Abstract
Musical training and performance require precise integration of multisensory and motor centres of the human brain and can be regarded as an epigenetic modifier of brain functions. Numerous studies have identified structural and functional differences between the brains of musicians and non-musicians and superior cognitive functions in musicians. Recently, music-listening and performance has also been shown to affect the regulation of several genes, many of which were identified in songbird singing. MicroRNAs affect gene regulation and studying their expression may give new insights into the epigenetic effect of music. Here, we studied the effect of 2 hours of classical music-performance on the peripheral blood microRNA expressions in professional musicians with respect to a control activity without music for the same duration. As detecting transcriptomic changes in the functional human brain remains a challenge for geneticists, we used peripheral blood to study music-performance induced microRNA changes and interpreted the results in terms of potential effects on brain function, based on the current knowledge about the microRNA function in blood and brain. We identified significant (FDR <0.05) up-regulation of five microRNAs; hsa-miR-3909, hsa-miR-30d-5p, hsa-miR-92a-3p, hsa-miR-222-3p and hsa-miR-30a-5p; and down-regulation of two microRNAs; hsa-miR-6803-3p and hsa-miR-1249-3p. hsa-miR-222-3p and hsa-miR-92a-3p putatively target FOXP2, which was found down-regulated by microRNA regulation in songbird singing. miR-30d and miR-222 corroborate microRNA response observed in zebra finch song-listening/learning. miR-222 is induced by ERK cascade, which is important for memory formation, motor neuron functions and neuronal plasticity. miR-222 is also activated by FOSL1, an immediate early gene from the FOS family of transcriptional regulators which are activated by auditory-motor stimuli. miR-222 and miR-92 promote neurite outgrowth by negatively regulating the neuronal growth inhibitor, PTEN, and by activating CREB expression and phosphorylation. The up-regulation of microRNAs previously found to be regulators of auditory and nervous system functions (miR-30d, miR-92a and miR-222) is indicative of the sensory perception processes associated with music-performance. Akt signalling pathway which has roles in cell survival, cell differentiation, activation of CREB signalling and dopamine transmission was one of the functions regulated by the up-regulated microRNAs; in accordance with functions identified from songbird learning. The up-regulated microRNAs were also found to be regulators of apoptosis, suggesting repression of apoptotic mechanisms in connection with music-performance. Furthermore, comparative analyses of the target genes of differentially expressed microRNAs with that of the song-responsive microRNAs in songbirds suggest convergent regulatory mechanisms underlying auditory perception.
Keywords: MicroRNA, Sequencing, Music-performance, Gene regulation, Peripheral blood, Cognition, Perception, Neuroplasticity
Introduction
Playing an instrument requires meticulous integration between the sensory (visual, auditory, tactile) and motor systems (timing, sequencing and spatial organisation of movements) (Zatorre, Chen & Penhune, 2007; Hyde et al., 2009). Further, orchestral performance requires synchronisation between fellow musicians and the conductor via non-verbal visuomotor and audiomotor communication, underscoring enhanced cooperation between musicians (Volpe et al., 2016). Long-term focused sensorimotor training often required for mastering a musical instrument causes structural and functional changes in the brain and cerebral cognitive networks (Schlaug, 2001; Gaser & Schlaug, 2003; Meyer, Elmer & Jäncke, 2012; Herholz & Zatorre, 2012; Moore et al., 2014). Accordingly, neural processing of music components and activation of different brain areas differ significantly between musicians and non-musicians (Park et al., 2014).
Research based on the effect of music training on the musician’s brain has often used neuroimaging techniques and descriptive analyses (Elbert et al., 1995; Schlaug, 2001; Gaser & Schlaug, 2003; Hyde et al., 2009; Meyer, Elmer & Jäncke, 2012; Herholz & Zatorre, 2012; Steele et al., 2013; Moore et al., 2014; Park et al., 2014). By applying genomic approaches, we and others have previously reported genetic predisposition in shaping human musical aptitude (Pulli et al., 2008; Park et al., 2012; Ukkola-Vuoti et al., 2013; Oikkonen et al., 2015). Music can also cause epigenetic alterations in the human body: we have shown that music-performance and music-listening affect expression of several genes, many of which are regulated by singing in songbirds (Kanduri et al., 2015a, 2015b). Indeed, songbirds offer important models for studying genomic mechanisms underlying sound perception and production (vocal communication) and social communication in humans (Whitney et al., 2014).
MicroRNAs are small non-coding RNAs of approximately 18–22 bp in length that can post-transcriptionally regulate approximately 60% of human genes, including those that play a critical role in neuronal functions like synaptic plasticity (Huntzinger & Izaurralde, 2011; Olde Loohuis et al., 2012; Wang, Kwon & Tsai, 2012). Further, studies on synaptic dynamics indicate that synaptic efficiency and size can be modified in a time frame of minutes (Regehr, 2012; Altenmüller & Schlaug, 2015). Interestingly, studies on songbirds indicate regulation of microRNAs in the auditory forebrain with functional implications on neuron differentiation and neuronal plasticity (Gunaratne et al., 2011). Here, we studied changes in the peripheral blood microRNA transcriptome after 2 hours of concert performance in professional musicians using high-throughput sequencing and computational methods. We have used peripheral whole blood as functional human brain tissues are inaccessible.
Materials and methods
Ethical permission
The study was conducted in accordance with the guidelines of Helsinki Declaration and ethical permission (number 233/13/03/2013) has been approved by the Ethical Committee of Helsinki University Central Hospital. Additionally, a written informed consent has been obtained from the participants.
Music-performance
The music-performance study was comprised of professional musicians (N = 10) from Tapiola sinfonietta who performed a Western classical music concert (duration approximately 2 hours) to the public (see Supplemental Methods). Seven of the 10 participants were also participants of our previous gene expression study (Kanduri et al., 2015a). The program for the concert comprised five music pieces described earlier (Kanduri et al., 2015a). We then collected verbal data from all the musicians regarding their activities before the concert (coffee/alcohol consumption), personal ratings (pleasantness of the concert and blood sample collections, conductor’s influence on the music-performance, mood of the musicians before and after the concert, fondness for the played music pieces and familiarity of the pieces played) and stress factors for the day before concert and for the concert day using a questionnaire, described in detail in Kanduri et al. (2015a).
Control study
The control group consisted of professional musicians (N = 10); but they neither played music nor listened to music during the control study (see Supplemental Methods). Instead, the musicians could choose either to attend a lecture or walk outside for 2 hours. The characteristics of the control group of professional musicians (N = 10) has been described earlier (Kanduri et al., 2015a). Using the same participants and music exposure as in the gene expression study (Kanduri et al., 2015a) enables to integrate the findings to understand the microRNA:mRNA interaction and the regulatory network in professional music-performance.
Sample collection, RNA extraction and sequencing
Peripheral whole blood samples (2.5 ml) were collected from the musicians immediately before and immediately after the music-performance and control study. The details of sample collection, microRNA extraction and sequencing are provided in the Supplemental Methods.
Pre-processing and statistical analysis of microRNA expression
The reads from the microRNA sequencing runs were quality controlled with FastQC version 11.3 (http://www.bioinformatics.babraham.ac.uk/projects/fastqc/) and trimmed to remove the 3′ adapter sequence and low quality reads using Trim Galore! Version 0.3.7 (http://www.bioinformatics.babraham.ac.uk/projects/trim_galore/). An adapter overlap of five bp, read length threshold of 15 bp, Phred score quality cut-off of 20 and an error rate threshold of 0.1 were applied for adapter and quality trimming. The reads were quality checked again using FastQC and aligned to the human genome reference (GRCh38, Ensembl release 76) using bowtie version 1.1.2 (Langmead et al., 2009) requiring perfect match for a seed length of 18 and selecting only unique alignments. The microRNA expressions were calculated from the alignments using HTSeq release 0.6.1p1 (Anders, Pyl & Huber, 2015) with annotations from the miRBase version 21 (Kozomara & Griffiths-Jones, 2014).
For the statistical analysis of microRNA expression differences over time (before vs. after 2 hours) across the music-performance and the control study, we used DESeq2 (version 1.22.1) (Love, Huber & Anders, 2014). DESeq2 performs very conservative testing based on negative binomial distribution of the sequencing counts with optimal balance between specificity and sensitivity (Khang & Lau, 2015; Tang et al., 2015) by: (1) estimating normalisation factors to account for sequencing depth differences between samples, (2) calculating microRNA-wise dispersions, (3) fitting negative binomial generalised linear model (GLM) and (4) by performing statistical significance testing. For this study, we used a GLM with a design formula containing the condition factor (music-performance and control activity (reference)), the time factor (Post and Pre (reference)), their interaction and the paired experimental design (R code: dds <- DESeqDataSet(dds, ∼ Pair + treatment + time + treatment:time)). We then performed the likelihood ratio test with a reduced model which does not contain the interaction term to find microRNAs that changed their expression over time (2 hours), as a result of music-performance, compared to the control activity (music-performance vs. control), as suggested in DESeq2 vignette (R code: dds <- DESeq(dds, test=‘LRT’, reduced = ∼ Pair + treatment+ time)). Additionally, to determine microRNAs with significant differential expression, we applied the Benjamini–Hochberg’s false discovery rate (FDR) threshold of 5%. Further, we selected only those differentially expressed microRNAs with a fold threshold of at least 1.3 over time (post vs. pre) across conditions (music-performance vs. controls), and post–pre threshold change of at least 15% for the music-performance study as candidate microRNAs.
Some important aspects of assigning fold change thresholds to individual studies should be taken into consideration here. The choice of thresholds to prioritise the top candidates from a study is based on the study question and the number of features (genes or microRNAs) identified as differentially expressed (Wu, Wang & Wu, 2015). Furthermore, our study represents gene–environment interaction where lower expression fold changes has been observed (Radom-Aizik et al., 2012, 2014; Black et al., 2013; Beech et al., 2014; Osterndorff-Kahanek et al., 2018).
Functional over-representation analysis of microRNAs
We assessed the functions of the differentially expressed (DE) microRNAs using two microRNA specific enrichment tools, TAM 2.0 (Li et al., 2018) and miRNA enrichment analysis and annotation tool (miEAA) (Backes et al., 2016) due to the bias observed in the functional enrichment analysis of the target genes of microRNAs by standard enrichment tools (Bleazard, Lamb & Griffiths-Jones, 2015). TAM 2.0 performs over-representation analysis (ORA) using hypergeometric test to test whether the input microRNAs are over or under-represented in the reference microRNA annotation sets (associated with functions and diseases) specifically curated for microRNA genes. TAM 2.0 also calculates the correlations between the input set of microRNAs and microRNAs which were found to be deregulated in other disease conditions based on literature findings. Whereas, miEAA performs ORA at the level of mature microRNAs for categories (ontology, pathway or diseases) and tests whether a category is significantly enriched (Fisher’s exact test) in a given microRNA set with respect to the reference using statistical test implemented in the gene set analysis toolkit, GeneTrail. It has to be noted here that majority of functions for the microRNAs in the reference annotation set used in miEAA for the gene ontology ORA are derived mainly based on the functions of their target genes.
Using TAM 2.0, we performed ORA of functions and diseases for our up-regulated and down-regulated microRNAs separately with default options (FDR <0.05) and with cancer terms masked. We then used miEAA for ORA of our up-regulated and down-regulated microRNAs separately with default options (FDR <0.05) selecting all the categories and used all annotated microRNAs/precursors collected in miEAA as reference set. To relate the microRNA expression patterns in blood to that of brain, we verified tissue specificity of the DE microRNAs and their target genes using microRNA databases, other databases (BBBomics) (Kalari et al., 2016) and literature searches.
MicroRNA target gene detection and their functional relevance
To understand microRNA regulatory mechanisms and networks in music-performance, we collected the validated target gene interactions for each DE microRNA with strong evidence (reporter assay or western blot) from miRTarBase Release 7.0 (Chou et al., 2018). Additionally, predicted target genes for each of the DE microRNAs were gathered from TargetScan algorithm Release 7.2 (Agarwal et al., 2015) using the below filtering criteria to reduce the false positive target genes. For the conserved and broadly conserved microRNA families, we chose only the target genes with conserved sites, having an aggregate probability of conserved targeting at least 0.2 and a total context++ score at most −0.15. For the poorly conserved DE microRNA families and those with other miRBase annotations, target genes with a total context++ score of less than −0.15 were selected (Agarwal et al., 2015). We did not consider predicted target genes of the DE microRNAs with non-canonical binding. Next, we extracted the latest validated ontology annotations derived using experimentally verified miRNA:target interaction data, named EBI-GOA-miRNA (Huntley et al., 2018), via the PSICQUIC web service. Additionally, we also searched the Rat Genome Database (Shimoyama et al., 2015) for annotations of our conserved microRNA orthologs, as per the Alliance of Genome Resources, from Rattus norvegicus.
Comparative analyses
We assessed conservativeness of the microRNA regulatory mechanisms associated with music perception and production in professional musicians by comparative analysis to the microRNA regulatory patterns identified in the songbird brain after song-listening (Gunaratne et al., 2011) and singing (Shi et al., 2013). We then compared target genes of the differentially regulated microRNAs after music-performance to the target genes of song-listening and singing responsive microRNAs from songbirds (Gunaratne et al., 2011; Shi et al., 2013; Lin, Balakrishnan & Clayton, 2014). Next, we tested the overlap between the target genes of the down-regulated microRNAs from our study and the genes stimulated by singing in songbirds. Towards this, we compiled a dataset comprising genes which were previously identified to show behavioural regulation (up-regulation) after singing in songbird brain based on findings from three studies (Wada et al., 2006; Hilliard et al., 2012; Whitney et al., 2014) and call it song production cum perception gene set. We then implemented a permutation test to determine whether the observed overlap between the song production cum perception gene set and the target genes of the down-regulated microRNAs from our study was more than that expected by chance through re-sampling (N = 10,000 times) of the datasets. Specifically, we randomly sampled (without replacement) the songbird genes of the same size as song production cum perception gene set using all the annotated genes from Taeniopygia guttata (N = 17,926) as Universe. Similarly, human genes were randomly sampled (without replacement) for the same size as the number of predicted and validated target genes of the down-regulated microRNAs using annotated human genes as the Universe (N = 20,219). We next estimated the overlap between these re-sampled datasets for each of the permutations of these datasets and calculated the p-value of the overlap. Similarly, we also analysed the statistical significance of the overlap between target genes of the up-regulated microRNAs from our study and the genes found inhibited after singing in songbird brain (Whitney et al., 2014) using re-sampling (N = 10,000).
Integrated analysis and putative regulatory network construction
Our objective with the integrated analysis of our microRNA results with the gene expression data in professional music-performance (Kanduri et al., 2015a) was to create a putative gene regulatory network that represents the molecular mechanisms underlying music-performance in professional musicians. We created microRNA–gene regulatory interactions using the target genes collected from miRTarBase, TargetScan and using Qiagen’s Ingenuity® Pathway Analysis (IPA®; Qiagen, Redwood City, CA, USA, www.qiagen.com/ingenuity). Only those target genes of the differentially expressed microRNAs from this study which showed inverse direction of regulation in the gene expression findings, from the same music-performance and control activity as this study (Kanduri et al., 2015a), were considered for generating microRNA–gene regulatory interactions.
Additionally, for constructing the putative regulatory network, we have considered microRNA-transcriptional factor (TF), TF-genes and TF-microRNA regulatory information. We considered microRNA-TF regulatory data as microRNAs, which can be under the regulation of transcriptional regulators, can regulate genes either directly or indirectly through the regulation of transcription factors (Otaegi et al., 2011; Li et al., 2017). The transcriptional regulatory data for the DE genes (TF-gene) were obtained from Table S4 of the same study (Kanduri et al., 2015a), which comprises the statistically significant upstream regulators for the DE genes. Additionally, for this study, we collected the regulatory effect (activation/inhibition) of the up-stream regulator on the target gene and their validation status for incorporating the information to the network. MicroRNA–TF interaction data were gathered from TargetScan, miRTarBase and literature for identifying the interaction between DE microRNAs and the over-represented transcriptional regulators of the DE genes (Kanduri et al., 2015a) after considering their putative regulatory effect on the DE genes. Validated TF-microRNA regulatory data were collected from TransmiR-2 (Wang et al., 2010) and literature search. Further, only those TFs previously found to be associated with musical traits (Oikkonen et al., 2016) and/or found in songbird singing or listening (Wada et al., 2006; Hilliard et al., 2012; Whitney et al., 2014) were considered for the putative regulatory network construction. This putative regulatory network was further extended with validated target genes of the DE microRNAs that were found both from miRTarBase and EBI-GOA-miRNA, their putative neighbours, co-expressions and indirect regulators gathered using the STRING database (Szklarczyk et al., 2017) and extensive literature search to connect the microRNA and gene expression findings and visualised with Cytoscape 3.7.0 (Shannon et al., 2003).
Results
Comparison of the music-performance and control study participants
We chose professional performing musicians as they start their music training during sensitive period of human brain development (approximately 6 year of age) (Steele et al., 2013), undergo rigorous training and constantly practice to maintain their expertise (Pallesen et al., 2010; Oechslin et al., 2013). Table 1 shows detailed demographic information for the professional performers (N = 10) and control musicians (N = 10) and their statistical comparison.
Table 1. Phenotypic characteristics of the participants.
| Characteristic | Music-performance (N = 10) | Control study (N = 10) |
|---|---|---|
| Female | 6 | 8 |
| Age | 48.3 ± 6.17; CI = 4.41 | 39.2 ± 11.31; CI = 8.09 |
| Age at commencing of music practice | 6.6 ± 1.71; CI = 1.23 | 5.7 ± 1.64; CI = 1.17 |
| Average hours of practise/day | 5.3 ± 0.82; CI = 0.59 | 4.3 ± 2.06; CI = 1.47 |
| Keyboard musicians | 0 | 3 |
| String musicians | 10 | 4 |
| Wind musicians | 0 | 3 |
| University degree (B.Mus./Diploma/Master’s) | 10 | 10 |
Note:
Participant demographic data are furnished in this table. Continuous explanatory variables—age, age at commencing of music practice and average hours of practice, are presented as the mean ± standard deviation with confidence interval (CI) at 95%. To assess the similarity of the music-performance and control participants, we performed two-sided t-tests to compare the age of commencement of musical practice, age of the participants and number of hours of musical practice per day. At 95% confidence, we did not find any significant (p < 0.05) differences in the age of commencement of musical practice, age of the participants and the number of hours of musical practice per day between music-performance and control groups. Additionally, we performed two-sided fisher’s exact test for count data to assess any statistically significant differences between the gender and music education level of the music-performance and control groups. However, at 95% confidence, we did not find any statistically significant differences in the gender of the participants between music-performance and control groups. Similarly, the education level of the performing musicians and the control group did not show any statistically significant differences from each other.
MicroRNA response to music-performance
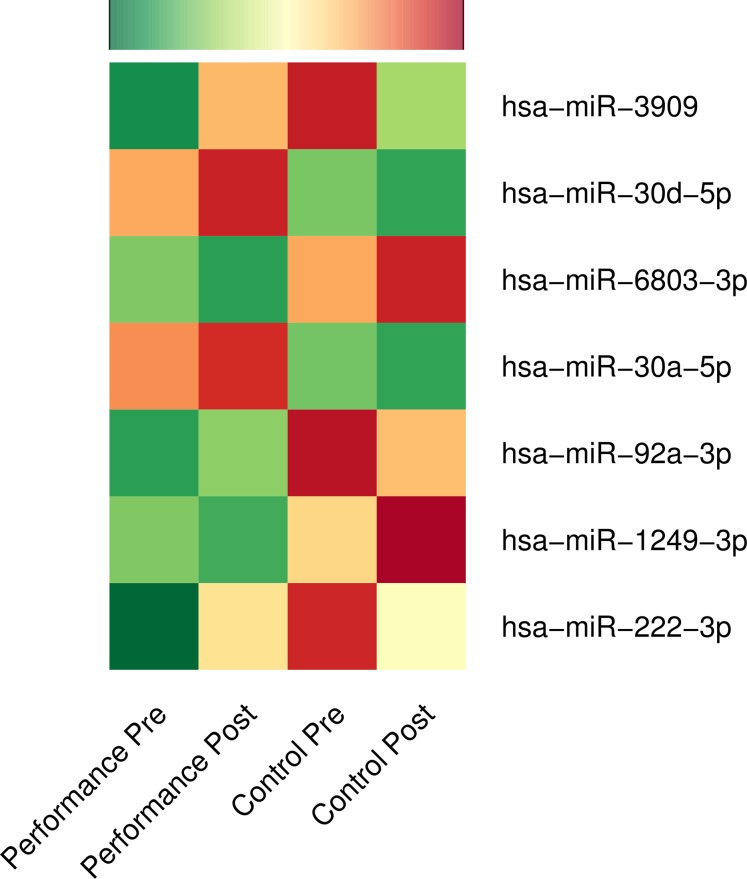
We compared the microRNA expressions over time (post vs. pre) across the music-performance and the control activity with DESeq2 (Love, Huber & Anders, 2014) owing to its strengths mentioned below. DESeq2 is one of the most sensitive statistical tools for performing differential expression analysis of high throughput sequencing experiments with a wide range of sample numbers as well as for those with a smaller fold changes (Love, Huber & Anders, 2014; Zhou, Lindsay & Robinson, 2014). Furthermore, systematic comparison of statistical tools for analysing differential expression shows DESeq2 giving a false positive rate as low as 0 and true positive rate above 80%, even with a log fold threshold as low as 0.5 and with a replicate number of 6 (Schurch et al., 2016), making DESeq2 a statistically appropriate tool for this study. At a stringent FDR threshold of 5%, we identified significant up-regulation of five microRNAs (hsa-miR-3909, hsa-miR-30d-5p, hsa-miR-92a-3p, hsa-miR-30a-5p, hsa-miR-222-3p) and down-regulation of two microRNAs (hsa-miR-6803-3p and hsa-miR-1249-3p) over time (2 hours) after music-performance, compared to the control activity (Fig. 1; Table S1). Genomic information for the DE microRNAs is provided in Table S2 and a concise list of putative functions of the up-regulated microRNAs and selected target genes of the down-regulated microRNAs in Table 2. The up-regulated microRNA gene hsa-mir-30d is located on 8q24.22, near to 8q24, which has been implicated in absolute pitch (Theusch, Basu & Gitschier, 2009) and auditory neuropathy (Rance, 2005). Likewise, hsa-mir-3909 is located on 22q12.3, whose deletions (22q12.3–q13.1) are associated with neurocristopathy and sensorineural hearing loss (Jelena et al., 2014).
Figure 1. Differential expression of microRNAs: music-performance vs. controls.
Normalised mean expressions of differentially regulated microRNAs before and after music-performance/control studies are represented using heatmap. Green, yellow and red colours denote low, moderate and high expression of microRNAs, respectively.
Table 2. Putative functions of the differentially regulated microRNAs and their target genes in music-performance.
| Up-regulated microRNAs | Putative functions |
|---|---|
| hsa-miR-30d-5p | Responsive to song-listening in songbird forebrain. Takes part in auditory, sensory, nervous system development and functioning, myelination, vascular remodelling. |
| hsa-miR-222-3p | Responsive to song-listening in songbird forebrain. Takes part in nervous system development, inner ear development, neuron differentiation, neuronal plasticity, maintenance of long-term potentiation, Schwann cell regeneration, sensory perception of sound, angiogenesis, cardiac growth, negative regulation of apoptosis. |
| hsa-miR-92a-3p | Motor neuron differentiation, synaptic plasticity, long-term synaptic potentiation, sensory perception of sound, negative regulation of apoptosis, regulation of heart contraction |
| hsa-miR-30a-5p | Schwann cell differentiation, neuron differentiation, myelination, negative regulation of apoptosis, vascular remodelling. |
| hsa-miR-3909 | Neuroprotection |
Note:
Functions of up-regulated microRNAs and their targets were gathered from literature. Down-regulated microRNAs are not well-characterised for functions or target genes.
Putative functions of the DE microRNAs
From the analysis of the DE microRNAs using TAM 2.0, the up-regulated microRNAs were found to have functions in aging, chondrocyte development, angiogenesis, regulation of Akt pathway and cell death (Data S1). The DE microRNAs, hsa-miR-222-3p and hsa-miR-92a-3p activates AKT (AKT serine/threonine kinase 1) signalling through their targeting of PTEN, which is an activator of apoptosis and inhibitor of AKT. Akt is activated by growth factors, insulin (Hayashi et al., 2017) and is regulated through phosphatidylinositol 3-OH kinase (PI3′K) signalling, implicated in dopamine neurotransmission (Tan et al., 2008) and promotes cell survival, cellular proliferation and growth signalling, in part, through the activation of the transcriptional regulator, CREB (Du & Montminy, 1998). According to Entrez gene annotation, activated AKT suppress apoptosis and is involved in the growth factor-mediated neuronal survival in the developing nervous system. This is consistent with microRNA findings from songbird learning (Gunaratne et al., 2011) where the target genes of the down-regulated microRNA activated MAPK signalling pathway, another pathway with roles in cellular proliferation and neuronal plasticity, and confirms some of the findings from human music-performance (Kanduri et al., 2015a) and music-perception (Kanduri et al., 2015b). Interestingly, conditional inactivation of AKT1 has been linked to the hyperphosphorylation of TAU protein which is implicated in Alzheimer’s disease (Wang et al., 2015). The up-regulated microRNAs repressed cell death and were found to be interfering with damaging cues.
Based on the analysis of the microRNAs using miEAA, the up-regulated microRNAs were found to be regulators of apoptotic processes implying pro-survival effects associated with music-performance. Other functions regulated by the up-regulated microRNAs include sensory perception, peripheral nervous system axon regeneration, intracellular transport, cytokine production and primary microRNA processing (Data S2). Furthermore, miR-30d and miR-222 in R. norvegicus are involved in long-term synaptic potentiation and sensory perception of sound (Smith et al., 2018). The functions and tissue expression data of the candidate microRNAs based on annotations from microRNA databases and from extensive literature analysis are given in Table S3.
Target genes and comparative analyses with genetic studies on song perception and production
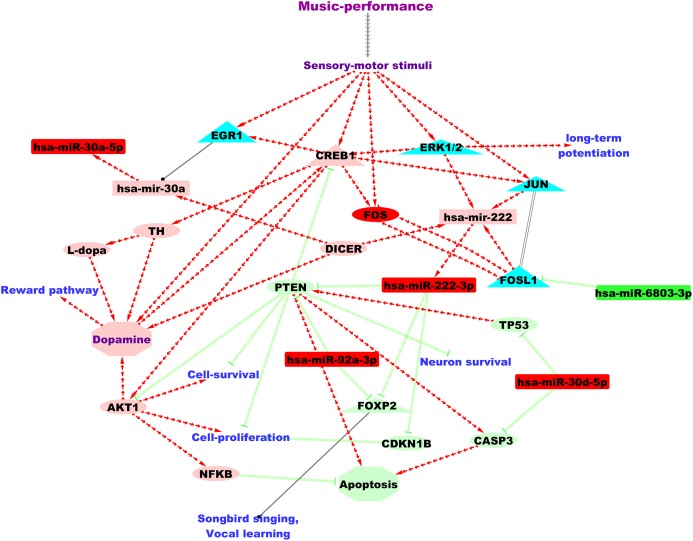
Altogether, we collected 2,027 and 3,825 target genes (validated and predicted inclusive), respectively, for the down-regulated and up-regulated microRNAs from both TargetScan and miRTarBase. Out of these, validated human target genes supported by strong experimental evidence were observed only for the up-regulated microRNAs (N = 143) from miRTarBase. Validated target genes and GO annotations as per EBI-GOA-miRNA dataset (Huntley et al., 2018) were only available for three of the seven DE microRNAs: hsa-miR-30a-5p, hsa-miR-222-3p and hsa-miR-92a-3p. Based on EBI-GOA-miRNA dataset, hsa-miR-92a-3p and hsa-miR-222-3p were involved in the negative regulation of inflammatory response (GO:0050728) and angiogenesis (GO:0016525). Additionally, we identified PTEN, a promoter of apoptotic processes, as the validated target shared by hsa-miR-222-3p and hsa-miR-92a-3p from EBI-GOA-miRNA dataset. A putative microRNA regulatory network underlying professional music-performance based on findings from the current study and its functional relevance is summarised in Fig. 2.
Figure 2. A putative microRNA regulatory network and summary of the findings from the current study based on the analysis of the DE microRNAs.
Green and red colour nodes, respectively, indicate down-regulated and up-regulated microRNAs from our study. Light-green and coral colour nodes denote putative repression and activation, respectively, based on findings from microRNA databases and literature. Annotation for edges: Red separate arrow edges denote activation; green backward slashes indicate repression; solid black edge denotes regulation and parallel lines indicate co-expression. MicroRNAs are represented using rectangles, genes using ellipses and transcriptional regulators using triangles.
Comparative analyses to songbirds identified three of the up-regulated microRNAs from this study (miR-30, miR-222 and miR-92) as regulated in the auditory forebrain of songbirds after song-learning (Gunaratne et al., 2011). Further, two of the microRNAs found up-regulated after music-performance in the professional musicians in our study, hsa-miR-92a-3p and hsa-miR-222-3p, target FOXP2 and constitute a microRNA-FOXP2 gene regulatory network. This is in accordance with findings from songbirds in which undirected singing behaviour up-regulated two microRNAs in the brain (miR-9 and miR-140-5p) that were regulators of the FOXP2 gene, which is important for vocal learning (Shi et al., 2013). Amongst the predicted target genes of the song-learning (listening) inhibited miR-2954 (Gunaratne et al., 2011), we identified TLK2 as putatively regulated by hsa-miR-6803-3p, which is down-regulated after music-performance. hsa-miR-6803-3p is also predicted to target PRKCA, which was up-regulated during song-responsive down-regulation of miR-2954 (Lin, Balakrishnan & Clayton, 2014). Similarly, we also observed putative targeting of RPS11 and RPL29, which were down-regulated after miR-2954 inhibition (Lin, Balakrishnan & Clayton, 2014), by hsa-miR-3909, one of the up-regulated microRNAs from this study.
From the analysis of the significance of the gene-set overlap based on permutation test, the overlap observed between target genes of the down-regulated microRNAs and the genes stimulated by singing in songbirds, song production cum perception gene set, is more than that expected by chance (p-value = 0.0017). Likewise, we also identified a statistically significant overlap between the genes down-regulated after songbird singing (Whitney et al., 2014) and the target genes of the up-regulated microRNAs (re-sampling p-value = 0, N = 10,000); 33.33% of the genes down-regulated after songbird singing (Whitney et al., 2014) were amongst target genes of the up-regulated microRNAs (Table S4).
Integrated analyses of music-performance responsive micrornas and genes
Differentially expressed microRNAs from this study were expression paired and assessed for inverse regulatory patterns with the RNA expression study, which studied the same music-performance in the same musicians (Kanduri et al., 2015a), with IPA and using the collected target genes. From this integrated analysis, we observed predicted interaction of the down-regulated hsa-miR-1249-3p with one of the up-regulated genes, OSBP2, from the gene expression analysis (Kanduri et al., 2015a) (Table 3). Likewise, we identified predicted interactions of the up-regulated microRNAs with down-regulated genes (Kanduri et al., 2015a); hsa-miR-222-3p with NBPF9; hsa-miR-3909 with NBPF9, CNPY3, CYTH4, ABI3, ADRB2; hsa-miR-30d-5p and hsa-miR-30a-5p with the down-regulated CHD9 and ADRB2 (Table 3). We also observed the differentially regulated microRNAs from our study to have predicted interactions with statistically over-represented upstream regulators of genes differentially regulated in professional music-performance (Table 3).
Table 3. MicroRNA:gene and microRNA:TF interactions (direct) associated with 2 hours of professional music-performance obtained by integrating microRNA expression profiles, gene expression profiles and statistically significant up-stream regulators of the DE genes.
| Comparison | Target genes for the DE microRNAs from the differentially regulated genes |
|---|---|
| Up-regulated microRNA interactions with down-regulated genes from (Kanduri et al., 2015a) | NBPF9, CNPY3, CHD9, CYTH4, ABI3, ADRB2 |
| Down-regulated microRNA interactions with up-regulated genes from (Kanduri et al., 2015a) | OSBP2 |
| Up-regulated microRNA | Validated and predicted interactions with upstream regulators of down-regulated genes from gene expression study (Kanduri et al., 2015a) |
| hsa-miR-222-3p | FOXP2 |
| hsa-miR-92a-3p | FOXP2 |
| hsa-miR-30a-5p and hsa-miR-30d-5p | NHLH2 |
| Down-regulated microRNA | Predicted interactions with upstream regulators of up-regulated genes from gene expression study (Kanduri et al., 2015a) |
| hsa-miR-6803-3p | FOSL1 |
Putative microrna regulatory network in professional music-performance
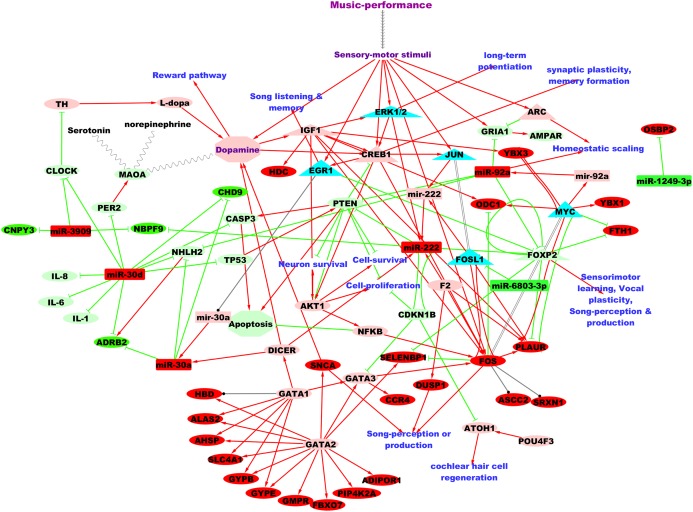
Figure 3 shows the putative microRNA regulatory network underlying 2 hours of professional music-performance generated from the integrated analysis with the RNA expression study (Kanduri et al., 2015a), incorporated with relevant findings from microRNA databases and the literature findings. Furthermore, the functional interactions between the genes which were up-regulated after music-performance (Kanduri et al., 2015a) from the integrated analysis (Fig. 3) and the putatively activated molecules from Fig. 3 (coral colour ellipses) were deduced using STRING and is given in Fig. S1. Interestingly, some of the molecules in the network were important for cell survival, cellular proliferation, neuron survival, long-term potentiation and dopamine signalling. Some of the molecules such as GATA2, GATA3 and miR-222 have functions in the inner ear development. Selected validated target genes from amongst this putative regulatory network (Fig. 3) are mentioned in detail in the Discussion.
Figure 3. Putative TF:microRNA:gene regulatory network in professional music-performance.
Candidate microRNAs are denoted by rectangles, target or other genes by ellipses and transcriptional regulators of candidate microRNAs by triangles. Green and red colour indicates down-regulation and up-regulation of the molecules (microRNAs and genes), respectively, after professional music-performance. Light-green and light-red colour nodes denote putative repression and activation, respectively, based on findings from microRNA databases and literature given in Discussion. Red arrows, green edges and black edges, respectively, show putative activation, repression and regulation. Solid and dotted arrows show direct and indirect interactions, respectively.
Discussion
Here, we report differential regulation of peripheral blood microRNAs in professional musicians post 2 hours of Western classical concert performance relative to a non-music control activity of the same duration using high-throughput sequencing and computational methods. Majority of the DE microRNAs were up-regulated, in accordance with microRNA regulation observed after singing in songbirds (Shi et al., 2013). The up-regulated microRNAs were found to repress apoptotic processes and inflammatory cytokines; activate (indirectly) the AKT signalling pathway and its transcriptional target, CREB (Du & Montminy, 1998), which are implicated in dopamine neurotransmission (Tan et al., 2008), cell survival (neuron), cellular proliferation and neuronal plasticity. These findings are consistent with the endogenous dopamine release observed during music-listening (Salimpoor et al., 2011) and the changes in brain plasticity identified in connection with professional music-training (Schlaug, 2001; Gaser & Schlaug, 2003; Groussard et al., 2010; Pantev & Herholz, 2011; Herholz & Zatorre, 2012). Here, we acknowledge that peripheral blood was used to study and interpret the changes in brain. This is facilitated by the highly similar (>80%) transcriptome shared by human peripheral whole blood and brain (Liew et al., 2006; Liang et al., 2007) and the positive correlation observed between peripheral blood and auditory forebrain transcriptomes (>70%) after song-listening in songbirds (Louder, Hauber & Balakrishnan, 2018). It is also plausible that environmental stimuli that elicit microRNA changes in the brain produce similar changes in the periphery, as illustrated in the brain-to-periphery model by Rao, Benito & Fischer (2013). Additionally, all the up-regulated microRNAs were expressed in the blood-brain barrier (Kalari et al., 2016) and various parts of the nervous system (Table S3), suggesting their brain expressions. In fact, multiple studies on human brain diseases have correlated the blood microRNA expression differences to their profile changes in the brain; for example, Parkinson’s disease (Martins et al., 2011), stroke (Liu et al., 2010) and Alzheimer’s (Leidinger et al., 2013). Accordingly, some of the observed microRNA changes and the regulatory mechanisms (Fig. 3) could reasonably reflect their changes in the brain.
We acknowledge here that factors that are not intrinsic to music such as the degree of emotional arousal of the participants during music-performance (Nakahara et al., 2011), stress level of the participants, familiarity of the music, mood of the musicians, pleasantness of the event etc. may have affected the results. To account for these, we collected some of these information from the music performers using a questionnaire (see Methods). Although the performing musicians reported to be slightly or moderately nervous before the performance, some of the music pieces they performed were very familiar to them and most musicians described their mood before and after the event as pleasant. We also acknowledge that these factors were not empirically studied in this work. Nonetheless, the long experience in performing as members of classical symphony orchestra (Tapiola sinfonietta) to the public suggested that the musicians were habituated to their venue of performance, well-versed with performing and had good rapport between their fellow musicians. Notably, all the musicians liked the music pieces they played, rehearsed the music pieces for the concert at the venue of their performance on the morning of the concert day and most musicians reported the whole event as relatively pleasant, supportive of the fact that these factors might have boosted their confidence, valence and quality of performance (Dib & Sturmey, 2011). Further focused studies using different settings are required to assess the effect of each of these different factors on the performing musicians.
Two of the microRNAs up-regulated after music-performance from this study, miR-30d and miR-222, corroborate findings from songbirds, the widely used neuroscientific model, in which they are differentially regulated in the auditory forebrain during song-learning/listening (Gunaratne et al., 2011). DE microRNAs from this study also show predicted interactions with some of the differentially regulated genes which are responsive to song-stimulated inhibition of miR-2954 (Gunaratne et al., 2011; Lin, Balakrishnan & Clayton, 2014). Further, putative target genes of miR-2954 and the up-regulated microRNAs from this study activate pathways and functions that promote cell survival and cellular proliferation related to neuronal plasticity (Gunaratne et al., 2011; Lin, Balakrishnan & Clayton, 2014).
The up-regulated microRNAs, hsa-miR-92a-3p and hsa-miR-222-3p target FOXP2, which plays crucial roles in human language or speech development (Mozzi et al., 2016) and song learning and singing in songbirds (Haesler et al., 2004; Teramitsu & White, 2006). In agreement with this, upstream regulators (microRNAs and EGR1) bind with FOXP2 promoter in singing songbirds, regulating the corticostriatal FOXP2 (Shi et al., 2013) for brain structure adaptations towards accurate song imitation (Heston & White, 2015), vocal complexity (Haesler et al., 2004) and stabilisation of mature song (Teramitsu et al., 2010). Further, FOXP2 is predominantly a transcriptional repressor and targets PLAUR, SELENBP1 and FTH1 (Vernes et al., 2007), which were up-regulated after music-performance (Kanduri et al., 2015a), suggesting indirect activation of FOXP2 target genes by microRNAs after music-performance.
miR-222 has been shown to be induced by ERK cascade (Terasawa et al., 2009), which is crucial for memory formation (Adams & Sweatt, 2002), striatal motor neuronal functions (Hutton et al., 2017) and neuronal plasticity (Kelleher et al., 2004). ERK–MAPK signalling pathway, with the help of cyclic adenosine monophosphate, is also involved in the proliferation of supporting cells of the auditory system (Bell & Oberholtzer, 2010). miR-222 also promotes neurite outgrowth by negatively regulating the neuronal growth inhibitor, PTEN, and by activating CREB expression and phosphorylation (Zhou et al., 2012). CREB signalling pathway modulates key processes involved in synaptic plasticity and memory formation and is activated by auditory-motor stimuli induced neuronal excitation (Waltereit & Weller, 2003). Interestingly, PTEN is also targeted by the up-regulated miR-92a (Zhang et al., 2014; Ke et al., 2015) and hippocampal PTEN knockdown inhibited neuronal apoptosis and CASP3 (caspase-3) (Zhu et al., 2006). Further, another candidate up-regulated microRNA, miR-30d also targets CASP3 and is implicated in auditory functions: miR-30d repression after acoustic over-stimulation caused increased caspase-3 expression and apoptosis induced sensory cell degeneration (Patel et al., 2013).
miR-92 and miR-30a are expressed in the brain and promote neuron differentiation (Sempere et al., 2004). miR-92a also plays a role in synaptic plasticity by targeting GRIA1, a glutamate receptor and component of AMPA receptor (AMPAR) that is activated by excitatory neurotransmission (Letellier et al., 2014). An immediate early gene, ARC, induced simultaneously by neuronal activity, is a key modulator of synaptic homeostatic scaling of AMPAR receptors and top candidate for musical ability (Oikkonen et al., 2016) located on 8q24, close to the up-regulated miR-30d (location:8q24.22).
miR-30 and miR-3909, up-regulated after music-performance, are also predicted to target CLOCK, a repressor of TH which converts tyrosine to the dopamine precursor, L-dopa (Parekh & McClung, 2015). miR-30 also targets, PER2, another CLOCK protein expressed in the striatum, which positively regulates MAOA expression in the mesolimbic dopaminergic system (Hampp et al., 2008; Parekh & McClung, 2015). MAOA is a critical enzyme that metabolises monoamines including serotonin, norepinephrine and dopamine and shows elevated expression in major depression disorders (Meyer et al., 2006). These findings are in accordance with the previous findings of activation of SNCA (important for neuron homeostasis, dopamine transmission, song perception of songbirds; Kimpo & Doupe, 1997; Wada et al., 2006) and dopaminergic pathway in connection with music/song perception and production (performance) in humans and in songbirds (zebra finch) (Whitney et al., 2014; Kanduri et al., 2015a, 2015b).
miR-222 is transcriptionally activated by FOSL1 (Wang et al., 2010), a member of the FOS family of transcription factors, which is co-expressed with FOS (up-regulated after music-performance), one of the most likely candidates associated with musical traits (Oikkonen et al., 2016). Also, FOSL1 and FOS are immediate early genes activated by neuronal and acoustic stimulation (Morgan & Curran, 1991) and FOSL1 is predicted to be regulated by hsa-miR-6803-3p. miR-222 also targets CDKN1B, the auditory hair cell cycle inhibitor (Walters et al., 2014; Chou et al., 2018), which encodes p27 protein with a direct repressive effect on GATA3, and indirectly activates GATA3 (Walters et al., 2017). Interestingly, GATA2 is located on the best associated region for musical aptitude (Oikkonen et al., 2015), transcriptionally activates GATA3 (Haugas et al., 2016) and SNCA co-expression network (Kanduri et al., 2015a, 2015b) and is essential for inner ear development (Lilleväli et al., 2004). Hence, putative repression of CDKN1B via miR-222 regulation might be important for the proper functioning of the auditory system (Walters et al., 2017).
Three of the up-regulated microRNAs in this study with a role in vascular remodelling (miR-222, miR-30d and miR-30a), were up-regulated after exercise (Baggish et al., 2011; McLean et al., 2015). This might reflect increased circulation because of the fine-tuning demanding physical activity and accurate motor functions while performing music by professional musicians. miR-30d-5p down-regulation and its inverse correlation between the levels of pro-inflammatory cytokines (IL-1, IL-6, IL-8 and others) were noted in severe sepsis (Caserta et al., 2016) suggesting that its up-regulation associated with music-performance might confer antagonistic effects consistent with findings from the RNA expression study (Kanduri et al., 2015a).
Here, we highlight some of the characteristics shared by professional performing musicians. Irrespective of their speciality, professional performing musicians usually start their music training at a very early age, undergoes rigorous training, constantly practice to maintain their standards excellent and share a multitude of structural and cognitive characteristics (Schlaug, 2001; Gaser & Schlaug, 2003; Meyer, Elmer & Jäncke, 2012; Herholz & Zatorre, 2012; Moore et al., 2014). For example, behavioural research indicate that consistent practice of a cognitive task, like music practice/performance by professional musicians, remodels it from controlled processing to an automated and effortless one with increased speed and accuracy (Jansma et al., 2001). Furthermore, analyses of the social data from musicians provide evidence that factors that might contribute to stress while performing music such as stage fright were found to be lowest in the professional musicians (Steptoe & Fidler, 1987).
Conclusions
To our knowledge, this is the first global expression profiling of human microRNAs to study professional music-performance at a concert. Our results provide new insights on behavioural microRNA regulatory mechanisms in human music genomics. Taken together, some of the candidate microRNAs and regulatory mechanisms associated with professional music-performance were found in songbird vocalisation and point to evolutionary conservation of sound perception and production. In addition, pathways and biological functions related to neurotransmission and sensorineural plasticity agree with previous studies on music perception and practice. As the genre of music and musical training were found to critically influence perceptual skills and neuronal processing of sound (Vuust et al., 2012), further studies with different genres of music-performance in different study populations are recommended. The impact of gene–environment interactions (including social interactions) on the microRNAs in professional musicians need to be further validated using animal models, preferably using songbirds. Specialised nature of the songbird neural circuits developed through singing experience resembles human music training and the social interaction involved in songbird singing has striking similarities to the non-verbal communication between professional musicians in performance, making songbird an ideal animal model for studying professional music-performance and other music-related human traits.
Supplemental Information
Nodes represent proteins. Edges represent protein–protein functional associations and the colour of the edges represent evidence including co-occurrence (Blue), co-expression (Black), experimental evidence (Purple), fusion evidence (Red), neighbourhood evidence (Green), database evidence (Light blue) and text mining (Yellow).
Acknowledgments
We are grateful to: Tapiola sinfonietta (https://www.tapiolasinfonietta.fi/en/frontpage/) for their participation; Professor Harri Lähdesmäki for the study design, bioinformatic analyses and critical reading of the manuscript; Dr. Eija Korpelainen and Dr. Chakravarthi Kanduri for critical reading of the manuscript; Hanna Kosonen, Helena Ruhkala, Sanna Pyy, Minna Varhala and Laura Salmela for expert technical help; and Wellcome Trust Centre for Human Genetics at the Oxford University, UK for the generation of the microRNA sequencing data.
Funding Statement
This work was supported by the Academy of Finland grant number 13371 and Biocentrum Helsinki Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information and Declarations
Competing Interests
The authors declare that they have no competing interests.
Author Contributions
Preethy Sasidharan Nair analysed the data, contributed reagents/materials/analysis tools, prepared figures and/or tables, authored or reviewed drafts of the paper, approved the final draft.
Tuire Kuusi conceived and designed the experiments, contributed reagents/materials/analysis tools, authored or reviewed drafts of the paper, approved the final draft.
Minna Ahvenainen performed the experiments, contributed reagents/materials/analysis tools, authored or reviewed drafts of the paper, approved the final draft.
Anju K. Philips performed the experiments, contributed reagents/materials/analysis tools, authored or reviewed drafts of the paper, approved the final draft.
Irma Järvelä conceived and designed the experiments, contributed reagents/materials/analysis tools, authored or reviewed drafts of the paper, approved the final draft.
Human Ethics
The following information was supplied relating to ethical approvals (i.e. approving body and any reference numbers):
The study was conducted in accordance with the guidelines of Helsinki Declaration and ethical permission (number 233/13/03/2013) has been approved by the Ethical Committee of Helsinki University Central Hospital. Additionally, written informed consent has been obtained from the participants.
Data Availability
The following information was supplied regarding data availability:
The data reported in this article have been deposited in the Gene Expression Omnibus database under the accession GSE115071.
References
- Adams & Sweatt (2002).Adams JP, Sweatt JD. Molecular psychology: roles for the ERK MAP kinase cascade in memory. Annual Review of Pharmacology and Toxicology. 2002;42(1):135–163. doi: 10.1146/annurev.pharmtox.42.082701.145401. [DOI] [PubMed] [Google Scholar]
- Agarwal et al. (2015).Agarwal V, Bell GW, Nam J-W, Bartel DP. Predicting effective microRNA target sites in mammalian mRNAs. eLife. 2015;4:e05005. doi: 10.7554/eLife.05005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altenmüller & Schlaug (2015).Altenmüller E, Schlaug G. Apollo’s gift: new aspects of neurologic music therapy. Progress in Brain Research. 2015;217:237–252. doi: 10.1016/bs.pbr.2014.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anders, Pyl & Huber (2015).Anders S, Pyl PT, Huber W. HTSeq-a Python framework to work with high-throughput sequencing data. Bioinformatics. 2015;31(2):166–169. doi: 10.1093/bioinformatics/btu638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Backes et al. (2016).Backes C, Khaleeq QT, Meese E, Keller A. miEAA: microRNA enrichment analysis and annotation. Nucleic Acids Research. 2016;44(W1):W110–W116. doi: 10.1093/nar/gkw345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baggish et al. (2011).Baggish AL, Hale A, Weiner RB, Lewis GD, Systrom D, Wang F, Wang TJ, Chan SY. Dynamic regulation of circulating microRNA during acute exhaustive exercise and sustained aerobic exercise training. Journal of Physiology. 2011;589(16):3983–3994. doi: 10.1113/jphysiol.2011.213363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beech et al. (2014).Beech RD, Leffert JJ, Lin A, Hong KA, Hansen J, Umlauf S, Mane S, Zhao H, Sinha R. Stress-related alcohol consumption in heavy drinkers correlates with expression of miR-10a, miR-21, and components of the TAR-RNA-binding protein-associated complex. Alcoholism: Clinical and Experimental Research. 2014;38(11):2743–2753. doi: 10.1111/acer.12549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell & Oberholtzer (2010).Bell TJ, Oberholtzer JC. cAMP-induced auditory supporting cell proliferation is mediated by ERK MAPK signaling pathway. Journal of the Association for Research in Otolaryngology. 2010;11(2):173–185. doi: 10.1007/s10162-009-0205-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black et al. (2013).Black DS, Cole SW, Irwin MR, Breen E, St.Cyr NM, Nazarian N, Khalsa DS, Lavretsky H. Yogic meditation reverses NF-κB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology. 2013;38(3):348–355. doi: 10.1016/j.psyneuen.2012.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bleazard, Lamb & Griffiths-Jones (2015).Bleazard T, Lamb JA, Griffiths-Jones S. Bias in microRNA functional enrichment analysis. Bioinformatics. 2015;31(10):1592–1598. doi: 10.1093/bioinformatics/btv023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caserta et al. (2016).Caserta S, Kern F, Cohen J, Drage S, Newbury SF, Llewelyn MJ. Circulating plasma microRNAs can differentiate human sepsis and Systemic Inflammatory Response Syndrome (SIRS) Scientific Reports. 2016;6(1):28006. doi: 10.1038/srep28006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou et al. (2018).Chou C-H, Shrestha S, Yang C-D, Chang N-W, Lin Y-L, Liao K-W, Huang W-C, Sun T-H, Tu S-J, Lee W-H, Chiew M-Y, Tai C-S, Wei T-Y, Tsai T-R, Huang H-T, Wang C-Y, Wu H-Y, Ho S-Y, Chen P-R, Chuang C-H, Hsieh P-J, Wu Y-S, Chen W-L, Li M-J, Wu Y-C, Huang X-Y, Ng FL, Buddhakosai W, Huang P-C, Lan K-C, Huang C-Y, Weng S-L, Cheng Y-N, Liang C, Hsu W-L, Huang H-D. miRTarBase update 2018: a resource for experimentally validated microRNA-target interactions. Nucleic Acids Research. 2018;46(D1):D296–D302. doi: 10.1093/nar/gkx1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dib & Sturmey (2011).Dib NE, Sturmey P. Effects of general-case training, instructions, rehearsal, and feedback on the reduction of sight-reading errors by competent musicians. Journal of Applied behavior Analysis. 2011;44(3):599–604. doi: 10.1901/jaba.2011.44-599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du & Montminy (1998).Du K, Montminy M. CREB is a regulatory target for the protein kinase Akt/PKB. Journal of Biological Chemistry. 1998;273(49):32377–32379. doi: 10.1074/jbc.273.49.32377. [DOI] [PubMed] [Google Scholar]
- Elbert et al. (1995).Elbert T, Pantev C, Wienbruch C, Rockstroh B, Taub E. Increased cortical representation of the fingers of the left hand in string players. Science. 1995;270(5234):305–307. doi: 10.1126/science.270.5234.305. [DOI] [PubMed] [Google Scholar]
- Gaser & Schlaug (2003).Gaser C, Schlaug G. Brain structures differ between musicians and non-musicians. Journal of Neuroscience. 2003;23(27):9240–9245. doi: 10.1523/JNEUROSCI.23-27-09240.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groussard et al. (2010).Groussard M, La Joie R, Rauchs G, Landeau B, Chételat G, Viader F, Desgranges B, Eustache F, Platel H. When music and long-term memory interact: Effects of musical expertise on functional and structural plasticity in the hippocampus. PLOS ONE. 2010;5(10):e13225. doi: 10.1371/journal.pone.0013225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunaratne et al. (2011).Gunaratne PH, Lin Y-C, Benham AL, Drnevich J, Coarfa C, Tennakoon JB, Creighton CJ, Kim JH, Milosavljevic A, Watson M, Griffiths-Jones S, Clayton DF. Song exposure regulates known and novel microRNAs in the zebra finch auditory forebrain. BMC Genomics. 2011;12(1):277. doi: 10.1186/1471-2164-12-277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haesler et al. (2004).Haesler S, Wada K, Nshdejan A, Morrisey EE, Lints T, Jarvis ED, Scharff C. FoxP2 expression in avian vocal learners and non-learners. Journal of Neuroscience. 2004;24(13):3164–3175. doi: 10.1523/JNEUROSCI.4369-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hampp et al. (2008).Hampp G, Ripperger JA, Houben T, Schmutz I, Blex C, Perreau-Lenz S, Brunk I, Spanagel R, Ahnert-Hilger G, Meijer JH, Albrecht U. Regulation of monoamine oxidase A by circadian-clock components implies clock influence on mood. Current Biology. 2008;18(9):678–683. doi: 10.1016/j.cub.2008.04.012. [DOI] [PubMed] [Google Scholar]
- Haugas et al. (2016).Haugas M, Tikker L, Achim K, Salminen M, Partanen J. Gata2 and Gata3 regulate the differentiation of serotonergic and glutamatergic neuron subtypes of the dorsal raphe. Development. 2016;143(23):4495–4508. doi: 10.1242/dev.136614. [DOI] [PubMed] [Google Scholar]
- Hayashi et al. (2017).Hayashi Y, Yamamoto N, Nakagawa T, Omori K, Ito J. Activation of IGF1 signaling in the cochlea induces the transcription of its mediators during the protection of cochlear hair cells against aminoglycoside. Otology & Neurotology. 2017;38(2):278–282. doi: 10.1097/MAO.0000000000001276. [DOI] [PubMed] [Google Scholar]
- Herholz & Zatorre (2012).Herholz SC, Zatorre RJ. Musical training as a framework for brain plasticity: behavior, function, and structure. Neuron. 2012;76(3):486–502. doi: 10.1016/j.neuron.2012.10.011. [DOI] [PubMed] [Google Scholar]
- Heston & White (2015).Heston JB, White SA. Behavior-linked FoxP2 regulation enables zebra finch vocal learning. Journal of Neuroscience. 2015;35(7):2885–2894. doi: 10.1523/JNEUROSCI.3715-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilliard et al. (2012).Hilliard AT, Miller JE, Fraley ER, Horvath S, White SA. Molecular microcircuitry underlies functional specification in a basal ganglia circuit dedicated to vocal learning. Neuron. 2012;73(3):537–552. doi: 10.1016/j.neuron.2012.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huntley et al. (2018).Huntley RP, Kramarz B, Sawford T, Umrao Z, Kalea A, Acquaah V, Martin MJ, Mayr M, Lovering RC. Expanding the horizons of microRNA bioinformatics. RNA. 2018;24(8):1005–1017. doi: 10.1261/rna.065565.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huntzinger & Izaurralde (2011).Huntzinger E, Izaurralde E. Gene silencing by microRNAs: contributions of translational repression and mRNA decay. Nature Reviews Genetics. 2011;12(2):99–110. doi: 10.1038/nrg2936. [DOI] [PubMed] [Google Scholar]
- Hutton et al. (2017).Hutton SR, Otis JM, Kim EM, Lamsal Y, Stuber GD, Snider WD. ERK/MAPK signaling is required for pathway-specific striatal motor functions. Journal of Neuroscience. 2017;37(34):8102–8115. doi: 10.1523/JNEUROSCI.0473-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyde et al. (2009).Hyde KL, Lerch J, Norton A, Forgeard M, Winner E, Evans AC, Schlaug G. Musical training shapes structural brain development. Journal of Neuroscience. 2009;29(10):3019–3025. doi: 10.1523/JNEUROSCI.5118-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jansma et al. (2001).Jansma JM, Ramsey NF, Slagter HA, Kahn RS. Functional anatomical correlates of controlled and automatic processing. Journal of Cognitive Neuroscience. 2001;13(6):730–743. doi: 10.1162/08989290152541403. [DOI] [PubMed] [Google Scholar]
- Jelena et al. (2014).Jelena B, Christina L, Eric V, Fabiola Q-R. Phenotypic variability in Waardenburg syndrome resulting from a 22q12.3-q13.1 microdeletion involving SOX10. American Journal of Medical Genetics Part A. 2014;164(6):1512–1519. doi: 10.1002/ajmg.a.36446. [DOI] [PubMed] [Google Scholar]
- Kalari et al. (2016).Kalari KR, Thompson KJ, Nair AA, Tang X, Bockol MA, Jhawar N, Swaminathan SK, Lowe VJ, Kandimalla KK. BBBomics-human blood brain barrier transcriptomics hub. Frontiers in Neuroscience. 2016;10:71. doi: 10.3389/fnins.2016.00071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanduri et al. (2015a).Kanduri C, Kuusi T, Ahvenainen M, Philips AK, Lähdesmäki H, Järvelä I. The effect of music performance on the transcriptome of professional musicians. Scientific Reports. 2015a;5(1):9506. doi: 10.1038/srep09506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanduri et al. (2015b).Kanduri C, Raijas P, Ahvenainen M, Philips AK, Ukkola-Vuoti L, Lähdesmäki H, Järvelä I. The effect of listening to music on human transcriptome. PeerJ. 2015b;3(2):e830. doi: 10.7717/peerj.830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ke et al. (2015).Ke T-W, Wei P-L, Yeh K-T, Chen WT-L, Cheng Y-W. MiR-92a promotes cell metastasis of colorectal cancer through PTEN-mediated PI3K/AKT pathway. Annals of Surgical Oncology. 2015;22(8):2649–2655. doi: 10.1245/s10434-014-4305-2. [DOI] [PubMed] [Google Scholar]
- Kelleher et al. (2004).Kelleher RJ, III, Govindarajan A, Jung H-Y, Kang H, Tonegawa S. Translational control by MAPK signaling in long-term synaptic plasticity and memory. Cell. 2004;116(3):467–479. doi: 10.1016/S0092-8674(04)00115-1. [DOI] [PubMed] [Google Scholar]
- Khang & Lau (2015).Khang TF, Lau CY. Getting the most out of RNA-seq data analysis. PeerJ. 2015;3(Suppl 9):e1360. doi: 10.7717/peerj.1360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimpo & Doupe (1997).Kimpo RR, Doupe AJ. FOS is induced by singing in distinct neuronal populations in a motor network. Neuron. 1997;18(2):315–325. doi: 10.1016/S0896-6273(00)80271-8. [DOI] [PubMed] [Google Scholar]
- Kozomara & Griffiths-Jones (2014).Kozomara A, Griffiths-Jones S. miRBase: annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Research. 2014;42(D1):D68–D73. doi: 10.1093/nar/gkt1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langmead et al. (2009).Langmead B, Trapnell C, Pop M, Salzberg SL. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biology. 2009;10(3):R25. doi: 10.1186/gb-2009-10-3-r25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leidinger et al. (2013).Leidinger P, Backes C, Deutscher S, Schmitt K, Mueller SC, Frese K, Haas J, Ruprecht K, Paul F, Stähler C, Lang CJG, Meder B, Bartfai T, Meese E, Keller A. A blood based 12-miRNA signature of Alzheimer disease patients. Genome Biology. 2013;14(7):R78. doi: 10.1186/gb-2013-14-7-r78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letellier et al. (2014).Letellier M, Elramah S, Mondin M, Soula A, Penn A, Choquet D, Landry M, Thoumine O, Favereaux A. miR-92a regulates expression of synaptic GluA1-containing AMPA receptors during homeostatic scaling. Nature Neuroscience. 2014;17(8):1040–1042. doi: 10.1038/nn.3762. [DOI] [PubMed] [Google Scholar]
- Li et al. (2018).Li J, Han X, Wan Y, Zhang S, Zhao Y, Fan R, Cui Q, Zhou Y. TAM 2.0: tool for MicroRNA set analysis. Nucleic Acids Research. 2018;46(W1):W180–W185. doi: 10.1093/nar/gky509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li et al. (2017).Li C, Zhang K, Chen J, Chen L, Wang R, Chu X. MicroRNAs as regulators and mediators of forkhead box transcription factors function in human cancers. Oncotarget. 2017;8(7):12433–12450. doi: 10.18632/oncotarget.14015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang et al. (2007).Liang Y, Ridzon D, Wong L, Chen C. Characterization of microRNA expression profiles in normal human tissues. BMC Genomics. 2007;8(1):166. doi: 10.1186/1471-2164-8-166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew et al. (2006).Liew C-C, Ma J, Tang H-C, Zheng R, Dempsey AA. The peripheral blood transcriptome dynamically reflects system wide biology: a potential diagnostic tool. Journal of Laboratory and Clinical Medicine. 2006;147(3):126–132. doi: 10.1016/j.lab.2005.10.005. [DOI] [PubMed] [Google Scholar]
- Lilleväli et al. (2004).Lilleväli K, Matilainen T, Karis A, Salminen M. Partially overlapping expression of Gata2 and Gata3 during inner ear development. Developmental Dynamics. 2004;231(4):775–781. doi: 10.1002/dvdy.20185. [DOI] [PubMed] [Google Scholar]
- Lin, Balakrishnan & Clayton (2014).Lin Y-C, Balakrishnan CN, Clayton DF. Functional genomic analysis and neuroanatomical localization of miR-2954, a song-responsive sex-linked microRNA in the zebra finch. Frontiers in Neuroscience. 2014;8(43):409. doi: 10.3389/fnins.2014.00409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu et al. (2010).Liu D-Z, Tian Y, Ander BP, Xu H, Stamova BS, Zhan X, Turner RJ, Jickling G, Sharp FR. Brain and blood microRNA expression profiling of ischemic stroke, intracerebral hemorrhage, and kainate seizures. Journal of Cerebral Blood Flow & Metabolism. 2010;30(1):92–101. doi: 10.1038/jcbfm.2009.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Louder, Hauber & Balakrishnan (2018).Louder MIM, Hauber ME, Balakrishnan CN. Early social experience alters transcriptomic responses to species-specific song stimuli in female songbirds. Behavioural Brain Research. 2018;347:69–76. doi: 10.1016/j.bbr.2018.02.034. [DOI] [PubMed] [Google Scholar]
- Love, Huber & Anders (2014).Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biology. 2014;15(12):550. doi: 10.1186/s13059-014-0550-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martins et al. (2011).Martins M, Rosa A, Guedes LC, Fonseca BV, Gotovac K, Violante S, Mestre T, Coelho M, Rosa MM, Martin ER, Vance JM, Outeiro TF, Wang L, Borovecki F, Ferreira JJ, Oliveira SA. Convergence of miRNA expression profiling, α-synuclein interaction and GWAS in Parkinson’s disease. PLOS ONE. 2011;6(10):e25443. doi: 10.1371/journal.pone.0025443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLean et al. (2015).McLean CS, Mielke C, Cordova JM, Langlais PR, Bowen B, Miranda D, Coletta DK, Mandarino LJ. Gene and microRNA expression responses to exercise; relationship with insulin sensitivity. PLOS ONE. 2015;10(5):e0127089. doi: 10.1371/journal.pone.0127089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer, Elmer & Jäncke (2012).Meyer M, Elmer S, Jäncke L. Musical expertise induces neuroplasticity of the planum temporale. Annals of the New York Academy of Sciences. 2012;1252(1):116–123. doi: 10.1111/j.1749-6632.2012.06450.x. [DOI] [PubMed] [Google Scholar]
- Meyer et al. (2006).Meyer JH, Ginovart N, Boovariwala A, Sagrati S, Hussey D, Garcia A, Young T, Praschak-Rieder N, Wilson AA, Houle S. Elevated monoamine oxidase a levels in the brain: an explanation for the monoamine imbalance of major depression. Archives of General Psychiatry. 2006;63(11):1209–1216. doi: 10.1001/archpsyc.63.11.1209. [DOI] [PubMed] [Google Scholar]
- Moore et al. (2014).Moore E, Schaefer R, Bastin M, Roberts N, Overy K. Can musical training influence brain connectivity? Evidence from diffusion tensor MRI. Brain Sciences. 2014;4(2):405–427. doi: 10.3390/brainsci4020405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan & Curran (1991).Morgan JI, Curran T. Stimulus-transcription coupling in the nervous system: involvement of the inducible proto-oncogenes fos and jun. Annual Review of Neuroscience. 1991;14(1):421–451. doi: 10.1146/annurev.ne.14.030191.002225. [DOI] [PubMed] [Google Scholar]
- Mozzi et al. (2016).Mozzi A, Forni D, Clerici M, Pozzoli U, Mascheretti S, Guerini FR, Riva S, Bresolin N, Cagliani R, Sironi M. The evolutionary history of genes involved in spoken and written language: beyond FOXP2. Scientific Reports. 2016;6(1):22157. doi: 10.1038/srep22157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakahara et al. (2011).Nakahara H, Furuya S, Masuko T, Francis PR, Kinoshita H. Performing music can induce greater modulation of emotion-related psychophysiological responses than listening to music. International Journal of Psychophysiology. 2011;81(3):152–158. doi: 10.1016/j.ijpsycho.2011.06.003. [DOI] [PubMed] [Google Scholar]
- Oechslin et al. (2013).Oechslin MS, Van De Ville D, Lazeyras F, Hauert C-A, James CE. Degree of musical expertise modulates higher order brain functioning. Cerebral Cortex. 2013;23(9):2213–2224. doi: 10.1093/cercor/bhs206. [DOI] [PubMed] [Google Scholar]
- Oikkonen et al. (2015).Oikkonen J, Huang Y, Onkamo P, Ukkola-Vuoti L, Raijas P, Karma K, Vieland VJ, Järvelä I. A genome-wide linkage and association study of musical aptitude identifies loci containing genes related to inner ear development and neurocognitive functions. Molecular Psychiatry. 2015;20(2):275–282. doi: 10.1038/mp.2014.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oikkonen et al. (2016).Oikkonen J, Onkamo P, Järvelä I, Kanduri C. Convergent evidence for the molecular basis of musical traits. Scientific Reports. 2016;6(1):39707. doi: 10.1038/srep39707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olde Loohuis et al. (2012).Olde Loohuis NFM, Kos A, Martens GJM, Van Bokhoven H, Nadif Kasri N, Aschrafi A. MicroRNA networks direct neuronal development and plasticity. Cellular and Molecular Life Sciences. 2012;69(1):89–102. doi: 10.1007/s00018-011-0788-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osterndorff-Kahanek et al. (2018).Osterndorff-Kahanek EA, Tiwari GR, Lopez MF, Becker HC, Harris RA, Mayfield RD. Long-term ethanol exposure: temporal pattern of microRNA expression and associated mRNA gene networks in mouse brain. PLOS ONE. 2018;13(1):e0190841. doi: 10.1371/journal.pone.0190841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otaegi et al. (2011).Otaegi G, Pollock A, Hong J, Sun T. MicroRNA miR-9 modifies motor neuron columns by a tuning regulation of FoxP1 levels in developing spinal cords. Journal of Neuroscience. 2011;31(3):809–818. doi: 10.1523/JNEUROSCI.4330-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pallesen et al. (2010).Pallesen KJ, Brattico E, Bailey CJ, Korvenoja A, Koivisto J, Gjedde A, Carlson S. Cognitive control in auditory working memory is enhanced in musicians. PLOS ONE. 2010;5(6):e11120. doi: 10.1371/journal.pone.0011120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantev & Herholz (2011).Pantev C, Herholz SC. Plasticity of the human auditory cortex related to musical training. Neuroscience & Biobehavioral Reviews. 2011;35(10):2140–2154. doi: 10.1016/j.neubiorev.2011.06.010. [DOI] [PubMed] [Google Scholar]
- Parekh & McClung (2015).Parekh PK, McClung CA. Circadian mechanisms underlying reward-related neurophysiology and synaptic plasticity. Frontiers in Psychiatry. 2015;6:187. doi: 10.3389/fpsyt.2015.00187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park et al. (2014).Park M, Gutyrchik E, Bao Y, Zaytseva Y, Carl P, Welker L, Pöppel E, Reiser M, Blautzik J, Meindl T. Differences between musicians and non-musicians in neuro-affective processing of sadness and fear expressed in music. Neuroscience Letters. 2014;566:120–124. doi: 10.1016/j.neulet.2014.02.041. [DOI] [PubMed] [Google Scholar]
- Park et al. (2012).Park H, Lee S, Kim H-J, Ju YS, Shin J-Y, Hong D, Von Grotthuss M, Lee D-S, Park C, Kim JH, Kim B, Yoo YJ, Cho S-I, Sung J, Lee C, Kim J-I, Seo J-S. Comprehensive genomic analyses associate UGT8 variants with musical ability in a Mongolian population. Journal of Medical Genetics. 2012;49(12):747–752. doi: 10.1136/jmedgenet-2012-101209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel et al. (2013).Patel M, Cai Q, Ding D, Salvi R, Hu Z, Hu BH. The miR-183/Taok1 target pair is implicated in cochlear responses to acoustic trauma. PLOS ONE. 2013;8(3):e58471. doi: 10.1371/journal.pone.0058471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pulli et al. (2008).Pulli K, Karma K, Norio R, Sistonen P, Göring HHH, Järvelä I. Genome-wide linkage scan for loci of musical aptitude in Finnish families: evidence for a major locus at 4q22. Journal of Medical Genetics. 2008;45(7):451–456. doi: 10.1136/jmg.2007.056366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radom-Aizik et al. (2014).Radom-Aizik S, Zaldivar FP, Jr, Haddad F, Cooper DM. Impact of brief exercise on circulating monocyte gene and microRNA expression: implications for atherosclerotic vascular disease. Brain, Behavior, and Immunity. 2014;39:121–129. doi: 10.1016/j.bbi.2014.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radom-Aizik et al. (2012).Radom-Aizik S, Zaldivar F, Jr, Leu S-Y, Adams GR, Oliver S, Cooper DM. Effects of exercise on microRNA expression in young males peripheral blood mononuclear cells. Clinical and Translational Science. 2012;5(1):32–38. doi: 10.1111/j.1752-8062.2011.00384.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rance (2005).Rance G. Auditory neuropathy/Dys-synchrony and its perceptual consequences. Trends in Amplification. 2005;9(1):1–43. doi: 10.1177/108471380500900102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao, Benito & Fischer (2013).Rao P, Benito E, Fischer A. MicroRNAs as biomarkers for CNS disease. Frontiers in Molecular Neuroscience. 2013;6:39. doi: 10.3389/fnmol.2013.00039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Regehr (2012).Regehr WG. Short-term presynaptic plasticity. Cold Spring Harbor Perspectives in Biology. 2012;4(7):a005702. doi: 10.1101/cshperspect.a005702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salimpoor et al. (2011).Salimpoor VN, Benovoy M, Larcher K, Dagher A, Zatorre RJ. Anatomically distinct dopamine release during anticipation and experience of peak emotion to music. Nature Neuroscience. 2011;14(2):257–262. doi: 10.1038/nn.2726. [DOI] [PubMed] [Google Scholar]
- Schlaug (2001).Schlaug G. The brain of musicians. A model for functional and structural adaptation. Annals of the New York Academy of Sciences. 2001;930(1):281–299. doi: 10.1111/j.1749-6632.2001.tb05739.x. [DOI] [PubMed] [Google Scholar]
- Schurch et al. (2016).Schurch NJ, Schofield P, Gierliński M, Cole C, Sherstnev A, Singh V, Wrobel N, Gharbi K, Simpson GG, Owen-Hughes T, Blaxter M, Barton GJ. How many biological replicates are needed in an RNA-seq experiment and which differential expression tool should you use? RNA. 2016;22(6):839–851. doi: 10.1261/rna.053959.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sempere et al. (2004).Sempere LF, Freemantle S, Pitha-Rowe I, Moss E, Dmitrovsky E, Ambros V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biology. 2004;5(3):R13. doi: 10.1186/gb-2004-5-3-r13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shannon et al. (2003).Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Research. 2003;13(11):2498–2504. doi: 10.1101/gr.1239303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi et al. (2013).Shi Z, Luo G, Fu L, Fang Z, Wang X, Li X. miR-9 and miR-140-5p target FoxP2 and are regulated as a function of the social context of singing behavior in zebra finches. Journal of Neuroscience. 2013;33(42):16510–16521. doi: 10.1523/JNEUROSCI.0838-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimoyama et al. (2015).Shimoyama M, De Pons J, Hayman GT, Laulederkind SJF, Liu W, Nigam R, Petri V, Smith JR, Tutaj M, Wang S-J, Worthey E, Dwinell M, Jacob H. The rat genome database 2015: genomic, phenotypic and environmental variations and disease. Nucleic Acids Research. 2015;43(D1):D743–D750. doi: 10.1093/nar/gku1026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith et al. (2018).Smith CL, Blake JA, Kadin JA, Richardson JE, Bult CJ, the Mouse Genome Database Group Mouse Genome Database (MGD)-2018: knowledgebase for the laboratory mouse. Nucleic Acids Research. 2018;46(D1):D836–D842. doi: 10.1093/nar/gkx1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steele et al. (2013).Steele CJ, Bailey JA, Zatorre RJ, Penhune VB. Early musical training and white-matter plasticity in the corpus callosum: evidence for a sensitive period. Journal of Neuroscience. 2013;33(3):1282–1290. doi: 10.1523/JNEUROSCI.3578-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steptoe & Fidler (1987).Steptoe A, Fidler H. Stage fright in orchestral musicians: a study of cognitive and behavioural strategies in performance anxiety. British Journal of Psychology. 1987;78(Pt 2):241–249. doi: 10.1111/j.2044-8295.1987.tb02243.x. [DOI] [PubMed] [Google Scholar]
- Szklarczyk et al. (2017).Szklarczyk D, Morris JH, Cook H, Kuhn M, Wyder S, Simonovic M, Santos A, Doncheva NT, Roth A, Bork P, Jensen LJ, Von Mering C. The STRING database in 2017: quality-controlled protein–protein association networks, made broadly accessible. Nucleic Acids Research. 2017;45(D1):D362–D368. doi: 10.1093/nar/gkw937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan et al. (2008).Tan H-Y, Nicodemus KK, Chen Q, Li Z, Brooke JK, Honea R, Kolachana BS, Straub RE, Meyer-Lindenberg A, Sei Y, Mattay VS, Callicott JH, Weinberger DR. Genetic variation in AKT1 is linked to dopamine-associated prefrontal cortical structure and function in humans. Journal of Clinical Investigation. 2008;118(6):2200–2208. doi: 10.1172/JCI34725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang et al. (2015).Tang M, Sun J, Shimizu K, Kadota K. Evaluation of methods for differential expression analysis on multi-group RNA-seq count data. BMC Bioinformatics. 2015;16(1):360. doi: 10.1186/s12859-015-0794-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teramitsu et al. (2010).Teramitsu I, Poopatanapong A, Torrisi S, White SA. Striatal FoxP2 is actively regulated during songbird sensorimotor learning. PLOS ONE. 2010;5(1):e8548. doi: 10.1371/journal.pone.0008548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teramitsu & White (2006).Teramitsu I, White SA. FoxP2 regulation during undirected singing in adult songbirds. Journal of Neuroscience. 2006;26(28):7390–7394. doi: 10.1523/JNEUROSCI.1662-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terasawa et al. (2009).Terasawa K, Ichimura A, Sato F, Shimizu K, Tsujimoto G. Sustained activation of ERK1/2 by NGF induces microRNA-221 and 222 in PC12 cells. FEBS Journal. 2009;276(12):3269–3276. doi: 10.1111/j.1742-4658.2009.07041.x. [DOI] [PubMed] [Google Scholar]
- Theusch, Basu & Gitschier (2009).Theusch E, Basu A, Gitschier J. Genome-wide study of families with absolute pitch reveals linkage to 8q24.21 and locus heterogeneity. American Journal of Human Genetics. 2009;85(1):112–119. doi: 10.1016/j.ajhg.2009.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ukkola-Vuoti et al. (2013).Ukkola-Vuoti L, Kanduri C, Oikkonen J, Buck G, Blancher C, Raijas P, Karma K, Lähdesmäki H, Järvelä I. Genome-wide copy number variation analysis in extended families and unrelated individuals characterized for musical aptitude and creativity in music. PLOS ONE. 2013;8(2):e56356. doi: 10.1371/journal.pone.0056356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vernes et al. (2007).Vernes SC, Spiteri E, Nicod J, Groszer M, Taylor JM, Davies KE, Geschwind DH, Fisher SE. High-throughput analysis of promoter occupancy reveals direct neural targets of FOXP2, a gene mutated in speech and language disorders. American Journal of Human Genetics. 2007;81(6):1232–1250. doi: 10.1086/522238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volpe et al. (2016).Volpe G, D’Ausilio A, Badino L, Camurri A, Fadiga L. Measuring social interaction in music ensembles. Philosophical Transactions of the Royal Society B: Biological Sciences. 2016;371(1693):20150377. doi: 10.1098/rstb.2015.0377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vuust et al. (2012).Vuust P, Brattico E, Seppänen M, Näätänen R, Tervaniemi M. Practiced musical style shapes auditory skills. Annals of the New York Academy of Sciences. 2012;1252(1):139–146. doi: 10.1111/j.1749-6632.2011.06409.x. [DOI] [PubMed] [Google Scholar]
- Wada et al. (2006).Wada K, Howard JT, McConnell P, Whitney O, Lints T, Rivas MV, Horita H, Patterson MA, White SA, Scharff C, Haesler S, Zhao S, Sakaguchi H, Hagiwara M, Shiraki T, Hirozane-Kishikawa T, Skene P, Hayashizaki Y, Carninci P, Jarvis ED. A molecular neuroethological approach for identifying and characterizing a cascade of behaviorally regulated genes. Proceedings of the National Academy of Sciences of the United States of America. 2006;103(41):15212–15217. doi: 10.1073/pnas.0607098103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waltereit & Weller (2003).Waltereit R, Weller M. Signaling from cAMP/PKA to MAPK and synaptic plasticity. Molecular Neurobiology. 2003;27(1):99–106. doi: 10.1385/mn:27:1:99. [DOI] [PubMed] [Google Scholar]
- Walters et al. (2017).Walters BJ, Coak E, Dearman J, Bailey G, Yamashita T, Kuo B, Zuo J. In vivo interplay between p27(Kip1), GATA3, ATOH1, and POU4F3 converts non-sensory cells to hair cells in adult mice. Cell Reports. 2017;19(2):307–320. doi: 10.1016/j.celrep.2017.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walters et al. (2014).Walters BJ, Liu Z, Crabtree M, Coak E, Cox BC, Zuo J. Auditory hair cell-specific deletion of p27Kip1 in postnatal mice promotes cell-autonomous generation of new hair cells and normal hearing. Journal of Neuroscience. 2014;34(47):15751–15763. doi: 10.1523/JNEUROSCI.3200-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang et al. (2015).Wang L, Cheng S, Yin Z, Xu C, Lu S, Hou J, Yu T, Zhu X, Zou X, Peng Y, Xu Y, Yang Z, Chen G. Conditional inactivation of Akt three isoforms causes tau hyperphosphorylation in the brain. Molecular Neurodegeneration. 2015;10(1):33. doi: 10.1186/s13024-015-0030-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Kwon & Tsai (2012).Wang W, Kwon EJ, Tsai L-H. MicroRNAs in learning, memory, and neurological diseases. Learning & Memory. 2012;19(9):359–368. doi: 10.1101/lm.026492.112. [DOI] [PubMed] [Google Scholar]
- Wang et al. (2010).Wang J, Lu M, Qiu C, Cui Q. TransmiR: a transcription factor-microRNA regulation database. Nucleic Acids Research. 2010;38(suppl_1):D119–D122. doi: 10.1093/nar/gkp803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitney et al. (2014).Whitney O, Pfenning AR, Howard JT, Blatti CA, Liu F, Ward JM, Wang R, Audet J-N, Kellis M, Mukherjee S, Sinha S, Hartemink AJ, West AE, Jarvis ED. Core and region-enriched networks of behaviorally regulated genes and the singing genome. Science. 2014;346(6215):1256780. doi: 10.1126/science.1256780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, Wang & Wu (2015).Wu H, Wang C, Wu Z. PROPER: comprehensive power evaluation for differential expression using RNA-seq. Bioinformatics. 2015;31(2):233–241. doi: 10.1093/bioinformatics/btu640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zatorre, Chen & Penhune (2007).Zatorre RJ, Chen JL, Penhune VB. When the brain plays music: auditory-motor interactions in music perception and production. Nature Reviews Neuroscience. 2007;8(7):547–558. doi: 10.1038/nrn2152. [DOI] [PubMed] [Google Scholar]
- Zhang et al. (2014).Zhang L, Zhou M, Qin G, Weintraub NL, Tang Y. MiR-92a regulates viability and angiogenesis of endothelial cells under oxidative stress. Biochemical and Biophysical Research Communications. 2014;446(4):952–958. doi: 10.1016/j.bbrc.2014.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, Lindsay & Robinson (2014).Zhou X, Lindsay H, Robinson MD. Robustly detecting differential expression in RNA sequencing data using observation weights. Nucleic Acids Research. 2014;42(11):e91. doi: 10.1093/nar/gku310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou et al. (2012).Zhou S, Shen D, Wang Y, Gong L, Tang X, Yu B, Gu X, Ding F. microRNA-222 targeting PTEN promotes neurite outgrowth from adult dorsal root ganglion neurons following sciatic nerve transection. PLOS ONE. 2012;7(9):e44768. doi: 10.1371/journal.pone.0044768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu et al. (2006).Zhu Y, Hoell P, Ahlemeyer B, Krieglstein J. PTEN: a crucial mediator of mitochondria-dependent apoptosis. Apoptosis. 2006;11(2):197–207. doi: 10.1007/s10495-006-3714-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Nodes represent proteins. Edges represent protein–protein functional associations and the colour of the edges represent evidence including co-occurrence (Blue), co-expression (Black), experimental evidence (Purple), fusion evidence (Red), neighbourhood evidence (Green), database evidence (Light blue) and text mining (Yellow).
Data Availability Statement
The following information was supplied regarding data availability:
The data reported in this article have been deposited in the Gene Expression Omnibus database under the accession GSE115071.