Abstract
Extracts of the cherry blossom plant have been reported to exert various biological effects on human cells. However, no previous investigations have examined the antioxidant and anti-apoptotic effects of these extracts on ultraviolet B (UVB) radiation-induced skin aging. This study explores the underlying mechanisms of the antioxidant and anti-apoptotic effects of cherry blossom extract (CBE) in human keratinocyte (HaCaT) cells. HaCaT cells were treated with CBE at concentrations of 0.5, 1.0, and 2.0% for 24 h and then irradiated with UVB (40 mJ/cm2). CBE effectively and dose-dependently decreased the levels of reactive oxygen species and malondialdehyde, while increasing the activities of superoxide dismutase and glutathione peroxidase. Pretreatment with 1 and 2% CBE attenuated UVB-induced DNA damage by reducing the formation of cyclobutane pyrimidine dimers and 8-hydroxy-20-deoxyguanosine. Furthermore, CBE also prevented UVB-induced apoptosis and significantly downregulated B cell lymphoma 2 (Bcl-2)-associated X, cytochrome-c, and caspase-3 expression, while upregulating Bcl-2 expression. Taken together, these results indicate that CBE protects HaCaT cells from UVB-induced oxidative stress and apoptosis and suggest that CBE could be a potent antioxidant against skin aging.
Electronic supplementary material
The online version of this article (10.1007/s10616-018-0215-7) contains supplementary material, which is available to authorized users.
Keywords: Skin aging, Cherry blossom extract, Antioxidant, Anti-apoptosis, HaCaT cells
Introduction
The skin is the largest and the most important protective organ in the human body, providing a dynamic barrier that prevents water evaporation and protects against noxious substances and pathogens (Swann 2010; Grice and Segre 2011). However, its protective functions can be compromised by the aging process, which leads to frequent skin inflammation, slow repair of wounds, and an increased risk of skin cancers (Maru et al. 2014). Skin aging is a complex and multi-factorial process, which is induced by intrinsic factors, such as gene mutations, cellular metabolism, and hormones, as well as extrinsic factors, such as ultraviolet (UV) irradiation, chemicals, and pollutants (Makrantonaki and Zouboulis 2007; Kammeyer and Luiten 2015). Skin aging results in defects in the structural integrity of the skin and a loss of its physiological functions (Landau 2007). UV irradiation is the primary extrinsic factor responsible for skin aging. UV radiation stimulates excessive reactive oxygen species (ROS) generation, which overwhelms cellular antioxidant defense systems and leads to oxidative stress in the skin (Hewage et al. 2015; Ding et al. 2015). These defense systems include protective enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px), as well as non-enzymatic mechanisms (Piao et al. 2012). Meanwhile, an excessive accumulation of ROS can cause oxidative damage to cellular macromolecules, including DNA modification, lipid peroxidation, and apoptotic signal transduction, which alters cellular function (Fernando et al. 2016; Tu and Tawata 2015; Briganti and Picardo 2003). These damages are usually accompanied by an increase in keratinocyte apoptosis and a decrease in epidermal cell proliferation, which may accelerate skin aging and increase the risk of some skin disorders, including skin cancer (Park et al. 2016).
Numerous studies have indicated that active antioxidant supplements can maintain skin structure and function, as well as delaying skin aging by protecting skin cells from oxidative damage (Baumann 2005). Therefore, various active antioxidant compounds are added to cosmetic products for cytoprotection against UV-induced oxidation to prevent skin aging (Baxter 2008; Popoola et al. 2015). Both synthetic and natural antioxidants are considered effective or potential additives for anti-aging cosmetics. Compared to synthetic antioxidants, natural antioxidants are more widely available, less likely to cause adverse reactions, and more cost-effective (Ding et al. 2015). Thus, studies identifying natural antioxidant products, such as wild plant sources, are necessary to replace the synthetic antioxidants currently used to prevent the skin aging effects of UV exposure (Tu and Tawata 2015; Chang et al. 2016; Jayatissa et al. 2016).
Prunus yedoensis belongs to the Rosaceae family, and is widely cultivated in Japan and Korea. Previous studies have revealed that cherry blossom (P. yedoensis flower) extract (CBE) exerts various physiological effects, including antioxidant, anti-inflammatory, and anti-cancer effects (Zhang et al. 2013, 2014; Lee et al. 2007). Researchers have demonstrated that CBE may be a promising agent against oxidative injury, owing to the synergistic effects of various phenolic compounds (Zhang et al. 2013), such as kaempferol 3-O-β-arabinofuranoside, kaempferol 3-O-β-xylopyranoside, kaempferol 3-O-β-glucopyranoside, and kaempferol 3-O-β-xylopyranosyl-(1 → 2)-β-glucopyranoside (Jung et al. 2002). The major components of CBE, according to a high-performance liquid chromatography (HPLC) assay conducted before this study, are caffeoyl glucoside, prunasinamide, and betulalbuside A (Fig. S1, Supplementary Materials). Similar to Jung et al. (2002), we found that caffeoyl glucoside (a phenolic compound) and prunasinamide can reduce the ROS generation of H2O2-induced normal human dermal fibroblast cells (data not shown). However, the antioxidant and anti-apoptotic mechanisms of CBE in the skin have not been reported. Therefore, this study aims to investigate the protective effects of CBE against UVB-induced oxidative damage in human keratinocyte (HaCaT) cells by assessing its effects on ROS generation and antioxidant-defense systems, including the activities of SOD and GSH-Px, and the production of malondialdehyde (MDA), as well as on DNA damage and apoptosis. To further explore the anti-apoptotic effects of CBE in UVB-irradiated HaCaT cells, we also measured the relative levels of mRNA and protein expression in the apoptotic pathway, including those of B-cell lymphoma 2 (Bcl-2), Bcl-2-associated X (Bax), cytochrome-c, and caspase-3.
Materials and methods
Materials and reagents
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), hydrogen peroxide, 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA), and enhanced chemiluminescence (ECL) reagents were purchased from Beyotime Biotechnology Ltd. (Shanghai, China). Dulbecco’s modified Eagle’s medium (DMEM) high glucose, fetal bovine serum (FBS), penicillin, and streptomycin were purchased from Gibco Life Technologies (Carlsbad, CA, USA). A bicinchoninic acid (BCA) assay kit was purchased from Dahui Biotechnology Ltd. (Guangzhou, China). SOD and GSH-Px activities and MDA levels were determined using relevant commercial kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Cyclobutane pyrimidine dimers (CPD) and 8-hydroxy-20-deoxyguanosine (8-OHdG) were determined using relevant ELISA kits (Cosmobio Co., Ltd, Tokyo, Japan). Trizol reagent was obtained from Invitrogen Co. (Carlsbad, CA, USA). The secondary antibodies, HRP-linked goat anti-mouse and anti-rabbit antibodies, were purchased from Santa Cruz Biotechnology Inc. (CA, USA). Radioimmunoprecipitation assay (RIPA) protein lysate and primary antibodies against β-actin, Bcl-2, Bax, cytochrome-c, and caspase-3 were obtained from Santa Cruz Biotechnology Inc. (CA, USA). A fluorescein isothiocyanate labeled annexin V (annexin V-FITC)/propidium iodide (PI) apoptosis detection kit was purchased from Phygene Ltd. (Fujian, China).
Sample preparation
Cherry blossoms (P. yedoensis flowers) were collected from Jeju Island, South Korea. The dried blossoms were extracted with a 1:20 extraction ratio using 40% 1, 3-propanediol (DuPont Tate & Lyle Bio Products, Tennessee, USA) and 60% distilled water by ultrasonic extraction for three days. The extract was then filtered, and the filtrate was taken to be at a concentration of 100%.
Cell culture
The HaCaT cell line was purchased from Guangzhou Jenniobio Biotechnology Co., Ltd. (Guangzhou, China). Cells were maintained in DMEM supplemented with 10% FBS, 1% penicillin, and 1% streptomycin in a humidified atmosphere with 95% air and 5% CO2 at 37 °C.
UVB irradiation
HaCaT cells were seeded on culture dishes, washed with phosphate-buffered saline (PBS) once after incubation, and then irradiated with 2 mW/cm2 UVB for 200 s, that is 40 mJ/cm2 UVB (Hou et al. 2015). The UVB source was a SH7B UV lamp (Sigmamed, Shanghai, China). After UVB irradiation, cells were incubated in DMEM for 24 h. In parallel, control cells were treated to the same conditions without UVB irradiation.
Cell viability assay
Cell viability was examined using the MTT assay. Briefly, cells were seeded in 96-well plates at a density of 4 × 104 cells per well and treated with 0.5, 1, or 2% CBE for 24 h before UVB exposure. After irradiation, the cells were incubated with 100 μL of 5 mg/mL MTT reagent for an additional 4 h. Subsequently, the absorbance of each sample was measured at 570 nm using a microplate reader (MK3, Thermo Fisher Scientific, MA, USA).
Intracellular ROS measurement
Intracellular ROS levels were evaluated using a DCFH-DA assay (Rosenkranz et al. 1992). Briefly, cells were seeded in 96-well culture plates at a density of 5 × 104 cells per well and treated with CBE at concentrations of 0.5, 1, or 2% for 24 h. Cells were then irradiated with 40 mJ/cm2 UVB. Subsequently, cells were incubated with 50 μL of 10 μmol/L DCFH-DA for 20 min and then washed three times with DMEM. Fluorescence intensity was then detected at 488 nm excitation/525 nm emission and quantified using a fluorescence microplate reader (Synergy HT, BioTek, VT, USA).
Determination of the activities of SOD and GSH-Px and the content of MDA
Cells were seeded in 96-well plates at a density of 1 × 105 cells per well with or without pre-treatment with 0.5, 1, or 2% CBE for 24 h, followed by UVB (40 mJ/cm2) irradiation. The activities of SOD and GSH-Px and the content of MDA were determined using relevant commercial kits, in accordance with the manufacturer’s instructions.
Quantification of the amount of CPD and 8-OHdG
The amounts of CPD and 8-OHdG were determined by enzyme-linked immunosorbent (ELISA) (Mori et al. 1991; Wei et al. 2010). Cells were pre-treated with CBE at concentrations of 0.5, 1.0, or 2.0% for 24 h incubation and then exposed to UVB (40 mJ/cm2). The amounts of CPD and 8-OHdG were determined using relevant ELISA kits in accordance with the manufacturer’s instructions.
Flow cytometric analysis
An Annexin V-FITC apoptosis detection kit was used to determine the number of apoptotic cells. Cells were pre-treated with CBE at concentrations of 0.5, 1.0, or 2.0% for 24 h and then irradiated with UVB (40 mJ/cm2). After exposure, the cells were collected and centrifuged at 800 rpm for 3–4 min, washed three times with cold PBS and resuspended in 100 μL binding buffer solution. Finally, the cells were incubated with Annexin V-FITC (5 μL) and PI (5 μL) at room temperature for 15 min in the dark. The fluorescence of the cells was immediately assessed using flow cytometry (FACSCalibur, Beckton Dickinson, CA, USA). In FACS diagram, the early apoptotic cells and the late apoptotic cells are respectively represented in the lower right quadrant and upper right quadrant. Total apoptosis rates were calculated using the following formula: total apoptosis rate (%) = early apoptosis rate + late apoptosis rate.
Quantitative real-time PCR detection
Cells were pre-treated with or without CBE for 24 h and then irradiated with UVB (40 mJ/cm2). Total RNA was then extracted using Trizol reagent. The RNA was reverse transcribed into cDNA using oligo-(dT) primers and Bcl-2, Bax, cytochrome-c, and caspase-3 gene sequences were amplified. A polymerase chain reaction (PCR) was performed using 5 μL cDNA in a total reaction volume of 50 μL. The PCR conditions were as follows: initial denaturation at 93 °C for 2 min, followed by 40 cycles of 93 °C for 1 min, 55 °C for 1 min, and 72 °C for 1 min. PCR products were analyzed and quantified using an AFD 4800 Real-Time PCR System (Roche, Basel, Switzerland). Each targeted mRNA expression level was normalized to the mRNA expression level of housekeeping gene, β-actin.
Western blot analysis
Cells were pre-treated with or without CBE for 24 h and then irradiated with UVB (40 mJ/cm2). The treated cells were washed twice with cold PBS and homogenized in 1 mL RIPA protein lysate at 12,000 rpm for 30 min at 4 °C. The protein concentration was measured using a BCA protein assay kit. Aliquots of the supernatant containing equal amounts of protein were electrophoresed on 10% sodium dodecyl sulfate–polyacrylamide gel and then transferred onto nitrocellulose membranes (Amersham Pharmacia Biotech., England, UK). The membranes were washed three times with Tris-buffered saline (TBS) and then incubated in TBS with Tween-20 (TBST) and 5% skim milk for 1 h at room temperature. Subsequently, the membranes were transferred into TBST and incubated for 2 h at room temperature with appropriate concentrations of primary antibodies directed against β-actin, Bcl-2, Bax, cytochrome-c, or caspase-3. The probed membranes were washed with TBST for 10 min three times and then incubated with secondary antibodies for 2 h at room temperature. Western blotting was repeated three times independently with the same conditions for the assessment of intra-experimental variability. Immunobands were detected with ECL reagent and exposed to x-ray film. Band density was quantified using a Tanon T600 image analysis system (Shanghai Tanon Technology Ltd., Shanghai, China), and the the relative amounts of proteins associated with specific antibodies were normalized according to the intensities of β-actin.
Statistical analysis
The results are expressed as the mean ± SD (n = 3) and the analyses were conducted using SPSS software (version 22.0; SPSS Inc., Chicago, IL, U.S.A.). Statistical comparisons were analyzed using two-tailed Student’s t tests. P < 0.05 was considered to be statistically significant.
Results
Effects of CBE on UVB-induced cytotoxicity in HaCaT cells
To determinate the protective effects of CBE on HaCaT viability under UVB radiation (40 mJ/cm2), an MTT assay was performed. As shown in Fig. 1, treatment of HaCaT cells with UVB significantly reduced cell viability to 53.67% compared with the control cells. However, the viability of the HaCaT cells which were treated with CBE at concentrations of 0.5, 1.0, and 2.0% before UVB irradiation, significantly increased to 65.61, 69.42, and 80.64%, respectively.
Fig. 1.
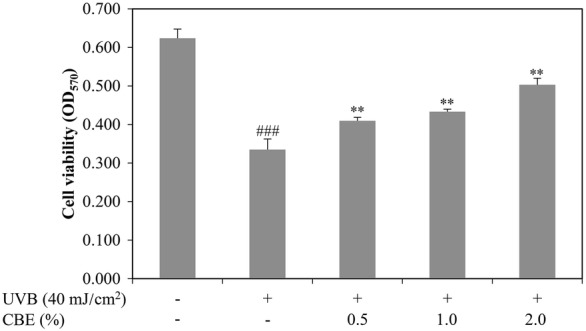
Effects of CBE on UVB-induced HaCaT cell viability. Cells were treated with different concentrations of CBE (0.5, 1.0, and 2.0%) for 24 h and then irradiated with UVB (40 mJ/cm2). Cell viability was measured by MTT assay. Values are shown as the mean ± SD (n = 3). ###P < 0.001 versus the control; *P < 0.05, **P < 0.01, and ***P < 0.001 versus the UVB-alone group. Control: 100%, UVB: 53.67%, UVB + CBE (0.5%): 65.61%, UVB + CBE (1.0%): 69.42%, UVB + CBE (2.0%): 80.64%
Effects of CBE on ROS generation in UVB-irradiated HaCaT cells
To assess the effects of CBE on the inhibition of ROS generation, we examined intercellular ROS levels by DCFH-DA fluorescence intensity. As shown in Fig. 2, a 2.04-fold increase in ROS levels was observed after treating cells with UVB radiation, while treatment with 0.5, 1.0, and 2.0% CBE markedly inhibited UVB-induced ROS generation.
Fig. 2.
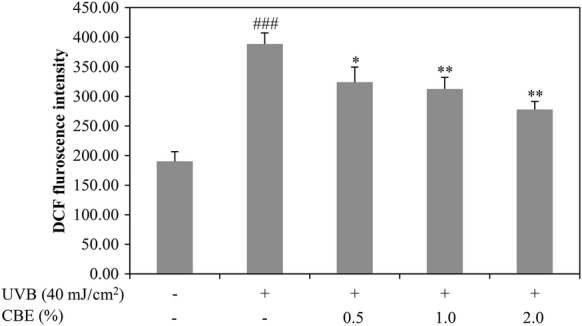
Effects of CBE on ROS generation in UVB-induced HaCaT cells. Cells were treated with different concentrations of CBE (0.5, 1.0, and 2.0%) for 24 h and then irradiated with UVB (40 mJ/cm2). ROS generation was determined by the fluorescence intensity of DCFH-DA. Values are mean ± SD (n = 3). ###P < 0.001 versus the control; *P < 0.05, **P < 0.01, and ***P < 0.001 versus the UVB-alone group. AU: Arbitrary units
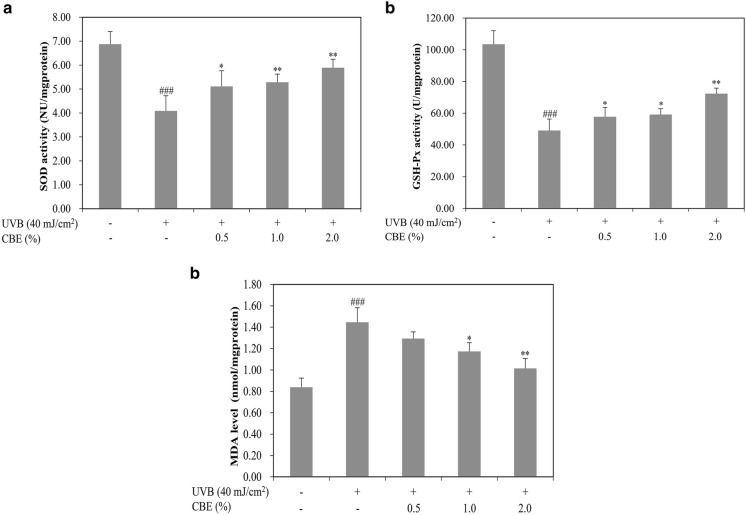
Effects of CBE on SOD and GSH-Px activities and MDA content in UVB-induced HaCaT cells
To explore the protective effects of CBE on antioxidant-defense systems, we investigated the activities of SOD and GSH-Px and the content of MDA using relevant kits. As shown in Fig. 3, HaCaT cells irradiated with UVB alone showed significantly decreased activities of SOD and GSH-Px, as well as increased MDA content, compared to the controls. After treatment with 0.5, 1.0, and 2.0% CBE, the SOD activity in the UVB-induced HaCaT cells showed dose-dependent and statistically significant increases compared with that in the UVB-treated group (Fig. 3a). Similarly, GSH-Px activity in UVB-induced HaCaT cells treated with CBE also increased in a dose-dependent manner (Fig. 3b). Additionally, the MDA content in UVB-treated HaCaT cells treated with 1.0 and 2.0% CBE was significantly lower than that in UVB-treated group (Fig. 3c).
Fig. 3.
Effects of CBE on SOD and GSH-Px activities and MDA content in UVB-induced HaCaT cells. Cells were treated with different concentrations of CBE (0.5, 1.0, and 2.0%) for 24 h and then irradiated with UVB (40 mJ/cm2). SOD activity (a), GSH-Px activity (b) and MDA content (c) were measured using relevant kits. Values are shown as the mean ± SD (n = 3). ###P < 0.001 versus the control; *P < 0.05, and **P < 0.01 versus the UVB-alone group
Effects of CBE on DNA damage in UVB-induced HaCaT cells
To investigate the effects of CBE on DNA damage, we measured the amount of CPD and 8-OHdG in UVB-exposed HaCaT cells using ELISA. As presented in Fig. 4, exposure to UVB significantly increased CPD levels compared with those in the controls. After treatment with 1 and 2% CBE, CPD levels in the UVB-exposed HaCaT cells showed statistically significant decreases in a dose-dependent manner. Similarly, 8-OHdG levels were significantly increased in the UVB-induced cells but treatment with 1 and 2% CBE significantly attenuated this increase.
Fig. 4.
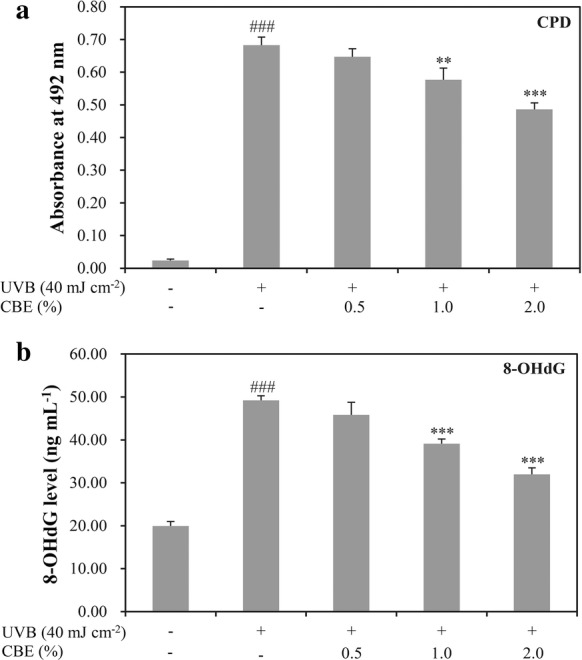
Effects of CBE on UVB-induced CPD and 8-OHdG formation in HaCaT cells. Cells were treated with different concentrations of CBE (0.5, 1.0, and 2.0%) for 24 h and then irradiated with UVB (40 mJ/cm2). The amount of CPD (a) and 8-OHdG (b) was measured by ELISA. Values are shown as mean ± SD (n = 3). ###P < 0.001 versus the control; *P < 0.05, **P < 0.01, and ***P < 0.001 versus the UVB-alone group
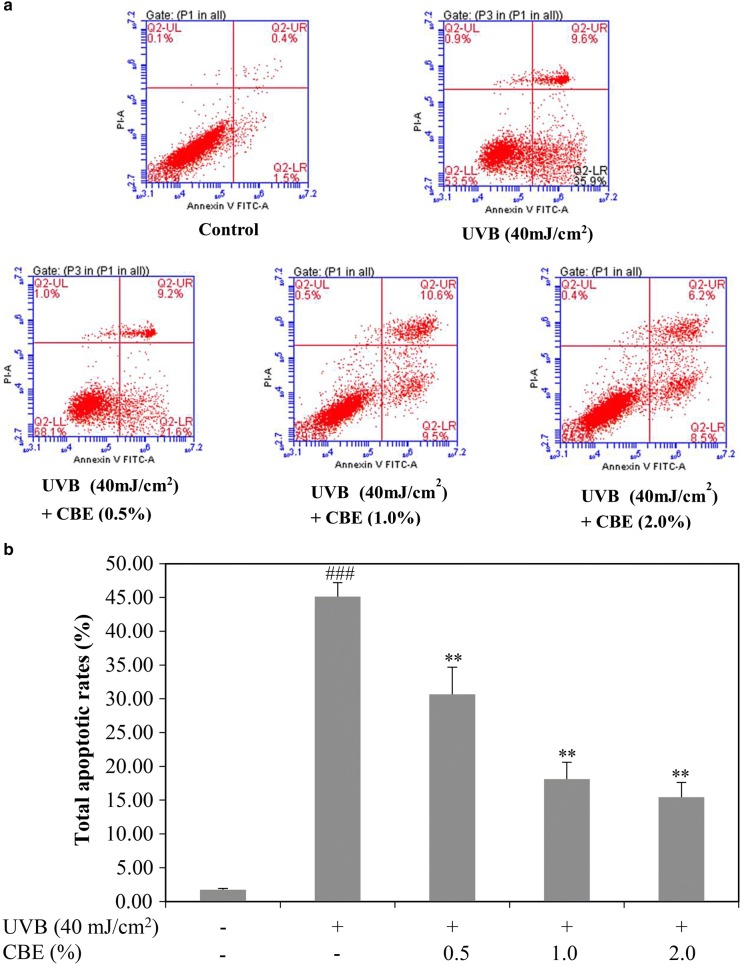
Effects of CBE on UVB-induced cell apoptosis
To investigate the effects of CBE on apoptosis, we measured the apoptosis of HaCaT cells by flow cytometry coupled with Annexin V-FITC and PI double staining. As shown in Fig. 5, exposure to UVB significantly increased HaCaT apoptosis rates from 1.7 ± 0.2 to 45.1 ± 2.1%. Treatment with 0.5, 1.0, and 2.0% CBE significantly reduced the apoptotic rates to 30.7 ± 4.0, 18.1 ± 2.5, and 15.4 ± 2.2%, respectively.
Fig. 5.
Effects of CBE on UVB-induced cell apoptosis in HaCaT cells. Cells were treated with different concentrations of CBE (0.5, 1.0, and 2.0%) for 24 h and then irradiated with UVB (40 mJ/cm2). After 24 h, the apoptotic cells were stained with Annexin V-FITC and PI and analyzed by flow cytometry (a). The histogram illustrates the apoptosis rate of the HaCaT cells (b). Values are shown as mean ± SD (n = 3). ###P < 0.001 versus the control; *P < 0.05 and **P < 0.01 versus the UVB-alone group
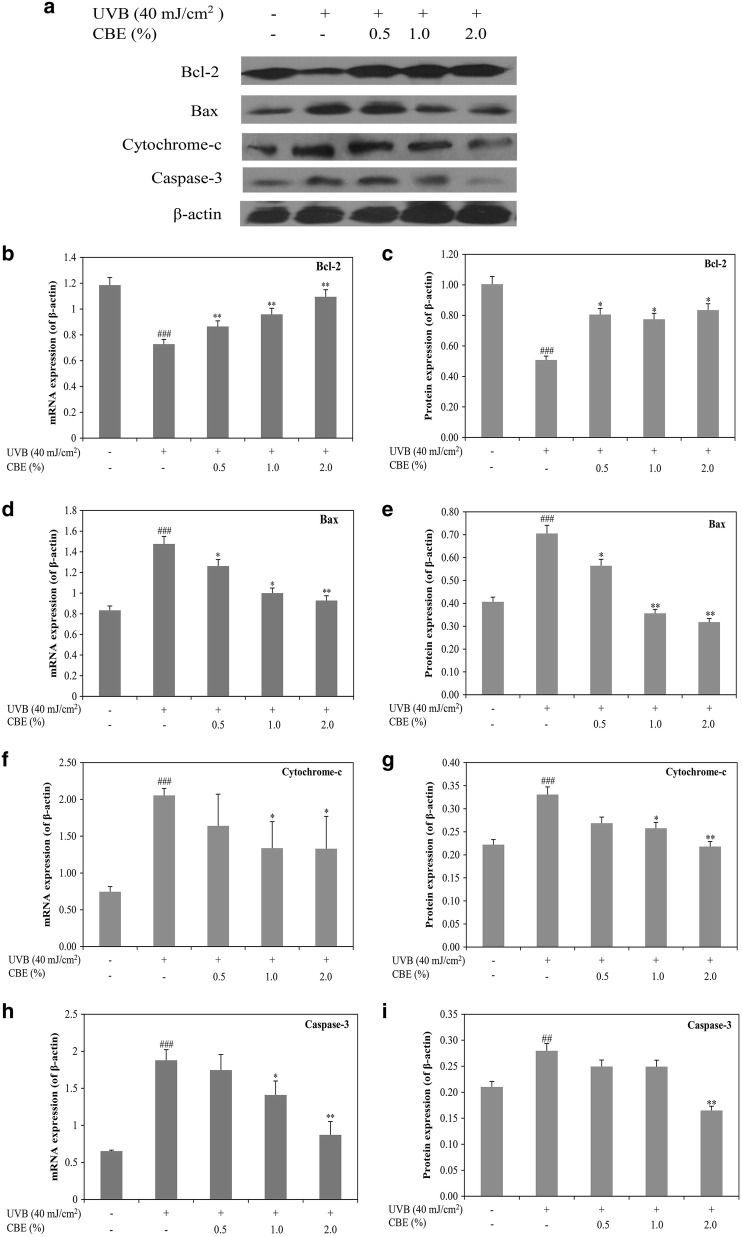
Effects of CBE on the expression of Bcl-2 family proteins, cytochrome-c, and caspase-3 in UVB-induced HaCaT cells
To investigate the molecular basis for the inhibition of UVB-induced apoptosis by CBE in HaCaT cells, we examined the mRNA and protein expressions of the apoptosis pathway genes Bcl-2, Bax, cytochrome-c, and caspase-3 using real-time PCR and western blot, respectively. As shown in Fig. 6b, c, UVB irradiation significantly decreased the mRNA and protein expression of Bcl-2, and increased the mRNA and protein expression of Bax (Fig. 6d, e). In contrast, treatment with 0.5, 1.0, and 2.0% CBE significantly increased the mRNA and protein expression of Bcl-2 and decreased the mRNA and protein expression of Bax in a dose-dependent manner. Additionally, UVB irradiation significantly increased the mRNA and protein levels of cytochrome-c. Treatment with CBE at concentrations of 1 and 2% before UVB exposure effectively attenuated this increase (Fig. 6f, g). Similarly, the mRNA and protein expression of caspase-3 were significantly increased in the UVB-induced cells and this increase was markedly attenuated by treatment with CBE at a concentration of 2.0% (Fig. 6h, i).
Fig. 6.
Effects of CBE on mRNA and protein expression of apoptotic regulators in UVB-induced HaCaT cells. Cells were treated with different concentrations of CBE (0.5, 1.0, and 2.0%) for 24 h and then irradiated with UVB (40 mJ/cm2). Cell lysates were subjected to electrophoresis and western blot analysis (a). The mRNA and protein expression of Bcl-2 (b, c) and Bax (d, e), cytochrome-c (f, g) and caspase-3 (h, i) were measured. Values are shown as the mean ± SD (n = 3). ###P < 0.001 and ##P < 0.001 versus the control; * P < 0.05 and ** P < 0.01 versus the UVB-alone group
Discussion
UV radiation is known as the primary extrinsic factor which causes skin aging (also referred to as photoaging) because of its ability to induce oxidative stress and DNA damage (Choi et al. 2015). Although the skin is equipped with a complex enzymatic and non-enzymatic defense system to maintain its intracellular redox balance, prolonged UV exposure can disrupt this system (Hewage et al. 2015). Particularly, UVB can damage the epidermis and result in extremely rapid photoaging (Ichihashi et al. 2009). The molecular mechanisms underlying UVB-induced apoptosis include ROS generation, DNA strand damage, and the accumulation of death receptors on the cell surface (Fernando et al. 2016). Recent observations have demonstrated that overaccumulation of intracellular ROS accelerates the skin aging process (Baumann. 2005). Therefore, it is important to explore novel and effective antioxidant agents that can protect against premature skin aging. This study aimed to investigate the effects of CBE on both the antioxidant system and the apoptotic pathway, thereby evaluating its ability to protect human epidermal keratinocytes from UVB-induced skin damage and aging.
CBE has been shown to have various pharmacological activities, such as antioxidant, anti-inflammatory, and anti-cancer activities (Zhang et al. 2013, 2014; Lee et al. 2007). These studies used different cell types, such as RAW 264.7 macrophage cells, and show that CBE is not cytotoxic at concentrations of 0.5, 1.0, and 2.0%. However, no previous studies have reported the effects of CBE on HaCaT cells. The present work evaluated the protective effect of CBE at concentrations of 0.5, 1.0, and 2.0% in UVB-irradiated HaCaT cells. Our results show that CBE treatment effectively prevented the loss of cell viability induced by UVB irradiation.
Our previous study reported that CBE markedly inhibited ROS levels in RAW264.7 macrophage cells (Zhang et al. 2014). However, the antioxidant mechanisms of CBE against UVB-induced oxidative stress in keratinocytes are currently unclear. To systemically evaluate the antioxidant effects of CBE, we measured reactive oxygen species (ROS) generation and the capacity of the cell’s antioxidant-defense system. UVB is well known to play a crucial role in the intracellular generation of ROS, which causes premature skin aging (Rittié and Fisher 2002). In this study, the suppression of ROS generation indicated that certain concentrations of CBE can decrease skin damage and protect keratinocytes from the oxidative stress induced by UVB irradiation. Similarly, other natural extracts, such as those from Nymphaea tetragona Georgi and Schisandra chinensis (Turcz.) Baill (from which deoxyschisandrin was isolated), have been shown to possess ROS scavenging antioxidant properties (Park et al. 2016; Hou et al. 2015). When ROS generation and antioxidant capacity are in relative balance, the expression of specific nuclear genes can be modulated to maintain the energy metabolism necessary for the cell’s survival. Once ROS levels exceed the endogenous antioxidant capacity, oxidative stress occurs within the cells (Lee and Wei 2005). Recent discoveries have indicated that antioxidant agents execute their functions by inhibiting ROS generation and increasing the activities of antioxidant enzymes (Piao et al. 2011). To investigate the effects of CBE on the antioxidant-defense system, we measured the activity of the major enzymatic ROS scavengers, SOD and GSH-Px, and found that they eliminate most of the side-effects caused by elevated ROS levels. SOD, a front-line among antioxidant enzymes, converts the superoxide (O2−) into hydrogen peroxide (H2O2) (Ding et al. 2015). GSH-Px can convert toxic peroxides into nontoxic hydroxyl compounds by permitting a reaction with the thiol-group of glutathione, thereby preventing structural damage to and functional loss of the cell membrane by H2O2 (Kammeyer and Luiten 2015). In addition, an increase in ROS levels has two potential influences: damaging cell components and triggering the activation of specific signaling pathways (Finkel and Holbrook 2000). For example, a high ROS level can stimulate lipid peroxidation in the cells. Hence, we also measured MDA content to confirm the effect of CBE on lipid peroxidation. MDA is thought to be the final product of lipid peroxidation and is poisonous to cells. Thus, its accumulation can directly reflect the status of lipid peroxidation and indirectly reflect the status of cell damage resulting from ROS (Ye et al. 2014; Liu et al. 2013). We found that CBE could effectively enhance the activities of SOD and GSH-Px and decrease the content of MDA. The protective effects against oxidative stress provided by CBE might be attributed to its enhancement of the antioxidant ability of the antioxidant-defense system in human epidermal keratinocytes in parallel with decreasing ROS generation.
UVB irradiation is capable of inducing DNA damage both directly, by forming helix-distorting photoproducts such as CPD, and indirectly, via ROS producing oxidative DNA adducts such as 8-OHdG (Emanuele et al. 2014; Afaq et al. 2007). CPD is the primary apoptosis-inducing lesion in DNA repair and its formation is understood to initiate skin carcinogenesis by inducing gene mutations (Batista et al. 2009). 8-OHdG, as a product of oxidative DNA damage by ROS, is related to mutagenesis, carcinogenesis, and premature aging (Gong et al. 2012). Our results show that UVB irradiation significantly increased the formation of CPD and 8-OHdG, however, pretreatment with 1 and 2% CBE can preventing the UVB-induced DNA damage by enhancing the removal of CPD and 8-OHdG. A study, performed in hairless mouse skin, observed similar results after treating the skin with delphinidin, and suggested that the reduced formation of CPDs and 8-OHdG could be attributed to ROS scavenging by delphinidin (Gong et al. 2012).
Determining the amount of UV-induced DNA damage has been suggested to be a crucial stage in determining the onset of the apoptotic program (Takasawa et al. 2005). Apoptosis is designed to remove DNA-damaged cells, when DNA damage is too severe to be repaired (Batista et al. 2009). Furthermore, ROS generation is considered to be an additional critical mediator in triggering the apoptotic machinery (Ichihashi et al. 2003). Recent studies demonstrated that ROS may cause oxidative damage to the DNA, RNA, proteins, and lipids of affected cells and induce apoptosis through the induction of mitochondrial membrane permeability transition and the release of pro-apoptotic proteins when ROS levels exceed a certain threshold (Park et al. 2016; Ueda et al. 2002; Maraldi et al. 2009). In brief, excessive ROS levels downregulate anti-apoptotic proteins and upregulate pro-apoptotic proteins, resulting in interference with the normal release of cytochrome-c, which activates the caspase family proteins, and finally induces cell death (Ryu et al. 2015). In the present study, UVB irradiation was found to markedly increase the apoptosis rate of HaCaT cells. The alterations in UV-induced apoptosis regulation may have an important impact on the induction of skin photoaging and skin cancer. Previous studies have demonstrated that the inhibition of excessive apoptosis and the accelerated elimination of DNA damage induced by UVB could protect the skin from UVB-induced aging and carcinogenesis (El-Mahdy et al. 2008). Our results showed that CBE could effectively attenuated UVB-induced apoptosis. Taken together, the results suggest that CBE may inhibit UVB-induced oxidative stress as well as DNA damage, thereby protecting the skin from UVB-induced apoptosis.
To further evaluate the effect of CBE on UVB-induced apoptosis signaling in HaCaT cells, we assayed the mRNA and protein expressions of the Bcl-2 family genes (Bcl-2 and Bax), the release of cytochrome-c, and the mRNA and protein expression of caspase-3 with and without CBE treatment. The Bcl-2 family includes anti- and pro-apoptotic proteins that can promote cell survival and elicit cell death, respectively. Bcl-2, one of the anti-apoptotic proteins of the Bcl-2 family, suppresses cell death by preventing mitochondrial membrane depolarization and cytochrome-c release (Romano et al. 2008). In contrast, Bax, a pro-apoptotic protein, induces mitochondrial membrane depolarization and cytochrome-c release (Romano et al. 2008). After cytochrome-c is released into the cytosol, it mediates caspase protein family activation, which then results in apoptosis. Caspase-3 serves as an effector caspase to execute apoptosis (Nys et al. 2012). Our results show that UVB irradiation increased the apoptosis rate of HaCaT cells, downregulated Bcl-2, upregulated Bax and caspase-3, and promoted cytochrome-c release. However, treatment with CBE prevented UVB-treated HaCaT cells from undergoing apoptosis by modulating the expressions of Bcl-2 and Bax, suppressing cytochrome-c release into the cytosol, and blocking the caspase-3 pathway. Similar results were also obtained in a study of the anti-apoptotic properties of water lily extracts (Park et al. 2016).
Conclusion
In summary, we demonstrated for the first time the protective effects of CBE on UVB-induced oxidative stress and apoptosis in human epidermal keratinocytes. CBE effectively alleviated UVB-induced oxidative stress by inhibiting excess ROS and MDA generation and increasing antioxidant enzyme activities in human epidermal keratinocytes. CBE also attenuated UVB-induced DNA damage by inhibiting the formation of CPD and 8-OHdG. Our study furthers the understanding of the protective effects of CBE, which attenuated UVB-induced apoptosis of keratinocytes by regulating the Bcl-2 family proteins, cytochrome-c release, and the caspase-3 pathway. Collectively, our results suggest that CBE might be a potent antioxidant agent for slowing the aging process in skin cells. The anti-aging efficacy of skincare products with added CBE on the human body will need to be evaluated in future studies.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We are grateful to The Garden of Natural Solution Co., Ltd. for supporting this work.
Compliance with ethical standards
Conflict of interests
The authors have declared that no competing interests exist.
Contributor Information
Weixuan Li, Phone: +86-757-83162332, Email: 13902803396@163.com.
Lei Guan, Phone: +86-20-85166421, Email: guanlei@landproof.com.
References
- Afaq F, Syed DN, Malik A, Hadi N, Sarfaraz S, Kweon MH, Khan N, Zaid MA, Mukhtar H. Delphinidin, an anthocyanidin in pigmented fruits and vegetables, protects human hacat keratinocytes and mouse skin against UVB-mediated oxidative stress and apoptosis. J Invest Dermatol. 2007;127:222–232. doi: 10.1038/sj.jid.5700510. [DOI] [PubMed] [Google Scholar]
- Batista LFZ, Kaina B, Meneghini R, Menck CF. How DNA lesions are turned into powerful killing structures: insights from UV-induced apoptosis. Mutat Res. 2009;681:197–208. doi: 10.1016/j.mrrev.2008.09.001. [DOI] [PubMed] [Google Scholar]
- Baumann L. How to prevent photoaging? J Invest Dermatol. 2005;125:xii–xiii. doi: 10.1111/j.0022-202X.2005.23810.x. [DOI] [PubMed] [Google Scholar]
- Baxter RA. Anti-aging properties of resveratrol: review and report of a potent new antioxidant skin care formulation. J Cosmet Dermatol. 2008;7:2–7. doi: 10.1111/j.1473-2165.2008.00354.x. [DOI] [PubMed] [Google Scholar]
- Briganti S, Picardo M. Antioxidant activity, lipid peroxidation and skin diseases. What’s new. J Eur Acad Dermatol Venereol. 2003;17:663–669. doi: 10.1046/j.1468-3083.2003.00751.x. [DOI] [PubMed] [Google Scholar]
- Chang YJ, Lee DU, Nam JH, Kim WK. Inhibitory effect of a grimonia pilosa leaf extract on the UV-Induced photoaging-related ion channel, ORAI1, and the enzymes tyrosinase and elastase. J Food Biochem. 2016;40:2–9. doi: 10.1111/jfbc.12171. [DOI] [Google Scholar]
- Choi HS, Park ED, Park Y, Suh HJ. Spent coffee ground extract suppresses ultraviolet B-induced photoaging in hairless mice. J Photochem Photobiol B. 2015;153:164–172. doi: 10.1016/j.jphotobiol.2015.09.017. [DOI] [PubMed] [Google Scholar]
- Ding Q, Yang D, Zhang W, Lu Y, Zhang M, Wang L, Li X, Zhou L, Wu Q, Pan W, Chen Y. Antioxidant and anti-aging activities of the polysaccharide TLH-3 from Tricholoma lobayense. Int J Biol Macromol. 2015;85:133–140. doi: 10.1016/j.ijbiomac.2015.12.058. [DOI] [PubMed] [Google Scholar]
- El-Mahdy MA, Zhu Q, Wang Q, Gulzar W, Srinivas P, Zhao Q, Arafa ES, Barakat B, Mir SN, Wani AA. Naringenin protects hacat human keratinocytes against UVB-induced apoptosis and enhances the removal of cyclobutane pyrimidine dimers from the genome. Photochem Photobiol. 2008;84:307. doi: 10.1111/j.1751-1097.2007.00255.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emanuele E, Spencer JM, Braun M. From DNA repair to proteome protection: new molecular insights for preventing non-melanoma skin cancers and skin aging. J Drugs Dermatol. 2014;13:274–281. [PubMed] [Google Scholar]
- Fernando PM, Piao MJ, Hewage SR, Kang HK, Yoo ES, Koh YS, Ko MH, Ko CS, Byeon SH, Mun SR, Lee NH, Hyun JW. Photo-protective effect of sargachromenol against UVB radiation-induced damage through modulating cellular antioxidant systems and apoptosis in human keratinocytes. Environ Toxicol Pharamacol. 2016;43:112–119. doi: 10.1016/j.etap.2016.02.012. [DOI] [PubMed] [Google Scholar]
- Finkel T, Holbrook NJ. Oxidants, oxidative stress and the biology of ageing. Nature. 2000;408:239–247. doi: 10.1038/35041687. [DOI] [PubMed] [Google Scholar]
- Gong C, Tao G, Yang L, Liu J, He H, Zhuang Z. The role of reactive oxygen species in silicon dioxide nanoparticle-induced cytotoxicity and DNA damage in hacat cells. Mol Biol Rep. 2012;39:4915–4925. doi: 10.1007/s11033-011-1287-z. [DOI] [PubMed] [Google Scholar]
- Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol. 2011;9:244–253. doi: 10.1038/nrmicro2537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hewage MSR, Piao MJ, Kim KC, Cha JW, Choi YH, Chae SW, Hyun WJ. Galangin (3,5,7-Trihydroxyflavone) shields human keratinocytes from ultraviolet B-induced oxidative stress. Biomol Ther. 2015;23:165–173. doi: 10.4062/biomolther.2014.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hou W, Gao W, Wang D, Liu Q, Zheng S, Wang Y. The protecting effect of deoxyschisandrin and schisandrin B on HaCaT cells against UVB-induced damage. PLoS ONE. 2015;10:1–11. doi: 10.1371/journal.pone.0127177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ichihashi M, Ueda M, Budiyanto A, Bito T, Oka M, Fukunaga M, Tsuru K, Horikawa T. UV-induced skin damage. Toxicology. 2003;189:21–39. doi: 10.1016/S0300-483X(03)00150-1. [DOI] [PubMed] [Google Scholar]
- Ichihashi M, Ando H, Yoshida M, Niki Y, Matsui M. Photoaging of the skin. J Anti Aging Med. 2009;6:46–59. doi: 10.3793/jaam.6.46. [DOI] [Google Scholar]
- Jayatissa FPMD, Jing PM, Kang KA, Seong RY, Wook CS, Won HJ. Rosmarinic acid attenuates cell damage against uvb radiation-induced oxidative stress via enhancing antioxidant effects in human hacat cells. Biomol Ther. 2016;24:75–84. doi: 10.4062/biomolther.2015.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung HA, Kim AR, Chung HY, Choi JS. In vitro antioxidant activity of some selected Prunus species in Korea. Arch Pharm Res. 2002;25:865–872. doi: 10.1007/BF02977006. [DOI] [PubMed] [Google Scholar]
- Kammeyer A, Luiten RM. Oxidation events and skin aging. Ageing Res Rev. 2015;21:16–29. doi: 10.1016/j.arr.2015.01.001. [DOI] [PubMed] [Google Scholar]
- Landau M. Exogenous factors in skin aging. Curr Probl Dermatol. 2007;35:1–13. doi: 10.1159/000106405. [DOI] [PubMed] [Google Scholar]
- Lee HC, Wei YH. Mitochondrial biogenesis and mitochondrial DNA maintenance of mammalian cells under oxidative stress. Int J Biochem Cell Biol. 2005;37:822–834. doi: 10.1016/j.biocel.2004.09.010. [DOI] [PubMed] [Google Scholar]
- Lee BB, Cha MR, Kim SY, Park E, Park HR, Lee SC. Antioxidative and anticancer activity of extracts of cherry (Prunus serrulata var. spontanea) blossoms. Plant Foods Hum Nutr. 2007;62:79–84. doi: 10.1007/s11130-007-0045-9. [DOI] [PubMed] [Google Scholar]
- Liu D, Hu HY, Lin ZX. Quercetin deformable liposome: preparation and efficacy against ultraviolet B induced skin damages in vitro and in vivo. J Photochem Photobiol B. 2013;127C:8–17. doi: 10.1016/j.jphotobiol.2013.07.014. [DOI] [PubMed] [Google Scholar]
- Makrantonaki E, Zouboulis CC. Molecular mechanisms of skin aging: state of the art. Ann N Y Acad Sci. 2007;1119:40–50. doi: 10.1196/annals.1404.027. [DOI] [PubMed] [Google Scholar]
- Maraldi T, Prata CD, Zambonin L, Landi L, Hakim G. Induction of apoptosis in a human leukemic cell line via reactive oxygen species modulation by antioxidants. Free Radical Biol Med. 2009;46:244–252. doi: 10.1016/j.freeradbiomed.2008.10.027. [DOI] [PubMed] [Google Scholar]
- Maru GB, Gandhi K, Ramchandani A, Kumar G. The role of inflammation in skin cancer. Adv Exp Med Biol. 2014;816:437–469. doi: 10.1007/978-3-0348-0837-8_17. [DOI] [PubMed] [Google Scholar]
- Mori T, Nakane M, Hattori T, Matsunaga T, Ihara M, Nikaido O. Simultaneous establishment of monoclonal antibodies specific for either cyclobutane pyrimidine dimer or (6-4) photoproduct from the same mouse immunized with ultraviolet-irradiated DNA. Photochem Photobiol. 1991;54:225–232. doi: 10.1111/j.1751-1097.1991.tb02010.x. [DOI] [PubMed] [Google Scholar]
- Nys K, Maes H, Andrei G, Snoeck R, Garmyn M, Agostinis P. Skin mild hypoxia enhances killing of UVB-damaged keratinocytes through reactive oxygen species-mediated apoptosis requiring Noxa and Bim. Free Radical Biol Med. 2012;52:1111–1120. doi: 10.1016/j.freeradbiomed.2011.12.017. [DOI] [PubMed] [Google Scholar]
- Park G, Sim Y, Lee W, Sung SH, Oh MS. Protection on skin aging mediated by antiapoptosis effects of the water lily (Nymphaea Tetragona Georgi) via reactive oxygen species scavenging in human epidermal keratinocytes. Pharmacology. 2016;97:282–293. doi: 10.1159/000444022. [DOI] [PubMed] [Google Scholar]
- Piao MJ, Yoo ES, Koh YS, Kang HK, Kim J, Kim YJ, Kang HH, Hyun JW. Antioxidant effects of the ethanol extract from flower of Camellia japonica via scavenging of reactive oxygen species and induction of antioxidant enzymes. Int J Mol Sci. 2011;12:2618–2630. doi: 10.3390/ijms12042618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piao MJ, Hyun YJ, Cho SJ, Kang HK, Yoo ES, Koh YS, Lee NH, Ko MH, Hyun JW. An Ethanol extract derived from Bonnemaisonia hamifera scavenges ultraviolet B (UVB) radiation-induced reactive oxygen species and attenuates UVB-Induced cell damage in human keratinocytes. Mar Drugs. 2012;10:2826–2845. doi: 10.3390/md10122826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Popoola OK, Marnewick JL, Rautenbach F, Ameer F, Iwuoha EI, Hussein AA. Inhibition of oxidative stress and skin aging-related enzymes by prenylated chalcones and other flavonoids from Helichrysum teretifolium. Molecules. 2015;20:7143–7155. doi: 10.3390/molecules20047143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rittié L, Fisher GJ. UV-light-induced signal cascades and skin aging. Ageing Res Rev. 2002;1:705–720. doi: 10.1016/S1568-1637(02)00024-7. [DOI] [PubMed] [Google Scholar]
- Romano G, Briguori C, Quintavalle C, Zanca C, Rivera NV. Contrast agents and renal cell apoptosis. Eur Heart J. 2008;29:2569–2576. doi: 10.1093/eurheartj/ehn197. [DOI] [PubMed] [Google Scholar]
- Rosenkranz AR, Schmaldienst S, Stuhlmeier KM, Chen W, Knapp W, Zlabinger GJ. A microplate assay for the detection of oxidative products using 2,7-dichlorofluorescin-diacetate. J Immunol Methods. 1992;156:39–45. doi: 10.1016/0022-1759(92)90008-H. [DOI] [PubMed] [Google Scholar]
- Ryu BM, Ahn BN, Kang KH, Kim YS, Li YX, Kong CS, Kim SK, Kim DG. Dioxinodehydroeckol protects human keratinocyte cells from UVB-induced apoptosis modulated by related genes Bax/Bcl-2 and caspase pathway. J Photochem Photobiol B. 2015;153:352–357. doi: 10.1016/j.jphotobiol.2015.10.018. [DOI] [PubMed] [Google Scholar]
- Swann G. The skin is the body’s largest organ. J Vis Commun Med. 2010;33:148–149. doi: 10.3109/17453054.2010.525439. [DOI] [PubMed] [Google Scholar]
- Takasawa R, Nakamura H, Mori T, Tanuma S. Differential apoptotic pathways in human keratinocyte hacat cells exposed to UVB and UVC. Apoptosis. 2005;10:1121–1130. doi: 10.1007/s10495-005-0901-8. [DOI] [PubMed] [Google Scholar]
- Tu PT, Tawata S. Anti-oxidant, anti-aging, and anti-melanogenic properties of the essential oils from two varieties of Alpinia zerumbet. Molecules. 2015;20:16723–16740. doi: 10.3390/molecules200916723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueda S, Masutani H, Nakamura H, Tanaka T, Ueno M, Yodoi J. Redox control of cell death. Antioxid Redox Sign. 2002;4:405. doi: 10.1089/15230860260196209. [DOI] [PubMed] [Google Scholar]
- Wei G, Yu A, Jiang L, Geng C, Zhong L. The protective effects of hydroxytyrosol against UVB-induced dna damage in hacat cells. Phytother Res. 2010;24:352–359. doi: 10.1002/ptr.2943. [DOI] [PubMed] [Google Scholar]
- Ye Y, Jia RR, Tang L, Chen F. In vivo antioxidant and anti-skin-aging activities of ethyl acetate extraction from Idesia polycarpa defatted fruit residue in aging mice induced by D-galactose evidence-based complementary and alternative. Evid Based Complement Alternat Med. 2014;2014:746. doi: 10.1155/2014/185716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang YQ, Yin SC, Zhang S, Chang MS, Tang JW, Guan L. Anti-oxidative activity and safety of cherry blossom extracts in vitro. J Diagn Ther Derma-Venereol. 2013;20:392–395. [Google Scholar]
- Zhang YQ, Guan L, Zhong ZY, Chang M, Zhang DK, Li H, Lai W. The anti-inflammatory effect of cherry blossom extract (Prunus yedoensis) used in soothing skincare product. Int J Cosmet Sci. 2014;36:527–530. doi: 10.1111/ics.12149. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.