Abstract
The POZ-ZF transcription factor Kaiso was discovered two decades ago as a binding partner for p120ctn. Since its discovery, roles for Kaiso in diverse biological processes (epithelial-to-mesenchymal transition, apoptosis, inflammation) and several signalling pathways (Wnt/β-catenin, TGFβ, EGFR, Notch) have emerged. While Kaiso’s biological role in normal tissues has yet to be fully elucidated, Kaiso has been increasingly implicated in multiple human cancers including colon, prostate, ovarian lung, breast and chronic myeloid leukemia. In the majority of human cancers investigated to date, high Kaiso expression correlates with aggressive tumor characteristics including proliferation and metastasis, and/or poor prognosis. More recently, interest in Kaiso stems from its apparent correlation with racial disparities in breast and prostate cancer incidence and survival outcomes in people of African Ancestry. This review discusses Kaiso’s role in various cancers, and Kaiso’s potential for driving racial disparities in incidence and/or outcomes in people of African ancestry.
Keywords: Kaiso, POZ-ZF transcription factors, Racial Disparities in Cancer, EMT, Inflammation
1. INTRODUCTION
The POZ-ZF transcription factor Kaiso (whose unique name is derived from the “Kaiso” genre of Caribbean music that has its origins in West Africa and is associated with dancing) was discovered almost two decades ago in association with p120 catenin (p120ctn), an Armadillo catenin responsible for regulating E-cadherin stability and turnover in adherens junctions [1]. At the time, several members of the POZ-ZF transcription factor family had been characterized as mediators of vertebrate development and cancer, hinting that Kaiso may also function in these processes [2]. As more evidence emerged surrounding the tumor suppressing role of E-cadherin, and the critical role of p120ctn in regulating E-cadherin function, significant efforts were made and are still ongoing to determine how Kaiso might function in tumorigenesis, perhaps as a regulator of E-cadherin– or p120ctn– related functions. Interestingly, early studies of Kaiso’s function in Xenopus development identified a role for Kaiso in antagonizing canonical Wnt signalling [3–5] and this set the stage for studies aimed at investigating Kaiso’s modulation of this pathway in the context of cancer. To date, multiple studies have implicated Kaiso in several different human cancers, however characterizing Kaiso as a bona fide tumor suppressor or oncogene has been challenging, as Kaiso’s function appears to be highly context-dependent. Nonetheless, diverse roles for Kaiso in key cancer-related processes and signalling pathways have been identified, revealing exciting avenues for further research. In this review, we will discuss some of the insight that has been gleaned into Kaiso’s structure and function and explore its identified roles in tumorigenesis as well as potential areas for continued study.
1.1. An overview of Kaiso structure, DNA binding and transcriptional activity
As is characteristic of the POZ-ZF family of proteins, Kaiso possesses an amino-terminal BTB/POZ (Broad complex, Tramtrack and Bric à brac/Poxvirus and Zinc finger, hereafter POZ) protein-protein interaction domain and a carboxy-terminal zinc finger (ZF) domain (Figure 1). The highly conserved POZ domain mediates homo- and heterodimeric interactions with other POZ and non-POZ proteins, whereas the ZF domain mediates DNA-binding, reviewed in [2]. Kaiso also contains several putative Serine/Threonine phosphorylation sites that have remained relatively understudied, although we and others have routinely observed a doublet consistent with a phosphorylated form of Kaiso in immunoblot analysis. Indeed, preliminary studies from the Daniel Lab have determined that Kaiso is predominantly phosphorylated on Serine/Threonine residues (our unpublished data).
Figure 1: Kaiso as a transcriptional activator and repressor.
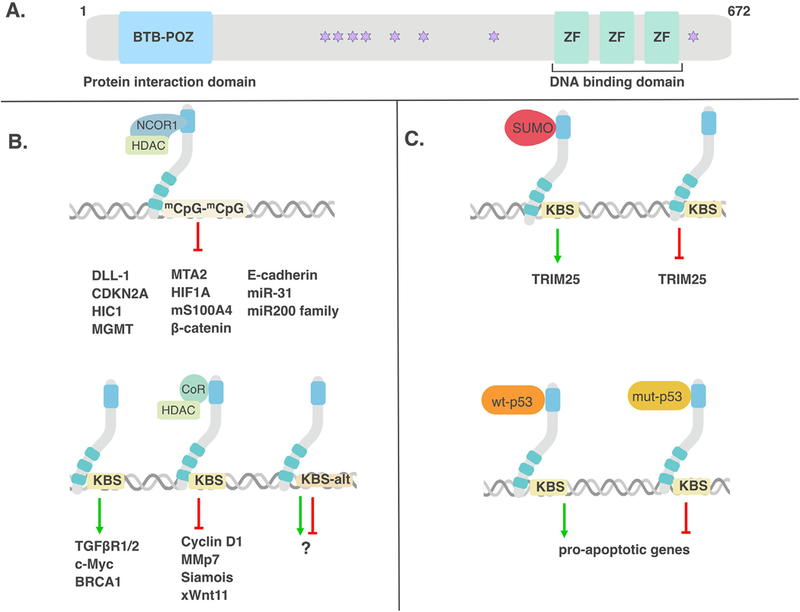
(A) Kaiso possesses a BTB/POZ protein-protein interaction domain and three DNA-binding ZFs. (B) Kaiso negatively regulates expression of many genes in a methylation-dependent manner but also positively regulates other genes via the core KBS. (C) Kaiso also switches between an activator or repressor dependent on its SUMOylation status. When SUMOylated, Kaiso acts as an activator but when de-SUMOylated, Kaiso acts as a repressor. Moreover, when Kaiso interacts with wild-type p53, it was found to activate pro-apoptotic genes whereas when associated with mutant p53, Kaiso repressed pro-apoptotic genes.
In our effort to fully describe the complexity of Kaiso’s biological function, it would be remiss not to mention the ongoing debates surrounding Kaiso’s DNA binding site(s) and its function as a transcriptional activator or repressor. Three different DNA sites have been reported as bona fide Kaiso binding sites, although to date, there is a lack of agreement as to which is the most favored or preferred Kaiso binding site. During our initial characterization of Kaiso, we identified a consensus DNA binding site, TCCTGCNA (core sequence bolded; N is any nucleotide) that was termed the Kaiso Binding Site (KBS) and we demonstrated an interaction between recombinant Kaiso proteins and KBS’s in the matrilysin promoter by EMSA [6]. In the same study, we also confirmed Kaiso’s binding to methylated CpG dinucleotides in oligonucleotides derived from the S100A4/Metastasin gene promoter [6], which was confirmed by Prokhortchouk et al. in an independent study [7]. These findings led to Kaiso being classified as a dual-specificity DNA-binding transcription factor [6]. Further analyses by our lab to determine the relative affinity of Kaiso for the KBS versus methyl-CpG (me-CpG) dinucleotides suggested that Kaiso possessed higher affinity for the KBS than for me-CpG dinucleotides [6]. However, a 2012 study by Raghav et al. – which found that promotor-proximal tethering of SMRT in terminal adipogenesis is mediated by Kaiso through a conserved, methylated TCTCGCGAGA motif – demonstrated a stronger interaction between Kaiso and the methylated TCTCGCGAGA motif than between Kaiso and the originally identified KBS [8]. In this study, the nucleotides flanking the core methylated CGCG motif were also shown to strengthen Kaiso’s binding to the me-CpG motif as mutation of these flanking sequences reduced the affinity of Kaiso for this site [8]. Following these early studies however, the relative affinity of Kaiso for the KBS versus me-CpG dinucleotides continues to be a topic of debate.
Through the analysis of Kaiso ChIP-seq datasets downloaded from the UCSC (University of California Santa Cruz) browser (from the ENCODE consortium), Blattler et al. demonstrated three key attributes of Kaiso’s endogenous interaction with DNA [9]. First, ~ 77% of Kaiso peaks were shown to overlap with RNA polymerase II peaks and enriched for active histone modifications, which suggested that Kaiso mostly binds to DNA regions that are actively transcribed or primed for transcription. Second, 36 – 43% of Kaiso peaks contained the TCTCGCGAGA motif identified by Raghav et al. [8], while the original KBS was not identified in any of the Kaiso peak sets. This finding suggests that additional studies should be conducted to clarify Kaiso’s DNA-binding properties and the importance/contributions of these different motifs to Kaiso’s respective downstream biological functions. Notably, most sites bound by Kaiso, including those containing the TCTCGCGAGA motif, were found to be unmethylated, indicating that Kaiso may prefer to bind to unmethylated DNA sites for transcriptional activity [9].
Irrespective of which site is the preferred or “correct” site for Kaiso binding, further research to clarify and identify Kaiso’s binding sites is warranted to enable a complete understanding of Kaiso’s transcriptional properties and function. This will also require an in-depth analysis of the role of the Kaiso-p120ctn interaction, since the interaction of p120ctn with Kaiso’s DNA-binding domain results in inhibition of Kaiso’s transcriptional activity and repression of target genes in the context of KBS-containing gene promoters [1, 10]. The cellular contexts under which p120ctn interacts with Kaiso are still not fully understood, and thus determining the upstream signalling pathways that regulate the Kaiso-p120ctn interaction will be essential for fully understanding Kaiso’s role in normal and tumorigenic processes.
Another layer of complexity with regards to Kaiso’s transcriptional role in normal and tumorigenic processes was heralded in 2006 upon the identification of two Kaiso-like proteins, ZBTB4 and ZBTB38 [11]. Intriguingly, ZBTB4, like Kaiso, was found to exhibit dual-specificity DNA binding to the KBS consensus sequence and me-CpG dinucleotides, while ZBTB38 was found to only bind me-CpG dinucleotides [11, 12]. Despite their structural similarity, Kaiso did not heterodimerize with ZBTB38 or ZBTB4, although an interaction between ZBTB38 and ZBTB4 was observed. Furthermore, no interaction between p120ctn and either of these two Kaiso-like proteins was observed [11]. Nonetheless, Kaiso, ZBTB4 and ZBTB38 were classified as a new family of methylated DNA-binding transcription factors that regulate gene expression and may function redundantly depending on cell or tissue context [11].
1.2. Kaiso - transcriptional repressor, activator or both?
Kaiso’s binding to methylated CpG dinucleotides, coupled with studies demonstrating interactions between Kaiso and SMRT-containing corepressor complexes (Figure 1), led to its initial characterization as a transcriptional repressor [7, 8]. Indeed, most studies to date have reported on Kaiso’s transcriptional repression of target genes including E-cadherin [13, 14], Wnt 11 [4], matrilysin [15], HIF1A [16], CDKN2A [17], miR-31 [18] and the miR-200 family [19]. While fewer studies have reported on Kaiso-mediated transcriptional activation of target genes [20, 21], recent studies suggest that transcriptional activation may be Kaiso’s preferred mode of transcriptional regulation. In support of this notion, we have found that twice as many genes are downregulated in response to Kaiso depletion in HCT-116 colon carcinoma cells than those that are upregulated (our unpublished data).
The notion that Kaiso may function as both an activator and repressor of transcription is not unprecedented since another POZ-ZF protein, Miz-1, has been reported to demonstrate both transcriptional repression and activation activities depending on the cofactors with which it interacts [22]. We have observed a similar phenomenon with Kaiso; its interaction with wild-type p53 activates transcription of pro-apoptotic genes, while its interaction with mutant p53 potentially represses transcription of pro-apoptotic genes (Figure 1) [23, 24]. Additionally, a recent study showed that while Kaiso’s transcriptional activities were mostly repressive in HeLa cells, many pathways were also activated in the presence of Kaiso [25]. This study also found opposing transcriptional activities of Kaiso between two cell lines, confirming that Kaiso DNA-binding and transcriptional activity is context and cell-type specific [25].
A recent study by Zhenilo et al. investigated the effect of post translational modifications on Kaiso’s transcriptional activity [26] and found that covalent linkage of small ubiquitin-like modifier (SUMO) polypeptides functioned as a molecular switch to regulate Kaiso’s function as an activator or repressor [26]. When SUMOylated, Kaiso acted as an activator but when deSUMOylated, Kaiso acted as a repressor (Figure 1). Using HEK293 cells, the authors demonstrated that Kaiso is SUMOylated under normal conditions at lysine 93 (K93); however, under hyperosmotic stress, Kaiso was found to be deSUMOylated [26]. This was the first study to elucidate a role for post-translational modifications on Kaiso function and indicates that further work is needed to examine other mechanisms (e.g. phosphorylation?) through which Kaiso could switch between its activating and repressive functions.
It is noteworthy that to date, studies reporting on Kaiso’s transcriptional activation all focus on Kaiso’s role when bound to the KBS. Thus, future studies are needed to elucidate whether the TCTCGCGAGA motif is also functionally linked to Kaiso-mediated transcriptional activation. Regardless of Kaiso’s specific transcriptional properties or preferred mode of DNA binding, it is clear that Kaiso and many of its putative target genes are implicated in or play key roles in tumorigenesis.
2. A Jack of all trades: the multifaceted functions of Kaiso in tumor development and progression
In a seminal review, Hanahan and Weinberg summarized the key acquired capabilities that are necessary for tumor growth and progression [27]. As we continue to unravel the details of Kaiso’s function, it has become evident that Kaiso is able to mediate several of these vital tumor-acquired capabilities including invasion, metastasis, apoptosis, cell proliferation, and inflammation (Figure 2; key references listed in Table 2). Thus, not surprisingly, Kaiso has been increasingly implicated in various human cancers (colon, prostate, lung, breast, ovarian, chronic myeloid leukemia) [28–32]. Here we review in depth, studies investigating Kaiso’s role in various cancers and tumor-related pathways.
Figure 2. Kaiso regulates several tumor-related processes as defined by Hanahan & Weinberg.
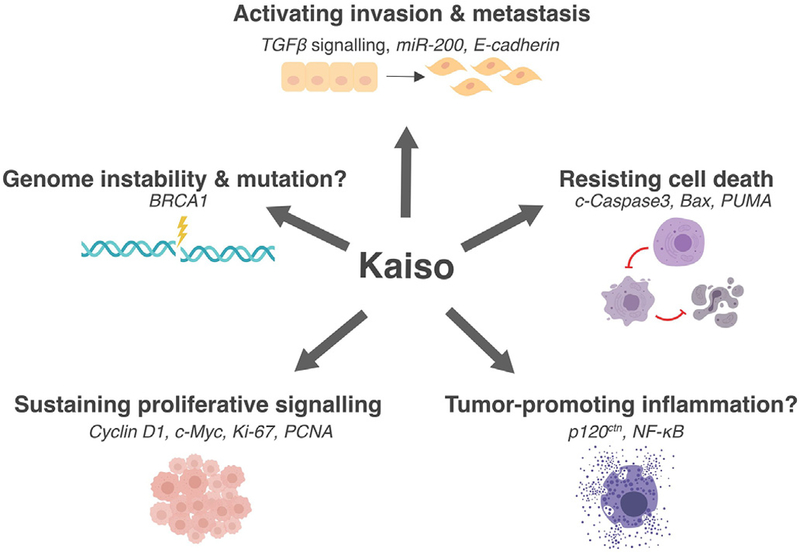
[27]. Kaiso activates tumor cell invasion and metastasis via regulation of TGFβ signalling, miR-200 family and E-cadherin expression. Kaiso sustains proliferative signalling through Cyclin-D1, Myc, Ki-67 and PCNA. As observed with several mouse models, Kaiso promotes intestinal inflammation. Kaiso is also implicated in resisting cell death through the regulation of c-Caspase3, Bax and PUMA. Finally, Kaiso is implicated in genome instability through its regulation of BRCA1.
Table 2-. Summary of cancer-related processes and genes regulated by Kaiso.
| Cancer site | Process affected as highlighted by research study |
Gene(s)/Protein(s) Implicated | Findings | References |
|---|---|---|---|---|
| Colorectal | Survival, tumor burden | - | Delayed polyp onset, extended lifespan & smaller polyps upon Kaiso depletion in ApcMin/+ mice | Prokhortchouk et al. [32] |
| Chemotherapeutic response | CDKN2A, HIC1 and &MGMT | Increased expression of tumor suppressor genes CDKN2A, HIC-1 & MGMT in Kaiso depleted CRC cell lines; Kaiso-depleted cells were sensitized to the cytotoxic drug emiddleoside | Lopes et al. [17] | |
| Inflammation | - | Chronic intestinal inflammation & crypt hyperplasia on intestinal-specific overexpression of Kaiso in C57BL/6J mice | Chaudhary et al. [46] | |
| Survival, tumor burden, inflammation | Wnt target genes; Pro-inflammatory markers | Shortened lifespan & increased polyp burden upon intestinal-specific overexpression of Kaiso in ApcMin/+ mice; increased expression of Wnt target genes & pro-inflammatory proteins | Pierre et al. [49] | |
| - | JAG1 | Positive regulation of JAG1 observed in both mice & CRC cell lines; JAG1 expression associated with Notch-mediated colon cancer | Robinson et al. [53] | |
| Prostate | Migration, invasion | E-cadherin | Reduced migration & invasion of Kaiso-depleted prostate cancer (PCa) cells with increased E-cadherin expression | Jones et al. [14] |
| Migration, invasion, proliferation | miR-31 | Reduced migration & invasion of Kaiso-depleted PCa cells with increased miR-31 expression | Wang et al. [16] | |
| Metastasis, tumor burden | miR-200 family; EMT regulators | Increased expression of miR-200 family (associated with suppression of EMT) and decreased expression of EMT regulators in Kaiso-depleted PCa cells; decreased tumor growth & fewer metastases in Kaiso-depleted PC3 xenografts | Abisoye-Ogunniyan et al. [19] | |
| Lung | Proliferation, invasion | - | Increased proliferation & invasion in Kaiso-depleted lung cancer cell lines | Dai et al. [28] |
| - | Cyclin-D1 | Kaiso overexpression decreased Cyclin-D1 & Cyclin-E1 expression | Jiang et al. [38] | |
| - | β-catenin | Kaiso overexpression reduced β-catenin expression | Liu et al. [70] | |
| CML | Proliferation and differentiation | SCF, Wnt11, C/EBPα, c-Myb, GATA-2, PU.1, CD15, CD11b, CD33 and CD117 | Kaiso depletion increased proliferation and decreased expression of differentiation markers. | Cofre et al. [30] |
| Breast | EMT, metastasis | EMT regulators; Cell adhesion factors | Induction of MET phenotype with decreased expression of key EMT markers & enhanced expression of cell-adhesion proteins in Kaiso-depleted TNBC cell lines; reduced metastasis of xenografted Kaiso-depleted TNBC cell lines. | Jones et al. [13] Bassey-Archibong et al. [83] |
| EMT, metastasis | TGFβR½ | Decreased expression of TGFβR½ with attenuation of TGFβ signalling in Kaiso-depleted TNBC cells; Constitutively active TGFβR½ rescued TGFβ signalling but not metastatic potential of xenografted Kaiso-depleted TNBC cells. | Bassey-Archibong et al. [83] | |
| Proliferation | CyclinD1, Myc, Ki76, PCNA | Reduced proliferation & restored anchorage dependence of Kaiso-depleted TNBC cell lines; reduced tumor volume of xenografted Kaiso-depleted TNBC cells; reduced expression of proliferation markers | Bassey-Archibong et al. [23] | |
| Apoptosis | C-Caspase3, Bax, PUMA | Induction of apoptosis with increased expression of pro-apoptotic proteins in mutant p53 Kaiso-depleted TNBC cells; enhanced sensitivity to cisplatain | ||
| DNA Repair | BRCA1 | Increased expression of BRCA1 in Kaiso-depleted TNBC cell lines | ||
| Glioma | Proliferation | PCNA, CDK2, CDK4, cyclin D1, cyclin E1, p21 and p27 | Kaiso depletion inhibited proliferation of a glioma cell line accompanied by reduced expression of cell cycle regulators PCNA, CDK2, CDK4, Cyclin D1 and Cyclin E1 and increased expression of p21 and p27. | Wang et al. [92] |
| Metastasis | E-cadherin, Vimentin, N-cadherin | Kaiso depletion resulted in reduced expression of mesenchymal markers (Vimentin, N-cadherin) and increased expression of epithelial markers (E-cadherin) | ||
2.1. Kaiso in Colorectal Cancer - Wnt pathway regulation or inflammation?
During the initial characterization of Kaiso as a transcription factor, several independent studies reported that Kaiso repressed a subset of Wnt/β-catenin target genes and inhibited Wnt signalling in several model systems [15, 33]. Park and colleagues were the first to report a role for Kaiso in the Wnt signalling cascade, a well-established cascade whose malfunction plays a key role in colon cancer [34]. Using a Xenopus model, they demonstrated that morpholino-mediated Kaiso depletion resulted in increased Wnt reporter activity and increased expression of several Wnt signalling targets including siamois, c-Myc, c-Fos and cyclinD1 [33]. Notably, Kaiso-mediated inhibition of Wnt target gene expression was attenuated by p120ctn overexpression [33]. Importantly, Kaiso was shown to rescue the duplicate-axis phenotype induced by constitutive Wnt signalling [33]. Based on these studies it was postulated that cross-talk existed between the p120ctn-Kaiso signalling trajectory and the canonical Wnt signalling pathway [35]. However, the interplay of Kaiso and p120ctn with canonical Wnt signalling in Xenopus development was further complicated by a later study that implicated Kaiso as a bimodal regulator of Wnt signalling [36]. In this study, both Kaiso depletion and overexpression inhibited Wnt signalling, while mild ectopic Kaiso expression resulted in increased Wnt signalling activity [36]. Unfortunately, none of these studies examined the expression levels or relevance of the Kaiso-like protein ZBTB4 on Wnt signaling in these models, and thus their apparent paradoxical findings may be explained by functional redundancy or competition with ZBTB4. Notwithstanding this paradox, Kaiso’s role as a regulator of Wnt signalling in Xenopus was solidified by a 2009 study that investigated a molecular mechanism for the interaction of the Xenopus homolog of Kaiso (xKaiso) with xTcf3 [5]. Ruzov et al. demonstrated that xKaiso interacted directly with xTcf3, leading to xTcf3 dissociation from the promoters of Wnt target genes [5]. These seminal studies examining Kaiso’s function in Xenopus development and Wnt signalling provided the rationale for further exploring Kaiso’s role in Wnt signalling in the context of mammalian model systems.
Studies in mammalian cell culture models further supported a bona fide role for Kaiso as a modulator of canonical Wnt signalling. Our lab and others reported that Kaiso represses a subset of Wnt target genes (cyclinD1, MMP7) in mammalian cultured cells [15, 37, 38]. Cell culture studies also hinted at a negative feedback loop whereby activation of the Wnt signalling pathway would inhibit Kaiso’s repressive effects on Wnt target genes [39]. Specifically, CK1ε phosphorylates p120ctn in response to Wnt stimulation, which promotes p120ctn’s association with Kaiso, and inhibits Kaiso’s transcriptional functions (Figure 3). In an extension of the studies performed by Ruzov et al., Kaiso was also found to interact with TCF4 and β-catenin in a manner that was mutually exclusive from its association with p120ctn [40]. Consequently, the Kaiso-p120ctn interaction hinders Kaiso’s ability to bind TCF4 and β-catenin to modulate Wnt signalling [39].
Figure 3-. Possible mechanisms of action of Kaiso in EMT.
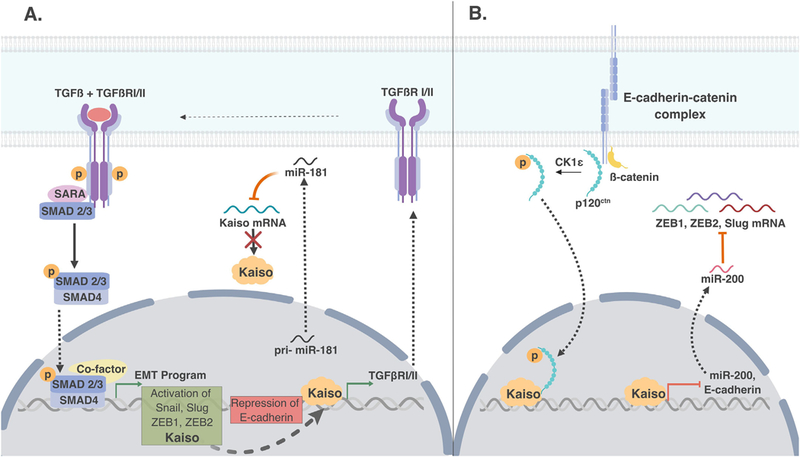
(A) Kaiso regulates TGF-β-mediated EMT and is itself regulated by the TGFβ signaling cascade which activates the expression of other pro-metastatic genes in triple negative breast tumors. The increase in Kaiso expression by TGFβ could be via the Smad transcription complex. Kaiso also appears to function in a positive feedback loop to increase TGFβ signaling (via increased TGFβRI & II expression) through as yet unidentified mechanism(s). (B) Kaiso represses E-cadherin expression through repression of miR-200 and possibly via sequestration of p120ctn in the nucleus. Phosphorylated p120ctn binds Kaiso, which leads to its sequestration in the nucleus and E-cadherin degradation.
Given the evidence linking Kaiso to Wnt signalling and the role of Wnt signalling in intestinal homeostasis and disease, the mammalian intestine represented an obvious choice for further studies into the physiological relevance of the Kaiso-Wnt relationship. Most colorectal cancers (CRCs), ~70%, are sporadic and driven by point mutations that follow a specific succession, with the first mutation occurring in the adenomatous polyposis coli (APC) tumor suppressor gene [41]. APC is a component of a large multi-protein complex that regulates the stability of β-catenin, the downstream effector of the canonical Wnt signalling cascade [42]. Most APC mutations that have been characterized in CRC result in a truncated APC protein, such that even in the absence of ligand-induced activation of the Wnt pathway, β-catenin is stabilized and translocates to the nucleus where it activates a plethora of tumorigenesis-promoting Wnt target genes [42–44].
The first study to examine the role of Kaiso in intestinal tumorigenesis was conducted in 2005, when Prokhortchouk et al. examined the effect of Kaiso depletion on polyp formation in the ApcMin/+ mouse model of intestinal neoplasia [32]. Surprisingly, Kaiso-deficient ApcMin/+ mice exhibited longer lifespans, delayed tumor onset and smaller tumors compared to their ApcMin/+ counterparts, suggesting that Kaiso functions in a pro-tumorigenic capacity in the intestinal setting [32]. Notably, the phenotype of Kaiso-deficient mice mimicked that of mice depleted for Mbd2, a methyl-CpG binding repressor [45]. Hypermethylation of tumor suppressor genes in various cancers is well documented [20], and has been proposed as a mechanism by which Kaiso may exert its effects in CRC [17]. Indeed, Kaiso depletion in CRC cell lines resulted in increased expression of three hypermethylated tumor suppressor genes, CDKN2A, HIC1, and MGMT and sensitized cells to the cytotoxic drug etoposide [17]. The expression of Kaiso in human intestinal tissue and in the Muc2−/− mouse model, which develops invasive colorectal tumors akin to those of patients with inflammatory bowel disease, was also investigated. Remarkably, Kaiso expression was found to be increased in the tumors of Muc2−/−mice compared to controls, although no difference was observed in Kaiso expression between matched normal and tumor human intestinal tissues in this study by Prokhortchouk [32].
In a quest to further understand the role of Kaiso in intestinal tumorigenesis and canonical Wnt pathway signaling, we generated an intestinal-specific Kaiso overexpressing mouse model (KaisoTg/+) [46]. While Kaiso overexpression alone was not sufficient to drive the development of tumors in the murine intestine, KaisoTg/+ mice developed crypt hyperplasia and chronic intestinal inflammation [46], which is an established risk factor for CRC. In fact, in patients with inflammatory bowel disease, the cumulative risk of developing colitis-associated cancer (CAC) ranges between 18–20% [16]. The development of chronic intestinal inflammation in Kaiso overexpressing mice is intriguing on two fronts. First, in contrast to sporadic CRC, Apc mutation occurs only in advanced stages of IBD- associated carcinogenesis if at all [16], which suggests a Wnt-independent mechanism by which Kaiso may promote intestinal tumorigenesis. Secondly, it suggests opposing roles for Kaiso (pro-) and p120ctn (anti-) in intestinal inflammation/tumorigenesis since limited ablation of p120ctn in murine intestines induced an inflammatory response characterized by an epithelial barrier defect with infiltrating neutrophils [47, 48], and promoted intestinal adenomas [48]. Most importantly, these findings hint that p120ctn and Kaiso may play a role in IBD progression to CAC.
We expanded our studies into the effect of Kaiso overexpression on intestinal homeostasis by crossing our KaisoTg/+ mice with ApcMin/+ mice [16, 49]. Consistent with earlier studies by Prokhortchouk et al., KaisoTg/+:ApcMin/+ mice exhibited significantly reduced lifespans and an approximately 3-fold increase in polyp number relative to control mice [49]. KaisoTg/+:ApcMin/+ mice also developed chronic intestinal inflammation, albeit at a younger age than KaisoTg/+ mice [49]. We also investigated the expression of Wnt target genes in the intestines of KaisoTg/+ and KaisoTg/+:ApcMin/+ mice [49]. Notably, Wnt target gene expression was increased rather than decreased in both KaisoTg/+ and KaisoTg/+:ApcMin/+ mice; however the increases noted in KaisoTg/+ mice compared to control mice were not significant [16, 49]. We also observed that increased Kaiso expression positively correlates with tumor stage in a human CRC tissue microarray (TMA) [49] and analysis of publicly available CRC microarray datasets revealed a statistically significant positive correlation between increased Kaiso expression and colon tumor tissues compared to normal tissues [16, 49], in contrast to the Prokhortchouk study [32]. Collectively, these studies indicate that in mammalian models, Kaiso promotes rather than suppresses tumorigenesis as was initially anticipated based on our findings regarding Kaiso and Wnt signalling in Xenopus.
Another striking phenotype that we observed in KaisoTg/+ mice was the expansion of the secretory cell population (i.e. goblet, Paneth and enteroendocrine cells) in the small and large intestines, which was accompanied by an overall decrease in cell proliferation [46]. This phenotype was reminiscent of that observed upon inhibition of the Notch signalling pathway in the intestine [50–52]. Indeed, expression of the Notch pathway target gene Hes1 was decreased in KaisoTg/+ mice [46], hinting at a novel role for Kaiso in regulating cell fate decisions through Notch signalling in the murine intestine. Notably, Kaiso represses the expression of both the Notch1 receptor and the Notch ligand Dll-1 in CRC cell lines [53] but promoted Jagged-1 expression, which is implicated in colon cancer progression. This finding provides an additional mechanism whereby Kaiso may promote CRC and adds another layer of complexity to Kaiso’s role in intestinal homeostasis and colon cancer.
While no studies to date have pinpointed the precise molecular mechanisms by which Kaiso potentiates intestinal tumorigenesis, and a definitive link between Kaiso and Wnt signaling in intestines has not been established, it is clear that in the context of the intestine, Kaiso functions in a pro-tumorigenic manner. Thus, ongoing efforts in our lab and others seek to characterize Kaiso’s role in intestinal inflammation and investigate its contribution to CAC and CRC using various mouse models.
2.2. Kaiso as a regulator of EMT and Metastasis in Prostate Cancer
The first report examining Kaiso’s role in prostate cancer (PCa) surveyed Kaiso expression and subcellular localization in various tumor types including a small cohort of PCa patients [54]. In a subsequent study with a larger patient cohort that included normal, adjacent normal, and benign prostatic hyperplasia (BPH) samples, Kaiso was found to be overexpressed in malignant prostate tumors and its high expression correlated significantly with tumor grade, Gleason score and race [14]. Specifically, low-Gleason grade tumors exhibited predominantly cytoplasmic Kaiso expression, while the high-Gleason grade and metastatic tumors displayed increased nuclear Kaiso expression. Two additional noteworthy observations from this study were that, i) African-American (AA) men expressed more nuclear Kaiso compared to Caucasian men, and ii) nuclear Kaiso was present in cells within the basal layer of adjacent normal tissue. Interestingly, multiple reports have indicated that adult prostate stem cells are present in the basal layer of both human and mouse prostate tissues and serve as developmental stimulants in PCa progression [40]. Indeed, a recent study by Yates et al. utilized Keratin-14-CreER/ROSA-LacZ and Keratin-5-tTA-TRE-H2BGFP lineage tracing mouse models and reported that Keratin-5 and Keratin-14 positive basal cells survive multiple rounds of castration and hormone manipulation, thus highlighting their stem cell characteristics [40]. It is unclear whether Kaiso expression in the basal layer is playing a role in castration resistance and/or PCa progression, but further investigation should be done to fully elucidate the role of Kaiso in the basal layer.
Previous studies suggest that DNA methylation is responsible for modulating gene expression that ultimately affects morphological changes in PCa cells and epithelial-to-mesenchymal transition (EMT) [55–57]. This is especially important in PCa, as PCa cells frequently adopt an EMT phenotype [58–60]. EGFR has been well characterized as a promoter of EMT in multiple tumor types, including PCa [61]. To gain insight into Kaiso’s mechanism of action in metastatic PCa, several well-characterized PCa cell culture models of EMT (LNCaP, DU-145, DU-145WT - which genetically overexpress EGFR, and PC-3, which exhibit a similar pattern of gene expression with human prostate tumor tissues), were examined and it was found that the more invasive and metastatic cell lines demonstrated increased nuclear Kaiso expression [14]. Interestingly, Kaiso expression was increased and Kaiso localized to the nucleus when EGFR was overexpressed or cells were treated with EGF. Furthermore, Kaiso bound directly to CpG islands in the E-cadherin promoter of PC-3 cells and Kaiso depletion resulted in robust re-expression of E-cadherin, similar to that observed on treatment with the demethylation agent 5-aza-CdR [14]. Intriguingly, a previous report by Yates et al. found that inhibition of the autocrine EGFR loop (and likely the EGFR-induced hepatocyte growth factor/c-met autocrine loop), either by direct disruption of the signalling loop or by secondary site signalling trans-attenuation, resulted in decreased cell motility and invasiveness, concomitant with E-cadherin re-expression in both DU-145 and PC-3 cells. Inhibition of the autocrine EGFR loop was observed in Kaiso-depleted cells hinting at a role for Kaiso in the EGFR-driven EMT phenotype of aggressive PCa.
To gain further insight into the mechanisms underlying Kaiso-mediated PCa development and progression, Wang et al. examined miRNA arrays and found that several miRNAs were upregulated in PCa [18]. Thirteen miRNAs, including miR-31, were significantly differentially expressed in Kaiso-depleted PC3 cells and a direct, methylation-dependent association between Kaiso and the endogenous miR-31 promoter was reported. PCa patients with Kaiso high and miR-31 low tumors had worse overall survival relative to patients with only Kaiso high tumors or miR-31 low tumors, suggesting that Kaiso promotes poor PCa survival via regulation of miR-31 expression. Most recently, Kaiso was also implicated in regulating the miR-200 family that suppresses EMT in PCa cells [19]. Upon Kaiso depletion in selected PCa cells, there was a significant increase of these miRNAs—similar to what was observed upon demethylation—and a decrease in downstream EMT targets such as ZEB1/2, Twist and Snail. Furthermore, using an in vivo mouse xenograft model, Kaiso-depleted PC3 cells subcutaneously injected into mice exhibited decreased tumor growth and less metastases compared to mice injected with control PC3 cells [19]. Collectively, these data further highlight Kaiso’s role in EMT and PCa tumor metastasis. Since acquisition of the mesenchymal phenotype by carcinoma cells is not permanent and tumor cells that have undergone EMT may later revert to an epithelial state at the secondary metastatic site through a mesenchymal-epithelial-transition (MET) [62–66], Kaiso could play a major role in mediating these transitional states of the cell throughout tumor progression and metastasis.
2.3. Kaiso and lung cancer
Two research groups recently investigated the role of Kaiso in lung cancer development and patient outcomes. Analysis of representative tissue cores from 294 non-small cell lung cancer (NSCLC) patients by Dai and colleagues, showed significantly increased cytoplasmic expression of Kaiso in NSCLC tissues compared to normal bronchial epithelium (NBE) tissues [28]. Dai et al. also found that high Kaiso expression correlated significantly with advanced stage and lymph node metastasis in a cohort of lung cancer patients [28]. Furthermore, patients with high levels of cytoplasmic Kaiso expression had significantly lower survival rates compared to those with no cytoplasmic Kaiso expression. Notably, nuclear Kaiso was only detected in a few NSCLC cases (~5%) but it was not associated with any clinicopathological features. In contrast to what was observed in NSCLC and NBE tissues, Dai et al. found that Kaiso was primarily localised in the nucleus of three lung cancer cell lines used (BE1, LETP-A-2 and SPC-A-1). Using proliferation and invasion assays, they found that down-regulation of nuclear Kaiso expression led to increased proliferation and invasion of the three lung cancer cell lines used [28]. In a subsequent study investigating Kaiso function in NSCLC, cytoplasmic Kaiso expression was positively correlated with high TNM stage, lymph node metastases and poor prognosis of NSCLC patients [67]. Notably, high cytoplasmic co-expression of Kaiso and δ-catenin was not detected in normal lung tissue [67].
The dynamic subcellular localization of Kaiso in NSCLC as described above is regulated by its interaction with p120ctn. In a study investigating the role of the Kaiso-p120ctn interaction in various lung cancer cell lines, it was found that Kaiso bound primarily to p120ctn isoform 3 (p120–3A) and not isoform 1 (p120–1A) in these cell lines [68]. Transient transfection studies using A549 lung cancer cells revealed that both cytoplasmic and nuclear Kaiso expression was increased by p120–3A overexpression but was unchanged by p120–1A overexpression [68]. Moreover p120–3A-bound Kaiso was shuttled out of the nucleus through the chromosomal region maintenance export pathway [68]. Zhang et al. also found that Kaiso’s binding to p120ctn was dependent on p120ctn serine-288 phosphorylation (Ps288) in lung cancer cells and tissues. They further showed that higher levels of Kaiso co-precipitated with phosphorylated p120ctn compared to unphosphorylated p120ctn, suggesting that serine-288 phosphorylation may act as the molecular switch that mediates p120ctn-Kaiso binding in lung cancer cells [68]. Most recently, Iderzorig et al. [69] found that p120ctn co-localizes with Kaiso in the nucleus in tyrosine kinase inhibitor (TKI)-resistant NSCLC cells, while in TKI-sensitive NSCLC cells reduced co-localization was observed. Interestingly, TKI-resistant NSCLC cells also exhibited low levels of E-cadherin expression and high expression of EMT markers such as Twist, Slug and Snail. This is in contrast to TKI-sensitive NSCLC cells, where E-cadherin was expressed and low levels of the aforementioned EMT markers were observed [69]. Given the dependence of the Kaiso-p120ctn interaction on the status of p120ctn phosphorylation [68], it is possible that p120ctn is hyper-phosphorylated in TKI-resistant NSCLC cells, resulting in its sequestration by Kaiso and consequently, a reduction in E-cadherin protein stability concomitant with increased cell motility or metastasis. This phenomenon, though not explored, is likely in TKI-resistant NSCLC tumors, since Dai et al. have reported overexpression of Kaiso in NSCLC [28].
Additional studies to elucidate Kaiso’s role in lung cancer have proposed a role for Kaiso in regulating β-catenin expression in a methylation-dependent manner in lung cancer [70]. Notably, increased β-catenin expression in the nucleus and cytoplasm correlates with more aggressive clinical course and lower survival, and is extensively reviewed in [71]. Liu et al. found that treatment of lung cancer cell lines with the demethylation agent 5-Aza-CdR, resulted in increased β-catenin expression. However, high Kaiso expression significantly downregulated β-catenin mRNA expression and suggested that Kaiso may be regulating β-catenin expression. Indeed, ChIP analysis revealed that Kaiso associated with the endogenous β-catenin promoter via methylated CpG dinucleotides [70].
Recently, studies by Zhang et al. investigating Kaiso’s role in lung cancer found that cigarette smoke induced nuclear-to-cytoplasmic shuttling of Kaiso, thus reducing Kaiso’s ability to transcriptionally silence the expression of its pro-tumorigenic target genes (e.g. c-Fos) [72]. Cigarette smoke promoted complex formation between p120ctn and the cytoplasmic tail of Mucin-1 (MUC1-CT) and enhanced Kaiso binding to p120ctn, an association that resulted in Kaiso shuttling to the cytoplasm. The nuclear-to-cytoplasmic shuttling of Kaiso was dependent on p120ctn as lung epithelial cells lacking p120ctn did not exhibit any shuttling of Kaiso from the nucleus [72].
Perhaps the most crucial findings that have been gleaned from the study of Kaiso in lung cancer cells is that Kaiso’s subcellular localization and consequently, its function, can be altered through its interaction with phosphorylated p120ctn. These findings lend insight into the upstream signalling events that can regulate Kaiso function, which have remained relatively understudied.
2.4. Kaiso as a regulator of cell differentiation and proliferation in chronic myeloid leukemia (CML)
The role of Kaiso’s cytoplasmic localization was further explored in CML by Cofre et al. [30]. Using immunofluorescence and immunoblot analyses, Kaiso was observed to be highly expressed in the cytoplasm of these cells rather than the nucleus. Similar to what was found in lung cancer cells, knock down of Kaiso (and/or p120ctn) led to increased proliferation in these cells, possibly through increased expression of the proliferation marker SCF, and hinted at a possible role for Kaiso by itself or in association with p120ctn for CML proliferation [30]. Examination of the expression of hematopoietic differentiation genes Wnt11, C/EBPα, c-Myb, GATA-2 and PU.1 in Kaiso and/or p120ctn knock-down CML cells revealed that c-MyB expression was increased while Wnt 11, PU-1, C/EBPα and Gata-2 expression were decreased when compared to scrambled knock-down cells [30]. Knock down of Kaiso and/or p120ctn also led to decreased expression of the global cell differentiators CD15, CD11b, CD33 and CD117. Notably in the clinical context, high expression of cytoplasmic Kaiso was observed in the more aggressive form of CML [30]. These findings were the first to implicate Kaiso in CML and further investigation is warranted to fully assess Kaiso’s role and mechanism of action in CML.
2.5. Kaiso and breast cancer
Mounting evidence suggests that Kaiso functions as an important driver of aggressive breast cancers like the triple negative breast cancer (TNBC) subtype. Interestingly, TNBCs are characterized by high proliferation indices, high rates of recurrence and a propensity to metastasize [73–75]. Currently, TNBCs lack targeted therapies due to their lack of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor-2 (HER2) expression. [76]. Although some therapeutic response is achieved through chemotherapy, patients with TNBC often relapse, thus associating this BC subtype with poor outcomes and shortened overall survival [77–80]. Notably, TNBC, is more frequently diagnosed in women of African ancestry, and Kaiso’s role in TNBC racial disparity in incidence and outcomes will be discussed in a later section.
Vermeulen et al. examined Kaiso expression and localization in a large cohort of normal and tumor breast tissues and found that high-grade breast tumors and tumors with a high mitotic activity index exhibited more nuclear Kaiso expression compared to low-grade tumors [81]. They further found that Kaiso expression negatively correlated with ERα positivity, and that nuclear Kaiso was significantly higher in aggressive BC, particularly the basal/triple negative and HER2-amplified breast cancers compared to luminal-type BC [81]. Notably, ~70% of hereditary (BRCA1-associated) breast cancers exhibited nuclear Kaiso expression compared to ~30% of sporadic carcinomas [81].
In a subsequent study by Jones et al., it was found that high nuclear Kaiso expression positively correlated with local invasion, lymph node metastases and poorer overall survival in a large cohort of invasive ductal carcinoma (IDC) tumor tissue samples [13]. Jones et al. also found TNBC cell lines (MDA-MB-231 & MDA-MB-468, hereafter MDA-231 & MDA-468) exhibited higher nuclear expression of Kaiso compared to the non-metastatic MCF-7 breast cancer cell line where Kaiso mainly localized to the cytoplasm [13]. Kaiso depletion in the highly metastatic MDA-231 and MDA-468 cell lines resulted in reduced cell migration in both wound healing and Boyden chamber assays, and reduced invasion through matrigel assays, while Kaiso overexpression in MCF-7 cells resulted in their increased migration and invasion [13]. These findings hinted that Kaiso may be involved in the regulation of the EMT process that is characterized by loss of E-cadherin expression and is a key contributor to metastases and poor outcomes of cancer patients [13]. Indeed, Kaiso depletion in MDA-231 and MDA-468 cells that typically lack E-cadherin expression, resulted in re-expression of E-cadherin with an associated reduction in expression of mesenchymal-related factors including N-cadherin, Cadherin 11 and Vimentin [13]. Chromatin immunoprecipitation assays also revealed that Kaiso binds the E-cadherin promoter, but treatment of cells with the demethylating agent 5-Aza-CdR abolished Kaiso binding, suggesting that this interaction is methylation-dependent [13].
Multiple signal transduction pathways regulate EMT, but one of the most studied is the TGFβ signalling pathway, which directly activates the expression of key transcriptional mediators of EMT such as Snail, Slug, ZEB1, ZEB2, and Twist [82]. Notably, we found that Kaiso-depleted MDA-231 breast tumor cells exhibited decreased TGFβ signalling, proliferated slowly in vivo and unlike parental MDA-231 cells, did not metastasize to lungs or liver in mouse xenograft models [83]. Kaiso depletion also resulted in increased expression of the TGFβ receptors, TGFβR1 and TGFβR2, and Kaiso was found to bind to KBS sites in the promoter regions of both genes [83]. Remarkably, TGFβ signaling induced Kaiso expression in TNBC cells at both the transcript and protein levels [83], which hinted at a positive feedback loop between Kaiso and TGFβ signalling in TNBC cells. Notably, TGFβ signalling plays a paradoxical role in breast cancer; in early stage breast cancer, TGFβ acts as a tumor suppressor by inhibiting cell proliferation and inducing apoptosis, but in advanced and aggressive breast cancers, TGFβ promotes progression and metastasis partly through induction of EMT [84]. Thus, our findings suggested that Kaiso may be a key driver of metastasis in TNBC (Figure 3).
Beyond its role in TNBC metastasis, we more recently demonstrated that Kaiso also plays a critical role in TNBC cell proliferation and survival [23]. Kaiso-depleted MDA-231 cells exhibited delayed tumor onset and reduced expression of the cell proliferation markers Ki67 and PUMA in a murine xenograft model [23]. Kaiso depletion also resulted in increased apoptosis of TNBC cells expressing mutant p53 and up-regulation of the pro-apoptotic genes PUMA and Bax [23]. Intriguingly, BRCA1 expression was reduced in Kaiso-depleted Hs587T and MDA-231 cells, and high co-expression of Kaiso and BRCA1 correlated with poor overall survival of TNBC patients. Notably, some TNBC subtypes demonstrate decreased sensitivity to chemotherapeutic agents [85]. Thus our finding that Kaiso-depleted Hs578T and MDA-231 cells exhibit increased Cisplatin sensitivity suggest that Kaiso may promote BRCA-1-mediated chemotherapy resistance in TNBC [23].
It is noteworthy that in breast tumor cells expressing wild-type p53, Kaiso functions in a pro-apoptotic capacity [23]. This supports independent studies showing that Kaiso activates the pro-apoptotic gene APAF1 by forming a protein complex with wild-type p53, which in turn binds and activates the APAF1 promoter [24]. Another mechanism by which Kaiso may promote apoptosis of breast tumor cells is through methylation-dependent repression of the glucocorticoid receptor (GR) [86]. Indeed, Kaiso overexpression reduced the anti-apoptotic effect of GR activation, thus highlighting a possible adjuvant role for Kaiso in breast cancer patients treated with glucocorticoid combined chemotherapy [86]. Collectively, these studies highlight paradoxical roles for Kaiso in apoptosis and indicates that even within the same tissue type, Kaiso’s role is highly complex and context-dependent.
2.6. Kaiso, miR-181 and EMT in glioma
Further support of Kaiso’s role as a regulator of EMT and proliferation in cancer cells was recently demonstrated in glioma cell lines. The miR-181 family inhibits proliferation, invasion, migration and EMT of gliomas by targeting several genes [87–91]. High Kaiso expression was found in glioma tissue compared to adjacent normal tissues and importantly, a significant negative correlation was found between Kaiso and miR-181 expression in high grade gliomas [92]. miR-181a bound to Kaiso’s 3’ UTR and ectopic expression of a miR-181a mimic resulted in reduced expression of both Kaiso mRNA and protein suggesting that Kaiso is a miR-181a target. Furthermore, Kaiso overexpression rescued the anti-proliferative and anti-invasive effects of a miR-181a mimic suggesting that miR-181 elicits its inhibitory effects on proliferation, invasion, migration and EMT through Kaiso suppression [92]. Similar to findings in breast tumor cell lines, Kaiso depletion inhibited the proliferation of a glioma cell line accompanied by reduced expression of cell cycle regulators PCNA, CDK2, CDK4, Cyclin D1 and Cyclin E1 and increased expression of p21 and p27. Reduced expression of mesenchymal markers (Vimentin, N-cadherin) and increased expression of epithelial markers (E-cadherin) was also observed upon depletion of Kaiso in glioma cells [92], similar to what has been reported in both breast and prostate cancer cells [19, 83]. In addition to reinforcing Kaiso’s role as a regulator of EMT, these studies have also identified the miR-181 family as novel regulators of Kaiso expression.
3. Kaiso and racial disparities in cancer
In the past decade, increasing evidence indicates that genetically based racial disparities exist in various human cancers including breast and prostate. The high BC mortality rates in women of African ancestry (WAA) despite having lower incidence rates and lifetime risks of BC than Caucasian women, initially led many to think that this disparity was linked to socio-economic status [93–97]. However, several studies now suggest that genetic risk factors/predisposition contribute to the racial disparity in the prevalence and mortality of patients diagnosed with the TNBC subtype [95, 97, 98]. Hence, the recent discovery that high Kaiso expression correlates with aggressive BC subtypes like TNBC and shorter metastasis-free survival in WAA [13, 81], as well as with aggressive PCa in African American (AA) men [14] suggests that high Kaiso expression may be linked to the racial disparity in prevalence and/or outcomes of aggressive cancers in people of African ancestry.
As a first step in testing this hypothesis, the Daniel and Yates labs independently examined a cohort of breast cancer patients and observed that nuclear Kaiso is significantly overexpressed in breast tumor tissues of patients with an African ancestry [13, 99]. Jones et al. further observed that nuclear Kaiso was overexpressed in primary tumor and paired lymph node metastases of AA women compared to Caucasian women (CaW) [13], and AA women with nuclear Kaiso had a decreased overall survival compared to CaW with nuclear Kaiso expression. We extended these findings in a pilot human tissue microarray (TMA) study where we examined Kaiso expression and subcellular localization in a TMA comprised of TNBC tissues from West African (Nigeria) and Caribbean (Barbadian) women. These patient cohorts were selected based on the premise that they represent more homogeneous populations of African ancestry than African Americans. Bassey-Archibong et al. found that Nigerian and Barbadian women were diagnosed with TNBC at much younger ages than AA and CauW [99], and that nuclear Kaiso levels were significantly higher in Nigerian, Barbadian and AA women compared with CauW. The high levels of nuclear Kaiso expression in women of African Ancestry compared to their Caucasian counterparts, suggests a role for Kaiso in TNBC racial disparity, and forms the premise for ongoing studies in the Daniel lab to further decipher this phenomenon. Collectively, these studies raise the exciting possibility that Kaiso may be a useful prognostic marker for TNBC, especially in WAA.
Notably, high Kaiso expression also correlates significantly with aggressive prostate tumors in AA men who have more aggressive tumors compared to Caucasian men [14]. Kaiso expression, particularly nuclear Kaiso, is increased in AA PCa patients, and is associated with a worse overall survival in AA men compared to Caucasian men [14]. Finally, high Kaiso expression has also been linked to racial disparities in pancreatic ductal adenocarcinoma (PDAC) [100]. Jones et al. observed higher Kaiso expression in PDAC tissues of AA patients when compared with normal/adjacent tissues [100]. They also found a significant relationship between high Kaiso expression, higher grade and tumor size in AA PDAC patients. Collectively these studies highlight an unexpected and unique role for Kaiso in racial disparities in cancer.
4. CONCLUDING REMARKS
In the past decade, significant strides have been made in understanding how Kaiso contributes to various human cancers. Generally, high Kaiso expression correlates with poor prognosis and worse clinical outcomes in colorectal, prostate, lung and breast cancers. We also have more insight into the molecular mechanisms via which Kaiso may drive these outcomes, since it is now appreciated that Kaiso regulates several tumor-associated processes including EMT, proliferation, apoptosis and inflammation.
Contrary to early reports that characterized Kaiso as a transcriptional repressor, it is evident that the manner in which Kaiso functions to modulate transcription may depend on several different factors including its interaction partners (e.g. wild-type vs. mutant p53) as well as the site at which it binds DNA. Most molecular studies to date have investigated Kaiso binding to the consensus KBS or in the context of methylation; however, few studies have examined the Kaiso binding motif identified by Raghav et al. [8]. Novel regulatory pathways may be identified using this motif and further enhance our understanding of Kaiso’s roles in tumorigenic processes. Other factors that regulate the activity of Kaiso include (i) changes in subcellular localization, which can occur upon binding to phosphorylated p120ctn, (ii) phosphorylation, which may alter Kaiso’s protein interactions, subcellular localization and transcriptional activity and (ii) SUMOylation, which alters Kaiso’s transcriptional activity. However, it is still unclear the precise cascade of events that lead to p120ctn-, phosphorylation- or SUMO-mediated regulation of Kaiso and hence these factors represent exciting avenues for further investigation. Furthermore, Kaiso’s regulation by miR181a and other as yet unknown miRNAs demand further investigation in various cell lines and contexts.
Another exciting avenue of study revolves around the findings that Kaiso can promote inflammation in the intestine. The mechanisms and molecular pathways via which Kaiso promotes inflammation, Kaiso’s interplay with p120ctn in the inflammatory process and whether Kaiso elicits its pro-inflammatory effects in other tissues are all questions that warrant further investigation.
With increasing data demonstrating that high Kaiso expression correlates with aggressive tumor characteristics, most likely through Kaiso’s regulation of EMT and cell proliferation in multiple cancers, Kaiso represents a candidate for further investigation as a diagnostic or prognostic marker and potentially a therapeutic target. One important question to be addressed is what causes high Kaiso expression in aggressive tumors. Finally, the link between high Kaiso expression and the racial disparities in cancer outcomes was an unexpected finding and ongoing studies seek to determine the molecular mechanism, if any, underlying this phenomenon. Two decades worth of progress into understanding Kaiso’s relevance in various cancers, has led to many intriguing findings and has indeed primed us for an exciting new era of scientific discovery around Kaiso function.
Table 1: Correlation of Kaiso expression with histopathological/clinical features of cancers.
| Cancer Site | Methodology | Histopathological/ Clinical Correlation | References |
|---|---|---|---|
| Colorectal | Colon cancer TMA, GEO database | Higher Kaiso expression observed in primary & metastatic tumor biopsies compared to normal colon tissues | Pierre et al. [49] |
| Prostate | PCa TMA | Higher Kaiso expression malignant prostate tumors compared to BPH; high Kaiso expression correlated with high tumor grade, Gleason score & AAs |
Jones et al. [14] |
| TCGA database | High Kaiso/low mir-31 expression correlated with lower overall survival relative to patients with either high Kaiso only or miR-31 low only tumors | Wang et al. [16] | |
| Lung | NSCLC TMA | Higher cymiddlelasmic expression correlated with advanced stage lung cancer & poorer overall patient survival; higher cymiddlelasmic expression in tissues of NSCLC patients compared to NBE patient tissues | Dai et al. [28] |
| Pancreas | TMA; ONCOMINE database | High Kaiso expression in PDAC tissues compared with low Kaiso expression in normal/adjacent tissues; high Kaiso expression in PDAC tissues correlated with higher grade & tumor size | Jones et al. [100] |
| Breast | Breast cancer TMA | Higher nuclear Kaiso expression associated with higher nuclear tumor grade | Vermeulen et al. [29] |
| Breast cancer TMA | High nuclear Kaiso expression positively correlated with invasion, lymph node metastases & poorer /reduced overall survival of patients with IDC | Jones et al. [13] | |
| TCGA database | High Kaiso/BRCA1 expression correlated with lower overall survival of patients diagnosed with TNBC & other BC subtypes | Bassey-Archibong et al. [23] | |
| TNBC TMA | Higher nuclear Kaiso expression observed in TNBC tissues obtained from WAA compared to Caucasian women | Bassey-Archibong et al. [99] | |
ACKNOWLEDGEMENTS
All images created with BioRender.
Funding
JMD was funded by CIHR, NSERC, JHCCF, CBCF/CCS; CCP was supported by The Richard Fuller Memorial Scholarship for Excellence in Science; CY was funded U54-MD007585–26 (NIH/RCMI) [CY], U54 CA118623 (NIH/NCI) [CY], (NIH/NCI) 1 R21 CA188799-01 [CY]; and a Department of Defense Grant, PC120913, W81XWH-10-1-0543.
REFERENCES
- 1.Daniel JM and Reynolds AB, The catenin p120(ctn) interacts with Kaiso, a novel BTB/POZ domain zinc finger transcription factor. Mol Cell Biol, 1999. 19(5): p. 3614–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kelly KF and Daniel JM, POZ for Effect- POZ-ZF Transcription Factors in Cancer and Development. Trends in Cell Biology, 2006. 16(11): p. 578–587. [DOI] [PubMed] [Google Scholar]
- 3.Kim SW, et al. , Isolation and characterization of XKaiso, a transcriptional repressor that associates with the catenin Xp120(ctn) in Xenopus laevis. J Biol Chem, 2002. 277(10): p. 8202–8. [DOI] [PubMed] [Google Scholar]
- 4.Kim SW, et al. , Non-canonical Wnt signals are modulated by the Kaiso transcriptional repressor and p120-catenin. Nat Cell Biol, 2004. 6(12): p. 1212–20. [DOI] [PubMed] [Google Scholar]
- 5.Ruzov A, et al. , The interaction of xKaiso with xTcf3: a revised model for integration of epigenetic and Wnt signalling pathways. Development, 2009. 136(5): p. 723–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Daniel JM, et al. , The p120(ctn)-binding partner Kaiso is a bi-modal DNA-binding protein that recognizes both a sequence-specific consensus and methylated CpG dinucleotides. Nucleic Acids Res, 2002. 30(13): p. 2911–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Prokhortchouk A, et al. , The p120 catenin partner Kaiso is a DNA methylation-dependent transcriptional repressor. Genes Dev, 2001. 15(13): p. 1613–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Raghav SK, et al. , Integrative genomics identifies the corepressor SMRT as a gatekeeper of adipogenesis through the transcription factors C/EBPbeta and KAISO. Mol Cell, 2012. 46(3): p. 335–50. [DOI] [PubMed] [Google Scholar]
- 9.Blattler A, et al. , ZBTB33 binds unmethylated regions of the genome associated with actively expressed genes. Epigenetics Chromatin, 2013. 6(1): p. 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kelly KF, et al. , NLS-dependent nuclear localization of p120ctn is necessary to relieve Kaiso-mediated transcriptional repression. Journal of Cell Science, 2004. 117: p. 2675–2686. [DOI] [PubMed] [Google Scholar]
- 11.Filion GJ, et al. , A family of human zinc finger proteins that bind methylated DNA and repress transcription. Mol Cell Biol, 2006. 26(1): p. 169–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sasai N, Nakao M, and Defossez PA, Sequence-specific recognition of methylated DNA by human zinc-finger proteins. Nucleic Acids Res, 2010. 38(15): p. 5015–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jones J, et al. , Nuclear localization of Kaiso promotes the poorly differentiated phenotype and EMT in infiltrating ductal carcinomas. Clin Exp Metastasis, 2014. 31(5): p. 497–510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jones J, et al. , Nuclear Kaiso indicates aggressive prostate cancers and promotes migration and invasiveness of prostate cancer cells. Am J Pathol, 2012. 181(5): p. 1836–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Spring CM, et al. , The catenin p120ctn inhibits Kaiso-mediated transcriptional repression of the beta-catenin/TCF target gene matrilysin. Exp Cell Res, 2005. 305(2): p. 253–65. [DOI] [PubMed] [Google Scholar]
- 16.Saraggi D, et al. , The molecular landscape of colitis-associated carcinogenesis. Dig Liver Dis, 2017. 49(4): p. 326–330. [DOI] [PubMed] [Google Scholar]
- 17.Lopes EC, et al. , Kaiso contributes to DNA methylation-dependent silencing of tumor suppressor genes in colon cancer cell lines. Cancer Res, 2008. 68(18): p. 7258–63. [DOI] [PubMed] [Google Scholar]
- 18.Wang H, et al. , Kaiso, a transcriptional repressor, promotes cell migration and invasion of prostate cancer cells through regulation of miR-31 expression. Oncotarget, 2016. 7(5): p. 5677–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Abisoye-Ogunniyan A, et al. , Transcriptional repressor Kaiso promotes epithelial to mesenchymal transition and metastasis in prostate cancer through direct regulation of miR-200c. Cancer Lett, 2018. 431: p. 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Madakashira BP and Sadler KC, DNA Methylation, Nuclear Organization, and Cancer. Front Genet, 2017. 8: p. 76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rodova M, et al. , Regulation of the rapsyn promoter by kaiso and delta-catenin. Mol Cell Biol, 2004. 24(16): p. 7188–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wiese KE, et al. , The role of MIZ-1 in MYC-dependent tumorigenesis. Cold Spring Harb Perspect Med, 2013. 3(12): p. a014290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bassey-Archibong BI, et al. , Kaiso depletion attenuates the growth and survival of triple negative breast cancer cells. Cell Death Dis, 2017. 8(3): p. e2689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Koh DI, et al. , Transcriptional activation of APAF1 by KAISO (ZBTB33) and p53 is attenuated by RelA/p65. Biochim Biophys Acta, 2015. 1849(9): p. 1170–8. [DOI] [PubMed] [Google Scholar]
- 25.Pozner A, Terooatea TW, and Buck-Koehntop BA, Cell-specific Kaiso (ZBTB33) Regulation of Cell Cycle through Cyclin D1 and Cyclin E1. J Biol Chem, 2016. 291(47): p. 24538–24550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhenilo S, et al. , DeSUMOylation switches Kaiso from activator to repressor upon hyperosmotic stress. Cell Death Differ, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hanahan D and Weinberg RA, Hallmarks of cancer: the next generation. Cell, 2011. 144(5): p. 646–74. [DOI] [PubMed] [Google Scholar]
- 28.Dai SD, et al. , Cytoplasmic Kaiso is associated with poor prognosis in non-small cell lung cancer. BMC Cancer, 2009. 9: p. 178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vermeulen JF, et al. , Nuclear Kaiso Expression is Associated with High Grade and Triple-Negative Invasive Breast Cancer. PLoS One, 2012. 7(5): e37864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cofre J, et al. , Knock-down of Kaiso induces proliferation and blocks granulocytic differentiation in blast crisis of chronic myeloid leukemia. Cancer Cell Int, 2012. 12(1): p. 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lopes EC, et al. , Kaiso contributes to DNA methylation-dependent silencing of tumor suppressor genes in colon cancer cell lines. Cancer Res., 2008. 68(18): p. 7258–7263. [DOI] [PubMed] [Google Scholar]
- 32.Prokhortchouk A, et al. , Kaiso-deficient mice show resistance to intestinal cancer. Mol Cell Biol, 2006. 26(1): p. 199–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Park JI, et al. , Kaiso/p120-catenin and TCF/beta-catenin complexes coordinately regulate canonical Wnt gene targets. Dev Cell, 2005. 8(6): p. 843–54. [DOI] [PubMed] [Google Scholar]
- 34.Schatoff EM, Leach BI, and Dow LE, Wnt Signaling and Colorectal Cancer. Curr Colorectal Cancer Rep, 2017. 13(2): p. 101–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Daniel JM, Dancing in and out of the nucleus: p120(ctn) and the transcription factor Kaiso. Biochim Biophys Acta, 2007. 1773(1): p. 59–68. [DOI] [PubMed] [Google Scholar]
- 36.Iioka H, Doerner SK, and Tamai K, Kaiso is a bimodal modulator for Wnt/beta-catenin signaling. FEBS Lett, 2009. 583(4): p. 627–32. [DOI] [PubMed] [Google Scholar]
- 37.Donaldson NS, et al. , Kaiso Represses the Cell Cycle Gene cyclin D1 via Sequence-Specific and Methyl-CpG-Dependent Mechanisms. PLoS One, 2012. 7(11): p. e50398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jiang G, et al. , P120-catenin isoforms 1 and 3 regulate proliferation and cell cycle of lung cancer cells via beta-catenin and Kaiso respectively. PLoS One, 2012. 7(1): p. e30303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Del Valle-Perez B, et al. , Wnt controls the transcriptional activity of Kaiso through CK1epsilon-dependent phosphorylation of p120-catenin. J Cell Sci, 2011. 124(Pt 13): p. 2298–309. [DOI] [PubMed] [Google Scholar]
- 40.Zhao C, et al. , Dual regulatory switch through interactions of Tcf7l2/Tcf4 with stage-specific partners propels oligodendroglial maturation. Nat Commun, 2016. 7: p. 10883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Marmol I, et al. , Colorectal Carcinoma: A General Overview and Future Perspectives in Colorectal Cancer. Int J Mol Sci, 2017. 18(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fearon ER, Molecular genetics of colorectal cancer. Annu Rev Pathol, 2011. 6: p. 479–507. [DOI] [PubMed] [Google Scholar]
- 43.Gryfe R, Inherited colorectal cancer syndromes. Clin Colon Rectal Surg, 2009. 22(4): p. 198–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Behrens J and Lustig B, The Wnt connection to tumorigenesis. Int J Dev Biol, 2004. 48(5–6): p. 477–87. [DOI] [PubMed] [Google Scholar]
- 45.Sansom OJ, et al. , Deficiency of Mbd2 suppresses intestinal tumorigenesis. Nat Genet, 2003. 34(2): p. 145–7. [DOI] [PubMed] [Google Scholar]
- 46.Chaudhary R, et al. , The POZ-ZF transcription factor Kaiso (ZBTB33) induces inflammation and progenitor cell differentiation in the murine intestine. PLoS One, 2013. 8(9): p. e74160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Smalley-Freed WG, et al. , p120-catenin is essential for maintenance of barrier function and intestinal homeostasis in mice. J Clin Invest, 2010. 120(6): p. 1824–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Smalley-Freed WG, et al. , Adenoma formation following limited ablation of p120-catenin in the mouse intestine. PLoS One, 2011. 6(5): p. e19880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pierre CC, et al. , Kaiso overexpression promotes intestinal inflammation and potentiates intestinal tumorigenesis in Apc(Min/+) mice. Biochim Biophys Acta, 2015. 1852(9): p. 1846–55. [DOI] [PubMed] [Google Scholar]
- 50.Fre S, et al. , Notch signals control the fate of immature progenitor cells in the intestine. Nature, 2005. 435(7044): p. 964–8. [DOI] [PubMed] [Google Scholar]
- 51.Jensen J, et al. , Control of endodermal endocrine development by Hes-1. Nat Genet, 2000. 24(1): p. 36–44. [DOI] [PubMed] [Google Scholar]
- 52.Ueo T, et al. , The role of Hes genes in intestinal development, homeostasis and tumor formation. Development, 2012. 139(6): p. 1071–82. [DOI] [PubMed] [Google Scholar]
- 53.Robinson SC, et al. , Kaiso differentially regulates components of the Notch signaling pathway in intestinal cells. Cell Commun Signal, 2017. 15(1): p. 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Soubry A, et al. , Expression and nuclear location of the transcriptional repressor Kaiso is regulated by the tumor microenvironment. Cancer Res, 2005. 65(6): p. 2224–33. [DOI] [PubMed] [Google Scholar]
- 55.Ross-Adams H, et al. , HNF1B variants associate with promoter methylation and regulate gene networks activated in prostate and ovarian cancer. Oncotarget, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Palma G, et al. , Triple negative breast cancer: looking for the missing link between biology and treatments. Oncotarget, 2015. 6(29): p. 26560–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Majid S, et al. , miR-23b represses proto-oncogene Src kinase and functions as methylation-silenced tumor suppressor with diagnostic and prognostic significance in prostate cancer. Cancer Res, 2012. 72(24): p. 6435–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Josson S, et al. , Tumor-Stromal Interactions Influence Radiation Sensitivity in Epithelial- versus Mesenchymal-Like Prostate Cancer Cells. J Oncol, 2010. 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yates C, et al. , Novel three-dimensional organotypic liver bioreactor to directly visualize early events in metastatic progression. Adv Cancer Res, 2007. 97: p. 225–46. [DOI] [PubMed] [Google Scholar]
- 60.Yates CC, et al. , Co-culturing human prostate carcinoma cells with hepatocytes leads to increased expression of E-cadherin. Br J Cancer, 2007. 96(8): p. 1246–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Khan MI, et al. , Role of epithelial mesenchymal transition in prostate tumorigenesis. Curr Pharm Des, 2015. 21(10): p. 1240–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ruiz-Narvaez EA, et al. , Genetic variation in the insulin, insulin-like growth factor, growth hormone, and leptin pathways in relation to breast cancer in African-American women: the AMBER consortium. NPJ Breast Cancer, 2016. 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Senguven B, et al. , Comparison of methods for the extraction of DNA from formalin-fixed, paraffin-embedded archival tissues. Int J Med Sci, 2014. 11(5): p. 494–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Siegel RL, Miller KD, and Jemal A, Cancer Statistics, 2017. CA Cancer J Clin, 2017. 67(1): p. 7–30. [DOI] [PubMed] [Google Scholar]
- 65.Ahn SG, et al. , Molecular Classification of Triple-Negative Breast Cancer. J Breast Cancer, 2016. 19(3): p. 223–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jeong HM, et al. , Targeted exome sequencing of Korean triple-negative breast cancer reveals homozygous deletions associated with poor prognosis of adjuvant chemotherapy-treated patients. Oncotarget, 2017. 8(37): p. 61538–61550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Dai SD, et al. , Upregulation of δ-catenin is associated with poor prognosis and enhances transcriptional activity through Kaiso in non-small-cell lung cancer. Cancer Sci, 2011. 102(1): p. 95–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhang PX, et al. , p120-catenin isoform 3 regulates subcellular localization of Kaiso and promotes invasion in lung cancer cells via a phosphorylation-dependent mechanism. Int J Oncol, 2011. 38(6): p. 1625–35. [DOI] [PubMed] [Google Scholar]
- 69.Iderzorig T, et al. , Comparison of EMT mediated tyrosine kinase inhibitor resistance in NSCLC. Biochem Biophys Res Commun, 2018. 496(2): p. 770–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Liu Y, et al. , Kaiso interacts with p120-catenin to regulate beta-catenin expression at the transcriptional level. PLoS One, 2014. 9(2): p. e87537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jin J, et al. , Prognostic significance of β-catenin expression in patients with non-small cell lung cancer: a meta-analysis. Transl Lung Cancer Res, 2017. 6(1): p. 97–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zhang L, et al. , Cigarette Smoke Mediates Nuclear to Cytoplasmic Trafficking of Transcriptional Inhibitor Kaiso through MUC1 and P120-Catenin. Am J Pathol, 2016. 186(12): p. 3146–3159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lindner R, et al. , Molecular phenotypes in triple negative breast cancer from African American patients suggest targets for therapy. PLoS One, 2013. 8(11): p. e71915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Iqbal J, et al. , Differences in breast cancer stage at diagnosis and cancer-specific survival by race and ethnicity in the United States. Jama, 2015. 313(2): p. 165–73. [DOI] [PubMed] [Google Scholar]
- 75.Keenan T, et al. , Comparison of the Genomic Landscape Between Primary Breast Cancer in African American Versus White Women and the Association of Racial Differences With Tumor Recurrence. J Clin Oncol, 2015. 33(31): p. 3621–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Clark O, et al. , Targeted therapy in triple-negative metastatic breast cancer: a systematic review and meta-analysis. Core Evid, 2014. 9: p. 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kassam F, et al. , Survival outcomes for patients with metastatic triple-negative breast cancer: implications for clinical practice and trial design. Clin Breast Cancer, 2009. 9(1): p. 29–33. [DOI] [PubMed] [Google Scholar]
- 78.Houvenaeghel G, et al. , Axillary lymph node micrometastases decrease triple-negative early breast cancer survival. Br J Cancer, 2016. 115(9): p. 1024–1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wushou A, et al. , Development of triple-negative breast cancer radiosensitive gene signature and validation based on transcriptome analysis. Breast Cancer Res Treat, 2015. 154(1): p. 57–62. [DOI] [PubMed] [Google Scholar]
- 80.Sun W, et al. , Prognostic analysis of triple-negative breast cancer patients treated with adjuvant chemotherapy of fluorouracil, epirubicin and cyclophosphamide. Oncol Lett, 2016. 11(3): p. 2320–2326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Vermeulen JF, et al. , Nuclear Kaiso expression is associated with high grade and triple-negative invasive breast cancer. PLoS One, 2012. 7(5): p. e37864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Katsuno Y, Lamouille S, and Derynck R, TGF-beta signaling and epithelial-mesenchymal transition in cancer progression. Curr Opin Oncol, 2013. 25(1): p. 76–84. [DOI] [PubMed] [Google Scholar]
- 83.Bassey-Archibong BI, et al. , Kaiso depletion attenuates transforming growth factor-beta signaling and metastatic activity of triple-negative breast cancer cells. Oncogenesis, 2016. 5: p. e208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zarzynska JM, Two Faces of TGF-Beta1 in Breast Cancer %J Mediators of Inflammation. 2014. 2014: p. 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.O’Reilly EA, et al. , The fate of chemoresistance in triple negative breast cancer (TNBC). BBA Clin, 2015. 3: p. 257–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zhou L, et al. , Kaiso represses the expression of glucocorticoid receptor via a methylation-dependent mechanism and attenuates the anti-apoptotic activity of glucocorticoids in breast cancer cells. BMB Rep, 2016. 49(3): p. 167–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Huang SX, et al. , The correlation of microRNA-181a and target genes with poor prognosis of glioblastoma patients. Int J Oncol, 2016. 49(1): p. 217–24. [DOI] [PubMed] [Google Scholar]
- 88.Shi ZM, et al. , MiRNA-181b suppresses IGF-1R and functions as a tumor suppressor gene in gliomas. RNA, 2013. 19(4): p. 552–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ayala-Ortega E, et al. , Epigenetic silencing of miR-181c by DNA methylation in glioblastoma cell lines. BMC Cancer, 2016. 16: p. 226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wang XF, et al. , MiR-181d acts as a tumor suppressor in glioma by targeting K-ras and Bcl-2. J Cancer Res Clin Oncol, 2012. 138(4): p. 573–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Shi L, et al. , hsa-mir-181a and hsa-mir-181b function as tumor suppressors in human glioma cells. Brain Res, 2008. 1236: p. 185–93. [DOI] [PubMed] [Google Scholar]
- 92.Wang L, et al. , Kaiso (ZBTB33) Downregulation by Mirna-181a Inhibits Cell Proliferation, Invasion, and the Epithelial-Mesenchymal Transition in Glioma Cells. Cell Physiol Biochem, 2018. 48(3): p. 947–958. [DOI] [PubMed] [Google Scholar]
- 93.DeSantis C, et al. , Breast cancer statistics, 2011. CA Cancer J Clin, 2011. 61(6): p. 409–18. [DOI] [PubMed] [Google Scholar]
- 94.Society, A.C., http://www.cancer.org/Cancer/BreastCancer/ 2011.
- 95.Amirikia KC, et al. , Higher population-based incidence rates of triple-negative breast cancer among young African-American women : Implications for breast cancer screening recommendations. Cancer, 2011. 117(12): p. 2747–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Huo D, et al. , Population differences in breast cancer: survey in indigenous African women reveals over-representation of triple-negative breast cancer. J Clin Oncol, 2009. 27(27): p. 4515–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Stark A, et al. , African ancestry and higher prevalence of triple-negative breast cancer: findings from an international study. Cancer, 2010. 116(21): p. 4926–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Khosravi-Shahi P, Cabezon-Gutierrez L, and Custodio-Cabello S, Metastatic triple negative breast cancer: Optimizing treatment options, new and emerging targeted therapies. Asia Pac J Clin Oncol, 2017. [DOI] [PubMed] [Google Scholar]
- 99.Bassey-Archibong BI, et al. , Kaiso is highly expressed in TNBC tissues of women of African ancestry compared to Caucasian women. Cancer Causes Control, 2017. 28(11): p. 1295–1304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Jones J, et al. , African Americans with pancreatic ductal adenocarcinoma exhibit gender differences in Kaiso expression. Cancer Lett, 2016. 380(2): p. 513–22. [DOI] [PMC free article] [PubMed] [Google Scholar]


