Abstract
Context: Coptidis rhizome (CR), also known as Huanglian in Chinese, is the rhizome of Coptis chinensis Franch., C. deltoidea C.Y. Cheng et Hsiao, or C. teeta Wall (Ranunculaceae). It has been widely used to treat bacillary dysentery, diabetes, pertussis, sore throat, aphtha, and eczema in China.
Objectives: The present paper reviews the latest advances of CR, focusing on the botany, phytochemistry, traditional usages, pharmacokinetics, pharmacology and toxicology of CR and its future perspectives.
Methods: Studies from 1985 to 2018 were reviewed from books; PhD. and MSc. dissertations; the state and local drug standards; PubMed; CNKI; Scopus; the Web of Science; and Google Scholar using the keywords Coptis, Coptidis Rhizoma, Huanglian, and goldthread.
Results: Currently, 128 chemical constituents have been isolated and identified from CR. Alkaloids are the characteristic components, together with organic acids, coumarins, phenylpropanoids and quinones. The extracts/compounds isolated from CR cover a wide pharmacological spectrum, including antibacterial, antivirus, antifungal, antidiabetic, anticancer and cardioprotective effects. Berberine is the most important active constituent and the primary toxic component of CR.
Conclusions: As an important herbal medicine in Chinese medicine, CR has the potential to treat various diseases. However, further research should be undertaken to investigate the clinical effects, toxic constituents, target organs and pharmacokinetics, and to establish criteria for quality control, for CR and its related medications. In addition, the active constituents, other than alkaloids, in both raw and processed products of CR should be investigated.
Keywords: Huanglian, morphology, alkaloids, berberine, pharmacokinetics, toxicity
Introduction
Coptidis rhizome (CR), also known as Huanglian in Chinese, is the rhizome of Coptis chinensis Franch. (Weilian in Chinese), C. deltoidea C.Y. Cheng et Hsiao (Yalian in Chinese), or C. teeta Wall. (Yunlian in Chinese) (Ranunculaceae) (Chinese Pharmacopoeia Commission 2015). Moreover, C. japonica Makino and its variants are also used in Japan (Cho et al. 2001). Large quantities of CR are consumed in Asian countries, such as China, Japan, Malaysia, Singapore and India, but only a small amount is used in European countries (Kong et al. 2013).
CR has been used to treat various inflammatory disorders and related diseases for a thousand years, and has functions of clearing heat, drying dampness and detoxification according to the traditional Chinese Medicinal theory. The medicinal use of this plant was first listed in Shennong’s Classic of Materia Medica in China, which was written during the Han Dynasty. More than 32,000 Chinese Medical formulas mention CR, usually in the form of a powder, pill, decoction or tablet (Wu et al. 2015). It is often utilized to treat diarrhoea, vomiting, abdominal fullness, jaundice, high fever coma, toothache, diabetes and eczema. Modern studies have demonstrated that CR has wide pharmacological activities, including antibacterial, antifungal, antiviral, antihepatic steatosis, anti-atherosclerosis, antimyocardial ischaemia/reperfusion injury, antidiabetic, antiarrhythmia, antihypertention, anti-inflammation, antioxidation and antitumour effects (Ma and Ma 2013; Wang 2016; Dan et al. 2017; Liu D et al. 2017). Currently, over 120 chemical components have been isolated and identified from CR. Apart from its main composition of alkaloids, it also contains organic acids, lignans, flavones, volatile oils, etc. (Yoshikawa et al. 1997a, 1997b; Wang et al. 2014; Chen et al. 2016). The present review provides the overview of CR from 1985 to 2018 in terms of its botany, phytochemistry, traditional usages, pharmacology, pharmacokinetics and toxicology. We also offer some perspectives about the future research into this herbal medicine.
Traditional usages
The rhizome is the main medicinal part of CR, and it is processed by 28 methods before clinical use, some of which are taken from ancient Chinese medicines books (Table 1). From these methods, we observed that CR processing has changed from simple to complex and then from complex to simple (Mei 2008). Nowadays, CR is commonly processed with wine, Zingiber officinale Rosc. (Zingiberaceae) juice, and Evodia rutaecarpa (Juss.) Benth. (Rutaceae) to exert different functions including treating insomnia, sore mouth, red and swelling eyes, preventing vomiting, expelling phlegm and curing diarrhoea (Lei and Dun 2002; Lu 2004; Li 2013; Chinese Pharmacopoeia Commission 2015).
Table 1.
Different processing methods of CR.
| Processing method | Purpose of processing | Dynasty | Reference |
|---|---|---|---|
| Rubbing the fibrous roots with cloth, washing | Removing non-medicinal parts and impurities to ensure curative effect | Before the Tang Dynasty | (Lei 1985) |
| Stir-baking to dark brown | Enhancing the efficacy of digestion and invigorating the function of spleen | Song Dynasty | (Wang 1991) |
| Carbonizing by stir-frying | Producing hemostatic effect | Qing Dynasty | (Chen 2006) |
| Stir-baking with loess | Invigorating the function of spleen andstomach | Jin Yuan Period,Ming Dynasty | (Zhu 2012) |
| Stir-frying with wine | Treating insomnia, sore mouth, red and swelling eyes | Song Dynasty, Jin Yuan Period | (Zhu 2015) |
| Stir-frying with Ginger | Enhancing the effect preventing vomitting, and expelling phlegm | Song Dynasty | (Wang 1991) |
| Stir-frying with bile | Enhancing the function of clearing the fire of the liver and galllbladder | Ming Dynasty | |
| Immersing into rice water | Strengthening the role of nourishing the spleen and harmonizing the spleen and stomach | Song Dynasty, Qing Dynasty | (Qian & Wang 2008) |
| Stir-baking with Evodiae Fructus | Curing diarrhea | Yuan Dynasty | (Zhu 2012) |
| Stir-baking with Rhizoma Zingiberis Recens | Enhancing the effect of preventing vomitting | Song Dynasty | (Dong 2003; Tang 2011) |
| Stir-baking with Sophorae Flos | Treatment of dysentery | Ming Dynasty | (Zhang 1996) |
| Steaming with wine | Curing diarrhea | Ming Dynasty | |
| Steaming with milk | Curing acute conjunctivitis | Ming Dynasty | (Han 1985) |
The medicinal value of CR is worth affirming. Relevant statistics show that in 13 prescriptions before the Song Dynasty, more than 32,000 Chinese Medical formulae mentioned CR. Currently, CR is commonly used as a main traditional Chinese medicine (TCM) to treat respiratory diseases (including tuberculous empyema, whooping cough, and pulmonary candidiasis caused by pneumonia), digestive diseases (including diarrhoea, chronic colitis and upper gastrointestinal infection), paediatric diseases (including hyperthermia of infantile external sensation, dyspepsia and urticaria), dermatological diseases (including acne, psoriasis, dermatitis and tinea pedis), and nervous system diseases (Wu et al. 2015). CR has been employed in the form of powders, pills or decoctions (Table 2).
Table 2.
The traditional and clinical uses of CR in China.
| Preparation | Main compositions | Traditional and clinical uses | References |
|---|---|---|---|
| An Gong Niu Huang Pills | Coptidis Rhizoma, Bovis Calculus, Condensed powder of Bubali Corun, Moschusm or Artificial Moschusm, Margarita, Cinnabaris, Realgar, Scutellariae Radix, Gardeniae Fructus, Curcumae Radix, Syntheticum Borneolum | Curing febrile convulsions, delirious, and gibberish | (Chinese Pharmacopoeia Commission 2015) |
| Dang Gui Long Hui Pills | Coptidis Rhizoma, Angelicae Sinensis Radix, Gentianae Radix et Rhizoma, Rhei Radix et Rhizoma, Scutellariae Radix, Phellodendri Chinensis Cortex, Aloe, Ineigo Naturalis, Gardeniae Fructus, Aucklandiae Radix, Artificial Moschusm | Curing dizziness, tinnitus, deafness, rib pain, abdominal distension pain and constipation | |
| Fu Fang Qing Dai Pill | Coptidis Rhizoma, Cnidii Frucutus, Sophorae Flavescentis Radix, Pseudolaricis Radix, Catechu, Alumen | Treating mycotic vaginitis, trichomonas vaginitis, and nonspecific vaginitis | |
| Huang Lian Shang Qing Pills | Coptidis Rhizoma, Gardeniae Fructus, Forsythiae Fructus, Viticis Fructus, Saposhnikoviae Radix, Schizonepetae Spica, Angelicae Dahuricae Radix, Scutellariae Radix, Rhei Radix et Rhizoma, Chrysanthemi Flos, Menthae Haplocalycis Herba, Phellodendri Chinensis Cortex, Platycodonis Radix, Chuanxiong Rhizoma | Treating dizziness, tooth pain, tongue sores, sore throat, ear pain tinnitus and constipation | |
| Huang Lian Yang Gan Pills | Coptidis Rhizoma, Rhizoma Picrohizae, Scutellariae Radix, Phellodendri Chinensis Cortex, Gentianae Radix et Rhizoma, Bupleuri Radix, Citri Reticulatae Pericarpium Viride, Equiseti Hiemalis Herba, Buddlejae Flos, Leonuri Fructus, Cassiae Semen, Haliotidis Concha | Treating red sore, eye, blurred vision | |
| Kai Guang Fu Ming Pills | Coptidis Rhizoma, Gardeniae Fructus, Phellodendri Chinensis Cortex, Scutellariae Radix, Rhei Radix et Rhizoma, Saposhnikoviae Radix, Chrysanthemi Flos, Gentianae Radix et Rhizoma, Scrophulariae Radix, Paeoniae Radix Rubra, Alismatis Rhizoma, Rehmanniae Radix | Clearing heat and improving eyesight | |
| Mu Xiang Bing Lang Pills | Coptidis Rhizoma, Aucklandiae Radix, Arecae Semen, Aurantii Fructus, Citri Reticulatae Pericarpium, Citri Reticulatae Pericarpium Viride, Cyperi Rhizoma, Sparganii Rhizoma, Curcumae Rhizoma, Phellodendri Chinensis Cortex, Rhei Radix et Rhizoma, Natrii Sulfas, Pharbitidis Semen | Treating abdominal distension pain and constipation | |
| Niu Huang Qian Jin Powder | Coptidis Rhizoma, Scorpio, Bombyx Batryticatus, Bovis Calculus, Cinnabaris, Borneolum Syntheticum, Arisaema Cum Bile, Gastrodiae Rhizoma, Glycyrrhizae Radix et Rhizoma | Clearing heat and detoxifying, calming nerves, curing children convulsion with high fever, hand and foot convulsions | |
| Qin Lian Tablets | Coptidis Rhizoma, Phellodendri Chinensis Cortex, Forsythiae Fructus, Scutellariae Radix, Paeoniae Radix Rubra, Glycyrrhizae Radix et Rhizoma | Treating headache and red eye, mouth and nose sores, hot dysentery, abdominal pain | |
| Shen Shuai Ning Capsules | Coptidis Rhizoma, Radix Pseudostellariae, Praeparatum Pinelliae Rhizoma, Citri Reticulatae Pericarpium, Poria, Rhei Radix et Rhizoma, Glycyrrhizae Radix et Rhizoma, Salviae Miltiorrhizae Radix et Rhizoma, Achyranthis Radix, Carthami Flos | Curing nausea, vomiting, poor appetite, bad urine, stool viscous | |
| Wan Shi Niu Huang Qing Xin Pills | Coptidis Rhizoma, Bovis Calculus, Cinnabaris, Scutellariae Radix, Gardeniae Fructus, Curcumae Radix | Curing high fever irritability, insanity and children febrile convulsion | |
| Wu Mei Pills | Coptidis Rhizoma, Mume Fructus, Asari Radix et Rhizoma, Zingiberis Rhizoma, Aconiti Lateralis Radix Praeparata, Zanthoxyli Pericarpium, Cinnamomi Ramulus, Ginseng Radix et Rhizoma, Phellodendri Chinensis Cortex, Angelicae Sinensis Radix | Curing abdominal pain, headache, mania, vomiting, and limbs cold. | |
| Xiao Ke Ping Tablets | Coptidis Rhizoma, Ginseng Radix et Rhizoma, Trichosanthis Radix, Asparagi Radix, Astragali Radix, Salviae Miltiorrhizae Radix et Rhizoma, Lycii Fructus, Astragali Complanati Semen, Puerariae Lobatae Radix, Anemarrhenae Rhizoma, Galla Chinensis, Schisandrae Chinensis Fructus | Curing diabetes | |
| Xiang Lian Pills | Coptidis Rhizoma, Aucklandiae Radix | Curing enteritis, and bacillary dysentery, relieving pain | |
| Xiong Ju Shang Qing Pills | Coptidis Rhizoma, Chuanxiong Rhizoma, Scutellariae Radix, Viticis Fructus, Menthae Haplocalycis Herba, Schizonepetae Spica, Ligustici Rhizoma et Radix, Saposhnikoviae Radix, Angelicae Dahuricae Radix, Chrysanthemi Flos, Gardeniae Fructus, Forsythiae Fructus, Platycodonis Radix, Glycyrrhizae Radix et Rhizoma, Notopterygii Rhizoma et Radix |
Treating migraine headache, nasal flow toothache, sore throat |
|
| Yi Qing Granules | Coptidis Rhizoma, Rhei Radix et Rhizoma, Scutellariae Radix | Treating pharyngitis, tonsil inflammation and gum inflammation | |
| Zhu Che Pills | Coptidis Rhizoma, Zingiberis Rhizoma Praeparatum, Angelicae Sinensis Radix, Moschus | Nourishing Yin and stopping dysentery, curing abdominal pain, diarrhea | |
| Zuo Jin Pills | Coptidis Rhizoma, Euodiae Fructus | Purging fire, soothing the liver, reconciling the intestines and stomach, analgesiastomach ache, mouth bitter noise, and vomiting | |
| Niu Huang Qing Re Powder | Coptidis Rhizoma, Scutellariae Radix, Gardeniae Fructus, Curcumae Radix, Bovis Calculus, Bubali Corun, Cinnabaris, Borneolum Syntheticum | Treating high fever spasm, limbs twitch, irritability restless, and phlegm turbid congestion | (Zhong 1991) |
| Niu Huang Xing Nao Pills | Coptidis Rhizoma, Bovis Calculus, Bubali Corun, Borneolum Syntheticum, Scutellariae Radix, Gardeniae Fructus, Moschusm, Cinnabaris, Margarita, Curcumae Radix | Curing high fever, coma convulsion, irritable restlessness, infantile convulsionand insomnia | (Zhong 1998) |
| Qing Wei Huang Lian Pills | Coptidis Rhizoma, Glycyrrhizae Radix et Rhizoma, Platycodonis Radix, Gypsum Fibrosum, Anemarrhenae Rhizoma, Moutan Cortex, Trichosanthis Radix, Forsythiae Fructus, Scutellariae Radix, Gardeniae Fructus, Phellodendri Chinensis Cortex | Curing tongue sores, and sore throat | (Zhong 1998) |
| San Huang Pills | Coptidis Rhizoma, Rhei Radix et Rhizoma, Huang Cao | Curing dysentery, vomiting , hemoptysis and constipation | |
| Xiao Er Qing Re Zhen Jing Powder | Coptidis Rhizoma, Arisaema Cum Bile, Scorpio, Bombyx Batryticatus, Glycyrrhizae Radix et Rhizoma, Bovis Calculus, Cinnabaris, Borneolum Syntheticum, Bambusae Concretio Silicea | Curing hot convulsion, hand-foot convulsions, cough, irritability and thirst | (Zhong 1991) |
| San Huang Qing Jie Pills | Coptidis Rhizoma, Scutellariae Radix, Forsythiae Fructus, Phellodendri Chinensis Cortex, Lonicerae Japonicae Flos | Curing fever, cough, sore throat, hot leaching and diarrhea | (Guo 2002) |
| Xie Li Xiao Pills | Coptidis Rhizoma, Atractylodis Rhizoma, Alba Paeoniae Radix, Aucklandiae Radix, Euodiae Fructus, Magnoliae Officinalis Cortex, Arecae Semen, Aurantii Fructus, Citri Reticulatae Pericarpium, Alismatis Rhizoma, Poria, Glycyrrhizae Radix et Rhizoma | Treating acute enteritis, colitis, and dysentery | |
| Huang Lian Jie Du Pills | Coptidis Rhizoma, Phellodendri Chinensis Cortex, Scutellariae Radix, Rhei Radix et Rhizoma, Talcum, Clematidis Armandii Caulis, Gardeniae Fructus | Treating sore mouth, headache, constipation, red eyes, heartburn, sore throat, | |
| Geng Nian Xin Capsules | Coptidis Rhizoma, Cinnamomi Cortex, Alpiniae Oxyphyliae Fructus, Lycii Fructus, Corni Fructus, Ligustri Lucidi Frucrus, Cuscutae Semen, Acori Tatarinowii Rhizoma, Rehmanniae Radix, Polygalae Radix, Ziziphi Spinosae Semen, Citri Reticulatae Pericarpium, Alismatis Rhizoma | Curing heart palpitations, insomnia, dizziness, tinnitus, and backache | (Chinese Pharmacopoeia Commission 2008) |
Botany
Coptis chinensis (Figure 1(A)) is a perennial herb with yellow, branched rhizomes. The leaves are slightly leathery, with three lobes (Xiao 2002). The scapes are 12–25 cm high. In addition, 3–8 flowers are clustered into a dichasium or pleiochasium. The five sepals, 9–12.5 mm in length, 2–3 mm in width, are greenish yellow and oblong ovate. There are approximately 20 stamens with 8–12 carpels, which are slightly curved outside. The 6–12 follicles are 6–8 mm in length with a thin handle. There are 7–8 brown, oblong seeds that are 2 mm long and 8 mm wide. Flowering occurs from February to March, and the fruit is commonly harvested from April to June. It is distributed in Sichuan, Guizhou, Hunan, Hubei, and southern Shaanxi in China. This plant grows in mountain forests or valleys at an altitude of approximately 500–2000 m ( Flora 2004).
Figure 1.
The whole plants and rhizomes of C. chinensis (A), C. chinensis (B) and C. teeta (C).
C. deltoidea (Figure 1(B)) is also a perennial herb with unbranched or few branched yellow rhizomes. The 3–11 leaves are oval and slightly leathery, are 16 cm long and 15 cm wide and are finely divided into three parts. The one or two scapes are slightly longer than the leaves. The plant produces 4–8 flowers, which are clustered into a blue-green inflorescence. Sepals are yellow-green, narrow ovoid, 8–12.5 mm long, and 2–2.5 mm wide. There are approximately 20 stamens, which are about half the length of the petals. The anther is yellow, and the filament is narrowly linear. The flowering period is March and April and the fruit are harvested from April to June. It is native to the areas of Emei and Hongya in Sichuan province. This plant grows in mountain forests with an altitude approximately 1600–2200 m (Flora 2004).
C. teeta (Figure 1(C)) is an often used as a folk medicine in Yunnan Province of China. It is a perennial herb with yellow rhizomes yellow, dense internodes and mostly fibrous roots. The blade comprises oval-shaped triangles that are 6–12 cm long and 5–9 cm wide, with a triple fissure. C. teeta has one or two scapes and is 15–25 cm high during the fruiting period. It has a blue-green inflorescence with 3–5 flowers. The yellow-green, oval calyx is 7.5–8 mm long and 2.5–3 mm wide. The anther is about 0.8 mm long and filament is 2–2.5 mm long. C. teeta is commonly distributed in Yunnan and Tibet provinces of China, and in Burma. C. teeta commonly grows in the shade of cold and damp mountainous areas with an altitude of approximately 1500–2300 m ( Flora 2004) .
The major morphological differences among the rhizomes of these three plants is that Weilian is curved, branched, clustered, and shaped like chicken’s feet; Yalian is less branched and cylindrical; while Yunlian is the smallest and is shaped like a scorpion’s tail. In this review, we will mainly discuss the advances in research into CR from Coptis chinensis, which is the most common source for CR.
Phytochemistry
The first investigation concerning the chemical components of CR, which succeeded in isolating berberine (1), was reported in 1862 from C. teeta (Perrins 1862). To date, over 100 chemical constituents have been isolated and identified. Alkaloids are the most abundant among these chemical components and are considered as the main active ingredients of CR. Besides alkaloids, CR contains organic acids, coumarins, phenylpropanoids, quinones and other chemical components. In this section, the structures of the main compounds of CR are described and drawn (Table 3; Figures 2–10).
Table 3.
Partial list of chemical compounds isolated from CR.
| Classification | Number | Ingredient name | Reference |
|---|---|---|---|
| Alkaloids | 1 | Berberine | (Noguchi et al. 1978) |
| 2 | Berberrubine | (Li ZF et al. 2012) | |
| 3 | Coptisine | (Wang et al. 2014) | |
| 4 | Palmatine | ||
| 5 | Epiberberine | (Mizuno et al. 1992) | |
| 6 | Columbamine | (Ikuta and Itokawa 1989) | |
| 7 | Tetradehydroscoulerine | (Chen et al. 2008) | |
| 8 | Jatrorrhizine | (Li ZF et al. 2012) | |
| 9 | Groenlandicine | ||
| 10 | Berberastine | (Li ZF et al. 2012) | |
| 11 | Worenine | ||
| 12 | 8-Oxyberberine | (Wang et al. 2014) | |
| 13 | 8-Oxycoptisine | ||
| 14 | 3-Hydroxy-2-methoxy-9,10-methylenedioxy-8-oxyprotoberberine | (Zhao et al. 2010) | |
| 15 | 8-Oxyepiberberine | (Yang et al. 2014) | |
| 16 | 8-Oxyberberrubine | ||
| 17 | (−)-5-Hydroxyl-8-oxyberberine | (Wang et al. 2014) | |
| 18 | (+)-5-Hydroxyl-8-oxyberberine | ||
| 19 | Tetrahydroberberine | (Wang et al. 2014) | |
| 20 | 8,13-Dioxocoptisine hydroxide | (Yang et al. 2014) | |
| 21 | 1,3-Dioxolo[4,5-g]isoquinolin-5(6H)-one | (Wang et al. 2007) | |
| 22 | Noroxyhydrastinine | ||
| 23 | Corydaldine | (Ma et al. 2013) | |
| 24 | Thalifoline | (Li ZF et al. 2012) | |
| 25 | 6-([1,3]Dioxolo[4,5-g]isoquinoline-5-carbonyl)-2,3-dimethoxy benzoic acid methyl ester | (Wang et al. 2014) | |
| 26 | Berbithine | ||
| 27 | Coptisonine | (Yang et al. 2014) | |
| 28 | Tetrandrine | ||
| 29 | Obamegine | ||
| 30 | Magnoflorine | (Tomita and Kura 1956) | |
| 31 | Sanguinarine | (Mizuno et al. 1988) | |
| 32 | Norsanguinarine | ||
| 33 | Oxysanguinarine | ||
| 34 | 6-Acetonyl-5,6-dihydrosanguinarine | ||
| 35 | Chilenine | (Yang et al. 2014) | |
| 36 | Z-N-Ferulyltyramine | (Li ZF et al. 2012) | |
| 37 | E-N-Feruloyltyramine | (Ma H et al. 2013) | |
| 38 | 3-Hydroxy-1-(4-hydroxyphenethyl) pyrrolidine-2,5-dione | (Li ZF et al. 2013) | |
| 39 | 4′-[Formyl-5-(hydroxymethyl)-1H-pyrrol-1-yl] butanoate | (Ma H et al. 2013) | |
| 40 | 8,9-Dihydroxy-1,5,6,10-β-tetrahydro-2H-pyrrolo[2,1-α]-isoquinolin-5-one | (Li, et al. 2012) | |
| 41 | Ehyl-2-pyrrolidinone-5(S)-carboxylate | ||
| 42 | Methyl-5-hydroxy-2-pyridinecarboxylate | ||
| 43 | 1H-indole-3-carboxaldehyde | ||
| 44 | Choline | (Chen L et al. 2012) | |
| Lignans | 45 | Woorenogenin | (Chen et al. 2016) |
| 46 | Woorenoside I | (Yoshikawa et al. 1995) | |
| 47 | Longifolroside A | (Meng et al. 2013) | |
| 48 | Woorenoside II | (Yoshikawa et al. 1995) | |
| 49 | Woorenoside V | ||
| 50 | Woorenoside III | ||
| 51 | Woorenoside IV | ||
| 52 | (+)-Pinoresinol | ||
| 53 | (+)-Medioresinol | ||
| 54 | (+)-Pinoresinol glucoside | ||
| 55 | (+)-Pinoresinol-4,4′-O-β-d-diglucopyranoside | (Yoshikawa et al.1997) | |
| 56 | (+)-Syringaresinol glucoside | (Meng et al. 2013) | |
| 57 | (+)-Lariciresinol | (Hirano et al. 1997) | |
| 58 | (±)-5,5′-Dimethoxylariciresinol | (Li XG et al. 2012) | |
| 59 | (+)-5′-Methoxylariciresinol | (Chen L et al. 2012) | |
| 60 | (+)-Lariciresinol glucoside | (Chen et al. 2016) | |
| 61 | 7S, 8R, 8′R-(+)-Lariciresinol-4,4′-O-β-d-diglucopyranoside | (Yoshikawa et al.1997) | |
| 62 | Lanicepside A | (Chen et al. 2016) | |
| 63 | 9-Acetyl lanicepside B | ||
| 64 | (+)-Isolariciresinol | ||
| 65 | Isolarisiresinol-9-O-β-d-glucopyranoside | (Li XG et al. 2012) | |
| 66 | Woorenoside XI | (Yoshikawa et al.1997) | |
| 67 | Cleomiscosin A | (Mizuno et al. 1992) | |
| 68 | Aquillochin | ( Min et al. 1987) | |
| 69 | 2,3-bis[(4-Hydroxy-3,5-dimethoxyphenyl)-methyl]-1,4-butanediol | ||
| 70 | secoisolariciresinol | (Li XG et al. 2012) | |
| 71 | Erythro-gaiacylglycerol-8-O-4′-(coniferylalcohol) ether | (Chen L et al. 2012) | |
| 72 | Threo-guaiacylglycerol-8-O-4′-(coniferyl alcohol) ether | ||
| 73 | Woorenoside X | (Yoshikawa et al.1997) | |
| 74 | Dihydrodehydrodiconiferyl alcohol | (Li XG et al. 2012) | |
| 75 | Wooreno | (Yoshikawa et al.1997) | |
| Simple phenylpropanoids | 76 | Z-Octadecyl cafeate | (Yang et al. 2014) |
| 77 | E-3-Methoxycinnamic acid | (Ma H et al. 2013) | |
| 78 | Ferulic acid | (Li XG et al. 2012) | |
| 79 | Ethyl ferulate | (Yoshikawa et al. 1995) | |
| 80 | N-Butyl ferulate | (Ma H et al. 2013) | |
| 81 | p-Hydroxyphenethyl E-ferulate | (Hirano et al. 1997) | |
| 82 | E-3,4-Dimethoxycinnamic acid | (Ma H et al. 2013) | |
| 83 | 4-O-Feruloylquinic acid | (Li XG et al. 2012) | |
| 84 | Methyl 4-O-feruloylquicinate | (Li XG et al. 2012) | |
| 85 | Ethyl 4-O-feruloylquicinate | (Ma H et al. 2013) | |
| 86 | 4-O-Feruloylquinic acid butyl ester | (Li XG et al. 2012) | |
| 87 | 5-O-Feruloylquinic acid | (Li XG et al. 2012) | |
| 88 | Methyl 5-O-feruloylquicinate | ||
| 89 | Ethyl 5-O-feruloylquicinate | ||
| 90 | 5-O-Feruloylquinic acid butyl ester | (Ma H et al. 2013) | |
| 91 | Chlorogenic acid | (Chen L et al. 2012) | |
| 92 | Methyl 3-O-feruloylquicinate | (Li XG et al. 2012) | |
| 93 | N-Butyl 3-O-feruloylquicinate | (Ma H et al. 2013) | |
| 94 | 3-(4′-Hydroxyphenyl)-(2R)-lactic acid | (Li XG et al. 2012) | |
| 95 | 3-(3′,4′-Hydroxyphenyl)-(2R)-lactic acid | (Yahara et al. 1985) | |
| 96 | 3-(3′,4′-Dihydroxyphenyl)-(2R)-lactic acid-4′-O-β-d-glucopyranoside | ||
| 97 | Methyl-3-(4′-O-β-d-glucopyranosyl-3′,4′-dihydroxyphenyl)-lactate | (Yoshikawa et al.1997) | |
| 98 | Methyl-3,4-dihydroxyphenyl lactate | (Li XG et al. 2012) | |
| 99 | Ethyl-3,4-dihydroxyphenyl lactate | (Ma H et al. 2013) | |
| 100 | N-Butyl-3,4-dihydroxyphenyl lactate | ||
| 101 | 3-(2,3,4-Trihydroxyphenyl) propanoic acid | (Li XG et al. 2012) | |
| Flavonoids | 102 | 6,8-Dimethyl-3,5,7-trihydroxyfavone | (Meng et al. 2013) |
| 103 | Rhamnetin | (Chen L et al. 2012) | |
| 104 | Wogonin | ||
| 105 | 7,4′-Dihydroxy-5-methoxyfavanone | (Min et al. 1987) | |
| 106 | 2′,4,4′-Trihydroxy-6′-methoxydihydrochalcone | ||
| 107 | Coptiside I | (Fujiwara et al. 1976) | |
| 108 | Coptiside II | ||
| 109 | Woorenoside XII | (Yoshikawa et al.1997) | |
| Other compounds | 110 | Limonin | (Wang et al. 2007) |
| 111 | 3,4-Dihydroxyphenylethyl alcohol | (Li XG et al. 2012) | |
| 112 | 3′,4′-Dihydroxyphenethyl alcohol 1-O-β-d-glucopyranoside | (Yahara et al. 1985) | |
| 113 | 3,5-Dihydroxyphenethyl alcohol-3-O-β-d-glucopyranoside | (Meng et al. 2013) | |
| 114 | Protocatechuic aldehyde | (Ma H et al. 2013) | |
| 115 | Gentisic acid-5-O-β-d-glucopyranoside | (Yahara et al. 1985) | |
| 116 | Apocynol | (Ma H et al. 2013) | |
| 117 | 1,2-Dihydroxy-benzene | (Li ZF et al. 2012) | |
| 118 | Protocatechuic acid | (Meng et al. 2013) | |
| 119 | Vanillic acid | (Li ZF et al. 2012) | |
| 120 | Vanillic acid-4-O-β-d-glucopyranoside | ||
| 121 | Protocatechuic acid methyl ester | (Ma H et al. 2013) | |
| 122 | Protocatechuic acid ethyl ester | (Wang et al. 2012) | |
| 123 | Woorenoside VI | (Yoshikawa et al.1997) | |
| 124 | Woorenoside VII | ||
| 125 | Woorenoside VIII | ||
| 126 | Woorenoside IX | ||
| 127 | cyclo-(Phe-Val) | (Li ZF et al. 2012) | |
| 128 | cyclo-(Phe-Leu) | ||
| 129 | β-Sitosterol | (Yang et al. 2014) |
Figure 2.
Subtypes of protoberberines in Coptidis Rhizoma.
Figure 3.
Matrices of isoquinolines in Coptidis Rhizoma.
Figure 4.
Alkaloids numbered 1–27 in Coptidis Rhizoma.
Figure 5.
Alkaloids numbered 28–44 in Coptidis Rhizoma.
Figure 6.
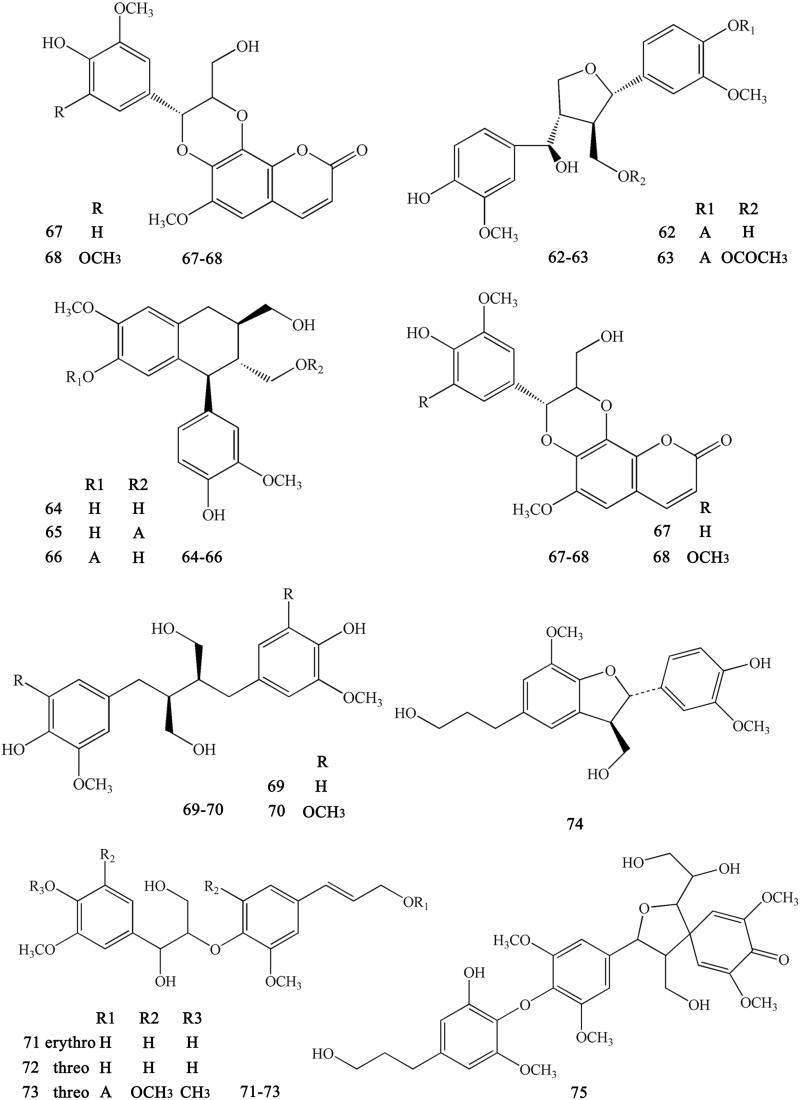
Lignans numbered 45–66 in Coptidis Rhizoma.
Figure 7.
Lignans numbered 67–75 in Coptidis Rhizoma.
Figure 8.
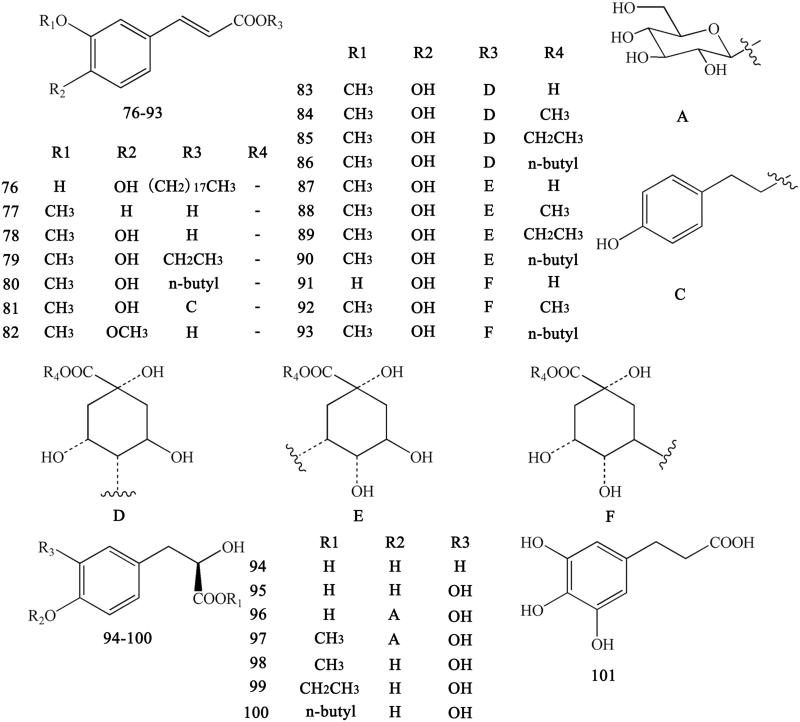
Simple phenylpropanoids in Coptidis Rhizoma.
Figure 9.
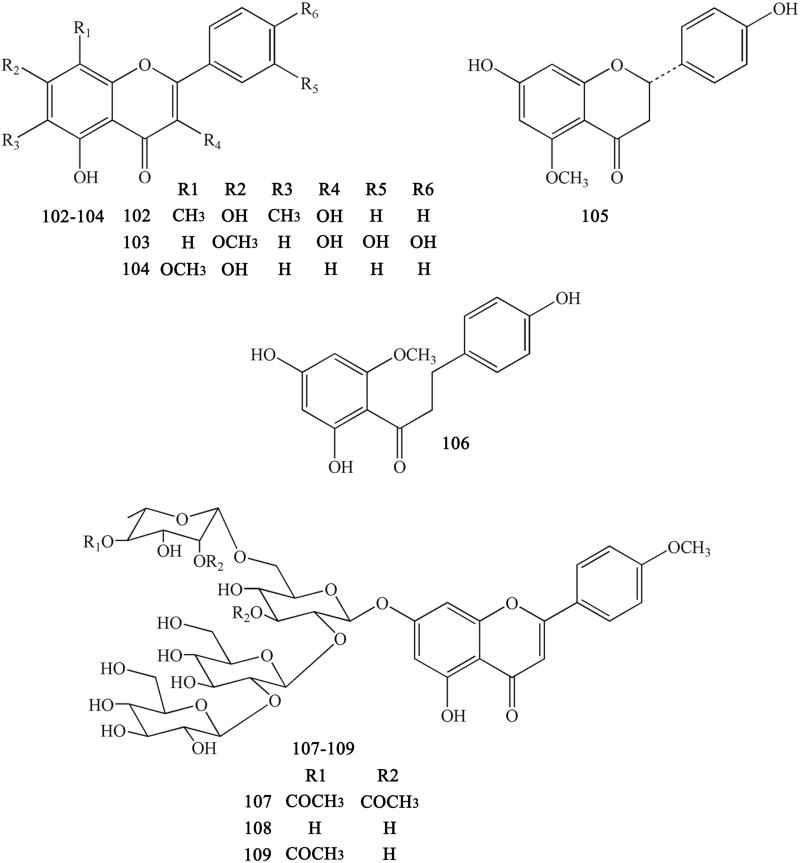
Flavonoids in Coptidis Rhizoma.
Figure 10.
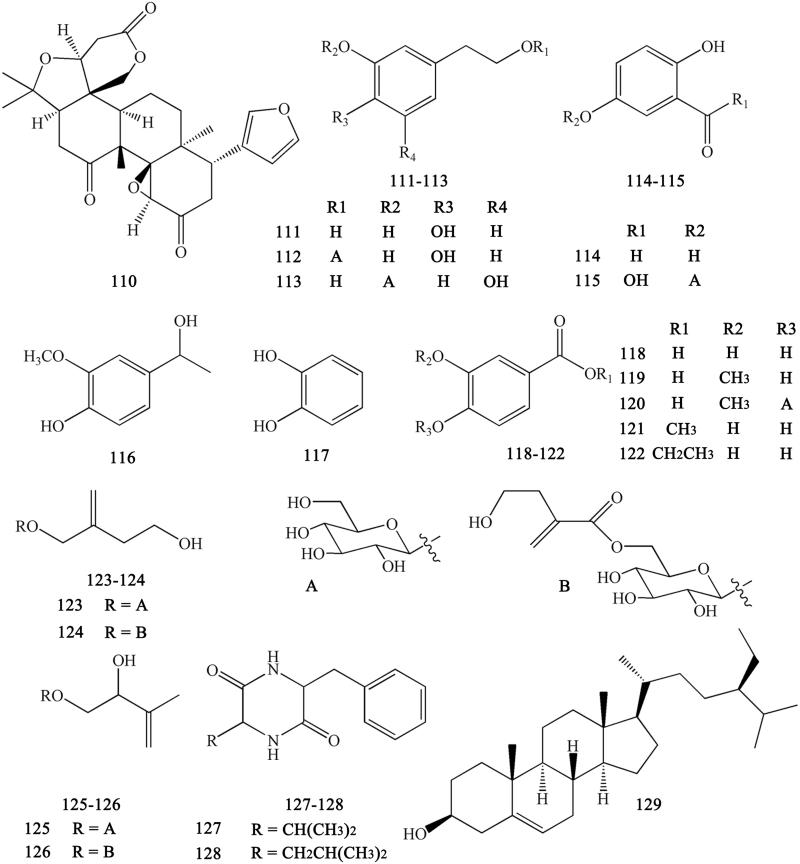
Other compounds in Coptidis Rhizoma.
Alkaloids
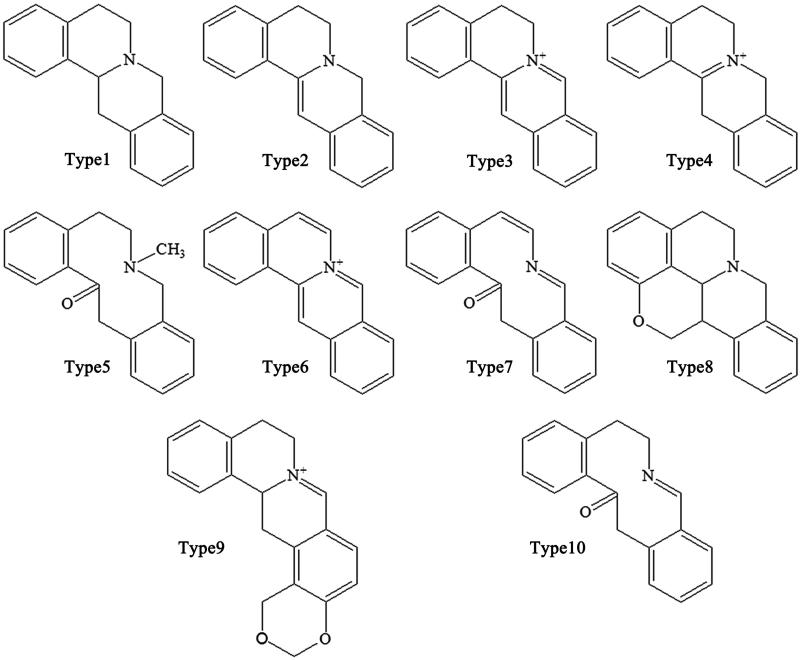
Alkaloids are the main active ingredients of coptidis, and isoquinoline alkaloids account for a large proportion, with berberine (1) as the most representative compound. Berberine is one of the most abundant ingredients (Cooper et al. 1970) at 4.5–8%, although this varies in different varieties of CR. In addition to berberine, CR contains over 30 different kinds of isoquinoline alkaloids, which can be divided into the following subtypes according to their structures: protoberberines, simple isoquinolines, aporphines and benzylisoquinolines (Figures 2–5).
Protoberberines
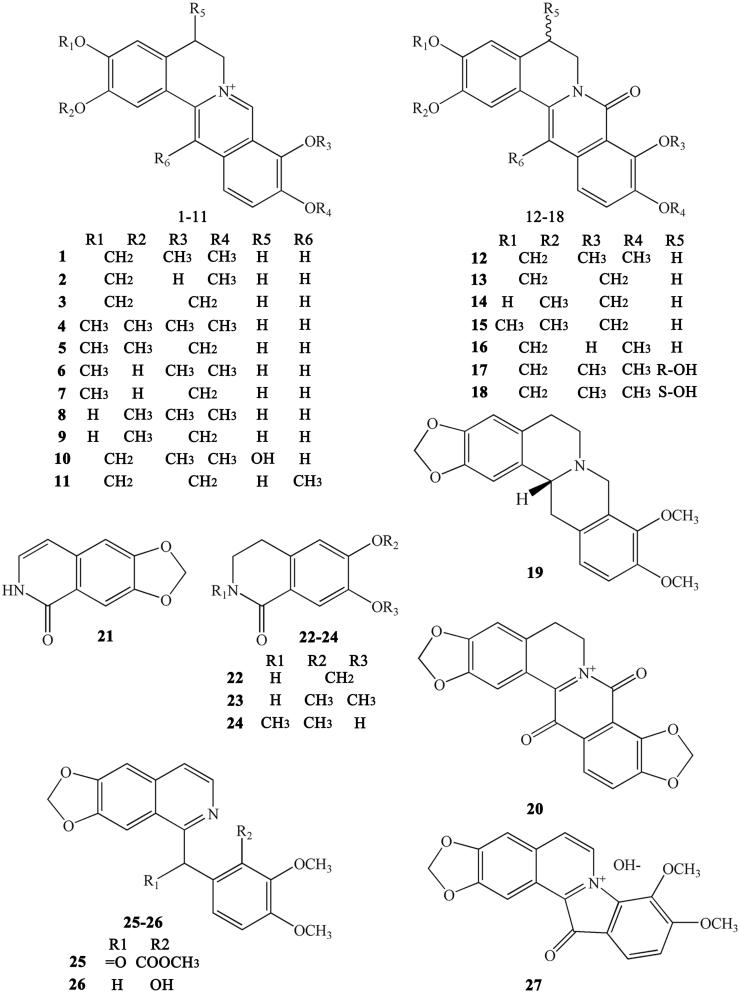
The protoberberine alkaloids are derived from benzylisoquinolines through phenolic oxidation and coupling with the isoquinoline N-methyl group, which becomes the ‘berberine bridge’ carbon. Tetracyclic rings, which are based on the dibenzo quinolizidine system, form the main matrices of protoberberine (Cooper et al. 1970). According to the position of the double bond and whether the nitrogen atom has a positive charge, the protoberberines can be divided into 10 subtypes, as shown in Figure 2. The following is a list of 20 representative protoberberine compounds that can be found in CR. Among these subtypes, type 3 is the most common one in CR: Berberine (1), berberrubine (2), coptisine (3), palmatine (4), epiberberine (5), columbamine (6), tetradehydroscoulerine (7), jatrorrhizine (8), groenlandicine (9), berberastine (10), worenine (11), 8-oxyberberine (12), 8-oxycoptisine (13), 3-hydroxy-2-methoxy-9,10-methylenedioxy-8-oxyprotoberberine (14), 8-oxyepiberberine (15), 8-oxyberberrubine (16), (-)-5-hydroxyl-8-oxyberberine (17), (+)-5-hydroxyl-8-oxyberberine (18), tetrahydroscoulerine (19), and 8,13-dioxocoptisine hydroxide (20) (Yoshikawa et al. 1995; Wang et al. 2007; Li ZF et al. 2012; Fan et al. 2014; Wang et al. 2014).
Simple isoquinolines
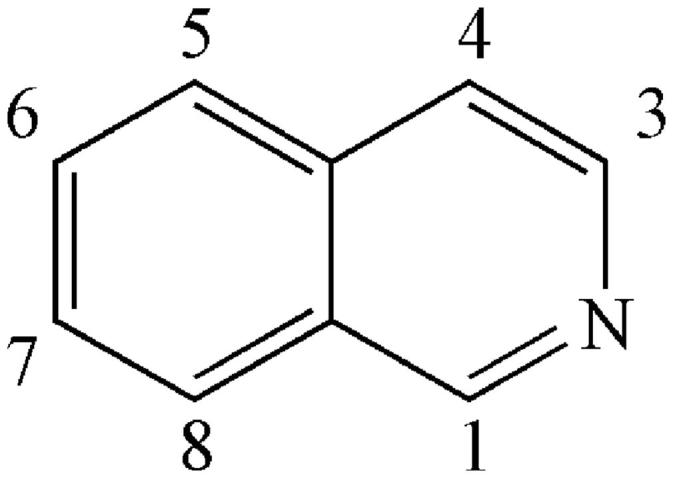
Alkaloids belonging to this subtype are fused together by a benzene ring and a pyridine; the nitrogen atom is in position 2 (which differs from quinoline) (Figure 3). Simple isoquinolines usually have a smaller in molecular weight and have no complex branched chains. The simple isoquinolines in CR include 1,3-dioxolo[4,5-g]isoquinolin-5(6H)-one (21), noroxyhydrastinine (22), corydaldine (23), and thalifoline (24) (Wang et al. 2007; Li ZF et al. 2012; Fan et al. 2014).
Benzylisoquinolines
Benzylisoquinolines are divided into 1-benzylisoquinolines and bis-benzylisoquinolines. 1-Benzylisoquinolines are compounds with isoquinoline matrices and a benzyl group at position 1. Furthermore, bis-benzylisoquinolines are formed by a combination of two 1-benzylisoquinolines via 1-3 ether bonds, such as 6-([1,3]dioxolo[4,5-g]isoquinoline-5-carbonyl)-2,3-dimethoxy benzoic acid methyl ester (25), berbithine (26), coptisonine (27), tetrandrine (28), and obamegine (29) (Wang et al. 2007).
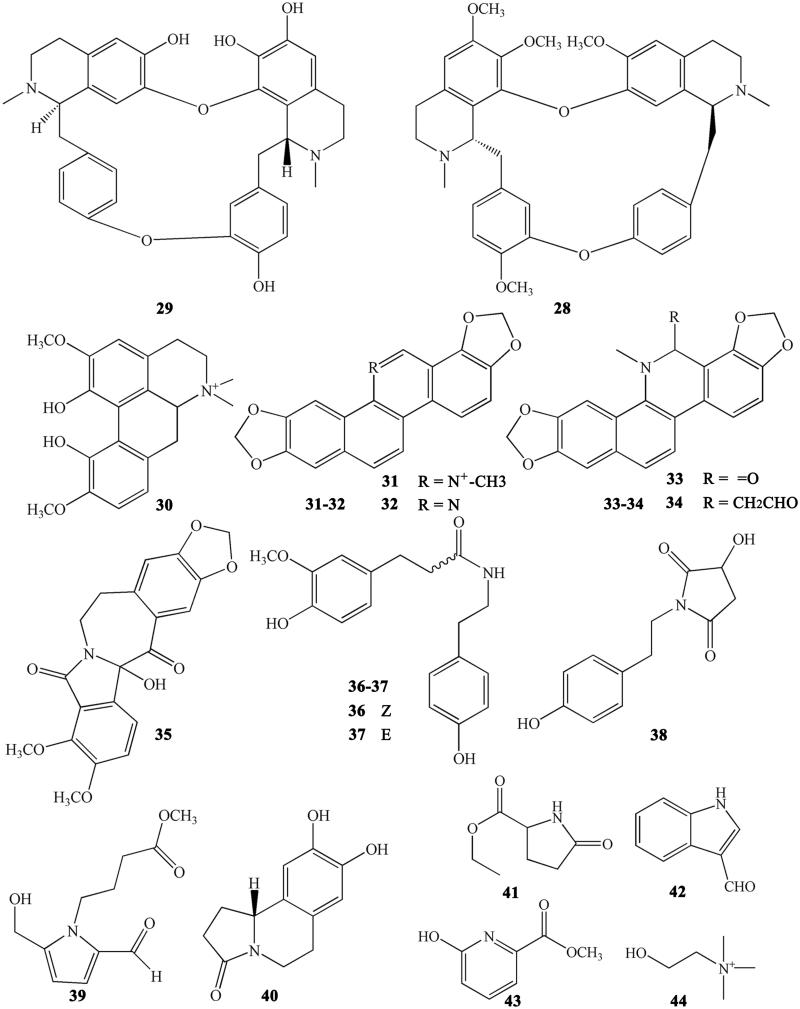
Other alkaloids
CR also contains other subtypes of alkaloids, such as magnoflorine (30) (Tomita and Kura 1956), which is an active ingredient belonging to the aporphine alkaloids. Moreover, some benzophenanthridine alkaloids can also be found in certain specific CR varieties. For example, sanguinarine (31), norsanguinarine (32), oxysanguinarine (33), and 6-acetonyl-5,6-dihydrosanguinarine (34) can be found in C. japonica (Maiti et al. 1982). CR also includes some small alkaloids, which are not representative compounds, such as chilenine (35) (Fan et al. 2014), Z-N-ferulyltyramine (36), E-N-feruloyltyramine (37), 3-hydroxy-1-(4-hydroxyphenethyl) pyrrolidine-2,5-dione (38), and 4′-[formyl-5-(hydroxymethyl)-1-pyrrol-1-yl] butanoate (39) (Wang et al. 2007); and 8,9-dihydroxy-1,5,6,10-β-tetrahydro-2H-pyrrolo[2,1-α]-isoquinolin-5-one (40), ethyl-2-pyrrolidinone-5(S)-carboxylate (41) (Li et al. 2012), methyl-5-hydroxy-2-pyridinecarboxylate (42), 1H-indole-3-carboxaldehyde (43), and choline (44) (Chen et al. 2012; Li XG et al. 2012; Li ZF et al. 2012; Ma H et al. 2013).
Phenylpropanoids
Phenylpropanoids are a class of compounds that are linked together by a benzene ring and three-carbon chains. They are a large class of organic compounds that exist widely exist in natural medicines and can be subdivided into many different subclasses. The molecular weight of phenylpropanoids in CR varies greatly, as do their structures. Both phenylpropanoids and their glycosides were reported in CR.
Lignans
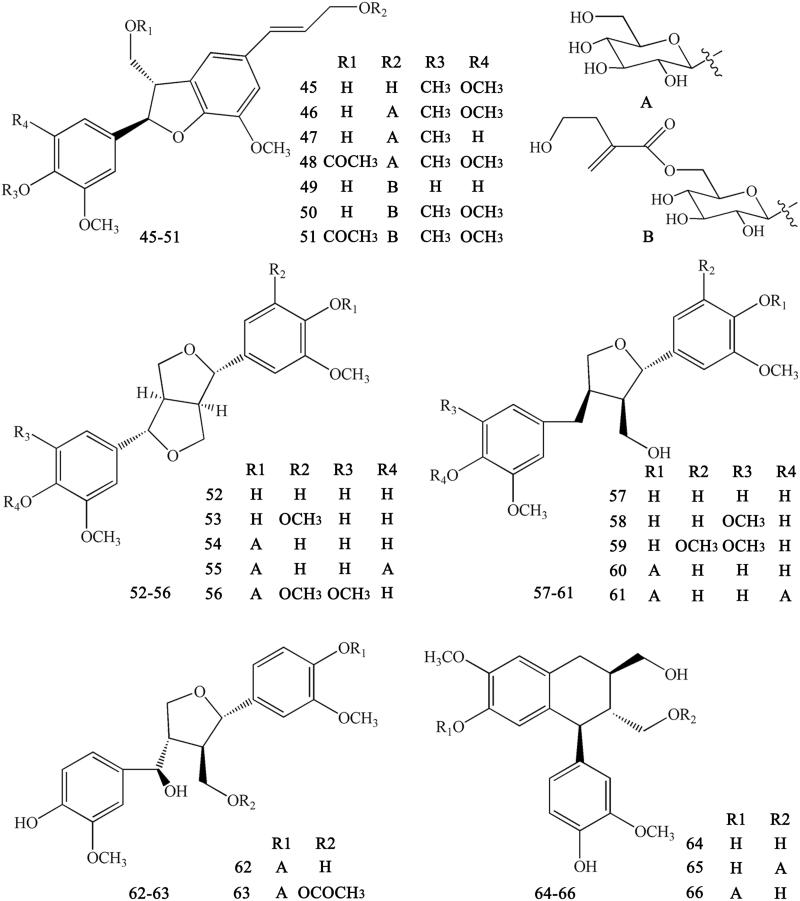
Lignans are important natural constituents with various pharmacological activities. Special kinds of phenylpropanoids, which are a combination of two or more simple phenylpropanoids, were comprehensively investigated and isolated from CR (Min et al. 1987;; Hirano et al. 1997; Yoshikawa 1997a; Chen L et al. 2012; Li XG et al. 2012; Wang et al. 2012). These constituents include woorenogenin (45), woorenoside I (46), longifolroside A (47), woorenoside II (48), woorenoside V (49), woorenoside III (50), woorenoside IV (51), (+)-pinoresinol (52), (+)-medioresinol (53), (+)-pinoresinol glucoside (54), (+)-pinoresinol-4,4′-O-β-d-diglucopyranoside (55), (+)-syringaresinol glucoside (56), (+)-lariciresinol (57), (±)-5,5′-dimethoxylariciresinol (58), (+)-5′-methoxylariciresinol (59), (+)-lariciresinol glucoside (60), 7S, 8 R, 8′R-(+)-lariciresinol-4,4′-O-β-d-diglucopyranoside (61), lanicepside A (62), 9-acetyl lanicepside B (63), (+)-isolariciresinol (64), isolarisiresinol-9-O-β-d-glucopyranoside (65), woorenoside XI (66), cleomiscosin A (67), aquillochin (68), 2,3-bis-[(4-hydroxy-3,5-dimethoxyphenyl)-methyl]-1,4-butanediol (69), secoisolariciresinol (70), erythro-gaiacylglycerol-8-O-4′-(coniferylalcohol) ether (71), threo-guaiacylglycerol-8-O-4′-(coniferyl alcohol) ether (72), woorenoside X (73), dihydrodehydrodiconiferyl alcohol (74), and wooreno (75) (Figures 6–7).
Simple phenylpropanoids
Ferulic acid and its derivatives are the most common simple phenylpropanoids in herbal medicine. In addition to ferulic acid, we can also found other simple phenylpropanoids. These derivatives usually form esters with carboxyl groups (Yahara et al. 1985; Yoshikawa et al. 1995, 1997a; Hirano et al. 1997; Chen L et al. 2012; Li et al. 2012; Meng et al. 2013; Fan et al. 2014). These compounds include Z-octadecyl cafeate (76), E-3-methoxycinnamic acid (77), ferulic acid (78), ethyl ferulate (79), N-butyl ferulate (80), p-hydroxyphenethyl E-ferulate (81), E-3,4-dimethoxycinnamic acid (82), 4-O-feruloylquinic acid (83), methyl 4-O-feruloylquicinate (84), ethyl-4-O-feruloylquicinate (85), 4-O-feruloylquinic acid butyl ester (86), 5-O-feruloylquinic acid (87), methyl-5-O-feruloylquicinate (88), ethyl-5-O-feruloylquicinate (89), 5-O-feruloylquinic acid butyl ester (90), chlorogenic acid (91), methyl-3-O-feruloylquicinate (92), N-butyl-3-O-feruloylquicinate (93), 3-(4′-hydroxyphenyl)-(2R)-lactic acid (94), 3-(3′,4′-hydroxyphenyl)-(2R)-lactic acid (95), 3-(3′,4′-dihydroxyphenyl)-(2R)-lactic acid-4′-O-β-d-glucopyranoside (96), methyl-3-(4′-O-β-d-glucopyranosyl-3′,4′-dihydroxyphenyl)-lactate (97), methyl-3,4-dihydroxyphenyl lactate (98), ethyl-3,4-dihydroxyphenyl lactate (99 ), N-butyl-3,4-dihydroxyphenyl lactate (100), and 3-(2,3,4-trihydroxyphenyl) propanoic acid (101) (Figure 8).
Flavonoids
Previous research reported that CR also contains certain flavonoids, mainly including 6,8-dimethyl-3,5,7-trihydroxyfavone (102), rhamnetin (103), wogonin (104) (Meng et al. 2013), 7,4′-dihydroxy-5-methoxyfavanone (105), 2′,4,4′-trihydroxy-6′-methoxydihydrochalcone (106) (Min et al. 1987) , coptiside I (107), coptiside II (108) and woorenoside XII (109) (Fujiwara et al. 1976; Yoshikawa et al. 1997b) (Figure 9).
Other compounds
Other compounds isolated from CR include limonin (110), 3,4-dihydroxyphenylethyl alcohol (111), 3′,4′-dihydroxyphenethyl alcohol 1-O-β-d-glucopyranoside (112), 3,5-dihydroxyphenethyl alcohol-3-O-β-d-glucopyranoside (113), protocatechuic aldehyde (114), gentisic acid-5-O-β-d-glucopyranoside (115), apocynol (116), 1,2-dihydroxy-benzene (117), protocatechuic acid (118), vanillic acid (119), vanillic acid-4-O-β-d-glucopyranoside (120), protocatechuic acid methyl ester (121), protocatechuic acid ethyl ester (122), woorenoside VI (123), woorenoside VII (124), woorenoside VIII (125), woorenoside IX (126), cyclo-(Phe-Val) (127), cyclo-(Phe-Leu) (128), and β-sitosterol (129) (Yahara et al. 1985; Yoshikawa et al. 1997; Wang et al. 2007; Li XG et al. 2012; Li ZF et al. 2012; Ma H et al. 2013; Meng et al. 2013; Yang et al. 2014) (Figure 10).
Pharmacology
Anti-pathogenic microorganism activity
Increasing research has been devoted to investigating the anti-pathogenic microorganism effects of CR, and its antibacterial, antiviral, and antifungal effects have been comprehensively studied and validated. Importantly, berberine has been recognized as the most important active monomer in this plant (Table 4).
Table 4.
Anti-pathogenic microorganism effect.
| Pathogenic microorganism | Extract/compounds (number) | In vivo/In vitro | Mechanism | Minimal active concentration/dose | Reference |
|---|---|---|---|---|---|
| Streptococcus agalactiae | 1 | in vitro | Damaging the structure of bacterial cell membrane and inhibiting synthesis of protein and DNA | MIC = 231.9 µM | (Peng et al. 2015) |
| Actinobacillus pleuropneumoniae | 1 | in vitro | Restraining DNA and protein syntheses, inhibiting the cleavage of bacteria, blocking the division and development of bacteria | MIC = 929.1 μM | (Kang et al. 2015) |
| Staphylococcus aureus | CRE | in vitro | Not mentioned | MIC = 77.8 μg/mL | (Feng et al. 2011) |
| Coagulase-negative Staphylococcus strains | 1 | in vitro | Not mentioned | MIC = 47.6 – 1522.2 μM | (Wojtyczka et al. 2014) |
| Shigella dysenteriae | 1 | in vitro | Not mentioned | MIC = 74.3 μM | (Kong et al. 2010) |
| Escherichia coli | 1 | in vitro | Heavily perturbing the formation of the Z-rings, inhibiting the cell division protein FtsZ | MIC = 1.5-4.5 mM | (Boberek et al. 2010) |
| Salmonella typhimuriummice | 1 | in vivo | As a LPS antagonist and blocking the LPS/TLR4 signaling | 0.20 g/kg (mice), 0.05 g/kg (rabbit) | (Chu et al. 2014) |
| Helicobacter pylori | 5 | in vitro | Binding to the active-site sulfydryl groups | IC50 = 3.0 μM for HPU and 2.3 μM for JBU | (Tan et al. 2017) |
| Helicobacter pylori | 1 | in vitro | Not mentioned | 74.3-743.2 μM | (Song et al. 2014) |
| Salmonella Typhimurium | CRE | in vitro | Regulation of the immune response | MBC = 12.5 mg/mLfor S. Typhimurium MBC = 25 mg/mL forST21 |
(Chang et al. 2014) |
| Aeromonas hydrophila | Total alklaoids | in vitro | Injuring membrane by increasing membrane lipid fluidity and changing conformation of membrane proteins, and reducing the secretion of virulence factors | MIC = 62.5 mg/L | (Xue D et al. 2015) |
| 1 | MIC = 371.6 μM | ||||
| Staphylococcus aureus | 1 | in vitro | Not mentioned | IC50 = 169.5 μM | (Fan et al. 2008) |
| 3 | IC50 = 209.2 μM | ||||
| 4 | IC50 = 337.7 μM | ||||
| 5 | IC50 = 636.2 μM | ||||
| 8 | IC50 = 803.8 μM | ||||
| Herpes simplex virus | 1 | in vitro | Downregulation of JNK and NF-kappa B Activation | EC50 = 6.77 µM for HSV-1 | (Song et al. 2014) |
| EC50 = 5.04 µM for HSV-2 | |||||
| Influenza virus | 1 | in vitro and in vivo | Inhibiting the virus infection, repressing inflammatory substances release | In vivo: 5 mg/kg; | (Wu et al. 2011) |
| In vitro: IC50 = 74 µM | |||||
| Respiratory syncytial virus | berberine chloride | in vitro | Inhibition of RSV-mediated early p38 MAPK activation | 25 μM or 100 μM | (Shin et al. 2015) |
| Chikungunya virus | 1 | in vitro | Predominantly targeting the ERK arm of MAPK signaling | EC50 = 4.5 μM in human embryonic kidney cells | (Varghese et al. 2016b) |
| EC50 = 12.2 μM in human osteosarcoma cells EC50 = 35.3 μM in CRL-2522 cells | |||||
| Enterovirus 71 | 1 | in vitro | Downregulating autophagy and MEK/ERK signaling pathway | IC50 = 7.43-10.25 μM | (Wang HQ et al. 2017) |
| H1N1 neuraminidase (NA-1) | CRE | in vitro | Inhibiting H1N1 neuraminidase (NA-1) | IC50 = 96.1 μg/mL | (Zhou et al. 2017) |
| 4 | IC50 = 50.5 μM | ||||
| 8 | IC50 = 67 μM | ||||
| 5 | IC50 = 99.9 μM | ||||
| 1 | IC50 = 233.7μM | ||||
| coptisine | IC50 = 326.5 μM | ||||
| Coronavirus | CRE | in vitro | Inhibition of RNA-dependent RNA polymerase or proteases, affecting virus assembly or release | EC50 = 2.0 mg/mL | (Kim et al. 2008) |
| Human cytomegalovirus (HCMV) | berberine chloride | in vitro | Interfering with intracellular events after virus penetration into the host cells and before viral DNA synthesis | IC50 = 0.68 μM | (Hayashi et al. 2007) |
| Candida albicans | 1 | in vitro | Impairment mitochondrial function, generation of ROS, targeting cell wall integrity pathway and also affecting HSF1 | Not mentioned | (Dhamgaye et al. 2014) |
Antibacterial effect
Berberine can inhibit Gram-positive (G+) bacteria such as Streptococcus agalactiae, Staphylococcus aureus, S. mutans, Bacillus anthracis, S. suis, and Enterococcus faecium (Choi et al. 2007; Fan et al. 2008; Wang et al. 2014; Peng et al. 2015); and Gram-negative (G-) bacteria such as Actinobacillus pleuropneumoniae (Kang et al. 2015), Shigella dysenteriae (Kong et al. 2010), and Escherichia coli (Boberek et al. 2010). Interestingly, alkaloids isolated from CR, especially epiberberine, can act as urease inhibitors to treat Helicobacter pylori infection (Tan et al. 2017). In 2014, Chen et al. reported that CR extracts (CRE) significantly inhibited Salmonella typhimurium with a minimum bactericidal concentration (MBC) of 12.5 mg/mL. Another study reported that although CRE had no effect on bacteria such as Pseudomonas aeruginosa, Proteus mirabilis, and Proteus vulgaris, after processing with ginger, it showed a marked inhibitory effect against these bacteria, especially P. aeruginosa (Li 2015).
Previous studies revealed that the antibacterial effects of CR and its active constituents were attributed to damaging the cell membrane, inhibiting protein and DNA synthesis, blocking bacterial division and development, and disturbing the formation of the Z-rings to inhibit the cell division protein FtsZ (Chu et al. 2014; Xue D et al. 2015; Ming et al. 2016). The antibacterial effect of CR alkaloids against G+ bacteria was stronger than that against G- bacteria, which could be explained by different the cell membrane structures of the pathogens (Yong et al. 2007). Kong W et al. (2009) performed a comprehensive analysis including the growth rate constant k, maximum power output of the log phase P m,log, total heat output of the log phase Q t,log, generation time t g, growth inhibitory ratio I, and half-inhibitory concentration of the drugs (IC50), and revealed that the anti-bacterial activities against E. coli of the four alkaloids from CR were in the order of berberine > coptisine > palmatine≫ jatrorrhizine.
Antiviral effect
Previous investigations revealed that CR and berberine have inhibitory effects against respiratory syncytial virus, influenza virus, enterovirus 71, herpes simplex virus, coronavirus and cytomegalovirus. In addition, studies showed that the inhibitory effects of berberine were mediated by downregulating cellular c-Jun N-terminal protein kinase (JNK) and NF-kappa B activation (Hayashi et al. 2007), suppressing mitogen-activated protein kinase (MAPK) or MAPK/ERK kinase 1 (MEK)/extracellular signal-regulated kinase (ERK) signalling (Shin et al. 2015; Varghese et al. 2016). Furthermore, berberine could suppress the EV71-induced autophagy by activating the AKT protein and inhibiting the phosphorylation of JNK and phosphatidylinositol-4,5-bisphosphate 3-kinase III (PI3KIII) (Wang HQ et al. 2017). H1N1 infection could be also suppressed by a water extract of CR, during which the main alkaloids served as neuraminidase inhibitors, and among them, palmatine was the most effective, with an IC50 of 50.5 µM (Zhou et al. 2017). The specific inhibition of West Nile virus (WNV) NS2B-NS3 protease and viral propagation by palmatine, with an IC50 of 96 mM, was investigated. Palmatine was also effective against dengue virus and yellow fever virus (Jia et al. 2010).
Antifungal effect
Berberine showed a weak inhibitory effect on C. albicans when used alone; while combined with fluconazole, the MIC value decreased sharply to 14.27 μM (Iwazaki et al. 2010). Other research showed that the antifungal effect of berberine was based on its ability to impair mitochondrial function, the generation of reactive oxygen species (ROS), targeting the cell wall integrity pathway, and affecting heat shock transcription factor 1 (HSF1) (Dhamgaye et al. 2014).
Protective effects on the cardiovascular system
Cardiovascular diseases (CVDs) involving the heart or blood vessels are the leading cause of death in worldwide. It is estimated that by 2030, over 23 million people will die from CVDs each year (Mendis et al. 2011). Importantly, CR can exert significant beneficial effects on major risk factors of CVDs, including anti-atherosclerotic, antihyperlipidemic, antidiabetic, antihepatic steatototic effects. Recent studies have shown that alkaloids in CR can protect against CVDs, such as coronary heart diseases, myocardial ischemia-reperfusion injury, heart failure, arrhythmia, and hypertension (Feng 2008; Mei 2011; Yong et al. 2011) (Table 5).
Table 5.
Protecting cardiovascular system related diseases effect.
| Pharmacological effects | Extract/compounds | Material or model | Mechanism | Dose | Reference |
|---|---|---|---|---|---|
| Lipid lowering effect | 8 | High fat (HF) diet C57BL/6J mice | Suppressing of lipogenesis and the enhancement of lipid oxidation in the liver | 100 mg/kg, 56 d | (Yang et al. 2016) |
| Hypolipidemic Effect | 3, 1, 8, 4, 5, Total alkaloids of CR (TACR) | High fat and high choesterol (HFHC) diet hamsters | Down-regulating the expression of HMGCR and up-regulating the expression of LDLR and CYP7A1 as well as promoting the excretion of TBA in the feces | 46.7 mg/kg, for 140 d | (He et al. 2016) |
| Synergetic cholesterol-lowering effects of main alkaloids | 3, 1, 8, 4, 5, TACR | HC diet hamsters; HepG2 cell | Up-regulating LDLR and CYP7A1, down-regulating HMGCR | in vivo: 70.05 mg/kg, 28 d; in vitro: 5 μg/mL | (Kou et al. 2016) |
| Antihyperlipidemia | 4, 8, 1, 5, | Diabetic KK-Ay mice; HepG2 cell | not mentioned | in vivo: 225 mg/kg, 40 d; in vitro: 5 μg/mL | (Ma et al. 2016) |
| Antihypercholesterole | berbamine | HC diet adult zebrafish; zebrafish larvae; embryos | Up-regulating cholesterol transport and bile acid synthesis, inhibiting cholesterol synthesis and lipoprotein assembly or secretion | Adult: 2.25, 4.5 or 9 mg/fish, 28 d; Larvae: 10, 20 or 40 μg/mL, 10 d; embryo: 5, 10, 20, 40 or 80 μg/mL In vitro: 5, 10, 20, 40 or 80 μg/mL |
(Han et al. 2017) |
| Antihyperlipidemia | 3, 1, 4, TACR | HF diet C57BL/6J mice | Modulating of the enterohepatic circulation of bile acids and cross-talk between the gut microbiota and the liver | 140 mg/kg, 35 d | (He et al. 2017) |
| Treating obesity | Ethanol extracts of CR, 1 | HF diet C57BL/6J mice | Decreasing degradation of dietary polysaccharides, lowering potential calorie intake, activating mitochondrial energy metabolism, regulating on gut microbes | 200 mg/kg, 42 d | (Xie W et al. 2011) |
| Treating obesity | 1 | High fat and high carbohydrate diet Wistar rats | Increasing the production of adiponectin and regulating the AMPK mechanism | 380 mg/kg, 56 d | (Wu et al. 2016) |
| Anti-adipogenic activity | 1, 3, 4, 5, 30 | 3T3-L1 cells | Downregulating C/EBP-α and PPAR-gamma | 12.5-50 μM | (Choi et al. 2014) |
| Anti-adipogenic effect | 5 | 3T3-L1 cells | Downregulating Raf/MEK1/ERK1/2 and AMPKα/Akt pathways during 3T3-L1 adipocyte differentiation | 12.5, 25, 50 μM | (Choi et al. 2015) |
| Supressing adipocyte differentiation | 1 | 3T3-L1 cells | Inhibiting cAMP/PKA-mediated CREB pathway | 5 μM | (Zhang et al. 2015) |
| Treating atherosclerosis and other chronic inflammatory disease | 3 | ApoE(-/-) mice | Inhibiting activation of MAPK signaling pathways and NF-kappa B nuclear translocation | 150 mg/kg, 84 d | (Feng et al. 2017) |
| Anti-atherosclerosis | 1 | Apolipoprotein E-deficient mice | Inhibiting oxidation and inflammation cytokine expressions | 150 mg/kg, 84 d | (Feng et al.2016) |
| Suppressing atherogenesis | 1 | Western diet ApoE (ApoE-/-) mice and ApoE-/-/AMPK alpha 2-/- mice; HUVECs | Suppressing atherogenesis via stimulation of AMPK-dependent UCP2 expression | in vivo: 1 mM in drinking water, 56 d; in vitro: 10 μM | (Wang et al. 2011) |
| Anti-atherogenic effect | 1 | THP-1-derived macrophages | Activating AMPK-SIRT1-PPAR-γ pathway and diminishing the uptake of ox-LDL | 14.9, 29.7, 59.5 mg/L | (Chi et al. 2014) |
| Anti-atherogenesis | 1 | THP-1 cells | Suppressing the activation of p38 pathway | 5, 10, 25, 50 μM | (Huang et al. 2011) |
| Against I/R injury | 1 | T2DM Wistar rats exposed to I/R | AMPK activation, AKT phosphorylation, and GSK3 inhibition in the nonischemic areas of the diabetic heart | 100 mg/kg, 7 d | (Chang et al. 2016) |
| Alleviating cardiac I/R injury | 1 | I/R C57BL/6 mice, H9c2 myocytes, | Suppressing autophagy activation by decreasing the expression of SIRT1, BNIP3, and Beclin- p-AMPK and p-mTORC2 (Ser2481) | in vivo: 5 mg/kg, 10 mg/kg; in vitro: 5, 10, 20 μM | (Huang et al. 2015) |
| Anti- I/R injury | 1 | I/R SD rats | Attenuating mitochondrial dysfunction and myocardial apoptosis | 200 mg/kg, 28 d | (Wang Y et al. 2015) |
| Anti-I/R injury | 1 | I/R SD rats, SIR H9c2 cells | Modulating Notch1/Hes1-PTEN/Akt signaling | in vivo: 200 mg/kg, 14 d; in vitro: 50 μM | (Yu et al. 2015) |
| Anti- I/R injury | 1 | I/R SD rats, SI/R H9c2 cells | Activating the JAK2/STAT3 signaling pathway and attenuating ER stress-induced apoptosis | in vivo: 200 mg/kg, 14 d; in vitro: 50 μM | (Zhao et al. 2016) |
| Anti- I/R injury | 1 | I/R SD rats | Suppressing the activation of PI3K/AKT signaling, | 100 mg/kg, 14 d | (Zhu and Li2016) |
| Anti-cardiac I/R injury | 1 | H/R H9c2 cells | Inhibiting apoptosis through the activation of Smad7 | 50 μM | (Yao et al. 2017) |
| Inhibition of autophagy induced by hypoxia | 1 | H9c2 cells under hypoxia | Inhibition of autophagy and suppression of AMPK activation | 5, 10 or 25 µM | (Jia et al. 2017) |
| Attenuating MI/R injury | 1 | I/R SD rats, SI/R H9c2 | Reducing oxidative damage and inflammation response, and SIRT1 signaling plays a key role | in vivo: 200 mg/kg, 14 d; in vitro: 50 μM | (Yu et al. 2016) |
| Anti- hypertrophy | 1 | High Glucose-and Insulin-Induced Cardiomyocyte | Activating the PPARα/NO signaling pathway | 0.01-10 μM. | (Wang M et al. 2013) |
| Anti- acute myocardial ischemia | 1 | SD rats with isoproterenol | Anti-inflammatory and antioxidative activity through regulating HMGB1-TLR4 Axis | 30, 60 mg/kg, 14 d | (Zhang T et al. 2014) |
| Anti-H/R damage | 3 | H/R H9c2 cell | Inhibition of autophagy | 0.3, 1, 3, 10 μM | (Wang Y et al. 2017) |
| Anti-I/R injury | 3 | I/R SD rats | Suppressing myocardial apoptosis and inflammation by inhibiting the Rho/ROCK pathway | 3, 10, and 30 mg/kg | (Guo et al. 2013) |
| Reducing I/R injury | 4 | I/R SD rats, HAEC cells, RAW 264.7 cells | Reducing oxidative stress and modulating inflammatory mediators | in vivo: 25, 50 mg/kg; in vitro: 1, 2, 5, 10 μM in HAEC; 1, 5, 10 μM in RAW 264.7 cells | (Kim et al. 2009) |
| Anti-nonalcoholic steatohepatitis | 1 | HF diet Balc/c mice | Normalizing gut microbiota, decreasing expression of endotoxin receptor, inflammatory cytokines | 200 mg/kg, 56 d | (Cao et al. 2016) |
| Decreasing hepatic steatosis | 1 | HF C57BL/6J mice, H4IIE cells | Anti-inflammation | in vivo: 100 mg/kg, 28 d; in vitro: 10, 25, 50 μM | (Guo et al. 2016) |
| Attenuating hepatic steatosis | 1 | High fat and high-sucrose C57BL/6 mice, mouse primary hepatocytes, HepG2 cells | Inducing autophagy and fibroblast growth factor 21 in SIRT1-dependent manner | in vivo: 5 mg/kg, ip., 35 d; in vitro: 10 μM | (Sun et al. 2017) |
| Attenuating hepatic steatosis | 1 | HF diet SD rats, Huh7 cells | Global modulation of hepatic mRNA and lncRNA expression profiles | in vivo: 200 mg/kg, 112 d; in vitro: 10 μM | (Yuan et al. 2015) |
| Attenuating hepatic steatosis | 1 | Db/db mice and methionine-choline-deficient diet mice, tunicamycin-induced mice, HepG2 cells | Reducing endoplasmic reticulum stress through the ATF6/SREBP-1c pathway | in vivo: 200 mg/kg, 35 or 20 or 3 d respectively; in vitro: 5 μM | (Zhang et al. 2016) |
Anti-atherosclerotic effect
Atherosclerosis (AS) commonly occurs in the subendothelial space (intima) of arteries and is triggered by endothelial dysfunction and subendothelial lipoprotein retention (Tabas et al. 2015). It has been reported that CR and its main alkaloids, such as berberine and coptisine, could effectively prevent the development of AS, and the potential mechanisms are correlated with suppressing ROS mediated oxidation (Xu RX et al. 2017), and halting chronic inflammatory reactions via inhibition of intracellular inflammation signaling pathways (Feng et al. 2016, 2017 ). In particular, berberine could inhibit atherogenesis by reducing oxidative stress and the expression of adhesion molecules in the aorta, and increasing the levels of uncoupling protein 2 (UCP2) (Wang et al. 2011). Another CR component, magnoflorine, could inhibit the copper-mediated (Cu2+) oxidation of various low-density lipoprotein (LDL) forms by increasing the lag time of conjugated diene formation and suppressing the generation of thiobarbituric acid reactive substances (TBARS) (Hung et al. 2007). The accumulation of foam cells in the subendothelial space is an indispensable step for the initiation and progression of AS. Berberine treatment could suppress foam cell formation, as well as the accumulation of lipid and cholesterol. The mechanism involves the activation of adenosine 5-monophosphate (AMP)-activated protein kinase (AMPK)-SIRT1-peroxisome proliferators-activated receptor γ2 (PPAR-γ) pathway and a decrease in ox-LDL uptake (Chi et al. 2014). Berberine can stabilize atherosclerotic plaques by inhibiting the expressions of matrix metalloproteinase 9 (MMP-9) and extracellular matrix metalloproteinase inducer (EMMPRIN) by suppressing activation of the p38 pathway (Huang et al. 2011).
Anti-hyperlipidemic effect
Hyperlipidemia, characterized by increased levels of blood lipids, has been implicated as a contributing factor to the development of cardiovascular diseases. The main mechanism of resisting hyperlipidemia is related to inhibiting lipogenesis and promoting the use, conversion and excretion of lipid (Iii et al. 2014). Alkaloids derived from CR, including berberine, coptisine, palmatine, epiberberine and jatrorrhizine, appeared to prevent body weight gain, reduce serum levels of total cholesterol (TC), triglyceride (TG) and low-density lipoprotein–cholesterol (LDL-c) and increase high-density lipoprotein–cholesterol (HDL-c) and promoted the excretion of total bile acids (TBA) in faeces (He et al. 2016; Yang W et al. 2016). The effect of berberine is mainly related to upregulating the LDL receptor (LDLR) and Cytochrome P450 7A1 (CYP7A1), while downregulating 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (HMGCR) (Ma et al. 2016). In addition, palmatine and epibeberine, which could also be beneficial to treat hyperlipidaemia and downregulate apical sodium dependent bile acid transporter (ASBT) (Zou et al. 2016; He et al. 2017). The sterol regulatory element-binding proteins (SREBPs) are transcription factors that regulate cholesterol by binding to the promoters of genes such as those encoding LDLR and HMG-CoA synthase. Interestingly, administration of coptisine, berberine and palmatine could activate SREBP2 (Kai et al. 2016). Besides these main alkaloids of CR, some minor alkaloids, such as berbamine, could also exert effects on hypercholesterolemic zebrafish by upregulating cholesterol transport and bile acid synthesis (Han et al. 2017).
Anti-obesity
Obesity is a pathological condition characterized by excessive body fat that often leads to cardiovascular diseases (Ashraf and Baweja 2013). 3T3-L1 cells are commonly used to detect fat metabolism. Previous studies revealed that five CR alkaloids (berberine, coptisine, palmatine, epiberberine and magnoflorine) could inhibit adipocyte differentiation and cellular triglyceride accumulation in 3T3-L1 cells, and downregulated adipocyte marker genes [including PPAR-γ and CCAAT/enhancer binding protein (C/EBP)] (Choi et al. 2014, 2015; Zhang et al. 2015). Lipolysis is the process of breaking down lipids and has been regarded as a target for treating obesity. Adiponectin, which is involved in the regulation of metabolic processes, binds to two main receptors (AdipoR1 and AdipoR2), whose expression levels are decreased during the development of obesity. Berberine treatment upregulated the expression of AdipoR1 and AdipoR2, which consequently elevated adiponectin production and induced lipolysis. Berberine could also directly upregulate lipolysis-related genes such as those encoding LPL, PPARα, carnitine palmitoyltransferase 1 (CPT1), and medium-chain acyl-CoA dehydrogenase (MCAD) (Wu et al. 2016).
Nonalcoholic fatty liver disease is a type of hepatic steatosis, which is always involved in obesity. It was reported that mice gut microbiota could be restored by gavage of 200 mg/kg of berberine for 8 weeks, resulting in alleviation of the predisposing factors for liver steatosis. These effects could be mediated by decreasing endotoxin receptor CD14 and inflammatory cytokines such as interleukin (IL)-1, IL-6, and tumour necrosis factor alpha (TNF-α) (Cao et al. 2016). This finding is consistent with another study that suggested that berberine’s actions are largely based on suppressing inflammation, independent of AMPK (Guo et al. 2016). Berberine could also attenuate hepatic steatosis and enhance energy expenditure in mice by inducing autophagy and fibroblast growth factor 21 (FGF21) expression; however, these effects were abolished by a deficiency of the nutrient sensor SIRT1 (Sun et al. 2017). Furthermore, increasing evidence suggests that the mechanism may correlate with global modulation of hepatic mRNA and long noncoding RNA (lncRNA) expression profiles, reducing endoplasmic reticulum stress (ER) stress through the ATF6/SREBP-1c pathway (Yuan et al. 2015; Zhang et al. 2016).
Protective effect against ischaemic heart disease
Cardiac ischemia is characterized by the deficient supply of blood flow and energy generating nutrients to the myocardium (Steenbergen and Frangogiannis 2012). The most effective treatment for ischaemic heart disease (IHD) is to re-perfuse the heart. However, re-perfusion could lead to series of additional injuries, termed ischaemia reperfusion injury (IRI) (Wijck and Buurman 2002). CR and its active compounds could reduce apoptosis, excessive autophagy, and inflammatory response, regulate energy metabolism, improve mitochondrial function, as well as alleviate ER stress, all of which might combine to alleviate IRI.
Berberine treatment could improve myocardial infarction and injury to cardiomyocytes, as indicated by the decrease of creatine kinase isoenzyme (CK-MB), lactate dehydrogenase (LDH), and cardiac troponin (cTnI); reducing oxidative stress by suppressing malondialdehyde (MDA) production; and promoting superoxide dismutase (SOD) (Liu XT et al. 2010; Zhang T et al. 2014; Wang Y et al. 2015). In vivo and in vitro experiments showed that berberine could reduce the myocardial infarct size, improve cardiac function; and suppress myocardial apoptosis, oxidative damage, and ER stress through activating the JAK2/STAT3 signalling pathway (Zhao et al. 2016). Activation of the AMPK signalling pathway and silent information regulator 1 (SIRT1) signalling might be involved in the anti-autophagy and anti-apoptosis effect of berberine (Yu et al. 2016; Jia et al. 2017).
In pressure-overload-induced cardiac hypertrophy, berberine inhibited the mTOR, p38, and ERK1/2 MAPK signaling pathways to enhance autophagy, consequently attenuating left ventricular remodeling and cardiomyocyte apoptosis (Li MH et al. 2014). However, excessive autophagy activity can also cause cell death, termed ‘autophagic cell death’, also known as type-II programed cell death (Li S et al. 2017). It has been reported that berberine could reduce excessive autophagy by suppressing autophagy-related proteins, such as LC3-II, SIRT1, BNIP3 and Beclin-1, thus protecting H9c2 cells from hypoxia/reoxygenization (HR)-induced cell death (Huang et al. 2015). In non-ischemic areas of diabetic animal hearts, berberine increased myocardial glucose uptake, glycolysis, and fatty acid oxidation (Chang et al. 2016). The observation that berberine could act as an M2 muscarinic agonist, which reduced the spontaneous contraction rate of cardiomyocytes in culture might contribute to our understanding of berberine’s complex actions on the heart (Salehi and Filtz 2011).
Studies have shown the berberine could reduce the release of TNF-α, IL-6, IL-β and HMGB1 to attenuate ischemic heart injury. TLR4, which is activated by HMGB1, is also reduced by berberine (Zhang T et al. 2014). Preconditioning with berberine for 14 days before the induction of I/R significantly attenuated myocardial I/R injury, as manifested by a reduction in the incidence of ventricular arrhythmia and the amelioration of myocardial histological changes. These effects were associated with the suppression of the PI3K/AKT signalling pathway and subsequent reduction of the expression of related inflammatory cytokinesis in the serum and myocardial tissue (Zhu and Li 2016).
Berberine could inhibit high glucose and insulin-induced cardiomyocyte hypertrophy, accompanied by increasing nitric oxide synthase (NOS) activity and NO concentration, which elevated PPARα and eNOS (Wang M et al. 2013). Coptisine also has an effect against myocardial ischemia reperfusion (MI/R) injury by suppressing myocardial apoptosis and inflammation via inhibition of the Rho/ROCK pathway, and inhibiting autophagosome formation rather than induction of autolysosomes in autophagy events (Guo et al. 2013; Wang Y et al. 2017).
Maintenance of mitochondrial integrity is one of the critical aspects of protecting the myocardium (Calo et al. 2013). Berberine could improve mitochondrial dysfunction, as indicated by increasing mitochondrial membrane potential, mitochondrial complex activity and decreasing the release of cytochrome C from mitochondria (Wang Y et al. 2015).
Antidiabetes
Diabetes mellitus (DM) is a common chronic diseases characterized by disorders of glucose metabolism that seriously threaten human health and longevity (Shi and Hu 2014). As early as the Wei and Jin Dynasties, Ming Yi Bie Lu recorded the treatment of CR for Xiaoke, which has been proven to be DM. CR and its components exert anti-diabetic effects by improving glucose metabolism, insulin resistance (IR), pancreatic beta cells and modulating the gut microbiota (Table 6).
Table 6.
Antidiabetes effect.
| Pharmacological effects | Extract/compounds | Material or mode | Mechanism | Dose | Reference |
|---|---|---|---|---|---|
| Antihyperglycemia | 1, 3, 4, 5, 8 | Diabetic KK-Ay Mice; HepG2 cells | Not mentioned | in vivo: 225 mg/kg, 40 d; in vitro: 5 μg/mL | (Ma et al. 2016) |
| Lowering glucose concentration | 1 | HepG2 cells and betaTC3 cells | insulin independent but has no effect on insulin secretion | 5 to 200 μM | (Xie X et al. 2011) |
| Activating glucose uptake | 1 | 3T3-L1 adipocytes | Activating GLUT1 through AMPK stimulation | 1, 5 μM | (Kim et al. 2007) |
| Treating type 2 DM | 1 | HF diet C57BL/6J mice, NIT-1 cells | Inhibiting mouse insulin gene promoter through activation of AMPK and exerting beneficial effect on pancreatic β-cell |
in vivo: 50 mg/kg, 70 d; in vitro: 0.01-10 μM |
(Shen et al. 2012) |
| Against insulin resistance | 1 | HF-diet and STZ induced Wistar rats, KK-Ay mice, HepG2, Bel-7402, L6 cells |
Through PKC–dependent up-regulation of insulin receptor expression | in vivo: 150, 300 mg/kg, 15 d, 200 mg/kg, 21 d; in vitro: 22.3 μM | (Kong WJ et al. 2009) |
| Antihyperglycemic | 1 | HF diet C57BL/6J mice, db/db mice, 3T3-L1 and L6 cells | Inhibiting PTP1B activity and mimicing insulin action |
in vivo: 100 mg/kg, 14 d; in vitro: 1.25-100 μM |
(Chen et al. 2010) |
| Increasing glucose uptake | 1 | Insulin-sensitive and insulin-resistant rat skeletal muscle cells | Improving tyrosine-phosphorylation of IRS-1 and the recruitment of p85 to IRS-1, PKC and PKB activity, inhibiting mTOR |
14.8 μM | (Liu LZ et al. 2010) |
| Treating type 2 DM | 1 | Alloxan-induced Wistar rats | Hypoglycemic effect, modulating lipids metabolic effects and to scavenge free radical |
100, 200 mg/kg, 21 d | (Tang et al. 2006) |
| Insulinotropic effect | 1 | Primary rat islets | Activating HNF4α and GK | 1, 3, 10 and 30 μM | (Wang et al. 2008) |
| Treating T1DM | 1 | Nonobese diabetic (NOD) mice | Protcting pancreatic islets and serum lipids | 50, 150, 500 mg/kg, 98 d | (Chueh & Lin 2011) |
| Protecting pancreatic islets | 1 | STZ-treated primary pancreatic islet cells |
Down-regulating Bax/Bcl-2 gene expression ratio | 1, 3, 5 μM | (Chueh & Lin 2012) |
| Treating T2DM | 1 | HF diet and STZ induced rats | Lowering RBP4 levels and up-regulating the expression of GLUT4 protein in tissues | 380 mg/kg, 28 d | (Zhang et al. 2008) |
| Antidiabetic effects | 1 | SD rats, NCI-H716 cells | Promoting GLP-1 secretion and GLP-1 biosynthesis in PKC-dependent pathway | in vivo: 60, 120 mg/kg, 35 d; in vitro: 1, 10, 100 μM | (Yu Y et al. 2010) |
| Ameliorating insulin resistance | 1 | HepG2 cells | Improving insulin sensitivity via its anti-inflammatory activity | 0.1, 1, 10 μM | (Lou et al. 2011) |
| Treating T2DM | 1 | RAW264.7 microphages | Attenuating inflammation by SIRT1 | 5 μM | (Chuanchong 2016) |
| Treating T2DM | 1 | STZ induced ddY mice | Antioxidative stress via down regulating GPx and up-regulating CuZn-SOD | 200 mg/kg, 14 d | (Lao-Ong et al. 2012) |
| Treating T2DM | 1 | high-carbohydrate/high-fat diet Wistar rats | Antioxidation and up-regulating P-TEFb expression | 75, 150, 300 mg/kg, 42 d | (Zhou and Zhou 2011) |
| Treating diabetic neuropathy | 1 | SH-SY5Y cells | As an Nrf2 activator | 0.1-10 nM | (Hsu et al. 2013) |
| Hypoglycemic | 1 | STZ induced diabetic SD rats, Caco-2 cells | Suppressing disaccharidase activities and the mRNA expression of SI complex in PKA-dependent pathway | in vivo: 100, 200 mg/kg, 35 d; in vitro: 2, 10, 50 μM | (Liu L et al. 2010) |
| Moderating glucose metabolism | 1 | HF diet SD rats | Regulating the MAPK and GnRh-Glp-1 pathways in the ileum | 120, 240 mg/kg, 56 d | (Zhang Q et al. 2014) |
Improving glucose metabolism
The expression of the glucose transporter protein (GLUT) is a key factor in the intracellular transport of glucose and is closely linked to cellular energy metabolism (Huang 2013). A previous report revealed that after treatment with berberine, the glucose uptake in L929 fibroblast cells, a cell line that express only GLUT1, reached maximum stimulation. Moreover, significant activation was observed within 5 min and reached a maximum at 30 min, which was attributed to the acute activation of the transport activity of GLUT1 (Cok et al. 2011). The level of GLUT1 protein was increased in 3T3-L1 cells, which was stated to be associated with the activation of AMPK stimulation (Kim et al. 2007). The upregulation of GLUT4 expression and downregulation of Retinol-binding protein 4 (RBP4) are also involved in glucose uptake (Zhang et al. 2008). HepG2 and βTC3 cell lines were used to test glucose consumption and insulin release, respectively. The results showed that glucose consumption by HepG2 cells was increased from 32% to 60% by berberine, which was insulin independent but had no influence on insulin secretion (Xie et al. 2011). Another study showed the GnRH-glucagon-like peptide-1 (GLP-1) and MAPK pathways in the intestines might be involved in the mechanisms of berberine to modulate glucose metabolism (Zhang Q et al. 2014).
Improving insulin resistance
Insulin resistance (IR) is a pathological condition in which cells fail to respond to the normal actions of the hormone insulin. IR increases the risk of developing pre-diabetes and type-2 DM. Treatment with berberine at 50 mg/kg/day for 2 weeks was effective against the features of IR syndrome, and could improve levels of IR parameters, such as body weight, hyperglycemia, hyperinsulinemia, hypercholesterolemia, and hypertriglyceridemia (Ye et al. 2016). Shen et al. (2012) revealed that berberine could decrease insulin levels in pancreatic islet β-cells via reversible the concentration-dependent inhibition of the INS2 promoter. Increasing the expression of insulin receptor (INSR) is also regarded as a target of berberine to increase insulin sensitivity. This effect is related to a protein kinase C (PKC)-dependent activation of its promoter (Kong WJ et al. 2009). In some insulin-resistant patients with diabetes, there is a phenomenon of increased INSR dephosphorylation by protein tyrosine phosphatase 1B (PTP1B). Interestingly, berberine can suppress the activation of PTP1B to increase the phosphorylation of INSR (Chen et al. 2010). Insulin receptor substrate (IRS) is a key molecule that acts after the insulin receptor and mediates insulin signalling. In insulin signalling, the levels of phosphorylated AKT and IRS were significantly increased by berberine in alloxan-induced diabetic mice (Xie X et al. 2011). In insulin-resistant cells, berberine improved insulin-induced tyrosine-phosphorylation of IRS-1 and the recruitment of p85 to IRS-1, which was related to the inhibition of mTOR (Liu LZ et al. 2010).
Improving pancreatic β cells and promoting the secretion of insulin
Some studies reported that berberine could promote the secretion of insulin by increasing GLP-1 release or by stimulating pancreatic cells (Wang et al. 2008; Yu Y et al. 2010). Intragastric administration of berberine restored the damage to pancreas tissues and reversed the decreased in the number of islets in rats with DM (Tang et al. 2006; Chueh and Lin 2011). Berberine significantly downregulated the ratio of BAX/BCL-2 to block streptozotocin (STZ)-induced apoptosis in mouse pancreatic islets (Chueh and Lin 2012). Berberine and CRE exerted similar protective effect on islet β cells by improving islet β cell proliferation and the protein level of PARP1 (Jiang et al. 2017). Inflammation and oxidation are closely associated with DM. After treatment with berberine, decrease levels of proinflammatory cytokines, such as TNF-α, IL-6, iNOS, MCP-1 and COX-2, were observed (Jeong et al. 2009; Lou et al. 2011), while IL-10 levels were elevated in diabetic animals, in related cells, and in patients (Sun 2017). The levels of AR, SOD, GSH-px and GSH increased, while MDA decreased, indicating that oxidation was inhibited (Zhou and Zhou 2011; Lao-Ong et al. 2012). Multiple cellular kinases, as well as signalling pathways (such as MAPKs, AMPK, Nrf2/HO, NF-κB, and Rho GTPase pathways) were verified to be pivotal for berberine’s activity in reducing oxidative stress and inflammation to treat DM (Wang et al. 2009; Xie et al. 2013; Mo et al. 2014). However, some studies showed that berberine could decrease hyperglycaemia and improve impaired glucose tolerance but did not increase insulin release and synthesis (Yin et al. 2002; Chen et al. 2010). In addition to berberine, recent studies showed that polysaccharides in CR increased glucose uptake, recovered glucose tolerance, inhibited the formation of advanced glycation end products, and reduced oxidation (Jiang et al. 2015; Cui et al. 2016; Yang Y et al. 2016).
Modulating gut microbiota
In recent years, berberine has been demonstrated to treat DM by modulating the structure and diversity of gut microbiota, including enrichment of beneficial microbes and inhibition of harmful microbes (Liu L et al. 2010). The bioavailability of berberine is very low, and the absorption rate is only 5–10% in the intestinal tract. However, it can significantly reduce the activity of disaccharidase and α-glucosidase in the intestinal tract, resulting in a reduction the absorption of glucose and postprandial hyperglycemia (Liu L et al. 2010; Li ZQ et al. 2012). CR alkaloid treatment avoided a decline in the diversity of gut microbes in obese mice and favoured the maintenance of a stable and healthy bacterial community in high-fat high cholesterol (HFHC)-fed animals (Kai 2017). Berberine can lead to an increase in the abundance of probiotics such as Blautia, Bacteroides, Bifidobacteria and Lactobacillus, and a decrease in relative abundance of Firmicutes and Bacteroides in the intestinal tract of animals (Meng et al. 2016; Gu et al. 2017).
Another study showed that the berberine selectively enriched the propionic acid producing bacteria and intestinal barrier repair bacteria Ackermansia; a CR decoction promoted butyric acid producing bacteria, such as Coprococcus, Faecalibacterium and Oscillospira. Compared with berberine, the CR decoction induced higher flora diversity, and the flora structure was closer to that of normal animals (Ti 2017). The increase of GLP-1 and short-chain fatty acids in the gut may account for the structural and diversity changes to the microbiota induced by berberine (Sun et al. 2016).
Anticancer effect
Cancer is the second leading cause of death globally and was responsible for 8.8 million deaths in 2015. Globally, nearly 1 in 6 deaths are caused by cancer, as reported by the World Health Organization. Studies showed that CR and berberine are effective against multiple types of human cancer, including bladder, breast, cervix, cholangiocarcinoma, colon, Ehrlich, gastric, glioma, intestine, kidney, leukemia, liver, lung, nasopharyngeal, melanoma, myeloma, ovary, pancreas, prostate and sarcoma (Ho et al. 2009; Wang N et al. 2015). CR and its active ingredients can prevent cancer by blocking the cell cycle, inhibiting tumor cell proliferation, inducing apoptosis, inhibiting migration and invasion, and enhancing the body’s immune function (Table 7).
Table 7.
Anticancer effect.
| Pharmacological effects | Extract or compounds | Material or model | Mechanism | Dose | Reference |
|---|---|---|---|---|---|
| Treating melanoma | 1 | A375 cells | Up-regulating p38 MAPK, GR and down-regulating DHODH | 5, 10, 20, 40, 80 μM | (Liu B et al. 2017) |
| Treating melanoma | 1 | B16 cells | Modulating the PI3K/Akt pathway, RARα/RARβ expression | 10, 20, 40 μM | (Kou et al. 2016) |
| Treating hepatocellular carcinoma | 1 | HepG2 cells | Promoting apoptosis through the NF-κB p65 pathway | 10, 50, 100 μM | (Li M et al. 2017) |
| Treating hepatocellular carcinoma | CRE | HepG2 cells, MHCC97-L and HepG2 cells xenograft mice | Suppressing vascular endothelial growth factor via inactivation of eukaryotic elongation factor 2 | in vitro: IC50 of 500 and 150 µg/mL at 24 and 48 h in MHCC97L cells, 250 and 120 µg/mL in HepG2 cells; in vivo: 50 mg/kg/2 d, 21 d | (Tan et al. 2014) |
| Treating hepatocellular carcinoma | CRE | MHCC97-L cells | Downregulating the Rho/ROCK signaling pathway | 300, 150 μM at 24 h and 48 h | (Wang N et al. 2010) |
| Treating hepatocellular carcinoma | 1 | HepG2 and MHCC97-L cells | Increasing Bax expression, activating Beclin‐1, inhibiting mTOR‐signaling pathway by suppressing the activity of Akt and up‐regulating P38 MAPK signaling | IC50: 100 µM in HepG2 cells, 250 µM in MHCC97‐L cells | |
| Treating hepatocellular carcinoma | berberine hydrochloride | HepG2 cells | AMPK activation | 50, 100 μM | (Yu et al. 2014) |
| Treating hepatocellular carcinoma | 3 | SMMC7721 xenograft mice, SMMC7721 and HepG2 cells | Induction of apoptosis through a 67LR/cGMP pathway | in vivo: 50 mg/kg; in vitro: 12.5, 25, 50,100 μM; | (Zhang et al. 2018) |
| Treating glioma | Coptis Chinensis granules | Xenograft mice, U87 cells | Down-regulating phosphorylation of STAT3 by reducing HDAC3 | in vivo: 20, 10 mg/per mouse, 1 month at intervals of 1 d; in vitro: 0.625, 1.25, 2.5, 5 or 10 mg/mL; | (La et al. 2017) |
| Inducing autophagic cell death | 1 | HCT-116, HepG2, DLD1cells | enhancing GRP78 levels and The ability of GRP78 to bind to VPS34 | IC50: 80, 100, 200 μM in HCT-116, HepG2, DLD1 respectively | |
| Treating osteosarcoma | 1 | Xenograft mice, Saos-2 and MG-63 cells | Downregulating caspase-1/IL-1β inflammatory signaling | in vivo: 20 mg/kg, 21 d; in vitro: 50 μM | (Jin et al. 2016) |
| Treating osteosarcoma | 1 | MG-63 cells | Inducing apoptosis and DNA damage | 20, 40, 60, 80 μM | (Zhu et al. 2014) |
| Treating osteosarcoma | 3 | xenografted mice; MG63, SW1353, Saos-2, and U-2OS cells | Downregulating the expression of CDK4, cyclin D1, VE-cadherin and integrin ß3, diminishing STAT3 phosphorylation | in vivo: 50 mg/kg, 24 d; in vitro: IC50: 12.99 ± 0.77, 14.10 ± 2.17, 22.56 ± 2.94, and 28.54 ± 5.71 µM, respectively | (Yu D et al. 2014) |
| Treating esophageal cancer | 1 | KYSE-30 cells | Anti-migration and anti-metastasis mediated by chemokine receptors | IC50: 60, 45 and 40 µM after 24, 48 and 72 h, respectively | (Mishan et al. 2015) |
| Treating lung cancer | 1 | H460, H1975 cells | Suppressing both phosphorylated and total levels of STAT3 protein and promoting STAT3 degradation by enhancing ubiquitination | IC50: 13.4 and 62.43 μM for H460 and H1975 cells | (Zhu et al. 2015) |
| Treating human gastric cancer | 1 | xenograft nude mice, BGC-823 and SGC7901 cells; |
Ihibiting the Akt/mTOR/p70S6/S6 pathway | in vivo: 10 mg/kg injected intratumorally once every 3 d for 18 d in vitro: 10, 25, 50, 75, 100 μM | (Yi et al. 2015) |
| Treating breast cancer | 3 | MDA-MB-231 cells | Anti-metastatic function through down-regulation of MMP-9 in combination with the increase of TIMP-1 | 16, 32, 64 μM | (Li J et al. 2014) |
| Treating nasopharyngeal carcinoma | berberine hydrochloride | CNE-1 cells | Blocking proliferation, migration and invasion, inducing apoptosis | IC50: 31.5 µM | (Li CH et al. 2014) |
| Treating tongue squamous cancer | 1 | SCC-4 cells | Inhibiting FAK, IKK, NF-kappaB, u-PA and MMP-2 and -9 | 62.5, 125 μM | (Ho et al. 2009) |
| Treating human colon cancer | 1 | SW620 cells | Generating reactive oxygen species and activation of JNK/p38 MAPK and FasL | 50 μM | (Hsu et al. 2007) |
| Treating colorectal cancer | 3 | Xenograft mice; HCT116 cells | Inhibiting epithelial–mesenchymal transition, the growth, adhesion and metastasis, down-regulating MFG-E8 | in vivo: 30, 60, 90 mg/mL, 14 d; in vitro: 62.4, 124.8, 249.6 μM; | (Cao et al. 2018) |
| Treating pancreatic cancer | 4 | PSC cells, HPNE, MIA PaCa-2, CFPaC-1 and PANC-1 cells | Inhibiting glutamine-mediated PSC-PCC interaction through simultaneous inhibition of survivin and COL1A1 | 212.8, 425.6 μM | (Chakravarthy et al. 2018) |
| Treating esophageal cancer | Coptidis Rhizoma, 1 | Xenograft mice, YES-2, YES-2 cells | Down-regulating tumor IL-6 production | in vivo: oral food supplement of final concentration of 1%; in vitro: 8-32 mM | (Iizuka et al. 2000) |
Inducing apoptosis
Berberine induces apoptosis in human colonic carcinoma cell line SW620; in the pancreatic cancer cell lines PANC-1 and MIA-PaCa2; and in breast cancer MCF-7 cells through the generation of ROS. Moreover, berberine had a greater apoptotic effect in PANC-1 cells than gemcitabine (Hsu et al. 2007; Xie et al. 2012; Park et al. 2015). When compared with chemical drugs (meloxicam and rosiglitazone) and berberine, total alkaloids showed a greater apoptosis-inducing effect (Ke 2007). Various apoptotic modulating signals are involved the induction of apoptosis by berberine. Berberine could markedly inhibit the expression of survivin in MGC-803gastric cancer cells, in SKOV3ovarian cancer cells (Zhang et al. 2013; Ma et al. 2015); and activated caspase-3, caspase-8, caspase-7 and caspase-9 in FaDu head and neck squamous cell carcinoma cells and malignant pleural mesothelioma (Yao 2014; Seo et al. 2015). Berberine also regulated the activities of Bcl-2 and Bax in colon cancer cells (Chidambara et al. 2012), FoxO1 and FoxO3 in HepG2 cells (Shukla et al. 2014), and p53 in MCF-7 and MDA-MB231breast cancer cells (Kim et al. 2012). Additionally, cPLA-COX2 and JAK2/STAT3 signalling was inhibited in liver cancer cells and colon cancer cells HT-29 (Li O et al. 2013; Li C et al. 2014). Berberine also promoted the Fas/FasL signalling pathway, and then triggered the activation of caspase-8 and caspase-9 precursors to induce apoptosis in human oral cancer cells (Kim et al. 2015). In HCT-116 colon cancer cells, berberine enhanced GRP78 activity by binding to and forming complexes with GRP78, which increased the ability of GRP78 to bind to VPS34. This suggested berberine could induce autophagic cancer cell death (La et al. 2017). In vitro and in vivo experiments showed that coptisine inhibited the proliferation, growth and migration of HCC cells and colorectal cancer cells, and promoted their apoptosis. Other studies showed that coptisine activated microRNA miR-122 (Chai et al. 2018) and the 67-kDa Laminin Receptor (Zhou et al. 2018), and inhibited MFG-E8 (Cao et al. 2018).
Cell cycle arrest
Berberine inhibited the expression of Cyclin D1 and the activity of the related AP-1 and Wnt pathways. Berberine prevented the proliferation of lung cancer PG cells by inhibiting Cyclin D1, increasing the number of cells in the Go/G1 phase, and decreasing the number of cells in the S phase and G2/M phase (Ye 2007). Berberine blocked human gastric carcinoma cell entrance into the cell cycle in the G0/G1 phase, and inhibited colorectal adenocarcinoma growth by inducing G2/M phase arrest (Sha et al. 2011; Cai et al. 2014). However, CR and berberine decreased the number of CNZ-2Z cells in the Go/G1 phase significantly, while the number of cells in the S phase increased significantly, indicating that the cell cycle was blocked in the S phase (Cui et al. 2008). In osteosarcoma, berberine treatment led to G1/S cell cycle arrest in p53-presenting cells, but may cause G2/M arrest in p53-deficient cells, suggesting that p53 may play diverse roles in the cell cycle distribution in berberine-treated cancer cells (Liu et al. 2009). In addition, another CR component, jatrorrhizine, could inhibit the proliferation and neovascularization of C8161 human metastatic melanoma cells by inducing cell cycle arrest at the G0/G1 transition (Liu et al. 2013). Moreover, columbamine could suppress proliferation and neovascularization of metastatic osteosarcoma U2OS cells with low cytotoxicity and induced cell cycle arrest at the G2/M transition, which was associated with attenuation of CDK6 gene expression, STAT3 phosphorylation and MMP2 expression (Bao et al. 2012).
Inhibiting tumour metastasis
Urokinase-type plasminogen activator (uPA) and MMPs play important roles in cancer metastasis and angiogenesis, and inhibition of uPA and MMP could inhibit the migration and invasion of cancer cells. Berberine affected JNK, ERK1/2, p38 MAPK, P13K-Akt and NF-κB signalling pathways to inhibit the actions of MMP-2, MMP-9, MMP-1, and uPA in SCC-4 human tongue squamous carcinoma cells, hepatoma cells, and breast cancer cells (Ho et al. 2009; Bing et al. 2011; Kim et al. 2012; Kuo et al. 2012). NM23-H1 and SDF-1 are potential genes associated with tumour cell metastasis and previous research indicated that berberine could decrease NM23-H1 and SDF-1 expression; thus reducing the metastasis of leukaemia cells (Li, Guo, et al. 2008; Liu et al. 2008). It was reported that berberine (50 μM) could act as a RhoGTPases inhibitor in HONE1 human nasopharyngeal carcinoma cell (Tang et al. 2009). Inhibition of RhoGTPase by CRE, as well as by berberine (100-200 μM), might also result in blockade of ROCK signalling in hepatoma cells (Wang et al. 2010). The expression levels of two chemokine receptors (CXCR4 and CCR7), which are involved in the migration and metastasis of esophageal cancer cells, were decreased following the berberine treatment (Mishan et al. 2015).
Tumour angiogenesis, a process associated with invasion and metastasis, is an essential link in the control of tumour progression (Zhao and Adjei 2015). In tumour angiogenesis, VEGF and hypoxia-inducible factor-1α (H1F-1α) play a key role in tumour progression. In vivo and in vitro studies revealed that the antiangiogenic activity of berberine was mediated by downregulating the expression of H1F-1, VEGF and proinflammatory mediators in hepatocellular carcinoma cells and breast cancer cells (Jie et al. 2011; Hamsa & Kuttan 2012; Kim et al. 2013). Berberine may also inhibit the adhesion of gastric cancer cells to endothelial cells by increasing the proportion of intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1), thus reducing the risk of tumour angiogenesis induced by evodiamine (Shi et al. 2013). In addition, berberine showed anti-angiogenesis effects on animals that were orthotopically implanted with hepatocellular carcinoma (Tsang et al. 2015). Coptisine at 150 mg/kg may reduce cancer metastasis risk by inhibiting the RAS-ERK pathway in HCT116 bearing mice (Huang et al. 2017).
Chinese medicinal herbs can enhance the body’s immune function, by inducing cytokines, interferon (IFN), lymphocyte-activated killer cells production and natural killer (NK) cell proliferation, thereby mediating tumor cell apoptosis. Importantly, CRE could markedly increase the IFN-β and TNF-α mRNA expression in breast cancer MCF-7 oestrogen receptor-positive cells (Kang et al. 2005) Furthermore, berberine was also capable of reducing the expression of caspase-1 and IL-1β in osteosarcoma cells, and inhibiting the growth of tumour cells, suggesting that the mechanism might involve downregulation of the caspase-1/IL-1β inflammatory signalling axis (Jin et al. 2016). A recent study showed that palmatine disrupted the interaction between pancreatic stellate cells and cancer cells in the tumour microenvironment, consequently resulting in the inhibition of cancer growth and migration, while inducing apoptosis by inhibiting survivin (Chakravarthy et al. 2018).
Other pharmacological effects
Experimental studies showed that CR and its compounds could be used to treat diseases of nervous system, digestive system, skeleton, and skin and hepatotoxicity, nephrotoxicity and aging-related disorders (Lee et al. 2010; Su et al. 2017). Berberine could ameliorate β-amyloid pathology, gliosis, and cognitive impairment in an Alzheimer’s disease transgenic mouse model through the PI3K/AKT/GSK3 signalling pathway and induced 6-hydroxydopamine-induced human dopaminergic neuronal cell death through the induction of heme-oxygenase-1 and exert antidepressant action through inhibition of organic cation transporter 2 and 3 (Durairajan et al. 2012; Bae et al. 2013; Sun et al. 2014). Furthermore, CR could treat Alzheimer’s disease via the significant inhibition of acetylcholinesterase (AchE) (Kaufmann et al. 2016). CRE, coptisine and jatrorrhizine displayed neuroprotective effect by alleviating oxidative stress (Friedemann et al. 2015, 2016; Luo et al. 2016). Berberine prevented glucocorticoid-induced bone loss in lumbar spongy bone by promoting bone formation and inhibiting bone resorption (Bilian et al. 2011). CRE had a radioprotective effect against radiation-induced skin damage in rats by modulating oxidative stress in skin and in aging-related diseases via antioxidation and AMPK activation (Wang XJ et al. 2013; Xu Z et al. 2017). In the digestive system, CR extracts could exert an analgesic effect on a rat model of irritable bowel syndrome by decreasing serotonin release and cholecystokinin expression (Tjong et al. 2011). Coptisine showed a significant gastric mucosal protective effect on stress gastric ulcers in mice. However, the protective effect of coptisine (57 mg/kg) on the gastric mucosa was significantly better than that of 100 mg/kg berberine (Feng et al. 2007). Jatrorrhizine delayed gastric emptying and intestinal transit in postoperative ileus (Zhang et al. 2012). Berberine has the potential to alleviate premenopausal syndrome by decreasing oxidative stress, LDL, triglycerides, insulin resistance and improving mood (Caliceti et al. 2015).
Pharmacokinetics
Currently, pharmacokinetics research on CR has mainly focused on the protoberberine alkaloids. After oral intake, blood exposure and absolute bioavailability are extremely low. During absorption, 50% of berberine undergoes extensive first-pass elimination (Liu Y et al. 2010). Then, the absorbed alkaloids are quickly and widely distributed in tissues, such as the brain, intestine, stomach, pancreas, heart, kidney, liver, spleen, lung, testicles and uterus, among which the liver has the highest concentration (Ma et al. 2010). Furthermore, the concentrations of the alkaloids in tissues are not only higher than those in circulation, but also are eliminated at a slower rate (Liu Y et al. 2010). Researchers have analysed metabolites from urine, feces, plasma, and intestinal flora and found that they mainly comprise the sulphate and glucuronide conjugates of the CR alkaloids or the Phase I metabolites of the alkaloids (Yang et al. 2010). In liver microsomes, cytochrome P450 isoenzymes (CYPs) play a major role. The intestinal flora also exerts significant effect on the enterohepatic circulation of the metabolites, which may be related to the multiple peaks phenomenon of the pharmacokinetics of the CR alkaloids (Zuo et al. 2006). Berberine is usually excreted in urine and bile. Other studies showed, only 0.013% of berberine is eliminated directly in urine after oral administration (Yu et al. 2000). The metabolites are mainly eliminated via urine (Yang et al. 2010), and aproportion of them are also eliminated through bile (Zuo et al. 2006). However, in some pathological conditions, such as diabetes mellitus, PI-IBS (post-inflammation irritable bowel syndrome) and lipopolysaccharide-related diseases, the pharmacokinetic processes are altered. In 2008, Yu et al. showed a higher exposure of berberine, palmatine, coptisine, epiberberine and jatrorrhizine, with 170–330% increases in C max (maximum concentration) and 150–350% increases in AUC0-24 (area under curve) in diabetic rats, after oral administration of CRE (1.3 g/kg). Then, in 2010, they discovered that impairment of the function and expression of P-glycoprotein in the intestine partly contributed to the increased exposure of the five protoberberine alkaloids (Yu et al. 2010).
Table 8.
The pharmacokinetic parameters of component in animals and humans.
| Subjects/animals | Drug administered | Dosages | Detected compounds | Pharmacokinetic parameters | References |
|---|---|---|---|---|---|
| Male SD rats | TACR (i.g.) | 1.3 g/kg | 1 | AUC0–24 (ng·h·mL−1): 147.66 ± 14.19, | (Yu et al. 2007) |
| Cmax (ng·mL-1): 11.39 ± 1.62, | |||||
| Tmax (h): 3.40 ± 1.47, | |||||
| MRT (h): 9.31 ± 0.81 | |||||
| 3 | AUC0–24 (ng·h/mL): 11.74 ± 7.24, | ||||
| Cmax (ng/mL): 1.52 ± 0.74, | |||||
| Tmax (h): 2.25 ± 1.82, | |||||
| MRT (h): 6.69 ± 2.07 | |||||
| 5 | AUC0–24 (ng·h/mL): 10.19 ± 6.67, | ||||
| Cmax (ng/mL): 2.40 ± 0.88, | |||||
| Tmax (h): 1.30 ± 1.56, | |||||
| MRT (h): 5.09 ± 2.44 | |||||
| 8 | Cmax (ng/mL): 0.84 ± 0.47, | ||||
| Tmax (h): 1.69 ± 1.72 | |||||
| 4 | AUC0–24 (ng·h/mL): 14.80 ± 2.25, | ||||
| Cmax (ng/mL): 1.74 ± 0.56, | |||||
| Tmax (h): 0.90 ± 0.95, | |||||
| MRT (h): 10.12 ± 1.16 | |||||
| Male diabetic SD rats | CRE (i.g.) | 1.3 g/kg | 1 | AUC0–24 (ng·h/mL): 255.10 ± 8.04 | (Yu et al. 2008) |
| Cmax (ng/mL): 24.97 ± 8.39 | |||||
| Tmax (h): 3.10 ± 1.52 | |||||
| MRT (h): 9.44 ± 1.52 | |||||
| 3 | AUC0–24 (ng·h/mL): 35.53 ± 10.32 | ||||
| Cmax (ng/mL): 4.88 ± 1.40 | |||||
| Tmax (h): 2.80 ± 1.44 | |||||
| MRT (h): 8.17 ± 1.30 | |||||
| 5 | AUC0–24 (ng·h/mL): 35.97 ± 11.14 | ||||
| Cmax (ng/mL): 6.26 ± 2.37 | |||||
| Tmax (h): 2.80 ± 1.44 | |||||
| MRT (h): 7.28 ± 1.71 | |||||
| 8 | AUC0–24 (ng·h/mL): 8.17 ± 3.30 | ||||
| Cmax (ng/mL): 1.30 ± 0.23 | |||||
| Tmax (h): 3.00 ± 1.06 | |||||
| MRT (h): 6.84 ± 1.45 | |||||
| 4 | AUC0–24 (ng·h/mL): 22.02 ± 4.39 | ||||
| Cmax (ng/mL): 2.93 ± 1.14 | |||||
| Tmax (h): 4.00 ± 3.79 | |||||
| MRT (h): 9.30 ± 0.61 | |||||
| Male pseudo germ-free Wistar rats | 1 (i.g.) | 40 mg/kg | 1 | AUC0-limt (ng·h/mL): 40.89 | (Zuo et al. 2006) |
| MTT (h): 10.25 | |||||
| Berberrubie, | AUC0-limt (ng·h/mL): 437.29, | ||||
| MTT (h): 7.45 | |||||
| Thalifendine | AUC0-limt (ng·h/mL): 287.85 | ||||
| MTT (h): 4.84 | |||||
| Emethylen-eberberine | AUC0-limt (ng·h/mL): 735.22 | ||||
| MTT (h): 15.08 | |||||
| 8 | AUC0-limt (ng·h/mL): 101.98 | ||||
| MTT (h): 4.05 | |||||
| Male Wistar rats | 1 (i.g.) | 40 mg/kg | 1 | AUC0-limt (ng·h/mL): 37.42, | |
| MTT (h): 10.53 | |||||
| Berberrubie | AUC0limt (ng·h/mL): 1879.64, | ||||
| MTT (h): 18.32 | |||||
| Thalifendine | AUC0-limt (ng·h/mL): 811.05, | ||||
| MTT (h): 7.62 | |||||
| Emethylen-eberberine | AUC0-limt (ng·h/mL): 1763.62, | ||||
| MTT (h): 24.68 | |||||
| 8 | AUC0-limt (ng·h/mL): 356.05, | ||||
| MTT (h): 10.32 | |||||
| Post inflammation irritable bowel syndrome male Wistar rats | berberine hydrochloride (i.g.) | 25 mg/kg | 1 | AUC0-t (ng·min/mL): 2,763.43 ± 203.14 | (Gong et al. 2014) |
| Cmax (ng/mL): 18.53 ± 0.61 | |||||
| Tmax (min): 15.00 ± 0.00 | |||||
| T1/2,λz (min): 941.45 ± 60.39 | |||||
| Vd/Fλz (L/kg): 41,202.89 ± 4,112.68 | |||||
| CL/F (L/h/kg): 3,270.57 ± 58.32 | |||||
| Male SD rats | berberine hydrochloride (i.g.) | 25 mg/kg | 1 | AUC0-t (ng·min/mL): 2,039.49 ± 492.24Cmax (ng·mL-1): 1 | |
| 6.74 ± 4.47 | |||||
| Tmax (min): 15.00 ± 0.00 | |||||
| T1/2,λz (min): 770.36 ± 65.01 | |||||
| Vd/Fλz (L/kg): 60,036.51 ± 19,704.59 | |||||
| CL/F (L/h/kg): 4,999.34 ± 1,198.79 | |||||
| Male Beagle Dog | 1 (i.v.) | 100 mg/kg | 1 | AUC (mg/h/L): 1979.31 ± 1140.31, | (Sheng et al. 1993) |
| Ka (h): 10.2843 ± 2.5, | |||||
| t1/2α(h): 0.15 ± 0.26, | |||||
| t1/2β(h): 12.59 ± 8.83, | |||||
| CL (h): 60.70 ± 24.38, | |||||
| Vd (L): 699.53 ± 219.05 | |||||
| 1 (i.g.) | 280 mg/kg | 1 | AUC (μg·h/L): 777.29 ± 150.10, | ||
| Cmax (μg/L): 15.46 ± 4.20, | |||||
| Tmax (h): 3.71 ± 0.95, | |||||
| t1/2α(h): 0.63 ± 0.14, | |||||
| t1/2β (h): 34.82 ± 14.36, | |||||
| CL (h): 2.64 ± 0.55, | |||||
| Vd (L): 125.41 ± 32.55 | |||||
| Male Wistar rats | CRE (i.g.) | 1.2 g/kg | 1 | AUC (h·ng/mL): 707.91, | (Bao et al. 2010) |
| T1/2 (h): 1.89, | |||||
| Cmax (ng/mL): 315.78, | |||||
| Tmax (h): 1 | |||||
| 4 | AUC (h·ng/mL): 130.29, | ||||
| T1/2 (h): 1.71, | |||||
| Tmax (h): 1 | |||||
| 2.4 g/kg | 1 | AUC (h·ng/mL): 1220.32, | |||
| T1/2 (h): 2.29, | |||||
| Cmax (ng/mL): 501.58, | |||||
| Tmax (h): 1 | |||||
| 4 | AUC (h·ng/mL): 348.61, | ||||
| T1/2 (h): 2.64, | |||||
| Tmax (h): 1 | |||||
| 4.8 g/kg | 1 | AUC (h·ng/mL): 2424.62, | |||
| T1/2 (h): 4.79, | |||||
| Cmax (ng/mL): 584.57, | |||||
| Tmax (h): 1 | |||||
| 4 | AUC (h·ng/mL): 872.76, | ||||
| T1/2 (h): 5.89, | |||||
| Tmax (h): 1 | |||||
| Male Wistar rats | CRE (i.v.) | 10.2 mg/kg | 1 | Hippocampus: | (Wang et al. 2005) |
| AUC (ng·h/g): 6940 ± 206, | |||||
| Cmax (ng/g): 272 ± 12, | |||||
| T1/2Kα (h): 4.48 ± 1.7 | |||||
| T1/2α (h): 0.215 ± 0.063 | |||||
| T1/2β (h): 12.0 ± 1.5, | |||||
| Tmax (h): 3.67 ± 0.48 | |||||
| Plasma: | |||||
| AUC (ng·h/mL): 473 ± 18, | |||||
| T1/2α (h): 0.227 ± 0.017, | |||||
| T1/2β (h): 1.13 ± 0.18 | |||||
| Vd (mL/kg): 2400 ± 300 | |||||
| CL (mL·kg/h): 6400 ± 200 | |||||
| Male healthy volunteers | 300 mg 1 (p.o.) | 300 mg/kg | 1 | AUC (mg·h/L): 2799 ± 1128.5, | (Li & Zhang 1997) |
| Cmax (mg/L): 394.7 ± 155.4, | |||||
| Tmax (h): 2.37 ± 0.04, | |||||
| T1/2Kα(h): 0.87 ± 0.03, | |||||
| T1/2β (h): 2.94 ± 0.14 | |||||
| Male Wistar rats | CRE (i.g.) | 300 mg/kg | 1 | AUC0–t (ng·h/mL): 92.71 ± 15.03, | (Liu et al. 2014) |
| Cmax (ng/mL): 12.27 ± 3.30, | |||||
| Tmax (h): 1.90 ± 0.58, | |||||
| T1/2 (h): 3.96 ± 0.92 | |||||
| 4 | AUC0–t (ng·h/mL): 4.72 ± 0.79, | ||||
| Cmax (ng/mL): 0.74 ± 0.44, | |||||
| Tmax (h): 1.30 ± 0.40, | |||||
| T1/2 (h): 3.91 ± 0.80 | |||||
| 3 | AUC0–t (ng·h/mL): 1.39 ± 0.60, | ||||
| Cmax (ng/mL): 1.38 ± 0.54, | |||||
| Tmax (h): 14.28 ± 2.38, | |||||
| T1/2 (h): 4.02 ± 1.83 | |||||
| 5 | AUC0–t (ng·h/mL): 6.63 ± 1.70, | ||||
| Cmax (ng/mL): 1.05 ± 0.75, | |||||
| Tmax (h): 1.38 ± 0.54, | |||||
| T1/2 (h): 5.28 ± 1.44 | |||||
| 8 | AUC0–t (ng·h/mL): 7.11 ± 0.65, | ||||
| Cmax (ng/mL): 0.67 ± 0.23, | |||||
| Tmax (h) 1.38 ± 0.54, | |||||
| T1/2 (h): 4.35 ± 0.67 |
Notes: AUC: area under curve; C max: maximum concentration; CL: body clearance; i.g.: intragastric; i.v.: intravenous; MTT: mean transit time; p.o.: per os; T 1/2: half-life; T 1/2a: distribution half-life; T 1/2β: elimination half-life; T 1/2Kα: absorption half-life; T max: time to peak concentration; Vd: volume of distribution.
After oral intake of berberine, the AUC0-t in mice with PI-IBS was higher than that in normal mice, while the total body clearance decreased significantly (Gong et al. 2014). In a pharmacokinetic study, magnoflorine showed lower bioavailability and faster absorption and elimination. However, pharmacokinetic parameters altered remarkably when magnoflorine was administered in a CR decoction. Oral gavage of a CR decoction decreased the absorption and elimination rates of magnoflorine, which revealed the pharmacokinetic interactions between magnoflorine and the rest of ingredients in CR (Xue B et al. 2015). Berberine in plasma was quickly eliminated after intravenous injection of CR; however, berberine could penetrate the blood-brain barrier (BBB) and reached the hippocampus with a rapid increase and slow elimination (Wang et al. 2005; Table 8).
Toxicology
CR has been banned in Singapore in recent decades because of the suggestion that berberine aggravated jaundice and kernicterus in neonates with glucose-6-phosphate dehydrogenase deficiency (Wong 1980). In 2012, researchers found no organ toxicity or electrolyte imbalance in 20 patients administered with CR at a daily dose of 3 g for 1055 patient-days (Linn et al. 2012). In 2016, the ban of Chinese herbal medicines rich in berberine was officially lifted. Nevertheless, toxicity cannot be ignored. An acute toxicity study showed that the oral medial lethal dose (LD50) of the fibrous roots of CR was greater than 7000 mg/kg body weight in Kunming mice. A sub-chronic toxicity study showed that the no-observed-adverse effect level (NOAEL) was 1.88 g/kg body weight in rats, whereas 3.76 g/kg body weight resulted in liver and lung damage. An Ames test, a mouse micronucleus test, and a mouse sperm abnormality test provided negative results (Ning et al. 2015). The median acute oral lethal dose of the CRE was 2.95 g/kg in mice; however, the alkaloid-rich extract was much more toxic than the total extract of CR (Ma et al. 2010). In another study, the LD50 values of four alkaloids (berberine, coptisine, palmatine and epiberberine) were determined as 713.57, 852.12, 1533.68 and 1360 mg/kg, respectively. Likewise, the cytotoxicity of berberine was the highest and that of palmatine was the lowest toward HepG2 and 3T3-L1 cells. In a subchronic toxicity study, no mortality or morbidity was observed (Yi et al. 2013). To determine the NOAEL and the toxicity of CR, rats received repeated oral administration of CR for 13 weeks. No mortality or remarkable clinical signs were observed during this 13-week study. The NOAEL of CR was determined as 667 mg/kg/day for male rats and 2000 mg/kg/day for female rats (Lee et al. 2014). Oral berberine has caused respiratory failure, extrapyramidal system reactions, severe arrhythmia, liver function injury and even death in clinics in China (Li et al. 2008), which as believed to caused by its inhibitory effect on the human eag-related gene (hERG) potassium channel and induction of mitochondrial dysfunction (Pereira et al. 2008; Schramm et al. 2011). Furthermore, the authors reported that an AChE inhibitor significantly increased the acute toxicity of the CRE, whereas a cholinesterase reactivator significantly decreased the acute toxicity. Therefore, the authors suggested that the acute toxicity of the oral CR extract was related to AChE inhibition (Ma et al. 2011) Taking these findings together, we concluded that the toxic constituents of CR were the alkaloids, mainly berberine. However, the toxic mechanism of the CR alkaloids may be complicated and remains to be determined. The currently recommended doses of CR alkaloids and CR consumption are relatively safe (Ho et al. 2014). In fact, CR is seldom used alone in clinics; instead, it is usually prescribed with other medicines that could reduce its toxic effect.
Future perspectives and conclusions
Herbal medicines, including TCMs, are considered useful agents to treat various human diseases (Li et al. 2009; Peng et al. 2018). CR has a long history of being used as an important herbal medicine in Asian countries because of its reliable curative effects against various diseases. Nowadays, the most predominant traditional uses of CR have been confirmed by modern pharmacological research. So far, these investigations have reported that CR contains abundant isoquinoline alkaloids (especially berberine), which are also the active substances responsible for the pharmacological effects of this TCM. CR and berberine have a broad-spectrum antibacterial effect, manifesting as bacteriostasis at low concentrations and sterilization at high concentrations. This suggests that a combination of berberine or CR and conventional antibacterial drugs might exert a greater effect. Intensive research has indicated that CR has potential as a cardioprotective agent. In addition to reducing the incidence, it also protects the heart from MI/R injury. These properties are mainly attributed to berberine, coptisine, palmatine, epiberberine, jatrorrhizine and magnoflorine. Many studies have demonstrated modulation of the composition of the gut microbiota (enrichment of beneficial microbiota and inhibition of harmful microbiota) as one of the most important aspect for treating obesity, diabetes, and other metabolic disorders. As a natural compound with both anti-inflammatory and antitumor activities, berberine shows great potential in cancer treatment. However, the effects of berberine are not strong; therefore, structural modification of berberine is required. Moreover, CR containing various active components may be more effective than its single component berberine and could provide multiple therapeutic effects. There is a significant difference between the blood concentration and the tissue concentration. Therefore, to find a suitable pharmacokinetic marker for CR may be challenging but is necessary. Moreover, the pharmacokinetics of TCM should try to elucidate all the chemical components entering the body and their processes in the body (absorption, distribution, metabolism and excretion), with the aim of building a bridge between the complex chemical components and the systemic clinical effects, to reveal the underlying mechanism(s). Additionally, related target-organ toxicity evaluations are lacking. Thus, more work should be devoted to investigating the pharmacokinetics and features of CR and its active components, and further clinical studies are required to evaluate the potential curative effects and possible toxicities of CR and its active components toward the target organs. In addition, according to the current pharmacological research, berberine is not only the main active component but also the primary toxic component of CR. Consequently, it is crucial to develop a strategy to balance the pharmacological effects and toxicity of berberine. Besides, current reports on the original plants used to make CR, including C. chinensis, C. deltoidea and C. teeta, commonly focus on the chemical components and pharmacological effects of the roots because of their traditional use in TCM, and the other parts of the plants are often ignored and disposed of without pretreatment (Shen 2006). However, some previous reports revealed that the leaves of the CR plants also contain berberine (Li et al. 2004; Liu T et al. 2010). Therefore, further research is required to investigate the chemical constituents and pharmacological activities of the other parts of the original CR plants.
This present study systematically reviewed the traditional uses, botany, phytochemistry, pharmacology, and toxicology of CR to provide comprehensive information regarding this herbal medicine, which could be beneficial for highlighting the importance of CR and providing some clues for the future research of this herbal medicine.
Consent for publication
All authors have provided consent for publication in Pharmaceutical Biology.
Funding Statement
This work was supported by the Sichuan Provincial Administration of Traditional Chinese Medicine (No. 2018JC011).
Disclosure statement
The authors have no personal or financial conflict of interests associated with this work.
References
- Ashraf MJ, Baweja P. 2013. Obesity: the ‘huge' problem in cardiovascular diseases. Missouri Medicine. 110:499–504. [PMC free article] [PubMed] [Google Scholar]
- Bae J, Lee D, Yun KK, Gil M, Lee JY, Lee KJ. 2013. Berberine protects 6-hydroxydopamine-induced human dopaminergic neuronal cell death through the induction of heme oxygenase-1. Mol Cells. 35:151–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bao M, Cao Z, Yu D, Fu S, Zhang G, Yang P, Pan Y, Yang B, Han H, Zhou Q. 2012. Columbamine suppresses the proliferation and neovascularization of metastatic osteosarcoma U2OS cells with low cytotoxicity. Toxicol Lett. 215:174–180. [DOI] [PubMed] [Google Scholar]
- Bao TD, L YJ, Yang Q, Weng XG, Zhang YF, Dong Y, Wang YW, Zhu XX. 2010. LC/MS determination of berberine and palmatin in rats plasma after oral administration of extracts Rhizoma Coptidis and its pharmacokinetics study. Chin J Exp Trad Med Formul. 16:186–189. [Google Scholar]
- Bilian X, Daohua X, Wenshuang C. 2011. Effects of berberine on cancellous and cortical bone in rats induced by glucocorticoid. Chin Pharm Bull. 27:965–970. [Google Scholar]
- Bing L, Wang G, Jie Y, Pan X, Yang Z, Zang L. 2011. Berberine inhibits human hepatoma cell invasion without cytotoxicity in healthy hepatocytes. PLoS One. 6:e21416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boberek JM, Stach J, Good L. 2010. Genetic evidence for inhibition of bacterial division protein FtsZ by berberine. PLoS One. 5:e13745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Y, Xia Q, Luo R, Huang P, Sun Y, Shi Y, Jiang W. 2014. Berberine inhibits the growth of human colorectal adenocarcinoma in vitro and in vivo . J Nat Med. 68:53–62. [DOI] [PubMed] [Google Scholar]
- Caliceti C, Rizzo P, Cicero AFG. 2015. Potential benefits of berberine in the management of perimenopausal syndrome. Oxid Med Cell Longev. 2015:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calo L, Dong Y, Kumar R, Przyklenk K, Sanderson TH. 2013. Mitochondrial dynamics: An emerging paradigm in ischemia-reperfusion injury. Curr Pharm Des. 19:6848–6857. [DOI] [PubMed] [Google Scholar]
- Cao Y, Pan Q, Cai W, Shen F, Chen GY, Xu LM, Fan JG. 2016. Modulation of gut microbiota by berberine improves steatohepatitis in high-fat diet-fed BALB/C mice. Arch Iran Med. 19:197–203. [PubMed] [Google Scholar]
- Cao Q, Hong S, Li Y, Chen H, Shen Y, Shao K, Lu M, Dai H, Ma S, Dai G. 2018. Coptisine suppresses tumor growth and progression by down-regulating MFG-E8 in colorectal cancer. RSC Adv. 8:30937–30945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chai FN, Ma WY, Zhang J, Xu HS, Li YF, Zhou QD, Li XG, Ye XL. 2018. Coptisine from Rhizoma Coptidis exerts an anti-cancer effect on hepatocellular carcinoma by up-regulating miR-122. Biomed Pharm. 103:1002–1011. [DOI] [PubMed] [Google Scholar]
- Chakravarthy D, Muñoz AR, Su A, Hwang RF, Keppler BR, Chan DE, Halff G, Ghosh R, Kumar AP. 2018. Palmatine suppresses glutamine-mediated interaction between pancreatic cancer and stellate cells through simultaneous inhibition of survivin and COL1A1. Cancer Lett. 419:103–115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang W, Li K, Guan F, Yao F, Yu Y, Zhang M, Hatch GM, Chen L. 2016. Berberine pretreatment confers cardioprotection against ischemia-reperfusion injury in a rat model of type 2 diabetes. J Cardiovasc Pharmacol Ther. 21:486–494. [DOI] [PubMed] [Google Scholar]
- Chang CH, Yu B, Su CH, Chen DS, Hou YC, Chen YS, Hsu YM. 2014. Coptidis rhizome and Si Jun Zi Tang can prevent Salmonella enterica Serovar Typhimurium infection in mice. PLoS One. 9:e105362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z. 2006. Collation of complete effective preions for women’s diseases. Beijing: People's Medical Publishing House; [Google Scholar]
- Chen J, Wang F, Liu J, Lee SC, Wang X, Yang H. 2008. Analysis of alkaloids in Coptis chinensis Franch. by accelerated solvent extraction combined with ultra-performance liquid chromatographic analysis with photodiode array and tandem mass spectrometry detections. Anal Chim Acta. 613:184–195. [DOI] [PubMed] [Google Scholar]
- Chen L, Wang L, Zhang Q, Zhang S, Ye W. 2012. Non-alkaloid chemical constituents from Coptis chinensis . Chin J Chin Mater Med. 37:1241–1244. [PubMed] [Google Scholar]
- Chen R, Wu Y, Tian W. 2016. Research progress in chemical constituents and related metabolic pathways of Coptidis Rhizoma. Hunan J Trad Chin Med. 32:190–192. [Google Scholar]
- Chen HY, Ye XL, Cui XL, He K, Jin YN, Chen Z, Li XG. 2012. Cytotoxicity and antihyperglycemic effect of minor constituents from Rhizoma Coptis in HepG2 cells. Fitoterapia. 83:67–73. [DOI] [PubMed] [Google Scholar]
- Chen C, Zhang Y, Huang C. 2010. Berberine inhibits PTP1B activity and mimics insulin action. Biochem Biophy Res Commun. 397:543–547. [DOI] [PubMed] [Google Scholar]
- Chi L, Peng L, Pan N, Hu X, Zhang Y. 2014. The anti-atherogenic effects of berberine on foam cell formation are mediated through the upregulation of sirtuin 1. Int J Mol Med. 34:1087–1093. [DOI] [PubMed] [Google Scholar]
- Chidambara Murthy KN, Jayaprakasha GK, Patil BS. 2012. The natural alkaloid berberine targets multiple pathways to induce cell death in cultured human colon cancer cells. Eur J Pharmacol. 688:14–21. [DOI] [PubMed] [Google Scholar]
- Chinese Pharmacopoeia Commission 2008. Xin yao zhuan zheng biao zhun. Vol. 70 Beijing: China Medical Science Press. [Google Scholar]
- Chinese Pharmacopoeia Commission 2015. Chinese pharmacopoeia. Vol. 1 Beijing: Medical Science Press. [Google Scholar]
- Cho JY, Kim AR, Park MH. 2001. Lignans from the rhizomes of Coptis japonica differentially act as anti-inflammatory principles. Planta Med. 67:312–316. [DOI] [PubMed] [Google Scholar]
- Choi JS, Kim JH, Ali MY, Jung HJ, Min BS, Choi RJ, Kim GD, Jung HA. 2015. Anti-adipogenic effect of epiberberine is mediated by regulation of the RAF/MEK1/2/ERK1/2 and AMPKα/AKT pathways. Arch Pharm Res. 38:2153–2162. [DOI] [PubMed] [Google Scholar]
- Choi JS, Kim JH, Ali MY, Min BS, Kim GD, Jung HA. 2014. Coptis chinensis alkaloids exert anti-adipogenic activity on 3T3-L1 adipocytes by downregulating C/EBP-α and PPAR-γ. Fitoterapia. 98:199–208. [DOI] [PubMed] [Google Scholar]
- Choi UK, Kim MH, Lee NH. 2007. Optimization of antibacterial activity by gold-thread (Coptidis Rhizoma Franch) against streptococcus mutans using evolutionary operation-factorial design technique. J Microbiol Biotechnol. 17:1880–1884. [PubMed] [Google Scholar]
- Chu M, Ding R, Chu ZY, Zhang MB, Liu XY, Xie SH, Zhai YJ, Wang YD. 2014. Role of berberine in anti-bacterial as a high-affinity LPS antagonist binding to TLR4/MD-2 receptor. BMC Complement Altern Med. 14:89–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuanchong X. 2016. Berberine inhibits macrophages inflammatory depending on sirti [Master’s thesis]. Nanjing: Nanjing University of Chinese Medicine.
- Chueh WH, Lin JY. 2011. Berberine, an isoquinoline alkaloid in herbal plants, protects pancreatic islets and serum lipids in nonobese diabetic mice. J Agri Food Chem. 59:8021–8027. [DOI] [PubMed] [Google Scholar]
- Chueh WH, Lin JY. 2012. Berberine, an isoquinoline alkaloid, inhibits streptozotocin-induced apoptosis in mouse pancreatic islets through down-regulating Bax/Bcl-2 gene expression ratio. Food Chem. 132:252–260. [DOI] [PubMed] [Google Scholar]
- Cok A, Plaisier C, Salie MJ, Oram DS, Chenge J, Louters LL. 2011. Berberine acutely activates the glucose transport activity of GLUT1. Biochimie. 93:1187–1192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper SF, Mockle JA, Beliveau J. 1970. Alkaloids of Coptis groenlandica . Planta Med. 19:23–29. [DOI] [PubMed] [Google Scholar]
- Cui G, Huang X, Zhou K. 2008. Inhibitory effect of Rhizoma Coptidis and its main components berberine on the growth of human nasopharyngeal carcinoma CNE-2Z. Guangdong Med J. 29:737–739. [Google Scholar]
- Cui L, Liu M, Chang X, Sun K. 2016. The inhibiting effect of the Coptis chinensis polysaccharide on the type II diabetic mice. Biomed Pharmacother. 81:111–119. [DOI] [PubMed] [Google Scholar]
- Dan L, Guangshang C, Xixi S, Qianqian C, Hongsheng S. 2017. An overview of the antiarrhythmic study of alkaloids in Coptidis Rhizoma. Shandong J Trad Chin Med. 2017:164–166. [Google Scholar]
- Dhamgaye S, Devaux F, Vandeputte P, Khandelwal NK, Sanglard D, Mukhopadhyay G, Prasad R. 2014. Molecular mechanisms of action of herbal antifungal alkaloid berberine, in Candida albicans . PLoS One. 9:e104554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong J. 2003. Lv she bei yao fang. Shanghai: Shanghai science and technology publishing house. [Google Scholar]
- Durairajan SSK, Liu LF, Lu JH, Chen LL, Yuan Q, Chung SK, Huang L, Li XS, Huang JD, Li M. 2012. Berberine ameliorates β-amyloid pathology, gliosis, and cognitive impairment in an Alzheimer's disease transgenic mouse model. Neurobiol Aging. 33:2903–2919. [DOI] [PubMed] [Google Scholar]
- Fan G, Tang C, Yan L, Yang YD, Zhang Y. 2014. Analysis of monosaccharide compositions of polysaccharides in Coptidis Rhizoma by pre-column derivatization HPLC method. Chin J Exp Trad Med Formul. 20:74–78. [Google Scholar]
- Fan DL, Xiao XH, Ma XJ. 2008. Calorimetric study of the effect of protoberberine alkaloids in Coptis chinensis Franch on Staphylococcus aureus growth. Thermochim Acta. 480:49–52. [Google Scholar]
- Feng J. 2008. Clinical control study and propranolol in the treatment of arrhythmia associated with large doses of berberine and oryzanol and coenzyme Q10. Asia Pac Trad Med. 4:51–53. [Google Scholar]
- Feng M, Kong SZ, Wang ZX, He K, Zou ZY, Hu YR, Ma H, Li XG, Ye XL. 2017. The protective effect of coptisine on experimental atherosclerosis Apoe(-/-) mice is mediated by MAPK/NF-κB-dependent pathway. Biomed Pharmacother. 93:721–729. [DOI] [PubMed] [Google Scholar]
- Feng X, Yan D, Zhao KJ, Luo JY, Ren YS, Kong WJ, Han YM, Xiao XH. 2011. Applications of microcalorimetry in the antibacterial activity evaluation of various Rhizoma Coptidis. Pharmac Biol. 49:348–353. [DOI] [PubMed] [Google Scholar]
- Feng M, Zou Z, Zhou X, Hu Y, Ma H, Xiao Y, Li X, Ye X. 2017. Comparative effect of berberine and its derivative 8-cetylberberine on attenuating atherosclerosis in ApoE-/- mice. Int Immunopharmacol. 43:195–202. [DOI] [PubMed] [Google Scholar]
- Feng L, Hao Z, Hua H. 2007. The protective effect of coptisine on gastric mucosal injury induced by irritable mice. West Chin J Pharm Sci. 22:713–714. [Google Scholar]
- Flora of China Committee of Chinese Academy of Sciences 2004. Flora of China. Vol. 27 Beijing: Science Press; p. 593. [Google Scholar]
- Friedemann T, Schumacher U, Tao Y, Leung KM, Schröder S. 2015. Neuroprotective activity of coptisine from Coptis chinensis (Franch). Evid Based Complement Alternat Med. 2015:827308–827316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedemann T, Ying Y, Wang W, Kramer ER, Schumacher U, Fei J, Schröder S. 2016. Neuroprotective effect of Coptis chinensis in MPP+ and MPTP-Induced Parkinson's Disease Models. Am J Chinese Med. 44:907–925. [DOI] [PubMed] [Google Scholar]
- Fujiwara H, Nonaka G, Yagi A, Nishioka I. 1976. Studies on the components of the leaves of Coptis japonica Makino. I. The structures of coptiside I and II. Chem Pharm Bull. 24:407–413. [DOI] [PubMed] [Google Scholar]
- Gong Z, Chen Y, Zhang R, Wang Y, Guo Y, Yang Q, Zhang H, Dong Y, Weng X, Gao S, et al. 2014. Pharmacokinetic comparison of berberine in rat plasma after oral administration of berberine hydrochloride in normal and post inflammation irritable bowel syndrome rats. Int J Mol Sci. 15:456–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu N, Zhang X, Yu H. 2017. Based on sequencing of 16s rRNA gene elaborate effect of Coptis chinensis on intestinal microbial diversity in type 2 diabetes mellitus. Chin Trad Herbal Drugs. 48:3998–4004. [Google Scholar]
- Guo T, Shih-Lung W, Guo X, Li H, Zheng J, Rachel B, Liu M, Pei Y, Xu H, Cai Y. 2016. Berberine ameliorates hepatic steatosis and suppresses liver and adipose tissue inflammation in mice with diet-induced obesity. Sci Rep. 6:22612–22620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo J, Wang SB, Yuan TY, Wu YJ, Yan Y, Li L, Xu XN, Gong LL, Qin HL, Fang LH. 2013. Coptisine protects rat heart against myocardial ischemia/reperfusion injury by suppressing myocardial apoptosis and inflammation. Atherosclerosis. 231:384–391. [DOI] [PubMed] [Google Scholar]
- Guo 2002. Guo jia zhong cheng yao biao zhun. Beijing: The State Drug Administration.
- Hamsa TP, Kuttan G. 2012. Antiangiogenic activity of berberine is mediated through the downregulation of hypoxia-inducible factor-1, VEGF, and proinflammatory mediators. Drug Chem Toxicol. 35:57–70. [DOI] [PubMed] [Google Scholar]
- Han B, Kou S, He K, Han Y, Wang Y, Huang T, Zhou X, Xiao Y, Ye X, Li X. 2017. Anti-hypercholesterolemic effect of berbamine isolated from Rhizoma Coptidis in hypercholesterolemic zebrafish induced by high-cholesterol diet. Iran J Pharm Res. 17:292–306. [PMC free article] [PubMed] [Google Scholar]
- Hayashi K, Minoda K, Nagaoka Y, Hayashi T, Uesato S. 2007. Antiviral activity of berberine and related compounds against human cytomegalovirus. Bioorganic Med Chem Lett. 17:1562–1564. [DOI] [PubMed] [Google Scholar]
- He K, Kou SM, Zou ZY, Hu YR, Feng M, Han B, Li XG, Ye XL. 2016. Hypolipidemic effects of alkaloids from Rhizoma Coptidis in diet-induced hyperlipidemic hamsters. Planta Med. 82:690–697. [DOI] [PubMed] [Google Scholar]
- He K, Ma H, Xu HS, Zou ZY, Feng M, Li X, Ye XL. 2017. Anti-hyperlipidemic effects of Rhizoma Coptidis alkaloids are achieved through modulation of the enterohepatic circulation of bile acid and cross-talk between the gut microbiota and the liver. J Funct Foods. 35:205–215. [Google Scholar]
- Hirano H, Tokuhira T, Yokoi T, Shingu T. 1997. Isolation of free radical scavenger from Coptidis Rhizoma. Nat Med. 51:539–540. [Google Scholar]
- Ho CE, Goh YL, Zhang C. 2014. From prejudice to evidence: the case of Rhizoma Coptidis in Singapore. Evid Based Complement Alternat Med. 2014:871720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho YT, Yang JS, Li TC, Lin JJ, Lin JG, Lai KC, Ma CY, Wood WG, Chung JG. 2009. Berberine suppresses in vitro migration and invasion of human SCC-4 tongue squamous cancer cells through the inhibitions of FAK, IKK, NF-kappaB, U-PA and MMP-2 and -9. Cancer Lett. 279:155–162. [DOI] [PubMed] [Google Scholar]
- Hsu WH, Hsieh YS, Kuo HC, Teng CY, Huang H, Wang CJ, Yang SF, Liou YS, Kuo WH. 2007. Berberine induces apoptosis in SW620 human colonic carcinoma cells through generation of reactive oxygen species and activation of JNK/p38 MAPK and Fasl. Arch Toxicol. 81:719–728. [DOI] [PubMed] [Google Scholar]
- Hsu YY, Tseng YT, Lo YC. 2013. Berberine, a natural antidiabetes drug, attenuates glucose neurotoxicity and promotes Nrf2-related neurite outgrowth. Toxicol Appl Pharmacol. 272:719–728. [DOI] [PubMed] [Google Scholar]
- Huang Q. 2013. Effect of blood glucose on the expression of GLUT1 and GLUT4 in the myocardial of diabetic rats [Master's thesis]. Jinan: Shandong University.
- Huang Z, Han Z, Ye B, Dai Z, Shan P, Lu Z, Dai K, Wang C, Huang W. 2015. Berberine alleviates cardiac ischemia/reperfusion injury by inhibiting excessive autophagy in cardiomyocytes. Eur J Pharmacol. 762:1–10. [DOI] [PubMed] [Google Scholar]
- Huang Z, Wang L, Meng S, Wang Y, Chen T, Wang C. 2011. Berberine reduces both MMP-9 and emmprin expression through prevention of p38 pathway activation in PMA-induced macrophages. Int J Cardiol. 146:153–158. [DOI] [PubMed] [Google Scholar]
- Huang T, Xiao Y, Yi L, Li L, Wang M, Tian C, Ma H, He K, Wang Y, Han B, et al. 2017. Coptisine from Rhizoma Coptidis suppresses HCT-116 cells-related tumor growth in vitro and in vivo . Sci Rep. 7:38524–38534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hung TM, Na M, Min BS, Zhang X, Lee I, Ngoc TM, Thuong PT, Sok DE, Bae K. 2007. Protective effect of magnoflorine isolated from Coptidis Rhizoma on Cu2+-induced oxidation of human low density lipoprotein. Planta Med. 73:1281–1284. [DOI] [PubMed] [Google Scholar]
- Iii APO, Byrnes JR, Mackman N. 2014. Hyperlipidemia, tissue factor, coagulation, and simvastatin. Trends Cardiovasc Med. 24:95–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iizuka N, Miyamoto K, Hazama S, Yoshino S, Yoshimura K, Okita K, Fukumoto T, Yamamoto S, Tangoku A, Oka M. 2000. Anticachectic effects of Coptidis Rhizoma, an anti-inflammatory herb, on esophageal cancer cells that produce Interleukin 6. Cancer Lett. 158:35–41. [DOI] [PubMed] [Google Scholar]
- Ikuta A, Itokawa H. 1989. Protoberberine alkaloids from Coptis quinquefolia . Plant Med. 43:81–82. [Google Scholar]
- Iwazaki RS, Endo EH, Ueda-Nakamura T, Nakamura CV, Garcia LB, Filho BPD. 2010. In vitro antifungal activity of the berberine and its synergism with fluconazole. Antonie Van Leeuwenhoek. 97:201–205. [DOI] [PubMed] [Google Scholar]
- Jeong HW, Hsu KC, Lee JW, Ham M, Huh JY, Shin HJ, Kim WS, Kim JB. 2009. Berberine suppresses proinflammatory responses through AMPK activation in macrophages. Am J Physiol Endocrinol Metab. 296:E955–E964. [DOI] [PubMed] [Google Scholar]
- Jia Z, Lin L, Huang S, Zhu Z, Huang W, Huang Z. 2017. Inhibition of autophagy by berberine enhances the survival of H9C2 myocytes following hypoxia. Mol Med Rep. 16:1677–1684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia F, Zou G, Fan J, Yuan Z. 2010. Identification of palmatine as an inhibitor of West Nile Virus. Arch Virol. 155:1325–1329. [DOI] [PubMed] [Google Scholar]
- Jiang YY, Cui HM, Wang JL, Liu H, Dang MM, Zhang QY, Yang F, Kou JT, Tong XL. 2017. Protective role of berberine and Coptis chinensis extract on T2MD rats and associated islet Rin-5f cells. Mol Med Rep. 16:6981–6991. [DOI] [PubMed] [Google Scholar]
- Jiang S, Wang Y, Ren D, Li J, Yuan G, An L, Du P, Ma J. 2015. Antidiabetic mechanism of coptis chinensis polysaccharide through its antioxidant property involving the JNK pathway. Pharmaceutical Biol. 53:1022–1029. [DOI] [PubMed] [Google Scholar]
- Jie S, Li H, Tian Y, Guo D, Zhu J, Gao S, Jiang L. 2011. Berberine inhibits angiogenic potential of Hep G2 cell line through VEGF down-regulation in vitro. J Gastroenterol Hepatol. 26:179–185. [DOI] [PubMed] [Google Scholar]
- Jin H, Jin X, Cao B, Wang W. 2016. Berberine affects osteosarcoma via downregulating the caspase-1/IL-1β signaling axis. Oncol Rep. 37:729–736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kai H. 2017. Rhizoma Coptidis alkaloids exert their anti-hyperlipidemic effects through modulation of bile acids signaling and gut microbiota in hyperlipidemia C57BL/6J mice [Dissertation]. Chongqing: Southwest University.
- Kai H, Hu Y, Hang M, Zou Z, Xiao Y, Yong Y, Min F, Li X, Ye X. 2016. Rhizoma Coptidis alkaloids alleviate hyperlipidemia in B6 mice by modulating gut microbiota and bile acid pathways. Biochim Biophys Acta. 1862:1696–1709. [DOI] [PubMed] [Google Scholar]
- Kang JX, Liu J, Wang J, He C, Li FP. 2005. The extract of huanglian, a medicinal herb, induces cell growth arrest and apoptosis by upregulation of interferon-beta and TNF-alpha in human breast cancer cells. Carcinogenesis. 26:1934–1939. [DOI] [PubMed] [Google Scholar]
- Kang S, Li Z, Yin Z, Jia R, Song X, Li L, Chen Z, Peng L, Qu J, Hu Z, et al. 2015. The antibacterial mechanism of berberine against Actinobacillus Pleuropneumoniae . Nat Prod Res. 29:2203–2206. [DOI] [PubMed] [Google Scholar]
- Kaufmann D, Kaur DA, Tahrani A, Herrmann F, Wink M. 2016. Extracts from traditional Chinese medicinal plants inhibit acetylcholinesterase, a known Alzheimer's disease target. Molecules. 21:1161–1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ke W. 2007. Study on the effect of Rhizoma Coptidis alkaloids on colon cancer and its molecular mechanism [Master's thesis]. Chongqing: ChongQing Medical University.
- Kim HY, Shin HS, Park H, Kim YC, Yun YG, Park S, Shin HJ, Kim K. 2008. In vitro inhibition of coronavirus replications by the traditionally used medicinal herbal extracts, Cimicifuga Rhizoma, Meliae Cortex, Coptidis Rhizoma, and Phellodendron Cortex. J Clin Virol. 41:122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim YM, Ha YM, Jin YC, Shi LY, Lee YS, Kim HJ, Seo HG, Choi JS, Kim YS, Kang SS, et al. 2009. Palmatine from Coptidis Rhizoma reduces ischemia-reperfusion-mediated acute myocardial injury in the rat. Food Chem Toxicol. 47:2097–2102. [DOI] [PubMed] [Google Scholar]
- Kim S, Han J, Kim NY, Lee SK, Cho DH, Choi MY, Kim JS, Kim JH, Choe JH, Nam SJ, et al. 2012. Effect of berberine on p53 expression by TPA in breast cancer cells. Oncol Rep. 27:210–215. [DOI] [PubMed] [Google Scholar]
- Kim S, Oh SJ, Lee J, Han J, Jeon M, Jung T, Lee SK, Bae SY, Kim J, Gil WH, et al. 2013. Berberine suppresses TPA-induced fibronectin expression through the inhibition of VEGF secretion in breast cancer cells. Cell Physiol Biochem. 32:1541–1550. [DOI] [PubMed] [Google Scholar]
- Kim JS, Oh D, Yim MJ, Park JJ, Kang KR, Cho IA, Moon SM, Oh JS, You JS, Kim CS, et al. 2015. Berberine induces Fasl-related apoptosis through p38 activation in KB human oral cancer cells. Oncol Rep. 33:1775–1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SH, Shin EJ, Kim ED, Bayaraa T, Frost SC, Hyun CK. 2007. Berberine activates GLUT1-mediated glucose uptake in 3T3-L1 adipocytes. Biol Pharma Bull. 30:2120–2125. [DOI] [PubMed] [Google Scholar]
- Kong W, Li Z, Xiao X, Zhao Y, Zhang P. 2010. Activity of berberine on Shigella Dysenteriae investigated by microcalorimetry and multivariate analysis. J Therm Anal Calorim. 102:331–336. [Google Scholar]
- Kong D, Lu L, Teng S, Sun T, Deng Z, Li Y. 2013. Research on export current situation and countermeasure of Coptis chinensis from Shizhu county. Morder Chin Med. 15:701–703. [Google Scholar]
- Kong W, Zhao Y, Xiao X, Cheng J, Liu Y, Li Z. 2009. Comparison of anti-bacterial activity of four kinds of alkaloids in Rhizoma Coptidis based on microcalorimetry. Chinese J Chem. 27:1186–1190. [Google Scholar]
- Kong WJ, Zhang H, Song DQ, Xue R, Zhao W, Wei J, Wang YM, Shan N, Zhou ZX, Yang P, et al. 2009. Berberine reduces insulin resistance through protein kinase C-dependent up-regulation of insulin receptor expression. Metab Clin Exp. 58:109–119. [DOI] [PubMed] [Google Scholar]
- Kou S, Bing H, Yue W, Tao H, Kai H, Han Y, Xia Z, Ye X, Li X. 2016. Synergetic cholesterol-lowering effects of main alkaloids from Rhizoma Coptidis in HepG2 cells and hypercholesterolemia hamsters. Life Sci. 151:50–60. [DOI] [PubMed] [Google Scholar]
- Kou Y, Li L, Li H, Tan Y, Li B, Wang K, Du B. 2016. Berberine suppressed epithelial mesenchymal transition through cross-talk regulation of PI3k/AKT and RARα/RARβ in melanoma cells. Biochem Biophys Res Commun. 479:290–296. [DOI] [PubMed] [Google Scholar]
- Kuo HP, Chuang TC, Tsai SC, Tseng HH, Hsu SC, Chen YC, Kuo CL, Kuo YH, Liu JY, Kao MC. 2012. Berberine, an isoquinoline alkaloid, inhibits the metastatic potential of breast cancer cells via AKT pathway modulation. J Agric Food Chem. 60:9649–9658. [DOI] [PubMed] [Google Scholar]
- La X, Zhang L, Li Z, Peng Y, Wang Y. 2017. Berberine-induced autophagic cell death by elevating GRP78 levels in cancer cells. Oncotarget. 8:20909–20924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lao-Ong T, Chatuphonprasert W, Nemoto N, Jarukamjorn K. 2012. Alteration of hepatic glutathione peroxidase and superoxide dismutase expression in streptozotocin-induced diabetic mice by berberine. Pharm Biol. 50:1007–1012. [DOI] [PubMed] [Google Scholar]
- Lee WC, Kim JK, Kang JW, Oh WY, Jung JY, Kim YS, Jung HA, Choi JS, Lee SM. 2010. Palmatine attenuates d-galactosamine/lipopolysaccharide-induced fulminant hepatic failure in mice. Food Chem Toxicol. 48:222–228. [DOI] [PubMed] [Google Scholar]
- Lee YH, Kim D, Lee MJ, Kim MJ, Jang HS, Park SH, Lee JM, Lee HY, Han BS, Son WC. 2014. Subchronic toxicity study of Coptidis Rhizoma in rats. J Ethnopharmacol. 152:457–463. [DOI] [PubMed] [Google Scholar]
- Lei X. 1985. Master lei's discourse on drug processing. Jinan: Phoenix Science Press. [Google Scholar]
- Lei G, Dun B. 2002. Technical guide for the processing of traditional Chinese Medicine. Xi'an: World Book Publishing Xi'an Company. [Google Scholar]
- Li S. 2013. Processing technology of traditional Chinese medicine. Beijing: Chemical Industry Press. [Google Scholar]
- Li X. 2015. Coptis chinensis and its processed products and the material basis of comparative study on the antibacterial spectrum. [Master's thesis]. Wuhan: Hubei University of Traditional Medicine.
- Li H, Cai X, Li XF. 2008. New applications and the side effects of Rhizoma Coptidis in clinic. Med J Chin People Armed Police Forces. 19:661–663. [Google Scholar]
- Li H, Guo L, Jie S, Liu W, Zhu J, Du W, Fan L, Wang X, Fu B, Huang S. 2008. Berberine inhibits SDF-1-induced aml cells and leukemic stem cells migration via regulation of SDF-1 level in bone marrow stromal cells. Biomed Pharm. 62:573–578. [DOI] [PubMed] [Google Scholar]
- Li S, Li H, Yang D, Yu X, Irwin DM, Niu G, Tan H. 2017. Excessive autophagy activation and increased apoptosis are associated with palmitic acid-induced cardiomyocyte insulin resistance. J Diabetes Res. 2017:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li O, Pan Y, Wang Z. 2013. Berberine induced apoptosis of hepatoma cells by inhibiting the arachidonic acid metabolic pathway CPLA-COX2. Pharmacol Commun Chin. 4:31–32. [Google Scholar]
- Li J, Qiu DM, Chen SH, Cao SP, Xia XL. 2014. Suppression of human breast cancer cell metastasis by coptisine in vitro . Asian Pac J Cancer Prev. 15:5747 [DOI] [PubMed] [Google Scholar]
- Li ZF, Wang Q, Chen G, Hua HM, Yang SL, Feng YL, Pei YH. 2013. A new pyrrolidine derivative from the rhizome of Coptis chinensis . Chem Nat Compd. 49:493–494. [Google Scholar]
- Li YM, Wang LQ, Deng F, Zhou Q, Sun SQ. 2004. Analysis and assessment of Coptis chinensis for different parts, ages, and heights using fourier transform infrared spectroscopy. Acta Acad Med Sin. 26:614–617. [PubMed] [Google Scholar]
- Li ZF, Wang Q, Feng YL, Luo XJ, Fan MM, Yang SL. 2012. Chemical constituents from Coptis chinensis . Zhong Yao Cai. 35:1438–1441. [PubMed] [Google Scholar]
- Li CH, Wu DF, Ding H, Zhao Y, Zhou KY, Xu DF. 2014. Berberine hydrochloride impact on physiological processes and modulation of twist levels in nasopharyngeal carcinoma CNE-1 cells. Asian Pac J Cancer Prev. 15:1851–1857. [DOI] [PubMed] [Google Scholar]
- Li C, Xiong J, Cheng Z, Cheng S. 2014. Effect of berberine on proliferation and apoptosis of colon cancer HT-29 cells. World Chin J Digestol. 22:2572–2577. [Google Scholar]
- Li XG, Yang LG, Chen LX, Qiu F. 2012. Chemical constituents from the decoction of Coptis chinensis Franch. J Shenyang Pharm Univ. 29:193–198. [Google Scholar]
- Li B, Zhang M, Bao L. 1995. Study on the pharmacokinetics of berberine after oral administration in human being. J Harbin Med Univ. 29:382–385. [Google Scholar]
- Li M, Zhang M, Zhang ZL, Liu N, Han XY, Liu QC, Deng WJ, Liao CX. 2017. Induction of apoptosis by berberine in hepatocellular carcinoma HepG2 cells via downregulation of NF-κB. Oncol Res. 25:233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Zhou M, Zhao A, Jia W. 2009. Traditional Chinese medicine: balancing the gut ecosystem. Phytother Res. 23:1332–1335. [DOI] [PubMed] [Google Scholar]
- Li ZQ, Zuo DY, Qie XD, Qi H, Zhao MQ, Wu YL. 2012. Berberine acutely inhibits the digestion of maltose in the intestine. J Ethnopharmacol. 142:474–480. [DOI] [PubMed] [Google Scholar]
- Linn YC, Lu J, Lim LC, Sun H, Sun J, Zhou Y, Ng HS. 2012. Berberine-induced haemolysis revisited: Safety of Rhizoma Coptidis and Cortex Phellodendri in chronic haematological diseases. Phytotherapy Res. 26:682–686. [DOI] [PubMed] [Google Scholar]
- Liu R, Cao Z, Pan Y, Zhang G, Yang P, Guo P, Zhou Q. 2013. Jatrorrhizine hydrochloride inhibits the proliferation and neovascularization of C8161 metastatic melanoma cells. Anticancer Drugs. 24:667–676. [DOI] [PubMed] [Google Scholar]
- Liu D, Cao G, Si X, Chen Q, Sun H. 2017. An overview of the antiarrhythmic study of alkaloids in Coptidis Rhizoma. Shandong J Trad Chin Med. 3:164–166. [Google Scholar]
- Liu LZ, Cheung SC, Lan LL, Ho SK, Xu HX, Chan JC, Tong PC. 2010. Berberine modulates insulin signaling transduction in insulin-resistant cells. Mol Cell Endocrinol. 317:148–153. [DOI] [PubMed] [Google Scholar]
- Liu Y, Hao H, Xie H, Lai L, Wang Q, Liu C, Wang J. 2010. Extensive intestinal first-pass elimination and predominant hepatic distribution of berberine explain its low plasma levels in rats. Drug Metab Disp Bio Fate Chem. 38:1779–1784. [DOI] [PubMed] [Google Scholar]
- Liu G, He W, Cai H, Sun X, Hou W, Lin M, Xie Z, Liao Q. 2014. The simultaneous determination of berberine, palmatine, coptisine, epiberberine and jatrorrhizine in rat plasma by LC-MS/MS and a pharmacokinetic comparison after the oral administration of Rhizoma Coptidis and Jiao-Tai-Wan extract. Anal Methods. 6:2998. [Google Scholar]
- Liu T, Liang ZS, Ding MH, Zhao YH. 2010. Optimal extraction process of berberine hydrochloride from the leaves of Coptis chinensis . J Northwest Forest Univ. 25:144–147. [Google Scholar]
- Liu Z, Qiao L, Bing X, Wu J, Guo C, Zhu F, Yang Q, Gao G, Gong Y, Shao C. 2009. Berberine induces p53-dependent cell cycle arrest and apoptosis of human osteosarcoma cells by inflicting DNA damage. Mutat Res. 662:75–83. [DOI] [PubMed] [Google Scholar]
- Liu SJ, Sun YM, Tian DF, He YC, Zeng L, He Y, Ling CQ, Sun SH. 2008. Downregulated MM23-H1 expression is associated with intracranial invasion of nasopharyngeal carcinoma. Br J Cancer. 98:363–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Yu YL, Yang JS, Li Y, Liu YW, Liang Y, Liu XD, Xie L, Wang GJ. 2010. Berberine suppresses intestinal disaccharidases with beneficial metabolic effects in diabetic states, evidences from in vivo and in vitro study. Naunyn Schmiedebergs Arch Pharmacol. 381:371–381. [DOI] [PubMed] [Google Scholar]
- Liu XT, Chen X, Jin R. 2010. Protective effects of Rhizoma Coptidis on acute myocardial ischemia injured cardiomyocytes in vivo and in vitro . Chin J Infor Trad Chin Med. 28–30. [Google Scholar]
- Liu B, Fu XQ, Li T, Su T, Guo H, Zhu PL, Tse AKW, Liu SM, Yu ZL. 2017. Computational and experimental prediction of molecules involved in the anti-melanoma action of berberine. J Ethnopharmacol. 208:225–235. [DOI] [PubMed] [Google Scholar]
- Li MH, Zhang YJ, Yu YH, Yang SH, Iqbal J, Mi QY, Li B, Wang ZM, Mao WX, Xie HG, et al. 2014. Berberine improves pressure overload-induced cardiac hypertrophy and dysfunction through enhanced autophagy. Eur J Pharmacol. 728:67–76. [DOI] [PubMed] [Google Scholar]
- Lou T, Zhang Z, Xi Z, Liu K, Li L, Liu B, Huang F. 2011. Berberine inhibits inflammatory response and ameliorates insulin resistance in hepatocytes. Inflammation. 34:659–667. [DOI] [PubMed] [Google Scholar]
- Lu YX. 2004. The complete works of Chinese traditional crafts the preparation of Chinese Medicine. Zhengzhou: Elephant Press. [Google Scholar]
- Luo T, Shen X, Li S, Ouyang T, Wang H. 2016. The protective effect of jatrorrhizine against oxidative stress in primary rat cortical neurons. CNS Neurol Disord Drug Targets. 16:617–623. [DOI] [PubMed] [Google Scholar]
- Ma H, Chen G, Pei YH. 2013. Isolation and identification of chemical constituents from rhizoma of Coptis chinensis and their cytotoxic activities. J Shenyang Pharm Univ. 63:1–9. [Google Scholar]
- Ma H, Hu YR, Zou ZY, Feng M, Ye XL, Li XG. 2016. Antihyperglycemia and antihyperlipidemia effect of protoberberine alkaloids from Rhizoma Coptidis in HepG2 cell and diabetic kk-ay mice. Drug Develop Res. 77:163–170. [DOI] [PubMed] [Google Scholar]
- Ma X, Liu H, Chen G. 2015. Effects of berberine combined with photodynamic on apoptosis of gastric cancer MGC-803 cell. Chin J Clin Pharm Ther. 20:961–966. [Google Scholar]
- Ma BL, Ma YM. 2013. Pharmacokinetic properties, potential herb-drug interactions and acute toxicity of oral Rhizoma Coptidis alkaloids. Exp Opin Drug Metab Toxicol. 9:51–61. [DOI] [PubMed] [Google Scholar]
- Ma BL, Ma YM, Gao CL, Wu JS, Qiu FR, Wang CH, Wang XH. 2011. Lipopolysaccharide increased the acute toxicity of the Rhizoma Coptidis extract in mice by increasing the systemic exposure to Rhizoma Coptidis alkaloids. J Ethnopharmacol. 138:169–174. [DOI] [PubMed] [Google Scholar]
- Ma BL, Ma YM, Shi R, Wang TM, Zhang N, Wang CH, Yang Y. 2010. Identification of the toxic constituents in Rhizoma Coptidis. J Ethnopharmacol. 128:357–364. [DOI] [PubMed] [Google Scholar]
- Ma B, Zhu L, Zang X, Chen Y, Li D, Wang Y. 2013. Coptis chinensis inflorescence and its main alkaloids protect against ultraviolet-B-induced oxidative damage. J Func Foods. 5:1665–1672. [Google Scholar]
- Maiti M, Nandi R, Chaudhuri K. 1982. Sanguinarine: a monofunctional intercalating alkaloid. FEBS Lett. 142:280–284. [DOI] [PubMed] [Google Scholar]
- Mei J. 2008. Study on processing technology and quality control of Coptidis Rhizoma [Dissertation]. Wuhan: Hubei College of Traditional Chinese Medicine.
- Mei L. 2011. Analysis of 37 cases of frequent atrial premature beats in the treatment of berberine. Chin J Misdiagn. 11:4467–4467. [Google Scholar]
- Mendis S, Puska P, Norrving B. 2011. Global atlas on cardiovascular disease prevention and control. WHO. World Heart Federation and World Stroke Organization.
- Meng L, Shu X, Xu H, Zhang C, Yang L, Li Z, Ji G. 2016. Integrative analysis of metabolome and gut microbiota in diet-induced hyperlipidemic rats treated with berberine compounds. J Trans Med. 14:237–248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng F, Wang L, Zhang J, Yin Z, Zhang Q, Ye W. 2013. Non-alkaloid chemical constituents from the rhizome of Coptis teeta . J Chin Pharm Univ. 44:307–310. [Google Scholar]
- Min ZD, Murata H, Mizuno M, Kojima H, Tanaka T, Iinuma M, Kimura R. 1987. Phenolic constituents from seeds of Coptis japonica var. Dissecta. Phytochemistry. 26:2071–2074. [Google Scholar]
- Ming C, Zhang M, Liu Y, Kang J, Chu Z, Yin K, Ding L, Ran D, Xiao R, Yin Y. 2016. Role of berberine in the treatment of methicillin-resistant Staphylococcus aureus infections. Sci Rep. 6:24748–24756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishan MA, Ahmadiankia N, Matin MM, Heirani-Tabasi A, Shahriyari M, Bidkhori HR, Naderi-Meshkin H, Bahrami AR. 2015. Role of berberine on molecular markers involved in migration of esophageal cancer cells. Cell Mol Biol (Noisy-le-Grand). 61:37–43. [PubMed] [Google Scholar]
- Mizuno M, Kojima H, Iinuma M, Tanaka T, Goto K. 1992. Coumarin derivatives in Coptis trifolia . Ann Proc Gifu Coll Pharm. 42:717–719. [Google Scholar]
- Mizuno M, Kojima H, Tanaka T, Iinuma M, Min ZD, Murata H. 1988. Benzophenanthridine alkaloids from the seeds of Coptis japonica var. Gifu: Disscta Annual Proceedings of Gifu College of Pharmacy, 37.
- Mo C, Wang L, Zhang J, Numazawa S, Tang H, Tang X, Han XJuan, Li J, Yang M, Wang Z, et al. 2014. The crosstalk between Nrf2 and AMPK signal pathways is important for the anti-inflammatory effect of berberine in LPS-stimulated macrophages and endotoxin-shocked mice. Antioxid Redox Signal. 20:574–588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ning N, Wang YZ, Zou ZY, Zhang DZ, Wang DZ, Li XG. 2015. Pharmacological and safety evaluation of fibrous root of Rhizoma Coptidis. Environ Toxicol Pharmacol. 39:53–69. [DOI] [PubMed] [Google Scholar]
- Noguchi M, Kubo M, Hayashi T, Ono M. 1978. Studies on the pharmaceutical quality evaluation of crude drug preparations used in orient medicine "kampoo". Iii. Precipitation reaction of glycyrrhizin with alkaloids or alkaloidal crude drugs in aqueous solution. Chem Pharm Bull. 26:3652–3657. [DOI] [PubMed] [Google Scholar]
- Park SH, Sung JH, Kim EJ, Chung N. 2015. Berberine induces apoptosis via ros generation in PANC-1 and MIA-PaCa2 pancreatic cell lines. Braz J Med Biol Res. 48:111–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng LC, Kang S, Yin ZQ, Jia RY, Song X, Li L, Li ZW, Zou YF, Liang XX, Li LX. 2015. Antibacterial activity and mechanism of berberine against Streptococcus agalactiae . Int J Clin Exp Pathol. 8:5217–5223. [PMC free article] [PubMed] [Google Scholar]
- Peng W, Shen H, Lin B, Han P, Li CH, Zhang QY, Ye BZ, Rahman K, Xin HL, Qin LP, et al. 2018. Docking study and antiosteoporosis effects of a dibenzylbutane lignan isolated from Litsea cubeba targeting Cathepsin K and MEK1. Med Chem Res. 27:2062–2070. [Google Scholar]
- Pereira CV, Machado NG, Oliveira PJ. 2008. Mechanisms of berberine (natural yellow 18)-induced mitochondrial dysfunction: Interaction with the adenine nucleotide translocator. Toxicol Sci. 105:408–417. [DOI] [PubMed] [Google Scholar]
- Perrins JD. 1862. XLIII.-On berberine-contributions to its history and revision of its formula. J Chem Soc. 15:339–356. [Google Scholar]
- Qin-Wei Z, Yong-Guang LI. 2016. Berberine attenuates myocardial ischemia reperfusion injury by suppressing the activation of PI3K/AKT signaling. Exp Ther Med. 11:978–984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salehi S, Filtz TM. 2011. Berberine possesses muscarinic agonist-like properties in cultured rodent cardiomyocytes. Pharm Res. 63:335–340. [DOI] [PubMed] [Google Scholar]
- Schramm A, Baburin I, Hering S, Hamburger M. 2011. Herg channel inhibitors in extracts of Coptidis Rhizoma. Planta Med. 77:692–697. [DOI] [PubMed] [Google Scholar]
- Seo YS, Yim MJ, Kim BH, Kang KR, Lee SY, Oh JS, You JS, Kim SG, Yu SJ, Lee GJ, et al. 2015. Berberine-induced anticancer activities in fadu head and neck squamous cell carcinoma cells. Oncol Rep. 34:3025–3034. [DOI] [PubMed] [Google Scholar]
- Sha SM, Zhang YG, Bin XU, Wang HH, Kong XY. 2011. Effect of berberine on cell proliferation and apoptosis in gastric carcinoma cells. J Moder Oncol. 37:2109–2120. [Google Scholar]
- Shen J. 2006. The resource utilization and research progess of Coptidis Rhizoma. J Mod Med Health. 22:1038–1039. [Google Scholar]
- Shen N, Huan Y, Shen ZF. 2012. Berberine inhibits mouse insulin gene promoter through activation of amp activated protein kinase and may exert beneficial effect on pancreatic β-cell. Eur J Pharmacol. 694:120–126. [DOI] [PubMed] [Google Scholar]
- Sheng M, Sun Q, Wang H. 1993. Study on the pharmacokinetics of berberine hydrochloride in beagle dog vein and oral administration. Chin Pharmacol Bull. 9:64–67. [Google Scholar]
- Shi Y, Hu FB. 2014. The global implications of diabetes and cancer. Lancet. 383:1947–1948. [DOI] [PubMed] [Google Scholar]
- Shi HL, Wu XJ, Liu Y, Xie JQ. 2013. Berberine counteracts enhanced IL-8 expression of AGS cells induced by evodiamine. Life Sci. 93:830–839. [DOI] [PubMed] [Google Scholar]
- Shin HB, Choi MS, Yi CM, Lee J, Kim NJ, Inn KS. 2015. Inhibition of respiratory syncytial virus replication and virus-induced p38 kinase activity by berberine. Int Immunopharmacol. 27:65–68. [DOI] [PubMed] [Google Scholar]
- Shukla S, Rizvi F, Raisuddin S, Kakkar P. 2014. Foxo proteins′ nuclear retention and BH3-only protein Bim induction evoke mitochondrial dysfunction-mediated apoptosis in berberine-treated HepG2 cells. Free Radical Biol Med. 76:185–199. [DOI] [PubMed] [Google Scholar]
- Song SW, Qiu M, Chu Y, Chen DY, Wang XH, Su AR, Wu ZW. 2014. Downregulation of cellular c-Jun N-terminal protein kinase and NF-kappa B activation by berberine may result in inhibition of herpes simplex virus replication. Antimicrob Agents Chemother. 58:5068–5078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steenbergen C, Frangogiannis NG. 2012. Chapter 36 - Ischemic heart disease In: Joseph A. Hill, Eric N. Olson, editors. New York (NY): Muscle, Academic Press; p. 495–521. [Google Scholar]
- Su G, Wang H, Gao Y, Chen G, Pei Y, Bai J. 2017. 1H-NMR-based metabonomics of the protective effect of Coptis chinensis and berberine on cinnabar-induced hepatotoxicity and nephrotoxicity in rats. Molecules. 22:459–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun H, Wang N, Cang Z, Zhu C, Zhao L, Nie X, Cheng J, Xia F, Zhai H, Lu Y. 2016. Modulation of microbiota-gut-brain axis by berberine resulting in improved metabolic status in high-fat diet-fed rats. Obesity Facts. 9:365–378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun S, Wang K, Lei H, Li L, Tu M, Zeng S, Zhou H, Jiang H. 2014. Inhibition of organic cation transporter 2 and 3 may be involved in the mechanism of the antidepressant-like action of berberine. Progr Neuropsychopharmacol Biol Psychiatry. 49:1–6. [DOI] [PubMed] [Google Scholar]
- Sun Y, Xia M, Yan H, Han Y, Zhang F, Hu Z, Cui A, Ma F, Liu Z, Gong Q. 2017. Berberine attenuates hepatic steatosis and enhances energy expenditure in mice by inducing autophagy and fibroblast growth factor 21. Br J Pharmacol. 12:2139–2148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabas I, García-Cardeña G, Owens GK. 2015. Recent insights into the cellular biology of atherosclerosis. J Cell Biol. 209:13–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan HY, Wang N, Tsao SW, Zhang Z, Feng Y. 2014. Suppression of vascular endothelial growth factor via inactivation of eukaryotic elongation factor 2 by alkaloids in Coptidis Rhizome in hepatocellular carcinoma. Integr Cancer Ther. 13:425–434. [DOI] [PubMed] [Google Scholar]
- Tang S. 2011. Classified Materia Medica from historical classics for emergency. Beijing: China Medical Press. [Google Scholar]
- Tang LQ, Wei W, Chen LM, Liu S. 2006. Effects of berberine on diabetes induced by alloxan and a high-fat/high-cholesterol diet in rats. J Ethnopharmacol. 108:109–115. [DOI] [PubMed] [Google Scholar]
- Tang F, Wang D, Duan C, Huang D, Wu Y, Chen Y, Wang W, Xie C, Meng J, Wang L, et al. 2009. Berberine inhibits metastasis of nasopharyngeal carcinoma 5-8F cells by targeting Rho kinase-mediated Ezrin phosphorylation at Threonine 567. J Biol Chem. 284:27456–27466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan L, Li C, Chen H, Mo Z, Zhou J, Liu Y, Ma Z, Xu Y, Yang X, Xie J, et al. 2017. Epiberberine, a natural protoberberine alkaloid, inhibits urease of Helicobacter pylori and jack bean: Susceptibility and mechanism. Eur J Pharm Sci. 110:77–86. [DOI] [PubMed] [Google Scholar]
- Ti H. 2017. Effect of Rhizoma Coptidis decoction on intestinal flora and GPR43 pathway GLP-1 and PYY in SD rats with metabolic syndrome [Master's thesis]. Beijing: Beijing University of Chinese Medicine.
- Tjong Y, Ip S, Lao L, Fong HHS, Sung JJY, Berman B, Che C. 2011. Analgesic effect of Coptis chinensis rhizomes (Coptidis Rhizoma) extract on rat model of irritable bowel syndrome. J Ethnopharmacol. 135:754–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomita M, Kura S. 1956. Isolation of magnoflorine from Coptis japonica Makino. J Pharm Soc Jap. 76:1425–1426. [Google Scholar]
- Tsang CM, Cheung KC, Cheung YC, Man K, Lui VW, Feng Y, Tsao SW. 2015. Berberine suppresses ID-1 expression and inhibits the growth and development of lung metastases in hepatocellular carcinoma. Biochim Biophys Acta. 1852:541–551. [DOI] [PubMed] [Google Scholar]
- Varghese FS, Thaa B, Amrun SN, Simarmata D, Rausalu K, Nyman TA, Merits A, Mcinerney GM, Ng LF, Ahola T. 2016. The antiviral alkaloid berberine reduces chikungunya virus-induced mitogen-activated protein kinase signaling. J Virol. 90:9743–9757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang G. 1991. Bo ji fang. Beijing: Zhonghua Book Company. [Google Scholar]
- Wang W. 2016. A review on pharmacologic effects of effective ingredients in huanglian. Clin J Chin Med. 8:147–148. [Google Scholar]
- Wang N, Feng Y, Lau EP, Tsang C, Ching Y, Man K, Tong Y, Nagamatsu T, Su W, Tsao S. 2010. F-actin reorganization and inactivation of Rho signaling pathway involved in the inhibitory effect of Coptidis Rhizoma on hepatoma cell migration. Integr Cancer Ther. 9:354–364. [DOI] [PubMed] [Google Scholar]
- Wang Y, Huang Y, Lam KSL, Li Y, Wong WT, Ye H, Lau CW, Vanhoutte PM, Xu A. 2009. Berberine prevents hyperglycemia-induced endothelial injury and enhances vasodilatation via adenosine monophosphate-activated protein kinase and endothelial nitric oxide synthase. Cardiovasc Res. 82:484–492. [DOI] [PubMed] [Google Scholar]
- Wang HQ, Li K, Ma LL, Wu S, Hu J, Yan HY, Jiang JD, Li YH. 2017. Berberine inhibits enterovirus 71 replication by downregulating the MEK/ERK signaling pathway and autophagy. Virol J. 14:2–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang XJ, Lin S, Kang HF, Dai ZJ, Bai MH, Ma XL, Ma XB, Liu MJ, Liu XX, Wang BF. 2013. The effect of Rhizoma Coptidis and Coptis chinensis aqueous extract on radiation-induced skin injury in a rat model. BMC Compl Alt Med. 13:105–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Liu J, Ma A, Chen Y. 2015. Cardioprotective effect of berberine against myocardial ischemia/reperfusion injury via attenuating mitochondrial dysfunction and apoptosis. International J Clin Exp Med. 8:14513–14519. [PMC free article] [PubMed] [Google Scholar]
- Wang ZQ, Lu FE, Leng SH, Fang XS, Chen G, Wang ZS, Dong LP, Yan ZQ. 2008. Facilitating effects of berberine on rat pancreatic islets through modulating hepatic nuclear factor 4 alpha expression and glucokinase activity. World J Gastroenterol. 14:6004–6011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang N, Tan HY, Li L, Yuen MF, Feng Y. 2015. Berberine and Coptidis Rhizoma as potential anticancer agents: recent updates and future perspectives. J Ethnopharmacol. 176:35–48. [DOI] [PubMed] [Google Scholar]
- Wang M, Wang J, Tan R, Wu Q, Qiu H, Yang J, Jiang Q. 2013. Effect of berberine on PPAR α /NO activation in high glucose- and insulin-induced cardiomyocyte hypertrophy. Evid Based Complement Altern Med. 2013:285489–285497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Wang R, Xing D, Su H, Ma C, Ding Y, Du L. 2005. Kinetic difference of berberine between hippocampus and plasma in rat after intravenous administration of Coptidis Rhizoma extract. Life Sci. 77:3058–3067. [DOI] [PubMed] [Google Scholar]
- Wang L, Zhang SY, Chen L, Huang XJ, Zhang QW, Jiang RW, Yao F, Ye WC. 2014. New enantiomeric isoquinoline alkaloids from Coptis chinensis . Phytochem Lett. 7:89–92. [Google Scholar]
- Wang W, Zhang QW, Wen-Cai YE. 2007. Isoquinoline alkaloids from the rhizoma of Coptis chinensis . Chin J Nat Med. 5:348–350. [Google Scholar]
- Wang Q, Zhi-Feng LI, Chen G, Feng YL, M Ma M F, Pei YH. 2012. Chemical constituents from Coptis chinensis Franch. Chin J Exp Trad Medi Form. 35:1438–1441. [PubMed] [Google Scholar]
- Wang Y, Wang Q, Zhang L, Ke Z, Zhao Y, Wang D, Chen H, Jiang X, Gu M, Fan S, et al. 2017. Coptisine protects cardiomyocyte against hypoxia/reoxygenation-induced damage via inhibition of autophagy. Biochem Biophys Res Commun. 490:231–238. [DOI] [PubMed] [Google Scholar]
- Wang Q, Zhang M, Liang B, Shirwany N, Zhu Y, Zou MH. 2011. Activation of AMP-activated protein kinase is required for berberine-induced reduction of atherosclerosis in mice: The role of uncoupling protein 2. PLoS One. 6:e25436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wijck KV, Buurman WA. 2002. Ischemia-reperfusion injury. J Surg Res. 105:248–306.12125704 [Google Scholar]
- Wojtyczka RD, Dziedzic A, Kępa M, Kubina R, Kabała-Dzik A, Mularz T, Idzik D. 2014. Berberine enhances the antibacterial activity of selected antibiotics against coagulase-negative staphylococcus strains in vitro. Molecules. 19:6583–6596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong HB. 1980. Singapore kernicterus. Singapore Med J. 21:556–567. [PubMed] [Google Scholar]
- Wu Y, Chen Z, Huang X, Liu J, Cha Y, Wang F, Xu J, Sheng L, Ding H. 2016. Berberine protected rats against adiposity induced by high-fat diets. Int J Clin Exp Med. 9:148–155. [Google Scholar]
- Wu Y, Li JQ, Kim YJ, Wu J, Wang Q, Hao Y. 2011. In vivo and in vitro antiviral effects of berberine on influenza virus. Chin J Integr Med. 17:444–452. [DOI] [PubMed] [Google Scholar]
- Wu S, Xu L, Liu H, Tong X. 2015. Clinical application and dosage of Coptidis Rhizoma. Chin Clin Doctor. 43:92–94. [Google Scholar]
- Xiao P. 2002. Modern Chinese Materia Medica. Vol. 3 Beijing: Chemical Industry Press. [Google Scholar]
- Xie X, Chang X, Chen L, Huang K, Huang J, Wang S, Shen X, Liu P, Huang H. 2013. Berberine ameliorates experimental diabetes-induced renal inflammation and fibronectin by inhibiting the activation of Rhoa/Rock signaling. Mol Cell Endocrinol. 381:56 [DOI] [PubMed] [Google Scholar]
- Xie W, Gu D, Li J, Cui K, Zhang Y. 2011. Effects and action mechanisms of berberine and Rhizoma Coptidis on gut microbes and obesity in high-fat diet-fed c57bl/6j mice. PLoS One. 6:e24520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie J, Huang XY, Yin-Yan XU, Wang L. 2012. Berberine induces apoptosis of breast cancer MCF-7 cells:Its related oxidative stress mechanism. J Med Postgrad. 2:135–139. [Google Scholar]
- Xie X, Li W, Lan T, Liu W, Peng J, Huang K, Huang J, Shen X, Liu P, Huang H. 2011. Berberine ameliorates hyperglycemia in alloxan-induced diabetic C57BL/6 mice through activation of AKT signaling pathway. Endocrine J. 58:761–768. [DOI] [PubMed] [Google Scholar]
- Xu Z, Feng W, Shen Q, Yu N, Yu K, Wang S, Chen Z, Shioda S, Guo Y. 2017. Rhizoma Coptidis and berberine as a natural drug to combat aging and aging-related diseases via anti-oxidation and AMPK activation. Aging Dis. 8:760–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu RX, Sun XC, Ma CY, Yao YH, Li XL, Guo YL, Zhang Y, Li S, Li JJ. 2017. Impacts of berberine on oxidized ldl-induced proliferation of human umbilical vein endothelial cells. Am J Transl Res. 9:4375–4389. [PMC free article] [PubMed] [Google Scholar]
- Xue D, Zou Z, Chen B. 2015. Study on membrane injury mechanism of total alkaloids and berberine from Coptidis Rhizoma on aeromonas hydrophila. Chin J Chin Mater Med. 40:1787–1792. [PubMed] [Google Scholar]
- Xue B, Zhao Y, Miao Q, Miao P, Yang X, Sun G, Su J, Ye J, Wei B, Zhang Y, et al. 2015. In vitro and in vivo identification of metabolites of magnoflorine by LC LTQ-Orbitrap MS and its potential pharmacokinetic interaction in Coptidis Rhizoma decoction in rat. Biomed Chromatogr. 29:1235–1248. [DOI] [PubMed] [Google Scholar]
- Yahara S, Satoshiro M, Nishioka I, Nagasawa T, Oura H. 1985. Isolation and characterization of phenolic compounds from Coptidis Rhizoma. Chem Pharm Bull. 33:527–531. [Google Scholar]
- Yang TC, Chao HF, Shi LS, Chang TC, Lin HC, Chang WL. 2014. Alkaloids from Coptis chinensis root promote glucose uptake in C2C12 myotubes. Fitoterapia. 93:239–244. [DOI] [PubMed] [Google Scholar]
- Yang Y, Kang N, Xia H, Li J, Cheng L, Qiu F. 2010. Metabolites of protoberberine alkaloids in human urine following oral administration of Coptidis Rhizoma decoction. Planta Med. 16:1859–1863. [DOI] [PubMed] [Google Scholar]
- Yang Y, Li Y, Yin D, Chen S, Gao X. 2016. Coptis chinensis polysaccharides inhibit advanced glycation end product formation. J Med Food. 19:593–600. [DOI] [PubMed] [Google Scholar]
- Yang W, She L, Yu K, Yan S, Zhang X, Tian X, Ma S, Zhang X. 2016. Jatrorrhizine hydrochloride attenuates hyperlipidemia in a high-fat diet-induced obesity mouse model. Mol Med Rep. 14:3277–3284. [DOI] [PubMed] [Google Scholar]
- Yao Z. 2014. The prognosis role of prognostic nutritional index in malignant pleural mesothelioma and the mechanisms of berberine-induccd apoptosis and autophagy [Master's thesis]. Jinan: Shandong University.
- Yao W, Wang X, Xiao K. 2018. Protective effect of berberine against cardiac ischemia/reperfusion injury by inhibiting apoptosis through the activation of Smad7. Mole Cell Prob. 38:38–44. [DOI] [PubMed] [Google Scholar]
- Ye L. 2007. The study of berberine inhibition of Cyclin D1 related signaling pathway in tumor cells [Master's thesis]. Beijing: Beijing University of Chinese Medicine.
- Ye L, Liang S, Guo C, Yu X, Zhao J, Zhang H, Shang W. 2016. Inhibition of M1 macrophage activation in adipose tissue by berberine improves insulin resistance. Life Sci. 166:82–91. [DOI] [PubMed] [Google Scholar]
- Yi J, Ye X, Wang D, He K, Yang Y, Liu X, Li X. 2013. Safety evaluation of main alkaloids from Rhizoma Coptidis. J Ethnopharmacol. 145:303–310. [DOI] [PubMed] [Google Scholar]
- Yi T, Zhuang L, Song G, Zhang B, Li G, Hu T. 2015. AKT signaling is associated with the berberine-induced apoptosis of human gastric cancer cells. Nutr Cancer. 67:523–531. [DOI] [PubMed] [Google Scholar]
- Yin J, Hu R, Chen M, Tang J, Li F, Yang Y, Chen J. 2002. Effects of berberine on glucose metabolism in vitro . Metabolism. 51:1439–1443. [DOI] [PubMed] [Google Scholar]
- Yong Y, Baoshun Z, Chunya C. 2011. Cardiovascular pharmacological activity of berberine. Chin Trad Pat Med. 33:867–869. [Google Scholar]
- Yong Y, Xiaoli Y, Xuegang L. 2007. Antimicrobial effect of four alkaloids from Coptidis Rhizome. Lishizhen Med Mater Med Res. 18:3013–3014. [Google Scholar]
- Yoshikawa K, Hiroshi Kinoshita A, Arihara S. 1997a. Woorenol, a novel sesquineolignan with a unique spiro skeleton, from the rhizomes of Coptis japonica var. J Nat Prod. 60:511–513. [Google Scholar]
- Yoshikawa K, Hiroshi Kinoshita A, Arihara S. 1997b. Non-basic components of Coptis Rhizoma: Four new hemiterpenoid glucosides, two new phenylpropanoid glucosides and a new flavonoid glycoside from Coptis japonica var. dissecta. Nat Med. 51:244–248. [Google Scholar]
- Yoshikawa K, Hiroshi Kinoshita A, Kan Y, Arihara S. 1995. Neolignans and phenylpropanoids from the rhizomes of Coptis japonica var. Dissecta. Chem Pharm Bull. 43:578–581. [Google Scholar]
- Yu D, Fu S, Cao Z, Bao M, Zhang G, Pan Y, Liu W, Zhou Q. 2014. Unraveling the novel anti-osteosarcoma function of coptisine and its mechanisms. Toxicol Lett. 226:328–336. [DOI] [PubMed] [Google Scholar]
- Yu L, Li Q, Yu B, Yang Y, Jin Z, Duan W, Zhao G, Zhai M, Liu L, Yi D, et al. 2016. Berberine attenuates myocardial ischemia/reperfusion injury by reducing oxidative stress and inflammation response: Role of silent information regulator 1. Oxidative Med Cell Long. 2016:1689602–1689617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu L, Li F, Zhao G, Yang Y, Jin Z, Zhai M, Yu W, Zhao L, Chen W, Duan W. 2015. Protective effect of berberine against myocardial ischemia reperfusion injury: Role of notch1/hes1-pten/AKT signaling. Apoptosis. 20:796–810. [DOI] [PubMed] [Google Scholar]
- Yu Y, Liu L, Wang X, Liu X, Liu X, Xie L, Wang G. 2010. Modulation of glucagon-like peptide-1 release by berberine: in vivo and in vitro studies. Biochem Pharmacol. 79:1000–1006. [DOI] [PubMed] [Google Scholar]
- Yu S, Pang X, Deng Y, Liu L, Liang Y, Liu X, Xie L, Wang G, Wang X. 2007. A sensitive and specific liquid chromatography mass spectrometry method for simultaneous determination of berberine, palmatine, coptisine, epiberberine and jatrorrhizine from Coptidis Rhizoma in rat plasma. Int J Mass Spectrom. 268:30–37. [Google Scholar]
- Yu S, Yu Y, Liu L, Wang X, Lu s, Liang Y, Liu X, Xie L, Wang G. 2010. Increased plasma exposures of five protoberberine alkaloids from Coptidis Rhizoma in streptozotocin-induced diabetic rats: is P-GP involved? Planta Med. 76:876–881. [DOI] [PubMed] [Google Scholar]
- Yu S, Yu Y, Lu S, Liu L, Liu X. 2008. Pharmacokinetics of berberine, palmatine, coptisine, epiberberine and jatrorr-hizine from Coptidis Rhizoma in diabetic rats. J Chin Pharm Univ. 39:526–529. [Google Scholar]
- Yu C, Zhang H, Pan J, Hong Y, Ren J, Zhu D, Xiu H. 2000. Determination and preliminary studies of metabolism of berberine in human urine after oral administration. Chin J Clin Pharmacol. 1:36–39. [Google Scholar]
- Yu R, Zhang ZQ, Wang B, Jiang HX, Cheng L, Shen LM. 2014. Berberine-induced apoptotic and autophagic death of HepG2 cells requires AMPK activation. Cancer Cell Int. 14:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan X, Wang J, Tang X, Li Y, Xia P, Gao X. 2015. Berberine ameliorates nonalcoholic fatty liver disease by a global modulation of hepatic mRNA and lncRNA expression profiles. J Transl Med. 13:24–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J. 1996. Jingyue’s complete works. Shanghai: Shanghai Scientific & Technical Publishers. [Google Scholar]
- Zhang B, Cao A, Zhou J, Hu Z, Wu D. 2012. Effect of jatrorrhizine on delayed gastrointestinal transit in rat postoperative ileus. J Pharm Pharmacol. 64:413–419. [DOI] [PubMed] [Google Scholar]
- Zhang C, Jing P, Li N. 2013. Berberine inhibits the proliferation and induces apoptosis of human ovarian cancer SKOV3 cells. Basic Clin Med. 33:225–226. [Google Scholar]
- Zhang J, Tang H, Deng R, Wang N, Zhang Y, Wang Y, Liu Y, Li F, Wang X, Zhou L. 2015. Berberine suppresses adipocyte differentiation via decreasing CREB transcriptional activity. PLoS One. 10:e0125667–e0125675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Q, Xiao X, Li M, Li W, Yu M, Zhang H, Fan P, Wang Z, Zheng J. 2014. Berberine moderates glucose metabolism through the GNRH-GLP-1 and MAPK pathways in the intestine. BMC Complement Altern Med. 14:188–197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T, Yang S, Du J. 2014. Protective effects of berberine on isoproterenol-induced acute myocardial ischemia in rats through regulating HMGB1-TLR4 axis. Evid Based Complement Alternat Med. 2014:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z, Li B, Meng X, Yao S, Jin L, Yang J, Wang J, Zhang H, Zhang Z, Cai D, et al. 2016. Berberine prevents progression from hepatic steatosis to steatohepatitis and fibrosis by reducing endoplasmic reticulum stress. Sci Rep. 6:20848–20860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ZHANG W, XU Yc, GUO Fj, MENG Y, LI Ml. 2008. Anti-diabetic effects of cinnamaldehyde and berberine and their impacts on retinol-binding protein 4 expression in rats with type 2 diabetes mellitus. Chin Med J. 121:2124–2128. [PubMed] [Google Scholar]
- Zhao Y, Adjei AA. 2015. Targeting angiogenesis in cancer therapy: moving beyond vascular endothelial growth factor. Oncologist. 20:660–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao M, Xian YF, Ip SP, Fong HH, Che CT. 2010. A new and weakly antispasmodic protoberberine alkaloid from Rhizoma Coptidis. Phytother Res. 24:1414–1416. [DOI] [PubMed] [Google Scholar]
- Zhao Gl, Yu Lm, Gao Wl, Duan Wx, Jiang B, Liu Xd, Zhang B, Liu Zh, Zhai Me, Jin Zx, et al. 2016. Berberine protects rat heart from ischemia/reperfusion injury via activating JAK2/STAT3 signaling and attenuating endoplasmic reticulum stress. Acta Pharmacol Sin. 37:354–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong 1991. Zhong yao cheng fang zhi ji. Vol 4. Beijing: Pharmacopoeia Committee of the People's Republic of China Ministry of Health.
- Zhong 1998. Zhong yao cheng fang zhi ji. Vol 17. Beijing: Pharmacopoeia Committee of the People's Republic of China Ministry of Health.
- Zhou L, Yang F, Li G, Huang J, Liu Y, Zhang Q, Tang Q, Hu C, Zhang R. 2018. Coptisine induces apoptosis in human hepatoma cells through activating 67-kDa Laminin receptor/cGMP signaling. Front Pharmacol. 9:517–533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou JY, Zhou SW. 2011. Protective effect of berberine on antioxidant enzymes and positive transcription elongation factor b expression in diabetic rat liver. Fitoterapia. 82:184–189. [DOI] [PubMed] [Google Scholar]
- Zhou X, Li H, Shi Z, Gao S, Wei S, Li K, Wang J, Li J, Wang R, Gong M, et al. 2017. Inhibition activity of a traditional chinese herbal formula Huang-Lian-Jie-Du-Tang and its major components found in its plasma profile on neuraminidase-1. Sci Rep. 7:15549–15558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu DX. 2012. Danxi's mastery of medicine. Beijing: China Medical Science Press; [Google Scholar]
- Zhu G. 2015. Lei zheng huo ren shu. Tianjin: Tianjin Scientific & Techenical Press; [Google Scholar]
- Zhu T, Li LL, Xiao GF, Luo QZ, Liu QZ, Yao KT, Xiao GH. 2015. Berberine increases doxorubicin sensitivity by suppressing STAT3 in lung cancer. Am J Chinese Med. 43:1487–1502. [DOI] [PubMed] [Google Scholar]
- Zhu Q, Li Y. 2016. Berberine attenuates myocardial ischemia reperfusion injury by suppressing the activation of PI3K/AKT signaling. Exp Ther Med. 11:978–984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Ma N, Li HX, Tian L, Ba YF, Hao B. 2014. Berberine induces apoptosis and DNA damage in MG-63 human osteosarcoma cells. Mol Med Rep. 10:1734–1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou ZY, Hu YR, Ma H, Feng M, Li XG, Ye XL. 2016. Epiberberine reduces serum cholesterol in diet-induced dyslipidemia syrian golden hamsters via network pathways involving cholesterol metabolism. Eur J Pharmacol. 774:1–9. [DOI] [PubMed] [Google Scholar]
- Zuo F, Nakamura N, Akao T, Hattori M. 2006. Pharmacokinetics of berberine and its main metabolites in conventional and pseudo germ-free rats determined by liquid chromatography/ion trap mass spectrometry. Drug Metab Dispos. 34:2064–2072. [DOI] [PubMed] [Google Scholar]