Abstract
The second half of the 20th century witnessed the emergence and the triumph of cardiac surgery. One of the legends of the period is Sir Magdi H. Yacoub (1935–), pioneer surgeon, scientist, master craftsman, and philanthropist. Yacoub established heart transplantation in the United Kingdom and introduced a variety of new concepts and new operations. His recent work focuses on molecular cardiology. This personal reminiscence highlights his personality, his surgical dexterity, and his many accomplishments and honors.
Keywords: Cardiac surgery, heart transplantation, Magdi H. Yacoub
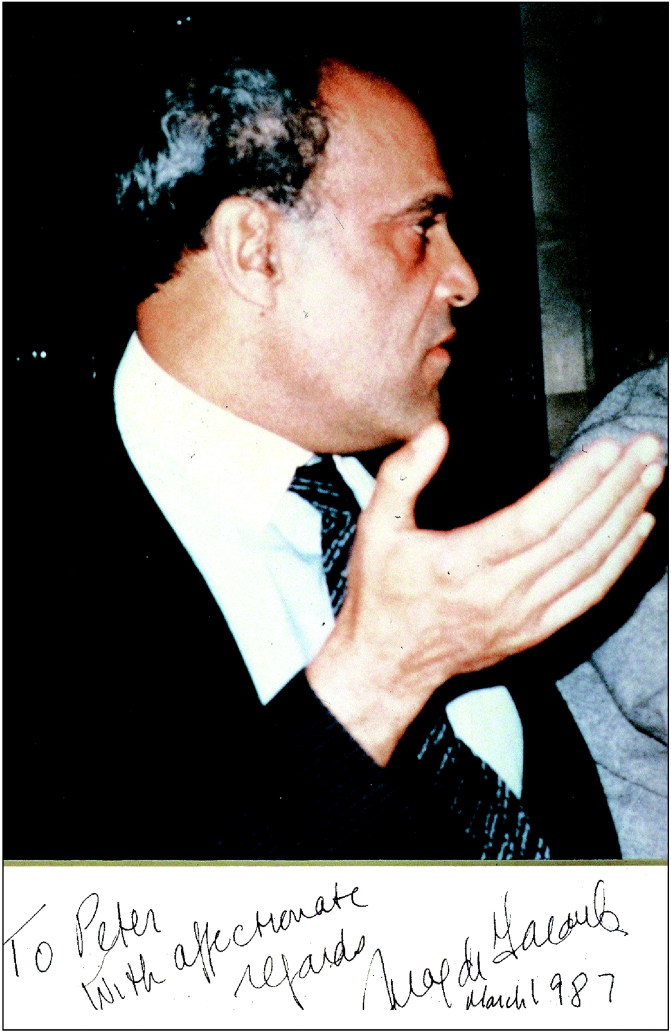
I met Magdi Yacoub (Figure 1) in his private consulting rooms near Harley Street in Central London in late July 1982, after finishing a year of pediatric cardiac surgery at the prestigious Great Ormond Street Hospital for Sick Children. He received me in a simple, friendly manner, sitting behind a large desk devoid of paperwork. Knowing him better later on, I would say that he had to receive me somewhere and chose this virtually empty room. It was better like that, because I was able to concentrate on his appearance without being distracted by a diploma or a photograph on the wall. The impression was astounding, as if some pharaoh had risen from the grave! The same swarthy skin, the same ancient, calm gaze in a perfectly noble face gave the impression that my interlocutor had lived and seen everything 3000 years before. As a surgeon I was impressed by his exceptionally delicate fingers, with unusual flexibility and dexterity. After two or three routine questions, he asked when I could start. The answer was “immediately!”
Figure 1.
Sir Magdi H. Yacoub, the “Leonardo” of cardiac surgery.
Two days later I met his team at the private Hillside Hospital, a large two-story residence that had been transformed to accommodate his foreign patients. The “international” flavor was enhanced by a constant stream of European and overseas visitors, who would spend 6 months to a year with the master. In addition to the family atmosphere in this small facility, everyone’s admiration of Magdi was striking, despite the small moans about his indifference to punctuality. It could happen that we started surgery at 7 pm and he would come at 9 pm and continue well past midnight. Bernie Logan, his robust, regular anesthesiologist, took it well, but the diminutive Allan Gillston asked venomously: “When will the pharaoh deign to come to Killside?”—using a witty pun to make a rather vitriolic joke.
BACKGROUND AND TRAINING
Magdi Yacoub (“M.Y.” to his colleagues), an Egyptian and a Christian Copt (b. 1935), came to England to train as a surgeon in 1962, because the Muslim regime in his country would not have allowed him to develop his talents. He worked first with Sir Russell Brock, well known for his pulmonary valvotomies at Guy’s Hospital. The two most famous students of Sir Russell, afterward Lord Bock, were Donald Ross, a South African surgeon and fellow student of Christiaan Barnard, and Magdi Yacoub. As Ross wrote, their boss, although a genius in the development of new ideas, was technically not particularly good.1 To the same category belong Owen Wangensteen, the famous professor at the University of Minnesota, teacher of Lillehei and Shumway, and Alfred Blalock of Johns Hopkins, professor of Denton Cooley.2 This bears out an American colleague’s pithy comment: “Great surgeons do not have ‘good hands,’ they’ve got ‘guts.’”
At the end of the 1960s, Magdi applied for a position at the historic Royal Brompton Hospital. Unfortunately for the Brompton, instead of the budding genius they chose an English surgeon with a historic name. And so only in 1973 was Magdi made a consultant at Harefield Hospital, west of London, which, due to its position in the country, was also a sanatorium. Built during the First World War, Harefield is a complex of small houses connected by long corridors. In this insignificant, anachronistic environment, Magdi Yacoub was to make major contributions to cardiac surgery and then create the greatest service for heart and lung transplants in the world, with more than 2500 cases up to the end of his career. Of course, no one could have predicted his meteoric rise, not even the people he worked with. He once suggested to Sir Peter Morris, the famous Oxford professor and authority on kidney transplantation, that together they should create a program at Harefield, perhaps the largest in the world, only to receive Morris’s condescending reply: “Are you being serious, Magdi? Can you imagine me at Harefield?” (M. Yacoub, personal communication).
SURGERY AT HAREFIELD
At Harefield, Magdi very quickly showed that he was an intellectual surgeon. He did not hesitate to apply new techniques or question established ones. He was also unbelievably skillful. His closest colleague there, the pediatric cardiology consultant Rosemary Radley-Smith, said: “Most good surgeons are only good technically. Magdi, on the other hand, has both innovative ideas and is a very fine surgeon.” And then she added: “Finesse is a characteristic of the very good surgeon.”
Although Donald Ross introduced antibiotic preserved “homografts” (more accurately allografts) to treat valvular diseases,3 it took Yacoub’s industry to establish their widespread use.4–7 The grafts were easily available (from the morgue). They were cheap and afforded excellent hemodynamics for the first few years. Implanted and unstented aortic homografts demonstrated their superiority over mechanical Starr-Edwards prostheses.8 Initially there was great excitement, but after 5 years new diastolic murmurs were detected and acute heart failure developed because of torn cusps. Failed excised valves showed an intact structure but the collagen lattice was totally acellular; that is, they were not populated by the patient’s own cells.1 Eventually, in the 1980s, the introduction of the Carpentier-Edwards bioprosthesis provided long and reliable service, even 15 years after implantation.9–11
In the mid-1970s, successful correction of complete (“D”) transposition of the great arteries was reported (the so-called switch operation).12 However, it was soon found that if surgery was undertaken several weeks after birth, the left ventricle would lose mass in facing the low-pressure pulmonary circuit and would not sustain the burden of systemic circulation after the repair.13 Early results of the switch proved disappointing. At this point, Magdi Yacoub introduced a two-stage operation for correcting the anomaly.14 The initial surgery consisted of banding the pulmonary artery proximally to increase the afterload to the left ventricle and strengthen it for the future definitive surgery. At the same time, an aortopulmonary Blalock-Hanlon shunt was constructed to improve oxygenation. Then, 4 months later, anatomical correction (“switch”) of the transposition would successfully be performed.15
His stamina was monumental. He could work days and nights on end. Finishing at 3 am, he would snatch a couple of hours of sleep before starting the new day at Harefield or the National Heart Hospital. In the evening, after more than fulfilling his obligations to the National Health Service, he would operate at the Harley Street Clinic, the Princess Grace Hospital, or Hillside. He was always calm and had the utmost concentration, without making mistakes or losing his temper with coworkers because of lack of sleep. Even after two sleepless nights he was able to discuss data regarding some project we were about to present. His superhuman strength and tranquility disarmed everyone and I, of course, was no exception.
This complete imperturbability he maintained above all in the operating room. A characteristic reaction occurred the day that Paolo, a “svelte” Italian colleague from Naples, upon opening a chest for reoperation, managed to tear the patient’s right ventricle, superior vena cava, and aorta with his saw. This was an unprecedented catastrophe that Harken would surely have christened “the Paolo maneuver”!16 Blood was running all over the floor, and Magdi was summoned. Donning gown and gloves but not stopping to scrub, he took the surgeon’s place. I expected the gates of hell to open for Paolo, but Magdi didn’t say a word. With the fingers of his left hand he hastily plugged the holes, and with his right hand he managed to place the purse strings and the cannulae and go on bypass in record time. Only when the patient was stable did he say: “Today, Paolo, you have been very naughty!” This incident only enhanced my growing admiration for the man.
Although he was absolutely calm in the operating room, he could not tolerate the slightest noise because, as he said, it stopped him “from thinking.” For him, surgery was not a simple mechanical process. His brain worked continuously, sifting through details and predicting the possible outcome. It was not unusual for him to finish the operation before completing all of the objectives we had set out. He would say: “This patient won’t tolerate any more. It’s better for him to come out alive and never mind if the operation wasn’t so perfect”—something that my perfectionist American teachers would not have espoused. Results proved him right.
HEART TRANSPLANTATION
Soon after, I moved on to Harefield Hospital as a clinical assistant, so I saw transplants performed by Magdi after he had harvested the heart (Figure 2). Fourteen cases had been done in 1980 following a moratorium imposed on heart transplantation in the United Kingdom due to poor results. Eleven more cases were done in 1981, with a small increase expected in 1982. I soon realized that the results were unsatisfactory because they were still following the protocols in use before the introduction of cyclosporine. They were a recipe for devastating infections, as I knew from my days at the Medical College of Virginia. That fall, therefore, the wonder drug was used for the first time. The instructions called for administering a generous loading dose before the operation and then large maintenance doses to prevent rejection. Cyclosporine’s harmful effect on the kidneys was not yet fully appreciated, and in a very short time recipients presented with acute renal failure.17 Some required dialysis in London, from which they never returned. The two transplant registrars (senior residents) resigned in quick succession. I had no experience of cyclosporine but I knew something about other things, so Magdi was relieved to accept my offer to take an active part in the transplants.
Figure 2.
A heart in the hands of Sir Magdi, ready for implantation (1984).
The first donor heart Magdi and I harvested was from the London Royal Free Hospital, mainstay of the National Health Service and later an important center for liver transplants. While Rosemary prepared the cold solution to preserve the heart, Magdi and I scrubbed. Very quickly he removed the heart, cutting the pulmonary veins and the inferior vena cava, then slicing through the pulmonary artery, aorta, and superior vena cava. He plunged the still-beating heart into the bowl with the cold solution and, clamping off the aorta, infused 2 L of cardioplegia solution, ensuring its protection for the next 3 or 4 hours. We packed the heart into three plastic bags, placed it in the small refrigerator, and went down quickly. Outside, a patrol car of the Metropolitan Police was waiting for us with two unsmiling policemen. The car was a Rover Vitesse, souped up and lowered for more speed and stability. Before we set off, the driver asked Magdi: “Do you wish a quick trip or a fast trip, Sir?” “Very fast, as fast as possible, please,” replied Magdi. Without comment they turned on the patrol car’s sirens and lights. The car flung itself downhill toward the London Orbital. My stomach fell to the floor, but the driver calmly pushed the other cars to the side of the road, easily taking the turns at 90 miles an hour. I drove this route every day, because we lived in north London, so I knew that we would shortly come to a 90-degree bend. I imagined that the driver would slow down a little. Nothing of the sort! He continued undaunted and the car went round the bend on two wheels! We were literally flying, and I saw Magdi clinging tightly to the handhold above the door. At the junction with the Oxford motorway, police motorcyclists were waiting and had stopped traffic so we could continue our journey at breakneck speed. The speedometer stayed at 110 to 130 miles per hour. When we arrived at Harefield, the two policemen smoothly opened the doors for us and we thanked them “for the nice trip.” After a few steps, Magdi said, “That was not driving; that was sheer madness.” We went into the operating room and he calmly performed the heart transplant. The following evening he asked me with a twinkle in his eye: “Have you recovered from yesterday’s trip? I’m still trying!”
This was the second incident in my relationship with Magdi that sealed my admiration. Here was the brightest star of European cardiac surgery risking his life to ensure the proper harvesting of the graft. This was real commitment!
One day we received a message that there was a donor in a provincial hospital when Magdi was still doing a difficult case on a child. Because he always gave priority to transplants, he prepared to hand the case over to the senior registrar, at which point Rosemary became almost hysterical. Telling him that leaving the case would be tantamount to “killing” the child, she said: “Let Peter go if the heart cannot wait!” Up to then no one else had harvested the heart. He hesitated but gave in to Rosemary’s insistence. All went well, and from that day forward I became the official surgeon for the harvesting of hearts.
This was one aspect of my work. The other was to staff and organize the transplant team. Thanks to my American training, I knew how to get people to work together. My predecessors had been on bad terms with Ariela Pomerance, a great pathologist, indispensable to our work.18 The consequence had been a delay in the results of the myocardial biopsies. I offered to go to her to look at the biopsies, at the same time explaining each clinical problem. She was overjoyed and our cooperation continued without interruption until I left. Later I invited her to Baylor where she gave a lecture. With the immunologist Marlene Rose,19 we already had good communication, and little by little we began to bring in the other subspecialties from Harefield and the neighboring hospital of Mount Vernon. In this way the team was put together, with the participation of the nursing staff, the social worker, and even the hospital dentist, so that the prospective patient would undergo a detailed examination before being seen by Magdi for the final decision.
At that point we began to collate the documents regarding the selection of patient and donor and the immunosuppression regimen. Magdi was informed and gave his approval, pleased with the progress. During this time heart transplants came thick and fast. When I joined the team in the fall of 1982, 40 transplants had been performed; the survival rate before the introduction of cyclosporine was only 30%. In 1983, we did 34 cases with a spectacular increase in survival. It was clear that there would be an increase in the number of procedures in the next few years. Despite the justifiable optimism, Magdi made a historic prediction: cyclosporine would not be a substitute for clinical excellence. He also expressed the opinion that steroids, as a part of the immunosuppression protocol, might not be necessary or might even be harmful.20,21 Following his lead, steroid taper and even discontinuation became a reality in the following years.22 Future developments proved how right he was on both counts.
HETEROTOPIC AND COMBINED TRANSPLANTATION
During this period, 1982 to 1984, Yacoub embraced heterotopic transplantation. Christiaan Barnard had suggested it to keep the patient alive following irreversible loss of the donor heart due to rejection.23 Yacoub conceived the idea that, given a period of rest afforded by the donor heart, the recipient’s cardiomyopathic heart would recover. It took several years and the establishment of long-term mechanical circulatory support to achieve clinically meaningful improvement due to structural and functional “reverse remodeling.”24 A similar hypothesis had been tested in the animal laboratory for short-term support with an allograft after temporary exclusion of the recipient left ventricle.25
In November 1983, Magdi progressed to a new procedure, the first combined transplantation of a heart and two lungs in Europe. The patient was a Swedish journalist, 33 years old, who had already been rejected by surgeons at Stanford as a lost cause. He asked Yacoub to operate on him, knowing that he would be his first such patient. Yacoub did not go to the animal laboratory as my American training led me to expect. The operation was a “one-off,” the only difference being that we brought the donor intubated to Harefield from the other hospital. In two adjacent rooms, Yacoub first removed the heart and lungs from the donor, immediately afterward doing the same thing with the recipient. He then proceeded with the transplant operation without the slightest hesitation, underlining the difference between a meticulous, well-prepared surgeon and a genius. Unfortunately, in the long run the patient did not survive—not unexpected because Norman Shumway himself had given the case his “seal” of unsuitability. Of course, this did not stop Yacoub from continuing, and even today Harefield still has the greatest experience in the world in this combined transplantation. A few years later, Yacoub introduced the concept of a “domino” transplant, in which the recipient of the combined heart–lung graft becomes a donor by giving his own healthy heart to another patient.26 This feat was repeated later at Baylor.27
In April 1984, when I left to start the heart transplantation program at Baylor University Medical Center, 95 transplants had been carried out. From the day I joined Harefield, we had done 55 operations with a short-term survival rate of 83%.
A RENAISSANCE GENIUS
Sir Magdi Yacoub practiced the whole gamut of cardiac surgery, including adults, children, and transplants, unattainable even for Denton Cooley. Despite the technical perfection of the latter, Magdi was the greater genius—not only because he introduced new concepts and new operations, especially in pediatric surgery, but because he could change the plan with the greatest skill if a different situation emerged, suiting the operation to the patient and not, as so often happens, bringing the patient “in line with” the operation. This is the supreme test of a really great surgeon!
In surgical dexterity, Magdi’s technique was unlike any other: it was completely his own. It was like looking at a work of art by Michelangelo or Raphael that “shouts out” who the master is. His movements, without being in any way theatrical, had a princely grace and delicacy. When he operated, he created. You could not be unmoved by the perfection of what he was doing, especially when he was operating on a child with congenital heart disease. I have never seen anyone else who could cut a circular patch to close a hole in the heart while looking at the hole and not the patch! He had a supernatural three-dimensional perception that enabled him to cut it the right size and shape without looking at it, his eyes glued to the hole! I do not exaggerate when I call Sir Magdi Yacoub the real Leonardo da Vinci of heart surgery, and I consider myself fortunate to have worked those 2 years with him (Figure 3).
Figure 3.
The simplicity of genius: Sir Magdi on a visit to Baylor (1995).
Speaking of working with a renaissance genius, the whole ambiance of Harefield and Hillside was that of such a workshop, with the visitors and disciples buzzing around the master craftsman. It was a fascinating experience, yet the writing on the wall was clear: we all worked for the master, not with him, and at some point had to move on with our lives—which I did.
Magdi never stayed on the beaten track. He immediately adopted mechanical support of the circulation as the alternative solution to transplantation. Going more deeply into the matter of molecular cardiology than anyone else, he proved the theory that hearts supported mechanically have a chance of recovering.28–30 At a lecture that he gave years ago in Athens, we witnessed the impressive exchange of opinions, actually a duel, with the famous James T. Willerson, editor of Circulation. I do not think anyone understood what they were arguing about, but the ease with which the “surgeon” stood up to the “scientist” made a lasting impression.
In 2001 Yacoub became the founder and director of the Science Center at Harefield, for research into the causes of heart diseases through studies into molecular and cellular biology. There, a new valve from stem cells was developed.31
In 1986 Yacoub was appointed professor at Imperial College, London, and in 1992 he was knighted. The greatest honor came in 1999, when he became a member of the Royal Society of Great Britain. Having created the Chain of Hope charity for the performance of cardiac surgery in developing countries, such as Mozambique, Ethiopia, and Jamaica, Sir Magdi was presented with the Order of Merit by Queen Elizabeth in 2014. He received numerous American honors as well, such as the Texas Heart Institute Award (1998), the Cleveland Clinic Foundation Heart Failure Award (2001), the International Society for Heart and Lung Transplantation Lifetime Achievement Award (2004), and the American College of Cardiology Legend of Cardiovascular Medicine Award (2012).
What is Sir Magdi’s hope for the future? Achieving immune tolerance as envisioned by the great Sir Peter Medawar. We are not there yet, and we may have to wait longer. Quoting Winston S. Churchill, Magdi Yacoub noted that the present achievements are “not the beginning of the end, but it is, perhaps, the end of the beginning.”32
References
- 1.Stoney WS, Ross DA In Pioneers of Cardiac Surgery. Nashville, TN: Vanderbilt University Press; 2008:285–296. [Google Scholar]
- 2.Stoney WS, Cooley DA In Pioneers of Cardiac Surgery. Nashville, TN: Vanderbilt University Press; 2008:244–255. [Google Scholar]
- 3.Ross DN. Homograft replacement of the aortic valve. Lancet. 1962;2(7254):487. doi: 10.1016/S0140-6736(62)90345-8 [DOI] [PubMed] [Google Scholar]
- 4.Yacoub M, Kittle CF. Sterilization of valve homografts by antibiotic solutions. Circulation. 1970;41(5 Suppl):II29–II32. [DOI] [PubMed] [Google Scholar]
- 5.Yacoub M, Knight E, Towers M. Proceedings: aortic valve replacement using fresh unstented homografts. Thoraxchir Vask Chir. 1973;21(5):451–457. doi: 10.1055/s-0028-1099071. [DOI] [PubMed] [Google Scholar]
- 6.Gula G, Ahmed M, Thompson RH, Radley-Smith R, Yacoub MH. Combined homograft replacement of the aortic valve and aortic root with reimplantation of the coronary arteries. Circulation. 1976;54(suppl II):11. [Google Scholar]
- 7.Thompson R, Knight E, Ahmed M, Somerville W, Towers M, Yacoub M. The use of “fresh” unstented homograft valves for replacement of the aortic valve: analysis of 6 1/2 years experience. Circulation. 1977;56(5):837–841. doi: 10.1161/01.CIR.56.5.837. [DOI] [PubMed] [Google Scholar]
- 8.Hollinrake K, Baidya M, Yacoub MH. Haemodynamic changes in patients with high pulmonary vascular resistance after mitral valve replacement. Comparative study between use of unstented aortic homograft valves and Starr-Edwards prostheses. Br Heart J. 1973;35(10):1047–1054. doi: 10.1136/hrt.35.10.1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Corbineau H, De La Tour B, Verhoye JP, Langanay T, Lelong B, Leguerrier A. Carpentier-Edwards supraannular porcine bioprosthesis in aortic position: 16-year experience. Ann Thorac Surg. 2001;71(5):S228–S231. doi: 10.1016/S0003-4975(01)02545-0. [DOI] [PubMed] [Google Scholar]
- 10.Spencer FC, Baumann FG, Grossi EA, Culliford AT, Galloway AC. Experiences with 1643 porcine prosthetic valves in 1492 patients. Ann Surg. 1986;203(6):691–700. doi: 10.1097/00000658-198606000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moon MR, Miller DC, Moore KA, et al. Treatment of endocarditis with valve replacement: the question of tissue versus mechanical prosthesis. Ann Thorac Surg. 2001;71(4):1164–1171. doi: 10.1016/S0003-4975(00)02665-5. [DOI] [PubMed] [Google Scholar]
- 12.Jatene AD, Fontes VF, Paulista PP, et al. Successful anatomic correction of transposition of the great vessels. A preliminary report. Arq Bras Cardiol. 1975;28(4):461–464. [PubMed] [Google Scholar]
- 13.Editorial: transposition of the great arteries. BMJ. 1976;1(6018):1104–1105. doi: 10.1136/bmj.1.6018.1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yacoub MH, Radley-Smith R, Maclaurin R. Two-stage operation for anatomical correction of transposition of the great arteries with intact interventricular septum. Lancet. 1977;309(8025):1275–1278. doi: 10.1016/S0140-6736(77)91317-4. [DOI] [PubMed] [Google Scholar]
- 15.Yacoub M, Bernhard A, Lange P, et al. Clinical and hemodynamic results of the two-stage anatomic correction of simple transposition of the great arteries. Circulation. 1980;62(2 Pt 2):I190–I196. [PubMed] [Google Scholar]
- 16.Alivizatos P. Dwight Emary Harken, MD, an all-American surgical giant: pioneer cardiac surgeon, teacher, mentor. Proc (Bayl Univ Med Cent). 2018;22(4):554-557. doi: 10.1080/08998280.2018.1471896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Greenberg A, Thompson ME, Griffith BJ, et al. Cyclosporine nephrotoxicity in cardiac allograft patients—a seven-year follow-up. Transplantation. 1990;50(4):589–593. doi: 10.1097/00007890-199010000-00012. [DOI] [PubMed] [Google Scholar]
- 18.Pomerance A, Davies MJ. The Pathology of the Heart. Oxford: Blackwell Scientific Publications; 1975. [Google Scholar]
- 19.Rose ML, Yacoub MH. Immunocytochemical analysis of transplanted heart and lung In: Rose M, Yacoub M, eds. Immunology of Heart and Lung Transplantation. London: Edward Arnold; 1993. [Google Scholar]
- 20.Yacoub MH, Alivizatos PA, Radley-Smith R, Khaghani A. Are steroids really necessary? J Am Coll Cardiol. 1985;5(2):533. [Google Scholar]
- 21.Yacoub MH, Alivizatos PA, Khaghani A, Mitchell A. The use of cyclosporine, azathioprine and antithymocyte globulin with or without low-dose steroids for immunosuppression of cardiac transplant patients. Transplant Proc. 1985;17:221–222. [Google Scholar]
- 22.Rosenbaum DH, Adams BC, Mitchell JD, et al. Effects of early steroid withdrawal after heart transplantation. Ann Thorac Surg. 2006;82(2):637–644. doi: 10.1016/j.athoracsur.2006.03.067. [DOI] [PubMed] [Google Scholar]
- 23.Novitzky D. The surgical techniques of orthotopic and heterotopic heart transplantation In Cooper DKC, Lanza RP, eds. Heart Transplantation: The Present Status of Orthotopic and Heterotopic Heart Transplantation. Surrey, UK: MTP Press; 1984:113–127. [Google Scholar]
- 24.Yacoub MH. A novel strategy to maximize the efficacy of left ventricular assist devices as a bridge to recovery. Eur Heart J. 2001;22(7):534–540. doi: 10.1053/euhj.2001.2613. [DOI] [PubMed] [Google Scholar]
- 25.Alivizatos PA, Guerraty AJ, Lower RR. Circulatory assistance with a cardiac allograft after exclusion of the canine left ventricle. Ann Thorac Surg. 1982;34(2):132–137. doi: 10.1016/S0003-4975(10)60873-9. [DOI] [PubMed] [Google Scholar]
- 26.Yacoub MH, Banner NR, Khaghani A, et al. Heart-lung transplantation for cystic fibrosis and subsequent domino heart transplantation. J Heart Transplant. 1990;9(5):459–466. [PubMed] [Google Scholar]
- 27.Alivizatos PA. A Baylor milestone: combined heart/lung-heart “domino” transplant. Proc (Bayl Univ Med Cent). 2018;31(1):123–125. doi: 10.1080/08998280.2017.1391037.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Birks EJ, Tansley PD, Hardy J, et al. Left ventricular assist device and drug therapy for the reversal of heart failure. N Engl J Med. 2006;355(18):1873–1884. doi: 10.1056/NEJMoa053063. [DOI] [PubMed] [Google Scholar]
- 29.Birks EJ, George RS, Hedger M, et al. Reversal of severe heart failure with a continuous-flow left ventricular assist device and pharmacological therapy: a prospective study. Circulation. 2011;123(4):381–390. doi: 10.1161/CIRCULATIONAHA.109.933960. [DOI] [PubMed] [Google Scholar]
- 30.Drakos SG, Mehra MR. Clinical myocardial recovery during long-term mechanical support in advanced heart failure: insights into moving the field forward. J Heart Lung Transplant. 2016;35(4):413–420. doi: 10.1016/j.healun.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 31.Yacoub M. Taking translational research to heart: an interview with Sir Magdi Yacoub. Dis Model Mech. 2009;2(9-10):433–435. doi: 10.1242/dmm.004176. [DOI] [PubMed] [Google Scholar]
- 32.Yacoub M. Heart transplantation: the end of the beginning. Am J Transplant. 2008;8(9):1767–1768. doi: 10.1111/j.1600-6143.2008.02366.x. [DOI] [PubMed] [Google Scholar]