Abstract
Background
Smoking is one of the well-established risk factors for gastric cancer incidence, yet whether men are more or equally susceptible to gastric cancer due to smoking compared with women is a matter of controversy. The aim of this study was to investigate and compare the effect of sex on gastric cancer risk associated with smoking.
Methods
We conducted a systemic literature search in MEDLINE, EMBASE, and the Cochrane CENTRAL databases to identify studies published from inception to December 2018. We included prospective observational studies which reported effect estimates with 95% confidence intervals (CIs) for associations of current or former smokers with the incidence of gastric cancer by sex. We calculated the ratio of relative risk (RRR) with corresponding 95% CI based on sex-specific effect estimates for current or former smokers versus non-smokers on the risk of gastric cancer.
Results
We included 10 prospective studies with 3,381,345 participants in our analysis. Overall, the summary RRR (male to female) for gastric cancer risk in current smokers was significantly increased compared with non-smokers (RRR: 1.30; 95% CI: 1.05–1.63; P = 0.019). Furthermore, there was no significant sex difference for the association between former smokers and gastric cancer risk (RRR: 1.20; 95% CI: 0.92–1.55; P = 0.178). However, the result of sensitivity analysis indicated the pooled result was not stable, which was altered by excluding a nested case-control study (RRR: 1.31; 95% CI: 1.10–1.57; P = 0.002).
Conclusion
This systematic review showed a potential sex difference association between current smokers and the risk of gastric cancer. The sex differential in smokers can give important clues for the etiology of gastric cancers and should be examined in further studies.
Electronic supplementary material
The online version of this article (10.1186/s12885-019-5601-9) contains supplementary material, which is available to authorized users.
Keywords: Smoking, Gastric cancer, Cancer risk, Sex, Meta-analysis
Background
Gastric cancer is the fifth most common cancer and the third leading cause of cancer mortality worldwide, despite its decreasing incidence in recent decades [1, 2]. Moreover, there is a sex-specific disparity in gastric cancer incidence. Incidence rates are 2-fold higher in men than in women worldwide [1]. Gastric cancer is a multifactorial disease, and both environmental and genetic factors have a role in its etiology. Common risk factors include older age, Helicobacter pylori infection, coffee, dairy products, red meat consumption, tobacco smoking, radiation, high body mass index, and family history [3–8]. There are also geographic, ethnic, and sex differences in the incidence of gastric cancer.
Previous studies have indicated that environmental factors could affect gastric cancer risk more prominently than genetic factors [9–11]. Several studies have suggested that smoking was associated with a higher risk of gastric cancer and a previous meta-analysis considered smoking to be most important behavioral risk factor for gastric cancer [12]. However, the role of sex differences remain controversial. Clarifying the association of smoking status with the risk of gastric cancer in men compared with women is particularly important since the prevalence of smoking in women is increasing and now tobacco use is seen as a “contemporary epidemic” in women in the United States and many other countries. A potential sex difference could help identify high-risk population groups for gastric cancer in smokers, allowing for the formulation of effective primary prevention strategies. Therefore, we performed a large-scale examination of the available prospective observational studies to explore the association between smoking status and gastric cancer risk by sex. We further evaluated the sex difference according to the baseline characteristics of the participants.
Methods
Data sources, search strategy, and selection criteria
This systematic review was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) Statement issued in 2009 [13]. Relevant articles were systematically searched in MEDLINE, EMBASE, and the Cochrane CENTRAL electronic databases from database inception to December 2018. We included studies that investigated humans without language restrictions and regardless of publication status (published, in the press, or in progress). The studies reporting associations between smoking status and gastric cancer risk were searched using strategies of a combined text and medical subjects headings (MeSH): (“smoke” OR “smoking” OR “nicotine” OR “tobacco” OR “lifestyle” OR “lifestyles” OR “cigarette”) AND (“gastric” OR “stomach” OR “cardia”) AND (“cancer” OR “tumor” OR “neoplasm”) AND (“nested case control” OR “cohort” OR “prospective”). Furthermore, we also manually checked the reference lists of identified reports for other potentially relevant studies. If the same population was reported more than once, the most comprehensive and recently published article was used. The study topic, study design, exposure, population, and reported outcomes were used to identify relevant studies.
Two authors independently performed a literature search and study selection, and disagreements between two authors were settled by a discussion in a group until a consensus was reached. A study was deemed eligible if it met the following inclusion criteria: (1) the study design was a prospective observational study; (2) the study evaluated the association of smoking status with gastric cancer risk; and (3) the associations between smoking status and gastric cancer risk in men and women were both reported.
Data collection and quality assessment
Two authors independently collected and extracted data from the included studies, and disagreements were resolved by a group discussion. The data collected from the included studies contained the following items: first author, publication year, country, sex, sample size of men and women, mean age for men and women, number of participants who had never smoked (non-smokers) for men and women, number of former smokers in men and women, number of current smokers in men and women, follow-up duration, reported outcomes, and adjusted factors.
We assessed the methodological quality of the included studies using the Newcastle-Ottawa Scale (NOS) [14], which has been partially validated for evaluating the quality of observational studies included in meta-analyses. The NOS is based on selection (4 items), comparability (1 item), and outcome (3 items), and provides a “star system” range of 0–9 to evaluate study quality. Two authors independently performed quality assessments and disagreements were settled by a group discussion.
Statistical analysis
The associations between smoking status and gastric cancer risk in men and women were determined based on the relative risk (RR), hazard ratio (HR), or odds ratio (OR), and the 95% confidence intervals (CIs) in each individual study. HR is considered equivalent to RR in prospective observational studies, and OR could also be assumed to be equivalent to the RR due to the low incidence of gastric cancer. We calculated the ratio of RRs (RRR) for current or former smokers versus non-smokers and the risk of gastric cancer based on sex-specific RRs in individual studies [15]. We used random-effects models to calculate the summary RRR and compared the sex differences in gastric cancer risk in current smokers, former smokers, or non-smokers [16, 17].
Heterogeneity among studies was shown by the I2 and Q statistics, and P values < 0.10 mean significant heterogeneity [18, 19]. A sensitivity analysis was performed by systematically excluding each study individually to evaluate its influence on the meta-analysis [20]. The potential sources of heterogeneity in estimates of the impact of current and former smokers based on follow-up duration were explored by using univariate meta-regression [21]. Subgroup analyses for the sex differences in the association between smoking status and gastric cancer risk were based on publication year, country, follow-up duration, reported outcomes, whether or not the studies adjusted for BMI or alcohol consumption, and study quality. Publication bias was explored visually using funnel plots and statistically using Egger’s and Begg’s tests [22, 23]. All P values were two-sided with significance defined as P < 0.05. Statistical analyses were conducted using STATA software (version 10.0; Stata Corporation, College Station, TX, USA).
Results
Literature search
A total of 1517 records from the initial search were identified, including 691 from MEDLINE, 757 from EMBASE, and 69 from the Cochrane CENTRAL. After discarding 1423 irrelevant or duplicate studies, 94 potential studies were selected for further reading. After detailed evaluating, 10 prospective observational studies were selected into the quantitative analysis [24–33]. The manual search of the reference lists of these studies did not yield any new eligible studies. The systematic review selection process is shown in Fig. 1, and the general characteristics of the included studies are displayed in Table 1.
Fig. 1.
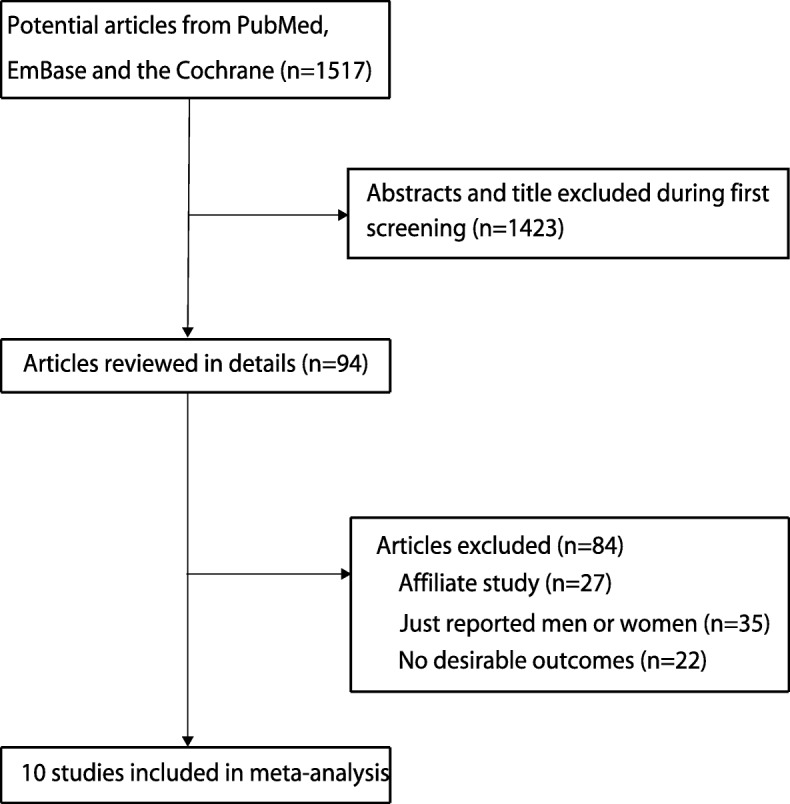
Flow diagram of the literature search and trial selection process
Table 1.
Baseline characteristic of studies included in the systematic review and meta-analysis
| Study | Country | Sex | Sample size | Age (years) | Never smoker | Former smoker | Current smoker | Follow-up (years) | Reported outcomes | Adjusted factors | NOS score |
|---|---|---|---|---|---|---|---|---|---|---|---|
| JACC 2005 [24] | Japan | Men | 43,482 | 57.4 | 9014 | 11,471 | 22,997 | 9.9 | GC mortality | Age, smoking, alcohol intake, educational level, preference for salty foods and green-tea intake. | 7 |
| Women | 43,482 | 57.4 | 9014 | 11,471 | 22,997 | ||||||
| Kato 1992 [25] | Japan | Men | NA | > 30.0 | NA | NA | NA | 6.0 | GC mortality | Age | 6 |
| Women | NA | > 30.0 | NA | NA | NA | ||||||
| Tverdal 1993 [26] | Norway | Men | 44,290 | 35.0–49.0 | 9334 | 10,467 | 18,400 | 13.3 | GC mortality | Age, cholesterol, SBP, PI during leisure, BMI, height and number of cigarettes. | 7 |
| Women | 24,535 | 35.0–49.0 | 12,369 | 3079 | 8954 | ||||||
| Engeland 1996 [27] | Norway | Men | 8905 | 23.0–57.0 | NA | NA | NA | 28.0 | GC | NA | 7 |
| Women | 10,169 | 18.0–57.0 | NA | NA | NA | ||||||
| Lindblad 2005 [28] | UK | Men | 6860 | 40.0–84.0 | 2956 | 678 | 1212 | NA | GC | Age, calendar year, BMI, alcohol consumption and reflux | 6 |
| Women | 3335 | 40.0–84.0 | 1744 | 168 | 395 | ||||||
| Jee 2004 [29] | Korea | Men | 830,139 | 45.0 | 166,858 | 190,932 | 473,179 | 8.0 | GC mortality | Age | 8 |
| Women | 382,767 | 48.6 | 352,528 | 9569 | 20,669 | ||||||
| Chao 2002 [30] | US | Men | 467,788 | 57.0 | 117,968 | 179,833 | 151,406 | 14.0 | GC mortality | Age, race, education, family history of stomach cancer, consumption of high-fiber grain foods, vegetables, citrus fruits or juices, and use of vitamin C, multivitamins, and aspirin | 8 |
| Women | 588,053 | 56.0 | 326,835 | 122,455 | 122,465 | ||||||
| Akiba 1990 [31] | Japan | Men | 122,261 | > 40.0 | NA | NA | NA | 16.0 | GC mortality | Prefecture of residence, occupation, attained age, and observation period | 6 |
| Women | 142,857 | > 40.0 | NA | NA | NA | ||||||
| Gonzalez 2003 [32] | Europe (10 countries) | Men | 148,182 | 51.6 | 49,678 | 36,791 | 36,643 | 5.0 | GC | Age, sex, vegetables, fruits, processed meat, alcohol, BMI and educational level | 7 |
| Women | 322,046 | 51.6 | 191,037 | 41,887 | 58,319 | ||||||
| Nomura 2012 [33] | US | Men | 82,683 | 60.1 | 25,466 | 42,251 | 14,966 | 7.3 | GC | Age at cohort entry as a continuous variable, ethnicity as a strata variable, education, processed meat intake, BMI, alcohol intake, aspirin use, and family history of gastric cancer | 8 |
| Women | 99,758 | 59.6 | 56,463 | 28,930 | 14,365 |
GC gastric cancer, SBP systolic blood pressure, PI physical activity, BMI body mass index
Study characteristics
Ten studies with a total of 3,381,345 participants were included in our analysis. Among the studies, nine were prospective cohort studies [24–27, 29–33] and one was a nested case-control study [28]. The duration of follow-up for participants was 5.0–28.0 years, while 9753-1,212,906 individuals were included in each study. Three studies were conducted in Japan [24, 25, 31], one in Korea [30], two in Norway [26, 27], one in the UK [28], two in the US [30, 33], and one in 10 European countries [32]. The main study outcome in 6 studies was gastric cancer mortality, and the remaining 4 studies reported gastric cancer incidence. NOS scores were used to evaluate study quality [14], and a score ≥ 7 was regarded as high quality. Overall, three studies had scores of 8, four studies had scores of 7, and the remaining three studies had scores of 6.
Sex differences for gastric Cancer risk in current smokers
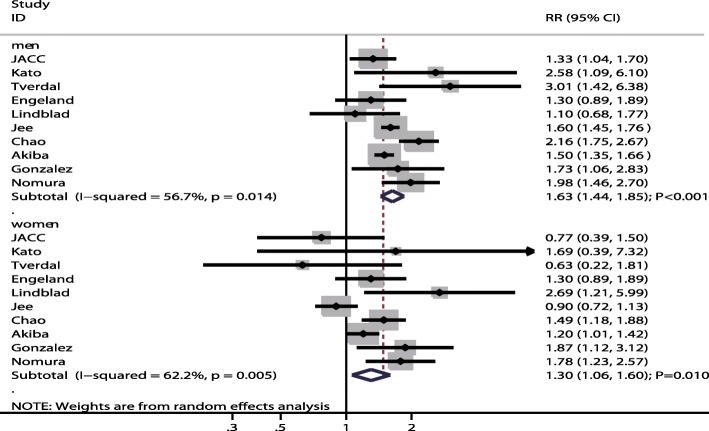
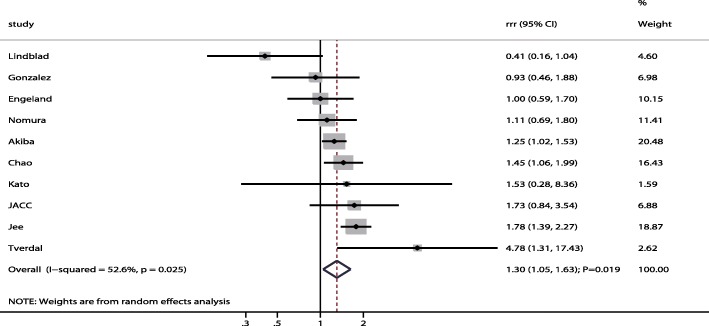
All included studies reported sex differences in the association between gastric cancer risk and current smokers compared with non-smokers. We noted current smokers were associated with higher risk of gastric cancer when compared with non-smokers in men (RR: 1.63; 95% CI: 1.44–1.85; P < 0.001; Fig. 2) and women (RR: 1.30; 95% CI: 1.06–1.60; P = 0.010; Fig. 2). Further, the increased risk of gastric cancer in current smokers compared to non-smokers was higher in men than in women (RRR: 1.30; 95% CI: 1.05–1.63; P = 0.019; Fig. 3), with significant heterogeneity (I2 = 52.6%; P = 0.025). The result of the sensitivity analysis indicated that the sex differences in the association between current smokers and gastric cancer were affected by the exclusion of multiple studies due to the small numbers of cohorts included (Table 2). The results of the meta-regression analysis showed that follow-up duration was not a significant factor contributing to the sex differences of the association between current smokers and gastric cancer (Additional file 1). We used subgroup analyses to minimize heterogeneity among the included studies and evaluate the sex differences in subpopulations (Table 3). The summary RRR (male to female) for current smokers indicated an increased risk of gastric cancer in men when the study was conducted in Asia (RRR: 1.50; 95% CI: 1.17–1.91; P = 0.001), regardless of follow-up duration (follow-up duration ≥10.0 years [RRR: 1.33; 95% CI: 1.02–1.74; P = 0.037]; follow-up duration < 10.0 years [RRR: 1.46; 95% CI: 1.11–1.91; P = 0.006]), when the study reported gastric cancer mortality (RRR: 1.53; 95% CI: 1.24–1.89; P < 0.001), when the study did not adjust for BMI (RRR: 1.47; 95% CI: 1.24–1.74; P < 0.001), when the study did not adjust for alcohol consumption (RRR: 1.53; 95% CI: 1.20–1.94; P = 0.001), and when the study had a NOS score of 7 or 8 (RRR: 1.42; 95% CI: 1.11–1.81; P = 0.005).
Fig. 2.
The associations of current smokers with the risk of gastric cancer in men and women separately
Fig. 3.
Sex difference of the association between current smokers and the risk of gastric cancer
Table 2.
Sensitivity analysis for sex difference of gastric cancer (current smoker versus never smoker and former smoker versus never smoker)
| Outcomes | Excluding study | RRR and 95% CI | P value | Heterogeneity (%) | P value for heterogeneity |
|---|---|---|---|---|---|
| Current smoker versus never smoker | JACC 2005 | 1.27 (1.01–1.61) | 0.045 | 56.8 | 0.018 |
| Kato 1992 | 1.30 (1.03–1.63) | 0.026 | 57.8 | 0.015 | |
| Tverdal 1993 | 1.27 (1.04–1.57) | 0.022 | 47.8 | 0.053 | |
| Engeland 1996 | 1.34 (1.06–1.70) | 0.015 | 54.5 | 0.024 | |
| Lindblad 2005 | 1.38 (1.15–1.66) | 0.001 | 35.7 | 0.132 | |
| Jee 2004 | 1.22 (0.97–1.53) | 0.089 | 38.1 | 0.114 | |
| Chao 2002 | 1.27 (0.97–1.66) | 0.080 | 57.4 | 0.016 | |
| Akiba 1990 | 1.30 (0.98–1.73) | 0.068 | 55.1 | 0.023 | |
| Gonzalez 2003 | 1.34 (1.06–1.69) | 0.014 | 55.0 | 0.023 | |
| Nomura 2012 | 1.33 (1.04–1.70) | 0.023 | 56.1 | 0.020 | |
| Former smoker versus never smoker | JACC 2005 | 1.20 (0.90–1.59) | 0.220 | 48.4 | 0.060 |
| Kato 1992 | 1.20 (0.92–1.58) | 0.175 | 46.9 | 0.068 | |
| Tverdal 1993 | 1.20 (0.91–1.58) | 0.187 | 47.7 | 0.063 | |
| Engeland 1996 | 1.15 (0.87–1.53) | 0.332 | 47.7 | 0.063 | |
| Lindblad 2005 | 1.31 (1.10–1.57) | 0.002 | 5.7 | 0.386 | |
| Jee 2004 | 1.10 (0.78–1.56) | 0.592 | 46.2 | 0.072 | |
| Chao 2002 | 1.17 (0.83–1.64) | 0.364 | 46.0 | 0.073 | |
| Gonzalez 2003 | 1.26 (0.96–1.64) | 0.095 | 40.8 | 0.107 | |
| Nomura 2012 | 1.11 (0.85–1.44) | 0.445 | 28.8 | 0.199 |
Table 3.
Subgroup analysis for sex difference of gastric cancer (current smoker versus never smoker)
| Group | RRR and 95% CI | P value | Heterogeneity (%) | P value for heterogeneity | P value for interaction test |
|---|---|---|---|---|---|
| Publication year | |||||
| 2000 or after | 1.27 (0.93–1.74) | 0.130 | 60.6 | 0.027 | 0.921 |
| Before 2000 | 1.32 (0.89–1.96) | 0.172 | 38.2 | 0.183 | |
| Country | |||||
| Asia | 1.50 (1.17–1.91) | 0.001 | 40.2 | 0.171 | 0.306 |
| Europe or US | 1.12 (0.77–1.64) | 0.548 | 58.2 | 0.035 | |
| Follow-up duration (years) | |||||
| 10 or greater | 1.33 (1.02–1.74) | 0.037 | 44.7 | 0.143 | 0.573 |
| < 10 | 1.46 (1.11–1.91) | 0.006 | 22.4 | 0.272 | |
| Outcomes | |||||
| GC incidence | 0.92 (0.66–1.29) | 0.628 | 16.5 | 0.309 | 0.023 |
| GC mortality | 1.53 (1.24–1.89) | < 0.001 | 39.3 | 0.143 | |
| Adjusted BMI or not | |||||
| Yes | 1.07 (0.53–2.15) | 0.848 | 68.0 | 0.025 | 0.527 |
| No | 1.47 (1.24–1.74) | < 0.001 | 20.2 | 0.286 | |
| Adjusted alcohol consumption | |||||
| Yes | 0.99 (0.61–1.61) | 0.971 | 49.6 | 0.114 | 0.331 |
| No | 1.53 (1.20–1.94) | 0.001 | 50.2 | 0.090 | |
| NOS score | |||||
| 7 or 8 | 1.42 (1.11–1.81) | 0.005 | 43.4 | 0.101 | 0.169 |
| < 7 | 0.91 (0.41–2.02) | 0.814 | 62.7 | 0.068 | |
Sex differences in gastric Cancer risk for former smokers
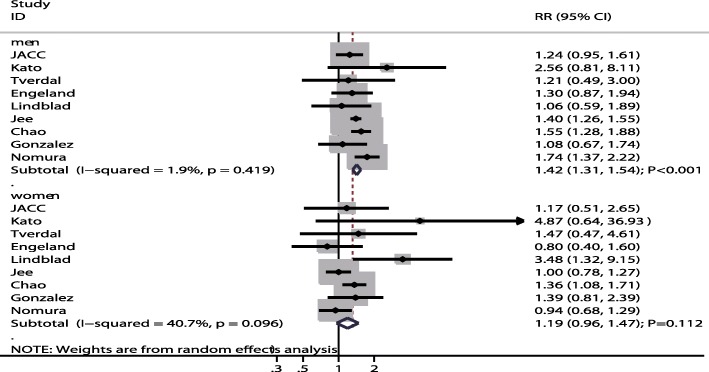
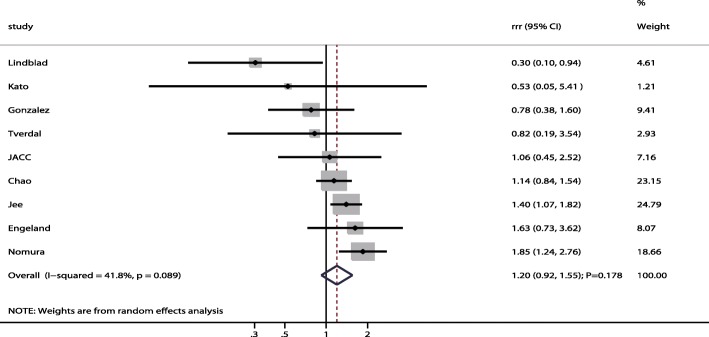
A total of 9 studies reported sex differences in the relation between gastric cancer risk in former smokers compared to non-smokers. The summary result indicated former smokers were associated with an increased risk of gastric cancer in men (RR: 1.42; 95% CI: 1.31–1.54; P < 0.001; Fig. 4), while this association was not associated with statistically significant in women (RR: 1.19; 95% CI: 0.96–1.47; P = 0.112; Fig. 4). There was no significant difference for gastric cancer risk between former smokers and non-smokers in men compared with women (RRR: 1.20; 95% CI: 0.92–1.55; P = 0.178; Fig. 5), and potential significant heterogeneity was observed among the included studies (I2 = 41.8%; P = 0.089). Following the result of the sensitivity analysis, we excluded the study by Lindblad et al. [28], which used a nested case control design. After this exclusion, we could conclude that male former smokers had a significantly increased risk of gastric cancer over non-smokers compared to female former smokers (RRR: 1.31; 95% CI: 1.10–1.57; P = 0.002; Table 2). Meta-regression analysis indicated follow-up duration did not contribute a significant role with the sex difference of the relation between former smokers and gastric cancer (Additional file 1). Subgroup analyses indicated a higher risk of gastric cancer in male verses female former smokers when the study was conducted in Asia (RRR: 1.35; 95% CI: 1.05–1.74; P = 0.019; Table 4), follow-up duration < 10.0 years (RRR: 1.35; 95% CI: 1.02–1.80; P = 0.038), when the study reported gastric cancer mortality (RRR: 1.25; 95% CI: 1.03–1.51; P = 0.022; Table 4), when the study did not adjust for BMI (RRR: 1.26; 95% CI: 1.04–1.53; P = 0.019; Table 4), when the study did not adjust for alcohol consumption (RRR: 1.26; 95% CI: 1.04–1.53; P = 0.020; Table 4), and when the study had high study quality (RRR: 1.32; 95% CI: 1.09–1.59; P = 0.004; Table 4). Furthermore, male former smokers were associated with a lower risk of gastric cancer if the study had lower study quality (RRR: 0.34; 95% CI: 0.12–0.93; P = 0.036).
Fig. 4.
The associations of former smokers with the risk of gastric cancer in men and women separately
Fig. 5.
Sex difference of the association between former smoker and the risk of gastric cancer
Table 4.
Subgroup analysis for sex difference of gastric cancer (former smoker versus never smoker)
| Group | RRR and 95% CI | P value | Heterogeneity (%) | P value for heterogeneity | P value for interaction test |
|---|---|---|---|---|---|
| Publication year | |||||
| 2000 or after | 1.17 (0.85–1.59) | 0.332 | 60.0 | 0.029 | 0.731 |
| Before 2000 | 1.28 (0.65–2.51) | 0.471 | 0.0 | 0.534 | |
| Country | |||||
| Asia | 1.35 (1.05–1.74) | 0.019 | 0.0 | 0.606 | 0.528 |
| Europe or US | 1.10 (0.73–1.66) | 0.648 | 59.7 | 0.029 | |
| Follow-up duration (years) | |||||
| 10 or greater | 1.17 (0.89–1.55) | 0.252 | 0.0 | 0.638 | 0.417 |
| < 10 | 1.35 (1.02–1.80) | 0.038 | 27.3 | 0.240 | |
| Outcomes | |||||
| GC incidence | 1.03 (0.51–2.07) | 0.936 | 73.9 | 0.009 | 0.436 |
| GC mortality | 1.25 (1.03–1.51) | 0.022 | 0.0 | 0.726 | |
| Adjusted BMI or not | |||||
| Yes | 0.86 (0.38–1.93) | 0.714 | 74.2 | 0.009 | 0.171 |
| No | 1.26 (1.04–1.53) | 0.019 | 0.0 | 0.630 | |
| Adjusted alcohol consumption | |||||
| Yes | 0.92 (0.45–1.89) | 0.828 | 73.9 | 0.009 | 0.213 |
| No | 1.26 (1.04–1.53) | 0.020 | 0.0 | 0.592 | |
| NOS score | |||||
| 7 or 8 | 1.32 (1.09–1.59) | 0.004 | 12.1 | 0.337 | 0.001 |
| < 7 | 0.34 (0.12–0.93) | 0.036 | 0.0 | 0.680 | |
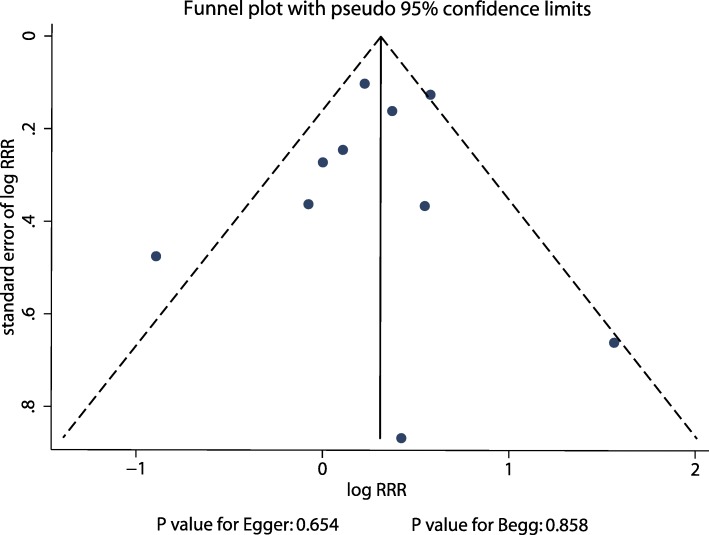
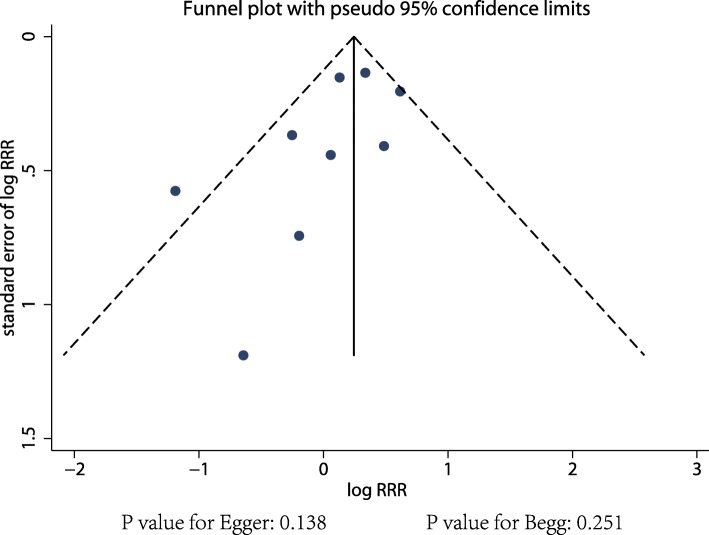
Publication Bias
Reviewing the funnel plots could not rule out the potential publication bias contributing to the sex differences in gastric cancer risk. The Egger’s and Begg’s test results showed no evidence of publication bias for sex differences in the association between current smokers and gastric cancer risk (Fig. 6). Moreover, there was no significant publication bias for former smokers and gastric cancer risk (Fig. 7).
Fig. 6.
Funnel plots for current smokers versus non-smokers
Fig. 7.
Funnel plots for former smokers versus non-smokers
Discussion
This analysis explored sex differences in the associations between smoking status and gastric cancer risk based on 10 prospective observational studies. In total, 3,381,345 participants from 9 prospective cohort studies and 1 nested case-control study were included with a broad range of characteristics. The results of our study suggested that male current smokers had a significantly higher risk of gastric cancer compared to women, while no sex differences were found for the association between former smokers and gastric cancer risk. Sensitivity and subgroup analyses might prove variable due to different baseline characteristics.
A previous study indicated that current smokers in men (RR: 1.62; 95% CI: 1.50–1.75) and women (RR: 1.20; 95% CI: 1.01–1.43) were associated with a significantly increased risk of gastric cancer when compared to non-smokers [12]. Furthermore, Tredaniel et al. indicated that the risk of gastric cancer among smokers was significantly increased compared with non-smokers, and the summary RR was higher in men than women (RR: 1.59 vs 1.11) [9]. Koizumi et al. pooled analyses of two prospective cohort studies in Japan and concluded that gastric cancer risk for current smokers (RR: 1.84; 95% CI: 1.39–2.43) and former smokers (RR: 1.77; 95% CI: 1.29–2.43) were increased compared to non-smokers [34]. Nishino et al. found that current smoking significantly increased the risk of gastric cancer in men (RR: 1.79; 95% CI: 1.51–2.12) and women (RR: 1.22; 95% CI: 1.07–1.38) [35]. The inherent limitations of those previous meta-analyses included the following: (1) although the risk of gastric cancer was different between men and women, the results from different studies might contribute important heterogeneity due to different populations; (2) the included participants were not reported with separate effect estimates in men and women, and there was no direct comparison in sex differences; (3) they did not calculate the associations between smoking status and gastric cancer risk in men and women; and (4) the study combined retrospective and prospective observational studies, which might introduce potential confounders. Therefore, we conducted a meta-analysis of prospective observational studies to evaluate sex differences in the relation between smoking status and gastric cancer risk.
The summary RRR indicated that male current smokers had a greater risk of gastric cancer than women. However, several studies included in our study reported inconsistent results. Lindblad et al. indicated that female current or former smokers were significantly associated with a higher risk of gastric cancer than non-smokers, while this association was not statistically significant in men [28]. The reason for this difference might be because this study was specifically designed as a nested case-control study, and there were imbalances in the number of participants in each smoking category. Gonzalez et al. indicated that current smokers associated with a higher risk of gastric cancer for both men and women, while there was no significant difference finding for gastric cancer risk between former smokers and non-smokers in men or women [32]. Engeland et al. indicated that current smokers or former smokers had no significant change in the risk for gastric cancer when compared with non-smokers for men or women [27]. Our analysis found that male current smokers had a significantly increased risk of gastric cancer, while no significant effect was shown in women [24, 25]. This higher risk of gastric cancer in male smokers than in female smokers might be attributed to a fewer number of cigarettes smoked and shorter smoking duration for women than men. In addition, the risk of gastric cancer due to smoking was higher in men than women, which might affect the sex difference of smoking status and subsequent risk of gastric cancer [36]. Finally, the high rate of alcohol consumption in men was significantly associated with the prevalence of smoking, especially for alcoholism, which was associated with an increased risk of gastric cancer [37].
The findings of the subgroup analyses indicated that the sex differences in gastric cancer risk for current smokers might be affected by country, reported outcomes, whether BMI or alcohol were adjusted for, and study quality. Male former smokers were associated with a higher risk of gastric cancer than female former smokers when the study was conducted in Asia, the outcome was gastric cancer mortality, the study did not adjust for BMI or alcohol consumption, and the study had high study quality. However, female former smokers were associated with a higher risk of gastric cancer than men when the included studies had lower study quality. One possible reason for the locational difference could be that different types of tobacco available between Asian and Western countries, which could have different effects on gastric cancer risk. Furthermore, men might smoke more cigarettes and have a longer duration of smoking than women, which might affect the gastric cancer mortality. Finally, the findings of the subgroup analyses may be variable due to the small cohorts included for several subsets. Therefore, a synthetic and comprehensive review was provided in this study.
We had three strengths in our study that should be highlighted. First, only prospective observational studies were included, which should eliminate the selection and recall biases inherent in retrospective observational studies. Second, the large sample size allowed us to quantitatively assess the association of smoking status and risk of gastric cancer, thus our findings are potentially more robust than the individual studies. Third, sex differences in the associations between smoking status and the risk of gastric cancer were directly compared among individual studies.
The limitations of our study were as follows: (1) the adjusted models were different in the included studies, and these factors might play essential roles in the development of gastric cancer; (2) the history of Helicobacter pylori infection is an important factor which is associated with a higher risk of gastric cancer, but none of the included studies adjusted for Helicobacter pylori and corresponding treatment strategies [3]; (3) the sex differences of the association between smoking status and gastric cancer risk were using dose-response meta-analytic approach, while cigarette smoke exposure as a continuous variable was not available in included studies; (4) although we did not find significant bias in our present work, publication bias was still an inevitable problem in a meta-analysis of published studies; and (5) the analysis used pooled data (individual data were not available) could not provide a more detailed relevant analysis and more comprehensive results.
Conclusion
The results of this study suggested that current smoking might have a more important effect on gastric cancer risk in men than women, while no sex differences were found for the association between former smokers and gastric cancer risk. Furthermore, potential sex difference for the association between former smokers and gastric cancer risk was observed through sensitivity analysis. In addition, this significant sex-difference mainly focused on gastric cancer mortality, while no sex-difference of current or former smoking on gastric cancer incidence. Several factors might affect this sex difference in the risk of gastric cancer, and future studies should focus on other impact factors to analyze the sex difference of gastric cancer.
Additional file
Supplemental 1. Meta-regression analyses for the sex difference of the association between current or former smokers and gastric cancer based on follow-up duration. (DOC 6972 kb)
Acknowledgements
Not applicable
Funding
This work was supported by the National Natural Science Foundation of China (No. 81272718, 81302125 and 81372550). The sponsors played no role in the study design, data collection, or analysis, or decision to submit the article for publication.
Availability of data and materials
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Abbreviations
- CIs
confidence intervals
- HR
hazard ratio
- MeSH
medical subjects headings
- NOS
Newcastle-Ottawa Scale
- OR
odds ratio
- PRISMA
reporting items for systematic reviews and meta-Analysis
- RR
relative risk
- RRR
ratio of relative risk
Authors’ contributions
WYL and ZFM designed this study. YNH, YYX, YXS and HX collected and analyzed the data. SCY, WYL and ZFM drafted and revised the manuscript. XYL, HMX and ZNW interpreted and revised the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Wen-Ya Li, Email: liwycmu@163.com.
Yunan Han, Email: yunanhan@wustl.edu.
Hui-Mian Xu, Email: 13898829926@163.com.
Zhen-Ning Wang, Email: josieon826@sina.cn.
Ying-Ying Xu, Email: yyxu_cmu@163.com.
Yong-Xi Song, Email: songyongxi840309@126.com.
Hao Xu, Email: 13840210201@163.com.
Song-Cheng Yin, Email: 707820302@qq.com.
Xing-Yu Liu, Email: vivavg@163.com.
Zhi-Feng Miao, Phone: 8624-83283556, Email: zfmiao@cmu.edu.cn.
References
- 1.Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Ferlay J, Colombet M, Soerjomataram I, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2018. 10.1002/ijc.31937. [DOI] [PubMed]
- 3.Bae JM, Kim EH. Helicobacter pylori infection and risk of gastric Cancer in Korea: a quantitative systematic review. J Prev Med Public Health. 2016;49:197–204. doi: 10.3961/jpmph.16.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Deng W, Yang H, Wang J, et al. Coffee consumption and the risk of incident gastric cancer--a meta-analysis of prospective cohort studies. Nutr Cancer. 2016;68:40–47. doi: 10.1080/01635581.2016.1115093. [DOI] [PubMed] [Google Scholar]
- 5.Sun Y, Lin LJ, Sang LX, et al. Dairy product consumption and gastric cancer risk: a meta-analysis. World J Gastroenterol. 2014;20:15879–15898. doi: 10.3748/wjg.v20.i42.15879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Song P, Lu M, Yin Q, et al. Red meat consumption and stomach cancer risk: a meta-analysis. J Cancer Res Clin Oncol. 2014;140:979–992. doi: 10.1007/s00432-014-1637-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen Y, Liu L, Wang X, et al. Body mass index and risk of gastric cancer: a meta-analysis of a population with more than ten million from 24 prospective studies. Cancer Epidemiol Biomark Prev. 2013;22:1395–1408. doi: 10.1158/1055-9965.EPI-13-0042. [DOI] [PubMed] [Google Scholar]
- 8.Bonequi P, Meneses-González F, Correa P, et al. Risk factors for gastric cancer in Latin America: a meta-analysis. Cancer Causes Control. 2013;24:217–231. doi: 10.1007/s10552-012-0110-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tredaniel J, Boffetta P, Buiatti E, et al. Tobacco smoking and gastric cancer: review and meta-analysis. Int J Cancer. 1997;72:565–573. doi: 10.1002/(SICI)1097-0215(19970807)72:4<565::AID-IJC3>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 10.Barstad B, Sorensen TI, Tjonneland A, et al. Intake of wine, beer and spirits and risk of gastric cancer. Eur J Cancer Prev. 2005;14:239–243. doi: 10.1097/00008469-200506000-00007. [DOI] [PubMed] [Google Scholar]
- 11.Franke A, Teyssen S, Singer MV. Alcohol-related diseases of the esophagus and stomach. Dig Dis. 2005;23:204–213. doi: 10.1159/000090167. [DOI] [PubMed] [Google Scholar]
- 12.Ladeiras-Lopes R, Pereira AK, Nogueira A, et al. Smoking and gastric cancer: systematic review and meta-analysis of cohort studies. Cancer Causes Control. 2008;19:689–701. doi: 10.1007/s10552-008-9132-y. [DOI] [PubMed] [Google Scholar]
- 13.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wells G, Shea Bm O'Connell D. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm. 2009.
- 15.Huxley R, Barzi F, Woodward M. Excess risk of fatal coronary heart disease associated with diabetes in men and women: meta-analysis of 37 prospective cohort studies. BMJ. 2006;332:73–78. doi: 10.1136/bmj.38678.389583.7C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 17.Ades AE, Lu G, Higgins JP. The interpretation of random-effects metaanalysis in decision models. Med Decis Mak. 2005;25:646–654. doi: 10.1177/0272989X05282643. [DOI] [PubMed] [Google Scholar]
- 18.Deeks JJ, Higgins JP, Altman DG. Analyzing data and undertaking meta-analyses. In: Higgins J, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions 5.0.1. Oxford, UK: The Cochrane Collaboration: 2008; chap 9.
- 19.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tobias A. Assessing the influence of a single study in meta-analysis. Stata Tech Bull. 1999;47:15–17. [Google Scholar]
- 21.Thompson SG, Higgins JP. How should meta-regression analyses be undertaken and interpreted? Stat Med. 2002;21:1559–1573. doi: 10.1002/sim.1187. [DOI] [PubMed] [Google Scholar]
- 22.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–1101. doi: 10.2307/2533446. [DOI] [PubMed] [Google Scholar]
- 24.Fujino Y, Mizoue T, Tokui N, et al. Cigarette smoking and mortality due to stomach Cancer: findings from the JACC study. J Epidemiol. 2005;15(Suppl 2):S113–S119. doi: 10.2188/jea.15.S113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kato I, Tominaga S, Matsumoto K. A prospective study of stomach cancer among a rural Japanese population: a 6-year survey. Jpn J Cancer Res. 1992;83:568–575. doi: 10.1111/j.1349-7006.1992.tb00127.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tverdal A, Thelle D, Stensvold I, et al. Mortality in relation to smoking history: 13 years follow-up of 68,000 Norwegian men and women 35-49 years. J Clin Epidemiol. 1993;46:475–487. doi: 10.1016/0895-4356(93)90025-V. [DOI] [PubMed] [Google Scholar]
- 27.Engeland A, Andersen A, Haldorsen T, et al. Smoking habits and risk of cancers other than lung cancer: 28 years' follow-up of 26,000 Norwegian men and women. Cancer Causes Control. 1996;7:497–506. doi: 10.1007/BF00051881. [DOI] [PubMed] [Google Scholar]
- 28.Lindblad M, LA G’a R’g, Lagergren J. Body mass, tobacco and alcohol and risk of esophageal, gastric cardia, and gastric non-cardia adenocarcinoma among men and women in a nested case-control study. Cancer Causes Control. 2005;16:285–294. doi: 10.1007/s10552-004-3485-7. [DOI] [PubMed] [Google Scholar]
- 29.Jee SH, Samet JM, Ohrr H, et al. Smoking and cancer risk in Korean men and women. Cancer Causes Control. 2004;15:341–348. doi: 10.1023/B:CACO.0000027481.48153.97. [DOI] [PubMed] [Google Scholar]
- 30.Chao A, Thun MJ, Henley SJ, et al. Cigarette smoking, use of other tobacco products and stomach cancer mortality in US adults: the Cancer prevention study II. Int J Cancer. 2002;101:380–389. doi: 10.1002/ijc.10614. [DOI] [PubMed] [Google Scholar]
- 31.Akiba S, Hirayama T. Cigarette smoking and cancer mortality risk in Japanese men and women--results from reanalysis of the six-prefecture cohort study data. Environ Health Perspect. 1990;87:19–26. doi: 10.1289/ehp.908719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.González CA, Pera G, Agudo A, et al. Smoking and the risk of gastric cancer in the European prospective investigation into Cancerand nutrition (EPIC) Int J Cancer. 2003;107:629–634. doi: 10.1002/ijc.11426. [DOI] [PubMed] [Google Scholar]
- 33.Nomura AM, Wilkens LR, Henderson BE, et al. The association of cigarette smoking with gastric cancer: the multiethnic cohort study. Cancer Causes Control. 2012;23:51–58. doi: 10.1007/s10552-011-9854-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Koizumi Y, Tsubono Y, Nakaya N, et al. Cigarette smoking and the risk of gastric cancer: a pooled analysis of two prospective studies in Japan. Int J Cancer. 2004;112:1049–1055. doi: 10.1002/ijc.20518. [DOI] [PubMed] [Google Scholar]
- 35.Nishino Y, Inoue M, Tsuji I, et al. Tobacco smoking and gastric cancer risk: an evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn J Clin Oncol. 2006;36:800–807. doi: 10.1093/jjco/hyl112. [DOI] [PubMed] [Google Scholar]
- 36.Hoshiyama Y, Sasaba T. A case-control study of stomach cancer and its relation to diet, cigarettes and alcohol consumption in Saitama prefecture, Japan. Cancer Causes Control. 1992;3:441–448. doi: 10.1007/BF00051357. [DOI] [PubMed] [Google Scholar]
- 37.Tramacere I, Negri E, Pelucchi C, et al. A meta-analysis on alcohol drinking and gastric cancer risk. Ann Oncol. 2012;23:28–36. doi: 10.1093/annonc/mdr135. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental 1. Meta-regression analyses for the sex difference of the association between current or former smokers and gastric cancer based on follow-up duration. (DOC 6972 kb)
Data Availability Statement
All data generated or analyzed during this study are included in this published article [and its supplementary information files].