Abstract
Prognosticating idiopathic pulmonary fibrosis (IPF) is challenging, in part due to a lack of sensitive biomarkers. A recent article in Thorax described how hyperpolarised xenon magnetic resonance spectroscopy may quantify regional gas exchange in IPF lungs. In a population of patients with IPF, we find that the xenon signal from red blood cells diminishes relative to the tissue/plasma signal over a 12-month time period, even when the diffusion factor for carbon monoxide is static over the same time period. We conclude that hyperpolarised 129Xe MR spectroscopy may be sensitive to short-term changes in interstitial gas diffusion in IPF.
Keywords: idiopathic pulmonary fibrosis, interstitial fibrosis, imaging/ct mri etc, lung physiology
Introduction
Despite significant developments in our understanding of the pathogenesis of idiopathic pulmonary fibrosis (IPF) and the emergence of efficacious treatments,1 2 prognostication remains challenging. Pulmonary function tests (PFTs), including forced vital capacity (FVC) and diffusing capacity of the lungs for carbon monoxide (DLCO), form the basis of assessing progression and treatment response, but are insensitive to longitudinal change.3 An FVC decline of ≥10% is a validated primary endpoint for disease progression and is predictive of mortality,4 but longitudinal data demonstrate substantial intrapatient variability.3 New biomarkers may help to assess disease progression. MRI with hyperpolarised helium (3He) or xenon (129Xe) gas can reveal subtle changes in lung ventilation and microstructure.5 Of particular interest for pulmonary gas exchange assessment is the solubility of xenon in lung parenchyma and blood.6 7 129Xe shows distinct signals from the red blood cells (RBCs) and interstitial tissue/plasma (TP) compartments by virtue of its environment-dependent resonant frequency, which enables evaluation of gas exchange efficiency by MR spectroscopy (MRS). Wang et al recently used 129Xe MRS to identify regions of gas transfer deficit in IPF.8 Herein, we describe preliminary findings evaluating the sensitivity of 129Xe MRS to longitudinal physiological changes in patients with IPF.
Methods
In a study with National Health Service Research Ethics Committee approval, 18 participants with a multidisciplinary diagnosis of IPF were recruited and provided prospective informed written consent. Five patients were taking antifibrotic treatment on recruitment to the study (four on pirfenidone and one on nintedanib). During the study, a further three patients commenced pirfenidone and two patients commenced nintedanib. All underwent baseline imaging and 10 underwent repeated scans 1–3 hours later. Fourteen participants returned at 6 months (195±24 days) and 13 returned at 12 months (361±31 days). Each underwent pulmonary MRS with hyperpolarised 129Xe gas on a 1.5T whole-body MRI scanner. Scans were well tolerated and completed in all, except one participant at the 12-month visit, who completed neither DLCO, nor the MR breath-hold manoeuvre.
129Xe was polarised under regulatory licence and 600 mL of isotopically enriched xenon was balanced with nitrogen to a total inhaled dose of 1 L. Whole lung 129Xe MRS was performed during a 10–15 s breath-hold after inhaling the gas mixture from functional residual capacity. A pulse-acquire sequence was used to acquire whole-lung spectra of 129Xe in the TP and RBC compartments (bandwidth: 1.8 kHz; flip angle: 90°; repetition time: 1 s). Data analysis was performed using MATLAB. After discarding the first spectrum, spectra were averaged and zeroth-order phased, and integrals over the RBC and TP spectral peaks were evaluated to derive the ratio RBC:TP.
PFTs were performed on the same day as MR. Spearman’s r determined the strength of correlations. Friedman tests evaluated the significance of differences in baseline, 6 and 12-month metrics. Intraclass correlation coefficient and Bland-Altman analyses assessed reproducibility of RBC:TP. Two-tailed p values of <0.05 determined statistical significance.
Results
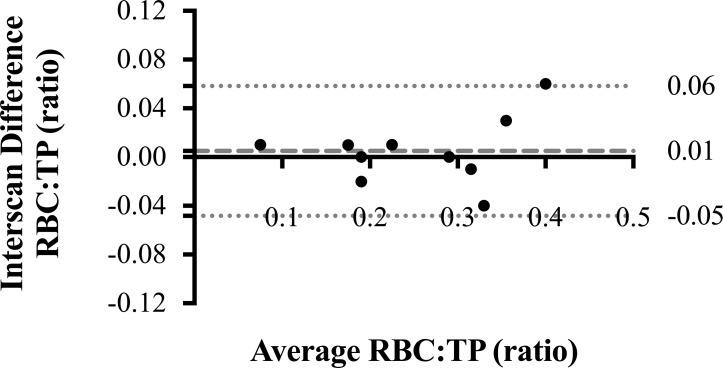
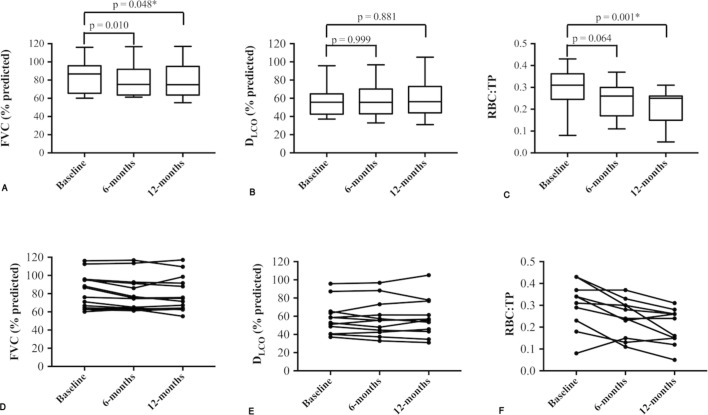
Summary statistics of demographics, PFTs and RBC:TP measurements (median (IQR)) are as follows: age (years) 71.5 (67.8–74.3); FVC (%-predicted) 75.1 (64.3–95.1); DLCO (%-predicted) 42.0 (34.6–56.3); baseline RBC:TP 0.26 (0.18–0.34). Same-day repeat scanning yielded highly reproducible intraparticipant RBC:TP ratios, (see Bland-Altman plot, figure 1; intraclass correlation 0.963). A statistically significant correlation (r; 95% CI) was observed between baseline RBC:TP and DLCO (r=0.677; 0.293 to 0.873), but not FVC (r=0.336; −0.170 to 0.702). Correlations with DLCO (%-predicted) remained significant at 6 months (r=0.831; 0.525 to 0.947) and 12 months (r=0.760; 0.312 to 0.931). Statistically significant changes (median change (95% CI); Friedman p value) were seen over 12 months in FVC (−4.2%, −7.8 to −0.2%; p=0.048) and RBC:TP (−0.08, −0.04 to −0.14; p=0.001), but not DLCO (−0.5%, −5.6% to 5.8%; p=0.881), see figure 2. Although the mean observed decline in FVC was small, five patients demonstrated a relative FVC decline ≥10%, defined as clinically significant.4
Figure 1.
Bland-Altman plot of 129Xe RBC:TP at baseline. Bland-Altman plot of intraparticipant, interscan RBC:TP ratio reproducibility in 10 participants, with the y-axis representing percentage error. Dashed line represents bias, with dotted lines representing 95% CIs. RBC, red blood cell; TP, tissue/plasma.
Figure 2.
Longitudinal change in FVC, DLCO and RBC:TP. (A–C): Box and whisker diagrams with associated Friedman’s test p values, representing 6-month and 12-month follow-up change in FVC, DLCO and 129-xenon MR-derived RBC:TP ratio. Note that a statistically significant difference was not demonstrated in DLCO (B), whereas FVC (A) and RBC:TP (C) declined significantly over 12 months. Individual participants’ metrics of FVC, DLCO and RBC:TP at baseline, 6 and 12 months are plotted in (D), (E) and (F), respectively. DLCO, diffusing capacity of the lungs for carbon monoxide; FVC, forced vital capacity; RBC:TP, red blood cell to tissue/plasma.
Discussion
Hyperpolarised 129Xe MRS is highly reproducible and sensitive to longitudinal changes in pulmonary gas exchange efficiency in IPF. The correlation between RBC:TP and DLCO suggests the techniques reflect similar underlying gas exchange pathophysiology, although with gases that have different membrane diffusion properties. While DLCO showed no longitudinal change, RBC:TP demonstrated a trend to decline over 6 months and a statistically significant decline over 12 months. Our baseline RBC:TP and DLCO correlation is slightly weaker than reported in8 possibly due to differences in MRS parameters, reference equations for DLCO, or inclusion of healthy volunteer data in previous correlations.8
DLCO is calculated from exhaled gas concentrations measured at the mouth, while MRS obtains 129Xe measurements directly from the alveoli, alveolar-capillary membrane and RBCs. The observed reduction in RBC:TP when DLCO is stable may therefore result from differing gas properties and measurement procedure, reflecting different pathophysiology. These differences may be emphasised by the postures used when measuring DLCO (sitting) and 129Xe RBC:TP (supine). The supine posture may cause a partial redistribution of ventilation and perfusion to regions of the lung less affected by disease. Indeed, Wang et al demonstrated how TP signal increases and RBC signal falls in lung tissue afflicted with IPF and how RBC signal is almost absent in areas severely afflicted.8 The 129Xe signal from the tissue and RBC compartments is weighted by a combination of specific ventilation, interstitial thickening, delayed gas diffusion and perfusion deficit. In future 129Xe spectroscopic studies, additional measurement the 129Xe alveolar gas signal as a reference, alongside contrast enhanced perfusion MRI may provide complementary physiological information to help understand the observed RBC:TP decline. Additionally, acquisition of longitudinal CT data could yield insight into the mechanism of RBC:TP change. We also note that inflation level may influence RBC:TP ratio in a non-negligible manner.9 Thus, future studies should consider varying the inhaled xenon gas dose with lung capacity.
Despite relatively limited participant numbers, we were able to identify a statistically significant decline in RBC:TP, indicating the power of this approach. The observed FVC decline is small, but consistent with treatment intervention studies1 2 and suggests a clinically significant decline in lung function and parallel gas exchange impairment. The decline observed in MRS-derived RBC:TP is underpinned by different pathophysiological mechanisms to that in FVC, and thus, RBC:TP may be additive to our current repertoire of physiological assessment tools. With further development, this technique may offer a sensitive endpoint for early-stage clinical trials.
Acknowledgments
The authors would like to thank all members of the POLARIS group. In particular, Leanne Armstrong and Jennifer Rodgers for administrative support.
Footnotes
Contributors: Conception and design: NDW, NJS, SAR, SMB and JMW. MRI experiments: NDW, NJS, H-FC, GN, JMW, GC, MR and HM. Postprocessing and statistical analysis: NDW and NJS. Pulmonary function tests: MA and LJS. Manuscript drafting for intellectual content: All authors. Manuscript editing: All authors.
Funding: Funding for this study was provided by National Institute for Health Research; Grant number: NIHR- RP-R3-12-027 and Medical Research Council; Grant number: MR/M008894/1.
Disclaimer: The views expressed in this publication are those of the authors and not necessarily those of the National Health Service, the National Institute for Health Research or the Department of Health.
Competing interests: None declared.
Patient consent: Not required.
Ethics approval: Liverpool Central NHS REC.
Provenance and peer review: Not commissioned; externally peer reviewed.
Correction notice: This article has been corrected since it was published Online First. The copyright licence was changed from CC BY-NC to CC-BY.
References
- 1. Noble PW, Albera C, Bradford WZ, et al. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomised trials. Lancet 2011;377:1760–9. 10.1016/S0140-6736(11)60405-4 [DOI] [PubMed] [Google Scholar]
- 2. Richeldi L, du Bois RM, Raghu G, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med 2014;370:2071–82. 10.1056/NEJMoa1402584 [DOI] [PubMed] [Google Scholar]
- 3. Nathan SD, Albera C, Bradford WZ, et al. Effect of continued treatment with pirfenidone following clinically meaningful declines in forced vital capacity: analysis of data from three phase 3 trials in patients with idiopathic pulmonary fibrosis. Thorax 2016;71:429–35. 10.1136/thoraxjnl-2015-207011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. King TE, Safrin S, Starko KM, et al. Analyses of efficacy end points in a controlled trial of interferon-gamma1b for idiopathic pulmonary fibrosis. Chest 2005;127:171–7. 10.1378/chest.127.1.171 [DOI] [PubMed] [Google Scholar]
- 5. Fain SB, Korosec FR, Holmes JH, et al. Functional lung imaging using hyperpolarized gas MRI. J Magn Reson Imaging 2007;25:910–23. 10.1002/jmri.20876 [DOI] [PubMed] [Google Scholar]
- 6. Stewart NJ, Leung G, Norquay G, et al. Experimental validation of the hyperpolarized 129Xe chemical shift saturation recovery technique in healthy volunteers and subjects with interstitial lung disease. Magn Reson Med 2015;74:196–207. 10.1002/mrm.25400 [DOI] [PubMed] [Google Scholar]
- 7. Qing K, Ruppert K, Jiang Y, et al. Regional mapping of gas uptake by blood and tissue in the human lung using hyperpolarized xenon-129 MRI. J Magn Reson Imaging 2014;39:346–59. 10.1002/jmri.24181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Wang JM, Robertson SH, Wang Z, et al. Using hyperpolarized 129Xe MRI to quantify regional gas transfer in idiopathic pulmonary fibrosis. Thorax 2018;73:21–8. 10.1136/thoraxjnl-2017-210070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Qing K, Mugler JP, Altes TA, et al. Assessment of lung function in asthma and COPD using hyperpolarized 129Xe chemical shift saturation recovery spectroscopy and dissolved-phase MRI. NMR Biomed 2014;27:1490–501. 10.1002/nbm.3179 [DOI] [PMC free article] [PubMed] [Google Scholar]