Abstract
Background
Down's syndrome occurs when a person has three copies of chromosome 21 (or the specific area of chromosome 21 implicated in causing Down's syndrome) rather than two. It is the commonest congenital cause of mental disability. Non‐invasive screening based on biochemical analysis of maternal serum or urine, or fetal ultrasound measurements, allows estimates of the risk of a pregnancy being affected and provides information to guide decisions about definitive testing.
Before agreeing to screening tests, parents need to be fully informed about the risks, benefits and possible consequences of such a test. This includes subsequent choices for further tests they may face, and the implications of both false positive (i.e. invasive diagnostic testing, and the possibility that a miscarried fetus may be chromosomally normal) and false negative screening tests (i.e. a fetus with Down’s syndrome will be missed). The decisions that may be faced by expectant parents inevitably engender a high level of anxiety at all stages of the screening process, and the outcomes of screening can be associated with considerable physical and psychological morbidity. No screening test can predict the severity of problems a person with Down's syndrome will have.
Objectives
To estimate and compare the accuracy of first and second trimester serum markers with and without first trimester ultrasound markers for the detection of Down’s syndrome in the antenatal period, as combinations of markers.
Search methods
We conducted a sensitive and comprehensive literature search of MEDLINE (1980 to 25 August 2011), Embase (1980 to 25 August 2011), BIOSIS via EDINA (1985 to 25 August 2011), CINAHL via OVID (1982 to 25 August 2011), the Database of Abstracts of Reviews of Effectiveness (the Cochrane Library 25 August 2011), MEDION (25 August 2011), the Database of Systematic Reviews and Meta‐Analyses in Laboratory Medicine (25 August 2011), the National Research Register (Archived 2007), and Health Services Research Projects in Progress database (25 August 2011). We did not apply a diagnostic test search filter. We did forward citation searching in ISI citation indices, Google Scholar and PubMed ‘related articles’. We also searched reference lists of retrieved articles
Selection criteria
Studies evaluating tests of combining first and second trimester maternal serum markers in women up to 24 weeks of gestation for Down's syndrome, with or without first trimester ultrasound markers, compared with a reference standard, either chromosomal verification or macroscopic postnatal inspection.
Data collection and analysis
Data were extracted as test positive/test negative results for Down's and non‐Down's pregnancies allowing estimation of detection rates (sensitivity) and false positive rates (1‐specificity). We performed quality assessment according to QUADAS criteria. We used hierarchical summary ROC meta‐analytical methods to analyse test performance and compare test accuracy. Analysis of studies allowing direct comparison between tests was undertaken. We investigated the impact of maternal age on test performance in subgroup analyses.
Main results
Twenty‐two studies (reported in 25 publications) involving 228,615 pregnancies (including 1067 with Down's syndrome) were included. Studies were generally high quality, although differential verification was common with invasive testing of only high risk pregnancies. Ten studies made direct comparisons between tests. Thirty‐two different test combinations were evaluated formed from combinations of eight different tests and maternal age; first trimester nuchal translucency (NT) and the serum markers AFP, uE3, total hCG, free βhCG, Inhibin A, PAPP‐A and ADAM 12. We looked at tests combining first and second trimester markers with or without ultrasound as complete tests, and we also examined stepwise and contingent strategies.
Meta‐analysis of the six most frequently evaluated test combinations showed that a test strategy involving maternal age and a combination of first trimester NT and PAPP‐A, and second trimester total hCG, uE3, AFP and Inhibin A significantly outperformed other test combinations that involved only one serum marker or NT in the first trimester, detecting about nine out of every 10 Down's syndrome pregnancies at a 5% false positive rate. However, the evidence was limited in terms of the number of studies evaluating this strategy, and we therefore cannot recommend one single screening strategy.
Authors' conclusions
Tests involving first trimester ultrasound with first and second trimester serum markers in combination with maternal age are significantly better than those without ultrasound, or those evaluating first trimester ultrasound in combination with second trimester serum markers, without first trimester serum markers. We cannot make recommendations about a specific strategy on the basis of the small number of studies available.
Keywords: Female; Humans; Pregnancy; Nuchal Translucency Measurement; Biomarkers; Biomarkers/blood; Chorionic Gonadotropin; Chorionic Gonadotropin/blood; Down Syndrome; Down Syndrome/blood; Down Syndrome/diagnosis; Down Syndrome/diagnostic imaging; Estriol; Estriol/blood; False Positive Reactions; Inhibins; Inhibins/blood; Maternal Age; Pregnancy Trimester, First; Pregnancy Trimester, First/blood; Pregnancy Trimester, Second; Pregnancy Trimester, Second/blood; Pregnancy‐Associated Plasma Protein‐A; Pregnancy‐Associated Plasma Protein‐A/analysis; Sensitivity and Specificity; alpha‐Fetoproteins; alpha‐Fetoproteins/analysis
Plain language summary
Screening tests for Down’s syndrome in the first 24 weeks of pregnancy
Background Down's syndrome (also known as Down's or Trisomy 21) is an incurable genetic disorder that causes significant physical and mental health problems, and disabilities. However, there is wide variation in how Down's affects people. Some individuals are severely affected whilst others have mild problems and are able to lead relatively normal lives. There is no way of predicting how badly a baby might be affected. Expectant parents are given the choice to be tested for Down’s syndrome during pregnancy to assist them in making decisions. If a mother is carrying a baby with Down’s syndrome, then there is the decision about whether to terminate or continue with the pregnancy. The information offers parents the opportunity to plan for life with a child with Down’s syndrome. The most accurate tests for Down’s syndrome involve testing fluid from around the baby (amniocentesis) or tissue from the placenta (chorionic villus sampling (CVS)) for the abnormal chromosomes associated with Down’s syndrome. Both these tests involve inserting needles through the mother's abdomen and are known to increase the risk of miscarriage. Thus, the tests may not be suitable for all pregnant women. Rather, tests that measure markers in the mother’s blood, urine, or on ultrasound scans of the baby are used for screening. These screening tests are not perfect as they can miss cases of Down’s syndrome and also give high risk test results to a number of women whose babies are not affected by Down’s syndrome. Thus, pregnancies identified as high risk using these screening tests require further testing using amniocentesis or CVS to confirm a diagnosis of Down’s syndrome.
What we did We assessed combinations of first trimester (up to 14 weeks' gestation) and second trimester serum screening tests (up to 24 weeks' gestation), with or without first trimester ultrasound screening tests, Our aim was to identify the most accurate test(s) for predicting the risk of a pregnancy being affected by Down's syndrome. We looked at one ultrasound marker (nuchal translucency) and seven different serum markers (PAPP‐A, total hCG, free βhCG, uE3, AFP, inhibin A, ADAM 12) that can be used alone, in ratios or in combination, taken before 24 weeks' gestation, thus creating 32 screening tests for Down’s. We found 22 studies, involving 228,615 pregnancies (including 1067 fetuses affected by Down's syndrome).
What we found For Down's syndrome screening, where tests were carried out in the first and second trimester and combined to give an overall risk, we found that a test comprised of first trimester nuchal translucency and PAPP‐A, and second trimester total hCG, uE3, AFP and Inhibin A was the most sensitive test, detecting nine out of 10 pregnancies affected by Down's syndrome. Five per cent of pregnant women receiving a high risk test result based on this combination would not be affected by Down's syndrome. There were relatively few studies assessing these tests and therefore we cannot make a strong recommendation about the best test. Other important information to consider The ultrasound tests themselves have no adverse effects for the woman, and blood tests can cause discomfort, bruising and, rarely, infection. However, some women who have a high risk screening test result, and are given amniocentesis or CVS have a risk of miscarrying a baby unaffected by Down’s. Parents will need to weigh up this risk when deciding whether or not to have an amniocentesis or CVS following a high risk screening test result.
Summary of findings
Summary of findings 1. Performance of the six most evaluated first and second trimester serum test strategies with or without ultrasound.
| Test strategy (with maternal age) | Studies | Women (cases) | Sensitivity (95% CI) at a 5% FPR | Test* |
| First trimester PAPP‐A and second trimester total hCG, uE3 and AFP | 4 | 2474 (236) | 85 (78, 89) | P = 0.014 |
| First trimester PAPP‐A and second trimester total hCG, uE3, AFP and inhibin A | 3 | 35,361 (217) | 87 (81, 91) | |
| First trimester NT and second trimester total hCG and AFP | 4 | 22,793 (135) | 85 (77, 91) | |
| First trimester NT and second trimester total hCG, uE3 and AFP | 4 | 13,708 (136) | 86 (78, 92) | |
| First trimester NT and PAPP‐A, and second trimester total hCG, uE3, AFP and inhibin A | 3 | 39,670 (184) | 95 (90, 97) | |
| First trimester NT and PAPP‐A, and second trimester free ßhCG, uE3, AFP and inhibin A | 4 | 40,348 (266) | 92 (88, 95) |
*Likelihood ratio test for the difference in accuracy between the six test strategies compared in a single meta‐analytic model
AFP = alpha‐fetoprotein; ßhCG = beta human chorionic gonadotrophin; FPR = false positive rate;hCG = human chorionic gonadotrophin; NT = nuchal translucency; PAPP‐A = pregnancy‐associated plasma protein‐A; uE3 = unconjugated oestriol
CI = confidence interval
Summary of findings 2. Performance of other first and second trimester serum strategies with or without ultrasound.
| Test | Studies | Women (cases) | Sensitivity* (95% CI) | Specificity* (95% CI) | Threshold |
| Without maternal age and ultrasound | |||||
| Single tests | |||||
| ADAM 12 second trimester to first trimester ratio | 1 | 579 (17) | 53 (28, 77) | 95 (93, 97) | 5% FPR |
| With maternal age and without ultrasound | |||||
| Triple tests | |||||
| First trimester PAPP‐A and second trimester total hCG and AFP | 1 | 1188 (98) | 83 (74, 90) | 95 (93, 96) | 5% FPR |
| First trimester PAPP‐A and second trimester free ßhCG and AFP | 2 | 2197 (94) | 83 to 85 | 94 to 95 | 5% FPR, 1:300 risk |
| Quadruple tests | |||||
| First trimester PAPP‐A and second trimester free ßhCG, uE3 and AFP | 1 | 1188 (98) | 86 (77, 92) | 95 (93, 96) | 5% FPR |
| Quintuple tests | |||||
| First trimester PAPP‐A and second trimester free ßhCG, uE3, AFP and inhibin A | 1 | 1188 (98) | 90 (82, 95) | 95 (93, 96) | 5% FPR |
| First trimester PAPP‐A and second trimester total hCG, uE3, AFP and PAPP‐A | 2 | 707 (121) | 78 (66, 86) | 98 (96, 99) | 1:200 risk |
| First trimester PAPP‐A and total hCG, and second trimester total hCG, uE3 and AFP | 2 | 707 (121) | 80 (68, 88) | 97 (94, 98) | 1:200 risk |
| First trimester PAPP‐A and uE3, and second trimester total hCG, uE3 and AFP | 2 | 707 (121) | 80 (68, 88) | 96 (93, 98) | 1:200 risk |
| Sextuple tests | |||||
| First trimester AFP, free ßhCG and uE3, and second trimester total hCG, uE3 and AFP | 1 | 12,339 (34) | 82 (65, 93) | 94 (93, 94) | 1:250 risk |
| First trimester PAPP‐A and second trimester total hCG, uE3, AFP, inhibin A and PAPP‐A | 1 | 540 (32) | 84 (67, 95) | 96 (94, 98) | 1:250 risk |
| Septuple tests | |||||
| First trimester PAPP‐A, total hCG and uE3, and second trimester total hCG, uE3, AFP and PAPP‐A | 2 | 707 (121) | 49 (36, 61) | 98 (96, 99) | 1:200 risk |
| With maternal age and ultrasound | |||||
| Triple tests | |||||
| First trimester NT and second trimester free ßhCG and AFP | 2 | 6616 (105) | 83 (70, 91) | 95 | 5% FPR |
| Quadruple tests | |||||
| First trimester NT and second trimester free ßhCG, uE3 and AFP | 1 | 1110 (85) | 88 (79, 94) | 95 (94, 96) | 5% FPR |
| First trimester NT and PAPP‐A, and second trimester total hCG and AFP | 1 | 1110 (85) | 91 (82, 96) | 95 (94, 96) | 5% FPR |
| First trimester NT and PAPP‐A, and second trimester free ßhCG and AFP | 2 | 3400 (93) | 88 to 91 | 95 to 98 | 5% FPR, 1:300 risk |
| Quintuple tests | |||||
| First trimester NT and second trimester total hCG, uE3, AFP and inhibin A | 1 | 1110 (85) | 91 (82, 96) | 95 (94, 96) | 5% FPR |
| First trimester NT and second trimester free ßhCG, uE3, AFP and inhibin A | 1 | 1110 (85) | 91 (82, 96) | 95 (94, 96) | 5% FPR |
| First trimester NT and PAPP‐A, and second trimester free ßhCG, uE3 and AFP | 1 | 1100 (85) | 92 (84, 97) | 95 (94, 96) | 5% FPR |
| First trimester NT and PAPP‐A, and second trimester total hCG, uE3 and AFP | 2 | 33,337 (171) | 88 to 92 | 95 to 97 | 5% FPR, 1:200 risk |
| Sextuple tests | |||||
| First trimester NT, PAPP‐A and free ßhCG, and second trimester total hCG, uE3 and AFP | 1 | 5060 (13) | 100 (75, 100) | 97 (96, 97) | 1:250 risk |
| Septuple tests | |||||
| First trimester NT, PAPP‐A and free ßhCG, and second trimester uE3, AFP, total hCG and inhibin A | 1 | 33,546 (87) | 94 (87, 98) | 89 (89, 89) | 1:150 risk |
| Contingent tests | |||||
| First trimester NT, PAPP‐A and free ßhCG, if risk 1:30‐1:1500, second trimester total hCG, uE3, AFP and inhibin A | 1 | 32,355 (86) | 91 (82, 96) | 95 (95, 96) | 1:270 risk |
| First trimester NT, PAPP‐A and free ßhCG, if risk 1:30‐1:1500, second trimester free ßhCG, uE3, AFP and inhibin A | 1 | 7842 (59) | 95 (86, 99) | 95 (94, 95) | 5% FPR |
| Stepwise tests | |||||
| First trimester NT and PAPP‐A, if risk < 1:100, second trimester free ßhCG, uE3 and AFP | 1 | 1507 (12) | 92 (62, 100) | 97 [(96, 98) | 1:250 risk |
| First trimester NT, PAPP‐A and free ßhCG, if risk < 1:30, second trimester total hCG, uE3, AFP and inhibin A | 1 | 32,355 (86) | 92 (84, 97) | 95 (95, 95) | 1:270 risk |
| First trimester NT, PAPP‐A and free ßhCG, if risk < 1:30, second trimester free ßhCG, uE3, AFP and 2T inhibin A | 1 | 7842 (59) | 97 (88, 100) | 95 (94, 95) | 5% FPR |
*Tests evaluated by at least one study are presented in the table. Where there were two studies at the same threshold, estimates of summary sensitivity and summary specificity were obtained by using univariate fixed‐effect logistic regression models to pool sensitivities and specificities separately. if the threshold used was a 5% FPR, then only the sensitivities were pooled. The range of sensitivities and specificities are presented where there were two studies and the thresholds used were different.
AFP = alpha‐fetoprotein; ßhCG = beta human chorionic gonadotrophin; FPR = false positive rate; hCG = human chorionic gonadotrophin; NT = nuchal translucency; PAPP‐A = pregnancy‐associated plasma protein‐A; uE3 = unconjugated oestriol
CI = confidence interval
Background
This is one of a series of reviews on antenatal screening for Down's syndrome following a generic protocol (Alldred 2010) ‐ see Published notes for more details.
Target condition being diagnosed
Down’s syndrome
Down’s syndrome affects approximately one in 800 live born babies (Cuckle 1987a). It results from a person having three, rather than two, copies of chromosome 21 – or the specific area of chromosome 21 implicated in causing Down's syndrome – as a result of trisomy (an additional copy of the whole chromosome) or translocation (duplication of part of the chromosome caused by rearrangements of parts of different chromosomes, resulting in three copies of information responsible for Down's syndrome). If not all cells are affected, the pattern is described as 'mosaic'. Down’s syndrome can cause a wide range of physical and mental problems. It is the commonest cause of mental disability, and is also associated with a number of congenital malformations, notably affecting the heart. There is also an increased risk of cancers such as leukaemia, and numerous metabolic problems including diabetes and thyroid disease. Some of these problems may be life‐threatening, or lead to considerable ill health, while some individuals with Down’s syndrome have only mild problems and can lead a relatively normal life.
There is no cure for Down’s syndrome, and antenatal diagnosis allows for preparation for the birth and subsequent care of a baby with Down’s syndrome, or for the offer of a termination of pregnancy. Having a baby with Down’s syndrome is likely to have a significant impact on family and social life, relationships and parents’ work. Special provisions may need to be made for education and care of the child, as well as accommodating the possibility of periods of hospitalisation.
Definitive invasive tests (amniocentesis and chorionic villus sampling (CVS)) exist that allow the diagnosis of Down's syndrome before birth but carry a risk of miscarriage. No test can predict the severity of problems a person with Down’s syndrome will have. Non‐invasive screening tests based on biochemical analysis of maternal serum or urine, or fetal ultrasound measurements, allow an estimate of the risk of a pregnancy being affected and provide parents with information to enable them to make choices about definitive testing. Such screening tests are used during the first and second trimester of pregnancy.
Screening tests for Down's syndrome
Initially, screening was determined solely by using maternal age to classify a pregnancy as high or low risk for trisomy 21, as it was known that older women had a higher chance of carrying a baby with Down’s syndrome (Penrose 1933).
Further advances in screening were made in the early 1980s, when Merkatz and colleagues investigated the possibility that low maternal serum alpha‐fetoprotein (AFP), obtained from maternal blood in the second trimester of pregnancy could be associated with chromosomal abnormalities in the fetus. Their retrospective case‐control study showed a statistically significant relationship between fetal trisomy, such as Down’s syndrome, and lowered maternal serum AFP (Merkatz 1984). This was further explored by Cuckle and colleagues in a larger retrospective trial using data collected as part of a neural tube defect (NTD) screening project (Cuckle 1984a). This work was followed by calculation of risk estimates using maternal serum AFP values and maternal age, which ultimately led to the introduction of the two screening parameters in combination (Alfirevic 2004).
In 1987, in a small case‐control study of women carrying fetuses with known chromosomal abnormalities, Bogart and colleagues investigated maternal serum levels of human chorionic gonadotrophin (hCG) as a possible screening tool for chromosomal abnormalities in the second trimester (Bogart 1987). This followed the observations that low hCG levels were associated with miscarriages, which are commonly associated with fetal chromosomal abnormalities. They concluded that high hCG levels were associated with Down’s syndrome and because hCG levels plateau at 18 to 24 weeks, that this would be the most appropriate time for screening. Later work suggested that the ß sub‐unit of hCG (free βhCG) was a more effective marker than total hCG (Macri 1990; Macri 1993).
Second trimester unconjugated oestriol (uE3), produced by the fetal adrenals and the placenta, was also evaluated as a potential screening marker. In another retrospective case‐control study, uE3 was shown to be lower in Down’s syndrome pregnancies compared with unaffected pregnancies. When used in combination with AFP and maternal age, it appeared to identify more pregnancies affected by Down’s syndrome than AFP and age alone (Canick 1988). Further work suggested that all three serum markers (AFP, hCG and uE3) showed even higher detection rates when combined with maternal age (Wald 1988a; Wald 1988b) and appeared to be a cost‐effective screening strategy (Wald 1992a).
Three other serum markers, produced by the placenta, have been linked with Down’s syndrome, namely pregnancy‐associated plasma protein A or PAPP‐A, inhibin A and a disintegrin and metalloprotease 12 (ADAM12). PAPP‐A has been shown to be reduced in the first trimester of Down’s syndrome pregnancies, with its most marked reduction in the early first trimester (Bersinger 1995). Inhibin A is high in the second trimester in pregnancies affected by Down’s syndrome (Cuckle 1995; Wallace 1995). There are some issues concerning the biological stability ‐ for example, delay in samples arriving in the laboratory ‐ and hence reliability of this marker, and the effect this will have on individual risk. ADAM 12 has been shown to be a potential first trimester marker with reduced maternal serum levels in pregnancy prior to 10 weeks (Laigaard 2003; Spencer 2008a).
In 1992, Nicolaides and colleagues demonstrated an association between increased nuchal translucency (NT) and chromosomal abnormalities (Nicolaides 1992). Nuchal translucency measurement requires an ultrasound scan of the fluid at the fetal neck between 10 and 13+6 weeks' gestation. If the amount is large, it suggests an increased risk of Down’s syndrome. This study was small (827 women), but led to further research into the use of NT scanning and its value when combined with serum tests. Other first trimester ultrasound markers, such as absent nasal bone, abnormal ductus venosus flow velocity and tricuspid regurgitation, have also been investigated.
In addition to serum and ultrasound markers for Down’s syndrome, work has been carried out looking at urinary markers. These markers include invasive trophoblast antigen, ß‐core fragment, free ßhCG and total hCG (Cole 1999). There is controversy about their value (Wald 2003a).
Screening and parental choice
Antenatal screening is used for several reasons (Alfirevic 2004), but the most important is to enable parental choice regarding pregnancy management and outcome. Before a woman and her partner opt to have a screening test, they need to be fully informed about the risks, benefits and possible consequences of such a test. This includes the choices they may have to face should the result show that the woman has a high risk of carrying a baby with Down’s syndrome and the implications of both false positive and false negative screening tests. They need to be informed of the risk of a miscarriage due to invasive diagnostic testing, and the possibility that a miscarried fetus may be chromosomally normal. If, following invasive diagnostic testing, the fetus is shown to have Down’s syndrome, further decisions need to be made about continuation or termination of the pregnancy, the possibility of adoption and finally, preparation for parenthood. Equally, if a woman has a test that shows she is at a low risk of carrying a fetus with Down’s syndrome, it does not necessarily mean that the baby will be born with a normal chromosomal make up. This possibility can only be excluded by an invasive diagnostic test (Alfirevic 2003). The decisions that may be faced by expectant parents inevitably engender a high level of anxiety at all stages of the screening process, and the outcomes of screening can be associated with considerable physical and psychological morbidity. No screening test can predict the severity of problems a person with Down's syndrome will have.
Index test(s)
This review examined serum screening tests used in the first and second trimester of pregnancy (up to 24 weeks' gestation) with and without first trimester ultrasound tests (up to 14 weeks' gestation). The review examined the following individual markers; NT measurement in the first trimester, ADAM 12, AFP, uE3, total hCG, free βhCG, Inhibin A and PAPP‐A. These markers can be used individually, in combination with age, and can also be used in combination with each other. The risks are calculated by comparing a woman's test result for each marker with values for an unaffected population, and multiplying this with her age‐related risk. Where several markers are combined, risks are computed using risk equations (often implemented in commercial software) that take into account the correlational relationships between the different markers and marker distributions in affected and unaffected populations.
Stepwise testing allows for triage of women into risk categories at two stages. Women found to be very high risk at the end of first trimester screening are offered invasive testing, whereas those women deemed to be lower risk are then screened again in the second trimester and a further overall risk is calculated once both stages of the test are completed.
Contingent screening is similar, however at the completion of first trimester screening women are classified into three groups – high risk, medium risk and low risk. High risk women are offered invasive testing at this stage, low risk women undergo no further screening and medium risk women are offered second trimester serum tests and calculation of a further overall risk once both stages of the test are completed.
Alternative test(s)
Down’s syndrome can be detected during pregnancy with invasive diagnostic tests such as amniocentesis or CVS, with or without prior screening. The ability to determine fetal chromosomal make up (also known as a karyotype) from amniotic fluid samples was demonstrated in 1966 by Steele and Breg (Steele 1966), and the first antenatal diagnosis of Down’s syndrome was made in 1968 (Valenti 1968). Amniocentesis is an invasive procedure which involves taking a small sample of the amniotic fluid (liquor) surrounding the baby, using a needle which goes through the abdominal wall into the uterus, and is usually performed after 15 weeks' gestation. CVS involves taking a sample of the placental tissue using a needle which goes through the abdominal wall and uterus or a cannula through the cervix. It is usually performed between 10 and 13 weeks' gestation. Amniocentesis and CVS are both methods of obtaining fetal chromosomes material, which are then used to diagnose Down’s syndrome. Both tests use ultrasound scans to guide placement of the needle. Amniocentesis carries a risk of miscarriage in the order of 1%; transabdominal CVS may carry a similar risk (Alfirevic 2003). Recent developments in the use of cell‐free fetal DNA detection in maternal serum are paving the way for non‐invasive diagnosis of Down's syndrome and other trisomies, however these tests were not used as reference standards in any of the studies examined.
There are many different screening tests which are available and offered which are the subject of additional Cochrane reviews and there are other reviews looking at this area. Tests being assessed in the other Cochrane reviews include first trimester serum tests (Alldred 2015); urine tests (Alldred 2015a); second trimester serum markers (Alldred 2012); and first trimester ultrasound tests alone, or in combination with first trimester serum tests (in press). Second trimester ultrasound markers have been assessed in a previous systematic review (Smith‐Bindman 2001).
Rationale
This is one of a suite of Cochrane reviews, the aim of which is to identify all screening tests for Down's syndrome used in clinical practice, or evaluated in the research setting, in order to try to identify the most accurate test(s) available, and to provide clinicians, policy makers and women with robust and balanced evidence on which to base decisions about interpreting test results and implementing screening policies to triage the use of invasive diagnostic testing. The full set of reviews is described in the generic protocol (Alldred 2010).
A systematic review of second trimester ultrasound markers for detection of Down’s syndrome concluded that nuchal fold thickening may be useful in detecting Down’s syndrome, but that it was not sensitive enough to be used as a screening test (Smith‐Bindman 2001). The review concluded that other second trimester ultrasound markers did not usefully distinguish between Down’s syndrome and pregnancies without Down’s syndrome. There has been no systematic review and meta‐analysis of serum, urine and first trimester ultrasound markers to enable rigorous and robust conclusions to be made about the diagnostic accuracy of available Down’s syndrome screening tests.
The topic has been split into several different reviews to allow for greater ease of reading and greater accessibility of data, and also to allow the reader to focus on separate groups of tests, for example, first trimester serum tests alone, first trimester ultrasound alone, first trimester serum and ultrasound, second trimester serum alone, first and second trimester serum, combinations of serum and ultrasound markers and urine markers alone. An overview review will compare the best tests, focusing on commonly used strategies, from each of these groups to give comparative results between the best tests in the different categories. This review is written with the global perspective in mind, rather than to conform with any specific local or national policy, as not all tests will be available in all areas where screening for Down's syndrome is carried out.
Objectives
The aim of this review was to estimate and compare the accuracy of first and second trimester serum markers with and without first trimester ultrasound markers for the detection of Down’s syndrome in the antenatal period, as combinations of markers. Individual markers are described in the other reviews belonging to this suite. Accuracy is described by the proportion of fetuses with Down’s syndrome detected by screening before birth (sensitivity or detection rate) and the proportion of women with a low risk (normal) screening test result who subsequently had a baby unaffected by Down's syndrome (specificity).
Investigation of sources of heterogeneity
We planned to investigate whether a uniform screening test is suitable for all women, or whether different screening methods are more applicable to different groups, defined by advanced maternal age, ethnic groups and aspects of the pregnancy and medical history such as multiple pregnancy, diabetes and family history of Down's syndrome. We also considered whether there existed evidence of overestimation of test accuracy in studies evaluating risk equations in the derivation sample rather than in a separate validation sample.
Methods
Criteria for considering studies for this review
Types of studies
We included studies in which all women from a given population had one or more index test(s) compared to a reference standard. Both consecutive series and diagnostic case‐control study designs were included. Randomised trials where individuals were randomised to different screening strategies and all verified using a reference standard were also eligible for inclusion. Studies in which test strategies were compared head‐to‐head either in the same women, or between randomised groups were identified for inclusion in separate comparisons of test strategies. Studies were excluded if they included less than five Down's syndrome cases, or more than 20% of participants were not followed up.
Participants
Pregnant women up to 24 weeks' gestation confirmed by ultrasound, who had not undergone previous testing for Down’s syndrome in their pregnancy were eligible. Studies were included if the pregnant women were unselected, or if they represented groups with increased risk of Down’s syndrome, or difficulty with conventional screening tests including maternal age greater than 35 years old, multiple pregnancy, diabetes mellitus and family history of Down’s syndrome.
Index tests
The following index tests were examined; nuchal translucency (NT) scanning, ADAM12, AFP, uE3,total hCG, free βhCG, Inhibin A, PAPP‐A, and combinations of these markers with maternal age. Combinations without maternal age were excluded.
We looked at comparisons of tests in isolation and in various combinations. All strategies included first and second trimester serum tests, and some included additional first trimester ultrasound markers. The maximum number of markers in any one test was seven, in combination with maternal age.
Where tests were used in comparison we looked at the performance of test comparisons according to predicted probabilities computed using risk equations and dichotomised into high risk and low risk (and medium risk, where applicable).
Target conditions
Down's syndrome in the fetus due to trisomy, translocation or mosaicism.
Reference standards
We considered several reference standards, involving chromosomal verification and postnatal macroscopic inspection.
Amniocentesis and chorionic villus sampling (CVS) are invasive chromosomal verification tests undertaken during pregnancy. They are highly accurate, but the process carries a 1% miscarriage rate, and therefore they are only used in pregnancies considered to be at high risk of Down's, or at the mother's request. All other types of testing (postnatal examination, postnatal karyotyping, birth registers and Down’s syndrome registers) are based on information available at the end of pregnancy. The greatest concern is not their accuracy, but the loss of the pregnancy to miscarriage between the serum test and the reference standard. Miscarriage with cytogenetic testing of the fetus is included in the reference standard where available. We anticipated that older studies, and studies undertaken in older women are more likely to have used invasive chromosomal verification tests in all women.
Studies undertaken in younger women and more recent studies were likely to use differential verification as they often only used prenatal karyotypic testing on fetuses considered screen positive/high risk according to the screening test; the reference standard for most unaffected infants being observing a phenotypically normal baby. Although the accuracy of this combined reference standard is considered high, it is methodologically a weaker approach as pregnancies that miscarry between the index test and birth are likely to be lost from the analysis, and miscarriage is more likely to occur in Down's than normal pregnancies. We investigated the impact of the likely missing false negative results in sensitivity analyses.
Search methods for identification of studies
We used one generic search strategy to identify studies for all reviews in this series.
Electronic searches
We applied a sensitive search strategy to search the following databases using the text words and MeSH terms detailed in Appendix 1, adapting the search strategy for each different database.
The following databases were searched.
MEDLINE via OVID (1980 to 25 August 2011)
Embase via Dialog Datastar (1980 to 25 August 2011)
BIOSIS via EDINA (1985 to 25 August 2011)
CINAHL via OVID (1982 to 25 August 2011)
The Database of Abstracts of Reviews of Effectiveness (25 August 2011)
MEDION (25 August 2011)
The Database of Systematic Reviews and Meta‐Analyses in Laboratory Medicine (www.ifcc.org/) (25 August 2011)
The National Research Register (Archived 2007)
Health Services Research Projects in Progress database (HSRPROJ) (25 August 2011)
The search strategy combined three sets of search terms (see Appendix 1). The first set was made up of named tests, general terms used for screening/diagnostic tests and statistical terms. Note that the statistical terms were used to increase sensitivity and were not used as a methodological filter to increase specificity. The second set was made up of terms that encompass Down's syndrome and the third set made up of terms to limit the testing to pregnant women. All terms within each set were combined with the Boolean operator OR and then the three sets were combined using AND. The terms used were a combination of subject headings and free‐text terms. The search strategy was adapted to suit each database searched.
We attempted to identify cumulative papers that reported data from the same data set, and contacted authors to obtain clarification of the overlap between data presented in these papers, in order to prevent data from the same women being analysed more than once.
Searching other resources
In addition, we examined references cited in studies identified as being potentially relevant, and those cited by previous reviews. We contacted authors of studies where further information was required.
We carried out forward citation searching of relevant items, using the search strategy in ISI citation indices, Google scholar and Pubmed ‘related articles’.
We did not apply language restrictions to the search.
Data collection and analysis
Selection of studies
Two review authors screened the titles and abstracts (where available) of all studies identified by the search strategy. Full‐text versions of studies identified as being potentially relevant were obtained and independently assessed by two review authors for inclusion, using a study eligibility screening pro forma according to the pre‐specified inclusion criteria. Any disagreement between the two review authors was settled by consensus, or where necessary, by a third party.
Data extraction and management
A data extraction form was developed and piloted using a subset of 20 identified studies (from all identified studies in this suite of reviews). Two review authors independently extracted data, and where disagreement or uncertainty existed, a third review author validated the information extracted.
Data on each marker were extracted as binary test positive/test negative results for Down's and non‐Down's pregnancies, with a high risk result ‐ as defined by each individual study ‐ being regarded as test positive (suggestive or diagnostic of Down's syndrome), and a low risk result being regarded as test negative (suggestive of absence of Down's syndrome). Where results were reported at several thresholds, we extracted data at each threshold.
We noted those in special groups that posed either increased risk of Down’s syndrome or difficulty with conventional screening tests including maternal age greater than 35 years old, multiple pregnancy, diabetes mellitus and family history of Down’s syndrome.
Assessment of methodological quality
We used a modified version of the QUADAS tool (Whiting 2003), a quality assessment tool for use in systematic reviews of diagnostic accuracy studies, to assess the methodological quality of included studies. We anticipated that a key methodological issue would be the potential for bias arising from the differential use of invasive testing and follow‐up for the reference standard according to index test results, bias arising due to higher loss to miscarriage in false negatives than true negatives. We chose to code this issue as originating from differential verification in the QUADAS tool: we are aware that it could also be coded under delay in obtaining the reference standard, and reporting of withdrawals. We omitted the QUADAS item assessing quality according to length of time between index and reference tests, as Down's syndrome is either present or absent rather than a condition that evolves and resolves, and disregarding the differential reference standard issue thus any length of delay is acceptable. Two review authors assessed each included study separately. Any disagreement between the two authors was settled by consensus, or where necessary, by a third party. Each item in the QUADAS tool was marked as ‘yes’, ‘no’ or ‘unclear’, and scores were summarised graphically. We did not use a summary quality score.
QUADAS criteria included the following 10 questions.
Was the spectrum of women representative of the women who will receive the test in practice? (Criteria met if the sample was selected from a wide range of childbearing ages, or selected from a specified ‘high risk’ group such as over 35s, family history of Down’s syndrome, multiple pregnancy or diabetes mellitus, provided all affected and unaffected fetuses included that could be tested at the time point when the screening test would be applied; criteria not met if the sample taken from a select or unrepresentative group of women (i.e. private practice), was an atypical screening population or recruited at a later time point when selection could be affected by selective fetal loss.)
Is the reference standard likely to correctly classify the target condition? (Amniocentesis, chorionic villus sampling, postnatal karyotyping, miscarriage with cytogenetic testing of the fetus, a phenotypically normal baby or birth registers are all regarded as meeting this criteria.)
Did the whole sample or a random selection of the sample receive verification using a reference standard of diagnosis?
Did women receive the same reference standard regardless of the index test result?
Was the reference standard independent of the index test result (i.e. the index test did not form part of the reference standard)?
Were the index test results interpreted without knowledge of the results of the reference standard?
Were the reference standard results interpreted without knowledge of the results of the index test?
Were the same clinical data (i.e. maternal age and weight, ethnic origin, gestational age) available when test results were interpreted as would be available when the test is used in practice?
Were uninterpretable/intermediate test results reported?
Were withdrawals from the study explained?
Statistical analysis and data synthesis
We initially examined each test or test strategy at each of the common risk thresholds used to define test positivity by plotting estimates of sensitivity and specificity from each study on forest plots and in receiver operating characteristic (ROC) space. Test strategies were selected for further investigation if they were evaluated in four or more studies or, if there were three or fewer studies, but the individual study results indicated performance likely to be superior to a sensitivity of 70% and specificity of 90%.
Estimation of average sensitivity and specificity
The analysis for each test strategy was undertaken first, by restricting to studies that reported a common threshold to estimate average sensitivity and specificity for each test at each threshold. Although data on all thresholds were extracted, we present only key common thresholds close to risks of 1:384, 1:250 and the 5% false positive rate (FPR), unless other thresholds were more commonly reported. Where combinations of tests were used in a risk score, we extracted the result for the test combination using the risk score and not the individual components that made up the test.
Meta‐analyses were undertaken using hierarchical summary ROC (HSROC) models, which included estimation of random‐effects in accuracy and threshold parameters when there were four or more studies. Otherwise, average sensitivity and specificity values were computed by using univariate random‐effects logistic regression models to average logit sensitivity and logit specificity separately because of insufficient number of studies to reliably estimate all the parameters in the HSROC model. It is common in this field for studies to report sensitivity for a fixed specificity (usually a 5% FPR). This removes the requirement to account for the correlation between sensitivity and specificity across studies by using a bivariate meta‐analytical method since all specificities are the same value. Thus, at a fixed specificity value, logit sensitivities were pooled using a univariate random‐effects model. This model was further simplified to a fixed‐effect model when there were only two or three studies and heterogeneity was not observed on the SROC plot. All analyses were undertaken using the NLMIXED procedure in SAS (version 9.2; SAS Institute, Cary, NC) and the xtmelogit command in Stata version 11.2 (Stata‐Corp, College Station, TX, USA).
Comparisons between tests
Comparisons between tests were first made utilising all available studies, selecting one threshold from each study to estimate a summary ROC curve without restricting to a common threshold. The threshold was chosen for each study according to the following order of preference: a) the risk threshold closest to one in 250; b) a multiples of the median (MoM) or presence/absence threshold; c) the performance closest to a 5% FPR or 95th percentile. The 5% FPR was chosen as a cut‐off point as this is the cut‐off most commonly reported in the literature. The analysis that used all available studies was performed by including the six most evaluated test strategies in a single HSROC model. The model included two indicator terms for each test to allow for differences in accuracy and threshold. As there were few studies for each test, a single summary ROC shape parameter was included in the model such that the fitted summary ROC curves did not cross. An estimate of the sensitivity of each test for a 5% FPR was derived from the summary ROC curve, and associated confidence intervals were obtained using the delta method.
Direct comparisons between tests were based on results of very few studies, and were analysed using a fixed‐effect HSROC model with symmetrical underlying summary ROC curves because the number of studies was insufficient to estimate between‐study heterogeneity in accuracy and threshold or asymmetry in the shape of the summary ROC curves. A separate model was used to make each pair‐wise comparison. Comparisons between tests were assessed by using likelihood ratio tests to test if the differences in accuracy were statistically significant or not. The differences were expressed as relative diagnostic odds ratios and were reported with 95% confidence intervals. As studies rarely report data cross‐classified by both tests for Down's and normal pregnancies, the analytical method did not take full account of the pairing of test results, but the restriction to direct head‐to‐head comparisons should have removed the potential confounding of test comparisons with other features of the studies. The strength of evidence for differences in performance of test strategies relied on evidence from both the direct and indirect comparisons.
Investigations of heterogeneity
If there were 10 or more studies available for a test, we planned to investigate heterogeneity by adding covariate terms to the HSROC model to assess the effect of a covariate on accuracy and threshold.
Sensitivity analyses
Mothers with pregnancies identified as high risk for Down's syndrome by ultrasound and serum testing are often offered immediate definitive testing by amniocentesis, whereas those considered low risk are assessed for Down's syndrome by inspection at birth. Such delayed and differential verification will introduce bias, most likely through there being greater loss to miscarriage in the Down's syndrome pregnancies that were not detected by the ultrasound and serum testing (the false negative diagnoses). Testing and detection of miscarriages is impractical in many situations, and no clear data are available on the magnitude of these miscarriage rates.
To account for the possible bias introduced by such a mechanism, we planned to perform sensitivity analyses by increasing the percentage of false negatives in studies where delayed verification in test negatives occurred (Mol 1999). We planned to incrementally increase the percentage from 10% to 50%, the final value representing a scenario where a third of more Down's pregnancies than normal pregnancies were likely to miscarry, thought to be higher than the likely value. We intended to conduct the sensitivity analyses on the analysis investigating the effect of maternal age on test sensitivity.
Assessment of reporting bias
Assessment of reporting bias was not performed.
Results
Results of the search
The search for the whole suite of reviews identified a total of 15,394 papers, once the results from each bibliographic database were combined and duplicates were removed. After screening out obviously irrelevant papers based on their title and abstract, 1145 papers remained and we obtained full‐text copies for formal assessment of eligibility. From these a total of 269 papers were deemed eligible and were included in the suite of reviews. A total of 22 studies (reported in 25 publications) were included in this review of first and second trimester serum screening, with and without ultrasound, involving 228,615 pregnancies including 1067 Down's syndrome pregnancies.
A total of 32 different test strategies combinations were evaluated in the 22 studies. The tests were produced from combinations of eight different tests, with and without maternal age; first trimester nuchal translucency (NT) and the serum markers AFP, uE3, total hCG, free βhCG, Inhibin A, PAPP‐A and ADAM 12. We examined tests combining first and second trimester markers with or without ultrasound as complete tests, and also examined stepwise and contingent strategies. The studies evaluated the following serum‐only tests: one single test without maternal age, and one septuple test, two sextuple tests, five quintuple tests, two quadruple tests and two triple test in combination with maternal age. Serum and ultrasound markers were evaluated in combination with maternal age: one study of seven markers, three studies of six markers, four studies of five markers, four studies of four markers and two studies of three markers. In addition, there were two contingent and three stepwise test strategies. Twelve of the 22 studies only evaluated the performance of a single test or test strategy, five compared two tests, two compared three tests, two compared five tests, and one compared 20 tests (Wald 2003b).
The following test combinations were the most evaluated and were each evaluated in four studies.
Six markers
First trimester NT, first trimester PAPP‐A , second trimester free ßhCG, second trimester uE3, second trimester AFP, second trimester Inhibin A, and maternal age (four studies; 40,348 women including 266 Down's syndrome pregnancies)
Four markers
First trimester PAPP‐A, second trimester total hCG, second trimester uE3, second trimester AFP and maternal age (four studies; 2474 women, including 236 Down's syndrome pregnancies)
First trimester NT, second trimester total hCG, second trimester uE3, second trimester AFP and maternal age (four studies; 13,708 women, including 136 Down's syndrome pregnancies)
Three markers
First trimester NT, second trimester total hCG, second trimester AFP and maternal age (four studies; 22,793 women, including 135 Down's syndrome pregnancies)
Of the remaining 28 test combinations, two were evaluated in three studies, eight were evaluated in two studies and the remaining 18 in single studies only.
Methodological quality of included studies
Methodological quality of the studies was judged to be high in half of the categories (Figure 1). Due to the nature of testing for Down's syndrome screening and the potential side effects of invasive testing, differential verification is almost universal in the general screening population, as most women whose screening test result is defined as low risk will have their screening test verified at birth, rather than by invasive diagnosis in the antenatal period. Additionally, it was not possible to ascertain from the included studies whether or not the results of index tests and reference standards were blinded. It would be difficult to blind clinicians performing invasive diagnostic tests (reference standards) to the index test result, unless all women received the same reference standard, which would not be appropriate in most scenarios. Any biases secondary to a lack of clinician blinding are likely to be minimal.
1.
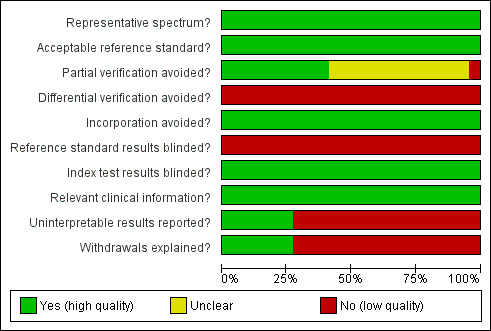
Methodological quality graph: review authors' judgements about each methodological quality item presented as percentages across all included studies.
Although not explicitly stated, most studies seemed to indicate 100% follow‐up. Inevitably there will be losses to follow‐up due to, for example, women moving out of the area of a study. It was therefore difficult to measure reporting of uninterpretable tests and hence reporting of withdrawals. Studies usually accounted for these and it is unlikely to have introduced significant bias. There was definitely under‐ascertainment of miscarriage, and very few papers accounted for miscarriage or performed tissue karyotyping in pregnancies resulting in miscarriage. Some studies attempted to adjust for predicted miscarriage rate and the incidence of Down's syndrome in this specific population, but most did not. We have not attempted to adjust for expected miscarriage rate in this review. This issue has the potential to have more influence with first trimester testing due to a higher miscarriage rate per se in this trimester.
Some studies that provided estimates of risk using multivariable equations used the same data set to evaluate performance of the risk equation as was used to derive the equation. This is often thought to lead to over‐estimation of test performance.
Findings
The results for the six most evaluated test strategies are presented in Table 1. Additional information is provided below.
1) First trimester nuchal translucency, first trimester PAPP‐A, second trimester free ßhCG, second trimester uE3, second trimester AFP, second trimester Inhibin A, and maternal age
Four studies (Aagaard‐Tillery 2009; Bestwick 2010; Wald 2003b; Wald 2009) evaluated this test strategy. The studies included 40,348 women in whom 266 pregnancies were affected by Down's syndrome. Over half the data were provided by Bestwick 2010 (22,746 women, including 106 Down's syndrome pregnancies). Studies presented data for different cut‐points but three (Aagaard‐Tillery 2009; Bestwick 2010; Wald 2003b) of the four studies also presented data for a 5% false positive rate (FPR). At a fixed cut‐point of 5% FPR on the summary ROC curve, the estimated sensitivity based on all four studies was 92% (95% confidence interval (CI) 88 to 95).
2) First trimester PAPP‐A, second trimester total hCG, second trimester uE3, second trimester AFP and maternal age
Four studies (Baviera 2010; Wald 2003b; Wright 2010 FASTER trial; Wright 2010 North York) evaluated this test strategy. The studies included 2474 women in whom 236 pregnancies were affected by Down's syndrome. Most of the data were provided by Wald 2003b (118 women, including 98 Down’s syndrome pregnancies). Studies presented data for cut‐points of 5% FPR (two studies Baviera 2010; Wald 2003b) and 1:250 risk (two studies Wright 2010 FASTER trial; Wright 2010 North York). At a fixed cut‐point of 5% FPR, the estimated sensitivity was 85% (95% CI 78 to 89).
3) First trimester nuchal translucency, second trimester total hCG, second trimester uE3, second trimester AFP and maternal age
Results for this test strategy were derived from four studies (Babbur 2005; Herman 2002; Schuchter 2001; Wald 2003b) and included 13,708 women in whom 136 pregnancies were known to be affected by Down's syndrome. Schuchter 2001 contributed 9342 pregnancies to the data. Studies presented data for cut‐points of 5% FPR (two studies: Schuchter 2001; Wald 2003b) and 1:250 risk (two studies:Babbur 2005; Herman 2002). At a fixed cut‐point of 5% FPR, the estimated sensitivity was 86% (95% CI 78 to 92).
4) First trimester nuchal translucency, second trimester total hCG, second trimester AFP and maternal age
Results were derived from four studies (Audibert 2001; Benattar 1999; Lam 2002; Wald 2003b) and included 22,793 women in whom 135 pregnancies were known to be affected by Down's syndrome. Lam 2002 contributed 16,237 pregnancies to the data. Studies presented data for cut‐points of 5% FPR (two studies: Lam 2002; Wald 2003b;) and 1:250 risk (two studies: Audibert 2001; Benattar 1999). At a fixed cut‐point of 5% FPR, the estimated sensitivity was 85% (CI 77 to 91).
5) Other test combinations
Of the 28 test combinations evaluated in three or fewer studies, 25 test combinations demonstrated estimated sensitivities of at least 70% and estimated specificities of more than 90%. Sixteen of these were evaluated in single studies only (see Table 2). Of the remaining nine test combinations evaluated in two or three studies, data were pooled for the following six tests.
First trimester PAPP‐A and second trimester total hCG, uE3, AFP and PAPP‐A, and maternal age evaluated in two studies (Wright 2010 FASTER trial; Wright 2010 North York) estimated a sensitivity of 78% (CI 66 to 86) and specificity of 98% (CI 96 to 99) at a cut‐point of 1:200 risk.
First trimester PAPP‐A and second trimester total hCG, uE3, AFP and inhibin A, and maternal age evaluated in three studies (Malone 2005; Palomaki 2006; Wald 2003b) estimated a sensitivity of 87% (CI 81 to 91) at a cut‐point of 5% FPR.
First trimester PAPP‐A and total hCG, and second trimester total hCG, uE3 and AFP evaluated in two studies (Wright 2010 FASTER trial; Wright 2010 North York) estimated a sensitivity of 80% (CI 68 to 88) and specificity of 97% (CI 94 to 98) at a cut‐point of 1:200 risk.
First trimester PAPP‐A and uE3, and second trimester total hCG, uE3 and AFP evaluated in two studies (Wright 2010 FASTER trial; Wright 2010 North York) estimated a sensitivity of 80% (CI 68 to 88) and specificity of 96% (CI 93 to 98) at a cut‐point of 1:200 risk.
First trimester NT and second trimester free ßhCG and AFP, and maternal age evaluated in two studies (Rozenberg 2002; Wald 2003b) estimated a sensitivity of 83% (CI 70 to 91) at a cut‐point of 5% FPR.
First trimester NT and PAPP‐A, and second trimester total hCG, uE3, AFP and Inhibin A, and maternal age evaluated in three studies (Malone 2005; Wald 2003b; Wald 2009) estimated a sensitivity of 95% (CI 90 to 97) at a cut‐point of 5% FPR.
Comparative analysis of the six selected test strategies
For each test, we obtained the detection rate (sensitivity) for a fixed false positive rate (FPR) (1‐specificity), a metric which is commonly used in Down’s syndrome screening to describe test performance. We chose to estimate detection rates at a 5% FPR in common with much of the literature. Figure 2 shows point estimates of the detection rate (and their 95% CIs) at a 5% FPR based on all available data for the six test strategies; the test strategies are ordered according to decreasing detection rates. The plot shows that all six test strategies have detection rates of 85% and above. The six marker maternal age‐adjusted combination of first trimester NT and PAPP‐A with second trimester total hCG, uE3, AFP and inhibin A showed the highest detection rate with an estimated detection rate of 95% (95% CI 90 to 97) based on data from three studies with 184 affected cases out of a total of 39,670 pregnancies. The next best performing strategy was a test combination with the same markers except that it included free ßhCG instead of total hCG. For this combination, the estimated detection rate was 92% (95% CI 88 to 95) based on data from four studies with 266 affected cases out of a total of 40,348 pregnancies. The remaining four test strategies showed similar detection rates.
2.
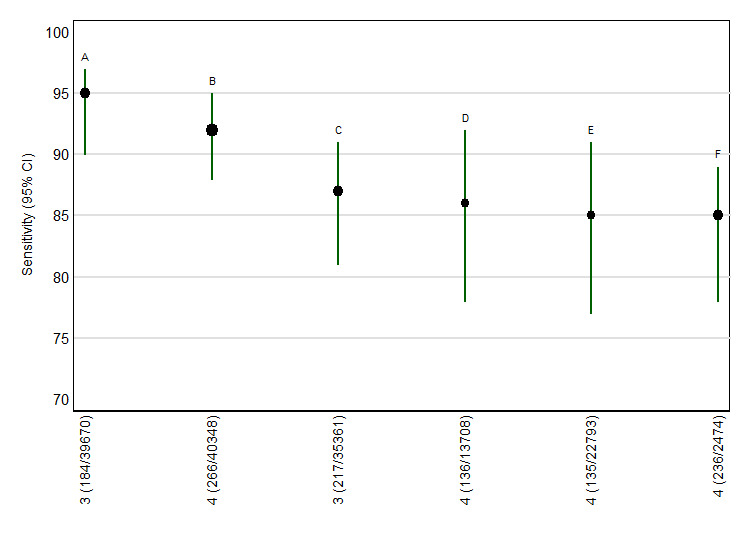
Detection rates (% sensitivity) at a 5% false positive rate for the six most evaluated test strategies (estimates from summary ROC curves).
A = First trimester NT and PAPP‐A , second trimester total hCG, uE3, AFP and inhibin A;B = First trimester NT and PAPP‐A , second trimester free ßhCG, uE3, AFP and inhibin A; C = First trimester PAPP‐A , second trimester total hCG, uE3, AFP and inhibin A; D = First trimester NT, second trimester total hCG, uE3 and AFP; E = First trimester NT, second trimester total hCG and AFP; and F = First trimester PAPP‐A , second trimester total hCG, uE3 and AFP.
All test combinations include maternal age. Each circle represents the summary sensitivity for a test strategy at a 5% false positive rate. The size of each circle is proportional to the number of Down's cases. The estimates are shown with 95% confidence intervals. The test strategies are ordered on the plot according to decreasing detection rate. The number of studies, cases and women included for each test strategy are shown on the horizontal axis.
The strength of evidence for differences in the diagnostic performance of the six test strategies relied on evidence from both direct and indirect comparisons. Table 3 shows pair‐wise direct comparisons (head‐to‐head) where studies were available. Such comparisons are regarded as providing the strongest evidence as differences between tests are unconfounded by study characteristics. The table shows the number of studies (K), the ratios of diagnostic odds ratios (DORs) with 95% CI and P values for each test comparison. There were no statistically significant differences in accuracy between any pair of tests. However, all comparisons in this table were based on one or two studies and so are unlikely to be powered to detect differences in accuracy.
1. Direct comparisons of the diagnostic accuracy of the six most evaluated test strategies.
|
Ratio of DORs (95% CI); P value (Studies) |
1T PAPP‐A, 2T total hCG, 2T uE3 and 2T AFP | 1T PAPP‐A, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A | 1T NT, 2T total hCG and 2T AFP | 1T NT, 2T total hCG, 2T uE3 and 2T AFP | 1T NT, 1T PAPP‐A, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A |
| 1T PAPP‐A, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A | 1.43 (0.39, 5.25); P = 0.49 (K = 1) |
||||
| 1T NT, 2T total hCG and 2T AFP | 0.86 (0.25, 2.96); P = 0.75 (K = 1) |
0.60 (0.16, 2.22); P = 0.34 (K = 1) |
|||
| 1T NT, 2T total hCG, 2T uE3 and 2T AFP | 1.23 (0.33, 4.57); P = 0.68 (K = 1) |
0.86 (0.22, 3.43); P = 0.78 (K = 1) |
1.44 (0.38, 5.41); P = 0.49 (K = 1) |
||
| 1T NT, 1T PAPP‐A, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A | 2.97 (0.53, 16.6); P = 0.15 (K = 1) |
2.08 (0.35, 12.3); P = 0.32 (K = 1) |
3.48 (0.62,19.6); P = 0.12 (K = 1) |
2.41 (41, 14.3); P = 0.24 (K = 1) |
|
| 1T NT, 1T PAPP‐A, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A | 2.41 (0.53, 11.0); P = 0.18 (K = 1) |
1.69 (0.35, 8.16); P = 0.41 (K = 2) |
2.82 (0.61, 13.0); P = 0.13 (K = 1) |
1.96 (0.40, 9.53); P = 0.30 (K = 1) |
1.87 (0.57, 6.06); P = 0.26 (K = 2) |
Direct comparisons were made using only data from studies that compared each pair of tests in the same population. Ratio of diagnostic odds ratios (DORs) were computed by division of the DOR for the test in the row by the DOR for the test in the column. If the ratio of DORs is greater than one, then the diagnostic accuracy of the test in the row is higher than that of the test in the column; if the ratio is less than one, the diagnostic accuracy of the test in the column is higher than that of the test in the row. All test combinations include maternal age. All test comparisons that were evaluated by only one study were from Wald 2003b.
1T = first trimester; 2T = second trimester; K = number of studies; CI = confidence interval
AFP = alpha‐fetoprotein; ßhCG = beta human chorionic gonadotrophin; FPR = false positive rate; hCG = human chorionic gonadotrophin; NT = nuchal translucency; PAPP‐A = pregnancy‐associated plasma protein‐A; uE3 = unconjugated oestriol.
Table 4 shows the same comparisons made using all available data. Results are generally in agreement with the direct comparisons, and in addition, showed some statistically significance differences (P < 0.05) suggesting that the six marker maternal age‐adjusted combination of first trimester NT and PAPP‐A with second trimester total hCG, uE3, AFP and inhibin A outperformed all the other test strategies except when compared with a similar strategy that included free ßhCG instead total hCG.
2. Indirect comparisons of the diagnostic accuracy of the six most evaluated test strategies.
|
Ratio of DORs (95% CI); P value |
1T PAPP‐A, 2T total hCG, 2T uE3 and 2T AFP | 1T PAPP‐A, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A | 1T NT, 2T total hCG and 2T AFP | 1T NT, 2T total hCG, 2T uE3 and 2T AFP | 1T NT, 1T PAPP‐A, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A | |
|
DOR (95% CI) Studies |
96 (48, 190) K =4 |
114 (62, 210) K = 3 |
103 (49, 215) K = 4 |
109 (51, 233) K = 4 |
214 (125, 367) K = 4 |
|
| 1T PAPP‐A, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A | 114 (62, 210) K = 3 |
1.19 (0.61, 2.32); P = 0.58 | ||||
| 1T NT, 2T total hCG and 2T AFP | 103 (49, 215) K = 4 |
1.08 (0.51, 2.36); P = 0.83 | 0.91 (0.43, 1.90); P = 0.78 | |||
| 1T NT, 2T total hCG, 2T uE3 and 2T AFP | 109 (51, 233) K = 4 |
1.14 (0.54, 2.42); P = 0.71 | 0.96 (0.45, 2.03); P = 0.90 | 1.06 (0.47, 2.41); P = 0.88 |
||
| 1T NT, 1T PAPP‐A, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A | 214 (125, 367) K = 4 |
2.24 (1.00, 5.00); P = 0.049 | 1.88 (0.88, 3.99); P = 0.094 | 2.08 (0.89, 4.87); P = 0.09 |
1.96 (0.82, 4.67); P = 0.12 |
|
| 1T NT, 1T PAPP‐A, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A | 339 (163, 705) K = 3 |
3.55 (1.28, 9.89); P = 0.019 | 2.98 (1.14; 7.80); P = 0.029 | 3.29 (1.15, 9.47); P = 0.030 |
3.11 (1.07, 9.07); P = 0.039 |
1.58 (0.64, 3.95); P = 0.30 |
Indirect comparisons were made using all available data. Ratio of diagnostic odds ratios (DORs) were computed by division of the DOR for the test in the row by the DOR for the test in the column. If the ratio of DORs is greater than one, then the diagnostic accuracy of the test in the row is higher than that of the test in the column; if the ratio is less than one, the diagnostic accuracy of the test in the column is higher than that of the test in the row. All test combinations include maternal age.
1T = first trimester; 2T = second trimester; K = number of studies; CI ‐ confidence interval.
AFP = alpha‐fetoprotein; ßhCG = beta human chorionic gonadotrophin; FPR = false positive rate; hCG = human chorionic gonadotrophin; NT = nuchal translucency; PAPP‐A = pregnancy‐associated plasma protein‐A; uE3 = unconjugated oestriol.
Comparison of integrated, contingent and stepwise strategy for a septuple combination of serum tests and first trimester nuchal translucency
Table 5 shows the results of two studies that assessed integrated, contingent or stepwise strategies. Integrated testing involves performing first trimester NT, PAPP‐A and free ßhCG, and second trimester uE3, AFP, total hCG and inhibin A, without disclosure of the first trimester result. The strategy was evaluated in one study (Malone 2005) that reported a 94% sensitivity (95% CI 87 to 98) and 89% specificity (95% CI 89 to 89) for a cut‐point of 1:150.
3. Comparison of integrated, contingent and stepwise strategy for a septuple combination of serum tests and first trimester nuchal translucency.
| Test combination | Screening policy | Study | Women (cases) | Sensitivity (95% CI) | Specificity (95% CI) | Threshold |
| First trimester NT, PAPP‐A and free ßhCG, and second trimester uE3, AFP, total hCG and inhibin A | Integrated | Malone 2005 | 33,546 (87) | 94 (87, 98) | 89 (89, 89) | 1:150 risk |
| First trimester NT, PAPP‐A and free ßhCG, if risk <1:30 invasive testing is offered, if risk 1:30‐1:1500, second trimester total hCG, uE3, AFP and inhibin A is performed | Contingent | Cuckle 2008 | 32,355 (86) | 91 (82, 96) | 95 (95, 96) | 1:270 risk |
| First trimester NT, PAPP‐A and free ßhCG, if risk <1:30 invasive testing is offered, if ≥ 1:30 second trimester total hCG, uE3, AFP and inhibin A is performed | Stepwise | Cuckle 2008 | 32,355 (86) | 92 (84, 97) | 95 (95, 95) | 1:270 risk |
AFP = alpha‐fetoprotein; ßhCG = beta human chorionic gonadotrophin; FPR = false positive rate; hCG = human chorionic gonadotrophin; NT = nuchal translucency; PAPP‐A = pregnancy‐associated plasma protein‐A; uE3 = unconjugated oestriol.
CI ‐ confidence interval.
In one study (Cuckle 2008), stepwise and contingent tests were compared in the same patient population, with similar detection rates (stepwise 91% (95% CI 84 to 97); contingent 92% (95% CI 82 to 96)) and identical false positive rates of 5% at cut‐points of 1:270.
The perceived advantages of the stepwise and contingent methods are that women deemed to be very high risk are offered invasive testing in the first trimester, allowing for earlier detection of Down's syndrome and subsequent management. Termination of pregnancy in the first trimester of pregnancy is safer than at later gestations. With contingent screening, where women are deemed to be low risk with a numerical risk of < 1:1500, no further testing is offered, and it does not appear to adversely affect the detection rate. In those women who are considered to be intermediate risk, additional second trimester serum tests may detect cases of Down's syndrome that would have been missed. Of note, in the study evaluated, all of the women found to have a risk of > 1:30 on first trimester screening were found to be high risk upon completion of the full contingent screening package. This type of screening may facilitate patient decision making, however further evaluation needs to be carried out.
It is difficult to make a comparison between the integrated method and the stepwise and contingent methods in practical terms, as the non‐disclosure of the first trimester result means that women would not be offered earlier diagnostic testing. More information is required about all three methods of testing in order to make a recommendation, as not all methods will be acceptable to women.
Investigation of heterogeneity and sensitivity analyses
The key characteristics of the 22 included studies is summarised in Table 6 with further details available in the Characteristics of included studies table. None of the tests was evaluated by 10 or more studies and so we were unable to investigate the effect of any potential source of heterogeneity. The planned sensitivity analyses were also not possible.
4. Maternal age, reference standard and study design characteristics of included studies.
| Study | Maternal age (years)* | Reference standard† | Withdrawals explained? | Study design |
| Aagaard‐Tillery 2009 | 30.6 (SD 6.1) | Karyotyping or follow‐up to birth | Of 33,546 trial participants only 7842 women with complete information for all screening tests and genetic sonography were included in the study. | Prospective cohort |
| Audibert 2001 | 30.1, all < 38, 86% < 35, 14% ≥35 | Prenatal karyotype conducted (in 7.6% of patients) depending on presence of risk >1/125, high maternal age, parental anxiety, history of chromosomal defects or parental translocation or abnormal second trimester scan. Cytogenetic testing of newborns with suspected abnormalities. Postmortum on terminations of pregnancy or miscarriages. Follow‐up to neonatal examination in newborns. | 35 women were lost to follow‐up (they had all had normal NT results). 340 women who did not want second trimester serum screening withdrew from that part of the study. Women lost to follow‐up were excluded in the final analysis. All detected cases were terminated. | Prospective consecutive series |
| Babbur 2005 | Median 37 (range 19 to 46) | Invasive testing offered to women with NT > 3 mm or risk > 1:250 as defined by combined NT and serum results CVS from 11 weeks, amniocentesis from 15 weeks). Rapid in situ hybridisation test in patients with risk > 1:30. No details given of any follow‐up to birth | 463 patients having NT did not go on to have second trimester serum testing. Women with miscarriages excluded. | Prospective cohort |
| Baviera 2010 | 35.3 for Down's cases, 30.4 for controls | Amniocentesis or follow‐up to birth | No details of withdrawals given. | Case control |
| Benattar 1999 | 32 (16 to 46), 8.3% > 35 | Amniocentesis due to maternal age > 38 years (6.1% or women). Karyotyping encouraged for women with positive result on one or more index test. No details of reference standard for index test negative women. | No details of withdrawals given. 12 patients were lost to follow‐up due to miscarriages | Prospective cohort |
| Bestwick 2010 | Median 39 for Down's cases, 34 for non‐Down's cases | Karyotyping or follow‐up to birth | No details of withdrawals given. | Retrospective cohort |
| Cuckle 2008 | Not reported | Karyotyping or follow‐up to birth | No details of withdrawals given. | Prospective cohort |
| Goh 1996 | 33 | Karyotyping or follow‐up to birth | No details of withdrawals given. | Cohort |
| Guanciali‐Franchi 2010 | 31.8 | Karyotyping or follow‐up to birth | No details of withdrawals given. | Prospective cohort |
| Habayeb 2010 | Median 35.4 (range 18 to 49) | Karyotyping or follow‐up to birth | No details of withdrawals given. | Cohort |
| Herman 2002 | Not reported | Karyotyping or follow‐up to birth | No details of withdrawals given. | Case control |
| Lam 2002 | 30.5 (19% ≥35) (unaffected pregnancies) | Women considered high risk offered CVS (0.7%) or amniocentesis (11.8%). Follow‐up to birth | Details given for patients excluded and those without follow‐up data. | Prospective cohort |
| Malone 2005 | 21.6% aged 35 and above | Amniocentesis (offered to women with positive results from any screening test) or follow‐up to birth. | Details given for patients who did not undergo different index tests. Unclear which patients did not have follow‐up data. Appears that aborted/miscarried foetuses did not have follow‐up. | Prospective cohort |
| Okun 2008 Integrated | 32 | Karyotyping or follow‐up to birth | 2614 (8%) of women undergoing integrated screening did not return for the second trimester part of the test. | Prospective cohort |
| Palomaki 2006 | 33.9 (SD 4.4) for Down's cases, 35.9 (SD 3.6) for controls | Karyotyping or follow‐up to birth | No details of withdrawals given. | Case control |
| Rodrigues 2009 | 30.6 for integrated screening, 30.9 for serum integrated screening | Karyotyping or follow‐up to birth | No details of withdrawals given. | Retrospective cohort |
| Rozenberg 2002 | 30.5 (18 to 37) | Amniocentesis offered to patients with NT > 3 mm or serum marker risk was > 1:250. Follow‐up to birth. | No details of withdrawals given. 3.4% of patients were lost to follow‐up and were excluded from the study. This included 113 women (1.2%) with miscarriages. | Prospective cohort |
| Schuchter 2001 | 28 (range 15 to 46), 10.7% aged 35 and above | CVS (offered to patients with first trimester NT > 3.5 mm), amniocentesis (offered to patients with first trimester NT 2.5 to 3.4, high risk on second trimester serum testing (> 1:250) and those > 35 years) or follow‐up to birth. | No details of withdrawals given. Women having miscarriages were excluded from the study. | Retrospective cohort |
| Wald 2003b | Not reported | Invasive testing (following second trimester screening) or follow‐up to birth. | No details of withdrawals given. | Case control |
| Wald 2009 | Median 33 (range 15 to 51), 20% aged 37 and above | Karyotyping or follow‐up to birth | No details of withdrawals given. | Retrospective cohort |
| Wright 2010 FASTER trial | Not reported | Karyotyping or follow‐up to birth | No details of withdrawals given. | Case control |
| Wright 2010 North York | Not reported | Karyotyping or follow‐up to birth | No details of withdrawals given. | Case control |
CVS = chorionic villus sampling; NT = nuchal translucency; SD = standard deviation
*Mean maternal age presented unless otherwise indicated.
†In all studies the choice of reference standard was dependent on the results of the index test.
Discussion
Summary of main results
We found 22 studies evaluating first and second trimester Down’s syndrome serum screening tests, with or without first trimester nuchal translucency (NT). Few studies provided unconfounded comparisons of test strategies by applying and comparing several strategies using the same serum sample, the majority of studies only evaluating a single test combination. A summary of results for the six most commonly evaluated test strategies is given in Table 1, and the remaining 26 test strategies are given in Table 2.
Three key findings were noted.
The combined test comprised of first trimester NT and PAPP‐A, and second trimester total hCG, uE3, AFP and Inhibin A, and maternal age evaluated in three studies (Malone 2005; Wald 2003b; Wald 2009) estimated a sensitivity of 95% (confidence interval (CI) 90 to 97) at a cut‐point of 5% FPR. In indirect comparisons this test combination significantly outperformed all others, except the test combination of first trimester NT, first trimester PAPP‐A, second trimester free ßhCG, second trimester uE3, second trimester AFP, second trimester Inhibin A, and maternal age with a sensitivity of 92% (95% CI 88 to 95) for a fixed 5% FPR.
In direct comparisons of tests in the same population of women, no test was found to be significantly better. These comparisons were based on one or two studies, and are therefore unlikely to be powered to detect differences.
Stepwise and contingent screening strategies show promising detection rates for fixed FPRs, however due to the nature of the test strategies it is not appropriate to make comparisons between these tests and those that do not involved stratification or risk at several different points in the screening journey. These test strategies warrant further study.
Strengths and weaknesses of the review
This review is the first comprehensive review of first and second trimester serum and ultrasound screening. We examined papers from around the world, covering a wide cross‐section of women in varying populations. We contacted authors to verify data where necessary to give as complete a picture as possible while trying to avoid replication of data.
There were a number of factors that made meta‐analysis of the data difficult, which we tried to adapt for in order to allow for comparability of data presented in different studies.
There were many different cut‐points used to define pregnancies as high or low risk for Down's syndrome. This means that direct comparison is more difficult than if all studies used the same cut‐point to dichotomise their populations.
There were many different risk equations and software applications in use for combination of multiple markers, which were often not described in the papers. This means that risks may be calculated by different formulae and they may not be directly comparable for this reason. It is possible that this is responsible for unexplained heterogeneity in results.
Different laboratories and clinics run different assays and use different machines and methods. This may influence raw results and subsequent risk calculations. Many laboratories have a quality assessment or audit trail, however, this may not necessarily be standard across the board. For example, how many assays are run, how often medians are calculated and adjusted for a given population and how quickly samples are tested from initially being taken.
Few studies made direct comparisons between tests, making it difficult to detect if a real difference exists between tests (i.e. how different tests perform in the same population). There were differences in populations, with assay medians being affected, for example, by race. It is not certain whether it is appropriate to make comparisons between populations which are inherently different.
We were unable to perform the investigations of heterogeneity that we had originally intended to because the data simply were not available. The vast majority of papers looking at pregnancies conceived by IVF, affected by diabetes, multiple gestation or a family history of Down's syndrome involved unaffected pregnancies only.
In addition, the search for this review was last updated in August 2011, and it is possible that new studies may have been published which have not been included. Since the search was completed we have kept a watching brief on outputs and are not aware of any studies with large sample sizes which could substantially affect the findings.
Applicability of findings to the review question
Potentially, when planning screening policy or a clinical screening programme, clinicians and policy makers need to make decisions about a finite number of tests or type of tests that can be offered. These policies are often driven by both the needs of a specific population and by financial resources. Economic analysis was considered to be outside of the scope of this review. Many of the tests examined as part of this review are already commercially available and in use in the clinical setting. The studies were carried out on populations of typical pregnant women and therefore, the results should be considered comparable with most pregnant populations encountered in every day clinical practice.
We were unable to extract information about harms of testing, information about miscarriage rates and uptake of definitive testing as the data were not available the majority of the time. While it is unlikely that major differences between the tests evaluated here exist in terms of direct harms of testing, as they are all based on ultrasound, with or without a blood sample, differences in accuracy may lead to differences in the use of definitive testing and its consequent adverse outcomes.
In some countries with a defined screening policy (i.e. the UK), first trimester screening plays a major role, usually in combination with first trimester ultrasound scanning, and second trimester serum screening is also readily available. In other countries however, there may only be a limited range of tests or markers available—often second trimester markers, rather than first trimester markers. The results of this review should be interpreted and applied in the context of test availability and local restrictions, populations or policies.
Authors' conclusions
Implications for practice.
The evidence supports the use of the six marker maternal age‐adjusted combination of first trimester nuchal translucency (NT) and pregnancy‐associated plasma protein‐A (PAPP‐A) with second trimester total human chorionic gonadotrophin (hCG), unconjugated oestriol (uE3), alpha‐fetoprotein (AFP) and inhibin A, which outperformed other test strategies. However the evidence was limited, based on small numbers of studies and the finding was not demonstrated in direct comparison of markers in the same populations of women. We cannot recommend a single test combination for Down’s syndrome screening. The choice of multiple markers will depend on the availability of certain assays in local laboratories. There is little evidence to recommend the use of first and second trimester serum markers without the addition of first trimester ultrasound. We would not recommend that these tests be introduced into wider clinical practice without careful consideration of cost.
Implications for research.
Further evaluation of test combinations involving contingent and stepwise strategies are required to determine whether they offer superior test performance.
Future studies should ensure that adequate sample sizes are recruited, and take opportunities to make comparisons of test performance testing several alternative test combinations on the same population. Such direct comparison removes issues of confounding when making test comparisons, and allows a clear focus on testing the incremental benefit of increasingly complex and expensive testing strategies. The reporting of studies of test accuracy can be improved and more closely adhere to the standards for the reporting of diagnostic accuracy studies (STARD) guideline. Three key aspects of this are: 1) formally testing the statistical significance of differences in test performance in direct comparisons and estimating incremental changes in detection rates (together with confidence intervals); 2) clearly reporting the number of mothers studied and their results; and 3) reporting the numbers of women who are lost to follow‐up. Many authors reported results of extrapolating findings to age‐standardised national cohorts to demonstrate the performance of the test, and failed to report the actual numbers studied and evaluated.
For the purposes of meta‐analysis and to allow for comparisons to be made between different tests and combinations, we would recommend the publication of consensus standard algorithms for estimating risk, and reporting of test performance at a standard set of thresholds. This would be difficult to achieve and implement, but an attempt at consensus should be made.
Notes
This review belongs to a suite of reviews examining antenatal screening for Down's syndrome which includes:
First trimester serum tests for Down's syndrome screening (Alldred 2015);
Urine tests for Down's syndrome screening (Alldred 2015a)
Second trimester serum tests for Down's syndrome screening (Alldred 2012);
First trimester ultrasound tests alone or in combination with first trimester serum tests for Down's syndrome screening (in press)
First and second trimester serum tests with and without first trimester ultrasound tests for Down's syndrome screening (this review).
The plans for these reviews were described in a generic protocol (Alldred 2010) published in the Cochrane Library in 2010. The project as a whole has been much larger than initially anticipated, both in terms of size and statistical complexity. The initial search was completed in 2007 and an updated search in August 2011. After identifying studies appropriate for inclusion, a significant amount of time has been devoted to data management and analysis.
The authors are conscious of the time lag from the latest literature search to publication, and the potential for the introduction of new urine tests in this time frame. The authors are also conscious of the potential for publication of new data pertaining to tests included in this review. Whilst not fulfilling the usual Cochrane up‐to‐date criteria, this review is published because it provides historical context in what is a rapidly‐changing field, and because it is unlikely to ever be repeated.
Acknowledgements
We acknowledge the assistance of the Pregnancy and Childirth Cochrane Review Group editorial base with writing the searches and other aspects of this review.
As part of the pre‐publication editorial process, this review has been commented on by two peers (an editor and referee who is external to the editorial team) and a member of the Pregnancy and Childbirth Group's international panel of consumers.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Pregnancy and Childbirth. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Appendices
Appendix 1. Search Strategy
Database: Ovid MEDLINE
‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐
1 exp Prenatal Diagnosis/
2 nuchal translucency.mp.
3 exp Pregnancy‐Associated Plasma Protein‐A/
4 pregnancy associated plasma protein a.mp.
5 papp‐a.mp.
6 exp Chorionic Gonadotropin, beta Subunit, Human/
7 (b‐hcg or bhcg).mp.
8 human chorionic gonadotropin.mp.
9 exp alpha‐Fetoproteins/
10 alphafetoprotein$.mp.
11 alpha‐fetoprotein$.mp.
12 afp.mp.
13 (unconjugated estriol or unconjugated oestriol).mp.
14 ue3.mp.
15 exp INHIBINS/
16 inhibin a.mp.
17 ultrasound.mp.
18 amniocentesis/
19 chorion$ vill$ sampling.mp.
20 Chorionic Villi‐Sampling/
21 nasal bone.mp.
22 tricuspid regurgitation.mp.
23 ductus venosus.mp
24 marker$.mp.
25 screen$.mp.
26 detect$.mp.
27 accura$.mp.
28 predict$.mp.
29 ROC.mp.
30 ROC curve/
31 AUC.mp.
32 Area under curve/
33 exp false negative reactions/ or exp false positive reactions/
34 (false positive$ or false negative$).mp.
35 likelihood ratio$.mp.
36 sensitiv$.mp.
37 specific$.mp.
38 diagnos$.ti,ab.
39 "reproducibility of results".mp.
40 reference value$.mp.
41 reference standard$.mp.
42 exp Down Syndrome/
43 downs syndrome.mp.
44 down syndrome.mp.
45 trisomy 21.mp.
46 Aneuploidy/
47 aneuploidy.mp.
48 Mosaicism/
49 mosaicism.mp.
50 or/1‐41
51 or/42‐49
52 50 and 51
53 (antenatal$ or prenatal$ or trimester$ or pregnan$ or fetus or foetus or fetal or foetal).mp.
54 52 and 53
55 animal/ not (humans/ and animal/)
56 54 not 55
*******************************************************
EMBASE via Dialog Datastar
1. PRENATAL‐DIAGNOSIS#.DE.
2. FETUS‐ECHOGRAPHY#.DE.
3. PREGNANCY‐ASSOCIATED‐PLASMA‐PROTEIN‐A#.DE.
4. CHORIONIC‐GONADOTROPIN‐BETA‐SUBUNIT#.DE.
5. HCG.AB.
6. PAPP.AB.
7. ALPHA‐FETOPROTEIN#.DE.
8. AFP.AB.
9. ALPHA ADJ FETOPROTEIN$
10. ALPHAFETOPROTEIN$
11. BETA ADJ HUMAN ADJ CHORIONIC ADJ GONADOTROPIN
12. PREGNANCY ADJ ASSOCIATED ADJ PLASMA ADJ PROTEIN
13. (UNCONJUGATED ADJ ESTRIOL OR UNCONJUGATED ADJ OESTRIOL).TI.
14. (UNCONJUGATED ADJ ESTRIOL OR UNCONJUGATED ADJ OESTRIOL).AB.
15. UE3
16. INHIBIN‐A#.DE.
17. INHIBIN ADJ A
18. ULTRASOUND
19. AMNIOCENTESIS
20. CHORION‐VILLUS‐SAMPLING.DE.
21. NASAL ADJ BONE
22. TRICUSPID ADJ REGURGITATION
23. DUCTUS ADJ VENOSUS
24. MARKER OR MARKERS
25. SCREEN OR SCREENING
26. DETECT OR DETECTING OR DETECTION
27. FALSE ADJ POSITIVE$
28. FALSE ADJ NEGATIVE$
29. SENSITIVITY OR SENSITIVE OR SENSITIVITIES
30. SPECIFICITY OR SPECIFICITIES
31. (DIAGNOSE OR DIAGNOSIS OR DIAGNOSTIC OR DIAGNOSTICS OR DIAGNOSES
OR DIAGNOSED).TI.
32. (DIAGNOSE OR DIAGNOSIS OR DIAGNOSTIC OR DIAGNOSTICS OR DIAGNOSES
OR DIAGNOSED).AB.
33. ROC.AB.
34. AUC.AB.
35. AREA‐UNDER‐THE‐CURVE.DE.
36. ROC‐CURVE.DE.
37. ACCURA$
38. PREDICT$
39. REPRODUCIBILITY.DE.
40. REFERENCE ADJ VALUE$
41. REFERENCE‐VALUE.DE.
42. REFERENCE ADJ STANDARD$
43. DOWN‐SYNDROME#.DE.
44. DOWN ADJ SYNDROME OR DOWNS ADJ SYNDROME
45. TRISOMY ADJ '21'
46. MOSAICISM
47. ANEUPLOIDY
48. ANTENATAL$ OR PRENATAL$ OR PREGNANCY OR PREGNANT OR TRIMESTER$ OR MATERNAL OR FETUS OR FOETUS OR FOETAL OR FETAL
49. 1 OR 2 OR 3 OR 4 OR 5 OR 6 OR 7 OR 8 OR 9 OR 10 OR 11 OR 12 OR 13 OR 14 OR 15 OR 16 OR 17 OR 18 OR 19 OR 20 OR 21 OR 22 OR 23 OR 24 OR 25 OR 26 OR 27 OR 28 OR 29 OR 30 OR 31 OR 32 OR 33 OR 34 OR 35 OR 36 OR 37 OR 38 OR 39 OR 40 OR 42
50. 43 OR 44 OR 45 OR 46 OR 47
51. 48 AND 49 AND 50
52. HUMAN=YES
53. 51 AND 52
ADJ = adjacent AB = abstract
TI = title $ = truncation symbol DE = descriptor (similar to MeSH)
*******************************************************
CINAHL via OVID
‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐
1 exp Prenatal Diagnosis/
2 nuchal translucency.mp.
3 pregnancy associated plasma protein.mp.
4 papp$.ti,ab.
5 exp Gonadotropins, chorionic/
6 (b‐hcg or bhcg).mp.
7 human chorionic gonadotropin.mp.
8 exp alpha‐Fetoproteins/
9 alphafetoprotein$.mp.
10 alpha‐fetoprotein$.mp.
11 afp.mp.
12 (unconjugated estriol or unconjugated oestriol).mp.
13 ue3.mp.
14 inhibin$.mp.
15 ultrasound.mp.
16 amniocentesis/
17 chorion$ vill$ sampling.mp.
18 Chorionic Villi‐Sampling/
19 nasal bone.mp.
20 tricuspid regurgitation.mp.
21 ductus venosus.mp.
22 marker$.mp.
23 screen$.mp.
24 detect$.mp.
25 accura$.mp.
26 predict$.mp.
27 ROC.mp.
28 ROC curve/
29 AUC.mp.
30 "area under curve".mp.
31 exp false negative reactions/ or exp false positive reactions/
32 (false positive$ or false negative$).mp.
33 likelihood ratio$.mp.
34 sensitiv$.mp.
35 specific$.mp.
36 diagnos$.ti,ab.
37 "reproducibility of results".mp.
38 reference value$.mp.
39 reference standard$.mp.
40 exp Down Syndrome/
41 downs syndrome.mp.
42 down syndrome.mp.
43 trisomy 21.mp.
44 aneuploidy.mp.
45 mosaicism.mp.
46 (antenatal$ or prenatal$ or trimester$ or pregnan$ or fetus or foetus or fetal or foetal).mp.
47 or/1‐39
48 or/40‐45
49 47 and 48 and 46
*******************************************************
Search terms and instructions for Biosis
The following search terms were entered separately in standard search box (select ‘Titles/subject/abstract’ from the drop‐down box on the right of the search box).
“reference standard*”
“reference value*”
“reproducibility of results”
diagnos*
sensitiv*
specific*
“likelihood ratio*”
“false negative*
“false positive”
“area under curve”
ROC
AUC
predict*
detect*
marker*
screen*
accura*
“ductus venosus”
“nasal bone”
“tricuspid regurgitation”
“chorion* vill* sampling”
amniocentesis
ultrasound
inhibin*
“unconjugaed oestriol”
“unconjugated estriol”
afp
“alpha fetoprotein*”
alphafetoprotein*
“ bhcg”
“human chorionic gonadotrophin”
“papp a”
“pregnancy associated plasma protein”
“nuchal translucency”
foetal
fetal
foetus
foetal
prenatal*
antenatal*
pregnan*
maternal*
“trisomy 21”
mosaicism
“down* syndrome”
The search then used the history function to combine terms:
1‐34 – combine using OR
35 – 42 – combine using OR
43 – 45 – combine using OR
The three sets were combined using AND
The combined search strategy had the form
(((((((al: "trisomy 21") or (al: (mosaicism))) or (al: "down* syndrome"))) and (((((((((((((((((((((((((((((((((((al: "reference standard*") or (al: "reference value*")) or (al: "reproducibility of results")) or (al: (diagnos*))) or (al: (specific*))) or (al: (sensitiv*))) or (al: "likelihood ratio*")) or (al: "false negative*")) or (al: "false positive*")) or (al: "area under curve")) or (al: (auc))) or (al: (roc))) or (al: (predict*))) or (al: (accura*))) or (al: (detect*))) or (al: (screen*))) or (al: (marker*))) or (al: "ductus venosus")) or (al: "tricuspid regurgitation")) or (al: "nasal bone")) or (al: "chorion* vill* sampling")) or (al: (amniocentesis))) or (al: (ultrasound))) or (al: (inhibin*))) or (al: "unconjugated oestriol")) or (al: "unconjugated estriol")) or (al: (afp))) or (al: "alpha feto protein*")) or (al: "alpha fetoprotein*")) or (al: "b hcg")) or (al: "human chorionic gonadotropin")) or (al: "papp a")) or (al: "pregnancy associated plasma protein")) or (al: "nuchal translucency")))) and (((((((((al: (foetal)) or (al: (fetal))) or (al: (foetus))) or (al: (fetus))) or (al: (pregnan*))) or (al: (trimester*))) or (al: (prenatal*))) or (al: (antenatal*))))))
*******************************************************
The Database of Abstracts of Reviews of Effectiveness (DARE), National Research Register and Health Services Research Projects in Progress database
:
Down syndrome (MeSH)
down* next syndrome
trisomy
aneuploidy
mosaicism
OR/ 1‐5
*******************************************************
MEDION (http://www.mediondatabase.nl/)
ICPC code for pregnancy – ‘W’.
*******************************************************
The Database of Systematic Reviews and Meta‐Analyses in Laboratory Medicine – download the database to a .pdf file and search for the following terms separately:
Down
Trisomy
Aneuploidy
Pregnant
Pregnancy
Pregnancies
Mosaicism
*******************************************************
Appendix 2. Glossary of terms (adapted in part from the UK National Screening Committee Glossary)
| Abnormal ductus venosus flow velocity | The ductus venosus is a vessel in the fetus which allows oxygenated blood from the placenta to bypass the fetal liver and flow straight to the heart. In conditions such as Down’s syndrome the pressure in this vessel can be abnormally high. |
| Absent nasal bone | Absence of the bone that forms the bridge of the nose, which may be detected at ultrasound scan during early pregnancy. |
| Affected individuals | Those individuals who are affected by the disorder for which they are being screened. |
| Amniocentesis | Amniocentesis is an invasive procedure which involves taking a small sample of the amniotic fluid (liquor) surrounding the baby, using a needle which goes through the abdominal wall into the uterus, and is usually performed after 15 weeks' gestation. |
| Chorionic villus sampling (CVS) | Chorionic villus sampling involves taking a sample of the placental tissue using a needle which goes through the abdominal wall and uterus or a cannula through the cervix. It is usually performed between 10 and 13 weeks' gestation. |
| Combined test | First trimester test (up to 13 + 6 weeks of pregnancy) based on combining nuchal translucency measurement with free beta‐hCG, pregnancy‐associated plasma protein A (PAPP‐A) and the woman’s age. |
| Diagnostic accuracy | The amount of agreement between the information from the index test and the reference standard (see below). |
| Diagnostic test | A definitive test, performed after a positive screening test result that gives a diagnosis (i.e. yes or no). |
| Double test | Second trimester test (from 13 + 6 up to 24 weeks of pregnancy) based on the measurement of alpha‐fetoprotein (AFP), human chorionic gonadotrophin (hCG ß either free beta‐hCG or total hCG), together with the woman’s age. |
| First trimester | Pregnancy from conception up to 13 weeks and 6 days. |
| Iatrogenic | A disease or condition in a patient occurring as a result of treatment. |
| Index test | A test or group of tests being evaluated in a systematic review. |
| Integrated test | Measurements performed at different times of pregnancy combined into a single test result. Unless otherwise specified, 'integrated test' refers to the combination of nuchal translucency measurement and PAPP‐A in the first trimester, with the quadruple test (see below) in the second. |
| Mosaicism | This is a condition in which person has some cells containing a normal number of chromosomes, and some containing an abnormal number. The more abnormal cells there are, the greater the effect. |
| Multiple of the median (MOM) | The serum test concentration for a pregnant woman divided by the average (median) for unaffected pregnancies in a defined population at the same stage of pregnancy. |
| Quadruple test | Second trimester test (from 13 + 6 up to 24 weeks of pregnancy) based on the measurement of AFP, uE3, free beta‐hCG (or total hCG), and inhibin‐A together with the woman’s age. |
| Reference Standard | The best available method for establishing the presence or absence of the target disease or condition. |
| Second trimester | Pregnancy from 14 weeks to 28 weeks' gestation. Note that for the purposes of this Cochrane review, second trimester testing refers to the period of 14 to 24 weeks' gestation. |
| Tricuspid regurgitation | Leakiness of or backflow of blood through the tricuspid valve of the heart. The tricuspid valve separates the upper and lower chambers of the right side of the heart. |
| Triple test | Second trimester test (from 14 up to 24 weeks of pregnancy) based on the measurement of AFP, unconjugated oestriol (uE3), and hCG (either total hCG or free beta‐hCG) together with the woman's age. |
| Trisomy | The presence of an extra chromosome resulting in three copies of a particular chromosome instead of the normal two. |
| Translocation | Part of one chromosome is broken off and attached to another chromosome. This does not usually cause the individual any problems as they have a normal amount of chromosomes, but in an abnormal arrangement. It can be passed on as an extra chromosome to offspring, resulting in conditions such as Down's syndrome. |
Data
Presented below are all the data for all of the tests entered into the review.
Tests. Data tables by test.
1. Test.

Age, 1T PAPP‐A , 2T free ßhCG and 2T AFP at 5% FPR.
2. Test.

Age, 1T PAPP‐A , 2T free ßhCG and 2T AFP, risk 1:300.
3. Test.

Age, 1T PAPP‐A , 2T total hCG, and 2T AFP at 5% FPR.
4. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3 and 2T AFP at 5% FPR.
5. Test.
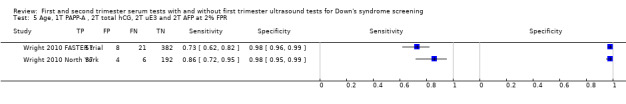
Age, 1T PAPP‐A , 2T total hCG, 2T uE3 and 2T AFP at 2% FPR.
6. Test.
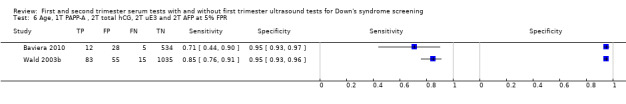
Age, 1T PAPP‐A , 2T total hCG, 2T uE3 and 2T AFP at 5% FPR.
7. Test.
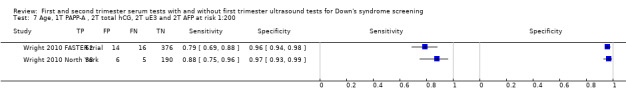
Age, 1T PAPP‐A , 2T total hCG, 2T uE3 and 2T AFP at risk 1:200.
8. Test.
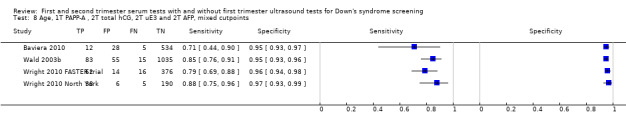
Age, 1T PAPP‐A , 2T total hCG, 2T uE3 and 2T AFP, mixed cutpoints.
9. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at 5% FPR.
10. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:50.
11. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:100.
12. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:150.
13. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:200.
14. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:250.
15. Test.

Age, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:300.
16. Test.
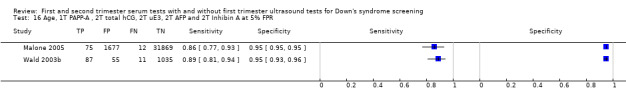
Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A at 5% FPR.
17. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:100.
18. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:150.
19. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:200.
20. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A at risk 1:250.
21. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A, mixed cutpoints.
22. Test.
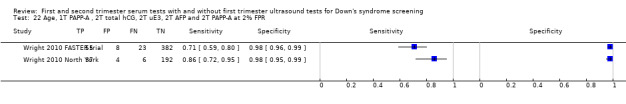
Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T PAPP‐A at 2% FPR.
23. Test.
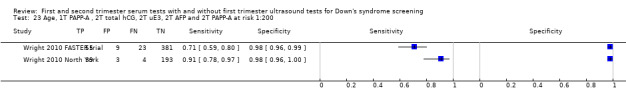
Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T PAPP‐A at risk 1:200.
24. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP, 2T Inhibin A and 2T PAPP‐A at risk 1:100.
25. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP, 2T Inhibin A and 2T PAPP‐A at risk 1:150.
26. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP, 2T Inhibin A and 2T PAPP‐A at risk 1:200.
27. Test.

Age, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP, 2T Inhibin A and 2T PAPP‐A at risk 1:250.
28. Test.
Age, 1T PAPP‐A , 1T total hCG, 2T total hCG, 2T uE3 and 2T AFP at 2% FPR.
29. Test.
Age, 1T PAPP‐A , 1T total hCG, 2T total hCG, 2T uE3 and 2T AFP at risk 1:200.
30. Test.
Age, 1T PAPP‐A , 1T uE3, 2T total hCG, 2T uE3 and 2T AFP at 2% FPR.
31. Test.
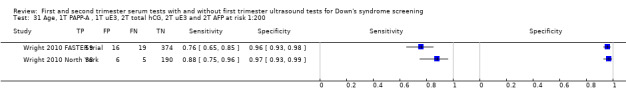
Age, 1T PAPP‐A , 1T uE3, 2T total hCG, 2T uE3 and 2T AFP at risk 1:200.
32. Test.
Age, 1T PAPP‐A , 1T total hCG, 1T uE3, 2T total hCG, 2T uE3, 2T AFP and 2T PAPP‐A at 2% FPR.
33. Test.
Age, 1T PAPP‐A , 1T total hCG, 1T uE3, 2T total hCG, 2T uE3, 2T AFP and 2T PAPP‐A at risk 1:200.
34. Test.

Age, 1T AFP, 1T free ßhCG, 1T uE3, 2T total hCG, 2T uE3 and 2T AFP at risk 1:250.
35. Test.

Age, 1T AFP, 1T free ßhCG, 1T uE3, 2T total hCG, 2T uE3 and 2T AFP at risk 1:384.
36. Test.
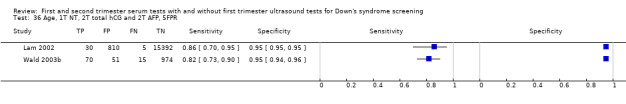
Age, 1T NT, 2T total hCG and 2T AFP, 5FPR.
37. Test.
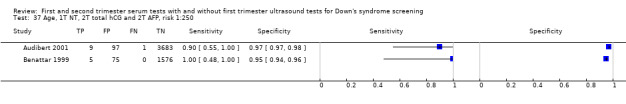
Age, 1T NT, 2T total hCG and 2T AFP, risk 1:250.
38. Test.
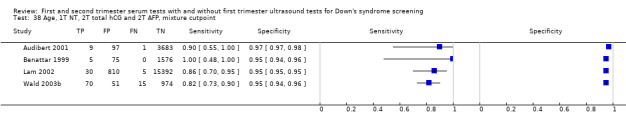
Age, 1T NT, 2T total hCG and 2T AFP, mixture cutpoint.
39. Test.
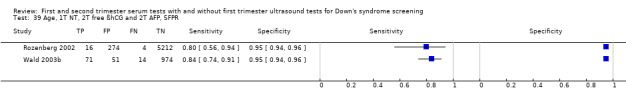
Age, 1T NT, 2T free ßhCG and 2T AFP, 5FPR.
40. Test.
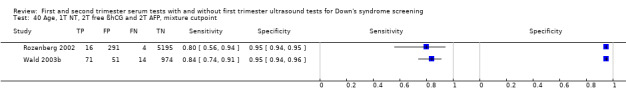
Age, 1T NT, 2T free ßhCG and 2T AFP, mixture cutpoint.
41. Test.

Age, 1T NT, 2T free ßhCG, 2T uE3 and 2T AFP, 5FPR.
42. Test.

Age, 1T NT, 2T total hCG, 2T uE3 and 2T AFP, 5FPR.
43. Test.

Age, 1T NT, 2T total hCG, 2T uE3 and 2T AFP, risk 1:250.
44. Test.
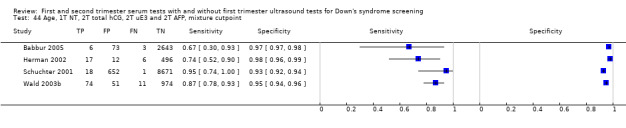
Age, 1T NT, 2T total hCG, 2T uE3 and 2T AFP, mixture cutpoint.
45. Test.

Age, 1T NT, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A, 5FPR.
46. Test.

Age, 1T NT, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A, 5FPR.
47. Test.

Age, 1T NT, 2T free ßhCG, 2T uE3, 2T AFP and 1T PAPP‐A , 5FPR.
48. Test.

Age, 1T NT, 2T free ßhCG, 2T uE3, 2T AFP and 1T PAPP‐A , risk 1:250.
49. Test.

Age, 1T NT, 1T PAPP‐A , 2T total hCG and 2T AFP, 5FPR.
50. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG and 2T AFP, 5FPR.
51. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG and 2T AFP,risk 1:250.
52. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG and 2T AFP, risk 1:300.
53. Test.

Age, 1T NT, 1T PAPP‐A, 2T total hCG, 2T uE3 and 2T AFP 5FPR.
54. Test.

Age, 1T NT, 1T PAPP‐A , 2T total hCG, 2T uE3 and 2T AFP, risk 1:200.
55. Test.
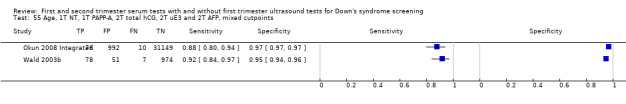
Age, 1T NT, 1T PAPP‐A, 2T total hCG, 2T uE3 and 2T AFP, mixed cutpoints.
56. Test.
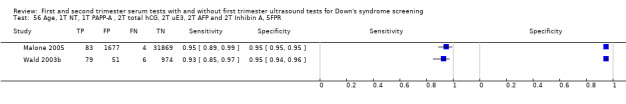
Age, 1T NT, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A, 5FPR.
57. Test.

Age, 1T NT, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A, risk 1:150.
58. Test.

Age, 1T NT, 1T PAPP‐A , 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A, mixed cutpoints.
59. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A,risk 1:300.
60. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A, 1:270.
61. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A,risk 1:250.
62. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A,risk 1:200.
63. Test.
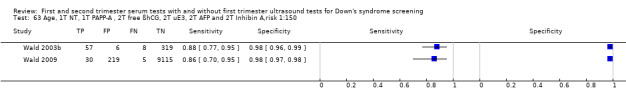
Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A,risk 1:150.
64. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A,risk 1:100.
65. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A,risk 1:50.
66. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A, 5FPR.
67. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A, 3FPR.
68. Test.

Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A, 1FPR.
69. Test.
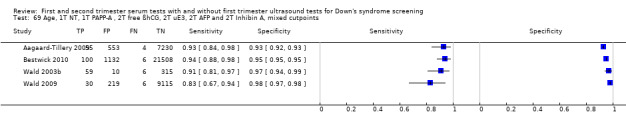
Age, 1T NT, 1T PAPP‐A , 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A, mixed cutpoints.
70. Test.

Age, 1T NT, 1T PAPP‐A, 1T free ßhCG, 2T total hCG, 2T uE3 and 2T AFP, risk 1:250.
71. Test.

Age, 1T NT, 1T PAPP‐A, 1T free ßhCG, 2T uE3, 2T AFP, 2T total hCG and 2T Inhibin A, risk 1:150.
72. Test.

ADAM 12 2T TO 1T RATIO.
73. Test.

Stepwise: Age, 1T NT, 1T PAPP‐A , 1T free ßhCG, if risk <1/30, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A risk 1:270.
74. Test.

Stepwise: Age, 1T NT, 1T PAPP‐A , 1T free ßhCG, if risk <1/30, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A risk 1:270.
75. Test.

Stepwise: Age, 1T NT, 1T PAPP‐A , 1T free ßhCG, if risk <1/30, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A 5% FPR.
76. Test.

Stepwise: Age, 1T NT, 1T PAPP‐A , if risk <1:100, 2T free ßhCG, 2T uE3, 2T AFP, risk 1:250.
77. Test.

Contingent: Age, 1T NT, 1T PAPP‐A , 1T free ßhCG, if risk 1/30‐1/1500, 2T total hCG, 2T uE3, 2T AFP and 2T Inhibin A risk 1:270.
78. Test.

Contingent: Age, 1T NT, 1T PAPP‐A , 1T free ßhCG, if risk 1/30‐1/1500, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A risk 1:270.
79. Test.

Contingent: Age, 1T NT, 1T PAPP‐A , 1T free ßhCG, if risk 1/30‐1/1500, 2T free ßhCG, 2T uE3, 2T AFP and 2T Inhibin A 5%FPR.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Aagaard‐Tillery 2009.
| Clinical features and settings | Routine screening | |
| Participants | 7842 participants who underwent both first and second trimester screening and a second trimester genetic sonogram USA ‐ The First and Second Trimester Evaluation of Risk (FASTER) trial (13 centres) Dates not specified Pregnant women Mean maternal age 30.6 years (SD 6.1 years) Singleton pregnancies 11‐13 and 15‐23 weeks' gestation |
|
| Study design | Prospective cohort study | |
| Target condition and reference standard(s) | Down's syndrome: 59 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT, PAPP‐A and free ßhCG (details not reported) Second trimester AFP, uE3, free ßhCG and inhibin‐A (details not reported) Second trimester genetic sonogram Detection rate for a 5% false positive rate and for fixed 1:270 cut‐off |
|
| Follow‐up | Details of follow‐up not reported | |
| Aim of study | To estimate the effectiveness of second trimester genetic sonography in modifying Down's syndrome screening results | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | Yes | Of 33,546 trial participants only 7842 women with complete information for all screening tests and genetic sonography were included in the study |
Audibert 2001.
| Clinical features and settings | Routine screening | |
| Participants | 4130 participants France ‐ single centre May 1994 to December 1997 Pregnant women Mean maternal age 30.1 years (all under 38 years) Singleton pregnancies 12‐13 weeks' gestation Crown rump length between 38 mm and 84 mm |
|
| Study design | Prospective consecutive series study | |
| Target condition and reference standard(s) | Down's syndrome: 12 cases Reference standards: prenatal karyotype conducted (in 7.6% of patients) depending on presence of risk > 1/125, high maternal age, parental anxiety, history of chromosomal defects or parental translocation or abnormal second trimester scan Cytogenetic testing of newborns with suspected abnormalities Postmortum on terminations of pregnancy or miscarriages Follow‐up to neonatal examination in newborns |
|
| Index and comparator tests | Maternal age First trimester NT planned at 12‐13 weeks, 3 mm cut‐off Second trimester serum hCG between 14 and 17 weeks (Amerlite, Orthoclinical diagnostics machine) Second trimester serum AFP between 14 and 17 weeks (Amerlite, Orthoclinical diagnostics machine) Serum tests in 3790 women Risk cut off 1:250 |
|
| Follow‐up | Outcome assessed at delivery and postnatal paediatric examination. 35 women were lost to follow‐up and excluded from the analysis. 340 women had first trimester NT but not second trimester serum testing | |
| Aim of study | To compare first trimester NT and second trimester maternal serum measurements as alternative methods of antenatal screening in a low risk population and to evaluate the consequence of combining the results in the estimation of risk | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | Women lost to follow‐up were excluded in the final analysis. All detected cases resulted in termination. | |
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | Yes | NT was not measured or not recorded in 219 women and these patients were excluded from the study |
| Withdrawals explained? All tests | Yes | 35 women were lost to follow‐up (they had all had normal NT results). 340 women who did not want second trimester serum screening withdrew from that part of the study |
Babbur 2005.
| Clinical features and settings | Women requesting screening (self‐paying service) and women attending on account of previous pregnancy history of fetal abnormality | |
| Participants | 3,188 participants UK ‐ Maternity Hospital August 2001 ‐ March 2004 Pregnant women Singleton pregnancies Median maternal age 37 years (range 19‐46 years) 11‐14 weeks' gestation 45 mm to 84 mm crown rump length Viable fetus |
|
| Study design | Prospective cohort study | |
| Target condition and reference standard(s) | Down's syndrome: 25 cases Reference standards: Invasive testing offered to women with NT > 3 mm or risk > 1:250 as defined by combined NT and serum results (CVS from 11 weeks, amniocentesis from 15 weeks). Rapid in situ hybridisation test in patients with risk > 1:30. No details given of any follow‐up to birth |
|
| Index and comparator tests | First trimester NT in all women (FMF methods) Second trimester serum uE3, AFP and hCG (AutoDELFIA(TM) time‐resolved fluoroimmunoassay (Perkin Elmer)) at 14 weeks. Offered to patients with negative first trimester NT (n = 2725, 85% accepted) |
|
| Follow‐up | Details of follow‐up not reported | |
| Aim of study | To determine the detection and false positive rates for trisomy 21 using 2‐stage combined NT and triple testing whilst disclosing abnormal NT measurements at the scan | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | Women with miscarriages excluded | |
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Selective testing of high risk women as done in practice |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | Yes | 463 patients having NT did not go on to have second trimester serum testing |
Baviera 2010.
| Clinical features and settings | Routine screening | |
| Participants | 579 participants: 17 cases and 562 controls matched for gestational age Italy ‐ single centre December 2006 ‐ May 2009 Pregnant women Mean maternal age 35.3 years (cases) and 30.4 years (controls) Singleton pregnancies 7‐10 and 14‐17 weeks' gestation |
|
| Study design | Case‐control study | |
| Target condition and reference standard(s) | Down's syndrome: 17 cases (14 identified by amniocentesis, 3 from follow‐up to birth) Reference standard: amniocentesis or follow‐up to birth |
|
| Index and comparator tests | Frozen serum samples tested for: First trimester and second trimester ADAM12s (time resolved fluorescence immunoassay, DELFIA assay kit, Perkin Elmer Life and Analytical Sciences) First trimester PAPP‐A (details not reported) Second trimester AFP, uE3 and hCG (details not reported) |
|
| Follow‐up | Details of follow‐up not reported | |
| Aim of study | To demonstrate the potential value of repeated measures of ADAM12s for the screening of Down's syndrome | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Benattar 1999.
| Clinical features and settings | Routine screening | |
| Participants | 1656 participants France ‐ single centre January to December 1995 Pregnant women Singleton pregnancies Mean maternal age 32 years (range 16‐46 years) Enrolled before 13 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 5 cases Reference standards: amniocentesis due to maternal age > 38 years (6.1% or women). Karyotyping encouraged for women with positive result on 1 or more index test. No details of reference standard for index test negative women |
|
| Index and comparator tests | Maternal age NT at 12‐14 weeks (Toshiba SSA 270), cut‐point 1:250 First trimester (12‐14 weeks) serum AFP and free ßhCG (Elsa AFP and Elsa free BhCG; Cis‐Bio International) Second trimester (15‐18 weeks) serum AFP and total hCG (AFP‐2T and hCG‐60; Ortho‐Clinical Diagnostics) All women had NT and serum testing |
|
| Follow‐up | Details of follow‐up not reported. 12 patients were lost to follow‐up due to miscarriages | |
| Aim of study | To evaluate the sequential combination of ultrasound screening for fetal aneuploidy at 11‐14 weeks with maternal biochemistry at 12‐14 and 15‐18 weeks of gestation | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Bestwick 2010.
| Clinical features and settings | Routine screening | |
| Participants | 22,746 participants UK ‐ 2 clinics January 2003 ‐ December 2008 Pregnant women Median maternal age 39 years (Down's syndrome) and 34 years (non‐Down's syndrome) 11‐13 and 14‐22 weeks' gestation |
|
| Study design | Retrospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 106 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT, PAPP‐A and free ßhCG (details not reported) Second trimester AFP, uE3, free ßhCG and inhibin‐A (details not reported) |
|
| Follow‐up | Data obtained from the Hospitals, the regional cytogenetic unit and the National Down Syndrome Cytogenetic Register | |
| Aim of study | To determine whether the SD of NT measurements has decreased over time and, if so, to revise the estimate and assess the effect of revising the estimate of the SD on the performance of antenatal screening for Down's syndrome | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Cuckle 2008.
| Clinical features and settings | Routine screening | |
| Participants | 36,740 participants undergoing first trimester screening (32,355 also underwent second trimester screening) USA ‐ 15 centres, FASTER trial Pregnant women Singleton pregnancies Maternal age not reported 11‐13 and 15‐18 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 116 cases (86 cases had both first trimester and second trimester screening) Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT, PAPP‐A and free ßhCG (details not reported) Second trimester AFP, total hCG, uE3 and inhibin‐A (details not reported) |
|
| Follow‐up | Details of follow‐up not reported | |
| Aim of study | To compare the contingent, step‐wise and integrated screening policies | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Goh 1996.
| Clinical features and settings | Routine screening | |
| Participants | 11,964 participants Singapore ‐ University Hospital 1989 to 1992 Pregnant women Singleton pregnancies Median maternal age 35 years (mean 33 years) 12‐22 weeks' gestation |
|
| Study design | Cohort | |
| Target condition and reference standard(s) | Down's syndrome: 34 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester and second trimester AFP and hCG (EIA method, kits from Abbot Laboratory, USA) and uE3 (In‐house indirect, extraction radioimmunoassay) Risk cut‐points of 1:250 and 1:384 |
|
| Follow‐up | No details of methods of follow‐up | |
| Aim of study | To appraise the potential effectiveness of implementing a prenatal screening programme on a local population in Singapore | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Guanciali‐Franchi 2010.
| Clinical features and settings | Routine screening | |
| Participants | 5060 participants Italy ‐ Genetic unit January 2006 ‐ April 2009 Pregnant women Mean maternal age 31.8 years Singleton pregnancies 10‐12 and 15‐17 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 13 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (by certified sonographers) First trimester PAPP‐A and free ßhCG (details not reported) Second trimester AFP, hCG and uE3 (details not reported) Cross‐trimester test: all first trimester and second trimester tests Cut‐point 1:250 |
|
| Follow‐up | Stated that follow‐up until delivery was available for all women | |
| Aim of study | To evaluate the effectiveness of cross‐trimester testing in selecting high risk pregnant women to undergo invasive prenatal diagnosis | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Habayeb 2010.
| Clinical features and settings | Routine screening | |
| Participants | 1507 participants UK ‐ Fetal medicine unit September 2007 ‐ December 2008 Pregnant women Median maternal age 35.4 years (range 18‐49 years) 9‐10, 11‐13 and > 14 weeks' gestation |
|
| Study design | Cohort | |
| Target condition and reference standard(s) | Down's syndrome: 12 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age Early first trimester PAPP‐A (9 weeks' gestation) (AutoDELFIA PAPP‐A kit, PerkinElmer LAS (UK) Ltd) First trimester NT (11‐13 weeks' gestation) (General Electric E8, Voluson 730 Pro, GE Healthcare) Second trimester AFP, free ßhCG and uE3 (at or after 14 weeks' gestation) (AutoDELFIA(TM) time‐resolved fluoroimmunoassay, PerkinElmer Life Sciences) Second trimester tests given if first trimester risk low (< 1:100) or invasive testing declined Cut‐point for second‐stage risk 1:250 |
|
| Follow‐up | Data recorded on a fetal medicine database and combined with data held on separate databases for pregnancy outcome and the regional cytogenetic laboratory. Cytogenetic test results available for all women delivering in the region | |
| Aim of study | To audit a model combining early PAPP‐A with NT and early triple test | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Herman 2002.
| Clinical features and settings | Routine screening | |
| Participants | 531 participants: 23 cases and 508 consecutive controls Israel ‐ Medical centre Pregnant women 10‐14 and 16‐19 weeks' gestation |
|
| Study design | Case‐control | |
| Target condition and reference standard(s) | Down's syndrome: 23 cases Reference standard: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT () Second trimester AFP, hCG and uE3 () |
|
| Follow‐up | Some cases obtained through follow‐up to birth. No details of follow‐up in controls reported | |
| Aim of study | To compare the results of the disclosure and non‐disclosure approaches, using the clinical data of first trimester ultrasound and second trimester serum screening tests among the same groups of normal and trisomy 21‐affected pregnancies | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Lam 2002.
| Clinical features and settings | Routine screening | |
| Participants | 16,237 participants undergoing NT and biochemical testing Hong Kong ‐ multi‐centre study 1997 to 2000 Pregnant women Mean maternal age 30.5 years (19% > 35 years) 10‐14 weeks and 15‐18 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 35 cases Reference standards: women considered high risk offered CVS (0.7%) or amniocentesis (11.8%). Follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (FMF methods) Second trimester free ßhCG and AFP (methods not reported) |
|
| Follow‐up | By review of hospital and laboratory records and by directly telephoning women. Participants who defaulted the second trimester serum tests (n = 1015) and those who miscarried after NT but before serum testing (n = 91) were excluded from the study. Outcome obtained in 15,253 patients (93.9%) | |
| Aim of study | To report data on participants undergoing both first and second trimester methods of screening to assess the relative efficacy of different methods of screening | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | Yes | NT successful in 99.8% of cases |
| Withdrawals explained? All tests | Yes | Details given for patients excluded and those without follow‐up data |
Malone 2005.
| Clinical features and settings | Routine screening | |
| Participants | 38,033 participants USA ‐ multi‐centre study (15 centres) October 1999 to December 2002 Pregnant women 21.6% of women aged ≥ 35 years Singleton pregnancies Live fetuses 10‐13 and 15‐18 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down’s syndrome: 92 cases (87 had first trimester and second trimester screening) Reference standards: amniocentesis (offered to women with positive results from any screening test) or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT in 36,306 patients (92.9%) First trimester PAPP‐A and free ßhCG in 37,843 patients (99.5%) Second trimester AFP, total hGC, uE3 and inhibin‐A in 35,236 patients (92.6%) All tests done in 33,546 patients (88.2%) |
|
| Follow‐up | Follow‐up with computerised tracking system. Medical records were reviewed in cases of 1) possible medical problem suspected 2) positive screening test results with no karyotype data, 3) 10% random sample of all enrolled patients. Follow‐up to birth complete in 36, 378 patients (97%) | |
| Aim of study | To evaluate first trimester and/or second trimester screening tools for Down's syndrome | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | Unclear which types of patients did not have follow‐up data. Appears that aborted/miscarried fetuses did not have follow‐up (note in table) | |
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | No | Not all women received a reference standard (3% had no ascertainment of pregnancy outcome, patients not excluded from study) |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | Yes | NT failed or rejected at review in 7.1% or women |
| Withdrawals explained? All tests | Yes | Details given for patients who did not undergo different index tests |
Okun 2008 Integrated.
| Clinical features and settings | Routine screening | |
| Participants | 32,227 participants undergoing integrated screening (a separate cohort evaluated for first trimester screening) January 2003 ‐ December 2005 Canada ‐ 2 hospitals Pregnant women Mean maternal age 32 years 11‐14 and 15‐18 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 86 affected cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (most sonographers had FMF certification) First trimester free ßhCG and PAPP‐A (DSX Four Plate Automated ELISA Processing system, Dynex Technologies and DPC Immulite 2000 automated immunoassay analyser, Siemens Medical Solutions Diagnostics) Second trimester hCG, AFP and uE3 (Time‐resolved fluoroimmunoassay, PerkinElmer AutoDelfia) Risk cut‐point 1:200 or NT ≥ 3.5 mm Results presented with and without adjustment for bias due to miscarriages (viability bias) |
|
| Follow‐up | From cytogenetics databases in both Hospitals, the Canadian Institute for Health Information, labour and delivery databases, written and phone follow‐up with care providers and phone follow‐up with women after birth | |
| Aim of study | To evaluate the performance of integrated prenatal screening and first trimester combined screening for trisomy 21 in a large Canadian urban centre | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | Yes | 2614 (8%) of women undergoing integrated screening did not return for the second trimester part of the test |
Palomaki 2006.
| Clinical features and settings | Routine screening | |
| Participants | 540 participants: 32 cases and 508 controls selected from same time period (within 1 month) New York ‐ General Hospital Singleton pregnancies Pregnant women Mean maternal age cases 33.9 years (SD 4.4 years) and controls 35.9 years (SD 3.6 years) 10‐13 and 14‐20 weeks' gestation |
|
| Study design | Case‐control study | |
| Target condition and reference standard(s) | Down's syndrome: 32 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age Fresh samples tested for first trimester PAPP‐A and Second trimester AFP, uE3 and hCG (PerkinElmer Life and Analytical Sciences, Woodbridge, Ontario, Canada) Frozen samples thawed and tested for second trimester inhibin‐A (Diagnostic Systems Laboratories, Webster, TX) and PAPP‐A (PerkinElmer) Cut‐points of 1:100, 1:150, 1:200 and 1:250 |
|
| Follow‐up | Outcome of pregnancy available from the Ontario Multiple Marker Screening Database | |
| Aim of study | To confirm that measuring pregnancy‐associated plasma protein‐A in both first and second trimester serum samples improves Down's syndrome screening | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | Yes | 2 cases and 6 controls had insufficient sample to measure second trimester inhibin‐A and were removed from the analysis |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Rodrigues 2009.
| Clinical features and settings | Routine screening | |
| Participants | 3299 participants: 2290 undergoing integrated and 1009 undergoing serum integrated screening Portugal ‐ screening programme March 2003 ‐ August 2007 Pregnant women Median maternal age: integrated screening 30.6 years, serum integrated screening 30.9 years First and second trimester |
|
| Study design | Retrospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 14 cases (integrated screening 8, serum integrated screening 6) Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (details not reported) First trimester PAPP‐A and second trimester free ßhCG and AFP (TRACE technology, Brahms Kryptor Systems) Risk cut‐point 1:300 for integrated and serum integrated screening |
|
| Follow‐up | Detail of follow‐up not reported | |
| Aim of study | To report an audit of an integrated and serum integrated screening programme | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Rozenberg 2002.
| Clinical features and settings | Routine screening | |
| Participants | 9118 participants France ‐ 2 tertiary and 4 primary referral centres March 1994 ‐ December 1997 Pregnant women Median age 30.5 years (18‐37 years) Singleton pregnancies 12‐14 and 14‐17 weeks' gestation |
|
| Study design | Prospective cohort | |
| Target condition and reference standard(s) | Down’s syndrome: 21 cases Reference standards: amniocentesis offered to patients with NT > 3 mm or serum marker risk was >1:250. Follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT in 98.6% of women (FMF methods) Second trimester free ßhCG (beta hCG ELISA immunoradiometric assay) and AFP (AFP ELISA immunoradiometric assay) in 91.1% of women Both NT and biochemical testing in 60.4% of women |
|
| Follow‐up | Details of follow‐up not reported. 3.4% of patients were lost to follow‐up and were excluded from the study. This included 113 women (1.2%) with miscarriages | |
| Aim of study | To assess the performance of combined first trimester sonographic screening and second trimester serum screening | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | Includes cost effectiveness analysis | |
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | Yes | NT was not able to be measured in 93 women (1.5%) |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Schuchter 2001.
| Clinical features and settings | Routine screening | |
| Participants | 9342 participants Austria ‐ single institution January 1994 to December 1998 Pregnant women Mean maternal age 28 years (range 15‐46 years), 10.7% ≥ 35 years 10‐13 weeks' gestation |
|
| Study design | Retrospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 19 cases Reference standards: CVS (offered to patients with first trimester NT > 3.5 mm), amniocentesis (offered to patients with first trimester NT 2.5‐3.4, high risk on second trimester serum testing (> 1:250) and those > 35 years) or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (all women) (5‐MHz transducer, Acuson Corp) Second trimester AFP, E2 and hGC (triple test) offered to patients not undergoing first trimester invasive testing (99.7% of women) (AMERLEX‐M 2nd Trimester kits, Ortho Clinical Diagnostics) |
|
| Follow‐up | Patients included in study if they were delivered in the same hospital where they were screened. It is stated that all newborns were examined for malformations by a paediatrician after delivery | |
| Aim of study | To evaluate screening for trisomy 21 in a low risk population utilising a combination of NT measurement in the first trimester and the triple test in the second trimester | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | Women having miscarriages were excluded from the study | |
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | Details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Wald 2003b.
| Clinical features and settings | Routine screening | |
| Participants | 606 participants: 101 cases, 505 controls matched for gestation, duration of storage and centre UK and Austria ‐ multi‐centre trial September 1996 to April 2000 Pregnant women 9‐13 and 14‐20 weeks' gestation |
|
| Study design | Case‐control study | |
| Target condition and reference standard(s) | Down's syndrome: 101 cases Rerence standards: invasive testing (following second trimester screening) or follow‐up to birth |
|
| Index and comparator tests | First trimester NT (midsagittal section, optimal magnification of thickness of translucent space between inner skin surface and fascia covering cervical spine (white black interface (outer) ‐ black white interface (inner), 41 models of ultrasound machine, 20 minutes allotted scanning time) First trimester and second trimester serum AFP, hCG, UE3, PAPPA, free ßhCG (time resolved fluoroimmunoassay, AutoDELFIA) First trimester and second trimester inhibin A (Sandwich enzyme linked immunosorbent assay, Oxford Bioinnovation) First trimester and second trimester urinary beta core fragment, total‐hCG, ITA and free ßhCG (ITA and beta core fragment, Quest diagnostics USA) |
|
| Follow‐up | Follow‐up by: 1) Staff at local hospitals completed a study outcome form at, or just after. delivery, 2) Study records of CVS, amniocentesis or karyotype at birth linked to information from cytogenic laboratories, 3) Study records linked to records of cases of Down's syndrome from the National Down's Syndrome Cytogenetic Register, 4) Information obtained from local obstetrical outcome records, 5) Forms sent to all women with a request to return details of the outcome of their pregnancy, 6) Individual searches in respect of women whose outcomes of pregnancy had not been obtained by any of the previous methods. 96% Birth/Karyotype full outcome documentation obtained | |
| Aim of study | To identify the most effective, safe and cost effective strategy for antenatal screening for Down's syndrome using NT, maternal serum and urine markers in the first and second trimesters of pregnancy and maternal age in various combinations | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | Performance of screening assessed at 17 weeks' gestation. Study tried to be non‐interventional in the first trimester ‐ second trimester testing was aimed to be used as the basis for any referral for invasive testing | |
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | 4% of total patient cohort did not have a documented outcome of pregnancy. Unclear if any of these included in nested case‐control study |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | Yes | Rates of NT failure on average 9%. Pre‐10 weeks' gestation, > 33% failure rate, declined to 7% at 12 weeks |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Wald 2009.
| Clinical features and settings | Routine screening | |
| Participants | 14,296 participants in whom screening for all markers were measured UK ‐ 2 Hospitals 2003 ‐ 2007 (2004 ‐ 2007 for 1 hospital) Pregnant women Singleton pregnancies Median maternal age 33 years (range 15‐51 years), 20% ≥ 37 years 10‐13 and 14‐22 weeks' gestation |
|
| Study design | Retrospective cohort | |
| Target condition and reference standard(s) | Down's syndrome: 47 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (details not reported) First trimester PAPP‐A (details not reported) Second trimester AFP, uE3, hCG, free ßhCG and, at one hospital, inhibin‐A (details not reported) Integrated test (at 1 of the hospitals women were given the option of having only the combined test and earlier test results) Cut‐point 1:150 |
|
| Follow‐up | Down's syndrome pregnancies, including those missed by screening, were ascertained from hospital records, cytogenetic laboratories and by linking data with the National Down Syndrome Cytogenetics Register | |
| Aim of study | To present a medical audit of screening using the Integrated test at 2 hospitals | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Unclear | Unclear if all women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Wright 2010 FASTER trial.
| Clinical features and settings | Routine screening | |
| Participants | 468 participants: 78 cases and 390 controls matched for gestational and maternal age, ethnicity and storage duration The First and Second Trimester Evaluation of Risk (FaSTER) dataset USA ‐ 15 screening centres October 1999 ‐ December 2002 Pregnant women Singleton pregnancies 11‐13 and 15‐18 weeks' gestation |
|
| Study design | Case‐control study | |
| Target condition and reference standard(s) | Down's syndrome: 78 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age First trimester NT (details not reported) Fresh samples tested for: First trimester PAPP‐A (details not reported) Second trimester AFP, uE3, hCG and inhibin A (details not reported) Frozen serum samples tested for: First trimester hCG and uE3 (details not reported) Second trimester PAPP‐A (details not reported) Frozen samples tested blind to other results and pregnancy outcome |
|
| Follow‐up | Details not reported | |
| Aim of study | To provide estimates and confidence intervals for the performance (detection and false positive rates) of screening for Down's syndrome using repeated measures of biochemical markers from first and second trimester maternal serum samples taken from the same woman | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
Wright 2010 North York.
| Clinical features and settings | Routine screening | |
| Participants | 239 participants: 43 cases and 196 controls (35 cases and 173 controls with second trimester testing) matched for maternal and gestational age and sample date USA ‐ The North York General Hospital dataset December 1999 ‐ November 2007 Pregnant women Singleton pregnancies 11‐13 and 14‐20 weeks' gestation |
|
| Study design | Case‐control study | |
| Target condition and reference standard(s) | Down's syndrome: 43 cases Reference standards: karyotyping or follow‐up to birth |
|
| Index and comparator tests | Maternal age Fresh samples tested for: First trimester PAPP‐A (PerkinElmer) Second trimester AFP, uE3, and hCG (PerkinElmer) Frozen serum samples tested for: First trimester hCG and uE3 (details not reported) Second trimester PAPP‐A (details not reported) Frozen samples tested blind to other results and pregnancy outcome |
|
| Follow‐up | Details not reported | |
| Aim of study | To provide estimates and confidence intervals for the performance (detection and false positive rates) of screening for Down's syndrome using repeated measures of biochemical markers from first and second trimester maternal serum samples taken from the same woman | |
| Test characteristics | ||
| Reference standard used | ||
| Notes | ||
| Table of Methodological Quality | ||
| Item | Authors' judgement | Description |
| Representative spectrum? All tests | Yes | Routine screening of typical pregnant population |
| Acceptable reference standard? All tests | Yes | Karyotyping or follow‐up to birth |
| Partial verification avoided? All tests | Yes | All women received a reference standard |
| Differential verification avoided? All tests | No | Choice of reference standard depended on index test results |
| Incorporation avoided? All tests | Yes | Reference standard was independent of the index test |
| Reference standard results blinded? All tests | No | Reference standard interpreted with knowledge of index test results |
| Index test results blinded? All tests | Yes | Index test interpreted without knowledge of reference standard results |
| Relevant clinical information? All tests | Yes | Information available as would be in standard clinical practice |
| Uninterpretable results reported? All tests | No | No details given for test failures/uninterpretable measurements |
| Withdrawals explained? All tests | No | No details of withdrawals given |
AFP: alpha‐fetoprotein ß hCG: beta human chorionic gonadotrophin CVS: chorionic villus sampling ELISA: enzyme‐linked immunosorbent assay hCG: human chorionic gonadotrophin NT: nuchal translucency PAPP‐A: pregnancy‐associated plasma protein‐A SD: standard deviation uE3: unconjugated oestriol
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Aagaard‐Tillery 2010 | Results presented in another study |
| Abbas 1995 | Unable to extract useful data |
| Abdul‐Hamid 2004 | No Down's syndrome pregnancies |
| Abraha 1999 | Unable to extract useful data |
| Abu‐Rustum 2010 | Not Down’s syndrome specific |
| Achiron 2010 | Study only includes cases of Down’s syndrome |
| Adekunle 1999 | Unable to extract useful information |
| Aitken 1993 | Unable to extract useful data |
| Aitken 1996 | Fewer than 80% of pregnancies had gestational age confirmed by USS |
| Aitken 1996a | Fewer than 80% of pregnancies had gestational age confirmed by USS |
| Ajayi 2011 | No diagnostic data |
| Akbas 2001 | Less than 5 Down's syndrome pregnancies |
| Alexioy 2009 | Study only includes test positives |
| Allingham‐Hawkins 2011 | Quantitative fluorescent polymerase chain reaction study |
| American College 2009 | Discussion article |
| Antona 1998 | Likely fewer than 80% of pregnancies dated by USS |
| Antsaklis 1999 | Women screened at greater than 24 weeks' gestation |
| Anuwutnavin 2009 | Second trimester ultrasound |
| Ashwood 1987 | Unable to extract useful data |
| Asrani 2005 | Review article |
| Audibert 2001b | Unable to ascertain whether part of screening population in Rozenberg et al. No response from authors therefore excluded to reduce risk of data replication |
| Axt‐Fleidner 2006 | Unable to extract useful data |
| Azuma 2002 | Unable to extract useful data |
| Baghagho 2004 | Unable to obtain paper |
| Bahado‐Singh 1995 | USS markers greater than 14 weeks' gestation |
| Bahado‐Singh 1996 | USS markers greater than 14 weeks' gestation |
| Bahado‐Singh 1999 | USS markers greater than 14 weeks' gestation |
| Bahado‐Singh 2002 | USS markers greater than 14 weeks' gestation |
| Bahado‐Singh 2003 | Review article |
| Ball 2007 | Data from the FASTER trial |
| Bar‐Hava 2001 | No Down's pregnancies in study population |
| Barkai 1996 | No Down's pregnancies in study population |
| Barnabei 1995 | No Down's pregnancies in study population |
| Bartels 1988 | Unable to extract useful data |
| Bartels 1993 | No Down's pregnancies in study population |
| Barth 1991 | Second trimester ultrasound study |
| Bas‐Budecka 2007 | No diagnostic data |
| Baviera 2004 | Unclear method of confirmation of gestational age |
| Bazzett 1998 | Male versus female fetuses |
| Beke 2008 | Results are not specific to Down’s syndrome |
| Bellver 2005 | No Down's syndrome pregnancies in study |
| Benn 1995 | Less than 80% follow‐up |
| Benn 1996 | Less than 80% follow‐up |
| Benn 1997 | No Down's pregnancies in study population |
| Benn 1998 | Less than 80% follow‐up |
| Benn 2001 | Statistical modelling (computer simulation) |
| Benn 2002 | Modelled data |
| Benn 2003 | Less than 80% of pregnancies dated by USS |
| Benn 2003a | Editorial |
| Benn 2005 | No Down's pregnancies included |
| Benn 2005a | Mathematical model |
| Benn 2007 | No follow‐up information |
| Berry 1995 | Less than 80% of pregnancies USS dated |
| Berry 1997 | Less than 80% of pregnancies USS dated |
| Bersinger 1994 | Gestational age not USS estimated |
| Bersinger 2000 | Unable to extract useful data |
| Bersinger 2001 | No Down's syndrome pregnancies in study population |
| Bersinger 2003 | Unable to extract useful data |
| Bersinger 2004 | No Down's syndrome pregnancies in study population |
| Bersinger 2005 | No Down's syndrome pregnancies in study population |
| Bestwick 2008 | All healthy pregnancies |
| Biggio 2004 | Cost‐effectiveness analysis |
| Bilardo 2011 | Not a proper sample ‐ most had elevated NT |
| Bindra 2002 | Review article |
| Blundell 1999 | Unable to extract useful data |
| Boormans 2010 | Study of testing on amniocentesis samples |
| Boots 1989 | Population risk factor calculations |
| Bornstein 2009a | No diagnostic data |
| Bornstein 2009b | No diagnostic data |
| Bornstein 2010 | No diagnostic data |
| Borowski 2007 | No diagnostic data |
| Borrell 2007 | No follow‐up data |
| Borrell 2009 | Based on SURUSS (Serum, Urine and Ultrasound Screening Study) data ‐ second trimester serum parameters not actually measured |
| Borruto 2002 | Unable to extract useful data |
| Bottalico 2009 | Second trimester ultrasound |
| Boue 1990 | Review article |
| Bradley 1994 | Screen negative population gestations not confirmed by ultrasound |
| Braithwaite 1996 | Review article |
| Brambati 1995 | USS screening inclusive of women greater than 14 weeks' gestation |
| Brambati 1996 | Review article |
| Brizot 1995 | Unable to extract useful data |
| Brizot 1995a | Unable to extract useful data |
| Brizzi 1989a | Second trimester ultrasound |
| Brock 1990 | Unable to extract useful data |
| Calda 2010 | No data for false positive rates |
| Campogrande 2001 | Unable to extract useful data |
| Canick 1988 | Unable to extract useful data |
| Canick 1995 | Unable to extract useful data |
| Canini 2002 | No Down's syndrome pregnancies in study population |
| Cans 1998 | Second trimester ultrasound |
| Carreras 1991 | Second trimester ultrasound |
| Caughey 2007 | No diagnostic data |
| Cebesoy 2008 | No diagnostic data |
| Chelli 2008 | No follow‐up for false negatives |
| Chen 1999 | Review article |
| Chen 2002 | No Down's syndrome pregnancies in study population |
| Chen 2004 | Less than 5 Down's cases in study population |
| Chen 2005 | Unable to extract useful data |
| Chen 2008 | No diagnostic data |
| Cheng 1993 | Likely that fewer than 80% of gestational age confirmed by USS |
| Cheng 1999 | Case series No Down's syndrome pregnancies in study population |
| Cheng 2004a | No Down's syndrome pregnancies in study population |
| Cheng 2004b | No Down's syndrome pregnancies in study population |
| Chitayat 2002 | Less than 5 Down's cases in study population |
| Chiu 2011 | Study of maternal DNA testing |
| Cho 2009 | Study of testing amniotic fluid |
| Chou 2009 | Not possible to calculate specificity |
| Christiansen 2002 | Unable to extract useful data |
| Christiansen 2007 | Unable to extract useful data |
| Christiansen 2008 | No diagnostic data |
| Chung 2000 | Less than 5 Down's syndrome pregnancies in study population |
| CNGOF 1996 | Unable to obtain translation |
| Cocciolone 2008 | Unable to extract useful data ‐ attempted to contact author |
| Cole 1996 | Review article |
| Comas 2001 | USS at greater than 14 weeks |
| Comas 2002a | USS at greater than 14 weeks |
| Comas 2002b | USS at greater than 14 weeks |
| Comstock 2006 | Unable to extract useful data |
| Conde 1998 | Review article |
| Cowans 2011 | No diagnostic data |
| Crossley 1991 | Less than 80% of pregnancies had gestational age confirmation by ultrasound |
| Crossley 1993 | Less than 80% of pregnancies had gestational age confirmation by ultrasound |
| Crossley 1996 | No Down's syndrome pregnancies in study population |
| Crossley 2002 | Adjustment factors for smokers |
| Cuckle 1984b | Gestational age not confirmed by USS |
| Cuckle 1987a | Gestational age not confirmed by USS |
| Cuckle 1987b | No gestational age limits given |
| Cuckle 1990 | Paper presenting adjustment factors |
| Cuckle 1996a | Data modelled on 4 meta‐analysed studies |
| Cuckle 1999b | Unable to extract useful data |
| Cuckle 1999c | Review article |
| Cullen 1990 | Abnormal scans only in study population |
| Cusick 2004 | Less than 5 Down's syndrome pregnancies in study population |
| Cusick 2007 | ST ultrasound |
| Dancoine 2001 | No Down's syndrome pregnancies in study population |
| Dane 2008 | Not specific to Down’s syndrome |
| De Biasio 2000 | Unable to extract useful information |
| De Biasio, 1999 | Unable to ascertain whether overlapping populations between several papers ‐ attempted to contact author with no response |
| De Biasio, 2001 | Unable to ascertain whether overlapping populations between several papers ‐ attempted to contact author with no response |
| De Graaf 1991 | Unable to extract useful data |
| De Graaf 1999 | Modelled data |
| Del Carmen Saucedo 2009 | No follow‐up information |
| DeVore 2001 | Second trimester ultrasound |
| Dhaifalah 2007a | Unable to obtain translation |
| Dhaifalah 2007b | Unable to obtain translation |
| Dhallan 2007 | DNA testing of blood samples from parents |
| Dickerson 1994 | Comment |
| Dimaio 1987 | Gestational age by USS only in screen positive population |
| Doran 1986 | Ultrasound confirmation of gestational age performed in screen positive women only |
| Dreux 2008 | No information for specificity |
| Drugan 1996a | Second trimester ultrasound |
| Drugan 1996b | Unable to extract useful data |
| Drysdale 2002 | Fewer than 5 Down's syndrome pregnancies in population |
| Dugoff 2008 | Not specific to Down’s syndrome |
| Ebell 1999 | Review article |
| Economides 1998 | Unable to extract useful data |
| Erickson 2004 | No Down's syndrome pregnancies in population |
| Evans 1996 | No Down's syndrome pregnancies in population |
| Evans 2007 | Data previously presented in another study |
| Falcon 2005 | Unable to extract useful data |
| Falcon 2006 | Unable to extract useful data |
| Ford 1998 | Audit |
| Frishman 1997 | No Down's syndrome pregnancies in population |
| Fukada 2000 | Unable to extract useful data |
| Gaudry 2009 | Study of karyotyping |
| Gebb 2009 | Study only examines screen positives |
| Geerts 2008 | Study only examines abnormal foetuses |
| Geipel 2010 | ST ultrasound |
| Gekas 2009 | Diagnostic data from other studies |
| Gekas 2011a | Diagnostic data from other studies |
| Gekas 2011b | Diagnostic parameters from other studies |
| Gerovassili 2007 | No diagnostic data |
| Ghidini 1998 | Comparison of male versus female fetuses |
| Goetzinger 2010 | Second trimester ultrasound |
| Goldie 1995 | Fewer than 80% of study population and gestational age confirmed by USS |
| Gollo 2008 | Only 1 case of Down’s syndrome |
| Gonçalves 2004 | Greater than 14 weeks USS screening |
| Goodburn 1994 | Likely that fewer than 80% of pregnancies had gestational age estimated by USS |
| Gorduza 2007 | Study of FISH technique |
| Grace 2010 | ST ultrasound |
| Grati 2010 | No diagnostic data |
| Gray 2009 | ST ultrasound |
| Gregor 2007 | Unable to obtain translation |
| Gregor 2009 | Unable to obtain translation |
| Grether 2009 | Systematic review and guidelines |
| Grozdea 2002 | Unable to extract useful data |
| Guo 2010 | Study of fetal samples |
| Gyselaers 2004a | Less than 80% follow‐up |
| Gyselaers 2004b | Less than 80% follow‐up |
| Gyselaers 2006a | Unaffected pregnancies only |
| Gyselaers 2006b | Unable to extract useful data |
| Hackshaw 1995 | No Down's syndrome pregnancies in population |
| Hackshaw 2001 | No Down's syndrome pregnancies in population |
| Haddow 1992 | Less than 80% of pregnancies had gestational age confirmed by ultrasound scan |
| Hadzsiev 2007 | Study of FISH technique |
| Hafner 1995 | Less than 5 Down's pregnancies in study population |
| Hallahan 1998 | Gestational age greater than 24 weeks |
| Han 2008 | Study of findings on amniocentesis |
| Harper 2010 | Second trimester ultrasound |
| Harrison 2006 | Less than 80% of pregnancies had gestational age confirmed by ultrasound scan |
| Harry 2006 | Editorial |
| Hayashi 1995 | Unable to extract useful data |
| Hayashi 1996 | Less than 5 Down's pregnancies in study population |
| Heikkila 1997 | Fewer than 80% of pregnancies had gestational age confirmed by USS |
| Heinig 2007 | No Down’s syndrome data |
| Heinonen 1996 | No Down's syndrome pregnancies in population |
| Herman 2000 | No Down's syndrome pregnancies in study population |
| Herman 2003 | Correlation between markers, not evaluation of screening tests |
| Herrou 1992 | Unable to extract useful data |
| Hershey 1985 | Gestation unclear |
| Hershey 1986 | Gestation based on LMP |
| Hewitt 1993 | Unable to extract useful data |
| Hills 2010 | Study of testing on CVS and amniocentesis samples |
| Ho 2010 | Study of FISH diagnosis |
| Hogdall 1992 | Unclear method of determination of gestational age Unable to extract useful data |
| Hong Kong Practitioner | CME |
| Hoogendoorn 2008 | Diagnostic data from other studies used |
| Howe 2000 | Second trimester ultrasound scans |
| Hsiao 1991 | Unable to obtain translation |
| Hsieh 1999 | No Down's syndrome pregnancies in study population |
| Hsu 1997a | Adjustment factors |
| Hsu 1998a | No Down's syndrome pregnancies in study population |
| Hsu 1999b | No Down's pregnancies |
| Hu 2007 | Same data as Liu 2010 |
| Huang 2003 | No Down's syndrome pregnancies in study population |
| Huang 2007a | Not possible to obtain detection rate |
| Huang 2007b | No diagnostic data |
| Huggon 2004 | Study of cardiac function in pregnancies with normal and abnormal NT results |
| Hui 2003 | No Down's syndrome pregnancies in population |
| Hui 2005 | No Down's syndrome pregnancies in population |
| Hultén 2004 | Editorial/commentary |
| Hung 2003 | Modelling |
| Hung 2008 | Second trimester ultrasound |
| Hurley 1993 | Unable to extract useful data |
| Huttly 2004 | No Down's syndrome pregnancies in population |
| Hwa 2004 | Less than 5 Down's pregnancies in population |
| Iles 1996 | Review |
| Ind 1994 | Unable to extract useful data |
| Ivorra‐Deleuze 2010 | No diagnostic data |
| Jakobsen 2011 | Not Down’s syndrome specific |
| Jean‐Pierre 2005 | Review article |
| Johnson 1991 | Gestatiojnal age estimated by USS in fewer than 80% of cases |
| Johnson 1993 | Normal pregnancies only |
| Jorgensen 1999 | Gestation greater than 14 weeks for USS |
| Jorgez 2007 | Study of DNA testing on maternal blood |
| Josefsson 1998 | No Down's syndrome pregnancies in study population |
| Jou 2001 | Less than 5 Down's syndrome pregnancies in study population |
| Jun‐Tao 2003 | Unable to obtain translation |
| Jung 2007 | ST ultrasound |
| Kagan 2006 | Screen positive pregnancies only |
| Kagan 2007 | No diagnostic data |
| Kagan 2008 | Not Down’s syndrome detection |
| Kalelioglu 2007 | ST ultrasound |
| Kautzmann 1995 | Fewer than 80% pregnancies had gestational age estimated by USS |
| Kazerouni 2009 | Not possible to obtain complete diagnostic data |
| Keith 1992 | Summary article |
| Kelekci 2004 | Less than 5 Down's syndrome pregnancies in population |
| Kellner 1995a | Less than 5 Down's syndrome pregnancies in population |
| Kellner 1995b | Less than 80% follow‐up Unable to ascertain proportion of population with gestational age confirmed by USS |
| Kellner 1997 | Assumption of normal karyotype without reference standard in significant proportion of control pregnancies |
| Kirkegaard 2008 | FPR only calculated for subset of the cohort |
| Kjaergaard 2008 | Unable to obtain translation |
| Knight 1990 | Review article |
| Knight 2001 | Validation of a specific assay |
| Knight 2005 | Less than 80% of pregnancies had gestational age confirmed by ultrasound scan |
| Koos 2006 | Review article |
| Kornman 1996 | Less than 5 Down's syndrome pregnancies in population |
| Kornman 1997 | Unable to extract useful information |
| Kotaska 2007 | No new data |
| Kramer 1998 | No Down's syndrome pregnancies in study population |
| Krantz 1996 | Modelled data |
| Krantz 2005 | Adjustment factor |
| Krantz 2007 | Uses data from other published studies |
| Kulch 1993 | No Down's cases in population |
| Lai 1998 | Modelled population |
| Lai 2003 | No Down's syndrome pregnancies in study population |
| Laigaard 2006a | Unable to extract useful data |
| Laigaard 2006b | Simulation |
| Lam 1997 | Unable to extract useful data |
| Lam 1998 | Fewer than 80% pregnancies had gestational age estimated by USS |
| Lam 1999a | No Down's syndrome pregnancies in population |
| Lam 1999b | Unable to extract useful data |
| Lam 2000 | Study of women's decisions about screening |
| Lam 2001 | Male versus female fetuses |
| Lambert‐Messerlian 1996 | Fewer than 80% of pregnancies USS dated |
| Lambert‐Messerlian 1998 | Unable to extract useful data |
| Lauria 2007 | No diagnostic data |
| Lehavi 2005 | Down's syndrome pregnancies only |
| Leung 2006 | Unable to separate twins from singletons therefore unable to extract useful data |
| Leymarie 1993 | Appears to be a review article (French) |
| Li 1998 | Unable to obtain translation |
| Li 1999 | Unable to obtain translation |
| Li 2010 | No diagnostic data |
| Liao 1997 | Unable to obtain translation |
| Liao 2001 | Unable to extract useful data |
| Lim 2002 | Second trimester ultrasound |
| Lippman 1987 | Editorial |
| Liu 2010 | Not possible to separate out data for cases of Down’s syndrome |
| Lo 2010 | Pooled test results |
| Lustig 1988 | Gestational age by LMP only |
| Luthgens 2008 | FPR and DR obtained from different cohorts |
| MacDonald 1991 | Fewer than 80% of gestational ages estimated by USS |
| Macintosh 1994 | Unable to extract useful data |
| Macintosh 1997 | Unable to extract useful data |
| MacRae 2010 | Pooled test results |
| Macri 1994 | Likely fewer than 80% evaluated for gestational age by ultrasound examination |
| Macri 1996 | Likely fewer than 80% evaluated for gestational age by ultrasound examination |
| Malone 1998 | Review article |
| Malone 2003 | Review article |
| Mandryka‐Stankewycz 2009 | No diagnostic data |
| Mangione 2001 | Abnormal screening results only |
| Markov 2008 | Unable to obtain paper |
| Maymon 2001a | No Down's syndrome pregnancies in study population |
| Maymon 2001b | No normal test results included therefore unable to extract meaningful data |
| Maymon 2002 | No Down's syndrome pregnancies in study population |
| Maymon 2004 | No Down's syndrome pregnancies in study population |
| Maymon 2005 | Modelled data |
| McDuffie 1996 | USS dating on screen positive women only |
| Meier 2002 | Observed versus expected cases of Down's syndrome in a population |
| Merkatz 1984 | Gestational age not confirmed by ultrasound scan |
| Merz 2005 | Editorial |
| Merz 2008 | First trimester only |
| Metzenbauer 2001 | Normal pregnancies only |
| Metzenbauer 2002 | Unable to extract useful data |
| Mikic 1999 | No Down's syndrome pregnancies in study population |
| Miller 1991 | Unable to extract useful data |
| Milunsky 1989 | Fewer than 80% gestational age estimated by USS |
| Milunsky 1996 | Fewer than 80% gestational age estimated by USS |
| Minobe 2002 | Gestational age greater than specified limits |
| Miron 2008 | No diagnostic data |
| Miron 2009 | No diagnostic data |
| Miron 2010 | No diagnostic data |
| Miyamura 1999 | Unable to extract useful data |
| Moghadam 1998 | Unable to extract useful data |
| Monni 2000 | Less than 5 Down's syndrome pregnancies |
| Monni 2002 | Review article |
| Mooney 1994 | Greater than 24 weeks' gestation |
| Muhcu 2008 | No diagnostic data |
| Muller 1994 | No Down's syndrome pregnancies in study population |
| Muller 1996 | Unable to extract useful data |
| Muller 1999 | Unable to extract useful data |
| Muller 2002a | Gestational age greater than 24 weeks |
| Muller 2002b | Unable to extract meaningful data ‐ unable to separate double and triple test data |
| Muller 2003 | No Down's syndrome pregnancies in study population |
| Murta 2002 | Unable to extract useful data |
| Musone 2000 | Unable to extract useful data |
| Musto 1986 | Fewer than 80% USS dated |
| Myrick 1990 | Unable to extract useful data |
| Naidoo 2008 | Not specific Down’s syndrome results |
| Nau 2009 | No diagnostic data |
| Nau 2009a | No diagnostic data |
| Neveux 1996a | No Down's syndrome pregnancies in population |
| Neveux 1996b | Unable to extract useful data |
| Ng 2004 | Unable to extract useful data |
| Nicolaides 1992 | Study of outcomes of abnormal NT results |
| Nicolaides 2000 | Review article |
| Nicolaides 2004 | Review article |
| Nicolaides 2005a | Unable to obtain translation ‐ appears to be a review article |
| Nicolaides 2005b | Unable to obtain translation ‐ appears to be a review article |
| Nicolaides 2005c | Unable to obtain translation ‐ appears to be a review article |
| Nicolaides 2005d | Unable to obtain translation ‐ appears to be a review article |
| Nicolaides 2005e | Unable to obtain translation ‐ appears to be a review article |
| Nicolaides 2005f | Review article |
| Niemimaa 2001 | No Down's pregnancies in study population |
| Niemimaa 2002 | No Down's syndrome pregnancies in population |
| Niemimaa 2003 | No Down's syndrome pregnancies in population |
| Noble 1997 | Unable to extract useful data |
| Norgaard 1990 | Less than 80% of gestational ages confirmed by USS |
| Norton 1992 | Unable to extract useful data |
| Novakov‐Mikic 2007 | Out of FT screening time frame |
| O'Brien 1997a | No Down's syndrome pregnancies in population |
| O'Brien 1997b | No Down's syndrome pregnancies in population |
| Odibo 2004 | Gestational age of greater than 14 weeks in USS population |
| Odibo 2007 | ST ultrasound |
| Odibo 2008 | ST ultrasound |
| Odibo 2009 | No results presented |
| Offerdal 2008 | ST ultrasound |
| Ognibene 1999 | Unable to extract useful data |
| Oh 2007 | No diagnostic data |
| Olajide 1989 | Unable to extract useful data |
| Onda 1996 | Unable to extract useful data |
| Onda 1998 | Unable to extract useful data |
| Onda 2000 | Less than 80% follow‐up |
| Orlandi 2002 | No Down's syndrome pregnancies in study population |
| Ottavio 1997 | Second trimester USS |
| Ozkaya 2010 | Only healthy pregnancies |
| Paladini 2007 | No diagnostic data |
| Palka 1998 | Twin data used in calculation of the median |
| Palomaki 1989 | Fewer than 80% USS dated |
| Palomaki 1993 | No Down's syndrome pregnancies in population |
| Palomaki 1994 | No Down's syndrome pregnancies in population |
| Palomaki 1996 | Meta‐analysis |
| Palomaki 2005 | Unable to extract meaningful data |
| Panburana 2001 | Less than 5 Down's syndrome pregnancies in population |
| Pandya 1994 | Study of outcomes of abnormal NT results |
| Pandya 1995b | Review article |
| Papadopoulou 2008 | No diagnostic data |
| Parra‐Cordero 2007 | ST ultrasound |
| Paterlini‐Brechot 2007 | Editorial, no new data |
| Paul 2001 | Unable to extract useful data |
| Peralta 2005 | Unable to extract useful data |
| Perenc 1998 | No Down's syndrome pregnancies in study population |
| Perheentupa 2002 | No Down's syndrome pregnancies in population |
| Perona 1998 | Smokers versus non smokers |
| Persico 2008 | ST ultrasound |
| Petervari 2000 | Unable to extract useful data |
| Petrocik 1989 | Likely fewer than 80% USS dated |
| Phillips 1992 | Gestational age confirmed by USS in less than 80% of population |
| Phillips 1993 | Gestational age confirmed by USS in less than 80% of population |
| Pihl 2008 | Only 2 cases of Down's syndrome |
| Pinette 2003 | Women screened prior to recruitment |
| Platt 2004 | Unable to extract useful data |
| Podobnik 1995 | Abnormal results only |
| Poon 2009 | No diagnostic data |
| Prefumo 2002 | Comparison of prevalence and prediction |
| Prefumo 2004 | Comparison of a marker in women of different ethnic origins |
| Price 1998 | Unable to extract useful data |
| Páez 2004 | Unable to obtain translation |
| Rembouskos 2004 | Unable to extract useful data |
| Ren 1992 | Review article |
| Renier 1998 | Method of ascertainment of gestational age unclear Twin gestations included in general population |
| Resta 1990 | Second trimester USS |
| Reynders 1997 | Fewer than 5 Down's cases |
| Reynolds 1989 | Explanation of mathematical techniques |
| Reynolds 1999 | Unable to extract useful data |
| Reynolds 2008 | Not full diagnostic data |
| Ribbert 1996 | No Down's syndrome pregnancies in study population |
| Rice 2005 | Down's syndrome pregnancies excluded from study |
| Rich 1991 | Unable to extract useful data |
| Roberts 1995 | No Down's syndrome pregnancies in study population |
| Robertson 1991 | Editorial |
| Rode 2003 | No Down's pregnancies |
| Ronge 2006 | Editorial ‐ summary of FASTER results |
| Rose 1995 | Review article |
| Ross 1997 | Review article |
| Rotmensch 1996 | Unable to extract useful data |
| Rotmensch 1999 | No Down's syndrome pregnancies in study population |
| Rozenberg 2006 | USS greater than 14 weeks' gestation |
| Rudnicka 2002 | No Down's syndrome pregnancies in population |
| Ryall 1992 | Unable to determine method of confirmation of gestational age |
| Ryall 2001 | High‐risk results only included (i.e. no screen negative group for comparison) |
| Räty 2000 | No Down's syndrome pregnancies in population |
| Räty 2002 | No Down's pregnancies in population |
| Sabriá 2002 | Unable to ascertain how numbers calculated and from which populations |
| Sacchini 2003 | Unable to extract useful data |
| Sahota 2009 | No diagnostic data |
| Sahota 2010 | Included in Sahota 2010 |
| Salazar 2007 | Unable to obtain paper |
| Salazar 2008 | Only 1 case of Down’s syndrome |
| Saller 1997 | Down's syndrome secondary to Robertsonian translocation only. No controls |
| Salomon 2001 | No Down's syndrome pregnancies in population |
| Salonen 1997 | Fewer than 80% had gestational age estimated by USS |
| Saltvedt 2005 | Gestation greater than 14 weeks for nuchal scanning |
| Saridogan 1996 | Down's syndrome and Edward's syndrome affected pregnancies only |
| Savoldelli 1993 | Unable to extract useful data |
| Schielen 2009 | Full study information not given |
| Schiott 2006 | Unable to extract useful data |
| Schmidt 2007a | Not specific to Down’s syndrome |
| Schmidt 2007b | No separate Down’s syndrome data |
| Schmidt 2007c | No diagnostic data |
| Schmidt 2008a | Not specific to Down’s syndrome |
| Schmidt 2008b | Not specific to Down’s syndrome |
| Schmidt 2008c | Not specific to Down’s syndrome |
| Schmidt 2010 | No follow‐up data for test negatives |
| Schuchter 1998 | No Down's pregnancies in study population |
| Scott 1995 | Less than 5 Down's syndrome pregnancies in study population |
| Seeds 1990 | Review article |
| Seki 1995 | No Down's syndrome pregnancies in study population |
| Shenhav 2003 | No Down's syndrome pregnancies |
| Shintaku 1989 | Unable to extract useful data |
| Shulman 2003 | No Down's syndrome pregnancies in population |
| Sieroszewski 2008 | No Down’s syndrome specific information for specificity |
| Simon‐Bouy 1999 | Review article |
| Simpson 1986 | Gestational age confirmed by USS in less than 80% of population |
| Smith 1990 | Analysis of screen positive results |
| Smith 1996 | Review/meta‐analysis |
| Smith 1999 | Unable to extract useful data |
| Smith‐Bindman 2001 | Meta‐analysis of second trimester ultrasound markers |
| Smith‐Bindman 2003 | Population study, not examining DTA |
| Snijders 1995 | Study of prevalence, not screening |
| Snijders 1999 | Study of prevalence, not screening |
| Soergel 2006 | Less than 80% follow‐up |
| Sokol 1998 | Observation of Down's prevalence stratified by age |
| Sonek 2003 | Editorial |
| Sonek 2007 | ST ultrasound |
| Sood 2010 | No diagnostic data |
| Sooklim 2010 | ST ultrasound |
| Spencer 1985 | Fewer than 80% USS dated |
| Spencer 1991a | Likely fewer than 80% USS dated |
| Spencer 1991b | Unable to extract useful data |
| Spencer 1992 | Unable to extract useful data |
| Spencer 1993a | Fewer than 80% USS dated |
| Spencer 1993b | No Down's pregnancies in study population |
| Spencer 1993c | Unable to extract useful data |
| Spencer 1993d | Fewer than 80% of pregnancies had gestational age confirmed by USS |
| Spencer 1993e | Unable to extract useful data |
| Spencer 1995 | No Down's pregnancies in population |
| Spencer 1996 | Fewer than 80% of pregnancies had gestational age confirmed by USS |
| Spencer 1997 | Statistical modelling, aneuploid pregnancies only in study population |
| Spencer 1998a | No Down's pregnancies in population |
| Spencer 1998b | Unable to extract useful data |
| Spencer 1999a | Review |
| Spencer 1999b | Statistical methods paper |
| Spencer 2000a | Examination of median shifts rather than an evaluation of screening |
| Spencer 2000b | No Down's syndrome pregnancies in population |
| Spencer 2000c | No Down's syndrome pregnancies in population |
| Spencer 2000d | No Down's cases |
| Spencer 2000e | Male versus female fetuses |
| Spencer 2000f | No Down's cases in population |
| Spencer 2000g | No Down's pregnancies in population |
| Spencer 2000h | No Down's pregnancies in population |
| Spencer 2000i | Comparsison of fetal sex |
| Spencer 2001a | No Down's syndrome pregnancies in population |
| Spencer 2001b | Unable to extract useful data |
| Spencer 2001c | Unable to extract useful data |
| Spencer 2001d | Unable to extract useful data |
| Spencer 2001e | No Down's syndrome pregnancies in population |
| Spencer 2002a | No Down's pregnancies |
| Spencer 2002b | Risk validation study |
| Spencer 2002c | No Down's syndrome pregnancies in population |
| Spencer 2002d | Demonstration of median changes with time, rather than evaluation of screening |
| Spencer 2003a | No Down's pregnancies in population |
| Spencer 2003b | No Down's pregnancies in population |
| Spencer 2003c | Calculation of weight correction factor |
| Spencer 2003d | Fewer than 5 Down's syndrome pregnancies |
| Spencer 2004 | Calculation of smoking correction factor |
| Spencer 2005a | No Down's pregnancies |
| Spencer 2005b | No Down's pregnancies |
| Spencer 2005c | Comparison of two different assays ‐ not actual screening evaluation |
| Spencer 2008b | Unable to extract appropriate data for unaffected pregnancies |
| Spong 1999 | Comparison of male and female fetuses |
| Staboulidou 2009 | No diagnostic data |
| Stevens 1998 | Literature review |
| Stoll 1992 | Review article |
| Stressig 2011 | ST ultrasound |
| Su 2002 | Unable to extract useful data |
| Suchet 1995 | Review article |
| Suchy 1990 | Unable to ascertain method of confirmation of gestational age |
| Summers 2003a | Only 55% gestational ages estimated by USS |
| Summers 2003b | No Down's syndrome pregnancies in study population |
| Suntharasaj 2005 | Examination of inter‐observer variation in NT scanning |
| Susman 2010 | No diagnostic data |
| Sutton 2004 | Unable to extract useful data |
| Suzuki 1998 | Unable to extract useful data |
| Tabor 1987 | Geststional age not confirmed by USS |
| Tanski 1999 | Information on screen positive pregnancies only |
| Thilaganathan 1998 | No Down's syndrome pregnancies in study population |
| Thilaganathan 1999 | Editorial |
| Tislaric 2002 | No Down's syndrome pregnancies in population |
| Torok 1997 | Unable to extract useful data |
| Torring 2009 | Not possible to obtain full diagnostic data |
| Trninic‐Pjevic 2007 | Unable to obtain translation |
| Tsai 2001 | Less than 5 Down's syndrome pregnancies in study population |
| Valerio 1996 | Fewer than 80% pregnancies had gestational age estimated by USS |
| Van Blerk 1992 | Unable to extract useful data |
| Van Dyke 2007 | Not possible to obtain full diagnostic data |
| Van Heesch, 2006 | No Down's syndrome pregnancies in study population Software comparison study |
| Van Lith 1991 | Unable to extract useful data |
| Van Lith 1993 | Unable to extract useful data |
| Van Lith 1994 | Unable to extract useful data |
| Veress 1986 | Unable to extract useful data |
| Veress 1988 | Unable to extract useful data |
| Vergani 2008 | ST ultrasound |
| Vintzileos 2003 | Second trimester USS |
| Wald 1988a | Less than 80% had gestational age confirmed by ultrasound |
| Wald 1988b | Gestational age not confirmed by USS |
| Wald 1991 | No Down's pregnancies in study |
| Wald 1992a | Less than 80% had gestational age confirmed by ultrasound |
| Wald 1992b | No Down's pregnancies in study |
| Wald 1992c | No Down's pregnancies in study |
| Wald 1993 | No USS dating |
| Wald 1994a | No Down's syndrome pregnancies in population |
| Wald 1994b | Review article |
| Wald 1996a | No Down's pregnancies |
| Wald 1996b | Dated by LMP |
| Wald 1996c | No Down's syndrome pregnancies in population |
| Wald 1996d | Gestational age greater than 24 weeks |
| Wald 1997 | Data modelled on 3 separate populations of women |
| Wald 1998 | Unable to extract useful data |
| Wald 1999a | Unable to extract useful data |
| Wald 1999b | Gestational age not confirmed by USS |
| Wald 1999c | No Down's syndrome pregnancies |
| Wald 1999d | Modelled on several studies, some of which have no USS dating |
| Wald 2003c | No cases |
| Wald 2003d | Less than 80% had gestational age confirmed by USS |
| Wald 2006 | Modelled on SURRUS data |
| Wallace 1994 | Unable to extract useful data |
| Wallace 1997 | No Down's syndrome pregnancies in study population |
| Wang 2010 | ST ultrasound |
| Ward 2005 | Review article |
| Watt 1996 | No Down's syndrome pregnancies in study population |
| Watt 1996a | No Down's syndrome pregnancies in study population |
| Wax 2007 | No diagnostic data |
| Weinans 2001 | Unable to extract useful data |
| Weinans 2004 | Study of women's views on screening |
| Weisz 2007 | Cohort split into people having different tests and non‐representative samples of women assessed for each test |
| Welborn 1994 | Abnormal results only (cystic hygroma) |
| Wenstrom 1993 | Less than 80% of pregnancies had gestational age confirmed by USS |
| Wenstrom 1995a | Adjustment factors |
| Wenstrom 1995b | Less than 80% of pregnancies had gestational age confirmed by USS |
| Wetta 2011 | No diagnostic data |
| Whitlow 1998a | Unable to extract useful data |
| Whitlow 1998b | Unable to extract useful data |
| Whitlow 1999 | Unable to extract useful data |
| Williamson 1994 | Likely fewer than 80% USS dated |
| Wilson 2000 | Review |
| Wojdemann 2001 | No Down's syndrome pregnancies in study population |
| Wong 2003 | Less than 5 Down's syndrome pregnancies in population |
| Wright 2006 | Mathematical model |
| Wright 2007 | Simulation study, no new data |
| Xie 2010 | Only cases of false negatives and true negatives included |
| Yagel 1998 | Second trimester USS |
| Yamamoto 2001a | Unable to extract useful data |
| Yamamoto 2001b | Method of determination of gestational age unclear |
| Yamamoto 2001c | Unable to extract useful data |
| Yaron 2001 | Male versus female fetuses |
| Ye 1995 | Unable to obtain translation |
| Yoshida 2000 | Fewer than 80% pregnancies had gestational age estimated by USS |
| Zalel 2008 | No diagnostic data |
| Zeitune 1991 | Only aneuploid pregnancies included in study |
| Zelop 2005 | No Down's cases in population |
| Zhang 2011 | No diagnostic data |
| Zhao 1998 | Unable to obtain translation |
| Zhong 2011 | Second trimester ultrasound |
| Zoppi 2003 | Inappropriate study design |
CVS: CVS: chorionic villus sampling FISH: Fluorescence in situ hybridisation FPR: false positive rate LMP: last menstrual period NT: nuchal transparency SURUSS: Serum, Urine and Ultrasound Screening Study USS: ultrasound screening
Differences between protocol and review
The protocol intended to investigate several additional outcomes downstream from test accuracy, should they be reported in the test accuracy studies. When we attempted to extract this information however, it was found to be available in very few studies, and where such information was found it was difficult to extract meaningful data to allow for comparison between studies, as data were not reported in a universal manner. In several studies such outcomes were estimated rather than measured. Often they were not reported at all. The outcomes stated in the protocol which have not been included are: harms of testing; need for further testing; side effects of test; interventions and side effects; other abnormalities detected by testing; spontaneous miscarriage; miscarriage subsequent to invasive procedure, with or without normal karyotype; fetal karyotype; termination of pregnancy (prior to definitive testing or in a karyotypically normal pregnancy and following confirmation of Down’s syndrome or following detection of other chromosomal abnormalities); stillbirth; livebirth of affected and unaffected fetus; uptake of definitive testing by women.
The following refinements to the eligibility criteria were imposed to ensure that the quality of the included literature remained high. We excluded studies that identified fewer than five Down's syndrome pregnancies in their study population. We excluded studies that had less than 80% follow‐up of participants.
In addition, the analytical strategy was informed by the volume of tests and studies included, and developed so that we focused on key tests and test combinations by a) only meta‐analysed tests that were included in four or more studies or b) showed more than 70% sensitivity for more than 95% specificity. In addition, a requirement that a minimum of 10 studies for a single test was required before subgroup analysis was undertaken. Consequently several possible sources of heterogeneity were not investigated due to lack of data.
Contributions of authors
KA undertook the searches, applied eligibility criteria, extracted and entered data and wrote the first and second draft of the review.
ZA applied eligibility criteria, provided senior clinical input, oversaw the review process, and approved the final draft of the review.
JD supervised and planned the review, checked data extraction, supervised statistical analyses and wrote the second draft of the review.
JP applied eligibility criteria, provided senior clinical input, oversaw the review process, and approved the final draft of the review.
BG checked data extraction and undertook statistical analyses.
MP applied eligibility criteria, extracted and entered data for the updated literature search, and entered characteristics of studies information.
YT checked data extraction, undertook statistical analyses and wrote parts of the first draft of the review.
Sources of support
Internal sources
-
University of Birmingham, UK.
- Funding of the research time of JD and BG
External sources
-
NIHR Health Technology Assessment Programme, UK.
Project grant
-
NIHR Health Technology Assessment Programme, UK.
- Funding for the Cochrane Reviews of Diagnostic Test Accuracy Support Unit, based at the University of Birmingham (JD).
Declarations of interest
S Kate Alldred was supported by a project grant from the NIHR Health Technology Assessment Programme.
Boliang Guo: none known.
Jonathan J Deeks : none known.
Zarko Alfirevic (ZA) is Director of Harris Wellbeing Preterm Birth Centre which is grant funded by the charity Wellbeing of Women. This grant is administered by University of Liverpool and Zarko Alfirevic is not paid directly. He is the principal investigator or co‐investigator on several grants from public funders including National Institute of Health Research, British Medical Association, European Commission and WHO. He has received research support in the past from Perkin Elmer and Alere for research related to pre‐eclampsia and preterm birth prevention. These grants were administered by his employers and ZA did not benefit directly. ZA is also a Co‐coordinating Editor of Cochrane Pregnancy and Childbirth.
James P Neilson received an award from the UK NIHR to facilitate a panel of Cochrane systematic reviews on Down's syndrome.
Mary Pennant: none known.
Yemisi Takwoingi was supported by an award from the United Kingdom National Institute for Health Research [DRF‐2011‐04‐135].
New
References
References to studies included in this review
Aagaard‐Tillery 2009 {published data only}
- Aagaard‐Tillery KM, Malone FD, Nyberg DA, Porter TF, Cuckle HS, Fuchs K, et al. Role of second‐trimester genetic sonography after Down syndrome screening. Obstetrics & Gynecology 2009;114(6):1189‐96. [DOI] [PMC free article] [PubMed] [Google Scholar]
Audibert 2001 {published data only}
- Audibert F, Dommergues M, Benattar C, Taieb J, Thalabard JC, Frydman R. Screening for Down syndrome using first‐trimester ultrasound and second‐trimester maternal serum markers in a low‐risk population: a prospective longitudinal study. Ultrasound in Obstetrics & Gynecology 2001;18(1):26‐31. [DOI] [PubMed] [Google Scholar]
Babbur 2005 {published data only}
- Babbur V, Lees CC, Goodburn SF, Morris N, Breeze AC, Hackett GA. Prospective audit of a one‐centre combined nuchal translucency and triple test programme for the detection of trisomy 21. Prenatal Diagnosis 2005;25(6):465‐9. [DOI] [PubMed] [Google Scholar]
Baviera 2010 {published data only}
- Baviera G, Chimicata S, Domenico R, Granese R, Carbone C, Dugo N, et al. First‐ and second‐trimester ADAM12s in Down syndrome screening. Clinical Chemistry 2010;56(8):1355‐7. [DOI] [PubMed] [Google Scholar]
Benattar 1999 {published data only}
- Benattar C, Audibert F, Taieb J, Ville Y, Roberto A, Lindenbaum A, et al. Efficiency of ultrasound and biochemical markers for Down's syndrome risk screening. A prospective study. Fetal Diagnosis and Therapy 1999;14(2):112‐7. [DOI] [PubMed] [Google Scholar]
Bestwick 2010 {published data only}
- Bestwick JP, Huttly WJ, Wald NJ. Distribution of nuchal translucency in antenatal screening for Down's syndrome. Journal of Medical Screening 2010;17(1):8‐12 (Erratum in Journal of Medical Screening 2010;17(2):106). [DOI] [PMC free article] [PubMed] [Google Scholar]
Cuckle 2008 {published data only}
- Cuckle HS, Malone FD, Wright D, Porter TF, Nyberg DA, Comstock CH, et al. Contingent screening for Down syndrome‐results from the FaSTER trial. Prenatal Diagnosis 2008;28(2):89‐94. [DOI] [PubMed] [Google Scholar]
Goh 1996 {published data only}
- Goh HH, Anandakumr C, Tain CF, Leong WP, Aw BL, Wong YC, et al. Establishment and appraisal of a prenatal screening test for Down's syndrome based on the triple test of maternal serum parameters in a Singapore population. Singapore Journal of Obstetrics and Gynaecology 1996;27(2):81‐8. [Google Scholar]
Guanciali‐Franchi 2010 {published data only}
- Guanciali‐Franchi P, Iezzi I, Matarrelli B, Morizio E, Calabrese G, Palka G. Effectiveness of crosstrimester test in selecting high‐risk pregnant women to undergo invasive prenatal diagnosis. Prenatal Diagnosis 2010;30(8):795‐6. [DOI] [PubMed] [Google Scholar]
Habayeb 2010 {published data only}
- Habayeb O, Goodburn S, Chudleigh T, Brockelsby J, Missfelder‐Lobos H, Hackett G, et al. The NTplus method of screening for Down syndrome: achieving the 2010 targets?. Prenatal Diagnosis 2010;30(5):434‐7. [DOI] [PubMed] [Google Scholar]
Herman 2002 {published data only}
- Herman A, Dreazen E, Tovbin J, Weinraub Z, Bukovsky Y, Maymon R. Comparison between disclosure and non‐disclosure approaches for trisomy 21 screening tests. Human Reproduction (Oxford, England) 2002;17(5):1358‐62. [DOI] [PubMed] [Google Scholar]
Lam 2002 {published data only}
- Lam YH, Lee CP, Sin SY, Tang R, Wong HS, Wong SF, et al. Comparison and integration of first trimester fetal nuchal translucency and second trimester maternal serum screening for fetal Down syndrome. Prenatal Diagnosis 2002;22(8):730‐5. [DOI] [PubMed] [Google Scholar]
Malone 2005 {published data only}
- Malone FD, Canick JA, Ball RH, Nyberg DA, Comstock CH, Bukowski R, et al. First‐trimester or second‐trimester screening, or both, for Down's syndrome (see comment). New England Journal of Medicine 2005;353(19):2001‐11. [DOI] [PubMed] [Google Scholar]
Okun 2008 Integrated {published data only}
- Okun N, Summers AM, Hoffman B, Huang T, Winsor E, Chitayat D, et al. Prospective experience with integrated prenatal screening and first trimester combined screening for trisomy 21 in a large Canadian urban center. Prenatal Diagnosis 2008;28(11):987‐92. [DOI] [PubMed] [Google Scholar]
Palomaki 2006 {published data only}
- Palomaki GE, Wright DE, Summers AM, Neveux LM, Meier C, O'Donnell A, et al. Repeated measurement of pregnancy‐associated plasma protein‐A (PAPP‐A) in Down syndrome screening: a validation study. Prenatal Diagnosis 2006;26(8):730‐9. [DOI] [PubMed] [Google Scholar]
Rodrigues 2009 {published data only}
- Rodrigues LC, Ramos‐Dias AM, Carvalho V, Cirurgiao F. Evaluation of four years of prenatal screening for aneuploidies in Hospital S. Francisco Xavier using the integrated test. Journal of Medical Screening 2009;16(1):46‐7. [DOI] [PubMed] [Google Scholar]
Rozenberg 2002 {published data only}
- Rozenberg P, Malagrida L, Cuckle H, Durand‐Zaleski I, Nisand I, Audibert F, et al. Down's syndrome screening with nuchal translucency at 12(+0)‐14(+0) weeks and maternal serum markers at 14(+1)‐17(+0) weeks: a prospective study. Human Reproduction 2002;17(4):1093‐8. [DOI] [PubMed] [Google Scholar]
Schuchter 2001 {published data only}
- Schuchter K, Hafner E, Stangl G, Ogris E, Philipp K. Sequential screening for trisomy 21 by nuchal translucency measurement in the first trimester and maternal serum biochemistry in the second trimester in a low‐risk population. Ultrasound in Obstetrics & Gynecology 2001;18(1):23‐5. [DOI] [PubMed] [Google Scholar]
Wald 2003b {published data only}
- Wald NJ, Rodeck C, Hackshaw AK, Rudnicka A. SURUSS in perspective. Seminars in Perinatology 2005;29(4):225‐35. [DOI] [PubMed] [Google Scholar]
- Wald NJ, Rodeck C, Hackshaw AK, Rudnicka A. SURUSS in perspective (see comment). BJOG: an international journal of obstetrics and gynaecology 2004;111(6):521‐31. [DOI] [PubMed] [Google Scholar]
- Wald NJ, Rodeck C, Hackshaw AK, Walters J, Chitty L, Mackinson AM. First and second trimester antenatal screening for Down's syndrome: the results of the Serum, Urine and Ultrasound Screening Study (SURUSS). Journal of Medical Screening 2003;10(2):56‐104. [DOI] [PubMed] [Google Scholar]
- Wald NJ, Rodeck C, Hackshaw AK, Walters J, Chitty L, Mackinson AM, et al. First and second trimester antenatal screening for Down's syndrome: the results of the Serum, Urine and Ultrasound Screening Study (SURUSS). Health Technology Assessment (Winchester, England) 2003;7(11):1‐77. [DOI] [PubMed] [Google Scholar]
Wald 2009 {published data only}
- Wald NJ, Huttly WJ, Murphy KW, Ali K, Bestwick JP, Rodeck CH. Antenatal screening for Down's syndrome using the Integrated test at two London hospitals. Journal of Medical Screening 2009;16(1):7‐10. [DOI] [PubMed] [Google Scholar]
Wright 2010 FASTER trial {published data only}
- Wright D, Bradbury I, Malone F, D'Alton M, Summers A, Huang T, et al. Cross‐trimester repeated measures testing for Down's syndrome screening: an assessment. Health Technology Assessment (Winchester, England) 2010;14(33):1‐80. [DOI] [PubMed] [Google Scholar]
Wright 2010 North York {published data only}
- Wright D, Bradbury I, Malone F, D'Alton M, Summers A, Huang T, et al. Cross‐trimester repeated measures testing for Down's syndrome screening: an assessment. Health Technology Assessment (Winchester, England) 2010;14(33):1‐80. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Aagaard‐Tillery 2010 {published data only}
- Aagaard‐Tillery KM, Flint Porter T, Malone FD, Nyberg DA, Collins J, Comstock CH, et al. Influence of maternal BMI on genetic sonography in the FaSTER trial. Prenatal Diagnosis 2010;30(1):14‐22. [DOI] [PubMed] [Google Scholar]
Abbas 1995 {published data only}
- Abbas A, Chard T, Nicolaides K. Fetal and maternal hCG concentration in aneuploid pregnancies. British Journal of Obstetrics and Gynaecology 1995;102(7):561‐3. [DOI] [PubMed] [Google Scholar]
Abdul‐Hamid 2004 {published data only}
- Abdul‐Hamid S, Fox R, Martin I. Maternal serum screening for trisomy 21 in women with a false positive result in last pregnancy. Journal of Obstetrics & Gynaecology 2004;24(4):374‐6. [DOI] [PubMed] [Google Scholar]
Abraha 1999 {published data only}
- Abraha HD, Noble PL, Nicolaides KH, Sherwood RA. Maternal serum S100 protein in normal and Down syndrome pregnancies. Prenatal Diagnosis. 1999;19(4):334‐6. [PubMed] [Google Scholar]
Abu‐Rustum 2010 {published data only}
- Abu‐Rustum RS, Daou L, Abu‐Rustum SE. Role of first‐trimester sonography in the diagnosis of aneuploidy and structural fetal anomalies. Journal of Ultrasound in Medicine 2010;29(10):1445‐52. [DOI] [PubMed] [Google Scholar]
Achiron 2010 {published data only}
- Achiron R, Gindes L, Gilboa Y, Weissmann‐Brenner A, Berkenstadt M. Umbilical vein anomaly in fetuses with Down syndrome. Ultrasound in Obstetrics & Gynecology 2010;35(3):297‐301. [DOI] [PubMed] [Google Scholar]
Adekunle 1999 {published data only}
- Adekunle O, Gopee A, El‐Sayed M, Thilaganathan B. Increased first trimester nuchal translucency: pregnancy and infant outcomes after routine screening for Down's syndrome in an unselected antenatal population. British Journal of Radiology 1999;72(857):457‐60. [DOI] [PubMed] [Google Scholar]
Aitken 1993 {published data only}
- Aitken DA, McCaw G, Crossley JA, Berry E, Connor JM, Spencer K, Macri JN. First‐trimester biochemical screening for fetal chromosome abnormalities and neural tube defects. Prenatal Diagnosis 1993;13(8):681‐9. [DOI] [PubMed] [Google Scholar]
Aitken 1996 {published data only}
- Aitken DA, Syvertsen BS, Crossley JA, Berry E, Connor JM. Heat‐stable and immunoreactive placental alkaline phosphatase in maternal serum from Down's syndrome and trisomy 18 pregnancies.[see comment]. Prenatal Diagnosis 1996;16(11):1051‐4. [DOI] [PubMed] [Google Scholar]
Aitken 1996a {published data only}
- Aitken DA, Wallace EM, Crossley JA, Swanston IA, Pareren Y, Maarle M, et al. Dimeric Inhibin A as a marker for Down's syndrome in early pregnancy. New England Journal of Medicine 1996;334(19):1231‐6. [DOI] [PubMed] [Google Scholar]
Ajayi 2011 {published data only}
- Ajayi GO. Is there any effect of fetal gender on the markers of first trimester Down's syndrome screening?. Clinical & Experimental Obstetrics & Gynecology 2011;38(2):162‐4. [PubMed] [Google Scholar]
Akbas 2001 {published data only}
- Akbas SH, Ozben T, Alper O, Ugur A, Yucel G, Luleci G. Maternal serum screening for Down's syndrome, open neural tube defects and trisomy 18. Clinical Chemistry & Laboratory Medicine 2001;39(6):487‐90. [DOI] [PubMed] [Google Scholar]
Alexioy 2009 {published data only}
- Alexioy E, Alexioy E, Trakakis E, Kassanos D, Farmakidis G, Kondylios A, et al. Predictive value of increased nuchal translucency as a screening test for the detection of fetal chromosomal abnormalities. Journal of Maternal‐Fetal & Neonatal Medicine 2009;22(10):857‐62. [DOI] [PubMed] [Google Scholar]
Allingham‐Hawkins 2011 {published data only}
- Allingham‐Hawkins DJ, Chitayat D, Cirigliano V, Summers A, Tokunaga J, Winsor E, et al. Prospective validation of quantitative fluorescent polymerase chain reaction for rapid detection of common aneuploidies. Genetics in Medicine 2011;13(2):140‐7. [DOI] [PubMed] [Google Scholar]
American College 2009 {published data only}
- American College of Nurse‐Midwives. Share with women. Prenatal tests for Down syndrome [Comparitir con muejeres. Pruebas prenatales para detectar el sindrome de Down]. Journal of Midwifery & Women's Health 2009;54(6):527‐8. [PubMed] [Google Scholar]
Antona 1998 {published data only}
- Antona D, Wallace EM, Shearing C, Ashby JP, Groome NP. Inhibin A and pro‐alphaC Inhibin A in Down syndrome and normal pregnancies. Prenatal Diagnosis 1998;18(11):1122‐6. [DOI] [PubMed] [Google Scholar]
Antsaklis 1999 {published data only}
- Antsaklis A, Papantoniou N, Mesogitis S, Michalas S, Aravantinos D. Pregnant women of 35 years of age or more: Maternal serum markers or amniocentesis?. Journal of Obstetrics and Gynaecology 1999;19(3):253‐6. [DOI] [PubMed] [Google Scholar]
Anuwutnavin 2009 {published data only}
- Anuwutnavin S, Wanitpongpan P, Chanprapaph P. Specificity of fetal tricuspid regurgitation in prediction of Down syndrome in Thai fetuses at 17‐23 weeks of gestation. Journal of the Medical Association of Thailand 2009;92(9):1123‐30. [PubMed] [Google Scholar]
Ashwood 1987 {published data only}
- Ashwood ER, Cheng E, Luthy DA. Maternal serum alpha‐fetoprotein and fetal trisomy‐21 in women 35 years and older: implications for alpha‐fetoprotein screening programs. American Journal of Medical Genetics 1987;26(3):531‐9. [DOI] [PubMed] [Google Scholar]
Asrani 2005 {published data only}
- Asrani CH. Triple marker. National Journal of Homoeopathy 2005;7(3):174. [Google Scholar]
Audibert 2001b {published data only}
- Audibert F, Dommergues M, Benattar C, Taieb J, Thalabard JC, Frydman R. Screening for Down syndrome using first‐trimester ultrasound and second‐trimester maternal serum markers in a low‐risk population: a prospective longitudinal study. Ultrasound in Obstetrics & Gynecology 2001;18(1):26‐31. [DOI] [PubMed] [Google Scholar]
Axt‐Fleidner 2006 {published data only}
- Axt‐Fliedner R, Schwarze A, Kreiselmaier P, Krapp M, Smrcek J, Diedrich K. Umbilical cord diameter at 11‐14 weeks of gestation: Relationship to nuchal translucency, ductus venous blood flow and chromosomal defects. Fetal Diagnosis and Therapy 2006;21(4):390‐5. [DOI] [PubMed] [Google Scholar]
Azuma 2002 {published data only}
- Azuma M, Yamamoto R, Wakui Y, Minobe S, Satomura S, Fujimoto S. A novel method for the detection of Down syndrome with the use of four serum markers. American Journal of Obstetrics & Gynecology 2002;187(1):197‐201. [DOI] [PubMed] [Google Scholar]
Baghagho 2004 {published data only}
- Baghagho EE, Kharboush IF, El‐Kaffash DM, KarKour TA, Ismail SR, Mortada MM. Maternal serum alpha fetoprotein among pregnant females in Alexandria. Journal of the Egyptian Public Health Association 2004;79(1‐2):59‐81. [PubMed] [Google Scholar]
Bahado‐Singh 1995 {published data only}
- Bahado‐Singh RO, Goldstein I, Uerpairojkit B, Copel JA, Mahoney MJ, Baumgarten A. Normal nuchal thickness in the midtrimester indicates reduced risk of Down syndrome in pregnancies with abnormal triple‐screen results. American Journal of Obstetrics and Gynecology 1995;173(4):1106‐10. [DOI] [PubMed] [Google Scholar]
Bahado‐Singh 1996 {published data only}
- Bahado‐Singh RO, Tan A, Deren O, Hunter D, Copel J, Mahoney MJ. Risk of Down syndrome and any clinically significant chromosome defect in pregnancies with abnormal triple‐screen and normal targeted ultrasonographic results. American Journal of Obstetrics and Gynecology 1996;175(4 I):824‐9. [DOI] [PubMed] [Google Scholar]
Bahado‐Singh 1999 {published data only}
- Bahado‐Singh RO, Oz AU, Flores D, Cermik D, Acuna E, Mahoney MJ, et al. Nuchal thickness, urine ß‐core fragment level, and maternal age for down syndrome screening. American Journal of Obstetrics and Gynecology 1999;180(2 I):491‐5. [DOI] [PubMed] [Google Scholar]
Bahado‐Singh 2002 {published data only}
- Bahado‐Singh RO, Shahabi S, Karaca M, Mahoney MJ, Cole L, Oz UA. The comprehensive midtrimester test: High‐sensitivity Down syndrome test. American Journal of Obstetrics and Gynecology 2002;186(4):803‐8. [DOI] [PubMed] [Google Scholar]
Bahado‐Singh 2003 {published data only}
- Bahado‐Singh RO, Cheng CC, Matta P, Small M, Mahoney MJ. Combined serum and ultrasound screening for detection of fetal aneuploidy. Seminars in Perinatology 2003;27(2):145‐51. [DOI] [PubMed] [Google Scholar]
Ball 2007 {published data only}
- Ball RH, Caughey AB, Malone FD, Nyberg DA, Comstock CH, Saade GR, et al. First‐ and second‐trimester evaluation of risk for Down syndrome. Obstetrics & Gynecology 2007;110(1):10‐7. [DOI] [PubMed] [Google Scholar]
Bar‐Hava 2001 {published data only}
- Bar‐Hava I, Yitzhak M, Krissi H, Shohat M, Shalev J, Czitron B, et al. Triple‐test screening in in vitro fertilization pregnancies. Journal of Assisted Reproduction and Genetics 2001;18(4):226‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Barkai 1996 {published data only}
- Barkai G, Goldman B, Ries L, Chaki R, Dor J, Cuckle H. Down's syndrome screening marker levels following assisted reproduction. Prenatal Diagnosis 1996;16(12):1111‐4. [DOI] [PubMed] [Google Scholar]
Barnabei 1995 {published data only}
- Barnabei VM, Krantz DA, Macri JN, Larsen JW Jr. Enhanced twin pregnancy detection within an open neural tube defect and Down syndrome screening protocol using free‐ß hCG and AFP. Prenatal Diagnosis 1995;15(12):1131‐4. [DOI] [PubMed] [Google Scholar]
Bartels 1988 {published data only}
- Bartels I, Lindemann A. Maternal levels of pregnancy‐specific ß 1‐glycoprotein (SP‐1) are elevated in pregnancies affected by Down's syndrome. Human Genetics 1988;80(1):46‐8. [DOI] [PubMed] [Google Scholar]
Bartels 1993 {published data only}
- Bartels I, Hoppe‐Sievert B, Bockel B, Herold S, Caesar J. Adjustment formulae for maternal serum alpha‐fetoprotein, human chorionic gonadotropin, and unconjugated oestriol to maternal weight and smoking. Prenatal Diagnosis 1993;13(2):123‐30. [DOI] [PubMed] [Google Scholar]
Barth 1991 {published data only}
- Barth WH Jr, Frigoletto FD Jr, Krauss CM, MacMillin MD, Stryker JM, Benacerraf BR. Ultrasound detection of fetal aneuploidy in women with elevated maternal serum alpha‐fetoprotein. Obstetrics & Gynecology 1991;77(6):897‐900. [PubMed] [Google Scholar]
Bas‐Budecka 2007 {published data only}
- Bas‐Budecka E, Perenc M, Sieroszewski P. [Abnormal second trimester screening for fetal chromosomal abnormalities as a predictor of adverse pregnancy outcome]. [Polish]. Ginekologia Polska 2007;78(11):877‐80. [PubMed] [Google Scholar]
Baviera 2004 {published data only}
- Baviera G, Carbone C, Corrado F, Mastrantonio P. Placental growth hormone in Down's syndrome screening. Journal of Maternal‐Fetal & Neonatal Medicine 2004;16(4):241‐3. [DOI] [PubMed] [Google Scholar]
Bazzett 1998 {published data only}
- Bazzett LB, Yaron Y, O'Brien JE, Critchfield G, Kramer RL, Ayoub M, et al. Fetal gender impact on multiple‐marker screening results. American Journal of Medical Genetics 1998;76(5):369‐71. [PubMed] [Google Scholar]
Beke 2008 {published data only}
- Beke A, Barakonyi E, Belics Z, Joo JG, Csaba A, Papp C, et al. Risk of chromosome abnormalities in the presence of bilateral or unilateral choroid plexus cysts. Fetal Diagnosis and Therapy 2008;23(3):185‐91. [DOI] [PubMed] [Google Scholar]
Bellver 2005 {published data only}
- Bellver J, Lara C, Soares SR, Ramirez A, Pellicer A, Remohi J, et al. First trimester biochemical screening for Down's syndrome in singleton pregnancies conceived by assisted reproduction. Human Reproduction. 2005;20(9):2623‐7. [DOI] [PubMed] [Google Scholar]
Benn 1995 {published data only}
- Benn PA, Horne D, Briganti S, Greenstein RM. Prenatal diagnosis of diverse chromosome abnormalities in a population of women identified by triple‐marker testing as screen positive for Down syndrome. American Journal of Obstetrics and Gynecology 1995;173(2):496‐501. [DOI] [PubMed] [Google Scholar]
Benn 1996 {published data only}
- Benn PA, Horne D, Craffey A, Collins R, Ramsdell L, Greenstein R. Maternal serum screening for birth defects: results of a Connecticut regional program. Connecticut Medicine 1996;60(6):323‐7. [PubMed] [Google Scholar]
Benn 1997 {published data only}
- Benn PA, Clive JM, Collins R. Medians for second‐trimester maternal serum alpha‐fetoprotein, human chorionic gonadotropin, and unconjugated estriol; differences between races or ethnic groups. Clinical Chemistry 1997;43(2):333‐7. [PubMed] [Google Scholar]
Benn 1998 {published data only}
- Benn PA. Preliminary evidence for associations between second‐trimester human chorionic gonadotropin and unconjugated oestriol levels with pregnancy outcome in Down syndrome pregnancies. Prenatal Diagnosis 1998;18(4):319‐24. [PubMed] [Google Scholar]
Benn 2001 {published data only}
- Benn PA, Ying J, Beazoglou T, Egan JF. Estimates for the sensitivity and false‐positive rates for second trimester serum screening for Down syndrome and trisomy 18 with adjustment for cross‐identification and double‐positive results. Prenatal Diagnosis 2001;21(1):46‐51. [PubMed] [Google Scholar]
Benn 2002 {published data only}
- Benn PA, Kaminsky LM, Ying J, Borgida AF, Egan JF. Combined second‐trimester biochemical and ultrasound screening for Down syndrome. Obstetrics and Gynecology 2002;100(6):1168‐76. [DOI] [PubMed] [Google Scholar]
Benn 2003 {published data only}
- Benn PA, Fang M, Egan JFX, Horne D, Collins R. Incorporation of inhibin‐A in second‐trimester screening for Down syndrome. Obstetrics and Gynecology 2003;101(3):451‐4. [DOI] [PubMed] [Google Scholar]
Benn 2003a {published data only}
- Benn P. Improved antenatal screening for Down's syndrome. Lancet 2003;361(9360):794‐5. [DOI] [PubMed] [Google Scholar]
Benn 2005 {published data only}
- Benn P, Wright D, Cuckle H. Practical strategies in contingent sequential screening for Down syndrome. Prenatal Diagnosis 2005;25(8):645‐52. [DOI] [PubMed] [Google Scholar]
Benn 2005a {published data only}
- Benn P, Donnenfeld AE. Sequential Down syndrome screening: the importance of first and second trimester test correlations when calculating risk. Journal of Genetic Counseling 2005;14(6):409‐13. [DOI] [PubMed] [Google Scholar]
Benn 2007 {published data only}
- Benn PA, Campbell WA, Zelop CM, Ingardia C, Egan JF. Stepwise sequential screening for fetal aneuploidy. American Journal of Obstetrics and Gynecology 2007;197(3):312‐5. [DOI] [PubMed] [Google Scholar]
Berry 1995 {published data only}
- Berry E, Aitken DA, Crossley JA, Macri JN, Connor JM. Analysis of maternal serum alpha‐fetoprotein and free ß human chorionic gonadotrophin in the first trimester: implications for Down's syndrome screening. Prenatal Diagnosis 1995;15(6):555‐65. [DOI] [PubMed] [Google Scholar]
Berry 1997 {published data only}
- Berry E, Aitken DA, Crossley JA, Macri JN, Connor JM. Screening for Down's syndrome: changes in marker levels and detection rates between first and second trimesters. British Journal of Obstetrics & Gynaecology 1997;104(7):811‐7. [DOI] [PubMed] [Google Scholar]
Bersinger 1994 {published data only}
- Bersinger NA, Brizot ML, Johnson A, Snijders RJ, Abbott J, Schneider H, et al. First trimester maternal serum pregnancy‐associated plasma protein A and pregnancy‐specific ß 1‐glycoprotein in fetal trisomies. British Journal of Obstetrics & Gynaecology 1994;101(11):970‐4. [DOI] [PubMed] [Google Scholar]
Bersinger 2000 {published data only}
- Bersinger NA, Xin WZ. Glycosylation of pregnancy‐associated plasma protein a (PAPP‐A) and pregnancy‐specific (ß)(1)‐glycoprotein (SP1): Relevance for fetal down syndrome screening and for placental function studies. Immuno‐Analyse et Biologie Specialisee 2000;15(6):402‐8. [Google Scholar]
Bersinger 2001 {published data only}
- Bersinger NA, Chanson A, Crazzolara S, Hänggi W, Pescia G, Scheier M, et al. Serum levels of placenta protein markers: The relevance of differences between spontaneous and after in vitro fertilization pregnancies for fetal trisomy screening. Journal fur Fertilitat und Reproduktion 2001;11(3):7‐13. [Google Scholar]
Bersinger 2003 {published data only}
- Bersinger NA, Noble P, Nicolaides KH. First‐trimester maternal serum PAPP‐A, SP1 and M‐CSF levels in normal and trisomic twin pregnancies. Prenatal Diagnosis 2003;23(2):157‐62. [DOI] [PubMed] [Google Scholar]
Bersinger 2004 {published data only}
- Bersinger NA, Wunder D, Vanderlick F, Chanson A, Pescia G, Janecek P, et al. Maternal serum levels of placental proteins after in vitro fertilisation and their implications for prenatal screening. Prenatal Diagnosis 2004;24(6):471‐7. [DOI] [PubMed] [Google Scholar]
Bersinger 2005 {published data only}
- Bersinger NA, Vanderlick F, Birkhäuser MH, Janecek P, Wunder D. First trimester serum concentrations of placental proteins in singleton and multiple IVF pregnancies: Implications for Down syndrome screening. Immuno‐Analyse et Biologie Specialisee 2005;20(1):21‐7. [Google Scholar]
Bestwick 2008 {published data only}
- Bestwick JP, Huttly WJ, Wald NJ. First trimester Down's syndrome screening marker values and cigarette smoking: new data and a meta‐analysis on free beta human chorionic gonadotophin, pregnancy‐associated plasma protein‐A and nuchal translucency. Journal of Medical Screening 2008;15(4):204‐6. [DOI] [PubMed] [Google Scholar]
Biggio 2004 {published data only}
- Biggio Jr, Morris TC, Owen J, Stringer JSA. An outcomes analysis of five prenatal screening strategies for trisomy 21 in women younger than 35 years. American Journal of Obstetrics and Gynecology 2004;190(3):721‐9. [DOI] [PubMed] [Google Scholar]
Bilardo 2011 {published data only}
- Bilardo CM, Timmerman E, Medina PG, Clur SA. Low‐resistance hepatic artery flow in first‐trimester fetuses: an ominous sign. Ultrasound in Obstetrics & Gynecology 2011;37(4):438‐43. [DOI] [PubMed] [Google Scholar]
Bindra 2002 {published data only}
- Bindra R, Heath V, Nicolaides KH. Screening for chromosomal defects by fetal nuchal translucency at 11 to 14 weeks. Clinical Obstetrics and Gynecology 2002;45(3):661‐70. [DOI] [PubMed] [Google Scholar]
Blundell 1999 {published data only}
- Blundell G, Ashby JP, Martin C, Shearing CH, Langdale‐Brown B, Keeling J, et al. Clinical follow‐up of high mid‐trimester maternal serum intact human chorionic gonadotrophin concentrations in singleton pregnancies. Prenatal Diagnosis 1999;19(3):219‐23. [DOI] [PubMed] [Google Scholar]
Boormans 2010 {published data only}
- Boormans EM, Birnie E, Oepkes D, Galjaard RJ, Schuring‐Blom GH, Lith JM, et al. Comparison of multiplex ligation‐dependent probe amplification and karyotyping in prenatal diagnosis. Obstetrics and Gynecology 2010;115(2 Pt 1):297‐303. [DOI] [PubMed] [Google Scholar]
Boots 1989 {published data only}
- Boots LR, Davis RO, Foster JM, Goldenberg RL. Maternal serum alpha‐fetoprotein prenatal screening for Down syndrome. Alabama Medicine 1989;59(1):25‐7. [PubMed] [Google Scholar]
Bornstein 2009a {published data only}
- Bornstein E, Lenchner E, Donnenfeld A, Barnhard Y, Seubert D, Divon MY. Advanced maternal age as a sole indication for genetic amniocentesis; risk‐benefit analysis based on a large database reflecting the current common practice. Journal of Perinatal Medicine 2009;37(2):99‐102. [DOI] [PubMed] [Google Scholar]
Bornstein 2009b {published data only}
- Bornstein E, Lenchner E, Donnenfeld A, Kapp S, Keeler SM, Divon MY. Comparison of modes of ascertainment for mosaic vs complete trisomy 21. American Journal of Obstetrics and Gynecology 2009;200(4):440‐5. [DOI] [PubMed] [Google Scholar]
Bornstein 2010 {published data only}
- Bornstein E, Lenchner E, Donnenfeld A, Jodicke C, Keeler SM, Kapp S, et al. Complete trisomy 21 vs translocation Down syndrome: a comparison of modes of ascertainment. American Journal of Obstetrics and Gynecology 2010;203(4):391‐5. [DOI] [PubMed] [Google Scholar]
Borowski 2007 {published data only}
- Borowski D, Czuba B, Cnota W, Hincz P, Czekierdowski A, Gajewska J, et al. [Evaluation of pregnancy‐associated plasma protein A (PAPP‐A) and free beta subunit of human chorionic gonadotropin (beta hCG) levels and sonographic assessement of fetal nuchal translucency (NT) in singleton pregnancies between 11 and 14 weeks of gestation‐‐Polish multi‐centre research]. [Polish]. Ginekologia Polska 2007;78(5):384‐7. [PubMed] [Google Scholar]
Borrell 2007 {published data only}
- Borrell A, Mercade I, Casals E, Borobio V, Seres A, Soler A, et al. Combining fetal nuchal fold thickness with second‐trimester biochemistry to screen for trisomy 21. Ultrasound in Obstetrics & Gynecology 2007;30(7):941‐5. [DOI] [PubMed] [Google Scholar]
Borrell 2009 {published data only}
- Borrell A, Borobio V, Bestwick JP, Wald NJ. Ductus venosus pulsatility index as an antenatal screening marker for Down's syndrome: use with the Combined and Integrated tests. Journal of Medical Screening 2009;16(3):112‐8. [DOI] [PubMed] [Google Scholar]
Borruto 2002 {published data only}
- Borruto F, Comparetto C, Acanfora L, Bertini G, Rubaltelli FF. Role of ultrasound evaluation of nuchal translucency in prenatal diagnosis. Clinical & Experimental Obstetrics & Gynecology 2002;29(4):235‐41. [PubMed] [Google Scholar]
Bottalico 2009 {published data only}
- Bottalico JN, Chen X, Tartaglia M, Rosario B, Yarabothu D, Nelson L. Second‐trimester genetic sonogram for detection of fetal chromosomal abnormalities in a community‐based antenatal testing unit. Ultrasound in Obstetrics & Gynecology 2009;33(2):161‐8. [DOI] [PubMed] [Google Scholar]
Boue 1990 {published data only}
- Boue A, Muller F. Screening for Down's syndrome with maternal serum human chorionic gonadotropin at midtrimester. Current Opinion in Pediatrics 1990;2(6):1157‐60. [Google Scholar]
Bradley 1994 {published data only}
- Bradley LA, Horwitz JA, Dowman AC, Ponting NR, Peterson LM. Triple marker screening for fetal Down syndrome. International Pediatrics 1994;9(3):168‐74. [Google Scholar]
Braithwaite 1996 {published data only}
- Braithwaite JM, Economides DL. Nuchal translucency and screening for Down's syndrome. Contemporary Reviews in Obstetrics and Gynaecology 1996;8(2):75‐81. [Google Scholar]
Brambati 1995 {published data only}
- Brambati B, Cislaghi C, Tului L, Alberti E, Amidani M, Colombo U, et al. First‐trimester Down's syndrome screening using nuchal translucency: a prospective study in women undergoing chorionic villus sampling. Ultrasound in Obstetrics & Gynecology 1995;5(1):9‐14. [DOI] [PubMed] [Google Scholar]
Brambati 1996 {published data only}
- Brambati B, Tului L, Alberti E. Sonography in the first trimester screening of trisomy 21 and other fetal aneuploidies. [Review] [73 refs]. Early Pregnancy 1996;2(3):155‐67. [PubMed] [Google Scholar]
Brizot 1995 {published data only}
- Brizot ML, Bersinger NA, Xydias G, Snijders RJ, Nicolaides KH. Maternal serum Schwangerschafts protein‐1 (SP1) and fetal chromosomal abnormalities at 10‐13 weeks' gestation. Early Human Development 1995;43(1):31‐6. [DOI] [PubMed] [Google Scholar]
Brizot 1995a {published data only}
- Brizot ML, Kuhn P, Bersinger NA, Snijders RJ, Nicolaides KH. First trimester maternal serum alpha‐fetoprotein in fetal trisomies. British Journal of Obstetrics & Gynaecology 1995;102(1):31‐4. [DOI] [PubMed] [Google Scholar]
Brizzi 1989a {published data only}
- Brizzi L, Cariati E, Periti E, Nannini R, Torricelli F, Cappelli G, et al. Evaluation of maternal serum alpha‐fetoprotein and ultrasound examination to screen fetal chromosomal abnormalities. Journal of Nuclear Medicine & Allied Sciences 1989;33(3 Suppl):85‐8. [PubMed] [Google Scholar]
Brock 1990 {published data only}
- Brock DJ, Barron L, Holloway S, Liston WA, Hillier SG, Seppala M. First‐trimester maternal serum biochemical indicators in Down syndrome. Prenatal Diagnosis 1990;10(4):245‐51. [DOI] [PubMed] [Google Scholar]
Calda 2010 {published data only}
- Calda P, Sipek A, Gregor V. Gradual implementation of first trimester screening in a population with a prior screening strategy: population based cohort study. Acta Obstetricia et Gynecologica Scandinavica 2010;89(8):1029‐33. [DOI] [PubMed] [Google Scholar]
Campogrande 2001 {published data only}
- Campogrande M, Viora E, Errante G, Bastonero S, Sciarrone A, Grassi Pirrone P, et al. Correlations between first and second trimester markers for Down's syndrome screening. Journal of Medical Screening 2001;8(3):163‐4. [DOI] [PubMed] [Google Scholar]
Canick 1988 {published data only}
- Canick JA, Knight GJ, Palomaki GE, Haddow JE, Cuckle HS, Wald NJ. Low second trimester maternal serum unconjugated oestriol in pregnancies with Down's syndrome. British Journal of Obstetrics & Gynaecology 1988;95(4):330‐3. [DOI] [PubMed] [Google Scholar]
Canick 1995 {published data only}
- Canick JA, Kellner LH, Saller DN Jr, Palomaki GE, Walker RP, Osathanondh R. Second‐trimester levels of maternal urinary gonadotropin peptide in down syndrome pregnancy. Prenatal Diagnosis 1995;15(8):739‐44. [DOI] [PubMed] [Google Scholar]
Canini 2002 {published data only}
- Canini S, Prefumo F, Famularo L, Venturini PL, Palazzese V, Biasio P. Comparison of first trimester, second trimester and integrated Down's syndrome screening results in unaffected pregnancies. Clinical Chemistry & Laboratory Medicine 2002;40(6):600‐3. [DOI] [PubMed] [Google Scholar]
Cans 1998 {published data only}
- Cans C, Amblard F, Devillard F, Pison H, Jalbert P, Jouk PS. Population screening for aneuploidy using maternal age and ultrasound. Prenatal Diagnosis 1998;18(7):683‐92. [PubMed] [Google Scholar]
Carreras 1991 {published data only}
- Carreras de Paz JJ, Silva Mendoza JM, Violante Diaz M, Cerrillo Hinojosa M, Ahued Ahued JR. Proposed normal values for alpha fetoprotein in maternal serum for the detection of neural tube closure defects and Down syndrome. Preliminary study [Propuesta de valores normales de alfa fetoproteina (AFP) en suero materno para la deteccion de defectos en el cierre del tubo neural (DCTN) y sindrome de Down (SD). Estudio preliminar]. Ginecologia y Obstetricia de Mexico 1991;59:261‐4. [PubMed] [Google Scholar]
Caughey 2007 {published data only}
- Caughey AB, Musci TJ, Belluomini J, Main D, Otto C, Goldberg J. Nuchal translucency screening: how do women actually utilize the results?. Prenatal Diagnosis 2007;27(2):119‐23. [DOI] [PubMed] [Google Scholar]
Cebesoy 2008 {published data only}
- Cebesoy FB. Combining 'nasal bone length assessment as MoM' with other markers for trisomy 21 screening: could it be more effective?. American Journal of Obstetrics and Gynecology 2008;198(6):726‐7. [DOI] [PubMed] [Google Scholar]
Chelli 2008 {published data only}
- Chelli D, Dimassi K, Chaabouni M, Ben Saad M, Mssaed H, Bchir F, et al. [Prenatal diagnosis of trisomy 21: the Tunisian experience]. [French]. Sante 2008;18(4):199‐203. [PubMed] [Google Scholar]
Chen 1999 {published data only}
- Chen FM. Integrated screening for Down's syndrome. Journal of Family Practice 1999;48(11):846‐7. [PubMed] [Google Scholar]
Chen 2002 {published data only}
- Chen M, Lam YH, Tang MH, Lee CP, Sin SY, Tang R, et al. The effect of ethnic origin on nuchal translucency at 10‐14 weeks of gestation. Prenatal Diagnosis 2002;22(7):576‐8. [DOI] [PubMed] [Google Scholar]
Chen 2004 {published data only}
- Chen M, Lam YH, Lee CP, Tang MHY. Ultrasound screening of fetal structural abnormalities at 12 to 14 weeks in Hong Kong. Prenatal Diagnosis 2004;24(2):92‐7. [DOI] [PubMed] [Google Scholar]
Chen 2005 {published data only}
- Chen CP, Lin CJ, Wang W. Impact of second‐trimester maternal serum screening on prenatal diagnosis of Down syndrome and the use of amniocentesis in the Taiwanese population. Taiwanese Journal of Obstetrics and Gynecology 2005;44(1):31‐5. [Google Scholar]
Chen 2008 {published data only}
- Chen M, Lee CP, Lam YH, Tang RY, Chan BC, Wong SF, et al. Comparison of nuchal and detailed morphology ultrasound examinations in early pregnancy for fetal structural abnormality screening: a randomized controlled trial. Ultrasound in Obstetrics & Gynecology 2008;31(2):136‐46. [DOI] [PubMed] [Google Scholar]
Cheng 1993 {published data only}
- Cheng EY, Luthy DA, Zebelman AM, Williams MA, Lieppman RE, Hickok DE. A prospective evaluation of a second‐trimester screening test for fetal Down syndrome using maternal serum alpha‐fetoprotein, hCG, and unconjugated estriol. Obstetrics & Gynecology 1993;81(1):72‐7. [PubMed] [Google Scholar]
Cheng 1999 {published data only}
- Cheng PJ, Liu CM, Chang SD, Lin YT, Soong YK. Elevated second‐trimester maternal serum hCG in women undergoing haemodialysis. Prenatal Diagnosis 1999;19(10):955‐8. [DOI] [PubMed] [Google Scholar]
Cheng 2004a {published data only}
- Cheng CC, Bahado‐Singh RO, Chen SC, Tsai MS. Pregnancy outcomes with increased nuchal translucency after routine Down syndrome screening. International Journal of Gynaecology & Obstetrics 2004;84(1):5‐9. [DOI] [PubMed] [Google Scholar]
Cheng 2004b {published data only}
- Cheng PJ, Chu DC, Chueh HY, See LC, Chang HC, Weng DR. Elevated maternal midtrimester serum free ß‐human chorionic gonadotropin levels in vegetarian pregnancies that cause increased false‐positive Down syndrome screening results. American Journal of Obstetrics and Gynecology 2004;190(2):442‐7. [DOI] [PubMed] [Google Scholar]
Chitayat 2002 {published data only}
- Chitayat D, Farrell SA, Huang T, Meier C, Wyatt PR, Summers AM. Double‐positive maternal serum screening results for down syndrome and open neural tube defects: An indicator for fetal structural or chromosomal abnormalities and adverse obstetric outcomes. American Journal of Obstetrics and Gynecology 2002;187(3):758‐63. [DOI] [PubMed] [Google Scholar]
Chiu 2011 {published data only}
- Chiu RW, Akolekar R, Zheng YW, Leung TY, Sun H, Chan KC, et al. Non‐invasive prenatal assessment of trisomy 21 by multiplexed maternal plasma DNA sequencing: large scale validity study. BMJ 2011;342:c7401. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cho 2009 {published data only}
- Cho EH, Park BY, Kang YS, Lee EH. Validation of QF‐PCR in a Korean population. Prenatal Diagnosis 2009;29(3):213‐6. [DOI] [PubMed] [Google Scholar]
Chou 2009 {published data only}
- Chou CY, Hsieh FJ, Cheong ML, Lee FK, She BQ, Tsai MS. First‐trimester Down syndrome screening in women younger than 35 years old and cost‐effectiveness analysis in Taiwan population. Journal of Evaluation in Clinical Practice 2009;15(5):789‐96. [DOI] [PubMed] [Google Scholar]
Christiansen 2002 {published data only}
- Christiansen M, Hogdall EV, Larsen SO, Hogdall C. The variation of risk estimates through pregnancy in second trimester maternal serum screening for Down syndrome. Prenatal Diagnosis 2002;22(5):385‐7. [DOI] [PubMed] [Google Scholar]
Christiansen 2007 {published data only}
- Christiansen M, Spencer K, Laigaard J, Cowans NJ, Larsen SO, Wewer UM. ADAM 12 as a second‐trimester maternal serum marker in screening for Down syndrome. Prenatal Diagnosis 2007;27(7):611‐5. [DOI] [PubMed] [Google Scholar]
Christiansen 2008 {published data only}
- Christiansen M, Sorensen TL, Larsen SO, Norgaard‐Pedersen B. First‐trimester maternal serum progesterone in aneuploid pregnancies. Prenatal Diagnosis 2008;28(4):319‐22. [DOI] [PubMed] [Google Scholar]
Chung 2000 {published data only}
- Chung BL, Kim YP, Nam MH. The application of three‐dimensional ultrasound to nuchal translucency thickness measurement at 10‐14 weeks of gestation. Prenatal and Neonatal Medicine 2000;5(1):17‐21. [Google Scholar]
CNGOF 1996 {published data only}
- Anon. Blood screening of Down's syndrome (Trisomy 21) and reimbursement of karyotype for women under 38. Revue Francaise de Gynecologie et d'Obstetrique 1996;91(11):575‐7. [Google Scholar]
Cocciolone 2008 {published data only}
- Cocciolone R, Brameld K, O'Leary P, Haan E, Muller P, Shand K. Combining first and second trimester markers for Down syndrome screening: think twice. Australian & New Zealand Journal of Obstetrics & Gynaecology 2008;48(5):492‐500. [DOI] [PubMed] [Google Scholar]
Cole 1996 {published data only}
- Cole L, Isozaki T, Palomaki G, Canick J, Iles R, Kellner L, et al. Detection of ß‐core fragment in second trimester Down's syndrome pregnancies. [Review]. Early Human Development 1996;47(Suppl):S47‐8. [DOI] [PubMed] [Google Scholar]
Comas 2001 {published data only}
- Comas C, Antolín E, Torrents M, Muñoz A, Figueras F, Echevarría M, et al. Early screening for chromosomal abnormalities: New strategies combining biochemical, sonographic and doppler parameters. Prenatal and Neonatal Medicine 2001;6(2):95‐102. [Google Scholar]
Comas 2002a {published data only}
- Comas C, Torrents M, Munoz A, Antolin E, Figueras F, Echevarria M. Measurement of nuchal translucency as a single strategy in trisomy 21 screening: should we use any other marker?. Obstetrics & Gynecology 2002;100(4):648‐54. [DOI] [PubMed] [Google Scholar]
Comas 2002b {published data only}
- Comas C, Carrera JM. Early sonographic screening for chromosomal abnormalities. Ultrasound Review of Obstetrics and Gynecology 2002;2(2):88‐91. [Google Scholar]
Comstock 2006 {published data only}
- Comstock CH, Malone FD, Ball RH, Nyberg DA, Saade GR, Berkowitz RL, et al. Is there a nuchal translucency millimeter measurement above which there is no added benefit from first trimester serum screening?. American Journal of Obstetrics and Gynecology. 2006;195(3):843‐7. [DOI] [PubMed] [Google Scholar]
Conde 1998 {published data only}
- Conde‐Agudelo A, Kafury‐Goeta AC. Triple‐marker test as screening for down syndrome: A meta‐analysis. Obstetrical and Gynecological Survey 1998;53(6):369‐76. [DOI] [PubMed] [Google Scholar]
Cowans 2011 {published data only}
- Cowans NJ, Stamatopoulou A, Torring N, Spencer K. Early first‐trimester maternal serum placental growth factor in trisomy 21 pregnancies. Ultrasound in Obstetrics & Gynecology 2011;37(5):515‐9. [DOI] [PubMed] [Google Scholar]
Crossley 1991 {published data only}
- Crossley JA, Aitken DA, Connor JM. Prenatal screening for chromosome abnormalities using maternal serum chorionic gonadotrophin, alpha‐fetoprotein, and age. Prenatal Diagnosis 1991;11(2):83‐101. [DOI] [PubMed] [Google Scholar]
Crossley 1993 {published data only}
- Crossley JA, Aitken DA, Connor JM. Second‐trimester unconjugated oestriol levels in maternal serum from chromosomally abnormal pregnancies using an optimized assay.[see comment]. Prenatal Diagnosis 1993;13(4):271‐80. [DOI] [PubMed] [Google Scholar]
Crossley 1996 {published data only}
- Crossley JA, Berry E, Aitken DA, Connor JM. Insulin‐dependent diabetes mellitus and prenatal screening results: current experience from a regional screening programme. Prenatal Diagnosis 1996;16(11):1039‐42. [DOI] [PubMed] [Google Scholar]
Crossley 2002 {published data only}
- Crossley JA, Aitken DA, Waugh SM, Kelly T, Connor JM. Maternal smoking: age distribution, levels of alpha‐fetoprotein and human chorionic gonadotrophin, and effect on detection of Down syndrome pregnancies in second‐trimester screening. Prenatal Diagnosis 2002;22(3):247‐55. [DOI] [PubMed] [Google Scholar]
Cuckle 1984b {published data only}
- Cuckle HS, Wald NJ, Lindenbaum RH. Maternal serum alpha‐fetoprotein measurement: a screening test for Down syndrome. Lancet 1984;i(8383):926‐9. [DOI] [PubMed] [Google Scholar]
Cuckle 1987a {published data only}
- Cuckle HS, Wald NJ, Thompson SG. Estimating a woman's risk of having a pregnancy associated with Down's syndrome using her age and serum alpha‐fetoprotein level. British Journal of Obstetrics & Gynaecology 1987;94(5):387‐402. [DOI] [PubMed] [Google Scholar]
Cuckle 1987b {published data only}
- Cuckle HS, Nanchahal K, Wald NJ. Maternal serum alpha‐fetoprotein and ethnic origin. British Journal of Obstetrics and Gynaecology 1987;94(11):1111‐2. [DOI] [PubMed] [Google Scholar]
Cuckle 1990 {published data only}
- Cuckle HS, Wald NJ, Densem JW, Royston P, Knight GJ, Haddow JE, et al. The effect of smoking in pregnancy on maternal serum alpha‐fetoprotein, unconjugated oestriol, human chorionic gonadotrophin, progesterone and dehydroepiandrosterone sulphate levels. British Journal of Obstetrics & Gynaecology 1990;97(3):272‐4. [DOI] [PubMed] [Google Scholar]
Cuckle 1996a {published data only}
- Cuckle HS, Holding S, Jones R, Groome NP, Wallace EM. Combining Inhibin A with existing second‐trimester markers in maternal serum screening for Down's syndrome. Prenatal Diagnosis 1996;16(12):1095‐100. [DOI] [PubMed] [Google Scholar]
Cuckle 1999b {published data only}
- Cuckle HS, Sehmi I, Jones R, Evans LW. Maternal serum activin A and follistatin levels in pregnancies with Down syndrome. Prenatal Diagnosis 1999;19(6):513‐6. [PubMed] [Google Scholar]
Cuckle 1999c {published data only}
- Cuckle HS, Lith JM. Appropriate biochemical parameters in first‐trimester screening for Down syndrome.[see comment]. Prenatal Diagnosis 1999;19(6):505‐12. [DOI] [PubMed] [Google Scholar]
Cullen 1990 {published data only}
- Cullen MT, Gabrielli S, Green JJ, Rizzo N, Mahoney MJ, Salafia C, et al. Diagnosis and significance of cystic hygroma in the first trimester. Prenatal Diagnosis 1990;10(10):643‐51. [DOI] [PubMed] [Google Scholar]
Cusick 2004 {published data only}
- Cusick W, Provenzano J, Sullivan CA, Gallousis FM, Rodis JF. Fetal nasal bone length in euploid and aneuploid fetuses between 11 and 20 weeks' gestation: a prospective study. Journal of Ultrasound in Medicine 2004;23(10):1327‐33. [DOI] [PubMed] [Google Scholar]
Cusick 2007 {published data only}
- Cusick W, Shevell T, Duchan LS, Lupinacci CA, Terranova J, Crombleholme WR. Likelihood ratios for fetal trisomy 21 based on nasal bone length in the second trimester: how best to define hypoplasia?. Ultrasound in Obstetrics & Gynecology 2007;30(3):271‐4. [DOI] [PubMed] [Google Scholar]
Dancoine 2001 {published data only}
- Dancoine F, Couplet G, Mainardi A, Sukno F, Jaumain P, Nowak E, et al. Antenatal screening for Down's syndrome with serum markers: Influence of maternal weight, smoking habits and diabetes. Immuno‐Analyse et Biologie Specialisee 2001;16(6):381‐9. [Google Scholar]
Dane 2008 {published data only}
- Dane B, Dane C, Cetin A, Kiray M, Sivri D, Yayla M. Pregnancy outcome in fetuses with increased nuchal translucency. Journal of Perinatology 2008;28(6):400‐4. [DOI] [PubMed] [Google Scholar]
De Biasio, 1999 {published data only}
- De Biasio, Siccardi M, Volpe G, Famularo L, Santi F, Canini S. First‐trimester screening for down syndrome using nuchal translucency measurement with free ß‐hCG and PAPP‐A between 10 and 13 weeks of pregnancy ‐ The combined test. Prenatal Diagnosis 1999;19(4):360‐3. [DOI] [PubMed] [Google Scholar]
De Biasio, 2001 {published data only}
- De Biasio, Ferrero S, Prefumo F, Canini S, Marchini P, Bruzzone I, et al. Down's syndrome: First trimester approach. Italian Journal of Gynaecology and Obstetrics 2001;13(1):22‐6. [Google Scholar]
De Biasio 2000 {published data only}
- Biasio P, Canini S, Prefumo F, Famularo L, Venturini PL. Extent of correlation between first and second trimester markers for Down's syndrome screening. Journal of Medical Screening 2000;7(3):163. [DOI] [PubMed] [Google Scholar]
De Graaf 1991 {published data only}
- Graaf I, Cuckle HS, Pajkrt E, Leschot NJ, Bleker OP, Lith JM. Co‐variables in first trimester maternal serum screening. Prenatal Diagnosis 1991;20(3):186‐9. [PubMed] [Google Scholar]
De Graaf 1999 {published data only}
- Graaf I, Pajkrt E, Bilardo CM, Leschot NJ, Cuckle HS, Lith JM. Early pregnancy screening for fetal aneuploidy with serum markers and nuchal translucency. Prenatal Diagnosis 1999;19(5):458‐62. [DOI] [PubMed] [Google Scholar]
Del Carmen Saucedo 2009 {published data only}
- Carmen Saucedo M, DeVigan C, Vodovar V, Lelong N, Goffinet F, Khoshnood B. Measurement of nuchal translucency and the prenatal diagnosis of Down syndrome. Obstetrics & Gynecology 2009;114(4):829‐38. [DOI] [PubMed] [Google Scholar]
DeVore 2001 {published data only}
- DeVore GR, Romero R. Combined use of genetic sonography and maternal serum triple‐marker screening: an effective method for increasing the detection of trisomy 21 in women younger than 35 years.[see comment]. Journal of Ultrasound in Medicine 2001;20(6):645‐54. [DOI] [PubMed] [Google Scholar]
Dhaifalah 2007a {published data only}
- Dhaifalah I, Mickova I, Vrbicka D, Santavy J, Curtisova V. [Advanced maternal age as an indication for invasive prenatal diagnostics?]. [Czech]. Ceska Gynekologie 2007;72(3):181‐4. [PubMed] [Google Scholar]
Dhaifalah 2007b {published data only}
- Dhaifalah I, Mickova I, Santavy J, Vrbicka D, Zapletalova D, Curtisova V. [Efficiency of measuring nasal bone as an ultrasound marker of Down syndrome in 11th to 13th+6 week of pregnancy]. [Czech]. Ceska Gynekologie 2007;72(1):19‐23. [PubMed] [Google Scholar]
Dhallan 2007 {published data only}
- Dhallan R, Guo X, Emche S, Damewood M, Bayliss P, Cronin M, et al. A non‐invasive test for prenatal diagnosis based on fetal DNA present in maternal blood: a preliminary study. Lancet 2007;369(9560):474‐81. [DOI] [PubMed] [Google Scholar]
Dickerson 1994 {published data only}
- Dickerson VM. Multiple marker screening. Western Journal of Medicine 1994;161(2):161. [PMC free article] [PubMed] [Google Scholar]
Dimaio 1987 {published data only}
- Dimaio MS, Baumgarten A, Greenstein RM, Saal HM, Mahoney MJ. Screening for fetal Down's syndrome in pregnancy by measuring maternal serum alpha‐fetoprotein levels. New England Journal of Medicine 1987;317(6):342‐6. [DOI] [PubMed] [Google Scholar]
Doran 1986 {published data only}
- Doran TA, Cadesky K, Wong PY, Mastrogiacomo C, Capello T. Maternal serum alpha‐fetoprotein and fetal autosomal trisomies. American Journal of Obstetrics and Gynecology 1986;154(2):277‐81. [DOI] [PubMed] [Google Scholar]
Dreux 2008 {published data only}
- Dreux S, Olivier C, Dupont JM, Leporrier N, Study Group, Oury JF, et al. Maternal serum screening in cases of mosaic and translocation Down syndrome. Prenatal Diagnosis 2008;28(8):699‐703. [DOI] [PubMed] [Google Scholar]
Drugan 1996a {published data only}
- Drugan A, Reichler A, Bronstein M, Johnson MP, Sokol RJ, Evans MI. Abnormal biochemical serum screening versus 2nd‐trimester ultrasound‐detected minor anomalies as predictors of aneuploidy in low‐risk women. Fetal Diagnosis and Therapy 1996;11(5):301‐5. [DOI] [PubMed] [Google Scholar]
Drugan 1996b {published data only}
- Drugan A, O'Brien JE, Dvorin E, Krivchenia EL, Johnson MP, Sokol RJ, et al. Multiple marker screening in multifetal gestations: failure to predict adverse pregnancy outcomes. Fetal Diagnosis and Therapy 1996;11(1):16‐9. [DOI] [PubMed] [Google Scholar]
Drysdale 2002 {published data only}
- Drysdale K, Ridley D, Walker K, Higgins B, Dean T. First‐trimester pregnancy scanning as a screening tool for high‐risk and abnormal pregnancies in a district general hospital setting. Journal of Obstetrics & Gynaecology 2002;22(2):159‐65. [DOI] [PubMed] [Google Scholar]
Dugoff 2008 {published data only}
- Dugoff L, Cuckle HS, Hobbins JC, Malone FD, Belfort MA, Nyberg DA, et al. Prediction of patient‐specific risk for fetal loss using maternal characteristics and first‐ and second‐trimester maternal serum Down syndrome markers. American Journal of Obstetrics and Gynecology 2008;199(3):290‐6. [DOI] [PubMed] [Google Scholar]
Ebell 1999 {published data only}
- Ebell M. Is the integrated test better for screening for Down's syndrome than the traditional triple test?. Evidence‐Based Practice 1999;2(11):4‐5. [Google Scholar]
Economides 1998 {published data only}
- Economides DL, Whitlow BJ, Kadir R, Lazanakis M, Verdin SM. First trimester sonographic detection of chromosomal abnormalities in an unselected population. British Journal of Obstetrics & Gynaecology 1998;105(1):58‐62. [DOI] [PubMed] [Google Scholar]
Erickson 2004 {published data only}
- Erickson JA, Ashwood ER, Gin CA. Evaluation of a dimeric inhibin‐A assay for assessing fetal Down syndrome: establishment, comparison, and monitoring of median concentrations for normal pregnancies. Archives of Pathology & Laboratory Medicine 2004;128(4):415‐20. [DOI] [PubMed] [Google Scholar]
Evans 1996 {published data only}
- Evans MI, O'Brien JE, Dvorin E, Krivchenia EL, Drugan A, Hume RF Jr, et al. Similarity of insulin‐dependent diabetics' and non‐insulin‐dependent diabetics' levels of ß‐hCG and unconjugated estriol with controls: no need to adjust as with alpha‐fetoprotein. Journal of the Society for Gynecologic Investigation 1996;3(1):20‐2. [PubMed] [Google Scholar]
Evans 2007 {published data only}
- Evans MI, Galen RS. Comparison of serum markers in first‐trimester down syndrome screening. Obstetrics & Gynecology 2007;109(3):782. [DOI] [PubMed] [Google Scholar]
Falcon 2005 {published data only}
- Falcon O, Cavoretto P, Peralta CF, Csapo B, Nicolaides KH. Fetal head‐to‐trunk volume ratio in chromosomally abnormal fetuses at 11 + 0 to 13 + 6 weeks of gestation. Ultrasound in Obstetrics & Gynecology 2005;26(7):755‐60. [DOI] [PubMed] [Google Scholar]
Falcon 2006 {published data only}
- Falcon O, Faiola S, Huggon I, Allan L, Nicolaides KH. Fetal tricuspid regurgitation at the 11 + 0 to 13 + 6‐week scan: association with chromosomal defects and reproducibility of the method. Ultrasound in Obstetrics & Gynecology 2006;27(6):609‐12. [DOI] [PubMed] [Google Scholar]
Ford 1998 {published data only}
- Ford C, Moore AJ, Jordan PA, Bartlett WA, Wyldes MP, Jones AF, et al. The value of screening for Down's syndrome in a socioeconomically deprived area with a high ethnic population.[see comment]. British Journal of Obstetrics & Gynaecology 1998;105(8):855‐9. [DOI] [PubMed] [Google Scholar]
Frishman 1997 {published data only}
- Frishman GN, Canick JA, Hogan JW, Hackett RJ, Kellner LH, Saller DN Jr. Serum triple‐marker screening in in vitro fertilization and naturally conceived pregnancies. Obstetrics & Gynecology 1997;90(1):98‐101. [DOI] [PubMed] [Google Scholar]
Fukada 2000 {published data only}
- Fukada Y, Takizawa M, Amemiya A, Yoda H, Kohno K, Hoshi K. Detection of aneuploidy with fetal nuchal translucency and maternal serum markers in Japanese women. Acta Obstetricia et Gynecologica Scandinavica 2000;79(12):1124‐5. [PubMed] [Google Scholar]
Gaudry 2009 {published data only}
- Gaudry P, Lebbar A, Choiset A, Girard S, Lewin F, Tsatsaris V, et al. Is rapid aneuploidy screening used alone acceptable in prenatal diagnosis? An evaluation of the possible role of ultrasound examination. Fetal Diagnosis and Therapy 2009;25(2):285‐90. [DOI] [PubMed] [Google Scholar]
Gebb 2009 {published data only}
- Gebb J, Dar P. Should the first‐trimester aneuploidy screen be maternal age adjusted? Screening by absolute risk versus risk adjusted to maternal age. Prenatal Diagnosis 2009;29(3):245‐7. [DOI] [PubMed] [Google Scholar]
Geerts 2008 {published data only}
- Geerts L. Prenatal diagnosis of chromosomal abnormalities in a resource‐poor setting. International Journal of Gynaecology & Obstetrics 2008;103(1):16‐21. [DOI] [PubMed] [Google Scholar]
Geipel 2010 {published data only}
- Geipel A, Willruth A, Vieten J, Gembruch U, Berg C. Nuchal fold thickness, nasal bone absence or hypoplasia, ductus venosus reversed flow and tricuspid valve regurgitation in screening for trisomies 21, 18 and 13 in the early second trimester. Ultrasound in Obstetrics & Gynecology 2010;35(5):535‐9. [DOI] [PubMed] [Google Scholar]
Gekas 2009 {published data only}
- Gekas J, Gagne G, Bujold E, Douillard D, Forest JC, Reinharz D, et al. Comparison of different strategies in prenatal screening for Down's syndrome: cost effectiveness analysis of computer simulation. BMJ 2009;338:b138. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gekas 2011a {published data only}
- Gekas J, Berg DG, Durand A, Vallee M, Wildschut HI, Bujold E, et al. Rapid testing versus karyotyping in Down's syndrome screening: cost‐effectiveness and detection of clinically significant chromosome abnormalities. European Journal of Human Genetics 2011;19(1):3‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gekas 2011b {published data only}
- Gekas J, Durand A, Bujold E, Vallee M, Forest JC, Rousseau F, et al. Cost‐effectiveness and accuracy of prenatal Down syndrome screening strategies: should the combined test continue to be widely used?. American Journal of Obstetrics and Gynecology 2011;204(2):175‐8. [DOI] [PubMed] [Google Scholar]
Gerovassili 2007 {published data only}
- Gerovassili A, Garner C, Nicolaides KH, Thein SL, Rees DC. Free fetal DNA in maternal circulation: a potential prognostic marker for chromosomal abnormalities?. Prenatal Diagnosis 2007;27(2):104‐10. [DOI] [PubMed] [Google Scholar]
Ghidini 1998 {published data only}
- Ghidini A, Spong CY, Grier RE, Walker CN, Pezzullo JC. Is maternal serum triple screening a better predictor of Down syndrome in female than in male fetuses?. Prenatal Diagnosis 1998;18(2):123‐6. [DOI] [PubMed] [Google Scholar]
Goetzinger 2010 {published data only}
- Goetzinger KR, Dicke JM, Gray DL, Stamilio DM, Macones GA, Odibo AO. The effect of fetal gender in predicting Down syndrome using long bone ultrasonographic measurements. Prenatal Diagnosis 2010;30(10):950‐5. [DOI] [PubMed] [Google Scholar]
Goldie 1995 {published data only}
- Goldie DJ, Astley JP, Beaman JM, Bickley DA, Gunneberg A, Jones SR. Screening for Down's syndrome: the first two years experience in Bristol. Journal of Medical Screening 1995;2(4):207‐10. [DOI] [PubMed] [Google Scholar]
Gollo 2008 {published data only}
- Gollo CA, Murta CG, Bussamra LC, Santana RM, Moron AF. [Predictive value for fetal outcome of Doppler velocimetry of the ductus venosus between the 11th and the 14th gestation week]. [Portuguese]. Revista Brasileira de Ginecologia e Obstetricia 2008;30(1):5‐11. [DOI] [PubMed] [Google Scholar]
Gonçalves 2004 {published data only}
- Gonçalves LF, Espinoza J, Lee W, Schoen ML, Devers P, Mazor M, et al. Phenotypic characteristics of absent and hypoplastic nasal bones in fetuses with down syndrome: Description by 3‐dimensional ultrasonography and clinical significance. Journal of Ultrasound in Medicine 2004;23(12):1619‐27. [DOI] [PubMed] [Google Scholar]
Goodburn 1994 {published data only}
- Goodburn SF, Yates JR, Raggatt PR, Carr C, Ferguson‐Smith ME, Kershaw AJ, et al. Second‐trimester maternal serum screening using alpha‐fetoprotein, human chorionic gonadotrophin, and unconjugated oestriol: experience of a regional programme. Prenatal Diagnosis 1994;14(5):391‐402. [DOI] [PubMed] [Google Scholar]
Gorduza 2007 {published data only}
- Gorduza EV, Onofriescu M, Martiniuc V, Grigore M, Mihalceanu E, Iliev G. [FISH technique in aneuplodies prenatal diagnosis]. [Romanian]. Revista Medico‐Chirurgicala a Societatii de Medici Si Naturalisti Din Iasi 2007;111(4):990‐5. [PubMed] [Google Scholar]
Grace 2010 {published data only}
- Grace D, Eggers P, Glantz JC, Ozcan T. Mitral valve‐tricuspid valve distance as a sonographic marker of trisomy 21. Ultrasound in Obstetrics & Gynecology 2010;35(2):172‐7. [DOI] [PubMed] [Google Scholar]
Grati 2010 {published data only}
- Grati FR, Barlocco A, Grimi B, Milani S, Frascoli G, Meco AM, et al. Chromosome abnormalities investigated by non‐invasive prenatal testing account for approximately 50% of fetal unbalances associated with relevant clinical phenotypes. American Journal of Medical Genetics 2010;Part A. 152A(6):1434‐42. [DOI] [PubMed] [Google Scholar]
Gray 2009 {published data only}
- Gray DL, Dicke JM, Dickerson R, McCourt C, Odibo AO. Reevaluating humeral length for the detection of fetal trisomy 21. Journal of Ultrasound in Medicine 2009;28(10):1325‐30. [DOI] [PubMed] [Google Scholar]
Gregor 2007 {published data only}
- Gregor V, Sipek A, Horacek J. [Birth defects in the Czech Republic‐‐the prenatal diagnostic]. [Czech]. Ceska Gynekologie 2007;72(4):262‐8. [PubMed] [Google Scholar]
Gregor 2009 {published data only}
- Gregor V, Sipek A, Sipek A Jr, Horacek J, Langhammer P, Petrzilkova L, et al. [Prenatal diagnostics of chromosomal aberrations Czech Republic: 1994‐2007]. [Czech]. Ceska Gynekologie 2009;74(1):44‐54. [PubMed] [Google Scholar]
Grether 2009 {published data only}
- Grether Gonzalez P, Aguinaga Rios M, Colegio Mexicano de Especialistas en Ginecologia. [Prenatal genetic screening: biochemical markers of the first and second quarter]. [Spanish]. Ginecologia y Obstetricia de Mexico 2009;77(2):S27‐46. [PubMed] [Google Scholar]
Grozdea 2002 {published data only}
- Grozdea J, Farge F, Bourrouillou G, Calot M, Cambus JP, Valdiguie P. Maternal serum urea resistant alkaline phosphatase in Down syndrome pregnancy. Early Human Development 2002;67(1‐2):55‐9. [DOI] [PubMed] [Google Scholar]
Guo 2010 {published data only}
- Guo Q, Zhou Y, Wang X, Li Q. Simultaneous detection of trisomies 13, 18, and 21 with multiplex ligation‐dependent probe amplification‐based real‐time PCR. Clinical Chemistry 2010;56(9):1451‐9. [DOI] [PubMed] [Google Scholar]
Gyselaers 2004a {published data only}
- Gyselaers WJ, Vereecken AJ, Herck EJ, Straetmans DP, Martens GE, Jonge ET, et al. Screening for trisomy 21 in Flanders: a 10 years review of 40.490 pregnancies screened by maternal serum. European Journal of Obstetrics, Gynecology, & Reproductive Biology 2004;115(2):185‐9. [DOI] [PubMed] [Google Scholar]
Gyselaers 2004b {published data only}
- Gyselaers WJA, Vereecken AJ, Van Herck, Straetmans DPL, De Jonge, Ombelet WUA, et al. Single‐step maternal serum screening for trisomy 21 in the era of combined or integrated screening. Gynecologic and Obstetric Investigation 2004;58(4):221‐4. [DOI] [PubMed] [Google Scholar]
Gyselaers 2006a {published data only}
- Gyselaers WJ, Vereecken AJ, Herck EJ, Straetmans DP, Ombelet WU, Nijhuis JG. Nuchal translucency thickness measurements for fetal aneuploidy screening: Log NT‐MoM or Delta‐NT, performer‐specific medians and ultrasound training. Journal of Medical Screening 2006;13(1):4‐7. [DOI] [PubMed] [Google Scholar]
Gyselaers 2006b {published data only}
- Gyselaers WJ, Roets ER, Holsbeke CD, Vereecken AJ, Herck EJ, Straetmans DP, et al. Sequential triage in the first trimester may enhance advanced ultrasound scanning in population screening for trisomy 21. Ultrasound in Obstetrics & Gynecology 2006;27(6):622‐7. [DOI] [PubMed] [Google Scholar]
Hackshaw 1995 {published data only}
- Hackshaw AK, Densem J, Wald NJ. Repeat maternal serum testing for Down's syndrome screening using multiple markers with special reference to free alpha and free ß‐hCG. Prenatal Diagnosis 1995;15(12):1125‐30. [DOI] [PubMed] [Google Scholar]
Hackshaw 2001 {published data only}
- Hackshaw AK, Wald NJ. Repeat testing in antenatal screening for Down syndrome using dimeric inhibin‐A in combination with other maternal serum markers. Prenatal Diagnosis 2001;21(1):58‐61. [PubMed] [Google Scholar]
Haddow 1992 {published data only}
- Haddow JE, Palomaki GE, Knight GJ, Williams J, Pulkkinen A, Canick J, et al. Prenatal screening for Down's syndrome with use of maternal serum markers. New England Journal of Medicine 1992;327(9):588‐93. [DOI] [PubMed] [Google Scholar]
Hadzsiev 2007 {published data only}
- Hadzsiev K, Czako M, Veszpremi B, Kosztolanyi G. [Rapid diagnosis of fetal chromosomal abnormalities by fluorescence in situ hybridization]. [Hungarian]. Orvosi Hetilap 2007;148(30):1401‐4. [DOI] [PubMed] [Google Scholar]
Hafner 1995 {published data only}
- Hafner E, Schuchter K, Philipp K. Screening for chromosomal abnormalities in an unselected population by fetal nuchal translucency. Ultrasound in Obstetrics & Gynecology 1995;6(5):330‐3. [DOI] [PubMed] [Google Scholar]
Hallahan 1998 {published data only}
- Hallahan TW, Krantz DA, Tului L, Alberti E, Buchanan PD, Orlandi F, et al. Comparison of urinary free ß (hCG) and ß‐core (hCG) in prenatal screening for chromosomal abnormalities. Prenatal Diagnosis 1998;18(9):893‐900. [DOI] [PubMed] [Google Scholar]
Han 2008 {published data only}
- Han SH, An JW, Jeong GY, Yoon HR, Lee A, Yang YH, et al. Clinical and cytogenetic findings on 31,615 mid‐trimester amniocenteses. Korean Journal of Laboratory Medicine 2008;28(5):378‐85. [DOI] [PubMed] [Google Scholar]
Harper 2010 {published data only}
- Harper LM, Gray D, Dicke J, Stamilio DM, Macones GA, Odibo AO. Do race‐specific definitions of short long bones improve the detection of down syndrome on second‐trimester genetic sonograms?. Journal of Ultrasound in Medicine 2010;29(2):231‐5. [DOI] [PubMed] [Google Scholar]
Harrison 2006 {published data only}
- Harrison G, Goldie D. Second‐trimester Down's syndrome serum screening: double, triple or quadruple marker testing?. Annals of Clinical Biochemistry 2006;43(1):67‐72. [DOI] [PubMed] [Google Scholar]
Harry 2006 {published data only}
- Harry WG, Reed KL. Nuchal translucency and first‐trimester screening. Journal of the Society for Gynecologic Investigation 2006;13(3):153‐4. [DOI] [PubMed] [Google Scholar]
Hayashi 1995 {published data only}
- Hayashi M, Kozu H. Maternal urinary ß‐core fragment of hCG/creatinine ratios and fetal chromosomal abnormalities in the second trimester of pregnancy. Prenatal Diagnosis 1995;15(1):11‐6. [DOI] [PubMed] [Google Scholar]
Hayashi 1996 {published data only}
- Hayashi M, Kozu H, Takei H. Maternal urinary free ß‐subunit of human chorionic gonadotrophin: Creatinine ratios and fetal chromosomal abnormalities in the second trimester of pregnancy. British Journal of Obstetrics and Gynaecology 1996;103(6):577‐80. [DOI] [PubMed] [Google Scholar]
Heikkila 1997 {published data only}
- Heikkila A, Ryynanen M, Kirkinen P, Saarikoski S. Results and views of women in population‐wide pregnancy screening for trisomy 21 in east Finland. Fetal Diagnosis and Therapy 1997;12(2):93‐6. [DOI] [PubMed] [Google Scholar]
Heinig 2007 {published data only}
- Heinig J, Steinhard J, Schmitz R, Nofer JR, Kiesel L, Klockenbusch W. Maternal serum free beta‐hCG and PAPP‐A in patients with habitual abortion‐influence on first‐trimester screening for chromosomal abnormalities. Prenatal Diagnosis 2007;27(9):814‐6. [DOI] [PubMed] [Google Scholar]
Heinonen 1996 {published data only}
- Heinonen S, Ryynanen M, Kirkinen P, Hippelainen M, Saarikoski S. Effect of in vitro fertilization on human chorionic gonadotropin serum concentrations and Down's syndrome screening. Fertility and Sterility 1996;66(3):398‐403. [DOI] [PubMed] [Google Scholar]
Herman 2000 {published data only}
- Herman A, Weinraub Z, Dreazen E, Arieli S, Rozansky S, Bukovsky I, et al. Combined first trimester nuchal translucency and second trimester biochemical screening tests among normal pregnancies. Prenatal Diagnosis 2000;20(10):781‐4. [DOI] [PubMed] [Google Scholar]
Herman 2003 {published data only}
- Herman A, Dreazen E, Tovbin Y, Reish O, Bukovsky I, Maymon R. Correlation and overlapping between nuchal translucency and triple test among Down syndrome‐affected pregnancies. Fetal Diagnosis and Therapy 2003;18(3):196‐200. [DOI] [PubMed] [Google Scholar]
Herrou 1992 {published data only}
- Herrou M, Leporrier N, Leymarie P. Screening for fetal Down syndrome with maternal serum hCG and oestriol: a prospective study. Prenatal Diagnosis 1992;12(11):887‐92. [DOI] [PubMed] [Google Scholar]
Hershey 1985 {published data only}
- Hershey DW, Crandall BF, Schroth PS. Maternal serum alpha‐fetoprotein screening of fetal trisomies. American Journal of Obstetrics and Gynecology 1985;153(2):224‐5. [DOI] [PubMed] [Google Scholar]
Hershey 1986 {published data only}
- Hershey DW, Crandall BF, Perdue S. Combining maternal age and serum alpha‐fetoprotein to predict the risk of Down syndrome. Obstetrics & Gynecology 1986;68(2):177‐80. [PubMed] [Google Scholar]
Hewitt 1993 {published data only}
- Hewitt B. Nuchal translucency in the first trimester. Australian & New Zealand Journal of Obstetrics & Gynaecology 1993;33(4):389‐91. [DOI] [PubMed] [Google Scholar]
Hills 2010 {published data only}
- Hills A, Donaghue C, Waters J, Waters K, Sullivan C, Kulkarni A, et al. QF‐PCR as a stand‐alone test for prenatal samples: the first 2 years' experience in the London region. Prenatal Diagnosis 2010;30(6):509‐17. [DOI] [PubMed] [Google Scholar]
Ho 2010 {published data only}
- Ho SS, Choolani MA. FlashFISH: "same day" prenatal diagnosis of common chromosomal aneuploidies. Methods in Molecular Biology 2010;659:261‐8. [DOI] [PubMed] [Google Scholar]
Hogdall 1992 {published data only}
- Hogdall CK, Hogdall EV, Arends J, Norgaard‐Pedersen B, Smidt‐Jensen S, Larsen SO. CA‐125 as a maternal serum marker for Down's syndrome in the first and second trimesters. Prenatal Diagnosis 1992;12(3):223‐7. [DOI] [PubMed] [Google Scholar]
Hong Kong Practitioner {published data only}
- Anon. Screening tests in pregnancy. Hong Kong Practitioner 2001;23(10):461‐5. [Google Scholar]
Hoogendoorn 2008 {published data only}
- Hoogendoorn M, Evers SM, Schielen PC, Genugten ML, Wit GA, Ament AJ. Costs and effects of prenatal screening methods for Down syndrome and neural tube defects. Community Genetics 2008;11(6):359‐67. [DOI] [PubMed] [Google Scholar]
Howe 2000 {published data only}
- Howe DT, Gornall R, Wellesley D, Boyle T, Barber J. Six year survey of screening for Down's syndrome by maternal age and mid‐trimester ultrasound scans.[see comment]. BMJ 2000;320(7235):606‐10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hsiao 1991 {published data only}
- Hsiao KJ, Lee SY, Chuang HC. [Antenatal screening of maternal alpha‐fetoprotein with dried‐blood spot samples on filter paper]. [Chinese]. Journal of the Formosan Medical Association 1991;90(6):598‐604. [PubMed] [Google Scholar]
Hsieh 1999 {published data only}
- Hsieh TT, Hsu JJ, Lo LM, Liou JD, Soong YK. Maternal urine alpha‐fetoprotein concentrations between 14 and 21 weeks of gestation. Changgeng Yi Xue Za Zhi 1999;22(2):234‐9. [PubMed] [Google Scholar]
Hsu 1997a {published data only}
- Hsu JJ, Hsieh TT, Soong YK. Influence of maternal age and weight on second‐trimester serum alpha‐fetoprotein, total and free ß human chorionic gonadotropin levels. Changgeng Yi Xue Za Zhi 1997;20(3):181‐6. [PubMed] [Google Scholar]
Hsu 1998a {published data only}
- Hsu JJ, Hsieh TT, Hung TH, Chiang CH. Midtrimester maternal serum free ß‐human chorionic gonadotropin levels: normal reference values for Taiwanese women. Changgeng Yi Xue Za Zhi 1998;21(3):277‐82. [PubMed] [Google Scholar]
Hsu 1999b {published data only}
- Hsu JJ, Hsieh TT, Hung TH, Chen KC, Soong YK. Urine free ß‐human chorionic gonadotropin levels between 14 and 21 weeks of gestation in Taiwanese pregnancies. Changgeng Yi Xue Za Zhi 1999;22(1):11‐6. [PubMed] [Google Scholar]
Hu 2007 {published data only}
- Hu YL, Birth Defect Intervention Group of Jiangsu Province. [Serum screening of fetal chromosome abnormality during second pregnancy trimester: results of 26,803 pregnant women in Jiangsu Province]. [Chinese]. Chung‐Hua i Hsueh Tsa Chih [Chinese Medical Journal] 2007;87(35):2476‐80. [PubMed] [Google Scholar]
Huang 2003 {published data only}
- Huang T, Summers AM, Wyatt PR, Meier C, Cote GB. Maternal serum marker medians in Aboriginal Canadian women. Prenatal Diagnosis 2003;23(2):98‐100. [DOI] [PubMed] [Google Scholar]
Huang 2007a {published data only}
- Huang T, Boucher K, Summers AM. Second trimester prenatal screening for Down syndrome: the associations between the levels of serum markers in successive pregnancies. Prenatal Diagnosis 2007;27(12):1138‐42. [DOI] [PubMed] [Google Scholar]
Huang 2007b {published data only}
- Huang T, Wang FL, Boucher K, O'Donnell A, Rashid S, Summers AM. Racial differences in first trimester nuchal translucency. Prenatal Diagnosis 2007;27(12):1174‐6. [DOI] [PubMed] [Google Scholar]
Huggon 2004 {published data only}
- Huggon IC, Turan O, Allan LD. Doppler assessment of cardiac function at 11‐14 weeks' gestation in fetuses with normal and increased nuchal translucency. Ultrasound in Obstetrics & Gynecology 2004;24(4):390‐8. [DOI] [PubMed] [Google Scholar]
Hui 2003 {published data only}
- Hui PW, Tang MH, Lam YH, Ng EH, Yeung WS, Ho PC. Maternal serum hCG and alpha‐fetoprotein levels in pregnancies conceived after IVF or ICSI with fresh and frozen‐thawed embryos. Human Reproduction 2003;18(3):572‐5. [DOI] [PubMed] [Google Scholar]
Hui 2005 {published data only}
- Hui PW, Tang MH, Lam YH, Yeung WS, Ng EH, Ho PC. Nuchal translucency in pregnancies conceived after assisted reproduction technology. Ultrasound in Obstetrics & Gynecology 2005;25(3):234‐8. [DOI] [PubMed] [Google Scholar]
Hultén 2004 {published data only}
- Hultén M. Combined serum and nuchal translucency screening in the first trimester achieves 85% to 90% detection rate for Down and Edward syndromes. Evidence‐Based Healthcare 2004;8(2):82‐4. [Google Scholar]
Hung 2003 {published data only}
- Hung JH, Fu CY, Yuan CC, Chen CL, Yang ML, Shu LP, et al. Nuchal translucence incorporated into a one‐stage multifactorial screening model for Down syndrome prediction at second‐trimester pregnancy. Ultrasound in Medicine & Biology 2003;29(12):1667‐74. [DOI] [PubMed] [Google Scholar]
Hung 2008 {published data only}
- Hung JH, Fu CY, Chen CY, Chao KC, Hung J. Fetal nasal bone length and Down syndrome during the second trimester in a Chinese population. Journal of Obstetrics & Gynaecology Research 2008;34(4):518‐23. [DOI] [PubMed] [Google Scholar]
Hurley 1993 {published data only}
- Hurley PA, Ward RH, Teisner B, Iles RK, Lucas M, Grudzinskas JG. Serum PAPP‐A measurements in first‐trimester screening for Down syndrome. Prenatal Diagnosis 1993;13(10):903‐8. [DOI] [PubMed] [Google Scholar]
Huttly 2004 {published data only}
- Huttly W, Rudnicka A, Wald NJ. Second‐trimester prenatal screening markers for Down syndrome in women with insulin‐dependent diabetes mellitus. Prenatal Diagnosis 2004;24(10):804‐7. [DOI] [PubMed] [Google Scholar]
Hwa 2004 {published data only}
- Hwa HL, Yen MF, Hsieh FJ, Ko TM, Chen TH. Evaluation of second trimester maternal serum screening for Down's Syndrome using the Spiegelhalter‐Knill‐Jones (S‐KJ) approach. Journal of Perinatal Medicine 2004;32(5):407‐12. [DOI] [PubMed] [Google Scholar]
Iles 1996 {published data only}
- Iles RK. Urinary analysis for Down's syndrome: Is the measurement of urinary ß‐core the future of biochemical screening for Down's syndrome. Early Human Development 1996;47(Suppl.):S41‐S45. [DOI] [PubMed] [Google Scholar]
Ind 1994 {published data only}
- Ind TEJ, Iles RK, Cuckle HS, Chard T. Second trimester maternal serum placental alkaline phosphatase concentrations in Down's syndrome. Journal of Obstetrics and Gynaecology 1994;14(5):305‐8. [Google Scholar]
Ivorra‐Deleuze 2010 {published data only}
- Ivorra‐Deleuze D, Bretelle F, Heinemann M, Levy A, Toga C, Philip N, et al. [Combined screening for Down syndrome in Marseille multidisciplinary prenatal centers]. [French]. Gynecologie, Obstetrique & Fertilite 2010;38(12):786‐8. [DOI] [PubMed] [Google Scholar]
Jakobsen 2011 {published data only}
- Jakobsen TR, Sogaard K, Tabor A. Implications of a first trimester Down syndrome screening program on timing of malformation detection. Acta Obstetricia et Gynecologica Scandinavica 2011;90(7):728‐36. [DOI] [PubMed] [Google Scholar]
Jean‐Pierre 2005 {published data only}
- Jean P. Fetal nasal bone: Review of first trimester findings. Ultrasound Review of Obstetrics and Gynecology 2005;5(2):102‐4. [Google Scholar]
Johnson 1991 {published data only}
- Johnson A, Cowchock FS, Darby M, Wapner R, Jackson LG. First‐trimester maternal serum alpha‐fetoprotein and chorionic gonadotropin in aneuploid pregnancies. Prenatal Diagnosis 1991;11(7):443‐50. [DOI] [PubMed] [Google Scholar]
Johnson 1993 {published data only}
- Johnson MP, Johnson A, Holzgreve W, Isada NB, Wapner RJ, Treadwell MC, et al. First‐trimester simple hygroma: cause and outcome. American Journal of Obstetrics and Gynecology 1993;168(1):156‐61. [DOI] [PubMed] [Google Scholar]
Jorgensen 1999 {published data only}
- Jorgensen FS, Valentin L, Salvesen KA, Jorgensen C, Jensen FR, Bang J, et al. MULTISCAN‐‐a Scandinavian multicenter second trimester obstetric ultrasound and serum screening study. Acta Obstetricia et Gynecologica Scandinavica 1999;78(6):501‐10. [PubMed] [Google Scholar]
Jorgez 2007 {published data only}
- Jorgez CJ, Dang DD, Wapner R, Farina A, Simpson JL, Bischoff FZ. Elevated levels of total (maternal and fetal) beta‐globin DNA in maternal blood from first trimester pregnancies with trisomy 21. Human Reproduction 2007;22(8):2267‐72. [DOI] [PubMed] [Google Scholar]
Josefsson 1998 {published data only}
- Josefsson A, Molander E, Selbing A. Nuchal translucency as a screening test for chromosomal abnormalities in a routine first trimester ultrasound examination. Acta Obstetricia et Gynecologica Scandinavica 1998;77(5):497‐9. [PubMed] [Google Scholar]
Jou 2001 {published data only}
- Jou HJ, Shih JC, Wu SC, Li TC, Tzeng CY, Hsieh FJ. First‐trimester Down's syndrome screening by fetal nuchal translucency measurement in Taiwan. Journal of the Formosan Medical Association 2001;100(4):257‐61. [PubMed] [Google Scholar]
Jung 2007 {published data only}
- Jung E, Won HS, Lee PR, Kim A. Ultrasonographic measurement of fetal nasal bone length in the second trimester in Korean population. Prenatal Diagnosis 2007;27(2):154‐7. [DOI] [PubMed] [Google Scholar]
Jun‐Tao 2003 {published data only}
- Liu JT, Hao N, Sun NH, Wang FY, Xu YH, Gai MY, et al. [Screening by maternal serum markers for Down's syndrome]. [Chinese]. Chung‐Kuo i Hsueh Ko Hsueh Yuan Hsueh Pao Acta Academiae Medicinae Sinicae 2003;25(2):156‐9. [PubMed] [Google Scholar]
Kagan 2006 {published data only}
- Kagan KO, Avgidou K, Molina FS, Gajewska K, Nicolaides KH. Relation between increased fetal nuchal translucency thickness and chromosomal defects.[see comment]. Obstetrics & Gynecology 2006;107(1):6‐10. [DOI] [PubMed] [Google Scholar]
Kagan 2007 {published data only}
- Kagan KO, Frisova V, Nicolaides KH, Spencer K. Dose dependency between cigarette consumption and reduced maternal serum PAPP‐A levels at 11‐13+6 weeks of gestation. Prenatal Diagnosis 2007;27(9):849‐53. [DOI] [PubMed] [Google Scholar]
Kagan 2008 {published data only}
- Kagan KO, Anderson JM, Anwandter G, Neksasova K, Nicolaides KH. Screening for triploidy by the risk algorithms for trisomies 21, 18 and 13 at 11 weeks to 13 weeks and 6 days of gestation. Prenatal Diagnosis 2008;28(13):1209‐13. [DOI] [PubMed] [Google Scholar]
Kalelioglu 2007 {published data only}
- Kalelioglu IH. Humerus length measurement in Down syndrome screening. Clinical & Experimental Obstetrics & Gynecology 2007;34(2):93‐5. [PubMed] [Google Scholar]
Kautzmann 1995 {published data only}
- Kautzmann M, Solis RL, Luberta A, Fernandez JL, Navarro J, Rodriguez L, et al. Study of the efficiency of screening for trisomy 21 based on maternal serum levels of AFP and hCG combined with maternal age. Journal of Clinical Ligand Assay 1995;18(3):181‐5. [Google Scholar]
Kazerouni 2009 {published data only}
- Kazerouni NN, Currier B, Malm L, Riggle S, Hodgkinson C, Smith S, et al. Triple‐marker prenatal screening program for chromosomal defects. Obstetrics & Gynecology 2009;114(1):50‐8. [DOI] [PubMed] [Google Scholar]
Keith 1992 {published data only}
- Keith D. Maternal serum screening for neural tube defects and Down syndrome. Clinical Laboratory Science 1992;5(5):274‐6. [PubMed] [Google Scholar]
Kelekci 2004 {published data only}
- Kelekci S, Yazicioglu HF, Oguz S, Inan I, Yilmaz B, Sonmez S. Nasal bone measurement during the 1st trimester: is it useful?. Gynecologic & Obstetric Investigation 2004;58(2):91‐5. [DOI] [PubMed] [Google Scholar]
Kellner 1995a {published data only}
- Kellner LH, Weiner Z, Weiss RR, Neuer M, Martin GM, Mueenuddin M, et al. Triple marker (alpha‐fetoprotein, unconjugated estriol, human chorionic gonadotropin) versus alpha‐fetoprotein plus free‐ß subunit in second‐trimester maternal serum screening for fetal Down syndrome: a prospective comparison study.[see comment]. American Journal of Obstetrics and Gynecology 1995;173(4):1306‐9. [DOI] [PubMed] [Google Scholar]
Kellner 1995b {published data only}
- Kellner LH, Weiss RR, Weiner Z, Neuer M, Martin GM, Schulman H, et al. The advantages of using triple‐marker screening for chromosomal abnormalities. American Journal of Obstetrics and Gynecology 1995;172(3):831‐6. [DOI] [PubMed] [Google Scholar]
Kellner 1997 {published data only}
- Kellner LH, Canick JA, Palomaki GE, Neveux LM, Saller DN Jr, Walker RP, et al. Levels of urinary ß‐core fragment, total oestriol, and the ratio of the two in second‐trimester screening for Down syndrome. Prenatal Diagnosis 1997;17(12):1135‐41. [DOI] [PubMed] [Google Scholar]
Kirkegaard 2008 {published data only}
- Kirkegaard I, Petersen OB, Uldbjerg N, Torring N. Improved performance of first‐trimester combined screening for trisomy 21 with the double test taken before a gestational age of 10 weeks. Prenatal Diagnosis 2008;28(9):839‐44. [DOI] [PubMed] [Google Scholar]
Kjaergaard 2008 {published data only}
- Kjaergaard S, Hahnemann JM, Skibsted L, Jensen LN, Sperling L, Zingenberg H, et al. [Prenatal diagnosis of chromosome aberrations after implementation of screening for Down's syndrome]. [Danish]. Ugeskrift for Laeger 2008;170(14):1152‐6. [PubMed] [Google Scholar]
Knight 1990 {published data only}
- Knight GJ, Palomaki GE. Maternal serum alpha fetoprotein screening for fetal down syndrome. Journal of Clinical Immunoassay 1990;13(1):23‐9. [Google Scholar]
Knight 2001 {published data only}
- Knight GJ, Palomaki GE, Neveux LM, Haddow JE, Lambert‐Messerlian GM. Clinical validation of a new dimeric inhibin‐A assay suitable for second trimester Down's syndrome screening. Journal of Medical Screening 2001;8(1):2‐7. [DOI] [PubMed] [Google Scholar]
Knight 2005 {published data only}
- Knight GJ, Palomaki GE, Neveux LM, Smith DE, Kloza EM, Pulkkinen A, et al. Integrated serum screening for Down syndrome in primary obstetric practice. Prenatal Diagnosis 2005;25(12):1162‐7. [DOI] [PubMed] [Google Scholar]
Koos 2006 {published data only}
- Koos BJ. First‐trimester screening: Lessons from clinical trials and implementation. Current Opinion in Obstetrics and Gynecology 2006;18(2):152‐5. [DOI] [PubMed] [Google Scholar]
Kornman 1996 {published data only}
- Kornman LH, Morssink LP, Beekhuis JR, Wolf BT, Heringa MP, Mantingh A. Nuchal translucency cannot be used as a screening test for chromosomal abnormalities in the first trimester of pregnancy in a routine ultrasound practice.[see comment]. Prenatal Diagnosis 1996;16(9):797‐805. [DOI] [PubMed] [Google Scholar]
Kornman 1997 {published data only}
- Kornman LH, Morssink LP, Wortelboer MJ, Beekhuis JR, Wolf BT, Pratt JJ, et al. Maternal urinary ß‐core hCG in chromosomally abnormal pregnancies in the first trimester. Prenatal Diagnosis 1997;17(2):135‐9. [DOI] [PubMed] [Google Scholar]
Kotaska 2007 {published data only}
- Kotaska A. Prenatal screening for fetal aneuploidy. Journal of Obstetrics & Gynaecology Canada: JOGC 2007;29(6):499‐500. [DOI] [PubMed] [Google Scholar]
Kramer 1998 {published data only}
- Kramer RL, Yaron Y, O'Brien JE, Critchfield G, Ayoub M, Johnson MP, et al. Effect of adjustment of maternal serum alpha‐fetoprotein levels in insulin‐dependent diabetes mellitus. American Journal of Medical Genetics 1998;75(2):176‐8. [PubMed] [Google Scholar]
Krantz 1996 {published data only}
- Krantz DA, Larsen JW, Buchanan PD, Macri JN. First‐trimester Down syndrome screening: free ß‐human chorionic gonadotropin and pregnancy‐associated plasma protein A. American Journal of Obstetrics and Gynecology 1996;174(2):612‐6. [DOI] [PubMed] [Google Scholar]
Krantz 2005 {published data only}
- Krantz DA, Hallahan TW, Macri VJ, Macri JN. Maternal weight and ethnic adjustment within a first‐trimester Down syndrome and trisomy 18 screening program. Prenatal Diagnosis 2005;25(8):635‐40. [DOI] [PubMed] [Google Scholar]
Krantz 2007 {published data only}
- Krantz DA, Hallahan TW, Macri VJ, Macri JN. Genetic sonography after first‐trimester Down syndrome screening. Ultrasound in Obstetrics & Gynecology 2007;29(6):666‐70. [DOI] [PubMed] [Google Scholar]
Kulch 1993 {published data only}
- Kulch P, Keener S, Matsumoto M, Crandall BF. Racial differences in maternal serum human chorionic gonadotropin and unconjugated oestriol levels. Prenatal Diagnosis 1993;13(3):191‐5. [DOI] [PubMed] [Google Scholar]
Lai 1998 {published data only}
- Lai FM, Yeo GS. Down syndrome screening in Singapore‐‐the effectiveness of a second trimester serum screening policy modelled on 29,360 pregnancies in KK Women's and Children's Hospital. Singapore Medical Journal 1998;39(2):69‐75. [PubMed] [Google Scholar]
Lai 2003 {published data only}
- Lai TH, Chen SC, Tsai MS, Lee FK, Wei CF. First‐trimester screening for down syndrome in singleton pregnancies achieved by intrauterine insemination. Journal of Assisted Reproduction and Genetics 2003;20(8):327‐31. [DOI] [PMC free article] [PubMed] [Google Scholar]
Laigaard 2006a {published data only}
- Laigaard J, Cuckle H, Wewer UM, Christiansen M. Maternal serum ADAM12 levels in Down and Edwards' syndrome pregnancies at 9‐12 weeks' gestation. Prenatal Diagnosis 2006;26(8):689‐91. [DOI] [PubMed] [Google Scholar]
Laigaard 2006b {published data only}
- Laigaard J, Spencer K, Christiansen M, Cowans NJ, Larsen SO, Pedersen BN, et al. ADAM 12 as a first‐trimester maternal serum marker in screening for Down syndrome. Prenatal Diagnosis 2006;26(10):973‐9. [DOI] [PubMed] [Google Scholar]
Lam 1997 {published data only}
- Lam YH, Tang MH, Tang LC, Lee CP, Ho PK. Second‐trimester maternal urinary gonadotrophin peptide screening for fetal Down syndrome in Asian women. Prenatal Diagnosis 1997;17(12):1101‐6. [PubMed] [Google Scholar]
Lam 1998 {published data only}
- Lam YH, Ghosh A, Tang MH, Tang LC, Lee CP, Sin SY, et al. Second‐trimester maternal serum alpha‐fetoprotein and human chorionic gonadotrophin screening for Down's syndrome in Hong Kong. Prenatal Diagnosis 1998;18(6):585‐9. [PubMed] [Google Scholar]
Lam 1999a {published data only}
- Lam YH, Yeung WS, Tang MH, Ng EH, So WW, Ho PC. Maternal serum alpha‐fetoprotein and human chorionic gonadotrophin in pregnancies conceived after intracytoplasmic sperm injection and conventional in‐vitro fertilization. Human Reproduction 1999;14(8):2120‐3. [DOI] [PubMed] [Google Scholar]
Lam 1999b {published data only}
- Lam YH, Tang MH. Second‐trimester maternal serum inhibin‐A screening for fetal Down syndrome in Asian women. Prenatal Diagnosis 1999;19(5):463‐7. [DOI] [PubMed] [Google Scholar]
Lam 2000 {published data only}
- Lam YH, Tang MH, Lee CP, Sin SY, Tang R, Wong HS, et al. Acceptability of serum screening as an alternative to cytogenetic diagnosis of down syndrome among women 35 years or older in Hong Kong. Prenatal Diagnosis 2000;20(6):487‐90. [DOI] [PubMed] [Google Scholar]
Lam 2001 {published data only}
- Lam YH, Tang MH. The effect of fetal gender on second‐trimester maternal serum inhibin‐A concentration. Prenatal Diagnosis 2001;21(8):662‐4. [PubMed] [Google Scholar]
Lambert‐Messerlian 1996 {published data only}
- Lambert‐Messerlian GM, Canick JA, Palomaki GE, Schneyer AL. Second trimester levels of maternal serum Inhibin A , total inhibin, alpha Inhibin Aprecursor, and activin in Down's syndrome pregnancy. Journal of Medical Screening 1996;3(2):58‐62. [DOI] [PubMed] [Google Scholar]
Lambert‐Messerlian 1998 {published data only}
- Lambert Messerlian G, Luisi S, Florio P, Mazza V, Canick JA, Petraglia F. Second trimester levels of maternal serum total activin A and placental inhibin/activin alpha and ßA subunit messenger ribonucleic acids in Down syndrome pregnancy. European Journal of Endocrinology 1998;138(4):425‐9. [DOI] [PubMed] [Google Scholar]
Lauria 2007 {published data only}
- Lauria MR, Branch MD, LaCroix VH, Harris RD, Baker ER. Clinical impact of systematic genetic sonogram screening in a low‐risk population. Journal of Reproductive Medicine 2007;52(5):359‐64. [PubMed] [Google Scholar]
Lehavi 2005 {published data only}
- Lehavi O, Aizenstein O, Evans MI, Yaron Y. 2nd‐trimester maternal serum human chorionic gonadotropin and alpha‐fetoprotein levels in male and female fetuses with Down syndrome. Fetal Diagnosis and Therapy 2005;20(3):235‐8. [DOI] [PubMed] [Google Scholar]
Leung 2006 {published data only}
- Leung TY, Spencer K, Leung TN, Fung TY, Lau TK. Higher median levels of free ß‐hCG and PAPP‐A in the first trimester of pregnancy in a Chinese ethnic group. Implication for first trimester combined screening for Down's syndrome in the Chinese population. Fetal Diagnosis and Therapy 2006;21(1):140‐3. [DOI] [PubMed] [Google Scholar]
Leymarie 1993 {published data only}
- Leymarie P, Leporrier N. Maternal serum markers and prenatal screening for Down syndrome. Archives Francaises de Pediatrie 1993;50(5):455‐7. [PubMed] [Google Scholar]
Li 1998 {published data only}
- Li G, Huang X. [Clinical uses of maternal serum markers in the prenatal diagnosis] [Chinese]. Chung‐Hua Fu Chan Ko Tsa Chih 1998;33(4):252‐4. [PubMed] [Google Scholar]
Li 1999 {published data only}
- Li W, Zhou Y. [Measurement of pregnancy‐associated plasma protein A in maternal peripheral blood and Down syndrome] [Chinese]. Chung‐Hua Fu Chan Ko Tsa Chih 1999;34(10):631‐3. [PubMed] [Google Scholar]
Li 2010 {published data only}
- Li HW, Hui PW, Tang MH, Lau ET, Yeung WS, Ho PC, et al. Maternal serum anti‐Mullerian hormone level is not superior to chronological age in predicting Down syndrome pregnancies. Prenatal Diagnosis 2010;30(4):320‐4. [DOI] [PubMed] [Google Scholar]
Liao 1997 {published data only}
- Liao S, Wang Y, Ye G. [AFP, uE3, ß‐hCG levels applied for prenatal diagnosis of Down's syndrome]. [Chinese]. Chung‐Hua Fu Chan Ko Tsa Chih 1997;32(11):655‐8. [PubMed] [Google Scholar]
Liao 2001 {published data only}
- Liao AW, Heath V, Kametas N, Spencer K, Nicolaides KH. First‐trimester screening for trisomy 21 in singleton pregnancies achieved by assisted reproduction. Human Reproduction 2001;16(7):1501‐4. [DOI] [PubMed] [Google Scholar]
Lim 2002 {published data only}
- Lim KI, Pugash D, Dansereau J, Wilson RD. Nuchal index: a gestational age independent ultrasound marker for the detection of Down syndrome. Prenatal Diagnosis 2002;22(13):1233‐7. [DOI] [PubMed] [Google Scholar]
Lippman 1987 {published data only}
- Lippman A, Evans JA. Screening for maternal serum alpha‐fetoprotein: what about the low side?. CMAJ 1987;136(8):801‐4. [PMC free article] [PubMed] [Google Scholar]
Liu 2010 {published data only}
- Liu YH, Li LF, Wu YM. [Analysis of Down syndrome screening by maternal serum detection in mid‐pregnancy]. [Chinese]. Nan Fang Yi Ke Da Xue Xue Bao = Journal of Southern Medical University 2010;30(3):532‐4. [PubMed] [Google Scholar]
Lo 2010 {published data only}
- Lo TK, Lai FK, Leung WC, Lau WL, Tang LC, Chin RK. A new policy for prenatal screening and diagnosis of Down syndrome for pregnant women with advanced maternal age in a public hospital. Journal of Maternal‐Fetal & Neonatal Medicine 2010;23(8):914‐9. [DOI] [PubMed] [Google Scholar]
Lustig 1988 {published data only}
- Lustig L, Clarke S, Cunningham G, Schonberg R, Tompkinson G. California's experience with low MS‐AFP results. American Journal of Medical Genetics 1988;31(1):211‐22. [DOI] [PubMed] [Google Scholar]
Luthgens 2008 {published data only}
- Luthgens K. Comparison of the new PRC software with the established algorithm of the FMF UK for the detection of trisomy 21 and 18/13. Fetal Diagnosis and Therapy 2008;24(4):376‐84. [DOI] [PubMed] [Google Scholar]
MacDonald 1991 {published data only}
- MacDonald ML, Wagner RM, Slotnick RN. Sensitivity and specificity of screening for Down syndrome with alpha‐fetoprotein, hCG, unconjugated estriol, and maternal age.[see comment]. Obstetrics & Gynecology 1991;77(1):63‐8. [PubMed] [Google Scholar]
Macintosh 1994 {published data only}
- Macintosh MCM, Iles R, Teisner B, Sharma K, Chard T, Grudzinskas J, et al. Maternal serum human chorionic gonadotrophin and pregnancy‐associated plasma protein A, markers for fetal Down syndrome at 8‐14 weeks. Prenatal Diagnosis 1994;14(3):203‐8. [DOI] [PubMed] [Google Scholar]
Macintosh 1997 {published data only}
- Macintosh MCM, Nicolaides KH, Noble P, Chard T, Gunn L, Iles R. Urinary ß‐core hCG: Screening for aneuploidies in early pregnancy (11‐14 weeks' gestation). Prenatal Diagnosis 1997;17(5):401‐5. [PubMed] [Google Scholar]
MacRae 2010 {published data only}
- MacRae AR, Chodirker BN, Davies GA, Palomaki GE, Knight GJ, Minett J, et al. Second and first trimester estimation of risk for Down syndrome: implementation and performance in the SAFER study. Prenatal Diagnosis 2010;30(5):459‐66. [DOI] [PubMed] [Google Scholar]
Macri 1994 {published data only}
- Macri JN, Kasturi RV, Krantz DA, Cook EJ, Moore ND, Young JA, et al. Maternal serum Down syndrome screening: free ß‐protein is a more effective marker than human chorionic gonadotropin.[see comment]. American Journal of Obstetrics & Gynecology 1990;163(4):1248‐53. [DOI] [PubMed] [Google Scholar]
- Macri JN, Spencer K, Garver K, Buchanan PD, Say B, Carpenter NJ, et al. Maternal serum free ß hCG screening: results of studies including 480 cases of Down syndrome.[see comment]. Prenatal Diagnosis 1994;14(2):97‐103. [DOI] [PubMed] [Google Scholar]
- Spencer K, Macri JN. Early detection of Down's syndrome using free ß human choriogonadotropin. Annals of Clinical Biochemistry 1992;19(3):349‐50. [DOI] [PubMed] [Google Scholar]
Macri 1996 {published data only}
- Macri JN, Anderson RW, Krantz DA, Larsen JW, Buchanan PD. Prenatal maternal dried blood screening with alpha‐fetoprotein and free ß‐human chorionic gonadotropin for open neural tube defect and Down syndrome. American Journal of Obstetrics and Gynecology 1996;174(2):566‐72. [DOI] [PubMed] [Google Scholar]
Malone 1998 {published data only}
- Malone FD, D'Alton ME. Ultrasound clinics. Fetal nuchal fold translucency screening. Contemporary OB/GYN 1998;43(3):117‐8. [Google Scholar]
Malone 2003 {published data only}
- Malone FD, D'Alton ME. First‐trimester sonographic screening for Down syndrome. Obstetrics and Gynecology 2003;102(5):1066‐79. [DOI] [PubMed] [Google Scholar]
Mandryka‐Stankewycz 2009 {published data only}
- Mandryka‐Stankewycz S, Perenc M, Dec G, Sieroszewski P. [Noninvasive prenatal test in the first trimester of pregnancy (NT and estimation of beta‐hCG and PAPP‐A) in the diagnosis of fetal abnormalities in Polish population‐‐comparison of the biochemistry own normal ranges and literature reported data]. [Polish]. Ginekologia Polska 2009;80(11):851‐5. [PubMed] [Google Scholar]
Mangione 2001 {published data only}
- Mangione R, Guyon F, Taine L, Wen ZQ, Roux D, Vergnaud A, et al. Pregnancy outcome and prognosis in fetuses with increased first‐trimester nuchal translucency. Fetal Diagnosis and Therapy 2001;16(6):360‐3. [DOI] [PubMed] [Google Scholar]
Markov 2008 {published data only}
- Markov D, Dimitrova V. [Ultrasound screening for chromosomal anomalies by assessment of the fetal nasal bone during 11‐14 weeks of gestation‐‐a pilot study]. [Bulgarian]. Akusherstvo i Ginekologiia 2008;47(1):3‐9. [PubMed] [Google Scholar]
Maymon 2001a {published data only}
- Maymon R, Shulman A. Comparison of triple serum screening and pregnancy outcome in oocyte donation versus IVF pregnancies. Human Reproduction 2001;16(4):691‐5. [DOI] [PubMed] [Google Scholar]
Maymon 2001b {published data only}
- Maymon R, Dreazen E, Buckovsky I, Weinraub Z, Herman A. Does a 'notched' nuchal translucency indicate Down syndrome fetuses or other adverse pregnancy outcome?. Prenatal Diagnosis 2001;21(5):403‐8. [DOI] [PubMed] [Google Scholar]
Maymon 2002 {published data only}
- Maymon R, Shulman A. Serial first‐ and second‐trimester Down's syndrome screening tests among IVF‐versus naturally‐conceived singletons. Human Reproduction 2002;17(4):1081‐5. [DOI] [PubMed] [Google Scholar]
Maymon 2004 {published data only}
- Maymon R, Shulman A. Integrated first‐ and second‐trimester Down syndrome screening test among unaffected IVF pregnancies. Prenatal Diagnosis 2004;24(2):125‐9. [DOI] [PubMed] [Google Scholar]
Maymon 2005 {published data only}
- Maymon R, Cuckle H, Jones R, Reish O, Sharony R, Herman A. Predicting the result of additional second‐trimester markers from a woman's first‐trimester marker profile: A new concept in Down syndrome screening. Prenatal Diagnosis 2005;25(12):1102‐6. [DOI] [PubMed] [Google Scholar]
McDuffie 1996 {published data only}
- McDuffie RS Jr, Haverkamp AD, Stark CF, Haverkamp C, Barth CK. Prenatal screening using maternal serum alpha‐fetoprotein, human chorionic gonadotropin, and unconjugated estriol: Two‐year experience in a health maintenance organization. Journal of Maternal‐Fetal Medicine 1996;5(2):70‐3. [DOI] [PubMed] [Google Scholar]
Meier 2002 {published data only}
- Meier C, Huang T, Wyatt PR, Summers AM. Accuracy of expected risk of Down syndrome using the second‐trimester triple test. Clinical Chemistry 2002;48(4):653‐5. [PubMed] [Google Scholar]
Merkatz 1984 {published data only}
- Merkatz IR, Nitowsky HM, Macri JN, Johnson WE. An association between low maternal serum alpha‐fetoprotein and fetal chromosomal abnormalities. American Journal of Obstetrics and Gynecology 1984;148(7):886‐94. [DOI] [PubMed] [Google Scholar]
Merz 2005 {published data only}
- Merz E. The fetal nasal bone in the first trimester ‐ Precise assessment using 3D sonography. Ultraschall in der Medizin 2005;26(5):365‐6. [DOI] [PubMed] [Google Scholar]
Merz 2008 {published data only}
- Merz E, Thode C, Alkier A, Eiben B, Hackeloer BJ, Hansmann M, et al. A new approach to calculating the risk of chromosomal abnormalities with first‐trimester screening data. Ultraschall in der Medizin 2008;29(6):639‐45. [DOI] [PubMed] [Google Scholar]
Metzenbauer 2001 {published data only}
- Metzenbauer M, Hafner E, Hoefinger D, Schuchter K, Stangl G, Ogris E, et al. Three‐dimensional ultrasound measurement of the placental volume in early pregnancy: method and correlation with biochemical placenta parameters. Placenta 2001;22(6):602‐5. [DOI] [PubMed] [Google Scholar]
Metzenbauer 2002 {published data only}
- Metzenbauer M, Hafner E, Schuchter K, Philipp K. First‐trimester placental volume as a marker for chromosomal anomalies: preliminary results from an unselected population. Ultrasound in Obstetrics & Gynecology 2002;19(3):240‐2. [DOI] [PubMed] [Google Scholar]
Mikic 1999 {published data only}
- Mikic TS, Johnson P. Second trimester maternal serum ß human chorionic gonadotrophin and pregnancy outcome. British Journal of Obstetrics & Gynaecology 1999;106(6):598‐600. [DOI] [PubMed] [Google Scholar]
Miller 1991 {published data only}
- Miller CH, O'Brien TJ, Chatelain S, Butler BB, Quirk JG. Alteration in age‐specific risks for chromosomal trisomy by maternal serum alpha‐fetoprotein and human chorionic gonadotropin screening. Prenatal Diagnosis 1991;11(3):153‐8. [DOI] [PubMed] [Google Scholar]
Milunsky 1989 {published data only}
- Milunsky A, Jick SS, Bruell CL, Maclaughlin DS, Tsung Y‐K, Jick H, et al. Predictive values relative risks and overall benefits of high and low maternal serum alpha fetoprotein screening in singleton pregnancies ‐ new epidemiological data. American Journal of Obstetrics and Gynecology 1989;161(2):291‐7. [DOI] [PubMed] [Google Scholar]
Milunsky 1996 {published data only}
- Milunsky A, Nebiolo L. Maternal serum triple analyte screening and adverse pregnancy outcome. Fetal Diagnosis and Therapy 1996;11(4):249‐53. [DOI] [PubMed] [Google Scholar]
Minobe 2002 {published data only}
- Minobe S. [A study on the screening of prenatal trisomy 21 using the fucosylated alpha‐fetoprotein ratio measured by a liquid‐phase binding assay]. [Japanese]. Hokkaido Igaku Zasshi ‐ Hokkaido Journal of Medical Science 2002;77(6):527‐32. [PubMed] [Google Scholar]
Miron 2008 {published data only}
- Miron P, Cote YP, Lambert J. Effect of maternal smoking on prenatal screening for Down syndrome and trisomy 18 in the first trimester of pregnancy. Prenatal Diagnosis 2008;28(3):180‐5. [DOI] [PubMed] [Google Scholar]
Miron 2009 {published data only}
- Miron P, Cote YP, Lambert J. Nuchal translucency thresholds in prenatal screening for Down syndrome and trisomy 18. Journal of Obstetrics & Gynaecology Canada: JOGC 2009;31(3):227‐35. [DOI] [PubMed] [Google Scholar]
Miron 2010 {published data only}
- Miron P, Lambert J, Marcil A, Cowans NJ, Stamatopoulou A, Spencer K. Maternal plasma levels of follistatin‐related gene protein in the first trimester of pregnancies with Down syndrome. Prenatal Diagnosis 2010;30(3):224‐8. [DOI] [PubMed] [Google Scholar]
Miyamura 1999 {published data only}
- Miyamura T, Saito N, Touno A, Nagata S, Hidaki T, Ishimaru T, et al. Multicenter study for maternal serum triple markers to establish Japanese standards: Maternal serum marker study group, Japan Association of Prenatal Diagnostics. Acta Obstetrica et Gynaecologica Japonica 1999;51(11):1042‐18. [Google Scholar]
Moghadam 1998 {published data only}
- Moghadam S, Engel W, Bougoussa M, Hennen G, Igout A, Sancken U. Maternal serum placental growth hormone and insulinlike growth factor binding proteins 1 and 3 in pregnancies affected by fetal aneuploidy and other abnormalities: implications for prenatal diagnosis of trisomy 21. Fetal Diagnosis and Therapy 1998;13(5):291‐7. [DOI] [PubMed] [Google Scholar]
Monni 2000 {published data only}
- Monni G, Zoppi MA, Ibba RM, Putzolu M, Floris M. Nuchal translucency in multiple pregnancies. Croatian Medical Journal 2000;41(3):266‐9. [PubMed] [Google Scholar]
Monni 2002 {published data only}
- Monni G, Zoppi MA. New ultrasonographic markers of aneuploidies: Nasal bones. Ultrasound Review of Obstetrics and Gynecology 2002;2(4):229‐34. [Google Scholar]
Mooney 1994 {published data only}
- Mooney RA, Peterson CJ, French CA, Saller DN Jr, Arvan DA. Effectiveness of combining maternal serum alpha‐fetoprotein and hCG in a second‐trimester screening program for Down syndrome. Obstetrics and Gynecology 1994;84(2):298‐303. [PubMed] [Google Scholar]
Muhcu 2008 {published data only}
- Muhcu M, Mungen E, Atay V, Ipcioglu OM, Dundar O, Ergur R, et al. First trimester screening for Down syndrome in rhesus negative women. Prenatal Diagnosis 2008;28(5):404‐7. [DOI] [PubMed] [Google Scholar]
Muller 1994 {published data only}
- Muller F, Bussieres L, Pelissier MC, Oury JF, Boue C, Uzan S, et al. Do racial differences exist in second‐trimester maternal hCG levels? A study of 23,369 women. Prenatal Diagnosis 1994;14(7):633‐6. [DOI] [PubMed] [Google Scholar]
Muller 1996 {published data only}
- Muller F, Dommergues M, Bussieres L, Aegerter P, Fiblec B, Uzan S, et al. Prenatal screening for Down syndrome: should first trimester ultrasound replace maternal serum screening?. Early Human Development 1996;47(Suppl):S37‐9. [DOI] [PubMed] [Google Scholar]
Muller 1999 {published data only}
- Muller F, Ngo S, Rebiffe M, Oury JF, Uzan S, Satge D. Maternal serum s100b protein is ineffective for Down syndrome screening. Prenatal Diagnosis 1999;19(11):1086. [DOI] [PubMed] [Google Scholar]
Muller 2002a {published data only}
- Muller F, Dreux S, Oury JF, Luton D, Uzan S, Uzan M, et al. Down syndrome maternal serum marker screening after 18 weeks' gestation. Prenatal Diagnosis 2002;22(11):1001‐4. [DOI] [PubMed] [Google Scholar]
Muller 2002b {published data only}
- Muller F, Forestier F, Dingeon B, ABA Study Group. Second trimester trisomy 21 maternal serum marker screening. Results of a countrywide study of 854,902 women.[see comment]. Prenatal Diagnosis 2002;22(10):925‐9. [DOI] [PubMed] [Google Scholar]
Muller 2003 {published data only}
- Muller F, Dreux S, Lemeur A, Sault C, Desgres J, Bernard MA, et al. Medically assisted reproduction and second‐trimester maternal serum marker screening for Down syndrome. Prenatal Diagnosis 2003;23(13):1073‐6. [DOI] [PubMed] [Google Scholar]
Murta 2002 {published data only}
- Murta CG, Moron AF, Avila MA, Weiner CP. Application of ductus venosus Doppler velocimetry for the detection of fetal aneuploidy in the first trimester of pregnancy. Fetal Diagnosis and Therapy 2002;17(5):308‐14. [DOI] [PubMed] [Google Scholar]
Musone 2000 {published data only}
- Musone R, Bonafiglia R, Menditto A, Paccone M, Cassese E, Russo G, et al. Fetuses with cystic hygroma. A retrospective study. Panminerva Medica 2000;42(1):39‐43. [PubMed] [Google Scholar]
Musto 1986 {published data only}
- Musto JD, Pizzolante JM, Chesarone VP, Sassi AM, Sane R. Alpha‐fetoprotein: an enhanced‐sensitivity assay for neural tube defect and Down syndrome evaluation. Clinical Chemistry 1986;32(7):1412. [PubMed] [Google Scholar]
Myrick 1990 {published data only}
- Myrick JE, Caudill SP, Hubert IL, Robinson MK, Adams MJ Jr, Pueschel SM. Identification of haptoglobin alpha‐2FF variants in mid‐trimester maternal serum as potential markers for Down syndrome. Applied & Theoretical Electrophoresis 1990;1(5):233‐41. [PubMed] [Google Scholar]
Naidoo 2008 {published data only}
- Naidoo P, Erasmus I, Jeebodh J, Nicolaou E, Gelderen CJ. Nuchal translucency as a method of first‐trimester screening for aneuploidy. South African Medical Journal 2008;Suid‐Afrikaanse Tydskrif Vir Geneeskunde. 98(4):295‐9. [PubMed] [Google Scholar]
Nau 2009 {published data only}
- Nau JY. [Screening for trisomy 21 in France]. [French]. Revue Medicale Suisse 2009;5(211):1531. [PubMed] [Google Scholar]
Nau 2009a {published data only}
- Nau JY. [Trisomy 21, after a half century]. [French]. Revue Medicale Suisse 2009;5(190):380. [PubMed] [Google Scholar]
Neveux 1996a {published data only}
- Neveux LM, Palomaki GE, Larrivee DA, Knight GJ, Haddow JE. Refinements in managing maternal weight adjustment for interpreting prenatal screening results. Prenatal Diagnosis 1996;16(12):1115‐9. [DOI] [PubMed] [Google Scholar]
Neveux 1996b {published data only}
- Neveux LM, Palomaki GE, Knight GJ, Haddow JE. Multiple marker screening for Down syndrome in twin pregnancies. Prenatal Diagnosis 1996;16(1):29‐34. [DOI] [PubMed] [Google Scholar]
Ng 2004 {published data only}
- Ng EK, El‐Sheikhah A, Chiu RW, Chan KC, Hogg M, Bindra R, et al. Evaluation of human chorionic gonadotropin ß‐subunit mRNA concentrations in maternal serum in aneuploid pregnancies: a feasibility study. Clinical Chemistry 2004;50(6):1055‐7. [DOI] [PubMed] [Google Scholar]
Nicolaides 1992 {published data only}
- Nicolaides KH, Zar G, Snijders RJM, Gosden CM. Fetal nuchal oedema associated malformations and chromosomal defects. Fetal Diagnosis and Therapy 1992;7(2):123‐31. [DOI] [PubMed] [Google Scholar]
Nicolaides 2000 {published data only}
- Nicolaides KH, Cicero S, Liao AW. One‐stop clinic for assessment of risk of chromosomal defects at 12 weeks of gestation. Prenatal and Neonatal Medicine 2000;5(3):145‐54. [DOI] [PubMed] [Google Scholar]
Nicolaides 2004 {published data only}
- Nicolaides KH. Nuchal translucency and other first‐trimester sonographic markers of chromosomal abnormalities. American Journal of Obstetrics and Gynecology 2004;191(1):45‐67. [DOI] [PubMed] [Google Scholar]
Nicolaides 2005a {published data only}
- Nicolaides KH, Wegrzyn P. [First trimester diagnosis of chromosomal defects][Polish]. Ginekologia Polska 2005;76(1):1‐8. [PubMed] [Google Scholar]
Nicolaides 2005b {published data only}
- Nicolaides KH, Wegrzyn P. [Sonographic features of chromosomal defects at 11(+0) to 13(+6) weeks of gestation] [Polish]. Ginekologia Polska 2005;76(6):423‐30. [PubMed] [Google Scholar]
Nicolaides 2005c {published data only}
- Nicolaides KH, Wegrzyn P. [Increased nuchal translucency with normal karyotype]. [Polish]. Ginekologia Polska 2005;76(8):593‐601. [PubMed] [Google Scholar]
Nicolaides 2005d {published data only}
- Nicolaides KH, Wegrzyn P. [Fetal nuchal translucency]. [Polish]. Ginekologia Polska 2005;76(3):179‐86. [PubMed] [Google Scholar]
Nicolaides 2005e {published data only}
- Nicolaides KH, Wegrzyn P. [Fetal nuchal translucency thickness and risk for chromosomal defects]. [Polish]. Ginekologia Polska 2005;76(4):257‐63. [PubMed] [Google Scholar]
Nicolaides 2005f {published data only}
- Nicolaides Kypros H. First‐trimester screening for chromosomal abnormalities. Seminars in Perinatology (Philadelphia) 2005;29(4):190‐4. [DOI] [PubMed] [Google Scholar]
Niemimaa 2001 {published data only}
- Niemimaa M, Heinonen S, Seppala M, Hippelainen M, Martikainen H, Ryynanen M. First‐trimester screening for Down's syndrome in in vitro fertilization pregnancies. Fertility & Sterility 2001;76(6):1282‐3. [DOI] [PubMed] [Google Scholar]
Niemimaa 2002 {published data only}
- Niemimaa M, Suonpaa M, Heinonen S, Seppala M, Bloigu R, Ryynanen M. Maternal serum human chorionic gonadotrophin and pregnancy‐associated plasma protein A in twin pregnancies in the first trimester. Prenatal Diagnosis 2002;22(3):183‐5. [PubMed] [Google Scholar]
Niemimaa 2003 {published data only}
- Niemimaa M, Heinonen S, Seppala M, Ryynanen M. The influence of smoking on the pregnancy‐associated plasma protein A, free ß human chorionic gonadotrophin and nuchal translucency. BJOG: an international journal of obstetrics and gynaecology 2003;110(7):664‐7. [PubMed] [Google Scholar]
Noble 1997 {published data only}
- Noble PL, Snijders RJ, Abraha HD, Sherwood RA, Nicolaides KH. Maternal serum free ß‐hCG at 10 to 14 weeks of gestation in trisomic twin pregnancies. British Journal of Obstetrics & Gynaecology 1997;104(6):741‐3. [DOI] [PubMed] [Google Scholar]
Norgaard 1990 {published data only}
- Norgaard Pedersen B, Larsen SO, Arends J, Svenstrup B, Tabor A. Maternal serum markers in screening for Down syndrome. Clinical Genetics 1990;37(1):35‐43. [DOI] [PubMed] [Google Scholar]
Norton 1992 {published data only}
- Norton ME, Golbus MS. Maternal serum CA 125 for aneuploidy detection in early pregnancy. Prenatal Diagnosis 1992;12(9):779‐81. [DOI] [PubMed] [Google Scholar]
Novakov‐Mikic 2007 {published data only}
- Novakov‐Mikic A, Potic Z, Pjevic A. [Ultrasound screening program for chromosomal abnormalities‐‐the first 2000 women]. [Serbian]. Medicinski Pregled 2007;60(1‐2):66‐70. [DOI] [PubMed] [Google Scholar]
O'Brien 1997a {published data only}
- O'Brien JE, Dvorin E, Yaron Y, Ayoub M, Johnson MP, Hume RF Jr, et al. Differential increases in AFP, hCG, and uE3 in twin pregnancies: Impact on attempts to quantify Down syndrome screening calculations. American Journal of Medical Genetics 1997;73(2):109‐12. [DOI] [PubMed] [Google Scholar]
O'Brien 1997b {published data only}
- Brien JE, Dvorin E, Drugan A, Johnson MP, Yaron Y, Evans MI. Race‐ethnicity‐specific variation in multiple‐marker biochemical screening: Alpha‐fetoprotein, hCG, and estriol. Obstetrics and Gynecology 1997;89(3):355‐8. [DOI] [PubMed] [Google Scholar]
Odibo 2004 {published data only}
- Odibo AO, Sehdev HM, Dunn L, McDonald R, Macones GA. The association between fetal nasal bone hypoplasia and aneuploidy. Obstetrics & Gynecology 2004;104(6):1229‐33. [DOI] [PubMed] [Google Scholar]
Odibo 2007 {published data only}
- Odibo AO, Sehdev HM, Stamilio DM, Cahill A, Dunn L, Macones GA. Defining nasal bone hypoplasia in second‐trimester Down syndrome screening: does the use of multiples of the median improve screening efficacy?. American Journal of Obstetrics and Gynecology 2007;197(4):361‐4. [DOI] [PubMed] [Google Scholar]
Odibo 2008 {published data only}
- Odibo AO, Sehdev HM, Gerkowicz S, Stamilio DM, Macones GA. Comparison of the efficiency of second‐trimester nasal bone hypoplasia and increased nuchal fold in Down syndrome screening. American Journal of Obstetrics and Gynecology 2008;199(3):281‐5. [DOI] [PubMed] [Google Scholar]
Odibo 2009 {published data only}
- Odibo AO, Schoenborn JA, Haas K, Macones GA. Does the combination of fronto‐maxillary facial angle and nasal bone evaluation improve the detection of Down syndrome in the second trimester?. Prenatal Diagnosis 2009;29(10):947‐51. [DOI] [PubMed] [Google Scholar]
Offerdal 2008 {published data only}
- Offerdal K, Blaas HG, Eik‐Nes SH. Prenatal detection of trisomy 21 by second‐trimester ultrasound examination and maternal age in a non‐selected population of 49 314 births in Norway. Ultrasound in Obstetrics & Gynecology 2008;32(4):493‐500. [DOI] [PubMed] [Google Scholar]
Ognibene 1999 {published data only}
- Ognibene A, Ciuti R, Tozzi P, Messeri G. Maternal serum superoxide dismutase (SOD): a possible marker for screening Down syndrome affected pregnancies.[see comment]. Prenatal Diagnosis 1999;19(11):1058‐60. [PubMed] [Google Scholar]
Oh 2007 {published data only}
- Oh C, Harman C, Baschat AA. Abnormal first‐trimester ductus venosus blood flow: a risk factor for adverse outcome in fetuses with normal nuchal translucency. Ultrasound in Obstetrics & Gynecology 2007;30(2):192‐6. [DOI] [PubMed] [Google Scholar]
Olajide 1989 {published data only}
- Olajide F, Kitau MJ, Chard T. Maternal serum AFP levels in the first trimester of pregnancy. European Journal of Obstetrics, Gynecology, & Reproductive Biology 1989;30(2):123‐8. [DOI] [PubMed] [Google Scholar]
Onda 1996 {published data only}
- Onda T, Kitagawa M, Takeda O, Sago H, Kubonoya K, Iinuma K, et al. Triple marker screening in native Japanese women. Prenatal Diagnosis 1996;16(8):713‐7. [DOI] [PubMed] [Google Scholar]
Onda 1998 {published data only}
- Onda T, Tanaka T, Takeda O, Kitagawa M, Kuwabara Y, Yamamoto H, et al. Agreement between predicted risk and prevalence of Down syndrome in second‐trimester triple‐marker screening in Japan. Prenatal Diagnosis 1998;18(9):956‐8. [PubMed] [Google Scholar]
Onda 2000 {published data only}
- Onda T, Tanaka T, Yoshida K, Nakamura Y, Kudo R, Yamamoto H, et al. Triple marker screening for trisomy 21, trisomy 18 and open neural tube defects in singleton pregnancies of native Japanese pregnant women. Journal of Obstetrics & Gynaecology Research 2000;26(6):441‐7. [DOI] [PubMed] [Google Scholar]
Orlandi 2002 {published data only}
- Orlandi F, Rossi C, Allegra A, Krantz D, Hallahan T, Orlandi E, et al. First trimester screening with free ß‐hCG, PAPP‐A and nuchal translucency in pregnancies conceived with assisted reproduction. Prenatal Diagnosis 2002;22(8):718‐21. [DOI] [PubMed] [Google Scholar]
Ottavio 1997 {published data only}
- Ottavio GD, Meir YJ, Rustico MA, Pecile V, Fischer‐Tamaro L, Conoscenti G, et al. Screening for fetal anomalies by ultrasound at 14 and 21 weeks. Ultrasound in Obstetrics and Gynecology 1997;10(6):375‐80. [DOI] [PubMed] [Google Scholar]
Ozkaya 2010 {published data only}
- Ozkaya O, Sezik M, Ozbasar D, Kaya H. Abnormal ductus venosus flow and tricuspid regurgitation at 11‐14 weeks' gestation have high positive predictive values for increased risk in first‐trimester combined screening test: results of a pilot study. Taiwanese Journal of Obstetrics & Gynecology 2010;49(2):145‐50. [DOI] [PubMed] [Google Scholar]
Páez 2004 {published data only}
- Páez L, Peña E, González F, Bello F, Bellorín J, Espinoza F, et al. Plasma protein "A" and chorionic gonadotropin at first trimester pregnancy. Informe Medico 2004;6(2):99‐109. [Google Scholar]
Paladini 2007 {published data only}
- Paladini D, Sglavo G, Penner I, Pastore G, Nappi C. Fetuses with Down syndrome have an enlarged anterior fontanelle in the second trimester of pregnancy. Ultrasound in Obstetrics & Gynecology 2007;30(6):824‐9. [DOI] [PubMed] [Google Scholar]
Palka 1998 {published data only}
- Palka G, Guanciali Franchi P, Papponetti M, Marcuccitti J, Morizio E, Calabrese G, et al. Prenatal diagnosis using the triple test. Minerva Ginecologica 1998;50(10):411‐5. [PubMed] [Google Scholar]
Palomaki 1989 {published data only}
- Palomaki GE, Williams J, Haddow JE. Combining maternal serum alpha‐fetoprotein measurements and age to screen for Down syndrome in pregnant women under age 35. American Journal of Obstetrics and Gynecology 1989;160(3):575‐81. [DOI] [PubMed] [Google Scholar]
Palomaki 1993 {published data only}
- Palomaki GE, Knight GJ, Haddow JE, Canick JA, Wald NJ, Kennard A. Cigarette smoking and levels of maternal serum alpha‐fetoprotein, unconjugated estriol, and hCG: Impact on Down syndrome screening. Obstetrics and Gynecology 1993;81(5):675‐8. [PubMed] [Google Scholar]
Palomaki 1994 {published data only}
- Palomaki GE, Knight GJ, Haddow JE. Human chorionic gonadotropin and unconjugated oestriol measurements in insulin‐dependent diabetic pregnant women being screened for fetal Down syndrome. Prenatal Diagnosis 1994;14(1):65‐8. [DOI] [PubMed] [Google Scholar]
Palomaki 1996 {published data only}
- Palomaki GE, Neveux LM, Haddow JE. Can reliable Down's syndrome detection rates be determined from prenatal screening intervention trials?. Journal of Medical Screening 1996;3(1):12‐7. [DOI] [PubMed] [Google Scholar]
Palomaki 2005 {published data only}
- Palomaki GE, Knight GJ, Neveux LM, Pandian R, Haddow JE. Maternal serum invasive trophoblast antigen and first‐trimester Down syndrome screening. Clinical Chemistry 2005;51(8):1499‐504. [DOI] [PubMed] [Google Scholar]
Panburana 2001 {published data only}
- Panburana P, Ajjimakorn S, Tungkajiwangoon P. First trimester Down Syndrome screening by nuchal translucency in a Thai population. International Journal of Gynaecology & Obstetrics 2001;75(3):311‐2. [DOI] [PubMed] [Google Scholar]
Pandya 1994 {published data only}
- Pandya PP, Brizot ML, Kuhn P, Snijders RJ, Nicolaides KH. First‐trimester fetal nuchal translucency thickness and risk for trisomies. Obstetrics & Gynecology 1994;84(3):420‐3. [PubMed] [Google Scholar]
Pandya 1995b {published data only}
- Pandya PP, Santiago C, Snijders RJM, Nicolaides KH. First trimester fetal nuchal translucency. Current Opinion in Obstetrics and Gynecology 1995;7(2):95‐102. [DOI] [PubMed] [Google Scholar]
Papadopoulou 2008 {published data only}
- Papadopoulou E, Sifakis S, Giahnakis E, Fragouli Y, Karkavitsas N, Koumantakis E, et al. Human placental growth hormone is increased in maternal serum in pregnancies affected by Down syndrome. Fetal Diagnosis and Therapy 2008;23(3):211‐6. [DOI] [PubMed] [Google Scholar]
Parra‐Cordero 2007 {published data only}
- Parra‐Cordero M, Quiroz L, Rencoret G, Pedraza D, Munoz H, Soto‐Chacon E, et al. Screening for trisomy 21 during the routine second‐trimester ultrasound examination in an unselected Chilean population. Ultrasound in Obstetrics & Gynecology 2007;30(7):946‐51. [DOI] [PubMed] [Google Scholar]
Paterlini‐Brechot 2007 {published data only}
- Paterlini‐Brechot P. [Non invasive prenatal diagnosis of trisomy 21: dream or reality?]. [French]. M S‐Medecine Sciences 2007;23(6‐7):592‐4. [DOI] [PubMed] [Google Scholar]
Paul 2001 {published data only}
- Paul C, Krampl E, Skentou C, Jurkovic D, Nicolaides KH. Measurement of fetal nuchal translucency thickness by three‐dimensional ultrasound. Ultrasound in Obstetrics & Gynecology 2001;18(5):481‐4. [DOI] [PubMed] [Google Scholar]
Peralta 2005 {published data only}
- Peralta CF, Falcon O, Wegrzyn P, Faro C, Nicolaides KH. Assessment of the gap between the fetal nasal bones at 11 to 13 + 6 weeks of gestation by three‐dimensional ultrasound. Ultrasound in Obstetrics & Gynecology 2005;25(5):464‐7. [DOI] [PubMed] [Google Scholar]
Perenc 1998 {published data only}
- Perenc M, Dudarewicz L, Kaluzewski B. Analysis of triple test results in 27 cases of twin pregnancies. Acta Geneticae Medicae et Gemellologiae 1998;47(3‐4):249‐54. [DOI] [PubMed] [Google Scholar]
Perheentupa 2002 {published data only}
- Perheentupa A, Ruokonen A, Tuomivaara L, Ryynänen M, Martikainen H. Maternal serum (ß)‐HCG and (alpha)‐fetoprotein concentrations in singleton pregnancies following assisted reproduction. Human Reproduction 2002;17(3):794‐7. [DOI] [PubMed] [Google Scholar]
Perona 1998 {published data only}
- Perona M, Mancini G, Dall'Amico D, Guaraldo V, Carbonara A. Influence of smoking habits on Down's syndrome risk evaluation at mid‐trimester through biochemical screening. International Journal of Clinical & Laboratory Research 1998;28(3):179‐82. [DOI] [PubMed] [Google Scholar]
Persico 2008 {published data only}
- Persico N, Borenstein M, Molina F, Azumendi G, Nicolaides KH. Prenasal thickness in trisomy‐21 fetuses at 16‐24 weeks of gestation. Ultrasound in Obstetrics & Gynecology 2008;32(6):751‐4. [DOI] [PubMed] [Google Scholar]
Petervari 2000 {published data only}
- Petervari L, Varga A, Tanko A, Szabo L, Godo G. [Significance of nuchal edema in fetuses of pregnant women under 35 years of age]. [Hungarian]. Orvosi Hetilap 2000;141(8):399‐402. [PubMed] [Google Scholar]
Petrocik 1989 {published data only}
- Petrocik E, Wassman ER, Kelly JC. Prenatal screening for Down syndrome with maternal serum human chorionic gonadotropin levels.[see comment]. American Journal of Obstetrics and Gynecology 1989;161(5):1168‐73. [DOI] [PubMed] [Google Scholar]
Phillips 1992 {published data only}
- Phillips OP, Elias S, Shulman LP, Andersen RN, Morgan CD, Simpson JL. Maternal serum screening for fetal Down syndrome in women less than 35 years of age using alpha‐fetoprotein, hCG, and unconjugated estriol: a prospective 2‐year study. Obstetrics & Gynecology 1992;80(3):353‐8. [PubMed] [Google Scholar]
Phillips 1993 {published data only}
- Phillips OP, Shulman LP, Elias S, Simpson JL. Maternal serum screening for fetal Down syndrome using alpha‐ fetoprotein, human chorionic gonadotrophin, and unconjugated estriol in adolescents. Adolescent and Pediatric Gynecology 1993;6(2):91‐4. [Google Scholar]
Pihl 2008 {published data only}
- Pihl K, Larsen T, Jonsson L, Hougaard D, Krebs L, Norgaard‐Pedersen B, et al. [Quality control of prenatal screening]. [Danish]. Ugeskrift for Laeger 2008;170(35):2691‐5. [PubMed] [Google Scholar]
Pinette 2003 {published data only}
- Pinette MG, Egan JF, Wax JR, Blackstone J, Cartin A, Benn PA. Combined sonographic and biochemical markers for Down syndrome screening. Journal of Ultrasound in Medicine 2003;22(11):1185‐90. [DOI] [PubMed] [Google Scholar]
Platt 2004 {published data only}
- Platt LD, Greene N, Johnson A, Zachary J, Thom E, Krantz D, et al. Sequential pathways of testing after first‐trimester screening for trisomy 21. Obstetrics and Gynecology 2004;104(4):661‐6. [DOI] [PubMed] [Google Scholar]
Podobnik 1995 {published data only}
- Podobnik M, Singer Z, Podobnik Sarkanji S, Bulic M. First trimester diagnosis of cystic hygromata using transvaginal ultrasound and cytogenetic evaluation. Journal of Perinatal Medicine 1995;23(4):283‐91. [DOI] [PubMed] [Google Scholar]
Poon 2009 {published data only}
- Poon LC, Chelemen T, Minekawa R, Frisova V, Nicolaides KH. Maternal serum ADAM12 (A disintegrin and metalloprotease) in chromosomally abnormal pregnancy at 11‐13 weeks. American Journal of Obstetrics and Gynecology 2009;200(5):508‐6. [DOI] [PubMed] [Google Scholar]
Prefumo 2002 {published data only}
- Prefumo F, Thilaganathan B. Agreement between predicted risk and prevalence of Down syndrome in first trimester nuchal translucency screening. Prenatal Diagnosis 2002;22(10):917‐8. [DOI] [PubMed] [Google Scholar]
Prefumo 2004 {published data only}
- Prefumo F, Sairam S, Bhide A, Penna L, Hollis B, Thilaganathan B. Maternal ethnic origin and fetal nasal bones at 11‐14 weeks of gestation. BJOG: and international journal of obstetrics and gynaecology 2004;111(2):109‐12. [DOI] [PubMed] [Google Scholar]
Price 1998 {published data only}
- Price KM, Lith JM, Silman R, Mantingh A, Grudzinskas JG. First trimester maternal serum concentrations of fetal antigen 2 in normal pregnancies and those affected by trisomy 21. Human Reproduction 1998;13(6):1706‐8. [DOI] [PubMed] [Google Scholar]
Räty 2000 {published data only}
- Räty R, Virtanen A, Koskinen P, Laitinen P, Forsstrom J, Salonen R, et al. Maternal midtrimester serum AFP and free ß‐hCG levels in in vitro fertilization twin pregnancies. Prenatal Diagnosis 2000;20(3):221‐3. [PubMed] [Google Scholar]
Räty 2002 {published data only}
- Räty R, Virtanen A, Koskinen P, Anttila L, Forsström J, Laitinen P, et al. Serum free (ß)‐HCG and alpha‐fetoprotein levels in IVF, ICSI and frozen embryo transfer pregnancies in maternal mid‐trimester serum screening for Down's syndrome. Human Reproduction 2002;17(2):481‐4. [DOI] [PubMed] [Google Scholar]
Rembouskos 2004 {published data only}
- Rembouskos G, Cicero S, Longo D, Vandecruys H, Nicolaides KH. Assessment of the fetal nasal bone at 11‐14 weeks of gestation by three‐dimensional ultrasound. Ultrasound in Obstetrics & Gynecology 2004;23(3):232‐6. [DOI] [PubMed] [Google Scholar]
Ren 1992 {published data only}
- Ren S‐G, Braunstein GD. Human chorionic gonadotropin. Seminars in Reproductive Endocrinology 1992;10(2):95‐105. [Google Scholar]
Renier 1998 {published data only}
- Renier MA, Vereecken A, Herck E, Straetmans D, Ramaekers P, Buytaert P. Second trimester maternal dimeric inhibin‐A in the multiple‐marker screening test for Down's syndrome. Human Reproduction 1998;13(3):744‐8. [DOI] [PubMed] [Google Scholar]
Resta 1990 {published data only}
- Resta RG, Nyberg D. The role of ultrasound in screening for Down syndrome. Birth Defects: Original Article Series 1990;26(3):104. [PubMed] [Google Scholar]
Reynders 1997 {published data only}
- Reynders CS, Pauker SP, Benacerraf BR. First trimester isolated fetal nuchal lucency: significance and outcome. Journal of Ultrasound in Medicine 1997;16(2):101‐5. [DOI] [PubMed] [Google Scholar]
Reynolds 1989 {published data only}
- Reynolds TM, Penney MD. The mathematical basis of multivariate risk screening: with special reference to screening for Down's syndrome associated pregnancy. Annals of Clinical Biochemistry 1989;27(5):452‐8. [DOI] [PubMed] [Google Scholar]
Reynolds 1999 {published data only}
- Reynolds TM, Schaeffer HJ, Schlensker S. Estimation of Down's syndrome risks in the first trimester of pregnancy: Experience of testing with PAPP‐A, total hCG and free ß‐ hCG levels in maternal blood samples in a German population. Clinical Laboratory 1999;45(1‐2):49‐53. [Google Scholar]
Reynolds 2008 {published data only}
- Reynolds TM, Aldis J. Median parameters for Down's syndrome screening should be calculated using a moving time‐window method. Annals of Clinical Biochemistry 2008;45(Pt 6):567‐70. [DOI] [PubMed] [Google Scholar]
Ribbert 1996 {published data only}
- Ribbert LS, Kornman LH, Wolf BT, Simons AH, Jansen CA, Beekhuis JR, et al. Maternal serum screening for fetal Down syndrome in IVF pregnancies. Prenatal Diagnosis 1996;16(1):35‐8. [DOI] [PubMed] [Google Scholar]
Rice 2005 {published data only}
- Rice JD, McIntosh SF, Halstead AC. Second‐trimester maternal serum screening for Down syndrome in in vitro fertilization pregnancies. Prenatal Diagnosis 2005;25(3):234‐8. [DOI] [PubMed] [Google Scholar]
Rich 1991 {published data only}
- Rich N, Boots L, Davis R, Finley S. Efficiency of maternal serum hCG AFP and free estriol in the identification of trisomy 21 and other complications of pregnancy. Journal of the Alabama Academy of Science 1991;62(2‐3):135. [Google Scholar]
Roberts 1995 {published data only}
- Roberts LJ, Bewley S, Mackinson AM, Rodeck CH. First trimester fetal nuchal translucency: problems with screening the general population. 1. British Journal of Obstetrics & Gynaecology 1995;102(5):381‐5. [DOI] [PubMed] [Google Scholar]
Robertson 1991 {published data only}
- Robertson EF. Maternal serum screening for neural tube defects and Down's syndrome.[see comment]. Medical Journal of Australia 1991;155(2):67‐8. [PubMed] [Google Scholar]
Rode 2003 {published data only}
- Rode L, Wojdemann KR, Shalmi AC, Larsen SO, Sundberg K, Norgaard‐Pedersen B, et al. Combined first‐ and second‐trimester screening for Down syndrome: an evaluation of proMBP as a marker. Prenatal Diagnosis 2003;23(7):593‐8. [DOI] [PubMed] [Google Scholar]
Ronge 2006 {published data only}
- Ronge R. Combined first trimester screening for Down's syndrome is superior to quadruple test. Geburtshilfe und Frauenheilkunde 2006;66(4):332. [Google Scholar]
Rose 1995 {published data only}
- Rose NC, Mennuti MT. Multiple marker screening for women 35 and older. Contemporary OB/GYN 1995;40(9):55‐6. [Google Scholar]
Ross 1997 {published data only}
- Ross HL, Elias S. Maternal serum screening for fetal genetic disorders. Obstetrics & Gynecology Clinics of North America 1997;24(1):33‐47. [DOI] [PubMed] [Google Scholar]
Rotmensch 1996 {published data only}
- Rotmensch S, Liberati M, Kardana A, Copel JA, Ben‐Rafael Z, Cole LA. Nicked free ß‐subunit of human chorionic gonadotropin: A potential new marker for Down syndrome screening. American Journal of Obstetrics and Gynecology 1996;174(2):609‐11. [DOI] [PubMed] [Google Scholar]
Rotmensch 1999 {published data only}
- Rotmensch S, Celentano C, Shalev J, Vishne TH, Lipitz S, Ben‐Rafael Z, et al. Midtrimester maternal serum screening after multifetal pregnancy reduction in pregnancies conceived by in vitro fertilization. Journal of Assisted Reproduction and Genetics 1999;16(1):8‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
Rozenberg 2006 {published data only}
- Rozenberg P, Bussieres L, Chevret S, Bernard JP, Malagrida L, Cuckle H, et al. Screening for Down syndrome using first‐trimester combined screening followed by second‐trimester ultrasound examination in an unselected population. American Journal of Obstetrics and Gynecology 2006;195(5):1379‐87. [DOI] [PubMed] [Google Scholar]
Rudnicka 2002 {published data only}
- Rudnicka AR, Wald NJ, Huttly W, Hackshaw AK. Influence of maternal smoking on the birth prevalence of Down syndrome and on second trimester screening performance. Prenatal Diagnosis 2002;22(10):893‐7. [DOI] [PubMed] [Google Scholar]
Ryall 1992 {published data only}
- Ryall RG, Staples AJ, Robertson EF, Pollard AC. Improved performance in a prenatal screening programme for Down's syndrome incorporating serum‐free hCG subunit analyses. Prenatal Diagnosis 1992;12(4):251‐61. [DOI] [PubMed] [Google Scholar]
Ryall 2001 {published data only}
- Ryall RG, Callen D, Cocciolone R, Duvnjak A, Esca R, Frantzis N, et al. Karyotypes found in the population declared at increased risk of Down syndrome following maternal serum screening. Prenatal Diagnosis 2001;21(7):553‐7. [DOI] [PubMed] [Google Scholar]
Sabriá 2002 {published data only}
- Sabriá J, Cabrero D, Bach C. Aneuploidy screening: Ultrasound versus biochemistry. Ultrasound Review of Obstetrics and Gynecology 2002;2(4):221‐8. [Google Scholar]
Sacchini 2003 {published data only}
- Sacchini C, El‐Sheikhah A, Cicero S, Rembouskos G, Nicolaides KH. Ear length in trisomy 21 fetuses at 11‐14 weeks of gestation. Ultrasound in Obstetrics & Gynecology 2003;22(5):460‐3. [DOI] [PubMed] [Google Scholar]
Sahota 2009 {published data only}
- Sahota DS, Leung TY, Chan LW, Law LW, Fung TY, Chan OK, et al. First‐trimester fetal nasal bone length in an ethnic Chinese population. Ultrasound in Obstetrics & Gynecology 2009;34(1):33‐7. [DOI] [PubMed] [Google Scholar]
Sahota 2010 {published data only}
- Sahota DS, Leung TY, Chen M, Chan LW, Fung TY, Lau TK. Comparison of likelihood ratios of first‐trimester nuchal translucency measurements: multiples of median, delta or mixture. Ultrasound in Obstetrics & Gynecology 2010;36(1):15‐9. [DOI] [PubMed] [Google Scholar]
Salazar 2007 {published data only}
- Salazar Lopez R, Ibarra Gallardo AL, Iduma Melendrez M, Leyva Bojorquez R. [Specificity of biochemical markers of pregnancy second trimester]. [Spanish]. Ginecologia y Obstetricia de Mexico 2007;75(10):608‐14. [PubMed] [Google Scholar]
Salazar 2008 {published data only}
- Salazar Lopez R, Ibarra Gallardo AL, Iduma Melendrez M, Leyva R. [Evaluation of plasmatic A protein as only marker during first trimester of pregnancy]. [Spanish]. Ginecologia y Obstetricia de Mexico 2008;76(10):576‐81. [PubMed] [Google Scholar]
Saller 1997 {published data only}
- Saller DN Jr, Canick JA, Kellner LH, Rose NC, Garza J, French CA, et al. Maternal serum analyte levels in pregnancies with fetal Down syndrome resulting from translocations. American Journal of Obstetrics and Gynecology 1997;177(4):879‐81. [DOI] [PubMed] [Google Scholar]
Salomon 2001 {published data only}
- Salomon LJ, Bernard JP, Taupin P, Benard C, Ville Y. Relationship between nuchal translucency at 11‐14 weeks and nuchal fold at 20‐24 weeks of gestation. Ultrasound in Obstetrics & Gynecology 2001;18(6):636‐7. [DOI] [PubMed] [Google Scholar]
Salonen 1997 {published data only}
- Salonen R, Turpeinen U, Kurki L, Lappalainen M, Ammala P, Hiilesmaa V, et al. Maternal serum screening for Down's syndrome on population basis. Acta Obstetricia et Gynecologica Scandinavica 1997;76(9):817‐21. [DOI] [PubMed] [Google Scholar]
Saltvedt 2005 {published data only}
- Saltvedt S, Almstrom H, Kublickas M, Valentin L, Bottinga R, Bui TH, et al. Screening for Down syndrome based on maternal age or fetal nuchal translucency: a randomized controlled trial in 39,572 pregnancies. Ultrasound in Obstetrics & Gynecology 2005;25(6):537‐45. [DOI] [PubMed] [Google Scholar]
Saridogan 1996 {published data only}
- Saridogan E, Djahanbakhch O, Naftalin AA. Screening for Down's syndrome: experience in an inner city health district. British Journal of Obstetrics & Gynaecology 1996;103(12):1205‐11. [DOI] [PubMed] [Google Scholar]
Savoldelli 1993 {published data only}
- Savoldelli G, Binkert F, Achermann J, Schmid W. Ultrasound screening for chromosomal anomalies in the first trimester of pregnancy. Prenatal Diagnosis 1993;13(6):513‐8. [DOI] [PubMed] [Google Scholar]
Schielen 2009 {published data only}
- Schielen PC, Wildschut HI, Loeber JG. Down syndrome screening: determining the cutoff level of risk for invasive testing. Prenatal Diagnosis 2009;29(2):190‐2. [DOI] [PubMed] [Google Scholar]
Schiott 2006 {published data only}
- Schiott KM, Christiansen M, Petersen OB, Sorensen TL, Uldbjerg N. The "Consecutive Combined Test"‐‐using double test from week 8 + 0 and nuchal translucency scan, for first trimester screening for Down syndrome. Prenatal Diagnosis 2006;26(12):1105‐9. [DOI] [PubMed] [Google Scholar]
Schmidt 2007a {published data only}
- Schmidt P, Rom J, Maul H, Vaske B, Hillemanns P, Scharf A. Advanced first trimester screening (AFS): an improved test strategy for the individual risk assessment of fetal aneuploidies and malformations. Archives of Gynecology & Obstetrics 2007;276(2):159‐66. [DOI] [PubMed] [Google Scholar]
Schmidt 2007b {published data only}
- Schmidt P, Staboulidou I, Soergel P, Wustemann M, Hillemanns P, Scharf A. Comparison of Nicolaides' risk evaluation for Down's syndrome with a novel software: an analysis of 1,463 cases. Archives of Gynecology & Obstetrics 2007;275(6):469‐74. [DOI] [PubMed] [Google Scholar]
Schmidt 2007c {published data only}
- Schmidt P, Pruggmayer M, Steinborn A, Schippert C, Staboulidou I, Hillemanns P, et al. Are nuchal translucency, pregnancy associated plasma protein‐A or free‐beta‐human chorionic gonadotropin depending on maternal age? A multicenter study of 8,116 pregnancies. Archives of Gynecology & Obstetrics 2007;276(3):259‐62. [DOI] [PubMed] [Google Scholar]
Schmidt 2008a {published data only}
- Schmidt P, Hormansdorfer C, Pruggmayer M, Schutte C, Neumann A, Gerritzen A, et al. Improved prenatal aneuploidy screening using the novel advanced first‐trimester screening algorithm: a multicenter study of 10,017 pregnancies. Journal of Clinical Ultrasound 2008;36(7):397‐402. [DOI] [PubMed] [Google Scholar]
Schmidt 2008b {published data only}
- Schmidt P, Staboulidou I, Elsasser M, Vaske B, Hillemanns P, Scharf A. How imprecise may the measurement of fetal nuchal translucency be without worsening first‐trimester screening?. Fetal Diagnosis and Therapy 2008;24(3):291‐5. [DOI] [PubMed] [Google Scholar]
Schmidt 2008c {published data only}
- Schmidt P, Hormansdorfer C, Oehler K, Hartel H, Hillemanns P, Scharf A. [Three‐dimensional scatter plot analysis to estimate the risk of foetal aneuloidy]. [German]. Zeitschrift fur Geburtshilfe und Neonatologie 2008;212(4):127‐35. [DOI] [PubMed] [Google Scholar]
Schmidt 2010 {published data only}
- Schmidt P, Hormansdorfer C, Golatta M, Scharf A. Analysis of the distribution shift of detected aneuploidies by age independent first trimester screening. Archives of Gynecology & Obstetrics 2010;281(3):393‐9. [DOI] [PubMed] [Google Scholar]
Schuchter 1998 {published data only}
- Schuchter K, Wald N, Hackshaw AK, Hafner E, Liebhart E. The distribution of nuchal translucency at 10‐13 weeks of pregnancy. Prenatal Diagnosis 1998;18(3):281‐6. [PubMed] [Google Scholar]
Scott 1995 {published data only}
- Scott F, Boogert A, Smart S, Anderson J. Maternal serum screening and routine 18‐week ultrasound in the detection of all chromosomal abnormalities. Australian & New Zealand Journal of Obstetrics & Gynaecology 1995;35(2):165‐8. [DOI] [PubMed] [Google Scholar]
Seeds 1990 {published data only}
- Seeds JW, Watson WJ. Ultrasound and maternal serum alpha‐fetoprotein screening: A complementary relationship. Ultrasound Quarterly 1990;8(2):145‐66. [Google Scholar]
Seki 1995 {published data only}
- Seki K, Mitsui C, Nagata I. Measurement of urinary free ß‐human chorionic gonadotropin by immunoradiometric assay. Gynecologic and Obstetric Investigation 1995;40(3):162‐7. [DOI] [PubMed] [Google Scholar]
Shenhav 2003 {published data only}
- Shenhav S, Gemer O, Sherman DJ, Peled R, Segal S. Midtrimester triple‐test levels in women with chronic hypertension and altered renal function. Prenatal Diagnosis 2003;23(2):166‐7. [DOI] [PubMed] [Google Scholar]
Shintaku 1989 {published data only}
- Shintaku Y, Takabayashi T, Sasaki H, Ozawa N, Shinkawa O, Hamazaki Y, et al. [Screening for chromosomal anomalies with maternal serum alpha‐fetoprotein]. [Japanese]. Nippon Sanka Fujinka Gakkai Zasshi ‐ Acta Obstetrica et Gynaecologica Japonica 1989;41(2):185‐90. [PubMed] [Google Scholar]
Shulman 2003 {published data only}
- Shulman A, Maymon R. Mid‐gestation Down syndrome screening test and pregnancy outcome among unstimulated assisted‐conception pregnancies. Prenatal Diagnosis 2003;23(8):625‐8. [DOI] [PubMed] [Google Scholar]
Sieroszewski 2008 {published data only}
- Sieroszewski P, Perenc M, Budecka EB, Sobala W, Deutinger J. Sonographical integrated test for detection of chromosomal aberrations. Ultraschall in der Medizin 2008;29(2):190‐6. [DOI] [PubMed] [Google Scholar]
Simon‐Bouy 1999 {published data only}
- Simon‐Bouy B. [Markers for trisomy 21][French]. Fertilite Contraception Sexualite 1999;27(9):289‐91. [PubMed] [Google Scholar]
Simpson 1986 {published data only}
- Simpson JL, Baum LD, Marder R, Elias S, Ober C, Martin AO. Maternal serum alpha‐fetoprotein screening: low and high values for detection of genetic abnormalities. American Journal of Obstetrics and Gynecology 1986;155(3):593‐7. [DOI] [PubMed] [Google Scholar]
Smith 1990 {published data only}
- Smith C, Grube GL, Wilson S. Maternal serum alpha‐fetoprotein screening and the role of ultrasound. Journal of Diagnostic Medical Sonography 1990;6(6):312‐6. [Google Scholar]
Smith 1996 {published data only}
- Smith ER, Petersen J, Okorodudu AO, Bissell MG. Does the addition of unconjugated estriol in maternal serum screening improve the detection of trisomy 21? A meta‐analysis. Clinical Laboratory Management Review 1996;10(2):176‐81. [PubMed] [Google Scholar]
Smith 1999 {published data only}
- Smith NC, Hau C. A six year study of the antenatal detection of fetal abnormality in six Scottish health boards. British Journal of Obstetrics & Gynaecology 1999;106(3):206‐12. [DOI] [PubMed] [Google Scholar]
Smith‐Bindman 2001 {published data only}
- Smith‐Bindman R, Hosmer W, Feldstein VA, Deeks JJ, Goldberg JD. Second‐trimester ultrasound to detect fetuses with Down syndrome: a meta‐analysis.[see comment]. JAMA 2001;285(8):1044‐55. [DOI] [PubMed] [Google Scholar]
Smith‐Bindman 2003 {published data only}
- Smith‐Bindman R, Chu P, Bacchetti P, Waters JJ, Mutton D, Alberman E. Prenatal screening for Down syndrome in England and Wales and population‐based birth outcomes. American Journal of Obstetrics and Gynecology 2003;187(4):980‐5. [DOI] [PubMed] [Google Scholar]
Snijders 1995 {published data only}
- Snijders RJM, Sebire NJ, Nicolaides KH. Maternal age and gestational age‐specific risk for chromosomal defects. Fetal Diagnosis and Therapy 1995;10(6):356‐67. [DOI] [PubMed] [Google Scholar]
Snijders 1999 {published data only}
- Snijders RJM, Sundberg K, Holzgreve W, Henry G, Nicolaides KH. Maternal age‐ and gestation‐specific risk for trisomy 21. Ultrasound in Obstetrics and Gynecology 1999;13(3):167‐70. [DOI] [PubMed] [Google Scholar]
Soergel 2006 {published data only}
- Soergel P, Pruggmayer M, Schwerdtfeger R, Muhlhaus K, Scharf A. Screening for trisomy 21 with maternal age, fetal nuchal translucency and maternal serum biochemistry at 11‐14 weeks: a regional experience from Germany. Fetal Diagnosis and Therapy 2006;21(3):264‐8. [DOI] [PubMed] [Google Scholar]
Sokol 1998 {published data only}
- Sokol AI, Kramer RL, Yaron Y, O'Brien JE, Muller F, Johnson MP, et al. Age‐specific variation in aneuploidy incidence among biochemical screening programs. American Journal of Obstetrics and Gynecology 1998;179(4):971‐3. [DOI] [PubMed] [Google Scholar]
Sonek 2003 {published data only}
- Sonek JD. Nasal bone evaluation with ultrasonography: A marker for fetal aneuploidy. Ultrasound in Obstetrics and Gynecology 2003;22(1):11‐5. [DOI] [PubMed] [Google Scholar]
Sonek 2007 {published data only}
- Sonek J, Borenstein M, Downing C, McKenna D, Neiger R, Croom C, et al. Frontomaxillary facial angles in screening for trisomy 21 at 14‐23 weeks' gestation. American Journal of Obstetrics and Gynecology 2007;197(2):160‐5. [DOI] [PubMed] [Google Scholar]
Sood 2010 {published data only}
- Sood M, Rochelson B, Krantz D, Ravens R, Tam Tam H, Vohra N, et al. Are second‐trimester minor sonographic markers for Down syndrome useful in patients who have undergone first‐trimester combined screening?. American Journal of Obstetrics and Gynecology 2010;203(4):408‐4. [DOI] [PubMed] [Google Scholar]
Sooklim 2010 {published data only}
- Sooklim R, Manotaya S. Fetal facial sonographic markers for second trimester Down syndrome screening in a Thai population. International Journal of Gynaecology & Obstetrics 2010;111(2):144‐7. [DOI] [PubMed] [Google Scholar]
Spencer 1985 {published data only}
- Spencer K, Carpenter P. Screening for Down's syndrome using serum alpha fetoprotein: a retrospective study indicating caution. British Medical Journal Clinical Research Education 1985;290(6486):1940‐3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spencer 1991a {published data only}
- Spencer K. Evaluation of an assay of the free ß‐subunit of choriogonadotropin and its potential value in screening for Down's syndrome. Clinical Chemistry 1991;37(6):809‐14. [PubMed] [Google Scholar]
Spencer 1991b {published data only}
- Spencer K. Maternal serum CA125 is not a second trimester marker for Down's syndrome. Annals of Clinical Biochemistry 1991;28(3):299‐300. [DOI] [PubMed] [Google Scholar]
Spencer 1992 {published data only}
- Spencer K, Coombes EJ, Mallard AS, Ward AM. Free ß human choriogonadotropin in Down's syndrome screening: a multicentre study of its role compared with other biochemical markers.[see comment]. Annals of Clinical Biochemistry 1992;29(5):506‐18. [DOI] [PubMed] [Google Scholar]
Spencer 1993a {published data only}
- Spencer K, Carpenter P. Prospective study of prenatal screening for Down's syndrome with free ß human chorionic gonadotrophin.[see comment]. BMJ 1993;307(6907):764‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spencer 1993b {published data only}
- Spencer K, Macri JN, Carpenter P, Anderson R, Krantz DA. Stability of intact chorionic gonadotropin (hCG) in serum, liquid whole blood, and dried whole‐blood filter‐paper spots: impact on screening for Down syndrome by measurement of free ß‐hCG subunit. Clinical Chemistry 1993;39(6):1064‐8. [PubMed] [Google Scholar]
Spencer 1993c {published data only}
- Spencer K, Wood PJ, Anthony FW. Elevated levels of maternal serum Inhibin Aimmunoreactivity in second trimester pregnancies affected by Down's syndrome. Annals of Clinical Biochemistry 1993;30(2):219‐20. [DOI] [PubMed] [Google Scholar]
Spencer 1993d {published data only}
- Spencer K, Macri JN, Anderson RW, Aitken DA, Berry E, Crossley JA, et al. Dual analyte immunoassay in neural tube defect and Down's syndrome screening: results of a multicentre clinical trial. Annals of Clinical Biochemistry 1993;30(4):394‐401. [DOI] [PubMed] [Google Scholar]
Spencer 1993e {published data only}
- Spencer K. Free alpha‐subunit of human chorionic gonadotropin in Down syndrome. American Journal of Obstetrics and Gynecology 1993;168(1):132‐5. [DOI] [PubMed] [Google Scholar]
Spencer 1995 {published data only}
- Spencer K. The influence of gravidity on Down's syndrome screening with free ß hCG. Prenatal Diagnosis 1995;15(1):87‐9. [DOI] [PubMed] [Google Scholar]
Spencer 1996 {published data only}
- Spencer K, Wallace EM, Ritoe S. Second‐trimester dimeric inhibin‐A in Down's syndrome screening. Prenatal Diagnosis 1996;16(12):1101‐10. [DOI] [PubMed] [Google Scholar]
Spencer 1997 {published data only}
- Spencer K, Noble P, Snijders RJ, Nicolaides KH. First‐trimester urine free ß hCG, ß core, and total oestriol in pregnancies affected by Down's syndrome: implications for first‐trimester screening with nuchal translucency and serum free ß hCG. Prenatal Diagnosis 1997;17(6):525‐38. [DOI] [PubMed] [Google Scholar]
Spencer 1998a {published data only}
- Spencer K. The influence of smoking on maternal serum AFP and free ß hCG levels and the impact on screening for Down syndrome. Prenatal Diagnosis 1998;18(3):225‐34. [DOI] [PubMed] [Google Scholar]
Spencer 1998b {published data only}
- Spencer K, Carpenter P. Is prostate‐specific antigen a marker for pregnancies affected by Down syndrome?. Clinical Chemistry 1998;44(11):2362‐5. [PubMed] [Google Scholar]
Spencer 1999a {published data only}
- Spencer K. Second trimester prenatal screening for Down's syndrome using alpha‐fetoprotein and free ß hCG: a seven year review. British Journal of Obstetrics & Gynaecology 1999;106(12):1287‐93. [DOI] [PubMed] [Google Scholar]
Spencer 1999b {published data only}
- Spencer K. Accuracy of Down's syndrome risks produced in a prenatal screening program. Annals of Clinical Biochemistry 1999;36(1):101‐3. [DOI] [PubMed] [Google Scholar]
Spencer 2000a {published data only}
- Spencer K, Berry E, Crossley JA, Aitken DA, Nicolaides KH. Is maternal serum total hCG a marker of trisomy 21 in the first trimester of pregnancy?. Prenatal Diagnosis 2000;20(4):311‐7. [DOI] [PubMed] [Google Scholar]
Spencer 2000b {published data only}
- Spencer K. Screening for trisomy 21 in twin pregnancies in the first trimester using free ß‐hCG and PAPP‐A, combined with fetal nuchal translucency thickness. Prenatal Diagnosis 2000;20(2):91‐5. [DOI] [PubMed] [Google Scholar]
Spencer 2000c {published data only}
- Spencer K. The influence of smoking on maternal serum PAPP‐A and free ß hCG levels in the first trimester of pregnancy. Prenatal Diagnosis 1999;19(11):1065‐6. [DOI] [PubMed] [Google Scholar]
Spencer 2000d {published data only}
- Spencer K, Ong CY, Liao AW, Nicolaides KH. The influence of parity and gravidity on first trimester markers of chromosomal abnormality. Prenatal Diagnosis 2000;20(10):792‐4. [PubMed] [Google Scholar]
Spencer 2000e {published data only}
- Spencer K. The influence of fetal sex in screening for Down syndrome in the second trimester using AFP and free ß‐hCG. Prenatal Diagnosis 2000;20(8):648‐51. [DOI] [PubMed] [Google Scholar]
Spencer 2000f {published data only}
- Spencer K, Ong CY, Liao AW, Nicolaides KH. The influence of ethnic origin on first trimester biochemical markers of chromosomal abnormalities. Prenatal Diagnosis 2000;20(6):491‐4. [PubMed] [Google Scholar]
Spencer 2000g {published data only}
- Spencer K, Tul N, Nicolaides KH. Maternal serum free ß‐hCG and PAPP‐A in fetal sex chromosome defects in the first trimester. Prenatal Diagnosis 2000;20(5):390‐4. [DOI] [PubMed] [Google Scholar]
Spencer 2000h {published data only}
- Spencer K. Second‐trimester prenatal screening for Down syndrome and the relationship of maternal serum biochemical markers to pregnancy complications with adverse outcome. Prenatal Diagnosis 2000;20(8):652‐6. [PubMed] [Google Scholar]
Spencer 2000i {published data only}
- Spencer K, Ong CY, Liao AW, Papademetriou D, Nicolaides KH. The influence of fetal sex in screening for trisomy 21 by fetal nuchal translucency, maternal serum free ß‐hCG and PAPP‐A at 10‐14 weeks of gestation. Prenatal Diagnosis 2000;20(8):673‐5. [PubMed] [Google Scholar]
Spencer 2001a {published data only}
- Spencer K. Age related detection and false positive rates when screening for Down's syndrome in the first trimester using fetal nuchal translucency and maternal serum free ßhCG and PAPP‐A. BJOG 2001;108(10):1043‐6. [DOI] [PubMed] [Google Scholar]
Spencer 2001b {published data only}
- Spencer K, Liao AW, Ong CY, Geerts L, Nicolaides KH. First trimester maternal serum placenta growth factor (PIGF)concentrations in pregnancies with fetal trisomy 21 or trisomy 18. Prenatal Diagnosis 2001;21(9):718‐22. [DOI] [PubMed] [Google Scholar]
Spencer 2001c {published data only}
- Spencer K, Liao AW, Ong CY, Geerts L, Nicolaides KH. Maternal serum levels of dimeric Inhibin A in pregnancies affected by trisomy 21 in the first trimester. Prenatal Diagnosis 2001;21(6):441‐4. [DOI] [PubMed] [Google Scholar]
Spencer 2001d {published data only}
- Spencer K, Liao AW, Skentou H, Ong CY, Nicolaides KH. Maternal serum levels of total activin‐A in first‐trimester trisomy 21 pregnancies. Prenatal Diagnosis 2001;21(4):270‐3. [DOI] [PubMed] [Google Scholar]
Spencer 2001e {published data only}
- Spencer K. Screening for trisomy 21 in twin pregnancies in the first trimester: does chorionicity impact on maternal serum free ß‐hCG or PAPP‐A levels?. Prenatal Diagnosis 2001;21(9):715‐7. [DOI] [PubMed] [Google Scholar]
Spencer 2002a {published data only}
- Spencer K, Nicolaides KH. A first trimester trisomy 13/trisomy 18 risk algorithm combining fetal nuchal translucency thickness, maternal serum free ß‐hCG and PAPP‐A. Prenatal Diagnosis 2002;22(10):877‐9. [DOI] [PubMed] [Google Scholar]
Spencer 2002b {published data only}
- Spencer K. Accuracy of Down syndrome risks produced in a first‐trimester screening programme incorporating fetal nuchal translucency thickness and maternal serum biochemistry. Prenatal Diagnosis 2002;22(3):244‐6. [DOI] [PubMed] [Google Scholar]
Spencer 2002c {published data only}
- Spencer K, Cuckle HS. Screening for chromosomal anomalies in the first trimester: does repeat maternal serum screening improve detection rates?. Prenatal Diagnosis 2002;22(10):903‐6. [DOI] [PubMed] [Google Scholar]
Spencer 2002d {published data only}
- Spencer K, Crossley JA, Aitken DA, Nix AB, Dunstan FD, Williams K. Temporal changes in maternal serum biochemical markers of trisomy 21 across the first and second trimester of pregnancy. Annals of Clinical Biochemistry 2002;39(6):567‐76. [DOI] [PubMed] [Google Scholar]
Spencer 2003a {published data only}
- Spencer K, Crossley JA, Aitken DA, Nix AB, Dunstan FD, Williams K. The effect of temporal variation in biochemical markers of trisomy 21 across the first and second trimesters of pregnancy on the estimation of individual patient‐specific risks and detection rates for Down's syndrome. Annals of Clinical Biochemistry 2003;40(3):219‐31. [DOI] [PubMed] [Google Scholar]
Spencer 2003b {published data only}
- Spencer K. The influence of different sample collection types on the levels of markers used for Down's syndrome screening as measured by the Kryptor Immunosassay system. Annals of Clinical Biochemistry 2003;40(2):166‐8. [DOI] [PubMed] [Google Scholar]
Spencer 2003c {published data only}
- Spencer K, Bindra R, Nicolaides KH. Maternal weight correction of maternal serum PAPP‐A and free ß‐hCG MoM when screening for trisomy 21 in the first trimester of pregnancy. Prenatal Diagnosis 2003;23(10):851‐5. [DOI] [PubMed] [Google Scholar]
Spencer 2003d {published data only}
- Spencer K, Nicolaides KH. Screening for trisomy 21 in twins using first trimester ultrasound and maternal serum biochemistry in a one‐stop clinic: a review of three years experience. BJOG: an international journal of obstetrics and gynaecology 2003;110(3):279‐80. [PubMed] [Google Scholar]
Spencer 2004 {published data only}
- Spencer K, Bindra R, Cacho AM, Nicolaides KH. The impact of correcting for smoking status when screening for chromosomal anomalies using maternal serum biochemistry and fetal nuchal translucency thickness in the first trimester of pregnancy. Prenatal Diagnosis 2004;24(3):169‐73. [DOI] [PubMed] [Google Scholar]
Spencer 2005a {published data only}
- Spencer K, Cicero S, Atzei A, Otigbah C, Nicolaides KH. The influence of maternal insulin‐dependent diabetes on fetal nuchal translucency thickness and first‐trimester maternal serum biochemical markers of aneuploidy. Prenatal Diagnosis 2005;25(10):927‐9. [DOI] [PubMed] [Google Scholar]
Spencer 2005b {published data only}
- Spencer K, Heath V, El‐Sheikhah A, Ong CY, Nicolaides KH. Ethnicity and the need for correction of biochemical and ultrasound markers of chromosomal anomalies in the first trimester: a study of Oriental, Asian and Afro‐Caribbean populations. Prenatal Diagnosis 2005;25(5):365‐9. [DOI] [PubMed] [Google Scholar]
Spencer 2005c {published data only}
- Spencer K. First trimester maternal serum screening for Down's syndrome: an evaluation of the DPC Immulite 2000 free ß‐hCG and pregnancy‐associated plasma protein‐A assays.[see comment]. Annals of Clinical Biochemistry 2005;42(1):30‐40. [DOI] [PubMed] [Google Scholar]
Spencer 2008b {published data only}
- Spencer K, Cowans NJ, Uldbjerg N, Vereecken A, Torring N. First trimester intact hCG as an early marker of trisomy 21: a promise unrecognised?. Prenatal Diagnosis 2008;28(12):1156‐9. [DOI] [PubMed] [Google Scholar]
Spong 1999 {published data only}
- Spong CY, Ghidini A, Stanley‐Christian H, Meck JM, Seydel FD, Pezzullo JC. Risk of abnormal triple screen for Down syndrome is significantly higher in women with female fetuses. Prenatal Diagnosis 1999;19(4):337‐9. [DOI] [PubMed] [Google Scholar]
Staboulidou 2009 {published data only}
- Staboulidou I, Galindo A, Maiz N, Karagiannis G, Nicolaides KH. First‐trimester uterine artery Doppler and serum pregnancy‐associated plasma protein‐a in preeclampsia and chromosomal defects. Fetal Diagnosis and Therapy 2009;25(3):336‐9. [DOI] [PubMed] [Google Scholar]
Stevens 1998 {published data only}
- Stevens SL. The use of nuchal lucency as a screening tool in first trimester sonography. Journal of Diagnostic Medical Sonography 1998;14(6):251‐4. [Google Scholar]
Stoll 1992 {published data only}
- Stoll C. A new approach of prenatal prevention of constiutional disabilities ‐ the study of markers of maternal serum. Journal de Medecine de Strasbourg 1992;23(1):25‐7. [Google Scholar]
Stressig 2011 {published data only}
- Stressig R, Kozlowski P, Froehlich S, Siegmann HJ, Hammer R, Blumenstock G, et al. Assessment of the ductus venosus, tricuspid blood flow and the nasal bone in second‐trimester screening for trisomy 21. Ultrasound in Obstetrics & Gynecology 2011;37(4):444‐9. [DOI] [PubMed] [Google Scholar]
Su 2002 {published data only}
- Su YN, Hsu JJ, Lee CN, Cheng WF, Kung CC, Hsieh FJ. Raised maternal serum placenta growth factor concentration during the second trimester is associated with Down syndrome. Prenatal Diagnosis 2002;22(1):8‐12. [DOI] [PubMed] [Google Scholar]
Suchet 1995 {published data only}
- Suchet IB. Ultrasonography of the fetal neck in the first and second trimesters. Part 2. Anomalies of the posterior nuchal region. Canadian Association of Radiologists Journal 1995;46(5):344‐52. [PubMed] [Google Scholar]
Suchy 1990 {published data only}
- Suchy SF, Yeager MT. Down syndrome screening in women under 35 with maternal serum hCG. Obstetrics & Gynecology 1990;76(1):20‐4. [PubMed] [Google Scholar]
Summers 2003a {published data only}
- Summers AM, Farrell SA, Huang T, Meier C, Wyatt PR. Maternal serum screening in Ontario using the triple marker test. Journal of Medical Screening 2003;10(3):107‐11. [DOI] [PubMed] [Google Scholar]
Summers 2003b {published data only}
- Summers AM, Huang T, Meier C, Wyatt PR. The implications of a false positive second‐trimester serum screen for Down syndrome. Obstetrics & Gynecology 2003;101(6):1301‐6. [DOI] [PubMed] [Google Scholar]
Suntharasaj 2005 {published data only}
- Suntharasaj T, Ratanasiri T, Chanprapaph P, Kengpol C, Kor‐anantakul O, Leetanaporn R, et al. Variability of nuchal translucency measurement: a multicenter study in Thailand. Gynecologic & Obstetric Investigation 2005;60(4):201‐5. [DOI] [PubMed] [Google Scholar]
Susman 2010 {published data only}
- Susman MR, Amor DJ, Muggli E, Jaques AM, Halliday J. Using population‐based data to predict the impact of introducing noninvasive prenatal diagnosis for Down syndrome. Genetics in Medicine 2010;12(5):298‐303. [DOI] [PubMed] [Google Scholar]
Sutton 2004 {published data only}
- Sutton JM, Cole LA. Sialic acid‐deficient invasive trophoblast antigen (sd‐ITA): a new urinary variant for gestational Down syndrome screening. Prenatal Diagnosis 2004;24(3):194‐7. [DOI] [PubMed] [Google Scholar]
Suzuki 1998 {published data only}
- Suzuki Y, Takada J, Iwaki T, Isaka K, Takayama M. Screening for trisomy 21 in the first trimester by measurement of serum PAPP‐A and free ß‐hCG. Acta Obstetrica et Gynaecologica Japonica 1998;50(1):37‐40. [Google Scholar]
Tabor 1987 {published data only}
- Tabor A, Larsen SO, Nielsen J, Nielsen J, Philip J, Pilgaard B, et al. Screening for Down's syndrome using an iso‐risk curve based on maternal age and serum alpha‐fetoprotein level. British Journal of Obstetrics & Gynaecology 1987;94(7):636‐42. [DOI] [PubMed] [Google Scholar]
Tanski 1999 {published data only}
- Tanski S, Rosengren SS, Benn PA. Predictive value of the triple screening test for the phenotype of Down syndrome. American Journal of Medical Genetics 1999;85(2):123‐6. [DOI] [PubMed] [Google Scholar]
Thilaganathan 1998 {published data only}
- Thilaganathan B, Khare M, Williams B, Wathen NC. Influence of ethnic origin on nuchal translucency screening for Down's syndrome. Ultrasound in Obstetrics & Gynecology 1998;12(2):112‐4. [DOI] [PubMed] [Google Scholar]
Thilaganathan 1999 {published data only}
- Thilaganathan B. First‐trimester nuchal translucency and maternal serum biochemical screening for Down's syndrome: A happy union?. Ultrasound in Obstetrics and Gynecology 1999;13(4):229‐30. [DOI] [PubMed] [Google Scholar]
Tislaric 2002 {published data only}
- Tislaric D, Brajenovic‐Milic B, Ristic S, Latin V, Zuvic‐Butorac M, Bacic J, et al. The influence of smoking and parity on serum markers for Down's syndrome screening. Fetal Diagnosis and Therapy 2002;17(1):17‐21. [DOI] [PubMed] [Google Scholar]
Torok 1997 {published data only}
- Torok O, Veress L, Szabo M, Zsupan I, Buczko Z, Bolodar A, et al. [Biochemical and ultrasonic screening of chromosomal aneuploidies in the second trimester of pregnancy]. [Hungarian]. Orvosi Hetilap 1997;138(3):123‐7. [PubMed] [Google Scholar]
Torring 2009 {published data only}
- Torring N. Performance of first‐trimester screening between gestational weeks 7 and 13. Clinical Chemistry 2009;55(8):1564‐7. [DOI] [PubMed] [Google Scholar]
Trninic‐Pjevic 2007 {published data only}
- Trninic‐Pjevic A, Novakov‐Mikic A. [First trimester ultrasound screening of chromosomal abnormalities]. [Serbian]. Srpski Arhiv Za Celokupno Lekarstvo 2007;135(3‐4):153‐6. [DOI] [PubMed] [Google Scholar]
Tsai 2001 {published data only}
- Tsai MS, Huang YY, Hwa KY, Cheng CC, Lee FK. Combined measurement of fetal nuchal translucency, maternal serum free ß‐hCG, and pregnancy‐associated plasma protein A for first‐trimester Down's syndrome screening. Journal of the Formosan Medical Association 2001;100(5):319‐25. [PubMed] [Google Scholar]
Valerio 1996 {published data only}
- Valerio D, Aiello R, Altieri V, Fagnoni P. Maternal serum screening of fetal chromosomal abnormalities by AFP, UE3, hCG and free‐ß hCG. Prospective and retrospective results. Minerva Ginecologica 1996;48(5):169‐73. [PubMed] [Google Scholar]
Van Blerk 1992 {published data only}
- Blerk M, Smitz J, Catte L, Kumps C, Elst J, Steirteghem AC. Second‐trimester cancer antigen 125 and Down's syndrome.[see comment]. Prenatal Diagnosis 1992;12(12):1062‐6. [DOI] [PubMed] [Google Scholar]
Van Dyke 2007 {published data only}
- Dyke DL, Ebrahim SA, Al Saadi AA, Powell SA, Zenger‐Hain JL, Micale MA, et al. The impact of maternal serum screening programs for Down syndrome in southeast Michigan, 1988‐2003. Prenatal Diagnosis 2007;27(6):583‐4. [DOI] [PubMed] [Google Scholar]
Van Heesch, 2006 {published data only}
- Heesch PN, Schielen PC, Wildhagen MF, Hollander K, Steegers EA, Wildschut HI. Combined first trimester screening for trisomy 21: Lack of agreement between risk calculation methods. Journal of Perinatal Medicine 2006;34(2):162‐5. [DOI] [PubMed] [Google Scholar]
Van Lith 1991 {published data only}
- Lith JM, Mantingh A, Beekhuis JR, Bruijn HW, Breed AS. First trimester CA 125 and Down's syndrome.[see comment]. British Journal of Obstetrics & Gynaecology 1991;98(5):493‐4. [DOI] [PubMed] [Google Scholar]
Van Lith 1993 {published data only}
- Lith JM, Mantingh A, Bruijn HW. Maternal serum CA 125 levles in pregnancies with chromosomally‐normal and ‐abnormal fetuses. Prenatal Diagnosis 1993;13(12):1123‐31. [DOI] [PubMed] [Google Scholar]
Van Lith 1994 {published data only}
- Lith JM, Mantingh A, Pratt JJ. First‐trimester maternal serum immunoreactive inhibin in chromosomally normal and abnormal pregnancies. Obstetrics and Gynecology 1994;83(5 I):661‐4. [PubMed] [Google Scholar]
Veress 1986 {published data only}
- Veress L, Szabo M, Horvath K, Polgar K, Papp Z. [Low maternal serum alpha‐fetoprotein concentration and Down syndrome]. [Hungarian]. Orvosi Hetilap 1986;127(20):1232‐3. [PubMed] [Google Scholar]
Veress 1988 {published data only}
- Veress L, Szabo M, Polgar K, Takacs L, Papp Z. [Prenatal screening for Down's syndrome by measuring the AFP concentration in the maternal serum]. [Hungarian]. Orvosi Hetilap 1988;129(31):1677. [PubMed] [Google Scholar]
Vergani 2008 {published data only}
- Vergani P, Ghidini A, Weiner S, Locatelli A, Pozzi E, Biffi A. Risk assessment for Down syndrome with genetic sonogram in women at risk. Prenatal Diagnosis 2008;28(12):1144‐8. [DOI] [PubMed] [Google Scholar]
Vintzileos 2003 {published data only}
- Vintzileos A, Walters C, Yeo L. Absent nasal bone in the prenatal detection of fetuses with trisomy 21 in a high‐risk population. Obstetrics & Gynecology 2003;101(5):905‐8. [DOI] [PubMed] [Google Scholar]
Wald 1988a {published data only}
- Wald NJ, Cuckle HS, Densem JW, Nanchahal K, Royston P, Chard T, et al. Maternal serum screening for Down's syndrome in early pregnancy. BMJ 1988;297(6653):883‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wald 1988b {published data only}
- Wald NJ, Cuckle HS, Densem JW, Nanchahal K, Canick JA, Haddow JE, et al. Maternal serum unconjugated oestriol as an antenatal screening test for Down's syndrome. British Journal of Obstetrics & Gynaecology 1988;95(4):334‐41. [DOI] [PubMed] [Google Scholar]
Wald 1991 {published data only}
- Wald N, Cuckle H, Wu TS, George L. Maternal serum unconjugated oestriol and human chorionic gonadotrophin levels in twin pregnancies: implications for screening for Down's syndrome. British Journal of Obstetrics & Gynaecology 1991;98(9):905‐8. [DOI] [PubMed] [Google Scholar]
Wald 1992a {published data only}
- Wald NJ, Kennard A, Densem JW, Cuckle HS, Chard T, Butler L. Antenatal maternal serum screening for Down's syndrome: results of a demonstration project.[see comment]. BMJ 1992;305(6850):391‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wald 1992b {published data only}
- Wald NJ, Cuckle HS, Densem JW, Stone RB. Maternal serum unconjugated oestriol and human chorionic gonadotrophin levels in pregnancies with insulin‐dependent diabetes: implications for screening for Down's syndrome. British Journal of Obstetrics & Gynaecology 1992;99(1):51‐3. [DOI] [PubMed] [Google Scholar]
Wald 1992c {published data only}
- Wald NJ, Cuckle HS, Densem JW, Kennard A, Smith D. Maternal serum screening for Down's syndrome: the effect of routine ultrasound scan determination of gestational age and adjustment for maternal weight.[see comment]. British Journal of Obstetrics & Gynaecology 1992;99(2):144‐9. [DOI] [PubMed] [Google Scholar]
Wald 1993 {published data only}
- Wald N, Densem J, Stone R, Cheng R. The use of free ß‐hCG in antenatal screening for Down's syndrome.[see comment]. British Journal of Obstetrics & Gynaecology 1993;100(6):550‐7. [DOI] [PubMed] [Google Scholar]
Wald 1994a {published data only}
- Wald NJ, Densem JW. Maternal serum free alpha‐human chorionic gonadotrophin levels in twin pregnancies: implications for screening for Down's syndrome. Prenatal Diagnosis 1994;14(8):717‐9. [DOI] [PubMed] [Google Scholar]
Wald 1994b {published data only}
- Wald NJ, Watt HC. Choice of serum markers in antenatal screening for Down's syndrome. Journal of Medical Screening 1994;1(2):117‐20. [DOI] [PubMed] [Google Scholar]
Wald 1996a {published data only}
- Wald NJ, Watt HC. Serum markers for Down's syndrome in relation to number of previous births and maternal age. Prenatal Diagnosis 1996;16(8):699‐703. [DOI] [PubMed] [Google Scholar]
Wald 1996b {published data only}
- Wald NJ, George L, Smith D, Densem JW, Petterson K. Serum screening for Down's syndrome between 8 and 14 weeks of pregnancy. International Prenatal Screening Research Group.[see comment]. British Journal of Obstetrics & Gynaecology. 1996;103(5):407‐12. [DOI] [PubMed] [Google Scholar]
Wald 1996c {published data only}
- Wald NJ, Watt HC, George L. Maternal serum inhibin‐A in pregnancies with insulin‐dependent diabetes mellitus: implications for screening for Down's syndrome. Prenatal Diagnosis 1996;16(10):923‐6. [DOI] [PubMed] [Google Scholar]
Wald 1996d {published data only}
- Wald NJ, Densem JW, George L, Muttukrishna S, Knight PG. Prenatal screening for Down's syndrome using inhibin‐A as a serum marker. Prenatal Diagnosis 1996;16(2):143‐53. [DOI] [PubMed] [Google Scholar]
Wald 1997 {published data only}
- Wald NJ, Hackshaw AK. Combining ultrasound and biochemistry in first‐trimester screening for Down's syndrome.[see comment]. Prenatal Diagnosis 1997;17(9):821‐9. [PubMed] [Google Scholar]
Wald 1998 {published data only}
- Wald NJ, Watt HC, Haddow JE, Knight GJ. Screening for Down syndrome at 14 weeks of pregnancy. Prenatal Diagnosis 1998;18(3):291‐3. [DOI] [PubMed] [Google Scholar]
Wald 1999a {published data only}
- Wald NJ, Hackshaw AK, Diamandis EP, Melegos DN. Maternal serum prostate‐specific antigen and Down syndrome in the first and second trimesters of pregnancy. Prenatal Diagnosis 1999;19(7):674‐6. [PubMed] [Google Scholar]
Wald 1999b {published data only}
- Wald NJ, Watt HC, Norgaard Pederson, Christiansen M. SP1 in pregnancies with Down syndrome in the first trimester of pregnancy. Prenatal Diagnosis 1999;19(6):517‐20. [PubMed] [Google Scholar]
Wald 1999c {published data only}
- Wald NJ, White N, Morris JK, Huttly WJ, Canick JA. Serum markers for Down's syndrome in women who have had in vitro fertilisation: implications for antenatal screening. British Journal of Obstetrics & Gynaecology 1999;106(12):1304‐6. [DOI] [PubMed] [Google Scholar]
Wald 1999d {published data only}
- Wald NJ, Watt HC, Hackshaw AK. Integrated screening for Down's syndrome on the basis of tests performed during the first and second trimesters.[see comment]. New England Journal of Medicine 1999;341(7):461‐7. [DOI] [PubMed] [Google Scholar]
Wald 2003c {published data only}
- Wald NJ, Rish S, Hackshaw AK. Combining nuchal translucency and serum markers in prenatal screening for Down syndrome in twin pregnancies. Prenatal Diagnosis 2003;23(7):588‐92. [DOI] [PubMed] [Google Scholar]
Wald 2003d {published data only}
- Wald NJ, Huttly WJ, Hackshaw AK. Antenatal screening for Down's syndrome with the quadruple test.[see comment]. Lancet 2003;361(9360):835‐6. [DOI] [PubMed] [Google Scholar]
Wald 2006 {published data only}
- Wald NJ, Rudnicka AR, Bestwick JP. Sequential and contingent prenatal screening for Down syndrome. Prenatal Diagnosis 2006;26(9):769‐77. [DOI] [PubMed] [Google Scholar]
Wallace 1994 {published data only}
- Wallace EM, Harkness LM, Burns S, Liston WA. Evaluation of maternal serum immunoreactive Inhibin Aas a first trimester marker of Down's syndrome. Clinical Endocrinology 1994;41(4):483‐6. [DOI] [PubMed] [Google Scholar]
Wallace 1997 {published data only}
- Wallace EM, Crossley JA, Ritoe SC, Groome NP, Aitken DA. Maternal serum inhibin‐A in pregnancies complicated by insulin dependent diabetes mellitus. British Journal of Obstetrics & Gynaecology 1997;104(8):946‐8. [DOI] [PubMed] [Google Scholar]
Wang 2010 {published data only}
- Wang E, Chen C, Glimco E, Grobman W. The performance of second trimester long bone ratios for Down syndrome screening is influenced by gestational age. Journal of Maternal‐Fetal & Neonatal Medicine 2010;23(7):642‐5. [DOI] [PubMed] [Google Scholar]
Ward 2005 {published data only}
- Ward A. Nuchal translucency measurement. Synergy (http://www.highbeam.com/doc/1P3‐866108421.html) (accessed 2007) 2005.
Watt 1996 {published data only}
- Watt HC, Wald NJ, Smith D, Kennard A, Densem J. Effect of allowing for ethnic group in prenatal screening for Down's syndrome. Prenatal Diagnosis 1996;16(8):691‐8. [DOI] [PubMed] [Google Scholar]
Watt 1996a {published data only}
- Watt HC, Wald NJ, George L. Maternal serum inhibin‐A levels in twin pregnancies: implications for screening for Down's syndrome. Prenatal Diagnosis 1996;16(10):927‐9. [DOI] [PubMed] [Google Scholar]
Wax 2007 {published data only}
- Wax JR, Pinette MG, Cartin A, Blackstone J. Optimal crown‐rump length for measuring the nuchal translucency. Journal of Clinical Ultrasound 2007;35(6):302‐4. [DOI] [PubMed] [Google Scholar]
Weinans 2001 {published data only}
- Weinans MJN, Pratt JJ, De Wolf, Mantingh A. First‐trimester maternal serum human thyroid‐stimulating hormone in chromosomally normal and Down syndrome pregnancies. Prenatal Diagnosis 2001;21(9):723‐5. [PubMed] [Google Scholar]
Weinans 2004 {published data only}
- Weinans MJN, Kooij L, Müller MA, Bilardo KM, Van Lith, Tymstra T. A comparison of the impact of screen‐positive results obtained from ultrasound and biochemical screening for Down syndrome in the first trimester: A pilot study. Prenatal Diagnosis 2004;24(5):347‐51. [DOI] [PubMed] [Google Scholar]
Weisz 2007 {published data only}
- Weisz B, Pandya P, Chitty L, Jones P, Huttly W, Rodeck C. Practical issues drawn from the implementation of the integrated test for Down syndrome screening into routine clinical practice. BJOG: an international journal of obstetrics and gnaecology 2007;114(4):493‐7. [DOI] [PubMed] [Google Scholar]
Welborn 1994 {published data only}
- Welborn JL, Timm NS. Trisomy 21 and cystic hygromas in early gestational age fetuses. American Journal of Perinatology 1994;11(1):19‐20. [DOI] [PubMed] [Google Scholar]
Wenstrom 1993 {published data only}
- Wenstrom KD, Williamson RA, Grant SS, Hudson JD, Getchell JP. Evaluation of multiple‐marker screening for Down syndrome in a statewide population. American Journal of Obstetrics and Gynecology 1993;169(4):793‐7. [DOI] [PubMed] [Google Scholar]
Wenstrom 1995a {published data only}
- Wenstrom KD, Owen J, Boots L, Ethier M. The influence of maternal weight on human chorionic gonadotropin in the multiple‐marker screening test for fetal Down syndrome. American Journal of Obstetrics and Gynecology 1995;173(4):1297‐300. [DOI] [PubMed] [Google Scholar]
Wenstrom 1995b {published data only}
- Wenstrom KD, Desai R, Owen J, Dubard MB, Boots L. Comparison of multiple‐marker screening with amniocentesis for the detection of fetal aneuploidy in women greater than or equal35 years old. American Journal of Obstetrics and Gynecology 1995;173(4):1287‐92. [DOI] [PubMed] [Google Scholar]
Wetta 2011 {published data only}
- Wetta L, Biggio J Jr, Owen J. Use of ethnic‐specific medians for Hispanic patients reduces ethnic disparities in multiple marker screening. Prenatal Diagnosis 2011;31(4):331‐3. [DOI] [PubMed] [Google Scholar]
Whitlow 1998a {published data only}
- Whitlow BJ, Lazanakis ML, Kadir RA, Chatzipapas I, Economides DL. The significance of choroid plexus cysts, echogenic heart foci and renal pyelectasis in the first trimester. Ultrasound in Obstetrics & Gynecology 1998;12(6):385‐90. [DOI] [PubMed] [Google Scholar]
Whitlow 1998b {published data only}
- Whitlow BJ, Economides DL. First trimester detection of fetal abnormalities in an unselected population. Contemporary Reviews in Obstetrics and Gynaecology 1998;10(4):245‐53. [DOI] [PubMed] [Google Scholar]
Whitlow 1999 {published data only}
- Whitlow BJ, Chatzipapas IK, Lazanakis ML, Kadir RA, Economides DL. The value of sonography in early pregnancy for the detection of fetal abnormalities in an unselected population. British Journal of Obstetrics & Gynaecology 1999;106(9):929‐36. [DOI] [PubMed] [Google Scholar]
Williamson 1994 {published data only}
- Williamson R. Expanded maternal serum alpha fetoprotein screening. Iowa Medicine 1994;84(9):397‐400. [PubMed] [Google Scholar]
Wilson 2000 {published data only}
- Wilson K. New first‐trimester prenatal screening for down syndrome. Laboratory Medicine 2000;31(11):591. [Google Scholar]
Wojdemann 2001 {published data only}
- Wojdemann KR, Larsen SO, Shalmi A, Sundberg K, Christiansen M, Tabor A. First trimester screening for Down syndrome and assisted reproduction: no basis for concern. Prenatal Diagnosis 2001;21(7):563‐5. [DOI] [PubMed] [Google Scholar]
Wong 2003 {published data only}
- Wong SF, Choi H, Ho LC. Nasal bone hypoplasia: is it a common finding amongst chromosomally normal fetuses of southern Chinese women?. Gynecologic & Obstetric Investigation 2003;56(2):99‐101. [DOI] [PubMed] [Google Scholar]
Wright 2006 {published data only}
- Wright D, Bradbury I, Cuckle H, Gardosi J, Tonks A, Standing S, et al. Three‐stage contingent screening for Down syndrome. Prenatal Diagnosis 2006;26(6):528‐34. [DOI] [PubMed] [Google Scholar]
Wright 2007 {published data only}
- Wright D, Spencer K, Nix B. First trimester screening for Down syndrome using free beta hCG, total hCG and PAPP‐A: an exploratory study. Prenatal Diagnosis 2007;27(12):1118‐22. [DOI] [PubMed] [Google Scholar]
Xie 2010 {published data only}
- Xie Z, Lu S, Li H. Contingent triple‐screening for Down syndrome in the second trimester: a feasibility study in Mainland Chinese population. Prenatal Diagnosis 2010;30(1):74‐6. [DOI] [PubMed] [Google Scholar]
Yagel 1998 {published data only}
- Yagel S, Anteby EY, Hochner‐Celnikier D, Ariel I, Chaap T, Ben Neriah Z. The role of midtrimester targeted fetal organ screening combined with the "triple test" and maternal age in the diagnosis of trisomy 21: a retrospective study. American Journal of Obstetrics and Gynecology 1998;178(1):40‐4. [DOI] [PubMed] [Google Scholar]
Yamamoto 2001a {published data only}
- Yamamoto R, Azuma M, Kishida T, Yamada H, Satomura S, Fujimoto S. Total alpha‐fetoprotein and Lens culinaris agglutinin‐reactive alpha‐fetoprotein in fetal chromosomal abnormalities. BJOG: an international journal of obstetrics and gynaecology 2001;108(11):1154‐8. [DOI] [PubMed] [Google Scholar]
Yamamoto 2001b {published data only}
- Yamamoto R, Azuma M, Hoshi N, Kishida T, Satomura S, Fujimoto S. Lens culinaris agglutinin‐reactive alpha‐fetoprotein, an alternative variant to alpha‐fetoprotein in prenatal screening for Down's syndrome. Human Reproduction 2001;16(11):2438‐44. [DOI] [PubMed] [Google Scholar]
Yamamoto 2001c {published data only}
- Yamamoto R, Azuma M, Wakui Y, Kishida T, Yamada H, Okuyama K, et al. Alpha‐fetoprotein microheterogeneity: a potential biochemical marker for Down's syndrome. Clinica Chimica Acta 2001;304(1‐2):137‐41. [DOI] [PubMed] [Google Scholar]
Yaron 2001 {published data only}
- Yaron Y, Wolman I, Kupferminc MJ, Ochshorn Y, Many A, Orr‐Urtreger A. Effect of fetal gender on first trimester markers and on Down syndrome screening. Prenatal Diagnosis 2001;21(12):1027‐30. [DOI] [PubMed] [Google Scholar]
Ye 1995 {published data only}
- Ye Guoling Liao Shixiu, Zhao Xiaolan. The possibility of prenatal screening for fetal abnormalities in second‐trimester pregnancies by measuring AFP, ß‐HCG and uE‐3 levels. Xi'an Yike Daxue Xuebao 1995;16(4):408‐11. [Google Scholar]
Yoshida 2000 {published data only}
- Yoshida K, Kuwabara Y, Tanaka T, Onda T, Kudo R, Yamamoto H, et al. Dimeric Inhibin A as a fourth marker for Down's syndrome maternal serum screening in native Japanese women. Journal of Obstetrics and Gynaecology Research 2000;26(3):171‐4. [DOI] [PubMed] [Google Scholar]
Zalel 2008 {published data only}
- Zalel Y, Achiron R, Yagel S, Kivilevitch Z. Fetal aberrant right subclavian artery in normal and Down syndrome fetuses. Ultrasound in Obstetrics & Gynecology 2008;31(1):25‐9. [DOI] [PubMed] [Google Scholar]
Zeitune 1991 {published data only}
- Zeitune M, Aitken DA, Crossley JA, Yates JRW, Cooke A, Ferguson‐Smith MA. Estimating the risk of a fetal autosomal trisomy at mid‐trimester using maternal serum alpha‐fetoprotein and age: A retrospective study of 142 pregnancies. Prenatal Diagnosis 1991;11(11):847‐57. [DOI] [PubMed] [Google Scholar]
Zelop 2005 {published data only}
- Zelop CM, Milewski E, Brault K, Benn P, Borgida AF, Egan JFX. Variation of fetal nasal bone length in second‐trimester fetuses according to race and ethnicity. Journal of Ultrasound in Medicine 2005;24(11):1487‐9. [DOI] [PubMed] [Google Scholar]
Zhang 2011 {published data only}
- Zhang J, Lambert‐Messerlian G, Palomaki GE, Canick JA. Impact of smoking on maternal serum markers and prenatal screening in the first and second trimesters. Prenatal Diagnosis 2011;31(6):583‐8. [DOI] [PubMed] [Google Scholar]
Zhao 1998 {published data only}
- Zhao Xiaolan, Ye Guoling, Liu Qi. Using maternal serum PAPP‐A and other pregnancy‐associated proteins in screening for fetal abnormalities. Xi'an Yike Daxue Xuebao 1998;19(1):94‐6, 110. [Google Scholar]
Zhong 2011 {published data only}
- Zhong Y, Longman R, Bradshaw R, Odibo AO. The genetic sonogram: comparing the use of likelihood ratios versus logistic regression coefficients for Down syndrome screening. Journal of Ultrasound in Medicine 2011;30(4):463‐9. [DOI] [PubMed] [Google Scholar]
Zoppi 2003 {published data only}
- Zoppi MA, Ibba RM, Floris M, Manca F, Axiana C, Monni G. Changes in nuchal translucency thickness in normal and abnormal karyotype fetuses. BJOG: an international journal of obstetrics and gynaecology 2003;110(6):584‐8. [PubMed] [Google Scholar]
Additional references
Alfirevic 2003
- Alfirevic Z, Sundberg K, Brigham S. Amniocentesis and chorionic villus sampling for prenatal diagnosis. Cochrane Database of Systematic Reviews 2003, Issue 3. [DOI: 10.1002/14651858.CD003252] [DOI] [PMC free article] [PubMed] [Google Scholar]
Alfirevic 2004
- Alfirevic Z, Neilson JP. Antenatal screening for Down's syndrome. BMJ 2004;9(329(7470)):811‐2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Alldred 2010
- Alldred SK, Deeks JJ, Neilson JP, Alfirevic Z. Antenatal screening for Down's syndrome: generic protocol. Cochrane Database of Systematic Reviews 2010, Issue 4. [DOI: 10.1002/14651858.CD007384.pub2] [DOI] [Google Scholar]
Alldred 2012
- Alldred SK, Deeks JJ, Guo B, Neilson JP, Alfirevic Z. Second trimester serum tests for Down's Syndrome screening. Cochrane Database of Systematic Reviews 2012, Issue 6. [DOI: 10.1002/14651858.CD009925] [DOI] [PMC free article] [PubMed] [Google Scholar]
Alldred 2015
- Alldred SK, Takwoingi Y, Guo B, Pennant M, Deeks JJ, Neilson JP, et al. First trimester serum tests for Down's syndrome screening. Cochrane Database of Systematic Reviews 2015, Issue 11. [DOI: 10.1002/14651858.CD011975] [DOI] [PMC free article] [PubMed] [Google Scholar]
Alldred 2015a
- Alldred SK, Guo B, Takwoingi Y, Pennant M, Wisniewski S, Deeks JJ, Neilson JP, et al. Urine tests for Down's syndrome screening. Cochrane Database of Systematic Reviews 2015, Issue 12. [DOI: 10.1002/14651858.CD011984] [DOI] [PMC free article] [PubMed] [Google Scholar]
Bersinger 1995
- Bersinger NA, Zakher A, Huber U, Pescia G, Schneider H. A sensitive enzyme immunoassay for pregnancy‐associated plasma protein A (PAPP‐A): a possible first trimester method of screening for Down syndrome and other trisomies. Archives of Gynecology and Obstetrics 1995;256(4):185‐92. [DOI] [PubMed] [Google Scholar]
Bogart 1987
- Bogart MH, Pandian MR, Jones OW. Abnormal maternal serum chorionic gonadotropin levels in pregnancies with fetal chromosome abnormalities. Prenatal Diagnosis 1987;7(9):623‐30. [DOI] [PubMed] [Google Scholar]
Cole 1999
- Cole LA, Rinne KM, Mahajan SM, Oz UA, Shahabi S, Mahoney MJ, et al. Urinary screening tests for fetal Down syndrome: I. Fresh beta‐core fragment (see comment). Prenatal Diagnosis 1999;19(4):340‐50. [DOI] [PubMed] [Google Scholar]
Cuckle 1984a
- Cuckle HS, Wald NJ, Lindenbaum RH. Maternal serum alpha‐fetoprotein measurement: a screening test for Down syndrome. Lancet 1984;1(8383):926‐9. [DOI] [PubMed] [Google Scholar]
Cuckle 1995
Laigaard 2003
- Laigaard J, Sorensen T, Frohlich C, Pedersen BN, Christiansen M, Schiott K, et al. ADAM12: a novel first‐trimester maternal serum marker for Down syndrome. Prenatal Diagnosis 2003;23(13):1086‐91. [PUBMED: 14691998] [DOI] [PubMed] [Google Scholar]
Macri 1990
- Macri JN, Kasturi RV, Krantz DA, Cook EJ, Moore ND, Young JA, et al. Maternal serum Down syndrome screening: free beta‐protein is a more effective marker than human chorionic gonadotropin. American Journal of Obstetrics and Gynecology 1990;163(4 Pt 1):1248‐53. [DOI] [PubMed] [Google Scholar]
Macri 1993
- Macri JN, Spencer K, Aitken D, Garver K, Buchanan PD, Muller F, et al. First‐trimester free beta (hCG) screening for Down syndrome. Prenatal Diagnosis 1993;13(7):557‐62. [DOI] [PubMed] [Google Scholar]
Mol 1999
- Mol BW, Lijmer JG, Meulen J, Pajkrt E, Bilardo CM, Bossuyt PM. Effect of study design on the association between nuchal translucency measurement and Down syndrome. Obstetrics and Gynecology 1999;94(5 Pt 2):864‐9. [DOI] [PubMed] [Google Scholar]
Penrose 1933
- Penrose LS. The relative effects of parental and maternal age in mongolism. Journal of Genetics 1933;27:219‐24. [DOI] [PubMed] [Google Scholar]
Spencer 2008a
- Spencer K, Vereecken A, Cowans NJ. Maternal serum ADAM12s as a potential marker of trisomy 21 prior to 10 weeks of gestation. Prenatal Diagnosis 2008;28(3):209‐11. [PUBMED: 18264948] [DOI] [PubMed] [Google Scholar]
Steele 1966
- Steele MW, Breg WR. Chromosome analysis of human amniotic‐fluid cells. Lancet 1966;1:383‐5. [DOI] [PubMed] [Google Scholar]
Valenti 1968
- Valenti C, Schutta EJ, Kehaty T. Prenatal diagnosis of Down's syndrome. Lancet 1968;2:220. [DOI] [PubMed] [Google Scholar]
Wald 2003a
- Wald NJ, Rodeck C, Hackshaw AK, Walters J, Chitty L, Mackinson AM. First and second trimester antenatal screening for Down's syndrome: the results of the Serum, Urine and Ultrasound Screening Study (SURUSS). Health Technology Assessment 2003;7(11):1‐77. [DOI] [PubMed] [Google Scholar]
Wallace 1995
- Wallace EM, Grant VE, Swanston IA, Groome NP. Evaluation of maternal serum dimeric inhibin A as a first‐trimester marker of Down's syndrome. Prenatal Diagnosis 1995;15(4):359‐62. [DOI] [PubMed] [Google Scholar]
Whiting 2003
- Whiting P, Rutjes AW, Reitsma JB, Bossuyt PM, Kleijnen J. The development of QUADAS: A tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC Medical Research Methodology 2003;3:25. [DOI] [PMC free article] [PubMed] [Google Scholar]


