Abstract
Local insulin delivery has been shown to improve osseous healing in diabetic animals. The purpose of this study was to quantify the effects of local intramedullary delivery of saline or Ultralente insulin (UL) on various fracture healing parameters using an in vivo non-diabetic BB Wistar rat model. Quantitation of local insulin levels showed a rapid release of insulin from the fractured femora, demonstrating complete release at 2 days. RT-PCR analysis revealed that the expression of early osteogenic markers (Col1a2, osteopontin) was significantly enhanced with UL treatment when compared with saline controls (p < 0.05). Significant differences in VEGF + cells and vascularity were evident between the treatment and control groups at day 7 (p < 0.05). At day 21, histomorphomet- ric analysis demonstrated a significant increase in percent mineralized tissue in the UL-treated animals compared with controls (p < 0.05), particularly within the subperiosteal region of the fracture callus. Mechanical testing at 4 weeks showed significantly greater mechanical strength for UL-treated animals (p < 0.05), but healing in control animals caught up at 6 weeks post-fracture. These results suggest that the primary osteogenic effect of UL during the early stages of fracture healing (1–3 weeks) is through an increase in osteogenic gene expression, subperiosteal angiogenesis, and mineralized tissue formation.
Keywords: fracture, BB wistar rat, insulin, local delivery, bone regeneration
Insulin is a polypeptide hormone that directly and indirectly modulates bone metabolism and skeletal regeneration.1–3 Functional insulin receptors on rat osteoblasts have been identified that, when activated, stimulate osteoblast proliferation and increase collagen production in vitro.4 Insulin treatment also increases collagen synthesis in bone organ culture5 and amplifies glucose transporter gene expression in osteoblast-like UMR 106 cells.6 Conversely, diminished insulin levels or signaling capacity associated with diabetes mellitus is known to impair osseous healing in animal models and humans.3,7 One of the features of impaired fracture healing observed in the diabetic rat models is decreased synthesis of collagen secondary to impaired cellular proliferation or migration.7,8
Several studies have indicated that local insulin treatment may potentially enhance osseous or fracture healing in non-diabetic animals. In non-diabetic mice, injection of insulin over the calvarium increased osteoid area, osteoblast surface area, and osteoblast number.9 Dedania et al. found that local insulin delivery significantly increased percent mineralized tissue within the endosteal region of the healing defect at 4 and 6 weeks post-osteotomy, compared with saline control animals, in a non-critical size bone defect non-diabetic rat model.2
To test this hypothesis, early and late parameters of fracture healing were measured in non-diabetic BB Wistar rats treated with a single intramedullary injection of Ultralente insulin (UL) immediately before producing a closed, femur fracture.
MATERIALS AND METHODS
Animal Model
One hundred and fifty-eight normal, non-diabetic BB Wistar rats were used, 18 of these were excluded because of anesthetic (3 rats) and surgical (4 rats) complications or because the fracture was not ideally located (11 rats). The rats were obtained from a breeding colony at University of Medicine and Dentistry of New Jersey—New Jersey Medical School that was established with animals from BioBreeding (Toronto, Canada). BB Wistar rats that are resistant to the development of diabetes were chosen as the appropriate breed in the present study to permit comparison with our previous work in spontaneously type-1 diabetic BB Wistar rats.3 All research protocols were approved by the Institutional Animal Care and Use Committee. This study employed parallel group randomized controlled trials to evaluate outcome parameters for UL-treated compared with saline control rats. Rats were randomly assigned to each group, stratified by animal weight prior to surgery and group size was determined by power analyses.
Closed Femur Fracture Procedure and Intramedullary Insulin Delivery
A closed mid-diaphyseal fracture surgery was created in the right femur using a modification of the method described by Bonnarens and Einhorn.3,7 Briefly, after exposing the distal femur, the intramedullary canal was entered by retrograde drilling through the intercondylar notch to the greater trochanter.
In the experimental group, human insulin (Humulin U, Ultralente, Eli Lilly and Company, Indianapolis, IN; 10 units; 0.45 mg) was injected into the intact space of the femoral intramedullary canal prior to insertion of a stainless steel wire to stabilize the impending fracture. Humulin U is a suspension of crystalline recombinant human insulin with zinc that provides for a longer duration of action, nominally 28 h in humans.10 Saline (100 μl) was administered in a similar manner in control animals. We did not observe any noticeable reaction to the human product. Surgical tools and UL insulin were maintained sterile prior to use and 2.5 mg/kg Baytril (Bayer, Mission, KS) was administered following surgery to reduce probability of inflammation-associated infection. In addition, all animals were closely observed within the early post-operative period.
Insulin Quantification
Local and systemic insulin quantification was performed using an enzyme-linked immunosorbent assay (ELISA; EZRMI-13K, Linco Research, Inc., St. Charles, MO) using previously described methods.2 The ELISA kit approached 100% sensitivity to both rat and human insulin. Local insulin quantification was measured at 12, 24, 48, and 96 h after fracture in the femora of animals treated with UL, and normalized to total protein concentration using a bicinchoninic acid assay (Pierce, Rockford, IL). Blood glucose levels were monitored within the first 48 h to evaluate whether locally delivered insulin was released systemically.
Histomorphometry
Rats were euthanized at 4, 7, 21, and 28 days following surgery for histomorphometric analysis of fracture healing. At 4 and 7 days post-fracture, fractured femora were harvested, fixed in 4% formalin, decalcified, embedded in paraffin, serially sectioned longitudinally along the femur from end to end, and stained with Masson’s Trichrome (Accustain™ Trichrome Staining kit, Sigma Diagnostics, St. Louis, MO) as described.10 At 3 and 4 weeks post-fracture, fractured femora were harvested, fixed in 4% formalin, embedded in polymethylmethacrylate, serially sectioned longitudinally along the femur from end to end, and stained with Stevenel’s blue and Van Gieson’s picrofuchsin as described.2,3 The histomorpho-metric analysis measured cartilage area and newly formed mineralized tissue (woven bone) as a percentage of total fracture callus area using an image analysis software (ImagePro Version 5, Media Cybernetics, Inc., Silver Spring, MD). Analysis of all samples at each time point was performed by two independent reviewers in a blinded fashion. All measurements were subsequently averaged. The data provided herein represent the mean values.
Mechanical Testing
Fractured and contralateral, non-fractured femora were harvested at 4 and 6 weeks after fracture and stripped of soft tissue. The peak torque, torsional rigidity, shear modulus, and maximum shear stress were determined through standard equations modeling the femur as a hollow ellipse.9,11
Gene Expression Analysis
Total RNA was isolated from fracture calluses at 4 and 7 days post-fracture and cDNA was obtained by reverse transcription as described previously.12 Target cDNAs were quantified by real-time PCR using SYBR Green I fluorescence as measure of DNA amplification in 25 μl reactions (ABgene, Rochester, NY). The amplifications were performed in an Applied Biosystems 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA) using a two-step protocol that alternated between 95°C for 15 s and the annealing temperature for 1 min. Fluorescence was measured after each of the 40 amplification cycles. Each reaction yielded an amplification plot and melting curve. The melting curve was used to verify the integrity of the quantitative real-time PCR (qPCR) reaction while the amplification plot was used to determine the cycle threshold (Ct) needed to quantify target DNA expression. Relative gene expression was calculated by normalizing values to GAPDH housekeeping gene expression using the software supplied with the instrument. The effects of insulin treatment on callus cartilage and osteoid synthesis was assessed by measuring the expression of Type I (Col1α2), Type II (Col2α1), Type X (Col10α1) collagen, osteo-pontin, and osteocalcin mRNA using RT-qPCR. Target mRNAs, primer sequences, amplification conditions, and annealing temperatures are listed in Supplementary Table I
Immunohistochemistry to Assess Cellular Proliferation and Vascularity
Rats were euthanized 2, 4, and 7 days after fracture and paraffin sections were analyzed for cellular proliferation with an antibody specific for proliferating cell nuclear antigen (PCNA) (EPOS Clone PCIO, DAKO, Denmark), as described previously.13 Positively stained cells were counted manually under 100 × magnification. Data are expressed as number of proliferating cells per unit callus area (cells/mm2). To measure vascularity at 7-days post-fracture, histological sections were examined using immunohistochemical staining with an antibody specific for platelet/endothelial cell adhesion molecule 1 and vascular endothelial growth factor-C (PECAM-1 and VEGF-C, Santa Cruz Biotechnology, Santa Cruz, CA). Positively stained cells were counted manually under 100× magnification. Vascular density was expressed as the number of positively stained cells per area of periosteum/cartilage junction, excluding a vascular cartilage. A rectangular area of 0.8 mm × 4 mm adjacent to the cortex was used to count positively stained cells on the anterior and posterior exterior callus. Specificity of staining was verified by examining slides not exposed to the primary antibody for each immuno-histochemical process (data not shown). Areas of cell analysis were separated into either subperiosteal (intramembranous bone formation) or gap (cartilage formation) callus. Cell counts were normalized to callus area within each region of interest. Analysis of all samples at each timepoint was performed by two independent reviewers in a blinded fashion. All measurements were subsequently averaged. The data provided herein represent the mean values.
Statistical Analysis
A Student’s t-test was performed to identify differences between the experimental and control groups (SigmaStat version 3.0, SPSS Inc., Chicago, IL) at each time point. Significance was established at p ≤ 0.05.
RESULTS
General Health of Animals
The age of the BB Wistar rats at the time of fracture surgery varied between 90 and 137 days for the study. However, animals were age-matched for each experiment and percent weight change was similar between groups. The average blood glucose level 12 h after local insulin treatment was 67 mg/dl, compared to 95 mg/dl for saline control animals.
Histomorphometric Analysis
Histomorphometric analysis revealed that at days 4 and 7 post-fracture, no difference in percent cartilage, percent mineralized tissue or area of the callus existed between insulin treated and control animals (Table 1). By day 21, percent-mineralized tissue in insulin-treated animals was significantly greater than controls (p = 0.021) (Table 1; Fig. 1). However, this effect was not sustained at later time points, as both groups displayed comparable histomorphometric parameters at 4 weeks post-fracture.
Table 1.
Insulin Treatment Enhances Fracture Callus Histomorühometrv
| 7 Days Post-Fracture |
21 Days Post-Fracture |
28 Days Post-Fracture |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Callus Area (mm2) |
% Mineralized Tissue |
% Cartilage | Callus Area (mm2) |
% Mineralized Tissue |
% Cartilage | Callus Area (mm2) |
% Mineralized Tissue |
% Cartilage | |
| Saline control | 20.5 ± 3.9 | 15.0 ± 5.4 | 8.2 ± 4.9 | 19.0 ± 6.6 | 29.0 ± 6.5 | 9.9 ± 4.0 | 16.4 ± 6.1 | 41.0 ± 3.5 | 4.1 ± 3.7 |
| (n = 5) | (n = 5) | (n = 5) | (n = 6) | (n = 6) | (n = 6) | (n = 6) | (n = 6) | (n = 6) | |
| 10 Units local | 21.8 ± 3.5 | 21.6 ± 8.5 | 5.5 ± 1.3 | 16.3 ± 3.3 | 38.7 ± 5.7a | 11.7 ± 2.7 | 18.8 ± 5.3 | 41.7 ± 7.1 | 7.2 ± 2.5 |
| Ultralente insulin | (n = 4) | (n = 4) | (n = 4) | (n = 6) | (n = 6) | (n = 6) | (n = 7) | (n = 7) | (n = 7) |
| p-values | 0.620 | 0.197 | 0.325 | 0.391 | 0.021a | 0.382 | 0.463 | 0.831 | 0.100 |
The data represent mean values (±SD).
Represent values statistically higher than control, p < 0.05 versus control.
Figure 1.
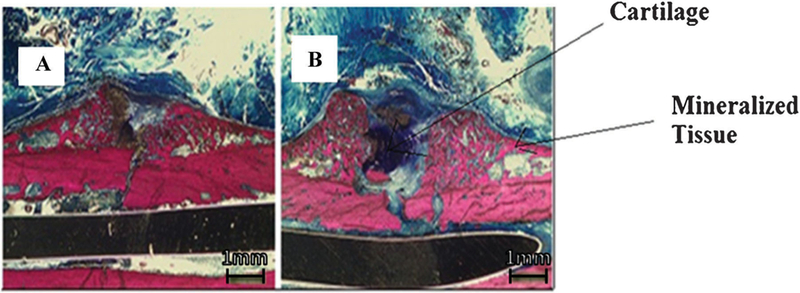
Histological comparison between Ultralente insulin and saline control. (A) 10 units Ultralente insulin at 2.5×; (B) saline control at 2.5× at 3 weeks post-fracture. Scale bar is equivalent to 1 mm as visualized under stereomicroscope.
Mechanical Testing
The effect of local insulin therapy on healing of femur fractures was measured by torsional mechanical testing. At 4 weeks post-fracture, fractured femora of insulin-treated animals displayed improved mechanical properties compared to controls. The maximum torque to failure was greater by 39% (p = 0.008) and maximum shear stress by 58% (p = 0.018) in insulin-treated animals, when compared to saline controls. However, torsional rigidity and shear modulus were not significantly different between groups (Table 2). By 6 weeks post-fracture, no significant differences with respect to mechanical properties were evident (Table 2). Radiographs taken at 4 weeks post-fracture demonstrate a subtle difference between groups, with a greater degree of early callus formation in UL-treated animals (Fig. 2).
Table 2.
Insulin Treatment Enhances Fracture Callus Biomechanics
| Fractured Femur Mechanical Testing Values | ||||||||
|---|---|---|---|---|---|---|---|---|
| 4 Weeks Post-Fracture |
6 Weeks Post-Fracture |
|||||||
| Maximum Torque to Failure (Nmm) |
Maximum Torsional Rigidity (Nmm2/rad) |
Shear Modulus (MPa) |
Maximum Shear Stress (MPa) |
Maximum Torque to Failure (Nmm) |
Maximum Torsional Rigidity (Nmm2/rad) |
Shear Modulus (MPa) |
Maximum Shear Stress (MPa) |
|
| Saline control (n = 5 | 5) 178 ± 38 | 9.3 ± 5.0 × 103 | 235 ± 102 | 19 ± 3 | 498 ± 81 | 4.7 ± 2.0 × 104 | 2.9 ± 1.7 × 103 | 88 ± 31 |
| 10 Units local | 248 ± 17a | 1.9 ± 1.1 × 104 | 685 ± 542 | 30 ± 8a | 537 ± 91 | 5.3 ± 1.6 × 104 | 4.2 ± 2.2 × 103 | 114 ± 39 |
| Ultralente | ||||||||
| insulin (n = 6) | ||||||||
| p-values | 0.008a | 0.105 | 0.103 | 0.018a | 0.472 | 0.566 | 0.298 | 0.275 |
The data represent mean values (±S.D.).
Represent values statistically higher than control, p < 0.05 versus control.
Figure 2.
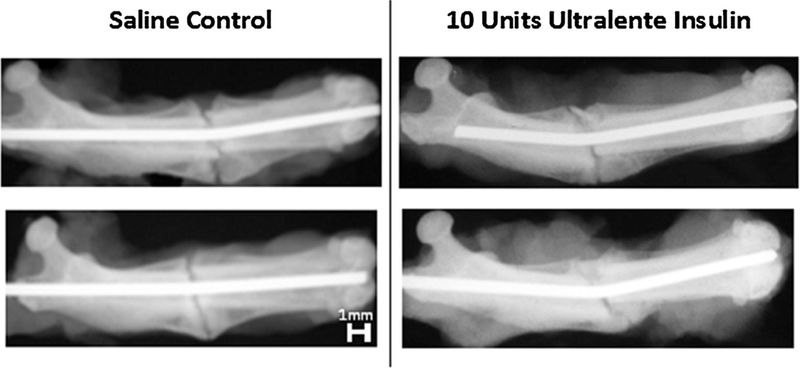
Four-week radiographs (insulin). Radiographs of three saline control, (B) 10 units Ultralente insulin.
Gene Expression Analysis
A significant increase in osteopontin mRNA levels of the insulin-treated (0.36 ± 0.14) relative to the control animals (0.15 ± 0.05) was observed 4 days following fracture (p = 0.015). By day 7 post-fracture, a significant increase in the Col1α2 mRNA level of the insulin-treated specimens (0.051 ± 0.012) relative to the control specimens (0.023 ± 0.004) was evident (p = 0.001). However, no differences in Col2α1 (0.073 ± 0.050 vs. 0.090 ± 0.093) or Col10α1 (0.016 ± 0.003 vs. 0.015 ± 0.003) expression were detected between the control and insulin-treated specimens. These data correlate with the large increase in percent-mineralized tissue observed in fracture calluses of insulin-treated animals at day 7 post-fracture.
PCNA Immunohistochemistry for Proliferating Cells
In animals treated with local UL, no significant differences in cell proliferation existed at 2, 4, or 7 days post-fracture in either the subperiosteal region or the gap region of the fracture callus, normalized to the area of the overall fracture callus (Table 3; Fig. 3).
Table 3.
Insulin Treatment Does Not Effect Early Cell Proliferation
| 2 Days Post-Fracture |
4 Days Post-Fracture |
7 Days Post-Fracture |
||||
|---|---|---|---|---|---|---|
| Positive Proliferating Cells per Unit Periosteal Region Area (Cells/mm2) |
Positive Proliferating Cells per Unit Gap Region Area (Cells/mm2) |
Positive Proliferating Cells per Unit Periosteal Region Area (Cells/mm2) |
Positive Proliferating Cells per Unit Gap Region Area (Cells/mm2) |
Positive Proliferating Cells per Unit Periosteal Region Area (Cells/mm2) |
Positive Proliferating Cells per Unit Gap Region Area (Cells/mm2) |
|
| Control | 507 ± 104 | 449 ± 65 | 925 ± 135 | 820 ±119 | 49 ± 18 | 100 ± 25 |
| (n = 5) | (n = 5) | (n = 5) | (n = 5) | (n = 6) | (n = 6) | |
| 10 Units local | 578 ± 104 | 512 ± 92 | 850 ± 47 | 754 ± 41 | 116 ± 79 | 187 ± 112 |
| Ultralente insulin | (n = 5) | (n = 5) | (n = 5) | (n = 5) | (n = 6) | (n = 6) |
| p-values | 0.312 | 0.246 | 0.274 | 0.275 | 0.070 | 0.093 |
The data represent mean values (±SD). No values were found to be statistically significant.
Figure 3.
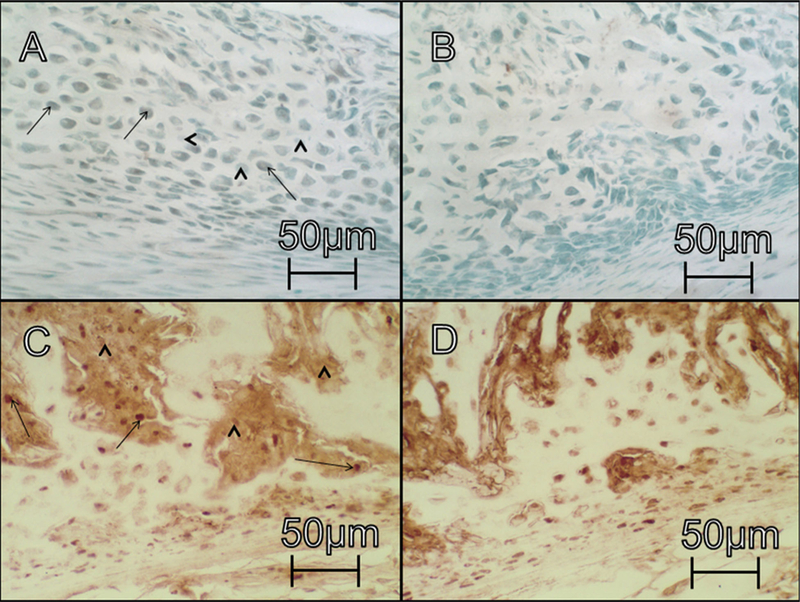
Immunohistochemical comparison between UL treated and saline control rats: panels A and B show (VEGF-C stain at 50×) show cross-sections of UL and saline control groups, respectively, at day 7 post-fracture. Panels C and D (PCNA stain at 50×) show cross-sections of UL and saline control groups, respectively, at day 7 post-fracture. Scale bar is equivalent to 50 mm as visualized under the light microscope. Strait arrows and arrowheads indicate representative cells that were positive and negative, respectively, for each stain.
VEGF/PECAM-1 Immunohistochemistry of Vascular Cells
At day 7, the average blood vessel (BV) density (number of BVs/mm2 callus area) within the subperiosteal region was significantly greater in the insulin-treated group compared with controls (p = 0.008; Table 4). This represents a 75% increase in vascularity for the insulin-treated group after only 7 days. Similarly, the number of VEGF-C+ cells within the subperiosteal region (normalized to the callus area) was significantly greater in insulin-treated animals than controls (p < 0.001; Table 4; Fig. 3). Meanwhile, the number of VEGF+ cells within the gap region was significantly higher for saline controls than insulin-treated animals (p = 0.014).
Table 4.
Insulin Treatment Enhances Early Fracture Callus Vascularity
| 7 Days Post-Fracture |
||||
|---|---|---|---|---|
| Number of Blood Vessels per Unit Area Subperiosteal Region (Vessels/mm2) |
Number of Blood Vessels per per Unit Area Gap Region (Vessels/mm2) |
Number of VEGF + Cells per Unit Area Subperiosteal Region (Cells/mm2) |
Number of VEGF + Cells per Unit Area Gap Region (Cells/mm2) |
|
| Control | 6.0 ± 1.4 (n = 5) | 4.5 ± 2.1 (n = 5) | 2.6 ± 0.9 (n = 5) | 242 ± 34b (n = 5) |
| 10 Units local | 10.5 ± 2.3a (n = 4) | 4.2 ± 2.7 (n = 4) | 7.8 ± 1.2a (n = 3) | 141 ± 50 (n = 3) |
| ultralente insulin | ||||
| p-values | 0.008a | 0.856 | 0.001a | 0.014b |
The data represent mean values (±SD).
Represent values statistically higher than control, p < 0.05 versus control.
Represent values statistically lower than control, p < 0.05 versus control
Insulin Quantification
Differences in local insulin levels were detected between the right, fractured femora and left, intact femora of the rats treated with insulin within the first 12–48 h (significantly higher at 48 h [p = 0.045]), but were largely depleted by 96 h (Fig. 4).
Figure 4.
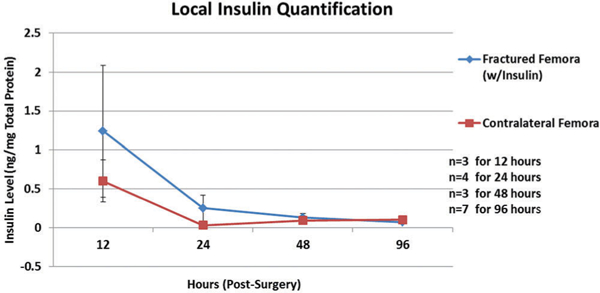
Quantification of local insulin levels Femur bone insulin concentrations (ng/mg total protein) at 12, 24, 48, and 96 h after surgery.
DISCUSSION
Local delivery of insulin was investigated to test its efficacy in accelerating femur fracture healing in a non-diabetic BB Wistar rat model. Prolonged release of insulin at the fracture site was critical in accelerating early osteogenic gene expression, subperiosteal an-giogenesis, and enhancing the mechanical strength of healing bones. The results from the present study suggest that local administration of insulin has the potential to become an option for treating fractures, and potentially as an adjunct for patients with risk factors for non-union (i.e., advanced age, diabetes, etc.).
Histomorphometric analysis of healing fractures following local UL treatment demonstrated an increase in mineralized tissue formation, similar to studies that examined the effects of systemic insulin treatment on non-diabetic fracture healing.14 A study by Stuck noted accelerated trabecular bone formation and the initiation of bone remodeling at 24 days post-fracture for non-diabetic rabbits, following daily injection of insulin (1.5 units) in the lateral ear vein.14 A time course evaluation of both groups revealed that rabbits receiving daily insulin administration had comparable histological sections at 22 days post-fracture to untreated controls at 33 days post-fracture.14 Results from the present study achieved the largest difference in percent mineralized tissue within the fracture callus beween local UL treated animals and saline controls at 21 days post-fracture. Control animals demonstrated comparable healing parameters at 28 days post-fracture. These findings suggest that local delivery of insulin may accelerate the early phases of fracture healing; yet non-diabetic control animals will eventually achieve similar healing outcomes.
Biomechanical parameters of healing fractures following local UL treatment demonstrated significantly enhanced maximum torque to failure and torsional rigidity. These findings support results from a study conducted by Schmidmaier et al. which examined the effects of IGF-1 on non-diabetic rat tibia fracture healing, delivered locally through a poly(D,L-lactide) coated representative samples for each group of fractured femur bone (A) titanium k-wire as the drug carrier.15 Schmidmaier et al. reported that femurs of animals locally treated with IGF-1 demonstrated significantly enhanced percent maximum torque to failure and percent torsional stiffness of the intact contralateral limb compared to saline controls at 4 weeks post-fracture. The IGF-1 administered in Schmidmaier’s study has a similar protein structure to insulin and may target the insulin-signaling pathway. Alternatively, Gregory found no difference in the breaking strength of healing rat femora following daily subcutaneous insulin administration at high doses (10 units/day) by day 7 postfracture.16 These conflicting results may be due to the chronic insulin dosing regimen or the early endpoint at which Gregory evaluated breaking strength.16
While the current study did find significant increases in early osteogenic gene expression, no such changes were evident for chondrogenic gene expression. This finding is possibly due to the rapid release of insulin from the fractured femora. Perhaps sustained release of insulin at the fracture site would have affected chondrogenesis and cell proliferation.
Local UL treatment did not significantly increase cellular proliferation within the fracture callus, contrary to previous studies examining the effects of insulin and IGF-1 on fracture healing.17,18 Others studies suggest that insulin treatment in a non-diabetic population may not necessarily induce mitogenesis.19,20 Fulzele et al. found that insulin treatment did not affect osteoblast cell proliferation in culture media, compared to untreated osteoblasts.20
Early vascular analysis of healing fractures following local UL treatment demonstrated significant increases in number of BVs and VEGF-C positive cells within the subperiosetal region of fracture callus. Previous studies examining the effects of insulin and IGF-1 on fracture healing reported similar findings. Local insulin therapy, while dissipating within 96 h of administration, may thus promote the early osteogenic stages of fracture healing by altering the response of callus cells to growth factors, or by inducing the expression of growth factors, such as IGF-I and VEGFs. Increases in levels of VEGF following trauma have been shown to elevate 5–10 days post-fracture in a rat model,21 while diminishing after 10 days. Local insulin may therefore augment VEGF levels, leading to a mature callus with a high mineralized tissue density.21 Kayal et al. suggest that VEGFs are mediators that play important roles in endochondral bone formation and fracture callus remodeling.22 Similar to the present study, Kayal found that the strepotozotocin (a chemical that is specifically toxic to the beta cells of the pancreas) induced type-I diabetic tibia fracture mouse model, associated with a deficiency of systemic insulin, significantly alters VEGF-C mRNA levels at both 12 and 22 days post-fracture, compared with non-diabetic mice.22 In vitro studies that evaluated the effect of IGF-1 on osteoblast-like cells revealed increases in VEGF mRNA levels and mineralized nodule formation.23,24 Clearly, the concept of local growth factors affecting the paracrine environment after augmentation in an effort to accelerate osseous healing is not novel, yet has numerous potential applications.15,21
Although we did not evaluate the effects of insulin on osteoclastogenesis and associated remodeling during fracture healing, Kayal et al. found that insulin treatment (in a type-I diabetic tibia fracture mouse model) ameliorated the increased cartilage resorption and increased density of osteoclasts within the healing fracture callus at day 16 after fracture, and decreased mineralized tissue formation at days 16 and 22 post-fracture, observed in non-insulin treated diabetic mice.25
Several potential mechanisms exist through which local UL could enhance growth factor expression, vascularity and subperiosteal osteogenesis. Our immunohistochemistry and gene expression findings suggest that local insulin treatment may enhance fracture healing by influencing early cell signaling within the subperiosteal region of the fracture callus which may affect early osteogeneisis/angiogenesis. Other studies26–28 have noted similar findings, wherein changes in angiogenesis/osteogenesis within the subperiosteal region led to strong effects on mineralized tissue formation and strength differences in the entire callus. We speculate differences that were detected exclusively in the subperiosteal region may be related to a cell-signaling target within this tissue, such as pre-osteoblasts. The sequence of cell signaling following local insulin distribution at the fracture site has yet to be investigated. It is possible that insulin affects cross-talk between endothelial cells and osteoblasts. Osteoblasts are known to produce paracrine factors which may be affected by the administration of insulin, thus enhancing VEGF production and endothelial cell proliferation.24,29
Significant increases in osteogenic gene expression following local UL treatment support the theory that the primary mechanism by which insulin exerts an osteogenic effect may be via osteoblast-mediated VEGF production, with a subsequent enhancement of subperiosteal bone formation. However, it is unclear whether insulin administration during fracture healing is exclusively directed by osteoblast or endothelial cell signaling, or the accelerated bone repair is a result of cross-talk between these cells.
A limitation of this study is that it was designed to determine whether local UL insulin affected fracture healing but not the mechanisms through which local UL works to affect healing. Potential mechanisms by which insulin may mediate skeletal remodeling have been explored by other studies.20,30 In addition, the present study did not investigate changes in early signaling following short-term administration of insulin. An in vitro osteoblast cell culture study that examines the effects of UL on early cytokine/growth factor expression may help elucidate the effects of UL on early signaling and may be correlated to fracture healing outcomes seen weeks after insulin delivery. Furthermore, the present study examined angiogenesis at a single time point (week 1) and mineralized tissue formation at three time points (weeks 1, 3 and 4). Further investigation of angiogenesis and osteogenesis at additional time points (i.e., 10, 14, and 17 days) would provide supplementary data to suggest a link between early angiogenesis and increased callus mineralization.
Although results from this study suggest that local UL treatment for fracture healing is a promising potential therapy, processing restrictions, storage issues, and the risk of hypoglycemia following administration requires further investigation. The mechanism of action, appropriate dosing, and optimal route of delivery of insulin in this application need to be characterized. The current study only examined a single dose (10 units) of local UL. A dose-response study for local UL might reveal a more optimal dose to further enhance the osteogenic effects that were reported in the present study.
To the best of our knowledge, the present in vivo study is the first to investigate the effect of local insulin delivery on fracture healing in non-diabetic animals. This study demonstrated that acute, local insulin treatment immediately after fracture promoted healing in non-diabetic rats.
Supplementary Material
ACKNOWLEDGMENTS
Sheldon S. Lin, Loay AI-Zube, J. Patrick O’Connor, Eric Breitbart, and Joseph Benevenia are listed as inventors on U.S. Patent No. 7763582, issued June 27, 2010.
Footnotes
Additional supporting information may be found in the online version of this article.
REFERENCES
- 1.Thrailkill KM, Lumpkin CK Jr, Bunn RC, et al. 2005. Is insulin an anabolic agent in bone? Dissecting the diabetic bone for clues. Am J Physiol Endocrinol Metab 289:E735–E745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dedania J, Borzio R, Paglia D, et al. 2011. Role of local insulin augmentation upon allograft incorporation in a rat femoral defect model. J Orthop Res 29:92–99. [DOI] [PubMed] [Google Scholar]
- 3.Gandhi A, Beam HA, O’Connor JP, et al. 2005. The effects of local insulin delivery on diabetic fracture healing. Bone 37:482–490. [DOI] [PubMed] [Google Scholar]
- 4.Levy JR, Murray E, Manolagas S, et al. 1986. Demonstration of insulin receptors and modulation of alkaline phosphatase activity by insulin in rat osteoblastic cells. Endocrinology 119:1786–1792. [DOI] [PubMed] [Google Scholar]
- 5.Craig RG, Rowe DW, Petersen DN, et al. 1989. Insulin increases the steady state level of alpha-1(I) procollagen mRNA in the osteoblast-rich segment of fetal rat calvaria. Endocrinology 125:1430–1437. [DOI] [PubMed] [Google Scholar]
- 6.Kim SJ, Kim KH. 1997. Insulin rapidly stimulates ERK2 in the membrane of osteoblast-like UMR-106 cell. Biochem Mol Biol Int 43:1023–1031. [DOI] [PubMed] [Google Scholar]
- 7.Beam HA, Parsons JR, Lin SS. 2002. The effects of blood glucose control upon fracture healing in the BB Wistar rat with diabetes mellitus. J Orthop Res 20:1210–1216. [DOI] [PubMed] [Google Scholar]
- 8.Gooch HL, Hale JE, Fujioka H, et al. 2000. Alterations of cartilage and collagen expression during fracture healing in experimental diabetes. Connect Tissue Res 41:81–91. [DOI] [PubMed] [Google Scholar]
- 9.Cornish J, Callon KE, Reid IR. 1996. Insulin increases histo-morphometric indices of bone formation in vivo. Calcif Tissue Int 59:492–495. [DOI] [PubMed] [Google Scholar]
- 10.White JR Jr, Campbell RK, Hirsch I. 1997. Insulin analogues: new agents for improving glycemic control. Postgrad Med 101:58–60 63–65, 70. [DOI] [PubMed] [Google Scholar]
- 11.Engesaeter LB, Ekeland A, Langeland N. 1978. Methods for testing the mechanical properties of the rat femur. Acta Orthop Scand 49:512–518. [DOI] [PubMed] [Google Scholar]
- 12.Balaburski G, O’Connor JP. 2003. Determination of variations in gene expression during fracture healing. Acta Orthop Scand 74:22–30. [DOI] [PubMed] [Google Scholar]
- 13.Gebauer GP, Lin SS, Beam HA, et al. 2002. Low-intensity pulsed ultrasound increases the fracture callus strength in diabetic BB Wistar rats but does not affect cellular proliferation. J Orthop Res 20:587–592. [DOI] [PubMed] [Google Scholar]
- 14.Stuck W 1932. The effect of insulin on the healing of experimental fractures in the rabbit. J Bone Joint Surg 14:109–115. [Google Scholar]
- 15.Schmidmaier G, Wildemann B, Gabelein T, et al. 2003. Synergistic effect of IGF-I and TGF-beta1 on fracture healing in rats: single versus combined application of IGF-I and TGF- beta1. Acta Orthop Scand 74:604–610. [DOI] [PubMed] [Google Scholar]
- 16.Gregory WB Jr. 1965. Effect of insulin on the healing of bone wounds in albino rats. J Dent Res 44:487–492. [DOI] [PubMed] [Google Scholar]
- 17.Phornphutkul C, Wu KY, Gruppuso PA. 2006. The role of insulin in chondrogenesis. Mol Cell Endocrinol 249:107–115. [DOI] [PubMed] [Google Scholar]
- 18.Phornphutkul C, Wu KY, Yang X, et al. 2004. Insulin-like growth factor-I signaling is modified during chondrocyte differentiation. J Endocrinol 183:477–486. [DOI] [PubMed] [Google Scholar]
- 19.Bellows CG, Jia D, Jia Y, et al. 2006. Different effects of insulin and insulin-like growth factors I and II on osteopro-genitors and adipocyte progenitors in fetal rat bone cell populations. Calcif Tissue Int 79:57–65. [DOI] [PubMed] [Google Scholar]
- 20.Fulzele K, DiGirolamo DJ, Liu Z, et al. 2007. Disruption of the insulin-like growth factor type 1 receptor in osteoblasts enhances insulin signaling and action. J Biol Chem 282: 25649–25658. [DOI] [PubMed] [Google Scholar]
- 21.Wildemann B, Lubberstedt M, Haas NP, et al. 2004. IGF-I and TGF-beta 1 incorporated in a poly(D,L-lactide) implant coating maintain their activity over long-term storage-cell culture studies on primary human osteoblast-like cells. Biomaterials 25:3639–3644. [DOI] [PubMed] [Google Scholar]
- 22.Kayal RA, Tsatsas D, Bauer MA, et al. 2007. Diminished bone formation during diabetic fracture healing is related to the premature resorption of cartilage associated with increased osteoclast activity. J Bone Miner Res 22:560–568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Akeno N, Robins J, Zhang M, et al. 2002. Induction of vascular endothelial growth factor by IGF-I in osteoblast-like cells is mediated by the PI3K signaling pathway through the hyp-oxia-inducible factor-2alpha. Endocrinology 143:420–425. [DOI] [PubMed] [Google Scholar]
- 24.Deckers MM, Karperien M, van der Bent C, et al. 2000. Expression of vascular endothelial growth factors and their receptors during osteoblast differentiation. Endocrinology 141:1667–1674. [DOI] [PubMed] [Google Scholar]
- 25.Kayal RA, Alblowi J, McKenzie E, et al. 2009. Diabetes causes the accelerated loss of cartilage during fracture repair which is reversed by insulin treatment. Bone 44:357–363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Holstein JH, Klein M, Garcia P, et al. 2008. Rapamycin affects early fracture healing in mice. Br J Pharmacol 154: 1055–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li M, Amizuka N, Oda K, et al. 2004. Histochemical evidence of the initial chondrogenesis and osteogenesis in the periosteum of a rib fractured model: implications of osteocyte involvement in periosteal chondrogenesis. Microsc Res Tech 64:330–342. [DOI] [PubMed] [Google Scholar]
- 28.Paglia DN, Wey A, Park AG, et al. 2012. The Effects of local vanadium treatment on angiogenesis and chondrogenesis during fracture healing. J Orthop Res 30:1971–1978. [DOI] [PubMed] [Google Scholar]
- 29.Fiorelli G, Orlando C, Benvenuti S, et al. 1994. Characterization, regulation, and function of specific cell membrane receptors for insulin-like growth factor I on bone endothelial cells. J Bone Miner Res 9:329–337. [DOI] [PubMed] [Google Scholar]
- 30.Ferron M, Wei J, Yoshizawa T, et al. 2010. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell 142:296–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


