Abstract
Background
Tumour lysis syndrome (TLS) is a serious complication of malignancies and can result in renal failure or death. Previous reviews did not find clear evidence of benefit of urate oxidase in children with cancer. This review is the second update of a previously published Cochrane review.
Objectives
To assess the effects and safety of urate oxidase for the prevention and treatment of TLS in children with malignancies.
Search methods
In March 2016 we searched CENTRAL, MEDLINE, Embase, and CINAHL. In addition, we searched the reference lists of all identified relevant papers, trials registers and other databases. We also screened conference proceedings and we contacted experts in the field and the manufacturer of rasburicase, Sanofi‐aventis.
Selection criteria
Randomised controlled trials (RCT) and controlled clinical trials (CCT) of urate oxidase for the prevention or treatment of TLS in children under 18 years with any malignancy.
Data collection and analysis
Two review authors independently extracted trial data and assessed individual trial quality. We used risk ratios (RR) for dichotomous data and mean difference (MD) for continuous data.
Main results
We included seven trials, involving 471 participants in the treatment groups and 603 participants in the control groups. No new studies were identified in the update. One RCT and five CCTs compared urate oxidase and allopurinol. Three trials tested Uricozyme, and three trials tested rasburicase for the prevention of TLS.
The RCT did not evaluate the primary outcome (incidence of clinical TLS). It showed no clear evidence of a difference in mortality (both all‐cause mortality (Fisher's exact test P = 0.23) and mortality due to TLS (no deaths in either group)), renal failure (Fisher's exact test P = 0.46), and adverse effects between the treatment and the control groups (Fisher's exact test P = 1.0). The frequency of normalisation of uric acid at four hours (10 out of 10 participants in the treatment group versus zero out of nine participants in the control group, Fisher's exact test P < 0.001) and area under the curve of uric acid at four days (MD ‐201.00 mg/dLhr, 95% CI ‐258.05 mg/dLhr to ‐143.95 mg/dLhr; P < 0.00001) were significantly better in the treatment group.
One CCT evaluated the primary outcome; no clear evidence of a difference was identified between the treatment and the control groups (RR 0.77, 95% CI 0.44 to 1.33; P = 0.34). Pooled results of three CCTs showed significantly lower mortality due to TLS in the treatment group (RR 0.05, 95% CI 0.00 to 0.89; P = 0.04); no clear evidence of a difference in all‐cause mortality was identified between the groups (RR 0.19, 95% CI 0.01 to 3.42; P = 0.26). Pooled results from five CCTs showed significantly lower incidence of renal failure in the treatment group (RR 0.26, 95% CI 0.08 to 0.89; P = 0.03). Results of CCTs also showed significantly lower uric acid in the treatment group at two days (three CCTs: MD ‐3.80 mg/dL, 95% CI ‐7.37 mg/dL to ‐0.24 mg/dL; P = 0.04), three days (two CCTs: MD ‐3.13 mg/dL, 95% CI ‐6.12 mg/dL to ‐0.14 mg/dL; P = 0.04), four days (two CCTs: MD ‐4.60 mg/dL, 95% CI ‐6.39 mg/dL to ‐2.81 mg/dL; P < 0.00001), and seven days (one CCT: MD ‐1.74 mg/dL, 95% CI ‐3.01 mg/dL to ‐0.47 mg/dL; P = 0.007) after therapy, but not one day (three CCTs: MD ‐3.00 mg/dL, 95% CI ‐7.61 mg/dL to 1.60 mg/dL; P = 0.2), five days (one CCT: MD ‐1.02 mg/dL, 95% CI ‐2.24 mg/dL to 0.20 mg/dL; P = 0.1), and 12 days (one CCT: MD ‐0.80 mg/dL, 95% CI ‐2.51 mg/dL to 0.91 mg/dL; P = 0.36) after therapy. Pooled results from three CCTs showed higher frequency of adverse effects in participants who received urate oxidase (RR 9.10, 95% CI 1.29 to 64.00; P = 0.03).
Another included RCT, with 30 participants, compared different doses of rasburicase (0.2 mg/kg versus 0.15 mg/kg). The primary outcome was not evaluated. No clear evidence of a difference in mortality (all‐cause mortality (Fisher's exact test P = 1.0) and mortality due to TLS (no deaths in both groups)) and renal failure (no renal failure in both groups) was identified. It demonstrated no clear evidence of a difference in uric acid normalisation (RR 1.07, 95% CI 0.89 to 1.28; P = 0.49) and uric acid level at four hours (MD 8.10%, 95% CI ‐0.99% to 17.19%; P = 0.08). Common adverse events of urate oxidase included hypersensitivity, haemolysis, and anaemia, but no clear evidence of a difference between treatment groups was identified (RR 0.54, 95% CI 0.12 to 2.48; P = 0.42).
The quality of evidence ranks from very low to low because of imprecise results, and all included trials were highly susceptible to biases.
Authors' conclusions
Although urate oxidase might be effective in reducing serum uric acid, it is unclear whether it reduces clinical TLS, renal failure, or mortality. Adverse effects might be more common for urate oxidase compared with allopurinol. Clinicians should weigh the potential benefits of reducing uric acid and uncertain benefits of preventing mortality or renal failure from TLS against the potential risk of adverse effects.
Plain language summary
Urate oxidase for the prevention and treatment of complications from massive lysis (breakdown) of tumour cells in children with cancer
Review question
We reviewed the evidence of the effects and safety of urate oxidase for the prevention and treatment of tumour lysis syndrome (TLS) in children with malignancies.
Background
TLS occurs when uric acid and other cellular substances are rapidly released into the circulation when tumour cells are broken down spontaneously or during treatment. Uric acid does not dissolve easily, therefore it can build up in the kidney, resulting in kidney failure and possibly death. Urate oxidase is an enzyme that can be administered to people at risk of TLS to convert uric acid to allantoin, which is easily dissolved and then readily excreted by the kidneys. Therefore, urate oxidase may be able to prevent or treat TLS in people with malignancies.
Study characteristics
The evidence is current to March 2016.
We found seven trials (1074 participants). No new studies were identified in this update. Six studies compared urate oxidase and allopurinol. Three trials tested Uricozyme, and three trials tested rasburicase. One study (30 participants) compared different doses of rasburicase.
Key results
The current systematic review of (randomised) controlled clinical trials found that although urate oxidase might be effective in reducing serum uric acid level, it has not been confirmed to reduce renal failure or mortality from TLS in children with cancer. Adverse effects might be more common in people who receive urate oxidase compared with allopurinol. Urate oxidase needs to be further evaluated, especially in high‐risk patients, such as those with high‐risk leukaemia and lymphoma.
Quality of the evidence
The quality of evidence ranks from very low to low because of imprecise results, and all included trials were highly susceptible to biases.
Summary of findings
Background
Tumour lysis syndrome (TLS) is a serious complication of malignancies that can occur spontaneously in the presence of rapidly proliferating tumour cells or during treatment because of rapid cell lysis, leading to release of intracellular components that may result in hyperkalaemia, hyperphosphataemia, hypocalcaemia, or hyperuricaemia. Hyperuricaemia and hyperphosphataemia can result in crystallisation in the renal tubules causing obstructive uropathy and renal failure. Other severe consequences of TLS include cardiac arrhythmia and sudden death from hyperkalaemia (Navolanic 2003; Rampello 2006). The Cairo‐Bishop definition for laboratory TLS is the development of any two or more of the following four criteria within three days before or seven days after the initiation of chemotherapy: uric acid level of 8 mg/dL or more; potassium level of 6 mmol/L or more; phosphate level of 6.65 mg/dL or more; and calcium level of 7 mg/dL or less. A 25% increase from baseline for uric acid, potassium, or phosphate levels or a 25% decrease from baseline for calcium level is an alternative threshold (Cairo 2004). The Cairo‐Bishop definition for clinical TLS is the presence of laboratory TLS and one or more of the following three criteria: serum creatinine level 1.5 times or more than the upper limit of normal; cardiac arrhythmias; sudden death; or seizures.
Risk factors for TLS include high proliferation rate, large tumour burden, and high chemosensitivity. A high white blood cell (WBC) count in leukaemia (> 50 x 10⁹/L) or a high lactate dehydrogenase (LDH) level in lymphoma indicates high tumour burden. Certain malignancies, such as Burkitt's lymphoma, are associated with a very high risk of TLS because of rapid tumour cell turnover (Wössmann 2003). The incidence of TLS varies among studies. A retrospective review of acute lymphoblastic leukaemia, acute myeloid leukaemia, and non‐Hodgkin's lymphoma found that the frequency of TLS was 3.4%, 5.2%, and 6.1%, respectively, and it accounts for 0.9% of cancer mortality (Annemans 2003a). The mortality rate of TLS has been estimated to be about 17.5% (Annemans 2003a). The medical costs of hyperuricaemia and TLS are substantial. The cost of hyperuricaemia without TLS has been estimated to be EUR 672 per patient for the whole treatment, and the cost of TLS, EUR 7342 (Annemans 2003a). The cost of TLS requiring dialysis has been shown to be even higher (EUR 17,706 on average) (Annemans 2003a).
Aggressive hydration and allopurinol, with or without urinary alkalinization with bicarbonate, is the standard prophylaxis for TLS. Allopurinol is a xanthine oxidase inhibitor, which prevents the formation of uric acid but does not catabolise (so degrade and detoxify) existing uric acid. Allopurinol is therefore not an effective treatment for established TLS, since it does not promote uric acid clearance. Because of the inhibition of xanthine oxidase, allopurinol increases the level of uric acid precursors, hypoxanthine and xanthine. As xanthine is less soluble than uric acid, it may precipitate in renal tubules causing xanthine nephropathy (kidney disease) or xanthine stones (Greene 1969).
Urate oxidase is an alternative agent used for the treatment or prophylaxis of hyperuricaemia in people who are at high risk of TLS. Urate oxidase converts uric acid to allantoin, which is five to 10 times more soluble than uric acid and readily excreted in urine. A non‐recombinant form of urate oxidase has been available in Europe for more than 20 years, but it is associated with acute hypersensitivity reactions in 4.5% of patients (Yim 2003). Rasburicase, a relatively new, recombinant urate oxidase enzyme produced by a genetically modified Saccharomyces cerevisiae strain, has now replaced the older agent and is widely used. Reported advantages of urate oxidase over allopurinol include its ability to catabolise existing uric acid in established TLS; no increased risk of xanthine stone formation; no requirement for dose adjustment in acute renal failure; lack of clinically relevant drug‐drug interaction; and lower incidence of adverse reactions, such as skin rash, fever, eosinophilia, and Stevens Johnson syndrome (Gutierrez‐Macias 2005; Sanofi 2011). Anecdotal reports and case series have indicated that urate oxidase may be effective in the prevention and treatment of TLS (Bosly 2003; Coiffier 2003; Hummel 2003; Hutcherson 2006; Jeha 2005; Lascombes 1998; Lee 2003; Liu 2005; McDonnell 2006; Pui 2001a; Pui 2001b; Shin 2006; Trifilio 2006; Wang 2006), resulting in a significant reduction of serum uric acid level and a low incidence of renal failure requiring dialysis. In addition, the use of urate oxidase has been reported to be cost‐effective for the prevention and treatment of TLS in both children and adults (Annemans 2003b). However, it is not entirely certain whether the existing evidence is sufficiently rigorous to support the routine use of urate oxidase as prophylaxis in children with malignancies at risk of TLS, or as a treatment for established laboratory or clinical TLS. It is also uncertain whether single or multiple doses of urate oxidase should be used or which types of high‐risk patients benefit most from prophylactic administration of urate oxidase. Although there are consensus guidelines developed for the management of TLS (Cairo 2010; Coiffier 2008; Tosi 2008), they did not include the latest evidence from systematic review. Therefore, we examined the efficacy and safety of urate oxidase in children with malignancies in a systematic review of RCTs and CCTs (Cheuk 2010; Cheuk 2014). This is the second update of that systematic review.
Objectives
To assess the effects and safety of urate oxidase for the prevention and treatment of TLS in children with malignancies.
Methods
Criteria for considering studies for this review
Types of studies
We included randomised controlled trials (RCTs) in the review. We also planned to include controlled clinical trials (CCTs) if no (or few) RCTs were available. A CCT is a study that compares one or more intervention groups to one or more control groups. We included historical controlled studies.
Types of participants
We included participants under 18 years of age with all types of cancer, including haematological malignancies and solid tumours.
Types of interventions
We included trials evaluating all preparations of urate oxidase. The control interventions could be placebo; no treatment; or other treatment, such as allopurinol. We also included trials comparing urate oxidase combined with other treatment versus the same other treatment alone, and trials comparing different doses or different preparations of urate oxidase.
Types of outcome measures
Prevention
For evaluation of urate oxidase as prevention for TLS, we assessed the following outcome measures.
Primary outcome
Incidence of clinical TLS according to Cairo‐Bishop definition (Cairo 2004)
Secondary outcomes
Incidence of laboratory TLS according to Cairo‐Bishop definition (Cairo 2004)
Mortality associated with TLS and combined with other reasons
Incidence of renal failure requiring renal replacement therapy associated with TLS
Frequency of normalisation of serum uric acid level
Duration before normalisation of serum uric acid level
Change in serum uric acid level
Area under the curve (AUC) of uric acid level
Frequency of adverse effects
Treatment
For the evaluation of urate oxidase as treatment for TLS, we assessed the following outcome measures.
Primary outcome
Mortality associated with TLS and combined with other reasons
Secondary outcomes
Incidence of renal failure requiring renal replacement therapy associated with TLS
Frequency of normalisation of serum uric acid level
Duration before normalisation of serum uric acid level
Change in serum uric acid level
AUC of uric acid level
Change in serum phosphate level
Change in serum potassium level
Change in serum creatinine level
Change in serum calcium level
Frequency of adverse effects
Search methods for identification of studies
There was no language restriction in the search and inclusion of studies.
Electronic searches
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library (issue 2, 2016), MEDLINE/PubMed (1966 to 14 March 2016), Embase (Ovid) (1980 to 14 March 2016), and CINAHL (Cumulative Index to Nursing and Allied Health Literature) (EBSCO) (1982 to 14 March 2016).
The search strategies used for the different electronic databases (using a combination of controlled vocabulary and text word terms) are shown in the Appendices (Appendix 1; Appendix 2; Appendix 3; Appendix 4).
We also explored other internet sources (on 15 March 2016), using keywords in Appendix 5:
the NHS' National Research Register (www.ukctg.nihr.ac.uk/);
the US National Institutes of Health Ongoing Trials Register (www.clinicaltrials.gov/);
World Health Organization International Clinical Trials Registry Platform (apps.who.int/trialsearch/)
metaRegister of Controlled Trials (www.isrctn.com/page/mrct); and
ProQuest Dissertations & Theses Database (www.proquest.com/).
Searching other resources
We searched the reference lists of all identified relevant papers for further studies. We also handsearched abstracts from the meetings of the ASCO (American Society of Clinical Oncology), ESMO (European Society for Medical Oncology), and SIOP (International Society of Paediatric Oncology) from 1993 up to and including 2015, using keywords in Appendix 6.
We also included articles published only in abstract form if we could contact the authors to provide essential details for appraisal and analysis. If the process of searching many different sources brought to light direct or indirect references to unpublished studies, we planned to obtain copies of such unpublished material. In addition, we contacted colleagues and experts in the field to ascertain any unpublished or ongoing studies. We also contacted the manufacturer of rasburicase, Sanofi‐aventis, for published and unpublished clinical studies.
Data collection and analysis
Selection of studies
Two authors (the first and second authors) independently reviewed titles and abstracts of references retrieved from the searches and selected all potentially relevant studies. The same authors obtained copies of these articles and reviewed them independently against the above‐mentioned inclusion and exclusion criteria for study selection. Authors were not blinded to the names of the trial authors, institutions, or journal of publication. We planned for the third author to resolve any discrepancies regarding selection of studies if necessary, but there was no discrepancy and the third author was not called upon. We have constructed a flow diagram. We excluded multiple publications reporting the same group of participants or its subsets.
Data extraction and management
Two authors extracted data from included trials, independently. We planned for the third author to resolve any discrepancies regarding data extraction if necessary, but there was no discrepancy and the third author was not called upon.
We extracted the following data.
-
Study methods
design (i.e. RCT or CCT)
randomisation method (including list generation)
method of allocation concealment
blinding method
stratification factors
-
Participants
inclusion/exclusion criteria
number of participants entering the trial, number of participants randomised, number of excluded participants (with reasons), and number of evaluable participants
age and gender distribution
type of malignancies
treatments for the malignancies (chemotherapy, radiotherapy, autologous stem cell transplant, allogeneic stem cell transplant)
baseline renal function, uric acid level, potassium level, phosphate level, calcium level, lactate dehydrogenase (LDH) level, white blood cell (WBC) counts (for leukaemia), rate of decrease of WBC (for leukaemia), and sizes of the liver and spleen
-
Intervention and control
type of uric oxidase
type of control treatment
details of administration of urate oxidase, including dosage and schedules
details of co‐interventions
-
Follow‐up data
duration of follow‐up
loss to follow up
-
Outcome data
serial uric acid levels measurement
days to normalisation of uric acid level
number of criteria of laboratory TLS according to Cairo‐Bishop definition (Cairo 2004)
number of criteria of clinical TLS according to Cairo‐Bishop definition (Cairo 2004)
change in serum potassium, calcium, phosphorus, and creatinine levels
adverse effects
-
Analysis data
methods of analysis (intention‐to‐treat or per‐protocol analysis)
comparability of groups at baseline (yes/no);
statistical methods
One author entered the data into Review Manager 5 (RevMan 5) (RevMan 2014); the other authors then checked the data.
Assessment of risk of bias in included studies
Two authors (the first and second authors) independently assessed the methodological quality of each eligible trial. We planned for the third author to resolve any discrepancies regarding risk of bias assessment if necessary, but there was no discrepancy and the third author was not called upon. Where necessary, we sought additional information from the principal investigator of the trial concerned.
We included the following items to assess the methodological quality of RCTs in the update of the review, according to the latest recommendation in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011) and the most recent 'Risk of bias' criteria as recommended by Cochrane Childhood Cancer (Kremer 2016):
random sequence generation (selection bias);
allocation concealment (selection bias);
blinding of participants and personnel (performance bias);
blinding of outcome assessment (detection bias) ‐ each outcome was assessed separately;
incomplete outcome data (attrition bias) ‐ each outcome was assessed separately;
selective reporting (reporting bias); and
other bias.
Similarly, we assessed controlled clinical trials for the above‐mentioned types of biases; we expected them not to incorporate random allocation of treatment groups or perform allocation concealment. Because of non‐random treatment group allocation, they were also susceptible to confounding, and we examined possible confounding factors, including age of the participant, types of malignancies, baseline renal function, WBC counts, LDH level, uric acid levels, and intensity of chemotherapy.
Measures of treatment effect
We used risk ratio (RR) estimations with 95% confidence intervals (CI) for dichotomous outcomes. We did not calculate a RR if there was only one study available for a particular outcome, and there was no event in one of the groups. We used the Fisher's exact test (performed using SPSS version 19 (SPSS 2010)) to determine the P value in such situations. We used mean difference (MD) estimations with 95% CI for continuous outcomes. We analysed all participants in the treatment groups to which they were allocated (intention‐to‐treat (ITT) analyses) if there were no missing data; we planned to perform per‐protocol analyses if information for intention‐to‐treat analyses was lacking. We did not impute missing data. We planned to consider cost‐effectiveness of interventions if relevant data were available.
Dealing with missing data
We contacted the authors of included studies to ask them to supply missing data. We assessed missing data and dropouts/attrition for each included study and assessed and discussed the extent to which the missing data could alter the results/conclusions of the review. If, for a particular outcome, less than 70% of participants allocated to the treatments were reported on at the end of the trial, we reported those data, but considered them prone to bias.
Assessment of reporting biases
We planned to generate a funnel plot (effect size against standard error) if we found sufficient studies (more than five). Asymmetry could be due to publication bias, but could also be due to a relationship between trial size and effect size. In the event that we found a relationship, we planned to examine clinical diversity of the studies (Egger 1997). However, there were not enough studies available to prepare a reliable funnel plot.
Data synthesis
Where the interventions were the same or similar enough, we synthesised results in a meta‐analysis if there was no important clinical heterogeneity. If no significant statistical heterogeneity was present, we synthesised the data using a fixed‐effect model. If there was unexplained heterogeneity, we used a random‐effects model in the meta‐analysis. We produced a 'Summary of findings' table for each comparison according to the recommendations in the Cochrane Handbook of Systematic Reviews of Interventions (Schünemann 2011a), to summarise the results for the outcomes (incidence of clinical TLS, mortality due to TLS, renal failure requiring renal replacement therapy, normalisation of serum uric acid, percentage reduction in serum uric acid level, AUC of serum uric acid level, and frequency of adverse events). We provided grading of the quality of evidence according to the GRADE system (Schünemann 2011b) using the GRADE profiler software (GRADEpro 2015). The GRADE system includes five criteria (study limitations, consistency of effect, imprecision, indirectness and publication bias) to assess the quality of evidence (Schünemann 2011b).
Subgroup analysis and investigation of heterogeneity
If data permitted, we conducted subgroup analyses for the following:
different types of malignancies (acute leukaemia, lymphoma, solid tumour);
different number of doses of urate oxidase (single dose, two doses, three or more doses); and
different levels of risk of TLS (participants with rapid cell turnover, high LDH, or baseline hyperuricaemia).
If two or more included trials reported the same outcomes for the same subgroups, and no significant heterogeneity was present, we combined their results in meta‐analyses. We assessed clinical heterogeneity by comparing the distribution of important participant factors between trials (age, type of malignancies) and trial factors (randomisation concealment, blinding of outcome assessment, losses to follow‐up, treatment regimens). We assessed statistical heterogeneity of RCTs by examining the I² statistic (Higgins 2002), a quantity that describes approximately the proportion of variation in point estimates that is due to heterogeneity rather than sampling error. If significant heterogeneity was present (i.e. I² ≥ 50% (Deeks 2011), we explored the trials to investigate possible explanations.
Sensitivity analysis
We planned to conduct sensitivity analyses to assess the impact of study quality, including the following:
all studies; and
only those studies with adequate allocation concealment.
We also planned to conduct sensitivity analyses to assess the impact of heterogeneity, by excluding those with outlying results.
Results
Description of studies
Results of the search
On August 25th 2009, we retrieved 17 articles from the Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library. We excluded 16 based on title or abstract, and we included one (Goldman 2001). The electronic search retrieved 68 articles from MEDLINE. We excluded 64 based on title or abstract, because of obvious irrelevance. We examined the full texts of the four remaining articles. Two were RCTs satisfying the inclusion criteria (Goldman 2001; Kikuchi 2009), of which Goldman 2001 had already been identified in the CENTRAL search. The other two were controlled clinical trials (CCTs) (Renyi 2007; Wossmann 2003). We included these two CCTs in the review because we only identified two RCTs. We also retrieved 100 articles from Embase. We excluded all but two articles after examining the title and abstract. The MEDLINE search also identified these two articles (Kikuchi 2009; Renyi 2007). We retrieved 40 articles from CINAHL, excluding 37 based on title or abstract. The remaining three articles were the same as those identified and included from CENTRAL and MEDLINE (Goldman 2001; Renyi 2007; Wossmann 2003). We identified and included one more CCT (Patte 2002) after checking the reference lists of the other included studies. We identified no additional completed or ongoing trials after checking internet sources and conference proceedings and contacting experts. There was no discrepancy in the independent selection of included studies among the two authors, and a third author was not necessary in this process.
With the updated electronic search strategy on 26 February 2013, we retrieved 16 articles from CENTRAL, 53 articles from MEDLINE, 18 articles from Embase, and no articles from CINAHL. We found five studies from checking references of included studies and two studies from the US National Institutes of Health Ongoing Trials Register. We identified no studies by scanning the conference proceedings and contacting experts. In summary, we found a total of 94 studies in the update search in February 2013. After we removed duplicates, we screened 81 articles for eligibility. We excluded 69 articles based on title or abstract. We obtained the full text of the remaining 12 articles. We included seven studies in this review, including the five studies included in the original search (Goldman 2001; Kikuchi 2009; Patte 2002; Rényi 2007; Wössmann 2003) and two additional studies in the update (Pui 1997; Sánchez Tatay 2010). Two studies were ongoing (see the Characteristics of ongoing studies tables) and did not have results available, and we excluded three studies (see the Characteristics of excluded studies tables). There was no discrepancy in the independent selection of included studies among the two authors, and a third author was not necessary in this process.
With the updated electronic search on 14 March 2016, we retrieved additional 24 records from CENTRAL, 15 records from MEDLINE, 11 records from Embase and six records from CINAHL. We found no additional relevant studies in searching the clinical trials databases and scanning the conference proceedings and contacting experts. In summary, we found an additional 56 records in the update search in March 2016. After we removed duplicates, we screened 43 records for eligibility and none of these records were included based on the title or abstract. Since there was no new relevant study, we did not check references in this update.
In summary (see Figure 1), we included seven studies (five identified in the original review and two in the update 2013 and none in the update 2016). Among the seven included studies, two were RCTs satisfying the inclusion criteria (Goldman 2001; Kikuchi 2009), and the other five were CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003).
1.

Study flow diagram of review update
Included studies
All seven included trials evaluated urate oxidase as a preventive measure for TLS. We identified no clinical trial investigating urate oxidase for treatment of TLS. We have given details of the included trials in the 'Characteristics of included studies' tables and summarise the details below.
Six included trials compared urate oxidase against allopurinol as the control treatment in parallel‐group designs (Goldman 2001; Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003). The remaining one included trial compared different doses of urate oxidase (Kikuchi 2009). Four trials used rasburicase (Goldman 2001; Kikuchi 2009; Rényi 2007; Sánchez Tatay 2010) while the remaining three trials used Uricozyme (Patte 2002; Pui 1997; Wössmann 2003). Four included trials (Goldman 2001; Patte 2002; Pui 1997; Rényi 2007) used a standard alkaline hyperhydration regimen in both the intervention and the control groups, while one trial (Wössmann 2003) used alkalinization only in the control group. The remaining two trials (Kikuchi 2009; Sánchez Tatay 2010) did not mention alkalinization. Six trials (Goldman 2001; Kikuchi 2009; Patte 2002; Pui 1997; Rényi 2007; Wössmann 2003) initiated urate oxidase before the start of chemotherapy, lasting for three to seven days. One trial (Sánchez Tatay 2010) did not mention the duration of therapy.
None of the five included non‐randomised, CCTs used a concurrent control group. Four trials used a historical control group (Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003), and one trial used aggregate participant data from trials of other study groups as a retrospective analysis (Patte 2002).
The RCT (Goldman 2001) comparing rasburicase with allopurinol included a total of 27 participants in the intervention group and 25 participants in the control group. This trial included children only (aged 0.3 to 17 years) (Goldman 2001). The RCT comparing different doses of rasburicase included a total of 15 participants in the low‐dose group (0.15 mg/kg) and 15 participants in the high‐dose group (0.2 mg/kg) (Kikuchi 2009). This trial also included children only (aged 0 to 17 years) (Kikuchi 2009). The five CCTs comparing urate oxidase with allopurinol included a total of 429 participants in the intervention groups and 563 participants in the control groups. All five CCTs included children only. Two of these trials reported a median age of 4.5 to 5.6 years in the intervention groups and 5.7 to 6 years in the control groups (Pui 1997; Rényi 2007). The remaining three trials (Patte 2002; Sánchez Tatay 2010; Wössmann 2003) did not mention the age distribution of the participants.
The types of malignancies included in all trials were similar. The RCT comparing rasburicase and allopurinol recruited participants with stage three or four non‐Hodgkin's lymphoma, acute lymphoblastic leukaemia (ALL) with high WBC counts, and participants with leukaemia or lymphoma with hyperuricaemia (Goldman 2001). The RCT comparing high‐dose and low‐dose rasburicase recruited participants with stage four non‐Hodgkin's lymphoma, stage three non‐Hodgkin's lymphoma with large lymph node or high LDH, and acute leukaemia with high WBC counts (Kikuchi 2009). All five CCTs included participants with haematological malignancies who were at high risk of TLS, with just minor differences in the inclusion criteria among these trials (for details, please refer to the Characteristics of included studies tables).
For outcome measures, only one study reported incidence of clinical TLS (Wössmann 2003). Five studies (Goldman 2001; Kikuchi 2009; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) reported on all‐cause mortality. Five studies (Goldman 2001; Kikuchi 2009; Patte 2002; Rényi 2007; Sánchez Tatay 2010) also reported on mortality due to TLS. All seven included studies reported frequency of renal failure requiring renal replacement therapy (Goldman 2001; Kikuchi 2009; Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003), two studies reported frequency of normalisation of serum uric acid (Goldman 2001; Kikuchi 2009), one study reported AUC of serum uric acid (Goldman 2001), and four studies reported serial uric acid levels (Kikuchi 2009; Pui 1997; Rényi 2007; Sánchez Tatay 2010). Five studies reported adverse events (Goldman 2001; Kikuchi 2009; Pui 1997; Rényi 2007; Sánchez Tatay 2010).
Excluded studies
We excluded three RCTs evaluating urate oxidase for prevention of TLS as they recruited adult participants only and did not include paediatric participants (Cortes 2010; Ishizawa 2009; Vadhan‐Raj 2012).
Risk of bias in included studies
In general, none of the included trials were of high methodological quality. The two RCTs were quite small, recruiting only 30 to 52 participants (Goldman 2001; Kikuchi 2009). We describe the risk of bias in the 'Characteristics of included studies' tables, Figure 2, and Figure 3.
2.

'Risk of bias' graph: review authors' judgements about each 'Risk of bias' item presented as percentages across all included studies
3.
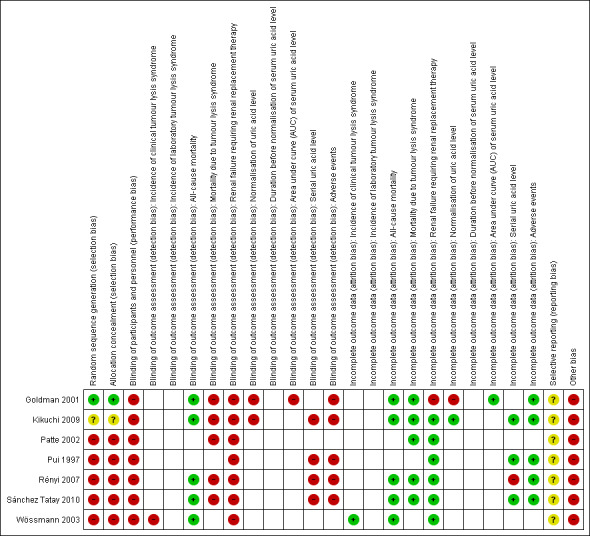
'Risk of bias' summary: review authors' judgements about each 'Risk of bias' item for each included study
Allocation
All five CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) included in this review were not randomised trials and selection bias was likely present. The RCT Kikuchi 2009 did not report random sequence generation or concealment. We were uncertain whether there was high risk of selection bias. Random sequence generation and randomisation concealment were likely to be adequate in Goldman 2001 as the randomisation code was computer‐generated.
Blinding
There was no blinding of participants or care providers in all five included CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003). Also, the two included RCTs (Goldman 2001; Kikuchi 2009) did not blind participants or care providers, which might have introduced performance bias.
We assessed detection biases for each outcome separately (except all‐cause mortality) and they are summarised in Figure 2 and Figure 3. There was no blinding of outcome assessors in all included RCTs and CCTs and most reported outcomes were considered to carry high risk of bias. Only the outcome on all‐cause mortality was considered to carry low risk of detection bias as assessment of death is unlikely to be mistaken or biased, even though outcome assessors knew which treatment participants were assigned.
Incomplete outcome data
We assessed attrition biases for each outcome separately and we have summarised them in Figure 2 and Figure 3. There were dropouts with incomplete data in both included RCTs (Goldman 2001; Kikuchi 2009). However, dropouts constituted a very low proportion of participants in one study (Kikuchi 2009) and were unlikely to cause significant attrition bias in the reported outcomes. Nevertheless, we considered the RCT by Goldman to have a high risk of bias in the outcomes of renal failure and normalisation of uric acid as there were differences in the dropout rate between the intervention and the control groups, and more than 10% of the participants in the control group had incomplete follow‐up (Goldman 2001). One CCT also had incomplete data for serum uric acid in a large proportion of participants and we considered it to have high risk of attrition bias (Rényi 2007). The remaining four CCTs had no dropouts and we considered them to have low risk of attrition biases in reported outcomes (Patte 2002; Pui 1997; Sánchez Tatay 2010; Wössmann 2003).
Selective reporting
It was unclear whether there was selective reporting of outcomes in all included studies as the trial protocols were not available (Goldman 2001; Kikuchi 2009; Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003).
Other potential sources of bias
In all studies, there was a high risk of other bias. The intervention and comparison groups were not comparable at baseline in four studies, which made them at high risk of bias (Goldman 2001; Kikuchi 2009; Rényi 2007; Wössmann 2003). Two studies (Patte 2002; Sánchez Tatay 2010) did not report some important baseline characteristics, so the comparability of their intervention and comparison groups was not certain. For non‐randomised controlled trials, failure of adjustment of potential confounders resulted in high risk of bias (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003). The use of historical controls in four trials (Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) may have biased the results in favour of the newer treatment because of improvement in supportive care. In one CCT (Patte 2002), chemotherapy treatments were different in different centres in different locations, and this may have caused bias.
Effects of interventions
Summary of findings for the main comparison. Summary of findings: urate oxidase compared with allopurinol.
| Urate oxidase compared with allopurinol for prevention of tumour lysis syndrome | ||||||
|
Patient or population: children with haematological malignancy at risk of tumour lysis syndrome Settings: hospital inpatient Intervention: urate oxidase Comparison: allopurinol | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Allopurinol | Urate oxidase | |||||
|
Incidence of clinical tumour lysis syndrome (follow‐up: unknown) |
161 per 1000 | 124 per 1000 (71 to 214) | RR 0.77 (0.44 to 1.33) | 348 (1) | ⊕⊕⊝⊝ lowa | |
|
Mortality due to tumour lysis syndrome (follow‐up: 4 to 12 days) |
51 per 1000b | 3 per 1000 (0 to 45) | RR 0.05 (0 to 0.89) | 396 (3) | ⊕⊕⊝⊝ lowa | |
|
Renal failure requiring renal replacement therapy (follow‐up: 4 to 13 days) |
92 per 1000 | 24 per 1000 (7 to 82) | RR 0.26 (0.08 to 0.89) | 992 (5) | ⊕⊝⊝⊝ very lowc | |
| Normalisation of serum uric acid | 0 per 1000 |
1000 per 1000d (confidence interval not estimable) |
Not applicable | 19 (1) |
⊕⊕⊝⊝ lowa | None of 9 participants in the control group and all 10 participants in the intervention group had normalisation of serum uric acid |
| Percentage reduction in serum uric acid level | Not reported | Not reported | Not applicable | Not applicable | Not applicable | None of the studies reported on this outcome measure |
|
AUC of serum uric acid level at 4 days (follow‐up: 2 weeks) |
The mean AUC of serum uric acid level in the control group was 329 mg/dLhr | The mean AUC of serum uric acid level in the intervention groups was 201 mg/dLhr lower (144 mg/dLhr to 258 mg/dLhr lower) | 52 (1) | ⊕⊕⊝⊝ lowa | ||
|
Frequency of adverse events (follow‐up: 4 to 13 days) |
0 per 1000 | 52 per 1000e (0 to 167) | RR 9.10 (1.29 to 64) | 345 (3) | ⊕⊕⊝⊝ lowa | None of participants in the control groups had adverse events |
| *The basis for the assumed risk is the median control group risk across studies unless otherwise specified. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). AUC: area under the curve; CI: Confidence interval; RR: Risk Ratio | ||||||
| GRADE Working Group grades of evidence High quality: we are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of effect, but there is a possibility that it is substantially different. Low quality: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect Very low quality: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect | ||||||
aEvidence from RCT downgraded by two levels because of high risk of bias in study design and imprecise result. bSince two of three studies had zero control group risks, we used mean instead of median control group risk across studies as the assumed risk. cEvidence from RCT downgraded by three levels because of high risk of bias in study design, imprecise result and high heterogeneity. dSince control group risk (assumed risk) was zero, corresponding risk estimation was based on risk in the intervention group in the included study. eSince control group risk (assumed risk) was zero, corresponding risk estimation was based on the median and the range of risks in the intervention groups in included studies.
Summary of findings 2. Summary of findings: high‐dose urate oxidase compared with low‐dose urate oxidase.
| High‐dose urate oxidase compared with low‐dose urate oxidase for prevention of tumour lysis syndrome | ||||||
|
Patient or population: children with haematological malignancy at risk of tumour lysis syndrome Settings: hospital inpatient Intervention: high‐dose urate oxidase Comparison: low‐dose urate oxidase | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Low‐dose urate oxidase | High‐dose urate oxidase | |||||
| Incidence of clinical tumour lysis syndrome | Not reported | Not reported | Not applicable | Not applicable | Not applicable | None of the studies reported on this outcome measure |
| Mortality due to tumour lysis syndrome | 0 per 1000 | 0 per 1000a | Not applicable | 30 (1) | ⊕⊕⊝⊝ lowb | None of participants died in both the intervention and the control groups |
| Renal failure requiring renal replacement therapy | 0 per 1000 | 0 per 1000a | Not applicable | 29 (1) | ⊕⊕⊝⊝ lowb | None of participants had renal failure in either the intervention or the control groups |
|
Normalisation of serum uric acid (follow‐up: 5 weeks) |
933 per 1000 | 998 per 1000 (830 to 1000) | RR 1.07 (0.89 to 1.28) | 29 (1) | ⊕⊕⊝⊝ lowb | |
|
Percentage reduction in serum uric acid level at 4 hours (follow‐up: 4 hours) |
The mean percentage reduction in serum uric acid level at 4 hours in the control group was 84.8%. | The mean percentage reduction in serum uric acid level at 4 hours in the intervention group was 8.1% higher (0.99% lower to 17.19% higher) | 29 (1) | ⊕⊕⊝⊝ lowb | ||
| AUC of serum uric acid level | Not reported | Not reported | Not applicable | Not applicable | Not applicable | None of the studies reported on this outcome measure |
|
Frequency of adverse events (follow‐up: 5 weeks) |
267 per 1000 | 144 per 1000 (32 to 662) | RR 0.54 (0.12 to 2.48) | 29 (1) | ⊕⊕⊝⊝ lowb | |
| *The basis for the assumed risk is the control group risk in the included study. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). AUC: area under the curve; CI: Confidence interval; RR: Risk Ratio | ||||||
| GRADE Working Group grades of evidence High quality: we are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of effect, but there is a possibility that it is substantially different. Low quality: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect Very low quality: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect | ||||||
aSince control group risk (assumed risk) is zero, corresponding risk estimation is based on risk in the intervention group in the included study. bEvidence from RCT downgraded by two levels because of high risk of bias in study design and imprecise result.
Urate oxidase versus allopurinol
Six included studies compared urate oxidase with allopurinol for prevention of TLS. One study was an RCT (Goldman 2001), and the other five studies were CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003).
Primary outcome
Incidence of clinical TLS
One CCT (Wössmann 2003) that reported this outcome did not find any significant difference between the group that received Uricozyme and the group that received allopurinol. (Sixteen out of 130 participants in the Uricozyme group versus 35 out of 218 participants in the allopurinol group developed TLS; risk ratio (RR) 0.77, 95% confidence intervals (CI) 0.44 to 1.33; P = 0.34; intention‐to‐treat (ITT) analysis; low quality of evidence; Analysis 1.1; Figure 4.) This study also reported results for the subgroup of participants with acute B lymphoblastic leukaemia (B‐ALL) and found no significant difference between the intervention and the control groups. (Five out of 53 participants in the Uricozyme group versus 16 out of 78 participants in the allopurinol group developed TLS; RR 0.46, 95% CI 0.18 to 1.18; P = 0.11; ITT analysis; Analysis 1.2).
1.1. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 1 Incidence of clinical tumour lysis syndrome.
4.

Forest plot of comparison: 1 Urate oxidase compared with allopurinol, outcome: 1.1 Incidence of clinical tumour lysis syndrome
1.2. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 2 Incidence of clinical tumour lysis syndrome in B‐ALL subgroup.
Secondary outcomes
Incidence of laboratory TLS
None of the included trials reported this outcome.
All‐cause mortality
The RCT (Goldman 2001) showed slightly lower mortality in the group that received rasburicase compared with the group that received allopurinol, but this was not statistically significant. (None of the 27 participants in the rasburicase group versus 2 out of 25 participants in the allopurinol group died; Fisher's exact test P = 0.23; ITT analysis). The two participants in the allopurinol group died from Pseudomonas sepsis and intracerebral haemorrhage, respectively. All‐cause mortality was available in three CCTs, two of which did not have any mortality (Rényi 2007; Sánchez Tatay 2010), and the third one reported four deaths in the control group (Wössmann 2003). The study reported the deaths to be treatment‐related, but did not specify the actual cause. The pooled result of the three CCTs showed no significant difference in all‐cause mortality between the intervention and the control groups. (None of the 158 participants in the urate oxidase group versus 4 out of 248 participants in the allopurinol group died; RR 0.19, 95% CI 0.01 to 3.42; P = 0.26; ITT analysis; Analysis 1.3).
1.3. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 3 All‐cause mortality.
Mortality due to TLS
The RCT (Goldman 2001) did not find any mortality due to TLS in either the intervention group (total of 27 participants) or control group (total of 25 participants) (ITT analysis). However, pooled results of three CCTs (Patte 2002; Rényi 2007; Sánchez Tatay 2010) showed a significantly lower mortality due to TLS in the group that received Uricozyme compared with the group that received allopurinol. (None of the 180 participants in the intervention group versus 11 out of 216 participants in the control group died due to TLS; RR 0.05, 95% CI 0.00 to 0.89; P = 0.04; ITT analysis; low quality of evidence; Analysis 1.4; Figure 5).
1.4. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 4 Mortality due to tumour lysis syndrome.
5.

Forest plot of comparison: 1 Urate oxidase compared with allopurinol, outcome: 1.4 Mortality due to tumour lysis syndrome
Renal failure requiring renal replacement therapy
The RCT (Goldman 2001) showed no significant difference in the frequency of renal failure between the intervention and the control groups. (None of the 26 participants in the rasburicase group versus 1 out of 22 participants in the allopurinol group had renal failure; Fisher's exact test P = 0.46; not ITT analysis.) In contrast, pooled results of five CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) showed significantly lower frequency of renal failure requiring renal replacement therapy in participants who received urate oxidase compared with those who received allopurinol. (Twelve out of 429 participants in the intervention group versus 65 out of 563 participants in the control group developed renal failure; I² = 62%; RR 0.26, 95% CI 0.08 to 0.89; P = 0.03; ITT analysis; very low quality of evidence; Analysis 1.5; Figure 6.) One CCT (Wössmann 2003) reported results of a subgroup of participants with B‐ALL and showed lower frequency of renal failure in the intervention group, but the difference was not statistically significant. (Two out of 53 participants in the intervention group versus 12 out of 78 participants in the control group developed renal failure; RR 0.25, 95% CI 0.06 to 1.05; P = 0.06; ITT analysis; Analysis 1.6).
1.5. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 5 Renal failure requiring renal replacement therapy.
6.

Forest plot of comparison: 1 Urate oxidase compared with allopurinol, outcome: 1.5 Renal failure requiring renal replacement therapy
1.6. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 6 Renal failure requiring renal replacement therapy in B‐ALL subgroup.
Normalisation of uric acid level
The RCT (Goldman 2001) showed significantly higher frequency of uric acid normalisation at four hours in the participants who received rasburicase compared with participants who received allopurinol. (Ten out of 10 participants in the intervention group versus zero out of nine participants in the control group had normalisation of uric acid level; Fisher's exact test P < 0.001; not ITT analysis; low quality of evidence).
Duration before normalisation of serum uric acid level
None of the included trials reported this outcome.
Area under the curve (AUC) of serum uric acid level
The RCT (Goldman 2001) reported a significantly lower AUC of serum uric acid at four days in the intervention group compared with the control group (mean AUC 128 mg/dLhr in 27 participants in the intervention group versus 329 mg/dLhr in 25 participants in the control group; mean difference (MD) ‐201.00 mg/dLhr, 95% CI ‐258.05 mg/dLhr to ‐143.95 mg/dLhr; P < 0.00001; ITT analysis; low quality of evidence; Analysis 1.7.) The RCT also reported results of different subgroups and found significantly lower AUC of serum uric acid at four days in the intervention group compared with the control group in participants with leukaemia (mean AUC 141 mg/dLhr in 20 participants in the intervention group versus 361 mg/dLhr in 19 participants in the control group; MD ‐220.00 mg/dLhr, 95% CI ‐286.67 mg/dLhr to ‐153.33 mg/dLhr; P < 0.00001; ITT analysis; Analysis 1.8), lymphoma (mean AUC 92 mg/dLhr in seven participants in the intervention group versus 224 mg/dLhr in six participants in the control group; MD ‐132.00 mg/dLhr, 95% CI ‐185.47 mg/dLhr to ‐78.53 mg/dLhr; P < 0.00001; ITT analysis; Analysis 1.9), baseline hyperuricaemia participants (mean AUC 162 mg/dLhr in 10 participants in the intervention group versus 440 mg/dLhr in nine participants in the control group; MD ‐278.00 mg/dLhr, 95% CI ‐373.69 mg/dLhr to ‐182.31 mg/dLhr; P < 0.00001; ITT analysis; Analysis 1.10), and participants with normal baseline uric acid (mean AUC 108 mg/dLhr in 17 participants in the intervention group versus 348 mg/dLhr in 16 participants in the control group; MD ‐240.00 mg/dLhr, 95% CI ‐340.95 mg/dLhr to ‐139.05 mg/dLhr, P < 0.00001; ITT analysis; Analysis 1.11).
1.7. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 7 AUC of serum uric acid level at 4 days.
1.8. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 8 AUC of serum uric acid level at 4 days in leukaemia participants.
1.9. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 9 AUC of serum uric acid level at 4 days in lymphoma participants.
1.10. Analysis.
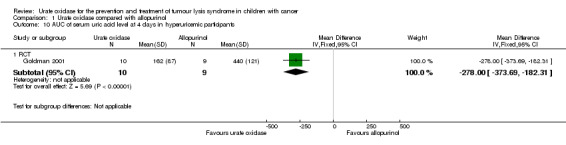
Comparison 1 Urate oxidase compared with allopurinol, Outcome 10 AUC of serum uric acid level at 4 days in hyperuricemic participants.
1.11. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 11 AUC of serum uric acid level at 4 days in normouricemic participants.
Serial uric acid level
Three CCTs reported serial uric acid levels (Pui 1997; Rényi 2007; Sánchez Tatay 2010). All three CCTs reported results on the first two days after urate oxidase, while two studies (Rényi 2007; Sánchez Tatay 2010) reported results up to four days and one study (Rényi 2007) reported results up to 12 days. The pooled results showed significantly lower uric acid level in the intervention group compared with the control group at two days (mean uric acid level 1.02 mg/dL in 147 participants the intervention group versus 3.25 mg/dL in 147 participants in the control group; I² = 87%; MD ‐3.80 mg/dL, 95% CI ‐7.37 mg/dL to ‐0.24 mg/dL; P = 0.04; not ITT analysis; < 70% of participants had outcomes available for Rényi 2007; results susceptible to bias; Analysis 1.12), three days (mean uric acid level 0.52 mg/dL in 28 participants in the intervention group versus 4.66 mg/dL in 19 participants in the control group; I² = 89%; MD ‐3.13 mg/dL, 95% CI ‐6.12 mg/dL to ‐0.14 mg/dL; P = 0.04; not ITT analysis; < 70% of participants had outcomes available for Rényi 2007; results susceptible to bias; Analysis 1.13), four days (mean uric acid level 0.24 mg/dL in 28 participants in the intervention group versus 4.41 mg/dL in 17 participants in the control group; MD ‐4.60 mg/dL, 95% CI ‐6.39 mg/dL to ‐2.81 mg/dL; P < 0.00001; not ITT analysis; < 70% of participants had outcomes available for Rényi 2007; results susceptible to bias; Analysis 1.14), and seven days (mean uric acid level 1.43 mg/dL in 12 participants in the intervention group versus 3.17 mg/dL in four participants in the control group; MD ‐1.74 mg/dL, 95% CI ‐3.01 mg/dL to ‐0.47 mg/dL; P = 0.007; not ITT analysis; < 70% of participants had outcomes available for Rényi 2007; results susceptible to bias; Analysis 1.15), but not significant at one day (mean uric acid level 1.37 mg/dL in 147 participants in the intervention group versus 3.94 mg/dL in 151 participants in the control group; I² = 94%; MD ‐3.00 mg/dL, 95% CI ‐7.61 mg/dL to 1.60 mg/dL; P = 0.2; not ITT analysis; < 70% of participants had outcomes available for Rényi 2007; results susceptible to bias; Analysis 1.16), five days (mean uric acid level 0.44 mg/dL in 12 participants in the intervention group versus 1.46 mg/dL in two participants in the control group; MD ‐1.02 mg/dL, 95% CI ‐2.24 mg/dL to 0.20 mg/dL; P = 0.1; not ITT analysis; < 70% of participants had outcomes available for Rényi 2007; results susceptible to bias; Analysis 1.17), and 12 days (mean uric acid level 2.34 mg/dL in 12 participants in the intervention group versus 3.14 mg/dL in eight participants in the control group; MD ‐0.80 mg/dL, 95% CI ‐2.51 mg/dL to 0.91 mg/dL; P = 0.36; not ITT analysis; Analysis 1.18).
1.12. Analysis.
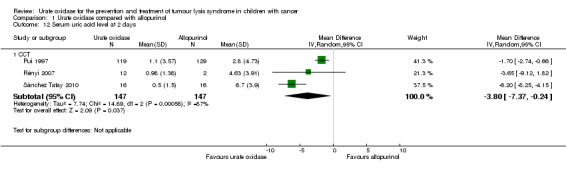
Comparison 1 Urate oxidase compared with allopurinol, Outcome 12 Serum uric acid level at 2 days.
1.13. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 13 Serum uric acid level at 3 days.
1.14. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 14 Serum uric acid level at 4 days.
1.15. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 15 Serum uric acid level at 7 days.
1.16. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 16 Serum uric acid level at 1 day.
1.17. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 17 Serum uric acid level at 5 days.
1.18. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 18 Serum uric acid level at 12 days.
Adverse events
The RCT (Goldman 2001) and three CCTs (Pui 1997; Rényi 2007; Sánchez Tatay 2010) reported frequency of adverse effects in the intervention and control groups. The RCT showed no significant differences between the intervention and the control groups. (One out of 27 participants in the intervention group versus none of the 25 participants in the control group had an adverse event; Fisher's exact test P = 1.0; ITT analysis.) The adverse event reported was haemolysis. The pooled results from CCTs showed significantly higher frequency of adverse effects in participants who received urate oxidase. (Thirteen out of 186 participants in the intervention group versus none of the 159 participants in the control group had adverse events; RR 9.10, 95% CI 1.29 to 64.00; P = 0.03; I² = 0%; ITT analysis; low quality of evidence; Analysis 1.19.) Adverse events reported in the intervention group included allergic reaction (six participants), methaemoglobinaemia (one participant), fever (two participants), nausea (one participant), abdominal pain (one participant), and mucositis (two participants). It should be noted that the Pui 1997 and Rényi 2007 studies included additional participants in this analysis (see the Characteristics of included studies tables for more information).
1.19. Analysis.

Comparison 1 Urate oxidase compared with allopurinol, Outcome 19 Frequency of adverse events.
High‐dose urate oxidase versus low‐dose urate oxidase
One RCT (Kikuchi 2009) compared urate oxidase (rasburicase) given in high dose (0.2 mg/kg/day for five days) versus low dose (0.15 mg/kg/day for five days).
Primary outcome
Incidence of clinical TLS
The RCT did not report this outcome.
Secondary outcomes
Incidence of laboratory TLS
The RCT did not report this outcome.
All‐cause mortality
The RCT (Kikuchi 2009) reported no significant difference in all‐cause mortality between the high‐dose and the low‐dose groups. (None of the 15 participants in the high‐dose group versus one of the 15 participants in the low‐dose group died; Fisher's exact test P = 1.0; ITT analysis.) The death in the low‐dose group was due to cerebral haemorrhage, brain oedema, and brain herniation.
Mortality due to TLS
The RCT (Kikuchi 2009) reported no mortality due to TLS in both the high‐dose group (15 participants) and the low‐dose group (15 participants) (ITT analysis; low quality of evidence).
Renal failure requiring renal replacement therapy
The RCT (Kikuchi 2009) reported no renal failure due to TLS in both the high‐dose group (14 participants) and the low‐dose group (15 participants) (not ITT analysis; low quality of evidence).
Normalisation of uric acid level
The RCT (Kikuchi 2009) showed no significant difference between the high‐dose and the low‐dose groups. (All participants (14) in the high‐dose group versus 14 out of 15 participants in the low‐dose group had normalisation of uric acid level; RR 1.07, 95% CI 0.89 to 1.28; P = 0.49; not ITT analysis; low quality of evidence; Analysis 2.1).
2.1. Analysis.

Comparison 2 High‐dose urate oxidase compared with low‐dose urate oxidase, Outcome 1 Normalisation of serum uric acid.
Duration before normalisation of serum uric acid level
The included RCT did not report this outcome.
AUC of serum uric acid level
The included RCT did not report this outcome.
Serial uric acid level
The RCT (Kikuchi 2009) reported the percentage reduction of uric acid level at four hours and did not find any significant difference between the high‐dose and the low‐dose groups (mean percentage reduction in uric acid level 92.9% in 14 participants in the high‐dose group versus 84.8% in 15 participants in the low‐dose group; MD 8.10%, 95% CI ‐0.99% to 17.19%; P = 0.08; not ITT analysis; low quality of evidence; Analysis 2.2).
2.2. Analysis.

Comparison 2 High‐dose urate oxidase compared with low‐dose urate oxidase, Outcome 2 Percentage reduction in serum uric acid level at 4 hours.
Adverse events
The RCT (Kikuchi 2009) did not show any significant difference in the frequency of adverse events between the high‐dose group (two out of 14 participants) and the low‐dose groups (four out of 15 participants) (RR 0.54, 95% CI 0.12 to 2.48; P = 0.42; not ITT analysis; low quality of evidence; Analysis 2.3). Adverse events included allergic reaction (three participants), haemolysis (one participant), and anaemia (two participants).
2.3. Analysis.

Comparison 2 High‐dose urate oxidase compared with low‐dose urate oxidase, Outcome 3 Frequency of adverse events.
Sensitivity analysis and cost‐benefit analysis
We planned to do sensitivity analysis for heterogeneous results by excluding outlying results. We could not identify any obvious outliers, and therefore did not perform this sensitivity analysis. Since there was only one RCT with adequate allocation concealment, which we did not include in a pooled analysis, we did not perform sensitivity analysis for this. Since we identified no high‐quality data on effectiveness, we did not perform a cost‐benefit analysis.
Discussion
Summary of main results
This is the second update of the original systematic review. No new studies were included and conclusions did not change. Although numerous uncontrolled studies have found that urate oxidase can lower serum uric acid levels quickly and sometimes dramatically, we found little evidence from randomised controlled trials (RCTs) or controlled clinical trials (CCTs) supporting its effectiveness in preventing or treating tumour lysis syndrome (TLS) in children with cancer. Only two RCTs (Goldman 2001; Kikuchi 2009) and five CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) were available on the prophylaxis of tumour lysis syndrome. There is currently no trial evaluating urate oxidase for treatment of established TLS.
Urate oxidase versus allopurinol
Six included studies compared urate oxidase with allopurinol for the prevention of tumour lysis syndrome. One study was an RCT (Goldman 2001), and the other five studies were CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003). One CCT (Wössmann 2003) reported the incidence of clinical TLS and did not find significant difference between the group that received Uricozyme and the group that received allopurinol. The RCT (Goldman 2001) showed no significant difference in all‐cause mortality between the group that received rasburicase and the group that received allopurinol. The pooled result of the three CCTs (Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) also showed no significant difference in all‐cause mortality between the intervention and the control groups. The RCT (Goldman 2001) did not find any mortality due to TLS in either the intervention group or the control group. However, the pooled result of the three CCTs (Patte 2002; Rényi 2007; Sánchez Tatay 2010) showed a significantly lower mortality due to TLS in the group that received Uricozyme compared with the group that received allopurinol. The RCT (Goldman 2001) showed no significant difference in the frequency of renal failure between the intervention and the control groups. In contrast, pooled results of five CCTs (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) showed significantly lower frequency of renal failure requiring renal replacement therapy in participants who received urate oxidase compared with those who received allopurinol. However, heterogeneity was present in this analysis.
The RCT (Goldman 2001) showed significantly higher frequency of uric acid normalisation at four hours in the participants who received rasburicase compared with participants who received allopurinol. All included trials did not report the duration before normalisation of serum uric acid level. The RCT (Goldman 2001) reported a significantly lower AUC of serum uric acid at four days in the intervention group compared with the control group. Three CCTs reported serial uric acid levels (Pui 1997; Rényi 2007; Sánchez Tatay 2010). All three CCTs reported results on the first two days after urate oxidase, while two studies (Rényi 2007; Sánchez Tatay 2010) reported results up to four days, and one study (Rényi 2007) reported results up to 12 days. The pooled results showed significantly lower uric acid levels in the intervention group compared with the control group at two days, three days, four days, and seven days, but the differences were not significant at one day, five days, and 12 days. Heterogeneity was present in some of these analyses. The RCT (Goldman 2001) and three CCTs (Pui 1997; Rényi 2007; Sánchez Tatay 2010) reported frequency of adverse effects in the intervention and the control groups. The RCT showed no significant differences between the intervention and the control groups. The adverse event reported was haemolysis. The pooled results from the three CCTs showed significantly higher frequency of adverse effects in participants who received urate oxidase. Adverse events reported in the intervention group included allergic reaction (six participants), methaemoglobinaemia (one participant), fever (two participants), nausea (one participant), abdominal pain (one participant), and mucositis (two participants).
High‐dose versus low‐dose urate oxidase
We included one RCT that compared high‐dose versus low‐dose rasburicase (Kikuchi 2009). The trial did not report the incidence of TLS. The trial reported no significant difference in all‐cause mortality between the high‐dose and the low‐dose groups. There was no mortality due to TLS in both the high‐dose and the low‐dose groups. There was no renal failure due to TLS in both groups. There was no significant difference in normalisation of uric acid level between the high‐dose and the low‐dose groups. The included RCT did not report the duration before normalisation of serum uric acid level or the AUC of serum uric acid level. The trial reported the percentage reduction of uric acid level at four hours and did not find significant difference between the two groups. There was no significant difference in the frequency of adverse events between the two groups. Adverse events included allergic reaction (three participants), haemolysis (one participant), and anaemia (two participants).
Overall completeness and applicability of evidence
Although urate oxidase is widely used in children with cancer for prevention of TLS, high‐quality evidence of its efficacy is limited. The only RCT comparing urate oxidase with allopurinol (Goldman 2001) did not report the important outcome of clinical TLS. Although participants who received rasburicase had significantly lower exposure to uric acid (lower AUC and higher chance of uric acid normalisation) compared with participants who received allopurinol in the RCT (Goldman 2001), we were not entirely certain whether this translated into significant clinical benefits. Although hyperuricaemia is related to TLS, this trial failed to show any significant difference between the treatment and the control groups in all‐cause mortality or mortality related to TLS or renal failure requiring renal replacement therapy. Because of the paucity of evidence from RCTs, we also included CCTs in the current systematic review. However, all five CCTs identified were of unsatisfactory methodological quality. Although mortality due to TLS and incidence of renal failure were found to be significantly lower in participants who received urate oxidase, the conclusion from CCTs has to be treated with caution in view of high risk of biases.
On the other hand, due to inadequate sample size in the existing trials, the absence of significant clinical benefits of urate oxidase may be a false negative result. Therefore, we cannot ignore the potential benefits of urate oxidase in children with malignancy based on the current available evidence, especially in view of its probable effectiveness in reducing serum uric acid, which is an important surrogate outcome. Further trials of larger sample size are needed to clarify the role of urate oxidase. Assuming a mortality rate of 0.9% (Annemans 2003a) and that urate oxidase is effective in reducing mortality by half, the number of people with cancer needed to treat to prevent one death is 223, and the sample size required to achieve a power of 80% in detecting a reduction in mortality at a 5% level of significance is estimated to be 856 people.
There was only one RCT comparing different doses of rasburicase (Kikuchi 2009). The results were consistent with previous uncontrolled studies and CCTs; comparing rasburicase at 0.15 mg/kg and 0.2 mg/kg showed a dramatic reduction in serum uric acid level in both arms. However, the RCT did not report the important outcome of incidence of clinical TLS, and this small trial was not adequately powered to address the other clinically important outcomes of mortality or renal failure. Although there was no significant difference in any of the outcomes between the two groups, we are not certain whether a higher and lower dose of rasburicase are really equivalent because of the small sample size. Likewise, there is uncertainly about whether the higher dose is associated with more adverse effects.
Although not eligible for inclusion in this review, there is a study that has addressed the cost‐effectiveness of rasburicase (Annemans 2003b). This study concluded that rasburicase was cost‐effective for prevention of TLS in children, and rasburicase for the treatment of TLS in children was cost‐saving. However, this conclusion was based on the assumption that rasburicase is 60% to 100% effective in the prevention of TLS, which was not in fact based on high‐quality trial evidence. As the effectiveness of urate oxidase in the prevention or treatment of TLS has yet to be established, its cost‐effectiveness remains uncertain.
Quality of the evidence
Apart from limitations in the number of RCTs reporting clinically relevant outcomes and inadequate power to evaluate these outcomes, the trials included in the current review had a number of methodological flaws and were prone to bias. We considered none of the included studies to have low risk of bias in all aspects assessed. In both RCTs, the treatment and the control groups were not comparable at baseline, which cast doubt on the success of randomisation and increased the probability of confounding. One of the RCTs included did not report the random sequence generation or allocation concealment, which are important to minimise selection bias (Kikuchi 2009). The other RCT had more dropouts in the control arm than in the treatment arm, which might have caused attrition bias (Goldman 2001). None of the included RCTs attempted to blind the participants, physicians, or outcome assessors, which might have introduced performance and detection biases. Trial protocols were not available, and it was uncertain whether there was reporting bias in the RCTs.
In addition, all five CCTs included were also of unsatisfactory methodological quality (Patte 2002; Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003). There was a high risk of selection bias as participants were not randomly allocated to treatment groups. Performance and detection biases were also likely as there was no blinding. Reporting bias was uncertain as trial protocols were not available. There was a high risk of attrition bas in one CCT, which had a lot of missing data (Rényi 2007). The results from the four historical controlled trials (Pui 1997; Rényi 2007; Sánchez Tatay 2010; Wössmann 2003) were prone to bias from the advancement of supportive care with time. The remaining CCT (Patte 2002) was actually a retrospective review of data from trials of chemotherapy protocols comparing different treatments from different study groups at different locations; therefore, it suffered from bias due to different practices in different centres. The intervention and comparison groups were not comparable at baseline in two studies, which might result in high risk of bias (Rényi 2007; Wössmann 2003). Two studies (Patte 2002; Sánchez Tatay 2010) did not report some important baseline characteristics, so comparability of the intervention and comparison groups was uncertain. None of the included CCTs took into consideration and adjusted for potential confounding factors in their analyses; hence, their results were susceptible to confounding by known and unknown factors.
In conclusion, the quality of evidence for the use of urate oxidase for prevention of TLS in children with cancer ranks from very low to low, because of high risk of bias in existing trials and imprecise results.
Potential biases in the review process
We focused our search on major English language electronic databases; therefore, non‐English literature might be under‐represented and missed in the review. Because the search was focused on RCTs and CCTs, we could have missed some further historical controlled trials. Publication bias was also possible.
Agreements and disagreements with other studies or reviews
As far as we know, this is the only systematic review evaluating the effectiveness of urate oxidase for prevention and treatment of TLS in children with cancer. There was a review on TLS with targeted therapy and the role of rasburicase (Bose 2011). The authors performed a search on MEDLINE in February 2011 and included RCTs, CCTs, and single‐arm studies of rasburicase in both children and adults. That review had a similar conclusion to the current review, that although there was a wealth of evidence suggesting that rasburicase is effective in correcting hyperuricaemia, prospective trials showing that it improves hard outcomes, such as acute renal failure, need for dialysis, and mortality, are lacking. More randomised controlled trials evaluating clinically relevant outcomes are needed.
Authors' conclusions
Implications for practice.
Thus far, the paucity of high‐quality studies precludes firm recommendations. Although there is some low‐quality evidence that urate oxidase might be more effective than allopurinol in reducing the frequency of hyperuricaemia and the exposure to high serum uric acid, it is still uncertain whether the routine use of urate oxidase is effective for the prevention or treatment of tumour lysis syndrome (TLS), or a reduction in mortality or renal failure associated with TLS in children with cancer. The potential benefit of urate oxidase might be its effectiveness in reducing serum uric acid, which is an important surrogate outcome. It is unclear which type of urate oxidase (rasburicase or Uricozyme) is superior in terms of efficacy and what dosage regimen or treatment duration is optimal. On the other hand, urate oxidase may be associated with potential adverse effects, such as haemolysis or hypersensitivity. Clinicians who wish to use urate oxidase should weigh the potential benefits of reducing serum uric acid levels and the uncertain benefits in preventing renal failure or mortality from TLS against the potential risk of adverse effects.
Implications for research.
There is a paucity of evidence from randomised controlled trials (RCT) assessing urate oxidase for the prevention and treatment of TLS in children with cancer. The existing trials are of small size and low methodological quality. Further high‐quality RCTs of larger sample size are needed to assess the effectiveness of urate oxidase in children, especially high‐risk patients who are more likely to benefit. High‐risk patients can include those with high tumour burden or turn‐over (such as high initial white blood cell counts for leukaemia, or Burkitt lymphoma, high‐stage lymphoma, or lymphoma with bulky disease), and people with baseline hyperuricaemia, renal impairment, hypocalcaemia, or hypophosphataemia. Trials should assess patient‐orientated outcomes, such as incidence of clinical TLS, mortality, or frequency of renal failure. Although blinding of participants and clinicians for comparison of intravenous urate oxidase and oral allopurinol is difficult, it can be attempted with the use of a double placebo, to minimise performance biases. The effectiveness and safety of different forms of urate oxidase in different dosage regimens should also be investigated further.
What's new
| Date | Event | Description |
|---|---|---|
| 14 March 2016 | New search has been performed | The search for eligible studies was updated to March 2016. |
| 14 March 2016 | New citation required but conclusions have not changed | Unfortunately, no new studies could be included in the review. As a result the conclusions have not changed. In this version of the review, we have used the most recent 'Risk of bias' criteria as recommended by Cochrane Childhood Cancer and we have included 'Summary of findings' tables and performed a GRADE assessment. |
History
Protocol first published: Issue 1, 2008 Review first published: Issue 6, 2010
| Date | Event | Description |
|---|---|---|
| 1 August 2013 | New citation required and conclusions have changed | Two new studies were found and included in the update. Conclusions regarding efficacy outcomes did not change, whereas for adverse effects they did. |
| 26 February 2013 | New search has been performed | The search for eligible studies was updated to February 2013. |
| 17 June 2008 | Amended | Converted to new review format. |
Acknowledgements
We are grateful to Dr Edit Bardi for providing us with missing data on the trial her group has published (Rényi 2007). We would like to thank the Editorial Base of Cochrane Childhood Cancer for their advice and support. The Editorial Base of Cochrane Childhood Cancer is funded by ‘Stichting Kinderen Kankervrij’ (KiKa), the Netherlands.
Appendices
Appendix 1. Search strategy for Cochrane Central Register of Controlled Trials (CENTRAL)
The following text words were used:
urate oxidase OR uricase OR uricas* rasburicase OR rasburicas* OR elitek OR fasturtec OR fasturt* OR uox
The search was performed in title, abstract or keywords
[*=zero or more characters]
Appendix 2. Search strategy for PubMed
1. For Urate oxidase the following MeSH headings and text words were used:
urate oxidase OR uricase OR uricas* OR oxidase, urate OR EC 1.7.3.3. OR rasburicase OR rasburicas* OR elitek OR fasturtec OR fasturt* OR uox
2. For Children the following MeSH headings and text words were used:
infant OR infan* OR newborn OR newborn* OR new‐born* OR baby OR baby* OR babies OR neonat* OR child OR child* OR schoolchild* OR schoolchild OR school child OR school child* OR kid OR kids OR toddler* OR adolescent OR adoles* OR teen* OR boy* OR girl* OR minors OR minors* OR underag* OR under ag* OR juvenil* OR youth* OR kindergar* OR puberty OR puber* OR pubescen* OR prepubescen* OR prepuberty* OR pediatrics OR pediatric* OR paediatric* OR peadiatric* OR schools OR nursery school* OR preschool* OR pre school* OR primary school* OR secondary school* OR elementary school* OR elementary school OR high school* OR highschool* OR school age OR schoolage OR school age* OR schoolage* OR infancy OR schools, nursery OR infant, newborn
3. For Cochrane RCTs/CCTs the following MeSH headings and text words were used in the original version of the review:
(randomized controlled trial [pt] OR controlled clinical trial [pt] OR randomized controlled trials [mh] OR random allocation [mh] OR double‐blind method [mh] OR single‐blind method [mh] OR clinical trial [pt] OR clinical trials [mh] OR ("clinical trial" [tw]) OR ((singl* [tw] OR doubl* [tw] OR trebl* [tw] OR tripl* [tw]) AND (mask* [tw] OR blind* [tw])) OR (placebos [mh] OR placebo* [tw] OR random* [tw] OR research design [mh:NoExp] OR comparative study [mh] OR evaluation studies [mh] OR follow‐up studies [mh] OR prospective studies [mh] OR control* [tw] OR prospectiv* [tw] OR volunteer* [tw]) NOT (animals [mh] NOT humans [mh]))
For the update in 2013 the following search strategy for identifying RCTs and CCTs was used:
((randomized controlled trial[pt]) OR (controlled clinical trial[pt]) OR (randomized[tiab]) OR (placebo[tiab]) OR (drug therapy[sh]) OR (randomly[tiab]) OR (trial[tiab]) OR (groups[tiab])) AND (humans[mh])
Final search: 1 AND 2 AND 3
[CCT = controlled clinical trial; RCT = randomized controlled trial; mh = Medical Subject Heading (MeSH) term; pt =publication type; tw: text word]
Appendix 3. Search strategy for Embase (OVID)
1. For Urate oxidase the following Emtree terms and text words were used:
1. urate oxidase.mp. or Urate Oxidase/ 2. (rasburicase or uox).mp. 3. (uricase or elitek).mp. 4. (9002‐12‐4 or 352311‐12‐7).rn. or EC 1733.mp. 5. (uricas$ or rasburicas$ or fasturtec or fasturt$).mp. 6. or/1‐5
2. For Children the following Emtree terms and text words were used:
1. infant/ or infancy/ or newborn/ or baby/ or child/ or preschool child/ or school child/ 2. adolescent/ or juvenile/ or boy/ or girl/ or puberty/ or prepuberty/ or pediatrics/ 3. primary school/ or high school/ or kindergarten/ or nursery school/ or school/ 4. or/1‐3 5. (infant$ or (newborn$ or new born$) or (baby or baby$ or babies) or neonate$).mp. 6. (child$ or (school child$ or schoolchild$) or (school age$ or schoolage$) or (pre school$ or preschool$)).mp. 7. (kid or kids or toddler$ or adoles$ or teen$ or boy$ or girl$).mp. 8. (minors$ or (under ag$ or underage$) or juvenil$ or youth$).mp. 9. (puber$ or pubescen$ or prepubescen$ or prepubert$).mp. 10. (pediatric$ or paediatric$ or peadiatric$).mp. 11. (school or schools or (high school$ or highschool$) or primary school$ or nursery school$ or elementary school or secondary school$ or kindergar$).mp. 12. or/5‐11 13. 4 or 12
3. For RCTs/CCTs the following Emtree terms and text words were used in the original version of the review:
1. Clinical Trial/ 2. Controlled Study/ 3. Randomized Controlled Trial/ 4. Double Blind Procedure/ 5. Single Blind Procedure/ 6. Comparative Study/ 7. RANDOMIZATION/ 8. Prospective Study/ 9. PLACEBO/ 10. Phase 2 Clinical Trial/ 11. phase 3 clinical study.mp. 12. phase 4 clinical study.mp. 13. Phase 3 Clinical Trial/ 14. Phase 4 Clinical Trial/ 15. or/1‐14 16. allocat$.mp. 17. blind$.mp. 18. control$.mp. 19. placebo$.mp. 20. prospectiv$.mp. 21. random$.mp. 22. ((singl$ or doubl$ or trebl$ or tripl$) and (blind$ or mask$)).mp. 23. (versus or vs).mp. 24. (randomized controlled trial$ or randomised controlled trial$).mp. 25. controlled clinical trial$.mp. 26. clinical trial$.mp. 27. or/16‐27 28. Human/ 29. Nonhuman/ 30. ANIMAL/ 31. Animal Experiment/ 32. or/29‐31 33. 32 not 28 34. (15 or 27) not 33
For the update in 2013 the following search strategy for identifying RCTs and CCTs was used:
1. Randomized Controlled Trial/ 2. Controlled Clinical Trial/ 3. randomized.ti,ab. 4. placebo.ti,ab. 5. randomly.ti,ab. 6. trial.ti,ab. 7. groups.ti,ab. 8. drug therapy.sh. 9. or/1‐8 10. Human/ 11. 9 and 10
Final search: 1 and 2 and 3 (Urate oxidase AND Children AND RCT)
[mp = title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name; sh = subject heading; ti,ab = title, abstract; / = Emtree term; $ = zero or more characters; rn = registry number; RCT = randomized controlled trial; CCT = controlled clinical trial]
Appendix 4. Search strategy for CINAHL
1. For Urate oxidase the following text words were used:
urate oxidase OR rasburicase OR uox OR uricase OR elitek OR fasfurtec
2. For Children the following text words were used:
neonate OR infant OR newborn OR baby OR child OR preschool OR school OR adolescent OR juvenile OR boy OR girl OR puberty OR pediatric OR kindergarten OR nursery OR kid OR minors
3. For RCTs/CCTs the following text words were used:
trial OR control OR placebo OR random OR prospective study OR comparative study
In the final search the three searches were combined: 1 AND 2 AND 3
Appendix 5. Search strategy for internet sources
(urate oxidase OR rasburicase OR uricase OR elitek OR fasfurtec) AND child AND tumour lysis
Appendix 6. Search strategy for conference proceedings
(urate oxidase OR rasburicase OR uricase OR elitek OR fasfurtec) AND child AND tumour lysis AND trial
Data and analyses
Comparison 1. Urate oxidase compared with allopurinol.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Incidence of clinical tumour lysis syndrome | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 CCT | 1 | 348 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.44, 1.33] |
| 2 Incidence of clinical tumour lysis syndrome in B‐ALL subgroup | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 CCT | 1 | 131 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.18, 1.18] |
| 3 All‐cause mortality | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 3.1 CCT | 3 | 406 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.19 [0.01, 3.42] |
| 4 Mortality due to tumour lysis syndrome | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.1 CCT | 3 | 396 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.05 [0.00, 0.89] |
| 5 Renal failure requiring renal replacement therapy | 5 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 5.1 CCT | 5 | 992 | Risk Ratio (M‐H, Random, 95% CI) | 0.26 [0.08, 0.89] |
| 6 Renal failure requiring renal replacement therapy in B‐ALL subgroup | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 6.1 CCT | 1 | 131 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.25 [0.06, 1.05] |
| 7 AUC of serum uric acid level at 4 days | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 RCT | 1 | 52 | Mean Difference (IV, Fixed, 95% CI) | ‐201.0 [‐258.05, ‐143.95] |
| 8 AUC of serum uric acid level at 4 days in leukaemia participants | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 8.1 RCT | 1 | 39 | Mean Difference (IV, Fixed, 95% CI) | ‐220.0 [‐286.67, ‐153.33] |
| 9 AUC of serum uric acid level at 4 days in lymphoma participants | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 9.1 RCT | 1 | 13 | Mean Difference (IV, Fixed, 95% CI) | ‐132.0 [‐185.47, ‐78.53] |
| 10 AUC of serum uric acid level at 4 days in hyperuricemic participants | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 10.1 RCT | 1 | 19 | Mean Difference (IV, Fixed, 95% CI) | ‐278.0 [‐373.69, ‐182.31] |
| 11 AUC of serum uric acid level at 4 days in normouricemic participants | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 11.1 RCT | 1 | 33 | Mean Difference (IV, Fixed, 95% CI) | ‐240.0 [‐340.95, ‐139.05] |
| 12 Serum uric acid level at 2 days | 3 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 12.1 CCT | 3 | 294 | Mean Difference (IV, Random, 95% CI) | ‐3.80 [‐7.37, ‐0.24] |
| 13 Serum uric acid level at 3 days | 2 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 13.1 CCT | 2 | 47 | Mean Difference (IV, Random, 95% CI) | ‐3.13 [‐6.12, ‐0.14] |
| 14 Serum uric acid level at 4 days | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 14.1 CCT | 2 | 45 | Mean Difference (IV, Fixed, 95% CI) | ‐4.6 [‐6.39, ‐2.81] |
| 15 Serum uric acid level at 7 days | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 15.1 CCT | 1 | 16 | Mean Difference (IV, Fixed, 95% CI) | ‐1.74 [‐3.01, ‐0.47] |
| 16 Serum uric acid level at 1 day | 3 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 16.1 CCT | 3 | 298 | Mean Difference (IV, Random, 95% CI) | ‐3.00 [‐7.61, 1.60] |
| 17 Serum uric acid level at 5 days | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 17.1 CCT | 1 | 14 | Mean Difference (IV, Fixed, 95% CI) | ‐1.02 [‐2.24, 0.20] |
| 18 Serum uric acid level at 12 days | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 18.1 CCT | 1 | 20 | Mean Difference (IV, Fixed, 95% CI) | ‐0.80 [‐2.51, 0.91] |
| 19 Frequency of adverse events | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 19.1 CCT | 3 | 345 | Risk Ratio (M‐H, Fixed, 95% CI) | 9.10 [1.29, 64.00] |
Comparison 2. High‐dose urate oxidase compared with low‐dose urate oxidase.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Normalisation of serum uric acid | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 RCT | 1 | 29 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.07 [0.89, 1.28] |
| 2 Percentage reduction in serum uric acid level at 4 hours | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 RCT | 1 | 29 | Mean Difference (IV, Fixed, 95% CI) | 8.10 [‐0.99, 17.19] |
| 3 Frequency of adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 3.1 RCT | 1 | 29 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.54 [0.12, 2.48] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Goldman 2001.
| Methods | Design: RCT Randomisation method: stratified randomisation, computer‐generated randomisation code Stratification factor: according to uric acid level (< 8 mg/dL or ≥ 8 mg/dL) | |
| Participants | Inclusion criteria: paediatric participants < 18 years with leukaemia and lymphoma deemed to have a high risk of tumour lysis syndrome: Murphy stage III or IV non‐Hodgkin's lymphoma, ALL with WBC ≥ 25,000/uL, childhood lymphoma or leukaemia with uric acid level of ≥ 8 mg/dL. Participants must have received chemotherapeutic agents not investigational in nature, with minimum life expectancy of 4 weeks, and ECOG performance scale ≤ 3 or Karnofsky scale ≥ 30% Exclusion criteria: participants previously treated with rasburicase or Uricozyme, treatment with allopurinol within 7 days, significant history of documented asthma, atopy, or G6PD deficiency Number of participants (intervention/comparison): 27/25 Number of boys (intervention/comparison): 16/18 Age (intervention/comparison): (mean) 7.1 (range = 0.3 to 17)/7.8 (range = 0.5 to 16) years Underlying haematological malignancies (intervention/comparison): leukaemia 20/9; lymphoma 7/6 Baseline creatinine (intervention/comparison): (mean) 0.6 (SD 0.33)/0.61 (SD 0.3) mg/dL Baseline uric acid (intervention/comparison): (mean) 7.7 (SD 3.5)/6.8 (SD 3.4) mg/dL Baseline potassium (intervention/comparison): (mean) 4.18 (SD 0.71)/3.85 (SD 0.52) mg/dL Baseline phosphate (intervention/comparison): (mean) 4.62 (SD 1.39)/4.15 (SD 1.11) mg/dL Baseline calcium (intervention/comparison): (mean) 8.92 (SD 0.74)/8.67 (SD 0.7) mg/dL Baseline LDH (intervention/comparison): (mean) 1599 (SD 1022)/1393 (SD 1438) U/L Baseline WBC (intervention/comparison): (mean) 83.2 (SD 81)/91 (SD 115) x 109/L |
|
| Interventions | Intervention (type of urate oxidase): rasburicase Comparison (type of control): allopurinol Treatment regime in intervention group: rasburicase 0.2 mg/kg ivi over 30 min daily for 5‐7 days Treatment regime in comparison group: allopurinol 300 mg/m²/d or 10 mg/kg/d divided every 8 h for 5‐7 days Cointerventions: hydration 3 L/m²/d, iv sodium bicarbonate at investigator's discretion | |
| Outcomes |
|
|
| Notes | Duration of follow up: 2 weeks
Number of dropouts (intervention/comparison): 1 (haemolysis)/3 (2 died; 1 did not start chemotherapy) Potential confounders were not described or adjusted The 2 groups may not be comparable at baseline because serum uric acid level was higher in the treatment group Whether outcomes were clearly defined: yes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "...treatment (rasburicase or allopurinol) was randomly allocated to patients according to a computer‐generated randomization code schema" |
| Allocation concealment (selection bias) | Low risk | Quote: "Via telephone entry, treatment (rasburicase or allopurinol) was randomly allocated to patients according to a computer‐generated randomization code schema" |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Participants knew which treatment they were allocated, and care providers knew which treatment a participant was allocated |
| Blinding of outcome assessment (detection bias) All‐cause mortality | Low risk | Assessment of death is unlikely to be mistaken or biased, even though outcome assessors knew which treatment participants were assigned |
| Blinding of outcome assessment (detection bias) Mortality due to tumour lysis syndrome | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Normalisation of uric acid level | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Area under curve (AUC) of serum uric acid level | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Adverse events | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Incomplete outcome data (attrition bias) All‐cause mortality | Low risk | This outcome was reported for all participants |
| Incomplete outcome data (attrition bias) Mortality due to tumour lysis syndrome | Low risk | This outcome was reported for all participants |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | High risk | 4% of participants in the treatment group and 12% of participants in the control group did not complete treatment or follow up and outcome was not certain for them |
| Incomplete outcome data (attrition bias) Normalisation of uric acid level | High risk | Only a subset of randomised participants (37% of the treatment group and 36% of the control group) who had baseline hyperuricaemia were analysed and reported |
| Incomplete outcome data (attrition bias) Area under curve (AUC) of serum uric acid level | Low risk | This outcome was reported for all participants |
| Incomplete outcome data (attrition bias) Adverse events | Low risk | This outcome was reported for all participants |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | The 2 groups may not be comparable at baseline because serum uric acid level was higher in the treatment group, which might introduce bias |
Kikuchi 2009.
| Methods | Design: RCT Randomisation method: central randomisation, details not available Stratification factor: baseline body weight (< 10 or ≥ 10 kg) | |
| Participants | Inclusion criteria: paediatric participants < 18 years with newly diagnosed haematological malignancies with hyperuricaemia (uric acid > 7.5 mg/dL for participants ≥ 13 years; uric acid > 6.5 mg/dL for participants < 13 years) or with a high tumour burden (defined as non‐Hodgkin's lymphoma stage IV; NHL stage III with ≥ 1 lymph node or mass > 5 cm, or LDH ≥ 3 times the upper limit of normal) or acute leukaemia with WBC ≥ 50,000/mm³ and LDH ≥ 3 times the upper limit of normal ECOG performance scale ≤ 3 or Lansky score ≥ 30 Life expectancy ≥ 45 days Exclusion criteria: administration of allopurinol within 72 h; known history of severe allergy, severe asthma, or both; low birth weight (< 2500 g) or gestational age < 37 weeks; previous therapy with urate oxidase; positive HBsAg, HCV antibodies, HIV‐1 or HIV‐2 antibodies; severe disorders of the liver or kidney (ALT > 5 times the upper limit of normal, total bilirubin > 3 times the upper limit of normal; creatinine > 3 times the upper limit of normal); uncontrollable infection including viral infection; G6PD deficiency; known family history of G6PD deficiency; known history of methaemoglobinaemia and haemolysis Number of participants (low‐dose group/high‐dose group): 15/15 Number of boys (low‐dose group/high‐dose group): 9/10 Age (low‐dose group/high‐dose group): (median) 11 (range = 1‐17)/7 (range = 0‐16) years Underlying haematological malignancies (low‐dose group/high‐dose group): acute leukaemia 9/13; lymphoma 6/2 Baseline hyperuricaemia (low‐dose group/high‐dose group): 8/5 Baseline creatinine (low‐dose group/high‐dose group): (mean) 52.3 (SD 22.6)/44.4 (SD 19.1) mg/dL |
|
| Interventions | Intervention (type of urate oxidase): rasburicase in both groups (0.15 mg/kg versus 0.2 mg/kg) Treatment regime in low‐dose group: rasburicase 0.15 mg/kg ivi over 30 min daily for 5 d Treatment regime in high‐dose group: rasburicase 0.2 mg/kg ivi over 30 min daily for 5 d Cointerventions: chemotherapy started 4‐24 h after the first dose of rasburicase in both groups |
|
| Outcomes |
|
|
| Notes | Duration of follow up: 5 weeks
Number of dropouts (low‐dose group/high‐dose group): 1 (3 concomitant grade 4 adverse events)/1 (lack of WBC result at baseline) The 2 groups may not be comparable at baseline because body weight was higher in the low‐dose group; baseline hyperuricaemia and diagnosis of lymphoma were more frequent in the low‐dose group Whether outcomes were clearly defined: yes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Participants were randomly allocated to the 2 treatment groups by central randomisation, but details were not available |
| Allocation concealment (selection bias) | Unclear risk | This was not mentioned |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Participants knew which treatment they were allocated, and care providers knew which treatment a participant was allocated |
| Blinding of outcome assessment (detection bias) All‐cause mortality | Low risk | Assessment of death is unlikely to be mistaken or biased, even though outcome assessors knew which treatment participants were assigned |
| Blinding of outcome assessment (detection bias) Mortality due to tumour lysis syndrome | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Normalisation of uric acid level | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Serial uric acid level | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Blinding of outcome assessment (detection bias) Adverse events | High risk | Outcome assessors knew which treatment participants were assigned as this was an open‐label trial |
| Incomplete outcome data (attrition bias) All‐cause mortality | Low risk | This outcome was reported for all participants |
| Incomplete outcome data (attrition bias) Mortality due to tumour lysis syndrome | Low risk | This outcome was reported for all participants |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | Low risk | 6.7% of participants in the high‐dose group did not complete treatment and this outcome for not reported. The dropout was explained. The low proportion of dropout is unlikely to cause significant bias |
| Incomplete outcome data (attrition bias) Normalisation of uric acid level | Low risk | 6.7% of participants in the high‐dose group did not complete treatment and this outcome for not reported. The dropout was explained. The low proportion of dropout is unlikely to cause significant bias |
| Incomplete outcome data (attrition bias) Serial uric acid level | Low risk | 6.7% of participants in the high‐dose group did not complete treatment and this outcome for not reported. The dropout was explained. The low proportion of dropout is unlikely to cause significant bias |
| Incomplete outcome data (attrition bias) Adverse events | Low risk | 6.7% of participants in the high‐dose group did not complete treatment and this outcome for not reported. The dropout was explained. The low proportion of dropout is unlikely to cause significant bias |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | The 2 groups may not be comparable at baseline because body weight was higher in the low‐dose group; baseline hyperuricaemia and diagnosis of lymphoma were more frequent in the low‐dose group |
Patte 2002.
| Methods | Design: CCT Stratification factor: not applicable | |
| Participants | Inclusion criteria: paediatric participants with stage III and IV B‐cell NHL or L3 ALL treated with the LMB89 protocol (intervention group), paediatric participants with stage IV B‐cell NHL or ALL treated with UKCCSG protocol (comparison group 1), or paediatric participants with stage IV B‐cell NHL or ALL treated with POG protocol (comparison group 2) Exclusion criteria: participants not treated in France excluded from intervention group Number of participants (intervention/comparison 1/comparison 2): 152/63/123 Number of boys (intervention/comparison 1/comparison 2): not available Age (intervention/comparison 1/comparison 2): not available Underlying haematological malignancies: only data for intervention group available: B‐NHL stage III 257 out of 410; B‐NHL stage IV 57 out of 410; L3 ALL 96 out of 410 Baseline renal failure (intervention/comparison 1/comparison 2): 21/410/not available Baseline elevated LDH ≥ 2 x normal (intervention/comparison 1/comparison 2): 234/410/not available |
|
| Interventions | Intervention (type of urate oxidase): Uricozyme Comparison 1 and 2 (type of control): allopurinol Treatment regime in intervention group: Uricozyme 50‐100 U/kg/d for 5‐7 d Treatment regime in comparison groups: not available Cointerventions: intervention group: alkaline hyperhydration 3 L/m²/d to obtain urine output 100‐120 ml/m²/h and urine pH 7; not stated in comparison groups | |
| Outcomes |
|
|
| Notes | Duration of follow up: 7 d Number of dropouts (intervention/comparison 1/comparison 2): 0/0/0 Potential confounders were not described or adjusted Comparability of the treatment groups at baseline was uncertain because some important baseline characteristics of comparison groups were not available Whether outcomes were clearly defined: yes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | This was not a randomised controlled trial |
| Allocation concealment (selection bias) | High risk | This was not a randomised controlled trial, and no allocation concealment was used |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Mortality due to tumour lysis syndrome | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Blinding was not used |
| Incomplete outcome data (attrition bias) Mortality due to tumour lysis syndrome | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | Low risk | The outcome data were complete for the groups of participants analysed |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | Comparability of the treatment groups at baseline was uncertain because baseline characteristics of comparison groups were not available. Potential confounders were not adjusted. Chemotherapy treatments were different in different centres in different locations, which may cause bias |
Pui 1997.
| Methods | Design: CCT Stratification factor: not applicable | |
| Participants | Inclusion criteria: paediatric participants with non‐B‐cell ALL Exclusion criteria: participants with history of allergy, G6PD deficiency or pregnancy, or who had not received methotrexate or 6‐mercaptopurine as preinduction therapy Number of participants (intervention/comparison): 119/129 Number of boys (intervention/comparison): not available Age (intervention/comparison): (median) 5.6/5.7 years Underlying haematological malignancies (intervention/comparison): all non‐B‐cell ALL Baseline WBC (intervention/comparison): (median) 11.7/13.8 x 10⁹/L Baseline uric acid (intervention/comparison): (median) 4.3/4.3 mg/dL Baseline lactate dehydrogenase (intervention/comparison): (median) 1243/957 U/L Baseline BUN (intervention/comparison): (median) 8.0/8.0 mg/dL Baseline creatinine (intervention/comparison): (median) 0.5/0.5 mg/dL Baseline calcium (intervention/comparison): (median) 9.4/9.4 mg/dL Baseline phosphate (intervention/comparison): (median) 4.9/4.7 mg/dL |
|
| Interventions | Intervention (type of urate oxidase): Uricozyme Comparison (type of control): allopurinol Treatment regime in intervention group: Uricozyme 100 units/kg ivi over 30 min daily for 5 d Treatment regime in comparison group: allopurinol 300 mg/m²/d orally for 5‐13 d Cointerventions: hydration with NaHCO₃ to maintain urine pH ≥ 6.5; oral phosphate binders (aluminium hydroxide or calcium carbonate) were given to participants as indicated | |
| Outcomes |
|
|
| Notes | Duration of follow up: 13 days
Number of dropouts (intervention/comparison): 0/0 Potential confounders were adjusted The intervention and the control groups appeared comparable at baseline Whether outcomes were clearly defined: yes The report of adverse events included additional 15 participants who had received an incomplete course of Uricozyme |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | This was not a randomised controlled trial |
| Allocation concealment (selection bias) | High risk | This was not a randomised controlled trial, and no allocation concealment was used |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Serial uric acid level | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Adverse events | High risk | Blinding was not used |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Serial uric acid level | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Adverse events | Low risk | The outcome data were complete for the groups of participants analysed |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | Potential confounders were not adjusted. Use of historical control may bias the results in favour of the newer treatment because of improvement in supportive care |
Rényi 2007.
| Methods | Design: CCT Stratification factor: not applicable | |
| Participants | Inclusion criteria: paediatric participants aged 6 months‐18 years with a recent diagnosis of B‐cell lineage ALL with an initial WBC ≥ 25,000/uL, or high‐grade non‐Hodgkin's lymphoma, or any type of ALL or NHL with a plasma uric acid ≥ 480 mmol/L and LDH > 500 IU/L, or either a creatinine or an LDH concentration > twice the upper limit of normal Exclusion criteria: history of clinically significant atopic allergy, bronchial asthma, G6PD deficiency or any type of haemolytic anaemia, previous treatment with rasburicase or non‐recombinant urate oxidase, hypersensitivity reaction against ingredients of the present preparation used in the study, participation in another drug experiment, pregnancy or lactation Number of participants (intervention/comparison): 12/14 Number of boys (intervention/comparison): 6/6 Age (intervention/comparison): (median) 4.5/6 Underlying haematological malignancies (intervention/comparison): leukaemia 8/13; lymphoma 4/1 Baseline creatinine (intervention/comparison): median 65 (range = 32‐85)/80 (range = 17‐353) umol/L Baseline uric acid (intervention/comparison): median 323 (range = 139‐1059)/207 (range = 51‐785) umol/L Baseline phosphate (intervention/comparison): median 1.32 (range = 0.97‐1.64)/1.62 (range = 0.98‐1.33) mmol/L Baseline LDH (intervention/comparison): 1909 (range = 497‐9760)/3193 (236‐20,560) U/L Baseline WBC (intervention/comparison): 51.8 (range = 2‐651)/56 (range = 0.4‐551) x 109/L |
|
| Interventions | Intervention (type of urate oxidase): rasburicase Comparison (type of control): allopurinol Treatment regime in intervention group: rasburicase 0.2 mg/kg ivi over 30 min daily for 5 d from day 1 of antineoplastic treatment Treatment regime in comparison group: allopurinol 300 mg/m²/d or 10 mg/kg/d divided every 8 h for 5‐7 d Cointerventions: hydration 3 L/m²/d, iv sodium bicarbonate 20‐40 mmol/L to maintain urine pH 6.5‐7 | |
| Outcomes |
|
|
| Notes | Duration of follow up: 12 d Number of dropouts (intervention/comparison): 0/0 Potential confounders were not described or adjusted The 2 groups may not be comparable at baseline because the treatment group had more participants with lymphoma, higher uric acid level, lower LDH level, lower serum creatinine, and phosphorus Whether outcomes were clearly defined: yes The report of adverse events included additional 24 participants who had received rasburicase in other centres |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | This was not a randomised controlled trial |
| Allocation concealment (selection bias) | High risk | This was not a randomised controlled trial, and no allocation concealment was used |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) All‐cause mortality | Low risk | Assessment of death is unlikely to be mistaken or biased, even though outcome assessors knew which treatment participants were assigned |
| Blinding of outcome assessment (detection bias) Mortality due to tumour lysis syndrome | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Serial uric acid level | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Adverse events | High risk | Blinding was not used |
| Incomplete outcome data (attrition bias) All‐cause mortality | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Mortality due to tumour lysis syndrome | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Serial uric acid level | High risk | Serum uric acid data at 24‐288 hours post‐treatment were missing in 6‐12 participants (43%‐86%) |
| Incomplete outcome data (attrition bias) Adverse events | Low risk | The outcome data were complete for the groups of participants analysed |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | The 2 groups may not be comparable at baseline because the treatment group had more participants with lymphoma, higher uric acid level, lower LDH level, lower serum creatinine, and phosphorus. Potential confounders were not adjusted. Use of historical control may bias the results in favour of the newer treatment because of improvement in supportive care |
Sánchez Tatay 2010.
| Methods | Design: CCT Stratification factor: not applicable | |
| Participants | Inclusion criteria: paediatric participants with haematological malignancies with TLS or at risk of TLS, with at least 1 of the following criteria: WBC > 50,000/uL or lactate dehydrogenase > 500 U/L; uric acid level ≥ 8 mg/dL or creatinine > 2 mg/dL; history of TLS in previous cycles of chemotherapy Exclusion criteria: participants with history of hypersensitivity to rasburicase or allopurinol, asthma, atopy, G6PD deficiency, or other metabolic causes of haemolytic anaemia Number of participants (intervention/comparison): 16/16 Number of boys (intervention/comparison): not available Age (intervention/comparison): not available Underlying haematological malignancies (intervention/comparison): not available Baseline uric acid (intervention/comparison): (median) 10.6 (SD 3.2)/11.3 (SD 5.8) mg/dL Baseline creatinine (intervention/comparison): (median) 0.93 (SD 0.81)/1.01 (SD 0.51) mg/dL Baseline phosphate (intervention/comparison): (median) 6.28 (SD 2.29)/6.72 (SD 5.02) mg/dL |
|
| Interventions | Intervention (type of urate oxidase): rasburicase Comparison (type of control): allopurinol Treatment regime in intervention group: rasburicase 0.2 mg/kg ivi daily (duration of treatment was not mentioned) Treatment regime in comparison group: allopurinol 10 mg/kg/d divided every 8 h (duration of treatment was not mentioned) Cointerventions: not mentioned | |
| Outcomes |
|
|
| Notes | Duration of follow up: 4 d
Number of dropouts (intervention/comparison): 0/0 Potential confounders were not described or adjusted Comparability of the treatment groups at baseline was uncertain because some important baseline characteristics of comparison groups were not available Whether outcomes were clearly defined: yes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | This was not a randomised controlled trial |
| Allocation concealment (selection bias) | High risk | This was not a randomised controlled trial, and no allocation concealment was used |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) All‐cause mortality | Low risk | Assessment of death is unlikely to be mistaken or biased, even though outcome assessors knew which treatment participants were assigned |
| Blinding of outcome assessment (detection bias) Mortality due to tumour lysis syndrome | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Serial uric acid level | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Adverse events | High risk | Blinding was not used |
| Incomplete outcome data (attrition bias) All‐cause mortality | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Mortality due to tumour lysis syndrome | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Serial uric acid level | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Adverse events | Low risk | The outcome data were complete for the groups of participants analysed |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | Comparability of the treatment groups at baseline was uncertain because some important baseline characteristics of comparison groups were not available. Potential confounders were not adjusted. Use of historical control may bias the results in favour of the newer treatment because of improvement in supportive care |
Wössmann 2003.
| Methods | Design: CCT Stratification factor: not applicable | |
| Participants | Inclusion criteria: paediatric participants ≤ 18 years with B‐ALL or stage III and IV B‐NHL and LDH ≥ 500 U/L treated in the trials NHL‐BFM 90 and 95, during the period November 1997‐December 2001 (intervention group) or April 1990‐March 1996 (comparison group) Exclusion criteria: nil Number of participants (intervention/comparison): 130/218 Number of boys (intervention/comparison): N/A Age (intervention/comparison): N/A Underlying haematological malignancies: B‐ALL 53/78; B‐NHL 77/140 Baseline elevated LDH > 1000 U/L (intervention/comparison): 49.6%/47.2% |
|
| Interventions | Intervention (type of urate oxidase): Uricozyme Comparison (type of control): allopurinol and alkalinization Treatment regime in intervention group: Uricozyme 3 x 50 U/kg/d for 3‐4 d Treatment regime in comparison group: allopurinol 10 mg/kg/d + alkalinization to maintain urine pH 7 Cointerventions: hydration 3 to 4.5 L/m²/d | |
| Outcomes |
|
|
| Notes | Duration of follow up not reported Number of dropouts (intervention/comparison): 0/0 Potential confounders were not described or adjusted The 2 groups may not be comparable at baseline because the treatment group had fewer participants who were critically ill or had complications after initial surgery Whether outcomes were clearly defined: yes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | This was not a randomised controlled trial |
| Allocation concealment (selection bias) | High risk | This was not a randomised controlled trial, and no allocation concealment was used |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) Incidence of clinical tumour lysis syndrome | High risk | Blinding was not used |
| Blinding of outcome assessment (detection bias) All‐cause mortality | Low risk | Assessment of death is unlikely to be mistaken or biased, even though outcome assessors knew which treatment participants were assigned |
| Blinding of outcome assessment (detection bias) Renal failure requiring renal replacement therapy | High risk | Blinding was not used |
| Incomplete outcome data (attrition bias) Incidence of clinical tumour lysis syndrome | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) All‐cause mortality | Low risk | The outcome data were complete for the groups of participants analysed |
| Incomplete outcome data (attrition bias) Renal failure requiring renal replacement therapy | Low risk | The outcome data were complete for the groups of participants analysed |
| Selective reporting (reporting bias) | Unclear risk | The study protocol was not available, and it was unclear whether there was selective reporting of outcomes |
| Other bias | High risk | The 2 groups may not be comparable at baseline because the treatment group had fewer participants who were critically ill or had complications after initial surgery. Potential confounders were not adjusted. Use of historical control may bias the results in favour of the newer treatment because of improvement in supportive care |
ALL: acute lymphoblastic leukaemia ALT: alanine aminotransferase B‐NHL: B cell non‐Hodgkin lymphoma BUN: blood urea nitrogen CCT: controlled clinical trial ECOG: Eastern Cooperative Oncology Group G6PD: glucose‐6‐phosphate dehydrogenase deficiency HBsAg: hepatitis B virus surface antigen HCV: hepatitis C virus iv: intravenous ivi: intravenous infusion LDH: lactate dehydrogenase N/A: information not available NHL: non‐Hodgkin lymphoma RCT: randomised controlled trial SD: standard deviation TLS: tumour lysis syndrome WBC: white blood cell (count) UKCCSG: United Kingdom Children's Cancer Study Group
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Cortes 2010 | This was an RCT recruiting adult participants only and no paediatric participants |
| Ishizawa 2009 | This was an RCT recruiting adult participants only and no paediatric participants |
| Vadhan‐Raj 2012 | This was an RCT recruiting adult participants only and no paediatric participants |
RCT: randomised controlled trial.
Characteristics of ongoing studies [ordered by study ID]
NCT00199043.
| Trial name or title | Randomised phase III trial of effectivity and safety of rasburicase compared with allopurinol for treatment of hyperuricemia in patients with acute lymphoblastic leukemia or high‐grade NHL with high risk of tumour lysis syndrome (> 15 yrs) |
| Methods | RCT |
| Participants | Participants aged above 15 years, participating in the GMALL B‐ALL/NHL‐Study 2002; the GMALL‐Study 07/2003; or the GMALL‐Study Elderly 1/2003 that fulfil the following criteria: bulky disease (> 7.5 cm), high LDH (> 2 times the upper limit of normal), uric acid > 8 mg/dl (> 475 µmol/L) at diagnosis, and leukocytes > 30000/µL |
| Interventions | Arm 1: allopurinol Arm 2: rasburicase |
| Outcomes | Primary outcomes: renal function, uric acid, electrolytes, adverse events, mortality in pre‐phase and the 2 following cycles of chemotherapy, time and dose compliance of chemotherapy Secondary outcomes: response rate, incidence of TLS |
| Starting date | May 2003 |
| Contact information | Dieter Hoelzer, University Hospital, Medical Dept. II, Frankfurt, Germany, 60590 |
| Notes | Study completed. Results not available yet. Not all participants are eligible for this review (for example, elderly participants may be included in this study) |
NCT01200485.
| Trial name or title | A randomized phase 2 study to evaluate the efficacy of rasburicase in patients at risk for TLS during two cycles of chemotherapy |
| Methods | RCT |
| Participants | Participants that are high risk for TLS or potential/intermediate risk for TLS as described below. (a) High risk: hyperuricaemia of malignancy (uric acid levels > 7.5 mg/dL); or diagnosis of very aggressive lymphoma/leukaemia based on Revised European‐American Lymphoma (REAL) classification; acute myeloid leukaemia, CML in blast crisis; high‐grade myelodysplastic syndrome only if they have > 10% bone marrow blast involvement and given aggressive treatment similar to acute myeloid leukaemia. (B) Potential risk: diagnosis of aggressive lymphoma/leukaemia based on REAL classification, plus 1 or more of the following criteria: LDH >/= 2 times the upper limit of normal; stage III‐IV disease; stage I‐II disease with at least 1 lymph node/tumour > 5 cm in diameter. For participants with potential/intermediate risk for TLS, only those planned to receive alternating regimens (or non‐standard regimens) in 2 cycles (example; R‐Hyper‐CVAD alternating with MTX/ARA‐C) will be eligible |
| Interventions | Arm A: Cycle 2 chemotherapy, rasburicase 0.15 mg/kg IV d 1, additional dose on days 2‐5 at physician's discretion Arm B: Cycle 2 chemotherapy, allopurinol 300 mg/d IV days 1‐5 + rasburicase 0.15 mg/kg IV d 1 if uric acid blood levels dictate single dose or more |
| Outcomes | Primary outcome: incidence of laboratory TLS during cycle 2 (as defined by the Cairo‐Bishop criteria) |
| Starting date | May 2011 |
| Contact information | Saroj Vadhan‐Raj, UT MD Anderson Cancer Center, Houston, Texas, United States, 77030 Telephone: +1 713 792 7966 |
| Notes | Ongoing study recruiting participants. Not all participants are eligible for this review (for example, elderly participants may be included in this study) |
CML: chronic myelogenous leukaemia R‐Hyper‐CVAD: chemotherapy regimen consisting of rituximab, cyclophosphamide, vincristine, adriamycin, dexamethasone LDH: lactate dehydrogenase MTX/ARA‐C: methotrexate and cytarabine NHL: non‐Hodgkin lymphoma RCT: randomised controlled trial TLS: tumour lysis syndrome
Differences between protocol and review
We used the mean difference for continuous outcomes instead of the weighted mean difference. We updated the 'Risk of bias' assessment according to the latest version of the Cochrane Handbook for Systematic Reviews of Interventions (version 5.1.0). We still reported outcomes with less than 70% of participants' data, but we considered them to be prone to bias. We did not calculate risk ratio if there was only one study available for a particular outcome and there was no event in one of the groups; we used the Fisher's exact test (performed using SPSS 19) to determine the P value in such situations. In the update 2013, we changed the unit of uric acid measurements to mg/dL, instead of micromol/L in the original review, as mg/dL appeared to be more widely used. In the update 2016, we included GRADE assessment and 'Summary of findings' table and updated risk of bias assessment.
Contributions of authors
Cheuk DKL: protocol development, searching for trials, 'Risk of bias' assessment and GRADE assessment of trials, data extraction, data input, data analyses, development of final review, preparation of 'Summary of findings' table, corresponding author. Chiang AKS: protocol development, 'Risk of bias' assessment of trials, data extraction, data input, data analyses, development of final review. Chan GCF: protocol development, development of final review. Ha SY: protocol development, development of final review.
Sources of support
Internal sources
-
The Library of The University of Hong Kong, Hong Kong.
The Library of The University of Hong Kong provided support in retrieving full‐texts of studies.
External sources
No sources of support supplied
Declarations of interest
Daniel KL Cheuk: nothing to declare Alan KS Chiang: nothing to declare Godfrey CF Chan: nothing to declare Shau Yin Ha: nothing to declare
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Goldman 2001 {published data only}
- Goldman SC, Holcenberg JS, Finklestein JZ, Hutchinson R, Kreissman S, Johnson FL, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood 2001;97(10):2998‐3003. [DOI] [PubMed] [Google Scholar]
Kikuchi 2009 {published data only}
- Kikuchi A, Kigasawa H, Tsurusawa M, Kawa K, Kikuta A, Tsuchida M, et al. A study of rasburicase for the management of hyperuricemia in pediatric patients with newly diagnosed hematologic malignancies at high risk for tumor lysis syndrome. International Journal of Hematology 2009;90(4):492‐500. [DOI] [PubMed] [Google Scholar]
Patte 2002 {published data only}
- Patte C, Sakiroglu C, Ansoborlo S, Baruchel A, Plouvier E, Pacquement H, et al. Urate‐oxidase in the prevention and treatment of metabolic complications in patients with B‐cell lymphoma and leukemia, treated in the Société Française d'Oncologie Pédiatrique LMB89 protocol. Annals of Oncology 2002;13(5):789‐95. [DOI] [PubMed] [Google Scholar]
Pui 1997 {published data only}
- Pui CH, Relling MV, Lascombes F, Harrison PL, Struxiano A, Mondesir JM, et al. Urate oxidase in prevention and treatment of hyperuricemia associated with lymphoid malignancies. Leukemia 1997;11(11):1813‐6. [DOI] [PubMed] [Google Scholar]
Rényi 2007 {published data only}
- Rényi I, Bárdi E, Udvardi E, Kovács G, Bartyik K, Kajtár P, et al. Prevention and treatment of hyperuricemia with rasburicase in children with leukemia and non‐Hodgkin's lymphoma. Pathology Oncology Research 2007;13(1):57‐62. [DOI] [PubMed] [Google Scholar]
Sánchez Tatay 2010 {published data only}
- Sánchez Tatay V, López Castilla JD, Carmona Ponce JM, Pérez Hurtado JM, Quiroga Cantero E, Loscertales Abril M. Rasburicase versus allopurinol in the treatment of hyperuricaemia in tumour lysis syndrome [Rasburicasa versus alopurinol como tratamientode la hiperuricemia en el sı´ndrome de lisis tumoral]. Anales de Pediatría 2010;72(2):103‐10. [DOI] [PubMed] [Google Scholar]
Wössmann 2003 {published data only}
- Wössmann W, Schrappe M, Meyer U, Zimmermann M, Reiter A. Incidence of tumor lysis syndrome in children with advanced stage Burkitt's lymphoma/leukemia before and after introduction of prophylactic use of urate oxidase. Annals of Hematology 2003;82(3):160‐5. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Cortes 2010 {published data only}
- Cortes J, Moore JO, Maziarz RT, Wetzler M, Craig M, Matous J, et al. Control of plasma uric acid in adults at risk for tumor lysis syndrome: efficacy and safety of rasburicase alone and rasburicase followed by allopurinol compared with allopurinol alone‐results of a multicenter phase III study. Journal of Clinical Oncology 2010;28(27):4207‐13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ishizawa 2009 {published data only}
- Ishizawa K, Ogura M, Hamaguchi M, Hotta T, Ohnishi K, Sasaki T, et al. Safety and efficacy of rasburicase (SR29142) in a Japanese phase II study. Cancer Science 2009;100(2):357‐62. [DOI] [PMC free article] [PubMed] [Google Scholar]
Vadhan‐Raj 2012 {published data only}
- Vadhan‐Raj S, Fayad LE, Fanale MA, Pro B, Rodriguez A, Hagemeister FB, et al. A randomized trial of a single‐dose rasburicase versus five‐daily doses in patients at risk for tumor lysis syndrome. Annals of Oncology 2012;23(6):1640‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to ongoing studies
NCT00199043 {published data only}
- NCT00199043. Randomised phase III trial of effectivity and safety of rasburicase compared with allopurinol for treatment of hyperuricemia in patients with acute lymphoblastic leukemia or high‐grade NHL with high risk of tumor lysis syndrome (> 15 yrs). clinicaltrials.gov/ct2/show/NCT00199043?term=NCT00199043&rank=1 (accessed 26 February 2013).
NCT01200485 {published data only}
- NCT01200485. A randomized phase 2 study to evaluate the efficacy of rasburicase in patients at risk for TLS during two cycles of chemotherapy. clinicaltrials.gov/ct2/show/NCT01200485?term=NCT01200485&rank=1 (accessed 26 February 2013).
Additional references
Annemans 2003a
- Annemans L, Moeremans K, Lamotte M, Garcia Conde J, Berg H, Myint H, et al. Incidence, medical resource utilisation and costs of hyperuricemia and tumour lysis syndrome in patients with acute leukaemia and non‐Hodgkin's lymphoma in four European countries. Leukemia & Lymphoma 2003;44(1):77‐83. [DOI] [PubMed] [Google Scholar]
Annemans 2003b
- Annemans L, Moeremans K, Lamotte M, Garcia Conde J, Berg H, Myint H, et al. Pan‐European multicentre economic evaluation of recombinant urate oxidase (rasburicase) in prevention and treatment of hyperuricaemia and tumour lysis syndrome in haematological cancer patients. Supportive Care in Cancer 2003;11(4):249‐57. [DOI] [PubMed] [Google Scholar]
Bose 2011
- Bose P, Qubaiah O. A review of tumour lysis syndrome with targeted therapies and the role of rasburicase. Journal of Clinical Pharmacy and Therapeutics 2011;36(3):299‐326. [DOI] [PubMed] [Google Scholar]
Bosly 2003
- Bosly A, Sonet A, Pinkerton CR, McCowage G, Bron D, Sanz MA, et al. Rasburicase (recombinant urate oxidase) for the management of hyperuricemia in patients with cancer: report of an international compassionate use study. Cancer 2003;98(5):1048‐54. [DOI] [PubMed] [Google Scholar]
Cairo 2004
- Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. British Journal of Haematology 2004;127(1):3‐11. [DOI] [PubMed] [Google Scholar]
Cairo 2010
- Cairo MS, Coiffier B, Reiter A, Younes A, TLS Expert Panel. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases: an expert TLS panel consensus. British Journal of Haematology 2010;149(4):578‐86. [DOI] [PubMed] [Google Scholar]
Coiffier 2003
- Coiffier B, Mounier N, Bologna S, Fermé C, Tilly H, Sonet A, et al. Efficacy and safety of rasburicase (recombinant urate oxidase) for the prevention and treatment of hyperuricemia during induction chemotherapy of aggressive non‐Hodgkin's lymphoma: results of the GRAAL1 (Groupe d'Etude des Lymphomes de l'Adulte Trial on Rasburicase Activity in Adult Lymphoma) study. Journal of Clinical Oncology 2003;21(23):4402‐6. [DOI] [PubMed] [Google Scholar]
Coiffier 2008
- Coiffier B, Altman A, Pui CH, Younes A, Cairo MS. Guidelines for the management of pediatric and adult tumor lysis syndrome: an evidence‐based review. Journal of Clinical Oncology 2008;26(16):2767‐78. [DOI] [PubMed] [Google Scholar]
Deeks 2011
- Deeks JJ, Higgins JPT, Altman DG (editors). Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.handbook.cochrane.org.
Egger 1997
- Egger M, Davey‐Smith G, Schneider M, Minder C. Bias in meta‐analysis detected by a simple, graphical test. BMJ 1997;315:629‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
GRADEpro 2015 [Computer program]
- McMaster University, Evidence Prime, Inc.. GRADEpro Guideline Development Tool. McMaster University, Evidence Prime, Inc., 2015.
Greene 1969
- Greene ML, Fujimoto WY, Seegmiller JE. Urinary xanthine stones ‐ a rare complication of allopurinol therapy. The New England Journal of Medicine 1969;280(8):426‐7. [DOI] [PubMed] [Google Scholar]
Gutierrez‐Macias 2005
- Gutierrez‐Macias A, Lizarralde‐Palacios E, Martinex‐Odriozola P, Miguel‐De la Villa F. Fatal allopurinol hypersensitivity syndrome after treatment of asymptomatic hyperuricemia. BMJ 2005;331(7517):623‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2002
- Higgins JP, Thompson SG. Quantifying heterogeneity in a meta‐analysis. Statistics in Medicine 2002;21(11):1539‐58. [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Hummel 2003
- Hummel M, Buchheidt D, Reiter S, Bergmann J, Hofheinz R, Hehlmann R. Successful treatment of hyperuricemia with low doses of recombinant urate oxidase in four patients with hematologic malignancy and tumor lysis syndrome. Leukemia 2003;17(12):2542‐4. [DOI] [PubMed] [Google Scholar]
Hutcherson 2006
- Hutcherson DA, Gammon DC, Bhatt MS, Faneuf M. Reduced‐dose rasburicase in the treatment of adults with hyperuricemia associated with malignancy. Pharmacotherapy 2006;26(2):242‐7. [DOI] [PubMed] [Google Scholar]
Jeha 2005
- Jeha S, Kantarjian H, Irwin D, Shen V, Shenoy S, Blaney S, et al. Efficacy and safety of rasburicase, a recombinant urate oxidase (Elitek), in the management of malignancy‐associated hyperuricemia in pediatric and adult patients: final results of a multicenter compassionate use trial. Leukemia 2005;19(1):34‐8. [DOI] [PubMed] [Google Scholar]
Kremer 2016
- Kremer LCM, Jellema P, Leclercq E, Noorman JK, Dalen EC. Cochrane Childhood Cancer Group. About The Cochrane Collaboration (Cochrane Review Groups (CRGs)) 2016:Issue 7. Art. No.: CHILDCA.
Lascombes 1998
- Lascombes F, Sommelet D, Gebhard F, Leverger G, Schaison G, Pinkerton R, et al. High efficacy of recombinant urate oxidase in prevention of renal failure related to tumor lysis syndrome. Blood 1998;92(Suppl 1):237b. [Google Scholar]
Lee 2003
- Lee AC, Li CH, So KT, Chan R. Treatment of impending tumor lysis with single‐dose rasburicase. The Annals of Pharmacotherapy 2003;37(11):1614‐7. [DOI] [PubMed] [Google Scholar]
Liu 2005
- Liu CY, Sims‐McCallum RP, Schiffer CA. A single dose of rasburicase is sufficient for the treatment of hyperuricemia in patients receiving chemotherapy. Leukemia Research 2005;29(4):463‐5. [DOI] [PubMed] [Google Scholar]
McDonnell 2006
- McDonnell AM, Lenz KL, Frei‐Lahr DA, Hayslip J, Hall PD. Single‐dose rasburicase 6 mg in the management of tumor lysis syndrome in adults. Pharmacotherapy 2006;26(6):806‐12. [DOI] [PubMed] [Google Scholar]
Navolanic 2003
- Navolanic PM, Pui CH, Larson RA, Bishop MR, Pearce TE, Cairo MS, et al. Elitek‐rasburicase: an effective means to prevent and treat hyperuricemia associated with tumor lysis syndrome, a Meeting Report, Dallas, Texas, January 2002. Leukemia 2003;17(3):499‐514. [DOI] [PubMed] [Google Scholar]
Pui 2001a
- Pui CH, Mahmoud HH, Wiley JM, Woods GM, Leverger G, Camitta B, et al. Recombinant urate oxidase for the prophylaxis or treatment of hyperuricemia in patients with leukemia or lymphoma. Journal of Clinical Oncology 2001;19(3):697‐704. [DOI] [PubMed] [Google Scholar]
Pui 2001b
- Pui CH, Jeha S, Irwin D, Camitta B. Recombinant urate oxidase (rasburicase) in the prevention and treatment of malignancy‐associated hyperuricemia in pediatric and adult patients: results of a compassionate‐use trial. Leukemia 2001;15(10):1505‐9. [DOI] [PubMed] [Google Scholar]
Rampello 2006
- Rampello E, Fricia T, Malaguarnera M. The management of tumor lysis syndrome. Nature Clinical Practice Oncology 2006;3(8):438‐47. [DOI] [PubMed] [Google Scholar]
RevMan 2014 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Sanofi 2011
- Sanofi‐Synthelabo. Elitek (rasburicase) product monograph. products.sanofi‐aventis.us/elitek/elitek.html (accessed 23 July 2012).
Schünemann 2011a
- Schünemann HJ, Oxman AD, Higgins JPT, Vist GE, Glasziou P, Guyatt GH. Chapter 11: Presenting results and 'Summary of findings' tables. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Schünemann 2011b
- Schünemann HJ, Oxman AD, Vist GE, Higgins JPT, Deeks JJ, Glasziou P, et al (editors). Chapter 12: Interpreting results and drawing conclusions. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Shin 2006
- Shin HY, Kang HJ, Park ES, Choi HS, Ahn HS, Kim SY, et al. Recombinant urate oxidase (rasburicase) for the treatment of hyperuricemia in pediatric patients with hematologic malignancies: results of a compassionate prospective multicenter study in Korea. Pediatric Blood and Cancer 2006;46(4):439‐45. [DOI] [PubMed] [Google Scholar]
SPSS 2010 [Computer program]
- IBM Corporation. IBM SPSS Statistics for Windows, Version 19.0. Armonk, NY: IBM Corporation, 2010.
Tosi 2008
- Tosi P, Barosi G, Lazzaro C, Liso V, Marchetti M, Morra E, et al. Consensus conference on the management of tumor lysis syndrome. Haematologica 2008;93(12):1877‐85. [DOI] [PubMed] [Google Scholar]
Trifilio 2006
- Trifilio S, Gordon L, Singhal S, Tallman M, Evens A, Rashid K, et al. Reduced‐dose rasburicase (recombinant xanthine oxidase) in adult cancer patients with hyperuricemia. Bone Marrow Transplantation 2006;37(11):997‐1001. [DOI] [PubMed] [Google Scholar]
Wang 2006
- Wang LY, Shih LY, Chang H, Jou ST, Lin KH, Yeh TC, et al. Recombinant urate oxidase (rasburicase) for the prevention and treatment of tumor lysis syndrome in patients with hematologic malignancies. Acta Haematologica 2006;115(1‐2):35‐8. [DOI] [PubMed] [Google Scholar]
Yim 2003
- Yim BT, Sims‐McCallum RP, Chong PH. Rasburicase for the treatment and prevention of hyperuricemia. The Annals of Pharmacotherapy 2003;37(7‐8):1047‐54. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Cheuk 2008
- Cheuk DKL, Chiang AKS, Chan GCF, Ha SY. Urate oxidase for prevention and treatment of tumor lysis syndrome in children with cancer (Protocol). Cochrane Database of Systematic Reviews 2008, Issue 1. [DOI: 10.1002/14651858.CD006945] [DOI] [Google Scholar]
Cheuk 2010
- Cheuk DKL, Chiang AKS, Chan GCF, Ha SY. Urate oxidase for the prevention and treatment of tumor lysis syndrome in children with cancer. Cochrane Database of Systematic Reviews 2010, Issue 6. [DOI: 10.1002/14651858.CD006945.pub2] [DOI] [PubMed] [Google Scholar]
Cheuk 2014
- Cheuk DKL, Chiang AKS, Chan GCF, Ha SY. Urate oxidase for the prevention and treatment of tumour lysis syndrome in children with cancer. Cochrane Database of Systematic Reviews 2014, Issue 8. [DOI: 10.1002/14651858.CD006945.pub3] [DOI] [PubMed] [Google Scholar]


