 1.3 headaches/month; this translates to a reduction from 4.8 to 3.5 headaches a month. Subjects given propranolol were more likely to report at least 50% reduction in headaches and to reduce their consumption of analgesic medications. In addition to reducing the number of headaches, the residual headaches were less severe and shorter in duration compared to those receiving placebo. Outcomes from the propranolol comparisons to placebo were rated as high-quality evidence. In three trials, metoprolol also reduced headache frequency, though the reduction was less than 1 headache a month. Conclusions regarding the efficacy of other beta-blockers is less certain, as most were studied in just one trial each. Atenolol, bisoprolol and timolol had weak evidence of benefit. Acebutolol, alprenolol and nadolol appeared to be ineffective in migraine prophylaxis. This is unlikely to be due to properties of the beta-blockers. Propranolol is nonselective as is alprenolol and nadolol. Metoprolol, also effective is a ß-1 selective drug as is atenolol, bisoprolol and acebutolol. Given that acebutolol, alprenolol and nadolol were only studied in one trial each, it is possible that this may be either random variation in outcomes or a problem with the trials (such as dose or duration). The network analysis suggests that the benefit of beta-blocker may be a class effect.
The literature comparing beta-blockers to other modalities consisted mostly of single-trials with the exception of the comparison of propranolol to metoprolol (moderate quality, no difference) and to flunarizine (high quality, no difference). Flunarizine, not available in the United States, is well-established as effective in treating migraine headache. Universally, beta-blockers were associated with bradycardia and with lower average pulse rates than placebo trials. This is not surprising given their impact on chronotropy. Other side effects more common among participants taking beta-blockers included dizziness and fatigue, though subjects on beta-blockers were not more likely to withdraw from the studies.
While these conclusions are similar to previous reviews, this analysis is a unique contribution in many ways, first it included many more trials than previously reported, including Chinese trials that had previously not been included. Secondly, all beta-blockers were carefully parsed by type of headache (tension v. migraine, episodic v. chronic). Third, this study examined outcomes at the specific times reported, it is common for meta-analyses to pool trials at the last time point, regardless of whether there were significant differences in that time-point. Fourth, trial sequential analysis that demonstrated the adequacy of the current database for propranolol, suggesting that there is low likelihood of type 1 error in the conclusions. Fifth, the network meta-analysis didn’t show clear benefit of one beta-blocker over another, suggesting a class effect, though other beta-blockers have weaker evidence for benefit.
An important question, unanswered in this review, is how effective beta-blockers are compared to other commonly used prophylactic regimens. Propranolol, metoprolol and flunarizine appear to have similar efficacy. The other comparisons were all single-trial comparisons, making definitive conclusions impossible. In a previous review, tricyclic antidepressants resulted in a reduction in headache frequency for patients experiencing chronic migraines of 1.3 SMD, compared to placebo, a large effect [17]. In this study, there was only had one trial on chronic migraines, and the calculated SMD was 0.58, about half of the effect previously reported for TCAs. In a network meta-analysis of chronic migraines, tricyclic antidepressants were more effective than propranolol [17] but propranolol was similar in efficacy to antiepileptics and flunarizine, similar to findings in this study, though it is important to note that the majority of trials for beta-blockers are for episodic rather than chronic migraines. Definitive answer to this comparative effectiveness question requires additional studies that directly compare the different prophylactic modalities.
This review has several important limitations. First, while the quality of evidence for the comparison between propranolol and placebo was high, in general, the remaining comparisons were of low quality, consisting mostly of underpowered single randomized trials. While all the included trials were randomized, there were significant methodologic problems; combining poorly designed studies can lead to questionable results. It is important to note that most of the comparisons were graded as being of low-quality evidence because of the paucity of studies and concern about study problems. Secondly, studies were inconsistent in reporting outcomes, so even when there were more than one trial available, specific outcomes may only be provided by a single study. Moreover, there were significant problems with selective reporting of outcomes and many studies did not collect information on headache frequency, the measure preferred by the International Headache Society. Third, the number of studies available precluded sensitivity analyses, such as assessing for publication bias or exploring sources of heterogeneity. Fourth, beta-blockers have been studied almost exclusively in the management of episodic migraine headaches. Their benefit for chronic episodic or tension-type headaches is uncertain. Fifth, because of the paucity of trials for most beta-blockers the network analysis was underpowered to show differences between beta-blockers, Sixth, the average age of participants was 38, and mostly female. While this reflects the demographics of headache, it limits applicability to older adults.
Conclusions
Propranolol is effective in reducing the burden of patients with episodic migraine headaches, reducing headaches from 5 to 3 headaches a month. This means that migraine sufferers given propranolol will have substantial residual headache burden. Propranolol reduces headaches by more than 50% as well as reducing the number of analgesic medication doses required. It also reduces the severity or duration of the headaches experienced. Propranolol and metoprolol exert similar effects and propranolol is as effective as flunarizine. The data for other beta-blockers and other comparisons are less clear.
Supporting information
(DOC)
(DOCX)
(DOCX)
Acknowledgments
The views reflected in this manuscript are those of the authors and should not be construed, in any way, to be those of the Department of Veterans Affairs.
Data Availability
Dara are available at https://doi.org/10.6084/m9.figshare.7674092.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Rasmussen BK. Epidemiology of headache. Cephalalgia 2001; 21(7):774–777. 10.1177/033310240102100708 [DOI] [PubMed] [Google Scholar]
- 2.Wang SJ. Epidemiology of migraine and other types of headache in Asia. Current Neurology & Neuroscience Reports 2003; 3(2):104–108. [DOI] [PubMed] [Google Scholar]
- 3.Radtke A, Neuhauser H. Prevalence and burden of headache and migraine in Germany. Headache 2009; 49(1):79–89. 10.1111/j.1526-4610.2008.01263.x [DOI] [PubMed] [Google Scholar]
- 4.Karli N, Zarifoglu M, Ertafs M, Saip S, Ozturk V, Bicakci S et al. Economic impact of primary headaches in Turkey: a university hospital based study: part II. J Headache Pain 2006; 7(2):75–82. 10.1007/s10194-006-0273-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Falavigna A, Teles AR, Velho MC, Vedana VM, Silva RC, Mazzocchin T et al. Prevalence and impact of headache in undergraduate students in Southern Brazil. Arq Neuropsiquiatr 2010; 68(6):873–877. [DOI] [PubMed] [Google Scholar]
- 6.Lipton RB, Bigal ME, Kolodner K, Stewart WF, Liberman JN, Steiner TJ. The family impact of migraine: population-based studies in the USA and UK. Cephalalgia 2003; 23(6):429–440. 10.1046/j.1468-2982.2003.00543.x [DOI] [PubMed] [Google Scholar]
- 7.Lipton RB, Liberman JN, Kolodner KB, Bigal ME, Dowson A, Stewart WF. Migraine headache disability and health-related quality-of-life: a population-based case-control study from England. Cephalalgia 2003; 23(6):441–450. 10.1046/j.1468-2982.2003.00546.x [DOI] [PubMed] [Google Scholar]
- 8.Adams AM, Serrano D, Buse DC, Reed ML, Marske V, Fanning KM et al. The impact of chronic migraine: The Chronic Migraine Epidemiology and Outcomes (CaMEO) Study methods and baseline results. Cephalalgia 2015; 35(7):563–578. 10.1177/0333102414552532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Buse DC, Manack AN, Fanning KM, Serrano D, Reed ML, Turkel CC et al. Chronic migraine prevalence, disability, and sociodemographic factors: results from the American Migraine Prevalence and Prevention Study. Headache 2012; 52(10):1456–1470. 10.1111/j.1526-4610.2012.02223.x [DOI] [PubMed] [Google Scholar]
- 10.Blumenfeld AM, Varon SF, Wilcox TK, Buse DC, Kawata AK, Manack A et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS). Cephalalgia 2011; 31(3):301–315. 10.1177/0333102410381145 [DOI] [PubMed] [Google Scholar]
- 11.Leonardi M, Raggi A, Bussone G, D'Amico D. Health-related quality of life, disability and severity of disease in patients with migraine attending to a specialty headache center. Headache 2010; 50(10):1576–1586. 10.1111/j.1526-4610.2010.01770.x [DOI] [PubMed] [Google Scholar]
- 12.Raggi A, Leonardi M, Bussone G, D'Amico D. Value and utility of disease-specific and generic instruments for assessing disability in patients with migraine, and their relationships with health-related quality of life. Neurol Sci 2010. [DOI] [PubMed] [Google Scholar]
- 13.Tepper SJ. A pivotal moment in 50 years of headache history: the first American Migraine Study. Headache 2008; 48(5):730–731. 10.1111/j.1526-4610.2008.01117_1.x [DOI] [PubMed] [Google Scholar]
- 14.Freitag FG. The cycle of migraine: patients' quality of life during and between migraine attacks. Clin Ther 2007; 29(5):939–949. 10.1016/j.clinthera.2007.05.008 [DOI] [PubMed] [Google Scholar]
- 15.Hu XH, Markson LE, Lipton RB, Stewart WF, Berger ML. Burden of migraine in the United States: disability and economic costs. Archives of Internal Medicine 1999; 159(8):813–818. [DOI] [PubMed] [Google Scholar]
- 16.Goldberg LD. The cost of migraine and its treatment. Am J Manag Care 2005; 11(2 Suppl):S62–S67. [PubMed] [Google Scholar]
- 17.Jackson JL, Cogbill E, Santana-Davila R, Eldredge C, Collier W, Gradall A et al. A Comparative Effectiveness Meta-Analysis of Drugs for the Prophylaxis of Migraine Headache. Plos One 2015; 10(7):e0130733 10.1371/journal.pone.0130733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ferrante T, Manzoni GC, Russo M, Camarda C, Taga A, Veronesi L et al. Prevalence of tension-type headache in adult general population: the PACE study and review of the literature. Neurological Sciences 2013; 34 Suppl 1:S137–S138. [DOI] [PubMed] [Google Scholar]
- 19.Lyngberg AC, Rasmussen BK, Jorgensen T, Jensen R. Has the prevalence of migraine and tension-type headache changed over a 12-year period? European Journal of Epidemiology 2005; 20(3):243–249. [DOI] [PubMed] [Google Scholar]
- 20.Rasmussen BK, Jensen R, Schroll M, Olesen J. Epidemiology of headache in a general population—A prevalence study. Journal of Clinical Epidemiology 1991; 44(11):1147–1157. [DOI] [PubMed] [Google Scholar]
- 21.Schwartz BS, Stewart WF, Simon D, Lipton RB. Epidemiology of tension-type headache. JAMA 1998; 279(5):381–383. [DOI] [PubMed] [Google Scholar]
- 22.Stovner LJ, Hagen K, Jensen J, Katsarava Z, Lipton RB, Scher AI et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia 2007; 27(3):193–210. 10.1111/j.1468-2982.2007.01288.x [DOI] [PubMed] [Google Scholar]
- 23.Abu Bakar N, Tanprawate S, Lambru G, Torkamani M, Jahanshahi M, Matharu M. Quality of life in primary headache disorders: A review. Cephalalgia 2016; 36(1):67–91. 10.1177/0333102415580099 [DOI] [PubMed] [Google Scholar]
- 24.Latinovic R, Gulliford M, Ridsdale L. Headache and migraine in primary care: consultation, prescription, and referral rates in a large population. J Neurol Neurosurg Psychiatry 2006; 77(3):385–387. 10.1136/jnnp.2005.073221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Linde M, Gustavsson A, Stovner LJ, Steiner TJ, Barr AJ., Katsarava Z et al. The cost of headache disorders in Europe: the Eurolight project. European Journal of Neurology 2012; 19(5):703–711. 10.1111/j.1468-1331.2011.03612.x [DOI] [PubMed] [Google Scholar]
- 26.Chronicle E, Mulleners W. Anticonvulsant drugs for migraine prophylaxis. Cochrane Library 2004;(3). [DOI] [PubMed] [Google Scholar]
- 27.Linde K, Rossnagel K. Propranolol for migraine prophylaxis. Cochrane Database Syst Rev 2004;(2):CD003225 10.1002/14651858.CD003225.pub2 [DOI] [PubMed] [Google Scholar]
- 28.Jackson JL, Kuriyama A, Hayashino Y. Botulinum toxin A for prophylactic treatment of migraine and tension headaches in adults: a meta-analysis. JAMA 2012; 307(16):1736–1745. 10.1001/jama.2012.505 [DOI] [PubMed] [Google Scholar]
- 29.Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ 2010; 182(7):E269–E276. 10.1503/cmaj.081657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lance JW, Anthony M. Clinical trial of a new serotonin antagonist, BC105, in the prevention of migraine. Med J Aust 1968; 1(2):54–55. [DOI] [PubMed] [Google Scholar]
- 31.Moja PL, Cusi C, Sterzi RR, Canepari C. Selective serotonin re-uptake inhibitors (SSRIs) for preventing migraine and tension-type headaches. Cochrane Database Syst Rev 2005;(3):CD002919 10.1002/14651858.CD002919.pub2 [DOI] [PubMed] [Google Scholar]
- 32.Jackson JL, Shimeall W, Sessums L, Dezee KJ, Becher D, Diemer M et al. Tricyclic antidepressants and headaches: systematic review and meta-analysis. BMJ 2010; 341:c5222 10.1136/bmj.c5222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rapaport AM. Recurrent migraine: cost-effective care. Neurology 1994; 44(5 Suppl 3):S25–S28. [PubMed] [Google Scholar]
- 34.Silberstein SD, Holland S, Freitag F, Dodick DW, Argoff C, Ashman E. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology 2012; 78(17):1337–1345. 10.1212/WNL.0b013e3182535d20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ferrari A, Pasciullo G, Savino G, Cicero AF, Ottani A, Bertolini A et al. Headache treatment before and after the consultation of a specialized centre: a pharmacoepidemiology study. Cephalalgia 2004; 24(5):356–362. 10.1111/j.1468-2982.2004.00678.x [DOI] [PubMed] [Google Scholar]
- 36.Takaki H, Onzuka D, Hagihara A. Migraine-preventive prescription patters by specialty in ambulatory care settings in the United States. Prev Med 2017; 9:62–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hepp Z, Dodick D. Persistence and switching patterns of oral migraine prophylactic medications among patients with chronic migraine: a retrospective claims analysis. Cephalagia 2017; 37(5):470–485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mafi JN, Edwards ST, Pedersen NP, Davis RB, McCarthy EP, Landon BE. Trends in the ambulatory management of headache: analysis of NAMCS and NHAMCS data 1999–2010. J Gen Intern Med 2015; 30(5):548–555. 10.1007/s11606-014-3107-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holroyd KA, Penzien DB, Cordingley GE. Propranolol in the management of recurrent migraine: a meta-analytic review. Headache 1991; 31(5):333–340. [DOI] [PubMed] [Google Scholar]
- 40.Linde K, Rossnagel K. Propranolol for migraine prophylaxis. Cochrane Database Syst Rev 2004;(2):CD003225 10.1002/14651858.CD003225.pub2 [DOI] [PubMed] [Google Scholar]
- 41.Shamliyan TA, Choi JY, Ramakrishnan R, Miller JB, Wang SY, Taylor FR et al. Preventive pharmacologic treatments for episodic migraine in adults. J Gen Intern Med 2013; 28(9):1225–1237. 10.1007/s11606-013-2433-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Moher D, Liverati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLOS Medicine 2009; 6(7):e1000097 10.1371/journal.pmed.1000097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018; 38(1):1–211. 10.1177/0333102417738202 [DOI] [PubMed] [Google Scholar]
- 44.Bendtsen L, Bigal ME, Cerbo R, et al. Guidelines for controlled trials of drugs in tension-type headache: Second edition. Cephalalgia 2010; 30(1):1–16. 10.1111/j.1468-2982.2009.01948.x [DOI] [PubMed] [Google Scholar]
- 45.Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.2. www.cochrane-handbook.org. 2011. Ref Type: Generic [Google Scholar]
- 46.Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 1996; 17(1):1–12. [DOI] [PubMed] [Google Scholar]
- 47.Kazis LE, Anderson JJ, Meenan RF. Effect sizes for interpreting changes in health status. Medical Care 1989; 27 Suppl 3:S178–S189. [DOI] [PubMed] [Google Scholar]
- 48.Follmann D, Elliott P, Suh I, Cutler J. Variance imputation for overviews of clinical trials with continuous response. Journal of Clinical Epidemiology 1992; 45(7):769–773. [DOI] [PubMed] [Google Scholar]
- 49.Galbraith RF. A note on graphical presentation of estimated odds ratios from several clinical trials. Statistics in Medicine 1988; 7(8):889–894. [DOI] [PubMed] [Google Scholar]
- 50.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003; 327(7414):557–560. 10.1136/bmj.327.7414.557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clinical Trials 1986; 7(3):177–188. [DOI] [PubMed] [Google Scholar]
- 52.Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L. Comparison of two methods to detect publication bias in meta-analysis. JAMA 2006; 295(6):676–680. 10.1001/jama.295.6.676 [DOI] [PubMed] [Google Scholar]
- 53.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997; 315(7109):629–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wetterslev J, Thorlund K, Brok J, Gluud C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. J Clin Epidemiol 2008; 61(1):64–75. 10.1016/j.jclinepi.2007.03.013 [DOI] [PubMed] [Google Scholar]
- 55.Knapp G, Hartung J. Improved tests for a random effects meta-regression with a single covariate. Stat Med 2003; 22(17):2693–2710. 10.1002/sim.1482 [DOI] [PubMed] [Google Scholar]
- 56.White I. Multivariate random-effects meta-analysis. Stata Journal 2009; 9:40–56. [Google Scholar]
- 57.Agius AM, Jones NS, Muscat R. A Randomized Controlled Trial comparing the efficacy of low-dose amitriptyline, amitriptyline with pindolol and surrogate placebo in the treatment of chronic tension-type facial pain. 2013; 51(2):143–153. [DOI] [PubMed] [Google Scholar]
- 58.Ahuja GK, Verma AK. Propranolol in prophylaxis of migraine. Indian J Med Res 1985; 82:263–265. [PubMed] [Google Scholar]
- 59.al-Qassab HK, Findley LJ. Comparison of propranolol LA 80 mg and propranolol LA 160 mg in migraine prophylaxis: a placebo controlled study. Cephalalgia 1993; 13(2):128–131. 10.1046/j.1468-2982.1993.1302128.x [DOI] [PubMed] [Google Scholar]
- 60.Albers GW, Simon LT, Hamik A, Peroutka SJ. Nifedipine versus propranolol for the initial prophylaxis of migraine. Headache 1989; 29(4):215–218. [DOI] [PubMed] [Google Scholar]
- 61.Andersson PG, Petersen EN. Propranolol and femoxetine, a HT-uptake inhibitor, in migraine prophylaxis. A double-blind crossover study. Acta Neurol Scand 1981; 64(4):280–288. [DOI] [PubMed] [Google Scholar]
- 62.Andersson PG, Dahl S, Hansen JH, Hansen PE, Hedman C, Kristensen TN et al. Prophylactic treatment of classical and non-classical migraine with metoprolol—a comparison with placebo. Cephalalgia 1983; 3(4):207–212. 10.1046/j.1468-2982.1983.0304207.x [DOI] [PubMed] [Google Scholar]
- 63.Ashtari F, Shaygannejad V, Akbari M. A double-blind, randomized trial of low-dose topiramate vs propranolol in migraine prophylaxis. 2008; 118(5):301–305. [DOI] [PubMed] [Google Scholar]
- 64.Baldrati A, Cortelli P, Procaccianti G, Gamberini G, D'Alessandro R, Baruzzi A et al. Propranolol and acetylsalicylic acid in migraine prophylaxis. Double-blind crossover study. Acta Neurol Scand 1983; 67(3):181–186. [DOI] [PubMed] [Google Scholar]
- 65.Behan PO, Reid M. Propranolol in the treatment of migraine. Practitioner 1980; 224(1340):201–203. [PubMed] [Google Scholar]
- 66.Bonuso S, Di SE, Marano E, de AS, Amato D, Scellini T et al. Long-term outcome of migraine therapy: predictive value of the frontotemporal nitroglycerin test. Neurology 1998; 51(5):1475–1478. [DOI] [PubMed] [Google Scholar]
- 67.Bordini CA, Arruda MA, Ciciarelli MC, Speciali JG. Propranolol vs flunarizine vs flunarizine plus propranolol in migraine without aura prophylaxis. A double-blind trial. Arq Neuropsiquiatr 1997; 55(3B):536–541. [DOI] [PubMed] [Google Scholar]
- 68.Borgesen SE, Nielsen JL, Moller CE. Prophylactic treatment of migraine with propranolol. A clinical trial. Acta Neurol Scand 1974; 50(5):651–656. [DOI] [PubMed] [Google Scholar]
- 69.Briggs RS, Millac PA. Timolol in migraine prophylaxis. Headache 1979; 19:379–381. [DOI] [PubMed] [Google Scholar]
- 70.Carroll JD, Reidy M, Savundra PA, Cleave N, McAinsh J. Long-acting propranolol in the prophylaxis of migraine: a comparative study of two doses. Cephalalgia 1990; 10(2):101–105. 10.1046/j.1468-2982.1990.1002101.x [DOI] [PubMed] [Google Scholar]
- 71.Dahlof C. No clearcut longterm prophylactic effect of one month of treatment with propranolol in migraineurs. Cephalalgia 1987; 7:459–460. [Google Scholar]
- 72.Diener HC, Foh M, Iaccarino C, Wessely P, Isler H, Strenge H et al. Cyclandelate in the prophylaxis of migraine: a randomized, parallel, double-blind study in comparison with placebo and propranolol. The Study group. Cephalalgia 1996; 16(6):441–447. 10.1046/j.1468-2982.1996.1606441.x [DOI] [PubMed] [Google Scholar]
- 73.Diener HC, Hartung E, Chrubasik J, Evers S, Schoenen J, Eikermann A et al. A comparative study of oral acetylsalicyclic acid and metoprolol for the prophylactic treatment of migraine. A randomized, controlled, double-blind, parallel group phase III study. Cephalalgia 2001; 21(2):120–128. 10.1046/j.1468-2982.2001.00168.x [DOI] [PubMed] [Google Scholar]
- 74.Diener HC, Matias-Guiu J, Hartung E, Pfaffenrath V, Ludin HP, Nappi G et al. Efficacy and tolerability in migraine prophylaxis of flunarizine in reduced doses: a comparison with propranolol 160 mg daily. Cephalalgia 2002; 22(3):209–221. 10.1046/j.1468-2982.2002.t01-1-00309.x [DOI] [PubMed] [Google Scholar]
- 75.Diener HC, Tfelt-Hansen P, Dahlof C, Lainez MJ, Sandrini G, Wang SJ et al. Topiramate in migraine prophylaxis—results from a placebo-controlled trial with propranolol as an active control. J Neurol 2004; 251(8):943–950. 10.1007/s00415-004-0464-6 [DOI] [PubMed] [Google Scholar]
- 76.Domingues RB, Pirajá Da Silva AL, Domingues SA, Aquino CCH, Kuster GW. A double-blind randomized controlled trial of low doses of propranolol, nortriptyline, and the combination of propranolol and nortriptyline for the preventive treatment of migraine. 2009; 67(4):973–977. [DOI] [PubMed] [Google Scholar]
- 77.Ekbom K, Lundberg PO. Clinical trial of LB-46 (d, 1-4-(2-hydroxy-3-isopropylaminopropoxy)indol. An adrenergic beta-receptor blocking agent in migraine prophylaxis. Headache 1972; 12(1):15–17. [DOI] [PubMed] [Google Scholar]
- 78.Ekbom K. Alprenolol for migraine prophylaxis. Headache 1975; 15(2):129–132. [DOI] [PubMed] [Google Scholar]
- 79.Ekbom K, Zetterman M. Oxprenolol in the treatment of migraine. Acta Neurol Scand 1977; 56(2):181–184. [DOI] [PubMed] [Google Scholar]
- 80.Formisano R, Falaschi P, Cerbo R, Proietti A, Catarci T, D'Urso R et al. Nimodipine in migraine: clinical efficacy and endocrinological effects. Eur J Clin Pharmacol 1991; 41(1):69–71. 10.1007/BF00280110 [DOI] [PubMed] [Google Scholar]
- 81.Forssman B, Henriksson KG, Johannsson V. Propranolol for migraine prophylaxis. Headache 1976; 16:238–245. [DOI] [PubMed] [Google Scholar]
- 82.Forssman B, Lindblad CJ, Zbornikova V. Atenolol for migraine prophylaxis. Headache 1983; 23(4):188–190. [DOI] [PubMed] [Google Scholar]
- 83.Freitag FG, Diamond S. Nadolol and placebo comparison study in the prophylactic treatment of migraine. J Am Osteopath Assoc 1984; 84(4):343–347. [PubMed] [Google Scholar]
- 84.Gawel MJ, Kreeft J, Nelson RF, Simard D, Arnott WS. Comparison of the efficacy and safety of flunarizine to propranolol in the prophylaxis of migraine. Can J Neurol Sci 1992; 19(3):340–345. [PubMed] [Google Scholar]
- 85.Gerber WD, Diener HC, Scholz E, Niederberger U. Responders and non-responders to metoprolol, propranolol and nifedipine treatment in migraine prophylaxis: a dose-range study based on time-series analysis. Cephalalgia 1991; 11(1):37–45. 10.1046/j.1468-2982.1991.1101037.x [DOI] [PubMed] [Google Scholar]
- 86.Gerber WD, Schellenberg R, Thom M, Haufe C, Bolsche F, Wedekind W et al. Cyclandelate versus propranolol in the prophylaxis of migraine—a double-blind placebo-controlled study. Funct Neurol 1995; 10(1):27–35. [PubMed] [Google Scholar]
- 87.Ghobadi SH, Jivad N. The prophylactic activity of propranol and nimodipineon migraine headache. 2013; 8(2):144–146. [Google Scholar]
- 88.Grotemeyer KH, Husstedt IW, Schlake HP. [Betablocker vs placebo in vasomotor headache. A double-blind crossover study]. Dtsch Med Wochenschr 1987; 112(45):1740–1743. 10.1055/s-2008-1068322 [DOI] [PubMed] [Google Scholar]
- 89.Grotemeyer KH, Schlake HP, Husstedt IW. [Prevention of migraine with metoprolol and flunarizine. A double-blind crossover study]. Nervenarzt 1988; 59(9):549–552. [PubMed] [Google Scholar]
- 90.Grotemeyer KH, Scharafinski HW, Schlake HP, Husstedt IW. Acetylsalicylic acid vs. metoprolol in migraine prophylaxis—a double-blind cross-over study. Headache 1990; 30(10):639–641. [DOI] [PubMed] [Google Scholar]
- 91.Havanka-Kanniainen H, Hokkanen E, Myllyla VV. Long acting propranolol in the prophylaxis of migraine. Comparison of the daily doses of 80 mg and 160 mg. Headache 1988; 28(9):607–611. [DOI] [PubMed] [Google Scholar]
- 92.Hedman C, Winther K, Knudsen JB. The difference between non-selective and beta 1-selective beta-blockers in their effect on platelet function in migraine patients. Acta Neurol Scand 1986; 74(6):475–478. [DOI] [PubMed] [Google Scholar]
- 93.Hesse J, Mogelvang B, Simonsen H. Acupuncture versus metoprolol in migraine prophylaxis: a randomized trial of trigger point inactivation. J Intern Med 1994; 235(5):451–456. [DOI] [PubMed] [Google Scholar]
- 94.Holdorff B, Sinn M, Roth G. Propranolol for prophylaxis of migraine. Medizinische Klinik 1977; 72(25):1115–1118. [PubMed] [Google Scholar]
- 95.Holroyd KA, Cottrell CK, O'Donnell FJ, Cordingley GE, Drew JB, Carlson BW et al. Effect of preventive (Î2 blocker) treatment, behavioural migraine management, or their combination on outcomes of optimised acute treatment in frequent migraine: Randomised controlled trial. 2010; 341(7776):769 10.1136/bmj.c4871 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Johannsson V, Nilsson LR, Widelius T, Javerfalk T, Hellman P, Akesson JA et al. Atenolol in migraine prophylaxis a double-blind cross-over multicentre study. Headache 1987; 27(7):372–374. [DOI] [PubMed] [Google Scholar]
- 97.Johnson RH, Hornabrook RW, Lambie DG. Comparison of mefenamic acid and propranolol with placebo in migraine prophylaxis. Acta Neurol Scand 1986; 73(5):490–492. [DOI] [PubMed] [Google Scholar]
- 98.Kangasniemi P, Hedman C. Metoprolol and propranolol in the prophylactic treatment of classical and common migraine. A double-blind study. Cephalalgia 1984; 4(2):91–96. 10.1046/j.1468-2982.1984.0402091.x [DOI] [PubMed] [Google Scholar]
- 99.Kangasniemi P, Andersen AR, Andersson PG, Gilhus NE, Hedman C, Hultgren M et al. Classic migraine: effective prophylaxis with metoprolol. Cephalalgia 1987; 7(4):231–238. 10.1046/j.1468-2982.1987.0704231.x [DOI] [PubMed] [Google Scholar]
- 100.Kangasniemi PJ, Nyrke T, Lang AH, Petersen E. Femoxetine—a new 5-HT uptake inhibitor—and propranolol in the prophylactic treatment of migraine. Acta neurologica Scandinavica 1983; 68:262–267. [DOI] [PubMed] [Google Scholar]
- 101.Kaniecki RG. A comparison of divalproex with propranolol and placebo for the prophylaxis of migraine without aura. Arch Neurol 1997; 54(9):1141–1145. [DOI] [PubMed] [Google Scholar]
- 102.Kass B, Nestvold K. Propranolol (Inderal) and clonidine (Catapressan) in the prophylactic treatment of migraine. A comparative trial. Acta Neurol Scand 1980; 61(6):351–356. [DOI] [PubMed] [Google Scholar]
- 103.Kaushik R, Kaushik RM, Mahajan SK, Rajesh V. Biofeedback assisted diaphragmatic breathing and systematic relaxation versus propranolol in long term prophylaxis of migraine. Complement Ther Med 2005; 13(3):165–174. 10.1016/j.ctim.2005.04.004 [DOI] [PubMed] [Google Scholar]
- 104.Kjaersgard Rasmussen MJ, Holt LB, Borg L, Soelberg SP, Hansen PE. Tolfenamic acid versus propranolol in the prophylactic treatment of migraine. Acta Neurol Scand 1994; 89(6):446–450. [DOI] [PubMed] [Google Scholar]
- 105.Klapper JA. An open label cross-over comparison of divalproex sodium and propranolol HCl in the prevention of migraine headaches. HEADACHE Q 1994; 5:50–53. [Google Scholar]
- 106.Kozubski W, Prusinski A. [Sodium valproate versus propranolol in the prophylactic treatment of migraine]. Neurol Neurochir Pol 1995; 29(6):937–947. [PubMed] [Google Scholar]
- 107.Kuritzky A, Hering R. Prophylactic treatment of migraine with long acting propranolol—a comparison with placebo. Cephalalgia 1987; 7:457–458. [Google Scholar]
- 108.Langohr HD, Gerber WD, Koletzki E, Mayer K, Schroth G. Clomipramine and metoprolol in migraine prophylaxis—a double-blind crossover study. Headache 1985; 25(2):107–113. [DOI] [PubMed] [Google Scholar]
- 109.Louis P, Schoenen J, Hedman C. Metoprolol v. clonidine in the prophylactic treatment of migraine. Cephalalgia 1985; 5(3):159–165. 10.1046/j.1468-2982.1985.0503159.x [DOI] [PubMed] [Google Scholar]
- 110.Lucking CH, Oestreich W, Schmidt R, Soyka D. Flunarizine vs. propranolol in the prophylaxis of migraine: two double-blind comparative studies in more than 400 patients. Cephalalgia 1988; 8 Suppl 8:21–26. [DOI] [PubMed] [Google Scholar]
- 111.Ludin HP. Flunarizine and propranolol in the treatment of migraine. Headache 1989; 29(4):219–224. [DOI] [PubMed] [Google Scholar]
- 112.Maissen CP, Ludin HP. [Comparison of the effect of 5-hydroxytryptophan and propranolol in the interval treatment of migraine]. Schweiz Med Wochenschr 1991; 121(43):1585–1590. [PubMed] [Google Scholar]
- 113.Malvea BP, Gwon N, Graham JR. Propranolol prophylaxis of migraine. Headache 1973; 12(4):163–167. [DOI] [PubMed] [Google Scholar]
- 114.Mathew NT. Prophylaxis of migraine and mixed headache. A randomized controlled study. Headache 1981; 21(3):105–109. [DOI] [PubMed] [Google Scholar]
- 115.Mikkelsen B, Pedersen KK, Christiansen LV. Prophylactic treatment of migraine with tolfenamic acid, propranolol and placebo. Acta Neurol Scand 1986; 73(4):423–427. [DOI] [PubMed] [Google Scholar]
- 116.Nadelmann JW, Phil M, Stevens J, Saper JR. Propranolol in the prophylaxis of migraine. Headache 1986; 26(4):175–182. [DOI] [PubMed] [Google Scholar]
- 117.Nambiar NJ, Aiyappa C, Srinivasa R. Oral riboflavin versus oral propranolol in migraine prophylaxis: An open label randomized controlled trial. 2011; 16(3):223–229. [Google Scholar]
- 118.Nanda RN, Johnson RH, Gray J, Keogh HJ, Melville ID. A double blind trial of acebutolol for migraine prophylaxis. Headache 1978; 18(1):20–22. [DOI] [PubMed] [Google Scholar]
- 119.Olerud B, Gustavsson CL, Furberg B. Nadolol and propranolol in migraine management. Headache 1986; 26(10):490–493. [DOI] [PubMed] [Google Scholar]
- 120.Olsson JE, Behring HC, Forssman B, Hedman C, Hedman G, Johansson F et al. Metoprolol and propranolol in migraine prophylaxis: a double-blind multicentre study. Acta Neurol Scand 1984; 70(3):160–168. [DOI] [PubMed] [Google Scholar]
- 121.Palferman TG, Gibberd FB, Simmonds JP. Prophylactic propranolol in the treatment of headache. Br J Clin Pract 1983; 37(1):28–29. [PubMed] [Google Scholar]
- 122.Pita E, Higueras A, Bolanos J, Perez N, Mundo A. Propranolol and migraine. A clinical trial. Arch Farmacol Toxicol 1977; 3(3):273–278. [PubMed] [Google Scholar]
- 123.Pradalier A, Serratrice G, Collard M, Hirsch E, Feve J, Masson M et al. Long-acting propranolol in migraine prophylaxis: results of a double-blind, placebo-controlled study. Cephalalgia 1989; 9(4):247–253. 10.1046/j.1468-2982.1989.0904247.x [DOI] [PubMed] [Google Scholar]
- 124.Ryan RE Sr., Ryan RE Jr., Sudilovsky A. Nadolol and placebo comparison study in the prophylactic treatment of migraine. Panminerva Med 1982; 24(2):89–94. [PubMed] [Google Scholar]
- 125.Ryan RE Sr. Comparative study of nadolol and propranolol in prophylactic treatment of migraine. Am Heart J 1984; 108(4 Pt 2):1156–1159. [DOI] [PubMed] [Google Scholar]
- 126.Sargent J, Solbach P, Damasio H, Baumel B, Corbett J, Eisner L et al. A comparison of naproxen sodium to propranolol hydrochloride and a placebo control for the prophylaxis of migraine headache. Headache 1985; 25(6):320–324. [DOI] [PubMed] [Google Scholar]
- 127.Schellenberg R, Lichtenthal A, Wohling H, Graf C, Brixius K. Nebivolol and metoprolol for treating migraine: an advance on beta-blocker treatment? Headache 2008; 48(1):118–125. 10.1111/j.1526-4610.2007.00785.x [DOI] [PubMed] [Google Scholar]
- 128.Shimell CJ, Fritz VU, Levien SL. A comparative trial of flunarizine and propranolol in the prevention of migraine. S Afr Med J 1990; 77(2):75–77. [PubMed] [Google Scholar]
- 129.Silberstein SD, Dodick DW, Lindblad AS, Holroyd K, Harrington M, Mathew NT et al. Randomized, placebo-controlled trial of propranolol added to topiramate in chronic migraine. Neurology 2012; 78(13):976–984. 10.1212/WNL.0b013e31824d5846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Siniatchkin M, Andrasik F, Kropp P, Niederberger U, Strenge H, Averkina N et al. Central mechanisms of controlled-release metoprolol in migraine: a double-blind, placebo-controlled study. Cephalalgia 2007; 27(9):1024–1032. 10.1111/j.1468-2982.2007.01377.x [DOI] [PubMed] [Google Scholar]
- 131.Sorensen PS, Larsen BH, Rasmussen MJ, Kinge E, Iversen H, Alslev T et al. Flunarizine versus metoprolol in migraine prophylaxis: a double-blind, randomized parallel group study of efficacy and tolerability. Headache 1991; 31(10):650–657. [DOI] [PubMed] [Google Scholar]
- 132.Soyka D, Oestreich W. Flunarizine versus propranolol in interval treatment of migraine. Nervenheilkunde 1990; 9:45–51. [Google Scholar]
- 133.Standnes B. The prophylactic effect of timolol versus propranolol and placebo in common migraine: beta-blockers in migraine. Cephalalgia 1982; 2(3):165–170. 10.1046/j.1468-2982.1982.0203165.x [DOI] [PubMed] [Google Scholar]
- 134.Steiner TJ, Joseph R, Hedman C, Rose FC. Metoprolol in the prophylaxis of migraine: parallel-groups comparison with placebo and dose-ranging follow-up. Headache 1988; 28(1):15–23. [DOI] [PubMed] [Google Scholar]
- 135.Stellar S, Ahrens SP, Meibohm AR, Reines SA. Migraine prevention with timolol. A double-blind crossover study. JAMA 1984; 252(18):2576–2580. [PubMed] [Google Scholar]
- 136.Stensrud P, Sjaastad O. Short-term clinical trial of phopranolol in racemic form (Inderal), D-propranolol and placebo in migraine. Acta Neurol Scand 1976; 53(3):229–232. [DOI] [PubMed] [Google Scholar]
- 137.Stensrud P, Sjaastad O. Comparative trial of Tenormin (atenolol) and Inderal (propranolol) in migraine. Headache 1980; 20(4):204–207. [DOI] [PubMed] [Google Scholar]
- 138.Stovner LJ, Linde M, Gravdahl GB, Tronvik E, Aamodt AH, Sand T et al. A comparative study of candesartan versus propranolol for migraine prophylaxis: A randomised, triple-blind, placebo-controlled, double cross-over study. Cephalalgia 2013; 34(7):523–532. 10.1177/0333102413515348 [DOI] [PubMed] [Google Scholar]
- 139.Streng A, Linde K, Hoppe A, Pfaffenrath V, Hammes M, Wagenpfeil S et al. Effectiveness and tolerability of acupuncture compared with metoprolol in migraine prophylaxis. Headache 2006; 46(10):1492–1502. 10.1111/j.1526-4610.2006.00598.x [DOI] [PubMed] [Google Scholar]
- 140.Sudilovsky A, Elkind AH, Ryan RE Sr., Saper JR, Stern MA, Meyer JH. Comparative efficacy of nadolol and propranolol in the management of migraine. Headache 1987; 27(8):421–426. [DOI] [PubMed] [Google Scholar]
- 141.Tfelt-Hansen P, Standnes B, Kangasneimi P, Hakkarainen H, Olesen J. Timolol vs propranolol vs placebo in common migraine prophylaxis: a double-blind multicenter trial. Acta Neurol Scand 1984; 69(1):1–8. [DOI] [PubMed] [Google Scholar]
- 142.van de Ven LL, Franke CL, Koehler PJ. Prophylactic treatment of migraine with bisoprolol: a placebo-controlled study. Cephalalgia 1997; 17(5):596–599. 10.1046/j.1468-2982.1997.1705596.x [DOI] [PubMed] [Google Scholar]
- 143.Vilming S, Standnes B, Hedman C. Metoprolol and pizotifen in the prophylactic treatment of classical and common migraine. A double-blind investigation. Cephalalgia 1985; 5(1):17–23. 10.1046/j.1468-2982.1985.0501017.x [DOI] [PubMed] [Google Scholar]
- 144.Weber RB, Reinmuth OM. The treatment of migraine with propranolol. Neurology 1971; 21:404–405. [DOI] [PubMed] [Google Scholar]
- 145.Wideroe TE, Vigander T. Propranolol in the treatment of migraine. Br Med J 1974; 2(5921):699–701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Worz R, Reinhardt-Benmalek B, Foh M, Grotemeyer KH, Scharafinski HW. [Prevention of migraine using bisoprolol. Results of a double-blind study versus metoprolol]. Fortschr Med 1992; 110(14):268–272. [PubMed] [Google Scholar]
- 147.Nair KG. A pilot study of the value of propranolol in migraine. J Postgrad Med 1975; 21(3):111–113. [PubMed] [Google Scholar]
- 148.Chen J. Clinical study of topiramate in preventing migraine attacks. Sichuan Medical Journal 2009; 30(1):119. [Google Scholar]
- 149.Ziegler DK, Hurwitz A, Preskorn S, Hassanein R, Seim J. Propranolol and amitriptyline in prophylaxis of migraine. Pharmacokinetic and therapeutic effects. Arch Neurol 1993; 50(8):825–830. [DOI] [PubMed] [Google Scholar]
- 150.Gong X, Zhang J. Therapeutic effect of metoprolol tartrate combined with flunarizine on migraine in the treatment of migraine. Journ of Integrative Medicine, Cardiovascular and Cerebrovascular Diseases 2016; 25(14):24–2957. [Google Scholar]
- 151.Ke D. Sibiling combined with propranolol to treat migraine. Modern Practical Medicine 2003; 15(12):760. [Google Scholar]
- 152.Li P. Double-blind therapeutic effect of â-blockers on menopausal migraine. Chinese Journal of Practical Nervous Diseases 2006; 9(5):120–121. [Google Scholar]
- 153.Li S. Therapeutic effect of sodium valproate on prevention of migraine recurrence. Sichuan Medical Journal 2004; 25(4):416. [Google Scholar]
- 154.Ma W. Therapeutic effect of metoprolol tartrate combined with flunarizine on migraine in the treatment of migraine. Modern Practical Medicine 2011; 23(11):1275–1276. [Google Scholar]
- 155.Wen JB. Clinical effect analysis of sibelium prophylactic treatment of migraine. Contemporary Medicine 2016; 22(6):141. [Google Scholar]
- 156.Yang Y, Wang G. Observation on the efficacy of propranolol in preventing menopausal migraine. Chinese Journal of Misdiagnosis 2006; 6(24):4760–4761. [Google Scholar]
- 157.Yang Y. The effect of metoprolol combined with fluoxetine on preventing migraine and its effect on serotonin. Chinese Journal of Modern Drug Application 2016; 10(6):109–110. [Google Scholar]
- 158.Yuan Q, Lu X. Preliminary randomized controlled trial of topiramate and propranolol to prevent migraine attacks. Huaxi Pharmaceutical Journal 2005; 20(2):182–183. [Google Scholar]
- 159.Zhou J, Zhu Y, Zheng W, Chen Y, Lin L, Tian X. The efficacy of metoprolol combined with fluoxetine in preventing migraine. Journal of Clinical and Experimental Medicine 2015; 14(10):836–839. [Google Scholar]
- 160.Zhu J, Wei Q, Yan T. Therapeutic effect of sibelium on migraine. Journal of Medical Forum 2005; 26(1):51–52. [Google Scholar]
- 161.Li Z, Li X, Xiong X. A clinical study of Sibelium combining Topiramate in prophylactic treatment of migraine. Chinese Journal of Primary Medicine and Pharmacy 2002; 19(5):419–420. [Google Scholar]
- 162.Sjaastad O, Stensrud P. Clinical trial of a beta-receptor blocking agent (LB 46) in migraine prophylaxis. Acta Neurol Scand 1972; 48(1):124–128. [DOI] [PubMed] [Google Scholar]
- 163.Dongxiang H. Clinical trial of propranolol and amitriptyline in the treatment of migraine. Yunnan Medicine 2010; 31(5):523–525. [Google Scholar]
- 164.Li Z, Li X, Xiong X. A clinical study of sibelium combining topiramate in prophylactic treatment of migraine. Chinese JMAP 2002; 19(5):419–421. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOCX)
(DOCX)
Data Availability Statement
Dara are available at https://doi.org/10.6084/m9.figshare.7674092.