Abstract
Background:
Primary amenorrhea is one of the most common disorders seen as gynecological problems in adolescent girls. It refers to the participants who did not attain menarche by the age of 11–15 years. Chromosome abnormalities contribute as one of the etiological factors in patients with primary amenorrhea.
Aims:
The aim of this study was to evaluate the frequency of chromosomal abnormalities and to investigate the abnormal karyotypes in patients referred with the symptom of primary amenorrhea for better management and counseling.
Setting and Design:
One hundred and seventy-four cases of primary amenorrhea were referred from the obstetrics and gynecology department to our cytogenetic laboratory for chromosomal analysis. G-banded chromosomes were karyotyped, and chromosomal analysis of all patients was done.
Results:
Out of 174 patients, we observed 23 (13.22%) participants with abnormal karyotype. In 23 cases of chromosomal abnormalities, 10 cases were sex reversal female (46,XY) and Turner karyotype (45,X) in 6 females. Other numerical and structural abnormalities were also seen such as 47,XXX; 45,X/47,XXX; 45,X/46, X,dic(X); 46,XX, inv (9); 45,X/46,X,i(Xq); 46,X,mar(X); and 45,X/46,XY in the primary amenorrhea cases.
Conclusion:
This study definitely attests the importance of chromosomal analysis in the etiologic diagnosis of primary amenorrhea patients. Karyotyping will help to counsel and manage the cases of primary amenorrhea in a better way. This study reveals the frequencies and different types of chromosomal abnormalities found in primary amenorrhea individuals and that might help to make the national database on primary amenorrhea in relation to chromosomal aberrations.
KEYWORDS: Chromosomal abnormalities, karyotyping, primary amenorrhea
INTRODUCTION
Primary amenorrhea is defined as the participants who did not attain spontaneous menarche by the age of 11–15 years with or without the normal development of secondary sexual characteristics.[1,2] Primary amenorrhea is only a symptom and not a disease entity.[3] It is one of the most common disorders seen as gynecological problems in adolescent girls.[4] The clinical findings of primary amenorrhea include the developmental impairments such as Mullerian agenesis, gonadal dysgenesis, streak gonad, vaginal atresia, hypoplastic uterus, and imperforate hymen.
The causes of primary amenorrhea can be categorized as the functional or anatomic defect of the hypothalamus, the functional or anatomic defect of the pituitary, the functional or anatomic defect of the uterus or ovaries, and genetic defect which may be either at chromosomal or at a gene level.[5,6] The genetic or chromosomal abnormality is the most important cause of primary amenorrhea, and the presence of chromosomal abnormality affects subsequent management.[7] It has been reported in different studies that the percentage of chromosomal aberration varies from 10% to 25% in patients with primary amenorrhea.[2]
A number of studies have been reported to ascertain the contribution of sex chromosome abnormalities in patients with primary amenorrhea. In primary amenorrhea cases, the sex chromosomal abnormalities may be of numerical and structural types. The sex chromosomal abnormalities may be numerical such as 45, X; 47, XXX or mosaic and structural with patients having abnormally small X chromosome due to deletions or abnormal large X chromosome, and some types of mosaicism of the X chromosome such as X/XXX and X/XX can also lead to primary amenorrhea.[7] Cytogenetic studies have shown different types of sex chromosome abnormal karyotype in primary amenorrhea females.[7,8,9,10,11,12,13,14,15] Cytogenetic investigations have confirmed the role of chromosomal abnormalities as a cause of amenorrhea. The wide variations in frequencies obtained are largely due to the degree of stringency in the selection of study population and small sample size studied in some surveys. The aim of our study was primarily to determine the frequency and types of chromosomal abnormalities among women with this disorder for better management and counseling.
MATERIALS AND METHODS
The study was carried out for the last 18 years (2000–2017) on patients with the symptom of primary amenorrhea. Patients were referred from the obstetrics and gynecology department to cytogenetic laboratory of our institute. Complete case history of patients was recorded. Heparinized peripheral blood was collected from the participants, and lymphocytes cultures were set up with whole blood sample. Lymphocyte cultures were set up according to our laboratory standardized technique,[16] i.e., adding 0.5-ml heparinized whole blood to 5 ml of RPMI-1640 medium (HiMedia, India) supplemented with 12.0% fetal calf serum (Biological Laboratories, Israel), 2.0% phytohemagglutinin M (Difco, USA), 100 units/ml penicillin, and 100 μg/ml streptomycin. All cultures were incubated in the presence of 5% carbon dioxide at 37°C with humidity of >90 for 72 h. One hour before harvesting aqueous colchicine (0.25 μg/ml) was added. Cultures were harvested following conventional process and flame-dried slides were prepared. Slides were preserved for Giemsa-Trypsin-Giemsa (GTG) banding and stained with Giemsa. Fifty well-spread metaphases were screened and analyzed to find the abnormality. The microphotography has been carried out with a Trinocular Research Microscope (Olympus, model CX 31) by a Digital Camera (Samsung, model SDC-312). Karyotype was prepared following the guidelines of the International System for Human Cytogenetic Nomenclature.[17]
RESULTS
Out of a total 174 primary amenorrhea cases studied, 151 (86.78%) cases were with normal karyotype and remaining 23 (13.22%) cases were with chromosomal abnormalities or abnormal karyotypes involving sex chromosomal numerical or structural changes. The most frequent abnormality seen among patients with primary amenorrhea was sex reversal female (46, XY) in 10 (5.74%) cases. The second most common abnormality found in patients with primary amenorrhea was X chromosome monosomy, i.e., pure Turner syndrome (TS) (45, X) in 6 (3.44%) cases. One case (0.57%) was detected with trisomy of X chromosome (47, XXX). Another case (0.57%) was observed as mosaic Turner with two cell lines: one cell line with monosomy of X chromosome and the other with trisomy of X chromosome and thus making a mosaic form as 45, X/47, XXX. In one case (0.57%), we noted X chromosome monosomy with an unbalanced structural abnormality of X chromosome, i.e., isochromosome Xq (45, X/46, X, i[Xq]). Interestingly, another patient (0.57%) with primary amenorrhea was presented as Turner mosaic with two cell lines: one cell line with monosomy of X chromosome and other cell line containing X and Y chromosome and making mosaic karyotype of 45, X/46, XY. We also found one each case (0.57%) with 46, XX, inv (9); 46, X, mar(X); and 45, X/46, X, dic(X) karyotype [Table 1 and Figures 1, 2]. Most of the cases of primary amenorrhea were presented with hypoplastic or absent uterus and/or small or absent ovaries sonologically.
Table 1.
Chromosomal abnormalities observed in the primary amenorrhea cases
| Total number of primary amenorrhea cases | Cytogenetic category | Karyotype | Number of cases (%) | |
|---|---|---|---|---|
| 174 | Normal 151 (86.78%) | 46,XX | 151 (86.78) | |
| Total abnormalities: 23 (13.22%) | XY female (sex reversal) | 46,XY | 10 (5.74) | |
| Numerical abnormality of X (trisomy of X chromosome) | 47,XXX | 1 (0.57) | ||
| Turner | 45,X | 6 (3.44) | ||
| Mosaic turner with numerical aberration of X | 45,X/47,XXX | 1 (0.57) | ||
| Turner with structural abnormality of X | 46,X, mar (X) | 1 (0.57) | ||
| Mosaic turner with structural abnormalites of X | 45,X/46,X,dic(X) | 1 (0.57) | ||
| 45,X/46,X,i(Xq) | 1 (0.57) | |||
| Inversion | 46,XX,inv(9) | 1 (0.57) | ||
| Mosaic XY female | 45,X/46,XY | 1 (0.57) | ||
Figure 1.
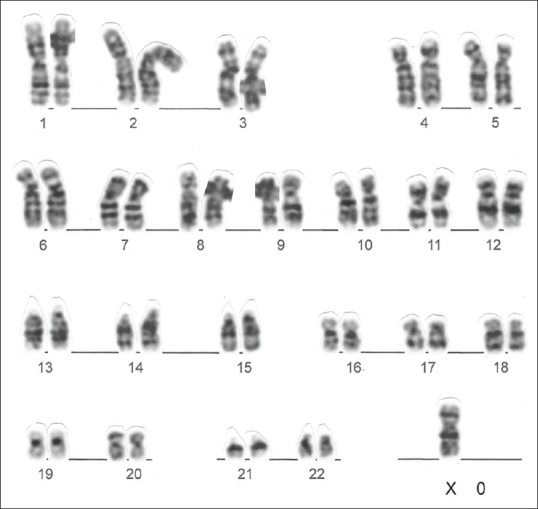
Metaphase of primary amenorrhea patient showing karyotype 45, X
Figure 2.
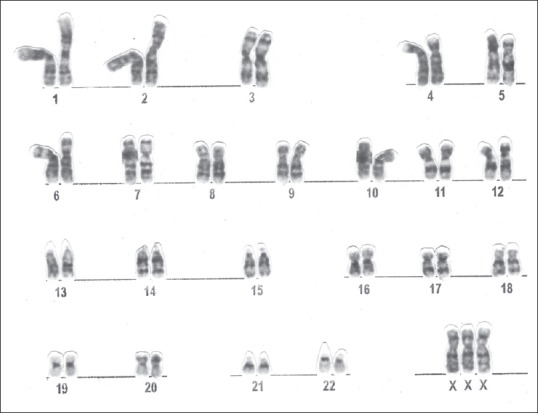
Metaphase of primary amenorrhea patient showing karyotype 47, XXX
DISCUSSION
Primary amenorrhea is a menstrual disorder. Normal menstruation of female participants indicates potent fertile candidate in future. Normal karyotype or normal genome is necessary for the normal development of an individual. Abnormal karyotype is an indication of abnormal development or disorders or diseases. Over the years, the global reports on the genetic basis of menstrual disorders suggest the relationship between the primary amenorrhea and abnormal karyotypes involving sex chromosomes and changes in sex chromosome may be of numerical or structural. Review of literature on the cytogenetic studies of primary amenorrhea[6,7,8,9,10,11,12,13,14,15] showed the involvement of sex chromosomes in primary amenorrhea cases [Table 2]. In different studies, the frequencies of abnormal karyotypes in primary amenorrhea differ and range between 10% and 25%. The distribution frequencies of different abnormal karyotypes (46, XY;45, X;47, XXX;45, X/47, XXX; 45, X/46, X, dic(X);46, XX, inv(9); 45, X/46, X, i(Xq); 46, X, mar(X); and 45, X/46, XY) have also some similarities in different studies. Our findings on chromosomal aberrations in primary amenorrhea cases showed overall 13.22% abnormality and the same trends of different types of chromosomal aberrations as shown in other studies [Table 2].
Table 2.
Comparison of results of our study with other studies
| Present study | Kara N et al. | Mohajertehran F et al. | El-Dahtory | Vijayalakshmi et al. | Kalavathi V. et al. | Akbar Safaei et al. | Joseph A and Thomas IM | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Year | 2017 | 2012 | 2012 | 2011 | 2010 | 2010 | 2009 | 1982 | ||
| Number of cases | 174 | 105 | 180 | 223 | 140 | 852 | 220 | 63 | ||
| Normal karyotype (%) | 151 (86.78) | 90 (85.7) | 136 (75.55) | 177 (79.37) | 101 (72.1) | 632 (74.18) | 176 (80) | 53 (84.12) | ||
| Total abnormal karyotype (%) | 23 (13.21) | 15 (14.3) | 44 (24.45) | 46 (20.63) | 39 (27.9) | 220 (25.82) | 44 (20) | 10 (15.87) | ||
| XY female (%) | 46,XY | 10 (5.74) | 2 (1.9) | 8 (4.4) | 2 (0.89) | 7 (5) | 52 (6.1) | 12 (5.5) | 1 (1.58) | |
| Numerical abnormality of X (%) | 47,XXX | 1 (0.57) | Not found | Not found | 2 (0.89) | Not found | 2 (0.23) | Not found | 1 (1.58) | |
| Turner (%) | 45,X | 6 (3.44) | 5 (4.8) | 21 (11.7) | 21 (9.42) | 11 (7.8) | 50 (5.87) | 19 (8.6) | 2 (3.17) | |
| Mosaic turner with numerical aberration of X (%) | Total mosaic turner: 6 (3.65) | 45,X/47,XXX | 1 (0.57) | Total mosaic turner: 1 (0.95) | Not found | Not found | Not found | 2 (0.23) | 1 (0.4) | Not found |
| Mosaic turner with structural abnormalities of X (%) | 45,X/46, X,iso(X) | 1 (0.57) | Not found | Not found | Not found | Not found | Not found | Not found | ||
| 46,XX,inv(9) | 1 (0.57) | Not found | Not found | Not found | Not found | Not found | Not found | |||
| 45,X/46,X, i(Xq) | 1 (0.57) | 1 (0.5) | Not found | 4 (4.9) | Not found | 1 (0.4) | Not found | |||
| Mosaic XY female (%) | 45,X/46,XY | 1 (0.57) | 2 (1.1) | Not found | 1 (0.7) | Not found | 2 (0.9) | Not found | ||
| Turner with structural abnormality (%) | 46,X,mar(X) | 1 (0.57) | Not found | Not found | Not found | Not found | Not found | Not found | Not found | |
Mainly, in our study, primary amenorrhea cases are due to aneuploidy (numerical changes) or structural changes of X chromosomes and sex reversal female participant (46, Y) with an abnormal Y chromosome. In general, primary amenorrhea cases show aneuploidy in karyotype (numerical changes) involving X chromosome and changes give rise to different kinds of syndrome patients such as TS (45, X) and different types of mosaic Turner with different karyotypes (45, X/47, XXX; 45, X/46, X, dic(X); 45, X/46, X, i(Xq); and 45, X/46, XY). Segregational anomalies of chromosomes during meiosis period of gametogenesis give rise to abnormal gametes, and after fertilization of abnormal gametes, the embryo develops with an aneuploidy karyotype. Mosaicism is due to postzygotic abnormal mitotic division. Occasionally, structural changes of X chromosome give rise to primary amenorrhea condition. The karyotype with X chromosome structural changes, i.e., 45, X/46, X, i(Xq) is due to initial X chromosome breakage and fusion and eventually unipolar segregation. Till the 6th week of embryonic development, the developing gonad is bipotential, irrespective of whether the inherited chromosome is XX or XY. Further differentiation is determined by a group of genes. Male sexual differentiation is initiated by the SRY gene (sex-determining region of Y chromosome) expression on the Y chromosome leading to the development of testis. In the absence of SRY, the bipotential gonad develops into ovary.[18] Female sex-reversed, Swyer syndrome, is a unique condition with female phenotype but karyotype is 46, XY. Swyer syndrome is a female phenotypes with normally developed Mullerian ducts, streak gonads, poorly developed breast, and primary amenorrhea, but the syndrome is characterized by 46, XY karyotype, i.e., sex-reversed female. Surprisingly, the high percentage of sex-reversed females with 46, XY karyotype was noted. Sex-reversed female may arise by the mutation or deletion of SRY gene present in the Y chromosome. Mutation might be a de novo one arising during the spermatogenesis or deletion might be due to unexpected cross-over of SRY gene to X chromosome during meiotic process of spermatogenesis and thus eventually give rise to a sperm with Y chromosome devoid of SRY gene or mutated gene. Fertilization of this Y-bearing sperm (mutated SRY or deleted SRY) with normal X-bearing ovum give rise to a sex-reversed female with karyotype 46, XY. Even though the sex-reversed females carry both X and Y sex chromosome, the undifferentiated gonad fails to develop into testis due to the absence of SRY gene. Testosterone and anti-Mullerian hormone (AMH) are not produced which lead to the absence of virilization and the development of female phenotype. As Wolffian duct fails to develop, the internal male organs are not formed. The absence of AMH leads to the development of the uterus, fallopian tube, cervix, and vagina. Possibly, in the mutation or deletion of SRY, the molecular genetic accident happens due to environmental pollution and changed or modern lifestyle. In 10%–15% of cases of female sex reversal, mutation of the SRY gene was seen. The remaining cases may be due to mutation of other genes involved in sex differentiation pathways such as the autosomal genes SOX9 and WT1 and the DAX1 gene on the X chromosome.[18,19,20] Some clinically diagnosed primary amenorrhea cases were referred to our laboratory, but the chromosomal analysis confirmed 46, XY karyotype. They had female external genitalia, but ultrasonography confirmed the presence of testis, and these were the cases of androgen insensitivity syndrome (previously known as testicular feminization). Hence, the cases were not considered as primary amenorrhea cases and were not included in the results.
Actually, for normal female development, two X chromosomes are necessary. Idiopathic primary amenorrhea with normal karyotype (46, XX) is difficult to explain, but with all possibilities, they must have some mutations in the genome or abnormal epigenetic changes of the genome. Possibly, environmental pollution or mutagens have a great role in the mutation, abnormal epigenetic changes, or endocrine disruptions which lead to primary amenorrhea. Clinically, it is impossible to assess the genetic makeup of amenorrhea cases. As like that of other studies, primary amenorrhea patients with karyotype abnormality from our study showed variations in the expression of secondary sexual characters.[14] Four patients with 46, XY karyotype had normal development of the mammary gland, and other 19 patients had poorly developed mammary glands. Pure TS patients were short stature with streak gonads sonologically. Turner mosaics were showing a wide range of phenotypic features from Turner to normal female. XY sex-reversed females were with moderate height and sonologically diagnosed absent or hypoplastic uterus and ovaries. The sex reversed female (46, XY) have completely underdeveloped or streak gonads which do not secrete hormones. Such patients with specific Y chromosome in their karyotype have an increased risk of developing germ cell tumor. These germ cell tumors arise from primordial germ cells or gonocytes. A high incidence of germ cell tumors in streak gonads of patients with the XY karyotype was also reported in the literature.[21,22] There is the necessity to remove such gonads at the time of diagnosis as there is a risk of development of malignancy in such patients.[10]
Autosomal genes have a crucial role in reproductive development. Kalantaridou and Chrousos in 2002 reported mutations of the GnRH receptor, FSHB, FSHR, and LHR gene leading to primary amenorrhea [Table 3].[23] In a study of primary amenorrhea, balanced autosomal translocation abnormality between chromosome 11 and 20 and between 14 and 21 was noted.[24] In an another report, the authors observed the absence of endometrial tissue due to balanced translocation (t[4;20]) resulting in primary amenorrhea.[25] Consanguinity has been reported to be the primary etiological factors resulting in homozygous condition for recessive autosomal/deleterious genes.[26] We also observed four cases of consanguinity out of 174 cases of primary amenorrhea.
Table 3.
Gene mutations leading to amenorrhea
| Defect | Location of gene | Phenotype | Key features | Inheritance |
|---|---|---|---|---|
| GnRH receptor gene mutations | 4q13.1 | Female | Amenorrhea | Autosomal recessive |
| FSHB gene mutations | 11p13 | Female | Primary amenorrhea | Autosomal recessive |
| FSHR gene | 2p21 | Female | Primary amenorrhea | Autosomal recessive |
| LHR gene mutations | 2p16-21 | Female | Primary amenorrhea | Autosomal recessive |
There is no curative treatment for genetic disorders or diseases, and only counseling can be provided for prevention and management of such diseases. After exclusion of the nongenetic causes by the gynecologists, cases with symptoms of primary amenorrhea should promptly be referred to the genetic laboratory for genetic study. Prompt referral and early diagnosis of primary amenorrhea cases is also necessary to plan a hormonal treatment of such patients to promote timely development of secondary sexual characters and to avoid serious health risk estrogen deficiency-related problems.[27,28] Primary amenorrhea may lead to anxiety, depression, and suicidal tendencies in affected individuals.[29] Providing psychological support during counseling is also very important in patients with Turner's syndrome with primary amenorrhea and sterility problem.[30] Because of high psychological and social impact, patients seeking genetic evaluation are a small fraction of affected women, and hence, the exact incidence or prevalence of amenorrhea is not known.[14] In our study, only 46 primary amenorrhea patients seek genetic advice before the age of 16 years and rest 128 were approached for medical advice after 16 years of age. During counseling of primary amenorrhea patients, mechanism of origin of such anomalies, the recurrence risk in sex chromosomal anomalies, the hormonal therapy, education/hobby/career, marriage, or reproductive options should be considered.[31] The early cytogenetic investigation (karyotyping) of the primary amenorrhea patients will help to understand the genetic makeup and accordingly will play an important role to counsel and manage the patients of primary amenorrhea in a better way.
CONCLUSION
Chromosomal abnormalities and abnormal karyotypes are one of the major etiological conditions for primary amenorrhea cases. We emphasize that an early diagnosis by karyotype analysis of all cases with primary amenorrhea or in suspected cases in early age or before puberty is essential for correct diagnosis and counseling in a better way. Such patients can be managed in a good way with possible therapies such as hormonal therapy and marriage counseling and also provided psychological support during counseling. Such kind of studies on primary amenorrhea will be helpful for making a national database.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgment
The authors are thankful to all the faculty of the department of obstetrics and gynecology for their constant support and to Mr. V. P. Kavinesan for technical support.
REFERENCES
- 1.Patavegar BN, Rasheed N, Pathak R, Kapilashrami MC, Farookee AB. Menstrual pattern and menstrual disorders among school going adolescent girls in Delhi. J Applied Basic Res Int. 2015;11:241–6. [Google Scholar]
- 2.Birmingham Alabama. Current evaluation of amenorrhea. Fertil Steril. 2008;90:219–25. doi: 10.1016/j.fertnstert.2008.08.038. [DOI] [PubMed] [Google Scholar]
- 3.Ten SK, Chin YM, Noor PJ, Hassan K. Cytogenetic studies in women with primary amenorrhea. Singapore Med J. 1990;31:355–9. [PubMed] [Google Scholar]
- 4.Mujumdar P, Ghosh S, Day SK. Association between primary amenorrhea and early maternal age: A population study. Indian J Sci Technol. 2015;8:1–6. [Google Scholar]
- 5.Merin T, Rema D, Preetha T, Amudha S, Jayalakshamma J, Mary M. Amenorrhea: Cytogenetic studies and beyond. Am J Mol Cell Biol. 2012;1:25–32. [Google Scholar]
- 6.Malla TM, Dar FA, Pandith AA, Zargar MH. Frequency and pattern of cytogenetic alterations in primary amenorrhea cases of Kashmir, North India. Egypt J Med Hum Genet. 2016;17:25–31. [Google Scholar]
- 7.Safaei A, Vasei M, Ayatollahi H. Cytogenetic analysis of patients with primary amenorrhea in Southwest of Iran. Iran J Pathol. 2010;5:121–5. [Google Scholar]
- 8.Kara N, Tural S, Elbistan M, Karakus N, Guven D, Kocak I. Cytogenetic findings of patients with Amenorrhea in Turkish population: A retrospective study. Indian J Hum Genet. 2012;12:87–92. [Google Scholar]
- 9.Mohajertehran F, Ghodsi K, Hafizi L, Rezaee A. Frequency and the type of chromosomal abnormalities in patients with primary amenorrhea in Northeast of Iran. Iran J Basic Med Sci. 2013;16:643–7. [PMC free article] [PubMed] [Google Scholar]
- 10.El-Dahtory F. Chromosomal abnormalities and hormonal disorders of primary amenorrhea patients in Egypt. Indian J Hum Genet. 2012;18:183–6. doi: 10.4103/0971-6866.100758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vijayalakshmi J, Koshy T, Kaur H, Mary FA, Selvi R, Deepa Parvathi V, et al. Cytogenetic analysis of patients with primary amenorrhea. Int J Hum Genet. 2010;10:71–6. [Google Scholar]
- 12.Kalavathi V, Chandra N, Nambiar GR, Shanker J, Sugunashankari P, Meena J, et al. Chromosomal abnormalities in 979 cases of amenorrhea: A review. Int J Hum Genet. 2010;10:65–9. [Google Scholar]
- 13.Wong MS, Lam ST. Cytogenetic analysis of patients with primary and secondary amenorrhoea in Hong Kong: Retrospective study. Hong Kong Med J. 2005;11:267–72. [PubMed] [Google Scholar]
- 14.Laxmi KV, Satyanarayana M. Cytogenetic analysis of primary amenorrhea cases. Indian J Hum Genet. 1997;3:95–100. [Google Scholar]
- 15.Joseph A, Thomas IM. Cytogenetic investigations in 150 cases with complaints of sterility or primary amenorrhea. Hum Genet. 1982;61:105–9. doi: 10.1007/BF00274197. [DOI] [PubMed] [Google Scholar]
- 16.Ambulkar PS, Ghosh SK, Ingole IV, Pal AK. Genotoxic and cytotoxic effects of antibacterial drug, ciprofloxacin, on human lymphocytes in vitro. Nepal Med Coll J. 2009;11:147–51. [PubMed] [Google Scholar]
- 17.McGowan-Jordan J, Simons A, Schmid M, editors. An International System for Human Cytogenomic Nomenclature. Basel, Freiburg: Karger; 2016. [Google Scholar]
- 18.John AM, Natarajan V, Srivastava V, George A, Rajaratnam S. A case of primary amenorrhoea. J Assoc Physicians India. 2014;62:753–5. [PubMed] [Google Scholar]
- 19.Hanley NA, Hagan DM, Clement-Jones M, Ball SG, Strachan T, Salas-Cortés L, et al. SRY, SOX9, and DAX1 expression patterns during human sex determination and gonadal development. Mech Dev. 2000;91:403–7. doi: 10.1016/s0925-4773(99)00307-x. [DOI] [PubMed] [Google Scholar]
- 20.Chander A, Shri R, Muthuvel A, Veluswamy C. Rare case of complete dysgenesis 46XY, Swyer syndrome. Int J Res Med Sci. 2017;5:714–6. [Google Scholar]
- 21.DU X, Zhang X, Li Y, Han Y. 46, XY female sex reversal syndrome with bilateral gonadoblastoma and dysgerminoma. Exp Ther Med. 2014;8:1102–4. doi: 10.3892/etm.2014.1922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mann JR, Corkery JJ, Fisher HJ, Cameron AH, Mayerová A, Wolf U, et al. The X linked recessive form of XY gonadal dysgenesis with a high incidence of gonadal germ cell tumours: Clinical and genetic studies. J Med Genet. 1983;20:264–70. doi: 10.1136/jmg.20.4.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kalantaridou SN, Chrousos GP. Clinical review 148: Monogenic disorders of puberty. J Clin Endocrinol Metab. 2002;87:2481–94. doi: 10.1210/jcem.87.6.8668. [DOI] [PubMed] [Google Scholar]
- 24.Kopakka N, Dalvi R, Shetty DL, Das BR, Mandava S. Balanced autosomal translocation and double robertsonian translocation in cases of primary amenorrhea in an Indian population. Int J Gynaecol Obstet. 2012;116:253–7. doi: 10.1016/j.ijgo.2011.09.029. [DOI] [PubMed] [Google Scholar]
- 25.Nigam A, Ahmad A, Batra S. Absent endometrium due to balanced translocation [t(4;20)] presenting as primary amenorrhea. J Hum Reprod Sci. 2014;7:63–5. doi: 10.4103/0974-1208.130867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Baba SM, Sameer AS, Siddiqi MA. A case of primary amenorrhea with 46+XY genotype from Kashmir Valley. Indian J Hum Genet. 2013;19:360–2. doi: 10.4103/0971-6866.120816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chung SH, Kim TH, Lee HH, Jeon DS. Primary amenorrhea of 16 year old girl that no chromosomal abnormalities. World Res J Med. 2013;1:4–5. [Google Scholar]
- 28.Pandurangi M, Tamizharasi M, Reddy NS. Pregnancy outcome of assisted reproductive technology cycle in patients with hypogonadotropic hypogonadism. J Hum Reprod Sci. 2015;8:146–50. doi: 10.4103/0974-1208.165141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Anitha GS, Tejeswini KK, Shivamurthi G. A clinical study of primary amenorrhea. J South Asian Fed Obstet Gynaecol. 2015;7:158–66. [Google Scholar]
- 30.Butnariu L, Covic M, Ivanov I, Bujoran C, Gramescu M, Gorduza EV. Clinical and cytogenetic correlation in primary and secondary amenorrhea: Retrospective study on 531 patients. Rev Romana Med Lab. 2011;19:149–60. [Google Scholar]
- 31.Rajangam S, Nanjappa L. Cytogenetic studies in amenorrhea. Saudi Med J. 2007;28:187–92. [PubMed] [Google Scholar]


