Abstract
The dense stroma in pancreatic cancer tumours is rich in secreted extracellular matrix proteins and proteoglycans. Secreted hyaluronan, osteopontin and type IV collagen sustain oncogenic signalling by interactions with CD44s and its variant isoform CD44v6 on cancer cell membranes. Although well established in animal and in vitro models, this oncogenic CD44‐stromal ligand network is less explored in human cancer. Here, we use a pancreatic cancer tissue microarray from 69 primary tumours and 37 metastatic lymph nodes and demonstrate that high tumour cell expression of CD44s and, surprisingly, low stromal deposition of osteopontin correlate with poor survival independent of established prognostic factors for pancreatic cancer. High stromal expression of hyaluronan was a universal trait of both primary tumours and metastatic lymph nodes. However, hyaluronan species of different molecular mass are known to function differently in pancreatic cancer biology and immunohistochemistry cannot distinguish between them. Using gas‐phase electrophoretic molecular mobility analysis, we uncover a shift towards high molecular mass hyaluronan in pancreatic cancer tissue compared to normal pancreas and at a transcriptional level, we find that hyaluronan synthesising HAS2 correlates positively with CD44. The resulting prediction that high molecular mass hyaluronan would then correlate with poor survival in pancreatic cancer was confirmed in serum samples, where we demonstrate that hyaluronan >27 kDa measured before surgery is an independent predictor of postoperative survival. Our findings confirm the prognostic value of CD44 tissue expression and highlight osteopontin tissue expression and serum high molecular mass hyaluronan as novel prognostic markers in pancreatic cancer.
Keywords: pancreatic cancer, CD44, hyaluronan, type IV collagen, osteopontin, biomarkers
Introduction
Pancreatic ductal adenocarcinoma (pancreatic cancer) is generally diagnosed at a late metastatic stage and patients often die just months after their first symptoms appear 1, 2. Even patients diagnosed at an early stage have a median survival of less than 2 years despite surgery and chemotherapy with curative intent 3, 4. The tumours are characterised by an abundant desmoplastic stroma rich in extracellular matrix components 5. These include fibrillar collagens (type I and type III collagen) and hyaluronan but also basement membrane proteins including type IV collagen and matricellular proteins such as osteopontin 5, 6, 7. These secreted extracellular matrix components promote proliferation, migration and survival through cell adhesion receptors 6, 8.
The cell adhesion receptor CD44 is widely expressed on both normal and malignant cells. Upon ligand binding, CD44 recruits receptor tyrosine kinases, which in turn promotes pancreatic cancer cell survival and migration in vitro and in mouse studies 8, 9, 10. In human pancreatic cancer, high expression of CD44s and the variant isoform 6 (CD44v6) have been associated with poor survival 9, 11, 12, 13, 14.
Hyaluronan promotes cancer cell survival and migration in various cancer types through CD44 interactions 15. Synthesis and degradation produce hyaluronan species of different sizes. Low molecular mass hyaluronan promotes pancreatic cancer cell migration 16, while high molecular mass hyaluronan acts as an osmotic space occupant causing blood vessel compression and reduced chemotherapy delivery in pancreatic cancer mouse models 17, 18. In pancreatic cancer patients, high tumour expression of hyaluronan has been associated with poor survival 19, 20. Other stromal CD44 ligands include type IV collagen 21 and osteopontin 22. Both type IV collagen and osteopontin promote pancreatic cancer cell invasion in vitro 23, 24 and are increased in the circulation in pancreatic cancer patients 25.
In this study, we hypothesise that the CD44 receptor and its stromal ligands constitute a tumour‐promoting network in pancreatic cancer and aim to find prognostic markers within this context.
Materials and methods
Ethics statement
All subjects provided written informed consent. The study was conducted in accordance with the Helsinki Declaration of 1975 and was approved by the regional research ethics board of northern Sweden (Dnr. 09‐175M/2009‐1378‐31).
Tissue samples
A previously described tissue microarray was used for the tissue studies 26. The tissue microarray has core areas of 1 mm in diameter selected by an experienced pathologist (WW). It includes three cores from each primary tumour (n = 69) and one to three cores from metastatic lymph nodes (n = 32) (Table 1). Due to sample loss during sectioning, the number of analysed tumour cores varied between 65 and 68 for primary tumours and between 24 and 32 for metastatic lymph nodes. Normal pancreatic tissue was collected from patients undergoing pancreatic surgery for non‐malignant conditions (n = 4). The staining analysis was performed under blinded conditions.
Table 1.
Clinical characteristics of the tissue microarray cohort and the cases and controls included in serum hyaluronan measurements
| Variable (n, %) | Tissue cases (n = 69) | Serum cases (n = 44) | Serum controls (n = 22) |
|---|---|---|---|
| Gender | |||
| Male | 33 (47.8%) | 24 (54.5%) | 10 (45.5%) |
| Female | 36 (52.2%) | 20 (45.5%) | 12 (55.5%) |
| Age (years) | |||
| Mean (95% CI) | 64.4 (62.4–66.5) | 66.9 (64.5–69.2) | 60.8 (57.1–64.5) |
| Tumour grade | |||
| Grade 1 | 17 (24.6%) | 9 (20.5%) | |
| Grade 2 | 42 (60.9%) | 18 (40.9%) | |
| Grade 3 | 8 (11.6%) | 14 (31.8%) | |
| NA | 2 (2.9%) | 3 (6.8%) | |
| Resection margin | |||
| R1–2 | 44 (63.8%) | 36 (81.8%) | |
| R0 | 20 (29.0%) | 8 (18.2%) | |
| NA | 5 (7.2%) | – | |
| TNM stage | |||
| Stage I | 16 (23.2%) | 12 (27.3%) | |
| Stage II | 37 (53.6%) | 29 (70.3%) | |
| Stage III | 4 (5.8%) | – | |
| Stage IV | 1 (1.5%) | – | |
| NA | 11 (16.0%) | 1 (2.3%) | |
| Neoadjuvant treatment | |||
| None | 59 (85.5%) | 44 (100%) | |
| Chemotherapy | 2 (2.9%) | – | |
| Radiotherapy | 1 (1.5%) | – | |
| Preop IP 5‐FU* | 7 (10.1%) | – | |
| Adjuvant treatment | |||
| Yes | 23 (33.4%) | 23 (52.3%) | |
| No | 39 (56.5%) | 11 (25.0%) | |
| NA | 7 (10.1%) | 10 (22.7%) | |
| Survival (months) | |||
| Median (range) | 12.4 (0.5–140.9) | 24.5 (0.5–79) | |
Intraperitoneal 5‐fluorouracil given as a single bolus the day before surgery
Immunohistochemical staining
5 μm tissue microarray sections were stained for CD44s, CD44v6, osteopontin and type IV collagen expression using an automated Ventana Benchmark staining machine (Ventana Medical Systems, Tucson, AZ, USA). The following antigen retrieval methods were applied: (1) citrate buffer at pH 6.0 for 8 min at 94 °C for CD44s, (2) EDTA buffer for 4 min at 100 °C for CD44v6 and (3) none for type IV collagen and osteopontin stainings. See supplementary material, Table S1 for more information on the primary antibodies and concentrations used. Hyaluronan was stained using a biotinylated hyaluronan‐binding protein, isolated from bovine nasal cartilage (Corgenix, Broomfield, CO, USA), according to a previously described protocol 27. Tissues serving as negative controls were incubated with hyaluronidase from Streptomyces hyalurolyticus (H‐1136; Sigma‐Aldrich, St. Louis, MO, USA) for 4 h at 37 °C to ensure assay specificity. In the final steps, all stains were developed with 3,3‐diaminobenzidine and counterstained with Mayer's haematoxylin. Human skin served as positive controls.
Scoring of tissue expression
CD44s and CD44v6: The percentages of positive cancer cells were measured using the cell counter tool in ImageJ software (version 2.0). The core with the highest percentage of positive cells represented the score for each individual patient.
Osteopontin, hyaluronan and type IV collagen: Stromal expression was scored semi‐quantitatively by two independent observers. Each observer scored individually and cores that were scored differently by the two observers were re‐scored by each observer. Persistent differences after the second round were discussed between the investigators and a final score was obtained by consensus. The mean score of the three primary tumour cores represented the score for each individual patient.
Osteopontin and hyaluronan: An intensity score (0 = negative, 1 = positive, 2 = strong staining) multiplied by a distribution score (0 = 0–10%, 1 = 10–50%, 2 = 50–90%, 3 > 90% positive) was used. Osteopontin scores were divided into high/low using a score of 2 as a cut‐off. Hyaluronan scores were divided into high/low using a score of 3 as a cut‐off.
Basement membrane expression of type IV collagen has been suggested to be disrupted or lost and accumulates in the desmoplastic stroma in pancreatic cancer tissue 28. Hence, type IV collagen expression was scored by two methods. In primary tumour and lymph nodes, basement membrane structures were scored as 0 = negative, 1 = positive but not fully circumferential around tumour clusters and 2 = positive and fully circumferential around tumour clusters. The stromal expression was scored as: 0 = negative, 1 = positive or 2 = strong.
Cut‐off values for all markers were determined by testing different cut‐off values in relation to survival in a subset of cases (n = 21). If an ‘optimal cut‐off’ was found that divided patients into groups with different survival rates, that cut‐off was used in the analysis of the entire cohort (n = 69). If no such optimal cut‐off was found, the cut‐off in primary tumours was determined as the score closest to the median score using the entire cohort and in lymph nodes, the cut‐off was determined as positive/negative.
mRNA data from The Cancer Genome Atlas
We extracted clinical data and mRNA expression data from all the available primary pancreatic cancer tissue samples in The Cancer Genome Atlas (n = 146), https://cancergenome.nih.gov/ (March 2017). mRNA expression data were generated by first aligning RNA‐Seq reads with a reference genome followed by normalisation using the fragments per kilobase of transcript per million mapped reads, the upper quartile method 29, 30. The data were log transformed. Two patients were excluded from all survival analyses (one with missing data on survival time and one with survival <1 month). Two patients were excluded from analyses involving hyaluronan synthase 1 and one patient was excluded from analysis of TP53 mutational status due to missing data. The cohort was divided into high versus low expression by quartiles of mRNA expression levels.
Serum samples
Serum samples were obtained from a biobank collecting tissue and blood samples from patients undergoing pancreatic surgery at Umeå University Hospital. We included patients with a histopathologically verified diagnosis of pancreatic ductal adenocarcinoma between August 2009 and July 2014. Postoperative follow‐up samples (≥28 days) were included when available. Patients with a history of malignant disease, systemic inflammatory disease or liver disease were excluded. Clinical characteristics and serum bilirubin levels were obtained from medical records. Patients undergoing surgery for non‐malignant conditions or those who had an endoscopic examination without pathological findings served as controls. None of the controls had a previous history of malignant disease.
Measurements of serum hyaluronan
Serum hyaluronan levels were measured with two hyaluronan‐binding protein (HABP)‐based ELISA‐like kits: (1) a competitive assay (HA‐ELISA K1200; Eschelon Biosciences, Salt Lake City, UT, USA) and (2) a sandwich assay (HA Test Kit; Corgenix, Broomfield, CO, USA), according to the manufacturers' instructions. The kits differ in that the sandwich assay is molecular mass restricted and unable to detect low molecular mass hyaluronan (<27 kDa) but is more accurate in detecting high molecular mass hyaluronan compared to the molecular mass promiscuous competitive assay 31. All samples were run in duplicate and coefficient of variation percentage (CV%) <10% was considered acceptable. Four samples were run on all plate assays and were used to normalise for inter‐plate variation. Absorbance was measured on a Thermo Multiskan Ascent (Thermo Fisher Scientific, Waltham, MA, USA) and plotted against the concentration of the standard curve using four‐parameter logistic regression.
Hyaluronan molecular mass profile analysis
Hyaluronan was isolated from normal pancreas tissue (n = 2) and cancer tissue (n = 2) using a previously described method 32 with slight modifications. Frozen tissue samples were sectioned (100 times 20 μm) in a cryostat followed by drying using a vacuum rotary evaporator. Before the first and after the last section used, 5 μm cryostat sections were H&E stained and analysed to confirm tissue morphology (see supplementary material, Figure S1). Then, proteins were digested with Proteinase K (Sigma–Aldrich) at 55 °C overnight. Next, nucleic acids were digested using benzonase nuclease (Sigma–Aldrich) for 5 h at 37 °C. Next, chondroitin was digested using Chondroitinase ABC (Sigma–Aldrich) for 10 min at 37 °C. At the end of each day, chloroform was added to each sample and the aqueous phase was dialysed against 0.1 m NaCl using Amicon Ultra 3K concentration units (Millipore, MA, USA) followed by precipitation overnight in 99% ethanol. Samples were loaded on anion exchange mini‐spin columns (Thermo Fisher Scientific) and centrifuged to wash out sulfated glycosaminoglycans and any remaining non‐hyaluronan contaminants based on NaCl binding. Finally, the sample was dialysed against 20 mm ammonium acetate (pH 8.0) in Amicon Ultra 3K concentration units to remove salt.
Hyaluronan molecular mass profiles were quantified using gas‐phase electrophoretic mobility molecular analysis (GEMMA) (TSI Corp., Shoreview, MN, USA) 33. Hyaluronan standards of 30 and 1000 kDa (Hyalose LLC, Oklahoma, OC, USA) were used as reference samples.
Statistics
Statistical analysis was performed using STATA 12.1 (StataCorp, College Station, TX, USA) and Prism 5 (GraphPad Software, La Jolla, CA, USA). Continuous variables were analysed with two‐tailed Student's t‐test (parametric data), Wilcoxon rank sum test (non‐parametric data), or Wilcoxon signed rank test (paired non‐parametric data). Categorical variables were compared using chi‐squared test or Fisher's exact test when appropriate. Correlations were analysed using Spearman's rank test or Pearson's correlations when appropriate. Survival analysis was performed with Kaplan–Meier curves and log‐rank and Wilcoxon–Breslow estimates. The Cox proportional hazard method was used for multivariable survival analysis. Patients still alive at analysis and patients who died within 1 month after surgery due to surgical complications were censored. Ordinal regression was performed to adjust for age in the serum‐hyaluronan results. p < 0.05 was used as a cut‐off for significance testing. The optimal cut‐off in receiver operating characteristics (ROC) analysis was calculated as the maximal value for sensitivity + specificity − 1. Combined ROC curves were constructed using the predicted probability from a logistic regression model.
Results
High CD44 expression is associated with poor survival
Normal pancreatic acinar cells stained strongly for CD44s in cell membranes and weakly for CD44v6 in the cytoplasm (see supplementary material, Figure S2A,B). Interestingly, while normal acinar and ductal cells stained for CD44v6 in the cytoplasm (see supplementary material, Figure S2B), cancer cells showed a marked membranous staining pattern (Figure 1B). Stromal cells stained positively for CD44s in cancerous tissue (Figure 1A).
Figure 1.
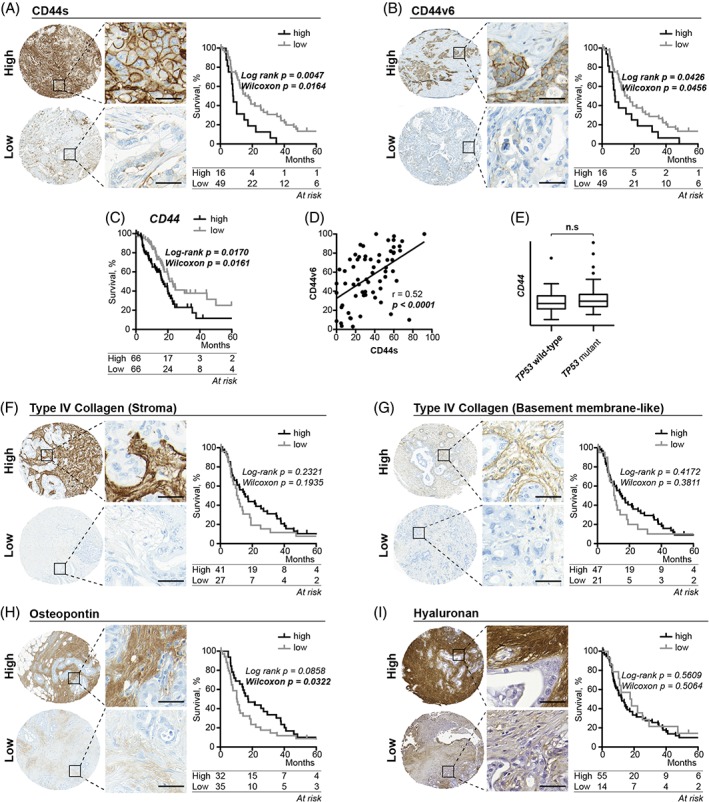
High versus low expression and associated Kaplan–Meier estimates for the primary tumour expression of (A,B) CD44 receptors, (F–G) type IV collagen, (H) osteopontin and (I) hyaluronan. Tissue microarray cores are 1 mm in diameter. Scale bar = 50 μm in highlighted areas. *Compared using Wilcoxon–Breslow estimates. (C) Kaplan–Meier survival estimates for patients with high versus low expression of CD44 mRNA transcripts. (D) Plot showing the correlation between CD44s and CD44v6 expression in primary tumours. (E) Box plots of CD44 mRNA transcripts in TP53 wild type and TP53 mutant primary tumours.
We divided the cohort into high/low expression groups for both CD44s and CD44v6 using the 75th percentile as a cut‐off (Figure 1A,B). High expression of both CD44 isoforms was associated with shorter median survival (8.1 versus 16.6 months for CD44s and 8.1 versus 14.9 months for CD44v6) (Figure 1A,B). Similarly, high CD44 mRNA expression in primary tumours was associated with shorter median survival (16.0 versus 21.1 months) (Figure 1C). We found a significant correlation between expression of the two CD44 isoforms (r = 0.52, p < 0.0010) (Figure 1D). We then compared CD44 mRNA levels in tumour suppressor TP53 mutant versus wild‐type tumours because previous reports have suggested that CD44 expression is negatively regulated by p53 34. However, we found no correlation between CD44 mRNA levels and TP53 status (Figure 1E).
Survival associations for stromal CD44 ligands
We then investigated survival associations with the stromal CD44 ligands type IV collagen, osteopontin and hyaluronan.
In normal pancreas tissue, type IV collagen stained positively in basement membranes around acini and in connective tissue around ducts (see supplementary material, Figure S2C). In tumours, we detected type IV collagen‐positive basement membranes in 69.1% of the cases (47/68) and type IV collagen‐positive stroma in 73.1% (49/68) of the cases (Figure 1F,G). Retained basement membrane expression correlated significantly with the stromal expression (see supplementary material, Figure S3A). However, we found no significant association between either of the type IV collagen expression patterns and patient survival (Figure 1F,G). The same was also true at the transcriptional level (see supplementary material, Figure S3B).
In normal pancreas tissue, osteopontin localised in ductal connective tissue and, to a varying degree, in connective tissue around acini (see supplementary material, Figure S2D). We detected stromal osteopontin in 93% of the tumours (62/67) but with large variations in intensity and staining distribution. We found a significant association between low osteopontin expression and shorter postoperative survival (median survival 10.3 versus 17.4 months) when using Wilcoxon–Breslow estimates but not when using Log‐Rank estimates (Figure 1H). This suggests a protective role of stromal osteopontin protein in pancreatic cancer tumours only in the initial years after surgery as Wilcoxon–Breslow estimates put more weight on early time points. Osteopontin mRNA expression was not associated with postoperative survival (see supplementary material, Figure S3C).
Hyaluronan stained sparsely in normal pancreatic ducts (see supplementary material, Figure S2E). By contrast, all tumours stained positively for hyaluronan in the stroma and the majority (53/67) stained strongly. Similarly, all metastatic lymph nodes stained positively and most (20/28) of them stained strongly. We found no association between the degree of stromal hyaluronan expression and patient survival (Figure 1I).
We detected no significant survival correlations to metastatic lymph node expression for any analysed markers, including the CD44 receptors (see supplementary material, Figure S4).
High molecular mass hyaluronan in PDAC tissue
As the molecular mass of hyaluronan has been reported to influence pancreatic cancer cell behaviour 16, we reasoned that hyaluronan mass profiles could be more informative than total hyaluronan amount. We therefore assessed the molecular mass profiles from two pancreatic cancer tissue samples and two normal pancreatic tissue samples. Interestingly, the two pancreatic cancer tissue samples were more abundant in high molecular mass hyaluronan compared to normal pancreatic tissue samples (Figure 2A).
Figure 2.
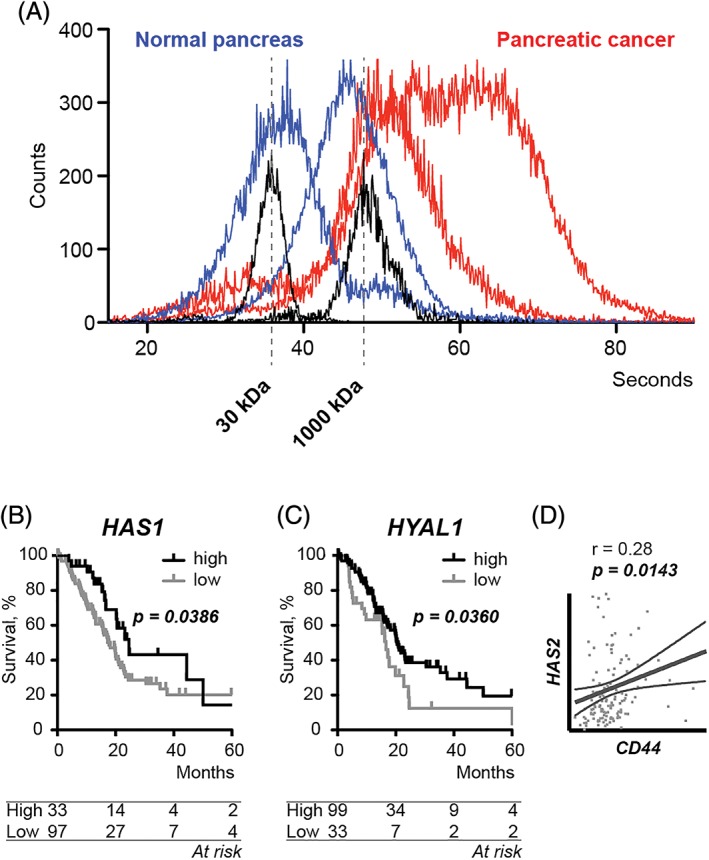
(A) Hyaluronan molecular mass profiles in two normal pancreas tissue samples (blue) and two pancreatic cancer tissue samples (red) detected by gas‐phase electrophoretic molecular mobility analysis. With increasing time (x‐axis), hyaluronan molecules of increasing molecular mass are counted (y‐axis). Hyaluronan standards (black) indicate 30 and 1000 kDa. (B,C) Kaplan–Meier survival estimates for high versus low mRNA levels of (B) hyaluronan synthase 1 (HAS1) and (C) hyaluronidase 1 (HYAL1). P = log‐rank test. (D) Correlation between CD44 and hyaluronan synthase 2 (HAS2) mRNA levels. r = Pearson's r. The thin lines indicate confidence intervals.
By investigating three hyaluronan‐synthesising proteins, hyaluronan synthases (HAS) 1–3 and two hyaluronan‐degrading enzymes, hyaluronidases (HYAL) 1 and 2, we found that high HAS1 mRNA levels (above the 75th percentile) and high HYAL1 levels (above the 25th percentile) were associated with longer survival (24.6 versus 17.7 months and p = 0.036 for HAS1 and 20.5 versus 16.6 months and p = 0.0386 for HYAL1) (Figure 2B,C). Interestingly, although mRNA levels of HAS 2 were not associated with survival, a positive correlation with CD44 expression was found (Figure 2D).
Pancreatic cancer patients have increased levels of serum hyaluronan
Next, we hypothesised that the high tissue expression of hyaluronan would be reflected in the circulation to serve as a non‐invasive biomarker. To test this, we compared serum hyaluronan levels in 44 pancreatic cancer patients (including 42 preoperative and 18 postoperative samples) and 22 healthy controls (Table 1) using two hyaluronan‐binding protein‐based ELISA assays. The assays differ in that the sandwich assay is restricted to detect high molecular mass hyaluronan (<27 kDa), while the competitive assay detects hyaluronan of any molecular mass (high molecular mass promiscuous) 31.
Independent of assay, preoperative serum hyaluronan levels were significantly higher in patients than in healthy controls (Figure 3A,B). The area under the curve (AUC) was 0.84 for both hyaluronan measurements and 0.91 for Ca 19‐9 (Figure 3C,D). The combination of hyaluronan and Ca 19‐9 measurements increased the AUC to 0.92 for the molecular mass promiscuous assay and 0.94 for the high mass restricted assay (Figure 3C,D). Postoperative serum hyaluronan levels were also significantly elevated compared to healthy controls but did not differ compared to preoperative levels (Figure 3A).
Figure 3.
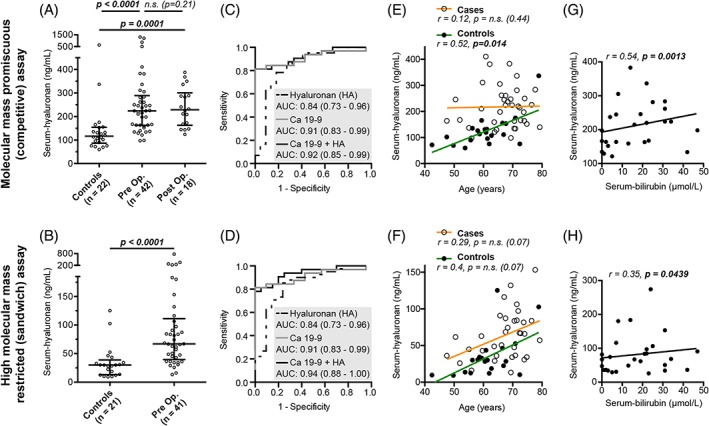
(A,B) Serum‐hyaluronan measurements in pancreatic cancer patients and healthy controls measured with (A) a molecular mass promiscuous (competitive) assay and (B) a high molecular mass restricted (sandwich) assay. (C,D) Receiver operating characteristics curves discriminating preoperative pancreatic cancer patients from healthy controls with serum‐hyaluronan, Ca 19‐9 and Hyaluronan + Ca 19‐9. AUC = area under the curve. 95% CI is within brackets. (E, F) Scatterplot showing the correlation between age and serum‐hyaluronan and linear regression lines for cases and controls measured with (E) the molecular mass promiscuous assay and (F) the high molecular mass restricted assay. Outliers (serum‐hyaluronan >500 ng/ml) are removed from the figure (not in the statistical analysis). The linear regression lines reflect that the effect of age on serum‐hyaluronan is different between cases and controls and intercept at ~80 years of age when measured with the molecular mass promiscuous assay (E) but not when measured with the high molecular mass restricted assay (F). (G,H) The correlation between serum‐hyaluronan and serum‐bilirubin. Outliers are removed from the figure (not in the statistical analysis).
The pancreas patients were significantly older than controls (p = 0.0047) (Table 1) and as serum hyaluronan levels increased with age in control samples (Figure 3E,F), we suspected that age might have confounded our results. However, after adjusting for age, hyaluronan remained significantly increased in patients in both assays (see supplementary material, Table S2). However, serum hyaluronan measured with the molecular mass promiscuous assay did not increase with age in patients and the regression lines for patients and controls intersected at 80 years of age when including an interaction term between age and group designation in the ordinal model (see supplementary material, Table S3). This was not the case for serum hyaluronan measured with the high molecular mass restricted assay. Similar relationships were evident when plotting the linear correlation between hyaluronan levels and age in the two assays (Figure 3E,F). In addition, hyaluronan levels correlated significantly with bilirubin in both assays (Figure 3G,H), suggesting that bile duct obstruction might also influence serum hyaluronan levels.
High mass serum hyaluronan, CD44 and OPN are independent predictors of PDAC survival
To adjust for clinical variables not accounted for in Kaplan–Meier analysis, we performed multivariable Cox regression including the following variables: CD44s protein expression, osteopontin protein expression, gender, age, tumour grade, TNM stage, R1 resection, neoadjuvant chemotherapy and adjuvant chemotherapy. In the multivariable model, high CD44s and low osteopontin independently predicted poor survival, as did poor differentiation, higher TNM stage, receiving preoperative intraperitoneal 5‐FU and not receiving adjuvant chemotherapy (Table 2).
Table 2.
Multivariable Cox regression model including the tissue expression of CD44s and OPN
| Variable | Hazard ratio (95% CI) | p |
|---|---|---|
| CD44s score (per 25% positive cells) | 1.9 (1.13–3.22) | 0.015 |
| Osteopontin score | 0.8 (0.62–0.99) | 0.046 |
| Male | 1.2 (0.63–2.42) | 0.548 |
| Age (per 10 years) | 0.9 (0.55–1.59) | 0.803 |
| Tumour grade | ||
| Grade 2 | 1.3 (0.61–2.92) | 0.465 |
| Grade 3 | 18.2 (3.91–84.6) | <0.0001 |
| Non‐radical resection | 1.1 (0.53–2.46) | 0.733 |
| Stage II–IV | 3.14 (1.27–7.79) | 0.013 |
| Neoadjuvant treatment | ||
| Preop IP 5‐FU | 4.4 (1.13–17.3) | 0.033 |
| Chemotherapy | 0.3 (0.10–3.03) | 0.488 |
| Adjuvant treatment | 0.3 (0.11–0.73) | 0.009 |
Significant P values are shown in bold underlined font.
We then performed a separate multivariable Cox regression model including high molecular mass serum hyaluronan, measured with the high molecular mass restricted assay. High preoperative levels of high molecular mass hyaluronan predicted poor postoperative survival independent of age, gender, grade, TNM stage, R1 resection and adjuvant treatment (Table 3). By contrast, neither preoperative nor postoperative serum‐hyaluronan levels measured with the molecular mass promiscuous assay were associated with survival (see supplementary material, Tables S4 and S5).
Table 3.
Cox regression models including preoperative S‐HA levels measured with the high molecular mass restricted (sandwich) assay
| Variable | Hazard ratio (95% CI) | p |
|---|---|---|
| Serum‐hyaluronan (per 100 ng/ml) | 1.3 (1.02–1.77) | 0.033 |
| Male | 1.2 (0.44–3.20) | 0.744 |
| Age (per 10 years) | 1.3 (0.64–2.70) | 0.452 |
| Tumour grade | ||
| Grade 2 | 6.8 (1.54–29.6) | 0.011 |
| Grade 3 | 21.0 (4.63–95.1) | <0.0001 |
| TNM stage II | 8.5 (2.51–28.7) | 0.001 |
| Non‐radical resection | 8.0 (1.96–32.2) | 0.004 |
| Adjuvant treatment | 0.1 (0.03–0.32) | <0.0001 |
Significant P values are shown in bold underlined font.
Discussion
The present study focuses on the CD44 receptor and its stromal ligands with the aim to find prognostic markers of postoperative survival in PDAC. We show that high expression of CD44s in pancreatic cancer cells and low stromal osteopontin expression independently predict poor postoperative survival, while hyaluronan and type IV collagen have limited potential as tissue‐based biomarkers. On the other hand, we show that hyaluronan is highly expressed in almost all pancreatic ductal adenocarcinomas and is significantly increased in the serum of pancreatic cancer patients. In addition, we uncover a shift towards a high molecular mass profile of hyaluronan in pancreatic cancer that can be detected in the circulation, where increased levels of high molecular mass hyaluronan predict poor postoperative survival.
The association between CD44s expression and survival is in line with several previous reports 9, 11, 12, 13, 14. We found that high expression of CD44, both at the protein and mRNA levels, is associated with worse patient outcome. This highlights CD44 as a potential therapeutic target in pancreatic cancer. However, while preclinical studies support this notion 10, 35, a phase I clinical trial evaluating an antibody interfering with the hyaluronan‐binding domain of CD44 had to be terminated due to lack of clinical response 36. This trial, however, included 65 patients with 20 different tumour forms and such a liberal inclusion criterion might have obscured clinical responses pertinent to certain cancer forms. In breast cancer, CD44 is negatively regulated by the tumour suppressor p53 34. No such correlation was, however, found in our material and hence, other pathways must be critical in CD44 regulation in pancreatic cancer.
We found that high osteopontin expression is an independent predictor of longer survival. Collins et al found similar results when comparing osteopontin‐positive versus negative pancreatic cancer, although they scored cancer cells instead of stromal expression 37. However, we found no association between osteopontin mRNA levels and survival, which is likely due to the high degree of post‐translational regulation of osteopontin 38.
We have previously shown that type IV collagen is expressed in pancreatic cancer tissue in basement membrane‐like structures close to cancer cells and that it stimulates cancer cell proliferation, migration and survival in vitro 23, 39. The lack of association with survival in the present study pinpoints a discrepancy between tumour‐residing and ‐circulating type IV collagen with regard to prognostic value. Hence, type IV collagen leakage rather than production appears to be a malignant trait of pancreatic cancer.
High hyaluronan expression in pancreatic cancer was associated with shorter postoperative survival in two previous studies 19, 20. However, the study by Cheng et al relied on an antibody‐based method to detect hyaluronan and as hyaluronan is a non‐immunogenic molecule 40, the specificity of antibody‐based staining is highly questionable 19. The other study used the ratio between hyaluronan staining and total tumour area to divide tumours into high versus low expression 20. That scoring method is inevitably affected by sample cellularity. Here, we used a HABP staining method, which specifically stains hyaluronan and scored the amount of positive stroma. In our hands, hyaluronan was strongly expressed in almost all primary tumours and metastases, with little variation and no correlation with any clinical parameter, including survival. This suggests that hyaluronan deposition is a universal trait of the desmoplastic pancreatic cancer stroma. Depletion of stromal hyaluronan is a promising therapeutic strategy in preclinical studies 18, 41. If stromal hyaluronan depletion is proven beneficial in human pancreatic cancer, our results suggest that such a therapy could aid a majority of patients owing to the ubiquitous expression of hyaluronan in the tumours. The strong expression also suggests that CD44‐hyaluronan signalling is limited by receptor expression rather than ligand availability.
In pancreatic cancer, high HAS2 and low HYAL1 expression has been associated with shorter survival 19. Similarly, we find that low HYAL1 and also low HAS1 mRNA expression in tissue correlate with shorter survival, suggestive of protective effects from a high HA turnover. But importantly, HAS and HYAL enzyme activities are extensively modulated post‐transcriptionally and such layers of hyaluronan biogenesis are beyond the scope of this manuscript. Nevertheless, our results suggest that the hyaluronan molecular mass profile in pancreatic cancer might be of greater importance than the total amount of hyaluronan expression. Indeed, we find that high molecular mass hyaluronan may be more abundant in pancreatic cancer tissue than in normal tissue, which calls for more extensive hyaluronan size profiling in pancreatic cancer.
We show that serum hyaluronan is significantly higher in pancreatic cancer patients compared to healthy controls. We also show that the AUC is higher when adding hyaluronan measurements to Ca 19‐9. Interestingly, while both assays demonstrated that serum hyaluronan is elevated in pancreatic cancer, our result suggests that hyaluronan measured with the molecular mass promiscuous assay cannot be adjusted for age in the elderly. Bile duct obstruction might confound serum hyaluronan as we found a correlation between serum hyaluronan and bilirubin. Sinusoidal endothelial cells in the liver degrade >90% of circulating hyaluronan 42 and bile duct obstruction is associated with impaired hepatic sinusoidal cell function 43. Importantly, elevated serum hyaluronan has been demonstrated in other cancer forms and in liver cirrhosis 44, 45 and hence, this finding is not specific for pancreatic cancer. Thus, its potential to discriminate disease and predict outcome must be evaluated in a larger cohort with the confounders presented here considered.
In summary, tissue expression of both CD44 receptors and stromal CD44 ligands confers prognostic information in pancreatic cancer. High cancer cell expression of CD44s and low stromal expression of osteopontin are independent markers of poor prognosis, underscoring CD44 as a potential treatment target and the need to revisit the role of osteopontin in pancreatic cancer progression and metastasis. The CD44 ligand hyaluronan is strongly expressed in the stroma in almost all pancreatic ductal adenocarcinomas and lymph node metastases and is elevated in serum from pancreatic cancer patients. In addition, high preoperative serum high molecular mass hyaluronan (>27 kDa) predicts poor postoperative survival independent of other, established survival predictors.
Author contributions statement
OF, OB, DÖ, UH and MS participated in planning the study and the study design. OF, DÖ and AB participated in immunohistochemistry scoring. OF, DÖ and WW participated in the planning and construction of the tissue microarray. OF, OB and CH participated in planning, extraction and analysing mRNA data. UH performed hyaluronan mass distribution analysis. OF and OB performed data analysis. OF, OB, DÖ, UH and MS wrote the manuscript. The final text was read and critically reviewed by all authors. All authors gave their final approval of the text.
Supporting information
Figure S1 Tissue sections before the first and after the last sections used in the hyaluronan molecular mass profile analysis
Figure S2 Protein expression in normal pancreatic tissue
Figure S3 Correlation and survival data for type IV collagen and osteopontin
Figure S4 Kaplan–Meier survival estimates comparing metastatic lymph node expression of CD44s, CD44v6, type IV collagen, osteopontin and hyaluronan
Table S1 Primary antibodies used for immunohistochemistry
Table S2 Ordinal regression to adjust serum‐hyaluronan (dependent variable) for age differences in patients and controls
Table S3 Ordinal regression to adjust serum‐hyaluronan (dependent variable) for age differences in patients and controls and the interaction term between age and patient/control designation
Table S4 Cox regression models including preoperative serum‐hyaluronan levels measured with the molecular mass promiscuous (competitive) assay
Table S5 Cox regression models including postoperative S‐HA levels measured with the molecular mass promiscuous (competitive) assay
Acknowledgements
We thank Christina Lundin for help with the serum measurements of hyaluronan and Ca 19‐9 as well as Susanne Gidlund for help with the tissue microarray construction. This study was funded by The Swedish Research Council (2011‐3089 for MS, 2017‐01531 for DÖ), the Swedish Cancer society (110679 and 120135 for MS, CAN 2017/332 and CAN 2017/827 for DÖ), the Swedish Society of Medicine (SLS‐591551 and SLS‐691681for DÖ), the County Council of Västerbotten (MS and VLL‐643451 for DÖ), the Swedish Society of Medicine (SLS‐691681 for DÖ), the Knut and Alice Wallenberg foundation (for DÖ), The Swedish Foundation for International Cooperation in Research and Higher Education (PT2015‐6432 for DÖ), Cancer Research Foundation in Northern Sweden (AMP15‐793 and AMP17‐877 for DÖ), JC Kempe Memorial Foundation (OF and DÖ) and grants from the medical faculty at Umeå University (223‐1828‐13 OF)
No conflicts of interest were declared.
References
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin 2016; 66: 7–30. [DOI] [PubMed] [Google Scholar]
- 2. Bilimoria KY, Bentrem DJ, Ko CY, et al Validation of the 6th edition AJCC Pancreatic Cancer Staging System: report from the National Cancer Database. Cancer 2007; 110: 738–744. [DOI] [PubMed] [Google Scholar]
- 3. Neoptolemos JP, Stocken DD, Bassi C, et al Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: a randomized controlled trial. JAMA 2010; 304: 1073–1081. [DOI] [PubMed] [Google Scholar]
- 4. Oettle H, Neuhaus P, Hochhaus A, et al Adjuvant chemotherapy with gemcitabine and long‐term outcomes among patients with resected pancreatic cancer: the CONKO‐001 randomized trial. JAMA 2013; 310: 1473–1481. [DOI] [PubMed] [Google Scholar]
- 5. Feig C, Gopinathan A, Neesse A, et al The pancreas cancer microenvironment. Clin Cancer Res 2012; 18: 4266–4276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Grzesiak JJ, Bouvet M. Determination of the ligand‐binding specificities of the alpha2beta1 and alpha1beta1 integrins in a novel 3‐dimensional in vitro model of pancreatic cancer. Pancreas 2007; 34: 220–228. [DOI] [PubMed] [Google Scholar]
- 7. Wong GS, Rustgi AK. Matricellular proteins: priming the tumour microenvironment for cancer development and metastasis. Br J Cancer 2013; 108: 755–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Jung T, Gross W, Zoller M. CD44v6 coordinates tumor matrix‐triggered motility and apoptosis resistance. J Biol Chem 2011; 286: 15862–15874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lee JJ, Perera RM, Wang H, et al Stromal response to Hedgehog signaling restrains pancreatic cancer progression. Proc Natl Acad Sci U S A 2014; 111: E3091–E3100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Molejon MI, Tellechea JI, Loncle C, et al Deciphering the cellular source of tumor relapse identifies CD44 as a major therapeutic target in pancreatic adenocarcinoma. Oncotarget 2015; 6: 7408–7423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Pothula SP, Xu Z, Goldstein D, et al Hepatocyte growth factor inhibition: a novel therapeutic approach in pancreatic cancer. Br J Cancer 2016; 114: 269–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Habbe N, Koorstra JB, Mendell JT, et al MicroRNA miR‐155 is a biomarker of early pancreatic neoplasia. Cancer Biol Ther 2009; 8: 340–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Xiaoping L, Xiaowei Z, Leizhen Z, et al Expression and significance of CD44 and p‐AKT in pancreatic head cancer. World J Surg Oncol 2015; 13: 334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Abue M, Yokoyama M, Shibuya R, et al Circulating miR‐483‐3p and miR‐21 is highly expressed in plasma of pancreatic cancer. Int J Oncol 2015; 46: 539–547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Toole BP, Slomiany MG. Hyaluronan: a constitutive regulator of chemoresistance and malignancy in cancer cells. Semin Cancer Biol 2008; 18: 244–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cheng XB, Kohi S, Koga A, et al Hyaluronan stimulates pancreatic cancer cell motility. Oncotarget 2016; 7: 4829–4840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Jacobetz MA, Chan DS, Neesse A, et al Hyaluronan impairs vascular function and drug delivery in a mouse model of pancreatic cancer. Gut 2012; 62: 112–120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Provenzano PP, Cuevas C, Chang AE, et al Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 2012; 21: 418–429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cheng XB, Sato N, Kohi S, et al Prognostic impact of hyaluronan and its regulators in pancreatic ductal adenocarcinoma. PLoS One 2013; 8: e80765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Whatcott CJ, Diep CH, Jiang P, et al Desmoplasia in primary tumors and metastatic lesions of pancreatic cancer. Clin Cancer Res 2015; 21: 3561–3568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ishii S, Ford R, Thomas P, et al CD44 participates in the adhesion of human colorectal carcinoma cells to laminin and type IV collagen. Surg Oncol 1993; 2: 255–264. [DOI] [PubMed] [Google Scholar]
- 22. Ahmed M, Sottnik JL, Dancik GM, et al An osteopontin/CD44 axis in RhoGDI2‐mediated metastasis suppression. Cancer Cell 2016; 30: 432–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Ohlund D, Franklin O, Lundberg E, et al Type IV collagen stimulates pancreatic cancer cell proliferation, migration, and inhibits apoptosis through an autocrine loop. BMC Cancer 2013; 13: 154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kolb A, Kleeff J, Guweidhi A, et al Osteopontin influences the invasiveness of pancreatic cancer cells and is increased in neoplastic and inflammatory conditions. Cancer Biol Ther 2005; 4: 740–746. [DOI] [PubMed] [Google Scholar]
- 25. Franklin O, Ohlund D, Lundin C, et al Combining conventional and stroma‐derived tumour markers in pancreatic ductal adenocarcinoma. Cancer Biomark 2015; 15: 1–10. [DOI] [PubMed] [Google Scholar]
- 26. Morin E, Sjoberg E, Tjomsland V, et al VEGF receptor‐2/neuropilin1 trans‐complex formation between endothelial and tumor cells is an independent predictor of pancreatic cancer survival. J Pathol 2018; 246: 311–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Hellstrom M, Johansson B, Engstrom‐Laurent A. Hyaluronan and its receptor CD44 in the heart of newborn and adult rats. Anat Rec A Discov Mol Cell Evol Biol 2006; 288: 587–592. [DOI] [PubMed] [Google Scholar]
- 28. Linder S, Castanos‐Velez E, von Rosen A, et al Immunohistochemical expression of extracellular matrix proteins and adhesion molecules in pancreatic carcinoma. Hepatogastroenterology 2001; 48: 1321–1327. [PubMed] [Google Scholar]
- 29. Bullard JH, Purdom E, Hansen KD, et al Evaluation of statistical methods for normalization and differential expression in mRNA‐Seq experiments. BMC Bioinformatics 2010; 11: 94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Mortazavi A, Williams BA, McCue K, et al Mapping and quantifying mammalian transcriptomes by RNA‐Seq. Nat Methods 2008; 5: 621–628. [DOI] [PubMed] [Google Scholar]
- 31. Haserodt S, Aytekin M, Dweik RA. A comparison of the sensitivity, specificity, and molecular weight accuracy of three different commercially available Hyaluronan ELISA‐like assays. Glycobiology 2011; 21: 175–183. [DOI] [PubMed] [Google Scholar]
- 32. Tolg C, Hamilton SR, Zalinska E, et al A RHAMM mimetic peptide blocks hyaluronan signaling and reduces inflammation and fibrogenesis in excisional skin wounds. Am J Pathol 2012; 181: 1250–1270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Malm L, Hellman U, Larsson G. Size determination of hyaluronan using a gas‐phase electrophoretic mobility molecular analysis. Glycobiology 2012; 22: 7–11. [DOI] [PubMed] [Google Scholar]
- 34. Godar S, Ince TA, Bell GW, et al Growth‐inhibitory and tumor‐ suppressive functions of p53 depend on its repression of CD44 expression. Cell 2008; 134: 62–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Li L, Hao X, Qin J, et al Antibody against CD44s inhibits pancreatic tumor initiation and postradiation recurrence in mice. Gastroenterology 2014; 146: 1108–1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. der Houven van Oordt CW, Gomez‐Roca C, Herpen CV, et al First‐in‐human phase I clinical trial of RG7356, an anti‐CD44 humanized antibody, in patients with advanced, CD44‐expressing solid tumors. Oncotarget 2016; 7: 80046–80058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Collins AL, Rock J, Malhotra L, et al Osteopontin expression is associated with improved survival in patients with pancreatic adenocarcinoma. Ann Surg Oncol 2012; 19: 2673–2678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Shevde LA, Samant RS. Role of osteopontin in the pathophysiology of cancer. Matrix Biol 2014; 37: 131–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ohlund D, Lundin C, Ardnor B, et al Type IV collagen is a tumour stroma‐derived biomarker for pancreas cancer. Br J Cancer 2009; 101: 91–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. de la Motte CA, Drazba JA. Viewing hyaluronan: imaging contributes to imagining new roles for this amazing matrix polymer. J Histochem Cytochem 2011; 59: 252–257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Chauhan VP, Martin JD, Liu H, et al Angiotensin inhibition enhances drug delivery and potentiates chemotherapy by decompressing tumour blood vessels. Nat Commun 2013; 4: 2516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Fraser JR, Alcorn D, Laurent TC, et al Uptake of circulating hyaluronic acid by the rat liver. Cellular localization in situ. Cell Tissue Res 1985; 242: 505–510. [DOI] [PubMed] [Google Scholar]
- 43. Yoshidome H, Miyazaki M, Shimizu H, et al Obstructive jaundice impairs hepatic sinusoidal endothelial cell function and renders liver susceptible to hepatic ischemia/reperfusion. J Hepatol 2000; 33: 59–67. [DOI] [PubMed] [Google Scholar]
- 44. Gudowska M, Gruszewska E, Panasiuk A, et al Hyaluronic acid concentration in liver diseases. Clin Exp Med 2016; 16: 523–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Wu M, Cao M, He Y, et al A novel role of low molecular weight hyaluronan in breast cancer metastasis. FASEB J 2015; 29: 1290–1298. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1 Tissue sections before the first and after the last sections used in the hyaluronan molecular mass profile analysis
Figure S2 Protein expression in normal pancreatic tissue
Figure S3 Correlation and survival data for type IV collagen and osteopontin
Figure S4 Kaplan–Meier survival estimates comparing metastatic lymph node expression of CD44s, CD44v6, type IV collagen, osteopontin and hyaluronan
Table S1 Primary antibodies used for immunohistochemistry
Table S2 Ordinal regression to adjust serum‐hyaluronan (dependent variable) for age differences in patients and controls
Table S3 Ordinal regression to adjust serum‐hyaluronan (dependent variable) for age differences in patients and controls and the interaction term between age and patient/control designation
Table S4 Cox regression models including preoperative serum‐hyaluronan levels measured with the molecular mass promiscuous (competitive) assay
Table S5 Cox regression models including postoperative S‐HA levels measured with the molecular mass promiscuous (competitive) assay


