Abstract
Hydroxytyrosol (HT) and Punicalagin (PC) exert cardioprotective and anti-atherosclerotic effects. This study evaluates the effect of oral supplementation with HT and PC (SAx) on early atherosclerosis markers in middle-aged, seemingly healthy adults. A randomized, double-blinded, placebo-controlled, crossover trial was performed for 20 weeks. There were two treatment sequences (Placebo/SAx, n = 41; SAx/Placebo, n = 43) for which the intervention periods (Placebo and SAx) were 8 weeks long, followed by a 4-week wash out period. The supplement was composed of 9.9 mg of HT and 195 mg of PC, and the placebo was composed of maltodextrin. SAx increased endothelial function (Flow-mediated dilatation [FMD]: 2.36%; p < 0.001) in the endothelial dysfunction subgroup compared to the placebo (2.36 ± 3.9 vs. 0.76 ± 3.5%, p < 0.05). SAx also reduced oxLDL by −28.74 ng/mL (p < 0.05) in subjects with higher levels of oxLDL, which was an improvement compared with the placebo (−28.74 ± 40.2 vs. 25.64 ± 93.8 ng/mL, p < 0.001). The prehypertension and hypertension subgroups exhibited decreased systolic (−15.75 ± 9.9 mmHg; p < 0.001) and diastolic (−6.36 ± 8.7 mmHg; p < 0.001) blood pressure after SAx consumption. Moreover, the systolic prehypertension and hypertension subgroups presented significant differences in systolic blood pressure compared to the placebo (−15.75 ± 9.9 vs. −2.67 ± 12.0 mmHg, p < 0.05). In conclusion, the supplement exerted anti-atherosclerotic effects by improving endothelial function, blood pressure, and levels of circulating oxLDL, especially for persons in whom these parameters were altered.
Keywords: atherosclerosis, hydroxytyrosol, punicalagin, endothelial dysfunction, oxidative stress, prehypertension, hypertension
1. Introduction
Cardiovascular disease (CVD) is the leading cause of death worldwide. The annual mortality from CVD is higher than from any other cause [1,2]. In the last 40 years, a rapid increase in the prevalence of cardiovascular diseases has been observed. In 2030, it is estimated that approximately 23.3 million people will die from CVD, which is likely to remain the world’s foremost cause of death [3]. Most cases of CVD, however, are preventable through the reduction of behavioural risks (tobacco use, unhealthy diet, physical inactivity or harmful use of alcohol) [1,4].
Atherosclerosis, which is also known as atherosclerotic vascular disease (ASVD), is one of the most important causes of CVD. The development of atherosclerosis involves a number of pathophysiological mechanisms [5,6], including endothelial dysfunction (ED) [7]. ED, which is greatly underdiagnosed in the general population, plays a pivotal role in the early stages of ASVD, and is later associated with plaque progression and the occurrence of cardiovascular (CV) events [8]. Flow-mediated dilatation (FMD) is an accepted technique to quantify endothelial function and has been shown to have prognostic value to future CV events in symptomatic [9,10] and apparently healthy or asymptomatic subjects [11,12,13].
With regard to blood pressure, it is well known that prehypertension often leads to hypertension [14,15] and an increased risk of CVD and future CV events [16]. Furthermore, the relationship between blood pressure and the risk of CV events is continuous, consistent, and independent of other cardiovascular risk factors [17]. Therefore, the prevention and treatment of hypertension is key for reducing the burden of CV morbi-mortality [18,19].
The role of oxidative stress (OS) in the pathophysiology of CVD is well established [6,20,21]. OS is one of the primary mechanisms involved in the development of ASVD. The circulating level of oxidised low-density lipoproteins (oxLDL) is one of the most important markers in the atherogenic process [22]. These lipoproteins contribute significantly to the development of ASVD, and they not only promote the formation of atherosclerotic plaque [23], but also participate in its progression [24,25,26] and destabilization [27,28,29,30]. This action is carried out through several mechanisms, including the promotion of ED [31,32,33,34]. Consequently, elevated levels of circulating oxLDL are associated with all stages of atherosclerosis, from the peripheral early atherogenesis stage, through coronary artery disease, until acute coronary syndromes and ischaemic stroke [22]. Therefore, the level of circulating oxLDL is a predictor of future CV events in apparently healthy or asymptomatic subjects [35] and in symptomatic subjects [27,29].
Polyphenols are gaining increasing acceptance as therapeutic agents for use in diverse diseases, including CVD [36] and its risk factors [37,38,39,40,41,42]. Several studies have reported an inverse correlation between the consumption of polyphenols and the risk of CV events [36] and overall mortality [40]. Among these compounds, hydroxytyrosol (HT, from olives) [43,44,45] and punicalagin (PC, from pomegranates) [46,47] have been attributed as having cardioprotective properties. The intake of adequate amounts of these bioactive compounds may therefore help reduce the morbidity-mortality associated with CVD.
The aim of the present work was to determine the effect of supplementation with HT and PC on endothelial dysfunction and other early atherosclerosis markers in apparently healthy middle-aged subjects.
2. Materials and Methods
The present study was registered at http://clinicaltrials.gov under the number NCT02042742.
2.1. Study Subjects
One hundred and five subjects aged 45–65 years were recruited for the present study by the Nutrition Department of La Paz University Hospital (HULP), Madrid (Spain). The inclusion criteria to be eligible for the study were as follows: aged between 45 and 65 years, having a suitable understanding of the clinical trial level, agreeing to voluntarily participate in the study, and signing the informed consent. The exclusion criteria were as follows: a BMI of ≥30 kg/m2; subjects receiving drug treatment for CV risk (dyslipidaemia, hypertension, diabetes mellitus, and others); those who had been diagnosed with metabolic syndrome, who had a mental illness or low cognitive ability; and those who suffered severe liver or kidney disease, or cancer. Subjects were also excluded if they had a family background of premature vascular disease, or if they were taking supplements (antioxidants, ω-3, vitamins, minerals, or probiotics). Women still experiencing menstrual cycles and subjects who planned to stop smoking or lose weight during the study were also excluded, as were subjects who had problems with complying with the general dietary recommendations, those who undertook intensive physical activity, those who consumed >30 g/day alcohol, and those who had allergies to olive and pomegranate by-products.
All subjects gave their informed consent to take part in the study, which was approved by the Scientific Research and Ethics Committee of the HULP (Code 3799) in accordance with the ethical standards of the Declaration of Helsinki [48].
2.2. Study Design
The study took the form of a randomized, controlled, double-blind, crossover clinical trial lasting 20 weeks. Subjects (n = 84) were randomly assigned (maintaining the gender ratio of the sample) to one of two treatment sequences involving an oral supplementation (9.9 mg of HT plus 195 mg of PC, and 995.1 mg of maltodextrin per day) or a placebo (1.200 mg of maltodextrin per day) for 8 weeks, followed by a 4-week wash-out interval and then a cross-over. During the 8-week intervention period, subjects consumed 3 capsules/day (containing either the oral supplement SAx or the placebo) with their meals. Neither the researchers nor the subjects knew which treatment sequence the subjects had been assigned to; the researchers were unblinded only at the end of the study.
2.3. Supplement and Placebo Capsules
The doses administered in the present work were in agreement with those reported in the literature [44,49,50,51]. Each supplement capsule (SAx) contained 3.3 mg of HT from a standardized olive fruit extract (Mediteanox®, Euromed S.A., Barcelona, Spain), 65 mg of PC from a standardized pomegranate fruit extract (Pomanox® P30, Euromed S.A., Barcelona, Spain), and 331.7 mg of maltodextrin. Each Placebo capsule contained 400 mg of maltodextrin. Probelte Pharma S.A. (Murcia, Spain) prepared the supplement capsules (with specified concentrations of HT and PC) and the placebo capsules specifically for this study. Both types of capsules were packaged in blister packs of 15, and the blister packs were labelled as either L1 or L2 to maintain blinded conditions. Subjects received all the capsules needed to complete the 8-week intervention period (SAx or placebo) during repeat appointments at the HULP.
2.4. Methods
2.4.1. Diet
All subjects were told to maintain their normal dietary habits and not to increase their consumption of foods rich in antioxidants. The diet of each subject was recorded during the week prior to the beginning and end of each intervention period. All food and beverages consumed inside and outside the home were recorded over three consecutive days (including one day of the weekend) [52]. Subjects were instructed to record the weight of the food consumed or, if this was not possible, to record household measurements (spoonfuls, cups, etc.). At each visit, all records were thoroughly reviewed by a nutritionist in the presence of the subject to ensure that the information collected was complete. The energy and nutritional content of the foods and beverages consumed were then calculated using DIAL software (Alce Ingeniería, Madrid, Spain).
2.4.2. Anthropometric Variables
Anthropometric measurements were taken at the beginning and end of each intervention period using standard techniques, adhering to international norms set out by the WHO [53]. All measurements were made by trained personnel in the morning with the subject barefoot and wearing only underwear. Body composition was determined using a specialized bioelectrical impedance analyser, the EFG ElectroFluidGraph analyser (Akern s.r.l., Florence, Italy). Height was determined using a height meter with an accuracy of 1 mm (range, 80–200 cm). BMI was calculated using the following formula: [weight (kg)/height (m)2].
2.4.3. Health Variables
Information was collected on medical conditions and the consumption of medications. Blood pressure and heart rate were measured on the right arm using a Spot Vital Signs 420 automatic monitor (Welch Allyn, Madrid, Spain) (accuracy ±5 mmHg). Three measurements were taken at 5-min intervals, and the means were calculated. Populations were classified according to whether the subjects exhibited prehypertension or high blood pressure that was non-diagnosed and was not being treated pharmacologically.
2.4.4. Determination of Vascular Function Variables
Flow-mediated dilatation (FMD) in the brachial artery was used as an indicator of endothelial function. FMD has been shown to be an independent predictor of CV events [9,10], even in apparently healthy subjects [11,12,13]. Measurements were performed based on recommendations described in published guidelines [54] using a Doppler ultrasound device with a 10-MHz linear probe (Biosound MyLab 25 Portable Ultrasound System, Esaote, Genoa, Italy). Briefly, the subject lay in the supine position in a room at 20–22 °C. A sphygmomanometer cuff was positioned just below the axilla, and the Doppler probe was placed 3 cm above the right elbow (with the arm extended) at an angle of 60 °. After a 10-min resting period, basal capillary flow was measured (t0). Thereafter, distal ischaemia was induced for 5 min by inflating the cuff to a pressure of 250 mmHg. The cuff was then deflated and, after 70 s, the flow was recorded (td). All images were captured by the same qualified operator. All variables were measured in triplicate, and their means were calculated. The area under the curve (AUC) for t0 and td was then determined. The FMD was calculated using the following equation: FMD = (AUCtd − AUCt0) × 100/AUCt0, and the subjects were classified according to the presence or absence of ED (ED = FMD < 10%) [55].
2.4.5. Biochemical Data
At the beginning and end of each intervention period, blood samples were collected early in the morning at the La Paz University Hospital Extraction Unit. Samples were kept at 4–6 °C until analysis, which was always performed within 48 h. The concentration of plasma-soluble vascular cell adhesion molecule-1 (sVCAM-1) was determined using a Luminex® 200TM multianalyte profiling system and commercially available immunoassay panels in the HCVD2MAG-67K Milliplex Map Kit (EMP Millipore Corp., Boston, MA, USA). Plasma Ox-LDL levels were measured using ELISA with human apoB-100 modified by Malondialdehyde (MDA-LDL) (Immundiagnostik AG, Bensheim, Germany).
Total plasma lipid peroxides were measured using the thiobarbituric acid reactive substances (TBARS) method [56]. Plasma antioxidant capacity was analysed using a ferric reducing antioxidant power (FRAP) assay [57]. Quantitative measurements of Paraoxonase-1 (PON-1) were made using a Human Serum PON-1 ELISA Kit (Aviscera Bioscience, Santa Clara, CA, USA). Plasma 8-isoprostanes (8-iso-PGF2α) was measured using an OxiSelectTM 8-iso-PGF2α ELISA kit (Cell Biolabs, San Diego, CA, USA). Nitrate/Nitrite (NOx) was analysed in plasma using a commercially available colorimetric assay kit (Cayman Chemical Company, Ann Arbor, MI, USA) according to the Griess method [58].
2.4.6. Compliance and Adverse Events
Subjects received the exact number of capsules (in blister packaging) required for each intervention period during a pre-period appointment, and they were asked to return all empty and non-empty blister packages. Compliance was measured at the middle of each intervention period during an interview and at the end of each intervention period by comparing the number of capsules provided and the number returned. A subject was considered compliant when he/she consumed ≥90% of the capsules provided. Adverse events were recorded on the final visit of each intervention period. An adverse event was defined as any unfavourable, unintended effect. All such events were recorded along with the symptoms involved (nausea, vomiting, diarrhoea, and constipation).
2.5. Statistical Analysis
A sample size of 38 patients was calculated as the sample size that was necessary to provide 90% power (at α = 0.05) to determine an absolute difference of 2% in FMD [59] (potential 20% dropout included). Quantitative data are presented as the means ± standard deviations (SD). Qualitative data are presented as counts and percentages. The Kolmogorov–Smirnov test was used to determine whether the data were normally distributed. Levene’s test was used to assess the equality of variance. The denominator degree of freedom was approximated using the Satterthwaite formula. The type class variables considered in the primary model included the treatment, period, treatment × period interaction, risk, and the treatment × risk interaction; these variables were added as fixed effects. The type class variables considered in the second model included the start–end, treatment, start–end × treatment interaction, period, treatment × period interaction, start–end × treatment × period interaction, the risk, treatment × risk interaction, and the start–end × treatment × risk interaction; these variables were added as fixed effects. For both models, the subject was considered in sequence as a random effect and a “variance components” covariance matrix. The type III tests of fixed effects table lists the hypothesis tests for the significance of each of the fixed effects specified in both models; in particular, the carryover effect was assessed in the primary model by the treatment × period interaction effect. If the carryover effect was significant for a given variable, then only the first period was considered for the analysis. Least squares means of the primary model were computed and compared for the treatment and the treatment × risk interaction variables. Least squares means of the second model were computed and compared for the treatment × start–end interaction and the start–end × treatment × risk interaction. Multiple comparisons were adjusted using the Bonferroni method. Two-sided tests were used, and a p-value < 0.05 was considered statistically significant. Statistical analyses were performed using the linear mixed model in the SAS statistical software package, version 9.3 (SAS Institute Inc., Cary, NC, USA).
3. Results
3.1. Recruitment and Study Population
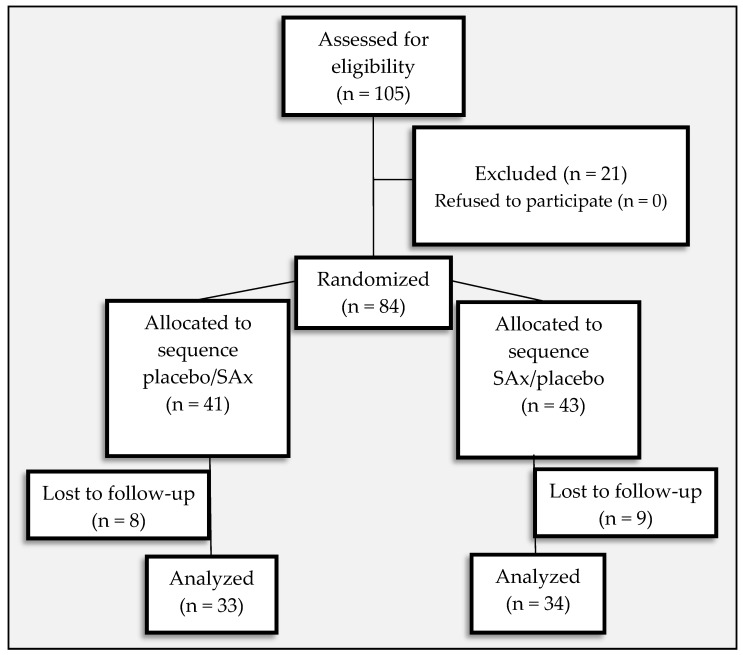
The study was performed between February and June 2013. Eighty-four apparently healthy subjects (17 men [20.2%], 67 women [79.8%]) were eligible for inclusion. Seventeen subjects were lost to follow-up (8 in the placebo/SAx sequence and 9 in the SAx/placebo sequence) due to personal causes (n = 15) and failure to follow treatment instructions (n = 2). Thus, 67 subjects (14 men [20.9%], 53 women [79.1%]) completed the 20-week study, and only their results were included in the subsequent analyses (Figure 1).
Figure 1.
Flow chart describing the present trial.
3.2. Baseline Characteristics
The mean age of the population was 53.0 ± 4.5 years old. The mean BMI was 24.6 ± 3.1 kg/m2. At the start of the study, no significant differences existed between subjects assigned to the supplementation (SAx) and placebo sequences in terms of their anthropometric, vascular function, oxidative status, and other variables (gender, age, and smoking) (Table 1).
Table 1.
Baseline characteristics of the subjects.
| Placebo/SAx (n = 33) | SAx/Placebo (n = 34) | ||
|---|---|---|---|
| Gender | (Female %, n) | 78.79 (26) | 79.41 (27) |
| Age | (years) | 53.21 ± 4.2 | 52.79 ± 4.8 |
| Smoking | (Smokers %, n) | 18.18 (6) | 26.47 (9) |
| Weight | (kg) | 66.26 ± 11.8 | 64.08 ± 10.9 |
| BMI | (kg/m2) | 24.64 ± 2.9 | 24.56 ± 3.2 |
| Waist circumference | (cm) | 80.51 ± 9.2 | 82.58 ± 9.8 |
| FM | (%) | 29.18 ± 6.7 | 28.76 ± 6.4 |
| FFM | (%) | 70.82 ± 6.7 | 71.24 ± 6.4 |
| MM | (%) | 48.03 ± 7.7 | 47.87 ± 5.5 |
| SBP | (mmHg) | 110.3 ± 13.1 | 110.9 ± 12.9 |
| DBP | (mmHg) | 74.06 ± 10.8 | 73.75 ± 9.5 |
| HR | (bpm) | 67.36 ± 8.9 | 70.41 ± 7.5 |
| FMD | (%) | 8.464 ± 4.0 | 7.962 ± 3.8 |
| sVCAM-1 | (pg/mL) | 679.9 ± 191.8 | 661.5 ± 183.1 |
| oxLDL | (ng/mL) | 118.2 ± 144.4 | 103.4 ± 113.9 |
| TBARS | (µmol MDA eq/mL) | 0.622 ± 0.8 | 0.572 ± 0.7 |
| FRAP | (µmol Trolox eq/mL) | 0.410 ± 0.1 | 0.425 ± 0.1 |
| 8-iso-PGF2α | (ng/mL) | 58.55 ± 12.6 | 56.95 ± 10.3 |
| PON-1 | (ng/mL) | 5.777 ± 6.5 | 4.383 ± 3.9 |
| NOx | (µM) | 23.28 ± 9.0 | 27.16 ± 19.9 |
Data are expressed as the means ± SDs. SAx: Oral Supplementation with HT and PC; BMI: Body Mass Index; FM: Fat mass; FFM: Fat-Free Mass; MM: Muscle Mass; SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HR: Heart Rate; FMD: Flow Mediated Dilatation; sVCAM-1: soluble Vascular Cell Adhesion Molecule-1; oxLDL: Oxidised Low-Density Lipoprotein; TBARS: Thiobarbituric Acid Reactive Substances; FRAP: Ferric Reducing Antioxidant Power; 8-iso-PGF2α: 8-iso-prostaglandinF2α; PON-1: Paraoxonase-1; NOx: Nitrates & Nitrites. There are not significant differences between the start and end of the intervention periods or in the change recorded between the intervention periods.
3.3. Dietetic and Anthropometric Variables
No significant differences were detected in any dietetic or anthropometric variables between the start and end of the intervention periods, nor between the periods in terms of changes in these variables (Table 2).
Table 2.
Dietetic or anthropometric variables at the start and end of the supplementation and placebo periods.
| SAx | Placebo | |||
|---|---|---|---|---|
| (n = 67) | (n = 67) | |||
| Energy | (kcal/day) | Start | 1923 ± 513.6 | 1864 ± 471.9 |
| End | 1891 ± 549.4 | 1881 ± 569.5 | ||
| Change | −31.88 ± 463.2 | 17.07 ± 355.5 | ||
| Carbohydrates | (%) | Start | 38.06 ± 6.5 | 38.46 ± 8.9 |
| End | 37.62 ± 6.3 | 39.06 ± 6.5 | ||
| Change | −0.439 ± 5.7 | 0.598 ± 8.1 | ||
| Proteins | (%) | Start | 17.24 ± 3.7 | 17.44 ± 2.9 |
| End | 17.43 ± 3.6 | 17.60 ± 3.1 | ||
| Change | 0.193 ± 4.4 | 0.164 ± 3.5 | ||
| Lipids | (%) | Start | 41.42 ± 6.3 | 40.48 ± 8.8 |
| End | 41.36 ± 5.7 | 40.19 ± 5.9 | ||
| Change | −0.067 ± 5.3 | −0.287 ± 8.0 | ||
| Weight | (kg) | Start | 65.10 ± 11.3 | 65.10 ± 11.2 |
| End | 64.93 ± 11.2 | 64.85 ± 11.3 | ||
| Change | −0.173 ± 1.3 | −0.249 ± 1.0 | ||
| BMI | (kg/m2) | Start | 24.58 ± 3.0 | 24.63 ± 3.0 |
| End | 24.51 ± 3.0 | 24.48 ± 3.0 | ||
| Change | −0.068 ± 0.5 | −0.151 ± 0.6 | ||
| Waist | (cm) | Start | 81.85 ± 9.0 | 81.24 ± 9.8 |
| circumference | End | 81.82 ± 9.6 | 81.25 ± 9.6 | |
| Change | −0.034 ± 2.9 | 0.008 ± 3.7 | ||
| FM | (%) | Start | 29.16 ± 6.6 | 28.90 ± 6.5 |
| End | 29.56 ± 6.8 | 29.79 ± 7.2 | ||
| Change | 0.400 ± 2.8 | 0.891 ± 3.7 | ||
| FFM | (%) | Start | 70.84 ± 6.6 | 71.10 ± 6.5 |
| End | 70.44 ± 6.8 | 70.21 ± 7.2 | ||
| Change | −0.400 ± 2.8 | −0.891 ± 3.7 | ||
| MM | (%) | Start | 47.63 ± 6.2 | 47.52 ± 6.5 |
| End | 46.67 ± 5.7 | 46.44 ± 5.8 | ||
| Change | −0.970 ± 5.1 | −1.082 ± 5.8 |
Data are expressed as the means ± SDs. SAx: Oral Supplementation with HT and PC; BMI: Body Mass Index; FM: Fat mass; FFM: Fat-Free Mass; MM: Muscle Mass. There are not significant differences between the start and end of the intervention periods or in the change recorded between the intervention periods.
3.4. Vascular Function Variables
Table 3 shows the values of the vascular function variables examined. A significant reduction in systolic blood pressure (SBP) was observed at the end of the SAx period (SAx period—start 111.3 ± 12.9, end 101.9 ± 12.0 mmHg, p < 0.001). The reduction in SBP observed after the supplementation treatment was significantly greater than that recorded for the placebo treatment (SAx −9.42 ± 10.3 vs. placebo −2.79 ± 11.2 mmHg, p < 0.05). After the SAx treatment, subjects with systolic prehypertension or hypertension exhibited significantly decreased SBP (SAx period—start 123.9 ± 4.2, end 108.2 ± 10.8 mmHg, p < 0.001); no such significant reduction was observed at the end of the placebo period (from 129.8 ± 2.8 to 127.2 ± 10.4 mmHg). In addition, in these subjects with systolic prehypertension or hypertension, the reduction in SBP that was observed following the SAx treatment was significantly greater than that recorded for the placebo treatment (SAx −15.75 ± 9.9 vs. placebo −2.67 ± 12.0 mmHg, p < 0.05).
Table 3.
Vascular function variables at the start and end of the supplementation and placebo periods.
| SAx | Placebo | |||
|---|---|---|---|---|
| (n = 67) | (n = 67) | |||
| SBP | (mmHg) | Start | 111.3 ± 12.9 | 110.2 ± 13.3 |
| End | 101.9 ± 12.0 *** | 107.4 ± 14.8 | ||
| Change | −9.419 ± 10.3 # | −2.792 ± 11.2 | ||
| DBP | (mmHg) | Start | 74.34 ± 10.1 | 72.68 ± 10.1 |
| End | 71.60 ± 10.4 *** | 71.42 ± 9.7 | ||
| Change | −2.742 ± 8.4 | −1.258 ± 8.1 | ||
| HR | (bpm) | Start | 68.63 ± 10.7 | 69.13 ± 9.4 |
| End | 67.67 ± 9.8 | 67.88 ± 8.4 | ||
| Change | −0.962 ± 8.3 | −1.250 ± 9.1 | ||
| FMD | (%) | Start | 8.036 ± 4.0 | 8.077 ± 3.3 |
| End | 9.462 ± 4.0 * | 8.619 ± 4.0 | ||
| Change | 1.427 ± 3.7 | 0.542 ± 3.4 | ||
| sVCAM-1 | (pg/mL) | Start | 650.8 ± 192.9 | 635.3 ± 174.9 |
| End | 625.0 ± 163.9 | 588.7 ± 136.7 | ||
| Change | −25.84 ± 175.1 | −46.66 ± 126.6 |
Data are expressed as the means ± SDs. SAx: Oral Supplementation with HT and PC; SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; HR: Heart Rate; FMD: Flow Mediated Dilatation; sVCAM-1: soluble Vascular Cell Adhesion Molecule-1. Significant differences between the start and end of the intervention periods (* p < 0.05, *** p < 0.001). Significant differences in the change recorded between the intervention periods (# p < 0.05).
A significant reduction in diastolic blood pressure (DBP) was also observed at the end of the SAx period (from 74.34 ± 10.1 to 71.60 ± 10.4 mmHg, p < 0.001); no such significant reduction was observed at the end of the placebo period (from 72.68 ± 10.1 to 71.42 ± 9.7 mmHg). These improvements were observed as well in the subgroup of diastolic prehypertension and hypertension after SAx treatment (from 84.4 ± 6.5 to 78.04 ± 10.8 mmHg, p < 0.001); no such improvement was observed after the placebo period (from 83.0 ± 5.6 to 79.9 ± 8.8 mmHg, NS). Subjects with normal blood pressure remained stable.
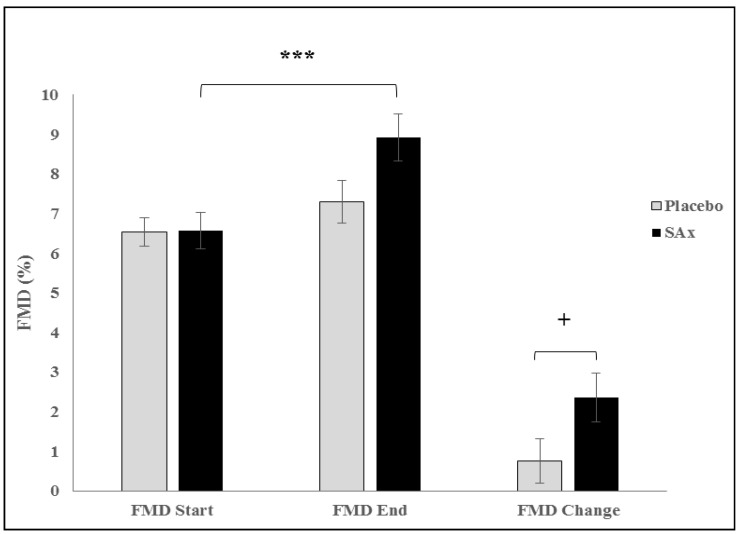
Significant differences were also observed in the FMD between the start and the end of the SAx period (from 8.04 ± 4.0 to 9.46 ± 4.0%, p < 0.05); no such improvement was observed for the placebo period (from 8.08 ± 3.3 to 8.62 ± 4.0%, NS). In subjects with ED, a significant increase in FMD was observed after the SAx period (from 6.57 ± 2.9 to 8.93 ± 3.8%, p < 0.001) but not after the placebo period (from 6.54 ± 2.3 to 7.30 ± 3.4%, NS). The increase in FMD that was observed following the SAx treatment was significantly greater than that recorded for the placebo treatment (SAx 2.36 ± 3.9 vs. placebo 0.76 ± 3.5 %, p < 0.05) (Figure 2). No significant changes in FMD were observed in subjects without ED in either intervention period (SAx from 10.86 ± 4.5 to 10.70 ± 4.2%; placebo from 11.16 ± 2.9 to 11.26 ± 3.7%). No differences were recorded in heart rate (HR) or soluble sVCAM-1 in any comparison.
Figure 2.
Endothelial Function (FMD), which is a predictor of future cardiovascular events in symptomatic and asymptomatic subjects, significantly increased following SAx (Oral Supplementation with HT and PC) treatment (black color) in apparently healthy subjects (*** p < 0.001), and this improvement was significantly different compared to that observed following the placebo (grey) (+p < 0.05). Forty-five subjects had endothelial dysfunction. Data represent the adjusted means (±SD) from multivariate models.
3.5. Oxidative Status Variables
Table 4 shows the values of the oxidative status variables recorded. Circulating oxLDL levels were reduced significantly after the SAx period (from 108.9 ± 126.2 to 97.44 ± 121.7 ng/mL, p < 0.05); however, no significant reduction was recorded after the placebo period (from 98.86 ± 128.1 to 105.9 ± 139.9 ng/mL). The reduction in oxLDL observed after the SAx treatment was significantly greater than that recorded for the placebo treatment (SAx −11.46 ± 28.1 vs. placebo 7.05 ± 55.6 ng/mL, p < 0.05). In subjects with higher levels of oxLDL, these levels were reduced significantly after the SAx treatment (from 258.2 ± 138.0 to 229.5 ± 149.5 ng/mL, p < 0.05); no significant reductions were observed after the placebo period (from 235.4 ± 162.5 to 261.0 ± 170.8 ng/mL). In these subjects, the reduction in oxLDL that was observed following the SAx period was significantly greater than that recorded for the placebo treatment (−28.74 ± 40.2 vs. 25.64 ± 93.8 ng/mL, p < 0.001). No significant differences were observed for the remaining variables.
Table 4.
Oxidative status at the start and end of the supplementation and placebo periods.
| SAx | Placebo | |||
|---|---|---|---|---|
| (n = 67) | (n = 67) | |||
| oxLDL | (ng/mL) | Start | 108.9 ± 126.2 | 98.86 ± 128.1 |
| End | 97.44 ± 121.7 * | 105.9 ± 139.9 | ||
| Change | −11.46 ± 28.1 # | 7.05 ± 55.6 | ||
| TBARS | (µmol MDA eq/mL) | Start | 0.696 ± 0.9 | 0.673 ± 0.9 |
| End | 0.635 ± 0.9 | 0.574 ± 0.8 | ||
| Change | −0.061 ± 1.2 | −0.099 ± 1.1 | ||
| FRAP | (µmol Trolox eq/mL) | Start | 0.421 ± 0.1 | 0.422 ± 0.1 |
| End | 0.425 ± 0.1 | 0.433 ± 0.1 | ||
| Change | 0.005 ± 0.1 | 0.011 ± 0.0 | ||
| 8-iso-PGF2α | (ng/mL) | Start | 52.54 ± 11.7 | 54.06 ± 13.6 |
| End | 50.68 ± 14.0 | 50.55 ± 11.7 | ||
| Change | −1.858 ± 13.7 | −3.507 ± 14.9 | ||
| PON-1 | (ng/mL) | Start | 4.187 ± 4.4 | 4.130 ± 5.5 |
| End | 3.567 ± 5.1 | 4.514 ± 7.0 | ||
| Change | −0.619 ± 6.5 | 0.384 ± 7.7 | ||
| NOx | (µM) | Start | 25.34 ± 16.2 | 23.57 ± 13.5 |
| End | 26.39 ± 16.9 | 22.52 ± 9.7 | ||
| Change | 1.053 ± 14.4 | −1.055 ± 13.1 |
Data are expressed as the means ± SDs. SAx: Oral Supplementation with HT and PC; OxLDL: Oxidised Low-Density Lipoprotein; TBARS: Thiobarbituric Acid Reactive Substances; FRAP: Ferric Reducing Antioxidant Power; 8-iso-PGF2α: 8-iso-prostaglandinF2α; PON-1: Paraoxonase-1; NOx: Nitrates & Nitrites. Significant difference between the start and end of the intervention periods (* p < 0.05). Significant differences in the change recorded between the intervention periods (# p < 0.05).
3.6. Compliance and Adverse Events
All subjects ingested >90% of the capsules provided. No significant differences were observed in the number of capsules consumed between the different intervention periods or treatment sequences. No adverse events resulting from the intake of either type of treatment capsule were reported.
4. Discussion
This is the first clinical trial to study the effect of the regular intake of a supplement rich in HT and PC on early atherosclerosis markers in middle-age subjects. The consumption of three capsules per day containing HT and PC (9.9 mg of HT and 195 mg of PC) for 8 weeks significantly improved endothelial function and blood pressure and reduced LDL oxidation in middle-aged subjects. These improvements were observed primarily in subjects who were at risk of developing cardiovascular disease. No adverse effects were observed.
Several studies have shown that HT [43,44,45,60,61] and PC [46,47] possess cardioprotective properties that may reduce the risk of developing CVD. However, most of these studies were performed in vitro or with experimental animals [24,25,26,27,28]; some studies have investigated the effects of these compounds in humans [60,62], and no studies have examined the combined effect of HT and PC.
The present results reveal a significant improvement in FMD among subjects with ED. One of the major objectives of a CVD detection programme must be to identify those apparently healthy subjects who have asymptomatic arterial disease; such identification may reduce the risk, delay the onset, and perhaps even induce the regression of atherosclerotic disease. The importance of new imaging methods for the detection of subjects at a high risk for suffering CV events has been recognized [63]. FMD is a non-invasive imaging technique that is accepted for quantifying endothelial function; in addition, FMD is the most widely used technique for testing endothelial function. FMD has been shown to have prognostic value for predicting future CV events in symptomatic subjects [9,10] and apparently healthy or asymptomatic subjects [11,12,13]. In fact, Kuvin et al. [55] observed that FMD measurements of the brachial artery were highly predictive of CVD, with an increase in the odds ratio of 1.32 for each percentage decrease in FMD below 10% [55]. The FMD also has a significant potential as a screening tool for CVD in individuals with an apparent low CV risk assessed by traditional risk factors [55]. The improvement in endothelial function from HT, PC, or foods rich in these compounds has been observed in many studies [41,47,62,64]. In addition, some authors suggest a possible inverse relationship between FMD and oxLDL levels [31,41]. In the present study, simultaneous improvements in FMD and oxLDL were observed.
It therefore appears that subjects with ED could benefit from supplementation with HT and PC, even if they are not diagnosed. In this regard, there remains a high prevalence of undiagnosed ED. Indeed, in this study of apparently healthy subjects, the percentage of subjects who were eventually diagnosed with ED reached 69.2%. In subjects with ED, these benefits could potentially help to reduce their risk of CVD and the future burden they may pose to the health system. In the present work, SBP and DBP significantly improved in the total population after intake of the supplement for 8 weeks. However, this effect was observed primarily in subjects suffering from prehypertension or hypertension. This finding is important because, in addition to hypertension, prehypertension also increases the risk of CVD and CV events [16]. Hypertension is usually inadequately controlled [65], and antihypertensive drugs can produce adverse effects that often lead to patient abandonment [66]. Therefore, the prevention and treatment of hypertension is key for reducing the burden of CV morbi-mortality [18,19]. In this context, various studies in which HT or PC were provided via functional foods or foods rich in these compounds also reported significant improvements in blood pressure [39,67]. Even the European Medicines Agency (EMA) recognized the hypotensive activity of olive leaf; however, the EMA mentioned that there are not sufficient data from well-designed clinical trials to support this indication [68]. Therefore, the present study contributes additional evidence suggesting that the hypotensive action after the consumption of foods, functional foods, or supplements containing HT or PC (or both) may help regulate blood pressure.
On the other hand, numerous studies have reported a reduction in oxLDL levels after the intake of foods rich in HT or PC [40,44,67,69,70,71]. In a crossover clinical trial in healthy subjects, it was reported that the consumption of 25 mL/day virgin olive oil (~3.75 mg/day phenolic compounds) for 3 weeks significantly reduced plasma oxLDL levels [69]. In the EUROLIVE study, a crossover clinical trial in healthy subjects reported that the consumption of 25 mL/day high-phenolic-content virgin olive oil (366 mg/L phenolic content) for 3 weeks induced a greater reduction in oxLDL levels compared with olive oils with a lower phenolic content [40]. In this regard, the EFSA recognized a cause and effect relationship between the daily consumption of 5 mg of HT and its derivatives from olive oil and the protection of c-LDL particles from oxidative damage [44]. The results of the present work are in line with these studies regarding HT as a protection strategy against c-LDL oxidative damage [44]. One mechanism through which HT may reduce oxLDL levels is by modulating the expression of genes involved in atherogenesis [45,72]. Other authors have reported that the long-term consumption of 50 mL/day pomegranate juice (~85 mg of PC) significantly reduces oxLDL levels in patients with carotid artery stenosis [67]. PC may provide this benefit through a direct antioxidant effect on c-LDL, by increasing serum PON-1 levels, and/or by reducing the capacity of macrophages to oxidize c-LDL [67,70,71]. However, in the present work, although supplementation with HT and PC reduced the formation of oxLDL, no change was observed in serum PON-1 levels or, indeed, in any other oxidative status variables (TBARS, FRAP, 8-iso-PGF2α, and NOx). This discrepancy may be explained by the fact that previous studies have involved subjects with CVD-related pathologies, whereas the subjects in the present study were apparently healthy. Similarly, PON-1 activity has been observed to be decreased in subjects with diabetes [73] but not in those with pre-diabetes or a new diagnosis of diabetes [74].
A high level of circulating oxLDL is a marker of ED [31] and a predictor of future CVD events in apparently healthy or asymptomatic subjects [35]. Therefore, the regular intake of supplements containing both HT and PC, which reduce the formation of oxLDL in subjects with elevated oxLDL levels, may help to reduce the CV risk in these subjects.
One possible limitation of this study is the sample size.
5. Conclusions
In summary, the consumption of a supplement containing HT and PC (9.9 mg of HT and 195 mg of PC per day) for 8 weeks could help reducing c-LDL oxidation and improved SBP, DBP, and FMD in middle-aged subjects. These improvements in FMD, blood pressure, and circulating oxLDL levels were most pronounced in subjects with alterations in these atherosclerotic markers. Therefore, the regular intake of a supplement containing HT and PC may reduce the risk of CV in these subjects. Further clinical trials are warranted to confirm the beneficial effect of these polyphenols in humans.
Acknowledgments
We thank Probelte Pharma S.A. for the manufacture and supply of the supplement and placebo capsules (Pomanox® and Mediteanox® are now manufactured and owned by EUROMED S.A.). Also, thanks to the Biostatistics Department at the HULP for their continuous support, and the members of the Internal Medicine Unit at the HULP for their assistance with the Doppler ultrasonography.
Abbreviations
| 8-iso-PGF2α | 8-isoprostanes |
| ASVD | Atherosclerotic Vascular Disease |
| AUC | Area Under the Curve |
| CV | Cardiovascular |
| CVD | Cardiovascular Disease |
| DBP | Diastolic Blood Pressure |
| ED | Endothelial Dysfunction |
| ELISA | Enzyme-Linked-Immunosorbent-Assay |
| FMD | Flow-Mediated Dilatation |
| FRAP | Ferric Reducing Antioxidant Power |
| HR | Heart Rate |
| HT | Hydroxytyrosol |
| HULP | University Hospital La Paz |
| OS | Oxidative Stress |
| oxLDL | oxidized Low Density Lipoprotein Cholesterol |
| PC | Punicalagin |
| PON-1 | Paraoxonase-1 |
| SBP | Systolic Blood Pressure |
| sVCAM-1 | soluble Vascular Cell Adhesion Molecule-1 |
| TBARS | Thiobarbituric Acid Reactive Substances |
Author Contributions
Conceptualization, B.L.-P. and C.G.-C.; Methodology, L.M.B.; Formal Analysis, R.Q.-F. and B.L.-P.; Investigation, B.L.-P., L.M.B., C.G.-C. and R.Q.-F.; Resources, S.P.-M.; Data Curation, R.Q.-F.; Writing—Original Draft Preparation, R.Q.-F. and B.L.-P.; Writing—Review and Editing, B.L.-P., L.M.B., C.G.-C. and R.Q.-F.; Visualization, S.P.-M.; Supervision, C.G.-C. and B.L.-P.; Project Administration, B.L.-P., L.M.B. and C.G.-C.
Funding
This study was supported by the Probelte Pharma S.A. group through the HENUFOOD project (CEN-20101016) from the CENIT program of Economy and Competitiveness Ministry of Spain (MINECO).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- 1.WHO . Global Status Report on Noncommunicable Diseases 2010. WHO; Geneva, Switzerland: 2011. (The World Health Organization Technical Report Series). [Google Scholar]
- 2.WHO . Global Atlas on Cardiovascular Disease Prevention and Control. WHO; Geneva, Switzerland: 2011. The World Health Organization Press in Collaboration with the World Heart Federation and the World Stroke Organization. [Google Scholar]
- 3.Mathers C.D., Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3:e442. doi: 10.1371/journal.pmed.0030442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.WHO . 2008–2013 Action Plan for the Global Strategy for the Prevention and Control of Noncommunicable Diseases. WHO; Geneva, Switzerland: 2009. (The World Health Organization Technical Report Series). [Google Scholar]
- 5.Salisbury D., Bronas U. Inflammation and immune system contribution to the etiology of atherosclerosis: Mechanisms and methods of assessment. Nurs. Res. 2014;63:375–385. doi: 10.1097/NNR.0000000000000053. [DOI] [PubMed] [Google Scholar]
- 6.Brown D.I., Griendling K.K. Regulation of signal transduction by reactive oxygen species in the cardiovascular system. Circ. Res. 2015;116:531–549. doi: 10.1161/CIRCRESAHA.116.303584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bonetti P.O., Lerman L.O., Lerman A. Endothelial dysfunction: A marker of atherosclerotic risk. Arterioscler. Thromb. Vasc. Biol. 2003;23:168–175. doi: 10.1161/01.ATV.0000051384.43104.FC. [DOI] [PubMed] [Google Scholar]
- 8.Gutierrez E., Flammer A.J., Lerman L.O., Elizaga J., Lerman A., Fernandez-Aviles F. Endothelial dysfunction over the course of coronary artery disease. Eur. Heart J. 2013;34:3175–3181. doi: 10.1093/eurheartj/eht351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ras R.T., Streppel M.T., Draijer R., Zock P.L. Flow-mediated dilation and cardiovascular risk prediction: A systematic review with meta-analysis. Int. J. Cardiol. 2013;168:344–351. doi: 10.1016/j.ijcard.2012.09.047. [DOI] [PubMed] [Google Scholar]
- 10.Xu Y., Arora R.C., Hiebert B.M., Lerner B., Szwajcer A., McDonald K., Rigatto C., Komenda P., Sood M.M., Tangri N. Non-invasive endothelial function testing and the risk of adverse outcomes: A systematic review and meta-analysis. Eur. Heart J. Cardiovasc. Imaging. 2014;15:736–746. doi: 10.1093/ehjci/jet256. [DOI] [PubMed] [Google Scholar]
- 11.Shechter M., Issachar A., Marai I., Koren-Morag N., Freinark D., Shahar Y., Shechter A., Feinberg M.S. Long-term association of brachial artery flow-mediated vasodilation and cardiovascular events in middle-aged subjects with no apparent heart disease. Int. J. Cardiol. 2009;134:52–58. doi: 10.1016/j.ijcard.2008.01.021. [DOI] [PubMed] [Google Scholar]
- 12.Shechter M., Shechter A., Koren-Morag N., Feinberg M.S., Hiersch L. Usefulness of brachial artery flow-mediated dilation to predict long-term cardiovascular events in subjects without heart disease. Am. J. Cardiol. 2014;113:162–167. doi: 10.1016/j.amjcard.2013.08.051. [DOI] [PubMed] [Google Scholar]
- 13.Corrado E., Rizzo M., Coppola G., Muratori I., Carella M., Novo S. Endothelial dysfunction and carotid lesions are strong predictors of clinical events in patients with early stages of atherosclerosis: A 24-month follow-up study. Coron. Artery Dis. 2008;19:139–144. doi: 10.1097/MCA.0b013e3282f3fbde. [DOI] [PubMed] [Google Scholar]
- 14.De Marco M., de Simone G., Roman M.J., Chinali M., Lee E.T., Russell M., Howard B.V., Devereux R.B. Cardiovascular and metabolic predictors of progression of prehypertension into hypertension: The strong heart study. Hypertension. 2009;54:974–980. doi: 10.1161/HYPERTENSIONAHA.109.129031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vasan R.S., Larson M.G., Leip E.P., Kannel W.B., Levy D. Assessment of frequency of progression to hypertension in non-hypertensive participants in the Framingham heart study: A cohort study. Lancet. 2001;358:1682–1686. doi: 10.1016/S0140-6736(01)06710-1. [DOI] [PubMed] [Google Scholar]
- 16.Wang S., Wu H., Zhang Q., Xu J., Fan Y. Impact of baseline prehypertension on cardiovascular events and all-cause mortality in the general population: A meta-analysis of prospective cohort studies. Int. J. Cardiol. 2013;168:4857–4860. doi: 10.1016/j.ijcard.2013.07.063. [DOI] [PubMed] [Google Scholar]
- 17.Chobanian A.V., Bakris G.L., Black H.R., Cushman W.C., Green L.A., Izzo J.L., Jr., Jones D.W., Materson B.J., Oparil S., Wright J.T., Jr., et al. The seventh report of the joint National committee on prevention, detection, evaluation, and treatment of high blood pressure: The JNC 7 report. JAMA. 2003;289:2560–2572. doi: 10.1001/jama.289.19.2560. [DOI] [PubMed] [Google Scholar]
- 18.Ingelsson E., Gona P., Larson M.G., Lloyd-Jones D.M., Kannel W.B., Vasan R.S., Levy D. Altered blood pressure progression in the community and its relation to clinical events. Arch. Intern. Med. 2008;168:1450–1457. doi: 10.1001/archinte.168.13.1450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Perry H.M., Jr., Davis B.R., Price T.R., Applegate W.B., Fields W.S., Guralnik J.M., Kuller L., Pressel S., Stamler J., Probstfield J.L. Effect of treating isolated systolic hypertension on the risk of developing various types and subtypes of stroke: The Systolic Hypertension in the Elderly Program (SHEP) JAMA. 2000;284:465–471. doi: 10.1001/jama.284.4.465. [DOI] [PubMed] [Google Scholar]
- 20.Griendling K.K., FitzGerald G.A. Oxidative stress and cardiovascular injury: Part I: Basic mechanisms and in vivo monitoring of ROS. Circulation. 2003;108:1912–1916. doi: 10.1161/01.CIR.0000093660.86242.BB. [DOI] [PubMed] [Google Scholar]
- 21.Griendling K.K., FitzGerald G.A. Oxidative stress and cardiovascular injury: Part II: Animal and human studies. Circulation. 2003;108:2034–2040. doi: 10.1161/01.CIR.0000093661.90582.c4. [DOI] [PubMed] [Google Scholar]
- 22.Trpkovic A., Resanovic I., Stanimirovic J., Radak D., Mousa S.A., Cenic-Milosevic D., Jevremovic D., Isenovic E.R. Oxidized low-density lipoprotein as a biomarker of cardiovascular diseases. Crit. Rev. Clin. Lab. Sci. 2015;52:70–85. doi: 10.3109/10408363.2014.992063. [DOI] [PubMed] [Google Scholar]
- 23.Maiolino G., Rossitto G., Caielli P., Bisogni V., Rossi G.P., Calo L.A. The role of oxidized low-density lipoproteins in atherosclerosis: The myths and the facts. Mediat. Inflamm. 2013;2013:714653. doi: 10.1155/2013/714653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huang H., Ma R., Liu D., Liu C., Ma Y., Mai W., Dong Y. Oxidized low-density lipoprotein cholesterol and the ratio in the diagnosis and evaluation of therapeutic effect in patients with coronary artery disease. Dis. Mark. 2012;33:295–302. doi: 10.1155/2012/167143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Koenig W., Karakas M., Zierer A., Herder C., Baumert J., Meisinger C., Thorand B. Oxidized LDL and the risk of coronary heart disease: Results from the MONICA/KORA Augsburg study. Clin. Chem. 2011;57:1196–1200. doi: 10.1373/clinchem.2011.165134. [DOI] [PubMed] [Google Scholar]
- 26.Toshima S., Hasegawa A., Kurabayashi M., Itabe H., Takano T., Sugano J., Shimamura K., Kimura J., Michishita I., Suzuki T., et al. Circulating oxidized low density lipoprotein levels. A biochemical risk marker for coronary heart disease. Arterioscler. Thromb. Vasc. Biol. 2000;20:2243–2247. doi: 10.1161/01.ATV.20.10.2243. [DOI] [PubMed] [Google Scholar]
- 27.Tsutsui T., Tsutamoto T., Wada A., Maeda K., Mabuchi N., Hayashi M., Ohnishi M., Kinoshita M. Plasma oxidized low-density lipoprotein as a prognostic predictor in patients with chronic congestive heart failure. J. Am. Coll. Cardiol. 2002;39:957–962. doi: 10.1016/S0735-1097(02)01721-7. [DOI] [PubMed] [Google Scholar]
- 28.Nishi K., Itabe H., Uno M., Kitazato K.T., Horiguchi H., Shinno K., Nagahiro S. Oxidized LDL in carotid plaques and plasma associates with plaque instability. Arterioscler. Thromb. Vasc. Biol. 2002;22:1649–1654. doi: 10.1161/01.ATV.0000033829.14012.18. [DOI] [PubMed] [Google Scholar]
- 29.Ehara S., Ueda M., Naruko T., Haze K., Itoh A., Otsuka M., Komatsu R., Matsuo T., Itabe H., Takano T., et al. Elevated levels of oxidized low density lipoprotein show a positive relationship with the severity of acute coronary syndromes. Circulation. 2001;103:1955–1960. doi: 10.1161/01.CIR.103.15.1955. [DOI] [PubMed] [Google Scholar]
- 30.Yamashita H., Ehara S., Yoshiyama M., Naruko T., Haze K., Shirai N., Sugama Y., Ikura Y., Ohsawa M., Itabe H., et al. Elevated plasma levels of oxidized low-density lipoprotein relate to the presence of angiographically detected complex and thrombotic coronary artery lesion morphology in patients with unstable angina. Circ. J. 2007;71:681–687. doi: 10.1253/circj.71.681. [DOI] [PubMed] [Google Scholar]
- 31.Gradinaru D., Borsa C., Ionescu C., Prada G.I. Oxidized LDL and NO synthesis—Biomarkers of endothelial dysfunction and ageing. Mech. Ageing Dev. 2015;151:101–113. doi: 10.1016/j.mad.2015.03.003. [DOI] [PubMed] [Google Scholar]
- 32.Ma F.X., Zhou B., Chen Z., Ren Q., Lu S.H., Sawamura T., Han Z.C. Oxidized low density lipoprotein impairs endothelial progenitor cells by regulation of endothelial nitric oxide synthase. J. Lipid Res. 2006;47:1227–1237. doi: 10.1194/jlr.M500507-JLR200. [DOI] [PubMed] [Google Scholar]
- 33.Matsumoto T., Takashima H., Ohira N., Tarutani Y., Yasuda Y., Yamane T., Matsuo S., Horie M. Plasma level of oxidized low-density lipoprotein is an independent determinant of coronary macrovasomotor and microvasomotor responses induced by bradykinin. J. Am. Coll. Cardiol. 2004;44:451–457. doi: 10.1016/j.jacc.2004.03.064. [DOI] [PubMed] [Google Scholar]
- 34.Bottino D.A., Lopes F.G., de Oliveira F.J., Ade S.M., Clapauch R., Bouskela E. Relationship between biomarkers of inflammation, oxidative stress and endothelial/microcirculatory function in successful aging versus healthy youth: A transversal study. BMC Geriatr. 2015;15:41. doi: 10.1186/s12877-015-0044-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Meisinger C., Baumert J., Khuseyinova N., Loewel H., Koenig W. Plasma oxidized low-density lipoprotein, a strong predictor for acute coronary heart disease events in apparently healthy, middle-aged men from the general population. Circulation. 2005;112:651–657. doi: 10.1161/CIRCULATIONAHA.104.529297. [DOI] [PubMed] [Google Scholar]
- 36.Tresserra-Rimbau A., Rimm E.B., Medina-Remon A., Martinez-Gonzalez M.A., de la Torre R., Corella D., Salas-Salvado J., Gomez-Gracia E., Lapetra J., Aros F., et al. Inverse association between habitual polyphenol intake and incidence of cardiovascular events in the PREDIMED study. Nutr. Metab. Cardiovasc. Dis. 2014;24:639–647. doi: 10.1016/j.numecd.2013.12.014. [DOI] [PubMed] [Google Scholar]
- 37.Szulińska M., Skrypnik D., Michałowska J., Bogdański P. Non-pharmacological modification of endothelial function: An important lesson for clinical practice. Postepy. Hig. Med. Dosw. 2018;72:89–100. doi: 10.5604/01.3001.0011.5963. [DOI] [Google Scholar]
- 38.Cicero A.F.G., Caliceti C., Fogacci F., Giovannini M., Calabria D., Colletti A., Veronesi M., Roda A., Borghi C. Effect of apple polyphenols on vascular oxidative stress and endothelium function: A translational study. Mol. Nutr. Food. Res. 2017;61:11. doi: 10.1002/mnfr.201700373. [DOI] [PubMed] [Google Scholar]
- 39.Medina-Remon A., Tresserra-Rimbau A., Pons A., Tur J.A., Martorell M., Ros E., Buil-Cosiales P., Sacanella E., Covas M.I., Corella D., et al. Effects of total dietary polyphenols on plasma nitric oxide and blood pressure in a high cardiovascular risk cohort. The PREDIMED randomized trial. Nutr. Metab. Cardiovasc. Dis. 2015;25:60–67. doi: 10.1016/j.numecd.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 40.Covas M.I., Nyyssonen K., Poulsen H.E., Kaikkonen J., Zunft H.J., Kiesewetter H., Gaddi A., de la Torre R., Mursu J., Baumler H., et al. The effect of polyphenols in olive oil on heart disease risk factors: A randomized trial. Ann. Intern. Med. 2006;145:333–341. doi: 10.7326/0003-4819-145-5-200609050-00006. [DOI] [PubMed] [Google Scholar]
- 41.Valls R.M., Farras M., Suarez M., Fernandez-Castillejo S., Fito M., Konstantinidou V., Fuentes F., Lopez-Miranda J., Giralt M., Covas M.I., et al. Effects of functional olive oil enriched with its own phenolic compounds on endothelial function in hypertensive patients. A randomised controlled trial. Food Chem. 2015;167:30–35. doi: 10.1016/j.foodchem.2014.06.107. [DOI] [PubMed] [Google Scholar]
- 42.Tresserra-Rimbau A., Rimm E.B., Medina-Remon A., Martinez-Gonzalez M.A., Lopez-Sabater M.C., Covas M.I., Corella D., Salas-Salvado J., Gomez-Gracia E., Lapetra J., et al. Polyphenol intake and mortality risk: A re-analysis of the PREDIMED trial. BMC Med. 2014;12:77. doi: 10.1186/1741-7015-12-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bulotta S., Celano M., Lepore S.M., Montalcini T., Pujia A., Russo D. Beneficial effects of the olive oil phenolic components oleuropein and hydroxytyrosol: Focus on protection against cardiovascular and metabolic diseases. J. Transl. Med. 2014;12:219. doi: 10.1186/s12967-014-0219-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.EFSA Scientific opinion on the substantiation of health claims related to quercetin and protection of DNA, proteins and lipids from oxidative damage. EFSA J. 2011;9:1–25. doi: 10.2903/j.efsa.2011.2067. [DOI] [Google Scholar]
- 45.45 Zrelli H., Matsuoka M., Kitazaki S., Zarrouk M., Miyazaki H. Hydroxytyrosol reduces intracellular reactive oxygen species levels in vascular endothelial cells by upregulating catalase expression through the AMPK-FOXO3a pathway. Eur. J. Pharmacol. 2011;660:275–282. doi: 10.1016/j.ejphar.2011.03.045. [DOI] [PubMed] [Google Scholar]
- 46.de Nigris F., Williams-Ignarro S., Sica V., Lerman L.O., D’Armiento F.P., Byrns R.E., Casamassimi A., Carpentiero D., Schiano C., Sumi D., et al. Effects of a pomegranate fruit extract rich in punicalagin on oxidation-sensitive genes and eNOS activity at sites of perturbed shear stress and atherogenesis. Cardiovasc. Res. 2007;73:414–423. doi: 10.1016/j.cardiores.2006.08.021. [DOI] [PubMed] [Google Scholar]
- 47.Vilahur G., Padro T., Casani L., Mendieta G., Lopez J.A., Streitenberger S., Badimon L. Polyphenol-enriched diet prevents coronary endothelial dysfunction by activating the Akt/eNOS pathway. Rev. Esp. Cardiol. (Engl. Ed.) 2015;68:216–225. doi: 10.1016/j.recesp.2014.03.023. [DOI] [PubMed] [Google Scholar]
- 48.Puri K.S., Suresh K.R., Gogtay N.J., Thatte U.M. Declaration of Helsinki, 2008: Implications for stakeholders in research. J. Postgrad. Med. 2009;55:131–134. doi: 10.4103/0022-3859.52846. [DOI] [PubMed] [Google Scholar]
- 49.Blekas G., Vassilakis C., Harizanis C., Tsimidou M., Boskou D.G. Biophenols in table olives. J. Agric. Food Chem. 2002;50:3688–3692. doi: 10.1021/jf0115138. [DOI] [PubMed] [Google Scholar]
- 50.Brenes M., Garcia A., Garcia P., Rios J.J., Garrido A. Phenolic compounds in Spanish olive oils. J. Agric. Food Chem. 1999;47:3535–3540. doi: 10.1021/jf990009o. [DOI] [PubMed] [Google Scholar]
- 51.Gil M.I., Tomas-Barberan F.A., Hess-Pierce B., Holcroft D.M., Kader A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000;48:4581–4589. doi: 10.1021/jf000404a. [DOI] [PubMed] [Google Scholar]
- 52.Ortega R.M., Requejo A.M., López-Sobaler A.M. Models of questionnaires for dietary studies, in the assessment of nutritional status. In: Ortega R.M., Requejo A.M., editors. Nutriguía Manual of Clinical Nu-trition in Primary Care. Complutense; Madrid, Spain: 2006. pp. 456–467. [Google Scholar]
- 53.WHO/FAO . Methodology of Nutritional Surveillance. Report of a Joint FAO/UNICEF/WHO Expert Committee. WHO/FAO; Geneva, Switzerland: 1976. (The World Health Organization Technical Report Series). [PubMed] [Google Scholar]
- 54.Corretti M.C., Anderson T.J., Benjamin E.J., Celermajer D., Charbonneau F., Creager M.A., Deanfield J., Drexler H., Gerhard-Herman M., Herrington D., et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: A report of the International brachial artery reactivity task force. J. Am. Coll. Cardiol. 2002;39:257–265. doi: 10.1016/S0735-1097(01)01746-6. [DOI] [PubMed] [Google Scholar]
- 55.Kuvin J.T., Patel A.R., Sliney K.A., Pandian N.G., Rand W.M., Udelson J.E., Karas R.H. Peripheral vascular endothelial function testing as a noninvasive indicator of coronary artery disease. J. Am. Coll. Cardiol. 2001;38:1843–1849. doi: 10.1016/S0735-1097(01)01657-6. [DOI] [PubMed] [Google Scholar]
- 56.Yagi K. Simple assay for the level of total lipid peroxides in serum or plasma. Methods Mol. Biol. 1998;108:101–106. doi: 10.1385/0-89603-472-0:101. [DOI] [PubMed] [Google Scholar]
- 57.Benzie I.F., Strain J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996;239:70–76. doi: 10.1006/abio.1996.0292. [DOI] [PubMed] [Google Scholar]
- 58.Ricart-Jane D., Llobera M., Lopez-Tejero M.D. Anticoagulants and other preanalytical factors interfere in plasma nitrate/nitrite quantification by the Griess method. Nitric Oxide. 2002;6:178–185. doi: 10.1006/niox.2001.0392. [DOI] [PubMed] [Google Scholar]
- 59.Sorensen K.E., Celermajer D.S., Spiegelhalter D.J., Georgakopoulos D., Robinson J., Thomas O., Deanfield J.E. Non-invasive measurement of human endothelium dependent arterial responses: Accuracy and reproducibility. Br. Heart J. 1995;74:247–253. doi: 10.1136/hrt.74.3.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.de Bock M., Derraik J.G., Brennan C.M., Biggs J.B., Morgan P.E., Hodgkinson S.C., Hofman P.L., Cutfield W.S. Olive (Olea europaea L.) leaf polyphenols improve insulin sensitivity in middle-aged overweight men: A randomized, placebo-controlled, crossover trial. PLoS ONE. 2013;8:e57622. doi: 10.1371/journal.pone.0057622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Achmon Y., Fishman A. The antioxidant hydroxytyrosol: Biotechnological production challenges and opportunities. Appl. Microbiol. Biotechnol. 2015;99:1119–1130. doi: 10.1007/s00253-014-6310-6. [DOI] [PubMed] [Google Scholar]
- 62.Stockton A., Farhat G., McDougall G.J., Al-Dujaili E.A.S. Effect of pomegranate extract on blood pressure and anthropometry in adults: A double-blind placebo-controlled randomised clinical trial. J. Nutr. Sci. 2017;6:e39. doi: 10.1017/jns.2017.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Graham I., Atar D., Borch-Johnsen K., Boysen G., Burell G., Cifkova R., Dallongeville J., De Backer G., Ebrahim S., Gjelsvik B., et al. European guidelines on cardiovascular disease prevention in clinical practice: Executive summary. Fourth joint task force of the European society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts) Eur. J. Cardiovasc. Prev. Rehabil. 2007;14(Suppl. 2):E1–E40. doi: 10.1097/01.hjr.0000277984.31558.c4. [DOI] [PubMed] [Google Scholar]
- 64.Moreno-Luna R., Munoz-Hernandez R., Miranda M.L., Costa A.F., Jimenez-Jimenez L., Vallejo-Vaz A.J., Muriana F.J., Villar J., Stiefel P. Olive oil polyphenols decrease blood pressure and improve endothelial function in young women with mild hypertension. Am. J. Hypertens. 2012;25:1299–1304. doi: 10.1038/ajh.2012.128. [DOI] [PubMed] [Google Scholar]
- 65.Whitworth J.A. 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J. Hypertens. 2003;21:1983–1992. doi: 10.1097/01.hjh.0000084751.37215.d2. [DOI] [PubMed] [Google Scholar]
- 66.Thomopoulos C., Parati G., Zanchetti A. Effects of blood-pressure-lowering treatment in hypertension: 9. Discontinuations for adverse events attributed to different classes of antihypertensive drugs: Meta-analyses of randomized trials. J. Hypertens. 2016;34:1921–1932. doi: 10.1097/HJH.0000000000001052. [DOI] [PubMed] [Google Scholar]
- 67.Aviram M., Rosenblat M., Gaitini D., Nitecki S., Hoffman A., Dornfeld L., Volkova N., Presser D., Attias J., Liker H., et al. Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clin. Nutr. 2004;23:423–433. doi: 10.1016/j.clnu.2003.10.002. [DOI] [PubMed] [Google Scholar]
- 68.EMA . Assessment Report on Olea Europaea L., Folium. The European Medicines Agency Science Medicines Health; London, UK: 2011. [Google Scholar]
- 69.Marrugat J., Covas M.I., Fito M., Schroder H., Miro-Casas E., Gimeno E., Lopez-Sabater M.C., de la Torre R., Farre M. Effects of differing phenolic content in dietary olive oils on lipids and LDL oxidation—A randomized controlled trial. Eur. J. Nutr. 2004;43:140–147. doi: 10.1007/s00394-004-0452-8. [DOI] [PubMed] [Google Scholar]
- 70.Aviram M., Dornfeld L., Kaplan M., Coleman R., Gaitini D., Nitecki S., Hofman A., Rosenblat M., Volkova N., Presser D., et al. Pomegranate juice flavonoids inhibit low-density lipoprotein oxidation and cardiovascular diseases: Studies in atherosclerotic mice and in humans. Drugs Exp. Clin. Res. 2002;28:49–62. doi: 10.1016/s0021-9150(00)80502-x. [DOI] [PubMed] [Google Scholar]
- 71.Rosenblat M., Hayek T., Aviram M. Anti-oxidative effects of pomegranate juice (PJ) consumption by diabetic patients on serum and on macrophages. Atherosclerosis. 2006;187:363–371. doi: 10.1016/j.atherosclerosis.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 72.Castaner O., Covas M.I., Khymenets O., Nyyssonen K., Konstantinidou V., Zunft H.F., de la Torre R., Munoz-Aguayo D., Vila J., Fito M. Protection of LDL from oxidation by olive oil polyphenols is associated with a downregulation of CD40-ligand expression and its downstream products in vivo in humans. Am. J. Clin. Nutr. 2012;95:1238–1244. doi: 10.3945/ajcn.111.029207. [DOI] [PubMed] [Google Scholar]
- 73.Tsuzura S., Ikeda Y., Suehiro T., Ota K., Osaki F., Arii K., Kumon Y., Hashimoto K. Correlation of plasma oxidized low-density lipoprotein levels to vascular complications and human serum paraoxonase in patients with type 2 diabetes. Metabolism. 2004;53:297–302. doi: 10.1016/j.metabol.2003.10.009. [DOI] [PubMed] [Google Scholar]
- 74.Kopprasch S., Pietzsch J., Kuhlisch E., Graessler J. Lack of association between serum paraoxonase 1 activities and increased oxidized low-density lipoprotein levels in impaired glucose tolerance and newly diagnosed diabetes mellitus. J. Clin. Endocrinol. Metab. 2003;88:1711–1716. doi: 10.1210/jc.2002-021561. [DOI] [PubMed] [Google Scholar]