Abstract
Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a rare and aggressive hematologic malignancy for which there is still no effective therapy. In order to identify genetic alterations useful for a new treatment design, we used whole-exome sequencing to analyze 14 BPDCN patients and the patient-derived CAL-1 cell line. The functional enrichment analysis of mutational data reported the epigenetic regulatory program to be the most significantly undermined (P<0.0001). In particular, twenty-five epigenetic modifiers were found mutated (e.g. ASXL1, TET2, SUZ12, ARID1A, PHF2, CHD8); ASXL1 was the most frequently affected (28.6% of cases). To evaluate the impact of the identified epigenetic mutations at the gene-expression and Histone H3 lysine 27 trimethylation/acetylation levels, we performed additional RNA and pathology tissue-chromatin immunoprecipitation sequencing experiments. The patients displayed enrichment in gene signatures regulated by methylation and modifiable by decitabine administration, shared common H3K27-acetylated regions, and had a set of cell-cycle genes aberrantly up-regulated and marked by promoter acetylation. Collectively, the integration of sequencing data showed the potential of a therapy based on epigenetic agents. Through the adoption of a preclinical BPDCN mouse model, established by CAL-1 cell line xenografting, we demonstrated the efficacy of the combination of the epigenetic drugs 5’-azacytidine and decitabine in controlling disease progression in vivo.
Introduction
Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a rare malignancy derived from precursors of plasmacytoid dendritic cells.1–4 It has no clear racial or ethnic predisposition and more often affects elderly males (male/female ratio 3.3:1; mean/median age at diagnosis: 61–67 years). BPDCN patients usually respond to first-line chemotherapy, but despite this they almost invariably relapse and display a dismal prognosis with a median overall survival (OS) ranging from 10 to 19 months.2 No standardized therapeutic approach has so far been established for BPDCN, even if hematopoietic stem cell transplantation has been shown to achieve remission in selected patients.5–6 Therefore, the development of effective treatments still represents an unmet need.7 The pathobiology of BPDCN is poorly understood and the number of reports exploring its molecular features is still limited.8–21 Recent advances in the understanding of the BPDCN molecular landscape have paved the way for novel treatment approaches based on the inhibition of the BCL2 protein,22 the activation of the cholesterol efflux,23 the repression of the Bromodomain-containing protein 4 (BRD4),24 and binding to the interleukin-3 receptor (IL3R).25 All these potential therapeutic options (which are worthy of further evaluation) have mainly emerged from the analysis of the BPDCN transcriptome or from its antigenic repertoire. The genomic landscape of BPDCN has not been well investigated, and only a few studies have explored the mutational events occurring in BPDCN, mainly through targeted sequencing approaches.14,16,19,20
Unfortunately, these have not offered any novel prospects of treatment options.
In this study, we performed whole-exome sequencing (WES) of 14 BPDCN samples and of the BPDCN-derived CAL-1 cell line to look for specific BPDCN genetic vulnerabilities that may support the design of new therapeutic strategies. The WES mutational findings were complemented by copy number variant (CNV) analysis, RNA and pathology tissue-chromatin immunoprecipitation (PAT-ChIP) sequencing results. The integration of data allowed us to identify a successful combinatorial therapy based on epigenetic drugs able to control disease progression in a rapidly progressive BPDCN xenograft model.
Methods
Blastic plasmacytoid dendritic cell neoplasm samples
We collected 14 BPDCN cryopreserved cutaneous biopsies at diagnosis, 9 matched saliva samples and the BPDCN patient-derived cell line, CAL-1. The pathological cases were evaluated as previously described17 and diagnosed by experienced hematopathologists (CA, EB, FF, LC, MP, ES, CT, MT, and SAP) according to World Health Organization diagnostic criteria.2 Informed consent was obtained from each patient in accordance with the Ethical Review Board of the Department of Experimental, Diagnostic, and Specialty Medicine of the University of Bologna, Italy, and the Declaration of Helsinki. DNA was extracted as reported in the Online Supplementary Appendix. The main clinical, immunohistochemical and cytogenetic features of the BPDCN patients are shown in Online Supplementary Tables S1 and S2.
Whole-exome sequencing analysis
We performed paired-end sequencing of matched tumor/normal DNA samples (9 cases), tumor only DNA samples (5 cases), and the CAL-1 cell line (Online Supplementary Table S3) using the TruSeq Exome Kit and Nextera Rapid Capture Exome kit (Illumina). Further details are available in the Online Supplementary Appendix.
Sanger sequencing
We used Sanger sequencing to analyze two candidate nonsense somatic mutations of SUZ12 and ASXL1 occurring in 2 patients, respectively, as described in the Online Supplementary Appendix.
Targeted sequencing
We performed MiSeq targeted sequencing (Illumina) of the 14 BPDCN tumor patients, 7 normal matched saliva samples and the CAL-1 cell line, already analyzed by WES. More bioinformatics details are provided in the Online Supplementary Appendix and Online Supplementary Tables S4 and S5.
RNA sequencing analysis
Five BPDCN cases studied by WES and targeted sequencing had sufficient material for RNA extraction and sequencing; these samples represented the RNA sequencing (RNA-seq) discovery set. We also collected an additional 4 BPDCN cryopreserved cutaneous biopsies, sufficient only for RNA sequencing experiments, used as an RNA-seq extension set. RNA of 4 normal plasmacytoid dendritic cell (pDCs) samples was purchased from AllCells (Alameda, CA, US) and used for comparison. For details, see Online Supplementary Table S6 and the Online Supplementary Appendix.
Pathology tissue-chromatin immunoprecipitation sequencing
The BPDCN_25 and BPDCN_37 patients were provided with one skin biopsy: half was cryopreserved and used for WES, targeted and RNA sequencing analysis, and the other half was fixed in formalin, included in paraffin and used for pathology tissue-chromatin immunoprecipitation (PAT-ChIP) sequencing analysis. PAT-ChIP experiments were performed as in Fanelli et al.26 Further details are available in the Online Supplementary Appendix.
CAL-1 cell line
CAL-1, a BPDCN cell line27 was cultured as reported previously.18 The CAL-1 gene expression profile of a previous study was used17 (http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE62014).
Mouse model and in vivo treatments
Experiments were carried out on 6–8-week old non-obese diabetic severe combined immunodeficient NOD/SCID interleukin-2 receptor g (IL-2Rg)–null (NSG) mice, as previously reported.13 All animal experiments were carried out in accordance with the Italian laws in force (Legislative Decree 26/14 and subsequent amendments) and institutional guidelines. All in vivo studies were ratified by the Italian Ministry of Health. For induction of BPDCN in mice, 5000 CAL-1 cells were injected intravenously (i.v.) through the lateral tail vein in non-irradiated mice. Engrafted mice were then treated with bortezomib, 5’-azacytidine, decitabine and romidepsin, as detailed in the Online Supplementary Appendix.
Results
Whole-exome sequencing reveals the epigenetic program dysregulation as the main theme of the blastic plasmacytoid dendritic cell neoplasm mutational landscape
We collected 14 BPDCN cases with a mean age of 56 years at diagnosis (range 9–89 years), a male-to-female ratio of 10:4, and the classical BPDCN presentation (Online Supplementary Tables S1 and S2).1 The enrolled patients underwent different treatment regimens and 78.5% (11 out of 14) died of the disease 6.3–76 months after the diagnosis or were lost at follow up. Most patients who underwent autologous and/or allogeneic hematopoietic stem cell transplantation experienced a prolonged survival.
We performed WES on 14 BPDCN cases, and on the BPDCN patient-derived CAL-1 cell line and identified 1302 non-synonymous single nucleotide variants (SNVs) represented predominantly by missense SNVs (n=1251), nonsense SNVs (n=47), and frameshift insertions/deletions (n=7). To verify the robustness of the WES analysis, 2 randomly-selected variants occurring in the ASXL1 and SUZ12 genes, respectively, were validated by Sanger sequencing (Online Supplementary Figure S1). To extend the validation to a higher number of samples and mutations, a targeted sequencing approach was adopted: 21 SNVs were interrogated by MiSeq Illumina technology in the same BPDCN cases analyzed by WES and a concordance of 95.2% (20 out of 21 SNVs) was achieved, underlining a high consistency of data (Online Supplementary Table S7). The 1302 non-synonymous SNVs detected by WES affected 1166 genes, all but 7 known to be related with pathological conditions and reported as mutated in the Catalogue of Somatic Mutations in Cancer (COSMIC_v66).
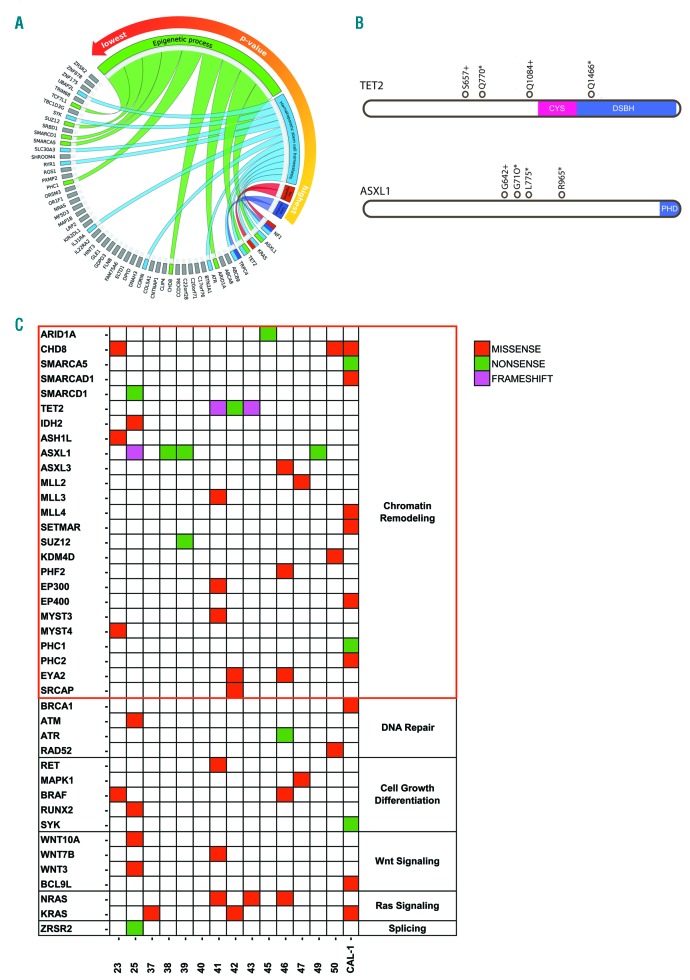
To identify the biological processes that were most altered by the mutational events, we performed a functional enrichment analysis of the 9 genes that were recurrently mutated (≥ 3 samples) and the 45 genes impacted by deleterious (nonsense or frameshift) SNVs. Among the top 10 significantly enriched biological processes, the epigenetic program was the most represented (P=0.0001), followed by hematopoietic stem cell homeostasis, Rac signaling and gamma-aminobutyric acid (GABA) secretion (Figure 1A and Online Supplementary Table S8). The ASXL1 gene proved to be the most frequently mutated (28%, 4 out of 14 samples), followed by TET2 (21%, 3 out of 14 samples); both genes displayed mainly nonsense or frameshift SNVs located within or upstream of the catalytic domain of the proteins, potentially leading to their functional disruption (Figure 1B and Online Supplementary Table S9). We consulted the Gene Ontology database28 to identify among the 1166 BPDCN mutated genes those implicated in the epigenetic regulation. We found 25 mutated epigenetic modifier genes controlling chromatin accessibility (ARID1a, CHD8, SMARCA1), DNA methylation (TET2, IDH2), or histone post-transcriptional modifications [methylation (ASXL1, SUZ12, MLL family), demethylation (KDM4D), acetylation (EP300, EP400), ubiquitination (PHC1, PHC2), dephosphorylation (EYA2), and exchange (SRCAP)]. Of note, 12 out of 14 BPDCN samples (86%) harbored at least one of the 25 epigenetic regulator genes mutated, and specifically 8 out of 14 patients (57.14%) presented a deleterious lesion (nonsense/frameshift SNV) (Online Supplementary Table S10). Many SNVs clustered in the histone methylation pathway, specifically in genes belonging to the Polycomb-repressive complex 2 (ASXL1, ASXL3, SUZ12) and in histone methyltransferases (ASHL1, SETMAR, MLL), possibly compromising the integrity of the methylation program. Besides genetic lesions targeting epigenetic regulators, we also detected mutations potentially affecting molecular programs commonly deregulated in myeloid malignancies, such as RAS signaling29 (hot-spot SNVs on KRAS or NRAS, alternatively), DNA repair/cycle progression30 (SNVs on BRCA1, ATM, ATR, and RAD52), Wnt-signaling31 (SNVs on WNT3, WNT7B, WNT10 and BCL9L), cell growth32 (SNVs on RUNX2, MAPK1), and splicing machinery33 (an SNV on ZRSR2) (Figure 1C).
Figure 1.
The genomic characterization of blastic plasmacytoid dendritic cell neoplasm (BPDCN). (A) Circos plot graphical representation of the functional analysis performed on 54 genes recurrently mutated and/or affected by nonsense and frameshift single nucleotide variants (SNVs) in BPDCN whole-exome sequencing (WES) samples. The four biological processes most significantly enriched are reported in the counterclockwise order from the highest to the lowest P-value: the gamma-aminobutyric acid (GABA) secretion (in violet), the Rac signaling (in red), the hematopoietic stem cell homeostasis (in light blue) and the epigenetic process (in green). The genes are colored according to their belonging to one or more of the biological processes represented. Genes not involved are in gray. (B) Overview of the TET2 and ASXL1 mutations identified. Structure of ASXL1 protein with C-terminal plant homeodomain catalytic region and structure of TET2 protein showing the catalytic core region: the cysteine-rich (Cys) and double-stranded β-helix (DSBH) domains. Empty circles: somatic SNVs. + : frameshift SNV; *nonsense SNV. (C) Heatmap representation of SNVs in BPDCN WES samples and its distribution among selected pathways commonly mutated in myeloid disorders. The SNVs, the affected genes and the related pathways are reported in rows, while, the BPDCN samples are in columns.
Whole-exome sequencing data were also used for cytogenetic CNV analysis, which highlighted extensive losses along the chromosome 9 and the associated deletion of the tumor suppressor CDKN2A gene in 8 out of 14 BPDCN samples (57%) (Online Supplementary Figure S2), as already reported in the literature.12,15,20 In addition, CNV analysis showed that deletions affected six of the nine genes recurrently mutated; deletions were always mutually exclusive with mutations (Online Supplementary Figure S3). However, no significant correlation was found between genetic lesions and the clinical data.
Blastic plasmacytoid dendritic cell neoplasm transcriptome profiling confirms the dysregulation of epigenetic programs
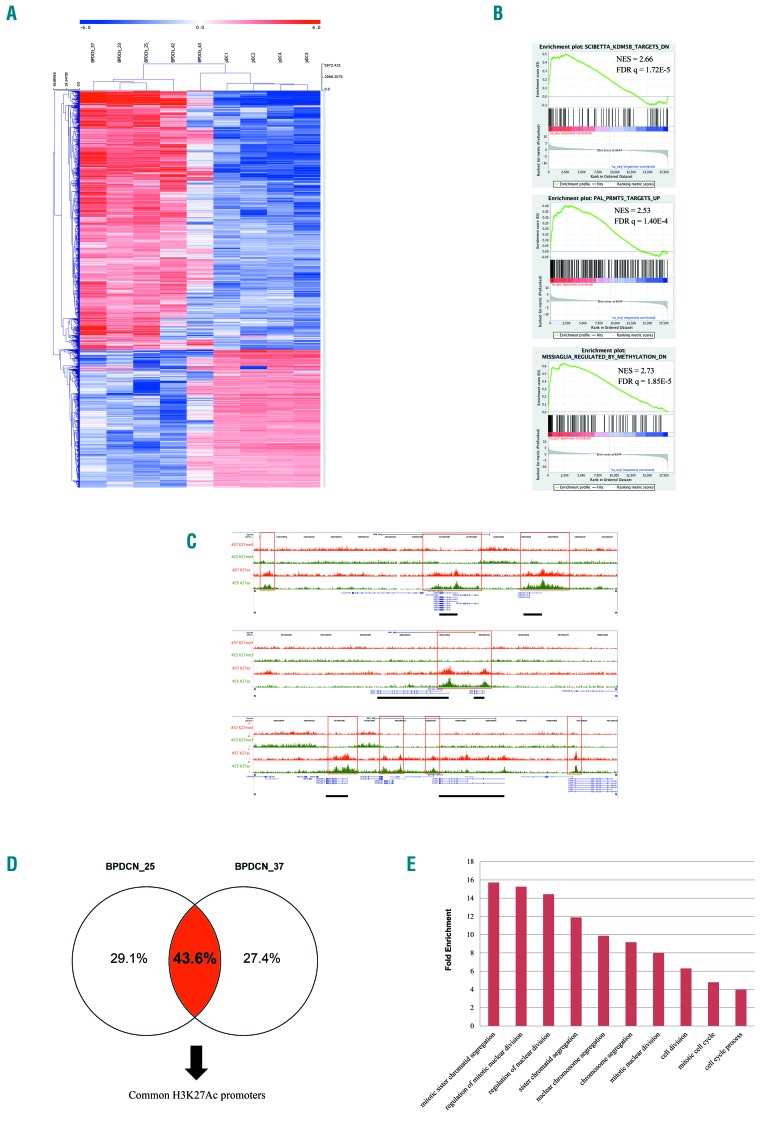
Genetic lesions in key epigenetic modifier genes and in related regulatory networks can induce profound perturbations in the transcriptional homeostasis of the cell. To further substantiate the impact of mutations affecting the chromatin remodeling pathway in BPDCN, we performed RNA sequencing of 5 BPDCNs, considered as the discovery set, already studied by WES and MiSeq targeted sequencing. We compared the patients’ transcriptomes with those of 4 normal plasmacytoid dendritic cell (pDC) samples isolated from the peripheral blood of healthy individuals and used as controls. BPDCN tumor samples and pDCs segregated separately according to their gene expression profiles (Figure 2A). Two thousand and thirty-four genes (2034) were significantly deregulated among patients, and approximately half of them were up-regulated (46%) in the BPDCN setting. Gene set enrichment analysis (GSEA) reported the significant deregulation of two genetic signatures involved in the methylation process, driven by the KDM5B34 histone demethylase and PRMT535 methyltransferase-associated genes, respectively. Of note, GSEA also detected the significant enrichment of a set of genes associated with the response to a DNA demethylating agent,36 namely decitabine (Figure 2B). The GSEA results [normalized enrichment score (NES) ≥2; false discovery rate (FDR) q-value ≤0.0001] were then validated in an extension set of 4 BPDCN samples and in a CAL-1 cell line (Online Supplementary Figures S4 and S5).
Figure 2.
The transcriptome and H3K27 trymethylation/acetylation profiling of blastic plasmacytoid dendritic cell neoplasm (BPDCN). (A) Unsupervised hierarchical clustering performed on 5 BPDCN samples and 4 plasmacytoid dendritic cell (pDCs) samples according to the expression level of the RNA sequencing data. In the heat-map each row represents a gene and each column a sample. The color scale exemplifies the relative expression level of a gene across all samples: (red) represented genes with an expression level above the mean; (blue) the genes with an expression level lower than the mean. Tumors (BPDCNs) and controls (pDCs) cluster in two distinct groups. (B) Gene Set Enrichment Analysis (GSEA) plot illustrating the enrichment of the KDM5B and PRMT5 gene signatures in BPDCN patients reported in literature34–36 as well as the enrichment of a set of genes, described by Missiaglia et al.37 as responsive to hypomethylating treatment, namely decitabine. Normalized enrichment score (NES) ≥ 2; false discovery rate (FDR) q-value false discovery rate ≤0.0001. (C) Visualization of anti-H3K27ac and anti-H3K27me3 normalized pathology tissue-chromatin immunoprecipitation (PAT-ChIP) sequencing profiles in the UCSC Genome Browser showing genomic regions from patient BPDCN_25 and BPDCN_37. (Red boxes) Exemplificative regions displaying a similar level of anti-H3K27ac in both patients. (Black solid rectangles) Genes in correspondence of the anti-H3K27ac peaks. (D) The cases BPDCN_25 and BPDCN_37 share common H3k27ac regions. (E) Histogram representation of the top 10 significant biological processes emerged by Gene Ontology (GO) analysis of 86 up-regulated genes marked by H3K27ac in their promoters. GO categories are shown in x-axis and the fold enrichment values of observed versus expected genes are reported in the y-axis (FDR q-value <0.001).
Genome-wide ChIP-sequencing substantiates epigenetic dysregulation of cell cycle program in blastic plasmacytoid dendritic cell neoplasms
To investigate if the transcriptional deregulation of BPDCNs could be linked to specific epigenetic features, we analyzed the histone acetylation/methylation profiles of 2 selected BPDCN patients (BPDCN_25 and BPDCN_37). The trimethylation at lysine 27 of histone 3 (H3K27me3) is closely associated with inactive gene promoters, while its acetylation (H3K27ac) closely correlates with gene activation, the two epigenetic modifications being mutually exclusive. Given this, we analyzed the genome-wide distribution of trimethylation and acetylation profiles of H3K27 in BPDCN cases. The analysis of PAT-ChIP sequencing data demonstrated that the 2 patients converged on the same pattern of histone acetylation, sharing as much as 43.6% of the acetylated promoters (Figure 2C and D). PAT-ChIP sequencing results were then integrated into the RNA sequencing data leading to the identification of a signature of 86 genes marked by promoter acetylation and significantly over-expressed in the BPDCN RNA sequencing sets. Gene Ontology analysis of the 86 selected genes highlighted the enrichment in biological processes involved in cell cycle progression (FDR q-value <0.001) (Figure 2E and Online Supplementary Table S11).
In vivo blastic plasmacytoid dendritic cell neoplasm modeling demonstrates combined epigenetic therapy as effective in controlling disease progression
The integration of results obtained from WES, RNA sequencing and PAT-ChIP-sequencing experiments clearly pointed to a fundamental role for epigenetic dysregulation in BPDCN and allowed us to hypothesize that this malignancy could display susceptibility to drugs active on the epigenetic regulation. Following the demonstration that the CAL-1 cell line, like primary BPDCN samples, had mutations clustering in chromatin remodeling pathway (Figure 1C) and enrichment in the same epigenetic programs (Online Supplementary Figure S5), we developed an in vivo CAL-1 xenograft BPDCN-like model to explore the effects of treatments targeting the acetylation, methylation, and also the NF-κB pathways, according to previous results.17,18 To this end, we focused on four different FDA-approved compounds: 5’-azacytidine, decitabine, romidepsin and bortezomib.
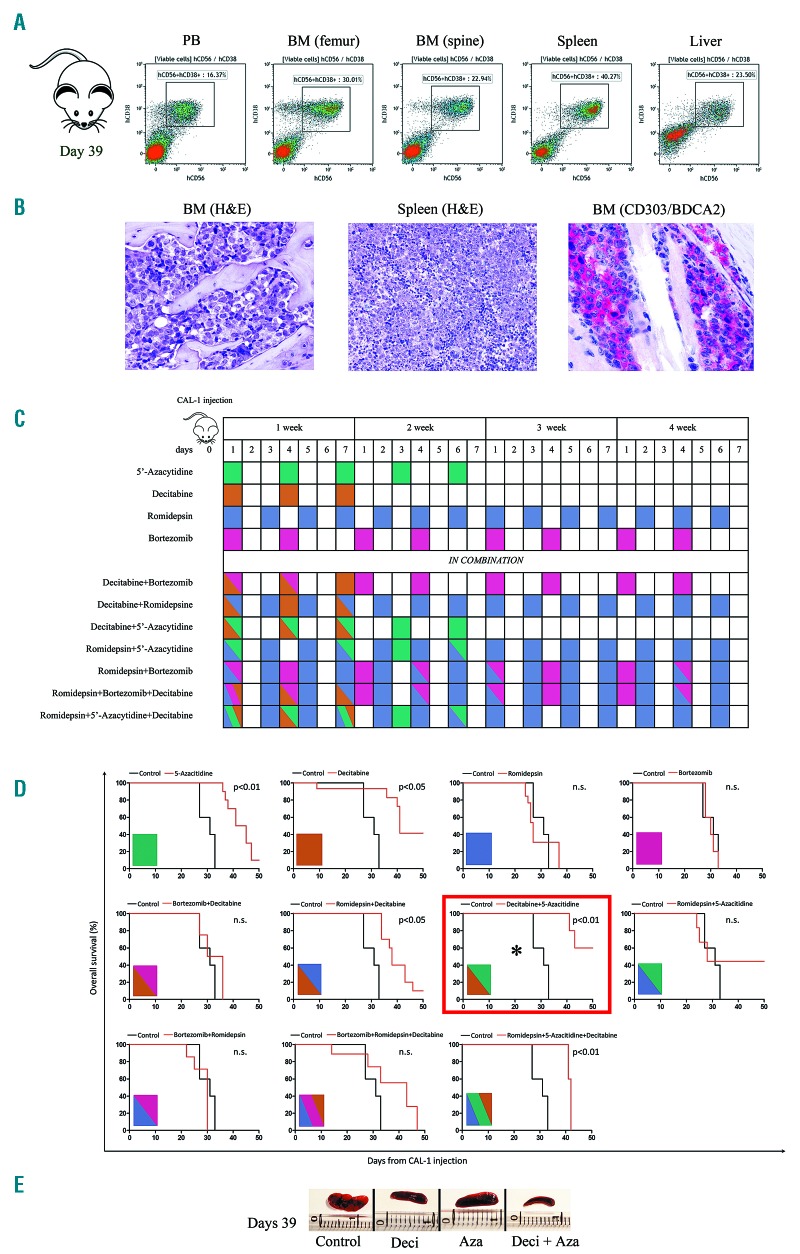
NSG mice intravenously injected with 5×103 CAL-1 cells rapidly developed a systemic BPDCN-like progressive disease, which was defined by the flow cytometry identification of human CD56+CD38+ malignant cells in the peripheral blood, bone marrow, spleen and liver, as evaluated at 39 days after injection (Figure 3A). The pathological infiltration by malignant BPDCN cells in the mouse model was also confirmed at the same time point by the histopathological analysis of the bone marrow and spleen samples, which showed the presence of atypical cells with blastic morphology and expressing the human CD303/BDCA2 pDC marker (Figure 3B). Xenografted mice were divided into 11 treatment groups (n=110 mice) one day after CAL-1 injection and treated with either saline or with the hypomethylating agents 5’-azacitidine or decitabine, the proteasome inhibitor bortezomib, and the histone deacetylase inhibitor romidepsin, used as single agents or in combination, according to the treatment schedule summarized in Figure 3C. The administration of 5’-azacytidine and decitabine used as single agents significantly prolonged OS of the mice when compared with saline (median survival 43.6 days vs. 32 days, P<0.01 for 5’-azacytidine; median survival 44.7 days vs. 32 days, P<0.05 for decitabine) while neither bortezomib nor romidepsin alone showed beneficial effects on disease outcome. When the same agents were associated in combined treatment experiments, three different combinations were seen to significantly prolong mouse survival: i) the association of romidepsin and decitabine (median survival 42.8 days vs. 32 days, P<0.05); ii) the combination of romidepsin, decitabine, and 5’-azacytidine (median survival 41.8 days vs. 32 days, P<0.01); and iii) the association of decitabine and 5’-azacytidine (median survival 52.8 days vs. 32 days, P<0.01), which achieved the best result in terms of survival (Figure 3D). Consistently, 5’-azacytdine and decitabine administered alone reduced the CAL-1-induced splenomegaly as evaluated at day 39 post injection and their combination proved to be even more effective (Figure 3E).
Figure 3.
The efficacy of epigenetic agents in a preclinical blastic plasmacytoid dendritic cell neoplasm (BPDCN) mouse model. (A) Pharmacodynamic assessment of the percentage of human CD56+CD38+ cells in the peripheral blood (PB), bone marrow (BM) of the femur and spine, spleen, and liver of a representative vehicle-treated BPDCN mouse model, 39 days after CAL-1 injection. The cytofluorometric assays shows the tumor dissemination in all the tissues analyzed. (B) Hematoxylin & eosin (H&E) staining of BM and spleen samples collected in a representative vehicle-treated NSG mouse 39 days after CAL-1 injection (H&E; x400; Olympus DP2-SAL). The histological assay shows a marked dissemination of blast elements. The immunohistochemistry detection of the CD303 (BDCA-2) antigen, in the murine BM, indicates the presence of specific BPDCN blasts cells (Immmuno-alkaline phosphatase; Gill’s hematoxylin nuclear counterstaining; x400; Olympus DP2-SAL). These results further confirmed the effective engraftment of CAL-1 cell line. (C) Graphical representation of the treatment schedules observed in a BPDCN mouse model. Each treatment is represented by a single color or by a combination of colors and was administered for four weeks as follows: 5’-azacytidine 5 mg/kg 5 doses at 2-day intervals (green), decitabine 2.5 mg/kg 3 doses at 2-day intervals (light brown), romidepsin 0.5 mg/kg every day (violet), bortezomib 0.5 mg/kg two times weekly (fuchsia). The same doses were also administered in various combinations. (D) Kaplan-Meier curves comparing overall survival of BPDCN mice models respectively treated with vehicle or the above reported treatments. Each treatment is summarized by a box colored as described above. *Indicates that the combination of decitabine and 5’-azacytidine was the most effective in prolonging mice survival. Curves were compared by log-rank test, n=5 mice/treatment arm. (E) Pharmacodynamic assessment of spleen size in 4 representative NSG mice CAL-1 injected after 39 days of treatment with vehicle (mouse Control), Decitabine (mouse Deci), 5’-azacytidine (mouse Aza), and 5’-azacytidine plus decitabine (mouse Deci+Aza) according to the dosing schedule reported above. ns: not significant.
Discussion
Blastic plasmacytoid dendritic cell neoplasm is a rare myeloid malignancy with dismal prognosis and no standard therapy. In the present study, we performed WES on the largest series of BPDCNs that, to the best of our knowledge, has so far been reported in the literature. Thanks to the integration of WES with RNA and PAT-ChIP sequencing, we provide new insights into BPDCN pathobiology by highlighting the dysregulation of the epigenetic program as a hallmark of the disease and suggest possible novel therapeutic interventions.
We found BPDCN patients extensively affected by mutations of genes involved in the epigenetic regulation: 25 epigenetic modifiers were mutated in almost all BPDCN patients (13 out of 14) and the CAL-1 cell line. In more than half of the patients (8 out of 14), the mutations heralded damaging functional alterations (Figure 1C). Some of the mutated genes have already been reported in previous studies (e.g. ASXL1, RAS, ATM, ARID1A, and IDH2), although, at times, at different rates than in our series (see ASXL1 and TET2, which were found to be mutated in 28.6% and 21.4% of our samples vs. 32% and 36% of those of Menezes et al.19). In this respect, it should be remembered that the aim of our study was not only to extensively explore the mutational landscape of BPDCN, but also to possibly translate molecular notions into a preclinical approach. In any case, thanks to the employment of a WES approach, which did not limit our investigation to a priori-selected genes, we recognized additional mutated epigenetic factors that have never been described before but which are potentially relevant in the context of BPDCN, like PHF2 histone demethylase, that enhances the TP53-tumor suppressor activity,37 and the CHD8 Chromodomain helicase DNA-binding protein-8, that promotes the E2F-dependent transcription and cell cycle progression.38 Besides the epigenetic pathway, we also detected mutations affecting programs common to other myeloid malignancies, such as DNA repair process,30 Wnt/β-catenin signaling,31 and the differentiation pathway.32
Importantly, the functional enrichment analysis of WES data showed that among all genes/pathways explored the epigenetic program was the most deregulated (P<0.0001).
To evaluate the impact of the identified epigenetic mutations at gene expression level, we analyzed the transcriptome of samples studied by WES. Among up-regulated genes, GSEA revealed the significant enrichment of two methylation pathways, driven respectively by the KDM5B histone-demethylase34 and by the PRMT5 arginine methyltransferase-5;35 these two epigenetic modifiers are reported to be over-expressed in several cancer types and also represent promising therapeutic targets.39 Blockade of the PRMT5 activity reduces cell survival in chronic myelogenous leukemia40 and inhibition of KDM5B demethylation correlates with cell growth arrest in hepatocellular carcinoma and breast cancers.41,42 We also identified the overexpression of one gene signature36 specifically responsive to the administration of the hypomethylating agent decitabine; a molecular finding bearing important therapeutic implications (FDR q=1.85E-5). To gain a functional insight into the epigenetic landscape of BPDCN samples, we performed PAT-ChIP sequencing of H3K27-acetylation/trymethylation signals of 2 BPDCN patients. The trimethylation of H3K27 marks inactive gene promoters and enhancers, while its acetylation correlates with gene activation.43 PAT-ChIP sequencing data showed that the 2 patients converged on the same epigenetic pattern sharing approximately half of the identified H3K27-acetylated promoters. Interestingly, the common acetylated regions comprised 10 super-enhancers (SE) bound by the Bromodomain-containing protein 4 (BRD4), as described by Ceribelli et al. in a recent work on BPDCN (data not shown).24
The integration of PAT-ChIP and the RNA sequencing data highlighted a set of 86 genes involved in the cell-cycle progression aberrantly over-expressed and marked by H3K27-promoter acetylation. This finding suggests that the cell-cycle deregulation could be driven by H3K27-acetylation signals, a hypothesis meriting future ad hoc studies that could help to clarify the mechanism of proliferation of this largely obscure disease.
The rarity of the disease (with an incidence of 0.000045%) and its extremely aggressive behavior (OS 10–19 months) limits the number of available patients included in biological and/or clinical studies. For these reasons, not surprisingly, BPDCN is still an orphan tumor lacking a standardized and effective therapeutic approach. In the last few years, new molecular studies have opened the way to innovative target therapies (e.g. bortezomib,17,18 venetoclax,22 BET-inhibitors,24 SL-40125) being used in clinical trials. Some of these are showing promising results, although still concerns remain regarding their safety. Of note, all the treatments proposed are mainly the result of investigation into the RNA transcriptome, while the DNA features of BPDCN patients have barely been evaluated.
We therefore decided to tackle this yet incurable disease by designing the first therapeutic strategy modeled on the DNA mutational status of BPDCN patients, analyzed by WES. The WES mutational findings enhanced by the RNA and PAT-ChIP sequencing results clearly evidenced the prominent role of the epigenetic program dysregulation among BPDCN patients and guided our therapeutic approach towards the use of epigenetic agents. In particular, we tested in vivo the efficacy of US Food and Drug Administration-approved epigenetic drugs which could be considered for potential repositioning in clinical trials: two hypomethylating agents such as decitabine and 5’-azacytidine, and the histone deacetylase inhibitor romidepsin. We hypothesized that these drugs could impact on tumor progression because: i) BPDCN patients displayed potential sensitivity to hypomethylating agents, particularly to decitabine, as detected by GSEA analysis; ii) both 5’-azacytidine and decitabine are currently used for the treatment of myelodysplastic syndromes,44,45 which are myeloid neoplasms sharing many epigenetic mutated genes with BPDCN; iii) preclinical studies on several malignancies demonstrated that the action of decitabine is synergized by romidepsin.46 In the light of this, our experimental design focused on epigenetic drugs with a large-scale activity, aiming to explore whether we might induce cell death by perturbation of the malignant epigenetic programme. In addition to the epigenetic drugs, we also verified the efficacy of bortezomib, a proteasome inhibitor, which had previously been shown to significantly induce in vitro and in vivo BPDCN cell death.17,18 Our experiments revealed that the treatment with 5’-azacytidine in combination with decitabine significantly inhibits disease progression and extends survival (P<0.01) in a preclinical mouse model. In the past, two reports experimented the use of 5’-azacytidine in elderly BPDCN patients, though this therapeutic choice was not yet sustained by a molecular rationale.47,48 Here we demonstrate that 5’-azacytidine is more effective in tumor eradication when combined with decitabine. Further studies are ongoing to elucidate the synergistic mechanisms between the two drugs.
In conclusion, we have identified the deregulation of the epigenetic program as a genetic hallmark of BPDCN and suggest a novel therapeutic approach based on the combination of two hypomethylating agents, 5’-azacytidine and decitabine, to be tested in future clinical trials.
Supplementary Material
Footnotes
Check the online version for the most updated information on this article, online supplements, and information on authorship & disclosures: www.haematologica.org/content/104/4/729
Funding
The present work was supported by the AIRC grants IG 15762 and 5×1000 10007 “Genetics-driven targeted management of lymphoid malignancies” and the Grant “Innovative approaches to the diagnosis and pharmacogenetic-based therapies of primary hepatic tumours, peripheral B and T-cell lymphomas and lymphoblastic leukaemias” Strategic Programme 2010–2012 Regione Emilia Romagna - Università (all to SAP).
References
- 1.Chaperot L, Bendriss N, Manches O, et al. Identification of a leukemic counterpart of the plasmacytoid dendritic cells. Blood. 2001;97(10):3210–3217. [DOI] [PubMed] [Google Scholar]
- 2.Swerdlow SH, Campo E, Hazzis NL, Facchetti F, Jones D, Petrella T, et al. Blastic plasmacytoid dendritic cell neoplasm. In: Swerdlow SH, Campo E, Hazzis NL, et al., eds. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. Lyon: IARC Press; 2008:145–147. [Google Scholar]
- 3.Facchetti F, Cigognetti M, Fisogni S, Rossi G, Lonardi S, Vermi W. Neoplasms derived from plasmacytoid dendritic cells. Mod Pathol. 2016;29(2):98–111. [DOI] [PubMed] [Google Scholar]
- 4.Garnache-Ottou F, Feuillard J, Ferrand C, et al. Extended diagnostic criteria for plasmacytoid dendritic cell leukaemia. Br J Haematol. 2009;145(5):624–636. [DOI] [PubMed] [Google Scholar]
- 5.Pagano L, Valentini CG, Pulsoni A, et al. Blastic plasmacytoid dendritic cell neoplasm with leukemic presentation: an Italian multi-center study. Haematologica. 2013;98(2): 239–246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Roos-Weil D, Dietrich S, Boumendil A, et al. Stem cell transplantation can provide durable disease control in blastic plasmacytoid dendritic cell neoplasm: a retrospective study from the European Group for Blood and Marrow Transplantation. Blood. 2013;121(3):440–446. [DOI] [PubMed] [Google Scholar]
- 7.Pemmaraju N. Blastic plasmacytoid dendritic cell neoplasm. Clin Adv Hematol Oncol. 2016;14(4):220–222. [PubMed] [Google Scholar]
- 8.Petrella T, Dalac S, Maynadie M, et al. CD4+ CD56+ cutaneous neoplasms: a distinct hematological entity? Groupe Francais d’Etude des Lymphomes Cutanes (GFELC). Am J Surg Pathol. 1999;23(2):137–146. [DOI] [PubMed] [Google Scholar]
- 9.Leroux D, Mugneret F, Callanan M, et al. CD4(+), CD56(+) DC2 acute leukemia is characterized by recurrent clonal chromosomal changes affecting 6 major targets: a study of 21 cases by the Groupe Francais de Cytogenetique Hematologique. Blood. 2002;99(11):4154–4159. [DOI] [PubMed] [Google Scholar]
- 10.Reichard KK, Burks EJ, Foucar MK, et al. CD4(+) CD56(+) lineage-negative malignancies are rare tumors of plasmacytoid dendritic cells. Am J Surg Pathol. 2005;29(10):1274–1283. [DOI] [PubMed] [Google Scholar]
- 11.Dijkman R, van Doorn R, Szuhai K, Willemze R, Vermeer MH, Tensen CP. Gene-expression profiling and array-based CGH classify CD4+CD56+ hematodermic neoplasm and cutaneous myelomonocytic leukemia as distinct disease entities. Blood. 2007;109(4):1720–1727. [DOI] [PubMed] [Google Scholar]
- 12.Wiesner T, Obenauf AC, Cota C, Fried I, Speicher MR, Cerroni L. Alterations of the cell-cycle inhibitors p27(KIP1) and p16(INK4a) are frequent in blastic plasmacytoid dendritic cell neoplasms. J Invest Dermatol. 2010;130(4):1152–1157. [DOI] [PubMed] [Google Scholar]
- 13.Agliano A, Martin-Padura I, Marighetti P, et al. Therapeutic effect of lenalidomide in a novel xenograft mouse model of human blastic NK cell lymphoma/blastic plasmacytoid dendritic cell neoplasm. Clin Cancer Res. 2011;17(19):6163–6173. [DOI] [PubMed] [Google Scholar]
- 14.Jardin F, Ruminy P, Parmentier F, et al. TET2 and TP53 mutations are frequently observed in blastic plasmacytoid dendritic cell neoplasm. Br J Haematol. 2011;153(3):413–416. [DOI] [PubMed] [Google Scholar]
- 15.Lucioni M, Novara F, Fiandrino G, et al. Twenty-one cases of blastic plasmacytoid dendritic cell neoplasm: focus on biallelic locus 9p21.3 deletion. Blood. 2011;118(17):4591–4594. [DOI] [PubMed] [Google Scholar]
- 16.Alayed K, Patel KP, Konoplev S, et al. TET2 mutations, myelodysplastic features, and a distinct immunoprofile characterize blastic plasmacytoid dendritic cell neoplasm in the bone marrow. Am J Hematol. 2013;88(12):1055–1061. [DOI] [PubMed] [Google Scholar]
- 17.Sapienza MR, Fuligni F, Agostinelli C, et al. Molecular profiling of blastic plasmacytoid dendritic cell neoplasm reveals a unique pattern and suggests selective sensitivity to NF-kB pathway inhibition. Leukemia. 2014;28(8):1606–1616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Philippe L, Ceroi A, Bole-Richard E, et al. Bortezomib as a new therapeutic approach for blastic plasmacytoid dendritic cell neoplasm. Haematologica. 2017;102(11):1861–1868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Menezes J, Acquadro F, Wiseman M, et al. Exome sequencing reveals novel and recurrent mutations with clinical impact in blastic plasmacytoid dendritic cell neoplasm. Leukemia. 2014;28(4):823–829. [DOI] [PubMed] [Google Scholar]
- 20.Stenzinger A, Endris V, Pfarr N, et al. Targeted ultra-deep sequencing reveals recurrent and mutually exclusive mutations of cancer genes in blastic plasmacytoid dendritic cell neoplasm. Oncotarget. 2014;5(15): 6404–6413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Emadali A, Hoghoughi N, Duley S, et al. Haploinsufficiency for NR3C1, the gene encoding the glucocorticoid receptor, in blastic plasmacytoid dendritic cell neoplasms. Blood. 2016;127(24):3040–3053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Montero J, Stephansky J, Cai T, et al. Blastic Plasmacytoid Dendritic Cell Neoplasm Is Dependent on BCL2 and Sensitive to Venetoclax. Cancer Discov. 2017;7(2):156–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ceroi A, Masson D, Roggy A, et al. LXR agonist treatment of blastic plasmacytoid dendritic cell neoplasm restores cholesterol efflux and triggers apoptosis. Blood. 2016;128(23):2694–2707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ceribelli M, Hou ZE, Kelly PN, et al. A Druggable TCF4- and BRD4-Dependent Transcriptional Network Sustains Malignancy in Blastic Plasmacytoid Dendritic Cell Neoplasm. Cancer Cell. 2016;30(5):764–778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Angelot-Delettre F, Roggy A, Frankel AE, et al. In vivo and in vitro sensitivity of blastic plasmacytoid dendritic cell neoplasm to SL-401, an interleukin-3 receptor targeted biologic agent. Haematologica. 2015;100(2): 223–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fanelli M, Amatori S, Barozzi I, et al. Pathology tissue-chromatin immunoprecipitation, coupled with high-throughput sequencing, allows the epigenetic profiling of patient samples. Proc Natl Acad Sci U S A. 2010;107(50):21535–21540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Maeda T, Murata K, Fukushima T, et al. A novel plasmacytoid dendritic cell line, CAL-1, established from a patient with blastic natural killer cell lymphoma. Int J Hematol. 2005;81(2):148–154. [DOI] [PubMed] [Google Scholar]
- 28.Ashburner M, Ball CA, Blake JA, et al. Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet. 2000;25(1):25–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ward AF, Braun BS, Shannon KM. Targeting oncogenic Ras signaling in hematologic malignancies. Blood. 2012; 120(17):3397–3406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Guarini A, Marinelli M, Tavolaro S, et al. ATM gene alterations in chronic lymphocytic leukemia patients induce a distinct gene expression profile and predict disease progression. Haematologica. 2012;97(1):47–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Simon M, Grandage VL, Linch DC, Khwaja A. Constitutive activation of the Wnt/beta-catenin signalling pathway in acute myeloid leukaemia. Oncogene. 2005; 24(14):2410–2420. [DOI] [PubMed] [Google Scholar]
- 32.Kuo YH, Zaidi SK, Gornostaeva S, Komori T, Stein GS, Castilla LH. Runx2 induces acute myeloid leukemia in cooperation with Cbfbeta-SMMHC in mice. Blood. 2009;113(14):3323–3332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Togami K, Madan V, Li J, et al. Blastic Plasmacytoid Dendritic Cell Neoplasm (BPDCN) Harbors Frequent Splicesosome Mutations That Cause Aberrant RNA Splicing Affecting Genes Critical in pDC Differentiation and Function. Blood. 2016; 128(22):738.28092897 [Google Scholar]
- 34.Scibetta AG, Santangelo S, Coleman J, et al. Functional analysis of the transcription repressor PLU-1/JARID1B. Mol Cell Biol. 2007;27(20):7220–7235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pal S, Vishwanath SN, Erdjument-Bromage H, Tempst P, Sif S. Human SWI/SNF-associated PRMT5 methylates histone H3 arginine 8 and negatively regulates expression of ST7 and NM23 tumor suppressor genes. Mol Cell Biol. 2004;24(21):9630–9645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Missiaglia E, Donadelli M, Palmieri M, Crnogorac-Jurcevic T, Scarpa A, Lemoine NR. Growth delay of human pancreatic cancer cells by methylase inhibitor 5-aza-2′-deoxycytidine treatment is associated with activation of the interferon signalling pathway. Oncogene. 2005;24(1):199–211. [DOI] [PubMed] [Google Scholar]
- 37.Lee KH, Park JW, Sung HS, et al. PHF2 histone demethylase acts as a tumor suppressor in association with p53 in cancer. Oncogene. 2015;34(22):2897–2909. [DOI] [PubMed] [Google Scholar]
- 38.Subtil-Rodriguez A, Vazquez-Chavez E, Ceballos-Chavez M, et al. The chromatin remodeller CHD8 is required for E2F-dependent transcription activation of S-phase genes. Nucleic Acids Res. 2014;42(4):2185–2196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rotili D, Mai A. Targeting Histone Demethylases: A New Avenue for the Fight against Cancer. Genes Cancer. 2011;2(6): 663–679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jin Y, Zhou J, Xu F, et al. Targeting methyltransferase PRMT5 eliminates leukemia stem cells in chronic myelogenous leukemia. J Clin Invest. 2016;126(10):3961–3980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yamane K, Tateishi K, Klose RJ, et al. PLU-1 is an H3K4 demethylase involved in transcriptional repression and breast cancer cell proliferation. Mol Cell. 2007;25(6):801–812. [DOI] [PubMed] [Google Scholar]
- 42.Tang B, Qi G, Tang F, et al. JARID1B promotes metastasis and epithelial-mesenchymal transition via PTEN/AKT signaling in hepatocellular carcinoma cells. Oncotarget. 2015;6(14):12723–12739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang T, Cooper S, Brockdorff N. The interplay of histone modifications - writers that read. EMBO Rep. 2015;16(11):1467–1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Quintas-Cardama A, Santos FP, Garcia-Manero G. Therapy with azanucleosides for myelodysplastic syndromes. Nat Rev Clin Oncol. 2010;7(8):433–444. [DOI] [PubMed] [Google Scholar]
- 45.Jabbour E, Short NJ, Montalban-Bravo G, et al. Randomized phase 2 study of low-dose decitabine vs low-dose azacitidine in lower-risk MDS and MDS/MPN. Blood. 2017;130(13):1514–1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kalac M, Scotto L, Marchi E, et al. HDAC inhibitors and decitabine are highly synergistic and associated with unique gene-expression and epigenetic profiles in models of DLBCL. Blood. 2011;118(20):5506–5516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Laribi K, Denizon N, Ghnaya H, et al. Blastic plasmacytoid dendritic cell neoplasm: the first report of two cases treated by 5-azacytidine. Eur J Haematol. 2014;93(1):81–85. [DOI] [PubMed] [Google Scholar]
- 48.Khwaja R, Daly A, Wong M, Mahe E, Cerquozzi S, Owen C. Azacitidine in the treatment of blastic plasmacytoid dendritic cell neoplasm: a report of 3 cases. Leuk Lymphoma. 2016;57(11):2720–2722. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.