Abstract
Background
Cystic fibrosis is a multi‐system disease characterised by the production of thick secretions causing recurrent pulmonary infection, often with unusual bacteria. This leads to lung destruction and eventually death through respiratory failure. There are no antibiotics in development that exert a new mode of action and many of the current antibiotics are ineffective in eradicating the bacteria once chronic infection is established. Antibiotic adjuvants ‐ therapies that act by rendering the organism more susceptible to attack by antibiotics or the host immune system, by rendering it less virulent or killing it by other means, are urgently needed.
Objectives
To determine if antibiotic adjuvants improve clinical and microbiological outcome of pulmonary infection in people with cystic fibrosis.
Search methods
We searched the Cystic Fibrosis Trials Register which is compiled from database searches, hand searches of appropriate journals and conference proceedings.
Date of most recent search: 26 July 2012.
We also searched MEDLINE (all years) on 23 February 2013 and ongoing trials registers on 13 February 2013.
Selection criteria
Randomised controlled trials and quasi‐randomised controlled trials of a therapy exerting an antibiotic adjuvant mechanism of action compared to placebo or no therapy for people with cystic fibrosis.
Data collection and analysis
The authors independently assessed and extracted data from identified studies.
Main results
We identified eighteen studies of which four are included that examined antibiotic adjuvant therapies, three studies are ongoing. The included studies involve the assessment of β‐carotene, garlic and zinc supplementation and KB001 (a biological agent). No therapy demonstrated a significant effect upon pulmonary function, pulmonary exacerbations or quality of life. The study of zinc supplementation reports a reduction in the requirement of oral antibiotics but not of intravenous antibiotics, an effect that is difficult to understand.
Authors' conclusions
We could not identify an antibiotic adjuvant therapy that could be recommended for the treatment of lung infection in those with cystic fibrosis. The emergence of increasingly resistant bacteria makes the reliance on antibiotics alone challenging for cystic fibrosis teams. There is a need to explore alternative strategies, such as the use of adjuvant therapies. Further research is required to provide future therapeutic options.
Plain language summary
Non‐antibiotic agents to assist antibiotic treatment for lung infection in cystic fibrosis
People with cystic fibrosis suffer lung infections due to thick secretions and commonly suffer infections caused by unusual bacteria, including the bacteria Pseudomonas aeruginosa. These bacteria become resistant to treatment with antibiotics. Long‐term infection leads to a poorer quality of life and reduced lung function. There are no new antibiotics currently in development that use a new type of action. New agents ‐ antibiotic adjuvants ‐ are needed to make bacteria more sensitive to either antibiotics or to the immune system, and to interfere with the formation of colonies of bacteria in the lungs.
We identified seven approaches for enhancing antibiotic action including sugars, garlic, monoclonal antibodies (many copies of one type of antibody, each made in a laboratory from a single copy of a human antibody), chick yolk antibodies, bacteriophages (viruses that infect bacteria), beta‐carotene and zinc supplementation. We included four trials of these therapies in our review (one each of beta‐carotene, garlic, zinc and a monoclonal antibody). All trials compared the active treatment to a placebo and looked at continuing long‐term treatment rather than treatment for a short‐term severe infection (the shortest trial was for 28 days and the longest was for one year). Neither beta‐carotene or garlic showed a significant improvement in clinical state or the number of infections. The study of zinc supplementation showed fewer oral antibiotics were needed, but the same was not true for intravenous antibiotics. It is difficult to explain this discrepancy and so the significance of the effect of zinc remains uncertain. The zinc study published only selected results, however the authors have kindly provided additional data. The monoclonal antibody study (KB001) demonstrated safety and tolerability.
We were unable to identify any therapy to enhance the actions of antibiotics which showed a significant benefit for either lung function, rate of infection or quality of life. Further randomised controlled trials are required before the routine use of any of these therapies can be recommended.
Background
Description of the condition
Cystic fibrosis (CF) is a multi‐system disease characterised by the production of thick secretions causing recurrent pulmonary infection and pancreatic malabsorption. The altered lung environment in people with CF provides an ideal niche for bacteria such as Pseudomonas aeruginosa (P. aeruginosa) to flourish and chronic infection to develop. This results in damage to the airways leading to a decline in lung function that is largely responsible for the morbidity and mortality in CF. Over the last two decades more efficient treatment of pulmonary infections has contributed to a large increase in life‐expectancy (Dodge 2007). The mean life expectancy for a baby born in 2003 was 42 years for a boy and 36 years for a girl (Dodge 2007). However, the adaptive behaviour of P. aeruginosa enables it to rapidly evolve in response to selective pressures exerted by the host environment and the use of bactericidal antibiotics. Thus, the increased use of broad spectrum antimicrobials has resulted in a reduction in their efficacy and an increase in bacterial resistance. In addition to P. aeruginosa, other important, antibiotic‐resistant pathogens can be found in the lungs of people with CF such as Burkholderia cepacia (B. cepacia) and methicillin‐resistant Staphylococcus aureus (MRSA).
Description of the intervention
Novel agents are urgently needed, which act differently to antibiotics and can eradicate the organism without selecting for resistance and whilst re‐sensitising them to readily available antibiotics. Antibiotics commonly act by killing the organism or stopping its growth. Antibiotic adjuvants may exert their effect on the organism without killing it. Antibiotic adjuvant therapies are a diverse group of novel agents that are similar in that they act by interfering with a mechanism the organism uses to decrease its susceptibility to antibiotics; by reducing the organism's virulence; or by rendering the organism more susceptible to the host immune system. Such agents include: quorum sensing inhibitors (see below); agents that interfere with biofilm construction (the sugars fucose and galactose, and novel dendrimers acting on lectin blockade); efflux pump inhibitors that stop bacteria removing antibiotics from within the bacterial cell; glutamine as an amino acid supplement; and biological agents (such as bacteriophages) that infect bacteria causing their break‐down and demise. In the case of bacteriophages that may cause the death of the organism, they act by 'infecting' the bacteria and therefore act differently to, but alongside, conventional antibiotics.
Some of the agents that may be considered in the review are available direct to the consumer (e.g. garlic, zinc). It is therefore conceivable that the results of the study may alter patient behaviour directly.
How the intervention might work
Antibiotic adjuvants are varied in their design. Many of the bacteria causing infections in CF communicate via quorum sensing, such that the bacteria only produce virulence factors when the population of bacteria has reached a critical size. The organism can therefore remain invisible to the host immune system until the population is in a position to withstand such host attack. Some antibiotic adjuvant therapies interfere with this bacterial communication, thus potentiating the efficacy of conventional antibiotics (von Bodman 2008). Some approaches attach to the genetic material of the organism, preventing the bacteria from becoming more resistant to antibiotics and some therapies target other mechanisms exploited by bacteria, such as the ability to form biofilms. Other antibiotic adjuvant therapies include bacteriophages or their products, viruses that directly target the bacterial cell, invade it and kill it (Borysowski 2006).
Why it is important to do this review
Worldwide, there is a burgeoning interest in agents that may potentiate, refine or replace the action of antibiotics without exerting selective pressure for antibiotic resistance. It is suggested that many of these agents are less susceptible to becoming inactive (due to the organism developing resistance toward them) because in many cases they do not directly influence the organisms' ability to reproduce. Currently both chronic pulmonary infections and acute infective exacerbations are difficult to treat and eradication of infection may be only temporary (Langton‐Hewer 2009). With research proceeding along diverse paths and individuals making decisions regarding supplementing conventional treatment with high‐street products, there is a need to evaluate current evidence to advise patients and their clinicians and direct research targets.
Objectives
To determine if antibiotic adjuvant therapies improve clinical and microbiological outcome of pulmonary infection in people with CF.
Methods
Criteria for considering studies for this review
Types of studies
Randomised controlled trials and quasi‐randomised controlled trials.
Types of participants
Adults and children with CF, diagnosed using the Cystic Fibrosis Foundation consensus statement (Rosenstein 1998). Therefore, a diagnosis of CF should be based on:
presence of one or more characteristic phenotypic features;
or a positive newborn screening test result;
-
or a history of CF in a sibling and laboratory evidence of an abnormality in the cystic fibrosis transmembrane regulator (CFTR) as documented by:
elevated sweat chloride concentration; or
identification of mutations in each CFTR gene known to cause CF; or
in vivo demonstration of characteristic abnormalities in ion transport across the nasal epithelium.
As no standardised, validated definitions of acute exacerbation or chronic infection exists, we have employed the definitions employed by the CF Trust Antibiotic Working Group (UK Cystic Fibrosis Trust Antibiotic Working Group) alongside those identified by Rosenfeld (Rosenfeld 2001). An acute exacerbation will be defined as at least four of the following (UK Cystic Fibrosis Trust Antibiotic Working Group):
increased productive cough or breathlessness;
changes in the appearance or volume of sputum;
new signs on auscultation;
new chest radiograph signs;
loss of appetite;
fall in respiratory function;
fever requiring treatment with intravenous antibiotics.
Alternatively, an acute exacerbation was defined by the following score meeting or exceeding 2.6 (Rosenfeld 2001).
| Feature | Score |
| Decreased exercise tolerance | 1.8 |
| Increased cough | 1.5 |
| Increased sputum/cough congestion | 1.5 |
| School or work absenteeism | 1.6 |
| Increased adventitial sounds on lung examination | 1.2 |
| Decreased appetite | 1.1 |
A chronic P. aeruginosa infection will be defined by more than 50% of months in a year when samples had been taken beingP. aeruginosa culture‐positive (Lee 2004). We previously had used the UK Cystic Fibrosis Trust definition of two or more occasions of P. aeruginosa isolation over six months (UK Cystic Fibrosis Trust 2004); however, the definition cited above more closely replicates current clinical practice.
In the case of studies including both clinical scenarios or less defined criteria, we shall aim to manage these separately using a pragmatic approach.
Types of interventions
Antibiotic adjuvant agents compared to conventional antibiotics (either alone or in combination), placebo or no therapy via any route of administration.
Antibiotic adjuvant agents are those which augment the host immune response or potentiate antibiotic action. The adjuvants themselves may exert a direct killing or bacteriostatic effect on the organism but their primary role is to augment the co‐administered antibiotics' effect. Such adjuvants include those that exert an effect on bacterial susceptibility to antibiotics and include efflux pump inhibitors (agents that block the action of efflux pumps ‐ pumps on the cell membrane that remove toxic substances from within the bacterium) or quorum sensing inhibitors. Other adjuvants change the physical resistance of an infection (e.g. lectin inhibitors) or those that act at the genetic level of the organism to prevent the acquisition of antibiotic resistance (e.g. anti‐sense strategies).
Biological agents (such as bacteriophages) were eligible for inclusion in this review. Agents were considered which are intended to treat bacteria, fungi and viruses.
We excluded trials of agents that physically alter the host environment (e.g. gene therapy, immunotherapy); anti‐inflammatory agents (e.g. steroids, ibuprofen); agents that alter mucociliary clearance (e.g. mannitol, hypertonic saline and dornase alfa); physical interventions (such as physiotherapy and exercise); and environmental changes (such as an infection control policy).
Types of outcome measures
Primary outcomes
Pulmonary exacerbations (protocol defined)
-
Respiratory function
per cent predicted forced expiratory volume at one second (FEV1) values for age, sex and height
per cent predicted forced vital capacity (FVC) values for age, sex and height
other validated measures of respiratory function
Quality of life measures (CF‐validated)
Secondary outcomes
Time to next course of antibiotics (oral or intravenous)
Antibiotic consumption (days of antibiotic use)
Time off work or school
Emergence of new CF pathogens
Quantitative microbiology (change in numbers of pathogens isolated from respiratory tract secretion culture)
Development of allergic bronchopulmonary aspergillosis (ABPA) (increase in serum IgE with clinical or radiological features)
Treatment burden (e.g. Challenges of Living with CF evaluation)
-
Change in inflammatory markers
serum (C‐reactive protein (CRP), erythrocyte sedimentation rate (ESR))
sputum (IL‐8, leukotrienes, cytokines)
Mortality
-
Adverse events
mild (not requiring treatment)
moderate (requiring treatment or admission)
severe (life‐threatening)
These outcomes will be assessed for both chronic infection and acute infective exacerbation. Results will be assessed and analysed separately for the two clinical scenarios at the time‐points detailed below (Data extraction and management).
Search methods for identification of studies
Electronic searches
We searched for relevant studies from the Group's Cystic Fibrosis Trials Register using the terms 'pulmonary infection' AND 'non‐antibiotic'. The Cystic Fibrosis Trials Register is compiled from electronic searches of the Cochrane Central Register of Controlled Trials (CENTRAL) (updated each new issue of The Cochrane Library), quarterly searches of MEDLINE, a search of EMBASE to 1995 and the prospective hand‐searching of two journals ‐ Pediatric Pulmonology and the Journal of Cystic Fibrosis. Unpublished work is identified by searching the abstract books of three major cystic fibrosis conferences: the International Cystic Fibrosis Conference; the European Cystic Fibrosis Conference and the North American Cystic Fibrosis Conference. For full details of all searching activities for the register, please see the relevant sections of the Cochrane Cystic Fibrosis and Genetic Disorders Group Module.
Date of last search of the Cystic Fibrosis Trials Register: 26 July 2012.
We also searched MEDLINE separately; the search strategy is given in the appendices (Appendix 2).
Date of last search of MEDLINE: 23 February 2013.
We also searched the ongoing trials registries www.clinicaltrials.gov and http://isrctn.org/ on 23 February 2013 using the term 'cystic fibrosis' and limited to phase III and IV clinical trials.
Searching other resources
We hand‐searched reference lists of identified studies. We also contacted primary authors of identified trials and research institutions or biotech companies for unpublished studies.
Data collection and analysis
We used methods described in the Cochrane Handbook of Systematic Reviews of Interventions (Higgins 2011). We aimed to analyse acute infective exacerbations and chronic infection separately as defined previously in the section Types of participants. However, there were no two studies comparing the same intervention, or an intervention with outcomes in both acute and chronic conditions. We would aim to make these comparisons in future updates of this review if there are suitable new studies.
Selection of studies
Two authors (MH and DF) independently reviewed all potential studies for inclusion. These two authors examined the title and abstract of potential publications to remove those that did not meet inclusion criteria (e.g. single case reports, reviews etc.). We then examined the full text publications of the remaining studies to determine if they met the eligibility criteria. In the case that we were unable to reach agreement regarding the determination of eligibility by discussion, we would have sought the opinion of the third independent author (AS); however, there were no such occurrences. Authors examined publications potentially eligible for inclusion for duplication by comparing author, institution, study detail (intervention, dosing, timing etc.) and participant demographics.
Data extraction and management
Two authors (MH and DF) independently extracted information from the eligible studies using a data collection form. We discussed the results from each data extraction form to ensure agreement of interpretation; if there had been an absence of agreement, we planned to seek the opinion from a third author (AS).
It is possible that during a trial a participant may experience more than one exacerbation; in this event the shortest time between exacerbations would have been used.
Where studies, particularly with regard to chronic infection, take multiple assessments of individuals over a protracted time period, we would have defined time‐frames of follow‐up to represent short, medium and long term follow‐up. For the acute exacerbation studies we would have assessed data at time‐points of up to two weeks (standard duration of exacerbation therapy), over two weeks and up to six weeks (to assess efficacy of sustaining an effect). As previously discussed we would have assessed the outcome measure, time to next exacerbation as an indicator of long‐term effectiveness of antibiotic therapy. For the chronic infection studies, we would have taken assessment time points at one month, over one month and up to three months, over three months and up to six months, over six months and up to twelve months and annually thereafter. For future updates, if outcome data are recorded at other time points, we shall consider examining these as well.
Assessment of risk of bias in included studies
The authors assessed the risk of bias using the 'Risk of bias' assessment tool as documented in section 8.5 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). In particular, authors considered generation of allocation sequence and allocation concealment, blinding and incomplete outcome data. We also sought to identify selective outcome reporting by comparing those outcomes reported in the published paper to those considered in the protocol.
Measures of treatment effect
In identifying any treatment effect, we had planned to analyse the results of intervention measures separately for acute exacerbation and chronic infection. For dichotomous data we planned to analyse these by calculating the relative risk (RR) and its 95% confidence intervals (CIs) on an intention‐to‐treat basis (ITT). No studies reported mortality as an outcome measure, although we had planned to combine data in order to calculate the hazard ratio (HR) with 95% CIs, or if it is more appropriate, to describe the outcomes of mortality and adverse events descriptively.
For continuous outcome variables, we documented the mean difference (MD) of effect and standard deviation (SD) of each variable. We did not use a standardised scoring system; if studies had documented scores using the same scoring system, we had planned to calculate the MD and 95% CIs. If studies had used different validated scoring systems, we would have calculated the standardised mean difference (SMD) and 95% CIs.
No study reported data for 'time to next exacerbation'. If in future updates, such data become available, we plan to calculate the HR and its 95% CIs (appropriate as the risk of an exacerbation is not dependent upon other variables and should be uniform throughout the time period).
We describe count data narratively.
Unit of analysis issues
Studies with a cross‐over design are difficult to include as in many cases the duration of action of individual treatments may be prolonged. As a result we would only consider the first treatment phase of a cross‐over study and did not include such studies in full until such time that duration of action detail is understood, a washout period is provided, measures of treatment effect are available and the data are available from each treatment phase. When considering studies of long duration (and measures of number of exacerbations or requirement of antibiotics) repeated values will be available for each participant.
Dealing with missing data
In the case where it was obvious that any of the included publications missed important data from the review's primary or secondary outcome fields, the authors approached the primary investigator for clarification or more detail. This was the case for the study by Abdulhamid, who kindly provided the additional data (Abdulhamid 2008). If, in future, we do not receive any reply from primary investigators to possible enquires, we shall seek to account for all missing data by contact with the co‐investigators.
Assessment of heterogeneity
If we had identified more than one study of a common therapy, we would have performed a meta‐analysis of these studies. To assess heterogeneity between studies in the meta‐analysis, we would have used the I2 statistic (Higgins 2003). We would have based our interpretation of this statistic on the guide given in the Cochrane Handbook such that we would regard an I2 statistic below 40% as not important, between 30% and 60% as representing moderate heterogeneity, between 50% and 90% representing substantial and over 75% representing considerable heterogeneity (Higgins 2011). We would have interpreted this within the context of magnitude and direction of any effect and the strength of evidence for any heterogeneity, including a CI if possible for the I2 result.
Assessment of reporting biases
We contacted the primary author of each included study to determine if they knew of other studies which have been completed but may not have been published. This process might have identified some small studies and in this instance we could have determined the effects of these along with any reporting biases with the construction of funnel plots, accepting the multiple causes of asymmetry. When available, we made efforts to identify selective reporting of results by comparing the trial protocol (as published on a clinical trials register) with the final published results and by comparing the methods and results as detailed in the publication for any inconsistency in reporting.
Data synthesis
In the event of multiple studies we planned to analyse data using a fixed‐effects model unless the measures of heterogeneity were significantly high such that a random‐effects model would be most appropriate (RevMan 2011).
Subgroup analysis and investigation of heterogeneity
If we had identified a sufficient number of studies (more than ten), we planned to do the following subgroup analyses for each class of agent:
adults versus children;
-
presence or absence of chronic infection with
P. aeruginosa
Burkholderia cepacia (B. cepacia)
methicillin‐resistant Staphylococcus aureus (MRSA).
Sensitivity analysis
In the case that we had multiple analyses, we would have determined the effect our decisions made relating to arbitrary categorisations by repeating these analyses with different categorisations; for example, repeating the analyses of treatment effect with different measures of short, medium and long term. With regard to determining the effect of small studies on the end result, we would repeat the analyses without these small studies (e.g. participant numbers less than 20 in each group) to determine their effect.
Results
Description of studies
Results of the search
The search strategy identified a total of 27 studies. Of these studies, three investigating the use of antibiotic adjuvant therapies for chronic infection are included in the review (Abdulhamid 2008; Renner 2001; Smyth 2010) and 21 studies were excluded.
The remaining three studies are still ongoing and will be assessed for inclusion in the review when they are completed and results available (Molfino 2012; Walshaw 2011; Zabner 2009). The Molfino study is currently underway and is examining the efficacy of a biological agent in a Phase 2 trial in 180 participants with CF and Pseudomonas aeruginosa infection (Molfino 2012). The Walshaw study of OligG has been completed and the results are awaited (Walshaw 2011). OligoG is an oligosaccharide derived from alginate polysaccharide. It is suggested that this disrupts bacterial biofilm formation (a mode of bacterial growth associated with antibiotic tolerance and resistance). The Zabner study is evaluating xylitol (Zabner 2009). The putative mechanism of action is a reduction in airway surface liquid salt concentration, thus facilitating lysozyme and β‐defensins' protective action. One of the studies excluded on the basis of non‐randomisation (Durairaj 2007) is a pre‐cursor to the ongoing Zabner trial.
Included studies
Acute Respiratory Exacerbations
None of the included studies involved the treatment of acute infectious exacerbations.
Chronic infection
Four trials are included detailing studies of β‐carotene supplementation (Renner 2001), garlic supplementation (Smyth 2010), zinc supplementation (Abdulhamid 2008) and a safety study of a monoclonal antibody (a biological agent) (Milla 2013) which is currently the subject of a Phase 2 trial (Molfino 2012) and will be considered in future versions of this review. The trial of β‐carotene was reported in seven publications (Renner 2001).
All four trials were randomised, double‐blind, placebo‐controlled trials of parallel design. The ß‐carotene trial took place in Vienna, Austria (Renner 2001); the garlic trial was based in Nottingham, UK (Smyth 2010); the zinc supplementation trial was based in Michigan, USA (Abdulhamid 2008) and the KB001 trials was based in the USA (Milla 2013). All four trials were quite small, with the number of participants ranging from 24 to 34. Three trials and included both adults and children (age range 6.8 to 54 years) (Milla 2013; Renner 2001; Smyth 2010); the fourth trial recruited children only (age range 7 to 18 years) (Abdulhamid 2008). Duration of the trials ranged from 28 days (safety study Milla 2013) to one year (Abdulhamid 2008).
The dose regimens for each supplement are detailed in the tables (Characteristics of included studies). Three trials (Abdulhamid 2008; Renner 2001; Smyth 2010) reported pulmonary function tests, number of days of antibiotics required (Smyth only reported on intravenous antibiotics (Smyth 2010), but the other two trials reported both oral and intravenous antibiotics (Abdulhamid 2008; Renner 2001)) and plasma levels of the supplement. The safety study of KB001 included data on pulmonary exacerbations and comment on lung function, as well as comprehensive data on adverse effects (Milla 2013). Additional outcomes for each trial are listed in the tables (Characteristics of included studies).
Excluded studies
A total of 21 studies were excluded.
Acute Respiratory Exacerbation
One randomised trial examined the effect of immunoglobulin on the treatment of pulmonary exacerbations (Winnie 1989). This study was excluded as this was a trial of immunotherapy.
Chronic Infection
Publications that were obviously not relevant to the review after interrogation of the abstracts were excluded and are not listed in the review. A total of 21 studies are listed as excluded.
Ten studies were excluded for methodological reasons. Seven studies were not randomised or quasi‐randomised studies (Durairaj 2007; Homnick 1995; Kollberg 2003; Lands 2010; Safai‐Kutti 1991; Sagel 2011; Winklhofer‐Roob 1995) and one study was excluded as it was a single case report (Kutateladze 2008). One study randomised one arm to receive inhaled sugars (inhaled fucose and galactose) as treatment of their pulmonary exacerbation and the other arm received inhaled sugars accompanied by intravenous antibiotics (Hauber 2008). While these agents are specific substrates for the galactophilic and fucophilic lectins (cell surface proteins involved in bacterial aggregation) which are known in vitro to inhibit aggregation and promote dispersal of P. aeruginosa biofilms (Johansson 2008); these agents have a novel non‐osmotic mechanism of action and so were not immediately excluded. However, the study lacked a true control group and was excluded (Hauber 2008). A cross‐over trial of edetate sodium (EDTA) aerosolisation was excluded as data from the first phase of the study were unavailable for analysis (Brown 1985).
A further 10 studies were excluded as the interventions were not considered to be antibiotic adjuvant therapies. One study investigating the effect of polyunsaturated fats (Panchaud 2006) and another study looking at fatty acid supplements (Olveira 2010) were excluded as these appear to exert an independent anti‐inflammatory effect and so were considered not to meet our definition of an antibiotic adjuvant (as described in our exclusion criteria). A phase 2 study of a CXCR2 antagonist was excluded as the mechanism of action is anti‐inflammatory (Moss 2012). One study of magnesium (Gontijo‐Amaral 2012) and another of L‐arginine (Grasemann 2013) were excluded as an adjuvant effect does not appear to have been demonstrated in either study. Three studies of immunotherapy agents were also excluded (Kollberg 2010; Schuster 2011; Winnie 1989). Two studies of miglustat were excluded as this agent acts at the host genetic level and is not an adjuvant (Lebecque 2008; Leonard 2012).
Risk of bias in included studies
No trials were identified for acute respiratory exacerbations; all the following information relates to trials concerned with chronic infection. The risk of bias summary figure summarises these judgements (Figure 1).
1.

Methodological quality summary: review authors' judgements about each methodological quality item for each included study.
Allocation
Generation of sequence
The garlic study used a web‐based random allocation system provided by an external agency (Smyth 2010). The risk of bias was therefore low.
The β‐carotene study was described as 'randomised', unfortunately the randomisation method is not described (Renner 2001). In the zinc study, the assignment of study participants to the treatment group was reported to have occurred 'randomly' but no methods are described (Abdulhamid 2008) as is the case with the KB001 study (Milla 2013). We therefore judge these studies to have an unclear risk of bias.
Allocation concealment
From discussion with the trial authors, the garlic supplements were dispensed in coded opaque sealed containers. We judge the risk of bias to be low (Smyth 2010).
No details on allocation concealment are given for the β‐carotene study (Renner 2001), the zinc study (Abdulhamid 2008) or the KB001 study (Milla 2013), so we judge the risk of bias to be unclear for these.
Blinding
We judged two studies to have a low risk of bias due to blinding (Renner 2001; Smyth 2010). The β‐carotene study supplement was reported as being identical to a starch‐containing placebo (Renner 2001). The garlic supplement study detailed the flavour masking agent, described both active and placebo agents as identical and reported participant guesses of which treatment they thought they had received (no significant difference) (Smyth 2010).
The zinc supplement study detailed that the two groups were given either zinc gluconate or placebo preparations in capsules. The appearance of these capsules was unreported and so we judge the risk of bias to be unclear (Abdulhamid 2008). The KB001 study describe a placebo, but no other details are given and so we judge the risk of bias to be unclear (Milla 2013).
Incomplete outcome data
We judged two studies to have a low risk of bias due to incomplete outcome data (Renner 2001; Smyth 2010). Renner reports end results for all 24 participants identified in the methods section (Renner 2001). The garlic study randomised 34 participants of whom eight withdrew ‐ four in the garlic group ‐ one received lung transplant, one forgot to take capsules, one withdrew due to indigestion and one could not attend the second visit; four in the placebo group ‐ two forgot to take capsules, one withdrew due to halitosis and one could not attend the second visit. Data for the primary outcome (lung function) was missing for one participant in the placebo arm. Analysis was per protocol (Smyth 2010).
In the zinc study, 13 children in each group were randomised. One participant in the treatment group withdrew from the study and it is unclear whether this person was included in the final analysis (Abdulhamid 2008). We therefore judge there to be a high risk of bias since the incomplete outcome data were not addressed.
Selective reporting
Three studies were judged to have a low risk of bias from selective reporting (Milla 2013; Renner 2001; Smyth 2010). Without registration on a clinical trials registry it is difficult to determine the primary outcome measures for the β‐carotene study (Renner 2001); however, the impact of any bias due to this is likely to be minimal. The individual publications of the β‐carotene study (considered in isolation) each suffer from selective reporting as a result of duplicate publications with differing emphasis. The post‐hoc analysis describing an effect upon FEV1 in the 'younger' participants suffers from selective reporting as only data for the treatment group are described. However, with the post‐hoc analysis issue aside, when the individual reports are considered together, the results appear to not suffer from selective reporting, the risk of bias is therefore judged to be low (Renner 2001). For the garlic (Smyth 2010) and KB001 (Milla 2013) studies, the published protocols were compared to that of the trial publications and no issues of selective reporting were identified.
The study of zinc supplementation suffers considerably from selective reporting (Abdulhamid 2008). The groups were originally assigned to one of the two study arms 'randomly' without respect to their prior zinc status. However, the outcomes of these participants are reported in terms of prior zinc status. Zinc status was reported using a threshold of normal far below what is considered by their institutions' laboratory to be 'normal'. Indeed, they later state that their groups had normal zinc levels within the clinically acceptable normal range (Abdulhamid 2008). They justify their use of a higher range of 'normal' zinc level by citing their own previous work stating that those with lower zinc levels are more susceptible to infections. The authors also measured lung function as described in the protocol but this is also not reported. We therefore initially judged the bias introduced by this method of reporting to be high (Abdulhamid 2008). However, the authors provided the raw data without reference to prior zinc status, which has reduced our interpretation of the bias introduced.
Other potential sources of bias
Compliance to the β‐carotene protocol was assessed by patient report and capsule counting; however, the result was not reported in the study (Renner 2001). Again, compliance to the garlic protocol was assessed by participant report and capsule counting and was reported as equal in the garlic study (Smyth 2010). The risk of bias overall is therefore judged to be low.
Compliance to the zinc study protocol is reported only in terms of prior zinc status, the per protocol analysis of compliance is not provided (Abdulhamid 2008). Compiance to the safety study is not an issue as this was a study of a one‐dose infusion (Milla 2013). The sponsor of this study was the pharmaceutical company developing KB001.
Effects of interventions
Acute Respiratory Exacerbations
There were no trials included which considered acute respiratory exacerbations. All three included studies investigated antibiotic adjuvant therapies for chronic pulmonary infection (Abdulhamid 2008; Renner 2001; Smyth 2010).
Chronic Infection
For simplicity we have reported below the review's outcome measures that have been reported in the selected studies.
β‐carotene supplementation
One trial compared β‐carotene supplementation to placebo (Renner 2001).
Primary outcomes
1. Pulmonary exacerbations (protocol defined)
The trial investigators report a reduced pulmonary exacerbations rate in the treatment arm, defined as the number of days of antibiotic consumption, although this is not statistically significant; MD ‐8.00 (95% CI ‐18.78 to 2.78) (Analysis 1.1).
1.1. Analysis.
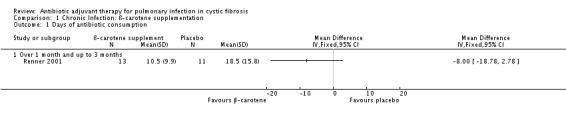
Comparison 1 Chronic Infection: ß‐carotene supplementation, Outcome 1 Days of antibiotic consumption.
2. Respiratory function
a. per cent predicted forced expiratory volume at one second (FEV1) values for age, sex and height
There appeared to be no effect upon lung function (FEV1 % predicted) between the two groups, MD ‐10.90% (95% CI ‐32.23% to 10.43%) (Analysis 1.2). The authors describe an apparently post‐hoc analysis of the 'younger patients' (undefined) and suggest their FEV1 'clearly improved'. Unfortunately only data for the treatment group are presented.
1.2. Analysis.
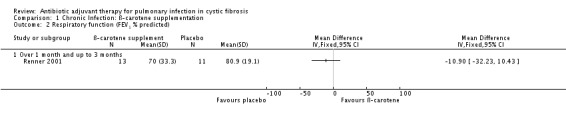
Comparison 1 Chronic Infection: ß‐carotene supplementation, Outcome 2 Respiratory function (FEV1 % predicted).
b. per cent predicted forced vital capacity (FVC) values for age, sex and height
This outcome was measured but not reported per protocol. The authors state that FVC did not change during the treatment (baseline: 87.6% ± 21.2%, week 12: 85.1% ± 24.6%, week 24: 85.6% ± 25.1%) (Renner 2001).
Secondary outcomes
2. Antibiotic consumption (days of antibiotic use)
As above, the protocol definition of pulmonary exacerbation was the antibiotic consumption rate in days (Analysis 1.1).
9. Mortality
There were no drop‐outs during the study and so we assume that no deaths occurred, although this wasn't explicitly discussed (Analysis 1.3).
1.3. Analysis.
Comparison 1 Chronic Infection: ß‐carotene supplementation, Outcome 3 Mortality.
10. Adverse events
There were no adverse events during the study. Some participants reported better tanning after exposure to sunlight (Renner 2001).
Garlic supplementation
One included trial investigated garlic supplementation (Smyth 2010). There were no statistically significant changes in clinical score and lung function. It was also not possible to detect a difference in quorum sensing molecules in plasma or sputum.
Primary outcomes
2. Respiratory function
a. per cent predicted forced expiratory volume at one second (FEV1) values for age, sex and height
The percentage change in predicted FEV1 between the treated and control groups did not reach significance; MD 1.59% (95% CI ‐7.49% to 10.67%) (Analysis 2.1)
2.1. Analysis.
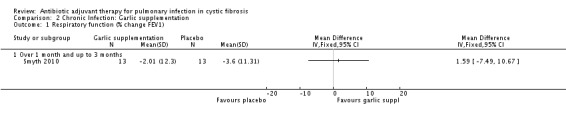
Comparison 2 Chronic Infection: Garlic supplementation, Outcome 1 Respiratory function (% change FEV1).
Secondary outcomes
2. Antibiotic consumption (days of antibiotic use)
Seven participants in the garlic group received intravenous antibiotics compared to five in the placebo group; however, days of antibiotic use was not reported.
9. Mortality
There were no deaths during the study period (Analysis 2.2) (Smyth 2010).
2.2. Analysis.
Comparison 2 Chronic Infection: Garlic supplementation, Outcome 2 Mortality.
10. Adverse events
a. mild (not requiring treatment)
Five participants in each group had abnormal liver function or triglyceride levels. Five participants in the garlic group reported minor adverse effects (diarrhoea (two participants), halitosis (two participants), abdominal pain (one participant), dysuria (one participant), minor haemoptysis (one participant)). One participant from the placebo group reported a minor haemoptysis (Analysis 2.3).
2.3. Analysis.
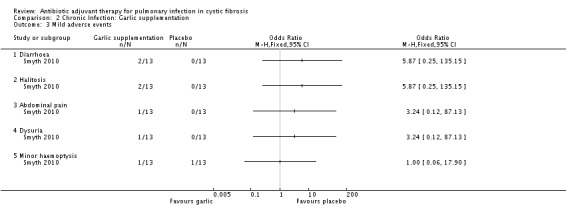
Comparison 2 Chronic Infection: Garlic supplementation, Outcome 3 Mild adverse events.
Zinc supplementation
One trial compared zinc supplementation to placebo (Abdulhamid 2008).
Primary outcomes
1. Pulmonary exacerbations (protocol defined)
The trial investigators do not report on their protocol definition of an exacerbation; they only report the number of episodes requiring intravenous and oral antibiotics (Abdulhamid 2008).
2. Respiratory function
a. per cent predicted forced expiratory volume at one second (FEV1) values for age, sex and height Measure given in the baseline characteristics, but the outcome is not reported in the published paper (Abdulhamid 2008). After contacting the authors, we received the data to allow us to analyse this outcome in the graphs and this showed a statistically non‐significant difference, MD ‐5.46 (95% CI ‐19.44 to 8.52) (Analysis 3.1).
3.1. Analysis.
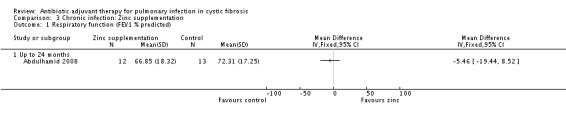
Comparison 3 Chronic infection: Zinc supplementation, Outcome 1 Respiratory function (FEV1 % predicted).
b. per cent predicted forced vital capacity (FVC) values for age, sex and height This measure given in the baseline characteristics, but the outcome was again not reported in the published paper (Abdulhamid 2008). Dr Abdulhamid supplied data to us for analysis and this showed a statistically non‐significant difference between groups, MD ‐1.75 (95% CI ‐13.09 to 9.59) (Analysis 3.2).
3.2. Analysis.
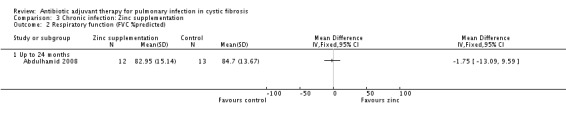
Comparison 3 Chronic infection: Zinc supplementation, Outcome 2 Respiratory function (FVC %predicted).
Secondary outcomes
2. Antibiotic consumption (days of antibiotic use)
The zinc supplement study reported that supplementation resulted in a borderline statistically significant reduction in the number of days of oral antibiotics (P = 0.05), the data are not given in the study, but were provided later (Abdulhamid 2008). Results showed a statistically significant result in favour of zinc supplementation for oral antibiotics alone, MD ‐17.74 (95% CI ‐26.98 to ‐8.50) (Analysis 3.3). However, the difference between groups was not statistically significant for duration of intravenous (IV) antibiotics, MD 0.52 (95% CI ‐3.07 to 4.11) (Analysis 3.4). When the data for both oral and IV antibiotics combined, supplied by Dr Abdulhamid, were analysed these showed a statistically significant difference between treatment and control groups of MD ‐17.22 (95% CI ‐27.06 to ‐7.38) (Analysis 3.5).
3.3. Analysis.
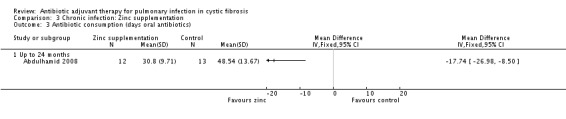
Comparison 3 Chronic infection: Zinc supplementation, Outcome 3 Antibiotic consumption (days oral antibiotics).
3.4. Analysis.
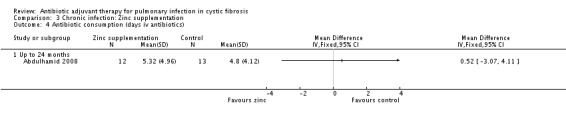
Comparison 3 Chronic infection: Zinc supplementation, Outcome 4 Antibiotic consumption (days iv antibiotics).
3.5. Analysis.
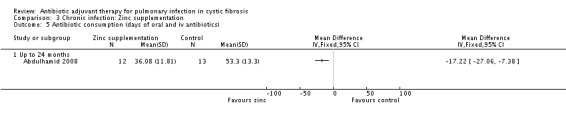
Comparison 3 Chronic infection: Zinc supplementation, Outcome 5 Antibiotic consumption (days of oral and iv antibiotics).
10. Adverse events
There were no adverse events reported in the zinc study.
KB001
One trial compared KB001 to placebo (Milla 2013). Data from this trial are only reported on the ongoing trials registry ClinicalTrials.gov.
Primary outcomes
1. Pulmonary exacerbations (protocol defined)
The trial investigators do not report on their protocol definition of an exacerbation, they report the number of events (Milla 2013). One participant who received a 3 mg/kg dose of KB001 experienced a pulmonary exacerbation, RR 1.58 (95% CI 0.07 to 35.32) (Analysis 4.1).
4.1. Analysis.
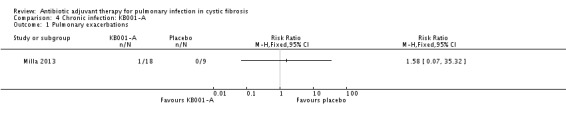
Comparison 4 Chronic infection: KB001‐A, Outcome 1 Pulmonary exacerbations .
2. Respiratory function
a. FEV1
Milla reports the number of episodes of a drop in lung function (FEV1); however, the lung function data in terms of the size of decrease in FEV1 are not reported (Milla 2013). Six participants who received a 3 mg/kg dose of KB001 experienced a decrease in FEV1, RR 4.74 (95% CI 0.28 to 79.44) (Analysis 4.2).
4.2. Analysis.
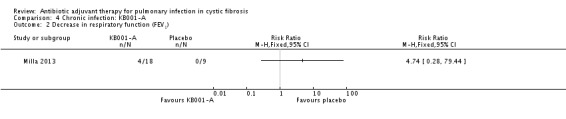
Comparison 4 Chronic infection: KB001‐A, Outcome 2 Decrease in respiratory function (FEV1).
Secondary outcomes
10. Adverse events
The primary outcome of this study was the number of participants experiencing adverse events. Seven of those who received placebo and all (18) of those who received KB001 experienced an adverse event, RR 1.30 (95% CI 0.90 to 1.87) (Analysis 4.3).
4.3. Analysis.

Comparison 4 Chronic infection: KB001‐A, Outcome 3 Number of participants experiencing an adverse event.
There is insufficient information to determine the severity of the events according to our classification of mild, moderate and severe, we have therefore used the classification reported in the trial ‐ 'serious' and 'other'. The trial reports the following serious adverse effects: a participant receiving a 3 mg/kg dose of KB001 developed bronchitis, RR 1.58 (95% CI 0.07 to 35.32); and a participant who received placebo developed sinusitis, RR 0.18 (95% CI 0.01 to 3.92) (Analysis 4.4). The 'other' events are described under group headings and presented in the analyses (Analysis 4.5; Analysis 4.6; Analysis 4.7; Analysis 4.8; Analysis 4.9; Analysis 4.10; Analysis 4.11; Analysis 4.12; Analysis 4.13; Analysis 4.14; Analysis 4.15).
4.4. Analysis.
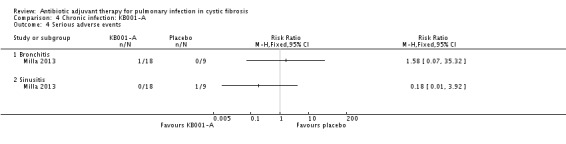
Comparison 4 Chronic infection: KB001‐A, Outcome 4 Serious adverse events.
4.5. Analysis.
Comparison 4 Chronic infection: KB001‐A, Outcome 5 Ear and labyrinth adverse effects.
4.6. Analysis.
Comparison 4 Chronic infection: KB001‐A, Outcome 6 Gastrointestinal adverse events.
4.7. Analysis.
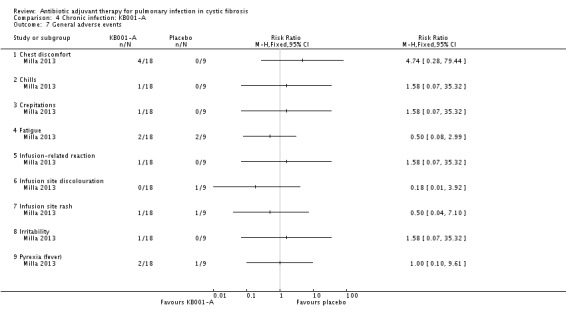
Comparison 4 Chronic infection: KB001‐A, Outcome 7 General adverse events.
4.8. Analysis.
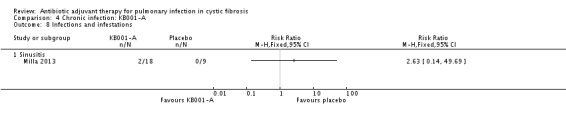
Comparison 4 Chronic infection: KB001‐A, Outcome 8 Infections and infestations.
4.9. Analysis.
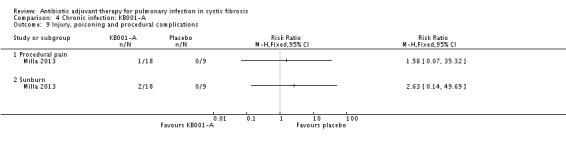
Comparison 4 Chronic infection: KB001‐A, Outcome 9 Injury, poisoning and procedural complications .
4.10. Analysis.
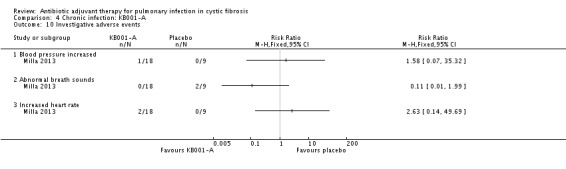
Comparison 4 Chronic infection: KB001‐A, Outcome 10 Investigative adverse events.
4.11. Analysis.
Comparison 4 Chronic infection: KB001‐A, Outcome 11 Nervous system adverse events.
4.12. Analysis.
Comparison 4 Chronic infection: KB001‐A, Outcome 12 Psychiatric adverse events.
4.13. Analysis.
Comparison 4 Chronic infection: KB001‐A, Outcome 13 Respiratory, thoracic and mediastinal adverse events.
4.14. Analysis.
Comparison 4 Chronic infection: KB001‐A, Outcome 14 Skin and subcutaneous tissue adverse events.
4.15. Analysis.
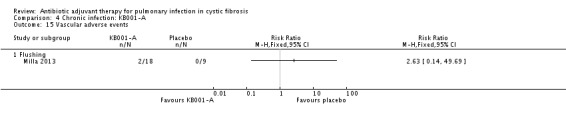
Comparison 4 Chronic infection: KB001‐A, Outcome 15 Vascular adverse events.
Discussion
Summary of main results
Various strategies have been investigated in an attempt to improve the current position where individuals are infected with resistant bacteria for which no successful treatment is available. Unfortunately, however, trials of these strategies have been of poor quality and often limited to observational or limited cohort studies. The more rigorous studies have suffered from either a lack of statistical power or robust outcome measures.
Neither the studies of β‐carotene supplementation or of garlic supplementation detected a statistically significant difference between those in the treatment and control groups (Renner 2001; Smyth 2010). It should also be noted that since the trial of β‐carotene was conducted, larger randomised trials with subsequent analysis have demonstrated an increased risk of development of certain malignancies (lung and stomach) in individuals with lung disease when given a daily 20 mg to 30 mg β‐carotene supplement (Druesne‐Pecollo 2010). The authors believe this precludes further investigation of the high‐dose supplementation regimen investigated in this review. The study of zinc supplementation suffers from selective reporting and so their finding of a reduction in the requirement of oral antibiotics of those in the supplemented group should be considered cautiously (Abdulhamid 2008). It is notable that the requirement of intravenous antibiotics did not differ between the two groups, an inconsistency that is difficult to explain. The study of KB001 was a safety study (Milla 2013) with limited data published online, but this has led to a repeat‐dose randomised controlled trial (Molfino 2012), where therapeutic efficacy will be considered as the primary outcome (measured by time‐to‐next antibiotic treatment) with safety and tolerability being secondary outcome measures.
Overall completeness and applicability of evidence
Some studies are yet to be completed and published and we await the results of these (Molfino 2012; Walshaw 2011; Zabner 2009). There is a preponderance to include participants in the younger age group ‐ children and adolescents. Potentially this group of younger individuals, prior to chronic colonisation with multiple‐resistant bacteria, could have most to gain from an intervention that might act to reduce infection and hence spare lung function and quality of life at an earlier age. However, the generalisability of this approach to the more unwell patients, who tend not to be included in such trials, is questionable.
Quality of the evidence
The quality of the evidence on which this review is based is poor and limited to four trials of four different interventions (Abdulhamid 2008; Milla 2013; Renner 2001; Smyth 2010).
Potential biases in the review process
One of the co‐authors of this review is lead investigator of one of the included trials (Smyth 2010).
Agreements and disagreements with other studies or reviews
There have been no previous systematic reviews of antibiotic adjuvant therapies for pulmonary infection in individuals with CF. Reviews of strategies in development stress the pre‐clinical nature of much of these approaches and unfortunately biotechnology companies developing new therapies may defer publication of commercially sensitive results.
Authors' conclusions
Implications for practice.
There continues to be no alternative to conventional antibiotics alone in the treatment of pulmonary infection in those with CF.
Implications for research.
Antibiotics remain the mainstay of treatment for pulmonary infection in CF. The emergence of increasingly resistant bacteria makes the reliance on antibiotics alone challenging for CF teams. There is a need to explore alternative strategies, such as the use of adjuvant therapies, that may prove a beneficial alternative. Novel strategies should aim to be founded on robustin vitro pre‐clinical development work that includes clinical isolates of mixed phenotypes of bacteria in models of infection that include both planktonic and biofilm modes of growth. Well‐designed placebo‐controlled double‐blind randomised trials of sufficient power and duration are required to determine the efficacy of any new strategy in both children and adults with CF. Such trials need to use outcome measures that are both objective and important to patients, specifically lung function, pulmonary exacerbation frequency, hospital attendance and measures of quality of life.
What's new
| Date | Event | Description |
|---|---|---|
| 29 June 2017 | Amended | Contact details updated. |
History
Protocol first published: Issue 4, 2009 Review first published: Issue 10, 2010
| Date | Event | Description |
|---|---|---|
| 7 May 2013 | New citation required but conclusions have not changed | No new studies were identified for inclusion in the review; limited results from a safety study previously listed as ongoing have been presented, but our conclusions have not changed. |
| 7 May 2013 | New search has been performed | A search of the Cystic Fibrosis Trials Register identified three references to two separate studies which were potentially eligible for inclusion in the review. Two of these references relate to the ongoing study identified for the initial version of the review (NCT00633191) now listed as (Kollberg 2010) which we have excluded as it refers to an immunotherapy. The remaining reference was excluded for the same reason (Winnie 1989). A renewed search of MEDLINE revealed six more studies. Two of these were excluded due to adjuvant activity being unclear (Gontijo‐Amaral 2012; Grasemann 2013). One study of immunotherapy (Schuster 2011) and one of anti‐inflammatory therapy (Moss 2012) were also excluded. Two studies were excluded as they were not randomised (Lands 2010; Sagel 2011). A search of ongoing trials registers identified two studies which are listed as ongoing and which will be assessed for inclusion when completed (Molfino 2012; Walshaw 2011). Four studies, which were listed as ongoing in the previous version of the review, have now been excluded. Two studies of miglustat were excluded as this is not an adjuvant (Lebecque 2008; Leonard 2012); the full publication of one of these studies was identified in the MEDLINE search (Leonard 2012). A safety study of a biological agent is now included (Milla 2013). The remaining cross‐over study was excluded after the study investigators confirmed that there were no first‐arm data available for analysis (Brown 1985). A study previously listed as 'Awaiting classification' has been excluded as the order of treatment was not randomised (Safai‐Kutti 1991). |
Acknowledgements
We would like to thank Dr Kevin Southern and Nikki Jahnke (Managing Editor, Cochrane Cystic Fibrosis & Genetic Disorders Group, Institute of Child Health) for their constructive advice in the preparation of this review and Natalie Yates (Trials Search Co‐ordinator) for her help in searching for studies.
Appendices
Appendix 1. Glossary
| Medical term | Explanation |
| antibiotic adjuvant | an agent that acts alongside an antibiotic, but itself has little or no bactericidal activity; it acts by increasing the organisms' susceptibility to the co‐administered antibiotic |
| bacteriophage | a virus that infects bacteria |
| biofilm | a thin layer of micro‐organisms (e.g. bacteria) that form on and coat various surfaces. Various substances coat the biofilm and provide protection to those organisms living within the biofilm |
| dendrimer | a large and complex molecule with a very well‐defined chemical structure |
| dysuria | painful urination |
| efflux pump | a mechanism located in the cell wall allowing substances toxic to the cell to be removed |
| efflux pump inhibitor | a molecule that interferes with the process of removing toxic substances and antibiotics from the cell |
| haemoptysis | coughing up of blood or of blood‐stained sputum |
| halitosis | bad breath |
| lectin | a sugar‐binding protein located on the surface off cells to allow cells to link to each other |
| quorum sensing inhibitors | molecules that interrupt the pathway of communication bacteria use to regulate expression of virulence factors |
| resistance | refers to the pathogens ability to withstand exposure to toxic substances, including antibiotics. |
| virulence factor | a substance produced by a pathogen that promotes its ability to cause disease |
Appendix 2. MEDLINE search strategy (1950 to present ) searched 11January 2010
| 1. | exp Cystic Fibrosis/ |
| 2. | cystic fibrosis.tw. |
| 3. | fibrocystic near disease near pancreas.tw. |
| 4. | mucoviscidos$.tw. |
| 5. | (cystic$ adj10 fibros$).tw. |
| 6. | 1 or 2 or 3 or 4 or 5 |
| 7. | (immune adj4 (stimulant$ or augment$ or activat$ or potentiat$ or modulat$)).tw. |
| 8. | exp Adjuvants, Immunologic/ |
| 9. | exp quorum sensing/ |
| 10. | Plant Extracts/ or Dietary Supplements/ or Vitamins/ or Phytotherapy/ or nutriceutical.mp. or Antioxidants/ or Nutritional Physiological Phenomena/ |
| 11. | (quorum and (sensing or quenching$)).tw. |
| 12. | (garlic or ajoene).tw. |
| 13. | exp Glutamine/ |
| 14. | glutamine.tw. |
| 15. | exp Bacteriophages/ |
| 16. | bacteriophage$.tw. |
| 17. | alginate lyase.tw. |
| 18. | exp endolysin/ |
| 19. | endolysin$.tw. |
| 20. | (IgY or immunoglobulin Y).tw. |
| 21. | ((efflux pump or lectin) adj inhibitor$).tw. |
| 22. | 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21 |
| 23. | 6 and 22 |
| 24. | limit 23 to (humans and clinical trial, all) |
Data and analyses
Comparison 1. Chronic Infection: ß‐carotene supplementation.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Days of antibiotic consumption | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Over 1 month and up to 3 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Respiratory function (FEV1 % predicted) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Over 1 month and up to 3 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Mortality | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 Over 1 month and up to 3 months | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 2. Chronic Infection: Garlic supplementation.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Respiratory function (% change FEV1) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Over 1 month and up to 3 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Mortality | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2.1 Over 1 month and up to 3 months | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Mild adverse events | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 Diarrhoea | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.2 Halitosis | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.3 Abdominal pain | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.4 Dysuria | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3.5 Minor haemoptysis | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 3. Chronic infection: Zinc supplementation.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Respiratory function (FEV1 % predicted) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Up to 24 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Respiratory function (FVC %predicted) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Up to 24 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Antibiotic consumption (days oral antibiotics) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Up to 24 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Antibiotic consumption (days iv antibiotics) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 Up to 24 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Antibiotic consumption (days of oral and iv antibiotics) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 5.1 Up to 24 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 4. Chronic infection: KB001‐A.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Pulmonary exacerbations | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2 Decrease in respiratory function (FEV1) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3 Number of participants experiencing an adverse event | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4 Serious adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 Bronchitis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 Sinusitis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Ear and labyrinth adverse effects | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 5.1 Tinnitus | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Gastrointestinal adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 6.1 Abdominal pain upper | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.2 Cheilitis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.3 Pancreatitis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.4 Vomiting | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 General adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 Chest discomfort | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.2 Chills | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.3 Crepitations | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.4 Fatigue | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.5 Infusion‐related reaction | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.6 Infusion site discolouration | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.7 Infusion site rash | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.8 Irritability | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.9 Pyrexia (fever) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Infections and infestations | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 8.1 Sinusitis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Injury, poisoning and procedural complications | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 9.1 Procedural pain | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.2 Sunburn | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Investigative adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 10.1 Blood pressure increased | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10.2 Abnormal breath sounds | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10.3 Increased heart rate | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11 Nervous system adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 11.1 Dizziness | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11.2 Headache | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11.3 Sinus headache | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12 Psychiatric adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 12.1 Anxiety | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12.2 Depression | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12.3 Stress | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13 Respiratory, thoracic and mediastinal adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 13.1 Asthma | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.2 Cough | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.3 Dysphonia | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.4 Dyspnoea | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.5 Haemoptysis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.6 Nasal congestion | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.7 Nausea | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.8 Pharyngolaryngeal pain | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.9 Productive cough | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.10 Rales | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.11 Respiratory tract congestion | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.12 Rhinorrhoea | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.13 Sinus congestion | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.14 Throat irritation | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.15 Wheezing | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14 Skin and subcutaneous tissue adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 14.1 Oral pruritus | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.2 Pruritus | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.3 Urticaria | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 15 Vascular adverse events | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 15.1 Flushing | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Abdulhamid 2008.
| Methods | Double‐blind randomised placebo‐controlled study. Study based in Michigan, USA. |
|
| Participants | 26 children (aged 7 ‐ 18 years) with CF and mild to moderate lung disease without a concurrent acute severe infection were recruited. | |
| Interventions | Zinc gluconate supplementation (30 mg daily dose) for 12 months vs placebo. | |
| Outcomes | Outcomes not stratified. At each visit, the patient’s interval medical history, height and weight, number of hospitalizations, the use of oral and intravenous antibiotics, pulmonary function test, and physical examination findings were recorded. Primary outcome appears to be number of days of oral antibiotics during active infection per year. Secondary outcomes were production of inflammatory cytokines and IL‐2. |
|
| Notes | Dr Abdulhamid has kindly provided a per‐protocol analysis of the data with analysis limited to results of the treatment group compared to that of the placebo group. No specific comment on P. aeruginosa infection. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised but method not described. |
| Allocation concealment (selection bias) | Unclear risk | Concealment of allocation not described |
| Blinding (performance bias and detection bias) PUFA first | Unclear risk | Both groups (treatment and placebo) were given capsules although these were not described. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 1 child withdrew (no reason provided) and does not contribute to the data analysis |
| Selective reporting (reporting bias) | Low risk | In the published study, there is considerable selective reporting with the outcomes reported as per a subgroup analysis comparing those that were previously 'zinc adequate' or 'zinc inadequate'. The thresholds used to define zinc adequacy varied considerably to the 'clinically acceptable normal range'. Dr Abdulhamid has kindly provided the per‐protocol data with groups defined by their treatment group (placebo / zinc supplementation). After the provision of this additional information we therefore judge the risk of selective reporting to be low. |
| Other bias | Unclear risk | The compliance with treatment is reported as per the subgroup analysis and not as per protocol which further introduces uncertainty. |
Milla 2013.
| Methods | Randomised double blind placebo‐controlled study. Study based at multiple site in the USA. |
|
| Participants | 27 participants (aged over 12 years) with P. aeruginosa infection | |
| Interventions | Placebo, KB001 (3mg/kg), KB001 (10mg/kg) KB001 is a monoclonal antibody that works by inactivating the infection mechanism of P. aeruginosa | |
| Outcomes | Safety outcomes | |
| Notes | Available results posted on clinicaltrials.gov only. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as "randomized" but no detail provided. |
| Allocation concealment (selection bias) | Unclear risk | No detail provided. |
| Blinding (performance bias and detection bias) PUFA first | Unclear risk | No detail provided but described as double‐blind (subject, investigator, outcomes assessor). |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No detail provided. 27 enrolled, stated that no participants did not complete. |
| Selective reporting (reporting bias) | Low risk | Protocol deposited on clinicaltrials.gov. Primary outcome safety and tolerability which are reported. |
| Other bias | Unclear risk | None identified. |
Renner 2001.
| Methods | Randomised, double‐blind placebo controlled trial. Study based in Vienna, Austria. |
|
| Participants | 24 participants with CF from 1 unit in Vienna. 13 (9 female) randomised to supplementation (mean age 12.8 years; range 6.8 ‐ 27.7) and 11 (9 female) randomised to placebo (mean age 10.5 years; range 6.7 ‐ 17.3). | |
| Interventions | 3‐month 'high dose supplementation' of β‐carotene at 1 mg/kg/day followed by low dose regimen of 10mg/day for 3 months vs starch‐containing placebo. | |
| Outcomes | Primary outcomes ‐ normalised plasma concentration of β‐carotene; number of pulmonary exacerbations, lung function. | |
| Notes | No specific data regarding bacteria presented ‐ author contacted for more information 26 Jan 10. The control group was on average 2.3 years younger than the intervention group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised, but method of randomisation not described. |
| Allocation concealment (selection bias) | Unclear risk | Capsules were of identical appearance. Procedure for dispensing preparation unclear. |
| Blinding (performance bias and detection bias) PUFA first | Low risk | Described as 'double‐blind' but method not described. Capsules were of identical appearance. Rust 2000: Described as double blind but no further detail. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | There were no drop‐outs. Two publications for this trial appear to report separate outcome data. |
| Selective reporting (reporting bias) | Low risk | Selective reporting not identified other than the separate reporting of different outcome data. |
| Other bias | Unclear risk | Patient compliance not reported. |
Smyth 2010.
| Methods | Pilot double‐blinded, placebo‐controlled randomised controlled trial of garlic as a quorum sensing inhibitor. Study based in Nottingham, UK. |
|
| Participants | 34 participants over 8 years of age with CF (definition given) and chronic pulmonary infection with P. aeruginosa and at the time of randomisation. | |
| Interventions | 8 weeks treatment with garlic capsule once daily vs placebo. | |
| Outcomes | Primary outcome: FEV1. Secondary outcomes: weight, clinical score, quorum sensing signal molecule level. |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Web‐based randomisation sequence. |
| Allocation concealment (selection bias) | Low risk | Procedure for dispensing preparation unclear ‐ unpublished details demonstrate the supplements were dispensed in coded sealed opaque containers. |
| Blinding (performance bias and detection bias) PUFA first | Low risk | Double blind, identical placebo. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 8 participants withdrew ‐ four in each group: Garlic ‐ 1 received lung transplant, 1 forgot to take capsules, 1 side effect of indigestion, 1 could not attend second visit; Placebo ‐ 2 forgot to take capsules, 1 side effect of halitosis, 1 could not attend visit 2. Analysis was per protocol. |
| Selective reporting (reporting bias) | Low risk | No selective reporting identified. |
| Other bias | Low risk | No other bias identified. |
CF: cystic fibrosis FEV1: forced expiratory volume at one second P. aeruginosa: Pseudomonas aeruginosa vs: versus
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Brown 1985 | Randomised cross‐over study but authors have confirmed that data from first phase of study are unavailable. |
| Durairaj 2007 | Safety study therefore not blinded, randomised or controlled. |
| Gontijo‐Amaral 2012 | Adjuvant effect does not appear to have been demonstrated. |
| Grasemann 2013 | Adjuvant effect does not appear to have been demonstrated. |
| Hauber 2008 | Lacked a control group that did not receive examined intervention. |
| Homnick 1995 | Non‐randomised study. |
| Kollberg 2003 | Non‐randomised study, immunotherapy. |
| Kollberg 2010 | Immunotherapy not an antibiotic adjuvant therapy. |
| Kutateladze 2008 | Single patient experience described. |
| Lands 2010 | Non‐randomised study. |
| Lebecque 2008 | Study of miglustat ‐ not an antibiotic adjuvant therapy. |
| Leonard 2012 | Study of miglustat ‐ not an antibiotic adjuvant therapy. |
| Moss 2012 | Immunotherapy not an antibiotic adjuvant therapy. |
| Olveira 2010 | The fatty acid supplement exerts an anti‐inflammatory effect and does not meet the definition of antibiotic adjuvant. |
| Panchaud 2006 | Independent anti‐inflammatory effect ‐ does not meet the definition of antibiotic adjuvant. |
| Safai‐Kutti 1991 | Non‐randomised study. |
| Sagel 2011 | Non‐randomised, open‐label study. |
| Schuster 2011 | Immunotherapy not an antibiotic adjuvant therapy. |
| Winklhofer‐Roob 1995 | Non‐randomised, healthy controls. |
| Winnie 1989 | Immunotherapy not an antibiotic adjuvant therapy. |
Characteristics of ongoing studies [ordered by study ID]
Molfino 2012.
| Trial name or title | Study to evaluate the effect of KB001‐A on time‐to‐need for antibiotic treatment. |
| Methods | A phase 2, randomized, double‐blind, placebo‐controlled, repeat‐dose study of KB001‐A in subjects with cystic fibrosis infected with Pseudomonas aeruginosa Parallel assignment. |
| Participants | 180 participants aged 12 to 80 years of age. Inclusion criteria:
Exclusion criteria:
|
| Interventions | 5 intravenous infusions of KB001‐A (a biological agent) at 10mg/kg versus placebo. |
| Outcomes | Primary outcome: time to requirement of antibiotics due to worsening respiratory tract signs and symptoms. Secondary outcome: adverse effects. |
| Starting date | December 2012. |
| Contact information | jpelham@kalobios.com |
| Notes | Primary completion date: November 2013. |
Walshaw 2011.
| Trial name or title | A double‐blind, randomized, placebo‐controlled, cross‐over study to evaluate the safety, tolerability and preliminary efficacy of alginate oligosaccharide (OligoG) administered for 28 days in subjects with cystic fibrosis chronically colonised with Pseudomonas aeruginosa. |
| Methods | Double‐blind (participant, caregiver, investigator, outcomes assessor), randomized, placebo‐controlled, cross‐over study. |
| Participants | 26 participants 18 years and older. Inclusion criteria:
Exclusion criteria:
|
| Interventions | Inhaled 6% OligoG versus 0.9% saline. |
| Outcomes | Primary outcome: safety and local tolerability of multiple dose administration of inhaled OligoG in people with CF. Secondary outcome: effect of multiple dose administration of inhaled OligoG on various efficacy variables. |
| Starting date | May 2011. |
| Contact information | alastair.woods@smerud.com |
| Notes | Primary completion date: July 2012. |
Zabner 2009.
| Trial name or title | Aerosolized hypertonic xylitol versus hypertonic saline in cystic fibrosis (CF) subjects. |
| Methods | Randomized, double‐blind (participant, caregiver, investigator), parallel assignment, safety/efficacy study. |
| Participants | CF patients, 16 years or older. |
| Interventions | Hypertonic xylitol versus hypertonic saline. |
| Outcomes | Primary outcome measures: FEV1 change from baseline, adverse events and respiratory symptom score. [Time Frame: 14 days]. Secondary outcome measures: density of colonization per gram of sputum, time to next exacerbation, sputum cytokines and revised CF quality of life questionnaire. [time frame: 14 days]. |
| Starting date | June 2009. |
| Contact information | joseph‐zabner@uiowa.edu. |
| Notes | Primary completion date: July 2013. |
ABPA: Allergic bronchopulmonary aspergillosis ABX: antibiotic treatment ATS: American Thoracic Society B. cepacia: Burkholderia cepacia CF: cystic fibrosis DNase: dornase alfa ERS: European Respiratory Society FEV1: forced expiratory volume at one second IgY: avian polyclonal antibodies for P. aeruginosa NPD: nasal potential difference NSAIDs: non‐steroidal anti‐inflammatory drugs P. aeruginosa: Pseudomonas aeruginosa TCS: total chloride secretion
Differences between protocol and review
We altered the title of the review to be more informative ‐ changing 'non‐antibiotic' terminology to a new term ‐ antibiotic adjuvants.
We have altered the definition of chronic infection to a definition that is most commonly used in routine clinical practice (Lee 2004) and have added requirement of antibiotics (days) as a secondary outcome measure.
Contributions of authors
MH, DF and AS conceived the title. MH wrote the protocol, completed a search of the literature, was an independent assessor of the studies identified and drafted the review. DF was the second independent assessor of the studies identified from the literature search and commented upon the drafts of the protocol and the substantive review. AS supervised, gave comments and advised on the protocol and review.
MH acts as guarantor of the review.
Sources of support
Internal sources
-
Nottingham Respiratory Biomedical Research Unit, UK.
MH and DF are funded by the Nottingham Respiratory BRU
External sources
No sources of support supplied
Declarations of interest
Dr Matthew Hurley Dr Hurley has received support from the Wellcome Trust as a Wellcome Turst Clinical Research Training Fellow.
Professor Alan Smyth Professor Smyth is lead investigator on one of the studies included in the review (Smyth 2010).
He has also acted as a consultant to the pharmaceutical industry for therapies in development for pulmonary infection with Pseudomonas aeruginosa in cystic fibrosis (CF):
Novartis ‐ dry powder inhaled tobramycin
Mpex Pharmaceuticals ‐ inhaled, nebulised levofloxacin
Biocontrol ‐ bacteriophage therapy
Professor Smyth has also received payment for commercial clinical trials:
Boehringer Ingelheim, Ltd. (UK) ‐ inhaled tiotropium as a long‐acting bronchodilator in CF.
Edited (no change to conclusions)
References
References to studies included in this review
Abdulhamid 2008 {published and unpublished data}
- Abdulhamid I, Beck FW, Millard S, Chen X, Prasad A. Effect of zinc supplementation on respiratory tract infections in children with cystic fibrosis. Pediatric Pulmonology 2008;43:281‐7. [DOI: 10.1002/ppul.20771] [DOI] [PubMed] [Google Scholar]
Milla 2013 {unpublished data only}
- Milla C, Molfino NA for KaloBios Pharmaceuticals. A phase I/II randomized, double‐blind, placebo‐controlled, single‐dose, dose escalation study of KB001 in cystic fibrosis patients infected with Pseudomonas aeruginosa. www.clinicaltrials.gov/ct2/show/study/NCT00638365?term=NCT00638365&rank=1 (accessed 21 March 2013).
Renner 2001 {published data only}
- Engl B, Rust P, Eichler I, Renner S, Elmadfa I. Bioavailability of therapeutic Beta‐Carotine (BC) in patients with cystic fibrosis (CF) and effects on anthropometrical parameters over 6 months [abstract]. Monatsschrift fur Kinderheilkunde 1997;145:S 134. [Google Scholar]
- Renner S, Rath R, Rust P, Lehr S, Frischer T, Elmadfa I, et al. Effects of beta‐carotene supplementation for six months on clinical and laboratory parameters in patients with cystic fibrosis. Thorax 2001;56(1):48‐52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renner S, Wojnarowski C, Koller DY, Rust P, Elmadfa I, Eichler I. Patients with cystic fibrosis (CF) benefit from ß‐Carotene supplementation for 6 months [abstract]. Pediatric Pulmonology 1997;24(Suppl 15):314. [Google Scholar]
- Renner S, Wojnarowski C, Koller DY, Rust P, Elmadfa I, Eichler I. Patients with cystic fibrosis (CF) benefit from ß‐Carotene supplementation for 6 months [abstract]. Proceedings of: 22nd European Cystic Fibrosis Conference; 1998 June 13‐19; Berlin, Germany. 1998:97.
- Rust P, Eichler I, Elmadfa I. Influence of an oral beta‐carotene supplementation on the antioxidant status of patients with cystic fibrosis [abstract]. Atemwegs und Lungenkrankheiten 1997;23:51. [Google Scholar]
- Rust P, Eichler I, Renner S, Elmadfa I. Effects of long‐term oral beta‐carotene supplementation on lipid peroxidation in patients with cystic fibrosis. International Journal for Vitamin and Nutrition Research 1998;68(2):83‐7. [PubMed] [Google Scholar]
- Rust P, Eichler I, Renner S, Elmadfa I. Long term beta‐carotene supplementation in patients with cystic fibrosis ‐ effects on oxidative status and pulmonary function.. Ann Nutr Metab 2000;44(1):30‐7. [PUBMED: 10838464] [DOI] [PubMed] [Google Scholar]
Smyth 2010 {published data only}
- Smyth A, Cifelli P, Lewis S, Erskine P, Holland E, Willaims P, et al. Quorum sensing molecules in sputum and plasma as biomarkers in patients with CF and chronic pseudomonas aeruginosa [abstract]. Pediatric Pulmonology 2008;43(Suppl 31):337. [Google Scholar]
- Smyth A, Cifelli P, Lewis S, Erskine P, Holland E, Williams P, et al. A randomized controlled trial of macerated garlic oil in patients with CF and chronic pseudomonas aeruginosa [abstract]. Pediatric Pulmonology 2008;43(Suppl 31):336. [Google Scholar]
- Smyth AR, Cifelli PM, Ortori CA, Righetti K, Lewis S, Erskine P, et al. Garlic as an inhibitor of Pseudomonas aeruginosa quorum sensing in cystic fibrosis – a pilot randomised controlled trial. Pediatric Pulmonology 2010;45(4):356‐62. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Brown 1985 {published data only}
- Brown J, Mellis CM, Wood RE. EDTA aerosol in pseudomonal lung infection [abstract]. Proceedings of 9th International Cystic Fibrosis Congress; 1984 June 9‐15; Brighton, England. 1984:4.10.
- Brown J, Mellis CM, Wood RE. Edetate sodium aerosol in Pseudomonas lung infection in cystic fibrosis. American Journal of Diseases of Children 1985;139(8):836‐9. [DOI] [PubMed] [Google Scholar]
Durairaj 2007 {published data only}
- Durairaj L, Launspach J, Watt JL, Mohamad Z, Kline J, Zabner J. Safety assessment of inhaled xylitol in subjects with cystic fibrosis. Journal of Cystic Fibrosis 2007 Jan;6(1):31‐4. [DOI] [PubMed] [Google Scholar]
Gontijo‐Amaral 2012 {published data only}
- Gontijo‐Amaral C, Guimaraes EV, Camargos P. Oral magnesium supplementation in children with cystic fibrosis improves clinical and functional variables: a double‐blind,randomized, placebo‐controlled crossover trial. American Journal of Clinical Nutrition 2012;96(1):50‐6. [DOI: 10.3945/ajcn.112.034207] [DOI] [PubMed] [Google Scholar]
Grasemann 2013 {published data only}
- Grasemann H, Tullis E, Ratjen F. A randomized controlled trial of inhaled L‐Arginine in patients with cystic fibrosis. Journal of Cystic Fibrosis 2013;pii: S1569‐1993(12):00241‐X. [DOI: 10.1016/j.jcf.2012.12.008] [DOI] [PubMed] [Google Scholar]
Hauber 2008 {published data only}
- Hauber H, Schulz M, Pforte A, Mack D, Zabel P, Schumacher U. Inhalation with fucose and galactose for treatment of Pseudomonas aeruginosa in cystic fibrosis patients. International Journal of Medical Sciences 2008;5(6):371‐6. [PUBMED: PMC2586681] [DOI] [PMC free article] [PubMed] [Google Scholar]
Homnick 1995 {published data only}
- Homnick DN, Spillers CR, Cox SR, Cox JH, Yelton LA, DeLoof MJ, et al. Single‐ and multiple‐dose‐response relationships of beta‐carotene in cystic fibrosis.[see comment]. Journal of Pediatrics 1995 Sep;127(3):491‐4. [DOI] [PubMed] [Google Scholar]
Kollberg 2003 {published data only}
- Kollberg H, Carlander D, Olesen H, Wejaker PE, Johannesson M, Larsson A. Oral administration of specific yolk antibodies (IgY) may prevent Pseudomonas aeruginosa infections in patients with cystic fibrosis: a phase I feasibility study. Pediatric Pulmonology 2003 Jun;35(6):433‐40. [DOI] [PubMed] [Google Scholar]
- Nilsson E, Kollberg H, Johannesson M, Wejaker PE, Carlander D, Larsson A. More than 10 years' continuous oral treatment with specific immunoglobulin Y for the prevention of Pseudomonas aeruginosa infections: a case report. Journal of Medicinal Food 2007;10(2):375‐8. [DOI] [PubMed] [Google Scholar]
- Nilsson E, Larsson A, Olesen HV, Wejaker PE, Kollberg H. Good effect of IgY against Pseudomonas aeruginosa infections in cystic fibrosis patients. Pediatric Pulmonology 2008;43(9):892‐9. [DOI] [PubMed] [Google Scholar]
Kollberg 2010 {published data only}
- Kollberg H, Larsson A, Nilsson E. Anti‐pseudomonas IGY ready for phase III [abstract]. Pediatric Pulmonology 2010;45(Suppl 33):343, Abstract no: 345. [CFGD Register: PI252a] [Google Scholar]
- Larsson A. Phase III study (IMPACTT) on anti‐pseudomonas IgY [abstract]. Journal of Cystic Fibrosis 2011;10(Suppl 1):S24, Abstract no: 92. [CFGD Register: PI252b] [Google Scholar]
Kutateladze 2008 {published data only}
- Kutateladze M, Adamia R. Phage therapy experience at the Eliava Institute. Medecine Et Maladies Infectieuses 2008;38(8):426‐30. [DOI] [PubMed] [Google Scholar]
Lands 2010 {published data only}
- Lands LC. Iskandar M. Beaudoin N. Meehan B. Dauletbaev N. Berthiuame Y. Dietary supplementation with pressurized whey in patients with cystic fibrosis. Journal of Medicinal Food 2010;13(1):77‐82. [DOI: 10.1089/jmf.2008.0326] [DOI] [PubMed] [Google Scholar]
Lebecque 2008 {unpublished data only}
- Lebecque P. Single center, double‐blind, randomized, placebo‐controlled, two‐period/two‐treatment crossover study investigating the effect of miglustat on the nasal potential difference in patients with cystic fibrosis homozygous for the F508del mutation. clinicaltrials.gov/ct2/show/NCT00742092?term=NCT00742092&rank=1 (accessed 21 March 2013).
Leonard 2012 {published data only}
- Leonard A, Lebecque P, Dingemanse J, Leal T. A randomized placebo‐controlled trial of miglustat in cystic fibrosis based on nasal potential difference. Journal of Cystic Fibrosis 2012;11(3):231‐6. [DOI: 10.1016/j.jcf.2011.12.004] [DOI] [PubMed] [Google Scholar]
Moss 2012 {published data only}
- Moss RB, Mistry SJ, Konstan MW, Pilewski JM, Kerem E, Tal‐Singer R, et al. Safety and early treatment effects of the CXCR2 antagonist SB‐656933 inpatients with cystic fibrosis. Journal of Cystic Fibrosis 2012;pii: S1569‐1993(12):00155‐5. [DOI: 10.1016/j.jcf.2012.08.016] [DOI] [PubMed] [Google Scholar]
Olveira 2010 {published data only}
- Olveira G, Olveira C, Acosta E, Espíldora F, Garrido‐Sánchez L, García‐Escobar E. Fatty acid supplements improve respiratory, inflammatory and nutritional parameters in adults with cystic fibrosis. Archivos de Bronconeumologia 2010;46(2):70‐7. [DOI] [PubMed] [Google Scholar]
Panchaud 2006 {published data only}
- Panchaud A, Sauty A, Kernen Y, Decosterd LA, Buclin T, Boulat O, et al. Biological effects of a dietary omega‐3 polyunsaturated fatty acids supplementation in cystic fibrosis patients: a randomized, crossover placebo‐controlled trial. Clinical Nutrition 2006; Vol. 25, issue 3:418‐27. [DOI: 10.1016/j.clnu.2005.10.011] [DOI] [PubMed]
Safai‐Kutti 1991 {published data only}
- Safai‐Kutti S, Selin E, Larsson S, Jagenburg R, Sten G, Kjellmer I. Zinc therapy in children with cystic fibrosis. Beitraege zur Infusionstherapie 1991;27:104‐14. [PubMed] [Google Scholar]
Sagel 2011 {published data only}
- Sagel SD, Sontag MK, Anthony MM, Emmett P, Paps KA. Effect of an antioxidant‐rich multivitamin supplement in cystic fibrosis. Journal of Cystic Fibrosis 2011;10(1):31‐6. [DOI: 10.1016/j.jcf.2010.09.005] [DOI] [PubMed] [Google Scholar]
Schuster 2011 {published data only}
- Schuster A. Phase III study to evaluate clinical efficacy and safety of avian polyclonal anti‐Pseudomonas antibodies (IgY) in prevention of recurrence of Pseudomonas aeruginosa infection in cystic fibrosis patients. http://clinicaltrials.gov/ct2/show/NCT01455675?term=NCT01455675&rank=1 (accessed 21 March 2013).
Winklhofer‐Roob 1995 {published data only}
- Winklhofer‐Roob BM, Puhl H, Khoschsorur G, van't Hof MA, Esterbauer H, Shmerling DH. Enhanced resistance to oxidation of low density lipoproteins and decreased lipid peroxide formation during beta‐carotene supplementation in cystic fibrosis. Free Radical Biology & Medicine 1995;18(5):849‐59. [DOI] [PubMed] [Google Scholar]
- Winklhofer‐Roob BM, Schlegel‐Haueter SE, Khoschsorur G, van't Hof MA, Suter S, Shmerling DH. Neutrophil elastase/alpha 1‐proteinase inhibitor complex levels decrease in plasma of cystic fibrosis patients during long‐term oral beta‐carotene supplementation. Pediatric Research 1996;40(1):130‐4. [DOI] [PubMed] [Google Scholar]
- Winklhofer‐Roob BM, van't Hof MA, Shmerling DH. Response to oral beta‐carotene supplementation in patients with cystic fibrosis: a 16‐month follow‐up study.[erratum appears in Acta Paediatr 1996 Jan;85(1):124]. Acta Paediatrica 1995 Oct;84(10):1132‐6. [DOI] [PubMed] [Google Scholar]
Winnie 1989 {published data only}
- Winnie GB, Cowan RG, Wade NA. Intravenous immune globulin treatment of pulmonary exacerbations in cystic fibrosis. The Journal of Pediatrics 1989;114(2):309‐14. [DOI: 10.1016/S0022-3476(89)80804-2] [DOI] [PubMed] [Google Scholar]
References to ongoing studies
Molfino 2012 {unpublished data only}
- Study to evaluate the effect of KB001‐A on time‐to‐need for antibiotic treatment.. Ongoing study December 2012..
Walshaw 2011 {unpublished data only}
- A double‐blind, randomized, placebo‐controlled, cross‐over study to evaluate the safety, tolerability and preliminary efficacy of alginate oligosaccharide (OligoG) administered for 28 days in subjects with cystic fibrosis chronically colonised with Pseudomonas aeruginosa.. Ongoing study May 2011..
Zabner 2009 {unpublished data only}
- Aerosolized hypertonic xylitol versus hypertonic saline in cystic fibrosis (CF) subjects.. Ongoing study June 2009..
Additional references
Borysowski 2006
- Borysowski J, Weber‐Dbrowska B, Górski A. Bacteriophage endolysins as a novel class of antibacterial agents. Experimental Biology and Medicine 2006;231(4):366‐77. [PUBMED: 16565432] [DOI] [PubMed] [Google Scholar]
Dodge 2007
- Dodge JA, Lewis PA, Stanton M, Wilsher J. Cystic fibrosis mortality and survival in the UK: 1947–2003. European Respiratory Journal 2007;29(3):522‐6. [DOI: 10.1183/09031936.00099506] [DOI] [PubMed] [Google Scholar]
Druesne‐Pecollo 2010
- Druesne‐Pecollo N, Latino‐Martel P, Norat T, Barrandon E, Bertrais S, Galan P, et al. Beta‐carotene supplementation and cancer risk: a systematic review and meta‐analysis of randomized controlled trials. International Journal of Cancer 2010;127(1):172‐84. [DOI: 10.1002/ijc.25008] [DOI] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327(7414):557‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Johansson 2008
- Johansson EM, Crusz SA, Kolomiets E, Buts L, Kadam RU, Cacciarini M, et al. Inhibition and dispersion of Pseudomonas aeruginosa biofilms by glycopeptide dendrimers targeting the fucose‐specific lectin LecB. Chemistry and Biology 2008;15(12):1249‐57. [DOI: 10.1016/j.chembiol.2008.10.009] [DOI] [PubMed] [Google Scholar]
Langton‐Hewer 2009
- Langton‐Hewer SC, Smyth AR. Antibiotic strategies for eradicating Pseudomonas aeruginosa in people with cystic fibrosis. Cochrane Database of Systematic Reviews 2009, Issue 4. [DOI: 10.1002/14651858.CD004197.pub3] [DOI] [PubMed] [Google Scholar]
Lee 2004
- Lee TWR, Brownlee KG, Denton M, Littlewood J, Conway SP. Reduction in prevalence of chronic Pseudomonas aeruginosa infection at a regional pediatric cystic fibrosis center. Pediatric Pulmonology 2004;37(2):104‐10. [DOI: 10.1002/ppul.10401] [DOI] [PubMed] [Google Scholar]
RevMan 2011 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.1. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2011.
Rosenfeld 2001
- Rosenfeld M, Emerson J, Williams‐Warren J, Pepe M, Smith A, Montgomery B, et al. Defining a pulmonary exacerbation in cystic fibrosis. Journal of Pediatrics 2001;139(3):359‐65. [DOI: 10.1067/mpd.2001.117288] [DOI] [PubMed] [Google Scholar]
Rosenstein 1998
- Rosenstein BJ, Cutting GP for the Cystic Fibrosis Foundation consensus panel. The diagnosis of cystic fibrosis: A consensus statement. Journal of Pediatrics 1998;132(4):589‐95. [DOI: 10.1016/S0022-3476(98)70344-0] [DOI] [PubMed] [Google Scholar]
UK Cystic Fibrosis Trust 2004
- UK Cystic Fibrosis Trust Infection Control Group. Pseudomonas aeruginosa infection in people with cystic fibrosis: Suggestions for prevention and infection control. Report of the UK Cystic Fibrosis Infection Control Group 2004. [ISBN: 0‐9540536‐9‐9]
UK Cystic Fibrosis Trust Antibiotic Working Group
- UK Cystic FIbrosis Antibiotic Working Group. Antibiotic Treatment for Cystic Fibrosis ‐ 3rd Edition. Cystic Fibrosis Trust 2009.
von Bodman 2008
- Bodman SB, Willey JM, Diggle SP. Cell‐Cell Communication in Bacteria: United We Stand. Journal of Bacteriology 2008;190(13):4377‐91. [DOI: 10.1128/JB.00486-08] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Hurley 2010
- Hurley MN, Forrester DL, Smyth AR. Antibiotic adjuvant therapy for pulmonary infection in cystic fibrosis. Cochrane Database of Systematic Reviews 2010, Issue 10. [DOI: 10.1002/14651858.CD008037.pub2] [DOI] [PubMed] [Google Scholar]


