Abstract
Background
Excision of the transformation zone of the cervix is the most commonly used approach to treat cervical precancerous lesions (cervical intraepithelial neoplasia (CIN)) to reduce the risk of developing cervical cancer. As the excision of the transformation zone leaves a raw area on the cervix, there is a risk of infection following the procedure. The incidence of infection after cold knife conization (CKC) is 36%, whereas the incidence for large loop excision of the transformation zone (LLETZ, also known as loop electrical excision procedure (LEEP)) is much lower (0.8% to 14.4%). Prophalytic antibiotics may prevent an infection developing and are often prescribed for CKC. However, there are no formal recommendations regarding the use of prophylactic antibiotics for infection prevention in women undergoing surgical excisional treatment for cervical precancerous lesions.
Objectives
To evaluate the effectiveness and safety of antibiotics for infection prevention following excision of the cervical transformation zone.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL) (2016, Issue 4), MEDLINE, Embase, LILACS to May 2016. We also checked registers of clinical trials, citation lists of included studies, key textbooks and previous systematic reviews for potentially relevant studies
Selection criteria
We included randomised controlled trials (RCTs) evaluating the effectiveness and safety of prophylactic antibiotics versus a placebo or no treatment in women having excision of the cervical transformation zone, regardless of the type of surgical excisional method used.
Data collection and analysis
We used standard methodological procedures expected by Cochrane. Two review authors independently selected potentially relevant trials, extracted data, and assessed risk of bias, compared results and resolved disagreements by discussion. We contacted investigators for additional data, where possible.
Main results
Of the 370 records that we identified as a result of the search (excluding duplicates), we regarded six abstracts and titles as potentially relevant studies. Of these six studies, three met the inclusion criteria involving 708 participants; most trials were at moderate or high risk of bias (risk mainly due to lack of blinding and high rate of incomplete data). We did not identify any ongoing trials. Although all included studies had been published in peer‐reviewed journals at the time of the search and data extraction, numerical data regarding the outcome measured in one trial involving 77 participants were insufficient for inclusion in a meta‐analyses.
The difference in the rates of prolonged vaginal discharge or presumed cervicitis (one study; 348 participants; risk ratio (RR), 1.29; 95% confidence interval (CI) 0.72 to 2.31; low‐quality evidence) and severe vaginal bleeding (two studies; 638 participants; RR 1.21; 95% CI 0.52 to 2.82; very low‐quality evidence) among the two comparison groups did not reach the level for clinically important effect. In addition, there was no difference in adverse events related to antibiotics i.e. nausea/vomiting, diarrhoea, and headache among the two comparison groups (two studies; 638 participants; RR 1.69; 95% CI 0.85 to 3.34; very low‐quality evidence). There were no differences in the incidence of fever (RR, 2.23; 95% CI 0.20 to 24.36), lower abdominal pain (RR, 1.03; 95% CI 0.61 to 1.72), unscheduled medical consultation (RR 2.68, 95% CI 0.97 to 7.41), and additional self‐medication (RR 1.22; 95% CI 0.56 to 2.67) between the two comparison groups (one study; 290 participants; low to very low‐quality evidence).
Authors' conclusions
As only limited data are available from three trials with overall moderate to high risk of bias, there is insufficient evidence to support use of antibiotics to reduce infectious complications following excision of the cervical transformation zone. In addition, there were minimal data about antibiotic‐related adverse events and no information on the risk of developing antibiotic resistance. Antibiotics given for infection prevention after excision of the cervical transformation zone should only be used in the context of clinical research, to avoid unnecessary prescription of antibiotics and to prevent further increases in antibiotic resistance.
Plain language summary
Prophylactic antibiotics to prevent infection after cervical excision
Background Pre‐cancerous cervical lesions can be treated by either excision or destruction of the abnormal cells from the cervix, to reduce the risk of developing cervical cancer in the future. The advantage of excisional treatment is that the abnormal cells are removed, rather than destroyed, so tissue can be sent for detailed examination to confirm the diagnosis histologically and make sure the affected area has been completely removed. As the excision of the transformation zone leaves a raw area on the cervix, there is a risk of getting an infection following the procedure. Antibiotics are sometimes given before surgical procedures to prevent infection developing (prophylactic), rather than to treat an existing infection. However, prophylactic antibiotics may not be necessary or effective. In addition, antibiotics can cause side effects (adverse events). Importantly, there are increasing concerns about antibiotic over‐use promoting antibiotic resistance in bacteria.
Review question Do prophylactic antibiotics prevent infection in women undergoing excision of the cervical transformation zone and what are the side effects?
Main findings We searched the literature to May 2016 and found three published randomised trials that met the review inclusion criteria. We did not identify any ongoing trials. The three included studies involved 708 participants who had undergone excisional treatment to the cervix (known as laser or large loop excision of transformation zone (LLETZ) or loop electrosurgical excision procedure (LEEP)). Two studies tested a antimicrobial vaginal pessary versus no treatment; the other tested oral antibiotics compared with placebo. We found that there was no benefit to prophylactic antibiotics after LLETZ to reduce or prevent prolonged vaginal discharge, severe vaginal bleeding, fever, lower abdominal pain, unscheduled medical consultation, and additional self‐medication. There was little information on antibiotic‐related adverse effects. The limited evidence available does not support routinely giving antibiotics for infection prevention after LLETZ. As there are growing concerns with antibiotic resistance, antibiotics for infection prevention after excision of the cervical transformation zones should only be used in the context of clinical trials.
Quality of the evidence The quality of the evidence regarding prophylactic antibiotics for preventing severe vaginal bleeding, fever, and adverse events was very low, with evidence from other comparisons being of low quality.
Summary of findings
for the main comparison.
| Antibiotics compared with placebo or no treatment for infection prevention after excision of the cervical transformation zone | ||||||
|
Patient or population: Women undergoing excision of the cervical transformation zone for cervical neoplasia Settings: Outpatients setting, the colposcopy clinic Intervention: Prophylactic antibiotics Comparison: Placebo or no treatment | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| [Placebo or no treatment] | [Antibiotics] | |||||
|
Number of participants who experienced prolonged vaginal discharge Follow‐up period: 2 weeks after the procedure |
103 per 1000 | 133 per 1000 (74 to 238) | RR 1.29 (0.72 to 2.31) | 348 (1 study) | ⊕⊕⊝⊝ low1,3 | |
|
Number of participants who had to be admitted for post‐procedure bleeding Follow‐up period: 2‐3 weeks after the procedure |
31 per 1000 | 38 per 1000 (16 to 87) |
RR 1.21 (0.52 to 2.82) |
638 (2 studies) |
⊕⊝⊝⊝ very low1,2,3,4 | |
|
Number of participants who developed fever Follow‐up period: 3 weeks after the procedure |
7 per 1000 | 16 per 1000 (1 to 171) |
RR 2.23 (0.20 to 24.36) |
290 (1 study) |
⊕⊝⊝⊝ very low2,3,4,5 | |
|
Number of participants who experienced lower abdominal pain Follow‐up period: 3 weeks after the procedure |
163 per 1000 | 168 per 1000 (99 to 289) |
RR 1.03 (0.61 to 1.72) |
290 (1 study) |
⊕⊕⊝⊝ low2,3 | |
|
Number of participants who experienced any adverse effects related to antibiotics Follow‐up period: 2‐3 weeks after the procedure |
37 per 1000 | 63 per 1000 (31 to 124) |
RR 1.69 (0.85 to 3.34) |
638 (2 studies) |
⊕⊝⊝⊝ very low1,2,3,4 | |
|
Number of participants who received additional medical consultant (for any reasons) Follow‐up period: 3 weeks after the procedure |
33 per 1000 | 88 per 1000 (32 to 255) |
RR 2.68 (0.97 to 7.41) |
290 (1 study) |
⊕⊕⊝⊝ low2,3 | |
|
Number of participants who had additional self‐medication Follow‐up period: 3 weeks after the procedure |
72 per 1000 | 88 per 1000 (40 to 192) |
RR 1.22 (0.56 to 2.67) |
290 (1 study) |
⊕⊕⊝⊝ very low1,2,3 | |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk Ratio | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1Based on the high risk of attrition bias (high rate of incomplete participants' record charts) 2 Based on high risk of detection bias (mainly due to lack of blinding of participants) 3 The analyses were not performed on an intention‐to‐treat basis 4 Small number of events in the analyses 5 Sparsness of data
Background
Description of the condition
Cervical cancer remains a major world public health problem, as there are 500,000 new cases diagnosed and 275,000 deaths from cervical cancer occurring among women worldwide each year (Wiebe 2012). Many cervical cancers can be prevented through screening programs designed to detect and treat cervical cancer precursors. At the present, cervical intraepithelial neoplasia (CIN) grade 2 to 3, or the so‐called 'high‐grade squamous intraepithelial lesions (HSILs)' and adenocarcinoma in situ (AIS) are acknowledged as precancerous lesions of squamous cell carcinoma and adenocarcinoma of the uterine cervix, respectively (Massad 2013).
Excision of the cervical transformation zone, an area where the majority of abnormalities occur, is the most commonly used approach to treat cervical precancerous lesions. The major advantage of excisional treatment is that the affected area is removed, so the excised specimen can be sent for detailed pathological examination to determine the severity of the lesion and ensure that abnormal lesion has been completely removed. The common surgical techniques for excision of the cervical transformation zone include large loop excision of the transformation zone (LLETZ), cold‐knife conization (CKC), and laser conization (LC). The terms LLETZ and loop electrosurgical excision procedure (LEEP) both have been used to describe a technique to excise the transformation zone using a fine wire loop with an electrical current (diathermy). This technique is also referred to as loop diathermy excision, loop biopsy, and loop cone. Current evidence obtained from a Cochrane review conducted to evaluate the effectiveness of various surgical techniques for treating cervical intraepithelial neoplasia concludes that there is no single method that is a superior surgical technique for treating cervical intraepithelial neoplasia (Martin‐Hirsch 2013).
Genital infections, as defined by cervical tenderness, fever, or wound infection requiring antibiotic therapy, have been reported to be as high as 36% after CKC (Janthanaphan 2009) and may have serious complications such as sepsis from lung and liver abscesses (Treszezamsky 2010). LLETZ has a lower incidence of infectious complications ranging from 0.8% to 14.4% (Kietpeerakool 2007; Lopez 1994; Mints 2006; Mitchell 1998; Prendiville 1989; Takac 1999). The incidence of infectious complications after excision of the cervical transformation zone would be much higher, if delayed vaginal bleeding (occurring after 24 hours of the procedure) and offensive discharge are also included (Chamot 2010). However, excisional procedures are often associated with prolonged vaginal discharge, which is not necessarily infective, but may be due to the healing processes and oedema secondary to the procedure.
Description of the intervention
The American College of Obstetricians and Gynecologists (ACOG) recommends single‐dose antibiotic prophylactic protocols using cephalosporin, ampicillin, gentamicin, metronidazole or antibiotics in the quinolone group prior to some major surgical procedures (ACOG 2009). However, there are no formal recommendations regarding the use of prophylactic antibiotics for LC, CKC or LLETZ, which are outpatient treatments that could be considered as contaminated procedures, since the vagina is not sterile. Therefore, the use of prophylactic antibiotics may be considered (Chamot 2010).
How the intervention might work
The excision of the transformation zone leaves a raw surface on the cervix, where vaginal flora or introduced bacteria may initiate infections. Theoretically, prophylactic antibiotics would prevent these infections by eliminating these bacteria at an early stage. Several studies have shown that prophylactic antibiotics can reduce the risk of infections among women undergoing intrauterine device insertion, surgical abortion, and repair of severe perineal tear (Buppasiri 2014; Grimes 1999; Sawaya 1996).
Why it is important to do this review
Standard clinical practice guidelines regarding the effectiveness and safety of prophylactic antibiotics for infection prevention after excision of the cervical transformation zone are lacking. A previous systematic review conducted to determine the safety of LLETZ identified that information regarding the use of prophylactic antibiotic therapy for infection prevention was inconsistently reported. Routine antibiotic therapy was described in only three out of 16 included studies (Chamot 2010). This systematic review, looking at the use of prophylactic antibiotics for the prevention of infection following the excision of the cervical transformation zone aims to evaluate the best and current evidence for the use of prophylactic antibiotics in this setting.
Objectives
To evaluate the effectiveness and safety of antibiotics for infection prevention following excision of the cervical transformation zone.
Methods
Criteria for considering studies for this review
Types of studies
We included randomised controlled trials (RCTs). We did not include quasi‐randomised trials as these may have been subjected to increased bias.
Types of participants
Women of any age undergoing excision of the cervical transformation zone for cervical intraepithelial neoplasia (CIN), irrespective of surgical techniques.
Types of interventions
Prophylactic antibiotics (irrespective of regimens) versus placebo or no treatment.
Classification of antibiotics were as follows (Marjoribanks 2004).
Cephalosporins
Penicillins
Macrolides
Fluoroquinolones
Sulfonamides
Tetracyclines
Aminogylocosides
Glycopeptides
Antiprotozoals
Combination drugs
Antibiotic regimen includes the following.
Administration route (for example intravenous, intramuscular, oral, vaginal)
Number of doses (for example single versus multiple doses)
Types of outcome measures
Primary outcomes
Infectious complications (defined as any documented sites of infection identified by cultivation or clinical symptoms and signs, or both), measured as the proportion of participants who developed each of the following within eight weeks of surgery: cervicitis, endometritis, pelvic inflammatory disease, prolonged vaginal discharge, severe vaginal bleeding, lower abdominal pain, fever, and additional antibiotic treatment.
Antibiotic resistance
Secondary outcomes
Adverse effects related to antibiotics: classified according to Common Terminology Criteria for Adverse Events (CTCAE 2010) as follows: gastrointestinal disorders (nausea, vomiting, anorexia, diarrhoea); immune system disorders (allergic reaction, anaphylaxis) blood and lymphatic system disorders (leucopenia, anaemia, thrombocytopenia, neutropenia); skin (stomatitis, mucositis, alopecia, allergy); nervous system disorders; genitourinary; and other side effects not categorised above
Unscheduled medical consultant (for any reasons).
Additional self‐medication.
As the association between vaginal bleeding, offensive vaginal discharge and secondary infection following excision of the cervical transformation zone has been observed in previous reports (Chan 2007; Doyle 1992), we chose severe vaginal bleeding and prolonged vaginal discharge as the surrogates for infectious complication. We did not include urinary tract infections and quality of life (QoL) in this review as these were not directly related to excision of the cervical transformation zone.
Search methods for identification of studies
Electronic searches
We searched the following electronic databases: Cochrane Central Register of Controlled Trials (CENTRAL; 2016, Issue 4) in the Cochrane Library, MEDLINE (1946 to May week 1, 2016), Embase (1980 to 2016, week 19), and LILACS (1970 to May, 2016).
Appendix 1, Appendix 2, Appendix 3, and Appendix 4 display the search strategies for CENTRAL, MEDLINE, Embase and LILACS, respectively.
Searching other resources
Unpublished and grey literature
We searched the following sources for ongoing trials.
ISRCTN registry, metaRegister of Controllled Trials (mRCT http://www.isrctn.com/page/mrct).
Physicians Data Query (https://www.cancer.gov/publications/pdq).
Clinical trials.gov http://www.clinicaltrials.gov.
International Clinical Trials Registry Platform (ICTRP) (http://www.who.int/ictrp/en/).
World Health Organization (WHO) International Clinical Trial Registry (http://apps.who.int/trialsearch/Default.aspx).
We searched electronic databases including Greynet.org (http:// www.greynet.org), the Ohio College Library Center (OCLC) WorldCat Dissertations and Theses (WorldCatDissertations; https://www.oclc.org/support/services/firstsearch/documentation/dbdetails/details/WorldCatDissertations.en.html) and Index to theses (ProQuest Dissertations & Theses: UK & Ireland) to identify the possible relevant conference abstracts and proceedings.
Handsearching
We handsearched reports of conferences from the following sources.
Gynecologic Oncology (Annual Meeting of the Society of Gynecologic Oncology)
International Journal of Gynecological Cancer (Annual Meeting of the International Gynecologic Cancer Society)
Annual Meeting of the European Society of Medical Oncology (ESMO)
Annual Meeting of the British Gynaecological Cancer Society (BGCS)
Biennial Meeting of the Asian Society of Gynecologic Oncology (ASGO)
Biennial Meeting of Asia and Oceania Federation of Obstetrics and Gynaecology (AOFOG)
Biennial Meeting of the European Society of Gynaecologic Oncology (ESGO)
We also checked the citation lists of the included studies and key textbooks for potentially relevant references. We searched for papers in all languages and would have had them translated, if necessary.
Data collection and analysis
Selection of studies
We downloaded all titles and abstracts retrieved by electronic searching to Endnote, and duplicates were removed. Two review authors (CK and BC) independently examined the remaining references. We excluded those studies that did not meet the inclusion criteria and obtained copies of the full text of potentially relevant references. We (CK and BC) independently assessed the eligibility of retrieved papers. We resolved disagreements by discussion and, if there was no consensus, by appeal to a third review author (JT or PL). We documented the reasons for exclusion (see Excluded studies and Characteristics of excluded studies).
Data extraction and management
We (CK and BC) abstracted data independently onto a data abstraction form specifically designed for the review. We resolved differences between review authors by discussion or by appeal to a third reviewer (JT or PL), if necessary. The same two review authors independently assessed the quality of the trials and abstracted data using forms specifically designed for the review, according to Cochrane guidelines (Higgins 2011).
Trial characteristics
Method of randomisation
Method of allocation concealment
Presence or absence of blinding of participants, clinicians and outcome assessors to treatment allocation
Number of participants randomised
Number of withdrawals (participants excluded from analysis or lost to follow‐up) and reasons
Whether an intention‐to‐treat (ITT) analysis was done
Duration, timing and location of the study
Characteristics of the study participants
Type of an excision of the cervical transformation zone undergone (either by CKC, laser conization, LEEP, LLETZ, or any other excision technique)
Inclusion criteria
Exclusion criteria
Whether the groups of participants were well balanced with regard to prognostic factors
Intervention used
Type of antibiotics used
Dose
Route
Single or multiple doses given
Duration of course of antibiotics
Number and timing of doses
Nature of comparison
No treatment, or placebo
Route of administration
Drug regimen
Outcomes
Methods used to measure genital infections
Methods used to evaluate adverse effects
Assessment of risk of bias in included studies
We assessed and reported on the methodological risk of bias of the included studies according to Chapter 8 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011), which recommends the explicit reporting of the following individual elements for RCTs.
Selection bias: random sequence generation and allocation concealment.
Performance bias: blinding of participants and personnel (participants and treatment providers).
Detection bias: blinding of outcome assessment.
Attrition bias: incomplete outcome data.
Reporting bias: selective reporting of outcomes.
Other bias
Two review authors (CK, BC) applied the ’Risk of bias’ tool independently, and we resolved differences by discussion or by appeal to a third review author (JT or PL). We judged each item as being at high, low or unclear risk of bias as set out in the criteria shown in Appendix 5 (Higgins 2011). We provided a quote from the study report or a statement as justification for our judgement. We summarised results in a ’Risk of bias’ summary figure.
Measures of treatment effect
We used the following measures of the effect of treatment.
For dichotomous data (e.g. adverse events or infections), we extracted the number of participants in each treatment arm who experienced the outcome of interest and the number of participants assessed at endpoint, in order to estimate a risk ratio (RR) with 95% confidence intervals (CIs).
For continuous data, we planned to use the mean difference (MD) or standardized mean difference (SMD) with 95% CIs.
Where possible RRs of individual studies were combined for meta‐analysis using RevMan 2014 software.
Dealing with missing data
We did not impute missing outcome data for the primary outcome and we attempted to contact study authors to obtain missing data. If data were missing to the extent that we could not have included the study in the analysis, we had planned to present the results in a narrative way.
Assessment of heterogeneity
We assessed heterogeneity using visual inspection of the forest plots. We also assessed statistical heterogeneity in each meta‐analysis using the I² statistic and Chi² test. We regarded heterogeneity as substantial if the I² statistic value was greater than 50%, or there was a low P value (< 0.10) in the Chi² test for heterogeneity (Deeks 2001; Higgins 2011). If there was substantial statistical heterogeneity, we planned to carry out subgroup analyses to assess differences between the included studies. However, if there had been clinical, methodological or considerable statistical heterogeneity (I² greater than 75%) across included studies, we had planned to use a narrative approach to data synthesis.
Assessment of reporting biases
We had planned to examine funnel plots corresponding to meta‐analysis of the primary outcome to assess the potential for small‐study effects, such as publication bias, if more than 10 studies were included. If asymmetry had been suggested by a visual assessment, we had planned to perform sensitivity analysis to investigate whether it affected the pooled results. (Sterne 2011).
Data synthesis
Where feasible, the results were pooled in a meta‐analyses. We used the random‐effects model with inverse variance weighting for all meta‐analyses (DerSimonian 1986). We performed statistical analysis using RevMan 2014.
For dichotomous outcomes, we calculated the RR for each study as well as for the pooled outcome.
For continuous outcomes, we calculated and pooled the mean difference between the treatment arms if all trials measured the outcome on the same scale; otherwise we pooled the standardised mean differences.
We prepared a ’Summary of findings’ table to present the results of the meta‐analysis, based on the methods described in Chapter 11 of the Cochrane Handbook for Systematic Reviews of Interventions (Schunemann 2011). For assessments of the overall certainty of evidence for each outcome that includes pooled data from RCTs only, we downgraded the evidence from 'high certainty' by one level for serious (or by two for very serious) study limitations (risk of bias), indirectness of evidence, serious inconsistency, imprecision of effect estimates or potential publication bias. The following outcomes were included in the 'Summary of findings' table:
prolonged vaginal discharge;
postoperative bleeding;
fever;
abdominal pain;
adverse effect related to antibiotic;
additional medical support;
additional self‐medication.
We presented the results of the meta‐analysis for the relevant outcomes and adverse events as outlined in the ’Types of outcome measures’ section.
Subgroup analysis and investigation of heterogeneity
As we found only a few trials, we did not perform any subgroup analyses. However, we considered factors such as type of excisional techniques (e.g. CKC, LC, or LLETZ/LEEP) and antibiotic regimen prescribed in the interpretation of review findings. In future updates, we will perform subgroup analysis based on these factors, if feasible.
Sensitivity analysis
As we found only three trials, we did not perform any sensitivity analysis. In future updates, if statistical heterogeneity is detected and there are sufficient trials included (greater than 10), we will conduct sensitivity analyses to examine the possible contribution of other clinical or methodological differences between the trials, specifically:
trials with adequate methodology versus those with poor methodology;
trials which seem to differ from the others in their clinical criteria for defining genital infections.
Results
Description of studies
Results of the search
We ran a broad search that yielded 390 references from the combined searches. We checked the reference lists and handsearched journals and congress abstracts, which identified two additional references. We did not identify any ongoing trials. After de‐duplication, we screened 370 references and excluded 364 references that obviously did not meet the inclusion criteria. Of the six studies that potentially met the review inclusion, we excluded three studies after reviewing the full texts (Doyle 1992; Gerli 2012; Minorchio 1990) (see Excluded studies and Characteristics of excluded studies). Figure 1 displays the PRISMA flowchart for study selection.
1.
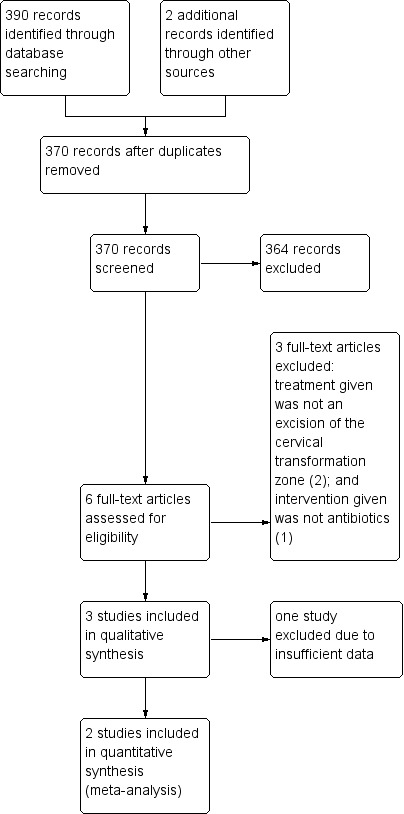
Study flow diagram.
Included studies
We found three studies that met the inclusion criteria (Chan 2007; Foden‐Shroff 1998; Gornall 1999). See 'Characteristics of included studies' for details of each included study.
Foden‐Shroff 1998 was conducted in UK from July 1994 to August 1996. Participants enrolled in the antibiotic group received ofloxacin 400 mg, once daily for five consecutive days, starting immediately after the procedure. Participants assigned to the control group received an identical placebo. All participants received a pictorial chart to record the amount of vaginal discharge and adverse events for two weeks after the procedure. Reported outcomes included: prevalence of prolonged vaginal discharge; postoperative vaginal bleeding requiring hospitalisation; and adverse events. From the 500 participants recruited, only 348 participants' charts were suitable for analyses (70% of total group).
Gornall 1999 was conducted in the UK. The study period was not reported. The participants allocated to the intervention group received Sultrin®, which is antimicrobial vaginal pessary containing sulphatiazole 3.4%, sulphacetamide 2.8%, and sulphabenzamide 3.7%, twice daily for five days. Participants assigned to the control group received no treatment. All participants received patient charts to record of their symptoms and requirement of unscheduled medical visits and additional antibiotic treatment, if any during the first four weeks after the procedure. Study outcomes were: (1) severity of vaginal bleeding, discharge, and pain; and (2) the unscheduled medical visits and additional antibiotic therapy. From the 100 participants recruited, 77 participants' charts were available for analyses, corresponding to a rate of 77%.
Chan 2007 was conducted in Hong Kong between May 2003 and August 2006. Of 321 participants complying with inclusion/exclusion criteria; 157 were randomly allocated to receive antibiotics and 164 who were assigned to the control group did not receive any medication. The intervention was an antimicrobial vaginal pessary containing 100 mg tetracycline and 50 mg amphotericin B (Talsutin®), given twice daily for 14 days, starting on the day of large loop excision of the transformation zone (LLETZ). All participants received a diary to record the daily amount of vaginal bleeding, vaginal discharge, and lower abdominal pain for three weeks after the procedure. Study outcomes were: (1) prevalence of vaginal bleeding that required admission to hospital, additional medical consultation, additional self‐medication, fever; (2) severity of vaginal bleeding, pain, and lower abdominal pain. The authors excluded 23 participants (12 in antibiotic group and 11 in control group) because they did not return the diary. In addition, the authors excluded eight participants assigned to antibiotic group due to non‐compliance with treatment, leaving 290 charts for final analyses (approximately 90% of total group).
See 'Characteristics of included studies' for full details of the included studies. Although all three included studies had been published in peer‐reviewed journals at the time of the search and data extraction, numerical data regarding the outcome measured in Gornall 1999 was insufficient for meta‐analyses. Attempts to obtain additional data from the investigators were not successful, since the authors did not reply to our inquiries. We therefore did not pool the results of this study and instead presented them in a narrative format.
Excluded studies
After excluding non‐relevant and duplicated records, we retrieved six possibly eligible studies for more detailed evaluation. Of the six potentially eligible studies that we assessed in full‐text format, we excluded three references for the following reasons (see 'Characteristics of excluded studies). One trial was excluded because it was a non‐randomised study. In addition, the treatment used in this study was cervical ablation, rather than excision, so it did not meet the review inclusion criteria (Minorchio 1990). Another reference was a randomised study evaluating effectiveness and safety of two preparations of vaginal antiseptic suppositories after cervical ablation using CO2 laser, thus it did not comply to the review inclusion criteria (Gerli 2012). The third was a randomised study, but it was excluded because the intervention that was evaluated in this study was a Monsel’s solution, rather than prophylactic antibiotics (Doyle 1992).
Risk of bias in included studies
Allocation
In two included studies (Chan 2007; Foden‐Shroff 1998), the participants were randomly allocated to the comparison groups with adequate allocation concealment. We therefore determined both trials as having low risk of bias. The other included study (Gornall 1999) used sealed envelopes for treatment allocation, but no details of randomisation were given. We determined this to indicate unclear risk of bias for random sequence generation and low risk of bias for allocation concealment (Figure 2).
2.

'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study.
Blinding
In Foden‐Shroff 1998, the authors provided the study medications containing either antibiotics or placebo with the same appearance and packaging. We determined this to indicate low risk of bias
In the remaining two included studies (Chan 2007; Gornall 1999), participants assigned to the control group did not have a placebo control, so they were aware about the treatment allocated. We therefore judged both trials as having high risk of bias (Figure 2).
Incomplete outcome data
In the studies of Foden‐Shroff 1998 and Gornall 1999 the rates of incomplete data were high (30% and 23%, respectively). Therefore we judged both studies as having high risk of bias. In Chan 2007 90% of participants had complete data, indicating low risk of bias (Figure 2).
Selective reporting
Although the study protocols of included studies were not available (Chan 2007; Foden‐Shroff 1998; Gornall 1999), the authors did report the expected relevant outcomes, so we judged this domain to be at a low risk of bias (Figure 2).
Other potential sources of bias
The analyses performed in all three included studies (Chan 2007; Foden‐Shroff 1998; Gornall 1999) did not follow an intention‐to‐treat basis. Additionally, in Gornall 1999, there were no data regarding the review inclusion and exclusion criteria applied. The authors also did not provide the data regarding the baseline characteristics of participants and number of participants allocated to each comparison group, which are mandatory for determining treatment outcomes and assessing the quality of study methodology. So, we deemed this domain to have a high risk of bias (Figure 2).
Effects of interventions
See: Table 1
See: Differences between protocol and review
Primary outcomes
Infectious complications
Prolonged vaginal discharge
Only Foden‐Shroff 1998 reported the incidence of prolonged vaginal discharge after cervical excision in the two comparison groups. Participants receiving prophylactic antibiotics had a higher incidence of prolonged vaginal discharge after LLETZ compared to those who were assigned to placebo group (13.3% versus 10.3%, respectively; Table 2). However, there was no statistical difference (one study; 348 participants; risk ratio (RR), 1.29; 95% confidence interval (CI) 0.72 to 2.31; low‐quality evidence). See Table 1; Analysis 1.1.
1. Extracted data from Chan 2007 and Foden‐Shroff 1998.
| Study | Antibiotic group | Placebo or no treatment |
| Number of participants who experienced prolonged vaginal discharge | ||
| Foden‐Shroff 1998 | 23/173 (13.3%) | 18/175 (10.3%) |
| Number of participants who had to be admitted for post‐procedure bleeding | ||
| Foden‐Shroff 1998 | 2/173 (1.2%) | 3/175 (1.7%) |
| Chan 2007 | 9/137 (6.6%) | 7/153 (4.6%) |
| Number of participants who experienced lower abdominal pain | ||
| Chan 2007 | 23/137 (16.8%%) | 25/153 (16.3%) |
| Number of participants who developed fever | ||
| Chan 2007 | 2/137 (1.5%) | 1/153 (0.7%) |
| Number of participants who experienced adverse events | ||
| Foden‐Shroff 1998 | 20/173 (11.6%) | 12/175 (6.9%) |
| Chan 2007 | 0/137 (0%) | 0/153 (0%) |
| Number of participants who required unscheduled medical consultation | ||
| Chan 2007 | 12/137 (8.8%) | 5/153 (3.3%) |
| Number of participants reported to have additional self‐medication | ||
| Chan 2007 | 12/137 (8.8%) | 11/153 (7.2%) |
1.1. Analysis.
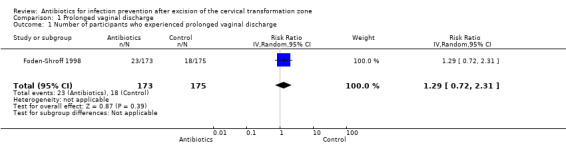
Comparison 1 Prolonged vaginal discharge, Outcome 1 Number of participants who experienced prolonged vaginal discharge.
Severe vaginal bleeding
All included studies reported the incidence of severe vaginal bleeding requiring hospital admission. However, numerical data regarding this outcome measured in the study Gornall 1999 was insufficient for meta‐analyses. In Foden‐Shroff 1998, only 1.2% of participants assigned to the antibiotic group experienced severe vaginal bleeding requiring hospitalisation, which was comparable to the 1.7% reported for participants in the placebo group (Table 2). In Chan 2007, participants receiving prophylactic antibiotics had a higher incidence of severe vaginal bleeding requiring hospitalisation compared to those who were assigned to placebo group (6.6% versus 4.6%, respectively; Table 2). After combining data from Foden‐Shroff 1998 and Chan 2007, there was no difference (638 participants; RR 1.21; 95% CI 0.52 to 2.82; very low‐quality evidence). See Table 1; Analysis 2.1.
2.1. Analysis.
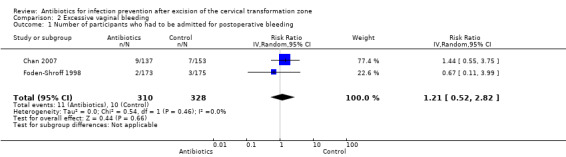
Comparison 2 Excessive vaginal bleeding, Outcome 1 Number of participants who had to be admitted for postoperative bleeding.
In Gornall 1999, of the 77 participants, two assigned to the control group were admitted because of excessive vaginal bleeding. None of the participants who received antibiotics had severe vaginal bleeding (Table 3).
2. Reported data from Gornall 1999.
| Outcomes reported in Gornall 1999 | Sultrin (sample size = n) | Control (sample size = 77‐n) | 95% confidence interval | P value |
| Mean duration of bleeding (days) | 15.2 | 11.2 | ‐7.7 to ‐0.2 | 0.04 |
| Mean duration of discharge (days) | 16.4 | 13.1 | ‐7.2 to 0.7 | 0.11 |
| Mean duration of pain (days) | 7.7 | 5.7 | ‐5.4 to 1.5 | 0.26 |
| Number of participants who received additional antibiotic therapy | 2 | 7 | Not reported | Not reported |
| Number of participants who had to be admitted for postoperative bleeding | 0 | 2 | Not reported | Not reported |
Numbers of the participant in each comparison group were not reported.
Fever
Only Chan 2007 reported the incidence of fever following excision of the cervical transformation zone and demonstrated no difference between the groups (one study; 290 participants; RR, 2.23; 95% CI 0.20 to 24.36; very low‐quality of evidence). See Table 1: Analysis 3.1.
3.1. Analysis.
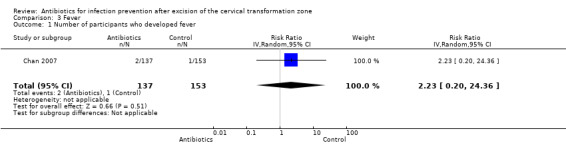
Comparison 3 Fever, Outcome 1 Number of participants who developed fever.
Lower abdominal pain
The incidence of postoperative lower abdominal pain was reported in one of three included studies (Chan 2007). Approximately 16.8% of participants assigned to the antibiotic group experienced lower abdominal pain following LLETZ, which was comparable to the 16.3% reported for participants in the control group (Table 2) (one study; 290 participants; RR, 1.03; 95% CI 0.61 to 1.72: low‐quality evidence). See Table 1; Analysis 4.1.
4.1. Analysis.
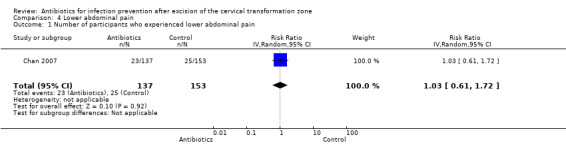
Comparison 4 Lower abdominal pain, Outcome 1 Number of participants who experienced lower abdominal pain.
Additional antibiotic treatment
Gornall 1999 reported that, of 77 participants available for the study analyses, two and seven participants assigned to the antibiotic and control group, respectively, received additional antibiotics. Because of missing data regarding the number of participants in each comparison group, the incidence of the requirement of additional antibiotic treatment among the two comparison groups and the relative effect of prophylactic antibiotics, could not be determined (Table 3). Foden‐Shroff 1998 and Chan 2007 did not report the requirement of additional antibiotic treatment in their studies.
Other infectious complications
The included studies did not report the incidence of cervicitis, endometritis, and pelvic inflammatory disease.
Antibiotic resistance
No information on antibiotic resistance was reported in any of the included studies.
Secondary outcomes
Adverse effects
No reported data on the rate of adverse events were available for Gornall 1999 (Table 3). In Chan 2007, no participants reported antibiotic adverse events (Table 2). Foden‐Shroff 1998 reported that 11.6% of participants who received antibiotic experienced adverse events, including nausea, vomiting, diarrhoea, and headache, which was higher than the 6.9% reported in participants assigned to placebo group. When we pooled the data from these two included studies (Chan 2007; Foden‐Shroff 1998), no difference was demonstrated (638 participants; RR 1.69; 95% CI 0.85 to 3.34; very low‐quality evidence). See Table 1: Analysis 5.1.
5.1. Analysis.
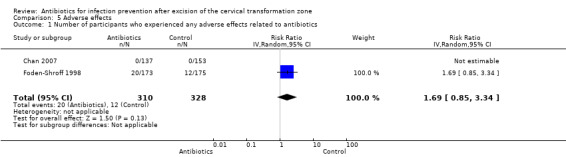
Comparison 5 Adverse effects, Outcome 1 Number of participants who experienced any adverse effects related to antibiotics.
Unscheduled medical consultation
Only Chan 2007 reported the rate of unscheduled medical consultation in the two comparison groups. Participants receiving antimicrobial vaginal pessary had a higher rate of unscheduled medical consultation than those who were assigned to the control arm (8.8% versus 3.3%, respectively), but this difference was not significant (one study, 290 participants; RR 2.68, 95% CI 0.97 to 7.41; low‐quality evidence). See Table 1; Analysis 6.1.
6.1. Analysis.
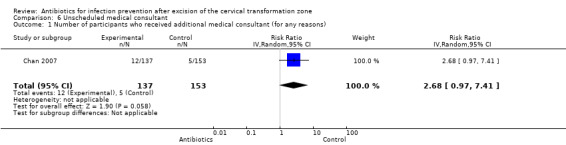
Comparison 6 Unscheduled medical consultant, Outcome 1 Number of participants who received additional medical consultant (for any reasons).
Additional self‐medication
The rate of additional self‐medication was reported in one study (Chan 2007). Approximately 8.8% of participants assigned to antibiotic group reported to have self‐medication which was comparable to the 7.2% reported for participants in the control group (one study; 290 participants; RR 1.22; 95%CI 0.56 to 2.67; very low‐quality evidence; Table 1; Analysis 7.1).
7.1. Analysis.
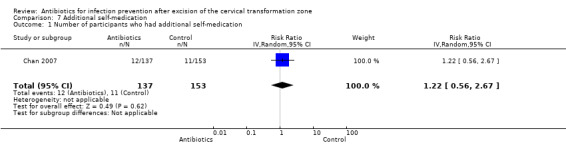
Comparison 7 Additional self‐medication, Outcome 1 Number of participants who had additional self‐medication.
Discussion
Summary of main results
We found three studies that met our inclusion criteria. However, numerical data regarding the outcome measured in one small study was insufficient for inclusion in the meta‐analyses. Evidence from this review indicates that there were no differences between women receiving prophylactic antibiotics and those receiving a placebo or no treatment in terms of prolonged vaginal discharge, severe vaginal bleeding, fever, lower abdominal pain, requirement of unscheduled medical consultation, and self‐medication. In addition, the number of adverse events did not differ between the two comparison groups. None of the three included studies reported data on antibiotic resistance. However, it is unknown whether these symptoms were due to infection, since samples for microbiological culture were not routinely taken in these studies and symptoms were largely self‐reported.
Overall completeness and applicability of evidence
This review included three randomised controlled trials (RCTs) evaluating prophylactic antibiotic in 708 participants who had undergone large loop excision of the transformation zone (LLETZ) procedure in the UK and Hong Kong. The interventions evaluated in the included studies were different antibiotic regimens to prevent infection following excision of the cervical transformation zone. Primary outcomes of this review were signs and symptoms suggesting infectious complications including: cervicitis; endometritis; pelvic inflammatory disease; prolonged vaginal discharge; severe vaginal bleeding; lower abdominal pain; fever; and additional antibiotic treatment received. Secondary outcomes included adverse events; antibiotic resistance; and incidence of unscheduled medical consultation and self‐medication. However, the included studies did not report the incidence of cervicitis, endometritis, pelvic inflammatory disease, and antibiotic resistance.
It is important to note that the excisional method used in all three included studies was LLETZ. Therefore, the same results may not be applicable to other excisional techniques. In addition, the two studies included in the review that tested drugs targeting Chlamydia trachomatis took place in a population with a prevalence of chlamydial infection that varied from 2% to 10% (Chan 2007; Foden‐Shroff 1998). The other antimicrobial used was vaginal pessary, which was a preparation used against Haemophilus (Garnerella) vaginalis (Gornall 1999).Therefore, generalisation of the results to different settings and different antibiotics or a combination of antibiotics may be limited.
Quality of the evidence
The greatest threat to the validity of the included study is likely to be the risk of bias (see Figure 2). In all included studies, treatment outcomes were measured using self‐reported data from participants. However, participants assigned to the control groups of two included studies did not receive a placebo. Thus, these participants were unblinded for treatment allocated, resulting in a high risk of performance and detection biases (Chan 2007; Gornall 1999). Another limitation for this review was the potential risk of attrition bias. Lack of data for final analyses were considerable, greater than 20% in two included studies (Foden‐Shroff 1998; Gornall 1999). In addition, analyses carried out in all included studies did not follow an intention‐to‐treat principle. Data from one study involving 77 participants were also limited. The authors of this study did not provide additional information. Baseline characteristics of participants, and number of participants allocated to each comparison group, which are necessary for assessing the effects of treatment and quality of study methodology, were not available (Gornall 1999).
Another major limitation of this review is the small number of included studies, which has the potential to affect the accuracy in determining statistical heterogeneity and thus we used random‐effects model for all meta‐analyses.
We assessed the quality of evidence using the GRADE approach for each outcome (see Table 1). Based on the concerns regarding the risk of the potential bias, we downgraded the evidence to low quality for the incidences of prolonged vaginal discharge, lower abdominal pain, unscheduled medical consultant, and self‐medication. We downgraded the evidence to very low quality for severe vaginal bleeding, postoperative fever, and adverse events due to the potential biases and sparseness of data. We found no available evidence on the potential risk of antibiotic resistance, one of the most serious health threats, following the use of antibiotic prophylaxis for excision of the cervical transformation zone.
Potential biases in the review process
With assistance from the Information Specialist, Cochrane Gynaecological, Neuro‐oncology & Orphan Cancer Group, we made every attempt to include global studies including a thorough search of the grey literature, conference proceedings, and ongoing trials. However, as there were only three studies that met the review inclusion criteria, there remains the possibility that there may be other unpublished trials of intervention that the review authors did not discover. This means that the review authors may unwittingly have perpetuated a publication bias. Another source of potential biases is our inability to obtain relevant incomplete data leading to the exclusion of one small study (Gornall 1999) from the meta‐analysis.
None of the review authors have any links to drug companies or a financial interest in the prescription of the drug under assessment, nor were they involved in the conduct of the included study. Thus, there were no issues associated to bias secondary to conflicts of interests in this review.
Agreements and disagreements with other studies or reviews
According to the surgical wound classification system, an excision of the cervical transformation zone is considered as a clean‐contaminated procedure. Although ACOG (ACOG 2009) and the National Institute for Health and Care Excellence (NICE 2008) generally recommend prophylactic antibiotics for clean‐contaminated procedures, findings from this review show that there is not sufficient evidence to support routine antibiotics for infection prevention after LLETZ procedure. This conclusion is in broad agreement with previously published reviews (Craciunas 2014; Morrill 2013).
Antibiotic resistance is one of the biggest threats to global health and requires urgent, co‐ordinated action across all healthcare sectors (Del Mar 2012; WHO 2015).The misuse of antibiotics accelerates the emergence of drug‐resistant bacteria (WHO 2015). As noted in previous reports in a variety of clinical settings, administration of antibiotic prophylaxis can be a risk factor for antibiotic resistance (Bitsori 2014; McMurray 2015; Minami 2014). Therefore, the potential benefits and possible harms of prophylactic antibiotics should be carefully assessed and considered, particularly for the risk of developing antibiotic resistance. However, there has been little discussion of potential risk of antibiotic resistance after administration of prophylactic antibiotics in previous reports (Buppasiri 2014; Grimes 1999; Morrill 2013; Sawaya 1996). In this review, none of the included studies reported data on antibiotic resistance.
Authors' conclusions
Implications for practice.
The limited evidence obtained from the three randomised controlled trials (RCTs) presented in this review found no benefit of antibiotics given to prevent infection following excision of the cervical transformation zone for cervical neoplasia. In addition, evidence for the safety of antibiotics used in this surgical setting is inconclusive, due to a small number of reported adverse events, incomplete data available for analyses, and lack of information on antibiotic resistance. The evidence does not support the routine administration of prophylactic antibiotics in women undergoing excision of the cervical transformation zone. At the present, antibiotics given for infection prevention after excision of the cervical transformation zone should be limited to appropriately conducted clinical research.
Implications for research.
The effectiveness and safety of antibiotics for infection prevention following excision of the cervical transformation zone were inadequately assessed in the included studies according to an intention‐to‐treat basis. Additionally, the trials in all of the comparisons were at high or moderate risk of bias, so the quality of the evidence was low or very low for these outcomes across all comparisons. Further research therefore is likely to have an important impact on our confidence in the estimates of effect and may alter the estimates in the treatment comparisons. However, this would be dependent on the studies being of high quality, adequately sized, placebo‐controlled, with a well‐defined protocol for appropriate outcomes measured and statistical analyses to delineate the effectiveness and safety of antibiotics. Any studies should include an assessment on the effect of antibiotic resistance.
What's new
| Date | Event | Description |
|---|---|---|
| 7 October 2019 | Amended | Most recent search 2 October 2019. No potentially relevant new studies identified. The conclusions of this Cochrane Review are therefore still considered up to date for this topic. A further search of the literature will be carried out in 2022. |
Acknowledgements
We thank Gail Quinn, Clare Jess and Tracey Harrison for their rigorous contribution to the editorial process; Jo Platt and Jane Hayes for designing the search strategies and running the search; and Jo Morrison for clinical and editorial advice. We thank the referees for many helpful suggestions and comments, some of these include Khadra Galaal and Jennifer Cove.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure to the Cochrane Gynaecological, Neuro‐oncology and Orphan Cancer Group. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Appendices
Appendix 1. CENTRAL search strategy
#1 MeSH descriptor Antibiotic Prophylaxis, this term only #2 MeSH descriptor Anti‐Infective Agents, this term only #3 MeSH descriptor Cephalosporins explode all trees #4 MeSH descriptor Penicillins explode all trees #5 MeSH descriptor Macrolides explode all trees #6 MeSH descriptor Fluoroquinolones explode all trees #7 MeSH descriptor Sulfonamides explode all trees #8 MeSH descriptor Tetracyclines explode all trees #9 MeSH descriptor Aminoglycosides explode all trees #10 MeSH descriptor Glycopeptides explode all trees #11 MeSH descriptor Antiprotozoal Agents explode all trees #12 (antibiotic* or antimicrob* or anti‐microb* or antibacteria* or anti‐bacteria* or antiinfect* or anti‐infect*) #13 ((prevent* or prophyla*) near/5 (bacteria* or microb* or infect*)) #14 (cef* or ceph* or loracarbef* or penicillin* or amoxicillin or erythromycin or clarithromycin or azithromycin or metronidazole or ciprofloxacin or levofloxacin or ofloxacin or co‐trimoxazole or cotrimoxazole or trimethoprim or tetracycline or doxycycline or gentamycin or gentamicin or vancomycin or augmentin) #15 (#1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14) #16 MeSH descriptor Uterine Cervical Neoplasms, this term only #17 MeSH descriptor Cervical Intraepithelial Neoplasia, this term only #18 MeSH descriptor Cervix Uteri, this term only #19 cervi* #20 (#16 OR #17 OR #18 OR #19) #21 Any MeSH descriptor with qualifier: SU #22 (surg* or excis* or laser* or conization) #23 MeSH descriptor Gynecologic Surgical Procedures explode all trees #24 (LEEP or LLETZ or LC or CKC) #25 (#21 OR #22 OR #23 OR #24) #26 (#15 AND #20 AND #25)
Appendix 2. MEDLINE search strategy
Medline (Ovid)
1 Antibiotic Prophylaxis/ 2 exp Anti‐Infective Agents/ 3 exp Cephalosporins/ 4 exp Penicillins/ 5 exp Macrolides/ 6 exp Fluoroquinolones/ 7 exp Sulfonamides/ 8 exp Tetracyclines/ 9 exp Aminoglycosides/ 10 exp Glycopeptides/ 11 exp Antiprotozoal Agents/ 12 (antibiotic* or antimicrob* or anti‐microb* or antibacteria* or anti‐bacteria* or antiinfect* or anti‐infect*).mp. 13 ((prevent* or prophyla*) adj5 (bacteria* or microb* or infect*)).mp. 14 (cef* or ceph* or loracarbef* or penicillin* or amoxicillin or erythromycin or clarithromycin or azithromycin or metronidazole or ciprofloxacin or levofloxacin or ofloxacin or co‐trimoxazole or cotrimoxazole or trimethoprim or tetracycline or doxycycline or gentamycin or gentamicin or vancomycin or augmentin).mp. 15 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 16 Uterine Cervical Neoplasms/ 17 Cervical Intraepithelial Neoplasia/ 18 Cervix Uteri/ 19 cervi*.mp. 20 16 or 17 or 18 or 19 21 surgery.fs. 22 (surg* or excis* or laser* or conization).mp. 23 exp Gynecologic Surgical Procedures/ 24 (LEEP or LLETZ or LC or CKC).mp. 25 21 or 22 or 23 or 24 26 15 and 20 and 25 27 randomized controlled trial.pt. 28 controlled clinical trial.pt. 29 randomized.ab. 30 placebo.ab. 31 drug therapy.fs. 32 randomly.ab. 33 trial.ab. 34 groups.ab. 35 27 or 28 or 29 or 30 or 31 or 32 or 33 or 34 36 26 and 35 37 exp animals/ not humans.sh. 38 36 not 37
key: mp=title, original title, abstract, name of substance word, subject heading word, unique identifier pt=publication type fs=floating subheading sh=subject heading
Appendix 3. Embase search strategy
Embase (Ovid)
1 antibiotic prophylaxis/ 2 exp antiinfective agent/ 3 exp cephalosporin derivative/ 4 exp penicillin derivative/ 5 exp macrolide/ 6 exp quinolone derivative/ 7 exp sulfonamide/ 8 exp tetracycline derivative/ 9 exp aminoglycoside/ 10 exp glycopeptide/ 11 exp antiprotozoal agent/ 12 (antibiotic* or antimicrob* or anti‐microb* or antibacteria* or anti‐bacteria* or antiinfect* or anti‐infect*).mp. 13 ((prevent* or prophyla*) adj5 (bacteria* or microb* or infect*)).mp. 14 (cef* or ceph* or loracarbef* or penicillin* or amoxicillin or erythromycin or clarithromycin or azithromycin or metronidazole or ciprofloxacin or levofloxacin or ofloxacin or co‐trimoxazole or cotrimoxazole or trimethoprim or tetracycline or doxycycline or gentamycin or gentamicin or vancomycin or augmentin).mp. 15 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 16 exp uterine cervix tumor/ 17 uterine cervix carcinoma in situ/ 18 exp uterine cervix/ 19 cervi*.mp. 20 16 or 17 or 18 or 19 21 su.fs. 22 (surg* or excis* or laser* or conization).mp. 23 exp gynecologic surgery/ 24 (LEEP or LLETZ or LC or CKC).mp. 25 21 or 22 or 23 or 24 26 15 and 20 and 25 27 crossover procedure/ 28 double‐blind procedure/ 29 randomized controlled trial/ 30 single‐blind procedure/ 31 random*.mp. 32 factorial*.mp. 33 (crossover* or cross over* or cross‐over*).mp. 34 placebo*.mp. 35 (double* adj blind*).mp. 36 (singl* adj blind*).mp. 37 assign*.mp. 38 allocat*.mp. 39 volunteer*.mp. 40 27 or 28 or 29 or 30 or 31 or 32 or 33 or 34 or 35 or 36 or 37 or 38 or 39 41 26 and 40 42 (exp Animal/ or Nonhuman/ or exp Animal Experiment/) not Human/ 43 41 not 42
key: [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword]
Appendix 4. LILACS search strategy
1 Antibiotic OR Anti‐bacterial agents/ 2 cephalosporins 3 penicillins 4 macrolides 5 fluoroquinolones 6 sulfonamides 7 aminogylocosides 8 glycopeptides 9 antiprotozoals 10 erythromycin 11 clarithromycin 12 metronidazole 13 azithromycin 14 methronidazole 15 ciprofloxacin 16 levofloxacin 17 ofloxacin 18 trimethoprim 19 tetracycycline 20 doxycycline 21 vancomycin 22 augmentin 23 #1 or # 2 or #3 or #4 or #5 or #6 or #7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20 or #21 or #22 24 infection or prevention 25 cervical intraepithelial surgery or cervix uteri surgery 26 randomized or clinical trials or controlled or trial 27 #23 and #24 and #25 and #26
Appendix 5. 'Risk of bias' assessment
Risk of bias assessment based on chapter 8 of Higgins 2011; • Random sequence generation i) Low risk of bias e.g. participants assigned to treatments on the basis of a computer‐generated random sequence or a table of random numbers ii) High risk of bias e.g. participants assigned to treatments on the basis of date of birth, clinic id‐number or surname, or no attempt to randomise participants iii) Unclear risk of bias e.g. not reported, information not available • Allocation concealment i) Low risk of bias e.g. where the allocation sequence could not be foretold ii) High risk of bias e.g. allocation sequence could be foretold by patients, investigators or treatment providers iii) Unclear risk of bias e.g. not reported. • Blinding of participants and personnel i) Low risk of bias if participants and personnel were adequately blinded ii) High risk of bias if participants were not blinded to the intervention that the participant received iii) Unclear risk of bias if this was not reported or unclear • Blinding of outcomes assessors i) Low risk of bias if outcome assessors were adequately blinded ii) High risk of bias if outcome assessors were not blinded to the intervention that the participant received iii) Unclear risk of bias if this was not reported or is unclear • Incomplete outcome data: we will record the proportion of participants whose outcomes were not reported at the end of the study. We will code a satisfactory level of loss to follow‐up for each outcome as: i) Low risk of bias e.g.if fewer than 20% of participants were lost to follow‐up and reasons for loss to follow‐up were similar in both treatment arms ii) High risk of bias e.g. if more than 20% of participants were lost to follow‐up or reasons for loss to follow‐up differed between treatment arms iii) Unclear risk of bias e.g. if loss to follow‐up was not reported • Selective reporting of outcomes i) Low risk of bias e.g. review reports all outcomes specified in the protocol ii) High risk of bias e.g. the study is suspected that outcomes have been selectively reported iii) Unclear risk of bias e.g. it is unclear whether outcomes have been selectively reported • Other bias i) Low risk of bias e.g. the review authors do not suspect any other source of bias and the trial appears to be methodologically sound ii) High risk of bias e.g. the review authors suspect that the trial was prone to an additional bias iii) Unclear risk of bias e.g. the review authors are uncertain whether an additional bias may have been present
Data and analyses
Comparison 1. Prolonged vaginal discharge.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who experienced prolonged vaginal discharge | 1 | 348 | Risk Ratio (IV, Random, 95% CI) | 1.29 [0.72, 2.31] |
Comparison 2. Excessive vaginal bleeding.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who had to be admitted for postoperative bleeding | 2 | 638 | Risk Ratio (IV, Random, 95% CI) | 1.21 [0.52, 2.82] |
Comparison 3. Fever.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who developed fever | 1 | 290 | Risk Ratio (IV, Random, 95% CI) | 2.23 [0.20, 24.36] |
Comparison 4. Lower abdominal pain.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who experienced lower abdominal pain | 1 | 290 | Risk Ratio (IV, Random, 95% CI) | 1.03 [0.61, 1.72] |
Comparison 5. Adverse effects.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who experienced any adverse effects related to antibiotics | 2 | 638 | Risk Ratio (IV, Random, 95% CI) | 1.69 [0.85, 3.34] |
Comparison 6. Unscheduled medical consultant.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who received additional medical consultant (for any reasons) | 1 | 290 | Risk Ratio (IV, Random, 95% CI) | 2.68 [0.97, 7.41] |
Comparison 7. Additional self‐medication.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants who had additional self‐medication | 1 | 290 | Risk Ratio (IV, Random, 95% CI) | 1.22 [0.56, 2.67] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Chan 2007.
| Methods | A randomised‐controlled trial conducted in the colposcopy clinic at Queen Mary Hospital, University of Hong Kong. Study duration: May 2003 to August 2006. |
|
| Participants | All women who attended the clinic for LLETZ. Exclusion criteria: history of antibiotic hypersensitivity, liver or renal disease, previous psychiatric problems or receiving antibiotics within the 2 weeks prior to the visit. All participants receive a diary to record the daily amount of vaginal bleeding, vaginal discharge, and lower abdominal pain for 3 weeks after the procedure Of 321 participants complying with inclusion/exclusion criteria; 157 were randomly allocated to receive antibiotics and 164 did not receive any medications. Mean age of participants in antibiotic and those in control groups were 38.4 years and 40.5 years, respectively. Approximately 57.7% and 61.4% of participants assigned to antibiotic and control groups, respectively, had positive endocervical swabs. Approximately 27.0% and 34.8% of participants assigned to antibiotic and control groups, respectively, had positive high vaginal swabs. Approximately 4.4% and 2.0% of participants assigned to antibiotic and control groups, respectively, had positive test for Chlamydia. Weight of excised specimens obtained among the participants allocated to antibiotic and control groups, respectively, was 2.2 g and 1.9 g. Approximately 67.2% and 63.4% of participants assigned to antibiotic and control groups, respectively, had high‐grade lesion on conization specimens. |
|
| Interventions | Antimicrobial vaginal pessary containing 100 mg tetracycline and 50 mg amphotericin B (Talsutin®, Bristol‐Myers Squibb New York, NY, USA) two times a day for 14 days, starting on the day of LLETZ. The control group did not receive any medications. Tetracycline is mainly an anti‐chlamydial agent. Amphotericin B is an antifungal antibiotic. | |
| Outcomes | The primary outcome was the incidence of post‐LLETZ bleeding that required medical attention. The secondary outcomes were the severity of postoperative vaginal bleeding, vaginal discharge, and lower abdominal pain in the 3 weeks after the procedure recorded in the participants' diary. All participants were assessed about the complications at 3 weeks after LLETZ. |
|
| Notes | The authors excluded 23 participants (12 in antibiotic group and 11 in control group) because they did not return the diary. In addition, the authors excluded 8 participants assigned to antibiotic group because of incomplete the course of treatment prescribed, leaving 290 charts for final analyses (approximately 90% of total group). Therefore, the analyses performed in this study did not follow an intention‐to‐treat basis. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | This trial used block randomisation with a randomised size of four. This study declared that "generation of randomisation schedule was performed by a person independent of the recruitment, and the seed from which the randomisation schedule was generated was kept securely by the randomiser." |
| Allocation concealment (selection bias) | Low risk | A quote from the study: "Sealed opaque envelopes containing the randomised treatment allocation was prepared and kept by the research assistant prior to the start of patient recruitment" |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | A quote from the study: "The result of the randomisation was blinded to the research assistant but not to the colposcopist who needed to prescribe the medication for the treatment group at the clinic immediately after the procedure". In addition, the authors did not use placebo for the participant assigned to the control group so the participants were not blinded to treatment allocated." |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | The study outcomes were based on the participants' self‐assessment on their symptoms after the procedure. As participants were aware about the treatment received, there is high risk of detection bias. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Twenty‐three participants did not return the diary (7.2%). |
| Selective reporting (reporting bias) | Low risk | All potential relevant outcomes were reported. |
| Other bias | High risk | This study excluded 23 participants due to not return their diary (12 in antibiotic group and 11 in control group) and 8 participants in the treatment group from the final analyses because of incomplete the course of treatment. So, the analyses performed in this study was not based on intention‐to‐treat. |
Foden‐Shroff 1998.
| Methods | A randomised double‐blind, placebo‐controlled study conducted in a large teaching hospital colposcopy clinic in UK. Study duration: July 1994 to August 1996 |
|
| Participants | The participants were women aged between 20 and 65 undergoing loop diathermy excision. Exclusion criteria: a history of antibiotic hypersensitivity, renal or hepatic diseases, previous psychiatric problems, and receiving antibiotic within the preceding 14 days. All participants received pictorial chart to record the amount of vaginal loss and adverse events for 2 weeks after the procedure.The participants had to return their chart in a stamped addressed envelopes which were provided by the trial authors. Of 500 participants complying with inclusion/exclusion criteria; 250 were randomly allocated to receive antibiotics and 250 to receive placebo. The mean ages for the participants in antibiotic and placebo groups were 37.0 and 37.1 years, respectively. Approximately 60.4% and 53.6% of participants assigned to antibiotic and control groups, respectively, had underlying high‐grade cervical lesion on excised specimens. Approximately 32.8% and 28.8% of participants enrolled to antibiotic and control groups, respectively, underwent multiple passes of loop excision. 418 participants (70%) returned pictorial chart, but 70 were subsequently excluded due to incomplete data, leaving 348 charts (173 in the antibiotic group and 175 in the placebo group) for final analyses. |
|
| Interventions | Participants enrolled in the antibiotic group received ofloxacin 400 mg (2 x 200 mg) once daily for five consecutive days,starting immediately after the procedure. Participants assigned to the control group received identical placebo given in the same fashion. Ofloxacin is antimicrobial antibiotics with special activity against Chlamydial infections. | |
| Outcomes | The amount of postoperative vaginal discharge estimated by participants' self assessment using pictorial chart. | |
| Notes | Of 500 participants recruited, only 348 participants' charts were eligible for study analyses, corresponding to a rate of approximately 70%. The authors stated that the prevalence of Chlamydial infections among their colposcopy population was 10%. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | A quote from the study: " the study medication had been randomly assigned to the patient numbers in advance by the manufacture on a 1:1 basis" |
| Allocation concealment (selection bias) | Low risk | Following the procedure, participants were allocated their study number and received the study medication carrying her number. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Investigators and participants involving in this trial were blinded to the treatment received. Patients received either antibiotic or identical placebo, which had been prepared in the same packaging. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | The study outcome was the participants' self‐assessment on postoperative vaginal loss using pictorial charts. As the participants were blinded about the treatment received, there is low risk of detection bias. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Of 500 participants enrolled, adequate information for study analyses were obtained from only 348 participants. |
| Selective reporting (reporting bias) | Low risk | All potential relevant outcomes were reported. |
| Other bias | High risk | This study excluded participants who did not return their charts and those with inadequate information on their charts.Thus, the analyses performed did not follow an intention‐to‐treat basis. |
Gornall 1999.
| Methods | A randomised‐controlled trial conducted in the colposcopy clinic at the Princess Anne Hospital, Southamton, UK. Study duration: not reported |
|
| Participants | The participants were women undergoing LLETZ. The trial authors did not stated exclusion criteria. All participants received patients' charts contained a daily record of vaginal bleeding, vaginal discharge, and pain for 4 weeks after the procedure. The participants were asked to record the requirement of unscheduled medical visits and additional antibiotic treatment, if any. The participants had to return their chart at 3 months following the procedure. Initially, this study recruited 100 participants but only 77 returned their charts for analyses. | |
| Interventions | Participants allocated to the intervention group received Sultrin which is antimicrobial vaginal pessary containing sulphatiazole 3.42%, sulphacetamide 2.86%, and sulphabenzamide 3.7%, twice daily for 5 days. Participants assigned as the control group received no treatment. Sultrin vaginal pessary is a preparation used intravaginally against Haemophilus (Garnerella)vaginalis. | |
| Outcomes |
|
|
| Notes | The authors did not reported the details regarding the processes of randomisation and treatment allocation. In addition, of 100 participants recruited, only 77 participants' charts were available for analyses, corresponding to a rate of 77%.The authors did not reported the baseline characteristics of the participants, treatment‐related events, and actual number of participants assigned in each comparison group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | The authors did not reported the details of the processes of randomisation. |
| Allocation concealment (selection bias) | Low risk | The authors used sealed envelope during randomisation |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | The authors did not use placebo for the participant assigned to the control group so the participants were not blinded to treatment received. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Because the outcome based on the participants' self‐assessment on their symptoms occurring after the procedure but the participants were aware about the treatment received. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | of 100 participants recruited, only 77 participants' charts were available for analyses, corresponding to a rate of incomplete outcome data of approximately 23%. |
| Selective reporting (reporting bias) | Low risk | All potential relevant outcomes were reported. |
| Other bias | High risk | This study excluded participants who did not return their charts. Therefore, the analyses carried out in this study was not based on an intention‐to‐treat. The authors did not state about the review inclusion and exclusion criteria applied in this study. The authors also did not report the baseline characteristics of participants and number of participants allocated to each comparison group which are mandatory for determining treatment outcomes and assessing the quality of study methodology. |
LLETZ: large loop excision of the transformation zone
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Doyle 1992 | This randomised study was excluded because the intervention used was a Monsel’s solution applied after cervical excision which was not a prophylactic antibiotics, the intervention that this review aims to address. |
| Gerli 2012 | This randomised study entitled "Antiseptic regimen in the surgical treatment of HPV generated cervical lesions: polyhexamethylene biguanide vs chlorhexidine. A randomised, double blind study" was conducted to determine the effectiveness and safety of polyhexamethylene biguanide‐based vaginal suppositories compared to chlorhexidine‐based preparation. However, the treatment used in this study was an ablation using CO2 laser which did not meet our review inclusion. |
| Minorchio 1990 | This study entitled "Advantages of topical therapy with polydeoxyribonucleotide in reparative processes after cauterization: Experience at a centre for early diagnosis of genital neoplasm" was conducted to evaluate the effectiveness of kanamycin sulphate alternated with placebo versus polydeoxyribonucleotide vaginal suppositories for preventing postoperative infection and promoting tissue healing after cauterisation of the uterine cervix. However, this study was a controlled clinical trail and the treatment used in this study was not an excisional method. |
Differences between protocol and review
Types of interventions
In the review, we additionally stated the details of antibiotic regimen as "Antibiotic regimen includes the followings: administration route (for example intravenous, intramuscular, oral, vaginal); number of doses (for example, single versus multiple doses).
Types of outcome measures
In the review protocol, we stated that primary outcome was genital infections occurring within seven days of excision of the cervical transformation zone including (a) cervix: cervicitis 24 hours following the procedure as defined by one or more of the symptoms and signs; delayed bleeding; redness; pus or offensive discharge; positive gram stain; positive culture; (b) uterus or adnexa: pelvic inflammatory disease (PID) assessed by vaginal examination for all women 24 hours after the procedure, found to have clinical evidence of PID (cervical excitation, pelvic organ tenderness, or both); and (c) sepsis or other serious infection or infectious complications, either with or without fever, defined as body temperature higher than 38o Celsius on at least two occasions more than four hours apart, 24 hours after the procedure. Secondary outcome was adverse effects of antibiotics including adverse drug reactions, which were defined as any medical complications related to drug metabolisms; allergy, liver failure, renal failure, etc. according to the CTCAE version 4 (CTCAE 2010) and drug interactions.
In attempts to cover a broad range of outcome measures for infectious complications following excision of the cervical transformation zone, we revised this section in this review as follows; Primary outcomes: (1) Infectious complications (defined as any documented sites of infection identified by cultivation or clinical symptoms and signs, or both), measured as the proportion of participants who developed each of the following within eight weeks of surgery: cervicitis, endometritis, pelvic inflammatory disease, prolonged vaginal discharge, severe vaginal bleeding, lower abdominal pain, fever, and additional antibiotic treatment; (2) Antibiotic resistance.
Secondary outcomes included (1) Adverse effects‐related to antibiotics: classified according to Common Terminology Criteria for Adverse Events (CTCAE 2010) as follows: gastrointestinal disorders (nausea, vomiting, anorexia, diarrhoea); immune system disorders (allergic reaction, anaphylaxis) blood and lymphatic system disorders (leucopenia, anaemia, thrombocytopenia, neutropenia); skin (stomatitis, mucositis, alopecia, allergy); nervous system disorders; genitourinary; and other side effects not categorised above; (2) unscheduled medical consultant (for any reasons); and (3) additional self‐medication .
Searching other source
In the review, we added Index to theses (ProQuest Dissertations & Theses: UK & Ireland) and the WHO International Clinical Trial Registry (http://apps.who.int/trialsearch/Default.aspx) as additional databases to be searched. In addition, we added the lists of conferences used for searching abstracts and proceedings
Data synthesis
We additionally stated in the review that "we prepared a ’Summary of findings’ table to present the results of the meta‐analysis, based on the methods described in Chapter 11 of the Cochrane Handbook for Systematic Reviews of Interventions (Schunemann 2011). We presented the results of the meta‐analysis for the primary outcome and adverse events as outlined in the 'Types of outcome measures’ section".
Assessment of reporting biases
As there was only three trials that met our inclusion criteria, we were unable to construct funnel plots to determine the possibility of publication bias as previously stated in the review protocol. In future update of this review, we will construct funnel plots corresponding to meta‐analyses of the primary outcome to assess the possibility of publication bias if we identify a sufficient number of included studies (i.e. more than 10). We will also carry out sensitivity analyses to investigate the effect on the pooled results if the funnel plots are asymmetrical.
Subgroup analysis and investigation of heterogeneity
All three included trials in this review relate primarily to cervical transformation zone excision using electrical wire loop. Therefore, subgroup analyses according to the types of excisional techniques as mentioned in the review protocol was not feasible.
Contributions of authors
Chumnan Kietpeerakool: developed and completed the review Bandit Chumworathayi: conceived the review question, developed, co‐ordinated the review. Jadsada Thinkamrop: performed part of the editing of the review and advised on part of the review. Butsakorn Ussahgij: searched for potential eligible studies and advised on part of the review Pisake Lumbiganon: co‐ordinated the development of the review, edited the review, and advised on the review. All authors approved the final version of the review prior to submission.
Sources of support
Internal sources
Department of Obstetrics and Gynaecology, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand.
Cochrane Thailand, Thailand.
External sources
Thailand Research Fund (Distinguished Professor Award), Thailand.
Declarations of interest
CK: None known BC: None known JT: None known BU: None known PL: None known
Edited (no change to conclusions)
References
References to studies included in this review
Chan 2007 {published data only}
- Chan KKL, Tam KF, Tse KY, Ngan HYS. The use of vaginal antimicrobial after large loop excision of transformation zone: a prospective randomised trial. BJOG: An International Journal of Obstetrics & Gynaecology 2007;114(8):970‐6. [DOI] [PubMed] [Google Scholar]
Foden‐Shroff 1998 {published data only}
- Foden‐Shroff J, Redman CW, Tucker H, Millinship J, Thomas E, Warwick A, et al. Do routine antibiotics after loop diathermy excision reduce morbidity?. British Journal of Obstetrics and Gynaecology 1998;105(9):1022‐5. [DOI] [PubMed] [Google Scholar]
Gornall 1999 {published data only}
- Gornall RJ, Beynon DGW, Shepherd NJ, Boyd IE. Topical antiseptic agent after large loop excision of the transformation zone: results of a randomised controlled trial. Journal of Obstetrics and Gynaecology 1999;19(5):509‐10. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Doyle 1992 {published data only}
- Doyle M, Warwick A, Redman C, Hillier C, Chenoy R, O'Brien S. Does application of Monsel’s solution after loop diathermy excision of the transformation zone reduce post‐operative discharge? Results of a prospective randomized controlled trial. British Journal of Obstetrics and Gynaecology 1992;99(12):1023‐4. [DOI] [PubMed] [Google Scholar]
Gerli 2012 {published data only}
- Gerli S, Bavetta F, Renzo GC. Antisepsis regimen in the surgical treatment of HPV generated cervical lesions: polyhexamethylene biguanide vs chlorhexidine. A randomized, double blind study. European Review for Medical and Pharmacological Sciences 2012;16(14):1994‐8. [PubMed] [Google Scholar]
Minorchio 1990 {published data only}
- Minorchio E, Bianco V, Corso F. Advantages of topical therapy with polydeoxyribonucleotide in reparative processes after cauterization: experience at a center for early diagnosis of genital neoplasms. Annali di Ostetricia, Ginecologia, Medicina Perinatale. 1990;111(6):388‐92. [PubMed] [Google Scholar]
Additional references
ACOG 2009
- ACOG Committee on Practice Bulletins‐‐Gynecology. ACOG practice bulletin No. 104: antibiotic prophylaxis for gynecologic procedures. Obstetrics & Gynecology 2009;113(5):1180‐9. [DOI] [PubMed] [Google Scholar]
Bitsori 2014
- Bitsori M, Maraki S, Galanakis E. Long‐term resistance trends of uropathogens and association with antimicrobial prophylaxis. Pediatric Nephrology 2014;29(6):1053‐8. [DOI] [PubMed] [Google Scholar]
Buppasiri 2014
- Buppasiri P, Lumbiganon P, Thinkhamrop J, Thinkhamrop B. Antibiotic prophylaxis for third‐ and fourth‐degree perineal tear during vaginal birth. Cochrane Database of Systematic Reviews 2014, Issue 10. [DOI: 10.1002/14651858.CD005125.pub4] [DOI] [PubMed] [Google Scholar]
Chamot 2010
- Chamot E, Kristensen S, Stringer JSA, Mwanahamuntu MH. Are treatments for cervical precancerous lesions in less‐developed countries safe enough to promote scaling‐up of cervical screening programs? A systematic review. BMC Women's Health 2010;10:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
Craciunas 2014
- Craciunas L, Tsampras N. A literature review of the current evidence for routine antibiotic prophylaxis after cervical tissue excisions. Journal of Obstetrics and Gynaecology 2014;34(8):700‐5. [DOI] [PubMed] [Google Scholar]
CTCAE 2010
- National Institutes of Health ‐ National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE) v4.03. National Institutes of Health, US Department of Health and Human Services 2010; publication no. 09‐7473. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010‐06‐14_QuickReference_8.5x11.pdf (accessed 08 December 2016).
Deeks 2001
- Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta‐analysis. In: Egger M, Davey Smith G, Altman DG editor(s). Systematic Reviews in Health Care: Meta‐Analysis in Context. 2nd Edition. London: BMJ Publication Group, 2001:285–312. [Google Scholar]
Del Mar 2012
- Mar C, Lasserson T. Procalcitoninc: hope in the fight against antibiotic resistance? [editorial]. Cochrane Database of Systematic Reviews 2012, issue 9. [DOI: 10.1002/14651858.ED000046] [DOI] [PMC free article] [PubMed]
DerSimonian 1986
- DerSimonian R, Laird N. Meta‐analysis in clinical trials. Controlled Clinical Trials 1986;7(3):177‐88. [DOI] [PubMed] [Google Scholar]
Grimes 1999
- Grimes DA, Lopez LM, Schulz KF. Antibiotic prophylaxis for intrauterine contraceptive device insertion. Cochrane Database of Systematic Reviews 1999, Issue 3. [DOI: 10.1002/14651858.CD001327] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011 Available from Cochrane handbook.org www.cochrane‐handbook.org.
Janthanaphan 2009
- Janthanaphan M, Wootipoom V, Tangsinmunkong K, Liabsuetrakul T. Comparison of success rate and complications of contour‐loop excision of the transformation zone (C‐LETZ) with cold knife conization (CKC) in high grade lesion (HGL) from colposcopic impression. Journal of the Medical Association of Thailand 2009;92(12):1573‐9. [PubMed] [Google Scholar]
Kietpeerakool 2007
- Kietpeerakool C, Srisomboon J, Suprasert P, Cheewakriangkrai C, Charoenkwan K, Siriaree S. Routine prophylactic application of Monsels solution after loop electrosurgical excision procedure of the cervix: is it necessary?. Journal of Obstetrics and Gynaecology Research 2007;33(3):299‐304. [DOI] [PubMed] [Google Scholar]
Lopez 1994
- Lopes A, Beynin G, Robertson G, Daras V, Monaghan JM. Short term morbidity following large loop excision of the cervical transformation zone. Journal of Obstetrics and Gynaecology 1994;14(3):197‐9. [Google Scholar]
Marjoribanks 2004
- Marjoribanks J, Calis KA, Jordan V. Antibiotic prophylaxis for elective hysterectomy. Cochrane Database of Systematic Reviews 2004, Issue 1. [DOI: 10.1002/14651858.CD004637] [DOI] [PMC free article] [PubMed] [Google Scholar]
Martin‐Hirsch 2013
- Martin‐Hirsch PP, Paraskevaidis E, Bryant A, Dickinson HO. Surgery for cervical intraepithelial neoplasia. Cochrane Database of Systematic Reviews 2013, Issue 12. [DOI: 10.1002/14651858.CD001318.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Massad 2013
- Massad LS, Einstein MH, Huh WK, Katki HA, Kinney WK, Schiffman M, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstetrics & Gynecology 2013;121(4):829‐46. [DOI] [PubMed] [Google Scholar]
McMurray 2015
- McMurray CL, Hardy KJ, Verlander NQ, Hawkey PM. Antibiotic surgical prophylaxis increases nasal carriage of antibiotic‐resistant staphylococci. Journal of Medical Microbiology 2015;64(12):1489‐95. [DOI] [PubMed] [Google Scholar]
Minami 2014
- Minami T, Sasaki T, Serikawa M, Ishigaki T, Murakami Y, Chayama K. Antibiotic prophylaxis for endoscopic retrograde chlangiopancreatography increases the detection rate of drug‐resistant bacteria in bile. Journal of Hepato‐Biliary‐Pancreatic Sciences 2014;21(9):712‐8. [DOI] [PubMed] [Google Scholar]
Mints 2006
- Mints M, Gaberi V, Andersson S. Miniconization procedure with C‐LETZ conization electrode for treatment of cervical intraepithelial neoplasia: a Swedish study. Acta Obstetricia et Gynecologica Scandinavica 2006;85(2):218‐23. [DOI] [PubMed] [Google Scholar]
Mitchell 1998
- Mitchell MF, Tortolero‐Luna G, Cook E, Whittaker L, Rhodes‐Morris H, Silva E. A randomized clinical trial of cryotherapy, laser vaporization, and loop electrosurgical excision for treatment of squamous intraepithelial lesions of the cervix. Obstetrics & Gynecology 1998;92(5):737‐44. [PubMed] [Google Scholar]
Morrill 2013
- Morrill MY, Schimp MO, Abed H, Carberry C, Margulies RU, White AB, et al. Antibiotic prophylaxis for selected gynecologic surgeries. International Journal of Gynecology and Obstetrics 2013;120(1):10‐5. [DOI] [PubMed] [Google Scholar]
NICE 2008
- NICE 2008. Surgical site infections: prevention and treatment: NICE guidelines [CG74]. https://www.nice.org.uk/guidance/cg74; accessed 08/12/2016. National Institute for Health and Clinical Excellence, 2008. [PubMed] [Google Scholar]
Prendiville 1989
- Prendiville W, Cullimore J, Norman S. Large loop excision of the transformation zone (LLETZ): A new method of management for women with cervical intraepithelial neoplasia. British Journal of Obstetrics and Gynaecology 1989;96(9):1054‐60. [DOI] [PubMed] [Google Scholar]
RevMan 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Sawaya 1996
- Sawaya GF, Grady D, Kerlikowske K, Grimes DA. Antibiotics at the time of induced abortion: the case for universal prophylaxis based on a meta‐analysis. Obstetrics & Gynecology 1996;87(5 Pt 2):884‐90. [PubMed] [Google Scholar]
Schunemann 2011
- Schünemann HJ, Oxman AD, Higgins JPT, Vist GE, Glasziou P, Guyatt GH. Chapter 11: Presenting results and ’Summary of findings’ tables.. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. [Google Scholar]
Sterne 2011
- Sterne J, Sutton A, Loannidis J, Terrin N, Jones D, Lau J, et al. Recommendations for examining and interpreting funnel plot asymmetry in meta‐analyses of randomized controlled trials. BMJ 2011;343:d4002. [DOI] [PubMed] [Google Scholar]
Takac 1999
- Takac I, Gorisek B. Cold knife conization and loop excision for cervical intraepithelial neoplasia. Tumori 1999;85(4):243‐6. [DOI] [PubMed] [Google Scholar]
Treszezamsky 2010
- Treszezamsky AD, Molina Boero MF, Mehta I. Cervical conization complicated by sepsis with lung and liver abscesses. Journal of Lower Genital Tract Disease 2010;14(2):130‐3. [DOI] [PubMed] [Google Scholar]
WHO 2015
- World Health Organization. Antimicrobial resistance: Fact sheet N°194. http://www.who.int/mediacentre/factsheets/fs194/en/; accessed 08/12/2016 2016.
Wiebe 2012
- Wiebe E, Denny L, Thomas G. Cancer of the cervix uteri. International Journal of Gynecology and Obstetrics 2012;119(Suppl 2):S100‐9. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Chumworathayi 2012
- Chumworathayi B, Udomthavornsuk B, Thinkhamrop J, Lumbiganon P. Antibiotics for infection prevention after excision of the cervical transformation zone. Cochrane Database of Systematic Reviews 2012, Issue 7. [DOI: 10.1002/14651858.CD009957] [DOI] [PMC free article] [PubMed] [Google Scholar]


