Abstract
Holt-Oram syndrome (HOS) is an autosomal dominant condition characterised by the association of congenital heart defect (CHD), with or without rhythm disturbances and radial defects, due to TBX5 variants. The diagnosis is challenged by the variability of expression and the large phenotypic overlap with other conditions, like Okihiro syndrome, TAR syndrome or Fanconi disease. We retrospectively reviewed 212 patients referred for suspicion of HOS between 2002 and 2014, who underwent TBX5 screening. A TBX5 variant has been identified in 78 patients, representing the largest molecular series ever described. In the cohort, 61 met the previously described diagnostic criteria and 17 have been considered with an uncertain HOS diagnosis. A CHD was present in 91% of the patients with a TBX5 variant, atrial septal defects being the most common (61.5%). The genotype–phenotype study highlights the importance of some critical features in HOS: the septal characteristic of the CHD, the bilateral and asymmetric characteristics of the radial defect and the presence of shoulder or elbow mobility defect. Besides, 21 patients presented with an overlapping condition. Among them, 13 had a typical HOS presentation. We discuss the strategies that could be adopted to improve the molecular delineation of the remaining typical patients.
Subject terms: Disease genetics, Congenital heart defects, Genetic counselling
Introduction
Congenital heart disease (CHD) is the most common congenital anomaly with an estimated prevalence of 9 per 1000 at birth [1]. Septal defects represent 47% of CHD [1]. Several genes contribute to heart development. Although NKX2–5 and GATA4 have been associated with isolated CHD, their partner TBX5 is implicated in Holt-Oram syndrome (HOS) (MIM #142900). This autosomal dominant condition is characterised by the association of CHD and anterior upper limb defect, with a high penetrance and a wide variability of expression [2]. Its prevalence has been estimated at 1 in 100,000 births [3]. A CHD is present in 70–95% of the individuals according to the literature [4–10]. The most common are septal defects: ostium secundum atrial septal defect (44.4%) and ventricular septal defect (29.4%). More severe CHD have also been reported: atrioventricular septal defect, Fallot tetralogy, left ventricle hypoplasia, and aortic coarctation. Rhythm disturbances (39%) can be found isolated or associated with a CHD, and may appear over the course of life. The most common are sinus bradycardia and right bundle branch block [4, 6, 7, 9, 11]. Upper limb involvement is constant but very variable. Its spectrum extends from an isolated thenar hypoplasia, a mobility defect affecting the thumb, elbow, or shoulder, up to phocomelia. The most common defects are thumb hypoplasia at various degrees or a triphalangeal/fingerlike thumb, associated or not with radial hypoplasia. A left-right asymmetry may be observed, with usually more severe defects on the left-hand side [4, 6]. No correlation between the severity of the skeletal and the cardiac defects has been observed [6].
Variants in the TBX5 gene (chromosome 12q24) have been found to cause HOS in 1997 [12, 13]. TBX5 encodes a transcription factor belonging to the T-box family, regulating a large variety of developmental processes in vertebrates [14]. During heart development, Tbx5 expression appears uniformly at the early stages, then is left-biased as the heart tube loops, being totally absent from the right ventricle [15]. Its role is crucial for cardiomyocytes proliferation and differentiation, septum formation and the establishment of the conduction system (reviewed in the ref. [16]). During limb development, Tbx5 is expressed in forelimbs and not in hindlimbs. Its action is essential at early stages of limb initiation (E9.5–E10.5) to trigger the epithelio-mesenchymal growth loop between Fgf10 in the mesenchyme and Fgf8 in the apical ectodermal ridge. At later stages (E11.2–E12.5), Tbx5 is involved in muscle and tendon patterning and no longer needed for limb outgrowth [17].
About 300 HOS patients have been described in the literature. However, for most of them, the diagnosis was only established on clinical features [4–11]. The molecular confirmation of HOS diagnosis is crucial because of the cardiac monitoring required by this condition, and its impact on the genetic counselling. The detection yield of TBX5 variants in HOS patients ranges from 22 to 74% depending on the studies [8, 11, 18–20]. The diagnostic criteria were consistent in most studies (uni or bilateral anterior upper limb defect, associated with a personal or familial history of CHD or rhythm disturbances). Mc Dermott et al. showed that the variant detection rate increases to 74% when the absence of associated features (except for spinal deformities) was added to the previous criteria. Of note, the molecular analysis methods were heterogeneous in the reported series of the literature and none of them included intragenic rearrangements screening.
Here, we report on the clinical and molecular characterisation of 78 unrelated HOS individuals harbouring a TBX5 variant, allowing the description of the largest molecular series so far. These data allow detailed genotype–phenotype correlations and support the idea that molecular analyses are essential to distinguish HOS from overlapping conditions, which could be clinically confused.
Patients and methods
Patients
A multicentre, retrospective study was set up to collect data on 228 patients addressed to our laboratory for suspicion of HOS, between 2002 and 2014. This collaboration involved 56 departments of clinical genetics. Sixteen patients were excluded because of a non-appropriate diagnosis of HOS, in presence of postaxial upper limb involvement, lower limb defect or multiple associated malformations. Patient phenotypes were divided into three groups: typical HOS with heart defect, typical HOS without heart defect, uncertain HOS. Typical HOS patients met the diagnostic criteria established by Mc Dermott et al. HOS was considered as uncertain, if the preaxial defect, associated with a heart defect, was unilateral and/or in presence of one or two minor additional features. The associated features were considered as minor if they were previously described in a patient with a TBX5 variant or if they are common in general population (growth retardation, urogenital malformations for example). Data from 212 patients were recorded. Clinical and radiological information were collected at the time of diagnosis and updated thanks to a clinical questionnaire as part of the study (available on request).
Ethics statement
Our study was performed using the Declaration of Helsinki protocol. Written informed consents were obtained from the patients or their parents prior to the molecular diagnostic analysis.
Genotyping
Peripheral blood was collected from patients and relatives. We performed Sanger sequencing of all TBX5 coding exons and their flanking intronic regions. Primers sequences are available on request. Variants are given according to reference sequence NM_000192.3 (Hg19). Large intragenic TBX5 rearrangements were tested by a multiplex ligation-dependent probe amplification (MLPA) kit (PE180 Limb malformations probemix, MRC-Holland). TBX5 variants were submitted to ClinVar database, available at the following link: http://www.ncbi.nlm.nih.gov/clinvar/?term=TBX5[gene]. For the interpretation of missense variants, pathogenicity prediction software (SIFT, Polyphen) and proteic structure prediction software (PHYRE2, SuperPose) were used.
Statistical analysis
Comparisons between the different groups of patients have been assessed by the Chi-square test, with Yates correction when the application conditions were not respected.
Results
Among the 212 patients, 105 met the established diagnostic criteria (79 with heart defect, 26 without heart defect) and 107 were considered with an uncertain HOS diagnosis. TBX5 molecular analysis identified 78 probands with TBX5 variants: 61 who meet the diagnostic criteria and 17 who were clinically considered as uncertain. According to the diagnostic criteria established by Mc Dermott et al., the detection rate of TBX5 variant was 58%. This detection rate was better in the subgroup associating bilateral anterior upper limb defect and CHD (70%).
Description of the patients with TBX5 variants
Within the 78 probands with TBX5 variants, 47 were sporadic cases. The clinical and radiological features observed in the patients and their 16 affected relatives are detailed in Tables 1 and 2. A cardiac defect was observed in 91% (71/78) of the patients. ASD was the most frequent (61.5% of the cases). Rhythm disturbances were observed in 30% of the living patients. The range of the limb phenotypes is depicted in Fig. 1. The upper limb defects were bilateral in 96% (75/78) of the patients. A left/right asymmetry was observed in 91% (31/34) of the patients when this information was available. The observed malformations were most often reduction defects (79.5%). None of the patients presented with preaxial polydactyly. Fourteen patients did not fit into the strict diagnostic criteria: 7 presented with the association of one or two skeletal features (scoliosis [4], pectus excavatum [3], costal deformities [1]), or one or two non-skeletal features (growth retardation [2], bilateral pyelic dilatation [1], familial form of Kallmann syndrome [1], laryngomalacia [1], pulmonary malformation [2], micropenis [1]). We describe 11 prenatal cases: 4 were revealed by an isolated upper limb defect (thumb agenesis, radial hypoplasia, phocomelia), 2 by an isolated CHD (VSD, AVD), 5 by the association of upper limb defect and CHD. The severity of the upper limb defects led to pregnancy termination for 6 foetuses. Data regarding the management and the evolution of the patients were often missing in the questionnaire filled in mainly by clinical geneticists. Orthopaedic surgery was reported for 25% (18/71) of the patients and consisted for 72% of them in pollicisation of the index finger. Cardiac surgery was performed in 47% (31/66) of the living patients with CHD and a pacemaker was placed for 38% (8/21) of the patients with rhythm disturbances. Two deaths occurred, including one during a cardiac surgery, due to the severity of the CHD consisting of atrioventricular septal defect either isolated or associated with a tight isthmic coarctation.
Table 1.
Frequency of cardiac phenotypes in the 78 probands and their affected relatives in our series, compared to the literature [4–11]
| Patients from our study | Literature [4–11] | ||
|---|---|---|---|
| Probands (N = 78) | Probands and relatives (N = 94) | ||
| Congenital heart disease (CHD) | |||
| General | |||
| Single septal defect | 38/78 (48.7) | 44/94 (46.8) | nd |
| Multiple septal defect | 12/78 (15.4) | 16/94 (17) | nd |
| Septal defect, associated with another CHD | 15/78 (19.2) | 17/94 (18) | nd |
| Isolated valvular defect | 2/78 (2.6) | 2/94 (2.1) | nd |
| Details | |||
| Septal defects | |||
| Atrial septal defect | 48/78 (61.5) | 58/94 (61.7) | 44.4% |
| Ventricular septal defect | 27/78 (34.6) | 32/94 (34) | 29.4% |
| Atrioventricular septal defect | 4/78 (5.1) | 4/94 (4.3) | 3.3% |
| Other | |||
| Mitral valve abnormalities | 3/78 (3.8) | 3/94 (3.2) | 4.1% |
| Patent ductus arteriosus | 3/78 (3.8) | 3/94 (3.2) | 3.6% |
| Pulmonary stenosis | 2/78 (2.6) | 3/94 (3.2) | 1.8% |
| Dextrocardia | 2/78 (2.6) | 2/94 (2.1) | 1.8% |
| Left ventricle hypoplasia | 2/78 (2.6) | 2/94 (2.1) | nd |
| Aortic coarctation | 4/78 (5.1) | 4/94 (4.3) | nd |
| Left superior vena cava | 3/78 (3.8) | 3/94 (3.2) | nd |
| Complex CHD | 1/78 (1.3) | 1/94 (1) | nd |
| Conduction disturbances | |||
| General | |||
| Isolated | 3/71 (4.2) | 5/87 (5.7) | nd |
| Associated with CHD | 18/71 (25.3) | 20/87 (23) | nd |
| Details | |||
| Sinus bradycardia | 6/71 (8.5) | 7/87 (8) | nd |
| Atrial flutter | 2/71 (2.8) | 2/87 (2.3) | nd |
| Atrioventricular block | 2/71 (2.8) | 4/87 (4.6) | nd |
| Right bundle branch block | 5/71 (7) | 6/87 (6.9) | nd |
| Junctional tachycardia | 5/71 (7) | 5/87 (5.7) | nd |
| Long QT | 1/71 (1.4) | 1/87 (1.1) | nd |
Numbers in brackets are expressed in percent
nd not determined
Table 2.
Frequency of skeletal phenotypes in the 78 probands and their affected relatives in our series, compared to the literature [4–11]
| Patients from our study | Literature [4–11] | ||||
|---|---|---|---|---|---|
| Probands (N = 78) | Probands and relatives (N = 94) | Right and left | |||
| Right | Left | Right | Left | ||
| Hand | |||||
| Reduction defect of the thumbs | |||||
| Thenar hypoplasia | 22/78 (28.2) | 20/78 (25.6) | 23/94 (24.4) | 21/94 (22.3) | 34.2% |
| Thumb hypoplasia | 23/78 (29.5) | 26/78 (33.3) | 26/94 (27.7) | 30/94 (31.9) | 31.9% |
| Thumb agenesis | 17/78 (21.8) | 25/78 (32.1) | 18/94 (19.1) | 27/94 (28.7) | 32.3% |
| I-II syndactyly | 12/78 (15.4) | 11/78 (14.1) | 14/94 (14.9) | 12/94 (12.8) | 13.3% |
| First metacarpal agenesis | 4/78 (5.1) | 3/78 (3.8) | 4/94 (4.3) | 3/94 (3.2) | nd |
| Additional defect of the thumbs | |||||
| Triphalangeal thumb | 21/78 (26.9) | 19/78 (24.3) | 21/94 (22.3) | 20/94 (21.3) | 23.6% |
| Digitalised thumb | 5/78 (6.4) | 2/78 (2.6) | 5/94 (5.3) | 4/94 (4.3) | 31.9% |
| Bifid thumb | – | – | – | – | 5% |
| II–III syndactyly | 2/78 (2.6) | 3/78 (3.8) | 2/94 (2.1) | 3/94 (3.2) | nd |
| Carpal bones malformation | 3/78 (3.8) | 3/78 (3.8) | 5/94 (5.3) | 5/94 (5.3) | 35.3% |
| Forearm | |||||
| Radial defects | |||||
| Hypoplasia | 17/78 (21.8) | 17/78 (21.8) | 19/94 (20.2) | 20/94 (20.2) | 34.2% |
| Agenesis | 4/78 (5.1) | 8/78 (10.3) | 4/94 (4.3) | 8/94 (8.5) | 15.2% |
| Ulnar defects | – | 1/78 (1.3) | – | 1/94 (1.1) | nd |
| Radio-ulnar synostosis | 3/78 (3.8) | 5/78 (6.4) | 3/94 (3.2) | 5/94 (5.3) | nd |
| Limitation of pro-supination | 14/78 (17.9) | 16/78 (20.5) | 19/94 (20.2) | 20/94 (21.3) | 25.8% |
| Limitation of elbow mobility | 9/78 (11.5) | 12/78 (15.4) | 12/94 (12.8) | 14/94 (14.9) | nd |
| Arm | |||||
| Humeral hypoplasia | – | 1/78 (1.3) | – | 1/94 (1) | 32% |
| Phocomelia | 3/78 (3.8) | 4/78 (5.1) | 4/94 (4.3) | 5/94 (5.3) | 8.4 % |
| Shoulder | |||||
| Limitation of elbow mobility | 4/78 (5.1) | 5/78 (6.4) | 4/94 (4.3) | 5/94 (5.3) | 28.6% |
| Sloping shoulders | 20/78 (25.6) | 18/78 (23) | 20/94 (20.2) | 18/94 (19.1) | 7.5% |
| Clavicle defects | 8/78 (10.3) | 8/78 (10.3) | 10/94 (10.6) | 10/94 (10.6) | 31.6% |
| Muscular hypoplasia | 5/78 (6.4) | 5/78 (6.4) | 5/94 (5.3) | 5/94 (5.3) | 38.2% |
Numbers in brackets are expressed in percent
nd not determined
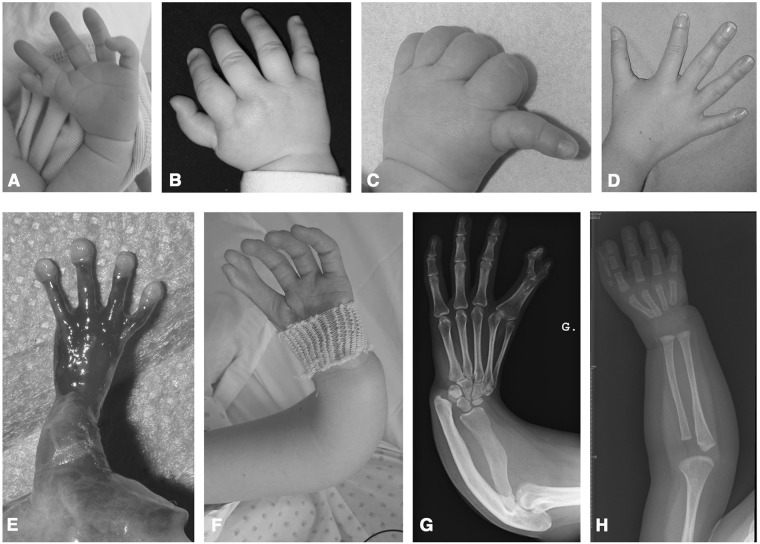
Fig. 1.
Clinical and radiological phenotypes in HOS patients. a Left triphalangeal thumb, I–II syndactyly, elbow mobility defect; b Right thumb hypoplasia; c Left thumb hypoplasia; d Left triphalangeal thumb; e Left thumb agenesis in a foetus: f Left digitalised thumb, I–II syndactyly, radial hypoplasia; g Radial and ulnar hypoplasia, I–II phalangeal syndactyly; h Right thumb hypoplasia
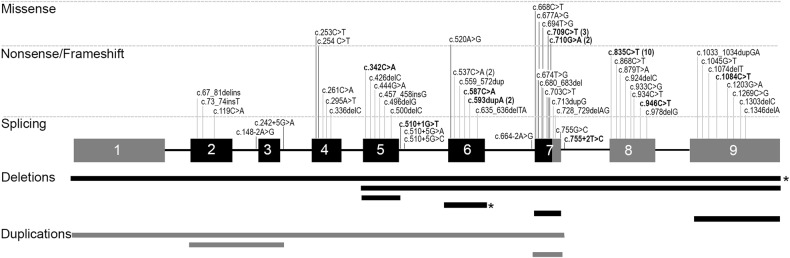
Among the observed TBX5 variants, 87% were point changes. Most of them were truncating (37% nonsense, 26% frameshift, 10% splice site), while 14% were missense variants. MLPA analysis identified TBX5 intragenic deletions in 8% and intragenic duplications in 4%. Finally, an apparently balanced translocation t(2;12)(q31;q24.3) implicating the TBX5 locus did segregate in 3 affected cases of the same family. All described variants are illustrated in the Fig. 2. We identified 49 novel variants. Two novel variants responsible for an extended TBX5 protein were found: one familial case (c.1303delC, p.(Leu435Trpfs*147), triphalangeal thumbs, radial hypoplasia, single auricle with sinusal bradycardia) and one sporadic foetus (c.1346delA, p.(Gln449Argfs*70) de novo, bilateral thumbs and radial hypoplasia, complex CHD and a micropenis). Eight distinct missense variants have been identified in 11 unrelated patients. Their pathogenicity was appreciated according to criteria developed in Supp table 1. Two of them occurred de novo in sporadic cases, therefore considered as likely pathogenic. Two of them concerned the amino acid 237, crucial for DNA interaction. Functional tests for these 2 variants have been previously published, allowing us to consider them as pathogenic. The remaining 4 variations have been considered to be of uncertain significance.
Fig. 2.
Representation of the TBX5 variants identified in our series of 78 HOS patients. Dark exons represent the T-box domain. Point variants are represented at the superior part of the figure. Intragenic deletions or duplications are represented at the inferior part. Variants in bold and deletions highlighted by a star have been described previously in the literature. The exons/introns are not at scale
Lack of penetrance—Mosaicism
In our series, 4 non-related parents of an affected child harboured the familial variant but no clinical HOS features. Two of them had normal skeletal X rays, cardiac ultrasound and electrocardiogram, whereas their children had typical features of HOS, up to phocomelia. This lack of penetrance was observed for 4 different variants: c.713dupG (p.(Ser239Glnfs*2)), duplication of exons 1 to 7 (c.(?_−667)_(755+1_756–1)dup), deletion of exon 7 (c.(663+1_664–1)_(755+1_756–1)del) and c.710G>A (p.(Arg237Gln)). Furthermore, one asymptomatic father of a proband presented with a somatic mosaicism (around 10% of the alleles in blood sample) for the variant identified in his son c.537C>A (p.(Tyr179*)).
Differential diagnosis
Among the 212 patients, 21 turned out to present a differential diagnosis (Supp table 2). Thirteen of them had typical presentation of HOS. Okihiro syndrome was confirmed molecularly for eight patients. Fanconi anemia was diagnosed in presence of increased chromosome breakage on karyotype with mitomycin C. TAR syndrome was confirmed in presence of a 1q21.1 microdeletion involving RBM8A, even if the second hit (hypomorphic SNP) was not observed.
Genotype–phenotype correlations
First, we compared the phenotypes between variant-positive and variant-negative patients, in the 3 phenotypic groups of patients. Some items were significantly more common in the variant-positive patients: the presence of a familial history, the presence of a septal defect, the association of a rhythm disturbance with a CHD, the bilateral characteristics of the radial defect and the presence of a shoulder or elbow mobility defect (p < 0.05) (Supp Table 3).
In addition, we compared the phenotypes according to the type of TBX5 variant: truncating (nonsense, frameshift, splice site point variants, intragenic deletions or duplications) versus missense variants. We observed that isolated septal CHD are more common in the truncating than in the missense variants (p = 0.02). Besides, complex CHD associated to septal defect seems to be more common in case of missense variants (p = 0.053). Thus, we observed a tendency of less severe CHD in patients with truncating variants compared to the patients with missense variants. No significant difference was observed in the type or the severity of upper limb defects between the two groups.
Discussion
This description of the largest molecular HOS series ever reported allows a better delineation of the HOS phenotype and genotype, and helps to direct appropriate molecular analyses by using more accurate diagnostic criteria for HOS and overlapping conditions.
Our clinical study highlights the importance of some critical features: the septal involvement for the CHD, the bilateral characteristics of the radial ray defects and the shoulder or elbow mobility defect (even in the absence of any skeletal involvement). Preaxial polydactyly, previously described in HOS in the literature [10], has not been observed in any of our patients with TBX5 variants. We observed that the presence of pectus excavatum, spinal deformities or pulmonary malformations should not rule out the HOS hypothesis. The presence of pectus excavatum in HOS has been largely overestimated (30%) in the clinical series of the literature, probably because some of these patients had an overlapping condition [4, 6]. In our series, only 3 patients out of 78 (4%) presented with pectus excavatum, which is still higher than its frequency in the general population (0.3%) [21]. This association can be explained by the involvement of Tbx5 in sternum development, demonstrated by the presence of abnormal sternal formation in a Tbx5 conditional mutant [22]. Four patients out of 78 (5%) presented with scoliosis, which is not significantly different than its frequency in the general population (2–3%) [23], suggesting that this feature is independent from HOS. Four patients from the literature presented with vertebral defects: fusion of cervical vertebrae, defect of anterior vertebral bodies, hemivertebra [4, 8, 10, 19], while Tbx5 expression has never been reported in the spinal column. Two patients of our series presented with a pulmonary malformation. A right lung agenesis, a right lung hypoplasia and a horseshoe lung have been previously described in HOS [24–26]. Tbx5 expression has been described in the mesenchyme of the developing lungs and trachea, and could participate, through Fgf10 pathway, to lung branching [27]. The other associated features were likely incidental.
We described a lack of penetrance in 4 different HOS families in our series, and a somatic mosaicism in the father of one proband. HOS has generally been described as a condition with complete penetrance, but 2 families presenting with missense variants and an incomplete penetrance have already been reported [8, 28]. Somatic mosaicism for a variant has previously been reported in an asymptomatic parent in the literature [9]. This highlights the importance of the parental molecular analyses for all the probands with TBX5 variants, even if the case seems to be sporadic, to be able to give an accurate genetic counselling to these families.
TBX5 variants are distributed throughout the gene, but the majority are found within the T-box DNA binding domain. Nonsense variants are the most common in our series (37%), whereas missense variants are the most frequently reported TBX5 variants in the literature (30%, versus 14% in our series), due to the larger propensity of publication of missense variant functional analyses (reviewed in the ref. [29]). Nonetheless, most of the reported TBX5 variants (70%) lead to haploinsufficiency by synthesis of a non-functional truncated protein or by triggering the nonsense-mediated mRNA decay, by nonsense, frameshift or splice site variants [29]. Missense TBX5 variants can cause loss-of-function or exceptionally gain-of-function. Sequence point variations leading to an extended protein and large intra and intergenic deletion/duplication have also been occasionally reported [29]. We observed a higher propensity of missense variants to be responsible for complex CHD associated to septal defect. In the literature, the few TBX5 variants described in patients presenting with a non-septal CHD (Fallot tetralogy, cardiomyopathy) are also missense variants. Also, frameshift variants leading to an extended TBX5 protein seem to be associated with more severe CHD and associated malformations [30]. This suggests a potential dominant-negative effect for these variants.
HOS is a challenging diagnosis by its variability of expression and the large phenotypic overlap with other conditions. Among the patients addressed to our laboratory for HOS suspicion, 21 turned out to present a differential diagnosis (Supp table 3). More than 50% of these patients met the HOS diagnostic criteria. The most common is Okihiro syndrome (MIM#607323), characterised by constant bilateral anterior upper limb defects, possibly associated with abnormality of the ocular motility named Duane anomaly (65% of the cases), morphological renal defects (28%), but also with cardiac (17%) or spinal defects and ear dysplasia or deafness [8, 31]. This autosomal dominant condition is due to variants affecting SALL4, which encodes a transcription factor acting synergistically with TBX5 during heart and limb development [32, 33]. HOS can be mistaken with many other overlapping conditions (summarized in Table 3) and clinical assessment of patients should be standardized, as detailed in Fig. 3.
Table 3.
Summary and frequency of associated features that can be observed in the main differential diagnoses for HOS
| MIM# | Gene/Locus | Inheritance | Cardiac anomalies | Radial defects | Characteristic clinical features | References | |
|---|---|---|---|---|---|---|---|
| Okihiro syndrome | 607323 | SALL4 | AD | 23% | 80% | Duane anomaly, renal anomalies | [8, 31] |
| Fanconi pancytopenia | 227650 | FA core complex genes | AR, XL | 6% | 35% | Pancytopenia, growth retardation, microcephaly | [38] |
| Polydactyly, preaxial type II Syndactyly type IV | 174500 and 186200 | ZRS locus | AD | – | 100% | Isolated pre-axial polydactyly with or without triphalangeal thumbs, mirror image polydactyly | [39] |
| Valproate embryopathy | – | – | sporadic | 14–26% | 30% | Developmental delay, neural tube defects, vertebral defects, hypospadias, cleft palate | [40, 41] |
| VACTERL association | 192350 | – | sporadic | 40–80% | 40–50% | Vertebral defects, anal atresia/stenosis, cardiac anomalies, tracheo-Esophageal fistula, renal anomalies, limb defects | [42] |
| Townes-Brockes syndrome | 107480 | SALL1 | AD | 9–40% | 50–90% | Anal atresia, ears malformations, deafness, triphalangeal thumbs, renal anomalies | [43] |
| TAR syndrome | 274000 | RBM8A | AR | 15–30% | 100% | Thrombocytopenia, preserved thumbs, lower limbs defects | [44, 45] |
| Nager syndrome | 154400 | SF3B4 | AD | 15% | 100% | Mandibulofacial dysostosis, deafness | [46] |
| LADD syndrome | 149730 | FGF10, FGFR2, FGFR3 | AD | - | 95% | Lacrymal ducts atresia, ears malformations, deafness, abnormal teeth, thumb anomalies | [47, 48] |
| Roberts syndrome | 268300 | ESCO2 | AR | 26% | 100% | Growth retardation, craniofacial malformations, upper and lower limbs reduction defects | [49, 5, 50] |
Fig. 3.

Assessment of patients suspected for HOS
Finally, some patients with typical HOS features had no TBX5 variant or identified overlapping condition after additional screening by array-CGH and the targeted high-throughput sequencing of 124 genes involved in limb development (data not shown). Previously, Terrett et al. identified 2 families with a typical presentation of HOS that were not linked to the 12q24 locus [34], supporting the hypothesis of a genetic heterogeneity. However, we performed whole exome sequencing in 13 selected typical cases, revealing no candidate gene (data not shown). Recent data showed that regulatory variants may explain a large part of congenital malformations [35]. Three TBX5 heart-specific enhancers have been described, and one homozygous rare variant in one of these sequences has been reported in a patient presenting with an isolated VSD [36]. A minimal regulatory sequence has been identified in intron 2, where Hox factors bind to activate Tbx5 expression during mouse upper limb initiation [37]. No variant in this element has been reported so far. Whole genome sequencing will probably help to unravel the variant-negative cases by allowing the exploration of regulatory elements, and the detailed screening of balanced chromosomal rearrangements.
Our study illustrates the inconsistencies and issues of the molecular diagnosis of HOS, by both its clinical (variability of expression, lack of penetrance, numerous overlapping conditions) and molecular aspects (diversity of TBX5 variants, potential genetic heterogeneity or regulatory variant). The diagnosis of HOS cannot be certainly affirmed or ruled out by strict diagnostic criteria and need to be confirmed by TBX5 molecular analysis, since the typical presentation of HOS can be sometimes incomplete or associated with other features in patients with TBX5 variants.
Electronic supplementary material
Acknowledgements
We greatly thank all the clinicians, the patients and their families for their contribution to this study.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
The online version of this article (10.1038/s41431-018-0303-3) contains supplementary material, which is available to authorized users.
References
- 1.van der Linde D, Konings EEM, Slager MA, et al. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. J Am Coll Cardiol. 2011;58:2241–7. doi: 10.1016/j.jacc.2011.08.025. [DOI] [PubMed] [Google Scholar]
- 2.Holt M, Oram S. Familial heart disease with skeletal malformations. Br Heart J. 1960;22:236–42. doi: 10.1136/hrt.22.2.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Elek C, Vitéz M, Czeizel E. Holt-Oram syndrome. Orv Hetil. 1991;132:73–74. [PubMed] [Google Scholar]
- 4.Smith AT, Sack GH, Taylor GJ. Holt-Oram syndrome. J Pediatr. 1979;95:538–43. doi: 10.1016/S0022-3476(79)80758-1. [DOI] [PubMed] [Google Scholar]
- 5.Hurst JA, Hall CM, Baraitser M. The Holt-Oram syndrome. J Med Genet. 1991;28:406–10. doi: 10.1136/jmg.28.6.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Newbury-Ecob RA, Leanage R, Raeburn JA, Young ID. Holt-Oram syndrome: a clinical genetic study. J Med Genet. 1996;33:300–7. doi: 10.1136/jmg.33.4.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Basson CT, Cowley GS, Solomon SD, et al. The clinical and genetic spectrum of the Holt-Oram syndrome (heart-hand syndrome) N Engl J Med. 1994;330:885–91. doi: 10.1056/NEJM199403313301302. [DOI] [PubMed] [Google Scholar]
- 8.Brassington AME, Sung SS, Toydemir RM, et al. Expressivity of Holt-Oram syndrome is not predicted by TBX5 genotype. Am J Hum Genet. 2003;73:74–85. doi: 10.1086/376436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Borozdin W, Bravo Ferrer Acosta AM, Bamshad MJ, et al. Expanding the spectrum of TBX5 mutations in Holt-Oram syndrome: detection of two intragenic deletions by quantitative real time PCR, and report of eight novel point mutations. Hum Mutat. 2006;27:975–6. doi: 10.1002/humu.9449. [DOI] [PubMed] [Google Scholar]
- 10.Barisic I, Boban L, Greenlees R, et al. Holt Oram syndrome: a registry-based study in Europe. Orphanet J Rare Dis. 2014;9:156. doi: 10.1186/s13023-014-0156-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Heinritz W, Moschik A, Kujat A, et al. Identification of new mutations in the TBX5 gene in patients with Holt-Oram syndrome. Heart Br Card Soc. 2005;91:383–4. doi: 10.1136/hrt.2004.036855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li QY, Newbury-Ecob RA, Terrett JA, et al. Holt-Oram syndrome is caused by mutations in TBX5, a member of the Brachyury (T) gene family. Nat Genet. 1997;15:21–29. doi: 10.1038/ng0197-21. [DOI] [PubMed] [Google Scholar]
- 13.Basson CT, Bachinsky DR, Lin RC, et al. Mutations in human TBX5 [corrected] cause limb and cardiac malformation in Holt-Oram syndrome. Nat Genet. 1997;15:30–35. doi: 10.1038/ng0197-30. [DOI] [PubMed] [Google Scholar]
- 14.Naiche LA, Harrelson Z, Kelly RG, Papaioannou VE. T-box genes in vertebrate development. Annu Rev Genet. 2005;39:219–39. doi: 10.1146/annurev.genet.39.073003.105925. [DOI] [PubMed] [Google Scholar]
- 15.Bruneau BG, Logan M, Davis N, et al. Chamber-specific cardiac expression of Tbx5 and heart defects in Holt-Oram syndrome. Dev Biol. 1999;211:100–8. doi: 10.1006/dbio.1999.9298. [DOI] [PubMed] [Google Scholar]
- 16.Greulich F, Rudat C, Kispert A. Mechanisms of T-box gene function in the developing heart. Cardiovasc Res. 2011;91:212–22. doi: 10.1093/cvr/cvr112. [DOI] [PubMed] [Google Scholar]
- 17.Hasson P, DeLaurier A, Bennett M, et al. Tbx4 and Tbx5 acting in connective tissue are required for limb muscle and tendon patterning. Dev Cell. 2010;18:148–56. doi: 10.1016/j.devcel.2009.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cross SJ, Ching YH, Li QY, et al. The mutation spectrum in Holt-Oram syndrome. J Med Genet. 2000;37:785–7. doi: 10.1136/jmg.37.10.785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McDermott DA, Bressan MC, He J, et al. TBX5 genetic testing validates strict clinical criteria for Holt-Oram syndrome. Pediatr Res. 2005;58:981–6. doi: 10.1203/01.PDR.0000182593.95441.64. [DOI] [PubMed] [Google Scholar]
- 20.Debeer P, Race V, Gewillig M, Devriendt K, Frijns JP. Novel TBX5 mutations in patients with Holt-Oram syndrome. Clin Orthop. 2007;462:20–26. doi: 10.1097/BLO.0b013e3181123ffe. [DOI] [PubMed] [Google Scholar]
- 21.Ellis DG. Chest wall deformities. Pediatr Rev. 1989;11:147–51. doi: 10.1542/pir.11-5-147. [DOI] [PubMed] [Google Scholar]
- 22.Bickley SRB, Logan MPO. Regulatory modulation of the T-box gene Tbx5 links development, evolution, and adaptation of the sternum. Proc Natl Acad Sci USA. 2014;111:17917–22. doi: 10.1073/pnas.1409913111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Weinstein SL, Dolan LA, Cheng JCY, Danielsson A, Morcuende JA. Adolescent idiopathic scoliosis. Lancet Lond Engl. 2008;371:1527–37. doi: 10.1016/S0140-6736(08)60658-3. [DOI] [PubMed] [Google Scholar]
- 24.Tseng YR, Su YN, Lu FL, et al. Holt-Oram syndrome with right lung agenesis caused by a de novo mutation in the TBX5 gene. Am J Med Genet A. 2007;143A:1012–4. doi: 10.1002/ajmg.a.31672. [DOI] [PubMed] [Google Scholar]
- 25.Qin X, Wei W, Fangqi G. Horseshoe lung associated with Holt-Oram syndrome. Iran J Pediatr. 2015;25:e251. doi: 10.5812/ijp.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Böhm J, Heinritz W, Craig A, et al. Functional analysis of the novel TBX5 c.1333delC mutation resulting in an extended TBX5 protein. BMC Med Genet. 2008;9:88. doi: 10.1186/1471-2350-9-88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Arora R, Metzger RJ, Papaioannou VE. Multiple roles and interactions of Tbx4 and Tbx5 in development of the respiratory system. PLoS Genet. 2012;8:e1002866. doi: 10.1371/journal.pgen.1002866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Boogerd CJJ, Dooijes D, Ilgun A, et al. Functional analysis of novel TBX5 T-box mutations associated with Holt-Oram syndrome. Cardiovasc Res. 2010;88:130–9. doi: 10.1093/cvr/cvq178. [DOI] [PubMed] [Google Scholar]
- 29.Barnett P, Postma AV. Molecular genetics of Holt-Oram syndrome. In: John Wiley & Sons Ltd (ed). eLS. Wiley, Chichester, 2014. http://doi.wiley.com/10.1002/9780470015902.a0024329. Accessed 31 May 2016.
- 30.Al-Qattan MM, Abou Al-Shaar H. Molecular basis of the clinical features of Holt-Oram syndrome resulting from missense and extended protein mutations of the TBX5 gene as well as TBX5 intragenic duplications. Gene. 2015;560:129–36. doi: 10.1016/j.gene.2015.02.017. [DOI] [PubMed] [Google Scholar]
- 31.Borozdin W, Wright MJ, Hennekam RCM, et al. Novel mutations in the gene SALL4 provide further evidence for acro-renal-ocular and Okihiro syndromes being allelic entities, and extend the phenotypic spectrum. J Med Genet. 2004;41:e102. doi: 10.1136/jmg.2004.019505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Al-Baradie R, Yamada K, St Hilaire C, et al. Duane radial ray syndrome (Okihiro syndrome) maps to 20q13 and results from mutations in SALL4, a new member of the SAL family. Am J Hum Genet. 2002;71:1195–9. doi: 10.1086/343821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Koshiba-Takeuchi K, Takeuchi JK, Arruda EP, et al. Cooperative and antagonistic interactions between Sall4 and Tbx5 pattern the mouse limb and heart. Nat Genet. 2006;38:175–83. doi: 10.1038/ng1707. [DOI] [PubMed] [Google Scholar]
- 34.Terrett JA, Newbury-Ecob R, Cross GS, et al. Holt-Oram syndrome is a genetically heterogeneous disease with one locus mapping to human chromosome 12q. Nat Genet. 1994;6:401–4. doi: 10.1038/ng0494-401. [DOI] [PubMed] [Google Scholar]
- 35.Spielmann M, Mundlos S. Looking beyond the genes: the role of non-coding variants in human disease. Hum Mol Genet. 2016;25:R157–R165. doi: 10.1093/hmg/ddw205. [DOI] [PubMed] [Google Scholar]
- 36.Smemo S, Campos LC, Moskowitz IP, Krieger JE, Pereira AC, Nobrega MA. Regulatory variation in a TBX5 enhancer leads to isolated congenital heart disease. Hum Mol Genet. 2012;21:3255–63. doi: 10.1093/hmg/dds165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Minguillon C, Nishimoto S, Wood S, Vendrell E, Gibson-Brown JJ, Logan MPO. Hox genes regulate the onset of Tbx5 expression in the forelimb. Dev Camb Engl. 2012;139:3180–8. doi: 10.1242/dev.084814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shimamura A, Alter BP. Pathophysiology and management of inherited bone marrow failure syndromes. Blood Rev. 2010;24:101–22. doi: 10.1016/j.blre.2010.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.VanderMeer JE, Ahituv N. cis-regulatory mutations are a genetic cause of human limb malformations. Dev Dyn Publ Am Assoc Anat. 2011;240:920–30. doi: 10.1002/dvdy.22535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kozma C. Valproic acid embryopathy: report of two siblings with further expansion of the phenotypic abnormalities and a review of the literature. Am J Med Genet. 2001;98:168–75. doi: 10.1002/1096-8628(20010115)98:2<168::AID-AJMG1026>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 41.Mohd Yunos H, Green A. Fetal valproate syndrome: the Irish experience. Ir J Med Sci. 2018 doi: 10.1007/s11845-018-1757-6. [DOI] [PubMed] [Google Scholar]
- 42.Carli D, Garagnani L, Lando M, et al. VACTERL (vertebral defects, anal atresia, tracheoesophageal fistula with esophageal atresia, cardiac defects, renal and limb anomalies) association: disease spectrum in 25 patients ascertained for their upper limb involvement. J Pediatr. 2014;164:458–462. doi: 10.1016/j.jpeds.2013.09.033. [DOI] [PubMed] [Google Scholar]
- 43.Kohlhase J, Taschner PE, Burfeind P, et al. Molecular analysis of SALL1 mutations in Townes-Brocks syndrome. Am J Hum Genet. 1999;64:435–45. doi: 10.1086/302238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hall JG. Thrombocytopenia and absent radius (TAR) syndrome. J Med Genet. 1987;24:79–83. doi: 10.1136/jmg.24.2.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Albers CA, Paul DS, Schulze H, et al. Compound inheritance of a low-frequency regulatory SNP and a rare null mutation in exon-junction complex subunit RBM8A causes TAR syndrome. Nat Genet. 2012;44:435–9, S1–2. doi: 10.1038/ng.1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Petit F, Escande F, Jourdain AS, et al. Nager syndrome: confirmation of SF3B4 haploinsufficiency as the major cause. Clin Genet. 2014;86:246–51. doi: 10.1111/cge.12259. [DOI] [PubMed] [Google Scholar]
- 47.Wiedemann HR, Drescher J. LADD syndrome: report of new cases and review of the clinical spectrum. Eur J Pediatr. 1986;144:579–82. doi: 10.1007/BF00496040. [DOI] [PubMed] [Google Scholar]
- 48.Rohmann E, Brunner HG, Kayserili H, et al. Mutations in different components of FGF signaling in LADD syndrome. Nat Genet. 2006;38:414–7. doi: 10.1038/ng1757. [DOI] [PubMed] [Google Scholar]
- 49.Vega H, Waisfisz Q, Gordillo M, et al. Roberts syndrome is caused by mutations in ESCO2, a human homolog of yeast ECO1 that is essential for the establishment of sister chromatid cohesion. Nat Genet. 2005;37:468–70. doi: 10.1038/ng1548. [DOI] [PubMed] [Google Scholar]
- 50.Vega H, Trainer AH, Gordillo M, et al. Phenotypic variability in 49 cases of ESCO2 mutations, including novel missense and codon deletion in the acetyltransferase domain, correlates with ESCO2 expression and establishes the clinical criteria for Roberts syndrome. J Med Genet. 2010;47:30–37. doi: 10.1136/jmg.2009.068395. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.