Abstract
Background
It is generally assumed by practitioners and guideline authors that combined modalities (methods of treatment) are more effective than single modalities in preventing venous thromboembolism (VTE), defined as deep vein thrombosis (DVT) or pulmonary embolism (PE), or both. This is an update of the review first published in 2008.
Objectives
The aim of this review was to assess the efficacy of combined intermittent pneumatic leg compression (IPC) and pharmacological prophylaxis versus single modalities in preventing venous thromboembolism.
Search methods
For this update the Cochrane Vascular Information Specialist (CIS) searched the Specialised Register (May 2016). In addition the CIS searched the Cochrane Register of Studies (CENTRAL (2016, Issue 4)). Clinical trials databases were searched for details of ongoing or unpublished studies.
Selection criteria
Randomized controlled trials (RCTs) or controlled clinical trials (CCTs) of combined IPC and pharmacological interventions used to prevent VTE.
Data collection and analysis
We independently selected trials and extracted data. Disagreements were resolved by discussion. We performed fixed‐effect model meta‐analyses with odds ratios (ORs) and 95% confidence intervals (CIs). We used a random‐effects model when there was heterogeneity.
Main results
We included a total of 22 trials (9137 participants) of which 15 were randomized trials (7762 participants). The overall risk of bias was mostly unclear or high due to selection and performance bias. We used GRADE to assess the quality of the evidence and this was downgraded from high to moderate or very low due to the risk of bias, imprecision or indirectness.
The rate of PE in the studies comparing IPC alone with combined IPC and pharmacological prophylaxis was low, underpowering the analyses. The incidence of symptomatic PE was 0.79% with IPC, but ranged between 0.1 to 1% with combined IPC and pharmacological prophylaxis (OR 0.49, 95% CI 0.18 to 1.34; 12 studies, 3017 participants, moderate quality evidence). The incidence of DVT was 4.10% in the IPC group and 2.19% in the combined group showing a reduced incidence of DVT in favour of the combined group (OR 0.52, 95% CI 0.33 to 0.82; 11 studies, 2934 participants, moderate quality evidence). The addition of an anticoagulant to IPC, however, increased the risk of any bleeding compared to IPC alone; 0.66% (7/1053) in the IPC group and 4.0% (44/1102) in the combined group (OR 5.04, 95% CI 2.36 to 10.77; 7 studies, 2155 participants, moderate quality evidence). Major bleeding followed a similar pattern; 0.1% (1/1053) in the IPC group to 1.5% (17/1102) in the combined group (OR 6.81, 95% CI 1.99 to 23.28; 7 studies, 2155 participants, moderate quality evidence).
We detected no difference between the type of surgery subgroups such as orthopedic and non‐orthopedic participants for DVT incidence (P = 0.16). Tests for differences between type of surgery subgroups were not possible for PE incidence.
Compared with pharmacological prophylaxis alone, the use of combined IPC and pharmacological prophylaxis modalities reduced the incidence of symptomatic PE from 2.92% to 1.20% (OR 0.39, 95% CI 0.23 to 0.64; 10 studies, 3544 participants, moderate quality evidence). The incidence of DVT was 6.2% in the pharmacological prophylaxis group and 2.9% in the combined group showing no difference between the combined and pharmacological prophylaxis groups (OR 0.42, 95% CI 0.18 to 1.03; 11 studies, 2866 participants, moderate quality evidence). Increased bleeding side effects were not observed for IPC when it was added to anticoagulation (bleeding: OR 0.80, 95% CI 0.30 to 2.14, very low quality evidence; major bleeding: OR 1.21, 95% CI 0.35 to 4.18, very low quality evidence, 3 studies, 244 participants).
No difference was detected between the type of surgery subgroups for PE incidence (P = 0.68) or for DVT incidence (P = 0.10).
Authors' conclusions
Moderate quality evidence suggests that combining IPC and pharmacological prophylaxis, compared with IPC or pharmacological prophylaxis alone, decreases the incidence of DVT when compared to compression, and incidence of PE when compared to anticoagulation. Moderate quality evidence suggests that there is no difference between combined and single modalities in the incidence of PE when compared with compression alone and DVT when compared with anticoagulation alone. The quality of evidence for PE or DVT was downgraded to moderate due to imprecision or risk of bias in study methodology, highlighting the need for further research. Moderate quality evidence suggests the addition of pharmacological prophylaxis to IPC, increased the risk of bleeding compared to IPC alone, a side effect not observed for IPC when added to pharmacological prophylaxis (very low quality evidence), as expected for a physical method of thromboprophylaxis. The quality of evidence for bleeding was downgraded to moderate due to indirectness or very low due to risk of bias in study methodology, indirectness and imprecision highlighting the need for further research. Nevertheless, the results of the current review agree with current guideline recommendations, which support the use of combined modalities in hospitalised patients (limited to those with trauma or undergoing surgery) at risk of developing VTE. More studies on the role of combined modalities in VTE prevention are needed.
Plain language summary
Combined intermittent pneumatic leg compression and medication for the prevention of deep vein thrombosis and pulmonary embolism
Background
Deep vein thrombosis (DVT) and pulmonary embolism (PE) are collectively known as venous thromboembolism (VTE), and occur when a blood clot develops inside the leg veins (DVT) and travels to the lungs (PE). They are possible complications of hospitalisation resulting from surgery or trauma. These complications extend hospital stay and are associated with long‐term disability and death. Patients undergoing total hip or knee replacement surgery or surgery for colorectal cancer are at high risk of venous thromboembolism. Sluggish venous blood flow, increased blood clotting and blood vessel wall injury are contributing factors. Treating more than one of these causes may improve prevention. Mechanical intermittent pneumatic leg compression (IPC) reduces sluggish blood flow (venous stasis) while medications such as aspirin and anticoagulants (low molecular weight heparin) reduce blood clotting. These medications are known as pharmacological prophylaxis (drugs used to prevent blood clots). However, these medications can also increase the risk of bleeding. This review is an update of a review first published in 2008.
Study characteristics and key results
We identified 22 trials with a total of 9137 participants to include in this review (current until May 2016). The mean age of participants, where reported, was 65.2 years. Most participants had either a high‐risk procedure or condition. The predisposing conditions were orthopedic surgery in 12 studies and urology, cardiothoracic, neurosurgery, trauma, general surgery, gynaecology or other types of participants in the remaining studies.
Compared to IPC alone, IPC plus medication did not show differences in the incidence (rate of new cases) of PE (12 studies with a total of 3017 participants). The incidence of DVT was reduced for IPC combined with medication when compared with IPC alone (11 studies with a total of 2934 participants). The addition of a medication to IPC, however, increased the risk for any bleeding compared to IPC alone, from 0.66% to 4.0%. Major bleeding followed a similar pattern, with an increase from 0.1% to 1.5%.Further analysis looking at different subgroups of participants (orthopedic and non‐orthopedic participants) did not show any overall difference in DVT while it was not possible to assess differences between subgroups for PE.
Compared with medication alone, combined IPC and medication reduced the incidence of PE (10 studies with 3544 participants). DVT incidence was not different between the medication and the combined IPC and medication group (11 studies with 2866 participants). No differences were observed in rates of bleeding (three studies with 244 participants). Further analysis looking at different subgroups of participants did not show any overall difference in incidence of PE and DVT between orthopedic and non‐orthopedic participants.
Quality of the evidence
The findings of this review show moderate quality evidence and agree with current guideline recommendations supporting the use of combined IPC and pharmacological prophylaxis, compared with IPC or pharmacological prophylaxis alone, to reduce the incidence of DVT and PE in hospitalized patients. Moderate quality evidence suggests the addition of pharmacological prophylaxis to IPC, increased the risk of bleeding compared to IPC alone, a side effect not observed for IPC when added to pharmacological prophylaxis (very low quality evidence), as expected for a physical method for preventing blood clots. The quality of the evidence was downgraded from high to moderate or very low for risk of bias and imprecision and indirectness between the studies.
Summary of findings
Background
Description of the condition
Deep vein thrombosis (DVT), i.e. the development of thrombi inside the deep veins of the legs (in most instances), is a potentially fatal disease as it can be complicated by pulmonary embolism (PE), resulting from the movement of thrombi from the leg veins to the pulmonary artery or its branches. The incidence of venous thromboembolism (VTE), DVT, PE or both, is still high despite the use of contemporary prophylactic measures. VTE risk is increased by the presence of certain risk factors, including, age, malignancy, immobilisation and the type of surgery. High‐risk patients include those undergoing total hip or knee replacement, or surgery for colorectal cancer (McLeod 2001). Experts in the field have indicated that this and similar observations are the result of failed and also omitted prophylaxis (Goldhaber 2001; Piazza 2007). The most recent guidelines recommend combined pharmacological and mechanical prophylaxis in high‐risk groups, in an effort to maximize thromboprophylaxis (Gould 2012; Nicolaides 2013). It is likely that mechanical methods increase the efficacy of thromboprophylaxis and reduce death and morbidity rates without increasing bleeding risk.
Description of the intervention
Intermittent pneumatic leg compression (IPC) involves wrapping the legs with inflatable sleeves, using commercially available devices. As a result of sleeve inflation, external pressure is exerted on the legs and its veins, resulting in an increase in blood flow and this reduction of blood stasis decreases the incidence of VTE. Pharmacological prophylaxis on the other hand is achieved by mostly small doses of anticoagulants given orally or subcutaneously; these also significantly reduce the incidence of VTE. Combined IPC and pharmacological prophylaxis in the form of dual modalities concurrently used for prevention of VTE may improve the efficacy of each method used alone.
How the intervention might work
Mechanical methods reduce VTE mainly by reducing venous stasis, while anticoagulants inhibit elements of the thrombosis cascade. Because single prophylactic modalities reduce but not completely eliminate VTE, combined modalities are expected to reduce further the frequency of VTE because of their different mechanisms of action.
Why it is important to do this review
Venous thromboembolism is the single most common, preventable cause of postoperative death. Better use of preventive resources is expected to reduce VTE events and mortality. Use of combined modalities is suggested by current guidelines in high risk patients, however the evidence supporting these recommendations requires better attention (Gould 2012; Nicolaides 2013). We performed this review to assess the breadth and strength of the best available evidence by pooling data from multiple studies to overcome limitations of small and underpowered studies. This review is an update of a review first published in 2008.
Objectives
The aim of this review was to assess the efficacy of combined intermittent pneumatic leg compression (IPC) and pharmacological prophylaxis versus single modalities in preventing venous thromboembolism.
Methods
Criteria for considering studies for this review
Types of studies
We included randomized controlled trials (RCTs) and controlled clinical trials (CCTs).
Types of participants
We included any type of hospitalized patient requiring prevention of venous thromboembolism (VTE) or at risk of developing VTE. We included patients undergoing surgery and trauma and ICU patients.
Types of interventions
We included studies which assessed the combined use of intermittent pneumatic leg compression (IPC) (including foot pumps and devices inflating calf sleeves) and pharmacological prophylaxis (including unfractionated heparin and low molecular weight heparin) compared with IPC or pharmacological prophylaxis alone. We excluded studies that used IPC for a short period of time (that is intraoperatively).
Types of outcome measures
Primary outcomes
Venous thromboembolism (symptomatic or asymptomatic) was the main outcome measure, with data on DVT and PE extracted as separate endpoints.
Outcomes were assessed by: ascending venography, I‐125 fibrinogen uptake test and ultrasound scanning for DVT; and pulmonary angiography or scintigraphy, computed tomography (CT), angiography and autopsy for PE.
Secondary outcomes
Bleeding was considered as a safety outcome and included all types reported i.e. any type, major bleeding (as defined by the study authors, but usually located at the surgical site or in a critical organ or site, requiring intervention or transfusion of at least two units of blood, or leading to death), and fatal bleeding reported separately. Fatal PE was an additional outcome.
Search methods for identification of studies
Electronic searches
For this update the Cochrane Vascular Information Specialist (CIS) searched the Specialised Register (May 2016). In addition the CIS searched the Cochrane Register of Studies (CRS) www.metaxis.com/CRSWeb/Index.asp (CENTRAL (2016, Issue 4)). See Appendix 1 for details of the search strategy used to search the CRS. The Specialised Register is maintained by the CIS and is constructed from weekly electronic searches of MEDLINE, EMBASE, CINAHL, AMED, and through handsearching relevant journals. The full list of the databases, journals and conference proceedings which have been searched, as well as the search strategies used are described in the Specialised Register section of the Cochrane Vascular module in the Cochrane Library (www.cochranelibrary.com).
The following trial databases were searched by the CIS (May 2016) for details of ongoing and unpublished studies using the terms pneumatic compression;
World Health Organization International Clinical Trials Registry (apps.who.int/trialsearch/)
ClinicalTrials.gov (clinicaltrials.gov/)
ISRCTN Register (www.isrctn.com/)
Searching other resources
We searched the reference lists of relevant articles and also similar systematic reviews and meta‐analyses to identify additional trials.
Data collection and analysis
Selection of studies
Two review authors (SK and IN) independently selected studies for inclusion on the basis of use of combined mechanical IPC and pharmacological modalities. Any disagreements were resolved by discussion.
Data extraction and management
SKK and two co‐authors (GG and IN) independently extracted the data. We used a data extraction form to record the type of patient or surgical procedure, total number of participants in the study (including those randomized, excluded and also withdrawn), the interventions used, the number of participants who reached an endpoint (DVT or PE) and the methodology used to establish this. A third author (JC) arbitrated any disagreements.
Assessment of risk of bias in included studies
We assessed the methodological quality of all included studies using Cochrane's 'Risk of bias' tool. . SKK and two co‐authors (GG and IN) independently performed the assessment according to Higgins 2011. The following domains were assessed: selection bias (random sequence generation, allocation concealment), performance bias (blinding of participants and personnel), detection bias (blinding of outcome assessment), attrition bias (incomplete outcome data), reporting bias (selective reporting) and other bias. We classified the domains as low risk, high risk, or unclear risk according to Higgins 2011. Any disagreements were resolved after discussion.
Measures of treatment effect
We performed separate analysis for the interventions of IPC versus combined modalities, and pharmacological prophylaxis versus combined modalities for the outcomes of PE and DVT. We used odds ratios (OR) and risk ratio (RR) with 95% confidence intervals (CIs) for assessment of dichotomous outcomes of all trials and RCTs only, respectively.
Unit of analysis issues
We excluded studies with non‐standard designs, such as cross‐over trials and cluster‐randomized trials. The individual patient was the unit of analysis.
Dealing with missing data
In case of missing participants due to drop‐out, we used intention‐to‐treat analysis. Where necessary, we contacted study authors to request that they provided any missing information.
Assessment of heterogeneity
We assessed statistical heterogeneity with the I2 test. I2 levels exceeding 50% were considered as substantial heterogeneity to justify the use of random‐effects analysis.
Assessment of reporting biases
We assessed publication bias with funnel plots when 10 or more studies were included in a comparison and contributed to the effect estimate; as described by Higgins 2011. Where the number of studies in each comparison was not greater than 10 the plots lack the power to distinguish chance from real asymmetry (Egger 1997).
Data synthesis
We used fixed‐effect models for each meta‐analysis to pool data, unless there was evidence of heterogeneity in which case we used a random‐effects model to calculate the RR or ORs and 95% CIs (see Assessment of heterogeneity)..
Subgroup analysis and investigation of heterogeneity
We performed subgroup analysis of the primary outcomes for surgery type (e.g. orthopedic surgery, etc), type of DVT (symptomatic) and type of IPC (foot IPC and other than foot IPC).
Sensitivity analysis
We performed sensitivity analysis of the primary outcomes by excluding studies with a high‐risk for bias, based on the 'Risk of bias' tool, and by excluding CCTs, in order to test the robustness of the evidence. Where significant heterogeneity was present, we performed sensitivity analysis to determine what effect this had on the results.
Summary of findings
We created 'Summary of findings' tables for the comparisons of IPC plus pharmacological prophylaxis versus IPC alone (Table 1) and IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone (Table 2). We used GRADEpro GDT software (GRADEpro GDT 2015) to present the main findings of the review. We included the primary outcomes DVT and PE and incidence of bleeding and major bleeding in the 'Summary of findings' tables. We calculated assumed control intervention risks from the mean number of events in the control groups of the selected studies for each outcome. The system developed by the Grading of Recommendation, Assessment, Development and Evaluation Working Group (GRADE working group) was used for grading the quality of evidence as high, moderate, low and very low, based on within‐study risk of bias, directness of evidence, heterogeneity, precision of effects estimates, and risk of publication bias (Atkins 2004).
Summary of findings for the main comparison. IPC plus pharmacological prophylaxis versus IPC alone.
| Does combined intermittent pneumatic compression (IPC) plus pharmacological prophylaxis increase prevention of venous thromboembolism compared with IPC alone? | ||||||
|
Patient or population: patients undergoing surgery or at risk of developing VTE because of other reasons (e.g. trauma) Settings: hospital (surgery, trauma or ICU stay) Intervention: combined IPC plus pharmacological prophylaxis Comparison: IPC alone | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Single modalities | Combined modalities | |||||
| Incidence of PEa | 8 per 1000 | 4 per 1000 (1 to 10) | OR 0.49 (0.18 to 1.34) | 3017 (12) | ⊕⊕⊕⊝ moderate1 | |
| Incidence of DVTb | 41 per 1000 | 22 per 1000 (14 to 34) | OR 0.52 (0.33 to 0.82) | 2934 (11) | ⊕⊕⊕⊝ moderate2 | |
| Incidence of bleedingc | 7 per 1000 | 33 per 1000 (16 to 67) | OR 5.04 (2.36 to 10.77) | 2155 (7) | ⊕⊕⊕⊝ moderate3 |
|
| Incidence of major bleedingd | 1 per 1000 | 6 per 1000 (2 to 22) | OR 6.81 (1.99 to 23.28) | 2155 (7) | ⊕⊕⊕⊝ moderate3 |
|
| * The basis for the assumed risk was the average risk in the single modalities group (i.e. the number of participants with events divided by total number of participants of the single modalities group included in the meta‐analysis). The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI) CI: confidence interval;DVT: deep vein thrombosis;IPC: intermittent pneumatic compression; OR: odds ratio; PE: pulmonary embolism; VTE: venous thromboembolism | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
a PE assessed by pulmonary angiography or scintigraphy, computed tomography (CT), angiography, or autopsy b DVT assessed by ascending venography, I‐125 fibrinogen uptake test, and ultrasound scanning c any type of bleeding as described by the study authors d major bleeding as defined by the study authors, but usually located at the surgical site or in a critical organ or site, requiring intervention or transfusion of at least units of blood, or leading to death 1 Downgraded by one level due to imprecision likely due to a type II error (few events and 4/12 studies contributing to effect estimate) 2 Downgraded by one level, due to risk of attrition bias, affecting effect estimate as shown by sensitivity analysis 3 Downgraded by one level due to indirectness (reporting of bleeding outcomes (major and minor bleeding) was not uniform across the studies, with some studies reporting on blood loss during the procedures or through the drains or providing rates for postoperative bleeding. Definitions used were also not uniform) Bleeding events may be affected by bias due to blinding. Only two out of seven studies are double blind. These are also the two largest studies in the analysis. When pooled they show a similar direction of effect (increased bleeding for combined modalities) as the overall effect for the seven studies in this comparison indicating that any potential risk of risk of performance or detection bias does not affect the results therefore not downgraded for risk of bias Wide confidence interval but upper and lower limits of corresponding risk and 95% confidence interval of effect both show the same message i.e. an increased risk of bleeding for combined modalities therefore not downgraded for imprecision
Summary of findings 2. IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone.
| Does combined intermittent pneumatic compression (IPC) plus pharmacological prophylaxis increase prevention of venous thromboembolism compared with pharmacological prophylaxis alone? | ||||||
|
Patient or population: patients undergoing surgery or at risk of developing VTE because of other reasons (e.g. trauma) Settings: hospital (surgery, trauma or ICU stay) Intervention: combined IPC plus pharmacological prophylaxis Comparison: pharmacological prophylaxis alone | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Single modalities | Combined modalities | |||||
| Incidence of PEa | 29 per 1000 | 12 per 1000 (7 to 19) | OR 0.39 (0.23 to 0.64) | 3544 (10) | ⊕⊕⊕⊝ moderate1 | |
| Incidence of DVTb | 62 per 1000 | 27 per 1000 (12 to 64) | OR 0.42 (0.18 to 1.03) | 2866 (11) | ⊕⊕⊕⊝ moderate2 | |
| Incidence of bleedingc | 81 per 1000 | 66 per 1000 (26 to 159) | OR 0.8 (0.3 to 2.14 | 244 (3) | ⊕⊝⊝⊝ very low3 |
|
| Incidence of major bleedingd | 41 per 1000 | 49 per 1000 (15 to 150) | OR 1.21 (0.35 to 4.18) | 244 (3) | ⊕⊝⊝⊝ very low3 |
|
| *The basis for the assumed risk was the average risk in the single modalities group (i.e. the number of participants with events divided by total number of participants of the single modalities group included in the meta‐analysis). The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI) CI: confidence interval;DVT: deep vein thrombosis;IPC: intermittent pneumatic compression; OR: odds ratio; PE: pulmonary embolism; VTE: venous thromboembolism | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
a PE assessed by pulmonary angiography or scintigraphy, computed tomography (CT), angiography, or autopsy b DVT assessed by ascending venography, I‐125 fibrinogen uptake test, and ultrasound scanning c any type of bleeding as described by the study authors d major bleeding as defined by the study authors, but usually located at the surgical site or in a critical organ or site, requiring intervention or transfusion of at least units of blood, or leading to death
1 Downgraded by one level due to risk of detection and attrition bias affecting effect estimate as shown by sensitivity analysis 2 Downgraded by one level, due to risk of selection, detection and other bias affecting effect estimate as shown by sensitivity analysis. Heterogeneity explained by detection and other bias 3 Downgraded by three levels due to risk of bias due to blinding (none of the studies in this comparison are double blind), indirectness (reporting of bleeding outcomes (major and minor bleeding) was not uniform across the studies, with some studies reporting on blood loss during the procedures or through the drains or providing rates for postoperative bleeding and definitions used were not uniform) and imprecision (small number of participants and relatively few events
Results
Description of studies
Results of the search
See Figure 1.
1.

Study flow diagram.
Included studies
For this update 11 additional studies were included (Cahan 2000; Dickinson 1998; Edwards 2008; Kurtoglu 2003; Sakai 2016; Siragusa 1994; Song 2014; Stannard 1996; Tsutsumi 2012; Windisch 2011; Yokote 2011) making a total of 22 studies that met the inclusion criteria, reporting outcomes on 9137 participants (Bigg 1992; Borow 1983; Bradley 1993; Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Kurtoglu 2003; Ramos 1996; Sakai 2016; Sieber 1997; Silbersack 2004; Siragusa 1994; Song 2014; Stannard 1996; Tsutsumi 2012; Turpie 2007; Westrich 2005; Westrich 2006; Windisch 2011; Woolson 1991; Yokote 2011). Four publications had three arms (Borow 1983; Cahan 2000; Dickinson 1998; Stannard 1996), using IPC, pharmacological prophylaxis and both, respectively. Fifteen studies were RCTs (Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Ramos 1996; Sakai 2016; Silbersack 2004; Siragusa 1994; Song 2014; Stannard 1996; Turpie 2007; Westrich 2006; Windisch 2011; Yokote 2011; Woolson 1991) that studied a total of 7762 participants. The remaining seven studies were CCTs, which were classified according to the draft guidelines of the Cochrane Non‐Randomised Studies Methods Group (NRSMG). These included three quasi‐randomized CCTs (Bigg 1992; Bradley 1993; Kurtoglu 2003) that studied a total of 222 participants, and four CCTs with concurrent controls (Borow 1983; Sieber 1997; Tsutsumi 2012; Westrich 2005) that involved a total of 1153 participants.
The included studies evaluated orthopedic patients (n = 12) (Bradley 1993; Edwards 2008; Eisele 2007; Sakai 2016; Silbersack 2004; Siragusa 1994; Stannard 1996; Westrich 2005; Westrich 2006; Windisch 2011; Woolson 1991; Yokote 2011); urology patients (n = 2) (Bigg 1992; Sieber 1997); cardiothoracic patients (n = 1) (Ramos 1996); neurosurgery patients (n = 1) (Dickinson 1998); trauma patients (n = 1) (Kurtoglu 2003) or general surgery, gynecology and other types of patients (n = 5) (Borow 1983; Cahan 2000; Song 2014; Tsutsumi 2012; Turpie 2007). Patient weighted mean age (in 17 studies that reported age, 5638 participants) was 65.2 years (Bigg 1992; Bradley 1993; Cahan 2000; Dickinson 1998; Edwards 2008; Ramos 1996; Sakai 2016; Sieber 1997; Silbersack 2004; Song 2014; Stannard 1996; Tsutsumi 2012; Westrich 2005; Westrich 2006; Windisch 2011; Woolson 1991; Yokote 2011).
Pharmacological prophylaxis included unfractionated heparin (UFH) (Bigg 1992; Bradley 1993; Cahan 2000; Ramos 1996; Sieber 1997; Siragusa 1994; Stannard 1996), low molecular weight heparin (LMWH) (Dickinson 1998; Edwards 2008; Kurtoglu 2003; Silbersack 2004; Song 2014; Westrich 2006; Windisch 2011), fondaparinux (Eisele 2007; Tsutsumi 2012; Turpie 2007), LMWH or fondaparinux (Yokote 2011), UFH or warfarin (Borow 1983), warfarin or aspirin (Westrich 2005; Woolson 1991) and edoxaban, a direct oral Factor Xa inhibitor (Sakai 2016).
IPC types included foot pumps (Bradley 1993; Sakai 2016; Stannard 1996; Windisch 2011), and devices inflating calf sleeves (Edwards 2008; Eisele 2007; Silbersack 2004; Westrich 2005; Westrich 2006), or thigh‐high sleeves (Bigg 1992; Borow 1983; Cahan 2000; Dickinson 1998; Ramos 1996; Sieber 1997; Song 2014; Woolson 1991). The exact IPC type was not reported in four studies (Kurtoglu 2003; Siragusa 1994; Tsutsumi 2012; Yokote 2011), while in one multi centre study the investigators were allowed to use the device type of their choice (Turpie 2007).
Four publications had three arms (Borow 1983; Cahan 2000; Dickinson 1998; Stannard 1996), using IPC, pharmacological prophylaxis and both, respectively. Of the remaining 18 publications, prophylactic methods in the control group included: IPC in 11 studies, either without aspirin (Bigg 1992; Kurtoglu 2003; Ramos 1996; Sieber 1997; Song 2014; Tsutsumi 2012; Turpie 2007; Woolson 1991; Yokote 2011) or with aspirin (Westrich 2005; Westrich 2006); and pharmacological prophylaxis in seven studies (Bradley 1993; Edwards 2008; Eisele 2007; Sakai 2016; Silbersack 2004; Siragusa 1994; Windisch 2011), with aspirin in one of them (Silbersack 2004). The intervention group in all studies used combined modalities and aspirin was used only in two studies (Stannard 1996; Woolson 1991).
Ultrasound was the main diagnostic modality to diagnose DVT and was used by most studies (Borow 1983; Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Kurtoglu 2003; Sakai 2016; Silbersack 2004; Siragusa 1994; Song 2014; Stannard 1996; Westrich 2005; Westrich 2006; Windisch 2011; Woolson 1991; Yokote 2011). PE, where reported, was diagnosed mainly with scintigraphy scanning (Bigg 1992; Ramos 1996; Turpie 2007; Woolson 1991), a pulmonary angiogram (Ramos 1996; Turpie 2007); or a CT pulmonary angiogram (Kurtoglu 2003; Sakai 2016; Silbersack 2004; Song 2014; Tsutsumi 2012; Turpie 2007; Westrich 2006; Windisch 2011; Yokote 2011).
Two studies did not report on DVT rates (Bigg 1992; Ramos 1996), and four studies did not report on PE rates (Bradley 1993; Dickinson 1998; Eisele 2007; Siragusa 1994).
Eleven studies reported on bleeding outcomes (Bigg 1992; Dickinson 1998; Sakai 2016; Song 2014; Tsutsumi 2012; Turpie 2007; Westrich 2005; Westrich 2006; Windisch 2011; Woolson 1991; Yokote 2011). For many studies no specific bleeding definitions were provided (Bigg 1992; Dickinson 1998; Song 2014; Westrich 2005; Westrich 2006; Windisch 2011; Woolson 1991). For the remaining studies that did provide bleeding definitions, the criteria were not uniform (Sakai 2016; Tsutsumi 2012; Turpie 2007; Yokote 2011).
Excluded studies
For this update an additional nine studies were excluded (Gagner 2012; Kiudelis 2010; Kumaran 2008; Lieberman 1994; Mehta 2010; Patel 2010; Wan 2015; Westrich 1996; Whitworth 2011); making a total of 21 excluded studies (Ailawadi 2001; Eskander 1997; Frim 1992; Gagner 2012; Gelfer 2006; Kamran 1998; Kiudelis 2010; Kumaran 2008; Lieberman 1994; Macdonald 2003; Mehta 2010; Nathan 2006; Patel 2010; Roberts 1975; Spinal cord injury investigators; Stannard 2006; Tsutsumi 2000; Wan 2015; Westrich 1996; Whitworth 2011; Winemiller 1999). Exclusions were due to: use of combined modalities was not concurrent or a different type of pharmacological prophylaxis was given in the two study groups (n = 5) (Eskander 1997; Gelfer 2006; Macdonald 2003; Spinal cord injury investigators; Stannard 2006); IPC use was limited to intraoperative use (n = 2) (Kiudelis 2010; Roberts 1975); they were controlled before and after studies (n = 3) (Frim 1992; Kamran 1998; Tsutsumi 2000); they were retrospective case‐control studies (n = 6) (Ailawadi 2001; Nathan 2006; Patel 2010; Wan 2015; Whitworth 2011; Winemiller 1999); a registry study (n = 1) (Gagner 2012); the single modalities group used either heparin or IPC (n = 1) (Kumaran 2008); aspirin was used for thromboprophylaxis (n = 2) (Lieberman 1994; Westrich 1996), and only aggregated VTE rates and not separate DVT and PE rates were provided (n = 1) (Mehta 2010).
Ongoing studies
Three additional studies were identified as ongoing (CHICTR‐IPR‐15007324; ISRCTN44653506 and NCT02040103; NCT00740987). See Characteristics of ongoing studies.
Risk of bias in included studies
2.
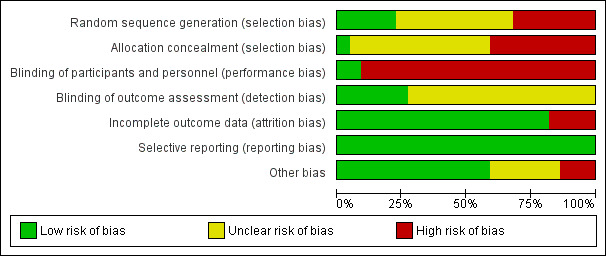
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Overall risk of bias was mostly unclear or high.
Allocation
The randomization method was unclear in 10 of the 15 RCTs (Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Silbersack 2004; Siragusa 1994; Stannard 1996; Westrich 2006; Windisch 2011; Yokote 2011). The studies that provided this information used random tables (Ramos 1996), a centralized computer‐generated schedule (Sakai 2016; Song 2014; Turpie 2007), and sealed envelopes (Woolson 1991). As a result, the quality of RCTs was mostly poor regarding selection bias, which was generally at high risk. By definition all quasi‐randomized trials and CCTs had a high risk for random sequence generation and therefore selection bias.
A high risk for allocation concealment was evident in nine studies (Bigg 1992; Borow 1983; Bradley 1993; Kurtoglu 2003; Sakai 2016; Sieber 1997; Song 2014; Tsutsumi 2012; Westrich 2005). Only Turpie 2007 had a low risk for allocation bias. In the remaining studies the risk of selection bias due to allocation concealment was unclear (Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Ramos 1996; Silbersack 2004; Siragusa 1994; Stannard 1996; Westrich 2006; Windisch 2011; Woolson 1991; Yokote 2011).
Blinding
A high risk of performance bias was evident in all studies except two recent RCTs, which were double‐blinded (Turpie 2007; Yokote 2011). The remaining studies were judged as being at high risk because of the lack of use of a placebo medication or device. Blinding of outcome assessment was reported by six studies (Bradley 1993; Kurtoglu 2003; Stannard 1996; Turpie 2007; Windisch 2011; Yokote 2011), while in the remaining studies there was unclear evidence of detection bias. This lack of blinding may have affected the detection of DVT or PE and potentially increase the heterogeneity of the results.
Incomplete outcome data
A total of 324 participants (3.5%) were excluded. One study excluded eight participants due to non‐compliance, confinement to bed for more than one week, premature transfer to a different institution, or re‐operation or discharge from hospital without ultrasonography (Silbersack 2004). One study excluded 11 participants because of a protocol violation (discharged before the ultrasound) (n = 6), or because they did not receive the correct study medication (n = 5) (Westrich 2006). A third study excluded 24 participants because inclusion or exclusion criteria were not met, informed consent was withdrawn, adverse events occurred, or for other reasons not stated (Turpie 2007). Three additional studies excluded 3, 43 and 235 participants respectively (Edwards 2008; Ramos 1996; Song 2014).
A total of 78 participants (0.85%) were lost to follow‐up. One study reported a 26.5% loss to follow up (Westrich 2006), which was 0.8% of the total number of participants in this systematic review; however, short‐term data were provided.
Selective reporting
No findings of selective reporting were identified.
Other potential sources of bias
Three studies were considered as being at high risk for other sources of bias (Dickinson 1998; Edwards 2008; Sakai 2016). Reasons for this were because they were prematurely stopped (Dickinson 1998; Sakai 2016); or had a large number of post‐randomization exclusions (Edwards 2008).
Effects of interventions
Intermittent pneumatic leg compression (IPC) plus pharmacological prophylaxis versus IPC alone
See Table 1.
Twelve of the included studies evaluated the role of combined modalities on the incidence of symptomatic PE (Bigg 1992; Borow 1983; Cahan 2000; Dickinson 1998; Kurtoglu 2003; Sieber 1997; Song 2014; Stannard 1996; Tsutsumi 2012; Turpie 2007; Woolson 1991; Yokote 2011). The incidence of PE was 0.79% (10/1281) in the control group and 0.46% (8/1736) in the combined group showing no difference between the combined and control groups; OR 0.49, 95% CI 0.18 to 1.34; participants = 3017; studies = 12; Analysis 1.1. Results did not demonstrate heterogeneity (I2 = 0%). The quality of the evidence was downgraded to moderate for imprecision likely due to a type II error. Fatal PE was not reported.
1.1. Analysis.
Comparison 1 IPC plus pharmacological prophylaxis versus IPC alone, Outcome 1 Incidence of PE.
Eleven studies investigated the role of combined modalities on the incidence of DVT (Borow 1983; Cahan 2000; Dickinson 1998; Kurtoglu 2003; Sieber 1997; Song 2014; Stannard 1996; Tsutsumi 2012; Turpie 2007; Woolson 1991; Yokote 2011). The incidence of DVT was 4.10% (51/1243) in the control group and 2.19% (37/1691) in the combined group showing a reduced incidence of DVT in favor of the combined modalities group (OR 0.52, 95% CI 0.33 to 0.82; participants = 2934; studies = 11), Analysis 1.2. Results did not demonstrate heterogeneity (I2 = 0%). The quality of the evidence was downgraded to moderate for risk of attrition bias (see sensitivity analysis below).
1.2. Analysis.
Comparison 1 IPC plus pharmacological prophylaxis versus IPC alone, Outcome 2 Incidence of DVT.
Six studies reported on the occurrence of symptomatic DVT (Cahan 2000; Sieber 1997; Song 2014; Tsutsumi 2012; Turpie 2007; Yokote 2011). The incidence of symptomatic DVT was 0.48% (5/1043) in the control group and 0.54% (8/1483) in the combined modalities group, showing no difference between the combined and control groups (OR 0.49, 95% CI 0.16 to 1.47; participants = 2526; studies = 6), Analysis 1.3. Results did not demonstrate heterogeneity (I2 = 0%).
1.3. Analysis.
Comparison 1 IPC plus pharmacological prophylaxis versus IPC alone, Outcome 3 Incidence of symptomatic DVT.
One study investigated the role of combined modalities on the incidence of DVT using a foot IPC (Stannard 1996), but because of a lack of events a risk estimate could not be calculated. Ten studies investigated the role of combined modalities on the incidence of DVT using IPC other than a foot IPC (Borow 1983; Cahan 2000; Dickinson 1998; Kurtoglu 2003; Sieber 1997; Song 2014; Tsutsumi 2012; Turpie 2007; Woolson 1991; Yokote 2011). The incidence of DVT was 4.19% (51/1218) in the control group and 2.22% (37/1666) in the combined group showing a reduced incidence of DVT in favor of the combined modalities group (OR 0.52, 95% CI 0.33 to 0.82; participants = 2884; studies = 10), Analysis 1.4. Results did not demonstrate heterogeneity (I2 = 0%).
1.4. Analysis.
Comparison 1 IPC plus pharmacological prophylaxis versus IPC alone, Outcome 4 Incidence of DVT by foot IPC or other IPC.
Seven studies reported on the incidence of bleeding in the combined modalities and IPC groups (Bigg 1992; Dickinson 1998; Song 2014; Tsutsumi 2012; Turpie 2007; Woolson 1991; Yokote 2011). The incidence of bleeding was 0.66% (7/1053) in the control (IPC) group and 4.0% (44/1102) in the combined group showing an increase in bleeding in the combined group (OR 5.04, 95% CI 2.36 to 10.77; participants = 2155; studies = 7), Analysis 1.5. Results did not demonstrate heterogeneity (I2 = 0%). The quality of the evidence was downgraded to moderate for indirectness. Major bleeding followed a similar pattern, with an incidence of 0.1% (1/1053) in the control (IPC) group and 1.5% (17/1102) in the combined group (OR 6.81, 95% CI 1.99 to 23.28; participants = 2155; studies = 7), Analysis 1.6. Results did not demonstrate heterogeneity (I2 = 0%). The quality of the evidence was downgraded to moderate for indirectness. Fatal bleeding during the intervention period was not reported.
1.5. Analysis.
Comparison 1 IPC plus pharmacological prophylaxis versus IPC alone, Outcome 5 Incidence of bleeding.
1.6. Analysis.
Comparison 1 IPC plus pharmacological prophylaxis versus IPC alone, Outcome 6 Incidence of major bleeding.
IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone
See Table 2.
Ten studies evaluated the role of combined modalities on the incidence of symptomatic PE (Borow 1983; Bradley 1993; Cahan 2000; Dickinson 1998; Edwards 2008; Ramos 1996; Sakai 2016; Silbersack 2004; Stannard 1996; Windisch 2011). The incidence of PE was 2.92% (50/1711) in the control group and 1.20% (22/1833) in the combined group showing a reduction in PE in favor of the combined modalities group (OR 0.39, 95% CI 0.23 to 0.64; participants = 3544; studies = 10), Analysis 2.1. Results did not demonstrate heterogeneity (I2 = 0%). The quality of the evidence was downgraded to moderate for risk of detection and attrition bias (see sensitivity analysis below). Fatal PE was not reported, except in one study, which did not provide the exact number of deaths or the treatment group they occurred in (Ramos 1996).
2.1. Analysis.
Comparison 2 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone, Outcome 1 Incidence of PE.
Eleven studies investigated the role of combined modalities on the incidence of DVT (Borow 1983; Bradley 1993; Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Sakai 2016; Silbersack 2004; Siragusa 1994; Stannard 1996; Windisch 2011). The incidence of DVT was 6.20 (90/1452) in the control group and 2.90% (41/1414) in the combined group showing no difference between the combined and control groups (OR 0.42, 95% CI 0.18 to 1.03; participants = 2866; studies = 11), Analysis 2.2. Results demonstrated significant heterogeneity (I2 = 68%). The quality of the evidence was downgraded to moderate for risk of selection, detection and other bias (see sensitivity analysis below).
2.2. Analysis.
Comparison 2 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone, Outcome 2 Incidence of DVT.
Five studies reported on the occurrence of symptomatic DVT (Cahan 2000; Edwards 2008; Eisele 2007; Sakai 2016; Windisch 2011). The incidence of symptomatic DVT was 0.43% (5/1157) in the control group and 0.43% (5/1155) in the combined group showing no difference between the combined and control groups (OR 1.02, 95% CI 0.29 to 3.54; participants = 2312; studies = 5), Analysis 2.3. Results did not demonstrate heterogeneity (I2 = 0%).
2.3. Analysis.
Comparison 2 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone, Outcome 3 Incidence of symptomatic DVT.
Four studies investigated the role of combined modalities on the incidence of DVT using a foot IPC (Bradley 1993; Sakai 2016; Stannard 1996; Windisch 2011). The incidence of DVT was 16.37% (28/171) in the control group and 13.07% (20/153) in the combined group showing no difference between the combined and control groups (OR 0.40, 95% CI 0.05 to 3.47; participants = 324; studies = 4). Results demonstrated significant heterogeneity (I2 = 81%). Seven studies investigated the role of combined modalities on the incidence of DVT using IPC other than a foot IPC (Borow 1983; Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Silbersack 2004; Siragusa 1994). The incidence of DVT was 4.83% (62/1281) in the control group and 1.67% (21/1261) in the combined group showing a reduced incidence of DVT in favor of the combined modalities group (OR 0.39, 95% CI 0.16 to 0.96; participants = 2542; studies = 7), Analysis 2.4. Results did not demonstrate significant heterogeneity (I2 = 51%).
2.4. Analysis.
Comparison 2 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone, Outcome 4 Incidence of DVT by foot IPC or other IPC.
Three studies reported on the incidence of bleeding in the combined and pharmacological prophylaxis groups (Dickinson 1998; Sakai 2016; Windisch 2011). These studies showed no difference in bleeding rates between the combined group (8/121, 6.6%) and the control group (10/123, 8.1%) (OR 0.80, 95% CI 0.30 to 2.14; participants = 244; studies = 3), Analysis 2.5. Results did not demonstrate heterogeneity (I2 = 0%). The quality of the evidence was downgraded to very low for risk of bias due to blinding, indirectness.and imprecision. There was also no difference in major bleeding rates between the combined group (6/121, 5.0%) and the control group (5/123, 4.1%) (OR 1.21, 95% CI 0.35 to 4.18; participants = 244; studies = 3), Analysis 2.6. Results did not demonstrate heterogeneity (I2 = 0%). Fatal bleeding during the intervention period was not reported.
2.5. Analysis.
Comparison 2 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone, Outcome 5 Incidence of bleeding.
2.6. Analysis.
Comparison 2 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone, Outcome 6 Incidence of major bleeding.
IPC plus pharmacological prophylaxis versus IPC plus aspirin
Three studies evaluated the role of combined IPC plus pharmacological prophylaxis versus IPC plus aspirin on the incidence of symptomatic PE (Westrich 2005; Westrich 2006; Woolson 1991). The studies showed a similar frequency of PE in the IPC plus aspirin control (2/268, 0.75%) and IPC plus pharmacological prophylaxis treatment groups (0/337, 0%) (OR 0.33, 95% CI 0.03 to 3.19; participants = 605; studies = 3), Analysis 3.1. Results did not demonstrate heterogeneity (I2 = 0%). Fatal PE was not reported.
3.1. Analysis.
Comparison 3 IPC plus pharmacological prophylaxis versus IPC plus aspirin, Outcome 1 Incidence of PE.
The same studies investigated the role of combined modalities on the incidence of DVT. The studies showed a similar frequency in DVT in the IPC plus aspirin control (32/268, 11.9%) and IPC plus pharmacological prophylaxis treatment groups (30/337, 8.9%) (OR 0.83, 95% CI 0.48 to 1.42; participants = 605; studies = 3), Analysis 3.2. Results did not demonstrate heterogeneity (I2 = 0%).
3.2. Analysis.
Comparison 3 IPC plus pharmacological prophylaxis versus IPC plus aspirin, Outcome 2 Incidence of DVT.
One study reported on the occurrence of symptomatic DVT (Westrich 2005), but because of the lack of events, a risk estimate could not be calculated (Analysis 3.3). No foot IPC was used in this comparison and therefore subgroup analysis was not possible.
3.3. Analysis.
Comparison 3 IPC plus pharmacological prophylaxis versus IPC plus aspirin, Outcome 3 Incidence of symptomatic DVT.
Three studies evaluated the role of combined IPC plus pharmacological prophylaxis versus IPC plus aspirin on the incidence of bleeding (Westrich 2005; Westrich 2006; Woolson 1991). The studies showed a similar frequency in bleeding in the IPC plus aspirin control (2/275, 0.7%) and IPC plus pharmacological prophylaxis treatment groups (4/341, 1.2%) (OR 1.23, 95% CI 0.27 to 5.53; participants = 616; studies = 3), Analysis 3.4. Results did not demonstrate heterogeneity (I2 = 0%). These studies showed also a similar frequency in major bleeding in the IPC plus aspirin control (2/275, 0.7%) and IPC plus pharmacological prophylaxis treatment groups (2/341, 0.6%) (OR 0.80, 95% CI 0.15 to 4.17; participants = 616; studies = 3), Analysis 3.5. Results did not demonstrate heterogeneity (I2 = 0%). Fatal bleeding during the intervention period was not reported.
3.4. Analysis.
Comparison 3 IPC plus pharmacological prophylaxis versus IPC plus aspirin, Outcome 4 Incidence of bleeding.
3.5. Analysis.
Comparison 3 IPC plus pharmacological prophylaxis versus IPC plus aspirin, Outcome 5 Incidence of major bleeding.
Subgroup analysis according to surgery type
IPC plus pharmacological prophylaxis versus IPC in orthopedic and non‐orthopedic participants
Three studies evaluated the role of combined modalities on the incidence of symptomatic PE in orthopedic participants (Stannard 1996; Woolson 1991; Yokote 2011). There were no events, which precluded any comparison (Analysis 4.1).
4.1. Analysis.
Comparison 4 IPC plus pharmacological prophylaxis versus IPC alone ‐ subgroups, Outcome 1 Incidence of PE.
Nine studies evaluated the role of combined modalities versus IPC alone on the incidence of symptomatic PE in participants not undergoing orthopedic surgery (Bigg 1992; Borow 1983; Cahan 2000; Dickinson 1998; Kurtoglu 2003; Sieber 1997; Song 2014; Tsutsumi 2012; Turpie 2007). These studies showed no difference in the incidence of PE ( 0.91% (10/1097) in the control group and 0.54% (8/1475) in the combined treatment group(OR 0.49, 95% CI 0.18 to 1.34; participants = 2572; studies = 9), Analysis 4.1. Results did not demonstrate heterogeneity (I2 = 0%).
It was not possible to test for differences between the orthopedic and non‐orthopedic subgroups for PE incidence as no PE events were reported in the orthopedic subgroup.
The same three studies evaluated the role of combined modalities on the incidence of DVT in orthopedic participants (Stannard 1996; Woolson 1991; Yokote 2011). These studies showed no difference in the incidence of DVT (8.15% (15/184) in the control group and 6.51% (17/261) in the combined treatment group OR 0.80, 95% CI 0.38 to 1.69; participants = 445; studies = 3), Analysis 4.2. Results did not demonstrate heterogeneity (I2 = 0%).
4.2. Analysis.
Comparison 4 IPC plus pharmacological prophylaxis versus IPC alone ‐ subgroups, Outcome 2 Incidence of DVT.
Eight studies evaluated the role of combined modalities on the incidence of DVT in participants not under going orthopedic surgery (Borow 1983; Cahan 2000; Dickinson 1998; Kurtoglu 2003; Sieber 1997; Song 2014; Tsutsumi 2012; Turpie 2007). These studies showed a reduction in DVT for the combined treatment group (3.40% (36/1059) in the control group and 1.40% (20/1430) in the combined treatment group, OR 0.41, 95% CI 0.23 to 0.73; participants = 2489; studies = 8), Analysis 4.2. Results did not demonstrate heterogeneity (I2 = 0%).
No difference was detected between the orthopedic and non‐orthopedic subgroups for DVT incidence (P = 0.16).
IPC plus pharmacological prophylaxis versus pharmacological prophylaxis in orthopedic and non‐orthopedic participants
Six studies evaluated the role of combined modalities on the incidence of symptomatic PE in orthopedic participants (Bradley 1993; Edwards 2008; Sakai 2016; Silbersack 2004; Stannard 1996; Windisch 2011). These trials showed no difference in PE (0.54% (2/370) in the control group and 0.28% (1/362) in the combined treatment group, OR 0.58, 95% CI 0.08 to 4.49; participants = 732; studies = 6), Analysis 5.1. Results did not demonstrate heterogeneity (I2 = 0%).
5.1. Analysis.
Comparison 5 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone ‐ subgroups, Outcome 1 Incidence of PE.
Four studies evaluated the role of combined modalities versus pharmacological prophylaxis alone on the incidence of symptomatic PE in participants not undergoing orthopedic surgery (Borow 1983; Cahan 2000; Dickinson 1998; Ramos 1996). These studies showed a reduction in incidence of PE in favor of the combined treatment group; 3.58% (48/1341) in the control group and 1.43% (21/1471) in the combined treatment group (OR 0.38, 95% CI 0.22 to 0.63; participants = 2812; studies = 4), Analysis 5.1. Heterogeneity could not be calculated as only one study reported any PE events (Ramos 1996).
No difference was detected between the orthopedic and non‐orthopedic subgroups for PE incidence (P = 0.68).
Eight studies investigated the role of combined modalities on the incidence of DVT in orthopedic participants (Bradley 1993; Edwards 2008; Eisele 2007; Sakai 2016; Silbersack 2004; Siragusa 1994; Stannard 1996; Windisch 2011). These trials showed a reduction in incidence of DVT in favor of the combined treatment group; 6.66% (87/1307) in the control group to 2.79% (36/1298) in the combined treatment group OR 0.32, 95% CI 0.12 to 0.86; participants = 2605; studies = 8), Analysis 5.2. Results did demonstrate significant heterogeneity (I2 = 73%), and a random‐effects model was used.
5.2. Analysis.
Comparison 5 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone ‐ subgroups, Outcome 2 Incidence of DVT.
Three studies investigated the role of combined modalities versus pharmacological prophylaxis alone on the incidence of DVT in participants not undergoing orthopedic surgery (Borow 1983; Cahan 2000; Dickinson 1998). These trials showed a similar frequency in DVT, 2.07% (3/145) in the control group and 4.31% (5/116) in the combined treatment group, OR 1.77, 95% CI 0.30 to 10.58; participants = 261; studies = 3), Analysis 5.2. Results demonstrated low heterogeneity (I2 = 14%).
No difference was detected between the orthopedic and non‐orthopedic subgroups for DVT incidence (P = 0.1).
IPC plus pharmacological prophylaxis versus IPC plus aspirin ‐ subgroups
The three studies in this comparison all included orthopedic participants and therefore subgroup analyses between orthopedic and non‐orthopedic groups were not possible (Westrich 2005; Westrich 2006; Woolson 1991). Details of the studies in this comparison are reported above and in Analysis 3.1 and Analysis 3.2.
Sensitivity analysis
Randomized controlled trials only ‐ intermittent pneumatic leg compression (IPC) plus pharmacological prophylaxis versus IPC alone
Seven RCTs evaluated the role of combined modalities on the incidence of symptomatic PE (Cahan 2000; Dickinson 1998; Song 2014; Stannard 1996; Turpie 2007; Woolson 1991; Yokote 2011). These trials showed no difference in incidence of PE; 0.41% (4/980) in the control group to 0.19% (2/1043) in the combined group, RR 0.51, 95% CI 0.09 to 2.76; participants = 2023; studies = 7), Analysis 6.1. Results did not demonstrate heterogeneity (I2 = 0%).
6.1. Analysis.
Comparison 6 IPC plus pharmacological prophylaxis versus IPC alone ‐ RCTs only, Outcome 1 Incidence of PE.
The same RCTs evaluated the role of combined modalities on the incidence of DVT (Cahan 2000; Dickinson 1998; Song 2014; Stannard 1996; Turpie 2007; Woolson 1991; Yokote 2011). These trials showed a reduction in DVT in favor of the combined treatment group; 4.40% (43/978) in the control group and 2.72% (28/1030) in the combined treatment group, RR 0.56, 95% CI 0.35 to 0.90; participants = 2008; studies = 7), Analysis 6.2. Low heterogeneity was present (I2 = 19%).
6.2. Analysis.
Comparison 6 IPC plus pharmacological prophylaxis versus IPC alone ‐ RCTs only, Outcome 2 Incidence of DVT.
Randomized controlled trials only ‐ IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone
Eight RCTs evaluated the effect of combined modalities on the incidence of symptomatic PE (Cahan 2000; Dickinson 1998; Edwards 2008, Ramos 1996; Sakai 2016; Silbersack 2004; Stannard 1996; Windisch 2011). These trials showed a reduction in the incidence of PE in favor of the combined treatment group; 3.20% (50/1560) in the control group and 1.28% (22/1725) in the combined treatment group, RR 0.40, 95% CI 0.24 to 0.65; participants = 3285; studies = 8), Analysis 7.1. Results did not demonstrate heterogeneity (I2 = 0%).
7.1. Analysis.
Comparison 7 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone ‐ RCTs only, Outcome 1 Incidence of PE.
Nine RCTs investigated the role of combined modalities on the incidence of DVT (Cahan 2000; Dickinson 1998; Edwards 2008, Eisele 2007; Sakai 2016; Silbersack 2004; Siragusa 1994; Stannard 1996; Windisch 2011). These trials showed no difference in incidence of DVT; 5.84% (76/1301) in the control group and 2.91% (38/1306) in the combined treatment group, RR 0.49, 95% CI 0.19 to 1.26; participants = 2607; studies = 9), Analysis 7.2. Substantial heterogeneity was present (I2 = 76%).
7.2. Analysis.
Comparison 7 IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone ‐ RCTs only, Outcome 2 Incidence of DVT.
Randomized controlled trials only ‐ IPC plus pharmacological prophylaxis versus IPC plus aspirin
Two RCTs evaluated the role of these combined modalities on the incidence of symptomatic PE (Westrich 2006; Woolson 1991). These trials showed no difference in PE; 1.00% (2/201) in the IPC plus aspirin control group and 0% (0/204) in the IPC plus pharmacological prophylaxis treatment group,RR 0.33, 95% CI 0.03 to 3.17; participants = 405; studies = 2), Analysis 8.1. Results did not demonstrate heterogeneity (I2 = 0%).
8.1. Analysis.
Comparison 8 IPC plus pharmacological prophylaxis versus IPC plus aspirin ‐ RCTs only, Outcome 1 Incidence of PE.
The same trials investigated the role of combined modalities on the incidence of DVT. These showed no difference in DVT; 14.92% (30/201) in the IPC plus aspirin control group and 12.25% (25/204) in the PC plus pharmacological prophylaxis treatment group, RR 0.81, 95% CI 0.50 to 1.33; participants = 405; studies = 2), Analysis 8.2. Results did not demonstrate heterogeneity (I2 = 0%).
8.2. Analysis.
Comparison 8 IPC plus pharmacological prophylaxis versus IPC plus aspirin ‐ RCTs only, Outcome 2 Incidence of DVT.
Exclusion of studies with risk of bias
Assessing the included trials for risk of bias showed a high number of studies at high risk for performance bias and at unclear risk for selection and detection bias. See Figure 2 and Figure 3.
Below we report those analyses that show a change in direction of the effect when high risk of bias studies were excluded from the analyses. No change in direction of effect were identified for the other analyses.
Sensitivity analysis excluding studies at high risk for selection bias (random sequence generation issues: Bigg 1992; Borow 1983; Bradley 1993; Kurtoglu 2003; Sieber 1997; Tsutsumi 2012; Westrich 2005), revealed in Analysis 5.2 that incidence of DVT in orthopedic participants (OR 0.32, 95% CI 0.12 to 0.86) was no longer significant (OR 0.34, 95% CI 0.11 to 1.03).
Sensitivity analyses excluding studies at high risk for selection bias (allocation concealment issues: Bigg 1992; Borow 1983; Bradley 1993; Kurtoglu 2003; Sakai 2016; Sieber 1997; Song 2014; Tsutsumi 2012; Westrich 2005) revealed that Analysis 2.2 (incidence of DVT for the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone (OR 0.42, 95% CI 0.18 to 1.03; P = 0.06) became significant (OR 0.32, 95% CI 0.12 to 0.84; P = 0.02). Analysis 7.2 showed that the incidence of DVT for the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone with RCTs only (RR 0.49, 95% CI 0.19 to 1.26) also became significant (RR 0.37, 95% CI 0.15 to 0.89).
Sensitivity analyses excluding studies with a high or unclear risk for blinding of outcome assessment (detection bias: Bigg 1992; Borow 1983; Cahan 2000; Dickinson 1998; Edwards 2008; Eisele 2007; Ramos 1996; Sakai 2016; Sieber 1997; Silbersack 2004; Siragusa 1994; Song 2014; Tsutsumi 2012; Westrich 2005; Westrich 2006; Woolson 1991), revealed that Analysis 2.1 (incidence of PE for the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone (OR 0.39, 95% CI 0.23 to 0.64; P = 0.0002) was no longer significant (OR 0.32, 95% CI 0.01 to 8.25; P = 0.49) and similarly in Analysis 7.1 for RCTs only (OR 0.33, 95% CI 0.01 to 7.81). In Analysis 2.2 (incidence of DVT for the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone (OR 0.42, 95% CI 0.18 to 1.03; P = 0.06) became significant (OR 0.15, 95% CI 0.04 to 0.62; P = 0.008).
Sensitivity analyses excluding studies with a high risk for attrition bias (incomplete outcome data) (Song 2014; Turpie 2007), revealed that Analysis 1.2 (incidence of DVT for the comparison IPC plus pharmacological prophylaxis versus IPC alone; OR 0.52, 95% CI 0.33 to 0.82; P = 0.005) was no longer significant (OR 0.72, 95% CI 0.41 to 1.26; P = 0.25). A similar change in the level of effect was seen in Analysis 4.2 for the subgroup of non‐orthopedic participants which showed OR 0.62, 95% CI 0.27 to 1.44), Analysis 6.2 (OR 0.89, 95% CI 0.48 to 1.64) and Analysis 7.1 (OR 0.59, 95% CI 0.08 to 4.40) both for RCTs only. However for Analysis 5.1, no studies were left for the non‐orthopedic subgroup after removing Ramos 1996. For Analysis 2.1 the incidence of PE for the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis (OR 0.39, 95% CI 0.23 to 0.64; P = 0.0002) was no longer significant (OR 0.58, 95% CI 0.08 to 4.49; P = 0.60).
Sensitivity analysis excluding studies with a high risk for other bias (Dickinson 1998; Edwards 2008; Sakai 2016) revealed that Analysis 2.2 (incidence of DVT for the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis) became significant (OR 0.20, 95% CI 0.11 to 0.37; I2 = 29%) and so did Analysis 7.2 (RR 0.22, 95% CI 0.06 to 0.79) comparing RCTs only.
Sensitivity analysis excluding studies with a high risk for performance bias (blinding of participants and personnel) was not performed because only two studies were deemed to be of low risk of performance bias (Turpie 2007; Yokote 2011).
Regarding the outcomes of bleeding and major bleeding, sensitivity analyses excluding studies with a high risk for all types of bias was performed only for the comparison of combined modalities versus IPC because the number of studies reporting on bleeding in the remaining comparisons was insufficient to show meaningful results. No changes were observed in the direction of effect of bleeding when excluding studies with a high risk for all types of bias.
Discussion
Summary of main results
Our review showed that combined modalities are more effective than single modalities in reducing the incidence of deep vein thrombosis (DVT) when compared to compression and incidence of pulmonary (PE) when compared to anticoagulation. Our review showed no difference between combined and single modalities in the incidence of PE when compared with compression alone and DVT when compared with anticoagulation alone. For PE this was likely caused by the low number of events and can be attributed to a type II error, i.e. an incorrect retention of the null hypothesis. The latter also applies to the studies that investigated the combination of compression plus anticoagulant with compression plus aspirin. However the quality of evidence for the research that supports these conclusions was judged to be moderate as a result of bias being present in several domains. The addition of an anticoagulant to IPC, however, increased the risk of bleeding compared to IPC alone, a side effect not observed for IPC when added to anticoagulation, as indeed expected for a physical method of thromboprophylaxis. These findings highlight the need to tailor the use of additional pharmacological thromboprophylaxis in patients at low risk for bleeding or those where bleeding does not have catastrophic consequences. This issue deserves further study since the criteria for major bleeding were not uniform across the studies, with some of them reporting on blood loss during the procedures or through the drains nor providing rates for postoperative bleeding.
The mechanism responsible for the improved effectiveness of combined modalities may be attributed to the fact that DVT is a multifactorial process. Virchow in 1856 suggested that venous stasis, coagulopathy and endothelial injury are all causes of VTE (Virchow 1856). By treating the different causes of VTE it is expected to improve efficacy in DVT prevention. Rosendaal more recently extended Virchow's theory by proposing a model of risk factors, which considered the importance of the additive role and interaction of multiple risk factors (multiple hit model) (Rosendaal 1999). Based on the additive role of mechanical and pharmacological modalities, the results of this review suggest that venous stasis and hypercoagulopathy are truly independent risk factors. IPC reduces venous stasis by producing active flow enhancement (Kakkos 2005), and also increases tissue factor pathway inhibitor (TFPI) plasma levels (Chouhan 1999). Unfractionated and low molecular weight heparin inhibit factor X. These totally different mechanisms of action are most likely responsible for the synergy between these two modality types.
Subgroup analysis confirmed the efficacy of adding compression to anticoagulation in DVT prevention in orthopedic participants, known to suffer from venous stasis, and also the efficacy of adding anticoagulation to compression in DVT prevention in non‐orthopedic participants, known to have frequently hypercoagulation due to the presence of sepsis, malignancy or other reasons. However the studies were underpowered regarding the other comparisons and also for the outcome of PE, with the exception of the comparison combined modalities versus anticoagulation in non‐orthopedic participants. Interestingly, some comparisons became significant after removing studies deemed to be high risk for bias, as presented in the results section of this review. Similarly, comparisons for symptomatic DVT were also underpowered and further studies may be required. Interestingly, the subgroup analysis of the comparison IPC plus pharmacological prophylaxis versus pharmacological prophylaxis, restricted to IPCs other than foot IPCs was significantly in favor of the combined modalities, unlike the analysis of all IPCs combined, perhaps because the potentially less effective foot IPCs were not included.
Pulmonary embolism risk‐reduction rates were mostly consistent across the studies with no heterogeneity, perhaps because symptomatic PE is a clinically significant complication. In contrast, some heterogeneity was noted in the results on DVT reduction, particularly in non‐orthopedic participants. This might have been related to the fact that methodological quality of the assessed studies was low, with allocation concealment usually being unclear and performance bias usually being high. An alternative explanation is that the heterogeneity of the included participants who underwent various surgical procedures resulted in a variable risk of DVT. Indeed subgroup analysis by patient characteristic provided results with better heterogeneity.
The results of our review endorse the recommendations of the venous thromboembolism prevention guidelines that high‐risk patients should receive multimodal prophylaxis (Gould 2012; NICE 2009; Nicolaides 2013).
Overall completeness and applicability of evidence
The studies included in this review were carried out in a wide range of patient groups undergoing orthopedic but also urological, cardiothoracic, general surgery neurosurgery, gynecology procedures and trauma patients. Most of the participants had a high‐risk procedure or condition and, therefore, the results of this review are not necessarily applicable to different patient groups, where a much lower risk may reduce the absolute risk reduction with combined modalities.
In an effort to investigate the applicability of combined modalities in orthopedic and non‐orthopedic participants, subgroup analysis was performed, confirming the efficacy of adding compression to anticoagulation in orthopedic patients, and the efficacy of adding anticoagulation to compression in non‐orthopedic patients, results that indicate a need for further research in particular patient populations. Since studies on combined modalities are mostly performed in patients at a high risk for VTE, the absolute benefit that would be observed is expected to be much lower in moderate‐risk patients, calling for cost‐effectiveness calculations and studies.
Additionally, it should be noted that the various IPC types may not have the same effectiveness and should not be used interchangeably, for example foot pumps versus calf or calf and thigh leggings, taking into account the results of Analysis 2.4.
A potential confounding factor in the present review is the concurrent use of elastic stockings, very often used together with IPC.
Also, it should be mentioned that medical (non‐surgical) patients were not included in the present review because such studies were not identified by the literature searches.
Reporting of bleeding outcomes (major and minor bleeding) was not uniform across the studies, with some studies reporting on blood loss during the procedures or through the drains or providing rates for postoperative bleeding. Definitions used were also not uniform. This issue deserves further study.
Quality of the evidence
This review included some 9137 participants who were studied in 22 trials (15 RCTs). This provided a body of evidence to investigate our hypothesis that combined modalities are more effective than their single counterparts. However, risk for performance bias was high in most studies and risk for selection and detection bias was mostly unclear. Nevertheless, the results of the present meta‐analysis update are generally consistent with a low amount of heterogeneity in almost all comparisons.
Using GRADE assessment, the quality of evidence for DVT and PE prevention with combined modalities is considered as being moderate. See Table 1 and Table 2. Regarding PE for the comparison of combined modalities with IPC alone, the quality of the evidence is downgraded due to very few events with only 4/12 studies contributing to the effect estimate leading to imprecision probably because of a type II error. For the comparison of combined modalities with anticoagulation, the quality of the evidence for PE is downgraded due to detection bias as a sensitivity analysis excluding studies at high or unclear risk of detection bias no longer show a reduction in the incidence of PE. Possible explanations for these two observations regarding PE include the elimination of low quality research leading to more accurate albeit negative results and also reduction of the number of studies and events (i.e. reduced the overall power to detect a difference). Since PE is a form of VTE, like DVT, we think that the second explanation is more rational than the first one. For DVT for both comparisons (combined modalities compared with IPC or anticoagulation) the quality of the evidence is downgraded to moderate due to risk of bias.
The quality of the evidence for bleeding and major bleeding for the comparison combined modalities versus IPC is moderate. The quality of the evidence was downgraded for indirectness as definitions of bleeding and the reporting of bleeding outcomes was not uniform across studies. The quality of the evidence for bleeding and major bleeding for the comparison combined modalities versus pharmacological prophylaxis is very low. The quality of the evidence is downgraded for risk of bias due to blinding, indirectness (because the definition of bleeding and reporting of bleeding outcomes was not uniform across studies) and imprecision (due to the small number of participants and few events).
Potential biases in the review process
The review authors have made an enormous effort to identify potential trials for inclusion in the present review. Publication bias still could have limited the validity of our results.
This review set out to assess RCTs and CCTs. Many of the CCTs are old and the reporting of the study methodology is often poor. In addition, patient care and standard practice has changed considerably since then. When assessing the incidence of DVT and PE in RCTs only the overall direction and size of the effects were not affected. This was likely caused by the fact that many CCTs did not contribute to the analysis due to small number of reported events. However, when sufficient RCTs become available to perform meaningful analyses of the planned subgroups we will consider including RCTs only.
The review assessed symptomatic or asymptomatic DVT and symptomatic PE as outcomes. In future updates, data permitting, we will add proximal DVT and clinically important VTE (proximal DVT and symptomatic PE) as additional important outcomes.
In order to be as inclusive as possible and because not all studies reported on the type of IPC devices used, we included all IPC devices. This resulted in including some devices that may no longer be used in some parts of the world.
We performed no formal assessment of side effects of IPC. However, from the included studies we note these were rarely encountered and recorded by the studies. We will look into this in more detail in future updates.
Agreements and disagreements with other studies or reviews
The results presented here agree with previous systematic meta‐analyses on this topic (Ho 2013; Kakkos 2012; Sobieraj 2013; Zareba 2014), which showed that combined modalities are significantly better than single prophylactic modalities. The studies included in these reviews were mostly restricted to a particular patient category or were limited by the fact that they used IPC interchangeably with elastic stockings, which is a limitation when interpretation of the results is attempted.
Authors' conclusions
Implications for practice.
Moderate quality evidence suggests that combining IPC with pharmacological prophylaxis, compared with IPC or pharmacological prophylaxis alone, decreases the incidence of DVT when compared to compression, and incidence of PE when compared to anticoagulation. Moderate quality evidence suggests that there is no difference between combined and single modalities in the incidence of PE when compared with compression alone and DVT when compared with anticoagulation alone. The quality of evidence for incidence of PE and DVTwas downgraded to moderate due to risk of bias in study methodology and imprecision, highlighting the need for further research. Moderate quality evidence suggests the addition of an pharmacological prophylaxis to IPC, increased the risk of bleeding compared to IPC alone, a side effect not observed for IPC when added to pharmacological prophylaxis (very low quality evidence), as expected for a physical method of thromboprophylaxis. The quality of evidence for bleeding was downgraded to moderate or very low due to risk of bias in study methodology and indirectness, highlighting the need for further research.The results of the current review agree with current guideline recommendations for hospitalised patients at risk of developing VTE. More studies on the role of combined modalities in VTE prevention are urgently needed, taking also into account the increased bleeding rates after anticoagulation is added to IPC.
Implications for research.
Most patients who received combined modalities in the studies reviewed were at high risk of developing VTE. Although the relative VTE reduction was large in this patient group, the same cannot be extrapolated for patients at moderate risk. Future studies should address this question and also take into account cost‐effectiveness issues; looking at benefits in terms of reduced hospital stay, rehabilitation, mortality and long‐term complications, for example post‐thrombotic syndrome, which add to the burden of disability in the community in the long term. Since the total number of RCTs evaluated in the current review was relatively small, particularly in certain subgroups, future research using RCTs in other patient groups (such as patients with stroke or medical ICU patients) and confirmatory RCTs are warranted. Nevertheless, cost‐effectiveness for combined modalities has been demonstrated in certain high risk groups by the NICE guidelines (NICE 2009). Cost‐effectiveness analysis should be performed in order to define the impact of this policy on health economics in both high‐risk and moderate‐risk patients.
More studies on the role of combined modalities (as opposed to pharmacological prophylaxis alone) in the prevention of pulmonary embolism are urgently needed.
Future research should also aim to use standardised bleeding criteria such as those defined by the International Society on Thrombosis and Haemostasis (Schulman 2010).
Further research should compare the efficacy of improved single modalities, including more effective schedule changes, with their combinations (Eriksson 2001; Kakkos 2005; King 2007; Eriksson 2008). Only one study in the present review used edoxaban, one of the new direct oral anticoagulants (Sakai 2016). Further research on the effect of combined use of recently introduced, improved prophylactic modalities is justified.
Feedback
Anticoagulant feedback, 14 February 2011
Summary
Feedback received on this review, and other reviews and protocols on anticoagulants, is available on the Cochrane Editorial Unit website at http://www.editorial‐unit.cochrane.org/anticoagulants‐feedback.
What's new
| Date | Event | Description |
|---|---|---|
| 3 May 2016 | New citation required but conclusions have not changed | Search updated. Eleven new studies included, nine new studies excluded and three ongoing studies identified. New author added. Cochrane Risk of bias assessments and 'Summary of findings' table added. Text amended to reflect current Cochrane standards. No change to conclusions. |
| 3 May 2016 | New search has been performed | Search updated. Eleven new studies included, nine new studies excluded and three ongoing studies identified. |
History
Protocol first published: Issue 2, 2005 Review first published: Issue 4, 2008
| Date | Event | Description |
|---|---|---|
| 14 February 2011 | Amended | Link to anticoagulant feedback added. |
| 16 June 2008 | Amended | Converted to new review format. |
Acknowledgements
We would like to thank Dr Janet Wale of the Consumer Network for her contribution to the plain language summary in previous versions of the review.
Appendices
Appendix 1. CRS search strategy
| #1 | MESH DESCRIPTOR Thrombosis | 1211 |
| #2 | MESH DESCRIPTOR Thromboembolism | 880 |
| #3 | MESH DESCRIPTOR Venous Thromboembolism | 220 |
| #4 | MESH DESCRIPTOR Venous Thrombosis EXPLODE ALL TREES | 1978 |
| #5 | (thrombus* or thrombopro* or thrombotic* or thrombolic* or thromboemboli* or thrombos* or embol*):TI,AB,KY | 16259 |
| #6 | MESH DESCRIPTOR Pulmonary Embolism EXPLODE ALL TREES | 719 |
| #7 | (PE or DVT or VTE):TI,AB,KY | 4239 |
| #8 | ((vein* or ven*) near thromb*):TI,AB,KY | 5893 |
| #9 | (blood near3 clot*):TI,AB,KY | 2331 |
| #10 | (pulmonary near3 clot*):TI,AB,KY | 5 |
| #11 | (lung near3 clot*):TI,AB,KY | 4 |
| #12 | MESH DESCRIPTOR Lower Extremity EXPLODE ALL TREES WITH QUALIFIERS BS | 1482 |
| #13 | #1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 | 22204 |
| #14 | MESH DESCRIPTOR Intermittent Pneumatic Compression Devices EXPLODE ALL TREES | 88 |
| #15 | MESH DESCRIPTOR Blood Flow Velocity EXPLODE ALL TREES | 2246 |
| #16 | ((pneumati* or sequential) near5 compres*):TI,AB,KY | 452 |
| #17 | ((calf near4 (inflat* or pump* or compres* or squeez*))):TI,AB,KY | 95 |
| #18 | ((foot near4 (inflat* or pump* or compres* or squeez*))):TI,AB,KY | 68 |
| #19 | ((leg near4 (inflat* or pump* or compres* or squeez*))):TI,AB,KY | 167 |
| #20 | ((circulat* near3 assist*)):TI,AB,KY | 31 |
| #21 | (a‐v impulse):TI,AB,KY | 28 |
| #22 | (av impulse):TI,AB,KY | 7 |
| #23 | (flowtron or revitaleg or presssion or plexipulse):TI,AB,KY | 4 |
| #24 | #14 OR #15 OR #16 OR #17 OR #18 OR #19 OR #20 OR #21 OR #22 OR #23 | 2907 |
| #25 | #13 AND #24 | 569 |
Appendix 2. Clinical trials database searches
Clinicaltrials.gov
66 studies found for: pneumatic compression
WHO trials database
68 records for 65 trials found for: pneumatic compression (45 in NCT and 6 in ISRCTN)
ISRCTN
7 results pneumatic compression
Data and analyses
Comparison 1. IPC plus pharmacological prophylaxis versus IPC alone.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 12 | 3017 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.18, 1.34] |
| 2 Incidence of DVT | 11 | 2934 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.33, 0.82] |
| 3 Incidence of symptomatic DVT | 6 | 2526 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.16, 1.47] |
| 4 Incidence of DVT by foot IPC or other IPC | 11 | 2934 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.33, 0.82] |
| 4.1 foot IPC | 1 | 50 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.2 other IPC | 10 | 2884 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.33, 0.82] |
| 5 Incidence of bleeding | 7 | 2155 | Odds Ratio (M‐H, Fixed, 95% CI) | 5.04 [2.36, 10.77] |
| 6 Incidence of major bleeding | 7 | 2155 | Odds Ratio (M‐H, Fixed, 95% CI) | 6.81 [1.99, 23.28] |
Comparison 2. IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 10 | 3544 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.39 [0.23, 0.64] |
| 2 Incidence of DVT | 11 | 2866 | Odds Ratio (M‐H, Random, 95% CI) | 0.42 [0.18, 1.03] |
| 3 Incidence of symptomatic DVT | 5 | 2312 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.29, 3.54] |
| 4 Incidence of DVT by foot IPC or other IPC | 11 | 2866 | Odds Ratio (M‐H, Random, 95% CI) | 0.42 [0.18, 1.03] |
| 4.1 foot IPC | 4 | 324 | Odds Ratio (M‐H, Random, 95% CI) | 0.40 [0.05, 3.47] |
| 4.2 other IPC | 7 | 2542 | Odds Ratio (M‐H, Random, 95% CI) | 0.39 [0.16, 0.96] |
| 5 Incidence of bleeding | 3 | 244 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.30, 2.14] |
| 6 Incidence of major bleeding | 3 | 244 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.21 [0.35, 4.18] |
Comparison 3. IPC plus pharmacological prophylaxis versus IPC plus aspirin.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 3 | 605 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.03, 3.19] |
| 2 Incidence of DVT | 3 | 605 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.48, 1.42] |
| 3 Incidence of symptomatic DVT | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4 Incidence of bleeding | 3 | 616 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.23 [0.27, 5.53] |
| 5 Incidence of major bleeding | 3 | 616 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.15, 4.17] |
Comparison 4. IPC plus pharmacological prophylaxis versus IPC alone ‐ subgroups.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 12 | 3017 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.18, 1.34] |
| 1.1 Orthopedic patients | 3 | 445 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.2 Non‐orthopedic patients | 9 | 2572 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.18, 1.34] |
| 2 Incidence of DVT | 11 | 2934 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.33, 0.82] |
| 2.1 Orthopedic patients | 3 | 445 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.38, 1.69] |
| 2.2 Non‐orthopedic patients | 8 | 2489 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.41 [0.23, 0.73] |
Comparison 5. IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone ‐ subgroups.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 10 | 3544 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.39 [0.23, 0.64] |
| 1.1 Orthopedic patients | 6 | 732 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.08, 4.49] |
| 1.2 Non‐orthopedic patients | 4 | 2812 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.38 [0.22, 0.63] |
| 2 Incidence of DVT | 11 | 2866 | Odds Ratio (M‐H, Random, 95% CI) | 0.42 [0.18, 1.03] |
| 2.1 Orthopedic patients | 8 | 2605 | Odds Ratio (M‐H, Random, 95% CI) | 0.32 [0.12, 0.86] |
| 2.2 Non‐orthopedic patients | 3 | 261 | Odds Ratio (M‐H, Random, 95% CI) | 1.77 [0.30, 10.58] |
Comparison 6. IPC plus pharmacological prophylaxis versus IPC alone ‐ RCTs only.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 7 | 2023 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.51 [0.09, 2.76] |
| 2 Incidence of DVT | 7 | 2008 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.35, 0.90] |
Comparison 7. IPC plus pharmacological prophylaxis versus pharmacological prophylaxis alone ‐ RCTs only.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 8 | 3285 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.40 [0.24, 0.65] |
| 2 Incidence of DVT | 9 | 2607 | Risk Ratio (M‐H, Random, 95% CI) | 0.49 [0.19, 1.26] |
Comparison 8. IPC plus pharmacological prophylaxis versus IPC plus aspirin ‐ RCTs only.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of PE | 2 | 405 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.03, 3.17] |
| 2 Incidence of DVT | 2 | 405 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.50, 1.33] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Bigg 1992.
| Methods | Study design: controlled clinical trial Method of randomisation: study was planned to be randomized and method of planned randomizations was stated as patient order Concealment of allocation: none stated Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 68, intervention group 32; control group 36 Age (mean, years): intervention group 67; control group 65 Sex: male Inclusion criteria: radical retropubic prostatectomy with bilateral pelvic prostatectomy for clinically localized prostate cancer Exclusion criteria: none stated | |
| Interventions | Intervention group: unfractionated heparin (5000 iu BID, subcutaneously) and sequential compression devices with elastic stockings Control group: sequential compression devices with elastic stockings | |
| Outcomes | Symptomatic PE, confirmed with ventilation‐perfusion scan | |
| Notes | The study was planned to be randomized but due to administrative errors the randomisation protocol was violated Sequential compression devices were started in the operating room and discontinued when the patients were ambulatory, usually 18 hours postoperatively Heparin was started two hours before the operation and was continued for 7 days or the time of discharge Study was discontinued because of bleeding complications associated with heparin use. No specific bleeding definitions were provided |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | The study was planned to be randomized but due to administrative errors the randomization protocol was violated. Method of planned randomizations was stated as patient order |
| Allocation concealment (selection bias) | High risk | Alternating patients received the study medication and in most cases the surgeon was aware of which patients received heparin |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | In most cases the surgeon was aware of which patients received heparin, and the same perhaps applies to the anesthesia personnel |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing the pulmonary ventilation‐perfusion scans or angiograms were aware of which patients received heparin |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No patients were lost to follow‐up |
| Selective reporting (reporting bias) | Low risk | PE was the only VTE event stated in methodology and was reported |
| Other bias | Unclear risk | No baseline characteristics, apart from age, were provided |
Borow 1983.
| Methods | Study design: controlled clinical trial Method of randomization: none Concealment of allocation: not reported Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 272, but only 237 of them were eligible for inclusion based on type of prophylaxis Age (mean, years): not reported Sex: not reported Inclusion criteria: general, surgery, orthopedics, gynecology, and vascular surgery Exclusion criteria: genitourinary surgery | |
| Interventions | Intervention group: sequential compression devices and pharmacological prophylaxis (unfractionated heparin or coumadin) Control group: sequential compression devices or pharmacological prophylaxis (unfractionated heparin or coumadin) | |
| Outcomes | DVT diagnosed with I‐125 fibrinogen scanning, IPG, Doppler ultrasound and venography | |
| Notes | Patients who received aspirin or dextran as an exclusive pharmacological modality or elastic stockings as an exclusive mechanical modality were not included in our review All modalities were started with the preoperative medication and continued until the patients were well ambulatory |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Patients were placed into each category in rotation by the vascular technicians |
| Allocation concealment (selection bias) | High risk | No details on the allocation procedure were provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Placebo medications or devices were not used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing DVT testing were aware of which patients received heparin |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All patients had an event reported |
| Selective reporting (reporting bias) | Low risk | DVT was the only VTE event stated in methodology and was reported |
| Other bias | Unclear risk | No baseline characteristics were provided |
Bradley 1993.
| Methods | Study design: controlled clinical trial Method of randomization: states that patients with an even date of birth were randomized to receive the plantar arteriovenous impulse system on the side to be operated on Concealment of allocation: not reported other than the radiologist who read the venograms was blinded to patient allocation Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: UK Number of participants: 74 Age (mean, years): 70 Sex: not reported Inclusion criteria: unilateral primary THA for osteoarthritis Exclusion criteria: non‐consenting patients | |
| Interventions | Intervention group: unfractionated heparin (5000 iu BID, subcutaneously), graduated compression stockings (TEDs), and pneumatic foot compression on the side to be operated on Control group: unfractionated heparin (5000 iu BID, subcutaneously) and graduated compression stockings (TEDs) | |
| Outcomes | DVT on bilateral lower extremity venography performed postoperative day 12 | |
| Notes | The foot pump started at the beginning of surgery and continued until discharge from the hospital. No details were provided for heparin | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Patients with an even date of birth were randomized to receive IPC |
| Allocation concealment (selection bias) | High risk | Patients with an even date of birth were allocated to receive IPC ‐ allocation therefore predictable |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo device was used |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | The radiologist who read the venograms was blinded to patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All study participants were reported in the results section |
| Selective reporting (reporting bias) | Low risk | DVT was the only VTE outcome event stated in methodology and was reported |
| Other bias | Low risk | Baseline characteristics were comparable |
Cahan 2000.
| Methods | Study design: randomized controlled trial Method of randomization: unclear Concealment of allocation: not reported Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 48 Age (mean, years): 67 Sex: 47 males, 1 female Inclusion criteria: major intra‐abdominal surgical procedures Exclusion criteria: pre‐existing venous disease, history of venous thromboembolism, preoperative or postoperative requirement for systemic anticoagulation (with the exception of the 12 patients undergoing aortic aneurysm repair, who did receive systemic doses of heparin intraoperatively) | |
| Interventions | Intervention group: subcutaneous heparin injections (5000 iu BID) combined with the use of a thigh‐length sequential pneumatic compression device (Kendall Health Care, Manchester, Mass, USA) Control groups: 1. subcutaneous heparin injections (5000 iu BID) 2. use of a thigh‐length sequential pneumatic compression device (Kendall Health Care, Manchester, Mass, USA) |
|
| Outcomes | DVT on duplex ultrasound and also clinically evident DVT and PE | |
| Notes | Investigation on the effect of study interventions on fibrinolytic activity, but also reported VTE outcomes DVT prophylaxis was initiated in the operating room after induction of anesthesia and continued until postoperative day 5 (or discharge, if this occurred sooner). If the patient remained hospitalized after postoperative day 5, DVT prophylaxis was left to the discretion of the primary surgeon, and the patient was no longer participating in the research study |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | Not provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo anticoagulants or IPC devices were used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel who performed the DVT screening were blinded to the treatment regimens |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All study participants were reported in the results section |
| Selective reporting (reporting bias) | Low risk | DVT was the only VTE outcome event stated in methodology and was reported |
| Other bias | Low risk | No significant baseline imbalances |
Dickinson 1998.
| Methods | Study design: randomized controlled trial Method of randomization: unclear Concealment of allocation: not reported Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 66 Age (mean, years): 47.4 (calculated) Sex: not reported Inclusion criteria: patients undergoing surgical treatment of intracranial neoplasms Exclusion criteria: history of DVT or pulmonary embolism, allergy to heparin or other anticoagulant agents, history of surgery or major trauma to the lower extremities, a concurrent condition requiring anticoagulation therapy, cranial base neoplasms and pituitary adenomas | |
| Interventions | Intervention group: Enoxaparin (Lovenox; Rhône‐Poulenc Rorer Pharmaceuticals) subcutaneously at a dose of 30 mg in the anesthesia holding room, and continued at a dose of 30 mg BID combined with thigh‐high SCDs (Kendall), a type of IPC, functioning on the patient before induction of anesthesia Control groups: 1. Enoxaparin subcutaneously at a dose of 30 mg and continued at a dose of 30 mg BID 2. thigh‐high SCDs (Kendall) | |
| Outcomes | DVT on duplex ultrasonography between days 1 and 3, between days 5 and 7, at the wound check appointment between days 10 and 14, and at the 1‐month follow‐up appointment Incidence of adverse events (including bleeding) was assessed by principal investigator by thorough review of medical records. No specific bleeding definitions were provided |
|
| Notes | The IPC devices functioned throughout the surgical procedure and remained on the patient during the postoperative period, until the patient was walking without assistance. If the patient remained nonambulatory, the devices were discontinued at the time of discharge from the Neurosurgery Service Enoxaparin was started in the anesthesia holding room and was discontinued at the time of discharge from the Neurosurgery Service |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | Not provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo anticoagulants or IPC devices were used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel who performed the DVT screening were blinded to the treatment regimens |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All study participants were reported in the results section |
| Selective reporting (reporting bias) | Low risk | DVT was the only VTE outcome event stated in methodology and was reported |
| Other bias | High risk | The trial stopped early (enrolment was planned for 120 subjects) |
Edwards 2008.
| Methods | Study design: randomized controlled trial
Method of randomization: unclear
Concealment of allocation: not reported
Exclusions: 10 consented patients cancelled their surgery; 33 patients were excluded for protocol violations, such as missed ultrasound (n = 9), surgery other than THA or TKA (n = 1), previous history of thrombosis (n = 12), prophylaxis other than LMWH (n = 8), and other protocol deviations (n = 3) Losses to follow up: none Intention‐to‐treat analysis: no |
|
| Participants | Country: USA Number of participants: 320 Age (mean, years): 67.3 (calculated) Sex: 162 females, 115 males Inclusion criteria: patients undergoing total hip or knee replacement Exclusion criteria: not provided | |
| Interventions | Intervention group: Enoxaparin (30 mg BID, starting the morning after surgery for 7 ‐ 8 days) combined with IPC (CECT device, ActiveCare DVT; Medical Compression Systems, Or Akiva, Israel) with calf sleeves Control group: Enoxaparin (30 mg BID, starting the morning after surgery for 7 ‐ 8 days) |
|
| Outcomes | DVT on duplex ultrasonography before discharge and also clinically evident DVT and PE at three months | |
| Notes | IPC was placed on the calves of the patient in the operating room and continued during hospitalisation Enoxaparin was started the morning after surgery and continued for 7 ‐ 8 days | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | Not provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo devices were used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel who performed the DVT screening were blinded to the treatment regimens |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All study participants were reported in the results section |
| Selective reporting (reporting bias) | Low risk | DVT and PE were VTE events stated in methodology and were reported |
| Other bias | High risk | A large number of post‐randomization exclusions |
Eisele 2007.
| Methods | Study design: randomized controlled trial Method of randomization: not stated Concealment of allocation: not reported Exclusions post randomization: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: Germany Number of participants: 1803 Age (mean, years): not reported Sex: not reported Inclusion criteria: total joint arthroplasty (24%); knee ligamentous and meniscal repair; tumor resection; open fixation of traumatic fractures; elective osteotomies to correct deformities of the femur, tibia, foot, and ankle; and to treat high‐impact contusion injuries of the lower extremity, pelvis, abdomen, spine, and chest Exclusion criteria: a surgery location that would interfere with the application of the pneumatic compression calf cuff and existing acute DVT | |
| Interventions | Intervention group: LMWH, certoparin (3000 iu 12 hours pre‐op, 12 post‐op then daily, subcutaneously), compression stockings (18 to 20 mmHg), and rapid‐inflation intermittent pneumatic compression Control group: LMWH, certoparin (3000 iu 12 hours pre‐op, 12 post‐op then daily, subcutaneously), and compression stockings (18 to 20 mmHg) | |
| Outcomes | Symptomatic DVT and DVT on duplex‐color coded ultrasound performed on the day of discharge | |
| Notes | "The DVT prophylaxis regimen was randomly assigned in the operating theatre at the time of completion of surgery and the randomisation was stratified by age." No information on PE was provided Patients in the intermittent pneumatic compression group had the intermittent pneumatic compression system applied to both calves in the recovery room shortly after the completion of surgery. Intermittent pneumatic compression therapy was applied daily during the time that the patient was confined to bed postoperatively, and it was terminated at the time that the patient was able to walk LMWH was started 12 hours preoperatively and continued throughout hospitalization |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided, apart from the information that it was stratified by patient age, so that an assumption that a computer generated sequence or the sealed envelope method was used may be made |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo device was used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel who performed the DVT screening were blinded to the treatment regimens |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All study participants were reported in the results section |
| Selective reporting (reporting bias) | Low risk | DVT was the only VTE event stated in methodology and was reported |
| Other bias | Low risk | Baseline number of risk factors for deep venous thrombosis per patient were comparable |
Kurtoglu 2003.
| Methods | Study design: quasi‐randomized controlled trial Method of randomization: by the last digit of year of birth Concealment of allocation: none Exclusions: not reported Losses to follow up: not reported Intention‐to‐treat analysis: yes | |
| Participants | Country: Turkey Number of participants: 80 Age (mean, years): not provided Sex: not provided Inclusion criteria: trauma patients, at high risk for bleeding Exclusion criteria: low risk for bleeding | |
| Interventions | Intervention group: LMWH (40 mg/day) combined with IPC Control group: IPC | |
| Outcomes | DVT on duplex ultrasonography and clinically evident DVT and PE | |
| Notes | Information on randomization and blinding was obtained from the study authors. No information on start and discontinuation of IPC or LMWH | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Quasi‐randomized trial, randomized by the last digit of year of birth |
| Allocation concealment (selection bias) | High risk | Quasi‐randomized trial so predictable |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo anticoagulants were used |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | The radiologist who performed the ultrasound tests was not aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All study participants were reported in the results section |
| Selective reporting (reporting bias) | Low risk | DVT and PE were the VTE events stated in methodology and results were provided |
| Other bias | Unclear risk | Insufficient details were provided to allow a conclusion to be made |
Ramos 1996.
| Methods | Study design: randomized controlled trial Method of randomization: table of random numbers Concealment of allocation: not reported Exclusions post randomization: intervention group 57; control group 178 Losses to follow up: yes Intention‐to‐treat analysis: no | |
| Participants | Country: USA Number of participants: randomized 2786, completed 2551 Age (mean, years): 63.9 Sex: male 1782; female 769 Inclusion criteria: open heart surgery Exclusion criteria: known prior DVT; bleeding complications; intraoperative death; intolerance to IPC; or withdrawal of prophylaxis before full ambulation | |
| Interventions | Intervention group: unfractionated heparin (5000 iu BID, subcutaneously) and sequential compression devices Control group: unfractionated heparin (5000 iu BID, subcutaneously) | |
| Outcomes | Symptomatic PE, confirmed by ventilation perfusion scan and/or pulmonary angiography | |
| Notes | Both prophylactic methods were started immediately after surgery and continued for 4 to 5 days or until patients were fully ambulatory | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | A table of random numbers was used |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo device was used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing the pulmonary ventilation‐perfusion scans or angiograms were aware of which patients used a compression device |
| Incomplete outcome data (attrition bias) All outcomes | High risk | A large number of patients were excluded after randomization |
| Selective reporting (reporting bias) | Low risk | PE was the only VTE event stated in methodology and was reported |
| Other bias | Low risk | Baseline characteristics were comparable |
Sakai 2016.
| Methods | Study design: randomized controlled trial Method of randomization: computer‐generated sequence Concealment of allocation: sealed envelopes Exclusions post randomization: none Losses to follow up: 2 patients Intention‐to‐treat analysis: no | |
| Participants | Country: Japan Number of participants: randomized 122, completed 120 Age (mean, years): 73.7 Sex: male 20; female 100 Inclusion criteria: patients (aged ≥ 20 years) undergoing knee replacement surgery for primary joint disease including osteoarthritis and rheumatoid arthritis Exclusion criteria: the presence of predefined risk factors for bleeding, coagulation disorders, heart failure (New York Heart Association class III or IV), significant renal dysfunction (creatinine clearance < 30 mL/min), and abnormalities in biochemical measurements (aspartate aminotransferase or alanine aminotransferase ≥ 5 times the upper limit of normal or total bilirubin ≥ 2 times the upper limit of normal). Patients were also excluded if they were scheduled to undergo bilateral joint replacement or reoperation, were unable to walk, or had uncontrolled cardiovascular disease | |
| Interventions | Intervention group: Edoxaban (15 mg or 30 mg OD) and a foot pump (A‐V Impulse System foot pump) Control group: Edoxaban (15 mg or 30 mg OD) |
|
| Outcomes | Symptomatic VTE by postoperative day 28 and asymptomatic DVT on compression ultrasonography on the postoperative day 10 Bleeding: major bleeding was defined as wound hematoma or hemorrhage occurring at a critical site and bleeding required for > 2 units of red blood cell concentrates. Minor bleeding was defined as bleeding that did not fulfil the criteria for major bleeding |
|
| Notes | Both groups also used bilateral knee‐high antithromboembolic stockings The foot pump was activated in the recovery room and used for four days Edoxaban started 12 hours postoperatively and was used for a mean of 11.5 days |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated sequence |
| Allocation concealment (selection bias) | High risk | Sealed enveloped contained the randomization slip, but no statement that these were opaque |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo device was used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | No information was provided on who performed the ultrasound and if that person was blinded to patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Minimal losses to follow‐up |
| Selective reporting (reporting bias) | Low risk | DVT and PE were VTE events stated in methodology and were reported |
| Other bias | High risk | Trial stopped prematurely |
Sieber 1997.
| Methods | Study design: controlled clinical trial Method of randomization: none Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 579 Age (mean, years): 65 Sex: male Inclusion criteria: patients who had pelvic lymphadenectomy with or without radical retropubic prostatectomy Exclusion criteria: none | |
| Interventions | Intervention group: unfractionated heparin (5000 iu BID, subcutaneously) and sequential compression devices Control group: sequential compression devices | |
| Outcomes | Symptomatic DVT or PE | |
| Notes | Participants were assigned to heparin and control groups by the primary surgeon Sequential compressive stockings were placed at the time of surgery and left in place for 48 hours after surgery for all patients Heparin was started preoperatively and continued for three days |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | The type of the study (CCT) makes it high risk for selection bias |
| Allocation concealment (selection bias) | High risk | The type of the study (CCT) makes it high risk for selection bias |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo injection for heparin was given |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No exclusions or withdrawals were reported |
| Selective reporting (reporting bias) | Low risk | DVT and PE were VTE events stated in methodology and were reported |
| Other bias | Unclear risk | Insufficient details were provided to allow a conclusion to be made |
Silbersack 2004.
| Methods | Study design: randomized controlled trial Method of randomization: not reported Concealment of allocation: not reported Exclusions post randomization: 8 Losses to follow up: none Intention‐to‐treat analysis: no | |
| Participants | Country: Germany Number of participants: 139 randomized Age (mean, years): 64 Sex: male 47; female 84 Inclusion criteria: primary unilateral THR or TKR Exclusion criteria: heart failure NYHA class III/IV; stage III chronic renal insufficiency; severe peripheral arterial disease; acute thrombophlebitis; neurological disorders or arthrodeses of the lower limbs; recent anticoagulation; hemorrhagic diathesis; allergy to heparins; or active malignant disease | |
| Interventions | Intervention group: LMWH, enoxaparin (40 mg daily, subcutaneously) and pneumatic sequential compression Control group: LMWH, enoxaparin (40 mg daily, subcutaneously) and class‐I graduated compression stockings | |
| Outcomes | Symptomatic and asymptomatic DVT (on ultrasound) | |
| Notes | The calf cuffs were applied to both lower limbs directly after the operation in the recovery room and the system was activated. The use of the IPC was continued until the tenth postoperative day whenever the patient was in bed Enoxaparin was started the evening before surgery and continued for 30 days |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | No details provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | A placebo device was not used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Color duplex ultrasonography was performed by an independent angiologist who was unaware of the patients' participation in the study or of the method of prophylaxis, but only to confirm the findings of compression ultrasonography, which was not reported to be performed by a blinded or not observer, hence unclear risk of bias |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Eight patients who were randomized were subsequently excluded (two from the LMWH/IPC and six from the LMWH/GCS group) for various reasons, but they represent a small percentage of the total patient number, unlikely to change they results and conclusions whatever their outcome might have been |
| Selective reporting (reporting bias) | Low risk | Thromboembolic (VTE) events were stated in methodology to be the outcome measures of the study and they were reported as such |
| Other bias | Low risk | Baseline characteristics were comparable |
Siragusa 1994.
| Methods | Study design: randomized controlled trial Method of randomization: unclear Concealment of allocation: unclear Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: Italy Number of participants: 70 Age (mean, years): not provided Sex: not provided Inclusion criteria: elective hip replacement Exclusion criteria: not provided | |
| Interventions | Intervention group: IPC + UFH Control group: UFH | |
| Outcomes | DVT on venography | |
| Notes | No information on start and discontinuation of IPC or UFH | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | No details provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | A placebo device was not used |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No exclusions or withdrawals were reported |
| Selective reporting (reporting bias) | Low risk | DVT was the only VTE event stated in methodology and was reported |
| Other bias | Unclear risk | Insufficient details were provided to allow a conclusion to be made |
Song 2014.
| Methods | Study design: randomized controlled trial Method of randomization: computer‐randomized treatment assignments Concealment of allocation: sequential sealed envelopes Exclusions: 15 patients did not have the planned ultrasound scan to detect DVT, although the exact reason for exclusion from the final analysis was provided only for three patients: one came down with heparin‐induced thrombocytopenia, one withdrew informed consent after surgery, and one underwent bypass surgery that led to noncurative operation Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: Korea Number of participants: 220 Age (mean, years): 57.6 Sex: 68.2 % (150) of the patients were male Inclusion criteria: gastric cancer patients with histologically proven adenocarcinoma undergoing surgery Exclusion criteria: history of PE or DVT in the previous 1 year; preoperative prolonged immobilization or being wheelchair bound; diseases of bleeding tendency; major surgery in the previous 6 months; cerebrovascular accident in the previous 3 months; uncontrolled hypertension; congestive cardiac failure; renal or liver impairment; allergy to heparin or heparin‐induced thrombocytopenia; varicose veins or chronic venous insufficiency; previous chemotherapy; radiotherapy; anticoagulation therapy; transfusion; body mass index (BMI) ≤ 18.5 kg/m2; pregnancy or plan to become pregnant | |
| Interventions | Intervention group: IPC combined with enoxaparin 40 mg OD Control group: IPC | |
| Outcomes | DVT on duplex ultrasound but also clinically evident DVT and PE Bleeding: major and minor, no specific bleeding definitions provided |
|
| Notes | Interim analysis The IPC was initiated preoperatively and continued until postoperative discharge Enoxaparin started postoperatively but the exact time of start and discontinuation was not provided |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐randomized treatment assignments |
| Allocation concealment (selection bias) | High risk | Sequential sealed envelopes, but no statement that these were opaque |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo injection was given |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | High risk | A relatively large number of patients in the combined group did not have duplex ultrasonography |
| Selective reporting (reporting bias) | Low risk | DVT and PE were stated in methodology to be the outcome measures of the study and results were reported |
| Other bias | Low risk | Baseline characteristics were comparable |
Stannard 1996.
| Methods | Study design: randomized controlled trial Method of randomization and concealment of allocation: unclear Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 75 Age (mean, years): 67.4 Sex: not reported Inclusion criteria: patients undergoing elective uncemented hip arthroplasty Exclusion criteria: not provided | |
| Interventions | Heparin/aspirin versus intermittent foot compression versus combined heparin/aspirin and intermittent foot compression | |
| Outcomes | Asymptomatic DVT, symptomatic DVT, any DVT, PE | |
| Notes | The pumps were started in the recovery room immediately after surgery and used until the end of the study, with the exact time not specified Heparin was started 8 hours before the operation and after 3 days of use it was replaced with 325 mg aspirin twice daily for an undefined duration |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | No details provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo for compression, heparin and aspirin |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | All duplex results and venograms were read by one of the authors who was blinded to the prophylactic modality used |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No patients were lost to follow‐up |
| Selective reporting (reporting bias) | Low risk | DVT was stated in methodology to be the outcome measure of the study and results were reported |
| Other bias | Low risk | Baseline characteristics were comparable |
Tsutsumi 2012.
| Methods | Study design: controlled clinical trial Method of randomization: none Concealment of allocation: not reported Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: Japan Number of participants: 137 Age (mean, years): 66.1 (calculated) Sex: 83 men, 54 women Inclusion criteria: patients with colorectal cancer undergoing elective resection surgery under general anesthesia, regardless of tumor stage Exclusion criteria: clinical signs of DVT, active bleeding, active GI ulceration, hemorrhagic stroke, contraindication for anticoagulation, indwelling epidural catheter, renal failure and inability to receive intermittent pneumatic compression | |
| Interventions | Intervention group: IPC (stopped 24 hours after surgery) combined with fondaparinux (subcutaneous injections of fondaparinux at 2.5 mg OD) Control group: IPC (stopped 24 hours after surgery) | |
| Outcomes | Clinically evident DVT and PE Bleeding: major bleeding was defined as bleeding that was fatal, retroperitoneal, intracranial, involving any other critical organ, led to intervention being discontinued, or was associated with a need for transfusion of more than 3 units of packed red blood cell. Other types of bleeding was included and defined as bleeding that did not fulfil the criteria for major bleeding |
|
| Notes | IPC was used for 24 hours after surgery, but no information on when it was started Fondaparinux was started 24 hours after surgery and was continued until days 5‐7 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | The type of the study (CCT) makes it high risk for selection bias |
| Allocation concealment (selection bias) | High risk | The type of the study (CCT) makes it high risk for selection bias |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No placebo for fondaparinux |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All enrolled patients had results reported |
| Selective reporting (reporting bias) | Low risk | Thromboembolic events (DVT and PE) were stated in methodology to be the outcome measures of the study and they were reported as such |
| Other bias | Low risk | Baseline characteristics were comparable |
Turpie 2007.
| Methods | Study design: randomized, double‐blind, placebo‐controlled, superiority trial Method of randomization: centralized computer‐generated schedule (1:1 randomization in blocks of four and stratified by centre) Concealment of allocation: yes Exclusions post randomization: 24 Losses to follow up: none Intention‐to‐treat analysis: no | |
| Participants | Country: USA Number of participants: 1309 randomized, 1285 randomized and treated Age: median age 59 and 60 years in the control and treatment groups, respectively Sex: male 635; female 650 Inclusion criteria: abdominal surgery expected to last longer than 45 min in patients aged over 40 years; or patients weighing over 50 kg Exclusion criteria: vascular surgery with evidence of leg ischemia caused by peripheral vascular disease; unable to receive intermittent pneumatic compression or elastic stockings; pregnant women and women of childbearing age not using effective contraception; life‐expectancy < 6 months; clinical signs of DVT and/or history of venous thromboembolism within the previous 3 months; active bleeding; documented congenital or acquired bleeding disorder; active ulcerative gastrointestinal disease (unless it was the reason for the present surgery); hemorrhagic stroke or surgery on the brain, spine or eyes within the previous 3 months; bacterial endocarditis or other contraindications for anticoagulant therapy; planned indwelling intrathecal or epidural catheter for more than 6 hours after surgical closure; unusual difficulty in achieving epidural or spinal anesthesia; known hypersensitivity to fondaparinux or iodinated contrast medium; current addictive disorders; serum creatinine concentration above 2.0 mg/dL in a well‐hydrated patient; platelet count below 100 000 mm; or patients requiring anticoagulant therapy or other pharmacologic prophylaxis besides intermittent pneumatic compression | |
| Interventions | Intervention group: fondaparinux and intermittent pneumatic compression Control group: intermittent pneumatic compression | |
| Outcomes | Venous thromboembolism (defined as DVT detected by mandatory screening and/or documented symptomatic DVT or PE, or both) and individual components up to day 10. Symptomatic venous thromboembolism up to day 10 and day 32 Major bleeding (defined as bleeding that was fatal, retroperitoneal, intracranial, or involved any other critical organ, led to intervention being discontinued, or was associated with a bleeding index of 2.0 or more) detected during the treatment period Death during the treatment period and up to day 32 |
|
| Notes | Study medications were packaged in boxes of identical appearance
Of the 1309 randomized patients, 842 (64.3%) had an evaluable venogram performed and were included in the primary efficacy analysis
Major bleeding occurred in 10 patients (1.6%) and 1 patient (0.2%) of the intervention and control groups, respectively (P = 0.006) During the on‐study‐drug period of 5–9 days, all patients were to receive venous thromboembolism prophylaxis with intermittent pneumatic compression using any type of device, except a foot pump, for a duration left to the investigator's discretion. The first injection of fondaparinux or placebo was scheduled 6–8 h after surgical closure, provided that hemostasis was achieved. The duration of the on‐study‐drug period was 5–9 days. If the patient was discharged from hospital before completing the on‐study‐drug period, a visiting nurse administered the remaining trial injections |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Centralized computer‐generated schedule randomization (1:1 randomization in blocks of four and stratified by centre) |
| Allocation concealment (selection bias) | Low risk | Centralized computer‐generated schedule randomization |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Use of placebo injections |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Reports double‐blind (use of placebo injections) but it is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | High risk | A large number of exclusions in both trial arms, around 35% of the total number of participants, mainly because of lack of mandatory or interpretable venography |
| Selective reporting (reporting bias) | Low risk | DVT and PE were the primary efficacy outcomes and they were reported in the results |
| Other bias | Low risk | Demographic variables and risk factors at baseline, type of anesthesia, and type and duration of surgery were similar in the two groups among both randomized and treated patients (Tables 1 and 2) and among patients analysed for primary efficacy |
Westrich 2005.
| Methods | Study design: controlled clinical trial Method of randomization: none Concealment of allocation:: none Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 200 Age (mean, years): 81.3 Sex: male 42; female 158 Inclusion criteria: patients older than 60 years who sustained a fragility fracture to the hip; and an ability and willingness to comply with the mechanical and chemical prophylaxis protocol Exclusion criteria: patients younger than 60 years; history of severe allergy to aspirin or warfarin; refusal to use the pneumatic compression device; multiple trauma injuries; or patients with a hip fracture that did not require surgical treatment | |
| Interventions | Intervention group: pneumatic sequential compression and warfarin Control group: pneumatic sequential compression and aspirin | |
| Outcomes | DVT on ultrasound of the ipsilateral lower external iliac, common femoral, superficial femoral, deep femoral, and popliteal veins Bleeding: all participants assessed for postoperative bleeding, no specific bleeding definition provided | |
| Notes | No symptomatic VTE was observed Three patients on warfarin developed bleeding complications The IPC device was applied over the duration of the patient's preoperative and postoperative stay until the time of discharge. Patients sent to a rehabilitation center were told to continue using the IPC until their final discharge home. Warfarin or aspirin started on the night before surgery but no duration of use was provided |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | The type of the study (CCT) makes it high risk for selection bias |
| Allocation concealment (selection bias) | High risk | The type of the study (CCT) makes it high risk for selection bias |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Not a double‐blind study |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No exclusions/participants lost to follow‐up |
| Selective reporting (reporting bias) | Low risk | DVT and PE were the main study outcomes and they were reported in the results |
| Other bias | Unclear risk | Insufficient details were provided to allow a conclusion to be made |
Westrich 2006.
| Methods | Study design: randomized controlled trial Method of randomization: not reported Concealment of allocation: not reported Exclusions post randomization: 11 Losses to follow up: 73 Intention‐to‐treat analysis: no | |
| Participants | Country: USA Number of participants: 275 Age (mean, years): 69 Sex: male 99; female 176 Inclusion criteria: unilateral TKA Exclusion criteria: allergies to aspirin; congenital or acquired bleeding disorders; active ulcerative or angiodysplastic gastrointestinal disease; multiple myeloma or other paraproteinemias; pheochromocytoma; hyperthyroidism; impaired renal function; known hepatic disease; past medical history of stroke; recent brain, spinal, or ophthalmologic surgery; hypersensitivity to enoxaparin; cardiac complications; severe peripheral vascular diseases; chronic heart failure; severe varicose veins; history of DVT and/or PE | |
| Interventions | Intervention group: pneumatic sequential compression and enoxaparin Control group: pneumatic sequential compression and aspirin | |
| Outcomes | DVT on ultrasound before discharge on postoperative days 3 to 5, and 4 to 6 weeks after surgery | |
| Notes | Bleeding complications were documented, no specific bleeding definitions provided Upon their arrival in the recovery room, the patients received a VenaFlow calf compression device that was placed on both of their lower extremities. The compression device was used during each patient's entire hospital stay Enoxaparin was initiated 2 hours after epidural catheter removal (approx. 48 hours postoperatively). Patients received 30 mg of enoxaparin twice daily until their hospital discharge; upon discharge, their dosage was changed to 40 mg once daily for 3 weeks. Aspirin started on the night of their surgery in the recovery room and was continued for 4 weeks postoperatively |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | No details provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Not a blinded trial |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | High risk | A large number of patients were lost to follow‐up, likely to affect outcome results |
| Selective reporting (reporting bias) | Low risk | DVT was the main study outcome and was reported in the results |
| Other bias | Low risk | Baseline characteristics were comparable |
Windisch 2011.
| Methods | Study design: randomized controlled trial Method of randomization: not provided Concealment of allocation: not provided Exclusions: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: Germany Number of participants: 80 Age (mean, years): 68.5 (calculated) Sex: not provided Inclusion criteria: patients undergoing total knee replacement (primary diagnosis of knee "arthritis") Exclusion criteria: patients aged younger than 60 years, body mass index (BMI) > 40 or < 25, existing acute DVT, thrombophlebitic varicosis (stages II–IV acc. Marshall), venous insufficiency (stages 2–3 according to Widmer) | |
| Interventions | Intervention group: IPC (foot pump) combined with enoxaparin (40 mg OD, beginning 24 hours prior to the operation) Control group: enoxaparin (40 mg OD, beginning 24 hours prior to the operation) | |
| Outcomes | DVT on duplex ultrasonography, but also clinically evident DVT and PE | |
| Notes | Reports none of the participants needed to be operated upon for hemarthrosis, no other details regarding bleeding were provided The AVI system was attached in the recovery room to both feet of the participants only shortly after completion of the operation; patients were free to discontinue its use at will Enoxaparin was started 24 hours before surgery, duration was not provided |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | No details provided |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | A placebo device was not used |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Sonographers were unaware of treatment allocations |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No exclusions/patients lost to follow‐up |
| Selective reporting (reporting bias) | Low risk | DVT and PE were VTE events stated in methodology and were reported |
| Other bias | Low risk | There were no baseline imbalances |
Woolson 1991.
| Methods | Study design: randomized controlled trial Method of randomization: sealed envelopes Concealment of allocation: sealed envelopes Exclusions post randomization: none Losses to follow up: none Intention‐to‐treat analysis: yes | |
| Participants | Country: USA Number of participants: 196 patients who had 217 procedures Age (mean, years): 65 Sex: male 95 procedures; female 122 procedures Inclusion criteria: primary or revision THA Exclusion criteria: allergy to aspirin or warfarin; recent peptic ulcer or other bleeding diathesis; receiving any drug that affects platelet function within two weeks before the operation; or patients expected to remain in bed for more than four days after the operation | |
| Interventions | Intervention group: pneumatic sequential compression, thigh‐high graduated elastic compression stockings, and warfarin (one group); or pneumatic sequential compression, thigh‐high graduated elastic compression stockings, and aspirin (second group) Control group: pneumatic sequential compression and thigh‐high graduated elastic compression stockings | |
| Outcomes | Proximal DVT on venography, B‐mode ultrasonography, or both, on discharge Symptomatic DVT or PE, objectively diagnosed | |
| Notes | Warfarin dose was 7.5 or 10 mg orally on the evening before the operation, then titrated to maintain the prothrombin time at 1.2 to 1.3 times the control value. Aspirin started the evening before surgery and continued at a dose of 650 mg twice daily. For both agents duration of use was not reported IPC was started in the operating theater, as soon as the patient was draped and used discharge Follow up was at least 3 months for all patients Bleeding: one patient in each of the three groups had a wound hematoma, that required evacuation in the two intervention group patients but not in the control group. No specific definition of bleeding provided No complications related to the use of the elastic stockings or pneumatic compression were reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Use of sealed envelope method |
| Allocation concealment (selection bias) | Unclear risk | Does not mention if the sealed envelopes were sequentially numbered and opaque |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Not a blinded trial |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | It is unclear if the personnel performing diagnostic testing were aware of patient allocation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No patients were lost to follow‐up |
| Selective reporting (reporting bias) | Low risk | DVT was the main study outcome and was reported in the results |
| Other bias | Low risk | There were no baseline imbalances |
Yokote 2011.
| Methods | Study design: randomized controlled trial Method of randomization: not provided Concealment of allocation: not provided Exclusions: none Losses to follow up: 3 patients withdrawn after randomization Intention‐to‐treat analysis: yes | |
| Participants | Country: Japan Number of participants: 255 Age (mean, years): 63.3 (calculated) Sex: 204 females and 46 males Inclusion criteria: elective primary unilateral total hip replacement Exclusion criteria: bilateral and revision procedures, patients who were less than 20 years of age, long‐term anticoagulation treatment such as unfractionated heparin, LMWH, vitamin K antagonists, antiplatelet agents for pre‐existing cardiac or cerebrovascular disease, a history of VTE, a coagulation disorder including antiphospholipid syndrome, the presence of a solid malignant tumor or a peptic ulcer, and major surgery in the preceding three months. Caucasian patients were also excluded | |
| Interventions | Intervention group: 1. Enoxaparin (20 mg BID) + IPC 2. Fondaparinux (2.5 mg OD) + IPC Control group: placebo + IPC |
|
| Outcomes | DVT on duplex ultrasonography and also clinically evident DVT and PE Any bleeding, both major or minor. Major bleeding: retro‐peritoneal, intracranial or intraocular, or if associated with either death, transfusion of more than two units of packed red blood cells or whole blood (except autologous), a reduction in the level of hemoglobin of > 2 g/dL, or a serious or life‐threatening clinical event requiring medical intervention. Suspected intra‐abdominal or intracranial bleeding was confirmed by ultrasonography, CT or MRI Minor bleeding: epistaxis lasting for more than five minutes or requiring intervention, ecchymosis or hematoma with a maximum size of > 5 cm, hematuria not associated with trauma from the urinary catheter, gastrointestinal hemorrhage not related to intubation or the passage of a nasogastric tube, a wound hematoma or hemorrhagic wound complications not associated with major hemorrhage or subconjunctival hemorrhage, requiring cessation of medication |
|
| Notes | The pneumatic devices were initiated in the operating theater (before surgery for the contralateral leg and just after surgery for the operated leg) and removed on the "second post‐operative day when the day of surgery was defined as post‐operative day 1" Pharmacological prophylaxis was started postoperatively |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No details provided |
| Allocation concealment (selection bias) | Unclear risk | No details provided |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Use of placebo |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Scans were read by experiences radiologist blinded to randomisation |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | A small percentage of exclusions (5/255, 2%) |
| Selective reporting (reporting bias) | Low risk | DVT and PE were the main study outcomes and they were reported in the results |
| Other bias | Low risk | Baseline characteristics were comparable |
BID: twice daily DVT: deep vein thrombosis IPC: intermittent pneumatic compression IPG: impedence plethysmography iu: international units LMWH: low molecular weight heparin mg: milligrams NYHA: New York Hospital Association OD: once daily PE: pulmonary embolism THA: total hip arthroplasty THR: total hip replacement TKR: total knee replacement
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Ailawadi 2001 | Retrospective case‐control study |
| Eskander 1997 | Use of combined modalities was not concurrent in the intervention group |
| Frim 1992 | Controlled before and after study |
| Gagner 2012 | Registry study, non‐randomized |
| Gelfer 2006 | Pharmacological prophylaxis was not the same in the two study groups |
| Kamran 1998 | Controlled before and after study |
| Kiudelis 2010 | Investigation restricted to intraoperative period up to 10 min after extubation |
| Kumaran 2008 | The control (single modality) group included patients who were allocated to heparin or pneumatic compression |
| Lieberman 1994 | Pharmacological prophylaxis consisted of aspirin, which has limited thromboprophylactic properties |
| Macdonald 2003 | Pharmacological prophylaxis was not the same in the two study groups |
| Mehta 2010 | Only aggregated VTE rates and not separate DVT and PE rates were provided and the authors did not reply when individual data were requested |
| Nathan 2006 | Prospective case‐control study |
| Patel 2010 | Retrospective study |
| Roberts 1975 | Pneumatic compression was used only intraoperatively |
| Spinal cord injury investigators | Pharmacological prophylaxis was not the same in the two study groups |
| Stannard 2006 | Use of enoxaparin was not concurrent in the two study groups |
| Tsutsumi 2000 | Controlled before and after study |
| Wan 2015 | Retrospective study |
| Westrich 1996 | Pharmacological prophylaxis consisted of aspirin, which has limited thromboprophylactic properties |
| Whitworth 2011 | Retrospective case‐control study investigating preoperative anticoagulation in patients on postoperative low molecular‐weight heparin and SCDs |
| Winemiller 1999 | Retrospective case‐control study |
DVT: deep vein thrombosis PE: pulmonary embolism SCD: sequential compression device VTE: venous thromboembolism
Characteristics of ongoing studies [ordered by study ID]
CHICTR‐IPR‐15007324.
| Trial name or title | The mechanical and medical prevention of lower extremity deep venous thrombosis formation post gynecologic pelvic surgery, a multiple center randomized case control study |
| Methods | Randomized parallel controlled trial |
| Participants | Women undergoing gynecologic pelvic surgery |
| Interventions | GCS: graduated compression stockings; GCS + LMWH: graduated compression stockings + low molecular weight heparin; GCS + IPC: graduated compression stockings + intermittent pneumatic compression; GCS + LMWH + IPC: graduated compression stockings + low molecular weight heparin + intermittent pneumatic compression |
| Outcomes | DVT on ultrasound of the leg veins (primary); hemoglobin; white blood cell count; hematocrit; platelets; PT; APTT; Fbg; TT; D‐Dimer; AT‐III; t‐PA; PAI; VIII factor; X factor; Protein c; Protein s; CTPA (all secondary) |
| Starting date | November 2015 |
| Contact information | Cuiqin Sang, 22 South Sanlitun Road, Beijing, China |
| Notes | Target sample size: GCS: 250; GCS + LMWH: 250; GCS + IPC: 250; GCS + LMWH + IPC: 250 |
ISRCTN44653506 and NCT02040103.
| Trial name or title | The PREVENT Trial: pneumatic compression for PREventing VENous Thromboembolism |
| Methods | RCT in ICU patients already receiving anticoagulants |
| Participants | No further information is provided |
| Interventions | Patients were randomized to use IPC or not |
| Outcomes | Incidence of proximal leg DVT up to 30 days (primary), PE up to 30 days (secondary), ICU and hospital mortality (secondary) |
| Starting date | December 2013 |
| Contact information | Dr Yaseen Arabi |
| Notes | Trial completed, no longer recruiting |
NCT00740987.
| Trial name or title | Efficacy of the association mechanical prophylaxis plus anticoagulant prophylaxis on venous thromboembolism incidence in intensive care unit (ICU) (CIREA2) |
| Methods | RCT in ICU patients without high risk of bleeding |
| Participants | 621 ICU patients |
| Interventions | Patients were randomized to use IPC or not |
| Outcomes | Primary outcome measures: combined criterion evaluated at day 6 ± 2 days after randomization: symptomatic venous thromboembolic event, non‐fatal, objectively confirmed; death related to PE; asymptomatic DVT of the lower limbs detected by CUS on day 6 (time frame: 6 ± 2 days) Secondary outcome measures: symptomatic thromboembolic events occurred between day 6 and day 90; total mortality evaluated at 1 month and 3 months (time frame: 6 days to 3 months) |
| Starting date | October 2007 |
| Contact information | Karine Lacut, MD. CHU Brest France, Univ Brest, EA 3878 |
| Notes | Study completed in January 2015, with no results being presented or published at the time of writing this review |
APTT: activated partial thromboplastin time CTPA: computed tomography pulmonary angiogram CUS: colour ultrasound DVT: deep vein thrombosis Fbg: fibrinogen GCS: graduated compression stockings ICU: intensive care unit IPC: intermittent pneumatic compression LMWH: low molecular weight heparin PAI: plasminogen activator inhibitor PE: pulmonary embolism PT: prothrombin time RCT: randomized controlled trial TT: thrombin time t‐PA: tissue plasminogen activator
Differences between protocol and review
The outcomes incidence of bleeding, incidence of major bleeding, and fatal bleeding are important adverse events of pharmacological prophylaxis and have therefore been added to the review.
The outcome fatal PE has been added to the review for completeness.
The method of evaluating study quality has changed since the protocol was published; we used the Cochrane 'Risk of bias' tool (Higgins 2011). We have also added 'Summary of findings' tables.
Because risk stratification of study participants was not provided nor based on modern or any methodology, all types of participants were included and not only those considered as being at high risk of developing VTE; however many studies included in this review included high risk patients such as those undergoing orthopedic surgery.
Contributions of authors
SKK: selected trials, assessed trial quality, and extracted data JC: arbitrated disagreements GG: selected trials, assessed trial quality, and extracted data AN: contributed to the text of the review GS: contributed to the text of the review DR: contributed to the text of the review IN: selected trials, assessed trial quality, and extracted data
Sources of support
Internal sources
No sources of support supplied
External sources
-
Chief Scientist Office, Scottish Government Health Directorates, The Scottish Government, UK.
The Cochrane Vascular editorial base is supported by the Chief Scientist Office.
-
National Institute for Health Research (NIHR), UK.
This project was supported by the NIHR, via Cochrane Incentive Award funding (15/81/10) to Cochrane Vascular. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Declarations of interest
SKK: has declared that he has received consulting fees from Covidien (small lecture fees from companies now taken over and incorporated by Covidien), Sanofi (research funding and travel grants), LEO (research funding), Bayer (research funding and travel grants), all unrelated to the subject of this review JC: has declared that he received consultancy fees from Janssen Research and Development (meeting to discuss protocol), Pfizer (meeting to discuss bleeding issues) and BMS Medtronics (advisory board meeting consultant). He received payment for lectures from Sanofi Poland (lecture to Vascular surgical society) and CHEST enterprises (lectures to Pharma group concerning guidelines). These were unrelated to the subject of this review GG: none known AN: none known GS: none known DR: none known IN: none known
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Bigg 1992 {published data only}
- Bigg SW, Catalona WJ. Prophylactic mini‐dose heparin in patients undergoing radical retropubic prostatectomy. A prospective trial. Urology 1992;39(4):309‐13. [DOI] [PubMed] [Google Scholar]
Borow 1983 {published data only}
- Borow M, Goldson HJ. Prevention of postoperative deep venous thrombosis and pulmonary emboli with combined modalities. American Surgeon 1983;49(11):599‐605. [PubMed] [Google Scholar]
Bradley 1993 {published data only}
- Bradley JG, Krugener GH, Jager HJ. The effectiveness of intermittent plantar venous compression in prevention of deep venous thrombosis after total hip arthroplasty. Journal of Arthroplasty 1993;8(1):57‐61. [DOI] [PubMed] [Google Scholar]
Cahan 2000 {published data only}
- Cahan MA, Hanna DJ, Wiley LA, Cox DK, Killewich LA. External pneumatic compression and fibrinolysis in abdominal surgery. Journal of Vascular Surgery 2000;32(3):537‐43. [DOI] [PubMed] [Google Scholar]
Dickinson 1998 {published data only}
- Dickinson LD, Miller LD, Patel CP, Gupta SK. Enoxaparin increases the incidence of postoperative intracranial hemorrhage when initiated preoperatively for deep venous thrombosis prophylaxis in patients with brain tumors. Neurosurgery 1998;43(5):1074–81. [DOI] [PubMed] [Google Scholar]
Edwards 2008 {published data only}
- Edwards JZ, Pulido PA, Ezzet KA, Copp SN, Walker RH, Colwell CW Jr. Portable compression device and low‐molecular‐weight heparin compared with low‐molecular‐weight heparin for thromboprophylaxis after total joint arthroplasty. Journal of Arthroplasty 2008;23(8):1122‐7. [DOI] [PubMed] [Google Scholar]
Eisele 2007 {published data only}
- Eisele R, Kinzl L, Koelsch T. Rapid‐inflation intermittent pneumatic compression for prevention of deep venous thrombosis. Journal of Bone and Joint Surgery ‐ American Volume 2007;89(5):1050‐6. [DOI] [PubMed] [Google Scholar]
Kurtoglu 2003 {published data only}
- Kurtoglu M, Aksoy M, Genc FA, Baktiroglu S. The efficacy of intermittent pneumatic compression devices in combination with or without low molecular weight heparin in the prophylaxis of venous thromboembolism in patients with trauma. Phlebology 2003;18(3):148. [Google Scholar]
- Yanar H, Kurtoglu M, Taviloglu K, Guloglu R, Ertekin C. Does intermittent pneumatic compression make low molecular weight heparin more efficient in the prophylaxis of venous thromboembolism in trauma patients. European Journal of Trauma and Emergency Surgery 2007;33(Suppl II):79‐80. [Google Scholar]
Ramos 1996 {published data only}
- Ramos R, Salem BI, Pawlikowski MP, Coordes C, Eisenberg S, Leidenfrost R. The efficacy of pneumatic compression stockings in the prevention of pulmonary embolism after cardiac surgery. Chest 1996;109(1):82‐5. [DOI] [PubMed] [Google Scholar]
Sakai 2016 {published data only}
- Sakai T, Izumi M, Kumagai K, Kidera K, Yamaguchi T, Asahara T, et al. Effects of a foot pump on the incidence of deep vein thrombosis after total knee arthroplasty in patients given edoxaban: a randomized controlled study. Medicine (Baltimore) 2016;95(1):e2247. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sieber 1997 {published data only}
- Sieber PR, Rommel FM, Agusta VE, Breslin JA, Harpster LE, Huffnagle HW, Stahl C. Is heparin contraindicated in pelvic lymphadenectomy and radical prostatectomy?. Journal of Urology 1997;158(3 pt1):869‐71. [DOI] [PubMed] [Google Scholar]
Silbersack 2004 {published data only}
- Silbersack Y, Taute BM, Hein W, Podhaisky H. Prevention of deep‐vein thrombosis after total hip and knee replacement. Low‐molecular‐weight heparin in combination with intermittent pneumatic compression. Journal of Bone and Joint Surgery ‐ British Volume 2004;86(6):809‐12. [DOI] [PubMed] [Google Scholar]
Siragusa 1994 {published data only}
- Siragusa S, Vicentini L, Carbone S, Barone M, Beltrametti C, Piovella F. Intermittent pneumatic leg compression (IPLC) and unfractionated heparin (UFH) in the prevention of post‐operative deep vein thrombosis in hip surgery: a randomized clinical trial. British Journal of Haematology 1994;87(Suppl 1):186. [Google Scholar]
Song 2014 {published data only}
- Song KY, Yoo HM, Kim EY, Kim JI, Yim HW, Jeon HM, et al. Optimal prophylactic method of venous thromboembolism for gastrectomy in Korean patients: an interim analysis of prospective randomized trial. Annals of Surgical Oncology 2014;21(13):4232‐8. [DOI] [PubMed] [Google Scholar]
Stannard 1996 {published data only}
- Stannard JP, Harris RM, Bucknell AL, Cossi A, Ward J, Arrington ED. Prophylaxis of deep venous thrombosis after total hip arthroplasty by using intermittent compression of the plantar venous plexus. American Journal of Orthopedics 1996;25:127–34. [PubMed] [Google Scholar]
Tsutsumi 2012 {published data only}
- Tsutsumi S, Yajima R, Tabe Y, Takaaki T, Fujii F, Morita H, et al. The efficacy of fondaparinux for the prophylaxis of venous thromboembolism after resection for colorectal cancer. Hepatogastroenterology 2012;59:2477‐9. [DOI] [PubMed] [Google Scholar]
Turpie 2007 {published data only}
- Priore G, Herzog TJ, Bauer KA, Caprini JA, Comp P, Gent M, et al. Fondaparinux combined with intermittent pneumatic compression (IPC) versus IPC alone in the prevention of venous thromboembolism after major abdominal surgery: results of the APOLLO study. Gynecologic Oncology 2006;101:S21. [Google Scholar]
- Turpie AGG, Bauer KA, Caprini JA, Comp PC, Gent M, Muntz JE, on behalf of the APOLLO Investigators. Fondaparinux combined with intermittent pneumatic compression vs. intermittent pneumatic compression alone for prevention of venous thromboembolism after abdominal surgery: a randomized, double‐blind comparison. Journal of Thrombosis and Haemostasis 2007;5(9):1854‐61. [DOI] [PubMed] [Google Scholar]
- Turpie AGG, Bauer KA, Caprini JA, Comp PP, Gent M, Muntz JE. Fondaparinux combined with intermittent pneumatic compression (IPC) versus IPC alone in the prevention of venous thromboembolism after major abdominal surgery: the randomized APOLLO study. Blood 2005;106(11):Abstract 279. [Google Scholar]
Westrich 2005 {published data only}
- Westrich GH, Rana AJ, Terry MA, Taveras NA, Kapoor K, Helfet DL. Thromboembolic disease prophylaxis in patients with hip fracture: a multimodal approach. Journal of Orthopaedic Trauma 2005;19(4):234‐40. [DOI] [PubMed] [Google Scholar]
Westrich 2006 {published data only}
- Westrich GH, Bottner F, Windsor RE, Laskin RS, Haas SB, Sculco TP. VenaFlow plus Lovenox vs VenaFlow plus aspirin for thromboembolic disease prophylaxis in total knee arthroplasty. Journal of Arthroplasty 2006;21(6 Suppl 2):139‐43. [DOI] [PubMed] [Google Scholar]
Windisch 2011 {published data only}
- Windisch C, Kolb W, Kolb K, Grützner P, Venbrocks R, Anders J. Pneumatic compression with foot pumps facilitates early postoperative mobilisation in total knee arthroplasty. International Orthopaedics 2011;35(7):995‐1000. [DOI] [PMC free article] [PubMed] [Google Scholar]
Woolson 1991 {published data only}
- Woolson ST, Watt JM. Intermittent pneumatic compression to prevent proximal deep venous thrombosis during and after total hip replacement. A prospective, randomized study of compression alone, compression and aspirin, and compression and low‐dose warfarin. Journal of Bone and Joint Surgery ‐ American Volume 1991;73(4):507‐12. [PubMed] [Google Scholar]
Yokote 2011 {published data only}
- Yokote R, Matsubara M, Hirasawa N, Hagio S, Ishii K, Takata C. Is routine chemical thromboprophylaxis after total hip replacement really necessary in a Japanese population?. Journal of Bone and Joint Surgery. British Volume 2011;93(2):251‐6. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Ailawadi 2001 {published data only}
- Ailawadi M, Priore G. A comparison of thromboembolic prophylaxis in gynecologic oncology patients. International Journal of Gynecological Cancer 2001;11(5):354‐8. [DOI] [PubMed] [Google Scholar]
Eskander 1997 {published data only}
- Eskander MB, Limb D, Stone MH, Furlong AJ, Shardlow D, Stead D, et al. Sequential mechanical and pharmacological thromboprophylaxis in the surgery of hip fractures: A pilot study. International Orthopaedics 1997;21(4):259‐61. [DOI] [PMC free article] [PubMed] [Google Scholar]
Frim 1992 {published data only}
- Frim DM, Barker FG 2nd, Poletti CE, Hamilton AJ. Postoperative low‐dose heparin decreases thromboembolic complications in neurosurgical patients. Neurosurgery 1992;30(6):830‐2. [PubMed] [Google Scholar]
Gagner 2012 {published data only}
- Gagner M, Selzer F, Belle SH, Bessler M, Courcoulas AP, Dakin GF, et al. Adding chemoprophylaxis to sequential compression might not reduce risk of venous thromboembolism in bariatric surgery patients. Surgery for Obesity and Related Diseases 2012;8(6):663‐70. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gelfer 2006 {published data only}
- Gelfer Y, Tavor H, Oron A, Peer A, Halperin N, Robinson D. Deep vein thrombosis prevention in joint arthroplasties: continuous enhanced circulation therapy vs low molecular weight heparin. Journal of Arthroplasty 2006;21(2):206‐14. [DOI] [PubMed] [Google Scholar]
Kamran 1998 {published data only}
- Kamran SI, Downey D, Ruff RL. Pneumatic sequential compression reduces the risk of deep vein thrombosis in stroke patients. Neurology 1998;50(6):1683‐8. [DOI] [PubMed] [Google Scholar]
Kiudelis 2010 {published data only}
- Kiudelis M, Gerbutavicius R, Gerbutaviciene R, Griniūte R, Mickevicius A, Endzinas Z, et al. A combinative effect of low‐molecular‐weight heparin and intermittent pneumatic compression device for thrombosis prevention during laparoscopic fundoplication. Medicina 2010;46:18‐23. [PubMed] [Google Scholar]
Kumaran 2008 {published data only}
- Kumaran R, Sokolovic M, Ashame E, George EM, Bernstein L, George L. Prevention of venous thromboembolism in critically ill medical patients. Chest 2008; Vol. 134, issue 4:36003s.
Lieberman 1994 {published data only}
- Lieberman JR, Huo MM, Hanway J, Salvati EA, Sculco TP, Sharrock NE. The prevalence of deep venous thrombosis after total hip arthroplasty with hypotensive epidural anesthesia. Journal of Bone and Joint Surgery 1994;76‐A(3):341–8. [DOI] [PubMed] [Google Scholar]
Macdonald 2003 {published data only}
- Macdonald RL, Amidei C, Baron J, Weir B, Brown F, Erickson RK, et al. Randomized, pilot study of intermittent pneumatic compression devices plus dalteparin versus intermittent pneumatic compression devices plus heparin for prevention of venous thromboembolism in patients undergoing craniotomy. Surgical Neurology 2003;59(5):363‐72; discussion 372‐4. [DOI] [PubMed] [Google Scholar]
Mehta 2010 {published data only}
- Mehta KV, Lee HC, Loh JSY. Mechanical thromboprophylaxis for patients undergoing hip fracture surgery. Journal of Orthopaedic Surgery (Hong Kong) 2010;18(3):287‐9. [DOI] [PubMed] [Google Scholar]
- Mehta KV, Lee HC, Loh JSY. Reply to a letter to the Editor re: Mechanical thromboprophylaxis for patients undergoing hip fracture surgery. Journal of Orthopaedic Surgery (Hong Kong) 2011;19(2):260‐2. [DOI] [PubMed] [Google Scholar]
Nathan 2006 {published data only}
- Nathan SS, Simmons KA, Lin PP, Hann LE, Morris CD, Athanasian EA, et al. Proximal deep vein thrombosis after hip replacement for oncologic indications. Journal of Bone and Joint Surgery ‐ American Volume 2006;88(5):1066‐70. [DOI] [PubMed] [Google Scholar]
Patel 2010 {published data only}
- Patel AR, Crist MK, Nemitz J, Mayerson JL. Aspirin and compression devices versus low‐molecular‐weight heparin and PCD for VTE prophylaxis in orthopedic oncology patients. Journal of Surgical Oncology 2010;102:276–81. [DOI] [PubMed] [Google Scholar]
Roberts 1975 {published data only}
- Roberts VC, Cotton LT. Failure of low‐dose heparin to improve efficacy of perioperative intermittent calf compression in preventing postoperative deep vein thrombosis. British Medical Journal 1975;3(5981):458‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spinal cord injury investigators {published data only}
- Spinal Cord Injury Thromboprophylaxis Investigators. Prevention of venous thromboembolism in the acute treatment phase after spinal cord injury: a randomized, multicenter trial comparing low‐dose heparin plus intermittent pneumatic compression with enoxaparin. Journal of Trauma ‐ Injury Infection and Critical Care 2003;54(6):1116‐24; discussion 1125‐6. [DOI] [PubMed] [Google Scholar]
Stannard 2006 {published data only}
- Stannard JP, Lopez‐Ben RR, Volgas DA, Anderson ER, Busbee M, Karr DK, et al. Prophylaxis against deep‐vein thrombosis following trauma: a prospective, randomized comparison of mechanical and pharmacologic prophylaxis. Journal of Bone and Joint Surgery ‐ American Volume 2006;88(2):261‐6. [DOI] [PubMed] [Google Scholar]
Tsutsumi 2000 {published data only}
- Tsutsumi K, Udagawa H, Kajiyama Y, Kinoshita Y, Ueno M, Nakamura T, et al. Pulmonary thromboembolism after surgery for esophageal cancer: its features and prophylaxis. Surgery Today 2000;30(5):416‐20. [DOI] [PubMed] [Google Scholar]
Wan 2015 {published data only}
- Wan B, Fu H‐Y, Yin J‐T, Ren G‐Q. Low‑molecular‑weight heparin and intermittent pneumatic compression for thromboprophylaxis in critical patients. Experimental and Therapeutic Medicine 2015;10(6):2331‐36. [DOI] [PMC free article] [PubMed] [Google Scholar]
Westrich 1996 {published data only}
- Westrich GH, Sculco TP. Prophylaxis against deep venous thrombosis after total knee arthroplasty. Pneumatic plantar compression and aspirin compared with aspirin alone. Journal of Bone and Joint Surgery 1996;78‐A(6):826‐34. [DOI] [PubMed] [Google Scholar]
Whitworth 2011 {published data only}
- Whitworth JM, Schneider KE, Frederick PJ, Finan MA, Reed E, Fauci JM, et al. Double prophylaxis for deep venous thrombosis in patients with gynecologic oncology who are undergoing laparotomy: does preoperative anticoagulation matter?. International Journal of Gynecological Cancer 2011;21(6):1131‐4. [DOI] [PubMed] [Google Scholar]
Winemiller 1999 {published data only}
- Winemiller MH, Stolp‐Smith KA, Silverstein MD, Therneau TM. Prevention of venous thromboembolism in patients with spinal cord injury: effects of sequential pneumatic compression and heparin. Journal of Spinal Cord Medicine 1999;22(3):182‐91. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
CHICTR‐IPR‐15007324 {published data only}
- CHICTR‐IPR‐15007324. The mechanical and medical prevention of lower extremity deep venous thrombosis formation post gynecologic pelvic surgery, a multiple center randomized case control study. http://apps.who.int/trialsearch/Trial2.aspx?TrialID=ChiCTR‐IPR‐15007324 (accessed April 2016).
ISRCTN44653506 and NCT02040103 {published data only}
- Arabi YM, Alsolamy S, Al‐Dawood A, Al‐Omari A, Al‐Hameed F, Burns KE, et al. Thromboprophylaxis using combined intermittent pneumatic compression and pharmacologic prophylaxis versus pharmacologic prophylaxis alone in critically ill patients: study protocol for a randomized controlled trial. Trials 2016;17:390. [DOI: 10.1186/s13063-016-1520-0] [DOI] [PMC free article] [PubMed] [Google Scholar]
- ISRCTN44653506. The PREVENT trial: pneumatic compression for preventing venous thromboembolism. http://www.isrctn.com/ISRCTN44653506 Accessed May 2016.
NCT00740987 {published data only}
- NCT00740987. Efficacy of the association mechanical prophylaxis + anticoagulant prophylaxis on venous thromboembolism incidence in intensive care unit (ICU) (CIREA2). https://clinicaltrials.gov/ct2/show/NCT00740987 (accessed April 2016).
Additional references
Atkins 2004
- Atkins D, Best D, Briss PA, Eccles M, Falck‐Ytter Y, Flottorp S, et al. Grading quality of evidence and strength of recommendations. British Medical Journal 2004;328(7454):1490‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Chouhan 1999
- Chouhan VD, Comerota AJ, Sun L, Harada R, Gaughan JP, Rao AK. Inhibition of tissue factor pathway during intermittent pneumatic compression: a possible mechanism for antithrombotic effect. Arteriosclerosis, Thrombosis and Vascular Biology 1999;19(11):2812‐7. [DOI] [PubMed] [Google Scholar]
Egger 1997
- Egger M, Davey SG, Schneider M, Minder C. Bias in meta‐analysis detected by a simple, graphical test. British Medical Journal (Clinical Research Ed.) 1997;315(7109):629‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
Eriksson 2001
- Eriksson BI, Bauer KA, Lassen MR, Turpie AG, for the Steering Committee of the Pentasaccharide in Hip‐Fracture Surgery Study. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after hip‐fracture surgery. New England Journal of Medicine 2001;345(18):1298‐304. [DOI] [PubMed] [Google Scholar]
Eriksson 2008
- Eriksson BI, Borris LC, Friedman RJ, Haas S, Huisman MV, Kakkar AK, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. New England Journal of Medicine 2008;358(26):2765‐75. [DOI] [PubMed] [Google Scholar]
Goldhaber 2001
- Goldhaber SZ. Prophylaxis of venous thrombosis. Current Treatment Options in Cardiovascular Medicine 2001;3(3):225‐35. [DOI] [PubMed] [Google Scholar]
Gould 2012
- Gould MK, Garcia DA, Wren SM, Karanicolas PJ, Arcelus JI, Heit JA, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. Chest 2012;141:e227S‐e277S. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. Available from www.cochrane‐handbook.org. 2011.
Ho 2013
- Ho KM, Tan JA. Stratified meta‐analysis of intermittent pneumatic compression of the lower limbs to prevent venous thromboembolism in hospitalized patients. Circulation 2013;128:1003‐20. [DOI] [PubMed] [Google Scholar]
Kakkos 2005
- Kakkos SK, Griffin M, Geroulakos G, Nicolaides AN. The efficacy of a new portable sequential compression device (SCD Express) in preventing venous stasis. Journal of Vascular Surgery 2005;42(2):296‐303. [DOI] [PubMed] [Google Scholar]
Kakkos 2012
- Kakkos SK, Warwick D, Nicolaides AN, Stansby GP, Tsolakis IA. Combined (mechanical and pharmacological) modalities for VTE prevention in joint arthroplasty. Journal of Bone and Joint Surgery. British Volume 2012;94‐B:729‐34. [DOI] [PubMed] [Google Scholar]
King 2007
- King CS. Twice vs three times daily heparin dosing for thromboembolism prophylaxis in the general medical population: a meta‐analysis. Chest 2007;131(2):507‐16. [DOI] [PubMed] [Google Scholar]
McLeod 2001
- McLeod RS, Geerts WH, Sniderman KW, Greenwood C, Gregoire RC, Taylor BM, et al. Canadian Colorectal Surgery DVT Prophylaxis Trial investigators. Subcutaneous heparin versus low‐molecular‐weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian colorectal DVT prophylaxis trial: a randomized, double‐blind trial. Annals of Surgery 2001;233(3):438‐44. [DOI] [PMC free article] [PubMed] [Google Scholar]
NICE 2009
- National Clinical Guideline Centre ‐ Acute and chronic conditions. Venous thromboembolism: reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital. https://www.nice.org.uk/guidance/cg92/ (accessed April 2016).
Nicolaides 2013
- Nicolaides AN, Fareed J, Kakkar AK, Comerota AJ, Goldhaber SZ, Hull R, et al. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). International Angiology 2013;32:111‐260. [PubMed] [Google Scholar]
NRSMG
- Cochrane Non‐Randomised Studies Methods Group. Draft chapters for the Guidelines on Non‐randomised studies in Cochrane reviews. http://www.cochrane.dk/nrsmg/guidelines.htm 2001 (accessed August 2007).
Piazza 2007
- Piazza G, Seddighzadeh A, Goldhaber SZ. Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent. Chest 2007;132(2):554‐61. [DOI] [PubMed] [Google Scholar]
Rosendaal 1999
- Rosendaal FR. Venous thrombosis: a multicausal disease. Lancet 1999;353(9159):1167‐73. [DOI] [PubMed] [Google Scholar]
Schulman 2010
- Schulman S, Angerås U, Bergqvist D, Eriksson B, Lassen MR, Fisher W, on behalf of the Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in surgical patients. Journal of Thrombosis and Haemostasis 2010;8(1):202‐4. [DOI] [PubMed] [Google Scholar]
Sobieraj 2013
- Sobieraj DM, Coleman CI, Tongbram V, Chen W, Colby J, Lee S, et al. Comparative effectiveness of combined pharmacologic and mechanical thromboprophylaxis versus either method alone in major orthopedic surgery: a systematic review and meta‐analysis. Pharmacotherapy 2013;33(3):275–83. [DOI] [PubMed] [Google Scholar]
Virchow 1856
- Virchow RLK. Thrombosis and emboli [Phlogose und thrombose im gefässsystem]. Gesammelte Adhandlungen zur Wissenschaftlichen Medicine. Frankfurt am Main: von Meidinger & Sohn, 1856:458‐636. [Google Scholar]
Zareba 2014
- Zareba P, Wu C, Agzarian J, Rodriguez D, Kearon C. Meta‐analysis of randomized trials comparing combined compression and anticoagulation with either modality alone for prevention of venous thromboembolism after surgery. British Journal of Surgery 2014;101:1053–62. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Kakkos 2005a
- Kakkos SK, Geroulakos G, Caprini J, Nicolaides AN, Stansby G. Combined intermittent pneumatic leg compression and pharmacological prophylaxis for prevention of venous thromboembolism in high risk patients. Cochrane Database of Systematic Reviews 2005, Issue 2. [DOI: 10.1002/14651858.CD005258] [DOI] [PubMed] [Google Scholar]
Kakkos 2008
- Kakkos SK, Caprini JA, Geroulakos G, Nicolaides AN, Stansby G, Reddy DJ. Combined intermittent pneumatic leg compression and pharmacological prophylaxis for prevention of venous thromboembolism in high‐risk patients. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD005258.pub2] [DOI] [PubMed] [Google Scholar]


