Abstract
Background
In people with acute pancreatitis, it is unclear what the role should be for medical treatment as an addition to supportive care such as fluid and electrolyte balance and organ support in people with organ failure.
Objectives
To assess the effects of different pharmacological interventions in people with acute pancreatitis.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL, 2016, Issue 9), MEDLINE, Embase, Science Citation Index Expanded, and trial registers to October 2016 to identify randomised controlled trials (RCTs). We also searched the references of included trials to identify further trials.
Selection criteria
We considered only RCTs performed in people with acute pancreatitis, irrespective of aetiology, severity, presence of infection, language, blinding, or publication status for inclusion in the review.
Data collection and analysis
Two review authors independently identified trials and extracted data. We did not perform a network meta‐analysis as planned because of the lack of information on potential effect modifiers and differences of type of participants included in the different comparisons, when information was available. We calculated the odds ratio (OR) with 95% confidence intervals (CIs) for the binary outcomes and rate ratios with 95% CIs for count outcomes using a fixed‐effect model and random‐effects model.
Main results
We included 84 RCTs with 8234 participants in this review. Six trials (N = 658) did not report any of the outcomes of interest for this review. The remaining 78 trials excluded 210 participants after randomisation. Thus, a total of 7366 participants in 78 trials contributed to one or more outcomes for this review. The treatments assessed in these 78 trials included antibiotics, antioxidants, aprotinin, atropine, calcitonin, cimetidine, EDTA (ethylenediaminetetraacetic acid), gabexate, glucagon, iniprol, lexipafant, NSAIDs (non‐steroidal anti‐inflammatory drugs), octreotide, oxyphenonium, probiotics, activated protein C, somatostatin, somatostatin plus omeprazole, somatostatin plus ulinastatin, thymosin, ulinastatin, and inactive control. Apart from the comparison of antibiotics versus control, which included a large proportion of participants with necrotising pancreatitis, the remaining comparisons had only a small proportion of patients with this condition. Most trials included either only participants with severe acute pancreatitis or included a mixture of participants with mild acute pancreatitis and severe acute pancreatitis (75 trials). Overall, the risk of bias in trials was unclear or high for all but one of the trials.
Source of funding: seven trials were not funded or funded by agencies without vested interest in results. Pharmaceutical companies partially or fully funded 21 trials. The source of funding was not available from the remaining trials.
Since we considered short‐term mortality as the most important outcome, we presented only these results in detail in the abstract. Sixty‐seven studies including 6638 participants reported short‐term mortality. There was no evidence of any differences in short‐term mortality in any of the comparisons (very low‐quality evidence). With regards to other primary outcomes, serious adverse events (number) were lower than control in participants taking lexipafant (rate ratio 0.67, 95% CI 0.46 to 0.96; N = 290; 1 study; very low‐quality evidence), octreotide (rate ratio 0.74, 95% CI 0.60 to 0.89; N = 770; 5 studies; very low‐quality evidence), somatostatin plus omeprazole (rate ratio 0.36, 95% CI 0.19 to 0.70; N = 140; 1 study; low‐quality evidence), and somatostatin plus ulinastatin (rate ratio 0.30, 95% CI 0.15 to 0.60; N = 122; 1 study; low‐quality evidence). The proportion of people with organ failure was lower in octreotide than control (OR 0.51, 95% CI 0.27 to 0.97; N = 430; 3 studies; very low‐quality evidence). The proportion of people with sepsis was lower in lexipafant than control (OR 0.26, 95% CI 0.08 to 0.83; N = 290; 1 study; very low‐quality evidence). There was no evidence of differences in any of the remaining comparisons in these outcomes or for any of the remaining primary outcomes (the proportion of participants experiencing at least one serious adverse event and the occurrence of infected pancreatic necrosis). None of the trials reported heath‐related quality of life.
Authors' conclusions
Very low‐quality evidence suggests that none of the pharmacological treatments studied decrease short‐term mortality in people with acute pancreatitis. However, the confidence intervals were wide and consistent with an increase or decrease in short‐term mortality due to the interventions. We did not find consistent clinical benefits with any intervention. Because of the limitations in the prognostic scoring systems and because damage to organs may occur in acute pancreatitis before they are clinically manifest, future trials should consider including pancreatitis of all severity but power the study to measure the differences in the subgroup of people with severe acute pancreatitis. It may be difficult to power the studies based on mortality. Future trials in participants with acute pancreatitis should consider other outcomes such as complications or health‐related quality of life as primary outcomes. Such trials should include health‐related quality of life, costs, and return to work as outcomes and should follow patients for at least three months (preferably for at least one year).
Plain language summary
Medical treatment for people with acute pancreatitis (sudden inflammation of the pancreas)
Background
The pancreas is an organ in the abdomen (tummy) that secretes several digestive enzymes (substances that enable and speed up chemical reactions in the body) into the pancreatic ductal system before it empties into the small bowel. It also contains the Islets of Langerhans, which secrete several hormones including insulin (helps regulate blood sugar). Acute pancreatitis is life‐threatening illness characterized by sudden inflammation of the pancreas, which can lead to failure of other organs, such as the lungs and kidneys. There is a lot of research into different medical treatments for the treatment of acute pancreatitis, but it is not clear what benefits each treatment has, or indeed if any medical treatment is beneficial apart from supportive treatment. This care includes body hydration and intensive care treatment for people with organ failure (to support the failing organs). We sought to resolve this issue by searching for existing studies on the topic. We included all randomised controlled trials (clinical studies where people are randomly put into one of two or more treatment groups) whose results were reported to 7 October 2016.
Study characteristics
We included 84 RCTs with 8234 participants in this review. Six trials (658 participants) did not report any of the outcomes of interest for this review. In the remaining 78 trials, 210 participants were excluded after randomisation. Thus, a total of 7366 participants in 78 trials contributed to one or more outcomes for this review. Apart from the comparison of whether antibiotics should be used, the other comparisons included only a small percentage of people with pancreatic necrosis (an extremely severe form of pancreatitis, which results in pancreatic destruction). Most trials included only the severe form of acute pancreatitis or included both mild and severe forms of pancreatitis.
Source of funding: seven trials were not funded or were funded by agencies without vested interest in results. Twenty‐one trials were partly or fully funded by pharmaceutical companies. The source of funding was not available from the remaining trials.
Quality of the evidence
The overall quality of evidence was low for all the measures because the trials were at unclear or high risk of bias (a systematic error or deviation from the truth that affects the results, favouring one treatment over another) and were small trials. As a result, further studies are required on this topic.
Key results
Sixty‐seven studies including 6638 participants reported short‐term deaths. Overall, an average 12% of people who received only supportive care died. There was no evidence that any of the treatments decreased short‐term deaths. There was evidence that various treatments might be beneficial in a number of outcomes; however, these results were not consistent, and we cannot make any conclusions as to whether any of the treatments may be beneficial. None of the trials reported health‐related quality of life.
In conclusion, based on low quality evidence, there is no evidence that any drug treatment added on to supportive care decreases short‐term deaths. Future trials in participants with acute pancreatitis should include health‐related quality of life, costs, and return to work as outcomes and should follow patients for at least three months (preferably for at least one year).
Summary of findings
Summary of findings for the main comparison. Summary of findings (mortality).
| Pharmacological interventions for treatment of acute severe pancreatitis (mortality) | |||||
| Patient or population: people with acute pancreatitis Settings: secondary or tertiary setting Intervention: various treatments Control: inactive control | |||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | |
|
Assumed risk Inactive control |
Corresponding risk Various treatments |
||||
|
Short‐term mortality Follow‐up: up to 3 months |
Antibiotics | OR 0.81 (0.57 to 1.15) | 1058 (17 studies) | ⊕⊝⊝⊝ Very lowa,b,c | |
| 120 per 1000 | 99 per 1000 (72 to 135) | ||||
| Antioxidants | OR 2.01 (0.53 to 7.56) | 163 (4 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 215 per 1000 (68 to 508) | ||||
| Aprotinin | OR 0.68 (0.40 to 1.14) | 651 (7 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 85 per 1000 (52 to 135) | ||||
| Calcitonin | OR 0.55 (0.15 to 2.00) | 125 (2 studies) | ⊕⊝⊝⊝ Very low1,²,3 | ||
| 120 per 1000 | 69 per 1000 (20 to 214) | ||||
| Cimetidine | OR 1.00 (0.06 to 17.18) | 40 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 120 per 1000 (8 to 701) | ||||
| EDTA | OR 0.94 (0.12 to 7.08) | 64 (1 study) | ⊕⊝⊝⊝ Very low1,²,3 | ||
| 120 per 1000 | 113 per 1000 (17 to 491) | ||||
| Gabexate | OR 0.79 (0.48 to 1.30) | 576 (5 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 98 per 1000 (62 to 151) | ||||
| Glucagon | OR 0.97 (0.51 to 1.87) | 409 (5 studies) | ⊕⊝⊝⊝ Very low1,²,3 | ||
| 120 per 1000 | 117 per 1000 (65 to 203) | ||||
| Iniprol | OR 0.14 (0.01 to 1.67) | 24 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 19 per 1000 (2 to 185) | ||||
| Lexipafant | OR 0.55 (0.30 to 1.01) | 423 (3 studies) | ⊕⊝⊝⊝ Very low1,²,3 | ||
| 120 per 1000 | 70 per 1000 (40 to 121) | ||||
| Octreotide | OR 0.76 (0.47 to 1.23) | 927 (6 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 94 per 1000 (60 to 143) | ||||
| Probiotics | OR 1.70 (0.87 to 3.30) | 358 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 120 per 1000 | 188 per 1000 (106 to 310) | ||||
| Activated protein C | OR 8.56 (0.41 to 180.52) | 32 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 539 per 1000 (52 to 961) | ||||
| Somatostatin | OR 0.57 (0.29 to 1.10) | 493 (6 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 72 per 1000 (39 to 130) | ||||
| Somatostatin plus omeprazole | OR 0.23 (0.05 to 1.11) | 140 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 30 per 1000 (6 to 132) | ||||
| Somatostatin plus ulinastatin | OR 0.43 (0.15 to 1.23) | 122 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 55 per 1000 (20 to 144) | ||||
| Thymosin | Not estimable | 24 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | not estimable | ||||
| Ulinastatin | OR 0.45 (0.12 to 1.72) | 132 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 120 per 1000 | 58 per 1000 (16 to 190) | ||||
| Long‐term mortality Follow‐up: 1 year | None of the trials with inactive treatment in the control group reported long‐term mortality. | ||||
| *The basis for the assumed risk is the average control group proportion across all comparisons. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence intervals; OR: odds ratio; EDTA: ethylenediaminetetraacetic acid. | |||||
| GRADE Working Group grades of evidence High quality: further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: we are very uncertain about the estimate. | |||||
aRisk of bias: downgraded by one level. bImprecision: downgraded one level for wide confidence intervals. cImprecision: downgraded one level for small sample size. dHeterogeneity: downgraded one level for lack of overlap of confidence intervals and high I².
Summary of findings 2. Summary of findings (other primary outcomes).
| Pharmacological interventions for treatment of acute severe pancreatitis (other outcomes) | |||||
| Patient or population: people with acute pancreatitis Settings: secondary or tertiary setting Intervention: various treatments Control: inactive control | |||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | |
| Assumed risk | Corresponding risk | ||||
| Inactive control | Various treatments | ||||
|
Serious adverse events (proportion) Follow‐up: up to 3 months |
Antibiotics | OR 0.65 (0.37 to 1.15) | 304 (5 studies) | ⊕⊝⊝⊝ Very lowa,b,c | |
| 147 per 1000 | 101 per 1000 (60 to 166) | ||||
| Antioxidants | OR 1.98 (0.48 to 8.13) | 82 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 147 per 1000 | 255 per 1000 (77 to 584) | ||||
| EDTA | OR 0.52 (0.11 to 2.39) | 64 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 147 per 1000 | 83 per 1000 (19 to 292) | ||||
| Gabexate | OR 1.31 (0.31 to 5.60) | 201 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 147 per 1000 | 185 per 1000 (51 to 492) | ||||
| Glucagon | OR 0.29 (0.01 to 7.46) | 127 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 147 per 1000 | 48 per 1000 (2 to 563) | ||||
| Octreotide | OR 1.73 (0.61 to 4.93) | 58 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 147 per 1000 | 230 per 1000 (95 to 460) | ||||
| Somatostatin | OR 1.07 (0.35 to 3.27) | 111 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 147 per 1000 | 156 per 1000 (57 to 361) | ||||
|
Serious adverse events (number) Follow‐up: up to 3 months |
Antibiotics | Rate ratio0.86 (0.68 to 1.07) | 716 (12 studies) | ⊕⊝⊝⊝ Very lowa,b,c | |
| 437 per 1000 | 374 per 1000 (298 to 469) | ||||
| Antioxidants | Rate ratio0.22 (0.02 to 2.21) | 71 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 94 per 1000 (9 to 967) | ||||
| Aprotinin | Rate ratio0.79 (0.49 to 1.29) | 264 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 345 per 1000 (212 to 562) | ||||
| Cimetidine | Rate ratio1.00 (0.20 to 4.95) | 60 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 437 per 1000 (88 to 2165) | ||||
| EDTA | Rate ratio0.94 (0.19 to 4.65) | 64 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 411 per 1000 (83 to 2034) | ||||
| Gabexate | Rate ratio0.86 (0.64 to 1.15) | 375 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 375 per 1000 (279 to 503) | ||||
| Glucagon | Rate ratio1.00 (0.02 to 50.40) | 68 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 437 per 1000 (9 to 22027) | ||||
| Lexipafant | rate ratio0.67 (0.46 to 0.96) | 290 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 292 per 1000 (203 to 420) | ||||
| Octreotide | Rate ratio0.74 (0.60 to 0.89) | 770 (5 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 321 per 1000 (264 to 391) | ||||
| Probiotics | Rate ratio0.94 (0.65 to 1.36) | 397 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 437 per 1000 | 412 per 1000 (286 to 595) | ||||
| Somatostatin | Rate ratio1.03 (0.66 to 1.59) | 257 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 437 per 1000 | 449 per 1000 (290 to 695) | ||||
| Somatostatin plus omeprazole | Rate ratio0.36 (0.19 to 0.70) | 140 (1 study) | ⊕⊕⊝⊝ Lowa,b | ||
| 437 per 1000 | 159 per 1000 (82 to 308) | ||||
| Somatostatin plus ulinastatin | Rate ratio0.30 (0.15 to 0.60) | 122 (1 study) | ⊕⊕⊝⊝ Lowa,b | ||
| 437 per 1000 | 133 per 1000 (68 to 262) | ||||
|
Organ failure Follow‐up: up to 3 months |
Antibiotics | OR 0.78 (0.44 to 1.38) | 258 (5 studies) | ⊕⊝⊝⊝ Very lowa,b,c | |
| 289 per 1000 | 241 per 1000 (152 to 360) | ||||
| Antioxidants | OR 0.92 (0.39 to 2.12) | 163 (4 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 289 per 1000 | 271 per 1000 (138 to 463) | ||||
| Gabexate | OR 0.32 (0.01 to 8.25) | 50 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 289 per 1000 | 115 per 1000 (5 to 770) | ||||
| Lexipafant | OR 0.68 (0.36 to 1.27) | 340 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 289 per 1000 | 216 per 1000 (128 to 341) | ||||
| Octreotide | OR 0.51 (0.27 to 0.97) | 430 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 289 per 1000 | 173 per 1000 (99 to 284) | ||||
| Probiotics | OR 0.80 (0.26 to 2.47) | 358 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 289 per 1000 | 246 per 1000 (95 to 501) | ||||
| Ulinastatin | OR 0.27 (0.01 to 6.67) | 129 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 289 per 1000 | 100 per 1000 (5 to 731) | ||||
|
Infected pancreatic necrosis Follow‐up: up to 3 months |
Antibiotics | OR 0.82 (0.53 to 1.25) | 714 (11 studies) | ⊕⊝⊝⊝ Very lowa,b,c | |
| 140 per 1000 | 118 per 1000 (80 to 169) | ||||
| Octreotide | OR 0.52 (0.04 to 6.06) | 58 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 140 per 1000 | 78 per 1000 (7 to 497) | ||||
| Probiotics | OR 1.10 (0.62 to 1.96) | 397 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 140 per 1000 | 152 per 1000 (92 to 243) | ||||
|
Sepsis Follow‐up: up to 3 months |
Antibiotics | OR 0.42 (0.11 to 1.60) | 60 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | |
| 122 per 1000 | 56 per 1000 (15 to 182) | ||||
| Aprotinin | OR 1.84 (0.49 to 6.96) | 103 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 122 per 1000 | 204 per 1000 (63 to 492) | ||||
| Gabexate | OR 1.10 (0.55 to 2.19) | 373 (3 studies) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 122 per 1000 | 133 per 1000 (71 to 233) | ||||
| Lexipafant | OR 0.26 (0.08 to 0.83) | 290 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 122 per 1000 | 35 per 1000 (12 to 103) | ||||
| Octreotide | OR 0.40 (0.05 to 3.53) | 340 (2 studies) | ⊕⊝⊝⊝ Very lowa,b,c,d | ||
| 122 per 1000 | 53 per 1000 (6 to 329) | ||||
| Probiotics | OR 0.36 (0.10 to 1.36) | 62 (1 study) | ⊕⊝⊝⊝ Very lowa,b,c | ||
| 122 per 1000 | 48 per 1000 (13 to 159) | ||||
| Health‐related quality of life | None of the trials reported this outcome. | ||||
| *The basis for the assumed risk is the average control group proportion across all comparisons. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence intervals; OR = odds ratio; EDTA = ethylenediaminetetraacetic acid. | |||||
| GRADE Working Group grades of evidence High quality: further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: we are very uncertain about the estimate. | |||||
aRisk of bias: downgraded by one level. bImprecision: downgraded one level for wide confidence intervals. cImprecision: downgraded one level for small sample size. dHeterogeneity: downgraded one level for lack of overlap of confidence intervals and high I².
Background
Description of the condition
The pancreas is an abdominal organ that secretes several digestive enzymes into the pancreatic ductal system before it empties into the small bowel. The pancreas also lodges the Islets of Langerhans, which secrete several hormones including insulin (NCBI 2014). Acute pancreatitis is a sudden inflammatory process in the pancreas, with variable involvement of nearby organs or other organ systems (Bradley 1993). The annual incidence of acute pancreatitis ranges from 5 to 30 per 100,000 population (Roberts 2013; Yadav 2006). There has been an increase in the incidence of acute pancreatitis in the last 10 to 20 years in the UK and USA (Roberts 2013; Yang 2008). Acute pancreatitis is the commonest gastrointestinal (digestive tract) cause of hospital admission in the USA (Peery 2012), and gallstones and alcohol are the two main causes. Approximately, 50% to 70% of acute pancreatitis is caused by gallstones (Roberts 2013; Yadav 2006); these slip into the common bile duct and obstruct the ampulla of Vater (a common channel formed by the union of common bile duct and pancreatic duct), resulting in obstruction to the flow of pancreatic enzymes and leading to activation of trypsinogen within the pancreas and acute pancreatitis (Sah 2013).
Advanced age, male sex, and lower socioeconomic class are associated with higher incidence of acute pancreatitis (Roberts 2013).
Clinicians generally diagnose acute pancreatitis when at least two of the following three features are present (Banks 2013).
Acute onset of a persistent, severe, epigastric pain, often radiating to the back.
Serum lipase activity (or amylase activity) at least three times greater than the upper limit of normal.
Characteristic findings of acute pancreatitis on contrast‐enhanced computed tomography (CECT) and less commonly magnetic resonance imaging (MRI) or transabdominal ultrasonography.
Depending upon the type of inflammation, acute pancreatitis can be classified into interstitial oedematous pancreatitis (diffuse (widespread) or occasionally localised enlargement of the pancreas due to inflammatory oedema as seen on CECT) or necrotising pancreatitis (necrosis involving either the pancreas, peripancreatic tissues, or both) (Banks 2013). Approximately 90% to 95% of people with acute pancreatitis have interstitial oedematous pancreatitis, while the remainder have necrotising pancreatitis (Banks 2013). Necrotising pancreatitis may be sterile or infected (Banks 2013). Various theories exist as to how pancreatic and peripancreatic tissues get infected. These include spread from blood circulation, lymphatics, bile, and the small bowel (duodenum) through the pancreatic duct, as well as movement (translocation) through the large bowel wall (Schmid 1999).
Local complications of acute pancreatitis include acute peripancreatic fluid collection, pancreatic pseudocyst, acute necrotic collection, and walled‐off necrosis (Banks 2013). The systemic complications of acute pancreatitis include worsening of pre‐existing illnesses such as heart or chronic lung disease (Banks 2013). The mortality rates following an attack of acute pancreatitis are between 6% and 20% (Roberts 2013; Yadav 2006), according to severity. Acute pancreatitis can be classified as mild, moderate, or severe, depending on the presence of local or systemic complications, transient organ failure involving one of more of lungs, kidneys, and cardiovascular system (heart and blood vessels) lasting up to 48 hours, or persistent failure of these organs lasting beyond 48 hours. Mild pancreatitis has the best prognosis, and there are no local or systemic complications or organ failure. In moderately severe acute pancreatitis, there may be local or systemic complications or transient organ failure. Severe acute pancreatitis carries the worst prognosis in terms of mortality, and there is persistent organ failure (Banks 2013).
The clinical manifestation of acute pancreatitis is believed to be caused by activation of inflammatory pathways either directly by the pathologic insult or indirectly by activation of trypsinogen (an enzyme that digests protein or a protease), resulting in formation of trypsin, a protease that can break down the pancreas (Sah 2013). This activation of inflammatory pathways manifests clinically as systemic inflammatory response syndrome (SIRS) (Banks 2013; Sah 2013; Tenner 2013). Systemic inflammatory response syndrome is characterised by two or more of the following criteria (Bone 1992).
Temperature of less than 36°C or more than 38°C.
Heart rate less than 90 beats/minute.
Respiratory rate more than 20/min or PCO₂ less than 32 mm Hg.
White blood cell count more than 12,000/mm³, less than 4000/mm³, or more than 10% immature (band) forms.
See Appendix 1 for a glossary of terms.
Description of the intervention
The main purpose of treatment is to decrease the mortality and morbidity associated with acute pancreatitis. The various pharmacological interventions that have been evaluated in the treatment of acute pancreatitis include agents such as somatostatin or octreotide that decrease pancreatic secretions; protease inhibitors such as gabexate mesilate, aprotinin, ulinastatin, and nafamostat; antioxidants such as vitamin C and selenium; platelet activating factor such as lexipafant; other agents that modulate the inflammatory pathway such as steroids and tumour necrosis factor‐alpha (TNF‐α) antibody; probiotics; and antibiotics (Bang 2008; Neumann 2011; Rada 2011; Yang 2011). We included any pharmacological intervention aimed at the treatment of acute pancreatitis.
We did not cover endoscopic sphincterotomy for the treatment of common bile duct stones (Ayub 2010), nor did we focus on endoscopic, radiology‐guided percutaneous treatments or surgical treatments for treatment of complications of acute pancreatitis (Tenner 2013). Furthermore, we did not cover the use of non‐steroidal anti‐inflammatory drugs (NSAIDs) or other drugs such as somatostatin analogues for preventing postendoscopic retrograde cholangiopancreatography (post‐ECRP)‐induced pancreatitis (Elmunzer 2012; Zhang 2009).
How the intervention might work
Somatostatin and its analogues decrease pancreatic secretion (Bang 2008). Since autodigestion (breakdown of pancreas) due to trypsinogen activation is one of the mechanisms believed to cause acute pancreatitis, decreasing pancreatic secretion can decrease the amount of trypsinogen. Inhibition of trypsin by protease inhibitors may result in decreased damage to the pancreas (Neumann 2011). Antioxidants, platelet‐activating factor inhibitors, steroids, and TNF‐α antibody are all aimed at decreasing the inflammatory response or at mitigating the damage resulting from the inflammatory response (Bang 2008). Probiotics decrease the bacterial colonisation of the gut, and antibiotics have antibacterial actions (Bang 2008).
Why it is important to do this review
Despite various pharmacological interventions being evaluated in acute pancreatitis, none is currently recommended in the treatment of acute pancreatitis, with the exception of antibiotics in infected necrotising pancreatitis (Tenner 2013). Systematic reviews and meta‐analyses increase the precision of the treatment effects (i.e. they provide a narrower range of the average treatment effect) (Higgins 2011), and so decrease the risk of a type II error (concluding that there is no difference between treatments when there is actually a difference). Systematic reviews also help in identifying the differences in the treatment effects between studies and allow exploration of the reasons behind these differences. Many studies have compared these interventions with placebo or with no treatment. It is therefore not possible to obtain accurate information on how one treatment compares with another treatment. Multiple treatment comparisons or a network meta‐analysis allow comparison of several treatments simultaneously and provide information on the relative effect of one treatment versus another, even when there is no direct comparison. There is no Cochrane Review or network meta‐analysis on this topic. So, we planned to perform a network meta‐analysis if the type of participants were included across all the comparisons. This systematic review will identify the relative effects of different treatments and identify any research gaps.
Objectives
To assess the effects of different pharmacological interventions in people with acute pancreatitis.
Methods
Criteria for considering studies for this review
Types of studies
We included only randomised controlled trials (RCTs). We included studies reported as full text, those published as abstract only, and unpublished data.
Types of participants
We included adults with acute pancreatitis irrespective of the severity (mild, moderately severe, or severe acute pancreatitis) or the type of acute pancreatitis (acute interstitial oedematous pancreatitis or necrotising pancreatitis).
Types of interventions
We included trials comparing any pharmacological interventions mentioned above with another, with placebo, or with no intervention, provided that the only difference between the randomised groups was the pharmacological intervention or interventions being assessed. Some of the interventions that we included are listed below.
Activated protein C.
Antibiotics.
Antioxidants.
Aprotinin.
Calcitonin.
Cimetidine.
EDTA (ethylenediaminetetraacetic acid).
Gabexate.
Glucagon.
Iniprol.
Lexipafant.
Octreotide.
Omeprazole.
Probiotics.
Somatostatin.
Thymosin.
Ulinastatin.
We did not combine the different somatostatin analogues (such as somatostatin or octreotide) as a single treatment in order to avoid further clinical heterogeneity. We assessed a combination of drugs as a separate treatment.
Types of outcome measures
Primary outcomes
-
Mortality.
Short‐term mortality (in‐hospital mortality or mortality within six months).
Long‐term mortality (at maximum follow‐up).
-
Serious adverse events (within six months). We accepted the definition of serious adverse events from the International Conference on Harmonisation ‐ Good Clinical Practice guideline (ICH‐GCP 1997): any untoward medical occurrence that results in death, is life‐threatening, requires inpatient hospitalisation or prolongation of existing hospitalisation, or results in persistent or significant disability/incapacity. Wealso accepted other variations of ICH‐GCP classifications such as Food and Drug Administration (FDA) classification (FDA 2006), Medicines and Healthcare products Regulatory Agency (MHRA) classification (MHRA 2013).
Proportion of people who developed serious adverse events (i.e. the percentage of people who developed one or more serious adverse events) and the number of serious adverse events (i.e. the total number of serious adverse events in each group regardless of the number of people in whom the serious adverse events developed).
Organ failure (however reported by authors).
Infected necrotising pancreatitis (cytology or positive culture).
Sepsis (however reported by authors).
-
Health‐related quality of life (using any validated scale).
Short‐term (four weeks to three months).
Medium‐term (three months to one year).
Long‐term (more than one year).
-
Health‐related quality of life (using any validated scale).
Short‐term (four weeks to three months).
Medium‐term (three months to one year).
Long‐term (more than one year).
Secondary outcomes
Adverse events (within six months). We accepted all adverse events reported by the trial authors, irrespective of the severity of the adverse event.
-
Measures of decreased complications and earlier recovery (within six months).
Length of hospital stay (including the index admission for acute pancreatitis and any disease‐related or intervention‐related readmissions including those for recurrent episodes).
Length of intensive care unit (ICU) stay (including the index admission for acute pancreatitis and any disease‐ or intervention‐related readmissions).
Requirement for additional invasive intervention such as necrosectomy for pancreatic necrosis, endoscopic or radiological drainage of collections.
Time to return to normal activity (return to pre‐acute pancreatitis episode mobility without any additional caregiver support).
Time to return to work (in those who were employed previously).
Costs (within six months).
We chose the above clinical outcomes based on the necessity to assess whether the pharmacological interventions were effective in decreasing complications, thereby decreasing the length of ICU and hospital stay, decreasing any additional interventions, and resulting in earlier return to normal activity and work as well as improvement in quality of life. The costs provide an indication of resource requirement.
We did not regard the reporting of the outcomes listed here as an inclusion criterion for the review.
Search methods for identification of studies
Electronic searches
We conducted a literature search to identify all published and unpublished randomised controlled trials. The literature search identified potential studies in all languages. We translated the non‐English language papers and fully assessed them for potential inclusion in the review as necessary.
We searched the following electronic databases for identifying potential studies.
Cochrane Central Register of Controlled Trials (CENTRAL; Issue 9, 2016; searched 7 October 2016; Appendix 2).
MEDLINE (1966 to 7 October 2016; Appendix 3).
Embase (1988 to 7 October 2016; Appendix 4).
Science Citation Index (1982 to 7 October 2016; Appendix 5).
We also conducted a search of ClinicalTrials.gov (Appendix 6) and World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) (Appendix 8) on 7 October 2016.
Searching other resources
We checked the reference lists of all primary studies and review articles for additional references. We contacted authors of identified trials and asked them to identify any other published and unpublished studies.
We searched for errata or retractions from eligible trials on www.ncbi.nlm.nih.gov/pubmed on 7 October 2016.
Data collection and analysis
Selection of studies
Two review authors (KG and AB) independently screened titles and abstracts of all the potential studies that we identified through the searches and coded them as 'retrieve' (eligible or potentially eligible/unclear) or 'do not retrieve'. We retrieved the full‐text study reports, and two review authors (KG and RK or EM) independently screened them and identified studies for inclusion; we identified and recorded reasons for exclusion of the ineligible studies. We resolved any disagreement through discussion. We identified and excluded duplicates and collated multiple reports of the same study so that each study rather than each report was the unit of interest in the review. We planned to contact the investigators of trials of unclear eligibility. We recorded the selection process in sufficient detail to complete a PRISMA flow diagram (Figure 1) and a 'Characteristics of excluded studies' table.
1.
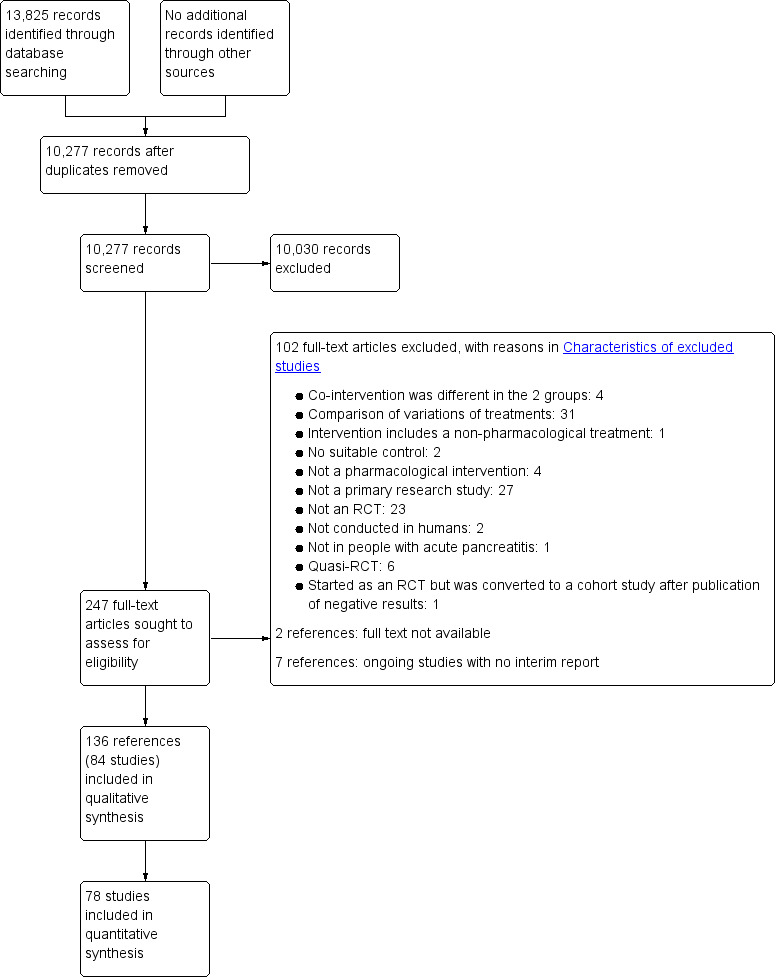
Study flow diagram.
Data extraction and management
We used a standard data collection form for study characteristics and outcome data, which had been piloted on three studies in the review. Two review authors (KG and RK or EM) independently extracted the following study characteristics.
Methods: study design, total duration study and run‐in, number of study centres and location, study setting, withdrawals, date of study.
Participants: number (N), mean age, age range, sex, severity and type of acute pancreatitis, inclusion criteria, exclusion criteria.
Interventions: intervention, comparison, co‐interventions, number of participants randomised to each group.
Outcomes: primary and secondary outcomes specified and collected, time points reported. For binary outcomes, we obtained the number of participants with events and the number of participants included in the analysis in each group. For continuous outcomes, we obtained the unit or scale of measurement, mean, standard deviation, and the number of participants included in the analysis for each group. For count outcomes, we obtained the number of events and number of participants included in the analysis in each group. For time‐to‐event outcomes, we obtained the proportion of people with events, the average duration of follow‐up of participants in the trial, and the number of participants included in the analysis for each group.
Notes: funding for trial, notable conflicts of interest of trial authors.
Two review authors (KG and RK or EM) independently extracted outcome data from included studies. If outcomes were reported at multiple time points, we planned to extract the data for all time points. We obtained information on the number of participants with adverse events (or serious adverse events) and the number of such events where applicable. We planned to extract all information on costs using the currency reported by the trial authors and planned to convert this to USD at the conversion rates on the day of the analysis. We extracted data for every trial arm that was an included intervention. If studies reported outcome data in an unusable way, we attempted to contact the trial authors and tried to obtain usable data. If we were unable to obtain usable data despite this, we planned to summarise the unusable data in an appendix. We resolved disagreements by consensus. One review author (EM) copied across the data for 'Characteristics of included studies' and 'Characteristics of excluded studies' from the data collection form into the Review Manager 5 (RevMan 5) file (RevMan 2014). One review author (KG) copied across the data for 'Data and analyses' from the data collection form into the RevMan 5 file. We double‐checked that the data were entered correctly by comparing the study reports with how the data were presented in the systematic review.
Assessment of risk of bias in included studies
Two review authors (KG and RK or EM) independently assessed the risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We resolved any disagreements by discussion. We assessed the risk of bias according to the following domains.
Random sequence generation.
Allocation concealment.
Blinding of participants and personnel.
Blinding of outcome assessment.
Incomplete outcome data.
Selective outcome reporting.
Bias due to funding source.
Other potential bias.
We graded each potential source of bias as high, low, or unclear and provided a quote from the study report together with a justification for our judgement in the 'Risk of bias' tables. We summarised the risk of bias judgements across different studies for each of the domains listed. We considered blinding separately for different key outcomes where necessary, for example, for unblinded outcome assessment, risk of bias for all‐cause mortality may be very different than for a participant‐reported pain scale. Where information on risk of bias relates to unpublished data or to correspondence with a trial author, we planned to note this in the 'Risk of bias' table. We presented the risk of bias in each pair‐wise comparison in Table 3.
1. Characteristics of included studies (ordered by comparisons).
| Study name | No of participants randomised | Postrandomisation dropouts | No of participants for whom outcome was reported | Treatment 1 | Treatment 2 | Selection bias | Performance and detection bias | Attrition bias | Selective reporting bias | Other bias |
| Pettila 2010 | 32 | 0 | 32 | Activated protein C | Placebo | Unclear | Low | Low | High | High |
| Barreda 2009 | 80 | 22 | 58 | Antibiotics | No active intervention | Unclear | Unclear | High | Low | Unclear |
| Delcenserie 1996 | 23 | 0 | 23 | Antibiotics | No active intervention | Unclear | Unclear | Low | Low | Unclear |
| Delcenserie 2001 | 81 | Not stated | 81 | Antibiotics | No active intervention | Unclear | Unclear | Unclear | Low | Unclear |
| Dellinger 2007 | 100 | 0 | 100 | Antibiotics | Placebo | Low | Low | Low | Low | High |
| Finch 1976 | 62 | 4 | 58 | Antibiotics | No active intervention | Unclear | Unclear | High | Low | Unclear |
| Garcia‐Barrasa 2009 | 46 | 5 | 41 | Antibiotics | Placebo | Unclear | Low | High | Low | Low |
| Hejtmankova 2003 | 41 | Not stated | 41 | Antibiotics | No active intervention | Unclear | Unclear | Unclear | Low | Unclear |
| Isenmann 2004 | 119 | 5 | 114 | Antibiotics | Placebo | Unclear | Low | High | High | High |
| Llukacaj 2012 | 80 | Not stated | 80 | Antibiotics | Placebo | Unclear | Low | Unclear | High | Unclear |
| Luiten 1995 | 109 | 7 | 102 | Antibiotics | No active intervention | Unclear | Unclear | High | Low | Unclear |
| Nordback 2001 | 90 | 32 | 58 | Antibiotics | Placebo | Unclear | Unclear | High | Low | Unclear |
| Poropat 2015 | 47 | 0 | 47 | Antibiotics | No active intervention | Unclear | Unclear | Low | Low | Unclear |
| Pederzoli 1993a | 74 | Not stated | 74 | Antibiotics | No active intervention | Unclear | Unclear | Low | Low | Unclear |
| Rokke 2007 | 73 | 0 | 73 | Antibiotics | No active intervention | Unclear | High | Low | Low | High |
| Sainio 1995 | 60 | 0 | 60 | Antibiotics | No active intervention | Unclear | Unclear | Low | Low | Unclear |
| Spicak 2002 | 63 | Not stated | 63 | Antibiotics | No active intervention | Unclear | Unclear | Unclear | Low | Unclear |
| Spicak 2003 | 41 | Not stated | 41 | Antibiotics | No active intervention | Unclear | Unclear | Unclear | Low | Unclear |
| Xue 2009 | 59 | 3 | 56 | Antibiotics | No active intervention | Unclear | Unclear | High | Low | Low |
| Bansal 2011 | 44 | 5 | 39 | Antioxidants | No active intervention | Unclear | High | High | Low | Low |
| Birk 1994 | 20 | Not stated | 20 | Antioxidants | No active intervention | Unclear | Unclear | Unclear | High | Unclear |
| Marek 1999 | 73 | 0 | 73 | Antioxidants | Placebo | Unclear | Unclear | Low | High | Unclear |
| Sateesh 2009 | 56 | 3 | 53 | Antioxidants | No active intervention | Unclear | High | High | Low | Unclear |
| Siriwardena 2007 | 43 | 0 | 43 | Antioxidants | Placebo | Low | Low | Low | Low | High |
| Vege 2015 | 28 | Not stated | 28 | Antioxidants | Placebo | Unclear | Low | Low | Low | Unclear |
| Chooklin 2007 | 34 | Not stated | 34 | Antioxidants plus Corticosteroids | No active intervention | Unclear | Unclear | Unclear | High | Unclear |
|
MRC Multicentre Trial 1977 (this is a 3‐armed trial; the numbers stated included all 3 arms) |
264 | 7 | 257 | Aprotinin | Placebo | Unclear | Low | High | High | High |
| Balldin 1983 | 55 | Not stated | 55 | Aprotinin | No active intervention | Unclear | Unclear | Unclear | Low | High |
| Berling 1994 | 48 | Not stated | 48 | Aprotinin | No active intervention | Unclear | Low | Low | Low | High |
| Imrie 1978 | 161 | Not stated | 161 | Aprotinin | Placebo | Unclear | Low | Unclear | Low | High |
| Imrie 1980 | 50 | Not stated | 50 | Aprotinin | Placebo | Unclear | Low | Unclear | High | Unclear |
| Storck 1968 | 43 | Not stated | 43 | Aprotinin | Placebo | Unclear | Low | Unclear | High | Unclear |
| Trapnell 1974 | 105 | Not stated | 105 | Aprotinin | Placebo | Low | Low | Unclear | High | High |
|
MRC Multicentre Trial 1977 (this is a 3‐armed trial; the numbers stated included all 3 arms) |
264 | 7 | 257 | Aprotinin | Glucagon | Unclear | Low | High | High | High |
| Goebell 1979 | 94 | Not stated | 94 | Calcitonin | Placebo | Unclear | Low | Unclear | Low | Unclear |
| Martinez 1984 | 31 | 0 | 31 | Calcitonin | Placebo | Unclear | Unclear | Low | High | Unclear |
| Perezdeoteyza 1980 | 40 | Not stated | 40 | Cimetidine | Placebo | Unclear | Low | Unclear | High | Unclear |
| Sillero 1981 | 60 | Not stated | 60 | Cimetidine | Placebo | Low | Unclear | Unclear | High | Unclear |
| Tykka 1985 | 64 | 0 | 64 | EDTA | Placebo | Unclear | Low | Low | Low | High |
| Frulloni 1994 | 116 | Not stated | 116 | Gabexate | Aprotinin | Unclear | Unclear | Unclear | Low | Unclear |
| Pederzoli 1993b | 199 | 17 | 182 | Gabexate | Aprotinin | Unclear | Low | High | Low | Unclear |
| Buchler 1993 | 223 | Not stated | 223 | Gabexate | Placebo | Low | Low | Low | Low | Unclear |
| Chen 2000 | 52 | Not stated | 52 | Gabexate | Placebo | Unclear | Unclear | Unclear | Low | Unclear |
| Freise 1986 | 50 | Not stated | 50 | Gabexate | Placebo | Unclear | Low | Unclear | Low | Unclear |
| Goebell 1988 | 162 | 11 | 151 | Gabexate | Placebo | Unclear | Low | High | Low | Unclear |
| Valderrama 1992 | 105 | 5 | 100 | Gabexate | Placebo | Low | Low | High | Low | High |
| Kirsch 1978 | 150 | Not stated | 150 | Glucagon | Atropine | Unclear | Unclear | Unclear | Low | Unclear |
|
MRC Multicentre Trial 1977 (this is a 3‐armed trial; the numbers stated included all 3 arms) |
264 | 7 | 257 | Glucagon | Placebo | Unclear | Unclear | Unclear | Low | High |
| Debas 1980 | 66 | Not stated | 66 | Glucagon | Placebo | Unclear | Low | Unclear | Low | Unclear |
| Dürr 1978 | 69 | Not stated | 69 | Glucagon | Placebo | Unclear | Low | Unclear | High | Unclear |
| Kalima 1980 | 80 | 9 | 71 | Glucagon | Placebo | Unclear | Unclear | High | Low | Unclear |
| Kronborg 1980 | 22 | Not stated | 22 | Glucagon | Placebo | Unclear | Low | Unclear | High | Unclear |
| Gilsanz 1978 | 62 | Not stated | 62 | Glucagon | Oxyphenonium | Unclear | Low | Unclear | Low | Unclear |
| Hansky 1969 | 24 | Not stated | 24 | Iniprol | No active intervention | Unclear | High | Unclear | High | High |
| Johnson 2001 | 291 | 1 | 290 | Lexipafant | Placebo | Unclear | Low | High | Low | High |
| Kingsnorth 1995 | 83 | Not stated | 83 | Lexipafant | Placebo | Unclear | Low | Unclear | High | High |
| McKay 1997b | 51 | 1 | 50 | Lexipafant | Placebo | Unclear | Low | High | High | High |
| Bredkjaer 1988 | 66 | 9 | 57 | NSAID | Placebo | Unclear | Unclear | Unclear | High | Unclear |
| Ebbehøj 1985 | 30 | 0 | 30 | NSAID | Placebo | Unclear | Low | Low | High | High |
| McKay 1997a | 58 | 0 | 58 | Octreotide | Placebo | Low | Low | Low | Low | Unclear |
| Ohair 1993 | 180 | Not stated | 180 | Octreotide | Placebo | Unclear | Unclear | Unclear | High | Unclear |
| Paran 1995 | 51 | 13 | 38 | Octreotide | No active intervention | Unclear | High | High | Low | Unclear |
| Uhl 1999 | 302 | 0 | 302 | Octreotide | Placebo | Unclear | Low | Low | Low | High |
| Wang 2013c | 372 | Not stated | 372 | Octreotide | No active intervention | Unclear | Unclear | High | Low | Low |
| Yang 2012 | 163 | 6 | 157 | Octreotide | No active intervention | Unclear | Unclear | High | High | Low |
| Wang 2013b | 354 | Not stated | 354 | Octreotide plus NSAID | Octreotide | Unclear | Unclear | Unclear | High | Unclear |
| Guo 2015 | 120 | Not stated | 120 | Octreotide plus ulinastatin | Octreotide | Unclear | Unclear | Unclear | Low | Unclear |
| Besselink 2008 | 298 | 2 | 296 | Probiotics | Placebo | Low | Low | High | Low | High |
| Olah 2007 | 83 | 21 | 62 | Probiotics | No active intervention | Unclear | Low | High | High | Unclear |
| Plaudis 2010 | 90 | Not stated | 58 | Probiotics | No active intervention | Unclear | Low | Unclear | High | Unclear |
| Sharma 2011 | 50 | 0 | 50 | Probiotics | Placebo | Unclear | Low | Low | High | High |
| Zhu 2014 | 39 | Not stated | 39 | Probiotics | Placebo | Unclear | Low | Unclear | High | Unclear |
| Grupo Español 1996 | 70 | 9 | 61 | Somatostatin | Placebo | Unclear | Low | High | High | Unclear |
| Choi 1989 | 71 | Not stated | 71 | Somatostatin | No active intervention | Unclear | Unclear | Unclear | Low | Unclear |
| Gjørup 1992 | 63 | Not stated | 63 | Somatostatin | Placebo | Unclear | Low | Unclear | Low | Unclear |
| Luengo 1994 | 100 | Not stated | 100 | Somatostatin | No active intervention | Unclear | Low | Unclear | High | Unclear |
| Moreau 1986 | 87 | 3 | 84 | Somatostatin | Placebo | Unclear | Low | Unclear | High | High |
| Usadel 1985 | 77 | Not stated | 77 | Somatostatin | Placebo | Unclear | Low | Unclear | High | Unclear |
| Wang 2013a (this is a 3‐armed trial; the numbers stated included all 3 arms) | 183 | Not stated | 183 | Somatostatin | No active intervention | Unclear | Low | Unclear | Low | Low |
| Yang 1999 | 48 | Not stated | 48 | Somatostatin | No active intervention | Unclear | Unclear | Unclear | High | Unclear |
| Xia 2014 | 140 | Not stated | 140 | Somatostatin plus omeprazole | No active intervention | Unclear | Unclear | Unclear | Low | Unclear |
| Wang 2013a (this is a 3‐armed trial; the numbers stated included all 3 arms) | 183 | Not stated | 183 | Somatostatin plus ulinastatin | Placebo | Unclear | Unclear | Unclear | High | Unclear |
| Wang 2013a (this is a 3‐armed trial; the numbers stated included all 3 arms) | 183 | Not stated | 183 | Somatostatin plus ulinastatin | Somatostatin | Unclear | Low | Unclear | Low | Low |
| Wang 2016 (this is a 4‐armed trial; the numbers stated included all 4 arms) | 492 | 0 | 492 | Somatostatin plus ulinastatin | Somatostatin | Low | Low | Low | Low | Low |
| Wang 2016 (this is a 4‐armed trial; the numbers stated included all 4 arms) | 492 | 0 | 492 | Somatostatin plus gabexate | Somatostatin | Low | Low | Low | Low | Low |
| Wang 2016 (this is a 4‐armed trial; the numbers stated included all 4 arms) | 492 | 0 | 492 | Somatostatin plus ulinastatin plus gabexate | Somatostatin | Low | Low | Low | Low | Low |
| Wang 2016 (this is a 4‐armed trial; the numbers stated included all 4 arms) | 492 | 0 | 492 | Somatostatin plus ulinastatin | Somatostatin plus gabexate | Low | Low | Low | Low | Low |
| Wang 2016 (this is a 4‐armed trial; the numbers stated included all 4 arms) | 492 | 0 | 492 | Somatostatin plus ulinastatin plus gabexate | Somatostatin plus gabexate | Low | Low | Low | Low | Low |
| Wang 2016 (this is a 4‐armed trial; the numbers stated included all 4 arms) | 492 | 0 | 492 | Somatostatin plus ulinastatin plus gabexate | Somatostatin plus ulinastatin | Low | Low | Low | Low | Low |
| Wang 2011 | 24 | Not stated | 24 | Thymosin | Placebo | Unclear | Low | Unclear | High | Unclear |
| Abraham 2013 | 135 | 6 | 129 | Ulinastatin | Placebo | Unclear | Low | High | Low | Unclear |
| Chen 2002a | 68 | 6 | 62 | Ulinastatin | Gabexate | Unclear | Unclear | High | High | Unclear |
| Chen 2002b | 26 | 1 | 25 | Ulinastatin | Octreotide | Unclear | Unclear | High | High | Unclear |
When considering treatment effects, we took into account the risk of bias for the studies that contribute to that outcome by a sensitivity analysis.
Assessment of bias in conducting the systematic review
We conducted the review according to the published protocol and reported any deviations from it in the 'Differences between protocol and review' section of this review.
Measures of treatment effect
For dichotomous variables (short‐term mortality, proportion of participants with adverse events, requirement for additional interventions), we calculated the odds ratio (OR) with 95% confidence interval (CI). For continuous variables, such as length of hospital stay, ICU stay, time to return to normal activity, time to return to work, and costs, we planned to calculate the mean difference (MD) with 95% CI. We planned to use standardised mean difference (SMD) with 95% CI for quality of life if different scales were used. For count outcomes such as the number of adverse events, we calculated the rate ratio with 95% CIs. For time‐to‐event data, such as long‐term mortality, we planned to use the hazard ratio (HR) with a 95% CI. However, only one trial reported mortality beyond 3 months and presented the number of deaths at two years. We analysed this information as binary data.
A common way that trial authors indicate when they have skewed data is by reporting medians and interquartile ranges. When we encountered this, we reported the difference in means or medians in a table.
Unit of analysis issues
The unit of analysis was individual participants with acute pancreatitis. As anticipated, we did not find any cluster‐randomised trials for this comparison.
In multi‐arm trials, the models account for the correlation between trial‐specific treatment effects from the same trial.
Dealing with missing data
We attempted to contact investigators or study sponsors in order to verify key study characteristics and obtain missing numerical outcome data where possible (e.g. when a study was identified as abstract only). For binary, count, and time‐to‐event outcomes, we performed an intention‐to‐treat analysis whenever possible (Newell 1992). Since this was not possible, we performed an available‐case analysis but planned to assess the impact of 'best‐best', 'best‐worst', 'worst‐best', and 'worst‐worst' scenario analyses on the results for binary outcomes. For continuous outcomes, we planned to perform an available‐case analysis. If we were unable to obtain the information from the investigators or study sponsors, we planned to impute the mean from the median (i.e. consider the median as the mean) and the standard deviation from the standard error, interquartile range, or P values according to the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011), but we planned to assess the impact of including such studies as indicated in a sensitivity analysis. If we were unable to calculate the standard deviation from the standard error, interquartile range, or P values, we planned to impute the standard deviation as the highest standard deviation in the remaining trials included in the outcome, being fully aware that this method of imputation would decrease the weight of the studies in the meta‐analysis of mean difference and shift the effect estimate towards no effect for standardised mean difference. We planned to assess the impact of including such studies by sensitivity analysis.
Assessment of heterogeneity
We assessed the heterogeneity in each pair‐wise comparison by assessing the Higgins I² (Higgins 2003), the Chi² test with significance set at a P value less than 0.10, and by visual inspection.
Assessment of reporting biases
We attempted to contact trial authors, asking them to provide missing outcome data. Where this was not possible, and if we thought that the missing data may introduce serious bias, we planned to explore the impact of including such studies in the overall assessment of results by a sensitivity analysis.
If we were able to pool more than 10 trials for a specific comparison, we created and examined a funnel plot to explore possible publication biases. We used Egger's test to determine the statistical significance of the reporting bias (Egger 1997). We considered a P value of less than 0.05 to indicate statistically significant reporting bias.
Data synthesis
We undertook meta‐analyses only where this was meaningful (i.e. if the treatments, participants and the underlying clinical question were similar enough for pooling to make sense). In general, we favoured performing a meta‐analysis and clearly highlighted the reason for not performing one if we decided against it. We used both the fixed‐effect and random‐effects model, reporting the fixed‐effect model when the choice of models did not alter the conclusion and the random‐effects model when it did. We did not perform a network meta‐analysis as planned because of the lack of information on potential effect modifiers and differences of type of participants included in the different comparisons, when information was available (i.e. the transitivity assumption was not satisfied).
Subgroup analysis and investigation of heterogeneity
We planned to perform the following subgroup analyses regardless of heterogeneity.
Different types of acute pancreatitis (acute interstitial oedematous pancreatitis or necrotising pancreatitis).
Different severity of acute pancreatitis (mild pancreatitis versus moderate or severe acute pancreatitis).
Presence of persistent organ failure (mild or moderate acute pancreatitis versus severe acute pancreatitis).
Presence of infection (infected necrotising pancreatitis versus non‐infected necrotising pancreatitis).
We planned to calculate the test for subgroup differences to identify differences between subgroups.
Sensitivity analysis
We planned to perform the following sensitivity analyses defined a priori to assess the robustness of our conclusions.
Excluding trials at unclear or high risk of bias (one or more of the 'Risk of bias' domains classified as unclear or high).
Excluding trials in which either the mean or the standard deviation or both were imputed.
Imputation of binary outcomes under 'best‐best', 'best‐worst', 'worst‐best', and 'worst‐worst' scenarios.
'Summary of findings' table
Although we planned to create a 'Summary of findings' table using all the outcomes, this would have resulted in a incomprehensible table. So, we presented the 'Summary of findings' table for the primary outcomes only. We used the five GRADE considerations (study limitations, inconsistency of effect, imprecision, indirectness and publication bias) to assess the quality of the body of evidence as it related to the studies contributing data to the meta‐analyses for the prespecified outcomes. We justified all decisions to down‐ or upgrade the quality rating of studies using footnotes, making comments to aid the reader's understanding of the review where necessary. We considered whether there was any additional outcome information that we were not able to incorporate into meta‐analyses and planned to note this in the comments, stating whether it supported or contradicted the information from the meta‐analyses.
Reaching conclusions
We based our conclusions only on findings from the quantitative or narrative synthesis of included studies for this review. We have avoided making recommendations for practice, and our implications for research give the reader a clear sense of where the focus of any future research in the area should be and what the remaining uncertainties are.
Results
Description of studies
Results of the search
We identified a total of 13,825 references through electronic searches of CENTRAL (1345 records), MEDLINE (5649 records), Embase (4102 records), Science Citation Index Expanded (2604 records), World Health Organization International Clinical Trials Registry Platform (78 records) and ClinicalTrials.gov (47 records). After removing 3548 duplicates, we obtained 10,277 references. We then excluded 10,030 clearly irrelevant references through screening titles and reading abstracts. We sought 247 references for further assessment but could not obtain 2 (Hansen 1966; Perez 1980). Seven references were ongoing trials, suspended trials, or completed trials identified from clinical registers with no interim reports available (ChiCTR‐IPR‐16008301; EUCTR2014‐004844‐37‐ES; NCT01132521; NCT02025049; NCT02212392; NCT02692391; NCT02885441). We did not identify any new trials by scanning reference lists of the identified randomised trials. We excluded 102 references for the reasons listed under the table 'Characteristics of excluded studies'. In total, 136 references (84 trials) met the inclusion criteria. The reference flow is summarised in the study flow diagram (Figure 1).
Included studies
A total of 8234 participants were included in these 84 trials. Six trials (N = 658) did not report any of the outcomes of interest for this review (Birk 1994; Chooklin 2007; Marek 1999; Moreau 1986; Plaudis 2010; Wang 2013b). The remaining 78 trials excluded 210 participants after randomisation. Thus, a total of 7366 participants in 78 trials contributed to one or more outcomes for this review.
One trial included only participants with acute interstitial oedematous pancreatitis (Chen 2002a); 12 trials included only participants with acute necrotising pancreatitis (Barreda 2009; Chen 2002b; Delcenserie 2001; Dellinger 2007; Frulloni 1994; Garcia‐Barrasa 2009; Llukacaj 2012; Nordback 2001; Pederzoli 1993a; Rokke 2007; Sainio 1995; Xue 2009); the remaining trials did not state clearly whether they included any participants with acute necrotising pancreatitis. All the trials that included acute necrotising pancreatitis either stated explicitly or implied that they excluded participants with infected necrotising pancreatitis.
Two trials included only participants with mild acute pancreatitis (Chen 2002a; Yang 2012). Twenty‐six trials included only severe acute pancreatitis (Balldin 1983; Berling 1994; Birk 1994; Chen 2000; Chen 2002b; Chooklin 2007; Delcenserie 1996; Dellinger 2007; Garcia‐Barrasa 2009; Grupo Español 1996; Guo 2015; Hejtmankova 2003; Luiten 1995; Martinez 1984; Olah 2007; Pettila 2010; Plaudis 2010; Rokke 2007; Spicak 2002; Spicak 2003; Wang 2011; Wang 2013a; Wang 2016; Xia 2014; Xue 2009; Zhu 2014). Two trials reported data separately for mild and severe acute pancreatitis (Abraham 2013; Wang 2013c). These trials presented the data separately for mild pancreatitis and acute severe pancreatitis. The remaining trials either included mild and severe acute pancreatitis or did not state the severity of pancreatitis in the participants. It should be noted that none of the trials used the current definition of severe acute pancreatitis (i.e. organ failure persisting for 48 hours or more).
The potential effect modifiers, arranged by comparisons, are shown in Table 4. As shown in the table, important potential effect modifiers were missing. In addition, it appeared that most trials in the comparison on antibiotics versus no active intervention included participants with necrotising pancreatitis. Because of this, there were serious concerns about the inclusion of similar participants in the different comparisons.
2. Potential effect modifiers (ordered by comparisons).
| Study name | Treatment 1 | Treatment 2 | Severe pancreatitis | Necrotising pancreatitis | Organ failure | Infection |
| Pettila 2010 | Activated protein C | Placebo | yes | not stated | not stated | not stated |
| Barreda 2009 | Antibiotics | No active intervention | not stated | yes | not stated | not stated |
| Delcenserie 1996 | Antibiotics | No active intervention | yes | not stated | not stated | not stated |
| Delcenserie 2001 | Antibiotics | No active intervention | not stated | yes | not stated | not stated |
| Dellinger 2007 | Antibiotics | Placebo | yes | yes | not stated | no |
| Finch 1976 | Antibiotics | No active intervention | not stated | not stated | not stated | not stated |
| Garcia‐Barrasa 2009 | Antibiotics | Placebo | yes | yes | not stated | not stated |
| Hejtmankova 2003 | Antibiotics | No active intervention | yes | not stated | not stated | not stated |
| Isenmann 2004 | Antibiotics | Placebo | not stated | not stated | not stated | not stated |
| Llukacaj 2012 | Antibiotics | Placebo | not stated | yes | not stated | no |
| Luiten 1995 | Antibiotics | No active intervention | yes | not stated | not stated | no |
| Nordback 2001 | Antibiotics | Placebo | not stated | yes | no | not stated |
| Pederzoli 1993a | Antibiotics | No active intervention | not stated | yes | not stated | not stated |
| Rokke 2007 | Antibiotics | No active intervention | yes | yes | not stated | not stated |
| Sainio 1995 | Antibiotics | No active intervention | not stated | yes | not stated | not stated |
| Spicak 2002 | Antibiotics | No active intervention | yes | not stated | not stated | not stated |
| Spicak 2003 | Antibiotics | No active intervention | yes | not stated | not stated | not stated |
| Xue 2009 | Antibiotics | No active intervention | yes | yes | not stated | no |
| Bansal 2011 | Antioxidants | No active intervention | not stated | not stated | not stated | not stated |
| Birk 1994 | Antioxidants | No active intervention | yes | not stated | not stated | not stated |
| Marek 1999 | Antioxidants | Placebo | not stated | not stated | not stated | not stated |
| Sateesh 2009 | Antioxidants | No active intervention | not stated | not stated | not stated | not stated |
| Siriwardena 2007 | Antioxidants | Placebo | not stated | not stated | not stated | not stated |
| Vege 2015 | Antioxidants | Placebo | not stated | not stated | not stated | not stated |
| Chooklin 2007 | Antioxidants plus corticosteroids | No active intervention | yes | not stated | not stated | not stated |
| Balldin 1983 | Aprotinin | No active intervention | yes | not stated | not stated | not stated |
| Berling 1994 | Aprotinin | No active intervention | yes | not stated | not stated | not stated |
| Imrie 1978 | Aprotinin | Placebo | not stated | not stated | not stated | not stated |
| Imrie 1980 | Aprotinin | Placebo | not stated | not stated | not stated | not stated |
| MRC Multicentre Trial 1977 | Aprotinin | Placebo | not stated | not stated | not stated | not stated |
| Storck 1968 | Aprotinin | Placebo | not stated | not stated | not stated | not stated |
| Trapnell 1974 | Aprotinin | Placebo | not stated | not stated | not stated | not stated |
| Goebell 1979 | Calcitonin | Placebo | not stated | not stated | not stated | not stated |
| Martinez 1984 | Calcitonin | Placebo | yes | not stated | not stated | not stated |
| Perezdeoteyza 1980 | Cimetidine | Placebo | not stated | not stated | not stated | not stated |
| Sillero 1981 | Cimetidine | Placebo | not stated | not stated | not stated | not stated |
| Tykka 1985 | EDTA | Placebo | not stated | not stated | not stated | not stated |
| Buchler 1993 | Gabexate | Placebo | not stated | not stated | not stated | not stated |
| Chen 2000 | Gabexate | Placebo | yes | not stated | yes | not stated |
| Freise 1986 | Gabexate | Placebo | not stated | not stated | not stated | not stated |
| Goebell 1988 | Gabexate | Placebo | not stated | not stated | not stated | not stated |
| Valderrama 1992 | Gabexate | Placebo | not stated | not stated | not stated | not stated |
| Debas 1980 | Glucagon | Placebo | not stated | not stated | not stated | not stated |
| Dürr 1978 | Glucagon | Placebo | not stated | not stated | not stated | not stated |
| Kalima 1980 | Glucagon | Placebo | not stated | not stated | not stated | not stated |
| Kronborg 1980 | Glucagon | Placebo | not stated | not stated | not stated | not stated |
| MRC Multicentre Trial 1977 | Glucagon | Placebo | not stated | not stated | not stated | not stated |
| Hansky 1969 | Iniprol | No active intervention | not stated | not stated | not stated | not stated |
| Johnson 2001 | Lexipafant | Placebo | not stated | not stated | not stated | not stated |
| Kingsnorth 1995 | Lexipafant | Placebo | not stated | not stated | not stated | not stated |
| McKay 1997b | Lexipafant | Placebo | not stated | not stated | not stated | not stated |
| Bredkjaer 1988 | NSAID | Placebo | not stated | not stated | not stated | not stated |
| Ebbehøj 1985 | NSAID | Placebo | not stated | not stated | not stated | not stated |
| McKay 1997b | Octreotide | Placebo | not stated | not stated | not stated | not stated |
| Ohair 1993 | Octreotide | Placebo | not stated | not stated | not stated | not stated |
| Paran 1995 | Octreotide | No active intervention | not stated | not stated | not stated | not stated |
| Uhl 1999 | Octreotide | Placebo | not stated | not stated | not stated | not stated |
| Wang 2013c (mild pancreatitis) | Octreotide | No active intervention | no | not stated | not stated | not stated |
| Wang 2013c (severe pancreatitis) | Octreotide | No active intervention | yes | not stated | not stated | not stated |
| Yang 2012 | Octreotide | No active intervention | no | not stated | not stated | not stated |
| Besselink 2008 | Probiotics | Placebo | not stated | not stated | not stated | not stated |
| Olah 2007 | Probiotics | No active intervention | yes | not stated | not stated | not stated |
| Plaudis 2010 | Probiotics | No active intervention | yes | not stated | not stated | not stated |
| Sharma 2011 | Probiotics | Placebo | not stated | not stated | not stated | not stated |
| Zhu 2014 | Probiotics | Placebo | yes | not stated | not stated | not stated |
| Choi 1989 | Somatostatin | No active intervention | not stated | not stated | not stated | not stated |
| Gjørup 1992 | Somatostatin | Placebo | not stated | not stated | not stated | not stated |
| Grupo Español 1996 | Somatostatin | Placebo | yes | not stated | not stated | not stated |
| Luengo 1994 | Somatostatin | No active intervention | not stated | not stated | not stated | not stated |
| Moreau 1986 | Somatostatin | Placebo | not stated | not stated | not stated | not stated |
| Usadel 1985 | Somatostatin | Placebo | not stated | not stated | not stated | not stated |
| Wang 2013a | Somatostatin | No active intervention | yes | not stated | not stated | not stated |
| Yang 1999 | Somatostatin | No active intervention | not stated | not stated | not stated | not stated |
| Xia 2014 | Somatostatin plus omeprazole | No active intervention | yes | not stated | not stated | not stated |
| Wang 2013a | Somatostatin plus ulinastatin | No active intervention | yes | not stated | not stated | not stated |
| Wang 2011 | Thymosin | Placebo | yes | not stated | not stated | not stated |
| Abraham 2013 (mild pancreatitis) | Ulinastatin | Placebo | no | not stated | not stated | no |
| Abraham 2013 (severe pancreatitis) | Ulinastatin | Placebo | yes | not stated | not stated | not stated |
| Frulloni 1994 | Gabexate | Aprotinin | not stated | yes | not stated | not stated |
| Pederzoli 1993b | Gabexate | Aprotinin | not stated | not stated | not stated | not stated |
| Kirsch 1978 | Glucagon | Atropine | not stated | not stated | not stated | not stated |
| Chen 2002a | Ulinastatin | Gabexate | no | no | no | not stated |
| MRC Multicentre Trial 1977 | Aprotinin | Glucagon | not stated | not stated | not stated | not stated |
| Guo 2015 | Octerotide plus ulinastatin | Octreotide | yes | not stated | not stated | not stated |
| Wang 2013b | Octreotide plus NSAID | Octreotide | not stated | not stated | not stated | not stated |
| Chen 2002b | Ulinastatin | Octreotide | yes | yes | not stated | not stated |
| Gilsanz 1978 | Glucagon | Oxyphenonium | not stated | not stated | not stated | not stated |
| Poropat 2015 | Antibiotics | No active intervention | not stated | not stated | not stated | no |
| Wang 2016 | Somatostatin plus gabexate | Somatostatin | yes | not stated | not stated | not stated |
| Wang 2013a | Somatostatin plus ulinastatin | Somatostatin | yes | not stated | not stated | not stated |
| Wang 2016 | Somatostatin plus ulinastatin | Somatostatin | yes | not stated | not stated | not stated |
| Wang 2016 | Somatostatin plus ulinastatin plus gabexate | Somatostatin | yes | not stated | not stated | not stated |
| Wang 2016 | Somatostatin plus ulinastatin | Somatostatin plus gabexate | yes | not stated | not stated | not stated |
| Wang 2016 | Somatostatin plus ulinastatin plus gabexate | Somatostatin plus gabexate | yes | not stated | not stated | not stated |
| Wang 2016 | Somatostatin plus ulinastatin plus gabexate | Somatostatin plus ulinastatin | yes | not stated | not stated | not stated |
Source of funding: seven trials were not funded or they were funded by agencies without vested interest in results (Bansal 2011; Garcia‐Barrasa 2009; Wang 2013a; Wang 2013c; Wang 2016; Xue 2009; Yang 2012). Pharmaceutical companies partially or fully funded 21 trials (Balldin 1983; Berling 1994; Besselink 2008; Dellinger 2007; Ebbehøj 1985; Hansky 1969; Imrie 1978; Isenmann 2004; Johnson 2001; Kingsnorth 1995; McKay 1997b; Moreau 1986; MRC Multicentre Trial 1977; Pettila 2010; Rokke 2007; Sharma 2011; Siriwardena 2007; Trapnell 1974; Tykka 1985; Uhl 1999; Valderrama 1992). The source of funding was not available from the remaining trials.
Excluded studies
None of the excluded studies were eligible for this review. The reasons for exclusion are listed in 'Characteristics of excluded studies'.
Risk of bias in included studies
We summarised the overall risk of bias in Figure 2 and Figure 3. Only Wang 2016 was at low risk of bias in all the domains and can be considered a trial at overall low risk of bias.
2.
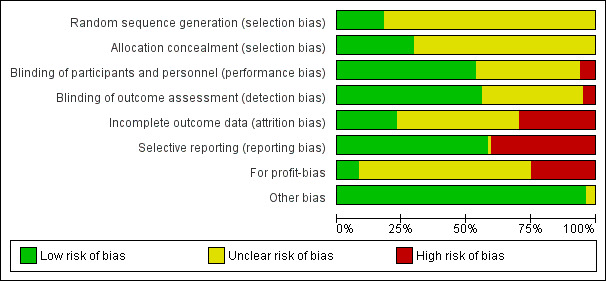
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.
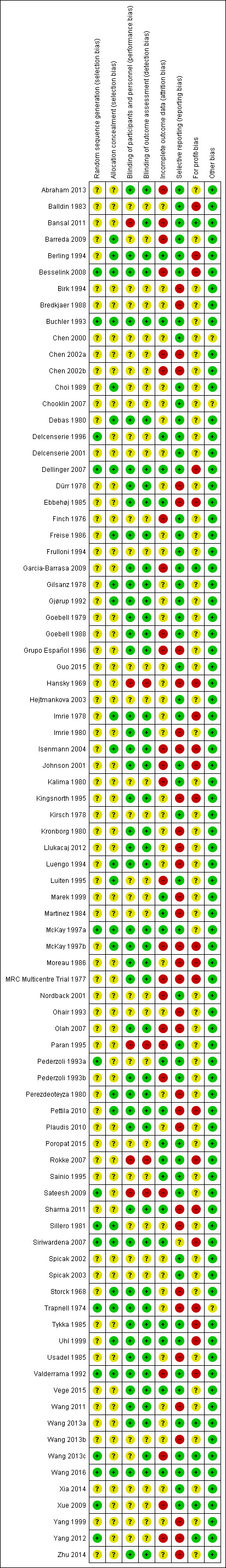
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Fifteen trials were at low risk of bias for random sequence generation (Besselink 2008; Buchler 1993; Delcenserie 1996; Dellinger 2007; McKay 1997a; Pederzoli 1993a; Sateesh 2009; Sillero 1981; Siriwardena 2007; Trapnell 1974; Valderrama 1992; Wang 2013c; Wang 2016; Xue 2009; Yang 2012). Twenty‐six trials were at low risk of bias for allocation concealment (Barreda 2009; Berling 1994; Besselink 2008; Buchler 1993; Choi 1989; Debas 1980; Dellinger 2007; Freise 1986; Gilsanz 1978; Gjørup 1992; Imrie 1978; Isenmann 2004; Luengo 1994; Luiten 1995; McKay 1997a; McKay 1997b; Perezdeoteyza 1980; Pettila 2010; Sharma 2011; Sillero 1981; Siriwardena 2007; Storck 1968; Trapnell 1974; Uhl 1999; Valderrama 1992; Wang 2016). Eight trials were at low risk of selection bias (Besselink 2008; Buchler 1993; Dellinger 2007; McKay 1997a; Siriwardena 2007; Trapnell 1974; Valderrama 1992; Wang 2016). The remaining trials were at unclear risk of selection bias since they did not describe random sequence generation or allocation concealment.
Blinding
Forty‐five trials were at low risk of bias for blinding of participants, healthcare providers, and outcomes assessors (Abraham 2013; Berling 1994; Besselink 2008; Buchler 1993; Debas 1980; Dellinger 2007; Dürr 1978; Ebbehøj 1985; Freise 1986; Garcia‐Barrasa 2009; Gilsanz 1978; Gjørup 1992; Goebell 1979; Goebell 1988; Grupo Español 1996; Imrie 1978; Imrie 1980; Isenmann 2004; Johnson 2001; Kingsnorth 1995; Kronborg 1980; Llukacaj 2012; Luengo 1994; McKay 1997a; McKay 1997b; Moreau 1986; MRC Multicentre Trial 1977; Olah 2007; Pederzoli 1993b; Perezdeoteyza 1980; Pettila 2010; Plaudis 2010; Sharma 2011; Siriwardena 2007; Storck 1968; Trapnell 1974; Tykka 1985; Uhl 1999; Usadel 1985; Valderrama 1992; Vege 2015; Wang 2011; Wang 2013a; Wang 2016; Zhu 2014). While Bansal 2011 and Wang 2013c were also at low risk of bias for the blinding of outcome assessors, Bansal 2011 was at high risk and Wang 2013c at unclear risk for the blinding of participants and healthcare providers. Overall, five trials were at high risk of bias due to lack of blinding (Bansal 2011; Hansky 1969; Paran 1995; Rokke 2007; Sateesh 2009). The remaining trials were at unclear risk of bias for blinding.
Incomplete outcome data
Nineteen trials were at low risk of attrition bias due to missing outcome data (Berling 1994; Buchler 1993; Delcenserie 1996; Dellinger 2007; Ebbehøj 1985; Marek 1999; Martinez 1984; McKay 1997a; Pederzoli 1993a; Pettila 2010; Poropat 2015; Rokke 2007; Sainio 1995; Sharma 2011; Siriwardena 2007; Tykka 1985; Uhl 1999; Vege 2015; Wang 2016). Twenty‐five trials were at high risk of attrition bias (Abraham 2013; Bansal 2011; Barreda 2009; Besselink 2008; Chen 2002a; Chen 2002b; Finch 1976; Garcia‐Barrasa 2009; Goebell 1988; Grupo Español 1996; Isenmann 2004; Johnson 2001; Kalima 1980; Luiten 1995; McKay 1997b; MRC Multicentre Trial 1977; Nordback 2001; Olah 2007; Paran 1995; Pederzoli 1993b; Sateesh 2009; Valderrama 1992; Wang 2013c; Xue 2009; Yang 2012). The remaining trials were at unclear risk of attrition bias.
Selective reporting
Forty‐nine trials were at low risk of selective reporting bias (Abraham 2013; Balldin 1983; Bansal 2011; Barreda 2009; Berling 1994; Besselink 2008; Buchler 1993; Chen 2000; Choi 1989; Debas 1980; Delcenserie 1996; Delcenserie 2001; Dellinger 2007; Finch 1976; Freise 1986; Frulloni 1994; Garcia‐Barrasa 2009; Gilsanz 1978; Gjørup 1992; Goebell 1979; Goebell 1988; Guo 2015; Hejtmankova 2003; Imrie 1978; Johnson 2001; Kalima 1980; Kirsch 1978; Luiten 1995; McKay 1997a; Nordback 2001; Paran 1995; Pederzoli 1993a; Pederzoli 1993b; Poropat 2015; Rokke 2007; Sainio 1995; Sateesh 2009; Siriwardena 2007; Spicak 2002; Spicak 2003; Tykka 1985; Uhl 1999; Valderrama 1992; Vege 2015; Wang 2013a; Wang 2013c; Wang 2016; Xia 2014; Xue 2009). The remaining trials were at high or unclear risk of reporting bias.
Other potential sources of bias
Source of funding bias: seven trials were at low risk of due to source of funding (Bansal 2011; Garcia‐Barrasa 2009; Wang 2013a; Wang 2013c; Wang 2016; Xue 2009; Yang 2012). Twenty‐one trials were at high risk of bias due to source of funding (Balldin 1983; Berling 1994; Besselink 2008; Dellinger 2007; Ebbehøj 1985; Hansky 1969; Imrie 1978; Isenmann 2004; Johnson 2001; Kingsnorth 1995; McKay 1997b; Moreau 1986; MRC Multicentre Trial 1977; Pettila 2010; Rokke 2007; Sharma 2011; Siriwardena 2007; Trapnell 1974; Tykka 1985; Uhl 1999; Valderrama 1992). The remaining trials were at unclear risk of bias due to the source of funding.
No other bias was noted in any of the trials.
Effects of interventions
Primary outcomes
Mortality
Short‐term mortality
A total of 67 studies (N = 6638) reported short‐term mortality (Abraham 2013; Balldin 1983; Bansal 2011; Barreda 2009; Berling 1994; Besselink 2008; Buchler 1993; Chen 2000; Choi 1989; Debas 1980; Delcenserie 1996; Delcenserie 2001; Dellinger 2007; Dürr 1978; Finch 1976; Freise 1986; Frulloni 1994; Garcia‐Barrasa 2009; Gjørup 1992; Goebell 1979; Goebell 1988; Grupo Español 1996; Guo 2015; Hansky 1969; Hejtmankova 2003; Imrie 1978; Imrie 1980; Johnson 2001; Kalima 1980; Kingsnorth 1995; Kirsch 1978; Kronborg 1980; Llukacaj 2012; Luengo 1994; Luiten 1995; Martinez 1984; McKay 1997a; McKay 1997b; MRC Multicentre Trial 1977; Nordback 2001; Olah 2007; Paran 1995; Pederzoli 1993a; Pederzoli 1993b; Perezdeoteyza 1980; Pettila 2010; Poropat 2015; Rokke 2007; Sainio 1995; Sateesh 2009; Siriwardena 2007; Spicak 2002; Spicak 2003; Storck 1968; Trapnell 1974; Tykka 1985; Uhl 1999; Usadel 1985; Valderrama 1992; Vege 2015; Wang 2011; Wang 2013a; Wang 2013c; Wang 2016; Xia 2014; Xue 2009; Yang 2012). There was no evidence of difference in any of the comparisons (Analysis 1.1).
1.1. Analysis.
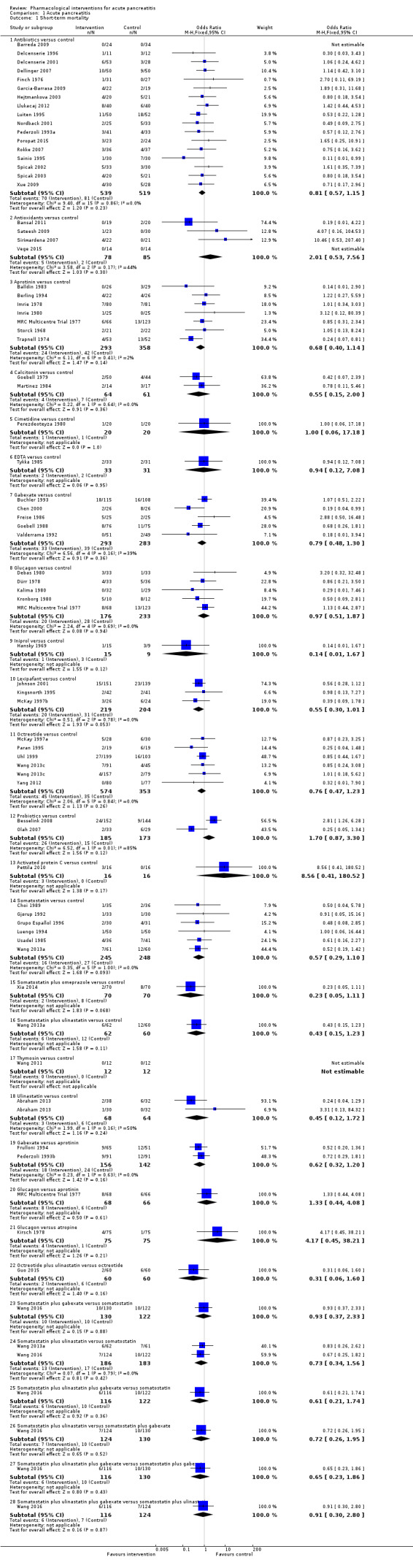
Comparison 1 Acute pancreatitis, Outcome 1 Short‐term mortality.
Long‐term mortality (maximum follow‐up)
Only one study (N = 62) reported mortality beyond six months (Gilsanz 1978). There was no evidence of difference in the only comparison possible.
Serious adverse events
A total of 17 studies (N = 1139) reported serious adverse events as a proportion or participants who experienced at least one serious adverse event (i.e. each person with a serious adverse event will be counted only once regardless of the number of serious adverse events that the person develops) (Bansal 2011; Chen 2002a; Debas 1980; Delcenserie 1996; Dellinger 2007; Freise 1986; Frulloni 1994; Garcia‐Barrasa 2009; Gjørup 1992; Goebell 1988; Kalima 1980; Llukacaj 2012; McKay 1997a; Sainio 1995; Siriwardena 2007; Tykka 1985; Yang 1999). There was no evidence of difference in any of the comparisons (Analysis 1.2).
1.2. Analysis.
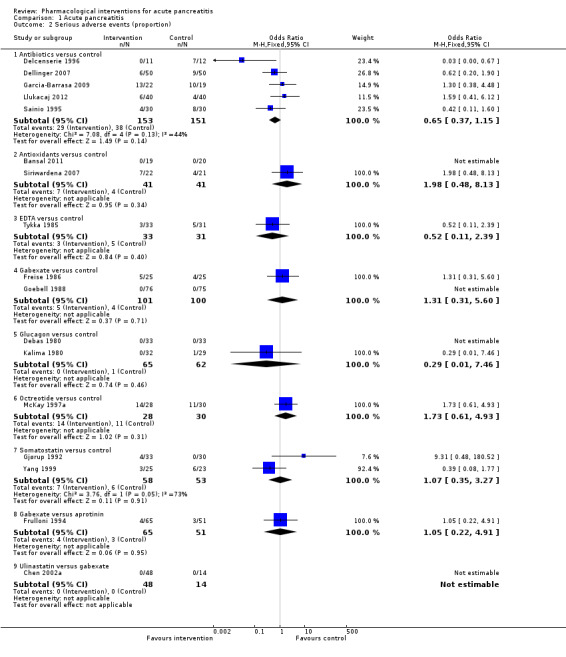
Comparison 1 Acute pancreatitis, Outcome 2 Serious adverse events (proportion).
A total of 37 studies (N = 3804) reported the number of serious adverse events observed in all participants (i.e. if a person develops more than one serious adverse event, the number of serious adverse events that the person develops is included) (Balldin 1983; Bansal 2011; Barreda 2009; Berling 1994; Besselink 2008; Buchler 1993; Chen 2000; Choi 1989; Debas 1980; Delcenserie 1996; Delcenserie 2001; Garcia‐Barrasa 2009; Gjørup 1992; Guo 2015; Imrie 1978; Isenmann 2004; Johnson 2001; Kirsch 1978; McKay 1997a; Nordback 2001; Olah 2007; Paran 1995; Pederzoli 1993a; Poropat 2015; Sainio 1995; Sillero 1981; Spicak 2002; Spicak 2003; Tykka 1985; Uhl 1999; Valderrama 1992; Vege 2015; Wang 2013a; Wang 2013c; Xia 2014; Xue 2009; Zhu 2014). There were fewer serious adverse events in participants receiving lexipafant (rate ratio 0.67, 95% CI 0.46 to 0.96; participants = 290; studies = 1), octreotide (rate ratio 0.74, 95% CI 0.60 to 0.89; participants = 770; studies = 5), somatostatin plus omeprazole (rate ratio 0.36, 95% CI 0.19 to 0.70; participants = 140; studies = 1), and somatostatin plus ulinastatin (rate ratio 0.30, 95% CI 0.15 to 0.60; participants = 122; studies = 1) than control. There were also fewer serious adverse events in participants taking octreotide plus ulinastatin compared to octreotide (rate ratio 0.30, 95% CI 0.17 to 0.51; participants = 120; studies = 1) and in participants taking somatostatin plus ulinastatin versus somatostatin (rate ratio 0.28, 95% CI 0.15 to 0.56; participants = 123; studies = 1). There was no evidence of difference in the remaining comparisons (Analysis 1.3).
1.3. Analysis.
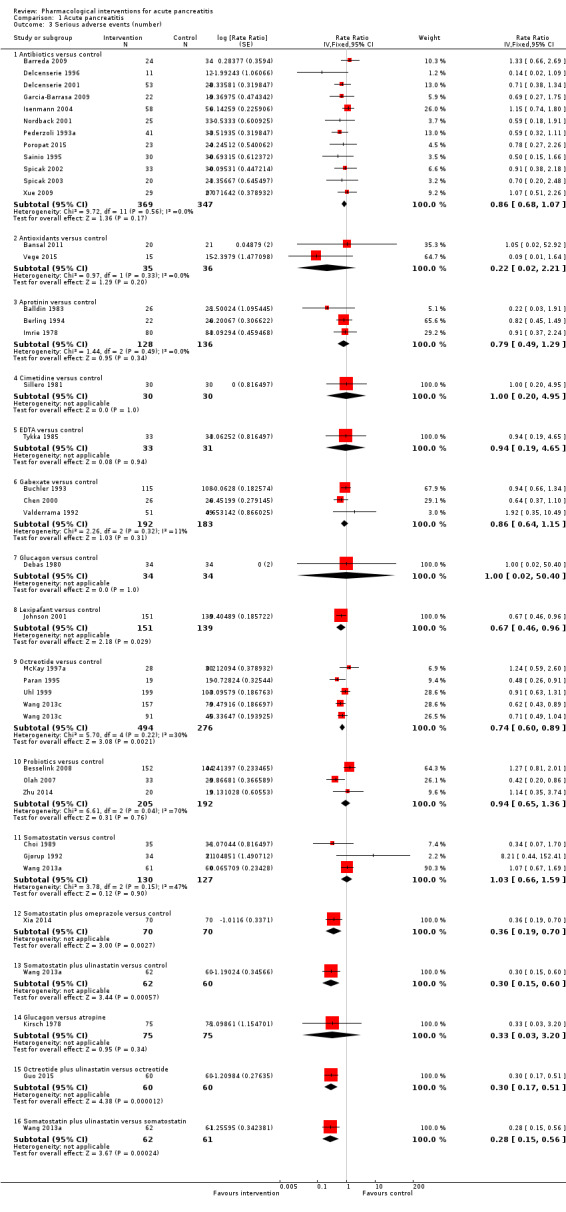
Comparison 1 Acute pancreatitis, Outcome 3 Serious adverse events (number).
Organ failure
A total of 18 studies (N = 2220) reported organ failure (Abraham 2013; Bansal 2011; Besselink 2008; Delcenserie 1996; Freise 1986; Garcia‐Barrasa 2009; Johnson 2001; McKay 1997a; McKay 1997b; Olah 2007; Pederzoli 1993a; Poropat 2015; Rokke 2007; Sateesh 2009; Siriwardena 2007; Vege 2015; Wang 2013c; Wang 2016). The proportion of people with organ failure was lower in the octreotide group than in control (OR 0.51, 95% CI 0.27 to 0.97; participants = 430; studies = 3). There was no evidence of difference in any of the remaining comparisons (Analysis 1.4).
1.4. Analysis.
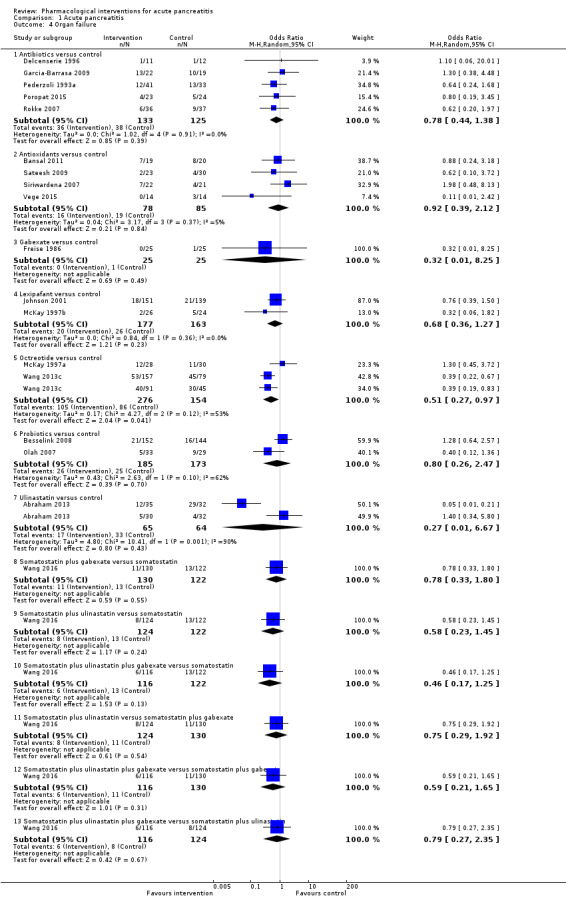
Comparison 1 Acute pancreatitis, Outcome 4 Organ failure.
Infected pancreatic necrosis
A total of 15 studies (N = 1173) reported infected pancreatic necrosis (Barreda 2009; Besselink 2008; Delcenserie 1996; Dellinger 2007; Garcia‐Barrasa 2009; Isenmann 2004; Llukacaj 2012; McKay 1997a; Olah 2007; Pederzoli 1993a; Poropat 2015; Rokke 2007; Spicak 2002; Spicak 2003; Zhu 2014). As shown in Analysis 1.5, there was no evidence of difference in any of the comparisons.
1.5. Analysis.
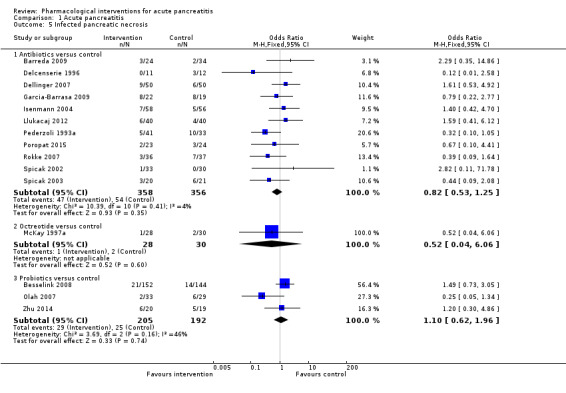
Comparison 1 Acute pancreatitis, Outcome 5 Infected pancreatic necrosis.
Sepsis
A total of 11 studies (N = 1350) reported sepsis (Balldin 1983; Berling 1994; Buchler 1993; Freise 1986; Frulloni 1994; Johnson 2001; Olah 2007; Paran 1995; Sainio 1995; Uhl 1999; Valderrama 1992). The proportion of people with sepsis was lower in those receiving lexipafant compared to control (OR 0.26, 95% CI 0.08 to 0.83; participants = 290; studies = 1). There was no evidence of difference in any of the remaining comparisons (Analysis 1.6).
1.6. Analysis.
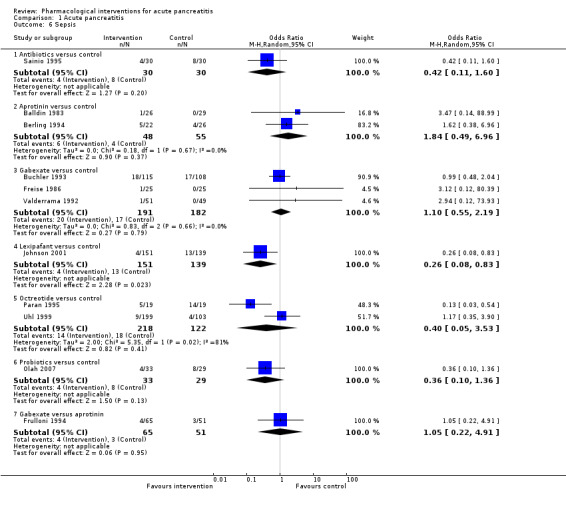
Comparison 1 Acute pancreatitis, Outcome 6 Sepsis.
Health‐related quality of life
None of the trials reported health‐related quality of life at any time point.
Secondary outcomes
Adverse events
A total of 27 studies (N = 2807) reported adverse events as a proportion or participants who experienced at least one adverse event (i.e. each person with an adverse event will be counted only once regardless of the number of adverse events that the person develops) (Bansal 2011; Buchler 1993; Chen 2002a; Chen 2002b; Debas 1980; Dellinger 2007; Finch 1976; Freise 1986; Frulloni 1994; Gjørup 1992; Goebell 1979; Kalima 1980; Kingsnorth 1995; Llukacaj 2012; McKay 1997a; Nordback 2001; Olah 2007; Paran 1995; Pederzoli 1993b; Rokke 2007; Sainio 1995; Tykka 1985; Uhl 1999; Valderrama 1992; Wang 2016; Xia 2014; Yang 1999). This proportion was lower in those receiving antibiotics (OR 0.51, 95% CI 0.32 to 0.80; participants = 429; studies = 6) and somatostatin plus omeprazole (OR 0.00, 95% CI 0.00 to 0.04; participants = 140; studies = 1) compared to control. There was no evidence of difference in the remaining comparisons (Analysis 1.7).
1.7. Analysis.
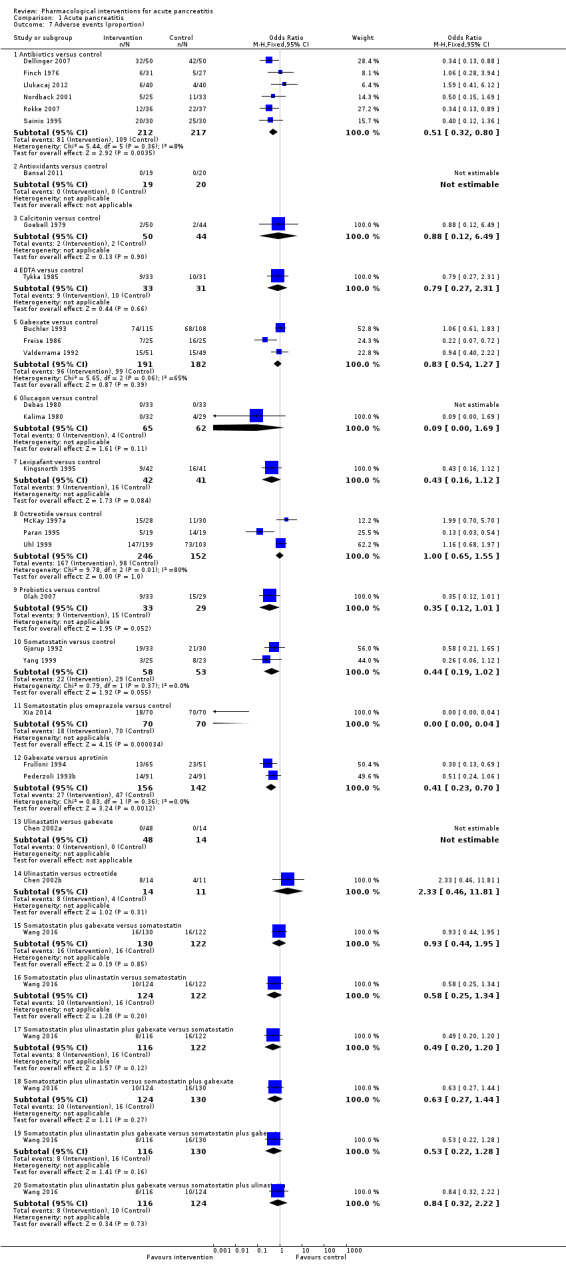
Comparison 1 Acute pancreatitis, Outcome 7 Adverse events (proportion).
A total of 40 studies (N = 3894) reported the number of adverse events observed in all participants (i.e. if a person develops more than one adverse event, the number of adverse events that the person develops is included) (Abraham 2013; Balldin 1983; Bansal 2011; Barreda 2009; Berling 1994; Besselink 2008; Buchler 1993; Chen 2000; Choi 1989; Debas 1980; Garcia‐Barrasa 2009; Gilsanz 1978; Gjørup 1992; Goebell 1979; Guo 2015; Hejtmankova 2003; Imrie 1978; Isenmann 2004; Johnson 2001; Kirsch 1978; Kronborg 1980; Luiten 1995; McKay 1997a; Nordback 2001; Olah 2007; Paran 1995; Pederzoli 1993a; Pederzoli 1993b; Poropat 2015; Sainio 1995; Sateesh 2009; Sillero 1981; Spicak 2002; Spicak 2003; Tykka 1985; Uhl 1999; Valderrama 1992; Wang 2013c; Xue 2009; Zhu 2014). Compared to control, there were fewer adverse events in participants receiving antibiotics (rate ratio 0.75, 95% CI 0.58 to 0.95; participants = 755; studies = 12), gabexate (rate ratio 0.76, 95% CI 0.61 to 0.95; participants = 375; studies = 3), and lexipafant (rate ratio 0.61, 95% CI 0.44 to 0.85; participants = 290; studies = 1). There were also fewer adverse events for the octreotide plus ulinastatin group versus ulinastatin alone (rate ratio 0.29, 95% CI 0.17 to 0.48; participants = 120; studies = 1). There was no evidence of difference in any of the remaining comparisons (Analysis 1.8).
1.8. Analysis.
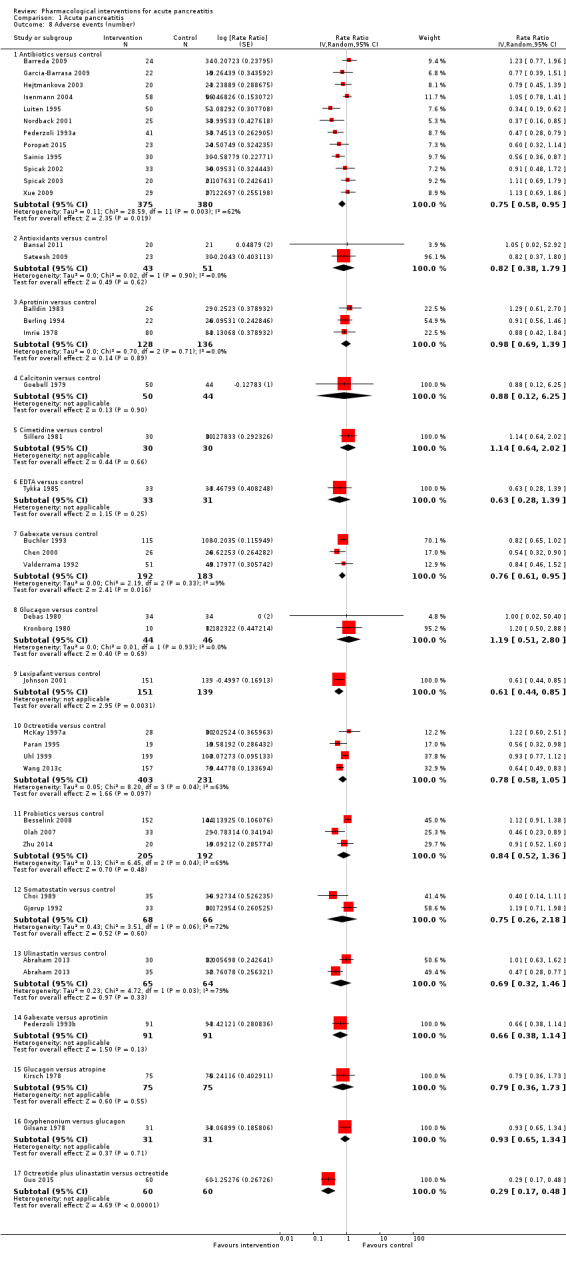
Comparison 1 Acute pancreatitis, Outcome 8 Adverse events (number).
Measures of decreased complication or earlier recovery
Length of hospital stay
Forty‐four trials (N = 4405) reported the length of hospital stay (Abraham 2013; Balldin 1983; Bansal 2011; Barreda 2009; Berling 1994; Besselink 2008; Bredkjaer 1988; Buchler 1993; Debas 1980; Delcenserie 1996; Dürr 1978; Ebbehøj 1985; Finch 1976; Garcia‐Barrasa 2009; Gjørup 1992; Goebell 1979; Guo 2015; Hansky 1969; Hejtmankova 2003; Isenmann 2004; Johnson 2001; Luengo 1994; Luiten 1995; Martinez 1984; McKay 1997a; McKay 1997b; Ohair 1993; Olah 2007; Paran 1995; Pettila 2010; Rokke 2007; Sainio 1995; Sateesh 2009; Sharma 2011; Siriwardena 2007; Spicak 2002; Spicak 2003; Uhl 1999; Vege 2015; Wang 2011; Wang 2013c; Wang 2016; Xue 2009; Yang 2012). Since most trials did not report the mean and standard deviation, we reported this outcome in Table 5. As seen in the table, none of the interventions consistently decreased length of hospital stay.
3. Length of hospital stay (days).
| Study name | Intervention | Comparator | Number of participants in intervention | Number of participants in control | Mean or median (standard deviation or interquartile range, if reported) hospital stay in intervention group | Mean or median (standard deviation or interquartile range, if reported) hospital stay in control group | Difference | Statistical significance (P‐value if reported) |
| Barreda 2009 | Antibiotics | No active intervention | 24 | 34 | 54 | 45 | 9 | Not significant |
| Delcenserie 1996 | Antibiotics | No active intervention | 11 | 12 | 27.8 | 22 | 5.8 | Not significant |
| Finch 1976 | Antibiotics | No active intervention | 31 | 27 | 10.4 | 11.3 | −0.9 | Not significant |
| Garcia‐Barrasa 2009 | Antibiotics | Placebo | 22 | 19 | 21 | 19 | 2 | Not significant (0.80) |
| Hejtmankova 2003 | Antibiotics | No active intervention | 20 | 21 | 18 (7.2) | 25 (14.8) | −7 | Not significant |
| Isenmann 2004 | Antibiotics | Placebo | 58 | 56 | 21 | 18 | 3 | Not significant |
| Luiten 1995 | Antibiotics | No active intervention | 50 | 52 | 30 | 32 | −2 | Not significant |
| Rokke 2007 | Antibiotics | No active intervention | 36 | 37 | 18 | 22 | −4 | Not significant (0.32) |
| Sainio 1995 | Antibiotics | No active intervention | 30 | 30 | 33.2 (22.1) | 43.8 (43.1) | −10.6 | Not significant (0.24) |
| Spicak 2002 | Antibiotics | No active intervention | 33 | 30 | 18.9 (8.1) | 23.8 (19.3) | −4.9 | Not significant |
| Spicak 2003 | Antibiotics | No active intervention | 20 | 21 | 18 (7.2) | 25 (14.8) | −7 | Not significant |
| Xue 2009 | Antibiotics | No active intervention | 29 | 27 | 28.3 | 30.7 | −2.4 | Not significant |
| Bansal 2011 | Antioxidants | No active intervention | 19 | 20 | 12.8 | 15.1 | −2.3 | Not significant |
| Sateesh 2009 | Antioxidants | No active intervention | 23 | 30 | 7.2 (5) | 10.3 (7) | −3.1 | Not significant (0.07) |
| Siriwardena 2007 | Antioxidants | Placebo | 22 | 21 | 20.4 (24.4) | 14.3 (15.7) | 6.1 | Not significant (0.34) |
| Vege 2015 | Antioxidants | Placebo | 14 | 14 | 3 | 5 | −2 | Not significant (0.06) |
| Balldin 1983 | Aprotinin | No active intervention | 26 | 29 | 17.3 | 16.5 | 0.8 | Not significant |
| Berling 1994 | Aprotinin | No active intervention | 22 | 26 | 25 (15‐32) | 33 (17‐38) | −8 | Not significant (0.24) |
| Goebell 1979 | Calcitonin | Placebo | 50 | 44 | 18.3 (6.4) | 20.2 (7.5) | −1.9 | Not significant |
| Martinez 1984 | Calcitonin | Placebo | 14 | 17 | 24 (20.2) | 30 (21.7) | −6 | Not significant |
| Buchler 1993 | Gabexate | Placebo | 115 | 108 | 26 (20‐43) | 23 (28‐34) | 3 | Not significant |
| Debas 1980 | Glucagon | Placebo | 33 | 33 | 26 (28.7) | 20 (19.2) | 6 | Not significant |
| Dürr 1978 | Glucagon | Placebo | 33 | 36 | 32.6 | 26.9 | 5.7 | Not significant |
| Hansky 1969 | Iniprol | No active intervention | 15 | 9 | 14.7 (9.3) | 18.7 (10.2) | −4 | Not significant |
| Johnson 2001 | Lexipafant | Placebo | 151 | 139 | 9 | 10 | −1 | Not significant |
| McKay 1997b | Lexipafant | Placebo | 26 | 24 | 13.3 | 14.9 | −1.6 | Not significant |
| Bredkjaer 1988 | NSAID | Placebo | 27 | 30 | 9 | 10 | −1 | Not significant |
| Ebbehøj 1985 | NSAID | Placebo | 14 | 16 | 13 | 15 | −2 | Not significant |
| McKay 1997a | Octreotide | Placebo | 28 | 30 | 10 | 10 | 0 | Not significant |
| Ohair 1993 | Octreotide | Placebo | 90 | 90 | 7.3 | 8.2 | −0.9 | Not significant |
| Paran 1995 | Octreotide | No active intervention | 19 | 19 | 17.9 (13.2) | 34.1 (22.7) | −16.2 | Significant (0.02) |
| Uhl 1999 | Octreotide | Placebo | 199 | 103 | 21.5 | 21 | 0.5 | Not significant |
|
Wang 2013c (mild acute pancreatitis) |
Octreotide | No active intervention | 157 | 79 | 14.4 | 15.37 | −0.97 | Not significant |
|
Wang 2013c (severe acute pancreatitis) |
Octreotide | No active intervention | 91 | 45 | 16 | 16 | 0 | Not significant |
| Yang 2012 | Octreotide | No active intervention | 80 | 77 | 7.4 (2) | 11.8 (4) | −4.4 | Significant |
| Besselink 2008 | Probiotics | Placebo | 152 | 144 | 28.9 (41.5) | 23.5 (25.9) | 5.4 | Not significant (0.98) |
| Olah 2007 | Probiotics | No active intervention | 33 | 29 | 14.9 | 19.7 | −4.8 | Not significant |
| Sharma 2011 | Probiotics | Placebo | 24 | 26 | 13.23 (18.19) | 9.69 (9.69) | 3.54 | Not significant (0.76) |
| Pettila 2010 | Activated protein C | Placebo | 16 | 16 | 17.1 | 34.4 | −17.3 | Significant (P < 0.05) |
| Gjørup 1992 | Somatostatin | Placebo | 33 | 30 | 12 | 10 | 2 | Not significant |
| Luengo 1994 | Somatostatin | No active intervention | 50 | 50 | 14.92 (11.46) | 20.28 (15) | −5.36 | Significant |
| Wang 2011 | Thymosin | Placebo | 12 | 12 | 37.1 (22.7) | 60.6 (32.9) | −23.5 | Not significant (0.06) |
|
Abraham 2013 (mild acute pancreatitis) |
Ulinastatin | Placebo | 30 | 32 | 7 (5‐22) | 8 (5‐15) | −1 | Not significant (0.07) |
|
Abraham 2013 (severe acute pancreatitis) |
Ulinastatin | Placebo | 35 | 32 | 9 (6‐22) | 10 (6‐22) | −1 | Not significant (0.21) |
| Guo 2015 | Octerotide plus ulinastatin | Octreotide | 60 | 60 | 11.8 (3.9) | 23.7 (16.3) | −11.9 | Significant |
| Wang 2016 | Somatostatin plus ulinastatin plus gabexate | Somatostatin | 116 | 122 | 17.7 (32.1) | 31.3 (37.6) | ‐13.6 | Significant |
| Wang 2016 | Somatostatin plus ulinastatin | Somatostatin | 124 | 122 | 22.6 (34.5) | 31.3 (37.6) | ‐8.7 | Significant |
| Wang 2016 | Somatostatin plus gabexate | Somatostatin | 130 | 122 | 23.2 (29.6) | 31.3 (37.6) | ‐8.1 | Significant |
| Wang 2016 | Somatostatin plus ulinastatin plus gabexate | Somatostatin plus gabexate | 116 | 130 | 17.7 (32.1) | 23.2 (29.6) | −5.5 | Significant |
| Wang 2016 | Somatostatin plus ulinastatin | Somatostatin plus gabexate | 124 | 130 | 22.6 (34.5) | 23.2 (29.6) | −0.6 | Significant |
| Wang 2016 | Somatostatin plus ulinastatin plus gabexate | Somatostatin plus ulinastatin | 116 | 124 | 17.7 (32.1) | 22.6 (34.5) | −4.9 | Significant |
NSAID: non‐steroidal anti‐inflammatory drug.
Length of intensive care unit stay
Thirteen trials (N = 1188) reported the length of intensive care unit (ICU) stay (Berling 1994; Besselink 2008; Garcia‐Barrasa 2009; Isenmann 2004; Johnson 2001; Nordback 2001; Rokke 2007; Sainio 1995; Sharma 2011; Siriwardena 2007; Spicak 2002; Vege 2015; Wang 2011). Since most trials did not report the mean and standard deviation, we reported the ICU stay in Table 6. As seen in the table, none of the interventions consistently decreased length of ICU stay.
4. Length of intensive care unit (ICU) stay (days).
| Study name | Intervention | Control | Number of participants in intervention | Number of participants in control | Mean or median (standard deviation or interquartile range, if reported) intensive care stay in intervention group | Mean or median (standard deviation or interquartile range, if reported) intensive care stay in control group | Difference | Statistical significance (P‐value, reported) |
| Garcia‐Barrasa 2009 | Antibiotics | Placebo | 22 | 19 | 17 | 18 | ‐1 | Not significant (P‐value = 0.83) |
| Isenmann 2004 | Antibiotics | Placebo | 58 | 56 | 8 | 6 | 2 | Not significant |
| Nordback 2001 | Antibiotics | Placebo | 25 | 33 | 8 | 8 | 0 | Not significant |
| Rokke 2007 | Antibiotics | No active intervention | 36 | 37 | 8 | 7 | 1 | Not significant (P‐value = 0.78) |
| Sainio 1995 | Antibiotics | No active intervention | 30 | 30 | 12.7 (10.7) | 23.6 (28.7) | ‐10.9 | Not significant (P‐value = 0.06) |
| Spicak 2002 | Antibiotics | No active intervention | 33 | 30 | 11.4 (5.4) | 15.9 (12) | ‐4.5 | Not significant |
| Siriwardena 2007 | Antioxidants | Placebo | 22 | 21 | 4 (10.3) | 0 (0) | 4 | Not significant (P‐value = 0.08) |
| Vege 2015 | Antioxidants | Placebo | 14 | 14 | 0 | 0 | 0 | Significant (P‐value = 0.03) |
| Berling 1994 | Aprotinin | No active intervention | 22 | 26 | 9.5 (4 ‐ 10) | 12 (3‐20) | ‐2.5 | Not significant (P‐value = 0.47) |
| Johnson 2001 | Lexipafant | Placebo | 151 | 139 | 9.5 | 11 | ‐1.5 | Not significant |
| Besselink 2008 | Probiotics | Placebo | 152 | 144 | 6.6 (17.1) | 3 (9.3) | 3.6 | Not significant (P‐value = 0.08) |
| Sharma 2011 | Probiotics | Placebo | 24 | 26 | 4.94 (9.54) | 4 (5.86) | 0.94 | Not significant (P‐value = 0.94) |
| Wang 2011 | Thymosin | Placebo | 12 | 12 | 24.6 (19.6) | 50.5 (25.7) | ‐25.9 | Significant (P‐value = 0.01) |
Requirement for additional invasive intervention
A total of 32 studies (N = 3495) reported requirement for additional invasive intervention (Barreda 2009; Berling 1994; Besselink 2008; Buchler 1993; Chen 2000; Delcenserie 1996; Dürr 1978; Garcia‐Barrasa 2009; Gilsanz 1978; Goebell 1979; Goebell 1988; Hejtmankova 2003; Isenmann 2004; Llukacaj 2012; Luengo 1994; Luiten 1995; Martinez 1984; MRC Multicentre Trial 1977; Nordback 2001; Ohair 1993; Olah 2007; Pederzoli 1993a; Pederzoli 1993b; Rokke 2007; Sainio 1995; Sillero 1981; Spicak 2002; Spicak 2003; Tykka 1985; Uhl 1999; Wang 2013c; Xue 2009). The proportion of people who needed an additional invasive intervention was lower in the gabexate group compared to control (OR 0.58, 95% CI 0.37 to 0.90; participants = 426; studies = 3). There was no evidence of difference in any of the remaining comparisons (Analysis 1.9).
1.9. Analysis.
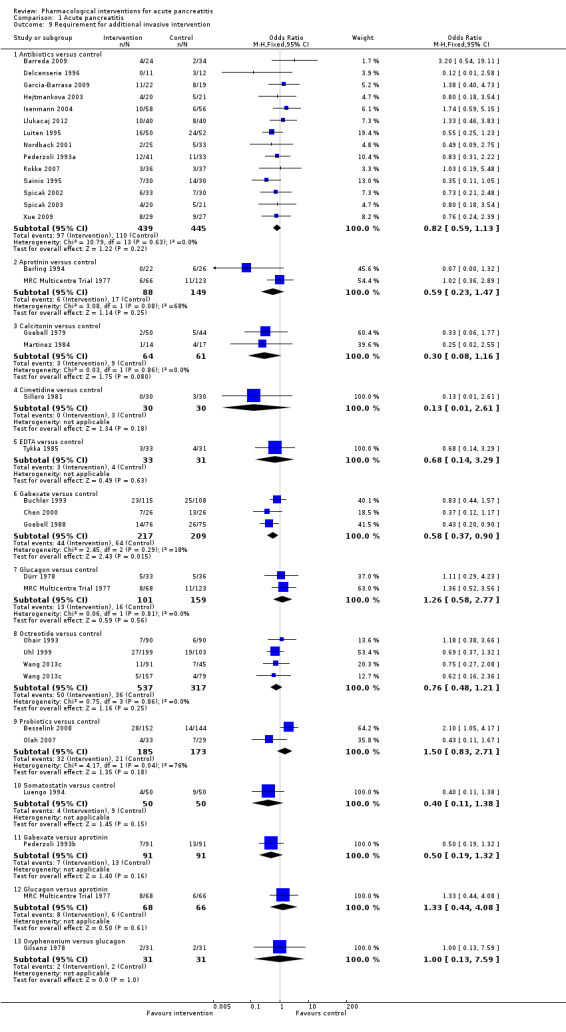
Comparison 1 Acute pancreatitis, Outcome 9 Requirement for additional invasive intervention.
Endoscopic or radiological drainage of collections
Three studies (N = 436) reported endoscopic or radiological drainage of collections (Delcenserie 1996; Wang 2013c; Zhu 2014). As shown in Analysis 1.10, there was no evidence of difference in any of the comparisons.
1.10. Analysis.
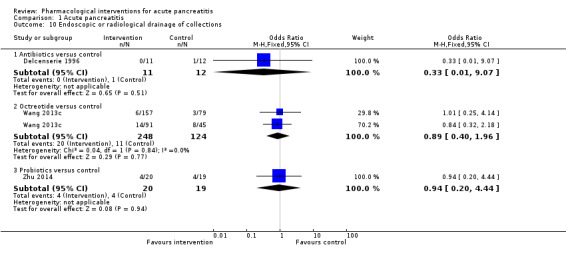
Comparison 1 Acute pancreatitis, Outcome 10 Endoscopic or radiological drainage of collections.
Time to return to normal activity
None of the trials reported this outcome.
Time to work
None of the trials reported this outcome.
Costs
None of the trials reported this outcome.
Subgroup analysis
Because of the paucity of data, we could only analyse a subgroup of acute necrotising pancreatitis and severe acute pancreatitis participants.
Acute necrotising pancreatitis
There was no evidence of difference in any of the outcomes (Analysis 2.1; Analysis 2.2; Analysis 2.3; Analysis 2.4; Analysis 2.5; Analysis 2.6).
2.1. Analysis.
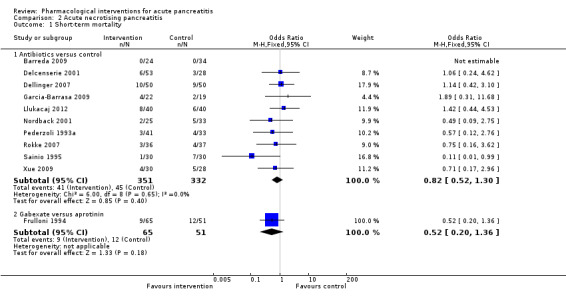
Comparison 2 Acute necrotising pancreatitis, Outcome 1 Short‐term mortality.
2.2. Analysis.
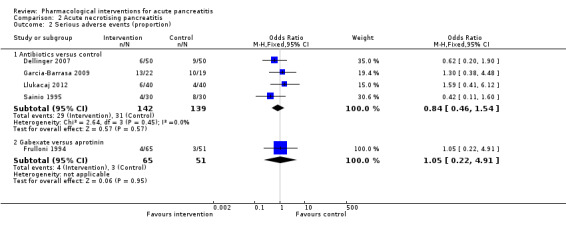
Comparison 2 Acute necrotising pancreatitis, Outcome 2 Serious adverse events (proportion).
2.3. Analysis.
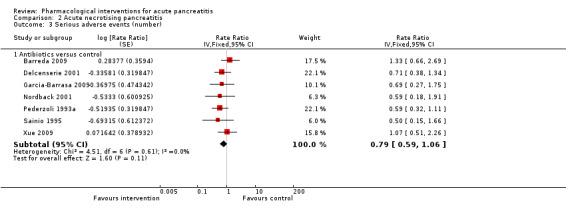
Comparison 2 Acute necrotising pancreatitis, Outcome 3 Serious adverse events (number).
2.4. Analysis.
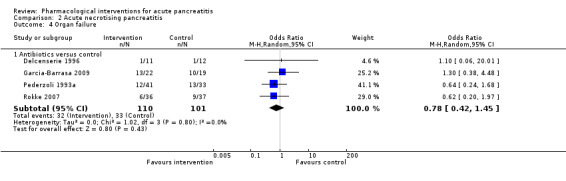
Comparison 2 Acute necrotising pancreatitis, Outcome 4 Organ failure.
2.5. Analysis.
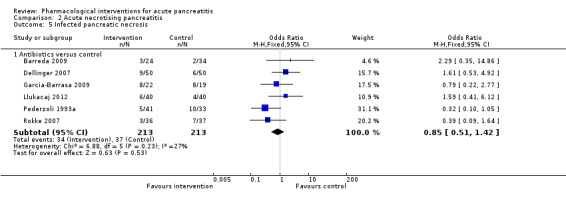
Comparison 2 Acute necrotising pancreatitis, Outcome 5 Infected pancreatic necrosis.
2.6. Analysis.
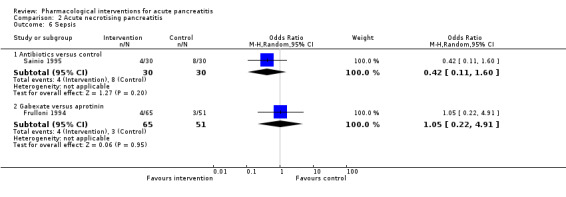
Comparison 2 Acute necrotising pancreatitis, Outcome 6 Sepsis.
Severe acute pancreatitis
Short‐term mortality was lower in the gabexate group versus control (OR 0.19, 95% CI 0.04 to 0.99; participants = 52; studies = 1) (Analysis 3.1)
3.1. Analysis.
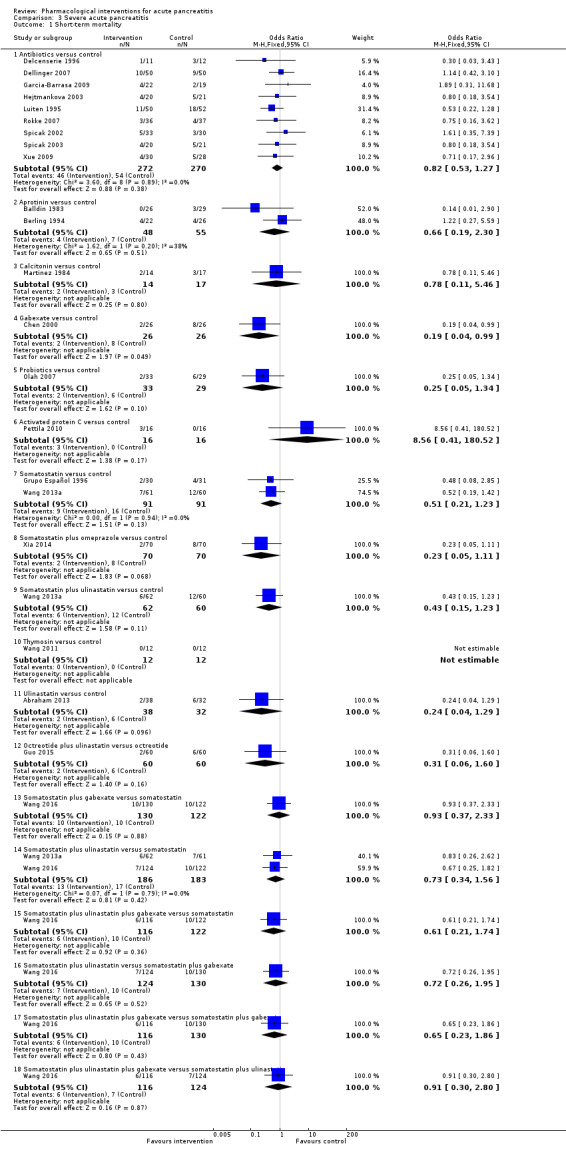
Comparison 3 Severe acute pancreatitis, Outcome 1 Short‐term mortality.
There was no evidence of difference in the proportion of participants experiencing serious adverse events in any of the comparisons (Analysis 3.2). The number of serious adverse events was lower in the somatostatin plus omeprazole group (rate ratio 0.36, 95% CI 0.19 to 0.70; participants = 140; studies = 1) and the somatostatin plus ulinastatin group (rate ratio 0.30, 95% CI 0.15 to 0.60; participants = 122; studies = 1) compared to control. There were also fewer serious adverse events in the somatostatin plus ulinastatin group versus somatostatin alone (rate ratio 0.28, 95% CI 0.15 to 0.56; participants = 123; studies = 1). There was no evidence of differences in other comparisons (Analysis 3.3). Organ failure was lower in the ulinastatin group than in control (OR 0.05, 95% CI 0.01 to 0.21; participants = 67; studies = 1). There was no evidence of differences between other comparisons (Analysis 3.4). There was no evidence of differences in infected pancreatic necrosis or sepsis in any of the comparisons (Analysis 3.5; Analysis 3.6).
3.2. Analysis.
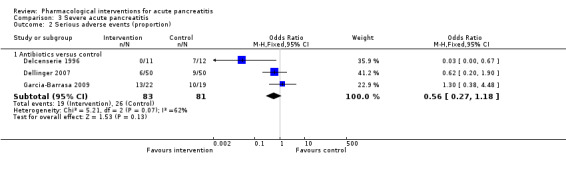
Comparison 3 Severe acute pancreatitis, Outcome 2 Serious adverse events (proportion).
3.3. Analysis.
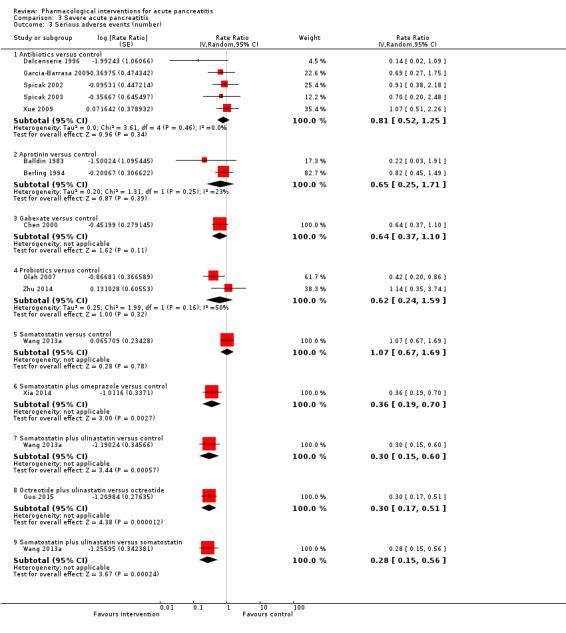
Comparison 3 Severe acute pancreatitis, Outcome 3 Serious adverse events (number).
3.4. Analysis.
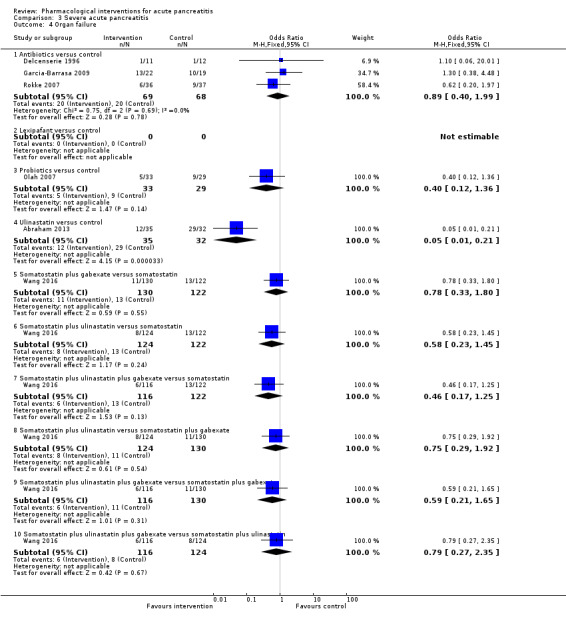
Comparison 3 Severe acute pancreatitis, Outcome 4 Organ failure.
3.5. Analysis.
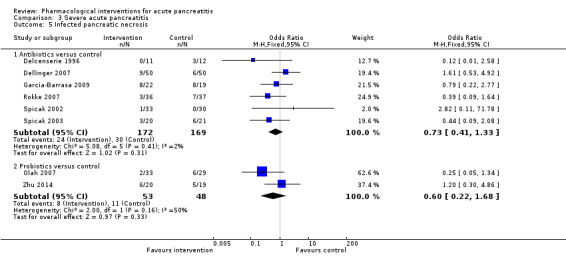
Comparison 3 Severe acute pancreatitis, Outcome 5 Infected pancreatic necrosis.
3.6. Analysis.
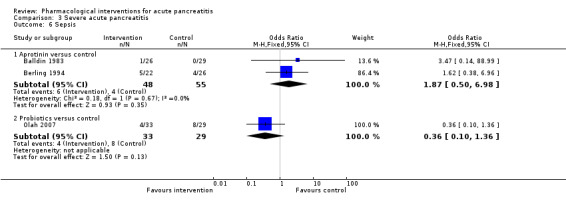
Comparison 3 Severe acute pancreatitis, Outcome 6 Sepsis.
Readers should keep in mind that all the comparisons in which there was evidence of difference are based on single trials at high risk of bias and with small sample size (i.e. random errors).
Sensitivity analysis
All the trials except one were at unclear or high risk of bias in one or more domains (Wang 2016). Since most trials reported median rather than mean for length of hospital stay and length of ICU stay, we did not perform a meta‐analysis by imputing mean and standard deviation. So, we did not perform a sensitivity analysis excluding trials in which either the mean or the standard deviation or both were imputed. We did not perform a sensitivity analysis imputing missing data based on different scenarios since the details of the postrandomisation dropouts were not available from the different trials in which there were postrandomisation dropouts.
Quality of evidence
Most of the comparisons in all the outcomes had low or very low quality evidence because of the risk of bias in the trials (downgraded by one level), imprecision (small sample size (downgraded by one level), and/or overlap of confidence intervals with clinically insignificant effect or no effect (downgraded by one level). There was evidence of heterogeneity in some of the outcomes, which resulted in further downgrading by one level for some comparisons.
Reporting bias
We evaluated the reporting bias for short‐term mortality, serious adverse events (number), infected pancreatic necrosis, adverse events (number), and the requirement for additional intervention for antibiotics versus control, the only comparisons with at least 10 trials. There was no evidence of reporting bias either on visual inspection or by Egger's test for the short‐term mortality, infected pancreatic necrosis, and requirement for additional intervention (Figure 4, P = 0.88; Figure 5, P = 0.74; and Figure 6, P = 0.98, respectively). There was evidence of reporting bias both on visual inspection and by Egger's test for number of serious adverse events (Figure 7; P = 0.021). There was evidence of reporting bias on visual inspection but not by Egger's test for number of adverse events (Figure 8; P = 0.079).
4.
Funnel plot of short‐term mortality indicating no evidence of reporting bias.
5.
Funnel plot of infected pancreatic necrosis indicating no evidence of reporting bias.
6.
Funnel plot of requirement for additional invasive intervention indicating no evidence of reporting bias.
7.
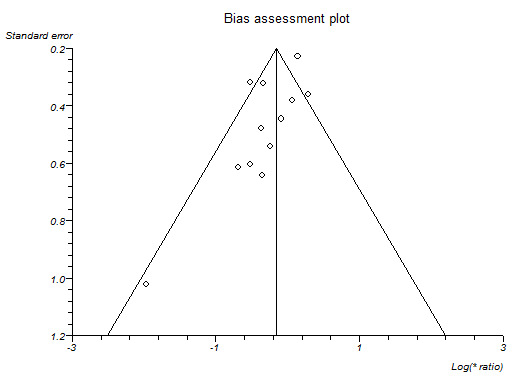
Funnel plot of serious adverse events (number) indicating that trials with lower precision favoured antibiotics without matching trials with lower precision which showed no effect or favouring control.
8.
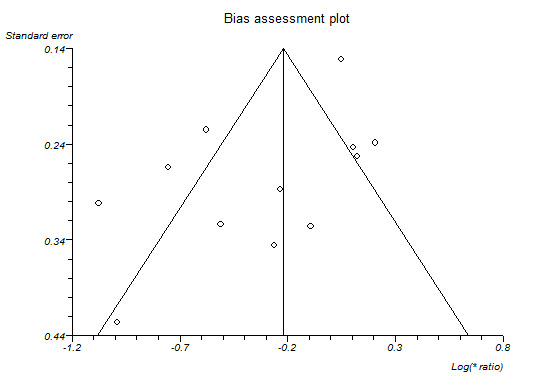
Funnel plot of adverse events (number) indicating that trials with lower precision favoured antibiotics while trials with greater precision favoured control.
Discussion
Summary of main results
A total of 7366 participants in 78 trials contributed to one or more outcomes for this review. The treatments assessed in these 78 trials included antibiotics, antioxidants, aprotinin, atropine, calcitonin, cimetidine, EDTA, gabexate, glucagon, iniprol, lexipafant, NSAID, octreotide, oxyphenonium, probiotics, activated protein C, somatostatin, somatostatin plus omeprazole, somatostatin plus ulinastatin, thymosin, ulinastatin, and inactive control.
Despite the number of trials included, network meta‐analysis was not performed because of major concerns about the transitivity assumption, that is, whether all participants in the network were sufficiently similar and therefore had an equal chance of receiving any of the treatments in the network . In particular, we highlight the fact that a total of 18 trials were included in the comparison under antibiotics versus inactive control (Delcenserie 1996; Delcenserie 2001; Dellinger 2007; Finch 1976; Garcia‐Barrasa 2009; Hejtmankova 2003; Isenmann 2004; Llukacaj 2012; Luiten 1995; Nordback 2001; Pederzoli 1993a; Poropat 2015; Rokke 2007; Sainio 1995; Spicak 2002; Spicak 2003; Xue 2009). Ten of these trials included only participants with acute necrotising pancreatitis (Barreda 2009; Delcenserie 2001; Dellinger 2007; Garcia‐Barrasa 2009; Llukacaj 2012Nordback 2001; Pederzoli 1993a; Rokke 2007; Sainio 1995; Xue 2009). Just two other trials that included only participants with acute necrotising pancreatitis were featured in all the other comparisons put together (Chen 2002b; Frulloni 1994). Thus, there is some clinical heterogeneity in the type of participants that were included in the different comparisons. As a result, we performed direct comparison only.
There was no evidence of difference in short‐term mortality between the groups in any of the comparisons. However, the confidence intervals were wide and consistent with significant benefits or harms of interventions. Because of the number of outcomes reported in the different trials, it is reasonable to expect that the beneficial effect is consistent across clinical outcomes. Interventions with at least two clinical benefits were: lexipafant, which was associated with fewer adverse events (and severe adverse events) and a lower proportion of people with sepsis; octreotide, which was associated with fewer serious adverse events and a lower proportion of people with organ failure; and gabexate, which was associated with fewer adverse events and a lower proportion of people requiring an additional invasive intervention compared to inactive intervention. However, because of the number of analyses performed ('Potential biases in the review process'), concerns about the availability of the drug ('Overall completeness and applicability of evidence'), and the quality of evidence ('Quality of the evidence'), further trials are required before recommending any of the interventions routinely.
Only one trial reported mortality beyond six months (Gilsanz 1978). The follow‐up in the remaining trials was three months in six trials (Besselink 2008; Buchler 1993; Chen 2000; Frulloni 1994; Goebell 1988; Pederzoli 1993b), while in the rest it was less than six weeks. A three‐month follow‐up would identify all the complications related to acute pancreatitis and most deaths related to these complications. However, a period less than three months is likely to miss a considerable proportion. None of the trials reported health‐related quality of life, costs, or other important socioeconomic measures such as return to work. Health‐related quality of life continues to improve between three months and one year after necrotising pancreatitis, although some impairment in quality of life may remain beyond then (Wright 2009). The quality of life after acute severe pancreatitis also appears to be impaired even several years after the acute pancreatitis episode (Hochman 2006; Pendharkar 2014). Future trials on acute pancreatitis should assess the health‐related quality of life for at least 3 months to 12 months and report socioeconomic measures so that it is possible to understand whether the treatments are cost‐effective.
We can only speculate on why no intervention showed any consistent benefit. One possible reason is that the trials were not powered to measure differences in short‐term mortality. The short‐term mortality in the inactive control group was 12% overall and 17.4% (102/586) in the subgroup of acute severe pancreatitis. To measure a 20% relative risk reduction in short‐term mortality using an alpha error of 5% and a beta error of 20%, 3422 participants are required. Clearly, the trials included only a small proportion of the required sample size, so the lack of evidence of difference may be due to random error. The complications related to mild pancreatitis are very infrequent, which means that an even greater sample size than 3422 is required to demonstrate a difference in clinical benefits. On the other hand, if the interventions are targeted against patients with severe pancreatitis, then it can take several hours or even days for the full picture of severe acute pancreatitis to develop. By this time, the damage may be too much for any treatment (other than supportive treatment including organ support) to make a difference. Several prognostic indexes exist for predicting whether the pancreatitis is mild or severe before the clinical picture fully emerges. However, these indexes have a modest sensitivity and specificity in predicting severe acute pancreatitis (Gao 2015a), so it may be reasonable to administer the treatment in all patients with acute pancreatitis and accept that only a proportion will benefit. The proportion of patients with severe pancreatitis in trials that included both mild and severe acute pancreatitis in this review ranged between 17% and 87% (median 35%). The sample size of the trial may have to be estimated on the basis that only the subgroup of severe acute pancreatitis will benefit. It is unlikely that trials powered to measure differences in mortality can be conducted in patients with acute pancreatitis. Using outcomes such as health‐related quality of life or clinically significant complications may allow clinically meaningful trials to be conducted in this population.
Overall completeness and applicability of evidence
This review included all pharmacological interventions without restriction by the year of publication of the trials or whether the drugs are currently licensed. The European Agency for the Evaluation of Medicinal Products (EMEA) had refused marketing authorisation for lexipafant in 1998 after reviewing the data submitted by the company (WHO 2001). Some of the reasons for this refusal included concerns about not having a functional independent data monitoring committee to monitor the results and allegations of financial misconduct by the company that manufactured lexipafant (Hampton 2000; Masood 1998).
Apart from the trials comparing antibiotics versus control, most of the remaining trials did not clearly state whether they included participants with necrotising pancreatitis. So, it is not clear whether this evidence is applicable to patients with acute necrotising pancreatitis. Most trials included a totality or at least a significant proportion of participants with severe acute pancreatitis, so the results of the review are applicable to patients with severe acute pancreatitis in addition to those with mild acute pancreatitis.
This review is only about pharmacological interventions for acute pancreatitis. We have not included any nutritional interventions or interventions on fluid management in this review. We are unable to comment on whether any of the above are effective in the treatment of acute pancreatitis based on the results of this review. We have only reviewed treatment of acute pancreatitis and not prophylaxis. Thus, our review is applicable only in people with acute pancreatitis.
Quality of the evidence
We assessed the quality of the evidence formally only for short‐term mortality, probably the most important outcome for patients with acute pancreatitis. This was low for most of the comparisons. The reason for this is that the risk of bias was unclear or high and because the results were imprecise. Overall, there was not much heterogeneity within each comparison or across comparisons as demonstrated by the I² and Chi² values within comparisons. There was no evidence of publication bias in the one comparison we could assess for short‐term mortality (antibiotics versus control). However, there was evidence of publication bias in serious adverse events (number).There was no indirectness in the short‐term mortality because of the nature of the outcome.
Although we did not undertake a formal assessment of the quality of evidence for the remaining outcomes, the quality of evidence is similarly low because of the issues discussed above, or possibly even lower (i.e. very low) because of having a smaller overall sample size. In addition, there appeared to be reporting bias for the number of both serious adverse events and all adverse events for the comparison antibiotics versus control, although Egger's test was statistically significant only for the number of serious adverse events.
Potential biases in the review process
We followed the Cochrane Handbook for Systematic Reviews of Interventions for the conduct of the direct comparison of the review. Two review authors selected studies and extracted data, reducing the errors in data collection. We used formal search strategies to identify the trials. While the likelihood of missing trials from the identified references was low, the review included the time frame before the mandatory trial registration era, and it was possible that some trials were not reported in journals because of their results. However, one has to be pragmatic and accept that this is the best level of evidence that is currently available.
Network meta‐analysis has its advantages in combining direct and indirect evidence (resulting in more precise evidence); however, when providing effect estimates in the absence of direct comparison and calculating the probability that an intervention is the best treatment, one has to be wary about the transitivity assumption (i.e. whether similar participants were included in the trials across all the comparisons and thus had an equal chance of being randomised to each treatment). As mentioned above, there is some clinical heterogeneity in the type of participants who were included in 'antibiotics versus control' (a high proportion of trials included only participants with acute necrotising pancreatitis) compared to other comparisons (only a very low proportion of trials included only participants with acute necrotising pancreatitis). In the presence of such heterogeneity, it is not appropriate to conduct a network meta‐analysis. In addition to the differences in the presence or absence of necrotising pancreatitis, the type of participants included in the trials were also different in terms of the severity of pancreatitis. We are not able to assess this fully since the definitions used in the trials were not the current definition of severe acute pancreatitis. So, there is likely to be heterogeneity in the type of participants included in the trials. In addition to the clinical heterogeneity in the type of participants included, there were variations in the treatments used in the trials; the definitions used for the different outcomes were not clear or were different in different trials. We did not find any systematic differences in the definitions used for specific comparisons; nevertheless, the lack of uniform definitions used in the trials along with other heterogeneity mentioned above is another potential bias in this review.
We included a number of outcomes to assess effectiveness. Although the outcomes are clinically significant, the outcomes reported in different trials were different. While we found evidence of reporting bias only in a few outcomes where it was possible to formally assess the reporting bias by funnel plots, there is a significant possibility that the outcomes reported in the trials were based on the results of the outcome. Examining a lot of outcomes can also lead to false positives because of multiplicity issues. However, we have decreased the impact of this by focusing on the most important outcome in acute pancreatitis, that is, mortality.
We were not able to obtain full texts for two references (Hansen 1966; Perez 1980). From the title, it appears that Perez 1980 was an abstract of an included trial (Perezdeoteyza 1980). The second reference was published 50 years ago and may or may not be a randomised controlled trial (Hansen 1966), but even if it were, it is unlikely to alter our conclusions.
Agreements and disagreements with other studies or reviews
This is the first attempted network meta‐analysis on this topic. We agree with Villatoro 2010 and Jiang 2012 in that there is no evidence that antibiotics decrease mortality or infected pancreatic necrosis in patients with acute pancreatitis.
Of the systematic reviews on other interventions, we agree with Xu 2013 that octreotide does not appear to be beneficial in major clinical outcomes related to acute pancreatitis and with Messori 1995 that gabexate might decrease the complications without affecting mortality. We disagree with Andriulli 1998 that somatostatin and octreotide decrease mortality. The differences in conclusions between Andriulli 1998 and this review may be due to the inclusion of non‐randomised studies and the publication of new trials subsequent to the conduct of the systematic review.
Authors' conclusions
Implications for practice.
Very low‐quality evidence suggests that no pharmacological treatment leads to a decrease in short‐term mortality in people with acute pancreatitis. However, the confidence intervals were wide and consistent with an increase or decrease in short‐term mortality. We did not find consistent clinical benefits with any intervention.
Implications for research.
Because of the limitations in the prognostic scoring systems and because damage to organs may occur in acute pancreatitis before they are clinically manifest, future trials should consider including pancreatitis of all severity but power the study to measure the differences in the subgroup of people with severe acute pancreatitis. It may be difficult to power the studies based on mortality. Future trials in patients with acute pancreatitis should consider other outcomes such as complications or health‐related quality of life as primary outcomes. Such trials should include health‐related quality of life, costs, and return to work as outcomes and should follow patients for at least three months (preferably for at least one year).
Acknowledgements
We thank Karin Dearness, Managing Editor, Cochrane Upper Gastrointestinal and Pancreatic Diseases (UGPD) Group for providing administrative and logistical support for the conduct of the current review, and Racquel Simpson, Trials Search Co‐ordinator, Cochrane Upper Gastrointestinal and Pancreatic Diseases (UGPD) Group for developing and executing the search strategies.
We thank the copy editors and Cochrane Editorial Unit for their comments.
Appendices
Appendix 1. Glossary of terms
Acute: sudden.
Analogues: a substance that is similar to another substance.
Antioxidants: substances that inhibit oxidation.
Autodigestion: Breakdown of the same organ that secretes the substance.
Bacterial colonisation: growth and multiplication of bacteria.
Cholangiopancreatography: fully known as endoscopic retrograde cholangiopancreatography (ERCP); a procedure carried out on the pancreatic and bile ducts using an endoscope and x‐rays.
Colonisation: presence of bacteria without causing illness (in this context).
Endoscopic sphincterotomy: endoscopic operation to cut the muscle surrounding the common bile duct and the pancreatic duct.
Endoscopic: with the help of an endoscope, a tube inserted into body (in this context, through the mouth and into the stomach and upper part of the small intestine).
Enzyme: substances that enable and speed up chemical reactions that are necessary for the normal functioning of the body.
Epigastric: upper central abdomen.
Epigastric pain: upper central abdominal pain.
Heterogeneity: variability.
Insulin: substance which helps regulate blood sugar.
Interstitial: space in between.
Morbidity: illness (in this context, it means complications).
Mortality: death.
Necrosectomy: removal of dead tissue.
Necrosis: death and decomposition of living tissue usually caused by lack of blood supply but can be caused by other pathological insult.
Necrotising : causing necrosis.
Oedematous: excessive accumulation of serous fluid in the intercellular spaces of tissues.
Pancreatic pseudocysts: fluid collections in the pancreas or the tissues surrounding the pancreas, surrounded by a well defined wall and contain only fluid with little or no solid material.
Pancreatitis: inflammation of the pancreas.
Pathologic insult: substance or mechanism that causes the condition.
Percutaneous: through the skin.
Peripancreatic tissues: tissues surrounding the pancreas.
Pharmacological: medicinal drugs.
Platelet activating factor: substance that causes platelets (cells responsible for clotting of blood) to clump together and is an intermediary substance in the inflammatory pathway.
Probiotics: microorganisms that are believed to provide health benefits when consumed.
Prognostic: to predict the likely outcome.
Protease inhibitors: substances that inhibit proteases.
Protease: an enzyme that digests protein.
Pseudocyst: a fluid‐filled cavity that resembles a cyst but lacks a wall or lining.
Radiology guided percutaneous treatments: treatments carried out by insertion of needle from the external surface of the body which are guided by a scan (usually an ultrasound or CT (computed tomography) scan).
Randomisation: using chance methods to assign people to treatments.
Retrograde: moving backwards.
Sepsis: life‐threatening illness due to blood infection with bacteria, fungus, or virus.
Serum: clear fluid that separates out when blood clots.
Sphincterotomy: a surgical procedure of the internal anal sphincter muscle.
Transabdominal: through the abdomen.
Transient: temporary.
Tumour necrosis factor‐alpha antibody: antibody to tumour necrosis factor‐alpha, an intermediary substance in the inflammatory pathway.
Appendix 2. CENTRAL search strategy
#1 MeSH descriptor: [Pancreatitis, Acute Necrotizing] this term only
#2 MeSH descriptor: [Pancreatitis] this term only and with qualifier(s): [Etiology ‐ ET]
#3 MeSH descriptor: [Pancreas] this term only and with qualifier(s): [Abnormalities ‐ AB, Pathology ‐ PA, Physiopathology ‐ PP]
#4 (acute near/3 pancrea*)
#5 (necro* near/3 pancrea*)
#6 (inflam* near/3 pancrea*)
#7 ((interstitial or edema* or oedema*) near/2 pancrea*)
#8 #1 or #2 or #3 or #4 or #5 or #6 or #7
Appendix 3. MEDLINE search strategy
1. Pancreatitis, Acute Necrotizing/
2. Pancreatitis/et
3. Pancreas/ab, pa, pp
4. (acute adj3 pancrea*).mp.
5. (necro* adj3 pancrea*).mp.
6. (inflam* adj3 pancrea$).mp.
7. ((interstitial or edema* or oedema*) adj2 pancrea*).mp.
8. 1 or 2 or 3 or 4 or 5 or 6 or 7
9. randomized controlled trial.pt.
10. controlled clinical trial.pt.
11. randomized.ab.
12. placebo.ab.
13. drug therapy.fs.
14. randomly.ab.
15. trial.ab.
16. groups.ab.
17. 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16
18. exp animals/ not humans.sh.
19. 17 not 18
20. 8 and 19
Appendix 4. Embase search strategy
1. acute hemorrhagic pancreatitis/
2. Pancreatitis/et
3. acute pancreatitis/
4. (acute adj3 pancrea*).mp.
5. (necro* adj3 pancrea*).mp.
6. (inflam* adj3 pancrea*).mp.
7. ((interstitial or edema* or oedema*) adj2 pancrea*).mp.
8. 1 or 2 or 3 or 4 or 5 or 6 or 7
9. Clinical trial/
10. Randomized controlled trial/
11. Randomization/
12. Single‐Blind Method/
13. Double‐Blind Method/
14. Cross‐Over Studies/
15. Random Allocation/
16. Placebo/
17. Randomi?ed controlled trial*.tw.
18. Rct.tw.
19. Random allocation.tw.
20. Randomly allocated.tw.
21. Allocated randomly.tw.
22. (allocated adj2 random).tw.
23. Single blind*.tw.
24. Double blind*.tw.
25. ((treble or triple) adj blind*).tw.
26. Placebo*.tw.
27. Prospective study/
28. or/9‐27
29. Case study/
30. Case report.tw.
31. Abstract report/ or letter/
32. or/29‐31
33. 28 not 32
34. 8 and 33
Appendix 5. Science Citation Index search strategy
# 1 TS=((acute or necro* or inflam* or interstitial or edema* or oedema*) near/3 pancrea*)
# 2 TS=(random* OR rct* OR crossover OR masked OR blind* OR placebo* OR meta‐analysis OR systematic review* OR meta‐analys*)
# 3 #2 AND #1
Appendix 6. ClinicalTrials.gov search strategy
"Interventional" [STUDY‐TYPES] AND acute pancreatitis [DISEASE] AND ( "Phase 2" OR "Phase 3" OR "Phase 4" ) [PHASE]
Appendix 7. Planned methods
We planned to conduct network meta‐analyses to compare multiple interventions simultaneously for each of the primary and secondary outcomes when there was direct and indirect evidence for at least one comparison. Network meta‐analysis combines direct evidence within trials and indirect evidence across trials (Mills 2012).
We planned to obtain a network plot (Figure 31) to ensure that the trials were connected by treatments using Stata/IC 11 (StataCorp LP) (see Appendix 9 for the Stata commands used). We planned to apply network meta‐analysis to each connected network. We planned to conduct a Bayesian network meta‐analysis using the Markov chain Monte Carlo method in WinBUGS 1.4. We planned to model the treatment contrast (e.g. log OR for binary outcomes, MD or SMD for continuous outcomes, rate ratio for count outcomes, HR for time‐to‐event outcomes) for any two interventions ('functional parameters') as a function of comparisons between each individual intervention and an arbitrarily selected reference group ('basic parameters') (Lu 2004). We planned to use inactive control (combination of placebo and no‐intervention) as the reference group. We planned to perform the network analysis as per the guidance from the NICE DSU documents (Dias 2013). We planned to perform the network meta‐analysis using arm level data. Further details of the codes we planned to use and the technical details of how we planned to perform the analysis are shown in Appendix 10 and Appendix 11. In short, we planned to use three chains and a burn in of 10,000 simulations to ensure convergence, and to obtain the posterior estimates after a further 20,000 simulations. We planned to run the fixed‐effect and random‐effects models (assuming homogeneous between‐trial variance across comparisons) for each outcome. We planned to choose the fixed‐effect model if it resulted in an equivalent or better fit (assessed by residual deviances, number of effective parameters, and deviance information criterion (DIC)) than the random‐effects model. A lower DIC indicates a better model fit. We planned to use the random‐effects model if it resulted in a better model fit as indicated by a DIC lower than that of the fixed‐effect model by at least three. In addition, we planned to perform a random‐effects inconsistency model suggested by NICE DSU (Dias 2012b). We planned to consider the inconsistency model to be better than the random‐effects consistency model (standard random‐effects network meta‐analysis model) if the model fit of the inconsistency model (as indicated by DIC) was at least three lower than the random‐effects consistency model.
9.
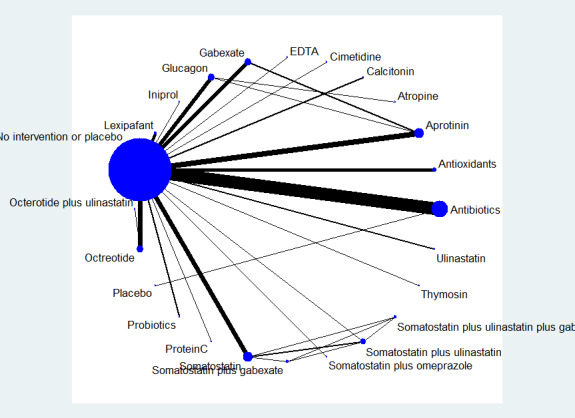
Network plot showing the treatment comparisons that included short‐term mortality. The circles represent treatments while the lines represent the comparisons between the treatments.
For multi‐arm trials, one can enter the data from all the arms in a trial as: the number of people with events and the number of people exposed to the event, using the binomial likelihood and logit link for binary outcomes; the mean and standard error using the normal likelihood and identity link for continuous outcomes requiring calculation of the mean difference; the mean and standard error of the treatment differences using the normal likelihood and identity link for continuous outcomes requiring calculation of the standardised mean difference; the number of events and the number of people exposed to the event using the Poisson likelihood and log link for count outcomes; the follow‐up time in the study, number of people with the event and the number of people exposed to the event using the binomial likelihood and cloglog link for time‐to‐event outcomes. We planned to report the treatment contrasts (e.g. log ORs for binary outcomes, MDs for continuous outcomes, and so on) of the different treatments in relation to the reference treatment (inactive intervention i.e. combined placebo and no‐intervention), the residual deviances, number of effective parameters, and DIC for the fixed‐effect model and the random‐effects model for each outcome. We also planned to report the parameters used to assess the model fit (i.e. residual deviances, number of effective parameters, and DIC) for the inconsistency model for all the outcomes and the between‐trial variance for the random‐effects model (Dias 2012a; Dias 2012b). If the inconsistency model resulted in a better model fit than consistency models, the transitivity assumption is likely to be untrue and the effect estimates obtained may not be reliable. We planned to highlight such outcomes where the inconsistency model results in a better model fit than consistency models.
We found significant clinical heterogeneity in the type of participants included under the different comparisons. To overcome the heterogeneity in the type of people included in different comparisons (See 'Included studies') we planned to perform a separate network meta‐analysis for interventions for mild pancreatitis separately from moderately severe or severe pancreatitis. This is because mild pancreatitis has no local or systemic complications and combining participants with mild and severe acute pancreatitis in the same network meta‐analysis may violate the transitivity assumption (the assumption that the participants included in the different studies with different treatments can be considered to be a part of a multi‐arm randomised controlled trial ‐ i.e. they should be reasonably similar in characteristics). We then planned to assess inconsistency again. However, this was not appropriate in the subgroup of severe acute pancreatitis because of the absence of any comparison in which direct and indirect comparison was available. If there was no evidence of inconsistency in the revised analysis, we planned to present the results of the analysis for mild and moderate or severe acute pancreatitis separately. If there was persistent evidence of inconsistency, we planned to present the results from the direct comparison in the 'Summary of findings' table.
We planned to calculate the 95% CrIs of treatment effects (e.g. ORs for binary outcomes, MDs for continuous outcomes, and so on) in the Bayesian meta‐analysis, which is similar in use to the 95% confidence intervals in the frequentist meta‐analysis. These are the 2.5th percentile and 97.5th percentiles of the simulations. We planned to report the mean effect estimate and the 95% CrI for each pair‐wise comparison in a table. We also planned to estimate the probability that each intervention ranks at one of the possible positions, and have presented this information in graphs. It should be noted that a less than 90% probability that the treatment is the best treatment is unreliable (i.e. one should not conclude that the treatment is the best treatment for that outcome if the probability of it being the best treatment is less than 90%) (Dias 2012a). We also planned to present the cumulative probability of the treatment ranks (i.e. the probability that the treatment is within the top two, the probability that the treatment is within the top three, etc.) in graphs. We also planned to plot the probability that each treatment is best for each of the different outcomes (rankograms) which are generally considered more informative (Dias 2012a; Salanti 2011). We planned to perform direct comparisons using the same codes. This would have allowed us to assess the heterogeneity in the comparisons and provide additional information in the 'Summary of findings' table. We also planned to use the Tau² statistic to measure heterogeneity among the trials in each analysis. The Tau² statistic provides a measure of the variability of the effect estimate across studies in a random‐effects model (Higgins 2011). If we identified substantial heterogeneity, we planned to explore it by meta‐regression. We also planned to assess the differences in the effect estimates between the subgroups using meta‐regression for each source of heterogeneity (i.e. one analysis for each source of heterogeneity) with the help of the code shown in Appendix 12. We planned to perform the following subgroup analyses regardless of heterogeneity. We planned to calculate the interaction term (Dias 2012c). If the 95% CrI of the regression coefficient of the interaction term does not overlap zero, we considered this statistically significant.
In the presence of adequate data where authors report the outcomes of participants at multiple follow‐up time points, we planned to follow the methods suggested by Lu 2007 to perform the meta‐analysis.
We planned to use methods and recommendations described for grading network meta‐analysis (Puhan 2014). This includes grading the quality for direct comparison, indirect comparison, and network meta‐analysis and presenting the information in tabular format.
Appendix 8. WHO ICTRP search strategy
Acute pancreatitis
Appendix 9. Stata code for network plot
networkplot t1 t2, labels(T1 T2 T3 ..)
Appendix 10. Winbugs code
Binary outcome
Binary outcome ‐ fixed‐effect model
# Binomial likelihood, logit link # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood # model for linear predictor logit(p[i,k]) <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] # expected value of the numerators rhat[i,k] <‐ p[i,k] * n[i,k] #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Binary outcome ‐ random‐effects model
# Binomial likelihood, logit link # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood logit(p[i,k]) <‐ mu[i] + delta[i,k] # model for linear predictor rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Binary outcome ‐ inconsistency model (random‐effects)
# Binomial likelihood, logit link, inconsistency model # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH trials delta[i,1]<‐0 # treatment effect is zero in control arm mu[i] ˜ dnorm(0,.0001) # vague priors for trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood logit(p[i,k]) <‐ mu[i] + delta[i,k] # model for linear predictor #Deviance contribution rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(d[t[i,1],t[i,k]] ,tau) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance for (c in 1:(nt‐1)) { # priors for all mean treatment effects for (k in (c+1):nt) { d[c,k] ˜ dnorm(0,.0001) } } sd ˜ dunif(0,5) # vague prior for between‐trial standard deviation var <‐ pow(sd,2) # between‐trial variance tau <‐ 1/var # between‐trial precision } # *** PROGRAM ENDS
Continuous outcome (mean difference)
Continuous outcome (mean difference) ‐ fixed‐effect model
# Normal likelihood, identity link # Fixed effect model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions y[i,k] ˜ dnorm(theta[i,k],prec[i,k]) # model for linear predictor theta[i,k] <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] #Deviance contribution dev[i,k] <‐ (y[i,k]‐theta[i,k])*(y[i,k]‐theta[i,k])*prec[i,k] } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for control arm # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # ranking on relative scale for (k in 1:nt) { rk[k] <‐ rank(d[],k) # assumes lower is better # rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (mean difference) ‐ random‐effects model
# Normal likelihood, identity link # Random effects model for multi‐arm trials model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions y[i,k] ˜ dnorm(theta[i,k],prec[i,k]) theta[i,k] <‐ mu[i] + delta[i,k] # model for linear predictor #Deviance contribution dev[i,k] <‐ (y[i,k]‐theta[i,k])*(y[i,k]‐theta[i,k])*prec[i,k] } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific MD distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of MD distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of MD distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for control arm # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # ranking on relative scale for (k in 1:nt) { rk[k] <‐ rank(d[],k) # assumes lower is better # rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (standardised mean difference)
The standardised mean difference and its standard error for each treatment comparison will be calculated using the statistical algorithms used by RevMan.
Continuous outcome (standardised mean difference) ‐ fixed‐effect model
# Normal likelihood, identity link # Trial‐level data given as treatment differences # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns2) { # LOOP THROUGH 2‐ARM STUDIES y[i,2] ˜ dnorm(delta[i,2],prec[i,2]) # normal likelihood for 2‐arm trials #Deviance contribution for trial i resdev[i] <‐ (y[i,2]‐delta[i,2])*(y[i,2]‐delta[i,2])*prec[i,2] } for(i in (ns2+1):(ns2+ns3)) { # LOOP THROUGH THREE‐ARM STUDIES for (k in 1:(na[i]‐1)) { # set variance‐covariance matrix for (j in 1:(na[i]‐1)) { Sigma[i,j,k] <‐ V[i]*(1‐equals(j,k)) + var[i,k+1]*equals(j,k) } } Omega[i,1:(na[i]‐1),1:(na[i]‐1)] <‐ inverse(Sigma[i,,]) #Precision matrix # multivariate normal likelihood for 3‐arm trials y[i,2:na[i]] ˜ dmnorm(delta[i,2:na[i]],Omega[i,1:(na[i]‐1),1:(na[i]‐1)]) #Deviance contribution for trial i for (k in 1:(na[i]‐1)){ # multiply vector & matrix ydiff[i,k]<‐ y[i,(k+1)] ‐ delta[i,(k+1)] z[i,k]<‐ inprod2(Omega[i,k,1:(na[i]‐1)], ydiff[i,1:(na[i]‐1)]) } resdev[i]<‐ inprod2(ydiff[i,1:(na[i]‐1)], z[i,1:(na[i]‐1)]) } for(i in 1:(ns2+ns3)){ # LOOP THROUGH ALL STUDIES for (k in 2:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions delta[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # ranking on relative scale for (k in 1:nt) { rk[k] <‐ nt+1‐rank(d[],k) # assumes higher HRQoL is “good” #rk[k] <‐ rank(d[],k) # assumes higher outcome is “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Continuous outcome (standardised mean difference) ‐ random‐effects model
# Normal likelihood, identity link # Trial‐level data given as treatment differences # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns2) { # LOOP THROUGH 2‐ARM STUDIES y[i,2] ˜ dnorm(delta[i,2],prec[i,2]) # normal likelihood for 2‐arm trials #Deviance contribution for trial i resdev[i] <‐ (y[i,2]‐delta[i,2])*(y[i,2]‐delta[i,2])*prec[i,2] } for(i in (ns2+1):(ns2+ns3)) { # LOOP THROUGH THREE‐ARM STUDIES for (k in 1:(na[i]‐1)) { # set variance‐covariance matrix for (j in 1:(na[i]‐1)) { Sigma[i,j,k] <‐ V[i]*(1‐equals(j,k)) + var[i,k+1]*equals(j,k) } } Omega[i,1:(na[i]‐1),1:(na[i]‐1)] <‐ inverse(Sigma[i,,]) #Precision matrix # multivariate normal likelihood for 3‐arm trials y[i,2:na[i]] ˜ dmnorm(delta[i,2:na[i]],Omega[i,1:(na[i]‐1),1:(na[i]‐1)]) #Deviance contribution for trial i for (k in 1:(na[i]‐1)){ # multiply vector & matrix ydiff[i,k]<‐ y[i,(k+1)] ‐ delta[i,(k+1)] z[i,k]<‐ inprod2(Omega[i,k,1:(na[i]‐1)], ydiff[i,1:(na[i]‐1)]) } resdev[i]<‐ inprod2(ydiff[i,1:(na[i]‐1)], z[i,1:(na[i]‐1)]) } for(i in 1:(ns2+ns3)){ # LOOP THROUGH ALL STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm for (k in 2:na[i]) { # LOOP THROUGH ARMS var[i,k] <‐ pow(se[i,k],2) # calculate variances prec[i,k] <‐ 1/var[i,k] # set precisions } for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific SMD distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of random effects distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of random effects distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # ranking on relative scale for (k in 1:nt) { rk[k] <‐ nt+1‐rank(d[],k) # assumes higher HRQoL is “good” # rk[k] <‐ rank(d[],k) # assumes higher outcome is “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Count outcome
Count outcome ‐ fixed‐effect model
# Poisson likelihood, log link # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dpois(theta[i,k]) # Poisson likelihood theta[i,k] <‐ lambda[i,k]*E[i,k] # failure rate * exposure # model for linear predictor log(lambda[i,k]) <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] #Deviance contribution dev[i,k] <‐ 2*((theta[i,k]‐r[i,k]) + r[i,k]*log(r[i,k]/theta[i,k])) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # pairwise RRs and LRRs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { rater[c,k] <‐ exp(d[k] ‐ d[c]) lrater[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Count outcome ‐ random‐effects model
# Poisson likelihood, log link # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dpois(theta[i,k]) # Poisson likelihood theta[i,k] <‐ lambda[i,k]*E[i,k] # failure rate * exposure # model for linear predictor log(lambda[i,k]) <‐ mu[i] + d[t[i,k]] ‐ d[t[i,1]] #Deviance contribution dev[i,k] <‐ 2*((theta[i,k]‐r[i,k]) + r[i,k]*log(r[i,k]/theta[i,k])) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) } } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ nt+1‐rank(d[],k) # assumes events are “good” rk[k] <‐ rank(d[],k) # assumes events are “bad” best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Time‐to‐event outcome
Time‐to‐event outcome ‐ fixed‐effect model
# Binomial likelihood, cloglog link # Fixed effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # Binomial likelihood # model for linear predictor cloglog(p[i,k]) <‐ log(time[i]) + mu[i] + d[t[i,k]] ‐ d[t[i,1]] rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for control arm # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ rank(d[],k) # assumes lower is better rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Time‐to‐event outcome ‐ random‐effects model
# Binomial likelihood, cloglog link # Random effects model model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # Binomial likelihood # model for linear predictor cloglog(p[i,k]) <‐ log(time[i]) + mu[i] + delta[i,k] rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions, with multi‐arm trial correction md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment, multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) #Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment # vague priors for treatment effects for (k in 2:nt){ d[k] ˜ dnorm(0,.0001) } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # ranking on relative scale for (k in 1:nt) { # rk[k] <‐ rank(d[],k) # assumes lower is better rk[k] <‐ nt+1‐rank(d[],k) # assumes lower outcome is worse best[k] <‐ equals(rk[k],1) #calculate probability that treat k is best for (h in 1:nt){ prob[h,k] <‐ equals(rk[k],h) } # calculates probability that treat k is h‐th best } } # *** PROGRAM ENDS
Appendix 11. Technical details of network meta‐analysis
The posterior probabilities (effect estimates or values) of the treatment contrast (i.e. log odds ratio, mean difference, standardised mean difference, rate ratio, or hazard ratio) may vary depending on the initial values to start the simulations. In order to control the random error due to the choice of initial values, we performed the network analysis for three different initial values (priors) as per the guidance from The National Institute for Health and Care Excellence (NICE) Decision Support Unit (DSU) documents (Dias 2013). If the results from three different priors are similar (convergence), then the results are reliable. It is important to discard the results of the initial simulations as they can be significantly affected by the choice of the priors and only include the results of the simulations obtained after the convergence. The discarding of the initial simulations is called 'burn in'. We ran the models for all outcomes for 10,000 simulations for 'burn in' for three different chains (a set of initial values). We ran the models for another 20,000 simulations to obtain the effect estimates. We obtained the effect estimates from the results of all the three chains (different initial values). We also ensured that the results in the three different chains are similar in order to control for random error due to the choice of initial values. This was done in addition to the visual inspection of convergence obtained after simulations in the burn in.
We ran three different models for each outcome. The fixed‐effect model assumes that the treatment effect is the same across studies. The random‐effects consistency model assumes that the treatment effect is distributed normally across the studies but assumes that the transitivity assumption is satisfied (i.e. the population studied, the definition of outcomes, and the methods used were similar across studies and that there is consistency between the direct comparison and indirect comparison). A random‐effects inconsistency model does not make the transitivity assumption. If the inconsistency model resulted in a better model fit than the consistency model, the results of the network meta‐analysis can be unreliable and so should be interpreted with extreme caution. If there is evidence of inconsistency, we planned to identify areas in the network where substantial inconsistency might be present in terms of clinical and methodological diversities between trials and, when appropriate, limit the network meta‐analysis to a more compatible subset of trials.
The choice of the model between fixed‐effect and random‐effects was based on the model fit as per the guidelines of the NICE TSU (Dias 2013). The model fit will be assessed by deviance residuals and Deviance Information Criteria (DIC) according to NICE TSU guidelines (Dias 2013). A difference of three or five in the DIC is not generally considered important (Dias 2012c). We used the simpler model, i.e. fixed‐effect model if the DIC are similar between the fixed‐effect and the random‐effects models. We used the random‐effects model if it results in a better model fit as indicated by a DIC lower than that of the fixed‐effect model by at least three.
We planned to calculate the effect estimates of the treatment and the 95% credible intervals using the following additional code. # pairwise ORs and MD for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { OR[c,k] <‐ exp(d[k] ‐ d[c]) #MD[c,k] <‐ (d[k]‐d[c]) } }
where c indicates control group, k indicates intervention group, OR indicates odds ratio or other ratios, and MD indicates mean difference or other differences.
Appendix 12. Winbugs code for subgroup analysis
Categorical covariate
Only the code for random‐effects model for a binary outcome is shown. The differences in the code are underlined. We planned to make similar changes for other outcomes.
# Binomial likelihood, logit link, subgroup # Random effects model for multi‐arm trials model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood # model for linear predictor, covariate effect relative to treat in arm 1 logit(p[i,k]) <‐ mu[i] + delta[i,k] + (beta[t[i,k]]‐beta[t[i,1]]) * x[i] rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment beta[1] <‐ 0 # covariate effect is zero for reference treatment for (k in 2:nt){ # LOOP THROUGH TREATMENTS d[k] ˜ dnorm(0,.0001) # vague priors for treatment effects beta[k] <‐ B[k] # exchangeable covariate effect B[k] ˜ dnorm(0,.0001) # vague prior for covariate effect } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # treatment effect when covariate = z[j] for (k in 1:nt){ # LOOP THROUGH TREATMENTS for (j in 1:nz) { dz[j,k] <‐ d[k] + (beta[k]‐beta[1])*z[j] } } # *** PROGRAM ENDS
Continuous covariate
# Binomial likelihood, logit link, continuous covariate # Random effects model for multi‐arm trials model{ # *** PROGRAM STARTS for(i in 1:ns){ # LOOP THROUGH STUDIES w[i,1] <‐ 0 # adjustment for multi‐arm trials is zero for control arm delta[i,1] <‐ 0 # treatment effect is zero for control arm mu[i] ˜ dnorm(0,.0001) # vague priors for all trial baselines for (k in 1:na[i]) { # LOOP THROUGH ARMS r[i,k] ˜ dbin(p[i,k],n[i,k]) # binomial likelihood # model for linear predictor, covariate effect relative to treat in arm 1 logit(p[i,k]) <‐ mu[i] + delta[i,k] + (beta[t[i,k]]‐beta[t[i,1]]) * (x[i]‐mx) rhat[i,k] <‐ p[i,k] * n[i,k] # expected value of the numerators #Deviance contribution dev[i,k] <‐ 2 * (r[i,k] * (log(r[i,k])‐log(rhat[i,k])) + (n[i,k]‐r[i,k]) * (log(n[i,k]‐r[i,k]) ‐ log(n[i,k]‐rhat[i,k]))) } # summed residual deviance contribution for this trial resdev[i] <‐ sum(dev[i,1:na[i]]) for (k in 2:na[i]) { # LOOP THROUGH ARMS # trial‐specific LOR distributions delta[i,k] ˜ dnorm(md[i,k],taud[i,k]) # mean of LOR distributions (with multi‐arm trial correction) md[i,k] <‐ d[t[i,k]] ‐ d[t[i,1]] + sw[i,k] # precision of LOR distributions (with multi‐arm trial correction) taud[i,k] <‐ tau *2*(k‐1)/k # adjustment for multi‐arm RCTs w[i,k] <‐ (delta[i,k] ‐ d[t[i,k]] + d[t[i,1]]) # cumulative adjustment for multi‐arm trials sw[i,k] <‐ sum(w[i,1:k‐1])/(k‐1) } } totresdev <‐ sum(resdev[]) # Total Residual Deviance d[1]<‐0 # treatment effect is zero for reference treatment beta[1] <‐ 0 # covariate effect is zero for reference treatment for (k in 2:nt){ # LOOP THROUGH TREATMENTS d[k] ˜ dnorm(0,.0001) # vague priors for treatment effects beta[k] <‐ B[k] # exchangeable covariate effect B[k] ˜ dnorm(0,.0001) # vague prior for covariate effect } sd ˜ dunif(0,5) # vague prior for between‐trial SD tau <‐ pow(sd,‐2) # between‐trial precision = (1/between‐trial variance) # treatment effect when covariate = z[j] (un‐centring treatment effects) for (k in 1:nt){ for (j in 1:nz) { dz[j,k] <‐ d[k] ‐ (beta[k]‐beta[1])*(mx‐z[j]) } } # pairwise ORs and LORs for all possible pair‐wise comparisons, if nt>2 for (c in 1:(nt‐1)) { for (k in (c+1):nt) { # at mean value of covariate or[c,k] <‐ exp(d[k] ‐ d[c]) lor[c,k] <‐ (d[k]‐d[c]) # at covariate=z[j] for (j in 1:nz) { orz[j,c,k] <‐ exp(dz[j,k] ‐ dz[j,c]) lorz[j,c,k] <‐ (dz[j,k]‐dz[j,c]) } } } } # *** PROGRAM ENDS
Data and analyses
Comparison 1. Acute pancreatitis.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term mortality | 67 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Antibiotics versus control | 17 | 1058 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.57, 1.15] |
| 1.2 Antioxidants versus control | 4 | 163 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.01 [0.53, 7.56] |
| 1.3 Aprotinin versus control | 7 | 651 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.40, 1.14] |
| 1.4 Calcitonin versus control | 2 | 125 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.15, 2.00] |
| 1.5 Cimetidine versus control | 1 | 40 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.06, 17.18] |
| 1.6 EDTA versus control | 1 | 64 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.12, 7.08] |
| 1.7 Gabexate versus control | 5 | 576 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.48, 1.30] |
| 1.8 Glucagon versus control | 5 | 409 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.51, 1.87] |
| 1.9 Iniprol versus control | 1 | 24 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.14 [0.01, 1.67] |
| 1.10 Lexipafant versus control | 3 | 423 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.30, 1.01] |
| 1.11 Octreotide versus control | 5 | 927 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.76 [0.47, 1.23] |
| 1.12 Probiotics versus control | 2 | 358 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.70 [0.87, 3.30] |
| 1.13 Activated protein C versus control | 1 | 32 | Odds Ratio (M‐H, Fixed, 95% CI) | 8.56 [0.41, 180.52] |
| 1.14 Somatostatin versus control | 6 | 493 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.57 [0.29, 1.10] |
| 1.15 Somatostatin plus omeprazole versus control | 1 | 140 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.23 [0.05, 1.11] |
| 1.16 Somatostatin plus ulinastatin versus control | 1 | 122 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.43 [0.15, 1.23] |
| 1.17 Thymosin versus control | 1 | 24 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.18 Ulinastatin versus control | 1 | 132 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.45 [0.12, 1.72] |
| 1.19 Gabexate versus aprotinin | 2 | 298 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.32, 1.20] |
| 1.20 Glucagon versus aprotinin | 1 | 134 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.33 [0.44, 4.08] |
| 1.21 Glucagon versus atropine | 1 | 150 | Odds Ratio (M‐H, Fixed, 95% CI) | 4.17 [0.45, 38.21] |
| 1.22 Octreotide plus ulinastatin versus octreotide | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.31 [0.06, 1.60] |
| 1.23 Somatostatin plus gabexate versus somatostatin | 1 | 252 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.37, 2.33] |
| 1.24 Somatostatin plus ulinastatin versus somatostatin | 2 | 369 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.73 [0.34, 1.56] |
| 1.25 Somatostatin plus ulinastatin plus gabexate versus somatostatin | 1 | 238 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.61 [0.21, 1.74] |
| 1.26 Somatostatin plus ulinastatin versus somatostatin plus gabexate | 1 | 254 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.72 [0.26, 1.95] |
| 1.27 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus gabexate | 1 | 246 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.23, 1.86] |
| 1.28 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus ulinastatin | 1 | 240 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.30, 2.80] |
| 2 Serious adverse events (proportion) | 17 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 Antibiotics versus control | 5 | 304 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.37, 1.15] |
| 2.2 Antioxidants versus control | 2 | 82 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.98 [0.48, 8.13] |
| 2.3 EDTA versus control | 1 | 64 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.11, 2.39] |
| 2.4 Gabexate versus control | 2 | 201 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.31 [0.31, 5.60] |
| 2.5 Glucagon versus control | 2 | 127 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.29 [0.01, 7.46] |
| 2.6 Octreotide versus control | 1 | 58 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.73 [0.61, 4.93] |
| 2.7 Somatostatin versus control | 2 | 111 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.07 [0.35, 3.27] |
| 2.8 Gabexate versus aprotinin | 1 | 116 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.22, 4.91] |
| 2.9 Ulinastatin versus gabexate | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Serious adverse events (number) | 37 | Rate Ratio (Fixed, 95% CI) | Subtotals only | |
| 3.1 Antibiotics versus control | 12 | 716 | Rate Ratio (Fixed, 95% CI) | 0.86 [0.68, 1.07] |
| 3.2 Antioxidants versus control | 2 | 71 | Rate Ratio (Fixed, 95% CI) | 0.22 [0.02, 2.21] |
| 3.3 Aprotinin versus control | 3 | 264 | Rate Ratio (Fixed, 95% CI) | 0.79 [0.49, 1.29] |
| 3.4 Cimetidine versus control | 1 | 60 | Rate Ratio (Fixed, 95% CI) | 1.0 [0.20, 4.95] |
| 3.5 EDTA versus control | 1 | 64 | Rate Ratio (Fixed, 95% CI) | 0.94 [0.19, 4.65] |
| 3.6 Gabexate versus control | 3 | 375 | Rate Ratio (Fixed, 95% CI) | 0.86 [0.64, 1.15] |
| 3.7 Glucagon versus control | 1 | 68 | Rate Ratio (Fixed, 95% CI) | 1.0 [0.02, 50.40] |
| 3.8 Lexipafant versus control | 1 | 290 | Rate Ratio (Fixed, 95% CI) | 0.67 [0.46, 0.96] |
| 3.9 Octreotide versus control | 4 | 770 | Rate Ratio (Fixed, 95% CI) | 0.74 [0.60, 0.89] |
| 3.10 Probiotics versus control | 3 | 397 | Rate Ratio (Fixed, 95% CI) | 0.94 [0.65, 1.36] |
| 3.11 Somatostatin versus control | 3 | 257 | Rate Ratio (Fixed, 95% CI) | 1.03 [0.66, 1.59] |
| 3.12 Somatostatin plus omeprazole versus control | 1 | 140 | Rate Ratio (Fixed, 95% CI) | 0.36 [0.19, 0.70] |
| 3.13 Somatostatin plus ulinastatin versus control | 1 | 122 | Rate Ratio (Fixed, 95% CI) | 0.30 [0.15, 0.60] |
| 3.14 Glucagon versus atropine | 1 | 150 | Rate Ratio (Fixed, 95% CI) | 0.33 [0.03, 3.20] |
| 3.15 Octreotide plus ulinastatin versus octreotide | 1 | 120 | Rate Ratio (Fixed, 95% CI) | 0.30 [0.17, 0.51] |
| 3.16 Somatostatin plus ulinastatin versus somatostatin | 1 | 123 | Rate Ratio (Fixed, 95% CI) | 0.28 [0.15, 0.56] |
| 4 Organ failure | 18 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Antibiotics versus control | 5 | 258 | Odds Ratio (M‐H, Random, 95% CI) | 0.78 [0.44, 1.38] |
| 4.2 Antioxidants versus control | 4 | 163 | Odds Ratio (M‐H, Random, 95% CI) | 0.92 [0.39, 2.12] |
| 4.3 Gabexate versus control | 1 | 50 | Odds Ratio (M‐H, Random, 95% CI) | 0.32 [0.01, 8.25] |
| 4.4 Lexipafant versus control | 2 | 340 | Odds Ratio (M‐H, Random, 95% CI) | 0.68 [0.36, 1.27] |
| 4.5 Octreotide versus control | 2 | 430 | Odds Ratio (M‐H, Random, 95% CI) | 0.51 [0.27, 0.97] |
| 4.6 Probiotics versus control | 2 | 358 | Odds Ratio (M‐H, Random, 95% CI) | 0.80 [0.26, 2.47] |
| 4.7 Ulinastatin versus control | 1 | 129 | Odds Ratio (M‐H, Random, 95% CI) | 0.27 [0.01, 6.67] |
| 4.8 Somatostatin plus gabexate versus somatostatin | 1 | 252 | Odds Ratio (M‐H, Random, 95% CI) | 0.78 [0.33, 1.80] |
| 4.9 Somatostatin plus ulinastatin versus somatostatin | 1 | 246 | Odds Ratio (M‐H, Random, 95% CI) | 0.58 [0.23, 1.45] |
| 4.10 Somatostatin plus ulinastatin plus gabexate versus somatostatin | 1 | 238 | Odds Ratio (M‐H, Random, 95% CI) | 0.46 [0.17, 1.25] |
| 4.11 Somatostatin plus ulinastatin versus somatostatin plus gabexate | 1 | 254 | Odds Ratio (M‐H, Random, 95% CI) | 0.75 [0.29, 1.92] |
| 4.12 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus gabexate | 1 | 246 | Odds Ratio (M‐H, Random, 95% CI) | 0.59 [0.21, 1.65] |
| 4.13 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus ulinastatin | 1 | 240 | Odds Ratio (M‐H, Random, 95% CI) | 0.79 [0.27, 2.35] |
| 5 Infected pancreatic necrosis | 15 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 Antibiotics versus control | 11 | 714 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.53, 1.25] |
| 5.2 Octreotide versus control | 1 | 58 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.04, 6.06] |
| 5.3 Probiotics versus control | 3 | 397 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.62, 1.96] |
| 6 Sepsis | 11 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 6.1 Antibiotics versus control | 1 | 60 | Odds Ratio (M‐H, Random, 95% CI) | 0.42 [0.11, 1.60] |
| 6.2 Aprotinin versus control | 2 | 103 | Odds Ratio (M‐H, Random, 95% CI) | 1.84 [0.49, 6.96] |
| 6.3 Gabexate versus control | 3 | 373 | Odds Ratio (M‐H, Random, 95% CI) | 1.10 [0.55, 2.19] |
| 6.4 Lexipafant versus control | 1 | 290 | Odds Ratio (M‐H, Random, 95% CI) | 0.26 [0.08, 0.83] |
| 6.5 Octreotide versus control | 2 | 340 | Odds Ratio (M‐H, Random, 95% CI) | 0.40 [0.05, 3.53] |
| 6.6 Probiotics versus control | 1 | 62 | Odds Ratio (M‐H, Random, 95% CI) | 0.36 [0.10, 1.36] |
| 6.7 Gabexate versus aprotinin | 1 | 116 | Odds Ratio (M‐H, Random, 95% CI) | 1.05 [0.22, 4.91] |
| 7 Adverse events (proportion) | 27 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 7.1 Antibiotics versus control | 6 | 429 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.51 [0.32, 0.80] |
| 7.2 Antioxidants versus control | 1 | 39 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Calcitonin versus control | 1 | 94 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.12, 6.49] |
| 7.4 EDTA versus control | 1 | 64 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.27, 2.31] |
| 7.5 Gabexate versus control | 3 | 373 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.54, 1.27] |
| 7.6 Glucagon versus control | 2 | 127 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.09 [0.00, 1.69] |
| 7.7 Lexipafant versus control | 1 | 83 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.43 [0.16, 1.12] |
| 7.8 Octreotide versus control | 3 | 398 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.65, 1.55] |
| 7.9 Probiotics versus control | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.35 [0.12, 1.01] |
| 7.10 Somatostatin versus control | 2 | 111 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.44 [0.19, 1.02] |
| 7.11 Somatostatin plus omeprazole versus control | 1 | 140 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.00 [0.00, 0.04] |
| 7.12 Gabexate versus aprotinin | 2 | 298 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.41 [0.23, 0.70] |
| 7.13 Ulinastatin versus gabexate | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.14 Ulinastatin versus octreotide | 1 | 25 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.33 [0.46, 11.81] |
| 7.15 Somatostatin plus gabexate versus somatostatin | 1 | 252 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.44, 1.95] |
| 7.16 Somatostatin plus ulinastatin versus somatostatin | 1 | 246 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.25, 1.34] |
| 7.17 Somatostatin plus ulinastatin plus gabexate versus somatostatin | 1 | 238 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.20, 1.20] |
| 7.18 Somatostatin plus ulinastatin versus somatostatin plus gabexate | 1 | 254 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.27, 1.44] |
| 7.19 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus gabexate | 1 | 246 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.53 [0.22, 1.28] |
| 7.20 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus ulinastatin | 1 | 240 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.32, 2.22] |
| 8 Adverse events (number) | 40 | Rate Ratio (Random, 95% CI) | Subtotals only | |
| 8.1 Antibiotics versus control | 12 | 755 | Rate Ratio (Random, 95% CI) | 0.75 [0.58, 0.95] |
| 8.2 Antioxidants versus control | 2 | 94 | Rate Ratio (Random, 95% CI) | 0.82 [0.38, 1.79] |
| 8.3 Aprotinin versus control | 3 | 264 | Rate Ratio (Random, 95% CI) | 0.98 [0.69, 1.39] |
| 8.4 Calcitonin versus control | 1 | 94 | Rate Ratio (Random, 95% CI) | 0.88 [0.12, 6.25] |
| 8.5 Cimetidine versus control | 1 | 60 | Rate Ratio (Random, 95% CI) | 1.14 [0.64, 2.02] |
| 8.6 EDTA versus control | 1 | 64 | Rate Ratio (Random, 95% CI) | 0.63 [0.28, 1.39] |
| 8.7 Gabexate versus control | 3 | 375 | Rate Ratio (Random, 95% CI) | 0.76 [0.61, 0.95] |
| 8.8 Glucagon versus control | 2 | 90 | Rate Ratio (Random, 95% CI) | 1.19 [0.51, 2.80] |
| 8.9 Lexipafant versus control | 1 | 290 | Rate Ratio (Random, 95% CI) | 0.61 [0.44, 0.85] |
| 8.10 Octreotide versus control | 4 | 634 | Rate Ratio (Random, 95% CI) | 0.78 [0.58, 1.05] |
| 8.11 Probiotics versus control | 3 | 397 | Rate Ratio (Random, 95% CI) | 0.84 [0.52, 1.36] |
| 8.12 Somatostatin versus control | 2 | 134 | Rate Ratio (Random, 95% CI) | 0.75 [0.26, 2.18] |
| 8.13 Ulinastatin versus control | 1 | 129 | Rate Ratio (Random, 95% CI) | 0.69 [0.32, 1.46] |
| 8.14 Gabexate versus aprotinin | 1 | 182 | Rate Ratio (Random, 95% CI) | 0.66 [0.38, 1.14] |
| 8.15 Glucagon versus atropine | 1 | 150 | Rate Ratio (Random, 95% CI) | 0.79 [0.36, 1.73] |
| 8.16 Oxyphenonium versus glucagon | 1 | 62 | Rate Ratio (Random, 95% CI) | 0.93 [0.65, 1.34] |
| 8.17 Octreotide plus ulinastatin versus octreotide | 1 | 120 | Rate Ratio (Random, 95% CI) | 0.29 [0.17, 0.48] |
| 9 Requirement for additional invasive intervention | 32 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 9.1 Antibiotics versus control | 14 | 884 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.59, 1.13] |
| 9.2 Aprotinin versus control | 2 | 237 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.23, 1.47] |
| 9.3 Calcitonin versus control | 2 | 125 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.30 [0.08, 1.16] |
| 9.4 Cimetidine versus control | 1 | 60 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.13 [0.01, 2.61] |
| 9.5 EDTA versus control | 1 | 64 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.14, 3.29] |
| 9.6 Gabexate versus control | 3 | 426 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.37, 0.90] |
| 9.7 Glucagon versus control | 2 | 260 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.26 [0.58, 2.77] |
| 9.8 Octreotide versus control | 3 | 854 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.76 [0.48, 1.21] |
| 9.9 Probiotics versus control | 2 | 358 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.50 [0.83, 2.71] |
| 9.10 Somatostatin versus control | 1 | 100 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.40 [0.11, 1.38] |
| 9.11 Gabexate versus aprotinin | 1 | 182 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.5 [0.19, 1.32] |
| 9.12 Glucagon versus aprotinin | 1 | 134 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.33 [0.44, 4.08] |
| 9.13 Oxyphenonium versus glucagon | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.13, 7.59] |
| 10 Endoscopic or radiological drainage of collections | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 10.1 Antibiotics versus control | 1 | 23 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 9.07] |
| 10.2 Octreotide versus control | 1 | 372 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.40, 1.96] |
| 10.3 Probiotics versus control | 1 | 39 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.20, 4.44] |
Comparison 2. Acute necrotising pancreatitis.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term mortality | 11 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Antibiotics versus control | 10 | 683 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.52, 1.30] |
| 1.2 Gabexate versus aprotinin | 1 | 116 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.20, 1.36] |
| 2 Serious adverse events (proportion) | 5 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 Antibiotics versus control | 4 | 281 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.46, 1.54] |
| 2.2 Gabexate versus aprotinin | 1 | 116 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.22, 4.91] |
| 3 Serious adverse events (number) | 7 | Rate Ratio (Fixed, 95% CI) | Subtotals only | |
| 3.1 Antibiotics versus control | 7 | Rate Ratio (Fixed, 95% CI) | 0.79 [0.59, 1.06] | |
| 4 Organ failure | 4 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 4.1 Antibiotics versus control | 4 | 211 | Odds Ratio (M‐H, Random, 95% CI) | 0.78 [0.42, 1.45] |
| 5 Infected pancreatic necrosis | 6 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 Antibiotics versus control | 6 | 426 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.51, 1.42] |
| 6 Sepsis | 2 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 6.1 Antibiotics versus control | 1 | 60 | Odds Ratio (M‐H, Random, 95% CI) | 0.42 [0.11, 1.60] |
| 6.2 Gabexate versus aprotinin | 1 | 116 | Odds Ratio (M‐H, Random, 95% CI) | 1.05 [0.22, 4.91] |
Comparison 3. Severe acute pancreatitis.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Short‐term mortality | 22 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Antibiotics versus control | 9 | 542 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.53, 1.27] |
| 1.2 Aprotinin versus control | 2 | 103 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.19, 2.30] |
| 1.3 Calcitonin versus control | 1 | 31 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.11, 5.46] |
| 1.4 Gabexate versus control | 1 | 52 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.19 [0.04, 0.99] |
| 1.5 Probiotics versus control | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.25 [0.05, 1.34] |
| 1.6 Activated protein C versus control | 1 | 32 | Odds Ratio (M‐H, Fixed, 95% CI) | 8.56 [0.41, 180.52] |
| 1.7 Somatostatin versus control | 2 | 182 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.51 [0.21, 1.23] |
| 1.8 Somatostatin plus omeprazole versus control | 1 | 140 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.23 [0.05, 1.11] |
| 1.9 Somatostatin plus ulinastatin versus control | 1 | 122 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.43 [0.15, 1.23] |
| 1.10 Thymosin versus control | 1 | 24 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.11 Ulinastatin versus control | 1 | 70 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.24 [0.04, 1.29] |
| 1.12 Octreotide plus ulinastatin versus octreotide | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.31 [0.06, 1.60] |
| 1.13 Somatostatin plus gabexate versus somatostatin | 1 | 252 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.37, 2.33] |
| 1.14 Somatostatin plus ulinastatin versus somatostatin | 2 | 369 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.73 [0.34, 1.56] |
| 1.15 Somatostatin plus ulinastatin plus gabexate versus somatostatin | 1 | 238 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.61 [0.21, 1.74] |
| 1.16 Somatostatin plus ulinastatin versus somatostatin plus gabexate | 1 | 254 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.72 [0.26, 1.95] |
| 1.17 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus gabexate | 1 | 246 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.23, 1.86] |
| 1.18 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus ulinastatin | 1 | 240 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.30, 2.80] |
| 2 Serious adverse events (proportion) | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 Antibiotics versus control | 3 | 164 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.27, 1.18] |
| 3 Serious adverse events (number) | 13 | Rate Ratio (Random, 95% CI) | Subtotals only | |
| 3.1 Antibiotics versus control | 5 | Rate Ratio (Random, 95% CI) | 0.81 [0.52, 1.25] | |
| 3.2 Aprotinin versus control | 2 | Rate Ratio (Random, 95% CI) | 0.65 [0.25, 1.71] | |
| 3.3 Gabexate versus control | 1 | Rate Ratio (Random, 95% CI) | 0.64 [0.37, 1.10] | |
| 3.4 Probiotics versus control | 2 | Rate Ratio (Random, 95% CI) | 0.62 [0.24, 1.59] | |
| 3.5 Somatostatin versus control | 1 | Rate Ratio (Random, 95% CI) | 1.07 [0.67, 1.69] | |
| 3.6 Somatostatin plus omeprazole versus control | 1 | Rate Ratio (Random, 95% CI) | 0.36 [0.19, 0.70] | |
| 3.7 Somatostatin plus ulinastatin versus control | 1 | Rate Ratio (Random, 95% CI) | 0.30 [0.15, 0.60] | |
| 3.8 Octreotide plus ulinastatin versus octreotide | 1 | Rate Ratio (Random, 95% CI) | 0.30 [0.17, 0.51] | |
| 3.9 Somatostatin plus ulinastatin versus somatostatin | 1 | Rate Ratio (Random, 95% CI) | 0.28 [0.15, 0.56] | |
| 4 Organ failure | 6 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.1 Antibiotics versus control | 3 | 137 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.40, 1.99] |
| 4.2 Lexipafant versus control | 0 | 0 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Probiotics versus control | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.40 [0.12, 1.36] |
| 4.4 Ulinastatin versus control | 1 | 67 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.05 [0.01, 0.21] |
| 4.5 Somatostatin plus gabexate versus somatostatin | 1 | 252 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.33, 1.80] |
| 4.6 Somatostatin plus ulinastatin versus somatostatin | 1 | 246 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.23, 1.45] |
| 4.7 Somatostatin plus ulinastatin plus gabexate versus somatostatin | 1 | 238 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.17, 1.25] |
| 4.8 Somatostatin plus ulinastatin versus somatostatin plus gabexate | 1 | 254 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.29, 1.92] |
| 4.9 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus gabexate | 1 | 246 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.21, 1.65] |
| 4.10 Somatostatin plus ulinastatin plus gabexate versus somatostatin plus ulinastatin | 1 | 240 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.27, 2.35] |
| 5 Infected pancreatic necrosis | 8 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 Antibiotics versus control | 6 | 341 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.73 [0.41, 1.33] |
| 5.2 Probiotics versus control | 2 | 101 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.60 [0.22, 1.68] |
| 6 Sepsis | 3 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 6.1 Aprotinin versus control | 2 | 103 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.87 [0.50, 6.98] |
| 6.2 Probiotics versus control | 1 | 62 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.36 [0.10, 1.36] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Abraham 2013.
| Methods | Randomised clinical trial | |
| Participants | Country: India Number randomised: 135 Postrandomisation dropouts: 6 (4.4%) Revised sample size: 129 Average age: 39 years Women: 13 (10.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 62 (48.1%) Moderate pancreatitis: not stated Severe pancreatitis: 67 (51.9%) Persistent organ failure: not stated Infected pancreatitis: 0 Inclusion criteria
|
|
| Interventions | Group 1: ulinastatin (n = 30), 200,000 IU twice daily for 5 days Group 2: placebo (n = 32) |
|
| Outcomes | Mortality, adverse events, organ failure, hospital stay Follow‐up: until discharge or maximum of 22 days |
|
| Notes | Reasons for postrandomisation dropouts: withdrew consent, screening error, died | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled, multi‐centre trial across 15 centres in India". |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled, multi‐centre trial across 15 centres in India". |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Balldin 1983.
| Methods | Randomised clinical trial | |
| Participants | Country: Sweden Number randomised: 55 Postrandomisation dropouts: not stated Revised sample size: 55 Average age: not stated Women: 15 (27.3%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 55 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: acute pancreatitis undergoing peritoneal lavage | |
| Interventions | Group 1: aprotinin (n = 26), 500,000 KIU in lavage fluid every 2 h for an average of 2.7 days Group 2: no intervention (n = 29) | |
| Outcomes | Mortality, serious adverse events, adverse events, sepsis, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Comment: supported by grants from the ….Bayer AG…. |
| Other bias | Low risk | Comment: no other risk of bias |
Bansal 2011.
| Methods | Randomised clinical trial | |
| Participants | Country: India Number randomised: 44 Postrandomisation dropouts: 5 (11.4%) Revised sample size: 39 Average age: 39 years Women: 9 (23.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis within 96 h of onset of symptoms Exclusion criteria
|
|
| Interventions | Group 1: antioxidants (n = 19): vitamin A, C, E ‐ initially parenterally and then orally when the participant could consume orally for a total of 14 days Group 2: no intervention (n = 20) | |
| Outcomes | Mortality, serious adverse events, adverse events, organ failure, hospital stay Follow‐up: until discharge |
|
| Notes | Reasons for postrandomisation dropouts: lost to follow‐up, withdrew consent | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "[t]his was a single‐center, prospective randomized, open‐label with blinded endpoint assessment study of antioxidant therapy". |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[t]his was a single‐center, prospective randomized, open‐label with blinded endpoint assessment study of antioxidant therapy". |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Low risk | Quote: "[s]ource of support: Nil". |
| Other bias | Low risk | Comment: no other risk of bias. |
Barreda 2009.
| Methods | Randomised clinical trial | |
| Participants | Country: Peru Number randomised: 80 Postrandomisation dropouts: 22 (27.5%) Revised sample size: 58 Average age: 50 years Women: 24 (41.4%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 58 (100%) Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with necrotising pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 24): imipenem 500 mg 4 times daily for 14 days Group 2: no intervention (n = 34) | |
| Outcomes | Mortality, serious adverse events, adverse events, infected pancreatic necrosis, requirement for additional intervention, length of hospital stay Follow‐up: 2 months |
|
| Notes | Reasons for postrandomisation dropouts: protocol violations | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "sealed envelopes". |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Berling 1994.
| Methods | Randomised clinical trial | |
| Participants | Country: multicentric, international Number randomised: 48 Postrandomisation dropouts: not stated Revised sample size: 48 Average age: 56 years Women: 17 (35.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 48 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: participants with acute severe pancreatitis with circulatory insufficiency or peritonitis Exclusion criteria
|
|
| Interventions | Group 1: aprotinin (n = 22), 20 million KIU in 7 lavages over 30 h Group 2: no intervention (n = 26) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, sepsis, hospital stay, ICU stay Follow‐up: 1 month |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he Bayer . . . and was also responsible for coding the bottles." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "prospective double‐blind randomized multicenter trial" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "prospective double‐blind randomized multicenter trial" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[t]his study was supported by grants from Bayer AG". |
| Other bias | Low risk | Comment: no other risk of bias |
Besselink 2008.
| Methods | Randomised clinical trial | |
| Participants | Country: Netherlands Number randomised: 298 Postrandomisation dropouts: 2 (0.7%) Revised sample size: 296 Average age: 60 years Women: 122 (41.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with predicted severe acute pancreatitis | |
| Interventions | Group 1: probiotics (n = 152): ecologic 641 (maximum of 28 days or until development of pancreatic necrosis or fluid collection) Group 2: placebo (n = 144) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, organ failure, infected pancreatic necrosis, hospital stay, ICU stay Follow‐up: 3 months |
|
| Notes | Reasons for postrandomisation dropouts: did not receive drug, wrong diagnosis of acute pancreatitis | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "[r]andomisation was done with a computer‐generated permuted‐block sequence.". |
| Allocation concealment (selection bias) | Low risk | Quote: "[b]oth the probiotic and placebo preparations were packaged in identical, numbered sachets that were stored in identical, numbered containers." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[a]ll doctors, nurses, research staff , and patients involved remained unaware of the actual product administered during the entire study period." |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[a]ll doctors, nurses, research staff , and patients involved remained unaware of the actual product administered during the entire study period." |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "HMT is an employee of Winclove Bio Industries, Amsterdam". |
| Other bias | Low risk | Comment: no other risk of bias |
Birk 1994.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 20 Postrandomisation dropouts: not stated Revised sample size: 20 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 20 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis | |
| Interventions | Group 1: antioxidants (n = 10): sodium selenite 600 μg/day for 8 days Group 2: no intervention (n = 10) | |
| Outcomes | None of the outcomes of interest were reported. Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias. |
Bredkjaer 1988.
| Methods | Randomised clinical trial | |
| Participants | Country: Denmark Number randomised: 66 Postrandomisation dropouts: 9 (13.6%) Revised sample size: 57 Average age: not stated Women: 26 (45.6%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria:
|
|
| Interventions | Group 1: NSAID (n = 27): indomethacin 100 mg rectal for 7 days Group 2: placebo (n = 30) | |
| Outcomes | The outcomes reported were: hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: chronic pancreatitis, wrong diagnosis, death | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Buchler 1993.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 223 Postrandomisation dropouts: not stated Revised sample size: 223 Average age: 50 years Women: 87 (39%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with moderate or severe acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: gabexate mesilate (n = 115), 53 mg/kg/day for 7 days Group 2: placebo (n = 108) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, sepsis, hospital stay Follow‐up: 3 months |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "[a] randomization list was applied to get a random sequence of GM and placebos for increasing package numbers." |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he drug packages for each hospital were numbered sequentially and the package number was used as patient number" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "randomized, double‐blind trial" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "randomized, double‐blind trial" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Chen 2000.
| Methods | Randomised clinical trial | |
| Participants | Country: Taiwan Number randomised: 52 Postrandomisation dropouts: not stated Revised sample size: 52 Average age: 44 years Women: 15 (28.8%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 0 (0%) Persistent organ failure: 52 (100%) Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis with organ failure | |
| Interventions | Group 1: gabexate mesilate (n = 26), 100 mg/h for 7 days Group 2: placebo (n = 26) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery Follow‐up: 3 months |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Unclear risk | Comment: this information was not available. |
Chen 2002a.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 68 Postrandomisation dropouts: 6 (8.8%) Revised sample size: 62 Average age: 53 years Women: 33 (53.2%) Acute interstitial oedematous pancreatitis: 62 (100%) Necrotising pancreatitis: 0 (0%) Mild pancreatitis: 62 (100%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 0 (0%) Persistent organ failure: 0 (0%) Infected pancreatitis: not stated Inclusion criteria: people with mild pancreatitis | |
| Interventions | Group 1: ulinastatin (n = 48), 50,000 IU twice daily for 3 days followed by once daily for 5 days Group 2: gabexate mesilate (n = 14), 100 mg twice daily for 3 days followed by once daily for 5 days | |
| Outcomes | Serious adverse events, adverse events Follow‐up: not stated (probably 2 weeks) |
|
| Notes | Reasons for postrandomisation dropouts: recent or current treatment with other drugs | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Chen 2002b.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 26 Postrandomisation dropouts: 1 (3.8%) Revised sample size: 25 Average age: 59 years Women: 12 (48%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 15 (60%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 25 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe necrotising pancreatitis | |
| Interventions | Group 1: ulinastatin (n = 14), 100,000 IU twice daily for 3 days followed by 50,000 IU once daily for 5‐10 days Group 2: octreotide (n = 11), 0.3 mg twice daily for 3 days followed by 0.1 mg once daily for 5 days | |
| Outcomes | Adverse events Follow‐up: not stated (probably 2 weeks) |
|
| Notes | Reasons for postrandomisation dropouts: death after starting treatment | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Choi 1989.
| Methods | Randomised clinical trial | |
| Participants | Country: Hong Kong, China
Number randomised: 71
Postrandomisation dropouts: not stated
Revised sample size: 71
Average age: 61 years
Women: 39 (54.9%)
Acute interstitial oedematous pancreatitis: not stated
Necrotising pancreatitis: not stated
Mild pancreatitis: not stated
Moderate pancreatitis: not stated
Severe pancreatitis: 15 (21.1%)
Persistent organ failure: not stated
Infected pancreatitis: not stated
Inclusion criteria: people with acute pancreatitis Exclusion criteria: people with acute pancreatitis caused by trauma, iatrogenic, or malignancy |
|
| Interventions | Group 1: somatostatin (n = 35), 250 μg bolus followed by 100 μg/h for 48 h Group 2: no intervention (n = 36) | |
| Outcomes | Mortality, serious adverse events, adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[r]andomisation was done by drawing sealed envelopes" |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Chooklin 2007.
| Methods | Randomised clinical trial | |
| Participants | Country: Ukraine Number randomised: 34 Postrandomisation dropouts: not stated Revised sample size: 34 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 34 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: antioxidants (N‐acetyl cysteine, unspecified dose and duration) plus corticosteroids (dexamethasone, unspecified dose and duration) (n = 16) Group 2: no intervention (n = 18) | |
| Outcomes | None of the outcomes of interest were reported. Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Unclear risk | Comment: no other risk of bias |
Debas 1980.
| Methods | Randomised clinical trial | |
| Participants | Country: Canada Number randomised: 66 Postrandomisation dropouts: not stated Revised sample size: 66 Average age: 53 years Women: 25 (37.9%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: glucagon (n = 33), 1 mg every 3 h (duration not stated) Group 2: placebo (n = 33) | |
| Outcomes | Mortality, serious adverse events, adverse events, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[o]nce we decided to enter a patient into the study, the hospital pharmacy randomly assigned…" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[p]rospective randomized double‐blind study" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[p]rospective randomized double‐blind study" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Delcenserie 1996.
| Methods | Randomised clinical trial | |
| Participants | Country: France Number randomised: 23 Postrandomisation dropouts: 0 (0%) Revised sample size: 23 Average age: 43 years Women: 2 (8.7%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: 23 (100%) Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 11), ceftazidime 2 g IV 3 times daily; amikacin 7.5 mg/kg IV BD; and metronidazole 0.5 g IV 3 times daily for 10 days Group 2: no intervention (n = 12) | |
| Outcomes | Mortality, serious adverse events, requirement for surgery, requirement for endoscopic or radiological drainage, organ failure, infected pancreatic necrosis, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "random‐number table" |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Delcenserie 2001.
| Methods | Randomised clinical trial | |
| Participants | Country: France Number randomised: 81 Postrandomisation dropouts: not stated Revised sample size: 81 Average age: 47 years Women: 14 (17.3%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 81 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 53): ciprofloxacin for 7 days or 21 days (random choice); dose not stated Group 2: no intervention (n = 28) | |
| Outcomes | Mortality, serious adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Dellinger 2007.
| Methods | Randomised clinical trial | |
| Participants | Country: multicentric, international Number randomised: 100 Postrandomisation dropouts: 0 (0%) Revised sample size: 100 Average age: 50 years Women: 30 (30%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 100 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 100 (100%) Persistent organ failure: not stated Infected pancreatitis: 0 Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 50): meropenem 1 g IV 3 times daily for 7‐21 days (recommended duration: 14 days) Group 2: placebo (n = 50) | |
| Outcomes | Mortality, serious adverse events, adverse events, infected pancreatic necrosis Follow‐up: 1.5 months |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "[t]he treatment given to each patient was determined by a random scheme prepared by the Biostatistics group at AstraZeneca (Wilmington, DE), using computer software that incorporates a standard procedure for generating random numbers" |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he treatment given to each patient was determined by a random scheme prepared by the Biostatistics group at AstraZeneca (Wilmington, DE), using computer software that incorporates a standard procedure for generating random numbers" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled study" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled study" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[s]upported by a grant from AstraZeneca Pharmaceuticals" |
| Other bias | Low risk | Comment: no other risk of bias |
Dürr 1978.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 69 Postrandomisation dropouts: not stated Revised sample size: 69 Average age: 49 years Women: 27 (39.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: glucagon (n = 33), 10 mg daily until surgery or at least 5 days in those who did not undergo surgery Group 2: placebo (n = 36) | |
| Outcomes | Mortality, requirement for surgery, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Ebbehøj 1985.
| Methods | Randomised clinical trial | |
| Participants | Country: Denmark Number randomised: 30 Postrandomisation dropouts: 0 (0%) Revised sample size: 30 Average age: 55 years Women: 10 (33.3%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: NSAID (n = 14), indomethacin 50 mg PR twice daily for 7 days Group 2: placebo (n = 16) | |
| Outcomes | Hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[c]ontrolled double‐blind trial". |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[c]ontrolled double‐blind trial". |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "[i]ndomethacin (Confortid) and placebo were generously supplied by Dumex Ltd, Denmark". |
| Other bias | Low risk | Comment: no other risk of bias |
Finch 1976.
| Methods | Randomised clinical trial | |
| Participants | Country: USA Number randomised: 62 Postrandomisation dropouts: 4 (6.5%) Revised sample size: 58 Average age: 36 years Women: 24 (41.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 31): ampicillin 500 mg to 1 g 4 times daily for 7 days (keflin 1 g 4 times daily for 7 days in people allergic to penicillin) Group 2: no intervention (n = 27) | |
| Outcomes | Mortality, adverse events, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: required surgery, developed pneumonia, went home against medical advice, malignancy | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "[o]n a randomized pre‐selected basis a card was drawn to determine in which group (antibiotic treatment or non‐antibiotic treatment) the patient was to be included." Comment: further details on whether the card was an open or held by a researcher not involved in recruitment are not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Freise 1986.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 50 Postrandomisation dropouts: not stated Revised sample size: 50 Average age: not stated Women: 17 (34%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: gabexate mesilate (n = 25), 150 mg IV 3 times daily for 7 days Group 2: placebo (n = 25) | |
| Outcomes | Mortality, serious adverse events, adverse events, organ failure, sepsis Follow‐up: not stated |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Comment: the drug code was concealed by third party. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Frulloni 1994.
| Methods | Randomised clinical trial | |
| Participants | Country: Italy Number randomised: 116 Postrandomisation dropouts: not stated Revised sample size: 116 Average age: 57 years Women: 49 (42.2%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 116 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: gabexate mesilate (n = 65), 3 g/day for 7 days Group 2: aprotinin (n = 51), 1.5 million KIU/day for 7 days | |
| Outcomes | Mortality, serious adverse events, adverse events, sepsis Follow‐up: 3 months |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Garcia‐Barrasa 2009.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 46 Postrandomisation dropouts: 5 (10.9%) Revised sample size: 41 Average age: 63 years Women: 12 (29.3%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 41 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 41 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute necrotising pancreatitis | |
| Interventions | Group 1: antibiotics (n = 22): ciprofloxacin 300 mg twice daily for 10 days Group 2: placebo (n = 19) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, organ failure, infected pancreatic necrosis, hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: 3 ‐ no confirmed necrosis; 2 fulminant pancreatitis | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[p]rospective, randomized, placebo‐controlled, double‐blind study" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[p]rospective, randomized, placebo‐controlled, double‐blind study" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Low risk | Quote: "[t]his study was promoted by the “Bellvitge Hospital” and has not received any grant or payment from the pharmaceutical industry". |
| Other bias | Low risk | Comment: no other risk of bias |
Gilsanz 1978.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 62. Postrandomisation dropouts: not stated Revised sample size: 62 Average age: 52 years Women: 44 (71%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: 48 (77.4%) Severe pancreatitis: 14 (22.6%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: glucagon (n = 31), 1 mg IV every 4 h (duration ‐ not stated) Group 2: oxyphenonium gromomethylate (n = 31), 1 mg IV every 4 h (duration ‐ not stated) | |
| Outcomes | Mortality, adverse events, requirement for surgery Follow‐up: 24 months |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "sealed envelope" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Gjørup 1992.
| Methods | Randomised clinical trial | |
| Participants | Country: Denmark Number randomised: 63 Postrandomisation dropouts: not stated Revised sample size: 63 Average age: 49 years Women: 22 (34.9%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: somatostatin (n = 33), 250 μg/h for 3 days Group 2: placebo (n = 30) | |
| Outcomes | Mortality, serious adverse events, adverse events, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "by selecting sealed envelopes" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blinded trial" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blinded trial" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Goebell 1979.
| Methods | Randomised clinical trial | |
| Participants | Country: multicentric, international Number randomised: 94 Postrandomisation dropouts: not stated Revised sample size: 94 Average age: 55 years Women: 37 (39.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 29 (30.9%) Moderate pancreatitis: 49 (52.1%) Severe pancreatitis: 16 (17%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: calcitonin (n = 50), synthetic salmon calcitonin 20 μg 3 times daily for 6 days Group 2: placebo (n = 44) | |
| Outcomes | Mortality, adverse events, requirement for surgery, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Goebell 1988.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 162 Postrandomisation dropouts: 11 (6.8%) Revised sample size: 151 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with moderate or severe pancreatitis | |
| Interventions | Group 1: gabexate mesilate (n = 76), 150 mg every 2 h followed by 0.5 mg/kg/h for 7 days Group 2: placebo (n = 75) | |
| Outcomes | Mortality, serious adverse events, requirement for surgery Follow‐up: 3 months |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Grupo Español 1996.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 70 Postrandomisation dropouts: 9 (12.9%) Revised sample size: 61 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 61 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis | |
| Interventions | Group 1: somatostatin (n = 30), 250 μg/h for 5 days Group 2: placebo (n = 31) | |
| Outcomes | Mortality Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: did not complete the study | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled, multi‐centre trial across 15 centres in India" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled, multi‐centre trial across 15 centres in India" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Guo 2015.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 120 Postrandomisation dropouts: not stated Revised sample size: 120 Average age: 46 years Women: 58 (48.3%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 120 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis | |
| Interventions | Group 1: octerotide plus ulinastatin (n = 60), 0.1 mg SC 3 times daily for 7‐14 days Group 2: octreotide (n = 60), 10 million units IV continuous for 7‐14 days | |
| Outcomes | Mortality, serious adverse events, adverse events, length of hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Hansky 1969.
| Methods | Randomised clinical trial | |
| Participants | Country: Australia Number randomised: 24 Postrandomisation dropouts: not stated Revised sample size: 24 Average age: not stated Women: 7 (29.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 3 (12.5%) Moderate pancreatitis: 15 (62.5%) Severe pancreatitis: 6 (25%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: iniprol (n = 15), single IV dose of 1 million units, followed by 500,000 units IV 4 times daily for 4‐8 days depending upon clinical course Group 2: no intervention (n = 9) | |
| Outcomes | Mortality, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "[t]he drug was not evaluated in a double‐blind manner". |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "[t]he drug was not evaluated in a double‐blind manner". |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "I am grateful to Difrex (Australia) laboratories for supplying . . ." |
| Other bias | Low risk | Comment: no other risk of bias |
Hejtmankova 2003.
| Methods | Randomised clinical trial | |
| Participants | Country: not stated Number randomised: 41 Postrandomisation dropouts: not stated Revised sample size: 41 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 41 (100%). Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis | |
| Interventions | Group 1: antibiotics (n = 20): meropenem 500 mg 3 times daily for 10 days Group 2: no intervention (n = 21) | |
| Outcomes | Mortality, adverse events, requirement for surgery, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Imrie 1978.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 161 Postrandomisation dropouts: not stated Revised sample size: 161 Average age: 51 years Women: 92 (57.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 60 (37.3%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: aprotinin (n = 80), 500 000 KIU bolus followed by 200 000 KIU 4 times daily for 5 days Group 2: placebo (n = 81) | |
| Outcomes | Mortality, serious adverse events, adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "sealed envelope". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind trial". |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind trial". |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[i]n addition to providing both Trasylol and placebo, Bayer Pharmaceuticals contributed the financial support of a research assistant". |
| Other bias | Low risk | Comment: no other risk of bias |
Imrie 1980.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 50 Postrandomisation dropouts: not stated Revised sample size: 50 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 29 (58%) Moderate pancreatitis: not stated Severe pancreatitis: 21 (42%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: aprotinin (n = 25), 2 million units KIU bolus followed by 400,000 KIU 4 h later Group 2: placebo (n = 25) | |
| Outcomes | Mortality Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind trial" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind trial" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Isenmann 2004.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 119 Postrandomisation dropouts: 5 (4.2%) Revised sample size: 114 Average age: 47 years Women: 27 (23.7%) Acute interstitial oedematous pancreatitis: 38 (33.3%) Necrotising pancreatitis: 76 (66.7%) Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with predicted severe pancreatitis | |
| Interventions | Group 1: antibiotics (n = 58): metronidazole 500 mg twice daily and ciprofloxacin 400 mg twice daily (duration not reported) Group 2: placebo (n = 56) | |
| Outcomes | Serious adverse events, adverse events, requirement for surgery, infected pancreatic necrosis, hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: lost to follow‐up, withdrawn from study prior to medication | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[s]tudy medication for each patient (verum or placebo) was packed in identical vials and labelled with consecutive patient numbers according to the randomization sequence". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind trial" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind trial" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "[s]upported by study medication provided from Bayer Vital and Ratiopharm as well as a financial grant from Bayer Vital" |
| Other bias | Low risk | Comment: no other risk of bias |
Johnson 2001.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 291 Postrandomisation dropouts: 1 (0.3%) Revised sample size: 290 Average age: 63 years Women: 124 (42.8%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria: age < 18 or > 80 years |
|
| Interventions | Group 1: lexipafant (n = 151), 100 mg daily for 7 days Group 2: placebo (n = 139) | |
| Outcomes | Mortality, serious adverse events, adverse events, organ failure, sepsis, hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: withdrew from the study | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind, placebo controlled, randomised, parallel group" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind, placebo controlled, randomised, parallel group" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[t]his study was funded by British Biotech Pharmaceuticals Ltd, Oxford, UK". |
| Other bias | Low risk | Comment: no other risk of bias |
Kalima 1980.
| Methods | Randomised clinical trial | |
| Participants | Country: Finland Number randomised: 80 Postrandomisation dropouts: 9 (11.3%) Revised sample size: 71 Average age: 46 years Women: 28 (39.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: glucagon (n = 32), 7.5 mg twice daily for 4‐5 days Group 2: placebo (n = 29) | |
| Outcomes | Mortality, serious adverse events, adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: underwent surgery, wrong diagnosis | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: although placebo was used, there was no mention of blinding. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: although placebo was used, there was no mention of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported |
| For profit‐bias | Unclear risk | Comment: this information was not available |
| Other bias | Low risk | Comment: no other risk of bias |
Kingsnorth 1995.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 83 Postrandomisation dropouts: not stated Revised sample size: 83 Average age: 59 years Women: 41 (49.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 54 (65.1%) Moderate pancreatitis: not stated Severe pancreatitis: 29 (34.9%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis within 48 h of onset of symptoms Exclusion criteria
|
|
| Interventions | Group 1: lexipafant (n = 42), 15 mg 4 times daily for 3 days Group 2: placebo (n = 41) | |
| Outcomes | Mortality, adverse events Follow‐up: 1 week |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "S.W.G. was supported by British Biotech, Oxford, UK" |
| Other bias | Low risk | Comment: no other risk of bias |
Kirsch 1978.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 150 Postrandomisation dropouts: not stated Revised sample size: 150 Average age: 53 years Women: 78 (52%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 35 (23.3%) Moderate pancreatitis: 61 (40.7%) Severe pancreatitis: 54 (36%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: glucagon (n = 75), 10 mg/day for 4 days Group 2: atropine (n = 75), 4 days (dose not stated) | |
| Outcomes | Mortality, serious adverse events, adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Kronborg 1980.
| Methods | Randomised clinical trial | |
| Participants | Country: Denmark Number randomised: 22 Postrandomisation dropouts: not stated Revised sample size: 22 Average age: not stated Women: 4 (18.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: 11 (50%) Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: glucagon (n = 10), 1 mg IV followed by 6 mg/day for 3 days Group 2: placebo (n = 12) | |
| Outcomes | Mortality, adverse events Follow‐up: until discharge |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: although authors stated they did not exclude any participants for wrong diagnosis, it was not clear whether they excluded participants for other reasons. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Llukacaj 2012.
| Methods | Randomised clinical trial | |
| Participants | Country: Albania Number randomised: 80 Postrandomisation dropouts: not stated Revised sample size: 80 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 80 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: 0 Inclusion criteria: people with non‐infected necrotising pancreatitis | |
| Interventions | Group 1: antibiotics (n = 40): imipenem 750 mg IV twice daily for 7 days Group 2: placebo (n = 40) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, infected pancreatic necrosis Follow‐up: 1 month |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: although authors stated they did not exclude any participants for wrong diagnosis, it was not clear whether they excluded participants for other reasons. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Luengo 1994.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 100 Postrandomisation dropouts: not stated Revised sample size: 100 Average age: 55 years Women: 39 (39%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 78 (78%) Moderate pancreatitis: not stated Severe pancreatitis: 22 (22%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: somatostatin (n = 50), 250 μg/h for 48 h following a 250 μg bolus Group 2: no intervention (n = 50) | |
| Outcomes | Mortality, requirement for surgery, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[p]atients were randomly divided by means of the sealed‐envelope method and grouped according to therapy". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: although authors stated they did not exclude any participants for wrong diagnosis, it was not clear whether they excluded participants for other reasons. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Luiten 1995.
| Methods | Randomised clinical trial | |
| Participants | Country: the Netherlands Number randomised: 109 Postrandomisation dropouts: 7 (6.4%) Revised sample size: 102 Average age: 55 years Women: 42 (41.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 102 (100%) Persistent organ failure: not stated Infected pancreatitis: 0 Inclusion criteria: people with severe pancreatitis | |
| Interventions | Group 1: antibiotics (n = 50): selective digestive decontamination using colistin 200 mg, amphotericin 500 mg, and norfloxacin 50 mg 4 times daily orally and as rectal enema along with short course of cefotaxime 500 mg IV 3 times daily until gram‐negative bacteria were eliminated from oral cavity and rectum. Total duration of treatment: until patient was extubated and taking oral feeds Group 2: no intervention (n = 52) | |
| Outcomes | Mortality, adverse events, requirement for surgery, hospital stay Follow‐up: until discharge |
|
| Notes | Reasons for postrandomisation dropouts: perioperatively proven infected pancreatic necrosis or wrong clinical diagnosis | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[a] 24‐hour randomization service was available to randomize patients with stratification per center". |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Marek 1999.
| Methods | Randomised clinical trial | |
| Participants | Country: Poland Number randomised: 73 Postrandomisation dropouts: 0 (0%) Revised sample size: 73 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 56 (76.7%) Moderate pancreatitis: not stated Severe pancreatitis: 17 (23.3%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: antioxidants (n = 35): vitamin C 500 mg IV 3 times daily for 5 days Group 2: placebo (n = 38) | |
| Outcomes | None of the outcomes of interest were reported. Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Martinez 1984.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 31 Postrandomisation dropouts: 0 (0%) Revised sample size: 31 Average age: 48 years Women: 6 (19.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 31 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis | |
| Interventions | Group 1: calcitonin (n = 14), synthetic salmon calcitonin 100 MRC units (equivalent to 100 IU) IV 3 times daily for 5 days or more Group 2: placebo (n = 17) | |
| Outcomes | Mortality, requirement for surgery, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: although some participants were excluded from hospital stay, they were included for mortality and requirement of surgical intervention. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
McKay 1997a.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 58 Postrandomisation dropouts: 0 (0%) Revised sample size: 58 Average age: 69 years Women: 32 (55.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with moderate or severe pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: octreotide (n = 28), 1 mg/day IV for 5 days Group 2: placebo (n = 30) | |
| Outcomes | Mortality, serious adverse events, adverse events, organ failure, infected pancreatic necrosis, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "[r]andomization was by the use of sequentially numbered treatment packs containing either octreotide or placebo as determined by a computer‐generated random code." |
| Allocation concealment (selection bias) | Low risk | Quote: "[r]andomization was by the use of sequentially numbered treatment packs containing either octreotide or placebo as determined by a computer‐generated random code." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[p]atients, investigators, and medical staff were blinded regarding the nature of the trial infusion". |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[p]atients, investigators, and medical staff were blinded regarding the nature of the trial infusion". |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
McKay 1997b.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 51 Postrandomisation dropouts: 1 (2%) Revised sample size: 50 Average age: 65 years Women: 21 (42%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with predicted severe pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: lexipafant (n = 26), 4 mg bolus IV followed by 4 mg/h by continuous infusion for 5‐7 days Group 2: placebo (n = 24) | |
| Outcomes | Mortality, organ failure, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: incorrect diagnosis | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[p]acks were numbered sequentially and prepared in advance by British Biotech (Oxford, UK)". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[i]nvestigators and patients were unaware of the nature of the trial infusion." |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[i]nvestigators and patients were unaware of the nature of the trial infusion." |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "[t]his study was supported by a grant from British Biotech". |
| Other bias | Low risk | Comment: no other risk of bias |
Moreau 1986.
| Methods | Randomised clinical trial | |
| Participants | Country: France Number randomised: 87 Postrandomisation dropouts: 3 (3.4%) Revised sample size: 84 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: somatostatin (n = 44), 400 μg for first 3 days, tapered and stopped on 4th day Group 2: placebo (n = 41) | |
| Outcomes | None of the outcomes of interest were reported. Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "Sonafi, kindly donated" |
| Other bias | Low risk | Comment: no other risk of bias |
MRC Multicentre Trial 1977.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 264 Postrandomisation dropouts: 7 (2.7%) Revised sample size: 257 Average age: not stated Women: 153 (59.5%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: aprotinin (n = 66), 500,000 IU IV followed by 300,000 units every 6 h for 5 days Group 2: glucagon (n = 68), 2 mg IV followed by 2 mg every 6 h for 5 days Group 3: placebo (n = 123) | |
| Outcomes | Mortality, requirement for surgery Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: initial amylase was too low | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled, multi‐centre trial across 15 centres in India" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[r]andomized, double‐blind, placebo‐controlled, multi‐centre trial across 15 centres in India" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Comment: the drugs and placebo were supplied by the pharmaceutical company. |
| Other bias | Low risk | Comment: no other risk of bias |
Nordback 2001.
| Methods | Randomised clinical trial | |
| Participants | Country: Finland Number randomised: 90 Postrandomisation dropouts: 32 (35.6%) Revised sample size: 58 Average age: 46 years Women: 7 (12.1%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 58 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: 0 (0%) Infected pancreatitis: not stated Inclusion criteria: people with acute necrotising pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 25): imipenem 1 g plus cilastatin IV 3 times daily; duration not stated Group 2: placebo (n = 33) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: older than 70 years of age, did not begin antibiotic as scheduled, criteria for pancreatic necrosis not fulfilled | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Ohair 1993.
| Methods | Randomised clinical trial | |
| Participants | Country: USA Number randomised: 180 Postrandomisation dropouts: not stated Revised sample size: 180 Average age: 37 years Women: 41 (22.8%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: octreotide (n = 90), 100 μg 3 times daily SC for duration of hospital stay Group 2: placebo (n = 90) | |
| Outcomes | Requirement for surgery, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Olah 2007.
| Methods | Randomised clinical trial | |
| Participants | Country: Hungary Number randomised: 83 Postrandomisation dropouts: 21 (25.3%) Revised sample size: 62 Average age: 47 years Women: 10 (16.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 62 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis Exclusion criteria: people with acute exacerbation of chronic pancreatitis. | |
| Interventions | Group 1: probiotics (n = 33): Synbiotic 2000 once daily for at least 1 week Group 2: no intervention (n = 29) Both groups received prebiotics (an intervention not of interest for this review). | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, organ failure, sepsis, infected pancreatic necrosis, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: because they were not severe acute pancreatitis after 48 h, did not tolerate jejunal feeding, participant removed the feeding tube | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Paran 1995.
| Methods | Randomised clinical trial | |
| Participants | Country: Israel Number randomised: 51 Postrandomisation dropouts: 13 (25.5%) Revised sample size: 38 Average age: 61 years Women: 18 (47.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: octreotide (n = 19), 01. mg SC 3 times daily for 14 days Group 2: no intervention (n = 19) | |
| Outcomes | Mortality, serious adverse events, adverse events, sepsis, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: failure to meet inclusion criteria, incomplete data, incorrect diagnosis | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "[a]s placebo vials were not available to us, the study was double blinded". |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "[a]s placebo vials were not available to us, the study was double blinded". |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Pederzoli 1993a.
| Methods | Randomised clinical trial | |
| Participants | Country: Italy Number randomised: 74 Postrandomisation dropouts: not stated Revised sample size: 74 Average age: 52 years Women: 30 (40.5%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 74 (100%) Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: antibiotics (n = 41): imipenem 0.5 g every 8 h for 2 weeks Group 2: no intervention (n = 33) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, organ failure, infected pancreatic necrosis Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "casual numbers table". |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Pederzoli 1993b.
| Methods | Randomised clinical trial | |
| Participants | Country: Italy Number randomised: 199 Postrandomisation dropouts: 17 (8.5%) Revised sample size: 182 Average age: 58 years Women: 78 (42.9%) Acute interstitial oedematous pancreatitis: 66 (36.3%) Necrotising pancreatitis: 116 (63.7%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: gabexate mesilate (n = 91), 3 g/day for 7 days Group 2: aprotinin (n = 91), 1,500,000 KIU/day for 7 days | |
| Outcomes | Mortality, adverse events, requirement for surgery Follow‐up: 3 months for mortality; all other complications ‐ 2 weeks |
|
| Notes | Reasons for postrandomisation dropouts: major protocol violations | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Perezdeoteyza 1980.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 40 Postrandomisation dropouts: not stated Revised sample size: 40 Average age: 56 years Women: 24 (60%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: cimetidine (n = 20), 1200 mg IV for 4‐5 days followed by 1000 mg oral for 10 days Group 2: placebo (n = 20) | |
| Outcomes | Mortality Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[r]andomisation code was held by pharmacy" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Pettila 2010.
| Methods | Randomised clinical trial | |
| Participants | Country: Finland Number randomised: 32 Postrandomisation dropouts: 0 (0%) Revised sample size: 32 Average age: 45 years Women: 3 (9.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 32 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: activated protein C (n = 16): drotrecogin alpha activated 24 μg/kg/h for 96 h Group 2: placebo (n = 16) | |
| Outcomes | Mortality, hospital stay Follow‐up: not stated (probably 2 weeks) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he code for study medication was concealed using sealed envelopes." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "Eli Lilly in part provided the study drug for this investigator‐initiated study". |
| Other bias | Low risk | Comment: no other risk of bias |
Plaudis 2010.
| Methods | Randomised clinical trial | |
| Participants | Country: Latvia Number randomised: 90 Postrandomisation dropouts: not stated Revised sample size: 58 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 58 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute severe pancreatitis | |
| Interventions | Group 1: probiotics (n = 30): 4 bioactive lactic acid bacteria Group 2: no intervention (n = 28) Both groups received prebiotics (an intervention not of interest for this review) | |
| Outcomes | None of the outcomes of interest were reported. Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Poropat 2015.
| Methods | Randomised clinical trial | |
| Participants | Country: Croatia Number randomised: 43 Postrandomisation dropouts: 0 (0%) Revised sample size: 43 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 23): imipenem 500 mg IV 3 times daily for 10 days Group 2: no intervention (n = 24) | |
| Outcomes | Mortality, serious adverse events, adverse events, infected pancreatic necrosis, and organ failure Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Rokke 2007.
| Methods | Randomised clinical trial | |
| Participants | Country: Norway Number randomised: 73 Postrandomisation dropouts: 0 (0%) Revised sample size: 73 Average age: 58 years Women: 24 (32.9%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 73 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 73 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 36): imipenem 0.5 g every 8 h for 5‐7 days Group 2: no intervention (n = 37) | |
| Outcomes | Mortality, adverse events, requirement for surgery, organ failure, infected pancreatic necrosis, hospital stay, ICU stay Follow‐up: not stated (probably 2 weeks) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "[t]he study was unblinded to all attending physicians". |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "[t]he study was unblinded to all attending physicians". |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[w]e are grateful to the pharmaceutical company MSD for economic support in organizing meetings for the Steering Committee". |
| Other bias | Low risk | Comment: no other risk of bias |
Sainio 1995.
| Methods | Randomised clinical trial | |
| Participants | Country: Finland Number randomised: 60 Postrandomisation dropouts: 0 (0%) Revised sample size: 60 Average age: 41 years Women: 7 (11.7%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 60 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with alcohol‐induced necrotising pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 30): cefuroxime 1.5 g IV 3 times daily continued until clinical recovery and fall to normal level of C‐reactive protein, after which oral administration of 250 mg twice daily until 14 days Group 2: no intervention (n = 30) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, sepsis, hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Sateesh 2009.
| Methods | Randomised clinical trial | |
| Participants | Country: India Number randomised: 56 Postrandomisation dropouts: 3 (5.4%) Revised sample size: 53 Average age: 39 years Women: 33 (62.3%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 10 (18.9%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: antioxidants (n = 23): vitamin C 500 mg once daily, N‐acteyl cysteine 200 mg 3 times daily, Antoxyl Forte 1 capsule 3 times daily); duration not stated Group 2: no intervention (n = 30) | |
| Outcomes | Mortality, adverse events, organ failure, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: did not receive allocated treatment, discontinued medication | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "according to a computer generated random number table" |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "[t]he study was unblinded". |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "[t]he study was unblinded". |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Sharma 2011.
| Methods | Randomised clinical trial | |
| Participants | Country: India Number randomised: 50 Postrandomisation dropouts: 0 (0%) Revised sample size: 50 Average age: 41 years Women: 27 (54%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 28 (56%) Moderate pancreatitis: not stated Severe pancreatitis: 22 (44%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: probiotics (n = 24): 2.5 billion bacteria per sachet and 25 mg of fructo‐oligosaccharide every day for 7 days Group 2: placebo (n = 26) | |
| Outcomes | Hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Quote: "[t]he method of allocation concealment was sequentially numbered sealed opaque envelopes technique". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "[t]he authors disclose that Alkem provided the probiotics and placebo on complimentary basis." |
| Other bias | Low risk | Comment: no other risk of bias |
Sillero 1981.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 60 Postrandomisation dropouts: not stated Revised sample size: 60 Average age: 52 years Women: 36 (60%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: cimetidine (n = 30): 1200 mg IV for 4 days followed by 1000 mg oral for 10 days Group 2: placebo (n = 30) | |
| Outcomes | Serious adverse events, adverse events, requirement for surgery Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "table of random numbers" |
| Allocation concealment (selection bias) | Low risk | Quote: "sealed envelopes" |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: although a placebo was used, it was not clear blinding was performed. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available |
| Other bias | Low risk | Comment: no other risk of bias |
Siriwardena 2007.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 43 Postrandomisation dropouts: 0 (0%) Revised sample size: 43 Average age: 67 years Women: 28 (65.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: antioxidants (n = 22) selenium started with 1000 mg and then tapered to 200 mg/day for a total duration of 7 days; vitamin C started with 2000 mg and then tapered to 1000 mg/day for a total duration of 7 days; N‐acetyl cysteine started with 300 mg and then tapered to 75 mg/day for a total duration of 7 days Group 2: placebo (n = 21) | |
| Outcomes | Mortality, serious adverse events, organ failure, hospital stay, ICU stay Follow‐up: until discharge |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "random number generation" Comment: probably computer‐generated |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he pharmacy administered the randomisation and storage of therapeutics for all participating centres". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Unclear risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "the costs of antioxidants and placebo were met by Pharmanord UK". |
| Other bias | Low risk | Comment: no other risk of bias |
Spicak 2002.
| Methods | Randomised clinical trial | |
| Participants | Country: Czech Republic Number randomised: 63 Postrandomisation dropouts: not stated Revised sample size: 63 Average age: 55 years Women: 25 (39.7%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 63 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 33): metronidazole 500 mg 3 times daily and ciprofloxacin 200 mg twice daily for 2 weeks Group 2: no intervention (n = 30) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, infected pancreatic necrosis, hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Spicak 2003.
| Methods | Randomised clinical trial | |
| Participants | Country: Czech Republic Number randomised: 41 Postrandomisation dropouts: not stated Revised sample size: 41 Average age: 58 years Women: 10 (24.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%). Moderate pancreatitis: 0 (0%) Severe pancreatitis: 41 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 20): meropenem 0.5 mg 3 times daily for 10 days Group 2: no intervention (n = 21) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, infected pancreatic necrosis, hospital stay Follow‐up: not stated |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | This information was not available. |
| Allocation concealment (selection bias) | Unclear risk | This information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | This information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | This information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | This information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | This information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Storck 1968.
| Methods | Randomised clinical trial | |
| Participants | Country: Sweden Number randomised: 43 Postrandomisation dropouts: not stated Revised sample size: 43 Average age: 59 years Women: 28 (65.1%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: aprotinin (n = 21), first half of the trial ‐ 50,000 to 100,000 units per day and then dose doubled for an average of 12 days Group 2: placebo (n = 22) | |
| Outcomes | Mortality Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[s]ealed envelopes" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Trapnell 1974.
| Methods | Randomised clinical trial | |
| Participants | Country: UK Number randomised: 105 Postrandomisation dropouts: not stated Revised sample size: 105 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: aprotinin (n = 53), 200,000 units IV stat followed by 200,000 units IV 4 times daily for 5 days Group 2: placebo (n = 52) | |
| Outcomes | Mortality Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "random numbers" |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he envelopes of allotment were placed in a recognized position in each hospital together with the packs of Trasylol". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | High risk | Quote: "[w]e are particularly indebted to Dr Brian Allen of Bayer Pharmaceuticals for the supplies of Trasylol and the preparation of the A and B ampoules". |
| Other bias | Unclear risk | Comment: no other risk of bias |
Tykka 1985.
| Methods | Randomised clinical trial | |
| Participants | Country: Finland Number randomised: 64 Postrandomisation dropouts: 0 (0%) Revised sample size: 64 Average age: 51 years Women: 23 (35.9%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: EDTA (n = 33), dose and duration not reported
Group 2: placebo (n = 31) Follow‐up: not stated (probably until discharge) |
|
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery | |
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[w]e are also grateful for the drugs and support from Sinclair Pharmaceutical Limited, England." Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Uhl 1999.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 302 Postrandomisation dropouts: 0 (0%) Revised sample size: 302 Average age: 50 years Women: 104 (34.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: 108 (35.8%) Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: octreotide (n = 199), 100 μg or 200 μg (randomised) SC 3 times daily for 7 days Group 2: placebo (n = 103) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, sepsis, hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Low risk | Quote: "[t]he packages were used sequentially as the patients were enrolled in the study". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[t]he preparation, randomisation, and delivery of the study medication, as well as the monitoring of the study centres by checking the information in the CRFs, were carried out by Novartis (formerly Sandoz), Nuremberg (Germany)". |
| Other bias | Low risk | Comment: no other risk of bias |
Usadel 1985.
| Methods | Randomised clinical trial | |
| Participants | Country: Germany Number randomised: 77 Postrandomisation dropouts: not stated Revised sample size: 77 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: somatostain (n = 36), 250 ng/h for 7 days Group 2: placebo (n = 41) | |
| Outcomes | Mortality Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Valderrama 1992.
| Methods | Randomised clinical trial | |
| Participants | Country: Spain Number randomised: 105 Postrandomisation dropouts: 5 (4.8%) Revised sample size: 100 Average age: 57 years Women: 53 (53%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: gabexate mesilate (n = 51), 12 mg/kg/day continuous IV for 4‐12 days based on disappearance of abdominal pain or requirement for surgery Group 2: placebo (n = 49) | |
| Outcomes | Mortality, serious adverse events, adverse events, sepsis Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: protocol violations | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "computer generated" |
| Allocation concealment (selection bias) | Low risk | Quote: "consecutively numbered boxes containing FOY or placebo" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | High risk | Quote: "[t]he authors thank Laboratorio Dr Esteve SA for supplies of gabexate mesylate (FOY)". |
| Other bias | Low risk | Comment: no other risk of bias |
Vege 2015.
| Methods | Randomised clinical trial | |
| Participants | Country: USA Number randomised: 28 Postrandomisation dropouts: not stated Revised sample size: 28 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: antioxidant (n = 14): pentoxifylline 400 mg oral 3 times daily for 3 days Group 2: placebo (n = 14) | |
| Outcomes | Mortality, serious adverse events, organ failure, hospital stay, ICU stay Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Wang 2011.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 24 Postrandomisation dropouts: not stated Revised sample size: 24 Average age: 46 years Women: 15 (62.5%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 24 (100%). Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis. | |
| Interventions | Group 1: thymosin alpha (n = 12), 3.2 mg twice daily for 7 days Group 2: placebo (n = 12) | |
| Outcomes | Mortality, hospital stay, ICU stay Follow‐up: 1 month |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Wang 2013a.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 183 Postrandomisation dropouts: not stated Revised sample size: 183 Average age: 42 years Women: 89 (48.6%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 159 (86.9%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: somatostatin plus ulinastatin (n = 62) Group 2: somatostatin (n = 61) Group 3: no intervention (n = 60) Somatostatin: 250 μg/h IV for 10 days. Ulinastatin: 10,000 units IV twice daily for 10 days | |
| Outcomes | Mortality, serious adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Low risk | Quote: "[t]he authors have no direct relationship with any of the companies mentioned in this article, either by employment or by receiving research grants". |
| Other bias | Low risk | Comment: no other risk of bias |
Wang 2013b.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 354 Postrandomisation dropouts: not stated Revised sample size: 354 Average age: not stated Women: not stated Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with predicted severe acute pancreatitis | |
| Interventions | Group 1: octreotide plus NSAID (n = not reported) Group 2: octreotide (n = not reported) Octreotide: 50 μg/h for first 3 days followed by 25 μg/h for next 4 days NSAID: celecoxib 200 mg twice daily for 7 days | |
| Outcomes | None of the outcomes of interest were reported. Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Wang 2013c.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 372 Postrandomisation dropouts: not stated Revised sample size: 372 Average age: 45 years Women: 174 (46.8%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 0 (0%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria: people with alcohol dependence |
|
| Interventions | Group 1: octreotide (n = 157), 50 μg/h for first 3 days followed by 25 μg/h for next 4 days or 25 μg/h for 7 days (randomised) Group 2: no intervention (n = 79) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, requirement for endoscopic or radiological drainage, organ failure, hospital stay Follow‐up: some outcomes were measured on 8th day and others at 1 month |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "computer‐generated randomization numbers" |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Quote: "[t]he physicians and nurses who managed the patients were blinded so that they did not know the patient has been allocated to and what treatment they had received". Comment: there is no mention of participant blinding. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[t]he physicians and nurses who managed the patients were blinded so that they did not know the patient has been allocated to and what treatment they had received". |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Low risk | Quote: "[t]his study was supported by a Key Grant #30330270 from the Natural Science Fund of China and the National Ministry of Health Fund for the Public Welfare 2‐13". |
| Other bias | Low risk | Comment: no other risk of bias |
Wang 2016.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 492 Postrandomisation dropouts: not stated Revised sample size: 492 Average age: 41 years Women: 238 (48.4%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 492 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with severe acute pancreatitis Exclusion criteria
|
|
| Interventions | Group 1: somatostatin plus ulinastatin plus gabexate (n = 116) Group 2: somatostatin plus ulinastatin (n = 124) Group 3: somatostatin plus gabexate (n = 130) Group 4: somatostatin (n = 122) Somatostatin: 3 mg IV for 10 days Ulinastatin: 10,000 units IV twice daily for 10 days Gabexate: 0.1 g IV 3 times daily for 10 days | |
| Outcomes | Mortality, adverse events, organ failure, length of hospital stay Follow‐up: not stated (probably until discharge) |
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "[a]ccording to a computerized random number generation . . ." |
| Allocation concealment (selection bias) | Low risk | Quote: "sealed envelopes" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "[t]his was a prospective and double‐blind study" Comment: a placebo was used to achieve blinding. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "[t]his was a prospective and double‐blind study" Comment: a placebo was used to achieve blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: there were no postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Low risk | Quote: "[t]his work was supported by National Natural Science Foundation of China, China (81360080, 81071594) and the Science Foundation of Science and Technology Hall of Jiangxi Province, China (20091391308000)." |
| Other bias | Low risk | Comment: no other risk of bias |
Xia 2014.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 140 Postrandomisation dropouts: not stated Revised sample size: 140 Average age: 43 years Women: 48 (34.3%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: 140 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
|
|
| Interventions | Group 1: somatostatin (3 mg IV twice daily for 7 days) plus omeprazole (40 mg IV twice daily for 7 days) (n = 70) Group 2: no intervention (n = 70) | |
| Outcomes | Mortality, serious adverse events, adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Xue 2009.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 59 Postrandomisation dropouts: 3 (5.1%) Revised sample size: 56 Average age: 48 years Women: 28 (50%) Acute interstitial oedematous pancreatitis: 0 (0%) Necrotising pancreatitis: 56 (100%) Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 56 (100%) Persistent organ failure: not stated Infected pancreatitis: 0 Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: antibiotics (n = 29): imipenem‐cilastatin 0.5 g every 8 h for 7‐14 days Group 2: no intervention (n = 27) | |
| Outcomes | Mortality, serious adverse events, adverse events, requirement for surgery, hospital stay Follow‐up: 1 month |
|
| Notes | Reasons for postrandomisation dropouts: death after starting treatment, transferred to operation | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "computer‐derived random number sequence" |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | Low risk | Comment: mortality and adverse events were reported. |
| For profit‐bias | Low risk | Quote: "[w]e thank Sichuan Province Science and Technology Tackling Key Project (no. 05SG011‐021‐1) for providing financial support for the trial and the publication of the paper". |
| Other bias | Low risk | Comment: no other risk of bias |
Yang 1999.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 48 Postrandomisation dropouts: not stated Revised sample size: 48 Average age: 45 years Women: 26 (54.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: not stated Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria: people with acute pancreatitis | |
| Interventions | Group 1: somatostatin (n = 25), 250 μg/h for 3‐4 days Group 2: no intervention (n = 23) | |
| Outcomes | Serious adverse events, adverse events Follow‐up: not stated (probably until discharge) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
Yang 2012.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 163 Postrandomisation dropouts: 6 (3.7%) Revised sample size: 157 Average age: 46 years Women: 71 (45.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 157 (100%) Moderate pancreatitis: not stated Severe pancreatitis: not stated Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: octreotide (n = 80), 50 μg/h for 3 days Group 2: no intervention (n = 77) | |
| Outcomes | Mortality, hospital stay Follow‐up: 1 month |
|
| Notes | Reasons for postrandomisation dropouts: loss to follow‐up; lack of data | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "computer‐generated randomization numbers" |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: there were postrandomisation dropouts. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Low risk | Quote: "[t]his study was supported by Key Grant #30330270 of the Natural Science Fund of China and the National Ministry of Health Fund for Public Welfare 2‐13." |
| Other bias | Low risk | Comment: no other risk of bias |
Zhu 2014.
| Methods | Randomised clinical trial | |
| Participants | Country: China Number randomised: 39 Postrandomisation dropouts: not stated Revised sample size: 39 Average age: 43 years Women: 18 (46.2%) Acute interstitial oedematous pancreatitis: not stated Necrotising pancreatitis: not stated Mild pancreatitis: 0 (0%) Moderate pancreatitis: 0 (0%) Severe pancreatitis: 39 (100%) Persistent organ failure: not stated Infected pancreatitis: not stated Inclusion criteria
Exclusion criteria
|
|
| Interventions | Group 1: probiotics (n = 20), 2 tablets twice daily for 14 days (Japanese preparation) Group 2: placebo (n = 19) | |
| Outcomes | Serious adverse events, adverse events, requirement for endoscopic or radiological drainage, infected pancreatic necrosis Follow‐up: not stated (probably 2 weeks) |
|
| Notes | Reasons for postrandomisation dropouts: not stated | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: this information was not available. |
| Allocation concealment (selection bias) | Unclear risk | Comment: this information was not available. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "double‐blind" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "double‐blind" |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: this information was not available. |
| Selective reporting (reporting bias) | High risk | Comment: either mortality or adverse events were not reported. |
| For profit‐bias | Unclear risk | Comment: this information was not available. |
| Other bias | Low risk | Comment: no other risk of bias |
ERCP: endoscopic retrograde cholangiopancreatography; ICU: intensive care unit; IU: international unit; IV: intravenous; KIU: kallikrein inhibitor units; MRC: Medical Research Council (1 MRC = 1 IU); PR: per rectum; SC: subcutaneous.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Akzhigitov 1968 | Not an RCT |
| Akzhigitov 1969 | Not an RCT |
| Al‐Leswas 2013a | Comparison of 2 different antioxidants |
| Al‐Leswas 2013b | Comparison of 2 different antioxidants |
| Al‐Leswas 2013c | Comparison of 2 different antioxidants |
| Al‐Leswas 2013d | Comparison of 2 different antioxidants |
| Al‐Leswas 2013e | Comparison of 2 different antioxidants |
| Al‐Leswas 2013f | Comparison of 2 different antioxidants |
| Al‐Leswas 2013g | Comparison of 2 different antioxidants |
| Amundsen 1972 | Not conducted in humans |
| Andersson 2008 | Not a primary research study (commentary) |
| Baden 1967 | Quasi‐RCT (allocation based on birth date) comparing 2 different preparations of aprotinin |
| Baden 1969 | Quasi‐RCT (allocation based on birth date) comparing 2 different preparations of aprotinin |
| Bai 2013 | Not an RCT |
| Bassi 1998 | Comparison of 2 different antibiotic regimens |
| Beechey‐Newman 1991 | Not an RCT |
| Beechey‐Newman 1993 | Not an RCT |
| Beger 2001 | Not a primary research study (commentary) |
| Bender 1992 | Not an RCT |
| Binder 1993 | Comparison of different doses of octreotide |
| Binder 1994 | Comparison of different doses of octreotide |
| Brown 2004 | Not a primary research study (editorial) |
| Buchler 1988 | Not an RCT |
| Cameron 1979 | Quasi‐randomised study (allocation by patient number) |
| Cheng 2008 | There was no control group for pharmacological intervention |
| Cullimore 2008 | Not a primary research study (letter to editor) |
| Curtis 1997 | Not a primary research study (review) |
| D'Amico 1990 | Not an RCT |
| Da Silvereira 2002 | Not a primary research study (commentary) |
| De Vries 2007 | Not a primary research study (systematic review) |
| Dikkenberg 2008 | Not a primary research study (commentary) |
| Dreiling 1977 | Not an RCT |
| Du 2002 | Comparison of 2 doses of vitamin C |
| Du 2003 | Comparison of 2 doses of vitamin C |
| Dürr 1985 | Quasi‐RCT (allocation by alternation) |
| Freise 1985 | Not an RCT |
| Friess 1994 | Not a primary research study (review) |
| Gabryelewicz 1968 | Not in humans |
| Gabryelewicz 1976 | Not an RCT |
| Gao 2015b | Not a pharmacological intervention |
| Garcia 2005 | Comparison of 2 variations of probiotics |
| Gostishchev 1977 | Not a primary research study (review) |
| Guo 2013 | Comparison of different doses of octreotide |
| Hajdu 2012 | Variations in nutritional supplementation |
| Harinath 2002 | Prophylactic intervention (not in people with acute pancreatitis) |
| Hart 2008 | Not a primary research study (review) |
| He 2004 | Not a pharmacological intervention |
| Helton 2001 | Not a primary research study (comment) |
| Hoekstra 2008 | Not a primary research study (letter to editor) |
| Holub 1974 | Not a primary research study (letter to editor) |
| Howard 2007 | Not a primary research study (editorial) |
| Howes 1975 | Quasi‐RCT (allocation by hospital number) |
| Huang 2008 | Variations in different types of nutritional supplementation |
| Issekutz 2002 | No suitable control (3 groups were: probiotics + fibre versus inactivated lactobacilli + fibre versus standard nutrition; it is not possible to obtain the effect estimate of probiotics alone from this comparison) |
| Ivanov 2002 | Not an RCT |
| Jiang 1988 | Not an RCT |
| Karakan 2007 | Not a pharmacological intervention (fibre supplementation only) |
| Karakoyunlar 1999 | Not an RCT |
| Karavanov 1966 | Not an RCT |
| Lasztity 2005a | Variations in fatty acids used in enteral nutrition |
| Lasztity 2005b | Variations in fatty acids used in enteral nutrition |
| Lasztity 2006 | Variations in fatty acids used in enteral nutrition |
| Lata 1998 | Not an RCT |
| Lata 2010 | This started as a RCT but was converted to a cohort study after publication of negative results |
| Lim 2015 | Not a primary research study (review) |
| Lu 2006 | Not a pharmacological intervention (variations in parenteral nutrition) |
| Lu 2008 | Intervention includes a non‐pharmacological treatment in addition to antioxidant |
| Manes 2003 | Comparison of 2 different antibiotics |
| Manes 2006 | Comparison of 2 different antibiotic regimens |
| McClave 2009 | Not a primary research study (editorial) |
| Mercadier 1973 | Not an RCT |
| Niu 2014 | Comparison of 2 different fats |
| Pearce 2006 | Variations in composition of enteral feeds |
| Pederzoli 1995 | Not primary research (review) |
| Pezzilli 1997 | Comparison of two doses of gabexate mesilate |
| Pezzilli 1999 | Comparison of 2 doses of gabexate mesilate |
| Pezzilli 2001 | Comparison of 2 doses of gabexate mesilate |
| Piascik 2010 | In addition to the difference in the groups in terms of whether the patients received protease inhibitor, the antibiotic regimen differed between the groups |
| Plaudis 2012 | Not an RCT |
| Rahman 2003 | Not a primary research study (letter to editor) |
| Ranson 1976 | Not an RCT |
| Reddy 2008 | Not a primary research study (letter to editor) |
| Santen 2008 | Not primary research (letter to editor) |
| Singer 1966 | No mention about randomisation |
| Skyring 1965 | No mention about randomisation |
| Tanaka 1979 | There were 2 trials reported in this publication. Of these, 1 was a quasi‐RCT (alternate allocation) and it was not clear whether the second trial was an RCT |
| Tang 2005 | Only the control group received Chinese medicines |
| Tang 2007 | Not an RCT |
| Ukai 2015 | Not a primary research study (review) |
| Usadel 1980 | Not a primary research study (letter to editor) |
| Venkatesan 2008 | Not a primary research study (commentary) |
| Villatoro 2010 | Not primary research (review) |
| Wang 2008 | Variations in composition of parenteral nutrition |
| Wang 2009 | Variations in composition of parenteral nutrition |
| Weismann 2010 | Not a primary research study (commentary) |
| Wyncoll 1998 | Not a primary research study (letter to editor) |
| Xiong 2009 | Variations in parenteral nutrition |
| Xu 2012 | Variations in parenteral nutrition |
| Yang 2008a | Not an RCT |
| Yang 2008b | Variations in total parenteral nutrition |
| Yang 2009 | Chinese medicines were given to the control group but not the intervention group |
| Zapater 2000 | The co‐interventions in the groups varied apart from the drug being evaluated (nasogastric suction was used only in the control group) |
RCT = randomised controlled trial
Characteristics of studies awaiting assessment [ordered by study ID]
Hansen 1966.
| Methods | Awaiting full text |
| Participants | — |
| Interventions | — |
| Outcomes | — |
| Notes | — |
Perez 1980.
| Methods | Awaiting full text |
| Participants | — |
| Interventions | — |
| Outcomes | — |
| Notes | — |
Characteristics of ongoing studies [ordered by study ID]
ChiCTR‐IPR‐16008301.
| Trial name or title | The effect of proton pump inhibitors on acute pancreatitis‐‐a randomly prospective control study |
| Methods | Randomised controlled trial |
| Participants | Adults with acute pancreatitis |
| Interventions | Proton pump inhibitor (omeprazole) versus placebo |
| Outcomes | Duration of hospital stay, gastrointestinal bleeding, and hospital costs |
| Starting date | September 2016 |
| Contact information | Xiao Ma (mxiao_9101@163.com) |
| Notes | — |
EUCTR2014‐004844‐37‐ES.
| Trial name or title | Trial of indomethacin in pancreatitis |
| Methods | Randomised controlled trial |
| Participants | Adults with acute pancreatitis |
| Interventions | Non‐steroidal anti‐inflammatory drugs (indomethacin) versus placebo |
| Outcomes | Mortality and organ failure |
| Starting date | May 2015 |
| Contact information | Enrique de Madaria Pascual (madaria@hotmail.com) |
| Notes | ChiCTR‐IPR‐16008301, NCT02692391 |
NCT01132521.
| Trial name or title | Ulinastatin in severe acute pancreatitis |
| Methods | Randomised controlled trial |
| Participants | Adults with severe acute pancreatitis |
| Interventions | Ulinastatin versus placebo |
| Outcomes | mortality, organ failure, requirement for additional invasive intervention, hospital stay, intensive care unit stay |
| Starting date | June 2010 |
| Contact information | Chunyou Wang (Wuhan Union Hospital, China) |
| Notes | The study is currently suspended. |
NCT02025049.
| Trial name or title | DP‐b99 in the treatment of acute high‐risk pancreatitis |
| Methods | Randomised controlled trial |
| Participants | Adults with predicted severe acute pancreatitis |
| Interventions | DP‐b99 versus placebo |
| Outcomes | Complications |
| Starting date | December 2013 |
| Contact information | Gilad Rosenberg (Wuhan Union Hospital, China) |
| Notes | The University Hospital Brno, Gastroenterology Clinic, Brno, Czech Republic, 62500 |
NCT02212392.
| Trial name or title | Comparing the outcome in patients of acute pancreatitis, with and without prophylactic antibiotics |
| Methods | Randomised controlled trial |
| Participants | Adults with acute pancreatitis |
| Interventions | Antibiotics (meropenem) versus no intervention |
| Outcomes | Infections and hospital stay |
| Starting date | Jan 2013 |
| Contact information | Fazal H Shah (Benazir Bhutto Hospital, Rawalpindi, Punjab, Pakistan, 46000) |
| Notes | — |
NCT02692391.
| Trial name or title | A randomized controlled pilot trial of indomethacin in acute pancreatitis |
| Methods | Randomised controlled trial |
| Participants | Adults with acute pancreatitis |
| Interventions | Non‐steroidal anti‐inflammatory drugs (indomethacin) versus placebo |
| Outcomes | Mortality and organ failure |
| Starting date | April 2014 |
| Contact information | Georgios I Papachristou (papachri@pitt.edu) |
| Notes | — |
NCT02885441.
| Trial name or title | Treatment of acute pancreatitis with ketorolac |
| Methods | Randomised controlled trial |
| Participants | Adults with predicted severe acute pancreatitis |
| Interventions | Non‐steroidal anti‐inflammatory drugs (ketorolac) versus placebo |
| Outcomes | New onset organ failure, pancreatic necrosis, and duration of hospital stay |
| Starting date | September 2016 |
| Contact information | Shaahin Shahbazi (mdkabe@gmail.com) |
| Notes | — |
Differences between protocol and review
We did not combine somatostatin and somatostatin analogues. This is to avoid further clinical heterogeneity.
We reported sepsis separately under serious adverse events due to its importance as an important clinical outcome.
Contributions of authors
EM selected studies and extracted the data for more than half the studies identified by screening and completed the tables detailing the characteristics of included and excluded studies. FF helped EM with data extraction. RK selected studies and extracted the data for the remaining studies. AB screened the references. SP and BRD critically commented on the review. KG screened the references, selected studies, extracted the data, analysed the data, and wrote the review.
Sources of support
Internal sources
University College London, UK.
External sources
National Institute for Health Research, UK.
Declarations of interest
This report is independent research funded by the National Institute for Health Research (NIHR Cochrane Programme Grants, 13/89/03 ‐ Evidence‐based diagnosis and management of upper digestive, hepato‐biliary, and pancreatic disorders). The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health.
New
References
References to studies included in this review
Abraham 2013 {published data only}
- Abraham P, Rodriques J, Moulick N, Dharap S, Chafekar N, Verma PK, et al. Efficacy and safety of intravenous ulinastatin versus placebo along with standard supportive care in subjects with mild or severe acute pancreatitis. Journal of the Association of Physicians of India 2013;61(8):535‐8. [PubMed] [Google Scholar]
Balldin 1983 {published data only}
- Balldin G, Borgström A, Genell S, Ohlsson K. The effect of peritoneal lavage and aprotinin in the treatment of severe acute pancreatitis. Research in Experimental Medicine. Zeitschrift für die Gesamte Experimentelle Medizin Einschliesslich Experimenteller Chirurgie 1983;183(3):203‐13. [DOI] [PubMed] [Google Scholar]
Bansal 2011 {published data only}
- Bansal D, Bhalla A, Bhasin DK, Pandhi P, Sharma N, Rana S, et al. Safety and efficacy of vitamin‐based antioxidant therapy in patients with severe acute pancreatitis: a randomized controlled trial. Saudi Journal of Gastroenterology: Official Journal of the Saudi Gastroenterology Association 2011;17(3):174‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Barreda 2009 {published data only}
- Barreda L, Targarona Modena J, Milian W, Portugal J, Sequeiros J, Pando E, et al. Is the prophylactic antibiotic therapy with imipenem effective for patients with pancreatic necrosis? [Es la antibioticoterapia profilactica con Imipenem efectiva en los pacientes con necrosis pancreatica?]. Acta Gastroenterologica Latinoamericana 2009;39(1):24‐9. [PubMed] [Google Scholar]
Berling 1994 {published data only}
- Berling R, Borgstrom A, Ohlsson K. Peritoneal lavage with aprotinin in patients with severe acute pancreatitis: effects on plasma and peritoneal levels of trypsin and leukocyte proteases and their major inhibitors. International Journal of Pancreatology 1998;24(1):9‐17. [DOI] [PubMed] [Google Scholar]
- Berling R, Genell IS, Ohlsson K. High‐dose intraperitoneal aprotinin treatment of acute severe pancreatitis: a double‐blind randomized multi‐center trial. Journal of Gastroenterology 1994;29(4):479‐85. [DOI] [PubMed] [Google Scholar]
- Berling R, Ohlsson K. Effects of high‐dose intraperitoneal aprotinin treatment on complement activation and acute phase response in acute severe pancreatitis. Journal of Gastroenterology 1996;31(5):702‐9. [DOI] [PubMed] [Google Scholar]
Besselink 2008 {published data only}
- Besselink MG, Santvoort HC, Boermeester MA, Fischer K, Renooij W, Smet MB, et al. Intestinal barrier dysfunction in a randomised placebo‐controlled trial of probiotic prophylaxis in acute pancreatitis. European Journal of Gastroenterology & Hepatology 2009;21(3):A2‐A. [Google Scholar]
- Besselink MG, Santvoort HC, Buskens E, Akkermans LM, Gooszen HG, Dutch Acute Pancreatitis Study Group. Probiotic prophylaxis in predicted severe acute pancreatitis ‐ reply. Lancet 2008;372(9633):114. [Google Scholar]
- Besselink MG, Santvoort HC, Buskens E, Boermeester MA, Goor H, Timmerman HM, et al. Dutch Acute Pancreatitis Study Group. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial. Erratum in: Lancet 2008;371(9620):1246. Lancet 2008;371(9613):651‐9. [DOI] [PubMed] [Google Scholar]
- Besselink MG, Santvoort HC, Buskens E, Boermeester MA, Goor H, Timmerman HM, et al. Probiotic prophylaxis in patients with predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial [Probioticaprofylaxe bij voorspeld ernstige acute pancreatitis: een gerandomiseerde, dubbelblinde, placebogecontroleerde trial.]. Nederlands Tijdschrift voor Geneeskunde 2008;152(12):685‐96. [PubMed] [Google Scholar]
- Besselink MG, Santvoort HC, Buskens E, Boermeester MA, Goor H, Timmerman HM, et al. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial. European Journal of Gastroenterology & Hepatology 2009;21(3):A8‐9. [Google Scholar]
- Besselink MG, Santvoort HC, Buskens E, Boermeester MA, Goor H, Timmerman HM, et al. Dutch Acute Pancreatitis Study Group. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial. Lancet 2008;371(9613):651‐9. [DOI] [PubMed] [Google Scholar]
- Besselink MG, Santvoort HC, Renooij W, Smet MB, Boermeester MA, Fischer K, et al. Intestinal barrier dysfunction in a randomized trial of a specific probiotic composition in acute pancreatitis. Annals of Surgery 2009;250(5):712‐9. [DOI] [PubMed] [Google Scholar]
- Besselink MGH, Timmerman HM, Buskens E, Nieuwenhuijs VB, Akkermans LMA, Gooszen HG. Probiotic prophylaxis in patients with predicted severe acute pancreatitis (propatria): design and rationale of a double‐blind, placebo controlled randomised multicenter trial [ISCRTN 38327949]. BMC Surgery 2004;4:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Besselink MGH, Santvoort HC, Buskens E, Boermeester MA, Dutch Acute Pancreatitis Study Group. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial. Lancet 2008;371(9620):1246. [DOI] [PubMed] [Google Scholar]
- The Editors of The Lancet. Expression of concern‐probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial. Lancet 2010;375(9718):875‐6. [DOI] [PubMed] [Google Scholar]
Birk 1994 {published data only}
- Birk D, Schoenberg MH, Adler G, Beger HG. Oxidative stress in acute‐pancreatitis ‐ results of a prospective randomized clinical pilot‐study. Gastroenterology 1994;106(4):A286‐A. [Google Scholar]
Bredkjaer 1988 {published data only}
- Bredkjaer HE, Bülow S, Ebbehøj N, Friis J, Lindewald H, Rasmussen SG, et al. Treatment of acute pancreatitis with indomethacin. A controlled study of the formation of pseudocysts and their subsequent course [Indometacinbehandling ved pancreatitis acuta. En kontrolleret undersogelse af forekomst og forlob af pseudocystedannelse]. Ugeskrift for Laeger 1988;150(47):2902‐3. [PubMed] [Google Scholar]
Buchler 1993 {published data only}
- Buchler M, Malfertheiner P, Uhl W, Scholmerich J, Stockmann F, Adler G, et al. Gabexate mesilate in human acute pancreatitis. German pancreatitis study group. Gastroenterology 1993;104(4):1165‐70. [DOI] [PubMed] [Google Scholar]
- Buchler M, Malfertheiner P, Uhl W, Scholmerich J, Stockmann F, Adler G, et al. Gabexate‐mesilate in human acute pancreatitis: results from the German multicenter trial with 4 g/day [abstract]. Digestion 1990;46(3):130. [Google Scholar]
Chen 2000 {published data only}
- Chen HM, Chen JC, Hwang TL, Jan YY, Chen MF. Prospective and randomized study of gabexate mesilate for the treatment of severe acute pancreatitis with organ dysfunction. Hepato‐Gastroenterology 2000;47(34):1147‐50. [PubMed] [Google Scholar]
Chen 2002a {published data only}
- Chen SY, Wang JY. Ulinastatin in the treatment of acute pancreatitis: a multicenter clinical trial. Chinese Journal of Digestive Diseases 2002;3(2):70‐4. [Google Scholar]
Chen 2002b {published data only}
- Chen SY, Wang JY. Ulinastatin in the treatment of acute pancreatitis: a multicenter clinical trial. Chinese Journal of Digestive Diseases 2002;3(2):70‐4. [Google Scholar]
Choi 1989 {published data only}
- Choi TK, Mok F, Zhan WH, Fan ST, Lai EC, Wong J. Somatostatin in the treatment of acute pancreatitis: a prospective randomised controlled trial. Gut 1989;30(2):223‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Chooklin 2007 {published data only}
- Chooklin S, Vatseba R. N‐acetylcysteine and dexamethasone in the prevention of respiratory complications in acute pancreatitis [abstract]. European Respiratory Journal 2007;30(Suppl 51):51s [E470]. [Google Scholar]
Debas 1980 {published data only}
- Debas HT, Hancock RJ, Soon‐Shiong P. Glucagon therapy in acute pancreatitis: prospective randomized double‐blind study. Canadian Journal of Surgery 1980;23(6):578‐80. [PubMed] [Google Scholar]
Delcenserie 1996 {published data only}
- Delcenserie R, Yzet T, Ducroix JP. Prophylactic antibiotics in treatment of severe acute alcoholic pancreatitis. Pancreas 1996;13(2):198‐201. [PubMed] [Google Scholar]
Delcenserie 2001 {published data only}
- Delcenserie R, Dellion‐Lozinguez MP, Pagenault M, Hastier P, Yzet T, Dupas JL, et al. Prophylactic ciprofloxacin treatment in acute necrotizing pancreatitis: a prospective randomized multicenter clinical trial. Gastroenterology 2001;120(5):A25‐A. [Google Scholar]
Dellinger 2007 {published data only}
- Dellinger EP, Tellado JM, Soto NE, Ashley SW, Barie PS, Dugernier T, et al. Early antibiotic treatment for severe acute necrotizing pancreatitis: a randomized, double‐blind, placebo‐controlled study. Annals of Surgery 2007;245(5):674‐83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dellinger EP, Tellado JM, Soto NE, Ashley SW, Barie PS, Dugernier T, et al. Re: Early antibiotic treatment for severe acute necrotizing pancreatitis ‐ reply. Annals of Surgery 2008;247(2):394‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dürr 1978 {published data only}
- Dürr HK, Kunz R, Zelder O. The treatment of acute pancreatitis with glucagon: a double blind study. Archives Francaises des Maladies de l'Appareil Digestif 1975;64(7 Suppl 4):349. [Google Scholar]
- Dürr HK, Maroske D, Zelder O, Bode Ch J. Glucagon therapy in acute pancreatitis. Report of a double‐blind trial. Gut 1978;19(3):175‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dürr HK, Zelder O, Maroske D, Bode JC. Treatment of acute pancreatitis with glucagon. Report on a double blind study [Zur behandlung der akuten pankreatitis mit glucagon. Bericht über eine doppelblindstudie]. Verhandlungen der Deutschen Gesellschaft für Innere Medizin 1976;82 Pt 1:970‐3. [PubMed] [Google Scholar]
Ebbehøj 1985 {published data only}
- Ebbehøj N, Bülow S, Friis J, Madsen P, Svendsen LB. Indomethacin treatment of acute pancreatitis. Scandinavian Journal of Gastroenterology Supplement 1984;19(Suppl 98):36. [DOI] [PubMed] [Google Scholar]
- Ebbehøj N, Friis J, Svendsen B. Indomethacin treatment of acute pancreatitis. A controlled double‐blind trial. Scandinavian Journal of Gastroenterology 1985;20(7):798‐800. [DOI] [PubMed] [Google Scholar]
Finch 1976 {published data only}
- Finch WT, Sawyers JL, Schenker S. A prospective study to determine the efficacy of antibiotics in acute pancreatitis. Annals of Surgery 1976;183(6):667‐71. [DOI] [PMC free article] [PubMed] [Google Scholar]
Freise 1986 {published data only}
- Freise J, Melzer P. Foy in the treatment of acute‐pancreatitis ‐ results of the Hannover multicentric double‐blind‐study with 50 patients. Zeitschrift Für Gastroenterologie 1985;23(9):429. [PubMed] [Google Scholar]
- Freise J, Melzer P, Schmidt FW, Horbach L. Gabexate mesilate in the treatment of acute pancreatitis ‐ results of the Hannover multicenter double‐blind trial with 50 patients [Gabexat mesilat in der behandlung der akuten pankreatitis. Ergebnisse der hannoverschen multizentrischen doppelblindstudie mit 50 patienten]. Zeitschrift Fur Gastroenterologie 1986;24(4):200‐11. [PubMed] [Google Scholar]
Frulloni 1994 {published data only}
- Frulloni L, Bassi C, Bovo P, Falconi M, Francesco V, Pederzoli P, et al. Gabexate mesilate vs aprotinin in the treatment of acute necrotizing pancreatitis. Argomenti di Gastroenterologia Clinica 1994;7(1):31‐6. [Google Scholar]
Garcia‐Barrasa 2009 {published data only}
- Garcia‐Barrasa A, Borobia FG, Pallares R, Jorba R, Poves I, Busquets J, et al. A double‐blind, placebo‐controlled trial of ciprofloxacin prophylaxis in patients with acute necrotizing pancreatitis. Journal of Gastrointestinal Surgery 2009;13(4):768‐74. [DOI] [PubMed] [Google Scholar]
Gilsanz 1978 {published data only}
- Gilsanz V, Oteyza CP, Rebollar JL. Glucagon vs anticholinergics in the treatment of acute pancreatitis. A double‐blind controlled trial. Archives of Internal Medicine 1978;138(4):535‐8. [PubMed] [Google Scholar]
Gjørup 1992 {published data only}
- Gjørup I, Roikjaer O, Andersen B, Burcharth F, Hovendal C, Pedersen SA, et al. A double‐blinded multicenter trial of somatostatin in the treatment of acute pancreatitis. Surgery, Gynecology & Obstetrics 1992;175(5):397‐400. [PubMed] [Google Scholar]
- Gjørup I, Roikjær O, Andersen B, Burcharth F, Hovendal C, Pedersen SA, et al. Somatostatin in the treatment of acute pancreatitis a double‐blind multicenter trial [abstract]. Digestion 1990;46(3):140. [Google Scholar]
Goebell 1979 {published data only}
- Goebell H, Ammann R, Akovbiantz A. Calcitonin in the treatment of acute pancreatitis. A multi centre double blind study. Irish Journal of Medical Science 1977;146(Suppl 1):105. [Google Scholar]
- Goebell H, Ammann R, Herfarth C. A double‐blind trial of synthetic salmon calcitonin in the treatment of acute pancreatitis. Scandinavian Journal of Gastroenterology 1979;14(7):881‐9. [DOI] [PubMed] [Google Scholar]
Goebell 1988 {published data only}
- Goebell H. Multicenter double‐blind trial of low‐dose intravenous proteinase‐inhibitor (foy‐r) in patients with acute‐pancreatitis. Zeitschrift Für Gastroenterologie 1988;26(9):447. [Google Scholar]
- Goebell H. Multicenter double‐blind‐study of gabexate‐mesilate (foy), given intravenously in low‐dose in acute‐pancreatitis. Digestion 1988;40(2):83‐. [Google Scholar]
Grupo Español 1996 {published data only}
- Grupo Español Cooperativo para el Estudio de la Somatostatina en el Tratamiento de la Pancreatitis Aguda Grave. [Somatostatina en el tratamiento de la pancreatitis aguda grave: ensayo clínico multicentrico, controlado, con asignación aleatoria y doble ciego. Presentación de resultados en el análisis secuencial]. Revista Española de Enfermedades Digestivas 1996;88(10):717‐8. [Google Scholar]
Guo 2015 {published data only}
- Guo H, Chen J, Suo D. Clinical efficacy and safety of ulinastatin plus octreotide for patients with severe acute pancreatitis. Zhonghua Yixue Zazhi [Chinese Medical Journal] 2015;95(19):1471‐4. [PUBMED: 26178495] [PubMed] [Google Scholar]
Hansky 1969 {published data only}
- Hansky J. The use of a peptidase inhibitor in the treatment of acute pancreatitis. Medical Journal of Australia 1969;1(25):1284‐5. [DOI] [PubMed] [Google Scholar]
Hejtmankova 2003 {published data only}
- Hejtmankova S, Cech P, Hoskovec D, Kostka R, Leffler J, Kasalicky M, et al. Antibiotic prophylaxis in severe acute pancreatitis: randomized multicenter prospective study with meropenem. Gastroenterology 2003;124(4):A85‐A. [Google Scholar]
Imrie 1978 {published data only}
- Imrie CW, Benjamin IS, Ferguson JC. A single centre double blind trial of trasylol therapy in primary acute pancreatitis. Irish Journal of Medical Science 1977;146(suppl.1):no.103. [DOI] [PubMed] [Google Scholar]
- Imrie CW, Benjamin IS, Ferguson JC. A single‐centre double‐blind trial of trasylol therapy in primary acute pancreatitis. British Journal of Surgery 1978;65(5):337‐41. [DOI] [PubMed] [Google Scholar]
- Imrie CW, Benjamin IS, Ferguson JC. Single centre double‐blind trial of trasylol therapy in primary acute pancreatitis. Gut 1977;18(11):A957‐A8. [DOI] [PubMed] [Google Scholar]
- Imrie CW, Benjamin IS, Ferguson JC, McKay AJ, Mackenzie I, Oneill J, et al. Single center double‐blind trial of trasylol therapy in primary acute‐pancreatitis. Gastroenterologie Clinique et Biologique 1978;2(11):954. [DOI] [PubMed] [Google Scholar]
- Imrie CW, Benjamin IS, Ferguson JC, Thomson WO, McKay AJ, Blumgart LH. Single‐center double‐blind trial of aprotinin (trasylol) therapy in primary acute‐pancreatitis. Annals of the Royal College of Surgeons of England 1978;60(2):142. [Google Scholar]
Imrie 1980 {published data only}
- Imrie CW, McKay AJ, Neill JO, Campbell FC, Gordon DA, Lang JA. Short duration megadosage iv trasylol in primary acute pancreatitis ‐ a double‐blind trial [abstract]. Gut 1980;21(Suppl 21):A457. [Google Scholar]
Isenmann 2004 {published data only}
- Beger HG, Isenmann R. Discussion on prophylactic antibiotic treatment in patients with predicted severe pancreatitis: a placebo‐controlled, double‐blind trial ‐ reply. Gastroenterology 2004;127(3):1016‐7. [DOI] [PubMed] [Google Scholar]
- Isenmann R, Ruenzi M, Kron M, Goebell H, Beger HG. Prophylactic antibiotics in severe acute pancreatitis. Results of a double‐blind, placebo‐controlled multicenter trial. Gastroenterology 2003;124(4):A32‐A. [DOI] [PubMed] [Google Scholar]
- Isenmann R, Runzi M, Kron M, Kahl S, Kraus D, Jung N. Prophylactic antibiotic treatment in patients with predicted severe acute pancreatitis: a placebo‐controlled, double‐blind trial. Gastroenterology 2004;126(4):997‐1004. [see comment]. [DOI] [PubMed] [Google Scholar]
Johnson 2001 {published data only}
- Johnson CD, Kingsnorth AN, Imrie CW, McMahon MJ, Neoptolemos JP, McKay C, et al. Double blind, randomised, placebo controlled study of a platelet activating factor antagonist, lexipafant, in the treatment and prevention of organ failure in predicted severe acute pancreatitis. Gut 2001;48(1):62‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kingsnorth AN. Early treatment with lexipafant, a platelet activating factor antagonist reduces mortality in acute pancreatitis: a double blind, randomized, placebo controlled study. Gastroenterology 1997;112(4):A453‐A. [Google Scholar]
- Toh SKC. Lexipafant, a platelet activation factor (paf) antagonist, reduces mortality in a randomised placebo‐controlled study in patients with severe acute pancreatitis [abstract]. Gut 1997;40(Suppl 1):A12. [Google Scholar]
Kalima 1980 {published data only}
- Kalima TV, Lempinen M. The effect of zinc‐protamine‐glucagon in acute pancreatitis. Annales Chirurgiae et Gynaecologiae 1980;69(6):293‐5. [PubMed] [Google Scholar]
Kingsnorth 1995 {published data only}
- Galloway SW, Formela L, Kingsnorth AN. A double blind placebo controlled study of bb‐882 (a potent paf antogonist) in human acute pancreatitis. Gut 1994;35(Suppl 5):T139. [Google Scholar]
- Galloway SW, Formela L, Kingsnorth AN. A double‐blind placebo controlled study of lexipafant (a potent paf antagonist) in acute pancreatitis. Gut 1995;36(3):A478. [Google Scholar]
- Kingsnorth AN, Galloway SW, Formela LJ. Randomized, double‐blind phase ii trial of lexipafant, a platelet‐activating factor antagonist, in human acute pancreatitis. British Journal of Surgery 1995;82(10):1414‐20. [DOI] [PubMed] [Google Scholar]
Kirsch 1978 {published data only}
- Kirsch A, Werner U, Heinze D. Proteinase inhibiting agents and glucagon in acute pancreatitis (author's transl). Zentralblatt für Chirurgie 1978;103(5):291‐303. [PubMed] [Google Scholar]
Kronborg 1980 {published data only}
- Kronborg O, Bulow S, Joergensen PM, Svendsen LB. A randomized double‐blind trial of glucagon in treatment of first attack of severe acute pancreatitis without associated biliary disease. American Journal of Gastroenterology 1980;73(5):423‐5. [PubMed] [Google Scholar]
- Kronborg O, Jorgensen PM, Bulow S. Controlled randomized trial of glucagon in treatment of 1st attack of severe acute‐pancreatitis without associated biliary disease, interim‐report. Scandinavian Journal of Gastroenterology 1976;11:13. [PubMed] [Google Scholar]
Llukacaj 2012 {published data only}
- Llukacaj A, Naco M, Mandi A, Rakipi B, Kodra N. Prophylactic antibiotic treatment for severe acute necrotizing pancreatitis. European Journal of Anaesthesiology 2012;29(Suppl 50):28. [Google Scholar]
Luengo 1994 {published data only}
- Luengo L, Gómez R, Castellote M, Ros S, Feliu F, Vadillo J. Influence of somatostatin in the evolution of acute pancreatitis. Cirugía Española 1994;56(Suppl 1):11‐2. [Google Scholar]
- Luengo L, Vicente V, Gris F, Coronas JM, Escuder J, Gomez JR, et al. Influence of somatostatin in the evolution of acute pancreatitis: a prospective randomized study. International Journal of Pancreatology 1994;15(2):139‐44. [DOI] [PubMed] [Google Scholar]
Luiten 1995 {published data only}
- Luiten EJ, Hop WC, Lange JF, Bruining HA. Controlled clinical trial of selective decontamination for the treatment of severe acute pancreatitis. Annals of Surgery 1995;222(1):57‐65. [DOI] [PMC free article] [PubMed] [Google Scholar]
Marek 1999 {published data only}
- Marek TA, Dziurkowska‐Marek A, Nowak A, Kacperek‐Hartleb T, Sierka E, Nowakowska‐Dulawa E. Prospective, randomized, placebo‐controlled trial of ascorbic acid in human acute pancreatitis. Gut 1999;45(Suppl. V):A315. [Google Scholar]
Martinez 1984 {published data only}
- Martinez E, Navarrete F. A controlled trial of synthetic salmon calcitonin in the treatment of severe acute pancreatitis. World Journal of Surgery 1984;8(3):354‐9. [DOI] [PubMed] [Google Scholar]
McKay 1997a {published data only}
- McKay C, Baxter J, Imrie C. A randomized, controlled trial of octreotide in the management of patients with acute pancreatitis. International Journal of Pancreatology 1997;21(1):13‐9. [DOI] [PubMed] [Google Scholar]
- McKay C, Baxter JN, Imrie CW. Octreotide in acute‐pancreatitis ‐ a randomized controlled trial. British Journal of Surgery 1994;81(12):1814‐. [Google Scholar]
McKay 1997b {published data only}
- Curran FJM, Sharples CE, Young CA, Curtis L, McKay CJ, Baxter JN, et al. Controlled trial of lexipafant in severe acute pancreatitis. Pancreas 1995;11(4):424. [Google Scholar]
- McKay CJ, Curran F, Sharples C, Baxter JN, Imrie CW. Prospective placebo‐controlled randomized trial of lexipafant in predicted severe acute pancreatitis. British Journal of Surgery 1997;84(9):1239‐43. [PubMed] [Google Scholar]
- McKay CJ, Imrie CW. Prospective placebo‐controlled randomized trial of lexipafant in predicted severe acute pancreatitis ‐ reply. British Journal of Surgery 1998;85(2):280. [PubMed] [Google Scholar]
Moreau 1986 {published data only}
- Moreau J, Bommelaer G, Buscail L, Benque A, Jacob C, Galleyrand J, et al. Preliminary‐results of a multicentric double‐blind trial of somatostatin (s) vs placebo (p) in acute‐pancreatitis (ap). Digestive Diseases and Sciences 1986;31(10):S24‐S. [Google Scholar]
MRC Multicentre Trial 1977 {published data only}
- Death from acute pancreatitis. M.R.C. Multicentre trial of glucagon and aprotinin. Lancet 1977;2(8039):632‐5. [PubMed] [Google Scholar]
Nordback 2001 {published data only}
- Nordback I, Sand J, Saaristo R, Paajanen H. Early treatment with antibiotics reduces the need for surgery in acute necrotizing pancreatitis‐‐a single‐center randomized study. Journal of Gastrointestinal Surgery: Official Journal of the Society for Surgery of the Alimentary Tract 2001;5(2):113‐8. [DOI] [PubMed] [Google Scholar]
Ohair 1993 {published data only}
- Ohair DP, Hoffman RG, Schroeder H, Wilson SD. Octreotide in the treatment of acute‐pancreatitis ‐ a prospective, randomized trial. Gastroenterology 1993;104(4):A326‐A. [Google Scholar]
Olah 2007 {published data only}
- Olah A, Belagyi T, Poto L, Romics Jr L, Bengmark S. Synbiotic control of inflammation and infection in severe acute pancreatitis: a prospective, randomized, double blind study. Hepato‐Gastroenterology 2007;54(74):590‐4. [PubMed] [Google Scholar]
Paran 1995 {published data only}
- Paran H, Neufeld D, Mayo A, Shwartz I, Singer P, Kaplan O, et al. Preliminary report of a prospective randomized study of octreotide in the treatment of severe acute pancreatitis. Journal of the American College of Surgeons 1995;181(2):121‐4. [PubMed] [Google Scholar]
Pederzoli 1993a {published data only}
- Bassi C, Cavallini G, Bovo P, Bonora A, Vesentini S, Pederzoli P. Gabexate mesilate in acute pancreatitis (ap). The Italian multicentric trial. International Journal of Pancreatology 1992;12(1):76. [Google Scholar]
- Bassi C, Vesentini S, Campedelli A, Nifosi F, Girelli R, Falconi M, et al. Imipenem prophylaxis in necrotizing pancreatitis: results of a multicenter study. International Journal of Pancreatology 1992;12(1):77. [Google Scholar]
- Pederzoli P, Bassi C, Vesentini S, Campedelli A. A randomized multicenter clinical trial of antibiotic prophylaxis of septic complications in acute necrotizing pancreatitis with imipenem. Surgery, Gynecology & Obstetrics 1993;176(5):480‐3. [PubMed] [Google Scholar]
Pederzoli 1993b {published data only}
- Bassi C, Cavallini G, Bovo P, Bonora A, Vesentini S, Pederzoli P. Gabexate mesilate in acute pancreatitis (ap). The Italian multicentric trial. International Journal of Pancreatology 1992;12(1):76. [Google Scholar]
- Bassi C, DiCarlo V, Zerbi A, Galloro V, Uomo G, Fontana G, et al. Role of imipenem (i) in preventing infected necrosis (in) during acute pancreatitis (np) results of the Italian multicenter study [abstract]. Digestion 1992;52(2):68. [Google Scholar]
- Pederzoli P, Cavallini G, Falconi M, Bassi C. Gabexate mesilate vs aprotinin in human acute pancreatitis (ga.Me.P.A.): A prospective, randomized, double‐blind multicenter study. International Journal of Pancreatology 1993;14(2):117‐24. [DOI] [PubMed] [Google Scholar]
Perezdeoteyza 1980 {published data only}
- Perezdeoteyza C, Rebollar JL, Ballarin M, Chantres MT, Alonso A, Marin J, et al. Controlled treatment of acute‐pancreatitis ‐ double‐blind‐study with cimetidine [Tratamiento controlado de la pancreatitis aguda. Ensayo doble ciego con cimetidina]. Revista Clinica Española 1980;158(6):263‐6. [PubMed] [Google Scholar]
Pettila 2010 {published data only}
- Kyhala L, Lindstrom O, Kylanpaa L, Mustonen H, Puolakkainen P, Kemppainen E, et al. Activated protein C retards recovery from coagulopathy in severe acute pancreatitis. Scandinavian Journal of Clinical and Laboratory Investigation 2016;76(1):10‐6. [DOI] [PubMed] [Google Scholar]
- Pettila V, Kyhala L, Kylanpaa ML, Leppaniemi A, Tallgren M, Markkola A, et al. APCAP ‐ activated protein C in acute pancreatitis: a double‐blind randomized human pilot trial. Critical Care 2010;14(4):R139. [DOI] [PMC free article] [PubMed] [Google Scholar]
Plaudis 2010 {published data only}
- Plaudis H, Boka V, Pupelis G. Early low volume oral synbiotic/prebiotic supplemented enteral stimulation of the gut in patients with sap: randomized double blind prospective study. HPB 2010;12(Suppl 1):49. [Google Scholar]
Poropat 2015 {published data only}
- Poropat G, Giljaca V, Licul V, Hauser G, Milic S, Stimac D. Imipenem prophylaxis for predicted severe acute pancreatitis‐preliminary results of a randomized clinical trial. United European Gastroenterology Journal 2015;3(5 Suppl):A172. [Google Scholar]
Rokke 2007 {published data only}
- Rokke O, Harbitz TB, Liljedal J, Pettersen T, Fetvedt T, Heen LO, et al. Early treatment of severe pancreatitis with imipenem: a prospective randomized clinical trial. Scandinavian Journal of Gastroenterology 2007;42(6):771‐6. [DOI] [PubMed] [Google Scholar]
Sainio 1995 {published data only}
- Sainio V, Kemppainen E, Puolakkainen P, Taavitsainen M, Kivisaari L, Valtonen V, et al. Early antibiotic treatment in acute necrotising pancreatitis. Lancet 1995;346(8976):663‐7. [DOI] [PubMed] [Google Scholar]
- Sainio V, Kemppainen E, Puolakkainen P, Taavitsainen M, Kivisaari L, Valtonen V, et al. Early antibiotic‐treatment in acute necrotizing pancreatitis ‐ a prospective randomized study. Gastroenterology 1994;106(4):A319‐A. [Google Scholar]
- Sainio V, Kemppainen E, Puolakkainen P, Taavitsainen M, Kivisaari L, Valtonen V, et al. Prophylactic antibiotic‐treatment in acute necrotizing pancreatitis ‐ a prospective randomized study. Gastroenterology 1993;104(4):A332‐A. [Google Scholar]
Sateesh 2009 {published data only}
- Sateesh J, Bhardwaj P, Singh N, Saraya A. Effect of antioxidant therapy on hospital stay and complications in patients with early acute pancreatitis: a randomised controlled trial. Tropical Gastroenterology 2009;30(4):201‐6. [PubMed] [Google Scholar]
Sharma 2011 {published data only}
- Sharma B, Srivastava S, Singh N, Sachdev V, Kapur S, Saraya A. Role of probiotics on gut permeability and endotoxemia in patients with acute pancreatitis: a double‐blind randomized controlled trial. Journal of Clinical Gastroenterology 2011;45(5):442‐8. [DOI] [PubMed] [Google Scholar]
Sillero 1981 {published data only}
- Sillero C, Perez‐Mateo M, Vazquez N, Martin A. Controlled trial of cimetidine in acute pancreatitis. European Journal of Clinical Pharmacology 1981;21(1):17‐21. [DOI] [PubMed] [Google Scholar]
Siriwardena 2007 {published data only}
- Sinwardena AK, Mason JM, Balachandra S, Bagul A, Galloway S, Formela L, et al. Randomized, double‐blind, placebo‐controlled, trial of high‐dose intravenous anti‐oxidant therapy in severe acute pancreatitis. Gastroenterology 2006;130(4):A83‐A. [Google Scholar]
- Siriwardena AK, Mason JM, Balachandra S, Bagul A, Galloway S, Formela L, et al. Randomised, double blind, placebo controlled trial of intravenous antioxidant (n‐acetylcysteine, selenium, vitamin c) therapy in severe acute pancreatitis. Gut 2007;56(10):1439‐44. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spicak 2002 {published data only}
- Hubaczova M, Spicak J, Antos F, Bartova I, Cech P, Kasalicky P, et al. The role of antibiotic treatment in severe form of acute pancreatitis: a randomized prospective study. Gastroenterology 2001;120(5):A645‐A. [Google Scholar]
- Hubaczová M, Spicák J, Antós F, Bártová I, Cech P, Kasalický M, et al. The role of antibiotic treatment in severe form of acute pancreatitis: a randomized prospective study [abstract]. Gut 2000;47(Suppl III):A142. [Google Scholar]
- Spicak J, Hubaczova M, Antos F, Bartova J, Cech P, Kasalicky M, et al. Antibiotics in the treatment of acute pancreatitis ‐ findings from a randomized multi‐centre prospective study [Antibiotika v léčbĕ akutní pankreatitidy ‐ poučení z randomizonvané multicentrické prospektivni studie]. Ceska a Slovenska Gastroenterologie a Hepatologie 2002;56(5):183‐9. [Google Scholar]
Spicak 2003 {published data only}
- Spicak J, Hejtmankova S, Hubaczova M, Antos F, Bartova J, Cech P, et al. Antibiotic prophylaxis of infectious complications of acute pancreatitis ‐ the results of randomised study by meropenem [Antibiotiká profylaxe infekĉních komplikací u akutní pankreatitidy ‐ výsledky randomizonvané studie s meropenemem]. Ceska a Slovenska Gastroenterologie a Hepatologie 2003;57(6):222‐7. [Google Scholar]
Storck 1968 {published data only}
- Storck G, Persson B. Trasylol treatment of acute pancreatitis. A double‐blind study [Trasylol vid akut pankreatit. En dubbel‐blindstudie]. Nordisk Medicin 1968;79(20):651‐3. [PubMed] [Google Scholar]
Trapnell 1974 {published data only}
- Trapnell JE. Controlled study of aprotinin in the treatment of acute pancreatitis [Etude controlee de l'aprotinine dans le traitement de la pancreatie aigue]. Annales de Chirurgie 1976;30(3):201. [PubMed] [Google Scholar]
- Trapnell JE, Rigby CC, Talbot CH, Duncan EH. A controlled trial of trasylol in the treatment of acute pancreatitis. British Journal of Surgery 1974;61(3):177‐82. [DOI] [PubMed] [Google Scholar]
- Trapnell JE, Rigby CC, Talbot CH, Duncan EH. Controlled study on trasylol in the treatment of acute pancreatitis [Eine kontrollierte prufung von trasylol bie der behandlung der akuten pankreatitis]. Die Medizinische Welt 1974;25(50):2106‐11. [PubMed] [Google Scholar]
- Trapnell JE, Talbot CH, Capper WM. Trasylol in acute pancreatitis. The American Journal of Digestive Diseases 1967;12(4):409‐12. [DOI] [PubMed] [Google Scholar]
Tykka 1985 {published data only}
- Tykka HT, Vaittinen EJ, Mahlberg KL. A randomized double‐blind study using cana2edta, a phospholipase a2 inhibitor, in the management of human acute pancreatitis. Scandinavian Journal of Gastroenterology 1985;20(1):5‐12. [DOI] [PubMed] [Google Scholar]
Uhl 1999 {published data only}
- Uhl W, Buchler MW, Malfertheiner P, Beger HG, Adler G, Gaus W. A randomised, double blind, multicentre trial of octreotide in moderate to severe acute pancreatitis. Gut 1999;45(1):97‐104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uhl W, Malfertheiner P, Adler G, Bruch HP, Lankisch PG, Lorenz D, et al. A randomized controlled multicentric trial on the role of octreotide in human acute pancreatitis. Gastroenterology 1997;112(4):A488‐A. [Google Scholar]
Usadel 1985 {published data only}
- Usadel KH, Uberla KK, Leuschner U. Treatment of acute‐pancreatitis with somatostatin ‐ results of the multicenter double‐blind trial (apts‐study). Digestive Diseases and Sciences 1985;30(10):992. [Google Scholar]
Valderrama 1992 {published data only}
- Valderrama R, Pérez‐Mateo M, Navarro S, Vázquez N, Sanjosé L, Adrián MJ, et al. Multicenter double‐blind trial of gabexate mesylate (foy) in unselected patients with acute pancreatitis. Digestion 1992;51(2):65‐70. [DOI] [PubMed] [Google Scholar]
Vege 2015 {published data only}
- Vege SS, Atwal T, Bi Y, Chari ST, Clemens MA, Enders FT. Pentoxifylline treatment in severe acute pancreatitis: a pilot, double‐blind, placebo‐controlled, randomized trial. Gastroenterology 2015;149(2):318‐20.e3. [DOI] [PubMed] [Google Scholar]
- Vege SS, Atwal T, Chari ST, Pearson RK, Loftus CG, Enders F, et al. Pentoxifylline treatment in predicted severe acute pancreatitis: a randomized double‐blind placebo‐controlled trial. Gastroenterology 2013;144(Suppl 1):S111‐2. [DOI] [PubMed] [Google Scholar]
Wang 2011 {published data only}
- Wang X, Li W, Niu C, Pan L, Li N, Li J. Thymosin alpha 1 is associated with improved cellular immunity and reduced infection rate in severe acute pancreatitis patients in a double‐blind randomized control study. Inflammation 2011;34(3):198‐202. [DOI] [PubMed] [Google Scholar]
Wang 2013a {published data only}
- Wang G, Wen J, Wilbur RR, Wen P, Zhou SF, Xiao X. The effect of somatostatin, ulinastatin and salvia miltiorrhiza on severe acute pancreatitis treatment. American Journal of the Medical Sciences 2013;346(5):371‐6. [DOI] [PubMed] [Google Scholar]
Wang 2013b {published data only}
- Wang R, Yang F, Guo Z, Yi Z, Huang L, Hu B, et al. Alleviation of acute pancreatitis with octreotide and celecoxib‐a randomized controlled trial. Journal of Gastroenterology and Hepatology 2013;28(Suppl):16‐7. [Google Scholar]
- Wang R, Yang F, Guo Z, Yi Z, Huang L, Tang C. Alleviation of acute pancreatitis with octreotide and celecoxib: a prospective randomized controlled trial. Gastroenterology 2013;144(Suppl 1):S139. [Google Scholar]
- Wang R, Yang F, Guo ZZ, Yi ZH, Huang LB, Hu B, et al. Alleviation of acute pancreatitis with octreotide and celecoxib: a prospective randomized controlled trial. Journal of Digestive Diseases 2014;15(Suppl 1):8. [Google Scholar]
Wang 2013c {published data only}
- Wang R, Yang F, Wu H, Wang Y, Huang Z, Hu B, et al. High‐dose versus low‐dose octreotide in the treatment of acute pancreatitis: a randomized controlled trial. Peptides 2013;40:57‐64. [DOI: 10.1016/j.peptides.2012.12.018] [DOI] [PubMed] [Google Scholar]
- Wang R, Yang F, Wu H, Wang YF, Hu B, Zhang MG, et al. Octreotide at high dose in the treatment of acute pancreatitis: a prospective randomized controlled trial. Gastroenterology 2012;142(5):S62. [Google Scholar]
Wang 2016 {published data only}
- Wang G, Liu Y, Zhou SF, Qiu P, Xu L, Wen P, et al. Effect of somatostatin, ulinastatin and gabexate on the treatment of severe acute pancreatitis. American Journal of the Medical Sciences 2016;351(5):506‐12. [DOI] [PubMed] [Google Scholar]
Xia 2014 {published data only}
- Xia YX, Liu XZ, Zhang XD, Shang PJ, Guo J. Efficacy and safety of omeprazole combined with somatostatin in treatment of severe acute pancreatitis. Shijie Huaren Xiaohua Zazhi [World Chinese Journal of Digestology] 2014;22(8):1179‐83. [Google Scholar]
Xue 2009 {published data only}
- Xue P, Deng LH, Zhang ZD, Yang XN, Wan MH, Song B, et al. Effect of antibiotic prophylaxis on acute necrotizing pancreatitis: Results of a randomized controlled trial. Journal of Gastroenterology and Hepatology (Australia) 2009;24(5):736‐42. [DOI] [PubMed] [Google Scholar]
Yang 1999 {published data only}
- Yang JQ, Wang P, Zhong J, Liu YK, Liu ZJ. A controlled study of somatostatin in the treatment of acute pancreatitis. Journal of Jinan University 1999;20(2):50‐2. [Google Scholar]
Yang 2012 {published data only}
- Yang F, Wu H, Li Y, Li Z, Wang C, Yang J, et al. Prevention of severe acute pancreatitis with octreotide in obese patients: a prospective multi‐center randomized controlled trial. Pancreas 2012;41(8):1206‐12. [DOI] [PubMed] [Google Scholar]
Zhu 2014 {published data only}
- Zhu YM, Lin S, Dang XW, Wang M, Li L, Sun RQ, et al. Effects of probiotics in treatment of severe acute pancreatitis. Shijie Huaren Xiaohua Zazhi [World Chinese Journal of Digestology] 2014;22(32):5013‐7. [Google Scholar]
References to studies excluded from this review
Akzhigitov 1968 {published data only}
- Akzhigitov GN. Use of monoaminoxidase inhibitor vetrazine in the treatment of acute pancreatitis [Primenenie ingibitora monoaminoksidazy vetrazina pri lechenii ostrogo pankreatita]. Terapevticheskii Arkhiv 1968;40(10):82‐4. [PubMed] [Google Scholar]
Akzhigitov 1969 {published data only}
- Akzhigitov GN, Grach Z, Rubtsova LK, Koroleva VG, Pozdniakova VP. Use of glycocycline in the treatment of patients with acute pancreatitis and cholepancreatitis. Antibiotiki 1969;14(2):174‐8. [PubMed] [Google Scholar]
Al‐Leswas 2013a {published data only}
- Al‐Leswas D, Arshad A, Eltweri A, Chung WY, Al‐Taan O, Pollard C, et al. An omega‐3 rich lipid emulsion is associated with improved clinical outcome in patients with severe acute pancreatitis: a randomised double‐blind controlled trial. British Journal of Surgery 2013;100(Suppl 4):2. [Google Scholar]
Al‐Leswas 2013b {published data only}
- Al‐Leswas D, Chung WY, Eltweri A, Arshad A, Al‐Taan O, Pollard C, et al. Evaluation of the acute inflammatory response to omega‐3 fatty acids in patients with severe acute pancreatitis: a randomised controlled trial. Pancreatology 2013;13(1):e6. [Google Scholar]
Al‐Leswas 2013c {published data only}
- Al‐Leswas D, Chung WY, Eltweri A, Stephenson J, Arshad A, Pollard C, et al. Evaluation of the cytokines response to omega‐3 fatty acids in patients with severe acute pancreatitis: a randomised controlled trial. Clinical Nutrition 2013;32(Suppl 1):S2. [DOI] [PubMed] [Google Scholar]
Al‐Leswas 2013d {published data only}
- Al‐Leswas D, Eltweri A, Arshad A, Chung WY, Al‐Taan O, Pollard C, et al. Progression of the early warning scores (ews) in severe acute pancreatitis patients treated with omega‐3 fish oil: a randomised control trial. Clinical Nutrition 2013;32(Suppl 1):S47‐S8. [Google Scholar]
Al‐Leswas 2013e {published data only}
- Al‐Leswas D, Eltweri A, Arshad A, Chung WY, Stephenson J, Pollard C, et al. An omega‐3 rich lipid emulsion is associated with fewer new organ failures, less systemic inflammatory response syndrome and improves the outcome in patients with severe acute pancreatitis: a randomised double‐blind, phase ii control trial. Pancreatology 2013;13(1):e2. [Google Scholar]
Al‐Leswas 2013f {published data only}
- Al‐Leswas D, Eltweri A, Chung WY, Al‐Taan O, Arshad A, Pollard C, et al. An omega‐3 rich lipid emulsion is associated with fewer new organ failures, less systemic inflammatory response syndrome and improves the outcome in patients with severe acute pancreatitis: A randomised double‐blind, phase ii control trial. Clinical Nutrition 2013;32(Suppl 1):S2‐3. [Google Scholar]
Al‐Leswas 2013g {published data only}
- Al‐Leswas D, Eltweri A, Hall T, Stephenson J, Pollard C, Garcea G, et al. Safety and tolerability of two parenteral lipid emulsions in patients with severe acute pancreatitis as measured by serum triglyceride and cholesterol levels: a randomised controlled trial. Clinical Nutrition 2013;32(Suppl 1):S49‐50. [Google Scholar]
Amundsen 1972 {published data only}
- Amundsen E. The use of protease inhibitors during acute haemorrhagic pancreatitis. Experimental findings and clinical implications. Annales Chirurgiae et Gynaecologiae Fenniae 1972;61(5):284‐7. [PubMed] [Google Scholar]
Andersson 2008 {published data only}
- Andersson RG. Probiotics in acute pancreatitis. British Journal of Surgery 2008;95(8):941‐2. [DOI] [PubMed] [Google Scholar]
Baden 1967 {published data only}
- Baden H, Jordal K, Lund F, Zachariae F. A double‐blind controlled clinical trial of trasylol. Preliminary results in acute pancreatitis and in prophylaxis against postoperative pancreatitis. Acta Chirurgica Scandinavica 1967;378(Suppl):97‐102. [PubMed] [Google Scholar]
Baden 1969 {published data only}
- Baden H, Jordal K, Lund F, Zachariae F. Prophylactic and curative action of trasylol in pancreatitis; a double blind trial. Scandinavian Journal of Gastroenterology 1969;4(3):291‐5. [PubMed] [Google Scholar]
Bai 2013 {published data only}
- Bai YT, Guo XZ, Li HY, Shao XD, Cui ZM, Wang D, et al. Therapeutic effect of ulinastatin vs gabexate mesilate in management of acute pancreatitis. Shijie Huaren Xiaohua Zazhi [World Chinese Journal of Digestology] 2013;21(14):1339‐42. [Google Scholar]
Bassi 1998 {published data only}
- Bassi C, Falconi M, Talamini G, Uomo G, Papaccio G, Dervenis C, et al. Controlled clinical trial of pefloxacin versus imipenem in severe acute pancreatitis. Gastroenterology 1998;115(6):1513‐7. [DOI] [PubMed] [Google Scholar]
Beechey‐Newman 1991 {published data only}
- Beechey‐Newman N, Lee W, Wilkinson M, Grogono J, McPherson GAD. Treatment with high dose octreotide improves the clinical course of acute pancreatitis [abstract]. Gut 1991;32(Suppl.5):A558. [Google Scholar]
Beechey‐Newman 1993 {published data only}
- Beechey‐Newman N. Controlled trial of high‐dose octreotide in treatment of acute pancreatitis. Evidence of improvement in disease severity. Digestive Diseases and Sciences 1993;38(4):644‐7. [DOI] [PubMed] [Google Scholar]
Beger 2001 {published data only}
- Beger HG. Early treatment with antibiotics reduces the need for surgery in acute necrotizing pancreatitis ‐ a single‐center randomized study ‐ commentary. Journal of Gastrointestinal Surgery 2001;5(2):119‐20. [DOI] [PubMed] [Google Scholar]
Bender 1992 {published data only}
- Bender HJ, Albrecht MD, Quintel M, Ackern K. Use of octreotidacetate (sandostatin) in the treatment of necrotising haemorrhagic pancreatitis. Der Anaesthesist 1992;41(Suppl 2):S130. [Google Scholar]
Binder 1993 {published data only}
- Binder M, Buchler M, Uhl W, Friess H, Dennler HJ, Beger HG. Octreotide in the treatment of acute pancreatitis: results of an unicentric prospective trial with three different octreotide dosages [Octreotide bei akuter pankreatitis: Ergebnisse einer unizentrischen prospektiven studie mit drei verschiedenen octreotide‐dosierungen]. Langenbecks Archiv fur Chirurgie 1993;Suppl 1 Forumband:233‐8. [Google Scholar]
Binder 1994 {published data only}
- Binder M, Uhl W, Friess H, Malfertheiner P, Buchler MW. Octreotide in the treatment of acute pancreatitis: results of a unicenter prospective trial with three different octreotide dosages. Digestion 1994;55(Suppl 1):20‐3. [DOI] [PubMed] [Google Scholar]
Brown 2004 {published data only}
- Brown A. Prophylactic antibiotic use in severe acute pancreatitis: Hemlock, help, or hype?. Gastroenterology 2004;126(4):1195‐8. [DOI] [PubMed] [Google Scholar]
Buchler 1988 {published data only}
- Buchler M, Malfertheiner P, Uhl W, Wolf HR, Schwab G, Beger HG. Gabexate mesilate in the therapy of acute pancreatitis. Multicenter study of tolerance of a high intravenous dose (4 g/day) [Gabexat‐mesilat in der therapie der akuten pankreatitis. Multicenterstudie zur vertraglichkeit einer hohen intravenosen dosis (4 g/Tag)]. Medizinische Klinik 1988;83(10):320‐4, 52. [PubMed] [Google Scholar]
Cameron 1979 {published data only}
- Cameron JL, Mehigan D, Zuidema GD. Evaluation of atropine in acute pancreatitis. Surgery Gynecology and Obstetrics 1979;148(2):206‐8. [PubMed] [Google Scholar]
Cheng 2008 {published data only}
- Cheng YX, Wang M, Cheng X. Effect of dachaihu decoction in treating acute mild pancreatitis of gan‐qi stagnant type. Zhongguo Zhongxiyi Jiehe Zqzhi [Chinese Journal of Integrated Traditional and Western Medicine] 2008;28(9):793‐6. [PubMed] [Google Scholar]
Cullimore 2008 {published data only}
- Cullimore J, Cotter L, Gonzalez A. Antibiotics in acute necrotising pancreatitis [author reply]. Lancet 2008;371(9618):1072. [DOI] [PubMed] [Google Scholar]
Curtis 1997 {published data only}
- Curtis LD. Lexipafant(bb‐882), a potent PAF antagonist in acute pancreatitis. Advances in Experimental Medicine and Biology 1997;416:361‐3. [DOI] [PubMed] [Google Scholar]
D'Amico 1990 {published data only}
- D'Amico D, Favia G, Biasiato R, Casaccia M, Falcone F, Fersini M, et al. The use of somatostatin in acute pancreatitis‐‐results of a multicenter trial. Hepato‐Gastroenterology 1990;37(1):92‐8. [PubMed] [Google Scholar]
Da Silvereira 2002 {published data only}
- Silveira EBV, Barkin JS. Antibiotic prophylaxis in acute necrotizing pancreatitis. American Journal of Gastroenterology 2002;97(6):1557‐9. [DOI] [PubMed] [Google Scholar]
De Vries 2007 {published data only}
- Vries AC, Besselink MG, Buskens E, Ridwan BU, Schipper M, Erpecum KJ, et al. Randomized controlled trials of antibiotic prophylaxis in severe acute pancreatitis: relationship between methodological quality and outcome. Pancreatology 2007;7(5‐6):531‐8. [DOI] [PubMed] [Google Scholar]
Dikkenberg 2008 {published data only}
- Dikkenberg GM. Probiotic prophylaxis in patients with predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial and informed consent procedure [author reply ‐ 4] [Probioticaprofylaxe bij voorspeld ernstige acute pancreatitis; gerandomiseerde, dubbelblinde, placebogecontroleerde trial en informed‐consentprocedure]. Nederlands Tijdschrift voor Geneeskunde 2008;152(28):1592. [PubMed] [Google Scholar]
Dreiling 1977 {published data only}
- Dreiling DA, Nacchiero M, Kaplan. I. Is there a place for steroids in the treatment of pancreatic inflammation?. American Journal of Gastroenterology 1977;67(1):21‐8. [PubMed] [Google Scholar]
Du 2002 {published data only}
- Du W, Shen D, Huang C, Zhou Y. Effect of high‐dose vitamin C on cellular immunity of patients with acute pancreatitis. Wei Chang Bing Xue [Chinese Journal of Gastroenterology] 2002;7(4):213‐5. [Google Scholar]
Du 2003 {published data only}
- Du WD, Yuan ZR, Sun J, Tang JX, Cheng AQ, Shen DM, et al. Therapeutic efficacy of high‐dose vitamin c on acute pancreatitis and its potential mechanisms. World Journal of Gastroenterology 2003;9(11):2565‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dürr 1985 {published data only}
- Dürr GH, Schaefers A, Maroske D, Bode JC. A controlled study on the use of intravenous fat in patients suffering from acute attacks of pancreatitis. Infusionstherapie und Klinische Ernahrung 1985;12(3):128‐33. [DOI] [PubMed] [Google Scholar]
Freise 1985 {published data only}
- Freise J, Schmidt FW, Maferstedt P, Schmid K. Gabexate mesilate and camostate: New inhibitors of phospholipase a2 and their influence on the alpha‐amylase activity in serum of patients with acute pancreatitis. Clinical Biochemistry 1985;18(4):224‐9. [DOI] [PubMed] [Google Scholar]
Friess 1994 {published data only}
- Friess H, Hofbauer B, Buchler MW. The role of somatostatin and octreotide in pancreatic surgery and in acute and chronic pancreatitis. Digestive Surgery 1994;11(3‐6):445‐50. [Google Scholar]
Gabryelewicz 1968 {published data only}
- Gabryelewicz A, Niewiarowski S. Activation of blood clotting and inhibition of fibrinolysis in acute pancreatitis. Thrombosis et Diathesis Haemorrhagica 1968;20(3):409‐14. [PubMed] [Google Scholar]
Gabryelewicz 1976 {published data only}
- Gabryelewicz A, Olszewski S, Szalaj W, Puchalski Z, Stasiewicz J. Heparin and glucagon treatment of acute pancreatitis‐‐results (author's transl) [Wyniki leczenia ostrego zapalenia trzustki za pomoca heparyny lub glukagonu]. Przeglad Lekarski 1976;33(2):323‐8. [PubMed] [Google Scholar]
Gao 2015b {published data only}
- Gao Q, Liang N. Integrated traditional Chinese medicine improves acute pancreatitis via the downregulation of prss1 and spink1. Experimental and Therapeutic Medicine 2015;9(3):947‐54. [DOI] [PMC free article] [PubMed] [Google Scholar]
Garcia 2005 {published data only}
- Garcia DA, Gonzalez JA, Rodriguez N, Britton CS, Hinojo E, Quintanilla C, et al. Utility of synbiotic therapy in acute pancreatitis. A double blind randomized clinical trial. Gastroenterology 2005;128(4 Suppl 2):A173‐A. [Google Scholar]
Gostishchev 1977 {published data only}
- Gostishchev VK, Lutsevich EV, Tolstykh PI, Vladimirov VG, Sergienko VI. [effectiveness of proteolysis inhibitors in treatment of acute pancreatitis]. Khirurgiia 1977, (7):87‐92. [PubMed] [Google Scholar]
Guo 2013 {published data only}
- Guo Z, Wang R, Tang C. Effect of octreotide on plasma levels of somatostatin, interleukin‐6, and tumor necrosis factor‐alpha in patients with acute pancreatitis: A prospective single‐center randomized controlled trial. Gastroenterology 2013;144(5 Suppl):S566. [Google Scholar]
Hajdu 2012 {published data only}
- Hajdu N, Belagyi T, Issekutz A, Bartek P, Gartner B, Olah A. Intravenous glutamine and early nasojejunal nutrition in severe acute pancreatitis ‐‐ a prospective randomized clinical study [Intravenas glutamin es korai nasojejunalis taplalas egyuttes alkalmazasa sulyos acut pancreatitisben ‐‐ prospektiv randomizalt kettos vak kontrollalt klinikai vizsgalat]. Magyar sebeszet 2012;65(2):44‐51. [DOI] [PubMed] [Google Scholar]
Harinath 2002 {published data only}
- Harinath G, O'Riordan B. Prospective randomized double‐blind placebo‐controlled trial of glyceryl trinitrate in endoscopic retrograde cholangiopacreatography‐induced pancreatitis (Br J Surg 2001;88:1178‐82). The British Journal of Surgery 2002;89(5):628‐9; discussion 9. [PubMed] [Google Scholar]
Hart 2008 {published data only}
- Hart PA, Bechtold ML, Marshall JB, Choudhary A, Puli SR, Roy PK. Prophylactic antibiotics in necrotizing pancreatitis: a meta‐analysis. Southern Medical Journal 2008;101(11):1126‐31. [DOI] [PubMed] [Google Scholar]
He 2004 {published data only}
- He XL, Ma QJ, Lu JG, Chu YK, Du XL. Effect of total parenteral nutrition (tpn) with and without glutamine dipeptide supplementation on outcome in severe acute pancreatitis (sap). Clinical Nutrition, Supplement 2004;1(1):43‐7. [Google Scholar]
Helton 2001 {published data only}
- Helton S, Nordback I, Rattner DW, Gloor B, Kumar A, Sarr MG, et al. Early treatment with antibiotics reduces the need for surgery in acute necrotizing pancreatitis ‐ a single‐center randomized study ‐ discussion. Journal of Gastrointestinal Surgery 2001;5(2):118‐9. [DOI] [PubMed] [Google Scholar]
Hoekstra 2008 {published data only}
- Hoekstra JH. Probiotic prophylaxis in patients with predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial and informed consent procedure [Probioticaprofylaxe bij voorspeld ernstige acute pancreatitis; gerandomiseerde, dubbelblinde, placebogecontroleerde trial en informed‐consentprocedure]. Nederlands Tijdschrift voor Geneeskunde 2008;152(28):1591‐2; author reply 3‐4. [PubMed] [Google Scholar]
Holub 1974 {published data only}
- Holub K, Om P. Treatment of acute pancreatitis using glucagon [Glukagonbehandlung der akuten pankreatitis]. Zentralblatt fur Chirurgie 1974;99(24):748‐50. [PubMed] [Google Scholar]
Howard 2007 {published data only}
- Howard TJ. As good as it gets: the study of prophylactic antibiotics in severe acute pancreatitis. Annals of Surgery 2007;245(5):684‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Howes 1975 {published data only}
- Howes R, Zuidema GD, Cameron JL. Evaluation of prophylactic antibiotics in acute pancreatitis. Journal of Surgical Research 1975;18(2):197‐200. [DOI] [PubMed] [Google Scholar]
Huang 2008 {published data only}
- Huang XX, Wang XP, Ma JJ, Jing DD, Wang PW, Wu K. Effects of enteral nutrition supplemented with glutamine and arginine on gut barrier in patients with severe acute pancreatitis: a prospective randomized controlled trial. National Medical Journal of China 2008;88(34):2407‐9. [PubMed] [Google Scholar]
Issekutz 2002 {published data only}
- Issekutz A, Olah A, Bengmark S. Early enteral supply of lactobacilli + fibre vs. Placebo + fibre in severe acute pancreatitis ‐ a prospective, randomized, controlled trial. Zeitschrift fur Gastroenterologie 2002;40(5):338. [Google Scholar]
Ivanov 2002 {published data only}
- Ivanov Iu V, Chudnykh SM, Mozgalin AG. Effectiveness of mexidol in acute pancreatitis [Effektivnost' meksidola pri ostrom pankreatite]. Klinicheskaia Meditsina 2002;80(9):44‐6. [PubMed] [Google Scholar]
Jiang 1988 {published data only}
- Jiang LS, Fang PS, Xu ZH. Controlled trial of calcium channel blocker, rou gui qing, and ‐receptor blocker for treatment of 80 patients with acute pancreatitis. Linchuang Gandanbing Zazhi [Chinese Journal of Clinical Hepatology] 1988;4(4):48. [Google Scholar]
Karakan 2007 {published data only}
- Karakan T, Ergun M, Dogan I, Cindoruk M, Unal S. Comparison of early enteral nutrition in severe acute pancreatitis with prebiotic fiber supplementation versus standard enteral solution: a prospective randomized double‐blind study. World Journal of Gastroenterology 2007;13(19):2733‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Karakoyunlar 1999 {published data only}
- Karakoyunlar O, Sivrel E, Tanir N, Denecli AG. High dose octreotide in the management of acute pancreatitis. Hepato‐Gastroenterology 1999;46(27):1968‐72. [PubMed] [Google Scholar]
Karavanov 1966 {published data only}
- Karavanov AG, Osingolts SL. The use of epsilon‐aminocaproic acid in the treatment of acute pancreatits [Primenenie epsilon‐aminokapronovoi kisloty (E‐AKK) v lechenii ostrykh pankreatitov]. Klinicheskaia Meditsina 1966;44(5):13‐5. [PubMed] [Google Scholar]
Lasztity 2005a {published data only}
- Lasztity N, Hamvas J, Biró L, Németh E, Marosvölgyi T, Decsi T, et al. Effect of enterally administered n‐3 polyunsaturated fatty acids in acute pancreatitis‐‐a prospective randomized clinical trial. Clinical Nutrition 2005;24(2):198‐205. [DOI] [PubMed] [Google Scholar]
Lasztity 2005b {published data only}
- Lasztity N, Pap A, Hamvas J, Biro L, Nemeth E, Marosvolgyi T, et al. Effect of enterally administered n‐3 polyunsaturated fatty acids in acute pancreatitis ‐ a prospective randomized clinical trial. Gastroenterology 2005;128(4):A174‐A. [DOI] [PubMed] [Google Scholar]
Lasztity 2006 {published data only}
- Lasztity N, Hamvas J, Biro L, Nemeth E, Marosvolgyi T, Decsi T, et al. Enteral administration of n‐3 polyunsaturated fatty acids in acute pancreatitis [Az n‐3 többszörösen telítetlen zsírsavak enteralis adása akut pancreatitisben ‐ előzetes eredmények]. Lege Artis Medicinae 2006;16(10):848‐54. [Google Scholar]
Lata 1998 {published data only}
- Lata J, Dít P, Julínková K, Precechtělová M, Prásek J. Effect of octreotide on the clinical course of acute pancreatitis and levels of free oxygen radicals and antioxidants [Vliv octreotidu na klinicky prubeh akutni pankreatitidy a hladinu volnych kyslikovych radikala a antioxidacnich latek]. Vnitrní lékarství 1998;44(9):524‐7. [PubMed] [Google Scholar]
Lata 2010 {published data only}
- Lata J, Juránková J, Stibůrek O, Príbramská V, Senkyrík M, Vanásek T. Probiotics in acute pancreatitis‐‐a randomised, placebo‐controlled, double‐blind study [Probiotika u akutni pankreatitidy‐‐randomizovana, placebem kontrolovana, dvojite slepa studie]. Vnitrní lékarství 2010;56(2):111‐4. [PubMed] [Google Scholar]
Lim 2015 {published data only}
- Lim CL, Lee W, Liew YX, Tang SS, Chlebicki MP, Kwa AL. Role of antibiotic prophylaxis in necrotizing pancreatitis: a meta‐analysis. Journal of Gastrointestinal Surgery 2015;19(3):480‐91. [DOI] [PubMed] [Google Scholar]
Lu 2006 {published data only}
- Lu J, Liu CW, Zheng YK, Hu W, Zhu KY, Hu WH. Effects of glutamine in hemodynamics and oxygen metabolism in patients with severe acute pancreatitis. Zhongguo Linchuang Yingyang Zazhi [Chinese Journal of Clinical Nutrition] 2006;14(4):227‐30. [Google Scholar]
Lu 2008 {published data only}
- Lu HG, Shi YB, Zhao LM, Bai C, Wang X. Role of enteral ebselen and ethylhydroxyethyl cellulose in pancreatitis‐associated multiple‐organ dysfunction in humans. Journal of Organ Dysfunction 2008;4(1):43‐50. [Google Scholar]
Manes 2003 {published data only}
- Manes G, Rabitti PG, Menchise A, Riccio E, Balzano A, Uomo G. Prophylaxis with meropenem of septic complications in acute pancreatitis: a randomized, controlled trial versus imipenem. Pancreas 2003;27(4):e79‐83. [DOI] [PubMed] [Google Scholar]
Manes 2006 {published data only}
- Manes G, Uomo I, Menchise A, Rabitti PG, Ferrara EC, Uomo G. Timing of antibiotic prophylaxis in acute pancreatitis: a controlled randomized study with meropenem. American Journal of Gastroenterology 2006;101(6):1348‐53. [DOI] [PubMed] [Google Scholar]
McClave 2009 {published data only}
- McClave SA, Heyland DK, Wischmeyer PE. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomized, double‐blind, placebo‐controlled trial. Journal of Parenteral and Enteral Nutrition 2009;33(4):444‐6. [DOI] [PubMed] [Google Scholar]
Mercadier 1973 {published data only}
- Mercadier M, Chigot JP, Clot JP. Has glucagon a value in the treatment of acute pancreatitis? [Le glucagon a‐t‐il un intert dans le traitement des pancreatites aigues]. Nouvelle Presse Medicale 1973;2(40):2692. [PubMed] [Google Scholar]
Niu 2014 {published data only}
- Niu G, Zhao R, Gao F, Zheng D, Liu X, Wu H, et al. Effect of omega‐3 polyunsaturated fatty acids on intestinal mucosal barrier of patients with severe acute pancreatitis. Zhongguo Linchuang Yingyang Zazhi [Chinese Journal of Clinical Nutrition] 2014;22(6):329‐33. [Google Scholar]
Pearce 2006 {published data only}
- Pearce CB, Sadek SA, Walters AM, Goggin PM, Somers SS, Toh SK, et al. A double‐blind, randomised, controlled trial to study the effects of an enteral feed supplemented with glutamine, arginine, and omega‐3 fatty acid in predicted acute severe pancreatitis. Journal of the Pancreas 2006;7(4):361‐71. [PubMed] [Google Scholar]
Pederzoli 1995 {published data only}
- Pederzoli P, Bassi C, Falconi M, Santis L, Uomo G, Rabitti PG, et al. Gabexate mesilate in the treatment of acute pancreatitis. Annali Italiani di Chirurgia 1995;66(2):191‐5. [PubMed] [Google Scholar]
Pezzilli 1997 {published data only}
- Pezzilli R, Dragonetti C, Innocenti P, Miglioli M. A multicenter, open label, copmarative, randomized study of two schedules of gabexate mesilate (1500 mg/day vs. 900 mg/day) for the treatment of severe acute pancreatitis. An interim report [abstract]. Italian Journal of Gastroenterology and Hepatology 1997;29(Suppl 1):A7. Abstract 17. [Google Scholar]
Pezzilli 1999 {published data only}
- Pezzilli R, Dragonetti C, Innocenti P, Miglioli M. Amulticenter, open label, comparative, randomized study of two schedules of gabexate mesilate for the treatment of severe acute pancreatitis.[abstract]. Gut 1999;45(Suppl V):A315. [Google Scholar]
Pezzilli 2001 {published data only}
- Pezzilli R, Miglioli M. Multicentre comparative study of two schedules of gabexate mesilate in the treatment of acute pancreatitis. Digestive and Liver Disease 2001;33(1):49‐57. [DOI] [PubMed] [Google Scholar]
Piascik 2010 {published data only}
- Piascik M, Rydzewska G, Milewski J, Olszewski S, Furmanek M, Walecki J, et al. The results of severe acute pancreatitis treatment with continuous regional arterial infusion of protease inhibitor and antibiotic: a randomized controlled study. Pancreas 2010;39(6):863‐7. [DOI] [PubMed] [Google Scholar]
Plaudis 2012 {published data only}
- Plaudis H, Pupelis G, Zeiza K, Boka V. Early low volume oral synbiotic/prebiotic supplemented enteral stimulation of the gut in patients with severe acute pancreatitis: a prospective feasibility study. Acta Chirurgica Belgica 2012;112(2):131‐8. [DOI] [PubMed] [Google Scholar]
Rahman 2003 {published data only}
- Rahman SH, Catton JA, McMahon MJ. Letter 2: Randomized clinical trial of specific lactobacillus and fibre supplement to early enteral nutrition in patients with acute pancreatitis (Br J Surg 2002; 89: 1103‐1107) [comment]. British Journal of Surgery 2003;90(1):123. [DOI] [PubMed] [Google Scholar]
Ranson 1976 {published data only}
- Ranson JH, Rifkind KM, Turner JW. Prognostic signs and nonoperative peritoneal lavage in acute pancreatitis. Surgery, Gynecology & Obstetrics 1976;143(2):209‐19. [PubMed] [Google Scholar]
Reddy 2008 {published data only}
- Reddy BS, MacFie J. Probiotic prophylaxis in predicted severe acute pancreatitis. Lancet 2008;372(9633):113‐. [DOI] [PubMed] [Google Scholar]
Santen 2008 {published data only}
- Santen GW, Benus RF, Werf TS. Probiotic prophylaxis in patients with predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial and informed consent procedure [Probioticaprofylaxe bij voorspeld ernstige acute pancreatitis; gerandomiseerde, dubbelblinde, placebogecontroleerde trial en informed‐consentprocedure]. Nederlands Tijdschrift voor Geneeskunde 2008;152(28):1591; author reply 3‐4. [PubMed] [Google Scholar]
Singer 1966 {published data only}
- Singer A, Tornya P, Skyring A. Double‐blind study of trasylol in treatment of acute pancreatitis. Gut 1966;7(3):304‐&. [Google Scholar]
Skyring 1965 {published data only}
- Skyring A, Singer A, Tornya P. Treatment of acute pancreatitis with trasylol: report of a controlled therapeutic trial. British Medical Journal 1965;2(5462):627‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Tanaka 1979 {published data only}
- Tanaka N, Tsuchiya R, Ishii K. Comparative clinical study of foy and trasylol in acute pancreatitis. Advances in Experimental Medicine and Biology 1979;120 B:367‐78. [PubMed] [Google Scholar]
Tang 2005 {published data only}
- Tang WF, Wan MH, Zhu L, Chen GY, Xia Q, Huang X. Immuno‐modulatory effect of somatostatin combined with traditional Chinese medicine on severe acute pancreatitis at early stage: a randomized control trial. Zhongxiyi Jiehe Xuebao [Journal of Chinese Integrative Medicine] 2005;3(2):103‐7. [DOI] [PubMed] [Google Scholar]
Tang 2007 {published data only}
- Tang WF, Wang YG, Zhu L, Wan MH, Chen GY, Xia Q, et al. Effect of somatostatin on immune inflammatory response in patients with severe acute pancreatitis. Multiphase Pumping and Technologies Conference and Exhibition 2007 2007;8(2):96‐102. [DOI] [PubMed] [Google Scholar]
Ukai 2015 {published data only}
- Ukai T, Shikata S, Inoue M, Noguchi Y, Igarashi H, Isaji S, et al. Early prophylactic antibiotics administration for acute necrotizing pancreatitis: A meta‐analysis of randomized controlled trials. Journal of Hepato‐biliary‐pancreatic Sciences 2015;22(4):316‐21. [DOI] [PubMed] [Google Scholar]
Usadel 1980 {published data only}
- Usadel KH, Leuschner U, Uberla KK. Treatment of acute pancreatitis with somatostatin: a multicenter double blind study. New England Journal of Medicine 1980;303(17):999‐1000. [DOI] [PubMed] [Google Scholar]
Venkatesan 2008 {published data only}
- Venkatesan T. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double‐blind, placebo‐controlled trial. Nutrition in Clinical Practice 2008;23(6):662‐3. [DOI] [PubMed] [Google Scholar]
Villatoro 2010 {published data only}
- Villatoro E, Mulla M, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database of Systematic Reviews 2010, Issue 5. [DOI: 10.1002/14651858.CD002941.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Wang 2008 {published data only}
- Wang X, Li W, Li N, Li J. Omega‐3 fatty acids ‐ supplemented parenteral nutrition decreases hyperinflammatory response and attenuates systemic disease sequelae in severe acute pancreatitis: a randomized and controlled study. Journal of Parenteral and Enteral Nutrition 2008;32(3):236‐41. [DOI] [PubMed] [Google Scholar]
Wang 2009 {published data only}
- Wang X, Li W, Zhang F, Pan L, Li N, Li J. Fish oil‐supplemented parenteral nutrition in severe acute pancreatitis patients and effects on immune function and infectious risk: a randomized controlled trial. Inflammation 2009;32(5):304‐9. [DOI] [PubMed] [Google Scholar]
Weismann 2010 {published data only}
- Weismann D, Maier SKG. Activated protein C in acute pancreatitis ‐ a double‐blind, randomized, placebo‐controlled study [Aktiviertes protein C bei akuter pankreatitis – eine doppelblinde, randomisierte, placebo‐kontrollierte studie]. Medizinische Klinik 2010;105(10):747. [Google Scholar]
Wyncoll 1998 {published data only}
- Wyncoll DL, Beale RJ. Prospective placebo‐controlled randomized trial of lexipafant in predicted severe acute pancreatitis. The British Journal of Surgery 1998;85(2):279‐80. [DOI] [PubMed] [Google Scholar]
Xiong 2009 {published data only}
- Xiong J, Zhu S, Zhou Y, Wu H, Wang C. Regulation of omega‐3 fish oil emulsion on the sirs during the initial stage of severe acute pancreatitis. Journal of Huazhong University of Science and Technology ‐ Medical Science 2009;29(1):35‐8. [DOI] [PubMed] [Google Scholar]
Xu 2012 {published data only}
- Xu QH, Cai GL, Lu XC, Hu CB, Chen J, Yan J. The effects of omega‐3 fish oil lipid emulsion on inflammation‐immune response and organ function in patients with severe acute pancreatitis. Zhonghua Neike Zazhi [Chinese Journal of Internal Medicine] 2012;51(12):962‐5. [PubMed] [Google Scholar]
Yang 2008a {published data only}
- Yang JS, Hou Y, Zuo Y. Effect of combined therapy with sandostatin and yiyan mixture in treating severe acute pancreatitis. Zhongguo Zhongxiyi Jiehe Zqzhi [Chinese Journal of Integrated Traditional and Western Medicine] 2008;28(8):708‐10. [PubMed] [Google Scholar]
Yang 2008b {published data only}
- Yang SQ, Xu JG. Effect of glutamine on serum interleukin‐8 and tumor necrosis factor‐alpha levels in patients with severe pancreatitis. Nanfang Yeie Daxue Xuebao [Journal of Southern Medical University] 2008;28(1):129‐31. [PubMed] [Google Scholar]
Yang 2009 {published data only}
- Yang XN, Deng LH, Xue P, Zhao L, Jin T, Wan MH, et al. Non‐preventive use of antibiotics in patients with severe acute pancreatitis treated with integrated traditional Chinese and western medicine therapy: a randomized controlled trial. Zhongxiyi Jiehe Xuebao [Journal of Chinese Integrative Medicine] 2009;7(4):330‐3. [DOI] [PubMed] [Google Scholar]
Zapater 2000 {published data only}
- Zapater P, Abad‐Santos F, Alcalde‐Rubio M, Moreno‐Otero R. Do muscarinic receptors play a role in acute pancreatitis? A randomised comparison of pirenzepine and nasogastric suction. Clinical Drug Investigation 2000;20(6):401‐8. [Google Scholar]
References to studies awaiting assessment
Hansen 1966 {published data only}
- Hansen HT, Drube HC, Brüning W, Borm D. Therapy of acute pancreatitis with and without proteinase inhibitor. Comparative clinical studies with trasylol [Behandlung der akuten pankreatitis mit und ohne proteinaseninhibitor. Vergleichende klinische untersuchungen mit trasylol]. Medizinische Klinik 1966;61(32):1254‐7. [PubMed] [Google Scholar]
Perez 1980 {published data only}
- Perez Oteyza C, Rebollar J, Ballarin M. Treatment of acute pancreatitis with cimetidine. Double‐blind controlled trial. Hepato‐Gastroenterology 1980;27(Suppl):F8.2. [Google Scholar]
References to ongoing studies
ChiCTR‐IPR‐16008301 {published data only}
- ChiCTR‐IPR‐16008301. The effect of proton pump inhibitors on acute pancreatitis‐‐a randomly prospective control study [The effect of proton pump inhibitors on acute pancreatitis‐‐a randomly prospective control study]. www.chictr.org.cn/showproj.aspx?proj=14089 (first received 18 April 2016).
EUCTR2014‐004844‐37‐ES {published data only}
- EUCTR2014‐004844‐37‐ES. Trial of indomethacin in pancreatitis [A randomized controlled pilot trial of indomethacin in acute pancreatitis]. www.clinicaltrialsregister.eu/ctr‐search/trial/2014‐004844‐37/ES (first received 8 May 2015).
NCT01132521 {published data only}
- NCT01132521. Ulinastatin in severe acute pancreatitis [Multicenter, double‐bind, randomised, placebo controlled study of ulinastatin in severe acute pancreatitis]. clinicaltrials.gov/show/NCT01132521 (first received 26 May 2010).
NCT02025049 {published data only}
- NCT02025049. DP‐b99 in the treatment of acute high‐risk pancreatitis [Pilot trial of intravenous DP‐b99 in the treatment of first‐ever episode of non‐obstructive acute high‐risk pancreatitis]. clinicaltrials.gov/show/NCT02025049 (first received 25 December 2013).
NCT02212392 {published data only}
- NCT02212392. Comparing the outcome in patients of acute pancreatitis, with and without prophylactic antibiotics [Comparing the outcome in patients of acute pancreatitis, with and without prophylactic antibiotics]. clinicaltrials.gov/show/NCT02212392 (first received 5 August 2014).
NCT02692391 {published data only}
- NCT02692391. A randomized controlled pilot trial of indomethacin in acute pancreatitis [A randomized controlled pilot trial of indomethacin in acute pancreatitis]. clinicaltrials.gov/show/NCT02692391 (first received 15 February 2016).
NCT02885441 {published data only}
- NCT02885441. Treatment of acute pancreatitis with ketorolac [Treatment of acute pancreatitis with ketorolac]. clinicaltrials.gov/show/NCT02885441 (first received 23 August 2016).
Additional references
Andriulli 1998
- Andriulli A, Leandro G, Clemente R, Festa V, Caruso N, Annese V, et al. Meta‐analysis of somatostatin, octreotide and gabexate mesilate in the therapy of acute pancreatitis. Alimentary Pharmacology & Therapeutics 1998;12(3):237‐45. [DOI] [PubMed] [Google Scholar]
Ayub 2010
- Ayub K, Slavin J, Imada R. Endoscopic retrograde cholangiopancreatography in gallstone‐associated acute pancreatitis. Cochrane Database of Systematic Reviews 2010, Issue 1. [DOI: 10.1002/14651858.CD003630.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Bang 2008
- Bang UC, Semb S, Nojgaard C, Bendtsen F. Pharmacological approach to acute pancreatitis. World Journal of Gastroenterology 2008;14(19):2968‐76. [DOI] [PMC free article] [PubMed] [Google Scholar]
Banks 2013
- Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, et al. Classification of acute pancreatitis‐‐2012: revision of the Atlanta classification and definitions by international consensus. Gut 2013;62(1):102‐11. [DOI] [PubMed] [Google Scholar]
Bone 1992
- Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 1992;101(6):1644‐55. [DOI] [PubMed] [Google Scholar]
Bradley 1993
- Bradley EL 3rd. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, GA, September 11‐13, 1992. Archives of Surgery 1993;128(5):586‐90. [DOI] [PubMed] [Google Scholar]
Dias 2012a
- Dias S, Welton NJ, Sutton AJ, Ades AE. NICE DSU Technical Support Document 1: Introduction to evidence synthesis for decision making, 2012. www.nicedsu.org.uk/TSD1%20Introduction.final.08.05.12.pdf (accessed 27 March 2014).
Dias 2012b
- Dias S, Welton NJ, Sutton AJ, Caldwell DM, Lu G, Ades AE. NICE DSU Technical Support Document 4: Inconsistency in networks of evidence based on randomised controlled trials, 2012. www.nicedsu.org.uk/TSD4%20Inconsistency.final.08.05.12.pdf (accessed 27 March 2014). [PubMed]
Dias 2012c
- Dias S, Sutton AJ, Welton NJ, Ades AE. NICE DSU Technical Support Document 3: Heterogeneity: subgroups, meta‐regression, bias and bias‐adjustment, 2012. www.nicedsu.org.uk/TSD3%20Heterogeneity.final%20report.08.05.12.pdf (accessed 27 March 2014).
Dias 2013
- Dias S, Welton NJ, Sutton AJ, Ades AE. NICE DSU technical support document 2: a generalised linear modelling framework for pairwise and network meta‐analysis of randomised controlled trials, 2013. www.nicedsu.org.uk/TSD2%20General%20meta%20analysis%20corrected%20Mar2013.pdf (accessed 27 March 2014). [PubMed]
Egger 1997
- Egger M, Davey SG, Schneider M, Minder C. Bias in meta‐analysis detected by a simple, graphical test. BMJ (Clinical Research Ed.) 1997;315(7109):629‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
Elmunzer 2012
- Elmunzer BJ, Scheiman JM, Lehman GA, Chak A, Mosler P, Higgins PD, et al. A randomized trial of rectal indomethacin to prevent post‐ERCP pancreatitis. New England Journal of Medicine 2012;366(15):1414‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
FDA 2006
- Center for Biologics Evaluation and Research, U.S. Food, Drug Administration. Guidance for industry adverse reactions section of labeling for human prescription drug and biological products — Content and format. www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm075057.pdf 2006 (accessed 4th July 2014).
Gao 2015a
- Gao W, Yang HX, Ma CE. The value of BISAP score for predicting mortality and severity in acute pancreatitis: a systematic review and meta‐analysis. PLOS ONE 2015;10(6):e0130412. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hampton 2000
- Hampton JR. Clinical trial safety committees: the devil's spoon. BMJ (Clinical research ed.) 2000;320(7229):244‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327(7414):557‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Hochman 2006
- Hochman D, Louie B, Bailey R. Determination of patient quality of life following severe acute pancreatitis. Canadian Journal of Surgery 2006;49(2):101‐6. [PMC free article] [PubMed] [Google Scholar]
ICH‐GCP 1997
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Code of Federal Regulation & ICH Guidelines. Pennsylvania: Barnett International/PAREXEL, 1997. [Google Scholar]
Jiang 2012
- Jiang K, Huang W, Yang XN, Xia Q. Present and future of prophylactic antibiotics for severe acute pancreatitis. World Journal of Gastroenterology 2012;18(3):279‐84. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lu 2004
- Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Statistics in Medicine 2004;23(20):3105‐24. [DOI] [PubMed] [Google Scholar]
Lu 2007
- Lu G, Ades AE, Sutton AJ, Cooper NJ, Briggs AH, Caldwell DM. Meta‐analysis of mixed treatment comparisons at multiple follow‐up times. Statistics in Medicine 2007;26(20):3681‐99. [DOI] [PubMed] [Google Scholar]
Masood 1998
Messori 1995
- Messori A, Rampazzo R, Scroccaro G, Olivato R, Bassi C, Falconi M, et al. Effectiveness of gabexate mesilate in acute pancreatitis. A metaanalysis. Digestive Diseases and Sciences 1995;40(4):734‐8. [DOI] [PubMed] [Google Scholar]
MHRA 2013
- Medicines and Healthcare products Regulatory Agency (MHRA). Clinical trials for medicines: Safety reporting ‐ SUSARs and DSURs. 2013. www.mhra.gov.uk/Howweregulate/Medicines/Licensingofmedicines/Clinicaltrials/Safetyreporting‐SUSARsandASRs/ (accessed 4 July 2014).
Mills 2012
- Mills EJ, Ioannidis JP, Thorlund K, Schunemann HJ, Puhan MA, Guyatt GH. How to use an article reporting a multiple treatment comparison meta‐analysis. JAMA 2012;308(12):1246‐53. [DOI] [PubMed] [Google Scholar]
NCBI 2014
- NCBI. MeSH. NLM Controlled Vocabulary. Pancreas. www.ncbi.nlm.nih.gov/mesh/68010179 (accessed 4 July 2014).
Neumann 2011
- Neumann I, Grassi B, Bdair F, Rada G. Antiproteases for acute pancreatitis. Cochrane Database of Systematic Reviews 2011, Issue 11. [DOI: 10.1002/14651858.CD009426] [DOI] [Google Scholar]
Newell 1992
- Newell DJ. Intention‐to‐treat analysis: implications for quantitative and qualitative research. International Journal of Epidemiology 1992;21(5):837‐41. [DOI] [PubMed] [Google Scholar]
Peery 2012
- Peery AF, Dellon ES, Lund J, Crockett SD, McGowan CE, Bulsiewicz WJ, et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology 2012;143(5):1179‐87. [DOI] [PMC free article] [PubMed] [Google Scholar]
Pendharkar 2014
- Pendharkar SA, Salt K, Plank LD, Windsor JA, Petrov MS. Quality of life after acute pancreatitis: a systematic review and meta‐analysis. Pancreas 2014;43(8):1194‐200. [DOI] [PubMed] [Google Scholar]
Puhan 2014
- Puhan MA, Schünemann HJ, Murad MH, Li T, Brignardello‐Petersen R, Singh JA, et al. A GRADE Working Group approach for rating the quality of treatment effect estimates from network meta‐analysis. BMJ (Clinical Research Ed.) 2014;349:g5630. [DOI] [PubMed] [Google Scholar]
Rada 2011
- Rada G, Neumann I, Roa M, Rojas L. Antioxidants for acute pancreatitis. Cochrane Database of Systematic Reviews 2011, Issue 3. [DOI: 10.1002/14651858.CD009049] [DOI] [Google Scholar]
RevMan 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Roberts 2013
- Roberts SE, Akbari A, Thorne K, Atkinson M, Evans PA. The incidence of acute pancreatitis: impact of social deprivation, alcohol consumption, seasonal and demographic factors. Alimentary Pharmacology and Therapeutics 2013;38(5):539‐48. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sah 2013
- Sah RP, Dawra RK, Saluja AK. New insights into the pathogenesis of pancreatitis. Current Opinion in Gastroenterology 2013;29(5):523‐30. [DOI] [PMC free article] [PubMed] [Google Scholar]
Salanti 2011
- Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple‐treatment meta‐analysis: an overview and tutorial. Journal of Clinical Epidemiology 2011;64(2):163‐71. [DOI] [PubMed] [Google Scholar]
Schmid 1999
- Schmid SW, Uhl W, Friess H, Malfertheiner P, Buchler MW. The role of infection in acute pancreatitis. Gut 1999;45(2):311‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Tenner 2013
- Tenner S, Baillie J, DeWitt J, Vege SS, American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis. American Journal of Gastroenterology 2013;108(9):1400‐15. [DOI] [PubMed] [Google Scholar]
WHO 2001
- World Health Organization. Essential drugs and medicines – quality assurance and safety of medicines health technology and pharmaceuticals. Pharmaceuticals: restrictions in use and availability. 2001. apps.who.int/medicinedocs/pdf/s2203e/s2203e.pdf (accessed on 5 October 2016).
WinBUGS 1.4 [Computer program]
- Imperial College and MRC. WinBUGS with DoodleBUGS. Version 1.4.3. London, UK: Imperial College and MRC, 2007.
Wright 2009
- Wright SE, Lochan R, Imrie K, Baker C, Nesbitt ID, Kilner AJ, et al. Quality of life and functional outcome at 3, 6 and 12 months after acute necrotising pancreatitis. Intensive Care Medicine 2009;35(11):1974‐8. [DOI] [PubMed] [Google Scholar]
Xu 2013
- Xu W, Zhou YF, Xia SH. Octreotide for primary moderate to severe acute pancreatitis: a meta‐analysis. Hepato‐gastroenterology 2013;60(126):1504‐8. [PubMed] [Google Scholar]
Yadav 2006
- Yadav D, Lowenfels AB. Trends in the epidemiology of the first attack of acute pancreatitis: a systematic review. Pancreas 2006;33(4):323‐30. [DOI] [PubMed] [Google Scholar]
Yang 2008
- Yang AL, Vadhavkar S, Singh G, Omary MB. Epidemiology of alcohol‐related liver and pancreatic disease in the United States. Archives of Internal Medicine 2008;168(6):649‐56. [DOI] [PubMed] [Google Scholar]
Yang 2011
- Yang X, Zeng X, Wu T. Lexipafant for acute pancreatitis. Cochrane Database of Systematic Reviews 2011, Issue 9. [DOI: 10.1002/14651858.CD009309] [DOI] [Google Scholar]
Zhang 2009
- Zhang Y, Chen QB, Gao ZY, Xie WF. Meta‐analysis: octreotide prevents post‐ERCP pancreatitis, but only at sufficient doses. Alimentary Pharmacology & Therapeutics 2009;29(11):1155‐64. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Gurusamy 2014
- Gurusamy KS. Pharmacological interventions for acute pancreatitis: a network meta‐analysis. Cochrane Database of Systematic Reviews 2014, Issue 11. [DOI: 10.1002/14651858.CD011384] [DOI] [Google Scholar]


