Key Points
CD37 is highly expressed across multiple types of B-cell lymphoma, representing a potential target for CAR-based therapy.
T cells expressing CD37CAR are efficiently redirected and can control B-cell lymphoma tumor progression in xenograft models.
Abstract
T cells modified to express chimeric antigen receptor (CAR) targeting CD19 (CD19CAR) have produced remarkable clinical responses in patients with relapsed/refractory B-cell acute lymphoblastic leukemia. CD19CAR T-cell therapy has also demonstrated prominent effects in B-cell non-Hodgkin lymphoma (B-NHL) patients. However, a subset of patients who relapse after CD19CAR T-cell therapy have outgrowth of CD19− tumor cells. Hence, development of alternative CARs targeting other B-cell markers represents an unmet medical need for B-cell acute lymphoblastic leukemia and B-NHL. Here, we confirmed previous data by showing that, overall, B-NHL has high expression of CD37. A second-generation CD37CAR was designed, and its efficacy in T cells was compared with that of CD19CAR. In vitro assessment of cytotoxicity and T-cell function upon coculture of the CAR T cells with different target B-cell lymphoma cell lines demonstrated comparable efficacy between the 2 CARs. In an aggressive B-cell lymphoma xenograft model, CD37CAR T cells were as potent as CD19CAR T cells in controlling tumor growth. In a second xenograft model, using U2932 lymphoma cells containing a CD19− subpopulation, CD37CAR T cells efficiently controlled tumor growth and prolonged survival, whereas CD19CAR T cells had limited effect. We further show that, unlike CD19CAR, CD37CAR was not sensitive to antigen masking. Finally, CD37CAR reactivity was restricted to B-lineage cells. Collectively, our results demonstrated that CD37CAR T cells also can effectively eradicate B-cell lymphoma tumors when CD19 antigen expression is lost and support further clinical testing for patients with relapsed/refractory B-NHL.
Visual Abstract
Introduction
The introduction of the anti-CD20 antibody rituximab as a single agent or in combination with conventional chemotherapy regimens has improved the clinical outcome for patients across multiple B-cell non-Hodgkin lymphoma (B-NHL) types, including diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and chronic lymphocytic leukemia (CLL). However, patients with primary chemotherapy refractory disease or patients who relapse often have inferior prognoses.1-3
Chimeric antigen receptor (CAR) T-cell therapy is emerging as a new treatment modality for relapsed/refractory patients. CD19-targeted CAR T cells have demonstrated remarkable response rates and induced long-term complete remissions in B-cell acute lymphoblastic leukemia (B-ALL) patients in multiple clinical trials.4-7 Recent studies have also shown efficacy against different types of B-cell lymphoma, leading to robust clinical responses7-15; however, despite initial clinical responses, a significant number of patients experience relapse.16,17 Two main types of relapses have been reported: the first type is linked to poor expansion and durability of CAR T cells in vivo, whereas the second type is linked to emergence of CD19− tumor cells.16 CARs targeting alternative B-cell–associated antigens are under development (reviewed in Fesnak et al18). This approach may help to rescue patients with CD19− tumor cell relapses or, in combination with CD19-targeted CAR (CD19CAR) T cells, may increase response rates.
CD37 is a tetraspanin membrane protein that is highly expressed on normal B cells but downregulated in plasma cells.19 Hematopoietic stem cells do not express CD37; however, low expression levels have been reported in T cells, macrophages, monocytes, dendritic cells, and natural killer (NK) cells.20,21 The biological function of CD37 has not been fully elucidated, but it might be linked to survival and apoptotic signals, as well as tumor suppression.22,23 High levels of expression have been shown across all types of B-NHL.19 Therefore, CD37 is a potential target for immunotherapy of B-cell malignancies. Several agents against CD37 are under development in phase 1 and phase 2 trials, including a naked antibody (BI836826), a homodimeric targeting peptide (otlertuzumab/TRU-016), antibodies coupled to toxins (IMGN529 and AGS67E), and a radioimmunoconjugate (177Lu-lilotomab; Betalutin).24,25 Importantly, the preclinical development of a CAR construct targeted against CD37 (CD37CAR) was recently reported and shown to be efficient in B- and T-cell malignancies.26
In this article, we present the development of a CD37CAR designed from the antibody clone HH1 and its preclinical validation using a transient expression setting. We first confirmed expression of CD37 in tumor biopsies from patients with different types of B-NHL and in B-lymphoma cell lines. We designed a second-generation CD37CAR construct and showed that it was efficiently expressed in T cells. Importantly, CD37CAR T cells demonstrated efficiency and specificity against B-cell lymphoma in vitro and in 2 mouse lymphoma xenograft models. We further studied the recently reported phenomenon of antigen masking27 and demonstrated that CD37CAR-expressing tumor cells did not become resistant to CD37CAR T cells, in contrast to what is observed with CD19CAR. We finally assessed the safety of our construct and observed a response restricted to the B-cell lineage. Taken together, our data confirm the preclinical validation reported by Maus and colleagues26 and pave the way for further clinical development of CD37CAR T-cell therapy in B-cell lymphoma.
Methods
Patient material and cell lines
The study was conducted in accordance with the Declaration of Helsinki and with approval from the Regional Committees for Medical and Health Research Ethics. Specimens were collected from 55 B-cell lymphoma patients before treatment, including FL (n = 19), DLBCL (n = 18), MCL (n = 10), marginal zone lymphoma (MZL; n = 2), and CLL (n = 6) samples. Tonsils from 15 healthy donors were also collected. The human cell lines BL-41, DAUDI, GRANTA 519, K422, K562, JEKO-1, Jurkat6, MAVER-1, MINO, RAJI, RAMOS, ROS-50, SC-1, SU-DHL6, and U2932 were obtained from Deutsche Sammlung von Mikroorganismen und Zellkulturen. The human cell lines SU-DHL4, Oci-Ly3, Oci-Ly7, and Oci-Ly10 were kind gifts from L. M. Staudt (National Cancer Institute, National Institutes of Health). Phoenix-AMPHO cells were obtained from American Type Culture Collection. All cell lines were routinely tested for the presence of mycoplasma (Venor GeM; Minerva Biolabs, Berlin, Germany). They were maintained in RPMI 1640 medium supplemented with 10% fetal calf serum (both from PAA Laboratories, Cölbe, Germany) and 0.05 mg/mL gentamycin (Garamycin; Schering-Plough Europe, Brussels, Belgium).
Immunophenotyping by flow cytometry
Patient specimens were stained with the following antibodies: CD19-BV510 (HIB19), immunoglobulin (Ig)λ–Pacific Blue (MHL-38), CD3 BV785 (OKT3) (all from BioLegend, San Diego, CA), CD20–APC–H7 (L27), CD22-APC (S-HCL-1), and Igκ–PerCPCy5.5 (G20-193) (all from BD Biosciences, San Diego, CA). CD37–Alexa Fluor 488 (HH1) was produced in-house. The cell lines were stained with CD19-PE (HIB19), CD20–Pacific Blue, CD22-FITC, and CD37–Alexa Fluor 647 (M-B371) (all from BD Biosciences), anti-CD19 (fmc63; EMD Millipore, Billerica, MA), and anti-CD37 (HH1; produced in-house); anti-mouse IgG-APC (BioLegend) was used as a secondary antibody. CD37CAR expression was detected by anti-mouse Fab antibody (Jackson ImmunoResearch, West Grove, PA), and CD19CAR was detected by biotinylated Protein L (GenScript, Piscataway, NJ) and streptavidin-PE (BD Biosciences). Flow cytometry was performed on a BD FACSCanto II or a BD LSR II (both from BD Biosciences) and analyzed with FlowJo software (TreeStar, Ashland, OR) or online Cytobank software (https://www.cytobank.org).
DNA constructs and retrovirus preparation
Total messenger RNA (mRNA) was isolated from HH1 hybridoma,28 and 5′–rapid amplification of complementary DNA ends was performed to identify the variable fragments of the heavy and light chains. A single-chain variable (scFv) construct was designed; the complete method will be published elsewhere (H.K., Elizabeth Baken, David John Warren, Geir Åge Løset, E.M.I., and S.W., manuscript in preparation). The second generation CD37CAR consists of an scFv designed as follows: leader sequence–light chain–(G4S)4–heavy chain, followed by a CD8 hinge and transmembrane domain (aa 128-210, UniProt P01732) fused to 4-1BB-CD3ζ signaling domains (aa 208-255, UniProt P07011 and aa 52-164, UniProt P20963, respectively). A codon-optimized sequence was generated and ordered (Eurofins, Hamburg, Germany). After subcloning into the Gateway system, the insert was subcloned into an mRNA synthesis vector, pCIpA102. The scFv of CD19CAR (a humanized version of the fmc63 hybridoma; a kind gift from Martin Pule, University College London, London, United Kingdom) was subcloned into our second-generation construct. We also generated a retroviral construct of the firefly luciferase-GFP (a kind gift from Rainer Löw, BioNTech IMFS, Idar-Oberstein, Germany)29 in the retroviral vector pMP71. B-lymphoma cell lines were transduced as previously described30 and sorted on the basis of GFP expression. Additionally, luciferase-GFP+ BL-41 cell lines were transduced to stably express CD19CAR or CD37CAR constructs in the same format and sorted by anti-mouse Fab.
Synthetic mRNA preparation
The in vitro transcribed (IVT) mRNA was synthesized using a RiboMA Large Scale RNA Production T7 System (Promega, Madison, WI), as previously described.31,32 Anti-Reverse Cap Analog [3′-O-Me-m7G(5′)ppp(5′)G; TriLink Biotechnologies, San Diego, CA] was used to cap the mRNA. The IVT mRNAs were qualitatively and quantitatively analyzed by agarose gel electrophoresis and NanoDrop (Thermo Fisher Scientific, Waltham, MA) prior to electroporation.
In vitro expansion and electroporation of human T cells
Human T cells were isolated from healthy donors under an approved institutional protocol and expanded with CD3/CD28 beads (Dynabeads ClinExVivo CD3/CD28; Thermo Fisher Scientific), as previously described.31,32 For electroporation, T cells were expanded for 10 days, collected, washed twice, and resuspended in CellGro DC Medium (CellGenix, Freiburg, Germany) at a final concentration of 62.5 × 106 cells per milliliter. Subsequently, the T-cell suspension was mixed with 100 µg/mL IVT mRNA (CD19CAR or CD37CAR) or only distilled water (mock) and electroporated in a 4-mm gap cuvette at 500 V and 2 milliseconds using a BTX 830 Square Wave Electroporation System (BTX Technologies, Hawthorne, NY). Immediately after electroporation, T cells were placed in complete CellGro DC Medium at 37°C in 5% CO2 overnight to allow CAR expression.
Bioluminescence-based cytotoxicity assay
The killing assay was performed as previously described.32 Briefly, luciferase-expressing target lymphoma cells were mixed with 75 µg/mL XenoLight D-Luciferin potassium salt (Perkin Elmer, Waltham, MA) and subsequently seeded in 96-well white plates in triplicate. Effector CAR T cells were added at the indicated effector-to-target (E:T) ratios and incubated in a 5% CO2 incubator at 37°C for the indicated time points. Bioluminescence (BLI) was measured with a luminometer (VICTOR Multilabel Plate Reader; Perkin Elmer) as relative light units (RLU). Triplicate wells were averaged, and lysis percentage was calculated using the following equation: percentage specific killing = 100 × (spontaneous cell death RLU − sample RLU)/(spontaneous death RLU − maximal killing RLU).
Degranulation and cytokine analysis
Effector CAR T cells and target lymphoma cells were plated in duplicates at an E:T ratio of 1:2 in complete RPMI medium in a 96-well plate. Then, PerCP-Cy5.5–labeled anti-CD107a antibody (BioLegend), GolgiStop (BD Biosciences), and GolgiPlug (BD Biosciences) were added, and the plates were incubated in a 5% CO2 incubator at 37°C for 6 hours. After incubation, cells were stained extracellularly with anti-CD3–BV421 antibody (BioLegend) and intracellularly with anti-interferon-γ–FITC and anti-tumor necrosis factor–PE/Cy7 antibodies (all from BD Biosciences), as described previously,32 and flow cytometry was performed. For cross-reactivity analysis, CAR T cells were plated in duplicates with autologous CD19+ B cells, CD3+ T cells, CD56+ NK cells, and CD14+ monocytes from 3 healthy donor peripheral blood mononuclear cells (PBMCs) and sorted with positive isolation kits (Dynabeads; Thermo Fisher Scientific) at E:T ratios of 1:2; subsequently, CD107a expression by effector CAR T cells was analyzed as mentioned above.
Mouse xenograft studies
NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) mice were bred in-house and maintained in pathogen-free conditions under an approved institutional animal care protocol. Six- to 10-week-old NSG mice were injected subcutaneously with 106 luciferase-expressing BL-41 or U2932 cells to establish flank tumors. After 4 or 12 days, 107 CAR T cells were injected intratumorally every 3 days for 2 weeks. Tumor growth was monitored by caliper.
Colony-forming cell assay
Bone marrow progenitor cells were incubated with the T cells for 14 days at 37°C, 5% CO2, and >95% humidity. Individual clusters of cells (colonies) were easily identified using a high-quality microscope. A 60-mm scoring grid was in the bottom of each dish, and all colonies were counted and divided into 3 groups: (1) colony-forming unit (CFU) erythroid with erythroblasts and/or burst-forming erythroid units, (2) CFU with granulocytes, macrophages, or cells of both lineages, and (3) CFU with erythroid-granulocyte-macrophage-megakaryocyte with erythroblasts and cells of at least 1 other lineage. Cell clusters were not counted as colonies unless >40 cells were visible.
Statistical analysis
Comparisons between 2 groups were assessed by the Student t test with Bonferroni correction or the Mann-Whitney U test. Survival studies were assessed by Kaplan-Meier curves and the log-rank (Mantel-Cox) test. Data were analyzed with Prism 6 software (GraphPad Software, La Jolla, CA). P < .05 was considered statistically significant.
Results
CD37 is widely expressed across multiple types of B-cell lymphoma
To identify relevant targets for CAR T-cell therapy, surface expression of the B-cell markers CD19, CD20, CD22, and CD37 was studied by flow cytometry analysis of viable tumor cell suspensions from FL, DLBCL, MCL, MZL, and CLL patients. B cells were identified as CD3−CD20+ cells, and malignant B cells were further identified based on Ig light chain restriction (supplemental Figure 1). B-NHL tumor cells displayed reduced expression levels of several B-cell markers, including CD19 (FL, DLBCL), CD20 (FL, CLL/MZL), and CD22 (FL, DLBCL, MCL), compared with healthy donor tonsillar B cells (Figure 1A). Interestingly, the expression level of CD37 across all types of B-NHL was high and comparable to healthy donor B cells (Figure 1A). For most of the antigens analyzed, variable expression levels were observed within non-Hodgkin lymphoma (NHL) subtypes (Figure 1A). Furthermore, tumor cell expression of CD19 and CD37 varied within individual patient samples in some cases and displayed different patterns (ie, high levels of both antigens, high levels of CD37 but low levels of CD19 or vice versa) (Figure 1B-C). Analyzing the percentage of positive tumor cells revealed that 9 of 41 cases had CD19 expression <95%, whereas only 4 cases had CD37 expression <95% (Figure 1D). We also observed variation in the expression levels of the same markers in B-cell lymphoma cell lines (supplemental Figure 2), but an overall presence of CD37. Taken together, because of its overall high expression across different types of B-NHL, the CD37 antigen is an attractive CAR target.
Figure 1.
Surface expression of CD19, CD20, CD22, and CD37 in multiple subtypes of B-NHL. Flow cytometry was used to analyze single-cell suspensions from FL, DLBCL, MCL, CLL, and MZL, as well as tonsils from healthy donors. Tumor cells were identified by gating on CD3−CD20+ B cells, followed by gating on tumor-restricted Ig light chain (Ig light chain–negative B cells in some cases). (A) Expression of CD37, CD19, CD20, and CD22 in tumor cells from NHL patients and in B cells from healthy donor tonsils. Relative protein expression was calculated through Cytobank using arcsinh transformation of median fluorescence intensity (MFI) of the cell population of interest as follows: fold change = arcsinh (MFI of protein in B cells/scale argument) − arcsinh (MFI of protein in T cells/scale argument). FL: n = 5 or n = 18 (CD19 and CD20), DLBCL: n = 18, MCL: n = 10, CLL/MZL: n = 8, tonsils: n = 8 or n = 15 (CD19 and CD20). (B) Association of CD37 and CD19 tumor cell expression levels in B-NHL. (C) Examples of CD19 and CD37 expression in tumor cells from 3 DLBCL patients. (D) Percent expression of CD19 and CD37 in individual samples from NHL patients. *P < .05, **P < .01, ****P < .0001, Mann-Whitney U nonparametric test.
CD37CAR T cells demonstrate potent in vitro activity against B-cell lymphoma cell lines
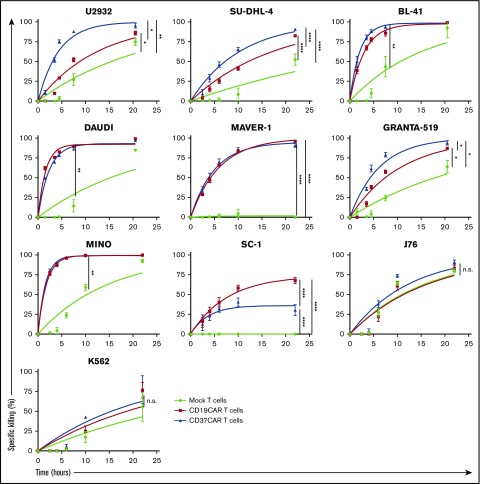
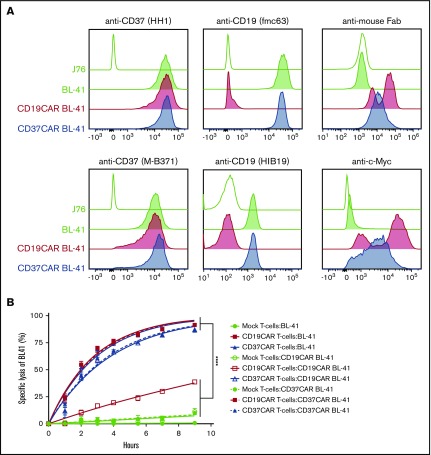
We isolated the coding sequence of the anti-CD37 antibody produced by the HH1 hybridoma.28 An scFv construct was synthesized and fused to the CD8 hinge and transmembrane domains, the 4-1BB costimulatory domain, and the CD3ζ subunit (see “Methods”). A CD19CAR containing humanized scFv from fmc63 hybridoma and an identical scaffold were prepared for comparison (Figure 2A). Electroporation of human peripheral blood T cells with mRNAs encoding these constructs resulted in high levels of CAR expression (>95% for both CARs; Figure 2B). We then evaluated their activity against several B-cell lymphoma cell lines expressing different levels of CD19 and CD37 surface proteins. CD19CAR and CD37CAR T cells generally demonstrated potent killing efficacy toward these cells (Figure 3). As expected, CD37CAR T cells outperformed CD19CAR T cells against the lymphoma cell line U2932 (Figure 3), which contained a subset of cells with loss of CD19 expression (supplemental Figure 2B). We also tested the selectivity of these constructs against CD19−/CD37− cell lines (K562 and J76) and did not observe any distinguishable differences in terms of functional activities. Importantly, as the result of variations in the luciferase levels for each target cell line and the variable donor T-cell alloreactivity (data not shown), the killing efficacy was validated only upon comparison between mock electroporated T cells and CAR T cells from the same donor. From these data, we concluded that CD37CAR T cells efficiently killed CD37+ B-cell lines with similar or higher efficiency than CD19CAR T cells, depending on the target cells’ expression levels of CD37 and CD19. CD37CAR was also potent against CD19− targets, suggesting that the loss of CD19 was not affecting CD37 presence, hence CD37CAR target recognition.
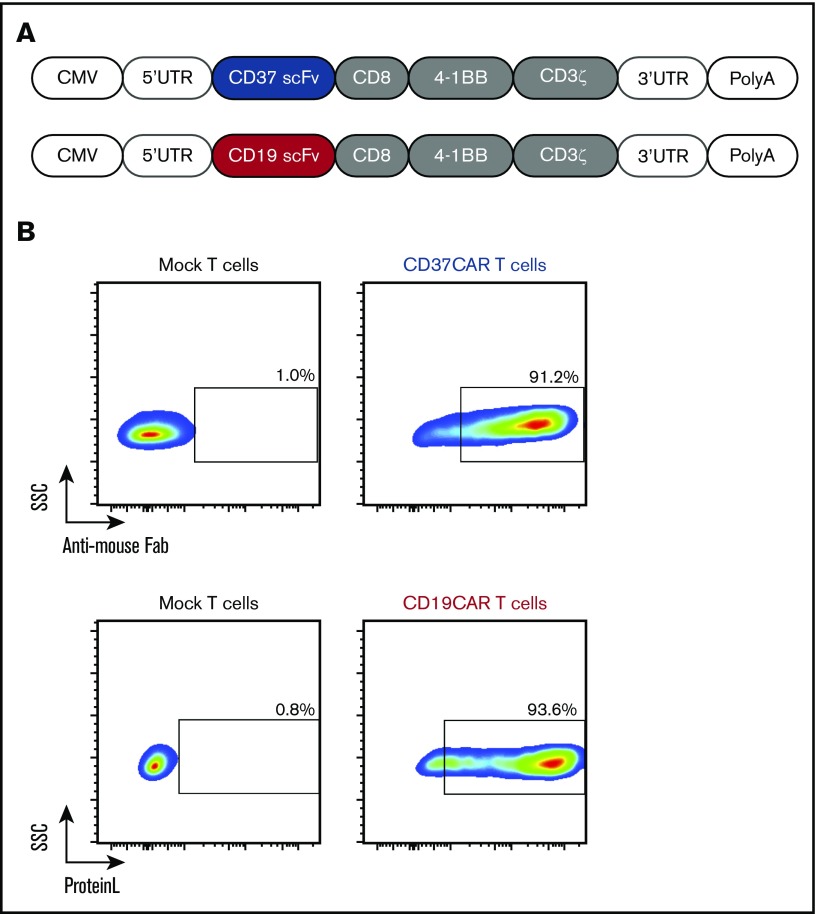
Figure 2.
Expression of CD19CAR and CD37CAR in human T cells. (A) A schematic representation of the CAR constructs. (B) mRNAs generated from CD19CAR and CD37CAR constructs (A) were electroporated into T cells. CD19CAR and CD37CAR expression was detected by Protein L staining and anti-mouse Fab antibody, respectively, 18 hours after electroporation.
Figure 3.
Comparison of CD19CAR T cells and CD37CAR T cells for antilymphoma activity in vitro. BLI-based measurement of cytotoxicity mediated by CD19CAR T cells or CD37CAR T cells when cocultured at an E:T ratio of 25:1 with B-cell lymphoma cell lines. Lysis was analyzed after 2, 4, 6, 10, or 22 hours of coculture. Data represent mean ± standard deviation of quadruplicates. Data from 1 of 2 experiments are shown. *P < .05, **P < .01, ****P < .0001, Student t test with Bonferroni correction. n.s., not significant.
CD37CAR T cells efficiently kill the CD19− subset of U2932 cells
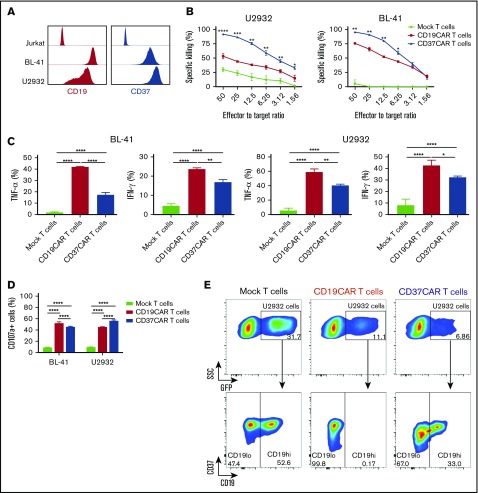
To further study CD37CAR T cells’ ability to kill B-cell lymphoma cells with reduced or missing CD19 expression, we used the CD19+/CD19− heterogeneous cell line U2932, which is homogenously CD37+ (Figure 4A; supplemental Figure 2B). As controls, we included the BL-41 cell line (CD19+/CD37+) and the T-cell line Jurkat (CD19−/CD37−). CD37CAR T cells outperformed CD19CAR T cells in terms of killing capacity against BL-41 and U2932 cell lines (Figure 4B) but displayed a similar degranulation pattern (Figure 4D). In contrast, CD19CAR T cells produced more interferon-γ and tumor necrosis factor-α than CD37CAR T cells when the CAR T cells were activated by BL-41 or U2932 cells (Figure 4C). To investigate selectivity in CAR T-cell killing, the CAR T cells were cocultured with GFP-transduced U2932 cells for 5 hours only, to allow some target cells to be detectable. This approach revealed that CD19CAR T cells restricted their killing to CD19+ cells, whereas CD37CAR T cells reacted against CD19+ and CD19− cells (Figure 4E). These results further support the potential role of CD37CAR T cells in B-NHL, especially in patients whose tumor cells contain no or low numbers of CD19 molecules.
Figure 4.
CD37CAR T cells exhibit enhanced effector functions, including specific killing of CD19−U2932 cells. (A) A representative analysis of CD19 and CD37 expression in Jurkat, BL-41, and U2932 cells by flow cytometry. (B) Specific killing mediated by CD19CAR or CD37CAR T cells against BL-41 and U2932 cells after 7 hours of coculture at the indicated E:T ratios. Data represent mean ± standard deviation of triplicates. Data from 1 of 2 experiments are shown. (C) Detection of intracellular cytokine production in CD19CAR or CD37CAR T cells after coincubation with target cells BL-41 or U2932 for 24 hours at an E:T ratio of 1:2. Data from 1 of 2 experiments are shown. Data represent mean ± standard deviation of quadruplicates. (D) Detection of degranulation in CD19CAR or CD37CAR T cells after coincubation with target cells BL-41 or U2932 for 6 hours at an E:T ratio of 1:2. Data from 1 of 2 experiments are shown. Data represent mean ± standard deviation of quadruplicates. (E) Flow cytometric analysis of CD19 and CD37 on the surface of GFP+ U2932 cells 5 hours after coculture with mock T cells, CD19CAR T cells, or CD37CAR T cells at an E:T ratio of 10:1. *P < .05, **P < .01, ***P < .001, ****P < .0001, Student t test with Bonferroni correction.
CD37CAR T cells mediate regression of established B-cell lymphoma tumors
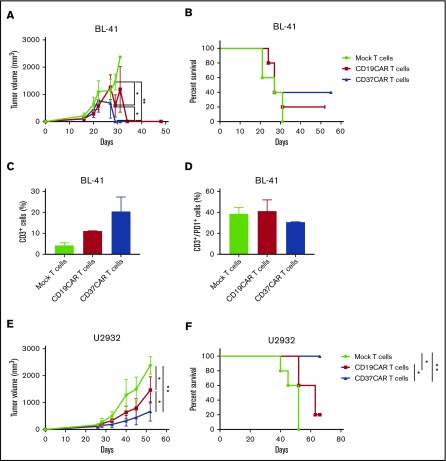
To confirm our in vitro data and validate the efficacy of CD37CAR T cells in a clinical setting, we established 2 murine xenograft models with BL-41 and U2932 lymphoma cells. We prepared the T cells as for a first-in-man trial in which the transient redirection method would be favored. Hence, a large batch of T cells was electroporated and stocked for multiple injections. We inoculated the cancer cells subcutaneously to distinguish a clear tumor burden. Mice were randomized and injected with the indicated CAR T cells or mock T cells.
When BL-41 tumors were treated, mock T cells failed to control the tumor growth, and the mice rapidly succumbed to cancer (Figure 5A-B, green group). In contrast, treatment with CD19CAR or CD37CAR T cells significantly reduced tumor growth of BL-41 tumors and tended to prolong survival (Figure 5A-B, red and blue groups, respectively). Upon analysis of the T-cell population that infiltrated the tumor tissues after 27 days of treatment, we observed more T cells in mice treated with CAR T cells than with mock T cells (Figure 5C). Because of the transient expression of CARs, tracking of CAR T cells was not possible at this time point. Further analysis did not reveal any differences in terms of expression of PD-1 exhaustion marker (Figure 5D). These data demonstrate that transient CD37CAR expression and multiple intratumoral injections were sufficient to slow the growth of an aggressive tumor as efficiently as CD19CAR-redirected T cells.
Figure 5.
CD37CAR T cells and CD19CAR T cells have comparable antitumor efficacy in vivo. NSG mice were engrafted with GFP/Luc+ BL-41 tumors or GFP/Luc+ U2932 tumors subcutaneously; 4 or 12 days, respectively, after tumor inoculation, mice were randomized and received intratumoral injections of mock T cells, CD19CAR T cells, or CD37CAR T cells (n = 5 for each group) every 3 days for 2 weeks. Tumor size was measured using a caliper. These experiments were reproduced twice. (A) BL-41 tumor growth curves after mock T cell, CD19CAR T cell, or CD37CAR T cell transfer. (B) Kaplan-Meier survival curves of mice shown in panel A. (C) Proportion of tumor-infiltrating lymphocytes, as detected by CD3 staining of tumor tissue single-cell suspension, using flow cytometry. The tumor samples were obtained at day 27. Data represent mean ± standard deviation of duplicates. (D) Percentage of tumor-infiltrating lymphocytes expressing PD-1 exhaustion marker. Data represent mean ± standard deviation of triplicates. (E) U2932 tumor growth curves after mock T cell, CD19CAR T cell, or CD37CAR T cell transfer. (F) Kaplan-Meier survival curves of mice shown in panel E. These experiments were reproduced twice. Survival curves were analyzed with a Mantel-Cox (log-rank) test. Data represent mean ± standard deviation. *P < .05, **P < .01.
We then studied the in vivo efficacy of CD37CAR T cells in NSG mice engrafted with U2932 tumors that contained a CD19− population. As expected, U2932 tumors progressively grew in the mice treated with mock T cells and killed them (Figure 5E-F, green group). CD19CAR T cells performed better than the mock cells but were not able to completely control tumor growth (Figure 5E-F, red group). In contrast, CD37CAR T cells drastically reduced tumor progression and significantly prolonged survival (Figure 5E-F, blue group). In summary, these findings demonstrate the antitumor efficacy of CD37CAR T-cell therapy in vivo and suggest that CD37CAR therapy could be an alternative to CD19CAR therapy in CD37+, but CD19− or CD19low, expressing B-NHL.
CD37CAR-expressing B cells do not prevent CD37CAR T-cell functionality
As recently reported by Ruella and colleagues,27 accidental CD19CAR expression in patient B cells led to CD19− relapse through masking of CD19 epitopes upon cis interaction with CD19CAR. We tested whether CD37CAR was also masking the target epitope when expressed in target cells. We first generated BL-41 cell lines stably expressing each CAR construct and showed that CD19CAR completely blocked CD19 surface detection without affecting CD37 signal (Figure 6A). Interestingly, CD37CAR expression in BL-41 cells did not mask CD37 or CD19 expression (Figure 6A). Importantly, these results were observed when using antibodies with the same origin as the ones used for the CAR designs or different antibody clones. We further analyzed the killing activity of CAR T cells against CAR-expressing tumor cells (Figure 6B). CD37CAR T cells mediated specific lysis, independent of whether the target cells expressed CD37CAR. This contrasted with CD19CAR T cells, whose cytotoxicity was significantly impaired by CD19CAR expression in tumor cells, again confirming the previous report.27 Moreover, when transposed to a system based on complete transient expression of the CAR (in target and effector cells), we observed similar results in terms of expression (supplemental Figure 3A-E), cytokine production (supplemental Figure 3F-G), and killing capacity (supplemental Figure 3H). These results reveal a clear safety advantage in the manufacturing of CD37CAR T cells. Further investigation will be necessary to understand the mechanism behind this effect.
Figure 6.
CD37CAR expression in BL-41 does not mask CD37. (A) Flow cytometric analysis of BL-41 cells transduced with CD19CAR or CD37CAR. Expression of CD19 and CD37 was detected by commercial antibody clones or by the corresponding antibody clones from which the CAR constructs were derived. The expression of CARs was also validated by anti-mouse Fab and anti–c-Myc antibodies. Data from 1 of 2 experiments are shown. (B) BLI-based measurement of cytotoxicity mediated by mock T cells, CD19CAR T cells, or CD37CAR T cells when cocultured at an E:T ratio of 10:1 with target cells BL-41, BL-41 CD19CAR, or BL-41 CD37CAR. Lysis was analyzed after 1, 2, 3, 4.5, 7, and 9 hours of coculture. Data represent mean ± standard deviation of quadruplicates. Representative data from 1 of 3 experiments are shown. ****P < .0001, Student t test with Bonferroni correction between effector condition and its respective negative control.
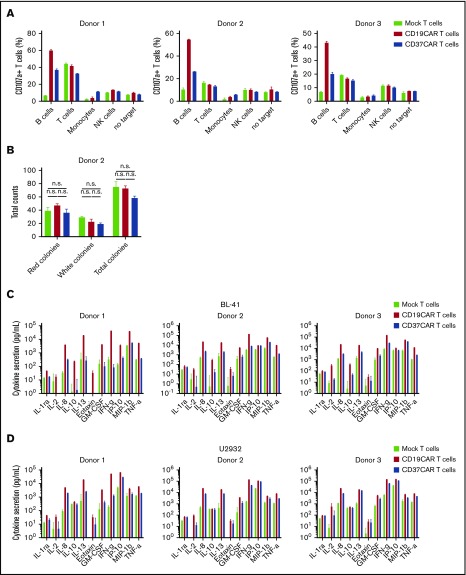
CD37CAR T cells are B-cell lineage specific
To complete our preclinical study, we also assessed the safety of CD37CAR. CD37 antigen was previously claimed to be expressed at low levels in other hematopoietic cells,21 a result that we could not confirm (data not shown). Therefore, we investigated the safety of CD37CAR T cells by testing their reactivity against autologous B cells, T cells, NK cells, and monocytes from 3 donors using the degranulation marker CD107a. We observed that CD37CAR T cells efficiently recognized B cells compared with mock T cells in all 3 donors, whereas no other cell types seemed to significantly increase stimulation (Figure 7A). CD107a was sometimes detected upon coincubation with T cells (donor 1, donor 2) or NK cells (donor 3) in all conditions (mock, CD19CAR, or CD37CAR), suggesting that these signals were unspecific. Small differences were also observed in monocytes (donor 1), but this was not above the background activity detected in the absence of target (Figure 7A). Next, we investigated the effect of CD19CAR and CD37CAR T cells on the colony-forming ability of bone marrow progenitor cells. Importantly, myeloid and erythroid colony formation in bone marrow samples was not affected following a coculture with CAR T cells at an E:T ratio of 5:1 (Figure 7B). These data show that CD37CAR T cells had no detectable off-target toxicity toward other hematopoietic lineages and suggest that killing guided by CD37CAR, as by CD19CAR, will be restricted to the B-cell lineage. Using the same donor CAR T cells, we performed Bio-Plex cytokine analysis of mock or CAR T cells after coculture with BL-41 or U2932 cells (Figure 7C-D; supplemental Figure 4). Overall, CD19CAR T cells exhibited higher cytokine secretion than CD37CAR T cells when cultured with target cells. Whether the lower cytokine secretion observed with CD37CAR T cells can also translate into reduced severity of side effects needs to be demonstrated in a clinical setting.
Figure 7.
CD37CAR T cells are B-cell lineage specific. (A) Primary CD19+ B cells, CD3+ T cells, CD14+ monocytes, and CD56+ NK cells were isolated from healthy donor PBMCs and cocultured with effector mock T cells, CD19CAR T cells, or CD37CAR T cells generated from the same donors at E:T ratios of 1:2 and 1:5. Activated T cells, used as target, were generated via CD3/CD28 stimulation. Six hours after coculture, CD107a expression on effector T cells was assessed by flow cytometry. Data represent mean ± standard deviation of duplicates. (B) Bone marrow progenitor cells were cocultured with CD19CAR or CD37CAR autologous T cells from a healthy donor for 6 hours at an E:T ratio of 5:1. The cells were then plated in semisolid methylcellulose progenitor culture for 14 days and scored for the presence of red (CFU erythroid), white (CFU with granulocytes, macrophages, or cells of both lineages), and total (CFU with granulocyte, erythroid, macrophage, megakaryocyte) colonies. Data represent mean ± standard deviation of hexaplicates. Representative data from 1 of 3 experiments are shown, P > .5 for all data. Cytokine and chemokine secretion was measured by Bio-Plex assay of supernatants from T cells from 3 healthy donors, transfected with CD19CAR or CD37CAR and activated by coculture with BL-41 cells (C) or U2932 cells (D) for 24 hours at an E:T ratio of 1:2. Data represent mean ± standard deviation of triplicates. Data from 1 of 2 experiments are shown.
Discussion
CD19CAR T-cell therapy has shown unprecedented efficacy in several clinical trials in chemoresistant B-ALL and B-cell lymphoma.33,34 These results led to US Food and Drug Administration approval, in 2017, of 2 CD19CAR products for the treatment of relapsed/refractory B-ALL and aggressive B-cell lymphoma.35,36 However, despite the fact that the majority of these patients initially responded to the therapy, a significant subset of them exhibited relapses characterized by the apparent loss of CD19 antigen expression.16,37 This may be an important tumor-escape mechanism, rendering the malignant B cells refractory to CD19-directed CAR T cells. The number of relapsed patients in B-cell lymphoma might also increase, because the follow-up times are still short. Thus, strategies designed to circumvent this escape mechanism would potentially improve patient outcomes.
A logical approach would be to target other surface markers expressed on malignant B cells, including CD20, CD22, CD123, or Ig-tumor restricted light chain (Igκ or Igλ). Clinical trials are underway with CD20- and CD22-targeted CAR T cells.38 Interestingly, CD22-targeted CAR T cells have shown clinical efficacy in B-ALL patients who developed resistance to anti-CD19 immunotherapy.39 The majority of these patients experienced relapse associated with reduced expression of CD22 on escaping clones. A recent study has shown that CAR T cells with dual targeting of CD19 and CD123 are able to induce regression of CD19− blasts in a preclinical model of B-ALL.40 Another emerging resistance mechanism, which has been reported in B-cell lymphoma, is upregulation of PD-L1 on tumor-associated macrophages and tumor cells after CD19CAR T cell therapy.10 Thus, combining CAR T-cell therapy with immune checkpoint blockade against the PD-1/PD-L1 axis could improve efficacy and overcome resistance following engineered T-cell therapy for B-cell lymphoma patients.
CD37 recently regained attention as a promising target in B-cell lymphoma but typically not for B-ALL or multiple myeloma, because the antigen is absent in early stages of B-cell differentiation and is also reduced in plasma cells.20,21 Here, we have shown that CD37 is highly expressed across FL, DLBCL, MCL, MZL, and CLL, which constitute the majority of B-NHLs. This is in contrast to a previous study reporting loss of CD37 expression in 60% of DLBCL patients, which was predictive of decreased survival.41 The study assessed a much larger number of patients than we did; however, all DLBCL samples tested were positive. The discrepancy could be due to a difference in epitope recognition or the dissimilar detection methods. The uniform and high expression would imply that CD37 could become an important target for immunotherapy in B-cell lymphoma. Screening of tumor specimens from B-cell lymphoma patients also revealed that some lymphoma cells were relatively high in CD37 expression and low in CD19 expression or vice versa. Given our observation that efficacy of CAR T cell therapy correlated with target antigen expression, this type of screening may become crucial to identify the patient groups that are likely to benefit from a targeted therapy.
To study the role of CD37CAR T cells in a model resembling relapse due to CD19 loss, we used the U2932 cell line, which consisted of 2 populations (CD19− and CD19+). Both subsets showed a uniform high expression of CD37. By using this cell line, we were able to show that CD37CAR T cells effectively killed U2932 cells and significantly prolonged survival in mice with established tumors, irrespective of CD19 expression. CD19+ U2932 cells harbor BCL2 amplification and BCL6 translocation, whereas CD19− cells harbor BCL2 amplification and MYC translocation.42 The latter subset may represent a good model for double-hit lymphoma, which is known to have an inferior outcome with standard therapies.43-45 Hence, our demonstration that CD37CAR T cells efficiently eradicated U2932 tumors is promising and should prompt further investigation of the usefulness of this novel concept in the treatment of hard-to-treat lymphomas.
Weak expression of CD37 by T cells, monocytes, macrophages, dendritic cells, and NK cells has been reported,21 but we could not confirm these data using different anti-CD37 antibodies (data not shown). One can speculate that some cells might express it in a cyclic way or under conditions that we did not test in our set-up. Our safety assessment against autologous PBMCs does not exclude CD37 expression in non-B cells, but the lack of CD37CAR T-cell reactivity suggests reactivity specifically toward B-lineage cells. In addition, CD37CAR T cells had no effect on CFU formation from bone marrow progenitors. We also observed some background activity with CD19CAR and CD37CAR, which we attribute to enhanced tonic signaling reminiscent of the overexpression of the CAR. Indeed, the presence of signaling domains, such as CD3ζ and CD28 or 4-1BB, have been shown by other investigators to influence T-cell biology, even in the absence of antigen stimulation.46,47 Although our data do not indicate cross-reactivity with other immune cells, unwanted off-target toxicity cannot be excluded before testing CD37CAR T cells in patients. Importantly, anti-CD37 antibody therapy, using the same antibody clone from which our CD37CAR construct is derived, conjugated with the isotope 177Lu, is currently under investigation in clinical phase 1/2 trials and was demonstrated to be safe and well tolerated.48 Furthermore, treatment with the anti-CD37 monoclonal antibody otlertuzumab, in combination with bendamustine, was well tolerated in refractory CLL patients.49 Still, to improve safety for first-in-man clinical studies, transient CD37CAR expression (mRNA electroporation) could be a valid option to evaluate potential off-tumor on-target toxicities in a more controlled manner.50,51
In summary, we describe a novel strategy for the treatment of B-cell lymphoma by targeting CD37 via CAR T cells. Another group has recently presented a CD37CAR construct and reached the same conclusions.26 Interestingly, they also demonstrated a potential usefulness for CD37CAR in the treatment of CD37+ T-cell lymphoma.26 CD37CAR T cells can eradicate B-cell lymphoma tumors, including those resistant to CD19-directed CAR T cells due to loss of CD19 antigen expression. Our present data are in line with their work, which supports the testing of CD37CAR in a clinical setting. Finally, in contrast to antigen masking, as observed for CD19CAR and CD22CAR,27 CD37CAR coexpression with CD37 did not affect CD37 detection. Thus, our data warrant clinical development of CD37CAR T cells in patients with recurrent and refractory B-NHL.
Supplementary Material
The full-text version of this article contains a data supplement.
Acknowledgments
The authors thank Hilde Almåsbak (Thermo Fisher Scientific) for valuable advice on the design of the experiments. They also thank the team at the Flow Cytometry Core Facility (Department of Core Facilities, Institute for Cancer Research) for their great support.
This work was supported by grants from the Research Council of Norway (244388 and 230817/F20), the Norwegian Cancer Society (6829007) (S.W.) and South-Eastern Norway Regional Health Authority (2016006 [H.K.] and 2017075 [S.M.M.]).
Authorship
Contribution: H.K. and P.D. designed and oversaw studies, conducted experiments, analyzed and interpreted data, and wrote the manuscript; S.E.J., S.M.M., S.P., and A.F. performed experiments and analyzed data; S.E.J., Y.N.B., K.H., E.M.I., and J.H.M. handled patient material and interpreted data; K.B., G.K., H.H., A.K., and E.B.S. provided key experimental material and interpreted data; J.H.M. and E.M.I analyzed and interpreted data and edited the manuscript; and S.W. designed and oversaw studies, analyzed and interpreted data, and edited the manuscript; and all authors critically reviewed the manuscript prior to submission.
Conflict-of-interest disclosure: E.B.S. owns stock in Nordic Nanovector. H.H., G.K., E.B.S., J.H.M., E.M.I., and S.W. have applied for a patent related to the results. The remaining authors declare no competing financial interests.
Correspondence: Sébastien Wälchli, Department of Cellular Therapy, Radiumhospitalet-OUS, Ullernchausséen 70, 0379 Oslo, Norway; e-mail: sebastw@rr-research.no.
References
- 1.Coiffier B, Lepage E, Briere J, et al. . CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346(4):235-242. [DOI] [PubMed] [Google Scholar]
- 2.Marcus R, Imrie K, Solal-Celigny P, et al. . Phase III study of R-CVP compared with cyclophosphamide, vincristine, and prednisone alone in patients with previously untreated advanced follicular lymphoma. J Clin Oncol. 2008;26(28):4579-4586. [DOI] [PubMed] [Google Scholar]
- 3.Forstpointner R, Dreyling M, Repp R, et al. ; German Low-Grade Lymphoma Study Group. The addition of rituximab to a combination of fludarabine, cyclophosphamide, mitoxantrone (FCM) significantly increases the response rate and prolongs survival as compared with FCM alone in patients with relapsed and refractory follicular and mantle cell lymphomas: results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood. 2004;104(10):3064-3071. [DOI] [PubMed] [Google Scholar]
- 4.Maude SL, Laetsch TW, Buechner J, et al. . Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med. 2018;378(5):439-448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maude SL, Frey N, Shaw PA, et al. . Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Davila ML, Riviere I, Wang X, et al. . Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med. 2014;6(224):224ra25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee DW, Kochenderfer JN, Stetler-Stevenson M, et al. . T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385(9967):517-528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Neelapu SS, Locke FL, Bartlett NL, et al. . Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377(26):2531-2544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kochenderfer JN, Somerville RPT, Lu T, et al. . Lymphoma remissions caused by anti-CD19 chimeric antigen receptor T cells are associated with high serum interleukin-15 levels. J Clin Oncol. 2017;35(16):1803-1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schuster SJ, Svoboda J, Chong EA, et al. . Chimeric antigen receptor T cells in refractory B-cell lymphomas. N Engl J Med. 2017;377(26):2545-2554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Turtle CJ, Hanafi LA, Berger C, et al. . Immunotherapy of non-Hodgkin’s lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor-modified T cells. Sci Transl Med. 2016;8(355):355ra116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Locke FL, Neelapu SS, Bartlett NL, et al. . Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang X, Popplewell LL, Wagner JR, et al. . Phase 1 studies of central memory-derived CD19 CAR T-cell therapy following autologous HSCT in patients with B-cell NHL. Blood. 2016;127(24):2980-2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kochenderfer JN, Dudley ME, Feldman SA, et al. . B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood. 2012;119(12):2709-2720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schuster SJ, Bishop MR, Tam CS, et al. ; JULIET Investigators. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380(1):45-56. [DOI] [PubMed] [Google Scholar]
- 16.Ruella M, Maus MV. Catch me if you can: leukemia escape after CD19-directed T cell immunotherapies. Comput Struct Biotechnol J. 2016;14:357-362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Orlando EJ, Han X, Tribouley C, et al. . Genetic mechanisms of target antigen loss in CAR19 therapy of acute lymphoblastic leukemia. Nat Med. 2018;24(10):1504-1506. [DOI] [PubMed] [Google Scholar]
- 18.Fesnak AD, June CH, Levine BL. Engineered T cells: the promise and challenges of cancer immunotherapy. Nat Rev Cancer. 2016;16(9):566-581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Barrena S, Almeida J, Yunta M, et al. . Aberrant expression of tetraspanin molecules in B-cell chronic lymphoproliferative disorders and its correlation with normal B-cell maturation. Leukemia. 2005;19(8):1376-1383. [DOI] [PubMed] [Google Scholar]
- 20.van Spriel AB, Sofi M, Gartlan KH, et al. . The tetraspanin protein CD37 regulates IgA responses and anti-fungal immunity. PLoS Pathog. 2009;5(3):e1000338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.de Winde CM, Zuidscherwoude M, Vasaturo A, van der Schaaf A, Figdor CG, van Spriel AB. Multispectral imaging reveals the tissue distribution of tetraspanins in human lymphoid organs. Histochem Cell Biol. 2015;144(2):133-146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lapalombella R, Yeh YY, Wang L, et al. . Tetraspanin CD37 directly mediates transduction of survival and apoptotic signals. Cancer Cell. 2012;21(5):694-708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.de Winde CM, Veenbergen S, Young KH, et al. . Tetraspanin CD37 protects against the development of B cell lymphoma. J Clin Invest. 2016;126(2):653-666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bertoni F, Stathis A. Staining the target: CD37 expression in lymphomas. Blood. 2016;128(26):3022-3023. [DOI] [PubMed] [Google Scholar]
- 25.de Winde CM, Elfrink S, van Spriel AB. Novel insights into membrane targeting of B cell lymphoma. Trends Cancer. 2017;3(6):442-453. [DOI] [PubMed] [Google Scholar]
- 26.Scarfò I, Ormhøj M, Frigault MJ, et al. . Anti-CD37 chimeric antigen receptor T cells are active against B- and T-cell lymphomas [published correction appears in Blood. 2018;132(23):2527]. Blood. 2018;132(14):1495-1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ruella M, Xu J, Barrett DM, et al. . Induction of resistance to chimeric antigen receptor T cell therapy by transduction of a single leukemic B cell. Nat Med. 2018;24(10):1499-1503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smeland E, Funderud S, Ruud E, Kiil Blomhoff H, Godal T. Characterization of two murine monoclonal antibodies reactive with human B cells. Their use in a high-yield, high-purity method for isolation of B cells and utilization of such cells in an assay for B-cell stimulating factor. Scand J Immunol. 1985;21(3):205-214. [DOI] [PubMed] [Google Scholar]
- 29.Loew R, Heinz N, Hampf M, Bujard H, Gossen M. Improved Tet-responsive promoters with minimized background expression. BMC Biotechnol. 2010;10(1):81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wälchli S, Løset GA, Kumari S, et al. . A practical approach to T-cell receptor cloning and expression. PLoS One. 2011;6(11):e27930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Inderberg EM, Wälchli S, Myhre MR, et al. . T cell therapy targeting a public neoantigen in microsatellite instable colon cancer reduces in vivo tumor growth. OncoImmunology. 2017;6(4):e1302631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Walseng E, Köksal H, Sektioglu IMM, et al. . A TCR-based chimeric antigen receptor. Sci Rep. 2017;7(1):10713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Aldoss I, Bargou RC, Nagorsen D, Friberg GR, Baeuerle PA, Forman SJ. Redirecting T cells to eradicate B-cell acute lymphoblastic leukemia: bispecific T-cell engagers and chimeric antigen receptors. Leukemia. 2017;31(4):777-787. [DOI] [PubMed] [Google Scholar]
- 34.Brudno JN, Kochenderfer JN. Chimeric antigen receptor T-cell therapies for lymphoma. Nat Rev Clin Oncol. 2018;15(1):31-46. [DOI] [PubMed] [Google Scholar]
- 35.Mullard A. FDA approves first CAR T therapy. Nat Rev Drug Discov. 2017;16(10):669. [DOI] [PubMed] [Google Scholar]
- 36.Roberts ZJ, Better M, Bot A, Roberts MR, Ribas A. Axicabtagene ciloleucel, a first-in-class CAR T cell therapy for aggressive NHL. Leuk Lymphoma. 2018;59(8):1785-1796. [DOI] [PubMed] [Google Scholar]
- 37.Brudno JN, Stroncek D, Pittaluga S, et al. . T cells expressing a novel fully-human anti-cd19 chimeric antigen receptor induce remissions of advanced lymphoma in a first-in-humans clinical trial [abstract]. Blood. 2016;128(22). Abstract 999. [Google Scholar]
- 38.Hartmann J, Schüßler-Lenz M, Bondanza A, Buchholz CJ. Clinical development of CAR T cells-challenges and opportunities in translating innovative treatment concepts. EMBO Mol Med. 2017;9(9):1183-1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fry TJ, Shah NN, Orentas RJ, et al. . CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat Med. 2018;24(1):20-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ruella M, Barrett DM, Kenderian SS, et al. . Dual CD19 and CD123 targeting prevents antigen-loss relapses after CD19-directed immunotherapies. J Clin Invest. 2016;126(10):3814-3826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xu-Monette ZY, Li L, Byrd JC, et al. . Assessment of CD37 B-cell antigen and cell of origin significantly improves risk prediction in diffuse large B-cell lymphoma. Blood. 2016;128(26):3083-3100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Quentmeier H, Amini RM, Berglund M, et al. . U-2932: two clones in one cell line, a tool for the study of clonal evolution. Leukemia. 2013;27(5):1155-1164. [DOI] [PubMed] [Google Scholar]
- 43.Valera A, López-Guillermo A, Cardesa-Salzmann T, et al. ; Grup per l’Estudi dels Limfomes de Catalunya i Balears (GELCAB). MYC protein expression and genetic alterations have prognostic impact in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Haematologica. 2013;98(10):1554-1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Green TM, Young KH, Visco C, et al. . Immunohistochemical double-hit score is a strong predictor of outcome in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Clin Oncol. 2012;30(28):3460-3467. [DOI] [PubMed] [Google Scholar]
- 45.Herrera AF, Mei M, Low L, et al. . Relapsed or refractory double-expressor and double-hit lymphomas have inferior progression-free survival after autologous stem-cell transplantation. J Clin Oncol. 2017;35(1):24-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Long AH, Haso WM, Shern JF, et al. . 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med. 2015;21(6):581-590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Frigault MJ, Lee J, Basil MC, et al. . Identification of chimeric antigen receptors that mediate constitutive or inducible proliferation of T cells. Cancer Immunol Res. 2015;3(4):356-367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kolstad A, Beasley M, Bayne M, et al. . 177 Lu-Lilotomab Satetraxetan, a novel CD37-targeted antibody-radionuclide conjugate in relapsed non-Hodgkin’s lymphoma (NHL): updated results of an ongoing phase I/II study (LYMRIT 37-01) [abstract]. Blood. 2017;130(suppl 1). Abstract 2769. [Google Scholar]
- 49.Robak T, Hellmann A, Kloczko J, et al. . Randomized phase 2 study of otlertuzumab and bendamustine versus bendamustine in patients with relapsed chronic lymphocytic leukaemia. Br J Haematol. 2017;176(4):618-628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Beatty GL, Haas AR, Maus MV, et al. . Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol Res. 2014;2(2):112-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Svoboda J, Rheingold SR, Gill SI, et al. . Nonviral RNA chimeric antigen receptor-modified T cells in patients with Hodgkin lymphoma. Blood. 2018;132(10):1022-1026. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.