Abstract
Background
The sustained interest in electronic assistive technology in dementia care has been fuelled by the urgent need to develop useful approaches to help support people with dementia at home. Also the low costs and wide availability of electronic devices make it more feasible to use electronic devices for the benefit of disabled persons. Information Communication Technology (ICT) devices designed to support people with dementia are usually referred to as Assistive Technology (AT) or Electronic Assistive Technology (EAT). By using AT in this review we refer to electronic assistive devices. A range of AT devices has been developed to support people with dementia and their carers to manage their daily activities and to enhance safety, for example electronic pill boxes, picture phones, or mobile tracking devices. Many are commercially available. However, the usefulness and user‐friendliness of these devices are often poorly evaluated. Although reviews of (electronic) memory aids do exist, a systematic review of studies focusing on the efficacy of AT for memory support in people with dementia is lacking. Such a review would guide people with dementia and their informal and professional carers in selecting appropriate AT devices.
Objectives
Primary objective
To assess the efficacy of AT for memory support in people with dementia in terms of daily performance of personal and instrumental activities of daily living (ADL), level of dependency, and admission to long‐term care.
Secondary objective
To assess the impact of AT on: users (autonomy, usefulness and user‐friendliness, adoption of AT); cognitive function and neuropsychiatric symptoms; need for informal and formal care; perceived quality of life; informal carer burden, self‐esteem and feelings of competence; formal carer work satisfaction, workload and feelings of competence; and adverse events.
Search methods
We searched ALOIS, the Specialised Register of the Cochrane Dementia and Cognitive Improvement Group (CDCIG), on 10 November 2016. ALOIS is maintained by the Information Specialists of the CDCIG and contains studies in the areas of dementia prevention, dementia treatment and cognitive enhancement in healthy people. We also searched the following list of databases, adapting the search strategy as necessary: Centre for Reviews and Dissemination (CRD) Databases, up to May 2016; The Collection of Computer Science Bibliographies; DBLP Computer Science Bibliography; HCI Bibliography: Human‐Computer Interaction Resources; and AgeInfo, all to June 2016; PiCarta; Inspec; Springer Link Lecture Notes; Social Care Online; and IEEE Computer Society Digital Library, all to October 2016; J‐STAGE: Japan Science and Technology Information Aggregator, Electronic; and Networked Computer Science Technical Reference Library (NCSTRL), both to November 2016; Computing Research Repository (CoRR) up to December 2016; and OT seeker; and ADEAR, both to February 2017. In addition, we searched Google Scholar and OpenSIGLE for grey literature.
Selection criteria
We intended to review randomised controlled trials (RCTs) and clustered randomised trials with blinded assessment of outcomes that evaluated an electronic assistive device used with the single aim of supporting memory function in people diagnosed with dementia. The control interventions could either be 'care (or treatment) as usual' or non‐technological psychosocial interventions (including interventions that use non‐electronic assistive devices) also specifically aimed at supporting memory. Outcome measures included activities of daily living, level of dependency, clinical and care‐related outcomes (for example admission to long‐term care), perceived quality of life and well‐being, and adverse events resulting from the use of AT; as well as the effects of AT on carers.
Data collection and analysis
Two review authors independently screened all titles and abstracts identified by the search.
Main results
We identified no studies which met the inclusion criteria.
Authors' conclusions
This review highlights the current lack of high‐quality evidence to determine whether AT is effective in supporting people with dementia to manage their memory problems.
Plain language summary
Assistive technology for memory support in dementia
Review question
We wanted to find out from a review of the evidence whether Assistive Technology can help people with dementia overcome some of the effects of their memory problems.
Background
Dementia causes memory problems that make it increasingly difficult to carry out everyday activities, for example cooking, remembering appointments, taking medication. The memory problems experienced can have a large negative impact on people with dementia, and may cause confusion, anxiety, embarrassment, or depression. This decreasing ability to carry out daily activities can cause stress to family carers who worry about the person’s safety and well‐being. A range of electronic devices — most commonly referred to as Assistive Technology (AT – used throughout this review), and sometimes as Electronic Assistive Technology (EAT) — have been developed to support people with dementia. The devices can be divided into four categories offering support with general and personal information (e.g. digital social charts); practical support with problems caused by the symptoms of dementia, especially memory loss (e.g. electronic pill dispenser boxes, electronic diaries); support with social contact and company (e.g. picture phones, interactive ‘pet’ robots); and support with perceived risks to health and safety (e.g. tracking devices, fall sensors). In this review, we concentrated on devices intended to help people manage their memory problems. We wanted to find our whether AT is effective at helping people who have memory problems due to dementia carry out their daily activities, and whether it makes them less dependent on others, improves their quality of life or has an effect on admission to institutional care. We also wanted to find out if it has any impact on family and paid carers.
Study characteristics
We searched systematically for all research studies that had evaluated AT by allocating people with dementia at random to an AT device or to 'usual care' or a non‐technological solution to support memory and then comparing outcomes. Our search included all the evidence available up to 10 November 2016.
Key results
We found no studies that met our criteria.
Quality of the evidence
The review identified a lot of literature on the development of AT, including reports of researchers working with people with dementia and their carers to determine the type and design of AT which might be useful. There was also a lot of guidance written for health professionals and potential users of AT. We found some small studies that had tested the effectiveness of selected AT devices, but the methods used were not of a high enough quality to meet the review criteria. Therefore we cannot be sure at the moment whether or not AT can really help people with dementia manage their memory problems. We believe more research should be done in this area.
Background
Daily life without Information Communication Technology (ICT) is almost unthinkable to many people nowadays. ICT serves many purposes including safety, navigation, or social contact and is applied in many environments, including health and social care settings. The cost of electronic devices have decreased over the years to a current level where even complex devices are affordable for most people. This raises a societal expectation to exploit the potential of these devices to support people with disabilities. New developments in health care to support people in improving their well‐being by means of ICT are encouraged by governments (Kamel Boulous 2009). Also, the European Commission stimulates the development of Assistive Technology to prevent people with disabilities being excluded from society by funding programmes like e‐Inclusion and Ambient Assisted Living (AAL) (European Commission 2010). Consequently, in the last two decades ICT has increasingly been developed to support people with cognitive impairment, including impairment related to dementia, in their daily lives.
ICT‐based devices developed for people with dementia are usually referred to as Assistive Technology (AT), but other terminology is also used, including Electronic Assistive Technology (EAT), telecare, cognitive prosthetics, technology‐based reminding support, and pervasive computing. AT has been developed to support people with dementia and their carers to manage their daily activities and to enhance safety. Several reports describe designing AT for groups with cognitive impairment (Cahill 2007; Hanson 2007; Meiland 2007; Mulvenna 2010; Nugent 2008; Rialle 2008; Sixsmith 2007; Span 2013; Sterns 2005; Van der Roest 2008). Some successful AT devices like electronic pill boxes, picture phones, or mobile tracking devices are already commercially available but, due to the lack of well‐designed trials and small sample sizes, their usefulness and effectiveness for people with dementia are not always clear. Furthermore, a wide range of devices and participants with a diversity of cognitive impairments are involved in the different studies, which makes it difficult to draw firm conclusions on the usefulness and effectiveness of AT for this group (Fleming 2014; Lauriks 2007; Topo 2009).
In their review, Lauriks 2007 described AT that is intended to support people with dementia in the four needs areas of: general and personalised information; practical support with regard to symptoms of dementia (including memory problems); social contact and company; and health monitoring and perceived safety. This review builds further on the work of Lauriks 2007 and focuses on AT for memory problems, one of the most common symptoms in people with dementia, which has a high impact on functioning in daily life. Many people with dementia, as well as their informal carers, report a lack of adequate support for memory problems. In fact, relative to other unmet needs, support for memory problems is the most frequently mentioned unmet need by people with dementia and carers (Van der Roest 2009). In addition to the more traditional means of memory support, for example diaries, written signs, journals or notes, ICT applications could potentially offer effective alternatives. It is expected that electronic memory support devices will enable people with dementia to live more independently and will alleviate carer burden (Cahill 2007).
Description of the condition
A dementia syndrome is usually caused by a chronic or progressive disease of the brain. The most common forms of dementia are Alzheimer's disease and vascular dementia. Dementia is characterized by impairment in higher cortical functioning, including memory, thinking, orientation, language, comprehension, action, and judgement. Cognitive impairment in dementia is often preceded or accompanied by the deterioration of emotional control, social behaviour, or motivation (WHO 2007). Functional decline is one of the core diagnostic criteria in all types of dementia (American Psychiatric Association 1994). Performing tasks of daily living becomes increasingly difficult. Initially the more complex instrumental activities of daily living (IADL) are affected and then later on also personal ADL tasks (Liu 2007; Öhman 2001; Sikkes 2009; Van Wielingen 2004). In the advanced stage of dementia people become totally dependent on the help of others (Agüero‐Torres 1998; Wimo 1999).
It is estimated that currently 44.4 million people worldwide live with dementia, and this total is expected to rise by 71% to reach 75.6 million by 2030 and 135.5 million by 2050 (Alzheimer's Disease International 2013). Many people in the advanced stage of dementia are admitted to a long‐term care facility to receive full‐time care. It is estimated that in high‐income countries approximately 34% of the people with dementia live in long‐term care facilities (Alzheimer's Disease International 2010).
People with dementia usually experience memory problems. The type of memory impairment experienced is dictated by the underlying condition, the associated site, and the extent of the brain lesion. Prospective memory (PM) is essential for living independently as it involves remembering to do things in the future without any prompting; whilst retrospective memory (RM) involves recalling or recognising information that one has acquired in the past (Maylor 2002).
Due to the progressive nature of dementia, people with dementia will increasingly depend on the support of others. This help is frequently provided by relatives, the so‐called informal carers, who provide unpaid care. If informal care is no longer sufficient, feasible, or available, paid staff (formal carers) step in to provide support. The estimated global costs for dementia are currently estimated to be USD 604 billion, of which the majority is attributed to informal care (42%) and social care (care provided by community care professionals and in long‐term care settings) (42%). The direct medical costs are much lower (16%) (Alzheimer's Disease International 2010). With an estimated increase in costs of 85% by the year 2030, Alzheimer's Disease International stresses the urgent need to develop cost‐effective packages of medical and social care for people with dementia.
Description of the intervention
Research has shown that people with mild dementia would appreciate devices to support both their prospective and retrospective memory. Examples of prospective memory support would be reminding them of actions needed at particular times, such as taking medication, eating, or keeping appointments. Retrospective memory support could include helping them to locate lost items, remember names of people, or keep track of the day and time (Nugent 2007).
Devices developed for different purposes have specific functional capabilities. Some ATs can be customised to react to the environment or the user in a dynamic way, for example sensors that activate a warning, alarm, or camera if no movement is detected in a defined period of time. Whilst some devices are stand‐alone, like electronic calendars, others are integrated into a more comprehensive, remotely configurable system, like the COGKNOW Day Navigator. As well as memory support, this also offers communication, activity and safety support (Meiland 2007). Other devices can support in ADL tasks, for instance the COACH that facilitates in handwashing by prompting (Mihailidis 2008). Devices can also be mobile, enabling the user to take the device with them outside their home; for example the 'Take Me Home' device, developed in the same COGKNOW project, guides people to their home by means of spoken instruction and images when they are lost. Since impairments in prospective memory are generally more often reported than retrospective memory impairments in people with dementia and seem to have greater impact (Smith 2000), AT devices that aim to support prospective memory functioning will usually need to be more advanced, because the first have to anticipate future events and changing environments. Effective prospective memory devices will have a larger impact on the daily lives of people with dementia than AT devices for retrospective memory problems, since prospective AT will enable people to function independently for a longer period of time.
How the intervention might work
People with memory problems often rely on others around them or on static reminders or cues, like written notes or diaries, to support their memory. By providing an AT device that reminds them of meaningful events, previous daytime activities, or guides them through complex situations or tasks, people with dementia may act more independently. They will attain their daily goals (for example appointments and activities), may be less agitated or confused, and will experience a better quality of life; and their informal carers may experience less burden (Cahill 2007). Ideally the assistive technology should be adapted and fine‐tuned to dementia‐related and other personal and context‐related factors (Dröes 2010). Levels of technology used for AT devices vary from low technology to higher level personalised technology and context‐aware (smart) environments. Electronic calendars are examples of low‐technology devices, as they do not take into account whether or not the person with dementia follows the given reminder. Mobile tracking devices that automatically provide a warning when a person with dementia is leaving his or her familiar area are examples of more context‐aware devices. Although technology for context‐aware environments does exist, it is not yet widely provided due to its current sensitivity to errors and false alarms.
Why it is important to do this review
Many assistive devices have been developed for memory support in people with cognitive impairment. However, in order for AT to be effective a device should be user friendly, so that a carer can manage or configure the device him‐ or herself; and the person with dementia should be able to use the device for his or her goals, regardless of their level of cognitive decline. To optimise the user friendliness people with dementia and informal carers were increasingly involved in the AT development process in the past decade. Methods for such design processes have been described (Meiland 2012; Meiland 2017; Span 2013). Despite the effort to carefully design AT and the conviction that AT can be beneficial for people with dementia in alleviating memory problems, robust evidence on its efficacy and effectiveness in everyday life and the user‐friendliness of these devices, especially for people with dementia, is scarce. Although reviews of (electronic) memory aids do exist (Bharucha 2009; Caprani 2006; Fleming 2014; Fritschy 2004; Lauriks 2007; Lindenberger 2008; Topo 2009), a systematic review of high‐quality studies focusing on the efficacy of AT for memory support in people with dementia is lacking. This review is intended to investigate the extent and quality of the evidence for existing devices intended to support people with dementia manage different types of memory problems. A comprehensive overview of evidence‐based devices will guide people with dementia and their informal and professional carers in selecting an appropriate memory device to match the user's needs. The review also provides useful information for AT developers in this rapidly growing area, by addressing the gaps in, and shortcomings of, existing AT for memory support. It highlights questions which need further evaluation using robust research methodologies.
Objectives
Primary objective
To assess the efficacy of AT for memory support in people with dementia in terms of daily performance of personal and instrumental activities of daily living (ADL), level of dependency, and admission to long‐term care.
Secondary objective
To assess the impact of AT on: users (autonomy, usefulness and user‐friendliness, adoption of AT); cognitive function and neuropsychiatric symptoms; need for informal and formal care; perceived quality of life; informal carer burden, self‐esteem and feelings of competence; formal carer work satisfaction, workload and feelings of competence; and adverse events.
Methods
Criteria for considering studies for this review
Types of studies
Randomised controlled trials (RCTs) and cluster randomised trials with blinded assessment of outcome were included (including those with inadequate sequence allocation). Studies reported in any language were eligible for inclusion.
Types of participants
Participants needed to be diagnosed with dementia according to the Diagnostic and Statistical Manual IV (American Psychiatric Association 1994) or ICD‐10 (WHO 2007). If participants' diagnostic information was not described in potential studies, primary authors were asked for additional information. If the information provided met the set criteria, studies were included. No further inclusion criteria for participants were applied.
Types of interventions
The review included studies that evaluated AT for people with dementia: devices driven by electronics and used with the single aim of supporting memory problems. The AT under evaluation could be stand alone or integrated in a service system (configured remotely); and stationary or mobile. The devices under study are most likely to require configuration or set‐up help by carers. The focus of the studies was to be primarily on the person with dementia, but the impact on carers was also reviewed. Studies that evaluated a combination of devices that were provided to meet different aims were not included. The control interventions could either be 'care (or treatment) as usual' or non‐technological psychosocial interventions (including interventions that use non‐electronic assistive devices) also specifically aimed at supporting memory problems.
Types of outcome measures
The primary outcome measures regarding the efficacy of the AT under study related to ADL and the level of dependency of people with dementia. The secondary outcome measures related to clinical and care‐related outcomes of the AT for people with dementia, to their perceived quality of life and well‐being, and also the effects of AT on their carers — informal carers for community‐based interventions and formal carers for institutional‐based interventions. Adverse events resulting from use of AT for people with dementia and carers (informal and formal) were to be included. All reported time frames were to be included; all outcomes measures were to be listed, and their validity and reliability considered.
Primary outcomes
Daily functioning
Activities of daily living (ADL): personal (PADL) and instrumental (IADL).
Level of dependency (self‐report or proxy report).
Admission to long‐term care (for community‐based interventions).
Secondary outcomes
User reports
Experienced autonomy (self‐report).
Experienced usefulness and user‐friendliness of AT (self‐report).
Adoption of AT.
Clinical
Cognitive functioning.
Neuropsychiatric symptoms (behavioural and mood problems).
Care
Need for informal care.
Need for formal care.
Well‐being
Perceived quality of life or well‐being (self‐report or proxy report).
Informal carer
Carer burden.
Self‐esteem.
Feelings of competence.
Formal carer
Work satisfaction.
Workload.
Feelings of competence.
Adverse events
Clinical.
Care.
Informal carer.
Formal carer.
Search methods for identification of studies
Electronic searches
The Cochrane Dementia and Cognitive Improvement Group Information Specialist searched ALOIS (www.medicine.ox.ac.uk/alois), the Cochrane Dementia and Cognitive Improvement Group’s Specialised Register, on 10 November 2016, using a search strategy devised by HvdR. The search terms used were: assistive OR technology OR device OR devices OR electronic OR locator OR "pill dispenser" OR calendar OR clock OR telecare.
ALOIS is maintained by the Cochrane Dementia and Cognitive Improvement Group Information Specialist. It contains studies in the areas of dementia prevention, dementia treatment and cognitive enhancement in healthy people. The studies are identified from:
monthly searches of a number of major healthcare databases: MEDLINE, Embase, CINAHL, PsycINFO and LILACS;
monthly searches of trial registers: ISRCTN; UMIN (Japan's Trial Register); the World Health Organization (WHO) portal (which covers ClinicalTrials.gov; ISRCTN; the Chinese Clinical Trials Register; the German Clinical Trials Register; the Iranian Registry of Clinical Trials and the Netherlands National Trials Register, plus others);
quarterly search of the Cochrane Library’s Central Register of Controlled Trials (CENTRAL);
six‐monthly searches of a number of grey literature sources: ISI Web of Knowledge Conference Proceedings; Index to Theses; Australasian Digital Theses.
To view a list of all sources searched for ALOIS see About ALOIS on the ALOIS web site.
Details of the search strategies used for the retrieval of trial reports from the healthcare databases, CENTRAL and conference proceedings can be viewed in the ‘Methods used in reviews’ section within the editorial information about the Dementia and Cognitive Improvement Group.
Additional searches were performed in many of the sources listed above to cover the period between the last search performed in ALOIS and completion of the review to ensure that the search was as up to date and as comprehensive as possible. The search strategies used can be seen in Appendix 1.
Two authors (HvdR and JW) carried out further searches in the following specialist sources, adapting the search strategy as needed.
PiCarta (to October 2016).
OTseeker (to February 2017).
ADEAR (to February 2017).
AgeInfo (to June 2016).
Social Care Online (to October 2016).
Centre for Reviews and Dissemination (CRD) Databases (to May 2016).
The Collection of Computer Science Bibliographies (to June 2016).
DBLP Computer Science Bibliography (to June 2016).
Networked Computer Science Technical Reference Library (NCSTRL) (to November 2016).
Computing Research Repository (CoRR) (to December 2016).
IEEE Computer Society Digital Library (to October 2016).
Springer Link Lecture Notes (to October 2016).
HCI Bibliography: Human‐Computer Interaction Resources (to June 2016).
Inspec (to October 2016).
J‐STAGE: Japan Science and Technology Information Aggregator, Electronic (to November 2016).
The search was adapted as needed for each database. There was no language restriction. See Appendix 2 for the search strategies used for these databases.
Searching other resources
Additionally, Google Scholar and OpenSIGLE were searched for grey literature. References of identified articles were handsearched using the snowball method in order to find other potentially relevant studies.
Data collection and analysis
HvdR and JW divided the databases between them and executed the search strategies as described.
Selection of studies
The search results were merged using reference management software and duplicate records were removed. HvdR and JW independently screened study titles and abstracts for appropriateness. We removed obviously irrelevant reports and linked multiple reports of the same study.
We obtained full‐text versions of potentially relevant reports. HvdR and JW examined these independently to assess compliance with the predefined eligibility criteria. If suitability of a study was unclear after examining the full text, we contacted the corresponding author to request clarification or additional information, or both. HvdR and JW compared and discussed their results. In the case of disagreement we examined abstracts and full papers in more detail until agreement was reached. Studies excluded for not meeting the predefined eligibility criteria are listed in the 'Characteristics of excluded studies' table together with the reasons for exclusion.
Results
Description of studies
See: Characteristics of excluded studies.
The initial search identified a total of 1246 citations after de‐duplication and first assessment by the CDCIG information specialist. After screening the titles and abstracts, nine full‐text papers were retrieved and one clinical trial was considered for further assessment. Two papers were looked at in more detail because they were reviews on assistive technology for cognition (Gillespie 2012) and late‐life mental disorders (Westphal 2010). Both reviews discriminated between studies of AT for dementia and AT for other disorders, however no studies were described that met the inclusion criteria for this review.
HvdR and JW independently evaluated the remaining papers and clinical trials for study design and methodological quality (Hofmann 2003; ISRCTN 86537017; Labelle 2006; Lancioni 2010; Mihailidis 2004; Mihailidis 2008; Ownby 2012; Spring 2011). It was agreed that none of the studies met the review eligibility criteria. See Figure 1 for an overview.
1.
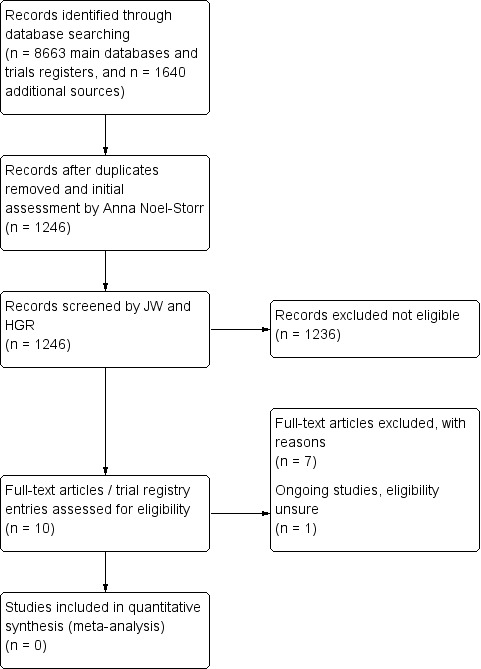
Flow diagram
Excluded studies
The following studies were evaluated in detail but excluded as discussed above.
Hofmann 2003 studied an interactive computer‐based training programme that relates to activities of daily living. This intervention did not meet the inclusion criteria and the study had a non‐randomised longitudinal group design. Lancioni 2010 described two studies on the use of verbal instructions and support technology to enable people with dementia to perform daily activities — making coffee or setting the table (Study I) and preparing a fruit or vegetable salad (Study II). Neither study had a randomised design. The study conducted by Labelle 2006 to evaluate an automated prompting system to support handwashing was not eligible for inclusion due to the multiple‐treatment, single‐subject design. Mihailidis 2004 and Mihailidis 2008 described two studies to assess the efficacy of COACH (Cognitive orthosis for assisting activities in the home), a computerised prompting system that supports people with dementia in the process of handwashing, but both were ineligible because they used a single subject design. Ownby 2012 used a randomised design to evaluate improvement of medication adherence using an electronic pill bottle, but the study was excluded as, although participants were clinically judged to have memory impairment, there was no validated dementia diagnosis. Spring 2011 described a computerised decision support system intervention used in primary care practice for treating mental illness; this study was excluded as neither the intervention nor the participants met the review eligibility criteria.
Ongoing studies
The ATTILA project (Assistive Technology and Telecare to maintain Independent Living At home for people with dementia), a registered clinical trial started in 2013, aims to support people with dementia to remain more independent for longer in their own home with the help of AT and telecare (ATT) (ISRCTN 86537017). The type of ATT used in the intervention will vary per participant, depending on individual needs. The study uses a multicentre randomised controlled trial design. The range of AT utilised within the study may not all be provided with the exclusive aim of supporting memory problems, so critical evaluation of the study design and results will be required to decide whether to include it or not. One of the two primary outcome measures — time from randomisation to admission to long‐term care — meets the review inclusion criteria, as do the secondary outcome measures: caregiver burden, quality of life, number and severity of adverse events, information on acceptability, applicability and reliability of the ATT offered. The trial is ongoing and no results are published yet.
Risk of bias in included studies
Not applicable since no studies met the inclusion criteria.
Effects of interventions
No studies met the inclusion criteria for this review.
Discussion
Summary of main results
We did not identify any completed studies that met the inclusion criteria for this review. One ongoing study, ATTILA, may meet the inclusion criteria, although the intervention will be tailored to the needs of the individual participant, and so will vary from person to person (ISRCTN 86537017). Prospective study results will need to be examined critically to decide on inclusion, as AT is not always provided with the sole aim of supporting memory problems, and will therefore not meet the review inclusion criteria. Studies that were evaluated in detail were excluded primarily because of study design, with longitudinal, non‐randomised and single‐subject designs being used. All of them had small sample sizes, for reasons not discussed in the publications. In general the AT could not be applied in large‐scale study designs because personalisation of the devices was required to meet the needs of their users: test settings, in which were installed sophisticated technical equipment to execute, monitor, and assess the interventions, were set up in the living environments of participants (Labelle 2006; Lancioni 2010); training was provided in some instances (Hofmann 2003); or many task rehearsals were needed (Lancioni 2010), as was intensive data collection in some studies (Lancioni 2010; Mihailidis 2004; Mihailidis 2008). The studies also used advanced technology to assess the performance of the people with dementia using the AT, making the studies expensive, and reducing the opportunity to include large study samples. It should be noted that all of the studies reported (moderately) positive results on functional domains.
AT to support people with dementia was first introduced into practice in approximately the year 2000, so this is a relatively new area. We therefore did not expect to find many randomised controlled trials at this stage, but the total lack of them is surprising. For every study including vulnerable groups, including people with cognitive impairment of whom some may be not mentally competent, gaining ethical governance is subject to strict regulations. Meeting these strict regulations can cause challenges in conducting studies in people with cognitive impairment and might be one of the reasons why no RCTs meeting the inclusion criteria were identified in this review. We also found that there is a lack of standardised terminology for AT. We had to use a large number of search terms, and there were a lot of false positives among the search results. Two prominent terms that were used in the literature were "technology" and "device", but in addition we also found terms such as "computerised" and "automated" that were used in combination with terminology addressed in the search strategy. Standardisation of terminology is needed to help identify studies in this developing area.
Quality of the evidence
No evidence was reviewed.
Agreements and disagreements with other studies or reviews
The lack of robust evidence for the effectiveness of AT for memory support for people with dementia is reflected across the spectrum of AT research, including other elderly populations. Various reviews of AT to support people with dementia have been conducted, and all report moderate or promising effects of small‐scale studies. Martin 2008 did not identify any eligible studies of smart home technologies for health and social care support of people with a physical disability, dementia or learning disability. Lauriks 2007 identified numerous small‐scale uncontrolled studies that evaluated the use of assistive devices providing practical support with the symptoms of dementia, (personalised) information, monitoring and perceived safety, and social contact and company. The same applies to Bemelmans 2012, Buettner 2010, Carswell 2009, Ciro 2014, Fleming 2014, Huschilt 2012, Mohktari 2012, Rigaud 2011, and Span 2013.
Peterson 2012 highlight the absence of an international consensus on a classification system for AT; and call for the several professional groups involved in developing and evaluating AT in dementia to use a common language to define and describe technology. The authors suggest that the societal impact of AT will emerge when the definitions, goals and outcomes are clearly set.
There is some published research on the involvement of people with dementia in developing AT to meet their own needs (e.g. Davies 2009; Dröes 2010; Mulvenna 2010; Sixsmith 2007; Span 2013; Topo 2009). Since needs are a consequence of the symptoms and impairments of dementia, they will vary per person, and so it is important that AT devices can be tailored to the individual user (Peterson 2012). AT devices are of no use if they exceed the skills level of their user (Dröes 2010; Mayer 2013; Meiland 2017).
Authors' conclusions
Implications for practice.
Published guidance on using assistive technology for people with dementia is available widely and can be found mainly in the grey literature (e.g. Improvement and Efficiency West Midlands 2013, Alzheimer's Society 2014, Alzheimer's Society 2015, Trent Dementia Services Development Centre 2016, Vilans 2016); and a much studied topic in research on AT for dementia is the development and design of AT (Meiland 2017). However, this review found that there is no high‐quality evidence to determine whether AT is an effective means of supporting people with dementia manage their memory problems.
Implications for research.
AT development is a very active area of research. There are a lot of qualitative studies aimed at identifying the type of activities that people with dementia want support for, and investigating device design and usage, but there is less work evaluating effectiveness.
The problem described by Martin 2008 and Peterson 2012 — a lack of consistent terminology to describe assistive technology — should be addressed. The lack of consistency makes identification of appropriate literature difficult and in future could hamper meta‐analysis. Other reviews have also highlighted the difficulty in comparing studies due to the variety of aims, technologies, design and outcome measures used (Topo 2009).
The evaluation of assistive technology is certainly complex. We support the suggestion made by Martin 2008: that future research could usefully be based on the discussion document published by the UK Medical Research Council (MRC) 'Developing and evaluating complex interventions' (Medical Research Council 2008). In addition to the methodological issues, it is also important to reduce the practical complexity of AT interventions. Technology is still evolving and becoming less intrusive, which makes it easier to apply and evaluate AT in the daily lives and natural environments of people with dementia.
Because of the need to personalise AT, RCTs in this field should be 'needs based', rather than 'intervention based'. Similar challenges have previously been met in RCTs of complex psychosocial interventions for people with dementia and their carers, which also have to address how to define the intervention precisely enough for it to be reproducible whilst building in the flexibility needed for it to be person‐centred and individualised. The goals and aims of trials must be clearly set. RCTs with a stratified block design (with skills level or specific impairments as strata) may be useful to investigate the overall effectiveness of the AT devices and the effectiveness in subgroups.
Support for memory problems is the unmet need most frequently mentioned by people with dementia and their carers. Small‐scale studies of memory support devices appear promising. There is now a need for more robust evaluation of effectiveness, addressing the complex methodological issues described.
Appendices
Appendix 1. Searches: main bibliographic databases
|
Source |
Search strategy | Hits retrieved |
| 1. ALOIS (www.medicine.ox.ac.uk/alois) [last searched: 10 November 2016] |
assistive OR technology OR device OR devices OR electronic OR locator OR "pill dispenser" OR calendar OR clock OR telecare | Feb 2013: 86 Feb 2014: 5 Feb 2015: 1 Feb 2016: 0 Nov 2016: 0 |
| 2. MEDLINE In‐process and other non‐indexed citations and MEDLINE 1950‐present (Ovid SP) [last searched: 10 November 2016] |
1. exp Dementia/ 2. Delirium/ 3. Wernicke Encephalopathy/ 4. Delirium, Dementia, Amnestic, Cognitive Disorders/ 5. dement*.mp. 6. alzheimer*.mp. 7. (lewy* adj2 bod*).mp. 8. deliri*.mp. 9. (chronic adj2 cerebrovascular).mp. 10. ("organic brain disease" or "organic brain syndrome").mp. 11. ("normal pressure hydrocephalus" and "shunt*").mp. 12. "benign senescent forgetfulness".mp. 13. (cerebr* adj2 deteriorat*).mp. 14. (cerebral* adj2 insufficient*).mp. 15. (pick* adj2 disease).mp. 16. (creutzfeldt or jcd or cjd).mp. 17. huntington*.mp. 18. binswanger*.mp. 19. korsako*.mp. 20. or/1‐19 21. technology.ti,ab. 22. ("information communications technology" or ICT).ti,ab. 23. Technology/ 24. exp Self‐Help Devices/ 25. orthot*.ti,ab. 26. device*.ti,ab. 27. telecare.ti,ab. 28. electronic.ti,ab. 29. "pill dispenser*".mp. 30. (organi?er or locator).ti,ab. 31. (calendar* or clock*).ti,ab. 32. or/21‐31 33. randomized controlled trial.pt. 34. controlled clinical trial.pt. 35. randomized.ab. 36. placebo.ab. 37. randomly.ab. 38. trial.ab. 39. groups.ab. 40. (animals not (humans and animals)).sh. 41. or/33‐39 42. 41 not 40 43. 20 and 32 and 42 |
Feb 2013: 500 Feb 2014: 81 Feb 2015: 79 Feb 2016: 79 Nov 2016: 117 |
| 3. Embase 1980 to 2016 November 09 (Ovid SP) [last searched: 10 November 2016] |
1. exp dementia/ 2. Lewy body/ 3. delirium/ 4. Wernicke encephalopathy/ 5. cognitive defect/ 6. dement*.mp. 7. alzheimer*.mp. 8. (lewy* adj2 bod*).mp. 9. deliri*.mp. 10. (chronic adj2 cerebrovascular).mp. 11. ("organic brain disease" or "organic brain syndrome").mp. 12. "supranuclear palsy".mp. 13. ("normal pressure hydrocephalus" and "shunt*").mp. 14. "benign senescent forgetfulness".mp. 15. (cerebr* adj2 deteriorat*).mp. 16. (cerebral* adj2 insufficient*).mp. 17. (pick* adj2 disease).mp. 18. (creutzfeldt or jcd or cjd).mp. 19. huntington*.mp. 20. binswanger*.mp. 21. korsako*.mp. 22. CADASIL.mp. 23. or/1‐22 24. technology.ti,ab. 25. ("information communications technology" or ICT).ti,ab. 26. assistive technology/ or assistive technology device/ or technology/ 27. Self‐Help Devices.mp. 28. orthot*.ti,ab. 29. device*.ti,ab. 30. telecare.ti,ab. 31. electronic.ti,ab. 32. "pill dispenser*".mp. 33. (organi?er or locator).ti,ab. 34. (calendar* or clock*).ti,ab. 35. or/24‐34 36. randomized controlled trial/ 37. controlled clinical trial/ 38. randomized.ab. 39. placebo.ab. 40. randomly.ab. 41. trial.ab. 42. groups.ab. 43. or/36‐42 44. 23 and 35 and 43 |
Feb 2013:1289 Feb 2014: 284 Feb 2015: 359 Feb 2016: 354 Nov 2016: 320 |
| 4. PsycINFO 1806 to November week 2 2016 (Ovid SP) [last searched: 10 November 2016] |
1. exp Dementia/ 2. exp Delirium/ 3. exp Huntingtons Disease/ 4. exp Kluver Bucy Syndrome/ 5. exp Wernickes Syndrome/ 6. exp Cognitive Impairment/ 7. dement*.mp. 8. alzheimer*.mp. 9. (lewy* adj2 bod*).mp. 10. deliri*.mp. 11. (chronic adj2 cerebrovascular).mp. 12. ("organic brain disease" or "organic brain syndrome").mp. 13. "supranuclear palsy".mp. 14. ("normal pressure hydrocephalus" and "shunt*").mp. 15. "benign senescent forgetfulness".mp. 16. (cerebr* adj2 deteriorat*).mp. 17. (cerebral* adj2 insufficient*).mp. 18. (pick* adj2 disease).mp. 19. (creutzfeldt or jcd or cjd).mp. 20. huntington*.mp. 21. binswanger*.mp. 22. korsako*.mp. 23. ("parkinson* disease dementia" or PDD or "parkinson* dementia").mp. 24. or/1‐23 25. technology.ti,ab. 26. ("information communications technology" or ICT).ti,ab. 27. Technology/ 28. Assistive Technology/ 29. orthot*.ti,ab. 30. device*.ti,ab. 31. telecare.ti,ab. 32. Telemedicine/ 33. electronic.ti,ab. 34. "pill dispenser*".mp. 35. (organi?er or locator).ti,ab. 36. (calendar* or clock*).ti,ab. 37. or/25‐36 38. 24 and 37 39. exp Clinical Trials/ 40. randomly.ab. 41. randomi?ed.ti,ab. 42. trial.ab. 43. groups.ab. 44. "double‐blind*".ti,ab. 45. "single‐blind*".ti,ab. 46. RCT.ti,ab. 47. or/39‐46 48. RCT.ti,ab. 49. or/39‐46 50. 38 and 49 |
Feb 2013: 356 Feb 2014: 49 Feb 2015: 85 Feb 2016: 69 Nov 2016: 44 |
| 5. CINAHL (EBSCOhost) [last searched: 10 November 2016] |
S1 (MH "Dementia+") S2 (MH "Delirium") or (MH "Delirium, Dementia, Amnestic, Cognitive Disorders") S3 (MH "Wernicke's Encephalopathy") S4 TX dement* S5 TX alzheimer* S6 TX lewy* N2 bod* S7 TX deliri* S8 TX chronic N2 cerebrovascular S9 TX "organic brain disease" or "organic brain syndrome" S10 TX "normal pressure hydrocephalus" and "shunt*" S11 TX "benign senescent forgetfulness" S12 TX cerebr* N2 deteriorat* S13 TX cerebral* N2 insufficient* S14 TX pick* N2 disease S15 TX creutzfeldt or jcd or cjd S16 TX huntington* S17 TX binswanger* S18 TX korsako* S19 S1 or S2 or S3 or S4 or S5 or S6 or S7 or S8 or S9 or S10 or S11 or S12 or S13 or S14 or S15 or S16 or S17 or S18 S20 AB technology S21 TX "information communications technology" OR ICT S22 (MM "Assistive Technology") S23 TX orthot* S24 TX device* S25 TX telecare OR telemedicine S26 TX electronic S27 TX "pill dispenser*" S28 TX organizer OR organiser OR locator S29 TX calendar* OR clock* S30 S20 or S21 or S22 or S23 or S24 or S25 or S26 or S27 or S28 or S29 S31 S19 and S30 S32 AB randomly S33 (MH "Clinical Trials") S34 TX RCT OR CCT S35 TX groups OR "control group" S36 TX "double‐blind*" OR "single‐blind*" S37 TX randomized OR randomized S38 S32 or S33 or S34 or S35 or S36 or S37 S39 S31 and S38 |
Feb 2013: 514 Feb 2014: 110 Feb 2015: 67 Feb 2016: 123 Nov 2016: 22 |
| 6. Web of Science and conference proceedings [last searched: 10 November 2016] |
Topic=(dementia OR alzheimer* OR lewy OR memory OR CJD OR JCD OR creutzfeldt OR binswanger OR korsakoff) AND Topic=(technology OR ICT OR device* OR telecare OR electronic OR "pill dispenser*" OR organizer OR organiser OR locator OR calendar* OR clock*) AND Topic=(randomly OR placebo OR trial OR RCT OR randomized OR randomised OR "double‐blind*" OR "single‐blind*" OR CCT OR "cross‐over" OR crossover) Timespan=All Years. Search language=English Lemmatization=On |
Feb 2013: 1506 Feb 2014: 388 Feb 2015: 468 Feb 2016: 481 Nov 2016: 452 |
| 7. LILACS (BIREME) [last searched: 10 November 2016] |
technology OR technologia OR tecnologia OR calendar OR calendario OR dispenser OR dispensador OR distribuidor OR electronic OR eletrônico OR electrónico OR device OR devices OR dispositivo [Words] and dementia OR alzheimer OR alzheimers OR lewy body OR "vascular impairment" OR "cerebral insufficiency" [Words] and randomly OR randomised OR randomized OR trial OR ensaio clínico OR control OR controlled [Words] | Feb 2013: 10 Feb 2014: 19 Feb 2015: 0 Feb 2016: 0 Nov 2016: 1 |
| 8. CENTRAL (The Cochrane Library) [last searched: 10 November 2016] |
#1 MeSH descriptor: [Dementia] explode all trees #2 MeSH descriptor: [Delirium] this term only #3 MeSH descriptor: [Wernicke Encephalopathy] this term only #4 MeSH descriptor: [Delirium, Dementia, Amnestic, Cognitive Disorders] this term only #5 dement* #6 alzheimer* #7 "lewy* bod*" #8 deliri* #9 "chronic cerebrovascular" #10 "organic brain disease" or "organic brain syndrome" #11 "normal pressure hydrocephalus" and "shunt*" #12 "benign senescent forgetfulness" #13 "cerebr* deteriorat*" #14 "cerebral* insufficient*" #15 "pick* disease" #16 creutzfeldt or jcd or cjd #17 huntington* #18 binswanger* #19 korsako* #20 #1 or #2 or #3 or #4 or #5 or #6 or #7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 #21 MeSH descriptor: [Self‐Help Devices] explode all trees #22 technology #23 telecare #24 device* #25 electronic #26 "pill dispenser*" #27 locator* #28 calender* #29 clock* #30 #21 or #22 or #23 or #24 or #25 or #26 or #27 or #28 or #29 in Trials #31 #20 and #30 in Trials |
Feb 2013: 161 Feb 2014: 51 Feb 2015: 35 Feb 2016: 31 Nov 2016: 25 |
| 9. ClinicalTrials.gov (www.clinicaltrials.gov) [last searched: 10 November 2016] |
Interventional Studies | dementia OR alzheimer OR alzheimers OR VCI OR vascular dementia OR VaD OR vascular cognitive impairment OR cadasil OR multi‐infarct OR binswanger | device OR devices OR technology OR telecare OR electronic OR calendar OR dispenser OR locator | Feb 2013: 129 Feb 2014: 2 Feb 2015: 0 Feb 2016: 7 Nov 2016: 3 |
| 10. ICTRP Search Portal (http://apps.who.int/trialsearch) [includes: Australian New Zealand Clinical Trials Registry; ClinicalTrilas.gov; ISRCTN; Chinese Clinical Trial Registry; Clinical Trials Registry – India; Clinical Research Information Service – Republic of Korea; German Clinical Trials Register; Iranian Registry of Clinical Trials; Japan Primary Registries Network; Pan African Clinical Trial Registry; Sri Lanka Clinical Trials Registry; The Netherlands National Trial Register] [last searched: 10 November 2016] |
dementia OR alzheimer OR alzheimers OR VCI OR vascular dementia OR VaD OR vascular cognitive impairment OR cadasil OR multi‐infarct OR binswanger | device OR devices OR technology OR telecare OR electronic OR calendar OR dispenser OR locator AND recruiting | Feb 2013: 97 Feb 2014: 3 Feb 2015: 0 Feb 2016: 2 Nov 2016: 1 |
| TOTAL before de‐duplication | Feb 2013: 4448 Feb 2014: 992 Feb 2015: 1094 Feb 2016: 1144 Nov 2016: 985 |
|
| TOTAL after de‐dupe and first assess | Feb 2013: 194 from bid databases//129 Trial register records Feb 2014: 53 Feb 2015: 24 Feb 2016: 33 Nov 2016: 29 |
|
Appendix 2. Searches: additional sources
| Search engine | Search strategy | Hits retrieved |
| 1. Picarta 10 October 2013 19 October 2016 |
dementia and technology | Search 1: 102 Search 2: 29 |
| 2. Centre for Reviews and Dissemeniation (CRD) Databases 24 October 2013 12 May 2016 |
(dement* OR alzheimer*) AND ((technol* OR device OR electr*) NOT (drug therapy)) AND (control* OR RCT) | Search 1: 104 Search 2: 24 |
| 3. Collection of Computer Science Bibliographies 22 October 2013 12 June 2016 |
dementia AND technol* AND (rct randomized) AND support NOT (drug* radiogr*) | Search 1: 44 Search 2: 21 |
| 4. DBLP Computer Science Bibliography 22 October 2013 27 June 2016 |
dementia technolog | Search 1: 27 Search 2: 15 |
| 5. Computing Research Repository (CoRR) 22 October 2013 12 December 2016 |
dementia OR Alzheimer | Search 1: 23 Search 2: 62 |
| 6. HCI Bibliography: Human Computer Interaction Resources 25 October 2013 27 June 2016 |
(dementia OR cognitive impairment OR alzheimer MCI OR vascular) AND (support OR assist*) AND (RCT OR control*) | Search 1: 16 Search 2: 6 |
| 7. J‐STAGE: Japan Science and Technology Information Aggregator, Electronic 25 October 2013 13 November 2016 |
Keyword(s): dementia OR Full Text: dementia OR MCI OR cognitive impairment OR alzheimer AND Full Text: technol* OR device |
Search 1: 62 Search 2: 42 |
| 8. Networked Computer Science Bibliographies (NCSTRL; hosted by EECS Technical Reports) 11 November 2013 13 November 2016 |
dement* | Search 1: 1 Search 2: 1 |
| 9. OT Seeker 11 November 2013 7 February 2017 |
Key words: dementia or Alzheimer Intervention: assistive technology / adaptive equipment Diagnosis / sub discipline: Alzheimer’s disease / dementia Method: randomised controlled trial |
Search 1: 6 Search 2: 1 |
| 10. ADEAR 11 November 2013 7 February 2017 |
assistive technology | Search 1: 19 Search 2: 20 |
| 11. AgeInfo 19 November 2013 27 June 2016 |
dementia technology | Search 1: 15 Search 2: 12 |
| 12. Inspec 22 November 2013 20 October 2016 |
(Dement* OR Alzheimer*) AND (technol*) AND RCT | Search 1: 3 Search 2: 0 |
| 13. Springer Link Lecture Notes for Computer Science 22 November 2013 30 October 2016 |
(dement* OR Alzheimer*) AND (technol*) and ("functional support" OR "memory support" OR "daily support") | Search 1: 81 Search 2: 39 |
| 14. Social Care online 27 November 2013 19 October 2016 |
dementia assistive technology RCT | Search 1: 75 Search 2: 40 |
| 15. IEEE Computer Society Digital Library 9 December 2013 20 October 2016 |
dementia and randomised | Search 1: 74 Search 2: 9 |
| 16. Google Scholar 3 March 2014 7 February 2017 |
dementia assistive technology rct OR randomized NOT review | Search 1: 178 Search 2: 102 |
| 17. OpenSIGLE (www.opengrey.eu) 6 March 2014 7 February 2017 |
dementia technology | Search 1: 11 Search 2: 2 |
| TOTAL before de‐duplication | Search 1: 841 Search 2: 425 |
|
| TOTAL after de‐duplication | Search 1: 799 Search 2: 404 |
|
Characteristics of studies
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Hofmann 2003 | Design and methodology. A non‐randomised longitudinal group design with three groups was used to test an Interactive Computer‐Based Training programme as a therapeutic tool. Although the training tasks related to ADL, the tool did not deliver actual support during ADL. |
| Labelle 2006 | Design. Eight participants were included in the study that used a multiple‐treatment, single‐subject research design with four phases. |
| Lancioni 2010 | Design and methodology. Study I used a non‐concurrent multiple baseline design, and Study II used a multiple probe across activities design. In both studies the interventions were based on a computerised verbal aid device, which provided instructions to support participants in performing daily activities. During the interventions verbal prompts were also given by a research assistant if instructions were not followed up by the participants. |
| Mihailidis 2004 | Design. The study used a withdrawal‐type ABAB single subject research design. |
| Mihailidis 2008 | Design. The study used a single subject research design with two phases. |
| Ownby 2012 | Participants. The obtainment of a formal diagnosis of dementia was no part of the data collection. |
| Spring 2011 | Participants and methodology. Participants between 18 and 75 years of age who met DSM‐IV criteria for panic, generalized or social anxiety, or post‐traumatic stress disorders, with or without major depression. The intervention was a treatment delivered by trained case managers who used the Coordinated Anxiety Learning and Management (CALM) system to track symptoms, deliver cognitive behavioural treatment, and guide medication management. |
Characteristics of ongoing studies [ordered by study ID]
ISRCTN 86537017.
| Trial name or title | The ATTILA Trial: Assistive Technology and Telecare to maintain Independent Living At home for people with dementia. |
| Methods | Randomised controlled multi‐centre clinical trial. |
| Participants | 500 participants with a Clinical Dementia Rating of 1, 2, or 3. |
| Interventions | Based on assessments of participants, specific areas where ATT can support participants and caregivers in the safe undertaking of occupational activities will be identified. The experimental group receives specific types of ATT that will meet the identified needs. Simple, battery operated, stand‐alone technologies and/or telecare will be installed at the home of participants. The control group receives no treatment. Study participants will be followed up for 2 years. |
| Outcomes | Primary outcome measures are time from randomisation to admission into long‐term care and cost‐effectiveness. Secondary outcome measures are caregiver burden; quality of life; number and severity of adverse events; and acceptability, applicability and reliability of the used devices. |
| Starting date | 1 June 2013 |
| Contact information | Prof. R Howard Department of Old Age Psychiatry The Institute of Psychiatry Box 070 De Crespigny Park London SE5 8AF United Kingdom robert.j.howard@kcl.ac.uk |
| Notes | Because of the heterogeneity of the AT devices used in the intervention it is uncertain whether this trial will meet the eligibility criteria for this review. |
Differences between protocol and review
The AgeLine database could not be accessed despite repeated attempts, and so it was not included in the search. No further alterations were made to the protocol. The following methods from the protocol could not be executed in this review, since no studies met the eligibility criteria.
Data extraction and management.
Assessment of risk of bias in included studies.
Measures of treatment effect.
Unit of analysis issues.
Dealing with missing data.
Assessment of heterogeneity.
Assessment of reporting bias.
Data synthesis.
Subgroup analysis and investigation of heterogeneity.
Sensitivity analysis.
Some textual edits have been made to the Background section and an additional reference was included, to provide the most recent numbers on the number of people with dementia worldwide. These edits have not altered the content of the Background section.
Contributions of authors
HvdR and JW: wrote the draft protocol, executed the search strategy, reviewed the search results and drafted the Results and Discussion sections.
CP: reviewed the search results.
HvdR: developed the search strategy.
RMD and MO: commented on, and edited the draft protocol and the review.
Sources of support
Internal sources
No sources of support supplied
External sources
Noaber Foundation, Netherlands.
-
NIHR, UK.
This review was supported by the National Institute for Health Research (NIHR), via Cochrane Infrastructure funding to the Cochrane Dementia and Cognitive Improvement group. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, National Health Service or the Department of Health
Declarations of interest
Henriëtte G Van der Roest – none known. Jennifer Wenborn – none known. Channah Pastink – none known. Rose‐Marie Dröes – none known. Martin Orrell – none known.
New
References
References to studies excluded from this review
Hofmann 2003 {published data only}
- Hofmann M, Rösler A, Schwarz W, Müller‐Spahn F, Krauchi K, Hock C, et al. Interactive computer‐training as a therapeutic tool in Alzheimer's disease. Comprehensive Psychiatry 2003;44(3):213‐9. [PUBMED: 12764709] [DOI] [PubMed] [Google Scholar]
Labelle 2006 {published data only}
- Labelle K, Mihailidis A. The use of automated prompting to facilitate handwashing in persons with dementia. American Journal of Occupational Therapy 2006;60(4):442‐50. [PUBMED: 16915875] [DOI] [PubMed] [Google Scholar]
Lancioni 2010 {published data only}
- Lancioni GE, Singh NN, O'Reilly MF, Sigafoos J, Tatulli E, Rigante V, et al. Technology‐aided verbal instructions to help persons with mild or moderate Alzheimer’s disease perform daily activities. Research in Developmental Disabilities 2010;31(6):1240‐50. [PUBMED: 20696547] [DOI] [PubMed] [Google Scholar]
Mihailidis 2004 {published data only}
- Mihailidis A, Barbenel JC, Fernie G. The efficacy of an intelligent cognitive orthosis to facilitate handwashing by persons with moderate to severe dementia. Neuropsychological Rehabilitation 2004;14(1‐2):135‐71. [DOI: 10.1080/09602010343000156] [DOI] [Google Scholar]
Mihailidis 2008 {published data only}
- Mihailidis A, Boger JN, Craig T, Hoey J. The COACH prompting system to assist older adults with dementia through handwashing: an efficacy study. BMC Geriatrics 2008;8:28. [PUBMED: 18992135] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ownby 2012 {published data only}
- Ownby RL, Hertzog C, Czaja SJ. Tailored information and automated reminding to improve medication adherence in Spanish‐ and English‐speaking elders treated for memory impairment. Clinical Gerontologist 2012;35(3):221‐38. [PUBMED: 24244067] [DOI] [PMC free article] [PubMed] [Google Scholar]
Spring 2011 {published data only}
- Spring B, Ferguson MJ. CALM technology‐supported intervention: Synopsis of evidence for an emerging class of practice tool. Translational Behavioral Medicine 2011;1(1):8‐9. [PUBMED: 24073025] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to ongoing studies
ISRCTN 86537017 {published data only}
- ISRCTN 86537017. The ATTILA Trial: Assistive Technology and Telecare to maintain Independent Living At home for people with dementia – a randomised controlled multi‐centre clinical trial. controlled‐trials.com/ISRCTN86537017 (first received 14 December 2012).
Additional references
Agüero‐Torres 1998
- Agüero‐Torres H, Fratiglioni L, Winblad B. Natural history of Alzheimer's disease and other dementias: review of the literature in the light of the findings from the Kungsholmen Project. International Journal of Geriatric Psychiatry 1998;13(11):755‐66. [PUBMED: 9850872] [DOI] [PubMed] [Google Scholar]
Alzheimer's Disease International 2010
- Wimo A, Prince M, Alzheimer's Disease International. World Alzheimer Report 2010. The global economic impact of dementia. https://www.alz.co.uk/research/world‐report‐2010 29 May 2011. [http://www.webcitation.org/6r3DHEv5j]
Alzheimer's Disease International 2013
- Prince M, Guerchet M, Prina M, Alzheimer's Disease International. The global impact of dementia 2013‐2050. https://www.alz.co.uk/research/GlobalImpactDementia2013.pdf 27 August 2014. [http://www.webcitation.org/6r3DhHJ7c]
Alzheimer's Society 2014
- Alzheimer's Society. Dementia‐friendly technology. A charter that helps every person with dementia benefit from technology that meets their needs. https://www.alzheimers.org.uk/download/downloads/id/2256/dementia‐friendly_technology_charter.pdf 13 November 2016. [http://www.webcitation.org/6r3DvhyCu]
Alzheimer's Society 2015
- Alzheimer's Society. Factsheet 437: Assistive technology – devices to help with everyday living. https://www.alzheimers.org.uk/download/downloads/id/1779/factsheet_assistive_technology_%E2%80%93_devices_to_help_with_everyday_living.pdf 19 February 2017. [http://www.webcitation.org/6oOTehw7L]
American Psychiatric Association 1994
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM‐IV). Washington DC: American Psychiatric Association, 1994. [Google Scholar]
Bemelmans 2012
- Bemelmans R, Gelderblom GJ, Jonker P, Witte LD. Socially assistive robots in elderly care: a systematic review into effects and effectiveness. Journal of the American Medical Directors Association 2012;13(2):114‐20. [PUBMED: 21450215] [DOI] [PubMed] [Google Scholar]
Bharucha 2009
- Bharucha AJ, Anand V, Forlizzi J, Dew MA, Reynolds CF 3rd, Stevens S, et al. Intelligent assistive technology applications to dementia care: current capabilities, limitations, and future challenges. American Journal of Geriatric Psychiatry 2009;17(2):88‐104. [PUBMED: 18849532] [DOI] [PMC free article] [PubMed] [Google Scholar]
Buettner 2010
- Buettner LL, Yu F, Burgener SC. Evidence supporting technology‐based interventions for people with early‐stage Alzheimer’s disease. Journal of Gerontological Nursing 2010;36(10):15‐9. [PUBMED: 20873690] [DOI] [PubMed] [Google Scholar]
Cahill 2007
- Cahill S, Macijauskiene J, Nygård A‐M, Faulkner J‐P, Hagen I. Technology in dementia care. Technology and Disability 2007;19(2):55‐60. [Google Scholar]
Caprani 2006
- Caprani N, Greaney J, Porter N. A review of memory aid devices for an ageing population. Psychology Journal 2006;4(3):205‐43. [Google Scholar]
Carswell 2009
- Carswell W, McCullagh PJ, Augusto JC, Martin S, Mulvenna MD, Zheng H, et al. A review of the role of assistive technology for people with dementia in the hours of darkness. Technology and Health Care 2009;17(4):281‐304. [PUBMED: 19822946] [DOI] [PubMed] [Google Scholar]
Ciro 2014
- Ciro CA. Maximizing ADL performance to facilitate aging in place for people with dementia. Nursing Clinics of North America 2014;49(2):157‐69. [PUBMED: 24846465] [DOI] [PubMed] [Google Scholar]
Davies 2009
- Davies RJ, Nugent CD, Donnelly M, Hettinga M, Meiland F, Moelaert F, et al. A user‐driven approach to develop a cognitive prosthetic to address unmet needs of people with mild dementia. Pervasive and Mobile Computing 2009;5(3):253‐67. [DOI: 10.1016/j.pmcj.2008.07.002] [DOI] [Google Scholar]
Dröes 2010
- Dröes RM, Bentvelzen SCL, Meiland F, Craig D. Dementia‐related and other factors to be taken into account when developing ICT‐support for people with dementia ‐ Lessons from field trials. In: Mulvenna MD, Nugent CD editor(s). Supporting people with dementia using pervasive health technologies. Series: Advanced information and knowledge processing. London: Springer‐Verlag, 2010:113‐30. [DOI: 10.1007/978-1-84882-551-2_8] [DOI] [Google Scholar]
European Commission 2010
- European Commission. e‐Inclusion. ec.europa.eu/information_society/activities/einclusion/index_en.htm 9 August 2010.
Fleming 2014
- Fleming A, Sum S. Empirical studies on the effectiveness of assistive technology in the care of people with dementia: a systematic review. Journal of Assistive Technology 2014;8(1):14‐34. [DOI: 10.1108/JAT-09-2012-0021] [DOI] [Google Scholar]
Fritschy 2004
- Fritschy EP, Kessels RP, Postma A. External memory aids for patients with dementia: a literature study on efficacy and applicability [Externe hulpmiddelen ter ondersteuning van het geheugen van patiënten met dementie: een literatuurstudie naar de effectiviteit en toepasbaarheid.]. Tijdschrift voor Gerontologie en Geriatrie 2004;35(6):234‐9. [PUBMED: 15704603] [PubMed] [Google Scholar]
Gillespie 2012
- Gillespie A, Best C, O'Neill B. Cognitive function and assistive technology for cognition: a systematic review. Journal of the International Neuropsychological Society 2012;18(1):1‐19. [PUBMED: 22152338] [DOI] [PubMed] [Google Scholar]
Hanson 2007
- Hanson E, Magnusson L, Arvidsson H, Claesson A, Keady J, Nolan M. Working together with persons with early stage dementia and their family members to design a user‐friendly technology‐based support service. Dementia 2007;6(3):411‐34. [DOI: 10.1177/1471301207081572] [DOI] [Google Scholar]
Huschilt 2012
- Huschilt J, Clune L. The use of socially assistive robots for dementia care. Journal of Gerontological Nursing 2012;38(10):15‐9. [PUBMED: 22998095] [DOI] [PubMed] [Google Scholar]
Improvement and Efficiency West Midlands 2013
- Association of Directors of Adult Social Services, Improvement, Efficiency West Midlands. Community Gateway CIC. Maximising the potential for the use of assistive technology: an information toolkit to support people with dementia, their carers and dementia services. Improvement and Efficiency West Midlands, 2013. [Google Scholar]
Kamel Boulous 2009
- Kamel Boulos MN, Lou RC, Anastasiou A, Nugent CD, Alexandersson J, Zimmermann G, et al. Connectivity for healthcare and well‐being management: examples from six European projects. International Journal of Environmental Research and Public Health 2009;6(7):1947‐71. [PUBMED: 19742164] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lauriks 2007
- Lauriks S, Reinersmann A, Roest HG, Meiland FJ, Davies RJ, Moelaert F, et al. Review of ICT‐based services for identified unmet needs in people with dementia. Ageing Research Reviews 2007;6(3):223‐46. [PUBMED: 17869590] [DOI] [PubMed] [Google Scholar]
Lindenberger 2008
- Lindenberger U, Lovden M, Schellenbach M, Li SC, Kruger A. Psychological principles of successful aging technologies: a mini‐review. Gerontology 2008;54(1):59‐68. [PUBMED: 18259095] [DOI] [PubMed] [Google Scholar]
Liu 2007
- Liu KP, Chan CC, Chu MM, Ng TY, Chu LW, Hui FS, et al. Activities of daily living performance in dementia. Acta neurologica Scandinavica 2007;116(2):91‐5. [PUBMED: 17661793] [DOI] [PubMed] [Google Scholar]
Martin 2008
- Martin S, Kelly G, Kernohan WG, McCreight B, Nugent C. Smart home technologies for health and social care support. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD006412.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mayer 2013
- Mayer JM, Zach J. Lessons learned from participatory design with and for people with dementia. MobileHCI 2013 Proceedings of the 15th international conference on Human‐computer interaction with mobile devices and services; 2013 Aug 27‐30; Munich, Germany. 2013:540‐5. [DOI: 10.1145/2493190.2494436] [DOI]
Maylor 2002
- Maylor EA, Smith G, Della Sala S, Logie RH. Prospective and retrospective memory in normal aging and dementia: an experimental study. Memory & Cognition 2002;30(6):871‐84. [PUBMED: 12450091] [DOI] [PubMed] [Google Scholar]
Medical Research Council 2008
- Craig P, Dieppe P, Michie S, Nazareth I, Petticrew M, on behalf of the MRC. Developing and evaluating complex interventions: new guidance. 2008. [DOI] [PMC free article] [PubMed]
Meiland 2007
- Meiland FJ, Reinersmann A, Bergvall‐Kareborn B, Craig D, Moelaert F, Mulvenna MD, et al. COGKNOW development and evaluation of an ICT‐device for people with mild dementia. Studies in Health Technology and Informatics 2007;127:166‐77. [PUBMED: 17901610] [PubMed] [Google Scholar]
Meiland 2012
- Meiland FJM, Bouman AIE, Sävenstedt S, Bentvelzen S, Davies RJ, Mulvenna MD, et al. Usability of a new electronic assistive device for community‐dwelling persons with mild dementia. Aging & Mental Health 2012;16(5):584‐91. [PUBMED: 22360649] [DOI] [PubMed] [Google Scholar]
Meiland 2017
- Meiland F, Innes A, Mountain G, Robinson L, Roest H, García‐Casal JA, et al. Technologies to support community‐dwelling persons with dementia: a position paper on issues regarding development, usability, (cost‐) effectiveness, deployment and ethics. JMIR Rehabilitation and Assistive Technologies 2017;4(1):e1. [DOI: 10.2196/rehab.6376] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mohktari 2012
- Mokhtari M, Aloulou H, Tiberghien T, Biswas J, Racoceanu D, Yap P. New trends to support independence in persons with mild dementia ‐ A mini‐review. Gerontology 2012;58(6):554‐63. [PUBMED: 22677914] [DOI] [PubMed] [Google Scholar]
Mulvenna 2010
- Mulvenna MD, Nugent CD. Supporting people with dementia using pervasive health technologies. 1st Edition. London: Springer, 2010:226. [DOI: 10.1007/978-1-84882-551-2] [DOI] [Google Scholar]
Nugent 2007
- Nugent C, Mulvenna M, Moelaert F, Bergvall‐Kåreborn B, Meiland F, Craig D, et al. Home based assistive technologies for people with mild dementia. In: Okadome T, Yamazaki T, Makhtari M editor(s). Pervasive Computing for Quality of Life Enhancement. Vol. 4541, Lecture Notes in Computer Science, Berlin and Heidelberg: Springer, 2007:63‐9. [DOI: 10.1007/978-3-540-73035-4_7] [DOI] [Google Scholar]
Nugent 2008
- Nugent CD, Davies RJ, Donnelly MP, Hallberg J, Hariz M, Craig D, et al. The development of personalised cognitive prosthetics. 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2008 August 20‐25. Vancouver, BC, Canada, 2008:787‐90. [PUBMED: 19162773] [DOI] [PubMed]
Peterson 2012
- Peterson CB, Prasad NR, Prasad, R. The future of assistive technologies for dementia. Gerontechnology 2012;11(2):259‐65. [DOI: 10.4017/gt.2012.11.02.427.742] [DOI] [Google Scholar]
Rialle 2008
- Rialle V, Ollivet C, Guigui C, Herve C. What do family caregivers of Alzheimer's disease patients desire in smart home technologies? Contrasted results of a wide survey. Methods of Information in Medicine 2008;47(1):63‐9. [PUBMED: 18213430] [PubMed] [Google Scholar]
Rigaud 2011
- Rigaud AS, Pino M, Wu YH, Rotrou J, Boulay M, Seux ML, et al. Support for patients with Alzheimer’s disease and their caregivers by gerontechnology [L’aide aux personnes souffrant de maladie d’Alzheimer et à leurs aidants par les gérontechnologies]. Gériatrie et psychologie neuropsychiatrie du vieillissement 2011;9(1):91‐100. [PUBMED: 21586382] [DOI] [PubMed] [Google Scholar]
Sikkes 2009
- Sikkes SA, Lange‐de Klerk ES, Pijnenburg YA, Scheltens P, Uitdehaag BM. A systematic review of Instrumental Activities of Daily Living scales in dementia: room for improvement. Journal of Neurology, Neurosurgery, and Psychiatry 2009;80(1):7‐12. [PUBMED: 19091706] [DOI] [PubMed] [Google Scholar]
Sixsmith 2007
- Sixsmith AJ, Gibson G, Orpwood RD, Torrington JM. Developing a technology 'wish‐list' to enhance the quality of life of people with dementia. Gerontechnology 2007;6(1):2‐19. [DOI: 10.4017/gt.2007.06.01.002.00] [DOI] [Google Scholar]
Smith 2000
- Smith G, Della Sala S, Logie RH, Mayor EA. Prospective and retrospective memory in normal ageing and dementia: a questionnaire study. Memory 2000;8(5):311‐21. [DOI] [PubMed] [Google Scholar]
Span 2013
- Span M, Hettinga M, Vernooij‐Dassen M, Eefsting J, Smits C. Involving people with dementia in the development of supportive IT applications: a systematic review. Ageing Research Reviews 2013;12(2):535‐51. [PUBMED: 23318684] [DOI] [PubMed] [Google Scholar]
Sterns 2005
- Sterns AA. Curriculum design and program to train older adults to use personal digital assistants. Gerontologist 2005;45(6):828‐34. [PUBMED: 16326666] [DOI] [PMC free article] [PubMed] [Google Scholar]
Topo 2009
- Topo P. Technology studies to meet the needs of people with dementia and their caregivers: A literature review. Journal of Applied Gerontology 2009;28(1):5‐37. [DOI: 10.1177/0733464808324019] [DOI] [Google Scholar]
Trent Dementia Services Development Centre 2016
- Trent Dementia Services Development Centre. AT Dementia. https://www.atdementia.org.uk/ 13 November 2016.
Van der Roest 2008
- Roest HG, Meiland FJ, Haaker T, Reitsma E, Wils H, Jonker C, et al. Finding the service you need: human centered design of a Digital Interactive Social Chart in DEMentia care (DEM‐DISC). Studies in Health Technology and Informatics 2008;137:210‐24. [PUBMED: 18560083] [PubMed] [Google Scholar]
Van der Roest 2009
- Roest HG, Meiland FJ, Comijs HC, Derksen E, Jansen AP, Hout HP, et al. What do community‐dwelling people with dementia need? A survey of those who are known to care and welfare services. International Psychogeriatrics / IPA 2009;21(5):949‐65. [PUBMED: 19602305] [DOI] [PubMed] [Google Scholar]
Van Wielingen 2004
- Wielingen LE, Tuokko HA, Cramer K, Mateer CA, Hultsch DF. Awareness of financial skills in dementia. Aging & Mental Health 2004;8(4):374‐80. [PUBMED: 15370053] [DOI] [PubMed] [Google Scholar]
Vilans 2016
- Vilans. Kenniscirkel Domotica. Dementia File living at home [Dossier Dementie thuiswonen]. http://www.domoticawonenzorg.nl/dwz/dossier‐dementie‐thuiswonen.html 13 November 2016. [www.webcitation.org/6lzYIyB5b]
Westphal 2010
- Westphal A, Dingjan P, Attoe R. What can low and high technologies do for late‐life mental disorders?. Current Opinion in Psychiatry 2010;23(6):510‐5. [PUBMED: 20717039] [DOI] [PubMed] [Google Scholar]
WHO 2007
- World Health Organization. International statistical Classification of Diseases and related health problems (10th Revision, Version for 2007). apps.who.int/classifications/apps/icd/icd10online/ 29 May 2011.
Wimo 1999
- Wimo A, Winblad B, Grafstrom M. The social consequences for families with Alzheimer's disease patients: potential impact of new drug treatment. International Journal of Geriatric Psychiatry 1999;14(5):338‐47. [PUBMED: 10389036] [DOI] [PubMed] [Google Scholar]
Öhman 2001
- Öhman A, Nygård L, Borell L. The vocational situation in cases of memory deficits or younger‐onset dementia. Scandinavian Journal of Caring Sciences 2001; Vol. 15, issue 1:34‐43. [DOI: 10.1046/j.1471-6712.2001.1510034.x] [DOI]


