Abstract
Background
Pancreatic pseudocysts are walled‐off peripancreatic fluid collections. There is considerable uncertainty about how pancreatic pseudocysts should be treated.
Objectives
To assess the benefits and harms of different management strategies for pancreatic pseudocysts.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) in The Cochrane Library 2015, Issue 9, and MEDLINE, EMBASE, Science Citation Index Expanded, and trials registers until September 2015. We also searched the references of included trials and contacted trial authors.
Selection criteria
We only considered randomised controlled trials (RCTs) of people with pancreatic pseudocysts, regardless of size, presence of symptoms, or aetiology. We placed no restrictions on blinding, language, or publication status of the trials.
Data collection and analysis
Two review authors independently identified trials and extracted data. We calculated the odds ratio (OR) and mean difference (MD) with 95% confidence intervals (CI) with RevMan 5, based on an available‐case analysis for direct comparisons, using fixed‐effect and random‐effect models. We also conducted indirect comparisons (rather than network meta‐analysis), since there were no outcomes for which direct and indirect evidence were available.
Main results
We included four RCTs, with 177 participants, in this review. After one participant was excluded, 176 participants were randomised to endoscopic ultrasound (EUS)‐guided drainage (88 participants), endoscopic drainage (44 participants), EUS‐guided drainage with nasocystic drainage (24 participants), and open surgical drainage (20 participants). The comparisons included endoscopic drainage versus EUS‐guided drainage (two trials), EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage alone (one trial), and open surgical drainage versus EUS‐guided drainage (one trial). The participants were mostly symptomatic, with pancreatic pseudocysts resulting from acute and chronic pancreatitis of varied aetiology. The mean size of the pseudocysts ranged between 70 mm and 155 mm across studies. Although the trials appeared to include similar types of participants for all comparisons, we were unable to assess this statistically, since there were no direct and indirect results for any of the comparisons.
All the trials were at unclear or high risk of bias, and the overall quality of evidence was low or very low for all outcomes. One death occurred in the endoscopic drainage group (1/44; 2.3%), due to bleeding. There were no deaths in the other groups. The differences in the serious adverse events were imprecise. Short‐term health‐related quality of life (HRQoL; four weeks to three months) was worse (MD ‐21.00; 95% CI ‐33.21 to ‐8.79; participants = 40; studies = 1; range: 0 to 100; higher score indicates better) and the costs were higher in the open surgical drainage group than the EUS‐guided drainage group (MD 8040 USD; 95% CI 3020 to 13,060; participants = 40; studies = 1). There were fewer adverse events in the EUS‐guided drainage with nasocystic drainage group than in the EUS‐guided drainage alone (OR 0.20; 95% CI 0.06 to 0.73; participants = 47; studies = 1), or the endoscopic drainage group (indirect comparison: OR 0.08; 95% CI 0.01 to 0.61). Participants with EUS‐guided drainage with nasocystic drainage also had shorter hospital stays compared to EUS‐guided drainage alone (MD ‐8.10 days; 95% CI ‐9.79 to ‐6.41; participants = 47; studies = 1), endoscopic drainage (indirect comparison: MD ‐7.10 days; 95% CI ‐9.38 to ‐4.82), or open surgical drainage group (indirect comparison: MD ‐12.30 days; 95% CI ‐14.48 to ‐10.12). The open surgical drainage group had longer hospital stays than the EUS‐guided drainage group (MD 4.20 days; 95% CI 2.82 to 5.58; participants = 40; studies = 1); the endoscopic drainage group had longer hospital stays than the open drainage group (indirect comparison: ‐5.20 days; 95% CI ‐7.26 to ‐3.14). The need for additional invasive interventions was higher for the endoscopic drainage group than the EUS‐guided drainage group (OR 11.13; 95% CI 2.85 to 43.44; participants = 89; studies = 2), and the open drainage group (indirect comparison: OR 23.69; 95% CI 1.40 to 400.71). The differences between groups were imprecise for the other comparisons that could be performed. None of the trials reported long‐term mortality, medium‐term HRQoL (three months to one year), long‐term HRQoL (longer than one year), time‐to‐return to normal activities, or time‐to‐return to work.
Authors' conclusions
Very low‐quality evidence suggested that the differences in mortality and serious adverse events between treatments were imprecise. Low‐quality evidence suggested that short‐term HRQoL (four weeks to three months) was worse, and the costs were higher in the open surgical drainage group than in the EUS‐guided drainage group. Low‐quality or very low‐quality evidence suggested that EUS‐guided drainage with nasocystic drainage led to fewer adverse events than EUS‐guided or endoscopic drainage, and shorter hospital stays when compared to EUS‐guided drainage, endoscopic drainage, or open surgical drainage, while EUS‐guided drainage led to shorter hospital stays than open surgical drainage. Low‐quality evidence suggested that there was a higher need for additional invasive procedures with endoscopic drainage than EUS‐guided drainage, while it was lower in the open surgical drainage than in the endoscopic drainage group.
Further RCTs are needed to compare EUS‐guided drainage, with or without nasocystic drainage, in symptomatic patients with pancreatic pseudocysts that require treatment. Future trials should include patient‐oriented outcomes such as mortality, serious adverse events, HRQoL, hospital stay, return‐to‐normal activity, number of work days lost, and the need for additional procedures, for a minimum follow‐up period of two to three years.
Plain language summary
Treatment methods for people with pancreatic pseudocysts (fluid collections around the pancreas)
Review question
How should people with pancreatic pseudocysts be treated?
Background
The pancreas is an abdominal organ that secretes several digestive enzymes (substances that enable and speed up chemical reactions in the body) into the pancreatic ductal system, which empties into the small bowel. It also contains the Islets of Langerhans, which secrete several hormones, including insulin (that helps to regulate blood sugar). Pancreatic pseudocysts are fluid collections around the pancreas. They arise due to sudden or long‐standing inflammation of the pancreas. While some will disappear when the inflammation of the pancreas settles down, others remain and cause symptoms such as abdominal pain, indigestion, vomiting, and weight loss. Treatments of pancreatic pseudocysts include conservative treatment (watchful monitoring), surgical drainage, which can be performed through a standard cut (open surgical drainage) or by key‐hole surgery (laparoscopic surgical drainage), or endoscopic drainage. In endoscopic drainage, a tube (stent) is inserted with the help of an endoscope (a tube passed through the mouth into the stomach, usually to visualise the abdominal organs from inside the body), that connects the pseudocyst to the stomach or the upper part of the small intestine. The insertion may be further helped by using an endoscopic ultrasound (an ultrasound probe attached to the endoscope; EUS‐guided drainage). Endoscopic ultrasound‐guided drainage may be further assisted by passing a tube through the nose and inserting it into the cyst during EUS‐guided drainage (EUS‐guided drainage with nasocystic drainage). The best way to treat pancreatic pseudocysts is not clear. We sought to resolve this by searching for existing studies on the topic. We included all randomised controlled trials whose results were reported up to 8 September 2015. Apart from using standard Cochrane methods, which allow comparison of only two treatments at a time (direct comparison), we used advanced methods, which allow individual comparison of the different treatments compared in the trials (indirect comparison).
Study characteristics
We included four trials, with 177 participants, in the review, 176 of whom were included in the analyses. The treatments compared in the four trials included endoscopic drainage (without EUS guidance), EUS‐guided drainage, EUS‐guided drainage with nasocystic drainage, and open surgical drainage. The participants were mostly people with pancreatic pseudocysts resulting from sudden onset or long‐term inflammation of the pancreas, from different causes.
Key results
One death occurred in the endoscopic drainage group, due to bleeding. The differences in the serious complications were imprecise. Short‐term health‐related quality of life (HRQoL; four weeks to three months) was worse, and the costs were higher in the open surgical drainage group than in the EUS‐guided drainage group. There were fewer complications of any severity (such as bleeding) that required additional treatment in the EUS‐guided drainage with nasocystic drainage group than in the EUS‐guided drainage alone or endoscopic drainage groups. Those who received EUS‐guided drainage with nasocystic drainage also had a shorter hospital stay compared to those who received EUS‐guided drainage alone, endoscopic drainage, or open surgical drainage. Those who received EUS‐guided drainage alone had shorter hospital stays than those with open surgical drainage. There was a higher need for additional invasive treatments to completely drain the pseudocyst with endoscopic drainage than EUS‐guided drainage alone. The differences for the other comparisons were imprecise. None of the trials reported long‐term deaths, medium‐term or long‐term HRQoL, time‐to‐return to normal activities, or time‐to‐return to work.
Quality of the evidence
The overall quality of evidence was low or very low for all the outcomes, because the trials were small and at high risk of bias (for example, prejudice of people who conduct the trial, and trial participants who prefer one treatment over another). As a result, further studies are required on this topic. Such studies should compare EUS‐guided drainage with or without nasocystic drainage in people who have symptoms from their pancreatic pseudocysts and need treatment. Such trials should measure patient‐oriented outcomes for a minimum follow‐up period of two to three years.
Summary of findings
Summary of findings for the main comparison. Management strategies for pancreatic pseudocysts: primary outcomes.
| Management strategies for pancreatic pseudocysts: a network meta‐analysis | |||||
| Patient or population: patients with pancreatic pseudocysts Settings: secondary or tertiary care Intervention: management strategies for pancreatic pseudocysts | |||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | |
| Assumed risk | Corresponding risk | ||||
| EUS‐guided drainage | Other methods of drainage | ||||
| Short‐term mortality | |||||
| Endoscopic drainage versus EUS‐guided drainage | 1 per 1000 | 3 per 1000 (0 to 74) | OR 3 (0.11 to 79.91) | 89 (2 studies) | ⊕⊝⊝⊝ very low1,2,3 |
| EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | not estimable since there was no short‐term mortality in either group | Not estimable | 47 (1 study) | ⊕⊝⊝⊝ very low1,2,3 | |
| Open surgical drainage versus EUS‐guided drainage | not estimable since there was no short‐term mortality in either group | Not estimable | 40 (1 study) | ⊕⊝⊝⊝ very low1,2,3 | |
| Serious adverse events | |||||
| Endoscopic drainage versus EUS‐guided drainage | 31 per 1000 | 72 per 1000 (16 to 268) | OR 2.42 (0.51 to 11.46) | 89 (2 studies) | ⊕⊝⊝⊝ very low1,2,3 |
| Open surgical drainage versus EUS‐guided drainage | 31 per 1000 | 208 per 1000 (13 to 845) | OR 8.2 (0.4 to 169.9) | 40 (1 study) | ⊕⊝⊝⊝ very low1,2,3 |
| Health‐related quality of life (4 weeks to 3 months) | |||||
| Open surgical drainage versus EUS‐guided drainage
Scale from: 0 to 100. Higher indicates better |
The mean health‐related quality of life in the EUS‐guided drainage was 71.4 | The mean health‐related quality of life in the open surgical drainage was 21 lower (33.21 to 8.79 lower) | 40 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| |||||
| *The basis for the assumed risk was the mean control group proportion for all outcomes other than short‐term mortality. For short‐term mortality, a short‐term mortality of 0.1% was used since there was no mortality in the control group. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; OR: Odds ratio. | |||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | |||||
1 Risk of bias was unclear or high in the trial(s). 2 Sample size was small. 3 Confidence intervals overlapped clinically important differences.
Summary of findings 2. Management strategies for pancreatic pseudocysts: a network meta‐analysis.
| Management strategies for pancreatic pseudocysts: a network meta‐analysis | |||||
| Patient or population: patients with pancreatic pseudocysts Settings: secondary or tertiary care Intervention: management strategies for pancreatic pseudocysts | |||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | |
| Assumed risk | Corresponding risk | ||||
| EUS‐guided drainage | Other methods of drainage | ||||
| Adverse events | |||||
| Endoscopic drainage versus EUS‐guided drainage | 171 per 1000 | 333 per 1000 (95 to 703) | OR 2.42 (0.51 to 11.46) | 89 (2 studies) | ⊕⊝⊝⊝ very low1,2,3 |
| EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | 171 per 1000 | 40 per 1000 (12 to 131) | OR 0.2 (0.06 to 0.73) | 47 (1 study) | ⊕⊕⊝⊝ low1,2 |
| Open surgical drainage versus EUS‐guided drainage | 171 per 1000 | 698 per 1000 (104 to 979) | OR 11.18 (0.56 to 222.98) | 40 (1 study) | ⊕⊝⊝⊝ very low1,2,3 |
| Hospital stay | |||||
| Endoscopic drainage versus EUS‐guided drainage | The median hospital stay in the EUS‐guided drainage group was 2.65 days | The mean hospital stay in the endoscopic drainage was 1 day less (2.53 less to 0.53 higher) | 29 (1 study) | ⊕⊝⊝⊝ very low1,2,3 | |
| EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | The median hospital stay in the EUS‐guided drainage group was 2.65 days | The mean hospital stay in the EUS‐guided drainage with nasocystic drainage was 8.1 days less (9.79 to 6.41 less) | 47 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| Open surgical drainage versus EUS‐guided drainage | The median hospital stay in the EUS‐guided drainage group was 2.65 days | The mean hospital stay in the open surgical drainage was 4.2 days more (2.82 to 5.58 more) | 40 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| Need for additional drainage | |||||
| Endoscopic drainage versus EUS‐guided drainage | 62 per 1000 | 424 per 1000 (159 to 742) | OR 11.13 (2.85 to 43.44) | 89 (2 studies) | ⊕⊝⊝⊝ very low1,2,4 |
| Open surgical drainage versus EUS‐guided drainage | 62 per 1000 | 30 per 1000 (3 to 273) | OR 0.47 (0.04 to 5.69) | 40 (1 study) | ⊕⊝⊝⊝ very low1,2,3 |
| Costs | |||||
| Open surgical drainage versus EUS‐guided drainage | The mean costs in the EUS‐guided drainage were 7010 US dollars | The mean costs in the open surgical drainage were 8040 higher (3020 to 13,060 higher) | 40 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| |||||
| *The basis for the assumed risk was the mean control group proportion. The corresponding risk (and its 95% confidence interval) was based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; OR: Odds ratio; | |||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | |||||
1 Risk of bias was unclear or high in the trial(s). 2 Sample size was small. 3 Confidence intervals overlapped clinically important differences. 4 There was moderate inconsistency as evidenced by I‐square, tau‐square, and lack of significant overlap of confidence intervals.
Background
Description of the condition
The pancreas is an abdominal organ that secretes several digestive enzymes into the pancreatic ductal system that empties into the small bowel. It also lodges the Islets of Langerhans, which secrete several hormones, including insulin (NCBI 2014). Pancreatic pseudocysts are fluid collections in the peripancreatic tissues, although they may be partly or wholly intra‐pancreatic (Banks 2013). They are surrounded by a well‐defined wall and contain only fluid, with little or no solid material (Banks 2013). They can arise after an episode of acute pancreatitis, sudden worsening of chronic pancreatitis, or pancreatic trauma (Cannon 2009). However, the development of a pancreatic pseudocyst is rare in acute pancreatitis (Banks 2013). Most fluid collections previously called pseudocysts after an episode of acute pancreatitis are now called acute peripancreatic fluid collections (which do not have well‐defined walls), acute necrotic collections (which do not contain a wall and contain solid material because of necrosis), or walled‐off necrosis (which have solid material because of necrosis; Banks 2013). Pancreatic pseudocysts are believed to arise from a disruption of the main pancreatic duct or its branches without any recognisable pancreatic parenchymal (pancreatic cellular tissue) necrosis (indicated by solid necrotic material), resulting in leakage of pancreatic juice into the retroperitoneum or the peripancreatic tissues, which leads to a persistent, localised fluid collection (Banks 2013; Cannon 2009). Occasionally, a pseudocyst may also arise in people with acute necrotising pancreatitis (inflammation of the pancreas with presence of necrosis in the pancreas or peripancreatic tissues) when the pancreatic parenchymal necrosis of the neck or body of the pancreas isolates a still‐functioning distal (left‐sided) pancreatic remnant (‘disconnected duct syndrome’; Banks 2013). Rarely, a pseudocyst may be evident many weeks after surgical debridement (removal of necrotic tissue) for acute pancreatic necrosis (necrosectomy), due to localised leakage of the disconnected duct into the necrosectomy cavity. Since the necrosis has been removed, the walled‐off cavity contains only fluid and hence, is called a pseudocyst (Banks 2013). The pancreatic pseudocystic fluid is usually rich in pancreatic amylase (Banks 2013; Cannon 2009). Diagnoses of pancreatic pseudocysts are made on the basis of the presence of cystic collection in the peripancreatic tissues with a well‐defined wall and with little or no solid material on an imaging modality, such as contrast‐enhanced computed tomography (CECT), magnetic resonance imaging (MRI), or an ultrasound (Banks 2013).
The true incidence of pancreatic pseudocysts is difficult to determine, because many of the previous studies calculating the incidence in acute pancreatitis might have included acute peripancreatic fluid collections, acute necrotic collections, or walled‐off necrosis. Symptoms related to pancreatic pseudocysts include persistent abdominal pain, gastric (stomach) outlet obstruction, jaundice (yellowish discolouration of the skin and dark urine), dyspepsia (indigestion), weight loss, and persistent fevers. They are believed to arise from the local mass effect (pressure on the surrounding structures) of the pseudocyst or the associated inflammatory response (Cannon 2009; Cheruvu 2003). Potential complications related to pancreatic pseudocysts include infection of the pseudocyst, bile duct or gastric outflow obstruction, free rupture of the pseudocyst into the peritoneal cavity, portal or splenic vein thrombosis leading to sinistral (left‐sided) portal hypertension, pseudocyst erosion into adjacent (nearby) vessels resulting in pseudoaneurysm formation, or even catastrophic haemorrhage into the gastrointestinal tract or peritoneal cavity (Cannon 2009; Vitas 1992). However, the frequency of these complications is not known, since the natural history of pancreatic pseudocysts is not well understood (Cannon 2009).
See Appendix 1 for a glossary of terms.
Description of the intervention
The main purpose of treatment is for the relief of symptoms related to pancreatic pseudocysts and to avoid the complications related to pseudocysts. The various treatment options include: conservative management; surgical management (cystogastrostomy or cystoenterostomy); radiological management (percutaneous drainage); endoscopic management (transpapillary), when the pancreatic pseudocyst communicates with the main pancreatic duct, transenteric approach, when an endoluminal bulge (within the cavity of the stomach or small intestine), or endoscopic ultrasound evidence of adherence between the gastric or duodenal wall and the cyst without associated necrosis, which can be achieved with or without an endoscopic ultrasound and with or without nasocystic drainage; Cannon 2009; Cheruvu 2003; Johnson 2009; Varadarajulu 2008; Varadarajulu 2013; Yuan 2015). Some of the treatment‐related complications include bleeding that requires additional treatment, and pancreatic stricture (Varadarajulu 2008; Varadarajulu 2013).
How the intervention might work
The interventions may work by decompressing the fluid, relieving the pressure of the pancreatic pseudocyst on the surrounding structures, relieving the inflammatory response that causes symptoms, and allowing an alternative route for the pancreatic juice to reach the small bowel, thereby allowing digestion and relieving pain that arises because of indigestion.
Why it is important to do this review
Currently, treatment of pseudocysts is only indicated in symptomatic patients (Cannon 2009). The traditional management of symptomatic pancreatic pseudocysts is open surgical drainage. The roles of laparoscopic drainage, endoscopic drainage using an oesophagoduodenoscope or an endoscopic ultrasound, percutaneous management, and conservative management are not clear. Multiple treatment comparison or network meta‐analysis allows the comparison of several treatments simultaneously and provides information on the relative effect of one treatment versus another, even when no direct comparison has been made. There is no Cochrane network meta‐analysis on this topic. This systematic review and network meta‐analysis will identify the relative effects of different treatments and identify any research gaps.
Objectives
To assess the benefits and harms of different management strategies for pancreatic pseudocysts.
Methods
Criteria for considering studies for this review
Types of studies
We included randomised controlled trials (RCTs). We included studies reported as full text, those published as abstract only, and unpublished data.
Types of participants
We included adults with pancreatic pseudocysts, regardless of the cause for the pseudocyst (acute pancreatitis, chronic pancreatitis, or trauma), the presence of symptoms, or the size and location of the cyst.
When an incidental pancreatic cyst is noted (i.e., people undergo a scan for other reasons and a pancreatic cyst is noted), there may be difficulty in distinguishing a pancreatic pseudocyst, arising in the background of chronic pancreatitis, from pancreatic cystic neoplasms (premalignant or malignant) that may cause local inflammation (Cannon 2009). Sometimes, it may not be possible to establish a clear diagnosis based on additional imaging, cytology of the fluid, or tumour marker levels in the fluid (Cannon 2009). In such situations, resection of the cyst and histopathological examination (examination under the microscope) may be performed purely to ensure that premalignant or malignant lesions are not missed. We excluded such participants from this review as they were not our main focus.
Types of interventions
We had planned to include trials comparing any of the following interventions with another intervention in the list, provided that the only difference between the randomised groups was the management intervention or interventions being assessed.
Open surgical drainage;
Laparoscopic surgical drainage;
Percutaneous radiological drainage;
Endoscopic drainage;
Endoscopic drainage with guidance from endoscopic ultrasound (EUS‐guided drainage);
EUS‐guided drainage with nasocystic drainage;
Conservative management.
Types of outcome measures
Primary outcomes
-
Mortality.
Short‐term mortality (in‐hospital mortality or mortality within three months);
Long‐term mortality (at maximum follow‐up).
-
Serious adverse events (within three months). We accepted the following definitions of serious adverse events:
International Conference on Harmonisation ‐ Good Clinical Practice guideline (ICH‐GCP 1996): Serious adverse events, defined as any untoward medical occurrence that results in death, is life‐threatening, requires inpatient hospitalisation or prolongation of existing hospitalisation, results in persistent or significant disability, incapacity, or both;
Other variations of ICH‐GCP classifications such as Food and Drug Administration (FDA) classification (FDA 2006), Medicines and Healthcare products Regulatory Agency (MHRA) classification (MHRA 2013);
Clavien‐Dindo classification (Clavien 2009; Dindo 2004): Grade III or more;
Individual complications that could be clearly classified as Grade III or more with Clavien‐Dindo classification or as a serious adverse event with ICH‐GCP classification.
-
Health‐related quality of life (HRQoL), using any validated scale.
Short‐term (four weeks to three months);
Medium‐term (three months to one year);
Long‐term (longer than one year).
Secondary outcomes
Adverse events (within three months). We accepted all adverse events reported by the study author regardless of the severity of the event.
-
Measures of decreased complications and earlier recovery (within three months).
Length of hospital stay (including the index admission for intervention and any disease‐related or intervention‐related readmissions);
Time‐to‐return to normal activity (return to pre‐intervention mobility without additional caregiver support);
Time‐to‐return to work (for those who were previously employed).
Need for additional invasive intervention, such as drainage or reoperation in the case of surgical interventions, and redrainage or surgical intervention in the case of other interventions (long‐term).
Costs (within three months).
The choice of these clinical outcomes is based on the necessity to assess whether the interventions are safe and effective in the treatment of pancreatic pseudocysts. The costs provide an indication of resource requirement.
We did not regard the reporting of the outcomes listed here as an inclusion criterion for the review.
Search methods for identification of studies
Electronic searches
We conducted a literature search to identify all published and unpublished randomised controlled trials in all languages. We had planned to translate the non‐English language papers and fully assess them for potential inclusion in the review as necessary.
We searched the following electronic databases:
Cochrane Central Register of Controlled Trials (CENTRAL; The Cochrane Library 2015, Issue 9; Appendix 2);
MEDLINE (1966 to September 2015; Appendix 3);
EMBASE (1988 to September 2015; Appendix 4); and
Science Citation Index (1982 to September 2015; Appendix 5).
We also searched ClinicalTrials.gov (Appendix 6) and the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) on 8 September 2015 (Appendix 7).
Searching other resources
We checked reference lists of all primary studies and review articles for additional references. We attempted to contact authors of identified trials and asked them to identify other published or unpublished studies.
We searched for errata or retractions from eligible trials www.ncbi.nlm.nih.gov/pubmed on 4 January 2016.
Data collection and analysis
Selection of studies
Two review authors (KG and EP) independently screened titles and abstracts of all the potential studies we identified, and coded them as 'retrieve' (eligible or potentially eligible or unclear) or 'do not retrieve'. We retrieved the full‐text study reports, and two review authors (KG and EP) independently screened them and identified studies for inclusion; we identified and recorded reasons for exclusion of the ineligible studies. We resolved disagreements through discussion. We identified and excluded duplicates, and collated multiple reports of the same study, so that each study, rather than each report, was the unit of interest in the review. We had planned to contact the authors of trials of unclear eligibility. We recorded the selection process in sufficient detail to complete a PRISMA flow diagram and a 'Characteristics of excluded studies' table.
Data extraction and management
We used a standard data collection form for study characteristics and outcome data, which had been piloted on at least one study in the review. Two review authors (KG and EP) extracted the following study characteristics:
Methods: study design, total duration of study and run in, number of study centres and location, study setting, withdrawals, date of study.
Participants: number (N), mean age, age range, gender, presence of symptoms, average size of the pancreatic pseudocyst, inclusion criteria, exclusion criteria.
Interventions: intervention, comparison, concomitant interventions, number of participants randomised to each group.
Outcomes: primary and secondary outcomes specified and collected, time points reported. For binary outcomes, we obtained the number of participants with events and the number of participants included in the analysis in each group. For continuous outcomes, we obtained the unit or scale of measurement, mean, standard deviation, and the number of participants included in the analysis for each group. For count outcomes, we had planned to obtain the number of events and the number of participants included in the analysis in each group. For time‐to‐event outcomes, we had planned to obtain the proportion of people with events, the average duration of follow‐up of participants in the trial, and the number of participants included in the analysis for each group.
Notes: funding for trial, notable conflicts of interest of trial authors.
Two review authors (KG and EP) independently extracted outcome data from the included studies. If outcomes had been reported at multiple time points, we would have extracted the data for all time points. We obtained information on the number of participants with adverse events (or serious adverse events) and the number of such events, where applicable. We extracted costs, using the currency reported by trial authors and had planned to convert them to US dollars at the conversion rates on the day of the analyses. We extracted data for every trial arm that was an included intervention. If outcome data were reported in an unusable way, we attempted to contact the trial authors to obtain usable data. If we were unable to obtain usable data, we had planned to summarise the unusable data in an appendix. We resolved disagreements by consensus. One review author (KG) copied the data from the data collection form into the Review Manager 5 file. We double‐checked that the data were entered correctly by comparing the study reports with the data in the systematic review.
Assessment of risk of bias in included studies
Two review authors (KG and EP) independently assessed the risk of bias for each study, using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We resolved disagreement by discussion. We assessed the risk of bias according to the following domains:
Random sequence generation;
Allocation concealment;
Blinding of participants and personnel;
Blinding of outcome assessment;
Incomplete outcome data;
Selective outcome reporting;
Other potential bias.
We classified each potential source of bias as high, low, or unclear, and provided a quote from the study report, together with a justification for our judgement, in the 'Risk of bias' table. We summarised the risk of bias judgements across all studies for each of the domains listed. We had planned to consider blinding separately for different key outcomes where necessary, e.g., for unblinded outcome assessment, risk of bias for all‐cause mortality may be very different than for a participant‐reported pain scale. Where information on risk of bias related to unpublished data or to correspondence with a trial author, we had planned to note this in the 'Risk of bias' table. We had planned to present the risk of bias in each pair‐wise comparison in separate tables. However, there were only four trials and all the trials were considered to be at unclear or high risk of bias, so, we did not present this information.
When considering treatment effects, we took into account the risk of bias for the studies that contributed to that outcome by completing a sensitivity analysis.
Assesment of bias in conducting the systematic review
We conducted the review according to the published protocol, and have reported any deviations from it in the 'Differences between protocol and review' section of the review (Gurusamy 2014).
Measures of treatment effect
For dichotomous variables, such as short‐term mortality, proportion of participants with adverse events, requirement for additional interventions, we planned to calculate the odds ratio (OR) with a 95% credible interval (CrI). For continuous variables, such as length of hospital stay, time‐to‐return to normal activity, time‐to‐return to work, and costs, we had planned to calculate the mean difference (MD) with a 95% CrI. We had planned to use the standardised mean difference (SMD) with a 95% CrI for quality of life if different scales were used. For count outcomes, such as the number of adverse events, we had planned to calculate the rate ratio (RaR) with a 95% CrI. For time‐to‐event data, such as long‐term mortality, and a requirement for additional invasive intervention, such as reoperation in the case of surgical interventions, and surgical intervention in the case of other interventions, we had planned to use the hazard ratio (HR) with a 95% CrI. However, we did not perform a Bayesian network analysis and performed a simple indirect comparison using frequentist methods. So, we calculated the above treatment effects when data was available with 95% confidence intervals. Please see 'Data synthesis' for the reason for not performing the network meta‐analysis.
A common way that trial authors indicate that they have skewed data is by reporting medians and interquartile ranges. When we encountered this, we had planned to report the median and interquartile range in a table.
Unit of analysis issues
The unit of analysis was individual participants with a pancreatic pseudocyst. As anticipated, there were no cluster‐randomised trials for this comparison, but if we had identified any, we had planned to obtain the effect estimate adjusted for the clustering effect. If this was not available from the report or from the trial authors, we had planned to exclude the trial from the meta‐analysis.
In multi‐arm trials, the models account for the correlation between trial‐specific treatment effects from the same trial.
Dealing with missing data
We attempted to contact investigators or study sponsors in order to verify key study characteristics, and obtain missing numerical outcome data, where possible (e.g., when a study was identified as an abstract only). For binary, count, and time‐to‐event outcomes, we had planned to perform an intention‐to‐treat analysis whenever possible (Newell 1992). If this was not possible, we would have performed an available‐case analysis, but had planned to assess the impact of 'best‐best', 'best‐worst', 'worst‐best', 'worst‐worst' scenario analyses on the results for binary outcomes. For continuous outcomes, we performed an available‐case analysis. if we were unable to obtain the information from the investigators or study sponsors, we imputed the mean from the median (i.e., considered the median as the mean) and the standard deviation from the standard error, interquartile range, or P values, according to the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011); we assessed the impact of including such studies in a sensitivity analysis, as indicated. If we had been unable to calculate the standard deviation from the standard error, inter‐quartile range, or P values, we had planned to impute the standard deviation as the highest standard deviation in the remaining trials included in the outcome, being fully aware that this method of imputation would decrease the weight of the studies in the meta‐analysis of mean difference and that it would shift the effect towards no effect for standardised mean difference. We had planned to assess the impact of including such studies by performing sensitivity analyses.
Assessment of heterogeneity
We assessed the heterogeneity in each pair‐wise comparison by assessing the Higgins I² (Higgins 2003), the Chi² test with significance set at a P value less than 0.10, and visual inspection, whenever applicable. We also used the Tau² statistic to measure heterogeneity among the trials in each analysis. The Tau² statistic provides a measure of the variability of the effect estimate across studies in a random‐effects model (Higgins 2011). If we had identified substantial heterogeneity, we had planned to explore it by performing a meta‐regression.
Assessment of reporting biases
We attempted to contact trial authors, asking them to provide missing outcome data. Where this was not possible, and the missing data were thought to introduce serious bias, we had planned to explore the impact of including such studies in the overall assessment of results with a sensitivity analysis.
If we had been able to pool more than 10 trials for a specific comparison, we had planned to create and examine a funnel plot to explore possible publication bias. We had planned to use Egger's test to determine the statistical significance of the reporting bias (Egger 1997). We had planned to consider a P value of less than 0.05 statistically significant of reporting bias.
Data synthesis
We undertook meta‐analyses only where this was meaningful, i.e., if the treatments, participants and the underlying clinical question were similar. In general, we favoured performing a meta‐analysis, and had planned to clearly highlight the reason for not performing one if we decided against it. We had planned to conduct network meta‐analyses to compare multiple interventions simultaneously for each of the primary and secondary outcomes whenever applicable. Network meta‐analysis combines direct evidence within trials and indirect evidence across trials (Mills 2012).
We had planned to obtain network plots to ensure that the trials were connected by treatments using Stata/IC 11 (StataCorp LP; please see Appendix 8 for the Stata command we had planned to use). We had planned to apply network meta‐analysis to each connected network, and conduct a Bayesian network meta‐analysis using the Markov chain Monte Carlo method in WinBUGS 1.4. We had planned to model the treatment contrast (e.g., log OR for binary outcomes and MD for continuous outcomes) for any two interventions ('functional parameters') as a function of comparisons between each individual intervention and an arbitrarily selected reference group ('basic parameters'; Lu 2004). We had planned to use EUS‐guided drainage as the reference group. We had planned to perform the network analysis as per the guidance from the National Institute for Health and Clinical Excellence Decision Support Unit (NICE DSU) documents (Dias 2013).
Further details of the codes used and the technical details of how we planned to perform the analysis are shown in Appendix 9 and Appendix 10. In short, we had planned to use three non‐informative priors, a burn‐in of 30,000 simulations to ensure convergence (we planned to use longer burn‐in if the models did not converge in 30,000 simulations), and obtain the posterior estimates after a further 100,000 simulations. We had planned to use the fixed‐effect and random‐effects models (assuming homogeneous between‐trial variance across comparisons) for each outcome. We had planned to choose the fixed‐effect model if results were an equivalent or better fit (assessed by residual deviances, number of effective parameters, and deviance information criterion (DIC)) than the random‐effects model. A lower DIC indicates a better model fit. We had planned to use the random‐effects model if it resulted in a better model fit, as indicated by a DIC lower than that of the fixed‐effect model by at least three. In addition, we had planned to perform a treatment‐by‐design random‐effects inconsistency model (Higgins 2012; White 2012). We had planned to consider that the inconsistency model was a better model than the random‐effects consistency model (standard random‐effects network meta‐analysis model) if the model fit of the inconsistency model (as indicated by the DIC) was at least three lower than the random‐effects consistency model.
For multi‐arm trials, one can enter the data from all arms in a trial. This is entered as the number of people with events and the number of people exposed to the event, using the binomial likelihood and logit link for binary outcomes; the mean and standard error using the normal likelihood and identity link for continuous outcomes requiring calculation of the mean difference; the mean and standard error of the treatment differences using the normal likelihood and identity link for continuous outcomes requiring calculation of the standardised mean difference; the number of events and the number of people exposed to the event using the Poisson likelihood and log link for count outcomes; and follow‐up time in the study, number of people with the event and the number of people exposed to the event using the binomial likelihood and cloglog link for time‐to‐event outcomes. However, we did not encounter any multi‐arm trials.
We had planned to report the treatment contrasts (e.g., log ORs for binary outcomes and MDs for continuous outcomes) of the different treatments in relation to the reference treatment (i.e., EUS‐guided drainage), the residual deviances, the number of effective parameters, and the DIC for a fixed‐effect model and a random‐effects model for each outcome. We had also planned to report the parameters used to assess the model fit (i.e., residual deviances, number of effective parameters, and DIC) for the inconsistency model for all the outcomes, and the between‐trial variance for the random‐effects model (Dias 2012a; Dias 2012b; Higgins 2012; White 2012). If the inconsistency model had resulted in a better model fit than the consistency models, the transitivity assumption was likely to be untrue and the effect estimates obtained may not have be reliable. We had planned to highlight such outcomes where the inconsistency model resulted in a better model fit than the consistency models. We had planned to perform a separate network meta‐analysis for different subgroups to assess the inconsistency again. If there was no evidence of inconsistency in the revised analysis, we had planned to present the results of the analysis for different subgroups separately. If there was persistent evidence of inconsistency, we had planned to present the results from the direct comparison in the 'Summary of findings' table.
We had planned to calculate the 95% CrIs of treatment effects (e.g. ORs for binary outcomes, MDs for continuous outcomes, and so on) in the Bayesian meta‐analysis, which is similar in use to the 95% confidence intervals in the Frequentist meta‐analysis. These are the 2.5th percentile and 97.5th percentiles of the simulations. We had planned to report the mean effect estimate and the 95% CrI for each pair‐wise comparison in a table. We had also planned to estimate the probability that each intervention ranked at one of the possible positions and would have presented this information in graphs. It should be noted that one should not conclude that a treatment is the best one for an outcome if the probability of being the best is less than 90% (Dias 2012a). We had also planned to present the cumulative probability of the treatment ranks (i.e., the probability that the treatment is within the top two, the top three, etc.) in graphs. We had also planned to plot the probability that each treatment was best for each of the different outcomes (rankograms), which are generally considered more informative (Dias 2012a; Salanti 2011). However, because of sparse data, trials with zero events and lack of direct and indirect evidence for any comparisons, we only performed indirect comparisons, using methods described by Bucher, et al (Bucher 1997). Although we had planned to perform the direct comparisons using the same codes, we used the RevMan statistical algorithm for direct comparisons, which allowed us to present information in the standard Cochrane way.
In the presence of adequate data, where authors reported the outcomes of participants at multiple follow‐up time points, we had planned to follow the methods suggested by Lu 2007 to perform the meta‐analysis.
'Summary of findings' table
We created a 'Summary of findings' table that reported on all the outcomes. We used the five GRADE considerations (study limitations, consistency of effect, imprecision, indirectness and publication bias) to assess the quality of the body of evidence as it related to the studies that contributed data to the meta‐analyses for the prespecified outcomes. We had planned to use methods and recommendations described in the GRADE Working Group approach for rating the quality of treatment effect estimates from network meta‐analysis (Puhan 2014). However, since the network meta‐analysis was not performed, we only presented the results of direct and indirect comparisons. We justified all decisions to down‐ or upgrade the quality rating of the evidence using footnotes, and made comments to aid the reader's understanding of the review where necessary. We considered whether there was any additional outcome information that we were not able to incorporate into meta‐analyses and noted this in the comments.
Subgroup analysis and investigation of heterogeneity
We had planned to assess the differences in the effect estimates between the following subgroups using meta‐regression, with the help of the code shown in Appendix 6, when at least one trial was included in each subgroup.
Symptomatic versus asymptomatic participants.
Reason for pancreatic pseudocyst (acute pancreatitis, chronic pancreatitis, trauma).
Mean size of the pseudocyst. Although size is not the main reason for the treatment, observational studies have found that the mean size of pseudocysts was higher in people who required surgery after conservative management, compared to those who did not require surgery after conservative management (7 cm versus 5 cm) (Vitas 1992).
We had planned to calculate the interaction term (Dias 2012c). If the 95% CrI of the interaction term did not overlap zero, we had planned to consider this statistically significant.
Sensitivity analysis
We had planned to perform the following sensitivity analyses defined a priori to assess the robustness of our conclusions.
Excluding trials at unclear or high risk of bias (one or more of the risk of bias domains classified as unclear or high).
Excluding trials in which either the mean, the standard deviation, or both were imputed.
Imputation of binary outcomes under 'best‐best', 'best‐worst', 'worst‐best', and 'worst‐worst' scenarios.
Reaching conclusions
We based our conclusions only on findings from the quantitative or narrative synthesis of included studies for this review. We avoided making recommendations for practice, and our implications for research give the reader a clear sense of the remaining uncertainties, and where future research in this area should focus.
Results
Description of studies
Results of the search
We identified 2467 references through electronic searches of Cochrane (Wiley; N = 200), MEDLINE (OvidSP; N = 1262), EMBASE (OvidSP; N = 736), Science Citation Index expanded (N = 255), ClinicalTrials.gov (N = 1), and WHO Trials register (N = 13). After removing duplicate references, there were 2052 references remaining. We excluded 2041 clearly irrelevant references by reading titles and abstracts. One reference was identified by reference searching. The full publication of 12 references were retrieved for further detailed assessment. One reference was excluded since it was a comment on an included study (Sauer 2010). One reference was an ongoing study from the WHO ICTRP trial register (NCT02041793). Although it appears to have completed recruitment, we were unable to find any written record of the results. In total, ten references describing four trials fulfilled the inclusion criteria (Characteristics of included studies). The reference flow is shown in Figure 1.
1.
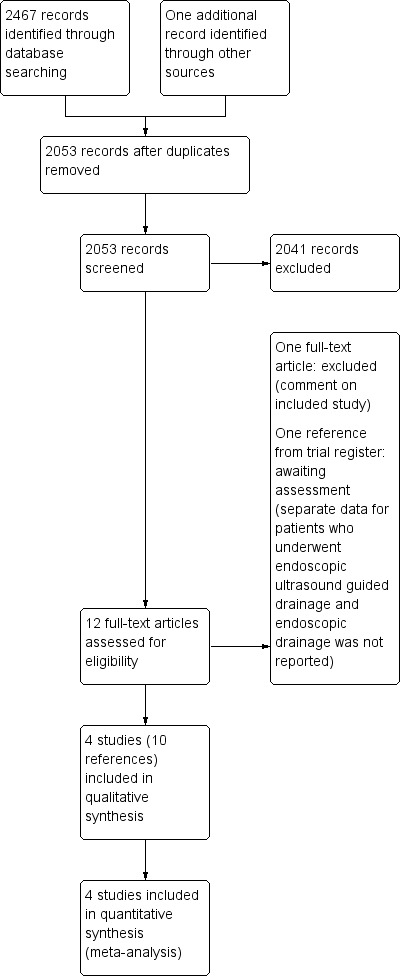
Study flow diagram.
Included studies
Four randomised controlled trials (RCT) were included in this review; they were all two‐armed trials (Park 2009; Varadarajulu 2008; Varadarajulu 2013; Yuan 2015). Three trials included only symptomatic patients with pseudocysts (Park 2009; Varadarajulu 2008; Varadarajulu 2013). This information was not available in Yuan 2015. Two studies reported that they included patients with pseudocysts from acute and chronic pancreatitis due to various aetiologies (Park 2009; Varadarajulu 2013). However, these studies did not report the data separately for people with acute and chronic pancreatitis. The remaining two trials did not report information on whether the pseudocyst was associated with acute or chronic pancreatitis. The mean size of the pseudocysts across studies ranged between 70 mm and 155 mm. No separate data were available for pseudocysts of different sizes. Infected pseudocysts were excluded in two trials (Park 2009; Varadarajulu 2008). This information was not available in the remaining two trials (Varadarajulu 2013; Yuan 2015).
A total of 177 participants were randomised in the four trials. One trial excluded a participant with suspicion of a pancreatic adenoma, leaving a total of 176 participants (Varadarajulu 2008). The interventions and length of follow‐up were as follows; all were compared to EUS‐guided drainage:
Park 2009: Endoscopic drainage (29 participants) versus EUS‐guided drainage (31 participants). Follow‐up: minimum six months.
Varadarajulu 2008: Endoscopic drainage (15 participants) versus EUS‐guided drainage (14 participants). Follow‐up: minimum six weeks.
Yuan 2015: EUS‐guided drainage with nasocystic drainage (24 participants) versus EUS‐guided drainage (23 participants). Follow‐up: minimum 24 months.
Varadarajulu 2013; Open surgical drainage (20 participants) versus EUS‐guided drainage (20 participants). Follow‐up: minimum 24 months.
Pseudocysts were drained into the stomach in two trials (Varadarajulu 2013; Yuan 2015), and into the stomach or duodenum in two trials (Park 2009; Varadarajulu 2008). Routine stent removal was reported in two trials (Varadarajulu 2008; Varadarajulu 2013). In Varadarajulu 2008, the stents were removed after six weeks in those with successful resolution of cysts in both groups. In Varadarajulu 2013, the stents were removed after eight weeks in the EUS‐guided drainage group following CT scan confirmation of the successful resolution of the cysts. The timing of stent removal in either group was not reported in the remaining trials.
Study details of the population, interventions, outcomes and risk of bias are listed in the Characteristics of included studies table and Table 3.
1. Characteristics table (arranged according to comparisons).
| Study name Inclusion and exclusion criteria | Number of people in intervention group | Number of people in control group | Risk of bias | ||||||
| Random sequence generation | Allocation concealment | Blinding of participants and personnel | Blinding of outcome assessment | Incomplete outcome data | Selective reporting | Other bias | |||
| Endoscopic drainage versus EUS guided drainage | |||||||||
| Park 2009 | 31 | 29 | Low | Unclear | High | High | Low | Low | Unclear |
| Inclusion criteria 1. History of pancreatitis. 2. Symptomatic pseudocysts with duration of longer than 4 weeks. Exclusion criteria 1. Age less than 18 years. 2. A pancreatic pseudocyst more than 4 cm in size and communicating with the pancreatic duct. 3. A suggested pancreatic abscess or necrosis, shown by computed tomography (CT). 4. A moderate thickness (> 10 mm) from the cyst wall to the intramural wall, shown by EUS. 5. Portal hypertension or coagulopathy. | |||||||||
| Varadarajulu 2008 | 14 | 15 | Low | Low | High | Unclear | High | Low | Unclear |
| Inclusion criteria 1. History of pancreatitis. 2. Symptomatic pseudocysts. Exclusion criteria 1. CT findings were suggestive of pathology other than a pseudocyst. 2. Pseudocyst was ≤ 4 cm in size. 3. Patients younger than 18 years of age. 4. Patients with pancreatic abscess or necrosis by CT. | |||||||||
| EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | |||||||||
| Yuan 2015 | 23 | 24 | Unclear | Low | Unclear | Unclear | Unclear | High | Unclear |
| Inclusion criteria 1. Ages 16 to 70 years. 2. Clinical presentation with abdominal distension with or without upper gastrointestinal obstruction. 3. Radiological findings suggestive of a pancreatic pseudocyst > 10 cm. Exclusion criteria 1. Cysts without adherence to the gastric wall. 2. Distance between pseudocyst and gastric wall greater than 1 cm. 3. Pseudocyst communication with the main pancreatic duct. 4. Pregnancy. 5. Pancreatic tumors. 6. Risk for anaesthesia and surgery. 7. Contraindications to magnetic resonance cholangiopancreatography and endoscopic retrograde cholangiopancreatography. 8. Liver cirrhosis. 9. Previous history of abdominal surgery (e.g. gastrectomy). 10. Inability to give informed consent. | |||||||||
| Open surgical drainage versus EUS‐guided drainage | |||||||||
| Varadarajulu 2013 | 20 | 20 | Low | Low | High | High | Low | Low | Low |
| Inclusion criteria 1. Diagnosis of pancreatic pseudocyst based on CT criteria. 2. Pseudocyst measuring >= 6 cm in size and located adjacent to the stomach. 3. Documented history of acute or chronic pancreatitis. 4. Persistent pancreatic pain requiring narcotics or analgesics. 5. Symptomatic gastric outlet or bile duct obstruction induced by the pseudocyst. Exclusion criteria 1. Age < 18 or > 80 years. 2. Contraindications to surgery: ASA class IV, severe portal hypertension. 3. Contraindication to endoscopic drainage: gastrectomy with Billroth II reconstruction, gastric bypass surgery, prior surgery for pancreas‐related complications. 4. Pregnancy. 5. Associated pancreatic necrosis on CT. 6. Pseudocyst not adjacent to the stomach. 7. Multiloculated pseudocyst or multiple pseudocysts. | |||||||||
Excluded studies
One reference was excluded since this was a comment on an included study (Sauer 2010).
Risk of bias in included studies
None of the included trials were at low risk of bias. The risk of bias in the individual domains are summarised in Figure 2 and Figure 3.
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Two trials reported the allocation sequence generation and allocation concealment adequately and are at low risk of selection bias (Varadarajulu 2008; Varadarajulu 2013). The remaining trials did not report the allocation sequence generation (Yuan 2015), or allocation concealment (Park 2009) and hence are at unclear risk of selection bias.
Blinding
It is not possible to blind the endoscopist or surgeons performing the procedures. However, it is possible to blind the participants (except for the comparison between endoscopic and surgical methods) and outcome assessors, endoscopists or surgeons who make clinical decisions after the procedure. None of the trials reported on blinding of the participants or outcome assessors. Hence, all the trials were at unclear risk of performance bias and unclear risk of detection bias for all outcomes other than mortality. All‐cause mortality was reported and it was unlikely that the lack of blinding introduced bias in the assessment of mortality.
Incomplete outcome data
Three trials reported the participant flow clearly (Park 2009; Varadarajulu 2008; Varadarajulu 2013). Of these, there were no post‐randomisation drop‐outs in two trials (Park 2009; Varadarajulu 2013). So, these two trials are at low risk of attrition bias. In one trial, one participant randomised to EUS‐guided drainage group was excluded from analysis since the EUS revealed septae in the cyst, leading to a suspicion of pancreatic adenoma (Varadarajulu 2008). However, identifying septae is only possible with EUS‐guided drainage and is a potential advantage of the procedure. Hence, the overall benefit of EUS‐guided drainage may have been underestimated. Nevertheless, the exclusion of this patient was unlikely to result in bias for any of the reported outcomes. So, this trial was also considered to be at low risk of attrition bias. The fourth trial was considered to be at unclear risk of attrition bias since it did not report the participant flow (Yuan 2015).
Selective reporting
None of the trials had a pre‐published protocol. Three trials reported the clinical outcomes that were likely to be measured in such clinical trials and were considered to be at low risk of selective reporting (Park 2009; Varadarajulu 2008; Varadarajulu 2013). Yuan 2015 did not report morbidity clearly and was considered to be at risk of selective outcome reporting bias.
Other potential sources of bias
One trial advised they had no external funding (Varadarajulu 2013). The source of funding in the remaining trials was not reported. There was no other bias noted in the remaining trials.
Effects of interventions
The effects of interventions have been summarised in Table 1 and Table 2, None of the trials reported the following outcomes: long‐term mortality, quality of life at medium‐term (three months to one year) or long‐term (more than one year), time‐to‐return to normal activity, and time‐to‐return to work.
Only one comparison (endoscopic drainage versus EUS‐guided drainage) was examined in more than one trial. So, heterogeneity and random‐effects model are only presented for this comparison, when both trials reported the outcome (Park 2009; Varadarajulu 2008).
Primary outcomes
1. Short‐term mortality
All four trials reported short‐term mortality (Park 2009; Varadarajulu 2008; Varadarajulu 2013; Yuan 2015). There was only one death reported in the endoscopic drainage group, due to major bleeding (Varadarajulu 2008). A direct comparison using the RevMan statistical algorithm revealed no statistically significant difference between the EUS‐guided drainage and endoscopic drainage groups (OR 3.00; 95% CI 0.11 to 79.91; participants = 89; studies = 2; Analysis 1.1). Since there was only one death in one arm of one study, a network meta‐analysis was not performed and the issues of fixed‐effect model versus random‐effects model and heterogeneity did not arise. The absolute proportions of short‐term mortality in different interventions are as follows.
1.1. Analysis.
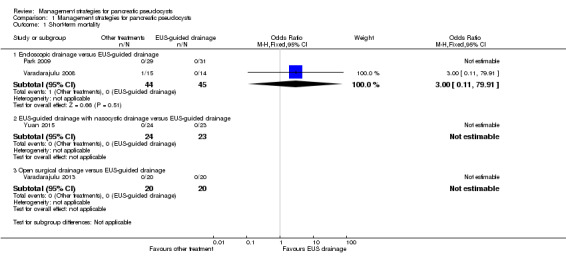
Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 1 Short‐term mortality.
EUS‐guided drainage 0% (0/88);
Endoscopic drainage 2.3% (1/44);
EUS‐guided drainage with nasocystic drainage 0% (0/24);
Open surgical drainage 0% (0/20).
Long‐term mortality
None of the trials reported long‐term mortality.
2. Serious adverse events
Three trials (129 participants) reported serious adverse events, which included: bleeding requiring additional intervention, pneumoperitoneum, stent migration, difficulty in eating requiring surgical placement of feeding tube, and pancreatic stricture requiring distal pancreatectomy (Park 2009; Varadarajulu 2008; Varadarajulu 2013).
Direct comparison
As shown in Analysis 1.2, there were no statistically significant differences in any of the direct comparisons (endoscopic drainage versus EUS‐guided drainage: OR 2.42; 95% CI 0.51 to 11.46; participants = 89; studies = 2; open surgical drainage versus EUS‐guided drainage: OR 8.20; 95% CI 0.40 to 169.90; participants = 40; studies = 1). There was no evidence of heterogeneity in the endoscopic drainage versus EUS‐guided drainage comparison (I² = 0; Chi² test for heterogeneity = 0.53), and no difference in the interpretation of results using either the fixed‐effect or random‐effects models.
1.2. Analysis.

Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 2 Serious adverse events.
The absolute unadjusted proportions of people with serious adverse events in different interventions were:
EUS‐guided drainage: 3.1% (2/65);
Endoscopic drainage: 11.4% (5/44);
Open surgical drainage: 15.0% (3/20).
Indirect comparison
As shown in Figure 4, there was no statistically significant difference in the proportion of people with serious adverse events between endoscopic drainage and open surgical drainage (OR 0.30; 95% CI 0.01 to 8.87).
4.
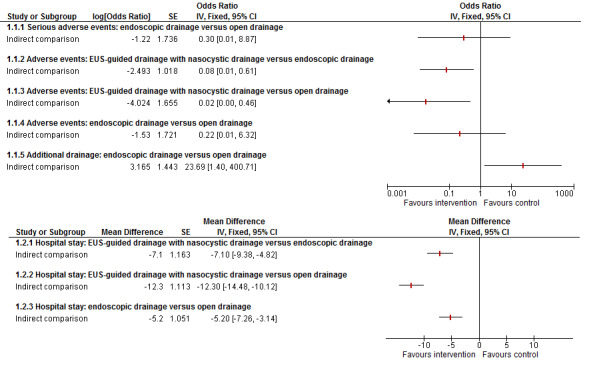
Indirect comparisons for adverse events and length of hospital stay
3. Health‐related quality of life (HRQoL)
Short‐term: four weeks to three months
Only one trial reported HRQoL, using the SF‐36 scale (Varadarajulu 2013). Data for general health perception were obtained to compare the HRQoL results (range of scores: 0 to 100, with 100 indicating maximum health). Health‐related quality of life was statistically significantly worse in the open surgical drainage group than in the EUS‐guided drainage group, using the RevMan statistical algorithm (MD ‐21.00; 95% CI ‐33.21 to ‐8.79; participants = 40; studies = 1; Analysis 1.3). Since this was the only trial reporting the outcome, a network meta‐analysis was not performed and the issues of fixed‐effect model versus random‐effects model and heterogeneity did not arise.
1.3. Analysis.
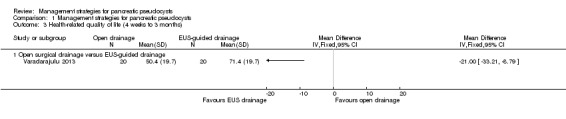
Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 3 Health‐related quality of life (4 weeks to 3 months).
Medium term: three months to one year and long‐term: more than one year
None of the trials reported HRQoL at medium‐term (three months to one year) or long‐term (more than one year).
Secondary outcomes
1. Adverse events
All four trials (176 participants) reported adverse events of any severity (Park 2009; Varadarajulu 2008; Varadarajulu 2013; Yuan 2015).
Direct comparison
As shown in Analysis 1.4, the proportion of people with any adverse events was statistically significantly lower with EUS‐guided drainage with nasocystic drainage compared to EUS‐guided drainage (OR 0.20; 95% CI 0.06 to 0.73; participants = 47; studies = 1). There were no statistically significant differences in any of the remaining direct comparisons (endoscopic drainage versus EUS‐guided drainage: OR 2.42; 95% CI 0.51 to 11.46; participants = 89; studies = 2; open surgical drainage versus EUS‐guided drainage: OR 11.18; 95% CI 0.56 to 222.98; participants = 40; studies = 1). There was no evidence of heterogeneity in the comparison of endoscopic drainage versus EUS‐guided drainage (I² = 0; Chi² test for heterogeneity = 0.53; Tau² statistic = 0; or visual inspection). There was no difference in the interpretation of results using fixed‐effect or random‐effects models.
1.4. Analysis.

Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 4 Adverse events.
The absolute unadjusted proportions of people with adverse events in different interventions are:
EUS‐guided drainage 17.0% (15/88);
Endoscopic drainage 11.4% (5/44);
EUS‐guided drainage with nasocystic drainage 20.8% (5/24);
Open surgical drainage 20.0% (4/20).
Indirect comparison
As shown in Figure 4, those who received EUS‐guided drainage with nasocystic drainage had fewer adverse events when compared to endoscopic drainage (OR 0.08; 95% CI 0.01 to 0.61) or open drainage (OR 0.02; 95% CI 0.00 to 0.46). There was no statistically significant differences in the proportion of people with adverse events between the endoscopic drainage and open drainage groups (OR 0.22; 95% CI 0.01 to 6.32).
2. Measures of decreased complications and earlier recovery (within three months)
2a. Length of hospital stay
Three trials (116 participants) reported length of hospital stay (Varadarajulu 2008; Varadarajulu 2013; Yuan 2015).
Direct comparison
As shown in Analysis 1.5, the length of hospital stay was statistically significantly shorter in the EUS‐guided drainage with nasocystic drainage compared to EUS‐guided drainage (MD ‐8.10 days; 95% CI ‐9.79 to ‐6.41; participants = 47; studies = 1), and statistically significantly longer in the open surgical drainage group compared to the EUS‐guided drainage group (MD 4.20 days; 95% CI 2.82 to 5.58; participants = 40; studies = 1). There was no statistically significant difference in the comparison between endoscopic drainage and EUS‐guided drainage (MD ‐1.00 days; 95% CI ‐2.53 to 0.53; participants = 29; studies = 1). Since there was only one study for the different comparisons, the issues of heterogeneity and fixed‐effect versus random‐effects model did not arise. Exclusion of one trial in which the mean was imputed from the median, and the standard deviation was calculated from the P value did not alter the interpretation of results, other than there was no information available in the comparison of hospital stays between the endoscopic drainage and EUS‐guided drainage groups (Varadarajulu 2008).
1.5. Analysis.
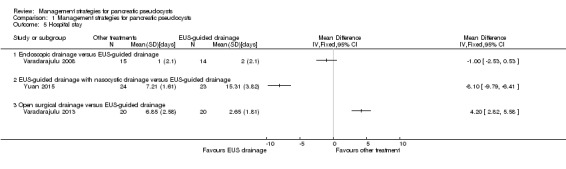
Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 5 Hospital stay.
Indirect comparison
As shown in Figure 4, the EUS‐guided drainage with nasocystic drainage group had a shorter hospital stay compared to the endoscopic drainage group (MD ‐7.10 days; 95% CI ‐9.38 to ‐4.82), or open surgical drainage group (MD ‐12.30 days; 95% CI ‐14.48 to ‐10.12). The endoscopic drainage group had a shorter hospital stay than the open drainage group (MD ‐5.20 days; 95% CI ‐7.26 to ‐3.14).
2b & 2c.Time‐to‐return to normal activity and time‐to‐return to work
None of the trials reported either the time‐to‐return to normal activity or the time‐to‐return to work.
3. Need for additional invasive intervention
Three trials (129 participants) reported that additional invasive interventions were required for drainage (Park 2009; Varadarajulu 2008; Varadarajulu 2013).
Direct comparison
As shown in Analysis 1.6, the proportion of people requiring additional invasive intervention for drainage was statistically significantly higher with endoscopic drainage than EUS‐guided drainage (OR 11.13; 95% CI 2.85 to 43.44; participants = 89; studies = 2). There was moderate heterogeneity (I² = 44%; Chi² test for heterogeneity = 0.18; Tau² statistic = 1.21; and visual inspection), but the heterogeneity appeared to be in the magnitude rather than direction of effect. There was no difference in the interpretation of results using either fixed‐effect or random‐effects models for this comparison. There were no statistically significant differences in the need for additional drainage between the open surgical and EUS‐guided drainage groups (OR 0.47; 95% CI 0.04 to 5.69; participants = 40; studies = 1).
1.6. Analysis.
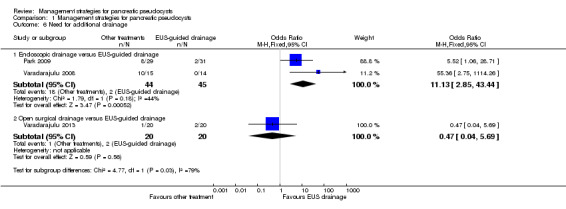
Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 6 Need for additional drainage.
The absolute unadjusted proportions of need for additional drainage in different interventions were:
EUS‐guided drainage 6.2% (4/65);
Endoscopic drainage 40.9% (18/44);
Open surgical drainage 5.0% (1/20).
Indirect comparison
As shown in Figure 4, the proportion of people requiring additional drainage was statistically significantly more in the endoscopic drainage group than in the open surgical drainage group (OR 23.69; 95% CI 1.40 to 400.71).
4. Costs
Only one trial reported costs (Varadarajulu 2013). Treatment costs were statistically significantly higher in the open surgical drainage group compared to the EUS‐guided drainage group, using the RevMan statistical algorithm (MD 8040 USD; 95% CI 3020 to 13,060; participants = 40; studies = 1; Analysis 1.7). Since this was the only trial reporting the outcome, we did not perform a network meta‐analysis, and the issues of fixed‐effect versus random‐effects model and heterogeneity did not arise.
1.7. Analysis.

Comparison 1 Management strategies for pancreatic pseudocysts, Outcome 7 Costs.
Subgroup analysis
We did not perform any of the planned subgroup analyses. Three trials included only symptomatic patients with pseudocysts (Park 2009; Varadarajulu 2008; Varadarajulu 2013). This information was not available in Yuan 2015. Two studies included patients with a pseudocyst from acute or chronic pancreatitis due to various aetiologies, but did not report the data separately (Park 2009; Varadarajulu 2013). The remaining two trials did not report whether the pseudocyst was associated with acute or chronic pancreatitis. None of the trials provided separate data for pseudocysts of different sizes.
Sensitivity analyses
We did not perform the sensitivity analysis of excluding trials at unclear or high risk of bias, since all the trials fell into this category. We did not perform a sensitivity analysis of imputation of binary outcomes under 'best‐best', 'best‐worst', 'worst‐best', or 'worst‐worst' scenarios because we did not consider that the exclusion of one patient due to pancreatic adenoma would result in attrition bias (please see 'Incomplete outcome data (attrition bias)'). The sensitivity analysis of excluding trials in which the mean, standard deviation, or both were imputed has been presented under the individual outcomes.
Reporting bias
We did not explore reporting bias using a funnel plot because we had fewer than 10 trials for each comparison.
Discussion
Summary of main results
We included four randomised controlled trials that included 176 participants and compared different methods of draining pseudocysts with EUS‐guided drainage (Park 2009; Varadarajulu 2008; Varadarajulu 2013; Yuan 2015). Network meta‐analysis provided inferior quality of evidence than direct comparison for all comparisons that used EUS‐guided drainage as the control group because of the indirectness it introduces, particularly since we were unable to assess the transitivity assumption. However, it provided the effect estimates for other pair‐wise comparisons when direct comparison was not available, the reliability of such effect estimates when the transitivity assumption could not be tested formally is not known.
There were no statistically significant differences between the treatments in short‐term mortality or the proportion of people with serious adverse events. Short‐term (four weeks to three months) health‐related quality of life (HRQoL) was worse and the costs were higher in the open surgical drainage group than in the EUS‐guided drainage group. Participants who received EUS‐guided drainage with nasocystic drainage had fewer adverse events when compared to those who received either EUS‐guided or endoscopic drainage. Participants who received EUS‐guided with nasocystic drainage also had shorter hospital stays when compared to EUS‐guided drainage, endoscopic drainage, or open surgical drainage, while those who received open surgical drainage had longer hospital stays than those who received EUS‐guided drainage. The need for additional invasive interventions was significantly higher with endoscopic drainage than with EUS‐guided drainage, while it was statistically significantly lower in those who received open surgical drainage than in those who received endoscopic drainage. There were no statistically significant differences in the other comparisons.
None of the trials reported on long‐term mortality, medium‐term HRQoL (three months to one year), long‐term HRQoL (longer than one year), time‐to‐return to normal activities, or time‐to‐return to work.
Overall completeness and applicability of evidence
Three trials only included symptomatic patients with pseudocysts (Park 2009; Varadarajulu 2008; Varadarajulu 2013), while Yuan 2015 did not state whether asymptomatic patients were included. So, the results of this study are only applicable for symptomatic patients with pseudocysts. Two trials clearly stated that they included pseudocysts that had resulted from acute or chronic pancreatitis due to various aetiologies (Park 2009; Varadarajulu 2013). The remaining two trials did not report on this association. Nevertheless, it appears that the results are applicable to patients with pseudocysts resulting from acute and chronic pancreatitis, the major causes of pseudocysts. The mean size of the pseudocysts ranged between 70 mm and 155 mm across all included studies. Thus, the results of this review are applicable for moderate to large pseudocysts. Infected pseudocysts were excluded in two trials (Park 2009; Varadarajulu 2008). This information was not available in the remaining two trials (Varadarajulu 2013; Yuan 2015). So, the results of this review are only applicable for non‐infected pseudocysts.
Quality of the evidence
The overall quality of evidence was low or very low for all the outcomes. There was no blinding of outcome assessors for any of the comparisons. All statistically significant outcomes were subjective outcomes and may be influenced by the lack of blinding. As a result, the risk of bias was unclear or high for most of the outcomes. It was not possible to assess the consistency of evidence since two comparisons, EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage and open surgical drainage versus EUS‐guided drainage had only one trial. There was no evidence of inconsistency in the comparison of endoscopic drainage with EUS‐guided drainage for serious adverse and adverse events, while there was moderate inconsistency for the need for additional drainage for the same comparison. However, the inconsistency in the need for additional drainage appears to be in the magnitude, rather than in the direction of effect. We were unable to assess the consistency for length of hospital stay since only one of the two trials comparing endoscopic drainage with EUS‐guided drainage reported it (Varadarajulu 2008). Another major issue was the small sample size in the trials, which resulted in imprecise results.
Potential biases in the review process
We followed the guidance in the NICE DSU documents and the Cochrane Handbook for Systematic Reviews of Interventions for the direct comparisons in the review (Dias 2013; Higgins 2011). Two people selected studies and extracted data, reducing the errors in data collection. We used formal search strategies to identify trials. While the likelihood of missing trials from identified references is low, the search strategy included the time‐frame before the mandatory trial registration era and it is possible that some trials were not reported in journals because of their results. However, one has to be pragmatic and accept that this is the best evidence that is currently available.
We imputed the mean and calculated the standard deviation for length of hospital stay for one trial from the median and P value (Varadarajulu 2008). Excluding this trial altered the conclusions by withdrawing information on the length of hospital stay for those who received endoscopic drainage.
While network meta‐analysis has its advantages in combining direct and indirect evidence (resulting in more precise evidence) and Bayesian network meta‐analysis allows the calculation of the probability of best treatments, these advantages were limited in this review because of the sparse data. All comparisons had at least one trial that had no events in one arm, disallowing direct and indirect evidence for a single comparison. So, we conducted direct comparisons using the Frequentist methods and conducted indirect comparisons using the methods described in Bucher 1997. The effect estimates of indirect comparisons and network meta‐analysis assume that the transitivity assumption is met. While the participants appeared to be similar across trials of various comparisons, we were unable to verify this using statistical methods, since none of the comparisons had direct and indirect evidence. So, we were not able to verify the transitivity assumption statistically. This decreases the confidence in indirect estimates.
Agreements and disagreements with other studies or reviews
This is the first systematic review of randomised controlled trials on pancreatic pseudocysts. There has been one previous review comparing open surgical drainage with endoscopic or EUS‐guided drainage for pancreatic pseudocysts, but this review included non‐randomised studies (Zhao 2016). The review authors concluded that the endoscopic group (endoscopic drainage or EUS‐guided drainage) was associated with a shorter length of hospital stay and lower hospital costs compared to the surgical group. We do not agree with this conclusion, since there is significant uncertainty in these statements due to the small sample size in the trials, and the lack of replication of the findings in two or more studies. In addition, only one trial included the costs of stent removal. None of the trials included the complications and length of hospital stay resulting from stent removal. Although stent removal can be considered a relatively minor procedure compared to the treatments, it does incur costs and may cause complications and longer hospital stays.
Authors' conclusions
Implications for practice.
Very low‐quality evidence suggested that the differences in mortality and serious adverse events between treatments were imprecise. Low‐quality evidence suggested that short‐term health‐related quality of life (four weeks to three months) was worse and the costs were higher in the open surgical drainage group than in the EUS‐guided drainage group. Low‐quality or very low‐quality evidence suggested that the EUS‐guided drainage with nasocystic drainage group had fewer adverse events compared to the EUS‐guided drainage or endoscopic drainage groups, and shorter hospital stays compared to those who received EUS‐guided drainage, endoscopic drainage, or open surgical drainage, while those who received EUS‐guided drainage had shorter hospital stays than those with open surgical drainage. Low‐quality evidence suggested that the need for additional invasive intervention was higher with endoscopic drainage than EUS‐guided drainage, while it was lower in the open surgical drainage than in the endoscopic drainage group.
Implications for research.
Further randomised controlled trials are needed in patients with symptomatic pancreatic pseudocysts requiring treatment. The most effective interventions in this review appear to be EUS‐guided drainage with or without nasocystic drainage, so, it is reasonable to start here. Other possible interventions include variations of EUS‐guided drainage (with or without nasocystic drainage) using self‐expanding stents (Huggett 2015), or EUS‐guided drainage with nasocystic drainage compared with laparoscopic drainage. Future trials should include patient‐oriented outcomes, such as mortality, serious adverse events, health‐related quality of life, length of hospital stay, return‐to‐normal activity, and the number of work days lost. All outcomes should be measured at least until the end of the draining procedure and the removal of the stent, with a minimum follow‐up period of two to three years, to ensure there are no recurrences of pseudocysts.
What's new
| Date | Event | Description |
|---|---|---|
| 13 September 2016 | Amended | A typographic error in the Plain Language Summary was corrected. |
Acknowledgements
We thank Karin Dearness, Managing Editor, Cochrane Upper Gastrointestinal and Pancreatic Diseases (UGPD) Group for providing administrative and logistical support for the conduct of the current review.
We thank the copy editors and Cochrane Editorial Unit for their comments.
Appendices
Appendix 1. Glossary of terms
Acute: sudden.
Aetiology: cause.
Amylase: an enzyme that breaks down carbohydrates.
Aspiration cytology: inserting a hollow needle into the body to obtain cells from an organ and examining the cells under microscopy to determine presence of cancer.
Cystoenterostomy: opening between the pseudocyst and small intestine.
Cystogastrostomy: opening between the pseudocyst and stomach.
Distal pancreatectomy: removal of body and tail of the pancreas.
Duodenal: relating to the first part of the small intestines.
Endoscopic: with the help of an endoscope, a tube inserted into the body (in this context, through the mouth and into the stomach and upper part of the small intestine).
Enzyme: substances that enable and speed up chemical reactions that are necessary for the normal functioning of the body.
Epigastric pain: upper central abdominal pain.
Gastric: related to stomach.
Gastrointestinal tract: digestive tract.
Haemorrhage: bleeding.
Insulin: substance which helps regulate blood sugar.
Intrapancreatic: within the pancreas.
Morbidity: illness (in this context, it means complications)
Mortality: death
Necrosis: death and decomposition of living tissue, usually caused by lack of blood supply, but can be caused by other pathological insult.
Neoplasm: tumour.
Oesophagoduodenoscope: inserting an endoscope, a tube inserted into the food pipe and stomach to view them internally (in this context).
Pathologic insult: substance or mechanism that causes the condition.
Percutaneous drainage: drainage carried out by insertion of a drain from the external surface of the body, usually guided by an ultrasound or CT (computed tomography) scan.
Peripancreatic tissues: tissues surrounding the pancreas.
Peritoneal cavity: abdominal cavity (tummy).
Portal vein: vein that transports the blood from the intestines to the liver.
Pneumoperitoneum: inflation of the abdomen (tummy) with gas, in this context, because of a hole in the bowel.
Pseudoaneurysm: false aneurysm (blood clot outside the arterial wall formed by a leak through a hole in the artery; the leak is contained by the surrounding tissues).
Protease: an enzyme that digests protein.
Retroperitoneum: behind the abdominal cavity.
Serum: clear fluid that separates out when blood clots.
Sinistral portal hypertension: left‐sided portal hypertension (high pressure in the portal vein)
Splenic vein: vein that drains the spleen.
Stricture: narrowing of a passage within the body.
Thrombosis: blood clot.
Transenteric endoscopic management: drainage of the pseudocyst through the intestines.
Transient: temporary.
Transpapillary endoscopic management: drainage of the pseudocyst through the papilla (ampulla of Vater) which is formed by the union of the pancreatic duct and the common bile duct.
Appendix 2. CENTRAL search strategy
#1 MeSH descriptor: [Pancreatic Pseudocyst] this term only
#2 (pancrea* near/2 pseudocyst*)
#3 MeSH descriptor: [Pancreatitis, Chronic] this term only and with qualifier(s): [Complications ‐ CO]
#4 MeSH descriptor: [Constriction, Pathologic] this term only and with qualifier(s): [Complications ‐ CO, Diagnosis ‐ DI, Etiology ‐ ET]
#5 MeSH descriptor: [Abscess] this term only
#6 pancrea*
#7 #5 and #6
#8 ((peri‐pancreatic or peripancreatic or pancrea*) near/2 (abscess or fluid))
#9 #1 or #2 or #3 or #4 or #7 or #8
Appendix 3. MEDLINE search strategy
1. Pancreatic Pseudocyst/
2. (pancrea* adj2 pseudocyst*).mp.
3. Pancreatitis, Chronic/co [Complications]
4. Constriction, Pathologic/co, di, et [Complications, Diagnosis, Etiology]
5. Abscess/
6. pancrea*.mp.
7. 5 and 6
8. ((peri‐pancreatic or peripancreatic or pancrea*) adj2 (abscess or fluid)).mp.
9. 1 or 2 or 3 or 4 or 7 or 8
10. randomized controlled trial.pt.
11. controlled clinical trial.pt.
12. randomized.ab.
13. placebo.ab.
14. drug therapy.fs.
15. randomly.ab.
16. trial.ab.
17. groups.ab.
18. 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17
19. exp animals/ not humans.sh.
20. 18 not 19
21. 9 and 20
Appendix 4. EMBASE search strategy
1. Pancreatic Pseudocyst/
2. (pancrea* adj2 pseudocyst*).mp.
3. Pancreatitis, Chronic/co [Complications]
4. common bile duct obstruction/
5. common bile duct stenosis/
6. pancreas duct stenosis/
7. ((peri‐pancreatic or peripancreatic or pancrea*) adj2 (abscess or fluid)).mp.
8. pancreas abscess/
9. pancreas juice/
10. 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9
11. Clinical trial/
12. Randomized controlled trial/
13. Randomization/
14. Single‐Blind Method/
15. Double‐Blind Method/
16. Cross‐Over Studies/
17. Random Allocation/
18. Placebo/
19. Randomi?ed controlled trial*.tw.
20. Rct.tw.
21. Random allocation.tw.
22. Randomly allocated.tw.
23. Allocated randomly.tw.
24. (allocated adj2 random).tw.
25. Single blind*.tw.
26. Double blind*.tw.
27. ((treble or triple) adj blind*).tw.
28. Placebo*.tw.
29. Prospective study/
30. or/11‐29
31. Case study/
32. Case report.tw.
33. Abstract report/ or letter/
34. or/31‐33
35. 30 not 34
36. 10 and 35
Appendix 5. Science Citation Index search strategy
# 1 TS=((pancrea* near/2 pseudocyst*) OR ((peri‐pancreatic or peripancreatic or pancrea*) near/2 (abscess or fluid)))
# 2 TS=(random* OR rct* OR crossover OR masked OR blind* OR placebo* OR meta‐analysis OR systematic review* OR meta‐analys*)
# 3 #2 AND #1
Appendix 6. ClinicalTrials.gov search strategy
"Interventional" [STUDY‐TYPES] AND pseudocyst [DISEASE] AND ( "Phase 2" OR "Phase 3" OR "Phase 4" ) [PHASE]
Appendix 7. WHO ICTRP search strategy
pseudocyst
Appendix 8. Stata code for network plot
networkplot t1 t2, labels(T1 T2 T3 ...)
Appendix 9. Winbugs code
Source of code:
Consistency models: Dias 2014
Inconsistency models: White 2012 (modifications were performed for continuous, count, and time‐to‐event outcomes)
Binary outcome
Binary outcome ‐ fixed‐effect model
# Binomial likelihood, logit link # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood # model for linear predictor logit(p[i,k]) <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] # expected value of the numerators rhat[i,k] <‐ p[i,k] * n[i,k] #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Binary outcome ‐ random‐effects model
# Binomial likelihood, logit link # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood logit(p[i,k]) <‐ mu[i] + delta[i,k] # model for linear predictor rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Binary outcome ‐ inconsistency model (random‐effects)
# Binomial likelihood, logit link, inconsistency model # Random effects model # Treatment by design interactions # ns = number of studies, nt = number of treatments, A = total number of treatment arms in all trials, and D = the number of designs have to be stated. # The main data are arranged with one record per arm: d and study indicate which design and study that arm belongs to, t indicates its treatment, and b indicates the first treatment in that design. r and n are the numbers of events and individuals in the arm. The supplementary data offset and offset.design list the rows in which the first arm of each trial and of each design is found. model { for(i in 1:ns) { eff.study[i, b[offset[i]], b[offset[i]]] <‐0 for(k in (offset[i] + 1):(offset[i + 1]‐1)) { eff.study[i,t[k],b[k]] <‐eff.des[d[k],t[k]] + RE[i,t[k]] ‐ RE[i,b[k]] } } # Random effects for heterogeneity for(i in 1:ns) { RE[i,1] <‐0 RE[i,2:nt] ˜ dmnorm(zero[], Prec[,]) } # Prec is the inverse of the structured heterogeneity matrix for(i in 1:(nt‐1)) { for(j in 1:(nt‐1)){ Prec[i,j] <‐2*(equals(i,j)‐1/nt)/(tau*tau) } } for(i in 1:A) { logit(p[i]) <‐mu[study[i]] + eff.study[study[i],t[i],b[i]] r[i] ˜ dbin(p[i],n[i])} # For computing DIC for(i in 1:A) { rhat[i] <‐p[i] * n[i] dev[i] <‐2 * (r[i] * (log(r[i])‐log(rhat[i])) + (n[i]‐r[i]) * (log(n[i]‐r[i]) ‐ log(n[i]‐ rhat[i]))) } devs <‐sum(dev[]) # Priors for(i in 1:ns) { mu[i] ˜ dnorm(0,0.01) } tau ˜ dunif(0,2) } # *** PROGRAM ENDS
Continuous outcome (mean difference)
Continuous outcome (mean difference) ‐ fixed‐effect model
# Normal likelihood, identity link # Fixed effect model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions y[i,k] ˜ dnorm(theta[i,k],prec[i,k]) # model for linear predictor theta[i,k] <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] #Deviance contribution dev[i,k] <‐ (y[i,k]‐theta[i,k])*(y[i,k]‐theta[i,k])*prec[i,k] } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for control arm # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # ranking on relative scale for (k in 1:nt) { rk[k] <‐ rank(d[],k) # assumes lower is better # rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (mean difference) ‐ random‐effects model
# Normal likelihood, identity link # Random effects model for multi‐arm trials model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions y[i,k] ˜ dnorm(theta[i,k],prec[i,k]) theta[i,k] <‐ mu[i] + delta[i,k] # model for linear predictor #Deviance contribution dev[i,k] <‐ (y[i,k]‐theta[i,k])*(y[i,k]‐theta[i,k])*prec[i,k] } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific MD distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of MD distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of MD distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for control arm # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # ranking on relative scale for (k in 1:nt) { rk[k] <‐ rank(d[],k) # assumes lower is better # rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (mean difference) ‐ inconsistency model (random‐effects)
# Normal likelihood, identity link, inconsistency model # Random effects model # Treatment by design interactions # ns = number of studies, nt = number of treatments, A = total number of treatment arms in all trials, and D = the number of designs have to be stated. # The main data are arranged with one record per arm: d and study indicate which design and study that arm belongs to, t indicates its treatment, and b indicates the first treatment in that design. y, se, and n are the mean, standard error, and number of individuals in the arm. The supplementary data offset and offset.design list the rows in which the first arm of each trial and of each design is found. model { for(i in 1:ns) { eff.study[i, b[offset[i]], b[offset[i]]] <‐0 for(k in (offset[i] + 1):(offset[i + 1]‐1)) { eff.study[i,t[k],b[k]] <‐eff.des[d[k],t[k]] + RE[i,t[k]] ‐ RE[i,b[k]] } } # Random effects for heterogeneity for(i in 1:ns) { RE[i,1] <‐0 RE[i,2:nt] ˜ dmnorm(zero[], Prec[,]) } # Prec is the inverse of the structured heterogeneity matrix for(i in 1:(nt‐1)) { for(j in 1:(nt‐1)){ Prec[i,j] <‐2*(equals(i,j)‐1/nt)/(tau*tau) } } for(i in 1:A) { var[i] <‐ pow(se[i],2) # calculate variances prec[i] <‐ 1/var[i] # set precisions y[i] ˜ dnorm(theta[i],prec[i]) # normal likelihood theta[i] <‐mu[study[i]] + eff.study[study[i],t[i],b[i]] # model for linear predictor } # For computing DIC for(i in 1:A) { dev[i] <‐ (y[i]‐theta[i])*(y[i]‐theta[i])*prec[i] } devs <‐sum(dev[]) # Priors for(i in 1:ns) { mu[i] ˜ dnorm(0,0.01) } tau ˜ dunif(0,2) for(i in 1:D) { for(k in (offset.design[i] + 1):(offset.design[i] + num.ests[i])) { eff.des[i,t[k]] ˜ dnorm(0,0.01) } } } # *** PROGRAM ENDS
Continuous outcome (standardised mean difference)
The standardised mean difference and its standard error for each treatment comparison will be calculated using the statistical algorithms used by RevMan (RevMan 2014).
Continuous outcome (standardised mean difference) ‐ fixed‐effect model
# Normal likelihood, identity link # Trial‐level data given as treatment differences # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns2) { # LOOP THROUGH 2‐ARM STUDIES y[i,2] ˜ dnorm(delta[i,2],prec[i,2]) # normal likelihood for 2‐arm trials #Deviance contribution for trial i resdev[i] <‐ (y[i,2]‐delta[i,2])*(y[i,2]‐delta[i,2])*prec[i,2] } for(i in (ns2+1):(ns2+ns3)) { # LOOP THROUGH THREE‐ARM STUDIES for (k in 1:(na[i]‐1)) { # set variance‐covariance matrix for (j in 1:(na[i]‐1)) { Sigma[i,j,k] <‐ V[i]*(1‐equals(j,k)) + var[i,k+1]*equals(j,k) } } Omega[i,1:(na[i]‐1),1:(na[i]‐1)] <‐ inverse(Sigma[i,,]) #Precision matrix # multivariate normal likelihood for 3‐arm trials y[i,2:na[i]] ˜ dmnorm(delta[i,2:na[i]],Omega[i,1:(na[i]‐1),1:(na[i]‐1)]) #Deviance contribution for trial i for (k in 1:(na[i]‐1)){ # multiply vector & matrix ydiff[i,k]<‐ y[i,(k+1)] ‐ delta[i,(k+1)] z[i,k]<‐ inprod2(Omega[i,k,1:(na[i]‐1)], ydiff[i,1:(na[i]‐1)]) } resdev[i]<‐ inprod2(ydiff[i,1:(na[i]‐1)], z[i,1:(na[i]‐1)]) } for(i in 1:(ns2+ns3)){ # LOOP THROUGH ALL STUDIES for (k in 2:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions delta[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # ranking on relative scale for (k in 1:nt) { rk[k] <‐ nt+1‐rank(d[],k) # assumes higher HRQoL is “good” #rk[k] <‐ rank(d[],k) # assumes higher outcome is “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (standardised mean difference) ‐ random‐effects model
# Normal likelihood, identity link # Trial‐level data given as treatment differences # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns2) { # LOOP THROUGH 2‐ARM STUDIES y[i,2] ˜ dnorm(delta[i,2],prec[i,2]) # normal likelihood for 2‐arm trials #Deviance contribution for trial i resdev[i] <‐ (y[i,2]‐delta[i,2])*(y[i,2]‐delta[i,2])*prec[i,2] } for(i in (ns2+1):(ns2+ns3)) { # LOOP THROUGH THREE‐ARM STUDIES for (k in 1:(na[i]‐1)) { # set variance‐covariance matrix for (j in 1:(na[i]‐1)) { Sigma[i,j,k] <‐ V[i]*(1‐equals(j,k)) + var[i,k+1]*equals(j,k) } } Omega[i,1:(na[i]‐1),1:(na[i]‐1)] <‐ inverse(Sigma[i,,]) #Precision matrix # multivariate normal likelihood for 3‐arm trials y[i,2:na[i]] ˜ dmnorm(delta[i,2:na[i]],Omega[i,1:(na[i]‐1),1:(na[i]‐1)]) #Deviance contribution for trial i for (k in 1:(na[i]‐1)){ # multiply vector & matrix ydiff[i,k]<‐ y[i,(k+1)] ‐ delta[i,(k+1)] z[i,k]<‐ inprod2(Omega[i,k,1:(na[i]‐1)], ydiff[i,1:(na[i]‐1)]) } resdev[i]<‐ inprod2(ydiff[i,1:(na[i]‐1)], z[i,1:(na[i]‐1)]) } for(i in 1:(ns2+ns3)){ # LOOP THROUGH ALL STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm for (k in 2:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions } for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific SMD distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of random effects distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of random effects distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # ranking on relative scale for (k in 1:nt) { rk[k] <‐ nt+1‐rank(d[],k) # assumes higher HRQoL is “good” # rk[k] <‐ rank(d[],k) # assumes higher outcome is “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (standardised mean difference) ‐ inconsistency model (random‐effects)
# Normal likelihood, identity link # Trial‐level data given as treatment differences # Random effects model model { for(i in 1:ns) { eff.study[i, t[i,1], t[i,1]] <‐0 for(k in 2:na[i]) { eff.study[i,t[i,k],t[i,1]] <‐eff.des[design[k],t[i,k]] + RE[i,t[i,k]] ‐ RE[i, t[i,1]] } } # Random effects for heterogeneity for(i in 1:ns) { RE[i,1] <‐0 RE[i,2:nt] ˜ dmnorm(zero[], Prec[,]) } # Prec is the inverse of the structured heterogeneity matrix for(i in 1:(nt‐1)) { for(j in 1:(nt‐1)){ Prec[i,j] <‐2*(equals(i,j)‐1/nt)/(tau*tau) } } for(i in 1:ns2) { # LOOP THROUGH 2‐ARM STUDIES y[i,2] ˜ dnorm(delta[i,2],prec[i,2]) # normal likelihood for 2‐arm trials #Deviance contribution for trial i resdev[i] <‐ (y[i,2]‐delta[i,2])*(y[i,2]‐delta[i,2])*prec[i,2] } for(i in (ns2+1):(ns2+ns3)) { # LOOP THROUGH THREE‐ARM STUDIES for (k in 1:(na[i]‐1)) { # set variance‐covariance matrix for (j in 1:(na[i]‐1)) { Sigma[i,j,k] <‐ V[i]*(1‐equals(j,k)) + var[i,k+1]*equals(j,k) } } Omega[i,1:(na[i]‐1),1:(na[i]‐1)] <‐ inverse(Sigma[i,,]) #Precision matrix # multivariate normal likelihood for 3‐arm trials y[i,2:na[i]] ˜ dmnorm(delta[i,2:na[i]],Omega[i,1:(na[i]‐1),1:(na[i]‐1)]) #Deviance contribution for trial i for (k in 1:(na[i]‐1)){ # multiply vector & matrix ydiff[i,k]<‐ y[i,(k+1)] ‐ delta[i,(k+1)] + eff.study[i,t[i,k],t[i,1]] z[i,k]<‐ inprod2(Omega[i,k,1:(na[i]‐1)], ydiff[i,1:(na[i]‐1)]) } resdev[i]<‐ inprod2(ydiff[i,1:(na[i]‐1)], z[i,1:(na[i]‐1)]) } for(i in 1:(ns2+ns3)){ # LOOP THROUGH ALL STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm for (k in 2:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions } for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific SMD distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of random effects distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of random effects distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) for(i in 1:D) { for(k in (offset.design[i] + 1):(offset.design[i] + num.ests[i])) { eff.des[i,t[i,k]] ˜ dnorm(0,0.01) } } } } # *** PROGRAM ENDS
Count outcome
Count outcome ‐ fixed‐effect model
# Poisson likelihood, log link # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dpois(theta[i,k]) # Poisson likelihood theta[i,k] <‐ lambda[i,k]*E[i,k] # failure rate * exposure # model for linear predictor log(lambda[i,k]) <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] #Deviance contribution dev[i,k] <‐ 2*((theta[i,k]‐r[i,k]) + r[i,k]*log(r[i,k]/theta[i,k])) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # pairwise RRs and LRRs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { rater[c,k] <‐ exp(d[k] ‐ d[c]) lrater[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Count outcome ‐ random‐effects model
# Poisson likelihood, log link # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dpois(theta[i,k]) # Poisson likelihood theta[i,k] <‐ lambda[i,k]*E[i,k] # failure rate * exposure # model for linear predictor log(lambda[i,k]) <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] #Deviance contribution dev[i,k] <‐ 2*((theta[i,k]‐r[i,k]) + r[i,k]*log(r[i,k]/theta[i,k])) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Count outcome ‐ inconsistency model (random‐effects)
# Poisson likelihood, log link, inconsistency model # Random effects model # Treatment by design interactions # ns = number of studies, nt = number of treatments, A = total number of treatment arms in all trials, and D = the number of designs have to be stated. # The main data are arranged with one record per arm: d and study indicate which design and study that arm belongs to, t indicates its treatment, and b indicates the first treatment in that design. r and E are the numbers of successes and exposures in the arm. The supplementary data offset and offset.design list the rows in which the first arm of each trial and of each design is found. model { for(i in 1:ns) { eff.study[i, b[offset[i]], b[offset[i]]] <‐0 for(k in (offset[i] + 1):(offset[i + 1]‐1)) { eff.study[i,t[k],b[k]] <‐eff.des[d[k],t[k]] + RE[i,t[k]] ‐ RE[i,b[k]] } } # Random effects for heterogeneity for(i in 1:ns) { RE[i,1] <‐0 RE[i,2:nt] ˜ dmnorm(zero[], Prec[,]) } # Prec is the inverse of the structured heterogeneity matrix for(i in 1:(nt‐1)) { for(j in 1:(nt‐1)){ Prec[i,j] <‐2*(equals(i,j)‐1/nt)/(tau*tau) } } for(i in 1:A) { r[i] ˜ dpois(theta[i]) # Poisson likelihood theta[i] <‐ lambda[i]*E[i] # failure rate * exposure log(lambda[i]) <‐mu[study[i]] + eff.study[study[i],t[i],b[i]] # model for linear predictor } # For computing DIC for(i in 1:A) { dev[i] <‐ 2*((theta[i]‐r[i]) + r[i]*log(r[i]/theta[i])) } devs <‐sum(dev[]) # Priors for(i in 1:ns) { mu[i] ˜ dnorm(0,0.01) } tau ˜ dunif(0,2) for(i in 1:D) { for(k in (offset.design[i] + 1):(offset.design[i] + num.ests[i])) { eff.des[i,t[k]] ˜ dnorm(0,0.01) } } } # *** PROGRAM ENDS
Time‐to‐event outcome
Time‐to‐event outcome ‐ fixed‐effect model
# Binomial likelihood, cloglog link # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # Binomial likelihood # model for linear predictor cloglog(p[i,k]) <‐ log(time[i]) + mu[i] + d[t[i,k]] ‐ d[t[i,1]] rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for control arm # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ rank(d[],k) # assumes lower is better rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Time‐to‐event outcome ‐ random‐effects model
# Binomial likelihood, cloglog link # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # Binomial likelihood # model for linear predictor cloglog(p[i,k]) <‐ log(time[i]) + mu[i] + delta[i,k] rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ rank(d[],k) # assumes lower is better rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Time‐to‐event outcome ‐ inconsistency model (random‐effects)
# Binomial likelihood, cloglog link, inconsistency model # Random effects model # Treatment by design interactions # ns = number of studies, nt = number of treatments, A = total number of treatment arms in all trials, and D = the number of designs have to be stated. # The main data are arranged with one record per arm: d and study indicate which design and study that arm belongs to, t indicates its treatment, and b indicates the first treatment in that design. r ,n, and time are the numbers of events, individuals, and follow‐up time in the arm. The supplementary data offset and offset.design list the rows in which the first arm of each trial and of each design is found. model { for(i in 1:ns) { eff.study[i, b[offset[i]], b[offset[i]]] <‐0 for(k in (offset[i] + 1):(offset[i + 1]‐1)) { eff.study[i,t[k],b[k]] <‐eff.des[d[k],t[k]] + RE[i,t[k]] ‐ RE[i,b[k]] } } # Random effects for heterogeneity for(i in 1:ns) { RE[i,1] <‐0 RE[i,2:nt] ˜ dmnorm(zero[], Prec[,]) } # Prec is the inverse of the structured heterogeneity matrix for(i in 1:(nt‐1)) { for(j in 1:(nt‐1)){ Prec[i,j] <‐2*(equals(i,j)‐1/nt)/(tau*tau) } } for(i in 1:A) { r[i] ˜ dbin(p[i],n[i]) # Binomial likelihood cloglog(p[i]) <‐ log(time[i]) + mu[study[i]] + eff.study[study[i],t[i],b[i]] # model for linear predictor } # For computing DIC for(i in 1:A) { dev[i] <‐ 2 * (r[i] * (log(r[i])‐log(rhat[i]))+ (n[i]‐r[i]) * (log(n[i]‐r[i]) ‐ log(n[i]‐rhat[i]))) } devs <‐sum(dev[]) # Priors for(i in 1:ns) { mu[i] ˜ dnorm(0,0.01) } tau ˜ dunif(0,2) for(i in 1:D) { for(k in (offset.design[i] + 1):(offset.design[i] + num.ests[i])) { eff.des[i,t[k]] ˜ dnorm(0,0.01) } } } # *** PROGRAM ENDS
Appendix 10. Technical details of network meta‐analysis
The posterior probabilities (effect estimates or values) of the treatment contrast (i.e., log odds ratio, mean difference, standardised mean difference, rate ratio, or hazard ratio) may vary depending upon the initial values to start the simulations. In order to control the random error due to the choice of initial values, we performed the network analysis for three different initial values (starting values) as per the guidance from The National Institute for Health and Care Excellence (NICE) Decision Support Unit (DSU) documents (Dias 2013). If the results from three different initial values were similar and stable (convergence), then the results are reliable. It is important to discard the results of the initial simulations, as they can be significantly affected by the choice of the initial values, and only include the results of the simulations obtained after the convergence. The discarding of the initial simulations is called 'burn in'. We had planned to run the models for all outcomes for 30,000 simulations for 'burn in' for three different chains (a set of initial values). We had planned to run the models for another 100,000 simulations to obtain the effect estimates. We had planned to obtain the effect estimates from the results of all the three chains (different initial values). We had planned to ensure that the results in the three different chains were similar in order to control for random error due to the choice of priors. We had planned to do this in addition to the visual inspection of convergence obtained after simulations in the 'burn in'.
We had planned to run three different models for each outcome. A fixed‐effect model assumes that the treatment effect is the same across studies. The random‐effects consistency model assumes that the treatment effect is distributed normally across the studies, but assumes that the transitivity assumption is satisfied (i.e., the population studied, the definition of outcomes, and the methods used were similar across studies and that there is consistency between the direct comparison and indirect comparison). A random‐effects inconsistency model does not make the transitivity assumption. If the inconsistency model resulted in a better model fit than the consistency model, the results of the network meta‐analysis could be unreliable and so should be interpreted with extreme caution. If there was evidence of inconsistency, we had planned to identify areas in the network where substantial inconsistency might be present in terms of clinical and methodological diversities between trials, and when appropriate, we had planned to limit network meta‐analysis to a more compatible subset of trials.
We had planned to base the choice of the model between fixed‐effect and random‐effects on the model fit as per the guidelines of the NICE TSU (Dias 2013). The model fit was assessed by deviance residuals and Deviance Information Criteria (DIC) according to NICE TSU guidelines (Dias 2013). A difference of three or five in the DIC is not generally considered important (Dias 2012c). We had planned to use the simpler model, i.e., the fixed‐effect model, if the DIC were similar between the fixed‐effect and the random‐effects models. We had planned to use the random‐effects model if it resulted in a better model fit, as indicated by a DIC that was lower than the fixed‐effect model by at least three.
We had planned to calculate the effect estimates of the treatment and the 95% credible intervals using the following additional code: # pairwise ORs and MD for all possible pairwise comparisons, if nt > 2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { OR[c,k] <‐ exp(d[k] ‐ d[c]) #MD[c,k] <‐ (d[k]‐d[c]) } }
where c indicates control group, k indicates intervention group, OR indicates odds ratio or other ratios, and MD indicates mean difference or other differences.
Appendix 11. Winbugs code for subgroup analysis
Source of code: Dias 2012c
Categorical covariate
Only the code for random‐effects model for a binary outcome is shown. The differences in the code are underlined. We had planned to make similar changes for other outcomes.
# Binomial likelihood, logit link, subgroup # Random effects model for multi‐arm trials model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood # model for linear predictor, covariate effect relative to treat in arm 1 logit(p[i,k]) <‐ mu[i] + delta[i,k] + (beta[t[i,k]]‐beta[t[i,1]]) * x[i] rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment beta[1] <‐ 0 # covariate effect is zero for reference treatment for (k in 2:nt){ # LOOP THROUGH TREATMENTS d[k] ˜ dnorm(0,.0001) # vague priors for treatment effects beta[k] <‐ B[k] # exchangeable covariate effect B[k] ˜ dnorm(0,.0001) # vague prior for covariate effect } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # treatment effect when covariate = z[j] for (k in 1:nt){ # LOOP THROUGH TREATMENTS for (j in 1:nz) { dz[j,k] <‐ d[k] + (beta[k]‐beta[1])*z[j] } } # *** PROGRAM ENDS
Continuous covariate
# Binomial likelihood, logit link, continuous covariate # Random effects model for multi‐arm trials model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood # model for linear predictor, covariate effect relative to treat in arm 1 logit(p[i,k]) <‐ mu[i] + delta[i,k] + (beta[t[i,k]]‐beta[t[i,1]]) * (x[i]‐mx) rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment beta[1] <‐ 0 # covariate effect is zero for reference treatment for (k in 2:nt){ # LOOP THROUGH TREATMENTS d[k] ˜ dnorm(0,.0001) # vague priors for treatment effects beta[k] <‐ B[k] # exchangeable covariate effect B[k] ˜ dnorm(0,.0001) # vague prior for covariate effect } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # treatment effect when covariate = z[j] (un‐centring treatment effects) for (k in 1:nt){ for (j in 1:nz) { dz[j,k] <‐ d[k] ‐ (beta[k]‐beta[1])*(mx‐z[j]) } } # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { # at mean value of covariate or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) # at covariate=z[j] for (j in 1:nz) { orz[j,c,k] <‐ exp(dz[j,k] ‐ dz[j,c]) lorz[j,c,k] <‐ (dz[j,k]‐dz[j,c]) } } } } # *** PROGRAM ENDS
Data and analyses
Comparison 1. Management strategies for pancreatic pseudocysts.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term mortality | 4 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Endoscopic drainage versus EUS‐guided drainage | 2 | 89 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.11, 79.91] |
| 1.2 EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | 1 | 47 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.3 Open surgical drainage versus EUS‐guided drainage | 1 | 40 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Serious adverse events | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 Endoscopic drainage versus EUS‐guided drainage | 2 | 89 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.42 [0.51, 11.46] |
| 2.2 Open surgical drainage versus EUS‐guided drainage | 1 | 40 | Odds Ratio (M‐H, Fixed, 95% CI) | 8.2 [0.40, 169.90] |
| 3 Health‐related quality of life (4 weeks to 3 months) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Open surgical drainage versus EUS‐guided drainage | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Adverse events | 4 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.1 Endoscopic drainage versus EUS‐guided drainage | 2 | 89 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.42 [0.51, 11.46] |
| 4.2 EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | 1 | 47 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.20 [0.06, 0.73] |
| 4.3 Open surgical drainage versus EUS‐guided drainage | 1 | 40 | Odds Ratio (M‐H, Fixed, 95% CI) | 11.18 [0.56, 222.98] |
| 5 Hospital stay | 3 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Endoscopic drainage versus EUS‐guided drainage | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.2 EUS‐guided drainage with nasocystic drainage versus EUS‐guided drainage | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.3 Open surgical drainage versus EUS‐guided drainage | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Need for additional drainage | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 6.1 Endoscopic drainage versus EUS‐guided drainage | 2 | 89 | Odds Ratio (M‐H, Fixed, 95% CI) | 11.13 [2.85, 43.44] |
| 6.2 Open surgical drainage versus EUS‐guided drainage | 1 | 40 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.47 [0.04, 5.69] |
| 7 Costs | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 7.1 Open surgical drainage versus EUS‐guided drainage | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Park 2009.
| Methods | Randomised controlled trial | |
| Participants | Country: South Korea.
Number randomised: 60.
Post‐randomisation drop‐outs: not stated.
Revised sample size: 60.
Average age: 48 years.
Females: 13 (21.7%).
Symptomatic patients: 60 (100%).
Acute pancreatitis: 27 (45%).
Chronic pancreatitis: 28 (46.7%).
Infected pseudocyst: 0 (0%).
Mean size of pseudocyst in mm: 78
Follow‐up time: minimum 6 months
Concomitant interventions: not stated Inclusion criteria 1. History of pancreatitis. 2. Symptomatic pseudocysts with duration of longer than 4 weeks. Exclusion criteria 1. Age less than 18 years. 2. A pancreatic pseudocyst more than 4 cm in size and communicating with the pancreatic duct. 3. A suggested pancreatic abscess or necrosis, shown by computed tomography (CT). 4. A moderate thickness (> 10 mm) from the cyst wall to the intramural wall, shown by EUS. 5. Portal hypertension or coagulopathy. |
|
| Interventions | Participants were randomly assigned to two groups. Group 1: Endoscopic EUS‐guided drainage (N = 31). Group 2: Endoscopic drainage (N = 29). | |
| Outcomes | The outcomes reported were short‐term mortality, adverse events, and requirement for additional procedures. | |
| Notes | Source of funding: not stated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "...randomly assigned by means of computer‐generated numbers to undergo….". |
| Allocation concealment (selection bias) | Unclear risk | Comment: This information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "... because this study was non‐blinded, the inherent possibility of bias may exist. However, blinding of an investigator to any technique to which patients are randomly assigned is impractical". |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "... because this study was non‐blinded, the inherent possibility of bias may exist. However, blinding of an investigator to any technique to which patients are randomly assigned is impractical". |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: There were no post‐randomisation drop‐outs. |
| Selective reporting (reporting bias) | Low risk | Comment: All important outcomes were reported. |
| Other bias | Low risk | Comment: No other source of bias. |
Varadarajulu 2008.
| Methods | Randomised controlled trial | |
| Participants | Country: USA.
Number randomised: 30.
Post‐randomisation drop‐outs: 1 (3.3%).
Revised sample size: 29.
Average age: 47 years.
Females: 11 (37.9%).
Symptomatic patients: 29 (100%).
Acute pancreatitis: not stated
Chronic pancreatitis: not stated
Infected pseudocyst: 0 (0%).
Mean size of pseudocyst in mm: 70
Follow‐up time: minimum 6 weeks
Concomitant interventions: not stated Inclusion criteria 1. History of pancreatitis. 2. Symptomatic pseudocysts. Exclusion criteria 1. CT findings were suggestive of pathology other than a pseudocyst. 2. Pseudocyst was ≤ 4 cm in size. 3. Patients younger than 18 years of age. 4. Patients with pancreatic abscess or necrosis by CT. |
|
| Interventions | Participants were randomly assigned to two groups. Group 1: Endoscopic EUS‐guided drainage (N = 14). Group 2: Endoscopic drainage (N = 15). | |
| Outcomes | The outcomes reported were short‐term mortality, adverse events, length of hospital stay, and requirement for additional procedures. | |
| Notes | Reasons for post‐randomisation drop‐outs: had septae on EUS and so the diagnosis was revised to pancreatic adenoma rather than pseudocyst, and patient underwent surgery. Source of funding: not stated. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "After ERCP, an endoscopy nurse opened a sealed envelope that contained computer‐generated randomization assignments". |
| Allocation concealment (selection bias) | Low risk | Quote: "After ERCP, an endoscopy nurse opened a sealed envelope that contained computer‐generated randomization assignments". |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "Furthermore, the blinding of an investigator to the technique to which the patient was randomized is neither feasible nor practical, and therefore, it is difficult to exclude the possibility of bias completely". Comment: It is impossible to blind the healthcare providers. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: This information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: There was one post‐randomisation drop‐out in the EUS‐guided drainage group since septae were noted and the diagnosis was revised to pancreatic adenoma. This is an inherent advantage of EUS‐guided drainage and exclusion of the patient may have underestimated the overall benefit of EUS‐guided drainage. However, this is unlikely to influence any of the reported outcomes. |
| Selective reporting (reporting bias) | Low risk | Comment: All important outcomes were reported. |
| Other bias | Low risk | Comment: No other source of bias. |
Varadarajulu 2013.
| Methods | Randomised controlled trial | |
| Participants | Country: USA.
Number randomised: 40.
Post‐randomisation drop‐outs: not stated.
Revised sample size: 40.
Average age: 50 years.
Females: 12 (30%).
Symptomatic patients: 40 (100%).
Acute pancreatitis: 13 (32.5%).
Chronic pancreatitis: 27 (67.5%).
Infected pseudocyst: not stated
Mean size of pseudocyst in mm: 108
Follow‐up time: minimum 24 months
Concomitant interventions: not stated Inclusion criteria 1. Diagnosis of pancreatic pseudocyst based on CT criteria. 2. Pseudocyst measuring ≥ 6 cm in size and located adjacent to the stomach. 3. Documented history of acute or chronic pancreatitis. 4. Persistent pancreatic pain requiring narcotics or analgesics. 5. Symptomatic gastric outlet or bile duct obstruction induced by the pseudocyst. Exclusion criteria 1. Age < 18 or > 80 years. 2. Contraindications to surgery: ASA class IV, severe portal hypertension. 3. Contraindication to endoscopic drainage: gastrectomy with Billroth II reconstruction, gastric bypass surgery, prior surgery for pancreas‐related complications. 4. Pregnancy. 5. Associated pancreatic necrosis on CT. 6. Pseudocyst not adjacent to the stomach. 7. Multiloculated pseudocyst or multiple pseudocysts. |
|
| Interventions | Participants were randomly assigned to two groups. Group 1: Endoscopic EUS‐guided drainage (N = 20). Group 2: Open surgical drainage (N = 20). | |
| Outcomes | The outcomes reported were short‐term mortality, adverse events, length of hospital stay, and requirement for additional procedures. | |
| Notes | Authors provided information on source of funding and length of hospital stay. Source of funding: no funding (author reply). |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Computer‐generated randomization assignments were provided by the statistician using a block randomization method". |
| Allocation concealment (selection bias) | Low risk | Quote: "The randomization assignments were placed in sequentially numbered, sealed, opaque envelopes and were opened by one of the study investigators to determine treatment allocation". |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "We conducted a single‐center, open‐label, randomized trial to compare endoscopic and surgical drainage….". |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "We conducted a single‐center, open‐label, randomized trial to compare endoscopic and surgical drainage….". |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: There were no post‐randomisation drop‐outs. |
| Selective reporting (reporting bias) | Low risk | Comment: All important outcomes were reported. |
| Other bias | Low risk | Comment: No other source of bias. |
Yuan 2015.
| Methods | Randomised controlled trial | |
| Participants | Country: China.
Number randomised: 47.
Post‐randomisation drop‐outs: not stated.
Revised sample size: 47.
Average age: 48 years.
Females: 18 (38.3%).
Symptomatic patients: not stated
Acute pancreatitis: not stated
Chronic pancreatitis: not stated
Infected pseudocyst: not stated
Mean size of pseudocyst in mm: 155
Follow‐up time: minimum 24 months
Concomitant interventions: not stated Inclusion criteria 1. Ages 16 to 70 years. 2. Clinical presentation with abdominal distension with or without upper gastrointestinal obstruction. 3. Radiological findings suggestive of a pancreatic pseudocyst > 10 cm. Exclusion criteria 1. Cysts without adherence to the gastric wall. 2. Distance between pseudocyst and gastric wall > 1 cm. 3. Pseudocyst communication with the main pancreatic duct. 4. Pregnancy. 5. Pancreatic tumours. 6. Risk for anaesthesia and surgery. 7. Contraindications to magnetic resonance cholangiopancreatography and endoscopic retrograde cholangiopancreatography. 8. Liver cirrhosis. 9. Previous history of abdominal surgery (e.g., gastrectomy). 10. Inability to give informed consent. |
|
| Interventions | Participants were randomly assigned to two groups. Group 1: Endoscopic EUS‐guided drainage (N = 23). Group 2: Endoscopic EUS‐guided drainage with nasocystic drainage (N = 24). | |
| Outcomes | The outcomes reported were short‐term mortality, recurrence, and length of hospital stay. | |
| Notes | Source of funding: not stated. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: This information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "Patients were randomized using sealed envelopes to undergo single‐step or 2‐step treatment". |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | This information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: This information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: This information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: Morbidity was not reported adequately. |
| Other bias | Low risk | Comment: No other source of bias. |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Sauer 2010 | Comment on an included study (Varadarajulu 2008) |
Characteristics of ongoing studies [ordered by study ID]
NCT02041793.
| Trial name or title | A Prospective Randomized Controlled Trial Comparing Laparoscopic Versus Endoscopic Drainage for Pseudocyst of the Pancreas |
| Methods | Randomised controlled trial |
| Participants | Country: India
Estimated enrolment:60 Inclusion criteria Symptomatic patients with pseudocyst of size more than 6 cm, more than 6‐8 weeks duration after an attack of acute pancreatitis. Exclusion criteria
|
| Interventions | Participants randomly assigned to two groups. Group 1: Laparoscopic cystogastrostomy Group 2: Endoscopic cystogastrostomy |
| Outcomes |
Primary outcome Resolution of pseudocyst. Secondary outcomes Adverse events (bleeding, sepsis, chest complications and other important events in the post procedure period requiring prolonged stay and/or repeat procedure) Cost per patient. Recurrence rate of pseudocyst. |
| Starting date | August 2011 |
| Contact information | Pramod K Garg, MD (pgarg10@hotmail.com) |
| Notes |
Differences between protocol and review
We included endoscopic‐guided drainage (EUS‐guided drainage) with nasocystic drainage as an additional intervention. Generally, a nasocystic catheter is used during EUS‐guided drainage to irrigate the cyst, if there is a suspicion of presence of debris. The nasocystic catheter is then generally removed, only retaining the stent between the stomach or duodenum and the pseudocyst. Yuan 2015 used routine nasocystic drainage. This was considered to be a significant variation from standard EUS‐guided drainage, so, it was included as an intervention.
While network meta‐analysis has its advantages in combining direct and indirect evidence (resulting in more precise evidence) and Bayesian network meta‐analysis allows the calculation of a probability of the best treatments, these advantages were limited in this review because of the sparse data, with zero event trials and lack of direct and indirect evidence for any comparisons. So, we used Frequentist methods, which allowed us to present information in the standard Cochrane format for direct comparisons; we also presented indirect comparisons when able.
We had planned to use conservative treatment as the reference group. However, none of the trials included conservative treatment as one of the arms. We plan to use EUS‐guided drainage as the reference group in future updates, since this was the intervention that was included in all the trials.
Contributions of authors
Conceiving the review: KG
Designing the review: KG
Co‐ordinating the review: KG
Designing search strategies: KG
Study selection: KG, EP
Data extraction: KG, EP
Writing the review: KG
Providing statistical advice on the review: NH
Providing general advice on the review: BRD, SP
Securing funding for the review: KG
Performing previous work that was the foundation of the current study: KG
Sources of support
Internal sources
-
University College London, UK.
Salary, equipment, and other resources to complete this review.
External sources
-
National Institute for Health Research, UK.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure, Cochrane Programme Grant or Cochrane Incentive funding to the Cochrane Hepato‐Biliary and Upper Gastrointestinal and Pancreatic Diseases Groups. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health. The funding covered salary, equipment, and other resources to complete this review. The review was also supported by the Complex Reviews Support Unit, also funded by the National Institute for Health Research (project number 14/178/29).
Declarations of interest
This report is independent research funded by the National Institute for Health Research (NIHR Cochrane Programme Grants, 13/89/03 ‐ Evidence‐based diagnosis and management of upper digestive, hepato‐biliary, and pancreatic disorders). The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health.
Edited (no change to conclusions)
References
References to studies included in this review
Park 2009 {published data only}
- Moon S‐H, Lee SS, Park DH, Choi SY, Kim JY, Jung SW, et al. Comparison of EUS‐guided one‐step transmural drainage of pancreatic pseudocysts and conventional transmural drainage: A prospective, non‐blinded, single center, randomized study. Gastrointestinal Endoscopy 2008;67(5):AB225. [Google Scholar]
- Park DH, Lee SS, Moon SH, Choi SY, Jung SW, Seo DW, et al. Endoscopic ultrasound‐guided versus conventional transmural drainage for pancreatic pseudocysts: A prospective randomized trial. Endoscopy 2009;41(10):842‐8. [DOI] [PubMed] [Google Scholar]
- Park DH, Lee SS, Moon SH, Choi SY, Jung SW, Seo DW, et al. Endoscopic ultrasound‐guided versus conventional transmural drainage for pancreatic pseudocysts: A prospective randomized trial. Endoskopie Heute 2009;22(4):243‐50. [DOI] [PubMed] [Google Scholar]
Varadarajulu 2008 {published data only}
- Varadarajulu S. Prospective randomized trial comparing EUS and EGD for transmural drainage of pancreatic pseudocysts: A need for a large randomized study response. Gastrointestinal Endoscopy 2010;71(2):432‐3. [DOI] [PubMed] [Google Scholar]
- Varadarajulu S, Christein JD, Tambane A, Drelichman ER, Wilcox CM. Prospective randomized trial comparing EUS and EGD for transmural drainage of pancreatic pseudocysts (with videos). Gastrointestinal Endoscopy 2008;68(6):1102‐11. [DOI] [PubMed] [Google Scholar]
- Varadarajulu S, Christein JD, Tamhane A, Drelichman ER, Wilcox CM. Prospective randomized trial comparing endoscopic ultrasound (EUS) and gastroscopy (EGD) for trans‐mural drainage of pancreatic pseudocysts. Gastrointestinal Endoscopy 2008;67(5):AB110. [DOI] [PubMed] [Google Scholar]
- Varadarajulu S, Christein JD, Tamhane A, Drelichman ER, Wilcox M. Prospective randomized trial comparing endoscopic ultrasound (EUS) and gastroscopy (EGD) for trans‐mural drainage of pancreatic pseudocysts. Gastrointestinal Endoscopy 2009;69(2):S228. [DOI] [PubMed] [Google Scholar]
Varadarajulu 2013 {published data only}
- Varadarajulu S, Bang JY, Sutton BS, Trevino JM, Christein JD, Wilcox CM. Equal efficacy of endoscopic and surgical cystogastrostomy for pancreatic pseudocyst drainage in a randomized trial. Gastroenterology 2013;145(3):583‐90.e1. [DOI] [PubMed] [Google Scholar]
- Varadarajulu S, Trevino J, Wilcox CM, Sutton B, Christein JD. Randomized trial comparing EUS and surgery for pancreatic pseudocyst drainage. Gastrointestinal Endoscopy 2010;71(5):AB116. [DOI] [PubMed] [Google Scholar]
Yuan 2015 {published data only}
- Yuan H, Qin M, Liu R, Hu S. Single‐step versus 2‐step management of huge pancreatic pseudocysts: A prospective randomized trial with long‐term follow‐up. Pancreas 2015;44(4):570‐3. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Sauer 2010 {published data only}
- Sauer B, Kahaleh M. Prospective randomized trial comparing EUS and EGD for transmural drainage of pancreatic pseudocysts: A need for a large randomized study. Gastrointestinal Endoscopy 2010;71(2):432. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
NCT02041793 {unpublished data only}
- CTRI/2012/09/002969. Laparoscopic cystogastrostomy versus endoscopic cystogastrostomy. http://www.ctri.nic.in/Clinicaltrials/pmaindet2.php?trialid=5181 (accessed 3 January 2016).
- NCT02041793. Laparoscopic cystogastrostomy versus endoscopic cystogastrostomy. https://clinicaltrials.gov/show/NCT02041793 (accessed 3 January 2016).
Additional references
Banks 2013
- Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, et al. Classification of acute pancreatitis‐‐2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013;62(1):102‐11. [DOI] [PubMed] [Google Scholar]
Bucher 1997
- Bucher HC, Guyatt GH, Griffith LE, Walter SD. The results of direct and indirect treatment comparisons in meta‐analysis of randomized controlled trials. Journal of Clinical Epidemiology 1997;50(6):683‐91. [DOI] [PubMed] [Google Scholar]
Cannon 2009
- Cannon JW, Callery MP, Vollmer CM Jr. Diagnosis and management of pancreatic pseudocysts: what is the evidence?. Journal of the American College of Surgeons 2009;209(3):385‐93. [DOI] [PubMed] [Google Scholar]
Cheruvu 2003
- Cheruvu CV, Clarke MG, Prentice M, Eyre‐Brook IA. Conservative treatment as an option in the management of pancreatic pseudocyst. Annals of the Royal College of Surgeons of England 2003;85(5):313‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Clavien 2009
- Clavien PA, Barkun J, Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien‐Dindo classification of surgical complications: five‐year experience. Annals of Surgery 2009;250(2):187‐96. [DOI] [PubMed] [Google Scholar]
Dias 2012a
- Dias S, Welton NJ, Sutton AJ, Ades AE. NICE DSU Technical Support Document 1: Introduction to evidence synthesis for decision making, 2012. www.nicedsu.org.uk/TSD1%20Introduction.final.08.05.12.pdf (accessed 27 March 2014).
Dias 2012b
- Dias S, Welton NJ, Sutton AJ, Caldwell DM, Lu G, Ades AE. NICE DSU Technical Support Document 4: Inconsistency in networks of evidence based on randomised controlled trials, 2012. www.nicedsu.org.uk/TSD4%20Inconsistency.final.08.05.12.pdf (accessed 27 March 2014). [PubMed]
Dias 2012c
- Dias S, Sutton AJ, Welton NJ, Ades AE. NICE DSU Technical Support Document 3: Heterogeneity: subgroups, meta‐regression, bias and bias‐adjustment, 2012. www.nicedsu.org.uk/TSD3%20Heterogeneity.final%20report.08.05.12.pdf (accessed 27 March 2014).
Dias 2013
- Dias S, Welton NJ, Sutton AJ, Ades AE. NICE DSU technical support document 2: a generalised linear modelling framework for pairwise and network meta‐analysis of randomised controlled trials, 2013. www.nicedsu.org.uk/TSD2%20General%20meta%20analysis%20corrected%20Mar2013.pdf (accessed 27 March 2014). [PubMed]
Dindo 2004
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Annals of Surgery 2004;240(2):205‐13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Egger 1997
- Egger M, Davey SG, Schneider M, Minder C. Bias in meta‐analysis detected by a simple, graphical test. BMJ (Clinical Research Ed.) 1997;315(7109):629‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
FDA 2006
- Center for Biologics Evaluation and Research, U.S. Food, Drug Administration. Guidance for industry adverse reactions section of labeling for human prescription drug and biological products — Content and format. www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm075057.pdf (accessed 4th July 2014).
Gurusamy 2014
- Gurusamy KS, Pallari E, Hawkins N, Pereira SP, Davidson BR. Management strategies for pancreatic pseudocysts: a network meta‐analysis. Cochrane Database of Systematic Reviews 2014, Issue 11. [DOI: 10.1002/14651858.CD011392] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327(7414):557‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Higgins 2012
- Higgins JPT, Jackson D, Barrett JK, Lu G, Ades AE, White IR. Consistency and inconsistency in network meta‐analysis: concepts and models for multi‐arm studies. Research Synthesis Methods 2012;3(2):98‐110. [DOI] [PMC free article] [PubMed] [Google Scholar]
Huggett 2015
- Huggett MT, Oppong KW, Pereira SP, Keane MG, Mitra V, Charnley RM, et al. Endoscopic drainage of walled‐off pancreatic necrosis using a novel self‐expanding metal stent. Endoscopy 2015;47(10):929‐32. [DOI] [PubMed] [Google Scholar]
ICH‐GCP 1996
- International Conference on Harmonisation of Technical Requiements for Registration of Pharmaceuticals for Human Use. Code of Federal Regulation & ICH Guidelines. Media: Parexel Barnett, 1996. [Google Scholar]
Johnson 2009
- Johnson MD, Walsh RM, Henderson JM, Brown N, Ponsky J, Dumot J, et al. Surgical versus nonsurgical management of pancreatic pseudocysts. Journal of Clinical Gastroenterology 2009;43(6):586‐90. [DOI] [PubMed] [Google Scholar]
Lu 2004
- Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Statistics in Medicine 2004;23(20):3105‐24. [DOI] [PubMed] [Google Scholar]
Lu 2007
- Lu G, Ades AE, Sutton AJ, Cooper NJ, Briggs AH, Caldwell DM. Meta‐analysis of mixed treatment comparisons at multiple follow‐up times. Statistics in Medicine 2007;26(20):3681‐99. [DOI] [PubMed] [Google Scholar]
MHRA 2013
- Medicines and Healthcare products Regulatory Agency (MHRA). Clinical trials for medicines: Safety reporting ‐ SUSARs and DSURs. www.mhra.gov.uk/Howweregulate/Medicines/Licensingofmedicines/Clinicaltrials/Safetyreporting‐SUSARsandASRs/ (accessed 4 July 2014).
Mills 2012
- Mills EJ, Ioannidis JP, Thorlund K, Schunemann HJ, Puhan MA, Guyatt GH. How to use an article reporting a multiple treatment comparison meta‐analysis. JAMA 2012;308(12):1246‐53. [DOI] [PubMed] [Google Scholar]
NCBI 2014
- NCBI. MeSH. NLM Controlled Vocabulary. Pancreas. www.ncbi.nlm.nih.gov/mesh/68010179 (accessed 4 July 2014).
Newell 1992
- Newell DJ. Intention‐to‐treat analysis: implications for quantitative and qualitative research. International Journal of Epidemiology 1992;21(5):837‐41. [DOI] [PubMed] [Google Scholar]
Puhan 2014
- Puhan MA, Schunemann HJ, Murad MH, Li T, Brignardello‐Petersen R, Singh JA, et al. A GRADE Working Group approach for rating the quality of treatment effect estimates from network meta‐analysis. BMJ (Clinical research edition) 2014;349:g5630. [DOI] [PubMed] [Google Scholar]
RevMan 2014 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan) [Computer program]. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Salanti 2011
- Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple‐treatment meta‐analysis: an overview and tutorial. Journal of Clinical Epidemiology 2011;64(2):163‐71. [DOI] [PubMed] [Google Scholar]
Vitas 1992
- Vitas GJ, Sarr MG. Selected management of pancreatic pseudocysts: operative versus expectant management. Surgery 1992;111(2):123‐30. [PubMed] [Google Scholar]
White 2012
- White IR, Barrett JK, Jackson D, Higgins JPT. Consistency and inconsistency in network meta‐analysis: model estimation using multivariate meta‐regression. Research Synthesis Methods 2012;3(2):111‐25. [DOI] [PMC free article] [PubMed] [Google Scholar]
WinBUGS 1.4 [Computer program]
- Imperial College and MRC, UK. WinBUGS with DoodleBUGS. Version 1.4.3. Imperial College and MRC, UK, 2007.
Zhao 2016
- Zhao X, Feng T, Ji W. Endoscopic versus surgical treatment for pancreatic pseudocyst. Digestive endoscopy 2016;28(1):83‐91. [DOI] [PubMed] [Google Scholar]


