Abstract
Background/Aims
Nonalcoholic fatty liver disease (NAFLD) is becoming a worldwide epidemic, and is frequently found in patients with chronic hepatitis B (CHB). We investigated the impact of histologically proven hepatic steatosis on the risk for hepatocellular carcinoma (HCC) in CHB patients without excessive alcohol intake.
Methods
Consecutive CHB patients who underwent liver biopsy from January 2007 to December 2015 were included. The association between hepatic steatosis (≥ 5%) and subsequent HCC risk was analyzed. Inverse probability weighting (IPW) using the propensity score was applied to adjust for differences in patient characteristics, including metabolic factors.
Results
Fatty liver was histologically proven in 70 patients (21.8%) among a total of 321 patients. During the median (interquartile range) follow-up of 5.3 (2.9–8.3) years, 17 of 321 patients (5.3%) developed HCC: 8 of 70 patients (11.4%) with fatty liver and 9 of 251 patients (3.6%) without fatty liver. The five-year cumulative incidences of HCC among patients without and with fatty liver were 1.9% and 8.2%, respectively (P=0.004). Coexisting fatty liver was associated with a higher risk for HCC (adjusted hazards ratio [HR], 3.005; 95% confidence interval [CI], 1.122–8.051; P=0.03). After balancing with IPW, HCC incidences were not significantly different between the groups (P=0.19), and the association between fatty liver and HCC was not significant (adjusted HR, 1.709; 95% CI, 0.404–7.228; P=0.47).
Conclusions
Superimposed NAFLD was associated with a higher HCC risk in CHB patients. However, the association between steatosis per se and HCC risk was not evident after adjustment for metabolic factors.
Keywords: Hepatitis B virus, Fatty liver, Nonalcoholic fatty liver disease (NAFLD), Metabolic syndrome, Liver cancer
INTRODUCTION
Chronic hepatitis B virus (HBV) infection is one of the most common chronic viral infections worldwide, and especially in Asian countries [1]. Patients with chronic hepatitis B (CHB) are at risk for developing liver cirrhosis, hepatic decompensation, and hepatocellular carcinoma (HCC) [2-4]. Although the long-term prognosis for patients with chronic HBV infection has been improved by antiviral therapy with potent nucleos(t)ide analogues (i.e., entecavir, tenofovir disoproxil or tenofovir alafenamide), the risk for developing HCC has not been eliminated in those patients, leading to a huge burden on public health [5]. Patients with HBV-related cirrhosis are at a particularly high risk for HCC development, with a yearly incidence of HCC in those patients ranging from 2% to 8% [5].
Nonalcoholic fatty liver disease (NAFLD) is another serious global health problem because of the increasing prevalence even in Asian countries. Although its pathogenesis is not fully understood, NAFLD is primarily associated with obesity and insulin resistance, and is regarded as a hepatic manifestation of metabolic syndrome [6]. A diverse spectrum of liver diseases including nonalcoholic steatohepatitis (NASH) and liver cirrhosis results from NAFLD, and NAFLD is a well-known risk factor for HCC [7]. Patients with NASH-related cirrhosis are at a greatly increased risk for HCC, with a yearly cumulative incidence of 2.6% [8]. Theoretically, NAFLD and chronic HBV infection may synergistically potentiate HCC development; however, few studies have evaluated the effect of coexisting NAFLD on the risk for developing HBV-related HCC. A recent study showed that CHB patients with histologically proven fatty liver had a 7.3–fold increased risk for developing HCC [9]. While age, long-term antiviral treatment, and cirrhosis were independent predictive factors for HCC development, metabolic factors such as diabetes, hypertension, and body mass index (BMI), were not. However, a previous Taiwanese study demonstrated that both concurrent obesity and diabetes were predictors of HCC risk in patients with CHB, suggesting a synergistic hepatocarcinogenic effect of metabolic factors and chronic viral hepatitis [10].
The present study was conducted to evaluate the effect of histologically proven fatty liver on the development of HCC in patients with chronic HBV infection. We adopted inverse probability weighting (IPW) based on each patient’s propensity score to rigorously adjust for possible confounding factors, including metabolic factors.
PATIENTS AND METHODS
Patient population
The study population consisted of consecutive CHB patients who underwent liver biopsy from January 2007 through December 2015 at CHA Bundang Medical Center (Seongnam, Korea). We excluded patients with significant alcohol intake (n=7), which was defined as alcohol consumption of more than 210 g per week in males and 140 g per week in females over a 2-year period preceding liver biopsy [11,12]. Patients who met any of the following criteria also were excluded from the study: (1) history of malignant disease, including HCC (n=11); (2) co-infection with hepatitis C virus (n=1); (3) a follow-up duration of less than 6 months (n=48); or (4) HCC diagnosed within 6 months from baseline (n=1) (Fig. 1). The study protocol was reviewed and approved by the Institutional Review Board of CHA Bundang Medical Center. Because this study was a retrospective cohort study based on pre-existing clinical data, which were analyzed anonymously, the need for obtaining a written informed consent was waived.
Figure 1.
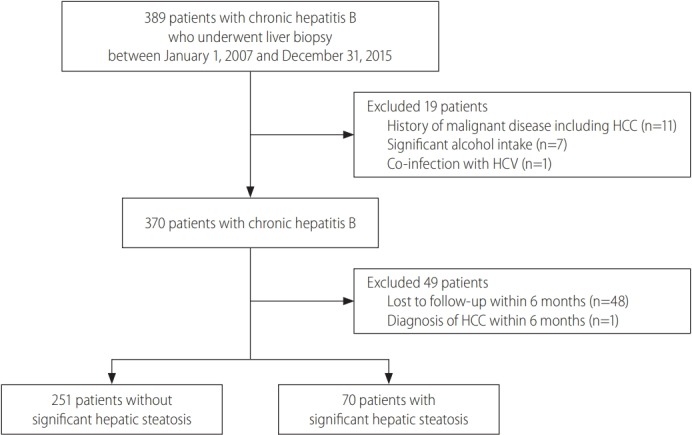
Flowchart of patient identification for the study. A total of 321 patients with chronic HBV infection were included in the analysis. HCC, hepatocellular carcinoma; HCV, hepatitis C virus; HBV, hepatitis B virus.
Clinical and histological assessment
All patients admitted for liver biopsy and clinical and laboratory parameters were assessed at baseline. BMI was calculated based on body weight and height at the time of admission. Overweight was defined as BMI ≥ 23 kg/m2, and obesity was defined as BMI ≥ 25 kg/m2 in accordance with BMI cut-off points for determining overweight and obesity in Asian populations [13]. The following laboratory parameters were evaluated at baseline: hepatitis B e antigen (HBeAg), HBV DNA levels, and blood chemistry parameters, including albumin, total bilirubin, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase (ALT), gamma-glutamyl transferase, fasting glucose, and lipid profiles. Dyslipidemia was defined as serum triglyceride ≥ 150 mg/dL or high-density lipoprotein (HDL) cholesterol < 40 mg/dL in males and < 50 mg/dL in females [14]. The study patients were usually assessed at 3- to 6-month intervals throughout the entire follow-up period. Follow-up duration was defined as the period between the date of liver biopsy and the date of HCC diagnosis or last visit. Patients were treated with nucleos(t)ide analogues based on international guidelines and the physician’s decision. Most patients underwent regular HCC surveillance with abdominal imaging (e.g., ultrasonography or computed tomography) and/or monitoring of alpha-fetoprotein levels every 6–12 months. HCC diagnosis was established radiologically or histologically according to practice guidelines [15-18].
Ultrasound-guided liver biopsy was performed during inpatient stay. Histopathologic diagnosis was made by an experienced hepatopathologist (K.I.K.) who had no clinical information regarding the study subjects. A liver biopsy specimen was considered adequate if it was longer than 1.5 cm and contained six or more portal triads [19]. Hepatic fibrosis was assessed semiquantitatively according to the Ishak scoring system (score 0–6), and cirrhosis was defined as severe fibrosis with Ishak score 5 or 6 [20]. Fatty liver was defined as the presence of ≥ 5% hepatic steatosis [11].
Statistical analysis
We compared the baseline demographic and clinical characteristics of patients with or without fatty liver. Continuous variables were expressed as median and interquartile range, and groupwise comparisons were performed using Student’s t-test. The chi-squared test was used to compare categorical variables. The cumulative incidence rates of HCC were plotted using the Kaplan-Meier method, and differences between groups were compared using the log-rank test. The Cox proportional hazards model was used to identify risk factors for development of HCC. Clinicopathological factors were tested in a univariate analysis; and then a multivariate analysis was performed with factors that showed a significant association in the univariate analysis. Presence of fatty liver was included in the multivariate analysis, even if it was not identified as a significant risk factor in the univariate analysis, to evaluate its adjusted predictive value for HCC.
We employed IPW using the propensity score to adjust for between-group differences in patient characteristics, including metabolic factors. Propensity scores were computed by fitting a logistic regression model, in which demographic and clinicopathological characteristics were included, and the presence of fatty liver was deemed an outcome. The balance of baseline characteristics between groups was re-evaluated after IPW; and thereafter, the weighted Cox proportional hazards model was used to explore independent risk factors for HCC development. All statistical analyses were conducted as 2-tailed tests using IBM SPSS, version 24.0 (IBM Corp., Armonk, NY, USA) and R language, version 3.4.2 (R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was declared with a P-value less than 0.05.
RESULTS
Characteristics of the study population
A total of 321 patients with chronic HBV infection were included in the analysis. Steatosis of ≥ 5% was identified in 70 patients (21.8%), and the median (interquartile range [IQR]) follow-up duration was 5.3 (2.9–8.3) years. None of the study patients died or underwent liver transplantation during the follow-up period. Baseline demographic and clinicopathological characteristics of the study subjects are summarized in Table 1. The median (IQR) age was 41 (33–49) years overall and the patients with fatty liver were significantly older than those without fatty liver (P=0.005). BMI was higher in patients with fatty liver than that of patients without fatty liver, and 55.7% of patients with fatty liver were obese (BMI ≥ 25 kg/m2). Diabetes (P=0.06) and hypertension (P=0.005) were more prevalent among patients with fatty liver than among those without fatty liver. Patients with fatty liver had significantly more metabolic risk factors, which included obesity, history of hypertension or diabetes mellitus, and higher serum levels of triglyceride and fasting glucose, lower serum levels of HDL cholesterol, compared to patients without fatty liver (P<0.001). Serum concentrations of total cholesterol, low-density lipoprotein cholesterol, and triglyceride were higher among patients with fatty liver (P<0.001). Fasting glucose concentrations were also higher in patients with fatty liver; however, this difference between the two groups was not statistically significant (P=0.09). Cirrhosis was more frequently diagnosed in patients with fatty liver, but the difference between groups was not significant (P=0.09). Almost all patients (n=302; 94.1%) received antiviral therapy, and 167 patients (52.0%) were treated for over 5 years. The majority of study patients were HBeAg-positive, and the proportion of HBeAg-positive patients and HBV DNA levels at baseline were similar between the groups (P=0.11 and P=0.26, respectively).
Table 1.
Baseline clinicopathological characteristics
| Characteristics | Overall (N=321) | Steatosis |
P-value | |
|---|---|---|---|---|
| < 5% (n=251) | ≥ 5% (n=70) | |||
| Age (median [IQR]) (years) | 41 (33–49) | 41 (32–48) | 45 (36–51) | 0.005 |
| Male | 196 (61.1) | 146 (58.2) | 50 (71.4) | 0.05 |
| BMI (kg/m2) | <0.001 | |||
| <23 (Normal) | 134 (41.7) | 118 (47.0) | 16 (22.9) | |
| ≥23 to <25 (Overweight) | 81 (25.2) | 66 (26.3) | 15 (21.4) | |
| ≥25 (Obese) | 106 (33.0) | 67 (26.7) | 39 (55.7) | |
| Diabetes mellitus | 21 (6.5) | 13 (5.2) | 8 (11.4) | 0.06 |
| Hypertension | 25 (7.8) | 14 (5.6) | 11 (15.7) | 0.005 |
| Number of metabolic risk factors* | <0.001 | |||
| <3 | 269 (83.8) | 223 (88.8) | 46 (65.7) | |
| ≥3 | 52 (16.2) | 28 (11.2) | 24 (34.3) | |
| Liver cirrhosis | 64 (19.9) | 45 (17.9) | 19 (27.1) | 0.09 |
| Duration of antiviral treatment | 0.88 | |||
| Never or <5 years | 154 (48.0) | 121 (48.2) | 33 (47.1) | |
| ≥5 years | 167 (52.0) | 130 (51.8) | 37 (52.9) | |
| Laboratory data (median [IQR]) | ||||
| Albumin (g/dL) | 4.0 (3.7–4.3) | 4.0 (3.7–4.3) | 4.2 (3.8–4.4) | 0.17 |
| Total bilirubin (mg/dL) | 0.6 (0.5–0.9) | 0.7 (0.5–0.9) | 0.6 (0.5–0.9) | 0.53 |
| ALP (IU/L) | 185 (154–241) | 191 (156–245) | 176 (150–227) | 0.17 |
| AST (IU/L) | 76 (47–140) | 85 (51–153) | 53 (36–86) | <0.001 |
| ALT (IU/L) | 87 (44–160) | 94 (45–171) | 71 (32–114) | 0.02 |
| GGT (IU/L) | 61 (32–112) | 66 (33–115) | 52 (30–104) | 0.51 |
| Fasting glucose (mg/dL) | 98 (90–109) | 98 (90–108) | 103 (92–117) | 0.09 |
| Total cholesterol (mg/dL) | 164 (140–183) | 159 (137–179) | 176 (155–203) | <0.001 |
| HDL cholesterol (mg/dL) | 47.3 (38.8–58.4) | 48.3 (39.4–59.4) | 44.4 (35.4–54.4) | 0.21 |
| LDL cholesterol (mg/dL) | 92 (76–112) | 90 (75–108) | 105 (87–129) | <0.001 |
| Triglyceride (mg/dL) | 84 (65–115) | 80 (63–109) | 103 (81–137) | <0.001 |
| HBeAg (positive) | 218 (67.9) | 176 (70.1) | 42 (60.0) | 0.11 |
| HBV DNA | 0.26 | |||
| <6 log10 copies/mL | 146 (45.5) | 110 (43.8) | 36 (51.4) | |
| ≥6 log10 copies/mL | 175 (54.5) | 141 (56.2) | 34 (48.6) | |
Data are given as number (%) of patients, unless otherwise noted.
IQR, interquartile range; BMI, body mass index; ALP, alkaline phosphatase; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gammaglutamyl transferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; HBeAg, hepatitis B e antigen; HBV, hepatitis B virus.
Metabolic risk factors included obesity, history of hypertension or diabetes mellitus, and serum levels of triglyceride, HDL cholesterol, and fasting glucose.
Development of HCC
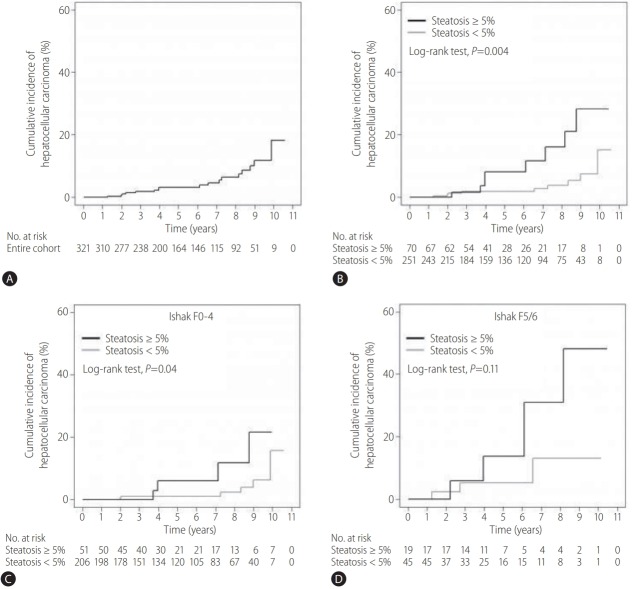
During the follow-up period, 17 patients (5.3%) were diagnosed with HCC: 8 (11.4%) among 70 patients with fatty liver and 9 (3.6%) among 251 patients without fatty liver. The 1-year, 3-year, and 5-year cumulative incidences of HCC were 0%, 1.8%, and 3.2%, respectively, and the rates differed significantly between the groups (P=0.004): 0%, 1.9%, and 1.9% among the patients without fatty liver and 0%, 1.6%, and 8.2% among those with fatty liver (Table 2, Fig. 2A, B). When the patients were divided into two subgroups according to the presence of cirrhosis, the 5-year cumulative incidences of HCC were 2.1% in the subgroup without cirrhosis (Ishak F0–4) and 8.0% in the subgroup with cirrhosis (Ishak F5/6) (Table 2). While the 5-year cumulative incidence rates were significantly higher among patients with fatty liver than among patients without fatty liver in the subgroup without cirrhosis (n=257: 6.0% vs. 1.1%; P=0.04) (Table 2, Fig. 2C), the 5-year cumulative incidence rates were not significantly different between the two groups within the subgroup with cirrhosis (n=64: 13.7% vs. 5.3%; P=0.11) (Table 2, Fig. 2D).
Table 2.
Development of HCC
| HCC development | Overall | Steatosis < 5% | Steatosis ≥ 5% | |
|---|---|---|---|---|
| Overall | ||||
| 1-year | Rate (%) | 0 | 0 | 0 |
| Sample size | 310 | 243 | 67 | |
| 3-year | Rate (%) | 1.8 | 1.9 | 1.6 |
| Sample size | 238 | 184 | 54 | |
| 5-year | Rate (%) | 3.2 | 1.9 | 8.2 |
| Sample size | 164 | 136 | 28 | |
| Ishak F0–4 | ||||
| 1-year | Rate (%) | 0 | 0 | 0 |
| Sample size | 248 | 198 | 50 | |
| 3-year | Rate (%) | 0.9 | 1.1 | 0 |
| Sample size | 191 | 151 | 40 | |
| 5-year | Rate (%) | 2.1 | 1.1 | 6.0 |
| Sample size | 141 | 120 | 21 | |
| Ishak F5/6 | ||||
| 1-year | Rate (%) | 0 | 0 | 0 |
| Sample size | 62 | 45 | 17 | |
| 3-year | Rate (%) | 5.6 | 5.3 | 5.9 |
| Sample size | 47 | 33 | 14 | |
| 5-year | Rate (%) | 8.0 | 5.3 | 13.7 |
| Sample size | 23 | 16 | 7 |
HCC, hepatocellular carcinoma.
Figure 2.
Incidence of HCC. (A) In the entire cohort. (B) Among patients with or without fatty liver. (C) Among patients with or without fatty liver in the subgroup without cirrhosis (Ishak F0–4). (D) Among patients with or without fatty liver in the subgroup with cirrhosis (Ishak F5/6). HCC, hepatocellular carcinoma.
A univariate Cox proportional hazards regression analysis identified age, diabetes mellitus, hypertension, cirrhosis, baseline serum ALT levels, and fatty liver as risk factors for HCC development (Table 3). A multivariate analysis with forward stepwise variable selection revealed a significant association between coexistence of fatty liver and an increased risk for HCC development (adjusted hazard ratio [HR], 3.005; 95% confidence interval [CI], 1.122–8.051; P=0.03) (Table 3). Age (adjusted HR, 1.109; 95% CI, 1.057–1.164; P<0.001) and cirrhosis (adjusted HR, 2.939; 95% CI, 1.088–7.935; P=0.03) were determined as independent risk factors for HCC development (Table 3).
Table 3.
Univariate and multivariate Cox analysis of the factors associated with HCC development
| Characteristics | HCC development |
|||
|---|---|---|---|---|
| Univariate analysis |
Multivariate analysis |
|||
| Hazard ratio (95% CI) | P-value | Adjusted hazard ratio (95% CI) | P-value | |
| Age (years) | 1.105 (1.057–1.155) | <0.001 | 1.109 (1.057–1.164) | <0.001 |
| Male | 1.403 (0.494–3.986) | 0.53 | ||
| BMI (kg/m2) | 0.52 | |||
| <23 (Normal) | 1 [Reference] | |||
| ≥23 to <25 (Overweight) | 1.851 (0.535–6.403) | 0.33 | ||
| ≥25 (Obese) | 1.826 (0.579–5.761) | 0.30 | ||
| Diabetes mellitus | 0.002 | |||
| No | 1 [Reference] | |||
| Yes | 6.019 (1.893–19.139) | |||
| Hypertension | 0.008 | |||
| No | 1 [Reference] | |||
| Yes | 4.138 (1.452–11.791) | |||
| Number of metabolic risk factors* | 0.95 | |||
| <3 | 1 [Reference] | |||
| ≥3 | 1.045 (0.299–3.651) | |||
| Liver cirrhosis | 0.007 | 0.03 | ||
| No | 1 [Reference] | 1 [Reference] | ||
| Yes | 3.880 (1.457–10.330) | 2.939 (1.088–7.935) | ||
| Histologic steatosis | 0.008 | 0.03 | ||
| < 5% | 1 [Reference] | 1 [Reference] | ||
| ≥ 5% | 3.669 (1.411–9.543) | 3.005 (1.122–8.051) | ||
| Duration of antiviral treatment | 0.43 | |||
| Never or <5 years | 1 [Reference] | |||
| ≥5 years | 0.635 (0.205–1.963) | |||
| Albumin (g/dL) | 0.874 (0.306–2.495) | 0.80 | ||
| Total bilirubin (mg/dL) | 0.902 (0.476–1.710) | 0.75 | ||
| ALP (IU/L) | 1.000 (0.993–1.006) | 0.91 | ||
| AST (IU/L) | 0.994 (0.986–1.002) | 0.17 | ||
| ALT (IU/L) | 0.990 (0.981–0.999) | 0.03 | ||
| GGT (IU/L) | 1.000 (0.997–1.004) | 0.83 | ||
| Fasting glucose (mg/dL) | 1.012 (0.999–1.024) | 0.07 | ||
| Total cholesterol (mg/dL) | 0.993 (0.978–1.008) | 0.33 | ||
| HDL cholesterol (mg/dL) | 1.010 (0.979–1.042) | 0.54 | ||
| LDL cholesterol (mg/dL) | 0.988 (0.970–1.008) | 0.23 | ||
| Triglyceride (mg/dL) | 0.995 (0.982–1.008) | 0.45 | ||
| HBeAg (positive) | 0.850 (0.313–2.305) | 0.75 | ||
| HBV DNA | 0.75 | |||
| <6 log10 copies/mL | 1 [Reference] | |||
| ≥6 log10 copies/mL | 0.841 (0.297–2.383) | |||
Data in parentheses are 95% CIs.
HCC, hepatocellular carcinoma; CI, confidence interval; BMI, body mass index; ALP, alkaline phosphatase; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; HBeAg, hepatitis B e antigen; HBV, hepatitis B virus.
Metabolic risk factors included obesity, history of hypertension or diabetes mellitus, and serum levels of triglyceride, HDL cholesterol, and fasting glucose.
HCC risk assessment using IPW
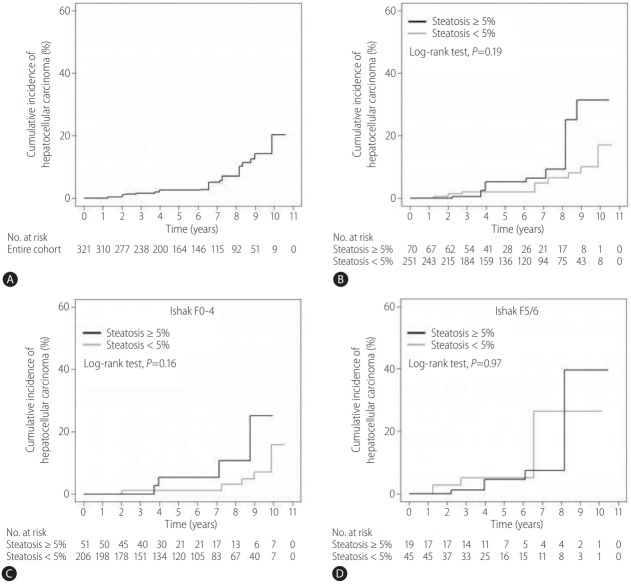
Baseline demographic and clinicopathological characteristics, including age, gender, BMI, diabetes, hypertension, and lipid profiles, became well balanced between the groups after adjustment for patient characteristics using IPW (Table 4). The weighted cumulative incidence rate of HCC at year 5 was 2.6% overall (Fig. 3A): 2.0% among the patients without fatty liver and 5.3% among those with fatty liver (Fig. 3B). There was no statistically significant difference in the 5-year cumulative risk for developing HCC between the two groups (P=0.19) (Fig. 3B). Moreover, the cumulative HCC incidence rates were similar between the groups in the subgroups without and with cirrhosis (P=0.16 and P=0.97, respectively) (Fig. 3C, D). When the weighted Cox proportional hazard models were fitted, a subsequent univariate analysis identified age, diabetes mellitus, and cirrhosis status as factors significantly associated with development of HCC (Table 5). In the multivariate Cox model, older age (adjusted HR, 1.063; 95% CI, 1.010–1.120; P=0.02) and status of diabetes mellitus (adjusted HR, 3.562; 95% CI, 1.117–11.359; P=0.03) were significantly associated with HCC risk, whereas coexistence of fatty liver (adjusted HR, 1.709; 95% CI, 0.404–7.228; P=0.47) and cirrhosis (adjusted HR, 3.554; 95% CI, 0.911–13.874; P=0.07) showed no significant association with HCC risk (Table 5).
Table 4.
Baseline clinicopathological characteristics after inverse probability weighting
| Characteristics | Overall (N=321) | Steatosis |
P-value | |
|---|---|---|---|---|
| < 5% (n=251) | ≥ 5% (n=70) | |||
| Age (median [IQR]) (years) | 41 (34–49) | 42 (33–49) | 40 (34–50) | 0.72 |
| Male | 186 (57.8) | 151 (60.7) | 35 (47.9) | 0.28 |
| BMI (kg/m2) | 0.74 | |||
| <23 (Normal) | 138 (42.8) | 104 (41.9) | 33 (45.8) | |
| ≥23 to <25 (Overweight) | 75 (23.2) | 62 (24.8) | 13 (17.6) | |
| ≥25 (Obese) | 110 (34.1) | 83 (33.4) | 27 (36.6) | |
| Diabetes mellitus | 18 (5.5) | 15 (5.8) | 3 (4.4) | 0.58 |
| Hypertension | 29 (8.9) | 23 (9.2) | 6 (8.2) | 0.83 |
| Number of metabolic risk factors* | 0.43 | |||
| <3 | 266 (82.7) | 209 (84.0) | 57 (78.1) | |
| ≥3 | 56 (17.3) | 40 (16.0) | 16 (21.9) | |
| Liver cirrhosis | 81 (25.0) | 51 (20.4) | 30 (41.1) | 0.08 |
| Duration of antiviral treatment | 0.42 | |||
| Never or <5 years | 158 (49.0) | 117 (46.9) | 41 (56.2) | |
| ≥5 years | 164 (51.0) | 132 (53.1) | 32 (43.8) | |
| Laboratory data (median [IQR]) | ||||
| Albumin (g/dL) | 4.0 (3.7–4.3) | 4 (3.7–4.4) | 3.9 (3.4–4.3) | 0.19 |
| Total bilirubin (mg/dL) | 0.7 (0.5–1.0) | 0.6 (0.5–1.0) | 0.8 (0.5–1.0) | 0.53 |
| ALP (IU/L) | 192 (157–250) | 189 (156–243) | 224 (169–250) | 0.31 |
| AST (IU/L) | 73 (47–136) | 79 (49–143) | 56 (47–106) | 0.30 |
| ALT (IU/L) | 78 (39–159) | 87 (43–169) | 65 (25–114) | 0.68 |
| GGT (IU/L) | 65 (33–126) | 61 (33–113) | 70 (33–178) | 0.33 |
| Fasting glucose (mg/dL) | 99 (90–112) | 98 (90–111) | 101 (92–124) | 0.49 |
| Total cholesterol (mg/dL) | 159 (136–180) | 163 (140–180) | 153 (136–181) | 0.44 |
| HDL cholesterol (mg/dL) | 49 (39.4–59.4) | 47.0 (39.4–58.5) | 54.0 (39.5–59.4) | 0.35 |
| LDL cholesterol (mg/dL) | 90 (74–110) | 92 (77–110) | 79 (69–105) | 0.29 |
| Triglyceride (mg/dL) | 87 (66–113) | 84 (65–114) | 94 (79–107) | 0.25 |
| HBeAg (positive) | 222 (68.9) | 167 (67.2) | 54 (74.7) | 0.37 |
| HBV DNA | 0.21 | |||
| <6 log10 copies/mL | 155 (48.2) | 112 (45.0) | 43 (59.2) | |
| ≥6 log10 copies/mL | 167 (51.8) | 137 (55.0) | 30 (40.8) | |
Data are given as number (%) of patients, unless otherwise noted.
IQR, interquartile range; BMI, body mass index; ALP, alkaline phosphatase; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gammaglutamyl transferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; HBeAg, hepatitis B e antigen; HBV, hepatitis B virus.
Metabolic risk factors included obesity, history of hypertension or diabetes mellitus, and serum levels of triglyceride, HDL cholesterol, and fasting glucose.
Figure 3.
Incidence of HCC after IPW. (A) In the entire cohort. (B) Among patients with or without fatty liver. (C) Among patients with or without fatty liver in the subgroup without cirrhosis (Ishak F0–4). (D) Among patients with or without fatty liver in the subgroup with cirrhosis (Ishak F5/6). HCC, hepatocellular carcinoma; IPW, inverse probability weighting.
Table 5.
Univariate and multivariate Cox analysis of factors associated with HCC development after inverse probability weighting
| Characteristics | HCC development |
|||
|---|---|---|---|---|
| Univariate analysis |
Multivariate analysis |
|||
| Hazard ratio (95% CI) | P-value | Adjusted hazard ratio (95% CI) | P-value | |
| Age (years) | 1.077 (1.025–1.132) | 0.003 | 1.063 (1.010–1.120) | 0.02 |
| Male | 1.349 (0.382–4.760) | 0.64 | ||
| BMI (kg/m2) | 0.93 | |||
| <23 (Normal) | 1 [Reference] | |||
| ≥23 to <25 (Overweight) | 1.300 (0.35–4.828) | 0.70 | ||
| ≥25 (Obese) | 1.123 (0.269–4.692) | 0.87 | ||
| Diabetes mellitus | 0.004 | 0.03 | ||
| No | 1 [Reference] | 1 [Reference] | ||
| Yes | 5.331 (1.686–16.853) | 3.562 (1.117–11.359) | ||
| Hypertension | 0.14 | |||
| No | 1 [Reference] | |||
| Yes | 2.951 (0.700–12.444) | |||
| Number of metabolic risk factors* | 0.98 | |||
| <3 | 1 [Reference] | |||
| ≥3 | 1.022 (0.192–5.433) | |||
| Liver cirrhosis | 0.01 | 0.07 | ||
| No | 1 [Reference] | 1 [Reference] | ||
| Yes | 4.371 (1.373–13.918) | 3.554 (0.911–13.874) | ||
| Histologic steatosis | 0.10 | 0.47 | ||
| < 5% | 1 [Reference] | 1 [Reference] | ||
| ≥ 5% | 2.620 (0.828–8.297) | 1.709 (0.404–7.228) | ||
| Duration of antiviral treatment | 0.77 | |||
| Never or <5 years | 1 [Reference] | |||
| ≥5 years | 0.838 (0.252–2.784) | |||
| Albumin (g/dL) | 0.689 (0.303–1.565) | 0.37 | ||
| Total bilirubin (mg/dL) | 0.953 (0.692–1.311) | 0.77 | ||
| ALP (IU/L) | 0.999 (0.992–1.005) | 0.72 | ||
| AST (IU/L) | 0.995 (0.988–1.003) | 0.25 | ||
| ALT (IU/L) | 0.994 (0.985–1.004) | 0.23 | ||
| GGT (IU/L) | 1.002 (0.999–1.006) | 0.22 | ||
| Fasting glucose (mg/dL) | 1.014 (1.000–1.028) | 0.05 | ||
| Total cholesterol (mg/dL) | 1.001 (0.989–1.013) | 0.92 | ||
| HDL cholesterol (mg/dL) | 1.007 (0.959–1.057) | 0.79 | ||
| LDL cholesterol (mg/dL) | 0.997 (0.97–1.025) | 0.83 | ||
| Triglyceride (mg/dL) | 1.002 (0.993–1.01) | 0.69 | ||
| HBeAg (positive) | 0.806 (0.232–2.804) | 0.74 | ||
| HBV DNA | 0.77 | |||
| <6 log10 copies/mL | 1 [Reference] | |||
| ≥6 log10 copies/mL | 0.814 (0.21–3.164) | |||
Data in parentheses are 95% CIs.
HCC, hepatocellular carcinoma; CI, confidence interval; BMI, body mass index; ALP, alkaline phosphatase; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; HBeAg, hepatitis B e antigen; HBV, hepatitis B virus.
Metabolic risk factors included obesity, history of hypertension or diabetes mellitus, and serum levels of triglyceride, HDL cholesterol, and fasting glucose.
DISCUSSION
In the present study, we demonstrated that the prevalence of histologically proven fatty liver was 21.8%, and coexistence of fatty liver was associated with a 3–fold increased risk for developing HCC in CHB patients without excessive alcohol consumption. Age and liver cirrhosis were additional independent risk factors for HCC in our study population. However, after rigorous adjustment for patient characteristics including metabolic factors using IPW, no significant association between coexistence of fatty liver and HCC development was observed. Instead, diabetes showed significant association with risk potentiating HCC development by 3.6–fold.
Because we aimed to accurately assess the hepatocarcinogenic effect of coexisting NAFLD in Korean patients with chronic HBV infection, patients undergoing liver biopsy and subsequent histologic diagnosis, which is the gold standard for assessment of hepatic steatosis [11], were selected for analysis. After adjusting for age and cirrhosis, histologically diagnosed NAFLD was determined to be an independent predictive factor of HCC development. Although diabetes and hypertension were identified as significant risk factors for HCC in a univariate analysis, these factors were excluded in the variable selection process for multivariate analysis. Intriguingly, the impact of fatty liver on the risk for developing HCC in CHB patients disappeared under conditions in which possible confounders, including metabolic factors, were thoroughly controlled. Cirrhosis, which is a well-known risk factor of HCC, was adjusted in the multivariate analysis and IPW, however, the impact of coexisting fatty liver might be attenuated in cirrhotic patients. Therefore, we repeated weighted analyses in the subgroup of non-cirrhotic patients (n=257). As results, the cumulative risks of developing HCC were not significantly different between patients without and with fatty liver (P=0.23 by weighted log-rank test). Moreover, coexistence of fatty liver was not associated with HCC risk (adjusted HR, 4.564; P=0.07) after adjustment for age (adjusted HR, 1.099; P=0.004), diabetes (adjusted HR, 7.018; P=0.007), and ALT (adjusted HR, 0.988; P=0.05). Overall, the obtained results were similar to those in the total study population. These findings suggest that coexisting NAFLD as a hepatic manifestation of metabolic syndrome potentiates the risk for HCC in CHB patients, but hepatic steatosis per se does not.
Consistent with our present study, Chan and colleagues recently reported that histologically proven fatty liver was associated with a 7.3–fold increased risk for HCC development in patients chronically infected with HBV [9]. In another study, when the influence of metabolic risk factors, such as obesity, diabetes mellitus, hypertension, and hypertriglyceridemia, on HCC risk and liver-related mortality in male patients with chronic HBV infection was analyzed, patients with ≥ 3 metabolic risk factors were at a 2.3–fold higher risk for HCC [21]. Particularly, the association of metabolic risk factors and insulin resistance with HCC risk was more evident in patients with low viral load (HBV DNA < 10,000 copies/mL), and this point supports findings in our present study, in which more than 90% of the study subjects were treated with antiviral therapies. Although results of the previous study drawn from a large Taiwanese cohort with long-term follow-up did not include data on the effects of histologically proven fatty liver, the results were in line with findings in our present study. Collectively, thorough screening and management of metabolic risk factors in CHB patients is crucial for preventing HCC, and ultimately improving the long-term clinical outcomes. In the present study, cirrhosis, a well-known major risk factor for HCC [5], was revealed to possess a significant association with a 2.9–fold increased risk for developing HCC. Coexisting fatty liver potentiated the HCC risk in patients without cirrhosis (Ishak F0–4), whereas did not in those with cirrhosis (Ishak F5/6). These results implicate that modifiable metabolic factors need be dealt with more strictly in young non-cirrhotic patients with chronic HBV infection.
In our study population, the prevalence of histologically proven fatty liver was 21.8%. Concurrent fatty liver was histologically diagnosed in 39.6% of the CHB patients included in the aforementioned study [9], and the prevalence of histologically proven fatty liver in Chinese CHB patients was reported to have gradually increased from 8.2% to 31.8% over 10 years [22]. Although the CHB patients included in our present study showed a lower prevalence of NAFLD when compared with those study population, NAFLD is becoming more prevalent as obesity becomes a worldwide epidemic. Therefore, physicians have to become more alert to the possibility of metabolic syndrome when treating patients with chronic HBV infection in the future.
There are several limitations to this study. First, we did not adopt the scoring systems for semiquantitative severity assessment of necroinflammation in NAFLD, such as NAFLD activity score and steatosis-activity-fibrosis score [23-25]. NAFLD is generally regarded as benign, and there are few reports of HCC developing from simple hepatic steatosis. However, the influence of NASH on the risk for HCC in CHB patients is worthy of further investigation. Second, because our data were collected retrospectively, insulin resistance, which plays a key role in glucose and lipid metabolism, could not be fully assessed in the present study. Moreover, the use of statin or metformin, a possible confounding factor, was not available for adjustment in our retrospective cohort. Prospective studies are warranted that evaluate the association between coexisting fatty liver and HCC risk, with adjustment for metabolic parameters reflecting insulin resistance, such as the homeostasis model assessment of insulin resistance index, and medication history [26]. Third, liver cirrhosis, a well-known risk factor for HCC, was not independent predictor of development of HCC after IPW, this finding might result from limited events during our study period and the association between liver cirrhosis and HCC risk would become evident with accumulation of events over time.
In conclusion, we demonstrated that coexisting fatty liver was associated with an increased risk for HCC development in patients with chronic HBV infection. However, considering that the association was not evident after adjusting for metabolic factors, fatty liver as a hepatic manifestation of metabolic syndrome, may possess important predictive value for HCC. Thorough screening and management of metabolic risk factors is needed to improve the long-term outcomes of patients chronically infected with HBV.
Abbreviations
- ALT
alanine aminotransferase
- BMI
body mass index
- CHB
chronic hepatitis B
- CI
confidence interval
- HBeAg
hepatitis B e antigen
- HBV
hepatitis B virus
- HCC
hepatocellular carcinoma
- HR
hazards ratio
- IPW
inverse probability weighting
- IQR
interquartile range
- NAFLD
nonalcoholic fatty liver disease
- NASH
nonalcoholic steatohepatitis
Study Highlights
·The prevalence of histologically proven fatty liver was 21.8%, and coexisting fatty liver was associated with a 3–fold increased risk for hepatocellular carcinoma (HCC) development in chronic hepatitis B (CHB) patients without excessive alcohol intake.
·However, when demographic and clinical characteristics including metabolic factors were balanced using inverse probability weighting based on each patient’s propensity score, the association between coexistence of fatty liver and HCC development was not evident.
·These findings suggest that superimposed nonalcoholic fatty liver disease (NAFLD) as a hepatic manifestation of metabolic syndrome, not hepatic steatosis per se, may possess important predictive value for HCC in CHB patients.
Footnotes
Author contribution
The corresponding author (Seong Gyu Hwang) had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Yun Bin Lee and Seong Gyu Hwang.
Provision of study materials or patients: Yun Bin Lee, Young Eun Chon, Mi Na Kim, Joo Ho Lee, Hana Park, Kyu Sung Rim, and Seong Gyu Hwang.
Collection and assembly of data: Yun Bin Lee, Yeonjung Ha, and Kwang-il Kim.
Data analysis and interpretation: Yun Bin Lee, Soo-Hwan Kim, and Seong Gyu Hwang.
Manuscript writing: Yun Bin Lee and Seong Gyu Hwang.
Final approval of manuscript: All authors.
Grant support
This study was supported by a grant of the Korea Healthcare Technology R&D project, funded by the Ministry for Health, Welfare & Family Affairs, Republic of Korea (HI13C1398).
Conflicts of Interest The authors declare that they have no competing interests.
REFERENCES
- 1.Schweitzer A, Horn J, Mikolajczyk RT, Krause G, Ott JJ. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. Lancet. 2015;386:1546–1555. doi: 10.1016/S0140-6736(15)61412-X. [DOI] [PubMed] [Google Scholar]
- 2.Beasley RP. Hepatitis B virus. The major etiology of hepatocellular carcinoma. Cancer. 1988;61:1942–1956. doi: 10.1002/1097-0142(19880515)61:10<1942::aid-cncr2820611003>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 3.McMahon BJ. The natural history of chronic hepatitis B virus infection. Semin Liver Dis. 2004;24 Suppl 1:17–21. doi: 10.1055/s-2004-828674. [DOI] [PubMed] [Google Scholar]
- 4.Hadziyannis SJ, Papatheodoridis GV. Hepatitis B e antigen-negative chronic hepatitis B: natural history and treatment. Semin Liver Dis. 2006;26:130–141. doi: 10.1055/s-2006-939751. [DOI] [PubMed] [Google Scholar]
- 5.Trépo C, Chan HL, Lok A. Hepatitis B virus infection. Lancet. 2014;384:2053–2063. doi: 10.1016/S0140-6736(14)60220-8. [DOI] [PubMed] [Google Scholar]
- 6.Sanyal AJ, American Gastroenterological Association AGA technical review on nonalcoholic fatty liver disease. Gastroenterology. 2002;123:1705–1725. doi: 10.1053/gast.2002.36572. [DOI] [PubMed] [Google Scholar]
- 7.Contos MJ, Sanyal AJ. The clinicopathologic spectrum and management of nonalcoholic fatty liver disease. Adv Anat Pathol. 2002;9:37–51. doi: 10.1097/00125480-200201000-00005. [DOI] [PubMed] [Google Scholar]
- 8.Ascha MS, Hanouneh IA, Lopez R, Tamimi TA, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology. 2010;51:1972–1978. doi: 10.1002/hep.23527. [DOI] [PubMed] [Google Scholar]
- 9.Chan AW, Wong GL, Chan HY, Tong JH, Yu YH, Choi PC, et al. Concurrent fatty liver increases risk of hepatocellular carcinoma among patients with chronic hepatitis B. J Gastroenterol Hepatol. 2017;32:667–676. doi: 10.1111/jgh.13536. [DOI] [PubMed] [Google Scholar]
- 10.Chen CL, Yang HI, Yang WS, Liu CJ, Chen PJ, You SL, et al. Metabolic factors and risk of hepatocellular carcinoma by chronic hepatitis B/C infection: a follow-up study in Taiwan. Gastroenterology. 2008;135:111–121. doi: 10.1053/j.gastro.2008.03.073. [DOI] [PubMed] [Google Scholar]
- 11.Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–357. doi: 10.1002/hep.29367. [DOI] [PubMed] [Google Scholar]
- 12.Korean Association for the Study of the Liver (KASL) KASL clinical practice guidelines: management of nonalcoholic fatty liver disease. Clin Mol Hepatol. 2013;19:325–348. doi: 10.3350/cmh.2013.19.4.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.WHO Expert Consultation Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–163. doi: 10.1016/S0140-6736(03)15268-3. [DOI] [PubMed] [Google Scholar]
- 14.Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–1645. doi: 10.1161/CIRCULATIONAHA.109.192644. [DOI] [PubMed] [Google Scholar]
- 15.Korean Liver Cancer Study Group and National Cancer Center, Korea Practice guidelines for management of hepatocellular carcinoma 2009. Korean J Hepatol. 2009;15:391–423. doi: 10.3350/kjhep.2009.15.3.391. [DOI] [PubMed] [Google Scholar]
- 16.Korean Liver Cancer Study Group (KLCSG) National Cancer Center, Korea (NCC) 2014 Korean Liver Cancer Study Group-National Cancer Center Korea practice guideline for the management of hepatocellular carcinoma. Korean J Radiol. 2015;16:465–522. doi: 10.3348/kjr.2015.16.3.465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bruix J, Sherman M, Practice Guidelines Committee. American Association for the Study of Liver Diseases Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236. doi: 10.1002/hep.20933. [DOI] [PubMed] [Google Scholar]
- 18.Bruix J, Sherman M, American Association for the Study of Liver Diseases Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022. doi: 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bravo AA, Sheth SG, Chopra S. Liver biopsy. N Engl J Med. 2001;344:495–500. doi: 10.1056/NEJM200102153440706. [DOI] [PubMed] [Google Scholar]
- 20.Ishak K, Baptista A, Bianchi L, Callea F, De Groote J, Gudat F, et al. Histological grading and staging of chronic hepatitis. J Hepatol. 1995;22:696–699. doi: 10.1016/0168-8278(95)80226-6. [DOI] [PubMed] [Google Scholar]
- 21.Yu MW, Lin CL, Liu CJ, Yang SH, Tseng YL, Wu CF. Influence of metabolic risk factors on risk of hepatocellular carcinoma and liver-related death in men with chronic hepatitis B: a large cohort study. Gastroenterology. 2017;153:1006–1017. doi: 10.1053/j.gastro.2017.07.001. e5. [DOI] [PubMed] [Google Scholar]
- 22.Wang MM, Wang GS, Shen F, Chen GY, Pan Q, Fan JG. Hepatic steatosis is highly prevalent in hepatitis B patients and negatively associated with virological factors. Dig Dis Sci. 2014;59:2571–2579. doi: 10.1007/s10620-014-3180-9. [DOI] [PubMed] [Google Scholar]
- 23.Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–1321. doi: 10.1002/hep.20701. [DOI] [PubMed] [Google Scholar]
- 24.Bedossa P, Poitou C, Veyrie N, Bouillot JL, Basdevant A, Paradis V, et al. Histopathological algorithm and scoring system for evaluation of liver lesions in morbidly obese patients. Hepatology. 2012;56:1751–1759. doi: 10.1002/hep.25889. [DOI] [PubMed] [Google Scholar]
- 25.Bedossa P, FLIP Pathology Consortium Utility and appropriateness of the fatty liver inhibition of progression (FLIP) algorithm and steatosis, activity, and fibrosis (SAF) score in the evaluation of biopsies of nonalcoholic fatty liver disease. Hepatology. 2014;60:565–575. doi: 10.1002/hep.27173. [DOI] [PubMed] [Google Scholar]
- 26.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]