Abstract
Aging is related to a number of functional and morphological changes leading to progressive decline of the biological functions of an organism. Reactive Oxygen Species (ROS), released by several endogenous and exogenous processes, may cause important oxidative damage to DNA, proteins, and lipids, leading to important cellular dysfunctions. The imbalance between ROS production and antioxidant defenses brings to oxidative stress conditions and, related to accumulation of ROS, aging-associated diseases. The purpose of this review is to provide an overview of the most relevant data reported in literature on the natural compounds, mainly phytochemicals, with antioxidant activity and their potential protective effects on age-related diseases such as metabolic syndrome, diabetes, cardiovascular disease, cancer, neurodegenerative disease, and chronic inflammation, and possibly lower side effects, when compared to other drugs.
1. Introduction
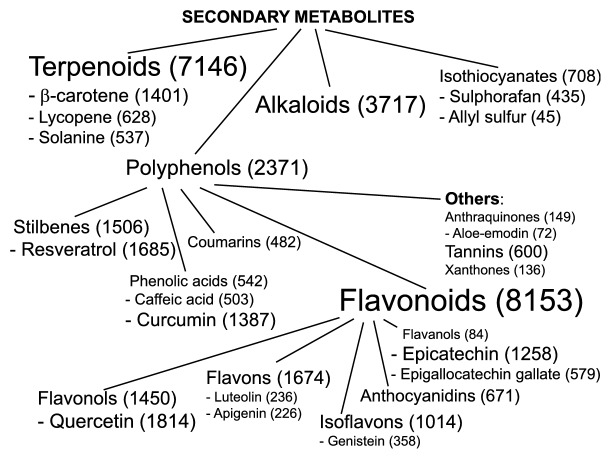
Aging in multicellular organisms is a biological process characterized by the progressive decline of cellular functions and by diminished tissue renewal capability, leading to a reduced ability to counteract the environmental stressors. Aging is controlled by several heterogeneous mechanisms involving genetic, epigenetic, and environmental factors [1, 2]. As largely demonstrated by literature data, aging has a strong relationship with several pathological conditions, including metabolic syndrome, obesity, cardiovascular diseases (CVD), cancer, and neurodegenerative diseases [3, 4]. Interestingly, the age-related pathologies are tightly associated with an increase in Reactive Oxygen Species (ROS) and subsequent oxidative stress [4]. In particular, a large number of studies emphasizes the antioxidant potential of natural compounds (“phytochemicals”) (Figure 1). Since oxidative stress and inflammation are closely related to pathophysiological processes, here we focused our attention on phytochemicals with well-known anti-inflammatory effects. Starting from the large available literature, the most used compounds and with the strongest evidence-based efficacy were summarized in Figure 2.
Figure 1.
A word-tree-cloud showing the body of published evidence indexed on PubMed up today, regarding the most studied phytochemicals as related to oxidative stress. Between brackets, the number of published manuscripts containing the name of each phytochemical and “oxidative stress”, within Title or Abstract, is reported.
Figure 2.
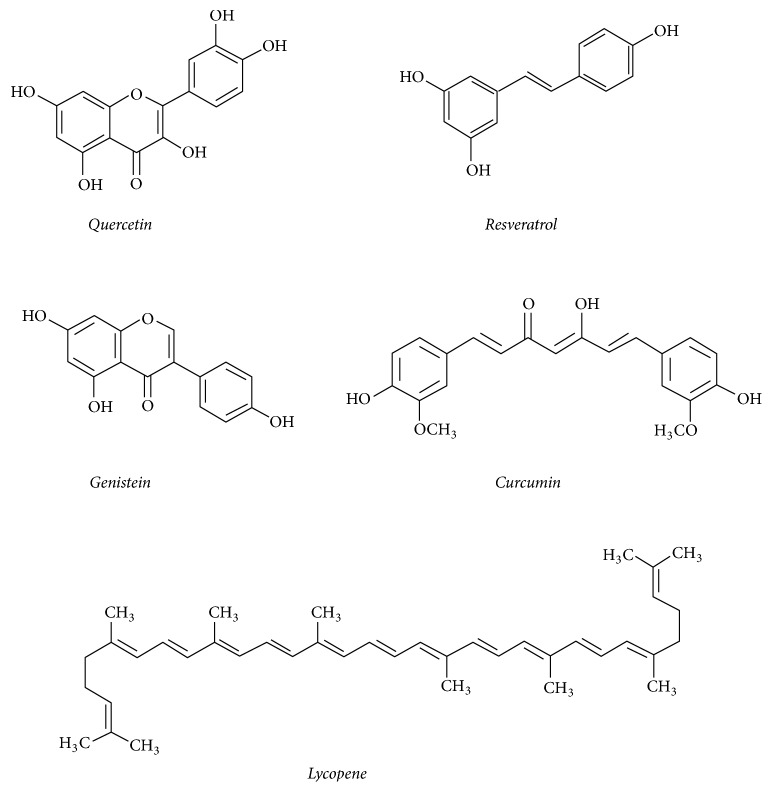
Structures of main secondary plant metabolites with demonstrated antioxidant and anti-inflammatory activity summarized in this review.
2. Plant Secondary Metabolites as Powerful Antioxidant Agents
Phytochemicals are a powerful group of compounds, belonging to secondary metabolites of plants and including a diverse range of chemical entities such as polyphenols, flavonoids, steroidal saponins, organosulphur compounds, and vitamins. They have important roles in plant development, being part of relevant physiological process, i.e., reproduction, symbiotic association, and interactions with other organisms and the environment. Even though most of these compounds occur constitutively, their synthesis can be enhanced under stress conditions, in a manner dependent on the growth conditions and on stressor [16–18]. In plants, following the exposure to stressful conditions, an oxidative burst may cause an imbalance between ROS production and scavenging, leading to the activation of reactive antioxidant enzymatic and non-enzymatic responses [16]. The first one includes changes in the activity of antioxidant enzymes, such as superoxide dismutase, peroxidases, and catalase, while the non-enzymatic response is related to the synthesis of low molecular (ascorbic acid, glutathione, carotenoids, phenolic acids, flavonoids, and others) and high molecular weight antioxidants (tannins).
Many of these plant metabolites have been tested on animal and human cells, showing very interesting biological activities (Table 1). They have been shown to be useful in pharmaceutical applications and in cosmetics, nutrition, and dietary supplements [19]. Plants have been always considered as source of food and medical compounds [20]: actually, up to 200 species are considered as medicinal plants and about 25% of the medicines have plants origins [21]. Most of phytochemicals, components of food, beverages, and herbal products are often reported in literature as “nutraceutical”, emphasizing their health promoting properties, including the prevention and treatment of pathologies like cancer, cardiovascular diseases, neural disorders, and Alzheimer's disease [22], as reported in the following paragraphs. Thus a plethora of information on the effects of phytochemicals on in vitro and in vivo systems is available in the literature. It is worthy to underline that the antioxidant activity of plant metabolites, detected by in vitro assays, does not always correspond to an effective action in vivo. This may be due to different metabolic processes that can affect their antioxidant activity [23] (see also paragraph 3 of this paper); thus some in vitro data have to be considered with caution.
Table 1.
Some of the plant metabolites possessing antioxidant and antitumor activities.
| Secondary metabolites | Common dietary sources | References |
|---|---|---|
| Polyphenols | Fruit, vegetables, coffee, tea, and cereals | [5, 6] |
|
| ||
| Anthocyanins | Strawberries, black rice, berries, cherry etc. | [7, 8] |
|
| ||
| Flavones | Blueberries, blood orange juice | [9] |
|
| ||
| Flavonols | Cherries, chokeberry, elderberries, Goji berry (wolfberry) | [9] |
|
| ||
| Resveratrol | Purple wine, peanuts | [10, 11] |
|
| ||
| Theaflavins | Black tea | [8, 12] |
|
| ||
| Carotenoids | Carrots, tomatoes, pumpkins, peppers, among others | [6, 13] |
|
| ||
| Lycopene | Tomatoes, watermelon, red peppers, papaya, apricot, pink grapefruit | [14, 15] |
The first antioxidant molecule discovered is ascorbic acid, i.e., vitamin C, that is, produced during aerobic metabolism, and reacts rapidly with O2•-, singlet oxygen and ozone (chemically), and H2O2 (enzymatically) through ascorbate peroxidase to neutralize their toxic effects. Besides this, in plants, such acid is also involved in the regeneration of carotenoids and vitamin E (tocopherol). The latter can also act as antioxidant and important liposoluble redox system, providing protection against lipid peroxidation [24]. Data from in vitro studies have shown that β-carotene can regenerate tocopherol from the tocopheroxyl radical; then the resulting carotenoid radical cation may be repaired by vitamin C [25]. The role of vitamins as potent antioxidants has been recently reviewed [26]; therefore, we decided to be more focused on other food-derived phytochemicals.
Carotenoids, responsible for many of the red, orange, and yellow hues of flowers, leaves, and fruits, are powerful antioxidants. Readily available due to their large occurrence in fruits and vegetables (Table 1), their antioxidant activity is based on the scavenging peroxyl radicals [27]. The number of conjugated double bonds of these molecules is related to the efficiency of quenching, i.e., α- and β-carotene, but also zeaxanthin, cryptoxanthin belong to the group of highly active quenchers of 1O2 [25]. For example, lycopene, an intermediate in biosynthetic pathway of carotenoids, acts as scavenger of ROS, lipid peroxyl radicals, and nitric oxide and may exert protective activity against cancer, atherosclerosis, diabetes, and diseases related to inflammatory processes [28]. Synergistic effects in scavenging reactive nitrogen species have been reported for β-carotene and vitamins C and E [25]. While several data show the antioxidant activity of carotenoids, controversy exists about their antioxidant potential in biological systems, since a number of factors may affect their efficacy and, in some cases, prooxidant effects at high concentration and oxygen pressure have been reported [29].
The most promising molecules for further health promoting studies are phenolic compounds. These phytochemicals comprehend a vast range of molecules (about 8000 different structures), playing important roles in the life of plants [30], where they are widely distributed. These compounds can be divided into phenolic acids, lignans, lignins, stilbenes, tannins, and flavonoids, as schematically summarized in Figure 1. Even though they are constitutively present, stressful growth conditions and/or changes of growth medium components may further induce their synthesis and can be used to enhance their production by in vitro plant cultures [31, 32]. In plants, phenolics are involved in H2O2 detoxification, providing protection against UV radiation, also acting as enzyme modulators and feeding deterrents for herbivores [33]. The broad spectrum of biological activities of phenolics, among which antioxidant (i.e., reducing agents, free radical scavenger, and quenchers of single oxygen formation) and antitumor properties, is widely acknowledged in several studies [23, 34]. The presence of at least one phenol ring is important for such activity, with hydroxyl, methyl, or acetyl groups replacing the hydrogen. An increased antioxidant activity has been related to the enhanced number of free hydroxyls and conjugation of side chains to aromatic rings [35].
Flavonoids contain the following subclasses: flavonols, flavones, flavanones, flavan-3-ols, isoflavones, and anthocyanidins. They have attracted the attention of the researchers because of their positive effects on a number of diseases as reported in this review. For instance, quercetin and anthocyanins have been reported to be effective in reducing the growth rate of malignant cells, influencing carcinogen metabolism, reducing parameters of tissue inflammation, and inhibiting angiogenesis [7, 36]. According to some authors, the antitumor activities of phenolic compounds may be related to apoptosis, scavenging of radicals, antioxidant, and prooxidant characteristics [37].
Terpenoids represent another very large family of plant secondary metabolites [38]. In vitro assays showed that monoterpenes, sesquiterpenes, and diterpenes extracted from aromatic plants have notable antioxidant activity [39]. The hypoglycemic and antioxidant activity of the alkaloid vindoline, vindolidine, vindolicine, and vindolinine, obtained from Catharanthus roseus leaves, have been reported [40]. Moreover, vindolicine shows the highest antioxidant effects, and also decreases H2O2-induced oxidative damage to pancreatic cells, suggesting it as a potential antidiabetic agent.
3. Natural Compounds and Inflammation
It is well known that ROS represent physiologic activators of transcription factors, such as Activator Protein-1 (AP-1) or Nuclear Factor κB (NF-κB), which in turn are able to modulate the transcription of proinflammatory cytokines such as Tumor Necrosis Factor α (TNF-α), Interleukin (IL)-6, IL-8, and IL-1 [41]. In fact, ROS, acting as an intracellular signaling component, are associated with inflammatory response and with autoimmune diseases [42]. Therefore, the use of natural products with antioxidant and anti-inflammatory activity represents an intriguing strategy for future clinical applications. These natural compounds have been shown to interfere with various proinflammatory mediators. Herbal medicines, nutraceuticals, which contain food or plant-derived constituents, or functional foods with anti-inflammatory features, can be used as complementary to anti-inflammatory drugs leading to the reduction in dose level of such drugs, thereby reducing their side effects. Phytochemicals with anti-inflammatory activities have been systematically reviewed [43].
A wide range of flavonoids with various chemical structures was associated with different anti-inflammatory mechanistic effects [44]. Glycosides of apigenin and luteolin are the most diffuse flavones. Important edible sources of flavones are parsley, rosemary, and celery [45]. Apigenin suppresses nitric oxide (NO) and prostaglandin production via inhibition of inducible nitric oxide synthase (iNOS) and COX-2, respectively [46]. Luteolin was also proved to inhibit chronic inflammation by in vitro co-culture of adipocytes and macrophages and the phosphorylation of JNK in macrophages [47]. One of the most studied flavonols is quercetin, which may be found in various vegetables and fruits, such as apples. Apple flavonoids have been associated with anti-inflammatory effects. In particular, quercetin and its glycosides were demonstrated to be potent anti-inflammatory agents on sarcoidosis patients and in vivo models of arthritis and allergic airway inflammation [48, 49].
Flavonoids and flavones from fruits of the Citrus spp. inhibit a range of pro-inflammatory mediators, including those derived from the arachidonic acid cascade [50]. In fact, Citrus flavanones (e.g., naringenin) mediate anti-inflammatory effects by modulating neuro-inflammation via interaction with p38 signaling cascades and STAT-1 [51] or suppressing the inflammatory response in an animal model of arthritis when administered orally [52].
Genistein, daidzein, and glycitein are isoflavones found almost exclusively in leguminous plants like soya (Glycine max). Genistein may inhibit inflammation inhibiting NF-κB activation, downregulating TNF-α and IL-6 expression and secretion, endothelin-1, and vascular cell adhesion molecule-1 (VCAM-1) expression [53]. In plants, flavanols can occur as monomers (e.g., catechin and epicatechin) and oligomers or polymers, described as proanthocyanidins or condensed tannins. Catechins interfere with the inflammatory processes that contribute to atherosclerosis progression [54], while, among the effects of flavan-3-ols, present in dietary plants, like tea (Camellia sinensis) and cocoa (Theobroma cacao), inhibition of eicosanoid production, reduction of platelet activation and modulation of NO-dependent mechanisms, and modulation of proinflamamtory cytokine production can be included [55]. Anthocyanins exert their anti-inflammatory effects, particularly via the mitogen-activated protein kinase (MAPK) pathway [56]. Mechanistic studies report the glycosides of malvidin, delphinidin, cyanidin, petunidin, and peonidin to dose-dependently reduce IL-1β -activation of NF-κ B in vitro [57].
Diterpenes from Stevia rebaudiana leaves, used as a source of natural sweeteners in the food industry, have also been shown to attenuate proinflammatory cytokines (TNF-α, IL-1β, and IL-6) production via modulation of the Iκ-Bα /NF-κB pathway [58]. Triterpenes from licorice root (Glycyrrhiza glabra) are glycyrrhizin and glycyrrhetinic acid: they have several effects, including gastric protection and modulation of blood pressure through their mineralocorticoid activity [59], but also anti-inflammatory function acting via the P13K/Akt/GSK3-β pathway to reduce cytokine production, while 18 β-glycyrrhetinic acid also blocks inflammation by causing dissociation of the glucocorticoid receptor [60].
Curcumin was demonstrated to inhibit LPS-induced production of TNF-α and IL-1β in a human monocytic macrophage cell line and, at the same millimolar concentration, to inhibit LPS-induced activation of NF-κB and reduce the biological activity of TNF-α [61].
Stilbenes are found in only low quantities in the human diet. Resveratrol inhibits TNF-α, IL-1β, and IL-6 expression [62] and its ability to suppress NF-κB activation, possibly via SIRT-1 activation, is suggested to be important in counteracting microglial inflammation. Rosmarinic acid is a phenolic acid occurring in herbs such as rosemary and sage. Its anti-inflammatory effects were shown by testing its topical application improving symptoms in atopic dermatitis patients [63].
Chronic inflammation is the main pathogenetic factor of many autoimmune diseases whose treatment is based on long or life-long administration of anti-inflammatory drugs. The possibility to use safe and effective natural products to reduce the dosage and side effects of conventional drugs represents an interesting field of study. Monoclonal antibodies against TNF-α or TNF-α soluble receptors are among the most efficient biological DMARDs (disease-modifying antirheumatic drugs) available for the chronic treatment of rheumatoid arthritis (RA) and other chronic inflammatory diseases. In nature, many natural compounds have been found to reduce the levels of TNF-α by interfering with various pro-inflammatory mediators and upstream targets. Thus, these compounds may represent alternative or adjunctive means of treating inflammatory diseases by modulating production, rather than activity, of TNF-α (reviewed in [64]).
On the other hand, fatty acids and their derivatives were shown to exert profound suppressive effects on the expression of iNOS, COX-II, IL-6, and TNF-α via a blockade of the NF-kB and AP-1 pathways. The strong anti-inflammatory potential and improved clinical parameters of RA of marine n-3 long-chain PUFA were also reported by Barden et al. [65].
Among flavonoids, luteolin and quercetin were the most potent TNF-α inhibitors [66]. The anti-inflammatory power of quercetin was clinically tested in women with RA demonstrating a significant effect in controlling inflammation and clinical symptoms [67]. Irrespective of their activity on the synthesis of TNF-α, green tea extract has also been proven in recent clinical trials to have anti-inflammatory and immunomodulatory properties in autoimmune disease [68].
Therefore, although natural product probably cannot substitute anti-inflammatory drugs, including DMARDs, they could significantly contribute to the reduction of dosage, for a more economic and safer treatment of autoimmune, as well as many other inflammation-related diseases.
4. Oxidative Stress, Metabolic Syndrome and Aging
Aging is a series of morphological and functional changes taking place over the time that can lead to deterioration of the biological functions of an organism [69]. ROS generated as byproducts of biological oxidations can induce severe oxidative damage to macromolecules eventually leading to cellular dysfunction [70]. Combination of aging, insulin resistance, and CVD can precipitate into metabolic syndrome [69, 71]. Although insulin resistance is considered as the main pathogenic mechanism-underlying onset of metabolic syndrome, a low-level chronic inflammation and oxidative stress have received much attention recently [72]. Additionally, evidences from experimental and clinical studies have shown that oxidative stress is a pivotal factor for obesity-associated diabetes, metabolic syndrome, and nonalcoholic steatohepatitis (NASH) [73, 74]. On the other hand, metabolic syndrome is the major health challenge of the 21st century that can significantly affect ever-increasing life and health spans in the developed world. Although the exact mechanism responsible is largely unknown, it is considered that metabolic syndrome can significantly advance aging.
Since the ancient times, natural products have always been suggested to improve longevity of an organism [75]. Epidemiological and experimental data suggest natural products to be powerful antioxidants that can improve stress-related diseases. Scientific literature also suggests potential of natural products in improving metabolic syndrome and aging [76, 77]. Therefore, detailed preclinical evaluation on underlying mechanism of action and bioactivities of the natural compounds may provide solid scientific foundation for clinical applications.
Polyphenols and in particular flavonoids have been shown to protect from various age-related disease [78]. Several studies have indicated that supplementation with dietary polyphenols such as (−)-epigallocatechin-3-gallate (EGCG) and curcumin can improve age-associated cellular damage by reducing generation of ROS [79]. On the other hand, resveratrol and pterostilbene are considered excellent as anti-aging natural compounds that can modulate oxidative damage, inflammation, and cell senescence; components associated with aging [80] as well as flavonoids have been shown to improve aging mainly by controlling metabolic syndrome [81]. Some of the commonly reported flavonoids that can tackle one or more components associated with aging or metabolic syndrome are hesperidin, hesperetin, naringin, and naringenin [82].
It is noteworthly that the literature on preclinical use of phytochemicals to treat various conditions associated with aging is ever expanding. However, a large number of phytochemicals have been tested successfully in clinical trials for age-related condition. It is noteworthy that some limitations of pre-clinical studies that can affect their translational significance are (1) choice of experimental models that is not clinically relevant, (2) poorly characterized mechanism of action, and (3) clinically irrelevant dosing/time points for data interpretation.
5. Natural Compounds and Vascular Diseases
Alteration of vascular function is a key pathogenic process common to many important and highly diffuse human pathologies [83]. The morpho-functional integrity of the vascular endothelium is a complex and highly homeostatic process involving maintenance of vasorelaxation ability as well as anti-inflammatory and barrier functions with important effects on atherogenesis and increased risk of cardiovascular diseases (CVD) [84, 85].
Aging and chronic inflammatory conditions, such as diabetes, alter vascular homeostasis disrupting the “protective” functions of the vascular endothelium, a mechanism known as vascular dysfunction [86]. Physiological aging progressively deteriorates vascular function; however, poor life style, hyperlipidemia, and hyperglycemia associated with oxidative stress can significantly accelerate these pathologic processes leading to CVD and macro- and microvascular complications of diabetes mellitus [87], including ocular pathologies such as ischemic retinopathies. Crucial to the development of vascular dysfunction in aging is the induction of oxidative/nitrative stress [88], which is also involved in the pathogenesis of other human diseases discussed in this review.
Vascular redox imbalance linked to aging and diabetes share important common features such as the induction of the so-called senescence-associated secretory phenotype (SASP) [89]. According to this emerging concept, aging and metabolic stress lead to redox imbalance and trigger enhanced expression of senescence markers and production/secretion of inflammatory cytokines [90, 91]. Suppression of sirtuins, class III NAD+-dependent protein histone deacetylases, is an important feature of SASP [92]. In particular, SIRT-1 plays a key role in maintenance of these cellular homeostasis and energy metabolism [93, 94]. The role of natural compounds as potent anti-oxidant players able to prevent, at least partially, pathologic processes associated with aging and metabolic diseases was recently reviewed [95]. Here we will focus on the therapeutic effects of omega 3 poly-unsaturated fatty acids (PUFA) and the flavonoid resveratrol because of their predominant effects on vascular dysfunction, SASP and CVD.
Omega 3 poly-unsaturated fatty acids (PUFAs) include alpha linoleic acid (18:3) (ALA), eicosapentaenoic acid (EPA) (20:5n-3), and docosaexhaenoic acid (DHA) (22-6n-3). ALA is not synthesized in humans and is considered plant-derived omega 3, whereas EPA and DHA are found predominantly in fish. The effects of omega 3 PUFAs are primarily attributed to their lipid lowering effects and consequent reduction of risk of atherosclerosis [96]. Omega 3 PUFAs have shown to reduce vascular inflammation by downregulating adhesion molecules and limiting leukocytes adhesion to the vascular wall [97]. This latter directly influences production of endothelial-derived nitric oxide due to stabilization of lipid rafts such as the endothelial cells caveolae, as demonstrated in retinal endothelial cells [98]. However, the experimental studies appear to be much more supportive of PUFA's positive effects than the clinical evidence. In fact, the omega 3 PUFA effects on increased endothelial regenerative capacity and maintenance of vascular endothelial cells homeostasis due to membrane stabilizing ability were demonstrated to haveimportant effects on prevention od CVD [98]. An extensive review of the literature, recently appeared on Cochrane Database Systematic Review [99] and summarized the results of a large number of randomized clinical trials assessing the effects of different doses of PUFA on CVD outcomes. The results of this study showed that higher PUFA intake only slightly reduces risk of coronary heart disease and CVD acute events (i.e., stroke) and mortality, but overall has not significant effects on all-cause or cardiovascular disease mortality. Most of the positive effects were associated with modulation of lipid metabolism [99]. In any case, even a slight but significant reduction of 10% of morbidity and mortality for CVD associated with PUFA supplementation remains a significant clinical outcome [100].
The cardioprotective effects of resveratrol in human studies have been reported [101]. However, the in vivo evidence and the clinical studies are less conclusive, mostly because of the poor intestinal absorption of these flavonoids and the extensive degradation in various phenolic acids, which may retain some antioxidant activity [102]. It is noteworthy that one additional function of resveratrol was linked to enhanced SIRT-1 expression and activity and was extensively linked to longevity [103], while a prooxidant activity of resveratrol under certain conditions was reported [104].
Ultimately, the deleterious effects of aging and metabolic diseases (i.e., diabetes) in promoting oxidative stress and vascular dysfunction can negatively affect the cardiovascular system by promoting vascular dysfunction. There is no doubt that a correct life style including a balanced healthy nutrition, physical exercise, and the use of nutraceuticals can positively impact longevity by preventing CVD. However, when CVD pathologies have been established, nutraceuticals such as omega 3 and resveratrol can still find therapeutic application as effective adjuvant therapies because of their numerous positive effects on vascular homeostasis.
Finally, other phytochemicals with antioxidant effects have been reported to play a protective action on the risk or the development of CVD and therefore were proposed as important factors in diet, like, for instance, β-carotene, curcuma, and others [105, 106]. In particular, several studies underline the anti-atherogenic effect of lycopene in association with the inhibition of proinflammatory cytokines secretion [107].
6. Phytochemicals for Neurodegenerative Diseases Prevention
Neurodegenerative diseases (NDDs) are a heterogeneous group of chronic and untreatable conditions, characterized by progressive functional impairment of the nervous system, induced by deterioration of neurons, myelin sheath, neurotransmission, and movement control. Among one of the most disabling of these, Alzheimer's disease (AD) is a NDD that destroys memory and other important mental functions. AD is characterized by the accumulation of amyloid-beta peptide (Aβ) in the brain, the presence of neurofibrillary tangles (NFTs) containing hyperphosphorylated tau fragments, and the loss of cortical neurons and synapses [108]. Parkinson's disease (PD) affects predominately dopaminergic neurons in the substantia nigra, associated with accumulation of Lewy bodies containing α-synuclein in neurons and increased neuroinflammatory cells. Huntington disease (HD) is a progressive brain disorder that causes uncontrolled movements, emotional problems, and loss of thinking ability and occurs in early middle life, even if it is recognized as a juvenile form. HD is an autosomal dominant NDD, characterized by the abnormal expansion of the CAG triplet repeats in the polyglutamine region of the huntingtin (HTT) gene [109]. Multiple sclerosis (MS) is a chronic, autoimmune, inflammatory disease that affects the brain and spinal cord, caused by autoimmune-mediated loss of myelin and axonal damage [110]. At present, there is no effective treatment for NDDs, and, in order to identify novel therapy or adjuvant strategy for NDDs, several natural medicinal plants have gained attention as potential neuroprotective agents, and a number of studies have suggested that a diet rich in vegetable products can prevent or delay the NDDs onset [111]. These properties might be due to the presence of polyphenols, an important group of phytochemicals that are abundantly present in fruits, vegetables, cereals, and beverages (Table 1) and already discussed in other sections of this review. In this chapter, we focused on the potential role of polyphenols for preventive and therapeutic purposes for NDDs treatment, based on related research evidence.
Resveratrol demonstrates significant neuroprotective activity both in vitro and in vivo. Several studies have demonstrated that resveratrol is cytoprotective in cells exposed to Aβ and/or to Aβ-metal complex via Sirt3-mediated mechanisms [112, 113]. In vivo, in a mouse model of cerebral amyloid deposition, orally administered resveratrol lowered microglial activation associated with cortical amyloid plaque formation [114]. Furthermore, long-term dietary resveratrol reduces cognitive impairment and has a neuroprotective role, decreasing the amyloid burden and reducing tau hyperphosphorylation in SAMP8 mice, a model of age-related AD [115]. Increasing evidence has also suggested that resveratrol shows enhanced benefits in cell and animal models of PD. In rat primary midbrain neuron-glia cultures, resveratrol protected dopaminergic neurons against lipopolysaccharide (LPS)-induced neurotoxicity in concentration- and time-dependent manners, through the inhibition of microglial activation and the subsequent reduction of proinflammatory factors release [116]. In vivo, resveratrol ameliorated both motor deficits and pathological changes in MPTP-treated mice via activation of SIRT-1 and subsequent LC3 deacetylation-mediated autophagic degradation of α-synuclein [117]. All the above findings suggest that resveratrol may be a potential prophylactic or therapeutic agent for NDDs, with the caution reported elsewhere in this review regarding intestinal absorption.
Curcuminoids consist of three components: curcumin (75%–80%), demethoxycurcumin (15%–20%), and bisdemethoxycurcumin (3%–5%). Curcumin also induces neuroprotective effects through the control of pathogenetic oxidative and inflammatory mechanisms both in vitro and in vivo models of AD and PD. In Neuro2a mouse neuroblastoma cells infected with Japanese encephalitis virus, curcumin enhances cell viability by decreasing ROS and inhibiting proapoptotic signals [118]. Furthermore, curcumin protects against α-synuclein-induced cytotoxicity in SH-SY5Y neuroblastoma cells decreasing cytotoxicity of aggregated α-synuclein, reducing intracellular ROS, and inhibiting caspase-3 activation [119]. In vivo, curcumin significantly alleviated spatial memory deficits in APP/PS1 mouse model of AD, promoting cholinergic neuronal function [120]. Curcumin also reduced the activation of microglia and astrocytes, as well as cytokine production and inhibited NF-κB signaling pathway, suggesting the beneficial effects of curcumin on AD mice are attributable to the suppression of neuroinflammation [120]. In the PD animal model induced by the neurotoxin MPTP, curcumin is neuroprotective and prevents glutathione depletion and lipid peroxidation induced by this toxin. More recently, curcumin restored motor deficits and enhanced the activities of antioxidant enzymes in rotenone induced Parkinson's in mice [121]. All these findings suggest a neuroprotective role of curcumin, and offer strong justification for the therapeutic prospective of this compound in the management of NDDs.
Pretreatment of primary hippocampal cultures with quercetin significantly attenuated Aβ(1-42)-induced cytotoxicity, protein oxidation, lipid peroxidation, and apoptosis by modulating oxidative stress [122]. More interestingly, quercetin decreases extracellular β-amyloidosis, tauopathy, astrogliosis, and microgliosis in the hippocampus and the amygdala and improves performance on learning and spatial memory tasks in aged triple transgenic Alzheimer's disease model mice [123].
Taken together, the above evidences suggest polyphenols as neuroprotective agents. The habitual consumption of dietary polyphenols is proven to inhibit various secondary sources of ROS and proinflammatory cytokines, thus reducing the risk of NDDs [124]. A beneficial clinical use of polyphenols to attenuate oxidative damage in aging and age-related disorders may be a viable and promising approach for the prevention and treatment of NDDs.
7. Natural Compounds as Anti-Cancer Agents
Although large progress was achieved, some tumors still present poor prognosis and research is currently geared towards the use of non-toxic doses of plant-extracted compounds. The road for a new therapeutic approach, based on natural molecules and drugs, was opened by the identification and use of natural chemotherapeutic agents like taxanes, vinca alkaloids, and anthracyclines [125]. Therefore, it is logical to hypothesize that compounds found in foods are likely to have some protective effects.
Numerous studies suggest that chronic inflammation is able to promote all stages of cancer development, including initiation, progression, and metastatic potential [126]. Moreover, recent data [127] show a close relationship between age-related pathologies, including cancer, and inflammation (Figure 3). ROS are mainly responsible of inflammation and cancer promotion by oxidative stress [128]. In particular, there are increasing evidences on the role of TNF-α, a well-known pro-inflammatory cytokine and a regulator of the generation of ROS, in the promotion of carcinogenesis through the activation of the transcription factor NF-κB [129].
Figure 3.
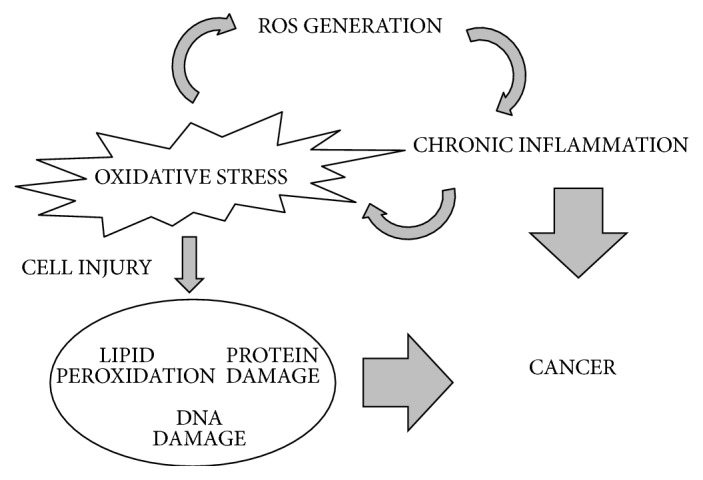
Schematic view of complex cross-talk between oxidative stress, chronic inflammation, and cancer.
Interestingly, recent literature data indicate an emerging role of polyamines metabolism as a novel target against inflammatory diseases. Polyamines are naturally occurring aliphatic compounds, ubiquitous to all living organisms, which interact with DNA, RNA and proteins and are required for eukaryotic cell growth, survival, and differentiation. Notably, excessive polyamine catabolism can lead to ROS formation, increasing oxidative stress, with subsequent enhancement of inflammatory response [130]. For that reason, polyamines metabolism represents an interesting target for anticancer therapy using natural compounds [131]. Therefore, the use of different natural substances, mainly polyphenols, coming from plants and foods, may exert promising results in antitumor therapy due to their anti-oxidant activities [132]. In particular, diet with high polyphenol content has been shown positive effects against cancer-related anorexia/cachexia syndrome and oxidative stress [133].
The flavonol quercetin is a well-known antioxidant molecule with well-documented anticancer activity [134]. Mechanisms underlying its anticancer activity are not completely elucidated; however, it is known that quercetin affects negatively the synthesis of polyamines, well known growth factors, by the inhibition of ornithine decarboxylase (ODC) expression [135]. Interestingly, it has been demonstrated that high flavonols intake, especially quercetin and kaempferol, is able to induce a significative reduction of serum IL-6 concentration, a well-known inflammation-reletated cytokine [136].
Also curcumin shows anti-inflammatory and anti-oxidant properties and potential anti-cancer activity [137]. Indeed, this molecule has been tested in a wide range of cancer cell lines, like cervical cancer [138], colorectal cancer [139], and breast cancer [140]. Moreover, curcumin has been shown to have effects on many signalling and polyamine pathways [141]. Although several studies underline curcumin therapeutic efficacy, its clinical administration is difficult due to its poor oral bioavailability, low solubility, and degradation [142]).
Resveratrol shows a potent cytotoxic effect on cancer cells [143]. For example, recently it was reported that this effect is abolished by Transglutaminase type 2 inhibition on cholangiocarcinoma and gallbladder cancer cell lines [144]. In the last decades, resveratrol has been one of the most studied natural compounds, often leading to contradictory results [145]. Although resveratrol is considered a good candidate as chemopreventive and synergistic agent, further studies are needed [146].
Genistein is a soybean isoflavone. The antitumor activity of genistein has been observed in various forms of cancer such as neuroblastoma and chronic lymphatic leukemia and in several organs such as breast, ovary, prostate, urinary bladder, colon, liver, and stomach [147]. Due to its structural similarity to mammalian estradiol, genistein is known as a phytoestrogen. A large number of studies suggest a beneficial role of this isoflavone in the inhibition of carcinogenesis in animal models. Although the effects of genistein as chemoprevention agent remain controversial [148], several human intervention studies have been undertaken. In fact, it has been demonstrated that dietary soy supplementation may reduce inflammatory processes related to prostate carcinogenesis [149].
One of the most well-known anticancer agents with anti-oxidant and anti-inflammatory properties is lycopene that exerts therapeutic effects on a large variety of cancers [150, 151]. In particular, it has been demonstrated that lycopene intake reduces prostate cancer risk [152].
During the last few decades, it has emerged how genomic instability, telomere attrition and epigenetic changes may underlie aging and senescence phenomena [153]. Telomeres are short tandem repeated sequences (TTAGGG) that are localized on the 5' ends of chromosomes. The length of telomeres is guaranteed by the activity of telomerase enzyme, widely expressed in tumor cells. The G-rich telomeric sequence can assume G-quadruplex DNA secondary structures, able to inhibit telomerase activity. Therefore, inhibition of telomerase or the stabilization of G-quadruplex by natural compounds may represent an important anti-cancer strategy [154, 155].
The identification of natural compounds has contributed to the improvement of cancer therapies and indeed many of these molecules are currently used in clinical practice. It is important, however, to emphasize that rigorous studies and preclinical investigations are needed to clarify their potential chemopreventive and antitumor activities.
8. Phytochemicals in Skin Diseases
According to the European Medicines agency (http://www.ema.europa.eu/ema/), several studies analyze in depth the actual efficacy of herbal medicinal products and derived molecules. Herbal products can be sorted according to their clinical use in several diseases such as appetite disorders, sleep disorders, pain and inflammation control, eye disorders, gastrointestinal disorders, and others (http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/landing/herbal_search.jsp&mid=WC0b01ac058001fa1d for a complete list). We focus here the attention on the beneficial use of herbal medicinal products in skin disorders, with a specific attention on pathologies often related to the skin-aging process [156]. Since in most cases they have been studied or proposed through topical applications, they are often referred to as oils or herbal preparations whose classification in terms of phytochemical content is rather complex.
Therapeutic applications to skin pathologies are proposed for Agrimoniae herba (Agrimonia eupatoria; in minor inflammation and superficial wounds), Echinacea purpurea (in small superficial wounds and mild acne), Soiae oleum (Glycine max; in mild recurrent eczema), Juglandis folium (Juglans regia; in minor skin inflammation), Matricariae aetheroleum (in anous and genitals irritation), Matricariae flos (Matricaria recrutita; in mild skin inflammation and sunburns and superficial wounds), Melaleuca spp. (in insects bites, mild acne, itching, minor skin inflammation), Meliloti herba (Melitotus officinalis; in minor skin inflammation), Origani dictamni herba and Origani majoranae herba (Origanum spp.; in minor skin inflammation and irritation), Rosae flos (Rosa spp.; in skin and mouth inflammation), and Solani dulcamarae stipites (Solanum dulcamara; in itchy and rash from mild eczema) (see http://www.ema.europa.eu/ema/index.jsp?currentCategory=Skin+disorders+%26+minor+wounds&curl=pages%2Fmedicines%2Flanding%2Fherbal_search.jsp&mid=WC0b01ac058001fa1d&searchType=Latin+name+of+herbal+substance&taxonomyPath=&keyword=Enter+keywords&alreadyLoaded=true&treeNumber=&searchkwByEnter=false&pageNo=2 for more details).
The European Medicines Agency official list of herbal substances, preparations, and combinations for use as traditional herbal medicinal products contains 12 substances, according to the European Community decision reported at the EU site (https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02008D0911-20180126).
The 12 herbal substances are as follows: Calendula officinalis, Echinacea purpurea, Eleutherococcus senticosus, Foeniculum vulgare, Hamamelis virginiana, Melaleuca spp., Mentha spp., Pimpinella anisum, Sideritis scardica, Thymus vulgaris, Valeriana officinalis, and Vitis vinifera.
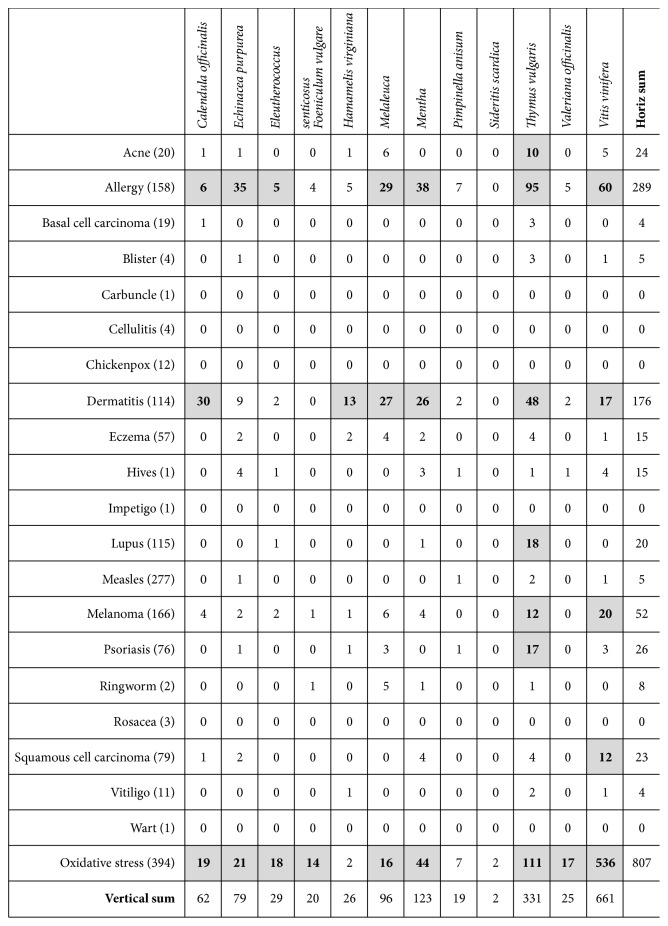
Their introduction within such official list is sustained by scientific reports demonstrating their therapeutic effectiveness in different pathological settings. We collected the scientific literature reported in PubMed for each of these substances co-occurring in the ALL fields with any of the common skin diseases reported in the following list: acne, allergy, basal cell carcinoma, blister, carbuncle, cellulitis, chickenpox, dermatitis, eczema, hives, impetigo, lupus, measles, melanoma, psoriasis, ringworm, rosacea, squamous cell carcinoma, vitiligo, and wart.
As reported in Figure 4, the investigated phytotherapeutic agents are mostly associated to allergy, dermatitis, and melanoma, related in most cases to either Melaleuca, or Mentha or Thymus vulgaris or Vitis vinifera.
Figure 4.
Number of published studies regarding plant-derived nutraceuticals and age-related skin diseases. In the first column, within brackets, the number of PubMed abstracts containing in the Title field the diseases name and the word “age” is reported. In several diseases a strong relation with the age is evident (namely, in allergy, basal cell carcinoma, chickenpox, dermatitis, eczema, lupus, measles, melanoma, psoriasis, squamous cell carcinoma, and oxidative stress). In the other columns, the number of PubMed abstracts containing the diseases name and the phytochemical name in All field is reported. Co-occurrence > 10 is highlighted in bold and gray background. The raw named Oxidative stress reports the number of studies indexed on PubMed containing experimental data which correlate each plant-derived nutraceutical with an “oxidative stress” (as present in All field of database).
More in detail, local application of Melaleuca alternifolia derived oils has been consistently reported to achieve a significant improvement of acne lesions, according to several independent studies [157]. The clinical efficacy of Melaleluca oil is likely related to its known antibacterial action [158] as well as anti-inflammatory activity [159]. The essential oil from Melaleuca alternifolia also shows antioxidant activities potentially useful in dermatitis and skin cancers [160]. Extracts from Melaleuca quinquenervia have been shown to inhibit melanin content in mouse melanoma cells, thus exerting potential cosmetic applications [161]. Minor side effects are associated with Melaleuca such as burning, scaling, itch, redness, dryness, pruritus, and stinging. Contact allergy to Melaleuca oil has been reported in some cases as well as dermatitis reactions indicating the essential oil from Melaleuca as a sensitizers and potentially irritant agent. Mentha-derived oil is used to relief skin inflammation and pruritus [162]; nevertheless, allergic reactions have been reported in some cases.
Finally, regarding Vitis vinifera, a meta-analysis recently published demonstrates Vitis vinifera as one of the effective components of medical devices useful in atopic dermatitis local treatment [163]. Interesting results have also been collected in oncological conditions; Vitis vinifera has shown some efficacy in reducing radiotherapy-induced dermatitis [164] and in inhibiting cell proliferation in melanoma [165, 166] and skin non-melanoma cancer [167], indicating grape seed proanthocyanidin as an apoptosis and autophagy inducer. Rare allergic reactions are reported for Vitis vinifera [168].
Hundreds of other phytochemicals are reported in literature with potential effects on skin, such as anti-age activity [169], photoprotection [170], wound healing [171], and anti-infection [172].
9. Conclusions
The body of evidence reported in this review demonstrates the large interest around phytochemicals and their potential use against oxidative stress-related human diseases, with a focus on those in which inflamed cells play a crucial and pivotal role on pathogenic mechanisms. Their potential use, in combination with drugs like for instance DMARDs, may be very useful to reduce side effects and be cost-effective. Further, socioeconomical issues are playing an even more important role on the rate of new drugs development. This is coupled to an increasing interest toward the repurposing and repositioning of old drugs or others largely used in traditional medicine. Therefore, it is expected that in the next future phytochemicals-based drugs will be object of a growing interest for inflammation and oxidative stress-related diseases.
Acknowledgments
The financial support from Italian Ministry of Health (Ricerca Corrente) is kindly acknowledged.
Disclosure
Giorgia Valle‘s present address is Via Giovanni Spadolini 2, 00046 Grottaferrata (Roma).
Conflicts of Interest
The authors report no conflicts of interest in this work.
References
- 1.Cardelli M. The epigenetic alterations of endogenous retroelements in aging. Mechanisms of Ageing and Development. 2018;174:30–46. doi: 10.1016/j.mad.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 2.Ciccarone F., Tagliatesta S., Caiafa P., Zampieri M. DNA methylation dynamics in aging: how far are we from understanding the mechanisms? Mechanisms of Ageing and Development. 2018;174:3–17. doi: 10.1016/j.mad.2017.12.002. [DOI] [PubMed] [Google Scholar]
- 3.Franceschi C., Garagnani P., Parini P., Giuliani C., Santoro A. Inflammaging: a new immune–metabolic viewpoint for age-related diseases. Nature Reviews Endocrinology. 2018;14(10):576–590. doi: 10.1038/s41574-018-0059-4. [DOI] [PubMed] [Google Scholar]
- 4.Liguori I., Russo G., Curcio F., et al. Oxidative stress, aging, and diseases. Clinical Interventions in Aging. 2018;13:757–772. doi: 10.2147/CIA.S158513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Neto C. C. Cranberry and blueberry: evidence for protective effects against cancer and vascular diseases. Molecular Nutrition & Food Research. 2007;51(6):652–664. doi: 10.1002/mnfr.200600279. [DOI] [PubMed] [Google Scholar]
- 6.Manganaris G. A., Goulas V., Mellidou I., Drogoudi P. Antioxidant phytochemicals in fresh produce: exploitation of genotype variation and advancements in analytical protocols. Frontiers in Chemistry. 2016;5, article 95 doi: 10.3389/fchem.2017.00095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Forni C., Braglia R., Mulinacci N., et al. Antineoplastic activity of Strawberry Crude Extracts on B16-F10 melanoma cells. Molecular BioSystems. 2014;10(6):1255–1263. doi: 10.1039/c3mb70316a. [DOI] [PubMed] [Google Scholar]
- 8.Peng C., Wang X., Chen J., et al. Biology of ageing and role of dietary antioxidants. BioMed Research International. 2014;2014:13. doi: 10.1155/2014/831841.831841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bhagwat S., Haytowitz D. B., Holden J. M. U.S. Department of Agriculture Agricultural Research Service. USDA Database for the Flavonoid Content of Selected Foods. Release 3.1. pp. 1-173, 2014. http://www.ars.usda.gov/nutrientdata.
- 10.Santamaria A. R., Mulinacci N., Valletta A., Innocenti M., Pasqua G. Effects of elicitors on the production of resveratrol and viniferins in cell cultures of vitis vinifera L. cv Italia. Journal of Agricultural and Food Chemistry. 2011;59(17):9094–9101. doi: 10.1021/jf201181n. [DOI] [PubMed] [Google Scholar]
- 11.Pervaiz S., Holme A. L. Resverastrol: its biologic targets and functional activity. Antioxidants & Redox Signaling. 2009;11(11):2851–2897. doi: 10.1089/ars.2008.2412. [DOI] [PubMed] [Google Scholar]
- 12.Takemoto M., Takemoto H. Synthesis of theaflavins and their functions. Molecules. 2018;23(4, article 918) doi: 10.3390/molecules23040918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ranard K. M., Jeon S., Mohn E. S., Griffiths J. C., Johnson E. J., Erdman J. W. Dietary guidance for lutein: consideration for intake reccomendations is scientifically supported. European Journal of Nutrition. 2017;56(S3):537–542. doi: 10.1007/s00394-017-1580-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wood L. G., Garg M. L., Smart J. M., Scott H. A., Barker D., Gibson P. G. Manipulating antioxidant intake in asthma: a randomized controlled trial. American Journal of Clinical Nutrition. 2012;96(3):534–543. doi: 10.3945/ajcn.111.032623. [DOI] [PubMed] [Google Scholar]
- 15.Liu Z., Ren Z., Zhang J., et al. Role of ROS and nutritional antioxidants in human diseases. Frontiers in Physiology. 2018;9, article 477 doi: 10.3389/fphys.2018.00477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mahajan S., Tuteja N. Cold, salinity and drought stresses: an overview. Archives of Biochemistry and Biophysics. 2005;444(2):139–158. doi: 10.1016/j.abb.2005.10.018. [DOI] [PubMed] [Google Scholar]
- 17.Forni C., Duca D., Glick B. R. Mechanisms of plant response to salt and drought stress and their alteration by rhizobacteria. Plant and Soil. 2017;410(1-2):335–356. doi: 10.1007/s11104-016-3007-x. [DOI] [Google Scholar]
- 18.Ramakrishna A., Ravishankar G. A. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signaling and Behavior. 2011;6(11):1720–1731. doi: 10.4161/psb.6.11.17613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hidalgo D., Sanchez R., Lalaleo L., Bonfill M., Corchete P., Palazon J. Biotechnological production of pharmaceuticals and biopharmaceuticals in plant cell and organ cultures. Current Medicinal Chemistry. 2018;25(30):3577–3596. doi: 10.2174/0929867325666180309124317. [DOI] [PubMed] [Google Scholar]
- 20.Phillipson J. D. Phytochemistry and medicinal plants. Phytochemistry. 2001;56(3):237–243. doi: 10.1016/s0031-9422(00)00456-8. [DOI] [PubMed] [Google Scholar]
- 21.Gurnani N., Mehta D., Gupta M., Mehta B. K. Natural products: source of potential drugs. African Journal of Basic & Applied Sciences. 2014;6(6):171–186. [Google Scholar]
- 22.Winter A. N., Brenner M. C., Punessen N., et al. Comparison of the neuroprotective and anti-inflammatory effects of the anthocyanin metabolites, protocatechuic acid and 4-hydroxybenzoic acid. Oxidative Medicine and Cellular Longevity. 2017;2017:13. doi: 10.1155/2017/6297080.6297080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kasote D. M., Katyare S. S., Hegde M. V., Bae H. Significance of antioxidant potential of plants and its relevance to therapeutic applications. International Journal of Biological Sciences. 2015;11(8):982–991. doi: 10.7150/ijbs.12096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Foyer C. H., Noctor G. Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. The Plant Cell. 2005;17(7):1866–1875. doi: 10.1105/tpc.105.033589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stahl W., Sies H. Antioxidant activity of carotenoids. Molecular Aspects of Medicine. 2003;24(6):345–351. doi: 10.1016/S0098-2997(03)00030-X. [DOI] [PubMed] [Google Scholar]
- 26.Ames B. N. Prolonging healthy aging: longevity vitamins and proteins. Proceedings of the National Acadamy of Sciences of the United States of America. 2018;105:10836–10844. doi: 10.1073/pnas.1809045115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jomova K., Valko M. Health protective effects of carotenoids and their interactions with other biological antioxidants. European Journal of Medicinal Chemistry. 2013;70:102–110. doi: 10.1016/j.ejmech.2013.09.054. [DOI] [PubMed] [Google Scholar]
- 28.Petyaev I. M. Lycopene deficiency in ageing and cardiovascular disease. Oxidative Medicine and Cellular Longevity. 2016;2016:6. doi: 10.1155/2016/3218605.3218605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Young A. J., Lowe G. M. Antioxidant and prooxidant properties of carotenoids. Archives of Biochemistry and Biophysics. 2001;385(1):20–27. doi: 10.1006/abbi.2000.2149. [DOI] [PubMed] [Google Scholar]
- 30.Hassan S., Mathesius U. The role of flavonoids in root–rhizosphere signalling: opportunities and challenges for improving plant–microbe interactions. Journal of Experimental Botany. 2012;63(9):3429–3444. doi: 10.1093/jxb/err430. [DOI] [PubMed] [Google Scholar]
- 31.Forni C., Frattarelli A., Lentini A., Beninati S., Lucioli S., Caboni E. Assessment of the antiproliferative activity on murine melanoma cells of extracts from elicited cell suspensions of strawberry, strawberry tree, blackberry and red raspberry. Plant Biosystems. 2016;150(5-6):1233–1239. doi: 10.1080/11263504.2015.1018981. [DOI] [Google Scholar]
- 32.Lucioli S., Di Bari C., Nota P., Frattarelli A., Forni C., Caboni E. Methyl jasmonate promotes anthocyanins production in Prunus salicina × Prunus persica in vitro shoot cultures. Plant Biosystems. 2017;151(5):788–791. doi: 10.1080/11263504.2016.1255267. [DOI] [Google Scholar]
- 33.Bennett R. N., Wallsgrove R. M. Secondary metabolites in plant defence mechanisms. New Phytologist. 1994;127(4):617–633. doi: 10.1111/j.1469-8137.1994.tb02968.x. [DOI] [PubMed] [Google Scholar]
- 34.Pandey K. B., Rizvi S. I. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Medicine and Cellular Longevity. 2009;2(5):270–278. doi: 10.4161/oxim.2.5.9498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moran J. F., Klucas R. V., Grayer R. J., Abian J., Becana M. Complexes of iron with phenolic compounds from soybean nodules and other legume tissues: prooxidant and antioxidant properties. Free Radical Biology & Medicine. 1997;22(5):861–870. doi: 10.1016/S0891-5849(96)00426-1. [DOI] [PubMed] [Google Scholar]
- 36.Bunea A., Ruginã D., Sconta Z., et al. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in B16-F10 metastatic murine melanoma cells. Phytochemistry. 2013;95:436–444. doi: 10.1016/j.phytochem.2013.06.018. [DOI] [PubMed] [Google Scholar]
- 37.Nandi S., Vracko M., Bagchi M. C. Anticancer activity of selected phenolic compounds: QSAR studies using ridge regression and neural networks. Chemical Biology & Drug Design. 2007;70(5):424–436. doi: 10.1111/j.1747-0285.2007.00575.x. [DOI] [PubMed] [Google Scholar]
- 38.Aharoni A., Jongsma M. A., Bouwmeester H. J. Volatile science? Metabolic engineering of terpenoids in plants. Trends in Plant Science. 2005;10(12):594–602. doi: 10.1016/j.tplants.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 39.Baratta M. T., Damien H. J., Deans S. G., Biondi D. M., Ruberto G. Chemical composition, antimicrobial and antioxidative activity of laure, sage, rosemary, oregano essential oils. Journal of Essential Oil Research. 1998;10(6):618–627. doi: 10.1080/10412905.1998.9700989. [DOI] [Google Scholar]
- 40.Tiong S. H., Looi C. Y., Hazni H., et al. Antidiabetic and antioxidant properties of alkaloids from Catharanthus roseus (L.) G. Don. Molecules. 2013;18(8):9770–9784. doi: 10.3390/molecules18089770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nordberg J., Arnér E. S. J. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radical Biology & Medicine. 2001;31(11):1287–1312. doi: 10.1016/S0891-5849(01)00724-9. [DOI] [PubMed] [Google Scholar]
- 42.Phull A.-R., Nasir B., Haq I. U., Kim S. J. Oxidative stress, consequences and ROS mediated cellular signaling in rheumatoid arthritis. Chemico-Biological Interactions. 2018;281:121–136. doi: 10.1016/j.cbi.2017.12.024. [DOI] [PubMed] [Google Scholar]
- 43.Howes M. J. R. Phytochemicals as anti-inflammatory nutraceuticals and phytopharmaceuticals. In: Chatterjee S., Jungraithmayr W., Bagchi D., editors. Immunity and Inflammation in Health and Disease: Emerging Roles of Nutraceuticals and Functional Foods in Immune Support. Cambridge, Mass, USA: Academic Press, Elsevier; 2017. pp. 363–388. [Google Scholar]
- 44.Gomes A., Fernandes E., Lima J. L. F. C., Mira L., Corvo M. L. Molecular mechanisms of anti-inflammatory activity mediated by flavonoids. Current Medicinal Chemistry. 2008;15(16):1586–1605. doi: 10.2174/092986708784911579. [DOI] [PubMed] [Google Scholar]
- 45.King H. G. C. Phenolic compounds of commercial wheat germ. Journal of Food Science. 1962;27(5):446–454. doi: 10.1111/j.1365-2621.1962.tb00125.x. [DOI] [Google Scholar]
- 46.Raso G. M., Meli R., di Carlo G., Pacilio M., di Carlo R. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 expression by flavonoids in macrophage J774A.1. Life Sciences. 2001;68(8):921–931. doi: 10.1016/S0024-3205(00)00999-1. [DOI] [PubMed] [Google Scholar]
- 47.Hirai S., Takahashi N., Goto T., et al. Functional food targeting the regulation of obesity-induced inflammatory responses and pathologies. Mediators of Inflammation. 2010;2010:8. doi: 10.1155/2010/367838.367838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Boots A. W., Drent M., de Boer V. C. J., Bast A., Haenen G. R. M. M. Quercetin reduces markers of oxidative stress and inflammation in sarcoidosis. Clinical Nutrition. 2011;30(4):506–512. doi: 10.1016/j.clnu.2011.01.010. [DOI] [PubMed] [Google Scholar]
- 49.Oliveira R. A. D., Fierro I. M. New strategies for patenting biological medicines used in rheumatoid arthritis treatment. Expert Opinion on Therapeutic Patents. 2018;28(8):635–646. doi: 10.1080/13543776.2018.1502748. [DOI] [PubMed] [Google Scholar]
- 50.Vafeiadou K., Vauzour D., Lee H. Y., Rodriguez-Mateos A., Williams R. J., Spencer J. P. E. The citrus flavanone naringenin inhibits inflammatory signalling in glial cells and protects against neuroinflammatory injury. Archives of Biochemistry and Biophysics. 2009;484(1):100–109. doi: 10.1016/j.abb.2009.01.016. [DOI] [PubMed] [Google Scholar]
- 51.Hämäläinen M., Nieminen R., Vuorela P., Heinonen M., Moilanen E. Anti-inflammatory effects of flavonoids: genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-kappaB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-kappaB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediators of Inflammation. 2007;2007:10. doi: 10.1155/2007/45673.45673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rogerio A. P., Dora C. L., Andrade E. L., et al. Anti-inflammatory effect of quercetin-loaded microemulsion in the airways allergic inflammatory model in mice. Pharmacological Research. 2010;61(4):288–297. doi: 10.1016/j.phrs.2009.10.005. [DOI] [PubMed] [Google Scholar]
- 53.Wang J., Zhang R., Xu Y., Zhou H., Wang B., Li S. Genistein inhibits the development of atherosclerosis via inhibiting NF-kappaB and VCAM-1 expression in LDLR knockout mice. Canadian Journal of Physiology and Pharmacology. 2008;86(11):777–784. doi: 10.1139/Y08-085. [DOI] [PubMed] [Google Scholar]
- 54.Selmi C., Mao T. K., Keen C. L., Schmitz H. H., Eric Gershwin M. The anti-inflammatory properties of cocoa flavanols. Journal of Cardiovascular Pharmacology. 2006;47(2):S163–S171. doi: 10.1097/00005344-200606001-00010. [DOI] [PubMed] [Google Scholar]
- 55.Khoo H. E., Azlan A., Tang S. T., Lim S. M. Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food & Nutrition Research. 2017;61 doi: 10.1080/16546628.2017.1361779.1361779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mena P., Domínguez-Perles R., Gironés-Vilaplana A., Baenas N., García-Viguera C., Villaño D. Flavan-3-ols, anthocyanins, and inflammation. IUBMB Life. 2014;66(11):745–758. doi: 10.1002/iub.1332. [DOI] [PubMed] [Google Scholar]
- 57.Murakami A., Ohnishi K. Target molecules of food phytochemicals: food science bound for the next dimension. Food & Function. 2012;3(5):462–476. doi: 10.1039/c2fo10274a. [DOI] [PubMed] [Google Scholar]
- 58.Momtazi-Borojeni A. A., Esmaeili S.-A., Abdollahi E., Sahebkar A. A review on the pharmacology and toxicology of steviol glycosides extracted from stevia rebaudiana. Current Pharmaceutical Design. 2017;23(11):1616–1622. doi: 10.2174/1381612822666161021142835. [DOI] [PubMed] [Google Scholar]
- 59.Kao T. C., Shyu M. H., Yen G. C. Glycyrrhizic acid and 18beta-glycyrrhetinic acid inhibit inflammation via PI3K/Akt/GSK3beta signaling and glucocorticoid receptor activation. Journal of Agricultural and Food Chemistry. 2010;58(15):8623–8629. doi: 10.1021/jf101841r. [DOI] [PubMed] [Google Scholar]
- 60.Salminen A., Lehtonen M., Suuronen T., Kaarniranta K., Huuskonen J. Terpenoids: natural inhibitors of NF-kappaB signaling with anti-inflammatory and anticancer potential. Cellular and Molecular Life Sciences. 2008;65(19):2979–2999. doi: 10.1007/s00018-008-8103-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liang G., Zhou H., Wang Y., et al. Inhibition of LPS-induced production of inflammatory factors in the macrophages by mono-carbonyl analogues of curcumin. Journal of Cellular and Molecular Medicine. 2009;13(9 B):3370–3379. doi: 10.1111/j.1582-4934.2009.00711.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhang F., Liu J., Shi J.-S. Anti-inflammatory activities of resveratrol in the brain: role of resveratrol in microglial activation. European Journal of Pharmacology. 2010;636(1–3):1–7. doi: 10.1016/j.ejphar.2010.03.043. [DOI] [PubMed] [Google Scholar]
- 63.Lee J., Jung E., Koh J., Kim Y. S., Park D. Effect of rosmarinic acid on atopic dermatitis. The Journal of Dermatology. 2008;35(12):768–771. doi: 10.1111/j.1346-8138.2008.00565.x. [DOI] [PubMed] [Google Scholar]
- 64.Murakami A., Nishizawa T., Egawa K., et al. New class of linoleic acid metabolites biosynthesized by corn and rice lipoxygenases: suppression of proinflammatory mediator expression via attenuation of MAPK- and Akt-, but not PPARgamma-, dependent pathways in stimulated macrophages. Biochemical Pharmacology. 2005;70(9):1330–1342. doi: 10.1016/j.bcp.2005.07.032. [DOI] [PubMed] [Google Scholar]
- 65.Veselinovic M., Vasiljevic D., Vucic V., et al. Clinical Benefits of n-3 PUFA and ɤ-Linolenic Acid in Patients with Rheumatoid Arthritis. Nutrients. 2017;9(4, article E325) doi: 10.3390/nu9040325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Xagorari A., Papapetropoulos A., Mauromatis A., Economou M., Fotsis T., Roussos C. Luteolin inhibits an endotoxin-stimulated phosphorylation cascade and proinflammatory cytokine production in macrophages. The Journal of Pharmacology and Experimental Therapeutics. 2001;296(1):181–187. [PubMed] [Google Scholar]
- 67.Javadi F., Ahmadzadeh A., Eghtesadi S., et al. The effect of quercetin on inflammatory factors and clinical symptoms in women with rheumatoid arthritis: a double-blind, randomized controlled trial. Journal of the American College of Nutrition. 2017;36(1):9–15. doi: 10.1080/07315724.2016.1140093. [DOI] [PubMed] [Google Scholar]
- 68.Shamekhi Z., Amani R., Habibagahi Z., Namjoyan F., Ghadiri A., Saki Malehi A. A randomized, double-blind, placebo-controlled clinical trial examining the effects of green tea extract on systemic lupus erythematosus disease activity and quality of life. Phytotherapy Research. 2017;31(7):1063–1071. doi: 10.1002/ptr.5827. [DOI] [PubMed] [Google Scholar]
- 69.Bonomini F., Rodella L. F., Rezzani R. Metabolic syndrome, aging and involvement of oxidative stress. Aging and Disease (A&D) 2015;6(2):109–120. doi: 10.14336/ad.2014.0305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Harman D. Aging: a theory based on free radical and radiation chemistry. Journal of Gerontology. 1956;11(3):298–300. doi: 10.1093/geronj/11.3.298. [DOI] [PubMed] [Google Scholar]
- 71.Guarner-Lans V., Rubio-Ruiz M. E., Pérez-Torres I., Baños de MacCarthy G. Relation of aging and sex hormones to metabolic syndrome and cardiovascular disease. Experimental Gerontology. 2011;46(7):517–523. doi: 10.1016/j.exger.2011.02.007. [DOI] [PubMed] [Google Scholar]
- 72.Wellen K. E., Hotamisligil G. S. Inflammation, stress, and diabetes. The Journal of Clinical Investigation. 2005;115(5):1111–1119. doi: 10.1172/JCI200525102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Furukawa S., Fujita T., Shimabukuro M., et al. Increased oxidative stress in obesity and its impact on metabolic syndrome. The Journal of Clinical Investigation. 2004;114(12):1752–1761. doi: 10.1172/JCI200421625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Portincasa P., Grattagliano I., Palmieri V. O., Palasciano G. Nonalcoholic steatohepatitis: recent advances from experimental models to clinical management. Clinical Biochemistry. 2005;38(3):203–217. doi: 10.1016/j.clinbiochem.2004.10.014. [DOI] [PubMed] [Google Scholar]
- 75.Dias D. A., Urban S., Roessner U. A Historical Overview of Natural Products in Drug Discovery. Metabolites. 2012;2(4):303–336. doi: 10.3390/metabo2020303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Patti A. M., Al-Rasadi K., Giglio R. V., et al. Natural approaches in metabolic syndrome management. Archives of Medical Science. 2018;14(2):422–441. doi: 10.5114/aoms.2017.68717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tabatabaei-Malazy O., Larijani B., Abdollahi M. Targeting metabolic disorders by natural products. Journal of Diabetes and Metabolic Disorders. 2015;14, article 57 doi: 10.1186/s40200-015-0184-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Truong V.-L., Jun M., Jeong W.-S. Role of resveratrol in regulation of cellular defense systems against oxidative stress. BioFactors. 2018;44(1):36–49. doi: 10.1002/biof.1399. [DOI] [PubMed] [Google Scholar]
- 79.Queen B. L., Tollefsbol T. O. Polyphenols and aging. Current Aging Science. 2010;3(1):34–42. doi: 10.2174/1874609811003010034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Li Y.-R., Li S., Lin C.-C. Effect of resveratrol and pterostilbene on aging and longevity. BioFactors. 2018;44(1):69–82. doi: 10.1002/biof.1400. [DOI] [PubMed] [Google Scholar]
- 81.Prasain J. K., Carlson S. H., Wyss J. M. Flavonoids and age-related disease: Risk, benefits and critical windows. Maturitas. 2010;66(2):163–171. doi: 10.1016/j.maturitas.2010.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Alam M. A., Subhan N., Rahman M. M., Uddin S. J., Reza H. M., Sarker S. D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Advances in Nutrition. 2014;5(4):404–417. doi: 10.3945/an.113.005603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Van Gaal L. F., Mertens I. L., De Block C. E. Mechanisms linking obesity with cardiovascular disease. Nature. 2006;444(7121):875–880. doi: 10.1038/nature05487. [DOI] [PubMed] [Google Scholar]
- 84.Grassi D., Desideri G., Ferri C. Cardiovascular risk and endothelial dysfunction: the preferential route for atherosclerosis. Current Pharmaceutical Biotechnology. 2011;12(9):1343–1353. doi: 10.2174/138920111798281018. [DOI] [PubMed] [Google Scholar]
- 85.Bordy R., Totoson P., Prati C., Marie C., Wendling D., Demougeot C. Microvascular endothelial dysfunction in rheumatoid arthritis. Nature Reviews Rheumatology. 2018;14(7):404–420. doi: 10.1038/s41584-018-0022-8. [DOI] [PubMed] [Google Scholar]
- 86.Gimbrone M. A., García-Cardeña G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circulation Research. 2016;118(4):620–636. doi: 10.1161/circresaha.115.306301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Head T., Daunert S., Goldschmidt-Clermont P. J. The aging risk and atherosclerosis: A fresh look at arterial homeostasis. Frontiers in Genetics. 2017;8, article 216 doi: 10.3389/fgene.2017.00216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lacroix S., Des Rosiers C., Tardif J.-C., Nigam A. The role of oxidative stress in postprandial endothelial dysfunction. Nutrition Research Reviews. 2012;25(2):288–301. doi: 10.1017/s0954422412000182. [DOI] [PubMed] [Google Scholar]
- 89.Wang M., Monticone R. E., Lakatta E. G. Arterial aging: a journey into subclinical arterial disease. Current Opinion in Nephrology and Hypertension. 2010;19(2):201–207. doi: 10.1097/MNH.0b013e3283361c0b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ventura M. T., Casciaro M., Gangemi S., Buquicchio R. Immunosenescence in aging: between immune cells depletion and cytokines up-regulation. Clinical and Molecular Allergy. 2017;15, article 15 doi: 10.1186/s12948-017-0077-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wei W., Ji S. Cellular senescence: molecular mechanisms and pathogenicity. Journal of Cellular Physiology. 2018;233(12):9121–9135. doi: 10.1002/jcp.26956. [DOI] [PubMed] [Google Scholar]
- 92.Hayakawa T., Iwai M., Aoki S., et al. SIRT1 suppresses the senescence-associated secretory phenotype through epigenetic gene regulation. PLoS ONE. 2015;10(1) doi: 10.1371/journal.pone.0116480.e0116480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Sosnowska B., Mazidi M., Penson P., Gluba-Brzózka A., Rysz J., Banach M. The sirtuin family members SIRT1, SIRT3 and SIRT6: Their role in vascular biology and atherogenesis. Atherosclerosis. 2017;265:275–282. doi: 10.1016/j.atherosclerosis.2017.08.027. [DOI] [PubMed] [Google Scholar]
- 94.Zhou M., Luo J., Zhang H. Role of sirtuin 1 in the pathogenesis of ocular disease (review) International Journal of Molecular Medicine. 2018;42(1):13–20. doi: 10.3892/ijmm.2018.3623. [DOI] [PubMed] [Google Scholar]
- 95.Seals D. R., Brunt V. E., Rossman M. J. Keynote lecture: Strategies for optimal cardiovascular aging. American Journal of Physiology-Heart and Circulatory Physiology. 2018;315(2):H183–H188. doi: 10.1152/ajpheart.00734.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wahle K. W. J., Caruso D., Ochoa J. J., Quiles J. L. Olive oil and modulation of cell signaling in disease prevention. Lipids. 2004;39(12):1223–1231. doi: 10.1007/s11745-004-1351-y.L9617 [DOI] [PubMed] [Google Scholar]
- 97.Baker E. J., Yusof M. H., Yaqoob P., Miles E. A., Calder P. C. Omega-3 fatty acids and leukocyte-endothelium adhesion: novel anti-atherosclerotic actions. Molecular Aspects of Medicine. 2018;64:169–181. doi: 10.1016/j.mam.2018.08.002. [DOI] [PubMed] [Google Scholar]
- 98.Colussi G., Catena C., Novello M., Bertin N., Sechi L. A. impact of omega-3 polyunsaturated fatty acids on vascular function and blood pressure: relevance for cardiovascular outcomes. Nutrition, Metabolism & Cardiovascular Diseases. 2017;27(3):191–200. doi: 10.1016/j.numecd.2016.07.011. [DOI] [PubMed] [Google Scholar]
- 99.Abdelhamid A. S., Martin N., Bridges C., et al. Interventions for fatigue and weight loss in adults with advanced progressive illness. Cochrane Database of Systematic Reviews. 2012;7 doi: 10.1002/14651858.CD012345 [DOI] [PubMed] [Google Scholar]
- 100.Kones R., Howell S., Rumana U. N-3 polyunsaturated fatty acids and cardiovascular disease: principles, practices, pitfalls, and promises - a contemporary review. Medical Principles and Practice. 2017;26(6):497–508. doi: 10.1159/000485837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hung L.-M., Chen J.-K., Huang S.-S., Lee R.-S., Su M.-J. Cardioprotective effect of resveratrol, a natural antioxidant derived from grapes. Cardiovascular Research. 2000;47(3):549–555. doi: 10.1016/S0008-6363(00)00102-4. [DOI] [PubMed] [Google Scholar]
- 102.Vitaglione P., Sforza S., Galaverna G., et al. Bioavailability of trans-resveratrol from red wine in humans. Molecular Nutrition & Food Research. 2005;49(5):495–504. doi: 10.1002/mnfr.200500002. [DOI] [PubMed] [Google Scholar]
- 103.Kao C.-L., Chen L.-K., Chang Y.-L., et al. Resveratrol protects human endothelium from H(2)O(2)-induced oxidative stress and senescence via SirT1 activation. Journal of Atherosclerosis and Thrombosis. 2010;17(9):970–979. doi: 10.5551/jat.4333. [DOI] [PubMed] [Google Scholar]
- 104.de La Lastra C. A., Villegas I. Resveratrol as an antioxidant and pro-oxidant agent: mechanisms and clinical implications. Biochemical Society Transactions. 2007;35(5):1156–1160. doi: 10.1042/bst0351156. [DOI] [PubMed] [Google Scholar]
- 105.Gammone M. A., Efthymakis K., Pluchinotta F. R., et al. Impact of chocolate on the cardiovascular health. Frontiers in Bioscience - Landmark. 2018;23(5):852–864. doi: 10.2741/4620. [DOI] [PubMed] [Google Scholar]
- 106.Zhang H., Liu H., Chen Y., Zhang Y. The curcumin-induced vasorelaxation in rat superior mesenteric arteries. Annals of Vascular Surgery. 2018;48:233–240. doi: 10.1016/j.avsg.2017.09.007. [DOI] [PubMed] [Google Scholar]
- 107.Gammone M. A., Pluchinotta F. R., Bergante S., Tettamanti G., D'Orazio N. Prevention of cardiovascular diseases with carotenoids. Frontiers in Bioscience - Scholar. 2017;9(1):165–171. doi: 10.2741/s480. [DOI] [PubMed] [Google Scholar]
- 108.Jeong S. Molecular and cellular basis of neurodegeneration in alzheimer's disease. Molecules and Cells. 2017;40(9):613–620. doi: 10.14348/molcells.2017.0096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Podvin S., Reardon H. T., Yin K., Mosier C., Hook V. Multiple clinical features of Huntington’s disease correlate with mutant HTT gene CAG repeat lengths and neurodegeneration. Journal of Neurology. 2019;266(3):551–564. doi: 10.1007/s00415-018-8940-6. [DOI] [PubMed] [Google Scholar]
- 110.Jones A. P., Kermode A. G., Lucas R. M., et al. Circulating immune cells in multiple sclerosis. Clinical & Experimental Immunology. 2017;187(2):193–203. doi: 10.1111/cei.12878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Joseph J., Cole G., Head E., Ingram D. Nutrition, brain aging, and neurodegeneration. The Journal of Neuroscience. 2009;29(41):12795–12801. doi: 10.1523/jneurosci.3520-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Granzotto A., Zatta P. Resveratrol acts not through Anti-Aggregative pathways but mainly via its scavenging properties against Aβ and Aβ-metal complexes toxicity. PLoS ONE. 2011;6(6) doi: 10.1371/journal.pone.0021565.e21565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Yan W., Liu R., Wang L., et al. Sirt3-mediated autophagy contributes to resveratrol-induced protection against ER stress in HT22 cells. Frontiers in Neuroscience. 2018;12, article 116 doi: 10.3389/fnins.2018.00116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Capiralla H., Vingtdeux V., Zhao H., et al. Resveratrol mitigates lipopolysaccharide- and Aβ-mediated microglial inflammation by inhibiting the TLR4/NF-κB/STAT signaling cascade. Journal of Neurochemistry. 2012;120(3):461–472. doi: 10.1111/j.1471-4159.2011.07594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Porquet D., Casadesús G., Bayod S., et al. Dietary resveratrol prevents alzheimer's markers and increases life span in SAMP8. Age (Dordr) 2013;35(5):1851–1865. doi: 10.1007/s11357-012-9489-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhang F., Shi J.-S., Zhou H., Wilson B., Hong J.-S., Gao H.-M. Resveratrol protects dopamine neurons against lipopolysaccharide-induced neurotoxicity through its anti-inflammatory actions. Molecular Pharmacology. 2010;78(3):466–477. doi: 10.1124/mol.110.064535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Guo Y.-J., Dong S.-Y., Cui X.-X., et al. Resveratrol alleviates MPTP-induced motor impairments and pathological changes by autophagic degradation of α-synuclein via SIRT1-deacetylated LC3. Molecular nutrition & food research. 2016;60(10):2161–2175. doi: 10.1002/mnfr.201600111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Dutta K., Ghosh D., Basu A. Curcumin protects neuronal cells from japanese encephalitis virus-mediated cell death and also inhibits infective viral particle formation by dysregulation of ubiquitin-proteasome system. Journal of Neuroimmune Pharmacology. 2009;4(3):328–337. doi: 10.1007/s11481-009-9158-2. [DOI] [PubMed] [Google Scholar]
- 119.Wang M. S., Boddapati S., Emadi S., Sierks M. R. Curcumin reduces alpha-synuclein induced cytotoxicity in Parkinson’s disease cell model. BMC Neuroscience. 2010;11, article 57:1–10. doi: 10.1186/1471-2202-11-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Liu Z., Li Z., Liu L., et al. Curcumin attenuates beta-amyloid-induced neuroinflammation via activation of peroxisome proliferator-activated receptor-gamma function in a rat model of alzheimer's disease. Frontiers in Pharmacology. 2016;7, article 261 doi: 10.3389/fphar.2016.00261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Khatri D. K., Juvekar A. R. Neuroprotective effect of curcumin as evinced by abrogation of rotenone-induced motor deficits, oxidative and mitochondrial dysfunctions in mouse model of Parkinson's disease. Pharmacology Biochemistry & Behavior. 2016;150-151:39–47. doi: 10.1016/j.pbb.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 122.Ansari M. A., Abdul H. M., Joshi G., Opii W. O., Butterfield D. A. Protective effect of quercetin in primary neurons against Abeta(1-42): relevance to Alzheimer's disease. The Journal of Nutritional Biochemistry. 2009;20(4):269–275. doi: 10.1016/j.jnutbio.2008.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Sabogal-Guáqueta A. M., Muñoz-Manco J. I., Ramírez-Pineda J. R., Lamprea-Rodriguez M., Osorio E., Cardona-Gómez G. P. The flavonoid quercetin ameliorates Alzheimer's disease pathology and protects cognitive and emotional function in aged triple transgenic Alzheimer's disease model mice. Neuropharmacology. 2015;93:134–145. doi: 10.1016/j.neuropharm.2015.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Gardener H., Caunca M. R. Mediterranean diet in preventing neurodegenerative diseases. Current Nutrition Reports. 2018;7(1):10–20. doi: 10.1007/s13668-018-0222-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Nobili S., Lippi D., Witort E., et al. Natural compounds for cancer treatment and prevention. Pharmacological Research. 2009;59(6):365–378. doi: 10.1016/j.phrs.2009.01.017. [DOI] [PubMed] [Google Scholar]
- 126.Shrihari T. Dual role of inflammatory mediators in cancer. ecancermedicalscience. 2017;11, article 721 doi: 10.3332/ecancer.2017.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Leonardi G. C., Accardi G., Monastero R., Nicoletti F., Libra M. Ageing: from inflammation to cancer. Immunity & Ageing. 2018;15, article 1 doi: 10.1186/s12979-017-0112-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Lin S., Li Y., Zamyatnin A. A., Werner J., Bazhin A. V. Reactive oxygen species and colorectal cancer. Journal of Cellular Physiology. 2018;233(7):5119–5132. doi: 10.1002/jcp.26356. [DOI] [PubMed] [Google Scholar]
- 129.Blaser H., Dostert C., Mak T. W., Brenner D. TNF and ROS crosstalk in inflammation. Trends in Cell Biology. 2016;26(4):249–261. doi: 10.1016/j.tcb.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 130.Hussain T., Tan B., Ren W., et al. Polyamines: therapeutic perspectives in oxidative stress and inflammatory diseases. Amino Acids. 2017;49(9):1457–1468. doi: 10.1007/s00726-017-2447-9. [DOI] [PubMed] [Google Scholar]
- 131.Lentini A., Tabolacci C., Provenzano B., Rossi S., Beninati S. Phytochemicals and protein-polyamine conjugates by transglutaminase as chemopreventive and chemotherapeutic tools in cancer. Plant Physiology and Biochemistry. 2010;48(7):627–633. doi: 10.1016/j.plaphy.2010.02.010. [DOI] [PubMed] [Google Scholar]
- 132.Mao X. Y., Jin M. Z., Chen J. F., Zhou H. H., Jin W. L. Live or let die: Neuroprotective and anti-cancer effects of nutraceutical antioxidants. Pharmacology & Therapeutics. 2018;183:137–155. doi: 10.1016/j.pharmthera.2017.10.012. [DOI] [PubMed] [Google Scholar]
- 133.Mantovani G., Madeddu C., Macciò A., et al. Cancer-related anorexia/cachexia syndrome and oxidative stress: an innovative approach beyond current treatment. Cancer Epidemiology, Biomarkers & Prevention. 2004;13(10):1651–1659. [PubMed] [Google Scholar]
- 134.Rauf A., Imran M., Khan I. A., et al. Anticancer potential of quercetin: a comprehensive review. Phytotherapy Research. 2018;32(11):2109–2130. doi: 10.1002/ptr.6155. [DOI] [PubMed] [Google Scholar]
- 135.Linsalata M., Orlando A., Messa C., Refolo M. G., Russo F. Quercetin inhibits human DLD-1 colon cancer cell growth and polyamine biosynthesis. Anticancer Reseach. 2010;30(9):3501–3507. [PubMed] [Google Scholar]
- 136.Bobe G., Albert P. S., Sansbury L. B., et al. Interleukin-6 as a potential indicator for prevention of high-risk adenoma recurrence by dietary flavonols in the polyp prevention trial. Cancer Prevention Research. 2010;3(6):764–775. doi: 10.1158/1940-6207.CAPR-09-0161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Anand P., Kunnumakkara A. B., Newman R. A., Aggarwal B. B. Bioavailability of curcumin: problems and promises. Molecular Pharmaceutics. 2007;4(6):807–818. doi: 10.1021/mp700113r. [DOI] [PubMed] [Google Scholar]
- 138.de Matos R. P. A., Calmon M. F., Amantino C. F., et al. Effect of curcumin-nanoemulsion associated with photodynamic therapy in cervical carcinoma cell lines. BioMed Research International. 2018;2018:11. doi: 10.1155/2018/4057959.4057959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Jalili-Nik M., Soltani A., Moussavi S., et al. Current status and future prospective of Curcumin as a potential therapeutic agent in the treatment of colorectal cancer. Journal of Cellular Physiology. 2018;233(9):6337–6345. doi: 10.1002/jcp.26368. [DOI] [PubMed] [Google Scholar]
- 140.Danafar H., Sharafi A., Kheiri S., Manjili H. K. Co-delivery of sulforaphane and curcumin with pegylated iron oxide-gold core shell nanoparticles for delivery to breast cancer cell line. Iranian Journal of Pharmaceutical Research. 2018;17(2):480–494. [PMC free article] [PubMed] [Google Scholar]
- 141.Murray-Stewart T., Casero R. A. Regulation of by cancer prevention and therapy. Medical Sciencies (Basel) 2017;4(4, article E38) doi: 10.3390/medsci5040038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Kumar S. S. D., Houreld N. N., Abrahamse H. Therapeutic potential and recent advances of curcumin in the treatment of aging-associated diseases. Molecules. 2018;23(4, article E835) doi: 10.3390/molecules23040835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Carter L. G., D'Orazio J. A., Pearson K. J. Resveratrol and cancer: focus on in vivo evidence. Endocrine-Related Cancer. 2014;21(3):R209–R225. doi: 10.1530/erc-13-0171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Roncoroni L., Elli L., Braidotti P., et al. Transglutaminase 2 mediates the cytotoxicity of resveratrol in a human cholangiocarcinoma and gallbladder cancer cell lines. Nutrition and Cancer. 2018;70(5):761–769. doi: 10.1080/01635581.2018.1470648. [DOI] [PubMed] [Google Scholar]
- 145.Pezzuto J. M. Resveratrol: twenty years of growth, development and controversy. Biomolecules & Therapeutics (Seoul) 2019;27:1–14. doi: 10.4062/biomolther.2018.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Xiao Q., Zhu W., Feng W., et al. A review of resveratrol as a potent chemoprotective and synergistic agent in cancer chemotherapy. Frontiers in Pharmacology. 2019;9, article 1534 doi: 10.3389/fphar.2018.01534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Mukund V., Mukund D., Sharma V., Mannarapu M., Alam A. Genistein: Its role in metabolic diseases and cancer. Critical Review in Oncology/Hematology. 2017;119:13–22. doi: 10.1016/j.critrevonc.2017.09.004. [DOI] [PubMed] [Google Scholar]
- 148.Kim S. H., Kim C. W., Jeon S. Y., Go R. E., Hwang K. A., Choi K. C. Chemopreventive and chemotherapeutic effects of genistein, a soy isoflavone, upon cancer development and progression in preclinical animal models. Laboratory Animal Research. 2014;30(4):143–150. doi: 10.5625/lar.2014.30.4.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Lesinski G. B., Reville P. K., Mace T. A., et al. Consumption of soy isoflavone enriched bread in men with prostate cancer is associated with reduced proinflammatory cytokines and immunosuppressive cells. Cancer Prevention Research. 2015;8(11):1036–1044. doi: 10.1158/1940-6207.CAPR-14-0464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Carini F., David S., Tomasello G., et al. Colorectal cancer: an update on the effects of lycopene on tumor progression and cell proliferation. Journal of Biological Regulators & Homeostatic Agents. 2017;31:769–774. [PubMed] [Google Scholar]
- 151.Jiang L., Liu Y., Li B. Lycopene exerts anti-inflammatory effect to inhibit prostate cancer progression. Asian Journal of Andrology. 2019;21(1):p. 80. doi: 10.4103/aja.aja_70_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Rowles J. L., Ranard K. M., Smith J. W., An R., Erdman J. W. Increased dietary and circulating lycopene are associated with reduced prostate cancer risk: a systematic review and meta-analysis. Prostate Cancer and Prostatic Diseases. 2017;20(4):361–377. doi: 10.1038/pcan.2017.25. [DOI] [PubMed] [Google Scholar]
- 153.Lidzbarsky G., Gutman D., Shekhidem H. A., Sharvit L., Atzmon G. Genomic instabilities, cellular senescence, and aging: in vitro, in vivo and aging-like human syndromes. Frontiers in Medicine. 2018;5, article 104 doi: 10.3389/fmed.2018.00104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Artese A., Costa G., Ortuso F., Parrotta L., Alcaro S. Identification of new natural DNA G-quadruplex binders selected by a structure-based virtual screening approach. Molecules. 2013;18(10):12051–12070. doi: 10.3390/molecules181012051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Ait-Ghezala G., Hassan S., Tweed M., et al. Identification of telomerase-activating blends from naturally occurring compounds. Alternative Therapies in Health and Medicine. 2016;2(2):6–14. [PubMed] [Google Scholar]
- 156.Ganceviciene R., Liakou A. I., Theodoridis A., Makrantonaki E., Zouboulis C. C. Skin anti-aging strategies. Dermato-Endocrinology. 2012;4(3):308–319. doi: 10.4161/derm.22804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Hammer K. A. Treatment of acne with tea tree oil (melaleuca) products: a review of efficacy, tolerability and potential modes of action. International Journal of Antimicrobial Agents. 2015;45(2):106–110. doi: 10.1016/j.ijantimicag.2014.10.011. [DOI] [PubMed] [Google Scholar]
- 158.Kwon H. H., Yoon J. Y., Park S. Y., Min S., Suh D. H. Comparison of clinical and histological effects between lactobacillus-fermented chamaecyparis obtusa and tea tree oil for the treatment of acne: an eight-week double-blind randomized controlled split-face study. Dermatology. 2014;229(2):102–109. doi: 10.1159/000362491. [DOI] [PubMed] [Google Scholar]
- 159.Ramage G., Milligan S., Lappin D. F., et al. Antifungal, cytotoxic, and immunomodulatory properties of tea tree oil and its derivative components: Potential role in management of oral candidosis in cancer patients. Frontiers in Microbiology. 2012;3, article 220 doi: 10.3389/fmicb.2012.00220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Pazyar N., Yaghoobi R., Bagherani N., Kazerouni A. A review of applications of tea tree oil in dermatology. International Journal of Dermatology. 2013;52(7):784–790. doi: 10.1111/j.1365-4632.2012.05654.x. [DOI] [PubMed] [Google Scholar]
- 161.Chao W.-W., Su C.-C., Peng H.-Y., Chou S.-T. Melaleuca quinquenervia essential oil inhibits α-melanocyte-stimulating hormone-induced melanin production and oxidative stress in B16 melanoma cells. Phytomedicine. 2017;34:191–201. doi: 10.1016/j.phymed.2017.08.024. [DOI] [PubMed] [Google Scholar]
- 162.Herro E., Jacob S. E. Mentha piperita (peppermint) Dermatitis. 2010;21(6):327–329. doi: 10.2310/6620.2011.10080. [DOI] [PubMed] [Google Scholar]
- 163.Micali G., Paternò V., Cannarella R., Dinotta F., Lacarrubba F. Evidence-based treatment of atopic dermatitis with topical moisturizers. Giornale Italiano di Dermatologia e Venereologia. 2018;153(3):396–402. doi: 10.23736/S0392-0488.18.05898-4. [DOI] [PubMed] [Google Scholar]
- 164.Di Franco R., Sammarco E., Calvanese M. G., et al. Preventing the acute skin side effects in patients treated with radiotherapy for breast cancer: the use of corneometry in order to evaluate the protective effect of moisturizing creams. Journal of Radiation Oncology. 2013;8, article 57 doi: 10.1186/1748-717x-8-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Turk S., Malkan U. Y., Ghasemi M., et al. Growth inhibitory activity of Ankaferd hemostat on primary melanoma cells and cell lines. SAGE Open Medicine. 2017;5:p. 205031211668951. doi: 10.1177/2050312116689519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Tartaglione L., Gambuti A., De Cicco P., et al. NMR-based phytochemical analysis of vitis vinifera cv Falanghina leaves. characterization of a previously undescribed biflavonoid with antiproliferative activity. Fitoterapia. 2018;125:13–17. doi: 10.1016/j.fitote.2017.12.009. [DOI] [PubMed] [Google Scholar]
- 167.Hah Y.-S., Kim J. G., Cho H. Y., Park J. S., Heo E. P., Yoon T.-J. Procyanidins from vitis vinifera seeds induce apoptotic and autophagic cell death via generation of reactive oxygen species in squamous cell carcinoma cells. Oncology Letters. 2017;14(2):1925–1932. doi: 10.3892/ol.2017.6422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Brito F. F., Gimeno P. M., Bartolomé B., et al. Vine pollen allergy in areas with a high density of vineyards. Annals of Allergy, Asthma & Immunology. 2008;100(6):596–600. doi: 10.1016/S1081-1206(10)60054-3. [DOI] [PubMed] [Google Scholar]
- 169.Kalyana Sundaram I., Sarangi D. D., Sundararajan V., George S., Sheik Mohideen S. Poly herbal formulation with anti-elastase and anti-oxidant properties for skin anti-aging. BMC Complementary and Alternative Medicine. 2018;18(1, article 33) doi: 10.1186/s12906-018-2097-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Prasad R., Singh T., Katiyar S. K. Honokiol inhibits ultraviolet radiation-induced immunosuppression through inhibition of ultraviolet-induced inflammation and DNA hypermethylation in mouse skin. Scientific Reports. 2017;7(1, article 1657) doi: 10.1038/s41598-017-01774-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Sarandy M. M., Novaes R. D., Xavier A. A., et al. Hydroethanolic extract of strychnos pseudoquina accelerates skin wound healing by modulating the oxidative status and microstructural reorganization of scar tissue in experimental type I diabetes. BioMed Research International. 2017;2017:11. doi: 10.1155/2017/9538351.9538351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Houston D. M. J., Robins B., Bugert J. J., Denyer S. P., Heard C. M. In vitro permeation and biological activity of punicalagin and zinc (II) across skin and mucous membranes prone to herpes simplex virus infection. European Journal of Pharmaceutical Sciences. 2017;96:99–106. doi: 10.1016/j.ejps.2016.08.013. [DOI] [PubMed] [Google Scholar]