Abstract
Amyloidosis, deposition of improperly folded insoluble proteins, may affect one organ or may be systemic. Although plasma cell dyscrasia is frequently implicated in etiology and is due to immunoglobulin light chain production (AL amyloidosis), age-related amyloidosis is believed to be secondary to transthyretin production (ATTR), chronic inflammation-related amyloidosis is thought due to acute phase reactants (AA amyloidosis), and dozens of others are also described. Clinical presentation is dependent upon the organ(s) involved and those associated with unexpected death are expected to involve the cardiovascular system. All cases received for forensic postmortem examination at the Medical University of South Carolina from 2008 to May 15, 2015 were searched to identify any in which amyloidosis was listed as a final diagnosis. Seven cases were identified that met the criteria and were reviewed for demographic information, presentation, cause and manner of death, and assessment of pertinence of the diagnosis of amyloidosis to the cause and manner of death. Interestingly, gross examination of the heart was suggestive of amyloidosis or other infiltrative process in only two of the cases reviewed and a history of myeloma was only noted in one individual. Common gross and microscopic findings are described and relevant medical history and toxicology findings are compared.
Keywords: Forensic pathology, Amyloid, Amyloidosis, Fatal, Postmortem, Restrictive cardiomyopathy, Sudden death, Transthyretin
Introduction
Amyloidosis results from the deposition of improperly folded insoluble proteins in the extracellular space. These insoluble proteins may interfere with the normal function of organs either due to physical disturbance of organ function or through direct toxicity of circulating proteins. The protein can be identified histologically because of its chemical nature (nonbranching fibrils arranged in a cross-β-pleated sheet) via Congo red staining and characteristic “apple-green” birefringence with polarized light. Formalin-fixed paraffin embedded sections stained with Congo red will also demonstrate positive red emissions filter fluorescence by ultraviolet light examination. In fact, the fluorescence assay provides higher sensitivity (1, 2). Electron microscopy demonstrates nonbranching 7.5-10 nm diameter rigid fibrils (3).
Plasma cell dyscrasias with consequent immunoglobulin light chain accumulation are frequently implicated in the etiology of amyloidosis (AL amyloidosis). Other causes include age-related amyloidosis secondary to normal or abnormal transthyretin (ATTR) and chronic inflammation-related amyloidosis due to acute phase reactants (AA amyloidosis). In fact, dozens of precursor proteins are described (4). Direct toxicity is more common in AL amyloidosis (3), particularly lambda subtype, which is significantly more common than the kappa subtype. AA amyloidosis and ATTR amyloidosis may be more difficult to identify clinically due to lack of known concurrent disease process (e.g., plasma cell dyscrasia in AL amyloidosis). Since symptoms are not specific, a high index of suspicion is needed and appropriate biopsies of affected tissue pursued.
More than 100 transthyretin mutations have been identified and may be pathogenic, neutral, or even protective (5). However, age-related accumulation of wild type transthyretin appears to be more common than any inherited event (6). In the absence of an inexpensive panel assay or positive family history with a known variant, identification of specific defects via molecular analysis remains low yield. However, the African-American population, which is reportedly more vulnerable to amyloidosis after age 60, frequently demonstrates the V221I TTR mutation. In one autopsy series, more than 20% of African-Americans with cardiomyopathy due to amyloidosis demonstrated this mutation (7, 8). Variable penetrance of amyloidosis and significant differences in population prevalence of familial variants compounds the difficulty of the molecular approach to diagnosis.
Clinical presentation is dependent upon the organ(s) involved and may be localized or systemic. The kidneys are most commonly affected and renal failure may occur. Spleen and liver involvement may result in organomegaly. Cardiac amyloidosis is classically described as a restrictive process but may also affect the conduction system and precipitate arrhythmias. Rapezzi et al. described several physical diagnostic clues:
… carpal tunnel syndrome in ATTR (particularly if bilateral in a male), history of unexplained neuropathic pain, orthostatic hypotension, and a diagnosis of 'hypertrophic cardiomyopathy' after the sixth decade (6).
At autopsy, if medical records are available, the presence of these findings may increase the pathologist's index suspicion for ATTR.
Amyloid fibril typing may be accomplished by mass spectrometry, which is commonly performed clinically on endocardial biopsies that have been shown to be Congo red-positive. However, the utility of subtyping amyloid in the postmortem setting is unclear unless a heritable form is identified. The costs of this reflex testing would be prohibitive for most forensic pathology offices.
The forensic autopsy database at the Medical University of South Carolina was searched for cases citing amyloid in the final diagnosis, and these cases were reviewed for demographic information, presentation, autopsy findings and cause and manner of death.
Methods
The electronic database housing all forensic autopsies performed at the Medical University of South Carolina (MUSC) from 2008 to May 15, 2015 was searched using keywords including “amyloid” and “amyloidosis” within either the cause of death statement or the final diagnosis section. Seven cases out of a total of 5055 forensic autopsies were identified and reviewed.
Results
Case 1
A 74-year-old black male presented to the hospital following a fall down three stairs. Multiple rib fractures, Klebsiella oxytoca urinary tract infection, elevated troponin I, decreased urinary output and an elevated creatinine of 2.3 mg/dL (which progressed to 4.4 mg/dL) were noted upon evaluation. Respiratory distress and arrest ensued and he died on hospital day three despite resuscitation attempts. Medical history was significant for congestive heart failure with an ejection fraction of 30%, mitral regurgitation, systemic and pulmonary hypertension, and chronic obstructive pulmonary disease.
Findings at autopsy included a calculated body mass index (BMI) of 26.8 kg/m2, cardiomegaly (650 g) with chamber dilation and diffuse pale brown coloration, and increased ventricular wall thicknesses (left ventricle, 1.7 cm; right ventricle, 0.8 cm; and interventricular septum, 2.3 cm). No significant coronary artery atherosclerosis was present. The lungs weighed 660 g together and bilateral pleural effusions were noted. Microscopy demonstrated cardiac myocyte hypertrophy and patchy, amorphous pink material in the myocardial interstitium (Image 1). This material stained positively with Congo-red and was also present in the pulmonary arterial walls and interstitium, and focally in the renal vasculature. Emphysematous changes of the lungs, focal bridging hepatic fibrosis, and chronic changes of the kidneys (interstitial fibrosis and tubular atrophy) were also described. Toxicology was not performed due to the extended hospitalization and lack of admission specimen. The cause of death was acute renal failure complicating cardiopulmonary amyloidosis. The fall down stairs with resultant rib fractures was considered contributory in this case, presumably due the musculoskeletal injury contributing to renal and pulmonary compromise. Therefore, the manner of death was best classified as accident.
Image 1.
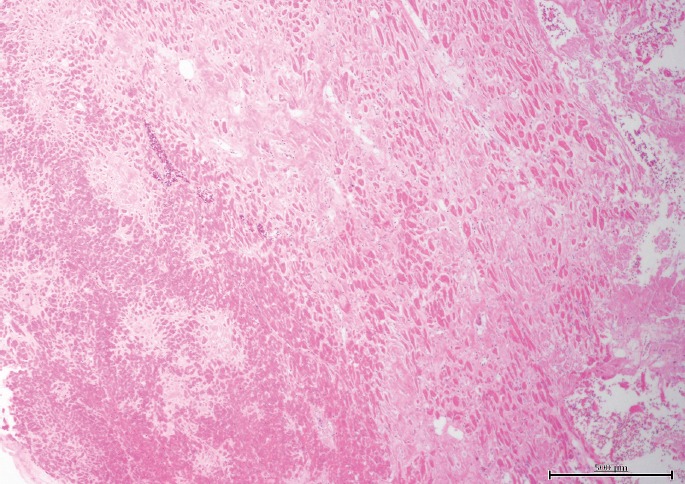
Case 1, heart with patchy, interstitial amyloid pattern (H&E, x40).
Case 2
A 63-year-old white female presented to the hospital with loss of appetite and shortness of breath. Laboratory data demonstrated acidosis, acute renal failure with a serum creatinine of 16.2 mg/dL, urea nitrogen of 142 mg/dL, and anemia for which a red blood cell transfusion was provided. She died within 24 hours of hospitalization.
Findings at autopsy included a BMI of 30.2 kg/m2, cardiomegaly (560 g) with normal appearing parenchyma on sectioning, fibrinous exudate on the pericardial and epicardial surfaces, no significant coronary artery atherosclerosis, and combined lung weight of 1540 g. The ventricular wall thicknesses were increased (left ventricle, 1.7 cm; right ventricle, 0.6 cm; and interventricular septum, 2.0 cm). Additional findings included pleural and peritoneal effusions, cholelithiasis, and colonic diverticula. Histologically, numerous organs sampled, including kidney, heart, liver, thyroid, and lung, demonstrated pink globular material in the blood vessel walls and interstitium. No Congo red stain was performed. Toxicology was not performed due to the length of hospitalization and lack of admission blood sample. The cause of death was acute renal failure due to systemic amyloidosis and the manner was natural.
Case 3
A 64-year-old black male was found dead on the floor of his residence bathroom. Medical history included hypertension, gout and back pain. Autopsy showed a BMI of 28.3 kg/m2, cardiomegaly (640 g) with a normal heart appearance (no wall measurements documented), no significant coronary artery atherosclerosis, edematous and emphysematous lungs with a combined weight of 1490 g, and, cystic “end-stage” kidneys. Small pleural and pericardial effusions were present. Histologically, serofibrinous acute pericarditis, peripheral pulmonary thromboemboli, and cardiac myocyte hypertrophy and interstitial fibrosis were identified (Image 2A). A Congo red stain demonstrated amyloid deposition in lung vessels, myocardium (Image 2B), and renal glomeruli and arterioles. Vitreous creatinine was 11.4 mg/dL. Toxicological analysis of peripheral blood demonstrated presence of delta-9 tetrahydrocannabinol, caffeine, theobromine, and cyclobenzaprine. The cause of death was complications of systemic amyloidosis and hypertensive cardiovascular disease was contributory; the manner was natural.
Image 2A).
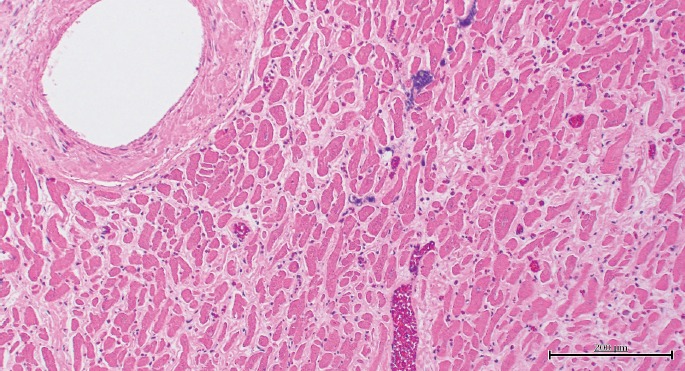
Case 3, heart with interstitial amyloid pattern (H&E, x100).
Image 2B).
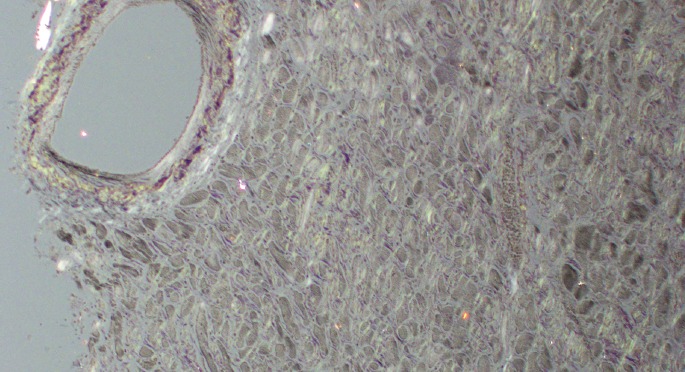
Case 3, heart under polarized light (Congo red, x100).
Case 4
A 72-year-old white female presented with severe fatigue, sweats, body aches, and light-headedness. Upon hospitalization, an abnormal electrocardiogram was noted and troponin was elevated at 0.38 ng/mL. B-type natriuretic protein concentration was 955 pg/mL and the serum creatinine was 1.0 mg/dL. Medical history was significant for hypertension, hypothyroidism, new onset atrial fibrillation, recent rotator cuff surgery, and remote multiple myeloma. She died within 24 hours of admission.
Gross findings at autopsy included a BMI of 18.9 kg/m2, heart weight of 300 g with a normal appearance of the parenchyma, and no significant atherosclerotic coronary artery disease. The combined lung weight was 1430 g. Pleural and peritoneal effusions, pulmonary edema, and severe spinal scoliosis were present. Histologically, amyloidosis confirmed by Congo red stains was in all organs examined, including heart, kidneys (Image 3), lungs (Images 4A and 4B), and spleen. The heart also showed myocytolysis and focal granulation tissue formation. Toxicological analysis of blood revealed the presence of acetaminophen, atenolol, and caffeine. The cause of death was acute myocardial ischemia due to systemic amyloidosis due to multiple myeloma and the manner was natural.
Image 3.
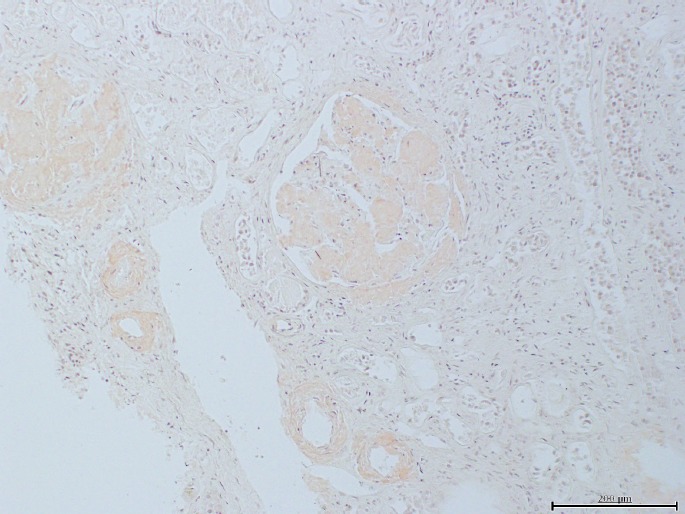
Case 4, renal amyloidosis with vascular and glomerular involvement (Congo red, x100).
Image 4A).
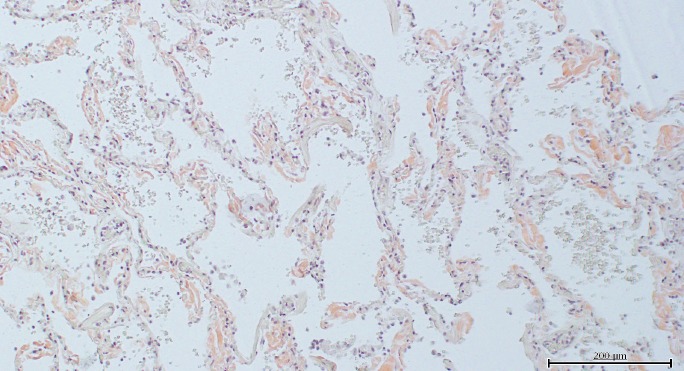
Case 4, lung with alveolar wall involvement (Congo red, x100).
Image 4B).

Case 4, lung under polarized light (Congo red, x100).
Case 5
A 67-year-old black male was found in an early state of decomposition in his residence yard. Medical history was significant for dyspnea upon exertion, hyperlipidemia, and hypertension, and a continuous positive airway pressure machine or CPAP was at the scene. Gross findings at autopsy included a BMI of 29.6 kg/m2, cardiomegaly (810 g), dark red brown and markedly firm and rubbery heart parenchyma, thickened heart wall thicknesses (left ventricle, 1.9 cm; right ventricle, 0.5 cm; and interventricular septum, 1.8 cm), no significant coronary artery atherosclerosis, and a combined lung weight of 1450 g. Bilateral pleural and pericardial effusions recorded. Histology demonstrated amorphous pink substance in the cardiac interstitium (Image 5A), Congo red stain highlighted the heart vessel walls and interstitium as did rhodamine fluorescence (Image 5B). No toxicology was performed. The cause of death was cardiomegaly due to amyloidosis and hypertensive cardiovascular disease was contributory. The manner was natural.
Image 5A).

Case 5, heart with patchy interstitial amyloid pattern (H&E, x100).
Image 5B).

Case 5, heart under rhodamine fluorescence (Congo red, x100).
Case 6
A 70-year-old black male with was found dead in a bathtub without immersion of his face. Medical history included hypertension, diabetes mellitus, and congestive heart failure as well as cocaine use. At autopsy, the BMI was 21.7 kg/m2 and the heart weighed 450 g and appeared normal on sectioning. Coronary artery atherosclerosis impeded the left anterior descending coronary artery lumen by 20% and the right coronary artery lumen by 60%; no heart wall measurements were documented. The combined lung weight was 1260 g and bilateral pleural and peritoneal effusions were noted. Histology of the heart demonstrated perivascular and interstitial fibrosis and granulation tissue as well as patches of pale pink fibers with amorphous pink material in vessel walls that was Congo red-positive (Image 6A and 6B). Renal glomeruli and vessel walls were also Congo red-positive. Vitreous creatinine was 0.7 mg/dL. Toxicological analysis of peripheral blood was positive for cocaine at 29 ng/mL, benzoylecgonine, levamisole, and trazodone at 0.63 μg/mL. The cause of death was acute cocaine toxicity and the manner was accident. Amyloidosis was considered an incidental finding in this case, and not contributory to the cause of death.
Image 6A).
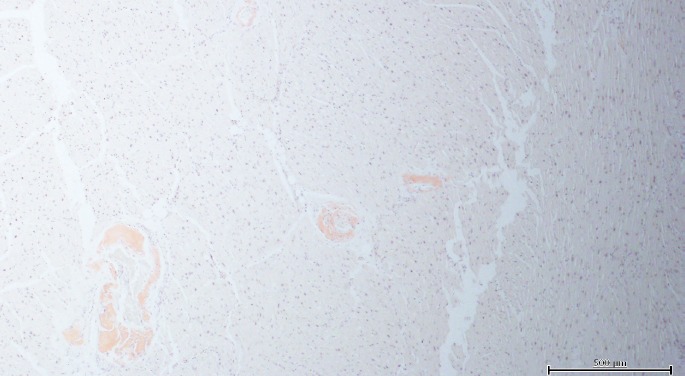
Case 6, cardiac amyloidosis with vascular pattern (Congo red, x40).
Image 6B).

Case 6, heart under polarized light (Congo red, x40).
Case 7
An 81-year-old white male was found dead after a nine foot fall off of a residence balcony. Medical history was significant for hyperlipidemia. Autopsy demonstrated a BMI of 25.8 kg/m2, blunt head trauma with subdural and subarachnoid hemorrhages, cerebral contusions and skull fracture, 300 mL hemopericardium, a superficial myocardial laceration, and sternal and rib fractures. The heart was 430 g and had a normal appearance. Mild coronary artery atherosclerosis and a 2380 g combined lung weight were recorded. Histology showed cardiac myocytolysis and multifocal areas of Congo red positivity in the myocardium surrounding myocytes. Vitreous creatinine was 0.4 mg/dL. Toxicology was positive for caffeine. The cause of death was blunt force trauma to the head due to a fall from height. The manner of death was accident. After review of the scene and circumstances surrounding the death, cardiac amyloidosis was considered contributory in that it presumably precipitated an arrhythmia resulting in the fall. No other pathology, intoxication, psychiatric disorder, or alternative explanation accounted for the decedent's fall from the balcony.
The details for the seven cases regarding presentation, medical history, autopsy findings, and cause and manner of death are summarized in Table 1.
Table 1.
Details For the Seven Cases
| Case | Presentation | Medical History | Cardiac Weight (g), Description | Amyloid | Other findings | Cause of Death | Contributory to Cause of Death | Manner of Death |
|---|---|---|---|---|---|---|---|---|
| 1 | Hospitalized for fall down stairs; elevated troponin; increasing creatinine | Congestive heart failure, ejection fraction=30%, mitral regurgitation, chronic obstructive pulmonary disease, hypertension | 650 Dilated, pale |
Heart Lung Kidney |
Pleural effusions Emphysema Hepatic fibrosis Toxicology not done |
Acute renal failure complicating cardiopulmonary amyloidosis | Fall down stairs with rib fractures | Accident |
| 2 | Hospitalized with loss of appetite, dyspnea, acidosis and renal failure | None |
560 Fibrinous pericarditis |
Heart Lung Kidney |
Pleural effusions Peritoneal effusion Toxicology not done |
Acute renal failure due to systemic amyloidosis | None | Natural |
| 3 | Found dead at home | Hypertension, gout, back pain |
640 Fibrinous pericarditis |
Heart Lung Kidney Spleen |
Pleural effusions Pericardial effusion Cystic kidneys Emphysema Peripheral pulmonary thromboembolism Toxicology: tetrahydrocannibinol, cyclobenzaprine, caffeine, theobromine |
Complications of systemic amyloidosis | Hypertensive cardiovascular disease | Natural |
| 4 | Hospitalized for fatigue, elevated troponin and B-type natriuretic peptide | Hypertension, hypothyroidism, new onset atrial fibrillation, remote history of myeloma | 300 Normal |
Heart Lung Kidney Spleen |
Pleural effusions Peritoneal effusion Pulmonary edema Nephrosclerosis Toxicology: caffeine, acetaminophen, atenolol |
Acute myocardial infarct due to systemic amyloidosis due to multiple myeloma | None | Natural |
| 5 | Found dead in yard with mild decomposition | Dyspnea on exertion, hyperlipidemia, hypertension, sleep apnea | 810 Firm and rubbery |
Heart Lung Kidney |
Pleural effusions Pericardial effusion Toxicology not done |
Cardiomegaly due to amyloidosis | Hypertensive cardiovascular disease | Natural |
| 6 | Found dead at home | Hypertension, congestive heart failure, diabetes, cocaine + | 430 Normal |
Heart Lung |
Pleural effusions Pericardial effusion Peritoneal effusion, mild coronary artery disease Toxicology: cocaine, benzoylecgonine, levamisole, trazodone |
Cocaine toxicity | None | Accident |
| 7 | Fell from 9′ balcony | Hyperlipidemia | 430 Normal |
Heart Lung Kidney |
Hemopericardium Blunt head and chest trauma, Mild coronary artery disease Toxicology: caffeine |
Blunt head trauma due to fall from height | Cardiac amyloidosis | Accident |
Discussion
During a 7.4-year period, only seven cases of amyloidosis were identified in our forensic autopsy population (0.1% of cases). Of these, amyloid was the cause of death or contributory to cause of death in 86% of cases (six of seven) and all demonstrated cardiac involvement. Not surprisingly, the manner of death was classified as natural in the majority of cases. However in the remaining 43% (three of seven), the manner of death was classified as accident. Two accidental deaths were at least partially attributable to blunt trauma sustained in falls and the third accidental death was due to cocaine toxicity with amyloidosis an incidental and noncontributory finding.
The majority (five of seven) of the deaths presented with apparent collapse or a fall with trauma. Sudden collapse is consistent with acute cardiac compromise that may be attributable to cardiac amyloidosis in these cases. Of the two cases presenting with a fall, one resulted in trauma that was nonfatal but considered contributory to the cause of death, and one resulted in fatal trauma that was thought to have been precipitated by a cardiac event secondary to amyloidosis. Cardiac amyloidosis precipitating an arrhythmic event clarified the context of the fall, as other circumstances did not adequately explain such (i.e., case 7).
All decedents were more than 60 years of age. The average age was 71 years with a range from 63 to 81 years. There was a slight African-American predominance (57%) and a male predominance (71%). None of the individuals had a medical history of amyloidosis and only one had a history of multiple myeloma. Only two decedents showed systemic symptoms such that medical attention was sought immediately prior to death. Creatinine levels showed renal dysfunction in three of the six for whom this datum was available.
The average heart weight was 548 g and the range was from 300 to 810 g. Surprisingly, the cardiac parenchyma appeared grossly normal in 71% of cases. No significant atherosclerotic disease was noted in the coronary circulation of any. However, histopathologically, all cases demonstrated involvement of the heart by amyloidosis. The pattern was variable with interstitial, patchy, and vascular deposition patterns noted. All patients showed multiple organ amyloid deposition including lung, kidney, and spleen when these organs were sampled.
Limitations of this series include the small number of cases, lack of toxicological analyses in three of seven cases, and the inability to distinguish a purported cardiac arrhythmia as hypertensive in nature or due to cardiac amyloidosis.
Conclusion
Amyloidosis is a rare cause of sudden unexpected death. Cardiac involvement classically results in a restrictive cardiomyopathy, but patients may also present with a hypertensive, ischemic appearance, or arrhythmia. Renal dysfunction is common. Since the organs may look grossly normal, microscopic sections and special stains may be justified, even in traumatic cases, to fully elucidate the circumstances surrounding the death. Despite the rarity of amyloid in the forensic setting, proper death certification is essential for epidemiological purposes. Additionally, diagnosis may have genetic implications for the family. The authors hope this short series increases the awareness of amyloidosis as a cause or contributory cause of death in a subset of the forensic population.
Footnotes
Disclosures
The authors have indicated that they do not have financial relationships to disclose that are relevant to this manuscript
ETHICAL APPROVAL
As per Journal Policies, ethical approval was not required for this manuscript
STATEMENT OF HUMAN AND ANIMAL RIGHTS
This article does not contain any studies conducted with animals or on living human subjects
STATEMENT OF INFORMED CONSENT
No identifiable personal data were presented in this manuscsript
DISCLOSURES & DECLARATION OF CONFLICTS OF INTEREST
The authors, reviewers, editors, and publication staff do not report any relevant conflicts of interest
References
- 1).Clement C.G., Truong L.D. An evaluation of Congo red fluorescence for the diagnosis of amyloidosis. Hum Pathol. 2014. Aug; 45(8): 1766–72. PMID: 24935061. 10.1016/j.humpath.2014.04.016. [DOI] [PubMed] [Google Scholar]
- 2).Sen S., Basdemir G. Diganosis of renal amyloidosis using Congo red fluorescence. Pathol Int. 2003. Aug; 53(8): 534–8. PMID: 12895232. 10.1046/j.1440-1827.2003.01513.x. [DOI] [PubMed] [Google Scholar]
- 3).Merlini G., Bellotti V. Molecular mechanisms of amyloidosis. N Engl J Med. 2003. Aug 7; 349(6): 583–96. PMID: 12904524. 10.1056/nejmra023144. [DOI] [PubMed] [Google Scholar]
- 4).Sipe J.D., Benson M.D., Buxbaum J.N. et al. Amyloid fibril protein nomenclature: 2012 recommendations from the Nomenclature Committee of the International Society of Amyloidosis. Amyloid. 2012. Dec; 19(4): 167–70. PMID: 23113696. 10.3109/13506129.2012.734345. [DOI] [PubMed] [Google Scholar]
- 5).Sekijima Y. Recent progress in the understanding and treatment of transthyretin amyloidosis. J Clin Pharm Ther. 2014. Jun; 39(3): 225–33. PMID: 24749898. 10.1111/jcpt.12145. [DOI] [PubMed] [Google Scholar]
- 6).Rapezzi C., Lorenzini M., Longhi S. et al. Cardiac amyloidosis: the great pretender. Heart Fail Rev. 2015. Mar; 20(2): 117–24. PMID: 25758359. 10.1007/s10741-015-9480-0. [DOI] [PubMed] [Google Scholar]
- 7).Connors L.H., Prokaeva T., Lim A. et al. Cardiac amyloidosis in African Americans: comparison of clinical and laboratory features of transthyretin V122I amyloidosis and immunoglobulin light chain amyloidosis. Am Heart J. 2009. Oct; 158(4): 607–14. PMID: 19781421. 10.1016/j.ahj.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 8).Jacobson D.R., Pastore R.D., Yaghoubian R. et al. Variant-sequence transthyretin (isoleucine 122) in late-onset cardiac amyloidosis in black Americans. N Engl J Med. 1997. Feb 13; 336(7): 466–73. PMID: 9017939. 10.1056/nejm199702133360703. [DOI] [PubMed] [Google Scholar]


