Abstract
OBJECTIVE:
To conduct a pooled analysis of three published trials of non-pharmacological interventions for menopausal hot flashes in order to compare the effectiveness of interventions.
METHODS:
Data from three randomized controlled trials of interventions for hot flashes (two acupuncture trials, one yoga trial) were pooled. All three studies recruited perimenopausal or postmenopausal women experiencing ≥4 hot flashes per day on average. The primary outcome for all three studies was frequency of hot flashes as measured by the Daily Diary of Hot Flashes. Study 1 participants were randomly assigned to 8 weeks of acupuncture treatments (active intervention), sham acupuncture (attention control), or usual care. Study 2 participants were randomly assigned to 10 weeks of yoga classes, health and wellness education classes (attention control), or waitlist control. Study 3 randomly assigned participants to 6 months of acupuncture or waitlist control. To standardize the time frame for these analyses, only the first 8 weeks of intervention from all three studies were used.
RESULTS:
The three active interventions and the two attention control groups had statistically similar trends in the percentage reduction of hot flashes over 8 weeks, ranging from 31% to 41%. These five groups did not differ significantly from each other, but all showed significantly greater reduction in hot flash frequency compared to the three usual care/waitlist groups.
CONCLUSION:
Acupuncture, yoga, and health and wellness education classes all demonstrated statistically similar effectiveness in reduction of hot flash frequency compared to controls.
Keywords: menopause, acupuncture, vasomotor symptoms, hot flashes, yoga, control groups
Vasomotor symptoms (VMS), which include hot flashes and night sweats, are the most common and troubling symptoms associated with menopause.1, 2 Although some women report experiencing the menopausal transition without any VMS,3 others experience frequent and severe symptoms that interfere with daily activities and quality of life.4–8 Recent estimates suggest that frequent menopausal VMS can continue for a median of 7.4 years and may last at least 14 years.9 VMS are the chief menopause-related problems for which US women seek medical attention.10, 11
Estrogen therapy, alone or in combination with progesterone, is currently the most effective treatment of VMS. Hormone therapy (HT), however, is associated with a number of risks12–16 and many women seek alternatives such as other pharmaceutical agents, herbal or dietary remedies, or behavioral therapies.17–20 Unfortunately, many of these agents also have side effects and/or have not been shown to be effective.19, 21–24
We have conducted three separate randomized trials of non-pharmacological interventions for the treatment of menopausal VMS.25–27 All three studies recruited peri and postmenopausal women experiencing ≥ 4 hot flashes per day. The primary outcome for all three studies was the percent reduction in hot flashes as recorded on daily dairies. Study 1 found that women randomized to 8 weeks of either true or sham acupuncture had a decrease of 40% from baseline in mean frequency of hot flashes.25 Both the true and sham acupuncture groups were significantly different from usual care, but not from each other. Study 2 compared 10 weeks of yoga with an attention control group of health and wellness education classes (HWE), and a waitlist control group. There was a significant decline in hot flash frequency for all groups, but the yoga and HWE groups followed similar patterns of 66% and 63% reduction of hot flashes from baseline and had a significantly greater decrease than the waitlist group.26 Study 3 was an effectiveness trial of acupuncture compared with a waitlist control where women randomized to acupuncture could receive up to 20 treatments within 6 months.27 At 8 weeks, the acupuncture group had a 33.9% reduction in VMS frequency compared to a 3.3% reduction in the waitlist control group.27
Although results of these studies have previously been published, we were interested in statistically comparing the effectiveness of the different interventions (including active controls) with each other. Given that all three studies used the same outcome measure and had similar eligibility criteria, we are able to combine these data into a single dataset to statistically compare the effectiveness of the five interventions and to control for any differences in sample characteristics. As suggested in a recent Editorial,28 such pooled analyses provide the opportunity to compare effects of different interventions. The objective of the present paper is to present findings from this pooled analysis comparing the reduction in VMS frequency among the three active interventions (two acupuncture groups, yoga), two attention control groups (sham acupuncture, HWE), and three passive controls groups (usual care and waitlist). The sham acupuncture and HWE classes will hereafter be referred to as attention control groups, and the waitlist and usual care groups will be referred to as passive control groups.
METHODS
Descriptions of all three studies have been previously published and are summarized in Table 1. For all studies, women were recruited through newspaper advertisements, radio announcements, and hospital postings. Initial eligibility was determined by telephone screener. Potentially eligible women were scheduled for an initial clinic visit where they completed self-administered baseline questionnaires and received instructions on keeping a 2-week hot flash diary, which they mailed back to verify eligibility. For all studies, women were required to report an average of ≥4 hot flashes per day to be eligible. All studies were approved by the Institutional Review Boards at all sites, and all participants provided written informed consent.
TABLE 1.
Trial designs
| Trial | Total Enroll ment |
Design | Eligibility | VMS Eligibility | Exclusion Criteria | Intervention Length |
|---|---|---|---|---|---|---|
| 1 Avis25 |
56 | 3-arms: acupuncture, sham acupuncture (attention control), usual care (passive control) |
Age 42–55 Perimenopausal or postmenopausal |
≥4 VMS/day | Use of hormone therapy, a selective estrogen receptor modulator, aromatase inhibitor, clonidine, Bellergal, antidepressant, or gabapentin (past 12 wks) Had received chemotherapy Significant psychiatric disorder Used acupuncture for any reason in past 4 wks Previous treatment for acupuncture for VMS Untreated thyroid disease Bleeding or clotting problem other than heavy periods Currently on prescribed medications that increase the risk of bleeding. |
8 wks. |
| 2 Avis26 |
54 | 3-arms: yoga, health and wellness education classes, (attention control) waitlist (passive control) |
Age 45–58 Late perimenopausal or postmenopausal |
≥4 VMS/day | Hormone therapy (past 12 wks) Hormone-modulating therapies (past 6 mos) History of cancer (past 5 yrs) Prior use of yoga for VMS Yoga or acupuncture (past 3 mos) Untreated thyroid disease Initiation of herbal or nutritional remedies (past 4 wks) Initiation of psychoactive medications (past 3 mos) |
10 wks. |
| 3 Avis27 |
209 | 2-arms: acupuncture, waitlist (passive control) |
Age 45–60 Perimenopausal or postmenopausal |
≥4 VMS/day | Initiation or change in dose of any tx for HF (past 4 wks) Initiation or change in dose of antidepressant (past 3 mos) Received acupuncture for any indication (past 4 wks) Received acupuncture from one of the study acupuncturists (prior 6 mos) Self-reported health status as poor or fair Diagnosis of hemophilia |
8 wks. |
VMS, vasomotor symptoms
The primary outcome in all studies was frequency of hot flashes as measured by the Daily Diary of Hot Flashes (DDHF).29 Baseline VMS frequency was calculated using the last fourteen days of diaries available prior to randomization. Intervention phases differed among the three studies; for the purpose of these analyses, we used data from only the first 8 weeks of intervention for all studies to correspond to the study with the shortest treatment phase.
Study 1: Acupuncture vs. Attention Control (Sham Acupuncture) vs Passive Control (Usual Care)
This study was a 2-site clinical trial using a 3-arm prospective, randomized single-blind, sham-control design and usual care only control (N=56).25 Women who were current or previous hormonal therapy (HT) users were not eligible. Eligible women were randomized to one of the three groups stratified by clinical center. Participants assigned to acupuncture (both true and sham) were seen twice a week for 8 weeks (a total of 16, 30 minute treatment sessions) by a trained acupuncturist. All participants were instructed not to take hormonal medications or initiate other treatments for their hot flashes during the study.
Acupuncture.
This group received needling at sites based on a “standardized individual” approach which combined standardized core acupuncture points supplemented with customized points based on a woman’s presentation.
Attention control group (sham acupuncture).
This group received shallow needling at bodily locations not corresponding to traditional acupuncture points that were thought to have minimal effects on hot flashes.
Passive control group (usual care).
This group did not receive any form of acupuncture needling. One clinical site was able to provide two acupuncture treatments to this group after two months.
Study 2: Yoga vs. Attention Control (Health and Wellness Education) vs. Passive Control (Waitlist)
This 3-arm randomized study consisted of three groups: yoga, health and wellness education (HWE) classes, and a usual care control group (N=54).26 Women who had HT use in the previous three months were not eligible. All study participants agreed not to take hormonal medications or initiate other treatments for their hot flashes during the study.
Yoga.
The study intervention consisted of 10 weekly, 90-minute classes of Integral Yoga which places a major emphasis on stress reduction and decreased likelihood of increasing heat. Each class included the same basic elements: (1) breathing (pranayama)/centering meditation; (2) physical postures (asanas); and (3) deep relaxation (Savasana). In addition to the group classes, participants were asked to practice yoga at home for 15 minutes ≥3 times per week using a videorecording produced for this study.
Attention Control group (HWE).
This control group was designed to control for time, attention, and social aspects of the yoga intervention. It was designed to be “believable” for menopausal women without being efficacious for reducing hot flashes. Classes consisted of 10 weekly, 90-minute didactic classes on different topics: menopause, stress management (with no breathing component) (2 sessions), bone health, cardiovascular health, nutrition, financial planning, skin care, aging and long-term health planning, and cancer prevention/screening. Women were given materials to read at home to match the time spent in home yoga practice.
Passive Control Group (Waitlist).
Participants in this group did not participate in any classes for 10 weeks. They were provided with up to four yoga or HWE classes after 10 weeks.
Study 3: Acupuncture vs. Passive Control (Waitlist)
In this pragmatic 2-site, 2-arm clinical trial, participants were randomized to either a 6-month course of up to 20 acupuncture treatments, or a waitlist group.27 The study did not include a sham acupuncture group as the purpose of the study was to better understand acupuncture as practiced in the real world. This study did not exclude HT users as we were interested in whether current HT use would be related to the effectiveness of acupuncture. However, only two participants were taking HT.
Acupuncture.
Study acupuncturists ascribed a Traditional Chinese Medicine diagnosis to each participant and were allowed to individualize treatment. The frequency of treatments was determined by the acupuncturist and study participant. Study acupuncturists were permitted to change acupuncture point selection and other aspects of treatment if clinically indicated at each treatment session. No restrictions were placed on duration of each treatment, or on the application of other treatment modalities such as moxibustion or manual or electrical stimulation of acupuncture needles at selected acupuncture point locations.
Passive Control Group (Waitlist).
Participants in this group did not receive any form of acupuncture needling during the first 6 months of study participation. After 6 months, they were provided up to 20 acupuncture treatments in the next 6 months.
Statistical Analysis
We computed basic descriptive data on key covariates: age (in years) at study enrollment, menopausal status (postmenopausal vs not postmenopausal), and race (white vs non-white).
Our outcome variable of interest was percentage change in weekly average number of daily hot flashes. For each of the 8 weeks following baseline, we computed an average number of daily hot flashes over the 7 days of that week. We then divided this weekly average by the average number of daily hot flashes reported during the baseline week to get a fractional proportion of baseline hot flashes. We subtracted this fraction from 1.0, and converted to percent to obtain percent reduction in average daily hot flashes from baseline week.
We first sought to determine whether we could combine the three passive control groups across studies into a single reference group based on the rationale that participants in these groups did not receive any type of study intervention. Prior to our main analyses, we statistically confirmed that there were no differences in the three passive control groups in terms of average overall reduction in hot flashes or in the pattern of reduction over the 8 weeks (p=0.98 for main effect of group and 0.60 for group*week interaction, in repeated measures mixed model containing three passive control groups only). We therefore combined these three passive control groups in our two main repeated measures models of interest.
Our first model tested the null hypothesis of no significant differences in level of hot flash reduction among the three active treatments and two attention control groups (collectively referred to as “intervention” groups, below). We compared the five groups in a single model controlling for the covariates of age (in years) at study enrollment, menopausal status, and race. Week was treated as an ordinal variable (taking on the values 1–8) after confirming monotonic trends in average hot flash reductions in all groups over time. We also included an interaction term between intervention group and week.
Our second model sought to determine if there was a significant difference between the five intervention groups and the three passive control groups. After confirming in our first model that there were no significant differences among the five intervention groups, we combined these five groups into one group and compared them to the group containing the three passive control recipients. Similar to the above, we controlled for the covariates of age at study enrollment, menopausal status, and race, and we again considered week as an ordinal variable. We again included the interaction term between group (combined intervention or combined passive control) and week.
All analyses were performed in SAS 9.4 (SAS Institute Inc, Cary, NC).
RESULTS
Participants in the three studies differed significantly (p<0.0001) on mean age at enrollment, with Study 1 having the youngest participants, on average, and Study 3 having the oldest (Table 2). The studies also differed significantly (p=0.008) in racial/ethnic composition: Study 1 had the highest percentage of white participants (85%), while Study 2 had a relatively low percentage of whites (60%). There were no significant differences among the three studies in percent postmenopausal or mean number of daily hot flashes reported at enrollment.
TABLE 2.
Baseline descriptive data from three studies
| Study 1 (n = 56) |
Study 2 (n = 54) |
Study 3 (n = 209) |
p-value | |
|---|---|---|---|---|
| Mean age at baseline (SD) | 51.2 (2.9) | 52.9 (2.9) | 53.8 (3.5) | <0.0001 |
| Mean number of daily hot flashes at baseline (SD) | 9.4 (5.1) | 9.3 (4.7) | 9.5 (5.0) | 0.97 |
| White, No. (%) | 48 (86) | 32 (60) | 157 (75.9) | 0.008 |
| Post-menopausal, No. (%) | 48 (86) | 41 (84) | 172 (83.1) | 0.90 |
SD, standard deviation
Results of the first repeated measures analysis examining differences among the five intervention groups are reported in Table 3 and shown in Figure 1. As seen in Table 3, there were no significant differences among these five groups in either the average level of percent change in number of hot flashes (p-value for group effect = 0.99) or change in number of hot flashes over time (p-value for group*time interaction = 0.95).
TABLE 3.
Results of repeated measures model examining effects of group and week on percent reduction in average number of weekly hot flashes among 5 intervention groups
| Variable | Parameter estimate | SE | p value |
|---|---|---|---|
| Age in years | −0.005 | 0.007 | 0.50 |
| Race | |||
| White | 0.027 | 0.05 | 0.60 |
| Nonwhite | (ref) | ||
| Menopause status | |||
| Perimenopausal | 0.02 | 0.06 | 0.69 |
| Postmenopausal | (ref) | ||
| Week | 0.02 | 0.003 | <0.0001 |
| Group | 0.99 | ||
| Study 1 acupuncture | 0.04 | 0.10 | |
| Study 1 active control (sham) | −0.02 | 0.10 | |
| Study 2 yoga | 0.02 | 0.10 | |
| Study 2 active control (HWE) | −0.03 | 0.10 | |
| Study 3 acupuncture | (ref) | ||
| Group x week interaction | 0.95 | ||
| Study 1 acupuncture x week | −0.003 | 0.01 | |
| Study 1 active control (sham) x week | 0.002 | 0.01 | |
| Study 2 yoga x week | −0.0001 | 0.01 | |
| Study 2 active control (HWE) x week | 0.008 | 0.01 | |
| Study 3 acupuncture x week | (ref) |
Figure 1.
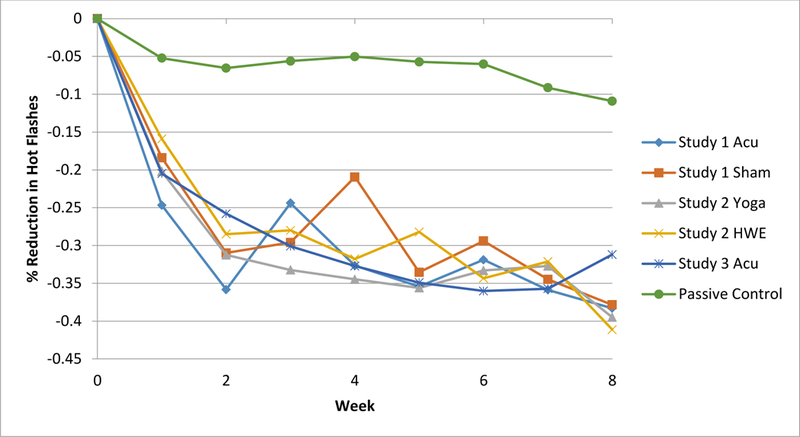
Estimated percent change in VMS frequency by group and weeks from randomization, controlling for baseline VMS frequency, season of randomization, site, prior use of acupuncture, use of hormone therapy, and menopause status.
Our second analysis compared the five intervention groups to the combined passive control group. Given the previous finding that there was no significant difference among the five intervention groups, we combined these groups into one group (“intervention group”) and compared this group to the group comprising the three passive control groups (“control group.”) As shown in Table 4, there was a significant group*time interaction in this model (p=0.02) with the intervention group showing significantly more decline over time than the control group. On average, and after adjustment for the included covariates, the intervention groups declined approximately 28% more by week 8 relative to the average among the control groups. Figure 1 illustrates this interaction using the unadjusted numbers: on average, women in the groups who received active treatment or attention control interventions reported 35–40% fewer hot flashes at the end of the 8 weeks compared to baseline, while those in the combined passive control groups reported close to 10% fewer hot flashes on average at the end of 8 weeks. This (unstandardized) effect size estimate at week 8 of a 28% difference between intervention and control was significantly different from 0 (in a contrast statement) at p<0.0001. As can be seen in Figure 1, lines for the five active groups lie upon each other, and all lie distinctly below the line for the passive control groups.
TABLE 4.
Results of repeated measures model examining effects of combined intervention (vs combined passive group) and week on percent reduction in average number of weekly hot flashes
| Variable | Parameter estimate | SE | p value |
|---|---|---|---|
| Age in years | −0.006 | 0.006 | 0.25 |
| Race | |||
| White | 0.01 | 0.04 | 0.81 |
| Nonwhite | (ref) | ||
| Menopause status | |||
| Perimenopausal | 0.06 | 0.05 | 0.25 |
| Postmenopausal | (ref) | ||
| Week | 0.006 | 0.004 | 0.15 |
| Group | 0.0001 | ||
| Combined intervention | 0.19 | 0.05 | |
| Combined passive control | (ref) | ||
| Week x group interaction | 0.02 | ||
| Combined intervention | 0.01 | 0.005 | |
| Combined passive control x week | (ref) |
DISCUSSION
This pooled analysis of three studies of non-pharmacological interventions for menopausal hot flashes shows that the active treatments (acupuncture and yoga) and attention control intervention groups (sham acupuncture and HWE classes) have statistically similar effects on the percentage reduction of hot flashes over 8 weeks, and that all five intervention groups are significantly better than the passive control groups. Yoga and acupuncture did not differ in their effectiveness in reducing hot flashes, and the two attention control groups did not differ from the two active interventions in effectiveness.
The acupuncture results support previous research showing a significant effect of acupuncture for reducing the frequency of VMS when compared to no treatment.25, 27, 30–35 Results of yoga studies are a bit more mixed, although most of studies do not measure frequency of hot flashes.36 More recently, the Menopause Strategies: Finding Lasting Answers for Symptoms and Health (MsFLASH) trial (a randomized controlled trial comparing yoga, exercise, and omega-3 supplements) found that study participants in the yoga group had a 35% reduction in hot flash frequency,37 which is equivalent to our results in the pooled analysis.
Importantly, this pooled analysis showed that the attention control groups were equally effective as the interventions of interest in reducing hot flash frequency. There are several potential explanations for this finding: 1) the two attention control groups were actually active treatments; 2) the non-specific effects of patient-provider interactions (attention) or other components of the control groups (ie, shallow needling at non-acupuncture points or participation in an educational program) are sufficient to reduce hot flashes; and/or 3) expectations of treatment effectiveness accounted for these results. Sham acupuncture is currently thought to have physiological effects and is not truly a placebo comparator, which would support the first explanation.38 Two reviews of acupuncture for menopausal hot flashes concluded that sham acupuncture could induce a treatment effect comparable to true acupuncture in the reduction of hot flash frequency.31, 35
Although it is somewhat less likely that the HWE classes served as an active intervention since none of these classes addressed management of hot flashes, we cannot rule out this explanation. Results for HWE classes (and sham acupuncture) might also argue that the non-specific effects of the classes and sham acupuncture contributed to the beneficial effects. Other studies have also found that such attention control groups can be as beneficial as the treatment group.39–41 In a systematic review of control condition designs for psychotherapy for depression, Mohr and colleagues found that studies with active control groups had much smaller effect sizes than those that had a usual care or waitlist control,42 suggesting that non-specific effects of such interventions can be effective.
Other studies have found that expectations of treatment effectiveness are important,43–46 but there are two arguments against the role of expectations in our results. First, Study 1 participants did not know if they were getting true or sham acupuncture, but Study 3 participants knew that they were getting true acupuncture. However, results were very similar for both groups. In addition, for both Studies 2 and 3, we asked participants at baseline to rate their expectations that the interventions would reduce the frequency of their hot flashes. Acupuncture received the highest rating (7.9 on a 10 pt. scale), whereas the mean rating for health and wellness was considerably lower (5.9). If expectations explained our findings, we would anticipate these mean expectation ratings to be similar.
Results of this pooled analysis point to the importance of including passive control groups in study designs. Had these groups not been included in our studies, we would not have known if the interventions had any benefit over and above the passage of time, and would have drawn very different conclusions. As discussed by others, the results of randomized controlled trials can depend as much on the selection and justification of the control group(s) as the experimental intervention.42, 47
A limitation of these analyses, which would apply to any pooled analyses, is that the study populations across the three studies are not perfectly matched. Even though we controlled for differences in education, age, and race, there may be unknown differences not accounted for. As previously noted, eligibility criteria around HT use differed among the three trials. However, given that each trial was randomized and that only two women among all of the women in all three trials were on HT at randomization, we believe it is unlikely that these differences in HT eligibility criteria affected results. There were also minor differences in treatment protocols between the two acupuncture studies and the passive controls varied slightly across studies. Further, it is not possible to rule out a general placebo effect such that women in all five intervention groups benefited simply by being in a study, or that longer follow-up would show somewhat different results.
This pooled analysis takes advantage of comparing three separate studies of interventions for menopausal hot flashes that all used the same eligibility criteria and primary outcome measure. Results showing that all interventions were equally effective have clinical significance for advising women bothered by hot flashes and searching for non-hormonal treatments.
CONCLUSION
Our pooled analysis of three randomized controlled trials demonstrates the effectiveness of acupuncture, yoga, and health and wellness education classes in the management of menopausal hot flashes compared with passive controls. Our analyses also provide evidence that attention control interventions may complicate the interpretation of comparisons between presumably active “treatment” interventions and interventions intended to be “inactive” or “inert,” but which still result in improvement in participants. We believe our findings illustrate the importance of including a control arm without any intervention in behavioral intervention trials.
Acknowledgments
Funding Support: Research supported by grants R01AT005854 and R21AT004234 from the National Center for Complementary and Integrative Health, and R21 CA104427 from the National Cancer Institute.
Footnotes
Clinical trials registration: http://www.ClinicalTrials.gov identifiers: NCT01073397 and NCT01276028.
Conflict of interest/financial disclosure: Dr. Coeytaux has a financial interest in two organizations involved in recruiting study participants and administering acupuncture treatments at one of the two study sites. A conflict of interest management plan was developed by Duke University and is available upon request.
References
- 1.Avis NE, Brockwell S, Colvin A. A universal menopausal syndrome? Am J Med 2005;118(12B):375–465. [DOI] [PubMed] [Google Scholar]
- 2.Kronenberg F Hot flashes: Epidemiology and physiology. Ann NY Acad Sci 1990;592:52–86. [DOI] [PubMed] [Google Scholar]
- 3.Avis NE, Crawford SL, McKinlay SM. Psychosocial, behavioral, and health factors related to menopause symptomatology. Womens Health 1997;3(2):103–20. [PubMed] [Google Scholar]
- 4.Avis NE, Ory M, Matthews KA, Schocken M, Bromberger J, Colvin A. Health-related quality of life in a multiethnic sample of middle-aged women: Study of Women’s Health Across the Nation (SWAN). Med Care 2003;41(11):1262–76. [DOI] [PubMed] [Google Scholar]
- 5.Blumel JE, Chedraui P, Baron G, et al. A large multinational study of vasomotor symptom prevalence, duration, and impact on quality of life in middle-aged women. Menopause 2011;18(7):778–85. [DOI] [PubMed] [Google Scholar]
- 6.Hays J, Ockene JK, Brunner RL, et al. Effects of estrogen plus progestin on health-related quality of life. N Engl J Med 2003;348(19):1839–54. [DOI] [PubMed] [Google Scholar]
- 7.Kumari M, Stafford M, Marmot M. The menopausal transition was associated in a prospective study with decreased health functioning in women who report menopausal symptoms. J Clin Epidemiol 2005;58(7):719–27. [DOI] [PubMed] [Google Scholar]
- 8.Williams RE, Levine KB, Kalilani L, Lewis J, Clark RV. Menopause-specific questionnaire assessment in US population-based study shows negative impact on health-related quality of life. Maturitas 2009;62(2):153–9. [DOI] [PubMed] [Google Scholar]
- 9.Avis NE, Crawford SL, Greendale G, et al. Duration of menopausal vasomotor symptoms over the menopause transition. JAMA Intern Med 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nicholson WK, Ellison SA, Grason H, Powe NR. Patterns of ambulatory care use for gynecologic conditions: A national study. Am J Obstet Gynecol 2001;184(4):523–30. [DOI] [PubMed] [Google Scholar]
- 11.Williams RE, Kalilani L, DiBenedetti DB, Zhou X, Fehnel SE, Clark RV. Healthcare seeking and treatment for menopausal symptoms in the United States. Maturitas 2007;58(4):348–58. [DOI] [PubMed] [Google Scholar]
- 12.Barnabei VM, Grady D, Stovall DW, et al. Menopausal symptoms in older women and the effects of treatment with hormone therapy. Obstet Gynecol 2002;100(6):1209–18. [DOI] [PubMed] [Google Scholar]
- 13.Grady D, Herrington D, Bittner V, et al. Cardiovascular disease outcomes during 6.8 years of hormone therapy: Heart and Estrogen/progestin Replacement Study follow-up (HERS II). JAMA 2002;288(1):49–57. [DOI] [PubMed] [Google Scholar]
- 14.Manson JE, Hsia J, Johnson KC, et al. Estrogen plus progestin and the risk of coronary heart disease. New Engl J Med 2003;349(6):523–34. [DOI] [PubMed] [Google Scholar]
- 15.Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA 2002;288(3):321–33. [DOI] [PubMed] [Google Scholar]
- 16.Wassertheil-Smoller S, Hendrix S, Limacher M, et al. Effect of estrogen plus progestin on stroke in postmenopausal women: the Women’s Health Initiative: a randomized trial. JAMA 2003;289(20):2673–84. [DOI] [PubMed] [Google Scholar]
- 17.Beal MW. Women’s use of complementary and alternative therapies in reproductive health care. J Nurse Midwifery 1998;43(3):224–34. [DOI] [PubMed] [Google Scholar]
- 18.Kessel B, Kronenberg F. The role of complementary and alternative medicine in management of menopausal symptoms. Endocrinol Metab Clin North Am 2004;33(4):717–39. [DOI] [PubMed] [Google Scholar]
- 19.Nedrow A, Miller J, Walker M, Nygren P, Huffman LH, Nelson HD. Complementary and alternative therapies for the management of menopause-related symptoms: a systematic evidence review. Arch Intern Med 2006;166(14):1453–65. [DOI] [PubMed] [Google Scholar]
- 20.Newton KM, Buist DS, Keenan NL, Anderson LA, LaCroix AZ. Use of alternative therapies for menopause symptoms: results of a population-based survey. Obstet Gynecol 2002;100(1):18–25. [DOI] [PubMed] [Google Scholar]
- 21.Huntley AL, Ernst E. A systematic review of herbal medicinal products for the treatment of menopausal symptoms. Menopause 2003;10(5):465–76. [DOI] [PubMed] [Google Scholar]
- 22.Kronenberg F, Fugh-Berman A. Complementary and alternative medicine for menopausal symptoms: A review of randomized, controlled trials. Ann Intern Med 2002;137(10):805–13. [DOI] [PubMed] [Google Scholar]
- 23.Loprinzi CL, Kubler JW, Sloan JA, et al. Venlafaxine in management of hot flashes in survivors of breast cancer: a randomized controlled trial. Lancet 2000;356(9247):2059–63. [DOI] [PubMed] [Google Scholar]
- 24.Nagamani M, Kelver ME, Smith ER. Treatment of menopausal hot flashes with transdermal administration of clonidine. Am J Obstet Gynecol 1987;156(3):561–5. [DOI] [PubMed] [Google Scholar]
- 25.Avis NE, Legault C, Coeytaux RR, et al. A randomized, controlled pilot study of acupuncture treatment for menopausal hot flashes. Menopause 2008;15(6):1070–8. [DOI] [PubMed] [Google Scholar]
- 26.Avis NE, Legault C, Russell G, Weaver K, Danhauer SC. Pilot study of integral yoga for menopausal hot flashes. Menopause 2014;21(8):846–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Avis NE, Coeytaux R, Isom S, Prevette K, Morgan T. Acupuncture in menopase (AIM): a pragmatic, randomized controlled trial. Menopause 2016;23(6):626–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Woods NF, Utian W. Quality of life, menopause, and hormone therapy: an update and recommendations for future research. Menopause 2018;25(7):713–20. [DOI] [PubMed] [Google Scholar]
- 29.Sloan JA, Loprinzi CL, Novotny PJ, Barton DL, Lavasseur BI, Windschitl H. Methodologic lessons learned from hot flash studies. J Clin Oncol 2001;19(23):4280–90. [DOI] [PubMed] [Google Scholar]
- 30.Borud EK, Alraek T, White A, et al. The Acupuncture on Hot Flushes Among Menopausal Women (ACUFLASH) study, a randomized controlled trial. Menopause 2009;16(3):484–93. [DOI] [PubMed] [Google Scholar]
- 31.Chiu HY, Pan CH, Shyu YK, Han BC, Tsai PS. Effects of acupuncture on menopause-related symptoms and quality of life in women on natural menopause: a meta-analysis of randomized controlled trials. Menopause 2015;22(2):1. [DOI] [PubMed] [Google Scholar]
- 32.Nedeljkovic M, Tian L, Ji P, et al. Effects of acupuncture and Chinese herbal medicine (Zhi Mu 14) on hot flushes and quality of life in postmenopausal women: results of a four-arm randomized controlled pilot trial. Menopause 2014;21(1):15–24. [DOI] [PubMed] [Google Scholar]
- 33.Painovich JM, Shufelt CL, Azziz R, et al. A pilot randomized, single blind, placebo-controlled trial of traditional acupuncture for vasomotor symptoms and mechanistic pathways of menopause. Menopause 2012;19(1):54–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vincent A, Barton D, Mandrekar J, Cha S, et al. Acupuncture for hot flashes: a randomized sham-controlled clinical study. Menopause 2007;14:45–52. [DOI] [PubMed] [Google Scholar]
- 35.Ee C, French SD, Xue CC, Pirotta M, Teede H. Acupuncture for menopausal hot flashes: clinical evidence update and its relevance to decision making. Menopause 2017;24(8):980–7. [DOI] [PubMed] [Google Scholar]
- 36.Lee MS, Kim JI, Ha JY, Boddy K, Ernst E. Yoga for menopausal symptoms: a systematic review. Menopause 2009;16(3):602–8. [DOI] [PubMed] [Google Scholar]
- 37.Newton KM, Reed SD, Guthrie KA, et al. Efficacy of yoga for vasomotor symptoms: a randomized controlled trial. Menopause 2014;21(4):339–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Birch S Sham acupuncture is not a placebo treatment- implications and problems in research. Japanese Acupuncture and Moxibustion 2012;8(1):4–8. [Google Scholar]
- 39.Jimenez DE, Begley A, Bartels SJ, et al. Improving health-related quality of life in older African American and non-Latino White patients. Am J Geriatr Psychiatry 2015;23(6):548–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kasckow J, Morse J, Begley A, et al. Treatment of post traumatic stress disorder symptoms in emotionally distressed individuals. Psychiatry Res. 2014;220(1–2):370–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Reynolds CF 3rd, Thomas SB, Morse JQ, et al. Early intervention to preempt major depression among older black and white adults. Psychiatric services 2014;65(6):765–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mohr DC, Ho J, Hart TL, et al. Control condition design and implementation features in controlled trials: a meta-analysis of trials evaluating psychotherapy for depression. Transl Behav Med 2014;4(4):407–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chen LX, Mao JJ, Fernandes S, et al. Integrating acupuncture with exercise-based physical therapy for knee osteoarthritis: a randomized controlled trial. J Clin Rheumatol 2013;19(6):308–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Colaguiri B, Smith CA. A systematic review of the effect of expectancy on treatment responses to acupunture. Evid-Based Compl Alt 2012;2012:857804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Keefe JR, Amsterdam J, Li QS, Soeller I, DeRubeis R, Mao JJ. Specific expectancies are associated with symptomatic outcomes and side effect burden in a trial of chamomile extract for generalized anxiety disorder. J Psychiatr Res 2017;84:90–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Linde K, Witt CM, Streng A, et al. The impact of patient expectations on outcomes in four randomized controlled trials of acupuncture in patients with chronic pain. Pain 2007;128(3):264–71. [DOI] [PubMed] [Google Scholar]
- 47.Mohr DC, Spring B, Freedland KE, et al. The selection and design of control conditions for randomized controlled trials of psychological interventions. Psychother Psychosom 2009;78(5):275–84. [DOI] [PubMed] [Google Scholar]


