Abstract
Non-serotype-specific protein-based pneumococcal vaccines have received extensive research focus due to the limitations of polysaccharide-based vaccines. Pneumococcal proteins (PnPs), universally expressed among serotypes, may induce broader immune responses, stimulating humoral and cellular immunity, while being easier to manufacture and less expensive. Such an approach has raised issues mainly associated with sequence/level of expression variability, chemical instability, as well as possible undesirable reactogenicity and autoimmune properties. A step forward employs the identification of highly-conserved antigenic regions within PnPs with the potential to retain the benefits of protein antigens. Besides, their low-cost and stable construction facilitates the combination of several antigenic regions or peptides that may impair different stages of pneumococcal disease offering even wider serotype coverage and more efficient protection. This review discusses the up-to-date progress on PnPs that are currently under clinical evaluation and the challenges for their licensure. Focus is given on the progress on the identification of antigenic regions/peptides within PnPs and their evaluation as vaccine candidates, accessing their potential to overcome the issues associated with full-length protein antigens. Particular mention is given of the use of newer delivery system technologies including conjugation to Toll-like receptors (TLRs) and reformulation into nanoparticles to enhance the poor immunogenicity of such antigens.
Keywords: protein-based pneumococcal vaccines, peptide antigens, vaccine adjuvants
1. Introduction
Streptococcus pneumoniae is a major cause of morbidity and mortality worldwide and antibiotic treatment is being compromised by the emergence of multidrug-resistant strains [1,2,3]. To date, 95 distinct capsular serotypes have been identified including the recently reported serotypes 6D, 11E, 20A/B, and 35D [4,5].
Two types of pneumococcal vaccines both targeting capsular polysaccharides are currently licensed: (a) the 23-valent pneumococcal polysaccharide-based vaccine (PPV-23) and (b) the 7-, 10-, and 13-valent pneumococcal conjugate vaccines (PCV-7, -10, -13). PPV-23 is poorly immunogenic in children under two years of age and generates neither an immune memory response nor herd immunity [6]. Conjugation of a pneumococcal polysaccharide to a carrier protein turned polysaccharide-based vaccines from T-cell independent to T-cell dependent antigens, enhancing their immunogenicity. Conjugate vaccines evoked an efficient immune memory and induced relevant herd immunity through a significant effect on nasopharyngeal carriage [7].
All currently available pneumococcal vaccines have limitations. Both PPV and PCVs are serotype-based vaccines and therefore they elicit only serotype-specific immunity. Emergence of replacement serotypes has repeatedly occurred after their introduction, even after the use of expanded valency PCVs [8,9,10,11], not to mention the vaccine-failures reported following the immunization with PCV-13 [12,13]. PCVs are less efficient against non-invasive pneumococcal disease (IPD) in comparison to IPD [14,15,16,17,18]. Their serotype coverage in developed countries is substantially higher compared to the developing world where pneumococcal disease is caused by a wider spectrum of serotypes [19]. Their debatable effect on pneumococcal disease has been well documented among high-risk individuals [20]. PCVs are very complicated and expensive to manufacture and thus unaffordable for those in greater need, especially in developing countries. Notably, PCV-13 is the second most expensive vaccine for the pediatric population among the 31 listed in Centres for Diseases Control and Prevention (CDC) (https://www.cdc.gov/vaccines/programs/vfc/awardees/vaccine-management/price-list/index.html#f5) [21].
An initial attempt to overcome the replacement phenomenon and the great geographical variation in circulating serotypes was the addition of new pneumococcal serotypes in the already licensed PCVs. PCV-15 is such a vaccine, containing the PCV-13 serotypes as well as 22F and 33F. It is currently on phase 3 clinical trial [22,23]. However, such vaccines will most likely retain all the shortcomings of PCVs mentioned above.
Another approach employs the use of next-generation whole-cell (killed) pneumococcal vaccines (WCPVs). So far, several methods including attenuation, chemical treatment, or preparations of whole-cell crude extracts have been used to reduce or inactivate a pathogen’s virulence for the development of vaccines. For example, both whole-cell and acellular purified protein-based pertussis vaccines provide efficient protection; however, whole-cell pertussis vaccines are more efficacious and produce longer lasting immunity than acellular vaccine. Based on such observations, WCPVs may be safe, provide multiple common surface proteins in native configurations and induce an immune response that is protective across different serotypes [24].
A WCPV may be the most suitable strategy for developing countries, as it is inexpensive and easy to construct and administer [25].
One such vaccine containing a non-capsulated pneumococcal strain has shown protection against nasopharyngeal (NP) colonization and sepsis in immunized mice [26]. Protection against NP colonization was shown to be CD4+ Interleukin 17A (IL-17A)-dependent and non-capsular antibody mediated [26,27].
The WCPV was used in two clinical trials to study safety, dose tolerance, and immunogenicity [28]. In healthy US adults (NCT01537185) WCPV was shown to be safe and well tolerated; significant IgG responses to pneumococcal antigens were elicited, including pneumococcal surface protein A (PspA) and pneumolysin, and functional antibody responses were detected in a pneumolysin toxin neutralizing assay [29]. Significant increases in T-cell cytokine responses, including IL-17A, were measured among subjects receiving the highest dose of WCPV. Passive transfer of human immune sera protected mice against fatal sepsis when challenged intravenously with a virulent strain of pneumococci [30]. Further studies are required to evaluate the safety, dose tolerance, and immunogenicity of such vaccines.
2. Protein-Based Pneumococcal Vaccines
2.1. Rationale towards Protein-Based Pneumococcal Vaccines
A promising alternative to capsular polysaccharide-based vaccines is the use of pneumococcal virulence proteins for the construction of novel vaccines. Pneumococcus has several proteins that are well-conserved among different serotypes, most of which are surface-displayed and thus antibody accessible [31]. Similar to capsular polysaccharide antigens, non-capsular protein antigens may induce the production of opsonophagocytic antibodies, with the potential to offer serotype-independent protection against pneumococcal disease [32]. Considering the fact that pneumococcal proteins contribute to bacterial virulence, as they are involved in several different stages of disease pathogenesis, anti-protein antibodies, unlike anti-capsular antibodies, may display additional protective mechanisms other than opsonophagocytosis. For instance, human antibodies against several pneumococcal proteins have been shown to reduce pneumococcal adherence to human lung epithelial cells and murine NP colonization by pneumococcus [33].
Soon, several epidemiological and experimental findings suggested that not only humoral but also cellular immunity generated by pneumococcal proteins is involved in the natural mechanism of acquired immunity to pneumococcal disease [34]. Prior to the PCV introduction, age-related decreases in IPD rates preceded the development of measurable serotype-specific antibodies [35]. IPD incidence peaks at around 10 months of age and then drops by 50% till the age of two years almost equally among all serotypes, well before the development of protective levels of anti-polysaccharide antibodies, arguing in favor of a serotype-independent mechanism [35]. This could be explained by the acquisition of antibodies to non-capsular conserved antigens, following repeated mucosal exposure to pneumococci, albeit such an assumption has not been confirmed so far. Indeed, the increased IPD susceptibility of not only pediatric, but also AIDS adults (i.e., HIV-infected adults), with low CD4+ T-cells implies a protective role of CD4 T-cells independent of antibodies [36,37,38]. Therefore, proteins as vaccine antigens are likely to induce both arms of immunity.
Many pneumococcal proteins have been undergoing preclinical evaluation as possible vaccine candidates [31]; Table 1 summarizes pneumococcal proteins that have been evaluated or are currently under evaluation as components of protein-subunit vaccines in humans. The majority of such formulations combines several proteins in order to expand their coverage and minimize the possibility of immune escape [39,40].
Table 1.
Summary of pneumococcal proteins that have been evaluated or are currently under evaluation as components of protein-subunit vaccines in humans.
| Vaccine | Assignee | Current Status | References |
|---|---|---|---|
| PhtD monovalent | GSK | Phase 2 completed | Leroux-Roels et al., 2014 [41] Seiberling et al., 2012 [42] |
| PhtD monovalent | Sanofi Pasteur | Phase 1 completed | Leroux-Roels et al., 2014 [41] |
| PhtD+dPly | GSK | Phase 2 completed | Seiberling et al., 2012 [42] |
| Protein D+PhtD+dPly trivalent | GSK | Phase 1 completed | Berglund et al., 2014 [43] |
| PhtD+dPly+PCV8 | GSK | Phase 1 completed | Seiberling et al., 2012 [42] |
| PhtD+dPly+PCV10 | GSK | Phase 2 completed | Seiberling et al., 2012 [42] |
| dPly, PhtD+dPly+PCV10 | GSK | Phase 1 completed | Seiberling et al., 2012 [42] |
| PspA | Sanofi Pasteur | Phase 1 completed | Frey et al., 2013 [44] |
| PlyD1 monovalent | Sanofi Pasteur | Phase 1 completed | Bologa et al., 2012 [45] Kamtchoua et al., 2013 [46] |
| PspA+PsaA | Sanofi-Pasteur/CDC | Phase 1 completed | Briles et al., 2000 [47,48] Nabors et al., 2000 [49] |
| PcpA | Sanofi Pasteur | Phase 1 completed | Bologa et al., 2012 [45] |
| PcpA+PhtD | Sanofi Pasteur | Phase 1 completed | Khan et al., 2012 [50] Bologa et al., 2012 [45] |
| PhtD+PcpA+PlyD1 | Sanofi Pasteur | Phase 1 completed | Bologa et al., 2012 [45] |
| PcsB, StkP, PsaA (IC-47) | Intercell AG/Novartis | Phase 1 completed | Kamtchoua et al., 2013 [46] |
2.2. Challenges Concerning the Licensure of Protein-Based Pneumococcal Vaccines
Protein-based pneumococcal vaccines face several challenges that remain to be addressed before licensure. The amino-acid sequence and level of expression vary among different serotypes [51]. The amino-acid sequence among pneumococcal proteins shows much less diversity compared to the biochemical structure among pneumococcal polysaccharides. However, some proteins contain regions with high sequence variability among different serotypes, which make them non-suitable candidates for the development of a serotype-independent vaccine. The level of protein expression among different strains/serotypes displays significant heterogeneity and variability. A recent study of the pneumococcal variome analyzed the distribution of proteins associated with virulence and host–pathogen interactions among 25 pneumococcal strains and revealed that only 26 of the 65 proteins are constitutively expressed without significant sequence variation in all strains [52]. Remarkably, only three of the above 26 widely expressed proteins (namely pneumococcal surface antigen A (PsaA), serine threonine kinase protein (StkP) and pneumolysin), have been so far included as candidate antigens in the vaccines that are currently under evaluation in clinical studies listed in Table 1. Sequence and expression variability could be overcome by combining more than one protein antigens in one molecule; though such an approach may raise difficulties concerning its high-cost and stable production. It is usually the protein’s 3-dimensional (3-D) structure that is immunogenic. The synthesis of recombinant protein antigens is characterized by a high degree of chemical instability that hampers their synthesis for mass production. Recombinant proteins, when overexpressed in a heterologous host, are often misfolded, exposing their hydrophobic regions and forming insoluble aggregates blocking their proper production [53]. Various cryptotopes may be exposed and immunodominant epitopes may be buried due to the denaturation procedure, preventing a more specific immunological response [54]. Pneumococcal proteins (i.e., pneumolycin) may be associated with undesirable reactogenicity or even toxicity if used in humans while the cost required for their detoxification is quite high [55,56]. Using whole proteins as vaccine antigens cannot exclude the possibility of a host autoimmune response caused by certain antigen fragments. For instance, a prior report suggested that some antibodies to the protection-eliciting coiled coil domain of PspA may react with denatured myosin at room temperature [55,56]. Although there is no compelling evidence for the clinical relevance of this observation, further research is required for the development of a PspA-based vaccine containing this domain [56,57].
Another issue worth mentioning is the different effects of polysaccharide-based and protein-based pneumococcal vaccines on NP colonization. NP colonization is an essential precursor to pneumococcal disease pathogenesis. In the NP, the expression of various pneumococcal virulence factors differs between transparent and opaque phase, facilitating the establishment of commensal carriage. Therefore, antibodies against capsular polysaccharides fail to protect against already established NP colonization, while antibodies against proteins may hamper initiation of colonization. Besides, antibodies against capsular polysaccharides prevent carriage of a specific serotype leading to a significant herd immunity effect; however, complete elimination of carriage in the NP and subsequent replacement of vaccine serotypes by non-vaccine serotypes has been an important issue. In contrast, protein-based pneumococcal vaccines, that are serotype independent, have been associated with intermittent priming and boosting to all pneumococcal serotypes through natural exposure [58]. This effect has been suggested to retain pneumococcal carriage density below disease threshold, instead of the total elimination induced by polysaccharide-based vaccines. Thus, such vaccines may induce more robust memory responses, and at the same time, prevent other pathogens emerging in the nasopharynx. A recent study among healthy infants showed that NP mucosal antibody levels to pneumococcal histidine triad D (PhtD), pneumococcal-choline binding protein A (PcpA), and pneumolysin detoxified derivative (PlyD1), induced through repeated colonization, correlated with protection against acute otitis media (AOM) but not protection against NP colonization [59]. Additionally, in a mouse influenzae/pneumococcal co-infection model it was shown that vaccination with PhtD prevented pneumococcal density from reaching a pathogenic threshold during a viral upper tract respiratory infection (URTI) without elimination of the pathogen from the NP [60]. However, things may differ in developing countries, where persistently high antigenic exposure attributed to the high pneumococcal burden might lead to immune hyporesponsiveness, resulting in low vaccine efficacy, since antigen specific CD4 T-cells can lose their ability to help B cells differentiate into antibody secreting cells [61].
AOM is the most suitable type of pneumococcal infection to assess the efficacy of pneumococcal vaccines due to its easy diagnosis using tympanocentesis and lower costs and shorter time to accumulate a sufficient number of subjects compared to IPD with concurrent PCV administration. Mucosal immunity plays a critical role in control of pneumococcus locally invasive infections including AOM. Capsular polysaccharide vaccines are less effective in protection against AOM than against IPD. As mentioned above, NP mucosal antibody levels to PhtD, PcpA, and PlyD1, induced through repeated colonization, correlated with protection against AOM [59]. Similarly, higher levels of mucosal antibodies against 6 pneumococcal protein antigens (PhtD, Pneumococcal histidine triad protein E (PhtE), the endo-β-N-acetylglucosaminidase (LytB), PcpA, PspA, and Ply) induced through natural exposure in healthy children were associated with reduced risk of AOM [33,62,63].
Currently, a clinical efficacy study is underway in an otitis-prone Native American population (NCT01545375). Nevertheless, for a vaccine to be efficient against AOM, special attention should be given to studies in the infection-prone child who has an immune response that is generally deficient compared to non-otitis prone children leading to susceptibility to recurrent AOM.
Therefore, while otitis-prone population may be attractive to assess the efficacy of protein-based pneumococcal vaccines against AOM, it should be realized that the response to such vaccines in otitis-prone populations may not be of the same magnitude as the general population.
3. Distinct Antigenic Fragments within Pneumococcal Proteins as Vaccine Antigens
3.1. Rationale towards the Use of Distinct Protein Fragments as Vaccine Antigens
To overcome the shortcomings of the protein-based pneumococcal vaccines mentioned above, extensive research has been directed towards the identification of highly-conserved antigenic regions, peptides, or even distinct B-cell epitopes within pneumococcal proteins that could serve for the design of novel pneumococcal vaccines. This approach is based on the theory that in order to initiate an efficient immune response against a specific protein, a vaccine principally needs to include only the minimal immunogenic peptide sequence which can be produced synthetically. These epitopes correspond to only a small region of the entire protein (polypeptide) antigen. Vaccines containing distinct antigenic protein fragments are capable of inducing immune responses through the T-cell-dependent pathway requiring both B-cell and T-cell peptide-epitopes specifically recognized by the T-cell and B-cell receptor (TCR and BCR respectively). Currently, several human peptide vaccines are in different stages of clinical testing while the only peptide vaccines in Phase III of clinical studies are cancer therapeutic vaccines [64].
3.2. Discovery of Antigenic Regions within Pneumococcal Proteins
Several methods have been used so far for the identification of antigenic regions within pneumococcal proteins. Among the standardized procedures, the most common one uses a panel of monoclonal antibodies to different domains and epitopes and independently evaluates their in vitro opsonophagocytic ability and in vivo protective efficacy. A more recent approach [65] uses human sera from individuals with natural infection as primary screening. Such an approach offers the possibility of identifying proteins that are not predicted by Open reading frame (ORF)-finding algorithms and allows a rapid selection of in vivo expressed antigens [66]. Following this, selected antigens are evaluated in preclinical studies to assess their potential as vaccine candidates. Based on this method, Giefing et al. identified large antigenic regions within several pneumococcal proteins (of a total length of 13494 amino acids), consistently recognized by adult patients with IPD [67]. Similarly, Beghetto et al. identified a panel of antigenic fragments within surface pneumococcal proteins; namely, the choline-binding protein D (CbpD), the pneumococcal histidine triad proteins PhtD and PhtE, the PspA, the plasminogen and fibronectin binding protein B (PfbB), and the zinc metalloproteinase B (ZmpB) [68]. However, in both reports mentioned above, the long length of the identified antigenic fragments hampers the construction of synthetic analogues that could be used as stable vaccine components. Therefore, a more recent study proceeded the identification of immunodominant B-cell epitopes within the antigenic regions previously identified by Beghetto et al. using a pediatric patient cohort with IPD [69]. The use of peptides in the design of pneumococcal vaccines is experiencing new enthusiasm due to the recent developments in peptide selection using different tools such as structural analysis by X-ray crystallography, Nuclear magnetic resonance (NMR) spectroscopy, mass spectrometry, and high-throughput screening techniques such as combinatorial chemistry, proteomics, genomics, and bioinformatics [70,71]. However, the selected peptides, regardless of the method used, require further evaluation of their immunogenicity and protective efficacy to confirm their potential as alternative vaccine candidates. Table 2 shows the most studied proteins regarding the identification of the antigenic regions they contain.
Table 2.
The most studied proteins regarding the identification of the antigenic regions they contain.
| Protein Name | Function | Virulence Factor | Location | 3-D structure of the Full Length Proteins |
|---|---|---|---|---|
| Pneumococcal surface protein A (PspA) | Cellular Metabolism and Immune Evasion | Choline-Binding Proteins (CBPs) | Bound to the cell wall via PCho moiety |
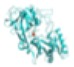
|
| Pneumococcal Choline-Binding Protein A (PcpA) | Protection Against Lung Infection and Sepsis | Choline-Binding Proteins (CBPs) | Bound to the cell wall via PCho moiety |

|
| Secreted 45-KDa Protein, Usp45-Hydrolase (PcsB) | Virulence | Non-Classical Surface-Exposed Proteins | Membrane |

|
| Serine/Threonine Protein Kinase (StkP) | Cellular Metabolism and Fitness | Non-Classical Surface-Exposed Proteins | Membrane |

|
| Peptide Permease Enzyme, Manganese ABC Transporter (PsaA) | Immune Evasion | Lipoproteins | Surface of the cell wall |

|
| Pneumolysin D (PlyD) | Cytolytic Toxin, Adherence, Immune Evasion, Invasion, Dissemination and Complement Activation | Non-Classical Surface-Exposed Proteins | Cytoplasmic toxin |

|
| Pneumolysin toxoid (dPly) | Toxoid | Non-Classical Surface-Exposed Proteins | Toxoid | N.A. * |
| Choline-binding protein D (CbpD) | Colonization | Choline-Binding Proteins (CBPs) | Bound to the cell wall via PCho moiety |

|
| Histidine triad protein D (PhtD) | Adherence and Immune Evasion | Lipoproteins | Surface of the cell wall |

|
| Histidine triad protein E (PhtE) | Adherence and Immune Evasion | Lipoproteins | Surface of the cell wall |

|
| Histidine triad protein A (PhtA) | Adherence and Immune Evasion | Lipoproteins | Surface of the cell wall | N.A. * |
| Histidine triad protein B (PhtB) | Adherence and Immune Evasion | Lipoproteins | Surface of the cell wall | N.A. * |
| Plasmin and Fibronectin-Binding Protein A (PfbB) | Adherence, Immune Evasion and Antiphagocytosis | LPxTG—Proteins | Anchored to well wall by the enzyme sortase A |

|
| Zinc metalloproteinase B (ZmpB) | Immune Evasion and Colonization | LPxTG—Proteins | Anchored to well wall by the enzyme sortase A |

|
* The 3-D structure of the corresponding protein has not been defined yet.
3.3. Potential Advantages of Antigenic Regions/Peptides within Pneumococcal Proteins Compared to Full-Length Proteins as Vaccine Candidates
Peptide-based vaccines offer several benefits compared to whole protein-based vaccines, including chemical stability and easier production processes, as they do not require folding into a tertiary 3-D structure [72]. Conjugation of various peptides from different antigens to the same carrier is much easier than conjugation of recombinant proteins. Such vaccines are considered safer than protein-based vaccines as they allow the exclusion of antigen fragments associated with autoimmune responses or toxicity. Synthetic peptides are inexpensive, requiring a much cheaper manufacturing process than recombinant proteins.
Synthetic peptides may induce better T-cell responses as vaccine antigens compared to full protein vaccines [73,74]. In fact, peptides are more efficiently endocytosed, processed, and presented on major histocompatibility complex (MHC) molecules compared to full-length proteins. Another, less investigated aspect of the antigen handling by antigen presenting cells (APCs), indicates that antigen cross-presentation—on a mole-for-mole ratio—is better for peptides than proteins. This is likely attributed to the efficient translocation of peptides into the cytoplasm from endosomes [75]. Sophisticated delivery systems can be easily applied in the design of peptide-based pneumococcal vaccines [72]. Their synthetic chemical stability allows an easier development of standardized in vitro assays in order to measure correlates of protection. Such immunological correlates may not be limited in the induction of opsonophagocytic antibodies, but also in the impairment of bacterial adherence to human nasopharyngeal and/or lung epithelial cells or in the induction of an efficient T-helper 17-mediated immune response to modulate neutrophil recruitment in the nasopharynx. A more comprehensive understanding of various factors influencing vaccine responses in a particular population/geography would be more easily defined using small synthetic peptides for vaccine development and deployment strategies instead of full-length recombinant proteins. Thus, the issue of constant natural exposure in developing countries associated with lower vaccine efficacy of protein-based pneumococcal vaccines would most likely be easier to overcome using peptides as vaccine antigens.
3.4. Protein Fragments as Vaccine Antigens
Significant progress has been achieved so far on the identification of antigenic regions, peptides, and/or B-cell epitopes within pneumococcal proteins and their evaluation as pneumococcal vaccine candidates.
PspA is a widely studied pneumococcal protein, expressed on the surface of all isolated strains, interfering with complement and C-reactive protein (CRP) deposition on the bacterial surface and immune adherence to erythrocytes [76,77,78,79]. Besides, PspA binding to lactoferrin via electrostatic interactions, specifically blocks this bactericidal peptide, preventing it from penetrating the bacterial membrane and killing pneumococcus [80]. PspA contains a variable α-helical N-terminal domain, an antigenically conserved proline-rich region (PRR), which is often interrupted by a non-proline block (NPB) and a choline-binding domain that anchors the protein to the bacterial outer cell wall [81,82]. Due to the high degree of amino-acid sequence variability among different pneumococcal strains, several PspA fragments mainly containing the N-terminal region, have been studied as vaccine candidates so far. Immunization of healthy adults with a single recombinant fragment of PspA containing the N-terminal region in a phase I clinical trial induced cross-reactive antibodies [49] that were able to induce passive protection in mice [47]. PspA fragments corresponding to the complete N-terminal regions of PspA protected mice against IPD caused by a strain expressing homologous PspA [83]. Remarkably, immunization of mice with a distinct 100-amino-acid fragment located within the more conserved N-terminal region of PspA induced protection in mice against pneumococcal lethal challenge [84,85]. In agreement with these reports, Roche et al. demonstrated the importance of C-terminal 104 and N-terminal 115 amino acids of the alpha-helical region of PspA in cross-protection of mice against IPD [83]. Considering that pneumococcal colonization is an essential precursor for disease pathogenesis, a more recent study reported that mice vaccination with recombinant salmonella outer membrane vehicles displaying the N-terminal part of the coiled-coil domain of PspA strongly reduces pneumococcal density in the nasopharynx, through an IL-17A dependent pathway [86]. The presence of one distinct epitope, common in different clinical isolates, strongly correlated with the induction of nasal IL-17A, that was, in turn, associated with a reduction of pneumococcal colonization [87].
Except from the N-terminal domains, immunization with recombinant PR (rPR) molecules and passive immunization with monoclonal antibodies reactive with either NPB or PR epitopes, alone or in combination with N-terminal fragments, were shown to be protective against IPD in mice [81,88,89]. However, there are concerns about PspA PR potential to cross react with the human protein myosin, which could induce autoimmune cardiac disorders [56]. Such an assumption has not been confirmed by Perciani et al., who reported that the presence of cross-reactive antibodies does not represent the risk of the development of an autoimmune response. A more recent study demonstrated that synthetic virus-like particles (SVLPs) carrying distinct B-cell epitope mimetics of length of no larger than 30-amino acids derived from the PR of PspA are highly immunogenic in mice without additional adjuvants and elicit cross-reactive significantly protective antibodies [90].
Histidine triad (Pht) protein family includes four highly conserved surface-exposed proteins (PhtA, PhtB, PhtD, and PhtE), characterized by the presence of five to six histidine triad (HxxHxH) motifs [91,92]. Pht proteins inhibit surface complement deposition and mediate bacterial adherence through zinc binding [91,92,93]. PhtD has been shown to display the least variability and is expressed by all pneumococcal strains screened thus far, with a 97–100% identity across different strains. Pht proteins have been widely considered as promising vaccine candidates. Indeed, immunization of mice with truncated derivatives from the C-terminal half of PhtD significantly increased their survival time after challenge with pneumococcus compared to immunization with the whole protein [94,95]. Further investigation revealed that immunization of mice with distinct B-cell epitopes within PhtD and PhtE proteins enhanced the survival of immunized mice compared to unimmunized controls when challenged with the high virulence pneumococcal serotype 3 [96]. Similar results were obtained for two distinct B-cell epitopes within CbpD involved in adherence, complement inhibition, and competence-induced cell lysis [97] and the bacterial adhesin ZmpB [98].
Finally, distinct antigenic regions have been identified within PsaA, mainly involved in manganese and zinc transportation, and a novel surface antigen encoded by ORF spr1875 in the R6 strain genome, involved in pneumococcal sepsis. In details, it has been reported that 15-mer peptides within PsaA protein selected through biopanning of a phage display library are immunogenic and protective in mice [99], while a 161 amino acid-long fragment within Spr1875 conferred immunoprotection in mice against experimental sepsis [100].
3.5. The Combination of Whole Proteins with Protein Fragments Demonstrated Protective Efficacy against Multiple Diseases in Mice
Another approach combines full-length proteins with distinct protein fragments in order to enhance their potential as vaccine antigens. Mice immunized with PsaA-PspA fragments (containing both family1 and 2 N-terminal region clade-defining region) were protected against fatal challenge with pneumococcal strains expressing different PspAs regardless of the challenge route [101]. Similarly, peptides derived from choline-binding protein A (CbpA), involved in adherence to cytokine-activated lung cells and complement deposition, genetically fused to detoxified pneumolycin, a cholesterol-dependent cytolysin that induces pore formation in the membrane of eukaryotic cells, protected mice from pneumococcal carriage, otitis media, pneumonia, bacteremia, meningitis, and meningococcal sepsis in active as well as passive immunization models [102]. Goulart et al. showed that PspA fragments including N-terminal domains plus PR regions fused to pneumolycin-derivatives increase the immune response mediated by cross-reactive antibodies and complement deposition to heterologous strains, confer protection against fatal challenge and reduce nasopharyngeal colonization in mice [103,104]. Chen et al. showed that multivalent pneumococcal protein vaccines comprising pneumolysoid with epitopes/fragments of CbpA and/or PspA elicit strong and broad protection in sepsis, meningitis, and focal pneumonia animal models [105].
3.6. Particular Adjuvants and Delivery Methods
Protein fragments as vaccine antigens, even those comprising optimal B-cell and T-cell epitopes, are poorly immunogenic. Suboptimal priming can be potentially circumvented using carriers and adjuvants that allow the targeting of APCs and contribute to an adequate immune stimulation. The most extensively used adjuvant so far is alum. Alum induces a TLR-dependent effect, increases antigen stability and enhances delivery to APCs. Although alum has been licensed for human vaccines for decades and is still widely used, its use in humans meets several obstacles. Alum does not have the ability to elicit T-helper 1 type immunity or cytotoxic T-cell responses and vaccines containing alum adjuvant cannot be sterilized by filtration, frozen, or lyophilized [106]. Another widely used immunostimulator in preclinical studies is Freud’s adjuvant. However, the highly effective Freund’s adjuvant is also reactogenic and frequently induces granulomas, sterile abscesses, and ulcerative necrosis at the site of inoculation, which precludes its use in human vaccines [107]. Another approach includes direct conjugation of long peptides to TLRs that may result in improved T-cell activation and antigen presentation leading to a more efficient vaccine. The presence of the antigen and the TLR ligand in the same endosomes determines the entrance to the presentation pathways [108]. Conjugation of antigenic peptides to TLR ligands like the TLR9-ligand CpG or the TLR1/2 heterodimer agonist Pam3CSK4 has been shown to strongly improve T-cell priming in vivo due to the combined effect of an increased uptake of long peptides and co-delivery with the immune-stimulatory signal [109,110]. Furthermore, the Pam3CSK4-conjugates were able to establish potent anti-tumor immune responses in multiple preclinical models and are now being tested in a phase I/II clinical trial evaluating synthetic peptide vaccination for treatment of Human Papillomavirus (HPV)-induced cancers [111,112]. Pneumococcal proteins when co-administrated with TLRs have been shown to induce significantly enhanced immunogenicity and protective efficacy in different animal models [113,114]. To the same direction, TLRs represent a promising delivery platform for the design of neo-epitope-based pneumococcal peptide vaccines. Furthermore, various advanced delivery systems have been studied so far to achieve the design of more efficient peptide-vaccines [115]. Over the past decade, nanoscale size (<1000 nm) materials such as virus-like particles (VLPs), outer-membrane vehicles (OMVs), liposomes, immune stimulating complexes (ISCOMs), polymeric, and non-degradable nanospheres have received attention as potential delivery vehicles for vaccine antigens which can both stabilize vaccine antigens and act as adjuvants. Besides, they offer the ability to design vaccines containing multiple protein antigenic fragments that specifically target immune cells, leading to more effective uptake by APCs [116,117,118,119,120]. VLPs, previously used as delivery system for pneumococcal protein antigens, are made from synthetic coiled-coil lipopeptides able to display multivalent epitope mimetics enhancing their presentation to the immune system, without the need for external adjuvants. VLPs are considered versatile tools in vaccine development due to their favorable immunological characteristics such as their size, repetitive surface geometry, their ability to induce both innate and adaptive immune responses, safety, and low cost. Several VLP-based vaccines are currently undergoing preclinical and clinical evaluation, [90,120] while VLP-based vaccines against HPV such as Cervarix®, Gardasil®, and Gardasil9® and Hepatitis B Virus (HBV) including the 3rd generation Sci-B-Vac™ are available on the market [121,122]. In addition, the first licensed malaria-VLP-based vaccine Mosquirix™ has been recently approved by the European regulators [123]. Bacterial OMVs represent another promising delivery system among nanoparticles used so far as delivery system for pneumococcal protein antigens. OMVs are vesicles of lipids released from the outer membranes of gram-negative bacteria to communicate among themselves and with other microorganisms in their environment. OMVs, displaying essential immunostimulatory properties, with the ability of antigen presentation on their surface and incorporation of heterologous antigens may be used as potent vaccine delivery systems. Such OMVs have been used for the design of the meningococcal B vaccine (Bexsero, Novartis) [124]. As for pneumococcus, PspA fragments and full-length proteins incorporated in Salmonella OMVs induced strong protection in a murine model of pneumococcal colonization, when intranasally administered, without the need for a mucosal adjuvant, in T-helper 17-mediated way [86,87].
Particular vaccine adjuvants or delivery systems used in peptide-based pneumococcal vaccines in pre-clinical studies so far are shown in Table 3.
Table 3.
Particular vaccine adjuvants or delivery systems used in peptide-based pneumococcal vaccines in pre-clinical studies so far.
| Protein | Fragment | Adjuvant | Reference |
|---|---|---|---|
| Pneumococcal surface proteins: CbpD, PhtD, PhtE, ZmpB | CbpD_pep4, PhtD_pep19, PhtE_pep40, ZmpB_pep125 and | Freund’s adjuvant | Papastamatiou et al., 2018 [96] |
| PspA | α1α2 fragment | OMVs (Salmonella Outer Membrane Vesicles) | Kuipers et al., 2017 [87] |
| PspA, PdT | PspA-PdT fusion protein | BCG (Bacillus Calmette-Guérin) | Goulart et al., 2017 [103] |
| PspA, PotD | PspA-PotD fusion protein | Al(OH)3 | Converso et al., 2017 [104] |
| PspA | linear peptides from the PRR or the NPB | SVLPs (synthetic Virus-Like Particles) | Tamborrini et al., 2015 [90] |
| PspA, Ply | N-terminal fragments of PspA, N-terminal & C-terminal fragments of Ply, fused to an Hbp-carrier | OMVs (Salmonella Outer Membrane Vesicles) | Kuipers et al., 2015 [86] |
| Ply, CbpA, PspA | combination of full-length or peptide regions of Ply, CbpA, or PspA | Alum | Chen et al., 2015 [105] |
| PspA, | PspA epitopes from the N-terminal region | Aluminum hydroxide | Vadesilho et al., 2014 [84] |
| SP_2108 and SP_0148 lipoproteins | 2108- 1912 (fusion construct of SP_2108 and SP_0148) | Cholera Toxin (CT)Aluminum hydroxide | Moffitt et al., 2014 [113] |
| PhtD | full length PhtD and truncated derivatives (PhtD, PhtD C1, PhtD N2 etc.) | E. coli labile toxin B subunit (LTB) | Plumptre et al., 2013 [95] |
| Protein encoded by the spr1875 of R6 strain genome | R4-GST fusion protein (glutathione S-transferase) (R4: 161 amino acid-long fragment) | Freund’s adjuvant | Cardaci et al., 2012 [100] |
| ZmpB, Ply, Dnal | rZmpB, rPly, rDnaJ, rZmpB + rPly, rZmpB + rDnaJ, rPly + rDnaJ, rZmpB + rPly + rDnaJ, | Freund’s adjuvant | Gong et al., 2011 [98] |
| PspA | N-terminal regions of clades 1,3 and 4 +PRR, N-terminal-PRR, N-terminal+ first block of PRR | Al(OH)3 | Darrieux et al, 2007 [83] |
| PspA | PR regions and NPB regions (PR+NPB _C, PR+NPB _N, PR+NPB, PR-NPB, or NPB) | Alum | Daniels et al., 2010 [82] |
| PspA | recombinant N-terminal His- tagged PspA protein | cholera toxin B subunit (CTB) | King et al., 2009 [89] |
| PspA | Recombinant PspAs | TLR-agonists: Pam3 Ultra Pure Escherichia coli K12 LPS, CSK4, Poly(I:C) and | Oma et al., 2009 [112] |
| Several proteins (i.e., PspA, PscB, StkP, PhtD etc) | N-terminal two thirds of PcsB, C-terminal half of StkP | Alum | Giefing et al., 2008 [67] |
| PspA | Six overlapping recombinant fragments of family 2, clade 3 PspA/EF3296 | AlOH3 | Roche et al., 2003 [125] |
| PhpA | Recombinant PhpA-79 | AlPO4 | Zhang et al., 2001 [92] |
| PsaA | peptide 43 (palmitoyl residue at the N-terminal end), 46 (same peptide without any ligands, nonlipidated), and PsaA | Aluminum hydroxide | Srivastava et al., 2000 [99] |
4. Conclusions
Current PCVs have been highly successful in reducing the incidence of IPD due to vaccine serotypes. However, several issues mainly associated with the emergence of non-vaccine serotypes have been a consistent, recurring problem.
Pneumococcal proteins that are universally expressed among all serotypes have been considered an interesting alternative for the development of serotype-independent pneumococcal vaccines. However, such an approach is reaching a crossroads regarding further evaluation.
A promising alternative may employ the use of antigenic protein fragments and peptides, several of which have been evaluated as vaccine antigens in pre-clinical studies, alone or in combination with whole pneumococcal proteins, as vaccine candidates. Such antigens retain the benefits of protein antigens i.e., immunogenicity in infants, induction of immune memory and natural serotype-independent “priming/boosting”. In the case of peptides, their low-cost and stable construction permits the combination of several highly-conserved antigens with the potential to activate both cellular and humoral immune responses and impair different stages of pneumococcal disease, offering even wider serotype coverage and more efficient protection. These vaccines would take advantage of newer delivery system technologies including conjugation to TLRs and reformulation of peptide antigens into nanoparticles that significantly enhance their immunogenicity. Such approaches would further benefit from the great advances in X-ray crystallography and NMR spectroscopy that allow precise structure determination, leading to synthesis of peptide analogs interfering with the immune system in the same way as native peptides of pneumococcal proteins.
Thorough understanding of the interaction of the immune system with the protein fragments is being currently elucidated. The possibilities for immune modulation using peptide antigens seem very broad and will depend on a more comprehensive knowledge of the underlying molecular mechanisms influencing this type of recognition. Innovative approaches in synthesis, structural analysis, conjugation, delivery, and immuno-monitoring will greatly increase the value of peptides in the design of novel pneumococcal vaccines.
Author Contributions
Each author made substantial contributions to the conception and design of the work. In detail, T.L. and P.B. collected the literature and drafted the article. J.R. revised the article and contributed to the design of the figures and tables. V.S. had the overall supervision and revised the article. All authors have approved the submitted version (and the version substantially edited by journal staff that involves the author’s contribution to the study) and agree to be personally accountable for the author’s own contributions and for ensuring that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and documented in the literature.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Ciapponi A., Elorriaga N., Rojas J.I., Romano M., Martí S.G., Bardach A., Ruvinsky S. Epidemiology of pediatric pneumococcal meningitis and bacteremia in Latin America and the caribbean: A systematic review and meta-analysis. Pediatr. Infect. Dis. J. 2014;33:971–978. doi: 10.1097/INF.0000000000000363. [DOI] [PubMed] [Google Scholar]
- 2.O’Brien K.L., Wolfson L.J., Watt J.P., Henkle E., Deloria-Knoll M., McCall N., Lee E., Mulholland K., Levine O.S., Cherian T. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: Global estimates. Lancet. 2009;374:893–902. doi: 10.1016/S0140-6736(09)61204-6. [DOI] [PubMed] [Google Scholar]
- 3.Mook-Kanamori B.B., Geldhoff M., van der Poll T., van de Beek D. Pathogenesis and pathophysiology of pneumococcal meningitis. Clin. Microbiol. Rev. 2011;24:557–591. doi: 10.1128/CMR.00008-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aligholi M., Emaneini M., Jabalameli F., Shahsavan S., Abdolmaleki Z., Sedaghat H., Jonaidi N. Antibiotic susceptibility pattern of gram-positive cocci cultured from patients in three university hospitals in Tehran, Iran during 2001–2005. Acta Med. Iran. 2009;47:329–334. [Google Scholar]
- 5.Bentley S.D., Aanensen D.M., Mavroidi A., Saunders D., Rabbinowitsch E., Collins M., Donohoe K., Harris D., Murphy L., Quail M.A., et al. Genetic analysis of the capsular biosynthetic locus from all 90 pneumococcal serotypes. PLoS Genet. 2006;2:262–269. doi: 10.1371/journal.pgen.0020031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cadoz M. Potential and limitations of polysaccharide vaccines in infancy. Vaccine. 1998;16:1391–1395. doi: 10.1016/S0264-410X(98)00097-8. [DOI] [PubMed] [Google Scholar]
- 7.Poolman J.T., Peeters C.C., Van Den Dobbelsteen G.P. The history of pneumococcal conjugate vaccine development: Dose selection. Expert Rev. Vaccines. 2013;12:1379–1394. doi: 10.1586/14760584.2013.852475. [DOI] [PubMed] [Google Scholar]
- 8.Kawaguchiya M., Urushibara N., Kobayashi N. Multidrug Resistance in Non-PCV13 Serotypes of Streptococcus pneumoniae in Northern Japan, 2014. Microb. Drug Resist. 2017;23:206–214. doi: 10.1089/mdr.2016.0054. [DOI] [PubMed] [Google Scholar]
- 9.McElligott M., Vickers I., Meehan M., Cafferkey M., Cunney R., Humphreys H. Noninvasive pneumococcal clones associated with antimicrobial nonsusceptibility isolated from children in the era of conjugate vaccines. Antimicrob. Agents Chemother. 2015;59:5761–5767. doi: 10.1128/AAC.00990-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Weinberger D.M., Malley R., Lipsitch M. Serotype replacement in disease following pneumococcal vaccination: A discussion of the evidence. Lancet. 2011;378:1962–1973. doi: 10.1016/S0140-6736(10)62225-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaur R., Casey J.R., Pichichero M.E. Emerging Streptococcus pneumoniae strains colonizing the nasopharynx in children after 13-valent pneumococcal conjugate vaccination in comparison to the 7-valent era, 2006–2015. Pediatr. Infect. Dis. J. 2016;35:901–906. doi: 10.1097/INF.0000000000001206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moraga-Llop F., Garcia-Garcia J.J., Díaz-Conradi A., Ciruela P., Martínez-Osorio J., González-Peris S., Hernández S., De Sevilla M.F., Uriona S., Izquierdo C., et al. Vaccine failures in patients properly vaccinated with 13-valent pneumococcal conjugate vaccine in Catalonia, a region with low vaccination coverage. Pediatr. Infect. Dis. J. 2016;35:460–463. doi: 10.1097/INF.0000000000001041. [DOI] [PubMed] [Google Scholar]
- 13.Mera R.M., Miller L.A., Daniels J.J., Weil J.G., White A.R. Increasing prevalence of multidrug-resistant Streptococcus pneumoniae in the United States over a 10-year period: Alexander Project. Diagn. Microbiol. Infect. Dis. 2005;51:195–200. doi: 10.1016/j.diagmicrobio.2004.10.009. [DOI] [PubMed] [Google Scholar]
- 14.Johnson H.L., Deloria-Knoll M., Levine O.S., Stoszek S.K., Hance L.F., Reithinger R., Muenz L.R., O’Brien K.L. Systematic evaluation of serotypes causing invasive pneumococcal disease among children under five: The pneumococcal global serotype project. PLoS Med. 2010;7 doi: 10.1371/journal.pmed.1000348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Balsells E., Guillot L., Nair H., Kyaw M.H. Serotype distribution of Streptococcus pneumoniae causing invasive disease in children in the post-PCV era: A systematic review and meta-analysis. PLoS ONE. 2017;12:e0177113. doi: 10.1371/journal.pone.0177113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Esposito S., Principi N. Direct and indirect effects of the 13-valent pneumococcal conjugate vaccine administered to infants and young children. Future Microbiol. 2015;10:1599–1607. doi: 10.2217/fmb.15.81. [DOI] [PubMed] [Google Scholar]
- 17.Kilpi T., Ahman H., Jokinen J., Lankinen K.S., Palmu A., Savolainen H., Grönholm M., Leinonen M., Hovi T., Eskola J., et al. Protective efficacy of a second pneumococcal conjugate vaccine against pneumococcal acute otitis media in infants and children: Randomized, controlled trial of a 7-valent pneumococcal polysaccharide-meningococcal outer membrane protein complex conjugate V. Clin. Infect. Dis. 2003;37:1155–1164. doi: 10.1086/378744. [DOI] [PubMed] [Google Scholar]
- 18.Fireman B., Black S.B., Shinefield H.R., Lee J., Lewis E., Ray P. Impact of the pneumococcal conjugate vaccine on otitis media. Pediatr. Infect. Dis. 2003;22:10–16. doi: 10.1097/00006454-200301000-00006. [DOI] [PubMed] [Google Scholar]
- 19.Rodgers G.L., Klugman K.P. Surveillance of the impact of pneumococcal conjugate vaccines in developing countries. Hum. Vaccines Immunother. 2016;12:417–420. doi: 10.1080/21645515.2015.1057671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Papadatou I., Spoulou V. Pneumococcal vaccination in high-risk individuals: Are we doing it right? Clin. Vaccine Immunol. 2016;23:388–395. doi: 10.1128/CVI.00721-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.CDC Vaccine Price List. [(accessed on 21 November 2018)]; Available online: https://www.cdc.gov/vaccines/programs/vfc/awardees/vaccine-management/price-list/index.html#f5.
- 22.McFetridge R., ter Meulen A.S., Folkerth S.D., Hoekstra J.A., Dallas M., Hoover P.A., Marchese R.D., Zacholski D.M., Watson W.J., Stek J.E., et al. Safety, tolerability, and immunogenicity of 15-valent pneumococcal conjugate vaccine in healthy adults. Vaccine. 2015;33:2793–2799. doi: 10.1016/j.vaccine.2015.04.025. [DOI] [PubMed] [Google Scholar]
- 23.Sobanjo-ter Meulen A., Vesikari T., Malacaman E.A., Shapiro S.A., Dallas M.J., Hoover P.A., McFetridge R., Stek J.E., Marchese R.D., Hartzel J., et al. Safety, tolerability and immunogenicity of 15-valent pneumococcal conjugate vaccine in toddlers previously vaccinated with 7-valent pneumococcal conjugate vaccine. Pediatr. Infect. Dis. J. 2015;34:186–194. doi: 10.1097/INF.0000000000000516. [DOI] [PubMed] [Google Scholar]
- 24.Moffitt K.L., Yadav P., Weinberger D.M., Anderson P.W., Malley R. Broad antibody and T-cell reactivity induced by a pneumococcal whole-cell vaccine. Vaccine. 2012;30:4316–4322. doi: 10.1016/j.vaccine.2012.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xu X., Meng J., Wang Y., Zheng J., Wu K., Zhang X., Yin Y., Zhang Q. Serotype-independent protection against pneumococcal infections elicited by intranasal immunization with ethanol-killed pneumococcal strain, SPY1. J. Microbiol. 2014;52:315–323. doi: 10.1007/s12275-014-3583-5. [DOI] [PubMed] [Google Scholar]
- 26.Lu Y.J., Lelte L., Gonçalves V.M., Dias Wde O., Liberman C., Fratelli F., Alderson M., Anderson P., Malley R. GMP-grade pneumococcal whole-cell vaccine injected subcutaneously protects mice from nasopharyngeal colonization and fatal aspiration-sepsis. Vaccine. 2010;28:7468–7475. doi: 10.1016/j.vaccine.2010.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lu Y.J., Gross J., Bogaert D., Finn A., Bagrade L., Zhang Q., Anderson P.W., Lipsitch M., Malley E. Interleukin-17A mediates acquired immunity to pneumococcal colonization. PLoS Pathog. 2008;4:e1000159. doi: 10.1371/journal.ppat.1000159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moffitt K., Malley R. Rationale and prospects for novel pneumococcal vaccines. Hum. Vaccines Immunother. 2016;12:383–392. doi: 10.1080/21645515.2015.1087625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Alderson M., Malley R., Anderson P., Thompson C., Morrison R., Briles D., King J., Goldblatt D., Green N., Hural J. A phase 1 study to assess the safety, tolerability and immunogenicity of inactivated non-encapsulated Streptococcus pneumoniae whole cell vaccine; Proceedings of the 9th International Symposium of Pneumococci and Pneumococcal Diseases; Hyderabad, India. 9–13 March 2014. [Google Scholar]
- 30.Briles D., King J., Hale Y., Malley R., Anderson P., Keech C., Tate A., Maisonneuve J., Alderson M. Immune sera from adults immunized with killed whole cell nonencapsulated vaccine protects mice from fatal infection with type 3 pneumococci; Proceedings of the 9th International Symposium of Pneumococci and Pneumococcal Diseases; Hyderabad, India. 9–13 March 2014. [Google Scholar]
- 31.Tai S.S. Streptococcus pneumoniae protein vaccine candidates: Properties, activities and animal studies. Crit. Rev. Microbiol. 2006;32:139–153. doi: 10.1080/10408410600822942. [DOI] [PubMed] [Google Scholar]
- 32.Feldman C., Anderson R. Review: Current and new generation pneumococcal vaccines. J. Infect. 2014;69:309–325. doi: 10.1016/j.jinf.2014.06.006. [DOI] [PubMed] [Google Scholar]
- 33.Kaur R., Surendran N., Ochs M., Pichichero M.E. Human antibodies to PhtD, PcpA, and ply reduce adherence to human lung epithelial cells and murine nasopharyngeal colonization by Streptococcus pneumoniae. Infect. Immun. 2014;82:5069–5075. doi: 10.1128/IAI.02124-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Robinson K.A., Baughman W., Rothrock G. Active Bacterial Core Surveillance (ABCs)/Emerging Infections Program Network. Epidemiology of invasive Streptococcus pneumoniae infections in the United States, 1995–1998: Opportunities for prevention in the conjugate vaccine era. JAMA. 2001;285:1729–1735. doi: 10.1001/jama.285.13.1729. [DOI] [PubMed] [Google Scholar]
- 35.Lipsitch M., Whitney C.G., Zell E., Kaijalainen T., Dagan R., Malley R. Are anticapsular antibodies the primary mechanism of protection against invasive pneumococcal disease? PLoS Med. 2005;2:e15. doi: 10.1371/journal.pmed.0020015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Malley R., Trzcinski K., Srivastava A., Thompson C.M., Anderson P.W., Lipsitch M. CD4+ T cells mediate antibody-independent acquired immunity to pneumococcal colonization. Proc. Natl. Acad. Sci. USA. 2005;102:4848–4853. doi: 10.1073/pnas.0501254102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Milner J.D., Brenchley J.M., Laurence A., Freeman A.F., Hill B.J., Elias K.M., Kanno Y., Spalding C., Elloumi H.Z., Paulson M.L., et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature. 2010;452:773–776. doi: 10.1038/nature06764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li Y., Gierahn T., Thompson C.M., Trzciński K., Ford C.B., Croucher N., Gouveia P., Flechtner J.B., Malley R., Lipsitch M. Distinct effects on diversifying selection by two mechanisms of immunity against Streptococcus pneumoniae. PLoS Pathog. 2012;8 doi: 10.1371/journal.ppat.1002989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Croucher N.J., Chewapreecha C., Hanage W.P., Harris S.R., McGee L., Van Der Linden M., Song J.H., Ko K.S., De Lencastre H., Turner C., et al. Evidence for soft selective sweeps in the evolution of pneumococcal multidrug resistance and vaccine escape. Genome Biol. Evol. 2014;6:1589–1602. doi: 10.1093/gbe/evu120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Croucher N.J., Harris S.R., Fraser C., Quail M.A., Burton J., Van Der Linden M., McGee L., Von Gottberg A., Song J.H., Ko K.S., et al. Rapid pneumococcal evolution in response to clinical interventions. Science. 2011;331:430–434. doi: 10.1126/science.1198545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Leroux-Roels G., Maes C., De Boever F., Traskine M., Rüggeberg J.U., Borys D. Safety, reactogenicity and immunogenicity of a novel pneumococcal protein-based vaccine in adults: A phase I/II randomized clinical study. Vaccine. 2014;32:6838–6846. doi: 10.1016/j.vaccine.2014.02.052. [DOI] [PubMed] [Google Scholar]
- 42.Seiberling M., Bologa M., Brookes R., Ochs M., Go K., Neveu D., Kamtchoua T., Lashley P., Yuan T., Gurunathan S. Safety and immunogenicity of a pneumococcal histidine triad protein D vaccine candidate in adults. Vaccine. 2012;30:7455–7460. doi: 10.1016/j.vaccine.2012.10.080. [DOI] [PubMed] [Google Scholar]
- 43.Berglund J., Vink P., Da Silva F.T., Lestrate P., Boutriau D. Safety, immunogenicity, and antibody persistence following an investigational Streptococcus pneumoniae and Haemophilus influenzae triple-protein vaccine in a phase 1 randomized controlled study in healthy adults. Clin. Vaccine Immunol. 2014;21:56–65. doi: 10.1128/CVI.00430-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Frey S.E., Lottenbach K.R., Hill H., Blevins T.P., Yu Y., Zhang Y., Brenneman K.E., Kelly-Aehle S.M., McDonald C., Jansen A., et al. A Phase, I.; dose-escalation trial in adults of three recombinant attenuated Salmonella Typhi vaccine vectors producing Streptococcus pneumoniae surface protein antigen PspA. Vaccine. 2013;31:4874–4880. doi: 10.1016/j.vaccine.2013.07.049. [DOI] [PubMed] [Google Scholar]
- 45.Bologa M., Kamtchoua T., Hopfer R., Sheng X., Hicks B., Bixler G., Hou V., Pehlic V., Yuan T., Gurunathan S. Safety and immunogenicity of pneumococcal protein vaccine candidates: Monovalent choline-binding protein A (PcpA) vaccine and bivalent PcpA-pneumococcal histidine triad protein D vaccine. Vaccine. 2012;30:7461–7468. doi: 10.1016/j.vaccine.2012.10.076. [DOI] [PubMed] [Google Scholar]
- 46.Kamtchoua T., Bologa M., Hopfer R., Neveu D., Hu B., Sheng X., Corde N., Pouzet C., Zimmermann G., Gurunathan S. Safety and immunogenicity of the pneumococcal pneumolysin derivative PlyD1 in a single-antigen protein vaccine candidate in adults. Vaccine. 2013;31:327–333. doi: 10.1016/j.vaccine.2012.11.005. [DOI] [PubMed] [Google Scholar]
- 47.Briles D.E., Hollingshead S.K., King J., Swift A., Braun P.A., Park M.K., Ferguson L.M., Nahm M.H., Nabors G.S. Immunization of humans with recombinant pneumococcal surface protein A (rPspA) elicits antibodies that passively protect mice from fatal infection with Streptococcus pneumoniae bearing heterologous PspA. J. Infect. Dis. 2000;182:1694–1701. doi: 10.1086/317602. [DOI] [PubMed] [Google Scholar]
- 48.Briles D.E., Ades E., Paton J.C., Sampson J.S., Carlone G.M., Huebner R.C., Virolainen A., Swiatlo E., Hollingshead S.K. Intranasal immunization of mice with a mixture of the pneumococcal proteins PsaA and PspA is highly protective against nasopharyngeal carriage of Streptococcus pneumoniae. Infect. Immun. 2000;68:796–800. doi: 10.1128/IAI.68.2.796-800.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nabors G.S., Braun P.A., Herrmann D.J., Heise M.L., Pyle D.J., Gravenstein S., Schilling M., Ferguson L.M., Hollingshead S.K., Briles D.E., et al. Immunization of healthy adults with a single recombinant pneumococcal surface protein A (PspA) variant stimulates broadly cross-reactive antibodies to heterologous PspA molecules. Vaccine. 2000;18:1743–1754. doi: 10.1016/S0264-410X(99)00530-7. [DOI] [PubMed] [Google Scholar]
- 50.Khan M.N., Pichichero M.E. Vaccine candidates PhtD and PhtE of Streptococcus pneumoniae are adhesins that elicit functional antibodies in humans. Vaccine. 2012;30:2900–2907. doi: 10.1016/j.vaccine.2012.02.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yun K.W., Choi E.H., Lee H.J. Genetic diversity of pneumococcal surface protein A in invasive pneumococcal isolates from Korean children, 1991–2016. PLoS ONE. 2017;12:e0183968. doi: 10.1371/journal.pone.0183968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gámez G., Castro A., Gómez-Mejia A., Gallego M., Bedoya A., Camargo M., Hammerschmidt S. The variome of pneumococcal virulence factors and regulators. BMC Genom. 2018;19:10. doi: 10.1186/s12864-017-4376-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Song J. Why do proteins aggregate_Intrinsically insoluble proteins and dark mediators revealed by studies on insoluble proteins solubilized in pure water. Version 1. F1000Research. 2013;2:94. doi: 10.12688/f1000research.2-94.v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Welsh R.M., Fujinami R.S. Pathogenic epitopes, heterologous immunity and vaccine design. Nature. 2007;5:555–563. doi: 10.1038/nrmicro1709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Alhamdi Y., Neill D.R., Abrams S.T., Malak H.A., Kadioglu A., Toh C.H. Circulating Pneumolysin is a potent inducer of cardiac injury during pneumococcal infection. PLoS Pathog. 2015;11:e1004836. doi: 10.1371/journal.ppat.1004836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Magyarics Z., Rouha H., Badarou A., Nielson N., Caccamo M., Weber S., Maierhofer B., Havlicek K., Dolezilkova I., Gross K., et al. Broadly cross-reactivity antibodies recognizing the proline-rich region of pneumococcal surface protein A variants show cross-reactivity with skeletal muscle; Proceedings of the 12th European Meeting on the Molecular Biology of the Pneumococcus; Oxford, UK. 7–10 July 2015. [Google Scholar]
- 57.Briles D., Hollingshead S.K. Euroconference on Infections and Lung Diseases (Paris, France) Institut Pasteur; Paris, France: 2006. Surface proteins of Streptococcus pneumoniae: Their roles in virulence and potential as vaccines. [Google Scholar]
- 58.McDaniel L.S., Swiatlo E. Should Pneumococcal Vaccines Eliminate Nasopharyngeal Colonization? MBio. 2016;7:e00545-16. doi: 10.1128/mBio.00545-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Singh M., Lingappan K. Whooping cough: The current scene. Chest. 2006;130:1547–1553. doi: 10.1016/S0012-3692(15)37335-9. [DOI] [PubMed] [Google Scholar]
- 60.Cohen J.M., Khandavill S., Camberiein E., Hyams C., Baxendale H.E., Brown J.S. Protective contributions against invasive Streptococcus pneumoniae pneumonia of antibody and Th17-cell responses to nasopharyngeal colonization. PLoS ONE. 2011;6:e25558. doi: 10.1371/journal.pone.0025558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ha S.-J., Mueller S.N., Wherry E.J., Barber D.L., Aubert R.D., Sharpe A.H., Freeman G.J., Ahmed R. Enhancing therapeutic vaccination by blocking PD-1–mediated inhibitory signals during chronic infection. J. Exp. Med. 2008;205:543–555. doi: 10.1084/jem.20071949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Xu Q., Casey J.R., Pichichero M.E. Higher levels of mucosal antibody to pneumococcal vaccine candidate proteins are associated with reduced acute otitis media caused by Streptococcus pneumoniae in young children. Mucosal Immunol. 2015;8:110–117. doi: 10.1038/mi.2015.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ren D., Almudevar A.L., Pichichero M.E. Synchrony in serum antibody response to conserved proteins of Streptococcus pneumoniae in young children. Hum. Vaccines Immunother. 2015;11:489–497. doi: 10.4161/21645515.2014.990861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tan A.C.L., Goubier A., Kohrt H.E. A quantitative analysis of therapeutic cancer vaccines in phase 2 or phase 3 trial. J. Immunother. Cancer. 2015;3 doi: 10.1186/s40425-015-0093-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rubinstein N.D., Mayrose I., Halperin D., Yekutieli D., Gershoni J.M., Pupko T. Computational characterization of B-cell epitopes. Mol. Immunol. 2008;45:3477–3489. doi: 10.1016/j.molimm.2007.10.016. [DOI] [PubMed] [Google Scholar]
- 66.Giefing C., Nagy E., von Gabain A. The antigenome: From protein subunit vaccines to antibody treatments of bacterial infections? Adv. Exp. Med. Biol. 2009;655:90–117. doi: 10.1007/978-1-4419-1132-2_9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Giefing C., Meinke A.L., Hanner M., Henics T., Minh D.B., Gelbmann D., Lundberg U., Senn B.M., Schunn M., Habel A., et al. Discovery of a novel class of highly conserved vaccine antigens using genomic scale antigenic fingerprinting of pneumococcus with human antibodies. J. Exp. Med. 2008;205:117–131. doi: 10.1084/jem.20071168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Beghetto E., Gargano N., Ricci S., Garufi G., Peppoloni S., Montagnani F., Oggioni M., Pozzi G., Felici F. Discovery of novel Streptococcus pneumoniae antigens by screening a whole-genome λ-display library. FEMS Microbiol. Lett. 2006;262:14–21. doi: 10.1111/j.1574-6968.2006.00360.x. [DOI] [PubMed] [Google Scholar]
- 69.Lagousi T., Routsias J., Piperi C., Tsakris A., Chrousos G., Theodoridou M., Spoulou V. Discovery of immunodominant B cell epitopes within surface pneumococcal virulence proteins in pediatric patients with invasive pneumococcal disease. J. Biol. Chem. 2015;290:27500–27510. doi: 10.1074/jbc.M115.666818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Khalili S., Jahangiri A., Borna H., Ahmadi Zanoos K., Amani J. Computational vaccinology and epitope vaccine design by immunoinformatics. Acta Microbiol. Immunol. Hung. 2014;61:285–307. doi: 10.1556/AMicr.61.2014.3.4. [DOI] [PubMed] [Google Scholar]
- 71.Sette A., Rappuoli R. Reverse vaccinology: Developing vaccines in the era of genomics. Immunity. 2010;33:530–541. doi: 10.1016/j.immuni.2010.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Skwarczynski M., Toth I. Peptide-based synthetic vaccines. Chem. Sci. 2016;7:842–854. doi: 10.1039/C5SC03892H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rosalia R.A., Quakkelaar E.D., Redeker A., Khan S., Camps M., Drijfhout J.W., Silva A.L., Jiskoot W., van Hall T., van Veelen P.A., et al. Dendritic cells process synthetic long peptides better than whole protein, improving antigen presentation and T-cell activation. Eur. J. Immunol. 2013;43:2554–2565. doi: 10.1002/eji.201343324. [DOI] [PubMed] [Google Scholar]
- 74.Zhang H., Hong H., Li D., Ma S., Di Y., Stoten A., Haig N., Di Gleria K., Yu Z., Xu X.N., et al. Comparing pooled peptides with intact protein for accessing cross-presentation pathways for protective CD8+ and CD4+ T cells. J. Biol. Chem. 2009;284:9184–9191. doi: 10.1074/jbc.M809456200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ménager J., Ebstein F., Oger R., Hulin P., Nedellec S., Duverger E., Lehmann A., Kloetzel P.M., Jotereau F., Guilloux Y. Cross-presentation of synthetic long peptides by human dendritic cells: A process dependent on ERAD component p97/VCP but not sec61 and/or Derlin-1. PLoS ONE. 2014;9 doi: 10.1371/journal.pone.0089897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Mukerji R., Mirza S., Roche A.M., Widener R.W., Croney C.M., Rhee D.-K., Weiser J.N., Szalai A.J., Briles D.E. Pneumococcal Surface Protein A Inhibits Complement Deposition on the Pneumococcal Surface by Competing with the Binding of C-Reactive Protein to Cell-Surface Phosphocholine. J. Immunol. 2012;189:5327–5335. doi: 10.4049/jimmunol.1201967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ren B., Li J., Genschmer K., Hollingshead S.K., Briles D.E. The absence of PspA or presence of antibody to PspA facilitates the complement-dependent phagocytosis of pneumococci In Vitro. Clin. Vaccine Immunol. 2012;19:1574–1582. doi: 10.1128/CVI.00393-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Mirza S., Wilson L., Benjamin W.H., Novak J., Barnes S., Hollingshead S.K., Briles D.E. Serine protease PrtA from Streptococcus pneumoniae plays a role in the killing of S. pneumoniae by apolactoferrin. Infect. Immun. 2011;79:2440–2450. doi: 10.1128/IAI.00489-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Shaper M., Hollingshead S.K., Benjamin W.H., Briles D.E. PspA protects Streptococcus pneumoniae from killing by apolactoferrin, and antibody to PspA enhances killing of pneumococci by apolactoferrin. Infect. Immun. 2004;72:5031–5040. doi: 10.1128/IAI.72.9.5031-5040.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lassing I., Schmitzberger F., Björnstedt M., Holmgren A., Nordlund P., Schutt C.E., Lindberg U. Molecular and Structural Basis for Redox Regulation of β-Actin. J. Mol. Biol. 2007;370:331–348. doi: 10.1016/j.jmb.2007.04.056. [DOI] [PubMed] [Google Scholar]
- 81.Hollingshead S.K., Becker R., Briles D.E. Diversity of PspA: Mosaic genes and evidence for past recombination in Streptococcus pneumoniae. Infect. Immun. 2000;68:5889–5900. doi: 10.1128/IAI.68.10.5889-5900.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Daniels C.C., Coan P., King J., Hale J., Benton K.A., Briles D.E., Hollingshead S.K. The proline-rich region of pneumococcal surface proteins A and C contains surface-accessible epitopes common to all pneumococci and elicits antibody-mediated protection against sepsis. Infect. Immun. 2010;78:2163–2172. doi: 10.1128/IAI.01199-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Darrieux M., Miyaji E.N., Ferreira D.M., Lopes M., Lopes A.P., Ren B., Briles D.E., Hollingshead S.K., Leite L.C. Fusion proteins containing family 1 and family 2 PspA fragments elicit protection against Streptococcus pneumoniae that correlates with antibody-mediated enhancement of complement deposition. Infect Immun. 2007;75:5930–5938. doi: 10.1128/IAI.00940-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Vadesilho C.F.M., Ferreira D.M., Gordon S.B., Briles D.E., Moreno A.T., Oliveira M.L.S., Ho P.L., Miyaji E.N. Mapping of epitopes recognized by antibodies induced by immunization of mice with PspA and PspC. Clin. Vaccine Immunol. 2014;21:940–948. doi: 10.1128/CVI.00239-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Olsen C.H. Review of the Use of Statistics in Infection and Immunity. Infect. Immun. 2003;71:6689–6692. doi: 10.1128/IAI.71.12.6689-6692.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kuipers K., Daleke-Schermerhorn M.H., Jong W.S.P., ten Hagen-Jongman C.M., van Opzeeland F., Simonetti E., Luirink J., de Jonge M.I. Salmonella outer membrane vesicles displaying high densities of pneumococcal antigen at the surface offer protection against colonization. Vaccine. 2015;33:2022–2029. doi: 10.1016/j.vaccine.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 87.Kuipers K., Jong W.S.P., van der Gaast-de Jongh C.E., Houben D., van Opzeeland F., Simonetti E., van Selm S., de Groot R., Koenders M.I., Azarian T., et al. Th17-mediated cross protection against pneumococcal carriage by vaccination with a variable antigen. Infect. Immun. 2017;85 doi: 10.1128/IAI.00281-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Moreno A.T., Oliveira M.L.S., Ferreira D.M., Ho P.L., Darrieux M., Leite L.C.C., Ferreira J.M.C., Pimenta F.C., Andrade A.L.S.S., Miyaji E.N. Immunization of mice with single PspA fragments induces antibodies capable of mediating complement deposition on different pneumococcal strains and cross-protection. Clin. Vaccine Immunol. 2010;17:439–446. doi: 10.1128/CVI.00430-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.King Q.O., Lei B., Harmsen A.G. Pneumococcal Surface Protein A Contributes to Secondary Streptococcus pneumoniae Infection after Influenza Virus Infection. J. Infect. Dis. 2009;200:537–545. doi: 10.1086/600871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Tamborrini M., Geib N., Marrero-Nodarse A., Jud M., Hauser J., Aho C., Lamelas A., Zuniga A., Pluschke G., Ghasparian A., et al. A Synthetic Virus-Like Particle Streptococcal Vaccine Candidate Using B-Cell Epitopes from the Proline-Rich Region of Pneumococcal Surface Protein A. Vaccines. 2015;3:850–874. doi: 10.3390/vaccines3040850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Rioux S., Neyt C., Di Paolo E., Turpin L., Charland N., Labbe S., Mortier M.C., Mitchell T.J., Feron C., Martin D., et al. Transcriptional regulation, occurrence and putative role of the Pht family of Streptococcus pneumoniae. Microbiology. 2011;157:335–348. doi: 10.1099/mic.0.042184-0. [DOI] [PubMed] [Google Scholar]
- 92.Zhang Y., Masi A.W., Barniak V., Mountzouros K., Hostetter M.K., Green B.A. Recombinant PhpA protein, a unique histidine motif-containing protein from Streptococcus pneumoniae, protects mice against intranasal pneumococcal challenge. Infect. Immun. 2001;69:3827–3836. doi: 10.1128/IAI.69.6.3827-3836.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kallio A., Sepponen K., Hermand P., Denoël P., Godfroid F., Melin M. Role of Pht proteins in attachment of Streptococcus pneumoniae to respiratory epithelial cells. Infect. Immun. 2014;82:1683–1691. doi: 10.1128/IAI.00699-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Godfroid F., Hermand P., Verlant V., Denoël P., Poolman J.T. Preclinical evaluation of the Pht proteins as potential cross-protective pneumococcal vaccine antigens. Infect. Immun. 2011;79:238–245. doi: 10.1128/IAI.00378-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Plumptre C.D., Ogunniyi A.D., Paton J.C. Vaccination against Streptococcus pneumoniae using truncated derivatives of polyhistidine triad protein D. PLoS ONE. 2013;8 doi: 10.1371/journal.pone.0078916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Papastamatiou T., Routsias J.G., Koutsoni O., Dotsika E., Tsakris A., Spoulou V. Evaluation of protective efficacy of selected immunodominant B-cell epitopes within virulent surface proteins of Streptococcus pneumonia. Infect. Immun. 2018;86 doi: 10.1128/IAI.00673-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kausmally L., Johnsborg O., Lunde M., Knutsen E., Håvarstein L.S. Choline-binding protein D (CbpD) in Streptococcus pneumoniae is essential for competence-induced cell lysis. J. Bacteriol. 2005;187:4338–4345. doi: 10.1128/JB.187.13.4338-4345.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Gong Y., Xu W., Cui Y., Zhang X., Yao R., Li D., Wang H., He Y., Cao J., Yin Y. Immunization with a ZmpB-based protein vaccine could protect against pneumococcal diseases in mice. Infect. Immun. 2011;79:867–878. doi: 10.1128/IAI.00717-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Srivastava N., Zeiler J.L., Smithson S.L., Carlone G.M., Ades E.W., Sampson J.S., Johnson S.E., Kieber-Emmons T., Westerink M.A. Selection of an immunogenic and protective epitope of the PsaA protein of Streptococcus pneumoniae using a phage display library. Hybridoma. 2000;19:23–31. doi: 10.1089/027245700315761. [DOI] [PubMed] [Google Scholar]
- 100.Cardaci A., Papasergi S., Midiri A., Mancuso G., Domina M., Cariccio V.L., Mandanici F., Galbo R., Lo Passo C., Pernice I., et al. Protective activity of Streptococcus pneumoniae Spr1875 protein fragments identified using a phage displayed genomic library. PLoS ONE. 2012;7:e36588. doi: 10.1371/journal.pone.0036588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lu J., Sun T., Wang D., Dong Y., Xu M., Hou H., Kong F.T., Liang C., Gu T., Chen P., et al. Protective immune responses elicited by fusion protein containing PsaA and PspA fragments. Immunol. Investig. 2015;44:482–496. doi: 10.3109/08820139.2015.1037956. [DOI] [PubMed] [Google Scholar]
- 102.Mann B., Thornton J., Heath R., Wade K.R., Tweten R.K., Gao G., El Kasmi K., Jordan J.B., Mitrea D.M., Kriwacki R., et al. Broadly protective protein-based pneumococcal vaccine composed of pneumolysin toxoid-CbpA peptide recombinant fusion protein. J. Infect. Dis. 2014;209:1116–1125. doi: 10.1093/infdis/jit502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Goulart C., Rodriguez D., Kanno A.I., Lu Y.J., Malley R., Leite L.C.C. Recombinant BCG expressing a PspA-PdT fusion protein protects mice against pneumococcal lethal challenge in a prime-boost strategy. Vaccine. 2017;35:1683–1691. doi: 10.1016/j.vaccine.2017.02.029. [DOI] [PubMed] [Google Scholar]
- 104.Converso T.R., Goulart C., Darrieux M., Leite L.C.C. A protein chimera including PspA in fusion with PhtD is protective against invasive pneumococcal infection and reduces nasopharyngeal colonization in mice. Vaccine. 2017;35:5140–5147. doi: 10.1016/j.vaccine.2017.08.010. [DOI] [PubMed] [Google Scholar]
- 105.Chen A., Mann B., Gao G., Heath R., King J., Maissoneuve J., Alderson M., Tate A., Hollingshead S.K., Tweten R.K., et al. Multivalent pneumococcal protein vaccines comprising pneumolysoid with epitopes/fragments of CbpA and/or PspA elicit strong and broad protection. Clin. Vaccine Immunol. 2015;22:1079–1089. doi: 10.1128/CVI.00293-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Magiri R., Mutwiri G., Wilson H.L. Recent advances in experimental polyphosphazene adjuvants and their mechanisms of action. Cell Tissue Res. 2018;374:465–471. doi: 10.1007/s00441-018-2929-4. [DOI] [PubMed] [Google Scholar]
- 107.Apostólico J.D.S., Alves V., Lunardelli S., Coirada F.C., Boscardin S.B., Rosa D.S. Adjuvants: Classification, Modus Operandi, and Licensing. J. Immunol. Res. 2016;2016:1459394. doi: 10.1155/2016/1459394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Blander J.M., Medzhitov R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature. 2006;440:808–812. doi: 10.1038/nature04596. [DOI] [PubMed] [Google Scholar]
- 109.Khan S., Bijker M.S., Weterings J.J., Tanke H.J., Adema G.J., Van Hall T., Drijfhout J.W., Melief C.J.M., Overkleeft H.S., Van Der Marel G.A., et al. Distinct uptake mechanisms but similar intracellular processing of two different toll-like receptor ligand-peptide conjugates in dendritic cells. J. Biol. Chem. 2007;282:21145–21159. doi: 10.1074/jbc.M701705200. [DOI] [PubMed] [Google Scholar]
- 110.Zom G.G., Welters M.J.P., Loof N.M., Goedemans R., Lougheed S., Valentijn R.R.P.M., Zandvliet M.L., Meeuwenoord N.J., Melief C.J.M., de Gruijl T.D., et al. TLR2 ligand-synthetic long peptide conjugates effectively stimulate tumor-draining lymph node T cells of cervical cancer patients. Oncotarget. 2016;7 doi: 10.18632/oncotarget.11512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Zom G.G., Khan S., Britten C.M., Sommandas V., Camps M.G.M., Loof N.M., Budden C.F., Meeuwenoord N.J., Filippov D.V., van der Marel G.A., et al. Efficient Induction of Antitumor Immunity by Synthetic Toll-like Receptor Ligand-Peptide Conjugates. Cancer Immunol. Res. 2014;2:756–764. doi: 10.1158/2326-6066.CIR-13-0223. [DOI] [PubMed] [Google Scholar]
- 112.Oma K., Zhao J., Ezoe H., Akeda Y., Koyama S., Ishii K.J., Kataoka K., Oishi K. Intranasal immunization with a mixture of PspA and a Toll-like receptor agonist induces specific antibodies and enhances bacterial clearance in the airways of mice. Vaccine. 2009;27:3181–3188. doi: 10.1016/j.vaccine.2009.03.055. [DOI] [PubMed] [Google Scholar]
- 113.Moffitt K., Skoberne M., Howard A., Gavrilescu L.C., Gierahn T., Munzer S., Dixit B., Giannasca P., Flechtner J.B., Malley R. Toll-like receptor 2-dependent protection against pneumococcal carriage by immunization with lipidated pneumococcal proteins. Infect. Immun. 2014;82:2079–2086. doi: 10.1128/IAI.01632-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Perrie Y., Kirby D., Bramwell V.W., Mohammed A.R. Recent Developments in Particulate-Based Vaccines. Recent Pat. Drug Deliv. Formul. 2007;1:117–129. doi: 10.2174/187221107780831897. [DOI] [PubMed] [Google Scholar]
- 115.Aikins M.E., Bazzill J., Moon J.J. Vaccine nanoparticles for protection against HIV infection. Nanomedicine. 2017;12:673–682. doi: 10.2217/nnm-2016-0381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Dowling D.J., Scott E.A., Scheid A., Bergelson I., Joshi S., Pietrasanta C., Brightman S., Sanchez-Schmitz G., Van Haren S.D., Ninković J., et al. Toll-like receptor 8 agonist nanoparticles mimic immunomodulating effects of the live BCG vaccine and enhance neonatal innate and adaptive immune responses. J. Allergy Clin. Immunol. 2017;140:1339–1350. doi: 10.1016/j.jaci.2016.12.985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zughaier S.M. Analysis of novel meningococcal vaccine formulations. Hum. Vaccines Immunother. 2017;13:1728–1732. doi: 10.1080/21645515.2017.1305528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Richard K., Mann B.J., Qin A., Barry E.M., Ernst R.K., Vogel S.N. Monophosphoryl lipid a enhances efficacy of a Francisella tularensis LVS-catanionic nanoparticle subunit vaccine against F. tularensis Schu S4 challenge by augmenting both humoral and cellular immunity. Clin. Vaccine Immunol. 2017;24 doi: 10.1128/CVI.00574-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Frietze K.M., Peabody D.S., Chackerian B. Engineering virus-like particles as vaccine platforms. Curr. Opin. Virol. 2016;18:44–49. doi: 10.1016/j.coviro.2016.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Mohsen M.O., Zha L., Cabral-Miranda G., Bachmann M.F. Major findings and recent advances in virus-like particle (VLP)-based vaccines. Semin Immunol. 2017;34:123–132. doi: 10.1016/j.smim.2017.08.014. [DOI] [PubMed] [Google Scholar]
- 121.Szarewski A. Drug Evaluation HPV vaccine: Cervarix. Expert Opin. Biol. Ther. 2010;10:477–487. doi: 10.1517/14712591003601944. [DOI] [PubMed] [Google Scholar]
- 122.Harper D.M., DeMars L.R. HPV vaccines—A review of the first decade. Gynecol. Oncol. 2017;146:196–204. doi: 10.1016/j.ygyno.2017.04.004. [DOI] [PubMed] [Google Scholar]
- 123.Wilby K.J., Lau T.T.Y., Gilchrist S.E., Ensom M.H.H. Mosquirix (RTS,S): A novel vaccine for the prevention of Plasmodium falciparum malaria. Ann. Pharmacother. 2012;46:384–393. doi: 10.1345/aph.1Q634. [DOI] [PubMed] [Google Scholar]
- 124.Holst J., Oster P., Arnold R., Tatley M.V., Næss L.M., Aaberge I.S., Galloway Y., Mcnicholas A., Hallahan J.O., Rosenqvist E., et al. Vaccines against meningococcal serogroup B disease containing outer membrane vesicles (OMV) Lessons from past programs and implications for the future. Hum. Vaccines Immunother. 2013;9:1241–1253. doi: 10.4161/hv.24129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Roche H., Håkansson A., Hollingshead S.K., Briles D.E. Regions of PspA/EF3296 best able to elicit protection against Streptococcus pneumoniae in a murine infection model. Infect. Immun. 2003;71:1033–1041. doi: 10.1128/IAI.71.3.1033-1041.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]


