Abstract
Gene expression is tightly regulated in time and space through a multitude of factors consisting of signaling molecules. Soluble N-ethylmaleimide-sensitive-factor attachment protein receptors (SNARE) are membrane proteins responsible for the intercellular trafficking of signals through endocytosis and exocytosis of vesicles. Altered expression of SNARE proteins in cellular communication is the major hallmark of cancer phenotypes as indicated in recent studies. SNAREs play an important role in maintaining cell growth and epithelial membrane permeability of the bladder and are not only involved in cancer progression but also metastatic cell invasion through SNARE-mediated trafficking. Synaptobrevin2/Vesicle associated membrane protein-2 (v-SNARE) and Syntaxin (t-SNARE) form a vesicular docking complex during endocytosis. Some earlier studies have shown a critical role of SNARE in colon, lungs, and breast cancer progression and metastasis. In this study, we analyzed the relative expression of the STX1A and VAMP2 (SYB2) for their possible association in the progression and metastasis of bladder cancer. The profiling of the genes showed a significant increase in STX1A and VAMP2 expression (p < 0.001) in high-grade tumor cells compared to normal and low-grade tumors. These findings suggest that elevated expression of STX1A and VAMP2 might have caused the abnormal progression and invasion of cancer cells leading to the transformation of cells into high-grade tumor in bladder cancer.
Keywords: SNARE, bladder cancer, vesicle fusion, gene expression
Introduction
Transitional cell carcinoma is the common form of histologic bladder cancer (90% cases) and has significant mortality rate (77.89% 5-year relative survival) (American Cancer Society, 2017). High grade tumors have high probability of recurrence with high percentage of progression while low grade tumors have low frequency of recurrence and are less progressive (Miyamoto et al., 2010). According to WHO in 2004 (WHO/International Society of Urological Pathology (ISUP) classification), the classification of bladder cancer is useful in differentiating carcinomas for prognostic evaluation (Pan et al., 2010). Low grade papillary urothelial carcinomas (LPUCs) and high grade papillary urothelial carcinomas (HPUCs) have distinct cancer progression categories and recurrence, and therefore, WHO recently recommended the staging of bladder cancer into only two categories: low grade and high grade (Miyamoto et al., 2010; Pan et al., 2010).
In almost all of the cancers, signal transduction dysregulation has a key role in triggering the cell for survival in malignant conditions (Bartsch et al., 2010). The tumor microenvironment plays a crucial role in maintaining the tumor growth, progression, and metastasis via exploiting growth factors, enzymes, and other signaling molecules that are preferably transported via exosomes (Kang et al., 2017). Proteome analysis of extracellular vesicles (EVs) secreted by the epithelial membrane in muscle invasive bladder cancer (MIBC) showed that these vesicles contain a number of proteins and signaling molecules that are transported to extracellular matrix (ECM) (Silvers et al., 2017). The vesicle trafficking is basically controlled by regulatory receptor proteins present on the membrane of the targeting cell, functioning with the aid of gated channels (Palfreyman and Jorgensen, 2008). Membrane trafficking in the eukaryotic cell is mediated by a SNARE complex [soluble (N-ethylmaleimide-sensitive factor) attachment protein receptors] (Shukla et al., 2000).
The SNARE complex is divided into two groups according to function and location. t-SNAREs are target membrane receptors while v-SNAREs are vesicular membrane proteins (Polchi et al., 2013). Syntaxin-1 (STX1) and SNAP25 are t-SNAREs, resident on the target cell membrane and participate in vesicle fusion whereas VAMPs (vesicle associated membrane proteins) are v-SNAREs, anchored to the membrane vesicles excreted or exocytosed by the cell (Haberman et al., 2012; Meng and Wang 2015). The vesicular fusion is accomplished by core SNARE complex comprising such as syntaxin (STX), SNAP25, and synaptobrevin2 (VAMP2) that mediate final vesicle fusion (Fang and Lindau, 2014).
SNARE proteins are known to actively derive vesicular trafficking between the cells to maintain the cell integrity via cell growth, migration, and wound healing in a regulative manner (Tian et al., 2014). Delivery of extracellular matrix (ECM) and integrins through vesicular transport is the fundamental function of SNARE proteins during cell proliferation and motility. Though SNARE-mediated exosome transport of integrins is critical for cancer development, epidermal growth factors at the cell surface have a major role in ECM regulation, cell survival, and progression (Enrich et al., 2015). SNARE proteins regulate matrix degradation and allow cell migration/invasion (Williams et al., 2014). Functional silencing of SNARE proteins decreases the ability of breast cancer cells to invade and migrate (Steffen et al., 2008). Inhibition of SNARE proteins impairs the development of invadopodium, disrupts cell invasion, and inhibits migration in tumors (Williams and Coppolino, 2014). Altered expression of the SNARE complex has been found critical for various cancers as they are the core signaling proteins involved in vesicular fusion and known to be good targets for cancer therapy (Meng and Wang, 2015).
STX1A and VAMP2 are known neuronal SNAREs that mediate synaptic vesicular fusion (Ramakrishnan et al., 2012). STX1A overexpression has also been observed in primary brain tumor and colorectal, lung, and breast cancers (Grabowski et al., 2002; Arsenault et al., 2013; Fernández-Nogueira et al., 2015). Blocking of STX1A inhibits tumor growth in glioblastoma (Ulloa et al., 2015). Little is known about the expression pattern of VAMP2 in breast and lung cancers and also in bladder cancer. However, loss of VAMP2 in neuronal tissue leads to endolysosomal degradation. VAMP2 relies on its sorting behavior for vesicle exocytosis and fusion with target sites. Decreased expression of VAMP2 causes abnormalities in the degradation pattern of useless proteins (Haberman et al., 2012). Heterogenic expression of VAMP2 and other SNARE proteins was found in undifferentiated colorectal carcinomas (Grabowski et al., 2002). Importantly, VAMP2 is known to be involved in the integrin trafficking and critical for cancer cell adhesion, survival, and migration (Hasan and Hu, 2010). SNAREs are thus basic complexes in exosome-mediated cellular communication that regulate the cell cycle and progression (Polchi et al., 2013). According to recent studies on the role of neuronal SNARE complex, specifically STX1A and VAMP2, in cancers other than brain tumors as regulators of important cellular mechanism of vesicular exocytosis, the heterogenic expression of these genes may cause cellular abnormalities leading to cancer development. Therefore, we aimed to determine the difference in expression of STX1A and VAMP2 in relation to tumor grades and pathological stages in bladder cancer.
Material and Methods
Tumor sampling
Tumor and normal tissue samples were collected from post-surgical bladder cancer specimens. The study was approved by the ethical review board (ERB) of COMSATS Institute of Information Technology (No. CIIT/Bio/ERB/18/76). The data were obtained with the written consent of the patients involved in the study. Disease histories were confirmed by the Department of Pathology, Pakistan Institute of Medical Science (PIMS). The total number of samples was 55, out of which 26 were paired. Surrounding normal tissue samples were used as controls. The histopathology reports of the patients were obtained from the Department of Urology PIMS and Shifa International Hospital, Islamabad, Pakistan, for categorizing the tumor samples according to their grades and cancer stage.
Quantitative PCR
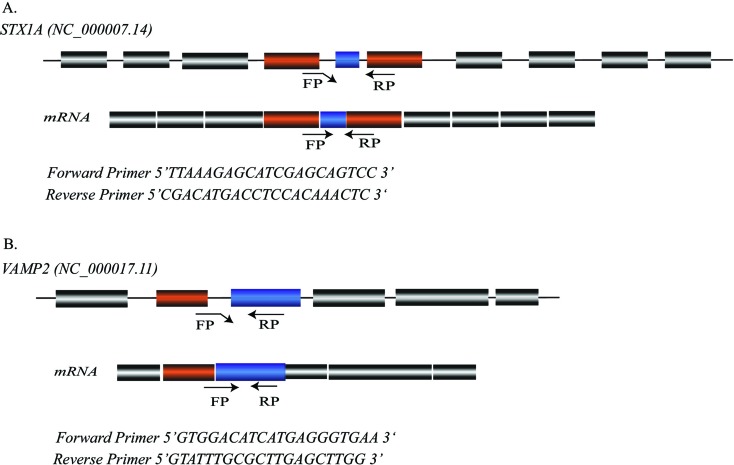
RNAlater® (Ambion, Thermo Fisher Scientific Inc. Waltham, MA USA) was used to preserve samples. RNA was isolated using Trizol (Thermo Fisher Scientific) reagent according to manufacturer’s protocol (Rio et al., 2010). Quantified RNA (1-2 μg) was used for cDNA synthesis (Thermo Fisher Scientific). Primers for the target genes STX1A, VAMP2 were designed using Primer Quest tool (Integrated DNA Technologies) and further edited to acquire specifications. TUB3 was used as the endogenous housekeeping gene. The primers designed were specific to STX1A-001 and VAMP2-001. Primer sequences and specification are given in Figure 1. UCSC in silico PCR was done using the set of primers to assure amplification. Quantitatve analysis of the expression level of the target genes was done by quantitative real-time PCR in a StepOnePlus Real-Time PCR system (Applied Biosystems). The experiment was run for three biological replicates with negative controls. A melting curve analysis for each sample was performed to check for non-targeted fragment amplification. The volume per reaction was adjusted to 25 μL using Maxima Syber Green/ROX qPCR Master Mix (Thermo Scientific), and cDNA was used at a concentration of 2 μg/μL for each sample. The relative fold-increase in the expression of the STX1A and VAMP2 genes was analyzed using the 2-DDCT method (Livak and Schmittgen, 2001). The data were normalized with the internal control TUB3 and the average fold-increase was determined by calculating the relative expressions of each tumor sample.
Figure 1. Primer location and specificity for qPCR analysis. (A) Primer pair sequence for STX1A (NC_000007.14) (FP 5’ TTAAAGAGCATCGAGCAGTCC 3’ and RP 5’ CGACATGACCTCCACAAACTC 3’), Amplicon size is 120 bp, TM=62 °C, location at GRCh38.p12; (Chr7: 73704218-73704237) (Chr7: 73704423-73704410). (B) VAMP2 (NC_000017.11) primer pair sequence; forward primer (5’ GTGGACATCATGAGGGTGAA 3’), reverse primer (5’GTATTTGCGCTTGAGCTTGG 3’). Amplicon size is 138 bp, TM=55 °C, location GRCh38.p12; (Chr17: 8161626-8161645), (Chr17: 8161763-8161744).
Statistical analysis
Statistical analyses were performed with OriginPro 2017 (OriginLab, Northampton, MA). For expression data, the Ct values of the target genes VAMP2 and STX1A were normalized with the control gene (TUB3) Ct. Normality of the data was assessed by the Shapiro-Wilk test. The correlation among different factors was assessed by the Spearman Correlation Coefficient test. Depending on the experiment, the statistical significance was determined using the Wilcoxon, Mann-Whitney, or Kruskal-Wallis ANOVA test, and specific comparisons were made by the Tukey’s Honestly Significant Difference (HSD) test. Fisher’s exact two-tailed test was performed to calculate patient data contingency, with p < 0.05 considered as significant.
Results
Tumor grade and stage
Out of 55 bladder tumor samples, 31 were high grade and 24 were low grade according to the WHO/ISUP classification system (Miyamoto et al., 2010). The histopathology reports of the patients were obtained from the hospitals (Pakistan Institute of Medical Sciences and Shifa International Hospital, Islamabad Pakistan) and, according to the histopathology examination; the tumors that had recurrent behavior were categorized as high grade tumors. The samples were confirmed as transitional cell carcinomas by the Department of Histopathology (PIMS). There were 10 high grade tumors that had metastasized to the pelvic wall and prostate gland. Among the low grade tumors there were seven tumors that had spread only to the sub-epithelial connective tissue of the bladder. The staging of tumors was based on the TNM staging system (Table 1). The significance and distribution of tumor grade among age intervals was calculated using the Chi-square test (Table 2).
Table 1. Histopathology of tumor samples according to TNM staging system.
| Histopathological staging | ||||||||
|---|---|---|---|---|---|---|---|---|
| I | II | III | ||||||
| Grades | Gender | Total NO. | Ta N0 M0 | Tis N0 M0 | T1 N1 M0 | T2 N2 M0 | T2a N1 M0 | T4a N3 M1 |
| High grade | Female | 11 | 0 | 1 | 2 | 3 | 3 | 2 |
| Male | 20 | 0 | 1 | 1 | 4 | 5 | 9 | |
| Low grade | Female | 8 | 2 | 4 | 2 | 0 | 0 | 0 |
| Male | 16 | 5 | 3 | 5 | 3 | 0 | 0 | |
Ta, Noninvasive papillary carcinoma; Tis, non-muscle-invasive bladder cancer; T4a, Tumor spread to the uterus or prostate; N0, Cancer not spread to regional lymph node/s; N1, Cancer spread to single regional lymph node in pelvis; M0, Non-metastasized; M1, Metastasized to pelvic organs.
Table 2. Significance and distribution of tumor grade in young age and older age group.
| Patient data | Age interval | p-value | |
|---|---|---|---|
| 35-40 years | > 40 years | ||
| Gender | |||
| Male | 24 | 12 | 0.5655 |
| Female | 11 | 8 | 0.5655 |
| High grade tumor | |||
| Muscle invasive | 17 | 9 | 0.1313 |
| Non-muscle invasive | 4 | 1 | 0.1313 |
| Low grade tumor | |||
| Muscle invasive | 1 | 2 | 0.1937 |
| Non-muscle invasive | 16 | 5 | 0.1937 |
Expression of STX1A and VAMP2
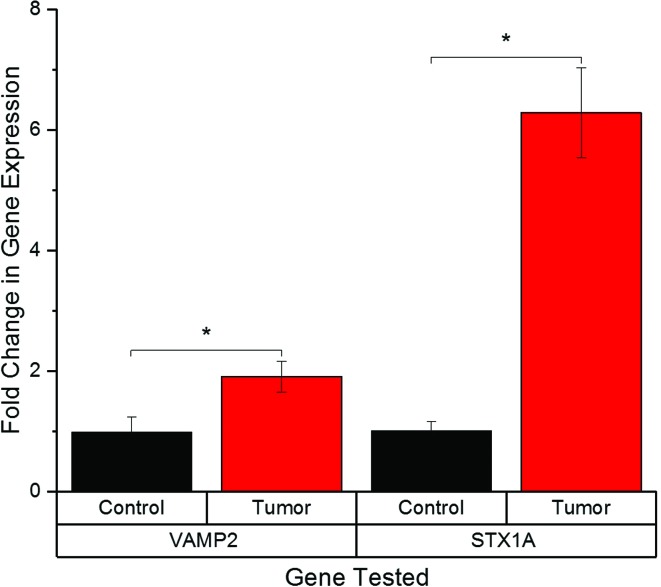
The relative fold-change in the expression of the STX1A was analyzed using the 2-DDCT method (Livak and Schmittgen, 2001). The data were normalized with the internal control gene tubulin (TUB3), and the average fold-change was determined by calculating the relative expressions of each tumor sample (Table S1 (70.2KB, pdf) ). The relative RNA level of STX1A showed a five-fold increase in tumors compared to their controls (p < 0.005). Similarly, the expression of VAMP2 was 2.9-fold higher in tumor samples compared to their controls (p < 0.001) (Figure 2).
Figure 2. Synaptobrevin2 and Syntaxin1A expression in tumor and adjacent normal bladder tissues. Bar graph of normalized (mean ± SE) gene expression of Synaptobrevin2 and Syntaxin1A, showing a significant increase in tumor tissue compared to adjacent normal tissue.
Expression of STX1A and VAMP2 relative to tumor grades
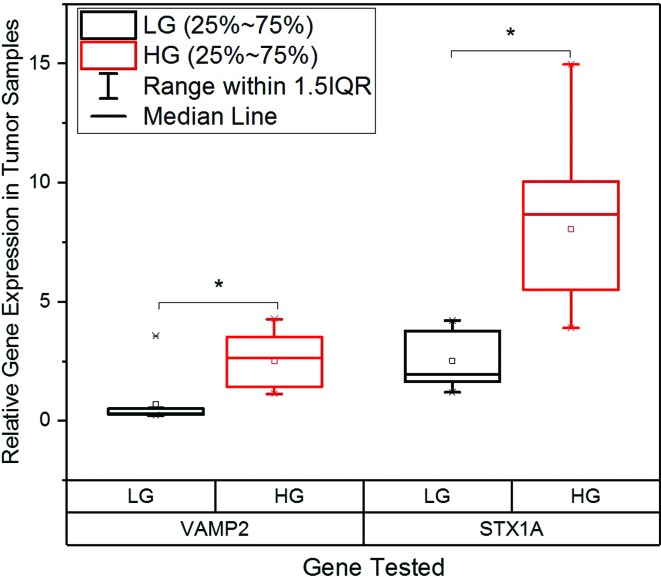
Expression levels of the genes were correlated to tumor grades (Table S2 (73.9KB, pdf) ). According to the pathological grading of bladder cancer, the expression of STX1A was highly increased in the high grade invasive tumors with distant metastasis. The expression of STX1A was eight-fold higher in high grade tumors and 2.5-fold higher in low grade tumors. Therefore, a significant difference of STX1A expression was observed between the high grade and low grade tumors (p < 0.001). VAMP2 expression was also significantly increased in high grade tumors compared to the low grade tumors (p < 0.001). Low grade tumors had a lesser fold-increase in the expression of STX1A and VAMP2, whereas high grade tumors showed relatively higher fold-increases in the expression of both genes (Figure 3). These results suggest that the genes had higher expression in higher grades tumors. The expression of both genes in the controls was normal, indicating that there wass no genetic aberration that might have caused the tumors to progress to high grade.
Figure 3. Expression of Synaptobrevin2 and Syntaxin1A in low and high grade tumor tissues. Boxplots of normalized (relative) gene expression of Synaptobrevin2 and Syntaxin1A showing significantly higher expression in high grade tumors compared to low grade tumors.
Expression of STX1A and VAMP2 relative to tumor stages
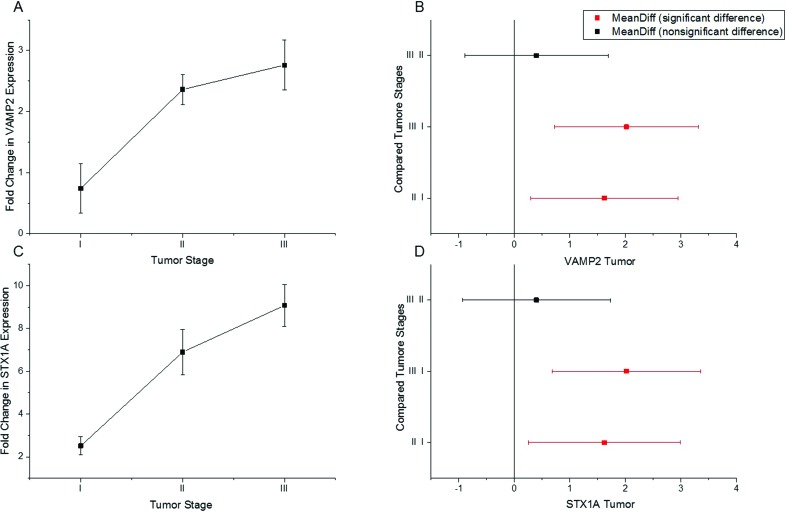
The increased expression of both genes was positively correlated to tumor stage (Table S3 (91.8KB, pdf) ). Our results showed that the expression of STX1A and VAMP2 increased progressively according to the stage of the tumor (Figure 4A, C). Stage II tumors are invasive and show invasion in the bladder muscles, while stage III tumors are highly invasive and tend to spread in adjacent organs. In another study, no change in expression levels was found for both genes between stage II and III (Lopez-Beltran 2008). In our study we observed a significant difference in the expression levels of the two genes between stage I and III, suggesting that the expression of STX1A and VAMP2 increases in a tumor in a stage-dependent manner (Figure 4B and 4D).
Figure 4 . Tumor stage-dependent gene expression. (A and C) Shown is the increase in expression of Synaptobrein2 and Syntaxin1A in a tumor stage-dependent manner (lowest in I and highest in III). (B) A significant difference in the expression of Synaptobrevin2 is seen between stages I and II, and stages I and III, whereas no difference is observed between stage II and III. (D) A significant difference is seen in the expression of Syntaxin1A between stages I and II, and stages I and III, whereas no difference is observed between stages II and III.
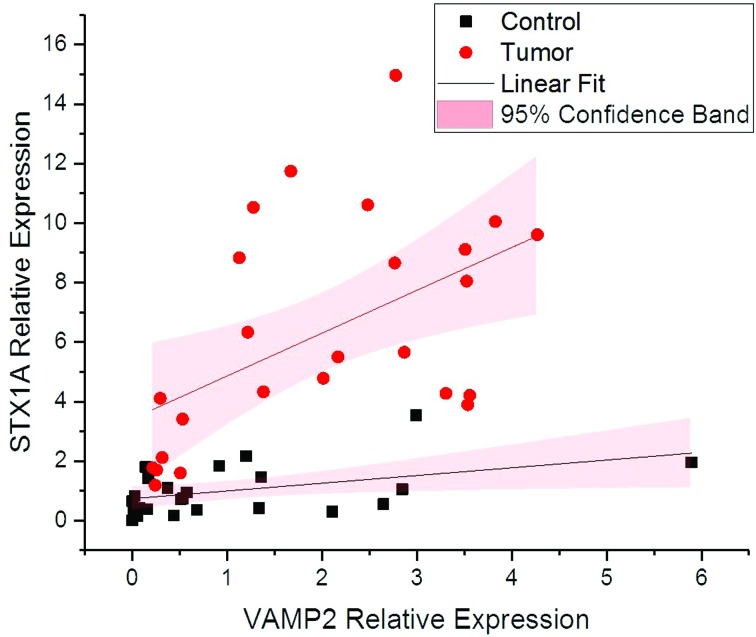
Expression correlation between STX1A and VAMP2
Both genes are a crucial part of SNAREs, as vesicle fusion only takes place followed by their interaction forming a vesicle fusion complex. It was previously reported that cancer progression might have a role in the increased expression of the two genes that mediate cell communication through vesicle fusion (Meng and Wang, 2015). These results suggest that the enhanced expression of STX1A and VAMP2 might have role in triggering tumor progression in high grade stage III tumors. In our correlation analysis results, the increase in the expression of both genes was linear (Table S4 (71.9KB, pdf) ) according to tumor grade and stage, which determines the strongly positive linear correlation between the two genes (Figure 5)
Figure 5. Correlation analysis between expression levels of the Synaptobrevin2 and Syntaxin1A in tumor and adjacent normal bladder tissue. The graph shows a significant positive correlation between expression of Synaptobrevin2 and Syntaxin1A in both tumor and adjacent normal bladder tissues.
Discussion
Epithelial cells along the inner surface of organs form a primary barrier where absorption, secretion, fusion of extracellular vesicles, and exocytosis of exosomes takes place. These cells have a regulatory vesicular communication that is accomplished by a complex of SNARE proteins. SNARE proteins are responsible for maintaining the permeability of the bladder epithelium (Born et al., 2003). The inner luminal membrane exposed to urine releases small vesicles, detaching the membrane that has been subjected to prolonged exposure to urine. Continuous extruding of the apical membrane is regulated by endocytosis of the vesicles (Hurst et al., 2015).
Vesicle trafficking is regulated by SNARE proteins. The SNARE proteins VAMPs (VAMP2) and SXT1A were found to be present in the epithelial fraction of the bladder (Born et al., 2003). STX1A, being t-SNARE, accomplishes vesicle fusion while VAMP2, a v-SNARE, plays a key role in Ca+2-dependent-exocytosis of the vesicles (Chang and Jackson, 2015). STX1A has been reported as tumor enhancer in brain cancers and small cell lung cancers, where its expression plays an important role in tumor formation. Increased expression of STX1A in neuronal cells was reported to be responsible for tumor formation in primary brain tumors (Ulloa et al., 2015). However, in some other cancers, like breast cancer, the expression of STX1A has been shown to be variable (Fernández-Nogueira et al., 2015). Our results showed the increased expression of both genes in bladder tumors compared to their normal adjacent tissue.
SNARE proteins are not only involved in the transport of neurotransmitters and neuropeptides, but also in the transfer of growth factors, recycled receptors, or integrins, and are involved in the secretion of matrix proteases that give the cell the capacity for invasion and migration. Thus, they are involved in cell progression in a regulative manner (Enrich et al., 2015). Apart from cell progression, STX1A and VAMP2 have been reported to be regulatory proteins of the SNARE complex, involved in cell navigation and migration and, hence, metastasis of cancer cells (Zylbersztejn and Galli, 2011; Friedl and Alexander, 2012). The Cancer Genome Atlas (TCGA) dataset of 406 bladder tumor samples revealed an average FPKM value of 3.3 for STX1A expression and 14.9 for VAMP2 expression (https://cancergenome.nih.gov). The Human Protein Atlas (HPA) and Genotype-Tissue Expression Dataset (GTEx) demonstrate a similar trend of expression for both genes in bladder cancer. Our data suggests that the expression of STX1A and VAMP2 was higher in high grade tumors exhibiting aggressive behavior. Overexpression of both genes in tumor cells suggests enhanced vesicular exocytosis that might have caused increased recycling of integrins and excretion of matrix proteases, resulting in a favorable tumor microenvironment for cancer cell development and metastasis.
Conclusions
As an important part of the core SNARE complex, STX1A and VAMP2 are associated with vesicular trafficking of growth factors, integrins, and proteases. Dysregulation of vesicular trafficking might cause multiple malignancies, more importantly cancer cell formation, altered cell adhesion, and alteration of the extracellular matrix, favoring tumor growth (Rainero et al., 2013). Vesicular trafficking is supported by F-actin., and STX1A and VAMP2 were shown to interact with F-actin for SNARE-dependent exocytosis (Daniel et al., 2002; Nevins and Thurmond, 2005). Enhanced expression of STX1A and VAMP2, as shown here in bladder cancer and in previous studies in other cancers (Grabowski et al., 2002; Steffen et al., 2008; Meng and Wang, 2015) suggest their involvement in abnormal vesicular trafficking that might have a critical role in tumor formation and metastasis.
Acknowledgments
We are thankful to the great assistance of doctors at the Department of Urology, Pakistan Institute of Medical Sciences (PIMS), and SHIFA International Hospital (Pakistan). This research was supported by a grant (CRGP) funded by COMSATS Institute of information Technology (Pakistan).
Supplementary material
The following online material is available for this article:
Footnotes
Associate Editor: Emmanuel Dias Neto
Conflict of Interest
The authors declare that there is no conflict of interest that could be perceived as prejudicial to the impartiality of the reported research.
Authors Contributions
SAR, SA, MJK and AH conceived the idea and designed the project. SAR, SA and MJK helped in experimentation and data acquisition. AY, AK, MN, AM contributed to clinical evaluation and sample provision. SAR, SA, STAS, AT, NB contributed to data analysis and the interpretation of the results. AH took the lead in writing the manuscript along SAR, SA, STAS, AT and NB. AH and MJK supervised the research. All authors reviewed and approved the manuscript.
References
- American Cancer Society . Cancer Facts & Figures. American Cancer Society; Atlanta: 2017. p. 71. [Google Scholar]; American Cancer Society (2017) Cancer Facts & Figures, American Cancer Society, Atlanta, 71 p.
- Arsenault J, Ferrari E, Niranjan D, Cuijpers SAG, Gu C, Vallis Y, O’Brien J, Davletov B. Stapling of the Botulinum Type A Protease to growth factors and neuropeptides allows selective targeting of neuroendocrine cells. J Neurochem. 2013;126:223–233. doi: 10.1111/jnc.12284. [DOI] [PMC free article] [PubMed] [Google Scholar]; Arsenault J, Ferrari E, Niranjan D, Cuijpers SAG, Gu C, Vallis Y, O’Brien J and Davletov B (2013) Stapling of the Botulinum Type A Protease to growth factors and neuropeptides allows selective targeting of neuroendocrine cells. J Neurochem 126:223–233. [DOI] [PMC free article] [PubMed]
- Bartsch G, Mitra AP, Cote RJ. Expression profiling for bladder cancer: Strategies to uncover prognostic factors. Expert Rev Anticancer Ther. 2010;10:1945–1954. doi: 10.1586/era.10.131. [DOI] [PMC free article] [PubMed] [Google Scholar]; Bartsch G, Mitra AP and Cote RJ (2010) Expression profiling for bladder cancer: Strategies to uncover prognostic factors. Expert Rev Anticancer Ther 10:1945–1954. [DOI] [PMC free article] [PubMed]
- Born M, Pahner I, Ahnert-Hilger G, Jöns T. The maintenance of the permeability barrier of bladder facet cells requires a continuous fusion of discoid vesicles with the apical plasma membrane. Eur J Cell Biol. 2003;82:343–350. doi: 10.1078/0171-9335-00326. [DOI] [PubMed] [Google Scholar]; Born M, Pahner I, Ahnert-Hilger G and Jöns T (2003) The maintenance of the permeability barrier of bladder facet cells requires a continuous fusion of discoid vesicles with the apical plasma membrane. Eur J Cell Biol 82:343–350. [DOI] [PubMed]
- Chang CW, Jackson MB. Synaptobrevin transmembrane domain influences exocytosis by perturbing vesicle membrane curvature. Biophys J. 2015;109:76–84. doi: 10.1016/j.bpj.2015.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]; Chang CW and Jackson MB (2015) Synaptobrevin transmembrane domain influences exocytosis by perturbing vesicle membrane curvature. Biophys J 109:76–84. [DOI] [PMC free article] [PubMed]
- Daniel S, Noda M, Cerione RA, Sharp GW. A link between Cdc42 and syntaxin is involved in Mastoparan-stimulated insulin release. Biochemistry. 2002;41:9663–9671. doi: 10.1021/bi025604p. [DOI] [PubMed] [Google Scholar]; Daniel S, Noda M, Cerione RA and Sharp GW (2002) A link between Cdc42 and syntaxin is involved in Mastoparan-stimulated insulin release. Biochemistry 41:9663-9671. [DOI] [PubMed]
- Enrich C, Rentero C, Hierro A, Grewal T. Role of cholesterol in SNARE-mediated trafficking on intracellular membranes. J Cell Sci. 2015;128:1071–1081. doi: 10.1242/jcs.164459. [DOI] [PubMed] [Google Scholar]; Enrich C, Rentero C, Hierro A and Grewal T (2015). Role of cholesterol in SNARE-mediated trafficking on intracellular membranes. J Cell Sci 128:1071–1081. [DOI] [PubMed]
- Fang Q, Lindau M. How could SNARE proteins open a fusion pore? Physiology. 2014;29:278–285. doi: 10.1152/physiol.00026.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]; Fang Q and Lindau M (2014) How could SNARE proteins open a fusion pore? Physiology 29:278–285. [DOI] [PMC free article] [PubMed]
- Fernández-Nogueira P, Bragado P, Almendro V, Ametller E, Rios J, Choudhury S, Mancino M, Gascón P. Differential expression of neurogenes among breast cancer subtypes identifies high risk patients. Oncotarget. 2015;7:5313–5326. doi: 10.18632/oncotarget.6543. [DOI] [PMC free article] [PubMed] [Google Scholar]; Fernández-Nogueira P, Bragado P, Almendro V, Ametller E, Rios J, Choudhury S, Mancino M and Gascón P (2015) Differential expression of neurogenes among breast cancer subtypes identifies high risk patients. Oncotarget 7:5313-5326. [DOI] [PMC free article] [PubMed]
- Friedl P, Alexander S. Cancer invasion and the microenvironment: Plasticity and reciprocity. Cell. 2012;147:992–1009. doi: 10.1016/j.cell.2011.11.016. [DOI] [PubMed] [Google Scholar]; Friedl P and Alexander S (2012) Cancer invasion and the microenvironment: Plasticity and reciprocity. Cell 147: 992–1009. [DOI] [PubMed]
- Grabowski P, Schönfelder S, Ahnert-Hilger G, Foss HD, Heine B, Schindler I, Stein H, Berger G, Zeitz M, Scherübl H. Expression of neuroendocrine markers: A signature of human undifferentiated carcinoma of the colon and rectum. Virchows Archiv. 2002;441:256–263. doi: 10.1007/s00428-002-0650-9. [DOI] [PubMed] [Google Scholar]; Grabowski P, Schönfelder S, Ahnert-Hilger G, Foss HD, Heine B, Schindler I, Stein H, Berger G, Zeitz M and Scherübl H (2002) Expression of neuroendocrine markers: A signature of human undifferentiated carcinoma of the colon and rectum. Virchows Archiv 441:256–263. [DOI] [PubMed]
- Haberman A, Williamson WR, Epstein D, Wang D, Rina S, Meinertzhagen IA, Hiesinger PR. The synaptic vesicle SNARE neuronal synaptobrevin promotes endolysosomal degradation and prevents neurodegeneration. J Cell Biol. 2012;196:261–276. doi: 10.1083/jcb.201108088. [DOI] [PMC free article] [PubMed] [Google Scholar]; Haberman A, Williamson WR, Epstein D, Wang D, Rina S, Meinertzhagen IA and Hiesinger PR (2012) The synaptic vesicle SNARE neuronal synaptobrevin promotes endolysosomal degradation and prevents neurodegeneration. J Cell Biol 196:261–276. [DOI] [PMC free article] [PubMed]
- Hasan N, Hu C. Vesicle-associated membrane protein 2 mediates trafficking of α5β1 integrin to the plasma membrane. Exp Cell Res. 2010;316:12–23. doi: 10.1016/j.yexcr.2009.10.007. [DOI] [PubMed] [Google Scholar]; Hasan N and Hu C (2010) Vesicle-associated membrane protein 2 mediates trafficking of α5β1 integrin to the plasma membrane. Exp Cell Res 316:12-23. [DOI] [PubMed]
- Hurst RE, Van Meerveld B, Wisniewski AB, VanGordon S, Lin H, Kropp BP, Towner RA. Increased bladder permeability in interstitial cystitis/painful bladder syndrome. Translat Androl Urol. 2015;45:563–571. doi: 10.3978/j.issn.2223-4683.2015.10.03. [DOI] [PMC free article] [PubMed] [Google Scholar]; Hurst RE, Van Meerveld B, Wisniewski AB, VanGordon S, Lin H, Kropp BP, and Towner RA (2015) Increased bladder permeability in interstitial cystitis/painful bladder syndrome. Translat Androl Urol 45:563–571. [DOI] [PMC free article] [PubMed]
- Kang HW, Kim WJ, Yun SJ. The role of the tumor microenvironment in bladder cancer development and progression. Translat Cancer Res. 2017;6(Suppl 4):S744–S758. [Google Scholar]; Kang HW, Kim WJ and Yun SJ (2017) The role of the tumor microenvironment in bladder cancer development and progression. Translat Cancer Res 6 Suppl 4: S744-S758.
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-DDCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]; Livak KJ and Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-DDCT method. Methods 25:402-408. [DOI] [PubMed]
- Lopez-Beltran A. Bladder cancer: Clinical and pathological profile. Scand J Urol Nephrol. 2008;42:95–109. doi: 10.1080/03008880802325226. [DOI] [PubMed] [Google Scholar]; Lopez-Beltran A (2008) Bladder cancer: Clinical and pathological profile. Scand J Urol Nephrol 42:95–109. [DOI] [PubMed]
- Meng J, Wang J. Role of SNARE proteins in tumourigenesis and their potential as targets for novel anti-cancer therapeutics. Biochim Biophys Acta. 2015;1856:1–12. doi: 10.1016/j.bbcan.2015.04.002. [DOI] [PubMed] [Google Scholar]; Meng J and Wang J (2015) Role of SNARE proteins in tumourigenesis and their potential as targets for novel anti-cancer therapeutics. Biochim Biophys Acta 1856:1–12. [DOI] [PubMed]
- Miyamoto H, Miller JS, Fajardo DA, Lee TK, Netto GJ, Epstein JI. Non-invasive papillary urothelial neoplasms: The 2004 WHO/ISUP classification system. Pathol Int. 2010;60:1–8. doi: 10.1111/j.1440-1827.2009.02477.x. [DOI] [PubMed] [Google Scholar]; Miyamoto H, Miller JS, Fajardo DA, Lee TK, Netto GJ and Epstein JI (2010) Non-invasive papillary urothelial neoplasms: The 2004 WHO/ISUP classification system. Pathol Int 60:1–8. [DOI] [PubMed]
- Nevins AK, Thurmond DC. A direct interaction between Cdc42 and vesicle-associated membrane protein 2 regulates SNARE-dependent insulin exocytosis. J Biol Chem. 2005;280:1944–1952. doi: 10.1074/jbc.M409528200. [DOI] [PubMed] [Google Scholar]; Nevins AK and Thurmond DC (2005) A direct interaction between Cdc42 and vesicle-associated membrane protein 2 regulates SNARE-dependent insulin exocytosis. J Biol Chem 280:1944-1952. [DOI] [PubMed]
- Palfreyman MT, Jorgensen EM. Roles of SNARE proteins in synaptic vesicle fusion. In: Wang ZW, editor. Molecular Mechanisms of Neurotransmitter Release. Humana Press; Totowa, NJ: 2008. pp. 35–59. [Google Scholar]; Palfreyman MT and Jorgensen EM (2008) Roles of SNARE proteins in synaptic vesicle fusion. In: Wang ZW (ed) Molecular Mechanisms of Neurotransmitter Release. Humana Press, Totowa, NJ, pp 35–59.
- Pan CC, Chang YH, Chen KK, Yu HJ, Sun CH, Ho DNT. Prognostic significance of the 2004 WHO/ISUP classification for prediction of recurrence, progression, and cancer-specific mortality of non-muscle-invasive urothelial tumors of the urinary bladder: A clinicopathologic study of 1,515 cases. Am J Clin Pathol. 2010;133:788–795. doi: 10.1309/AJCP12MRVVHTCKEJ. [DOI] [PubMed] [Google Scholar]; Pan CC, Chang YH, Chen KK, Yu HJ, Sun CH and Ho DNT (2010) Prognostic significance of the 2004 WHO/ISUP classification for prediction of recurrence, progression, and cancer-specific mortality of non-muscle-invasive urothelial tumors of the urinary bladder: A clinicopathologic study of 1,515 cases. Am J Clin Pathol 133:788–795. [DOI] [PubMed]
- Polchi A, Tancini B, Emiliani C. Signaling pathways in exosome biogenesis, secretion and fate. Genes. 2013;4:152–170. doi: 10.3390/genes4020152. [DOI] [PMC free article] [PubMed] [Google Scholar]; Polchi A, Tancini B and Emiliani C (2013) Signaling pathways in exosome biogenesis, secretion and fate. Genes 4:152–170. [DOI] [PMC free article] [PubMed]
- Rainero E, Van den Berghe P, Norman JC. Internalisation, endosomal trafficking and recycling of integrins during cell migration and cancer Invasion. In: Yarden Y, Tarcic G, editors. Vesicle Trafficking in Cancer. Springer; New York: 2013. pp. 327–359. [Google Scholar]; Rainero E, Van den Berghe P and Norman JC (2013) Internalisation, endosomal trafficking and recycling of integrins during cell migration and cancer Invasion. In: Yarden Y and Tarcic G (eds) Vesicle Trafficking in Cancer. Springer, New York, pp 327-359.
- Ramakrishnan NA, Drescher MJ, Drescher DG. The SNARE complex in neuronal and sensory cells. Mol Cell Neurosci. 2012;50:58–69. doi: 10.1016/j.mcn.2012.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]; Ramakrishnan NA, Drescher MJ and Drescher DG (2012) The SNARE complex in neuronal and sensory cells. Mol Cell Neurosci 50:58-69. [DOI] [PMC free article] [PubMed]
- Rio DC, Ares M, Hannon GJ, Nilsen TW. Purification of RNA using TRIzol (TRI Reagent) Cold Spring Harb Protoc. 2010;5:5440–5441. doi: 10.1101/pdb.prot5439. [DOI] [PubMed] [Google Scholar]; Rio DC, Ares M, Hannon GJ and Nilsen TW (2010) Purification of RNA using TRIzol (TRI Reagent). Cold Spring Harb Protoc 5: 5440–5441. [DOI] [PubMed]
- Silvers CR, Miyamoto H, Messing EM, Netto GJ, Lee YF. Characterization of urinary extracellular vesicle proteins in muscle-invasive bladder cancer. Oncotarget. 2017;8:91199–91208. doi: 10.18632/oncotarget.20043. [DOI] [PMC free article] [PubMed] [Google Scholar]; Silvers CR, Miyamoto H, Messing EM, Netto GJ and Lee YF (2017) Characterization of urinary extracellular vesicle proteins in muscle-invasive bladder cancer. Oncotarget 8:91199-91208. [DOI] [PMC free article] [PubMed]
- Shukla AL, Berglund L, Nielsen LP, Nielsen S, Hoffmann HJ, Dahl R. Regulated exocytosis in immune function: Are SNARE-proteins involved? Respir Med. 2000;94:10–17. doi: 10.1053/rmed.1999.0700. [DOI] [PubMed] [Google Scholar]; Shukla AL, Berglund L, Nielsen LP, Nielsen S, Hoffmann HJ and Dahl R (2000). Regulated exocytosis in immune function: Are SNARE-proteins involved? Respir Med 94:10–17. [DOI] [PubMed]
- Steffen A, Le Dez G, Poincloux R, Recchi C, Nassoy P, Rottner K, Galli T, Chavrier P. MT1-MMP-dependent invasion is regulated by TI-VAMP/VAMP7. Curr Biol. 2008;18:926–931. doi: 10.1016/j.cub.2008.05.044. [DOI] [PubMed] [Google Scholar]; Steffen A, Le Dez G, Poincloux R, Recchi C, Nassoy P, Rottner K, Galli T and Chavrier P (2008) MT1-MMP-dependent invasion is regulated by TI-VAMP/VAMP7. Curr Biol 18:926–931. [DOI] [PubMed]
- Tian AG, Tamori Y, Huang YC, Melendez NT, Deng WM. Efficient EGFR signaling and dorsal-ventral axis patterning requires syntaxin dependent Gurken trafficking. Dev Biol. 2014;373:349–358. doi: 10.1016/j.ydbio.2012.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]; Tian AG, Tamori Y, Huang YC, Melendez NT and Deng WM (2014) Efficient EGFR signaling and dorsal-ventral axis patterning requires syntaxin dependent Gurken trafficking. Dev Biol 373:349–358. [DOI] [PMC free article] [PubMed]
- Ulloa F, Gonzàlez-Juncà A, Meffre D, Barrecheguren PJ, Martínez-Mármol R, Pazos I, Olivé N, Cotrufo T, Seoane J, Soriano E. Blockade of the SNARE protein Syntaxin 1 inhibits glioblastoma tumor growth. PLoS One. 2015;10:e0119707. doi: 10.1371/journal.pone.0119707. [DOI] [PMC free article] [PubMed] [Google Scholar]; Ulloa F, Gonzàlez-Juncà A, Meffre D, Barrecheguren PJ, Martínez-Mármol R, Pazos I, Olivé N, Cotrufo T, Seoane J and Soriano E (2015) Blockade of the SNARE protein Syntaxin 1 inhibits glioblastoma tumor growth. PLoS One 10:e0119707. [DOI] [PMC free article] [PubMed]
- Williams KC, Coppolino MG. SNARE-dependent interaction of Src, EGFR and 1 Integrin regulates invadopodia formation and tumor cell invasion. J Cell Sci. 2014;127:1712–1725. doi: 10.1242/jcs.134734. [DOI] [PubMed] [Google Scholar]; Williams KC and Coppolino MG (2014) SNARE-dependent interaction of Src, EGFR and 1 Integrin regulates invadopodia formation and tumor cell invasion. J Cell Sci 127:1712–1725. [DOI] [PubMed]
- Williams KC, McNeilly RE, Coppolino MG. SNAP23, syntaxin, 4 and vesicle-associated membrane protein 7 (VAMP7) mediate trafficking of membrane type 1–matrix metalloproteinase (MT1-MMP) during invadopodium formation and tumor cell invasion. Mol Biol Cell. 2014;25:2061–2070. doi: 10.1091/mbc.E13-10-0582. [DOI] [PMC free article] [PubMed] [Google Scholar]; Williams KC, McNeilly RE and Coppolino MG (2014) SNAP23, syntaxin 4, and vesicle-associated membrane protein 7 (VAMP7) mediate trafficking of membrane type 1–matrix metalloproteinase (MT1-MMP) during invadopodium formation and tumor cell invasion. Mol Biol Cell 25:2061-2070. [DOI] [PMC free article] [PubMed]
- Zylbersztejn K, Galli T. Vesicular traffic in cell navigation. FEBS J. 2011;278:4497–4505. doi: 10.1111/j.1742-4658.2011.08168.x. [DOI] [PubMed] [Google Scholar]; Zylbersztejn K Galli T (2011) Vesicular traffic in cell navigation. FEBS J 278:4497–4505. [DOI] [PubMed]
Internet resources
- The Human Protien Atlas (HPA) [(accessed 12 March 2018).]. https://www.proteinatlas.org/ENSG00000220205-VAMP2/pathology/tissue/urothelial+cancer.; The Human Protien Atlas (HPA), https://www.proteinatlas.org/ENSG00000220205-VAMP2/pathology/tissue/urothelial+cancer (accessed 12 March 2018).
- The Human Protien Atlas (HPA) [(accessed 12 March 2018).]. https://www.proteinatlas.org/ENSG00000106089-STX1A/pathology/tissue/urothelial+cancer.; The Human Protien Atlas (HPA), https://www.proteinatlas.org/ENSG00000106089-STX1A/pathology/tissue/urothelial+cancer (accessed 12 March 2018).
- The Cancer Genome Atlas (TCGA) [(accessed 12 March 2018).]. https://cancergenome.nih.gov/cancersselected/UrothelialBladderCarcinoma.; The Cancer Genome Atlas (TCGA), https://cancergenome.nih.gov/cancersselected/UrothelialBladderCarcinoma (accessed 12 March 2018).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.