Abstract
Background
Post-transplant lymphoproliferative disorder (PTLD) is a rare complication following solid organ transplantation and allogeneic hematopoietic stem cell transplantation (Allo-HSCT), which gives rise to high mortality rates.
Material/Methods
This was a single-center retrospective analysis based on 27 patients who were diagnosed with PTLD following Allo-HSCT between January 1, 2007 and June 2018 at the Chinese PLA General Hospital. The purpose of this analysis was to investigate responses and prognostic factors of rituximab-based treatment.
Results
Twenty-seven patients were treated with rituximab. Among them, 20 of 27 patients (74.07%) had a complete response, 2 of 27 patients (7.41%) had a partial response, 5 of 27 patients (18.52%) had no response, and 22 of 27 patients (81.48%) cleared Epstein-Barr virus (EBV) copies. There were no obvious side effects. The 1-year overall survival (OS) estimate was 46.8% (95% CI, 23.1–65.5%). Univariate analysis revealed that lower OS was correlated with Eastern Cooperative Oncology Group (ECOG) score standard (3–4), Epstein-Barr virus (EBV) viral load (≥106 copies/mL), bacteria or fungal infection, and EBV reactivation were positive after treatment with 1 or 2 doses of rituximab (P<0.05). Multivariate analysis showed that each of the following were independently associated with lower OS (P<0.05): female, ECOG score standard (3–4), and EBV reactivation were positive after treatment with 1 or 2 doses of rituximab.
Conclusions
Our results demonstrated that rituximab-based treatment was a safe and effective strategy for patients who were diagnosed with PTLD following Allo-HSCT. The identified prognostic factors may help to detect which PTLD patients are at a higher risk of mortality.
MeSH Keywords: Epstein-Barr Virus Infections, Graft vs Host Disease, Hematopoietic Stem Cell Transplantation, Lymphoproliferative Disorders, Transplants
Background
Post-transplant lymphoproliferative disorder (PTLD) is a rare complication following solid organ transplantation and allogeneic hematopoietic stem cell transplantation (Allo-HSCT), which gives rise to high mortality rates [1,2]. PTLDs are a heterogeneous group of diseases [3]. According to the 2016 World Health Organization (WHO) lymphoma classification, PTLDs are divided into infectious mononucleosis PTLD, plasmacytic hyperplasia PTLD, classical Hodgkin lymphoma PTLD, florid follicular hyperplasia PTLD, monomorphic PTLD, and polymorphic PTLD [4].
It has been reported that many risk factors, such as HLA mismatch, T-cell depletion of donor grafts, and use of antithymocyte globulin, can increase the incidence of PTLD [5,6]. The pathogenesis for PTLD is assumed to be linked with impairment of cellular immunity and EBV reactivation [7,8]. A clinical symptom of PTLD is fever alone or with lymphadenopathy, which is frequently accompanied by extra nodal sites [9,10].
The treatment of PTLD varies depending on the classification and site of the disease, ranging from reducing immunosuppression to administration of rituximab alone or with chemotherapy or radiotherapy. Treatment can also be comprised of a combination of all of these approaches. Rituximab has been efficacious to the treatment of CD20 positive PTLD [11–13].
The purpose of this analysis was to investigate responses and prognostic factors of rituximab-based treatment.
Material and Methods
This was a single-center retrospective analysis based on 27 patients who were diagnosed with PTLD following Allo-HSCT between January 1, 2007 and June 1, 2018 at the Chinese PLA General Hospital. The analysis included the following clinical characteristics for each patient: age, diagnosis, gender, conditioning regimens, EBV and cytomegalovirus (CMV) reactivation, and grade of graft-versus-host disease (GVHD). Written informed consent was obtained from each patient. Patients who participated in this study were approved by the Chinese PLA General Hospital ethical committee.
Conditioning regimen
The conditioning regimens were as follows: 1) Bu/Cy-conditioning regimen consisted of busulfan (3.2 mg/kg/day for 3 days) together with cyclophosphamide (60 mg/kg for 2 days), 2) FC-conditioning regimen consisted of fludarabine (30 mg/m2 for 5 days) together with cyclophosphamide (50 mg/m2 for 4 days), and 3) FB-conditioning regimen consisted of fludarabine (30 mg/m2/day for 4 days) with busulfan (3.2 mg/kg/day IV for 3 days). The conditioning regimen for all 27 patients also included 2.5 mg/kg/day thymoglobulin (Sangstat, Lyon, France) for 4 days.
Infection and GVHD prophylaxis
From the first day of conditioning, all patients received antiviral prophylaxis with acyclovir, as well as pneumocystis prophylaxis with cotrimoxazole. No systematic antibacterial prophylaxis was given before stem cell engraftment.
Acute and chronic GVHD were evaluated according to the Seattle standard criteria. Prophylaxis against GVHD consisted of 0.5 g mycophenolate mofetil every 12 hours starting on day 1 for 28 days, followed by cyclosporine starting on day 6, and a short course of methotrexate (15 mg/m2 on day +1; 10 mg/m2 on days +3, +6, and +11). Grades II to IV acute GVHD were usually treated with methylprednisolone 1–2 mg/kg/day. Extensive chronic GVHD was treated with prednisone 1 mg/kg/day.
EBV-reactivation
Quantitative PCR for detection of EBV copies of plasma, was performed as described elsewhere [14], the results were expressed as viral copy number per mL. EBV DNA load was monitored at least once a week during the first 4 or more months after transplantation, until EBV DNA became undetectable [15].
Diagnosis and treatment of PTLD
PTLD diagnosis was based on histopathology obtained by biopsy. Routinely, diagnostic tissue samples were examined independently by 2 pathologists with agreement on the diagnoses and subtype according to the 2016 WHO lymphoma classification. In order to detect lymphoproliferative sites, all patients diagnosed with PTLD underwent blood tests, and computed tomography (CT) scans. PTLD can be diagnosed as either probable or proven. The following criteria lead to a probable PTLD diagnosis: significant lymphadenopathy, hepatosplenomegaly, or other organ manifestations (without tissue biopsy, but in the absence of other documented cause). Conversely, a proven PTLD diagnosis is defined as a histologically diagnosed PTLD.
Patients accepted 4 weekly doses of rituximab (375 mg/m2), alone or combined with 1 to 4 courses of standard CHOP chemotherapy (cyclophosphamide 750 mg/m2 on day 1, doxorubicin 50 mg/m2 on day 1, vincristine 1.4 mg/m2 on day 1, prednisone 50 mg/m2 on day 1 to day 5) or COP therapy (cyclophosphamide 750 mg/m2 on day 1, prednisone 50 mg/m2 on day 1 to day 5).
Definitions
Overall survival (OS) was calculated from the onset of transplantation to the date of the latest follow-up or to the date of death. Response criteria are defined according to the International Harmonization Project on Lymphoma [16]. Safety data were evaluated using the NCICTCAE criteria version 3.0. The virologic response was also assessed based on EBV DNA copies reduction.
Statistical analysis
Descriptive statistics were used to report the 27 patients’ general characteristics. Percentages were reported for categorical variables, whereas medians and ranges were reported for continuous variables. The prognostic factors of PTLD were treated with rituximab and were evaluated as a time-dependent variable in a Cox proportional-hazards regression model. The probabilities of OS were estimated by the Kaplan-Meier survival method.
The impact of the following variables was analyzed: age at transplant (≥50 versus <50 years), gender (female versus male), acute GVHD at the time of PTLD diagnosis (>grade II versus grade I–II, chronic or absent), time from transplant to PTLD diagnosis (≥100 days versus <100 days), maximum EBV viral load (≥106 versus <106 copies/mL), EBV reactivation turned negative after 2 doses of rituximab (no versus yes), bacterial or fungal infection (with versus without), Eastern Cooperative Oncology Group (ECOG) score standard (3–4 versus 0–2) and LDH level (high vs. normal). Multivariable analysis included only candidate variables which were statistically significant at a P<0.15 level from univariate analysis [16]. The level of significance in all cases was set at P<0.05. Statistical tests were run on SPSS (version 18.0). Kaplan-Meier survival-curve analyses were performed using Prism 6.0 software (GraphPad Software).
Results
Patient characteristics
Patient characteristics are presented in Table 1. Twenty-seven cases of PTLD were identified, of which, 8 were females and 19 were males. The median age was 27 years old (range, 12–61 years old). Among all patients, 25 individuals had a mismatched family donor, whereas 2 individuals had matched unrelated donors. Primary diseases included acute lymphoblastic leukemia (n=9), acute myeloblastic leukemia (n=11), non-Hodgkin lymphoma (n=1), myelodysplastic syndrome (n=2), and severe aplastic anemia (n=4). Among the 27 patients, 1 patient developed platelets graft failure, whereas all patients developed a granulocyte graft successfully. The median time to neutrophil recovery was 11 days (range, 8–9 days), whereas the median time to platelet recovery was 15 days (range, 9–47 days). Clinically significant grade I–II acute GVHD occurred in 16 cases (59.26%), and grade III–IV acute GVHD occurred in 2 cases (7.41%). Extensive chronic GVHD was diagnosed in 1 case (3.70%), 24 cases (88.89%) had concomitant CMV infection, and 9 cases (33.33%) had hemorrhagic cystitis.
Table 1.
Patient and transplantation characteristics of my study population.
| Characteristics | N | % |
|---|---|---|
| Patient age, median (range) | 27 (12–61) years | |
| Patient gender | ||
| Male | 19 | 70.37 |
| Female | 8 | 29.63 |
| Diagnosis | ||
| AA | 4 | 14.82 |
| ALL | 9 | 33.33 |
| AML | 11 | 40.74 |
| MDS | 2 | 7.41 |
| NHL | 1 | 3.70 |
| Conditioning regimen | ||
| With ATG | 27 | 100 |
| Without ATG | 0 | 0 |
| Source of stem cell | ||
| PB | 27 | 100 |
| BM | 0 | 0 |
| Donor | ||
| Sibling | 25 | 92.59 |
| MUD | 2 | 7.41 |
| ABO compatibility | ||
| Yes | 14 | 51.85 |
| No | 13 | 48.15 |
| Hemorrhagic cystitis | ||
| Yes | 9 | 33.33 |
| No | 18 | 66.67 |
| EBV reactivation | ||
| Positive | 27 | 100 |
| Negative | 0 | 0 |
| CMV reactivation | ||
| Positive | 24 | 88.89 |
| Negative | 3 | 11.11 |
| GVHD | ||
| Without GVHD | 8 | 29.63 |
| Acute I–II GVHD | 16 | 59.26 |
| Acute III–IV GVHD | 2 | 7.41 |
| Chronic GVHD | 1 | 3.70 |
| Use basiliximab | ||
| Yes | 3 | 11.11 |
| No | 24 | 88.89 |
| Times of transplantation | ||
| >One time | 2 | 7.41 |
| One time | 25 | 92.59 |
| Condition regimen | ||
| FB | 7 | 25.93 |
| FC | 2 | 7.41 |
| Bu/Cy | 18 | 66.66 |
| Status at transplantation | ||
| CR | 15 | 55.56 |
| NR | 8 | 29.63 |
| Untreated | 4 | 14.81 |
| PTLD classification | ||
| Probable PTLD | 19 | 70.37 |
| Proven PTLD | 8 | 27.59 |
| PTLD onset time | ||
| <100 days post SCT | 24 | 88.89 |
| ≥100 days post SCT | 3 | 11.11 |
| MNC median (range) ×108/kg | 8.75 (4.81–23.12) | |
| CD34+ cell count median (range) ×106/kg | 3.75 (0.93–10.87) | |
| Neutrophil recovery(days) >0.5×109/l median (range) | 11 (8–9) | |
| Platelets recovery(days) >0.5×109/l median (range) | 15 (9–47) | |
| Median day of PTLD diagnosis (range) | 58 (22–202) | |
| Median maximum EBV copies/ml (range) | 127300 (8346–17740000) | |
PTLD – post-transplant lymphoproliferative disorder; AA – aplastic anemia; ALL – acute lymphoblastic leukemia; AML – acute myeloblastic leukemia; MDS – myelodysplastic syndrome; NHL – non-Hodgkin lymphoma; ATG – antithymoglobulins; PB – peripheral blood; BM – bone marrow; MUD – matched unrelated donor; CMV – cytomegalovirus; EBV – Epstein-Barr Virus; GVHD – graft-vs.-host disease; Bu – busulfan; Cy – cyclophosphamide; FB – fludarabine+busulfan; Ara-C – cytarabine; FC – fludarabine+cyclophosphamide; MNC – mononuclear cells count; CR – complete remission; NR – none remission.
EBV-reactivation and PTLD
As Table 2 shows, 27 cases had detectable EBV reactivation and the median of peak EBV DNA copies was 12.73×104 per mL (range=8.346×103–1.774×107 per mL).
Table 2.
Characteristics of 27 patients of PTLD.
| No | Age | Gender | Diagnosis | Regimen | PTLD classification | Symptom | Treatment | Peak EBV DNAemia, copies/ml | Histology | Outcome, cause of death |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 43 | Male | AML | FB | Proven | Fever, lymphadenopathy | Rx5 | 130000 | Bunkitt’s lymphoma | Dead GVHD |
| 2 | 15 | Male | AA | Bu/Cy | Probable | Fever, lymphadenopathy | Rx4 | 127300 | – | Alive |
| 3 | 15 | Male | AML | FB | Proven | Fever, lymphadenopathy | Rx4 | 5114000 | DLBCL | Dead PTLD |
| 4 | 16 | Male | ALL | Bu/Cy | Probable | Fever, lymphadenopathy | Rx4 | 49510 | – | Alive |
| 5 | 19 | Female | AML | Bu/Cy | Probable | Lymphadenopathy | Rx3 | 362500 | – | Alive |
| 6 | 19 | Male | AA | Bu/Cy | Probable | Fever, lymphadenopathy | Rx4 | 3850000 | – | Alive |
| 7 | 22 | Male | AML | Bu/Cy | Probable | lymphadenopathy | Rx3 | 10440 | – | Alive |
| 8 | 22 | Female | ALL | Bu/Cy | Probable | Fever, lymphadenopathy | Rx3 | 4362000 | – | Dead GVHD |
| 9 | 27 | Female | ALL | Bu/Cy | Probable | Fever, lymphadenopathy | Rx3 | 42810 | – | Alive |
| 10 | 35 | Male | ALL | Bu/Cy | Probable | Fever | Rx2 | 62700 | – | Dead GVHD |
| 11 | 40 | Female | ALL | FB | Proven | Lymphadenopathy | Rx3+COPx1 | 8346 | DLBCL | Dead fungal pneumonia |
| 12 | 42 | Male | ALL | Bu/Cy | Probable | Lymphadenopathy | Rx3 | 64450 | – | Alive |
| 13 | 51 | Male | AML | FB | Probable | Fever, lymphadenopathy | Rx4 | 1571000 | – | Dead fungal pneumonia |
| 14 | 56 | Male | AML | FB | Proven | Fever, lymphadenopathy | Rx3 | 1465000 | Polymorphic | Alive |
| 15 | 61 | Male | AML | FB | Probable | Fever, lymphadenopathy | Rx1 | 236000 | – | Dead AML |
| 16 | 12 | Male | AML | FB | Proven | Fever, lymphadenopathy | Rx4 | 35100 | DLBCL | Alive |
| 17 | 35 | Female | MDS | Bu/Cy | Probable | Fever, lymphadenopathy | Rx2 | 34560 | – | Dead MDS |
| 18 | 31 | Male | NHL | Bu/Cy | Probable | Lymphadenopathy | Rx4+CTL | 12260000 | – | Dead PTLD |
| 19 | 24 | Male | AA | FC | Probable | Fever, lymphadenopathy | Rx2 | 17740000 | – | Dead PTLD |
| 20 | 32 | Male | ALL | Bu/Cy | Probable | Fever, lymphadenopathy | Rx1 | 604900 | – | Dead ALL |
| 21 | 26 | Female | MDS | Bu/Cy | Proven | Fever, lymphadenopathy | Rx3 | 478800 | Polymorphic | Dead PTLD |
| 22 | 35 | Female | AML | Bu/Cy | Probable | Diarrhea, lymphadenopathy | Rx4 | 92650 | – | Alive |
| 23 | 27 | Male | AML | Bu/Cy | Probable | Nausea, vomiting | Rx4 | 236600 | – | Alive |
| 24 | 33 | Male | ALL | Bu/Cy | Probable | Fever, hepatic dysfunction | Rx2 | 15520 | – | Alive |
| 25 | 35 | Female | AML | Bu/Cy | Probable | Diarrhea | Rx4 | 19470 | – | Dead AML |
| 26 | 19 | Male | ALL | Bu/Cy | Proven | Fever | Rx4 | 20790 | DLBCL | Alive |
| 27 | 21 | Male | AA | FC | Proven | Fever | Rx4 | 49700 | DLBCL | Alive |
AA – aplastic anemia; ALL – acute lymphoblastic leukemia; AML – acute myeloblastic leukemia; MDS – myelodysplastic syndrome; NHL – non-Hodgkin lymphoma; ATG – antithymoglobulins; EBV – Epstein-Barr Virus; Bu – busulfan; Cy – cyclophosphamide; FB – fludarabine+busulfan; FC – fludarabine+cyclophosphamide; R – rituximab; RI – reduction of immunosuppression; DLBCL – diffuse large B cell lymphoma; CTL – cytotoxic lymphocyte; COP – cyclophosphamide+vincristine+prednisone.
PTLD was proven by biopsy in 8 cases, whereas 19 cases had probable PTLD. The median time to PTLD onset was 58 days (range, 22–202 days) after transplantation. According to the WHO classification, pathologic findings varied in the following ways: 2 cases had polymorphic PTLD, 5 cases had diffuse large B-cell lymphoma, 1 case had Burkitt lymphoma, 19 patients had fever, and 21 patients had lymphadenopathy. Among 8 proven PTLD cases, CD20 status was positive in 100.0% of individuals. An extra nodal site was involved in 14.81% (n=4) of cases and the location of the extra nodal sites were as follows: gastrointestinal tract (n=2), central nervous system (n=1), and liver (n=1).
Outcome of treatments
As Table 2 shows, the following outcomes occurred at the end of the study: 22 of 27 patients cleared EBV after rituximab treatment, 20 of 27 patients had a complete response, 2 of 27 patients had a partial response, and 4 of 27 patients exhibited progressive disease. One patient responded well to rituximab – combined with COP followed by 2 doses of rituximab treatment – but died of fungal pneumonia. One patient was given an EBV-specific CTL lymphocyte infusion followed by rituximab treatment, but died of PTLD progression. Taken together, 14 of 27 patients were still alive at the end of study, 4 patients died from PTLD, 3 patients died from GVHD, 2 patients died from fungal pneumonia, and the remaining 4 patients died from relapse of the primary disease. Figure 1 shows OS analysis by the Kaplan-Meier survival method.
Figure 1.
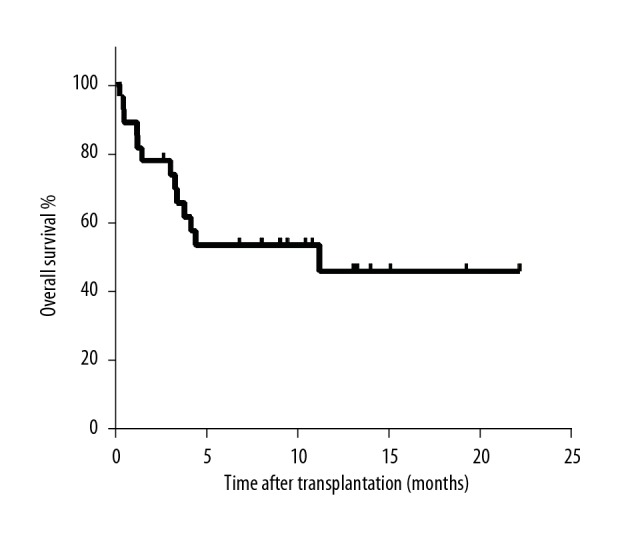
Kaplan-Meier survival curve of post-transplant lymphoproliferative disorder.
Side effects
There were no obvious side effects from rituximab infusion, and tolerance was generally good.
Prognosis factors for PTLD
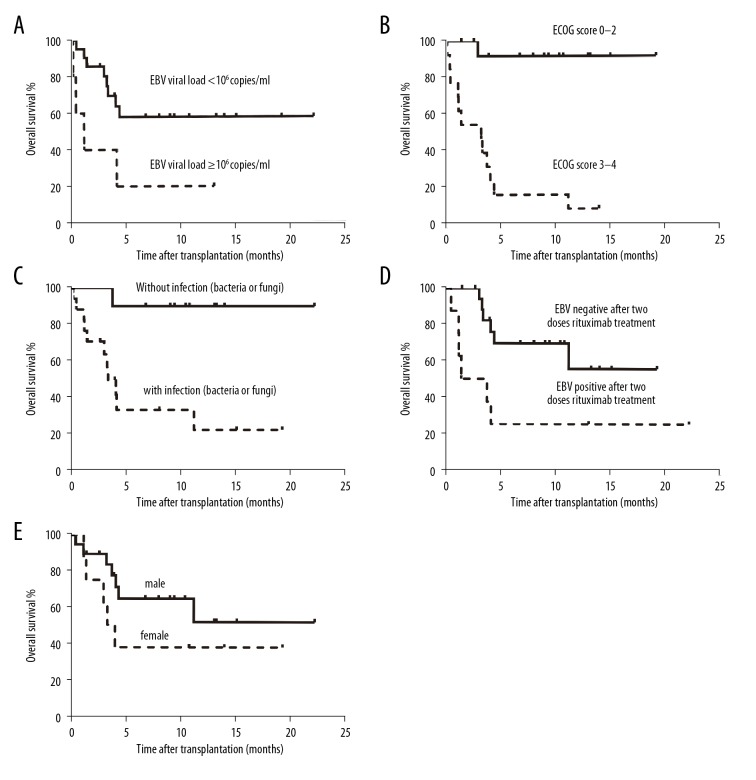
Univariate analysis revealed that lower OS was correlated with EBV was not negative after treatment with 1 or 2 doses of rituximab (P=0.007, HR=4.758,95%CI, 1.535–14.750), ECOG score standard ≥3–4 (P=0.002, HR=25.125, 95% CI, 3.190–197.884), EBV viral load ≥106 copies/mL (P=0.033, HR=3.408, 95% CI, 1.103–10.530) and with bacterial or fungal infection (P=0.004, HR=9.234, 95% CI, 1.995–42.743) (Table 3). Multivariate analysis identified that EBV was not negative after treatment with 1 or 2 doses of rituximab (P=0.033, HR=4.273,95% CI,1.076–16.974), ECOG score standard ≥3–4 (P=0.002, HR=36.986, 95% CI, 3.775–362.345), and females (P=0.018, HR=5.688, 95% CI, 1.352–23.921) were independently associated with lower OS (Table 4). Figure 2 shows the Kaplan-Meier survival-method analysis of 5 significant prognostic factors.
Table 3.
Univariate Cox proportional hazards regression techniques analyses for overall survival of PTLD.
| Factor | P | OR | 95%CI |
|---|---|---|---|
| Gender | 0.110 | 2.448 | 0.817–7.336 |
| Female | |||
| Male | |||
| Age | 0.272 | 2.350 | 0.511–10.806 |
| ≥50 years | |||
| <50 years | |||
| PTLD disease | 0.442 | 0.550 | 0.120–2.525 |
| <100 days post HSCT | |||
| ≥100 days post HSCT | |||
| EBV negative after two doses of rituximab | 0.007 | 4.758 | 1.535–14.750 |
| No | |||
| Yes | |||
| Acute GVHD III-IV | 0.616 | 1.352 | 0.415–4.401 |
| Yes | |||
| No | |||
| Infection (bacteria or fungi) | 0.004 | 9.234 | 1.995–42.743 |
| Yes | |||
| No | |||
| ECOG score standard | 0.002 | 25.125 | 3.190–197.884 |
| 3–4 | |||
| 0–2 | |||
| LDH | 0.754 | 1.387 | 0.179–10.771 |
| ≥250 U/L | |||
| <250 U/L | |||
| EBV viral load ≥106 copies/ml | 0.033 | 3.408 | 1.103–10.530 |
| Yes | |||
| No |
LDH – lactate dehydrogena.
Table 4.
Multivariate Cox proportional hazards regression techniques analyses for overall survival of PTLD.
| Factor | P | OR | 95%CI |
|---|---|---|---|
| Gender | 0.018 | 5.688 | 1.352–23.921 |
| Female | |||
| Male | |||
| EBV negative after two doses of rituximab | 0.039 | 4.273 | 1.076–16.974 |
| No | |||
| Yes | |||
| Infection (bacteria or fungi) | 0.254 | 2.726 | 0.486–15.285 |
| Yes | |||
| No | |||
| ECOG score standard | 0.002 | 36.986 | 3.775–362.345 |
| 3–4 | |||
| 0–2 | |||
| EBV viral load ≥106 copies/ml | 0.493 | 2.035 | 0.267–15.502 |
| Yes | |||
| No |
PTLD – post-transplant lymphoproliferative disorder; GVHD – graft-vs.-host disease.
Figure 2.
Overall survival (%) of post-transplant lymphoproliferative disorder under 5 prognostic factors following allogeneic stem cell transplantation. Significance is based on log-rank statistics, (A) stratified by Epstein-Barr virus (EBV) DNAemia copies, P=0.033; (B) stratified by Eastern Cooperative Oncology Group (ECOG) score standard, P=0; (C) stratified by infection, P=0.001; (D) stratified by EBV negative or positive after 2 doses rituximab treatment, P=0.003; (E) stratified by gender, P=0.099.
Discussion
PTLDs are dangerous and fatal complications after Allo-HSCTs. It has been reported that patients under 10 years of age, as well as patients over 60 years of age, are more likely to develop PTLDs [3,17,18]. “Early” PTLD occurs within the first year of transplantation and “late” PTLD occurs beyond the first year [11,19]. It has been reported that “early” PTLD may have a causative association with EBV reactivation [20,21]. In the present study, all patients had “early” PTLD and detectable EBV reactivation. Mortality of PTLD can be high, but varies among medical centers and also depends on the type of transplantation [22]. In the present study, 13 of 27 patients died, and the mortality rate was 48.15%.
The key of early diagnosis of PTLD is active awareness. When a patient exhibits adenopathy and fever after Allo-HSCT, a diagnosis of PTLD should be considered. To clarify the diagnoses, tissue biopsy is required. In addition, it is recommended that early diagnosis of PTLD be achieved through the monitoring of EBV DNA copies before onset of clinical symptoms.
Intense immunosuppression is one of the most common factors that stimulates PTLD in transplanted patients [17,23]. Strikingly, 55–65% of PTLDs are associated with EBV reactivation [24,25]. Immune control of EBV reactivation is mediated by T cell responses, but when patients are undergoing Allo-HSCT, the use of immunosuppression against GVHD leads to a profound deficit in cell response. This, in turn, leads to increased viral replication and to an accumulation of infected B-cells [26,27]. In the present study, EBV reactivation was detected in 29 of 29 patients (100%)
Current guidelines recommend that rituximab, reduction of immunosuppression, and EBV-specific cytotoxic-T-cell therapy are the first-line of therapies, while donor lymphocyte infusions (DLI) or chemotherapy are the second-line of therapies [15]. However, the response rates of reductions of immunosuppressive drugs are low [28]. Additionally, reduction of immunosuppression is known to be associated with a high risk of GVHD. EBV-specific cytotoxic-T-cell therapy is not available at most transplant centers. The remission rate of DLI is more than 70% for PTLD, but it can also increase the risk of GVHD [29]. Rituximab has become an available alternative therapy for PTLD, and may additionally reduce the risk of acute or chronic GVHD [30,31]. CD20 is the target of rituximab, and many reports demonstrate that rituximab can inhibit B-cell proliferation [32,33], which induces some patients to achieve long-term disease-free survival. In addition, rituximab-based treatment – following failure of reduced immunosuppression – is an alternative therapy of PTLD [34]. In this analysis, 20 of 27 patients had a complete response, whereas 2 of 27 patients had a partial response. One patient was diagnosed with central nervous system PTLD, for which there is no standard therapy, although possible therapeutic options include rituximab alone or combined with chemotherapy through either intravenous [35] or intrathecal injections [36]. After treatment with intravenous injection of rituximab alone, clinical symptoms of nausea and vomiting disappeared and cerebrospinal fluid EBV is negative. In summary, the therapeutic effect in the present study suggests that rituximab is an efficacious choice for PTLD patients. There were no obvious side effects, and tolerance of rituximab infusion was generally good. Additionally, there was no hematological toxicity and no patients experienced graft rejection.
In the era of PTLD treatments becoming rituximab-based, it is necessary to obtain detailed knowledge about prognostic factors. Poor PTLD prognosis is associated with poor performance status, CNS disease, late onset of disease, advanced age, disease involving multiple sites, and elevated LDH [37,38]. Additionally, the initial response to rituximab is regarded as a reliable prognostic factor for PTLD [39]. In this study, the initial responses of 10 patients to rituximab were positive and all were still alive. Univariate analysis revealed that lower OS was correlated with EBV was not negative after treatment with 1 or 2 dose of rituximab, ECOG score standard ≥3–4, EBV DNA load ≥106 copies/ml and with bacterial or fungal infection. Multivariate analysis identified that EBV was not negative after treatment with 1 or 2 doses of rituximab, ECOG score standard ≥3–4 and females were independently associated with lower OS.
Several concerns have been raised [40], including the fact that rituximab cannot restore immunity to EBV reactivation. Administration of rituximab to individuals who develop EBV copies exceed a threshold associated with PTLD, and it has led to an obvious reduction in mortality [41,42].
The major limitations of the present study were its small sample size and the short follow-up time. Despite these drawbacks, we identified that a rituximab-based treatment is a safe and effective strategy for patients who were diagnosed as PTLD. Further studies with more patients from multi-centers will be required.
Conclusions
The diagnosis of PTLD can be established on a proven or probable level. Rituximab-based treatment is a safe and effective strategy for patients who are diagnosed with PTLD following Allo-HSCT. Identified prognostic factors may help to identify patients who have a higher risk of mortality.
Footnotes
Source of support: Departmental sources
References
- 1.Baker KS, DeFor TE, Burns LJ, et al. New malignancies after blood or marrow stem-cell transplantation in children and adults: Incidence and risk factors. J Clin Oncol. 2003;21:1352–58. doi: 10.1200/JCO.2003.05.108. [DOI] [PubMed] [Google Scholar]
- 2.Landgren O, Kyle RA, Hoppin JA, et al. Pesticide exposure and risk of monoclonal gammopathy of undetermined significance in the Agricultural Health Study. Blood. 2009;113:6386–91. doi: 10.1182/blood-2009-02-203471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Al-Mansour Z, Nelson BP, Evens AM. Post-transplant lymphoproliferative disease (PTLD): Risk factors, diagnosis, and current treatment strategies. Curr Hematol Malig Rep. 2013;8:173–83. doi: 10.1007/s11899-013-0162-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375–90. doi: 10.1182/blood-2016-01-643569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Curtis RE, Travis LB, Rowlings PA, et al. Risk of lymphoproliferative disorders after bone marrow transplantation: A multi-institutional study. Blood. 1999;94:2208–16. [PubMed] [Google Scholar]
- 6.Bustami RT, Ojo AO, Wolfe RA, et al. Immunosuppression and the risk of post-transplant malignancy among cadaveric first kidney transplant recipients. Am J Transplant. 2004;4:87–93. doi: 10.1046/j.1600-6135.2003.00274.x. [DOI] [PubMed] [Google Scholar]
- 7.Hanto DW, Gajl-Peczalska KJ, Frizzera G, et al. Epstein-Barr virus (EBV) induced polyclonal and monoclonal B-cell lymphoproliferative diseases occurring after renal transplantation. Clinical, pathologic, and virologic findings and implications for therapy. Ann Surg. 1983;198:356–69. doi: 10.1097/00000658-198309000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pourfarziani V, Taheri S, Lessan-Pezeshki M, et al. Lymphoma after living donor kidney transplantation: An Iranian multicenter experience. Int Urol Nephrol. 2008;40:1089–94. doi: 10.1007/s11255-008-9377-0. [DOI] [PubMed] [Google Scholar]
- 9.Dodd GD, 3rd, Greenler DP, Confer SR. Thoracic and abdominal manifestations of lymphoma occurring in the immunocompromised patient. Radiol Clin North Am. 1992;30:597–610. [PubMed] [Google Scholar]
- 10.Bakker NA, van Imhoff GW, Verschuuren EA, et al. Early onset post-transplant lymphoproliferative disease is associated with allograft localization. Clin Transplant. 2005;19:327–34. doi: 10.1111/j.1399-0012.2005.00342.x. [DOI] [PubMed] [Google Scholar]
- 11.Kremer BE, Reshef R, Misleh JG, et al. Post-transplant lymphoproliferative disorder after lung transplantation: A review of 35 cases. J Heart Lung Transplant. 2012;31:296–304. doi: 10.1016/j.healun.2011.10.013. [DOI] [PubMed] [Google Scholar]
- 12.Kumarasinghe G, Lavee O, Parker A, et al. Post-transplant lymphoproliferative disease in heart and lung transplantation: Defining risk and prognostic factors. J Heart Lung Transplant. 2015;34:1406–14. doi: 10.1016/j.healun.2015.05.021. [DOI] [PubMed] [Google Scholar]
- 13.Trappe R, Oertel S, Leblond V, et al. Sequential treatment with rituximab followed by CHOP chemotherapy in adult B-cell post-transplant lymphoproliferative disorder (PTLD): The prospective international multicentre phase 2 PTLD-1 trial. Lancet Oncol. 2012;13:196–206. doi: 10.1016/S1470-2045(11)70300-X. [DOI] [PubMed] [Google Scholar]
- 14.Niesters HG, van Esser J, Fries E, et al. Development of a real-time quantitative assay for detection of Epstein-Barr virus. J Clin Microbiol. 2000;38:712–15. doi: 10.1128/jcm.38.2.712-715.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Styczynski J, van der Velden W, Fox CP, et al. Management of Epstein-Barr Virus infections and post-transplant lymphoproliferative disorders in patients after allogeneic hematopoietic stem cell transplantation: Sixth European Conference on Infections in Leukemia (ECIL-6) guidelines. Haematologica. 2016;101:803–11. doi: 10.3324/haematol.2016.144428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cheson BD, Pfistner B, Juweid ME, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007;25:579–86. doi: 10.1200/JCO.2006.09.2403. [DOI] [PubMed] [Google Scholar]
- 17.Cox KL, Lawrence-Miyasaki LS, Garcia-Kennedy R, et al. An increased incidence of Epstein-Barr virus infection and lymphoproliferative disorder in young children on FK506 after liver transplantation. Transplantation. 1995;59:524–29. [PubMed] [Google Scholar]
- 18.Opelz G, Dohler B. Lymphomas after solid organ transplantation: A collaborative transplant study report. Am J Transplant. 2004;4:222–30. doi: 10.1046/j.1600-6143.2003.00325.x. [DOI] [PubMed] [Google Scholar]
- 19.Thomas de Montpreville V, Le Pavec J, Le Roy Ladurie F, et al. Lymphoproliferative disorders after lung transplantation: clinicopathological characterization of 16 cases with identification of very-late-onset forms. Respiration. 2015;90:451–59. doi: 10.1159/000441064. [DOI] [PubMed] [Google Scholar]
- 20.Poirel HA, Bernheim A, Schneider A, et al. Characteristic pattern of chromosomal imbalances in posttransplantation lymphoproliferative disorders: Correlation with histopathological subcategories and EBV status. Transplantation. 2005;80:176–84. doi: 10.1097/01.tp.0000163288.98419.0d. [DOI] [PubMed] [Google Scholar]
- 21.Tsai DE, Hardy CL, Tomaszewski JE, et al. Reduction in immunosuppression as initial therapy for posttransplant lymphoproliferative disorder: Analysis of prognostic variables and long-term follow-up of 42 adult patients. Transplantation. 2001;71:1076–88. doi: 10.1097/00007890-200104270-00012. [DOI] [PubMed] [Google Scholar]
- 22.Soler MJ, Puig JM, Mir M, et al. Post-transplant lymphoproliferative disease: Treatment and outcome in renal transplant recipients. Transplant Proc. 2003;35:1709–13. doi: 10.1016/s0041-1345(03)00667-5. [DOI] [PubMed] [Google Scholar]
- 23.Shapiro R, Nalesnik M, McCauley J, et al. Posttransplant lymphoproliferative disorders in adult and pediatric renal transplant patients receiving tacrolimus-based immunosuppression. Transplantation. 1999;68:1851–54. doi: 10.1097/00007890-199912270-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oton AB, Wang H, Leleu X, et al. Clinical and pathological prognostic markers for survival in adult patients with post-transplant lymphoproliferative disorders in solid transplant. Leuk Lymphoma. 2008;49:1738–44. doi: 10.1080/10428190802239162. [DOI] [PubMed] [Google Scholar]
- 25.Nourse JP, Jones K, Gandhi MK. Epstein-Barr Virus-related post-transplant lymphoproliferative disorders: Pathogenetic insights for targeted therapy. Am J Transplant. 2011;11:888–95. doi: 10.1111/j.1600-6143.2011.03499.x. [DOI] [PubMed] [Google Scholar]
- 26.Burns DM, Crawford DH. Epstein-Barr virus-specific cytotoxic T-lymphocytes for adoptive immunotherapy of post-transplant lymphoproliferative disease. Blood Rev. 2004;18:193–209. doi: 10.1016/j.blre.2003.12.002. [DOI] [PubMed] [Google Scholar]
- 27.Kuehnle I, Huls MH, Liu Z, et al. CD20 monoclonal antibody (rituximab) for therapy of Epstein-Barr virus lymphoma after hemopoietic stem-cell transplantation. Blood. 2000;95:1502–5. [PubMed] [Google Scholar]
- 28.Swinnen LJ, LeBlanc M, Grogan TM, et al. Prospective study of sequential reduction in immunosuppression, interferon alpha-2B, and chemotherapy for posttransplantation lymphoproliferative disorder. Transplantation. 2008;86:215–22. doi: 10.1097/TP.0b013e3181761659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Heslop HE, Brenner MK, Rooney CM. Donor T cells to treat EBV-associated lymphoma. N Engl J Med. 1994;331:679–80. doi: 10.1056/NEJM199409083311017. [DOI] [PubMed] [Google Scholar]
- 30.Ratanatharathorn V, Ayash L, Reynolds C, et al. Treatment of chronic graft-versus-host disease with anti-CD20 chimeric monoclonal antibody. Biol Blood Marrow Transplant. 2003;9:505–11. doi: 10.1016/s1083-8791(03)00216-7. [DOI] [PubMed] [Google Scholar]
- 31.Ratanatharathorn V, Logan B, Wang D, et al. Prior rituximab correlates with less acute graft-versus-host disease and better survival in B-cell lymphoma patients who received allogeneic peripheral blood stem cell transplantation. Br J Haematol. 2009;145:816–24. doi: 10.1111/j.1365-2141.2009.07674.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Styczynski J, Gil L, Tridello G, et al. Response to rituximab-based therapy and risk factor analysis in Epstein Barr Virus-related lymphoproliferative disorder after hematopoietic stem cell transplant in children and adults: A study from the Infectious Diseases Working Party of the European Group for Blood and Marrow Transplantation. Clin Infect Dis. 2013;57:794–802. doi: 10.1093/cid/cit391. [DOI] [PubMed] [Google Scholar]
- 33.Ghobrial IM, Habermann TM, Ristow KM, et al. Prognostic factors in patients with post-transplant lymphoproliferative disorders (PTLD) in the rituximab era. Leuk Lymphoma. 2005;46:191–96. doi: 10.1080/10428190400012011. [DOI] [PubMed] [Google Scholar]
- 34.Gupta S, Fricker FJ, Gonzalez-Peralta RP, et al. Post-transplant lymphoproliferative disorder in children: Recent outcomes and response to dual rituximab/low-dose chemotherapy combination. Pediatr Transplant. 2010;14:896–902. doi: 10.1111/j.1399-3046.2010.01370.x. [DOI] [PubMed] [Google Scholar]
- 35.Hanel M, Fiedler F, Thorns C. Anti-CD20 monoclonal antibody (rituximab) and cidofovir as successful treatment of an EBV-associated lymphoma with CNS involvement. Onkologie. 2001;24:491–94. doi: 10.1159/000055132. [DOI] [PubMed] [Google Scholar]
- 36.Czyzewski K, Styczynski J, Krenska A, et al. Intrathecal therapy with rituximab in central nervous system involvement of post-transplant lymphoproliferative disorder. Leuk Lymphoma. 2013;54:503–6. doi: 10.3109/10428194.2012.718342. [DOI] [PubMed] [Google Scholar]
- 37.Ghobrial IM, Habermann TM, Maurer MJ, et al. Prognostic analysis for survival in adult solid organ transplant recipients with post-transplantation lymphoproliferative disorders. J Clin Oncol. 2005;23:7574–82. doi: 10.1200/JCO.2005.01.0934. [DOI] [PubMed] [Google Scholar]
- 38.Choquet S, Mamzer BM, Hermine O, et al. Identification of prognostic factors in post-transplant lymphoproliferative disorders. Recent Results Cancer Res. 2002;159:67–80. doi: 10.1007/978-3-642-56352-2_9. [DOI] [PubMed] [Google Scholar]
- 39.Trappe RU, Dierickx D, Zimmermann H, et al. Response to rituximab induction is a predictive marker in B-cell post-transplant lymphoproliferative disorder and allows successful stratification into rituximab or R-CHOP consolidation in an international, prospective, multicenter phase II trial. J Clin Oncol. 2017;35:536–43. doi: 10.1200/JCO.2016.69.3564. [DOI] [PubMed] [Google Scholar]
- 40.Heslop HE, Slobod KS, Pule MA, et al. Long-term outcome of EBV-specific T-cell infusions to prevent or treat EBV-related lymphoproliferative disease in transplant recipients. Blood. 2010;115:925–35. doi: 10.1182/blood-2009-08-239186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Carpenter B, Haque T, Dimopoulou M, et al. Incidence and dynamics of Epstein-Barr virus reactivation after alemtuzumab-based conditioning for allogeneic hematopoietic stem-cell transplantation. Transplantation. 2010;90:564–70. doi: 10.1097/TP.0b013e3181e7a3bf. [DOI] [PubMed] [Google Scholar]
- 42.Worth A, Conyers R, Cohen J, et al. Pre-emptive rituximab based on viraemia and T cell reconstitution: A highly effective strategy for the prevention of Epstein-Barr virus-associated lymphoproliferative disease following stem cell transplantation. Br J Haematol. 2011;155:377–85. doi: 10.1111/j.1365-2141.2011.08855.x. [DOI] [PubMed] [Google Scholar]