Abstract
Background
Traditional monitoring of ovarian hyperstimulation during in vitro fertilisation (IVF) and intra‐cytoplasmic sperm injection (ICSI) treatment has included transvaginal ultrasonography (TVUS) plus serum estradiol levels to ensure safe practice by reducing the incidence and severity of ovarian hyperstimulation syndrome (OHSS) whilst achieving the good ovarian response needed for assisted reproduction treatment. The need for combined monitoring (using TVUS and serum estradiol) during ovarian stimulation in assisted reproduction is controversial. It has been suggested that combined monitoring is time consuming, expensive and inconvenient for women and that simplification of IVF and ICSI therapy by using TVUS only should be considered.
Objectives
To assess the effect of monitoring controlled ovarian hyperstimulation (COH) in IVF and ICSI cycles in subfertile couples with TVUS only versus TVUS plus serum estradiol concentration, with respect to rates of live birth, pregnancy and OHSS.
Search methods
We searched the Menstrual Disorders and Subfertility Group Specialised Register of controlled trials, the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, CINAHL, PsycINFO, the National Research Register, and web‐based trial registers such as Current Controlled Trials. The last search was conducted in May 2014. There was no language restriction applied. All references in the identified trials and background papers were checked and authors were contacted to identify relevant published and unpublished data.
Selection criteria
Only randomised controlled trials that compared monitoring with TVUS only versus TVUS plus serum estradiol concentrations in women undergoing COH for IVF and ICSI treatment were included.
Data collection and analysis
Three review authors independently selected the studies, extracted data and assessed risk of bias. They resolved disagreements by discussion with the rest of the authors. Outcomes data were pooled and summary statistics were presented when appropriate. The quality of the evidence was rated using the GRADE methods.
Main results
With this update, four new studies were identified resulting in a total of six trials including 781 women undergoing monitoring of COH with either TVUS alone or a combination of TVUS and serum estradiol concentration during IVF or ICSI treatment.
None of the six studies reported our primary outcome of live birth rate. Pooled data showed no evidence of a difference in clinical pregnancy rate per woman between monitoring with TVUS only and combined monitoring (odds ratio (OR) 1.10; 95% confidence interval (CI) 0.79 to 1.54; four studies; N = 617; I² = 5%; low quality evidence). This suggests that compared with women with a 34% chance of clinical pregnancy using monitoring with TVUS plus serum estradiol, the clinical pregnancy rate in women using TVUS only was between 29% and 44%.
There was no evidence of a difference between the groups in the reported cases of OHSS (OR 1.03; 95% CI 0.48 to 2.20; six studies; N = 781; I² = 0%; low quality evidence), suggesting that compared with women with a 4% chance of OHSS using monitoring with TVUS plus serum estradiol, the OHSS rate in women monitored by TVUSS only was between 2% and 8%.
There was no evidence of a difference between the groups in the mean number of oocytes retrieved pre woman (mean difference (MD) 0.32; 95% CI ‐0.60 to 1.24; five studies; N = 596; I² = 17%; low quality evidence).
The evidence was low quality for all comparisons. Limitations included imprecision and potential bias due to unclear randomisation methods, allocation concealment and blinding, as well as differences in treatment protocols. Quality assessment was hampered by the lack of methodological descriptions in several studies.
Authors' conclusions
This review update found no evidence from randomised trials to suggest that combined monitoring by TVUS and serum estradiol is more efficacious than monitoring by TVUS alone with regard to clinical pregnancy rates and the incidence of OHSS. The number of oocytes retrieved appeared similar for both monitoring protocols. The data suggest that both these monitoring methods are safe and reliable. However, these results should be interpreted with caution because the overall quality of the evidence was low. Results were compromised by imprecision and poor reporting of study methodology. A combined monitoring protocol including both TVUS and serum estradiol may need to be retained as precautionary good clinical practice and as a confirmatory test in a subset of women to identify those at high risk of OHSS. An economic evaluation of the costs involved with the two methods and the views of the women undergoing cycle monitoring would be welcome.
Plain language summary
Monitoring of stimulated cycles in fertility treatment involving in vitro fertilisation (IVF) and intra‐cytoplasmic sperm injection (ICSI)
Review question: can ultrasound alone be used safely without adding estradiol blood test measurements to monitor women undergoing controlled ovarian hyperstimulation during IVF and ICSI? We reviewed the evidence on monitoring women undergoing controlled ovarian hyperstimulation as part of IVF or ICSI by transvaginal ultrasound (TVUS) only versus traditional combined monitoring (TVUS) and blood hormone (estradiol) levels.
Background: assisted reproduction techniques such as IVF and ICSI involve ovarian hyperstimulation. The ovaries are artificially stimulated to produce follicles and then ovulation (release of a mature ovum or egg) is induced so that eggs can be retrieved for use in either IVF or ICSI to produce embryos in the laboratory. Traditionally women undergoing controlled ovarian hyperstimulation prior to ovulation induction have been monitored by TVUS and measurement of the hormone estradiol level in their blood. The aim of monitoring is to detect the optimum time to induce ovulation (by the administration of human chorionic gonadotrophin or luteinising hormone) and to determine an adequate response to ovarian hyperstimulation to allow egg retrieval, but importantly also to identify women at risk of the potentially serious rare condition of ovarian hyperstimulation syndrome (OHSS). It has been suggested that a simplified protocol of monitoring by TVUS alone may reduce unnecessary anxiety and operational costs during IVF and ICSI.
Study characteristics: we included six randomised controlled trials conducted in the UK, France, Spain, Israel and the US, including 781 women. They compared monitoring with TVUS only versus TVUS plus serum estradiol concentration in women undergoing ovarian hyperstimulation for IVF and ICSI treatment. The evidence was current to May 2014.
Key results: none of the six studies reported our primary outcome of live birth rate. Pooled data showed no evidence of a difference in clinical pregnancy rate between monitoring with TVUS only and monitoring with TVUS plus estradiol measurement (odds ratio (OR) 1.10; 95% CI 0.79 to 1.54; four studies; N = 617; I² = 5%; low quality evidence). Our findings suggest that compared with women with a 34% chance of clinical pregnancy using monitoring with TVUS plus serum estradiol, the clinical pregnancy rate in women using TVUS only was between 29% and 44%. There was no evidence of a difference in OHSS between the two arms (OR 1.03; 95% CI 0.48 to 2.20; six studies; N = 781; I² = 0%; low quality evidence), suggesting that compared with women with a 4% chance of OHSS using monitoring with TVUS plus serum estradiol, the OHSS rate in women monitored by TVUS only was between 2% and 8%.
Quality of the evidence: the evidence was of low quality. Limitations included imprecision and potential bias due to unclear randomisation methods, allocation concealment and blinding, as well as differences in the treatment protocols. Quality assessment was hampered by a lack of methodological descriptions in several studies. Two studies reported funding by pharmaceutical companies, whereas the remaining four studies did not report their sources of funding.
Summary of findings
Summary of findings for the main comparison. Monitoring ovarian stimulation by transvaginal ultrasound (TVUS) only compared to monitoring with TVUS plus serum estradiol for women undergoing ovarian stimulation with gonadotrophins in IVF and ICSI procedures.
| Monitoring ovarian stimulation by transvaginal ultrasound (TVUS) only compared to monitoring with TVUS plus serum estradiol for women undergoing ovarian stimulation with gonadotrophins in IVF and ICSI procedures | ||||||
| Population: Women undergoing ovarian stimulation with gonadotrophins in IVF and ICSI procedures Settings: Hospital or outpatient assisted reproduction units Intervention: Monitoring ovarian stimulation by transvaginal ultrasound (TVUS) only Comparison: Monitoring with TVUS plus serum estradiol | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Monitoring with TVUS plus serum estradiol | Monitoring by TVUS only | |||||
| Live birth per woman | No evidence available, as none of the included studies reported live birth as an outcome | |||||
| Clinical pregnancy per woman | 337 per 1000 | 361 per 1000 (287 to 439) | OR 1.05 (0.79 to 1.54) | 617 (4 studies) | ⊕⊕⊝⊝ low1,6 | No difference demonstrated between monitoring by TVUS only and TVUS plus serum estradiol |
| Mean number of oocytes retrieved per woman | The mean number of oocytes retrieved per woman in the intervention groups was 0.32 higher (0.6 lower to 1.24 higher) | 595 (5 studies) | ⊕⊕⊝⊝ low2,7 | No difference demonstrated between monitoring by TVUS only and TVUS plus serum estradiol | ||
| Cycle cancellation rate per woman | 36 per 1000 |
17 per 1000 (3 to 140) |
OR 0.57 (0.07 to 4.39) |
115 (2 studies) |
⊕⊕⊝⊝ low3,6 | No difference demonstrated between monitoring by TVUS only and TVUS plus serum estradiol |
| OHSS rate (mild, moderate or severe) per woman | 36 per 1000 | 36 per 1000 (18 to 75) | OR 1.03 (0.48 to 2.20) | 781 (6 studies) | ⊕⊕⊝⊝ low4,5,6 | No difference demonstrated between monitoring by TVUS only and TVUS plus serum estradiol Two studies reported no incidence of OHSS |
| *The basis for the assumed risk is the median control group risk across studies. The corresponding risk (and its 95% CI) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; OR: Odds ratio | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1 Methods of allocation concealment inadequately described in the four trials; none of the trials adequately described blinding 2 Inadequate description of methods of randomisation in two of the five trials, allocation concealment all of the five trials and blinding in four of the five trials
3 Methods of randomisation and allocation concealment inadequately described in one of the two trials 4 Methods of randomisation inadequately described in three of the six trials, allocation concealment inadequately described in all the six trials and blinding inadequately described in five of the six trials 5 No definition of OHSS provided by authors of these 6 studies
6 Serious imprecision with wide confidence intervals
7 Unclear whether mean values correctly calculated (i.e. whether they include zero counts for cancelled cycles)
Background
The overall aim of in vitro fertilisation (IVF) and intra‐cytoplasmic sperm injection (ICSI) in assisted reproduction is to achieve pregnancy and ultimately live births. A successful outcome from IVF and ICSI is dependent upon a preliminary phase of controlled ovarian hyperstimulation (COH) using exogenous gonadotrophins. The aim of COH is to produce multiple follicles and thereafter to induce follicle maturation. Ovulation is then induced by administration of human chorionic gonadotrophin (hCG) or luteinizing hormone, which then allows optimal and multiple opportunities for subsequent fertilisation. However, during this process of COH and induction there exists the risk of ovarian hyperstimulation syndrome (OHSS), which is the most serious iatrogenic complication of ovarian stimulation. There is debate over whether women should be monitored during COH using a combination of transvaginal ultrasound (TVUS) and serum estradiol measurements or by TVUS alone.
Description of the condition
The aim of achieving clinical pregnancy and ultimately live births must be balanced with the risks associated with assisted reproductive techniques, and specifically those related to ovarian hyperstimulation. OHSS is a systemic condition associated with development of a large number of follicles and is a potentially fatal condition (Delvigne 2002; Jenkins 2006; Schenker 1978). OHSS can lead to ascites, pleural and pericardial effusion, haemoconcentration, venous thromboembolism, hepatorenal failure and coagulopathy (Borase 2012; Delvigne 2002; Jenkins 2006; Schenker 1978). It has been suggested that the condition is related to the release of vasoactive chemicals from the ovaries during COH and ovulation induction, which leads to increased vascular permeability and thus fluid shifts extravascularly (Jenkins 2006). However, the exact aetiology remains unknown. The incidence of OHSS is estimated to range from 0.2% to 2.7% of all assisted reproductive cycles, including intra‐uterine insemination (Asch 1991; Ferraretti 2012; MacDougall 1992; Nygren 2002; Roest 1996; Smitz 1990).
Description of the intervention
COH is monitored by means of TVUS in order to gain information on the number and size of developing follicles and to determine the optimal time for hCG administration prior to oocyte retrieval. Some fertility units measure serum estradiol concentration alongside TVUS during the course of COH to provide added information about the ovarian response and the potential risk of hyperstimulation. Combined monitoring with TVUS and serum estradiol concentrations has been suggested to be the gold standard for monitoring stimulated cycles in IVF and ICSI procedures (Rizk 1992).
How the intervention might work
The number and size of follicles visualised at TVUS provide an estimate of ovarian response and hCG is used to trigger ovulation when a certain number of follicles reach a certain size. Estradiol, which is produced by developing follicles, provides additional information which is believed to further improve the decision making process; follicle maturity is supported by adequate estradiol levels while there is an increased risk of OHSS in the presence of very high levels. The overall aim is to ensure optimal stimulation of the ovaries and to reduce the incidence and severity of OHSS whilst obtaining a successful treatment outcome.
TVUS monitoring alone has been reported to provide more accurate information on follicular number and size than can be obtained by serum estradiol concentration alone in women with anovulatory infertility undergoing gonadotrophin induction therapy (Haning 1982; Hardiman 1990; Shoham 1991).
A cohort study comparing TVUS only versus TVUS plus hormonal determinations, including serum estradiol concentrations, for ovarian monitoring in women undergoing IVF reported no differences in live birth rate and the incidence of OHSS, and there was a significant economic benefit in the TVUS only monitoring protocol (Murad 1998). Another cohort study reported that TVUS alone, performed during COH in IVF and intra‐uterine insemination, predicted 88% of cycle decisions as compared to 100% of cycle decisions that were predicted using combined monitoring (Confino 1996).
One non‐randomised study reported no differences in IVF outcomes and incidence of OHSS between women whose ovarian response was monitored by TVUS and serum estradiol concentration on the day of human chorionic gonadotrophin (hCG) administration and women who were monitored with TVUS only and had the serum estradiol concentration checked if the risk of OHSS was deemed to be high (Thomas 2002). A more recent non‐randomised study compared combined monitoring with ultrasound monitoring only in women undergoing IVF and ICSI and reported equivalence in results between the two arms for the primary outcome of number of mature oocytes at egg retrieval (Vandekerckhove 2014).
Why it is important to do this review
The need for combined monitoring during COH in IVF and ICSI is controversial. It has been suggested that close monitoring is time consuming, expensive and inconvenient for the woman (Howard 1988; Rainhorn 1987; Tan 1992). Simplification of IVF therapy by minimal monitoring has been reported to have no adverse effects on treatment outcome and the incidence of OHSS (Abdalla 1989; Roest 1995; Tan 1994) and some IVF programs have abandoned the use of the hormone assay completely (Kemeter 1989; Tan 1994; Vlaisavljevic 1992). This review is important to couples, clinicians and researchers because if there is evidence that estradiol measurements are unnecessary, hospitals and clinics could change their protocols thus reducing associated costs and anxiety to the couple undergoing IVF or ICSI.
Objectives
To assess the effect of monitoring controlled ovarian hyperstimulation (COH) in IVF and ICSI cycles in subfertile couples with TVUS only versus TVUS plus serum estradiol concentration, with respect to rates of live birth, pregnancy and OHSS.
Methods
Criteria for considering studies for this review
Types of studies
Randomised controlled trials only. Quasi‐randomised trials were excluded. Cross‐over trials were excluded as they are an inappropriate design for this question because ‘success’ in the first phase would prevent entry to subsequent phases of the study.
Types of participants
All women undergoing COH with gonadotrophins in IVF and ICSI procedures.
Types of interventions
Monitoring by transvaginal ultrasound (TVUS) only versus combined monitoring with TVUS plus serum estradiol concentrations.
Types of outcome measures
Primary outcomes
Live birth rate per woman
Secondary outcomes
Pregnancy rate and multiple pregnancy rate per woman, where clinical pregnancy is defined as a pregnancy diagnosed by ultrasonographic visualisation of one or more gestational sacs or definitive clinical signs of pregnancy. It includes ectopic pregnancy. Multiple gestational sacs are counted as one clinical pregnancy
Number of oocytes recovered per woman
Adverse outcomes: rate of ovarian hyperstimulation syndrome (OHSS) per woman; number of cancelled cycles per woman
Costs of monitoring with TVUS versus costs of monitoring with TVUS plus serum estradiol concentrations
Search methods for identification of studies
Electronic searches
We searched the Menstrual Disorders and Subfertility Group (MDSG) Specialised Register of controlled trials (Appendix 1) (May 2014) without language restriction and in consultation with the MDSG Trials Search Co‐ordinator.
We conducted searches in the following electronic databases:
1. CENTRAL (Appendix 2) (April 2014); 2. MEDLINE (Appendix 3) (May 2014); 3. EMBASE (Appendix 4) (May 2014); 4. PsycINFO (Appendix 5) (May 2014);
5.CINAHL (Appendix 6) (May 2014).
Searching other resources
Two review authors (AW, AK) searched online research databases without language or date restrictions (May 2014) including Clinicaltrials.gov, PubMed, LILACS/VHL, OpenGrey, ISI Web of Knowledge, World Health Organization (WHO) Clinical Trial Registries Platform and the Database of Abstracts of Reviews of Effects (DARE) in The Cochrane Library. We handsearched online relevant international and national peer‐reviewed journals for relevant studies. These included:
Human Reproduction;
Human Reproduction Update;
Molecular Human Reproduction (MHR);
Fertility and Sterility;
American Journal of Obstetrics and Gynecology;
Best Practice & Research. Clinical Obstetrics & Gynaecology;
Current Obstetrics and Gynaecology;
European Journal of Obstetrics, Gynecology, and Reproductive Biology;
European Journal of Obstetrics and Gynecology;
Infertility and Reproductive Medicine Clinics of North America;
International Journal of Gynaecology and Obstetrics;
Journal of Reproductive Immunology;
Middle East Fertility Society Journal;
Obstetrics and Gynecology Clinics of North America;
Placenta;
Obstetrics, Gynaecology and Reproductive Medicine;
Reproductive BioMedicine Online;
Reproductive Health Matters;
Sexual & Reproductive Healthcare;
Women's Health Issues;
Women's Health Medicine;
The Australian & New Zealand Journal of Obstetrics & Gynaecology;
Reproduction, Fertility, and Development;
American Journal Reproductive Immunology and Microbiology;
Biology of Reproduction;
Reproduction;
International Journal of Andrology.
In addition, we searched and checked reference lists of the included studies, and contacted authors for clarification of study methodology. We translated one article that was in French (Rongieres 2006) and one in Spanish (Aguirre 2010).
Data collection and analysis
Selection of studies
Three review authors (IK, AW, AK) independently examined the electronic search results for reports of possibly relevant trials. These reports were retrieved in full. Three review authors independently applied the selection criteria to the trial reports and resolved disagreements by discussion with the other review author (SB). AK and IK contacted authors for further clarification of study eligibility and methods.
Data extraction and management
Data collection and analysis were conducted in accordance with the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). Three review authors (IK, AW, AK) independently extracted the data and information using a data extraction form designed by the MDSG and pilot‐tested by the authors. The review authors were not blinded to the authors or publication journal when doing this. Results were compared and any differences resolved by discussion. Where there was insufficient information in the published report, AK and IK contacted the authors to request further information and clarification.
Assessment of risk of bias in included studies
Three authors (IK, AK, AW) independently assessed each trial for risk of bias according to the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We assessed random sequence generation, allocation concealment, blinding, completeness of outcome data (including use of intention‐to‐treat (ITT) analysis) and selective outcome reporting for each trial. Other potential sources of bias were also assessed. Each trial was categorised as at low, unclear or high risk of bias for each domain based on the standards described in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). Where the method was not clearly reported, AK and IK contacted the authors for clarification.
Measures of treatment effect
For dichotomous data we used the number of events in the control and intervention groups of each study to calculate Mantel‐Haenszel odds ratios (ORs). For continuous data we calculated the mean difference (MD) between treatment groups. We presented 95% confidence intervals (CIs) for all outcomes.
Unit of analysis issues
The primary analyses were per woman randomised. For reported data that did not allow valid analysis (for example 'per cycle' rather than 'per woman', where the same women may contribute more than one cycle) and when contact with authors yielded no additional data or when contacts were unsuccessful, we excluded the outcomes from the analyses.
Dealing with missing data
We analysed the outcomes data of women as they were randomised by intention to treat (ITT). Trial authors were contacted for any missing data. Where no additional information was forthcoming and when we were not able to impute any missing values from available data, we excluded these outcomes from the analyses.
Assessment of heterogeneity
We considered whether the clinical and methodological characteristics of the included studies were sufficiently similar for meta‐analysis to provide a clinically meaningful summary. For each meta‐analysis, we assessed statistical heterogeneity using the I² and Chi² statistics. Substantial heterogeneity was determined to be present if I² was greater than 50%, or if P < 0.05 in the Chi² test for heterogeneity (Higgins 2011).
Assessment of reporting biases
We planned that a funnel plot would be constructed if 10 or more studies presented data on the same outcome.
Data synthesis
Where appropriate, we combined dichotomous data for meta‐analysis using the Mantel‐Haenszel method to estimate pooled ORs with 95% CIs using a fixed‐effect model. For continuous data, we computed weighted MD with 95% CI also using a fixed‐effect model in the meta‐analysis.
Subgroup analysis and investigation of heterogeneity
We planned to conduct subgroup analysis to investigate heterogeneity, identified using the Chi² test (with P < 0.05 as evidence of significant heterogeneity) and the I² statistic (Higgins 2011), stratified by risk of bias in study design, where participants or assessors, or both, were blinded versus non‐blinded, as well as by clinical characteristics of study populations such as OHSS risks categories and first IVF versus previously failed IVF attempts. We would have conducted these analyses had there been sufficient data available.
Sensitivity analysis
As no included study reported our primary outcome of live birth rates per woman, we did not perform the planned sensitivity analysis to assess whether the findings were robust or whether the conclusions would have differed if eligibility was restricted to studies without a high risk of bias.
Summary of findings table
We prepared a summary of findings table using GRADEpro software. This table evaluated the overall quality of the body of evidence for the primary review outcomes using GRADE criteria (study limitations (that is risk of bias), consistency of effect, imprecision, indirectness and publication bias). Judgements about evidence quality (high, moderate or low) were justified, documented, and incorporated into reporting of results for each outcome. See Table 1.
Results
Description of studies
See Characteristics of included studies; Characteristics of excluded studies.
Results of the search
In the original review, our search strategy identified 1119 potentially eligible reports of which two (Golan 1994; Lass 2003) met our inclusion criteria. They involved 411 women who underwent COH monitoring. One study (Murad 1998) was excluded as it was not a randomised controlled trial (RCT). In the 2014 review update, an additional four studies (Aguirre 2010; Rongieres 2006; Strawn 2007; Wiser 2012) involving 370 women were identified. One of these included studies (Strawn 2007) was reported as a conference abstract. Three studies were excluded: one was a quasi‐randomised study (Schindler 2001); one was not a randomised study (Kably Ambe 1994); and the other was an RCT that compared combined monitoring with conventional two‐dimensional (2D) and three‐dimensional (3D) ultrasound instruments (Raine‐Fenning 2010). See Figure 1 for details of the screening and selection process.
1.
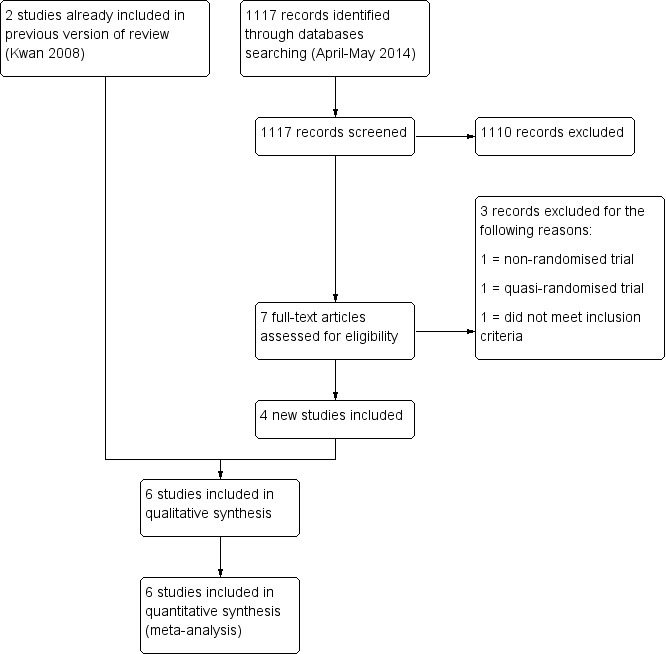
Study flow diagram.
Included studies
A total of six studies was included in this review (see Characteristics of included studies).
Study design and setting
The six included studies were all RCTs published between 1994 and 2012 (Aguirre 2010; Golan 1994; Lass 2003; Rongieres 2006; Strawn 2007; Wiser 2012) and conducted in UK (N = 1), France (N = 1), Spain (N = 1), Israel (N = 2) and the US (N = 1). One was a multi‐centre study involving four assisted conception units (Lass 2003). None of the trials specifically reported that the participants included egg donors. We did not identify any cross‐over trials.
Participants
The studies included 781 women (sample size ranged from 50 to 297), 394 in the control groups and 387 in the intervention groups, with an overall age range between 23 and 42 years. Two studies included only women admitted for the first IVF attempt (Golan 1994; Wiser 2012) and two studies included women who had had less than three previous unsuccessful IVF cycles (Aguirre 2010; Lass 2003). Women for ICSI were excluded from one study (Lass 2003) and so were women with a previous history of serious OHSS (Rongieres 2006). Most participants were women with subfertility problems due to tubal factors, endometriosis, polycystic ovary syndrome (PCOS), male factors or unexplained infertility.
Interventions
In this review, ovarian monitoring in the intervention group was conducted by TVUS only; and that of the control group by TVUS plus serum estradiol concentration. All six studies reported the use of TVUS to measure follicular growth. Details of COH protocols were not available for every study, and they differed in the use of agonist or antagonist gonadotrophin releasing hormone (GnRH), human menopausal gonadotrophin (hMG) or recombinant follicle stimulating hormone (rFSH). Three studies based the protocol used and dose according to individual response (Golan 1994) or as preferred or determined by the clinicians (Strawn 2007;Wiser 2012). In one study, all women had hormonal assays and ultrasound examinations but the results were only made known to the clinicians after oocyte retrieval (Golan 1994).
Outcomes
Primary outcomes: none of the six included studies reported the primary outcome of live birth rate. One study reported the ongoing pregnancy rate per cycle initiated (Strawn 2007).
Secondary outcomes: four studies reported the outcome clinical pregnancy rate per woman (Aguirre 2010; Lass 2003; Rongieres 2006; Wiser 2012). Two studies reported pregnancy rate per oocyte retrieval (Golan 1994; Strawn 2007) and they were excluded from our analysis as no further details were provided when the authors were contacted. Five studies reported the number of oocytes retrieved per woman (Aguirre 2010; Golan 1994; Lass 2003; Strawn 2007; Wiser 2012). Three studies reported the number of cycle cancellations: due to OHSS (Strawn 2007), insufficient treatment response (Wiser 2012), and one study reported that cycle cancellation was similar in both arms but no data were presented (Rongieres 2006). One study presented the likely costs which might be saved by avoiding serum hormone determination (Golan 1994). All six studies reported the number of cases of OHSS.
Excluded studies
We excluded three studies. One was a quasi‐randomised study (Schindler 2001); one was not a randomised study (Kably Ambe 1994); and one RCT (Raine‐Fenning 2010) compared combined monitoring with 3D ultrasound (US) versus 2D US (see Characteristics of excluded studies).
Risk of bias in included studies
See Risk of bias in included studies; Figure 2; Figure 3.
2.
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.
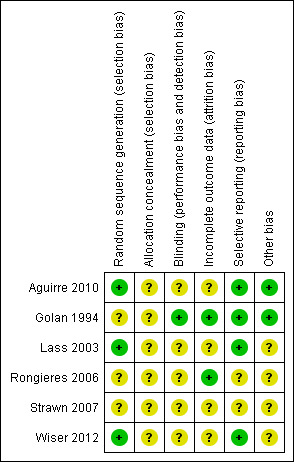
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Three studies were at low risk of selection bias related to random sequence generation as they used computer randomisation (Aguirre 2010; Lass 2003; Wiser 2012). In one study women were randomised by 'drawing of lots' (Rongieres 2006) and the methods of randomisation were not described in two studies (Golan 1994; Strawn 2007). These three studies were considered to be at an unclear risk of bias.
For allocation concealment, two studies used sealed envelopes (Wiser 2012) with unique identification (ID) (Lass 2003) but did not give details whether the sealed envelopes were consecutively numbered and opaque, so they were rated as at unclear risk of bias. Four studies did not describe allocation concealment and were therefore at unclear risk of this bias (Aguirre 2010; Golan 1994; Rongieres 2006; Strawn 2007). The authors were contacted but they were either not able to provide these methodological details or did not respond to our enquiry.
Blinding
For the women participating in the studies, we did not consider that blinding was likely to influence the risk of performance bias for the IVF outcomes assessed. However, there was potential for performance and detection bias in the outcomes of fertilisation and subsequent clinical pregnancy when the operators were not blinded to the allocation. One study reported that patients were blinded for the allocation (Lass 2003) but did not describe the method. The other three studies did not report methods of patient blinding (Aguirre 2010; Golan 1994; Wiser 2012).
In one study, women in the intervention and control groups received combined monitoring and results of estradiol levels were only made known to the clinicians after oocyte retrieval (Golan 1994).
Incomplete outcome data
Analysis of outcomes based on the ITT principle was conducted in one study (Golan 1994) where data were analysed as participants were randomised. Nine women had their treatment discontinued due to risk of OHSS: five from the TVUS group and three from the combined group (Lass 2003). Four women were excluded from the study due to non‐adherence to the protocol (Aguirre 2010), and one women in the TVUS group was excluded due to insufficient response to treatment (Wiser 2012). There was no report of attrition in one study (Rongieres 2006).
For the reporting of number of oocytes retrieved per woman, it was not clear from the included studies that the means for the number of oocytes were reported correctly, that is including zero counts for those women with cancelled cycles, as none of the included studies provided this information.
Selective reporting
All six studies reported outcomes pre‐specified in the methods section. One study reported comparable cycle cancellation rates and mean numbers of oocytes retrieved between the two arms but no data were presented (Rongieres 2006). Some outcomes were not of interest in this review, such as fertilisation rate, implantation rate, miscarriage rate, ectopic pregnancy rate, number of embryos transferred, duration of hMG treatment, number of hMG ampoules used and endometrial thickness, and these data were neither extracted nor analysed.
Other potential sources of bias
Use of a prospective power calculation was not reported in five studies (Aguirre 2010; Golan 1994; Rongieres 2006; Strawn 2007; Wiser 2012).
Defintions of clinical pregnancy were reported in three studies (Lass 2003; Rongieres 2006; Wiser 2012) but none of the six studies provided a definition of OHSS or clarified whether the number of oocytes retrieved per woman was calculated to include zero count.
Another potential source of bias, imbalance in baseline characteristics, was deemed at low risk in four included studies (Aguirre 2010; Golan 1994; Lass 2003; Wiser 2012) which reported similar baseline characteristics in relation to age, body mass index (BMI) and causes of infertility between the intervention and the control groups. Any baseline imbalance could be considered as a possible indicator of allocation bias. Two studies provided demographic details which were comparable between the two arms but there was no information on the causes of infertility (Rongieres 2006; Strawn 2007).
Ovarian stimulation protocols differed between the two arms in three studies: according to the individual’s response (Golan 1994) and the physician’s preference (Strawn 2007; Wiser 2012).
Effects of interventions
See: Table 1
See Table 1.
Transvaginal ultrasound (TVUS) only versus TVUS plus serum estradiol concentrations
Primary outcome
Live birth rate
None of the six included studies reported our primary outcome of live birth rate.
Secondary outcomes
1. Pregnancy rate
There was no evidence of a difference between ovarian monitoring with TVUS only and ovarian monitoring with TVUS plus estradiol measurement on the clinical pregnancy rate per woman. This outcome was reported in four studies (19/35 versus 14/35, 46/148 versus 49/149, 26/88 versus 30/97, and 19/34 versus 12/31, respectively; OR 1.10; 95% CI 0.79 to 1.54; N = 617; I² = 5%; low quality evidence) (Analysis 1.1, Figure 4) (Aguirre 2010; Lass 2003; Rongieres 2006; Wiser 2012). This suggested that compared with women with a 34% chance of clinical pregnancy using monitoring with TVUS plus serum estradiol, the clinical pregnancy rate in women using TVUS only was between 29% and 44%. Two studies measured the pregnancy rate per oocyte retrieval only and so were excluded from the analysis (Golan 1994; Strawn 2007).
1.1. Analysis.
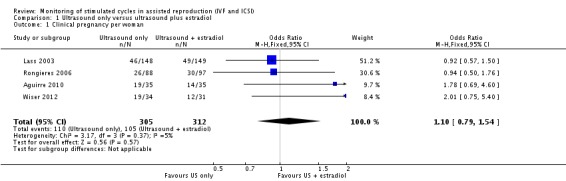
Comparison 1 Ultrasound only versus ultrasound plus estradiol, Outcome 1 Clinical pregnancy per woman.
4.
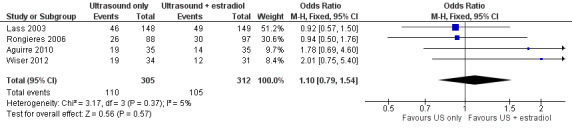
Forest plot of comparison: 1 Ultrasound only versus ultrasound plus estradiol, outcome: 1.1 Clinical pregnancy per woman.
2. Number of oocytes recovered per woman
There was no evidence of a difference in the mean number of oocytes retrieved between the two arms in the five studies (MD 9.9 (SD 3.1) versus 10.9 (SD 4.2), 13.4 (SD 7.5) versus 11.7 (SD 8.4), 11.7 (SD 5.9) versus 11.4 (SD 6.1), 11 (SD 7.0) versus 9 (SD 4.0), and 11.7 (SD 8.0) versus 10.0 (SD 5.5), respectively; MD 0.32; 95% CI ‐0.60 to 1.24; N = 596; I² = 17%; low quality evidence) (Analysis 1.2, Figure 5) (Aguirre 2010; Golan 1994; Lass 2003; Strawn 2007; Wiser 2012).
1.2. Analysis.
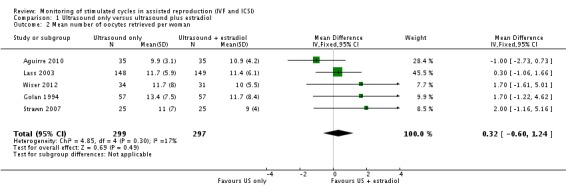
Comparison 1 Ultrasound only versus ultrasound plus estradiol, Outcome 2 Mean number of oocytes retrieved per woman.
5.
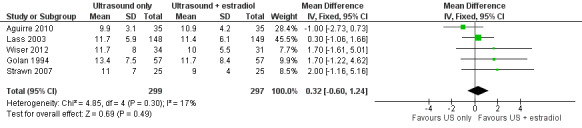
Forest plot of comparison: 1 Ultrasound only versus ultrasound plus estradiol, outcome: 1.2 Mean number of oocytes retrieved per woman.
One study reported a non‐significant difference in the number of oocytes retrieved but no data were presented (Rongieres 2006).
3. Adverse outcomes
3.1 Rate of ovarian hyperstimulation syndrome (OHSS), including severe OHSS
No incidence of OHSS was reported in two studies (Aguirre 2010; Wiser 2012). Combining the results of six studies, we found no difference in the total cases of OHSS between the two arms (0/35 versus 0/35, 0/34 versus 0/31, 1/25 versus 1/25, 4/57 versus 3/57, 7/148 versus 5/149, and 2/88 versus 5/97, respectively; OR 1.03; 95% CI 0.48 to 2.20; six studies; N = 781; I² = 0%; low quality evidence) (Analysis 1.3, Figure 6) (Aguirre 2010; Golan 1994; Lass 2003; Rongieres 2006; Strawn 2007; Wiser 2012), suggesting that compared with women with a 4% chance of OHSS using monitoring with TVUS plus serum estradiol, the OHSS rate in women monitored by TVUS only was between 2% and 8%.
1.3. Analysis.
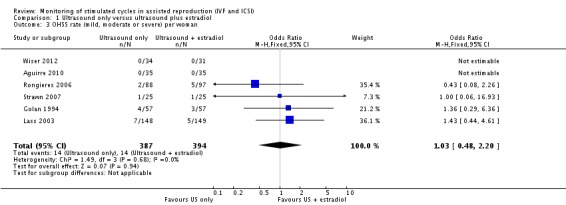
Comparison 1 Ultrasound only versus ultrasound plus estradiol, Outcome 3 OHSS rate (mild, moderate or severe) per woman.
6.
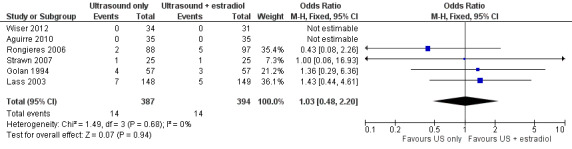
Forest plot of comparison: 1 Ultrasound only versus ultrasound plus estradiol, outcome: 1.3 OHSS rate (mild, moderate or severe) per woman.
Of the 28 cases of OHSS reported in all the six studies there were four cases of severe OHSS, which showed no difference between the two arms (1/57 versus 1/57 and 2/148 versus 0/149, respectively) (Golan 1994; Lass 2003).
3.2 Number of cancelled cycles
Three studies reported this outcome. The cycle cancellation rate was similar in the two arms of two studies (0/34 versus 1/31, 1/25 versus 1/25; OR 0.57; 95% CI 0.07 to 4.39; N = 115; I² = 0%; low quality evidence) (Analysis 1.4, Figure 7) (Strawn 2007; Wiser 2012). The third study reported comparable cancellation rates between the two arms but no data were presented (Rongieres 2006).
1.4. Analysis.
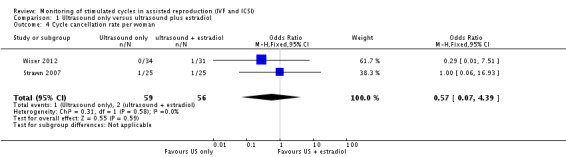
Comparison 1 Ultrasound only versus ultrasound plus estradiol, Outcome 4 Cycle cancellation rate per woman.
7.
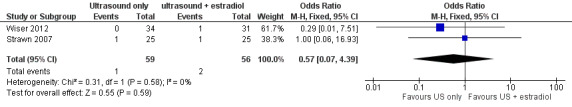
Forest plot of comparison: 1 Ultrasound only versus ultrasound plus estradiol, outcome: 1.4 Cycle cancellation rate per woman.
4. Costs of monitoring with TVUS versus monitoring with TVUS plus serum estradiol concentration
None of the six studies performed an economic analysis on the cost‐effectiveness of the two different types of ovarian monitoring. However, one study (Golan 1994) suggested that US monitoring could be highly cost‐effective and avoiding serum hormone determination might save over USD 150 (1994 prices) in each cycle and compensate for the cost of the GnRH analogue. No further details were available from the studies or the authors.
Discussion
Summary of main results
There were no data on live births. With respect to pregnancy outcomes, in pooling the data of these six trials involving 781 women we found no evidence that cycle monitoring by TVUS alone was any less effective or efficacious than combined monitoring by TVUS plus estradiol assay. However, this interpretation was limited by the imprecision of the findings and the overall low quality of the included studies.
Overall completeness and applicability of evidence
Although the pooled results of these six studies suggest that TVUS alone may potentially be a satisfactory monitoring tool for women undergoing COH for IVF or ICSI, the overall completeness and applicability of the evidence is limited.
None of the studies reported our primary outcome of live births and all of the studies were of low methodological quality. One weakness was the variability of the inclusion criteria for participants. The studies included women undergoing their first IVF attempts (Golan 1994; Wiser 2012) as well as women who had already received three previous IVF attempts (Aguirre 2010; Lass 2003). Women for both IVF and ICSI were included in one study (Rongieres 2006) and women for ICSI were excluded in one study (Lass 2003), as were women with previous serious OHSS (Rongieres 2006). Women undergoing ICSI (that is those with partners with male factor problems) may be younger than those receiving IVF. These different patient selection criteria may have led to bias in the results due to different responses to treatment. Excluding those with previous OHSS may lead to a spuriously lower proportion of women with this condition in a subsequent cycle. Women were enrolled for a single treatment cycle (Lass 2003), for more than one cycle (Aguirre 2010; Rongieres 2006), and three studies did not report the number of cycles (Golan 1994; Strawn 2007; Wiser 2012). The different inclusion criteria for women participating in the studies included in this review limit its generalisability.
Likewise, the criteria for stimulation protocols varied between the studies and it is not clear how this difference would influence the outcomes. In three studies all women were treated with a standardised protocol (Aguirre 2010; Golan 1994; Lass 2003) and in two trials differing protocols were offered depending on clinical assessment and physician preference (Strawn 2007; Wiser 2012). One study described a standardised protocol with all women receiving GnRH analogues initially and with 35% to 36% of the women receiving further GnRH antagonists depending on predefined criteria of estradiol level or presence of one follicle ≥ 13 mm (Rongieres 2006). Furthermore, the timing of TVUS in cycles varied between studies though the significance of this is uncertain and likely reflects the individual local protocols.
There was a lack of definitions for the outcomes assessed. For the three studies which presented the definitions of clinical pregnancy (Lass 2003; Rongieres 2006; Wiser 2012) they differed, ranging from 'fetal sac (with or without fetal heart activity) visualised by US on day 28 to 42 after hCG' (Lass 2003) to 'as confirmed by a fetal heartbeat determined by TVUS; approximately 32 days after embryo transfer' (Wiser 2012) and 'presence of an intra‐uterine gestational sac at US' (Rongieres 2006). The definition of OHSS was not presented in any of the studies and it was uncertain if the reported cases of 'severe' OHSS in two studies (Golan 1994; Lass 2003) were similar in nature. This lack of uniformity and consistency in outcomes definitions is an important shortcoming as there is potential for the assessed outcomes to be different, thus affecting the generalisability and applicability of the evidence.
Some studies either did not report or were unable to provide information to allow computation of outcomes 'per woman' when 'per cycle' data only were reported. This resulted in two studies being excluded from the analysis for the outcome on pregnancy rate (Golan 1994; Strawn 2007), further limiting the evidence base, its generalisability and applicability. Our attempts to contact the authors were unsuccessful and we received no response.
Cycle monitoring with both TVUS and estradiol measurement is likely to involve higher costs (to cover technicians and laboratory costs, outpatient attendance) when compared with TVUS alone. Hormonal assays involve repeated venepuncture, which may cause stress and anxiety to women. However, it is unclear if women would be more satisfied with combined monitoring and because of a placebo effect with the perception that they are being monitored more closely. In future research it would be useful to know which monitoring protocol women were more satisfied with.
Quality of the evidence
Using GRADE methods, the overall quality of the evidence was rated as low for all comparisons (Table 1). The main reasons for downgrading the quality of the evidence were risk of bias and imprecision. The methodological quality of the included studies was poor. This was largely due to some studies failing to describe their methods in adequate detail, such as methods of randomisation and allocation concealment and patient characteristics, resulting in the small quantity of relevant usable data that was available. In addition, five of the six studies did not conduct data analyses based on the ITT principle. Our efforts to contact study authors for further information were unsuccessful.
The addition of new trials in this update enables the aggregation of outcomes data, especially for pregnancy rate per woman. The methodological quality of the trials was variable (Figure 2, Figure 3). Population and treatment characteristics could influence the success of the IVF treatment. It is difficult to determine whether or not there was significant clinical heterogeneity as there was a lack of methodological details, particularly in the pilot study presented as a conference abstract (Strawn 2007). Overall, the included studies appeared to investigate a shared research question with the same intervention and comparator. Five of the six included studies took place in assisted reproduction units (Aguirre 2010; Golan 1994; Lass 2003; Rongieres 2006; Wiser 2012).
Blinding was variably reported in the studies. Women may feel either anxious or reassured if they receive combined monitoring and in this context an unblinded design would be potentially beneficial to measure certain outcomes. However, women's perception of anxiety or reassurance from venepuncture for estradiol measurement is unlikely to influence live birth and pregnancy rates. Assessor blinding would reduce performance and detection bias in oocyte retrieval and embryo transfer when they are not aware of the women's allocation status. In one study, women in the intervention and control groups received combined monitoring and results of the estradiol levels were made known to the clinicians after oocyte retrieval (Golan 1994). Studies whereby women are not blinded to their monitoring regime may be of clinical interest in the future.
There was a possibility that some of the randomised women may not have reached oocyte recovery stage due to cycles cancelled for inappropriate COH (either an under or over response). This is important in trials where ovarian response may be affected by the monitoring regimen. In one study (Lass 2003) nine women left the study before the hCG injection, six due to failure of human rFSH treatment, one because of risk of OHSS, one received saline instead of hCG, and one woman failed to follow the drug protocol. In this study a total of 42 women (14.1%) did not reach the embryo transfer stage, although the reasons for discontinuing treatment did not differ between the two groups.
For the reporting of number of oocytes retrieved per woman, it was not clear from the included studies that the means reported for oocytes correctly included zero counts for those with cancelled cycles. This would have the effect of both increasing the mean and reducing the standard deviation (SD), therefore exaggerating the statistical significance of the comparisons. Lack of this information from the studies and the authors increases the uncertainty of data reliability. In addition, the interpretation of US may potentially differ because it involves some degree of inter‐observer variability, especially in units where successive scans may be performed by different personnel.
We contacted all authors for missing and additional information about their studies but received no response. One author responded that the data were no longer available due to the long time lapse from when the study was conducted, 20 years ago.
Potential biases in the review process
We did not exclude studies on the grounds of language and included one Spanish and one French study. One study was a conference abstract. Despite our efforts to contact authors, we were unable to obtain further information from them and some bias in the review process could have arisen due to the inclusion of trials with insufficient information on methods and outcome data and where authors did not respond to our enquiries.
Agreements and disagreements with other studies or reviews
A recent systematic review of six studies reported that, compared to TVUS plus hormonal measurement, monitoring COH with TVUS only was unlikely to substantially change the chance of achieving clinical pregnancy outcomes in women undergoing IVF treatment (Martins 2013). They included five studies, all of which were included in this review except for one (Strawn 2007).
One RCT that compared combined monitoring with traditional two‐dimensional (2D) US versus automated three‐dimensional (3D) US reported comparable outcomes for clinical pregnancy, number of oocytes retrieved and number of fertilised oocytes (Raine‐Fenning 2010). This suggests that traditional 2D US is no more or less efficient than 3D ultrasound for monitoring stimulated cycles in assisted reproduction.
A recent study to validate the saliva‐based estradiol assay for intra‐ and inter‐assay variability found that it was associated with improved patient satisfaction and reduced anxiety (Zimon 2013), and this method may provide an equivalent but 'patient‐friendly' alternative to serum‐based assessment. However, in the absence of any effectiveness and cost‐effectiveness data from RCTs comparing the effects of serum estradiol and saliva‐based estradiol assays, the non‐invasive saliva‐based estradiol assay would still incur anxiety and costs. Serum estradiol may be perceived as a more reliable parameter, especially in women at risk of OHSS in whom decisions on cycle cancellation or elective freezing of oocytes or embryos may need to be taken. However, as the findings of this review show that current evidence is limited, high quality RCTs to assess monitoring methods would be welcome. A cycle monitoring protocol including both US and serum estradiol concentrations may need to be retained as precautionary good clinical practice and in a subset of women as a confirmatory test to identify those at high risk of OHSS, such as women with a history of OHSS or with polycystic ovaries, those exposed to ovulation induction, and younger women undergoing assisted reproduction techniques (Jenkins 2006).
No formal cost analysis was reported in these studies. The studies reviewed were not designed to test the cost‐effectiveness of the two interventions. However, it was suggested that avoiding serum hormone determination might save over USD 150 (at early 1990 prices) in each cycle and this would compensate for the cost of the GnRHa (Golan 1994).
Authors' conclusions
Implications for practice.
This review update found no evidence from randomised controlled trials to suggest that combined monitoring by TVUS and serum estradiol is more efficacious than monitoring by TVUS alone, with regard to clinical pregnancy rates and the incidence of OHSS. The number of oocytes retrieved appeared similar for both monitoring protocols. The data suggest that both of these monitoring methods are safe and reliable. However, these results should be interpreted with caution because the overall quality of the evidence was low. Results were compromised by imprecision and poor reporting of study methodology. A combined monitoring protocol including both TVUS and serum estradiol may need to be retained as precautionary good clinical practice and as a confirmatory test in a subset of women to identify those at high risk of OHSS.
Implications for research.
To find a difference in an important but rare outcome such as OHSS, a large randomised controlled study requiring the recruitment of approximately 5000 women is needed for each randomised arm, and this would pose a great methodological challenge. Based on the quoted risk of OHSS following IVF in Europe of 1% (Ferraretti 2012) and a hypothetical trial of an intervention for reduction of OHSS with a clinically meaningful effect size, meaning to halve this risk (from 1% to 0.5%) with 90% power and 95% confidence (alpha = 0.05), the trial would need 4675 women in each randomised arm. However, an economic evaluation of the costs involved in these two monitoring methods and assessing the views of the women undergoing cycle monitoring would be welcome.
What's new
| Date | Event | Description |
|---|---|---|
| 30 May 2014 | New citation required but conclusions have not changed | New studies included and summary of findings table added. |
| 30 May 2014 | New search has been performed | Four studies added to the review (Aguirre 2010; Rongieres 2006; Strawn 2007; Wiser 2012). |
History
Protocol first published: Issue 2, 2005 Review first published: Issue 2, 2008
| Date | Event | Description |
|---|---|---|
| 15 December 2013 | New citation required and conclusions have changed | Substantive amendment |
| 7 November 2008 | New search has been performed | Converted to new review format. |
Acknowledgements
We are grateful to Marian Showell for her help in updating the search strategy and carrying out the literature search; to Anne Lethaby for translating the Spanish paper and extracting data; and in particular to Helen Nagels and Jane Marjoribanks for their editorial assistance, help with GRADEpro, general co‐ordination and assisting with teleconferences among authors. We also thank Helen Nagels for her help in translating the French paper and extracting the data.
Appendices
Appendix 1. Menstrual Disorders and Subfertility Group (MDSG) search strategy (20 May 2014)
Keywords CONTAINS "IVF"or "ICSI"or"in vitro fertilisation"or "in vitro fertilization"or "in‐vitro fertilisation procedure"or"in‐vitro fertilisation techniques"or "intracytoplasmic sperm injection"or "Embryo"or "oocytes"or "ovarian hyperstimulation"or "ovarian hyperstimulation syndrome"or"ovarian stimulation"or"ovarian response"or"ovarian stimulation syndrome"
AND
Keywords CONTAINS "ultrasonography" or "ultrasound" or"ultrasound guidance" or "ultrasound, pelvic" or "monitoring" or Title CONTAINS "ultrasonography" or "ultrasound" or"ultrasound guidance" or "ultrasound, pelvic"or "monitoring"
Appendix 2. Cochrane Central Register of Controlled Trials (CENTRAL) search strategy (April 2014)
1 Ovulation Induction/ (981) 2 Ovarian Hyperstimulation Syndrome/ (140) 3 Ovarian Follicle/ (346) 4 Follicle Stimulating Hormone/ (1546) 5 Luteinizing Hormone/ (1451) 6 Gonadotropins/ (194) 7 (ovulat$ adj5 (induct$ or stimulat$)).tw. (651) 8 (ovar$ adj5 (induct$ or stimulat$ or follicle$)).tw. (1225) 9 ((ovar$ or ovulat$) adj5 (hyperstimulat$ or hyper stimulat$ or hyper‐stimulat$)).tw. (620) 10 gonadotrop?in$.tw. (2694) 11 (luteiniz$ adj5 hormone$).tw. (1225) 12 (stimulat$ adj5 (cycle$ or hormone$)).tw. (2651) 13 or/1‐12 (6658) 14 exp Fertilization in Vitro/ (1609) 15 (IVF or in vitro fertili?ation or in‐vitro fertili?ation or in‐vitro‐fertili?ation).tw. (2758) 16 Intracytoplasmic Sperm Injections/ (394) 17 ((intracytoplasmic or intra‐cytoplasmic) adj5 sperm inject$).tw. (516) 18 ICSI.tw. (801) 19 or/14‐18 (3300) 20 13 and 19 (1492) 21 (ultrasound or ultrasonograph$ or ultra‐sound or ultra‐sonograph$).tw. (10233) 22 ((ovar$ or ovulat$) adj5 (monitor$ or scan$ or measur$)).tw. (390) 23 exp Estradiol/ (3362) 24 ((oestrad$ or estrad$) adj5 (monitor$ or scan$ or measur$)).tw. (437) 25 (hormon$ adj5 (measure$ or monitor$ or scan$)).tw. (1520) 26 exp Ultrasonography/ (6733) 27 echograph$.tw. (160) 28 or/21‐27 (19410) 29 20 and 28 (470) 30 limit 29 to yr="2013 ‐Current" (10)
Appendix 3. Ovid MEDLINE search strategy (20 May 2014)
Database: Ovid MEDLINE(R) In‐Process & Other Non‐Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) <1946 to Present> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 Ovulation Induction/ (8926) 2 Ovarian Hyperstimulation Syndrome/ (1736) 3 Ovarian Follicle/ (15163) 4 Follicle Stimulating Hormone/ (32964) 5 Luteinizing Hormone/ (42967) 6 Gonadotropins/ (8402) 7 (ovulat$ adj5 (induct$ or stimulat$)).tw. (5299) 8 (ovar$ adj5 (induct$ or stimulat$ or follicle$)).tw. (16264) 9 ((ovar$ or ovulat$) adj5 (hyperstimulat$ or hyper stimulat$ or hyper‐stimulat$)).tw. (3965) 10 gonadotrop?in$.tw. (53846) 11 (luteiniz$ adj5 hormone$).tw. (25275) 12 (stimulat$ adj5 (cycle$ or hormone$)).tw. (45955) 13 or/1‐12 (145856) 14 exp Fertilization in Vitro/ (27947) 15 (IVF or in vitro fertili?ation or in‐vitro fertili?ation or in‐vitro‐fertili?ation).tw. (24620) 16 Intracytoplasmic Sperm Injections/ (4600) 17 ((intracytoplasmic or intra‐cytoplasmic) adj5 sperm inject$).tw. (5141) 18 ICSI.tw. (5486) 19 or/14‐18 (36686) 20 13 and 19 (10815) 21 (ultrasound or ultrasonograph$ or ultra‐sound or ultra‐sonograph$).tw. (216336) 22 ((ovar$ or ovulat$) adj5 (monitor$ or scan$ or measur$)).tw. (4931) 23 exp Estradiol/ (74460) 24 ((oestrad$ or estrad$) adj5 (monitor$ or scan$ or measur$)).tw. (3412) 25 (hormon$ adj5 (measure$ or monitor$ or scan$)).tw. (12149) 26 exp Ultrasonography/ (247866) 27 echograph$.tw. (8633) 28 or/21‐27 (470968) 29 20 and 28 (3305) 30 randomized controlled trial.pt. (373788) 31 controlled clinical trial.pt. (88373) 32 randomized.ab. (293663) 33 randomised.ab. (58854) 34 placebo.tw. (158182) 35 clinical trials as topic.sh. (169943) 36 randomly.ab. (212697) 37 trial.ti. (126393) 38 (crossover or cross‐over or cross over).tw. (60653) 39 or/30‐38 (943680) 40 exp animals/ not humans.sh. (3939073) 41 39 not 40 (870261) 42 29 and 41 (511) 43 (201310$ or 201311$ or 201312$).ed. (232178) 44 2014$.ed. (381204) 45 2014$.dp. (372749) 46 43 or 44 or 45 (913311) 47 42 and 46 (13)
Appendix 4. EMBASE search strategy (20 May 2014)
Database: Embase < 1980 to 2014 Week 20> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 Ovulation Induction/ (10545) 2 Ovarian Hyperstimulation Syndrome/ (6330) 3 Ovarian Follicle/ (13554) 4 Follicle Stimulating Hormone/ (40096) 5 Luteinizing Hormone/ (47094) 6 Gonadotropins/ (21901) 7 (ovulat$ adj5 (induct$ or stimulat$)).tw. (6292) 8 (ovar$ adj5 (induct$ or stimulat$ or follicle$)).tw. (19113) 9 ((ovar$ or ovulat$) adj5 (hyperstimulat$ or hyper stimulat$ or hyper‐stimulat$)).tw. (5426) 10 gonadotrop?in$.tw. (54983) 11 (luteiniz$ adj5 hormone$).tw. (23538) 12 (stimulat$ adj5 (cycle$ or hormone$)).tw. (47601) 13 or/1‐12 (161565) 14 exp Fertilization in Vitro/ (38208) 15 (IVF or in vitro fertili?ation or in‐vitro fertili?ation or in‐vitro‐fertili?ation).tw. (33773) 16 exp Intracytoplasmic Sperm Injection/ (12528) 17 ((intracytoplasmic or intra‐cytoplasmic) adj5 sperm inject$).tw. (6555) 18 ICSI.tw. (9464) 19 or/14‐18 (52926) 20 13 and 19 (16720) 21 (ultrasound or ultrasonograph$ or ultra‐sound or ultra‐sonograph$).tw. (287310) 22 ((ovar$ or ovulat$) adj5 (monitor$ or scan$ or measur$)).tw. (5859) 23 ((oestrad$ or estrad$) adj5 (monitor$ or scan$ or measur$)).tw. (3683) 24 (hormon$ adj5 (measure$ or monitor$ or assay$)).tw. (16839) 25 exp ultrasound/ (101105) 26 exp echography/ (501808) 27 echograph$.tw. (10048) 28 (hormon$ adj5 (measure$ or monitor$ or scan$)).tw. (13391) 29 or/21‐28 (678508) 30 20 and 29 (3133) 31 Clinical Trial/ (830705) 32 Randomized Controlled Trial/ (341422) 33 exp randomization/ (61977) 34 Single Blind Procedure/ (18225) 35 Double Blind Procedure/ (113073) 36 Crossover Procedure/ (38825) 37 Placebo/ (238864) 38 Randomi?ed controlled trial$.tw. (97655) 39 Rct.tw. (13725) 40 random allocation.tw. (1297) 41 randomly allocated.tw. (20015) 42 allocated randomly.tw. (1916) 43 (allocated adj2 random).tw. (710) 44 Single blind$.tw. (14129) 45 Double blind$.tw. (139503) 46 ((treble or triple) adj blind$).tw. (361) 47 placebo$.tw. (195795) 48 prospective study/ (249423) 49 or/31‐48 (1350956) 50 case study/ (25746) 51 case report.tw. (256378) 52 abstract report/ or letter/ (888025) 53 or/50‐52 (1164600) 54 49 not 53 (1313533) 55 30 and 54 (645) 56 (201310$ or 201311$ or 201312$).em. (67090) 57 2014$.em. (694933) 58 2014$.dp. (41843) 59 56 or 57 or 58 (764192) 60 55 and 59 (20)
Appendix 5. PsycINFO search strategy (20 May 2014)
Database: PsycINFO <1806 to May Week 2 2014> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 exp Reproductive Technology/ or exp Ovulation/ or exp Infertility/ (2907) 2 (ovulat$ adj5 (induct$ or stimulat$)).tw. (50) 3 (ovar$ adj5 (induct$ or stimulat$ or follicle$)).tw. (211) 4 ((ovar$ or ovulat$) adj5 (hyperstimulat$ or hyper stimulat$ or hyper‐stimulat$)).tw. (9) 5 (IVF or in vitro fertili?ation or in‐vitro fertili?ation or in‐vitro‐fertili?ation).tw. (613) 6 ((intracytoplasmic or intra‐cytoplasmic) adj5 sperm inject$).tw. (37) 7 ICSI.tw. (44) 8 or/1‐7 (3282) 9 (ultrasound or ultrasonograph$ or ultra‐sound or ultra‐sonograph$).tw. (2679) 10 ((ovar$ or ovulat$) adj5 (monitor$ or scan$ or measur$)).tw. (119) 11 ((oestrad$ or estrad$) adj5 (monitor$ or scan$ or measur$)).tw. (159) 12 (hormon$ adj5 (measure$ or monitor$ or scan$)).tw. (1100) 13 exp Ultrasound/ (983) 14 echograph$.tw. (50) 15 or/9‐14 (4266) 16 8 and 15 (51) 17 random.tw. (40490) 18 control.tw. (314333) 19 double‐blind.tw. (17824) 20 clinical trials/ (7555) 21 placebo/ (3760) 22 exp Treatment/ (578758) 23 or/17‐22 (883642) 24 16 and 23 (21) 25 limit 24 to yr="2013 ‐Current" (2)
Appendix 6. CINAHL search strategy (20 May 2014)
| # | Query | Results |
| S27 | S14 AND S26 | 117 |
| S26 | S22 AND S25 | 345 |
| S25 | S23 OR S24 | 1,505 |
| S24 | (MH "Ovarian Hyperstimulation Syndrome") OR "ovarian hyperstimulation syndrome" | 202 |
| S23 | TX ovulat* | 1,373 |
| S22 | S15 OR S16 OR S17 OR S18 OR S19 OR S20 OR S21 | 450,401 |
| S21 | TX echograph* | 173 |
| S20 | TX Measur* | 350,807 |
| S19 | TX scanned | 1,212 |
| S18 | TX scanning | 6,423 |
| S17 | TX scan | 12,299 |
| S16 | TX monitor* | 82,820 |
| S15 | (MH "Ultrasonography, Prenatal") OR (MH "Ultrasonography") OR "ultrasound" | 31,069 |
| S14 | S1 OR S2 or S3 or S4 OR S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 OR S12 OR S13 | Display |
| S13 | TX allocat* random* | Display |
| S12 | (MH "Quantitative Studies") | Display |
| S11 | (MH "Placebos") | Display |
| S10 | TX placebo* | Display |
| S9 | TX random* allocat* | Display |
| S8 | (MH "Random Assignment") | Display |
| S7 | TX randomi* control* trial* | Display |
| S6 | TX ( (singl* n1 blind*) or (singl* n1 mask*) ) or TX ( (doubl* n1 blind*) or (doubl* n1 mask*) ) or TX ( (tripl* n1 blind*) or (tripl* n1 mask*) ) or TX ( (trebl* n1 blind*) or (trebl* n1 mask*) ) | Display |
| S5 | TX ( (trebl* n1 blind*) or (trebl* n1 mask*) ) | Display |
| S4 | TX ( (trebl* n1 blind*) or (trebl* n1 mask*) ) | Display |
| S3 | TX clinic* n1 trial* | Display |
| S2 | PT Clinical trial | Display |
| S1 | (MH "Clinical Trials+") | Display |
Data and analyses
Comparison 1. Ultrasound only versus ultrasound plus estradiol.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Clinical pregnancy per woman | 4 | 617 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.79, 1.54] |
| 2 Mean number of oocytes retrieved per woman | 5 | 596 | Mean Difference (IV, Fixed, 95% CI) | 0.32 [‐0.60, 1.24] |
| 3 OHSS rate (mild, moderate or severe) per woman | 6 | 781 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.48, 2.20] |
| 4 Cycle cancellation rate per woman | 2 | 115 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.57 [0.07, 4.39] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Aguirre 2010.
| Methods | Randomisation: randomisation list Allocation concealment: not reported Blinding of participants/investigators: no Blinding of assessors: not reported Number randomised: 70 Number analysed: 66 Intention‐to‐treat analysis: no Power and sample calculation not reported Duration of study: conducted from January 2006 to June 2007 |
|
| Participants | Women undergoing IVF, between 25 to 36 years of age with normal follicular reserve, BMI and with less than 3 previous IVF cycles Mean age: 32‐33 years Cause of infertility: female factors, male factors, mixed factors, unexplained Similar demographic and infertility characteristics at baseline (age, BMI, basal FSH and causes of infertility) | |
| Interventions | 1. Control: monitoring of ovarian induction by TVUS plus serum estradiol determination (n=31) 2. Intervention: monitoring of ovarian induction by TVUS only (n=35) Ovarian stimulation protocol: GnRH agonist, triptorelin, followed by rFSH |
|
| Outcomes | 1. Primary: live birth rate not reported
2. Secondary: clinical pregnancy rate/woman, multiple pregnancy rate/woman, number of oocytes retrieved/woman, number of cases of OHSS Other outcomes reported: ICSI; fertilisation rate, implantation rate, miscarriage rate, ectopic pregnancy rate, number of embryos transferred |
|
| Notes | Country: Spain Single centre Unidad de Reproduccion Asistida Clinica Montpellier, Espana Funding: Schering Plough Definition of pregnancy not documented Definition of OHSS not reported Four women excluded due to non‐adherence to treatment protocol Number of cycles: 42 cycles for control group; 48 cycles for intervention group Paper in Spanish Author contacted: no response and no data details were available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation list, no details given |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Blinding of participants and assessors not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Four women excluded due to non‐adherence to treatment protocol |
| Selective reporting (reporting bias) | Low risk | All pre‐specified outcomes reported |
| Other bias | Low risk | Comparable demographic and infertility characteristics at baseline |
Golan 1994.
| Methods | Randomisation: methods unclear Allocation concealment: methods unclear Blinding of participants/investigators: patients not blind Blinding of assessors: yes Number randomised: 114 Number analysed: 114 Intention‐to‐treat analysis: yes Power and sample calculation not reported Duration of study: not stated |
|
| Participants | Women admitted for first IVF attempts Mean age: 30‐31 years Cause of infertility: tubal disease, male factor, endometriosis, unexplained Similar demographic and infertility characteristics at baseline (age, mean duration of infertility, and indications for IVF treatment) | |
| Interventions | 1. Control: monitoring of ovarian induction by TVUS plus serum estradiol determination (n=57) 2. Intervention: monitoring of ovarian induction by TVUS only (n=57) Ovarian stimulation protocol: human menopausal gonadotrophin (hMG), adjusting dose according to individual response |
|
| Outcomes | 1. Primary: live birth rate not reported
2. Secondary: pregnancy rate/oocyte retrieval, number of oocytes retrieved/woman, number of cases of OHSS, costs saved Other outcomes reported: duration of hMG treatment, number of hMG ampoules used, serum estradiol, embryos achieved, number of embryo replaced |
|
| Notes | Country: Israel Single centre Assaf Harofeh Medical Centre, affiliated to Sackler Faculty of Medicine, Tel Aviv University, Israel Funding: not stated Definition of pregnancy not documented Definition of OHSS not reported Number of cycles not reported Author contacted: responded that no further data details were available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Patients were randomly divided into 2 groups, no details given |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Low risk | Both control and intervention groups received ultrasonography and serum estradiol measurement, the results were only made known to the clinicians following oocyte retrieval |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All women randomised included in analysis |
| Selective reporting (reporting bias) | Low risk | All pre‐specified outcomes reported |
| Other bias | Low risk | Comparable demographic and infertility characteristics at baseline |
Lass 2003.
| Methods | Randomisation: computer‐generated randomisation Allocation concealment: individual sealed envelopes with unique identification Blinding of participants/investigators: patient‐blind Blinding of assessors: no Number randomised: 297 Number analysed: 288 Intention‐to‐treat analysis: no Power and sample calculation described Duration of study: one cycle only |
|
| Participants | Women undergoing IVF treatment who had no more than 3 previous unsuccessful attempts; women with PCOS were included; ICSI excluded
Mean age: 23‐39 years
Cause of infertility: not reported Similar demographic and infertility characteristics at baseline (age, BMI, duration of infertility and no. of previous ART treatments) |
|
| Interventions | 1. Control: monitoring of ovarian induction by TVUS plus serum estradiol determination (n=148)
2. Intervention: monitoring of ovarian induction by TVUS only (n=149) Ovarian stimulation protocol: pituitary down‐regulation achieved by daily injection of buserelin; recombinant human FSH dose according to clinical practice |
|
| Outcomes | 1. Primary: livebirth rate not reported
2. Secondary: clinical pregnancy rate/woman, number of oocytes retrieved/woman, number of cases of OHSS Other outcomes reported: dose of Gonal‐F, endometrial thickness, number of follicles, number of embryos transferred |
|
| Notes | Country: UK Multi‐centres: 4 assisted conception units Bourn Hall Clinic, Cambridge, UK Funding: author worked for Serono Definition of pregnancy: 'a pregnancy in which a fetal sac (with or without fetal heart activity) was visualized by ultrasound on day 28‐42 after hCG administration.' Definition of OHSS: not reported Nine women had their treatment discontinued due to the risk of OHSS, three from the estradiol plus ultrasound group and five from the ultrasound‐only group A total of 42 women (14.1%) did not reach the embryo transfer stage, 19 in control group and 23 in intervention group, reasons for discontinuing treatment did not differ between the two groups One cycle only Author contacted: no response and no data details were available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation list |
| Allocation concealment (selection bias) | Unclear risk | Randomisation code for each patient provided in Individual sealed envelopes with unique ID |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Participants blind Blinding of assessors not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | A total of 9 women (3%) did not complete study |
| Selective reporting (reporting bias) | Low risk | All pre‐specified outcomes reported |
| Other bias | Unclear risk | Causes of infertility not reported |
Rongieres 2006.
| Methods | Randomisation: by 'drawing of lots' Allocation concealment: methods unclear Blinding of participants/investigators: not reported Blinding of assessors: not reported Number randomised: 185 Number analysed: 185 Intention‐to‐treat analysis: yes Power and sample calculation: not described Duration of study: not reported |
|
| Participants | Women undergoing IVF and ICST treatment; women with previous serious OHSS excluded
Mean age: 24‐42 years
Cause of infertility: not reported Similar demographic at baseline (age, BMI, techniques (IVF, ICSI, TESSA)) |
|
| Interventions | 1. Control: monitoring of ovarian induction by TVUS plus serum estradiol determination (n=97)
2. Intervention: monitoring of ovarian induction by TVUS only (n=88) Ovarian stimulation protocol: GnRH agonist long protocol. 35% to 36% of the women also received GnRH antagonists depending on predefined criteria of estradiol level or presence of one follicle ≥ 13 mm |
|
| Outcomes | 1. Primary: live birth rate not reported
2. Secondary: clinical pregnancy rate/woman, number of oocytes retrieved/woman, rate of cycle cancellation/woman, number of cases of OHSS Other outcomes reported: units of GnRH used, fertilisation rate |
|
| Notes | Country: France Single centre Centre d'Assistance Médicale à la Procréation, Centre Hospitalier Universitaire, Strasbourg Funding: not reported Definition of pregnancy: 'presence of an intra‐uterine gestational sac at US' Definition of OHSS: not stated Number of cycles: 1.8±1.2 (control), 1.2±1.4 (intervention) Author contacted: no response and data details were available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | 'Drawing of lots', no details |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Blinding of participants and assessors not reported |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All women randomised included in analysis |
| Selective reporting (reporting bias) | Unclear risk | Not reported |
| Other bias | Unclear risk | Causes of infertility not reported |
Strawn 2007.
| Methods | Randomisation: methods unclear Allocation concealment: methods unclear Blinding of participants/investigators: not reported Blinding of assessors: not reported Number randomised: 50 Number analysed: 49 Intention‐to‐treat analysis: no Power and sample calculation not described Duration of study: not reported |
|
| Participants | Women undergoing IVF treatment
Mean age: 32 years
Cause of infertility: not reported Similar demographic characteristics at baseline (weight and BMI) |
|
| Interventions | 1. Control: monitoring of ovarian induction by TVUS plus estradiol assay (n=25)
2. Intervention: monitoring of ovarian induction by TVUS only (n=24) Ovarian stimulation protocol: based on practitioner preference, no details given |
|
| Outcomes | 1. Primary: live birth rate not reported
2. Secondary: ongoing pregnancy rate/cycle, number of oocytes retrieved/woman, cycle cancellation rate/woman, number of cases of OHSS Other outcomes reported: number of units of hMG used, level of estradiol, number of embryos transferred |
|
| Notes | Country: US Funding: Azko‐Nobel Pharamceutical corporation Definition of pregnancy: not reported Number of cycles: not reported One cycle was cancelled in the TVUS arm and one cycle cancelled in the estradiol plus TVUS arm Author contacted: no response and no data details were available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Methods unclear |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Blinding of participants and assessors not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | one cycle was cancelled in the TVUS arm and one cycle cancelled in the estradiol plus TVUS arm |
| Selective reporting (reporting bias) | Unclear risk | Not clear |
| Other bias | Unclear risk | Causes of infertility not reported; stimulation protocol based on practitioner preference |
Wiser 2012.
| Methods | Randomisation: computer‐generated Allocation concealment: sealed envelopes Blinding of participants/investigators: not reported Blinding of assessors: not reported Number randomised: 65 Number analysed: 63 Intention‐to‐treat analysis: no Power and sample calculation not reported Duration of study: conducted from 2007 to 2009 |
|
| Participants | Women undergoing first IVF attempts, younger than age 40 years
Mean age: 31‐32 years
Cause of infertility: male factor, PCOS, endometriosis, mechanical, unexplained Similar demographic and infertility characteristics at baseline (age, BMI, basal FSH, causes of infertility and ovarian reserve) |
|
| Interventions | 1. Control: monitoring of ovarian induction by TVUS plus serum estradiol and progesterone determination (n=30)
2. Intervention: monitoring of ovarian induction by TVUS only (n=33) Ovarian stimulation protocol: either long GnRH agonist protocol or antagonist protocol as determined by the physician |
|
| Outcomes | 1. Primary: live birth rate not reported
2. Secondary: clinical pregnancy rate/woman, number of oocytes retrieved/woman; number of cases of OHSS Other outcomes reported: number of induction days, number of gonadotrophin ampoules used, endometrial thickness, estradiol levels, number of embryos transferred, mean score of leading embryos transferred |
|
| Notes | Country: Israel Single centre Meir Medical Center, Kfar Saba, affiliated to Sackler Faculty of Medicine, Tel Aviv University, Israel Funding: not stated (authors declared no conflict of interest) Definition of clinical pregnancy: confirmed by 'by a fetal heartbeat determined by transvaginal ultrasound approximately 32 days after embryo transfer.' Definition of OHSS: not reported One patient (in the control group) was cancelled due to insufficient response to treatment Number of cycles not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random numbers |
| Allocation concealment (selection bias) | Unclear risk | Each patient chose a sealed envelope containing the randomised assignment to either the study or the control group |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Blinding of participants and assessors not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Two patients excluded after randomisation: one woman conceived spontaneously under suppression with GnRH agonist and one left the clinic before starting treatment One woman (in the control group) was cancelled due to insufficient response to treatment, results did not reflect this |
| Selective reporting (reporting bias) | Low risk | All pre‐specified outcomes reported |
| Other bias | Unclear risk | Comparable demographic and infertility characteristics at baseline, stimulation protocols varied as determined by the physician |
TVUS ‐ Transvaginal ultrasonography; OHSS ‐ Ovarian hyperstimulation syndrome; ICSI ‐ Intra‐cytoplasmic sperm injection; TESE: Testicular sperm extraction; hMG ‐ Human menopausal gonadotrophin; hCG ‐ Human chorionic gonadotrophin; GnRH ‐ Gonadotropin‐releasing hormone; FSH ‐ Follicle stimulating hormone; PCOS ‐ Polycystic ovary syndrome; BMI ‐ body mass index
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Kably Ambe 1994 | This is a non‐randomised trial |
| Raine‐Fenning 2010 | The intervention comprises estradiol measurement plus two‐dimensional ultrasound versus estradiol measurement plus SonoAVC (three‐dimensional ultrasound) and does not satisfy the inclusion criteria of the review |
| Schindler 2001 | This is a quasi‐randomised trial in which women were randomised by month of birth |
Differences between protocol and review
None
Contributions of authors
IK took the lead in writing the draft of the protocol, developed the intended methods of the review, was involved in selecting trials for inclusion, performed independent data extraction and quality assessment of included trials, contacted authors for additional information and was responsible for statistical analyses, and writing the final review.
SB helped to develop the protocol and intended methods of the review, was consultant on clinical issues, made significant contributions to the interpretation of results and improvement of the final review.
AW and AK were involved in selecting trials for inclusion, contacting authors for additional information, performed independent data extraction and quality assessment of included trials. AW contributed to the statistical analyses and interpretation of results, provided clinical input and helped to write the final review.
Sources of support
Internal sources
-
Review authors, Other.
The review authors had no specific funding for this review
External sources
None, Other.
Declarations of interest
None known
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Aguirre 2010 {published data only}
- Aguirre SG, Hevia IG, García‐Carpintero G, DelRosal Ma J. Measuring plasma estradiol levels in in vitro fertilization. Is it useful?. Revista Iberoamericana De Fertilidad y Reproduccion Humana 2010;27(1):43‐9. [Google Scholar]
Golan 1994 {published data only}
- Golan A, Herman A, Soffer Y, Bukovsky I, Ron‐El R. Ultrasonic control without hormone determination for ovulation induction in in‐vitro fertilization/embryo transfer with gonadotrophin‐releasing hormone analogue and human menopausal gonadotrophin. Human Reproduction 1994;9(9):1631‐3. [DOI] [PubMed] [Google Scholar]
Lass 2003 {published data only}
- Lass A, UK Timing of hCG Group. Monitoring of in vitro fertilization‐embryo transfer cycles by ultrasound versus by ultrasound and hormonal levels: a prospective, multicenter, randomized study. Fertility and Sterility 2003;80(1):80‐5. [DOI] [PubMed] [Google Scholar]
Rongieres 2006 {published data only}
- Rongieres C. Monitoring ovarian stimulation: are hormonal assessments necessary?. Journal de Gynécologie, Obstétrique et Biologie de la Reproduction 2006;35:2S39‐41. [PubMed] [Google Scholar]
Strawn 2007 {published data only}
- Strawn EY, Roesler M, Granlund A, Bohling S. In vitro fertilization can be successfully accomplished without routine estradiol monitoring: a randomized pilot study. Fertility and Sterility 2007;88 Suppl 1(P‐114):S147. [Google Scholar]
Wiser 2012 {published data only}
- Wiser A, Gonen O, Ghetler Y, Shavit T, Berkovitz A, Shulman A. Monitoring stimulated cycles during in vitro fertilization treatment with ultrasound only – preliminary results. Gynecological Endocrinology 2012;28(6):429–31. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Kably Ambe 1994 {published data only}
- Kably Ambe A, Carranza Lira S, Serviere Zaragoza C, Espinoza de los Monteros A, Coria Soto I, Alvarado Duran A. The evaluation of the usefulness of ultrasound independent of estradiol as a prognostic criterion for ovular capture and maturity A comparative double‐blind study. Ginecologia y Obstetricia de Mexico 1994;62:288‐91. [PubMed] [Google Scholar]
Raine‐Fenning 2010 {published data only}
- Raine‐Fenning N, Deb S, Jayaprakasan K, Clewes J, Hopkisson J, Campbell B. Timing of oocyte maturation and egg collection during controlled ovarian stimulation: a randomized controlled trial evaluating manual and automated measurements of follicle diameter. Fertility and Sterility 2010;94(1):184‐8. [DOI] [PubMed] [Google Scholar]
Schindler 2001 {published data only}
- Schindler L, Janny L, Canis M, Schubert B, Durakovic N. Oestradiol is useless in IVF monitoring: a prospective study. Human Reproduction 2001;16 Suppl 1(P‐269):204. [Google Scholar]
Additional references
Abdalla 1989
- Abdalla HI, Baber RJ, Leonard T, Kirkland A, Mitchell A, Power M, et al. Timed oocyte collection in an assisted conception programme using GnRH analogue. Human Reproduction 1989;4(8):927‐30. [DOI] [PubMed] [Google Scholar]
Asch 1991
- Asch RH, Li HP, Balmaceda JP, Weckstein LN, Stone SC. Severe ovarian hyperstimulation syndrome in assisted reproductive technology: definition of high risk groups. Human Reproduction 1991;6(10):1395‐9. [DOI] [PubMed] [Google Scholar]
Borase 2012
- Borase H, Mathur R. Ovarian hyperstimulation syndrome: clinical features, prevention and management. Obstetrics, Gynaecology and Reproductive Medicine 2012;22(7):186‐90. [Google Scholar]
Confino 1996
- Confino E, Binor Z, Molo MW, Rawlins R, Balos R, Mullaney K, et al. Sonographically monitored ovarian stimulation for assisted reproduction: a prospective, blind study. Journal of Reproductive Medicine 1996;41(1):7‐10. [PubMed] [Google Scholar]
Delvigne 2002
- Delvigne A, Rozenberg S. Epidemiology and prevention of ovarian hyperstimulation syndrome (OHSS): a review. Human Reproduction Update 2002;8(6):559‐77. [DOI] [PubMed] [Google Scholar]
Ferraretti 2012
- Ferraretti AP, Goossens V, Mouzon J, Bhattacharya S, Castilla JA, Korsak V, et al. The European IVF‐monitoring (EIM), and Consortium, for the European Society of Human Reproduction and Embryology (ESHRE). Human Reproduction 2012;27(9):2571‐84. [DOI] [PubMed] [Google Scholar]
Haning 1982
- Haning RV Jr, Austin CW, Kuzma DL, Shapiro SS, Zweibel WJ. Ultrasound evaluation of estrogen monitoring for induction of ovulation with menotropins. Fertility and Sterility 1982;37(5):627‐32. [DOI] [PubMed] [Google Scholar]
Hardiman 1990
- Hardiman P, Thomas M, Osgood V, Vlassopoulou V, Ginsburg J. Are estrogen assays essential for monitoring gonadotropin stimulant therapy?. Gynecological Endocrinology 1990;4(4):261‐9. [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins J PT, Green S, editors. Cochrane Handbook of Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org. [Google Scholar]
Howard 1988
- Howard WF, Chihal HJ, Strain CA, Smith M. Programmed follicular stimulation reduces cycle cost and stress with no compromise in cycle quality: use of a modified programmed protocol. Journal of In Vitro Fertilisation and Embryo Transfer 1988;5(6):343‐6. [DOI] [PubMed] [Google Scholar]
Jenkins 2006
- Jenkins JM, Drakeley AJ, Mathur RS. The management of ovarian hyperstimulation syndrome (RCOG Green‐top Guideline No. 5). London: Royal College of Obstetricians and Gynaecologists, 2006. [Google Scholar]
Kemeter 1989
- Kemeter P, Feichtinger W. Experience with a new fixed‐stimulation protocol without hormone determinations for programmed oocyte retrieval for in‐vitro fertilization. Human Reproduction 1989;4(8 Suppl):53‐8. [DOI] [PubMed] [Google Scholar]
MacDougall 1992
- MacDougall MJ, Tan SL, Jacobs HS. In‐vitro fertilization and the ovarian hyperstimulation syndrome. Human Reproduction 1992;7(5):597‐600. [DOI] [PubMed] [Google Scholar]
Martins 2013
- Martins WP, Vieira CV, Teixeira DM, Barbosa MA, Dassunção LA, Nastri CO. Ultrasound for monitoring controlled ovarian stimulation: a systematic review and meta‐analysis of randomized controlled trials. Ultrasound Obstetrics and Gynecology 2014;43(1):25‐33. [DOI] [PubMed] [Google Scholar]
Murad 1998
- Murad NM. Ultrasound or ultrasound and hormonal determinations for in vitro fertilization monitoring. International Journal of Gynaecology and Obstetrics 1998;63(3):271‐6. [DOI] [PubMed] [Google Scholar]
Nygren 2002
- Nygren KG, Andersen AN. Assisted reproductive technology in Europe, 1999. Results generated from European registers by ESHRE. Human Reproduction 2002;17(12):3260‐74. [DOI] [PubMed] [Google Scholar]
Rainhorn 1987
- Rainhorn JD, Forman RG, Belaisch‐Allart J, Hazout A, Fries N, Testart J, et al. One year's experience with programmed oocyte retrieval for IVF. Human Reproduction 1987;2(6):491‐4. [DOI] [PubMed] [Google Scholar]
Rizk 1992
- Rizk B, Smitz J. Ovarian hyperstimulation syndrome after superovulation using GnRH agonists for IVF and related procedures. Human Reproduction 1992;7(3):320‐7. [DOI] [PubMed] [Google Scholar]
Roest 1995
- Roest J, Verhoeff A, Heusden AM, Zeilmaker GH. Minimal monitoring of ovarian hyperstimulation: a useful simplification of the clinical phase of in vitro fertilization treatment. Fertility and Sterility 1995;64(3):552‐6. [DOI] [PubMed] [Google Scholar]
Roest 1996
- Roest J, Mous HV, Zeilmaker GH, Verhoeff A. The incidence of major clinical complications in a Dutch transport IVF programme. Human Reproduction Update 1996;2(4):345‐53. [DOI] [PubMed] [Google Scholar]
Schenker 1978
- Schenker JG, Weinstein D. Ovarian hyperstimulation syndrome: a current survey. Fertility and Sterility 1978;30(3):255‐68. [DOI] [PubMed] [Google Scholar]
Shoham 1991
- Shoham Z, Carlo C, Patel A, Conway GS, Jacobs HS. Is it possible to run a successful ovulation induction program based solely on ultrasound monitoring? The importance of endometrial measurements. Fertility and Sterility 1991;56(5):836‐41. [DOI] [PubMed] [Google Scholar]
Smitz 1990
- Smitz J, Camus M, Devroey P, Erard P, Wisanto A, Steirteghem AC. Incidence of severe ovarian hyperstimulation syndrome after GnRH agonist/HMG superovulation for in‐vitro fertilization. Human Reproduction 1990;5(8):933‐7. [DOI] [PubMed] [Google Scholar]
Tan 1992
- Tan SL, Balen A, Hussein E, Mills C, Campbell S, Yovich J, et al. A prospective randomized study of the optimum timing of human chorionic gonadotropin administration after pituitary desensitization in in vitro fertilization. Fertility and Sterility 1992;57(6):1259‐64. [DOI] [PubMed] [Google Scholar]
Tan 1994
- Tan SL. Simplifying in‐vitro fertilization therapy. Current Opinion in Obstetrics and Gynecology 1994;6(2):111‐4. [PubMed] [Google Scholar]
Thomas 2002
- Thomas K, Searle T, Quinn A, Wood S, Lewis‐Jones I, Kingsland C. The value of routine estradiol monitoring in assisted conception cycle. Acta Obstetricia et Gynecologica Scandinavica 2002;81(6):551‐4. [DOI] [PubMed] [Google Scholar]
Vandekerckhove 2014
- Vandekerckhove F, Gerris J, Vansteelandt S, Sutter P. Adding serum estradiol measurements to ultrasound monitoring does not change the yield of mature oocytes in IVF/ICSI. Gynecological Endocrinology 2014;Epub ahead of print (doi:10.3109/09513590.2014.912267):http://www.ncbi.nlm.nih.gov/pubmed/24811095#. [DOI] [PubMed] [Google Scholar]
Vlaisavljevic 1992
- Vlaisavljevic V, Kovacic B, Gavric V. In vitro fertilization program based on programmed cycles monitored by ultrasound only. Journal of the International Federation of Gynaecology and Obstetrics 1992;39(3):227‐31. [DOI] [PubMed] [Google Scholar]
Zimon 2013
- Zimon A, Lannon B, Sheller S, Sakkas D, Ulrich M, Alper M. Venopuncture‐free IVF: Measurement of estrogen in controlled ovarian stimulation IVF cycles using a "patient‐friendly" saliva‐based estradiol assay. Fertility and Sterility 2013;100(3 Suppl 1):S110. [Google Scholar]


