Abstract
Purpose
We previously reported that a specific treadmill running exercise regimen protects against light-induced retinal degeneration (LIRD) in mice. We hypothesized that this protective effect varies with running intensity. To test this, mice undergoing LIRD were run at different treadmill speeds and retinal function was assessed.
Methods
BALB/c mice were assigned to LIRD groups at varying treadmill speeds—0, 5, 10, or 20 m/min labeled inactive, low, medium, and high, respectively—and compared with naïve mice exposed to standard lighting (50 lux; naïve). Following 2 weeks of exercise, a subset of mice were exposed to toxic light (10,000 lux; LIRD) for 4 hours. After 5 additional days of exercise, retinal function was assessed by ERG. Corticosterone levels in serum and cathepsin B (CTSB) protein levels in muscle, brain, serum, and retina were measured. The retinal gene expression of complement factor 1qa (C1qa) and CTSB were measured.
Results
The low+LIRD and medium+LIRD exercise groups had greater a- and b-wave ERG amplitudes when compared with the inactive+LIRD group (P < 0.02). The high+LIRD mice only differed from the inactive+LIRD mice in their dark-adapted b-waves. Serum corticosterone increased in the high+LIRD mice (P < 0.006). Retinal CTSB protein levels were higher in the low+LIRD versus high+LIRD mice (P < 0.004) but were otherwise unchanged. Exercise of any intensity decreased C1qa gene expression.
Conclusions
Faster running did not additionally protect against LIRD, but it did increase serum corticosterone, suggesting stress-induced limits to exercise benefits. Unexpectedly, exercise did not increase CTSB proteins levels in muscle or serum, suggesting that it may not mediate exercise effects. Our results have implications for the use of low-intensity exercise as a vision loss treatment.
Keywords: exercise, retinal degeneration, cathepsin B, neuroprotection
Exercise has been shown to enhance cognitive performance and protect against a variety of degenerative diseases within the central nervous system (CNS). In rodents, exercise improves performance on memory1 and learning tasks2 and stimulated neurogenesis.3 In humans, exercise improves memory4,5 and learning5 and ameliorates cognitive dysfunction in patients with Alzheimer's6 and Parkinson's diseases.7,8
As part of the CNS, the retina is a target for exercise interventions, and emerging evidence shows that exercise could prevent and treat retinal degenerative diseases. Several studies suggest that increased physical activity is associated with reduced risk of development or progression of age-related macular degeneration, glaucoma, and diabetic retinopathy and that conversely reduced physical activity is associated with poorer outcomes in these disease groups (reviewed in Ref. 9). We find that increased physical activity is associated with greater self-reported visual function in patients with retinitis pigmentosa.10
Exercise preserves structure and function in animal models of several ocular diseases. We recently reported that treadmill exercise protects visual function in a rat model of streptozotocin (STZ)-induced diabetic retinopathy in pigmented Long-Evans rats,11 corroborating reports that treadmill exercise prevents retinal cell death in rats with diabetic retinopathy.12,13 Chrystomou et al.14,15 report that swim exercise protects retinal ganglion cells against cell loss and dysfunction following acute injury from elevated intraocular pressure, a possible model of glaucoma. Finally, we find that voluntary running wheel exercise protects the retina in an inherited model of retinal degeneration, the pigmented Pde6brd10/rd10 (rd10) mouse,16 and that forced treadmill exercise protects against light-induced retinal degeneration (LIRD) in albino mice,17 a commonly used model of retinal degeneration.18
Although numerous studies in humans and animal models are directed at determining whether varying exercise dose or modality produces different effects on CNS function (reviewed in Refs. 19–21), the optimal exercise intensity for benefit to retina is unstudied and unknown. Ocular blood flow increases with biking intensity in humans,22 and rod-mediated signaling decreases with greater exercise intensity, whereas cone-mediated signaling is unaffected.23 A variety of exercise regimens have been used in exercise studies, but the influence of varying exercise intensities on protection in retinal disease has not been systematically tested. In this study, we hypothesized that the protective effect of treadmill running exercise on the retina that we previously reported17 would vary with intensity, lessening at lower running rates and increasing with faster running rates.
Mechanisms underlying exercise-mediated retinal protection continue to be explored. Myokines, small proteins that respond within skeletal muscle to exercise and initiate signaling cascades throughout the body,24 could be partially responsible. Recently it was reported that one such myokine, cathepsin B (CTSB), increases in calf muscle, plasma, and the brain following exercise, stimulating neurotrophin production in the hippocampus that correlates with cognitive function.25 This suggests that exercise causes a systemic change in CTSB, which could travel to the brain and lead to neural benefits. Whether this relationship extends to the retina has not been studied.
In both humans and rodents, glucocorticoid levels, especially corticosterone, typically considered a stress hormone, rise during exercise.26–28 Glucocorticoids have been shown to influence neural function. When exposed to stress or exogenous corticosterone, neurotrophin mRNA and protein, including that of brain-derived neurotrophic factor, which we found in part mediates exercise-induced retinal protection,11,16,17 decreases in the rat hippocampus.29–31 Rats exercised at low intensities exhibit hippocampal neurogenesis, but this does not occur in rats exercising at a higher intensity that have a concurrent rise in corticosterone levels.27 It is possible that at lower intensities, exercise elicits a lower, therapeutic rise in glucocorticoids. Glucocorticoids possess anti-inflammatory properties32 and create a dose-dependent inhibitory effect on complement activation.33 Long-term exercise lowers systemic complement component 1qa (C1qa) levels and is associated with greater synaptic plasticity in the brain.34 It may be that low-intensity exercise is retinoprotective via modest increases in circulating glucocorticoid levels and decreased complement activation, but that beneficial effects decline at higher intensities of exercise as a result of noxious levels of stress-related glucocorticoids such as corticosterone. These relationships between exercise intensity, glucocorticoids, complement, and neural function have not been studied in the retina. Although secondary to the central question of whether protection varies with exercise intensity, our experimental design allowed for the correlative monitoring of these potential mediators.
To determine optimal levels of exercise for retinal function protection, we assigned varying forced treadmill exercise intensities to mice undergoing LIRD and measured retinal function using ERG. To determine the potential mechanisms of the beneficial effects of exercise, we examined systemic and retinal tissue for molecular changes, focusing on CTSB, corticosterone, and C1qa. By combining the study of exercise intensity and corresponding retinal functional and molecular responses, we aimed to further our understanding of the potential mechanisms contributing to exercise's protection in degenerative disease.
Methods
Ethical Approval
All procedures conformed to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research and were approved by the Atlanta Veterans Administration Institutional Animal Care and Use Committee.
Animals
BALB/c male mice (8–10 weeks old; Charles River, Wilmington, MA, USA) were housed under a 12:12 light:dark cycle with ad libitum access to standard mouse chow.
Experimental Design
The mice were randomly assigned to one of the following four groups: naïve (n = 16), inactive+LIRD (n = 14), or one of three active groups at varying treadmill running speeds, 5 m/min (low+LIRD; n = 18), 10 m/min (med+LIRD; n = 18), or 20 m/min (high+LIRD; n = 20). Four inactive+LIRD mice were excluded from the study, three because of procedural errors in LIRD treatment and one as a result of an absence of ERG response, predetermined criteria for exclusion. As illustrated in Figure 1, the mice were exercised once daily for 5 days per week for 2 weeks. On the day of LIRD, they were removed from the treadmills and exposed to toxic light within 30 minutes. Following 1 additional week of treadmill running, the ERGs were recorded. After 2 final days of exercise, the mice were staged so that each mouse could be euthanized immediately after the end of 1 hour of treadmill running. The mice were killed via cervical dislocation, and the retinas were dissected within 90 seconds and frozen on dry ice. Calf muscle, serum, and whole brain were collected within 2 minutes and also placed on dry ice. All tissue was stored in a −80° celsius freezer.
Figure 1.
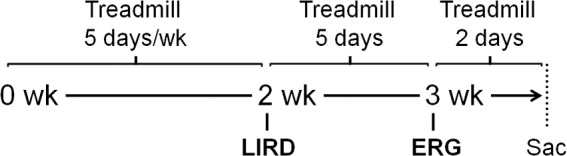
Experimental design. Timeline shown in the middle. Running procedure on top, and experimental procedures on bottom. Wk, week; sac, sacrifice.
Exercise Regimen and Light Exposure
In accordance with previous studies,17 the active mice ran 60 minutes per day between 10 AM and 12 PM on treadmills equipped with electric shock grating (Exer-3/6; Columbus Instruments, Columbus, OH, USA). The grating would inactivate if the mice received 10 shocks in a single session. Before daily exercise, the mice were trained for 5 to 10 minutes at an intensity of 5 to 10 m/min for 2 consecutive days on the treadmill. The mice learned to maintain running within the first few days of running, receiving very few shocks after the first 1 to 2 days. Inactive mice were placed on static treadmills. Following 2 weeks of exercise, the mice were exposed to typical laboratory lighting (50 lux) or toxic light (10,000 lux) for 4 hours using an light emitting diode (LED) light panel (LED500A; Fancierstudio, Hayward, CA, USA) placed atop a standard mouse cage.35 A small subset (n = 10, with representation from all LIRD treated groups) received 40,000 lux for 1 hour and were not statistically different in retinal function from mice with the 10,000 lux treatment and so were included in the study (two-way repeated-measures ANOVA for a- and b-waves, P value > 0.23).
Electroretinography (ERG)
At 1 week after LIRD, retinal function was measured with a commercial ERG system (UTAS 3000; LKC Technologies, Gaithersburg, MD, USA) as previously described.17,36 After overnight dark adaptation, the mice were anesthetized (ketamine [80 mg/kg]/xylazine [16 mg/kg]). The depth of anesthesia was considered appropriate to proceed when whisker movement had ceased and toe pinch reflex was absent. All procedures were performed under dim red light. The corneas were anesthetized (1% tetracaine; Alcon Laboratories, Ft. Worth, TX, USA), and the pupils were dilated (1% tropicamide; Alcon Laboratories). Body temperature was maintained with a heating plate (ATC 1000; World Precision Instruments, Sarasota, FL, USA) for the duration of the session. The ERG consisted of a nine-step series of full-field flash stimuli produced by a Ganzfeld dome under both dark-adapted (−3.4 to 2.5 log cd s/m2) and light-adapted conditions (0.5 to 1.4 log cd s/m2 single flashes and 2.0 log cd s/m2 6.1 Hz flicker with a 30 cd/m2 background light). As flash stimuli increased in brightness, the interstimulus interval progressively increased from 3 to 70 seconds. Custom gold-loop wire electrodes were placed on the center of each eye through a layer of 1% methylcellulose to measure the electrical response of the eye to each flash. Platinum needle reference electrodes (1 cm; Natus Medical Incorporated, Pleasanton, CA, USA) were inserted subcutaneously in each cheek. Following the ERG, anesthesia was reversed with atipamezole (0.5 mg/kg), saline eye drops were applied, and the animals were allowed to recover on a heating pad.
The a-waves, representing photoreceptor function,37 were measured from baseline to the bottom of the first trough. The b-waves, representing ON-bipolar cell function,38 were measured either from the bottom of the a-wave, or the baseline if the a-wave was not present, to the leading curve of the peak of the waveform. ERG responses were measured for both eyes and averaged together.
Enzyme-Linked Immunosorbent Assay (ELISA)
Tissue for ELISA was flash frozen and stored at −80°C. ELISA kits for CTSB (catalog #ab119585; Abcam, Cambridge, UK) and corticosterone (catalog #KA0468; Abnova Corporation, Taipei City, Taiwan) were used according to the manufacturer's instructions. Briefly, the samples were brought to room temperature and homogenized. For the CTSB assay, serum was diluted 1:1250 sample:diluent buffer, brain 1:1000, retina 1:20, and calf muscle 1:100. Sample diluent buffer was used for the negative control. For the corticosterone assay, the serum was diluted 1:10 sample:diluent buffer, and standard ELISA buffer diluent was used for the blank control. The samples were analyzed in triplicate.
Digital Droplet PCR (ddPCR)
The retinal gene expressions of CTSB and C1qa were assayed relative to hypoxanthine-guanine phosphoribosyltransferase (HPRT) expression by ddPCR. RNA isolation was performed using a Qiagen (Germantown, MD, USA) RNeasy Mini Qiacube kit with a Qiagen TissueLyser and Qiacube as per the manufacturer's protocol. Complementary DNA (cDNA) was generated using 500 ng of RNA and a Qiagen QuantiNova Reverse Transcriptase kit as per the manufacturer's protocol. For ddPCR, fluorescent amidite matrix (FAM) hydrolysis probe sets were ordered from Biorad (Hercules, CA, USA) or Integrated DNA Technologies (Coralville, IA, USA) for each target of interest and normalized to a hexachlorofluorescein (HEX) HPRT hydrolysis probe set from Integrated DNA Technologies. ddPCR was performed using 5 ng of cDNA and FAM hydrolysis probe sets for CTSB (dMmuCPE5095240; Biorad), C1qa (Mm.PT.58.7482171; Integrated DNA Technologies), and a HEX hydrolysis probe set for HPRT (Mm.PT.39a22214828; Integrated DNA Technologies) in Biorad ddPCR Supermix for Probes (no deoxyuridine triphosphate [dUTP]). The droplets were prepared using an Automated Droplet Generator model AutoDG (Biorad), plates were sealed with a PX1 plate sealer, PCR was performed using a C1000 Touch thermalcycler (Biorad), with deep well block, and read using a QX200 droplet reader (Biorad). Data were were analyzed using Biorad QuantaLife software and Microsoft (Redmond, WA, USA) Excel 2016. Analyses were performed in triplicate for each tissue sample.
Statistical Analysis
Sample size was determined based on our previously reported data showing that medium-intensity exercise is neuroprotective to retinal function (ERG b-wave amplitudes17). One-way and two-way repeated-measures ANOVAs were performed using SigmaPlot 13.0 (Systat Software, San Jose, CA, USA) with Holm-Sidak post hoc comparisons. For molecular data, significance was set at P < 0.05. For ERG data with multiple comparisons, significance was adjusted using the rough false discovery rate method39 (calculated as P×[#tests+1]/(2×[#tests]) to P < 0.026. The statistical differences for ERG are indicated by *P < 0.026, **P < 0.005, and ***P < 0.001. All data are reported as mean ± 95% confidence intervals, and error bars in all figures represent 95% confidence intervals.
Results
Low- to Medium-Intensity Exercise Protects Retinal Function From Light-Induced Retinal Degeneration
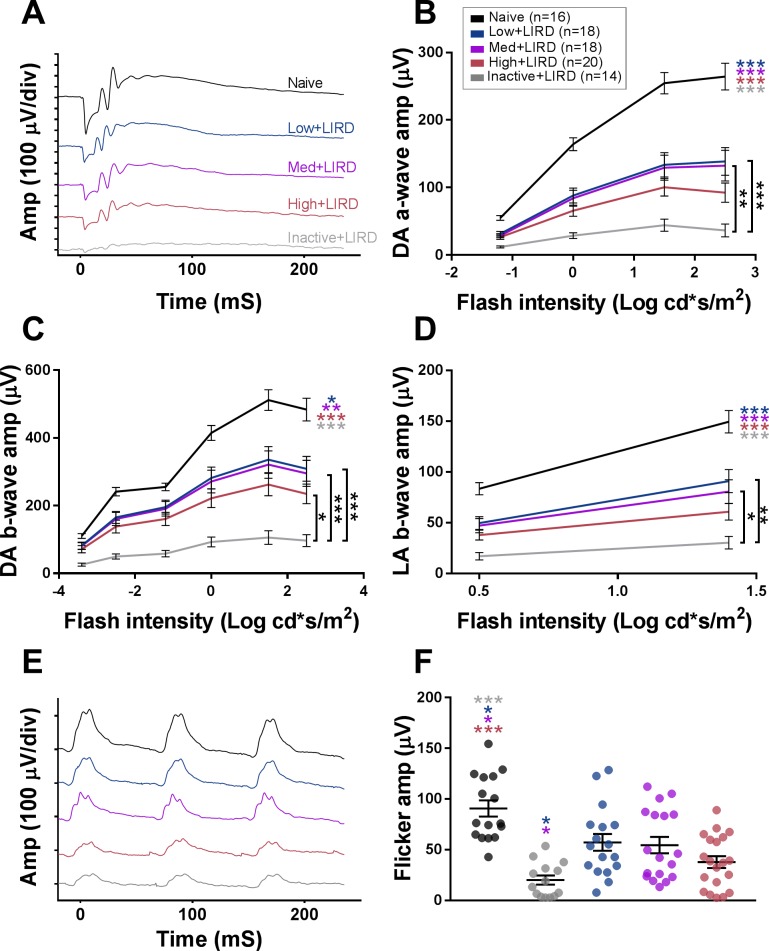
Representative waveforms (Figs. 2A, 2E) show toxic light exposure markedly reduced a- and b-wave amplitudes compared to naïve mice, consistent with previous studies.17 Exercised mice showed functional protection from LIRD. Low and medium exercise intensities gave nearly identical functional benefits, with similar ERG amplitudes on all ERG waves. Dark-adapted ERG a- and b-wave amplitudes were higher in mice in the low+LIRD and med+LIRD groups when compared with the inactive+LIRD group (a-wave, 3.8X inactive+LIRD mean amplitude on the brightest flash; P < 0.001; Fig. 2B; b-wave, 3.9X; P < 0.001; Fig. 2C). Light-adapted ERG b-waves and flicker b-wave amplitudes showed similar differences between the low+LIRD and med+LIRD groups when compared with the inactive+LIRD group (single flash, 3.55X; P < 0.001; Fig. 2D; flicker, 3.18X; P < 0.02). Interestingly, high-intensity exercise did not have the same positive effect as lower intensity exercise. Although ERG amplitudes from the high+LIRD mice trended slightly higher than the inactive+LIRD mice (average difference = 2.4X), they only reached statistical significance for the dark-adapted b-wave amplitude (2.4X, P < 0.007; Fig. 2C), but not for the dark-adapted a-wave, light-adapted b-wave, or flicker amplitudes.
Figure 2.
Effect of exercise intensity on retinal function. Representative ERG waveforms are shown for maximum dark-adapted (DA) stimuli (A, 2.5 log cd s/m2) and flicker (E, 2.0 log cd s/m2). Quantified ERG amplitudes are depicted for dark-adapted a-wave (B), dark-adapted b-wave (C), light-adapted (LA) b-wave (D), and flicker (F). Data are depicted as mean ± SEM. Circles on (F) represent measurements for individual animals. Colored asterisks refer to the difference between the group they are closest to and the group of the referenced color. Amp, amplitude, div, division. *P < 0.026, **P < 0.005, ***P < 0.001.
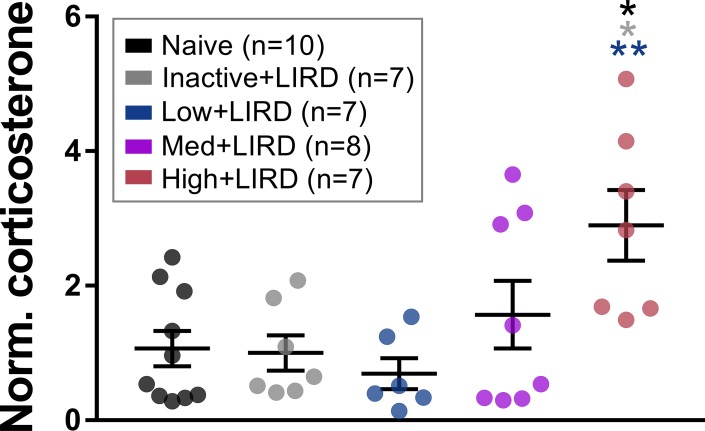
High-Intensity Exercise Elicits a Rise in Corticosterone
To evaluate the contribution of stress to the lack of functional protection in the high+LIRD mice, we evaluated serum corticosterone levels. Mice in the high+LIRD group had significantly higher serum corticosterone than the naïve (2.7X higher, P < 0.01), inactive+LIRD (2.9X higher, P < 0.01), and low+LIRD mice (4.2X higher, P < 0.006; Fig. 3).
Figure 3.
High-intensity exercise increases serum corticosterone. Serum corticosterone was elevated in mice exercised at high intensity, but not low or medium intensity. Data are depicted as mean ± SEM. Circles represent measurements for individual animals. Colored asterisks refer to the difference between the group they are closest to and the group of the referenced color. *P < 0.05, **P < 0.01.
Exercise Does Not Alter Circulating and Muscle Levels of CTSB Protein
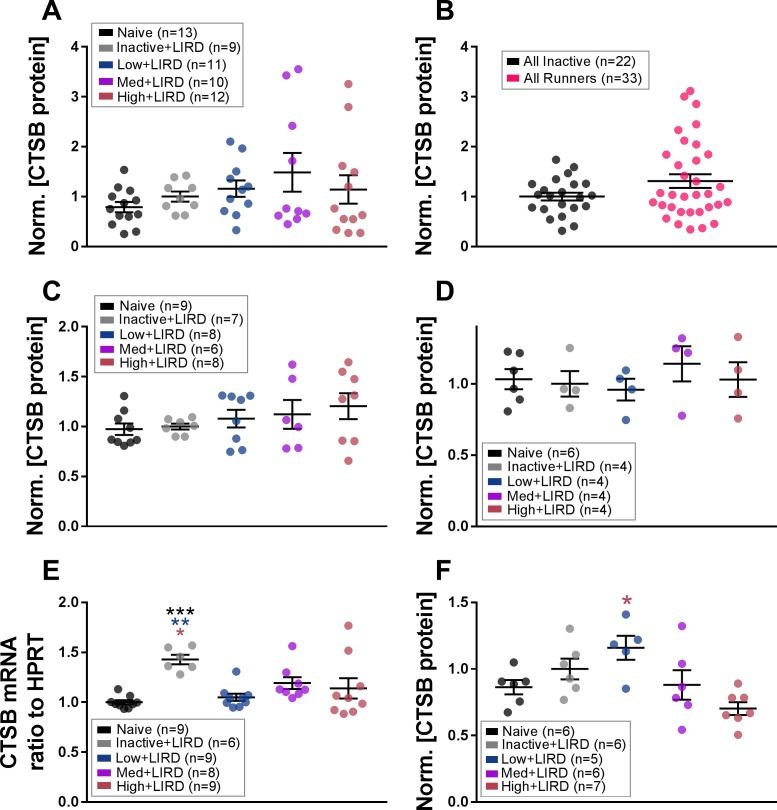
Although low- to medium-intensity exercise resulted in functional protection within the retina, there was no increase in CTSB protein levels within the calf muscle with exercise of any intensity (Fig. 4A), even when all exercised animals were compared against all sedentary animals (Fig. 4B). The CTSB protein levels were also unaltered in whole brain (Fig. 4C) and serum (Fig. 4D) with exercise.
Figure 4.
CTSB gene expression and protein levels altered in retina after exercise. CTSB protein in calf muscle was analyzed by exercise level (A) and when comparing all inactive (inactive+LIRD and naïve) mice to all exercised mice (B). CTSB protein levels were also examined in the brain (C) and serum (D). In the retina, CTSB gene expression (E) and protein levels (F) were measured. Data are depicted as mean ± SEM normalized to inactive+LIRD for A and C–E and to all inactive in B. Circles represent measurements for individual animals. Colored asterisks refer to the difference between the group they are closest to and the group of the referenced color. *P < 0.05, **P < 0.01, ***P < 0.001.
In contrast, CTSB gene expression and protein levels were altered in the retina. CTSB mRNA was elevated in inactive+LIRD mice, with ∼1.3X greater expression than naïve (P < 0.001), low-intensity exercised (P < 0.003), and high-intensity exercised mice (P < 0.03; Fig. 4E). The CTSB protein levels in the retina of exercised mice were not greater than the levels in the inactive+LIRD mice (Fig. 4F). However, the intensity of exercise produced a dose effect in retinal CTSB levels, with low+LIRD mice having significantly higher levels when compared with high+LIRD mice (1.6X higher, P < 0.004; Fig. 4F).
To determine if delayed elevations in CTSB protein occurred following the end of exercise, we exercised a small cohort of male BALB/c mice at 10 m/min for 1 or 3 weeks and killed them at 30 minutes and 1 and 3 hours after the final exercise session. There was no exercise-induced increase in CTSB at any time point in any tissue (Supplementary Fig. S1).
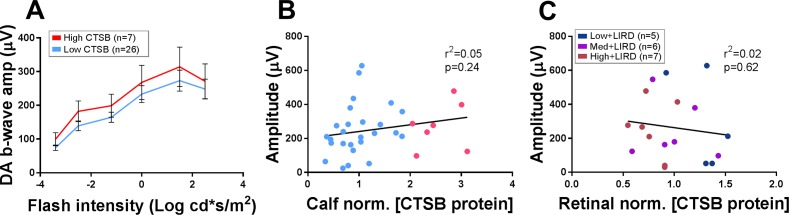
CTSB Protein Levels Not Associated With Exercise-Induced Retinal Protection
To determine if systemic CTSB might underlie retinal function protection, we examined whether mice with the highest levels of calf muscle CTSB showed greater ERG amplitudes. Animals with a calf muscle CTSB level 10% higher than the max inactive+LIRD animal (1.90) were classified as “high CTSB” and compared with those below the threshold, labeled “low CTSB.” These two groups were statistically indistinguishable for dark-adapted b-wave (P = 0.45; Fig. 5A) as well as all other ERG parameters tested (Supplementary Fig. S2). Neither calf muscle nor retinal CTSB levels correlated with the dark-adapted b-wave amplitudes elicited with the brightest dark-adapted stimuli tested (calf muscle CTSB, P = 0.24, Fig. 5B; retinal CTSB P = 0.62, Fig. 5C).
Figure 5.
No correlation in calf muscle or retinal CTSB to functional protection. (A) Active animals across all three exercise intensity groups were divided into high- and low-muscular CTSB levels. Animals with a calf muscle CTSB level 10% higher than the max inactive+LIRD animal were classified as “high CTSB” and compared with those below the threshold “low CTSB” for dark-adapted b-wave amplitude. The brightest flash dark-adapted b-wave amplitudes showed no correlation with calf muscle CTSB (B) and retinal CTSB protein (C). Circles represent measurements for individual animals. Data in A depicted as mean ± SEM. Linear regression line shown in B and C. Amp, amplitude.
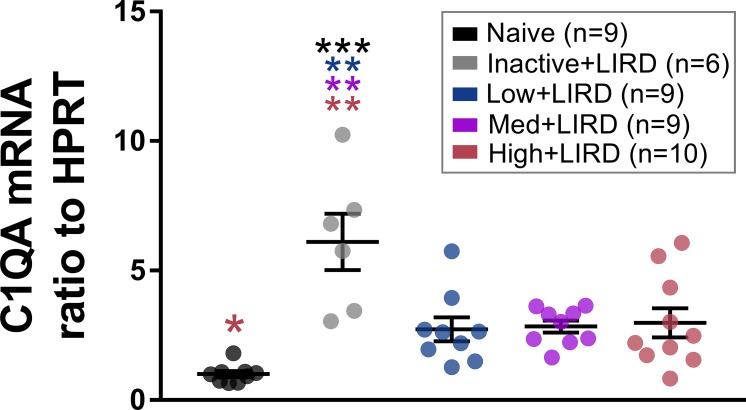
Complement C1qa Gene Expression Lower in Exercised Mice
Because complement factor levels increase during retinal degenerative diseases14,40 and exercise decreases systemic complement induction34 and C1q deposition in retinal ischemia, we hypothesized that exercise would decrease C1qa mRNA levels in the retina. As expected, retinal C1qa expression was elevated in the inactive+LIRD mice when compared with naïve controls (6.1X higher, P < 0.001). All exercise intensities decreased the expression of retinal C1qa when compared with inactive+LIRD mice (low+LIRD, 2.2X smaller, P < 0.001; med+LIRD, 2.2X smaller, P < 0.002; high+LIRD, 2X smaller, P < 0.002; Fig. 6). The high+LIRD expression level was slightly higher than the other exercise groups, reaching statistical difference when compared with naïve mice (P < 0.04).
Figure 6.
Any exercise intensity lowers retinal C1qa gene expression. Retinal C1qa mRNA was elevated in inactive+LIRD animals, but preserved at naïve levels in mice exercised at any of the three treadmill speeds. Data are depicted as mean ± SEM. Circles represent measurements for individual animals. Colored asterisks refer to the difference between the group they are closest to and the group of the referenced color. *P < 0.05, **P < 0.01, ***P < 0.001.
Discussion
Exercise shows promise as an intervention for retinal degenerative disease, but many questions remain about the nature of its protective effects. Consistent with previous studies,12–14,16,17,41,42 we confirmed that exercise protects retinal function in a retinal degeneration model. We advanced these findings by demonstrating that higher intensity exercise may not provide more benefit to retinal function after LIRD, potentially because of the increased stress as reflected by increased circulating corticosterone levels. Instead, we found that lower intensity exercise did not alter corticosterone levels and protected retinal function. Furthermore, although not tested as causative, our results show that the beneficial effects of exercise on retinal function are accompanied by (1) local retinal increases in CTSB protein but not circulating levels and (2) a decrease in retinal complement gene expression.
Across all ERG parameters, low- to medium-intensity exercise gave robust protection from LIRD, whereas high-intensity exercise did not (except for a small increase in dark-adapted b-wave amplitude). One possibility for reduced functional preservation in the high+LIRD mice is increased stress. Corticosterone, the major murine glucocorticoid released in response to stress,43 has been shown to inhibit neurogenesis at high exercise intensities.27 This relationship was confirmed in the current study, as the high+LIRD mice had a significant rise in serum corticosterone, but lacked functional protection.
Although the 10 m/min treadmill exercise regimens have previously been shown to protect against LIRD,17,42 it was not known if lower intensity exercise would be beneficial. One exciting application for this lower intensity but equally effective exercise treatment is for those suffering from age-related retinal degeneration. Although higher intensity exercise such as running would be challenging for a large portion of the older population affected by early retinal disease and other comorbidities, many would be able to engage in low-intensity exercise such as walking.
We were unable to replicate results reported by others that suggest exercise-induced benefit to neuronal function is mediated by CTSB released by skeletal muscle.25 In our study, exercise did not produce a significant increase in CTSB in muscle, brain, or serum. In addition, ERG amplitudes did not correlate with calf muscle CTSB levels; even mice with the highest levels of calf muscle CTSB were not found to have increased retinal function benefits. It is unlikely that CTSB elevations were delayed following exercise, as we did not observe any increase up to 3 hours following exercise. Collectively, these results do not support a role for systemic CTSB in exercise-mediated neural protection.
The dose-dependent relationship seen in the retinal CTSB protein levels with exercise intensity suggests that exercise may have a local effect on retina tissue. However, retinal CTSB protein levels did not correlate with ERG amplitudes (Fig. 4), and retinal CTSB gene expression was only elevated in inactive+LIRD mice (Fig. 3). Our results suggest that if CTSB influences the retina, it does so locally, and not through a systemic mechanism. Additional studies are needed to evaluate CTSB gene expression and protein in exercise and LIRD groups and determine the potential source of CTSB protein in the retina.
Exercise is sometimes considered a preconditioning stimulus.42 We may have detected elevated CTSB expression in inactive+LIRD animals because the tissue was still actively responding to injury when the mice were killed, whereas animals with exercise preconditioning were primed to handle the injury and would quickly return to homeostasis, as suggested for other potential mediators of light-induced stress responses.42 In support of this hypothesis, the inactive+LIRD animals had elevated retinal C1qa expression when compared with naïve mice, suggesting that the complement response in the retinas of sedentary animals was active a week after the injury. In comparison, the exercised mice had levels of retinal C1qa expression close to the levels observed in the naïve mice. This effect of treadmill running may generalize to other exercise modalities and other retinal damage models as it is reminiscent of outcomes reported in an retinal ganglion cell damage model, where the C1q deposition increase induced by acute elevation of intraocular pressure was prevented in mice that had undergone a swim exercise regiment.14 Measurements of the expression levels of these genes across time relative to light-induced injury are needed to more fully understand if a preconditioning effect is present in the retina in response to exercise.
In the current study, lower intensity exercise protected the retina from LIRD, whereas higher intensity exercise did not. Increased stress in mice undergoing higher intensity exercise may have diminished potential protection. No clear role for CTSB in functional protection was found. As true for more than 65 years in the wider field of exercise-induced CNS effects,44–46 further studies are needed to uncover the mechanism linking exercise with retinal neuroprotection. Low-intensity exercise was strengthened as a candidate for treating retinal degenerative disease in the current study, and its greater accessibility could make it an influential intervention.
Supplementary Material
Acknowledgments
Supported by Center for Visual and Neurocognitive Rehabilitation Pilot funds (Center Grant Number C9246C), Research Career Scientist Award RX003134, and SPiRE Award Number I21RX001924 from the United States Department of Veterans Affairs Rehabilitation R&D Service; National Institutes of Health R01EY028859 and P30EY006360; the Abraham J. & Phyllis Katz Foundation; and an unrestricted grant to the Department of Ophthalmology at Emory University from Research to Prevent Blindness, Inc.
Disclosure: L.M. Mees, None; M.M. Coulter, None; M.A. Chrenek, None; C.T. Motz, None; E.G. Landis, None; J.H. Boatright, None; M.T. Pardue, None
References
- 1.Radahmadi M, Alaei H, Sharifi MR, Hosseini N. Effect of forced exercise and exercise withdrawal on memory, serum and hippocampal corticosterone levels in rats. Exp Brain Res. 2015;233:2789–2799. doi: 10.1007/s00221-015-4349-y. [DOI] [PubMed] [Google Scholar]
- 2.Cetinkaya C, Sisman AR, Kiray M, et al. Positive effects of aerobic exercise on learning and memory functioning, which correlate with hippocampal IGF-1 increase in adolescent rats. Neurosci Lett. 2013;549:177–181. doi: 10.1016/j.neulet.2013.06.012. [DOI] [PubMed] [Google Scholar]
- 3.Nokia MS, Lensu S, Ahtiainen JP, et al. Physical exercise increases adult hippocampal neurogenesis in male rats provided it is aerobic and sustained. J Physiol. 2016;594:1855–1873. doi: 10.1113/JP271552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Roig M, Nordbrandt S, Geertsen SS, Nielsen JB. The effects of cardiovascular exercise on human memory: a review with meta-analysis. Neurosci Biobehav Rev. 2013;37:1645–1666. doi: 10.1016/j.neubiorev.2013.06.012. [DOI] [PubMed] [Google Scholar]
- 5.Cassilhas RC, Tufik S, de Mello MT. Physical exercise, neuroplasticity, spatial learning and memory. Cell Mol Life Sci. 2016;73:975–983. doi: 10.1007/s00018-015-2102-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Strohle A, Schmidt DK, Schultz F, et al. Drug and exercise treatment of Alzheimer disease and mild cognitive impairment: a systematic review and meta-analysis of effects on cognition in randomized controlled trials. Am J Geriatr Psychiatry. 2015;23:1234–1249. doi: 10.1016/j.jagp.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 7.Tabak R, Aquije G, Fisher BE. Aerobic exercise to improve executive function in Parkinson disease: a case series. J Neurol Phys Ther. 2013;37:58–64. doi: 10.1097/NPT.0b013e31829219bc. [DOI] [PubMed] [Google Scholar]
- 8.Reynolds GO, Otto MW, Ellis TD, Cronin-Golomb A. The therapeutic potential of exercise to improve mood, cognition, and sleep in Parkinson's disease. Mov Disord. 2016;31:23–38. doi: 10.1002/mds.26484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ong SR, Crowston JG, Loprinzi PD, Ramulu PY. Physical activity, visual impairment, and eye disease. Eye (London, England) 2018;32:1296–1303. doi: 10.1038/s41433-018-0081-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Levinson JD, Joseph E, Ward LA, et al. Physical activity and quality of life in retinitis pigmentosa. J Ophthalmol. 2017;2017:6950642. doi: 10.1155/2017/6950642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Allen RS, Hanif AM, Gogniat MA, et al. TrkB signalling pathway mediates the protective effects of exercise in the diabetic rat retina. Eur J Neurosci. 2018;47:1254–1265. doi: 10.1111/ejn.13909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ji ES, Ko IG, Cho JW, et al. Treadmill exercise inhibits apoptotic neuronal cell death with suppressed vascular endothelial growth factor expression in the retinas of the diabetic rats. J Exerc Rehabil. 2013;9:348–353. doi: 10.12965/jer.130043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim DY, Jung SY, Kim CJ, Sung YH, Kim JD. Treadmill exercise ameliorates apoptotic cell death in the retinas of diabetic rats. Mol Med Rep. 2013;7:1745–1750. doi: 10.3892/mmr.2013.1439. [DOI] [PubMed] [Google Scholar]
- 14.Chrysostomou V, Galic S, van Wijngaarden P, Trounce IA, Steinberg GR, Crowston JG. Exercise reverses age-related vulnerability of the retina to injury by preventing complement-mediated synapse elimination via a BDNF-dependent pathway. Aging Cell. 2016;15:1082–1091. doi: 10.1111/acel.12512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chrysostomou V, Kezic JM, Trounce IA, Crowston JG. Forced exercise protects the aged optic nerve against intraocular pressure injury. Neurobiol Aging. 2014;35:1722–1725. doi: 10.1016/j.neurobiolaging.2014.01.019. [DOI] [PubMed] [Google Scholar]
- 16.Hanif AM, Lawson EC, Prunty M, et al. Neuroprotective effects of voluntary exercise in an inherited retinal degeneration mouse model. Invest Ophthalmol Vis Sci. 2015;56:6839–6846. doi: 10.1167/iovs.15-16792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lawson EC, Han MK, Sellers JT, et al. Aerobic exercise protects retinal function and structure from light-induced retinal degeneration. J Neurosci. 2014;34:2406–2412. doi: 10.1523/JNEUROSCI.2062-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Organisciak DT, Vaughan DK. Retinal light damage: mechanisms and protection. Prog Retin Eye Res. 2010;29:113–134. doi: 10.1016/j.preteyeres.2009.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gomes-Osman J, Cabral DF, Morris TP, et al. Exercise for cognitive brain health in aging: a systematic review for an evaluation of dose. Neurol Clin Pract. 2018;8:257–265. doi: 10.1212/CPJ.0000000000000460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Herting MM, Chu X. Exercise, cognition, and the adolescent brain. Birth Defects Res. 2017;109:1672–1679. doi: 10.1002/bdr2.1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jadczak AD, Makwana N, Luscombe-Marsh N, Visvanathan R, Schultz TJ. Effectiveness of exercise interventions on physical function in community-dwelling frail older people: an umbrella review of systematic reviews. JBI Database System Rev Implement Rep. 2018;16:752–775. doi: 10.11124/JBISRIR-2017-003551. [DOI] [PubMed] [Google Scholar]
- 22.Hayashi N, Ikemura T, Someya N. Effects of dynamic exercise and its intensity on ocular blood flow in humans. Eur J Appl Physiol. 2011;111:2601–2606. doi: 10.1007/s00421-011-1880-9. [DOI] [PubMed] [Google Scholar]
- 23.Zwierko T, Czepita D, Lubinski W. The effect of physical effort on retinal activity in the human eye: rod and cone flicker electroretinogram studies. Graefes Arch Clin Exp Ophthalmol. 2010;248:659–666. doi: 10.1007/s00417-010-1305-1. [DOI] [PubMed] [Google Scholar]
- 24.Wrann CD, White JP, Salogiannnis J, et al. Exercise induces hippocampal BDNF through a PGC-1alpha/FNDC5 pathway. Cell Metab. 2013;18:649–659. doi: 10.1016/j.cmet.2013.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Moon HY, Becke A, Berron D, et al. Running-induced systemic cathepsin b secretion is associated with memory function. Cell Metab. 2016;24:332–340. doi: 10.1016/j.cmet.2016.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Neto RA, de Souza Dos Santos MC, Rangel IF, et al. Decreased serum T3 after an exercise session is independent of glucocorticoid peak. Horm Metab Res. 2013;45:893–899. doi: 10.1055/s-0033-1351279. [DOI] [PubMed] [Google Scholar]
- 27.Shih PC, Yang YR, Wang RY. Effects of exercise intensity on spatial memory performance and hippocampal synaptic plasticity in transient brain ischemic rats. PLoS One. 2013;8:e78163. doi: 10.1371/journal.pone.0078163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brown DA, Johnson MS, Armstrong CJ, et al. Short-term treadmill running in the rat: what kind of stressor is it? J Appl Physiol. 2007;103:1979–1985. doi: 10.1152/japplphysiol.00706.2007. [DOI] [PubMed] [Google Scholar]
- 29.Smith MA, Makino S, Kvetnansky R, Post RM. Stress and glucocorticoids affect the expression of brain-derived neurotrophic factor and neurotrophin-3 mRNAs in the hippocampus. J Neurosci. 1995;15(3 Pt 1):1768–1777. doi: 10.1523/JNEUROSCI.15-03-01768.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jacobsen JP, Mork A. Chronic corticosterone decreases brain-derived neurotrophic factor (BDNF) mRNA and protein in the hippocampus, but not in the frontal cortex, of the rat. Brain Res. 2006;1110:221–225. doi: 10.1016/j.brainres.2006.06.077. [DOI] [PubMed] [Google Scholar]
- 31.Schaaf MJ, de Jong J, de Kloet ER, Vreugdenhil E. Downregulation of BDNF mRNA and protein in the rat hippocampus by corticosterone. Brain Res. 1998;813:112–120. doi: 10.1016/s0006-8993(98)01010-5. [DOI] [PubMed] [Google Scholar]
- 32.Busillo JM, Cidlowski JA. The five Rs of glucocorticoid action during inflammation: ready, reinforce, repress, resolve, and restore. Trends Endocrinol Metab. 2013;24:109–119. doi: 10.1016/j.tem.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ferris LT, Williams JS, Shen CL. The effect of acute exercise on serum brain-derived neurotrophic factor levels and cognitive function. Med Sci Sports Exerc. 2007;39:728–734. doi: 10.1249/mss.0b013e31802f04c7. [DOI] [PubMed] [Google Scholar]
- 34.Soto I, Graham LC, Richter HJ, et al. APOE stabilization by exercise prevents aging neurovascular dysfunction and complement induction. PLoS Biol. 2015;13:e1002279. doi: 10.1371/journal.pbio.1002279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Boatright JH, Moring AG, McElroy C, et al. Tool from ancient pharmacopoeia prevents vision loss. Mol Vis. 2006;12:1706–1714. [PubMed] [Google Scholar]
- 36.Mocko JA, Kim M, Faulkner AE, Cao Y, Ciavatta VT, Pardue MT. Effects of subretinal electrical stimulation in mer-KO mice. Invest Ophthalmol Vis Sci. 2011;52:4223–4230. doi: 10.1167/iovs.10-6750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Penn RD, Hagins WA. Signal transmission along retinal rods and the origin of the electroretinographic a-wave. Nature. 1969;223:201–204. doi: 10.1038/223201a0. [DOI] [PubMed] [Google Scholar]
- 38.Robson JG, Frishman LJ. Dissecting the dark-adapted electroretinogram. Doc Ophthalmol. 1998;95:187–215. doi: 10.1023/a:1001891904176. [DOI] [PubMed] [Google Scholar]
- 39.Aung MH, Park HN, Han MK, et al. Dopamine deficiency contributes to early visual dysfunction in a rodent model of type 1 diabetes. J Neurosci. 2014;34:726–736. doi: 10.1523/JNEUROSCI.3483-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fernandez-Godino R, Garland DL, Pierce EA. A local complement response by RPE causes early-stage macular degeneration. Hum Mol Gen. 2015;24:5555–5569. doi: 10.1093/hmg/ddv287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kim CS, Park S, Chun Y, Song W, Kim HJ, Kim J. Treadmill exercise attenuates retinal oxidative stress in naturally-aged mice: an immunohistochemical study. Int J Mol Sci. 2015;16:21008–21020. doi: 10.3390/ijms160921008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chrenek MA, Sellers JT, Lawson EC, et al. Exercise and cyclic light preconditioning protect against light-induced retinal degeneration and evoke similar gene expression patterns. Adv Exp Med Biol. 2016;854:443–448. doi: 10.1007/978-3-319-17121-0_59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gong S, Miao YL, Jiao GZ, et al. Dynamics and correlation of serum cortisol and corticosterone under different physiological or stressful conditions in mice. PLoS One. 2015;10:e0117503. doi: 10.1371/journal.pone.0117503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vrba R. A source of ammonia and changes of protein structure in the rat brain during physical exertion. Nature. 1955;176:117–118. doi: 10.1038/176117a0. [DOI] [PubMed] [Google Scholar]
- 45.Vrba R. Significance of glutamic acid in metabolic processes in the rat brain during physical exercise. Nature. 1955;176:1258–1261. doi: 10.1038/1761258a0. [DOI] [PubMed] [Google Scholar]
- 46.Vrba R. On the participation of the glutamic acid-glutamine system in metabolic processes in the rat brain during physical exercise. J Neurochem. 1956;1:12–17. doi: 10.1111/j.1471-4159.1956.tb12049.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.