Abstract
Background
Implantable cardioverter-defibrillator (ICD) lead parameters may deteriorate due to right ventricular (RV) disease such as arrhythmogenic right ventricular cardiomyopathy (ARVC), with implications for safe delivery of therapies. We compared ICD and CRT-D (cardiac resynchronisation therapy-defibrillator) lead parameters in patients with ARVC and dilated cardiomyopathy (DCM).
Methods
RV lead sensing (R wave amplitude) and pacing (threshold and amplitude-pulse width product (APWP)), left ventricular (LV) pacing (APWP), and imaging parameter trends were assessed in 18 patients with ARVC and 18 with DCM.
Results
R wave amplitude did not change significantly over time in either group (over 5 years, ARVC -0.4 mV, 95% CI -3.8–3.0 mV; DCM -1.8 mV, 95% CI -5.0–1.3 mV). Within ARVC group, divergent trends were seen according to lead position. DCM patients experienced an increase in RV lead threshold (+1.1 V over 5 years, 95% CI + 0.5 to +1.7 V) and RV APWP (+0.48 Vms over 5 years, 95% CI + 0.24 to +0.71 Vms); ARVC patients had no change. ARVC patients had a higher LVEF at baseline than DCM patients (52 vs 20%, p < 0.001), though LVEF decreased over time for the former, while increasing for the latter. TAPSE did not change over time for ARVC patients.
Conclusions
Lead parameters in ARVC patients were stable over medium-term follow up. In DCM patients, RV lead threshold and RV and LV APWP increased over time. These differential responses for DCM and ARVC were not explained by imaging indices, and may reflect distinct patterns of disease progression.
Keywords: ICD, CRT, ARVC, Lead parameters, Dilated cardiomyopathy
1. Introduction
Cardiomyopathies such as arrhythmogenic (right ventricular) cardiomyopathy (ARVC) and dilated cardiomyopathy (DCM) often progress over time, with deterioration in myocyte contractile function, disruption of myocardial architecture, and associated electrophysiological changes. Implantable cardioverter defibrillators (ICDs) and cardiac resynchronisation therapy – defibrillators (CRT-Ds) are often implanted in patients with these diseases. Disease progression raises the possibility that sensing or pacing parameters in these devices may deteriorate, with potential implications for safe delivery of tachycardia therapies in conditions involving the right ventricle (RV) [7]. In ARVC, fibro-fatty change can develop progressively, though the interventricular septum and left ventricle (LV) are commonly spared [3]. Placement of defibrillator leads in regions of healthy tissue, or epicardially on the LV via the coronary sinus, could help ensure safe device function, and optimal current drain, thereby preserving longevity.
We hypothesised that sensed R wave voltage in the RV of patients with ARVC decreases over time. To investigate, we compared ICD and CRT-D lead parameters, and indices of ventricular function, in patients with ARVC and DCM, with the latter acting as a comparator group.
2. Methods
2.1. Study subjects
We reviewed records of patients undergoing ICD or CRT-D implant at St Bartholomew's and the Heart Hospitals, London, between 2011, when device check records first began to be stored electronically, and 2016. There were 1676 patients who underwent ICD or CRT-D implant in this period; of these, 18 patients with a definite diagnosis of ARVC receiving de novo ICD/CRT-D were identified, and compared with 18 consecutive patients with a definite diagnosis of idiopathic DCM who underwent de novo device implant over the same period. Patients receiving de novo devices were chosen to minimise interaction of infections and other procedures such as upgrades and lead revisions on lead parameters and outcomes. Diagnoses were established in a specialist heart muscle disease clinic according to contemporaneous diagnostic criteria [8]. Data from all included patients were used in the analyses. Patient information was de-identified.
2.2. Device data
Implant records were reviewed to determine type of device, and whether the RV lead used active or passive fixation. Site of RV lead position was determined from the implant report. The following ventricular lead parameters were determined at implant, at one month follow up, and then at six monthly follow up intervals thereafter: RV lead R wave amplitude, RV lead threshold (voltage amplitude and pulse width), and high voltage (HV) electrode impedance, LV lead threshold (voltage amplitude and pulse width). Amplitude-pulse width product (APWP) was calculated and used as a means of incorporating the much more variable pulse widths used with LV leads in threshold testing.
2.3. Imaging data
Cardiac imaging reports for echocardiography and MRI were reviewed to obtain the baseline LV ejection fraction (LVEF) and tricuspid annular plane systolic excursion (TAPSE) measurements, as indices of LV and RV systolic function, respectively. These indices were then recorded from follow-up imaging investigations to allow investigation of trends over time.
2.4. Statistics
All statistical analysis was performed with StataIC 14 (StataCorp LP, USA), and figures were produced with this. Multilevel mixed effects models were employed to analyse repeated measures data, due to their ability to model within-patient variation, and their tolerance of missing data. Statistical significance was assumed if p < 0.05.
3. Results
Group demographic and device characteristics are summarised in Table 1. Follow up data were available for a mean time of 30 months (standard deviation 18 months). For device lead parameters, the implant value and ≥1 follow up measurement were available for all 36 patients, except for RV threshold, RV APWP and HV impedance (data for 35 patients). Baseline and ≥1 follow up measurement of LVEF was available for 22 patients, and TAPSE measurements for 11 of the ARVC patients.
Table 1.
Patient characteristics by group.
| ARVC group | DCM group | |
|---|---|---|
| Age (mean) | 51 | 60 |
| Male | 9/18 | 12/18 |
| Device type | ||
| ICD (%) | 17 (94) | 4 (22) |
| CRT-D (%) | 1 (6) | 14 (78) |
| Secondary prevention (%) | 13 (28) | 15 (17) |
| Median no. of leads | 2 | 3 |
| RV lead site | ||
| Septal (%) | 13 (72) | 9 (50) |
| Apical/free wall (%) | 5 (28) | 9 (50) |
| Active RV lead fixation (%) | 17 (94) | 17 (94) |
3.1. Patient and device-related events
One patient had a generator re-siting a month after implant, though the leads were not repositioned. One patient with DCM and CRT-D underwent heart transplant over a year after implant, and the device was extracted at this time. Another patient with DCM and CRT-D died 13 months after implant. One patient with DCM and ICD required atrial lead repositioning 2 months after implant, and this patient died 14 months after implant. And one patient required extraction of their CRT-D 36 months after implant due to infection.
3.2. RV (defibrillator) lead R wave
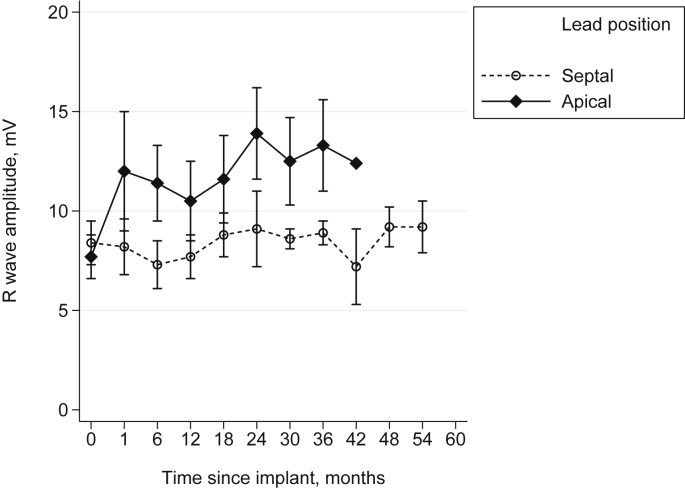
There was a significant difference in R wave amplitude between diseases at baseline, with R wave amplitudes in the ARVC group lower than those in the DCM group (Δ −5.6 mV, 95% confidence interval (CI) −8.8 to −2.5 mV, p < 0.001). For neither group was there a significant change in R wave over time (Fig. 1). Fig. 2 shows an example of the multilevel modelling, with the measured values and model's predicted values for each patient.
Fig. 1.
RV lead R wave amplitude trends over time for ARVC and DCM patients (means ± standard error on the mean (SEM)).
Fig. 2.
Multilevel model – measured and model's fitted values for R wave amplitude trends in ARVC and DCM patients.
When R wave trends were analysed according to lead position, in ARVC patients there was no difference in mean implant R wave amplitude between septal and non-septal leads (p = 0.463) (Fig. 3). Septal leads showed a small decrease in R wave over time (−2.0 mV over 5 years, 95% CI -3.8 to −0.2 mV, p = 0.026), while non-septal leads showed a small increase (+3.6 mV over 5 years, 95% CI 0.8–6.4 mV, p = 0.013). In DCM patients, there was no difference between sites in mean implant value (p = 0.730). There was no significant change in R wave over time for septal (+1.1 mV over 5 years, 95% CI -4.0–6.2 mV, p = 0.680), or non-septal leads (−4.3 mV over 5 years, 95% CI -9.0–0.4 mV, p = 0.071).
Fig. 3.
RV lead R wave amplitude trends over time for ARVC patients by lead position (means ± SEM).
3.3. RV lead threshold – voltage amplitude
There was a significant difference between diseases at baseline: thresholds in the ARVC group were significantly higher than those of the DCM group (Δ +0.2 V, 95% CI 0.0–0.4 V, p = 0.031) (Fig. 4). For DCM patients there was a significant increase in threshold over time, equivalent to +1.1 V over 5 years (95% CI 0.5–1.7 V, p = 0.001), while for ARVC patients there was no significant change seen (p = 0.336). When analysed according to lead position, in the ARVC group there was no change over time for either septal (p = 0.509), or non-septal leads (p = 0.811). In the DCM group, septally-positioned leads showed an increase in threshold over time (+1.5 V over 5 years, 95% CI 0.3–2.7 V, p = 0.016), whereas non-septal leads did not change significantly (+0.9 V, 95% CI -0.3–2.1 V, p = 0.150).
Fig. 4.
RV lead threshold voltage amplitude trends over time for ARVC and DCM patients (means ± SEM).
3.4. RV lead threshold – amplitude-pulse width product
RV lead APWP in the ARVC group was significantly larger than in the DCM group at baseline (Δ +0.11 Vms, 95% CI 0.02 to 0.20 Vms, p = 0.018) (Fig. 5). As for voltage amplitude threshold, there was a highly significant increase over time equivalent to +0.48 Vms over 5 years (95% CI 0.24 to 0.71 Vms, p < 0.001) for DCM patients, while for ARVC patients there was no significant change (p = 0.249).
Fig. 5.
RV lead threshold amplitude-pulse width product trends over time for ARVC and DCM patients (means ± SEM).
3.5. HV lead impedance
There was no significant difference between diseases at baseline.
For DCM patients there was a modest magnitude increase in HV over time which was significant (+7 Ω over 5 years, 95% CI 2–12 Ω, p = 0.003). For ARVC patients, there was a similar modest magnitude increase in HV over time which was also significant (+8 Ω, 95% CI 3–13 Ω, p = 0.002).
3.6. LV lead threshold – amplitude-pulse width product
Only 1 patient with ARVC had a CRT-D, so analysis was restricted to the DCM group in whom 14/18 patients had a CRT-D. For these patients the baseline value in LV APWP was 0.33 Vms, and there was a significant increase over time, equivalent to +0.58 Vms over 5 years (95% CI 0.05 to 1.11 Vms, p = 0.033).
3.7. LV ejection fraction
There was a highly significant difference between groups at baseline, with LVEF higher in the ARVC group (Δ +31%, 95% CI 19–43%, p < 0.001). However, there was a highly significant reduction of moderate magnitude over time for this group, equivalent to −8% over 5 years (95% CI -12 to −3%, p < 0.001), whilst for DCM patients there was a highly significant increase of moderate magnitude over time (+13% over 5 years, 95% CI 4–22%, p = 0.004) (Fig. 6).
Fig. 6.
LV ejection fraction trends over time for ARVC and DCM patients (means ± SEM).
3.8. Tricuspid annular plane systolic excursion
Only 3 DCM patients had 2 or more TAPSE measurements, so statistical analysis was restricted to the ARVC group in which 11/18 patients had ≥2 measurements. There was no significant change in TAPSE over time for these patients (p = 0.685) (Fig. 7).
Fig. 7.
Tricuspid annular plane systolic excursion (TAPSE) trend over time for ARVC patients (means ± SEM). (In the DCM group, only 3 patients had repeated TAPSE measurements).
3.9. Ventricular pacing percentage
The median ventricular pacing percentage in the ARVC group was 0% (interquartile range 0–0.7%), while that for the DCM group was 98.8% (interquartile range 94–99%).
4. Discussion
Currently there is no guideline-based recommendation of minimum R wave amplitude at implant for patients receiving an ICD or CRT-D. However, an amplitude of ≥ 5–7 mV is considered safe [16,17]. Lillo-Castellano et al. found that with an R wave amplitude of ≤2.5 mV there was a significant risk of undersensing in VF [7]. Ruetz et al. concluded that there was no minimum sinus rhythm R wave necessary to ensure reliable sensing of VF, though less than 4% of patients in their study had R wave amplitudes <3 mV [14].
We found a highly significant difference of moderate magnitude in R wave amplitude between patients with ARVC and DCM at baseline. The only other study to date to compare baseline characteristics between patients with ARVC and a comparator group found no difference [4]. This is difficult to explain, but the findings below do not support this being related to disease process.
In contrast to the study by Mugnai et al. [11], there was no significant change in RV R wave amplitude over time in our ARVC group. Interestingly, when this group was analysed according to lead position, for those placed on the septum there was a small but significant deterioration in R wave, while those in non-septal sites (4 out of 5 at the apex) showed a small but significant increase in R wave over time. This contrasts with the findings of Herman et al., who reported no change in R wave in septally-positioned leads, though a deterioration for apical leads [4]. Our findings were unexpected, but together with those of Herman et al., suggest that while sensing may change slightly over the medium term, and lead position may affect this, the magnitude of change is small and therefore may not be of clinical significance. More extended follow up is required to know whether this deterioration is important in the long term.
The lack of change in RV threshold indices in the ARVC group is concordant with other published reports in which either no, or minimal change in threshold was observed [4,11]. The DCM group on the other hand experienced a moderate increase in RV and LV threshold parameters over medium term follow up. Medi and Mond found stable lead parameters for pacemaker leads placed on the right ventricular outflow tract septum [9]. And Liang et al. compared septal and apical lead placement, and found that position in itself does not affect R wave amplitude or threshold significantly in patients with pacemakers [6], findings supported by Kristiansen et al. in the setting of CRT [5]. Thus, it seems the RV lead threshold changes observed in the DCM group in our study are likely disease, rather than lead–related.
In their study of coronary sinus LV leads, Steffel et al. noted no change in threshold energy over one year follow up [15]. However, La Rocca et al. noted an increase in mean pacing energy threshold over a mean of 41.7 months' follow up, concordant with our results [13].
LVEF was seen to fall slightly over the course of the study period in the ARVC group. A possible reason for this is left ventricular involvement of the disease. Whilst less commonly seen, it is nevertheless reported [3]. The lack of change of TAPSE suggests RV function did not decline dramatically. And the ventricular pacing burden of the group was low, making right ventricular pacing-induced LV dysfunction highly unlikely. On the other hand, the DCM group experienced an increase in LVEF, despite increases in RV and LV pacing threshold parameters. The most plausible explanations are an effect related to CRT itself [2,10], pharmacological heart failure therapy, or resolution of or compensation for the myocardial insult that had caused the DCM.
With regards to why threshold parameters increased in the DCM group but not in the ARVC group, differences in pathophysiology and tissue inflammatory response may account for part of this. For example, there is some evidence to suggest an inflammatory response forms part of the pathogenesis in ARVC [1]. While it may be expected that such a response would increase pacing thresholds, perhaps conversely it could improve current transfer at the electrode-tissue interface.
Our study has several strengths. Firstly, we assessed lead parameters at multiple time points for each patient, ensuring reliability in recorded measurements, and maximisation of use of data. Secondly, we recorded up to five years' follow up for patients with ARVC and DCM with either ICD or CRT-D devices. Thirdly, to our knowledge, this is the first report combining ICD/CRT-D lead and imaging follow up data. Finally, with APWP we have created a new method to account for the real-world variability of pulse-widths used in LV pacing with CRT devices.
4.1. Study limitations
There were reasonably small numbers of patients in each group, as only patients with a confirmed diagnosis of ARVC and DCM, and new device implant, were included.
5. Conclusions
Current guidelines do not make specific recommendations regarding the position of ICD RV lead placement for patients with ARVC [12]. Overall in our ARVC cohort, there was no significant change in sensing over medium term follow up. Unexpectedly, R wave amplitude for septally-positioned leads in this group deteriorated slightly, whilst it increased slightly for non-septal (mostly apical) leads. Over medium term follow up, these small changes in sensing do not appear to be clinically significant, and in our study did not result in the need for lead replacement. Therefore either position would appear to produce acceptable lead parameters, though it may be preferable to avoid implanting ICD leads at the true apex due to the risk of wall perforation. Longer term follow up is required to assess lead parameter trends in this predominantly young patient group.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Compliance with ethical standards
For this type of study, formal consent is not required.
Conflicts of interest
Disclosures: RJS has received research grants from Boston Scientific, Medtronic and St Jude. All other authors report no conflict of interest.
Footnotes
Peer review under responsibility of Indian Heart Rhythm Society.
References
- 1.Campuzano O., Alcalde M., Iglesias A., Barahona-Dussault C., Sarquella-Brugada G., Benito B., Arzamendi D., Flores J., Leung T.K., Talajic M., Oliva A., Brugada R. Arrhythmogenic right ventricular cardiomyopathy: severe structural alterations are associated with inflammation. J Clin Pathol. 2012;65:1077–1083. doi: 10.1136/jclinpath-2012-201022. [DOI] [PubMed] [Google Scholar]
- 2.Cleland JG, Daubert JC, Erdmann E, Freemantle N, Gras D, Kappenberger L, Tavazzi L, Cardiac Resynchronization-Heart Failure (CARE-HF) Study Investigators (2005) the effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med;352:1539–1549. [DOI] [PubMed]
- 3.Corrado D., Link M.S., Calkins H. Arrhythmogenic right ventricular cardiomyopathy. N Engl J Med. 2017;376:61–72. doi: 10.1056/NEJMra1509267. [DOI] [PubMed] [Google Scholar]
- 4.Herman A.R.M., Gardner M., Steinberg C., Yeung-Lai-Wah J.A., Healey J.S., Leong-Sit P., Krahn A.D., Chakrabarti S. Long-term right ventricular implantable cardioverter-defibrillator lead performance in arrhythmogenic right ventricular cardiomyopathy. Heart Rhythm. 2016;13:1964–1970. doi: 10.1016/j.hrthm.2016.06.021. [DOI] [PubMed] [Google Scholar]
- 5.Kristiansen H., Hovstad T., Vollan G., Faerestrand S. Right ventricular pacing and sensing function in high posterior septal and apical lead placement in cardiac resynchronization therapy. Indian Pacing Electrophysiol J. 2012;12:4–14. doi: 10.1016/s0972-6292(16)30458-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liang Y.H., Liu L., Chen D.L., Lin C.Y., Fei H.W., Chen S.L., Wu S.L. Right ventricular outflow tract septal pacing versus apical pacing: a prospective, randomized, single-blind 5-years follow-up study of ventricular lead performance and safety. J Huazhong Univ Sci Technol - Med Sci. 2015;35:858–861. doi: 10.1007/s11596-015-1518-y. [DOI] [PubMed] [Google Scholar]
- 7.Lillo-Castellano J.M., Marina-Breysse M., Gomez-Gallanti A., Martinez-Ferrer J.B., Alzueta J., Perez-Alvarez L., Alberola A., Fernandez-Lozano I., Rodriguez A., Porro R., Anguera I., Fontenla A., Gonzalez-Ferrer J.J., Canadas-Godoy V., Perez-Castellano N., Garofalo D., Salvador-Montanes O., Calvo C.J., Quintanilla J.G., Peinado R., Mora-Jimenez I., Perez-Villacastin J., Rojo-Alvarez J.L., Filgueiras-Rama D. Safety threshold of R-wave amplitudes in patients with implantable cardioverter defibrillator. Heart. 2016;102:1662–1670. doi: 10.1136/heartjnl-2016-309295. [DOI] [PubMed] [Google Scholar]
- 8.Marcus F.I., McKenna W.J., Sherrill D., Basso C., Bauce B., Bluemke D.A., Calkins H., Corrado D., Cox M.G.P.J., Daubert J.P., Fontaine G., Gear K., Hauer R., Nava A., Picard M.H., Protonotarios N., Saffitz J.E., Sanborn D.M.Y., Steinberg J.S., Tandri H., Thiene G., Towbin J.A., Tsatsopoulou A., Wichter T., Zareba W. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the task force criteria. Circulation. 2010;121:1533–1541. doi: 10.1161/CIRCULATIONAHA.108.840827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Medi C., Mond H.G. Right ventricular outflow tract septal pacing: long-term follow-up of ventricular lead performance. Pacing Clin Electrophysiol. 2009;32:172–176. doi: 10.1111/j.1540-8159.2008.02199.x. [DOI] [PubMed] [Google Scholar]
- 10.Moss A.J., Hall W.J., Cannom D.S., Klein H., Brown M.W., Daubert J.P., Estes N.A., 3rd, Foster E., Greenberg H., Higgins S.L., Pfeffer M.A., Solomon S.D., Wilber D., Zareba W., MADIT-CRT Trial Investigators Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med. 2009;361:1329–1338. doi: 10.1056/NEJMoa0906431. [DOI] [PubMed] [Google Scholar]
- 11.Mugnai G., Tomei R., Dugo C., Tomasi L., Morani G., Vassanelli C. Implantable cardioverter-defibrillators in patients with arrhythmogenic right ventricular cardiomyopathy: the course of electronic parameters, clinical features, and complications during long-term follow-up. J Intervent Card Electrophysiol. 2014;41:23–29. doi: 10.1007/s10840-014-9920-0. [DOI] [PubMed] [Google Scholar]
- 12.Priori S.G., Blomström-Lundqvist C., Mazzanti A., Blom N., Borggrefe M., Camm J., Elliott P.M., Fitzsimons D., Hatala R., Hindricks G., Kirchhof P., Kjeldsen K., Kuck K.-H., Hernandez-Madrid A., Nikolaou N., Norekvål T.M., Spaulding C., Van Veldhuisen D.J. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2015;36:2793–2867. doi: 10.1016/j.rec.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 13.La Rocca V., Bertaglia E., Solimene F., Accogli M., Iuliano A., Marrazzo N., De Luca De Masi G., Scaccia A., Donnici G., Chiariello P., Turco P., De Simone A., Ciardiello C., Stabile G. Clinical and technical determinants of long-term performance of coronary sinus leads. J Cardiovasc Med. 2010;11:40–44. doi: 10.2459/JCM.0b013e32833245df. [DOI] [PubMed] [Google Scholar]
- 14.Ruetz L.L., Koehler J.L., Brown M.L., Jackson T.E., Belk P., Swerdlow C.D. Sinus rhythm R-wave amplitude as a predictor of ventricular fibrillation undersensing in patients with implantable cardioverter-defibrillator. Heart Rhythm. 2015;12:2411–2418. doi: 10.1016/j.hrthm.2015.08.012. [DOI] [PubMed] [Google Scholar]
- 15.Steffel J., Hurlimann A., Starck C., Krasniqi N., Schmidt S., Luscher T.F., Duru F., Ruschitzka F., Holzmeister J., Hurlimann D. Long-term performance of modern coronary sinus leads in cardiac resynchronization therapy. Indian Pacing Electrophysiol J. 2014;14:112–120. doi: 10.1016/s0972-6292(16)30752-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Swerdlow C.D., Russo A.M., Degroot P.J. The dilemma of ICD implant testing. Pacing Clin Electrophysiol. 2007;30:675–700. doi: 10.1111/j.1540-8159.2007.00730.x. [DOI] [PubMed] [Google Scholar]
- 17.Wilkoff B.L., Fauchier L., Stiles M.K., Morillo C.A., Al-Khatib S.M., Almendral J., Aguinaga L., Berger R.D., Cuesta A., Daubert J.P., Dubner S., Gasparini M., Haines D.E., Healey J.S., Hurtwitz J.L., Keegan R., Kolb C., Kuck K.-H., Martinelli M., Mcguire M., Molina L.G., Okumura K., Proclemer A., Russo A.M., Wang C.-C., Zhang S. 2015 HRS/EHRA/APHRS/SOLAECE expert consensus statement on optimal implantable cardioverter-defibrillator programming and testing. Europace. 2016;18:159–183. doi: 10.1093/europace/euv411. [DOI] [PubMed] [Google Scholar]