Abstract
Transforming growth factor (TGF)-β1 is reported to be associated with the occurrence of atherosclerosis, although the mechanism remains unclear. Therefore, the present study aimed to investigate the involvement of TGF-β1 signaling in atherosclerosis. A total of 56 patients with atherosclerosis and 44 healthy volunteers were involved in this study. Serum expression of TGF-β1 and long non-coding RNA-ATB was detected by ELISA and quantitative polymerase chain reaction (qPCR). Receiver operating characteristic curve analysis was performed to analyze the diagnostic value of serum TGF-β1 and lncRNA-ATB for atherosclerosis. A human umbilical vein endothelial cell (HUVEC) line overexpressing lncRNA-ATB was constructed. The effects of TGF-β1 treatment and lncRNA-ATB overexpression on HUVEC cell proliferation and viability was detected with Cell Counting Kit-8 and MTT assays, respectively. Expression of TGF-β1 and pro-apoptotic Caspase-3 in lncRNA-ATB-overexpressing HUVECs was detected by western blotting. In addition, the expression of lncRNA-ATB in TGF-β1-treated HUVECs was detected by qPCR. It was demonstrated that serum TGF-β1 and lncRNA-ATB expression was significantly higher in atherosclerosis patients, compared with controls, and could be used to effectively distinguish patients from healthy individuals. TGF-β1 treatment and lncRNA-ATB overexpression reduced HUVEC viability and proliferation. TGF-β1 treatment increased the expression of lncRNA-ATB in HUVECs, while lncRNA-ATB overexpression had no significant effect on TGF-β1 expression. LncRNA-ATB silencing with small interfering RNA significantly reduced the effects of TGF-β1 treatment on the proliferation and viability of HUVECs. Furthermore, LncRNA-ATB overexpression upregulated the expression of caspase-3 in HUVECs. Therefore, it was concluded that TGF-β1 may have upregulated the expression of lncRNA-ATB to promote atherosclerosis, and lncRNA-ATB may serve as a potential therapeutic target for atherosclerosis. However, the mechanism remains to be further investigated.
Keywords: atherosclerosis, transforming growth factor-β1, long non-coding RNA-ATB
Introduction
Atherosclerosis as a chronic inflammatory disease caused by the formation of plaques in arteries, causing narrowing and the consequent development of debilitating conditions, such as stroke, kidney problems, coronary artery disease and peripheral artery disease (1,2). Abnormal cholesterol content has been shown to be a major cause of atherosclerosis, although the exact mechanisms remain unclear (3). Risk factors include diabetes, high blood pressure, family history, obesity and an unhealthy diet (4,5). Typically, atherosclerosis does not have well-defined symptoms, and the majority of patients are diagnosed in advanced stages, leading to poor treatment outcomes (6). Therefore, early diagnosis is critical for the effective treatment of atherosclerosis and prevention of atherosclerosis-associated complications.
Long non-coding RNAs (lncRNAs) are a group of transcripts composed of >200 nucleotides, with no protein-coding ability (7). It has been well established that various lncRNAs participate in the development of human diseases, by inhibiting or promoting their progression (8). In effect, certain lncRNAs have been proven to be a potential therapeutic target for the treatment of human diseases. The development of atherosclerosis is also accompanied by alterations in the lncRNA expression profile (9), indicating the involvement of certain lncRNAs in the pathogenesis of this disease. lncRNA-ATB has critical function in several types of human malignancies (10,11). Altered expression of lncRNA-ATB is closely correlated with the progression and prognosis of colon cancer (10). During the development of renal cell carcinoma, lncRNA-ATB is involved in tumor metastases possibly by promoting cancer cell migration and invasion (11). However, its involvement in atherosclerosis is unknown. The preliminary microarray analysis revealed that lncRNA-ATB was altered in atherosclerosis, indicating its potential involvement in this disease. Therefore, the present study aimed to investigate the functionality of lncRNA-ATB in atherosclerosis. The present study provided evidence of a novel biomarker for the diagnosis of atherosclerosis, and a novel target for the treatment of this disease.
Materials and methods
Subjects
Serum samples were collected from 56 patients with atherosclerosis (early stage) (12) and 44 healthy volunteers from the elbow vein in The Fourth People's Hospital of Jinan from January 2016 to January 2018 (Jinan, China). Patients with other severe diseases, such as cases of severe heart, lung and liver diseases, were not included in this study. All participants were willing to participate in this study. The 56 patients included 29 males and 27 females, with a mean age of 26.8±7.8 (range, 16–34). The 44 healthy volunteers included 24 males and 20 females, with a mean age of 25.7±9.1 (range, 18–32). No significant differences in age and gender were demonstrated between the patient group and healthy control group. The present study was approved by the Ethics Committee of The Fourth People's Hospital of Jinan (Jinan, China), and all patients provided written informed consent.
ELISA
Serum TGF-β1 was detected by ELISA using coating antibody (cat. no. MAB1835) and biotinylated detection antibody (cat. no. BAF240), obtained from R&D Systems, Inc. (Minneapolis, MN, USA). All operations were performed in strict accordance with manufacturer's protocol. Acid activation was performed to release biologically active TGF-β1, as described previously (13). Serum samples were diluted in Dulbecco's PBS (1:75) and added to the ELISA plate to measure the concentration of active TGF-β1.
Cell culture
Human umbilical vein endothelial cells [HUVECs; cat. no. CRL-1730; American Type Culture Collection (ATCC), Manassas, VA, USA] were cultured in F-12K medium (cat. no. 30-2004; ATCC) supplemented with 0.1 mg/ml heparin (cat. no. H3393; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany), endothelial cell growth supplement (1:100; cat. no. 354006; Corning Corporation, Corning, NY, USA) and 10% fetal bovine serum (cat. no. 30-2020; ATCC) at 37°C with 5% CO2. Cells were collected during the logarithmic growth phase for subsequent experiments.
Establishment of lncRNA-ATB overexpression and siRNA silencing in HUVECs
A EcoRI-EcoRI fragment containing full length lncRNA-ATB cDNA was inserted into pIRES2-EGFP vector (Clontech Laboratories, Inc., Mountainview, CA, USA) to establish a vector expressing lncRNA-ATB. Cells were cultured in the medium mentioned above at 37°C overnight to reach 70–80% confluence. Transfection was performed using Lipofectamine® 2000 (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) to transfect 10 nM vector, 50 nM lncRNA-ATB siRNA (5′-CCAUGAGGAGUACUGCCAATT-3′) or non-targeting control siRNA (5′-UUCUCCGAACGUGUCACGUtt-3′; Sangon, Biotech Co., Ltd., Shanghai, China) into 5×106 HUVECs. Cell suspensions were prepared by centrifuging cells at 1,000 × g for 10 min at room temperature to remove the supernatant, followed by the addition of fresh culture medium. Cells were collected 24 h following transfection for subsequent experiments. Control cells were treated with Lipofectamine® 2000 only. Cells transfected with empty vectors or non-targeting control siRNA were negative control cells.
MTT assay
Tetraethylammonium (10 mM; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added to cells (5×104 cells/ml) with lncRNA-ATB overexpression and siRNA silencing as well as control and negative control cells. Each well of a 96-well plate was filled with 100 µl cell suspension (5×103 cells/well), and cells were cultured at 37°C with 5% CO2 for 4 h, following which 10 µl MTT was added into each well for another 4 h at 37°C, DMSO was added to dissolve the formazan crystals, and the optical density was measured at 570 nm with a microplate reader.
Cell proliferation assay
Treated cells in a 100 µl suspension were added into each well of 96-well plates (4×104 cells/well). Cells were cultured in an incubator at 37°C with 5% CO2 and Cell Counting Kit-8 solution (10 µl) was added into each well 24, 48, 72 and 96 h later, and further cultured at 37°C for another 4 h. Optical density was measured at 450 nm with a microplate reader. In cases of TGF-β1 treatment, cells were treated with TGF-β1 (Sigma-Aldrich Merck KGaA) at a dose of 10 ng/ml for 1 h.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) from serum and treated cells and RNA samples were used to synthesize cDNA through reverse transcription. Reverse transcription was performed using SuperScript II Reverse Transcriptase kit (Thermo Fisher Scientific, Inc.) through the following conditions: 5 min at 25°C, 10 min at 55°C and 5 min at 80°C. Sequences of primers used in PCR reactions were: 5′-TCTGGCTGAGGCTGGTTGAC-3′ (forward) and 5′-ATCTCTGGGTGCTGGTGAAGG-3′ (reverse) for lncRNA-ATB; 5′-GACCTCTATGCCAACACAGT-3′ (forward) and 5′-AGTACTTGCGCTCAGGAGGA-3′ (reverse) for β-actin. SYBR® Green Quantitative RT-qPCR kit (Sigma-Aldrich; Merck KGaA) was used to prepare all PCR reaction systems. The PCR thermocycling conditions were: 95°C for 45 sec, followed by 40 cycles of 95°C for 10 sec and 60°C for 35 sec. Data were quantified using the 2−ΔΔCq method (14). Relative expression of lncRNA-ATB was normalized to β-actin.
Western blot analysis
Total protein was extracted from cells using radioimmunoprecipitation assay lysis buffer (Thermo Fisher Scientific, Inc.), and protein concentration was determined by a bicinchoninic acid protein assay. Proteins (20 µg/lane) were separated by 10% SDS-PAGE and transferred to polyvinylidene difluoride membranes. Membranes were blocked with 5% skimmed milk for 2 h at room temperature. Following three washes with PBS (10 min each time), membranes were incubated with primary antibodies including rabbit anti-TGF-β1 (1:2,000; cat. no. ab92486; Abcam, Cambridge, UK), anti-caspase-3 (1:1,500; cat. no. ab4051; Abcam) and anti-GAPDH (1:1,000; cat. no. ab9845; Abcam) overnight at 4°C. Membranes were washed again in PBS three times (10 min each time) and subsequently incubated with anti-rabbit horseradish peroxidase-conjugated secondary antibody (1:1,000; cat. no. MBS435036; MyBioSource, Inc., San Diego, CA, USA) at room temperature for 1.5 h. Following further washes as described above, bands were visualized with enhanced chemiluminescence reagent (Sigma-Aldrich; Merck KGaA). The relative expression of TGF-β1 was normalized to the endogenous control GAPDH using ImageJ v.148 software (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
GraphPad Prism 6 (GraphPad Software, Inc., La Jolla, CA, USA) and Origin v10 (OriginLab Corporation, Northampton, MA, USA) were used to perform statistical analyses. All experiments were performed in triplicate manner and data were expressed as mean ± standard deviation. Data comparisons between two groups were performed with the Student's t-test, and among multiple groups using one-way analysis of variance, followed by Tukey test. Chi-square test was used for the analysis of count data. ROC curve analysis was used to evaluate the diagnostic value of serum active TGF-β1 and lncRNA-ATB expression in atherosclerosis. P<0.05 was considered to indicate a statistically significant difference.
Results
TGF-β1 and lncRNA-ATB expression is increased in patients with atherosclerosis
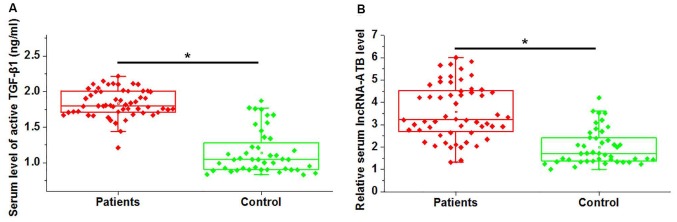
Serum expression of active TGF-β1 and lncRNA-ATB was measured in patients with atherosclerosis and healthy controls via ELISA and RT-qPCR, respectively. As presented in Fig. 1A, active TGF-β1 expression in serum samples was significantly higher in patients with atherosclerosis, compared with healthy controls (P<0.05). Similarly, significantly higher circulating lncRNA-ATB expression was detected in atherosclerosis patients, compared with healthy controls (P<0.05; Fig. 1B). It is of note that no significant differences in serum expression of total TGF-β1 were demonstrated between patient and control groups (data not shown). These data suggested that upregulation of TGF-β1 and lncRNA-ATB may be involved in the pathogenesis of atherosclerosis.
Figure 1.
Comparison of serum level of active TGF-β1 and lncRNA-ATB in patients with atherosclerosis and healthy controls. (A) Serum expression of active TGF-β1 and (B) lncRNA-ATB in patients with atherosclerosis and healthy individuals. *P<0.05. TGF-β1, transforming growth factor-β1; lncRNA, long non-coding RNA.
Serum expression of active TGF-β1 and lncRNA-ATB may be useful as diagnostic factors in atherosclerosis
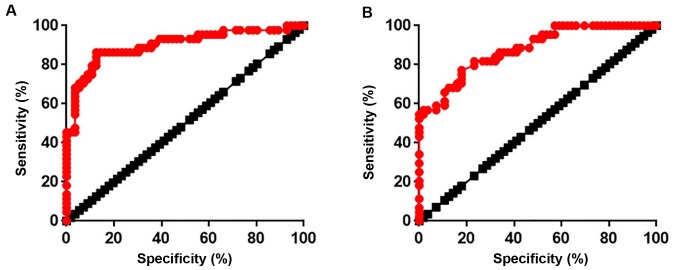
ROC curve analysis was used to evaluate the diagnostic value of serum active TGF-β1 and lncRNA-ATB expression in atherosclerosis. As presented in Fig. 2A, the area under the curve (AUC) for TGF-β1 was 0.9012 with a 95% confidence interval (CI) of 0.8364–0.9660 (P<0.0001). In addition, the AUC for lncRNA-ATB was 0.8787 with 95% CI of 0.8137–0.9436 (P<0.0001; Fig. 2B). These data indicated that TGF-β1 and lncRNA-ATB expression may be used to effectively distinguish patients with atherosclerosis from healthy controls.
Figure 2.
Diagnostic value of serum active TGF-β1 and lncRNA-ATB for atherosclerosis. (A) Receiver operating characteristic curve of serum active TGF-β1 and (B) lncRNA-ATB in atherosclerosis.
Effects of lncRNA-ATB overexpression and TGF-β1 treatment on the proliferation and viability of HUVECs
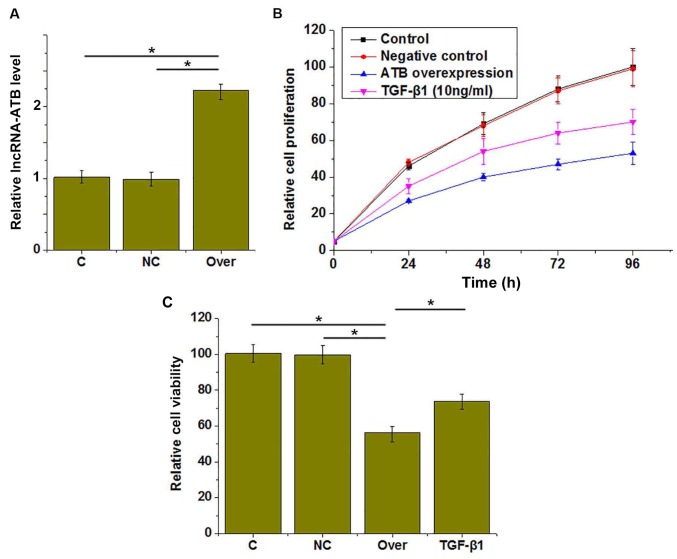
lncRNA-ATB overexpression was confirmed by RT-qPCR (Fig. 3A). CCK-8 assays were performed to investigate the effects of lncRNA-ATB overexpression on the proliferation and viability of HUVECs. The results revealed that proliferation was markedly inhibited (Fig. 3B) and viability was reduced (Fig. 3C) in HUVECs by lncRNA-ATB overexpression, compared with the control groups. In addition, treatment with TGF-β1 (10 ng/ml for 1 h) also inhibited the proliferation (Fig. 3B) and reduced the viability (Fig. 3C) of HUVECs compared with the control group.
Figure 3.
Effects of lncRNA-ATB overexpression and TGF-β1 treatment on the proliferation and viability of HUVECs. (A) lncRNA-ATB overexpression was successfully induced and the (B) proliferation and (C) viability of HUVECs under different conditions was assessed. *P<0.05. TGF-β1, transforming growth factor-β1; lncRNA, long non-coding RNA; HUVECs, human umbilical vascular endothelial cells; C, control; NC, negative control; Over, lncRNA-ATB overexpression.
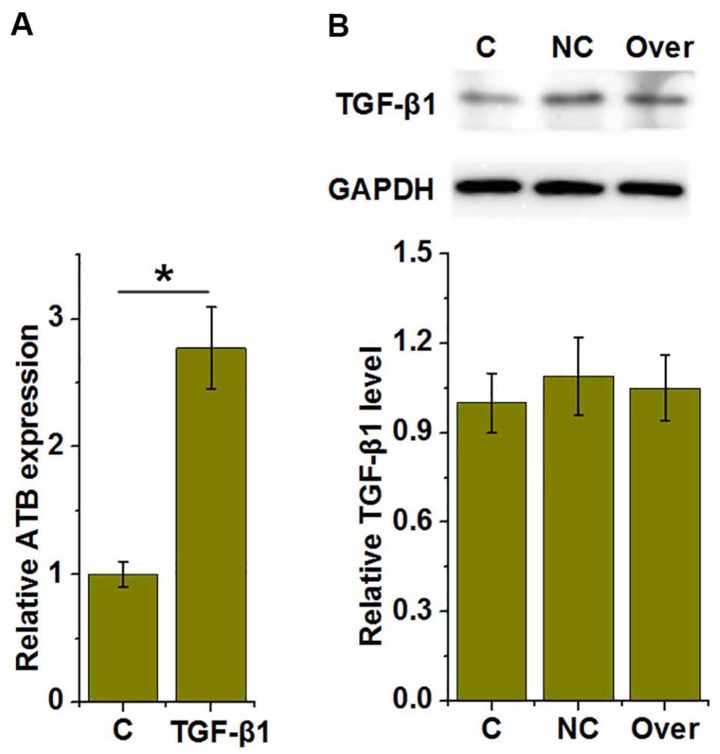
TGF-β1 is an upstream activator of lncRNA-ATB in HUVECs
As presented in Fig. 4A, treatment with TGF-β1 significantly increased the expression of lncRNA-ATB in HUVECs, compared with the control (P<0.05), whereas lncRNA-ATB overexpression had no significant effect on TGF-β1 expression (Fig. 4B), suggesting that TGF-β1 may be an upstream activator of lncRNA-ATB in HUVECs.
Figure 4.
TGF-β1 is an upstream activator of lncRNA-ATB in HUVECs. (A) Effects of TGF-β1 treatment on lncRNA-ATB and (B) the effect of lncRNA-ATB overexpression on TGF-β1 expression. *P<0.05. lncRNA, long non-coding RNA; HUVECs, human umbilical vascular endothelial cells; TGF-β1, transforming growth factor-β1; C, control; NC, negative control; Over, lncRNA-ATB overexpression.
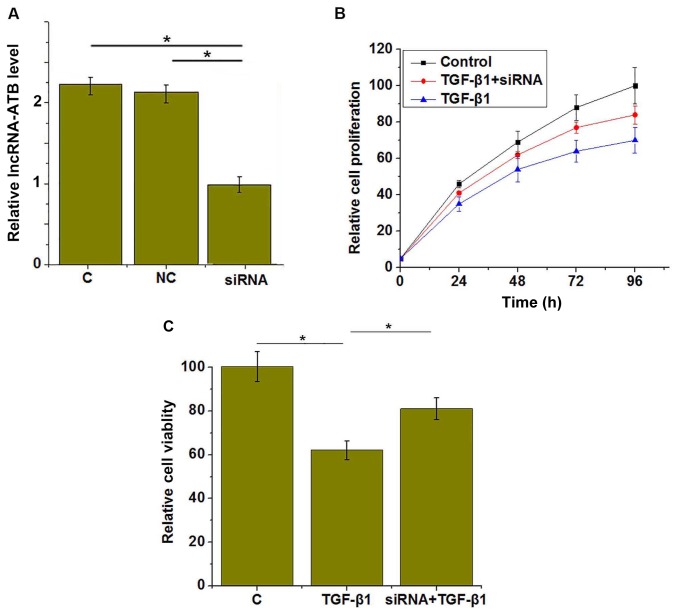
LncRNA-ATB siRNA inhibits the effects of TGF-β1 treatment on HUVEC proliferation and viability
The above data demonstrated that TGF-β1 was an upstream activator of lncRNA-ATB in HUVECs. To further investigate TGF-β1 signaling in HUVECs, lncRNA-ATB was silenced in HUVECs with siRNA, as confirmed by RT-qPCR (Fig. 5A). It was demonstrated that lncRNA-ATB siRNA significantly reduced the effects of TGF-β1 treatment on HUVEC proliferation (Fig. 5B) and viability (Fig. 5C).
Figure 5.
LncRNA-ATB siRNA silencing significantly reduced the effects of TGF-β1 treatment on proliferation and viability of HUVECs. (A) LncRNA-ATB silencing was successfully induced by siRNA, and the (B) proliferation and (C) viability of HUVECs under different conditions was assessed. *P<0.05. lncRNA, long non-coding RNA; HUVECs, human umbilical vascular endothelial cells; TGF-β1, transforming growth factor-β1; C, control; NC, negative control; siRNA, small interfering RNA.
LncRNA-ATB overexpression upregulates the expression of caspase-3
To investigate the effects of lncRNA-ATB on HUVEC apoptosis, the expression of pro-apoptotic protein caspase-3 was detected by western blotting. As presented in Fig. 6, lncRNA-ATB overexpression significantly upregulated the expression of pro-apoptotic caspase-3 in HUVECs (P<0.05).
Figure 6.
Long non-coding RNA-ATB overexpression upregulates the expression of proapoptotic caspase-3 in human umbilical vascular endothelial cells. Western blot analysis was performed to detect the expression of caspase-3. *P<0.05. C, control; NC, negative control; Over, long non-coding RNA-ATB overexpression.
Discussion
Although the pathogenesis of atherosclerosis has not been fully elucidated, the involvement of lncRNAs in the development and progression of the disease has been studied extensively. LncRNA-H19 has been reported to be significantly upregulated in atherosclerosis, and lncRNA-H19 overexpression promotes the progression of this disease through the activation of mitogen-activated protein kinase and nuclear factor-kB pathways (15). In addition, the expression of lncRNA-RNCR3 is significantly increased in human and mouse aortic atherosclerotic lesions, and is associated with endothelial cell dysfunction and vascular smooth muscle cell proliferation inhibition (16). Furthermore, lncRNA-TUG1 overexpression may be a promising target for the treatment of atherosclerosis (17). lncRNA-ATB is upregulated in several types of human malignancies (10,11). In the present study, serum lncRNA-ATB expression was significantly higher in patients with atherosclerosis, compared with healthy controls, indicating that upregulation of lncRNA-ATB may be involved in this disease. TGF-β1 signaling has critical function in the development and progression of atherosclerosis (18). In the present study, serum expression levels of active TGF-β1 were significantly increased in atherosclerosis patients compared with healthy controls, further confirming the involvement of TGF-β1 signaling in atherosclerosis.
Early diagnosis and treatment of atherosclerosis is critical, but this is hindered by the lack of classic symptoms (6). In the present study, all atherosclerosis patients included were in the early stage of disease, and ROC curve analysis demonstrated that serum active TGF-β1 and lncRNA-ATB may be used to active distinguish patients with atherosclerosis from healthy controls, indicating that serum active TGF-β1 and lncRNA-ATB may serve as promising biomarkers for the early diagnosis of atherosclerosis. It is of note that no significant differences in serum expression of total TGF-β1 between patient and control groups (data not shown), and ROC curve analysis suggested that serum total TGF-β1 may not be an effective diagnostic biomarker for atherosclerosis. Therefore, serum active TGF-β1 should be used.
TGF-β1 and lncRNA-ATB serve regulatory roles in the proliferation and apoptosis of various cell types. TGF-β1 has both antiproliferative and apoptotic effects in bovine mammary epithelial BME-UV1 cells (19) and the proapoptotic effect of TGF-β1 on cultured HUVECs has also been demonstrated (20); whereas lncRNA-ATB promotes papillary thyroid tumor growth (21). It is well-established that EC apoptosis serves a pivotal role in the pathogenesis of atherosclerosis (22). In the present study, lncRNA-ATB overexpression and TGF-β1 treatment inhibited the proliferation and reduced the viability of HUVECs, and promoted the expression of proapoptotic caspase-3, indicating the involvement of TGF-β1 signaling and lncRNA-ATB in the regulation of proliferation and apoptosis of key cells involved the pathogenesis of atherosclerosis. It has been reported that TGF-β signaling in hepatocellular carcinoma upregulates the expression of lncRNA-ATB to promote cancer progression (23). In the present study, TGF-β1 was demonstrated to upregulate lncRNA-ATB expression. Furthermore, lncRNA-ATB silencing significantly reduced the effect of TGF-β1 on the proliferation and viability of HUVECs. This suggested that lncRNA-ATB was upregulated by TGF-β1 to participate in the pathogenesis of atherosclerosis through apoptosis promotion and proliferation inhibition in HUVECs.
In conclusion, TGF-β1 and lncRNA-ATB expression was upregulated in patients with atherosclerosis, and increased expression levels of TGF-β1 and lncRNA-ATB may be used to effectively distinguish atherosclerosis patients from normal healthy individuals. Upregulation of lncRNA-ATB by TGF-β1 inhibited the proliferation and reduced the viability of HUVECs. The present study was challenged by a small sample size, and further studies with a large sample size are required to further confirm the conclusions. However, the present study failed to construct an atherosclerosis model using HUVECs due to the limited resources. Only cell proliferation and apoptosis, but not other cell behaviors were investigated. This study also failed to detect other inflammatory markers, such as C-reactive protein, P-selectin and TNF-α. Further studies are still needed.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
HY and CZ designed experiments. HY and SM performed experiments. LS and JG analyzed data. CZ wrote the paper. All autors reviewed and approved the paper.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of The Fourth People's Hospital of Jinan (Jinan, China), and all patients provided written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Li H, Horke S, Förstermann U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis. 2014;237:208–219. doi: 10.1016/j.atherosclerosis.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 2.Blankenberg S, Barbaux S, Tiret L. Adhesion molecules and atherosclerosis. Atherosclerosis. 2003;170:191–203. doi: 10.1016/S0021-9150(03)00097-2. [DOI] [PubMed] [Google Scholar]
- 3.Steinberg D. In celebration of the 100th anniversary of the lipid hypothesis of atherosclerosis. J Lipid Res. 2013;54:2946–2949. doi: 10.1194/jlr.R043414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Parrinello CM, Rastegar I, Godino JG, Miedema MD, Matsushita K, Selvin E. Prevalence of and racial disparities in risk factor control in older adults with diabetes: The Atherosclerosis Risk in Communities Study. Diabetes Care. 2015;38:1290–1298. doi: 10.2337/dc15-0016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McEvoy JW, Blaha MJ, DeFilippis AP, Lima JA, Bluemke DA, Hundley WG, Min JK, Shaw LJ, Lloyd-Jones DM, Barr RG, et al. Cigarette smoking and cardiovascular events: Role of inflammation and subclinical atherosclerosis: The multiethnic study of atherosclerosis. Arterioscler Thromb Vasc Biol. 2015;35:700–709. doi: 10.1161/ATVBAHA.114.304960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kubota Y, London SJ, Cushman M, Chamberlain AM, Rosamond WD, Heckbert SR, Zakai N, Folsom AR. Lung function, respiratory symptoms and venous thromboembolism risk: The atherosclerosis risk in communities study. J Thromb Haemost. 2016;14:2394–2401. doi: 10.1111/jth.13525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mattick JS, Makunin IV. Non-coding RNA. Hum Mol Genet. 2006;15:R17–R29. doi: 10.1093/hmg/ddl046. Spec No 1. [DOI] [PubMed] [Google Scholar]
- 8.Lalevée S, Feil R. Long noncoding RNAs in human disease: Emerging mechanisms and therapeutic strategies. Epigenomics. 2015;7:877–879. doi: 10.2217/epi.15.55. [DOI] [PubMed] [Google Scholar]
- 9.Meng F, Yan J, Ma Q, Jiao Y, Han L, Xu J, Yang F, Liu J. Expression status and clinical significance of lncRNA APPAT in the progression of atherosclerosis. PeerJ. 2018;6:e4246. doi: 10.7717/peerj.4246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iguchi T, Uchi R, Nambara S, Saito T, Komatsu H, Hirata H, Ueda M, Sakimura S, Takano Y, Kurashige J, et al. A long noncoding RNA, lncRNA-ATB, is involved in the progression and prognosis of colorectal cancer. Anticancer Res. 2015;35:1385–1388. [PubMed] [Google Scholar]
- 11.Xiong J, Liu Y, Jiang L, Zeng Y, Tang W. High expression of long non-coding RNA lncRNA-ATB is correlated with metastases and promotes cell migration and invasion in renal cell carcinoma. Jpn J Clin Oncol. 2016;46:378–384. doi: 10.1093/jjco/hyv214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schwartz CJ, Valente AJ, Sprague EA, Kelley JL, Cayatte AJ, Mowery J. Atherosclerosis: Potential targets for stabilization and regression. Circulation. 1992;86(Suppl 6):III117–III123. [PubMed] [Google Scholar]
- 13.Roberts AB, Sporn MB. Physiological actions and clinical applications of transforming growth factor-β (TGF-β) Growth Factors. 1993;8:1–9. doi: 10.3109/08977199309029129. [DOI] [PubMed] [Google Scholar]
- 14.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 15.Pan JX. LncRNA H19 promotes atherosclerosis by regulating MAPK and NF-kB signaling pathway. Eur Rev Med Pharmacol Sci. 2017;21:322–328. [PubMed] [Google Scholar]
- 16.Shan K, Jiang Q, Wang XQ, Wang YN, Yang H, Yao MD, Liu C, Li XM, Yao J, Liu B, et al. Role of long non-coding RNA-RNCR3 in atherosclerosis-related vascular dysfunction. Cell Death Dis. 2016;7:e2248. doi: 10.1038/cddis.2016.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen C, Cheng G, Yang X, Li C, Shi R, Zhao N. Tanshinol suppresses endothelial cells apoptosis in mice with atherosclerosis via lncRNA TUG1 up-regulating the expression of miR-26a. Am J Transl Res. 2016;8:2981–2991. [PMC free article] [PubMed] [Google Scholar]
- 18.Leonarduzzi G, Sevanian A, Sottero B, Arkan MC, Biasi F, Chiarpotto E, Basaga H, Poli G. Up-regulation of the fibrogenic cytokine TGF-β1 by oxysterols: A mechanistic link between cholesterol and atherosclerosis. FASEB J. 2001;15:1619–1621. doi: 10.1096/fj.00-0668fje. [DOI] [PubMed] [Google Scholar]
- 19.Kolek O, Gajkowska B, Godlewski MM, Motyl T. Antiproliferative and apoptotic effect of TGF-β1 in bovine mammary epithelial BME-UV1 cells. Comp Biochem Physiol C Toxicol Pharmacol. 2003;134:417–430. doi: 10.1016/S1532-0456(02)00249-1. [DOI] [PubMed] [Google Scholar]
- 20.Tsukada T, Eguchi K, Migita K, Kawabe Y, Kawakami A, Matsuoka N, Takashima H, Mizokami A, Nagataki S. Transforming growth factor β1 induces apoptotic cell death in cultured human umbilical vein endothelial cells with down-regulated expression of bcl-2. Biochem Biophys Res Commun. 1995;210:1076–1082. doi: 10.1006/bbrc.1995.1766. [DOI] [PubMed] [Google Scholar]
- 21.Fu XM, Guo W, Li N, Liu HZ, Liu J, Qiu SQ, Zhang Q, Wang LC, Li F, Li CL. The expression and function of long noncoding RNA lncRNA-ATB in papillary thyroid cancer. Eur Rev Med Pharmacol Sci. 2017;21:3239–3246. [PubMed] [Google Scholar]
- 22.Choy JC, Granville DJ, Hunt DW, McManus BM. Endothelial cell apoptosis: Biochemical characteristics and potential implications for atherosclerosis. J Mol Cell Cardiol. 2001;33:1673–1690. doi: 10.1006/jmcc.2001.1419. [DOI] [PubMed] [Google Scholar]
- 23.Yuan JH, Yang F, Wang F, Ma JZ, Guo YJ, Tao QF, Liu F, Pan W, Wang TT, Zhou CC, et al. A long noncoding RNA activated by TGF-β promotes the invasion-metastasis cascade in hepatocellular carcinoma. Cancer Cell. 2014;25:666–681. doi: 10.1016/j.ccr.2014.03.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.