This case-control study examines the association between the use of dipeptidyl peptidase 4 inhibitors and the risk of developing bullous pemphigoid in patients with diabetes.
Key Points
Questions
What is the association between use of dipeptidyl peptidase 4 inhibitors and the risk of developing bullous pemphigoid in patients with diabetes?
Findings
In this population-based, case-control study of insurance claims data from 1340 patients in Korea, the proportion of patients with diabetes among all patients with bullous pemphigoid increased between 2012 and 2016 and use of dipeptidyl peptidase 4 inhibitors was significantly associated with an increased risk of developing bullous pemphigoid in patients with diabetes compared with control patients. The risk of bullous pemphigoid was highest in male patients using vildagliptin.
Meaning
Use of dipeptidyl peptidase 4 inhibitors, particularly vildagliptin, may be associated with the development of bullous pemphigoid in male patients with diabetes.
Abstract
Importance
Recent studies suggest that dipeptidyl peptidase 4 (DPP-4) inhibitors are associated with an increased risk of developing bullous pemphigoid (BP). Population-based studies on the association between DPP-4 inhibitors and BP are limited.
Objective
To characterize the potential association between the use of DPP-4 inhibitors and an increased risk of developing BP.
Design, Setting, and Participants
This retrospective, nationwide, population-based, case-control study using Korean insurance claims data from January 1, 2012, to December 31, 2016, included patients with newly diagnosed BP and diabetes. Control patients with diabetes (and without BP) were randomly obtained after matching for age, sex, and year of diagnosis within the same period.
Main Outcomes and Measures
The number of patients with newly diagnosed BP and diabetes per year and annual changes in the proportion of patients with diabetes among all patients with BP were measured. The association between use of DPP-4 inhibitors and risk of developing BP was analyzed using univariate and multivariate logistic regression analyses.
Results
The study included 670 case patients (with diabetes and BP) and 670 control patients (with only diabetes) (mean [SD] age, 75.3 [10.0] years in each group; 342 [51.0%] male in each group). The number of patients with diabetes and BP more than doubled during the study period (from 77 in 2012 to 206 in 2016). The proportion of patients with diabetes among all patients with BP also increased (from 0.18 in 2012 to 0.33 in 2016). The use of DPP-4 inhibitors was associated with a significant increase in the risk of developing BP (adjusted odds ratio [aOR], 1.58; 95% CI, 1.25-2.00; P < .001); among all DPP-4 inhibitors used in Korea, the highest aOR was associated with the use of vildagliptin (aOR, 1.81; 95% CI, 1.31-2.50; P < .001). Subgroup analyses revealed a significant association in male patients (aOR, 1.91; 95% CI, 1.39-2.63; P < .001) and that vildagliptin was the most high-risk DPP-4 inhibitor (aOR, 2.70; 95% CI, 1.73-4.34; P < .001).
Conclusions and Relevance
The findings suggest that DPP-4 inhibitors are associated with a significantly increased risk of the development of BP in patients with diabetes. Of the DPP-4 inhibitors available in Korea, vildagliptin was associated with the highest risk, particularly in male patients. Practitioners should consider that DPP-4 inhibitors, particularly vildagliptin, may be associated with the development of BP in patients with diabetes. These nationwide, population-based results may serve as a foundation for further studies seeking to understand how DPP-4 inhibitors contribute to the development of BP.
Introduction
Bullous pemphigoid (BP) is a rare autoimmune bullous disease characterized by generalized pruritic tense blisters and erosions, mainly on the skin in elderly patients.1 It is characterized by an autoimmune response directed against hemidesmosomal proteins (BP180 and BP230) at the dermoepidermal junction.2 Although the exact pathogenesis of BP remains unknown, neurologic disorders (eg, dementia and Parkinson disease), psychiatric disorders, inflammatory skin diseases, and conditions associated with being bedridden are considered to be risk factors of BP.3,4,5,6 Various factors (eg, trauma, burns, radiotherapy, and drugs) may be involved in the development of BP.1
Dipeptidyl peptidase 4 (DPP-4) inhibitors are a new class of oral antidiabetic agents for patients with type 2 diabetes that can be used alone or in combination with metformin.7 DPP-4 inhibitors inhibit the degradation of incretins, thereby decreasing glucagon release, increasing insulin secretion, and decreasing blood glucose levels.8 Sitagliptin was the first DPP-4 inhibitor approved by the US Food and Drug Administration in 2006, followed by saxagliptin in 2009, linagliptin in 2011, and alogliptin in 2013. Additional DPP-4 inhibitors are also available in Korea, including vildagliptin, gemigliptin, anagliptin, and teneligliptin. Recent clinical reports suggest that the use of DPP-4 inhibitors is associated with an increased risk of developing BP, but large population-based studies are rare, and the association is not yet fully elucidated.9,10,11,12 In this study, we investigated the association between the use of DPP-4 inhibitors and BP using insurance claims data in Korea. In addition, we analyzed the association of patient age, sex, and a specific DPP-4 inhibitor with the risk of developing BP in patients with diabetes prescribed a DPP-4 inhibitor.
Methods
Data Source and Acquisition
We performed a retrospective, nationwide, population-based, case-control study by acquiring data from the Health Insurance Review & Assessment Service database in Korea, which covers all Koreans, from January 1, 2012, to December 31, 2016. The data included all medical records, including patient’s age, sex, and information related to diagnosis and medications. This study was approved by the institutional review board of Bundang CHA Medical Center, Seongnam, Republic of Korea, and patient consent was waived.
Study Population
Patients with diabetes and BP were identified by collecting data on patients who visited any type of medical institution with a principal diagnosis of BP and diabetes according to the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10). To improve the accuracy of the diagnosis, we included patients who had received a diagnosis of BP (ICD-10 code L12.0) and diabetes (ICD-10 codes E11, E13, or E14) as the principal diagnosis on at least 2 occasions during the study period. The control patients were randomly obtained from among patients with diabetes who had not previously received a diagnosis of bullous skin diseases after matching for age, sex, and year of diagnosis with the patients with diabetes and BP from January 1, 2012, to December 31, 2016. One control patient was matched for each patient with diabetes and BP.
For all case and control patients, comorbid disorders were investigated. Cardiovascular, neurologic, other endocrinologic (except for diabetes), malignant, nephrologic, psychological, rheumatic, and other dermatologic disorders were included.
We examined the number of patients who were prescribed DPP-4 inhibitors and the specific type(s) of DPP-4 inhibitors prescribed during the study period in both groups. We classified DPP-4 inhibitors into 4 groups; vildagliptin, sitagliptin, linagliptin, and others (ie, saxagliptin, teneligliptin, gemigliptin, and alogliptin). All DPP-4 inhibitors prescribed before the diagnosis of BP were included, and other cotreatments were also reviewed. The number of patients with newly diagnosed BP and diabetes per year and the proportion of patients with diabetes among all patients with BP were assessed.
Statistical Analysis
All patient characteristics were collected, and the results are expressed as mean (SD). Pearson χ2 or Fisher exact tests were used to directly compare the individual comorbid disorders and cotreatments between the case and control groups. All factors with a P < .05 are considered as possible confounding variables in multivariate logistic regression models. The factors for adjustment were neurologic, malignant, and psychological comorbid disorders and receipt of spironolactone or psycholeptics. To determine the association of DPP-4 inhibitors with BP in terms of adjusted odds ratios (aORs) and 95% CIs, the multivariate logistic regression model was used. A subgroup analysis by age, sex, and the specific type of DPP-4 inhibitor was also investigated. A 2-sided P < .05 was considered to be statistically significant. All data were analyzed using PASW Statistics, version 18.0 (IBM Co).
Results
Clinical Characteristics of Patients and Controls
In total, 670 patients with newly diagnosed BP and diabetes and 670 controls were included in the study period (mean [SD] age, 75.3 [10.0] years in each group; 342 [51.0%] male in each group). When analyzed according to age, 590 patients (88.1%) were 65 years or older, and the most common age group was between 75 and 84 years (298 [44.5%]). The most common comorbidities in patients were cardiovascular (423 [63.1%]), psychological (143 [21.3%]), and other endocrinologic disorders (140 [20.9%]). Neurologic and psychological disorders were significantly more common in the patient group than the control group; malignant tumors had the opposite pattern. The number of patients prescribed DPP-4 inhibitors was significantly higher in the patient group (260 [38.9%]) compared with the control group (188 [28.1%]) (P < .001). In both groups, vildagliptin was the most frequently prescribed DPP-4 inhibitor, followed by linagliptin and sitagliptin (Table 1 and Table 2). For the other cotreatments, spironolactone (63 cases [9.4%] vs 42 controls [6.3%]; P = .04) and psycholeptics (173 cases [25.8%] vs 122 controls [18.2%]; P < .001) were prescribed significantly more in the patient group than the control group. There were no differences in other antidiabetic medications between both groups (518 cases [77.3%] vs 558 controls [83.3%]; P = .13) (Table 2).
Table 1. Demographics and Comorbidities of Patients and Control Individualsa.
| Variable | Patients | Controls | P Valueb |
|---|---|---|---|
| Sex | |||
| Male | 342 (51.0) | 342 (51.0) | NA |
| Female | 328 (49.0) | 328 (49.0) | NA |
| Age at diagnosis, mean (SD), y | 75.3 (10.0) | 75.3 (10.0) | NA |
| <65 | 80 | 80 | NA |
| 65-74 | 196 | 196 | NA |
| 75-84 | 298 | 298 | NA |
| ≥85 | 96 | 96 | NA |
| Comorbidities | |||
| Cardiovascular | 423 (63.1) | 389 (58.1) | .07 |
| Neurologic | 82 (12.2) | 49 (7.3) | .003 |
| Endocrinologicc | 140 (20.9) | 140 (20.9) | >.99 |
| Malignant | 59 (8.8) | 86 (12.8) | .03 |
| Nephrologic | 110 (16.4) | 88 (13.1) | .11 |
| Psychological | 143 (21.3) | 106 (15.8) | .01 |
| Rheumatic | 9 (1.3) | 10 (1.5) | >.99 |
| Dermatologicd | 23 (3.4) | 27 (4.0) | .67 |
Abbreviation: NA, not applicable.
Data are presented as number (percentage) of patients or controls unless otherwise indicated.
P values determined with the χ2 test or Fisher exact test, as appropriate.
Except for diabetes.
Except for bullous pemphigoid.
Table 2. DPP-4 Inhibitor Prescriptions and Other Cotreatments of Cases and Control Individuals.
| Variable | No. (%) | P Valuea | |
|---|---|---|---|
| Patients | Controls | ||
| DPP-4 Inhibitor | |||
| None | 410 (61.1) | 482 (71.9) | NA |
| Yes | 260 (38.9) | 188 (28.1) | <.001 |
| Vildagliptin | 118 (17.6) | 71 (10.6) | |
| Sitagliptin | 89 (13.3) | 57 (8.5) | |
| Linagliptin | 90 (13.4) | 58 (8.7) | |
| Othersb | 34 (5.1) | 25 (3.7) | |
| Cotreatments | |||
| Diuretics | |||
| Spironolactone | 63 (9.4) | 42 (6.3) | .04 |
| Furosemide | 160 (23.9) | 136 (20.3) | .13 |
| Thiazide | 145 (21.6) | 124 (18.5) | .17 |
| Other cardiovascular drugs | |||
| ACE inhibitor or ARB inhibitor | 358 (53.4) | 323 (48.2) | .06 |
| Calcium channel blocker | 304 (45.4) | 274 (40.9) | .11 |
| β-Blocker | 146 (21.8) | 122 (18.2) | .12 |
| Statins | 359 (53.6) | 362 (54.0) | .91 |
| Antiplatelet or anticoagulant | 404 (60.3) | 371 (55.4) | .08 |
| Psycholepticsc | 173 (25.8) | 122 (18.2) | <.001 |
| Antiparkinson drugs | 54 (8.1) | 46 (6.9) | .47 |
| NSAIDs | 177 (26.4) | 150 (22.4) | .10 |
| Other antidiabetic agentsd | 518 (77.3) | 558 (83.3) | .13 |
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; DPP-4, dipeptidyl peptidase 4; NSAID, nonsteroidal anti-inflammatory drug.
P values determined with the χ2 test or Fisher exact test, as appropriate.
Others include patients prescribed saxagliptin, teneligliptin, gemigliptin, and alogliptin.
Psycholeptics (N05) include antipsychotics, anxiolytics, hypnotics, and sedatives.
Except for DPP-4 inhibitors.
Annual Changes in the Proportion of Patients With Diabetes Among All Patients With BP
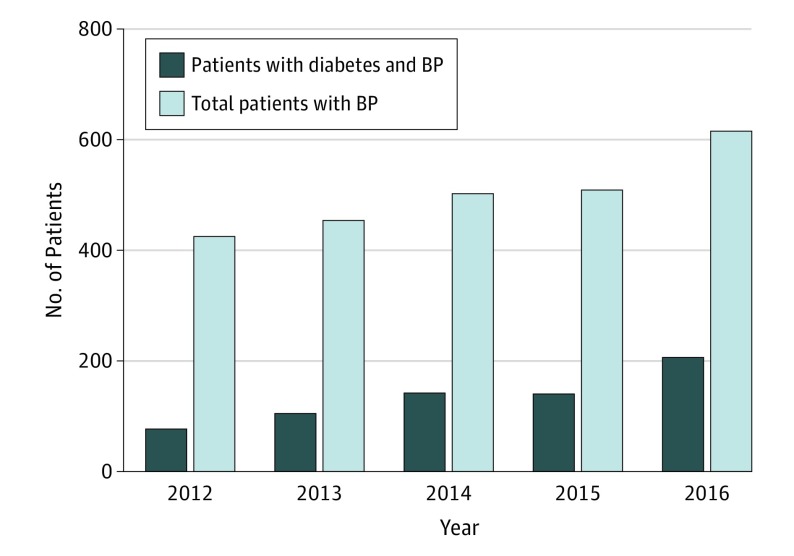
From 2012 to 2016, a total of 2505 patients were newly diagnosed with BP, which constitutes a gradual increase yearly within this timeframe. The number of patients with diabetes and BP more than doubled from 77 in 2012 to 206 in 2016. The proportion of patients with diabetes among all patients with BP increased from 0.18 in 2012 to 0.33 in 2016 (Figure).
Figure. Annual Number of Patients Newly Diagnosed With Bullous Pemphigoid (BP) and Patients With Diabetes and BP.
DPP-4 Inhibitors and BP
Univariate analysis revealed that the use of DPP-4 inhibitors in patients with diabetes was associated with a higher risk of BP (OR, 1.61; 95% CI, 1.28-2.03; P < .001). Multivariate logistic regression analysis revealed that the risk of BP was significantly higher in patients with diabetes who were prescribed a DPP-4 inhibitor compared with the controls (aOR, 1.58; 95% CI, 1.25-2.00; P < .001). Of the DPP-4 inhibitors available in Korea, use of vildagliptin had the greatest association with increased risk of BP (aOR, 1.81; 95% CI, 1.31-2.50; P < .001); sitagliptin (aOR, 1.70; 95% CI, 1.19-2.43; P = .004) and linagliptin (aOR, 1.64; 95% CI, 1.15-2.33; P = .006) were also significantly associated with the development of BP (Table 3).
Table 3. Univariate and Multivariate Analyses of the Association Between DPP-4 Inhibitors and the Development of Bullous Pemphigoid in Patients With Diabetes Mellitus.
| Drug, Patient Group | No. (%) | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|
| Crude OR (95% CI)a | P Value | Adjusted OR (95% CI)b | P Value | ||
| All DPP-4 inhibitors | |||||
| Patients | 260 (38.9) | 1.61 (1.28-2.03) | <.001 | 1.58 (1.25-2.00) | <.001 |
| Controls | 188 (28.1) | 1 [Reference] | 1 [Reference] | ||
| Vildagliptin | |||||
| Patients | 118 (17.6) | 1.84 (1.34-2.54) | <.001 | 1.81 (1.31-2.50) | <.001 |
| Controls | 71 (10.6) | 1 [Reference] | 1 [Reference] | ||
| Sitagliptin | |||||
| Patients | 89 (13.3) | 1.68 (1.18-2.39) | .004 | 1.70 (1.19-2.43) | .004 |
| Controls | 57 (8.5) | 1 [Reference] | 1 [Reference] | ||
| Linagliptin | |||||
| Patients | 90 (13.4) | 1.64 (1.16-2.32) | .006 | 1.64 (1.15-2.33) | .006 |
| Controls | 58 (8.7) | 1 [Reference] | 1 [Reference] | ||
| Others | |||||
| Patients | 34 (5.1) | 1.38 (0.81-2.34) | .23 | 1.25 (0.73-2.14) | .41 |
| Controls | 25 (3.7) | 1 [Reference] | 1 [Reference] | ||
Abbreviations: DPP-4, dipeptidyl peptidase 4; NA, not applicable; OR, odds ratio.
The ORs and 95% CIs were estimated using univariate logistic regression analysis.
The ORs and 95% CIs were estimated using multivariate logistic regression analysis adjusted for neurologic, malignant, and psychological comorbid disorders and receipt of spironolactone and psycholeptics.
Subgroup Analysis According to Age
We divided all patients into 2 groups: younger than 75 years and 75 years or older. Patients younger than 75 years who were taking DPP-4 inhibitors had an increased risk of developing BP (aOR, 1.76; 95% CI, 1.22-2.55; P = .003). The use of vildagliptin (aOR, 2.03; 95% CI, 1.24-3.32; P = .005) and sitagliptin (aOR, 1.80; 95% CI, 1.04-3.13; P = .04) were significantly associated with development of BP. Older patients (≥75 years of age) also had an increased risk of BP (aOR, 1.50; 95% CI, 1.11-2.04; P = .008) (Table 4).
Table 4. Subgroup Analysis of the Association Between Use of DPP-4 Inhibitors and the Development of Bullous Pemphigoid in Patients With Diabetes .
| Variable | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Crude OR (95% CI)a | P Value | Adjusted OR (95% CI)b | P Value | |
| Age <75 y | ||||
| All DPP-4 inhibitors | 1.75 (1.22-2.52) | .003 | 1.76 (1.22-2.55) | .003 |
| Vildagliptin | 2.07 (1.27-3.37) | .003 | 2.03 (1.24-3.32) | .005 |
| Sitagliptin | 1.76 (1.02-3.04) | .04 | 1.80 (1.04-3.13) | .04 |
| Linagliptin | 1.46 (0.82-2.59) | .20 | 1.47 (0.82-2.62) | .20 |
| Others | 4.14 (1.15-14.82) | .03 | 3.78 (1.05-13.70) | .04 |
| Age ≥75 y | ||||
| All DPP-4 inhibitors | 1.53 (1.14-2.06) | .005 | 1.50 (1.11-2.04) | .008 |
| Vildagliptin | 1.68 (1.10-2.57) | .02 | 1.68 (1.09-2.60) | .02 |
| Sitagliptin | 1.63 (1.03-2.58) | .04 | 1.67 (1.04-2.67) | .03 |
| Linagliptin | 1.75 (1.13-2.72) | .01 | 1.75 (1.12-2.75) | .02 |
| Others | 1.00 (0.54-1.84) | >.99 | 0.93 (0.50-1.74) | .83 |
| Male | ||||
| All DPP-4 inhibitors | 1.87 (1.36-2.57) | <.001 | 1.91 (1.39-2.63) | <.001 |
| Vildagliptin | 2.68 (1.71-4.18) | <.001 | 2.70 (1.73-4.34) | <.001 |
| Sitagliptin | 1.54 (0.96-2.49) | .07 | 1.62 (1.00-2.62) | .05 |
| Linagliptin | 1.68 (1.07-2.65) | .03 | 1.70 (1.07-2.69) | .02 |
| Others | 1.35 (0.56-3.24) | .51 | 1.32 (0.54-3.20) | .55 |
| Female | ||||
| All DPP-4 inhibitors | 1.37 (0.98-1.91) | .06 | 1.24 (0.88-1.75) | .21 |
| Vildagliptin | 1.19 (0.74-1.90) | .47 | 1.09 (0.67-1.76) | .74 |
| Sitagliptin | 1.86 (1.10-3.15) | .02 | 1.76 (1.03-3.01) | .04 |
| Linagliptin | 1.58 (0.91-2.75) | .10 | 1.48 (0.84-2.60) | .18 |
| Others | 1.40 (0.72-2.72) | .32 | 1.26 (0.64-2.48) | .50 |
Abbreviations: DPP-4, dipeptidyl peptidase 4; OR, odds ratio.
The ORs and 95% CIs were estimated using univariate logistic regression analysis.
The ORs and 95% CIs were estimated using multivariate logistic regression analysis adjusted for neurologic, malignant, and psychological comorbid disorders and receipt of spironolactone and psycholeptics.
Subgroup Analysis According to Sex
In male patients with BP and diabetes, we found a significant association between the use of DPP-4 inhibitors and BP (aOR, 1.91; 95% CI, 1.39-2.63; P < .001). Of the DPP-4 inhibitors available in Korea, use of vildagliptin had the greatest association with the development of BP (aOR, 2.70; 95% CI, 1.73-4.34; P < .001). No association was found between the use of DPP-4 inhibitors and increased risk of BP in female patients with BP and diabetes (Table 4).
Discussion
We performed a retrospective, population-based study of 670 patients with BP and diabetes and 670 controls using Korean insurance claims data to evaluate the association between DPP-4 inhibitors and the development of BP in patients with diabetes. Our study suggests that use of DPP-4 inhibitors is associated with an increased risk of developing BP (aOR, 1.58; 95% CI, 1.25-2.00). Vildagliptin, sitagliptin, and linagliptin are the most commonly prescribed DPP-4 inhibitors in Korea, and each was significantly associated with the development of BP. Recent studies11,13,14 have implicated DPP-4 inhibitors in the development of BP. Varpuluoma et al13 suggested that vildagliptin was significantly associated with increased risk of BP (aOR, 10.40; 95% CI, 4.56-23.80). Benzaquen et al11 reported that the use of DPP-4 inhibitors was associated with an increased risk for the development of BP (aOR, 2.64); vildagliptin yielded the highest risk (aOR, 3.57 [in 61 patients with BP and diabetes]). Sitagliptin and linagliptin were not associated with the development of BP in this study. In a recent study14 of 36 patients with BP, intake of DPP-4 inhibitors was associated with a 3-fold increased risk of BP (aOR, 3.2). In addition, that study reported that use of vildagliptin (n = 24) and linagliptin (n = 9) was associated with an increased risk of BP (aORs, 10.7 for vildagliptin and 6.7 for linagliptin). The use of sitagliptin (n = 6) was independent of the development of BP. Given the relatively small sample size, that study was underpowered to validate that vildagliptin and linagliptin are associated with the development of BP. Owing to a larger sample size, in the present study, the risk associated with each DPP-4 inhibitor could be assessed. Our study found that vildaglitin has a greater association with the development of BP compared with other DPP-4 inhibitors (aOR, 1.81; 95% CI, 1.31-2.50); however, the risks associated with specific DPP-4 inhibitors was lower in our study than what has been reported previously.11,13 Use of sitagliptin and linagliptin was associated with the development of BP (aORs, 1.70 [95% CI, 1.19-2.43] for sitagliptin and 1.64 [95% CI, 1.15-2.33] for linagliptin).
Subgroup analyses conducted in this study revealed that use of DPP-4 inhibitors was associated with an increased risk of developing BP in male but not female patients with diabetes. In particular, the risk associated with vildagliptin prescription was higher in male patients (aOR, 2.70; 95% CI, 1.73-4.34). In female patients, we found that use of DPP-4 inhibitor was not a significant risk factor for the development of BP. Similar to the results reported herein, a study11 of 61 patients also indicated that the association between DPP-4 inhibitors and increased risk of developing BP was observed only in male patients. Kridin and Bergman14 also reported that the association of DPP-4 inhibitors with BP was stronger among male patients.
According to the age-stratified subgroup analysis, we found that patients who received DPP-4 inhibitors were at a significantly increased risk of developing BP compared with control patients regardless of age, and the risk was slightly higher in those younger than 75 years. One previous study11 suggested that older age could be a risk factor for developing BP, but this finding was not evident in our study. In contrast, Kridin and Bergman14 suggested that the strongest association was observed in patients younger than 70 years. Recent studies10,14 have reported that male patients with diabetes commonly have a high risk of BP, but the association between age and increased risk of BP indicates the conflicting results. To validate these results, further large-scale cohort studies in various regions will be needed. Of interest, we observed that the risk of BP was high in patients younger than 75 years who received DPP-4 inhibitors, including saxagliptin, teneligliptin, gemigliptin, and alogliptin, but the small sample sizes make generating conclusive claims difficult.
Recent studies15,16 indicate that the annual number of patients with BP and diabetes and the prevalence of diabetes in patients newly diagnosed with BP have increased since 2010. Our study also found that the proportion of patients with diabetes among all patients with BP has increased since 2012. According to these results, we hypothesized that the increased proportion of diabetes in patients with BP might be attributable to the use of DPP-4 inhibitors. Several previous epidemiologic studies17,18,19 have found that BP was more common in females. Our study also found similar results, and the proportion of males to females was 0.78:1. The proportion of male to female patients with diabetes and BP was 1.04:1. In the European pharmacovigilance database, the development of BP in patients with diabetes as a result of a drug adverse effect was more common in men.20 There is a difference in sex distribution between total patients with BP and patients with BP and diabetes; these differences may be attributable to the increased risk of developing BP in male patients who received DPP-4 inhibitors, particularly vildagliptin. It is not clear why the use of DPP-4 inhibitors was associated with an increased risk of developing BP in only male patients.
The exact pathogenesis of how DPP-4 inhibitors might cause BP remains unclear. DPP-4 inhibitors may alter the antigenic properties of the epidermal basement membrane and lead to modifications of the immune response in genetically predisposed patients.21,22 Among the DPP-4 inhibitors available in Korea, vildagliptin use was associated with the greatest risk for the development of BP. The DPP family includes not only DPP-4 but also the structurally similar DPP-2, DPP-8, DPP-9, and fibroblast activation protein.23 Most DPP-4 inhibitors have high DPP-4 selectivity, but there are differences among different drugs of this class. Vildagliptin, which has been reported to have lower selectivity for DPP-4 than linagliptin and sitagliptin, significantly decreased DPP-8 and DPP-9 activity in an ex vivo study.24,25 The exact function of DPP-8 and DPP-9 is unknown, but DPP-8 and DPP-9 are considered to be a new checkpoints that hold procaspase-1. DPP-8 and DPP-9 inhibition results in the conversion of procaspase-1 into an activated form of procaspase-1.23,26 Remarkable upregulation of the nucleotide-binding domain, leucine-rich family protein 3–caspase-1–IL-18 axis was found in patients with BP and has been positively correlated with disease activity.27 We hypothesized that the inflammasome–caspase-1 pathway was associated with the BP pathogenesis, namely, that unwanted DPP-8 and DPP-9 inhibition induced activation of the caspase-1 pathway because of low selectivity of vildagliptin, which may contribute to an increased risk of developing BP.
Strengths and Limitations
The main strength of our study is that patient information, including all drug prescription details, was obtained from the Korean insurance claims data. Our study assessed the association of use of DPP-4 inhibitors with the risk of developing BP based on a nationwide cohort with BP and is one of the largest cohort studies of this condition to date. There are some limitations to the present study. First, we could not confirm the actual onset of the BP symptoms and the duration of use of DPP-4 inhibitors before the onset of BP. Second, DPP-4 inhibitors are frequently prescribed in combination with metformin; however, we did not consider the effect of metformin because it has not been implicated in the development of BP.14 Recent studies14,28 also suggest that antidiabetic agents other than DPP-4 inhibitors are not associated with BP and the association of DPP-4 inhibitors with BP is independent of the use of metformin.
Conclusions
Our nationwide, population-based study revealed an increased proportion of patients with diabetes among patients with BP and that DPP-4 inhibitors were associated with a significantly increased risk of the development of BP in patients with diabetes. Vildagliptin use had the greatest association with the development of BP compared with other DPP-4 inhibitors. The findings suggest that use of DPP-4 inhibitors is associated with the development of BP in patients with diabetes, particularly the use of vildagliptin in male patients. Further studies are required to understand the exact pathogenesis and further characterize the association between DPP-4 inhibitors and the development of BP.
References
- 1.Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381(9863):320-332. doi: 10.1016/S0140-6736(12)61140-4 [DOI] [PubMed] [Google Scholar]
- 2.Kasperkiewicz M, Zillikens D. The pathophysiology of bullous pemphigoid. Clin Rev Allergy Immunol. 2007;33(1-2):67-77. doi: 10.1007/s12016-007-0030-y [DOI] [PubMed] [Google Scholar]
- 3.Lai YC, Yew YW, Lambert WC. Bullous pemphigoid and its association with neurological diseases: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2016;30(12):2007-2015. doi: 10.1111/jdv.13660 [DOI] [PubMed] [Google Scholar]
- 4.Bastuji-Garin S, Joly P, Lemordant P, et al. ; French Study Group for Bullous Diseases . Risk factors for bullous pemphigoid in the elderly: a prospective case-control study. J Invest Dermatol. 2011;131(3):637-643. doi: 10.1038/jid.2010.301 [DOI] [PubMed] [Google Scholar]
- 5.Jeon HW, Yun SJ, Lee SC, Won YH, Lee JB. Mortality and comorbidity profiles of patients with bullous pemphigoid in Korea. Ann Dermatol. 2018;30(1):13-19. doi: 10.5021/ad.2018.30.1.13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chan LS, Vanderlugt CJ, Hashimoto T, et al. Epitope spreading: lessons from autoimmune skin diseases. J Invest Dermatol. 1998;110(2):103-109. doi: 10.1046/j.1523-1747.1998.00107.x [DOI] [PubMed] [Google Scholar]
- 7.Thornberry NA, Gallwitz B. Mechanism of action of inhibitors of dipeptidyl-peptidase-4 (DPP-4). Best Pract Res Clin Endocrinol Metab. 2009;23(4):479-486. doi: 10.1016/j.beem.2009.03.004 [DOI] [PubMed] [Google Scholar]
- 8.Muscelli E, Casolaro A, Gastaldelli A, et al. Mechanisms for the antihyperglycemic effect of sitagliptin in patients with type 2 diabetes. J Clin Endocrinol Metab. 2012;97(8):2818-2826. doi: 10.1210/jc.2012-1205 [DOI] [PubMed] [Google Scholar]
- 9.Béné J, Moulis G, Bennani I, et al. ; French Association of Regional PharmacoVigilance Centres . Bullous pemphigoid and dipeptidyl peptidase IV inhibitors: a case-noncase study in the French Pharmacovigilance Database. Br J Dermatol. 2016;175(2):296-301. doi: 10.1111/bjd.14601 [DOI] [PubMed] [Google Scholar]
- 10.García-Díez I, Ivars-Lleó M, López-Aventín D, et al. Bullous pemphigoid induced by dipeptidyl peptidase-4 inhibitors: eight cases with clinical and immunological characterization. Int J Dermatol. 2018;57(7):810-816. doi: 10.1111/ijd.14005 [DOI] [PubMed] [Google Scholar]
- 11.Benzaquen M, Borradori L, Berbis P, et al. Dipeptidyl peptidase IV inhibitors, a risk factor for bullous pemphigoid: retrospective multicenter case-control study from France and Switzerland. J Am Acad Dermatol. 2018;78(6):1090-1096. doi: 10.1016/j.jaad.2017.12.038 [DOI] [PubMed] [Google Scholar]
- 12.Yoshiji S, Murakami T, Harashima SI, et al. Bullous pemphigoid associated with dipeptidyl peptidase-4 inhibitors: a report of five cases. J Diabetes Investig. 2018;9(2):445-447. doi: 10.1111/jdi.12695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Varpuluoma O, Försti AK, Jokelainen J, et al. Vildagliptin significantly increases the risk of bullous pemphigoid: a Finnish nationwide registry study. J Invest Dermatol. 2018;138(7):1659-1661. doi: 10.1016/j.jid.2018.01.027 [DOI] [PubMed] [Google Scholar]
- 14.Kridin K, Bergman R. Association of bullous pemphigoid with dipeptidyl-peptidase 4 inhibitors in patients with diabetes: estimating the risk of the new agents and characterizing the patients. JAMA Dermatol. 2018;154(10):1152-1158. doi: 10.1001/jamadermatol.2018.2352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fania L, Di Zenzo G, Didona B, et al. Increased prevalence of diabetes mellitus in bullous pemphigoid patients during the last decade. J Eur Acad Dermatol Venereol. 2018;32(4):e153-e154. doi: 10.1111/jdv.14649 [DOI] [PubMed] [Google Scholar]
- 16.Gravani A, Gaitanis G, Tsironi T, Tigas S, Bassukas ID. Changing prevalence of diabetes mellitus in bullous pemphigoid: it is the dipeptidyl peptidase-4 inhibitors [published online March 23, 2018]. J Eur Acad Dermatol Venereol. [DOI] [PubMed] [Google Scholar]
- 17.Thorslund K, Seifert O, Nilzén K, Grönhagen C. Incidence of bullous pemphigoid in Sweden 2005-2012: a nationwide population-based cohort study of 3761 patients. Arch Dermatol Res. 2017;309(9):721-727. doi: 10.1007/s00403-017-1778-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kridin K, Bergman R. Ethnic variations in the epidemiology of bullous pemphigoid in Israel. Int J Dermatol. 2018;57(1):34-39. doi: 10.1111/ijd.13813 [DOI] [PubMed] [Google Scholar]
- 19.Hübner F, Recke A, Zillikens D, Linder R, Schmidt E. Prevalence and age distribution of pemphigus and pemphigoid diseases in Germany. J Invest Dermatol. 2016;136(12):2495-2498. doi: 10.1016/j.jid.2016.07.013 [DOI] [PubMed] [Google Scholar]
- 20.García M, Aranburu MA, Palacios-Zabalza I, Lertxundi U, Aguirre C. Dipeptidyl peptidase-IV inhibitors induced bullous pemphigoid: a case report and analysis of cases reported in the European pharmacovigilance database. J Clin Pharm Ther. 2016;41(3):368-370. doi: 10.1111/jcpt.12397 [DOI] [PubMed] [Google Scholar]
- 21.Skandalis K, Spirova M, Gaitanis G, Tsartsarakis A, Bassukas ID. Drug-induced bullous pemphigoid in diabetes mellitus patients receiving dipeptidyl peptidase-IV inhibitors plus metformin. J Eur Acad Dermatol Venereol. 2012;26(2):249-253. doi: 10.1111/j.1468-3083.2011.04062.x [DOI] [PubMed] [Google Scholar]
- 22.Forssmann U, Stoetzer C, Stephan M, et al. Inhibition of CD26/dipeptidyl peptidase IV enhances CCL11/eotaxin-mediated recruitment of eosinophils in vivo. J Immunol. 2008;181(2):1120-1127. doi: 10.4049/jimmunol.181.2.1120 [DOI] [PubMed] [Google Scholar]
- 23.Waumans Y, Baerts L, Kehoe K, Lambeir AM, De Meester I. The dipeptidyl peptidase family, prolyl oligopeptidase, and prolyl carboxypeptidase in the immune system and inflammatory disease, including atherosclerosis. Front Immunol. 2015;6:387. doi: 10.3389/fimmu.2015.00387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tatosian DA, Guo Y, Schaeffer AK, et al. Dipeptidyl peptidase-4 inhibition in patients with type 2 diabetes treated with saxagliptin, sitagliptin, or vildagliptin. Diabetes Ther. 2013;4(2):431-442. doi: 10.1007/s13300-013-0045-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Huan Y, Jiang Q, Liu JL, Shen ZF. Establishment of a dipeptidyl peptidases (DPP) 8/9 expressing cell model for evaluating the selectivity of DPP4 inhibitors. J Pharmacol Toxicol Methods. 2015;71:8-12. doi: 10.1016/j.vascn.2014.11.002 [DOI] [PubMed] [Google Scholar]
- 26.Okondo MC, Johnson DC, Sridharan R, et al. DPP8 and DPP9 inhibition induces pro-caspase-1-dependent monocyte and macrophage pyroptosis. Nat Chem Biol. 2017;13(1):46-53. doi: 10.1038/nchembio.2229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fang H, Shao S, Cao T, et al. Increased expression of NLRP3 inflammasome components and interleukin-18 in patients with bullous pemphigoid. J Dermatol Sci. 2016;83(2):116-123. doi: 10.1016/j.jdermsci.2016.04.009 [DOI] [PubMed] [Google Scholar]
- 28.Varpuluoma O, Försti AK, Jokelainen J, et al. Oral diabetes medications other than dipeptidyl peptidase 4 inhibitors are not associated with bullous pemphigoid: a Finnish nationwide case-control study. J Am Acad Dermatol. 2018;79(6):1034-1038.e5. doi: 10.1016/j.jaad.2018.05.030 [DOI] [PubMed] [Google Scholar]