Abstract
Background
Electromechanical‐ and robotic‐assisted gait‐training devices are used in rehabilitation and might help to improve walking after stroke. This is an update of a Cochrane Review first published in 2007.
Objectives
To investigate the effects of automated electromechanical‐ and robotic‐assisted gait‐training devices for improving walking after stroke.
Search methods
We searched the Cochrane Stroke Group Trials Register (last searched 9 August 2016), the Cochrane Central Register of Controlled Trials (CENTRAL) (the Cochrane Library 2016, Issue 8), MEDLINE in Ovid (1950 to 15 August 2016), Embase (1980 to 15 August 2016), CINAHL (1982 to 15 August 2016), AMED (1985 to 15 August 2016), Web of Science (1899 to 16 August 2016), SPORTDiscus (1949 to 15 September 2012), the Physiotherapy Evidence Database (PEDro) (searched 16 August 2016), and the engineering databases COMPENDEX (1972 to 16 November 2012) and Inspec (1969 to 26 August 2016). We handsearched relevant conference proceedings, searched trials and research registers, checked reference lists, and contacted authors in an effort to identify further published, unpublished, and ongoing trials.
Selection criteria
We included all randomised controlled trials and randomised controlled cross‐over trials in people over the age of 18 years diagnosed with stroke of any severity, at any stage, in any setting, evaluating electromechanical‐ and robotic‐assisted gait training versus normal care.
Data collection and analysis
Two review authors independently selected trials for inclusion, assessed methodological quality and risk of bias, and extracted the data. The primary outcome was the proportion of participants walking independently at follow‐up.
Main results
We included 36 trials involving 1472 participants in this review update. Electromechanical‐assisted gait training in combination with physiotherapy increased the odds of participants becoming independent in walking (odds ratio (random effects) 1.94, 95% confidence interval (CI) 1.39 to 2.71; P < 0.001; I² = 8%; moderate‐quality evidence) but did not significantly increase walking velocity (mean difference (MD) 0.04 m/s, 95% CI 0.00 to 0.09; P = 0.08; I² = 65%; low‐quality evidence) or walking capacity (MD 5.84 metres walked in 6 minutes, 95% CI ‐16.73 to 28.40; P = 0.61; I² = 53%; very low‐quality evidence). The results must be interpreted with caution because 1) some trials investigated people who were independent in walking at the start of the study, 2) we found variations between the trials with respect to devices used and duration and frequency of treatment, and 3) some trials included devices with functional electrical stimulation. Our planned subgroup analysis suggested that people in the acute phase may benefit, but people in the chronic phase may not benefit from electromechanical‐assisted gait training. Post hoc analysis showed that people who are non‐ambulatory at intervention onset may benefit, but ambulatory people may not benefit from this type of training. Post hoc analysis showed no differences between the types of devices used in studies regarding ability to walk, but significant differences were found between devices in terms of walking velocity.
Authors' conclusions
People who receive electromechanical‐assisted gait training in combination with physiotherapy after stroke are more likely to achieve independent walking than people who receive gait training without these devices. We concluded that seven patients need to be treated to prevent one dependency in walking. Specifically, people in the first three months after stroke and those who are not able to walk seem to benefit most from this type of intervention. The role of the type of device is still not clear. Further research should consist of large definitive pragmatic phase III trials undertaken to address specific questions about the most effective frequency and duration of electromechanical‐assisted gait training as well as how long any benefit may last.
Plain language summary
Automated training devices for improving walking after stroke
Review question
Do machine‐ and robot‐assisted walking training devices improve walking after stroke?
Background
Many people who have had a stroke have difficulties walking, and improving walking is one of the main goals of rehabilitation. Automated training devices assist walking practice.
Search date
The review is current to August 2016.
Study characteristics
We included 36 studies involving a total of 1472 participants over the age of 18 years with acute, postacute, or chronic ischaemic or haemorrhagic stroke. The mean age in the included studies ranged from 48 years to 76 years. The majority of studies were conducted in an inpatient setting.
Key results
We found moderate‐quality evidence that electromechanical‐assisted gait training combined with physiotherapy when compared with physiotherapy alone may improve recovery of independent walking in people after stroke.
We determined that for every seven patients treated with electromechanical‐ and robotic‐assisted gait training devices, just one prevention of dependency in walking occurs.
Specifically, people in the first three months after stroke and those who are not able to walk appear to benefit most from this type of intervention. The importance of the type of device is still not clear. Further research should address what frequency or duration of walking training might be most effective and how long the benefit lasts. It also remains unclear how such devices should be used in routine rehabilitation.
Quality of the evidence
The quality of the evidence for automated electromechanical‐ and robotic‐assisted gait‐training devices for improving walking after stroke was moderate. The quality of evidence was low for walking speed, very low for walking capacity, and low for adverse events and people discontinuing treatment.
Summary of findings
Summary of findings for the main comparison. Electromechanical‐ and robotic‐assisted gait training plus physiotherapy compared to physiotherapy (or usual care) for walking after stroke.
| Electromechanical‐ and robotic‐assisted gait training plus physiotherapy compared to physiotherapy (or usual care) for walking after stroke | ||||||
| Patient or population: walking after stroke Setting: inpatient and outpatient setting Intervention: electromechanical‐ and robotic‐assisted gait training plus physiotherapy Comparison: physiotherapy (or usual care) | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with physiotherapy (or usual care) | Risk with electromechanical‐ and robotic‐assisted gait training plus physiotherapy | |||||
| Independent walking at the end of intervention phase, all electromechanical devices used Assessed with FAC | Study population | OR 1.94 (1.39 to 2.71) | 1472 (36 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | ||
| 457 per 1000 | 615 per 1000 (530 to 693) | |||||
| Recovery of independent walking at follow‐up after study end Assessed with FAC | Study population | OR 1.93 (0.72 to 5.13) | 496 (6 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | ||
| 551 per 1000 | 703 per 1000 (469 to 863) | |||||
| Walking velocity (metres per second) at the end of intervention phase Assessed with timed measures of gait Scale: 0 to infinity | The mean walking velocity (metres per second) at the end of intervention phase was 0. | MD 0.04 higher (0 to 0.09 higher) | ‐ | 985 (24 RCTs) | ⊕⊕⊝⊝ LOW 1 2 | |
| Walking velocity (metres per second) at follow‐up Assessed with timed measures of gait Scale: 0 to infinity | The mean walking velocity (metres per second) at follow‐up was 0. | MD 0.07 higher (0.05 lower to 0.19 higher) | ‐ | 578 (9 RCTs) | ⊕⊕⊕⊝ MODERATE 1 | |
| Walking capacity (metres walked in 6 minutes) at the end of intervention phase Assessed with timed measures of gait Scale: 0 to infinity | The mean walking capacity (metres walked in 6 minutes) at the end of intervention phase was 0. | MD 5.84 higher (16.73 lower to 28.40 higher) | ‐ | 594 (12 RCTs) | ⊕⊝⊝⊝ VERY LOW 1 3 4 | |
| Walking capacity (metres walked in 6 minutes) at follow‐up | The mean walking capacity (metres walked in 6 minutes) at follow‐up was 0. | MD 0.82 lower (32.17 lower to 30.53 higher) | ‐ | 463 (7 RCTs) | ⊕⊝⊝⊝ VERY LOW 1 2 4 | |
| Acceptability of electromechanical‐assisted gait‐training devices during intervention phase Assessed with number of dropouts | Study population | OR 0.67 (0.43 to 1.05) | 1472 (36 RCTs) | ⊕⊕⊝⊝ LOW 1 5 | ||
| 131 per 1000 | 92 per 1000 (61 to 136) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FAC: Functional Ambulation Category; MD: mean difference; OR: odds ratio; RCT: randomised controlled trial; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect. Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect. Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect. | ||||||
1Downgraded due to several ratings of 'unclear' and 'high' risk of bias. 2Downgraded due to statistical heterogeneity and no overlap of several confidence intervals. 3Downgraded because the 95% confidence interval includes no effect and the upper confidence limit crosses the minimal important difference. 4Downgraded due to funnel plot asymmetry. 5Downgraded because the total number of events (157) is less than 300 (a threshold rule‐of‐thumb value).
Background
Description of the condition
A stroke is a sudden, non‐convulsive loss of neurological function due to an ischaemic or haemorrhagic intracranial vascular event (WHO 2006). In general, cerebrovascular accidents are classified by anatomic location in the brain, vascular distribution, aetiology, age of the affected individual, and haemorrhagic versus non‐haemorrhagic nature (Adams 1993). Stroke is a leading cause of death and serious long‐term disability in adults. Three months after stroke, 20% of people remain wheelchair bound, and approximately 70% walk at a reduced velocity and capacity (Jorgensen 1995). Restoration of walking ability and gait rehabilitation are therefore highly relevant for people who are unable to walk independently after stroke (Bohannon 1991), as well as for their relatives. To restore gait, modern concepts of rehabilitation favour a repetitive task‐specific approach (Carr 2003; French 2007). In recent years it has also been shown that higher intensities of walking practice (resulting in more repetitions trained) resulted in better outcomes for people after stroke (Kwakkel 1999; Van Peppen 2004).
Description of the intervention
As an adjunct to overground gait training (States 2009), in recent years treadmill training has been introduced for the rehabilitation of people after stroke (Mehrholz 2014). Treadmill training with and without partial body weight support enables the repetitive practice of complex gait cycles for these people. However, one disadvantage of treadmill training might be the effort required by therapists to set the paretic limbs and to control weight shift, thereby possibly limiting the intensity of therapy, especially in more severely disabled people. Automated electromechanical gait machines were developed to reduce dependence on therapists. They consist of either a robot‐driven exoskeleton orthosis or an electromechanical solution, with two driven foot plates simulating the phases of gait (Colombo 2000; Hesse 1999).
One example of automated electromechanical gait rehabilitation is the Lokomat (Colombo 2000). A robotic gait orthosis combined with a harness‐supported body weight system is used together with a treadmill. The main difference from treadmill training is that the patient's legs are guided by the robotic device according to a preprogrammed gait pattern. A computer‐controlled robotic gait orthosis guides the patient, and the process of gait training is automated.
A second example is the Gait Trainer GT I, which is based on a double crank and rocker gear system (Hesse 1999). In contrast to a treadmill, the electromechanical Gait Trainer GT I consists of two foot plates positioned on two bars, two rockers, and two cranks, which provide the propulsion. The harness‐secured patient is positioned on the foot plates, which symmetrically simulate the stance and swing phases of walking (Hesse 1999). A servo‐controlled motor guides the patient during walking exercise. Vertical and horizontal movements of the trunk are controlled in a phase‐dependent manner. Again, the main difference from treadmill training is that the process of gait training is automated and is supported by an electromechanical solution.
Other similar electromechanical devices that have been developed in recent years include the Haptic Walker (Schmidt 2005), the Anklebot (MIT 2005), and the LOPES (Lower Extremity Powered Exoskeleton) (Veneman 2005). More recently, new so‐called powered mobile solutions, Buesing 2015, Stein 2014, Watanabe 2014, and ankle robots, Forrester 2014, Waldman 2013, to improve walking have been described in the literature.
How the intervention might work
Electromechanical devices (such as those previously described) can be used to give non‐ambulatory patients intensive practice (in terms of high repetitions) of complex gait cycles. The advantage of these electromechanical devices compared with treadmill training with partial body weight support may be the reduced effort required of therapists, as they no longer need to set the paretic limbs or assist trunk movements (Hesse 2003).
Why it is important to do this review
Scientific evidence for the benefits of the above‐mentioned technologies may have changed since our Cochrane Review was first published in 2007 (Mehrholz 2007), and so an update of the review was required to justify the large equipment and human resource costs needed to implement electromechanical‐assisted gait devices, as well as to confirm the safety and acceptance of this method of training. The aim of this review was therefore to provide an update of the best available evidence about the above‐mentioned approach.
Objectives
To investigate the effects of automated electromechanical‐ and robotic‐assisted gait‐training devices for improving walking after stroke.
Methods
Criteria for considering studies for this review
Types of studies
We searched for all randomised controlled trials and randomised controlled cross‐over trials for inclusion in this review. If we included randomised controlled cross‐over trials, we planned to analyse only the first period as a parallel‐group trial.
Types of participants
We included studies with participants of any gender over 18 years of age after stroke, using the World Health Organization (WHO) definition of stroke or a clinical definition of stroke if the WHO definition was not specifically stated (WHO 2006).
Types of interventions
We included all trials that evaluated electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care) for regaining and improving walking after stroke. We also included automated electromechanical devices that were used in combination with therapies such as functional electrical stimulation applied to the legs during gait training (compared with therapies not using electromechanical devices). We defined an automated electromechanical device as any device with an electromechanical solution designed to assist stepping cycles by supporting body weight and automating the walking therapy process in people after stroke. This category included any mechanical or computerised device designed to improve walking function. We also searched for electromechanical devices such as robots for gait training after stroke (MIT 2005; Schmidt 2005; Veneman 2005).
Electromechanical devices can principally be differentiated into end‐effector and exoskeleton devices. Examples of end‐effector devices are the LokoHelp (Freivogel 2009), the Haptic Walker (Schmidt 2005), and the Gait Trainer GT I (Hesse 1999). The definition of an end‐effector principle is that a patient's feet are placed on foot plates, whose trajectories simulate the stance and swing phases during gait training (Hesse 2010). An example of exoskeleton devices is the Lokomat (Colombo 2000). Such exoskeletons are outfitted with programmable drives or passive elements, which move the knees and hips during the phases of gait (Hesse 2010).
We did not include non‐weight‐bearing interventions such as non‐interactive devices that deliver continuous passive motion only (Nuyens 2002). We excluded trials testing the effectiveness of treadmill training or other approaches such as repetitive task training in physiotherapy or electrical stimulation alone (French 2016; Pollock 2014), to prevent duplication with other Cochrane Reviews and protocols (e.g. Mehrholz 2014).
Types of outcome measures
Primary outcomes
Regaining the ability to walk is a very important goal for people after stroke (Bohannon 1988). We therefore defined the primary outcome as the ability to walk independently. We measured the ability to walk with the Functional Ambulation Category (FAC) (Holden 1984). A FAC score of 4 or 5 indicated independent walking over a 15‐metre surface, irrespective of aids used such as a cane. A FAC score of less than 4 indicates dependency in walking (supervision or assistance, or both must be given in performing walking).
If the included studies did not report FAC scores, we used alternative indicators of independent walking, such as:
a score of 3 on the ambulation item of the Barthel Index (Wade 1988); or
a score of 6 or 7 for the walking item of the Functional Independence Measure (Hamilton 1994); or
a 'yes' response to the item 'walking inside, with an aid if necessary (but with no standby help)' or 'yes' to 'walking on uneven ground' in the Rivermead Mobility Index (Collen 1991).
Secondary outcomes
We defined secondary outcomes as measures of activity limitations. We used walking speed (in metres per second), walking capacity (metres walked in 6 minutes), and the Rivermead Mobility Index score as relevant measures of activity limitations, if stated by the trialists. Additionally, we used death from all causes as a secondary outcome.
Adverse outcomes
We investigated the safety of electromechanical‐assisted gait‐training devices with the incidence of adverse outcomes such as thrombosis, major cardiovascular events, injuries, pain, and any other reported adverse events. To measure the acceptance of electromechanical‐assisted gait‐training devices in walking therapies, we used visual analogue scales or withdrawal from the study for any reason (dropout rates), or both during the study period, depending on data provided by the study authors.
Depending on the above‐stated categories and the availability of variables used in the included trials, we discussed and reached consensus on which outcome measures should be included in the analysis.
Search methods for identification of studies
See the 'Specialized register' section in the Cochrane Stroke Group module. We searched for trials in all languages and arranged for translation of relevant papers published in languages other than English.
Electronic searches
We searched the Cochrane Stroke Group Trials Register (last searched August 2016) and the following electronic bibliographic databases:
The Cochrane Central Register of Controlled Trials (CENTRAL) (the Cochrane Library, Issue 8, 2016) (Appendix 1);
MEDLINE in Ovid (1950 to 15 August 2016) (Appendix 2);
Embase (1980 to 15 August 2016) (Appendix 3);
CINAHL (Cumulative Index to Nursing and Allied Health Literature) in EBSCO (1982 to 15 August 2016) (Appendix 4);
AMED (Allied and Complementary Medicine Database) (1985 to 15 August 2016) (Appendix 5);
Web of Science (Science Citation Index Expanded, Social Sciences Citation Index, Arts and Humanities Citation Index) (1899 to 16 August 2016) (Appendix 6);
PEDro (Physiotherapy Evidence Database) (searched 16 August 2016) (Appendix 7);
COMPENDEX (1972 to 16 November 2012) (Appendix 8);
SPORTDiscus (1949 to 15 September 2012) (Appendix 9); and
Inspec (1969 to 26 August 2016) (Appendix 10).
We developed the search strategies with the help of the Cochrane Stroke Group Information Specialist and adapted the MEDLINE search strategy for the other databases.
We identified and searched the following ongoing trials and research registers:
International Standard Randomised Controlled Trial Number Register at www.isrctn.com/ (searched August 2016);
ClinicalTrials.gov at www.clinicaltrials.gov (searched 27 August 2016);
Stroke Trials Register at www.strokecenter.org (searched 27 August 2016); and
World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) at apps.who.int/trialsearch/ (searched 27 August 2016) (Appendix 11).
Searching other resources
We also:
-
handsearched the following relevant conference proceedings:
World Congress of NeuroRehabilitation (2002, 2006, 2008, 2010, 2012, 2014, and 2016);
World Congress of Physical Medicine and Rehabilitation (2001, 2003, 2005, 2007, 2009, 2011, 2013, and 2015);
World Congress of Physical Therapy (2003, 2007, 2011, and 2015);
Deutsche Gesellschaft für Neurotraumatologie und Klinische Neurorehabilitation (2001 to 2015);
Deutsche Gesellschaft für Neurologie (2000 to 2016);
Deutsche Gesellschaft für Neurorehabilitation (1999 to 2016); and
Asia‐Oceanian Conference of Physical & Rehabilitation Medicine (2008 to 2016).
screened reference lists of all relevant articles; and
contacted trialists, experts, and researchers in our field of study.
Data collection and analysis
Selection of studies
Two review authors (JM, BE) independently read the titles and abstracts of the identified references and eliminated obviously irrelevant studies. We obtained the full text for the remaining studies. Based on our inclusion criteria (types of studies, participants, aims of interventions, outcome measures), the same two review authors independently ranked these studies as relevant, irrelevant, or possibly relevant. We excluded all trials ranked initially as irrelevant but included all other trials at this stage. We excluded all trials of specific treatment components, such as electrical stimulation as stand‐alone treatment, treadmill training, and continuous passive motion treatment, because these have been the subject of other Cochrane Reviews (e.g. Mehrholz 2014). We resolved any disagreements through discussion between all four review authors. If we required further information to reach consensus, we contacted trialists in an attempt to obtain the missing information. We recorded the selection process in sufficient detail to complete a PRISMA flow diagram, and listed all studies that did not match our inclusion criteria regarding types of studies, participants, and aims of interventions in the Characteristics of excluded studies table.
Data extraction and management
Two review authors (JM, BE) independently extracted trial and outcome data from the selected trials. We established the characteristics of unpublished trials through correspondence with the trial co‐ordinator or principal investigator. If any review author was involved in any of the selected studies, another review author not involved in the study extracted the study information. If there was any doubt as to whether a study should be excluded, we retrieved the full text of the article. In cases of disagreement between the two review authors, a third review author (JK) reviewed the information to decide on inclusion or exclusion of a study. We used checklists to independently record the following details.
Methods of generating the randomisation schedule.
Method of concealment of allocation.
Blinding of assessors.
Use of an intention‐to‐treat analysis (all participants initially randomly assigned were included in the analyses as allocated to groups).
Adverse events and dropouts for all reasons.
Important imbalance in prognostic factors.
Participants (country, number of participants, age, gender, type of stroke, time from stroke onset to entry to the study, inclusion and exclusion criteria).
Comparison (details of the intervention in treatment and control groups, details of co‐intervention(s) in both groups, duration of treatment).
Outcomes and time points of measures (number of participants in each group and outcome, regardless of compliance).
The two review authors checked all of the extracted data for agreement, with a third review author (JK) arbitrating any items for which consensus could not be reached. If necessary, we contacted trialists to request more information, clarification, and missing data.
Assessment of risk of bias in included studies
Two review authors (JM, MP) independently evaluated the methodological quality of the included trials using the Cochrane 'Risk of bias' tool, as described in Chapter 8 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011a).
We checked all methodological quality assessments for agreement between review authors. We resolved disagreements by discussion. If one of the review authors was a co‐author of an included trial, another review author (BE or JK) conducted the methodological quality assessment for this trial in this case.
Measures of treatment effect
We planned to compare electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care) for primary and secondary outcome parameters. We used the effect measures odds ratio (OR) or mean difference (MD) in the meta‐analyses.
Unit of analysis issues
We analysed binary (dichotomous) outcomes with an OR, random‐effects model with 95% confidence intervals (CIs). We analysed continuous outcomes with MDs, using the same outcome scale. We used a random‐effects model for all analyses. We used Cochrane Review Manager 5 software for all statistical comparisons, (RevMan 2014).
Dealing with missing data
In the case of missing outcome data, we attempted to analyse data according to the intention‐to‐treat approach. We contacted the trial co‐ordinator or principal investigator if data were missing.
Assessment of heterogeneity
We used the I² statistic to assess heterogeneity. We used a random‐effects model, regardless of the level of heterogeneity.
Assessment of reporting biases
We inspected funnel plots to assess the risk of publication bias.
Data synthesis
GRADE and 'Summary of findings' table
We created two 'Summary of findings' tables using the following outcomes.
Primary outcome measure: Independent walking at the end of intervention phase, all electromechanical devices used. Scale from 0 to infinity.
Primary outcome measure: Recovery of independent walking at follow‐up after study end. Scale from 0 to infinity.
Primary outcome measure: Walking velocity (metres per second) at the end of intervention phase. Scale from 0 to infinity.
Secondary outcome measure: Walking velocity (metres per second) at follow‐up. Scale from 0 to infinity.
Secondary outcome measure: Walking capacity (metres walked in 6 minutes) at the end of intervention phase. Scale from 0 to infinity.
Secondary outcome measure: Walking capacity (metres walked in 6 minutes) at follow‐up. Scale from 0 to infinity.
Secondary outcome measure: Acceptability of electromechanical‐assisted gait‐training devices during intervention phase: number of dropouts.
We used the five GRADE considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) to assess the quality of a body of evidence as it relates to the studies that contribute data to the meta‐analyses for the prespecified outcomes (Atkins 2004). We used methods and recommendations described in Section 8.5 and Chapter 12 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011b), employing GRADEpro GDT software (GRADEpro GDT). We justified all decisions to down‐ or upgrade the quality of studies using footnotes, and made comments to aid the reader's understanding of the review where necessary.
Subgroup analysis and investigation of heterogeneity
As planned in our protocol (Mehrholz 2006), we performed a formal subgroup analysis following the guidance in the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2011), comparing participants treated in the acute and subacute phases of their stroke (within three months) with participants treated in the chronic phase (longer than three months).
Sensitivity analysis
As planned in our protocol, we performed a sensitivity analysis of methodological quality for each included study.
We carried out the following sensitivity analyses by including only those studies:
with an adequate sequence generation process;
with adequate concealed allocation;
with blinded assessors for the primary outcome; and
without incomplete outcome data.
We considered it necessary to do a further sensitivity analysis by removing the largest study, Pohl 2007, because some of the review authors (JM, MP, and CW) were investigators in this large trial. We carried out this sensitivity analysis by including all studies without the largest study (Pohl 2007).
We performed two further (post hoc) sensitivity analyses.
Ambulatory status at start of study (including only studies that included an independent walker; including only studies that included dependent and independent walkers; and including only studies that included a dependent walker).
Type of device used in trials (including only studies that used end‐effector devices and including only studies that used exoskeleton devices).
Results
Description of studies
See the Characteristics of included studies, Characteristics of excluded studies, and Characteristics of ongoing studies tables.
Results of the search
Figure 1 shows the flow diagram of the selection of studies for this update.
1.
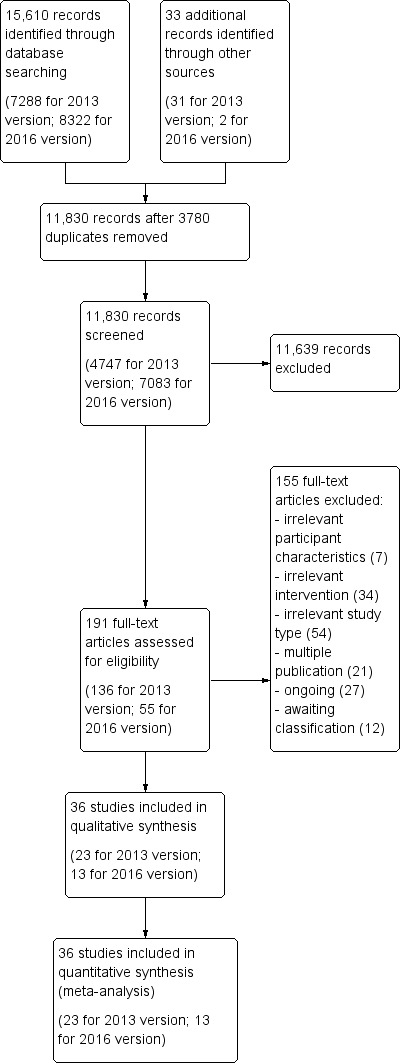
Study flow diagram.
Searches of the electronic databases and trials registers generated 7083 new unique references for screening. After excluding non‐relevant citations, we obtained the full text of 55 new papers, and from these identified and included 13 new trials in the review.
Included studies
We included 36 trials involving a total of 1472 participants (see the Characteristics of included studies, Figure 1, Table 7, and Table 8). All included studies investigated the effects of electromechanical‐ or robotic‐assisted gait‐training devices in improving walking after stroke.
1. Participant characteristics in studies.
| Study ID |
Experimental: age, mean (SD) |
Control: age, mean (SD) |
Experimental: time poststroke |
Control: time poststroke |
Experimental: sex |
Control: sex |
Experimental: side paresis |
Control: side paresis |
| Aschbacher 2006 | 57 years | 65 years | ≤ 3 months | ≤ 3 months | 2 female | 4 female | Not stated | Not stated |
| Bang 2016 | 54 years | 54 years | 12 months | 13 months | 5 male, 4 female | 4 male, 5 female | 4 right, 5 left | 4 right, 5 left |
| Brincks 2011 | 61 (median) years | 59 (median) years | 56 (median) days | 21 (median) days | 5 male, 2 female | 4 male, 2 female | 5 right, 2 left | 1 right, 5 left |
| Buesing 2015 | 60 years | 62 years | 7 years | 5 years | 17 male, 8 female | 16 male, 9 female | 13 right, 12 left | 12 right, 13 left |
| Chang 2012 | 56 (12) years | 60 (12) years | 16 (5) days | 18 (5) days | 13 male, 7 female | 10 male, 7 female | 6 right, 14 left | 6 right, 11 left |
| Cho 2015 | 55 (12) years | 55 (15) years | 15 months | 13 months | Not stated | Not stated | 6 right, 4 left (4 both) | 3 right, 1 left (3 both) |
| Chua 2016 | 62 (10) years | 61 (11) years | 27 (11) days | 30 (14) days | 35 male, 18 female | 40 male, 13 female | 24 right, 29 left | 21 right, 32 left |
| Dias 2006 | 70 (7) years | 68 (11) years | 47 (64) months | 48 (30) months | 16 male, 4 female | 14 male, 6 female | Not stated | Not stated |
| Fisher 2008 | Not stated | Not stated | Less than 12 months | Less than 12 months | Not stated | Not stated | Not stated | Not stated |
| Forrester 2014 | 63 years | 60 years | 12 days | 11 days | Not stated | Not stated | 9 right, 9 left | 7 right, 9 left |
| Geroin 2011 | 63 (7) years | 61 (6) years | 26 (6) months | 27 (6) months | 14 male, 6 female | 9 male, 1 female | Not stated | Not stated |
| Han 2016 | 68 (15) years | 63 (11) years | 22 (8) days | 18 (10) days | Not stated | Not stated | 20 right, 10 left | 14 right, 12 left |
| Hidler 2009 | 60 (11) years | 55 (9) years | 111 (63) days | 139 (61) days | 21 male, 12 female | 18 male, 12 female | 22 right, 11 left | 13 right, 17 left |
| Hornby 2008 | 57 (10) years | 57 (11) years | 50 (51) months | 73 (87) months | 15 male, 9 female | 15 male, 9 female | 16 right, 8 left | 16 right, 8 left |
| Husemann 2007 | 60 (13) years | 57 (11) years | 79 (56) days | 89 (61) days | 11 male, 5 female | 10 male, 4 female | 12 right, 4 left | 11 right, 3 left |
| Kim 2015 | 54 (13) years | 50 (16) years | 80 (60) days | 120 (84) days | 9 male, 4 female | 10 male, 3 female | 8 right, 5 left | 10 right, 3 left |
| Kyung 2008 | 48 (8) years | 55 (16) years | 22 (23) months | 29 (12) months | 9 male, 8 female | 4 male, 4 female | 9 right, 8 left | 4 right, 4 left |
| Mayr 2008 | Not stated | Not stated | Between 10 days and 6 months | Between 10 days and 6 months | Not stated | Not stated | Not stated | Not stated |
| Morone 2011 | 62 (11) years | 62 (14) years | 19 (11) days | 20 (14) days | 15 male, 9 female | 13 male, 11 female | 13 right, 11 left | 15 right, 9 left |
| Noser 2012 | 67 (9) years | 64 (11) years | 1354 days | 525 days | 7 male, 4 female | 6 male, 4 female | Not stated | Not stated |
| Ochi 2015 | 62 (8) years | 66 (12) years | 23 (7) days | 26 (8) days | 11 male, 2 female | 9 male, 4 female | 6 right, 7 left | 5 right, 8 left |
| Peurala 2005 | 52 (8) years | 52 (7) years | 2.5 (2.5) years | 4.0 (5.8) years | 26 male, 4 female | 11 male, 4 female | 13 right, 17 left | 10 right, 5 left |
| Peurala 2009 | 67 (9) years | 68 (10) years | 8 (3) days | 8 (3) days | 11 male, 11 female | 18 male, 16 female | 11 right, 11 left | 14 right, 20 left |
| Picelli 2016 | 62 (10) years | 65 (3) years | 6 (4) years | 6 (4) years | 7 male, 4 female | 9 male, 2 female | Not stated | Not stated |
| Pohl 2007 | 62 (12) years | 64 (11) years | 4.2 (1.8) weeks | 4.5 (1.9) weeks | 50 male, 27 female | 54 male, 24 female | 36 right, 41 left | 33 right, 45 left |
| Saltuari 2004 | 62 (13) years | 60 (19) years | 3.6 (4.6) months | 1.9 (0.8) months | 4 male, 4 female | 2 male, 6 female | Not stated | Not stated |
| Schwartz 2006 | 62 (9) years | 65 (8) years | 22 (9) days | 24 (10) days | 21 male, 16 female | 20 male, 10 female | 17 right, 20 left | 8 right, 22 left |
| Stein 2014 | 58 (11) years | 57 (15) years | 49 (39) months | 89 (153) months | Not stated | Not stated | Not stated | Not stated |
| Tanaka 2012 | 63 (10) years | 60 (9) years | 55 (37) months | 65 (67) months | 10 male, 2 female | 9 right, 3 left | ||
| Tong 2006 | 71 (14) years | 64 (10) years | 2.5 (1.2) weeks | 2.7 (1.2) weeks | 19 male, 11 female | 12 male, 8 female | 13 right, 17 left | 7 right, 13 left |
| Ucar 2014 | 56 years | 62 years | Not stated | Not stated | Not stated | Not stated | Not stated | Not stated |
| Van Nunen 2012 | 53 (10) years | 2.1 (1.3) months | 16 male, 14 female | Not stated | Not stated | |||
| Waldman 2013 | 51 (8) years | 53 (7) years | 41 (20) months | 30 (22) months | Not stated | Not stated | Not stated | Not stated |
| Watanabe 2014 | 67 (17) years | 76 (14) years | 59 (47) days | 51 (34) days | 7 male, 4 female | 4 male, 7 female | 6 right, 5 left | 5 right, 6 left |
| Werner 2002 | 60 (9) years | 60 (9) years | 7.4 (2.0) weeks | 6.9 (2.1) weeks | 8 male, 7 female | 5 male, 10 female | 8 right, 7 left | 8 right, 7 left |
| Westlake 2009 | 59 (17) years | 55 (14) years | 44 (27) months | 37 (20) months | 6 male, 2 female | 7 male, 1 female | 4 right, 4 left | 3 right, 5 left |
SD: standard deviation
2. Demographics of studies including dropouts and adverse events.
| Criteria | Stroke severity | Electromechanical device used | Duration of study intervention | Aetiology (ischaemic/haemorrhage) | Intensity of treatment per day | Description of the control intervention | Dropouts |
Reasons for dropout and adverse events in the experimental group |
Reasons for dropout and adverse events in the control group |
Source of information |
| Aschbacher 2006 | Not stated | Lokomat | 3 weeks | Not stated | 30 minutes, 5 times a week | Described as task‐oriented physiotherapy, 5 times a week for 3 weeks (2.5 hours a week) | 4 of 23 | Not stated | Not stated | Unpublished information in the form of a conference presentation |
| Bang 2016 | Unclear | Lokomat | 4 weeks | 13/5 | 60 minutes, 5 times a week (20 sessions) | Described as treadmill training without body weight support | 0 of 18 | ‐ | ‐ | Published information |
| Brincks 2011 | Mean FIM, 92 of 126 points | Lokomat | 3 weeks | Not stated | Not stated | Physiotherapy | 0 of 13 | ‐ | ‐ | Unpublished and published information provided by the authors. |
| Buesing 2015 | Unclear | Wearable exoskeleton Stride Management Assist system (SMA) | 6 to 8 weeks | Unclear | 3 times per week for a maximum of 18 sessions | Functional task‐specific training (intensive overground training and mobility training) | 0 of 50 | ‐ | ‐ | Published information |
| Chang 2012 | Not stated | Lokomat | 10 days | Not stated | 30 minutes daily for 10 days | Conventional gait training by physical therapists (with equal therapy time and same amount of sessions as experimental group) | 3 of 40 | Not described by group (3 participants dropped out: 1 due to aspiration pneumonia, and 2 were unable to co‐operate fully with the experimental procedure) |
Unpublished and published information provided by the authors. | |
| Cho 2015 | Mean Modified Barthel Index, 36 points | Lokomat | 8 weeks (2 phases, cross‐over after 4 weeks) | 4/14 (2 both) | 30 minutes, 3 times a week for 4 weeks | Bobath (neurophysiological exercises, inhibition of spasticity and synergy pattern) | 0 of 20 | ‐ | ‐ | Published information |
| Chua 2016 | Mean Barthel Index, 49 points | Gait Trainer | 8 weeks | Not stated | Not stated | Physiotherapy including 25 minutes of stance/gait, 10 minutes cycling, 10 minutes tilt table standing | 20 of 106 | 2 death, 3 refusal, 1 medical problem, 1 transport problem (1 pain as adverse event) |
1 death, 6 refusal, 3 medical problem, 1 administrative problem (no adverse events) |
Published information |
| Dias 2006 | Mean Barthel Index, 75 points | Gait Trainer | 4 weeks | Not stated | 40 minutes, 5 times a week | Bobath method, 5 times a week for 5 weeks | 0 of 40 | ‐ | ‐ | Unpublished and published information provided by the authors. |
| Fisher 2008 | Not stated | AutoAmbulator | 24 sessions | Not stated | Minimum of 3 sessions a week up to 5 sessions; number of minutes in each session unclear | "Standard" physical therapy, 3 to 5 times a week for 24 consecutive sessions | 0 of 20 | 14 adverse events, no details provided |
11 adverse events, no details provided |
Unpublished and published information provided by the authors. |
| Forrester 2014 | Mean FIM walk 1 point | Anklebot | 8 to 10 sessions (with ca. 200 repetitions) | Not stated | 60 minutes, 8 to 10 sessions | Stretching of the paretic ankle | 5 of 34 | Total of 5 dropouts in both groups (1 medical complication, 1 discharge prior study end, 2 time poststroke > 49 days, 1 non‐compliance) | Published information provided by the authors. | |
| Geroin 2011 | Mean European Stroke Scale, 80 points | Gait Trainer | 2 weeks | Not stated | 50 minutes, 5 times a week | Walking exercises according to the Bobath approach | 0 of 30 | ‐ | ‐ | Unpublished and published information provided by the authors. |
| Han 2016 | Not stated | Lokomat | 4 weeks | 33/23 | 30 minutes, 5 times a week | Neurodevelopmental techniques for balance and mobility | 4 0f 60 | ‐ | 4 unclear reasons | Published information provided by the authors. |
| Hidler 2009 | Not stated | Lokomat | 8 to 10 weeks (24 sessions) | 47/16 | 45 minutes, 3 days a week | Conventional gait training, 3 times a week for 8 to 10 weeks (24 sessions), each session lasted 1.5 hours | 9 of 72 | Not described by group (9 withdrew or were removed because of poor attendance or a decline in health, including 1 death, which according to the authors was unrelated to study) |
Unpublished and published information provided by the authors. | |
| Hornby 2008 | Not stated | Lokomat | 12 sessions | 22/26 | 30 minutes, 12 sessions | Therapist‐assisted gait training, 12 sessions, each session lasted 30 minutes | 14 of 62 | 4 participants dropped out (2 discontinued secondary to leg pain during training, 1 experienced pitting oedema, and 1 had travel limitations) |
10 participants dropped out (4 discontinued secondary to leg pain, 1 experienced an injury outside therapy, 1 reported fear of falling during training, 1 presented with significant hypertension, 1 had travel limitations, and 2 experienced subjective exercise intolerance) |
Published information provided by the authors. |
| Husemann 2007 | Median Barthel Index, 35 points | Lokomat | 4 weeks | 22/8 | 30 minutes, 5 times a week | Conventional physiotherapy, 30 minutes per day for 4 weeks | 2 of 32 | 1 participant enteritis | 1 participant pulmonary embolism | Information as provided by the authors |
| Kim 2015 | Mean Barthel Index, 20 points | Walkbot | 4 weeks | 13/13 | 30 minutes, 5 times a week | Conventional physiotherapy (bed mobility, stretching, balance training, strengthening, symmetry training, treadmill training) | 4 of 30 | 1 rib fracture, 3 decline in health condition | Information as provided by the authors | |
| Kyung 2008 | Not stated | Lokomat | 4 weeks | 18/7 | 45 minutes, 3 days a week | Conventional physiotherapy, received equal time and sessions of conventional gait training | 10 of 35 | 1 participant dropped out for private reasons (travelling); adverse events not described |
9 participants refused after randomisation (reasons not provided); adverse events not described | Unpublished and published information provided by the authors. |
| Mayr 2008 | Not stated | Lokomat | 8 weeks | Not stated | Not stated | Add‐on conventional physiotherapy, received equal time and sessions of conventional gait training | 13 of 74 | 4 participants dropped out (reasons not provided); adverse events not described | 9 participants dropped out (reasons not provided) | Unpublished and published information provided by the authors. |
| Morone 2011 | Canadian Neurological Scale, 6 points | Gait Trainer | 4 weeks | 41/7 | 40 minutes, 5 times a week | Focused on trunk stabilisation, weight transfer to the paretic leg, and walking between parallel bars or on the ground. The participant was helped by 1 or 2 therapists and walking aids if necessary. | 21 of 48 | 12 (hypotension, referred weakness, knee pain, urinary infection, uncontrolled blood pressure, fever, absence of physiotherapist) | 9 (hypotension, referred weakness, knee pain, ankle pain, uncontrolled blood pressure, fever, absence of physiotherapist) | Information as provided by the authors |
| Noser 2012 | Not stated | Lokomat | Unclear | Not stated | Not stated | Not stated | 1 of 21 | No dropouts; 2 serious adverse events (1 skin breakdown as a result of therapy, 1 second stroke during the post‐treatment phase) |
1 dropout due to protocol violation; 2 serious adverse events (1 sudden drop in blood pressure at participant's home leading to brief hospitalisation, 1 sudden chest pain before therapy leading to brief hospitalisation) |
Information as provided by the authors |
| Ochi 2015 | Not stated | Gait‐assistance robot (consisting of 4 robotic arms for the thighs and legs, thigh cuffs, leg apparatuses, and a treadmill) | 4 weeks | 10/16 | 20 minutes, 5 times a week for 4 weeks, in addition to rehabilitation treatment | Range‐of‐motion exercises, muscle strengthening, rolling over and sit‐to‐stand and activity and gait exercises | 0 of 26 | ‐ | ‐ | Published information |
| Peurala 2005 | Scandinavian Stroke Scale, 42 points | Gait Trainer | 3 weeks | 25/20 | 20 minutes, 5 times a week for 3 weeks, in addition to rehabilitation treatment | Walking overground; all participants practised gait for 15 sessions over 3 weeks (each session lasting 20 minutes) |
0 of 45 | ‐ | ‐ | Published information |
| Peurala 2009 | Not stated | Gait Trainer | 3 weeks | 42/14 | 20 minutes, 5 times a week for 3 weeks, in addition to rehabilitation treatment | Overground walking training; in the other control group, 1 or 2 physiotherapy sessions daily but not at the same intensity as in the other groups | 9 of 56 | 5 dropouts (2 situation worsened after 1 to 2 treatment days; 1 had 2 unsuccessful attempts in device; 1 had scheduling problems; 1 felt protocol too demanding) |
4 dropouts (1 felt protocol too demanding; 2 situation worsened after 1 to 2 treatment days; 1 death) |
Published information |
| Picelli 2016 | Not stated | G‐EO System Evolution | Experimental group (G‐EO) 30 minutes a day for 5 consecutive days | Not stated | 5 days in addition to botulinum toxin injection of calf muscles | None | 0 of 22 | ‐ | ‐ | Published information |
| Pohl 2007 | Mean Barthel Index, 37 points | Gait Trainer | 4 weeks | 124/31 | 20 minutes, 5 times a week | Physiotherapy every weekday for 4 weeks | 11 of 155 | 2 participants refused therapy, 1 increased cranial pressure, 1 relapsing pancreas tumour, 1 cardiovascular unstable |
4 participants refused therapy, 1 participant died, 1 myocardial infarction | Published information |
| Saltuari 2004 | Not stated | Lokomat | 2 weeks | 13/3 | A‐B‐A study: in phase A, 30 minutes, 5 days a week | Physiotherapy every weekday for 3 weeks (phase B) | 0 of 16 | None | None | Unpublished and published information provided by the authors. |
| Schwartz 2006 | Mean NIHSS, 11 points | Lokomat | 6 weeks | 49/67 | 30 minutes, 3 times a week | Physiotherapy with additional gait training 3 times a week for 6 weeks | 6 of 46 | 2 participants with leg wounds, 1 participant with recurrent stroke, 1 refused therapy |
1 participant with recurrent stroke, 1 with pulmonary embolism |
Unpublished and published information provided by the authors. |
| Stein 2014 | Not stated | Bionic leg device (AlterG) | 6 weeks | Not stated | 1 hour, 3 times a week for 6 weeks | Group exercises | 0 of 24 | ‐ | ‐ | Published information |
| Tanaka 2012 | Mean FIM, 79 points | Gait Master4 | 4 weeks | Not stated | 20 minutes, 2 or 3 times a week (12 sessions) | Non‐intervention (non‐training) | 0 of 12 | ‐ | ‐ | Published information |
| Tong 2006 | Mean Barthel Index, 51 points | Gait Trainer | 4 weeks | 39/11 | 20 minutes, 5 times a week | Conventional physiotherapy alone, based on Bobath concept | 4 of 50 | None | 2 participants discharged before study end, 1 participant readmitted to an acute ward, 1 participant deteriorating condition |
Published information |
| Ucar 2014 | Not stated | Lokomat | 2 weeks | Not stated | 30 minutes, 5 times a week | Conventional physiotherapy at home (focused on gait) | 0 of 22 | ‐ | ‐ | Published information |
| Van Nunen 2012 | Not stated | Lokomat | 8 weeks | Not stated | 30 minutes, twice a week | Overground therapy | 0 of 30 | ‐ | ‐ | Unpublished and published information provided by the author. |
| Waldman 2013 | Not stated | Portable rehab robot (ankle device) | 6 weeks | Not stated | 3 times a week, 18 sessions | Stretching the plantar flexors and active exercises for ankle mobility and strength | 0 of 24 | ‐ | ‐ | Published information |
| Watanabe 2014 | Not stated | Single‐leg version of the Hybrid Assistive Limb (HAL) | 4 weeks | 11/11 | 20 minutes, 12 sessions | Aimed to improve walking speed, endurance, balance, postural stability, and symmetry | 10 of 32 | 4 withdrew, 1 epilepsy, 1 technical reasons |
2 pneumonia, 2 discharged |
Published information |
| Werner 2002 | Mean Barthel Index, 38 points | Gait Trainer | 2 weeks | 13/12 | 20 minutes, 5 times a week | Gait therapy including treadmill training with body weight support | 0 of 30 | None | None | Published information |
| Westlake 2009 | Not stated | Lokomat | 4 weeks (12 sessions) | 8/8 | 30 minutes, 3 times a week | 12 physiotherapy sessions including manually guided gait training (3 times a week over 4 weeks) | 0 of 16 | None | None | Published information |
FIM: Functional Independence Measure NIHSS: National Institutes of Health Stroke Scale
For one of the included studies published only as an abstract we obtained at least some results through correspondence with the trial co‐ordinator or principal investigator (Mayr 2008). Another study was not yet published, but the results of the trial were presented orally, and we were able to obtain a handout with information about the study from the principal investigator (Aschbacher 2006).
A detailed description of all participant characteristics can be found in Table 7 and Table 8 (see also the Characteristics of included studies). The mean age in the included studies ranged from 48 years, in Kyung 2008, to 76 years, in Watanabe 2014 (Table 7). More males than females were included the studies (approximately 60% males). More participants with ischaemic stroke than haemorrhagic stroke lesions (approximately 70% ischaemic stroke) were included, and almost as many participants with left‐sided hemiparesis compared with participants with right‐sided hemiparesis (approximately 50% left‐sided) were included in the studies (see Table 7 and Table 8).
Twelve studies provided information about baseline stroke severity (Table 8), of which seven used the Barthel Index score, ranging from 20 Barthel Index points, in Kim 2015, to 75 of 100 Barthel Index points, in Dias 2006 (Table 8). Details of all inclusion and exclusion criteria used in the studies can be found in the Characteristics of included studies table.
The duration of study intervention (time frame during which experimental interventions were applied) was heterogeneous, ranging from 10 days, in Chang 2012, to eight weeks, in Mayr 2008. The study intervention period for most studies was three or four weeks (Table 8). Fifteen of the 36 studies included participants who could walk independently at the start of the study; a further nine studies included participants who were dependent and independent walkers (Analysis 4.1); and 12 studies included only non‐ambulatory participants (Analysis 4.1). The experimental intervention in 17 studies was the robotic‐assisted device Lokomat, and the experimental intervention in nine studies was the electromechanical‐assisted device Gait Trainer; a detailed description of devices used in studies can be found in Table 8.
4.1. Analysis.
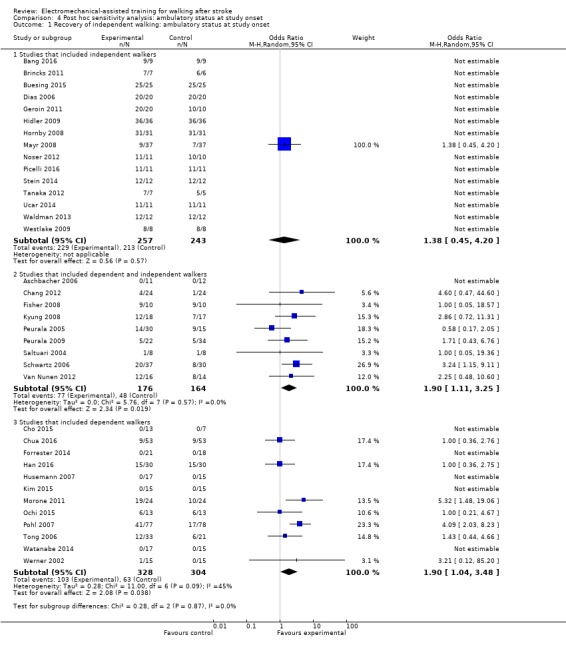
Comparison 4 Post hoc sensitivity analysis: ambulatory status at study onset, Outcome 1 Recovery of independent walking: ambulatory status at study onset.
Frequency (in terms of therapy provided per week) of treatment ranged from two or three times a week, in Tanaka 2012, to five times a week (Table 8). Intensity (in terms of duration of experimental therapy provided) of treatment ranged from 20 minutes, in Werner 2002, to 60 minutes, in Forrester 2014. In many studies, details of the interventions were unclear or incomplete, for example details about the intensity of the experimental treatment were unclear in some studies (Table 8). Except for Tanaka 2012 and Picelli 2016, the gait training time did not differ between control and experimental groups in the included studies. Eleven included studies used a follow‐up assessment after the study ended (Buesing 2015; Chua 2016; Dias 2006; Hidler 2009; Hornby 2008; Peurala 2005; Peurala 2009; Pohl 2007; Schwartz 2006; Stein 2014; Waldman 2013). Most studies investigated improvement in walking function as a primary outcome for analysis and used the Functional Ambulation Category (FAC) or comparable scales to assess independent walking. Furthermore, frequently investigated outcomes included assessment of walking function using gait velocity in metres per second. A more detailed description of the primary and secondary outcomes for each trial can be found in the Characteristics of included studies table.
We found the highest dropout rates for all reasons at the end of the treatment phase to be 23%, in Hornby 2008, and 29%, in Kyung 2008. Seventeen trialists reported no dropouts at scheduled follow‐up (Analysis 1.7; Table 8).
1.7. Analysis.
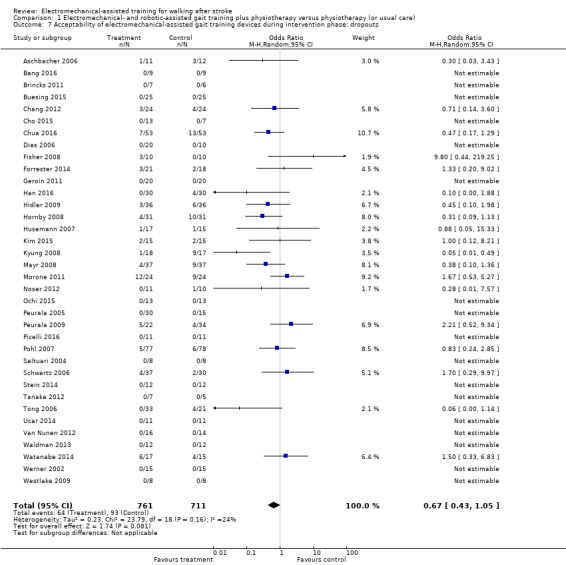
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 7 Acceptability of electromechanical‐assisted gait training devices during intervention phase: dropouts.
Excluded studies
We excluded 24 studies (see Characteristics of excluded studies and Figure 1 for further information).
Ongoing studies and studies awaiting assessment
We identified 16 ongoing studies (see Characteristics of ongoing studies). Thirteen studies for which we were unable to make contact with the trialists are still awaiting assessment (see Characteristics of studies awaiting classification).
Risk of bias in included studies
The risk of bias in included studies is described in greater detail in Characteristics of included studies and Figure 2.
2.
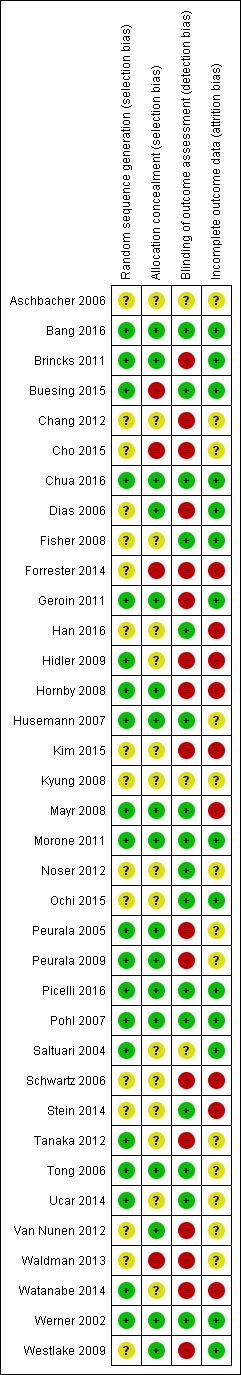
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
We wrote to the trialists of all included studies and studies awaiting assessment to request clarification of design features or for missing information to complete the quality ratings. We sent the correspondence via email or letter, followed by reminders every month if we received no response. Most trialists provided at least some of the requested data, but we were not able to obtain all of the required data.
Two review authors (JM, MP) used the 'Risk of bias' assessment tool to independently assess the methodological quality of the studies for the domains random sequence generation, allocation concealment, blinding of outcome assessment, and incomplete outcome data for all of the included trials except two (Pohl 2007; Werner 2002), which two other review authors (BE, JK) rated in an interview with the trialists. The review authors discussed all disagreements and sought arbitration by another review author (JK or BE) if necessary.
Allocation
Of the 36 included studies, 20 described adequate random sequence generation, and 17 described adequate allocation concealment.
Blinding
Of the 36 included studies, 6 reported blinding of the primary outcome assessment.
Incomplete outcome data
Of the 36 included studies, 14 reported incomplete outcome data (attrition bias).
Selective reporting
For the majority of studies, particularly the older trials, we did not find study protocols. Where study protocols were available, there was no evidence of selective reporting of outcomes relevant to this review.
Other potential sources of bias
Five out of 36 included trials used a cross‐over design with random allocation to the order of treatment sequences (Brincks 2011; Cho 2015; Saltuari 2004; Tanaka 2012; Werner 2002). We analysed only the first intervention period as a parallel‐group trial in this review. All other included studies used a parallel‐group design with true randomisation to group allocation.
Three studies used two experimental groups and one control group (Geroin 2011; Peurala 2005; Tong 2006), and one study used one experimental group and two control groups (Peurala 2009). In the former three studies (Geroin 2011; Peurala 2005; Tong 2006), additional functional electrical stimulation of leg muscles (or transcranial stimulation of the brain in Geroin 2011) during gait training was applied in one of the treatment groups. Because functional electrical stimulation or transcranial stimulation of the brain was done as an adjunct during electromechanical‐assisted gait training, and because the results in these experimental groups did not differ significantly, we combined the results of both experimental groups into one (collapsed) group and compared this with the results from the control group. In one study, an electromechanical‐assisted device was used in the experimental group and was compared with two control groups that did not use a device (Peurala 2009). Because we were interested in the effects of electromechanical‐ and robotic‐assisted gait‐training devices for improving walking after stroke, we combined the results of both control groups without devices into one (collapsed control) group and compared this with results of the one experimental group.
Effects of interventions
See: Table 1
Independent walking at the end of the intervention phase, all electromechanical devices used
Thrity‐six trials with a total of 1472 participants measured independent walking at study end, but for 21 included trials, no effect estimate (odds ratio (OR)) was feasible because no events (e.g. no participant reached the ability to walk) or only events (e.g. all participants regained walking) were reported (Analysis 1.1) (Deeks 2011).
1.1. Analysis.
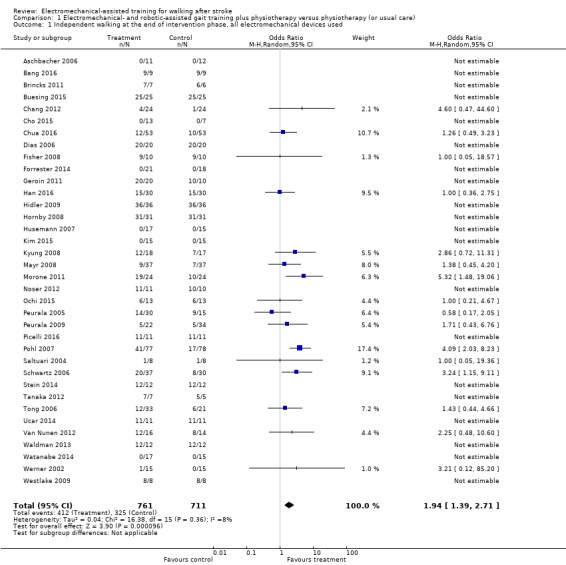
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 1 Independent walking at the end of intervention phase, all electromechanical devices used.
The use of electromechanical devices in gait rehabilitation for people after stroke increased the chance of walking independently (OR 1.94, 95% confidence interval (CI) 1.39 to 2.71; P < 0.001; level of heterogeneity I² = 8%; moderate‐quality evidence; Table 1). However, 15 out of 36 studies investigated at least some participants who were already independent in walking at the start of the study. A further nine studies included participants who were dependent and independent walkers, and 12 studies included only non‐ambulatory participants (Analysis 4.1). Of the total population of 1472 participants, approximately 39% were independent and approximately 59% were dependent walkers at the start of the study.
Recovery of independent walking at follow‐up after study end
Six trials with a total of 496 participants measured recovery of independent walking with follow‐up after the study end (Chua 2016; Hidler 2009; Hornby 2008; Peurala 2009; Pohl 2007; Tong 2006), but for two included trials (with 125 participants), no effect estimate (OR) was feasible because no events (e.g. no participant reached ability to walk) or only events (e.g. all participants regained walking) were reported (Analysis 1.2). The use of electromechanical devices for gait rehabilitation of people after stroke did not increase the chance of walking independently at follow‐up after study end (OR 1.93, 95% CI 0.72 to 5.13; P = 0.19; level of heterogeneity I² = 79%; moderate‐quality evidence). However, some included trials investigated participants who were already independent in walking at the start of the study. We could draw no definitive conclusion regarding a longer‐lasting effect of the use of electromechanical devices.
1.2. Analysis.
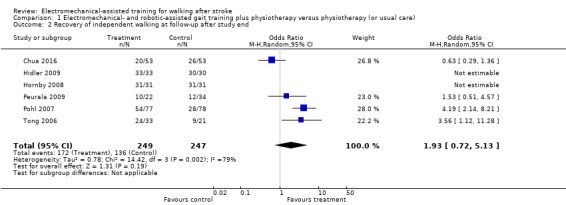
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 2 Recovery of independent walking at follow‐up after study end.
Walking velocity (metres per second) at the end of the intervention phase
Twenty‐four trials with a total of 985 participants provided data for walking velocity (m/s) at study end (Analysis 1.3). The use of electromechanical devices for gait rehabilitation did not significantly increase walking velocity. The pooled mean difference (MD) (random‐effects model) for walking velocity was 0.04 m/s (95% CI 0.00 to 0.09; P = 0.08; level of heterogeneity I² = 65%; low‐quality evidence). Participants who were unable to walk were regarded as having a walking velocity of zero metres per second.
1.3. Analysis.
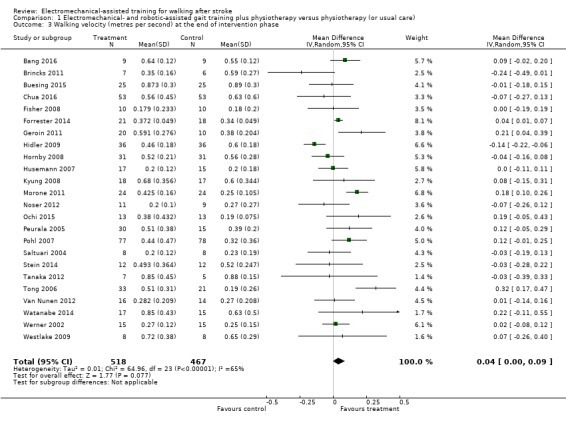
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 3 Walking velocity (metres per second) at the end of intervention phase.
Walking velocity (metres per second) at follow‐up
Nine trials with a total of 578 participants provided data for walking velocity (m/s) at follow‐up after study end (Buesing 2015; Chua 2016; Hidler 2009; Hornby 2008; Kyung 2008; Noser 2012; Pohl 2007; Stein 2014; Tong 2006). The use of electromechanical devices for gait rehabilitation did not significantly increase the walking velocity at follow‐up after study end. The pooled MD (random‐effects model) for walking velocity was 0.07 m/s (95% CI ‐0.05 to 0.19; P = 0.26; level of heterogeneity I² = 80%; moderate‐quality evidence; Analysis 1.4). Participants who were unable to walk were regarded as having a walking velocity of zero metres per second. We could draw no definitive conclusion regarding a longer‐lasting effect of the use of electromechanical devices for walking velocity.
1.4. Analysis.
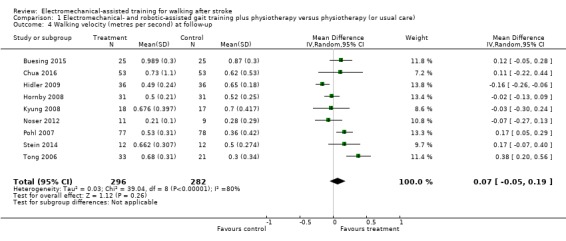
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 4 Walking velocity (metres per second) at follow‐up.
Walking capacity (metres walked in 6 minutes) at the end of the intervention phase
Twelve trials with a total of 594 participants provided data for walking capacity (metres walked in 6 minutes) at study end (Chua 2016; Hidler 2009; Hornby 2008; Noser 2012; Peurala 2005; Picelli 2016; Pohl 2007; Saltuari 2004; Stein 2014; Waldman 2013; Watanabe 2014; Westlake 2009). The use of electromechanical devices in gait rehabilitation did not increase the walking capacity of people after stroke. The pooled MD (random‐effects model) for walking capacity was 5.84 metres walked in 6 minutes (95% CI ‐16.73 to 28.40; P = 0.61; level of heterogeneity I² = 53%; very low‐quality evidence; Analysis 1.5).
1.5. Analysis.
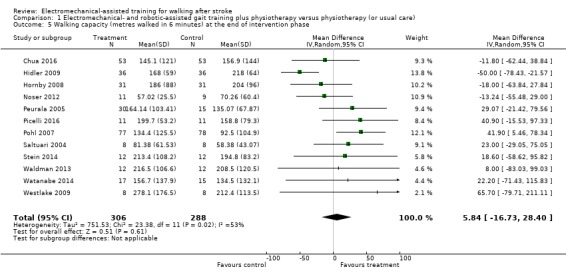
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 5 Walking capacity (metres walked in 6 minutes) at the end of intervention phase.
Walking capacity (metres walked in 6 minutes) at follow‐up
Seven trials with a total of 463 participants provided data for walking capacity (metres walked in 6 minutes) at follow‐up after study end (Chua 2016; Hidler 2009; Hornby 2008; Noser 2012; Pohl 2007; Stein 2014; Waldman 2013). The use of electromechanical devices for gait rehabilitation did not increase walking capacity at follow‐up after study end. The pooled MD (random‐effects model) for walking capacity was ‐0.82 metres walked in 6 minutes (95% CI ‐32.17 to 30.53; P = 0.96; level of heterogeneity I² = 58%; very low‐quality evidence; Analysis 1.6).
1.6. Analysis.
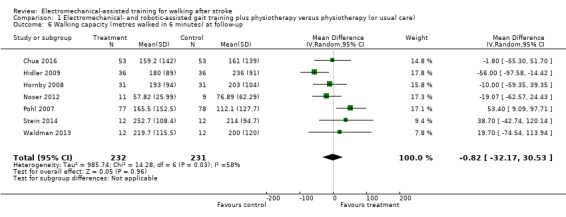
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 6 Walking capacity (metres walked in 6 minutes) at follow‐up.
Death from all causes until the end of the intervention phase
Only three larger trials reported any deaths during the intervention period (Chua 2016; Hidler 2009; Pohl 2007). In Pohl 2007 one participant in the control group died as the result of aspiration pneumonia, and one participant in the treatment group died due to recurrent stroke. In Hidler 2009, the group in which the death occurred was not stated. We therefore used a worst‐case (conservative) scenario and counted the one death for the experimental group. In the study of Chua 2016 the deaths occurred after the treatment period. The use of electromechanical devices for gait rehabilitation of non‐ambulatory people after stroke did not increase the risk of participants dying during the intervention period (risk difference (random‐effects model) 0.00, 95% CI ‐0.01 to 0.02; P = 0.77; level of heterogeneity I² = 0%; moderate‐quality evidence; Analysis 1.8).
1.8. Analysis.
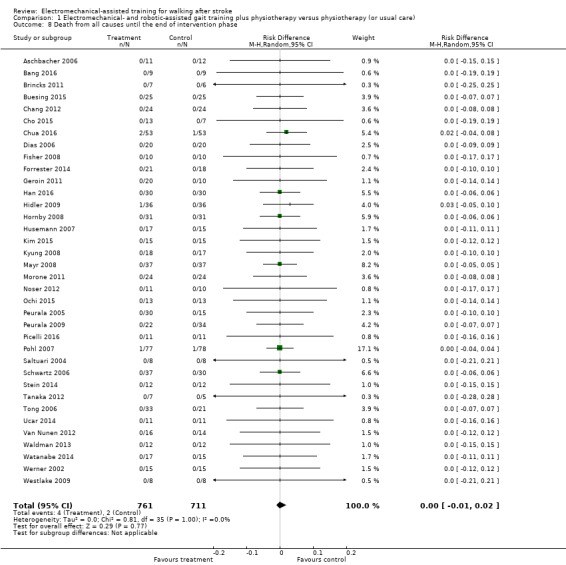
Comparison 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), Outcome 8 Death from all causes until the end of intervention phase.
Adverse outcomes: acceptability of electromechanical‐assisted gait‐training devices during the intervention phase in terms of dropouts
All trialists provided information about participants who dropped out from all causes during the trial period, but for 17 of the 36 included trials, no events/dropouts were reported (Analysis 1.7). The use of electromechanical devices for gait rehabilitation of non‐ambulatory people after stroke did not increase the risk of participants dropping out (OR (random‐effects model) 0.67, 95% CI 0.43 to 1.05; P = 0.08; level of heterogeneity I² = 24%; low‐quality evidence). The reasons for dropouts and all adverse events are described in detail for each trial in Table 8.
Regaining independent walking ability: planned sensitivity analysis by trial methodology
To examine the robustness of the results, we specified variables in a sensitivity analysis that we believed could influence the size of the observed effect (adequate sequence generation process, adequate concealed allocation, blinded assessors for the primary outcome, incomplete outcome data, and excluding the largest study). As stated above, for some of the included trials, no effect estimate (OR) was feasible (Analysis 2.1).
2.1. Analysis.

Comparison 2 Planned sensitivity analysis by trial methodology, Outcome 1 Regaining independent walking ability.
Studies with adequate sequence generation process
We included 20 trials with a total of 949 participants with an adequate sequence generation process (Figure 2). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.80, 95% CI 1.06 to 3.08; P = 0.03; level of heterogeneity I² = 38%).
Studies with adequate concealed allocation
We included 17 trials with a total of 831 participants with adequate concealed allocation (Figure 2). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.87, 95% CI 1.12 to 3.12; P = 0.02; level of heterogeneity I² = 37%).
Studies with blinded assessors for the primary outcome
Sixteen trials with a total of 762 participants had blinded assessors for the primary outcome (Figure 2). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.81, 95% CI 1.10 to 2.98; P = 0.02; level of heterogeneity I² = 31%).
Studies with complete outcome data
Fourteen trials with a total of 590 participants adequately described complete outcome data (Figure 2). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 2.23, 95% CI 1.16 to 4.29; P = 0.02; level of heterogeneity I² = 29%).
Excluding the largest study (Pohl 2007)
After excluding the largest study (Pohl 2007), 35 trials with a total of 1317 participants remained in this analysis. The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.65, 95% CI 1.17 to 2.34; P = 0.005; level of heterogeneity I² = 0%).
Subgroup analysis comparing participants in the acute and chronic phases of stroke
Independent walking at the end of the intervention phase, all electromechanical devices used
In our planned subgroup analysis comparing independent walking at the end of the intervention phase in people in the acute and chronic phases of stroke, we attempted to assign all included studies to one of two subgroups (acute and chronic phases).
Twenty trials with a total of 1143 participants investigated people in the acute or subacute phase, defined as less than or equal to three months after stroke (Analysis 3.1). As stated in the comparisons above, for some of the included trials no effect estimate (OR) was feasible (Analysis 3.1). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.90, 95% CI 1.38 to 2.63; P < 0.001; level of heterogeneity I² = 5%).
3.1. Analysis.
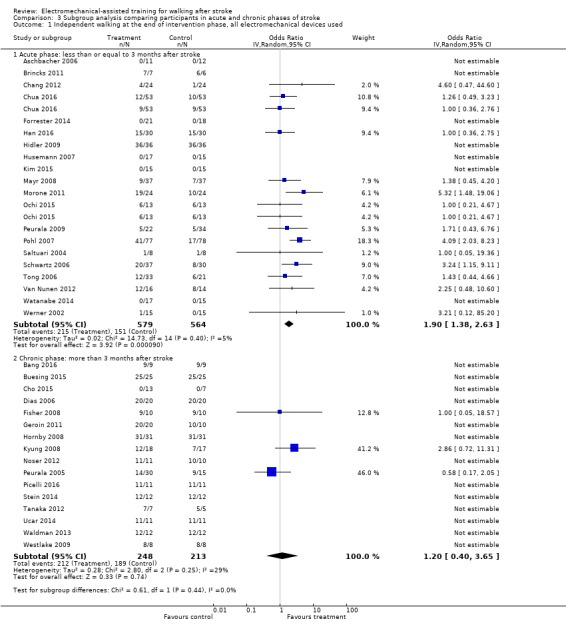
Comparison 3 Subgroup analysis comparing participants in acute and chronic phases of stroke, Outcome 1 Independent walking at the end of intervention phase, all electromechanical devices used.
Sixteen trials with a total of 461 participants investigated people in the chronic phase, defined as more than three months after stroke (Analysis 3.1). The use of electromechanical devices for gait rehabilitation of people after stroke did not increase the chance of walking independently (OR (random‐effects model) 1.20, 95% CI 0.40 to 3.65; P = 0.74; level of heterogeneity I² = 29%).
In a formal subgroup analysis, we did not find statistically significant differences in regaining independent walking between participants treated in the acute/subacute phase compared with participants treated in the chronic phase after stroke (Chi² = 0.61, df = 1; P = 0.44).
Post hoc sensitivity analysis by ambulatory status at study onset
Independent walking at the end of the intervention phase
To examine the robustness of the results and to explore the relationship between the main effect and walking status at the start of the study, we compared independent walking rates at the end of the intervention phase by ambulatory status at start of study.
Ambulatory participants at start of study
Fifteen trials with a total of 500 participants investigated independent walkers (Analysis 4.1). As stated in the comparisons above, for some of the included trials, no effect estimate (OR) was feasible; the conclusions are therefore based on one trial. The use of electromechanical devices for gait rehabilitation of people after stroke did not increase the chance of walking independently (OR (random‐effects model) 1.38, 95% CI 0.45 to 4.20; P = 0.57; level of heterogeneity I² = not applicable).
Ambulatory and non‐ambulatory participants at start of study
Nine trials with a total of 340 participants investigated a mixed population of dependent and independent walkers (Analysis 4.1). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.90, 95% CI 1.11 to 3.25; P = 0.02; level of heterogeneity I² = 0%).
Non‐ambulatory participants at start of study
Twelve trials with a total of 632 participants investigated dependent walkers (Analysis 4.1). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 1.90, 95% CI 1.04 to 3.48; P = 0.04; level of heterogeneity I² = 45%).
In a subgroup analysis, we did not find statistically significant differences between people who were dependent or independent walkers at the start of the study in regaining independent walking (Chi² = 0.28, df = 2; P = 0.87).
Walking speed at the end of the intervention phase
To examine the robustness of the results and to explore the relationship between walking velocity and ambulatory status at the start of the study, we compared achieved walking velocity at the end of the intervention phase by ambulatory status at the start of the study.
Ambulatory participants at start of study
Ten trials with a total of 317 participants investigated independent walkers at the start of the study and provided data for walking velocity (m/s) at study end (Analysis 4.2). The use of electromechanical devices for gait rehabilitation did not significantly increase walking velocity. The pooled MD (random‐effects model) for walking velocity was ‐0.02 m/s (95% CI ‐0.10 to 0.06; P = 0.66; level of heterogeneity I² = 59%).
4.2. Analysis.
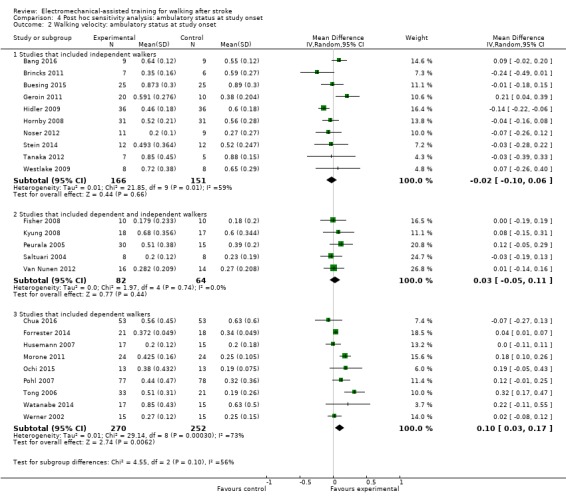
Comparison 4 Post hoc sensitivity analysis: ambulatory status at study onset, Outcome 2 Walking velocity: ambulatory status at study onset.
Ambulatory and non‐ambulatory participants at start of study
Five trials with a total of 146 participants investigated dependent and independent walkers at the start of the study and provided data for walking velocity (m/s) at study end (Analysis 4.2). The use of electromechanical devices for gait rehabilitation did not significantly increase walking velocity. The pooled MD (random‐effects model) for walking velocity was 0.03 m/s (95% CI ‐0.05 to 0.11; P = 0.44; level of heterogeneity I² = 0%).
Non‐ambulatory participants at start of study
Nine trials with a total of 522 participants investigated dependent walkers at the start of the study and provided data for walking velocity (m/s) at study end (Analysis 4.2). The use of electromechanical devices for gait rehabilitation significantly increased walking velocity. The pooled MD (random‐effects model) for walking velocity was 0.10 m/s (95% CI 0.03 to 0.17; P = 0.006; level of heterogeneity I² = 73%).
In a subgroup analysis, we did not find statistically significant differences in regaining independent walking between participants who were dependent or independent walkers at the start of the study (Chi² = 4.55, df = 2; P = 0.10).
Post hoc sensitivity analysis by type of electromechanical device
Independent walking at the end of the intervention phase
To examine the robustness of the results and to explore the relationship between independent walking and type of electromechanical device, we compared achieved independent walking rates at the end of the intervention phase by type of electromechanical device.
End‐effector devices
Eleven trials with a total of 598 participants used an end‐effector device as the experimental intervention (Table 8). As stated in the comparisons above, for some of the included trials, no effect estimate (OR) was feasible (Analysis 5.1). The use of electromechanical devices for gait rehabilitation of people after stroke did not increase the chance of walking independently (OR (random‐effects model) 1.90, 95% CI 0.99 to 3.63; P = 0.05; level of heterogeneity I² = 50%).
5.1. Analysis.
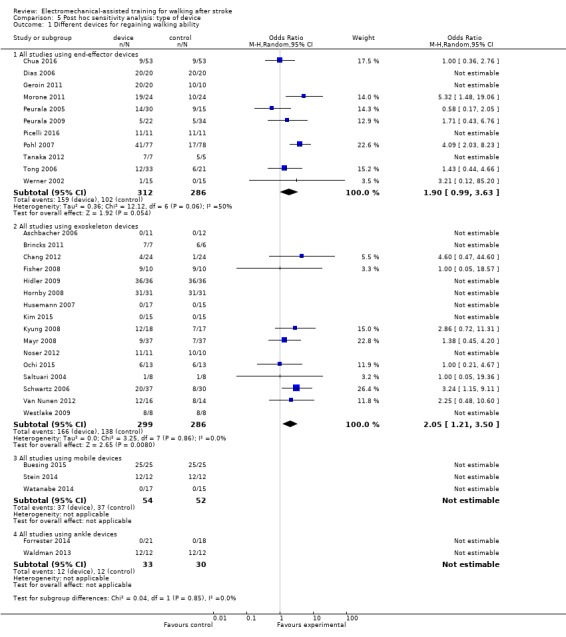
Comparison 5 Post hoc sensitivity analysis: type of device, Outcome 1 Different devices for regaining walking ability.
Exoskeleton devices
Sixteen trials with a total of 585 participants used an exoskeleton device as the experimental intervention (Table 8). The use of electromechanical devices for gait rehabilitation of people after stroke increased the chance of walking independently (OR (random‐effects model) 2.05, 95% CI 1.21 to 3.50; P = 0.008; level of heterogeneity I² = 0%).
We did not find statistically significant differences in regaining independent walking between participants treated with end‐effector or exoskeleton devices (Chi² = 0.04, df = 1; P = 0.85).
Mobile devices
Three trials with a total of 106 participants used powered mobile devices as the experimental intervention (Table 8), but the effects on walking ability were not estimable.
Ankle devices
Two trials with a total of 63 participants used ankle devices while sitting as the experimental intervention (Table 8), but the effects on walking ability were not estimable.
We did not find statistically significant differences in regaining independent walking by type of electromechanical device (end‐effector, exoskeleton, mobile or ankle device)(Chi² = 0.04, df = 1; P = 0.85).
Walking speed at the end of the intervention phase
To examine the robustness of the results and to explore the relationship between independent walking and type of electromechanical device, we compared the walking speed at the end of the intervention phase by type of electromechanical device.
End‐effector devices
Nine trials with a total of 519 participants used an end‐effector device as the experimental intervention and provided data for walking velocity (m/s) at study end (Analysis 5.2). The use of electromechanical devices for gait rehabilitation significantly increased walking velocity. The pooled MD (random‐effects model) for walking velocity was 0.11 m/s (95% CI 0.04 to 0.18; P = 0.003; level of heterogeneity I² = 73%).
5.2. Analysis.
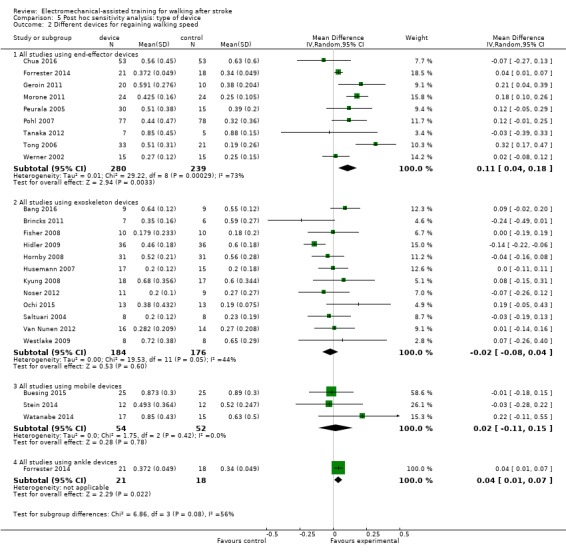
Comparison 5 Post hoc sensitivity analysis: type of device, Outcome 2 Different devices for regaining walking speed.
Exoskeleton devices
Twelve trials with a total of 360 participants used an exoskeleton device as the experimental intervention and provided data for walking velocity (m/s) at study end (Analysis 5.2). The use of electromechanical devices for gait rehabilitation did not increase walking velocity. The pooled MD (random‐effects model) for walking velocity was ‐0.02 m/s (95% CI ‐0.08 to 0.04; P = 0.60; level of heterogeneity I² = 44%).
In a formal subgroup analysis, we found statistically significant differences in improvement in walking velocity between participants treated with an end‐effector device or an exoskeleton device (Chi² = 6.79, df = 1; P = 0.009; I² = 85.3%).
Mobile devices
Three trials with a total of 106 participants used powered mobile devices as the experimental intervention and provided data for walking velocity (m/s) at study end (Analysis 5.2). The use of electromechanical devices for gait rehabilitation did not increase walking velocity. The pooled MD (random‐effects model) for walking velocity was 0.02 m/s (95% CI ‐0.11 to 0.15; P = 0.78; level of heterogeneity I² = 0%).
Ankle devices
One trial with 39 participants used an ankle mobile device as the experimental intervention and provided data for walking velocity (m/s) at study end (Analysis 5.2). The use of electromechanical devices for gait rehabilitation increased walking velocity. The MD (random‐effects model) for walking velocity was 0.04 m/s (95% CI 0.01 to 0.07; P = 0.02; level of heterogeneity not applicable).
In a subgroup analysis, we did not find statistically significant differences in improvement in walking velocity by type of electromechanical device (end‐effector, exoskeleton, mobile or ankle device)(Chi² = 6.56, df = 3; P = 0.09; I² = 54.3%).
Walking capacity at the end of the intervention phase
To examine the robustness of the results and to explore the relationship between independent walking and type of electromechanical device, we compared the walking capacity at the end of the intervention phase by type of electromechanical device.
End‐effector devices
Four trials with a total of 328 participants used an end‐effector device as the experimental intervention and provided data for walking capacity (metres) at study end (Analysis 5.3). The use of electromechanical devices for gait rehabilitation significantly increased walking capacity. The pooled MD (random‐effects model) for walking capacity was 27.5 m (95% CI 3.63 to 51.36; P = 0.02; level of heterogeneity I² = 4%).
5.3. Analysis.
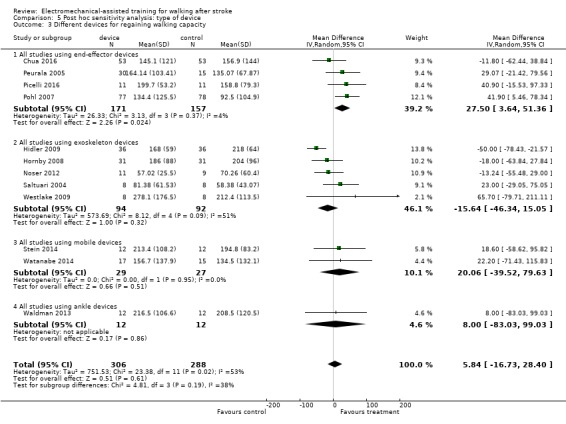
Comparison 5 Post hoc sensitivity analysis: type of device, Outcome 3 Different devices for regaining walking capacity.
Exoskeleton devices
Five trials with a total of 186 participants used an exoskeleton device as the experimental intervention and provided data for walking capacity (metres) at study end (Analysis 5.3). The use of electromechanical devices for gait rehabilitation did not increase walking capacity. The pooled MD (random‐effects model) for walking capacity was ‐15.64 m (95% CI ‐46.34 to 15.05; P = 0.32; level of heterogeneity I² = 51%).
In a formal subgroup analysis, we found statistically significant differences in improvement in walking capacity between participants treated with an end‐effector device or an exoskeleton device (Chi² = 4.73, df = 1; P = 0.03; I² = 78.9%).
Mobile devices
Two trials with a total of 56 participants used powered mobile devices as the experimental intervention and provided data for walking capacity (metres) at study end (Analysis 5.3). The use of electromechanical devices for gait rehabilitation did not increase walking capacity. The pooled MD (random‐effects model) for walking capacity was 20.06 m (95% CI ‐39.52 to 79.63; P = 0.51; level of heterogeneity I² = 0%).
Ankle devices
One trial with 24 participants used an ankle mobile device as the experimental intervention and provided data for walking capacity (metres) at study end (Analysis 5.3). The use of electromechanical devices for gait rehabilitation did not increase walking capacity. The MD (random‐effects model) for walking capacity was 8.0 m (95% CI ‐83.03 to 99.03; P = 0.86; level of heterogeneity not applicable).
In a subgroup analysis, we did not find statistically significant differences in improvement in walking capacity between participants treated by type of electromechanical device (end‐effector, exoskeleton, mobile or ankle device) (Chi² = 4.81, df = 3; P = 0.19; I² = 37.7%).
Discussion
Summary of main results
The aim of this review was to evaluate the effects of electromechanical‐ and robotic‐assisted gait‐training devices (with body weight support) for improving walking after stroke. We sought to estimate the likelihood or chance of becoming independent in walking as a result of these interventions, which is a main rehabilitation goal for people who have had a stroke (Bohannon 1988; Bohannon 1991).
We included 36 trials with a total of 1472 participants and found evidence that the use of electromechanical‐assisted devices in combination with physiotherapy in rehabilitation settings may improve walking function after stroke.
Furthermore, adverse events, dropouts, and deaths do not appear to be more frequent in participants who received electromechanical‐ or robotic‐assisted gait training, which indicates that the use of electromechanical‐assisted gait‐training devices was safe and acceptable to most participants included in the trials analysed by this review.
The exclusion of certain patient groups, such as people over 80 years of age, people with unstable cardiovascular conditions, people with cognitive and communication deficits, and people with a limited range of motion in the lower limb joints at the start of the intervention, may limit the general applicability of the findings. However, using the results from the primary outcomes, it is possible to explore the apparent effectiveness of electromechanical‐assisted devices for regaining walking ability. Of 761 participants in the treatment group, 412 (54%) were walking independently at the end of the intervention phase. We used the primary outcome of independently walking at the end of the intervention phase for all included participants (OR 1.94) to calculate the number needed to treat for an additional beneficial outcome (NNTB). Together with our control event rate of 45% (325 out of 711 control participants were independently walking), we calculated an NNTB of 7 (95% CI 6 to 8) (Sackett 1996). This means that every seventh dependency in walking ability after stroke could be avoided with the use of electromechanical‐assisted devices. However, the optimum amount of electromechanical‐assisted gait training (optimal frequency, optimal duration in the use of assistive technologies and timing of application) remains unclear.
It appears that people in the acute and subacute phases after stroke profit more than people treated more than three months poststroke from this type of therapy (Analysis 3.1). This means that people may benefit more from electromechanical‐ and robotic‐assisted gait training in the first three months after stroke than after three months poststroke.
We argue that 582 (39%) of the 1472 included participants were independently walking at baseline (see Description of studies and the Characteristics of included studies table). Because people who are already ambulatory cannot regain or recover independent walking, our effect estimate could have been influenced by performance bias. We therefore performed two further sensitivity analyses by ambulatory status at the start of the study (Analysis 4.1 and Analysis 4.2).
We found that studies that included mainly dependent walkers (i.e. participants who were non‐ambulatory at the start of the study) were more likely to report that these participants were able to walk at study end and to reach greater walking velocities at the end of the intervention phase compared with participants who were already ambulatory at the start of the study (Analysis 4.1; Analysis 4.2). This means that ambulatory people do not benefit from electromechanical‐ and robotic‐assisted gait training.
We found that the ability to walk at study end was not dependent on the type of device used in the studies (Analysis 5.1). However, walking velocities at the end of the intervention phase were higher when end‐effector devices were used compared with participants who received training by an exoskeleton device (Analysis 5.2), meaning that the type of device used could play a role in improving walking function after stroke. This is in line with the former version of this review from 2013, Mehrholz 2013, and another review that compared the effects of different types of devices on walking ability after stroke (Mehrholz 2012a). However, in the absence of a direct empirical comparison between electromechanical‐assisted gait‐training devices, this point warrants further investigation.
Overall completeness and applicability of evidence
In all systematic reviews the risk of publication bias is present. However, we performed an extensive search for relevant literature in electronic databases and handsearched conference abstracts. Additionally, we contacted and asked authors, trialists, and experts in the field for other unpublished and ongoing trials.
Upon visual inspection, we did not detect graphical evidence of publication bias (see Figure 3 and Figure 4).
3.
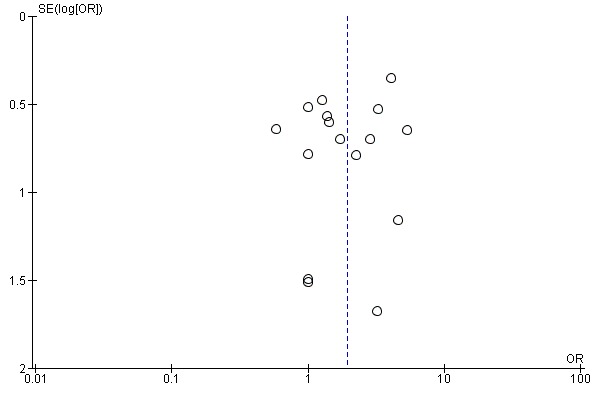
Funnel plot of comparison: 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), outcome: 1.1 Independent walking at the end of intervention phase, all electromechanical devices used.
4.
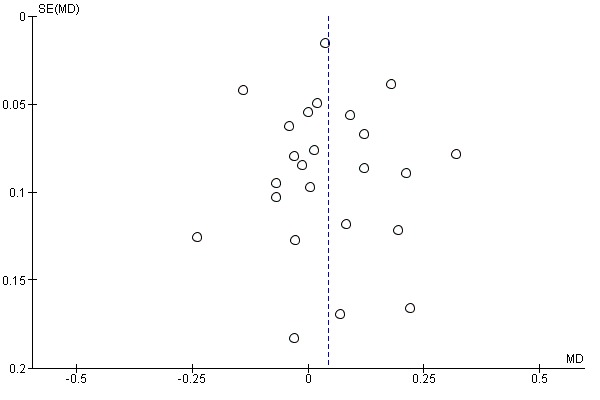
Funnel plot of comparison: 1 Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care), outcome: 1.3 Walking velocity (metres per second) at the end of intervention phase.
Given that we found several ongoing studies of substantial size, it is possible that these ongoing studies could potentially impact our overall conclusion when they are included in the review (see Characteristics of ongoing studies).
It is not clear whether the observed differences between experimental and control groups depend on the intensity of therapy, in terms of repetitions of gait practice. Time devoted to therapy is a crude measure of intensity. A 30‐minute therapy session could include no walking practice or high‐intensity walking practice with many steps taken. Reviews of the effectiveness of arm robotic therapy suggest that the positive benefit of robotic therapy may be lost when the intensity of practice is matched between experimental and control groups (Mehrholz 2012b). However, the numbers of repetitions in the experimental and control groups were not exactly counted in any of the included studies. Further studies should therefore ascertain whether the benefits described here are still apparent when the intensity of gait practice (e.g. step repetitions) is exactly matched between groups.
It should be mentioned that we do not know yet whether these devices provide any cost benefit. Further studies should investigate, under the premise that gait practice is matched in terms of objective measures of intensity, the long‐term costs of regaining walking ability and the cost‐effectiveness of these devices.
Quality of the evidence
There was heterogeneity between the trials in terms of trial design (two arms, three arms, parallel‐group or cross‐over trial, duration of follow‐up, and selection criteria for participants), characteristics of the therapy interventions (especially duration of the intervention), and participant characteristics (length of time since stroke onset and stroke severity at baseline). We noted methodological differences in the mechanisms of randomisation and in the allocation concealment methods used, as well as in the blinding of primary outcomes and the presence or use of intention‐to‐treat analysis.
After examining the effects of methodological quality on the odds of independence in walking, we found that the benefits were relatively robust when we removed trials with an inadequate sequence generation process, inadequate concealed allocation, no blinded assessors for the primary outcome, and incomplete outcome data (Analysis 2.1). However, we found that the odds of independence in walking were slightly lower after the largest included study (Pohl 2007, N = 155) was removed, but a statistically significant and clinically relevant benefit for participants is still observed.
Although the methodological quality of the included trials seemed generally good to moderate (Figure 2), trials investigating electromechanical‐ and robotic‐assisted gait‐training devices are subject to potential methodological limitations. These include inability to blind the therapist and participants, so‐called contamination (provision of the intervention to the control group), and co‐intervention (when the same therapist unintentionally provides additional care to either treatment or comparison group). All these potential methodological limitations introduce the possibility of performance bias. However, as discussed previously, this was not supported in our sensitivity analyses by methodological quality.
The quality of the evidence for automated electromechanical‐ and robotic‐assisted gait‐training devices for improving walking after stroke was moderate. The quality of the evidence was low for walking speed, very low for walking capacity, and low for adverse events and people discontinuing treatment.
Potential biases in the review process
In all systematic reviews the risk of publication bias is present. However, we performed an extensive search for relevant literature in electronic databases and handsearched conference abstracts. Additionally, we contacted and asked authors, trialists, and experts in the field for other unpublished and ongoing trials.
Upon visual inspection, we did not detect graphical evidence of publication bias (see Figure 3 and Figure 4).
Given that we found several ongoing studies of substantial size, it is possible that these ongoing studies could potentially impact our overall conclusion (see Characteristics of ongoing studies).
Agreements and disagreements with other studies or reviews
The most recent and relevant review describes the effects of new so‐called powered mobile solutions (Louie 2016). We included three studies of mobile devices in this update. When pooling these results, we did not find significant improvements in walking speed and walking capacity; this result is in agreement with the recent review of Louie 2016. Additionally, there are two new studies describing the effects of ankle robots (Forrester 2014; Waldman 2013) to improve walking were described. When pooling these studies, we did not find significant improvements in walking speed and walking capacity. We are not aware of any systematic review about this type of devices so far.
Authors' conclusions
Implications for practice.
This systematic review provides moderate‐quality evidence that the use of electromechanical‐assisted gait‐training devices in combination with physiotherapy increases the chance of regaining independent walking ability in people after stroke. These results could be interpreted as preventing one participant from remaining dependent in walking after stroke for every seven treated. However, this apparent benefit for patients is not supported by our secondary outcomes. Gait‐training devices were associated with neither improvements in walking velocity nor walking capacity (low‐ to very low‐quality evidence). It appears that the greatest benefits with regard to independence in walking and walking speed were achieved in participants who were non‐ambulatory at the start of the study and in those for whom the intervention was applied early poststroke.
Implications for research.
There is still a need for well‐designed, large‐scale, multicentre studies to evaluate the benefits and harms of electromechanical‐assisted gait training for walking after stroke, including only non‐ambulatory people in the very early stages after stroke. Comparisons between different devices are also currently lacking. Future research should include estimates of the costs (or savings) associated with electromechanical gait training. Further analyses should investigate whether non‐ambulatory or ambulatory people benefit most, and trials should include outcome measures in the activities of daily living and quality of life domains. In future updates of this review we will consider investigating the effects of different control interventions using subgroup analysis. Additionally, in the next update we will compare the effects of different duration and intensity of treatment (e.g. less than versus more than four weeks; five days per week versus less than five days).
Feedback
Feedback, 30 June 2010
Summary
It appears that the P value for the walking capacity outcome is incorrect in your abstract. The P value is reported as P = 0.073 in the abstract but is reported as P = 0.73 in the results section and in the forest plot.
Reply
The feedback from Meghan Malone‐Moses, above, is accurate. I am sorry for this error which occurred in the abstract. The printed P value in the abstract (P = 0.073) was not correct and has now been changed to P = 0.73 as reported correctly in the Results section and in the forest plot. There is no change to the conclusions because the P value for the walking capacity outcome remains non‐significant.
Contributors
Commenter: Meghan Malone‐Moses, MPH, Medical Writer, DynaMed Responder: Jan Mehrholz
What's new
| Date | Event | Description |
|---|---|---|
| 6 December 2016 | New search has been performed | We have updated the searches to September 2016 and revised the text as appropriate. We have included 36 studies with 1472 participants in this update, compared with 23 trials with 999 participants in the previous version of this review from 2013. |
| 6 December 2016 | New citation required and conclusions have changed | The conclusions of the review have changed. The previous version of this review concluded that, for the primary outcome (walking), the number needed to treat was five patients to prevent one dependency; this updated version of our review concludes that seven patients need to be treated to prevent one dependency in walking. |
History
Protocol first published: Issue 4, 2006 Review first published: Issue 3, 2007
| Date | Event | Description |
|---|---|---|
| 23 January 2013 | New citation required and conclusions have changed | The conclusions of the review have changed. The previous version of this review concluded that, for the primary outcome (walking), the number needed to treat was six patients to prevent one dependency; this updated version of our review concludes that five patients need to be treated to prevent one dependency in walking. |
| 14 January 2013 | New search has been performed | We have updated the searches to December 2012 and revised the text as appropriate. We have included 23 trials with 999 participants in this update, compared with 17 trials with 837 participants in the previous version of this review from 2009. |
| 28 July 2010 | Feedback has been incorporated | Feedback and author response included in the Feedback section, and error corrected in the Abstract. |
| 16 October 2009 | New search has been performed | We have updated the searches to April 2009 and revised the text as appropriate. The conclusions of the review have not changed. We have included 17 trials with 837 participants in this update, as compared with eight trials with 414 participants in the previous version of this review from 2007. The previous version of this review concluded that, for the primary outcome (walking), the number needed to treat was four patients to prevent one dependency; this updated version of our review concludes that six patients need to be treated to prevent one dependency in walking. |
| 6 August 2008 | Amended | Converted to new review format. |
Acknowledgements
We thank Brenda Thomas for help with developing the search strategy and for providing us with relevant trials and systematic reviews from CINAHL, AMED, SPORTDiscus, and Inspec; Hazel Fraser for providing us with relevant information about trials and systematic reviews from the Cochrane Stroke Group Trials Register; and Gabi Voigt for conducting research and for providing us with many helpful studies. We thank Stanley Fisher, Carmen Krewer, Jorge Lians, Andreas Mayer, Stefan Hesse, Joseph Hidler, George Hornby, Yun‐Hee Kim, Zeev Meiner, Sinnika Peurala, Leopold Saltuari, Isabella Schwartz, Raymond Tong, John Brincks, Michael van Nunen, and Naoki Tanaka for providing additional information or unpublished data for their trials.
Appendices
Appendix 1. CENTRAL search strategy
#1MeSH descriptor: [Cerebrovascular Disorders] explode all trees #2MeSH descriptor: [Brain Injuries] this term only #3MeSH descriptor: [Brain Injury, Chronic] this term only #4stroke* or cva or poststroke or post‐stroke #5cerebrovasc* or cerebral vascular #6cerebral or cerebellar or brain* or vertebrobasilar #7infarct* or isch?emi* or thrombo* or emboli* or apoplexy #8#6 and #7 #9cerebral or brain or subarachnoid #10haemorrhage or hemorrhage or haematoma or hematoma or bleed* #11#9 and #10 #12MeSH descriptor: [Hemiplegia] this term only #13MeSH descriptor: [Paresis] explode all trees #14hempar* or hemipleg* or brain injur* #15MeSH descriptor: [Gait Disorders, Neurologic] this term only #16#1 or #2 or #3 or #4 or #5 or #8 or #11 or #12 or #13 or #14 or #15 #17MeSH descriptor: [Physical Therapy Modalities] this term only #18MeSH descriptor: [Exercise Therapy] this term only #19MeSH descriptor: [Motion Therapy, Continuous Passive] this term only #20MeSH descriptor: [Musculoskeletal Manipulations] this term only #21MeSH descriptor: [Exercise] this term only #22MeSH descriptor: [Exercise Test] this term only #23MeSH descriptor: [Robotics] this term only #24MeSH descriptor: [Automation] this term only #25MeSH descriptor: [Orthotic Devices] this term only #26MeSH descriptor: [Man‐Machine Systems] this term only #27MeSH descriptor: [Self‐Help Devices] this term only #28MeSH descriptor: [Therapy, Computer‐Assisted] this term only #29MeSH descriptor: [Body Weight] this term only #30MeSH descriptor: [Weight‐Bearing] this term only #31((gait or locomot*) near/5 (train* or therapy or rehabilitat* or re‐educat* or machine* or powered or device*)) #32(electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device*) #33((body‐weight or body weight) near/3 (support* or relief)) #34(robot* or orthos* or orthotic or automat* or computer aided or computer assisted or power‐assist*) #35(bws or harness or treadmill or exercise* or fitness train* or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator) #36(continuous passive or cpm) near/3 therap* #37#17 or #18 or #19 or #20 or #21 or #22 or #23 or #24 or #25 or #26 or #27 or #28 or #29 or#30 or #31 or #32 or #33 or #34 or #35 or #36 #38MeSH descriptor: [Gait] this term only #39MeSH descriptor: [Walking] explode all trees #40MeSH descriptor: [Locomotion] this term only #41MeSH descriptor: [Range of Motion, Articular] this term only #42MeSH descriptor: [Recovery of Function] this term only #43walk* or gait* or ambulat* or mobil* or locomot* or balanc* or stride #44#38 or #39 or #40 or #41 or #42 or #43 #45#16 and #37 and #44 #46MeSH descriptor: [Animals] explode all trees #47#45 not #46 #48#47 in Trials #49#48 Publication Year from 2012 to 2016
Number of records retrieved until 2012: 1140
Number of records retrieved until 2016: 861
Appendix 2. MEDLINE search strategy (OvidSP)
1. exp cerebrovascular disorders/ or brain injury, chronic/
2. (stroke$ or cva or poststroke or post‐stroke).tw.
3. (cerebrovasc$ or cerebral vascular).tw.
4. (cerebral or cerebellar or brain$ or vertebrobasilar).tw.
5. (infarct$ or isch?emi$ or thrombo$ or emboli$ or apoplexy).tw.
6. 4 and 5
7. (cerebral or brain or subarachnoid).tw.
8. (haemorrhage or hemorrhage or haematoma or hematoma or bleed$).tw.
9. 7 and 8
10. hemiplegia/ or exp paresis/
11. (hempar$ or hemipleg$ or brain injur$).tw.
12. Gait Disorders, Neurologic/
13. 1 or 2 or 3 or 6 or 9 or 10 or 11 or 12
14. physical therapy modalities/ or exercise therapy/ or motion therapy, continuous passive/ or musculoskeletal manipulations/
15. *exercise/ or *exercise test/
16. robotics/ or automation/ or orthotic devices/ or man‐machine systems/ or self‐help devices/ or therapy, computer‐assisted/
17. body weight/ or weight‐bearing/
18. ((gait or locomot$) adj5 (train$ or therapy or rehabilitat$ or re‐educat$ or machine$ or powered or device$)).tw.
19. (electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device$).tw.
20. ((body‐weight or body weight) adj3 (support$ or relief)).tw.
21. (robot$ or orthos$ or orthotic or automat$ or computer aided or computer assisted or power‐assist$).tw.
22. (bws or harness or treadmill or exercise$ or fitness train$ or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator).tw.
23. ((continuous passive or cpm) adj3 therap$).tw.
24. or/14‐23
25. gait/ or exp walking/ or locomotion/
26. "Range of Motion, Articular"/
27. recovery of function/
28. (walk$ or gait$ or ambulat$ or mobil$ or locomot$ or balanc$ or stride).tw.
29. or/25‐28
30. 13 and 24 and 29
31. Randomized Controlled Trials as Topic/
32. random allocation/
33. Controlled Clinical Trials as Topic/
34. control groups/
35. clinical trials as topic/ or clinical trials, phase i as topic/ or clinical trials, phase ii as topic/ or clinical trials, phase iii as topic/ or clinical trials, phase iv as topic/
36. double‐blind method/
37. single‐blind method/
38. Placebos/
39. placebo effect/
40. cross‐over studies/
41. Therapies, Investigational/
42. Research Design/
43. evaluation studies as topic/
44. randomized controlled trial.pt.
45. controlled clinical trial.pt.
46. (clinical trial or clinical trial phase i or clinical trial phase ii or clinical trial phase iii or clinical trial phase iv).pt.
47. (evaluation studies or comparative study).pt.
48. random$.tw.
49. (controlled adj5 (trial$ or stud$)).tw.
50. (clinical$ adj5 trial$).tw.
51. ((control or treatment or experiment$ or intervention) adj5 (group$ or subject$ or patient$)).tw.
52. (quasi‐random$ or quasi random$ or pseudo‐random$ or pseudo random$).tw.
53. ((multicenter or multicentre or therapeutic) adj5 (trial$ or stud$)).tw.
54. ((control or experiment$ or conservative) adj5 (treatment or therapy or procedure or manage$)).tw.
55. ((singl$ or doubl$ or tripl$ or trebl$) adj5 (blind$ or mask$)).tw.
56. (coin adj5 (flip or flipped or toss$)).tw.
57. versus.tw.
58. (cross‐over or cross over or crossover).tw.
59. placebo$.tw.
60. sham.tw.
61. (assign$ or alternate or allocat$ or counterbalance$ or multiple baseline).tw.
62. or/31‐61
63. 30 and 62
64. exp animals/ not humans.sh.
65. 63 not 64
66. limit 65 to ed=20120719‐20160815
Number of records retrieved until 2012: 1928
Number of records retrieved until 2016: 1094
Appendix 3. Embase search strategy (OvidSP)
1. cerebrovascular disease/ or exp basal ganglion hemorrhage/ or exp brain hematoma/ or exp brain hemorrhage/ or exp brain infarction/ or exp brain ischemia/ or exp carotid artery disease/ or cerebral artery disease/ or exp cerebrovascular accident/ or exp intracranial aneurysm/ or exp occlusive cerebrovascular disease/ or stroke patient/
2. (stroke or poststroke or post‐stroke or cerebrovasc$ or brain vasc$ or cerebral vasc$ or cva$ or apoplex$ or SAH).tw.
3. ((brain$ or cerebr$ or cerebell$ or intracran$ or intracerebral) adj5 (isch?emi$ or infarct$ or thrombo$ or emboli$ or occlus$)).tw.
4. ((brain$ or cerebr$ or cerebell$ or intracerebral or intracranial or subarachnoid) adj5 (haemorrhage$ or hemorrhage$ or haematoma$ or hematoma$ or bleed$)).tw.
5. hemiparesis/ or hemiplegia/ or paresis/
6. (hemipleg$ or hemipar$ or paresis or paretic or hemineglect or hemi‐neglect or ((unilateral or spatial or hemi?spatial or visual) adj5 neglect)).tw.
7. Gait Disorders, Neurologic/
8. or/1‐7
9. physical therapy modalities/ or exercise therapy/ or motion therapy, continuous passive/ or musculoskeletal manipulations/
10. *exercise/ or *exercise test/
11. robotics/ or automation/ or orthotic devices/ or man‐machine systems/ or self‐help devices/ or therapy, computer‐assisted/
12. body weight/ or weight‐bearing/
13. ((gait or locomot$) adj5 (train$ or therapy or rehabilitat$ or re‐educat$ or machine$ or powered or device$)).tw.
14. (electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device$).tw.
15. ((body‐weight or body weight) adj3 (support$ or relief)).tw.
16. (robot$ or orthos$ or orthotic or automat$ or computer aided or computer assisted or power‐assist$).tw.
17. (bws or harness or treadmill or exercise$ or fitness train$ or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator).tw.
18. ((continuous passive or cpm) adj3 therap$).tw.
19. or/9‐18
20. gait/ or exp walking/ or locomotion/
21. "Range of Motion, Articular"/
22. recovery of function/
23. (walk$ or gait$ or ambulat$ or mobil$ or locomot$ or balanc$ or stride).tw.
24. or/20‐23
25. 19 and 24
26. Randomized Controlled Trial/ or "randomized controlled trial (topic)"/
27. Randomization/
28. Controlled clinical trial/ or "controlled clinical trial (topic)"/
29. control group/ or controlled study/
30. clinical trial/ or "clinical trial (topic)"/ or phase 1 clinical trial/ or phase 2 clinical trial/ or phase 3 clinical trial/ or phase 4 clinical trial/
31. Crossover Procedure/
32. Double Blind Procedure/
33. Single Blind Procedure/ or triple blind procedure/
34. placebo/ or placebo effect/
35. (random$ or RCT or RCTs).tw.
36. (controlled adj5 (trial$ or stud$)).tw.
37. (clinical$ adj5 trial$).tw.
38. ((control or treatment or experiment$ or intervention) adj5 (group$ or subject$ or patient$)).tw.
39. (quasi‐random$ or quasi random$ or pseudo‐random$ or pseudo random$).tw.
40. ((control or experiment$ or conservative) adj5 (treatment or therapy or procedure or manage$)).tw.
41. ((singl$ or doubl$ or tripl$ or trebl$) adj5 (blind$ or mask$)).tw.
42. (cross‐over or cross over or crossover).tw.
43. (placebo$ or sham).tw.
44. trial.ti.
45. (assign$ or allocat$).tw.
46. controls.tw.
47. or/26‐46
48. 8 and 25 and 47
49. (exp animals/ or exp invertebrate/ or animal experiment/ or animal model/ or animal tissue/ or animal cell/ or nonhuman/) not (human/ or normal human/ or human cell/)
50. 48 not 49
51. limit 50 to dd=20120719‐20160815
Number of records retrieved until 2012: 2315
Number of records retrieved until 2016: 1601
Appendix 4. CINAHL search strategy (via EBSCOHost)
S1 .(MH "Cerebrovascular Disorders") OR (MH "Basal Ganglia Cerebrovascular Disease+") OR (MH "Carotid Artery Diseases+") OR (MH "Cerebral Ischemia+") OR (MH "Cerebral Vasospasm") OR (MH "Intracranial Arterial Diseases+") OR (MH "Intracranial Embolism and Thrombosis") OR (MH "Intracranial Hemorrhage+") OR (MH "Stroke") OR (MH "Vertebral Artery Dissections")
S2 .(MH "Stroke Patients") OR (MH "Stroke Units")
S3 .TI ( stroke or poststroke or post‐stroke or cerebrovasc* or brain vasc* or cerebral vasc or cva or apoplex or SAH ) or AB ( stroke or poststroke or post‐stroke or cerebrovasc* or brain vasc* or cerebral vasc or cva or apoplex or SAH )
S4 .TI ( brain* or cerebr* or cerebell* or intracran* or intracerebral ) or AB ( brain* or cerebr* or cerebell* or intracran* or intracerebral )
S5 .TI ( ischemi* or ischaemi* or infarct* or thrombo* or emboli* or occlus* ) or AB ( ischemi* or ischaemi* or infarct* or thrombo* or emboli* or occlus* )
S6 .S4 and S5
S7 .TI ( brain* or cerebr* or cerebell* or intracerebral or intracranial or subarachnoid ) or AB ( brain* or cerebr* or cerebell* or intracerebral or intracranial or subarachnoid )
S8 .TI ( haemorrhage* or hemorrhage* or haematoma* or hematoma* or bleed* ) or AB ( haemorrhage* or hemorrhage* or haematoma* or hematoma* or bleed* )
S9 .S7 and S8
S10 .(MH "Hemiplegia")
S11 .TI ( hemipleg* or hemipar* or paresis or paretic ) or AB ( hemipleg* or hemipar* or paresis or paretic )
S12 .S1 OR S2 OR S3 OR S6 OR S9 OR S10 OR S11
S13 .(MM "PhysicalTherapy") OR (MM "TherapeuticExercise") OR (MM "Motion Therapy, Continuous Passive") OR (MM "Robotics") OR (MM "Automation") OR (MM "Orthoses") OR (MM "Assistive Technology Devices") OR (MM "Therapy, Computer Assisted") OR (MM "Body Weight") OR (MM "Weight‐Bearing")
S14 *exercise or (*exercise test)
S15 "man‐machine systems"
S16 ((gait or locomot*) N5 (train* or therapy or rehabilitat* or re‐educat* or machine* or powered or device*))
S17 electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device*
S18 ((body‐weight or body weight) N3 (support* or relief))
S19 robot* or orthos* or orthotic or automat* or computer aided or computer assisted or power‐assist*
S20 bws or harness or treadmill or exercise* or fitness train* or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator
S21 (continuous passive or cpm) N3 therap*
S22 S13 OR S14 OR S15 OR S16 OR S17 OR S18 OR S19 OR S20 OR S21
S23 (MM "Gait")
S24 (MH "Walking+")
S25 (MM "Locomotion")
S26 (MM "Range of Motion")
S27 (MM "Recovery")
S28 walk* or gait* or ambulat* or mobil* or locomot* or balanc* or stride
S29 S23 OR S24 OR S25 OR S26 OR S27 OR S28
S30 S22 AND S29
S31 .(MH "Randomized Controlled Trials") or (MH "Random Assignment") or (MH "Random Sample+")
S32 .(MH "Clinical Trials") or (MH "Intervention Trials") or (MH "Therapeutic Trials")
S33 .(MH "Double‐Blind Studies") or (MH "Single‐Blind Studies") or (MH "Triple‐Blind Studies")
S34 .(MH "Control (Research)") or (MH "Control Group") or (MH "Placebos") or (MH "Placebo Effect")
S35 .(MH "Crossover Design") OR (MH "Quasi‐Experimental Studies")
S36 .PT (clinical trial or randomized controlled trial)
S37 .TI (random* or RCT or RCTs) or AB (random* or RCT or RCTs)
S38 .TI (controlled N5 (trial* or stud*)) or AB (controlled N5 (trial* or stud*))
S39 .TI (clinical* N5 trial*) or AB (clinical* N5 trial*)
S40 .TI ((control or treatment or experiment* or intervention) N5 (group* or subject* or patient*)) or AB ((control or treatment or experiment* or intervention) N5 (group* or subject* or patient*))
S41 .TI ((control or experiment* or conservative) N5 (treatment or therapy or procedure or manage*)) or AB ((control or experiment* or conservative) N5 (treatment or therapy or procedure or manage*))
S42 .TI ((singl* or doubl* or tripl* or trebl*) N5 (blind* or mask*)) or AB ((singl* or doubl* or tripl* or trebl*) N5 (blind* or mask*))
S43 .TI (cross‐over or cross over or crossover) or AB (cross‐over or cross over or crossover)
S44 .TI (placebo* or sham) or AB (placebo* or sham)
S45 .TI trial
S46 .TI (assign* or allocat*) or AB (assign* or allocat*)
S47 .TI controls or AB controls
S48 .TI (quasi‐random* or quasi random* or pseudo‐random* or pseudo random*) or AB (quasi‐random* or quasi random* or pseudo‐random* or pseudo random*)
S49 .S31 OR S32 OR S33 OR S34 OR S35 OR S36 OR S37 OR S38 OR S39 OR S40 OR S41 OR S42 OR S43 OR S44 OR S45 OR S46 OR S47 OR S48
S50 .S12 AND S30 AND S49
S51 .EM 201207‐
S52 .S50 AND S51
Number of records retrieved until 2012: 787
Number of records retrieved until 2016: 382
Appendix 5. AMED search strategy (OvidSP)
1. cerebrovascular disorders/ or cerebral hemorrhage/ or cerebral infarction/ or cerebral ischemia/ or cerebrovascular accident/ or stroke/
2. (stroke or poststroke or post‐stroke or cerebrovasc$ or brain vasc$ or cerebral vasc$ or cva$ or apoplex$ or SAH).af.
3. ((brain$ or cerebr$ or cerebell$ or intracran$ or intracerebral) adj5 (isch?emi$ or infarct$ or thrombo$ or emboli$ or occlus$)).af.
4. ((brain$ or cerebr$ or cerebell$ or intracerebral or intracranial or subarachnoid) adj5 (haemorrhage$ or hemorrhage$ or haematoma$ or hematoma$ or bleed$)).af.
5. hemiplegia/
6. (hemipleg$ or hemipar$ or paresis or paretic or hemineglect or hemi‐neglect or ((unilateral or spatial or hemi?spatial or visual) adj5 neglect)).af.
7. or/1‐6
8. physical therapy modalities/ or exercise therapy/ or motion therapy, continuous passive.mp or musculoskeletal manipulations/
9. exercise/ or exercise test/
10. robotics/ or orthotic devices/ or myoelectric prosthesis/ or therapy, computer‐assisted/
11. body weight/ or weight‐bearing/
12. ((gait or locomot$) adj5 (train$ or therapy or rehabilitat$ or re‐educat$ or machine$ or powered or device$)).af.
13. (electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device$).af.
14. ((body‐weight or body weight) adj3 (support$ or relief)).af.
15. (robot$ or orthos$ or orthotic or automat$ or computer aided or computer assisted or power‐assist$).af.
16. (bws or harness or treadmill or exercise$ or fitness train$ or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator).af.
17. ((continuous passive or cpm) adj3 therap$).af.
18. or/8‐17
19. gait/ or exp walking/ or locomotion/
20. "Range of Motion"/
21. recovery of function/
22. (walk$ or gait$ or ambulat$ or mobil$ or locomot$ or balanc$ or stride).af.
23. or/19‐22
24. 7 and 18 and 23
25. exp animals/ not humans.sh.
26. 24 not 25
27. limit 26 to up=201207‐201609
Number of records retrieved until 2012: 383
Number of records retrieved until 2016: 392
Appendix 6. Web of Science search strategy
#1.TS=(stroke or poststroke or post‐stroke or cerebrovasc* or brain vasc* or cerebral vasc* or cva* or apoplex* or SAH)
#2.TS=((brain* or cerebr* or cerebell* or intracran* or intracerebral) NEAR/5 (isch$emi* or infarct* or thrombo* or emboli* or occlus*))
#3.TS=((brain* or cerebr* or cerebell* or intracran* or intracerebral) NEAR/5 (h$emorrhage* or h$ematoma* or bleed*))
#4.TS=(hemipleg* or hemipar* or paresis or paretic)
#5.#4 OR #3 OR #2 OR #1
#6.TS=(Neurologic NEAR/5 Gait NEAR/5 Disorders)
#7.TS= (robotics or automation or orthotic devices or man‐machine system* or self‐help devices or (computer‐assisted NEAR/5 therapy))
#8.TS=((gait or locomot*) NEAR/5 (train* or therapy or rehabilitat* or re‐educat* or machine* or powered or device$))
#9.TS=(electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device$)
#10.TS=((body‐weight or (body NEAR/3 weight)) NEAR/3 (support$ or relief))
#11.TS=(robot* or orthos* or orthotic or automat* or computer aided or computer assisted or power‐assist*)
#12.TS=(bws or harness or treadmill or exercise$ or fitness train* or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator)
#13.TS=(((continuous NEAR/3 passive) or cpm) NEAR/3 therap*)
#14. #6 or #7 or #8 or #9 or #10 or #11 or #12 or #13
#15.TS=((gait or walk* or locomot* or ambulat* or mobil* or balance* or stride) or (range of motion) or recovery of function)
#16.#14 and #15
#17.TS=(random* or RCT or RCTs)
#18.TS=(controlled NEAR/5 (trial$ or stud*))
#19.TS=(clinical* NEAR/5 trial*)
#20.TS=((control or treatment or experiment* or intervention) NEAR/5 (group$ or subject$ or patient$))
#21.TS=(quasi‐random* or quasi random* or pseudo‐random* or pseudo random*)
#22.TS=((control$ or experiment* or conservative) NEAR/5 (treatment or therapy or manage* or procedure))
#23.TS=((singl$ or doubl* or tripl* or trebl*) NEAR/5 (blind* or mask*))
#24.TS=(cross‐over or cross over or crossover)
#25.TS=(placebo* or sham)
#26.TI=trial
#27.TS=(assign* or allocat*)
#28.TS=controls
#29.#17 or #18 or #19 or #20 or #21 or #22 or #23 or #24 or #25 or #26 or #27 or #28
#30.#5 and #16 and #29
Number of records retrieved until 2016: 3806
Appendix 7. PEDro search strategy
Abstract&Title: Stroke AND gait Method: Clinical Trial
Published since: 2012 All other fields not mentioned here have been left blank.
Number of records retrieved until 2012: 165
Number of records retrieved until 2016: 166
Appendix 8. COMPENDEX search strategy (via DIALOG)
1. CEREBROVASCULAR (W) DISORDER? ?/TI,AB,DE 2. BRAIN (W) INJUR???/TI,AB,DE 3. CHRONIC (W) BRAIN (W) INJUR???/TI,AB,DE 4. (STROKE? OR CVA OR POSTSTROKE? OR CEREBROVASC?)/TI,AB,DE 5. CEREBRAL (W) VASCULAR/TI,AB,DE 6. (CEREBRAL OR CEREBELLAR OR BRAIN? OR 7. (INFARCT? OR ISCHAEMI? OR ISCHEMI? OR THROMBO? OR 8. S6 AND S7 9. (CEREBRAL OR BRAIN OR SUBARACHNOID)/TI,AB,DE 10. (HAEMORRHAGE OR HEMORRHAGE OR HAEMATOMA OR HEMATOMA OR 11. S9 AND S10 12. (HEMIPLEG? OR HEMIPAR? OR PARESIS)/TI,AB,DE 13. BRAIN (W) INJUR???/TI,AB,DE 14. GAIT (W) DISORDER? ?/TI,AB,DE 15. NEUROLOGIC??/TI,AB,DE 16. S14 AND S15 17. S1 OR S2 OR S3 OR S4 OR S5 OR S8 OR S16 OR S11‐S13 18. PHYSICAL (W) THERAPY (W) MODALIT???/TI,AB,DE 19. EXERCISE (W) THERAP?/TI,AB,DE 20. MOTION (W) THERAP? /TI,AB,DE 21. CONTINUOUS (3W) PASSIVE (3W) MOTION (3W) THERAP? 22. EXERCISE?/TI,AB,DE 23. (ROBOTICS OR AUTOMATION) /TI,AB,DE 24. ORTHOTIC (W) DEVICE? ?/TI,AB,DE 25. BODY (W) WEIGHT/TI,AB,DE 26. WEIGHT (W) BEARING/TI,AB,DE 27. GAIT (5N) TRAIN???/TI,AB,DE 28. GAIT (5N) THERAP?/TI,AB,DE 29. GAIT (5N) REHABILITAT?/TI,AB,DE 30. GAIT (5N) EDUCAT?/TI,AB,DE 31. LOCOMOT? (5N) TRAIN?/TI,AB,DE 32. LOCOMOT? (5N) THERAP?/TI,AB,DE 33. LOCOMOT? (5N) REHABILITAT?/TI,AB,DE 34. LOCOMOT? (5N) EDUCAT?/TI,AB,DE 35. (ELECTROMECHANICAL OR MECHANICAL OR MECHANI?ED OR 36. ((BODY (W) WEIGHT (3N) SUPPORT?))/TI,AB,DE 37. ((BODY (W) WEIGHT (3N) RELIEF))/TI,AB,DE 38. (ROBOT? OR ORTHOS? OR ORTHOTIC OR AUTOMAT?)/TI,AB,DE 39. COMPUTER (W) AIDED/TI,AB,DE 40. (COMPUTER (W) ASSISTED)/TI,AB,DE 41. (BWS OR HARNESS OR TREADMILL OR LO?OMAT OR GAITRAINER 42. FITNESS (W) TRAIN?/TI,AB,DE 43. CONTINUOUS (W) PASSIVE/TI,AB,DE 44. THERAP?/TI,AB,DE 45. S43 AND S44 46. CPM (3N) THERAP?/TI,AB,DE 47. S18‐S42 48. S48 OR S45 OR S46 49. (GAIT OR WALKING OR LOCOMOTION)/TI,AB,DE 50. RANGE (1W) MOTION/TI,AB,DE 51. ARTICULAR/TI,AB,DE 52. S51 AND S52 53. RECOVERY (3N) FUNCTION/TI,AB,DE 54. (WALK? OR GAIT? ? OR AMBULAT? OR MOBIL? OR LOCOMOT? OR 55. S50 OR S53 OR S54 OR S55 56. S17 AND S49 AND S56 57. S57 AND HUMAN
Number of records retrieved until 2012: 701
Appendix 9. SPORTDiscus search strategy (via EBSCOHost)
S1 .DE "CEREBROVASCULAR disease" OR DE "BRAIN ‐‐ Hemorrhage" OR DE "CEREBRAL embolism & thrombosis" S2 .DE "CEREBROVASCULAR disease ‐‐ Patients" S3 .DE "HEMIPLEGIA" OR DE "HEMIPLEGICS" S4 .TI ( stroke* or poststroke* cva* or cerebrovascular* or cerebral vascular ) OR AB ( stroke* or poststroke* cva* or cerebrovascular* or cerebral vascular ) S5 .TI ( cerebral or cerebellar or brain* or vertebrobasilar ) OR AB ( cerebral or cerebellar or brain* or vertebrobasilar ) S6 .TI ( infarct* or ischemi* or ischaemi* or thrombo* or emboli* or apoplexy ) OR AB ( infarct* or ischemi* or ischaemi* or thrombo* or emboli* or apoplexy ) S7 .S5 and S6 S8 .TI ( cerebral or brain or subarachnoid ) OR AB ( cerebral or brain or subarachnoid ) S9 .TI ( haemorrhage or hemorrhage or haematoma or hematoma or bleed* ) OR AB ( haemorrhage or hemorrhage or haematoma or hematoma or bleed* ) S10 .S8 and S9 S11 .TI ( hempar$ or hemipleg* or brain injur* ) OR AB ( hempar$ or hemipleg* or brain injur* ) S12 .DE "GAIT disorders" S13 .S1 or S2 or S3 or S4 or S7 or S10 or S11 or S12 S14 .DE "PHYSICAL therapy" S15 .DE "EXERCISE" OR DE "LEG exercises" OR DE "STRENGTH training" OR DE "TREADMILL exercise" S16 .DE "EXERCISE therapy" S17 .DE "MANIPULATION (Therapeutics)" S18 .TI ( gait or locomot* ) OR AB ( gait or locomot* ) S19 .TI ( train* or therapy or rehabilitat* or re‐educat* or machine* or powered or device* ) OR AB ( train* or therapy or rehabilitat* or re‐educat* or machine* or powered or device* ) S20 .S18 and S19 S21 .TI ( electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device* ) OR AB ( electromechanical or electro‐mechanical or mechanical or mechanised or mechanized or driven or assistive device* ) S22 .TI ( body‐weight or body weight ) OR AB ( body‐weight or body weight ) S23 .TI ( support* or relief ) OR AB ( support* or relief ) S24 .S22 and S23 S25 .TI ( robot* or orthos* or orthotic or automat* or computer aided or computer assisted or power‐assist* ) OR AB ( robot* or orthos* or orthotic or automat* or computer aided or computer assisted or power‐assist* ) S26 .TI ( bws or harness or treadmill or exercise* or fitness train* or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator ) OR AB ( bws or harness or treadmill or exercise* or fitness train* or Lokomat or Locomat or GaiTrainer or GT1 or Kinetron or Haptic Walker or Anklebot or LOPES or AutoAmbulator ) S27 .TI ( continuous passive or cpm ) OR AB ( continuous passive or cpm ) S28 .TI Therapy OR AB therapy S29 .S27 and S28 S30 .S14 or S15 or S16 or S17 or S20 or S21 or S24 or S25 or S26 or S29 S31 .DE "WALKING" OR DE "FITNESS walking" OR DE "GAIT in humans" S32 .DE "LOCOMOTION" OR DE "HUMAN locomotion" S33 .DE "JOINTS ‐‐ Range of motion" S34 .TI ( walk* or gait* or ambulat* or mobil* or locomot* or balanc* or stride ) OR AB ( walk* or gait* or ambulat* or mobil* or locomot* or balanc* or stride ) S35 .S31 or S32 or S33 or S34 S36 .S13 and S30 and S35 S37 .TI ( random* or RCT or trial* or placebo* or sham or double‐blind* or single‐blind or control or controls or assign* or allocat* ) OR AB ( random* or RCT or trial* or placebo* or sham or double‐blind* or single‐blind or control or controls or assign* or allocat* ) S38 .S36 and S37
Number of records retrieved until 2012: 461
Appendix 10. Inspec search strategy (TecFinder)
Robot AND gait AND stroke
publication year: 2012 ‐ 2016
Number of records retrieved until 2012: 81
Number of records retrieved until 2016: 20
Appendix 11. World Health Organization International Clinical Trials Registry Platform (WHO ICTRP)
Robot AND gait AND stroke
Robot AND Walking AND stroke
publication year: 2012 ‐ 2016
Number of records retrieved until 2016: 10
Data and analyses
Comparison 1. Electromechanical‐ and robotic‐assisted gait training plus physiotherapy versus physiotherapy (or usual care).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Independent walking at the end of intervention phase, all electromechanical devices used | 36 | 1472 | Odds Ratio (M‐H, Random, 95% CI) | 1.94 [1.39, 2.71] |
| 2 Recovery of independent walking at follow‐up after study end | 6 | 496 | Odds Ratio (M‐H, Random, 95% CI) | 1.93 [0.72, 5.13] |
| 3 Walking velocity (metres per second) at the end of intervention phase | 24 | 985 | Mean Difference (IV, Random, 95% CI) | 0.04 [‐0.00, 0.09] |
| 4 Walking velocity (metres per second) at follow‐up | 9 | 578 | Mean Difference (IV, Random, 95% CI) | 0.07 [‐0.05, 0.19] |
| 5 Walking capacity (metres walked in 6 minutes) at the end of intervention phase | 12 | 594 | Mean Difference (IV, Random, 95% CI) | 5.84 [‐16.73, 28.40] |
| 6 Walking capacity (metres walked in 6 minutes) at follow‐up | 7 | 463 | Mean Difference (IV, Random, 95% CI) | ‐0.82 [‐32.17, 30.53] |
| 7 Acceptability of electromechanical‐assisted gait training devices during intervention phase: dropouts | 36 | 1472 | Odds Ratio (M‐H, Random, 95% CI) | 0.67 [0.43, 1.05] |
| 8 Death from all causes until the end of intervention phase | 36 | 1472 | Risk Difference (M‐H, Random, 95% CI) | 0.00 [‐0.01, 0.02] |
Comparison 2. Planned sensitivity analysis by trial methodology.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Regaining independent walking ability | 36 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 All studies with adequate sequence generation process | 20 | 949 | Odds Ratio (M‐H, Random, 95% CI) | 1.80 [1.06, 3.08] |
| 1.2 All studies with adequate concealed allocation | 17 | 831 | Odds Ratio (M‐H, Random, 95% CI) | 1.87 [1.12, 3.12] |
| 1.3 All studies with blinded assessors for primary outcome | 16 | 762 | Odds Ratio (M‐H, Random, 95% CI) | 1.81 [1.10, 2.98] |
| 1.4 All studies without incomplete outcome data | 14 | 590 | Odds Ratio (M‐H, Random, 95% CI) | 2.23 [1.16, 4.29] |
| 1.5 All studies excluding the largest study Pohl 2007 | 35 | 1317 | Odds Ratio (M‐H, Random, 95% CI) | 1.65 [1.17, 2.34] |
Comparison 3. Subgroup analysis comparing participants in acute and chronic phases of stroke.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Independent walking at the end of intervention phase, all electromechanical devices used | 36 | Odds Ratio (IV, Random, 95% CI) | Subtotals only | |
| 1.1 Acute phase: less than or equal to 3 months after stroke | 20 | 1143 | Odds Ratio (IV, Random, 95% CI) | 1.90 [1.38, 2.63] |
| 1.2 Chronic phase: more than 3 months after stroke | 16 | 461 | Odds Ratio (IV, Random, 95% CI) | 1.20 [0.40, 3.65] |
Comparison 4. Post hoc sensitivity analysis: ambulatory status at study onset.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Recovery of independent walking: ambulatory status at study onset | 36 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 Studies that included independent walkers | 15 | 500 | Odds Ratio (M‐H, Random, 95% CI) | 1.38 [0.45, 4.20] |
| 1.2 Studies that included dependent and independent walkers | 9 | 340 | Odds Ratio (M‐H, Random, 95% CI) | 1.90 [1.11, 3.25] |
| 1.3 Studies that included dependent walkers | 12 | 632 | Odds Ratio (M‐H, Random, 95% CI) | 1.90 [1.04, 3.48] |
| 2 Walking velocity: ambulatory status at study onset | 24 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 2.1 Studies that included independent walkers | 10 | 317 | Mean Difference (IV, Random, 95% CI) | ‐0.02 [‐0.10, 0.06] |
| 2.2 Studies that included dependent and independent walkers | 5 | 146 | Mean Difference (IV, Random, 95% CI) | 0.03 [‐0.05, 0.11] |
| 2.3 Studies that included dependent walkers | 9 | 522 | Mean Difference (IV, Random, 95% CI) | 0.10 [0.03, 0.17] |
Comparison 5. Post hoc sensitivity analysis: type of device.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Different devices for regaining walking ability | 32 | Odds Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 1.1 All studies using end‐effector devices | 11 | 598 | Odds Ratio (M‐H, Random, 95% CI) | 1.90 [0.99, 3.63] |
| 1.2 All studies using exoskeleton devices | 16 | 585 | Odds Ratio (M‐H, Random, 95% CI) | 2.05 [1.21, 3.50] |
| 1.3 All studies using mobile devices | 3 | 106 | Odds Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 1.4 All studies using ankle devices | 2 | 63 | Odds Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Different devices for regaining walking speed | 24 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 2.1 All studies using end‐effector devices | 9 | 519 | Mean Difference (IV, Random, 95% CI) | 0.11 [0.04, 0.18] |
| 2.2 All studies using exoskeleton devices | 12 | 360 | Mean Difference (IV, Random, 95% CI) | ‐0.02 [‐0.08, 0.04] |
| 2.3 All studies using mobile devices | 3 | 106 | Mean Difference (IV, Random, 95% CI) | 0.02 [‐0.11, 0.15] |
| 2.4 All studies using ankle devices | 1 | 39 | Mean Difference (IV, Random, 95% CI) | 0.04 [0.01, 0.07] |
| 3 Different devices for regaining walking capacity | 12 | 594 | Mean Difference (IV, Random, 95% CI) | 5.84 [‐16.73, 28.40] |
| 3.1 All studies using end‐effector devices | 4 | 328 | Mean Difference (IV, Random, 95% CI) | 27.50 [3.64, 51.36] |
| 3.2 All studies using exoskeleton devices | 5 | 186 | Mean Difference (IV, Random, 95% CI) | ‐15.64 [‐46.34, 15.05] |
| 3.3 All studies using mobile devices | 2 | 56 | Mean Difference (IV, Random, 95% CI) | 20.06 [‐39.52, 79.63] |
| 3.4 All studies using ankle devices | 1 | 24 | Mean Difference (IV, Random, 95% CI) | 8.0 [‐83.03, 99.03] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Aschbacher 2006.
| Methods | RCT Method of randomisation: not stated Blinding of outcome assessors: stated as 'yes' by the investigator Adverse events: not stated Deaths: not stated Dropouts: 4 (1 in treatment group, 3 in control group) ITT: unclear | |
| Participants | Country: Switzerland 23 participants (12 in treatment group, 11 in control group) Not ambulatory at start of study Mean age: 57 to 67 years (control and treatment groups, respectively) Inclusion criteria: ≤ 3 months after stroke, ability to stand or walk 5 metres Exclusion criteria: orthopaedic problems, contractures, NYHA III‐IV | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 3 weeks and 6 months Primary outcomes: walking velocity, step length, endurance, walking ability (FAC) Secondary outcomes: isometric knee extension strength, patient acceptance and satisfaction (visual analogue scale) | |
| Notes | Unpublished data | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of randomisation not described |
| Allocation concealment (selection bias) | Unclear risk | Not described |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Unclear |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Bang 2016.
| Methods | RCT Method of randomisation: opaque, closed envelopes Blinding of outcome assessors: stated as 'yes' by the investigator Adverse events: not stated Deaths: not stated Dropouts: no |
|
| Participants | Country: Korea
18 participants (9 in treatment group, 9 in control group)
Ambulatory at start of study
Mean age: 54 years (control and treatment group) Inclusion criteria: > 6 months after stroke, ability to walk over 10 meters, gait speed > 0.4 m/s, MMSE ≥ 24 Exclusion criteria: uncontrolled health condition, comorbidity or disability other than stroke precluding gait training |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 2 weeks. Outcome measures: gait speed, cadence, step length, double support period (GAITRite system), balance, level of balance confidence (ABC scale) |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The randomisation was performed by selection of an opaque, closed envelope in which the group assignment was written, which was given to the physical therapist. |
| Allocation concealment (selection bias) | Low risk | By sealed envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Described as blinded by an assessor not participating in study |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No dropouts |
Brincks 2011.
| Methods | Randomised cross‐over trial Method of randomisation: shuffled envelopes Blinding of outcome assessors: no Adverse events: none Deaths: none Dropouts: none ITT: yes | |
| Participants | Country: Denmark 13 participants (7 in treatment group, 6 in control group) All participants were ambulatory at start of study Mean age: 59 to 61 years (control and treatment groups, respectively) Inclusion criteria: unknown Exclusion criteria: unknown | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline, after 3 and 6 weeks Primary outcomes: single support stance time in impaired extremity and gait asymmetry and swing time ratio Secondary outcomes: walking speed | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Shuffling envelopes |
| Allocation concealment (selection bias) | Low risk | Using sealed, shuffled envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No blinding was done. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | ITT |
Buesing 2015.
| Methods | RCT Method of randomisation: random number generator Blinding of outcome assessors: stated as 'yes' by the investigator Adverse events: no adverse events Deaths: not stated Dropouts: none |
|
| Participants | Country: USA
50 participants (25 in treatment group, 25 in control group)
Ambulatory at start of study
Mean age: 62 years experimental group and 60 years control group Inclusion criteria: > 12 months after stroke, medically stable, initial gait speed between 0.4 and 0.8 m/s, > 17 MMSE, sit unsupported for 30 s, walk ≥ 10 m with maximum 1 person, follow a 3‐step command |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after visit 10 and 18, 3‐month follow‐up Outcome measures: gait velocity, cadence, step time, step length, stride length, swing time, stance time, double support time (GAITRite) | |
| Notes | NCT01994395 | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Method of randomisation described as "random number generator". |
| Allocation concealment (selection bias) | High risk | Allocation concealment not described. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Evaluated by a research physical therapist, who was blinded to the participant’s training group |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No dropouts |
Chang 2012.
| Methods | RCT Method of randomisation: not stated Blinding of outcome assessors: not stated Adverse events: not stated Deaths: not stated Dropouts: 3 (2 in experimental group, 1 in control group) ITT analysis: not described | |
| Participants | Country: Republic of Korea 48 allocated participants (24 in treatment group, 24 in control group) 38 participants were non‐ambulatory at start of study Mean age: 58 years Inclusion criteria: first‐ever stroke, stroke onset within 1 month, supratentorial lesion, age > 20 years and < 65 years, not an independent ambulator (FAC < 2), and ability to co‐operate during exercise testing Exclusion criteria: people who met criteria for absolute and relative contraindications to exercise testing established by the American College of Sports Medicine (ACSM) were excluded. Also, people who met contraindications for Lokomat therapy or musculoskeletal disease involving the lower limbs, such as severe painful arthritis, osteoporosis, or joint contracture and other neurological diseases, were also excluded | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after training:
|
|
| Notes | This study describes the same study protocol and participants as described in the study Kim 2008, but provides further explanation of participant characteristics; the ID Chang 2012 therefore replaces the formerly review used ID Kim 2008. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of randomisation is unclear. |
| Allocation concealment (selection bias) | Unclear risk | Method of concealment is unclear. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Cho 2015.
| Methods | RCT, cross‐over
Method of randomisation: not stated
Blinding of outcome assessors: not stated
Adverse events: not stated
Deaths: not stated
Dropouts: not stated ITT: not stated |
|
| Participants | Country: Korea
20 participants (13 in treatment group, 7 in control group)
Not ambulatory at start of study
Mean age: 55 years in control and treatment group Inclusion criteria: onset period of > 6 months, FAC < 2, independent ambulation before stroke, ability to understand and execute RAGT, no orthopaedic or neurosurgical problems in the lower extremities Exclusion criteria: weight > 120 kg; femoral length < 35 cm or > 47 cm; history of low‐extremity fracture after stroke, instability or subluxation of the hip joint, or pressure ulcers on the hips or lower extremities; any underlying disease preventing execution of RAGT |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 4 and 8 weeks:
|
|
| Notes | Higher dose of intervention in experimental group compared to control group; group differences at baseline (modified forward reach) | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of randomisation not described. |
| Allocation concealment (selection bias) | High risk | Allocation not described. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not mentioned |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Chua 2016.
| Methods | RCT
Method of randomisation: computer‐generated sequence
Blinding of outcome assessors: stated as 'yes' by the investigator
Adverse events: no
Deaths: yes
Dropouts: yes (7 in treatment group, 13 in control group) ITT: yes |
|
| Participants | Country: Singapore
108 participants (53 in treatment group, 53 in control group)
Not ambulatory at start of study
Mean age: 62 years experimental and 61 years control group Inclusion criteria: unilateral haemorrhagic/ischaemic stroke, age between 18 and 80 years, independent ambulation pre‐stroke Exclusion criteria: > 8 weeks poststroke, FAC ≥ 4, cardiovascular instability, MMSE < 16, communication deficits, lower limb joint contractures |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and 4, 8, 12, 24, and 48 weeks after baseline Outcome measures: walking ability (FAC), Barthel Index, gait velocity (10‐metre walk test), gait endurance (6‐minute walk test), health status (Stroke Impact Scale) |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Authors state: "Randomization was performed using a computer‐generated sequence of random numbers." |
| Allocation concealment (selection bias) | Low risk | Authors state: "An independent department generated the random group allocation sequence and transferred the sequence to a series of serially numbered opaque envelopes, which were not opened and revealed until after acceptance into the study and the baseline tests, therefore ensuring allocation concealment." |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Authors state: "The data assessors were blinded to group allocation, but it was not possible to blind participants or the health care professionals providing interventions." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All data for all participants provided and analysed. Authors state: "An intention‐to‐treat approach was used. Data from subjects were analysed according to the group to which they were randomised, regardless of whether they completed the intervention. Participants failing to complete either intervention were asked to return for follow‐up." |
Dias 2006.
| Methods | RCT Method of randomisation: permuted block randomisation Blinding of outcome assessors: stated as blinded Adverse events: none stated Deaths: none Dropouts: none ITT: not stated but probably done because there were no dropouts | |
| Participants | Country: Portugal 40 participants (20 in treatment group, 20 in control group) Ambulatory at start of study Mean age: 69 years Inclusion criteria: first‐ever stroke patients > 12 months after stroke; age > 18 and < 80 years; cognitive (MMSE > 19) and communication capacities to understand the treatment; absence of cardiac, psychological, and orthopaedic contraindications Exclusion criteria: not stated | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 4 weeks and 3 months:
After study end and at follow‐up, participants rated satisfaction with and efficiency of treatment in a self questionnaire (Likert scale). |
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Unclear |
| Allocation concealment (selection bias) | Low risk | Central allocation |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not done |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing outcome data |
Fisher 2008.
| Methods | RCT Method of randomisation: blocked randomisation Blinding of outcome assessors: stated as 'yes' Adverse events: control group 14, experimental group 11 Deaths: none Dropouts: none ITT: stated as 'yes' | |
| Participants | Country: USA 20 participants (10 in treatment group, 10 in control group) 5 in treatment group and 7 in control group were ambulatory at start of study Mean age: not stated Inclusion criteria: subacute, < 2 months after stroke Exclusion criteria: not stated | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 24 sessions:
|
|
| Notes | This trial is described as ongoing; the results of the first 20 participants were reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Unclear |
| Allocation concealment (selection bias) | Unclear risk | Stated as concealed, but method not described. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Yes |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | ITT stated. |
Forrester 2014.
| Methods | RCT
Method of randomisation: not described
Blinding of outcome assessors: stated as 'no'
Adverse events: no
Deaths: not mentioned
Dropouts: 5 (3 in treatment group, 2 in control group) ITT: no |
|
| Participants | Country: USA
39 participants (21 in treatment group, 18 in control group)
Not ambulatory at start of study
Mean age: 63 years in experimental group and 60 years in control group Inclusion criteria: first stroke; residual lower extremity hemiparesis involving the ankle (1/5 to 4/5 MRC); capable of generating at least trace muscle activation in PF‐DF; adequate language and neurocognitive function; sit in the chair for 30 to 60 minutes per session of ankle training Exclusion criteria: total plegia (0/5) at paretic ankle; fixed or painful contractures; dementia; orthopaedic, arthritic, or inflammatory condition limiting ankle movement; deep venous thrombosis or pulmonary thromboembolism; vision impairment; severe receptive or global aphasia |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and at discharge. Outcome measures: walking ability (Functional Independence Measure walking), balance (Berg Balance Scale), walking velocity, active range of motion, muscle strength, spatiotemporal gait parameters (step time, step length, step symmetry), motor control variables (angular velocity, co‐ordination) |
|
| Notes | Unclear amount of therapy | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not clearly described; authors only state "blocked randomisation" |
| Allocation concealment (selection bias) | High risk | Not described |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Authors state "not blinded". |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 5 participants were excluded from analysis after randomisation. |
Geroin 2011.
| Methods | RCT Method of randomisation: software‐generated randomisation scheme Blinding of outcome assessors: no Adverse events: none Deaths: none Dropouts: none ITT: yes | |
| Participants | Country: Italy 30 participants (10 in treatment group 1, 10 in treatment group 2, and 10 in control group) 5 in both treatment groups and 7 in control group were ambulatory at start of study Mean age: not stated Inclusion criteria: at least 12 months from their first unilateral ischaemic stroke; age < 75 years; European Stroke Scale score between 75 and 85; MMSE score ≧ 24; ability to maintain standing position without aids for at least 5 minutes; ability to walk independently for at least 15 metres with the use of walking aids (cane and orthoses) Exclusion criteria: preceding epileptic fits; an electroencephalography suspect of elevated cortical excitability; metallic implants within the brain and previous brain surgery; medications altering cortical excitability or with a presumed effect on brain plasticity; posterior circulation stroke; deficits of somatic sensation involving the paretic lower limb; presence of vestibular disorders or paroxysmal vertigo; presence of severe cognitive or communicative disorders; presence of other neurological or orthopaedic conditions involving the lower limbs; presence of cardiovascular comorbidity; performance of any type of rehabilitation treatment in the 3 months before start of study | |
| Interventions | 3 arms:
All participants received 10 x 50‐minute treatment sessions, 5 days a week, for 2 consecutive weeks |
|
| Outcomes | Outcomes were recorded at baseline and after 2 weeks:
|
|
| Notes | We combined the results of both robotic‐assisted groups (arms 1 and 2) into a single group, which we compared with the results of the control group (arm 3). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Software‐generated list |
| Allocation concealment (selection bias) | Low risk | Central allocation |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not done |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing outcome data |
Han 2016.
| Methods | RCT
Method of randomisation: not stated
Blinding of outcome assessors: stated as 'yes'
Adverse events: none
Deaths: none
Dropouts: 4 in control group (2 refused and 2 dropped) ITT: no |
|
| Participants | Country: Republic of Korea 60 participants (30 in treatment group, 30 in control group) Non‐ambulatory at start of study Mean age: 68 years in experimental group and 63 years in control group Inclusion criteria: clinical diagnosis of stroke < 3 months after stroke onset, first‐ever stroke, dependent ambulation with severe gait impairment (FAC < 2), and sufficient cognition to understand procedures and provide informed consent Exclusion criteria: contraindications for RAGT therapy; cerebellar or brainstem lesions that could affect autonomic or balance function; musculoskeletal disease involving the lower limbs, such as severe painful arthritis, osteoporosis, amputation, or joint contracture; and other concurrent neurological diseases (e.g. Parkinson's disease, multiple sclerosis, etc.) |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after the 4‐week intervention; all outcome parameters were measured within 3 days before and after 20 sessions of training:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of randomisation not described. |
| Allocation concealment (selection bias) | Unclear risk | Allocation concealment not described. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | A physiatrist (rehabilitation doctor) who remained blinded to participant group and treatment throughout the entire study analysed outcome measures. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | No ITT |
Hidler 2009.
| Methods | RCT Method of randomisation: randomisation table Blinding of outcome assessors: not described Adverse events: control group 14; experimental group 11 Deaths: 1, which study arm not reported Dropouts: 9 ITT: no, described as analysis per protocol | |
| Participants | Country: USA 72 participants (36 in treatment group, 36 in control group); 63 participants completed all training sessions and were analysed as per protocol All participants were ambulatory at start of study Mean age: 60 years Inclusion criteria: hemiparesis resulting from unilateral ischaemic or haemorrhagic stroke, time since stroke less than 6 months, no prior stroke, age > 18 years, ability to ambulate 5 metres without physical assistance and a self selected walking speed between 0.1 and 0.6 m/s, not receiving any other physical therapy targeting the lower limbs Exclusion criteria: severe osteoporosis, contractures limiting range of motion in the lower extremities, not ambulating before stroke, severe cardiac disease (NYHA classification of II‐IV), uncontrolled hypertension (systolic > 200 mm Hg, diastolic > 110 mm Hg), stroke of the brainstem or cerebellar lesions, uncontrolled seizures, presence of lower limb non‐healing ulcers, lower limb amputation, uncontrolled diabetes, cognitive deficits (< 24 on the MMSE), symptoms of depression (≥ 16 on the Center for Epidemiological Studies Depression Scale) | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 12 and 24 sessions, and at 3‐month follow‐up:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation table |
| Allocation concealment (selection bias) | Unclear risk | Not described in sufficient detail |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not done |
| Incomplete outcome data (attrition bias) All outcomes | High risk | No ITT, analysis per protocol |
Hornby 2008.
| Methods | RCT Method of randomisation: opaque, sealed envelopes Blinding of outcome assessors: not done Adverse events: 8 events in control group and 3 events in experimental group Deaths: none Dropouts: 14 (10 in control group and 4 in experimental group) ITT: no ITT; analysis per protocol | |
| Participants | Country: USA 62 participants (31 in treatment group, 31 in control group), 48 participants completed all training sessions and were analysed as per protocol All participants were ambulatory at start of study Mean age: 57 years Inclusion criteria: hemiparesis of longer than 6 months' duration after patients with unilateral, supratentorial, ischaemic, or haemorrhage stroke were recruited; no evidence of bilateral or brainstem lesions; able to walk 10 metres overground without physical assistance at speeds of 0.8 m/s at self selected velocity, using assistive devices and bracing below the knee as needed Exclusion criteria: significant cardiorespiratory/metabolic disease or other neurological or orthopaedic injury that may limit exercise participation or impair locomotion, size limitations for the harness/counterweight system or robotic orthosis, botulinum toxin therapy in the lower limbs within 6 months before enrolment, scores lower than 23 on the MMSE, patients could not receive concurrent physical therapy | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 12 sessions and at 6‐month follow‐up:
|
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | (Probably) shuffling envelopes |
| Allocation concealment (selection bias) | Low risk | Opaque, sealed envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No blinding |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 'As‐treated' analysis done |
Husemann 2007.
| Methods | RCT Method of randomisation: opaque envelopes, stratified by side of paresis and aetiology Blinding of outcome assessors: yes Adverse events: 2 (1 in experimental group, 1 in control group) Deaths: none Dropouts: 2 (1 in experimental group, 1 in control group) ITT analysis: not provided for all dropouts | |
| Participants | Country: Germany 32 participants (17 in treatment group, 15 in control group) Non‐ambulatory at start of study Mean age: not provided Inclusion criteria: not provided Exclusion criteria: not provided | |
| Interventions | 2 arms:
Both groups received additional 30 minutes of physiotherapy daily. |
|
| Outcomes | Outcomes were recorded at baseline and after 4 weeks:
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Random numbers generated by a computer program, block randomisation. |
| Allocation concealment (selection bias) | Low risk | Sealed, opaque envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Evaluating therapist blinded for group allocation. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Insufficient reporting of attrition/exclusions to permit judgement of ‘low risk’ or ‘high risk’ |
Kim 2015.
| Methods | RCT
Method of randomisation: not described
Blinding of outcome assessors: not described
Adverse events: not mentioned
Deaths: none
Dropouts: 4 (2 in experimental group, 2 in control group) ITT: no |
|
| Participants | Country: Korea
30 participants (15 in treatment group, 15 in control group)
Not ambulatory at start of study
Mean age: 54 years in experimental group and 50 years in control group Inclusion criteria: first stroke < 1 year, plateau in recovery of the locomotor functions after a 30‐day conventional neurorehabilitation Exclusion criteria: severe spasticity (Modified Ashworth Scale 2), tremor, severe visual and cognitive impairments, musculoskeletal diseases, cardiopulmonary diseases, body weight of 135 kg; height of 150 cm |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 4 and 8 weeks. Outcome measures: walking ability (FAC), balance (Berg Balance Scale), modified Barthel Index, spasticity (Modified Ashworth Scale), quality of life (EuroQol‐5 dimension) |
|
| Notes | NCT02053233 | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method not described. |
| Allocation concealment (selection bias) | Unclear risk | Unclear |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not described |
| Incomplete outcome data (attrition bias) All outcomes | High risk | No ITT |
Kyung 2008.
| Methods | RCT Method of randomisation: unclear Blinding of outcome assessors: unclear Adverse events: unclear Deaths: unclear Dropouts: 3 (2 in experimental group, 1 in control group) ITT analysis: unclear | |
| Participants | Country: Republic of Korea 35 participants (18 in treatment group, 17 in control group) 10 participants in the experimental group and 7 participants in the control group were ambulatory at start of study Mean age: not stated Inclusion criteria: not stated Exclusion criteria: not stated | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after training
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Unclear |
| Allocation concealment (selection bias) | Unclear risk | Method neither described nor stated. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Unclear |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Mayr 2008.
| Methods | RCT Method of randomisation: unclear Blinding of outcome assessors: unclear Adverse events: not stated Deaths: unclear, probably none Dropouts: 13 (4 in experimental group, 9 in control group) ITT analysis: not stated | |
| Participants | Country: Austria 74 participants (37 in treatment group, 37 in control group) Most participants in both groups were non‐ambulatory at start of study Mean age: not stated Inclusion criteria: primary ischaemic lesion of the medial cerebral artery, between 10 days and 6 weeks after stroke, stable cardiovascular system, ability to walk with assistance of 1 therapist Exclusion criteria: brainstem lesions, thrombosis, severe contractures, good walking ability with standing only help by therapist | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after training phase:
|
|
| Notes | Published as conference abstract and unpublished data | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Software‐generated list |
| Allocation concealment (selection bias) | Low risk | Described concealed allocation |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Described as blinded evaluator |
| Incomplete outcome data (attrition bias) All outcomes | High risk | No ITT |
Morone 2011.
| Methods | RCT Method of randomisation: by computer program Blinding of outcome assessors: stated as 'yes' Adverse events: control group 4, experimental group 3 Deaths: none Dropouts: (defined in this study as discontinued intervention) 12 in robotic groups and 9 in control groups ITT: yes | |
| Participants | Country: Italy
48 participants (24 in treatment group , 24 in control group )
All participants were non‐ambulatory at start of study
Mean age: 62 years
Inclusion criteria: hemiplegia/hemiparesis in the subacute phase with significant gait deficits (FAC < 3) caused by a first‐ever stroke, lesions that were confirmed by computed tomography or magnetic resonance imaging, and age between 18 and 80 years Exclusion criteria: presence of subarachnoid haemorrhages, sequelae of prior cerebrovascular accidents or other chronic disabling pathologies, orthopaedic injuries that could impair locomotion, spasticity that limited lower extremity range of motion to less than 80%, sacral skin lesions, MMSE score < 24, and hemispatial neglect, as evaluated by a neuropsychologist |
|
| Interventions | 2 arms (including strata for motor function):
The standard physiotherapy, shared by both groups, was focused on facilitation of movement on the paretic side and upper limb exercises, as well as improving balance, standing, sitting, and transferring. |
|
| Outcomes | Outcomes were recorded by a physician who was blinded to the treatment at baseline, after 4 weeks of the intervention, and at hospital discharge:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Generated electronically by www.random.org |
| Allocation concealment (selection bias) | Low risk | Central allocation |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Yes |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | ITT done; missing outcome data balanced in numbers across intervention groups, with similar reasons for missing data across groups. |
Noser 2012.
| Methods | RCT Method of randomisation: unclear Blinding of outcome assessors: stated as 'yes' Adverse events: 4 (2 in experimental group and 2 in control group) Deaths: none stated Dropouts: 1 in the control group (protocol violation) ITT: stated as 'yes' | |
| Participants | Country: USA 21 participants (11 in treatment group, 10 in control group); 20 participants completed all training sessions, and 11 in treatment group and 9 in control group completed the study and were analysed as per protocol All participants were ambulatory at start of study Mean age: unclear Inclusion criteria: people with ischaemic or haemorrhagic stroke confirmed by cerebral CT or MRI scan; age > 18; at least 3 months' poststroke at time of enrolment into study; ability to walk at least 10 feet with maximum 1 person assist, but not to walk in the community independently; residual paresis in the lower extremity as defined by NIHSS lower extremity motor score 2 to 4; ability to perform Lokomat ambulation training with assistance of 1 therapist; ability to follow a 3‐step command; physician approval for patient participation; ability to give informed consent, completed rehabilitation services (i.e. not receiving concurrent physical, occupational, or speech therapy) Exclusion criteria: serious cardiac condition, uncontrolled blood pressure defined as > 200 or diastolic > 100 at rest, history of serious chronic obstructive pulmonary disease or oxygen dependence, severe weight‐bearing pain, lower extremity amputation, claudication while walking, life expectancy < 1 year, history of deep vein thrombosis or pulmonary embolism within 6 months, severe orthopaedic problem, any medical or psychiatric condition that the investigators believe would prevent participation in study | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and at postintervention, 3 months' postintervention
|
|
| Notes | NCT00975156; same study as Kelley 2013 | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Unclear |
| Allocation concealment (selection bias) | Unclear risk | Unclear |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Authors state: "Blinded assessors tested the participants at baseline, post‐intervention, and 3‐month follow‐up." |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | 1 participant in the control group was not analysed. |
Ochi 2015.
| Methods | RCT Method of randomisation: random number table Blinding of outcome assessors: stated as 'yes' Adverse events: none Deaths: none Dropouts: none |
|
| Participants | Country: Japan
26 participants (13 in treatment group, 13 in control group)
Not ambulatory at start of study
Mean age: 62 years in experimental group and 65 years in control group Inclusion criteria: first‐ever stroke < 5 weeks; age between 40 and 85 years; lower extremities Brunnstrom's recovery stage ≤ grade III; FAC ≤ 2; independence in walking before stroke Exclusion criteria: height between 145 and 180 cm; body weight over 100 kg; limitation in range of motion in the lower extremity; severe cardiovascular, respiratory, renal, or musculoskeletal disease; difficulty in communicating |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 4 weeks. Outcome measures: Fugl‐Meyer Assessment lower extremity, muscle torque, walking ability (FAC), 10‐metre walk test, Functional Independence Measure |
|
| Notes | Clinical group differences in FAC at baseline | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method of randomisation not described. |
| Allocation concealment (selection bias) | Unclear risk | No allocation concealment described. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Described as blinded assessors |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No dropouts, no participants excluded from analysis. |
Peurala 2005.
| Methods | RCT Method of randomisation: an investigator not involved in the study randomly assigned participants to groups using concealed envelopes Blinding of outcome assessors: no Adverse events: none Deaths: none Dropouts: none ITT analysis: not stated | |
| Participants | Country: Finland 45 participants (15 in treatment group A, 15 in treatment group B, 15 in control group) Ambulatory and non‐ambulatory at study onset Mean age: 52 years Inclusion criteria: first supratentorial stroke with duration of illness longer than 6 months, younger than 65 years of age, slow or difficult walking, no unstable cardiovascular disease, no severe malposition of joints, no severe cognitive or communicative disorders, written informed consent Exclusion criteria: not stated | |
| Interventions | 3 arms:
All participants practised gait for 15 sessions over 3 weeks (each session lasting 20 minutes) and received an additional 55 minutes daily physiotherapy. |
|
| Outcomes | Outcomes were recorded at baseline and after 2 and 3 weeks and 6 months:
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | An investigator not involved in the study randomly assigned participants to groups using concealed envelopes. |
| Allocation concealment (selection bias) | Low risk | Concealed envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not done |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear if reasons for missing outcome data are unlikely to be related to true outcome |
Peurala 2009.
| Methods | RCT Method of randomisation: sealed envelopes (stratified according to ability to walk) Blinding of outcome assessors: no Adverse events: 2 in treatment group A, 3 in control group Deaths: 1 in control group Dropouts: 5 in treatment group A, 1 in treatment group B, 3 in control group ITT analysis: not stated | |
| Participants | Country: Finland 56 participants (22 in treatment group A, 21 in treatment group B, 13 in control group) Non‐ambulatory at start of study Mean age: 68 years Inclusion criteria: first supratentorial stroke or no significant disturbance from an earlier stroke (Modified Rankin Scale 0 to 2), acute phase after stroke with a maximum duration of 10 days, FAC 0 to 3, voluntary movement in the leg of the affected side, Barthel Index 25 to 75 points, age 18 to 85 years, no unstable cardiovascular disease, body mass index < 32, no severe malposition of joints, no severe cognitive or communicative disorders Exclusion criteria: not stated | |
| Interventions | Between June 2003 and December 2004, random allocation to 2 arms took place (2 walking exercise groups)
All participants received 55 minutes daily gait‐oriented physiotherapy and additional gait training for 15 sessions over 3 weeks (each session lasting maximum of 20 minutes of walking). Between January 2005 and February 2007, random allocation to 3 arms took place (3 walking exercise groups)
All participants received 55 minutes daily gait‐oriented physiotherapy and additional gait training for 15 sessions over 3 weeks (each session lasting maximum of 20 minutes of walking). However, CT‐Group received 1 or 2 physiotherapy sessions daily but not at the same intensity as in the other groups. |
|
| Outcomes | Outcomes were recorded at baseline and after 3 weeks and 6 months:
|
|
| Notes | Because we were interested in the effects of automated electromechanical‐ and robotic‐assisted gait‐training devices for improving walking after stroke, we combined the results of the CT‐Group and the WALK‐Group as one group, which we compared with the results from the GT‐Group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | An investigator not involved in the study randomly assigned participants to groups using concealed envelopes. |
| Allocation concealment (selection bias) | Low risk | Allocation was performed by an independent person not otherwise involved with the participants. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear whether reasons for missing outcome data are unlikely to be related to true outcome |
Picelli 2016.
| Methods | RCT Method of randomisation: software generated Blinding of outcome assessors: yes Adverse events: none Deaths: none Dropouts: none ITT analysis: no dropouts | |
| Participants | Country: Italy 22 participants (11 in treatment group, 11 in control group) Ambulatory at study onset Mean age: 63 years Inclusion criteria: age > 18 years, leg spasticity, FAC > 4, duration of illness > 6 months Exclusion criteria: participation in other trials, deformities such as contractures, spasticity treatment before the study | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline, 1 month Primary outcome:
Secondary outcomes:
|
|
| Notes | 1 group received additional gait training (performance bias). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Software‐generated random order |
| Allocation concealment (selection bias) | Low risk | Sealed, opaque envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Blinded evaluator |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No dropouts |
Pohl 2007.
| Methods | RCT Method of randomisation: lots indicating A or B were prepared in sealed envelopes Blinding of outcome assessors: primary outcomes were evaluated by blinded assessors Adverse events: 4 (3 in experimental group, 1 in control group) Deaths: 2 (1 in experimental group, 1 in control group) Dropouts: 11 (5 in experimental group, 6 in control group) ITT analysis: yes | |
| Participants | Country: Germany 155 participants (77 in treatment group, 78 in control group) Non‐ambulatory at study onset Mean age: 63 years Inclusion criteria: first supratentorial stroke (ischaemic or haemorrhagic); age between 18 and 79 years; interval between stroke and study onset less than 60 days; able to sit unsupported (i.e. without holding onto supports such as the edge of the bed), with feet supported, could not walk at all, or required the help of 1 or 2 therapists irrespective of the use of an ankle‐foot orthosis or a walking aid (FAC 3 or less); understanding the meaning of the study and following instructions, providing written informed consent to participation in the study approved by the local ethical committee Exclusion criteria: unstable cardiovascular condition after a 12‐lead electrocardiogram examined by a cardiologist, restricted passive range of motion in the major lower limb joints (extension deficit > 20° for the affected hip or knee joints, or a dorsiflexion deficit > 20° for the affected ankle, tested while lying supine and on the non‐affected side), prevalence of other neurological or orthopaedic diseases impairing walking ability | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 4 weeks and 6 months. Primary outcomes:
Participants who were ambulatory (FAC 4 or 5) or reaching a Barthel Index > 75 were defined as responders to therapy. Secondary outcomes:
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Lots indicating A or B were prepared in sealed envelopes; a person not involved in the study allocated participants to groups using the concealed envelopes. |
| Allocation concealment (selection bias) | Low risk | Concealed envelopes |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Blinded primary outcomes (a person not involved in the study rated videotapes of participants) |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | ITT analysis done; missing outcome data balanced in numbers across intervention groups, with similar reasons for missing data across groups. |
Saltuari 2004.
| Methods | Cross‐over RCT Method of randomisation: by random numbers Blinding of outcome assessors: unclear Adverse events: none Deaths: none Dropouts: none ITT: yes | |
| Participants | Country: Austria 16 participants (8 in treatment group, 8 in control group) Ambulatory and non‐ambulatory at study onset Mean age: 61 years Inclusion criteria: not provided Exclusion criteria: not provided | |
| Interventions | 2 arms (A: Lokomat, B: physiotherapy):
|
|
| Outcomes | Outcomes were recorded at baseline and after 3 weeks (they were additionally recorded after 6 and 9 weeks, but only outcomes of the first phase were included in this review). | |
| Notes | Unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | By random numbers |
| Allocation concealment (selection bias) | Unclear risk | Unclear |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Unclear |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing outcome data |
Schwartz 2006.
| Methods | RCT Method of randomisation: block sampling method (each block contained 6 participants: 4 experimental group and 2 control group) Blinding of outcome assessors: no Adverse events: 5 (3 in experimental group, 2 in control group) Deaths: none Dropouts: 11 (8 in experimental group, 3 in control group) ITT: no (stated, but 2 participants from the control group were excluded from analysis) | |
| Participants | Country: Israel 67 participants (at October 2006) (37 in treatment group, 30 in control group) Non‐ambulatory at study onset Mean age: 60 years Inclusion criteria: first stroke, until 3 months after stroke Exclusion criteria: not provided | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 3, 6, and 9 weeks:
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Block sampling |
| Allocation concealment (selection bias) | Unclear risk | Unclear |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not done |
| Incomplete outcome data (attrition bias) All outcomes | High risk | For dichotomous outcome data, the proportion of missing outcomes compared with observed event risk was enough to induce clinically relevant bias into intervention effect estimate. |
Stein 2014.
| Methods | RCT
Method of randomisation: not stated
Blinding of outcome assessors: stated as 'yes'
Adverse events: none
Deaths: none
Dropouts: none immediately after study end (at 3‐month follow‐up: 2 in experimental group and 2 in control group) ITT: no |
|
| Participants | Country: USA
24 participants (12 in treatment group, 12 in control group)
Ambulatory at start of study
Mean age: 58 years in experimental group and 57 years in control group Inclusion criteria: single stroke, significant leg weakness and gait alterations < 6 months before study entry, independent in household ambulation Exclusion criteria: ongoing physical therapy for the leg and/or gait and mobility, botulinum toxin injections < 3 months before study entry, no further planned injections, other neurologic disorders, excessive spasticity of lower limb (Ashworth Scale > 3), uncontrolled hypertension, unstable coronary artery disease, contractures of lower limb, impaired cognition (MMSE score < 24) |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline, after 6, 10, and 19 weeks Outcome measures: Timed Up and Go Test, 10‐metre walk test, 6‐minute walk test, Five‐Times‐Sit‐to‐Stand Test, Berg Balance Scale, California Functional Evaluation, Emory Functional Ambulation Profile |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method unclear |
| Allocation concealment (selection bias) | Unclear risk | Unclear, not described |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Performed by 1 physical therapist blinded to group assignment |
| Incomplete outcome data (attrition bias) All outcomes | High risk | No ITT |
Tanaka 2012.
| Methods | Cross‐over RCT Method of randomisation: computer‐generated randomisation Blinding of outcome assessors: no Adverse events: none described Deaths: none Dropouts: none ITT: no | |
| Participants | Country: Japan 12 participants (7 in treatment group, 5 in control group) All were ambulatory at study onset Mean age: 62 years Inclusion criteria: first stroke; more than 6 months since stroke onset; slight‐to‐moderate motor deficit (Brunnstrom recovery stages III–VI); could walk with or without walking aids Exclusion criteria: higher brain function disorder or cognitive deficit affecting ability to understand and describe symptoms (< 24 on the MMSE); severe heart disorder affecting gait movement intensity; severe bone and joint disease affecting gait movement | |
| Interventions | 2 arms (only the first 12 weeks before cross‐over are described here; A: no training, B: gait training with Gait Master 4, 1 session: 20 minutes):
|
|
| Outcomes | Outcomes were recorded weekly over a 24‐week period:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation |
| Allocation concealment (selection bias) | Unclear risk | Unclear |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Tong 2006.
| Methods | RCT Randomisation was done by computer‐generated random numbers Blinding of outcome assessors: no (except for Barthel Index and Functional Independence Measure scores, which were performed by nurses who were blinded) Adverse events: 2 (none in experimental group, 2 in control group) Deaths: none Dropouts: 4 (none in experimental group, 4 in control group) ITT: yes | |
| Participants | Country: Hong Kong, China 50 participants (15 in treatment group A, 15 in treatment group B, 20 in control group) Non‐ambulatory at study onset Mean age: 68 years Inclusion criteria: diagnosis of first ischaemic brain injury or intracerebral haemorrhage shown by magnetic resonance imaging or computed tomography less than 6 weeks after onset of stroke; sufficient cognition to follow simple instructions and understand the content and purpose of the study (MMSE score > 21); ability to stand upright, supported or unsupported, for 1 minute; significant gait deficit (FAC score < 3); no skin allergy to electrical stimulation Exclusion criteria: recurrent stroke; other neurological, medical, or psychological deficit or condition that would affect ambulation ability or compliance with study protocol (such as Parkinson's disease, major depression, pain, cardiac arrhythmias); aphasia with an inability to follow 2 consecutive step commands or a cognitive deficit; severe hip, knee, or ankle contracture that would preclude passive range of motion of the leg | |
| Interventions | 3 arms:
The study consisted of 1 training session per weekday for 4 weeks. Experimental groups 1 and 2 underwent gait training for 20 minutes, with body weight support by an electromechanical gait trainer; group 2 also received functional electrical stimulation to the paretic lower limb during gait training. Participants in group 3 received physiotherapy overground gait training based on the principles of proprioceptive neuromuscular facilitation and Bobath concepts. |
|
| Outcomes |
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random numbers |
| Allocation concealment (selection bias) | Low risk | Concealed |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Yes for primary outcome |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | ITT unclear |
Ucar 2014.
| Methods | RCT
Method of randomisation: random number list Blinding of outcome assessors: yes Adverse events: not described Deaths: not described Dropouts: not described ITT: unknown |
|
| Participants | Country: Turkey
22 participants (11 in treatment group, 11 in control group)
Ambulatory at start of study
Mean age: 56 years in experimental group and 62 years in control group Inclusion criteria: adult male (> 18 years), ability to ambulate 10 metres without personal assistance, and not receiving any other physical therapy Exclusion criteria: body weight more than 300 pounds (135 kg), FAC score < 3 and not able to walk consistently or independently within the community, cognitive deficits, cardiac disease, spasticity of the lower limbs preventing robotic walking, traumatic stroke, intracranial space occupying lesion‐induced strokes, and seizures |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were assessed at baseline and after 2 and 8 weeks:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Random number list |
| Allocation concealment (selection bias) | Unclear risk | Not described |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Stated as blinded |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Van Nunen 2012.
| Methods | RCT Method of randomisation: stated as block randomisation Blinding of outcome assessors: no Adverse events: none described Deaths: none Dropouts: none ITT: unknown | |
| Participants | Country: Hong Kong, China
50 participants (30 in treatment group , 20 in control group)
Non‐ambulatory at study onset (intervention group: 3 , control group: 2 ) Mean age: 53 years Inclusion criteria: unknown Exclusion criteria: unknown |
|
| Interventions | 2 arms
|
|
| Outcomes | Outcomes were assessed at baseline and after 8 weeks:
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Unclear |
| Allocation concealment (selection bias) | Low risk | A person not involved in the study was asked to draw 1 of the opaque envelopes inside which group assignment was established each time a new participant entered the study. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Unclear |
Waldman 2013.
| Methods | RCT Method of randomisation: unclear Blinding of outcome assessors: no Adverse events: none described Deaths: none Dropouts: none ITT: unknown | |
| Participants | Country: USA
24 participants (12 in treatment group, 12 in control group)
Ambulatory at study onset Mean age: 51 years Inclusion criteria: people with stroke duration longer than 3 months, with reduced ankle range of motion and strength, and able to walk with or without assistant devices Exclusion criteria: unknown |
|
| Interventions | 2 arms
|
|
| Outcomes | Outcomes were assessed at baseline and after 6 and 12 weeks :
|
|
| Notes | Published and unpublished data provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method not described. |
| Allocation concealment (selection bias) | High risk | Not described |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not described |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | No ITT |
Watanabe 2014.
| Methods | RCT
Method of randomisation: computer generated
Blinding of outcome assessors: stated as 'no'
Adverse events: none
Deaths: none
Dropouts: 10 (6 in experimental group and 4 in control group) ITT: no |
|
| Participants | Country: Japan
32 participants (17 in treatment group, 15 in control group)
Not ambulatory at start of study
Mean age: 67 years in experimental group and 76 years in control group Inclusion criteria: stroke < 6 months Exclusion criteria: not ambulating prior to stroke, FAC 4 or 5, severe cardiac disease, NYHA III or IV, severe disturbance of consciousness (Japan Coma Scale II or III), size limitations for the robotic orthosis, skin disease, pacemaker |
|
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline, after 12 training sessions Primary outcome:
Secondary outcomes:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated sequence |
| Allocation concealment (selection bias) | Unclear risk | Odd‐numbered participants underwent gait training using the Hybrid Assistive Limb, and even‐numbered participants underwent conventional gait training (maybe even high risk of bias). |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Not done |
| Incomplete outcome data (attrition bias) All outcomes | High risk | No ITT |
Werner 2002.
| Methods | Cross‐over RCT Method of randomisation: participants randomly assigned to groups (group allocation in envelopes that were drawn by an independent person) Blinding of outcome assessors: yes Adverse events: none Deaths: none Dropouts: none ITT: yes | |
| Participants | Country: Germany 30 participants (15 in treatment group, 15 in control group) Non‐ambulatory at study onset Mean age: 60 years Inclusion criteria: first stroke, supratentorial lesion 4 to 12 weeks' poststroke, younger than 75 years of age, not able to walk (FAC of 2 or less), able to sit unsupported on the edge of a bed, able to stand for at least 10 seconds with help, able to provide and did provide written informed consent Exclusion criteria: hip and knee extension deficit > 20 degrees; passive dorsiflexion of the affected ankle to less than a neutral position; severe impairment of cognition or communication; evidence of cardiac ischaemia, arrhythmia, decompression, or heart failure; feeling of 'overexertion' or heart rate exceeding the age‐predicted maximum (i.e. 190 beats/minute minus age) during training; resting systolic blood pressure exceeding 200 mm Hg at rest or dropping by more than 10 mm Hg with increasing workload | |
| Interventions | 2 arms:
Treated as inpatients for five 15‐ to 20‐minute sessions per week for 2 weeks A: treadmill training with body weight support: participants walked on a treadmill with partial body weight support provided by a harness B: gait trainer with body weight support: participants walked on Gait Trainer with partial body weight support provided by a harness |
|
| Outcomes | Outcomes were recorded at baseline and after 2 weeks (additionally after 4 and 6 weeks, but only the first phase was included in this review):
|
|
| Notes | We used the first treatment phase only. Published and unpublished data provided by the authors. 0% dropouts at the end of first treatment phase (data were analysed as ITT) | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | By envelopes |
| Allocation concealment (selection bias) | Low risk | Concealed envelopes that were drawn by an independent person |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Yes |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data |
Westlake 2009.
| Methods | RCT Method of randomisation: computer‐generated random order (stratified by fast or slow walking) Blinding of outcome assessors: not described Adverse events: 1 in control group Deaths: none Dropouts: none ITT: yes | |
| Participants | Country: USA 16 participants (8 in treatment group, 8 in control group) All were ambulatory at study onset Mean age: 57 years Inclusion criteria: hemiparesis resulting from a single cortical or subcortical stroke > 6 months before the study, categorised as at least unlimited household ambulatory, written informed consent Exclusion criteria: unstable cardiovascular, orthopaedic, or neurological conditions; uncontrolled diabetes that would preclude exercise of moderate intensity; significant cognitive impairment affecting ability to follow directions | |
| Interventions | 2 arms:
|
|
| Outcomes | Outcomes were recorded at baseline and after 4 weeks
|
|
| Notes | — | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Unclear |
| Allocation concealment (selection bias) | Low risk | Randomisation list was overseen by 1 of the investigators who had no contact with participants until group assignment was revealed. Group assignment was not revealed to study personnel until the participant consented and baseline testing was complete. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data |
ABC: Activities‐specific Balance Confidence ADL: activities of daily living CT: computed tomography FAC: Functional Ambulation Category ITT: intention‐to‐treat MMSE: Mini–Mental State Examination MRC: Medical Research Council MRI: magnetic resonance imaging NIHSS: National Institutes of Health Stroke Scale NYHA: New York Heart Association PF‐DF: plantar flexion and dorsiflexion RAGT: robot‐assisted gait training RCT: randomised controlled trial SF‐36: 36‐Item Short Form Health Survey
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bae 2014 | Intervention: both groups received robot‐assisted gait training (the experimental group also received functional electrical stimulation on the ankle dorsiflexor of the affected side). |
| Byun 2011 | Uses a sliding rehabilitation machine, not robotic training as experimental condition |
| Caldwell 2000 | Did not investigate electromechanical‐ and robotic‐assisted gait‐training devices as stated in the protocol of this review: bicycle training versus treadmill walking versus variable surface training were investigated |
| Danzl 2013 | Investigates brain stimulation, and both groups participated in identical locomotor training with a robotic gait orthosis |
| David 2006 | Did not meet inclusion criteria of this review: not an RCT |
| Forrester 2016 | Compared different robotic applications |
| Gong 2003 | Did not investigate electromechanical‐ and robotic‐assisted gait‐training devices as stated in the protocol of this review: no electromechanical‐assisted devices were compared |
| Goodman 2014 | Did not meet inclusion criteria of this review: not an RCT |
| Hesse 2001 | Did not meet inclusion criteria of this review: not an RCT |
| Hsieh 2014 | Investigated upper limbs |
| Mirelman 2009 | Did not meet inclusion criteria of this review: experimental and control groups received a kind of assisted stepping therapy in a seated position. This study investigated the effects of virtual reality as an adjunct to stepping training. After discussion, we reached consensus to exclude this study from our review. |
| Morone 2016 | Investigated the i‐Walker, a rollator vehicle |
| NCT01337960 | Compared different robotic approaches |
| Page 2008 | Did not meet inclusion criteria of this review: the experimental group received a kind of assisted stepping therapy in a seated position. This study investigated the effect of the NuStep apparatus. After discussion, we reached consensus to exclude this study from our review. |
| Park 2015 | Uses a treadmill training approach as experimental condition (Gait Trainer 2 analysis system, Biodex Medical Systems, Inc., Shirley, NY, USA) |
| Patten 2006 | According to the information on ClinicalTrials.gov (NCT00125619), this is a 1‐arm, non‐randomised trial. |
| Pennati 2015 | Investigated upper limbs |
| Picelli 2015 | Both groups received the same robotic treatment. |
| Pitkanen 2002 | Did not meet inclusion criteria of this review: the study describes preliminary findings of an initial sample of 9 participants; the experimental group received treadmill training or gait training |
| Richards 1993 | Did not meet inclusion criteria of this review: the experimental group received a specialised locomotor training including early intensive physiotherapy with tilt table, limb load monitor, resistance exercises, and treadmills to promote functional recovery. After discussion, we reached consensus to exclude this study. |
| Richards 2004 | Did not meet inclusion criteria of this review: the experimental group received specialised locomotor training including early intensive physiotherapy with tilt table, limb load monitor, resistance exercises and treadmills to promote functional recovery. After discussion, we reached consensus to exclude this study. |
| Shirakawa 2001 | Not an RCT |
| Skvortsova 2008 | Not an RCT, control groups were age and sex matched |
| Stoller 2015 | Compared 2 treadmill exercise options |
| Wu 2014 | Compared different modes (resistance versus assistance training) of the same robotic device |
RCT: randomised controlled trial
Characteristics of studies awaiting assessment [ordered by study ID]
Chernikova 2014.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | — |
Globokar 2005.
| Methods | Probably an RCT |
| Participants | People after stroke, number unclear |
| Interventions | 2 arms:
|
| Outcomes | Unclear |
| Notes | This study was presented at the 5th World Congress of Physical Medicine and Rehabilitation. |
Golyk 2006.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | — |
Jang 2005.
| Methods | Probably an RCT |
| Participants | 34 non‐ambulatory stroke survivors |
| Interventions | 2 arms:
|
| Outcomes | Unclear |
| Notes | This study was presented at the 5th World Congress of Physical Medicine and Rehabilitation. |
Kim 2001.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | Study was found in the Proceedings of the 1st International Congress of International Society of Physical and Rehabilitation Medicine (ISPRM), 2001 July 7‐13. |
Kim 2014.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | — |
Koeneman 2004.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | — |
Mehrberg 2001.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | — |
Ohata 2015.
| Methods | Probably a randomised cross‐over trial |
| Participants | 23 participants with stroke (stroke onset < 6 months) Group 1 (N = 13; mean: 60.9 ± 9.6 years) Group 2 (N = 10; mean: 61.1 ± 14.6 years) |
| Interventions | Gait training with the Stride Management Assist device (Honda R&D Co., Ltd. Japan) (20 minutes/time, 5 times per week for 4 weeks) Conventional rehabilitation (40 minutes/time) 4 weeks |
| Outcomes |
|
| Notes | Published as abstract |
Sale 2012.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | No longer at ClinicalTrials.gov |
Wu 2012.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | ‐ |
Yoon 2015.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | N = 10 in gait training with active‐assistive gait device group N = 10 in control group |
| Outcomes |
|
| Notes | Conference abstract |
Zhu 2016.
| Methods | Probably an RCT |
| Participants | Unclear |
| Interventions | Unclear |
| Outcomes | Unclear |
| Notes | ‐ |
RCT: randomised controlled trial
Characteristics of ongoing studies [ordered by study ID]
Louie 2015.
| Trial name or title | Use of a powered robotic exoskeleton to promote walking recovery after stroke: study protocol for a randomised controlled trial |
| Methods | Single‐blind randomised controlled trial to evaluate the efficacy of a powered mobile exoskeleton (Ekso) on improving walking ability in people early after stroke |
| Participants | 50 individuals admitted for stroke rehabilitation in Canada, within 4 weeks' poststroke and needing second person assist to walk, will be randomly assigned to either a usual care group or exoskeleton group for 5 days/week for 4 weeks. |
| Interventions | 2 arms:
|
| Outcomes | Outcomes will be measured at baseline, 4 weeks later at discharge, and at 6 months after program ends. Primary outcome:
Secondary outcomes:
|
| Starting date | Unknown |
| Contact information | Louie DR1,2 1University of British Columbia, Vancouver, Canada 2Rehabilitation Research Program, Vancouver Coastal Health Research Institute, Vancouver, Canada |
| Notes | Presented at the Canadian Stroke Conference, Toronto, 2015 September 17 |
NCT00284115.
| Trial name or title | Efficacy of a mechanical gait repetitive training technique compared with a usual rehabilitation program on gait recovery in hemiparetic stroke patients |
| Methods | RCT with 2 arms |
| Participants | Country: France Inclusion criteria: men or women aged 18 years or older; hemiplegia secondary to stroke; interval between stroke and study inclusion of 2 months or less; first‐time supratentorial stroke; non‐ambulatory (FAC stage 0); able to sit unsupported at the edge of the bed; no severe impairment of cognition or communication; written informed consent provided Exclusion criteria: orthopaedic or rheumatological disease impairing mobility, or both; other neurologically associated disease; history of myocardial infarction or deep venous embolism or pulmonary embolism within 3 months before study inclusion; chronic pulmonary disease; intolerance to standing up |
| Interventions | 4‐week rehabilitation programme comparing physiotherapy and gait trainer therapy with physiotherapy alone |
| Outcomes | Primary outcomes: walking speed (time needed to walk 10 metres) after the 4‐week rehabilitation programme Secondary outcomes: FAC; walking endurance (6‐minute walk test); time to self sufficient gait recovery; spasticity (Modified Ashworth Scale); Motricity Index; need for mobility and self assistance (Barthel Index, PMSI‐SSR scores, need for physical assistance); economic evaluation (healthcare requirements, rehabilitation unit length of stay) |
| Starting date | March 2006 |
| Contact information | Principal Investigator: Régine Brissot, MD, Service de Médecine Physique et Réadaptation, Hôpital Pontchaillou, Rennes, 35033, France Tel: +33 2 9928 4219 email: regine.brissot@chu‐rennes.fr |
| Notes | Expected total enrolment: 122 participants Sponsored by: Rennes University Hospital Information derived from: ClinicalTrials.gov identifier: NCT00284115 |
NCT00530543.
| Trial name or title | Effects of gait training with assistance of a robot‐driven gait orthosis in hemiparetic patients after stroke |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions | Robot‐assisted gait training |
| Outcomes | Not provided |
| Starting date | Unknown status |
| Contact information | Not provided |
| Notes | The recruitment status of this study is unknown. The completion date has passed, and the status has not been verified in more than 2 years. |
NCT01146587.
| Trial name or title | Comparative study of GangTrainer GT1, Lokomat and conventional physiotherapy (GALOP) |
| Methods | RCT with 3 arms |
| Participants | Inclusion criteria: first supratentorial stroke (ischaemic, haemorrhagic, or intracerebral haemorrhage) resulting in hemiparesis; interval from stroke 3 to 12 weeks; non‐ambulatory (FAC < 3); free sitting on bedside for 1 minute, with both feet on the floor and holding onto bedside by hands; Barthel Index 25 to 65 Exclusion criteria: unstable cardiovascular system (in case of doubt, only after approval by an internist); manifested heart diseases like labile compensated cardiac insufficiency (NYHA III), angina pectoris, myocardial infarction 120 days before study onset, cardiomyopathy, severe cardiac arrhythmia, severe joint misalignment (severe constriction of movement for hip, knee, or ankle, or any combination of the 3: more than 20° fixed hip and knee extension deficit, or more than 20° fixed plantar flexion of the ankle); severe cognitive dysfunction that prevents comprehension of the aims of study; severe neurological or orthopaedic diseases (e.g. polio, Parkinson's disease) that massively affect mobility; deep vein thrombosis; severe osteoporosis; or malignant tumour disease |
| Interventions | Group A: 30 minutes of treatment on the GangTrainer GT1 and 30 minutes of conventional physiotherapy every workday for 8 weeks Group B: 30 minutes of treatment on the Lokomat and 30 minutes of conventional physiotherapy every workday for 8 weeks Group C: 60 minutes of conventional physiotherapy every workday for 8 weeks |
| Outcomes | Primary outcomes: FAC, modified Emory Functional Ambulation Secondary outcomes: Barthel Index, 10‐metre walk test, 6‐minute walk test on the floor, Medical Research Council, Rivermead Visual Gait Assessment, EuroQol‐5 Dimensions (EQ‐5D) |
| Starting date | August 2010 |
| Contact information | Contact: Andreas Waldner, MD; +39 0471 471 471; waldner.andreas@villamelitta.it Contact: Christopher Tomelleri, MSc; +39 0471 471 471; christopher.tomelleri@villamelitta.it |
| Notes | Estimated enrolment: 120 Estimated study completion date: August 2013 Estimated primary completion date: August 2013 (final data collection date for primary outcome measure) |
NCT01187277.
| Trial name or title | A randomised controlled trial on hemiplegic gait rehabilitation: robotic locomotor training versus conventional training in subacute stroke |
| Methods | RCT with 2 arms |
| Participants | Country: Thailand
Inclusion criteria: subacute first‐time stroke patients (haemorrhage and ischaemic), age 18 to 80 years, impaired FAC at initial score 0 to 2, cardiovascular stable, provided signed informed consent Exclusion criteria: unstable general medical condition, severe malposition or fixed contracture of joint with an extension deficit > 30°, any functional impairment before stroke, cannot adequately co‐operate in training, severe communication problems, severe cognitive‐perceptual deficits |
| Interventions | Group A: conventional therapy: 50 minutes individual physiotherapy and 60 minutes individual occupational therapy per workday (5 times per week) for 4 consecutive weeks Group B: conventional therapy plus robot‐assisted gait training: 30 minutes individual physiotherapy plus 20 minutes robotic‐assisted gait training (with Gait Trainer GT1) and 60 minutes individual occupational therapy per workday (5 times per week) for 4 consecutive weeks |
| Outcomes | Primary outcomes: FAC 0 to 5 and Barthel Index 0 to 100 Secondary outcome: Berg Balance Scale 0 to 56, Resistance to Passive Movement Scale (REPAS)‐Muscle tone 0 to 52 |
| Starting date | January 2011 |
| Contact information | Principal Investigator: Ratanapat Chanubol, MD, Rehabilitation Department, Prasat Neurological Institute, Mahidol University |
| Notes | Study Completion Date: July 2012 Enrolment: 60 |
NCT01678547.
| Trial name or title | Robot walking rehabilitation in stroke patients |
| Methods | RCT with 3 arms |
| Participants | Inclusion criteria: between the ages of 18 and 95 years, able to walk 25 feet unassisted or with assistance, first acute event of cerebrovascular stroke, unilateral paresis, ability to understand and follow simple instructions, ability to walk without assistance before stroke, endurance sufficient to stand at least 20 minutes unassisted per participant report Exclusion criteria: unable to understand instructions required by the study (Informed Consent Test of Comprehension), medical or neurological comorbidities that could contribute to significant gait dysfunction, uncontrolled hypertension > 190/110 mm Hg, significant symptoms of orthostasis when standing up, circulatory problems, history of vascular claudication or significant (+ 3) pitting oedema, lower extremity injuries or joint problems (hip or leg) that limit range of motion or function or cause pain with movement, bilateral impairment, severe sensory deficits in the paretic upper limb, cognitive impairment or behavioural dysfunction that would influence the ability to comprehend or participate in the study, women who are pregnant or lactating, or both |
| Interventions | Experimental group: Robot G‐EO: each participant will be asked to perform 15 sessions (3 to 5 days a week for 4 up to 5 weeks) consisting of a treatment cycle using the G‐EO system device, according to individually tailored exercise scheduling Control group: Treadmill training: each participant will be asked to perform 15 sessions (3 to 5 days a week for 4 up to 5 weeks) consisting of a treatment cycle using the treadmill system device, according to individually tailored exercise scheduling Control group: Ground treatment: each participant will be asked to perform 15 sessions (3 to 5 days a week for 4 up to 5 weeks) of traditional lower limb physiotherapy |
| Outcomes | |
| Starting date | September 2012 |
| Contact information | Contact: Patrizio Sale, MD; patrizio.sale@gmail.com Contact: Marco Franceschini, MD; marco.franceschini@sanraffaele.it |
| Notes | Estimated enrolment: 90 Estimated study completion date: September 2015 Estimated primary completion date: August 2014 (final data collection date for primary outcome measure) |
NCT01726998.
| Trial name or title | Effects of locomotion training with assistance of a robot‐driven gait orthosis in hemiparetic patients after subacute stroke |
| Methods | Randomised trial 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
72 first‐ever stroke patients who could not walk independently (FAC < 2), and suffered stroke within 6 months were enrolled and randomly assigned into 2 groups. People with congestive heart failure, malignancies, cardiopulmonary dysfunctions, and who could not walk independently before their stroke were excluded. |
| Interventions | 2 groups received 30 minutes of conventional gait training including neurodevelopmental treatment:
|
| Outcomes | Independent walking ability (FAC ≥ 3), FAC, Motricity index, Fugl‐Meyer Assessment, Modified Barthel Index, Medical Research Council were assessed for lower extremity muscles before, during (2 weeks), and after training. Independent walking ability was followed until 3 months. |
| Starting date | March 2012 |
| Contact information | Yonsei University |
| Notes |
NCT02114450.
| Trial name or title | Human‐machine system for the H2 lower limb exoskeleton (H2‐NeuroExo) |
| Methods | RCT with 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions | Experimental: robot‐assisted rehabilitation participants will receive robot‐assisted training with the H2 lower limb powered exoskeleton. They will perform walking and other lower limb exercises (as applicable) while wearing the H2 lower limb powered exoskeleton. Training will involve 3 sessions per week for 4 weeks, each lasting about 1.5 hours. Control: supervised motor practice participants will perform walking and other lower limb exercises (as applicable) under the supervision of a research physical therapist. Training will involve 3 sessions per week for 4 weeks, each lasting about 1.5 hours. |
| Outcomes | Primary outcome measures
Secondary outcome measures
|
| Starting date | March 2014 |
| Contact information | clinicaltrials.gov/ct2/show/study/NCT02114450#contacts |
| Notes |
NCT02471248.
| Trial name or title | Interactive exoskeleton robot for walking ‐ ankle joint |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions |
|
| Outcomes |
|
| Starting date | June 2015 |
| Contact information | Raymond KY Tong, Professor, Chinese University of Hong Kong |
| Notes |
NCT02483676.
| Trial name or title | Adaptive ankle robot control system to reduce foot‐drop in chronic stroke |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions |
|
| Outcomes |
|
| Starting date | September 2015 |
| Contact information | clinicaltrials.gov/ct2/show/study/NCT02483676?term=NCT02483676&rank=1#contacts |
| Notes |
NCT02545088.
| Trial name or title | New technology for individualised, intensive training of gait after stroke ‐ phase III trials, study II |
| Methods | Randomised 3 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions |
|
| Outcomes |
|
| Starting date | October 2015 |
| Contact information | clinicaltrials.gov/ct2/show/study/NCT02545088?term=NCT02545088&rank=1#contacts |
| Notes |
NCT02680691.
| Trial name or title | Robot assisted gait training in patients with infratentorial stroke |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions |
|
| Outcomes | Primary outcome measures
Secondary outcome measures
|
| Starting date | April 2015 |
| Contact information | clinicaltrials.gov/ct2/show/study/NCT02680691?term=NCT02680691&rank=1#contacts |
| Notes |
NCT02694302.
| Trial name or title | Clinical trial of robot‐assisted‐gait‐training (RAGT) in stroke patients (Walkbot) |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions |
|
| Outcomes | Primary outcome measure
Secondary outcome measures
|
| Starting date | March 2014 |
| Contact information | P&S Mechanics Co., Ltd. |
| Notes |
NCT02755415.
| Trial name or title | Clinical applicability of robot‐assisted gait training system in acute stroke patients |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions | Device: HIWIN Robotic Gait Training System˜: the HIWIN Robotic Gait Training System is an automatic training system that combines weight‐bearing standing, repetitive stepping, and gait training |
| Outcomes |
|
| Starting date | May 2016 |
| Contact information | China Medical University Hospital |
| Notes | Other study ID: CMUH105‐REC1‐037 |
NCT02781831.
| Trial name or title | Robot‐assisted gait training for patients with stroke |
| Methods | Randomised 2 arms |
| Participants | Inclusion criteria
Exclusion criteria
|
| Interventions | Standard hospital‐based rehabilitation for people with stroke |
| Outcomes |
|
| Starting date | May 2016 |
| Contact information | Principal Investigator: Nai‐Hsin Meng, MD, China Medical University Hospital |
| Notes | HIWIN‐CMU‐C‐105‐1 |
NCT02843828.
| Trial name or title | Gait pattern analysis and feasibility of gait training with a walking assist robot in stroke patients and elderly adults |
| Methods | Randomised 2 treatment groups |
| Participants | Number of participants: 54 (n = 27 per group) Inclusion criteria
Exclusion criteria
|
| Interventions | Group 1 : gait rehabilitation with hip assist robot (Samsung Hip Assist v1): 10 sessions (5 sessions: treadmill gait training/5 sessions: overground gait training), 30 minutes per session Group 2 : gait rehabilitation without hip assist robot: 10 sessions (5 sessions: treadmill gait training/5 sessions: overground gait training), 30 minutes per session |
| Outcomes |
|
| Starting date | September 2015 |
| Contact information | Yun‐Hee Kim, MD, PhD; 82‐2‐3410‐2824 yunkim@skku.edu |
| Notes |
FAC: Functional Ambulation Category NYHA: New York Heart Association RCT: randomised controlled trial
Differences between protocol and review
In our protocol we stated that we would use the PEDro Scale to assess the methodological quality of the included trials. However, Chapter 8 of the latest edition of the Cochrane Handbook for Systematic Reviews of Interventions suggests that scales that yield a summary score should be avoided (Higgins 2011a). In accordance with this suggestion, we no longer used the PEDro Scale to assess the methodological quality of the included trials, instead using the Cochrane 'Risk of bias' tool to analyse trial methodology.
In our protocol we planned to quantify heterogeneity with the I² statistic and to use a cutoff of I² = 50% for all comparisons. Additonally, we planned to calculate the overall effects using a random‐effects model instead of a fixed‐effect model when we found substantial heterogeneity. However, in this update we calculated the overall effects using a random‐effects model, irrespective of the level of heterogeneity.
In this review update we expanded our post‐hoc sensitivity analysis: type of device (Analysis 5.1; Analysis 5.2; Analysis 5.3) by introducing a new subgroup of studies using mobile and ankle devices and adding a new comparison in Analysis 5 (Analysis 5.3 Different devices for regaining walking capacity).
Contributions of authors
Jan Mehrholz (JM) contributed to the conception and the design of the protocol and drafted the protocol. He searched electronic databases and conference proceedings, screened titles and abstracts of references identified by the search, selected and assessed trials, extracted trial and outcome data, guided the analysis and interpretation of data, and contributed to and approved the final manuscript of the review.
Simone Thomas (ST) evaluated and extracted trial data, assessed the methodological quality of selected trials, contributed to the interpretation of data, and contributed to and approved the final manuscript of the review.
Cordula Werner (CW) screened the titles and abstracts of references identified by the search; located, selected, and assessed trials; extracted trial and outcome data; assessed the methodological quality of selected trials; contributed to the interpretation of data; and contributed to and approved the final manuscript of the review.
Joachim Kugler (JK) evaluated and extracted trial and outcome data, assessed the methodological quality of selected trials, contributed to the interpretation of data, and contributed to and approved the final manuscript of the review.
Marcus Pohl (MP) contributed to the conception and design of the review, drafted the protocol, and assessed the methodological quality of selected trials. Together with JM, he contacted trialists about unpublished data and entered the data, carried out statistical analysis, helped with the interpretation of the data, drafted the review, and approved the final manuscript of the review.
Bernhard Elsner (BE) searched electronic databases and conference proceedings, screened titles and abstracts of references identified by the search, selected and assessed trials, guided analysis and the interpretation of the data, and contributed to and approved the final manuscript of the review.
Sources of support
Internal sources
Klinik Bavaria Kreischa, Wissenschaftliches Institut, Germany.
Technical University Dresden, Lehrstuhl Public Health, Germany.
SRH Fachhochschule Gera, Lehrstuhl Therapiewissenschaften, Germany.
External sources
No sources of support supplied
Declarations of interest
Bernhard Elsner: none known.
Simone Thomas: none known.
Joachim Kugler: none known.
Marcus Pohl was author of one included trial (Pohl 2007).
Jan Mehrholz was co‐author of one included trial (Pohl 2007).
Cordula Werner was an author of two included trials (Pohl 2007; Werner 2002), and of one excluded trial (Hesse 2001).
These review authors (MP, JM, CW) did not participate in quality assessment and data extraction of these studies.
New search for studies and content updated (conclusions changed)
References
References to studies included in this review
Aschbacher 2006 {unpublished data only}
- Aschbacher B. Comparing gait training in patients after stroke with task oriented physiotherapy or robot‐assisted treadmill training: a feasibility study. Unpublished 2006.
Bang 2016 {published data only}
- Bang DH, Shin WS. Effects of robot‐assisted gait training on spatiotemporal gait parameters and balance in patients with chronic stroke: a randomized controlled pilot trial. NeuroRehabilitation 2016;38:343‐9. [DOI] [PubMed] [Google Scholar]
Brincks 2011 {published data only}
- Brincks J. The order of gait training, including Lokomat® and Physiotherapy, does not influence gait symmetry in subacute ambulatory persons with stroke. Physiotherapy, Supplement S1, 16th World Congress of Physical Therapy, 2011 June 20‐23 , Amsterdam. World Confederation of Physical Therapy, 2011; Vol. 97. [Number: RR‐PO‐208‐17‐Tue]
Buesing 2015 {published data only}
- Buesing C, Fisch G, O'Donnell M, Shahidi I, Thomas L, Mummidisetty CK, et al. Effects of a wearable exoskeleton stride management assist system (SMA) on spatiotemporal gait characteristics in individuals after stroke: a randomized controlled trial. Journal of NeuroEngineering and Rehabilitation 2015; Vol. 12, issue 69. [DOI] [PMC free article] [PubMed]
Chang 2012 {published data only}
- Chang WH, Kim MS, Huh JP, Lee PK, Kim YH. Effects of robot‐assisted gait training on cardiopulmonary fitness in subacute stroke patients: a randomized controlled study. Neurorehabilitation and Neural Repair 2012;26(4):318–24. [DOI] [PubMed] [Google Scholar]
- Kim M, Kim YH, Lee PKW, Hyong MK, Jung PH. Effect of robot‐assisted gait therapy on cardiopulmonary fitness in subacute stroke patients. Neurorehabilitation and Neural Repair 2008;22:594. [DOI] [PubMed] [Google Scholar]
Cho 2015 {published data only}
- Cho DY, Park SW, Lee MJ, Park DS, Kim EJ. Effects of robot‐assisted gait training on the balance and gait of chronic stroke patients: focus on dependent ambulators. Journal of Physical Therapy Science 2015;27(10):3053‐7. [0915‐5287] [DOI] [PMC free article] [PubMed] [Google Scholar]
Chua 2016 {published data only}
- Chua J, Culpan J, Menon E. Efficacy of an electromechanical gait trainer poststroke in Singapore: a randomized controlled trial. Archives of Physical Medicine and Rehabilitation 2016;97:683‐90. [DOI] [PubMed] [Google Scholar]
Dias 2006 {published data only}
- Dias D, Laíns J, Pereira A, Nunes R, Caldas J, Amaral C, et al. Partial body weight support in chronic hemiplegics: a randomized control trial. Europa Medicophysica 2007;43(4):499‐504. [PubMed] [Google Scholar]
Fisher 2008 {published and unpublished data}
- Fisher S. Use of autoambulator for mobility improvement in patients with central nervous system (CNS) injury or disease. Neurorehabilitation and Neural Repair 2008;22:556. [Google Scholar]
- Fisher S, Lucas L, Thrasher TA. Robot‐assisted gait training for patients with hemiparesis due to stroke. Topics in Stroke Rehabilitation 2011;18(3):269‐76. [DOI] [PubMed] [Google Scholar]
Forrester 2014 {published data only}
- Forrester LW, Roy A, Krywonis A, Kehs G, Krebs HI, Macko RF. Modular ankle robotics training in early subacute stroke: a randomized controlled pilot study. Neurorehabilitation and Neural Repair 2014; Vol. 28, issue 7:678‐87. [1552‐6844] [DOI] [PMC free article] [PubMed]
Geroin 2011 {published data only}
- Geroin C, Picelli A, Munari D, Waldner A, Tomelleri C, Smania N. Combined transcranial direct current stimulation and robot‐assisted gait training in patients with chronic stroke: a preliminary comparison. Clinical Rehabilitation 2011;25(6):537‐48. [DOI] [PubMed] [Google Scholar]
Han 2016 {published data only}
- Han EY, Im SH, Kim BR, Seo MJ, Kim MO. Robot‐assisted gait training improves brachial‐ankle pulse wave velocity and peak aerobic capacity in subacute stroke patients with totally dependent ambulation: Randomized controlled trial. Medicine 2016;95:e5078. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hidler 2009 {published data only (unpublished sought but not used)}
- Hidler J, Nichols D, Pelliccio M, Brady K, Campbell DD, Kahn JH, et al. Multicenter randomized clinical trial evaluating the effectiveness of the Lokomat in subacute stroke. Neurorehabilitation and Neural Repair 2009;23(1):5‐13. [DOI] [PubMed] [Google Scholar]
Hornby 2008 {published data only}
- Hornby TG, Campbell DD, Kahn JH, Demott T, Moore JL, Roth HR. Enhanced gait‐related improvements after therapist‐ versus robotic‐assisted locomotor training in subjects with chronic stroke: a randomized controlled study. Stroke 2008;39(6):1786‐92. [DOI] [PubMed] [Google Scholar]
Husemann 2007 {published data only}
- Husemann B, Müller F, Krewer C, Laß A, Gille C, Heller S, et al. Effects of locomotion training with assistance of a driven gait orthosis in hemiparetic patients after stroke. Neurologie & Rehabilitation 2004;10(4):25. [Google Scholar]
- Husemann B, Müller F, Krewer C, Laß A, Gille C, Heller S, et al. Effects of locomotion training with assistance of a robot‐driven gait orthosis in hemiparetic patients after stroke: a randomized controlled pilot study. Stroke 2007;38(2):349‐54. [DOI] [PubMed] [Google Scholar]
Kim 2015 {published data only}
- Kim S‐Y, Yang L, Park IJ, Kim EJ, Park MS, You SH, et al. Effects of innovative WALKBOT robotic‐assisted locomotor training on balance and gait recovery in hemiparetic stroke: a prospective, randomized, experimenter blinded case control study with a four‐week follow‐up. IEEE Transactions on Neural Systems & Rehabilitation Engineering 2015;23(4):636‐42. [1558‐0210] [DOI] [PubMed] [Google Scholar]
Kyung 2008 {published and unpublished data}
- Jung KH, Ha HG, Shin HJ, Ohn SH, Sung DH, Lee PKW, et al. Effects of robot‐assisted gait therapy on locomotor recovery in stroke patients. Journal of Korean Academy of Rehabilitation Medicine 2008;32:258‐66. [Google Scholar]
- Kyung HJ, Kim YH. Effects of robot‐assisted gait therapy on locomotor recovery in stroke patients. Asia‐Oceanian Conference of Physical & Rehabilitation Medicine; 2008 May 16‐19; Nanjing, China. 2008.
Mayr 2008 {published and unpublished data}
- Mayr A, Quirbach E, Picelli A, Kofler M, Smania N, Saltuari L. Effect of early robot‐assisted gait retraining in nonambulatory patients with stroke: a single‐blind randomized controlled trial. Unpublished 2016. [DOI] [PubMed]
- Mayr A, Saltuari L, Quirbach E. Impact of Lokomat training on gait rehabilitation. Neurorehabilitation and Neural Repair 2008;22(5):596. [DOI] [PubMed] [Google Scholar]
Morone 2011 {published and unpublished data}
- Morone G, Bragoni M, Iosa M, Angelis D, Venturiero V, Coiro P, et al. Who may benefit from robotic‐assisted gait training? A randomized clinical trial in patients with subacute stroke. Neurorehabilitation and Neural Repair 2011;25(7):636‐44. [DOI] [PubMed] [Google Scholar]
Noser 2012 {published data only (unpublished sought but not used)}
- NCT00975156. NCT00975156 [Improving ambulation post stroke with robotic training]. clinicaltrials.gov/show/NCT00975156 (first received: 10 September 2009).
Ochi 2015 {published data only}
Peurala 2005 {published data only}
- Peurala S, Tarkka I, Pitkänen K, Sivenius J. The effectiveness of body weight‐supported gait training and floor walking in patients with chronic stroke. Archives of Physical Medicine and Rehabilitation 2005;86:1557‐64. [DOI] [PubMed] [Google Scholar]
- Peurala SH, Pitkanen K, Sivenius J, Tarkka I. Body‐weight supported gait exercise compared with floor walking in chronic stroke patients. Archives of Physical Medicine and Rehabilitation 2004;85:E7. [DOI] [PubMed] [Google Scholar]
- Peurala SH, Pitkanen K, Sivenius J, Tarkka IM. Body‐weight supported gait trainer exercises with or without functional electrical stimulation improves gait in patients with chronic stroke. Neurorehabilitation and Neural Repair 2006;20(1):98. [Google Scholar]
Peurala 2009 {published and unpublished data}
- Peurala SH, Airaksinen O, Huuskonen P, Jäkälä P, Juhakoski M, Sandell K, et al. Effects of intensive therapy using Gait Trainer or floorwalking exercises early after stroke. Journal of Rehabilitation Medicine 2009;41(3):166‐73. [DOI] [PubMed] [Google Scholar]
Picelli 2016 {published data only}
- Picelli A, Bacciga M, Melotti C, Marchina E, Verzini E, Ferrari F, et al. Combined effects of robot‐assisted gait training and botulinum toxin type A effect on spastic equinus foot in patients with chronic stroke: a pilot, single blind, randomized controlled trial. European Journal of Physical and Rehabilitation Medicine 2016; Vol. 52, issue 6:759‐766. [PUBMED: 27098300] [PubMed]
Pohl 2007 {published data only}
- Pohl M, Werner C, Holzgraefe M, Kroczek G, Mehrholz J, Wingendorf I, et al. Repetitive locomotor training and physiotherapy improve walking and basic activities of daily living in subacute, non‐ambulatory stroke patients: a single‐blind, randomised multi‐centre trial (DEutsche GAngtrainerStudie, DEGAS). Clinical Rehabilitation 2007;21(1):17‐27. [DOI] [PubMed] [Google Scholar]
Saltuari 2004 {published data only}
- Saltuari L. Efficiency of Lokomat training in stroke patients. Neurologie & Rehabilitation 2004;10(4):S4. [Google Scholar]
Schwartz 2006 {unpublished data only}
- Schwartz I, Katz‐Leurer M, Fisher I, Sajin A, Shochina M, Meiner Z. The effectiveness of early locomotor therapy in patients with first CVA. Collaborative Evaluation of Rehabilitation in Stroke Across Europe (CERISE) Congress; 2006 February 10‐11; Leuven, Belgium. 2006.
Stein 2014 {published data only}
- Stein J, Bishop L, Stein DJ, Wong CK. Gait training with a robotic leg brace after stroke. American Journal of Physical Medicine & Rehabilitation 2014;93(11):987‐94. [0894‐9115] [DOI] [PubMed] [Google Scholar]
Tanaka 2012 {published data only}
- Tanaka N, Saitou H, Takao T, Iizuka N, Okuno J, Yano H, et al. Effects of gait rehabilitation with a footpad‐type locomotion interface in patients with chronic post‐stroke hemiparesis: a pilot study. Clinical Rehabiilation 2012;26(8):686‐95. [DOI] [PubMed] [Google Scholar]
Tong 2006 {published and unpublished data}
- Li LSW, Tong RYU, Ng MFW, So EFM. Effectiveness of gait trainer in stroke rehabilitation. Journal of the Neurological Sciences 2005;238(Suppl 1):S81. [Google Scholar]
- Ng MFW, Tong RKY, Li LSW. A pilot study of randomized clinical controlled trial of gait training in subacute stroke patients with partial body‐weight support electromechanical gait trainer and functional electrical stimulation: six‐month follow‐up. Stroke 2008;39:154‐60. [DOI] [PubMed] [Google Scholar]
- Ng MFW, Tong RKY, So EFM, Li LSW. The therapeutic effect of electromechanical gait trainer and functional electrical stimulation for patients with acute stroke. Neurorehabilitation and Neural Repair 2006;20(1):97 (Abstract F1D‐7). [Google Scholar]
- Tong RKY, Ng MF, Li LS. Effectiveness of gait training using an electromechanical gait trainer, with and without functional electric stimulation, in subacute stroke: a randomized controlled trial. Archives of Physical Medicine and Rehabilitation 2006;87(10):1298‐304. [DOI] [PubMed] [Google Scholar]
Ucar 2014 {published data only}
- Uçar DE, Paker N, Buğdaycı D. Lokomat: a therapeutic chance for patients with chronic hemiplegia. NeuroRehabilitation 2014;34(3):447‐53. [1053: 8135] [DOI] [PubMed] [Google Scholar]
Van Nunen 2012 {published data only}
- Nunen M. RCT evaluating the effectiveness of robot‐assisted treadmill training in restoring walking ability of stroke patients. 7th World Congress of Neurological Rehabilitation; 2012 May 16‐19; Melbourne, Australia. Melbourne: World Federation for Neurorehabilittaion, 2012:210. [Abstract ID: 537, Poster No. 296]
- Nunen MPM, Gerrits KHL, Konijnenbelt M, Janssen TWJ, Haan A. Recovery of walking ability using a robotic device in subacute stroke patients: a randomized controlled study. Disability & Rehabilitation: Assistive Technology 2015;10(2):141‐8. [1748‐3107] [DOI] [PubMed] [Google Scholar]
Waldman 2013 {published data only}
Watanabe 2014 {published data only}
- Watanabe H, Tanaka N, Inuta T, Saitou H, Yanagi H. Locomotion improvement using a hybrid assistive limb in recovery phase stroke patients: a randomized controlled pilot study. Archives of Physical Medicine and Rehabilitation 2014;95(11):2006‐12. [0003: 9993] [DOI] [PubMed] [Google Scholar]
Werner 2002 {published data only}
- Werner C, Frankenberg S, Treig T, Konrad M, Hesse S. Treadmill training with partial body weight support and an electromechanical gait trainer for restoration of gait in subacute stroke patients: a randomized crossover study. Stroke 2002;33(12):2895‐901. [DOI] [PubMed] [Google Scholar]
Westlake 2009 {published data only}
- Westlake K, Patten C. Pilot study of Lokomat versus manual‐assisted treadmill training for locomotor recovery post‐stroke. Journal of NeuroEngineering and Rehabilitation 2009;6:18. [DOI: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies excluded from this review
Bae 2014 {published data only}
- Bae Y, Ko Y, Chang W, Lee J, Lee K, Park Y, et al. Effects of robot‐assisted gait training combined with functional electrical stimulation on recovery of locomotor mobility in chronic stroke patients: a randomized controlled trial. Journal of Physical Therapy Science 2014; Vol. 26, issue 12:1949‐53. [0915: 5287] [DOI] [PMC free article] [PubMed]
Byun 2011 {published data only}
- Byun SD, Jung TD, Kim CH, Lee YS. Effects of the sliding rehabilitation machine on balance and gait in chronic stroke patients ‐ a controlled clinical trial. Clinical Rehabilitation 2011;25(5):408‐15. [1477‐0873: (Electronic)] [DOI] [PubMed] [Google Scholar]
Caldwell 2000 {published data only}
- Caldwell C, Medley A. Effects of bicycling, treadmill, and variable surfaces on gait in people following a CVA. Neurology Report 2000;24(5):203. [Google Scholar]
Danzl 2013 {published data only}
- Danzl MM, Chelette KC, Lee K, Lykins D, Sawaki L. Brain stimulation paired with novel locomotor training with robotic gait orthosis in chronic stroke: a feasibility study. NeuroRehabilitation 2013;33(1):67‐76. [1053‐8135] [DOI] [PMC free article] [PubMed] [Google Scholar]
David 2006 {published data only}
- David D, Regnaux JP, Lejaille M, Louis A, Bussel B, Lofasso F. Oxygen consumption during machine‐assisted and unassisted walking: a pilot study in hemiplegic and healthy humans. Archives of Physical Medicine and Rehabilitation 2006;87:482‐9. [DOI] [PubMed] [Google Scholar]
Forrester 2016 {published data only}
- Forrester LW, Roy A, Hafer‐Macko C, Krebs HI, Macko RF. Task‐specific ankle robotics gait training after stroke: a randomized pilot study. Journal of NeuroEngineering and Rehabilitation 2016; Vol. 13, issue 51. [DOI] [PMC free article] [PubMed]
Gong 2003 {published data only}
- Gong S, Zhang J. Effect of early rehabilitation training on daily life activity of patients with hemiplegia after stroke. Chinese Journal of Clinical Rehabilitation 2003;7(5):848‐9. [Google Scholar]
Goodman 2014 {published data only}
Hesse 2001 {published data only}
- Hesse S, Werner C, Uhlenbrock D, Frankenberg S, Bardeleben A, Brandl‐Hesse B. An electromechanical gait trainer for restoration of gait in hemiparetic stroke patients: preliminary results. Neurorehabilitation and Neural Repair 2001;15:37‐48. [DOI] [PubMed] [Google Scholar]
Hsieh 2014 {published data only}
- Hsieh YW, Lin KC, Horng YS, Wu CY, Wu TC, Ku FL. Sequential combination of robot‐assisted therapy and constraint‐induced therapy in stroke rehabilitation: a randomized controlled trial. Journal of Neurology 2014, (5):1037‐45. [DOI] [PubMed] [Google Scholar]
Mirelman 2009 {published data only}
- Mirelman A, Bonato P, Deutsch JE. Effects of training with a robot‐virtual reality system compared with a robot alone on the gait of individuals after stroke. Stroke 2009;40(1):169‐74. [DOI] [PubMed] [Google Scholar]
Morone 2016 {published data only}
- Morone G, Annicchiarico R, Iosa M, Federici A, Paolucci S, Cortes U, et al. Overground walking training with the i‐Walker, a robotic servo‐assistive device, enhances balance in patients with subacute stroke: a randomized controlled trial. Journal of NeuroEngineering and Rehabilitation 2016;13(47). [DOI] [PMC free article] [PubMed] [Google Scholar]
NCT01337960 {published data only}
- NCT01337960. Ankle robotics training after stroke. clinicaltrials.gov/show/NCT01337960 (first received 15 April 2011).
Page 2008 {published data only}
- Page SJ, Levine P, Teepen J, Hartman EC. Resistance‐based, reciprocal upper and lower limb locomotor training in chronic stroke: a randomized, controlled crossover study. Clinical Rehabilitation 2008;22(7):610‐7. [DOI] [PubMed] [Google Scholar]
Park 2015 {published data only}
- Park BS, Kim MY, Lee LK, Yang SM, Lee WD, Kim J. Effects of conventional overground gait training and a gait trainer with partial body weight support on spatiotemporal gait parameters of patients after stroke. Journal of Physical Therapy Science 2015;27:1603‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Patten 2006 {published data only}
- Westlake KP, Patten C. Pilot study of Lokomat versus manual‐assisted treadmill training for locomotor recovery post‐stroke. Journal of NeuroEngineering and Rehabilitation 2009;6:18. [DOI] [PMC free article] [PubMed] [Google Scholar]
Pennati 2015 {published data only}
- Pennati GV, Re C, Messineo I, Bonaiuti D. How could robotic training and botulinum toxin be combined in chronic post stroke upper limb spasticity? A pilot study. European Journal of Physical & Rehabilitation Medicine 2015;51(4):381‐7. [1973‐9095] [PubMed] [Google Scholar]
Picelli 2015 {published data only}
- Picelli A, Chemello E, Castellazzi P, Roncari L, Waldner A, Saltuari L, et al. Combined effects of transcranial direct current stimulation (tDCS) and transcutaneous spinal direct current stimulation (tsDCS) on robot‐assisted gait training in patients with chronic stroke: a pilot, double blind, randomized controlled trial. Restorative Neurology & Neuroscience 2015;33(3):357‐68. [1878‐3627] [DOI] [PubMed] [Google Scholar]
Pitkanen 2002 {published data only}
- Pitkanen K, Tarkka I, Sivenius J. Walking training with partial body weight support versus conventional walking training of chronic stroke patients: preliminary findings. Neurorehabilitation and Neural Repair 2001;15(4; SupplementAbstracts 3rd World Conference On Neurorehabilitation; April 2‐6, 2002 Venice, Italy):312. [http://journals.sagepub.com/doi/pdf/10.1177/154596830101500402] [Google Scholar]
Richards 1993 {published data only}
- Richards CL, Malouin F, Wood‐Dauphinee S, Williams JI, Bouchard JP, Brunet D. Task‐specific physical therapy for optimization of gait recovery in acute stroke patients. Archives of Physical Medicine and Rehabilitation 1993;74:612‐20. [DOI] [PubMed] [Google Scholar]
Richards 2004 {published data only}
- Richards CL, Malouin F, Bravo G, Dumas F, Wood‐Dauphinee S. The role of technology in task‐oriented training in persons with subacute stroke: a randomized controlled trial. Neurorehabilitation and Neural Repair 2004;18(4):199‐211. [DOI] [PubMed] [Google Scholar]
Shirakawa 2001 {published data only}
- Shirakawa R, Uchida SU, Okajima YO, Sakaki TS, Shutou HS. Therapeutic effects of power‐assist training combined with biofeedback on hemiplegia by Therapeutic Exercise Machine (TEM). 1st International Congress of International Society of Physical and Rehabilitation Medicine (ISPRM); 2001 July 7‐13; Amsterdam. 2001.
Skvortsova 2008 {published data only (unpublished sought but not used)}
- Skvortsova VI, Ivanova GE, Kovrazhkina EA, Rumiantseva NA, Staritsyn AN, Suvorov AIu, et al. The use of a robot‐assisted Gait Trainer GT1 in patients in the acute period of cerebral stroke: a pilot study. Zhurnal nevrologii i psikhiatrii imeni S.S. Korsakova 2008;Suppl 23:28‐34. [PubMed] [Google Scholar]
- Skvortsova VI, Ivanova GE, Kovrazhkina EA, Rumyantseva NA, Staritsin AN, Sogomonyan EK. The efficacy of gait rehabilitation after stroke training with assistance of a robotic device gait trainer: a pilot study. International Journal of Stroke 2008;3(Suppl 3):355. [Google Scholar]
Stoller 2015 {published data only}
- Stoller O, Bruin ED, Schindelholz M, Schuster‐Amft C, Bie RA, Hunt KJ. Feedback‐controlled robotics‐assisted treadmill exercise for cardiovascular training and assessment early after severe stroke. Cerebrovascular Diseases 2015;39:107. [1015‐9770] [Google Scholar]
- Stoller O, Bruin E, Schindelholz M, Schuster‐Amft C, Bie R, Hunt KJ. Efficacy of feedback‐controlled robotics‐assisted treadmill exercise to improve cardiovascular fitness early after stroke. Journal of Neurologic Physical Therapy 2015;39(3):156. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wu 2014 {published data only}
- Wu M, Landry J, Kim J, Schmit B, Yen S, MacDonald J. Robotic resistance/assistance training improves locomotor function in individuals poststroke: A randomized controlled study. Archives of Physical Medicine and Rehabilitation 2014; Vol. 95, issue 5:799‐806. [0003: 9993] [DOI] [PMC free article] [PubMed]
References to studies awaiting assessment
Chernikova 2014 {published data only}
- Chernikova LA, Klochkov AS. The influence of physical training with the use of a Lokomat robotic system on the walking ability of the patients with post‐stroke hemiparesis. Voprosy Kurortologii, Fizioterapii i Lechebnoi Fizicheskoi Kultury 2014, issue 3:13‐7. [0042‐8787] [PubMed]
Globokar 2005 {published data only}
- Globokar D. Gait trainer in neurorehabilitation of patients after stroke. 3rd World Congress of the International Society of Physical and Rehabilitation Medicine ISPRM; 2005 April 10‐15; Sao Paulo, Brazil. Sao Paulo, Brazil, 2005:987‐1.
Golyk 2006 {published data only (unpublished sought but not used)}
- Golyk VA, Pivnyk AP, Ipatov AV. Constraint‐induced movement therapy for walking improvement (comparison of two walking training machine modifications' efficacy) for stroke patients. European Journal of Neurology 2006;13(Suppl 2):263. [Google Scholar]
Jang 2005 {published data only}
- Jang SJ, Park SW, Kim ES, Wee HM, Kim YH. Electromechanical gait trainer for restoring gait in hemiparetic stroke patients. 3rd World Congress of the International Society of Physical and Rehabilitation Medicine ISPRM; 2005 April 10‐15; Sao Paulo, Brazil. Sao Paulo, Brazil, 2005:909‐1.
Kim 2001 {published data only}
- Kim BO, Lee JJ, Cho KH, Kim SH. Gait training robot (gaiTrainer) in rehabilitation. 1st International Congress of International Society of Physical and Rehabilitation Medicine (ISPRM); 2001 July 7‐13; Amsterdam. 2001. [Abstract Th.139.P]
Kim 2014 {published data only}
- Kim JH, Park HI, Jang CH, Lim YH. Effects of robot‐assisted therapy on lower limb in geriatric patients with subacute stroke. European Geriatric Medicine 2014; Vol. 5, issue Supplement 1:S174. [DOI: ] [DOI]
Koeneman 2004 {published data only (unpublished sought but not used)}
- Koeneman JB. Air muscle device for ankle stroke rehabilitation. www.sbir.gov/sbirsearch/detail/210093 (accessed May 2017).
Mehrberg 2001 {published data only (unpublished sought but not used)}
- Mehrberg RD, Flick C, Dervay J, Carmody J, Carrington C, Jermer M. Clinical evaluation of a new over ground partial body weight support assistive device in hemiparetic stroke patients. Archives of Physical Medicine and Rehabilitation 2001;82:1293 (Abstract 10). [Google Scholar]
Ohata 2015 {published data only}
- Ohata K, Tsuboyama T, Watanabe A, Takahashi H. Gait training using new robotics device for patients with hemiplegia after stroke: a randomized cross‐over trial. Physiotherapy 2015;101:eS1123‐4. [0031‐9406] [Google Scholar]
Sale 2012 {published data only}
- NCT01678547. Robot Walking Rehabilitation in Stroke Patients. clinicaltrials.gov/show/NCT01678547 (date first received 31 August 2012. [NCT01678547]
Wu 2012 {published data only}
- Wu H, Gu XD, Fu JM, Yao YH, Li JH, Xu ZS. Effects of rehabilitation robot for lower‐limb on motor function in hemiplegic patients after stroke. National Medical Journal of China 2012, issue 37:2628‐31. [PubMed]
Yoon 2015 {published data only}
- Yoon Y, Seok TY, Yu K, Lee KJ, Kang SK, Yun SB. Gait training with the newly developed active‐assistive system for gait is feasible for hemiplegic patients after stroke. PM&R 2015; Vol. 1:S115‐6. [193‐4148]
Zhu 2016 {published data only}
- Zhu L, Song W, Liu L, Zhang R, Zhang Y. Rehabilitation effect of lower limb rehabilitation training robot combined with task‐oriented training on walking ability after stroke. Chinese Journal of Cerebrovascular Diseases 2016; Vol. 13, issue 5:240‐4 and 248. [1672‐5921]
References to ongoing studies
Louie 2015 {published data only}
- Louie DR, Eng JJ, Mortenson WB, Yao J. Use of a powered robotic exoskeleton to promote walking recovery after stroke: study protocol for a randomized controlled trial. International Journal of Stroke. 2015; Vol. 10, Supplement 4:89. [http://onlinelibrary.wiley.com/doi/10.1111/ijs.12633_2/pdf]
NCT00284115 {unpublished data only}
- Brissot R, Laviolle B. Efficacy of a mechanical gait repetitive training technique in hemiparetic stroke patients. www.clinicaltrials.gov (last accessed September 2016).
NCT00530543 {published data only}
- NCT00530543. Effects of gait training with assistance of a robot‐driven gait orthosis in hemiparetic patients after stroke. clinicaltrials.gov 2007. [DOI] [PubMed]
NCT01146587 {published data only}
- Waldner A. Robot assisted therapy for acute stroke patients: a comparative study of GangTrainer GT I, Lokomat system and conventional physiotherapy. www.clinicaltrials.gov (last accessed September 2016).
NCT01187277 {published data only}
- Chanubol R. Robotic versus conventional training on hemiplegic gait. www.clinicaltrials.gov (last accessed September 2016).
- Chanubol R, Wongphaet P, Werner C, Chavanich N, Panichareon L. Gait rehabilitation in subacute hemiparetic stroke: robot‐assisted gait training versus conventional physical therapy. Journal of the Neurological Sciences 2013; Vol. 333, issue Suppl 1:e574.
NCT01678547 {published data only}
- Sale P. Effect of robot assisted treatment on gait performance in stroke patients. www.clinicaltrials.gov (last accessed December 2012).
NCT01726998 {published data only}
- NCT01726998. Effects of locomotion training with assistance of a robot‐driven gait orthosis in hemiparetic patients after subacute stroke. www.clinicaltrials.gov 2014. [DOI] [PubMed]
NCT02114450 {published data only}
- NCT02114450. Human‐machine system for the H2 lower limb exoskeleton. www.clinicaltrials.gov 2016.
NCT02471248 {published data only}
- NCT02471248. Interactive exoskeleton robot for walking ‐ ankle joint. www.clinicaltrials.gov 2016.
NCT02483676 {published data only}
- NCT02483676. Ankle robot to reduce foot‐drop in stroke. www.clinicaltrials.gov 2016.
NCT02545088 {published data only}
- NCT02545088. New technology for individualised, intensive training of gait after stroke ‐ study II (HAL‐RCT‐II). www.clinicaltrials.gov 2015.
NCT02680691 {published data only}
- NCT02680691. Robot assisted gait training in patients with infratentorial stroke. www.clinicaltrials.gov 2016.
NCT02694302 {published data only}
- NCT02694302. Clinical trial of robot‐assisted‐gait‐training (RAGT) in stroke patients. www.clinicaltrials.gov 2016.
NCT02755415 {published data only}
- NCT02755415. Clinical applicability of robot‐assisted gait training system in acute stroke patients. www.clinicaltrials.gov 2016.
NCT02781831 {published data only}
- NCT02781831. Robot‐assisted gait training for patients with stroke. www.clinicaltrials.gov 2016.
NCT02843828 {published data only}
- NCT02843828. Gait pattern analysis and feasibility of gait training with a walking assist robot in stroke patients and elderly adults. www.clinicaltrials.gov 2016.
Additional references
Adams 1993
- Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, et al. Classification of subtype of acute ischemic stroke: definitions for use in a multicenter clinical trial (TOAST: Trial of Org 10172 in Acute Stroke Treatment). Stroke 1993;24(1):35‐41. [DOI] [PubMed] [Google Scholar]
Atkins 2004
- Atkins D, Best D, Briss PA, Eccles M, Falck‐Ytter Y, Flottorp S, et al. Grading quality of evidence and strength of recommendations. BMJ 2004;328:1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bohannon 1988
- Bohannon R. Rehabilitation goals of patients with hemiplegia. International Journal of Rehabilitation Research 1988;11(2):181‐3. [Google Scholar]
Bohannon 1991
- Bohannon RW, Horton MG, Wikholm JB. Importance of four variables of walking to patients with stroke. International Journal of Rehabilitation Research 1991;14:246‐50. [DOI] [PubMed] [Google Scholar]
Carr 2003
- Carr J, Shepherd R. Stroke Rehabilitation: Guidelines for Exercises and Training. London: Butterworth Heinemann, 2003. [Google Scholar]
Collen 1991
- Collen FM, Wade DT, Robb GF, Bradshaw CM. The Rivermead Mobility Index: a further development of the Rivermead Motor Assessment. International Disability Studies 1991;13(2):50‐4. [DOI] [PubMed] [Google Scholar]
Colombo 2000
- Colombo G, Joerg M, Schreier R, Dietz V. Treadmill training of paraplegic patients using a robotic orthosis. Journal of Rehabilitation Research and Development 2000;37(6):693‐700. [PubMed] [Google Scholar]
Deeks 2011
- Deeks JJ, Higgins JPT, Altman DG. Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Freivogel 2009
- Freivogel S, Schmalohr D, Mehrholz J. Improved walking ability and reduced therapeutic stress with an electromechanical gait device. Journal of Rehabilitation Medicine 2009;41:734–9. [DOI] [PubMed] [Google Scholar]
French 2007
- French B, Thomas LH, Leathley MJ, Sutton CJ, McAdam J, Forster A. Repetitive task training for improving functional ability after stroke. Cochrane Database of Systematic Reviews 2007, Issue 4. [DOI: 10.1002/14651858.CD006073.pub2] [DOI] [PubMed] [Google Scholar]
French 2016
- French B, Thomas LH, Coupe J, McMahon NE, Connell L, Harrison J, et al. Repetitive task training for improving functional ability after stroke. Cochrane Database of Systematic Reviews 2016, Issue 11. [DOI: 10.1002/14651858.CD006073.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- McMaster University (developed by Evidence Prime, Inc.). Available from gradepro.org. GRADEpro GDT: GRADEpro Guideline Development Tool. McMaster University (developed by Evidence Prime, Inc.). Available from gradepro.org, 2015.
Hamilton 1994
- Hamilton BB, Laughlin JA, Fiedler RC, Granger CV. Interrater reliability of the 7‐level functional independence measure (FIM). Scandinavian Journal of Rehabilitation Medicine 1994;26(3):115‐9. [PubMed] [Google Scholar]
Hesse 1999
- Hesse S, Sarkodie‐Gyan T, Uhlenbrock D. Development of an advanced mechanised gait trainer, controlling movement of the centre of mass, for restoring gait in non‐ambulant subjects. Biomedizinische Technik [Biomedical Engineering] 1999;44(7‐8):194‐201. [DOI] [PubMed] [Google Scholar]
Hesse 2003
- Hesse S, Schmidt H, Werner C, Bardeleben A. Upper and lower extremity robotic devices for rehabilitation and for studying motor control. Current Opinion in Neurology 2003;16(6):705‐10. [DOI] [PubMed] [Google Scholar]
Hesse 2010
- Hesse S, Waldner A, Tomelleri C. Innovative gait robot for the repetitive practice of floor walking and stair climbing up and down in stroke patients. Journal of NeuroEngineering and Rehabilitation 2010;7:30. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011a
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Higgins 2011b
- Schünemann HJ, Oxman AD, Vist GE, Higgins JPT, Deeks JJ, Glasziou P, et al. Chapter 12: Interpreting results and drawing conclusions. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Holden 1984
- Holden MK, Gill KM, Magliozzi MR, Nathan J, Piehl‐Baker L. Clinical gait assessment in the neurologically impaired: reliability and meaningfulness. Physical Therapy 1984;64(1):35‐40. [DOI] [PubMed] [Google Scholar]
Jorgensen 1995
- Jorgensen H, Nakayama H, Raaschou H, Olsen T. Recovery of walking function in stroke patients: the Copenhagen stroke study. Archives of Physical Medicine and Rehabilitation 1995;76:27‐32. [DOI] [PubMed] [Google Scholar]
Kelley 2013
- Kelley CP, Childress J, Boake C, Noser EA. Over‐ground and robotic‐assisted locomotor training in adults with chronic stroke: a blinded randomized clinical trial. Disability & Rehabilitation: Assistive Technology. Philadelphia, Pennsylvania: Taylor & Francis Ltd, 2013; Vol. 8, issue 2:161‐8. [1748‐3107] [DOI] [PubMed]
Kim 2008
- Kim M, Kim YH, Lee PKWY, Hyong MK, Jung PH. Effect of robot‐assisted gait therapy on cardiopulmonary fitness in subacute stroke patients. Neurorehabilitation and Neural Repair 2008;22:594. [DOI] [PubMed] [Google Scholar]
Kwakkel 1999
- Kwakkel G, Wagenaar RC, Twisk JW, Lankhorst GJ, Koetsier JC. Intensity of leg and arm training after primary middle‐cerebral‐artery stroke: a randomised trial. Lancet 1999;354(9174):191‐6. [DOI] [PubMed] [Google Scholar]
Louie 2016
- Louie DR, Eng JJ. Powered robotic exoskeletons in post‐stroke rehabilitation of gait: a scoping review. Journal of NeuroEngineering and Rehabilitation 2016; Vol. 13, issue 1:1‐10. [MEDLINE: ; 1743‐0003] [DOI] [PMC free article] [PubMed]
Mehrholz 2012a
- Mehrholz J, Pohl M. Electromechanical‐assisted gait training after stroke: a systematic review comparing end‐effector and exoskeleton devices. Journal of Rehabilitation Medicine 2012;44(3):193‐9. [DOI] [PubMed] [Google Scholar]
Mehrholz 2012b
- Mehrholz J, Hädrich A, Platz T, Kugler J, Pohl M. Electromechanical and robot‐assisted arm training for improving generic activities of daily living, arm function, and arm muscle strength after stroke. Cochrane Database of Systematic Reviews 2012, Issue 6. [DOI: 10.1002/14651858.CD006876.pub3] [DOI] [PubMed] [Google Scholar]
Mehrholz 2014
- Mehrholz J, Pohl M, Elsner B. Treadmill training and body weight support for walking after stroke. Cochrane Database of Systematic Reviews 2014, Issue 1. [DOI: 10.1002/14651858.CD002840.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
MIT 2005
- Massachusetts Institute of Technology. MIT develops Anklebot for stroke patients. web.mit.edu/newsoffice/2005/stroke‐robot.html (accessed 20 December 2005).
Nuyens 2002
- Nuyens GE, Weerdt WJ, Spaepen AJ Jr, Kiekens C, Feys HM. Reduction of spastic hypertonia during repeated passive knee movements in stroke patients. Archives of Physical Medicine and Rehabilitation 2002;83(7):930‐5. [DOI] [PubMed] [Google Scholar]
Pollock 2014
- Pollock A, Farmer SE, Brady MC, Langhorne P, Mead GE, Mehrholz J. Interventions for improving upper limb function after stroke. Cochrane Database of Systematic Reviews 2014, Issue 11. [DOI: 10.1002/14651858.CD010820.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
RevMan 2014 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Sackett 1996
- Sackett DL, Deeks JJ, Altman DG. Down with odds ratios!. Evidence‐based Medicine 1996;1:164‐6. [Google Scholar]
Schmidt 2005
- Schmidt H, Hesse S, Bernhardt R, Krüger J. HapticWalker ‐ a novel haptic foot device. ACM Transactions on Applied Perception 2005;2(2):166‐80. [Google Scholar]
States 2009
- States RA, Pappas E, Salem Y. Overground physical therapy gait training for chronic stroke patients with mobility deficits. Cochrane Database of Systematic Reviews 2009, Issue 3. [DOI: 10.1002/14651858.CD006075.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Van Peppen 2004
- Peppen RP, Kwakkel G, Wood‐Dauphinee S, Hendriks HJ, Wees PJ, Dekker J. The impact of physical therapy on functional outcomes after stroke: what's the evidence?. Clinical Rehabilitation 2004;18(8):833‐62. [DOI] [PubMed] [Google Scholar]
Veneman 2005
- Veneman J, Kruidhof R, Helm FCT, Kooy H. Design of a Series Elastic‐ and Bowdencable‐based actuation system for use as torque‐actuator in exoskeleton‐type training robots. International Conference on Rehabilitation Robotics; 2005 June 28‐July 1; Chicago (IL). 2005.
Wade 1988
- Wade DT, Collin C. The Barthel ADL Index: a standard measure of physical disability?. International Disability Studies 1988;10(2):64‐7. [DOI] [PubMed] [Google Scholar]
WHO 2006
- World Health Organization. Cerebrovascular accident, stroke. www.who.int/topics/cerebrovascular_accident/en/ (accessed 1 February 2006).
References to other published versions of this review
Mehrholz 2006
- Mehrholz J, Werner C, Kugler J, Pohl M. Electromechanical‐assisted training for walking after stroke [Protocol]. Cochrane Database of Systematic Reviews 2006, Issue 4. [DOI: 10.1002/14651858.CD006185] [DOI] [PubMed] [Google Scholar]
Mehrholz 2007
- Mehrholz J, Werner C, Kugler J, Pohl M. Electromechanical‐assisted training for walking after stroke. Cochrane Database of Systematic Reviews 2007, Issue 4. [DOI: 10.1002/14651858.CD006185.pub2] [DOI] [PubMed] [Google Scholar]
Mehrholz 2013
- Mehrholz J, Elsner B, Werner C, Kugler J, Pohl M. Electromechanical‐assisted training for walking after stroke. Cochrane Database of Systematic Reviews 2013, Issue 7. [DOI: 10.1002/14651858.CD006185.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]


