Abstract
Aim
The aim of the study was to evaluate costs associated with colonic endoscopic submucosal dissection (ESD) for treatment of colorectal cancer.
Methods
The study is a retrospective analysis of data on 395 patients treated by colonic ESD.
Results
The operation, consumable items, and medication accounted for 71% of the total costs for colonic ESD treatment. Medication and consumable items' costs were higher if lesions occurred in the transverse colon and right hemicolon compared to the left hemicolon. Medication, consumable items, and total costs were higher for larger lesions. Lesion numbers and carcinoma were associated with higher medication, consumable items, operation, and total costs. Positive surgical margins and complications of hemorrhage or perforation were positively correlated with higher costs for medication, consumable items, and total costs.
Conclusion
Labor costs for doctors and nurses remain low in China. Costs for medication and consumable items were higher for treatment involving the transverse colon or right hemicolon (vs. the left hemicolon), larger lesions, carcinoma, and a positive surgical margin. A benchmark cost estimate for ESD treatment including 4 days of postoperative hospitalization was determined to be approximately 5400 USD.
1. Introduction
Colorectal cancer is among the top three causes of cancer mortality and is commonly diagnosed in both Western and Eastern nations, including China [1]. Based upon data collected in the USA and Japan, current levels of colorectal cancer reflect a sharp increase in incidence. High costs of colorectal cancer treatment have, accordingly, dramatically increased the global healthcare economic burden [2]. Early stage colorectal cancer can be effectively treated using advanced endoscopic procedures, including endoscopic submucosal dissection (ESD) [3]. ESD is a very promising approach that delivers favorable short-term outcomes. Multiple procedures can be performed simultaneously. ESD is minimally invasive and results in high radical cure rates, removal of pathological tissues, and rapid recovery [4]. Compared to conventional endoscopic procedures such as endoscopic mucosal resection, ESD exhibits higher en bloc resection rates and lower recurrence rates for early stage gastric and colorectal cancer [5, 6]. At the present time, optimized cost-benefit ratios for hospitalization time with respect to pre-ESD preparation and post-ESD complications remain undefined. To fill this knowledge gap, we performed a retrospective analysis of data collected on nearly 400 patients in order to examine the cost-effectiveness of colonic ESD.
2. Materials and Methods
2.1. Patient Cohort
The study is based on retrospective analysis of data from 395 patients undergoing colonic ESD between January 2015 and December 2017 at the Department of Gastroenterology, Renmin Hospital of Wuhan University, China.
2.2. Lesion and Operation Characteristics
Tumor size (13.9 ± 2.7 mm, median 24 mm, and range from 6 to 105 mm) was included. Pathologies of the neoplasm were summarized as polyp (hyperplastic, 29, 7.3%; inflammatory, 52, 13.2%), adenoma, carcinoid, and carcinoma. Moreover, macroscopic morphology of the lesions (LST types: LST-G, 27, 6.8%; LST-NG, 37, 9.4%) was presented as well (type 0-IIc 31, 7.8%; 0-IIa+IIc, 26, 6.6%; type 0-I, 7, 1.8%). The operators consisted of junior operators and senior professors. On one hand, there were three well-trained junior doctors (trained for more than three years and operated upon more than one hundred pig stomachs and colons) who participated in the beginning steps for 72 cases that were supervised by senior professors. On the other hand, four senior professors who practiced colonic ESD for more than 500 cases and more than ten years were enrolled. The indications strictly followed the colonic ESD guidelines including polys/LST bigger than 2 cm, a submucosal tumor, carcinoid or rectal carcinoid smaller than 2 cm, and early carcinoma. The operations (techniques and procedures) were conducted according to colonic ESD guidelines, such as lesion observation, marking, submucosal injection, incision, dissection, and wound management. Complications (intraprocedural and postoperative) were involved as well.
2.3. Cost Analysis
The analyzed colonic ESD patient medical costs spanned hospital admission to discharge. Medical costs were categorized as direct, indirect, and intangible. Direct costs were related to treatment and patient care including admission, medical procedures, consumable items, medications, and nursing. Indirect costs were those ascribed to disease-related morbidity and mortality. Intangible costs encompassed nonfinancial outcomes such as pain and mental suffering. Direct medical charges were obtained from hospitalized patient charge lists (CHIS 7.0, Founder International Co., Ltd., China).
2.4. Statistical Analysis
Statistical analysis was performed using SPSS version 17.0 (SPSS, Chicago, IL, USA). Numerical data are presented as mean ± SD. Student's t-test was used for comparison between groups. P<0.05 was considered statistically significant.
3. Results
Table 1 presents clinical features and proximal outcomes for 395 patients (214 male, 185 female) between the ages of 19 and 83 years undergoing colonic ESD. The procedure duration ranged from 11 to 172 min. Tumors varied in diameter from 6 to 105 mm and were variably located from the rectum to the ascending colon. Tumors comprised polyps (20.5%), adenomas (45.3%), carcinoids (22.0%), and carcinomas (12.2%). Nine complications arose during ESD and 56 were recorded after the procedure. Postprocedure and total duration of hospital stay were also recorded.
Table 1.
Clinical features and short-term outcomes for 395 patients treated with colonic ESD.
| Age (yr) | 51.9 ± 6.7 (19–83) |
| Gender | |
| Male (214) | |
| Female (181) | |
| Procedure time (min) | 47.7 ± 5.0 (11–172) |
| Tumor size (mm) | 13.9 ± 2.7 (6–105) |
| Tumor location | |
| Distance from anus (cm) | 36.2 ± 2.9 (5–72) |
| Pathology | |
| Polyp | 81 (20.5%) |
| Hyperplastic | 29 (7.3%) |
| Inflammatory | 52 (13.2%) |
| Adenoma | 185 (46.9%) |
| Carcinoid | 87 (22.0%) |
| Carcinoma | 42 (10.6%) |
| Complications | |
| Intraprocedure | 9 (2.3%) |
| Hemorrhage | 5 (1.3%) |
| Perforation | 3 (0.8%) |
| Infection | 1 (0.2%) |
| Abdominal pain | 0 (0.0%) |
| Postprocedure | 56 (14.2%) |
| Hemorrhage | 22 (5.6%) |
| Perforation | 11 (2.8%) |
| Infection | 11 (2.8%) |
| Abdominal pain | 12 (3.0%) |
| Total | 65 (16.5%) |
| Hospitalization (d) | |
| Postprocedure | 6.6 ± 1.0 (2–21) |
| Total | 8.4 ± 1.9 (4–27) |
We divided costs into several categories: hospitalization, laboratory, imaging, nursing, medication, anesthesia, consumable items, and operation. The hospital financial system precluded more specific cost stratification in this study. Total costs were divided into those covered by National Health Insurance (NHI) and those paid by the patients themselves. Operation, medication, and consumable items' costs accounted for 71% of the total costs for colonic ESD (Table 2 and Figure 1).
Table 2.
Medical costs by category.
| Category | USD |
|---|---|
| Hospitalization | 178 (117–242) |
| Laboratory | 452 (395–674) |
| Radiology | 474 (422–790) |
| Nursing | 191 (163–227) |
| Medication | 2151 (1135–3465) |
| Anesthesia | 385 (352–411) |
| Consumable items | 1563 (1043–2667) |
| Operation | 433 (379–796) |
| Covered by NHI | 2757 (2039–3370) |
| Paid by patient | 3070 (2882–3496) |
| Total | 5927 (4615–9984) |
Figure 1.
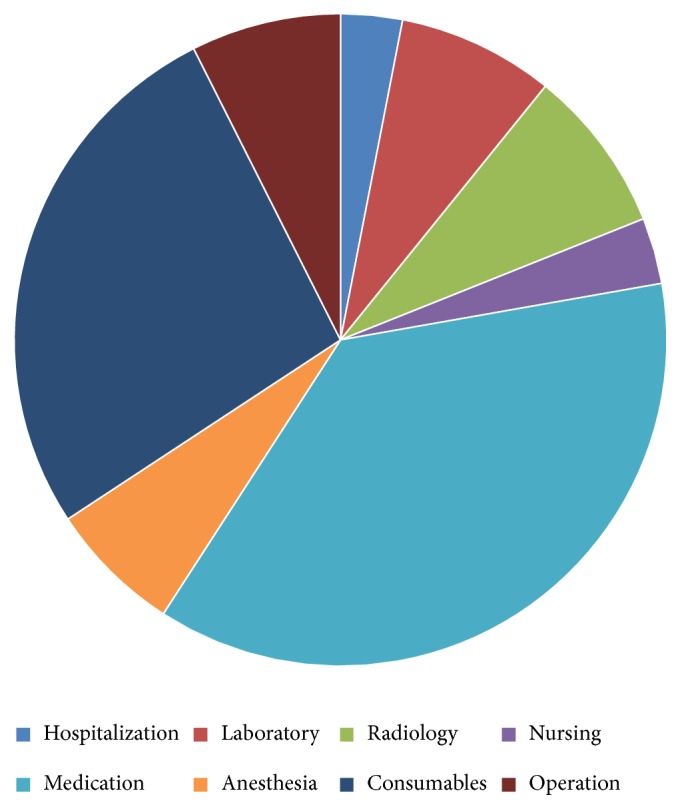
Distribution of medical costs during hospitalization.
We examined relationships between cost and lesion location. Medication and consumable items' costs were higher for lesions in the transverse colon and right hemicolon (>2300 USD) than for lesions in the left hemicolon (<1900 USD). Total costs showed the same trend, with costs exceeding 6700 USD for lesions in the transverse colon and right hemicolon but less than 5800 USD for lesions in the left hemicolon. No major differences in operation costs were observed for lesions in different locations (Table 3 and Figure 2).
Table 3.
Major costs based on lesion location.
| Classification | Rectum | Sigmoid colon | Descending colon | Transverse colon | Ascending colon | Cecum |
|---|---|---|---|---|---|---|
| n = 86 | n = 67 | n = 78 | n = 79 | n = 69 | n = 16 | |
| Medication | 1843 | 1835 | 1866 | 2397 | 2474 | 2341 |
| Consumable | 1546 | 1412 | 1574 | 1801 | 1851 | 1790 |
| Operation | 425 | 381 | 439 | 443 | 399 | 406 |
| Total | 4917 | 4898 | 5755 | 6752 | 7148 | 6933 |
Figure 2.

Distribution of major costs based on lesion location.
To determine the reasons for differential costs associated with different lesion locations, we examined the relationship between location and postprocedure complications. No significant differences were found for hemorrhage, perforation, or infection between different locations (P>0.05). However, abdominal pain occurred significantly more frequently following procedures for lesions of the ascending and transverse colon than for other locations (P<0.05) (Table 4 and Figure 3).
Table 4.
Complications based on lesion location.
| Complication | Location | |||||
|---|---|---|---|---|---|---|
| Rectum | Sigmoid colon | Descending colon | Transverse colon | Ascending colon | Cecum | |
| n = 86 | n = 67 | n = 78 | n = 79 | n = 69 | n = 16 | |
| Hemorrhage (n = 27) | 7 | 5 | 4 | 5 | 6 | 0 |
| Perforation (n = 14) | 2 | 4 | 2 | 2 | 3 | 1 |
| Infection (n = 12) | 3 | 4 | 1 | 2 | 2 | 0 |
| Abdominal pain (n = 12) | 1 | 1 | 0 | 4 | 5 | 1 |
Figure 3.

Complications based on lesion location.
We observed that costs for medication and consumable items and total costs were significantly higher for lesions in the size ranges of 1–5 cm and >5 cm than for lesions <1 cm (P<0.05). Operation costs, however, did not significantly differ according to lesion size (P> 0.05). The presence of multiple lesions (>1) was significantly correlated with higher costs for medication, consumable items, and operation and total costs compared to patients with a single lesion (P< 0.05). Carcinoma was associated with significantly (P< 0.05) higher costs for medication, consumable items, and operation and total costs compared to other pathologies (Table 5 and Figure 4).
Table 5.
Main costs based on lesion size, number, and pathology.
| Category | Size (cm) | Number | Pathology | ||||||
|---|---|---|---|---|---|---|---|---|---|
| < 1, | 1–5, | > 5, | 1, n=298 | >1, n=97 | Polyp | Adenoma | Carcinoid | Carcinoma | |
| n=77 | n=282 | n=36 | n=81 | n=185 | n=87 | n=42 | |||
| Medication | 1705 | 2249 | 2583 | 2030 | 2528 | 1909 | 2197 | 2043 | 3259 |
| Consumable items | 1446 | 1691 | 1802 | 1419 | 1832 | 1326 | 1452 | 1465 | 2508 |
| Operation | 426 | 430 | 438 | 421 | 462 | 397 | 432 | 450 | 741 |
| Total | 5249 | 5921 | 6897 | 5773 | 6758 | 5674 | 5990 | 6252 | 9147 |
Figure 4.

Distribution of main costs based on lesion size, number, and pathology.
Stratifying patients based on operation characteristics, we observed that positive surgical margins were associated with significantly higher medication, consumable items, and total costs compared to negative surgical margins (P< 0.05). With respect to complications, hemorrhage and perforation were associated with significantly higher medication, consumable items, and total costs compared to complications of infection and abdominal pain (Table 6 and Figure 5).
Table 6.
Main costs based on surgical margins and complications.
| Category | Surgical margin | Complication | ||||
|---|---|---|---|---|---|---|
| Negative | Positive | Hemorrhage | Perforation | Infection | Abdominal pain | |
| n=331 | n=64 | n=27 | n=14 | n=12 | n=12 | |
| Medication | 2012 | 2988 | 2797 | 3086 | 2490 | 2385 |
| Consumable items | 1475 | 2460 | 1994 | 2418 | 1562 | 1649 |
| Operation | 404 | 718 | 502 | 717 | 425 | 432 |
| Total | 5626 | 8759 | 6135 | 8704 | 6056 | 5992 |
Figure 5.

Distribution of main costs based on surgical margins and complications.
To better understand differential costs associated with duration of hospitalization, we analyzed the occurrence of post-ESD complications. Total post-ESD complications comprised hemorrhage (6.84%, 27/395), perforation (3.54%, 14/395), infection (3.04%, 12/395), and abdominal pain (3.04%, 12/395). The incidence of complications was significantly higher within 4 d after the procedure (87.7%) than at 4 d (12.3%) (Table 7). We examined costs relative to length of hospitalization (≤4 d, 4–10 d, and >10 d). Hospitalization of ≤4 d and >10 d was associated with the lowest and highest, respectively, medication, consumable items, and total costs (P< 0.05). Hospitalization of >10 d was correlated with significantly higher operation costs (Table 8 and Figure 6).
Table 7.
Complications based on hospitalization days after the procedure.
| Complication | Days | |||
|---|---|---|---|---|
| ≤1 | ≤4 | ≤10 | >10 | |
| Hemorrhage (n) | 5 | 18 | 1 | 3 |
| Perforation (n) | 5 | 8 | 1 | 0 |
| Infection (n) | 2 | 8 | 1 | 1 |
| Abdominal pain (n) | 7 | 4 | 1 | 0 |
Table 8.
Costs based on hospitalization days after the procedure.
| Category | Hospitalization days (post-ESD) | ||
|---|---|---|---|
| ≤4, n=84 | 4<n≤10, n=244 | >10, n=67 | |
| Medication | 1835 | 2479 | 3142 |
| Consumable items | 1390 | 1703 | 2004 |
| Operation | 408 | 426 | 579 |
| Total | 4811 | 5848 | 6925 |
Figure 6.

Distribution of costs based on hospitalization days after the procedure.
Lastly, in order to clarify the relationship between costs and outcomes, the costs have been analyzed for defined outcomes of colorectal ESD. More detailedly, the costs of uncomplicated curative ESD of cancer (and HGIN), uncomplicated noncurative ESD of cancer, and complicated ESD of cancer (and of other benign lesions) were analyzed, respectively (Table 9).
Table 9.
Costs based on defined outcomes.
| Classification | Uncomplicated curative ESD of cancer | Uncomplicated curative ESD of HGIN | Uncomplicated noncurative ESD of cancer | Complicated ESD of cancer | Complicated ESD of other benign lesions |
|---|---|---|---|---|---|
| Medication | 2557 | 2213 | 2748 | 3204 | 2717 |
| Consumable items | 1640 | 1502 | 1484 | 2276 | 1895 |
| Operation | 448 | 433 | 405 | 694 | 499 |
| Total | 6348 | 6185 | 8251 | 8739 | 7283 |
4. Discussion
Minimally invasive ESD is a promising technique for treatment of digestive tract diseases, especially polyps, adenoma, and early stage carcinoma [7]. Most well-trained gastrointestinal endoscopic physicians can correctly perform ESD operations [8]. Colonic ESD will certainly become more prevalent due to higher patient satisfaction, more comprehensive indications, shorter hospitalization times, and lower costs compared to traditional laparotomy surgery [9]. In China, NHI covers some costs associated with colonic ESD, except for some consumable items and medication [10]. The present study helps to clarify some cost issues related to colonic ESD treatment in China. More specifically, we examined costs of clinical characteristics including lesion location, size, number, and pathology; surgical margin and complications; and hospital stay after the procedure. Costs for medication and consumable items together accounted for >60% of total costs. Operation and nursing costs combined comprised only 10% of total costs, indicating that physician and nurse labor costs in China remain low.
Nearly 90% of complications occurred within 4 d after the procedure. With respect to lesion location, abdominal pain occurred more often when lesions were located in the ascending and transverse colon. This might be due to poor gas release from high positions of the colon, with the pneumogastric nerve only exiting above the splenic flexure [11, 12]. When operating on high colonic positions surgeons generally take more time and use more consumable items (such as metal clips) to avoid complications such as hemorrhage or perforation [13]. In contrast, studies showed that, even in the right colon, the complication rate should not be increased for ESD on the professional level [14, 15]. This in turn requires patients to stay longer in hospital for postoperative observation [16]. This could contribute to higher costs for medication and consumable items and total costs for high-position colon ESD. ESD to treat larger lesions also requires more consumable items, as well as more medication to promote wound healing and patient recovery [17, 18]. We made similar observations with respect to lesion number. Some carcinoma patients received surgical and additional oncology treatment, and thus costs for these patients were relatively high [19]. Positive surgical margins were associated with higher medical, consumable items, operation, and total costs [20, 21]. Patients with post-ESD complications of hemorrhage and perforation had higher costs associated with repeat endoscopy and/or surgical treatment [22, 23]. Based on defined outcomes, complicated colonic ESD of cancer occupied the highest position for costs of all aspects on account of more usage of medication and consumables, more complex operations and multiple therapies intervention, and so on [24]. However, the uncomplicated operations had lower medication, consumable items, operation, and total costs comparing to the complicated ones. The reason might be effective coaching and cost controlling in the process. Even when well-trained junior physicians participated in some parts of an ESD operation, senior professors supervised and step-by-step guided the whole procedure [14, 25]. Further efforts (randomized control trial) could be focused on costs saving of different treatment choices, such as cost savings of professional colorectal ESD as compared to elective laparoscopic operation or surgery for a specific indication [26]. Our study indicated only 10.6% malignant lesions were resected by colorectal ESD. Therefore, more profound thinking should be concentrated on the endoscopic diagnosis accuracy and indication criteria selection. Cheaper techniques (e.g., EMR) should be performed for benign neoplasia [27]. For instance, small and rectal carcinoid could be easily resected with much cheaper procedures, such as rubber band EMR or UEMR [28]. Our analysis suggests benchmark costs for colonic ESD of approximately 400 USD for the procedure itself and a total treatment cost of 5000 USD. The current study retrospectively examined costs for 395 patients treated at a single center. Additional prospective multicenter studies will be needed to fully evaluate issues of cost-effectiveness.
Data Availability
The data and material are available from the corresponding author under reasonable request.
Conflicts of Interest
The authors declare that they have no conflicts of interest with any other third party.
Authors' Contributions
Ning Cui finished the work of data collection and analysis. Ning Cui and Yu Zhao finished paper writing. Honggang Yu fulfilled the conception of this study.
References
- 1.Brody H. Colorectal cancer. Nature. 2015;521(7551):S1–S1. doi: 10.1038/521S1a. [DOI] [PubMed] [Google Scholar]
- 2.Shankaran V. Cost considerations in the evaluation and treatment of colorectal cancer. Current Treatment Options in Oncology. 2015;16(8, article 41) doi: 10.1007/s11864-015-0354-4. [DOI] [PubMed] [Google Scholar]
- 3.Fujihara S., Mori H., Kobara H., et al. Current innovations in endoscopic therapy for the management of colorectal cancer: from endoscopic submucosal dissection to endoscopic full-thickness resection. BioMed Research International. 2014;2014:12. doi: 10.1155/2014/925058.925058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tashima T., Ohata K., Sakai E., et al. Efficacy of an over-the-scope clip for preventing adverse events after duodenal endoscopic submucosal dissection: a prospective interventional study. Endoscopy. 2018;50(05):487–496. doi: 10.1055/s-0044-102255. [DOI] [PubMed] [Google Scholar]
- 5.Wagner A., Neureiter D., Kiesslich T., et al. Single-center implementation of endoscopic submucosal dissection (ESD) in the colorectum: low recurrence rate after intention-to-treat ESD. Digestive Endoscopy. 2018;30(3):354–363. doi: 10.1111/den.12995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang J., Zhang X.-H., Ge J., Yang C.-M., Liu J.-Y., Zhao S.-L. Endoscopic submucosal dissection vs endoscopic mucosal resection for colorectal tumors: a meta-analysis. World Journal of Gastroenterology. 2014;20(25):8282–8287. doi: 10.3748/wjg.v20.i25.8282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mizutani H., Ono S., Ohki D., et al. Recent development of techniques and devices in colorectal endoscopic submucosal dissection. Clinical Endoscopy. 2017;50(6):562–568. doi: 10.5946/ce.2017.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shiga H., Kuroha M., Endo K., et al. Colorectal endoscopic submucosal dissection (ESD) performed by experienced endoscopists with limited experience in gastric ESD. International Journal of Colorectal Disease. 2015;30(12):1645–1652. doi: 10.1007/s00384-015-2334-3. [DOI] [PubMed] [Google Scholar]
- 9.Nakamura F., Saito Y., Sakamoto T., et al. Potential perioperative advantage of colorectal endoscopic submucosal dissection versus laparoscopy-assisted colectomy. Surgical Endoscopy. 2015;29(3):596–606. doi: 10.1007/s00464-014-3705-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang Y., Tang W., Zhang X., Zhang Y., Zhang L., Wang Y. National health insurance development in China from 2004 to 2011: coverage versus benefits. PLoS ONE. 2015;10(5) doi: 10.1371/journal.pone.0124995.e0124995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mizushima T., Kato M., Iwanaga I., et al. Technical difficulty according to location, and risk factors for perforation, in endoscopic submucosal dissection of colorectal tumors. Surgical Endoscopy. 2015;29(1):133–139. doi: 10.1007/s00464-014-3665-9. [DOI] [PubMed] [Google Scholar]
- 12.Jabari S., da Silveira A. B., de Oliveira E. C., et al. Mucosal layers and related nerve fibres in non-chagasic and chagasic human colon—a quantitative immunohistochemical study. Cell and Tissue Research. 2014;358(1):75–83. doi: 10.1007/s00441-014-1934-5. [DOI] [PubMed] [Google Scholar]
- 13.Ko Y. B., Lee J., Kim W. S., et al. Unexpected delayed colon perforation after the endoscopic submucosal dissection with snaring of a laterally spreading tumor. Clinical Endoscopy. 2015;48(6):570–575. doi: 10.5946/ce.2015.48.6.570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gromski M. A., Cohen J., Saito K., et al. Learning colorectal endoscopic submucosal dissection: a prospective learning curve study using a novel ex vivo simulator. Surgical Endoscopy. 2017;31(10):4231–4237. doi: 10.1007/s00464-017-5484-2. [DOI] [PubMed] [Google Scholar]
- 15.Ebigbo A., Probst A., Römmele C., Messmann H. Step-up training for colorectal and gastric ESD and the challenge of ESD training in the proximal colon: results from a German Center. Endoscopy International Open. 2018;06(05):E524–E530. doi: 10.1055/a-0584-6457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tomiki Y., Kawai M., Takehara K., et al. Clinical pathway to discharge 3 days after colorectal endoscopic submucosal dissection. Digestive Endoscopy. 2015;27(6):679–686. doi: 10.1111/den.12468. [DOI] [PubMed] [Google Scholar]
- 17.Tsuji Y., Ohata K., Gunji T., et al. Endoscopic tissue shielding method with polyglycolic acid sheets and fibrin glue to cover wounds after colorectal endoscopic submucosal dissection (with video) Gastrointestinal Endoscopy. 2014;79(1):151–155. doi: 10.1016/j.gie.2013.08.041. [DOI] [PubMed] [Google Scholar]
- 18.Holmes I., Friedland S. Endoscopic mucosal resection versus endoscopic submucosal dissection for large polyps: a western colonoscopist’s view. Clinical Endoscopy. 2016;49(5):454–456. doi: 10.5946/ce.2016.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tanaka S., Kashida H., Saito Y., et al. JGES guidelines for colorectal endoscopic submucosal dissection/endoscopic mucosal resection. Digestive Endoscopy. 2015;27(4):417–434. doi: 10.1111/den.12456. [DOI] [PubMed] [Google Scholar]
- 20.Noh G. Y., Ku H. R., Kim Y. J., et al. Clinical outcomes of early gastric cancer with lymphovascular invasion or positive vertical resection margin after endoscopic submucosal dissection. Surgical Endoscopy. 2015;29(9):2583–2589. doi: 10.1007/s00464-014-3973-0. [DOI] [PubMed] [Google Scholar]
- 21.Ozawa S., Tanaka S., Hayashi N., et al. Risk factors for vertical incomplete resection in endoscopic submucosal dissection as total excisional biopsy for submucosal invasive colorectal carcinoma. International Journal of Colorectal Disease. 2013;28(9):1247–1256. doi: 10.1007/s00384-013-1701-1. [DOI] [PubMed] [Google Scholar]
- 22.Ogasawara N., Yoshimine T., Noda H., et al. Clinical risk factors for delayed bleeding after endoscopic submucosal dissection for colorectal tumors in Japanese patients. European Journal of Gastroenterology & Hepatology. 2016;28(12):1407–1414. doi: 10.1097/MEG.0000000000000723. [DOI] [PubMed] [Google Scholar]
- 23.Yoon J. Y., Kim J. H., Lee J. Y., et al. Clinical outcomes for patients with perforations during endoscopic submucosal dissection of laterally spreading tumors of the colorectum. Surgical Endoscopy. 2013;27(2):487–493. doi: 10.1007/s00464-012-2462-6. [DOI] [PubMed] [Google Scholar]
- 24.Gallegos-Orozco J. F., Gurudu S. R. Complex colon polypectomy. Journal of Gastroenterology and Hepatology. 2010;6(6):375–382. [PMC free article] [PubMed] [Google Scholar]
- 25.Yamamoto S., Uedo N., Ishihara R., et al. Endoscopic submucosal dissection for early gastric cancer performed by supervised residents: assessment of feasibility and learning curve. Endoscopy. 2009;41(11):923–928. doi: 10.1055/s-0029-1215129. [DOI] [PubMed] [Google Scholar]
- 26.Hon S. S. F., Ng S. S. M., Wong T. C. L., et al. Endoscopic submucosal dissection vs laparoscopic colorectal resection for early colorectal epithelial neoplasia. World Journal of Gastrointestinal Endoscopy. 2015;7(17):1243–1249. doi: 10.4253/wjge.v7.i17.1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Arezzo A., Passera R., Marchese N., Galloro G., Manta R., Cirocchi R. Systematic review and meta-analysis of endoscopic submucosal dissection vs endoscopic mucosal resection for colorectal lesions. United European Gastroenterology Journal. 2016;4(1):18–29. doi: 10.1177/2050640615585470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yamashina T., Tumura T., Maruo T., et al. Underwater endoscopic mucosal resection: a new endoscopic method for resection of rectal neuroendocrine tumor grade 1 (carcinoid) ≤ 10 mm in diameter. Endoscopy International Open. 2018;06(01):E111–E114. doi: 10.1055/s-0043-123467. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data and material are available from the corresponding author under reasonable request.


