Abstract
Although the extremely thermophilic archaea (Topt ≥ 70°C) may be the most primitive extant forms of life, they have been studied to a limited extent relative to mesophilic microorganisms. Many of these organisms have unique biochemical and physiological characteristics with important biotechnological implications. These include methanogens that generate methane, fermentative anaerobes that produce hydrogen gas with high efficiency, and acidophiles that can mobilize base, precious and strategic metals from mineral ores. Extremely thermophilic archaea have also been a valuable source of thermoactive, thermostable biocatalysts, but their use as cellular systems has been limited because of the general lack of facile genetics tools. This situation has changed recently, however, thereby providing an important avenue for understanding their metabolic and physiological details and also opening up opportunities for metabolic engineering efforts. Along these lines, extremely thermophilic archaea have recently been engineered to produce a variety of alcohols and industrial chemicals, in some cases incorporating CO2 into the final product. There are barriers and challenges to these organisms reaching their full potential as industrial microorganisms but, if these can be overcome, a new dimension for biotechnology will be forthcoming that strategically exploits biology at high temperatures.
Keywords: extremely thermophilic archaea, biotechnology, biocatalysis, biooxidation
New developments in molecular genetics tools have expanded the biotechnological scope of extremely thermophilic archaea to include metabolic engineering while enhancing opportunities already in place.
INTRODUCTION
Nearly four billion years ago, on an earth still cooling with a thin oxygen-free atmosphere, microbial life arose (Olsen, Woese and Overbeek 1994). While debate still surrounds the details of primordial biology, extremely thermophilic archaea are ‘living fossils’ and provide a glimpse into this critical period in evolution (Whitfield 2004). Often overshadowed by their prokaryotic cousins, the Bacteria, in terms of both public perception and scientific study, their biochemical and physiological features offer intriguing opportunities for biotechnology. These are directly related to their proposed primitive beginnings: the ability to inhabit and thrive at extreme temperature and pH, along with metabolizing simple but technologically important compounds, such as hydrogen gas and C1 chemicals (methane, carbon dioxide, carbon monoxide), present in volcanic vents in the Hadean Ocean. As another hint of their prehistoric nature, extremely thermophilic archaea typically have small genomes (∼2 Mb), potentially simplifying systems biology analysis and subsequent metabolic engineering efforts.
To date, archaea have lagged behind bacteria and eukaryotes on the industrial biotechnology stage, yet they have been important in several ways, most notably for their role in copper biomining (Wheaton et al.2015) and for their high-fidelity thermostable DNA polymerases in the polymerase chain reaction (PCR) (Pavlov et al.2004). Recent research breakthroughs have demonstrated that facile genetic systems developed for several archaea can be used for engineering non-native chemical production, in some cases from inorganic substrates, such as carbon dioxide (Keller et al.2013; Hawkins et al.2015; Zeldes et al.2015; Loder et al.2016). In applications where the most widely used mesophilic, metabolic engineering platforms, such as Escherichia coli and Saccharomyces cerevisiae, have failed or performed poorly, extremely thermophilic archaea offer a compelling option to overcome certain bioprocessing problems, such as contamination and phage infection.
A return to the extremely thermophilic archaea, typically located at the roots of phylogenetic trees, brings with it opportunity. A narrative that probably began billions of years ago in undersea volcanic vents enters the post-genomics era with momentum, promise and significance. Herein, we chronicle the contributions associated with extremely thermophilic archaea to current biotechnology and peer into the future to preview the promise of these microorganisms and their associated biomolecules.
The key to any biotechnological uses of the extremely thermophilic archaea is the isolation of these microorganisms from unusual environments. Following Woese's seminal proposal that the archaea constitute a third domain of life (Woese and Fox 1977), there was a dramatic increase in the isolation of new extremely thermophilic archaea, led by the pioneering efforts of Stetter (Stetter 1996), Jannasch (Jorgensen, Isaksen and Jannasch 1992) and other intrepid microbiologists. These archaea were obtained from a variety of globally diverse, natural thermal features, ranging from deep sea hydrothermal vents to shallow terrestrial hot springs. By 2000, the pace of finding new genera of extremely thermophilic archaea slowed considerably, in part due to some convergence in the physiological characteristics of microorganisms being isolated from disparate natural biotopes. In fact, had genome sequencing technology been more widely available sooner, it would have been clear that many ‘newly’ identified archaea were in fact very closely related to previous finds. Since 2000, there have been relatively few reports of extremely thermophilic archaea with definitively new properties. However, numerous biotechnological opportunities exist based on those archaea that have already been described and future discoveries of new archaea should be expected. In fact, as we become more knowledgeable about extreme thermophile physiology and metabolism, and as ‘omics’ tools and systems biology approaches become even more powerful, return trips to thermal biotopes previously examined could identify important archaea that were overlooked previously. In addition, re-examination of extreme thermophiles already ‘in captivity’ may reveal previously undiscovered features that have biotechnological importance.
BIOTECHNOLOGICAL POTENTIAL OF EXTREMELY THERMOPHILIC ARCHAEA
Table 1 lists representative extremely thermophilic archaea with biotechnological potential as sources of important enzymes or as a consequence of their metabolic and physiological features. Several have facile genetic systems, making them even more significant as promising metabolic engineering platforms. More detailed discussion of these, and other extremely thermophilic archaea, can be found in the following sections. Nevertheless, it would be useful to first provide some context about the approaches that have been employed and could be taken to exploit the potential of these microorganisms for biotechnology.
Table 1.
Selected extremely thermophilic archaea with biotechnological potential.
| Archaeon | Genome/ isolation reference | Topt | pHopt | Anaerobe | Aerobe | Growth on α,β-glucans | Growth on peptides | CO2 Fixation | CO Oxidation | Carboxydotrophic | Sulfur Oxidation | Iron Oxidation | H2 Production | Genetic System | Biotech relevance |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pyrococcus | Fiala and | 100 | 6–7 | X | X | X | X | X | ME Platform (3-HP, ethanol, butanol, | ||||||
| furiosus | Stetter (1986) | oxidize CO); source of proteases and glycoside hydrolases | |||||||||||||
| Thermococcus | Fukui et al. | 85 | 6–7 | X | X | X | X | X | ME platform; source of proteases | ||||||
| kodakarensis | (2005) | and glycoside hydrolases | |||||||||||||
| Thermococcus | Lee et al. | 80 | 8.5 | X | X | X | X | X | X | Demonstrated use in producing H2 | |||||
| onnurineus NA1 | (2008) | from steel mill gas effluents | |||||||||||||
| Methanococcus | Bult et al. | 85 | 5–7 | X | X | Methane generation from H2 and | |||||||||
| jannaschii | (1996) | CO2 | |||||||||||||
| Sulfolobus | She et al. | 80 | 2–4 | X | X | X | a | a | X | Source of proteases and glycoside | |||||
| solfataricus | (2001) | hydrolases; overexpression of biotech enzymes | |||||||||||||
| Sulfolobus | Chen et al. | 75 | 2–3 | X | X | a | a | X | Potential ME platform | ||||||
| acidocaldarius | (2005) | ||||||||||||||
| Sulfolobus | Huber and | 70 | 2–3 | X | X | X | X | Prolific iron and sulfur oxidizer | |||||||
| metallicus | Stetter (1991) | utilized in bioleaching of ores | |||||||||||||
| Metallosphaera | Auernik et al. | 73 | 2-–3 | X | X | X | X | bioleaching of ores | |||||||
| sedula | (2008) | bioleaching of ores | |||||||||||||
| Acidianus | Segerer et al. | 70 | 2 | X | X | X | X | X | X | Prolific iron and sulfur oxidizer | |||||
| brierelyi | (1986) | utilized in bioleaching of ores |
Reported for some species.
In the 1990s, interest in extremely thermophilic archaea was fueled by the potential of their intrinsically thermophilic enzymes as biocatalysts that would be robust in the face of biologically unfavorable industrial conditions (Adams 1994; Adams and Kelly 1995, 1998; Adams, Perler and Kelly 1995). When it became clear that the genes encoding these enzymes could be overexpressed and the resulting polypeptides folded into their functional form in mesophilic microbial hosts (Zwickl et al.1990), biotechnology ventures arose. Diversa Corporation was one example of such an enterprise that leveraged innovative methods for high-throughput screening from gene expression libraries to access biodiversity for a range of enzyme-based applications (Palackal et al.2004; Solbak et al.2005). However, one potential concern for large-scale applications of archaeal enzymes then, that remains today, is the challenge of obtaining the high overexpression levels needed to meet industrial demand on economic scale, which is typically multigrams of protein per liter.
In addition to being a source of thermostable and thermoactive biocatalysts, several of the archaea listed in Table 1 have industrial potential as sources of novel biosynthetic pathways for producing biobased fuels and chemicals that could be engineered into mesophilic microorganisms with highly developed genetic systems, such as E. coli or S. cerevisiae. However, the incompatibility of the host temperature range with the enzymes from the extreme thermophile is problematic. This may not be an insurmountable obstacle in all cases. Some extremely thermophilic enzymes are active in mesophilic temperature ranges, but clearly this will be a technological barrier if suboptimal temperatures are required for their application. The solution currently being pursued is to establish extreme thermophiles as metabolic engineering platforms through the development of molecular genetics tools for these microorganisms. There are many promising advances in this regard (Adams and Kelly 2017), such that certain extremely thermophilic archaea can already be metabolically engineered to produce industrial chemicals and fuels. These archaea have great promise as new platforms which can advantageously exploit elevated temperatures as well as their unique biological characteristics (Zeldes et al.2015; Counts et al.2017; Loder et al.2017).
H2 AND C1 METABOLISM IN EXTREMELY THERMOPHILIC ARCHAEA
From a biotechnological perspective, one of the most promising characteristics of extremely thermophilic archaea is their ability to use and produce simple chemicals, including C1 compounds (CO, formate, CO2, CH4) and molecular hydrogen (H2). As mentioned, this ability may map back to their primordial origins when these chemicals were the primary carbon and energy sources available. Now, as we understand more about the underlying metabolism involving these chemicals, avenues to biotechnology are emerging.
Molecular hydrogen (H2)
Hydrogen has potential as a renewable and carbon-neutral energy carrier. With the availability of the commercial fuel cell applications, the demand for renewable hydrogen gas is expected to increase dramatically in the near future. However, the current methods of industrial H2 production still heavily rely on fossil fuels, so alternative methods for renewable hydrogen production are desirable. For biohydrogen production, an increase in temperature from 37°C to 100°C enhances the entropy by 20%, as determined from the Gibbs free energy relationship. Therefore, elevated temperature benefits H2 production (Verhaart et al.2010). Extensive studies on H2 metabolism have focused on Pyrococcus furiosus, Thermococcus onnurineus NA1 and T. kodakarensis KOD1, aided by the availability of genetic systems (Kim et al.2010; Lipscomb et al.2011; Hileman and Santangelo 2012). These archaea have proven to be prolific H2 producers. For example, in continuous culture, P. furiosus grown on maltose and T. kodakarensis grown on pyruvate demonstrate similar specific H2 production rates of 0.16 and 0.12 g g−1 h−1, respectively (Schicho et al.1993; Kanai et al.2005). Similarly, the CO-dependent H2 production from T. onnurineus NA1 achieved 0.19 g g−1 h−1 with continuous CO feed (Kim et al.2013). Improvements in H2 production by these and related archaea will come with improved understanding of their metabolism to inform metabolic engineering efforts.
Hydrogenases, which catalyze the reversible reaction between protons, an electron source and hydrogen, can be classified into three types based on the metal content in the catalytic site: [NiFe], [FeFe] and mononuclear Fe hydrogenases but, for reasons that are not clear, the [FeFe]-enzymes have yet to be found in any of the archaea (Vignais and Billoud 2007; Sondergaard, Pedersen and Greening 2016). The NiFe-enzymes are ubiquitous and the extremely thermophilic archaeon P. furiosus possesses three such enzymes. One is membrane-bound hydrogenase (MBH) and two are in the cytoplasm (SHI and SHII). Most members of the Thermococcales contain genes encoding at least one MBH and one SH (Schut et al.2013). The physiological function of MBH is to generate hydrogen from the reducing equivalents generated by glycolysis as well as to produce a H+/Na+ gradient for energy conservation (Sapra, Bagramyan and Adams 2003). The deletion of the genes encoding MBH in P. furiosus abolished H2 production and eliminated growth in the absence of elemental sulfur as an external electron acceptor, while in T. kodakarensis the overexpression of MBH leads to increased H2 production (Schut et al.2012; Kanai et al.2015). The predicted function of SH is to oxidize the H2 that is produced and regenerate reduced nicotinamide cofactors, but the deletion of SH in P. furiosus did not affect growth, indicating that alternative systems are present to provide NADPH for biosynthesis (Lipscomb et al.2011). In P. furiosus, no significant change in H2 production rate was observed when the genes encoding both SHI and SHII were deleted, while a 10% increase in H2 production rate was obtained when the SH was knocked out in T. kodakarensis (Kanai et al.2011; Schut et al.2012). The type of glycosidic linkage in the carbon sources also affects H2 production in P. furiosus, where cellobiose-grown cultures have 50% higher specific H2 generation rates compared to maltose-grown cultures (Chou et al.2007). Efforts have been reported to utilize extremely thermophilic archaea for H2 production from waste materials, such as chitin, agricultural waste and feather meal (Balint et al.2005; Hensley, Moreira and Holden 2016; Aslam et al.2017), which can be re-visited if metabolic engineering strategies to improve the conversion of sugars and peptides become available.
In vivo, biological fermentation for H2 production is limited by the so called ‘Thauer Limit’, where the maximum theoretical yield is 4 H2 per glucose. Nevertheless, this could be overcome by cell-free in vitro synthetic systems that have a theoretical yield of up to 12 H2/glucose, with half of the hydrogen atoms provided by water. The full oxidation of all six carbons to CO2 allows this to be a thermodynamically feasible and redox balanced pathway. In addition, in vitro synthetic systems have an advantage of producing H2 as the primary product compared to biological fermentation systems. Because of its specificity for NADP+ and intrinsic stability, soluble hydrogenase I (SHI) from P. furiosus has been used in a wide range of applications, including the production of reduced nicotinamide cofactors and of hydrogen gas (Wu et al.2015). Among these applications, H2 production from sugars by an extensively studied synthetic pathway is one of the most promising methods for biohydrogen production currently. This in vitro synthetic system was originally designed to use glucose-6-phosphate (G6P) as the starting substrate to generate NADPH in a reconstructed pentose phosphate pathway using mesophilic enzymes from various sources, with H2 produced by SHI using the formed NADPH (Woodward et al.2000). This pathway was further modified to include a phosphorylation step to generate G6P from sugars (Zhang et al.2007). Over the last decade, this pathway has been engineered to use different enzymes to generate monosaccharides from diverse sugars as the energy source, including cellulosic materials (Ye et al.2009), xylose (Martin del Campo et al.2013), sucrose (Myung et al.2014), corn stover (Rollin et al.2015) and xylooligosaccharides (Moustafa et al.2016). Recently, instead of directly using NADPH for H2 production by SHI, a synthetic electron mediator, benzyl viologen (BV), was included in the pathway, where NADPH was used by NADPH rubredoxin oxidoreductase from P. furiosus to reduce BV that was further used by SHI for H2 production (Fig. 1) (Kim et al.2016a, 2017). Used in combination with enzymes from other thermophiles, the operating temperature was elevated to 50°C and the H2 productivity was enhanced more than 200-fold, to 0.18 g L−1 h−1, using starch as the energy source for in vitro H2 production (Zhang et al.2007; Kim et al.2017). This pathway also achieved the maximum yield of 12 H2 per glucose. A recently developed hybrid photocatalytic system consisting of nanocrystalline CdSe/CdS dot-in-rod coupled with SHI from P. furiosus also demonstrated that H2 could be produced in a high efficiency using light as the energy source (Chica et al.2017).
Figure 1.
In vitro synthetic pathway for hydrogen production, adapted from Myung et al. (2014), Kim et al. (2017). Enzymes from extremely thermophilic archaea: isoamylase from Sulfolobus tokodaii, 4-α-glucanotransferase from Thermococcus litoralis, NADPH:rubredoxin oxidoreducase and [NiFe]-hydrogenase from Pyrococcus furiosus.*Enzyme from extremely thermophilic archaeon.
Carbon monoxide (CO) and formate (HCOO−)
Despite its advantages as a renewable energy carrier, the issues of storage and delivery of H2 hinder potential applications, drawing interest to compounds such as formate as a potential hydrogen storage chemical (Joo 2008; Enthaler, von Langermann and Schmidt 2010). Two H2 production membrane complexes have been identified in the genus of T. onnurineus: formate hydrogen lyase (FHL) and carbon monoxide dehydrogenase (CODH) (Takacs et al.2008; Kim et al.2010, 2013; Kozhevnikova et al.2016). FHL and CODH are part of a modular family of MBH-type complexes that are involved in energy conservation by Na+-dependent H2 respiration (Schut et al.2016a). They have homologous subunits to MBH in P. furiosus, including the Na+/H+ transporter (Mrp) and the membrane-bound hydrogenase (Mbh). FHL contains subunits of formate dehydrogenase for conversion of formate to CO2, while CODH contains subunits of CO dehydrogenase that catalyze CO oxidation to CO2 (Lipscomb et al.2014; Schut et al.2016b). Production of H2 from T. onnurineus with substrates CO, formate or starch achieved modest specific production rates but yielded 98%,100% and 78% of theoretical conversion, respectively (Bae et al.2012). Growth conditions of T. onnurineus have been optimized for H2 production using formate as the energy source, and rates increased to 0.48 g L−1 h−1 (Bae et al.2015). When the cell density of T. onnurieneus increased, using formate as the energy source, a linear increase of volumetric H2 production rate with cell density was reported, where the productivity reached 5.70 g L−1 h−1 with a specific rate of 0.82 g g−1 h−1 (Lim et al.2012). The H2 productivity in T. onnurineus was further improved by overexpressing an F420-reducing class hydrogenase and the FHL complex to achieve a specific productivity of 1.01 g g−1 h−1, which is one of the highest productivities reported to date (Rittmann et al.2015). Three fhl operons were identified in the genome of T. onnurineus, with the fhl2 operon being essential for growth with formate as the energy source (Kim et al.2010). The operon encoding the 18-subunit fhl2 complex was heterologously expressed in P. furiosus and the amount of H2 production during the growth in the presence of formate and sugars was more than 2-fold higher than the parent strain (Lipscomb et al.2014).
The expression of CODH in T. onnurineus has been manipulated to also increase the H2 production rate from CO. The operon encoding CODH was overexpressed in T. onnurineus under the control of a strong promoter, and the H2 production increased by 3.8-fold over the wild type, reaching 0.27 g L−1 h−1. The specific rate was also 1.8-fold higher when the cells were grown in the presence of CO (Kim et al.2013). Thermococcus onnurineus can also utilize CO in waste gas from the steel industry for H2 production, although a reduction of the maximum rate by 30% was observed due to the relatively low content of CO in the waste gas (Kim et al.2013). Acetate production from steel mill waste gas has also been reported by using T. onnurineus in the first stage to which Linz-Donawitz converter gas (56% CO as the sole carbon source) was fed, followed by a bacterial homoacetogen (Thermoanaerobacter kivui) (Kim et al.2016b). Besides manipulating the expression of CODH, simply overexpressing a putative transcriptional regulator, TON1015, resulted in a 5-fold improvement in H2 productivity to 0.37 g L−1 h−1 in T. onnurineus compared to the wild type (Rittmann et al.2015). The 17-gene cluster encoding CODH in T. onnurineus has been heterologously expressed in P. furiosus, and when grown in the presence of CO, H2 production was enhanced by almost an order of magnitude compared to the growth in the absence of CO. The recombinant strain had also the ability to utilize CO as a respiratory energy source for growth (Schut et al.2016a).
Carbon dioxide (CO2)
Direct incorporation of CO2 into biobased fuels and chemicals is an aspirational goal of clean energy systems. This presents an important biotechnological opportunity for extremely thermophilic archaea capable of using CO2 as a carbon source. Of the six known carbon dioxide fixation cycles, two are found exclusively in extremely thermophilic acidophiles (Berg et al.2007) (Fig. 2). In contrast to photosynthetically powered carbon fixation characteristic of the Calvin-Benson-Bassham (CBB) cycle, thermoacidophilic archaea obtain energy to fix CO2 chemolithoautrophically, via aerobic oxidation of sulfur (i.e. Sulfolobus metallicus; Huber and Stetter 1991), oxidation of ferric iron (Fe2+) (i.e. Metallosphaera sedula; Huber et al.1989) or anaerobic reduction of sulfur with hydrogen (i.e. Ignococcus hospitalis; Paper et al.2007). While these pathways may appear to have a common root, most evidence points to convergent evolution, which involves substantially different enzymes to perform similar reactions (Braakman and Smith 2012).
Figure 2.
CO2 fixation cycles including those found exclusively in extremely thermophilic archaea. The DC/4-HB (blue) and 3-HP/4-HB (green) are found exclusively in extremely thermophilic archaea. These cycles share many intermediates with the reverse TCA (rTCA) (yellow) cycle as well as the 3-HP bicycle (gray) from green non-sulfur bacteria. TCA, tricarboxylic acid; 4-HB, hydroxybutyrate; 3-HP, 3-hydroxypropionate; DC, dicarboxylate
The 3-hydroxypropionate (3-HP) bicycle, found exclusively in the green non-sulfur bacteria family Chloroflexaceae, relies on two branches with overlap through a central chain from acetyl-CoA through propionyl-CoA (Strauss and Fuchs 1993) (see Fig. 2). The central branch is responsible for sequestering two carbons: one branch produces a two-carbon glycoxylate, while the second branch assimilates glycoxylate, producing pyruvate. Both branches generate two carbons as acetyl-CoA, heading the central trunk of the pathway. At present, primary interest in utilizing this pathway has centered upon mesophilic hosts, such as E. coli, which have produced 3-HP titers above 40 g L−1 (Liu et al.2016). Although this pathway contains 14 enzymes (Strauss and Fuchs 1993), its bioenergetic and redox requirements provide an advantage over related cycles (DC/4-HB and 3-HP/4-HB), as discussed below.
In the dicarboxylate cycle/4-hydroxybutyrate (DC/4-HB) cycle (see Fig. 2), the section of the pathway from succinyl-CoA to acetyl-CoA is nearly identical to the 3-HP/4-HB cycle with its most unique feature being the three-step carbon fixing pathway from acetyl-CoA to oxaloacetate (Huber et al.2008). In two steps, the DC/4-HB cycle sequesters two carbons at the expense of only one ATP and one reducing equivalent, nearly matching the efficiency of the reverse TCA cycle (rTCA). Thus, the DC/4-HB has been proposed as a potential thermophilic pathway to produce succinate, requiring two ATP and three reducing equivalents in addition to a source of acetyl-CoA to form succinate. The phosphoenolpyruvate carboxylase, the central enzyme in this pathway, could be used in conjunction with other synthetic pathways due to this energy conservation (Bar-Even et al.2010).
As with rTCA and DC/4-HB cycles, acetyl-CoA exits the 3-HP/4-HB cycle to meet cellular needs. As it requires 4 ATP and 4 reducing equivalents to fix a single carbon dioxide, it is the least energy efficient of the six carbon dioxide fixation pathways. This inefficiency is due to the lack of energy conservation during CoA transfers. Two of the four ATPs consumed in the cycle are required to fix carbonate, but the cycle also requires ATP at two separate points to transfer a free CoA onto a carboxylic acid. Given that it does not contain any intermediate that is only found in the cycle and its status as the least efficient carbon fixation pathway, at this point the biotechnological importance of the 3-HP/4-HB cycle stems from the three enzyme route from acetyl-CoA to 3-HP. Utilizing acetyl-CoA carboxylase and two successive NADPH-dependent reductases, 3-HP production has been demonstrated in a recombinant extremely thermophilic host, P. furiosus, which grows optimally near 100°C (Keller et al.2013; Hawkins et al.2015; Lian et al.2016). By insertion of the three enzymes from M. sedula, P. furiosus was able to produce up to 0.4 g L−1 3-HP at 72°C, a non-optimal temperature for the host, but the optimal temperature for the pathway (Lian et al.2016).
There are other features in extremely thermophilic archaea that relate to CO2 fixation in mesophilic organisms. The CBB cycle is responsible for the majority of the earth's carbon sequestration from the environment. The cycle depends upon Rubisco (ribulose-1,5-bisphosphate carboxylase/oxygenase) for the carboxylase activity, which is not only a relatively slow enzyme, but also suffers from catalyzing a competitive oxygenase reaction. In order to remove the bottleneck in CO2 fixation processes, significant efforts to enhance both the kinetics and specificity of Rubisco have been undertaken. While there are no known archaea utilizing the CBB cycle, type III Rubisco enzymes are found exclusively in archaea, possibly hinting at the evolutionary heritage of this important protein (Sato, Atomi and Imanaka 2007). Thermococcus kodakarensis contains such a version of this Rubisco that is neither affected by nor inactivated by oxygen (Ezaki et al.1999). Additionally, this enzyme has been shown to function in vivo when cloned into a mesophilic bacterium, Rhodopseudomonas palustris, demonstrating its potential to operate at ambient temperatures (Nishitani et al.2010). More recent efforts have attempted to understand these properties as well as explore mutations for optimization and insertion into photosynthetic mesophilic plants, bacteria or algae to enhance carbon fixation (Fujihashi et al.2016).
Analysis of the existing CO2 fixation cycles has inspired the design of synthetic carbon fixation pathways by combining enzymes from a variety of sources toward achieving a kinetically and thermodynamically favorable route for sequestering carbon into commercial products. In many cases, the carboxylation step—integration of carbon dioxide (or bicarbonate – HCO3−)—is the rate-limiting step. A survey carboxylase enzymes responsible for the fixation of CO2 or carbonate (HCO3−) suggested that an optimized synthetic pathway should contain phosphoenolpyruvate carboxylase (from DC/4-HB) and/or pyruvate carboxylase (in production of oxaloacetate) which possess the highest activity and affinity for the carbon species of those carboxylating enzymes yet reported (Bar-Even et al.2010).
The thermodynamics of many biological reactions are at, or near, equilibrium and a subtle shift in pH, temperature or other environmental factors can affect the favorability of certain pathways. These modifications in the environmental conditions in which the enzymes function could result in reactions with lower potential electron carriers or energy conserving steps. The archaeal carbon fixation cycles provide an enzymatic toolkit to construct hybrid pathways that function over a wide range of temperature and pH. Since many of these enzymes retain significant activity at lower temperatures, their potential use is not limited to thermophile metabolic engineering, but may find use in photosynthetic plants and algae (Wilson, Alonso and Whitney 2016).
Methane (CH4)
Methane is a valuable energy source and the biological production of this hydrocarbon offers biotechnological opportunities ranging from treating agricultural and domestic wastewater as well as the conversion of H2 into an energy carrier that is compatible with the existing natural gas infrastructure (Jentsch, Trost and Sterner 2014; Puyol et al.2016; Lecker et al.2017). All methane-generating organisms, including those responsible for methane generation in the human gut, are archaea. Methane production in pure culture can proceed through acetoclastic methanogenesis where acetate is converted to CO2 and CH4, methylotrophic methanogenesis where methylated compounds are converted to methyl-S-CoM and either reduced by H2 or disporportionated to CO2 and CH4, or through hydrogenotrophic methanogenesis where H2 reduces CO2 to CH4 (Costa and Leigh 2014). Acetoclastic and methylotrophic methanogenesis are performed only by the Methanosarcinales and are common in anaerobic digester communities as they catalyze the last step for treatment of agricultural and domestic waste (Ferry 1992; Fournier and Gogarten 2008). However, the Methanosarcinales grow at temperatures less than 70°C and have lower growth rates compared to extremely thermophilic methanogens, which limits acetoclastic and methylotrophic methanogenesis to mesophilic and thermophilic archaea (De Vrieze et al.2012).
While CO2 reduction by hydrogen is thermodynamically feasible, this reaction becomes less thermodynamically favorable at higher temperatures (Thauer et al.2008). Autotrophic methanogens attract biotechnological interest as they are able to use H2 to upgrade CO2 to methane from waste gases (Simon 2015). While not as efficient as using H2 directly as a fuel, it has the potential for reducing the effects of methane and CO2 emissions, since the methane combustion emissions are immediately re-captured. Several high temperature autotrophic, methanogenic archaea have been characterized including Methano(caldo)coccus jannaschii (Topt 80°C), Methanothermus fervidus (Topt 83°C), Methanocaldococcus villosus (Topt 80°C) and Methanopyrus kandleri (Topt 98°C). Despite the thermodynamic challenges of methanogenesis, these archaea can grow rapidly, in some cases with doubling times as under 1 h (Stetter et al.1981; Jones et al.1983; Kurr et al.1991; Bellack et al.2011). These organisms were isolated from thermal features and likely survive on the combination of H2 from thermal fluids and H2 production from heterotrophs (Ver Eecke et al.2012; Topçuoğlu et al.2016). Autotrophic methanogenesis and growth rates can be uncoupled due to factors such as temperature and hydrogen availability (Tsao et al.1994; De Poorter, Geerts and Keltjens 2007; Ver Eecke et al.2012). As a result, methane generation is dependent on these factors, along with reactor conditions including gas flow rates and agitation speeds (Rittmann 2015).
Biogas upgrading to improve energy content has been limited to mesophilic and moderately thermophilic systems, as few studies have considered more thermophilic methanogens. To optimize H2 mass transfer and methane production, several reactor configurations have been employed including trickle bed and upflow reactors (Lee et al.2012; Burkhardt, Koschack and Busch 2015). Although these studies were conducted with inocula from mesophilic anaerobic digesters, they were nonetheless able to produce high concentrations and productivities of methane. Reactor configurations that address mass transfer limitations for methane production have not been explored for more thermophilic archaea, although such approaches could provide enhanced volumetric productivities, considering the rapid growth rates of some extremely thermophilic methanogens.
SULFUR
Sulfur is an essential element in biology, existing in oxidation states ranging from –2 (H2S) to +6 (H2SO4), thereby contributing to redox reactions as well as a constituent of metal-sulfur clusters in electron carriers and catalytic sites in enzymes. Sulfur is also important in the chemical industry, with nearly 70 million metric tons produced worldwide in 2016 almost exclusively recovered as a by-product during fossil fuel processing. Sulfur is used industrially, primarily in the form of sulfuric acid, with the majority being used to produce fertilizers, but with large amounts also utilized in the processing of petrochemicals and metallic ores (Ober 2002).
While normally present in trace amounts in nature, sulfur can become enriched in extreme environments, such as those inhabited by extremely thermophilic archaea. The high temperature and pressure characteristics of hydrothermal features separate solutes based on volatility. Less volatile salts are concentrated in the liquid phase and result in discharges of brackish water, while nearby vapor-dominated discharges contain mostly CO2 and H2S, resulting in sulfur-dominated acid pools as H2S oxidizes to sulfuric acid (Nordstrom, Ball and McClesky 2005). Deep-sea hydrothermal systems also release substantial amounts of reduced sulfur compounds in the form of metal sulfides and hydrogen sulfide (Hannington et al.1995). Sulfides are also present in the earth's crust as metallic sulfide ores. In these specialized environments, sulfur becomes not just an important nutrient to microorganisms, but also a key source of energy. Depending upon the reducing environment, sulfur compounds can serve as electron acceptors or donors for both heterotrophic and chemolithotrophic microorganisms. While the ability to use sulfur in this way is not unique to archaea, their prevalence in extreme environments means that they have adapted a variety of approaches to take advantage of sulfur where it is plentiful. The relationship between high-temperature organisms and sulfur has been known for some time (Amend and Shock 2001). Sulfur metabolism may also have played a key role in the early evolution of life—the ‘Iron-Sulfur World’ hypothesis suggests that early life appeared under thermoacidophilic conditions, with hydrothermal H2S serving as the electron donor (Wachtershauser 1988), although this theory is controversial (Bada and Lazcano 2002).
Sulfur reduction
Elemental sulfur (S0) is used as an electron acceptor in lieu of oxygen by a number of anaerobic thermophilic archaea, both autotrophs and heterotrophs. The reduced sulfur compounds that result can precipitate dissolved metal ions, which has applications in biotechnology and bioremediation. Perhaps the most common energy metabolism at extremely high temperatures is the reduction of S0 by heterotrophs, where electrons derived from peptides and sugars are transferred to S0, generating H2S and oxidized organic fermentation products (acetate, alanine) in the process. Members of the archaeal order Thermococcales are the best-studied examples of this metabolism. As discussed earlier, some Thermococcales also grow in the absence of S0, generating H2 from reduced ferredoxin (Fdred), but this growth mode is only possible on sugars, since peptide oxidation proceeds through the less reduced NADPH electron carrier, from which H2 production is thermodynamically unfavorable (Schut et al.2014). The redox-responsive transcription factor responsible for the switch from H2 to H2S production in P. furiosus (Topt = 100°C), SurR, has been characterized in great detail (Lipscomb et al.2009, 2017b; Yang et al.2010). Direct physical contact between cells and S0 was found to be unnecessary for P. furiosus sulfur reduction, suggesting the true substrate is soluble polysulfide, which forms abiotically under standard growth conditions (Blumentals et al.1990). Gaps remain in the current understanding of S0 reduction in Thermococcales, but some of the key players have been identified. A membrane-bound oxidoreductase complex (MBX), with homology to the MBH complex mentioned previously in connection with hydrogen production, is upregulated during growth on S0. However, membrane fractions exhibit no sulfur reduction activity and it was proposed that, given its high homology to MBH, MBX is responsible for energy conservation through unknown mechanisms, while a cytoplasmic NADPH sulfur oxidoreductase (NSR) is directly involved in H2S production (Schut, Bridger and Adams 2007). While knockout strains confirmed that MBX was essential for growth on S0, strains lacking NSR grew similarly to the parent strain and continued to produce H2S (Bridger et al.2011; Santangelo, Cubonova and Reeve 2011). The exact mechanism by which S0 is reduced is therefore not clear at present. While Pyrococcus and Thermococcus species are the best studied, heterotrophic S0 reduction is also observed in the thermophilic archaeal genera Acidilobus, Caldisphaera, Caldivirga, Desulfurococcus, Hyperthermus, Palaeococcus, Pyrobaculum, Staphylothermus, Stetteria, Thermophilium, Thermoplasma, Thermoproteus and Vulcanisaeta (Kletzin 2007). Thermoproteus tenax (Topt = 80°C) has been reported to have the unusual ability to oxidize organic compounds completely to CO2 using S0 as the electron acceptor (Selig and Schonheit 1994).
Autotrophic growth via S0 reduction is carried out by some members of the extremely thermophilic archaea where hydrogen gas is the source of electrons rather than organic compounds. However, the mechanism of heterotrophic S0 reduction is completely different from that of the autotrophs. The model system examined for this mode of autotrophic growth among archaea is the thermoacidophilic crenarchaeon Acidianus ambivalens (Topt = 80°C, pHopt = 2). This microorganism uses two closely associated membrane proteins, a sulfur reductase (SR) and a hydrogenase (Hyd), to generate an electrochemical membrane gradient, similar to the mechanism found in the mesophilic bacterium Wolinella succinogenes (Laska, Lottspeich and Kletzin 2003). Members of the hyperthermophilic crenarchaeal genus Pyrodictium (Topt = 100°C) contain similar membrane complexes and are also capable of autotrophic S0 reduction (Dirmeier et al.1998), as are Ignicoccus species (Huber et al.2000).
Dissimilatory sulfate reduction (DSR), the anaerobic reduction of sulfate to H2S by reduction of H2 or organic compounds, is an energy yielding process, in contrast to assimilatory sulfate reduction, which encompasses the uptake and incorporation of sulfur into biomolecules. The first confirmed archaeal sulfate reducers were in the hyperthermophilic genus Archaeoglobus (Topt = 85°C) (Stetter et al.1987), which share gene homologs for sulfate adenyltransferase (Sat), adenylyl sulfate reductase (AprAB) and sulfite reductase (DsrABD) with bacteria (Klenk et al.1997); recent genome sequence data suggest that the archaeon T tenax has also the requisite genes for sulfate reduction (Siebers et al.2011). The Dsr A, B and C subunits from Archaeoglobus fulgidus were integral in recent work elucidating the energy conserving mechanism of DSR (Santos et al.2015).
Biotechnological applications of sulfur-based reductive metabolisms face a variety of challenges: the bioenergetic yield is low compared to oxidative pathways, the product (H2S) is toxic and currently plentiful in natural gas and petrochemical sources, and the feedstocks (H2 and organics) are likely to be more valuable than the product, except in specialized cases. One place where sulfur reducers show great promise is for bioremediation of water contaminated with heavy metals, since the biologically produced H2S would react with soluble metal ions to produce insoluble metal sulfides (Florentino et al.2016). Sulfur-reducing microorganisms have been commercialized for sulfate removal (SULFATEQ) and heavy metals removal (THIOTEQ Metal) from wastewater by Paques (Muyzer and Stams 2008; Paques 2017). Biologically generated H2S for metal precipitation can be produced safely on site from elemental sulfur, and individual metals can be recovered by tuning the reactor pH (Sanchez-Andrea et al.2016). Many sulfur-reducing species also exhibit other growth modes (P. furiosus reducing protons to H2, A. ambivalens oxidizing S0), which may have additional industrial applications.
Sulfur oxidation
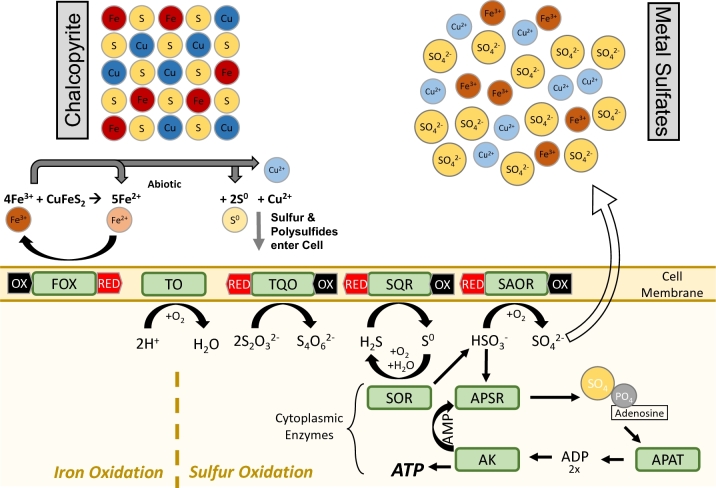
Autotrophic growth by the aerobic oxidation of S0 was a defining characteristic of the thermoacidophilic crenarchaeal order Sulfolobales (Brock et al.1972). This physiological feature is no longer considered universal for the order, but is still widespread (Albers and Siebers 2014). As with S0 reduction, the model for archaeal S0 oxidation is A. ambivalens, from which a variety of relevant enzymes have been characterized; unfortunately, a genome sequence for this archaeon is not currently available. Unlike the neutral and reduced growth conditions of S0 reducers, such as P. furiosus, oxygenated acidic environments do not favor the formation of soluble polysulfides. As a result, organisms like A. ambivalens must contend with S0 in its inert and minimally soluble elemental form. The key enzyme for oxidation of S0 is the cytoplasmic sulfur oxygenase reductase (SOR), though species that lack it may still oxidize other inorganic sulfur compounds. SOR disproportionates inert S0 into more reactive HSO32− and H2S in the presence of oxygen (Kletzin 1989). These sulfur compounds are then oxidized through a series of energy conserving steps, either involving membrane-associated or cytoplasmic enzymes (Zimmermann et al.1999). Membrane proteins that feed into the quinone pool (for eventual conversion to an electrochemical gradient via a terminal oxidase) include thiosulfate:quinone oxidoreductase (TQO) (Muller et al.2004), sulfide:quinone oxidoreductase (SQR) (Brito et al.2009) and sulfite:acceptor oxidoreductase (SAOR, no gene for which has yet been identified). The cytoplasmic pathway proceeds through an adenosine-5΄-phosphosulfate (APS) intermediate via APS reductase (APSR), APS:phosphate adenyltransferase (APAT) and adenylate kinase (AK) (see Fig. 3 and Table 2).
Figure 3.
Sulfur and iron oxidation pathways in extremely thermophilic archaeal species applicable to biomining. Biotic oxidation of ferrous iron (Fe2+) drives the supply of ferric iron (Fe3+) to abiotically dissolve the ore sulfides to elemental sulfur and polysulfides, rendering sulfur species available for biotic oxidation. Biotic and abiotic reactions are listed in Table 2.
Table 2.
Chemical/Biochemical sulfur oxidation reactions involving extremely thermophilic archaea.
| Species | Oxidation state of sulfur | Enzyme | Net Reaction | Comment |
|---|---|---|---|---|
| CuS | −2 | Abiotic | CuS + Fe3++ H+ Cu2+ + ½ H2Sn + Fe2+ Cu2+ + ½ H2Sn + Fe2+
|
– |
| H2Sn | 0 | Abiotic | ½ H2Sn + Fe3+ + H+ S0 + Fe2+ + H+ S0 + Fe2+ + H+
|
– |
| H2S | −2 | Sulfide quinone reductase (SQR) | H2S  S0 + 2H+ + 2e− S0 + 2H+ + 2e−
|
Reduced quinone to terminal oxidase |
| S0 | 0 | Sulfur oxygenase reductase (SOR) | 2S0 + ½ O2 +2H2O  H2S + HSO3− + H+ H2S + HSO3− + H+
|
– |
| Abiotic | S0 + ¾ H2O  ¼ SO32− + 4HS− + 8H+ ¼ SO32− + 4HS− + 8H+
|
 Favored at pH >7 Favored at pH >7 |
||
 Favored at pH <5 Favored at pH <5 |
||||
| Abiotic | S0 + HSO3− S2O32− + 8H+ S2O32− + 8H+
|
 Favored at pH >5 Favored at pH >5 |
||
 Favored at pH <4 Favored at pH <4 |
||||
| HSO3− | +4 | Adenylylsulfate reductase (APSR) | HSO3− + AMP  PAPS + 2e− PAPS + 2e−
|
ADP from AMP *Adenylate Kinase (AK) transforms (2 ADP  ATP + AMP) ATP + AMP)
|
| Adenylylsulate:phosphate adenylyltransferase (APAT) | PAPS + PO42− ADP + SO42- (PAPS = Phosphoadenylyl sulfate) ADP + SO42- (PAPS = Phosphoadenylyl sulfate) |
|||
| Sulfite:acceptor oxidoreductase (SAOR) | HSO3−+ H2O  SO42− + 3H+ 2e− SO42− + 3H+ 2e−
|
Reduced quinone to terminal oxidase | ||
| S2O32− | (–2,+6) | Thiosulfate:quinone oxidoreductase (TQO) | 2S2O32− S4O62−+ 2e− S4O62−+ 2e−
|
Reduced quinone to terminal oxidase |
| S4O62− | (+5,0,0,+5) | Abiotic | S4O62− + H2O  S0 + SO42− + S2O32−+ 2H+ S0 + SO42− + S2O32−+ 2H+
|
 Favored at pH >4 Favored at pH >4 |
| – | – | Terminal oxidase (TO) | 2H+ + ½ O2 + 2 e− H2O H2O |
ATP via ATP synthase |
Autotrophic oxidation of H2S has been reported in another member of the Sulfolobales, Su. metallicus (Morales et al.2011, 2012), possibly proceeding through a homolog of the SQR identified in A. ambivalens. This suggests potential applications in bioremediation, removing H2S from gas and water streams before they are released into the environment. THIOPAQ, a commercial technology for biological removal of H2S from gas streams (relying on a mesophilic bacterium), has been developed by Paques and applied to both biogas and fossil fuel gas streams (Janssen and Buisman 2001). A variety of bioreactor designs have been developed for H2S removal. While most rely on mesophilic bacteria, the use of thermophilic archaea in these systems would be advantageous, since the waste streams involved are often hot (>50°C) (Li et al.2015).
Sulfur oxidizers also play an important role in bioleaching, since sulfur forms a passivating layer on mineral ore surfaces, blocking access of metal oxidizers and inhibiting ore solubilization. Therefore, sulfur oxidizers are often beneficial contributors to microbial consortia in ore leaching, since they remove the sulfur barrier, while producing sulfuric acid to aid in solubilizing metal ions (more details in metals section). A similar role for sulfur oxidizers in the removal of sulfur impurities from coal was investigated (Peeples and Kelly 1993), although as coal power plants are phased out in favor of natural gas, the need for this sulfur removal could decline. Bioleaching is discussed in more detail in the next section.
The end product of sulfur oxidation is sulfuric acid, which as mentioned above is a key industrial chemical. The aqueous solutions produced biologically, even by acidophiles adapted to pH 1 and below (McCarthy et al.2015; Ai et al.2016), are too dilute for most industrial uses, but may have applications in lignocellulosic biomass pre-treatment and ore bioleaching operations. One advantage of biological sulfuric acid production is the ability to capture energy during its production, which can be utilized by sulfur-oxidizing autotrophs for carbon fixation. The resulting biomass, once separated from the acid stream, could serve as a protein supplement, or be converted to biogas by anaerobic digestion. If genetic systems were available, these autotrophs could even be engineered to produce a specific desired chemical from CO2 and sulfur.
METALS
Metals are essential in biological systems for many reasons, but in most cases they are found at the catalytic centers of enzymes. There are several interesting aspects of metals in extremely thermophilic archaea, including the use of tungsten as a metal center in central metabolic enzymes, such as the glyceraldehyde-3-phosphate oxidoreductase (Mukund and Adams 1996) and the aldehyde oxidoreductase (AOR) found in P. furiosus (Mukund and Adams 1990), and the unexpected range of metals identified in the metalloproteome (Cvetkovic et al.2010). From a biotechnological perspective, the ability of extremely thermoacidophilic archaea to oxidize metals is the basis for biohydrometallurgy applications related to the recovery of base, precious and strategic metals from mineral ores.
Metal biooxidation
Metal biooxidation is a particularly rare metabolic feature, limited mostly to acidophiles, whose environmental conditions favor the solubility of metal species, although some neutrophiles have been implicated in a similar process. The biochemical mechanism of biooxidation was first studied in the 1980s, when a redox-active protein structure was spectroscopically identified from iron-grown cells (Ingledew 1982; Barr, Ingledew and Norris 1990; Blake et al.1993). The mesophilic bacterium central to this study, Acidithiobacillus ferrooxidans (originally Thiobacillus ferrooxidans), is the most well characterized of the metal mobilizers, with an established working model for iron-mediated metal biooxidation (Quatrini et al.2009; Bonnefoy 2010). Membrane proteins shuttle electrons from extracellular ferrous iron via a cytochrome-c containing protein through a bifurcating pathway to either a type-aa3 terminal oxidase or an NAD+ reductase complex. The latter provides reducing potential, while the former provides energy via a membrane-associated ATPase, and may help to alleviate acid stress in the cytosol, which is maintained at neutral pH (Quatrini et al.2009). The two processes are driven by a modest electronic potential (from iron oxidation), resulting in the need for a large amount of substrate turnover and efficient fluxes to both pathways to maintain the vital pH homeostasis and provide energy for other cellular processes (Bonnefoy and Holmes 2012).
In contrast, much less is known about iron biooxidation under extremely thermophilic conditions (Wheaton et al.2015; Counts et al.2017). To date, the organisms that have been isolated from extremely thermoacidophilic environments have been limited to the crenarchaeal order Sulfolobales, with emphasis on the genera Sulfolobus, Acidianus and Metallopshaera. Most of our understanding of biooxidation at high temperatures has been limited to transcriptomic and comparative genomic data from only a few of these members, including Su. metallicus (Bathe and Norris 2007), M. yellowstonensis (Kozubal et al.2011) and M. sedula (Auernik and Kelly 2008). The central proteins for the process are encoded primarily in the fox stimulon, a well-conserved set of genes that demonstrate varying levels of transcriptomic activation in the presence of different iron substrates (Bathe and Norris 2007; Auernik and Kelly 2008; Auernik et al.2008) (see Fig. 3). Particularly responsive among these genes are foxC and foxD, which likely form a membrane-bound complex with a cytochrome b domain, that could presumably interact with iron as an initial electron acceptor. Additionally, foxG contains putative iron-sulfur domains, which could help with the flow of electrons to the multi-copper oxidase proteins. These genes are the only ones without demonstrated synteny within the order, but appear to be conserved among the metal mobilizers (Wheaton et al.2015) and highly responsive in the species M. yellowstonensis as well as in environmental metal-transcriptomic samples from hot spring iron mats (Kozubal et al.2011). In the final steps of the proposed pathway, electrons are either passed to a quinol pool, likely ending in a NAD+ reductase complex, or utilized to push protons from the interior to the exterior of the cell via FoxAB (against the natural gradient).
While the pathway can still be classified as hypothetical, the synteny of this stimulon among identified metal mobilizers, and its absence among non-metal mobilizers, provides further support for its function in iron biooxidation (Wheaton et al.2015). Even though the proposed mechanism relies on a similar scheme to that of Ac. ferrooxidans, there are key differences that highlight the vastly different evolutionary paths of these organisms, including but not limited to differences in membrane structures (archaea lack a periplasm), the presence of cytochrome b (as opposed to cytochrome c) and a possible bifurcating membrane soluble multi-copper oxidase (Ilbert and Bonnefoy 2013). The additional feature of thermal stability makes these proteins desirable targets for biotechnological innovations in the biomining field. The future of biomining was once described as ‘hot’ (Rawlings 2002), a designation that highlights the potential gains to be made from development of biomining technologies at higher temperatures.
Biomining
In the past century, significant focus was placed on the role of microbes in our natural geochemical environments. As a consequence, the field of biohydrometallurgy has matured to encompass all of the processes by which microbes interact with minerals. In particular, the role of microbes in mobilizing metals from metal-bearing ores has inspired the study of biomining, which involves a surprisingly large portion of the mineral processing industry. Recent estimates suggest that up to 20% of the copper and 5% of the gold produced globally relies on the intentional application of microbes (Johnson 2014; Watling 2016). These two minerals also represent two distinct applications of biomining. The first is bioleaching, which is the mobilization of target metal species by the microbe (e.g. copper, iron, nickel, etc.), whereas biooxidation occurs when a microbe interacts with occluding minerals that hinder the mobilization of other metal species (pyrite occlusion of gold or silver) (Brierley and Brierley 2013). A further distinction is the type of technology used in biomining: either heap or stirred-tank reactors. The former is the lower cost option that often involves stacking of large, milled, low-quality ore in beds, which can be operated in ambient conditions with aeration and percolated fluids, while the latter is a more capital intensive option, often reserved for high-value product streams where recovery is a primary focus (Schippers et al.2014). Both have been used successfully in industrial applications but have unique advantages, depending on the dynamics of the mineral system. In particular, stirred-tank biooxidation achieved early success as a means for improving gold yields via microbial pretreatment. It dates back to the 1980s and remains a common practice (Kaksonen, Mudunuru and Hackl 2014). In this case, microbes oxidize sulfur species (the details of which are provided in the previous section) in the bulk ore material in order to improve dissolution using lixiviants downstream (e.g. cyanide) (van Aswegen et al.2007). While this process was originally operated with mesophilic microbes, extremely thermophilic archaea can enhance the dissolution of sulfidic ores and consequently improve downstream yields (Lindström, Sandström and Sundkvist 2003; van Aswegen et al.2007; Astudillo and Acevedo 2009; Ciftci and Akcil 2013).
Copper has also been another focus of stirred-tank reactor technology, particularly with the adaptation of the BIOX process for a high-temperature copper extraction technology, BIOCOP (Batty and Rorke 2006). There have been numerous demonstrations of increased dissolution of copper sulfide ores, considered recalcitrant, with thermophiles in comparison to mesophiles (Sandström and Petersson 1997; Le Roux and Wakerley 1998; Dew et al.2000; d’Hughes and Foucher 2001; Gericke, Pinches and van Rooyen 2001; du Plessis, Batty and Dew 2007; Norris et al.2012; Li et al.2014). Due to the rising value of copper, interest in thermophilic biomining ramped up in the early 2000s, based on potential advantages at high temperatures, ranging from enhanced kinetics and favorable redox potentials (Ahonen and Tuovinen 1990; Gericke, Govender and Pinches 2010) to minimizing adverse effects resulting from passivation or electron interfacial changes (Khoshkhoo et al.2014; Crundwell 2015). As the quality of available ores trends toward lower grades and mixed sulfides, extreme thermophiles have been investigated for their niche advantages, such as co-solubilization of other useful metals (e.g. nickel or zinc) (Sandström and Petersson 1997; Konishi, Nishimura and Asai 1998; Langwaldt 2007; Gericke and Govender 2011; Vukovic et al.2014; Norris 2017) or even other high-value metals, such as silver (Norris et al.2017). Another distinct advantage is their ability to avoid the dissolution of microbially deleterious metal species, such as molybdenum (Romano et al.2001; Abdollahi et al.2014). Finally, these same organisms have shown promise in detoxifying arsenic species via oxidation in arsenopyritic sludge or in the presence of arsenic-containing copper ores (e.g. enargite), which are not conducive to traditional pyrometallurgy due to the chance of creating toxic metal dusts (Hita et al.2008; Takatsugi, Sasaki and Hirajima 2011).
For all of their advantages, several problems have been identified with using extremely thermophilic organisms in biomining operations (Donati, Castro and Urbieta 2016). Specifically, gas–liquid mass transfer can be diminished at high temperatures; this could lead to limited yield in both heaps and stirred-tank reactors (du Plessis, Batty and Dew 2007). Several studies have examined the role of O2 and CO2 on bioleaching with thermophiles, suggesting that strategies are needed to either enrich gas feed streams or improve gas transfer of both gas species (De Kock, Barnard and du Plessis 2004; Astudillo and Acevedo 2009). For many of these organisms, biooxidation is achieved in chemolithoautotrophic environments, where CO2 is required to build biomass and O2 is essential for maintaining pH homeostasis and electronic potential (Wheaton et al.2015). Another issue has been the inability of extreme thermophiles (in particular Su. metallicus) to function at high pulp densities, which are necessary to boost the economics of extremely thermophilic biomining. Several reasons for this have been suggested, such as sheer stress and metal toxicity (du Plessis, Batty and Dew 2007), while increased CO2 in feed streams, serial adaptation of cultures and particle size control have been suggested as solutions to this issue (Astudillo and Acevedo 2008, 2009; Jones, van Hille and Harrison 2012).
In summary, extremely thermophilic archaea have some distinct advantages in the field of biomining, ranging from selective dissolution of high-value minerals, inherent pathways for detoxification and robust ability to handle large fluctuations in temperature resulting from low-grade gauge materials. These observations warrant further investigation, which could lead to improved processes for existing mesophilic and moderately thermophilic industrial processes or new potential applications of thermophilic metal mobilizers. Furthermore, these extremely thermoacidophilic archaea deserve more attention as they come from a domain that has only become a focus of microbiologists in the previous half-century. They contain a plethora of enzymes and pathways for which we have limited knowledge, and the number of new islotes is continually growing as geothermal sites throughout the world are explored.
ENZYMES FOR MOLECULAR BIOLOGY
The earliest commercial use of enzymes from extremely thermophilic archaea involved molecular biological applications, specifically for the PCR. DNA polymerases from T. litoralis (Vent Polymerase) (Perler et al.1992) and P. furiosus (Pfu Polymerase) (Lundberg et al.1991) found wide use as higher fidelity alternatives to the bacterial Taq Polymerase, but there are also more recent applications based on polymerases, ligases and inteins from extremely thermophilic archaea (see Table 3).
Table 3.
Extremely thermophilic archaeal enzymes used in molecular biology.
| Enzyme | Origin | Application | References |
|---|---|---|---|
| DNA polymerases | Thermococcus barophilus Ch5 | PCR | Kwon et al. (2016) |
| Pyrococcus furiosus | PCR, error-prone PCR | Biles and Connolly (2004); Wang et al. (2004) | |
| Sulfolobus solfataricus | Error-prone PCR | McDonald et al. (2006) | |
| Thermococcus kodakaraensis | PCR, one-step RT-PCR | Elshawadfy et al. (2014); Okano et al. (2017) | |
| Thermococcus JDF-3 | DNA sequencing | Arezi, Hansen and Hogrefe (2002) | |
| Thermococcus 9°N-7 | DNA sequencing, sequence-by-synthesis | Gardner and Jack (2002); Ju et al. (2006) | |
| Ligases | Thermococcus sp. 1519 | Gibson Assembly | Smagin et al. (2008) |
| Staphylothermus marinus | Ligase chain reaction | Seo et al. (2007) | |
| Pyrococcus furiosus | Ligase chain reaction | Tanabe et al. (2012) | |
| Methanothermobacter thermautotrophicus | 5’-adenylation | Sriskanda et al. (2000) | |
| Thermococcus kodakaraensis | RNA sequencing | Zhang and Tripathi (2017) | |
| Inteins | Pyrococcus horikoshii | Protein purification | Lennon, Stanger and Belfort (2016) |
| Purococcus furiosus | Biosensor | Iwai, Lingel and Pluckthun (2001) |
DNA polymerases
Polymerases (pols) are prevalent in nature and are classified into seven families: A, B, C, D, X, Y and reverse transcriptase (RT) (Burgers et al.2001). Family A and B pols are the most extensively studied; most archaeal pols fall into the B, D and Y families. The development of PCR (Mullis et al.1986), a powerful technique to amplify a targeted DNA sequence, was a pivotal step in the emergence of molecular biotechnology and led to many other advanced methods, including DNA and RNA sequencing. Thermal cycling is key to PCR, and this was the reason why the thermostable DNA polymerase from Thermus aquaticus (Chien, Edgar and Trela 1976) (or Taq polymerase) has been widely used for this purpose. Better DNA pol options than Taq pol have been sought, including those with the ability to proofread (or possess 3΄-5΄ exonuclease activity), and to extend mismatched primers for longer DNA templates. To this end, as mentioned above, the archaeal polymerase from T. litoralis (Mattila et al.1991) and others from the genera Pyrococcus and Thermococcus were used for their more precise proofreading ability. DNA pol from T. kodakaraensis KOD1 has a low error rate, high processivity and high extension rate, thus making longer DNA target amplification more accurate, up to 6 kb (Takagi et al.1997; Hashimoto et al.1999, 2001). By addition of a small amount of an archaeal family B pol, with 3΄-5΄ exonuclease activity, to the Taq pol reaction, improvements in PCR were obtained. A DNA pol from T. barophilus Ch5, when used in a mixture with Taq DNA pol, amplified 25 kb DNA templates at lower error rates than Pfu pol (Kwon et al.2016). Through mutagenesis, a low-fidelity DNA pol mutant from P. furiosus found use in error-prone PCR, a technique used to introduce random mutations into a gene, while retaining thermal stability and activity (Biles and Connolly 2004).
Along these lines, the low-fidelity Dpo4 DNA pol from Su. solfataricus was used in error-prone PCR and/or amplification of damaged DNA (McDonald et al.2006). There have been efforts to engineer the current archaeal pols to improve processivity by fusing a DNA-binding domain to one end of DNA pol, referred to as ‘chimeric DNA pol’. A non-specific DNA-binding protein (Sso7d) from Su. solfataricus was fused to the C-terminus of Pfu DNA pol; the resulting fusion (Phusion) DNA pol was 10 times more processive with 6-fold higher fidelity than the wild-type enzyme (Wang et al.2004). Given its high performance and accuracy, Phusion DNA pol is useful for high-fidelity PCR and site-directed mutagenesis. The chimeric pol between a helix-hairpin-helix (HhH) DNA-binding motif and Pfu DNA pol, or ‘Pfu-HhH DNA’ pol, has increased polymerization rates, increased thermal stability and is highly resistant to salt and inhibitors, such as phenol, blood and DNA intercalating dyes (Pavlov et al.2002). The chimeric Pfu-KOD1 DNA pols were made by fusing the high-fidelity KOD1 DNA pol with the thermostable Pfu DNA pol. As a result, this fusion DNA pol has all the desired characteristics of the precursors: high fidelity, high performance and high stability at elevated temperature (Elshawadfy et al.2014).
For more advanced applications, such as DNA sequencing, archaeal DNA pols were not used initially, due to their poor ddNTP/dNTP selectivity and their intrinsic 3΄-5΄ exonuclease activity. However, several archaeal pols were engineered for efficient usage of ddNTPs and loss of, or reduced, 3΄-5΄ exonuclease activity, such as variants of the DNA pols from T. litoralis, P. furiosus, Thermococcus JDF-3 and Thermococcus 9°N-7 (Gardner and Jack 1999; Evans et al.2000; Arezi, Hansen and Hogrefe 2002). Additionally, the Themococcus JDF-3 DNA pol is significantly more processive than Pfu and Vent DNA pols (Arezi, Hansen and Hogrefe 2002). This DNA pol was further engineered to incorporate fluorescently tagged dNTPs. Another variant of the archaeal DNA pol from Thermococcus 9°N-7, commercially available as Therminator DNA pol, has enhanced the ability to incorporate modified nucleotides, e.g. dideoxynucleotides, ribonucleotides and acyclonucleotides (Gardner and Jack 2002). This Therminator DNA pol was further engineered by amino acid substitutions to make it more versatile and applicable to use for the sequence-by-synthesis method (Ju et al.2006). Next-generation sequencing (Metzker 2010; Mardis 2011) requires pols with higher efficiency. To this end, a variant of T. kodakarensis DNA pol was engineered for one-step RT-PCR, a technique not widely used with archaeal pols due to their low cDNA synthesis activity. This variant is stable up to 100°C and is sensitive enough, compared to the retroviral RT, to be used in the current RT-PCR conditions (Okano et al.2017). In general, archaeal DNA pols are thermostable and exhibit high fidelity, such that they will be continue to be targets for use in next-generation DNA sequencing (Pascal et al.2006).
Ligases
DNA and RNA ligases catalyze the formation of the phosphodiester bonds between the opposing 5΄ phosphate and the 3΄ hydroxyl termini of nucleic acids, and are an essential enzyme for biological processes, e.g. DNA replication, DNA recombination, genes rearrangement and DNA/RNA repairs. To date, approximately 25 archaeal ligases have been characterized (Tomkinson et al.2006), with structures of these enzymes reported from Ar. fulgidus (Kim et al.2009), P. furiosus (Nishida et al.2006; Tanabe et al.2014), Su. solfataricus (Pascal et al.2006), T. sibiricus (Petrova et al.2012b), Thermococcus sp. 1519 (Petrova et al. 2012a) and Sulfophobococcus zilligii (Supangat et al.2010). Ligases are used for cloning, plasmid/fosmid library construction, Gibson assembly and plasmid-based site-directed mutagenesis for re-circularizing linear DNA. Alternatives to Taq ligase for Gibson Assembly (Gibson et al.2009) were sought that are capable of ligating double-stranded, cohesive- and blunt-ended fragments. This effort identified ligases from Aeropyrum pernix (Jeon and Ishikawa 2003), Staphylothermus marinus (Seo et al.2007), Themococcus sp. 1519 (Smagin et al.2008) and T. fumicolans (Rolland et al.2004) that were able to join cohesive-ended fragments, and ligases from St. marinus and T. fumicolans that can ligate blunt-ended fragments. Thermococcus sp. 1519 ligase connects long cohesive ends (about 12-nucleotide overhangs), but not shorter cohesive ends (4-nucleotide) (Smagin et al.2008). Since this enzyme is also most active at 60°C–70°C, it is a good candidate to replace Taq ligase in Gibson Assembly. In another application involving the ligase chain reaction for detection of single nucleotide polymorphisms, hyperthermophilic ligases are used, since they are stable to survive the denaturing step at 95°C in the cycling protocol. For example, the DNA ligase from St. marinus has a half-life of almost 3 h at 100°C and can also catalyze both cohesive- and blunt-ended fragments (Seo et al.2007). To date, only the DNA ligase from P. furiosus has been the focus of developing enhanced activity (Tanabe et al.2012, 2014). Based on its structure, this ligase was modified by mutagenesis, particularly at the C-terminal helix that interacts with the oligonucleotide binding domain (OBD) and the adenylation domain (AdD), to enhance activity and a broadening of its temperature range from 20°C to 80°C (Tanabe et al.2012). The mutant achieved maximum amplification of ligated DNA product after only 3 cycles, whereas the wild-type enzyme required 10 cycles (Tanabe et al.2012). This result demonstrated that a rational design approach, involving the C-terminal helix with the OBD and the AdD, could be generalized to other archaeal ligase systems.
RNA ligases that are involved in RNA repair, splicing and editing are also important in molecular biotechnology. In fact, T4 RNA ligases I and II are the essential components for a subset of rapid amplification of cDNA, RNA labeling, and more recently, the preparation of miRNA sequencing libraries (Chambers and Patrick 2015). The first characterized archaeal RNA ligase from P. abyssi (Brooks et al.2008) was active on single-stranded RNA substrates only. The RNA ligase Methanothermobacter thermautotrophicus (Torchia, Takagi and Ho 2008) can adenylate both single-stranded RNA and DNA, making it useful for 5΄-adenylating single-stranded DNA adapters for construction of miRNA sequencing library. Usually, T4 DNA ligase is used for this 5΄ adenylation step (Chiuman and Li 2002). However, the adenylated product yield is low and the process is expensive. In contrast, the RNA ligase from Me. thermautotrophicus is more efficient (Sriskanda et al.2000) and currently available commercially. Furthermore, it was also shown that a single point mutation (K97A) completely stripped its adenylation activity, while retaining the ability to form phosphodiester bonds (Zhelkovsky and McReynolds 2012). Another archaeal RNA ligase, from T. kodakarensis (KOD), has a template dependency that is better than that of T. thermautotrophicus RNA ligase (Zhang and Tripathi 2017). KOD RNA ligase also possesses mismatch specificity, which is useful for RNA sequencing, since reduced background ligation will improve the fidelity of sequencing reads significantly and is tolerant of blood protein contaminant. In fact, KOD RNA ligase retains ligation activity in the presence of up to 5% human serum (Zhang and Tripathi 2017). Even though no structural information is available for KOD RNA ligase, it has great promise for use in RNA detection (as it is demonstrated to detect Ebola RNA transcripts), RNA modification and sequencing.
Inteins
Inteins (internvening protein) are genetic elements with intervening sequence that are capable of self-splicing post-translationally and many also contain exonuclease components capable of invading DNA (Mills, Johnson and Perler 2014; Shah and Muir 2014). It is a naturally occurring biochemical process that mediates post-translation conversion of a precursor polypeptide into a functional protein. Although inteins are found in all three domains of life, as well as in viruses and phages, they are present with the highest frequency in archaea (Novikova, Topilina and Belfort 2014). Additionally, most intein-containing proteins are involved in DNA-related processing, e.g. polymerases, helicases and topoisomerases (Novikova, Topilina and Belfort 2014). Initially, inteins were used for development of self-cleaving affinity tags to use in protein expression, releasing the tagless target upon intein splicing (Chong et al.1998). Ideally for this application, the intein will cause minimal cleavage during protein expression, but undergo cleavage rapidly and with high specificity to release the tagless protein once purification is completed. Understanding the mechanisms of intein function is essential. For example, the cleavage of RadA intein from P. horikoshii was shown to be highly specific and rapid with addition of single-stranded DNA (Topilina et al.2015). Inteins found in the MoaA precursor protein in P. abyssi and a radical S-adenosylmethionine domain protein of Ar. profundus are trapped in their precursor proteins by formation of disulfide bond with the internal cysteine residues involved in the splicing mechanism; as such, cleavage will not occur until a thiol is added (Callahan, Stanger and Belfort 2013). This insight inspired development of artificial inteins that utilize the disulfide bond to prevent premature cleavage. The split PI-Pfu intein from P. furiosus, though not used in protein purification, was the basis for a biosensor that can be expressed inside living cells. This intein was used to cyclize a green fluorescent protein in vivo, which is more stable than its linear counterpart (Iwai, Lingel and Pluckthun 2001). With the development of genetics systems for extremely thermophilic archaea, the prospect of understanding intein function has improved, which will help to unlock novel applications of inteins for biotechnology.
ENZYMES FOR BIOTRANSFORMATIONS
Once genome sequences of extremely thermophilic archaea became available in the mid-1990s (e.g. for Met. jannaschii and P. furiosus) (Bult et al.1996; Robb et al.2001), the opportunity arose to identify thermophilic homologs of industrially relevant, mesophilic enzymes, already in industrial use, from mesophilic sources arose (Adams and Kelly 1998). As a consequence, there were many reports focused on potential applications based on recombinant versions of these enzymes produced in mesophilic hosts (Adams, Perler and Kelly 1995). However, to date, there have not been many large-scale commercial processes using enzymes from extremely thermophilic archaea. There are several barriers to overcome, including the challenge of replacing existing enzymes entrenched in mature bioprocesses. The advent of metabolic engineering with extremely thermophilic archaea and new emerging concepts for high-temperature bioprocessing could provide further opportunities for commercial applications of enzymes from extremely thermophilic archaea. (see Table 4).
Table 4.
Extremely thermophilic archaeal biotechnology-related biotransformations.
| Enzyme | Origin | Application | References |
|---|---|---|---|
| Beta-Glucosidase | Pyrococcus, Sulfolobus, Thermococcus spp. | Flavanone and ginsenoside production; lactose removal from milk; steryl glucoside removal from biodiesel | Li et al. (2013); Shin, Nam and Oh (2013); Peiru et al. (2015) |
| Lactonase | Sulfolobus solfataricus | Quorum-sensing disruption, detoxification of organophosphates | Del Giudice et al. (2016); Remy et al. (2016) |
| Superoxide reductase | Pyrococcus furiosus | Reduced mortality of plants and insects to heat stress | Im et al. (2009); Geng et al. (2016); Jiang et al. (2017) |
| Ferritin | Pyrococcus furiosus | PO4, AsO4 removal from water; enzyme immobilization | Sevcenco et al. (2015); Tetter and Hilvert (2017) |
| Isoamylase | Sulfolobus tokodaii | Enhanced productivity from amylopectin component of starch for chemical and energy production | Cheng et al. (2015); You et al. (2017) |
| Glycolytic enzymes | Thermococcus kodakarenesis; Archaeoglobus fulgidus | Cell-free metabolic engineering for energy or chemical production | Fujisawa et al. (2017); You et al. (2017) |
| Transcription factors | Sulfolobus islandicus | Identification of anti-microbial and anti-cancer peptide; serve as potential templates for other medical uses | Notomista et al. (2015); Gaglione et al. (2017) |
| Chaperones | Pyrococcus furiosus; Methanocaldococcus jannaschii | Prevent protein misfolding in the presence of translation inhibitors; template for novel protein scaffolds | Peng et al. (2012), Glover and Clark (2015); Glover et al. (2016) |
| PfpI | Pyrococcus, Thermococcus | Homolog to protease important in human diseases | Halio et al. (1996); Wilson et al. (2003); Larson and McPherson (2017); Blumentals, Robinson and Kelly (1990) |
| Carbamate kinase | Thermococcus barophilus | N removal from wastewater | Hennessy et al. (2017) |
| Fuel cell | Pyrococcus furiosus | Bioelectricity | Sekar et al. (2017) |
| Xylanase | Thermococcus | Biomass conversion | Gavrilov et al. (2016) |
| Chitinase | Thermococcus sp. | Chitin hydrolysis | Horiuchi et al. (2016) |
| Alcohol dehydrogenase | Pyrococcus furiosus | Cofactor specificity | Solanki, Abdallah and Banta (2016) |
| Amylase | Thermococcus sp. | Commercial enzyme | Richardson et al. (2002) |
Protein expression
Proper folding and assembly of proteins from extreme thermophiles are presumably best performed in the native organism or in a closely related one. This had not been possible prior to the development of genetic systems for extremely thermophilic archaea, but there have been significant developments in this area (see section on Genetics). For example, a subunit of a chitinase from T. kodakarensis was overexpressed leading to an increase in chitinase activity over the protein when produced in a mesophilic host (Takemasa et al.2011). A His6-tagged P. furiosus RNA polymerase was overexpressed with a gluconeogenic promoter, demonstrating inducible protein expression (Waege et al.2010). An NADPH-dependent cytoplasmic [NiFe]-hydrogenase was overexpressed in P. furiosus, and with the addition of a His9-tag, the protein was purified to homogeneity by a single affinity chromatography step (Chandrayan et al.2015). With improvements in molecular genetics tools for extremely thermophilic archaea, the day may come when these microorganisms are utilized as overexpression hosts for proteins that require high temperatures for proper folding and function.
Glycoside hydrolases
Glycosid hydrolases (GHs) from extremely thermophilic archaea were among the first enzymes from these microorganisms to attract biotechnological interest, primarily due to their intrinsic thermostability, a desirable feature for many applications. For example, with an eye toward starch processing, many α-amylases have been characterized from Thermococcus, Pyrococcus and Sulfolobus species as possible replacements for the widely used α-amylase from Bacillus licheniformis (Brown, Costantino and Kelly 1990; Chung et al.1995; Dong et al.1997; Jorgensen, Vorgias and Antranikian 1997). While many of these processes operate at pH 4.5–5.5, the pH optimum of B. licheniformis α-amylase is around 6, requiring pH adjustment (Chung et al.1995; Shaw, Bott and Day 1999). However, a pH-stable and thermostable α-amylase was developed by using a gene shuffling approach based on the genes encoding three such enzymes from primary enrichments of deep-sea vent samples, which had greater than 85% sequence homology to those found in the Thermococcales (Richardson et al.2002). One identified variant had improved saccharification properties at pH 4.5 and 90°C for starch hydrolysis, which compared favorably to the B. licheniformis α-amylase. Ultimately, this became the basis for FuelZyme®, which was one of the first large-scale biotechnological products, other than molecular biological enzymes, based on extremely thermophilic archaea. Amylolytic enzymes continue to be an active focus of both discovery and improvement. Multi-functional hydrolases, such as the type III pullulanase from T. kodakarensis, which has a hydrolytic activity to both α-1,4 and α-1,6 bonds, have promise as an industrial enzyme since its thermostability and pH optimum are consistent with traditional starch processing conditions (Ahmad et al.2014). A key limitation for any industrial enzyme candidate is the inability to obtain multigram per liter expression levels, and this has been a concern for archaeal enzymes. To address this, efforts for improving expression of an α-amylase from P. furiosus in mesophilic organisms, such as B. subtilis (Jorgensen, Vorgias and Antranikian 1997), E. coli (Wang et al.2007) and B. amyloliquefaciens (Wang et al.2016), have been reported.
There has been interest in hydrolyzing polysaccharides other than starch at elevated temperatures. Microcrystalline cellulose degradation at high temperatures by enzymes is a long-sought property for lignocellulose processing (Blumer-Schuette et al.2008). An archaeal consortium growing on a filter paper at 90°C was isolated and the associated metagenome encoded a multidomain GH with a family GH5 domain, that is similar to an enzyme encoded in the extremely thermophilic bacterium, Caldicellulosiruptor saccharolyticus (e.g. Csac_2528) (Graham et al.2011). However, the metagenome had no GH48 or carbohydrate binding modules from family 3 domains, normally associated with crystalline cellulose degradation, and filter paper dissolution occurred relatively slowly at 90°C.
Chitin is the second most abundant polysaccharide after cellulose, but its use as a feedstock is limited by the inability to hydrolyze it into simple sugars. Given the marine biotopes of many extremely thermophilic archaea, it is not surprising that several chitinases have been characterized from these microorganisms (Tanaka et al.1999, 2003; Gao et al.2003). Recently, the first thermophilic chitinase able to hydrolyze the reducing end of chitin was reported in T. chitonophagus, potentially expanding the opportunities for using this material (Horiuchi et al.2016). Thermococcus sp. strain 2319 × 1, a hyperthermophilic archaeon isolated from a primary enrichment from a tidal vent off the coast of Kunashir Island, can grow on xylan as the sole carbon source, although the enzymatic basis for this capability is not yet clear. However, a novel multi-domain GH was identified in this species, containing GH5-GH12-GH12-CBM2-CBM2 domains, that had low activity on xylan but was highly active on mixed β-1,3 and β-1,4 glucans (Gavrilov et al.2016).
Other applications involving GHs involve in vivo biocatalysis. Recently, a P. furiosus-based microbial fuel cell demonstrated electricity production on maltose at a power density of 225 mW m−2 (Sekar et al.2017). While the power density is low, this proof of concept could potentially lead to exploiting the GH inventory from extremely thermophilic archaea to generate electricity from complex polysaccharides.
Emerging applications of β-glucosidases
β-Glucosidases catalyze the hydrolysis of β-1,4 linkages in glucans and have been well characterized in Pyrococcus, Thermococcus and Sulfolobus species (Cady et al.2001; Lebbink et al.2001; Moracci, Ciaramella and Rossi 2001). While these enzymes have applications in biomass processing, applications to food processing and biofuel production have emerged. The CelB from P. furiosus produced in Pichia pastoris hydrolyzed 90% of the lactose in milk within 30 min at 65°C (Li et al.2013). Recently, exo-β-glucosidases have been shown to have broad specificity for glycoconjugated substrates that could be converted into products that improve human health (Shin, Nam and Oh 2013; Shin et al.2015, 2017). Production of flavenoids and ginsenosides from citrus and ginseng extracts, respectively, has been demonstrated using the β-glucosidases from P. furiosus and Su. solfataricus. To overcome kinetic limitations, semirational design of the β-glucosidase from Su. solfataricus identified a W361F mutation that improved activity on ginseng extracts for compound K conversion by over 4-fold (Shin et al.2017). Furthermore, this mutant also had a higher activity on flavonoids, indicating that promiscuity of this engineered hydrolase can be exploited for several nutritional products (Shin et al.2017). Steryl glucosides (SGs) are insoluble contaminants made as a by-product of biodiesel that can cause it to fail quality tests. The β-glucosidase from T. litoralis removed nearly all of the SGs within 2 h (Peiru et al.2015), suggesting that enzymatic treatment, instead of distillation, could be used to reduce contaminant levels during biodiesel purification.
Wastewater treatment and organophosphorus decontamination
Several environmental applications for extremely thermophilic archaeal enzymes are emerging, including detoxification of organophosphorus compounds (Hiblot et al.2012a,b; Del Giudice et al.2016; Remy et al.2016), phosphate and arsenate removal in water treatment (Sevcenco et al.2015), and nitrogen removal from wastewater (Hennessy et al.2017). Carbamate kinases have gained interest for their ability to sequester ammonia and carbon dioxide from wastewater as carbamate phosphate, which can serve as a building block molecule for chemical synthesis. Carbamate kinases from T. barophilus and T. sibiricus had high activity in alkaline pH, indicating their potential applications for enhanced CO2 sequestration in ammonia (Hennessy et al.2017). Ferretin serves as an iron storage protein that is found in all three domains of life. Ferritin from P. furiosus has been demonstrated as a cage for iron nanoparticle formation, which has a high affinity for phosphate and arsenate (Sevcenco et al.2015). Furthermore, ferritin from Ar. fulgidus was shown to act as a cage for GFP fusion proteins (Tetter and Hilvert 2017), which holds promise for pharmaceutical applications as well as immobilization of polishing enzymes in water treatment. In addition, P. furiosus was recently shown to contain a completely new type of iron-storage protein termed IssA (for iron-sulfur storage protein A) (Vaccaro et al.2017). IssA contains iron and sulfur in the form of thioferrate, an inorganic anionic polymer previously unknown in biology. IssA forms nanoparticles reaching 300 nm in diameter and is the largest natural metalloprotein complex known. IssA nanoparticles are visible by electron microscopy as electron-dense bodies in the P. furiosus cytoplasm. Purified nanoparticles appear to be generated from 20 nm units containing ∼6400 Fe atoms and ∼170 IssA monomers. Whether IssA has any biotechnological relevance remains to be seen.
Other proteins from extremely thermophilic archaea with biotechnological importance include SsoPox, a bifunctional lactonase and phosphotriesterase isolated from Su. solfataricus that has been shown to have broad specificity (Hiblot et al.2012a,b). While the wild-type phosphotriesterase activity is low (Merone et al.2005), several mutants with improved activity have been developed (Merone et al.2010; Del Giudice et al.2016; Remy et al.2016). SsoPox triple mutant C258L/I261F/W263A had a 300-fold increased activity on paraoxon, a highly toxic metabolite of the organophosphate insecticide parathion, compared to wild-type Sso Pox, although the Topt of the mutant decreased from 80°C to 65°C (Del Giudice et al.2016). This mutant was able to quickly hydrolyze paraoxon from cotton tissues and the surface of apples without detergents, which makes it a promising enzyme for detoxification of agricultural products and chemical warfare agents. A quadruple mutant of SSo Pox was shown to have over 2000-fold higher activity on parathion compared to wild type, furthering the substrates on which mutagenized enzymes are active (Jacquet et al.2017). Additionally, mutagenized versions of SsoPox have been shown to retain their activity after sterilization, exposure to solvents and immobilization, which can expand the possible applications and processing conditions this enzyme can tolerate (Remy et al.2016). Furthermore, the W236I mutant was shown to have its lactonase activity restored by exposure to supernatants from biofilm-forming bacteria after heat inactivation. This could expand the use of SsoPox into biofilm disruption in pipelines and as a potential antimicrobial agent (Remy et al.2016).
Crop stress protection
As global temperatures increase, agricultural products must be engineered to mitigate drought and heat stressors to maintain yields. Heat stress induces the formation of reactive oxygen species (ROS). While ROS function as signaling molecules, they become toxic to cells in high concentrations due to the inability to manage the associated oxidative stress (Mittler 2002). A new type of oxidative stress was discovered in P. furiosus, which was subsequently found in virtually all anaerobes, involving the enzyme superoxide reductase (SOR) (Jenney et al.1999). In contrast to superoxide dismutase, SOR converts O2− into H2O2 without generating oxygen, an important feature for anaerobic organisms. The gene encoding the P. furiosus enzyme (PfSOR) has been overexpressed in tobacco cell culture (Im et al.2005), model and non-model plant systems (Im et al.2009; Geng et al.2016), and silkworms (Jiang et al.2017). Progressively increasing heat stress, up to 40°C for 24 h, on Cornus canadensis with PfSOR did not adversely affect growth or recovery from heat stress. Furthermore, there was less proline accumulation and lipid peroxidation in transgenic plants, indicating PfSOR can enhance ROS scavenging in non-model plants (Geng et al.2016). Overexpression of PfSOR in Bombyx mori resulted in a 30% decrease in mortality after a 44-h exposure to heat stress of 35°C without disruption to the cocoon production rates. Additionally, the transgenic silkworms with PfSOR showed delayed mortality when subjected to starvation conditions (Jiang et al.2017).
Biomedical applications
Applications related to proteases, chaperones and transcription factors from extremely thermophilic archaea are of increasing interest due to their potential biomedical implications. Sometimes features in extremely thermophilic archaea have wide-ranging evolutionary relationships with less thermophilic organisms and cells. For example, P. furiosus protease I (PfpI) is a cysteine protease that has been grouped into the DJ-1/ThiJ/PfpI superfamily (Blumentals, Robinson and Kelly 1990; Halio et al.1996, 1997; Bandyopadhyay and Cookson 2004; Larson and McPherson 2017) of relevance in human biology and disease. Mutations in, or downregulation of, the eukaryotic analog of PfpI, DJ-1, causes familial Parkinson's disease (Wilson et al.2003; Olzmann et al.2004), tumors (Le Naour et al.2001) and infertility (An et al.2011).
By preventing misfolding, thermostable chaperones have the potential to serve as therapeutics in protein aggregation diseases (Glover and Clark 2015; Peng et al.2017b). Hsp60, prefoldin and sHsp chaperones from P. furiosus have been expressed in E. coli to confer short-term resistance to translation inhibiting aminoglycoside antibiotics (Peng et al.2017b). Prefoldin and Hsp60 improved growth of E. coli in the presence of streptomycin due to a reduction in intracellular aggregation of misfolded proteins. Furthermore, the γ-prefoldin from Met. jannaschii forms highly stable filamentous structures with potential uses in tissue engineering and drug delivery (Glover et al.2016). Archaeal transcription factors can serve as templates for antimicrobial peptides, such as has been demonstrated for the VLL-28 peptide derived from the Stf76 transcription factor from Su. islandicus (Notomista et al.2015). This peptide was recently shown to selectively induce apoptosis in murine fibroblasts and human tumor lines, expanding the antimicrobial properties to a potential cancer therapeutic (Gaglione et al.2017).
In vitro biotransformations
As mentioned previously, enzymes from extreme thermophiles have also been used to construct components of in vitro systems that convert mixtures of simple (xylose, glucose) or complex sugars (starch, corn stover) to useful products with high productivities and yields. Using thermostable GHs with glycolytic enzymes has led to cell-free starch conversion for novel applications including production of sugar alcohols, such as myo-inositol (Fujisawa et al.2017; You et al.2017) and direct production of energy for enzymatic fuel cells (Zhu et al.2014; Cheng et al.2015). For all of these applications, the process is essentially the same. α-1,4 linkages of amylodextrin are broken by an α-glucan phosphorylase to produce glucose-1-phosphate. A phosphoglucomutase converts glucose-1-phosphate to glucose-6-phosphate. For energy production, glucose-6-phosphate is oxidized by NAD+ and G6P dehydrogenase. NADH is then oxidized by diaphorase which transfers electrons to the anode of the fuel cell (Zhu et al.2014; Cheng et al.2015). The proof-of-concept system yielded a consistent power density of 0.32 mW cm−2 over a 60-h period (Zhu et al.2014). Isoamylase from Su. tokodaii was added to debranch amylopectin, resulting in a doubling of the power output from the starch fuel cell (Cheng et al.2015). For cell-free myo-inositol production, glucose-6-phosphate is then converted to myo-inositol-3-phosphate by inositol synthase, then dephosphorylated to myo-inositol. Fujisawa et al. (2017) used the maltodextrin phosphorylase and phosphoglucomutase from T. kodakarensis and the inositol synthase from Ar. fulgidus . A similar pathway was used with the addition of both a 4-α-glucanotransferase from T. litoralis to remove free glucose and an isoamylase from Su. tokodaii, thereby eliminating the need to continuously add NAD+ and facilitating scale-up (You et al.2017).
While many archaeal enzymes can be directly used as components of in vitro pathways, engineered variants can provide more flexibility for in vitro pathway design. For example, the AdhD from P. furiosus preferentially uses NAD(H) over NADP(H) in the oxidation of secondary alcohols and reduction of ketones (Machielsen et al.2006). AdhD is active on numerous secondary alcohols and ketones, and is able to reduce ketones enantioselectively (Machielsen et al.2006). Modification of the cofactor binding pocket allowed for nicotinamide mononucleotide to serve as the cofactor in an enzymatic fuel cell, resulting in a 40% increase in current density (Campbell et al.2012; Solanki, Abdallah and Banta 2016). Because of their thermostability, ability to selectively synthesize chiral alcohols and capacity to use less expensive electron acceptors, engineered AdhD variants have applications in the synthesis of pharmaceuticals and speciality chemicals (Machielsen et al.2006; Solanki, Abdallah and Banta 2016).
GENETICS
Significant strides have been made to develop and improve molecular genetic techniques for extremely thermophilic archaea in the past decade. At present, genetic manipulations have been performed in 10 such archaea (see Table 5), including crenarchael Metallosphaera and Sulfolobus species and euryarchaeal Thermococcus and Pyrococcus species. While extremely thermophilic methanogenic archaea (e.g. Methanothermus fervidus, Methanocaldococcus jannaschii) are also of interest for biotechnological applications, no genetic systems have been developed, to date, in these species. Instead, archaeal methanogen engineering efforts have focused on mesophilic Met. maripaludis (Moore and Leigh 2005; VanDyke et al.2009) and Methanosarcina species (Pritchett, Zhang and Metcalf 2004; Buan, Kulkarni and Metcalf 2011; Mondorf, Deppenmeier and Welte 2012; Shea et al.2016), which are outside the scope of this review. Of the extremely thermophilic archaea with functional genetic systems, those of P. furiosus, T. kodakarensis, Su. solfataricus and Su. acidocaldarius stand out as being highly tractable model systems. The general molecular genetic strategies and techniques used in these systems are often able to be extended to related species. These systems also demonstrate methods for overcoming the challenges related to manipulating archaea in general. Thus, the proven methodologies and metabolic engineering successes in these model species will facilitate the development and improvement of other molecular genetic systems in a wider array of extremely thermophilic archaea. Additionally, new technologies and methodologies (e.g. DNA synthesis, transposon mutagenesis, CRISPR-Cas) are entering the molecular genetics toolbox for these organisms, enabling further advancements. Vector designs and workflow for genetic engineering, including many of the genetic tools (shuttle vectors, promoters, selectable markers, etc.), have been reviewed previously (Leigh et al.2011; Atomi, Imanaka and Fukui 2012; Farkas, Picking and Santangelo 2013; Loder et al.2017; Peng et al.2017a). Here, we will focus, in general, on the challenges for developing robust genetic tools and systems in extremely thermophilic archaea. Recent discoveries and technologies enabled by genetics in these microorganisms will also be discussed.
Table 5.
Reported genetic transformations with extremely thermophilic archaea.
| Organism | Topt | Uracil / 5-FOA | Tryptophan / 6-MP | Simvistatin | Agmatine | Hygromycin | Other selection methods | References |
|---|---|---|---|---|---|---|---|---|
| Metallosphaera sedula | 75°C | X | Huber et al. (1989)c, Maezato et al. (2012)d | |||||
| Sulfolobus acidocaldarius | 80°Cb | X | X | Alcohols, | Brock et al. (1972); Grogan (1989)c, Aravalli and Garrett | |||
| β-galactosidase screen | (1997); Grogan, Carver and Drake (2001); Kurosawa and Grogan (2005); Berkner et al. (2007, 2010); Wagner et al. (2009, 2012); Ellen, Albers and Driessen (2010); Suzuki and Kurosawa (2017)d | |||||||
| Sulfolobus islandicus | 78°Cb | X | X | X | Zillig et al. (1993)c; Deng et al. (2009); Peng et al. (2012); | |||
| Zheng et al. (2012); Zhang et al. (2013)b | ||||||||
| Sulfolobus solfataricus | 80°Cb | X | X | Lactose | Zillig et al. (1980); Grogan (1989)c, Cannio et al. (1998); | |||
| Jonuscheit et al. (2003); Worthington et al. (2003); Schelert et al. (2004); Albers et al. (2006); Berkner et al. (2007); Albers and Driessen (2008); Zhang et al. (2013)d | ||||||||
| Thermococcus kodakarensis a | 85°C | X | X | X | X | Arginine/ | Atomi et al. (2004)c, Sato et al. (2003, 2005); Matsumi | |
| citruline, β-glycosidase screen | et al. (2007); Fukuda et al. (2008); Santangelo, Cubonova and Reeve (2008, 2010); Yokooji et al. (2009); Santangelo and Reeve (2011); Hileman and Santangelo (2012)d | |||||||
| Thermococcus barophilus | 85°C | X | X | Histidine | Marteinsson et al. (1999)c; Thiel et al. (2014)d | |||
| Thermococcus onnurineus | 80°C | X | Bae et al. (2006); Lee et al. (2008)c; Kim et al. (2013)d | |||||
| Pyrococcus abyssi | 100°C | X | Erauso et al. (1993)c; Lucas et al. (2002)d | |||||
| Pyrococcus yayanosii | 98°C | X | X | Zeng et al. (2009); Birrien et al. (2011)c; Li et al. (2015)d | ||||
| Pyrococcus furiosus a | 100°C | X | X | Fiala and Stetter (1986)c; Waege et al. (2010); Lipscomb | ||||
| et al. (2011); Farkas et al. (2012)d |
Genetic manipulation workflows at high temperatures
The workflow of genetic manipulations in extremely thermophilic archaea varies slightly depending on the manipulation being performed (chromosomal deletion or insertion), whether the final modification is marked with a selectable marker and whether the host acceptor strain can be transformed with linear or circular DNA. For most applications, genetic manipulations to the chromosome are performed in a two-step process. First, an acceptor strain is transformed with a circular DNA construct bearing a selectable marker and regions of homology to the chromosome. An intermediate strain, which has taken up this DNA by homologous recombination, is generated and identified with a selectable marker out of the population. The second step involves counterselection for loss of the marker, resulting in either generation of the chromosomal modification or reversion to the parent genotype. Additional screening is required to identify the genetically modified strain from the revertant strain population. A final markerless strain allows for the same marker to be used iteratively to make multiple chromosomal modifications to a strain. For transformations involving linear DNA, where there is a need to recycle the selectable marker, a short pair of homologous DNA regions are placed on either side of the marker. After insertion of the DNA at the target location, the second step counterselects for marker loss or ‘pop-out’ via recombination at these short regions. Extrachromosomal expression of a gene can also be achieved in some genetic systems via use of an autonomously replicating plasmid.
Systems of genetic selection
Selective agents coupled with corresponding selectable genetic markers for positive and negative selection strategies are one of the keystone elements to a successful genetic system. The selection and/or counterselection ability of a marker, the selection stringency and flexibility for selection in different media types determine the utility of the selectable marker for engineering a species. Antibiotics used widely in bacterial genetic systems are largely ineffective in archaea, so other antibiotics have been developed for archaeal genetic systems. In extremely thermophilic archaea, the choice of selection strategy is further limited by the thermal stability of both the antibiotic agent and its corresponding resistance protein; therefore, only a few antibiotic strategies have been used successfully in extreme thermophiles so far. The statin-based antibiotics simvastatin and to a lesser extent mevinolin have been most widely used as positive selective agents, and these target the pathway involved in isoprenoid biosynthesis for archaeal phospholipid production. Overexpressing the gene for 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase confers resistance to these statin antibiotics in several systems: P. furiosus (Waege et al.2010), T. kodakarensis (Matsumi et al.2007), Su. islandicus (Zheng et al.2012). Another antibiotic strategy implemented only in T. kodakarensis involves negative selection on 6-methylpurine (6-MP) for the loss of a host marker gene (TK0664) involved in purine biosynthesis (Santangelo, Cubonova and Reeve 2010). Most antibiotic resistance selection methods offer the advantage of being compatible with use in rich media preparations, simplifying media formulation and potentially improving strain growth.
Because of the limited number of thermally stable antibiotics and their corresponding markers, nutritional selection strategies with auxotrophic mutant acceptor strains and defined media preparations have also been readily employed in extremely thermophilic archaea. Auxotrophic strains can be generated via selection of a random mutant or by targeted deletion employing antibiotic selection. The most widely used nutritional markers are the pyrF or pyrE genes in the uracil biosynthesis pathway, which when deleted produce uracil auxotrophic genetic background strains. These markers are particularly useful as they can be used in both positive and negative selection strategies. Complementation of the auxotrophic strain with the marker restores uracil prototrophy, which can be selected for in defined medium lacking uracil. For strains which require further manipulation with the marker, loss of pyrF or pyrE can be counterselected using 5-fluoroorotic acid (5-FOA), an analog of a uracil biosynthetic pathway intermediate. When both pyrE and pyrF are present, 5-FOA is converted to a compound toxic to the cells. Utilization of 5-FOA in the media allows for selection of those cells that have lost the marker via homologous recombination, and thus are no longer capable of converting to a toxic by-product. These uracil auxotrophy/5-FOA systems have been employed to modify almost all extremely thermophilic archaea to date (see Table 5).
Other nutritional selection auxotrophy/complementation selection strategies have also been employed. The majority of these have been used in T. kodakarensis, including tryptophan (Sato et al.2005; Santangelo, Cubonova and Reeve 2008), arginine/citrulline (Santangelo and Reeve 2011) and agmatine (Santangelo, Cubonova and Reeve 2010). Agmatine-based selection has also been used in Su. solfataricus (Zhang et al.2013), and positive selection with agamatine has the added advantage of being compatible with rich medium preparations. Histidine-based auxotrophy and complementation has been used in T. barophilus (Thiel et al.2014). Of the nutritional selection strategies, the counterselectable pyrF and pyrE markers have a distinct advantage for employing schemes which necessitate recycling of markers for iterative genetic manipulations. Otherwise, application of two different markers, one for positive selection and one for negative selection, can be employed to achieve the same result.
Growth requirement challenges during genetic manipulation
The growth requirements of extremely thermophilic archaea present a challenge for developing a selective system, but also represent a hurdle for achieving clonal isolation of selected variants using solid medium. Agar substitutes such as Phytagel and Gelrite are used to formulate solid medium that remains solid at high temperatures, and glass petri dishes must be used instead of plastic. Incubation of plates under anaerobic high-temperature conditions can be achieved using modified metal ‘paint canisters’. Some anaerobic species, like P. furiosus, tolerate manipulations at room temperature on the bench instead of in an anaerobic chamber. An additional challenge to manipulating these archaea is that colony formation requires incubation periods of several days, regardless of their relatively rapid growth in liquid culture. Pyrococcus furiosus can take 2 to 4 days to grow distinct colonies on plates, while some Sulfolobus species require up to 7 days for colony growth. The time investment in selecting for, and clonally purifying, modified strains on plates is one of the major limitations to rapidly engineering these organisms. This increases the necessity for good transformation efficiency, stringent selection mechanisms and well-understood foundational genetic background strains so genetic manipulations can be accomplished in a deliberate manner as opposed to one that is based on trial and error.
DNA transformation barriers
Overcoming barriers to DNA transformation are also of importance for developing a robust and efficient genetic system. Classical methodologies for transformation of microorganisms including CaCl2 incubation, heat shock and electroporation have all been used with varying success in extremely thermophilic archaea. Recently, a naturally competent P. furiosus strain COM1 was identified (Lipscomb et al.2011; Farkas et al.2012), which is transformable with linear DNA and has transformation efficiencies approaching those of naturally competent bacteria. Thermococcus kodakarensis KOD1 is also naturally competent for uptake of either linear or circular DNA, but with considerably lower transformation efficiencies than P. furiosus COM1 (Sato et al.2003; Hileman and Santangelo 2012).
Additional barriers to transformation are DNA restriction modification systems present in the host organisms. Sulfolobus acidocaldarius restriction enzyme SuaI is known to cut unmethylated CCGG motifs (Prangishvili et al.1985), while Su. islandicus E322S restriction enzyme Suil cuts GCwGC motifs (Sollner, Berkner and Lipps 2006). Plasmid DNA being transformed into Su. acidocaldarius can be methylated in E. coli prior to transformation to prevent degradation of the transformed DNA by SuaI (Kurosawa and Grogan 2005). Deletion of SuaI can also be performed to eliminate the need for methylation prior to transformation of Su. acidocaldarius (Suzuki and Kurosawa 2016). SMRT sequencing technologies, which can detect certain types of DNA methylation, are a valuable tool for determining the native DNA methylation patterns that may need to be overcome for the successful transformation of various species.
Of additional concern in developing genetic systems are species with genetic insertion elements which, under certain conditions, may become mobile, potentially generating random phenotypic effects. Multiple identical insertion elements within a genome can also complicate DNA sequence assembly, and in turn, complicate re-sequencing of modified strains. About 10% of the Su. solfataricus P2 genome is composed of insertion elements, and further, both Su. solfataricus and Su. islandicus have active mobile genetic elements (She et al.2001; Brugger et al.2004; Guo et al.2011). Thus, in the genus Sulfolobus, these two species are less desirable metabolic engineering hosts than Su. acidocaldarius, which does not suffer from the abundance and mobility of various insertion elements.
Developments enabled by improving genetic systems
In the face of formidable barriers previously discussed, genetic systems developed in extremely thermophilic archaea have enabled recent advancements in the understanding of the physiology of these organisms. For example, deletion of reverse gyrase in P. furiosus demonstrated the necessity of this DNA modification protein for growth at temperatures above 95°C (Lipscomb et al.2017a). The genetics of P. furiosus have also enabled in vivo study of PfAgo, an Argonaute DNA-guided nuclease (Swarts et al.2015). This PfAgo protein was recently developed into a biotechnological platform for creating ‘artificial restriction enzymes’ for cleavage of DNA at a programmable site of interest, guided by short DNA pieces (Enghiad and Zhao 2017). This technology could be transformative to classical molecular biology approaches using restriction enzymes, potentially allowing precise control of an enzyme to cut at a specific DNA sequence of interest, programmed by a small guide DNA. Another recent development using P. furiosus genetics was the demonstration of the ability to construct a random mutant library (Guschinskaya et al.2016). For the library construction, P. furiosus genomic DNA was randomly transposed in vitro and these marker-bearing fragments were then transformed into P. furiosus COM1. The ability to perform this mutant library construction is facilitated largely by the relatively high transformation efficiency of the naturally competent P. furiosus COM1 strain. A random mutant library in this archaeon would enable competitive fitness screening, which could help identify genes which positively or negatively affect strain fitness under a variety of conditions (e.g. growth substrates, growth modes, stresses). Genetics and transformation procedures have also enabled the study of the native CRISPR-Cas systems of P. furiosus (Elmore et al.2015; Swarts et al.2015; Shiimori et al.2017) and Sulfolobus species (Deng et al.2013; Peng et al.2015). This understanding of the CRISPR-Cas system of extremely thermophilic archaea could enable future genetic engineering efforts in these species. For example, using a native CRISPR-Cas system that targets mRNA in Su. solfataricus, post-translational gene silencing could be performed with gene knockdown greater than 90% in some cases (Zebec et al.2016). Genetics of T. kodakarensis have recently been used to study factor-independent transcriptional termination (Walker, Luyties and Santangelo 2017), to engineer strains for degrading and metabolizing chitin (Aslam et al.2017), and to study DNA replication (Gehring et al.2017). Other studies in T. kodakarensis have elucidated the endopeptidases which contribute to the maturation of a [NiFe]-hydrogenase in this species (Kanai et al.2017). Recently developed genetics in T. barophilus and P. yayanosii have enabled the study of stress responses and adaptation to piezophilic conditions in these species (Cario et al.2015, 2016; Li et al.2016). And, genetics in Su. acidocaldarius were recently used to better understand a Crenarchaeal system for exchange of DNA (termed Ced), which is involved in DNA import and is induced by UV-light treatment (van Wolferen et al.2016). Better understanding of DNA uptake in the crenarchaea may enable improved genetic transformation techniques for these species. The mechanisms of DNA transfer between archaea, including the Ced system, were recently reviewed (Wagner et al.2017). The genetic methodologies in extremely thermophilic archaea have brought many new insights to these fascinating organisms which would otherwise not have been possible.
It is indeed an exciting time for the study of these archaea, with so many new genetic technologies on the horizon. The discoveries and developments, made in recent years, have been enabled primarily by the increasing tractability of genetics in extremely thermophilic archaea. Work to add new techniques to the molecular genetic toolbox of the most tractable genetic systems, and to extend these techniques to other species of archaea is ongoing. Thus, the future of using molecular genetics in extremely thermophilic archaea for basic science discovery and metabolic engineering is extremely bright.
METABOLIC ENGINEERING
While recruitment of individual genes and pathways from archaea for metabolic engineering applications has been employed, there has been much less done with extreme thermophiles as the platform organisms to date (Zeldes et al.2015). In the last decade, numerous small molecule pathways have been demonstrated at titers of ≤1 g L−1, along with examples of protein overexpression, both homologous and heterologous (see Table 6). Progress in genetic system development has increased the pace at which extremely thermophilic archaea are being utilized as platform organisms, thereby opening up new avenues to metabolic engineering.
Table 6.
Genetically engineered products and protein overexpression involving extremely thermophilic archaea.
| Organism | Topt (°C) | Recombinant outcome | Sources |
|---|---|---|---|
| Sulfolobus solfataricus | 80 | Cellulose degradation with overexpressed endoglucanasea | Girfoglio, Rossi and Cannio (2012) |
| Sulfur oxygenase reductase (SOR)b | Albers et al. (2006); Girfoglio, Rossi and Cannio (2012) | ||
| ABC class Fe-S proteina | |||
| Membrane-associated ATPasesa | |||
| Sulfolobus acidocaldarius | 75 | Manipulate pentose uptake for biomass degradation applications | Wagner et al. (2017) |
| Heat stable green fluorescent protein expressionb | Henche et al. (2012) | ||
| Sulfolobus islandicus | 78 | Protein expressiona | Peng et al. (2012) |
| Thermococcus kodakarensis | 85 | Increased chitinase productionaProteasesa | Takemasa et al. (2011) |
| Increased H2 production via hydrogenase and reductase deletions | Santangelo, Cubonova and Reeve (2011) | ||
| Increased H2 production via overexpression of hydrogenasea | Kanai et al. (2015) | ||
| Thermococcus onnurineus | 80 | Increased H2 production from CO via overexpression of CO dehydrogenasea | Kim et al. (2013) |
| Pyrococcus furiosus | 100 | Lactate (0.3 g L−1) | Basen, Sun and Adams (2012) |
| 3-Hydroxypropionate (0.4 g L−1) | Keller et al. (2013); Lian et al. (2016) | ||
| Ethanol (1.1 g L−1) | Basen et al. (2014) | ||
| Butanol (0.07 g L−1) | Keller et al. (2015) | ||
| Acetoin (0.5 g L−1) | Nguyen et al. (2016) | ||
| H2 production from CO | Schut et al. (2016a) | ||
| Increased cytoplasmic hydrogenase expression | Chandrayan et al. (2012) | ||
| Overexpression of tagged RNA polymerasea | Waege et al. (2010) |
Homologous protein overexpression.
Heterologous protein overexpression.
There are examples of proteins encoded in extremely thermophilic archaeal genomes that have been recruited for moderately thermophilic metabolic engineering purposes. For example, genes encoding enzymes from T. kodakarensis and P. horikoshii were employed to construct a non-ATP-forming chimeric Embden-Meyerhof pathway expressed in the thermophilic bacterium Th. thermophilus (Ye et al.2012). Also, a portion of the archaeal isoprenoid ether lipid biosynthetic pathway was reconstructed in E. coli by expressing four genes from Ar. fulgidus to form digeranylgeranylglyceryl phosphate (Lai et al.2009). Moving forward, cases where the extreme thermophiles themselves will be used as the platform microorganism will be reported, based on the development and application of systems biology-based tools. For example, efforts to develop genome-scale models for archaea have been described that seek to relate growth characteristics to metabolic pathways that have the potential to steer metabolic engineering efforts for substrate utilization and product formation (Ulas et al.2012; Thor, Peterson and Luthey-Schulten 2017).
Core metabolism
Most extremely thermophilic archaea have a modified Embden-Meyerhof central glycolysis pathway which converts glyceraldehyde-3-phosphate (G3P) to 3-phosphoglycerate (3-PG) in a single step via a glyceraldehyde-3-phosphate ferredoxin oxidoreductase. This bypasses two steps normally found in E-M glycolysis, the reduction of GAP via glyceraldehyde-3-phosphate dehydrogenase and ATP production via phosphoglycerate kinase. Thus, the pathway from glucose to pyruvate is a net zero ATP process when considering only substrate phosphorylation. While utilizing such a pathway in vivo is not advantageous for ATP supply of cellular needs, this net zero ATP cycling can be strategic when constructing a pathway in vitro. By constructing an in vitro energy and reducing equivalent neutral pathway with enzymes from the thermophilic bacterium Th. thermophilus and hyperthermophilic archaea T. kodakarensis and P. horikoshii, a stoichiometric amount of lactate was produced from glucose (Ye et al.2012).
Metabolism can be significantly influenced by environmental conditions and stress, especially temperature. Growth of P. furiosus below 77°C resulted in 0.5 g L−1 acetoin while temperatures above 95°C, near optimal for the organism, did not result in detection of acetoin (Nguyen et al.2016). Additionally, a lactate dehydrogenase from a bacterium with optimal growth near 78°C was inserted into the P. furiosus genome under the control of a cold-shock promoter (Basen, Sun and Adams 2012). Approximately 0.3 g L−1 lactate was produced at 72°C while virtually no lactate was detected at 98°C, demonstrating yet another example of temperature related control of metabolism in extremely thermophilic archaea.
Products from biotechnology applications
A limited number of alcohols and other small molecules have been produced within extremely thermophilic archaeal hosts in recent years. In P. furiosus, ethanol was produced at 0.5 g L−1 via reduction of its acetate by-product with an unusual ferredoxin-dependent AOR producing acetaldehyde (Basen et al.2014). The insertion of an AdhA from Thermoanaerobacter species completed the ethanol production via reduction with NADPH. This pathway was also shown to reduce exogenously added organic acids, such as propionate and isobutyrate, producing the corresponding alcohol, demonstrating substrate promiscuity of both the AOR and AdhA. A synthetic pathway was constructed in P. furiosus to produce 1-butanol with four genes recruited from Caldanaerobacter subterraneus and one from each Spirochaeta thermophila and Thermoanaerobacter ethanolicus. Modest titers of 1-butanol (0.07 g L−1) were achieved, yet this is the most complex metabolic pathway engineered into an extreme thermophile to date (Keller et al.2015). Genes for three enzymes, a multisubunit carboxylase and two NADPH-dependent reductases, from the 3-HP/4-HB carbon fixation cycle found in M. sedula were engineered into P. furiosus to produce 3-hydroxypropionate at 0.05 g L−1 (Keller et al.2013). Further development of the pathway led to the addition of a biotin protein ligase, responsible for biotinylation of the carboxylase. A nearly 10-fold increase in titer was achieved with addition to the pathway producing nearly 0.4 g L−1 (Lian et al.2016). When utilizing an enzymatic pathway with activity well below the optimal temperature of the host organism, an operational strategy is necessary to ensure that the host reaches high cell density within a reasonable time frame (see Fig. 4). This was employed for a 3-hydroxypropionate pathway with demonstrated optimal activity at 72°C, nearly 30°C below the optimal temperature of the host, P. furiosus. A bioreactor scale temperature strategy allowed the organisms to multiply in expontential phase to nearly the maximal density (1 × 108 cells mL−1), before the temperature was lowered over a short period of time to 72°C. Prior to the temperature shift, 3-HP was not detected and acetate was the primary fermentation product. Following the temperature shift, 3-HP production commenced after a lag phase lasting approximately 6 h (Hawkins et al.2015). Transcriptomic analysis revealed that the foreign genes were expressed at very high levels during both growth and production phases, as expected given the strong, constituitive promoter used. Central metabolic genes were generally downregulated in the parent strain during the lower temperature production phase. But, surprisingly, these genes were highly transcribed in the recombinant strain during both phases, perhaps reflecting the bioenergetic needs to produce 3-HP (Fig. 4). While the temperature shift strategy presents an opportunity to exploit thermophily for high temperature metabolic engineering platforms, more work is needed to understand the physiological and metabolic consequences of this approach.
Figure 4.
Metabolic engineering temperature shift strategy demonstrated in P. furiosus. (a) Heterologous pathway from M. sedula inserted in P. furiosus; (b) temperature shift strategy with growth phase near Topt of host organism and production phase at Topt of pathway; (c) transcriptomes revealed that central metabolism in recombinant strain was minimally affected by temperature shift compared to parent strain. Mal-CoA = malonyl CoA reductase; Mal SA = malonyl semialdehyde reductase.
VIRUSES
Viruses associated with extremely thermophilic archaea co-exist and proliferate in the same environments as their hosts. Similar to their hosts, they demonstrate marked diversity and a reservoir of many uncharacterized enzymes and proteins (Prangishvili et al.2017). Archaeal-specific viruses are currently classified into 17 families, many containing thermophilic representatives. Many of these viruses have unique and previously unobserved morphologies, ranging from the champagne bottle-like Ampullaviridae (Haring et al.2005) to the spindle-shaped Fuselloviridae and Bicaudaviridae (Krupovic et al.2014) to the ‘tear drop’ shaped Guttaviridae (Mochizuki, Sako and Prangishvili 2011). On the other hand, some archaeal viruses closely resemble those observed in other domains of life. For instance, the globular viruses from Globuloviridae and the filamentous viruses from the families Clavaviridae, Rudiviridae, Lipothrixviridae and Tristromaviridae closely resemble the filamentous viruses seen in bacteria and eukaryotes in structure. However, they are highly divergent with respect to their central components (e.g. utilizing dsDNA versus ssDNA or RNA) (Krupovic et al.2018) and, in the case of the latter two families, a lipid envelope is present, derived from its hosts (Rensen et al.2016; Kasson et al.2017). Finally, there are numerous viruses that are dubbed ‘cosmopolitan’ in nature. This is due to their similarities to the viruses seen in other domains of life and to their ability to infect a multitude of hosts, such as the Turriviridae, which have an icosahedral capsid structure and are known to exclusively infect the Sulfolobales (Veesler et al.2013).
Even though many archaeal morphologies have been discovered, fewer archaeal viruses have been isolated and classified than bacterial viruses (roughly 2% as many) (Ackermann and Prangishvilli 2012), and still this diversity of discovered morphologies continues to grow (Prangishvili et al.2017). Beyond the detectable homologs seen among virus families in the archaea, few if any homology-based connections can be made to the better studied bacterial and eukaryotic viruses (Gliozzi et al.1983; Iranzo et al.2016). Additionally, there are some viruses that lack genes with any homologs (Prangishvili, Garrett and Koonin 2006). The fact that many of these viruses are co-habitating in extremely thermophilic hosts suggests that enzymes, with biotechnological relevance, remain to be discovered.
The robustness of these viral capsids has already inspired some biotech applications for thermophilic viruses. Specifically, the Su. monocaudavirus 1 (SMV1) was recently investigated as a nano-trafficker capable of surviving the acidic environment of the stomach in mice, while avoiding interactions with gut microbes and endothelial cells, and potentially unwanted immune response (Uldahl et al.2017). Furthermore, the unique morphologies of some of these viruses may play a role in their selective uptake in certain types of cells (Uldahl et al.2016). Another application suggested and explored is the ability of these extremely stable capsid proteins to self-assemble, forming novel viral nanoparticle materials (Steinmetz et al.2008).
An underappreciated application of archaea could involve their CRISPR systems, which to date are much less studied in comparison to those present in bacteria. In fact, archaea may be a better reservoir for new CRISPR applications and enzymes given that recent estimates suggest that 70% of archaeal genomes contain CRISPR systems, in comparison to 50% of bacterial genomes (Makarova et al.2011). In contrast, a recent report suggests the numbers are dramatically lower in uncultured prokaryotes (Burstein et al.2016). This higher prevalence appears to be a reasonable outcome of life at higher temperatures, where population sizes are lower, leading to lower viral mutation rates, and a stronger impetus for adaptive immunity; this contrasts well with mesophilic conditions where the arms race is much faster paced and cells may not derive as much evolutionary benefit from squandering resources on a quickly circumvented route to immunity (Weinberger et al.2012; Iranzo et al.2013). Add to this the fact that many archaea appear to have a ‘primed immunity’ fueled by low specificity for mismatches on some protospacers (Maniv et al.2016; Mousaei et al.2016), a not uncommon feature in type I and type III systems, the dominant systems in archaea (Makarova et al.2011).
These systems are more complex in structure (requiring more proteins, some of which are still being characterized and understood); however, in the niche world of extreme thermophiles, some are being utilized for molecular biology pursuits, such as genome editing (Peng et al.2015) and gene regulation (Zebec et al.2014; Li et al.2016) (specifically in the Sulfolobales). Nonetheless, Cas9 from Streptococcus pyogenes has remained a focal point of the development of Cas-mediated genome editing and gene silencing tools, which have revolutionized the field of molecular biology (Donohoue et al.2017). Although one advantage of the type II system is its simplicity (well-characterized activity from a single effector protein module), the lack of a thermally stable system presents challenges in making robust systems that could be used in medical and industrial applications. Thus, some efforts have moved to identifying more thermotolerant Cas9 proteins but thus far the limit has been a protein with short-term stability at a maximum of 70˚C from Geobacillus spp. (Harrington et al.2017). Therefore, the future of CRISPR biotechnological advancement could focus on more thermophilic organisms that appear to be a reservoir of novel CRISPR systems, whose enzymes are waiting to be better characterized for broader application.
LIPIDS
Archaeal lipids differ in structure from their bacterial and eukaryotic counterparts, in that they contain ether linkages along with isoprenoid moieties; the stereochemistry of these linkages is opposite that of their bacterial/eukaryotic counterparts. The structural features of archaeal lipids impart thermal stability and attributes that inspire biotechnological applications. These lipids can be utilized to encapsulate molecules and materials as liposomes or to create monolayer films. Archaeosomes, spherical vesicles comprising archaeal lipids, have been utilized as delivery systems for drugs, genes, antigens and proteins. Archaeal lipids have also been employed to create thin films for sensors and filtration membranes. In archaea, the lipids that comprise their membrane components are a function of the need to survive in such extreme habitats (Konings et al.2002).
Lipids from extremely thermophilic archaea contain C20-C40 isoprenoid chains with linkages to either glycerol and/or nonitol; these backbones may have either polar or non-polar headgroups (Hanford and Peeples 2002). This unique chemistry imparts structural advantages of relevance to biotechnology, including enhanced stability. Such durability and robustness is the result of many attributes including tetraether structure that spans the membrane becomes rigid at high temperature, the transmembrane cyclic structures reduce fluidity of the membrane, the alkyl covalent bonds increases the strength of the membrane at high temperature and the significant amount of glycosyl polar head groups that stabilize the membrane through hydrogen bonding (Ulrih, Gmajner and Raspor 2009). While diethers, such as archaeols, occur in almost all archaea, tetraethers, such as caldarchaeola and calditoglycerocaldarchaeols, occur in methanogenic, thermophilic and psychrophilic archaea (Sprott 1992). Other structures are observed more rarely (Langworthy 1977; De Rosa et al.1983; Gräther and Arigoni 1995). For example, tetraether backbones form monolayer membranes with the different polar head groups on each side (Gliozzi et al.1983; Jacquemet et al.2009). Many different lipid classes are observed in archaea, including phospholipids, phosphoglycolipids, glycolipids, sulpholipids and aminolipids (Jacquemet et al.2009). The architcture of archaeal monolayer membranes creates opportunities for covalent linkage in the middle layer, which protects against membrane lysis. Other noteworthy features of archaeal lipids include β-D-galactofuranosyl units that rapidly hydrolyze and cyclopentane rings increasingly occur in the lipid chains as environmental temperatures increase (Tomoaia-Cotisel et al.1992; Gambacorta, Gliozzi and De Rosa 1995; Itoh et al.2001).
Archaeal lipid structures improve membrane function in a variety of ways that could be of biotechnological benefit. Ether groups increase resistance to hydrolysis in comparison to ester linkages, imparting the toleration of a broader pH range. Branching methyl groups reduce crystallization and membrane permeability. Saturated chains provide protection against oxidative degradation. The stereochemistry of the glycerol backbone provides resistance against attack by phospholipases (Kates 1992). In extreme thermoacidophiles and methanogens, flexible cyclopentane units allow the membrane rigidity to remain constant with changing temperature and glycosylated lipids stabilize the membrane through glycosyl headgroup bonding (Benvegnu, Lemiegre and Cammas-Marion 2008). Large sugar head groups close to the convex surface promote asymmetric orientation to aid in membrane organization, cyclic diethers aid in survival under high pressures, and the structure of the monolayer also reduces permeation of protons and small molecules (Maccioni et al.1995; Patwardhan and Thompson 2000; Gliozzi, Relini and Chong 2001). Archaeal lipids are formed in the absence of cholesterol, and as such can be modified to provide more stability against enzymatic hydrolysis (Szoka and Papahadjopoulos 1980; Patel and Sprott 1999; Réthoré et al.2007; Benvegnu et al.2013). Archaeal lipids can be prepared and stored in the presence of oxygen, sterilized by autoclaving and freeze-dried without degradation (Brown et al.2009; Uhl et al.2016). All of these features combine to make extremely thermophilic archaeal lipids biotechnologically interesting.
Archaeosomes
Archaeosomes are liposomes made of archaeal lipids and are spherical vesicles constructed at least partially of lipid bilayers. Generally defined as vesicles formed by only archaeal lipids, archaeasomes can also be utilized in combination with conventional lipids and thus termed ‘mixed vesicles’. As with liposomes, archaeosomes and mixed vesicles can be formed as either unilamellar vesicles (ULV) or multilamellar vesicles (MLV) (Jacobsen et al.2017). Archaeosomes are typically formed by hydrating dry lipids, stirring the mixture for an extended time and then extruding the mixture through a membrane designed for liposome creation (Parmentier et al.2011a,b). To test their stability in vitro, markers such as 14C-sucrose, 5(6)-carboxyfluorescein and fluorescein isothiocyanate-dextran can be utilized to track their successful migration to the point of interest (Patel et al.2000; Benvegnu et al.2005; Parmentier et al.2011a).
The aforementioned structure of the membrane-spanning tetraethers causes archaeosomes to be more stable and less permeable than other liposomes (Szoka and Papahadjopoulos 1980; Lichtenberg and Barenholz 1988). Unlike other liposomes, which require a large proportion of cholesterol to form, minimal or no cholesterol is needed for archaeosomes (Lichtenberg and Barenholz 1988). Archaeosomes are much more stable in the presence of high temperature, acidic or alkaline pH, phospholipases, bile salts and serum media (Patel et al.2000; Benvegnu et al.2005). They can also be formed in the presence of oxygen at physiological temperatures or lower (Patel and Chen 2010). In vitro and in vivo studies have demonstarted that archaeosomes are safe and non-toxic in mice (Patel and Sprott 1999; Omri, Agnew and Patel 2003). This biocompatibility and the superior stability of archaeosomes makes them a promising candidate for applications in gene and vaccine and drug delivery.
Drug delivery
Archaeal lipids show promise as candidates for improving upon the drug delivery methods developed for liposomes that have demonstrated stability issues. Oral delivery of therapeutic proteins, peptides and small molecules must initially resist the harsh, acidic environment of the digestive tract (Jacobsen et al.2017). Lipids from extreme thermophiles are well suited for applications in such environments as they are derived from organisms whose very survival depends upon maintaining membrane integrity under such conditions (Parmentier et al. 2011a). For example, archaeosomes made from methanogen lipids were used to encase paclitaxel, a small molecule drug utilized in chemotherapy (Alavi et al.2014). The archaeosome treatment was found to increase the therapeutic value of the drug over its standard administration. Archaeosomes, 400 nm in diameter, have been prepared from the extreme thermophile Ae. pernix for encasement of molecules of various sizes (Zavec et al.2014). For delivery of indicator molecules like calcein into epithelial cells, 40% effectiveness has been reported.
Utilization of pure archaeal lipids may be prohibitively expensive, but their improved properties could be advantageous in lower fractions as stabilizers. A mixture of egg phosphatidylcholine (EPC), cholesterol and the total lipid extract (TLE) from Su. islandicus was examined in the presence of simulated intestinal fluid at various taurocholate (a bile salt) levels. The mixture containing 18% TLE from Su. islandicus showed minor reduction in loss of calcein, the fluorescent marker utilized for tracking (Jensen et al.2015). In a study with rats, a statin was encapsulated in a mixture of EPC and specific lipid components derived from Su. acidocaldarius, specifically dipalmitoyl phosphatidylcholine (DPPC) and glycerlcaldityl tetraether (GCTE). Radiolabeled tetraether lipids were incorporated into the archaesome mixture with radioactivity tracked after oral administration as a proxy for liposome degradation. The mixtures with increased GCTE to DPCC ratios outperformed the mixture of EPC and cholesterol. Addition of a synthetic analog of DPPC, produced by adding an amine containing branch to each end to impart a cationic nature, resulted in nearly five times lower plasma membrane concentrations of the radiolabeled statin (Parmentier et al.2011b)
Gene delivery
Many lipid–DNA complexes have been produced for lipofection, or the transfection of DNA into cells through the use of liposomes. However, the lipids employed, such as DOPE or cholesterol, were often unstable and clinically unsatisfactory (Barenholz 2001). The stability and rigidity of archaeosomes could overcome this barrier. Synthetic archaeal lipids are stable enough even for oral delivery, and they have also proven to be effective enhancing lipids for in vitro gene delivery when compared to DOPE and cholesterol (Brard et al.2007). To demonstrate transfection ability, a series of diether and tetraether archaeal-like lipids were synthesized and derivatized with polyethylene glycol and a folic acid group. Utilizing this mixture, a plasmid containing a gene for luciferase (fluorescent reporter protein) expression was transfected into HeLa cells (Laine et al.2008). The lipids derivatized with folate demonstrated improved transfection due to their ability to bind to receptors on the target cells. Thus, synthesized and derivatized archaeal lipids can be functionalized for specific targets in addition to harnessing their inherent improved stability.
Antigen delivery
Antigens delivered within archaeosomes have demonstrated effective immune response and subsequent antibody production, underlining their potential as antigen delivery systems. Phagocytic cells take in archaeosomes more than 3-fold faster than conventional ester-based liposomes (Tolson et al.1996). Utilizing archaeosomes as a delivery mechanism targeting phagocytic antigen processing cells, a superior immune response was demonstrated in mice. The lipid extracts from mesophilic methanogenic archaea, Methanobrevibacter smithii, Methanosarcina mazei and the thermophilic archaeon Thermoplasma acidophilum were utilized to construct separate BSA containing archaeosomes. Each displayed immune responses comparable to the effective but toxic immunopotentiator Freud's adjuvant (Sprott, Tolson and Patel 1997). These archaeosomes have shown good immunostimulation, whether delivered subcutaneously, intramuscularly or intraperitoneally. They are thus excellent candidates for the delivery of vaccines, and have also been successfully used as immunostimulatory carriers for tumor antigen cancer vaccines (Krishnan et al.2003).
Peptide and protein delivery
Archaeal lipids can be used both as a matrix and a delivery system for proteins and peptides. Experiments have shown successful in vitro reconstitution of proteins from their native lipid bilayers into archaeosomes (In’t Veld et al.1992; Elferink et al.1993). After their incorporation into archaeosomes, beef heart cytochrome-c-oxidase and leucine transport systems were active, showing transport functions and the creation of transmembrane potentials. Proton gradients have also been successfully generated by a bacteriorhodopsin in the main phospholipid from Th. acidophilum and coupled to ATP synthesis in an archaeosome (Freisleben et al.1995). These tetraether lipid-based formulations do form a matrix for proton pumps, but the rigidity of the membrane compromises their operation at high tetraether concentration. Isoprenylcysteine carbon methyl transferase activity was also successfully demonstrated in synthetic bipolar archaeosomes (Febo-Ayala et al.2006).
Archaeosomes could be useful in delivering peptide or proteins taken orally since they can withstand the stressful conditions of the stomach and intestines with minimal leakage. MLV liposomes, which retained more than 90% of their contents after 90 min at low pH, are generally more stable than ULV liposomes (Patel et al.2000). The nature of the head groups was also important, with amphiphilic phosphocholine groups contributing most to the stability of the liposome (Benvegnu et al.2005). Small molecules exit the archaeosome more easily than large molecules, such as peptides; this is initially favorable, but could also indicate a high permeability for protons that could denature the enclosed proteins (Parmentier et al.2011b). Mixed vesicles containing archaeal lipids and a bioenhancer, such as cholylsarcosine, octadecanethiol or TPGS 1000, are promising for the delivery of peptides, since they are stable under digestive conditions, non-toxic and safe when delivered both orally and intravenously. Insulin delivery to mice with induced diabetes was effective with archaeosomes made from the lipids of Su. acidocaldarius, producing a decline in glucose (Li et al.2010). While not as effective as intraperitoneally injected insulin, it was more effective than orally delivered encapsulated insulin or insulin enclosed in standard liposomes, which showed no effect.
Archaeolipid films
Archaeal lipids can also be used to make thin films, which can be useful in the creation of sensing elements or membranes in the study of transmembrane systems. Lipids derived from the extreme thermophile, Su. solfataricus, have been employed in conjunction with the cyclic polypeptide valinomycin, a compound highly selective for potassium over sodium ions, to create Langmuir–Blodgett films. These films have applications for potassium ion-selective membranes with applications in sensors for potassium ions (Sprott, Tolson and Patel 1997). The stable bipolar tetraether lipids derived from Su. acidocaldarius are also useful in the creation of free-standing planar membranes on micropores in printed circuit board fluidics (Ren et al.2014). Compared to other membranes, those constructed with archaeal lipids demonstrate higher resistance and better stability, which is useful in studying channel proteins and transmembrane events. This suggests biotechnological uses of tetraether lipids derived from extreme thermophiles in high-throughput drug screening, artificial photosynthesis, microfluidics and membrane-based lab-on-chip applications.
FUTURE PROSPECTS
Once a biological curiosity, the time is now ripe to consider extremely thermophilic archaea for biotechnological applications that go beyond their use as sources of intrinsically thermostable and thermoactive enzymes. In the post-genomics era, new tools have emerged for microbial biotechnology that have been used to examine the biochemical and physiological characteristics of extreme thermophiles, as well as their ecological roles. Molecular genetic systems and tools for several extreme thermophiles have matured to the point that they can be considered as metabolic engineering platforms. Now it is up to the imagination of biotechnologists to find ways to leverage the biological properties that define life at elevated temperatures to create new products and processes that exploit archaeal extreme thermophily.
FUNDING
This work was supported by grants to RMK and MWWA by the US Department of Energy Research ARPA-E Electrofuels Program (DE-AR0000081), the US National Science Foundation (CBET-1264052, CBET-1264053), to RMK by the US Air Force Office of Scientific Research (AFOSR) (FA9550-13-1-0236, FA9550-17-1-0268), and to MWWA by the US Department of Energy (DE-FG05-95ER20175). JMC, CTS and JRC acknowledge support from a US Department of Education GAANN Fellowship (P200A140020, P200A160061), JAC acknowledges support from a US NIH Biotechnology Traineeship (T32 GM008776-16).
REFERENCES
- Abdollahi H, Shafaei SZ, Noaparast M et al. Mesophilic and thermophilic bioleaching of copper from a chalcopyrite-containing molybdenite concentrate. Int J Miner Process 2014;128:25–32. [Google Scholar]
- Ackermann H-W, Prangishvilli D. Prokaryote viruses studied by electron microscopy. Arch Virol 2012;157:1843–9. [DOI] [PubMed] [Google Scholar]
- Adams MWW. Biochemical diversity among sulfur-dependent, hyperthermophilic microorganisms. FEMS Microbiol Rev 1994;15:261–77. [DOI] [PubMed] [Google Scholar]
- Adams MWW, Kelly RM. Enzymes from microorganisms in extreme environments. Chem Eng News 1995;73:32–42. [Google Scholar]
- Adams MWW, Kelly RM. Finding and using hyperthermophilic enzymes. Trends Biotechnol 1998;16:329–32. [DOI] [PubMed] [Google Scholar]
- Adams MWW, Kelly RM. The renaissance of life near the boiling point - at last, genetics and metabolic engineering. Microb Biotechnol 2017;10:37–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adams MWW, Perler FB, Kelly RM. Extremozymes—expanding the limits of biocatalysis. Biotechnology 1995;13:662–8. [DOI] [PubMed] [Google Scholar]
- Ahmad N, Rashid N, Haider MS et al. Novel maltotriose-hydrolyzing thermoacidophilic type III pullulan hydrolase from Thermococcus kodakarensis. Appl Environ Microb 2014;80:1108–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahonen L, Tuovinen OH. Silver catalysis of the bacterial leaching of chalcopyrite-containing ore material in column reactors. Miner Eng 1990;3:437–45. [Google Scholar]
- Ai C, McCarthy S, Eckrich V et al. Increased acid resistance of the archaeon, Metallosphaera sedula by adaptive laboratory evolution. J Ind Microbiol Biot 2016;43:1455–65. [DOI] [PubMed] [Google Scholar]
- Alavi SE, Mansouri H, Esfahani MK et al. Archaeosome: as new drug carrier for delivery of Paclitaxel to breast cancer. Ind J Clin Biochem 2014;29:150–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albers SV, Driessen AJ. Conditions for gene disruption by homologous recombination of exogenous DNA into the Sulfolobus solfataricus genome. Archaea 2008;2:145–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albers SV, Jonuscheit M, Dinkelaker S et al. Production of recombinant and tagged proteins in the hyperthermophilic archaeon Sulfolobus solfataricus. Appl Environ Microbiol 2006;72:102–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albers S-V, Siebers B. The family sulfolobaceae. In: Rosenberg E, DeLong EF, Lory S et al. (eds). The Prokaryotes. Berlin Heidelberg: Springer, 2014, 323–46. [Google Scholar]
- Amend JP, Shock EL. Energetics of overall metabolic reactions of thermophilic and hyperthermophilic Archaea and bacteria. FEMS Microbiol Rev 2001;25:175–243. [DOI] [PubMed] [Google Scholar]
- An CN, Jiang H, Wang Q et al. Down-regulation of DJ-1 protein in the ejaculated spermatozoa from Chinese asthenozoospermia patients. Fertil Steril 2011;96:19–23.e2e12. [DOI] [PubMed] [Google Scholar]
- Aravalli RN, Garrett RA. Shuttle vectors for hyperthermophilic archaea. Extremophiles 1997;1:183–92. [DOI] [PubMed] [Google Scholar]
- Arezi B, Hansen CJ, Hogrefe HH. Efficient and high fidelity incorporation of dye-terminators by a novel archaeal DNA polymerase mutant. J Mol Biol 2002;322:719–29. [DOI] [PubMed] [Google Scholar]
- Aslam M, Horiuchi A, Simons JR et al. Engineering of a hyperthermophilic archaeon, Thermococcus kodakarensis, that displays chitin-dependent hydrogen production. Appl Environ Microb 2017;83:e00280-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astudillo C, Acevedo F. Adaptation of Sulfolobus metallicus to high pulp densities in the biooxidation of a flotation gold concentrate. Hydrometallurgy 2008;92:11–5. [Google Scholar]
- Astudillo C, Acevedo F. Effect of CO2 air enrichment in the biooxidation of a refractory gold concentrate by Sulfolobus metallicus adapted to high pulp densities. Hydrometallurgy 2009;97:94–7. [Google Scholar]
- Atomi H, Fukui T, Kanai T et al. Description of Thermococcus kodakaraensis sp. nov., a well studied hyperthermophilic archaeon previously reported as Pyrococcus sp. KOD1. Archaea 2004;1:263–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atomi H, Imanaka T, Fukui T. Overview of the genetic tools in the Archaea. Front Microbiol 2012;3:337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auernik KS, Kelly RM. Identification of components of electron transport chains in the extremely thermoacidophilic crenarchaeon Metallosphaera sedula through iron and sulfur compound oxidation transcriptomes. Appl Environ Microb 2008;74:7723–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auernik KS, Maezato Y, Blum PH et al. The genome sequence of the metal-mobilizing, extremely thermoacidophilic archaeon Metallosphaera sedula provides insights into bioleaching-associated metabolism. Appl Environ Microb 2008;74:682–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bada JL, Lazcano A. Miller revealed new ways to study the origins of life. Nature 2002;416:475. [DOI] [PubMed] [Google Scholar]
- Bae SS, Lee HS, Jeon JH et al. Enhancing bio-hydrogen production from sodium formate by hyperthermophilic archaeon, Thermococcus onnurineus NA1. Bioprocess Biosyst Eng 2015;38:989–93. [DOI] [PubMed] [Google Scholar]
- Bae SS, Kim TW, Lee HS et al. H2 production from CO, formate or starch using the hyperthermophilic archaeon, Thermococcus onnurineus. Biotechnol Lett 2012;34:75–9. [DOI] [PubMed] [Google Scholar]
- Bae SS, Kim YJ, Yang SH et al. Thermococcus onnurineus sp nov., a hyperthermophilic Archaeon isolated from a deep-sea hydrothermal vent area at the PACMANUS field. J Microbiol Biotechnol 2006;16:1826–31. [Google Scholar]
- Balint B, Bagi Z, Toth A et al. Utilization of keratin-containing biowaste to produce biohydrogen. Appl Microbiol Biot 2005;69:404–10. [DOI] [PubMed] [Google Scholar]
- Bandyopadhyay S, Cookson MR. Evolutionary and functional relationships within the DJ1 superfamily. BMC Evol Biol 2004;4:6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bar-Even A, Noor E, Lewis NE et al. Design and analysis of synthetic carbon fixation pathways. Proc Natl Acad Sci USA 2010;107:8889–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barenholz Y. Liposome application: problems and prospects. Curr Opin Colloid In 2001;6:66–77. [Google Scholar]
- Barr DW, Ingledew WJ, Norris PR. Respiratory chain components of iron-oxidizing acidophilic bacteria. FEMS Microbiol Lett 1990;70:85–9. [Google Scholar]
- Basen M, Sun J, Adams MW. Engineering a hyperthermophilic archaeon for temperature-dependent product formation. mBio 2012;3:e00053–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basen M, Schut GJ, Nguyen DM et al. Single gene insertion drives bioalcohol production by a thermophilic archaeon. Proc Natl Acad Sci USA 2014;111:17618–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bathe S, Norris PR. Ferrous iron- and sulfur-induced genes in Sulfolobus metallicus. Appl Environ Microb 2007;73:2491–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batty JD, Rorke GV. Development and commercial demonstration of the BioCOP™ thermophile process. Hydrometallurgy 2006;83:83–9. [Google Scholar]
- Bellack A, Huber H, Rachel R et al. Methanocaldococcus villosus sp. nov., a heavily flagellated archaeon that adheres to surfaces and forms cell-cell contacts. Int J Syst Evol Micr 2011;61:1239–45. [DOI] [PubMed] [Google Scholar]
- Benvegnu T, Lemiegre L, Cammas-Marion S. Archaeal lipids: innovative materials for biotechnological applications. Eur J Org Chem 2008;28:4725–44. [Google Scholar]
- Benvegnu T, Lemiègre L, Dalençon S et al. Applications of extremophilic archaeal lipids in the field of nanocarriers for oral/topical drug delivery. Curr Biotechnol 2013;2:294–303. [Google Scholar]
- Benvegnu T, Rethore G, Brard M et al. Archaeosomes based on novel synthetic tetraether-type lipids for the development of oral delivery systems. Chem Commun 2005;44:5536–8. [DOI] [PubMed] [Google Scholar]
- Berg IA, Kockelkorn D, Buckel W et al. A 3-hydroxypropionate/4-hydroxybutyrate autotrophic carbon dioxide assimilation pathway in archaea. Science 2007;318:1782–6. [DOI] [PubMed] [Google Scholar]
- Berkner S, Grogan D, Albers S-V et al. Small multicopy, non-integrative shuttle vectors based on the plasmid pRN1 for Sulfolobus acidocaldarius and Sulfolobus solfataricus, model organisms of the (cren-)archaea. Nucleic Acids Res 2007;35:e88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berkner S, Wlodkowski A, Albers S-V et al. Inducible and constitutive promoters for genetic systems in Sulfolobus acidocaldarius. Extremophiles 2010;14:249–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biles BD, Connolly BA. Low-fidelity Pyrococcus furiosus DNA polymerase mutants useful in error-prone PCR. Nucleic Acids Res 2004;32:e176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birrien JL, Zeng X, Jebbar M et al. Pyrococcus yayanosii sp. nov., an obligate piezophilic hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent. Int J Syst Evol Microbiol 2011;61:2827–31. [DOI] [PubMed] [Google Scholar]
- Blake RC, Shute Ea, Greenwood MM et al. Enzymes of aerobic respiration on iron. FEMS Microbiol Rev 1993;11:9–18. [DOI] [PubMed] [Google Scholar]
- Blumentals I, Itoh M, Olson G et al. Role of polysulfides in reduction of elemental sulfur by the hyperthermophilic archaebacterium Pyrococcus furiosus. Appl Environ Microb 1990;56:1255–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumentals I, Robinson A, Kelly R. Characterization of sodium dodecyl sulfate-resistant proteolytic activity in the hyperthermophilic archaebacterium Pyrococcus furiosus. Appl Environ Microb 1990;56:1992–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumer-Schuette SE, Kataeva I, Westpheling J et al. Extremely thermophilic microorganisms for biomass conversion: status and prospects. Curr Opin Biotechnol 2008;19:210–7. [DOI] [PubMed] [Google Scholar]
- Bonnefoy V. Bioinformatics and genomics of iron- and sulfur-oxidizing acidophiles. In: Barton LMandl M, Loy A (eds.) Geomicrobiology: Molecular and Environmental Perspective. Netherlands, Dordrecht:Springer, 2010, 169–92. [Google Scholar]
- Bonnefoy V, Holmes DS. Genomic insights into microbial iron oxidation and iron uptake strategies in extremely acidic environments. Environ Microbiol 2012;14:1597–611. [DOI] [PubMed] [Google Scholar]
- Braakman R, Smith E. The emergence and early evolution of biological carbon-fixation. PLoS Comput Biol 2012;8:e1002455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brard M, Laine C, Rethore G et al. Synthesis of archaeal bipolar lipid analogues: a way to versatile drug/gene delivery systems. J Org Chem 2007;72:8267–79. [DOI] [PubMed] [Google Scholar]
- Bridger SL, Clarkson SM, Stirrett K et al. Deletion strains reveal metabolic roles for key elemental sulfur-responsive proteins in Pyrococcus furiosus. J Bacteriol 2011;193:6498–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brierley CL, Brierley JA. Progress in bioleaching: Part B: applications of microbial processes by the minerals industries. Appl Microbiol Biot 2013;97:7543–52. [DOI] [PubMed] [Google Scholar]
- Brito JA, Sousa FL, Stelter M et al. Structural and functional insights into sulfide:quinone oxidoreductase. Biochemistry 2009;48:5613–22. [DOI] [PubMed] [Google Scholar]
- Brock TD, Brock KM, Belly RT et al. Sulfolobus: a new genus of sulfur-oxidizing bacteria living at low pH and high temperature. Archiv Mikrobiol 1972;84:54–68. [DOI] [PubMed] [Google Scholar]
- Brooks MA, Meslet-Cladiere L, Graille M et al. The structure of an archaeal homodimeric ligase which has RNA circularization activity. Protein Sci 2008;17:1336–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown DA, Venegas B, Cooke PH et al. Bipolar tetraether archaeosomes exhibit unusual stability against autoclaving as studied by dynamic light scattering and electron microscopy. Chem Phys Lipids 2009;159:95–103. [DOI] [PubMed] [Google Scholar]
- Brown SH, Costantino HR, Kelly RM. Characterization of amylolytic enzyme activities associated with the hyperthermophilic archaebacterium Pyrococcus furiosus. Appl Environ Microb 1990;56:1985–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brugger K, Torarinsson E, Redder P et al. Shuffling of Sulfolobus genomes by autonomous and non-autonomous mobile elements. Biochem Soc Trans 2004;32:179–83. [DOI] [PubMed] [Google Scholar]
- Buan N, Kulkarni G, Metcalf W. Genetic methods for Methanosarcina species. Method Enzymol 2011;494:23–42. [DOI] [PubMed] [Google Scholar]
- Bult CJ, White O, Olsen GJ et al. Complete genome sequence of the methanogenic archaeon, Methanococcus jannaschii. Science 1996;273:1058–73. [DOI] [PubMed] [Google Scholar]
- Burgers PM, Koonin EV, Bruford E et al. Eukaryotic DNA polymerases: proposal for a revised nomenclature. J Biol Chem 2001;276:43487–90. [DOI] [PubMed] [Google Scholar]
- Burkhardt M, Koschack T, Busch G. Biocatalytic methanation of hydrogen and carbon dioxide in an anaerobic three-phase system. Bioresource Technol 2015;178:330–3. [DOI] [PubMed] [Google Scholar]
- Burstein D, Sun CL, Brown CT et al. Major bacterial lineages are essentially devoid of CRISPR-Cas viral defence systems. Nat Commun 2016;7:10613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cady SG, Bauer MW, Callen W et al. Beta-Endoglucanase from Pyrococcus furiosus. Method Enzymol 2001;330:346–54. [DOI] [PubMed] [Google Scholar]
- Callahan BP, Stanger M, Belfort M. A redox trap to augment the intein toolbox. Biotechnol Bioeng 2013;110:1565–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell E, Meredith M, Minteer SD et al. Enzymatic biofuel cells utilizing a biomimetic cofactor. Chem Commun 2012;48:1898–900. [DOI] [PubMed] [Google Scholar]
- Cannio R, Contursi P, Rossi M et al. An autonomously replicating transforming vector for Sulfolobus solfataricus. J Bacteriol 1998;180:3237–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cario A, Mizgier A, Thiel A et al. Restoration of the di-myo-inositol-phosphate pathway in the piezo-hyperthermophilic archaeon Thermococcus barophilus. Biochimie 2015;118:286–93. [DOI] [PubMed] [Google Scholar]
- Cario A, Jebbar M, Thiel A et al. Molecular chaperone accumulation as a function of stress evidences adaptation to high hydrostatic pressure in the piezophilic archaeon Thermococcus barophilus. Sci Rep 2016;6:29483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambers CR, Patrick WM. Archaeal nucleic acid ligases and their potential in biotechnology. Archaea 2015;2015:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandrayan SK, McTernan PM, Hopkins RC et al. Engineering hyperthermophilic archaeon Pyrococcus furiosus to overproduce its cytoplasmic [NiFe]-hydrogenase. J Biol Chem 2012;287:3257–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandrayan SK, Wu CH, McTernan PM et al. High yield purification of a tagged cytoplasmic [NiFe]-hydrogenase and a catalytically-active nickel-free intermediate form. Protein Expr Purif 2015;107:90–4. [DOI] [PubMed] [Google Scholar]
- Chen LM, Brugger K, Skovgaard M et al. The genome of Sulfolobus acidocaldarius, a model organism of the Crenarchaeota. J Bacteriol 2005;187:4992–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng K, Zhang F, Sun F et al. Doubling power output of starch biobattery treated by the most thermostable isoamylase from an archaeon Sulfolobus tokodaii. Sci Rep 2015;5:13184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chica B, Wu CH, Liu Y et al. Balancing electron transfer rate and driving force for efficient photocatalytic hydrogen production in CdSe/CdS nanorod–[NiFe] hydrogenase assemblies. Energy Environ Sci 2017;10:2245–55. [Google Scholar]
- Chien A, Edgar DB, Trela JM. Deoxyribonucleic acid polymerase from the extreme thermophile Thermus aquaticus. J Bacteriol 1976;127:1550–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiuman W, Li Y. Making AppDNA using T4 DNA ligase. Bioorg Chem 2002;30:332–49. [DOI] [PubMed] [Google Scholar]
- Chong S, Montello GE, Zhang A et al. Utilizing the C-terminal cleavage activity of a protein splicing element to purify recombinant proteins in a single chromatographic step. Nucleic Acids Res 1998;26:5109–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou CJ, Shockley KR, Conners SB et al. Impact of substrate glycoside linkage and elemental sulfur on bioenergetics of and hydrogen production by the hyperthermophilic archaeon Pyrococcus furiosus. Appl Environ Microb 2007;73:6842–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung YC, Kobayashi T, Kanai H et al. Purification and properties of extracellular amylase from the hyperthermophilic archaeon Thermococcus profundus dt5432. Appl Environ Microb 1995;61:1502–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciftci H, Akcil A. Biohydrometallurgy in Turkish gold mining: first shake flask and bioreactor studies. Miner Eng 2013;46–47:25–33. [Google Scholar]
- Costa KC, Leigh JA. Metabolic versatility in methanogens. Curr Opin Biotechnol 2014;29:70–5. [DOI] [PubMed] [Google Scholar]
- Counts JA, Zeldes BM, Lee LL et al. Physiological, metabolic and biotechnological features of extremely thermophilic microorganisms. WIRES Syst Biol Med 2017;9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crundwell FK. The semiconductor mechanism of dissolution and the pseudo-passivation of chalcopyrite. Can Metall Quart 2015;54:279–88. [Google Scholar]
- Cvetkovic A, Menon AL, Thorgersen MP et al. Microbial metalloproteomes are largely uncharacterized. Nature 2010;466:779–82. [DOI] [PubMed] [Google Scholar]
- d’Hughes PD, Foucher S. HIOX® Project: a bioleaching process for the treatment of chalcopyrite concentrates using extreme thermophiles. In: Ciminelli VST, Garcia O (eds) Biohydrometallurgy:Fundamentals, Technology and Sustainable Development. Amsterdam:Elsevier, 2001, 75–83. [Google Scholar]
- De Kock SH, Barnard P, du Plessis C. Oxygen and carbon dioxide kinetic challenges for thermophilic mineral bioleaching processes. Biochem Soc Trans 2004;32:273–5. [DOI] [PubMed] [Google Scholar]
- De Poorter LMI, Geerts WJ, Keltjens JT. Coupling of methanothermobacter thermautotrophicus methane formation and growth in Fed-Batch and continuous cultures under different H2 gassing regimens. Appl Environ Microb 2007;73:740–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Rosa M, Gambacorta A, Nicolaus B et al. Isoprenoid ethers; backbone of complex lipids of the archaebacterium Sulfolobus solfataricus. BBA- Lipid Lipid Met 1983;753:249–56. [Google Scholar]
- De Vrieze J, Hennebel T, Boon N et al. Methanosarcina: the rediscovered methanogen for heavy duty biomethanation. Bioresource Technol 2012;112:1–9. [DOI] [PubMed] [Google Scholar]
- Del Giudice I, Coppolecchia R, Merone L et al. An efficient thermostable organophosphate hydrolase and its application in pesticide decontamination. Biotechnol Bioeng 2016;113:724–34. [DOI] [PubMed] [Google Scholar]
- Deng L, Garrett RA, Shah SA et al. A novel interference mechanism by a type IIIB CRISPR-Cmr module in Sulfolobus. Mol Microbiol 2013;87:1088–99. [DOI] [PubMed] [Google Scholar]
- Deng L, Zhu HJ, Chen ZJ et al. Unmarked gene deletion and host-vector system for the hyperthermophilic crenarchaeon Sulfolobus islandicus. Extremophiles 2009;13:735–46. [DOI] [PubMed] [Google Scholar]
- Dew DW, Buuren CV, McEwan K et al. Bioleaching of base metal sulphide concentrates: a comparison of high and low temperature bioleaching. J S Afr I Min Metall 2000;100:409–14. [Google Scholar]
- Dirmeier R, Keller M, Frey G et al. Purification and properties of an extremely thermostable membrane-bound sulfur-reducing complex from the hyperthermophilic Pyrodictium abyssi. Eur J Biochem 1998;252:486–91. [DOI] [PubMed] [Google Scholar]
- Donati ER, Castro C, Urbieta MS. Thermophilic microorganisms in biomining. World J Microb Biot 2016;32:179. [DOI] [PubMed] [Google Scholar]
- Dong G, Vieille C, Savchenko A et al. Cloning, sequencing, and expression of the gene encoding extracellular alpha-amylase from Pyrococcus furiosus and biochemical characterization of the recombinant enzyme. Appl Environ Microb 1997;63:3569–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donohoue PD, Barrangou R, May AP. Advances in Industrial biotechnology using CRISPR-Cas systems. Trends Biotechnol 2017. [DOI] [PubMed] [Google Scholar]
- du Plessis CA, Batty JD, Dew DW. Commercial applications of thermophile bioleaching. In: Rawlings DE, Johnson DB (eds.) Biomining. Springer: Berlin, 2007, 57–80. [Google Scholar]
- Elferink MG, De Wit JG, Driessen AJ et al. Energy-transducing properties of primary proton pumps reconstituted into archaeal bipolar lipid vesicles. Eur J Biochem 1993;214:917–25. [DOI] [PubMed] [Google Scholar]
- Ellen AF, Albers SV, Driessen AJ. Comparative study of the extracellular proteome of Sulfolobus species reveals limited secretion. Extremophiles 2010;14:87–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmore J, Deighan T, Westpheling J et al. DNA targeting by the type I-G and type I-A CRISPR-Cas systems of Pyrococcus furiosus. Nucleic Acids Res 2015;43:10353–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elshawadfy AM, Keith BJ, Ee Ooi H et al. DNA polymerase hybrids derived from the family-B enzymes of Pyrococcus furiosus and Thermococcus kodakarensis: improving performance in the polymerase chain reaction. Front Microbiol 2014;5:224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enghiad B, Zhao H. Programmable DNA-guided artificial restriction enzymes. ACS Synth Biol 2017;6:752–7. [DOI] [PubMed] [Google Scholar]
- Enthaler S, von Langermann J, Schmidt T. Carbon dioxide and formic acid-the couple for environmental-friendly hydrogen storage? Energy Environ Sci 2010;3:1207–17. [Google Scholar]
- Erauso G, Reysenbach AL, Godfroy A et al. Pyrococcus abyssi sp. nov., a new hyperthermophilic archaeon isolated from a deep-sea hydrothermal vent. Arch Microbiol 1993;160:338–49. [Google Scholar]
- Evans SJ, Fogg MJ, Mamone A et al. Improving dideoxynucleotide-triphosphate utilisation by the hyper-thermophilic DNA polymerase from the archaeon Pyrococcus furiosus. Nucleic Acids Res 2000;28:1059–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezaki S, Maeda N, Kishimoto T et al. Presence of a structurally novel type ribulose-bisphosphate carboxylase/oxygenase in the hyperthermophilic archaeon, Pyrococcus kodakaraensis KOD1. J Biol Chem 1999;274:5078–82. [DOI] [PubMed] [Google Scholar]
- Farkas J, Stirrett K, Lipscomb GL et al. Recombinogenic properties of Pyrococcus furiosus strain COM1 enable rapid selection of targeted mutants. Appl Environ Microb 2012;78:4669–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farkas JA, Picking JW, Santangelo TJ. Genetic techniques for the archaea. Annu Rev Genet 2013;47:539–61. [DOI] [PubMed] [Google Scholar]
- Febo-Ayala W, Morera-Felix SL, Hrycyna CA et al. Functional reconstitution of the integral membrane enzyme, isoprenylcysteine carboxyl methyltransferase, in synthetic bolalipid membrane vesicles. Biochemistry 2006;45:14683–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferry JG. Methane from acetate. J Bacteriol 1992;174:5489–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiala G, Stetter KO. Pyrococcus furiosus Sp. nov. represents a novel genus of marine heterotrophic archaebacteria growing optimally at 100°C. Arch Microbiol 1986;145:56–61. [Google Scholar]
- Florentino AP, Weijma J, Stams AJM et al. Ecophysiology and application of acidophilic sulfur-reducing microorganisms. In: Rampelotto PH. (ed.) Biotechnology of Extremophiles. Cham: Springer International Publishing, 2016, 141–75. [Google Scholar]
- Fournier GP, Gogarten JP. Evolution of acetoclastic methanogenesis in Methanosarcina via horizontal gene transfer from cellulolytic clostridia. J Bacteriol 2008;190:1124–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freisleben HJ, Zwicker K, Jezek P et al. Reconstitution of bacteriorhodopsin and ATP synthase from Micrococcus luteus into liposomes of the purified main tetraether lipid from Thermoplasma acidophilum: proton conductance and light-driven ATP synthesis. Chem Phys Lipids 1995;78:137–47. [DOI] [PubMed] [Google Scholar]
- Fujihashi M, Nishitani Y, Kiriyama T et al. Mutation design of a thermophilic Rubisco based on three-dimensional structure enhances its activity at ambient temperature. Proteins 2016;84:1339–46. [DOI] [PubMed] [Google Scholar]
- Fujisawa T, Fujinaga S, Atomi H. An in vitro enzyme system for the production of MYO-Inositol from starch. Appl Environ Microb 2017;83:e00550-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda W, Morimoto N, Imanaka T et al. Agmatine is essential for the cell growth of Thermococcus kodakaraensis. FEMS Microbiol Lett 2008;287:113–20. [DOI] [PubMed] [Google Scholar]
- Fukui T, Atomi H, Kanai T et al. Complete genome sequence of the hyperthermophilic archaeon Thermococcus kodakaraensis KOD1 and comparison with Pyrococcus genomes. Genome Res 2005;15:352–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaglione R, Pirone L, Farina B et al. Insights into the anticancer properties of the first antimicrobial peptide from Archaea. BBA- Gen Subjects 2017;1861:2155–64. [DOI] [PubMed] [Google Scholar]
- Gambacorta A, Gliozzi A, De Rosa M. Archaeal lipids and their biotechnological applications. World J Microb Biot 1995;11:115–31. [DOI] [PubMed] [Google Scholar]
- Gao J, Bauer MW, Shockley KR et al. Growth of hyperthermophilic archaeon Pyrococcus furiosus on chitin involves two family 18 chitinases. Appl Environ Microb 2003;69:3119–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner AF, Jack WE. Determinants of nucleotide sugar recognition in an archaeon DNA polymerase. Nucleic Acids Res 1999;27:2545–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner AF, Jack WE. Acyclic and dideoxy terminator preferences denote divergent sugar recognition by archaeon and Taq DNA polymerases. Nucleic Acids Res 2002;30:605–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gavrilov SN, Stracke C, Jensen K et al. Isolation and characterization of the first xylanolytic hyperthermophilic euryarchaeon Thermococcus sp. strain 2319x1 and its unusual multidomain glycosidase. Front Microbiol 2016;7:552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gehring AM, Astling DP, Matsumi R et al. Genome replication in Thermococcus kodakarensis Independent of Cdc6 and an origin of replication. Front Microbiol 2017;8:2084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geng XM, Liu X, Ji M et al. Enhancing heat tolerance of the little dogwood Cornus canadensis L.f. with introduction of a superoxide reductase gene from the hyperthermophilic archaeon Pyrococcus furiosus. Front Plant Sci 2016;7:26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gericke M, Govender Y. Bioleaching strategies for the treatment of nickel–copper sulphide concentrates. Miner Eng 2011;24:1106–12. [Google Scholar]
- Gericke M, Govender Y, Pinches A. Tank bioleaching of low-grade chalcopyrite concentrates using redox control. Hydrometallurgy 2010;104:414–9. [Google Scholar]
- Gericke M, Pinches A, van Rooyen JV. Bioleaching of a chalcopyrite concentrate using an extremely thermophilic culture. Int J Miner Process 2001;62:243–55. [Google Scholar]
- Gibson DG, Young L, Chuang RY et al. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat Methods 2009;6:343–5. [DOI] [PubMed] [Google Scholar]
- Girfoglio M, Rossi M, Cannio R. Cellulose degradation by Sulfolobus solfataricus requires a cell-anchored endo-β-1-4-glucanase. J Bacteriol 2012;194:5091–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gliozzi A, Relini A, Chong PL. Structure and permeability properties of biomimetic membranes of bolaform archaeal tetraether lipids. J Membrane Sci 2002;206:131–47. [Google Scholar]
- Gliozzi A, Rolandi R, De Rosa M et al. Monolayer black membranes from bipolar lipids of archaebacteria and their temperature-induced structural changes. J Membrane Biol 1983;75:45–56. [DOI] [PubMed] [Google Scholar]
- Glover DJ, Clark DS. Oligomeric assembly is required for chaperone activity of the filamentous γ-prefoldin. FEBS J 2015;282:2985–97. [DOI] [PubMed] [Google Scholar]
- Glover DJ, Giger L, Kim SS et al. Geometrical assembly of ultrastable protein templates for nanomaterials. Nat Commun 2016;7:11771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham JE, Clark ME, Nadler DC et al. Identification and characterization of a multidomain hyperthermophilic cellulase from an archaeal enrichment. Nat Commun 2011;2:375. [DOI] [PubMed] [Google Scholar]
- Gräther OA, Arigoni D. Detection of Regioisomeric macrocyclic tetraethers in the lipids of Methanobacterium thermoautotrophicum and other archaeal organisms. J Chem Soc Chem Comm 1995;4;405–6. [Google Scholar]
- Grogan DW. Phenotypic characterization of the archaebacterial genus Sulfolobus: comparison of five wild-type strains. J Bacteriol 1989;171:6710–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grogan DW, Carver GT, Drake JW. Genetic fidelity under harsh conditions: analysis of spontaneous mutation in the thermoacidophilic archaeon Sulfolobus acidocaldarius. Proc Natl Acad Sci USA 2001;98:7928–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo L, Brugger K, Liu C et al. Genome Analyses of icelandic strains of Sulfolobus islandicus, model organisms for genetic and virus-host interaction studies. J Bacteriol 2011;193:1672–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guschinskaya N, Brunel R, Tourte M et al. Random mutagenesis of the hyperthermophilic archaeon Pyrococcus furiosus using in vitro mariner transposition and natural transformation. Sci Rep 2016;6:36711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halio SB, Blumentals II, Short SA et al. Sequence, expression in Escherichia coli, and analysis of the gene encoding a novel intracellular protease (PfpI) from the hyperthermophilic archaeon Pyrococcus furiosus. J Bacteriol 1996;178:2605–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halio SB, Bauer MW, Mukund S et al. Purification and characterization of two functional forms of intracellular protease PfpI from the hyperthermophilic archaeon Pyrococcus furiosus. Appl Environ Microb 1997;63:289–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanford MJ, Peeples TL. Archaeal tetraether lipids: unique structures and applications. Appl Biochem Biotechnol 2002;97:45–62. [DOI] [PubMed] [Google Scholar]
- Hannington MD, Jonasson IR, Herzig PM et al. Physical and chemical processes of seafloor mineralization at mid‐ocean ridges. In: Humphris SE, Zierenberg RA, Mullineaux LS (eds). Seafloor Hydrothermal Systems: Physical, Chemical, Biological, and Geological Interactions. 1995, 115–57. [Google Scholar]
- Haring M, Rachel R, Peng X et al. Viral diversity in hot springs of Pozzuoli, Italy, and characterization of a unique archaeal virus, Acidianus bottle-shaped virus, from a new family, the Ampullaviridae. J Virol 2005;79:9904–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrington LB, Paez-Espino D, Staahl BT et al. A thermostable Cas9 with increased lifetime in human plasma. Nat Commun 2017;8:1424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimoto H, Nishioka M, Fujiwara S et al. Crystal structure of DNA polymerase from hyperthermophilic archaeon Pyrococcus kodakaraensis KOD111. J Mol Biol 2001;306:469–77. [DOI] [PubMed] [Google Scholar]
- Hashimoto H, Matsumoto T, Nishioka M et al. Crystallographic studies on a family B DNA polymerase from hyperthermophilic archaeon Pyrococcus kodakaraensis strain KODI. J Biochem (Tokyo) 1999;125:983–6. [DOI] [PubMed] [Google Scholar]
- Hawkins AB, Lian H, Zeldes BM et al. Bioprocessing analysis of Pyrococcus furiosus strains engineered for CO2-based 3-hydroxypropionate production. Biotechnol Bioeng 2015;112:1533–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henche AL, Koerdt A, Ghosh A et al. Influence of cell surface structures on crenarchaeal biofilm formation using a thermostable green fluorescent protein. Environ Microbiol 2012;14:779–93. [DOI] [PubMed] [Google Scholar]
- Hennessy JE, Latter MJ, Fazelinejad S et al. Hyperthermophilic carbamate kinase stability and anabolic in vitro activity at alkaline pH. Appl Environ Microb 2017;84:e02250–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hensley SA, Moreira E, Holden JF. Hydrogen production and enzyme activities in the hyperthermophile Thermococcus paralvinellae grown on maltose, tryptone, and agricultural waste. Front Microbiol 2016;7:167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiblot J, Gotthard G, Chabriere E et al. Characterisation of the organophosphate hydrolase catalytic activity of SsoPox. Sci Rep 2012a;2:779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiblot J, Gotthard G, Chabriere E et al. Structural and enzymatic characterization of the lactonase SisLac from Sulfolobus islandicus. PLoS One 2012b;7:e47028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hileman TH, Santangelo TJ. Genetics techniques for Thermococcus kodakarensis. Front Microbiol 2012;3:195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hita R, Wang H, Bigham JM et al. Bioleaching of a pyritic sludge from the Aznalcóllar (Spain) mine spillage at ambient and elevated temperatures. Hydrometallurgy 2008;93:76–9. [Google Scholar]
- Horiuchi A, Aslam M, Kanai T et al. A structurally novel chitinase from the chitin-degrading hyperthermophilic archaeon Thermococcus chitonophagus. Appl Environ Microb 2016;82:3554–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber G, Stetter KO. Sulfolobus metallicus, sp. nov., a novel strictly chemolithoautotrophic thermophilic archaeal species of metal-mobilizers. Syst Appl Microbiol 1991;14:372–8. [Google Scholar]
- Huber G, Spinnler C, Gambacorta A et al. Metallosphaera sedula gen, and sp. nov. represents a new genus of aerobic, metal-mobilizing, thermoacidophilic Archaebacteria. Syst Appl Microbiol 1989;12:38–47. [Google Scholar]
- Huber H, Burggraf S, Mayer T et al. Ignicoccus gen. nov., a novel genus of hyperthermophilic, chemolithoautotrophic Archaea, represented by two new species, Ignicoccus Islandicus sp. nov. and Ignicoccus pacificus sp. nov. Int J Syst Evol Micr 2000;50(Pt 6):2093–100. [DOI] [PubMed] [Google Scholar]
- Huber H, Gallenberger M, Jahn U et al. A dicarboxylate/4-hydroxybutyrate autotrophic carbon assimilation cycle in the hyperthermophilic archaeum Ignicoccus hospitalis. Proc Natl Acad Sci USA 2008;105:7851–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ilbert M, Bonnefoy V. Insight into the evolution of the iron oxidation pathways. BBA- Bioenergetics 2013;1827:161–75. [DOI] [PubMed] [Google Scholar]
- Im YJ, Ji M, Lee AM et al. Production of a thermostable archaeal superoxide reductase in plant cells. FEBS Lett 2005;579:5521–6. [DOI] [PubMed] [Google Scholar]
- Im YJ, Ji M, Lee A et al. Expression of Pyrococcus furiosus superoxide reductase in Arabidopsis enhances heat tolerance. Plant Physiol 2009;151:893–904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- In’t Veld G, Elferink MG, Driessen AJ et al. Reconstitution of the leucine transport system of Lactococcus lactis into liposomes composed of membrane-spanning lipids from Sulfolobus acidocaldarius. Biochemistry 1992;31:12493–9. [DOI] [PubMed] [Google Scholar]
- Ingledew WJ. Thiobacillus ferrooxidans the bioenergetics of an acidophilic chemolithotroph. BBA- Rev Bioenergetics 1982;683:89–117. [DOI] [PubMed] [Google Scholar]
- Iranzo J, Koonin EV, Prangishvili D et al. Bipartite network analysis of the archaeal virosphere: evolutionary connections between viruses and capsidless mobile elements. J Virol 2016;90:11043–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iranzo J, Lobkovsky AE, Wolf YI et al. Evolutionary dynamics of the prokaryotic adaptive immunity system CRISPR-Cas in an explicit ecological context. J Bacteriol 2013;195:3834–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh YH, Sugai A, Uda I et al. The evolution of lipids. Adv Space Res 2001;28:719–24. [DOI] [PubMed] [Google Scholar]
- Iwai H, Lingel A, Pluckthun A. Cyclic green fluorescent protein produced in vivo using an artificially split PI-PfuI intein from Pyrococcus furiosus. J Biol Chem 2001;276:16548–54. [DOI] [PubMed] [Google Scholar]
- Jacobsen AC, Jensen SM, Fricker G et al. Archaeal lipids in oral delivery of therapeutic peptides. Eur J Pharm Sci 2017;108:101–10. [DOI] [PubMed] [Google Scholar]
- Jacquemet A, Barbeau J, Lemiegre L et al. Archaeal tetraether bipolar lipids: structures, functions and applications. Biochimie 2009;91:711–7. [DOI] [PubMed] [Google Scholar]
- Jacquet P, Hiblot J, Daude D et al. Rational engineering of a native hyperthermostable lactonase into a broad spectrum phosphotriesterase. Sci Rep 2017;7:16745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssen AJH, Buisman CJN. Process for biological removal of sulphide. 2001. U.S. 6,221,652 Paques April 24, 2001.
- Jenney FE, Verhagen MFJM, Cui X et al. Anaerobic microbes: oxygen detoxification without superoxide dismutase. Science 1999;286:306–9. [DOI] [PubMed] [Google Scholar]
- Jensen SM, Christensen CJ, Petersen JM et al. Liposomes containing lipids from Sulfolobus islandicus withstand intestinal bile salts: an approach for oral drug delivery? Int J Pharm 2015;493:63–9. [DOI] [PubMed] [Google Scholar]
- Jentsch M, Trost T, Sterner M. Optimal use of power-to-gas energy storage systems in an 85% renewable energy scenario. Energy Procedia 2014;46:254–61. [Google Scholar]
- Jeon SJ, Ishikawa K. A novel ADP-dependent DNA ligase from Aeropyrum pernix K1. FEBS Lett 2003;550:69–73. [DOI] [PubMed] [Google Scholar]
- Jiang L, Huang C, Wang B et al. Enhanced heat tolerance in transgenic silkworm via overexpression of Pyrococcus furiosus superoxide reductase. Insect Biochem Mol Biol 2018;92:40–4. [DOI] [PubMed] [Google Scholar]
- Johnson DB. Biomining—biotechnologies for extracting and recovering metals from ores and waste materials. Curr Opin Biotechnol 2014;30:24–31. [DOI] [PubMed] [Google Scholar]
- Jones GC, van Hille RP, Harrison STL. Reactive oxygen species generated in the presence of fine pyrite particles and its implication in thermophilic mineral bioleaching. Appl Microbiol Biot 2013;97:2735–42. [DOI] [PubMed] [Google Scholar]
- Jones WJ, Leigh JA, Mayer F et al. Methanococcus jannaschii sp. nov., an extremely thermophilic methanogen from a submarine hydrothermal vent. Arch Microbiol 1983;136:254–61. [Google Scholar]
- Jonuscheit M, Martusewitsch E, Stedman KM et al. A reporter gene system for the hyperthermophilic archaeon Sulfolobus solfataricus based on a selectable and integrative shuttle vector. Mol Microbiol 2003;48:1241–52. [DOI] [PubMed] [Google Scholar]
- Joo F. Breakthroughs in hydrogen Storage-Formic acid as a sustainable storage material for hydrogen. ChemSusChem 2008;1:805–8. [DOI] [PubMed] [Google Scholar]
- Jorgensen BB, Isaksen MF, Jannasch HW. Bacterial sulfate reduction above 100 C in deep-sea hydrothermal vent sediments. Science 1992;258:1756–7. [DOI] [PubMed] [Google Scholar]
- Jorgensen S, Vorgias CE, Antranikian G. Cloning, sequencing, characterization, and expression of an extracellular alpha-Amylase from the hyperthermophilic archaeon Pyrococcus furiosus in Escherichia coli and Bacillus subtilis. J Biol Chem 1997;272:16335–42. [DOI] [PubMed] [Google Scholar]
- Ju J, Kim DH, Bi L et al. Four-color DNA sequencing by synthesis using cleavable fluorescent nucleotide reversible terminators. Proc Natl Acad Sci USA 2006;103:19635–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaksonen AH, Mudunuru BM, Hackl R. The role of microorganisms in gold processing and recovery—A review. Hydrometallurgy 2014;142:70–83. [Google Scholar]
- Kanai T, Imanaka H, Nakajima A et al. Continuous hydrogen production by the hyperthermophilic archaeon, Thermococcus kodakaraensis KOD1. J Biotechnol 2005;116:271–82. [DOI] [PubMed] [Google Scholar]
- Kanai T, Matsuoka R, Beppu H et al. Distinct physiological roles of the three [NiFe]-hydrogenase orthologs in the hyperthermophilic archaeon Thermococcus kodakarensis. J Bacteriol 2011;193:3109–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanai T, Simons JR, Tsukamoto R et al. Overproduction of the membrane-bound [NiFe]-hydrogenase in Thermococcus kodakarensis and its effect on hydrogen production. Front Microbiol 2015;6:847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanai T, Yasukochi A, Simons JR et al. Genetic analyses of the functions of [NiFe]-hydrogenase maturation endopeptidases in the hyperthermophilic archaeon Thermococcus kodakarensis. Extremophiles 2017;21:27–39. [DOI] [PubMed] [Google Scholar]
- Kasson P, DiMaio F, Yu X et al. Model for a novel membrane envelope in a filamentous hyperthermophilic virus. Elife 2017;6:e26268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kates M. The Archaebacteria: biochemistry and biotechnology. Biochem Soc Symp 1992;58:1–207. [PubMed] [Google Scholar]
- Keller MW, Lipscomb GL, Loder AJ et al. A hybrid synthetic pathway for butanol production by a hyperthermophilic microbe. Metab Eng 2015;27:101–6. [DOI] [PubMed] [Google Scholar]
- Keller MW, Schut GJ, Lipscomb GL et al. Exploiting microbial hyperthermophilicity to produce an industrial chemical, using hydrogen and carbon dioxide. Proc Natl Acad Sci USA 2013;110:5840–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khoshkhoo M, Dopson M, Shchukarev A et al. Chalcopyrite leaching and bioleaching: an X-ray photoelectron spectroscopic (XPS) investigation on the nature of hindered dissolution. Hydrometallurgy 2014;149:220–7. [Google Scholar]
- Kim DJ, Kim O, Kim HW et al. ATP-dependent DNA ligase from Archaeoglobus fulgidus displays a tightly closed conformation. Acta Crystallogr F 2009;65:544–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EJ, Wu CH, Adams MW et al. Exceptionally high rates of biological hydrogen production by biomimetic in vitro synthetic enzymatic pathways. Chem Eur J 2016a;22:16047–51. [DOI] [PubMed] [Google Scholar]
- Kim JE, Kim EJ, Chen H et al. Advanced water splitting for green hydrogen gas production through complete oxidation of starch by in vitro metabolic engineering. Metab Eng 2017;44:246–52. [DOI] [PubMed] [Google Scholar]
- Kim MS, Bae SS, Kim YJ et al. CO-Dependent H2 production by genetically engineered thermococcus onnurineus NA1. Appl Environ Microb 2013;79:2048–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim TW, Bae SS, Lee JW et al. A biological process effective for the conversion of CO-containing industrial waste gas to acetate. Bioresource Technol 2016b;211:792–6. [DOI] [PubMed] [Google Scholar]
- Kim YJ, Lee HS, Kim ES et al. Formate-driven growth coupled with H2 production. Nature 2010;467:352–5. [DOI] [PubMed] [Google Scholar]
- Klenk HP, Clayton RA, Tomb JF et al. The complete genome sequence of the hyperthermophilic, sulphate-reducing archaeon Archaeoglobus fulgidus. Nature 1997;390:364–70. [DOI] [PubMed] [Google Scholar]
- Kletzin A. Coupled enzymatic production of sulfite, thiosulfate, and hydrogen sulfide from sulfur: purification and properties of a sulfur oxygenase reductase from the facultatively anaerobic archaebacterium Desulfurolobus ambivalens. J Bacteriol 1989;171:1638–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kletzin A. Metabolism of inorganic sulfur compounds in archaea. In: Klenk Ragh P (ed.). Archaea: Evolution, Physiology, and Molecular Biology. Archaea: Blackwell Publishing Ltd, 2007, 261–74. [Google Scholar]
- Konings WN, Albers SV, Koning S et al. The cell membrane plays a crucial role in survival of bacteria and archaea in extreme environments. Anton Van Leeuw 2002;81:61–72. [DOI] [PubMed] [Google Scholar]
- Konishi Y, Nishimura H, Asai S. Bioleaching of sphalerite by the acidophilic thermophile Acidianus brierleyi. Hydrometallurgy 1998;47:339–52. [Google Scholar]
- Kozhevnikova DA, Taranov EA, Lebedinsky AV et al. Hydrogenogenic and sulfidogenic growth of Thermococcus archaea on carbon monoxide and formate. Mikrobiologiia 2016;85:381–92. [PubMed] [Google Scholar]
- Kozubal MA, Dlakic M, Macur RE et al. Terminal oxidase diversity and function in Metallosphaera yellowstonensis: gene expression and protein modeling suggest mechanisms of Fe(II) oxidation in the Sulfolobales. Appl Environ Microb 2011;77:1844–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishnan L, Sad S, Patel GB et al. Archaeosomes induce enhanced cytotoxic T lymphocyte responses to entrapped soluble protein in the absence of interleukin 12 and protect against tumor challenge. Cancer Res 2003;63:2526–34. [PubMed] [Google Scholar]
- Krupovic M, Cvirkaite-Krupovic V, Iranzo J et al. Viruses of archaea: structural, functional, environmental and evolutionary genomics. Virus Res 2018;244:181–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krupovic M, Quemin ER, Bamford DH et al. Unification of the globally distributed spindle-shaped viruses of the Archaea. J Virol 2014;88:2354–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurosawa N, Grogan DW. Homologous recombination of exogenous DNA with the Sulfolobus acidocaldarius genome: properties and uses. FEMS Microbiol Lett 2005;253:141–9. [DOI] [PubMed] [Google Scholar]
- Kurr M, Huber R, König H et al. Methanopyrus kandleri, gen. and sp. nov. represents a novel group of hyperthermophilic methanogens, growing at 110°C. Arch Microbiol 1991;156:239–47. [Google Scholar]
- Kwon KM, Kang SG, Sokolova TG et al. Characterization of a family B DNA polymerase from Thermococcus barophilus Ch5 and its application for long and accurate PCR. Enzyme Microb Technol 2016;86:117–26. [DOI] [PubMed] [Google Scholar]
- Lai D, Lluncor B, Schroder I et al. Reconstruction of the archaeal isoprenoid ether lipid biosynthesis pathway in Escherichia coli through digeranylgeranylglyceryl phosphate. Metab Eng 2009;11:184–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laine C, Mornet E, Lemiegre L et al. Folate-equipped pegylated archaeal lipid derivatives: synthesis and transfection properties. Chem Eur J 2008;14:8330–40. [DOI] [PubMed] [Google Scholar]
- Langwaldt J. Bioleaching of multimetal black shale by thermophilic micro-organisms. Adv Mater Res 2007;20-21:167. [Google Scholar]
- Langworthy TA. Long-chain diglycerol tetraethers from Thermoplasma acidophilum. BBA-Lipid Lipid Metab 1977;487:37–50. [DOI] [PubMed] [Google Scholar]
- Larson SB, McPherson A. The structure of the Pfp1 protease from the hyperthermophilic archaeon Thermococcus thioreducens in two crystal forms. Acta Crystallogr D 2017;73:749–56. [DOI] [PubMed] [Google Scholar]
- Laska S, Lottspeich F, Kletzin A. Membrane-bound hydrogenase and sulfur reductase of the hyperthermophilic and acidophilic archaeon Acidianus ambivalens. Microbiology 2003;149:2357–71. [DOI] [PubMed] [Google Scholar]
- Le Naour F, Misek DE, Krause MC et al. Proteomics-based identification of RS/DJ-1 as a novel circulating tumor antigen in breast cancer. Clin Cancer Res 2001;7:3328–35. [PubMed] [Google Scholar]
- Lennon CW, Stanger M, Belfort M. Protein splicing of a recombinase intein induced by ssDNA and DNA damage. Genes & Dev 2016;30:2663–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Roux NW, Wakerley W. Leaching of chalcopyrite (CuFeS2) at 70°C using. In: Norris PR, Kelly DP (eds.) Sulfolobus. Proceedings of Biohydrometallurgy 87. Surrey, UK:Science and Technology Letters, 1998, 305–17. [Google Scholar]
- Lebbink JH, Kaper T, Kengen SW et al. Beta-Glucosidase CelB from Pyrococcus furiosus: production by Escherichia coli, purification, and in vitro evolution. Method Enzymol 2001;330:364–79. [DOI] [PubMed] [Google Scholar]
- Lecker B, Illi L, Lemmer A et al. Biological hydrogen methanation – a review. Bioresource Technol 2017;245:1220–8. [DOI] [PubMed] [Google Scholar]
- Lee HS, Kang SG, Bae SS et al. The complete genome sequence of Thermococcus onnurineus NA1 reveals a mixed heterotrophic and carboxydotrophic metabolism. J Bacteriol 2008;190:7491–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JC, Kim JH, Chang WS et al. Biological conversion of CO2 to CH4 using hydrogenotrophic methanogen in a fixed bed reactor. J Chem Technol Biot 2012;87:844–7. [Google Scholar]
- Leigh JA, Albers SV, Atomi H et al. Model organisms for genetics in the domain Archaea: methanogens, halophiles, Thermococcales and Sulfolobales. FEMS Microbiol Rev 2011;35:577–608. [DOI] [PubMed] [Google Scholar]
- Li B, Wang Z, Li S et al. Preparation of lactose-free pasteurized milk with a recombinant thermostable beta-glucosidase from Pyrococcus furiosus. BMC Biotechnol 2013;13:73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L, Zhang J, Lin J et al. Biological technologies for the removal of sulfur containing compounds from waste streams: bioreactors and microbial characteristics. World J Microb Biot 2015;31:1501–15. [DOI] [PubMed] [Google Scholar]
- Li S, Zhong H, Hu Y et al. Bioleaching of a low-grade nickel–copper sulfide by mixture of four thermophiles. Bioresource Technol 2014;153:300–6. [DOI] [PubMed] [Google Scholar]
- Li Z, Chen J, Sun W et al. Investigation of archaeosomes as carriers for oral delivery of peptides. Biochem Bioph Res Co 2010;394:412–7. [DOI] [PubMed] [Google Scholar]
- Li Z, Li X, Xiao X et al. An integrative genomic island affects the adaptations of the piezophilic hyperthermophilic archaeon Pyrococcus yayanosii to high temperature and high hydrostatic pressure. Front Microbiol 2016;7:1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lian H, Zeldes BM, Lipscomb GL et al. Ancillary contributions of heterologous biotin protein ligase and carbonic anhydrase for CO2 incorporation into 3-hydroxypropionate by metabolically engineered Pyrococcus furiosus. Biotechnol Bioeng 2016;113:2652–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lichtenberg D, Barenholz Y. Liposomes: preparation, characterization, and preservation. Methods Biochem Anal 1988;33:337–462. [DOI] [PubMed] [Google Scholar]
- Lim JK, Bae SS, Kim TW et al. Thermodynamics of formate-oxidizing metabolism and implications for H2 production. Appl Environ Microb 2012;78:7393–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindström EB, Sandström Å, Sundkvist J-E. A sequential two-step process using moderately and extremely thermophilic cultures for biooxidation of refractory gold concentrates. Hydrometallurgy 2003;71:21–30. [Google Scholar]
- Lipscomb GL, Hahn EM, Crowley AT et al. Reverse gyrase is essential for microbial growth at 95°C. Extremophiles 2017a;21:603–8. [DOI] [PubMed] [Google Scholar]
- Lipscomb GL, Keese AM, Cowart DM et al. SurR: a transcriptional activator and repressor controlling hydrogen and elemental sulphur metabolism in Pyrococcus furiosus. Mol Microbiol 2009;71:332–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipscomb GL, Schut GJ, Scott RA et al. SurR is a master regulator of the primary electron flow pathways in the order Thermococcales. Mol Microbiol 2017b;104:869–81. [DOI] [PubMed] [Google Scholar]
- Lipscomb GL, Schut GJ, Thorgersen MP et al. Engineering hydrogen gas production from formate in a hyperthermophile by heterologous production of an 18-subunit membrane-bound complex. J Biol Chem 2014;289:2873–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipscomb GL, Stirrett K, Schut GJ et al. Natural competence in the hyperthermophilic archaeon Pyrococcus furiosus facilitates genetic manipulation: construction of markerless deletions of genes encoding the two cytoplasmic hydrogenases. Appl Environ Microb 2011;77:2232–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Ding Y, Zhang R et al. Functional balance between enzymes in malonyl-CoA pathway for 3-hydroxypropionate biosynthesis. Metab Eng 2016;34:104–11. [DOI] [PubMed] [Google Scholar]
- Loder AJ, Han Y, Hawkins AB et al. Reaction kinetic analysis of the 3-hydroxypropionate/4-hydroxybutyrate CO2 fixation cycle in extremely thermoacidophilic archaea. Metab Eng 2016;38:446–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loder AJ, Zeldes BM, Conway JM et al. Extreme thermophiles as metabolic engineering platforms: strategies and current perspective. In: Liao CWJ. (ed.). Industrial Biotechnology. Wiley-VCH Verlag GmbH & Co. KGaA, 2017, 505–80. [Google Scholar]
- Lucas S, Toffin L, Zivanovic Y et al. Construction of a shuttle vector for, and spheroplast transformation of, the hyperthermophilic archaeon Pyrococcus abyssi. Appl Environ Microbiol 2002;68:5528–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lundberg KS, Shoemaker DD, Adams MW et al. High-fidelity amplification using a thermostable DNA polymerase isolated from Pyrococcus furiosus. Gene 1991;108:1–6. [DOI] [PubMed] [Google Scholar]
- McCarthy S, Johnson T, Pavlik BJ et al. Expanding the limits of thermoacidophily in the archaeon Sulfolobus solfataricus by adaptive evolution. Appl Environ Microb 2016;82:857–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maccioni E, Mariana P, Rustichelli F et al. X-ray diffraction structural analysis of Langmuir-Blodgett films using a pattern recognition approach. Thin Solid Films 1995;265:74–83. [Google Scholar]
- Machielsen R, Uria AR, Kengen SWM et al. Production and characterization of a thermostable alcohol dehydrogenase that belongs to the aldo-keto reductase superfamily. Appl Environ Microb 2006;72:233–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDonald JP, Hall A, Gasparutto D et al. Novel thermostable Y-family polymerases: applications for the PCR amplification of damaged or ancient DNAs. Nucleic Acids Res 2006;34:1102–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maezato Y, Johnson T, McCarthy S et al. Metal resistance and lithoautotrophy in the extreme thermoacidophile Metallosphaera sedula. J Bacteriol 2012;194:6856–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makarova KS, Haft DH, Barrangou R et al. Evolution and classification of the CRISPR–Cas systems. Nat Rev Microbiol 2011;9:467–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maniv I, Jiang W, Bikard D et al. Impact of different target sequences on type III CRISPR-Cas immunity. J Bacteriol 2016;198:941–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mardis ER. A decade's perspective on DNA sequencing technology. Nature 2011;470:198–203. [DOI] [PubMed] [Google Scholar]
- Marteinsson VT, Birrien JL, Reysenbach AL et al. Thermococcus barophilus sp. nov., a new barophilic and hyperthermophilic archaeon isolated under high hydrostatic pressure from a deep-sea hydrothermal vent. Int J Syst Bacteriol 1999;49:351–9. [DOI] [PubMed] [Google Scholar]
- Martin del Campo JS, Rollin J, Myung S et al. High-yield production of dihydrogen from xylose by using a synthetic enzyme cascade in a cell-free system. Angew Chem Int Ed 2013;52:4587–90. [DOI] [PubMed] [Google Scholar]
- Matsumi R, Manabe K, Fukui T et al. Disruption of a sugar transporter gene cluster in a hyperthermophilic archaeon using a host-marker system based on antibiotic resistance. J Bacteriol 2007;189:2683–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattila P, Korpela J, Tenkanen T et al. Fidelity of DNA synthesis by the Thermococcus litoralis DNA polymerase–an exttemely heat stable enzyme with proofreading activity. Nucleic Acids Res 1991;19:4967–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merone L, Mandrich L, Porzio E et al. Improving the promiscuous nerve agent hydrolase activity of a thermostable archaeal lactonase. Bioresource Technol 2010;101:9204–12. [DOI] [PubMed] [Google Scholar]
- Merone L, Mandrich L, Rossi M et al. A thermostable phosphotriesterase from the archaeon Sulfolobus solfataricus: cloning, overexpression and properties. Extremophiles 2005;9:297–305. [DOI] [PubMed] [Google Scholar]
- Metzker ML. Sequencing technologies - the next generation. Nat Rev Genet 2010;11:31–46. [DOI] [PubMed] [Google Scholar]
- Mills KV, Johnson MA, Perler FB. Protein splicing: how inteins escape from precursor proteins. J Biol Chem 2014;289:14498–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittler R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 2002;7:405–10. [DOI] [PubMed] [Google Scholar]
- Mochizuki T, Sako Y, Prangishvili D. Provirus induction in hyperthermophilic archaea: characterization of Aeropyrum pernix spindle-shaped virus 1 and Aeropyrum pernix ovoid virus 1. J Bacteriol 2011;193:5412–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mondorf S, Deppenmeier U, Welte C. A novel inducible protein production system and neomycin resistance as selection marker for Methanosarcina mazei. Archaea 2012;2012:973743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore BC, Leigh JA. Markerless mutagenesis in Methanococcus maripaludis demonstrates roles for alanine dehydrogenase, alanine racemase, and alanine permease. J Bacteriol 2005;187:972–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moracci M, Ciaramella M, Rossi M. Beta-glycosidase from Sulfolobus solfataricus. Methods Enzymol 2001;330:201–15. [DOI] [PubMed] [Google Scholar]
- Morales M, Arancibia J, Lemus M et al. Bio-oxidation of H2S by Sulfolobus metallicus. Biotechnol Lett 2011;33:2141–5. [DOI] [PubMed] [Google Scholar]
- Morales M, Silva J, Morales P et al. Biofiltration of hydrogen sulfide by Sulfolobus metallicus at high temperatures. Water Sci Technol 2012;66:1958–61. [DOI] [PubMed] [Google Scholar]
- Mousaei M, Deng L, She Q et al. Major and minor crRNA annealing sites facilitate low stringency DNA protospacer binding prior to Type I-A CRISPR-Cas interference in Sulfolobus. RNA Biol 2016;13:1166–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moustafa HMA, Kim EJ, Zhu ZG et al. Water splitting for high-yield hydrogen production energized by biomass xylooligosaccharides catalyzed by an enzyme cocktail. ChemCatChem 2016;8:2898–902. [Google Scholar]
- Mukund S, Adams MW. Characterization of a tungsten-iron-sulfur protein exhibiting novel spectroscopic and redox properties from the hyperthermophilic archaebacterium Pyrococcus furiosus. J Biol Chem 1990;265:11508–16. [PubMed] [Google Scholar]
- Mukund S, Adams MW. Molybdenum and vanadium do not replace tungsten in the catalytically active forms of the three tungstoenzymes in the hyperthermophilic archaeon Pyrococcus furiosus. J Bacteriol 1996;178:163–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller FH, Bandeiras TM, Urich T et al. Coupling of the pathway of sulphur oxidation to dioxygen reduction: characterization of a novel membrane-bound thiosulphate:quinone oxidoreductase. Mol Microbiol 2004;53:1147–60. [DOI] [PubMed] [Google Scholar]
- Mullis K, Faloona F, Scharf S et al. Specific enzymatic amplification of DNA in vitro: the polymerase chain reaction. Cold Spring Harb Symp Quant Biol 1986;51(Pt 1):263–73. [DOI] [PubMed] [Google Scholar]
- Muyzer G, Stams AJM. The ecology and biotechnology of sulphate-reducing bacteria. Nat Rev Microbiol 2008;6:441–54. [DOI] [PubMed] [Google Scholar]
- Myung S, Rollin J, You C et al. In vitro metabolic engineering of hydrogen production at theoretical yield from sucrose. Metab Eng 2014;24:70–7. [DOI] [PubMed] [Google Scholar]
- Nguyen DM, Lipscomb GL, Schut GJ et al. Temperature-dependent acetoin production by Pyrococcus furiosus is catalyzed by a biosynthetic acetolactate synthase and its deletion improves ethanol production. Metab Eng 2016;34:71–9. [DOI] [PubMed] [Google Scholar]
- Nishida H, Kiyonari S, Ishino Y et al. The closed structure of an archaeal DNA ligase from Pyrococcus furiosus. J Mol Biol 2006;360:956–67. [DOI] [PubMed] [Google Scholar]
- Nishitani Y, Yoshida S, Fujihashi M et al. Structure-based catalytic optimization of a type III Rubisco from a hyperthermophile. J Biol Chem 2010;285:39339–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nordstrom DK, Ball JW, McClesky RB. Ground water to surface water: chemistry of thermal outflows in Yellowstone National Park. Geothermal biology and geochemistry in Yellowstone National Park, US Geological Survey, 2005, 73–94. [Google Scholar]
- Norris PR. Selection of thermophiles for base metal sulfide concentrate leaching, Part II: nickel-copper and nickel concentrates. Miner Eng 2017;106:13–17. [Google Scholar]
- Norris PR, Calvo-Bado La, Brown CF et al. Ore column leaching with thermophiles: I, copper sulfide ore. Hydrometallurgy 2012;127-128:62–9. [Google Scholar]
- Norris PR, Laigle L, Ogden TJ et al. Selection of thermophiles for base metal sulfide concentrate leaching, Part I: effect of temperature on copper concentrate leaching and silver recovery. Miner Eng 2017;106:7–12. [Google Scholar]
- Notomista E, Falanga A, Fusco S et al. The identification of a novel Sulfolobus islandicus CAMP-like peptide points to archaeal microorganisms as cell factories for the production of antimicrobial molecules. Microb Cell Fact 2015;14:126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novikova O, Topilina N, Belfort M. Enigmatic distribution, evolution, and function of inteins. J Biol Chem 2014;289:14490–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ober J. Material Flows of Sulfur. US Geological Survey; 2002. [Google Scholar]
- Okano H, Baba M, Kawato K et al. High sensitive RNA detection by one-step RT-PCR using the genetically engineered variant of DNA polymerase with reverse transcriptase activity from hyperthermophilies. J Biosci Bioeng 2017;125:275–81. [DOI] [PubMed] [Google Scholar]
- Olsen GJ, Woese CR, Overbeek R. The winds of (evolutionary) change: breathing new life into microbiology. J Bacteriol 1994;176:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olzmann JA, Brown K, Wilkinson KD et al. Familial Parkinson's disease-associated L166P mutation disrupts DJ-1 protein folding and function. J Biol Chem 2004;279:8506–15. [DOI] [PubMed] [Google Scholar]
- Omri A, Agnew BJ, Patel GB. Short-term repeated-dose toxicity profile of archaeosomes administered to mice via intravenous and oral routes. Int J Toxicol 2003;22:9–23. [DOI] [PubMed] [Google Scholar]
- Palackal N, Brennan Y, Callen WN et al. An evolutionary route to xylanase process fitness. Protein Sci 2004;13:494–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paper W, Jahn U, Hohn MJ et al. Ignicoccus hospitalis sp. nov., the host of 'Nanoarchaeum equitans'. Int J Syst Evol Micr 2007;57:803–8. [DOI] [PubMed] [Google Scholar]
- Paques 2017. PAQUES - Biological and Wastewater Treatment.
- Parmentier J, Becker MM, Heintz U et al. Stability of liposomes containing bio-enhancers and tetraether lipids in simulated gastro-intestinal fluids. Int J Pharm 2011a;405:210–7. [DOI] [PubMed] [Google Scholar]
- Parmentier J, Thewes B, Gropp F et al. Oral peptide delivery by tetraether lipid liposomes. Int J Pharm 2011b;415:150–7. [DOI] [PubMed] [Google Scholar]
- Pascal JM, Tsodikov OV, Hura GL et al. A flexible interface between DNA ligase and PCNA supports conformational switching and efficient ligation of DNA. Mol Cell 2006;24:279–91. [DOI] [PubMed] [Google Scholar]
- Patel GB, Agnew BJ, Deschatelets L et al. In vitro assessment of archaeosome stability for developing oral delivery systems. Int J Pharm 2000;194:39–49. [DOI] [PubMed] [Google Scholar]
- Patel GB, Chen W. Archaeal lipid mucosal vaccine adjuvant and delivery system. Expert Rev Vaccines 2010;9:431–40. [DOI] [PubMed] [Google Scholar]
- Patel GB, Sprott GD. Archaeobacterial ether lipid liposomes (archaeosomes) as novel vaccine and drug delivery systems. Crit Rev Biotechnol 1999;19:317–57. [DOI] [PubMed] [Google Scholar]
- Patwardhan AP, Thompson DH. Novel flexible and rigid tetraether acyclic and macrocyclic bisphosphocholines: synthesis and monolayer properties. Langmuir 2000;16:10340–50. [Google Scholar]
- Pavlov AR, Belova GI, Kozyavkin SA et al. Helix-hairpin-helix motifs confer salt resistance and processivity on chimeric DNA polymerases. Proc Natl Acad Sci USA 2002;99:13510–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavlov AR, Pavlova NV, Kozyavkin SA et al. Recent developments in the optimization of thermostable DNA polymerases for efficient applications. Trends Biotechnol 2004;22:253–60. [DOI] [PubMed] [Google Scholar]
- Peeples TL, Kelly RM. Bioenergetics of the metal/sulfur-oxidizing extreme thermoacidophile, Metallosphaera sedula. Fuel 1993;72:1619–24. [Google Scholar]
- Peiru S, Aguirre A, Eberhardt F et al. An industrial scale process for the enzymatic removal of steryl glucosides from biodiesel. Biotechnol Biofuels 2015;8:223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng N, Deng L, Mei Y et al. A synthetic arabinose-inducible promoter confers high levels of recombinant protein expression in hyperthermophilic archaeon Sulfolobus islandicus. Appl Environ Microbiol 2012;78:5630–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng N, Han W, Li Y et al. Genetic technologies for extremely thermophilic microorganisms of Sulfolobus, the only genetically tractable genus of crenarchaea. Sci China Life Sci 2017a;60:370–85. [DOI] [PubMed] [Google Scholar]
- Peng S, Chu Z, Lu J et al. Heterologous expression of chaperones from hyperthermophilic archaea inhibits aminoglycoside-induced protein misfolding in Escherichia coli. Biochemistry (Mosc) 2017b;82:1169–75. [DOI] [PubMed] [Google Scholar]
- Peng W, Feng M, Feng X et al. An archaeal CRISPR type III-B system exhibiting distinctive RNA targeting features and mediating dual RNA and DNA interference. Nucleic Acids Res 2015;43:406–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perler FB, Comb DG, Jack WE et al. Intervening sequences in an Archaea DNA polymerase gene. Proc Natl Acad Sci USA 1992;89:5577–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrova T, Bezsudnova EY, Boyko KM et al. ATP-dependent DNA ligase from Thermococcus sp. 1519 displays a new arrangement of the OB-fold domain. Acta Crystallogr F 2012a;68:1440–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrova TE, Bezsudnova EY, Dorokhov BD et al. Expression, purification, crystallization and preliminary crystallographic analysis of a thermostable DNA ligase from the archaeon Thermococcus sibiricus. Acta Crystallogr F 2012b;68:163–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prangishvili D, Bamford DH, Forterre P et al. The enigmatic archaeal virosphere. Nat Rev Microbiol 2017;15:724–39. [DOI] [PubMed] [Google Scholar]
- Prangishvili D, Garrett RA, Koonin EV. Evolutionary genomics of archaeal viruses: unique viral genomes in the third domain of life. Virus Res 2006;117:52–67. [DOI] [PubMed] [Google Scholar]
- Prangishvili DA, Vashakidze RP, Chelidze MG et al. A restriction endonuclease Sua I from the thermoacidophilic archaebacterium Sulfolobus acidocaldarius. FEBS Lett 1985;192:57–60. [DOI] [PubMed] [Google Scholar]
- Pritchett MA, Zhang JK, Metcalf WW. Development of a markerless genetic exchange method for Methanosarcina acetivorans c2a and its use in construction of new genetic tools for methanogenic archaea. Appl Environ Microb 2004;70:1425–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puyol D, Batstone DJ, Hülsen T et al. Resource recovery from wastewater by biological technologies: opportunities, challenges, and prospects. Front Microbiol 2016;7:2106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quatrini R, Appia-Ayme C, Denis Y et al. Extending the models for iron and sulfur oxidation in the extreme acidophile Acidithiobacillus ferrooxidans. BMC Genomics 2009;10:394–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawlings DE. Heavy metal mining using microbes. Annu Rev Microbiol 2002;56:65–91. [DOI] [PubMed] [Google Scholar]
- Remy B, Plener L, Poirier L et al. Harnessing hyperthermostable lactonase from Sulfolobus solfataricus for biotechnological applications. Sci Rep 2016;6:37780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ren X, Liu K, Zhang Q et al. Design, fabrication, and characterization of archaeal tetraether free-standing planar membranes in a PDMS- and PCB-based fluidic platform. ACS Appl Mater Inter 2014;6:12618–28. [DOI] [PubMed] [Google Scholar]
- Rensen EI, Mochizuki T, Quemin E et al. A virus of hyperthermophilic archaea with a unique architecture among DNA viruses. Proc Natl Acad Sci USA 2016;113:2478–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Réthoré G, Montier T, Le Gall T et al. Archaeosomes based on synthetic tetraether-like lipids as novel versatile gene delivery systems. Chem Commun 2007;20:2054–6. [DOI] [PubMed] [Google Scholar]
- Richardson TH, Tan X, Frey G et al. A novel, high performance enzyme for starch liquefaction. J Biol Chem 2002;277:26501–7. [DOI] [PubMed] [Google Scholar]
- Rittmann SK, Lee HS, Lim JK et al. One-carbon substrate-based biohydrogen production: microbes, mechanism, and productivity. Biotechnol Adv 2015;33:165–77. [DOI] [PubMed] [Google Scholar]
- Rittmann SKMR. A critical assessment of microbiological biogas to biomethane upgrading systems. In: Guebitz GM, Bauer A, Bochmann G. et al (eds) Biogas Science and Technology 2015;151:117–35. [DOI] [PubMed] [Google Scholar]
- Robb FT, Maeder DL, Brown JR et al. Genomic sequence of hyperthermophile, Pyrococcus furiosus: implications for physiology and enzymology. Meth Enzymol 2001;330:134–57. [DOI] [PubMed] [Google Scholar]
- Rolland JL, Gueguen Y, Persillon C et al. Characterization of a thermophilic DNA ligase from the archaeon Thermococcus fumicolans. FEMS Microbiol Lett 2004;236:267–73. [DOI] [PubMed] [Google Scholar]
- Rollin JA, Martin del Campo J, Myung S et al. High-yield hydrogen production from biomass by in vitro metabolic engineering: mixed sugars coutilization and kinetic modeling. Proc Natl Acad Sci USA 2015;112:4964–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romano P, Blázquez ML, Alguacil FJ et al. Comparative study on the selective chalcopyrite bioleaching of a molybdenite concentrate with mesophilic and thermophilic bacteria. FEMS Microbiol Lett 2001;196:71–5. [DOI] [PubMed] [Google Scholar]
- Sanchez-Andrea I, Stams AJM, Weijma J et al. A case in support of implementing innovative bio-processes in the metal mining industry. FEMS Microbiol Lett 2016;363:fnw106. [DOI] [PubMed] [Google Scholar]
- Sandström Å, Petersson S. Bioleaching of a complex sulphide ore with moderate thermophilic and extreme thermophilic microorganisms. Hydrometallurgy 1997;46:181–90. [Google Scholar]
- Santangelo TJ, Cubonova L, Reeve JN. Shuttle vector expression in Thermococcus kodakaraensis: contributions of cis elements to protein synthesis in a hyperthermophilic archaeon. Appl Environ Microb 2008;74:3099–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santangelo TJ, Cubonova L, Reeve JN. Thermococcus kodakarensis genetics: TK1827-Encoded beta-glycosidase, new positive-selection protocol, and targeted and repetitive deletion technology. Appl Environ Microb 2010;76:1044–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santangelo TJ, Cubonova L, Reeve JN. Deletion of alternative pathways for reductant recycling in Thermococcus kodakarensis increases hydrogen production. Mol Microbiol 2011;81:897–911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santangelo TJ, Reeve JN. Genetic tools and manipulations of the hyperthermophilic heterotrophic archaeon Thermococcus kodakarensis. In: Horikoshi K (ed.) Extremophiles Handbook. Japan, Tokyo: Springer, 2011, 567–82. [Google Scholar]
- Santos AA, Venceslau SS, Grein F et al. A protein trisulfide couples dissimilatory sulfate reduction to energy conservation. Science 2015;350:1541–5. [DOI] [PubMed] [Google Scholar]
- Sapra R, Bagramyan K, Adams MW. A simple energy-conserving system: proton reduction coupled to proton translocation. Proc Natl Acad Sci USA 2003;100:7545–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato T, Atomi H, Imanaka T. Archaeal type III RuBisCOs function in a pathway for AMP metabolism. Science 2007;315:1003–6. [DOI] [PubMed] [Google Scholar]
- Sato T, Fukui T, Atomi H et al. Targeted gene disruption by homologous recombination in the hyperthermophilic archaeon Thermococcus kodakaraensis KOD1. J Bacteriol 2003;185:210–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato T, Fukui T, Atomi H et al. Improved and versatile transformation system allowing multiple genetic manipulations of the hyperthermophilic archaeon Thermococcus kodakaraensis. Appl Environ Microb 2005;71:3889–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schelert J, Dixit V, Hoang V et al. Occurrence and characterization of mercury resistance in the hyperthermophilic archaeon sulfolobus solfataricus by use of gene disruption. J Bacteriol 2004;186:427–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schicho RN, Ma K, Adams MW et al. Bioenergetics of sulfur reduction in the hyperthermophilic archaeon Pyrococcus furiosus. J Bacteriol 1993;175:1823–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schippers A, Hedrich S, Vasten J et al. Biomining: metal recovery from ores with microorganisms. Adv Biochem Eng Biotechnol 2014;141:1–47. [DOI] [PubMed] [Google Scholar]
- Schut GJ, Bridger SL, Adams MWW. Insights into the metabolism of elemental sulfur by the hyperthermophilic archaeon Pyrococcus furiosus: characterization of a coenzyme A- dependent NAD(P)H sulfur oxidoreductase. J Bacteriol 2007;189:4431–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schut GJ, Boyd ES, Peters JW et al. The modular respiratory complexes involved in hydrogen and sulfur metabolism by heterotrophic hyperthermophilic archaea and their evolutionary implications. FEMS Microbiol Rev 2013;37:182–203. [DOI] [PubMed] [Google Scholar]
- Schut GJ, Lipscomb GL, Han Y et al. The order thermococcales and the family thermococcaceae. In: Rosenberg E, DeLong EF, Lory S. et al. (eds). The Prokaryotes. Berlin Heidelberg: Springer, 2014, 363–83. [Google Scholar]
- Schut GJ, Lipscomb GL, Nguyen DM et al. Heterologous production of an energy-conserving carbon monoxide dehydrogenase complex in the hyperthermophile Pyrococcus furiosus. Front Microbiol 2016a;7:29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schut GJ, Nixon WJ, Lipscomb GL et al. Mutational analyses of the enzymes involved in the metabolism of hydrogen by the hyperthermophilic archaeon Pyrococcus furiosus. Front Microbiol 2012;3:163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schut GJ, Zadvornyy O, Wu CH et al. The role of geochemistry and energetics in the evolution of modern respiratory complexes from a proton-reducing ancestor. BBA-Bioenergetics 2016b;1857:958–70. [DOI] [PubMed] [Google Scholar]
- Segerer A, Neuner A, Kristjansson JK et al. Acidianus infernus Gen. nov., Sp. nov., and Acidianus brierleyi Comb. nov.: facultatively aerobic, extremely acidophilic thermophilic sulfur-metabolizing archaebacteria. Int J Syst Bacteriol 1986;36:559–64. [Google Scholar]
- Sekar N, Wu C-H, Adams MWW et al. Electricity generation by Pyrococcus furiosus in microbial fuel cells operated at 90°C. Biotechnol Bioeng 2017;114:1419–27. [DOI] [PubMed] [Google Scholar]
- Selig M, Schonheit P. Oxidation of organic compounds to CO2 with sulfur or thiosulfate as electron acceptor in the anaerobic hyperthermophilic archaea Thermoproteus tenax and Pyrobaculum islandicum proceeds via the citric acid cycle. Arch Microbiol 1994;162:286–94. [Google Scholar]
- Seo MS, Kim YJ, Choi JJ et al. Cloning and expression of a DNA ligase from the hyperthermophilic archaeon Staphylothermus marinus and properties of the enzyme. J Biotechnol 2007;128:519–30. [DOI] [PubMed] [Google Scholar]
- Sevcenco AM, Paravidino M, Vrouwenvelder JS et al. Phosphate and arsenate removal efficiency by thermostable ferritin enzyme from Pyrococcus furiosus using radioisotopes. Water Res 2015;76:181–6. [DOI] [PubMed] [Google Scholar]
- Shah NH, Muir TW. Inteins: nature's gift to protein chemists. Chem Sci 2014;5:446–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw A, Bott R, Day AG. Protein engineering of alpha-amylase for low pH performance. Curr Opin Biotechnol 1999;10:349–52. [DOI] [PubMed] [Google Scholar]
- She Q, Singh RK, Confalonieri F et al. The complete genome of the crenarchaeon Sulfolobus solfataricus P2. Proc Natl Acad Sci USA 2001;98:7835–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shea MT, Walter ME, Duszenko N et al. pNEB193-derived suicide plasmids for gene deletion and protein expression in the methane-producing archaeon, Methanosarcina acetivorans. Plasmid 2016;84-85:27–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiimori M, Garrett SC, Chambers DP et al. Role of free DNA ends and protospacer adjacent motifs for CRISPR DNA uptake in Pyrococcus furiosus. Nucleic Acids Res 2017;45:11281–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin KC, Choi HY, Seo MJ et al. Compound K production from red ginseng extract by beta-Glycosidase from Sulfolobus solfataricus supplemented with alpha-L-Arabinofuranosidase from Caldicellulosiruptor saccharolyticus. PLoS One 2015;10:e0145876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin K-C, Choi H-Y, Seo M-J et al. Improved conversion of ginsenoside Rb1 to compound K by semi-rational design of Sulfolobus solfataricus β-glycosidase. AMB Express 2017;7:186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin KC, Nam HK, Oh DK. Hydrolysis of flavanone glycosides by beta-Glucosidase from Pyrococcus furiosus and its application to the production of flavanone aglycones from citrus extracts. J Agr Food Chem 2013;61:11532–40. [DOI] [PubMed] [Google Scholar]
- Siebers B, Zaparty M, Raddatz G et al. The complete genome sequence of thermoproteus tenax: a physiologically versatile member of the Crenarchaeota. PLoS One 2011;6:e24222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon K, Rittman MR. A critical assessment of microbiological biogas to biomethane upgrading systems. In: Guebitz GMBauer A, Bochmann G et al. (eds). Biogas Science and Technology. Cham: Springer International Publishing, 2015; 151:117–35. [DOI] [PubMed] [Google Scholar]
- Smagin VA, Mardanov AV, Bonch-Osmolovskaia EA et al. Isolation and characteristics of new thermostable DNA ligase from archaea of the genus Thermococcus. Prikl Biokhim Mikrobiol 2008;44:523–8. [PubMed] [Google Scholar]
- Solanki K, Abdallah W, Banta S. Extreme makeover: Engineering the activity of a thermostable alcohol dehydrogenase (AdhD) from Pyrococcus furiosus. Biotechnol J 2016;11:1483–97. [DOI] [PubMed] [Google Scholar]
- Solbak AI, Richardson TH, McCann RT et al. Discovery of pectin-degrading enzymes and directed evolution of a novel pectate lyase for processing cotton fabric. J Biol Chem 2005;280:9431–8. [DOI] [PubMed] [Google Scholar]
- Sollner S, Berkner S, Lipps G. Characterisation of the novel restriction endonuclease SuiI from Sulfolobus islandicus. Extremophiles 2006;10:629–34. [DOI] [PubMed] [Google Scholar]
- Sondergaard D, Pedersen CN, Greening C. HydDB: A web tool for hydrogenase classification and analysis. Sci Rep 2016;6:34212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprott GD. Structures of archaebacterial membrane lipids. J Bioenerg Biomembr 1992;24:555–66. [DOI] [PubMed] [Google Scholar]
- Sprott GD, Tolson DL, Patel GB. Archaeosomes as novel antigen delivery systems. FEMS Microbiol Lett 1997;154:17–22. [DOI] [PubMed] [Google Scholar]
- Sriskanda V, Kelman Z, Hurwitz J et al. Characterization of an ATP-dependent DNA ligase from the thermophilic archaeon Methanobacterium thermoautotrophicum. Nucleic Acids Res 2000;28:2221–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinmetz NF, Bize A, Findlay KC et al. Site-Specific and spatially controlled addressability of a new viral nanobuilding block: Sulfolobus islandicus rod-shaped virus 2. Adv Funct Mater 2008;18:3478–86. [Google Scholar]
- Stetter KO. Hyperthermophilic procaryotes. FEMS Microbiol Rev 1996;18:149–58. [Google Scholar]
- Stetter KO, Lauerer G, Thomm M et al. Isolation of extremely thermophilic sulfate reducers: evidence for a novel branch of Archaebacteria. Science 1987;236:822–4. [DOI] [PubMed] [Google Scholar]
- Stetter KO, Thomm M, Winter J et al. Methanothermus fervidus, sp. nov., a novel extremely thermophilic methanogen isolated from an Icelandic hot spring. Zbl Bakt Mik Hyg I C 1981;2:166–78. [Google Scholar]
- Strauss G, Fuchs G. Enzymes of a novel autotrophic CO2 fixation pathway in the phototrophic bacterium Chloroflexus aurantiacus, the 3-hydroxypropionate cycle. Eur J Biochem 1993;215:633–43. [DOI] [PubMed] [Google Scholar]
- Supangat S, An YJ, Sun Y et al. Purification, crystallization and preliminary crystallographic analysis of a multiple cofactor-dependent DNA ligase from Sulfophobococcus zilligii. Acta Crystallogr F 2010;66:1583–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki S, Kurosawa N. Disruption of the gene encoding restriction endonuclease SuaI and development of a host–vector system for the thermoacidophilic archaeon Sulfolobus acidocaldarius. Extremophiles 2016;20:139–48. [DOI] [PubMed] [Google Scholar]
- Suzuki S, Kurosawa N. Development of the multiple gene knockout system with One-Step PCR in Thermoacidophilic Crenarchaeon Sulfolobus acidocaldarius. Archaea 2017;2017:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swarts DC, Hegge JW, Hinojo I et al. Argonaute of the archaeon Pyrococcus furiosus is a DNA-guided nuclease that targets cognate DNA. Nucleic Acids Res 2015;43:5120–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szoka F Jr, Papahadjopoulos D. Comparative properties and methods of preparation of lipid vesicles (liposomes). Annu Rev Biophys Bioeng 1980;9:467–508. [DOI] [PubMed] [Google Scholar]
- Takacs M, Toth A, Bogos B et al. Formate hydrogenlyase in the hyperthermophilic archaeon, Thermococcus litoralis. BMC Microbiol 2008;8:88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takagi M, Nishioka M, Kakihara H et al. Characterization of DNA polymerase from Pyrococcus sp. strain KOD1 and its application to PCR. Appl Environ Microb 1997;63:4504–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takatsugi K, Sasaki K, Hirajima T. Mechanism of the enhancement of bioleaching of copper from enargite by thermophilic iron-oxidizing archaea with the concomitant precipitation of arsenic. Hydrometallurgy 2011;109:90–6. [Google Scholar]
- Takemasa R, Yokooji Y, Yamatsu A et al. Thermococcus kodakarensis as a host for gene expression and protein secretion. Appl Environ Microb 2011;77:2392–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanabe M, Ishino S, Ishino Y et al. Mutations of Asp540 and the domain-connecting residues synergistically enhance Pyrococcus furiosus DNA ligase activity. FEBS Lett 2014;588:230–5. [DOI] [PubMed] [Google Scholar]
- Tanabe M, Ishino S, Yohda M et al. Structure-based mutational study of an archaeal DNA ligase towards improvement of ligation activity. ChemBioChem 2012;13:2575–82. [DOI] [PubMed] [Google Scholar]
- Tanaka T, Fujiwara S, Nishikori S et al. A unique chitinase with dual active sites and triple substrate binding sites from the hyperthermophilic archaeon Pyrococcus kodakaraensis KOD1. Appl Environ Microb 1999;65:5338–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka T, Fukui T, Atomi H et al. Characterization of an Exo-beta-D-Glucosaminidase involved in a novel chitinolytic pathway from the hyperthermophilic archaeon Thermococcus kodakaraensis KOD1. J Bacteriol 2003;185:5175–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tetter S, Hilvert D. Enzyme encapsulation by a ferritin cage. Angew Chem Int Ed 2017;56:14933–6. [DOI] [PubMed] [Google Scholar]
- Thauer RK, Kaster AK, Seedorf H et al. Methanogenic archaea: ecologically relevant differences in energy conservation. Nat Rev Microbiol 2008;6:579–1. [DOI] [PubMed] [Google Scholar]
- Thiel A, Michoud G, Moalic Y et al. Genetic manipulations of the hyperthermophilic piezophilic archaeon Thermococcus barophilus. Appl Environ Microb 2014;80:2299–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thor S, Peterson JR, Luthey-Schulten Z. Genome-scale metabolic modeling of archaea lends insight into diversity of metabolic function. Archaea 2017;2017:1–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tolson DL, Latta RK, Patel GB et al. Uptake of archaeobacterial liposomes and conventional liposomes by phagocytic cells. J Liposome Res 1996;6:755–76. [Google Scholar]
- Tomkinson AE, Vijayakumar S, Pascal JM et al. DNA ligases: structure, reaction mechanism, and function. Chem Rev 2006;106:687–99. [DOI] [PubMed] [Google Scholar]
- Tomoaia-Cotisel M, Chifu E, Zsako J et al. Monolayer properties of archaeol and caldarchaeol polar lipids of a methanogenic archaebacterium, Methanospirillum hungatei, at the air/water interface. Chem Phys Lipids 1992;63:131–8. [DOI] [PubMed] [Google Scholar]
- Topçuoğlu BD, Stewart LC, Morrison HG et al. Hydrogen limitation and syntrophic growth among natural assemblages of thermophilic methanogens at deep-sea hydrothermal vents. Front Microbiol 2016;7:1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topilina NI, Novikova O, Stanger M et al. Post-translational environmental switch of RadA activity by extein–intein interactions in protein splicing. Nucleic Acids Res 2015;43:6631–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torchia C, Takagi Y, Ho CK. Archaeal RNA ligase is a homodimeric protein that catalyzes intramolecular ligation of single-stranded RNA and DNA. Nucleic Acids Res 2008;36:6218–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsao J-H, Kaneshiro SM, Yu S-S et al. Continuous culture of Methanococcus jannaschii, an extremely thermophilic methanogen. Biotechnol Bioeng 1994;43:258–61. [DOI] [PubMed] [Google Scholar]
- Uhl P, Helm F, Hofhaus G et al. A liposomal formulation for the oral application of the investigational hepatitis B drug Myrcludex B. Eur J Pharm Biopharm 2016;103:159–66. [DOI] [PubMed] [Google Scholar]
- Ulas T, Riemer SA, Zaparty M et al. Genome-scale reconstruction and analysis of the metabolic network in the hyperthermophilic archaeon Sulfolobus solfataricus. PLoS One 2012;7:e43401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uldahl KB, Walk ST, Olshefsky SC et al. SMV1, an extremely stable thermophilic virus platform for nanoparticle trafficking in the mammalian GI tract. J Appl Microbiol 2017;123:1286–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uldahl KB, Wu L, Hall A et al. Recognition of extremophilic archaeal viruses by eukaryotic cells: a promising nanoplatform from the third domain of life. Sci Rep 2016;6:37966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulrih NP, Gmajner D, Raspor P. Structural and physicochemical properties of polar lipids from thermophilic archaea. Appl Microbiol Biot 2009;84:249–60. [DOI] [PubMed] [Google Scholar]
- Vaccaro BJ, Clarkson SM, Holden JF et al. Biological iron-sulfur storage in a thioferrate-protein nanoparticle. Nat Commun 2017;8:16110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Aswegen PC, van Niekerk J, Olivier W. The BIOX Process for the Treatment of Refractory Gold Concentrates. Rawlings DE, Johnson DB (eds.) Berlin:Springer, 2007, 1–34. [Google Scholar]
- van Wolferen M, Wagner A, van der Does C et al. The archaeal Ced system imports DNA. Proc Natl Acad Sci USA 2016;113:2496–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VanDyke DJ, Wu J, Logan SM et al. Identification of genes involved in the assembly and attachment of a novel flagellin N-linked tetrasaccharide important for motility in the archaeon Methanococcus maripaludis. Mol Microbiol 2009;72:633–44. [DOI] [PubMed] [Google Scholar]
- Veesler D, Ng T-S, Sendamarai AK et al. Atomic structure of the 75 MDa extremophile Sulfolobus turreted icosahedral virus determined by CryoEM and X-ray crystallography. Proc Natl Acad Sci USA 2013;110:5504–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ver Eecke HC, Butterfield DA, Huber JA et al. Hydrogen-limited growth of hyperthermophilic methanogens at deep-sea hydrothermal vents. Proc Natl Acad Sci USA 2012;109:13674–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verhaart MR, Bielen AA, van der Oost J et al. Hydrogen production by hyperthermophilic and extremely thermophilic bacteria and archaea: mechanisms for reductant disposal. Environ Technol 2010;31:993–1003. [DOI] [PubMed] [Google Scholar]
- Vignais PM, Billoud B. Occurrence, classification, and biological function of hydrogenases: an overview. Chem Rev 2007;107:4206–72. [DOI] [PubMed] [Google Scholar]
- Vukovic M, Strbac N, Sokic M et al. Bioleaching of pollymetallic sulphide concentrate using thermophilic bacteria. Hem Ind 2014;68:575–83. [Google Scholar]
- Wachtershauser G. Before enzymes and templates - theory of surface metabolism. Microbiol Rev 1988;52:452–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waege I, Schmid G, Thumann S et al. Shuttle vector-based transformation system for Pyrococcus furiosus. Appl Environ Microb 2010;76:3308–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner A, Whitaker RJ, Krause DJ et al. Mechanisms of gene flow in archaea. Nat Rev Microbiol 2017;15:492–501. [DOI] [PubMed] [Google Scholar]
- Wagner M, Berkner S, Ajon M et al. Expanding and understanding the genetic toolbox of the hyperthermophilic genus Sulfolobus. Biochm Soc Trans 2009;37:97–101. [DOI] [PubMed] [Google Scholar]
- Wagner M, van Wolferen M, Wagner A et al. Versatile genetic tool box for the crenarchaeote Sulfolobus acidocaldarius. Front Microbiol 2012;3:214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker JE, Luyties O, Santangelo TJ. Factor-dependent archaeal transcription termination. Proc Natl Acad Sci USA 2017;114:E6767–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Zhou QH, Chen HY et al. Efficient solubilization, purification of recombinant extracellular alpha-amylase from Pyrococcus furiosus expressed as inclusion bodies in Escherichia coli. J Ind Microbiol Biot 2007;34:187–92. [DOI] [PubMed] [Google Scholar]
- Wang P, Wang PL, Tian J et al. A new strategy to express the extracellular alpha-amylase from Pyrococcus furiosus in Bacillus amyloliquefaciens. Sci Rep 2016;6:22229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Prosen DE, Mei L et al. A novel strategy to engineer DNA polymerases for enhanced processivity and improved performance in vitro. Nucleic Acids Res 2004;32:1197–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watling H. Microbiological advances in biohydrometallurgy. Minerals 2016;6:49. [Google Scholar]
- Weinberger AD, Wolf YI, Lobkovsky AE et al. Viral diversity threshold for adaptive immunity in prokaryotes. mBio 2012;3:e00456–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheaton G, Counts J, Mukherjee A et al. The confluence of heavy metal biooxidation and heavy metal resistance: implications for bioleaching by extreme thermoacidophiles. Minerals 2015;5:397–451. [Google Scholar]
- Whitfield J. Born in a watery commune. Nature 2004;427:674–6. [DOI] [PubMed] [Google Scholar]
- Wilson MA, Collins JL, Hod Y et al. The 1.1-A resolution crystal structure of DJ-1, the protein mutated in autosomal recessive early onset Parkinson's disease. Proc Natl Acad Sci USA 2003;100:9256–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson RH, Alonso H, Whitney SM. Evolving Methanococcoides burtonii archaeal Rubisco for improved photosynthesis and plant growth. Sci Rep 2016;6:22284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woese CR, Fox GE. Phylogenetic structure of the prokaryotic domain: the primary kingdoms. Proc Natl Acad Sci USA 1977;74:5088–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward J, Orr M, Cordray K et al. Enzymatic production of biohydrogen. Nature 2000;405:1014–5. [DOI] [PubMed] [Google Scholar]
- Worthington P, Hoang V, Perez-Pomares F et al. Targeted disruption of the α-amylase gene in the hyperthermophilic archaeon Sulfolobus solfataricus. J Bacteriol 2003;185:482–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CH, McTernan PM, Walter ME et al. Production and application of a soluble hydrogenase from Pyrococcus furiosus. Archaea 2015;2015:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H, Lipscomb GL, Keese AM et al. SurR regulates hydrogen production in Pyrococcus furiosus by a sulfur-dependent redox switch. Mol Microbiol 2010;77:1111–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye X, Wang Y, Hopkins RC et al. Spontaneous high-yield production of hydrogen from cellulosic materials and water catalyzed by enzyme cocktails. ChemSusChem 2009;2:149–52. [DOI] [PubMed] [Google Scholar]
- Ye XT, Honda K, Sakai T et al. Synthetic metabolic engineering-a novel, simple technology for designing a chimeric metabolic pathway. Microb Cell Fact 2012;11:120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokooji Y, Tomita H, Atomi H et al. Pantoate kinase and phosphopantothenate synthetase, two novel enzymes necessary for CoA biosynthesis in the Archaea. J Biol Chem 2009;284:28137–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- You C, Shi T, Li Y et al. An in vitro synthetic biology platform for the industrial biomanufacturing of myo-inositol from starch. Biotechnol Bioeng 2017;114:1855–64. [DOI] [PubMed] [Google Scholar]
- Zavec AB, Ota A, Zupancic T et al. Archaeosomes can efficiently deliver different types of cargo into epithelial cells grown in vitro. J Biotechnol 2014;192(Pt A):130–5. [DOI] [PubMed] [Google Scholar]
- Zebec Z, Manica A, Zhang J et al. CRISPR-mediated targeted mRNA degradation in the archaeon Sulfolobus solfataricus. Nucleic Acids Res 2014;42:5280–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zebec Z, Zink IA, Kerou M et al. Efficient CRISPR-mediated post-transcriptional gene silencing in a hyperthermophilic archaeon using multiplexed crRNA expression. G3 2016;6:3161–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeldes BM, Keller MW, Loder AJ et al. Extremely thermophilic microorganisms as metabolic engineering platforms for production of fuels and industrial chemicals. Front Microbiol 2015;6:1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng X, Birrien JL, Fouquet Y et al. Pyrococcus CH1, an obligate piezophilic hyperthermophile: extending the upper pressure-temperature limits for life. ISME J 2009;3:873-6. [DOI] [PubMed] [Google Scholar]
- Zhang C, Cooper TE, Krause DJ et al. Augmenting the genetic toolbox for Sulfolobus islandicus with a stringent positive selectable marker for agmatine prototrophy. Appl Environ Microb 2013;79:5539–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Tripathi A. Archaeal RNA ligase from Thermoccocus kodakarensis for template dependent ligation. RNA Biol 2017;14:36–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang YH, Evans BR, Mielenz JR et al. High-yield hydrogen production from starch and water by a synthetic enzymatic pathway. PLoS One 2007;2:e456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhelkovsky AM, McReynolds LA. Structure-function analysis of Methanobacterium thermoautotrophicum RNA ligase – engineering a thermostable ATP independent enzyme. BMC Mol Biol 2012;13:24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng T, Huang Q, Zhang C et al. Development of a simvastatin selection marker for a hyperthermophilic acidophile, Sulfolobus islandicus. Appl Environ Microb 2012;78:568–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Z, Kin Tam T, Sun F et al. A high-energy-density sugar biobattery based on a synthetic enzymatic pathway. Nat Commun 2014;5:3026. [DOI] [PubMed] [Google Scholar]
- Zillig W, Kletzin A, Schleper C et al. Screening for Sulfolobales, their plasmids and their viruses in icelandic solfataras. Syst Appl Microbiol 1993;16:609-28. [Google Scholar]
- Zillig W, Stetter KO, Wunderl S et al. The Sulfolobus-“Caldariella” group: Taxonomy on the basis of the structure of DNA-dependent RNA polymerases. Arch Microbiol 1980;125:259–69. [Google Scholar]
- Zimmermann P, Laska S, Kletzin A. Two modes of sulfite oxidation in the extremely thermophilic and acidophilic archaeon Acidianus ambivalens. Arch Microbiol 1999;172:76–82. [DOI] [PubMed] [Google Scholar]
- Zwickl P, Fabry S, Bogedain C et al. Glyceraldehyde-3-phosphate dehydrogenase from the hyperthermophilic archaebacterium Pyrococcus woesei: characterization of the enzyme, cloning and sequencing of the gene, and expression in Escherichia coli. J Bacteriol 1990;172:4329–38. [DOI] [PMC free article] [PubMed] [Google Scholar]