Abstract
PIEZO channels are stretch-activated channels involved in wound sealing and cell proliferation in many cell types. A recent study focussing on lung cancer (LC), using next-generation sequencing analysis, has indicated that PIEZO functions were implicated in LC development. However, the expression and role of PIEZO channels in non-small cell LC (NSCLC) progression require elucidation. In the current study, we investigated the gene expression and alteration frequency in human NSCLC tissue, accessed the prognostic roles of PIEZO channels in NSCLC patients, and further studied the effect of PIEZOs in NSCLC cell proliferation and tumor growth in vivo. The mRNA expression of PIEZO1 and 2 was clearly decreased in NSCLC tumor tissue compared with that in matched adjacent non-tumor tissue. In human NSCLC tissues, PIEZO1 gene expression exhibits a highly deep deletion rate, and PIEZO2 mainly exhibits mutation in gene expression. High mRNA expression of PIEZO channels was found to correlate with better overall survival (OS) for NSCLC patients, especially for patients with lung adenocarcinoma (LUAD), but not for patients with lung squamous cell carcinoma (LUSC). The prognostic role of PIEZO channels was more sensitive in female patients than male patients, and more sensitive in patients at earlier stages than patients at latter stages. Knockdown of PIEZO1 or PIEZO2 in NSCLC cells significantly promoted cell migration in vitro and tumor growth in vivo. These results indicate the critical prognostic values of the PIEZO channels in NSCLC. This information will be beneficial to understand the pathological mechanism of NSCLC and to generate effective therapeutic approaches for NSCLC patients.
Keywords: cell migration, NSCLC, PIEZO, Prognostic roles
Introduction
Lung cancer (LC) is one of the major causes of deaths worldwide, killing 8.2 million people annually [1]. Like other cancers, it is characterized by the rapid division and uncontrolled growth of cells, in this instance, in lung tissue. Non-small cell LC (NSCLC) represents 85–90% of all LC, and mainly includes lung squamous cell carcinoma (LUSC) and lung adenocarcinoma (LUAD) [1]. Radiation, heavy metals, genotoxic agents, cigarette smoke, and other non-genetic factors are associated with NSCLC [2,3]. In terms of carcinogens, a covalent carcinogen—DNA adduct may result in LC by causing misincorporation leading to genetic mutations [4]. Research focussing on genetic reasons for NSCLC has demonstrated that epidermal growth factor receptor (EGFR) is the most commonly mutated protein that results in LC. Approximately 90% of EGFR mutations in LC are a result of deletion in exon 19 affecting either the conserved sequence LREA (delE746-A750) or Leucine to Arginine at 858 (L858R) [5]. However, the molecular mechanism resulting in the pathogenesis of LC is still not fully understood.
PIEZO homologs are found in diverse organisms, such as invertebrates, protozoa, and plants [6]. They are non-selective Ca2+-permeable cation channels that act as important mediators of various aspects of mechanotransduction [7,8], and with regard to mechanical functions, they are abundant in organs, including the skin, bladder, lungs, and somatosensory dorsal root ganglion (DRG) neurones [9]. Many studies have recently indicated that PIEZO channels are highly expressed in human lung tissue and would be an important factor in lung diseases, especially in LC. However, there is still little understanding of the function of PIEZO1 and 2 in NSCLC progression [10]. When it is expressed in endothelial cells, PIEZO1 plays a key role in sensing blood flow-caused shear stress, which is important for blood vessel development [11]. On the other hand, proprioception and touch sensation are mediated by PIEZO2 and specialized touch receptors that are located in primary sensory neurones and the skin. More importantly, various genetic diseases caused by alteration of channel properties are associated with mutations in human PIEZO1 and PIEZO2 genes [12]. PIEZO1 is able to control epithelial cell crowding and division, determine neural stem cell lineage [13], and regulate blood pressure and exercise performance [14,15]. According to recent research, mutations in the PIEZO1 gene of humans contribute to anemia (dehydrated stomatocytosis) and generalized lymphatic dysplasia [16,17]. In vitro experiments show that knockdown of PIEZO1 in lung epithelial cells promotes cell migration and reduces cell adherence, suggesting that lack of PIEZO1 expression may lead to cell migration and metastasis in lung tumors [18]. PIEZO2 is important in touch sensation and the airway stretch sensation mediated by sensory neurones [19–21]. PIEZO2 gene mutations are responsible for distal arthrogryposis and other diseases [22,23]. PIEZO2 knockdown has been shown to promote anchorage-independent growth in premalignant human fibroblasts [24]. Since there are no reports on the prognostic roles of PIEZOs in NSCLC patients, we investigated the expression and roles of PIEZOs in this patient group.
Materials and methods
Analysis of gene alteration frequency in NSCLC
We analyzed the gene alteration frequency of PIEZO1 and 2 in NSCLC patients from the The Cancer Genome Atlas (TCGA) database using the cBioportal for cancer genomics analysis (http://www.cbioportal.org/) [25].
Expression of PIEZO channels in NSCLC tissues
We queried the expression level of PIEZO1 and 2 in NSCLC from the Gene Expression Omnibus (GEO), and two original datasets were downloaded (GSE10072 and GSE19804). The differentially expressed mRNA for PIEZO1 and 2 in NSCLC samples (GSE10072 and GSE19804) and adjacent non-tumor tissues were used for analysis.
Collection of human LC tissue samples and ethics statement
LC tissues and paired adjacent non-tumor normal lung tissues from NSCLC patients were obtained at Jilin Province Cancer Hospital in 2012, as previously described [26]. All fresh tissues were stored at −80°C until subsequent experiments. The study was approved by the Ethical Committee of Jilin Province Cancer Hospital. An informed consent was obtained from all the participants before enrollment in the study. The entire study was performed based on the Declaration of Helsinki.
Human LC cell culture
Human LC cell (A549, CCL-185) was obtained from the American Type Culture Collection (ATCC) and cultured as described previously [26]. A549 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% FBS (Sigma–Aldrich, Inc., St. Louis, MO, U.S.A.) at 37°C in a 5% CO2 humidified atmosphere.
Real-time quantitative PCR analysis of gene expression
Total RNA from human tissue samples and cells was extracted using TRIzol reagent (Invitrogen, Inc; Carlsbad, CA, U.S.A.) and 1 µg of total mRNA was reverse-transcribed into cDNA by using a reverse transcription kit (Bio-Rad, Inc; Hercules, CA, U.S.A.). Real-time quantitative PCR (RT-qPCR) analysis of gene expression used the following primers: PIEZO1, forward primer: 5′-GGACTCTCGCTGGTCTACCT-3′; PIEZO1, reverse primer: 5′-GGGCACAATATGCAGGCAGA-3′; PIEZO2, forward primer: 5′-ATGGCCTCAGAAGTGGTGTG-3′; PIEZO2, reverse primer: 5′- ATGTCCTTGCATCGTCGTTTT-3′; glyceraldehyde-3-phosphate dehydrogenase (GAPDH), forward primer: 5′-ACAACTTTGGTATCGTGGAAGG-3′; GAPDH, reverse primer: 5′-GCCATCACGCCACAGTTTC-3′. RT-qPCR analysis was performed using SYBR Premix ExTaq (Takara). The levels of PCR products were monitored using an Mx3000P QPCR system (Agilent, Santa Clara, CA, U.S.A.). The thermal cycling conditions were as follows: 10 s at 95°C, 40 cycles of 5 s at 95°C, and 30 s at 60°C. The mRNA expression of PIEZO1 and PIEZO2 was normalized to the constitutive expression level of GAPDH mRNA.
Antibodies and Western blot analysis
Protease inhibitor cocktail tablets (EDTA-free complete) were from Sigma–Aldrich, Inc. (St. Louis, MO, U.S.A.). Rabbit anti-PIEZO1, anti-PIEZO2, and mouse anti-GAPDH antibodies were purchased from Thermo Fisher Scientific, Inc. (Waltham, MA, U.S.A.). Horseradish peroxidase (HRP)-linked anti-mouse IgG and anti-rabbit IgG antibodies were obtained from Bio-Rad Laboratories, Inc. (Hercules, CA, U.S.A.). Immunoblot analysis was performed as described previously [26]. Briefly, cell lysates were prepared in lysis buffer containing EDTA-free complete protease inhibitors, followed by centrifugation at 10000×g for 10 min, and boiled with Laemmli sample buffer for 5 min. Cell lysates (20 µg protein) were separated on 10% or 4–20% SDS/PAGE, then transferred to polyvinylidene difluoride (PVDF) membranes, and blocked with TBST containing 5% BSA prior to incubation with primary antibodies (1:1000 dilution) overnight, and secondary antibodies (1:2000 dilution) for 2 h at room temperature. Blots were developed using the ECL chemiluminescence kit, and the integrated density of pixels in each membrane was quantitated using Image Quant 5.2 software (Molecular Dynamics, Sunnyvale, CA, U.S.A.).
Prognostic role of PIEZO expression in NSCLC
We used a public database to study the relevance and significance of the PIEZO expression level to overall survival (OS) in NSCLC patients. The data regarding NSCLC patients used for the Kaplan–Meier (KM) plotter analysis were pooled from TCGA, (http://cancergenome.nih.gov), GEO (http://www.ncbi.nlm.nih.gov/geo/), European genome-phenome archive (EGA) (https://ega.crg.eu/), and PubMed (http://www.pubmed.com) [27]. The database included the mRNA expression of two PIEZO channels and survival information (20 years) from 1432 NSCLC patients.
To obtain KM plots for patient OS rates, we used the KM plotter database (http://kmplot.com/analysis/index.php?p=service&cancer=lung). Based on the expression of genes, the patients were divided into two groups (high expression: mRNA expression higher than the median separates; low expression: mRNA expression lower than the median separates). Hazard ratio (HR), 95% confidence intervals, and log rank P, were also calculated from the database, and are included in the figures and tables of this manuscript. P<0.05 was used to indicate a statistically significant difference [28].
shRNA transfection
Vehicle control shRNA (Veh shRNA, SHC016), human PIEZO1 sh-RNA (sh-PIEZO1, TRCN0000121969), and PIEZO2 sh-RNA (sh-PIEZO2, TRCN0000123253) were purchased from Sigma–Aldrich, Inc. (St. Louis, MI, U.S.A.). A549 cells cultured on six-well plates (50–60% confluence) were transiently transfected with Veh sh-RNA or sh-PIEZO1 or sh-PIEZO2 (3 µg/well) using a plasmid transfection kit (Qiagen, Inc., Gaithersburg, MD, U.S.A.) according to the manufacturer’s instructions. Forty-eight hours post-transfection, cells were selected and cultured in growth medium containing puromycin (1 µg/ml). Two weeks post-transfection, the expression of PIEZO1 and 2 in cells was analyzed using RT-qPCR.
Scratch assay
A549 cells, with stable transfection of shRNA (Veh shRNA, sh-PIEZO1, or sh-PIEZO2), were grown to confluence in complete DMEM containing 1 µg/ml puromycin. At time 0 h, a 2-mm scrape wound was created with a pipette tip. Dead and floating cells were washed out from the complete medium. The remaining cells were continuously cultured in complete medium containing Mitomycin C (1 μg/ml) [29]. Cell migration was recorded at 0 and 24 h from at least three independent experiments. Wound healing was quantitated using Image Quant 5.2 software (Molecular Dynamics, Sunnyvale, CA, U.S.A.) and normalized with the wound healing in the cells transfected with Veh shRNA. Briefly, % of wound healing = (wound area at 0 h – wound area at 24 h)/wound area at 0 h × 100% [30].
Mice and in vivo tumor growth
Nude mice (female, 6 weeks old) were obtained from the Animal Center of the Chinese Academy of Science (Shanghai, China), and maintained in the nude mice care center of the Jilin Cancer Hospital. A549 cells with stable transfection of Veh shRNA, sh-PIEZO1, and sh-PIEZO2 were subcutaneously injected into nude mice (1 × 107 cell/ml, 50 µl). After injection, the tumor nodules were measured every week with a caliper. The tumor volume was calculated using: tumor volume (mm3) = π/6 × a × b2, where a is the longest diameter and b is the shortest diameter. During the 8-week follow-up period, the tumor size of nude mice was recorded.
Statistical analysis
All data are expressed as means ± S.E.M. from at least three independent experiments. Results were subjected to statistical analysis using one-way ANOVA or two-tailed Student’s t test as described before [26]. Values of P<0.05 were considered significant.
Results
Decreased expression of PIEZO channels in NSCLC
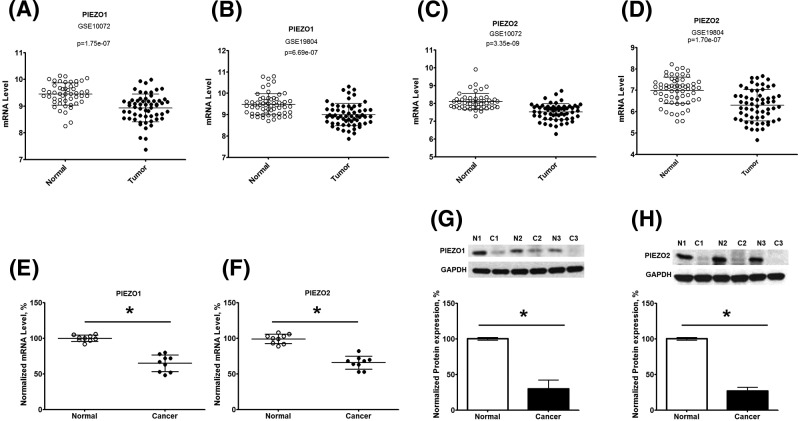
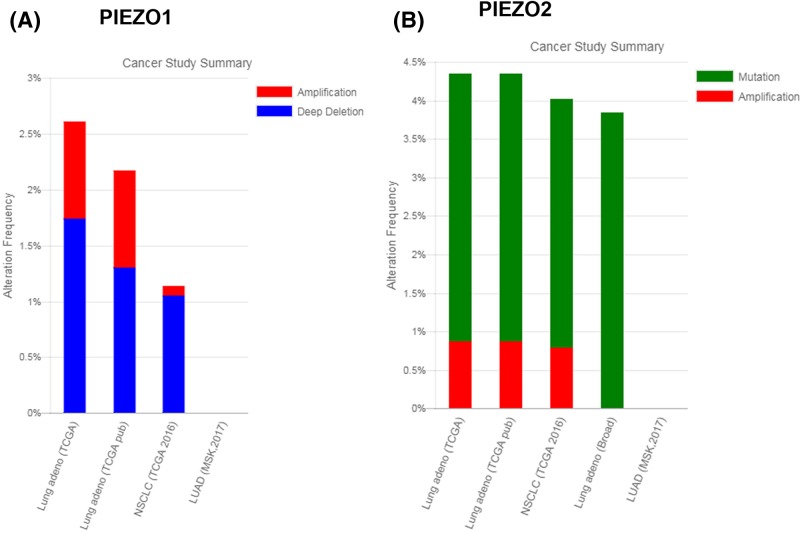
First, we analyzed the mRNA expression of PIEZO1 and 2 in NSCLC tissues and normal tissues from microarray. Raw microarray data were retrieved using the search terms ‘GSE#10072’ and ‘GSE19804’ in the GEO dataset. The analysis indicated that mRNA expression of PIEZO1 and 2 in NSCLC tissues was significantly lower than that in non-tumor tissues (P<0.0001; Figure 1A–D). Next, we used RT-qPCR to analyze the mRNA and protein expression of PIEZO1 and PIEZO2 in LC and adjacent control tissues from NSCLC patients, which were collected from our hospital in 2012. The mRNA expression of PIEZO1 and 2 in human tissues was analyzed by RT-qPCR and normalized to the expression of GAPDH. As shown in Figure 1E–H, the mRNA (E,F) and protein (G,H) expression of PIEZO1 (Figure 1E,G) and PIEZO2 (Figure 1F,H) in cancer tissue from NSCLC patients were significantly lower than that in the adjacent non-cancer tissues. Next, we analyzed the gene alteration frequency of PIEZO1 and 2 in NSCLC tissues. We found that there was a high deep deletion rate of PIEZO1 gene in NSCLC (Figure 2A), and a high gene mutation rate of the PIEZO2 gene in NSCLC (Figure 2B). These results indicate that PIEZO1 and 2 are decreased in NSCLC tissues in comparison with matched adjacent non-tumor tissue, which may be due to gene alteration of PIEZO channels during NSCLC progression.
Figure 1. Expression of PIEZO1 and 2 in normal and tumor tissues from human NSCLC patients.
mRNA and protein expression of PIEZO1 and 2 in lung tumors was compared with the expression in adjacent normal tissues. (A,B) Expression levels of PIEZO1 in human lc tissues compared with normal tissues in GSE10072 (A) and GSE19804 (B). (C,D) Expression levels of PIEZO2 in human LC tissues compared with normal tissues in GSE10072 (C) and GSE19804 (D). (E,F) mRNA expression of PIEZO1 (E) and PIEZO2 (F) in cancer tissue and adjacent normal tissues from human NSCLC patients were collected from Jilin Hospital. The mRNA expression was analyzed by RT-qPCR, and normalized to the expression of GAPDH. (G,H) Protein expression of PIEZO1 (G) and PIEZO2 (H) in cancer tissue and adjacent normal tissues. Upper panel indicates the representative images of Western blot from cancer tissue (C1–C3) and adjacent normal tissues (N1–N3), and the lower panel shows the quantitation of protein expression (n=5). The normalized expression of PIEZO1 and PIEZO2 is displayed as mean ± S.D. *P<0.05.
Figure 2. Alteration frequency of PIEZO1 and 2 in NSCLC.
The alteration of PIEZO1 and 2 were visualized using the cBioPortal for Cancer Genomics database. Mutation, deletion, and amplification are shown in different colors. (A) Alteration frequency of PIEZO1 in NSCLC. (B) Alteration frequency of PIEZO2 in NSCLC.
Association between the expression of PIEZO channels and clinicopathological characteristics of NSCLC patients
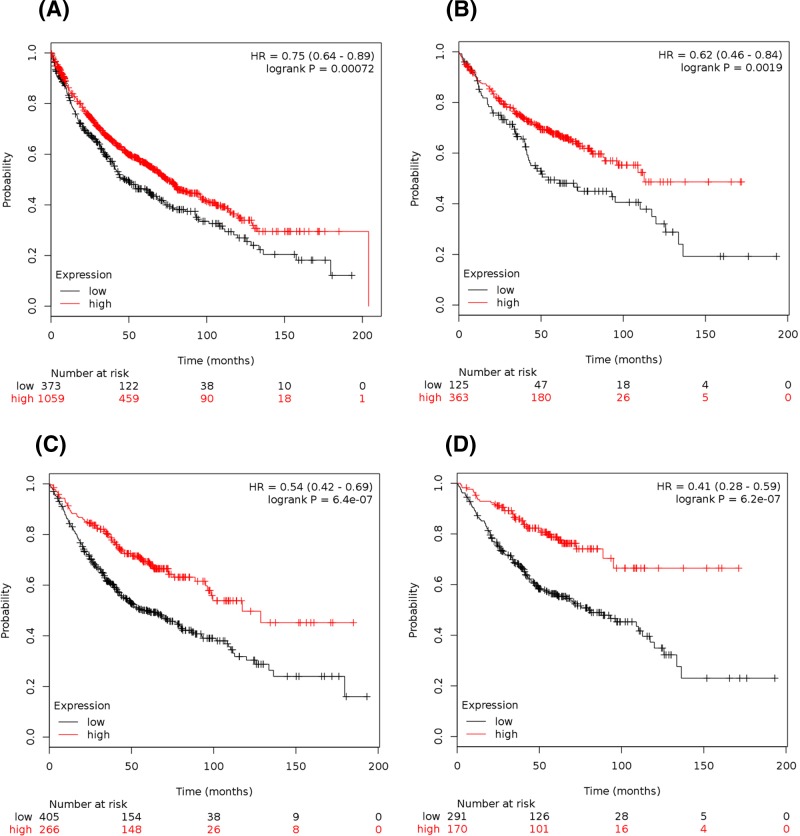
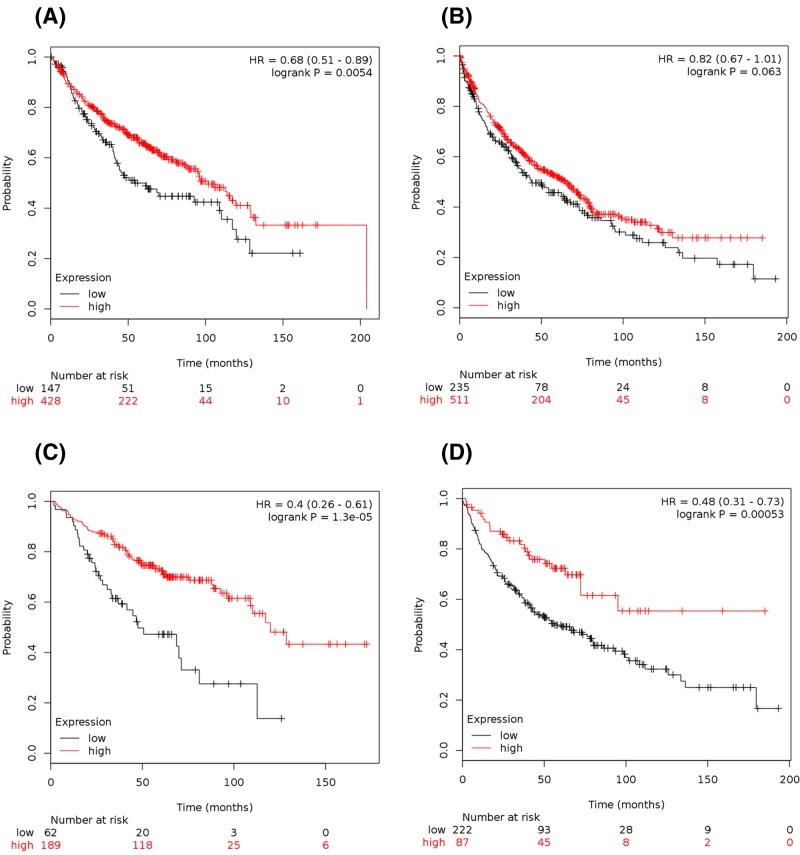
We used the KM plotter to determine the prognostic value of PIEZOs in the database. The Affymetrix IDs is valid: 202771_at (PIEZO1) and 222908_at (PIEZO2). Survival curves were drafted for PIEZO1 in all NSCLC patients (n=1432) (Figure 3A), LUAD patients (n=488) (Figure 3B), and LUSC patients (n=421) (data not shown). The high expression of PIEZO1 mRNA was correlated to better OS for all NSCLC patients, HR 0.79 (0.64–0.89), P=0.00072 (Figure 3A). In particular, the high expression of PIEZO1 mRNA was strongly correlated to better OS in LUAD patients, HR 0.62 (0.46–0.84), P=0.0019 (Figure 3B), but not in LUSC patients, HR 0.86 (0.65–1.12), P=0.26 (data not shown). The high expression of PIEZO1 mRNA was also strongly correlated to better OS in female patients, HR 0.68 (0.51–0.89), P=0.0054 (Figure 4A), but not in male patients, HR 0.82 (0.67–1.01), P=0.063 (Figure 4B).
Figure 3. Prognostic value of the expression of PIEZOs in NSCLC.
(A,B) Prognostic value of PIEZO1 expression in NSCLC patients (A, n=1432) and LUAD patients (B, n=488). (C,D) Prognostic value of PIEZO2 expression in NSCLC patients (C, n=671) and LUAD patients (D, n=461).
Figure 4. Prognostic value of the mRNA expression of PIEZOs in female and male patients.
(A,B) Prognostic value of PIEZO1 mRNA expression in female patients (A, n=575) and male patients (B, n=746). (C,D) Prognostic value of PIEZO2 mRNA expression in female patients (C, n=251) and male patients (D, n=309).
For PIEZO2, the survival curves were drafted in all NSCLC patients (n=671) (Figure 3C), in LUAD patients (n=461) (Figure 3D), and in LUSC patients (n=168) (data not shown). The high expression of PIEZO2 mRNA was correlated to better OS for all NSCLC patients, HR 0.54 (0.42–0.69), P=6.4 x 10-7 (Figure 3C). Interestingly, the high expression of PIEZO2 mRNA was strongly correlated to better OS in LUAD patients, HR 0.41 (0.28–0.59), P=6.2e-7 (Figure 3D), but not in LUSC patients, HR 1.39 (0.88–2.2), P=0.15 (data not shown). The high expression of PIEZO2 mRNA was strongly correlated to better OS in female patients, HR 0.4 (0.26–0.61), P=1.3 x 10-5 (Figure 4C), and in male patients, HR 0.48 (0.31–0.73), P=0.00053 (Figure 4D).
To further assess the association of PIEZOs with other clinicopathological profiles, we determined the correlation with the smoking status of the patients, different clinical stages, and surgical treatments (Table 1). As shown in Table 1, the high expression of PIEZO1 and 2 was strongly correlated with better OS in non-smoking patients, but not in patients who smoked. From Table 1, the low mRNA expression of PIEZO1 was associated with worse OS of patients with grades I and II, but not with grade III. The low mRNA expression of PIEZO2 was associated with worse OS of patients with grades I, II, and III. From Table 1, the low mRNA expression of PIEZO2, but not PIEZO1, was associated with worse OS in patients with negative surgical margins.
Table 1. Correlation of gene expression with NSCLC patients in different clinical variables.
| Clinical variables | Genes | Cases | HR | 95% CI | P-value | |
|---|---|---|---|---|---|---|
| Smoking status | Never smoked | PIEZO1 | 178 | 0.5 | 0.25–0.971 | 0.037 |
| Smoked | PIEZO1 | 669 | 1.14 | 0.9–1.46 | 0.28 | |
| Never smoked | PIEZO2 | 117 | 0.37 | 0.11–1.32 | 0.11 | |
| Smoked | PIEZO2 | 164 | 0.48 | 0.24–0.95 | 0.032 | |
| Clinical stage | I | PIEZO1 | 440 | 0.54 | 0.38–0.77 | 0.00046 |
| II | PIEZO1 | 185 | 0.51 | 0.31–0.84 | 0.0067 | |
| III | PIEZO1 | 67 | 1.42 | 0.72–2.811 | 0.31 | |
| I | PIEZO2 | 322 | 0.38 | 0.25–0.57 | 1.1 x 10-6 | |
| II | PIEZO2 | 107 | 0.56 | 0.32–1 | 0.045 | |
| III | PIEZO2 | 44 | 2.52 | 1.1–5.78 | 0.024 | |
| Negative surgical margins | PIEZO1 | 726 | 1.14 | 0.89–1.45 | 0.29 | |
| PIEZO2 | 204 | 0.28 | 0.13–0.62 | 0.00079 | ||
Knockdown of PIEZO genes promotes cell migration and tumor growth
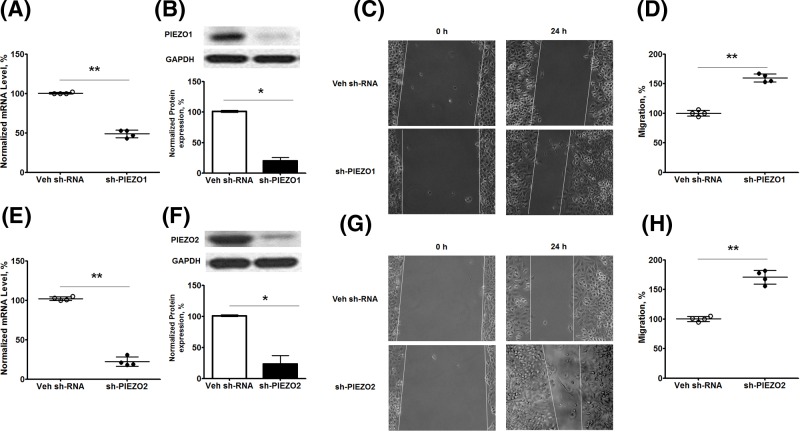
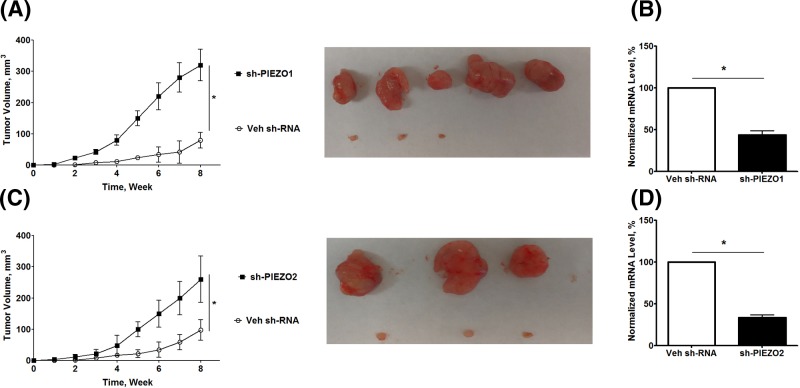
To investigate the potential mechanism of PIEZO in the development of NSCLC, we further studied the effects of PIEZO channels in NSCLC cell migration. As shown in Figure 5A,B,E, F, stable transfection of sh-PIEZO1 (Figure 5A,B) or sh-PIEZO2 (Figure 5E,F) significantly reduced the mRNA and protein expression of the target gene in A549 cells. In comparison with cells with transfection of Veh sh-RNA, cells with stable knockdown of PIEZO1 (Figure 5C,D) or PIEZO2 (Figure 5G,H) promoted cell migration in A549 cells. To check the role of PIEZOs in tumor growth, we inoculated A549 cells with stable transfection of Veh sh-RNA, sh-PIEZO1, or sh-PIEZO2 into nude mice. As shown in Figure 6A, the tumors derived after inoculation with sh-PIEZO1-transfected A549 cells grew faster and larger than the mice inoculated with Veh sh-RNA transfected A549 cells in vivo (Figure 6A). The qPCR data indicate that the xenografts from mice inoculated with A549 cells with stable knockdown of PIEZO1 showed a greatly decreased expression of human PIEZO1 in comparison with those xenografts grown from A549 cells with stable transfection of Veh sh-RNA (Figure 6B). Similarly, inoculation of A549 cells with stable transfection of sh-PIEZO2 also promoted tumor growth in nude mice (Figure 6C), and these xenografts showed less expression of human PIEZO2 (Figure 6D) in comparison with mice inoculated with control cells. These data indicate that the lower expression of PIEZOs resulted in worse OS in NSCLC patients, at least partly, through promoting cancer cell migration and tumor growth.
Figure 5. Knockdown of PIEZO gene expression promotes cell migration in A549 cells.
A549 cells were transfected with Veh shRNA and shRNA target PIEZO1 or PIEZO2, and the stable cell line was selected and maintained in culture medium with puromycin as described in the ‘Materials and methods’ section. The mRNA expression of PIEZO1 and 2 in these cells was analyzed using RT-qPCR, and the protein expression of GAPDH, PIEZO1, and 2 in these cells was analyzed using Western blot. Cell migration in A549 cells with and without knockdown of PIEZO genes was analyzed by scratch assay as described in the ‘Materials and methods’ section. (A,E) mRNA expression of PIEZO1 (A) and PIEZO2 (E) in A549 cells. (B,F) Protein expression PIEZO1 (B) and PIEZO2 (F) in A549 cells; upper panel indicates the representative images of Western blot, and the lower panel shows the quantitation of protein expression. (C,D) Cell migration in A549 cells with stable knockdown of PIEZO1. Representative images (C) and quantitation of migration (D) in A549 cells with stable knockdown of PIEZO1. (G,H) Cell migration in A549 cells with stable knockdown of PIEZO2. Representative images (G) and quantitation of migration (H) in A549 cells with stable knockdown of PIEZO2. Data are displayed as mean ± S.D. of gene expression in different cells. *, P<0.05; **, P<0.01, cells with stable knockdown of PIEZO1 or 2 compared with cells with stable transfection of Veh shRNA.;
Figure 6. Knockdown of the expression of PIEZOs promotes tumor growth in vivo.
Nude mice were subcutaneously inoculated with A549 cells stably transfected with sh-PIEZO1 (n=8), sh-PIEZO2 (n=8), or Veh shRNA (n=8). The subcutaneous tumor size was recorded every week. Data are presented as mean ± S.D. of the measurement of each group. The subcutaneous tumors were isolated and measured. After inoculation, the tumor volumes were measured every week. *P<0.05. (A) Tumor growth in nude mice inoculated with A549 cells with stable knockdown of PIEZO1. Right panel shows the xenografts from mice with subcutaneous inoculation of A549 cells with or without knockdown of PIEZO1. sh-PIEZO1 (n=5) and Veh shRNA (n=3). (B) qPCR quantitation of mRNA expression of human PIEZO1 from the xenografts with or without stable knockdown of PIEZO1. (C) Tumor growth in nude mice inoculated with A549 cells with stable knockdown of PIEZO2. Right panel shows the xenografts from mice with subcutaneous inoculation of A549 cells with or without knockdown of PIEZO2. sh-PIEZO1 (n=3) and Veh shRNA (n=3). (D) qPCR quantitation of mRNA expression of human PIEZO2 from the xenografts with or with stable knockdown of PIEZO2. *, P<0.05, cells with stable knockdown of PIEZO1 or 2 compared with cells with stable transfection of Veh shRNA.
Discussion
PIEZO channels, including PIEZO1 and PIEZO2, represent a new class of mechanosensitive channels, which respond to mechanical forces and allow Ca2+ to enter or exit the cell. PIEZO channels are large transmembrane proteins with more than 2500 amino acids and 24–36 putative transmembrane segments [31]. PIEZO channels are widely expressed in human tissues. Recent investigations using knockout mice have indicated the physiological importance of PIEZO channels in various mechanotransduction processes, such as touch, proprioception, hearing, and blood pressure regulation in mammals. In particular, in endothelial cells, PIEZO1 plays an essential role in shear stress-sensing caused by blood flow, which is important for proper blood vessel development [32]. In epithelial cells, PIEZO1 regulates cell crowding and division [14]. PIEZO1 also regulates blood pressure and exercise performance [15], and determines neural stem cell lineage [13]. PIEZO2 is important in touch sensation [21,33], mainly expressed in primary sensory neurones [34]. In the airway, PIEZO2 localized in sensory neurones is critical for the airway stretch sensation [19]. Furthermore, mutations in human PIEZO1 and PIEZO2 genes have been linked to various genetic diseases due to alterations in channel properties [22].
In many organs, mechanical cues are converted into biological signals through the detection of mechanotransduction via mechanosensitive channels, such as PIEZO1 and 2. Mechanosensitive channels are essential to mechanotransduction and are frequently located on the cell surface. Accumulated research indicates that the regulation of mechanosensitive adaptors is associated with the pathogenesis of various diseases in many organs. In the midgut of adult Drosophila, mechanical stress regulates stem cell differentiation through the stretch-activated ion channel PIEZO, which increases cytosolic Ca2+ in response to a direct mechanical stimulus [35]. In fibrotic tissues, mechanical signaling acts through the discoidin domain receptor 1 to regulate the interactions of cells with the extracellular matrix (ECM) [36]. The composition and physical property changes of ECM are associated with tumor progression in many organs including the lung [37]. Recently, a number of physiological functions of PIEZO channels have been identified through various biological researches. PIEZOs were proved to sense the stiffness of the surrounding substrate and respond to light touch in a number of different cells [38]. Stiffness plays a key role in regulating the matricellular protein CCN1/CYR61 in endothelial cells during tumor metastasis, suggesting that target stiffness‐induced changes is a potential mechanism to impair tumor metastasis [39]. The matrix stiffness of cancer tissue also affects the phenotypes and properties of many types of cancer cells. In LC cells, a stiff substrate was shown to enhance programmed death-ligand 1 (PD-L1) expression and regulate tumor growth [40]. Human PIEZO1 gene mutations resulted in anemia and generalized lymphatic dysplasia [16,17], and PIEZO2 gene mutations were proved to cause distal arthrogryposis and other diseases [22,23]. A recent study has shown that PIEZO1 regulates epithelial restitution and cell mobility in gastric cancer cells through interaction with trefoil factor family 1 (TFF1), a member of the TFF-domain peptide family [41]; and knockdown of PIEZO1 expression reduces cell migration in gastric cancer cell lines [42]. In small cell LC (SCLC) cell lines, the expression of PIEZO1 was lower in the SCLC cell lines in comparison with normal control cells [18]. Knockdown of the expression of PIEZO1 in SCLC cells, promoted cell migration and colony numbers in soft agar [18]. Similarly, in our study, we also found that the expression of PIEZO1 and PIEZO2, in the lung tissue from NSCLC patients, and knockdown of the expression of PIEZO1 or PIEZO2 increased cell migration in A549 cells (NSCLC cell line) in the 2D culture system.
PIEZO1 and 2 are highly expressed in lung tissue. Knockdown of PIEZO1 in lung epithelial cells attenuated cell adherence and stimulated cell migration [18]. In particular, how lung mechanotransduction regulates lung tumor progression remains elusive. We investigated the expression and prognostic roles of PIEZO channels in NSCLC patients. Our data indicate that the expression of PIEZOs decreased in lung tumor tissues, which may result in the impaired function of PIEZO channels in LC patients [6]. Similar to LC, the expression of PIEZO1 is also down-regulated in radiation-induced thyroid tumors [43]. The potential molecular mechanisms leading to the decreased expression of PIEZOs were different. For PIEZO1, gene expression shows high frequency of deep deletion in human LC tissue. However, PIEZO2 expression shows a high mutation ratio in cancer tissue from NSCLC patients. The predicted roles of PIEZOs in NSCLC patients, both PIEZO1 and 2, are those involved in the suppression of tumor progression. The higher expression of PIEZO1 and PIEZO2 was correlated to better OS for all NSCLC patients. Indeed, we provide compelling evidence that PIEZO1 and 2 have critical prognostic values in NSCLC. Meanwhile, Yoda1, an activator for activation or sensitization of membrane tension, and non-specific blockers of PIEZOs (such as gadolinium, ruthenium red, and streptomycin), have been reported to regulate PIEZO functions [44], which might be useful in understanding the heterogeneity and complexity of the molecular biological role of PIEZOs in the pathogenesis of NSCLC and other diseases. Our current study indicated that blocking the function of PIEZOs or knockout of the expression of PIEZOs promotes tumor formation, which may suggest that activators targetting PIEZOs would be a potential candidate for the treatment of NSCLC.
In summary, our investigation demonstrates that the expression of PIEZOs decreased in NSCLC tissues, which may be due to the alteration of gene expression. High mRNA expression of PIEZO channels was found to correlate with better OS for all NSCLC patients, especially for LUAD patients, which may be due to the regulation of cell migration and tumor growth. Our further investigation of the prognostic roles of PIEZO channels in different clinicopathological features indicated that there are critical prognostic values of PIEZO1 and 2 channels in NSCLC. This information suggests that PIEZO1 and 2 might be a potential drug target for NSCLC patients, and the expression of these genes would be useful to develop novel tools to effectively predict the prognosis of NSCLC.
Abbreviations
- DMEM
Dulbecco’s modified Eagle’s medium
- ECM
extracellular matrix
- EGFR
epidermal growth factor receptor
- GAPDH
glyceraldehyde-3-phosphate dehydrogenase
- GEO
Gene Expression Omnibus
- HR
hazard ratio
- KM plotter
Kaplan–Meier plotter
- LC
lung cancer
- LUAD
lung adenocarcinoma
- LUSC
lung squamous cell carcinoma
- NSCLC
non-small cell LC
- OS
overall survival
- RT-qPCR
real-time quantitative PCR
- SCLC
small cell LC
- TBST
Tris-buffered saline with 0.1% Tween 20
- TCGA
The Cancer Genome Atlas
Author contribution
Z.H., Z.S., H.L., and Y.L. designed the experiments, collected the data, wrote and revised the manuscript. Z.H., Z.S., X.Z., and K.N. contributed to the in vitro experiments and data analysis including cell culture, Western blot, and qPCR. Z.H., Z.S., Y.W., and J.Z. contributed to the in vivo animal experiments.
Funding
This work was supported by the China National Natural Science Foundation [grant number NSFC-81400047]; the Science and Technology Department of Jiangsu Province [grant number BK20150213]; and Grant from Health Commission of Jilin Provinvce [grant number 2018J026]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing interests
The authors declare that there are no competing interests associated with the manuscript.
References
- 1.Keith R.L. and Miller Y.E. (2013) Lung cancer chemoprevention: current status and future prospects. Nat. Rev. Clin. Oncol. 10, 334–343 10.1038/nrclinonc.2013.64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Field R.W. and Withers B.L. (2012) Occupational and environmental causes of lung cancer. Clin. Chest Med. 33, 681–703 10.1016/j.ccm.2012.07.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Marciniak S.J. (2017) Endoplasmic reticulum stress in lung disease. Eur. Respir. Rev. 26, 10.1183/16000617.0018-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pappu P., Madduru D., Chandrasekharan M., Modhukur V., Nallapeta S. and Suravajhala P. (2016) Next generation sequencing analysis of lung cancer datasets: a functional genomics perspective. Indian J. Cancer 53, 1–7 10.4103/0019-509X.180832 [DOI] [PubMed] [Google Scholar]
- 5.Liu Y. (2018) Small cell lung cancer transformation from EGFR-mutated lung adenocarcinoma: a case report and literatures review. Cancer Biol. Ther. 19 445–449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bagriantsev S.N., Gracheva E.O. and Gallagher P.G. (2014) Piezo proteins: regulators of mechanosensation and other cellular processes. J. Biol. Chem. 289, 31673–31681 10.1074/jbc.R114.612697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coste B., Mathur J., Schmidt M., Earley T.J., Ranade S., Petrus M.J.. et al. (2010) Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 330, 55–60 10.1126/science.1193270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Delmas P. and Coste B. (2013) Mechano-gated ion channels in sensory systems. Cell 155, 278–284 10.1016/j.cell.2013.09.026 [DOI] [PubMed] [Google Scholar]
- 9.Nilius B. (2010) Pressing and squeezing with Piezos. EMBO Rep. 11, 902–903 10.1038/embor.2010.181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Copp S.W., Kim J.S., Ruiz-Velasco V. and Kaufman M.P. (2016) The mechano-gated channel inhibitor GsMTx4 reduces the exercise pressor reflex in decerebrate rats. J. Physiol. 594, 641–655 10.1113/JP271714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li J. et al. (2014) Piezo1 integration of vascular architecture with physiological force. Nature 515, 279–282 10.1038/nature13701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ranade S.S., Syeda R. and Patapoutian A. (2015) Mechanically activated ion channels. Neuron 87, 1162–1179 10.1016/j.neuron.2015.08.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pathak M.M., Nourse J.L., Tran T., Hwe J., Arulmoli J., Le D.T.. et al. (2014) Stretch-activated ion channel Piezo1 directs lineage choice in human neural stem cells. Proc. Natl. Acad. Sci. U.S.A. 111, 16148–16153 10.1073/pnas.1409802111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gudipaty S.A., Lindblom J., Loftus P.D., Redd M.J., Edes K., Davey C.F.. et al. (2017) Mechanical stretch triggers rapid epithelial cell division through Piezo1. Nature 543, 118–121 10.1038/nature21407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rode B. et al. (2017) Piezo1 channels sense whole body physical activity to reset cardiovascular homeostasis and enhance performance. Nat. Commun. 8, 350 10.1038/s41467-017-00429-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fotiou E. et al. (2015) Novel mutations in PIEZO1 cause an autosomal recessive generalized lymphatic dysplasia with non-immune hydrops fetalis. Nat. Commun. 6, 8085 10.1038/ncomms9085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zarychanski R., Schulz V.P., Houston B.L., Maksimova Y., Houston D.S., Smith B.. et al. (2012) Mutations in the mechanotransduction protein PIEZO1 are associated with hereditary xerocytosis. Blood 120, 1908–1915 10.1182/blood-2012-04-422253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McHugh B.J., Murdoch A., Haslett C. and Sethi T. (2012) Loss of the integrin-activating transmembrane protein Fam38A (Piezo1) promotes a switch to a reduced integrin-dependent mode of cell migration. PLoS ONE 7, e40346 10.1371/journal.pone.0040346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nonomura K., Woo S.H., Chang R.B., Gillich A., Qiu Z., Francisco A.G.. et al. (2017) Piezo2 senses airway stretch and mediates lung inflation-induced apnoea. Nature 541, 176–181 10.1038/nature20793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rezaei N. and Saghazadeh A. (2017) The role of PIEZO2 in human mechanosensation. Acta Med. Iran. 55, 664 [PubMed] [Google Scholar]
- 21.Woo S.H. et al. (2014) Piezo2 is required for Merkel-cell mechanotransduction. Nature 509, 622–626 10.1038/nature13251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alper S.L. (2017) Genetic diseases of PIEZO1 and PIEZO2 dysfunction. Curr. Top. Membr. 79, 97–134 10.1016/bs.ctm.2017.01.001 [DOI] [PubMed] [Google Scholar]
- 23.Coste B. et al. (2013) Gain-of-function mutations in the mechanically activated ion channel PIEZO2 cause a subtype of Distal Arthrogryposis. Proc. Natl. Acad. Sci. U.S.A. 110, 4667–4672 10.1073/pnas.1221400110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Beitzinger M., Hofmann L., Oswald C., Beinoraviciute-Kellner R., Sauer M., Griesmann H.. et al. (2008) p73 poses a barrier to malignant transformation by limiting anchorage-independent growth. EMBO J. 27, 792–803 10.1038/emboj.2008.13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cerami E. et al. (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2, 401–404 10.1158/2159-8290.CD-12-0095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Huang Z., Yang C., Sun S., Nan Y., Lang Z., Wang X.. et al. (2017) Heat Shock Protein 27, a novel regulator of transforming growth factor beta induced resistance to cisplatin in A549 cell. Pharmacology 100, 283–291 10.1159/000479320 [DOI] [PubMed] [Google Scholar]
- 27.Gyorffy B., Surowiak P., Budczies J. and Lanczky A. (2013) Online survival analysis software to assess the prognostic value of biomarkers using transcriptomic data in non-small-cell lung cancer. PLoS ONE 8, e82241 10.1371/journal.pone.0082241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang L.S. et al. (2014) The mitochondrial cardiolipin remodeling enzyme lysocardiolipin acyltransferase is a novel target in pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 189, 1402–1415 10.1164/rccm.201310-1917OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen M., Hu C., Guo Y., Jiang R., Jiang H., Zhou Y.. et al. (2018) Ophiopogonin B suppresses the metastasis and angiogenesis of A549 cells in vitro and in vivo by inhibiting the EphA2/Akt signaling pathway. Oncol. Rep. 40, 1339–1347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yue P.Y., Leung E.P., Mak N.K. and Wong R.N. (2010) A simplified method for quantifying cell migration/wound healing in 96-well plates. J. Biomol. Screen. 15, 427–433 10.1177/1087057110361772 [DOI] [PubMed] [Google Scholar]
- 31.Xu X.Z. (2016) Demystifying mechanosensitive Piezo ion channels. Neurosci. Bull. 32, 307–309 10.1007/s12264-016-0033-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Beech D.J. (2018) Endothelial Piezo1 channels as sensors of exercise. J. Physiol. 596, 979–984 10.1113/JP274396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chesler A.T. et al. (2016) The role of PIEZO2 in human mechanosensation. N. Engl. J. Med. 375, 1355–1364 10.1056/NEJMoa1602812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ranade S.S. et al. (2014) Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature 516, 121–125 10.1038/nature13980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.He L., Si G., Huang J., Samuel A.D.T. and Perrimon N. (2018) Mechanical regulation of stem-cell differentiation by the stretch-activated Piezo channel. Nature 555, 103–106 10.1038/nature25744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Coelho N.M. and McCulloch C.A. (2018) Mechanical signaling through the discoidin domain receptor 1 plays a central role in tissue fibrosis. Cell Adh. Migr. 12, 348–362 10.1080/19336918.2018.1448353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cox T.R. and Erler J.T. (2011) Remodeling and homeostasis of the extracellular matrix: implications for fibrotic diseases and cancer. Dis. Model Mech. 4, 165–178 10.1242/dmm.004077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gottlieb P.A. (2017) A tour de force: the discovery, properties, and function of Piezo channels. Curr. Top. Membr. 79, 1–36 10.1016/bs.ctm.2016.11.007 [DOI] [PubMed] [Google Scholar]
- 39.Reid S.E. et al. (2017) Tumor matrix stiffness promotes metastatic cancer cell interaction with the endothelium. EMBO J. 36, 2373–2389 10.15252/embj.201694912 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Miyazawa A., Ito S., Asano S., Tanaka I., Sato M., Kondo M.. et al. (2018) Regulation of PD-L1 expression by matrix stiffness in lung cancer cells. Biochem. Biophys. Res. Commun. 495, 2344–2349 10.1016/j.bbrc.2017.12.115 [DOI] [PubMed] [Google Scholar]
- 41.Yang X.N., Lu Y.P., Liu J.J., Huang J.K., Liu Y.P., Xiao C.X.. et al. (2014) Piezo1 is as a novel trefoil factor family 1 binding protein that promotes gastric cancer cell mobility in vitro. Dig. Dis. Sci. 59, 1428–1435 10.1007/s10620-014-3044-3 [DOI] [PubMed] [Google Scholar]
- 42.Li C. et al. (2015) Piezo1 forms mechanosensitive ion channels in the human MCF-7 breast cancer cell line. Sci. Rep. 5, 8364 10.1038/srep08364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ory C. et al. (2011) Gene expression signature discriminates sporadic from post-radiotherapy-induced thyroid tumors. Endocr. Relat. Cancer 18, 193–206 10.1677/ERC-10-0205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Syeda R. et al. (2015) Chemical activation of the mechanotransduction channel Piezo1. Elife 4, 10.7554/eLife.07369 [DOI] [PMC free article] [PubMed] [Google Scholar]