Abstract
Recent evidence points at the role that human endogenous retroviruses (HERVs) may play through the activation of genes integrated across the human genome. Although a variety of genetic/epigenetic mechanisms maintain most HERVs silenced, independent environmental stimuli including infections may transactivate endogenous elements favoring pathogenic conditions. Several studies associated exposures to Mycobacterium avium subsp. paratuberculosis (MAP) with increased anti-MAP seroreactivity in T1D patients. Here, we assessed humoral responses against HERV envelope antigens (HERV-KEnv and HERV-WEnv) and four MAP-derived peptides with human homologs in distinct populations: Sardinian children at T1D risk (rT1D) (n = 14), rT1D from mainland Italy (n = 54) and Polish youths with T1D (n = 74) or obesity unrelated to autoimmunity (OB) (n = 26). Unlike Sardinian rT1D, youths displayed increased anti-HERV-WEnv Abs prevalence compared to age-matched OB or healthy controls (24.32 vs. 11.54%, p = 0.02 for Polish T1D/OB and 31.48 vs. 11.90%, p = 0.0025 for Italian rT1D). Anti-HERV-KEnv responses showed variable trends across groups. A strong correlation between Abs levels against HERV-WEnv and homologous peptides was mirrored by time-related Abs patterns. Elevated values registered for HERV-WEnv overlaped with or preceded the detection of T1D diagnostic autoantibodies. These results support the hypothesis of MAP infection leading to HERV-W antigen expression and enhancing the production of autoantibodies in T1D.
Subject terms: Viral infection, Pre-diabetes
Introduction
Genetic heterogeneity characterizing type 1 diabetes (T1D) enhanced in the recent years the research on environmental components considered as putatively decisive factors underlying development of autoimmunity. However, despite numerous variables including diet, toxins, gut dysbiosis and viral infections assessed to date, no causal relationship with disease progression has been established. Complex and unclear T1D etiology suggests the involvement of various contributors acting on specific endpoints in a series of events. A large body of evidence points at the role that human endogenous retroviruses (HERVs) may play through the activation of genes integrated in multiple copies across the human genome as traces of ancestral infections1. The envelope protein of HERV-W and HERV-K families has been detected in patients with several immune-mediated and neurodegenerative diseases2–4 and reported to function as a superantigen due to the ability of inducing strong T cell responses5. The specific role of HERV envelope proteins in immune and neurological disorders, including T1D, has been recently reviewed6. In T1D, the initially described association with HERV-K has been questioned based on the ubiquitous nature of its antigens7–9, whereas HERV-W remains a valid object of investigation. A high expression of HERV-W envelope protein (HERV-WEnv) observed in serum, peripheral blood mononuclear cells and pancreas of T1D patients has been recently correlated with macrophage infiltrates, inhibited insulin secretion in human Langerhans islets in vitro and corroborated by studies in transgenic mice10 which also develop autoantibodies against recombinant viral gp70 envelope protein increasing in titer along with disease progression11. GNbAC1 monoclonal antibody specifically targeting HERV-WEnv is currently under a phase-IIa clinical trial testing as a possible HERV-based therapeutic approach in T1D12. Although a variety of genetic and epigenetic mechanisms maintain most HERVs silenced, independent environmental stimuli such as infections may transactivate endogenous elements favoring pathogenic conditions13. Previously, we have described a significantly elevated seroreactivity of T1D subjects towards antigens sharing amino acid sequence homology derived from Mycobacterium avium subsp. paratuberculosis (MAP) and human proteins involved in the pathogenesis of diabetes: zinc transporter 8 (ZnT8) and proinsulin (PI)14,15. These epitopes were recognized by over 65% of T1D patients from independent cohorts in contrast to non-diabetic controls and patients affected by type 2 diabetes (T2D) who displayed specific responses to a very limited extent16. In the present study, we show a strong correlation between antibodies (Abs) directed against the assessed peptides and HERV-WEnv fragment in three populations at risk of T1D (rT1D) or with developed disease differing by age and biogeographic background. The observed time-related Abs patterns hint at a possible reactivation of HERV-WEnv following exposure to MAP that, through imbalance facilitated by cross-reacting homologous epitopes, may lead to the loss of immune tolerance.
Results
Seroreactivity against HERVEnv antigens in correlation with T1D clinical phase
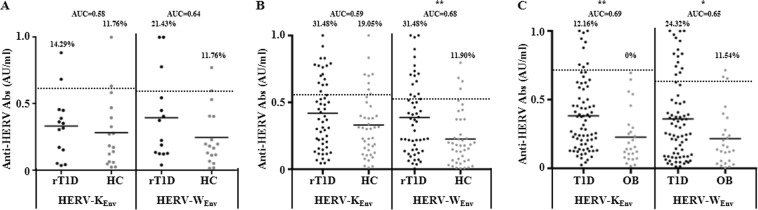
In all analyzed groups, subjects at risk of or affected by T1D displayed higher Abs values against HERVEnv epitopes compared to HC and OB patients (Fig. 1). More pronounced differences of seroreactivity against the two peptides were registered among the youngest participants from Sardinia, for which the prevalence of anti-HERV-KEnv Abs reached similar values between children at T1D risk or HC (14.29 vs. 11.76%, respectively) and corresponded to elevated values for HERV-WEnv among patients followed up for autoimmunity, with unchanged levels in HC (21.43 vs. 11.76%, respectively).
Figure 1.
Prevalence of Abs against HERVEnv antigens in Sardinian rT1D children (A), rT1D youths from mainland Italy (B) and Polish T1D youths (C) along with the respective control groups. The dotted lines represent positivity thresholds calculated by ROC analysis. Dashed lines indicate the respective mean AU/ml values. Area under the curve (AUC) and statistical significance, when attained, are reported for each distribution.
Youths from mainland Italy showed differing trends. Elevated responses to both HERV-WEnv and HERV-KEnv were observed in over 31% of rT1D subjects. In contrast, HC presented significantly lower levels of anti-HERV-WEnv Abs (11.9%, p = 0.0025) with somewhat higher reactivity towards HERV-KEnv (19.05%).
Less pronounced, although highly significant reactivity against HERVEnv antigens were registered among Polish T1D patients whose prevalence of anti-HERV-KEnv Abs accounted for 12.16%, whilst no positive subject was found in the OB group (p = 0.0034). HERV-WEnv triggered more elevated responses in both clinical conditions, however marked difference was still maintained (24.32% for T1D vs. 11.54% for OB, p = 0.02).
Correlation between reactivity against HERVEnv and homologous MAP/human epitopes
Previous screening for Abs against fragments of mycobacterial cation transporter MAP3865c133–141 and MAP3865c125–133, a hypothetical protein MAP2404c70–85, and a glucan branching protein MAP1,4αgbp157–173 alongside their respective human homologs ZnT8186–194, ZnT8178–186, PI46–61 and PI64–80 revealed elevated responsiveness of T1D and rT1D patients to the selected antigens when compared to reference controls17,18.
Even though Abs prevalence followed different patterns in the three assessed populations, values registered for either HERV-WEnv or HERV-KEnv correlated with MAP/PI epitopes regardless of the location and disease phase (Table 1). Particularly elevated R2 values were reached between HERV-WEnv and PI antigens in patients with established T1D. No correlation was found with single-point levels of diagnostic biomarkers or age groups within single cohorts, however the coefficient of determination increased proportionally considering the mean age of entire populations (children < younger youths (Italian) < older youths (Polish)). Less linear trends and slightly lower R2 were registered for HERV-KEnv among patients and for both antigens in youth control groups. In Sardinian healthy subjects, comparison between anti-HERV-KEnv and anti-MAP (PI homologs) Abs corresponded to higher coefficients than in relative rT1D children. Abs targeting MAP/ZnT8 homologous epitopes correlated overall to a weaker extent with each HERVEnv epitope but maintained high coefficient values for HERV-WEnv in Polish T1D group. As well, anti-ZnT8186–194 Abs corresponded to a marked correlation with reactivity towards HERV-WEnv in both populations of subjects at T1D risk.
Table 1.
Relationship between HERVEnv antigens and homologous epitope pairs derived from MAP and human proteins expressed as R2. Values were obtained based on all available samples for single populations. For each peptide, R2 corresponding to patients are reported with underneath coeffcients relative to the respective control groups.
| MAP/human antigen | HERV-KEnv | HERV-WEnv | ||||
|---|---|---|---|---|---|---|
| Sardinian | Italian | Polish | Sardinian | Italian | Polish | |
| MAP1,4αgbp157–173 | 0.7167 | 0.7154 | 0.6581 | 0.7008 | 0.8066 | 0.8149 |
| 0.8489 | 0.6457 | 0.5271 | 0.7716 | 0.6535 | 0.5569 | |
| PI64–80 | 0.7822 | 0.7130 | 0.7630 | 0.7547 | 0.8151 | 0.8617 |
| 0.4783 | 0.6725 | 0.5191 | 0.1507 | 0.6405 | 0.5871 | |
| MAP2404c70–85 | 0.7950 | 0.7155 | 0.7446 | 0.7984 | 0.7747 | 0.7963 |
| 0.8603 | 0.5363 | 0.7590 | 0.6219 | 0.5187 | 0.7718 | |
| PI46–61 | 0.7816 | 0.7123 | 0.7229 | 0.7768 | 0.7974 | 0.8209 |
| 0.7709 | 0.6009 | 0.7219 | 0.6709 | 0.5264 | 0.7560 | |
| MAP3865c133–141 | 0.6676 | 0.6586 | 0.6303 | 0.5790 | 0.6929 | 0.7102 |
| 0.3907 | 0.5207 | 0.5352 | 0.1459 | 0.3980 | 0.5887 | |
| ZnT8186–194 | 0.7847 | 0.6506 | 0.6457 | 0.7017 | 0.7530 | 0.7186 |
| 0.6430 | 0.6354 | 0.5996 | 0.5538 | 0.5591 | 0.6708 | |
| MAP3865c125–133 | 0.8335 | 0.6271 | 0.6746 | 0.8003 | 0.6815 | 0.7573 |
| 0.3677 | 0.4851 | 0.4913 | 0.5303 | 0.4646 | 0.6471 | |
| ZnT8178–186 | 0.7616 | 0.5817 | 0.6825 | 0.7984 | 0.6194 | 0.8153 |
| 0.7146 | 0.6213 | 0.4032 | 0.4333 | 0.5149 | 0.4742 | |
Temporal coincidence of Abs patterns
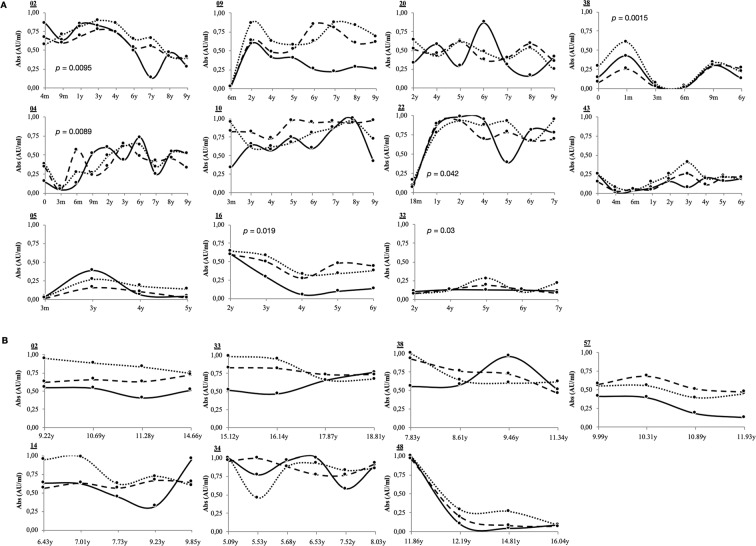
Upon time-point analysis of Abs status in subjects for which follow-up samples were available (n = 13 for Sardinian children and n = 26 for Italian youths at T1D risk), the obtained values were classified in two sets based on peptide sequence homology: MAP/ZnT8 and MAP/PI (Fig. 2). Further comparison with Abs against HERV-WEnv confirmed the correlation in a time-related manner. Interestingly, value reads for HERV-WEnv were prone to fluctuations mirrored by variable Abs status against MAP/human epitopes during long observation periods. Moreover, they tended to raise or decrease in concert with or subsequently to variations of Abs specific for the homologous peptides assessed. Statistically significant results were obtained in 6 out of 11 Sardinian patients but not in the Italian rT1D group which may be due a lower number of time-point samples available. This may result from shifted fluctuations during which the maximal peaks of anti-HERV-WEnv Abs not necessarily correspond to minimal responses against MAP/human homologous antigens.
Figure 2.
Time-related seroreactivity against HERV-WEnv and homologous MAP/human antigens in subjects at T1D risk from Sardinia (A) and mainland Italy (B). For graphical simplification, data relative to MAP/ZnT8 or MAP/PI sets are represented as separated means since values obtained for each of the four peptides included in their specific set fell into similar ranges. Results shown correspond to subjects for which at least 4 follow-up samples were available with patients’ identifiers reported above the Y axis of each graph. HERV-WEnv: continuous line. MAP/PI: dashed line. MAP/ZnT8: dotted line. m: months. y: years.
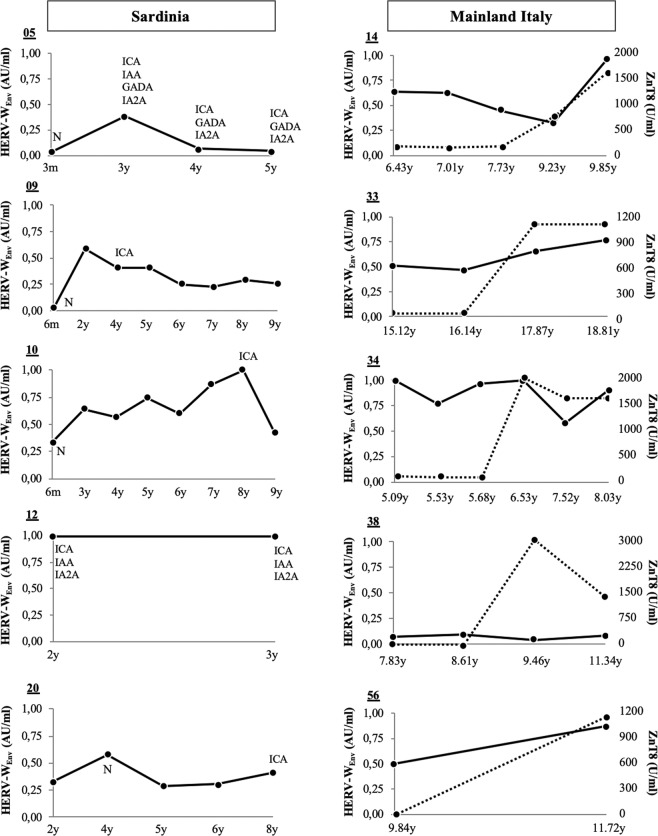
Among Italian subjects at T1D risk who over a period of 4 years developed high titers of standard Abs (n = 5), especially those specific for ZnT8 C-terminal region (>1000 U/ml), 80% showed a strong seroreactivity against HERV-WEnv and the homologous peptides (Fig. 3). This biomarker was not included for diagnostic purposes in the other study populations, however, in the Sardinian cohort, increased values registered for HERV-WEnv overlaped with or preceded the moment when diagnostic autoantibodies (ICA, IAA, IA2A and/or GADA) were detected.
Figure 3.
Development of standard Abs in relation to reactivity against HERV-WEnv in Sardinian children (A) and youths from mainland Italy (B) at T1D risk. Patients’ identifiers are reported above the Y axis of each graph. Black line: anti-HERV-WEnv Abs values. Dotted line: standard Abs against ZnT8. For Sardinian cohort, the type of diagnostic Abs is specified when positivity threshold has been exceeded. N: measurement not performed. m: months. y: years.
Discussion
In this prospective study, we investigated the production of Abs against two agents, namely HERV-W and MAP, in young populations at T1D risk from distinct geographical regions of Italy based on data collected over a 10-year period. Our results indicate a possible role of MAP in HERV-WEnv activation which correlates with follow-up levels of diagnostic biomarkers. In addition, the Abs status was evaluated in environmentally unrelated youths with clinical T1D or non-diabetic obesity in order to assess whether developed autoimmunity and risk for metabolic syndrome may determine characteristic serological features.
MAP infection is common only to some exposed subjects and hardly identifiable due to latency phases, extremely low growth and tropism to intestinal macrophages19. Although the most severe consequences of MAP proliferation occur in ruminants, the mycobacterium may be easily transmitted to humans with a wide range of contaminated animal products, while anti-MAP Abs have been detected in milk of breastfeeding mothers20,21. On the other hand, HERVs are intrinsic sequences disseminated throughout human genome that under certain circumstances may be activated from their silenced form and expressed generating a series of antigens primarily on monocytes and B cells22,23. A few studies performed to evaluate the association between HERVs and MAP in autoimmune diseases reported a partial decrease of anti-MAP Abs in multiple sclerosis (MS) following natalizumab treatment which efficiently reduced levels of Abs targeting HERV-WEnv24. No significant difference in specific humoral responses against MAP antigen was obtained when comparing rheumatoid arthritis (RA) patients under treatment with healthy controls25. Therapies employed to alleviate MS and RA symptoms act on the immune system, thereby influencing inflammatory responses in general. Outcomes of the present study are based on subjects free from therapy or in the course of insulin treatment, which however does not suppress the reactivity of immune cells. These differences may explain distinct results among the three autoimmune conditions and be the first step to suggest MAP infection as a plausible circumstance able to induce the expression of HERV-W envelope antigens which may result in a further interplay between mycobacterial, viral and human Abs leading in turn to immune imbalance. A possibility of such cascade of events to occur and its link with the development of autoimmunity requires further investigation.
Elevated reactivity and strong correlations between HERV-WEnv and MAP/human homologous peptides registered for rT1D youths are in line with this hypothesis. When Abs levels were distributed in more homogeneous profiles over time, values specific for the selected peptides tended to maintain similar trends. In subjects displaying high variations of responsiveness relative to MAP/human antigens, fluctuations specific for HERV-WEnv were particularly marked with peak values corresponding to decreasing levels of anti-MAP/human Abs and the lowest values correlating negatively. These shifts may be indicative of sequentiality characteristic for distinct epitopes resulting from a selective responsiveness; the crucial point is to understand whether MAP drives the immunity as a primary antigen. Interestingly, anti-HERV-WEnv Abs in T1D youths under insulin therapy equaled to a lower prevalence compared to age-matched rT1D. These subjects also showed significantly increased levels of Abs targeting HERV-KEnv even though seropositivity accounted for only 12.16% of the assessed population (Fig. 1C). Past studies based on the viral IDDMK1,2 22 sequence expression excluded the association between HERV-K and T1D7,9,26 after an initial report in favor of such a link27. Different loci encoding HERV genes may be involved in a potential immune reactivity28, nonetheless the absence of significant responsiveness in rT1D would not support a causal role of HERV-K family. In contrast, raised levels of Abs against other two HERV-W epitopes (HERV-WEnv93–108 and HERV-WEnv140–160) in diabetic and rT1D youths and a marked degree of correlation with MAP/PI peptides were observed in a pilot cohort (data not shown).
It has been recently reported that MAP infection in cattle affects the expression of immune regulatory genes, including Th17-derived cytokines, interferon regulatory factors, and calcium signaling-associated genes29. Both immune activation and inflammation can impair HERV transcription through interaction with specific binding sites in relative HERV promoter regions30. Similarly, oxidative stress also seems to contribute by hampering binding of methyl groups transferred by DNA methyltransferases and enhancing gene expression upon inhibition of histone deacytelases30. Increased activity of selenium-dependent glutathione peroxidase (GPx) indicative of oxidative processes has been reported in MAP-infected cattle and patients with Crohn’s disease31. Formerly, destruction of the exocrine pancreas was observed in a goat with confirmed MAP infection32. Recent studies in transgenic mice demonstrated that artificially induced expression of HERV-WEnv promotes hyperglycemia, decreased insulin production and immune cell infiltrates in the exocrine part10. The hypothesis of MAP as a plausible natural trigger of HERV-WEnv transactivation is supported also by the fact that macrophages activated in granulomatous lesions of infected goats highly express CD68+ indicative of elevated numbers of lysosomes and acid phosphatase activity33. A study in wild ruminants affected by Johne’s disease proved the presence of MAP and cell damage in the pancreatic lymph nodes34. Levet et al., 2017, reported increased CD68+ macrophage infiltrates in T1D exocrine pancreas correlated with HERV-WEnv expression10. In line with our earlier results, involvement of MAP in the pathogenesis of T1D may occur through a more complex network of interaction that previously thought.
As humans are not the primary target of MAP, it may act indirectly on immune homeostasis as a consequence of survival mechanisms eluding host defense against pathogens and conditions favoring the expression of HERV-W antigens.
Limitations of the study. In this pilot study, we investigated for the first time the interaction between MAP and HERV-W related to T1D, however some limitations couldn’t be avoided. The results were obtained based on three independent populations differing by mean age and the advancement of autoimmunity, each of them forming groups with relatively low numbers of participants. In particular, statistical significance was not attained for a single-point analysis of Sardinian rT1D population compared with age-matched healthy controls. The lack of follow-up samples until development of diabetes in Italian rT1D group and before manifestation of symptoms in Polish T1D population did not allow for a continuous determination of Abs status against the assessed epitopes. Prospective screening involving groups increased in the number of participants is therefore necessary to further assess Abs patterns and a possible transactivation of HERV-W following mycobacterial infection. Investigation of cell-mediated responses and application of in vivo models alongside the analysis of retroviral gene expression should be done in order to identify the most probable sequence of events which take place in nature. As well, development of serological markers in numerically extended cohorts of newborns and young children with regard to a possible vertical transmission of Abs needs further evaluation.
Materials and Methods
Subjects
Independent cohorts of rT1D subjects were enrolled in two separated centers: 54 children and youths (mean age 9.42 ± 3.84 years, 1:1 male/female ratio) attending the Tor Vergata University Hospital of Rome, Italy, and 14 children (1:2 male/female ratio, mean age 5.85 ± 2.19 years) attending the Department of Diabetes, St. Michele Hospital of Cagliari, Italy. Venous whole blood was collected for the purpose of prospective studies providing in total 103 and 87 follow-up samples for the respective groups over a 10-year period. T1D risk was verified by the presence of disease familiarity with first-degree relatives (parents or siblings), high risk HLA genotype and/or the presence of standard autoantibodies (ZnT8, GADA, IA2A, ICA and IAA). Healthy volunteers (HC) without known history of autoimmune disorders and recent inflammatory episodes were recruited as reference controls following periodic medical checks in the corresponding geographic areas (n = 42, mean age 6.94 ± 3.58 years, 1:1.5 male/female ratio, the Tor Vergata University Hospital of Rome; and n = 17, mean age 6.66 ± 2.59 years, 1:2.4 male/female ratio, Department of Endocrinology, University Hospital of Sassari, Italy). Additionally, a group composed of children/youths with established T1D under insulin therapy (n = 74, mean age 12.9 ± 3.51; females n = 47, males n = 27) and age-matched obese patients (OB; n = 26, mean age 12.54 ± 2.99, mean BMI 27.35 ± 4.53; females n = 16, males n = 10) was enrolled at the Clinic of Endocrinology and Diabetology, Children’s Memorial Health Institute in Warsaw, Poland, for single time-point sample collections in a distinct biogeographic background. General inflammatory background of obesity in an apparent healthy condition often affecting young populations nowadays was considered a valuable control. T1D onset was diagnosed based on the levels of standard biomarkers and levels of glycated hemoglobin, according to the American Diabetes Association criteria35. The study protocols were approved by delegated Bioethics Committees (Children’s Memorial Health Institute, Tor Vergata University Hospital, St. Michele Hospital and University Hospital of Sassari) and written informed consent from a parent or legal tutor was obtained for all study participants. All methods were performed in accordance with regional and national regulations.
Diagnostic autoantibodies
Levels of Abs specific to the ZnT8 C-terminal region (268–369, 325R or 325W) were determined in the sera by Protein A-radioimmunoprecipitation assays according to the protocol by Lampasona et al.36 with positivity threshold set at 30 U/mL. Abs to insulin, GAD65, and IA-2 were measured by radioligand assays using commercial kits (CentAK® IAA RT, CentAK® anti-GAD65, and CentAK® anti-IA2, Medipan, Germany) according to the manufacturer’s instruction. Values are expressed in arbitrary units with the respective Abs thresholds of >0.4, >0.9 and >0.75 U/mL.
Antigens
HERV-KEnv19-37 (SVWVPGPTDDRCPAKPEEE) fragment of envelope surface glycoprotein were selected based on significantly increased seroreactivity in MS and ALS patients observed by Arru et al.37. Other studies reported comparable responsiveness to a range of HERV-W envelope surface peptides38, therefore HERV-WEnv109–123 (FTQTGMSDGGGVQDQ) was used in the present assessment as a first-trial choice for a pilot correlation analysis. HERVEnv peptides along with homologous epitope pairs derived from MAP and human proteins ZnT8 and PI, were synthesized as formerly described17,37.
Serological assays and data analysis
The presence of Abs against the selected antigen set was assessed in separated serum samples through indirect enzyme-linked immunosorbent assay following in-house optimized protocols14. Data were normalized to a strongly positive control serum included in all assays with Abs reactivity set at 1.0 arbitrary units AU/mL. Optimal thresholds to discriminate between positive and negative mean values were identified based on the receiver operating characteristic (ROC) curves, setting specificity at 90%. GraphPad Prism ver. 6.0 was employed to determine levels of statistical significance through Mann-Whitney U test. Normality of the distribution was assessed by D’Agostino-Pearson test. Comparison of positivity between patients and reference controls was performed using Fisher’s exact test.
Acknowledgements
The authors would like to thank Ewa Balas from the Children’s Memorial Health Institute for her technical expertise and dedication to this study.
Author Contributions
M.N. was involved in the study design, carried out experimental work, performed data analysis and drafted the manuscript. M.W.C., G.S.P., J.T., J.M. and M.S. were involved in the selection and enrollment of Polish patients according to established criteria, collection of clinical data and preparation of biological material used in the present study. L.A.S. conceived the study and its experimental design, analyzed the results and approved the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Belshaw R, et al. Long-term reinfection of the human genome by endogenous retroviruses. Proc. Natl. Acad. Sci. USA. 2004;101:4894–4899. doi: 10.1073/pnas.0307800101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gröger V, Cynis H. Human endogenous retroviruses and their putative role in the Ddevelopment of autoimmune disorders such as multiple sclerosis. Front. Microbiol. 2018;9:265. doi: 10.3389/fmicb.2018.00265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hansen DT, Petersen T, Christensen T. Retroviral envelope proteins: Involvement in neuropathogenesis. J. Neurol. Sci. 2017;380:151–163. doi: 10.1016/j.jns.2017.07.027. [DOI] [PubMed] [Google Scholar]
- 4.Brüting C, Emmer A, Kornhuber ME, Staege MS. Cooccurrences of putative endogenous retrovirus-associated diseases. Biomed. Res. Int. 2017;2017:7973165. doi: 10.1155/2017/7973165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lavillette D, et al. The envelope glycoprotein of human endogenous retrovirus type W uses a divergent family of amino acid transporters/cell surface receptors. J. Virol. 2002;76:6442–6452. doi: 10.1128/JVI.76.13.6442-6452.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grandi N, Tramontano E. HERV envelope proteins: Physiological role and pathogenic potential in cancer and autoimmunity. Front. Microbiol. 2018;9:462. doi: 10.3389/fmicb.2018.00462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Conrad B, et al. A human endogenous retroviral superantigen as candidate autoimmune gene in type I diabetes. Cell. 1997;90:303–313. doi: 10.1016/S0092-8674(00)80338-4. [DOI] [PubMed] [Google Scholar]
- 8.Badenhoop K, et al. IDDM patients neither show humoral reactivities against endogenous retroviral envelope protein nor do they differ in retroviral mRNA expression from healthy relatives or normal individuals. Diabetes. 1999;48:215–8. doi: 10.2337/diabetes.48.1.215. [DOI] [PubMed] [Google Scholar]
- 9.Kim A, et al. Human endogenous retrovirus with a high genomic sequence homology with IDDMK(1,2)22 is not specific for Type I (insulin-dependent) diabetic patients but ubiquitous. Diabetologia. 1999;42:413–418. doi: 10.1007/s001250051173. [DOI] [PubMed] [Google Scholar]
- 10.Levet S, et al. An ancestral retroviral protein identified as a therapeutic target in type-1 diabetes. JCI Insight. 2017;2:17. doi: 10.1172/jci.insight.94387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bashratyan R, et al. Type 1 diabetes pathogenesis is modulated by spontaneous autoimmune responses to endogenous retrovirus antigens in NOD mice. Eur. J. Immunol. 2017;47:575–584. doi: 10.1002/eji.201646755. [DOI] [PubMed] [Google Scholar]
- 12.Curtin F, et al. A new therapeutic approach for type 1 diabetes: Rationale for GNbAC1, an anti-HERV-W-Env monoclonal antibody. Diabetes. Obes. Metab. 2018;20:2075–2084. doi: 10.1111/dom.13357. [DOI] [PubMed] [Google Scholar]
- 13.Nellåker C, et al. Transactivation of elements in the human endogenous retrovirus W family by viral infection. Retrovirology. 2006;3:44. doi: 10.1186/1742-4690-3-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Masala S, et al. Antibodies recognizing Mycobacterium avium paratuberculosis epitopes cross-react with the beta-cell antigen ZnT8 in Sardinian type 1 diabetic patients. PLoS One. 2011;6:e26931. doi: 10.1371/journal.pone.0026931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Masala S, et al. Proinsulin and MAP3865c homologous epitopes are a target of antibody response in new-onset type 1 diabetes children from continental Italy. Pediatr. Diabetes. 2015;16:189–195. doi: 10.1111/pedi.12269. [DOI] [PubMed] [Google Scholar]
- 16.Rosu V, et al. Specific immunoassays confirm association of Mycobacterium avium subsp. paratuberculosis with type-1 but not type-2 diabetes mellitus. PLoS One. 2009;4:e4386. doi: 10.1371/journal.pone.0004386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Niegowska M, et al. Type 1 diabetes at-risk children highly recognize Mycobacterium avium subspecies paratuberculosis epitopes homologous to human Znt8 and proinsulin. Sci. Rep. 2016;6:22266. doi: 10.1038/srep22266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Niegowska M, et al. Recognition of ZnT8, proinsulin, and homologous MAP peptides in Sardinian children at risk of T1D precedes detection of classical islet antibodies. J. Diabetes Res. 2016;2016:5842701. doi: 10.1155/2016/5842701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Arsenault JR, et al. From mouth to macrophage: mechanisms of innate immune subversion by Mycobacterium avium subsp. paratuberculosis. Vet. Res. 2014;45:54. doi: 10.1186/1297-9716-45-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Waddell L, Rajic A, Stärk K, McEwen SA. Mycobacterium avium ssp. paratuberculosis detection in animals, food, water and other sources or vehicles of human exposure: A scoping review of the existing evidence. Prev. Vet. Med. 2016;132:32–48. doi: 10.1016/j.prevetmed.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 21.Naser SA, Schwartz D, Shafran I. Isolation of Mycobacterium avium subsp paratuberculosis from breast milk of Crohn’s disease patients. Am. J. Gastroenterol. 2000;95:1094–1095. doi: 10.1111/j.1572-0241.2000.01954.x. [DOI] [PubMed] [Google Scholar]
- 22.Grandi N, Tramontano E, Type W. Hhuman endogenous retrovirus (HERV-W) integrations and their mobilization by L1 machinery: Contribution to the human transcriptome and impact on the host physiopathology. Viruses. 2017;9:162. doi: 10.3390/v9070162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brudek T, et al. B cells and monocytes from patients with active multiple sclerosis exhibit increased surface expression of both HERV-H Env and HERV-W Env, accompanied by increased seroreactivity. Retrovirology. 2009;6:104. doi: 10.1186/1742-4690-6-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Arru G, et al. Natalizumab modulates the humoral response against HERV-Wenv73–88 in a follow-up study of Multiple Sclerosis patients. J. Neurol. Sci. 2015;357:106–108. doi: 10.1016/j.jns.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 25.Bo M, et al. Rheumatoid arthritis patient antibodies highly recognize IL-2 in the immune response pathway involving IRF5 and EBV antigens. Sci. Rep. 2018;8:1789. doi: 10.1038/s41598-018-19957-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jaeckel E, et al. No evidence for association between IDDMK(1,2)22, a novel isolated retrovirus, and IDDM. Diabetes. 1999;48:209–214. doi: 10.2337/diabetes.48.1.209. [DOI] [PubMed] [Google Scholar]
- 27.Muir A, Ruan QG, Marron MP, She JX. The IDDMK(1,2)22 retrovirus is not detectable in either mRNA or genomic DNA from patients with type 1 diabetes. Diabetes. 1999;48:219–222. doi: 10.2337/diabetes.48.1.219. [DOI] [PubMed] [Google Scholar]
- 28.Grandi N, Cadeddu M, Blomberg J, Tramontano E. Contribution of type W human endogenous retroviruses to the human genome: characterization of HERV W proviral insertions and processed pseudogenes. Retrovirology. 2016;13:67. doi: 10.1186/s12977-016-0301-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Park HE, Park HT, Jung YH, Yoo HS. Gene expression profiles of immune-regulatory genes in whole blood of cattle with a subclinical infection of Mycobacterium avium subsp. paratuberculosis. PLoS One. 2018;13:e0196502. doi: 10.1371/journal.pone.0196502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Morris G, Maes M, Murdjeva M, Puri BK. Do human endogenous retroviruses contribute to multiple sclerosis, and if so, how? Mol. Neurobiol. 2018 doi: 10.1007/s12035-018-1255-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Qasem A, Abdel-Aty A, Abu-Suwa H, Naser SA. Oxidative stress due to Mycobacterium avium subspecies paratuberculosis (MAP) infection upregulates selenium-dependent GPx activity. Gut Pathog. 2016;8:12. doi: 10.1186/s13099-016-0090-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Al-Dubaib MA, Mahmoud O. Paratuberculosis of goats at Qassim region of Central Saudi Arabia. BJVM. 2008;11:65–69. [Google Scholar]
- 33.Valheim M, Sigurdardottir OG, Storset AK, Aune LG, Press CM. Characterization of macrophages and occurrence of T cells in intestinal lesions of subclinical paratuberculosis in goats. J. Comp. Path. 2004;131:221–232. doi: 10.1016/j.jcpa.2004.04.004. [DOI] [PubMed] [Google Scholar]
- 34.Williams ES, Snyder SP, Martin KL. Pathology of spontaneous and experimental infection of North American wild ruminants with Mycobacterium paratuberculosis. Vet. Pathol. 1983;20:274–291. doi: 10.1177/030098588302000304. [DOI] [PubMed] [Google Scholar]
- 35.American Diabetes Association Diagnosis and classification of diabetes mellitus. Diabetes Care. 2011;34:S62–S69. doi: 10.2337/dc11-S062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lampasona V, et al. Zinc transporter 8 antibodies complement GAD and IA-2 antibodies in the identification and characterization of adult-onset autoimmune diabetes: Non Insulin Requiring Autoimmune Diabetes (NIRAD) Diabetes Care. 2010;33:104–108. doi: 10.2337/dc08-2305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Arru G, et al. Humoral immunity response to human endogenous retroviruses K/W differentiates between amyotrophic lateral sclerosis and other neurological diseases. Eur. J. Neurol. 2018;25:1076–e84. doi: 10.1111/ene.13648. [DOI] [PubMed] [Google Scholar]
- 38.Mameli G, et al. Epitopes of HERV-Wenv induce antigen-specific humoral immunity in multiple sclerosis patients. J. Neuroimmunol. 2015;280:66–68. doi: 10.1016/j.jneuroim.2015.03.003. [DOI] [PubMed] [Google Scholar]