Abstract
We report a case of acute hemorrhagic leukoencephalitis in an adult man with a prodrome of “feeling unwell” two days prior to this death. At autopsy, external examination revealed minor external injuries including contusions on the scalp and left thigh and abrasions on the forehead and right eyebrow. Gross examination of the brain after coronal sectioning revealed multiple petechial hemorrhages in the white matter in the cerebral hemispheres, corpus callosum, basal ganglia, brainstem, and cerebellum. Microscopic examination of these lesions revealed demyelination, hemorrhage, and necrosis with fibrin exudation in a perivenular distribution with radial extension into the white matter. The remainder of the autopsy was unremarkable. This case highlights the death of a young man by a rare fatal complication of a natural disease only identified by a singular set of gross and microscopic findings at autopsy in circumstances that would otherwise suggest a nonnatural death. The case demonstrates the importance of a thorough autopsy in settings where the clinical history, scene, and circumstances may be misleading or absent.
Keywords: Forensic pathology, Perivenular hemorrhage, Demyelination, Acute disseminated encephalitis (ADEM), Acute hemorrhagic leukoencephalitis (AHL or AHLE)
Introduction
Acute hemorrhagic leukoencephalitis or acute hemorrhagic leucoencephalopathy (AHL or AHLE) is a rare entity that occurs in approximately two percent of patients with acute disseminated encephalomyelitis (ADEM) and is often considered to be a more severe and fulminant form (1). Furthermore, AHL is more commonly diagnosed in young adults (2). (1, 2)
While exact causes are unknown, ADEM and AHL are thought to be delayed hypersensitivity reactions to infections typically of the upper respiratory tract. The formation of immune complexes, consisting of circulating antibodies and myelin antigens, by the immune system in response to infection followed by the deposition of these immune complexes in the brain are thought to result in the hallmarks of AHL including perivenular demyelination, petechial hemorrhages, and tissue necrosis of the white matter (3). Numerous pathogens have been associated with AHL including measles, rubella, varicella, HIV, and some other systemic infections (4).
The clinical presentation of AHL includes multiple neurological signs including meningismus, headaches, seizures, asymmetrical neurological deficits, and coma (5). In clinical settings, magnetic resonance imaging is very useful to identify demyelinating lesions involving white matter sparing the cortex, punctate hemorrhages, and surrounding edema (6). Pathologically, AHL is characterized by rapid perivenular demyelination and diffuse inflammatory and hemorrhagic necrosis of the central nervous system (5, 7). If brought to medical attention, patients are treated with steroids or intravenous immunoglobulin to reduce inflammation as well as antibiotics or antiviral medications if an underlying infection is clinically suspected or confirmed by laboratory testing. Acute disseminated encephalomyelitis patients can recover slowly over four to six weeks with a mortality rate of up to 5% (8). The recovery of neurological function and prognosis for survival are worse for AHL with rapid deterioration and death usually occurring within days to one week after onset of symptoms. The overall mortality rate of AHL is 70% (9, 10).
In this report, we present a case of a young man who was found deceased in his apartment after being reported alive and feeling unwell by his girlfriend two days prior. The only relevant autopsy findings were that of diffuse petechial hemorrhages in the brain, which corresponded to areas of perivenular demyelination, necrosis, and hemorrhage, indicating AHL as the immediate cause of death.
CASE REPORT
Scene of Death
This 36-year-old man was found deceased, lying supine on his bed in his apartment. His last known contact was with his girlfriend two days prior who reported that he was feeling unwell. His medical history included depression, former alcohol abuse, and some reported seizures during the period of alcohol abuse. He was on several medications, including divalproex, venlafaxine, quetiapine, and a Symbicort inhaler.
Autopsy Findings
A complete medicolegal autopsy was performed. The external examination documented a well-nourished male appropriately developed for the reported age. No significant injuries were present. Internal examination revealed that all body cavities were unremarkable with no hemorrhage, blood, or effusion. The cardiovascular system was unremarkable except for mild atherosclerosis of the coronary arteries and aorta and the lungs exhibited congestion and edema. There was no gross or microscopic evidence of infarcts, tumors, or pneumonia. The soft tissues of the neck, tongue, hyoid, larynx, trachea, carina, and bronchi were all unremarkable. All remaining organs and tissues were also unremarkable. There was no evidence of bony, skeletal, or other deep soft tissue injury.
The brain weight was 1370 g and there was no evidence of cerebral edema, hemorrhage, or meningitis. Upon coronal sectioning, the brain showed multiple petechial hemorrhages in the white matter, involving the subcortical white matter, corpus callosum, basal ganglia, brainstem, and the cerebellum (Images 1 and 2). Microscopic examination of the lungs showed anthracosis, congestion, and pigmented macrophages in the alveoli, and the liver showed microsteatosis. Multiple sections of cerebellar cortex, basal ganglia, brainstem, and cerebellar white matter using routine stains (Hematoxylin and Eosin, H&E) showed perivenular hemorrhagic necrosis, necrotic vessels, and fibrinoid necrosis of the vessel walls (Image 3). Frozen sections were taken from affected areas and negative for fat (Oil Red O); special stains for fungus (periodic acid-Schiff and Grocott's methenamine silver) were negative. Multiple sections stained with H&E/luxol fast blue of the cerebellar cortex, basal ganglia, brainstem, and cerebellar white matter showed perivenular demyelination and diffuse hemorrhagic necrosis of the venules and adjacent brain tissue (Image 4). The purpose of submitting frozen tissue for Oil Red O was to exclude central nervous system (CNS) fat embolism. There was no microscopic evidence of parasitic infection such as CNS malaria or toxoplasmosis. Toxicological testing was noncontributory (only low concentrations of acetone and traces of gamma-hydroxybutyrate, betahydroxybutyrate, and codeine were detected in femoral blood). The immediate cause of death was given as acute hemorrhagic leukoencephalitis and the manner of death was certified as natural.
Image 1.
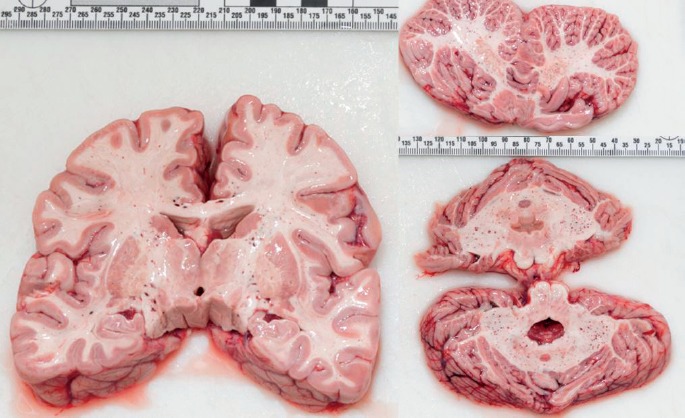
Coronal sections of the unfixed brain demonstrate diffuse petechial hemorrhages involving the white matter of the cerebral cortex, corpus callosum, basal ganglia, and cerebellum.
Image 2.
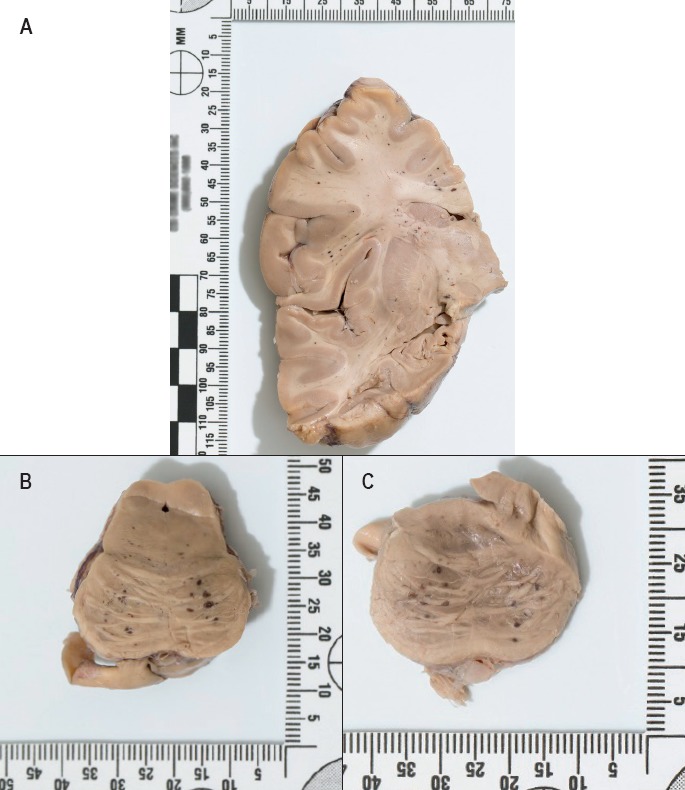
Closer views of the petechial hemorrhages in A) the cerebral cortex, B) brainstem, and C) pons after formalin fixation for 14 days.
Image 3.
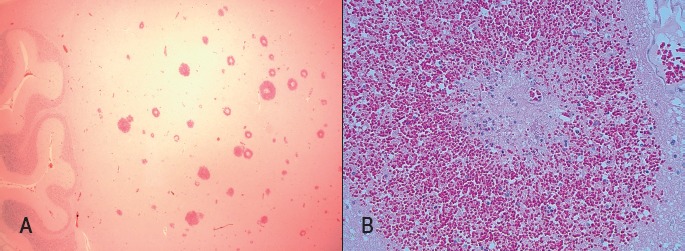
Microscopic sections of white matter of the cerebellum. A) Cerebellar folia on left side and multiple perivenular hemorrhages towards left side (H&E, x12.5). B) Necrotic venule with perivenular necrosis and hemorrhage with a minimal inflammatory infiltrate composed of neutrophils and cell debris (H&E, x200).
Image 4.

Microscopic sections of cerebral cortex at A) 25x magnification and B) 200x magnification stained with luxol fast blue and H&E/luxol fast blue. There is a perivenular distribution of demyelination, hemorrhage, and necrosis with a minimal inflammatory infiltrate.
Discussion
This case shows the importance of thorough gross and microscopic examination at autopsy and the need for the forensic pathologist to recognize rare diagnostic entities that would indicate a natural manner of death in cases where the available history, scene findings, and circumstances are ambiguous. In this particular case, the failure to recognize the gross and microscopic findings of AHL would have led to a more ambiguous determination of the cause and manner of death as the autopsy findings would have essentially been negative. Given the history of possible seizures and medication history of divalproex, sudden unexpected death in epilepsy (SUDEP) would have likely been entertained if AHL had not been recognized. This diagnosis, however, would have been controversial, especially in light that firm documentation of the man's seizure and medication history was unavailable at the time of autopsy.
Acute hemorrhagic leukoencephalitis is a rare diagnosis and it is important for pathologists to recognize the characteristic features of the disease while ruling out other more common differential diagnoses. The three key characteristic features of AHL are: diffuse cerebral edema, brain parenchymal necrosis, and white matter petechial hemorrhages (5). It is important to note that the severity of these characteristics can vary. For example, cerebral edema can range from slight with minimal changes to severe with herniation. In addition, the hemorrhages seen in the white matter may be elusive petechial hemorrhages or could present as an unmistakable hematoma. Histological findings of AHL include perivascular demyelination, fibrinoid perivascular necrosis, widespread perivascular mixed inflammatory infiltrates, and “ring and ball” hemorrhages (5, 7). The presence of all three gross autopsy findings and histological findings will lead to the diagnosis of AHL. Differential diagnoses include ADEM (2, 5) acute necrotizing encephalopathy (ANE) (11), and multiple sclerosis (MS) of Marburg type (3, 5).
Acute disseminated encephalomyelitis and AHL are sometimes thought to be on the same spectrum of demyelinating diseases, with AHL being the more fulminant subtype. The clinical and pathological findings of both disorders are similar and are usually preceded by a viral infection, upper respiratory infection, or vaccination; however, as previously discussed, the severity, mortality rate, and morbidity of AHL are much higher than ADEM (9). Another distinguishing factor between AHL and ADEM is the difference in types of central nervous system infiltrates as neutrophils are seen with AHL and lymphocytes are seen with ADEM (12). In this case, the inflammation was minimal and neutrophilic, favoring AHL.
Acute necrotizing encephalopathy is characterized by multiple symmetrical lesions in the white matter of the brain, usually associated with edematous necrosis and hemorrhage (11). Acute necrotizing encephalopathy also displays similar clinical symptoms and shares a similar etiology with AHL (13). For these reasons, its clinical and pathological features are similar and have the potential to be confused. The differentiating variance among the two disorders is the absence of inflammatory cells in ANE compared to the abundance of inflammatory cells in AHL (11).
Multiple sclerosis of Marburg type is a rare, idiopathic disease afflicting young adults, characterized by inflammatory demyelination and can clinically resemble AHL. Under the microscope, MS shows demyelination with infiltrates similar to AHL; however, MS does not show perivascular hemorrhage nor does it show fibrinoid necrosis of the vessels in the white matter (5). With respect to this case, MS of Marburg type would likely not present as a sudden unexpected death. It is important to exclude fat embolism and parasitic infections of the brain, such as cerebral malaria and toxoplasmosis. There was no evidence of fat embolism or parasitic infection in this case.
Conclusion
It is important for a pathologist to be aware of rare diagnostic entities that could indicate a natural manner of death in a medicolegal autopsy. The recognition of AHL, when the scene and circumstances of death do not point towards it, may be challenging. However, AHL and its differential diagnoses should be considered when a person dies rapidly from an encephalitic mechanism with no previous trauma, negative blood cultures, and negative cerebral spinal fluid studies. It is also important for pathologists to be able to distinguish AHL from other more common disorders that may appear the same clinically or pathologically, but are ultimately of different etiologies.
Footnotes
ETHICAL APPROVAL
As per Journal Policies, ethical approval was not required for this manuscript
STATEMENT OF HUMAN AND ANIMAL RIGHTS
This article does not contain any studies conducted with animals or on living human subjects
STATEMENT OF INFORMED CONSENT
No identifiable personal data were presented in this manuscsript
DISCLOSURES & DECLARATION OF CONFLICTS OF INTEREST
The authors, reviewers, editors, and publication staff do not report any relevant conflicts of interest
FINANCIAL DISCLOSURE The authors have indicated that they do not have financial relationships to disclose that are relevant to this manuscript
References
- 1.Tenembaum S. Chamoles N. Fejerman N.. Acute disseminated encephalomyelitis: a long-term follow-up study of 84 pediatric patients. Neurology. 2002. Oct 22; 59(8): 1224–31. PMID: 12391351. 10.1212/wnl.59.8.1224. [DOI] [PubMed] [Google Scholar]
- 2.Leake J.A. Billman G.F. Nespeca M.P. et al. Pediatric acute hemorrhagic leukoencephalitis: report of a surviving patient and review. Clin Infect Dis. 2002. Mar 1; 34(5): 699–703. PMID: 11810602. 10.1086/338718. [DOI] [PubMed] [Google Scholar]
- 3.Karussis D.. The diagnosis of multiple sclerosis and the various related demyelinating syndromes: a critical review. J Autoimmun. 2014. Feb-Mar; 48-49: 134–42. PMID: 24524923. 10.1016/j.jaut.2014.01.022. [DOI] [PubMed] [Google Scholar]
- 4.Tenembaum S. Chitnis T. Ness J. et al. Acute disseminated encephalomyelitis. Neurology. 2007. Apr 17; 68(16 Suppl 2): S23–36. PMID: 17438235. 10.1212/01.wnl.0000259404.51352.7f. [DOI] [PubMed] [Google Scholar]
- 5.Lann M.A. Lovell M.A. Kleinschmidt-DeMasters B.K.. Acute hemorrhagic leukoencephalitis: a critical entity for forensic pathologists to recognize. Am J Forensic Med Pathol. 2010. Mar; 31(1): 7–11. PMID: 20010289. 10.1097/PAF.0b013e3181c6be92. [DOI] [PubMed] [Google Scholar]
- 6.Tatco V. Elgendy A. et al. Acute haemorrhagic leucoencephalitis. Radiopaedia [Internet]. [cited 2017. Jun 16]. Available from: https://radiopaedia.org/articles/acute-haemorrhagic-leukoencephalitis.
- 7.Gibbs W.N. Kreidie M.A. Kim R.C. Hasso A.N.. Acute hemorrhagic leukoencephalitis: neuroimaging features and neuropathologic diagnosis. J Comput Assist Tomogr. 2005. Sep-Oct; 29(5): 689–93. PMID: 16163044. 10.1097/01.rct.0000173843.82364.db. [DOI] [PubMed] [Google Scholar]
- 8.Menge T. Hemmer B. Nessler S. et al. Acute disseminated encephalomyelitis: an update. Arch Neurol. 2005. Nov; 62(11): 1673–80. PMID: 16286539. 10.1001/archneur.62.11.1673. [DOI] [PubMed] [Google Scholar]
- 9.Archer H. Wall R.. Acute haemorrhagic leukoencephalopathy: two case reports and review of the literature. J Infect. 2003. Feb; 46(2): 133–7. PMID: 12634076. 10.1053/jinf.2002.1096. [DOI] [PubMed] [Google Scholar]
- 10.Davies N.W. Sharief M.K. Howard R.S.. Infection-associated encephalopathies: their investigation, diagnosis, and treatment. J Neurol. 2006. Jul; 253(7): 833–45. PMID: 16715200. 10.1007/s00415-006-0092-4. [DOI] [PubMed] [Google Scholar]
- 11.Neilson D.E.. The interplay of infection and genetics in acute necrotizing encephalopathy. Curr Opin Pediatr. 2010. Dec; 22(6): 751–7. PMID: 21610332. 10.1097/MOP.0b013e3283402bfe. [DOI] [PubMed] [Google Scholar]
- 12.Yildiz Ö Pul R. Raab P. et al. Acute hemorrhagic leukoencephalitis (Weston-Hurst syndrome) in a patient with relapse-remitting multiple sclerosis. J Neuroinflammation. 2015. Sep 17; 12: 175 PMID: 26376717. PMCID: PMC4574135. 10.1186/s12974-015-0398-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong A.M. Simon E.M. Zimmerman R.A. et al. Acute necrotizing encephalopathy of childhood: correlation of MR findings and clinical outcome. AJNR Am J Neuroradiol. 2006. Oct; 27(9): 1919–23. PMID: 17032866. [PMC free article] [PubMed] [Google Scholar]


