Abstract
Serum immunoglobulin A (IgA) is a biomarker of liver disease severity in adult nonalcoholic fatty liver disease (NAFLD). The objective of this study was to assess whether this is true in pediatric NAFLD. This was a retrospective study of children followed in the Steatohepatitis Center of a tertiary care center (2010–2017). Serum IgA levels, histology and biochemical markers of hepatocellular injury and metabolic dysregulation were collected. Of the 600 children with available serum IgA levels (mean age 14±3 years, 62% boys, 80% non-Hispanic), IgA was elevated in 4%. The group with elevated IgA levels was younger and had a greater representation of Hispanic ethnicity compared with those with normal or low IgA. Body mass index and aminotransferases were not different between the groups. Histology was available for 170 children and was not different in those with elevated, normal and low IgA. In contrast to adults, serum IgA is not a biomarker of liver disease severity in pediatric NAFLD.
Keywords: children, diagnosis, fibrosis, immunoglobulin A, nonalcoholic steatohepatitis
Nonalcoholic fatty liver disease (NAFLD) can lead to the development of hepatic fibrosis, which in adults is the single most important determinant of long-term patient outcomes (1). The presence and severity of fibrosis is most accurately determined with histology; however, practitioners are often reluctant to obtain liver biopsies, in part due to their invasive nature combined with the present lack of highly effective treatments for NAFLD (2,3). Laboratory-based approaches to non-invasively assessing fibrosis, such as the use of serum biomarkers, either in isolation or as part of fibrosis scores, have not been shown to perform well in pediatric NAFLD (4). Research is needed to identify accurate, non-invasive biomarkers of liver disease severity. Adult literature suggests that serum immunoglobulin A (IgA) levels may function as such a biomarker (5,6).
Serum IgA levels are often elevated in patients with alcoholic fatty liver disease (AFLD) (7). Given the similarities between AFLD and NAFLD this finding has prompted further investigations regarding the role of serum IgA in adults with NAFLD (5,6). IgA has been shown to be the immunoglobulin most frequently elevated in this context, seen in up to 44% of patients with NAFLD, compared to 10% for IgG elevation and 8% for IgM elevation(5). Furthermore, IgA levels are increased more often and to a greater extent in adults with nonalcoholic steatohepatitis (NASH) compared to those with non-alcoholic fatty liver (NAFL). Similarly, the percentage of patients with elevated serum IgA levels increases progressively with fibrosis stage, and the degree of IgA elevation correlates with fibrosis severity (5). The addition of serum IgA levels to a mathematical model combining patient characteristics (age and body mass index [BMI]) and simple laboratory investigations (alanine aminotransferase [ALT], aspartate aminotransferase [AST]) improves the accuracy of advanced fibrosis prediction to an AUROC of 0.87 (5). There are no data on the utility of serum IgA levels in predicting liver disease severity in children with NAFLD.
The objective of this study was to assess serum IgA levels in a large pediatric cohort with NAFLD and to investigate whether they are reflective of liver disease severity.
METHODS
We conducted a retrospective analysis of children with presumed or confirmed NAFLD evaluated in the Cincinnati Children’s Steatohepatitis Center between August 2010 and October 2017, following approval by the institutional review board and waiver of informed consent.
Inclusion criteria were a diagnosis of confirmed (with histology) or presumed NAFLD (elevated transaminases in the context of obesity with or without radiographic evidence of steatosis of the liver and a negative workup for other liver or endocrine disorders)(3). Exclusion criteria were diagnoses of immune dysregulation, rheumatologic conditions or inflammatory bowel disease, or secondary causes of fatty liver disease (eg, medications) or missing serum IgA levels.
Data collected for this study included baseline demographics, anthropometrics, results of total serum IgA levels, as well as serum ALT, AST, gamma glutamyl transferase [GGT], alkaline phosphatase, albumin, international normalized ratio [INR] and complete blood count levels obtained within 3 months of the IgA level. Increased serum levels of ALT, as well as AST and GGT, have been independently associated with increased histological severity of pediatric NAFLD (8–10) and were therefore used as surrogate markers of disease severity. In addition, data from available liver biopsies were collected. Liver disease severity was determined histologically using the scoring system developed by Kleiner et al (11). The biopsies were scored prospectively by experienced pathologists of a quaternary pediatric care center. The scores for steatosis, lobular inflammation and ballooning degeneration were added to calculate the NAFLD activity score (NAS). Significant fibrosis was defined as fibrosis stage 2 or higher.
Different age-specific cutoffs determined by the laboratory at Cincinnati Children’s Hospital Medical Center were used to determine the presence of low, high and normal serum IgA levels.
Statistical Analyses
Descriptive statistics were used and continuous data were presented as means with standard deviation (SD) or medians (range) depending on their distribution. Comparisons between groups were made using the Student’s t test for normally distributed continuous variables and the Mann-Whitney U test for not-normally distributed variables. Chi-square testing was used to compare proportions. Analysis of variance was used to compare continuous variables across different groups. Pearson correlation was used to identify associations between variables. Statistical analyses were performed using Stata MP 13.0 (College Station, TX).
RESULTS
Of 797 children and young adults (mean age 13±3 years, 62% male, 80% non-Hispanic, median BMI 34 kg/m2) who had screening bloodwork upon referral to the NASH clinics at CCHMC since August 2010, 600 (75%) had had serum IgA levels measured. The primary reason for missing IgA levels is that celiac disease screening, which is currently recommended by the most recent pediatric NAFLD guidelines for patients with presumed NAFLD(3), was not done routinely until 2012.
The median serum IgA level of the entire cohort was 147 mg/dL (range 6–446). According to age-specific cutoffs, 23 patients (4%) had elevated IgA and 30 (5%) were IgA deficient. The clinical and biochemical characteristics of patients grouped by IgA levels are shown in Table 1. Compared to those with normal or low IgA levels, the group of patients with high IgA levels had a greater representation of younger, Hispanic children. Biochemical markers of liver injury were not different between the groups.
TABLE 1.
Characteristics of patients grouped by immunoglobulin A category
| Variable | Low IgA (n = 30) | Normal IgA (n = 547) | Elevated IgA (n = 23) | P |
|---|---|---|---|---|
| Age (±SD), y | 14 ± 2 | 13 ± 3 | 11 ± 3 | 0.01 |
| Sex, n (%male) | 17 (57%) | 341 (62%) | 14 (61%) | 0.82 |
| Ethnicity (% non-Hispanic) | 28 (93%) | 440 (80%) | 13 (57%) | 0.02 |
| BMI, kg/m2 | 35 ± 6 | 35 ± 8 | 34 ± 9 | 0.68 |
| ALT, U/L | 64 ± 33 | 76 ± 57 | 76 ± 41 | 0.55 |
| AST, U/L | 30 ± 21 | 44 ± 30 | 46 ± 26 | 0.66 |
| GGT, U/L | 40 ± 22 | 44 ± 46 | 40 ± 22 | 0.83 |
| ALP, U/L | 205 ± 120 | 205 ± 112 | 231 ± 102 | 0.53 |
| Histology | Low IgA (n = 8) | Normal IgA (n = 160) | High IgA (n = 2) | p value |
| NAS | ||||
| <5 | 7 (87%) | 110 (69%) | 1 (50%) | 0.44 |
| ≥5 | 1 (13%) | 50 (31%) | 1 (50%) | |
| Ballooning score | ||||
| 0 | 2 (25%) | 68 (42%) | 1 (50%) | 0.69 |
| 1 | 6 (75%) | 80 (50%) | 1 (50%) | |
| 2 | 0 (0%) | 12 (8%) | 0 (0%) | |
| Fibrosis presence | ||||
| no | 4 (50%) | 78 (49%) | 0 (0%) | 0.87 |
| yes | 4 (50%) | 81 (51%) | 2 (100%) | |
| Fibrosis stage | ||||
| 0 | 4 (50%) | 78 (49%) | 0 (33%) | 0.42 |
| 1 | 3 (38%) | 51 (32%) | 1 (33%) | |
| 2 | 1 (12%) | 16 (10%) | 0 (0%) | |
| 3 | 0 (0%) | 14 (9%) | 1 (33%) |
Variables are shown as means ± (SD).
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BMI = body mass index; GGT = gamma glutamyl transferase; IgA = immunoglobulin A; NAFLD = nonalcoholic fatty liver disease; NAS = NAFLD Activity Score.
There was no correlation between serum IgA levels and ALT or AST (r = 0.03, P = 0.42; and r = 0.04, P = 0.29; respectively). Weak correlations were found between IgA levels and BMI (r = 0.14, P < 0.01), GGT (r = 0.11, P < 0.01) and alkaline phosphatase (r = −0.17, P < 0.01). When dividing the cohort by HbA1c levels, no difference in serum IgA levels was found between those without (HbA1c ≤ 5.7%) and those with evidence of pre-diabetes/diabetes (HbA1c > 5.8%; mean (±SD) IgA = 157±72 vs 170 ± 77 mg/dL, respectively; P = 0.17).
Histology was available for 170 patients (n = 8 of the 30 with low IgA [27%], n = 160 of the 547 with normal IgA [29%] and n = 2 of the 23 with elevated IgA [9%]). The overall proportion of low, normal and high IgA in the subset with histology was 5%, 94%, and 1%, respectively. The mean steatosis score of the entire cohort was1.9 ± 0.9 and the mean score for lobular inflammation was 1.2 ± 0.7. Significant fibrosis (fibrosis stage ≥ 2) was seen in 32 (19%) patients; the proportion of patients with fibrosis stages 0, 1, 2, and 3 was 48%, 33%, 10%, and 9%, respectively. No patient had stage 4 fibrosis. IgA levels were measured at a median time of 160 days before the liver biopsy (interquartile range 48–445). There was no correlation between serum IgA levels and time from blood-work to the liver biopsy (r = −0.10, P = 0.20).
There was no difference in steatosis severity (mean score:1.9 ± 1.0 vs 1.9 ± 0.9 vs 2.5 ± 0.7, respectively; P = 0.63), lobular inflammation severity (mean score: 0.9 ± 1.0 vs 1.2±0.7 vs1.5 ± 0.7, respectively; P = 0.35), or NAS (mean score 3.5±1.6 vs 3.7 ± 1.6 vs 4.5 ± 2.1, respectively; P = 0.90) between patients with low, normal and elevated serum IgA levels. The NAS was ≥5 in 51 (30%) patients. Serum IgA levels were not different in those with NAS < 5 compared to those with NAS ≥ 5 (151 ± 67 mg/dL vs 146 ± 70 mg/dL, respectively; P = 0.35). The distribution of IgA levels did not vary by NAS score (P = 0.86). The proportion of patients with NAS < 5 and ≥5 was also not different between the groups of patients with low, normal and elevated IgA levels (Table 1). There were no significant differences in IgA levels of patients with different degrees of portal inflammation (143 ± 67 mg/dL vs 156 ± 69 mg/dL vs 142 ± 64 in those with portal inflammation score of 0, 1, and 2, respectively; P = 0.45).
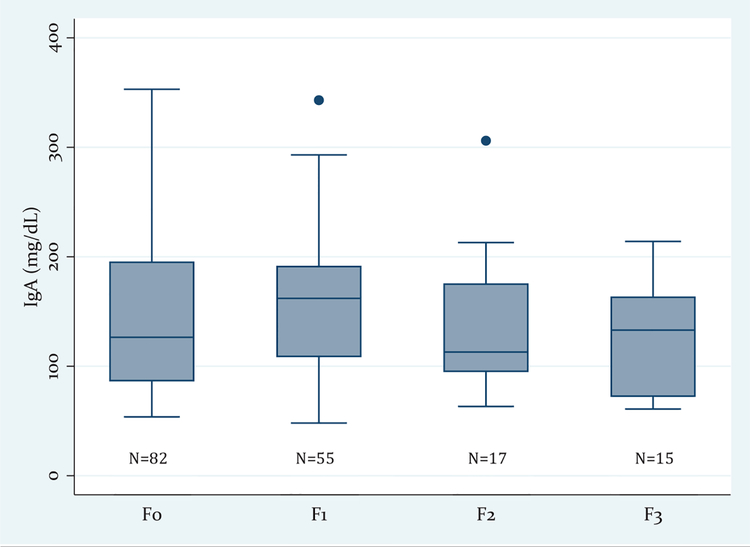
Serum IgA levels across different stages of fibrosis are shown in Figure 1. There was no difference in serum IgA levels between those with (F1-F3) and without (F0) fibrosis (146 ± 72 mg/dL vs 152 ± 63, P = 0.62). The proportion of patients with different fibrosis stages was not different between the groups (Table 1).
FIGURE 1.
Distribution of serum immunoglobulin A (IgA) levels across different fibrosis stages. The boxes represent interquartile ranges and the horizontal lines in the boxes represent median values.
DISCUSSION
In a large cohort of predominantly non-Hispanic, obese children with NAFLD, serum IgA levels were elevated in only 4% of patients. Children and adolescents with elevated IgA levels were more likely to be younger and of Hispanic ethnicity compared to those with normal or low IgA levels. In the entire cohort, IgA levels were not associated with metabolic dysregulation or liver disease severity, as reflected by degree of serum amino-transferase elevation. Furthermore, in a large sample of patients with histologically characterized NAFLD, the distribution of IgA levels did not differ by NAS and fibrosis severity, nor did the proportion of patients with high, normal or low IgA level differ by NAFLD severity.
To date, adult literature on serum IgA levels in the context of NAFLD has yielded somewhat conflicting results. McPherson et al found that almost half (46%) of adults with NAFLD in Australia had elevated serum IgA levels (5). A study from Iran however, failed to show a difference in serum IgA levels between patients with NAFLD and healthy controls (12). Both studies, as well as another report from Japan, reported higher IgA levels in patients with more advanced liver disease, particularly those with NASH and advanced fibrosis (5,6,12). In our large pediatric cohort, we found a low prevalence of high IgA levels. Further, both serum IgA levels, as well as the proportion of patients with elevated IgA were not different in those with more advanced liver disease.
It is not clear why patients with NAFLD would have elevated IgA levels and whether IgA plays a role in the pathogenesis of this condition. It has been postulated that dysbiosis seen in the context of NAFLD (13,14) and its impact on the intestinal barrier and the immune system may contribute to IgA release from the gut (5). Indeed, intestinal plasma cells are a major source of IgA synthesis(15). IgA secreted in the intestinal lumen not only prevents micro-organisms from crossing the gut barrier but also alters the intestinal microbiota composition, as shown in various animal models (16). One can also hypothesize that differences in the microbiome between adults and children may contribute to the discrepancies between the previously reported adult literature and the results of this study. Our study was not designed to address this hypothesis, which remains to be investigated further.
The histological data available for this study were obtained from patients who had undergone liver biopsies for clinical indications. At our institution, the decision to proceed with a liver biopsy is made when other liver diseases need to be excluded or when there is a concern regarding advanced liver disease, for example, in the context of persistently (>6 months) and significantly elevated (ALT > 50 U/L) or rising liver enzymes in patients with risk factors that have been associated with more severe disease (eg, dyslipidemia, prediabetes or type 2 diabetes, obstructive sleep apnea, etc). Interestingly, the results of our study revealed that, while approximately one-third of those with normal or low IgA had undergone liver biopsies, the patients with elevated serum IgA levels had undergone liver biopsies at a much lower frequency of only 9%. This may be a reflection of the younger age of this group of patients; however, it does also suggest that from a biochemical perspective there may have been less evidence of increased liver disease severity over time in this subgroup, further strengthening the results of our study that overall found no association between elevated IgA levels and increased NAFLD severity.
The strengths of our study include its novelty, as it is the first to determine prevalence of elevated IgA in a large cohort of children with NAFLD. We also had a large cohort of children with histologic confirmation and staging of liver disease severity, with a sample size comparable to other published studies that have identified clinical or biochemical characteristics associated with severity of NAFLD in children (cohort size ranging from 149 to 186 children) (8,17–19). Limitations of our study include its retrospective nature and the variable timeframe between measuring serum IgA levels and obtaining the histology data. In addition, the low prevalence of high IgA levels in the cohort in general, and among the subset with biopsy data, precluded multivariable analyses to study the association between IgA and serological or histologic features of NAFLD. Finally, our cohort is predominantly non-Hispanic, reflecting the demographics of our regional pediatric NAFLD population. Therefore, generalizability to Hispanic youth with NAFLD is unknown.
In summary, serum IgA is not an optimal biomarker of liver disease severity in pediatric NAFLD.
What Is Known
There are no accurate biochemical biomarkers of liver disease severity in pediatric nonalcoholic fatty liver disease.
Non-invasive biomarkers of liver disease severity are needed.
Serum immunoglobulin A levels are often elevated in adults with nonalcoholic fatty liver disease and correlate with liver disease severity.
What Is New
Only a minority of pediatric patients with nonalcoholic fatty liver disease have elevated serum immunoglobulin A levels.
Immunoglobulin A levels do not correlate with serum aminotransferase levels or histological liver disease severity in pediatric nonalcoholic fatty liver disease.
Immunoglobulin A levels should not be used as a disease biomarker in children with nonalcoholic fatty liver disease.
Footnotes
The authors report no conflicts of interest.
REFERENCES
- 1.Ekstedt M, Franzén LE, Mathiesen UL, et al. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology 2006;44:865–73. [DOI] [PubMed] [Google Scholar]
- 2.Mouzaki M, Ling SC, Schreiber RA, et al. Management of pediatric non-alcoholic fatty liver disease by academic hepatologists in Canada: a nationwide survey. J Pediatr Gastroenterol Nutr 2017;65: 380–3. [DOI] [PubMed] [Google Scholar]
- 3.Vos MB, Abrams SH, Barlow SE, et al. NASPGHAN clinical practice guideline for the diagnosis and treatment of nonalcoholic fatty liver disease in children: recommendations from the expert committee on NAFLD (ECON) and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN). J Pediatr Gastroenterol Nutr 2017;64:319–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jackson JA, Konomi JV, Mendoza MV, et al. Performance of fibrosis prediction scores in paediatric non-alcoholic fatty liver disease. J Paediatr Child Health 2018;54:172–6. [DOI] [PubMed] [Google Scholar]
- 5.McPherson S, Henderson E, Burt AD, et al. Serum immunoglobulin levels predict fibrosis in patients with non-alcoholic fatty liver disease. J Hepatol 2014;60:1055–62. [DOI] [PubMed] [Google Scholar]
- 6.Tomita K, Teratani T, Yokoyama H, et al. Serum immunoglobulin a concentration is an independent predictor of liver fibrosis in nonalcoholic steatohepatitis before the cirrhotic stage. Digest Dis Sci 2011;56:3648–54. [DOI] [PubMed] [Google Scholar]
- 7.van de Wiel A, Schuurman HJ, Kater L. Alcoholic liver disease: an IgA-associated disorder. Scand J Gastroenterol 1987;22:1025–30. [DOI] [PubMed] [Google Scholar]
- 8.Patton HM, Lavine JE, Van Natta ML, et al. Clinical correlates of histopathology in pediatric nonalcoholic steatohepatitis. Gastroenterology 2008;135:1961–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xanthakos SA, Jenkins TM, Kleiner DE, et al. High prevalence of nonalcoholic fatty liver disease in adolescents undergoing bariatric surgery. Gastroenterology 2015;149,:623–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schwimmer JB, Newton KP, Awai HI, et al. Paediatric gastroenterology evaluation of overweight and obese children referred from primary care for suspected non-alcoholic fatty liver disease. Aliment Pharmacol Ther 2013;38:1267–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005;41:1313–21. [DOI] [PubMed] [Google Scholar]
- 12.Maleki I, Aminafshari MR, Taghvaei T, et al. Serum immunoglobulin A concentration is a reliable biomarker for liver fibrosis in non-alcoholic fatty liver disease. World J Gastroenterol 2014;20:12566–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mouzaki M, Comelli EM, Arendt BM, et al. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology 2013;58: 120–7. [DOI] [PubMed] [Google Scholar]
- 14.Zhu L, Baker SS, Gill C, et al. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology 2013;57:601–9. [DOI] [PubMed] [Google Scholar]
- 15.Strugnell RA, Wijburg OL. The role of secretory antibodies in infection immunity. Nat Rev Microbiol 2010;8:656–67. [DOI] [PubMed] [Google Scholar]
- 16.Inamine T, Schnabl B. Immunoglobulin A and liver diseases. J Gastroenterol 2018;53:691–700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Suzuki A, Abdelmalek MF, Schwimmer JB, et al. Association between puberty and features of nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 2012;10:786–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Corey KE, Vuppalanchi R, Vos M, et al. Improvement in liver histology is associated with reduction in dyslipidemia in children with nonalcoholic fatty liver disease. J Pediatr Gastroenterol Nutr 2015;60:360–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vos MB, Belt P, Molleston JP, et al. Correlation of vitamin E uric acid, and diet composition with histologic features of pediatric NAFLD. J Pediatr Gastroenterol Nutr 2012;54:90–6. [DOI] [PMC free article] [PubMed] [Google Scholar]