Abstract
Rationale:
Hepatoid adenocarcinoma (HAC) of the fallopian tubes is a rare malignant tumor in the female reproductive system.
Patient concerns:
An 81-year-old Chinese woman presented with an elevated serum alpha-fetoprotein (AFP) level.
Diagnosis:
Positron emission tomography-computed tomography (PET-CT) scan revealed a mass of approximately 47 × 27 mm located in the right adnexa. The tumor was diagnosed as a HAC arising from fallopian tube by immunohistochemical and histochemical technique.
Interventions:
This patient underwent surgical treatment including a bilateral adnexectomy and appendectomy. In addition, the patient underwent 5 cycles of postoperative chemotherapy.
Outcomes:
The disease has recurred approximately six months after surgery and therefore, this patient will continue to be observed.
Lessons:
Up to this point, only 4 known cases of HAC originating in fallopian tube have been published in the English literature. Further studies are needed to better understand the clinical characteristics, the prognosis, and the pathological mechanism of HAC development in the fallopian tubes.
Keywords: alpha-fetoprotein, fallopian tube, hepatoid adenocarcinoma
1. Introduction
Primary fallopian tube carcinoma (PFTC) is thought to be an extremely rare tumor as it accounts for only 0.1% to 1.8% of all gynecological tumors.[1] Preoperative diagnosis of PFTC is difficult, and a definite diagnosis is mostly base on postoperative pathology. Hepatoid adenocarcinoma (HAC) is an aggressive, rare, and malignant cancer that develops in the extrahepatic organs or tissues, and has a hematogenous metastasis and early lymph node involvement. There are many similarities between tumor cells of HAC and hepatocytes in morphological and immunohistochemical features.[2] Here we present a very rare case of HAC that arose in the fallopian tube, and discuss the clinicoradiological and pathological findings of this tumor. To the best of our knowledge, only 4 cases of HAC originating in fallopian tube have been published in the English literature thus far.
2. Case report
An 81-year-old Chinese woman (gravida 3, para 2) was admitted to our hospital with an elevated serum alpha-fetoprotein (AFP) level. Approximately 12 months before the patient presented to us, her routine medical report indicated that her serum AFP level was slightly elevated (36.4 ng/mL). Eight months later, her serum AFP level rose significantly to above 1210.0 ng/mL; however, she did not have any symptoms such as jaundice, abdominal pain, gastrointestinal bleeding, or vaginal bleeding. She had experienced menopause approximately 32 years prior, before which, she had a normal menstrual cycle. She suffered from hypertension for more than 20 years but kept her blood pressure under control. She has not had a history of viral hepatitis or malignant cancer. She had no previous exposure to alcohol tobacco, and toxic carcinogens. Laboratory tests showed results within the normal range except her serum level of AFP were markedly elevated at 2594.5 ng/mL.
Positron emission tomography-computed tomography (PET-CT) scan revealed a mass of approximately 47 × 27 mm located in the right adnexa (Fig. 1A). The mass showed noticeable uptake of 18F-fluorodeoxyglucose (18F-FDG) on PET-CT, with the maximum standard uptake value (SUV) being 7.69 (Fig. 1C). The uptake of 18F-FDG in the uterus and the left adnexa was within normal limits. In addition, multiple lymph nodes surrounding the inferior vena cava showed remarkable uptake on PET-CT, with the maximal SUV being 6.12. The uptake of 18F-FDG in liver was within the normal limits (Fig. 1B).
Figure 1.
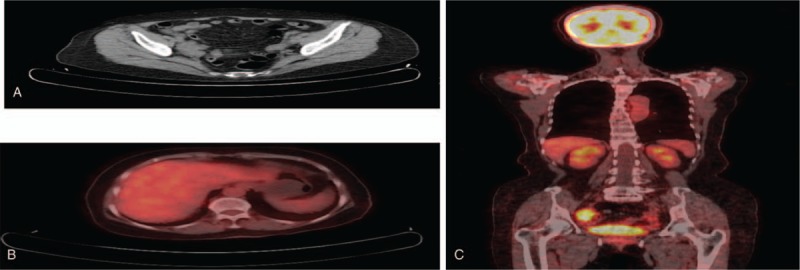
The Positron emission tomography-computed tomography (PET-CT) scan of upper abdomen found a mass about 47 × 27 mm located in the right adnexa (A); PET-CT scan demonstrated FDG normal uptake by the liver(B); PET-CT scan demonstrated 18F-FDG remarkable uptake by the mass(C). PET-CT = positron emission tomography- computed tomography.
A laparoscopic exploration was performed to confirm the patient's diagnosis and to determine the best treatment options. Intraoperatively, there was approximately 200 mL of fluid in the abdomen due to ascites, and an irregular friable mass was identified in the right fallopian tube, measuring 5 cm × 5 cm × 4 cm, which adhered to the right ovary, appendix, intestines, and right pelvic wall. No abnormalities were found in the liver, and its surface was smooth. Bilateral adnexectomy and appendectomy were performed. Pathological analysis of an intra-operative frozen section suggested a diagnosis of a malignant tumor.
Grossly, the specimen consisted of an irregular friable mass near the right adnexa, the left adnexa, and epityphlon. A histological examination revealed that the tumor was a hepatoid adenocarcinoma of the right fallopian tube and was also beginning to infiltrate the right ovary and appendix, but the left adnexa was not invaded. Microscopically, a large amount of round to polygonal adenocarcinoma cells were seen by the tumor. These poorly differentiated cells had large nuclei and were abundant, eosinophilic cytoplasm, but the cytoplasm of some cells was vacuous. Numerous Mallory bodies and hyaline globules were found in the cytoplasm of some tumor cells. These tumor cells arranged in tubular, sinusoidal, and nest bulk cords (Fig. 2A, B). In immunohistochemistry, tumor cells showed positive staining for AFP(+++) (Fig. 2C), EMA(+++) (Fig. 2D), Gly-3(++) (Fig. 2E), SALL-4(+++) (Fig. 2F), PLAP(++), P53(+++), P16(+++), Sox-2(+), CD15(+) and 85%Ki67, but negative staining for CK20, ER, PR, WT-1, CK7, Melan-A and D2-40.
Figure 2.

Hematoxylin-eosinstain stain and immunohistochemistry of the fallopian tube tumor. The tumor cells arranged in the tubular, sinusoidal and nest bulk cords (A, hematoxylin-eosinstain stain ×200); The tumor was composed of round to polygonal poorly differentiated cells (B, hematoxylin-eosinstain stain ×400); The tumor cells were positive for AFP (C, immunostaining ×200); The tumor cells were positive for EMA (D, immunostaining ×200); The tumor cells were positive for Gly-3 (E, immunostaining ×200); The tumor cells were positive for SALL-4(F, immunostaining ×200). AFP = alpha-fetoprotein, SALL4 = Sal-like protein 4.
In accordance with tubal cancer staging of the International Federation of Gynecology and Obstetrics (FIGO), the tumor was staged as IIIc. The patient underwent 3 cycles of postoperative chemotherapy with paclitaxel (PTX, 90 mg/day, day 1, 15, 30) and carboplatin (CBP, 150 mg/day, day 1, 15, 30) and after 3 cycles of chemotherapy with PTX and CBP, the serum AFP markedly was elevated (Fig. 3). Therefore, we changed the chemotherapy and the patient received a fourth cycle of vincristine (VCR, 1 mg/day, day1), kengshemycinl (KSM, 200 ug/day1, 300 ug/day2, 200 ug/day3), and cyclophosphamide (CTX, 200 mg/day1, 200 mg/day 2, 300 mg/day3). After the fourth cycle of chemotherapy, the patient had obvious side effects including nausea, vomiting and bone marrow suppression. Because she did not tolerate the adverse drug reaction, the fifth cycle of chemotherapy was changed with oxaliplatin (OXA, 50 mg/day, day 1), PTX (40 mg/day, day 2) and bevacizumab (BEV, 300 mg/day, day 2). After the 5 cycles chemotherapy, the transabdominal color doppler scanning found a 7.2 cm × 2.7 cm × 5.7 cm mass in her abdomen which was considered a recurrence. The disease has recurred approximately six months after surgery and therefore, this patient will continue to be observed. Our patient provided informed consent, and the research was approved by the appropriate ethics review board.
Figure 3.
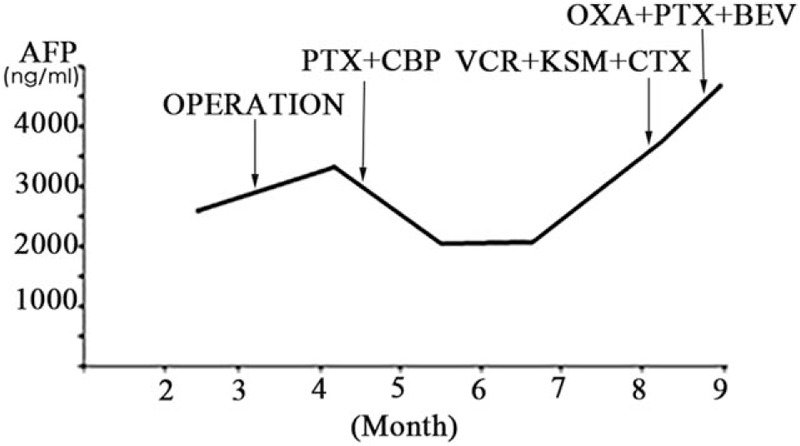
The change of serum APF in the therapeutic procedure. BEV = bevacizumab, CBP = carboplatin, CTX = cyclophosphamide, KSM = kengshemycinl, OXA = oxaliplatin, PTX = paclitaxel, VCR = vincristine.
3. Discussion
Primary HAC is a highly malignant disease and the prognosis is exceedingly poor. HAC often arises in extrahepatic organs or tissues such as the stomach, ovaries, lungs, gallbladder, pancreas, and uterus;[3] overall, the incidence of tubal HAC is low. Primary ovarian HAC is regularly found in post-menopausal women aged 42 to 78 years.[4] Only eight cases of HAC affecting the uterus have been reported to date. According to these studies, Primary uterine HAC commonly occurs in post-menopausal women aged 60 to 86 years.[5] The first case identified as tubal HAC was reported by Aoyama in 1996 and since then only 4 cases have been reported to be tubal HAC[6–9] (Table 1). According to these cases, tubal HAC is a serious disease with nonspecific symptoms. The average peak age of tubal HAC incidence is between 52 and 81 years.
Table 1.
Summary of the previous cases.

In histopathology, HAC is exceedingly similar to hepatocellular carcinoma (HCC). Our patient's tumor showed morphological and functional similarities to HCC such as a trabecular cellular growth pattern and paucity of mucin. Oncofetal proteins such AFP, PLAP, and Gly-3 were been long thought to be expressed by the fetal liver, yolk sac tumors, hepatoblastomas, and HCC.[10,11] Generally, Sal-like protein 4 (SALL4) expression is mainly discovered in primitive germ cell tumors and seldomly is found in normal liver tissue or HCCs.[11] The Immunohistochemistry results show that tumor cells are generally positive for AFP and PLAP but not for CK7 and CK20. Therefore, SALL4 expression may be useful for distinguishing HAC from HCCs.
Preoperative diagnosis of tubal HAC is exceeding difficult. The differential diagnosis of HAC should include ovarian HAC, a yolk sac tumor (YST) and metastatic hepatic carcinoma. Ovarian HAC also occurs in older women.[12] But based on the morphological and immunohistochemical features of the tumor cells, there are considered to arising from the fallopian tube. Because YST always has increasing of serum AFP, it is often associated with HAC. It is important to note that YST always appear in young women.[9] In addition, the pathologic findings and patient history in this case do not support the diagnosis of YST. For example, the patient does not have a history of hepatitis or HCC, and no liver tumors have been detected. The PET-CT showed that the uptake of 18F-fluorodeoxyglucose in the hepatic parenchyma was within a normal range.
Effective treatment of fallopian tube HAC is unclear and unified. Surgical resection combined with postoperative chemotherapy may reduce the risk of recurrence and prolong overall survival.[13] A hysterectomy and bilateral salpingo-oophorectomy were performed in the previous 4 cases (Table 1) but because our patient was a senior and had hypertension, a bilateral adnexectomy and appendectomy were performed. Postoperative chemotherapy may be imperative to reduce recurrence of this disease. Platinum-based drugs were administered in most of the previous cases, but the treatment of 1 case was unclear. Moreover, serum AFP was correlated with the prognosis of HAC.[4,14] After surgery, chemotherapy and targeted therapy were provided for our patient, the level of AFP was significantly fluctuant (Fig. 3). This tumor rapidly reappeared 6 months after surgery. In the Aoyama et al[6] study, they found that the patient survived more than 2 years with an HAC of the fallopian tube. Another study by Fukunaga et al showed that the patient had no progression for 10 months after surgery.[7] The median survival was approximately 2 years in patients diagnosed HAC of the ovaries according to Mazouz et al.[4] In the Ishibashi et al[5] study, the prognosis of uterine HAC was very poor and the patients died or the tumor rapidly relapsed within 12 months in almost studies included. Overall, the outcome, prognosis, and treatment effect of tubal, ovarian and uterine HAC was not optimal. However, further studies will need to be conducted to better understand the clinical characteristics, prognosis, and pathological mechanisms of tubal HAC development.
4. Conclusion
Tubal HAC is a rare malignant tumor in the female reproductive system and is a serious disease with nonspecific symptoms. Preoperative diagnosis of tubal HAC is extremely difficult, and definite diagnosis mostly base on postoperative pathology. The average peak age of tubal HAC incidence is between 52 and 81 years. Surgical resection combined with postoperative chemotherapy may reduce the risk of recurrence and prolong overall survival. In addition, targeted therapy may be an alternative treatment for those patients whose serum AFP is hardly decreased after surgery and chemotherapy. So far, only 5 studies regarding tubal HAC are found and further studies will need to be conducted to help better select the treatment of tubal HAC.
Author contributions
Conceptualization: Maomao Li, Ping Wang.
Data curation: Maomao Li.
Formal analysis: Maomao Li.
Funding acquisition: Ping Wang.
Investigation: Maomao Li.
Methodology: Maomao Li, Kaixuan Yang.
Project administration: Maomao Li.
Resources: Ping Wang.
Software: Maomao Li.
Supervision: Maomao Li.
Validation: Maomao Li.
Visualization: Maomao Li.
Writing – original draft: Maomao Li.
Writing – review & editing: Maomao Li.
Maomao Li orcid: 0000-0003-2429-5918.
Footnotes
Abbreviations: AFP = alpha-fetoprotein, BEV = bevacizumab, CBP = carboplatin, CTX = cyclophosphamide, HAC = hepatoid adenocarcinoma, HCC = hepatocellular carcinoma, KSM = kengshemycinl, OXA = oxaliplatin, PET-CT = positron emission tomography- computed tomography, PFTC = primary fallopian tube carcinoma, PTX = paclitaxel, SALL4 = Sal-like protein 4, SUV = standard uptake value, VCR = vincristine, YST = yolk sac tumor.
This work was supported by the Key Research Programs of Science and Technology Department of Sichuan Province (No. 2018FZ0057).
The authors have no conflicts of interest.
References
- [1].You D, Wang Q, Jiang W, et al. Primary leiomyosarcoma of the fallopian tube: a case report and literature review. Medicine (Baltimore) 2018;97:e0536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Yorita K, Sasaki S, Kawada A, et al. Hepatoid adenocarcinoma of the extrahepatic bile duct in a patient with polysplenia syndrome. Intern Med 2017;56:401–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Dogeas E, Peng L, Choti MA. Hepatoid adenocarcinoma of unknown primary masquerading as a pancreatic tumor. J Gastrointest Surg 2017;21:2132–4. [DOI] [PubMed] [Google Scholar]
- [4].Mazouz A, Amaadour L, Ameurtesse H, et al. Primary hepatoid carcinoma of the ovary: a case report. Pan Afr Med J 2015;20:93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Ishibashi K, Kishimoto T, Yonemori Y, et al. Primary hepatoid adenocarcinoma of the uterine corpus: a case report with immunohistochemical study for expression of liver-enriched nuclear factors. Pathol Res Pract 2011;207:332–6. [DOI] [PubMed] [Google Scholar]
- [6].Aoyama T, Mizuno T, Andoh K, et al. alpha-Fetoprotein-producing (hepatoid) carcinoma of the fallopian tube. Gynecol Oncol 1996;63:261–6. [DOI] [PubMed] [Google Scholar]
- [7].Fukunaga M, Fujiwara Y, Naito Z. Hepatoid carcinoma with serous component of the fallopian tube: a case report with immunohistochemical and ultrastructural studies. Int J Gynecol Pathol 2006;25:233–7. [DOI] [PubMed] [Google Scholar]
- [8].Ishiwata I, Yasuda M, Hirano T, et al. Establishment and characterization of HEPFT, a cell line derived from hepatoid carcinoma of the fallopian tube, with special reference to alpha-fetoprotein, lectin affinity and histogenesis. Hum Cell 2007;20:119–30. [DOI] [PubMed] [Google Scholar]
- [9].D’Antonio A, Sparano L, Addesso M, et al. Extraovarian yolk sac tumour in an elderly woman with an endometrioid-like pattern concurrent with a hepatoid component. J Obstet Gynaecol 2010;30:530–2. [DOI] [PubMed] [Google Scholar]
- [10].Wee A. Diagnostic utility of immunohistochemistry in hepatocellular carcinoma, its variants and their mimics. Appl Immunohistochem Mol Morphol 2006;14:266–72. [DOI] [PubMed] [Google Scholar]
- [11].Yoshizawa J, Ishizone S, Ikeyama M, et al. Gastric hepatoid adenocarcinoma resulting in a spontaneous gastric perforation: a case report and review of the literature. BMC Cancer 2017;17:368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Mahmood H, Fatima H, Faheem M. Metastatic hepatoid carcinoma of ovarian origin - a case report from northern Pakistan. Gynecol Oncol Rep 2017;21:24–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Jin R, Yu Q, Liang X. Ectopic hepatocellular carcinoma manifesting multiple abdominal masses: a case report. Medicine (Baltimore) 2017;96:e8968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Lee CH, Huang KG, Ueng SH, et al. A hepatoid carcinoma of the ovary. Acta Obstet Gynecol Scand 2002;81:1080–2. [DOI] [PubMed] [Google Scholar]


