Abstract
Background
Early natural menopause, the cessation of ovarian function before age 45 y, is positively associated with cardiovascular disease and other conditions. Dietary vitamin D intake has been inversely associated with early menopause; however, no previous studies have evaluated risk with regard to plasma 25-hydroxyvitamin D [25(OH)D] concentrations.
Objective
We prospectively evaluated associations of total and free 25(OH)D and vitamin D–binding protein (VDBP) concentrations and the risk of early menopause in a case-control study nested within the Nurses’ Health Study II (NHS2). We also considered associations of 25(OH)D and VDBP with anti-Müllerian hormone (AMH) concentrations.
Methods
The NHS2 is a prospective study in 116,430 nurses, aged 25–42 y at baseline (1989). Premenopausal plasma blood samples were collected between 1996 and 1999, from which total 25(OH)D and VDBP concentrations were measured and free 25(OH)D concentrations were calculated. Cases experienced menopause between blood collection and age 45 y (n = 328) and were matched 1:1 by age and other factors to controls who experienced menopause after age 48 y (n = 328). Conditional logistic regression models were used to estimate ORs and 95% CIs for early menopause according to each biomarker. Generalized linear models were used to estimate AMH geometric means according to each biomarker.
Results
After adjusting for smoking and other factors, total and free 25(OH)D were not associated with early menopause. Quartile 4 compared with quartile 1 ORs were 1.04 (95% CI: 0.60, 1.81) for total 25(OH)D and 0.70 (95% CI: 0.41, 1.20) for free 25(OH)D. 25(OH)D was unrelated to AMH concentrations. VDBP was positively associated with early menopause; the OR comparing the highest with the lowest quartile of VDBP was 1.80 (95% CI: 1.09, 2.98).
Conclusions
Our findings suggest that total and free 25(OH)D are not importantly related to the risk of early menopause. VDBP may be associated with increased risk, but replication is warranted.
Keywords: vitamin D, 25-hydroxyvitamin D, vitamin D–binding protein, early menopause, ovarian aging, anti-Müllerian hormone
Introduction
Early menopause, which is the cessation of ovarian function before the age of 45 y, affects 10% of women in Western populations (1). Women who experience early menopause are at increased risk of premature mortality and cognitive decline, osteoporosis, and cardiovascular disease, as well as other adverse health outcomes (2–5). Early menopause may also be problematic for couples trying to conceive, because female fertility declines drastically during the 10 y leading up to menopause (6). Couples unable to conceive as they wish may experience substantial financial and psychological consequences, particularly as women increasingly delay childbearing into the later reproductive years (1, 7). As such, it is important to identify modifiable lifestyle factors that may be related to early menopause risk, including diet.
Accelerated ovarian aging, the mechanism thought to underlie early menopause, is characterized by a decline in the quantity and quality of the ovarian follicle pool (7). During the reproductive years, primordial follicle growth and transition to primary follicles are inhibited by anti-Müllerian hormone (AMH), a glycoprotein secreted by the granulosa cells of primary, secondary, and small antral follicles. Notably, a vitamin D response element has been identified in the promoter region of the AMH gene (8), suggesting a potential role of vitamin D in AMH secretion and thus follicle recruitment. In line with this hypothesis, laboratory studies have shown that vitamin D upregulates AMH mRNA expression in both human prostate cells and granulosa cells of hens (9, 10).
Findings of a recent study in the Nurses’ Health Study (NHS) II (NHS2) cohort suggest that vitamin D intake from food sources, particularly dairy sources, is associated with a lower risk of early menopause (11). Because vitamin D is obtained through dietary intake as well as sunlight exposure, concentrations of 25-hydroxyvitamin D [25(OH)D], the primary circulating metabolite of vitamin D, is a better indicator of vitamin D status than dietary intake alone. To our knowledge, no previous studies have evaluated 25(OH)D concentrations and the risk of early menopause and findings of epidemiologic studies evaluating 25(OH)D concentrations and AMH are conflicting (12–15). One study found that AMH concentrations exhibit seasonal variation correlated with 25(OH)D concentrations and that vitamin D supplementation prevented a seasonal decline in AMH concentrations (13). An additional study in US women found 25(OH)D to be positively associated with AMH concentrations (12), whereas 3 more-recent studies reported no association (14, 15).
It is also currently unclear whether measures of total compared with free 25(OH)D concentrations may have different relations with ovarian aging. At any given time, >99% of vitamin D in the body is bound to vitamin D–binding protein (VDBP) or albumin, leaving <1% unbound (16). Because VDBP concentrations influence the bioavailability of vitamin D, assessing total compared with free 25(OH)D concentrations as well as concentrations of VDBP may be important for understanding the potential relation of vitamin D and early menopause. In fact, recent epidemiologic studies have observed that the free 25(OH)D fraction, as compared with total 25(OH)D, is more strongly associated with several health outcomes such as bone mineral density (17) and colorectal cancer (18).
In light of the lack of studies evaluating vitamin D status and early menopause, the aims of the present nested case-control study were thus to evaluate associations of total 25(OH)D, free 25(OH)D, and VDBP concentrations and the risk of early menopause among participants of the NHS2.
Methods
The NHS2 is a prospective study in 116,429 female US registered nurses who were 25–42 y old in 1989 when they responded to a mailed baseline questionnaire. Information with regard to lifestyle behaviors and medical conditions are collected through biennial questionnaires, for which the follow-up rate for each cycle has been ≥89%. The study protocol was approved by the institutional review board at Brigham and Women's Hospital in Boston, Massachusetts.
Case and control ascertainment
On the 1989 baseline questionnaire, nurses were asked if their menstrual periods had ceased permanently (i.e., no periods for ≥12 mo) and were provided the following response options: 1) no (premenopausal), 2) yes (no menstrual periods), 3) yes (had menopause but now have periods induced by hormones), and 4) not sure (e.g., started hormones before cessation of periods). Nurses who indicated that their periods had ceased were then asked the following questions: 1) At what age did your periods cease (open response) and 2) for what reason did your periods cease (response options were surgery, radiation or chemotherapy, and natural)? Women were also asked about their current and past use of menopausal hormone therapy (HT). Questions with regard to menopausal status and use of HT were then repeated on all subsequent questionnaires.
A small number of women reported being postmenopausal on one questionnaire after a long interval of amenorrhea (>12 mo), only to have periods return again and report being premenopausal on a subsequent questionnaire. For these women, we defined age at menopause as the age after which periods were absent for ≥12 mo, and then confirmed that this status persisted for ≥3 consecutive questionnaires in order to reduce potential for misclassification of early menopause.
Blood sample collection
Participants of the NHS2 who had not previously been diagnosed with cancer (n = 92,888) were invited to provide blood samples between 1996 and 1999. During this time period, participants were aged 32–54 y. Premenopausal women who were not pregnant and were not using oral contraceptives (OCs) or menopausal HT were asked to provide 2 samples during the span of one menstrual cycle. The first sample was to be collected during the follicular phase (days 3–5 of the menstrual cycle) and the second was to be collected during the luteal phase (7–9 d before the start of the next menses). Women who were using OCs or menopausal HT were asked to provide a single untimed sample, and women who reported irregular menstrual cycles were asked to collect a luteal phase sample 22 d after the last menses. To confirm timing and menstrual cycle phase of the samples, women completed a postcard at the start of their next menses. Upon receipt, blood samples were centrifuged, separated into plasma, buffy, and RBC components, and then stored at ≤ –130°C in nitrogen freezers.
Among eligible women, 29,611 provided a sample and ∼23,000 of these women were premenopausal at the time of blood draw. Women who provided samples were similar to the entire NHS2 cohort, having equivalent BMI (in kg/m2; 26 and 26) and parity (1.9 and 1.9 children) and comparable proportions of ever smoking (34% compared with 36%) and history of OC use (86% compared with 88%), as well as other factors (19).
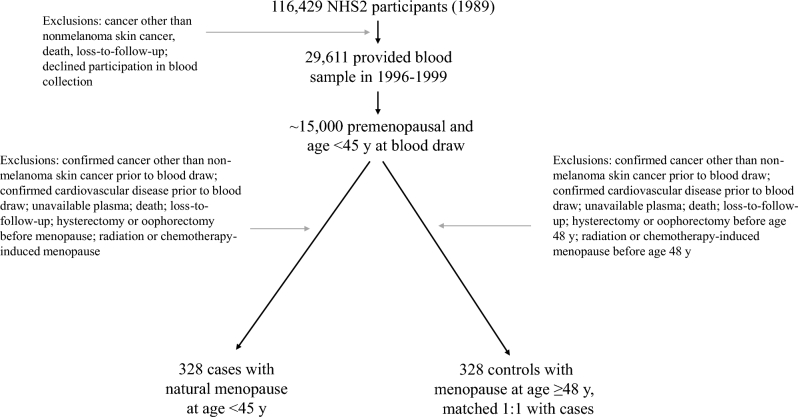
Because we were interested in prospectively evaluating the relation of plasma 25(OH)D concentrations and risk of early menopause, we limited eligibility to women who experienced menopause after blood draw. In addition, our study sample was limited to women without a previous diagnosis of cancer (other than nonmelanoma skin cancer), myocardial infarction, stroke, coronary artery bypass surgery, or percutaneous coronary intervention and women who had available plasma samples. Cases of early menopause were then defined as women reporting natural menopause (i.e., not due to surgery or chemotherapy) before the age of 45 y during follow-up. To reduce the potential for misclassification of early menopause status, eligible controls were women who experienced menopause after age 48 y (quartile 3 of the IQR). Eligible cases (n = 328) were then matched 1:1 to controls according to age at blood collection (within 4 mo), time of day of blood collection, month of collection (within 3 mo), sample type (luteal phase or untimed), and fasting status (Figure 1).
FIGURE 1.
Participant flowchart. NHS2, Nurses’ Health Study II.
Laboratory assays
Plasma 25(OH)D, VDBP, albumin, and AMH were measured in the laboratory of Nader Rifai at Boston's Children's Hospital. Immunoassays were used for the measurement of plasma 25(OH)D (Immunodiagnostic Systems, Inc.) and plasma VDBP (R&D Systems). The immunoassay used to measure plasma VDBP was a monoclonal antibody. Plasma AMH was measured by using the pico AMH assay from ANSH Labs.
With the use of measured values of plasma total 25(OH)D, VDBP, and albumin for each individual, we calculated each participant's plasma free 25(OH)D concentration according to the following equation (20):
 |
(1) |
To prevent potential exposure measurement error related to case status, laboratory personnel were blinded to case/control status for all assays. Samples were labeled by number; and matched case-control sets were handled together, shipped in the same batch, and assayed in the same analytical run. Masked quality-control samples were randomly interspersed among case-control samples and were analyzed in each batch. The CVs for biomarkers were 4.9% for total 25(OH)D, 7.2% for VDBP, and 8.6% for AMH.
Assessment of covariates
Variables used as matching factors, including age at blood collection, time of day of blood collection, month of collection, sample type (luteal phase or untimed), and fasting status, were assessed at the time of blood draw. We also considered the following variables for inclusion in multivariable models on the basis of previous studies of risk factors for early menopause: race/ethnicity, smoking, BMI, parity, physical activity, OC use and duration, duration of breastfeeding, alcohol intake, and vegetable protein intake. Height and race/ethnicity were assessed at baseline in 1989 and information with regard to smoking status, weight (to calculate BMI), alcohol intake, fasting status, and menstrual cycle phase were collected at the time of blood draw by separate questionnaire. Updated information with regard to smoking, weight, OC use, parity, and breastfeeding was assessed on biennial questionnaires beginning in 1989. Physical activity was assessed in 1989, 1991, 1997, 2001, and 2005 by using validated questionnaires (21). Finally, validated FFQs administered every 4 y were used to assess dietary intake of total calories, alcohol, and vegetable protein (22–24). Intakes of nutrients were adjusted for total energy using the residual method (25). For time-varying covariates, we modeled variables that corresponded to questionnaires closest in time to blood collection for each individual. Women with missing covariate data were assigned to a missing indicator.
Statistical analysis
We used chi-square and t tests to compare characteristics of early menopause cases and controls at the time of blood draw and histogram and normality plots to assess the normality of biomarker data. Participants were divided into quartiles of total 25(OH)D, free 25(OH)D, and VDBP on the basis of the distribution of these biomarkers in the control group. Likelihood ratio tests comparing nested models were used to assess the global significance of each biomarker. We also categorized participants according to the following cutoffs: <50, 50 to <75, and ≥75 nmol/L (26). For analyses using continuous biomarker data, we identified and removed outliers by using the Rosner extreme studentized deviate test and standardized continuous variables to aid in interpretability. Relations of total and free 25(OH)D with log(AMH) were evaluated by using multivariable generalized linear models. We then back-calculated AMH geometric means and 95% CIs according to each quartile of total and free 25(OH)D.
For each exposure, we used conditional logistic regression to calculate ORs and 95% CIs, adjusting for matching factors only. We then controlled for potential confounders of the 25(OH)D–early menopause relation using multivariable conditional logistic regression. Because none of the covariates that were tested produced a change in exposure estimates >10%, covariates in multivariable models were selected if they were importantly associated with the outcome in our population.
To assess potential nonlinear associations of each biomarker exposure with the risk of early menopause, we additionally conducted restricted cubic spline models. For these models, we specified 4 knots in order to evaluate the individual spline term contributions to the model fit and overall test for nonlinearity.
We additionally used restricted cubic spline models to assess potential nonlinear associations of each biomarker with the risk of early menopause. To assess potential variation in associations according to age, we stratified our analyses by median age at blood draw (40 y). Second, because vitamin D is sequestered in adipose tissue (27), we assessed potential BMI effect modification by stratifying according to 2 categories of BMI (<25 and ≥25). Third, to evaluate potential seasonal variation in the 25(OH)D–early menopause relation, we stratified our analyses by season of blood collection (winter/spring and summer/fall).
We also conducted sensitivity analyses to determine the robustness of estimates in primary analyses. Autoimmune conditions such as multiple sclerosis, rheumatoid arthritis, and lupus may be associated with lower 25(OH)D concentrations and earlier age at menopause. Accordingly, we conducted analyses excluding individuals diagnosed with these conditions at any point during follow-up. Similarly, we conducted analyses limited to women with luteal phase samples in order to address the potential impact of variation in biomarker measures due to menstrual cycle variability. We also conducted analyses limited to never smokers to evaluate potential residual confounding due to misclassification of smoking amount among smokers. All of the analyses were conducted with the use of SAS version 9.4 software (SAS Institute, Inc.). We used 2-sided statistical tests performed at the 0.05 significance level for all analyses.
Results
Characteristics of cases and controls at the time of blood collection are presented in Table 1. At the time of blood collection, cases were more likely to identify as nonwhite and to smoke and reported lower intakes of alcohol, supplemental calcium, and total vitamin D and calcium than controls.
TABLE 1.
Characteristics of early menopause cases and controls at blood draw (1996–1999): NHS21
| Characteristic | Cases (n = 328) | Controls (n = 328) | P 2 |
|---|---|---|---|
| Age,3 y | 40.2 ± 2.8 | 40.2 ± 2.8 | 0.99 |
| BMI, kg/m2 | 25.4 ± 0.3 | 25.0 ± 0.3 | 0.10 |
| Age at menarche, y | 12.4 ± 0.1 | 12.3 ± 0.1 | 0.57 |
| Physical activity, MET-h/wk | 81.5 ± 14.7 | 72.5 ± 12.9 | 0.12 |
| Parity, n | 1.8 ± 0.1 | 1.9 ± 0.1 | 0.55 |
| Duration of breastfeeding, mo | 4.8 ± 0.2 | 5.0 ± 0.2 | 0.25 |
| Alcohol intake, g/d | 3.3 ± 0.3 | 3.9 ± 0.4 | <0.01 |
| Vegetable protein intake, % total kcal/d | 5.2 ± 0.01 | 5.5 ± 0.01 | 0.09 |
| Total vitamin D intake, IU/d | 348 ± 13.3 | 381 ± 15.1 | 0.03 |
| Supplemental vitamin D intake | 125 ± 10.9 | 156 ± 11.7 | 0.19 |
| Dietary vitamin D intake | 218 ± 6.5 | 217 ± 6.4 | 0.81 |
| Dairy vitamin D intake | 122 ± 6.2 | 118 ± 5.7 | 0.15 |
| Total calcium intake, mg/d | 990 ± 24.5 | 1062 ± 27.6 | 0.03 |
| Supplemental calcium intake | 146 ± 15.7 | 186 ± 19.7 | <0.001 |
| Dietary calcium intake | 841 ± 18.3 | 865 ± 17.1 | 0.29 |
| Dairy calcium intake | 537 ± 18.6 | 542 ± 17.3 | 0.27 |
| Non-Hispanic white, % | 95.3 | 98.8 | 0.01 |
| Season of blood draw, % | 0.64 | ||
| Summer/fall | 50.0 | 50.0 | |
| Winter/spring | 51.8 | 48.2 | |
| Current smoker, % | 14.0 | 9.2 | 0.05 |
| Smoking duration,4 pack-years | 12.9 ± 1.0 | 10.5 ± 0.7 | 0.02 |
| Current OC user, % | 1.8 | 3.7 | 0.15 |
| OC use duration,5 mo | 59.6 ± 3.2 | 63.3 ± 3.5 | 0.11 |
1Values are means ± SEs unless otherwise indicated. MET, metabolic equivalent task; NHS2, Nurses’ Health Study II; OC, oral contraceptive.
2 P values correspond to t tests for continuous variables and chi-square tests for categorical variables.
3Values are means ± SDs.
4Among ever smokers only.
5Among ever OC users only.
In unadjusted analyses and analyses adjusting for important covariates, total 25(OH)D concentrations were not associated with risk (Table 2). For example, the OR comparing the first and fourth quartile of total 25(OH)D was 1.04 (95% CI: 0.60, 1.81) in our multivariable (MV) 1 model. Results for free 25(OH)D were suggestive of a lower risk at the highest concentrations, but CIs were wide and results were not significant (quartile 4 compared with quartile 1—OR: 0.70; 95% CI: 0.41, 1.20). High VDBP was associated with increased risk, and results were stronger after adjustment for variables in MV1, with some evidence of a threshold of higher risk for quartiles 3 and 4 compared with quartile 1 (quartile 3 compared with quartile 1—OR: 1.80; 95% CI: 1.10, 2.95; quartile 4 compared with quartile 1—OR: 1.80; 95% CI: 1.09, 2.98). Results comparing women with total 25(OH)D concentrations ≥75 nmol/L compared with those with concentrations <50 nmol/L were null (MV2—OR: 1.19; 95% CI: 0.57, 2.46) (Supplemental Table 1). For each exposure, results from restricted cubic spline models indicated no associations with early menopause—either linear or nonlinear (P for all exposures >0.10; complete results not shown).
TABLE 2.
Unadjusted and multivariable ORs (95% CIs) for early menopause according to quartile of total and free 25(OH)D and VDBP concentrations: NHS2 (1996–2011)1
| Quartile | ||||||
|---|---|---|---|---|---|---|
| Biomarker | n/Q1 = 169 | n/Q2 = 175 | n/Q3 = 153 | n/Q4 = 159 | P 2 | Continuous3 |
| Total 25(OH)D | ||||||
| Median, nmol/L | 44.2 | 59.8 | 71.7 | 90.4 | ||
| Cases:controls, n:n | 86:83 | 92:83 | 71:82 | 79:80 | ||
| Unadjusted OR4 (95% CI) | 1 | 1.07 (0.70, 1.64) | 0.81 (0.51, 1.29) | 0.91 (0.56, 1.48) | 0.69 | 1.02 (0.83, 1.25) |
| MV1 | 1 | 1.21 (0.75, 1.93) | 0.93 (0.56, 1.54) | 1.04 (0.60, 1.81) | 0.75 | 1.12 (0.89, 1.42) |
| MV2 | 1 | 1.18 (0.73, 1.89) | 0.88 (0.53, 1.46) | 0.96 (0.55, 1.69) | 0.70 | 1.09 (0.85, 1.38) |
| Free 25(OH)D | ||||||
| Median, pmol/L | 13.9 | 18.7 | 24.2 | 32.4 | ||
| Cases:controls, n:n | 94:83 | 91:85 | 81:80 | 62:80 | ||
| Unadjusted OR4 (95% CI) | 1 | 0.92 (0.60, 1.40) | 0.87 (0.56, 1.34) | 0.65 (0.41, 1.05) | 0.32 | 0.82 (0.67, 0.99) |
| MV1 | 1 | 1.02 (0.64, 1.62) | 0.98 (0.61, 1.57) | 0.70 (0.41, 1.20) | 0.44 | 0.85 (0.68, 1.05) |
| VDBP | ||||||
| Median, µg/mL | 145.4 | 203.0 | 248.2 | 305.3 | ||
| Cases:controls, n:n | 56:81 | 87:81 | 86:83 | 99:83 | ||
| Unadjusted OR4 (95% CI) | 1 | 1.54 (0.99, 2.41) | 1.49 (0.96, 2.34) | 1.75 (1.11, 2.77) | 0.10 | 1.16 (0.99, 1.36) |
| MV1 | 1 | 1.54 (0.94, 2.51) | 1.80 (1.10, 2.95) | 1.80 (1.09, 2.98) | 0.07 | 1.17 (0.99, 1.39) |
| MV2 | 1 | 1.50 (0.92, 2.46) | 1.77 (1.08, 2.90) | 1.77 (1.06, 2.94) | 0.09 | 1.16 (0.98, 1.38) |
1MV1 adjusted for matching factors plus physical activity (<9, 9 to <42, or ≥42 MET-h/wk), duration of breastfeeding at blood draw (0, >0–6, >6–18, or >18 mo), smoking status at blood draw (nonsmoker or current smoker), BMI (kg/m2; <25 or ≥25), and intakes of alcohol (0, 0.1–9.9, 10.0–29.9, or ≥30.0 g/d) and percentage of vegetable protein (continuous) at blood draw. MV2 adjusted for covariates in MV1 plus mutual adjustment for total 25(OH)D and VDBP. MET, metabolic equivalent task; MV, multivariable; NHS2, Nurses’ Health Study II; Q, quartile; VDBP, vitamin D–binding protein; 25(OH)D, 25-hydroxyvitamin D.
2 P values correspond to likelihood ratio tests comparing models with and without indicator variables for exposures.
3ORs correspond to a 1-SD increase in exposure.
4Model adjusted for matching factors only [i.e., age at blood collection (within 4 mo), time of day of blood collection, month of collection (within 3 mo), sample type (luteal phase or untimed), and fasting status].
We did not find that adjusted geometric means of AMH concentrations varied according to quartile of total (P = 0.55) or free 25(OH)D (P = 0.32) concentrations (Table 3). AMH geometric mean concentrations varied significantly across VDBP quartiles, with the lowest concentrations in quartiles 3 and 4 (P = 0.04).
TABLE 3.
Unadjusted and adjusted AMH geometric means (95% CIs) according to quartile of total and free 25(OH)D and VDBP: NHS2 (1996–2011)1
| AMH geometric mean (95% CI), ng/mL | ||||
|---|---|---|---|---|
| n | Range | Unadjusted | Adjusted2 | |
| Total 25(OH)D, nmol/L | ||||
| Q1 | 169 | 22.9–52.1 | 0.4 (0.3, 0.6) | 0.5 (0.4, 5.8) |
| Q2 | 175 | 52.2–65.8 | 0.4 (0.3, 0.5) | 0.4 (0.3, 4.6) |
| Q3 | 153 | 65.9–78.8 | 0.4 (0.3, 0.5) | 0.4 (0.3, 5.4) |
| Q4 | 159 | 78.8–159.0 | 0.4 (0.3, 0.5) | 0.4 (0.3, 4.8) |
| P 3 | 0.65 | 0.55 | ||
| Free 25(OH)D, pmol/L | ||||
| Q1 | 177 | 6.8–16.2 | 0.4 (0.3, 0.5) | 0.4 (0.3, 0.5) |
| Q2 | 176 | 16.3–21.4 | 0.4 (0.3, 0.5) | 0.4 (0.3, 0.5) |
| Q3 | 161 | 21.6–27.5 | 0.4 (0.3, 0.5) | 0.4 (0.3, 0.5) |
| Q4 | 142 | 27.5–88.9 | 0.5 (0.4, 0.6) | 0.5 (0.4, 0.6) |
| P 3 | 0.35 | 0.32 | ||
| VDBP, µg/mL | ||||
| Q1 | 137 | 48.7–177.7 | 0.5 (0.4, 0.7) | 0.5 (0.4, 0.7) |
| Q2 | 168 | 178.2–224.3 | 0.4 (0.3, 0.5) | 0.4 (0.3, 0.6) |
| Q3 | 169 | 224.8–272.0 | 0.4 (0.3, 0.5) | 0.4 (0.3, 0.5) |
| Q4 | 182 | 272.6–425.8 | 0.3 (0.3, 0.4) | 0.3 (0.3, 0.4) |
| P 3 | 0.07 | 0.04 | ||
1AMH, anti-Müllerian hormone; MET, metabolic equivalent task; NHS2, Nurses’ Health Study II; Q, quartile; VDBP, vitamin D–binding protein; 25(OH)D, 25-hydroxyvitamin D.
2Multivariable model 1 adjusted for matching factors plus physical activity (<9, 9 to <42, or ≥42 MET-h/wk), duration of breastfeeding at blood draw (0, >0–6, >6–18, or >18 mo), smoking status at blood draw (nonsmoker vs. current smoker), BMI (kg/m2; <25 or ≥25), and intakes of alcohol (0, 0.1–9.9, 10.0–29.9, or ≥30.0 g/d) and percentage of vegetable protein (continuous) at blood draw.
3 P values corresponds to type III P values.
Findings of the primary analyses were largely unchanged in models evaluating effect modification and in sensitivity analyses. Estimates from analyses of total 25(OH)D stratified by median age at blood draw (<40 or ≥40 y) were consistent with findings from main analyses (data not shown). Estimates for total 25(OH)D stratified by BMI (underweight/normal and overweight/obese) were unstable due to small numbers (data not shown). Season (summer/fall compared with winter/spring) did not modify associations for total 25(OH)D or free 25(OH)D (P-interaction = 0.31 and 0.85, respectively). Estimates were also similar after restricting analyses to nonsmokers (n = 222 cases and 216 controls), women who provided timed blood samples (n = 254 cases and 254 controls), and women without diagnoses of autoimmune conditions (n = 321 cases and 319 controls). We also conducted models using only “super-normal controls,” defined as women who experienced menopause at the population mean age of 51 y (n = 328 cases and 63 controls). Estimates from these models were not materially different than those of our main analyses. For example, the MV1 OR for each 1-SD increase in total 25(OH)D was 1.29 (95% CI: 0.87, 1.91) and for free 25(OH)D was 1.00 (95% CI: 0.68, 1.46).
Discussion
In this prospective study, we did not find vitamin D metabolite concentrations to be consistently or strongly associated with the risk of early menopause, or with concentrations of plasma AMH, a marker of ovarian aging. Conversely, high compared with low VDBP concentrations were associated with increased risk of early menopause and lower AMH concentrations.
To our knowledge, this is the first study to investigate the association of total and free 25(OH)D and VDBP concentrations and risk of early menopause. A recent study in NHS2 participants by our research group observed a significant 17% lower risk of early menopause among women who consumed the highest vitamin D from food sources (quintile 5 median = 528 IU/d), as compared with those who consumed the least (quintile 1 median = 148 IU/d) (11). In contrast, we observed that total vitamin D intake was not associated with risk, whereas supplemental vitamin D intake (≥600 compared with 0 IU/d) was associated with increased risk. The lack of strong and consistent associations of 25(OH)D with risk in the present analysis suggests that other dietary components or lifestyle factors correlated with vitamin D in foods may be associated with early menopause, rather than vitamin D itself. In our previous study, we noted a stronger association for dairy sources of vitamin D compared with nondairy dietary vitamin D. Other constituents of dairy, such as lactose, calcium, or progesterone, which are highly correlated with vitamin D in dairy, may have influenced the observed inverse association for dietary vitamin D. The potential importance of dairy foods generally, rather than vitamin D specifically, is also supported by findings of Carwile et al. (28), who observed that low-fat dairy intake was associated with later age at menopause, among women aged <51 y. To answer this question, future studies of dairy foods and dairy constituents and risk of early menopause are warranted.
Vitamin D has been hypothesized to be related to ovarian aging and menopause timing primarily through effects on AMH and follicular development. Our null findings for 25(OH)D and AMH are consistent with those of 3 cross-sectional studies (14, 15, 29). Pearce et al. (14) observed no association of 25(OH)D and AMH among 340 infertility treatment-seeking women <40 y of age living in South Australia. Similarly, Drakopoulos et al. (15) also observed no association of 25(OH)D concentrations and AMH among an infertile population of 283 Belgian women <42 y of age, and Kim et al. (29) observed no association of 25(OH)D and AMH concentrations in 291 Korean women 35–49 y of age.
In contrast, 2 other studies reported significant associations between 25(OH)D and AMH. In a clinical trial in 33 premenopausal women living in New Zealand, Dennis et al. (13) observed seasonal variation of AMH concentrations mirroring that of 25(OH)D in both direction and magnitude, as well as a stabilizing effect of cholecalciferol supplementation on the seasonality of AMH concentrations. This is in contrast to placebo and ergocalciferol supplementation groups, who experienced a significant decline in AMH concentrations from summer to winter months. In another small trial, Naderi et al. (30) reported a significant increase in both 25(OH)D and AMH concentrations after 3 mo of vitamin D supplementation among 30 infertile Iranian women with vitamin D insufficiency or deficiency. Likewise, in a cross-sectional study in US women, Merhi et al. (12) observed a significant 1.1% higher log-transformed AMH for each 1-ng/mL higher 25(OH)D concentration among 388 women aged ≥40 y, but no association among younger women.
The inconsistency in findings between studies of 25(OH)D and AMH may potentially be explained by 2 important factors. First, it is possible that 25(OH)D may only be associated with changes in AMH concentrations among vitamin D–deficient women. Both the Kim et al. trial (29) and the Merhi et al. (12) study were conducted in predominantly vitamin D–deficient women. The Dennis et al. trial (13) did not provide baseline 25(OH)D concentrations of participants, but given the high latitude of residence of the study population, these women may have been vitamin D deficient before receiving the supplement or placebo, and thus supplementation may have been beneficial in preventing a seasonal decline in 25(OH)D and AMH concentrations. If a 25(OH)D–AMH association is only observable at the low end of the vitamin D spectrum, then one would expect to see an association only in populations with a high prevalence of vitamin D deficiency, which the NHS2 is not. Given the small numbers of women with 25(OH)D concentrations <35 nmol/L (n = 28) in the NHS2, we were unable to address this possibility in our analyses.
It is also possible that the 25(OH)D–AMH relation may vary across racial groups due to differences in VDBP, because VDBP concentrations and binding affinity are determined almost entirely by genetic polymorphisms specific to race and ethnicity (31). The binding affinity and concentration of VDBP are directly related to the bioavailability of 25(OH)D within the body, and thus may be biologically relevant to the mechanisms involved in ovarian aging. In our study, we observed a threshold of higher risk for early menopause among individuals in quartiles 2 through 4 of VDBP compared with quartile 1, and a nonlinear relation of VDBP with AMH concentrations, suggesting that VDBP concentrations and/or their genetic determinants may be associated with risk. The NHS2, as well as the study populations of Pearce et al. (14) and Drakopoulos et al. (15) were almost entirely white, whereas that of Merhi et al. (12) was predominantly black and Hispanic. If 25(OH)D is only associated with AMH in specific racial/ethnic groups, demographic differences in study populations, and thus the underlying population heterogeneity of VDBP polymorphisms, may provide some explanation for inconsistent findings.
Our ability to evaluate associations of early menopause and AMH with free 25(OH)D and VDBP in nonwhite individuals was limited for 2 reasons. First, given the racial homogeneity of the NHS2, we used a monoclonal rather than a polyclonal assay to measure VDBP, which has been shown to underestimate free 25(OH)D concentrations in nonwhite individuals (32). Second, the NHS2 is composed mostly of white women, and thus numbers of nonwhite individuals were too small to conduct race-stratified analyses. In light of evidence that the 25(OH)D–AMH relation may vary by race potentially due to VDBP differences, additional evaluation of 25(OH)D, VDBP, and AMH in large, diverse populations is necessary.
It is important to note additional limitations of our study. First, we relied upon single measurements of 25(OH)D, VDBP, and AMH. Because 25(OH)D concentrations are influenced heavily by recent sun exposure and dietary intake, it is possible that within-person variability in 25(OH)D concentrations may have contributed measurement error, resulting in nondifferential misclassification. However, in the NHS, a similar population of female health professionals, the intraclass correlation coefficient for plasma 25(OH)D measured 2–3 y apart was 0.72 (P < 0.001) (33), and for measurements 10 y apart was 0.51 (95% CI: 0.42, 0.60) (34). In a comparable population, the intraclass correlation coefficient for VDBP measured over 1–3 y was 0.96 (P < 0.001) (35), indicating that these biomarkers are relatively stable over time. Furthermore, although some researchers have raised questions about potential misclassification of vitamin D deficiency status when 25(OH)D concentrations are measured by immunoassay, this technique is widely used and well validated for studies comparing disease risk across relative 25(OH)D concentrations, such as our study (36). These factors help minimize the possibility that measurement error in biomarker data would explain our null findings for total and free 25(OH)D.
Second, we relied upon self-report of age at menopause, defined as the age at which periods were absent for 12 mo, to ascertain cases and controls. Because women may experience amenorrhea and then have periods return again during perimenopause, some degree of measurement error in self-reported age at menopause is to be expected. Such measurement error would result in misclassification of early menopause cases and controls and produce a bias toward the null. However, among 6591 women in the comparable NHS population, 82% of women reported the same age at menopause over multiple questionnaire cycles, suggesting high reproducibility (37). Furthermore, we restricted the control group to women with age at menopause >48 y to reduce potential misclassification of the outcome. As such, misclassification of cases and controls would be an unlikely explanation for our findings.
Third, it is possible that residual confounding may have influenced our estimates. However, we were able to control for previously identified risk factors for early menopause such as smoking, vegetable protein intake, and BMI. None of the covariates included in multivariable models produced a change >10% in exposure estimates and unadjusted estimates were similar to fully adjusted estimates, suggesting that substantial bias due to residual confounding is unlikely.
There are also several notable strengths of our study, which extend on design limitations and findings of previous research in this area. First, this was the first study, to our knowledge, to evaluate the association of 25(OH)D and early menopause. Not only did we assess total 25(OH)D, we also evaluated VDBP and free 25(OH)D, which represents the biologically active vitamin D fraction and has been more strongly related to the risk of some health outcomes than total 25(OH)D alone. Second, the size of our study population was larger than previous studies evaluating 25(OH)D and AMH, which provided greater statistical power and also allowed us to consider a wide variety of potential confounders. Finally, because most previous studies of 25(OH)D and AMH have been conducted in women seeking treatment for infertility, we anticipate that our findings are more widely generalizable to healthy premenopausal women. In the context of our findings and those of the aforementioned studies, it appears that 25(OH)D is unlikely to be related to AMH or early menopause, at least among white premenopausal women without vitamin D deficiency.
In conclusion, the findings of our study do not suggest that 25(OH)D concentrations are importantly related to the risk of early menopause. Modest positive associations of VDBP concentrations with the risk of early menopause and AMH concentrations warrant further evaluation in large, ethnically diverse populations.
Supplementary Material
Acknowledgments
The authors’ responsibilities were as follows—JEM, SEH, BAR, and ERB-J: designed the research; ACP-S, BWW, and ERB-J: conducted the research and performed statistical analysis; JEM, SEH, and BAR: provided essential materials; ACP-S, BWW, JEM, and ERB-J: wrote the manuscript; LMT: interpreted study results, reviewed the manuscript for important intellectual content, and contributed knowledge of underlying biological mechanisms; ACP-S, JEM, and ERB-J: had primary responsibility for final content; and all authors: read and approved the final manuscript.
Notes
Supported by UM1CA176726 and R01HD078517 from the NIH, US Department of Health and Human Services.
Author disclosures: ACP-S, BWW, JEM, SEH, LMT, BAR, and ERB-J, no conflicts of interest.
Supplemental Table 1 is available from the “Supplementary data” link in the online posting of the article and from the same link in the online table of contents at https://academic.oup.com/jn/.
Abbreviations used:
- AMH
anti-Müllerian hormone
- HT
hormone therapy
- MV
multivariable
- NHS
Nurses’ Health Study
- OC
oral contraceptive
- VDBP
vitamin D–binding protein
- 25(OH)D
25-hydroxyvitamin D
References
- 1. Shuster LT, Rhodes DJ, Gostout BS, Grossardt BR, Rocca WA. Premature menopause or early menopause: long-term health consequences. Maturitas 2010;65:161–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wellons M, Ouyang P, Schreiner PJ, Herrington DM, Vaidya D. Early menopause predicts future coronary heart disease and stroke: the Multi-Ethnic Study of Atherosclerosis. Menopause 2012;19:1081–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. van Der Voort D, van Der Weijer P, Barentsen R. Early menopause: increased fracture risk at older age. Osteoporos Int 2003;14:525–30. [DOI] [PubMed] [Google Scholar]
- 4. Fauser B. Trilogy 8: Premature ovarian failure and perimenopause: female health implications of premature ovarian insufficiency. Proceedings of the International Federation of Fertility Societies 21st World Congress on Fertility and Sterilty and the 69th Annual Meeting of the American Society for Reproductive Medicine; Boston, MA; 2013. p. 137–8. [Google Scholar]
- 5. Bleil ME, Adler NE, Gregorich SE, Sternfeld B, Rosen MP, Cedars MI. Does accelerated reproductive aging underlie premenopausal risk for cardiovascular disease? Fertil Steril 2012;98:S43–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lobo RA. Potential options for preservation of fertility in women. N Engl J Med 2005;353:64–73. [DOI] [PubMed] [Google Scholar]
- 7. Broekmans F, Soules M, Fauser B. Ovarian aging: mechanisms and clinical consequences. Endocr Rev 2009;30:465–93. [DOI] [PubMed] [Google Scholar]
- 8. Malloy PJ, Peng L, Wang J, Feldman D. Interaction of the vitamin D receptor with a vitamin D response element in the Mullerian-inhibiting substance (MIS) promoter: regulation of MIS expression by calcitriol in prostate cancer cells. Endocrinology 2008;150:1580–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Krishnan AV, Moreno J, Nonn L, Malloy P, Swami S, Peng L, Peehl DM, Feldman D. Novel pathways that contribute to the anti-proliferative and chemopreventive activities of calcitriol in prostate cancer. J Steroid Biochem Mol Biol 2007;103:694–702. [DOI] [PubMed] [Google Scholar]
- 10. Wojtusik J, Johnson P. Vitamin D regulates anti-Mullerian hormone expression in granulosa cells of the hen. Biol Reprod 2012;86:91. [DOI] [PubMed] [Google Scholar]
- 11. Purdue-Smithe AC, Whitcomb BW, Szegda KL, Boutot ME, Manson JE, Hankinson SE, Rosner BA, Troy LM, Michels KB, Bert. one-Johnson ER. Vitamin D and calcium intake and risk of early menopause. Am J Clin Nutr 2017;105:1493–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Merhi ZO, Seifer DB, Weedon J, Adeyemi O, Holman S, Anastos K, Golub ET, Young M, Karim R, Greenblatt R et al. Circulating vitamin D correlates with serum antimüllerian hormone levels in late-reproductive-aged women: Women's Interagency HIV Study. Fertil Steril 2012;98:228–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Dennis NA, Houghton LA, Jones GT, van Rij AM, Morgan K, McLennan IS. The level of serum anti-Müllerian hormone correlates with vitamin D status in men and women but not in boys. J Clin Endocrinol Metab 2012;97:2450–5. [DOI] [PubMed] [Google Scholar]
- 14. Pearce K, Gleeson K, Tremellen K. Serum anti-Mullerian hormone production is not correlated with seasonal fluctuations of vitamin D status in ovulatory or PCOS women. Hum Reprod 2015;30:2171–7. [DOI] [PubMed] [Google Scholar]
- 15. Drakopoulos P, Van A de V, Schutyser V, Milatovic S, Anckaert E, Schiettecatte J, Blockeel C, Camus M, Tournaye H, Polyzos NP. The effect of serum vitamin D levels on ovarian reserve markers: a prospective cross-sectional study. Hum Reprod 2017;32:208–14. [DOI] [PubMed] [Google Scholar]
- 16. Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol 2014;21:319–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Li C, Chen P, Duan X, Wang J, Shu B, Li X, Ba Q, Li J, Wang Y, Wang H. Bioavailable 25(OH)D but not total 25(OH)D is an independent determinant for bone mineral density in Chinese postmenopausal women. EBioMedicine 2016;15:184–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ying H-Q, Sun H-L, He B-S, Pan Y-Q, Wang F, Deng Q-W, Chen J, Liu X, Wang S-K. Circulating vitamin D binding protein, total, free and bioavailable 25-hydroxyvitamin D and risk of colorectal cancer. Sci Rep 2015;5:7956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Tworoger S, Sluss P, Hankinson SE. Association bewteen plasma prolactin levels and breast cancer among predominantly premenopausal women. Cancer Res 2006;66:2476–82. [DOI] [PubMed] [Google Scholar]
- 20. Powe CE, Ricciardi C, Berg AH, Erdenesanaa D, Collerone G, Ankers E, Wenger J, Karumanchi SA, Thadhani R, Bhan I. Vitamin D–binding protein modifies the vitamin D–bone mineral density relationship. J Bone Miner Res 2011;26:1609–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Wolf AM, Hunter DJ, Colditz GA, Manson JE, Stampfer MJ, Corsano KA, Rosner B, Kriska A, Willett WC. Reproducibility and validity of a self-administered physical activity questionnaire. Int J Epidemiol 1994;23:991–9. [DOI] [PubMed] [Google Scholar]
- 22. Willett WC, Sampson L, Stampfer MJ, Rosner B, Bain C, Witschi J, Hennekens CH, Speizer FE. Reproducibility and validity of a semiquantitative food frequency questionnaire. Am J Epidemiol 1985;122:51–65. [DOI] [PubMed] [Google Scholar]
- 23. Salvini S, Hunter DJ, Sampson L, Stampfer MJ, Colditz GA, Rosner B, Willett WC. Food-based validation of a dietary questionnaire: the effects of week-to-week variation in food consumption. Int J Epidemiol 1989;18:858–67. [DOI] [PubMed] [Google Scholar]
- 24. Rimm EB, Giovannucci EL, Stampfer MJ, Colditz GA, Litin LB, Willett WC. Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am J Epidemiol 1992;135:1114–26. [DOI] [PubMed] [Google Scholar]
- 25. Willett WC. Nutritional epidemiology. 2nd ed New York: Oxford University Press; 1998. [Google Scholar]
- 26. Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2011;96:1911–30. [DOI] [PubMed] [Google Scholar]
- 27. Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr 2000;72:690–3. [DOI] [PubMed] [Google Scholar]
- 28. Carwile JL, Willett WC, Michels KB. Consumption of low-fat dairy products may delay natural menopause. J Nutr 2013;143:1642–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Kim S, Kim JJ, Kim M-J, Han KH, Lee JR, Suh CS, Choi YM, Kim SH. Relationship between serum anti-Mullerian hormone with vitamin D and metabolic syndrome risk factors in late reproductive-age women. Gynecol Endocrinol 2017;34:327–31. [DOI] [PubMed] [Google Scholar]
- 30. Naderi Z, Kashanian M, Chenari L, Sheikhansari N. Evaluating the effects of administration of 25-hydroxyvitamin D supplement on serum anti-Mullerian hormone (AMH) levels in infertile women. Gynecol Endocrinol 2017;35:409–12. [DOI] [PubMed] [Google Scholar]
- 31. Chun RF. New perspectives on the vitamin D binding protein. Cell Biochem Funct 2012;30:445–56. [DOI] [PubMed] [Google Scholar]
- 32. Nielson CM, Jones KS, Bouillon R. Role of assay type in determining free 25-hydroxyvitamin D levels in diverse populations. N Engl J Med 2016;374:1695–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kotsopoulos J, Tworoger SS, Campos H, Chung F-L, Clevenger CV, Franke AA, Mantzoros CS, Ricchiuti V, Willett WC, Hankinson SE et al. Reproducibility of plasma and urine biomarkers among premenopausal and postmenopausal women from the Nurses’ Health Studies. Cancer Epidemiol Biomark Prev 2010;19:938–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Eliassen AH, Warner ET, Rosner B, Collins LC, Beck AH, Quintana LM, Tamimi RM, Hankinson SE. Plasma 25-hydroxyvitamin D and risk of breast cancer in women followed over 20 years. Cancer Res 2016;76:5423–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Sonderman JS, Munro HM, Blot WJ, Signorello LB. Reproducibility of serum 25-hydroxyvitamin D and vitamin D-binding protein levels over time in a prospective cohort study of black and white adults. Am J Epidemiol 2012;176:615–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Lai JKC, Lucas RM, Clements MS, Harrison SL, Banks E. Assessing vitamin D status: pitfalls for the unwary. Mol Nutr Food Res 2010;54:1062–71. [DOI] [PubMed] [Google Scholar]
- 37. Colditz GA, Stampfer MJ, Willett WC, Stason WB, Rosner B, Hennekens CH, Speizer FE. Reproducibility and validity of self-reported menopausal status in a prospective cohort study. Am J Epidemiol 1987;126:319–25. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.