Abstract
Uridine diphosphate glycosyltransferases (UGTs) are multifunctional detoxification enzymes, which are involved in metabolizing various chemicals and contribute to the development of insecticide resistance. However, the possible roles of UGTs in chlorantraniliprole resistance in Chilo suppressalis have rarely been studied in detail. Based on genome data, 24 UGT genes in C. suppressalis belonging to 11 families were identified, which were designated by the UGT nomenclature committee. Synergism assay data suggested that UGTs are potentially involved in chlorantraniliprole resistance in C. suppressalis. CsUGT40AL1 and CsUGT33AG3 were significantly overexpressed in the chlorantraniliprole resistant strain (12.36- and 5.34-fold, respectively). The two UGTs were highly expressed in the larval Malpighian tubules, fat body, and midgut; however, expression was lowest in the head. Injection of individual dsRNAs reduced the expression of the two target genes (by 69.34% and 48.74%, respectively) and caused significant higher larval mortality (81.33% and 54.67%, respectively). Overexpression of CsUGT40AL1 and CsUGT33AG3 was potentially involved in chlorantraniliprole resistance in C. suppressalis, as confirmed by the RNAi assay. Our findings suggest that overexpression of UGTs may contribute to chlorantraniliprole resistance in C. suppressalis.
Keywords: Chilo suppressalis, UDP-glycosyltransferase, chlorantraniliprole, resistance, RNAi
1. Introduction
The striped rice stem borer, Chilo suppressalis (Walker), is an serious pest of agricultural grain crops, including rice, and it causes huge losses in yield throughout Asia [1,2]. Although it is known for its rapid development of resistance to many conventional insecticides, the management of C. suppressalis relies heavily on insecticides, which emphasizes the necessity for the understanding of its insecticide resistance mechanisms [3,4]. Chlorantraniliprole, as a novel diamide insecticide that targets the ryanodine receptor (RyR), has been extensively used for the control of caterpillars [5]. However, high-level chlorantraniliprole resistance of C. suppressalis has been reported after a few years of extensive use [6,7]. Understanding the chlorantraniliprole resistance mechanism can improve its application and prolong its use. Previous studies have found that three mutations (G4910E, Y4667D, and I4758M) in RyR might be involved in the chlorantraniliprole resistance in C. suppressalis [7,8]. Additionally, multiple overexpressed cytochrome P450-dependent monooxygenase genes (CYP6CV5, CYP9A68, CYP321F3, and CYP32412) potentially contributed to chlorantraniliprole resistance of C. suppressalis [9]. Enhancement of esterase activity might also confer C. suppressalis resistance to chlorantraniliprole [7,8]. These results showed complex resistance mechanisms in C. suppressalis to chlorantraniliprole. Therefore, the resistance mechanism of chlorantraniliprole in C. suppressalis should be clearly understood.
The insect detoxification systems that mediate diverse metabolic resistance mechanisms to insecticides are sophisticated, and different detoxification enzyme genes may be involved in insecticide resistance. In the three phases of detoxification, uridine diphosphate glycosyltransferases (UGTs) belong to phase II metabolizing enzymes that are found in all living organisms, which are encoded by a superfamily of genes [10]. The protein structure of UGTs contains two main parts: the aglycone substrate-binding domain at the N-terminus, and the UDP sugar donor-binding domain at the C-terminus. UGTs catalyze the transfer of glycosyl groups from a nucleotide sugar to a variety of small hydrophobic molecules (aglycones). This increases their water solubility and makes them easier to be excreted, resulting in the detoxification and elimination of substrates [11]. Therefore, glycosylation of toxins by UGTs is a particularly important detoxification mechanism. UGTs are important detoxification enzymes in insects which can metabolize insecticides, and consequently the involvement of insect UGTs in the development of insecticide resistance has been studied. Bull et al. (1972) found that UGTs could be involved in the metabolism of insecticides by enhancing glucosidic conjugation in the organophosphorus resistant Heliothis virescens [12]. Similarly, UGTs are important in pyraclofos metabolism in the resistant Musca domestica strain [13]. An important feature of detoxification enzyme–mediated metabolic resistance is the overexpression of detoxification enzyme genes at the transcriptional level in insecticide-resistant insects, which results in increased protein amounts and enzyme activity levels. Previous studies have suggested that gene overexpression is the cause of insecticide resistance. Overexpression of UGT2 contributes to imidacloprid resistance in the resistant strain of Leptinotarsa decemlineata [14]. The overexpression of multiple UGT genes was found to play an important role in the resistance of Aphis gossypii to thiamethoxam insecticides [15]. Overexpressed UGT2B17 in Plutella xylostella conferred high-level chlorantraniliprole resistance [16]. However, whether the chlorantraniliprole resistance in C. suppressalis is mediated by UGT genes needs to be investigated.
The study of the resistance mechanism in C. suppressalis will enhance the continued development of innovative alternative control measures and resistance management strategies. In the present study, UGT genes were identified in C. suppressalis based on genome data, and the phylogenetic relationships among these genes and their homologs in four other insects were analyzed. We performed synergism bioassays and screened overexpressed UGT genes using chlorantraniliprole-susceptible and chlorantraniliprole-resistant strains. Furthermore, we characterized tissue-specific expression profiles of overexpressed UGT genes. We also confirmed the function of UGTs in chlorantraniliprole resistance using RNA interference (RNAi). We aimed to elucidate the contribution of overexpressed UGTs to the chlorantraniliprole resistance mechanism in C. suppressalis.
2. Results
2.1. Chlorantraniliprole Toxicity and Synergists Assessment
The resistant strain (YYR) had developed a high resistance level to chlorantraniliprole (LC50 = 565.48 mg/L) with a resistance ratio (RR) of 44.32, while the susceptible strain (YYS) was sensitive to chlorantraniliprole. To investigate whether the UGTs were involved in the detoxification of chlorantraniliprole in the two strains of C. suppressalis, two chemicals, 5-nitrouracil (5-NU) and sulfinpyrazone (SUL), were selected as synergists for their good inhibition of UGT activity. In YYR, 5-NU increased the chlorantraniliprole toxicity by 3.38-fold, and SUL increased it by 3.56-fold. However, the toxicity of chlorantraniliprole was not significantly changed by 5-NU or SUL in YYS, and the synergistic ratios (SRs) were 1.22- and 1.13-fold, respectively (Table 1).
Table 1.
Synergism effects of 5-NU and SUL on the toxicities of chlorantraniliprole in the YYS and YYR strains.
| Strain | Treatment | LC50 (95% CL) (mg·L−1) | Slope ± SE | x 2 | RR | SR |
|---|---|---|---|---|---|---|
| YYS | Chlorantraniliprole | 12.76 (11.08–14.76) | 2.72 ± 0.31 | 5.96 | 1.00 | 1.00 |
| Chlorantraniliprole + 5-NU | 10.48 (8.95–12.27) | 2.42 ± 0.35 | 5.17 | - | 1.22 | |
| Chlorantraniliprole + SUL | 11.27 (9.76–13.03) | 2.70 ± 0.32 | 7.72 | - | 1.13 | |
| YYR | Chlorantraniliprole | 565.48 (510.42–626.47) | 3.67 ± 0.22 | 7.31 | 44.32 | 1.00 |
| Chlorantraniliprole + 5-NU | 167.47 (146.78–191.08) | 2.97 ± 0.29 | 2.68 | - | 3.38 * | |
| Chlorantraniliprole + SUL | 158.71 (139.32–180.79) | 3.08 ± 0.28 | 7.75 | - | 3.56 * |
* Indicates statistical significance. CL, Confidence limits; RR = LC50 of YYR strain/LC50 of YYS strain; SR = LC50 of chlorantraniliprole/LC50 of chlorantraniliprole + synergist.
2.2. Identification and Classification of UGT Genes
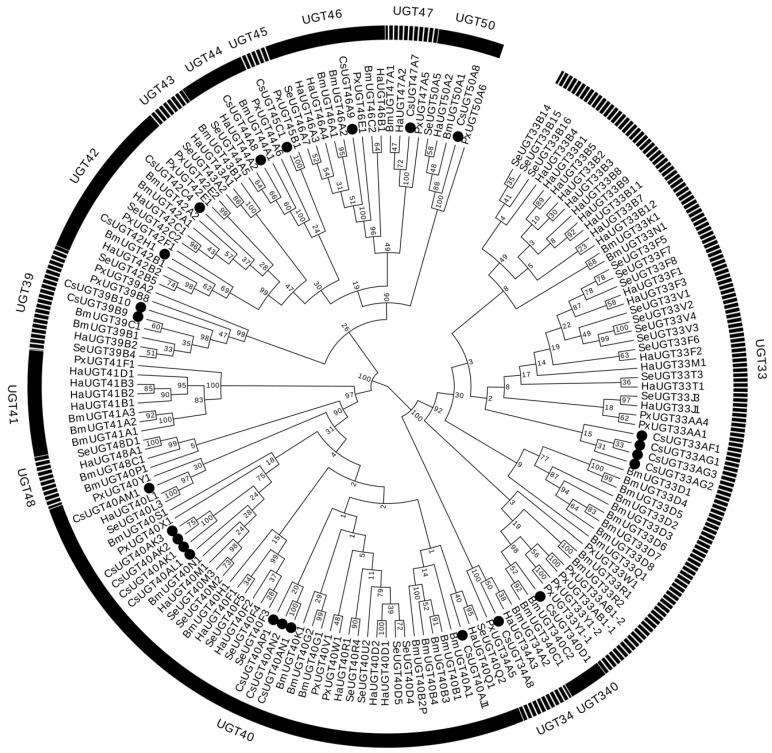
Twenty-four UGT genes with complete open reading frames (ORFs) were identified from the genomic database of C. suppressalis, and their cDNAs were cloned using reverse transcription polymerase chain reaction (RT-PCR) confirmation. Amino acid sequences of 24 UGTs were predicted uridine diphosphate glycosyltransferase conserved domains ‘UDPGT’ based on SMART online software (http://smart.embl-heidelberg.de/). The UGT gene names were constituted by a prefix ‘Cs’ and a specific name. The specific names of these UGT genes were designated by the UGT Nomenclature Committee [17]. The characteristics of these 24 UGTs are summarized in Table 2. These gene sequences were deposited into GenBank (GenBank accession nos. MK135471-MK135494). The ORFs of the UGTs ranged from 514 to 544 amino acids in length, and the predicted molecular weights (MWs) of deduced proteins ranged from 57.36 to 62.11 kDa. The isoelectric points (PIs) ranged from 6.77 to 9.32. Fifteen of the 24 UGTs had signal peptide (SP) sequences at the N-terminal end. To understand the evolutionary relationships of C. suppressalis UGT genes, a phylogenetic analysis was performed by combing these data with the UGT orthologs from the other four species. The 24 C. suppressalis UGTs were clustered into 11 families, of which four and nine UGTs were clustered within the UGT33 and UGT40 families, respectively. There were two UGTs in the UGT39 and UGT42 family, respectively, and only one each in the UGT34, UGT44, UGT45, UGT46, UGT47, UGT50, and UGT340 families (Figure 1).
Table 2.
Characteristics of the UGT genes identified in C. suppressalis.
| Gene Name | Accession Number | ORF Size (aa) | PI/MW (KDa) | SP | ‘UDPGT’ Domain | Best Blastx Match | ||
|---|---|---|---|---|---|---|---|---|
| Species Name 1 | GenBank Accession Number | Identity | ||||||
| CsUGT33AF1 | MK135471 | 518 | 7.77/59.32 | 1–19 | 22–517 | Ha | XP_021197637.1 | 62% |
| CsUGT33AG1 | MK135472 | 524 | 6.91/60.06 | 1–22 | 24–517 | Pp | XP_013139422.1 | 61% |
| CsUGT33AG2 | MK135473 | 520 | 6.77/59.27 | 1–20 | 24–517 | Ha | AEW43120.1 | 55% |
| CsUGT33AG3 | MK135474 | 520 | 9.14/59.67 | 1–23 | 25–519 | Ha | AEW43118.1 | 57% |
| CsUGT340D1 | MK135475 | 523 | 7.24/59.54 | 1–19 | 21–519 | Bm | NP_001243963.1 | 52% |
| CsUGT34A8 | MK135476 | 523 | 8.30/59.31 | – | 22–510 | At | XP_013184861.1 | 62% |
| CsUGT39B10 | MK135477 | 528 | 9.00/61.43 | – | 110–517 | Bm | NP_001243980.1 | 69% |
| CsUGT39B9 | MK135478 | 528 | 9.01/60.69 | 1–21 | 95–516 | Se | ANI21999.1 | 66% |
| CsUGT40AJ1 | MK135479 | 514 | 8.87/58.20 | – | 19–513 | Ha | XP_021183292.1 | 56% |
| CsUGT40AK1 | MK135480 | 519 | 8.39/59.76 | – | 29–518 | Ha | AEW43128.1 | 52% |
| CsUGT40AK2 | MK135481 | 519 | 8.83/59.17 | – | 37–515 | Ha | AEW43128.1 | 52% |
| CsUGT40AK3 | MK135482 | 520 | 8.76/59.33 | 1–18 | 27–516 | Ha | XP_021191114.1 | 54% |
| CsUGT40AL1 | MK135483 | 531 | 9.32/61.01 | – | 32–531 | Ha | AEW43128.1 | 54% |
| CsUGT40AM1 | MK135484 | 521 | 8.13/59.30 | 1–20 | 22–520 | Dp | OWR51610.1 | 59% |
| CsUGT40AN1 | MK135485 | 515 | 9.00/57.36 | – | 22–514 | At | XP_013186135.1 | 56% |
| CsUGT40AN2 | MK135486 | 514 | 8.94/57.42 | – | 22–513 | At | XP_013189977.1 | 56% |
| CsUGT40AP1 | MK135487 | 519 | 9.22/58.68 | 1–19 | 24–518 | Bm | XP_012550238.1 | 56% |
| CsUGT42C4 | MK135488 | 505 | 7.63/57.49 | 1–21 | 24–500 | Se | ANI22012.1 | 65% |
| CsUGT42H1 | MK135489 | 524 | 8.67/59.39 | 1–22 | 24–523 | Pm | XP_014358643.1 | 61% |
| CsUGT44A8 | MK135490 | 515 | 6.79/59.79 | – | 30–511 | Ha | XP_021186279.1 | 62% |
| CsUGT45C1 | MK135491 | 515 | 8.33/58.49 | 1–19 | 24–502 | Pp | XP_013141171.1 | 54% |
| CsUGT46A9 | MK135492 | 533 | 8.96/60.29 | 1–23 | 27–528 | At | XP_013183121.1 | 66% |
| CsUGT47A7 | MK135493 | 530 | 8.94/60.44 | 1–18 | 19–510 | At | XP_013192144.1 | 78% |
| CsUGT50A8 | MK135494 | 544 | 8.88/62.11 | 1–21 | 79–516 | Ha | XP_021184423.1 | 74% |
ORF, open reading frame; MW, molecular weight; PI, isoelectric point; SP, signal peptide. 1 Se (Spodoptera exigua), Ha (Helicoverpa armigera), Pp (Papilio polytes), Bm (Bombyx mori), At (Amyelois transitella), Dp (Danaus plexippus), Pm (Papilio machaon).
Figure 1.
Phylogenetic relationship of UGTs from different species using the complete amino acid sequences. Twenty-four UGT amino acid sequences from Chilo suppressalis (Cs before the name), 23 from Plutella xylostella (Px), 32 from Spodoptera exigua (Se), 40 from Helicoverpa armigera (Ha), and 44 from Bombyx mori (Bm). The tree was constructed with MEGA 5.0, using the neighbor-joining method with the Poisson correction model and 1000 bootstrap replicates. The identified UGTs are highlighted with solid black circles.
The amino acid sequence alignments of the 24 UGTs with human UGT2B7 revealed that these UGT proteins contained the two major parts: the highly variable N-terminal substrate-binding part, and the conserved C-terminal sugar donor-binding part. The two catalytic residues deduced from human UGT2B7 modeling (H and D) were located in the N-terminal substrate-binding part. In addition, the two predicted sugar donor-binding regions (DBR1 and DBR2) were found in the middle of the C-terminal part, and three important residues in DBR interacting with the sugar donor were also conserved. The UGT signature motif sequence, (FVA)-(LIVMF)-(TS)-(HQ)-(SGAC)-G-X(2)-(STG)-X(2)-(DE)-X(6)-P-(LIVMFA)-(LIVMFA)-X(2)-P-(LMVFIQ)-X(2)-(DE)-Q (where X is any amino acid), was found in the middle of the C-terminal part [10]. The transmembrane domain, followed by cytoplasmic tails, was identified at the C-terminal end (Figure 2).
Figure 2.
Multiple alignments of 24 UGTs in C. suppressalis with human UGT2B7. The transmembrane domain and cytoplasmic tails based on human UGT2B7 are underlined. Asterisks (*) below alignments indicate the important catalytic residues (H and D). Black and grey background indicate that residues strictly conserved and residues well conserved, respectively. Two donor-binding regions (DBR1 and DBR2) are overlined. The signature motif is boxed. Several important residues interacting with the sugar donor are indicated by letters (a, b, and c) under the alignment.
2.3. Screening of Overexpressed UGTs in C. suppressalis with Chlorantraniliprole Resistance
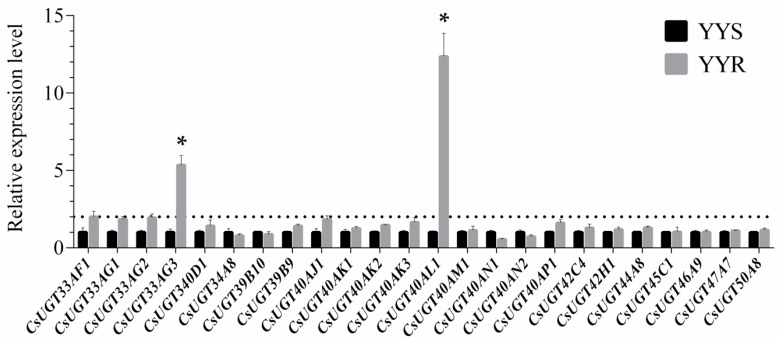
Among the 24 UGT genes, CsUGT40AL1 and CsUGT33AG3, which increased 12.36- and 5.34-fold in YYR, respectively, were significantly overexpressed in YYR compared to YYS (Figure 3). No significant differences between YYS and YYR were observed in the other 22 gene expressions.
Figure 3.
Relative expression levels of UGTs in YYS and YYR determined by real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR). Data were normalized to the expression of G3PDH and Actin A1. Error bars represent the standard deviation (SD) from the mean of three independent replicates. Thirty fourth instar larvae were sampled for each biological replicate. Asterisks (*) indicate a significant difference in the expression levels of UGTs between YYS and YYR using Student’s t-test (p < 0.05).
2.4. Expression Patterns of Overexpressed UGTs in Different Tissues
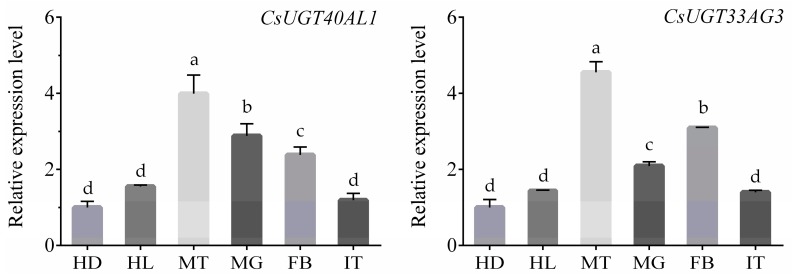
To investigate the tissue-specific expression patterns of the two overexpressed UGTs, qRT-PCR was carried out using RNA isolated from the head, hemolymph, Malpighian tubules, midgut, fat body, and integument tissue samples of C. suppressalis. CsUGT40AL1 and CsUGT33AG3 showed high expression levels in the Malpighian tubules (3.99- and 4.55-fold, respectively), midgut (2.88- and 2.09-fold, respectively), and fat body (2.38- and 3.09-fold, respectively), followed by the hemolymph (1.55- and 1.44-fold, respectively) and integument (1.19- and 1.40-fold, respectively). However, the relative expressions of the two UGTs were lowest in the head (Figure 4). The expression patterns of CsUGT40AL1 and CsUGT33AG3 may reflect their specialized roles in insecticide resistance.
Figure 4.
Expressions of CsUGT40AL1 and CsUGT33AG3 in different tissues of the fourth instar larvae of YYS. HD, head; HL, hemolymph; MT, Malpighian tubules; MG, midgut; FB, fat body; and IT, integument. The mRNA levels in the different tissues were normalized to the expressions in the head. Error bars represent the SD from the mean of three independent replicates. For each biological replicate, 30 fourth instar larvae were sampled. Different lowercase letters indicate significant differences among treatments as determined by one-way analysis of variance (ANOVA) followed by Tukey’s honest significant difference (Tukey’s HSD) test (p < 0.05).
2.5. Confirmation of Overexpressed UGT Genes Function
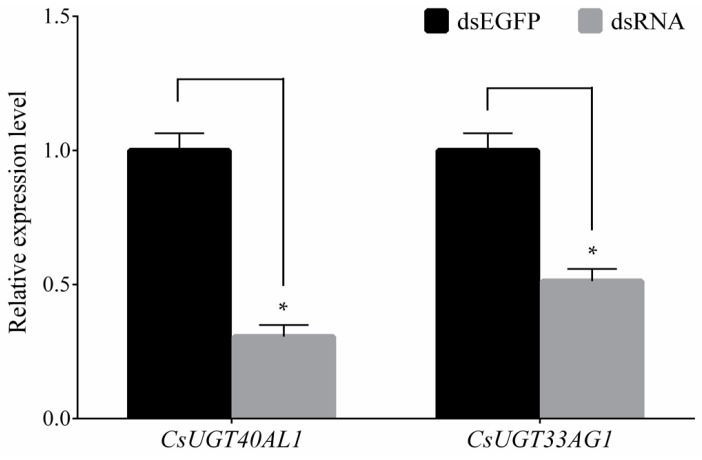
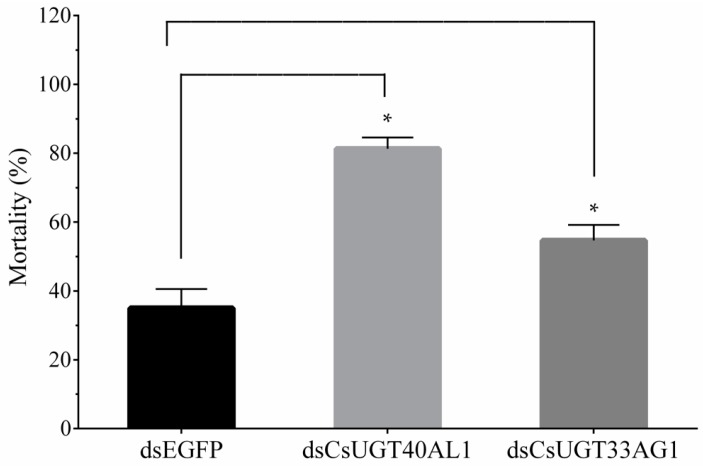
A further in vivo functional study was conducted using RNAi to silence the two overexpressed genes, followed by a chlorantraniliprole bioassay to evaluate the contribution of these genes to chlorantraniliprole resistance. After double-stranded RNA (dsRNA) injection, CsUGT40AL1 and CsUGT33AG3 expression greatly decreased by 69.34% and 48.74% compared to that after injection with double-stranded enhanced green fluorescent protein (dsEGFP), respectively (Figure 5). This indicated that dsRNA injection effectively silenced these target genes. The susceptibility of the dsRNA-injected YYR C. suppressalis to chlorantraniliprole was evaluated on the basis of fourth instar larval mortality. High mortality was observed after individual injection with CsUGT40AL1 (81.33%) and CsUGT33AG3 (54.67%), compared to that after dsEGFP injection (35.42%) (Figure 6). However, the mortalities showed no significant differences among the three dsRNA treatments in the absence of chlorantraniliprole (results not shown). These results suggested that the RNAi-mediated silencing of the two UGT genes could decrease the resistance of YYR to chlorantraniliprole.
Figure 5.
qRT-PCR analysis of UGT gene expression after individual UGT dsRNA injection in YYR of C. suppressalis. Data were normalized to the expression of G3PDH and Actin A1. Error bars represent the SD from the mean of three independent replicates. There were 30 fourth instar larvae for each biological replicate. Asterisks (*) indicate significant differences between treatments as determined by Student’s t-test (p < 0.05).
Figure 6.
Mortality of chlorantraniliprole-treated YYR strain of C. suppressalis larvae after individual dsRNA injection. Error bars represent the SD from the mean of three independent replicates. For each biological replicate, 30 fourth instar larvae were sampled. Asterisks (*) indicate significant differences between treatments as determined by Student’s t-test (p < 0.05).
3. Discussion
Since 2008, chlorantraniliprole has been registered and commercialized in China for agricultural usage to control the occurrence of several lepidopteran taxa, which provides a valid method for managing insecticide resistance [3]. The mode of its action can result in uncontrolled release of the calcium stores from muscle cells [7]. Although chlorantraniliprole is especially effective against C. suppressalis, frequent use of this insecticide has prompted rapid development of chlorantraniliprole resistance in this pest. A previous resistance monitoring study showed that C. suppressalis in the Jiangxi, Hunan, Henan, and Zhejiang provinces of China developed moderate to high resistance to chlorantraniliprole (64.10- to 249.60-fold) in the field [1,2,8]. In the present study, we found that C. suppressalis could develop high-level resistance to chlorantraniliprole in the laboratory under selection pressure, which was in line with previous findings [8,9]. The results indicated that C. suppressalis populations from different geographical areas seem to have a remarkable ability to develop resistance to this insecticide under continuous pressure. Additionally, several other lepidopteran pests, such as P. xylostella, Spodoptera exigue, and Spodoptera litura, developed high chlorantraniliprole resistance in China [18,19,20]. Despite the occurrence of resistance, chlorantraniliprole is still used in agriculture for lepidopteran pest management. To make good use of this insecticide and prolong its usage, an in-depth understanding of the resistance mechanisms is necessary. A susceptible YYS and a resistant YYR were established under intensive laboratory selection environments to decrease genetic diversity, which provided a genetic background to elucidate resistance mechanisms.
Understanding resistance mechanisms is a prerequisite for establishing resistance management strategies. An important metabolic resistance mechanism of chlorantraniliprole mediated by phase I enzyme P450s in C. suppressalis has been reported [9]; however, the increase in UGT enzyme activities is another main mechanism conferring insecticide resistance. The application of inhibitors is an effective way to verify an enzyme’s function. This study showed that the toxicities of chlorantraniliprole were significantly increased by the two UGT inhibitors (5-NU and SUL) in the YYR strains but not in the YYS strain, suggesting that enhanced UGTs enzyme activities may contribute to biochemical mechanisms of resistance in C. suppressalis. The two synergists had high potential for utilization in the management of chlorantraniliprole-resistant insects due to their significant synergistic effects. Therefore, a glycosylation by UGTs might play an important role in the detoxification of chlorantraniliprole in C. suppressalis.
Although UGT enzymes might be associated with chlorantraniliprole resistance in C. suppressalis, the specific genes that encoded this functional enzyme were not likely to have been previously identified. In this study, 24 UGT genes were identified by searching the C. suppressalis genome. The availability of whole genome and transcriptome sequences of various insects enables the characterization and comparative analysis of UGTs in these species. The total number of UGT genes identified was lower than that identified in three species, namely Helicoverpa armigera (40 genes), Bombyx mori (44 genes), and Drosophila melanogaster (34 genes), but higher than that in Spodoptera littoralis (11 genes) and similar to that in P. xylostella (23 genes) and Athetis lepigone (23 genes) [21,22,23,24,25,26]. Fifteen UGTs had signal peptide sequences at the N-terminal end. However, a signal peptide was absent from the other UGT genes, which were most likely non-secretory proteins, despite the complete sequence. Alignments of the C. suppressalis UGT amino acid sequences showed conserved domains, including DBR1 and DBR2 (important residues interacting with the sugar donor and catalytic residues), and the UGT motif signature sequences. These data suggested that C. suppressalis UGTs were likely active proteins, which is similar to observations reported for other lepidopteran taxa [25]. The evolutionary relationships of C. suppressalis UGTs suggest that the identified UGT transcripts form a complete UGT gene repertoire of C. suppressalis. The C. suppressalis UGTs were distributed into 11 families (i.e., UGT33, UGT34, UGT39, UGT40, UGT42, UGT44, UGT45, UGT46, UGT47, UGT50, and UGT340), which formed family clusters with other insect UGTs. Phylogenetic tree analysis of C. suppressalis UGTs revealed patterns of interspecific conservation and lineage-specific expansion of the gene families. Similar to other lepidopteran taxa, 4 and 9 UGTs were clustered in the UGT33 and UGT40 families, respectively, indicating that the divergence of these genes in the two families likely increased the range of compounds that could be detoxified or regulated by glycosylation [21,25]. Thus, these identified UGT genes in C. suppressalis could be used to further clarify their roles in chlorantraniliprole resistance.
The underlying UGTs that mediate metabolic resistance mechanisms were characterized by expression levels of UGT genes between the susceptible and resistant strains. In this study, CsUGT40AL1 and CsUGT33AG3 were significantly overexpressed in YYR, compared to YYS, which is in line with the synergism assays data, suggesting that overexpression of multiple UGT genes play a role in chlorantraniliprole resistance in C. suppressalis. The full-length cDNA sequences of the two UGT genes were obtained. The conserved signature motif position and conserved amino acid residues appeared to be diverse for each upregulated UGT gene, indicating the diversity of UGT genes involved in chlorantraniliprole resistance in C. suppressalis. The UGT33 and UGT40 family genes may have diverse functions, including detoxification of xenobiotics and insecticide resistance. Krempl et al. (2016) found that gossypol is partially metabolized by UGT40D1 via glycosylation in H. armigera and H. virescens, which might be a crucial step in gossypol detoxification in generalist herbivores utilizing cotton as host plant [27]. Some UGTs from the two families were overexpressed in the adult antennae of A. lepigone, suggesting that they contribute to the degradation of sex pheromones and plant volatiles, and/or insecticide detoxification [26]. UGT2B17 (renamed UGT33AA4) from the UGT33 family is involved in chlorantraniliprole resistance in P. xylostella [16,25]. The identified CsUGT40AL1 and CsUGT33AG3 sequences clustered into an own group separated from UGT2B17 (renamed UGT33AA4) from P. xylostella in phylogenetic tree, suggesting the specificity of UGTs in different insect species. Our study, combined with these examples, confirmed contributions of CsUGT40AL1 and CsUGT33AG3 in chlorantraniliprole resistance in C. suppressalis. Expression of CsUGT40AL1 and CsUGT33AG3 was high in the Malpighian tubules, midgut, and fat body, followed by the hemolymph and the integument but was significantly lower in the head. The insect Malpighian tubules, fat body, and midgut are well-known to play crucial roles in the metabolism of xenobiotics and restrict the spread of the insecticide and minimize its toxic effects [28]. Similar expression patterns of UGT genes were reported for P. xylostella [16], H. armigera [21], and B. mori [22]. This suggests that the two UGT genes highly expressed in those tissues may be involved in sequestering chlorantraniliprole and in the detoxification processes.
To confirm a functional role of the overexpressed UGT genes in chlorantraniliprole resistance, we performed gene-silencing RNAi assays. Such technology has previously been used to analyze gene functions in C. suppressalis and has shown potential for developing new pest control measures [9,16,29]. Our results showed that dsRNA injection significantly silenced the target genes. This notable knockdown of the target genes resulted in a higher larval mortality rate than control in the resistant strain, indicating a loss of resistance. The RNAi results indicated both UGTs contributed to chlorantraniliprole resistance. But the expression of CsUGT40AL1 was much higher than that of CsUGT33AG3, and tissue-specific expression patterns of CsUGT40AL1 and CsUGT33AG3 also showed significant differences. Therefore, further metabolism efficiencies were compared to evaluate the importance of the two UGT proteins in chlorantraniliprole resistance. The present study showed that UGTs mediated metabolic resistance of chlorantraniliprole in C. suppressalis, in addition to P450-mediated resistance.
4. Materials and Methods
4.1. Insects and Sample Preparation
The C. suppressalis colony was derived from a field population collected from a paddy field near Yuyao City, Zhejiang Province, China, in March 2016. After collection, a susceptible strain YYS was established by maintaining one part of the population on rice seedlings in the laboratory for 17 generations without exposure to insecticide. The rest of the population was continuously selected in the laboratory by treating fourth instar larvae with LC50 of chlorantraniliprole (using the diet surface overlay bioassay method [9]) for 17 generations to obtain a resistant strain of YYR. The C. suppressalis colony was reared on rice seedlings at 27 ± 1 °C with a humidity of 70–80% and 16-h light/8-h dark photoperiod in the laboratory.
Fourth instar larvae in YYS and YYR were collected to analyze tissue-specific expression patterns for the different strains. The fat body, midgut, hemolymph, integument, head, and Malpighian tubules were dissected from fourth instar larvae of the YYS strain, and washed with phosphate-buffered saline (PBS; 140 mM NaCl, 27 mM KCl, 8 mM Na2HPO4 and 1.5 mM KH2PO4 pH 7.4). Whole insect and tissue samples from those individuals were immediately frozen in liquid nitrogen and stored at −80 °C until RNA extraction. Each collection was replicated three times.
4.2. Toxicity and Synergism Bioassays
A stock solution of chlorantraniliprole (97.3%, TC; DuPont Trading Co., Ltd., Shanghai, China) was dissolved in N,N-dimethylformamide (Solarbio Science and Technology Co., Ltd., Beijing, China) and serially diluted into 5–6 concentrations with distilled water containing 0.1% Triton X-100 (Sinapharm Chemical Reagent Co., Ltd., Shanghai, China) as a cosolvent. The artificial diet was prepared as previously described [9]. The insecticide surface overlay diet was prepared by applying diluted chlorantraniliprole (100 μL) to the surface of the artificial diet (900 μL) in each well of a 24 well plate after the artificial diet solidified. A single fourth instar larva was transferred into each well when the liquid disappeared. Insects feeding the diet treated with water containing N,N-dimethylformamide and Triton X-100 were used as control. Each treatment was replicated three times for a total of 144 larvae per concentration. All treated insects were reared under the above rearing conditions. The mortality was checked after 72 h. Using a synergism bioassay, two UGT inhibitors, 5-NU and SUL (Sigma-Aldrich Trading Co., Ltd., Shanghai, China), were dissolved into a series of chlorantraniliprole concentrations with 0.1% Triton X-100 at a final concentration of 600 mg/L. The final concentrations of 5-NU and SUL were used in the maximum sublethal concentration that led to zero mortality in fourth instar larvae. Three replicates were performed for each concentration. All other procedures were the same as above. The LC50 values were calculated using probit analysis in the PoloPlus software (LeOra Software Inc., Petaluma, CA, USA). SRs were calculated as the LC50 values of the insecticide alone divided by the LC50 values of the insecticide with the synergist treatments. A significant difference was deemed to exist between LC50s when their 95% CL did not overlap.
4.3. RNA Isolation and cDNA Synthesis
Total RNA was extracted from samples using Trizol reagent (Life Technologies, Carlsbad, CA, USA) following the manufacturer’s instructions. The quality and quantity of RNA were checked using agarose gel electrophoresis (1.3%) and Nanodrop spectrophotometry (Thermo Fisher Scientific, Waltham, MA, USA). First-strand cDNA was synthesized by HiScript II Q RT SuperMix for qPCR (+gDNA wiper) (Vazyme biotech Co., Ltd., Nanjing, China) according to the manufacturer’s protocol.
4.4. Identification of UGT Genes and Phylogenetic Analysis
UGT gene sequences were obtained by querying genome databases of C. suppressalis (http://www.insect-genome.com/) with annotated gene names [30]. Cap3 software was used to identify the overlaps between different sequences and to remove redundant fragments (http://doua.prabi.fr/software/cap3). Uridine diphosphate glycosyltransferase conserved domains ‘UDPGT’ of UGT genes were analyzed by using the SMART online software (http://smart.embl-heidelberg.de/) [31]. Each of the putative UGT sequences was used as a query to search the NCBI non-redundant protein database using BLASTX to validate further the potential UGTs (http://www.ncbi.nlm.nih.gov/). Finally, the putative 24 UGT genes with complete ORFs were obtained. The accuracy of these putative UGT genes was substantiated by RT-PCR using the primers detailed in Table S1 (Supplementary Materials), and these were submitted to GenBank. The complete ORFs of C. suppressalis UGT genes were predicted using the ORF finder tool (http://www.ncbi.nlm.nih.gov/gorf/gorf.html). The SPs were predicted using SignalP 4.1 (http://www.cbs.dtu.dk/services/SignalP/). The PIs and MWs of deduced proteins were calculated using the ExPASy Compute pI/Mw tool (http://web.expasy.org/compute_pi/). A set of 163 UGT gene sequences from 5 species, namely S. exigua (32 sequences), B. mori (44 sequences), H. armigera (40 sequences), P. xylostella (23 sequences) and C. suppressalis (24 sequences), were obtained from the NCBI database. The names and accession numbers of the genes used for phylogenetic tree building are listed in Table S2 (Supplementary Materials). Amino acid sequences were aligned with ClustalX 2.0 and unrooted trees were constructed by MEGA 5.0 using the neighbor-joining method with Poisson correction of distances [32,33,34]. Node support was assessed using a bootstrap procedure based on 1000 replicates.
4.5. Expression Patterns of UGT Genes in Resistant and Susceptible Strains
qRT-PCR was performed to determine the expression patterns of UGT genes in different strains and tissues using an Applied Biosystems™ QuantStudio™ 6 Flex Real-Time PCR System. Primers were designed by Beacon Designer 7.0 (Premier Biosoft International, Palo Alto, CA, USA) and are listed in Table S3 (Supplementary Materials). A ChamQTM SYBR® qPCR Master Mix (Vazyme biotech Co., Ltd., Nanjing, China) was used according to the manufacturer’s protocol. Normalization of each experimental gene expression level was conducted based on the geometric mean of two selected reference genes, G3PDH and Actin A1 [8]. The expression stability of the two reference genes was analyzed with geNorm Software [35]. Data were calculated according to the 2−ΔΔCt method.
4.6. Functional Analysis of Overexpressed UGT Genes with RNAi
Total RNA was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA), and the first-strand cDNA was synthesized as previously described. The specific primers for dsRNA synthesis were designed on the basis of sequences of overexpressed UGT genes and the fragment sequence of an enhanced green fluorescent protein (EGFP), which contained T7 polymerase promoter sequence at both ends (Table S4, Supplementary Materials). The PCR products amplified by RT-PCR were purified using Wizard®SV gel and the PCR clean-up system (Promega, Madison, WI, USA) according to the manufacturer’s protocol and were used as templates for in vitro transcription using the T7 RiboMAXTM Express RNAi System (Promega, Madison, WI, USA) to synthesize dsRNA. All dsRNAs were resuspended in nuclease-free water and quantified spectrophotometrically. These dsRNAs were examined by agarose gel electrophoresis to verify that the products exhibited the expected sizes in a single band. Then they were stored at −80 °C until use.
RNAi experiments were performed using the dsRNA microinjection method. The dsRNA was dissolved in diethyl pyrocarbonate-treated water to a final concentration of 4 µg/µL. Then dsRNA (1 µL, 4 µg) was injected into the larvae using a microneedle. The needles were pulled from glass capillaries (outer diameter, 1 mm; inner diameter, 0.5 mm) using a micropipette puller (Model P-97, Sutter Instruments Co., Novato, CA, USA). The needles were held still at the injection point for 30 s to avoid small interfering RNA leakage. The dsRNA injection was performed using an Eppendorf InjectMan NI 2 microinjection system (Eppendorf, Hamburg, Germany). Thirty fourth instar larvae in YYR were used for each treatment, and all experiments were performed in triplicate.
Three hours after interfering, 30 surviving larvae from each treatment were treated according to the preliminary bioassay method described above. The treated C. suppressalis were kept under the rearing conditions described above, and the mortality was checked after 72 h.
To estimate the target gene silencing effect following RNAi, qRT-PCR was used to measure the expression levels of the target genes in YYR. qRT-PCR was performed as previously described. The primers used are listed in Table S4 (Supplementary Materials).
4.7. Statistical Analysis
Data are presented as mean ± standard deviation (SD). Student’s t-test was used for two sample comparisons. One-way analysis of variance (ANOVA) followed by Tukey’s honest significant difference (Tukey’s HSD) test was used for multiple group comparisons. All statistical analyses were performed using SPSS 20.0 software (SPSS Inc., Chicago, IL, USA). p < 0.05 was considered statistically significant.
5. Conclusions
The present study strongly supports the conclusion that the overexpression of UGTs is responsible for resistance to chlorantraniliprole in C. suppressalis. These results, in conjunction with the involvement of UGT in chlorantraniliprole resistance of P. xylostella, provide evidence that overexpression of UGTs in chlorantraniliprole resistance may be a common mechanism in lepidopteran pests. Future pest management decisions using chlorantraniliprole to control lepidopterans should take this into account.
Acknowledgments
We thank Michael H. Court (member of the UGT Nomenclature Committee) for the nomenclature of Chilo suppressalis UGTs.
Abbreviations
| UGT | Uridine diphosphate glycosyltransferase |
| UDP | Uridine diphosphate |
| RyR | Ryanodine receptor |
| RNAi | RNA interference |
| dsRNA | Double-stranded RNA |
| 5-NU | 5-nitrouracil |
| SUL | Sulfinpyrazone |
| RR | Resistance ratio |
| SR | Synergism ratio |
| CL | Confidence limits |
| ORF | Open reading frame |
| MW | Molecular weight |
| PI | Isoelectric point |
| SP | Signal peptide |
| EGFP | Enhanced green fluorescent protein |
| qRT-PCR | Real-time quantitative reverse transcription polymerase chain reaction |
| RT-PCR | Reverse transcription polymerase chain reaction |
| PBS | Phosphate-buffered saline |
| SD | Standard deviation |
| ANOVA | Analysis of variance |
| Tukey’s HSD | Tukey’s honest significant difference |
Supplementary Materials
Supplementary materials can be found at https://www.mdpi.com/1422-0067/20/5/1064/s1.
Author Contributions
Conceptualization, Z.H.; methodology, J.Z., Y.S., and L.X.; validation, J.Z., Y.S., P.S., and L.X.; formal analysis, J.Z., Y.S., and L.X.; investigation, J.Z., Y.S., P.S., and L.X.; resources, L.X. and Z.H.; data curation, Z.H.; writing—original draft preparation, J.Z. and L.X.; writing—review and editing, J.Z., Y.S., P.S., L.X., and Z.H.; visualization, J.Z., Y.S., P.S., and L.X.; supervision, Z.H.; project administration, Z.H.; funding acquisition, Z.H.
Funding
This work was supported by the Special Fund for Agro-scientific Research in the Public Interest of China (Grant No.201303017).
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Lu Y.H., Wang G.R., Zhong L.Q., Zhang F.C., Bai Q., Zheng X.S., Lu Z.X. Resistance monitoring of Chilo suppressalis (Walker) (Lepidoptera: Crambidae) to chlorantraniliprole in eight field populations from east and central China. Crop. Prot. 2017;100:196–202. doi: 10.1016/j.cropro.2017.07.006. [DOI] [Google Scholar]
- 2.Huang S.J., Chen Q., Qin W.J., Sun Y., Qin H.G. Resistance monitoring of four insecticides and a description of an artificial diet incorporation method for Chilo suppressalis (Lepidoptera: Crambidae) J. Econ. Entomol. 2017;110:2554–2561. doi: 10.1093/jee/tox266. [DOI] [PubMed] [Google Scholar]
- 3.Su J.Y., Zhang Z.Z., Wu M., Gao C.F. Geographic susceptibility of Chilo suppressalis Walker (Lepidoptera: Crambidae), to chlorantraniliprole in China. Pest Manag. Sci. 2014;70:989–995. doi: 10.1002/ps.3640. [DOI] [PubMed] [Google Scholar]
- 4.Sun Y., Huang S.J., Wang S.P., Guo D.H., Ge C., Xiao H.M., Jie W.C., Yang Q.P., Teng X.L., Li F. Large-scale identification of differentially expressed genes during pupa development reveals solute carrier gene is essential for pupal pigmentation in Chilo suppressalis. J. Insect Physiol. 2017;98:117–125. doi: 10.1016/j.jinsphys.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 5.Cordova D., Benner E.A., Sacher M.D., Rauh J.J., Sopa J.S., Lahm G.P., Selby T.P., Stevenson T.M., Flexner L., Gutteridge S., et al. Anthranilic diamides: A new class of insecticides with a novel mode of action, ryanodine receptor activation. Pestic. Biochem. Physiol. 2006;84:196–214. doi: 10.1016/j.pestbp.2005.07.005. [DOI] [Google Scholar]
- 6.He Y.P., Zhang J.F., Gao C.F., Su J.Y., Chen J.M., Shen J.L. Regression analysis of dynamics of insecticide resistance in field populations of Chilo suppressalis (Lepidoptera: Crambidae) during 2002–2011 in China. J. Econ. Entomol. 2013;106:1832–1837. doi: 10.1603/EC12469. [DOI] [PubMed] [Google Scholar]
- 7.Yao R., Zhao D.D., Zhang S., Zhou L.Q., Wang X., Gao C.F., Wu S.F. Monitoring and mechanisms of insecticide resistance in Chilo suppressalis (Lepidoptera: Crambidae), with special reference to diamides. Pest Manag. Sci. 2017;73:1169–1178. doi: 10.1002/ps.4439. [DOI] [PubMed] [Google Scholar]
- 8.Sun Y., Xu L., Chen Q., Qin W.J., Huang S.J., Jiang Y., Qin H.G. Chlorantraniliprole resistance and its biochemical and new molecular target mechanisms in laboratory and field strains of Chilo suppressalis (Walker) Pest Manag. Sci. 2018;74:1416–1423. doi: 10.1002/ps.4824. [DOI] [PubMed] [Google Scholar]
- 9.Xu L., Zhao J., Sun Y., Xu D., Xu G., Xu X., Zhang Y., Huang S., Han Z., Gu Z. Constitutive overexpression of cytochrome P450 monooxygenase genes contributes to chlorantraniliprole resistance in Chilo suppressalis (Walker) Pest Manag. Sci. 2019;75:718–725. doi: 10.1002/ps.5171. [DOI] [PubMed] [Google Scholar]
- 10.Mackenzie P.I., Owens I.S., Burchell B., Bock K.W., Bélanger A., Belanger A., Fournel-Gigleux S., Green M., Hum D.W., Iyanagi T., et al. The UDP glycosyltransferase gene superfamily: Recommended nomenclature update based on evolutionary divergence. Pharmacogenetics. 1997;7:255–269. doi: 10.1097/00008571-199708000-00001. [DOI] [PubMed] [Google Scholar]
- 11.Bock K.W. Vertebrate UDP-glucuronosyltransferases: functional and evolutionary aspects. Biochem. Pharmacol. 2003;66:691–696. doi: 10.1016/S0006-2952(03)00296-X. [DOI] [PubMed] [Google Scholar]
- 12.Bull D.L., Whitten C.J. Factors influencing organophosphorus insecticide resistance in tobacco budworms. J. Agric. Food. Chem. 1972;20:561. doi: 10.1021/jf60181a061. [DOI] [PubMed] [Google Scholar]
- 13.Lee S.W., Ohta K., Tashiro S., Shono T. Metabolic resistance mechanisms of the housefly (Musca domestica) resistant to pyraclofos. Pestic. Biochem. Physiol. 2006;85:76–83. doi: 10.1016/j.pestbp.2005.10.005. [DOI] [Google Scholar]
- 14.Kaplanoglu E., Chapman P., Scott I.M., Donly C. Overexpression of a cytochrome P450 and a UDP-glycosyltransferase is associated with imidacloprid resistance in the Colorado potato beetle, Leptinotarsa decemlineata. Sci. Rep. 2017;7:1762. doi: 10.1038/s41598-017-01961-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pan Y.O., Tian F.Y., Wei X., Wu Y.Q., Gao X.W., Xi J.H., Shang Q.L. Thiamethoxam resistance in Aphis gossypii glover relies on multiple UDP-glucuronosyltransferases. Front. Physiol. 2018;9:322. doi: 10.3389/fphys.2018.00322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li X.X., Zhu B., Gao X.W., Liang P. Over-expression of UDP-glycosyltransferase gene UGT2B17 is involved in chlorantraniliprole resistance in Plutella xylostella (L.) Pest Manag. Sci. 2017;73:1402–1409. doi: 10.1002/ps.4469. [DOI] [PubMed] [Google Scholar]
- 17.Mackenzie P.I., Bock K.W., Burchell B., Guillemette C., Ikushiro S., Iyanagi T., Miners J.O., Owens I.S., Nebert D.W. Nomenclature update for the mammalian UDP glycosyltransferase (UGT) gene superfamily. Pharmacogenet. Genom. 2005;15:677–685. doi: 10.1097/01.fpc.0000173483.13689.56. [DOI] [PubMed] [Google Scholar]
- 18.Wang X.L., Wu Y.D. High levels of resistance to chlorantraniliprole evolved in field populations of Plutella xylostella. J. Econ. Entomol. 2012;105:1019–1023. doi: 10.1603/EC12059. [DOI] [PubMed] [Google Scholar]
- 19.Lai T.C., Li J., Su J.Y. Monitoring of beet armyworm Spodoptera exigua (Lepidoptera: Noctuidae) resistance to chlorantraniliprole in China. Pestic. Biochem. Physiol. 2011;101:198–205. doi: 10.1016/j.pestbp.2011.09.006. [DOI] [Google Scholar]
- 20.Su J.Y., Lai T.C., Li J. Susceptibility of field populations of Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae) in China to chlorantraniliprole and the activities of detoxification enzymes. Crop Prot. 2012;42:217–222. doi: 10.1016/j.cropro.2012.06.012. [DOI] [Google Scholar]
- 21.Ahn S.J., Vogel H., Heckel D.G. Comparative analysis of the UDP-glycosyltransferase multigene family in insects. Insect Biochem. Mol. Biol. 2012;42:133–147. doi: 10.1016/j.ibmb.2011.11.006. [DOI] [PubMed] [Google Scholar]
- 22.Huang F.F., Chai C.L., Zhang Z., Liu Z.H., Dai F.Y., Lu C., Xiang Z.H. The UDP-glucosyltransferase multigene family in Bombyx mori. BMC Genomics. 2008;9:563. doi: 10.1186/1471-2164-9-563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Luque T., O’Reilly D.R. Functional and phylogenetic analyses of a putative Drosophila melanogaster UDP-glycosyltransferase gene. Insect Biochem. Mol. Biol. 2002;32:1597–1604. doi: 10.1016/S0965-1748(02)00080-2. [DOI] [PubMed] [Google Scholar]
- 24.Bozzolan F., Siaussat D., Maria A., Durand N., Pottier M.A., Chertemps T., Maïbèche-Coisne M. Antennal uridine diphosphate (UDP)-glycosyltransferases in a pest insect: diversity and putative function in odorant and xenobiotics clearance. Insect Mol. Biol. 2014;23:539–549. doi: 10.1111/imb.12100. [DOI] [PubMed] [Google Scholar]
- 25.Li X., Shi H., Gao X., Liang P. Characterization of UDP-glucuronosyltransferase genes and their possible roles in multi-insecticide resistance in Plutella xylostella (L.) Pest Manag. Sci. 2018;74:695–704. doi: 10.1002/ps.4765. [DOI] [PubMed] [Google Scholar]
- 26.Zhang Y.N., Ma J.F., Xu L., Dong Z.P., Xu J.W., Li M.Y., Zhu X.Y. Identification and expression patterns of UDP-glycosyltransferase (UGT) genes from insect pest Athetis lepigone (Lepidoptera: Noctuidae) J. Asia-Pac. Entomol. 2017;20:253–259. doi: 10.1016/j.aspen.2017.01.008. [DOI] [Google Scholar]
- 27.Krempl C., Sporer T., Reichelt M., Ahn S., Heidel-Fischer H., Vogel H., Heckel D.G., Joußen N. Potential detoxification of gossypol by UDP-glycosyltransferases in the two Heliothine moth species Helicoverpa armigera and Heliothis virescens. Insect Biochem. Mol. Biol. 2016;71:49–57. doi: 10.1016/j.ibmb.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 28.Xu L., Zhao C.Q., Xu D.J., Xu G.C., Xu X.L., Han Z.J., Zhang Y.N., Gu Z.Y. RNAi suppression of nuclear receptor genes results in increased susceptibility to sulfoxaflor in brown planthopper, Nilaparvata lugens. J. Asia-Pac. Entomol. 2017;20:645–653. doi: 10.1016/j.aspen.2017.03.022. [DOI] [Google Scholar]
- 29.Tian F., Wang Z., Li C., Liu J., Zeng X. UDP-Glycosyltransferases are involved in imidacloprid resistance in the Asian citrus psyllid, Diaphorina citri (Hemiptera: Lividae) Pestic. Biochem. Physiol. 2019;154:23–31. doi: 10.1016/j.pestbp.2018.12.010. [DOI] [PubMed] [Google Scholar]
- 30.Yin C.L., Shen G.Y., Guo D.H., Wang S.P., Ma X.Z., Xiao H.M., Liu J.D., Zhang Z., Liu Y., Zhang Y.Q., et al. InsectBase: a resource for insect genomes and transcriptomes. Nucleic Acids Res. 2016;44:D801–D807. doi: 10.1093/nar/gkv1204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang S., Liu Y., Zhou J.J., Yi J.K., Pan Y., Wang J., Zhang X.X., Wang J.X., Yang S., Xi J.H. Identification and tissue expression profiling of candidate UDP-glycosyltransferase genes expressed in Holotrichia parallela motschulsky antennae. Bull. Entomol. Res. 2018;108:807–816. doi: 10.1017/S0007485318000068. [DOI] [PubMed] [Google Scholar]
- 32.Larkin M.A., Blackshields G., Brown N.P., Chenna R., McGettigan P.A., McWilliam H., Valentin F., Wallace I.M., Wilm A., Lopez R., et al. Clustal W and Clustal X version 2.0. Bioinformatics. 2007;23:2947–2948. doi: 10.1093/bioinformatics/btm404. [DOI] [PubMed] [Google Scholar]
- 33.Tamura K., Peterson D., Peterson N., Stecher G., Nei M., Kumar S. MEGA5: molecular gvolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011;28:2731–2739. doi: 10.1093/molbev/msr121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Thompson J.D., Higgins D.G., Gibson T.J. Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994;22:4673–4680. doi: 10.1093/nar/22.22.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hellemans J., Mortier G., Paepe D.A., Speleman F., Vandesompele J. qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol. 2007;8:R19. doi: 10.1186/gb-2007-8-2-r19. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.