Key Points
Question
What is the source of errors in the initial misdiagnosis of optic nerve sheath meningiomas?
Findings
In this medical record review that included 35 patients with optic nerve sheath meningiomas, 25 initially received a misdiagnosis that resulted in diagnostic delay, most frequently due to clinician assessment failures and errors in diagnostic testing.
Meaning
Education regarding diagnostic strategies and proper neuroimaging for optic neuropathies may prevent vision loss from the initial misdiagnosis of optic nerve sheath meningiomas.
Abstract
Importance
Diagnostic errors can lead to the initial misdiagnosis of optic nerve sheath meningiomas (ONSM), which can lead to vision loss.
Objective
To identify factors contributing to the initial misdiagnosis of ONSM.
Design, Setting, and Participants
We retrospectively reviewed 35 of 39 patients with unilateral ONSM (89.7%) who were seen in the tertiary neuro-ophthalmology practice at Emory University School of Medicine between January 2002 and March 2017. The Diagnosis Error Evaluation and Research taxonomy tool was applied to cases with missed/delayed diagnoses.
Exposures
Evaluation in a neuro-ophthalmology clinic.
Main Outcomes and Measures
Identifying the cause of diagnostic errors for patients who initially received a misdiagnosis who were found to have ONSM.
Results
Of 35 patients with unilateral ONSM (30 women [85.7%]; mean [SD] age, 45.26 [15.73] years), 25 (71%) had a diagnosis delayed for a mean (SD) of 62.60 (89.26) months. The most common diagnostic error (19 of 25 [76%]) was clinician assessment failure (errors in hypothesis generation and weighing), followed by errors in diagnostic testing (15 of 25 [60%]). The most common initial misdiagnosis was optic neuritis (12 of 25 [48%]), followed by the failure to recognize optic neuropathy in patients with ocular disorders. Five patients who received a misdiagnosis (20%) underwent unnecessary lumbar puncture, 12 patients (48%) unnecessary laboratory tests, and 6 patients (24%) unnecessary steroid treatment. Among the 16 patients who initially received a misdiagnosis that was later correctly diagnosed at our institution, 11 (68.8%) had prior magnetic resonance imaging (MRI) results that were read as healthy; 5 (45.5%) showed ONSM but were misread by a non-neuroradiologist and 6 (54.5%) were performed incorrectly (no orbital sequence or contrast). Sixteen of the 25 patients (64%) had a poor visual outcome.
Conclusions and Relevance
Biased preestablished diagnoses, inaccurate funduscopic examinations, a failure to order the correct test (MRI brain/orbits with contrast), and a failure to correctly interpret MRI results were the most common sources of diagnostic errors and delayed diagnosis with worse visual outcomes and increased cost (more visits and tests). Easier access to neuro-ophthalmologists, improved diagnostic strategies, and education regarding neuroimaging should help prevent diagnostic errors.
This medical record review identifies factors and diagnostic errors that contribute to the initial misdiagnosis of optic nerve sheath meningiomas.
Introduction
Optic nerve sheath meningiomas (ONSMs) are rare tumors of the orbit and account for 1% to 2% of all meningiomas. However, they represent the second most common tumor of the optic nerve after gliomas, comprising about one-third of all primary optic nerve tumors.1,2 Most ONSMs are secondary tumors that extend from intracranial sites, whereas primary ONSMs arise from the intraorbital, or less commonly, intracanalicular dural sheath.2,3 Like all meningiomas, ONSMs usually affect middle-aged women, with a mean age at presentation of 40.8 years.1 Diagnosis is based on the characteristic clinical and imaging findings. Biopsies and surgical resection are associated with significant visual morbidity and therefore are usually avoided.3,4
The classic clinical presentation of ONSM is the triad of progressive visual loss, optic atrophy, and the presence of retinal-choroidal collaterals.3 However, the simultaneous occurrence of all 3 findings is rare. Patients usually present with painless progressive monocular visual loss, which may lead to blindness if left untreated. Therefore, although ONSMs are histologically benign and relatively rare tumors, timely diagnosis and appropriate management are crucial to ensure a favorable visual outcome. Early intervention with stereotactic fractionated radiation therapy is shown to be effective for preserving and even improving visual function.3,5,6,7,8,9 We sought to identify the factors that contribute to the initial misdiagnosis or delayed diagnosis of ONSM to provide strategies to avoid diagnostic errors and increase the likelihood of a favorable visual outcome.
Methods
This study was reviewed and approved by the Emory University School of Medicine institutional review board and informed consent was waived. We performed a retrospective review of all cases with ONSM seen in one neuro-ophthalmology clinic of a tertiary health care institution from January 2002 to March 2017.
We identified patients with primary ONSM using the electronic database of neuro-ophthalmology, neuroradiology, and radiation oncology departments at our institution. We then reviewed the electronic and paper medical records of patients with the final diagnosis of ONSM who were evaluated in the neuro-ophthalmology service.
We included all patients who were 16 years or older who were referred to us with the prior diagnosis of ONSM or who received a diagnosis of ONSM in the service. The diagnosis of ONSM was determined by experienced neuro-ophthalmologists (V.B., N.J.N.) on the basis of characteristic clinical symptoms and signs and typical magnetic resonance imaging (MRI) findings as interpreted by expert neuroradiologists, neurosurgeons, and radiation oncologists at our institution.2,3 All patients had full ocular and neuro-ophthalmic evaluations, visual field testing, and orbital and brain MRI with and without contrast during follow-up at our institution. Some patients also had a histopathological confirmation of ONSM. Primary intracranial meningiomas, other causes of optic neuropathy, and cases with uncertain diagnoses were excluded.
Demographic data including age, race/ethnicity (self-reported by patients), sex, and body mass index (calculated as weight in kilograms divided by height in meters squared) were recorded. Relevant information about patient history, examination findings, prior diagnostic tests and their reports, initial diagnoses, and treatments were obtained. The time to the final diagnosis and detailed referral data, including the specialties of all physicians who were seen before referral, were also retrieved from the medical records. We inquired if the brain MRI was ordered correctly (with and without contrast and including orbital views with fat suppression) and whether the interpreting radiologist was trained in neuroradiology.
For patients whose final diagnoses of ONSM were missed or delayed, the Diagnosis Error Evaluation and Research (DEER) taxonomy tool (Table 1) was applied and each case was classified according to the location and type of error in the diagnostic process.10 As we were dealing primarily with errors in the diagnostic assessment, we specifically concentrated on steps 2 through 6, which were directly associated with the diagnostic process. When there were multiple breakdowns in the diagnostic process, the factor that was considered to make the largest contribution to the misdiagnosis was assigned a primary DEER category whereas other errors were categorized as secondary or tertiary.11
Table 1. Diagnosis Error Evaluation and Research Taxonomy.
| Point in the Diagnostic Process (Anatomic Localization) | What Went Wrong (Lesion) | No. (%) | |
|---|---|---|---|
| Patients in Each Category (Primary/Major Diagnostic Error) | Frequency of Patients With a Misdiagnosis in Each Category | ||
| Access/presentation | |||
| A | Failure/delay in presentation | ||
| B | Failure/denied care access | ||
| History | 5 (20) | 6 (24) | |
| A | Failure/delay in eliciting critical piece of history data | ||
| B | Inaccurate/misinterpretation | 1 | |
| C | Failure in weighing | 4 | 5 |
| D | Failure/delay to follow-up | ||
| Physical examination | 6 (24) | 7 (28) | |
| A | Failure/delay in eliciting critical examination finding | 2 | |
| B | Inaccurate/misinterpreted | 2 | 4 |
| C | Failure in weighing | 2 | |
| D | Failure/delay to follow-up | ||
| Tests (laboratory/radiology) | 5 (20) | 15 (60) | |
| Ordering | |||
| A | Failure/delay in ordering needed test(s) | 1 | 3 |
| B | Failure/delay in performing ordered test(s) | ||
| C | Error in test sequencing | ||
| D | Ordering of the wrong test(s) | ||
| E | Test ordered the wrong way | 2 | 4 |
| Performance | |||
| F | Sample mix-up/mislabeled (eg, wrong patient/test) | ||
| G | Technical error/poor processing of specimen/test | ||
| H | Erroneous laboratory/radiology reading of test | 2 | 7 |
| I | Failed/delayed reporting of the result to the clinician | ||
| Clinician processing | |||
| J | Failed/delayed follow-up (abnormal) test result | 2 | |
| K | Error in clinician interpretation of test | ||
| Assessment | 9 (36) | 19 (76) | |
| Hypothesis generation | |||
| A | Failure/delay in considering the correct diagnosis | 3 | 7 |
| Suboptimal weighing/prioritizing | |||
| B | Too little consideration/weight given to the diagnosis | 1 | 3 |
| C | Too much weight on competing/coexisting diagnosis | 5 | 9 |
| Recognizing urgency/complications | |||
| D | Failure/delay to recognize/weigh urgency | ||
| E | Failure/delay to recognize/weigh complication(s) | ||
| Referral/consultation | 4 (16) | ||
| A | Failure in ordering referral | ||
| B | Inappropriate/unneeded referral | 4 | |
| C | Error in diagnostic consultation performance | ||
| D | Failed/delayed communication/follow-up of consultation | ||
| Follow-up | |||
| A | Failure to refer patient to close/safe setting/monitoring | ||
| B | Failure/delay in timely follow-up/rechecking of patient | ||
Statistical Analysis
To compare continuous variables, the Mann-Whitney U test was used. For categorical variable comparisons, we used the Fisher exact test.
Results
A total of 47 patients with the final diagnosis of primary ONSM were identified from the database of our institution. After excluding 8 patients who were not seen in the neuro-ophthalmology clinic, the medical records of 39 patients were reviewed. Because our primary concern was to assess the steps of the evolving diagnostic process, 2 patients with poor previous outside records and 1 patient who received an incidental diagnosis from a brain scan that was performed for screening for breast cancer metastases were excluded. One patient with atypical features and an uncertain diagnosis was also excluded.
The remaining 35 eligible cases were systematically analyzed and classified into 3 groups based on the diagnosis pattern. Group 1 consisted of 9 patients (25.7%) whose diagnoses were made by the referring physician (not missed); group 2 included 9 patients (25.7%) whose diagnoses were made by the referring physician after being missed by someone else; and group 3 included 17 patients (48.6%) who received their diagnoses at our institution. Group 3 was further subdivided into 1 group with a diagnosis missed before referral to our institution (group 3a) and another group of patients who had been referred with no previous workup (not missed) (group 3b) (Figure 1).
Figure 1. Classification of Patients According to the Diagnosis Pattern.
Patients were seen between 2002 and 2017. ONSM indicates optic nerve sheath meningioma.
White middle-aged women predominated with a female-to-male ratio of 6:1 (30 women [85.7%], 5 men [14.3%]) and a mean age of 45.26 years (range, 16-72 years) at the time of diagnosis. All patients had a unilateral ONSM (17 [48.6%] right, 18 [51.4%] left). Decreased vision in 1 eye was the most common presenting symptom (30 patients [85.7%]), followed by eye pain or pressure sensation behind the eye (4 patients [11.4%]) and binocular diplopia (1 patient [2.8%]).
Most patients were referred by ophthalmologists (19 patients [54.3%]). Four patients (11.4%), 2 of whom received a misdiagnosis, were referred by neuro-ophthalmologists. Seventeen patients (48.6%) eventually received their diagnoses at our institution, 7 patients (20.0%) received their diagnoses from outside neuro-ophthalmologists, 6 patients (17.1%) by ophthalmologists, and 2 (5.7%) by emergency medicine clinicians.
A total of 25 patients (71.4%) were found to have delayed or missed diagnoses, experiencing at least 1 error at some step of the diagnostic assessment process (the sum of groups 2 and 3b). They received a misdiagnosis either from the referring physician or before referral. Ten patients (28.5%) were determined to be assessed appropriately without any diagnostic error and received an accurate diagnosis (groups 1 and 3a).
The DEER taxonomy tool was applied to each of the 25 patients with delayed or missed diagnoses (Table 1). Clinician assessment failures (errors in hypothesis generation and weighing) were determined to account for most of the major diagnostic errors, being primarily responsible for the misdiagnosis in 9 of the 25 cases (36%). Six patients (24%) had a delayed or missed diagnosis primarily because of a failure in eliciting or a misinterpretation of a physical examination finding (fundus examination), whereas 5 cases (20%) were attributable to a failure in ordering or interpreting appropriate radiologic tests and another 5 (20%) were due to a poor history (especially a failure in weighing, such as overreliance on a coincidental event in cases with trauma history or misinterpreting eye pressure sensation as eye pain and assuming inflammatory optic neuritis).
Eighteen cases (51.4%) involved more than 1 diagnostic error that contributed directly to the diagnostic failure. When all these secondary and tertiary errors were considered, patients were found to experience a total of 53 diagnostic errors, most of which (19 of 53 [35.8%]) were associated with a failure in clinician assessment. Diagnostic errors that were directly linked to each other were not considered separate errors. For example, the failure to detect the lesion on MRI because of inappropriate imaging (eg, brain MRI without contrast and orbital images) was assigned as “wrongly ordered test (4E in Table 1),” not “erroneous reading of test (4H in Table 1).” Overall, 19 of the 25 misdiagnosed cases (76%) experienced clinician assessment failure, 15 cases (60%) were subject to errors in diagnostic testing, 7 cases (28%) involved a failure in administering an appropriate physical examination, 6 cases (24%) involved errors in history taking and interpretation, and 4 patients (16%) had inappropriate referrals that eventually contributed to the delayed or missed diagnosis (Table 1). In subgroup analysis of the errors, weighing the competing or coexisting diagnosis was the most common diagnostic error, which occurred in nearly half (12 of 25 [48%]) of the cases. Clinicians frequently misdiagnosed the cases because of overweighing coexisting ocular diseases, such as cataract, glaucoma, or retinal disease, without further evaluating the patients to rule out compressive optic neuropathies. They also had the tendency to diagnose optic neuritis in young to middle-aged women. In this group of 25 patients, optic neuritis was the most common misdiagnosis, followed by retinal macular disease (Table 2). Nearly half of the patients (12 of 25 [48%]) received a misdiagnosis of optic neuritis at some step during the diagnostic process.
Table 2. Misdiagnoses in 25 Patients.
| Misdiagnosis | No. of Patients (n = 25) |
|---|---|
| Optic neuritis | 12 |
| Retinal/macular disease | 5 |
| Cataract | 4 |
| Traumatic optic neuropathy | 3 |
| Refractive error | 2 |
| Ischemic optic neuropathy | 2 |
| Glaucoma | 2 |
| Optic nerve drusen | 2 |
| Idiopathic intracranial hypertension | 2 |
| Amblyopia | 2 |
| Oculomotor palsy | 1 |
| Allergy | 1 |
Of the 16 patients with a misdiagnosis who eventually received a diagnosis at our institution, 10 (62.5%) had been referred by ophthalmologists, 2 (12.5%) by neurologists, and 2 (12.5%) by neuro-ophthalmologists. Five (31.3%) of these patients had no MRI results, whereas the remaining 11 (68.7%) had their MRI results misread. Five (45.5%) of the misread MRIs were misinterpreted by radiologists who were not trained in neuroradiology and 6 (54.5%) were misinterpreted because the test was ordered incorrectly (not including orbital images and contrast administration).
In the total misdiagnosed group, 5 patients (20%) underwent unnecessary lumbar puncture. One patient had 2 lumbar punctures. Twelve patients (48%) had unnecessary extensive laboratory tests. Six patients (24%) had been treated with steroids (1 for presumed temporal arteritis and 5 for presumed optic neuritis), 1 of whom developed steroid-induced diabetes. One patient had retinal laser treatment because of a misdiagnosis of retinal disease.
When the patients with and without diagnostic errors were compared, patients with a misdiagnosis when compared with the accurately diagnosed group (Table 3) tended to be younger at symptom onset and the time of diagnosis (mean [SD] age at symptom onset, 38.2 [18.38] vs 49 [12.0] years; mean [SD] age at diagnosis, 43.6 [17.03] vs 49.2 [11.70] years) and had a longer time from symptom onset to the definitive diagnosis (mean time, 62.6 months vs 5.7 months). Patients with a misdiagnosis saw more clinicians (mean, 2.9 [range, 1-6]) and attended more office visits (mean, 4.31 [range, 1-11]) than patients who received a correct diagnosis of ONSM initially (mean, 1.2 [range, 1-2] for both).
Table 3. Characteristics of Patients With and Without Diagnostic Errors.
| Characteristic | Mean (Range) | P Value | |
|---|---|---|---|
| Misdiagnosed Cases (n = 25) | Accurately Diagnosed Cases (n = 10) | ||
| Age at symptom onset, y | 38.5 (12-72) | 49.0 (29-66) | .11 |
| Age at diagnosis, y | 43.7 (16-73) | 49.2 (30-66) | .35 |
| Time to accurate diagnosis, mo | 62.6 (0.7-348) | 5.7 (0-12) | .06 |
| Specialists seen before diagnosis. No. | 3 (1-6) | 1.5 (1-3) | .01 |
| Visits before diagnosis. No. | 4.31 (1-11) | 1.5 (1-3) | .004 |
| Visual acuity, No./total No. (%) | .13 | ||
| Stable or improved | 9 of 25 (36) | 7 of 10 (70) | |
| Declined | 16 of 25 (64) | 3 of 10 (30) | |
| 7 NLP, 2 HM | No NLP or HM | ||
Abbreviations: HM, hand motion; NLP, no light perception.
Regarding the outcome, 16 of the patients with a misdiagnosis (64%) experienced a deterioration of vision, with 10 (40%) having a very poor visual outcome (visual acuity of no light perception in 7 patients [70%], visual acuity of hand motion in 2 patients [20%], and visual acuity of counting fingers at 3 ft in 1 patient [10%]). Conversely, 3 patients (10%) without diagnostic errors had a deterioration of vision, with the worst final visual acuity being 20/70 in the affected eye. Seven patients (70%) without diagnostic delay had improvement in vision or remained stable (Table 3).
Discussion
This study showed that nearly three-quarters (71.4%) of patients with ONSM had delayed or initially missed diagnoses. This led to unnecessary laboratory tests in nearly half of the patients with a misdiagnosis (48%) and invasive procedures, such as lumbar puncture, in 5 (20%) and, most importantly, resulted in vision loss in 16 patients (64%).
When the DEER taxonomy tool was applied to classify the diagnostic errors, the most common reason for misdiagnosis was found to be clinician assessment failures (errors in hypothesis generation and weighing) followed by a failure in eliciting or misinterpretation of a physical examination finding (fundus examination). Regarding all the secondary and tertiary diagnostic errors, 76% of the misdiagnosed cases experienced clinician assessment failure and 60% of cases were subject to errors in diagnostic testing (neuroimaging).
A recent US National Academy of Medicine report defines a diagnostic error as the failure to establish an accurate and timely explanation of the patient’s health problem(s) or communicate that explanation to the patients.12 It emphasizes that “improving diagnosis is not only possible, but it also represents a moral, professional, and public health imperative.”12 Therefore, it is crucial to identify causes of diagnostic error and define strategies to improve diagnostic accuracy to achieve a better clinical outcome.13,14,15
Diagnostic errors are especially a problem in rare diseases.16,17 These diseases are usually underdiagnosed or mislabeled as a more frequently encountered disease. This heuristic leads to premature closure of thinking.18 Indeed, this study showed that nearly half of the patients with a misdiagnosis received a diagnosis of optic neuritis, a more commonly seen disease. Physicians usually overdiagnose more commonly seen diseases like optic neuritis and miss other optic neuropathies.19 This anchoring resulted in a confirmation bias with a tendency to interpret optic nerve sheath enhancement as optic neuritis and overinterpret white matter hyperintensities as being suggestive of demyelinating disease.
The next most common misdiagnosis was coexisting ocular diseases, such as cataract, glaucoma, or retinal disease. Cases involving these ocular diseases were prematurely closed without being evaluated to rule out compressive optic neuropathies. Clinician assessment failures, meaning the failure to consider or appropriately weigh alternative diagnoses, were the most common diagnostic errors in our study.
In addition to the failure to look for a relative afferent pupillary defect in patients with unilateral visual loss, nearly one-fourth of patients received a misdiagnosis primarily because of a failure to note the correct funduscopic examination findings. Performing ophthalmoscopy and correctly interpreting the findings are still a major clinical challenge.20,21
It is noteworthy that 11 of the 16 patients with a misdiagnosis (68.8%) who eventually received a diagnosis at our institution had their MRI results misinterpreted. Five of these MRIs were misread by radiologists who were not trained in neuroradiology and 6 were misread because the test was ordered incorrectly (not including orbital images and contrast administration). Radiologists have a key role in disease detection (Figure 2).22,23 For many neuro-ophthalmological diseases, neuroimaging is mandatory to accurately diagnose and manage the problem. Most radiological centers are equipped with advanced MRI devices; however, a lack of neuroradiology training among the interpreting radiologists may lead to devastating diagnostic errors.24,25 Inappropriate imaging protocol selection is another factor that leads to misdiagnosis. Our impression is that most clinicians fail to order orbital images and contrast-enhanced sequences for patients who present with progressive visual loss. Additionally, several insurance companies in the United States systematically deny dedicated orbital imaging when ordered in addition to a brain MRI, which contributes to inappropriate imaging techniques.
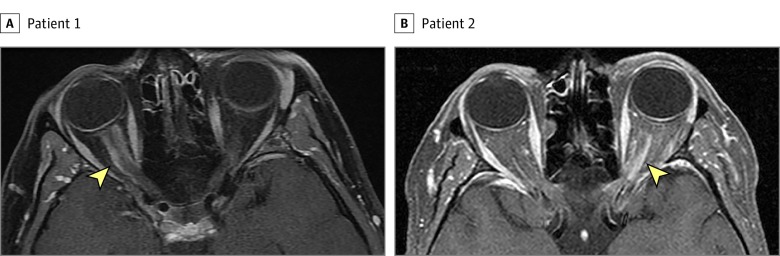
Figure 2. Optic Nerve Sheath Meningioma on Orbital Magnetic Resonance Imaging (MRI).
Axial T1-weighted contrast-enhanced orbital MRI with fat suppression shows an obvious right (A) and left (B) optic nerve sheath enhancement (arrowhead) that led to the correct diagnosis of optic nerve sheath meningioma. The optic nerve itself does not enhance. Both patients previously had a brain MRI without dedicated orbital views on which the optic nerve sheath meningioma was missed.
Limitations
Our study has several limitations, including its retrospective design. However, we had the advantage of gaining access to the previous medical records of the patients, which provided us many details of the diagnostic processes, and we excluded cases with insufficient data. It is also possible that there was some referral bias to our tertiary center, thereby potentially rendering our findings less applicable to the overall misdiagnosis of ONSM.
Conclusions
Considering compressive optic neuropathies in the differential diagnosis of monocular painless progressive vision loss and accordingly ordering the appropriate diagnostic tests are important. Recognizing an optic neuropathy, accurately performing funduscopy, and properly ordering and reviewing neuroimaging will improve the diagnostic process and help avoid unnecessary investigations and delayed treatment.
References
- 1.Dutton JJ. Optic nerve sheath meningiomas. Surv Ophthalmol. 1992;37(3):167-183. doi: 10.1016/0039-6257(92)90135-G [DOI] [PubMed] [Google Scholar]
- 2.Miller NR. New concepts in the diagnosis and management of optic nerve sheath meningioma. J Neuroophthalmol. 2006;26(3):200-208. doi: 10.1097/01.wno.0000235569.19131.ac [DOI] [PubMed] [Google Scholar]
- 3.Shapey J, Sabin HI, Danesh-Meyer HV, Kaye AH. Diagnosis and management of optic nerve sheath meningiomas. J Clin Neurosci. 2013;20(8):1045-1056. doi: 10.1016/j.jocn.2013.03.008 [DOI] [PubMed] [Google Scholar]
- 4.Schick U, Jung C, Hassler WE. Primary optic nerve sheath meningiomas: a follow-up study. Cent Eur Neurosurg. 2010;71(3):126-133. doi: 10.1055/s-0029-1246136 [DOI] [PubMed] [Google Scholar]
- 5.Adams G, Roos DE, Crompton JL. Radiotherapy for optic nerve sheath meningioma: a case for earlier intervention? Clin Oncol (R Coll Radiol). 2013;25(6):356-361. doi: 10.1016/j.clon.2013.02.004 [DOI] [PubMed] [Google Scholar]
- 6.Metellus P, Kapoor S, Kharkar S, et al. Fractionated conformal radiotherapy for management of optic nerve sheath meningiomas: long-term outcomes of tumor control and visual function at a single institution. Int J Radiat Oncol Biol Phys. 2011;80(1):185-192. doi: 10.1016/j.ijrobp.2010.01.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bloch O, Sun M, Kaur G, Barani IJ, Parsa AT. Fractionated radiotherapy for optic nerve sheath meningiomas. J Clin Neurosci. 2012;19(9):1210-1215. doi: 10.1016/j.jocn.2012.02.010 [DOI] [PubMed] [Google Scholar]
- 8.Saeed P, Rootman J, Nugent RA, White VA, Mackenzie IR, Koornneef L. Optic nerve sheath meningiomas. Ophthalmology. 2003;110(10):2019-2030. doi: 10.1016/S0161-6420(03)00787-5 [DOI] [PubMed] [Google Scholar]
- 9.Adeberg S, Welzel T, Rieken S, Debus J, Combs SE. Prior surgical intervention and tumor size impact clinical outcome after precision radiotherapy for the treatment of optic nerve sheath meningiomas (ONSM). Radiat Oncol. 2011;6:117. doi: 10.1186/1748-717X-6-117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schiff GD, Kim S, Abrams R, et al. Diagnosing diagnosis errors: lessons from a multi-institutional collaborative project In: Henriksen K, Battles JB, Marks ES, Lewin DI, eds. Advances in Patient Safety: From Research to Implementation. Rockville, MD: Agency for Healthcare Research and Quality; 2005:255-278. [PubMed] [Google Scholar]
- 11.Schiff GD, Hasan O, Kim S, et al. Diagnostic error in medicine: analysis of 583 physician-reported errors. Arch Intern Med. 2009;169(20):1881-1887. doi: 10.1001/archinternmed.2009.333 [DOI] [PubMed] [Google Scholar]
- 12.Balogh EP, Miller BT, Ball JR, eds. Improving Diagnosis in Health Care. Washington, DC: National Academies Press; 2015. [PubMed] [Google Scholar]
- 13.Bakradze E, Liberman AL. Diagnostic error in stroke—reasons and proposed solutions. Curr Atheroscler Rep. 2018;20(2):11. doi: 10.1007/s11883-018-0712-3 [DOI] [PubMed] [Google Scholar]
- 14.Tarnutzer AA, Lee SH, Robinson KA, Wang Z, Edlow JA, Newman-Toker DE. ED misdiagnosis of cerebrovascular events in the era of modern neuroimaging: a meta-analysis. Neurology. 2017;88(15):1468-1477. doi: 10.1212/WNL.0000000000003814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bhise V, Meyer AND, Singh H, et al. Errors in diagnosis of spinal epidural abscesses in the era of electronic health records. Am J Med. 2017;130(8):975-981. doi: 10.1016/j.amjmed.2017.03.009 [DOI] [PubMed] [Google Scholar]
- 16.Kliegman RM, Bordini BJ, Basel D, Nocton JJ. How doctors think: common diagnostic errors in clinical judgement—lessons form an undiagnosed and rare disease program. Pediatr Clin North Am. 2017;64(1):1-15. doi: 10.1016/j.pcl.2016.08.002 [DOI] [PubMed] [Google Scholar]
- 17.Bordini BJ, Stephany A, Kliegman R. Overcoming diagnostic errors in medical practice. J Pediatr. 2017;185:19-25.e1. doi: 10.1016/j.jpeds.2017.02.065 [DOI] [PubMed] [Google Scholar]
- 18.Hussain A, Oestreicher J. Clinical decision-making: heuristics and cognitive biases for the ophthalmologist. Surv Ophthalmol. 2018;63(1):119-124. doi: 10.1016/j.survophthal.2017.08.007 [DOI] [PubMed] [Google Scholar]
- 19.Stunkel L, Kung NH, Wilson B, McClelland CM, Van Stavern GP. Incidence and causes of overdiagnosis of optic neuritis. JAMA Ophthalmol. 2018;136(1):76-81. doi: 10.1001/jamaophthalmol.2017.5470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fisayo A, Bruce BB, Newman NJ, Biousse V. Overdiagnosis of idiopathic intracranial hypertension. Neurology. 2016;86(4):341-350. doi: 10.1212/WNL.0000000000002318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Biousse V, Bruce BB, Newman NJ. Ophthalmoscopy in the 21st century: The 2017 H. Houston Merritt Lecture. Neurology. 2018;90(4):167-175. doi: 10.1212/WNL.0000000000004868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Siegal D, Stratchko LM, DeRoo C. The role of radiology in diagnostic error: a medical malpractice claims review. Diagnosis (Berl). 2017;4(3):125-131. doi: 10.1515/dx-2017-0025 [DOI] [PubMed] [Google Scholar]
- 23.Allen B, Chatfield M, Burleson J, Thorwarth WT. Improving diagnosis in health care: perspectives from the American College of Radiology. Diagnosis (Berl). 2017;4(3):113-124. doi: 10.1515/dx-2017-0020 [DOI] [PubMed] [Google Scholar]
- 24.Elmalem VI, Hudgins PA, Bruce BB, Newman NJ, Biousse V. Underdiagnosis of posterior communicating artery aneurysm in noninvasive brain vascular studies. J Neuroophthalmol. 2011;31(2):103-109. doi: 10.1097/WNO.0b013e3181f8d985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Briggs GM, Flynn PA, Worthington M, Rennie I, McKinstry CS. The role of specialist neuroradiology second opinion reporting: is there added value? Clin Radiol. 2008;63(7):791-795. doi: 10.1016/j.crad.2007.12.002 [DOI] [PubMed] [Google Scholar]