Abstract
Purpose
There is paucity of data on the epidemiology of peripapillary choroidal neovascularisartion (PPCNV). Our aim was to determine prevalence of PPCNV in the elderly UK population of Bridlington residents aged ≥65 years.
Methods
Eyes with PPCNV in the Bridlington eye assessment project (BEAP) database of 3475 participants were analysed. PPCNV outline was drawn, its area measured, and clock-hour involvement of disc circumference recorded. Location and shortest distance from the lesion edge to fovea were recorded. Masked grading for age-related maculopathy (ARM)/reticular pseudodrusen (RPD) within the ETDRS grid was assigned for each eye using a modified Rotterdam scale. Peripapillary retinal pigment epithelial (RPE) changes/drusen were recorded. Visual acuity (VA) and demographic details analysed separately were merged with grading data.
Results
PPCNV were identified in ten subjects, and were bilateral in two (20%), a population prevalence of 0.29%, and 0.06% bilaterality. Gender-specific prevalence were 0.36% and 0.19% for females and males, respectively. Age ranged from 66 to 85 years [mean 76.3 (SD 6.4)]. PPCNV were located nasal to disc in 41.7%, measuring 0.46–7.93 mm2 [mean 2.81 mm2 (SD 2.82)]. All PPCNV eyes had peripapillary RPE changes. One subject had no ARM, 1 angioid streaks, and 30% RPD. No direct foveal involvement, or reduced VA attributable to PPCNV was observed.
Conclusion
PPCNV were infrequent in this population, more common in females, and often located nasal to the disc, without foveal extension. Peripapillary degenerative changes were universal, and strong association with ARM was observed in eyes with PPCNV. Typically, PPCNV were asymptomatic with VA preservation.
Introduction
Peripapillary choroidal neovascular membranes (PPCNV) form part of the spectrum of diseases that have potential to cause severe visual loss [1]. They are well-recognized but uncommon, reportedly accounting for less than 10% of all newly presenting choroidal neovascular membranes (CNV) [2, 3]. In a survey by Browning and Fraser [4], PPCNVs were associated with age-related macular degeneration (AMD) in 45% of cases, while 39% were considered idiopathic. PPCNV are also associated with conditions such as inflammatory diseases including presumed ocular histoplasmosis [5], uveitis [6–9], and chorioretinitis [10], and degenerative processes including myopia [11] and angioid streaks [4]. Choroidal osteoma, optic disc drusen and congenital disc anomalies are other rare associations. Our group has previously reported that PPCNV occurred in 9 out of 231 cases of newly presenting CNV in a UK hospital setting (3.9%) [2]. Previous reports on the prevalence and associations of PPCNV are based on case reports or small case series from hospital data. To the best of our knowledge, there are no reports on population prevalence of PPCNV except for our previous report of worse eye prevalence of 0.29% for Grade 4c (PPCNV), compared to 1.8% for neovascular AMD (nAMD) (grade 4b AMD) and 2.5% for geographic atrophy (GA) (grade 4a AMD) [12]. Furthermore, there are no data available in the literature on the population prevalence of asymptomatic PPCNV.
In this study, characteristics of eyes classified as having PPCNV from the Bridlington eye assessment project (BEAP) were investigated.
Methods
The BEAP study methods, including image acquisition and analysis, have been reported previously [12]. In summary, the BEAP is a single centre population-based prevalence study, with the primary objective to investigate the utility of screening for eye disease in an elderly population ≥65 years, using clinical examination by trained optometrists and digital imaging technology. All individuals registered with a General Practitioner in Bridlington and 65 years and older on the 5th November 2002 were eligible for inclusion in the project. Subjects known to be moving in or out of the area during the study, those that were registered blind or partially sighted, bed bound individuals or those known to have significant dementia were excluded from the study. Subjects were invited by letter on a street-by-street basis in ascending numerical order of postcode. When contacted, each subject was invited to telephone the BEAP to make an appointment to be examined. At its completion in March 2006 over 3500 subjects had been examined. All participants were interviewed, in person, by a trained research nurse using structured questionnaire, and examined by one of four specially trained optometrists with structured proforma completed by the research staff. Non-stereoscopic mydriatic fundus photography was performed with a Topcon fundus camera (model TRC NW6S) and a Nikon 10-megapixel camera. Each eye had a 30° colour fundus photograph (CFP) taken centred on the macula. Local ethics committee approval (Ref. no. PB/RH/02/288) was obtained and the research adhered to the tenets of the Declaration of Helsinki. All subjects provided informed consent. Masked image grading was performed in accordance with the International Classification System of AMD, using 30° non-mydriatic colour fundus photographs. All photographs of right and left eyes were graded by a single ophthalmologist (CW) who was trained in image grading at the Central angiographic reading facility (CARF), Belfast, Northern Ireland. One in ten (1 in 10) randomly selected right eye images were sent to the CARF for secondary masked grading by certified graders.
The diagnosis of PPCNV was clinical, and based on surrogate clinical features of CNV (as previously described) [12] directly adjacent to and contiguous with the optic disc. These include definite RPE detachment, haemorrhagic or serous, and/or subretinal or sub-RPE haemorrhages unassociated with any other vascular lesion and/or intraretinal, subretinal or sub-RPE glial tissue, and/or subretinal or sub-RPE neovascular membrane as characterized by grey/yellowish discoloration. All eyes identified as having PPCNV, or questionable lesions were reviewed and scrutinised by a retinal specialist (WMA). Any differences in opinion were sent to CARF for secondary grading. All images were analysed using the Topcon IMAGEnet 2000 programme. All eyes identified with PPCNV (and corresponding contralateral eyes) were subsequently reviewed in greater detail, with demographic details and visual acuity (VA) unknown to the grader. The outline of the PPCNV was drawn and area measured. Location of the lesion was also recorded. The PPCNV area was taken to include only visible membrane (as indicated by a grayish-brown or whitish-grey appearance) and/or areas of sub-retinal or sub-RPE haemorrhage. Areas of obvious peripapillary glial tissue, if associated with haemorrhage were also measured as part of the lesion. Often, the lesion would be located adjacent to the disc, with peripheral edge haemorrhage or exudate. Areas of subretinal fluid (SRF) without any haemorrhage were not in themselves measured as part of the lesion. Only areas of gross exudation immediately adjacent to haemorrhage or membrane were included in the measured area, whereas sparse distal exudates were not. The extent/severity of the PPCNV was recorded using clock hours of involvement of the optic disc circumference. An example of a PPCNV is shown in Fig. 1.
Fig. 1.
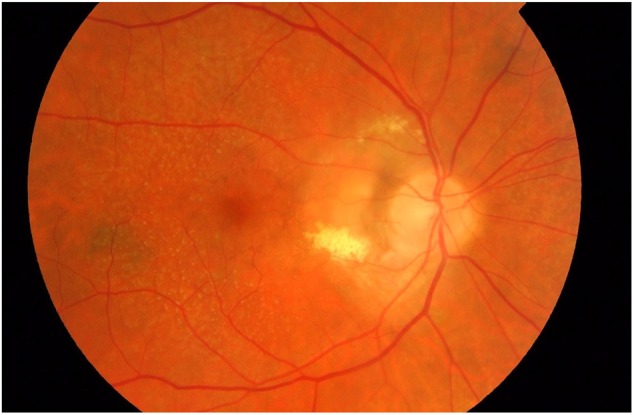
Non-stereoscopic colour fundus photograph. A peripapillary choroidal neovascular membrane involving the temporal 6 clock hours of the optic disc circumference is present, and associated with exudation. There are visible co-morbid reticular pseudodrusen present in the macula and elsewhere
Signs of age-related maculopathy (ARM) within an ETDRS macular grid were recorded using a modified Rotterdam grade as previously described [12]. Retinal pigment epithelial (RPE) changes and presence of drusen in the peripapillary area (one-disc diameter around the optic disc) were specifically recorded along with the closest distance from the edge of changes secondary to the PPCNV, including SRF to the fovea. ARM changes in the contralateral eyes were recorded.
VA and demographic details which were analysed separately were then merged with the image grading data.
Statistical analysis was performed using Stata 12.0 (StataCorp, College Station, TX) and SPSS v.22 (IBM Corp. Armonk, NY). Patient demographic characteristics that are categorical will be summarised using percentages and continuous normally distributed variables with the presentation of means, standard deviation and associated 95% confidence intervals.
Where the continuous variable is non-normally distributed we will present medians and associated quartiles (25th, 75th). Statistical significance will be shown when P-values < 0.05.
Results
Amongst the total of 3475 participants with gradable photographs in at least one eye, PPCNV were identified in ten subjects (Table 1), with bilateral occurrence in two individuals (20%). This resulted in a total of 12 eyes with PPCNV, and an overall population prevalence of 0.29%. PPCNV accounted for 12 (13.3%) of a total of 90 cases of CNVMs identified in the BEAP Study. Seven individuals (70%) were female resulting in gender-specific prevalence rates of 0.36% (7/1939) and 0.19% (3/1536) for females and males respectively. Bilateral involvement equated to a population prevalence of 0.06%. Ages ranged from 66 to 85 years, with a mean of 76.3 years (SD 6.4). There was no obvious increase in prevalence with age, as shown in Table 2 (p = 0.77).
Table 1.
Populatio-based prevalence of PPCNV in the Bridlington eye assessment project (BEAP). Data is number (%) [95%CI]
| Grade | Right eye (3340 gradable photos) | Left eye (3384 gradable photos) | Participants (3475 gradable photos) | Bilateral (3255 participants with gradable photos in both) |
|---|---|---|---|---|
| PPCNV/4c | 6 (0.18) [0.07–0.40] | 6 (0.18) [0.07–0.40] | 10 (0.29) [0.15–0.54] | 2 (0.06) [0.01–0.24] |
| Mean age (years) | 76.7 | 73.6 | 76.3 | 69.5 |
Table 2.
Age (years)-specific prevalence of PPCNV. Data is number (%) [95% CI]
| Age range (years) | 65–69 | 70–74 | 75–79 | 80–84 | 85–89 | >90 | Total |
|---|---|---|---|---|---|---|---|
| Number of PPCNV eyes (%) [95% CI] | 2 (0.24) [0.01–0.91] | 1 (0.09) [0.01–0.58] | 3 (0.37) [0.07–1.14] | 3 (0.56) [0.11–1.72] | 1 (0.55) [0.01– 3.34] | 0 (0.00) [0.00– 12.39] | 10 (0.29) [0.15–0.54] |
| Total gradable eyes available | 849 | 1069 | 808 | 533 | 183 | 33 | 3475 |
Eyes with PPCNV had a higher prevalence of the more advanced stages of AMD when compared to the overall BEAP cohort. The prevalence of Rotterdam grade 2, 3 or 4a within the macula in PPCNV eyes was considerably high at 42% compared to that in the general population (see Tables 3 and 4). Only 1 eye in the ten individuals with a PPCNV had minimal/no sign of ARM present (Rotterdam grade 0).
Table 3.
Summary of demographic details of eyes with PPCNV and associated macular age-related changes (Rotterdam Grades), with LogMAR visual acuity (VA)
| Number | Affected eye | Age at exam | Sex | (Macular status) AMD grade right | (Macular status) AMD grade left | Right VA (LogMAR) | Left VA (LogMAR) | Cause of vision loss |
|---|---|---|---|---|---|---|---|---|
| 1 | LE | 66 | F | 1a | 1a | 0.02 | 0.14 | Nil |
| 2 | LE | 76 | F | 1a | 1a | 0.30 | 0.42 | Other: cataracts |
| 3 | LE | 76 | F | 1a | 1a | 0.1 | 0.1 | Nil |
| 4 | LE | 85 | M | 4b | 4, reticular drusen | CF | 0.3 | ARM |
| 5 | RE | 83 | F | 0a | 0b | 0.2 | 0.2 | Nil |
| 6 | BE | 72 | F | 1a | 1a | 0.1 | 0.1 | Nil |
| 7 | RE | 77 | M | 2a, reticular drusen | 2a, reticular drusen | 0.2 | 0.2 | Nil |
| 8 | RE | 80 | M | 3, reticular drusen | 3, reticular drusen | 0.0 | 0.0 | Nil |
| 9 | BE | 67 | F | 4a | 4a | 0.3 | 0.4 | RE AMD-GA, LE diabetic CSMO |
| 10 (angioid streaks) | RE | 81 | F | 1a | Cannot grade (Non-AMD) | 0.22 | 0.36 | RE-ERM, LE-cataract |
Table 4.
Summary of the peripapillary retinal changes along with size (area) and locations of CNV in relation to the optic disc in all the eyes with PPCNV
| Number | Affected eye | Features of the peripapillary disc area ipsilateral eye | Features of the peripapillary disc area contralateral eye | Maximum drusen diameter (µm) ipsilateral eye | Maximum drusen diameter (µm) contralateral eye | Signs of PPCNVM | Area of PPCNVM (mm2) | Distance from edge of lesion to fovea (mm) | Clock hours (S = superior, I = inferior, N = nasal, T = temporal) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | LE | RPE atrophy | RPE hyperpigmentary crescent and some RPE atrophy | 90 | 210 | RPE changes and subretinal haemorrhage, visible membrane | 3.90 | 2.89 | 3 (11–2 S) |
| 2 | LE | Alpha zone changes with hypopigmentation and drusen visible | A pigmented crescent of RPE hyperpigmentation temporally with more widespread hypopigmentation | 250 | 130 | Peripapillary haemorrhage and RPE changes | 1.39 | 5.68 | 2.5 (6.30–9 IN) |
| 3 | LE | Hypopigmentation around disc with drusen | Focal area of RPE atrophy with scleral show. RPE hyperpigmentation | 250 | 160 | Multiple peripapillary subretinal haemorrhages and exudate | 6.49 | 2.18 | 5 (12.30–5.30 T) |
| 4 | LE | There is a rim of RPE atrophy around the disc with extensive atrophy beyond | Extensive atrophy | 150 | 0 (4b) | Haemorrhage surrounded by disciform scar and atrophy | 1.23 | 2.21 | 1.5 (2.30–4 T) |
| 5 | RE | There is a RPE hyperpigmentary crescent and areas of hypopigmentation and atrophy | Small area of RPE atrophy with more widespread hyperpigmentation/hypopigmentation | 0 | ≤63 | Subretinal haemorrhage | 1.42 | 4.87 | 2 (4-6 IN) |
| 6 | BE | RPE atrophy with hyperpigmentary crescent RE | RPE atrophy LE withy hyperpigmentation and hypopigmentation | 200 RE 220 LE | NA | RE-multiple haems and atrophy LE-Haemorrhages and atrophy | 0.58 RE 0.46 LE | 3.20 RE 3.34 LE | RE: 1.5 (7.5–9 IT) LE: 1.5 (3–4.30 ST) |
| 7 | RE | RPE atrophy with more widespread hypopigmentation | RPE atrophy with more widespread hypopigmentation | 190 | 210 | Subretinal haemorrhage | 0.63 | 5.07 | 2 (12–2 SN) |
| 8 | RE | RPE atrophy and reticular drusen | RPE atrophy and reticular drusen | 170 | 190 | Gross exudation, visible membrane, retinal thickening | 7.93 | 1.52 | 6 (12–6 T) |
| 9 | BE | RPE atrophy and hyperpigmentation with drusen RE | RPE atrophy and hyperpigmentation and hypopigmentation | 450 | 230 | RE: Gross exudation, haemorrhage and retinal thickening LE: subretinal haemorrhage and pigmentary changes | RE: 7.21 LE: 1.49 | RE: 6.29 LE: 3.19 | RE: 5 (12–5 N) LE: 1 (1-2 ST) |
| 10 | RE | Angioid streaks with RPE atrophy and hyperpigmentation | Angioid streaks | 265 | Cannot grade as poor quality photo, but questionable PPCNVM with haemorrhage | Haemorrhage with exudate | 0.94 | 5.59 | 1 (4.5–5.5 IN) |
One subject (10%) had identifiable angioid streaks. This individual was removed from our initial report of AMD prevalence [12] as the pathology was not felt to be purely age-related. The patient was 81 years of age and had co-morbid large soft drusen within the macula. No individuals with PPCNV had pathological myopia or optic disc pathology, such as disc drusen or disc swelling, or evidence of previous chorioretinitis. None of the eyes with PPCNV had direct involvement of their fovea with SRF, exudate or haemorrhage in the photographs. Visual function was good in all eyes, with none having poor VA secondary to PPCNV. Eyes with reduced vision had co-morbid conditions as summarised in Table 3. Patient number 10 (LogMAR VA 0.22, Snellen VA = 20/32) had a co-morbid epiretinal membrane. Patient 2 (LogMAR VA of 0.42 [Snellen 20/50]), subsequently underwent cataract surgery and postoperatively had a VA of 0.2 [Snellen 20/30]. Subject 4 had a LogMAR VA of 0.3 [Snellen 20/40] at presentation. This eye had a 23-months follow-up in a hospital eye service, after which the patient was reported to have a haemorrhagic peripapillary scar with atrophy and discharged from further follow-up. The VA reduction was attributed in part to secondary macula RPE atrophic changes.
Table 3 details demographic and macular changes in participants with PPCVN. Table 4 summarises the peripapillary retinal changes, along with size (area) and locations of CNV in relation to the optic disc in all eyes with PPCNV. Three eyes of 3 subjects (30%) with PPCNV had reticular pseudodrusen (RPD). Nine out of ten individuals (90%) had evidence of drusen ≥63 µm in size within the macular area, including the one with angioid streaks. One individual (10%) had contralateral nAMD; another had bilateral GA with multiple large drusenoid pigment epithelial detachments that appeared completely separate from any PPCNV (which was nasal to the disc). All eyes with PPCNV had RPE hyperpigmentary, hypopigmentary or atrophic changes around the optic disc. One eye (8.33%) had a large PPCNV involving ≥6 clock hours. Six of the 12 identified PPCNM had predominantly temporal location with another occurring superiorly. Five of 12 PPCNVs (41.7%) involved retina nasal to the optic disc only. The PPCNV size ranged from 0.46 mm2 to 7.93 mm2 with a mean of 2.81 mm2 (SD 2.82, 95% CI 1.1–4.6). The mean area of PPCNV in the four eyes with visible exudation was 5.64 mm2 (95% CI 0.57–10.72). For the eight eyes with no visible exudation, the membranes measured an average of 1.39 mm2 (95% CI 0.47–2.31).
Discussion
This study reports the prevalence of PPCNV within an elderly Caucasian population, using data from the largest UK population based screening study of AMD to date. To the best of our knowledge, this represents the first population study specifically to report on the prevalence of PPCNV, as well as describe the characteristics of PPCNV and concomitant ocular findings in affected and fellow eyes, as all previous publications on the subject were based on hospital cohorts, in predominantly symptomatic individuals, with inherent selection bias. Our results indicate that as many as two thirds of PPCNV may remain asymptomatic, compatible with the finding that 40% of the lesions are located nasal to the disc.
PPCNVs (grade 4c AMD) were an infrequent finding, considerably lower than the prevalence of grade 4b AMD, in the same population [12]. Our findings are similar to those of some previous published studies [13, 14], where there was a female preponderance. RPD are known to have such female predilection [12, 15]. However, it is unlikely that the increased female prevalence of PPCNV in this study is due to the presence of RPD, as the three patients with RPD and PPCNV in the series were male. We have previously reported that PPCNV accounted for 3.9% of newly diagnosed cases of CNV in AMD in a hospital eye service [2]. Other investigators have reported frequency of <10% of all CNVs [16]. This relatively low prevalence is confirmed in this study, where PPCNV accounted for 13.3% of all identified CNVs.
The majority of PPCNVs in this cross-sectional study were unilateral, similar to what was reported at baseline in hospital based longitudinal studies [4, 14]. In the Silvestri study [14], only two out of the 14 (14%) individuals had bilateral PPCNV at their initial presentation, whilst others developed contralateral eye involvement over follow up of up to 7 years resulting in 54% bilteralism [14]. Similarly, Browning et al. reported bilateral involvement in 19 of their 96 patients (19.8%) [4] after a median follow up of 2 years.
There is a myriad of reported associations between PPCNV and other conditions, most of which are based on single case reports or small case series. Larger hospital-based studies also exist, but may give a poor representation of the true spectrum of disease as small, nasal, age-related PPCNVs may remain asymptomatic. In a series of 115 eyes of 96 patients, Browning et al. reported ocular conditions associated with PPCNV as 45.2% ARM, 39.1% idiopathic, 4.3% multifocal choroiditis, 2.6% angioid streaks, 1.7% presumed ocular histoplasmosis, 1.7% choroidal osteoma, 0.9% optic disc drusen and 0.9% congenital disc anomalies [4]. The definition of ARM was however broad, and included all eyes with ≥1 of drusen > 63 µm, pigment clumps, mottled pigment epithelial atrophy, GA and signs of an exudative AMD, including disciform scars in both eyes [4]. They also reported that 39% of subjects with PPCNV had drusen on colour photography [4]. Kies and Bird similarly reported 15 of their 55 eyes (27%) with PPCNV had identifiable drusen [13], and Silvestri et al. reported that 60% of PPCNV were related to age-related degenerative changes [14]. In the present series, the presence of drusen ≥ 63 µm within the macular area in 90% of cases was much higher than previously published. This higher prevalence of PPCNV in eyes with the more advanced stages of AMD when compared to the overall BEAP cohort could be a chance finding, but suggests a stronger association of PPCNV with ARM than previously published. The lower prevalence of ARM reported within hospital populations could, in part, be explained by the fact that these are symptomatic, and more likely to be large lesions with macula involvement from exudation or SRF which could mask drusen or result in their regression. Alternatively, it could reflect the older age of the cohort included in the present study. RPD or subretinal drusenoid deposits (SDD) cover a large area of the retina including the peripapillary zone [15, 17], and are known to be associated with nAMD [18–20]. We have previously reported their prevalence in 22% of eyes with newly presenting nAMD in a hospital eye service [19], a finding similar to that in participants with PPCNV in this population.
Peripapillary degenerative changes in the present series were a more universal finding when compared to the presence of macular drusen. Previous studies on PPCNV have not reported the presence of drusen or pigmentary change in the immediate peripapillary area. Recent studies have reported the presence of peripapillary SDD in association with drusen and other age-related changes using multimodal imaging [15, 17]. As drusen and pigmentary changes within the macula are known hallmarks of both GA and CNV, and SDD predispose to CNV, it seems logical to consider these changes in the peripapillary area as potentially pathological for PPCNV. Older reports have speculated on potential relationship of previous unwitnessed episodes of multiple white dot syndromes in the aetiology of the relatively large cohort of presumed idiopathic PPCNVs [21, 22]. A more plausible explanation would be that localized, age-related changes occurring in the peripapillary area may predispose to localized breaks in Bruch’s membrane allowing CNV membranes to develop as reported by Sarks [23].
The association with angioid streaks and PPCNV is well established [24–27]. The number of such cases is small, but suggests that membranes occurring in the region of angioid streaks remain small and asymptomatic and, therefore, are less likely to present to a hospital eye service. Potential overlaps between the aetiologies of PPCNV in patients with angioid streaks and comorbid age-related changes are possible.
Some authors have defined a PPCNV as large if it covered more than 3.5 disc areas or involved over 50% of the disc circumference [28]. Only one subject (10%) in the current series had >6 clock hours involved. This figure is similar to the 15% prevalence of large membranes reported by Browning et al. [4], but less than the higher prevalence of 87% of large membranes involving more than 6 clock hours reported by Kies and Bird [13]. There was complete disc encirclement by the PPCNV in 0.9% of Browning’s cases [4] whilst 11% in Kies and Bird series [13] had similar features [4], probably reflecting selection bias. Caution should however be exercised when comparing the size of CNV lesions, given the different imaging modalities used in the different studies, especially as PPCNV may be larger in surgical excision and indocyanine green angiography than on FFA [13, 28–30].
The finding of asymptomatic PPCNVs in this study is interesting. Our literature search could not find any studies on the prevalence of asymptomatic PPCNV. Sarks provides the best insight on the subject, with a clinicopathological correlative study of 150 eyes of 80 patients obtained post-mortem [23], which identified CNVMs in the peripapillary area in 14%, macular area in 20%, and in the peripheral retina in 24.6% of eyes. Unlike the large temporal PPCNVs seen in eyes of symptomatic individuals which had extended to the fovea [14, 23, 31], Sarks suggested that in older subjects, small and frequently nasal asymptomatic PPCNV occurred with greater frequency than large, temporal membranes with macula involvement, and demonstrated that PPCNVs originate from choroidal vessels passing either through breaks in Bruch’s membrane (57%) or from vessels extending around the termination of Bruch’s membrane (43%) especially on the nasal side [23].
Limitations of our study include utilization only of CFP of field 2. Although the disc and nasal peripapillary areas were well included in these images, the addition of CFP of field 1, centered on the optic disc may have added clearer delineation of nasal PPCNV. Multimodal imaging including SD-OCT around the optic disc, combined with angiography in suspected cases would also have improved case detection, and are recommended in future population studies. Individuals registered as blind or partially sighted were excluded from study inclusion. However, our analysis showed that such subjects were few, and had subfoveal CNV. We cannot be sure that some of these individuals may have had poor vision secondary to PPCNV which could be bilateral or in combination with other visually significant ocular pathology. Such scenarios would make our prevalence measure an underestimation.
In conclusion, this study confirms that PPCNV are an infrequent finding in the elderly population compared to macular CNV, and have a clear female preponderance. PPCNV may be asymptomatic especially when small or located nasally. This finding is very distinct from that in previous hospital studies of symptomatic patients, and supports the early histopathological study [23] which suggested that in elderly individuals, PPCNV frequently occurred in a nasal position and are asymptomatic in the majority of cases. This study also reports the universal finding of peripapillary degenerative changes in all eyes with PPCNV, and a strong association between PPCNV and signs of ARM within both the macula and immediate peripapillary areas plus association with RPD. Multimodal imaging such as OCT, FFA and ICG would help identify possible phenotypic variants. More research is also required on the natural history of these lesions, including those that are located nasally.
Summary
What was known before
There are some data on the occurrence and natural history of peripapillary choroidal neovascularisation (PPCNV) in hospital settings but a paucity of data exists on the epidemiology of PPCNV in community populations.
There is no detailed description of the prevalence and features of asymptomatic PPCNV.
What this study adds
This represents the first study to report PPCNV occurs with a population prevalence of 0.29%, and is bilateral in 0.06% in the UK Caucasian population ≥65 years.
Gender-specific prevalence was 0.36% and 0.19% for females and males, respectively.
PPCNV are commonly located nasal to the optic disc and are small and asymptomatic early on.
Peripapillary RPE degenerative changes, drusen and RPD, as well as higher grade ARM predispose to PPCNV.
Acknowledgements
This research was funded in part by a Research Grant from the Macular Society UK, Andover, Hants, UK. The Bridlington Eye Assessment Project was funded by an unrestricted grant from Pfizer. We would also like to thank the following organisations for financial support of the Project: Pharmacia, Yorkshire Wolds and Coast Primary Care Trust, The Lords Feoffees of Bridlington, Bridlington Hospital League of Friends, The Hull and East Riding Charitable Trust, The National Eye Research Centre (Yorkshire), The Rotary Club of Bridlington, The Alexander Pigott Wernher Memorial Trust, Bridlington Lions Club, The Inner Wheel Club of Bridlington, Soroptimist International of Bridlington, and The Patricia and Donald Shepherd Charitable Trust. The authors thank Sheila MacNab (Project Manager), and Stephen Brown, Janet Button, Graham Langton, and Mark Kunz (Optometrists) for their work with the Project; John Bapty, Nigel Connell, Peter Jay, and Gillian Poole for their work as the charity trustees of the Bridlington Eye Assessment Project.
Author contributions
WMA: consultant for Abbvie, Alimera, Allergan Inc, Bayer, Novartis, Pfizer, Santen, and Thrombogenics, and has undertaken research sponsored by Allergan, Novartis, and Pfizer. He has received speaker fees and travel grants from Allergan, Bausch and Lomb, Bayer, Novartis and Pfizer. JGH: none. HKM: none. RM: none. MM: none. GM: none. AP: none. SAV: consultant for Allergan and Alcon, and has undertaken research sponsored by Allergan and Pfizer. He has received speaker fees from Allergan, Alcon, Pfizer, Optivue and Topcon. CW: none.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Lopez PF, Green WR. Peripapillary subretinal neovascularization. A review. Retina. 1992;12:147–71. doi: 10.1097/00006982-199212020-00012. [DOI] [PubMed] [Google Scholar]
- 2.Wilde C, Patel M, Lakshmanan A, Amankwah R, Dhar-Munshi S, Amoaku W. The diagnostic accuracy of spectral-domain optical coherence tomography for neovascular age-related macular degeneration: a comparison with fundus fluorescein angiography. Eye. 2015;29:602–9. doi: 10.1038/eye.2015.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ruben S, Palmer H, Marsh RJ. The visual outcome of peripapillary choroidal neovascular membranes. Acta Ophthalmol. 1994;72:118–21. doi: 10.1111/j.1755-3768.1994.tb02749.x. [DOI] [PubMed] [Google Scholar]
- 4.Browning DJ, Fraser CM. Ocular conditions associated with peripapillary subretinal neovascularization, their relative frequencies, and associated outcomes. Ophthalmology. 2005;112:1054–61. doi: 10.1016/j.ophtha.2004.11.062. [DOI] [PubMed] [Google Scholar]
- 5.Cantrill HL, Burgess D. Peripapillary neovascular membranes in presumed ocular histoplasmosis. Am J Ophthalmol. 1980;89:192–203. doi: 10.1016/0002-9394(80)90111-7. [DOI] [PubMed] [Google Scholar]
- 6.Arkfeld DF, Brockhurst RJ. Peripapillary subretinal neovascularization in peripheral uveitis. Retina. 1985;5:157–60. doi: 10.1097/00006982-198500530-00005. [DOI] [PubMed] [Google Scholar]
- 7.Garcia CA, Segundo Pde S, Garcia Filho CA, et al. Intermediate uveitis complicated by choroidal granuloma following subretinal neovascular membrane: case reports. Arq Bras Oftalmol. 2008;71:890–3. doi: 10.1590/S0004-27492008000600026. [DOI] [PubMed] [Google Scholar]
- 8.Mehta S, Hariharan L, Ho AC, et al. Peripapillary choroidal neovascularization in pars planitis. J Ophthalmic Inflamm Infect. 2013;3:13. doi: 10.1186/1869-5760-3-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shoughy SS, Jaroudi MO, Tabbara KF. Regression of peripapillary choroidal neovascular membrane in a patient with sarcoidosis after oral steroid therapy. Saudi J Ophthalmol. 2014;28:160–2. doi: 10.1016/j.sjopt.2014.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jampol LM, Orth D, Daily MJ, et al. Subretinal neovascularization with geographic (serpiginous) choroiditis. Am J Ophthalmol. 1979;88:683–9. doi: 10.1016/0002-9394(79)90665-2. [DOI] [PubMed] [Google Scholar]
- 11.Hotchkiss ML, Fine SL. Pathologic myopia and choroidal neovascularization. Am J Ophthalmol. 1981;91:177–83. doi: 10.1016/0002-9394(81)90170-7. [DOI] [PubMed] [Google Scholar]
- 12.Wilde C, Poostchi A, Mehta RL, MacNab HK, Hillman JG, Vernon SA, et al. Prevalence of age-related macular degeneration in an elderly UK Caucasian population—The Bridlington Eye Assessment Project: a cross-sectional study. Eye. 2017;31:1042–50. doi: 10.1038/eye.2017.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kies JC, Bird AC. Juxtapapillary choroidal neovascularization in older patients. Am J Ophthalmol. 1988;105:11–9. doi: 10.1016/0002-9394(88)90114-6. [DOI] [PubMed] [Google Scholar]
- 14.Silvestri G, Archer DB, Johnston PB. Peripapillary subretinal neovascular membranes: the natural history. Eye. 1993;7:398–402. doi: 10.1038/eye.1993.78. [DOI] [PubMed] [Google Scholar]
- 15.Zarubina AV, Neely DC, Clark ME, Huisingh CE, Samuels BC, Zhang Y, et al. Prevalence of subretinal drusenoid deposits in Older Persons with and without age-related macular degeneration by multimodal imaging. Ophthalmology. 2016;123:1090–1100. doi: 10.1016/j.ophtha.2015.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Berkow JW. Subretinal neovascularization in senile macular degeneration. Am J Ophthalmol. 1984;97:143–7. doi: 10.1016/S0002-9394(14)76083-0. [DOI] [PubMed] [Google Scholar]
- 17.Huisingh C, McGwin G, Jr, Neely D, Zarubina A, Clark M, Zhang Y, et al. The association between subretinal drusenoid deposits in older adults in normal macular health and incident age-related macular degeneration. Invest Ophthalmol Vis Sci. 2016;57:739–45. doi: 10.1167/iovs.15-18316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cohen SY, m Dubois L, Tadayani R, Delahaye-Mazza C, Debibie C, Quentel G. Prevalence of reticular pseudodrusen in age-related macular degeneration with newly diagnosed choroidal neovascularisation. Br J Ophthalmol. 2007;91:354–9. doi: 10.1136/bjo.2006.101022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wilde C, Patel M, Lakshmanan A, Morales MA, Dhar-Munshi S, Amoaku WM. Prevalence of reticular pseudodrusen in eyes with newly presenting neovascular age-related macular degeneration. Eur J Ophthalmol. 2015;26:128–34. doi: 10.5301/ejo.5000661. [DOI] [PubMed] [Google Scholar]
- 20.Hogg RE, Silva R, Staurenghi G, Murphy G, Santos AR, Rosina C, et al. Clinical characteristics of reticular pseudodrusen in the fellow eye of patients with unilateral neovascular age-related macular degeneration. Ophthalmology. 2014;121:1748–55. doi: 10.1016/j.ophtha.2014.03.015. [DOI] [PubMed] [Google Scholar]
- 21.Oh KT, Christmas NJ, Russell SR. Late recurrence and choroidal neovascularization in multiple evanescent white dot syndrome. Retina. 2001;21:182–4. doi: 10.1097/00006982-200104000-00017. [DOI] [PubMed] [Google Scholar]
- 22.Wyhinny GJ, Jackson JL, Jampol LM, Caro NC. Subretinal neovascularization following multiple evanescent white-dot syndrome. Arch Ophthalmol. 1990;108:1384–5. doi: 10.1001/archopht.1990.01070120030013. [DOI] [PubMed] [Google Scholar]
- 23.Sarks SH. New vessel formation beneath the retinal pigment epithelium in senile eyes. Br J Ophthalmol. 1973;57:951–65. doi: 10.1136/bjo.57.12.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ballatori N, Clarkson TW. Developmental changes in the biliary excretion of methylmercury and glutathione. Science. 1982;216:61–3. doi: 10.1126/science.7063871. [DOI] [PubMed] [Google Scholar]
- 25.Singerman LJ, Hatem G. Laser treatment of choroidal neovascular membranes in angioid streaks. Retina. 1981;1:75–83. doi: 10.1097/00006982-198101020-00001. [DOI] [PubMed] [Google Scholar]
- 26.Mansour AM, Shields JA, Annesley WH, Jr., et al. Macular degeneration in angioid streaks. Int J Ophthal. 1988;197:36–41. doi: 10.1159/000309915. [DOI] [PubMed] [Google Scholar]
- 27.Lim JI, Bressler NM, Marsh MJ, Bressler SB. Laser treatment of choroidal neovascularization in patients with angioid streaks. Am J Ophthal. 1993;116:414–23. doi: 10.1016/S0002-9394(14)71398-4. [DOI] [PubMed] [Google Scholar]
- 28.Binder S. Surgical treatment of peripapillary choroidal neovascularisation. Br J Ophthalmol. 2007;91:990–1. doi: 10.1136/bjo.2007.114009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kokame GT, Yamaoka S. Subretinal surgery for peripapillary subretinal neovascular membranes. Retina. 2005;25:564–9. doi: 10.1097/00006982-200507000-00004. [DOI] [PubMed] [Google Scholar]
- 30.Wolf S, Wald KJ, Remky A, Arend O, Reim M. Evolving peripapillary choroidal neovascular membrane demonstrated by indocyanine green choroidal angiography. Retina. 1994;14:465–7. doi: 10.1097/00006982-199414050-00014. [DOI] [PubMed] [Google Scholar]
- 31.Gass JD. Drusen and disciform macular detachment and degeneration. Arch Ophthalmol. 1973;90:206–17. doi: 10.1001/archopht.1973.01000050208006. [DOI] [PubMed] [Google Scholar]


