Abstract
Background/purpose
Granulomatosis with polyangiitis (GPA) is a type of primary systemic vasculitis associated with the presence of anti-neutrophil cytoplasmic antibodies (ANCA). GPA mainly affects medium and small vessels and may manifest in different organs, most commonly upper respiratory tract. Oral lesions occur in 6–13% of GPA patients and might be the first symptom observed by the patient. This study presents the evaluation of orofacial manifestations of patients with GPA.
Materials and methods
Prospective study was conducted between June 2014 and November 2017 in Department of Allergy and Immunology at University Hospital in Krakow. Patients diagnosed with GPA, after medical assessment, were examined including precise dental inspection, bacterial and fungal flora of oral cavity evaluation and Cone Beam Computed Tomography (CBCT) imaging.
Results
Nine patients were enrolled in the study. Characteristic for GPA strawberry gingivitis was observed in one patient. Bone destruction and inflammatory lesions in paranasal sinuses was confirmed by CBCT in 55.6% of patients. Fungal infection was revealed in 66.7% of patients.
Conclusion
These findings oblige dentists to consult patient with laryngologist or internal medicine physician to establish further diagnostic approach, because early diagnosis of GPA is crucial for implementing appropriate treatment and preventing chronic organ damage.
Keywords: ANCA, Gingivitis, Granulomatosis with polyangiitis, Oral mucosa, Vasculitis
Introduction
Granulomatosis with polyangiitis (GPA) is one of the three types of primary systemic vasculitis associated with the presence of anti-neutrophil cytoplasmic antibodies (ANCA). It is an autoimmune, rare disease with the incidence in Central Europe of 6–12 new cases/million/year in general population.1 GPA most often affects people between 45 and 60 years old, without significant differences between men and women.2 The incidence of primary systemic vasculitis increases with age. Given the fact that European population is ageing and more accurate diagnostic pathways are getting available, it is not a surprise that GPA morbidity has increased over the past twenty years.3
Pathogenesis of GPA is complex and still unclear with possible genetic,4 environmental5 and infectious factors.6 In some cases adverse drug reactions are mentioned.7 Many abnormalities are found in the immune system of GPA patients. The most characteristic one is the presence of ANCA, which are identified in the GPA as antibodies against proteinase 3 (anti-PR3).8 In their presence there is an uncontrolled activation of the neutrophils followed by recruitment of other inflammatory cells, with particular involvement of T-helper lymphocytes with subsequent inflammatory infiltration of the blood vessels.9,10 Typically small and medium-sized vessels are involved leading to organ damage in kidneys, lungs, mucous membranes or skin.11
Symptoms from the sinuses, ears, oral cavity and throat are present in 70–100% of GPA patients.12 The most common are rhinitis and sinusitis, including the feeling of blockage due to nasal mucosa edema, purulent or bloody chronic nasal discharge, nasal crusting and oral mucosa inflammation with ulcers. They may also be accompanied by symptoms of chronic otitis media with purulent discharge and conductive hearing loss. In milder manifestations, other symptoms may be absent, but in severe cases, the inflammatory process in the facial area can cause skeletal structures destruction with saddle nose and oronasal fistulas formation.13 Patients with GPA are more likely to have nasal cavity colonized by Staphylococcus aureus which is supposed to have an immunomodulatory effect during the development of the disease.14
Oral lesions occur in 6–13% of GPA patients.15,16 The first GPA manifestation can be limited to gingiva inflammation as so called “strawberry gingivitis”. Oral cavity manifestations are localized within the mouth mucosa (ulcers, delayed healing of lesions), gingiva (gingivitis, bleeding, ecchymosis, necrosis), tongue (ulceration, necrotic lesions), alveolar process (osteitis, resorption), palate (ulcers, osteonecrosis, oronasal fistulas), teeth (toothache, tooth loss), nasopharynx (ulcers), and lips (swelling, nodules, exfoliation).8
A rare manifestation of the GPA is parotitis, submucosal or sublingual salivary glands inflammation17,18 with its painful enlargement.19,20
The aim of the study was to present the clinical and radiological characteristics of the orofacial manifestations of GPA as it might be the symptom preceding other organ involvement and the first abnormality observed by the clinician.
Materials and methods
In this prospective study conducted between June 2014 and November 2017 in Department of Allergy and Immunology at University Hospital in Krakow from the group of 30 patients with established diagnosis of GPA, 9 patients accepted the enrollment to the oral examination.
Diagnosis of GPA was based on the American College of Rheumatology criteria for the classification of GPA,21 according to which in each patient at least two of the following findings were present:
-
1)
nasal or oral inflammation (oral ulcers or purulent or bloody nasal discharge)
-
2)
abnormal chest radiograph (presence of nodules, fixed infiltrates, or cavities)
-
3)
urinary sediment (>5 red blood cells per high power field) or red cell casts in urine sediment
-
4)
granulomatous inflammation on biopsy (granulomatous inflammation within the wall of an artery or in the perivascular or extravascular area)
Patients were treated according to current guidelines based on activity and course of the disease.22, 23, 24
Glucocorticosteroids in high doses (3 pulses 250–500 mg followed by 1 mg/kg of body weight of methylprednisolone i.v., then tempered) were used for the remission induction treatment combined with second immunosuppressive drug – cyclophosphamide. Maintenance treatment was based on small doses of glucocorticosteroids and azathioprine or methotrexate.
Patient demographics, clinical symptoms, disease history, localization of GPA lesions in various organs, treatment regimens, and results of selected laboratory tests were recorded.
Patients enrolled to the study had full dental examination. Bacterial and fungal cultures were taken from oral cavity and Cone Beam Computed Tomography (CBCT) scans were performed.
The results were analyzed using the Statistica software version 10.0 (StatSoft, Inc., Tulsa, Oklahoma, USA). The results were expressed as total counts (%) and median with interquartile range (IQR).
The study was conducted in full adherence with good clinical practice and the ethical principles of the Declaration of Helsinki 2013. The written informed consent was obtained from each participant before entering the study. The study protocol was approved by the Bioethics Committee at the Jagiellonian University in Krakow, Poland (KBET/51/B/2014).
Blood samples
Blood samples were collected from the ulnar vein of fasted participants in the morning hours between 8 and 10 a.m. for further analysis. Samples were centrifuged at 3000×g for 10 min and sera were transported on ice to the Laboratory of the 2nd Department of Medicine for further tests. ANCA serum titres were assessed by immunofluorescence method and anty-PR3 levels were examined by ELISA method.
Dental examination
Oral examination including detailed medical history and careful systematic assessment of the oral cavity was performed by two dentists (K.S., D.D.) according to the World Health Organization recommendations.25 Each patient was additionally asked about symptoms of salivary glands dysfunction.
The dental examination comprised of an external and internal examination of hard and soft tissues. It was conducted in a dental office according to the sanitary requirements, under artificial light, using a disposable dental mirror and explorer. Dental examination included the number of total teeth present and the presence of decayed teeth (D), missing teeth due to caries (M) and filled teeth (F). DMF-T index with the total numbers of D, M and F teeth was calculated, and the DMF-T value above zero indicates the present or past caries.
Oral swabs samples
Oral swabs samples were taken from mucous membrane of cheek and tongue of patients in the morning (8:00–10:00) before hygienic procedures and eating. Samples were transferred in ambient temperature to certified microbiological laboratory of the University Hospital in Krakow to obtain bacteriological and mycological cultures.
Radiological examination
CBCT was performed in all patients. An important advantage of Cone Beam CT is high quality of images with the significantly lower radiation dose compared to standard CT. The evaluation of the maxilla and mandible images was done by the radiology specialist from the Radiological Laboratory of the 2nd Department of Medicine.
Results
Nine GPA patients were enrolled to the study. The group consisted of 4 females and 5 males, age 24–68 years (median 57, IQR 50–64 years). The median of duration of GPA was 2 years (IQR 1–6 years).
The most common organ involved was lungs (77.8% of patients). All of the patients had manifestation in at least two organs. Six patients had remission of clinical symptoms, one had disease exacerbation and two patients were newly diagnosed during the time of the study. Patients’ characteristic is presented in Table 1.
Table 1.
Characteristics of patients with granulomatosis with polyangiitis (GPA).
| Number of patients | 9 | |
|---|---|---|
| Age (years): | min – max | 24–68 |
| Median (IQR) | 57 (50–64) | |
| Gender | Female | 4 |
| Male | 5 | |
| Smoking | Yes | 0 |
| No | 9 | |
| Disease duration (years) (IQR) | 2 (1–6) | |
| GPA manifestation (no. of cases) | ||
| Lungs | 7 | |
| Kidney | 5 | |
| Skin | 5 | |
| Joints | 3 | |
| Peripheral nervous system | 1 | |
| GPA activity (no. of cases) | ||
| Remission | 6 | |
| Exacerbation | 1 | |
| Currently diagnosed | 2 | |
| Treatment (no. of cases) | ||
| Glucocorticosteroids (GCs) | 2 | |
| Azathioprine | 2 | |
| Cyclophosphamide + GCs | 3 | |
| Methotrexate + GCs | 1 | |
| Rituximab | 2 | |
| ANCA titres1 (min–max) | 1:10–1:1280 | |
| PR3 (IU/ml)1 Mean (min–max) | 87 (4–156) | |
| anti-PR3 (IU/ml)1 Mean (min–max) | 49 (<0.4–156) | |
Footnote:1 highest values during disease course.
Abbreviations: ANCA – anti-neutrophil cytoplasmic antibodies; anti-PR3 – anti-proteinase 3 antibodies; PR3 – proteinase.
Serum ANCA antibodies in immunofluorescence tests were detected in all patients with GPA and elevated titers ranged from 1: 10 to 1: 1280. Other, typical for GPA findings, were elevated serum proteinase-3 levels with median value of 87 IU/ml, and range from 4 to 156 IU/ml. Median of anti-PR3 antibody concentrations was 49 IU/ml with range from less than 0.4–156 IU/ml (Table 1).
A detailed description of oral and sinus lesions in patients with GPA is presented in Table 2. Two patients were found to have papillary hypertrophy of the tongue, one GPA-specific strawberry gingivitis - confirmed by histopathological examination (Fig. 1a and b). Three patients had no oral lesions. Total incidence of oral lesions in GPA patients is shown in Table 3. Oral mucosa and tongue involvement was reported in 55.6% of the patients.
Table 2.
Results of intraoral and extraoral examinations, microbiological tests and cone beam computed tomography (CBCT) in patients with GPA.
| Patient, age (years) | Oral examination | Salivary glands | Microbiological tests |
Radiological findings | |
|---|---|---|---|---|---|
| Bacteria | Fungi | ||||
| AO, 67 | normal | normal | negative | Candida albicans | normal |
| CD, 59 | tongue filiform papillaes hypertrophy, petechiae of soft palate |
normal | negative | Candida albicans | Early stage of maxillary sinuses wall destruction |
| MM, 24 | tongue filiform papillaes hypertrophy, gingivitis |
normal | negative | negative | Bilateral maxillary sinuses wall destruction |
| AZ, 56 | normal | normal | negative | Candida albicans | normal |
| ZD, 52 | strawberry gingivitis, coated tongue |
normal | negative | Candida albicans | Minor mucosa thickening in the right maxillary sinus |
| KG, 67 | normal | normal | negative | Candida albicans | Mucosa thickening in the right maxillary sinus and bilateral smoothing in maxillary sinuses |
| MP, 59 | paleness of oral mucosa | normal | negative | Candida norvegensis | normal |
| IT, 34 | normal | normal | negative | negative | Bilateral maxillary sinuses wall destruction |
| TZ, 67 | petechiae of oral mucosa | normal | negative | negative | normal |
Fig. 1.
A. Healing stage of strawberry gingivitis in granulomatosis with polyangiitis (GPA) patient. B. Histopathological pictures of healing stage of strawberry gingivitis. Small vessels of mucosal stroma with features of wall hyalinisation, lumen obliteration and minor inflammatory infiltrate.
Table 3.
Frequency of oral lesions and results of tests in patients with GPA.
| Frequency (%) | |
|---|---|
| Oral lesions (total) | 55.6 |
| Oral mucosa paleness | 11.1 |
| Strawberry gingivitis | 11.1 |
| Gingivitis | 11.1 |
| Tongue papilleas hypertrophy | 22.2 |
| Petechiae | 22.2 |
| Coated tongue | 11.1 |
| Sinuses lesions | 55.6 |
| Candida positive cultures | 66.7 |
| Bacteria positive cultures | 0 |
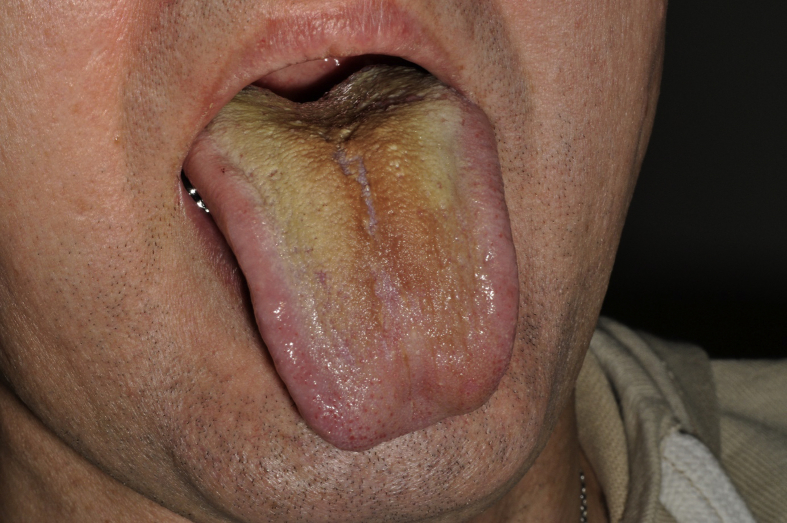
Dental caries was observed in all patients with GPA. The mean DMF-T score was 22. In the detailed examination by palpation of salivary glands no changes were noticed (including no enlargement nor pain). Microbiological evaluation of oral swabs did not reveal pathogenic bacteria. In six patients (66.7%) fungal cultures were isolated and identified. Candida albicans was found in five patients and Candida norvegensis in one (Fig. 2).
Fig. 2.
Coated tongue with Candida albicans infection in granulomatosis with polyangiitis (GPA) patient.
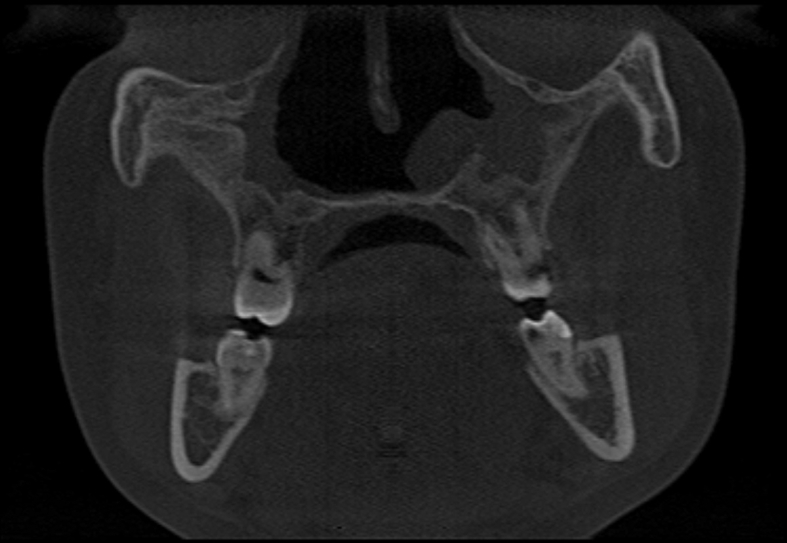
CBCT showed bone destruction and inflammatory lesions in paranasal sinuses in 5 patients (55.6%). Fig. 3, Fig. 4 presents CBCT images of GPA-specific maxillary sinusitis.
Fig. 3.
Cone beam computed tomography (CBCT) image. Sinus lesions in the course of granulomatosis with polyangiitis (GPA). Maxillary sinuses destruction with the remodeling and thickening of their walls. The lumen of the maxillary sinuses is significantly reduced on both sides, partially filled with bone masses and soft tissue masses. Partial bone loss of the nasal cavity lateral walls on both sides. On the left side, the soft tissue masses pass from the maxillary sinus to the lumen of the nasal cavity to form a longitudinal structure. No nasal turbinates on both sides.
Fig. 4.
Cone beam computed tomography (CBCT) image. The sinuses lesions that correspond to sinusitis in the course of granulomatosis with polyangiitis (GPA). Bilateral destruction and heterogeneous remodeling of bone structure with bone and soft tissue masses filling the sinuses.
Discussion
GPA is a rare, chronic inflammatory disease of unknown etiopathogenesis with autoimmune background. It was formerly known as Wegener's granulomatosis, described by the German physician Friedreich Wegener in 1936, as a distinctive form of vasculitis. GPA is characterized by the systemic vasculitis associated with the presence of ANCA in serum of patients.2,8,12,17 The disease affects many organs, particularly the upper respiratory tract including trachea, sinuses and nose, but also lungs, kidneys, nervous system, eyes, ears, heart, skin and oral cavity. The course of GPA is chronic with primarily rapid progression. It can be life-threatening leading to organ failure. Therefore, an early diagnosis and proper treatment is crucial for the patient's prognosis.16
In this study, 9 patients with GPA were identified based on generally accepted criteria.21, 22, 23, 24 This is the first in Poland presentation of oral cavity manifestation of this disease. Oral lesions were found in 55.6% of the patients and most often in form of filiform papillary hypertrophy of the tongue or white coating of the tongue caused by fungal infection. The infection was confirmed in 66.7% of patients, and C. albicans was the most frequently cultured fungus. Frequent fungal infections in patients with GPA may be associated with long, systemic treatment, especially with immunosuppressive agents such as glucocorticosteroids and might have important clinical and therapeutic implications.
In addition, the presence of petechiae on the oral cavity mucosa of patients with GPA and strawberry-like gingivitis were observed. According to the literature, this change is rarely seen, although it is the most characteristic manifestation observed in the oral cavity of patients with GPA. Strawberry gingivitis is usually located in the front of the jaw. Interdental gingiva is enlarged, erythematous with color from red to purple, with reddish or yellow spots, just like outer surface of a ripe strawberry - hence the name.8,26, 27, 28 The patient with strawberry gingivitis as a first clinical manifestation of GPA was recently observed by our group and described by Wawrzycka et al.29 This kind of oral lesion, which was histopathologically verified in a patient with GPA during the healing phase, is shown in Fig. 1.
GPA is a difficult disease to diagnose, especially in the early stages, when the symptoms may be absent or uncharacteristic. Mucocutaneous lesions are various including necrotizing ulcerations, purpura, nodules are not characteristic and can be seen in other systemic diseases.30,31 The oral lesions can be seen as an initial sign of GPA mostly in the form of diffuse gingivitis. Hernandez et al. presented in 2008 a case report of a rare type of strawberry gingivitis which was the first clinical manifestation of GPA then called Wegener's granulomatosis.30 Heera et al. described in 2012 the first case of strawberry gingivitis in India.27 The 54-year-old patient was a smoker, did not care for oral hygiene, and had no other health problems except for generalized gingiva hypertrophy in the jaw and mandible. Panoramic radiograph revealed generalized bone loss, more pronounced in the maxillary anterior region. The ANCA level in the patient's serum was normal, there was a mild leukocytosis, histological examination confirmed the GPA, and glucocorticosteroid therapy was effective. The authors highlight the importance of early diagnosis of this typical oral lesion in GPA.27
GPA is a vasculitis associated with ANCA antibodies, especially ones against proteinase-3 (PR-3).2,8 Although these antibodies are not directly pathogenic, they play significant role in immune cells activation leading to inflammation and granuloma formation. ANCA and anti-PR3 became important marker during diagnostic process.9 All patients enrolled to this study were ANCA positive with identified antibodies against PR3.
Radiological tests are crucial in assessing destructive changes of the cranial bones, including the major paranasal sinuses, where typical GPA lesions are localized.8,13 In this study, changes in facial skeleton were found in over half of the subjects (55.6%). It is highlighted in the literature that facial bone destruction in radiological examinations is very common and characteristic in patients with GPA.13
An interdisciplinary approach to the patient with GPA seems to be important and there is no full assessment of the patient without co-operation of the leading physician with the dentist, laryngologist and other specialists. Dentists should be aware of this disease, because lesions seen during routine radiological examinations performed in practice like extraoral X-ray picture have some limitations. Therefore, orthopantomogram or CBCT can reveal the cranial bones destruction, especially in paranasal sinuses, which are typical manifestations of early GPA. These findings oblige dentists to consult with laryngologist or internal medicine physician to establish further diagnostic path. Early diagnosis of GPA is crucial for implementing appropriate treatment and preventing chronic organ damage.
Conflicts of interest
All authors disclose no conflict of interests in relation to this study.
Acknowledgements
We acknowledge dr. Lukasz Chmura, Department of Pathomorphology, Jagiellonian University, Medical College, for providing us with histopathological pictures.
References
- 1.Reinhold-Keller E., Herlyn K., Wagner-Bastmeyer R., Gross W.L. Stable incidence of primary systemic vasculitides over five years: results from the German vasculitis register. Arthritis Rheum. 2005;53:93–99. doi: 10.1002/art.20928. [DOI] [PubMed] [Google Scholar]
- 2.Comarmond C., Cacoub P. Granulomatosis with polyangiitis (Wegener): clinical aspects and treatment. Autoimmun Rev. 2014;13:1121–1125. doi: 10.1016/j.autrev.2014.08.017. [DOI] [PubMed] [Google Scholar]
- 3.Herlyn K., Buckert F., Gross W.L., Reinhold-Keller E. Doubled prevalence rates of ANCA-associated vasculitides and giant cell arteritis between 1994 and 2006 in Northern Germany. Rheumatology (Oxford) 2014;53:882–889. doi: 10.1093/rheumatology/ket440. [DOI] [PubMed] [Google Scholar]
- 4.Merkel P.A., Xie G., Monach P.A. Identification of functional and expression polymorphisms associated with risk for antineutrophil cytoplasmic autoantibody-associated vasculitis. Arthritis Rheum. 2017;69:1054–1066. doi: 10.1002/art.40034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kerstein A., Schuler S., Cabral-Marques O. Environmental factor and inflammation-driven alteration of the total peripheral T-cell compartment in granulomatosis with polyangiitis. J Autoimmun. 2017;78:79–91. doi: 10.1016/j.jaut.2016.12.004. [DOI] [PubMed] [Google Scholar]
- 6.Pinching A.J., Rees A.J., Pussell B.A., Lockwood C.M., Mitchison R.S., Peters D.K. Relapses in Wegener's granulomatosis: the role of infection. Br Med J. 1980;281:836–838. doi: 10.1136/bmj.281.6244.836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bonaci-Nikolic B., Nikolic M.M., Andrejevic S., Zoric S., Bukilica M. Antineutrophil cytoplasmic antibody (ANCA)-associated autoimmune diseases induced by antithyroid drugs: comparison with idiopathic ANCA vasculitides. Arthritis Res Ther. 2005;7:R1072–R1081. doi: 10.1186/ar1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Almouhawis H.A., Leao J.C., Fedele S., Porter S.R. Wegener's granulomatosis: a review of clinical features and an update in diagnosis and treatment. J Oral Pathol Med. 2013;42:507–516. doi: 10.1111/jop.12030. [DOI] [PubMed] [Google Scholar]
- 9.van Paassen P., Tervaert J.W., Heeringa P. Mechanisms of vasculitis: how pauci-immune is ANCA-associated renal vasculitis? Nephron Exp Nephrol. 2007;105:e10–e16. doi: 10.1159/000096960. [DOI] [PubMed] [Google Scholar]
- 10.Schlesier M., Kaspar T., Gutfleisch J., Wolff-Vorbeck G., Peter H.H. Activated CD4+ and CD8+ T-cell subsets in Wegener's granulomatosis. Rheumatol Int. 1995;14:213–219. doi: 10.1007/BF00262300. [DOI] [PubMed] [Google Scholar]
- 11.Weidner S., Carl M., Riess R., Rupprecht H.D. Histologic analysis of renal leukocyte infiltration in antineutrophil cytoplasmic antibody-associated vasculitis: importance of monocyte and neutrophil infiltration in tissue damage. Arthritis Rheum. 2004;50:3651–3657. doi: 10.1002/art.20607. [DOI] [PubMed] [Google Scholar]
- 12.Trimarchi M., Sinico R.A., Teggi R., Bussi M., Specks U., Meroni P.L. Otorhinolaryngological manifestations in granulomatosis with polyangiitis (Wegener's) Autoimmun Rev. 2013;12:501–505. doi: 10.1016/j.autrev.2012.08.010. [DOI] [PubMed] [Google Scholar]
- 13.D'Anza B., Langford C.A., Sindwani R. Sinonasal imaging findings in granulomatosis with polyangiitis (Wegener granulomatosis): a systematic review. Am J Rhinol Allergy. 2017;31:16–21. doi: 10.2500/ajra.2017.31.4408. [DOI] [PubMed] [Google Scholar]
- 14.Popa E.R., Stegeman C.A., Kallenberg C.G., Tervaert J.W. Staphylococcus aureus and Wegener's granulomatosis. Arthritis Res. 2002;4:77–79. doi: 10.1186/ar392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lilly J., Juhlin T., Lew D., Vincent S., Lilly G. Wegener's granulomatosis presenting as oral lesions: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85:153–157. doi: 10.1016/s1079-2104(98)90418-8. [DOI] [PubMed] [Google Scholar]
- 16.Ponniah I., Shaheen A., Shankar K.A., Kumaran M.G. Wegener's granulomatosis: the current understanding. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;100:265–270. doi: 10.1016/j.tripleo.2005.04.018. [DOI] [PubMed] [Google Scholar]
- 17.Wojciechowska J., Krajewski W., Krajewski P., Krecicki T. Granulomatosis with polyangiitis in otolaryngologist practice: a review of current knowledge. Clin Exp Otorhinolaryngol. 2016;9:8–13. doi: 10.21053/ceo.2016.9.1.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ceylan A., Asal K., Celenk F., Koybasioglu A. Parotid gland involvement as a presenting feature of Wegener's granulomatosis. Singap Med J. 2013;54:e196–e198. doi: 10.11622/smedj.2013183. [DOI] [PubMed] [Google Scholar]
- 19.Berge S., Niederhagen B., von Lindern J.J., Appel T., Reich R.H. Salivary gland involvement as an initial presentation of Wegener's disease. A case report. Int J Oral Maxillofac Surg. 2000;29:450–452. [PubMed] [Google Scholar]
- 20.Specks U., Colby T.V., Olsen K.D., DeRemee R.A. Salivary gland involvement in Wegener's granulomatosis. Arch Otolaryngol Head Neck Surg. 1991;117:218–223. doi: 10.1001/archotol.1991.01870140106018. [DOI] [PubMed] [Google Scholar]
- 21.Leavitt R.Y., Fauci A.S., Bloch D.A. The American College of Rheumatology 1990 criteria for the classification of Wegener's granulomatosis. Arthritis Rheum. 1990;33:1101–1107. doi: 10.1002/art.1780330807. [DOI] [PubMed] [Google Scholar]
- 22.Mukhtyar C., Guillevin L., Cid M.C. EULAR recommendations for the management of primary small and medium vessel vasculitis. Ann Rheum Dis. 2009;68:310–317. doi: 10.1136/ard.2008.088096. [DOI] [PubMed] [Google Scholar]
- 23.Ntatsaki E., Carruthers D., Chakravarty K. BSR and BHPR guideline for the management of adults with ANCA-associated vasculitis. Rheumatology (Oxford) 2014;53:2306–2309. doi: 10.1093/rheumatology/ket445. [DOI] [PubMed] [Google Scholar]
- 24.Yates M., Watts R.A., Bajema I.M. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann Rheum Dis. 2016;75:1583–1594. doi: 10.1136/annrheumdis-2016-209133. [DOI] [PubMed] [Google Scholar]
- 25.World Health Organization - oral health surveys: basic methods - 5th edition. WHO; Geneva: 2013. [Google Scholar]
- 26.Beyer C., Klaus H., Vieth M., Manger B., Schett G., Zwerina J. Strawberry gingivitis. Joint Bone Spine. 2012;79:322. doi: 10.1016/j.jbspin.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 27.Heera R., Choudhary K., Beena V.T., Simon R. Strawberry gingivitis: a diagnostic feature of gingival Wegener's granulomatosis! Dent Res J (Isfahan) 2012;9:S123–S126. [PMC free article] [PubMed] [Google Scholar]
- 28.Ghiasi M. Strawberry gingivitis in granulomatosis with polyangiitis. N Engl J Med. 2017;377:2073. doi: 10.1056/NEJMicm1706987. [DOI] [PubMed] [Google Scholar]
- 29.Wawrzycka K., Szczeklik K., Darczuk D., Lipska W., Szczeklik W., Musial J. Strawberry gingivitis as the first manifestation of granulomatosis with polyangiitis. Pol Arch Med Wewn. 2014;124:551–552. doi: 10.20452/pamw.2468. [DOI] [PubMed] [Google Scholar]
- 30.Hernandez G., Serrano C., Porras L., Lopez-Pintor R., Rubio L., Yanes J. Strawberry-like gingival tumor as the first clinical sign of Wegener's granulomatosis. J Periodontol. 2008;79:1297–1303. doi: 10.1902/jop.2008.070572. [DOI] [PubMed] [Google Scholar]
- 31.Mach T., Szczeklik K., Garlicka M., Owczarek D. Oral ulceration in patient with active Crohn's disease. Przeglad Gastroenterol. 2007;2:210–213. [Google Scholar]