Abstract
Mastitis is the most economically important disease affecting dairy cattle worldwide. Staphylococcus aureus is a highly prevalent cause of mastitis, causing infections ranging from sub-clinical to gangrenous. However, the interaction between the genotype of the infecting strain of S. aureus and the host response remains largely uncharacterised. To better understand the variation in presentation and outcomes of S. aureus-mediated bovine mastitis, we studied the interaction of a panel of mastitis isolates from several prominent bovine-associated lineages with bovine mammary epithelial cells (bMEC) and neutrophils. Significant differences in immune gene expression by infected primary or immortalised bMEC, or their elaboration of neutrophil chemoattractants, were observed and were dependent on the lineage of the infecting strain. Differences were also apparent in the invasiveness of S. aureus strains and their ability to survive killing by neutrophils. Our results demonstrate that a range of immune responses occur, suggesting the importance of S. aureus strain in dictating mastitis disease course. S. aureus lineages may therefore have adopted differing strategies for exploitation of the intramammary niche. Consequently, improved diagnosis of infecting lineage may enable better prognosis for S. aureus mastitis and reduce morbidity and economic loss.
Introduction
Mastitis, commonly due to intramammary infection (IMI), occurs in dairy herds globally. Often requiring antibiotic intervention, it is a burden both to the wellbeing of the animal and the economic output of the herd through a reduction in milk yield, withholding of milk from antibiotic-treated cows and culling of animals in severe cases1. While the implementation of mastitis control programs at farm level has reduced the incidence of IMI in recent decades, it remains a significant problem in dairy production. A recent estimate of the cost of clinical mastitis occurring during the first 30 days of lactation in US herds suggests a loss per case of $444 with long-term indirect costs accounting for the majority of the losses2.
A wide range of microbial species have been reported to cause bovine IMI although a limited number account for the majority of cases. Staphylococcus aureus is among the major pathogens and is the most commonly isolated microorganism from cases of sub-clinical and clinical mastitis in Ireland, accounting for 20–30% of such cases3,4. A model of the economic cost of S. aureus IMI indicated that losses may be as high as €570 per cow infected, substantially higher than for other pathogens5.
IMI control strategies often include monitoring milk for somatic cell count (SCC), which mainly reflects the number of leukocytes in the udder. In the case of S. aureus infections in particular, milk-recording often reveals elevated SCC in animals otherwise lacking in clinical signs of IMI as this pathogen can persist in the intramammary environment. By contrast, infections with Escherichia coli are more typically acute and clinical in nature, clearing within a few days6. The molecular mechanism underlying the species-specific immune response to these important IMI-associated pathogens has been extensively investigated. Infection of bovine mammary epithelial cells (bMEC) with E. coli initiates Toll-like receptor (TLR) signalling, resulting in increased levels of active NF-κB and induction of a strong cytokine response. In contrast, S. aureus infection may fail to activate NF-κB transcription factors resulting in a muted cytokine, and hence SCC, response7,8.
Isolates of S. aureus associated with bovine mastitis predominantly belong to a number of genetically distinct bovine-adapted lineages9. Each lineage encodes a diverse assemblage of regulators and effectors of virulence which could individually or additively influence the host immune response and presentation of mastitis10. While inter-animal variation plays a role in determining S. aureus mastitis susceptibility, S. aureus strains and lineages also differ in their ability to form biofilm11,12, coagulate plasma, produce toxins13, and elicit an immune response from bMEC14,15. Failure to elicit a robust local pro-inflammatory response in bMECs that would result in attraction of immune cells to the site of infection could have important consequences for mastitis presentation and diagnosis.
The major lineages associated with IMI in Ireland are CC71, CC97, ST136 and CC15116. In this study, we examined in vitro the potential for lineage-specific virulence of a panel of strains of S. aureus in their interactions with the MAC-T immortalised bovine mammary epithelial cell line, as well as primary bMEC and neutrophils. These strains were isolated from cases of clinical mastitis in Ireland4 and comprised three isolates from each of the four major lineages. The ability of the strains to induce a pro-inflammatory immune response from bovine mammary epithelial cells resulting in neutrophil chemotaxis, as well as their propensity to invade bMEC, were characterised. We additionally examined survival of the strains during incubation with bovine granulocytes.
Results
Expression of pro-inflammatory immune genes differs by lineage of infecting strain
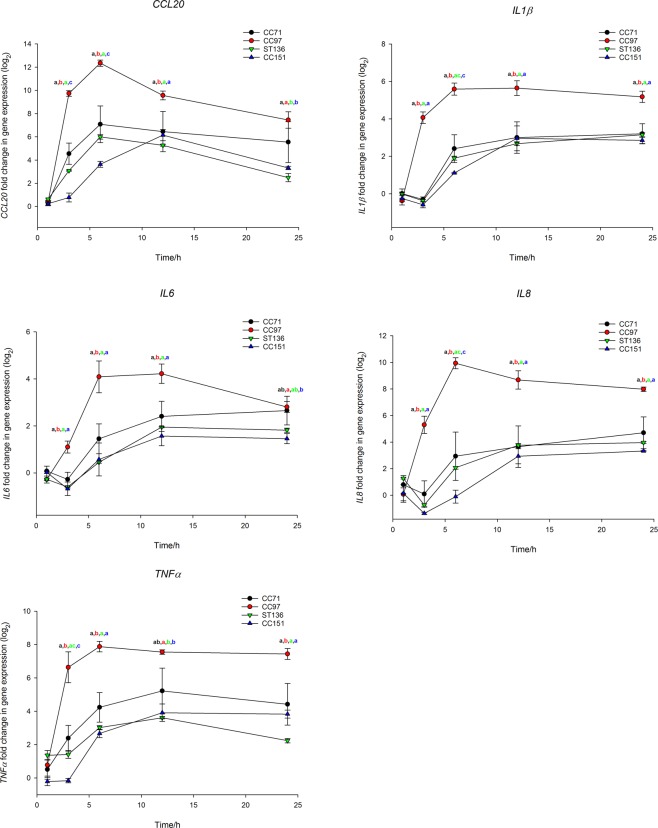
All strains of S. aureus induced CCL20, IL-1β, IL-6, IL-8 and TNFα expression in the MAC-T bovine mammary epithelial cell line (Supplementary File). Expression of TLR2 was not detectable, despite verification that the hydrolysis probe assay could detect the transcript. Expression peaked at 6–12 hours post-infection (hpi) and either remained elevated or declined. There were no significant differences between S. aureus strains in their ability to induce an immune response from MAC-T cells at 1 hpi; however, for all other time points there were significant differences between strains in immune gene induction (Supplementary File). Strains belonging to the same lineage displayed remarkably similar patterns of immune gene induction with the exception of CC71, strains of which were more variable. Strains from CC97 were the strongest inducers resulting in a rapid increase in expression. The largest incitement of inflammatory signalling was mediated by CC97 strain, MOK028, which induced a >7000-fold increase in CCL20 expression at 6 hpi. Moreover, both of the next most highly inducing strains also belonged to CC97. By contrast, ST136 and CC151 strains were relatively poor inducers with CC151 strains also displaying delayed immune gene induction for all genes except IL-6 (Supplementary File). At 1 hpi there were no significant differences between lineages in immune stimulating properties but differences were evident at all other time points; CC97 was the highest immune gene inducer and ST136 and CC151 the poorest (Fig. 1A–E).
Figure 1.
Gene expression in the MAC-T bovine mammary epithelial cell line in response to infection with S. aureus from bovine-adapted lineages. Expression of (A) CCL20, (B) IL1β, (C) IL6, (D) IL8 and (E) TNFα over time in MAC-T cells infected with strains from S. aureus lineages CC71 (black), CC97 (red), ST136 (green) and CC151 (blue). Expression was determined relative to uninfected cells. Data represents mean log2-transformed, normalised expression of 3 individual strains ± SEM. Means which share a superscript are not significantly different.
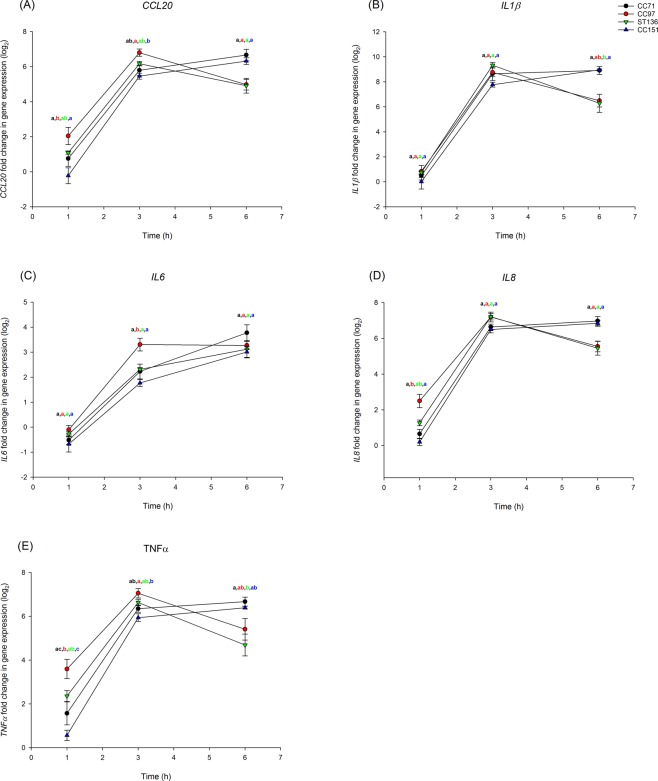
Some differences were evident in the transcriptional response of pbMECs to infection with S. aureus strains but surprisingly these were much less pronounced than for MAC-T cells. Each of the tested genes was up-regulated, with some significant differences between strains in TNFα expression; these differences generally involved strains belonging to CC97 inducing gene expression more highly than those belonging to CC151 (Supplementary File). Again, considering lineages, a trend similar to that seen for MAC-T cells was evident; CC97 induced an earlier and higher pro-inflammatory response than other lineages (Fig. 2).
Figure 2.
Gene expression in primary bovine mammary epithelial cells in response to infection with S. aureus from bovine-adapted lineages. Expression of (A) CCL20, (B) IL1β, (C) IL6, (D) IL8 and (E) TNFα over time in primary bMECs infected with strains from S. aureus lineages CC71 (black), CC97 (red), ST136 (green) and CC151 (blue). Expression was determined relative to uninfected cells. Data represents mean log2-transformed, normalised expression of 3 individual strains ± SEM. Means which share a superscript are not significantly different.
Secretion of cytokines and chemokines differs by lineage of infecting strain
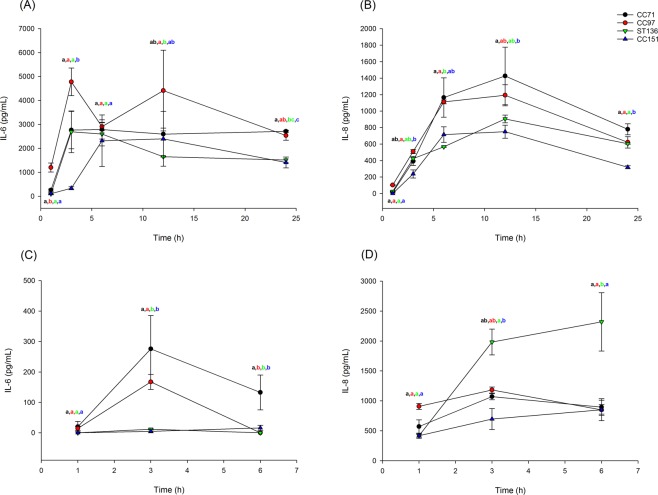
For each strain, the quantity of IL-6 and IL-8 secreted by epithelial cells during infection was determined. Uninfected MAC-T cells produced low levels of IL-6 (<110 pg/ml) at all time points except 24 h and low levels of IL-8 (<100 pg/ml) at all time points. Infection with all strains of S. aureus induced both IL-6 and IL-8 secretion from MAC-T cells (Supplementary File). Per lineage, CC97 induced significantly more IL-6 by 1 hpi than all other lineages and at 3 hpi all lineages induced more IL-6 than CC151. At 24 hpi CC97 and CC71 induced more IL-6 than CC151 (Fig. 3A). By 3 hpi CC97 also induced significantly more IL-8 than CC151 while at 24 hpi, all lineages had induced more IL-8 than CC151 (Fig. 3B).
Figure 3.
Secretion of IL-6 and IL-8 by bovine mammary epithelial cells in response to infection with S. aureus from bovine-adapted lineages. Secretion of IL-6 (A,C) and IL-8 (B,D) from MAC-T cells (A,B), or primary bMEC (C,D) over time in response to infection with strains from S. aureus lineages CC71 (black), CC97 (red), ST136 (green) and CC151 (blue). Data represent mean ± SEM of three strains per lineage. Means which share a superscript are not significantly different.
Uninfected pbMECs did not secrete IL-6 at any time point and secreted low levels of IL-8. Infected cells induced expression of both IL-6 and IL-8 although expression of IL-6 was induced to a much lower level in pbMECs than in MAC-T cells (Supplementary File). There were many significant differences between strains in ability to induce IL-6 secretion while the only significant difference for IL-8 was that MOK076 induced significantly more IL-8 than MOK023 and MOK124 at 6 hpi (Supplementary File). At 3 hpi, CC71 and CC97 induced significantly more IL-6 than CC151 or ST136, while at 6 hpi CC71 induced more IL-6 than the other 3 lineages (Fig. 3C). For IL-8, ST136 induced secretion more highly than CC151 at 3 hpi and more highly than all lineages at 6 hpi (Fig. 3D).
S. aureus lineages induce bMECs to produce chemotactic gradients of differing potency
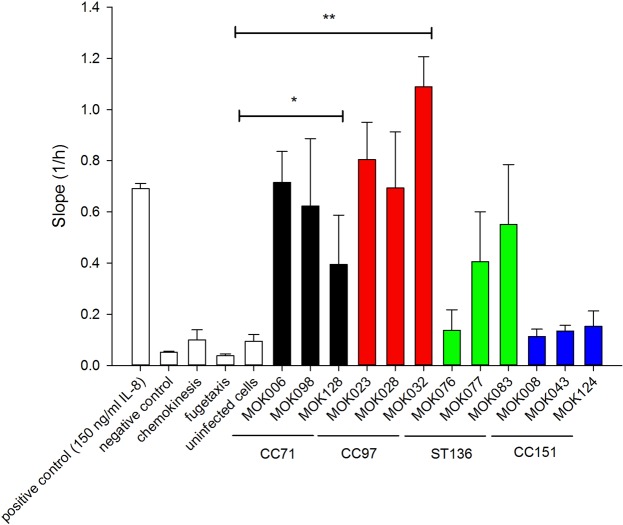
As individual strains, particularly those belonging to CC97, appeared to induce expression of pro-inflammatory mediators more highly than other strains, we sought to establish if this determined the ability of infected bMEC to attract immune cells. Conditioned media from MAC-T cells infected for 24 h with each strain of S. aureus were used as neutrophil chemoattractants in a real-time cell migration assay. All strains belonging to CC97 in addition to strains MOK006 and MOK098 (CC71) and MOK083 (ST136) were able to drive a migratory response that was significantly greater than conditioned medium from uninfected cells. Conditioned media from bMEC infected with CC151 strains displayed a striking lack of ability to stimulate neutrophil chemotaxis (Fig. 4). For the lineages, conditioned media from CC71 and CC97 resulted in significant chemotaxis (P = 0.02 and P = 0.006, respectively) while conditioned media from ST136 and CC151 did not (P = 0.09 and 0.82, respectively).
Figure 4.
Ability of conditioned media from MAC-T cells infected for 24 h with each strain of S. aureus to attract neutrophils. Chemotaxis was measured in real time in a two-chamber cell migration plate by cell impedance on an xCELLigence system (Roche). Positive control included 150 ng/ml IL-8 in the lower chamber; negative control included no IL-8; chemokinesis control included 150 ng/ml IL-8 in both upper and lower chambers; fugetaxis control included 150 ng/ml IL-8 in the upper chamber while uninfected controls included conditioned media from uninfected MAC-T cells in the lower chamber. Data represent the mean rate of migration ± SEM, from triplicate experiments.
Ability of S. aureus strains to survive killing by bovine granulocytes
Each of the S. aureus strains was highly susceptible to the antimicrobial activity of bovine granulocytes following 2 hours of co-culture; survival ranged from 4.2% (MOK076; ST136) to 16.9% (MOK032; CC97) (Fig. 5). There were no significant differences between strains in ability to survive killing by bovine granulocytes (P = 0.1). There were, however, significant differences in survival between lineages, strains belonging to ST136 had significantly poorer survival than CC97 and CC151 (P < 0.05).
Figure 5.
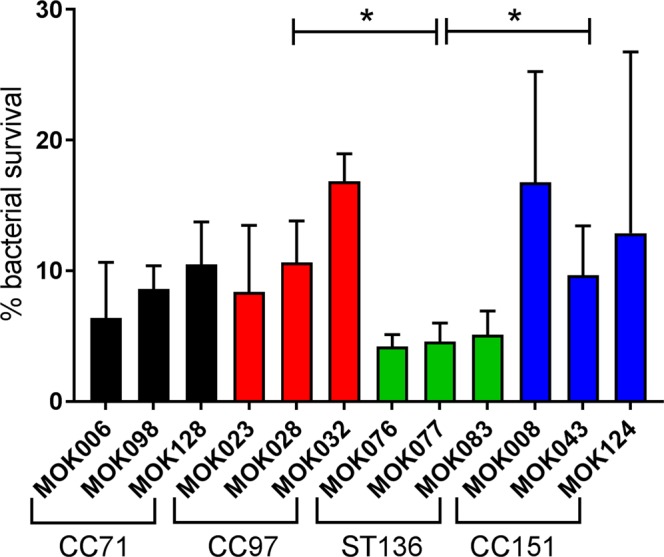
Survival of S. aureus incubated with bovine granulocytes. Percentage of each strain of S. aureus that survived 2 hours of co-culture with bovine granulocytes compared to cells incubated without granulocytes. Data represent the mean ± SD from triplicate experiments.
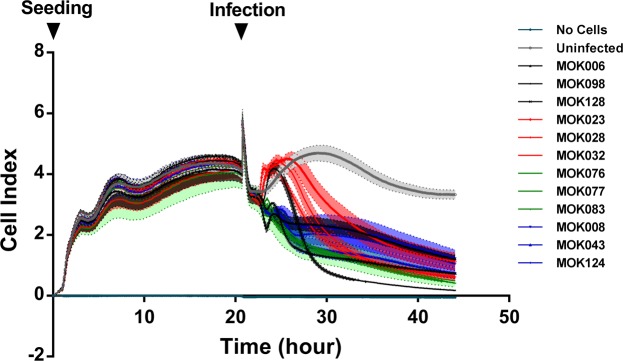
Acute infection causes a lineage-dependent effect on cell behaviour
In order to determine whether the strains differentially affected cell viability, monolayer cultures of MAC-T cells were established in E-plates. Cells were confluent by 20 h of culture. For this cell type, confluence corresponded to a cell index (CI) of ~4. A short-lived disruption in impedance was evident due to plate removal for replacement of media and inoculation of bacteria. For uninfected cells, CI remained steady over the remainder of the experiment. Infection with S. aureus caused a decline in cell index over time, indicating detachment of non-viable cells from the electrodes. Individual strains affected MAC-T cells differently; infection with all strains belonging to CC97 as well as MOK098 (CC71) resulted in a transient rise in cell index followed by a rapid decline while infection with the remaining strains resulted in a prolonged gradual decline in cell index (Fig. 6).
Figure 6.
Real-time measurement of adhesion, morphology and detachment of MAC-T cells pre- and post-infection with each of 12 strains of S. aureus. Cells were cultured to confluence in wells of an E-plate (Roche) over 20 h before addition of each S. aureus strain at an MOI of 10 (arrow indicates time of infection). Cell index, the relative change in electrical impedence and hence a measure of the strength of cell adhesion, was monitored in real-time for the following 24 hours post-infection using an xCELLigence system (Roche). Data represent mean cell index ± SD over time (N = 3 per strain).
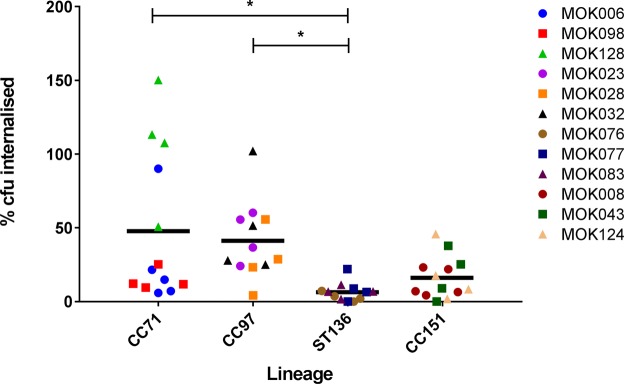
S. aureus strains show differences in their ability to internalise within epithelial cells
Invasion and persistence is a trait common to multiple strains of S. aureus. Hence, we determined whether the strains differed in their ability to invade MAC-T cells. Strain MOK128 (CC71) was significantly more effective at internalising into MAC-T cells than all strains belonging to ST136, while strains MOK023 and MOK032 internalised to a higher extent than MOK076 (P < 0.05) (Fig. 7). There were no significant differences between any other strains. Comparing between lineages, CC97 and CC71 strains internalised to a significantly greater extent than ST136 strains (P < 0.05).
Figure 7.
Percentage of each strain of S. aureus that internalised into MAC-T cells after 2 hours of co-incubation. Percentage internalisation was determined relative to bacterial proliferation in the absence of cells. Data represent mean ± SD, N = 4.
Differences between the lineages in ability to elicit the production of chemotactic factors from bMEC, ability to resist killing by bovine neutrophils and ability to internalise into bMEC are summarised in Table 1.
Table 1.
Summary characteristics of bovine-adapted S. aureus lineages.
| Lineage | Capsule type | bMEC Internalisation | Neutrophil Survival | Recruitment of neutrophils by bMEC conditioned media |
|---|---|---|---|---|
| CC71 | cap8 | 49.4%a | 8.5%ab | 0.55* |
| CC97 | cap5 | 41.3%a | 12.0%a | 0.80* |
| ST136 | cap5 | 6.4%b | 4.6%b | 0.36 |
| CC151 | cap8 | 16.4%ab | 13.1%a | 0.12 |
*Significantly different from uninfected control.
Means which share a superscript are not significantly different.
Discussion
Staphylococcus aureus mastitis is a global problem with substantial economic costs and negative impact on animal health and welfare. Current mastitis control methods such as disease detection, culling of persistently infected animals and genetic selection for mastitis resistance are primarily based on the host response to infection, determined by milk somatic cell count (SCC). Therefore the ability of S. aureus strains to interact in significantly different ways with the host immune system may impact diagnosis, treatment and control of disease. Mammary epithelial cells are one of the major host cell types responsible for initial detection of infecting S. aureus and the severity and outcome of infection is dependent on the ability of this cell type to signal to professional immune cells in a timely manner, initiating rapid and substantial neutrophil recruitment17. In this study we characterise the bMEC response to infection for 24 h with live S. aureus from different bovine-adapted lineages for the first time. We demonstrate in vitro an interaction between bMEC response and the genotype of S. aureus and demonstrate that this has downstream consequences for immune signalling.
Each of the examined S. aureus strains induced expression of immune signalling cytokines and chemokines in the bMEC cell line MAC-T. Substantial up-regulation occurred within 3 hpi, suggesting a robust, acute response by the cells to S. aureus. CC97 strains proved highly immunogenic, while strains from other lineages, most notably CC151 and ST136, were much less immune-stimulatory. Notably, gene induction was similar among strains that were of a common lineage, but differed significantly between those of different lineages. Strains of CC151 also appeared to display a somewhat delayed immune gene induction compared to other strains. This suggests that some lineages are more immunogenic than others and/or some may be immunosuppressive. A relative failure of the immune system to properly detect S. aureus compared to E. coli has been previously reported7,8,18–21; albeit in studies comparing heat-killed isolates, which may not fully reflect the dynamic interaction between host cells and live bacteria. These studies demonstrated that S. aureus was weakly agonistic for TLR2 and failed to induce NF-κB and its downstream effectors in pbMECs7,8,19. It was recently shown in vivo that this failure underpins the differential presentation of mastitis caused by these species with S. aureus being actively immune evasive through NF-κB suppression. In the present study, expression of TLR2 was not detected in response to any strain and so other effectors may be responsible for the observed differences in immunogenicity. Lack of TLR2 stimulation in human cells transfected with bovine TLR2 has been previously observed in response to other Gram positive bacteria which cause IMI such as S. uberis and S. agalactiae21. The expression of capsular polysaccharide by S. aureus has also been reported to interfere with bacterial recognition via TLR2 possibly by masking TLR2-active lipoproteins22.
In support of our observations regarding gene expression, we assayed the secretion by the cells of IL-6 and IL-8. IL-6 is a cytokine required for Th17 cell differentiation, a cell type that releases IL-17 instigating a signalling cascade that results in recruitment and activation of neutrophils23. IL-8 is a key chemokine required for neutrophil recruitment and both cytokines have previously been shown to be central to the mammary epithelial response to S. aureus mastitis8. Infected MAC-T cells secreted IL-6 and IL-8 over 24 hours of infection to different extents depending on the infecting strain, in agreement with the differential gene expression of IL-6 that we observed. Although both gene and protein expression assays generally found greatest cytokine production to be stimulated by CC97 or CC71 strains with a lesser pro-inflammatory response to CC151 strains, the concordance between mRNA expression and that of the cognate protein was not absolute. Such discrepancies are frequently reported and a variety of explanations are possible including mRNA stability, translational regulation and protein turnover. In this instance the reason for any discrepancies is unknown but importantly functional neutrophil chemotaxis assays confirmed that infection with strains from CC97 resulted in the strongest neutrophil recruitment while infection with strains from CC151 failed to result in neutrophil recruitment.
It has previously been shown for S. aureus strains that the magnitude of induction of IL-1β, IL-8 and TNFα from bMEC varied according to strain24. In the aforementioned study, however, extracellular S. aureus were killed at 3 hpi and gene expression returned to basal levels by 24 hpi, while in our study gene expression remained elevated (MAC-T) or infection resulted in cell death (pbMEC)24. Elsewhere, heat-inactivated S. aureus strains were shown to elicit differing immune responses from pbMECs15, with the more pro-inflammatory strains belonging to lineages CC8, CC20 and CC97, and the less agonistic strains all belonging to CC151, thus supporting our finding of relatively limited immune signalling mediated by this lineage. These studies support our finding that different strains of S. aureus can induce immune gene expression to differing extents and that this difference is likely mediated through lineage-specific factors. In many studies, only single strains of S. aureus are utilised to investigate the response of the bovine mammary epithelium to challenge with S. aureus8,18,19. Hence, these studies lack information about the potential for differing strains of S. aureus to present with differing immunologic profiles in the mammary gland. Limiting the study of S. aureus mastitis to one or a few closely related strains may overlook the range of immunogenicity of which the species is capable.
Variation in the immune response of pbMEC to S. aureus strains was also evident, albeit with somewhat lesser magnitude of expression and more moderate differences between lineages. This was surprising as it has been previously reported that while MAC-T cells faithfully reproduce key aspects of the parental cell immune response, their response tends to be weaker25. It is not clear why, in our hands, the pbMEC response was not completely concordant with that of MAC-T but it may reflect individual variability between MEC donors, a loss of response of our cells due to freezing and passage or the fact that infection with live bacteria (compared to heat-killed in25), which led to death of the infected monolayers, affected gene expression. Alternatively, as MAC-T and pbMEC were each cultured in a cell-specific medium this may have impacted gene expression; the composition of culture medium has been previously reported to modulate gene expression in S. aureus-infected MEC26.
Differences between the lineages in host cell interactions were further evident in their effect on cell morphology and viability. Infection of MAC-T cells with all CC97 strains, as well as with one CC71 strain, resulted in a transient increase in cell index (CI) during the initial phase post-infection. This was in contrast to the immediate decline in CI caused by the remaining S. aureus strains. There were also slight differences between strains in the rate of detachment, and some cells appeared detached by 24hpi. This indicates that the 24 hpi cytokine induction and secretion are caused by a smaller subset of cells, and also that some differences observed at this time point are caused by cell death processes. However, further studies are necessary to examine the viability of bMEC when infected with the different strains. The transient increase in CI most likely reflects a change in cellular morphology, such as cellular swelling mediated by pyroptosis pursuant to inflammatory signalling, or necrosis, in response to a factor expressed by these strains27,28.
S. aureus can internalise within host cells, including epithelial cells and phagocytes29–33, and can be found within epithelial cells and macrophages isolated from cows experiencing IMI34, or in the interstitial tissue particularly during chronic infection35. Here they may evade the immune system, establishing a reservoir from which to re-establish infection. A S. aureus strain with a reduced capacity for mammary epithelial cell internalization in vitro was also shown to have delayed mammary epithelial cell internalisation in vivo in a murine mastitis model36. In the present study a high degree of variability in the extent of the strains’ internalisation was evident not only between strains, but also for the strains themselves. With the exception that ST136 strains were consistently poorly internalised, within strain and lineage variation was large. This is consistent with a previous report of large variation in the ability of bovine S. aureus isolates to invade MAC-T cells37. Nevertheless, we observed the minimum internalisation of our strains to be 4.3 ± 2.6%, in the case of the ST136 strain MOK076. By contrast, some studies have reported invasion by geographically diverse mastitis isolates of S. aureus into MAC-T or pbMEC cells below 0.01% of initial inoculum, even when a high bacterial titre (109 cfu) was used33,38. It has been shown that the well-characterised bovine isolates, RF122 (CC151) and Newbould 305 (CC97), internalise into MAC-T cells up to ~1% and ~15% of their respective initial titres when added in a 100:1 MOI to the cells39. Thus, our findings support the previously reported differences in the proficiency of various strains to internalise into bMECs.
Neutrophils are the predominant infiltrating cells during the response to S. aureus intramammary infection, accounting for over 80% of milk somatic cells during acute infection40 and 66% of somatic cells in sub-clinical infection41, remaining dominant during persistent infection42. Importantly, as well as demonstrating that infection with CC97 strains resulted in upregulation of immune gene and protein expression while CC151 strains caused a delayed response of lower magnitude, we also demonstrated that this upregulation had a functional effect on neutrophil recruitment. Hence, the infecting strain determined the ability of bMEC to signal to professional immune cells. A delayed influx of neutrophils into the mammary gland negatively affects infection resolution, allowing the infection to become established which can lead to invasive disease. Therefore the delayed and muted immune response initiated by CC151 strains may be a strategy to allow their establishment in the intramammary environment. A number of epidemiological studies have examined the relationship between S. aureus genotype and infection characteristics although a more limited number have characterised the isolates by clonal complex; among these studies it has been reported that S. aureus transmission and cure depend on lineage43–46. The majority of in vivo S. aureus-challenge IMI studies have been conducted with strains belonging to ST8 (CC8), ST133 (CC133) or ST115 (CC97; the laboratory strain Newbould 305)35,47–49 and in some instances where a local isolate has been used the infecting strain genotype has been omitted50,51. The relevance of using Newbould 305 to study S. aureus-mediated bovine mastitis has been questioned on the basis of its mild induction of host response and the limited distribution of ST115 strains52. Interestingly, although it has been reported that experimental infection with S. aureus strain RF122 (CC151) causes a severe mastitis while Newbould 305 (CC97) causes mild mastitis13, RF122 has not been widely studied in vivo. A pilot study by Wilson53 described infection of 3 cows with RF122. In all instances infection was established and acute clinical mastitis was observed with an inoculum as low as 2 cfu. A further study by the same group reported clinical mastitis in 3 out of 4 cows infected with RF12254.
The ability of neutrophils to phagocytose and kill invading S. aureus is vital for infection resolution. In turn, some strains of S. aureus are capable of producing pore-forming leukocidins and high expression of the bovine LukMFʹ leukocidin has been associated with severe mastitis55. Carriage of the lukMFʹ genes is lineage-dependent and among our strains only CC151 strains encoded both genes required to form the two-component toxin16. Lineages CC97 and CC151 were more resistant to killing than ST136 but there were no significant differences between individual strains, suggesting that expression of lukMFʹ may not have been switched on under the assay conditions. Neutrophils recruited to the mammary gland show an immature phenotype, and their enzymatic activity is perhaps 10% that of blood-derived counterparts56 with milk neutrophils showing reduced capacity to kill S. aureus compared to blood derived neutrophils57. As such, the bacteria may have greater opportunity to survive in the udder, where strain-specific differences may be evident.
In the present study, the marked difference in the immunogenic character of the strains may be due to their lineage-dependent carriage of virulence traits including adhesins and toxins16. The CC97 strains harbour multiple Microbial Surface Components Recognising Adhesive Matrix Molecules (MSCRAMMs) and subsequently may be invasive and capable of producing persistent subclinical mastitis in vivo. Conversely, the CC151 strains do not encode a number of MSCRAMMs including fibronectin binding protein B (fnbB) and may therefore fail to readily adhere and internalise58. In this context, the expression of LukMFʹ by CC151 strains may dispose them to cause acute clinical mastitis59,60. Further to this, the CC151 strains are agr type II and cap8, a genetic profile suggested to be adapted to the extracellular niche37. CC151 isolates are also poor biofilm formers relative to CC97 isolates14. This could bring CC151 strains in direct contact with the host immune response; biofilm formation being associated with immune evasion and persistent intramammary infection11. Differences between strains within a lineage were expected, as the accessory genome of each differs to some extent. The redundancy of MSCRAMMs and toxins within the global S. aureus pangenome perhaps explains why no strain was entirely deficient in either invasion of MAC-T cells or induction of immune response genes. While it is compelling to consider that, in order to persist sub-clinically in the udder, S. aureus must be minimally immunogenic, this need not be the case if the bacteria survive intracellularly.
Greater variability was observed among CC71 strains than among strains of the other lineages in ability to internalise, elicit an immune response and stimulate neutrophil chemotaxis. Some CC71 strains also behaved similarly to CC97 strains in their ability to elicit expression of chemotactic factors and their effect on bMEC morphology. CC71 is a recently defined lineage which evolved from CC97 by multiple recombination events in an approximately 300 kb region spanning the origin of replication61,62. In this study, strains belonging to CC71 were of two different sequence types, ST71 and ST3173, which may explain in part the differences in host cell interaction observed. A previous study also demonstrated phenotypic variability between ST71 strains62. Therefore diversity of strains within the CC71 lineage may be greater than within other lineages, which warrants further examination.
Using a panel of S. aureus strains of known bovine-associated lineage, we have shown that strain interaction with bovine mammary epithelial cells and neutrophils varies according to bacterial genotype. The differences in bMEC interaction and bacterial survival indicate that each S. aureus lineage has a unique set of characteristics that may determine the outcome of infection in vivo. Further studies are required on the differences between strains in their ability to cause intramammary infection in cattle. This work also emphasises that study of single strains, even model-specific isolates, can misrepresent the breadth of possible outcomes of infection across a bacterial species. Associating the specificity in virulence with lineage and strain in future studies would enable more effective comparison of general trends in S. aureus virulence.
Methods
Bacteriological Culture
The strains were originally isolated each from a different cow presenting with clinical mastitis; in total 6 farms are represented. Isolation and characterisation of the strains has been previously described4,16. Bacteria were cultured until stationary phase in tryptic soy broth (TSB; BD Bioscience) at 37 °C, 220 rpm. Cultures were suspended in phosphate buffered saline (PBS; Gibco) to an optical density (OD600nm) of 1 prior to dilution in appropriate culture medium. Colony forming units were enumerated by manual count following serial dilution in PBS, spreading onto tryptic soy agar and overnight incubation at 37 °C. The S. aureus strains used in the study are described in Table 2. The genome of each strain has been sequenced and deposited in the NCBI Sequence Read Archive; accession numbers are listed in Table 2.
Table 2.
Lineage and Sequence Type of S. aureus strains used during this study.
| Strain† | Lineage | Sequence Type | Genome Accession Number |
|---|---|---|---|
| MOK006 | CC71 | ST3173 | SRS2841719 |
| MOK098 | CC71 | ST71 | SRS2841716 |
| MOK128 | CC71 | ST71 | SRS2841712 |
| MOK023 | CC97 | ST3170 | SRS775827 |
| MOK028 | CC97 | ST3221 | SRS2841721 |
| MOK032 | CC97 | ST3170 | SRS775824 |
| MOK076 | ST136 | ST136 | SRS2841714 |
| MOK077 | ST136 | ST136 | SRS2841715 |
| MOK083 | ST136 | ST136 | SRS2841717 |
| MOK008 | CC151 | ST151 | SRS2841718 |
| MOK043 | CC151 | ST151 | SRS2841720 |
| MOK124 | CC151 | ST151 | SRS2841713 |
†Full characterisation of strains in16.
Cell Culture
MAC-T cells, a bovine mammary epithelial cell line derived from a lactating Holstein cow63 were recovered from −150 °C (passage 9–10) and cultured in Dulbeccos’ Modified Eagle medium (DMEM; Sigma) with 10% foetal bovine serum (FBS; Sigma) at 37 °C, 5% CO2. Primary bovine mammary epithelial cells (pbMECs; AvantiCell Ltd) were derived from the udder parenchyma of a cow during the third trimester of pregnancy. Primary cells were cultured as described previously64. All cells were used within six passages following revival from storage at −150 °C.
Cell Infections and RNA Extraction
MAC-T cells were seeded into 12-well plates at 2 × 105 cells/well in 1 ml DMEM, 10% FBS. Primary cells were seeded into EHS matrix-coated 12-well plates at 8 × 105 cells/well in 1 ml of attachment-differentiation medium, replaced with serum-free lactogenic medium after 24 h. Cells were incubated at 37 °C, 5% CO2 until confluent. Monolayers were washed twice with pre-warmed infection medium (DMEM with 1% FBS for MAC-T; serum-free lactogenic medium for pbMECs) before inoculation with bacteria at a multiplicity of infection (MOI) of 10. Uninoculated wells served as uninfected controls. Following 1, 3, 6, 12 or 24 h of incubation at 37 °C, 5% CO2, medium was removed, clarified by centrifugation and stored at −20 °C. The viability of S. aureus infected MAC-T at 24 h post-infection was 80–90% using trypan blue exclusion and annexin V/PI staining while pbMEC were non-viable by 12 h post-infection. Wells were washed with 1 ml pre-warmed PBS and RNA was extracted using RNeasy kit (Qiagen) and stored at −80 °C.
Gene expression analysis
RNA was treated to remove residual DNA using the Turbo DNA-free kit (Ambion) and quantified using the RNA broad range assay kit (Invitrogen). RNA quality was verified with a 2100 bioanalyser (Agilent). Synthesis of cDNA was conducted on MAC-T samples having RIN values exceeding 7 (mean 9.6) and on primary cell samples having RIN values exceeding 5.4 (mean 8.4). RNA from each sample (500 ng) was reverse transcribed using the Superscript VILO kit (Invitrogen).
For each sample, cDNA (1 µl) was added to duplicate wells with 7 µl of DNase-free water, 10 µl of Taqman Fast Advanced master mix (Applied Biosystems) and 1 µl of each of 2 Taqman gene expression assays (Applied Biosystems) (Table 3). Lack of interference between assays was confirmed experimentally. Samples were incubated at 50 °C for 2 min followed by 95 °C for 20 s then cycled 40 times at 95 °C for 3 s and 60 °C for 30 s in a 7500 Fast thermocycler (Applied Biosystems).
Table 3.
Taqman assays used for analysis of gene expression in S. aureus-infected bovine mammary epithelial cells.
| Gene | Gene Accession | Taqman® Assay ID | Fluorophore | Amplicon Length |
|---|---|---|---|---|
| IL-6 | NM_173923.2 | Bt03211903_m1 | FAM | 69 |
| CCL20 | NM_174263.2 | Bt03223359_m1 | FAM | 73 |
| TLR2 | NM_174197.2 | Bt03223212_m1 | VIC | 109 |
| IL-8 | NM_173925.2 | Bt03211907_g1 | FAM | 105 |
| IL-1β | NM_174093.1 | Bt03212744_m1 | FAM | 69 |
| TNFα | NM_173966.3 | Bt03259155_g1 | VIC | 66 |
| ACTB | NM_173979.3 | Bt03279175_g1 | VIC | 144 |
| RPS24 | NM_001025339.2 | Bt03220533_g1 | VIC | 87 |
Target gene Cq values were normalised against the reference genes ACTB and RPS24, selected from 9 candidate genes whose stable expression in both MAC-T cells and pbMECs that were infected with S. aureus was evaluated using GeNorm, NormFinder and BestKeeper. The efficiency of all PCR reactions was between 98–102% (data not shown). Fold change in gene expression level was calculated by comparing to Cq values from uninfected cells at the same time point.
Quantification of secreted proteins by ELISA
For quantification of cytokine secretion, 100 µl of supernatants recovered post-infection was assayed by sandwich ELISA for the presence of IL-6 (ThermoFisher) and IL-865, per manufacturers’ instructions. All samples were assayed in duplicate and values below the limit of detection (39 pg/mL for IL-6 and 31 pg/mL for IL-8) were set to zero.
Neutrophil-mediated killing of bacteria
Peripheral blood was collected from four disease-free cattle. Serum was collected from the same four animals and pooled. Erythrocytes were lysed by suspension of blood for 10 min in four volumes of high-yield lyse solution (Invitrogen) at ambient temperature. Leukocytes were isolated by centrifugation at 300 × g for 10 min and washed in sterile PBS. Granulocytes were purified by magnetic-activated cell sorting, using mouse anti-bovine granulocyte IgG1 (clone CH138A Kingfisher Biotech Inc) and magnetic anti-mouse IgG1 microbeads (Miltenyi Biotec), following the manufacturers’ protocol (Miltenyi). Estimation of purity and enumeration of granulocytes was performed on an Attune flow cytometer (Applied Biosystems). Granulocytes were centrifuged for 10 min at 300 × g and re-suspended to 1 × 107 cells/ml in RPMI.
Fresh bacterial colonies were suspended in sterile PBS to an OD600nm of 0.45 and enumerated as described above. Each strain (7 × 106 cfu in 50 µl) was suspended in the presence or absence of 2.5 × 106 granulocytes (MOI ~3) to a total volume of 500 µl including 10% autologous bovine serum and incubated at 37 °C for 2 h, rotating end-over-end. Ultrapure water was then added directly to lyse granulocytes and suspensions were serially diluted in sterile PBS. Dilutions (50 µl) were spread onto triplicate TSA plates for enumeration of bacteria. The experiment was conducted in triplicate for each strain.
Real-time cell adhesion and morphology during infection
Wells of a 16-well E-plate (Roche Diagnostics GmBH) were equilibrated for 30 min at ambient temperature with 100 µl DMEM before adding 1 × 104 MAC-T cells/well in an additional 100 µl DMEM. One well per plate received DMEM only to control for background impedance. Cells were allowed to settle for 30 min before plates were inserted into an xCELLigence RTCA DP Analyser (ACEA Biosciences Inc.) at 37 °C, 5% CO2. Impedance was measured for 20 h. E-plate wells were confirmed visually to be confluent with cells. Medium was removed and each strain was added to a discrete well at an MOI of 10 in 200 µl DMEM. Bacteria were allowed to settle for 30 min and E-plates were returned to the instrument at 37 °C, 5% CO2 for a further 24 h. The experiment was conducted in triplicate for each strain.
Cell Invasion Assay
MAC-T cells were infected for 2 h with S. aureus as described above. Wells were washed with PBS and 1 ml of 100 µg/ml gentamicin in DMEM plus 1% FBS was added. Plates were incubated for a further 1 h then washed with PBS. Cells were lysed for 10 min in 1 ml 1% Triton X-100, 0.1% SDS, followed by repeated pipetting. Lysates were serially diluted in PBS and plated (50 µl) to enumerate internalised bacteria. The inoculum was similarly incubated for 2 h before dilution and plating. The experiment was conducted in quadruplicate for each strain.
Chemotaxis Assay
The chemotaxis assay was carried out as described elsewhere66. Briefly, cell invasion and migration plates (CIM-Plate 16; Roche) were coated with 20 μg/ml fibronectin (Sigma) in PBS at 37 °C, 30 min, then washed twice with 50 μl PBS. The lower chambers were filled with 160 µl of supernatants from MAC-T cells infected with the relevant strain of S. aureus for 24 h. Upper chambers received 35 µl DMEM with 1% FBS. After 1 h at room temperature, background impedence was obtained at 37 °C, 5% CO2. To the top chamber of each well, 100 µl of neutrophils (1.2 × 107 cells/ml in DMEM with 1% FBS) was added and impedence measured every 5 min for 7 h. Several controls were included: (i) positive control: neutrophils in upper chamber, 150 ng/ml bovine IL-8 (Kingfisher Biotech Inc) in lower chamber (ii) negative control: neutrophils in upper chamber, 0 ng/ml IL-8 in lower chamber (iii) chemokinesis: neutrophils in upper chamber, 150 ng/ml IL-8 in upper and lower chambers (iv) fugetaxis: neutrophils and 150 ng/ml IL-8 in upper chamber, medium only in lower chamber; (v) uninfected cells: neutrophils in upper chamber, medium from cells uninfected with S. aureus in lower chamber. The slope of the exponential part of the curve (1 h) was calculated using RTCA software 1.2.1.
Statistical Analysis
Models were fitted and residuals checked to ensure the assumptions of the analysis (normality and constant variance) were met; log transformation was used as appropriate. Transformations were applied to ELISA measurements (sqrt(x)), internalisation measurements (log10(x + 1)) and chemotaxis measurements (log10(x*100 + 1). For between strain analyses, data were analysed using Proc GLIMMIX (SAS v9.3); for two-factor experiments, a random time statement with a residual qualifier and unstructured covariance matrix accounted for repeated measures over time. For the between lineage analysis, data were also analysed using Proc GLIMMIX, with strain as a random term. Tukeys’ post-hoc multiple comparison test or Dunnetts’ comparison to a control were applied as appropriate. P values < 0.05 were considered significant.
Ethical approval
All procedures involving animals were conducted under ethical approval and experimental license from the Irish Health Products Regulatory Authority and performed according to European regulations regarding animal welfare and protection of animals used for experimental and other scientific purposes.
Supplementary information
Acknowledgements
The authors gratefully acknowledge the technical assistance of Margaret Murray and Ciara Carberry (Teagasc) and Jim Grant (Teagasc) for statistical advice. This study was funded by the Department of Agriculture, Food and the Marine, grant number 14/S/802.
Author Contributions
M.P.M. and D.A.N. performed the experiments. F.C.L. and O.M.K. were responsible for study conception. M.P.M., D.A.N., F.C.L. and O.M.K. analysed the data and wrote the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mark P. Murphy and Dagmara A. Niedziela contributed equally.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-42424-2.
References
- 1.Hogeveen H, Huijps K, Lam TJ. Economic aspects of mastitis: new developments. N. Z. Vet. J. 2011;59:16–23. doi: 10.1080/00480169.2011.547165. [DOI] [PubMed] [Google Scholar]
- 2.Rollin E, Dhuyvetter KC, Overton MW. The cost of clinical mastitis in the first 30 days of lactation: An economic modeling tool. Prev. Vet. Med. 2015;122:257–264. doi: 10.1016/j.prevetmed.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 3.Barrett DJ, Healy AM, Leonard FC, Doherty ML. Prevalence of pathogens causing subclinical mastitis in 15 dairy herds in the Republic of Ireland. Ir. Vet. J. 2005;58:333–337. doi: 10.1186/2046-0481-58-6-333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Keane OM, Budd KE, Flynn J, McCoy F. Pathogen profile of clinical mastitis in Irish milk-recording herds reveals a complex aetiology. Vet. Rec. 2013;173:17–21. doi: 10.1136/vr.101308. [DOI] [PubMed] [Google Scholar]
- 5.Sørensen LP, Mark T, Sørensen MK, Østergaard S. Economic values and expected effect of selection index for pathogen-specific mastitis under Danish conditions. J. Dairy Sci. 2010;93:358–369. doi: 10.3168/jds.2009-2506. [DOI] [PubMed] [Google Scholar]
- 6.Schukken YH, et al. Host-response patterns of intramammary infections in dairy cows. Vet. Immunol. Immunopathol. 2011;144:270–289. doi: 10.1016/j.vetimm.2011.08.022. [DOI] [PubMed] [Google Scholar]
- 7.Yang W, et al. Bovine TLR2 and TLR4 properly transduce signals from Staphylococcus aureus and E. coli, but S. aureus fails to both activate NF-κB in mammary epithelial cells and to quickly induce TNFα and interleukin-8 (CXCL8) expression in the udder. Mol. Immunol. 2008;45:1385–1397. doi: 10.1016/j.molimm.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 8.Günther J, et al. Comparative kinetics of Escherichia coli- and Staphylococcus aureus-specific activation of key immune pathways in mammary epithelial cells demonstrates that S. aureus elicits a delayed response dominated by interleukin-6 (IL-6) but not by IL-1A or tumor necrosis factor alpha. Infect. Immun. 2011;79:695–707. doi: 10.1128/IAI.01071-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sung JM, Lloyd DH, Lindsay JA. Staphylococcus aureus host specificity: comparative genomics of human versus animal isolates by multi-strain microarray. Microbiology. 2008;154:1949–1959. doi: 10.1099/mic.0.2007/015289-0. [DOI] [PubMed] [Google Scholar]
- 10.Foster, T. J. & Geoghegan, J. A. In Molecular Medical Microbiology (Second Edition) (ed. Yi-Wei TangMax SussmanDongyou LiuIan PoxtonJoseph Schwartzman) 655–674 (Academic Press, 2015).
- 11.Cucarella C, et al. Role of biofilm-associated protein bap in the pathogenesis of bovine Staphylococcus aureus. Infection and immunity. 2004;72:2177–2185. doi: 10.1128/IAI.72.4.2177-2185.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bardiau M, et al. Existence of two groups of Staphylococcus aureus strains isolated from bovine mastitis based on biofilm formation, intracellular survival, capsular profile and agr-typing. Vet. Microbiol. 2016;185:1–6. doi: 10.1016/j.vetmic.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 13.Peton V, et al. Fine-tuned characterization of Staphylococcus aureus Newbould 305, a strain associated with mild and chronic mastitis in bovines. Veterinary research. 2014;45:106. doi: 10.1186/s13567-014-0106-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Budd KE, Mitchell J, Keane OM. Lineage associated expression of virulence traits in bovine-adapted Staphylococcus aureus. Vet. Microbiol. 2016;189:24–31. doi: 10.1016/j.vetmic.2016.04.013. [DOI] [PubMed] [Google Scholar]
- 15.Zbinden Christina, Stephan Roger, Johler Sophia, Borel Nicole, Bünter Julia, Bruckmaier Rupert M., Wellnitz Olga. The Inflammatory Response of Primary Bovine Mammary Epithelial Cells to Staphylococcus aureus Strains Is Linked to the Bacterial Phenotype. PLoS ONE. 2014;9(1):e87374. doi: 10.1371/journal.pone.0087374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Budd KE, et al. Extensive Genomic Diversity among Bovine-Adapted Staphylococcus aureus: Evidence for a Genomic Rearrangement within CC97. PLoS One. 2015;10:e0134592. doi: 10.1371/journal.pone.0134592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rainard P, Riollet CL. Mobilization of neutrophils and defense of the bovine mammary gland. Reprod. Nutr. Dev. 2003;43:439–457. doi: 10.1051/rnd:2003031. [DOI] [PubMed] [Google Scholar]
- 18.Griesbeck-Zilch B, Meyer HHD, Kühn C, Schwerin M, Wellnitz O. Staphylococcus aureus and Escherichia coli cause deviating expression profiles of cytokines and lactoferrin messenger ribonucleic acid in mammary epithelial cells. J. Dairy Sci. 2008;91:2215–2224. doi: 10.3168/jds.2007-0752. [DOI] [PubMed] [Google Scholar]
- 19.Fu Y, et al. Staphylococcus aureus and Escherichia coli elicit different innate immune responses from bovine mammary epithelial cells. Vet. Immunol. Immunopathol. 2013;155:245–252. doi: 10.1016/j.vetimm.2013.08.003. [DOI] [PubMed] [Google Scholar]
- 20.Ibeagha-Awemu EM, Ibeagha AE, Messier S, Zhao X. Proteomics, genomics, and pathway analyses of Escherichia coli and Staphylococcus aureus infected milk whey reveal molecular pathways and networks involved in mastitis. J. Proteome Res. 2010;9:4604–4619. doi: 10.1021/pr100336e. [DOI] [PubMed] [Google Scholar]
- 21.Farhat K, et al. The response of HEK293 cells transfected with bovine TLR2 to established pathogen-associated molecular patterns and to bacteria causing mastitis in cattle. Veterinary immunology and immunopathology. 2008;125:326–336. doi: 10.1016/j.vetimm.2008.05.026. [DOI] [PubMed] [Google Scholar]
- 22.Hilmi D, et al. Heterogeneity of host TLR2 stimulation by Staphylocoocus aureus isolates. PloS one. 2014;9:e96416. doi: 10.1371/journal.pone.0096416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bettelli E, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 24.Lahouassa H, Moussay E, Rainard P, Riollet C. Differential cytokine and chemokine responses of bovine mammary epithelial cells to Staphylococcus aureus and Escherichia coli. Cytokine. 2007;38:12–21. doi: 10.1016/j.cyto.2007.04.006. [DOI] [PubMed] [Google Scholar]
- 25.Günther J, Koy M, Berthold A, Schuberth H-J, Seyfert H-M. Comparison of the pathogen species-specific immune response in udder derived cell types and their models. Vet. Res. 2016;47:22. doi: 10.1186/s13567-016-0307-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bauer, I., Gunther, J., Wheeler, T. T., Engelmann, S. & Seyfert, H.-M. Extracellular milieu grossly alters pathogen-specific immune response of mammary epithelial cells. BMC Vet. Res. 11 (2015). [DOI] [PMC free article] [PubMed]
- 27.Fink SL, Cookson BT. Caspase-1-dependent pore formation during pyroptosis leads to osmotic lysis of infected host macrophages. Cell. Microbiol. 2006;8:1812–1825. doi: 10.1111/j.1462-5822.2006.00751.x. [DOI] [PubMed] [Google Scholar]
- 28.Soong G, Chun J, Parker D, Prince A. Staphylococcus aureus activation of caspase 1/calpain signaling mediates invasion through human keratinocytes. J. Infect. Dis. 2012;205:1571–1579. doi: 10.1093/infdis/jis244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Anaya-López JL, et al. Invasive potential of bacterial isolates associated with subclinical bovine mastitis. Res. Vet. Sci. 2006;81:358–361. doi: 10.1016/j.rvsc.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 30.Gresham HD, et al. Survival of Staphylococcus aureus inside neutrophils contributes to infection. J. Immunol. 2000;164:3713–3722. doi: 10.4049/jimmunol.164.7.3713. [DOI] [PubMed] [Google Scholar]
- 31.Kubica M, et al. A potential new pathway for Staphylococcus aureus dissemination: The silent survival of S. aureus phagocytosed by human monocyte-derived macrophages. PLoS One. 2008;3:e1409. doi: 10.1371/journal.pone.0001409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tuchscherr L, et al. Staphylococcus aureus phenotype switching: an effective bacterial strategy to escape host immune response and establish a chronic infection. EMBO Mol. Med. 2011;3:129–141. doi: 10.1002/emmm.201000115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Almeida RA, Matthews KR, Cifrian E, Guidry AJ, Oliver SP. Staphylococcus aureus Invasion of Bovine Mammary Epithelial Cells. J. Dairy Sci. 1996;79:1021–1026. doi: 10.3168/jds.S0022-0302(96)76454-8. [DOI] [PubMed] [Google Scholar]
- 34.Hebert A, Sayasith K, Senechal S, Dubreuil P, Lagace J. Demonstration of intracellular Staphylococcus aureus in bovine mastitis alveolar cells and macrophages isolated from naturally infected cow milk. FEMS microbiology letters. 2000;193:57–62. doi: 10.1016/S0378-1097(00)00455-9. [DOI] [PubMed] [Google Scholar]
- 35.Hensen SM, Pavicic MJ, Lohuis JA, de Hoog JA, Poutrel B. Location of Staphylococcus aureus within the experimentally infected bovine udder and the expression of capsular polysaccharide type 5 in situ. Journal of dairy science. 2000;83:1966–1975. doi: 10.3168/jds.S0022-0302(00)75073-9. [DOI] [PubMed] [Google Scholar]
- 36.Brouillette E, Grondin G, Shkreta L, Lacasse P, Talbot BG. In vivo and in vitro demonstration that Staphylococcus aureus is an intracellular pathogen in the presence or absence of fibronectin-binding proteins. Microbial pathogenesis. 2003;35:159–168. doi: 10.1016/S0882-4010(03)00112-8. [DOI] [PubMed] [Google Scholar]
- 37.Bardiau M, Detilleux J, Farnir F, Mainil JG, Ote I. Associations between properties linked with persistence in a collection of Staphylococcus aureus isolates from bovine mastitis. Veterinary microbiology. 2014;169:74–79. doi: 10.1016/j.vetmic.2013.12.010. [DOI] [PubMed] [Google Scholar]
- 38.Hensen SM, Pavičić MJAMP, Lohuis JACM, Poutrel B. Use of bovine primary mammary epithelial cells for the comparison of adherence and invasion ability of Staphylococcus aureus strains. J. Dairy Sci. 2000;83:418–429. doi: 10.3168/jds.S0022-0302(00)74898-3. [DOI] [PubMed] [Google Scholar]
- 39.Bouchard DS, Rault L, Berkova N, Le Loir Y, Even S. Inhibition of Staphylococcus aureus invasion into bovine mammary epithelial cells by contact with live Lactobacillus casei. Appl. Environ. Microbiol. 2013;79:877–885. doi: 10.1128/AEM.03323-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Leitner G, Shoshani E, Krifucks O, Chaffer M, Saran A. Milk leucocyte population patterns in bovine udder infection of different aetiology. Journal of Veterinary Medicine. 2000;47:581–589. doi: 10.1046/j.1439-0450.2000.00388.x. [DOI] [PubMed] [Google Scholar]
- 41.Gonçalves JL, et al. Using milk leukocyte differentials for diagnosis of subclinical bovine mastitis. J. Dairy Res. 2017;84:309–317. doi: 10.1017/S0022029917000267. [DOI] [PubMed] [Google Scholar]
- 42.Riollet C, Rainard P, Poutrel B. Cell subpopulations and cytokine expression in cow milk in response to chronic Staphylococcus aureus infection. J. Dairy Sci. 2001;84:1077–1084. doi: 10.3168/jds.S0022-0302(01)74568-7. [DOI] [PubMed] [Google Scholar]
- 43.Boss R, et al. Bovine Staphylococcus aureus: Subtyping, evolution, and zoonotic transfer. Journal of dairy science. 2016;99:515–528. doi: 10.3168/jds.2015-9589. [DOI] [PubMed] [Google Scholar]
- 44.Graber HU, et al. Mastitis-related subtypes of bovine Staphylococcus aureus are characterized by different clinical properties. Journal of dairy science. 2009;92:1442–1451. doi: 10.3168/jds.2008-1430. [DOI] [PubMed] [Google Scholar]
- 45.Barlow JW, Zadoks RN, Schukken YH. Effect of lactation therapy on Staphylococcus aureus transmission dynamics in two commercial dairy herds. BMC veterinary research. 2013;9:28. doi: 10.1186/1746-6148-9-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.van den Borne BH, et al. Host adaptation of bovine Staphylococcus aureus seems associated with bacteriological cure after lactational antimicrobial treatment. Journal of dairy science. 2010;93:2550–2558. doi: 10.3168/jds.2009-2971. [DOI] [PubMed] [Google Scholar]
- 47.Petzl W, et al. Early transcriptional events in the udder and teat after intra-mammary Escherichia coli and Staphylococcus aureus challenge. Innate immunity. 2016;22:294–304. doi: 10.1177/1753425916640057. [DOI] [PubMed] [Google Scholar]
- 48.Petzl W, et al. Escherichia coli, but not Staphylococcus aureus triggers an early increased expression of factors contributing to the innate immune defense in the udder of the cow. Veterinary research. 2008;39:18. doi: 10.1051/vetres:2007057. [DOI] [PubMed] [Google Scholar]
- 49.Benjamin AL, Green BB, Hayden LR, Barlow JW, Kerr DE. Cow-to-cow variation in fibroblast response to a toll-like receptor 2/6 agonist and its relation to mastitis caused by intramammary challenge with Staphylococcus aureus. Journal of dairy science. 2015;98:1836–1850. doi: 10.3168/jds.2014-9075. [DOI] [PubMed] [Google Scholar]
- 50.Leitner G, et al. Development of a Staphylococcus aureus vaccine against mastitis in dairy cows. I. Challenge trials. Veterinary immunology and immunopathology. 2003;93:31–38. doi: 10.1016/S0165-2427(03)00051-5. [DOI] [PubMed] [Google Scholar]
- 51.Gronlund U, Hulten C, Eckersall PD, Hogarth C, Persson Waller K. Haptoglobin and serum amyloid A in milk and serum during acute and chronic experimentally induced Staphylococcus aureus mastitis. The Journal of dairy research. 2003;70:379–386. doi: 10.1017/S0022029903006484. [DOI] [PubMed] [Google Scholar]
- 52.Smith EM, et al. Multilocus sequence typing of intercontinental bovine Staphylococcus aureus isolates. Journal of clinical microbiology. 2005;43:4737–4743. doi: 10.1128/JCM.43.9.4737-4743.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wilson, G. Genome scale analysis of the role of superantigens in Staphylococcus aureus disease pathogenesis PhD thesis, University of Edinburgh (2011).
- 54.Wilson, G. J. et al. Bovine Staphylococcus aureus Superantigens Stimulate the Entire T Cell Repertoire of Cattle. Infection and immunity86 (2018). [DOI] [PMC free article] [PubMed]
- 55.Vrieling M, et al. LukMF′ is the major secreted leukocidin of bovine Staphylococcus aureus and is produced in vivo during bovine mastitis. Sci. Rep. 2016;6:37759. doi: 10.1038/srep37759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Prin-Mathieu C, et al. Enzymatic Activities of Bovine Peripheral Blood Leukocytes and Milk Polymorphonuclear Neutrophils during Intramammary Inflammation Caused by Lipopolysaccharide. Clin. Diagn. Lab. Immunol. 2002;9:812–817. doi: 10.1128/CDLI.9.4.812-817.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mehrzad J, Duchateau L, Burvenich C. Phagocytic and bactericidal activity of blood and milk-resident neutrophils against Staphylococcus aureus in primiparous and multiparous cows during early lactation. Vet. Microbiol. 2009;134:106–112. doi: 10.1016/j.vetmic.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 58.Dziewanowska K, et al. Fibronectin binding protein and host cell tyrosine kinase are required for internalization of Staphylococcus aureus by epithelial cells. Infection and immunity. 1999;67:4673–4678. doi: 10.1128/iai.67.9.4673-4678.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vrieling M, et al. LukMF’ is the major secreted leukocidin of bovine Staphylococcus aureus and is produced in vivo during bovine mastitis. Scientific reports. 2016;6:37759. doi: 10.1038/srep37759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hoekstra Jurriaan, Rutten Victor, Sommeling Laura, van Werven Tine, Spaninks Mirlin, Duim Birgitta, Benedictus Lindert, Koop Gerrit. High Production of LukMF’ in Staphylococcus aureus Field Strains Is Associated with Clinical Bovine Mastitis. Toxins. 2018;10(5):200. doi: 10.3390/toxins10050200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Budd, K. E. Staphylococcus aureus of bovine origin: genotypic diversity and phenotypic heterogeneity Doctor of Philosophy thesis, University College Dublin, (2015).
- 62.Spoor LE, et al. Recombination-mediated remodelling of host-pathogen interactions during Staphylococcus aureus niche adaptation. Microbial genomics. 2015;1:e000036. doi: 10.1099/mgen.0.000036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Huynh HT, Robitaille G, Turner JD. Establishment of bovine mammary epithelial cells (MAC-T): an in vitro model for bovine lactation. Exp. Cell Res. 1991;197:191–199. doi: 10.1016/0014-4827(91)90422-Q. [DOI] [PubMed] [Google Scholar]
- 64.Murphy MP, Niedziela DA, Keane OM. EHS matrix incubated in media containing penicillin retains sufficient concentrations of antibiotic to inhibit growth of susceptible microorganisms. J. Microbiol. Methods. 2017;139:103–106. doi: 10.1016/j.mimet.2017.05.011. [DOI] [PubMed] [Google Scholar]
- 65.Cronin JG, Hodges R, Pedersen S, Sheldon IM. Enzyme linked immunosorbent assay for quantification of bovine interleukin-8 to study infection and immunity in the female genital tract. Am. J. Reprod. Immunol. 2015;73:372–382. doi: 10.1111/aji.12344. [DOI] [PubMed] [Google Scholar]
- 66.Cano PM, Vargas A, Lavoie JP. A real-time assay for neutrophil chemotaxis. BioTechniques. 2016;60:245–251. doi: 10.2144/000114416. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.