Abstract
Background
Inhaled corticosteroids (ICS) form the basis of maintenance therapy in asthma and their efficacy is well established. However, the optimal starting dose of ICS is not clearly established. Recent reviews demonstrate a relatively flat efficacy curve for ICS and increasing side effects with increasing ICS doses. High doses are frequently prescribed and there are now reports of significant side effects occurring with high dose ICS use. These issues demonstrate the need to establish the optimal starting dose of ICS in asthma.
Objectives
To establish the optimal starting dose of ICS by evaluating the efficacy of initial high dose ICS with low dose ICS in subjects with asthma, not currently on ICS.
Search methods
We searched the Cochrane Airways Group trials register and reference lists of articles.Date of last search: January 2003
Selection criteria
Randomised controlled trials of two different doses of the same ICS in adults and children with asthma with no concomitant ICS or OCS.
Data collection and analysis
Trial quality was assessed and data were extracted independently by two reviewers. Study authors were contacted for confirmation. Trials were analysed according to the following ICS dose comparisons: step down vs constant dose ICS (n=7); high vs moderate (n=11); high vs low (n=9); moderate vs low (n=11); fold change in dose (all studies).
Main results
31 papers reporting the results of 26 trials were included in the review. For studies that compared a step down approach to a constant moderate/low ICS dose, there were no significant differences in lung function, symptoms, rescue medications or asthma control between the two treatment approaches. Significant but clinically small improvements in percent predicted FEV1 ( WMD 5.32, 95% CI 0.65 to 9.99) and non significant improvements in the change in morning PEF were found for high dose ICS compared to moderate dose ICS. There were no significant differences in efficacy between high and low dose ICS. For moderate dose ICS, compared to low dose ICS, there were significant improvements in the change in morning PEF L/min from baseline (WMD 11.14, 95% CI 1.34 to 20.93) and nocturnal symptoms (SMD ‐0.29, 95% CI ‐0.53 to ‐0.06 ) . Commencing ICS at double or quadruple a base moderate or low dose had no greater effect than commencing with the base dose. Several studies reported greater improvement in airway hyperresponsiveness for high dose ICS.
Authors' conclusions
For patients with asthma who require ICS, commencing with a moderate dose ICS is equivalent to commencing with a high dose ICS and down‐titrating. The small significant benefits of commencing with a high ICS dose are not of sufficient clinical benefit to warrant its use when compared to moderate or low dose ICS. Initial moderate ICS dose appears to be more effective than initial low ICS dose. High dose ICS may be more effective than moderate or low dose ICS for airway hyperresponsiveness. There is no benefit in doubling or quadrupling ICS in subjects with stable asthma.
Plain language summary
High dose versus low dose inhaled corticosteroid as initial starting dose for asthma in adults and children
Guidelines vary in their recommendations for the initial dose of inhaled corticosteroid (ICS) for asthma. This review compared initial ICS doses for asthma . The results showed that commencing with a moderate dose ICS is as effective as commencing with a high dose ICS and then reducing the dose whilst monitoring symptoms. These results also show that initial moderate dose ICS maybe more effective than initial low dose ICS. No extra benefit was gained by doubling or quadrupling the starting ICS dose. People with asthma should start their treatment with low to moderate doses of ICS.
Background
The use of inhaled corticosteroids (ICS) is very effective in reducing mortality and morbidity from asthma (Suissa 2001). Inhaled corticosteroids form the basis of maintenance therapy in asthma (NAC 2002; NIH 2002), and target the airway inflammatory process that is a key pathophysiological characteristic of asthma (Barnes 1998). Systematic reviews of randomised controlled trials of inhaled corticosteroid use have established their efficacy as maintenance treatment of asthma (Adams 2002; Adams 2002 (b); Adams 2002 (c)). However, the optimal starting dose of ICS is not clearly established.
A systematic review has shown that, with increasing doses of inhaled corticosteroids, there is an increase in side effects (Adams 2002). Reviews also demonstrate that there is a relatively flat efficacy curve for inhaled corticosteroid use where greater than 90% of the benefit is achieved at low doses (e.g. fluticasone propionate 250 mcg/day) (Holt 2001; Adams 2002; Powell 2003). Very high doses of inhaled corticosteroids are frequently prescribed in clinical practice (Black 2000) and there are now reports of significant side‐effects occurring with high dose inhaled corticosteroid use (Todd 2002). These issues demonstrate the need to establish the optimal starting dose of inhaled corticosteroid in asthma.
The Global Initiative against Asthma (GINA) guidelines for asthma management recommend a wide range of starting doses, between 200 to 1000 mcg beclomethasone‐equivalent per day (NIH 2002). Some guidelines recommend starting with a high dose of inhaled corticosteroid and then back‐titrating the dose (step down) (NAC 2002). In contrast, other guidelines either do not specify a starting dose (NIH 2002) or recommend starting with moderate to low doses of inhaled corticosteroid (BTS/SIGN 2003; NZGG 2003). Randomised controlled trials have compared the effect of starting with high dose inhaled corticosteroid, with some studies then back titrating, or starting with low dose inhaled corticosteroid. This review aims to assess the efficacy of initial inhaled corticosteroid doses for the treatment of asthma.
Objectives
The objective of this systematic review is to establish the optimal starting dose of inhaled corticosteroid by evaluating the efficacy of constant high dose, constant moderate dose, constant low dose, and initial high dose inhaled corticosteroid followed by low dose inhaled corticosteroids (step down) in subjects with asthma, not currently treated with inhaled corticosteroids.
Methods
Criteria for considering studies for this review
Types of studies
Prospective randomised controlled trials only were eligible for inclusion in the review. Trials using either a parallel or crossover design and double, single or unblinded studies were considered.
Types of participants
Adults and children with non‐oral steroid dependent asthma (defined by doctor's diagnosis, objective lung function or according to American Thoracic Society guidelines).
Types of interventions
Interventions that compared two different doses of the same inhaled corticosteroid with no concomitant oral or inhaled corticosteroid therapy, and no recent use of inhaled corticosteroids (at least 1 month) were permitted. Treatment duration was required to be a minimum of 4 weeks. Delivery may be by metered dose inhaler (pMDI) with or without a valved holding device (spacer), dry powder inhaler (DPI) or nebuliser.
Types of outcome measures
The following outcomes were evaluated: asthma symptoms, lung function, exacerbations, unscheduled visits to the doctor, ER visits, hospitalisation, airway hyperresponsiveness, asthma control.
Search methods for identification of studies
Electronic searches
Cochrane Airways Group's register derived from MEDLINE, EMBASE, CINAHL, hand searched respiratory journals and meeting abstracts. The register was searched using the following terms:
(asthma OR wheez*) AND (step* OR high* dose OR low* dose) AND (inhaled corticoster* OR beclometh* OR triamcin* OR flutic* OR budes* OR betameth* OR flunis* OR cicles* OR momet*)
Searching other resources
Full text versions of the relevant papers were obtained, and their bibliographic lists hand searched for additional articles.
Data collection and analysis
Selection of studies
The full text version of each potentially relevant article was obtained for assessment by two independent reviewers to establish whether it met the inclusion criteria.
Data extraction and management
Two authors extracted data from the studies independently. Study investigators were contacted to verify and provide further information about methodological approaches and outcomes data.
Assessment of risk of bias in included studies
Study quality was assessed independently and scored by two reviewers using two instruments. The first, the Jadad system (Jadad 1996), allows for a score between 0 and 5 with higher scores indicating a better description of the study. The Jadad system measures the clarity of the description of: randomisation (1 point, 2 points if well described and appropriate), blinding (1point, 2 points if well described and appropriate) and treatment of withdrawals and dropouts (1 point). One point is deducted if the method of randomisation or blinding is inappropriate. Studies were further assessed, using the Cochrane method, as "Adequate", "Inadequate" or "Unclear" according to the actual methods used for randomisation and concealment of allocation. In this assessment, if studies are either not truly randomised (e.g. alternated) or if allocation to treatment or control groups is not truly blinded, studies are considered "inadequate". If the author does not fully state these methods, the study is characterised as "unclear" until the author is contacted and clarification can be made.
Data synthesis
Outcomes were analysed as continuous and/or dichotomous variables, using standard statistical techniques. I) For continuous outcomes, the weighted mean difference or standardised mean difference with 95% confidence intervals was calculated as appropriate.
ii) For dichotomous outcomes, the relative risk was calculated with 95% confidence interval. Heterogeneity was examined using a Chi‐squared test and reasons for heterogeneity explored where appropriate. Equivalence between doses was considered to be present when the 95% confidence limit for the effect size was within the limits of clinical significance for the outcome of interest. These values were: PEF 50L/min; FEV1 200ml or 10% predicted.
Five primary comparisons were made. These were determined by the strength of the ICS dose and the type of intervention ‐ step down or constant dose being compared. ICS dose was classified according to British Thoracic Society guidelines (BTS/SIGN 2003) as: a) High dose ‐ Adults: > 400 mcg/day of fluticasone propionate (FP) or > 800 mcg/day budesonide (BUD) or beclomethasone (BDP) ‐ Children:> 200 mcg/day FP or > 400 mcg/day BUD or 400 mcg BDP. b) Moderate dose ‐ Adults: from 200 up to (but not including) 400 mcg/day FP or from 400 up to (but not including) 800 mcg/day BUD or BDP. ‐ Children: from 100 up to (but not including) 200 mcg/day FP or from 200 up to (but not including) 400 mcg/day BUD or BDP. c) Low dose ‐ < 200 mcg/day FP or < 400 mcg/day BUD or BDP. ‐ Children: < 100 mcg/day FP or < 200 mcg/day BUD or BDP.
The primary comparisons were: 1) Step Down vs Constant ICS Dose 2) High vs Moderate ICS Dose 3) High vs Low ICS Dose 4) Moderate vs Low ICS Dose 5) Fold difference in ICS dose: two or four fold increased ICS vs base ICS dose
Subgroup analysis and investigation of heterogeneity
Outcomes were analysed according to: I) Delivery device (nebuliser / pMDI / DPI) ii) Age (infants / children / adults) iii) Design (parallel / crossover) iv) Duration of treatment (<= 12 weeks / > 12 weeks) v) ICS type
Results
Description of studies
31 publications describing 26 RCTs met the inclusion criteria for the review. Full details of the studies are provided in the characteristics of the included studies table. Disagreement on eligibility for inclusion was resolved by consensus. Agreement was 94%, kappa 0.88.
PARTICIPANT CHARACTERISTICS
Twenty studies had a multicentre design. Three of these were conducted in general practice, one in a medical centre and eight in outpatient clinics. The remaining eight multicentre trials did not state where they were conducted. Two trials were conducted as single centre outpatient trials and the remaining four did not state where the trial was conducted.
Participants in seven studies were ICS naive. In four studies the inclusion criteria included no current ICS use and 15 studies excluded potential participants who had used ICS in the previous 4 weeks to six months prior to commencement of the study. One trial (Hofstra 2000) allowed participants to use intranasal ICS.
The majority of studies were conducted in adults (n = 17) with five studies conducted in children (Jonasson 1998; Hofstra 2000; Pedersen 1995; Nayak 2002; Visser 2001) and four in infants (Bisgaard 1993; Bisgaard 1999; Volovitz 1998; Wennergren 1996).
INTERVENTION
The majority of studies (n = 15) were comparisons of BUD dose. Seven studies compared different doses of FP, three studies compared beclomethasone (BDP) doses and one study incorporated both BDP and FP comparisons (Szefler 2002). In six studies, three arms of three doses of the same ICS were compared (Jonasson 1998; Miyamoto 2000; Pedersen 1995; Sheffer 1996; Szefler 2002; Wasserman 1996). Two studies included a third treatment arm of another asthma medication. This included theophylline (Pedersen 1996) and salbutamol (Majima 1993). A placebo arm as well as dose comparison was included in 15 studies.
DESIGN
Parallel group design was used in 24 studies. One of these studies, (Szefler 2002), had one arm each using FP in one and BDP in the other. These ICS doses were increased every six weeks for 18 weeks, and then a three week run out period on high dose FP. This study was not included in any meta analysis due to the differing study design. The two remaining studies were of crossover design (Bisgaard 1993; Pedersen 1995). These two studies had three arms with three four week treatment periods. Pedersen 1995 incorporated a two week run in before commencement of the intervention. Neither of these studies used a washout between treatment periods. These two studies (Bisgaard 1993; Pedersen 1995) did not provide sufficient data to be included in any meta analysis.
DOSE
Seven studies incorporated a step down versus constant ICS dose. Six of these used BUD, 800‐1600 mcg/day reducing to 200 mcg/day compared to a constant dose of 200‐400 mcg /day for the four adult trials (Campbell 1998; Chanez 2001; Pirozynski 1996; van der Molen 1998) and nebulised BUD 2 mg reducing to 0.5 mg vs 0.5 mg in the infant studies (Volovitz 1998; Wennergren 1996). One study compared FP 1000 mcg/day reducing to 100 mcg /day vs 200 mcg/day (Visser 2001).
Eleven studies compared high dose versus moderate dose ICS. Three of these studies used the step down approach and measured outcomes prior to stepping down ICS (Campbell 1998; Chanez 2001; Visser 2001). High vs low dose ICS were compared in nine studies, two of these using the step down approach and measuring outcomes prior to step down (Pirozynski 1996; van der Molen 1998). Moderate vs low dose ICS were compared in 11 trials. This included one trial that included two low dose ICS arms (Sheffer 1996). Four studies included three arms comparing high, moderate and low dose ICS (Wasserman 1996; Miyamoto 2000;Pedersen 1995; Szefler 2002). One study compared low, medium and high dose ICS in increasing doses (step up) (Szefler 2002). The comparison of two low ICS doses was made in two studies (Hampel 2000; Sheffer 1996).
Full details of the dose comparisons are described in Table 1.
1. Primary Comparisons.
| Study | Age | Drug | StepDown vs Constant | High vs Mod | High vs Low | Mod vs Low |
| Bisgaard 1993 | Infants | BUD | 800mcg vs 200mcg | |||
| Bisgaard 1999 | Infants | FP | 200mcg vs 100 mcg | |||
| Campbell 1998 | Adults | BUD | 800‐400mcg vs 400mcg | 800mcg vs 400mcg | ||
| Chanez 2001 | Adults | BUD | 1600‐200mcg vs 400mcg | 1600mcg vs 400mcg | ||
| Gershman 2000 | Adults | FP | 1000mcg vs 100mcg | |||
| Hampel 2000 | Adults | BDP | ||||
| Hofstra 2000 | Children | FP | 500mcg vs 200mcg | |||
| Jatakanon 1999 | Adults | BUD | 400mcg vs 100mcg | |||
| Jonasson 1998 | Children | BUD | 200mcg vs 100mcg | |||
| Lorentzson 1990 | Adults | BUD | 400mcg vs 200mcg | |||
| Majima 1993 | Adults | BDP | 800mcg vs 400mcg | |||
| Miyamoto 2000 | Adults | BUD | 800mcg vs 400mcg | 800mcg vs 200mcg | 400mcg vs 200mcg | |
| Nayak 2002 | Children | BDP | 160mcg vs 80mcg | |||
| Noonan 1998 | Adults | FP | 200mcg vs 100mcg | |||
| O'Byrne 1996 | Adults | BUD | 800mcg vs 400mcg | |||
| Pedersen 1995 | Children | BUD | 400mcg vs 200mcg | 400mcg vs 100mcg | 200mcg vs 100mcg | |
| Pedersen 1996 | Adults | BUD | 1600mcg vs 400mcg | |||
| Pirozynski 1996 | Adults | BUD | 800‐200mcg vs 200mcg | 800mcg vs 200mcg | ||
| Sheffer 1996 | Adults | FP | 200mcg vs 50mcg | |||
| Szefler 2002 | Adults | BDP | 1344mcg vs 672mcg | 1344mcg vs 168mcg | 672mcg vs 168mcg | |
| Szefler 2002 | Adults | FP | 704mcg vs 352mcg | 704mcg vs 88mcg | 352 vs 88mcg | |
| Tukiainen 2000 | Adults | BUD | 800mcg vs 200mcg | |||
| van der Molen 1998 | Adults | BUD | 800‐200mcg vs 200mcg | 800mcg vs 200mcg | ||
| Visser 2001 | Children | FP | 1000‐100mcg vs 200mcg | 1000mcg vs 200mcg | ||
| Volovitz 1998 | Infants | BUD | 2‐0.5mg vs 0.5mg | |||
| Wasserman 1996 | Adults | FP | 500mcg vs 200mcg | 500mcg vs 100mcg | 200mcg vs 100mcg | |
| Wennergren 1996 | Infants | BUD | 2mg‐0.5mg vs 0.5mg | |||
DURATION
Treatment periods ranged from a minimum of four weeks through to 24 months. The 24 month trial (Jonasson 1998) had an initial treatment period of 12 weeks with some participants continuing for 24 months. Outcome data was collected at 12 weeks. The majority of studies (n = 18) were conducted over a four to 12 week period. Another two studies had a duration of 16 weeks (Chanez 2001; O'Byrne 1996), one study of 18 weeks ( Wennergren 1996) and three studies of 24 weeks (Campbell 1998; Hofstra 2000; Szefler 2002). Two studies were conducted over nine and 12 months (Pedersen 1996; Visser 2001).
DEVICES
A dry powder inhaler (DPI) was used to deliver ICS in nine studies. Twelve studies used a metered dose inhaler (pMDI) with six of these also using a spacer device. A nebuliser was used in two of the infant studies(Volovitz 1998; Wennergren 1996). Three studies did not state the method of ICS delivery.
ASTHMA SEVERITY
Asthma severity was determined from the study reports using percent predicted FEV1 stated as an inclusion criteria or the mean percent predicted FEV1 of participating subjects. Asthma severity was defined according to Australian Asthma Management Guidelines (NAC 2002) as mild: FEV1 > 80% predicted, moderate: FEV1 60‐80% predicted and severe: FEV1 <60% predicted. The majority of studies included participants with mild to moderate asthma (n = 8). Mild asthma was represented in four studies, moderate asthma in four studies and four studies included participants with moderate to severe asthma (Bisgaard 1999; Nayak 2002; Sheffer 1996; Wasserman 1996). In six studies the level of asthma severity was unable to be determined. In three studies this was due to reporting mean FEV1 instead of percent predicted, one study reported entry criteria of symptoms present a minimum of six days per month (Wennergren 1996), Bisgaard 1993 included participants with recurrent wheeze, and one study's entry criteria were wheezing more than 40% of the days in the past three months (Volovitz 1998). In one study (Miyamoto 2000) 1% of participants had severe asthma.
Risk of bias in included studies
Methodological quality was good for the majority of studies. All the included trials were randomised controlled trials with 22 of them double blinded. Two trials did not specify the type of blinding (Majima 1993; Hampel 2000), one trial was 'open label' (Szefler 2002) and one single blind (Bisgaard 1993). Using the Jadad system, 81% of the studies scored three or higher, with four trials scoring two and one trial with a score of one (Majima 1993). It is possible that this trial was of higher quality but due to the limitations of translation this could not be determined. Quality assessed by the Cochrane method was unclear for 15 studies with 11 trials adequately reporting the method of allocation concealment.
Effects of interventions
STEP DOWN VERSUS CONSTANT ICS DOSE
In seven studies initial high dose ICS that was subsequently stepped down was compared to a constant ICS dose. 1396 participants were randomised and in six studies 844 participants completed the study. One study (Campbell 1998) reported a withdrawal rate of 33% due to adverse events, worsening asthma and non‐compliance (55% of withdrawals). The total duration of treatment varied from 10 weeks (Volovitz 1998) to 12 months (Visser 2001). The duration of high dose ICS prior to step down was one week (Volovitz 1998), three weeks (Wennergren 1996), four weeks (Chanez 2001; van der Molen 1998; Pirozynski 1996), six weeks (Campbell 1998) and two months (Visser 2001). In two studies, ICS dose was only reduced if participants fitted predefined criteria for dose reduction (Wennergren 1996) or their asthma was controlled (Chanez 2001). Two studies, at step down, reduced the dose from b.i.d to a once daily dose (van der Molen 1998; Campbell 1998). The step down ICS dose was reduced to a lower dose than the constant ICS dose in two studies (Chanez 2001; Visser 2001) and reduced to the same dose as the constant dose comparison in the remaining five studies. Four studies were conducted in adults, one in children and two in infants. Where appropriate, subgroup analyses were conducted for age, ICS type, device (if stated) and duration. These seven studies reported the following results:
Lung Function
FEV1, measured as percent predicted, was reported in four studies(Chanez 2001; Pirozynski 1996; Visser 2001; van der Molen 1998), however there was insufficient data provided to perform a meta analysis. All four studies reported no significant difference between the treatment groups.
Peak Flow (PEF)
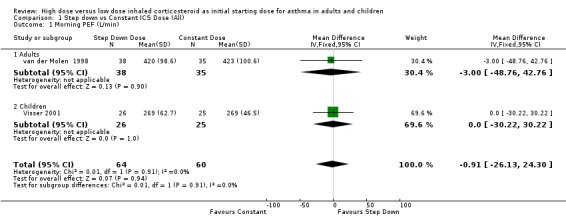
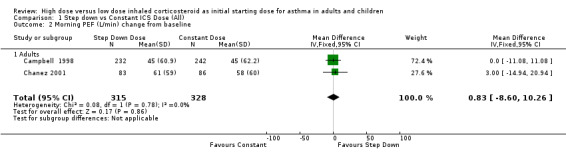
Five studies reported morning PEF, three as L/min (Pirozynski 1996; van der Molen 1998; Visser 2001) and two as change from baseline (Campbell 1998; Chanez 2001). There was no significant difference in morning PEF for step down ICS dose compared to a constant ICS dose. This non‐significance extended to both adults, children and ICS type separately for morning PEF L/min. There was no heterogeneity present. All studies included in these meta analyses were conducted for > 12 weeks and used either DPI or did not state the method of delivery (Visser 2001). Morning PEF L/min (adults and children): WMD ‐0.91 (95%CI ‐26.13 to 24.30) Morning PEF L/min change from baseline (adults): WMD 0.83 (95%CI ‐8.6 to 10.26) No significant difference between treatment groups for evening PEF L/min was reported in one study (Visser 2001).
Symptoms
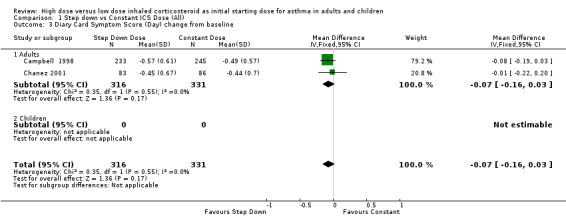
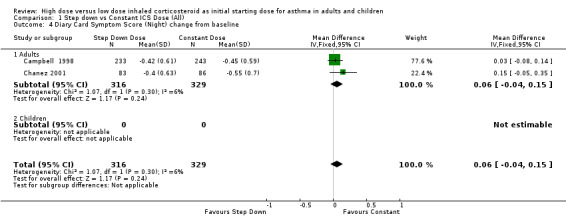
All seven studies reported symptoms. Overall, there was no significant difference between the two treatment approaches. Symptoms improved for both treatment groups with no significant difference between the groups in three studies (Pirozynski 1996; Visser 2001; Wennergren 1996). Symptoms were measured as a score in the remaining four studies. All four studies used a score of 0‐3 where no symptoms equalled a score of zero and severe symptoms a score of three. Volovitz 1998 reported a reduction in symptoms for both treatment groups that was greater for the step down group particularly for wheeze. Day and night symptom scores were reported in three studies, one as a mean score (van der Molen 1998) and two as diary card symptom score change from baseline (Campbell 1998; Chanez 2001). No significant difference between treatment groups in day and night symptoms was reported and no significant heterogeneity present with the pooled results. Both studies included in these meta analyses were conducted in adults using BUD via a DPI for > 12 weeks. Diary card symptom score (day) change from baseline (adults): WMD ‐0.07 (95% CI ‐0.16 to 0.03) Diary card symptom score (night) change from baseline (adults): WMD 0.06 (95% CI ‐0.04 to 0.15)
Rescue medications
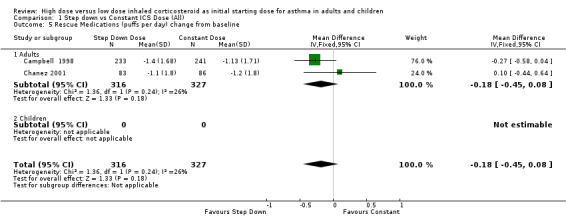
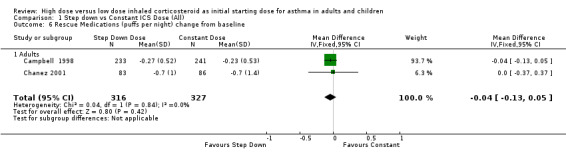
Rescue medication use was measured in five trials. Three trials reported a reduction in beta agonist use for both treatment groups (van der Molen 1998; Wennergren 1996; Pirozynski 1996). Day and night beta agonist use was reported separately in two trials, both in adults using BUD via a DPI for > 12 weeks, as the change from baseline in mean puffs per day or night (Campbell 1998; Chanez 2001). Meta analyses were able to be performed for these two outcomes. There was no significant difference for the change in nighttime or daytime beta agonist use. There was no heterogeneity present but some inconsistency was present (I2 = 26%) between studies for daytime beta agonist use. Rescue medications (day) change from baseline (adults): WMD ‐0.18 (95% CI ‐0.45 to 0.08) Rescue medications (night) change from baseline (adults): WMD ‐0.04 (95% CI ‐0.13 to 0.05)
Adverse events
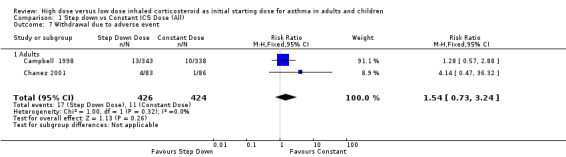
Adverse events were recorded as the number of participants who withdrew due to an adverse event in two studies in adults (Campbell 1998; Chanez 2001). When these results were pooled there was no significant difference in withdrawals due to adverse events for constant ICS compared to step down ICS. There was no heterogeneity present. Both studies included in the meta analysis were conducted in adults using BUD via a DPI for > 12 weeks. Withdrawal due to adverse event (adults): RR 1.54 (95% CI 0.73 to 3.24)
Asthma control
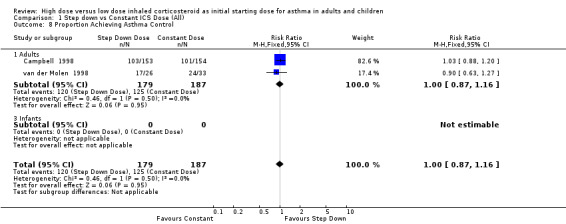
There was no reported difference in time taken to achieve control of asthma for step down or constant dose ICS in infants (Wennergren 1996) or adults (van der Molen 1998). Two studies in adults using BUD via a DPI for > 12 weeks, reported the number of participants achieving asthma control (van der Molen 1998; Campbell 1998). There was no significant difference and no heterogeneity present for these pooled results. Asthma control (adults): RR 1.0 (95% CI 0.87 to 1.16)
Airway hyperresponsiveness
In a study in children Visser 2001 reported an improvement in airway hyperresponsiveness for both stepdown and constant ICS groups. This was higher for the step down ICS at four months but there was no significant difference between the dose groups in airway hyperresponsiveness at completion of the study after 12 months.
HIGH VERSUS MODERATE ICS DOSE
High dose ICS were compared to moderate dose ICS in 11 studies. 1749 participants were randomised with 10 studies reporting 1294 participants completing the studies. Three studies that were included in the step down comparison have also been included in this comparison using outcome data collected prior to stepping down the high dose ICS at four, six and eight weeks respectively (Chanez 2001; Campbell 1998; Visser 2001). The remaining eight studies were between six weeks and nine months duration.
Three studies had three dosage arms of high, moderate and low dose ICS (Miyamoto 2000; Wasserman 1996; Pedersen 1995). The high and moderate ICS dose comparisons were used in this analysis. A parallel design was used in nine studies, crossover design of three four week treatment periods in one study (Pedersen 1995) and a step up design of two ICS in another (Szefler 2002). Subgroup analysis was conducted for age, ICS type, device(if stated) and duration of study.
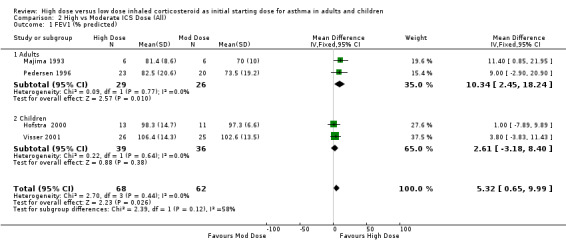
Lung Function FEV1 was reported in eight studies as litres, percent predicted and litres change from baseline. No significant difference between high and moderate dose ICS was reported in four studies (Szefler 2002; Wasserman 1996; Miyamoto 2000; Chanez 2001). Four studies reporting percent predicted FEV1 provided sufficient data to be pooled using a WMD. There was a significant improvement in FEV1 in favour of the high dose group with no statistical heterogeneity. FEV1 % predicted (All): WMD 5.32 (95% CI 0.65 to 9.99) When analysed by age this remained significant for adults but there was no significant difference between high and moderate dose ICS for children. The pooled sample sizes were small: adults = 55, children = 75. FEV1 % predicted (adults): WMD 10.34 (95% CI 2.45 to 18.24) FEV1 % predicted (children): WMD 2.61 (95% CI ‐3.18 to 8.4)
Morning Peak Flow
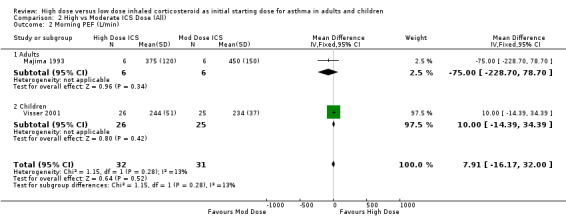
Four studies reported morning PEF as L/min.There was no significant difference between treatment groups in morning PEF reported in one study (Hofstra 2000) and a significant improvement for all treatment groups with difference between treatment groups not stated in an other study (Pedersen 1995). No significant difference was found when the results of two studies, both using a DPI and < 12 weeks duration were pooled. This was consistent for age and ICS type. There was no heterogeneity. Morning PEF (All): WMD 7.91 (95% CI ‐16.17 to 32.0)
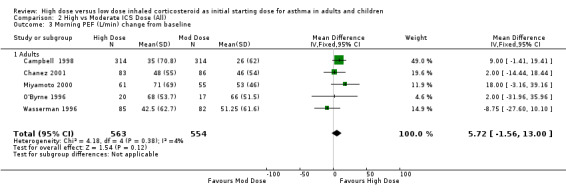
Morning PEF was reported as the change in L/min from baseline in five studies conducted in adults. When these studies were pooled there was no significant difference between the treatment groups in favour of high dose ICS. No heterogeneity was present. These results remained non significant when analysed by device and duration of the study. Morning PEF L/min change from baseline (All): WMD 5.72 (95% CI ‐1.56 to 13.0)
Evening Peak Flow
Four studies reported evening PEF as L/min (n=2) and L/min change from baseline (n=2). No significant difference between treatment groups was reported in all four studies. A significant test for trend suggesting a dose response relationship was reported in one study (Miyamoto 2000).
Symptoms
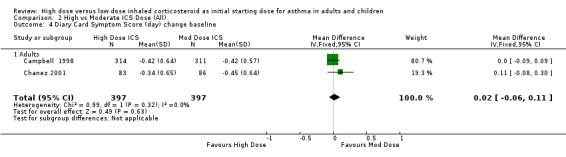
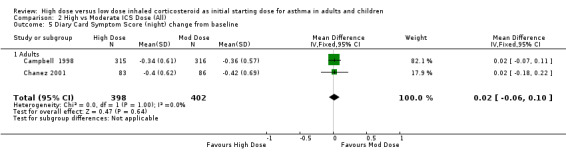
Symptoms were reported in seven studies as both day and night symptoms, proportion of participants reporting symptoms, symptom score and symptom score change from baseline. Miyamoto 2000 reported significant dose‐response relationships for symptom scores. Symptoms reduced for both treatment groups in four studies with no significant difference between treatment groups in two studies (Wasserman 1996; O'Byrne 1996) and significance not stated in two studies (Szefler 2002;Pedersen 1995). There was no significant difference in two studies, conducted in adults using BUD via a DPI for 12 weeks or less, when the results were pooled. Both studies used the same 0‐3 scale. No heterogeneity was present. Symptom score (day ) change from baseline: WMD 0.02 (95% CI ‐0.06 to 0.11) Symptom score (night) change from baseline: WMD 0.02 (95% CI ‐0.06 to 0.10)
Rescue medications
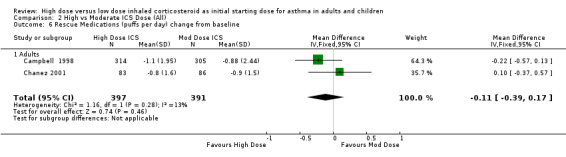
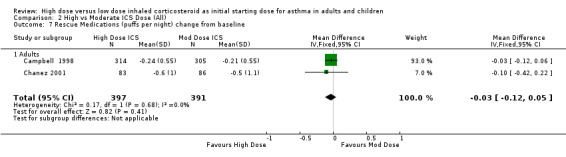
Five studies reported the use of rescue medication. Two studies, both in adults using BUD for 12 weeks or less, reported beta agonist use as the change in puffs per day or night from baseline (Campbell 1998;Chanez 2001). The pooled results showed no significant difference between treatment groups for day or night rescue medication use and no heterogeneity. Rescue medications (day) change from baseline: WMD ‐0.11(95% CI ‐0.39 to 0.17) Rescue medications (night) change from baseline: WMD ‐0.03(95% CI ‐0.12 to 0.05)
The remaining three studies reported reductions in rescue medication use for both treatment groups with no significant difference between groups in one study (Pedersen 1996) and significance not stated in two studies (Szefler 2002; Wasserman 1996).
Airway hyperresponsiveness
Airway hyperresponsiveness was measured in five studies either as PC20 or after exercise challenge. There was insufficient data provided to perform a meta analysis. In the three studies conducted in children, (Pedersen 1995) reported a significant difference between high and moderate dose BUD in fall in FEV1% after exercise in favour of the high dose group (p<0.0001). Visser 2001 reported an improvement in AHR for both FP treatment groups that was significantly higher for the high dose group at four months. In a study of FP conducted in children by Hofstra 2000 PD20 was higher for the high dose group at 24 weeks but not significant and AHR (exercise) was significantly reduced for both FP groups.
In the remaining two studies conducted in adults using FP and BDP ( Szefler 2002) and BUD ( Pedersen 1996), no significant differences between high and moderate dose ICS for AHR were reported at completion of the study. However in the study using BUD, although non significant, the improvement was greater in the high dose group.
Adverse Events
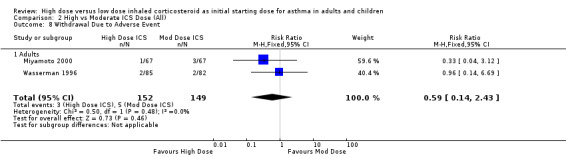
Withdrawal Due to Adverse Event Two studies ( Wasserman 1996; Miyamoto 2000) conducted in adults reported the number of participants who withdrew due to an adverse event. There was no significant difference between high or moderate BUD or FP and no heterogeneity. Both studies were less than 12 weeks duration and used a DPI for delivery of ICS. Withdrawal due to adverse event (adults): RR 0.59 (95%CI 0.14 to 2.43)
Oral Candidiasis
In the two studies reporting oral candidiasis Hofstra 2000 reported no cases in either treatment group and there was no significant difference in oral candidiasis between high and moderate FP in the study conducted by Wasserman 1996.
Inflammatory markers
Blood Eosinophils
Blood eosinophils decreased for both high and moderate dose BUD in one study ( Pedersen 1996). These decreases were more rapid in the high dose group than the moderate dose group despite there being no significant difference between treatment groups at the end of the study.
Serum ECP
Two studies reported the results of high and moderate ICS dose on serum ECP. No significant changes were reported for either FP group in a study conducted in children ( Hofstra 2000). ECP reduced after 9 months on low dose BUD and after 1 month on high dose BUD (Pedersen 1996).
HIGH VERSUS LOW ICS DOSE
In nine studies high dose ICS was compared to low dose ICS. Three of these studies included a third arm of moderate dose ICS which was analysed in the previous comparison (Miyamoto 2000; Wasserman 1996; Pedersen 1995). One study using the step down approach reported outcomes prior to step down (van der Molen 1998). 1136 participants were randomised with eight studies reporting 730 completing. Parallel design was used in six studies in adults, two studies in infants and children used a crossover design (Bisgaard 1993; Pedersen 1995), and one adult study stepped up medication every six weeks (Szefler 2002). Duration ranged from four to 24 weeks. Results were analysed by age, ICS type, device and duration.
Lung Function
Six studies reported FEV1 as percent predicted FEV1, litres or change from baseline. Two studies in adults of 12 weeks or less duration, reporting FEV1 as percent predicted and in litres were pooled in a meta analysis. There were no significant differences in FEV1 between high dose ICS and low dose ICS and no statistical heterogeneity was present. This was consistent when analysed by ICS type, device and duration of study. FEV1 (All):SMD ‐0.20 (95% CI ‐0.55 to 0.16)
No significant differences between treatment groups were reported in the remaining four studies for FEV1 (van der Molen 1998; Szefler 2002), or FEV1 change from baseline (Miyamoto 2000; Wasserman 1996).
Morning Peak Flow
Morning peak flow was measured in five studies as L/min. Morning PEF was not significantly different between high and low ICS dose (Tukiainen 2000) and improvements for both treatment groups were reported in another study (Pirozynski 1996). When the results of two studies conducted in adults, and less than 12 weeks duration, were pooled in a meta analysis there was no significant difference in morning PEF. This was consistent for both ICS type and device with no statistical heterogeneity. Morning PEF (All): WMD ‐18.05 (95% CI ‐56.04 to 19.95)
Two studies reported morning PEF L/min as the change from baseline. Both studies were conducted in adults using BUD via a DPI and for less than 12 weeks. When these results were pooled there was no significant improvement in morning PEF in favour of high dose BUD. Statistical heterogeneity was not present, however there was moderate inconsistency between the studies. Morning PEF change from baseline (adults): WMD 12.0 (95% CI ‐1.50 to 25.51). Chi2 =2.06, p=0.15, I2 = 51.6%.
Evening Peak Flow
Evening PEF was reported in two studies as L/min (Pedersen 1995; Tukiainen 2000) and change from baseline (Miyamoto 2000; Wasserman 1996). No significant difference between treatment groups was reported by all four studies. Miyamoto 2000 reported a significant test for trend suggesting a dose response relationship.
Symptoms
Seven studies reported symptoms both as day and night symptoms and symptom score . Symptoms reduced for both treatment groups with no significant difference between groups in five studies (Bisgaard 1993; Gershman 2000; Tukiainen 2000; van der Molen 1998; Wasserman 1996) and reduced in two studies with significance not stated (Szefler 2002; Pedersen 1995).
Rescue medications
The use of rescue medications was reported in five studies. There was no significant difference in beta agonist use (puffs/day) in adults and no heterogeneity present when the results of two studies of 12 weeks or less and reporting rescue medication use were pooled. Significant heterogeneity ws not present but there was moderate inconsistency between the two studies. Rescue Medications: WMD ‐0.04 (95%CI ‐0.65 to 0.56). Chi2 =2.36, p=0.12, I2 =57.6%. When analysed by ICS type there was a non significant trend for less rescue medication use in the high dose treatment group for the study conducted using FP via a pMDI and no significant difference between BUD treatment groups.
The three remaining studies (Bisgaard 1993; Szefler 2002; Wasserman 1996) reported reductions in rescue medication use with both treatments, and significance not stated.
Airway hyperresponsiveness
Four studies measured airway hyperresponsiveness either as PC20 or after exercise challenge. There was insufficient data provided to perform a meta analysis. In the study conducted in children, Pedersen 1995 reported a significant difference between high and low dose BUD in fall in FEV1% after exercise in favour of the high dose group (p<0.001).
In the remaining three studies conducted in adults the study using FP (Gershman 2000) reported no significant differences between high and low dose ICS for AHR. Szefler 2002, using both BDP and FP reported a maximum change in PC20 at low dose FP and at moderate dose BDP with significance between high and low dose ICS not stated. In a study of BUD Tukiainen 2000 reported, although non significant, an improvement that was greater in the high dose group.
Adverse events
Withdrawal Due to Adverse Event
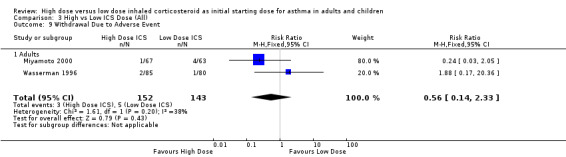
Two studies (Miyamoto 2000; Wasserman 1996) conducted in adults reported the number of participants who withdrew due to an adverse event. There was no significant difference between high or low BUD or FP. Significant heterogeneity was not present but there was low to moderate inconsistency between the studies. Both studies were less than 12 weeks duration and used a DPI for delivery of ICS. Withdrawal due to adverse event (adults): RR 0.56 (95%CI 0.14 to 2.33).Chi2 =1.61, p=0.20, I2 =38.0%.
Oral Candidiasis
There was no significant difference in oral candidiasis between high and low dose FP in one study (Wasserman 1996).
Inflammatory markers
Blood Eosinophils
Tukiainen 2000 reported a significantly greater decrease in blood eosinophils for the high dose BUD group compared to the low dose group.
Serum ECP
Two studies reported the results of high and moderate ICS dose on serum ECP. Serum ECP decreased significantly for the high dose BUD group with no change for the low dose group in one study (Tukiainen 2000). Gershman 2000 reported significant decreases for both FP groups but no significant differences between groups.
Sputum Eosinophils
Gershman 2000 reported a significant reduction in sputum eosinophils after 14 days treatment for both high and low FP groups.
MODERATE VERSUS LOW ICS DOSE
1971 participants were randomised in 11 studies to moderate or low dose ICS with 1743 completing the studies. Seven studies were conducted in adults, three in children and one in infants. The duration of treatment was from four to 24 weeks. Four studies included in the high vs moderate ICS dose comparison that had a third arm of low dose ICS were able to be analysed in this comparison (Miyamoto 2000; Pedersen 1995; Wasserman 1996; Szefler 2002). One study was a crossover design (Pedersen 1995) and one study of two treatments stepped up therapy every six weeks (Szefler 2002). Results were analysed by age, ICS type, device and duration.
Lung Function
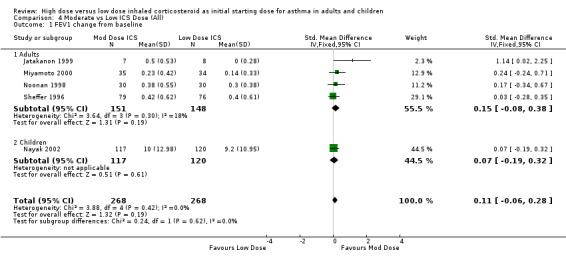
FEV1 was reported as the change from baseline in litres or percent predicted in six studies. There was no significant difference in moderate or low dose ICS for FEV1 change from baseline when analysed by age, ICS type, device or duration in the five studies that were pooled for this analysis. There was no heterogeneity. FEV1 change from baseline (adults & children): SMD 0.11 (‐0.06 to 0.28)
One study (Szefler 2002) reported FEV1 (L) reaching a maximum at moderate dose BDP and at low dose FP.
Morning PEF
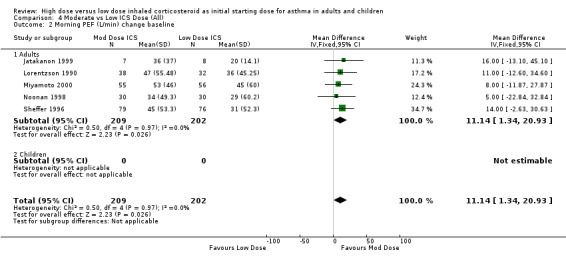
Three studies in children reported PEF L/min. Two of these studies (Nayak 2002; Pedersen 1995) did not state the difference between the treatment groups but compared them to a placebo arm. Jonasson 1998 reported no significant difference between the treatment groups for PEF. There was a significant improvement in PEF L/min change from baseline in favour of a moderate ICS dose for the five studies in adults, all less than 12 weeks duration, that were pooled for this analysis. No heterogeneity was present. Morning PEF L/min change from baseline (adults): WMD 11.14 (95% CI 1.34 to 20.93) When examined by individual ICS and device the improvement did not show any significant difference between FP and BUD. Morning PEF L/min change from baseline (BUD): WMD 10.69 (95% CI ‐2.78 to 24.16) Morning PEF L/min change from baseline (FP): WMD 11.63 (95% CI ‐2.64 to 25.91)
Evening PEF
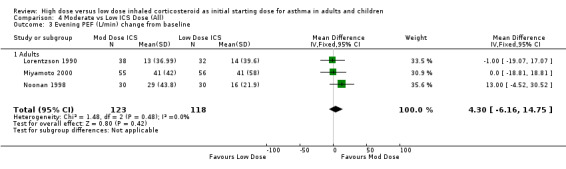
There was no significant difference in evening PEF L/min change from baseline between moderate and low dose ICS as reported in three adult studies of less than 12 weeks duration. This was consistent for device with no heterogeneity. Evening PEF L/min change from baseline (adults): WMD 4.30 (95%CI ‐6.16 to 14.75) When analysed by ICS type there was a non significant trend for improvement in evening PEF for FP but not BUD. Evening PEF L/min change from baseline (BUD): WMD ‐0.52 (95%CI ‐13.55 to 12.51) Evening PEF L/min change from baseline (FP): WMD 13.0 (95%CI ‐4.52 to 30.52)
PEF Diurnal Variation
PEF diurnal variation was measured in two studies in adults with BUD using a DPI and less than 12 weeks duration as L/min (Miyamoto 2000) and percent (Jatakanon 1999) change from baseline. The improvement in favour of moderate dose ICS was not significant and showed no heterogeneity. PEF diurnal variation change from baseline (adults): SMD ‐0.28 (95%CI ‐0.63 to 0.07)
Symptoms
Symptoms were reported in nine studies. Miyamoto 2000 reported significant dose‐response relationships for symptom scores. No significant differences were reported for moderate compared to low dose ICS for symptom scores (Noonan 1998), diary card symptoms (Bisgaard 1999), mean symptom free days (Miyamoto 2000) and symptom score change from baseline (Lorentzson 1990). Symptoms were reduced for all treatment groups in two studies (Jonasson 1998; Pedersen 1995). Two studies in adults using the same 0‐3 scale reported symptom score change from baseline(Noonan 1998; Jatakanon 1999). There was no significant difference in reduction in symptom score when these results were pooled, however significant heterogeneity and high inconsistency were present. Symptom Score change from baseline (adults):WMD ‐0.04 (95%CI ‐0.21 to 0.13). When examined by ICS type there was a significant improvement in symptom score from baseline for moderate dose BUD using a DPI (Jatakanon 1999) and no significant difference between moderate and low dose FP using a pMDI (Noonan 1998). Both studies were of less than 12 weeks duration.
There was a significant reduction of symptoms (night waking) from baseline in favour of moderate dose ICS when the results of three studies, two using the same 0 to 3 scaled symptom score and one study reporting as mean number of awakenings were pooled. There was no heterogeneity. All three studies were conducted in adults using a pMDI and less than 12 weeks duration. Symptom score (night waking): SMD ‐0.29 (95%CI ‐0.53 to ‐0.06).
Rescue Medication
Rescue medications were measured as the change in number of puffs from baseline in five studies. Lorentzson 1990 reported no significant difference between moderate and low dose BUD for reduction in day or night beta agonist use compared to baseline. When the results of the remaining four studies were pooled the reduction in beta agonist use for moderate dose ICS was not significant. This was consistent for age, ICS type and device with no heterogeneity. The studies were all less than 12 weeks in duration. Rescue Medications change from baseline (adults & children): WMD ‐0.35 (95% CI ‐0.98 to 0.28)
Airway hyperresponsiveness
There were no significant differences between moderate and low dose ICS for PD20 in three studies (Jatakanon 1999; Jonasson 1998; Noonan 1998). However Jatakanon 1999 reported a trend in favour of increasing BUD doses and Noonan 1998 reported this to be non significantly greater for the moderate dose group. Pedersen 1995 reported significant differences in favour of the moderate dose BUD group for AHR post exercise challenge.
Adverse events
There were no significant differences and no heterogeneity between moderate and low dose ICS for the seven studies that reported this outcome either as withdrawal due to adverse event or as adverse events. This was consistent for age, ICS type, device and duration of study. Withdrawal due to adverse event (all): RR 1.0 (95% CI 0.41 to 2.46) Adverse events (all): RR 1.04 (95% CI 0.93 to 1.17)
Oral Candidiasis
Two studies using FP reported the incidence of oral candidiasis, one in adults(Sheffer 1996) and one in infants (Bisgaard 1999). There was no significant difference between moderate and low dose ICS when these results were pooled. Statistical heterogeneity was present and there was high inconsistency between the two studies. Oral candidiasis (adults & infants): RR 0.90 (95% CI 0.34 to 2.39), Chi2 =3.91, p=0.05, I2 =74.4%. This was consistent for both device and duration of the study. When analysed by age, there were non significantly fewer reporting of oral candidiasis in the low dose ICS group for adults and the moderate dose ICS group for infants.
Exacerbations
Exacerbations were reported in two studies with no significant difference between moderate and low dose ICS for children or infants. This was consistent for ICS type, device and duration of study. No statistical heterogeneity was present. Exacerbations (children & infants): RR 0.82 (95% CI 0.47 to 1.43)
Plasma Cortisol
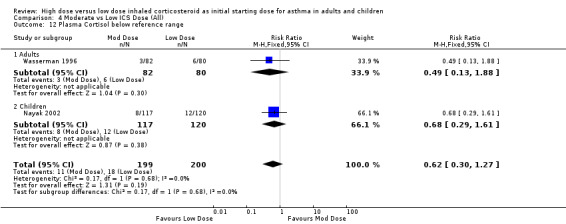
A non significant proportion of participants randomised to low dose ICS were reported to have a plasma cortisol level below the reference range in two studies conducted in adults and children. No heterogeneity was present and no significant difference in subgroup analysis. Plasma cortisol below reference range (adults & children): RR 0.62 (95% CI 0.30 to 1.27)
Other Outcomes
No significant differences between treatment groups were reported for serum eosinophils or ECP in one study in children (Jonasson 1998). In a study in adults Jatakanon 1999 reported significant reductions in sputum eosinophils and exhaled nitrous oxide with no significant difference between the treatment groups.
FOLD DIFFERENCE IN ICS DOSE
ICS dose can also be examined as a fold increase over the comparator. We therefore included an analysis examining fold difference in ICS dose. All included studies were analysed according to the fold difference in ICS dose compared to the ICS dose used in the control group. Studies were classified using either a two fold ICS dose increase (n=9), a four fold increase or greater (n=11) or both two and four fold ICS dose increases, when compared to the ICS dose used by the control group (n=5). One study compared a three fold increase in ICS (Hofstra 2000). For the studies using the step down approach the outcomes reported prior to step down were used. The following are the results of the pooled analyses.
Lung Function
Five studies measuring FEV1 as L/min or percent predicted were pooled using the SMD. There was no significant difference in FEV1 between the base ICS dose and a four fold increased ICS dose in four studies. No heterogeneity was present and there was low inconsistency between the studies. FEV1 (>= 4 Fold): SMD 0.04 (95% CI ‐0.23 to 0.31).
FEV1 reported as the change from baseline was not significantly greater for both a four fold increase in ICS (n=3) and a two fold increase in ICS dose (n=5). No heterogeneity was present, however there was moderate inconsistency between the four fold studies. FEV1 change from baseline (>= 4 Fold): SMD 0.18 (95% CI ‐0.08 to 0.43). Chi2 = 4.08, p = 0.13, I2 = 51.0%. FEV1 change from baseline (2 Fold): SMD 0.14 (95% CI ‐0.01 to 0.29).
Morning Peak Flow
Four studies reported morning PEF as L/min or L/second. There was no significant difference in morning PEF between a four fold increased ICS dose(n=3) or a two fold increased ICS dose (n=1) and the base dose. No heterogeneity was present. Morning PEF (>= 4 Fold): SMD ‐0.03 (95% CI ‐0.36 to 0.29) Morning PEF (2 Fold): SMD ‐0.51 (95% CI ‐1.67 to 0.65). There was a significant improvement in the change in morning PEF from baseline favouring both the four fold (n=5) and two fold (n=8) increased dose. There was no heterogeneity. Morning PEF change from baseline (>= 4 Fold): WMD 10.21(95% CI 1.75 to 18.67) Morning PEF change from baseline (2 Fold): WMD 6.8 (95% CI 0.75 to 12.84).
Evening Peak Flow
Evening PEF was reported as the change from baseline. In the one study that compared a four fold increase in ICS dose there was a non significant improvement in favour of the four fold dose. There was no significant difference in improvement in evening PEF from baseline between the two fold increased dose and the base dose when the results of four studies were pooled. Heterogeneity was not present. Evening PEF change from baseline (>= 4 Fold): SMD 15.0 (95% CI ‐6.39 to 36.39) Evening PEF change from baseline (2 Fold): SMD 2.37 (95% CI ‐5.78 to 10.53).
PEF Diurnal Variation
In the three studies measuring this outcome the improvement in PEF diurnal variation in favour of both a four fold and a two fold increase in ICS compared to the base dose was not significant. PEF Diurnal Variation (>= 4 Fold): SMD ‐0.32 (95% CI ‐0.66 to 0.03) PEF Diurnal Variation (2 Fold): SMD ‐0.21 (95% CI ‐0.59 to 0.16).
Symptoms
Symptoms were reported as the change in both day and night symptoms scores from baseline. There was no difference in improvement in daytime symptom score between a two fold increased ICS dose and the base ICS dose in the pooled result of two studies. In a single study comparing a four fold increased ICS dose there was a non significant improvement in favour of the base ICS dose. Symptom score (day ) change from baseline (>= 4 fold): WMD 0.11 (95%CI ‐0.08 to 0.30) Symptom score (day ) change from baseline ( 2 fold): WMD 0.0 (95%CI ‐0.09 to 0.09) There was an improvement in night time symptom score for all treatment groups with no significant difference between a two fold increase and a base dose (n=4) and a non significant difference in favour of a four fold increase (n=2), however heterogeneity was present in the four fold analysis. Symptom score (night) change from baseline (>= 4 fold): SMD ‐0.17 (95% ‐0.38 CI to 0.05), Chi2 =3.42, p=0.06, I2 =71% Symptom score (night) change from baseline (2 fold): SMD ‐0.02 (95% CI ‐0.15 to 0.11)
Rescue medications
Rescue medication use reported as the change from baseline of either the number of puffs or puffs per day, was no different for either a four fold ICS dose (n=2) or two fold ICS dose (n=4) compared to a base ICS dose and no heterogeneity. Rescue medications change from baseline (>= 4 fold): SMD ‐0.11 (95%CI ‐0.42 to 0.19) Rescue medications change from baseline (2 fold): SMD ‐0.01 (95%CI ‐0.16 to 0.15) This lack of a difference was also evident when rescue medications were measured as day and night use change from baseline. Rescue medications (day) change from baseline (>= 4 fold): WMD ‐0.10 (95%CI ‐0.37 to 0.57) Rescue medications (day) change from baseline (2 fold): WMD ‐0.21 (95%CI ‐0.55 to 0.14) Rescue medications (night)change from baseline (>= 4 fold): WMD ‐0.10(95%CI ‐0.42 to 0.22) Rescue medications (night) change from baseline (2 fold): WMD ‐0.03 (95%CI ‐0.11 to 0.06)
Adverse events
Withdrawal Due to Adverse Event
There was no significant difference in the risk of withdrawing due to an adverse event between a four fold increased ICS (n=2) or a two fold increased ICS (n=5) compared to base ICS dose and no statistical heterogeneity. However low to moderate inconsistency was present for the two four fold studies. Withdrawal due to adverse event (>= 4 fold): RR 0.56 (95% CI 0.14 to 2.33), Chi2 = 1.61, p = 0.2, I2 = 38.0%. Withdrawal due to adverse event (2 fold): RR 1.09 (95% CI 0.49 to 2.42)
Oral Candidiasis
There was no significant difference in incidences of oral candidiasis in the pooled results of four studies comparing a two fold ICS increase to a base dose and the two studies comparing a four fold increase in ICS dose and no heterogeneity. There was low inconsistency between the four fold studies and the two fold studies. Oral Candidiasis (>= 4 fold): RR 2.30 (95% CI 0.68 to 7.83), Chi2 = 1.46, p = 0.23, I2 = 31.4% Oral Candidiasis (2 fold): RR 0.66 (95% CI 0.27 to 1.64), Chi2 = 2.28, p = 0.32, I2 = 12.1%
Plasma Cortisol
A non significant greater proportion of participants were reported to have a plasma cortisol level below the reference range in the base dose groups for both the four fold and the two fold comparisons. Plasma Cortisol below ref range (>=4 fold): RR 0.63 (95%CI 0.18 to 2.14) Plasma Cortisol below ref range (2 fold): RR 0.62 (95%CI 0.30 to 1.27)
Discussion
This systematic review evaluated the results of 26 RCTs that compared initial ICS doses in asthma. We evaluated the effects of starting ICS at a high dose, plus or minus a subsequent step down, to commencing with a moderate or low dose ICS. In light of differing guideline recommendations, and the frequent prescription of high dose ICS with subsequent significant side effects, it is important to establish the optimal starting dose for ICS in asthma.
The overall results of the studies suggest that there is no significant difference between initial ICS doses for most asthma outcomes. It has previously been established that low to moderate ICS doses are highly effective for asthma (Adams 2002; Powell 2003). We have extended these observations to examine the efficacy of differing ICS doses as initial therapy for asthma. In most of the studies included in this review efficacy was shown in both treatment arms for the majority of outcomes. There was no clear benefit for starting at a high ICS dose. There was no heterogeneity amongst the studies and the confidence intervals were relatively tight. For the key outcome of PEF, the criteria for equivalence was met for each dose comparison.This did not differ when analysed by drug type, delivery device or duration of the study. There may be a benefit for airway hyperresponsiveness, and this outcome is discussed separately below.
The practice of starting with high dose ICS to gain control of asthma and then stepping down to a moderate or low maintenance dose is recommended in some asthma management guidelines (NAC 2002). When we reviewed the seven studies that compared this practice to a constant moderate or low ICS dose we found no significant benefit on lung function, symptoms, rescue medications or asthma control. There were some non significant effects in favour of a step down dose for both rescue medication use and symptoms during the day but this was not maintained at night. These results suggest that constant ICS doses have similar clinical efficacy to the more complex regimen of high ICS doses followed by a step down.
In reviewing the 11 studies that compared a constant high dose ICS to a moderate dose ICS no significant differences were found between commencing with high or moderate dose ICS for most asthma outcomes. There was a statistically significant difference for adults favouring high dose ICS for FEV1. The pooled sample size for the FEV1 analysis was small (55 adults), and the effect was not seen in children. Furthermore, the effect was not consistently seen in other measures of lung function, or in other dose comparisons. This small benefit in lung function needs to be considered against the risks of increased side effects with the use of constant high dose ICS (Adams 2002; Powell 2003).
For the nine studies that compared a constant high dose ICS to a low dose ICS no significant differences were found in asthma outcomes. There was a clinically small and non significant improvement in change in morning PEF from baseline. Due to differences in reporting most outcomes only two studies were able to be included in the meta analyses. However all the studies reported improvements for both low and high dose ICS groups.
For moderate dose ICS there was a significant improvement in the change in morning PEF from baseline and nocturnal symptoms in comparison to low dose ICS. There were also non significant improvements in FEV1, PEF diurnal variation, and the reduction of rescue medication use from baseline suggesting a superior effect for moderate dose ICS.
Many of the subjects entering the studies were required to have symptomatic asthma with features of poor asthma control. Clinicians frequently consider dose adjustment by a two fold or four fold increase in dose under these circumstances. We therefore performed an analysis that compared the efficacy of a baseline dose to a two or four fold increased dose. When we reviewed all the included studies as to whether they compared a two or four fold increase in ICS we found no greater benefit for either. The change in morning PEF from baseline was the only outcome to show a significant improvement for either a two or four fold increased ICS dose compared to a moderate or low base dose. Any other improvements were non significant and, in the majority of outcomes, equivalent for a two or four fold increase in dose. For several outcomes these increases in dose were equivalent to the base dose. There was no significant difference for a four fold increase in ICS for FEV1, morning PEF or rescue medication use. This would suggest that there is no extra benefit gained by quadrupling the ICS dose. There was also no extra benefit for doubling ICS for evening PEF, symptom score (day or night), rescue medication use or withdrawal due to adverse events. Several studies reported a possible dose response relationship for airway hyperresponsiveness. In the one stepdown study that measured this outcome in children there was a significant improvement for the high dose group prior to stepping down ICS dose which was not maintained once the dose was stepped down. Another study, also in children, reported significant differences between high moderate and low dose ICS after exercise challenge. These differences were significant for the higher dose for all possible comparisons. Non significant improvements in AHR for high dose ICS were also reported when compared to moderate dose ICS in children (n=1), adults (n=1) and to low dose in adults in another study. no significant difference in AHR between treatment groups was reported in four studies, three comparing moderate to low dose ICS (one study reported a trend in favour of increasing doses) and one comparing high to low dose ICS. The study incorporating a step up design did not report the significance between treatment groups but stated a maximum improvement in AHR was achieved at low dose FP and moderate dose BDP.
International guidelines for initial ICS therapy vary in their recommendations. The Australian Asthma Management Guidelines (AAMP) (NAC 2002) recommend for adults with moderate persistent asthma commencing ICS at a dose of 800 mcg BDP equivalent per day (high dose) to gain rapid control of symptoms and then reducing this dose once controlled to the minimum required to maintain control. Whilst the Global Initiative for Asthma (GINA) (NIH 2002) recommend less than or equal to 500 mcg BDP equivalent (moderate dose) for mild persistent asthma and a wide range of initial ICS for moderate persistent asthma ranging from 200‐1000 mcg BDP equivalent (low to high dose). Similarly, they recommend stepping down ICS once asthma is controlled to 500 mcg BDP equivalent. The British Thoracic Society (BTS/SIGN 2003) guidelines recommend starting with an ICS dose of 400 mcg BDP equivalent per day (moderate dose) but also recommend a range according to severity of 200‐800 mcg/day, then titrating down to the lowest effective dose to maintain control. This review examined initial starting ICS doses. We did not examine back‐titration of maintenance ICS doses in controlled asthma. The results of this review support initiating asthma therapy with low to moderate doses of ICS at a constant dose.
Authors' conclusions
Implications for practice.
The results of this review support initiating ICS at a constant moderate dose.
Commencing inhaled corticosteroids at a constant moderate or low dose is equally efficacious to commencing at a high dose and then down titrating.
The small significant benefits of commencing with a high ICS dose are not of sufficient clinical benefit to warrant its use when compared to moderate or low dose ICS.
Initial moderate ICS dose appears to be more effective than initial low ICS dose.
High dose ICS may be more effective than moderate or low dose ICS for airway hyperresponsiveness.
Implications for research.
Further research could examine the effects of initial ICS dose on other asthma outcomes such as airway hyperresponsiveness, longitudinal decline in lung function, and asthma pathology.
What's new
| Date | Event | Description |
|---|---|---|
| 24 April 2019 | Amended | Seven studies added to Studies awaiting classification. |
History
Protocol first published: Issue 1, 2003 Review first published: Issue 1, 2004
| Date | Event | Description |
|---|---|---|
| 25 July 2008 | Amended | Converted to new review format. |
| 20 August 2003 | New citation required and conclusions have changed | Substantive amendment |
Acknowledgements
We would like to thank the Cochrane Airways Review Group for their assistance with this review with data base searches, obtaining studies, translations and statistical assistance: Toby Lasserson, Karen Blackhall, Jo Picot and Chris Cates.
We would also like to thank the following authors for providing information regarding their studies: Dr B Volovitz, Dr WB Hofstra, Dr MJ Visser and Dr O Selroos for Dr Miyamoto.
We would like to thank the Cooperative Research Centre for Asthma for financial support. We would like to thank Claire Allen for comments on the synopsis for this review.
Data and analyses
Comparison 1. Step down vs Constant ICS Dose (All).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Morning PEF (L/min) | 2 | 124 | Mean Difference (IV, Fixed, 95% CI) | ‐0.91 [‐26.13, 24.30] |
| 1.1 Adults | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | ‐3.0 [‐48.76, 42.76] |
| 1.2 Children | 1 | 51 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [‐30.22, 30.22] |
| 2 Morning PEF (L/min) change from baseline | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | 0.83 [‐8.60, 10.26] |
| 2.1 Adults | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | 0.83 [‐8.60, 10.26] |
| 3 Diary Card Symptom Score (Day) change from baseline | 2 | 647 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.16, 0.03] |
| 3.1 Adults | 2 | 647 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.16, 0.03] |
| 3.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Diary Card Symptom Score (Night) change from baseline | 2 | 645 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [‐0.04, 0.15] |
| 4.1 Adults | 2 | 645 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [‐0.04, 0.15] |
| 4.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Rescue Medications (puffs per day) change from baseline | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.45, 0.08] |
| 5.1 Adults | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.45, 0.08] |
| 5.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per night) change from baseline | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.13, 0.05] |
| 6.1 Adults | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.13, 0.05] |
| 7 Withdrawal due to adverse event | 2 | 850 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.73, 3.24] |
| 7.1 Adults | 2 | 850 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.73, 3.24] |
| 8 Proportion Achieving Asthma Control | 2 | 366 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.87, 1.16] |
| 8.1 Adults | 2 | 366 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.87, 1.16] |
| 8.3 Infants | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
1.1. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 1 Morning PEF (L/min).
1.2. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 2 Morning PEF (L/min) change from baseline.
1.3. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 3 Diary Card Symptom Score (Day) change from baseline.
1.4. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 4 Diary Card Symptom Score (Night) change from baseline.
1.5. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 5 Rescue Medications (puffs per day) change from baseline.
1.6. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 6 Rescue Medications (puffs per night) change from baseline.
1.7. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 7 Withdrawal due to adverse event.
1.8. Analysis.
Comparison 1 Step down vs Constant ICS Dose (All), Outcome 8 Proportion Achieving Asthma Control.
Comparison 2. High vs Moderate ICS Dose (All).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 (% predicted) | 4 | 130 | Mean Difference (IV, Fixed, 95% CI) | 5.32 [0.65, 9.99] |
| 1.1 Adults | 2 | 55 | Mean Difference (IV, Fixed, 95% CI) | 10.34 [2.45, 18.24] |
| 1.2 Children | 2 | 75 | Mean Difference (IV, Fixed, 95% CI) | 2.61 [‐3.18, 8.40] |
| 2 Morning PEF (L/min) | 2 | 63 | Mean Difference (IV, Fixed, 95% CI) | 7.91 [‐16.17, 32.00] |
| 2.1 Adults | 1 | 12 | Mean Difference (IV, Fixed, 95% CI) | ‐75.0 [‐228.70, 78.70] |
| 2.2 Children | 1 | 51 | Mean Difference (IV, Fixed, 95% CI) | 10.0 [‐14.39, 34.39] |
| 3 Morning PEF (L/min) change from baseline | 5 | 1117 | Mean Difference (IV, Fixed, 95% CI) | 5.72 [‐1.56, 13.00] |
| 3.1 Adults | 5 | 1117 | Mean Difference (IV, Fixed, 95% CI) | 5.72 [‐1.56, 13.00] |
| 4 Diary Card Symptom Score (day) change baseline | 2 | 794 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.11] |
| 4.1 Adults | 2 | 794 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.11] |
| 5 Diary Card Symptom Score (night) change from baseline | 2 | 800 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.10] |
| 5.1 Adults | 2 | 800 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.10] |
| 6 Rescue Medications (puffs per day) change from baseline | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.39, 0.17] |
| 6.1 Adults | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.39, 0.17] |
| 7 Rescue Medications (puffs per night) change from baseline | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.12, 0.05] |
| 7.1 Adults | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.12, 0.05] |
| 8 Withdrawal Due to Adverse Event | 2 | 301 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.14, 2.43] |
| 8.1 Adults | 2 | 301 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.14, 2.43] |
2.1. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 1 FEV1 (% predicted).
2.2. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 2 Morning PEF (L/min).
2.3. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 3 Morning PEF (L/min) change from baseline.
2.4. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 4 Diary Card Symptom Score (day) change baseline.
2.5. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 5 Diary Card Symptom Score (night) change from baseline.
2.6. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 6 Rescue Medications (puffs per day) change from baseline.
2.7. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 7 Rescue Medications (puffs per night) change from baseline.
2.8. Analysis.
Comparison 2 High vs Moderate ICS Dose (All), Outcome 8 Withdrawal Due to Adverse Event.
Comparison 3. High vs Low ICS Dose (All).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 | 2 | 123 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.55, 0.16] |
| 1.1 Adults | 2 | 123 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.55, 0.16] |
| 1.2 Children | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Morning PEF (L/min) | 2 | 95 | Mean Difference (IV, Fixed, 95% CI) | ‐18.05 [‐56.04, 19.95] |
| 3.1 Adults | 2 | 95 | Mean Difference (IV, Fixed, 95% CI) | ‐18.05 [‐56.04, 19.95] |
| 3.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Morning PEF (L/min) change from baseline | 2 | 282 | Mean Difference (IV, Fixed, 95% CI) | 12.00 [‐1.50, 25.51] |
| 4.1 Adults | 2 | 282 | Mean Difference (IV, Fixed, 95% CI) | 12.00 [‐1.50, 25.51] |
| 4.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per day) | 2 | 95 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.65, 0.56] |
| 6.1 Adults | 2 | 95 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.65, 0.56] |
| 9 Withdrawal Due to Adverse Event | 2 | 295 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.14, 2.33] |
| 9.1 Adults | 2 | 295 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.14, 2.33] |
3.1. Analysis.
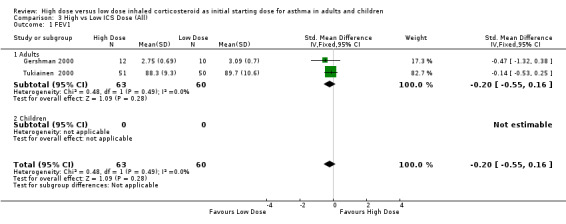
Comparison 3 High vs Low ICS Dose (All), Outcome 1 FEV1.
3.3. Analysis.
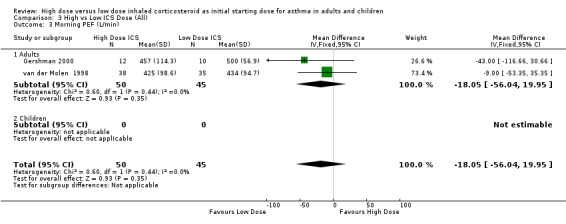
Comparison 3 High vs Low ICS Dose (All), Outcome 3 Morning PEF (L/min).
3.4. Analysis.
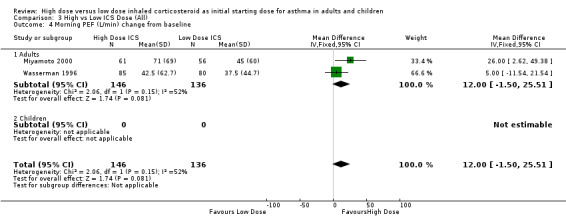
Comparison 3 High vs Low ICS Dose (All), Outcome 4 Morning PEF (L/min) change from baseline.
3.6. Analysis.
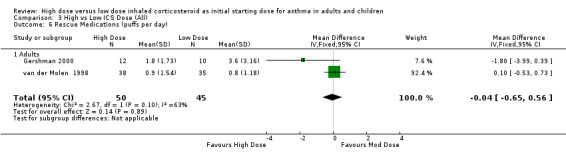
Comparison 3 High vs Low ICS Dose (All), Outcome 6 Rescue Medications (puffs per day).
3.9. Analysis.
Comparison 3 High vs Low ICS Dose (All), Outcome 9 Withdrawal Due to Adverse Event.
Comparison 4. Moderate vs Low ICS Dose (All).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 change from baseline | 5 | 536 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.11 [‐0.06, 0.28] |
| 1.1 Adults | 4 | 299 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.15 [‐0.08, 0.38] |
| 1.2 Children | 1 | 237 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.07 [‐0.19, 0.32] |
| 2 Morning PEF (L/min) change baseline | 5 | 411 | Mean Difference (IV, Fixed, 95% CI) | 11.14 [1.34, 20.93] |
| 2.1 Adults | 5 | 411 | Mean Difference (IV, Fixed, 95% CI) | 11.14 [1.34, 20.93] |
| 2.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Evening PEF (L/min) change from baseline | 3 | 241 | Mean Difference (IV, Fixed, 95% CI) | 4.30 [‐6.16, 14.75] |
| 3.1 Adults | 3 | 241 | Mean Difference (IV, Fixed, 95% CI) | 4.30 [‐6.16, 14.75] |
| 4 PEF Diurnal Variation change from baseline | 2 | 126 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐0.63, 0.07] |
| 4.1 Adults | 2 | 126 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐0.63, 0.07] |
| 5 Symptom Score change baseline | 2 | 75 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.21, 0.13] |
| 5.1 Adults | 2 | 75 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.21, 0.13] |
| 5.2 Children | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Symptom (Night Waking) change from baseline | 3 | 285 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.29 [‐0.53, ‐0.06] |
| 6.1 Adults | 3 | 285 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.29 [‐0.53, ‐0.06] |
| 7 Rescue Medications (no. of puffs) change baseline | 4 | 467 | Mean Difference (IV, Fixed, 95% CI) | ‐0.35 [‐0.98, 0.28] |
| 7.1 Adults | 3 | 230 | Mean Difference (IV, Fixed, 95% CI) | ‐0.35 [‐0.99, 0.29] |
| 7.2 Children | 1 | 237 | Mean Difference (IV, Fixed, 95% CI) | ‐0.32 [‐3.29, 2.65] |
| 8 Withdrawal due to Adverse Event | 3 | 523 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.41, 2.46] |
| 8.1 Adults | 1 | 130 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.16, 3.03] |
| 8.2 Children | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.06, 16.21] |
| 8.3 Infants | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.32 [0.37, 4.72] |
| 9 Adverse Events | 4 | 523 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.93, 1.17] |
| 9.1 Adults | 2 | 130 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.0 [0.19, 20.90] |
| 9.2 Children | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.86, 1.20] |
| 9.3 Infants | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.07 [0.92, 1.24] |
| 10 Oral Candidiasis | 2 | 311 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.34, 2.39] |
| 10.1 Adults | 1 | 155 | Risk Ratio (M‐H, Fixed, 95% CI) | 8.66 [0.47, 158.21] |
| 10.3 Infants | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.39 [0.11, 1.43] |
| 11 Exacerbations | 2 | 393 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.47, 1.43] |
| 11.1 Adults | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 Children | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.26, 9.04] |
| 11.3 Infants | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.42, 1.35] |
| 12 Plasma Cortisol below reference range | 2 | 399 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.30, 1.27] |
| 12.1 Adults | 1 | 162 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.13, 1.88] |
| 12.2 Children | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.29, 1.61] |
4.1. Analysis.
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 1 FEV1 change from baseline.
4.2. Analysis.
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 2 Morning PEF (L/min) change baseline.
4.3. Analysis.
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 3 Evening PEF (L/min) change from baseline.
4.4. Analysis.
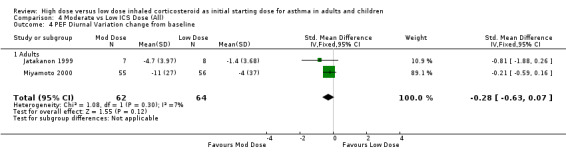
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 4 PEF Diurnal Variation change from baseline.
4.5. Analysis.
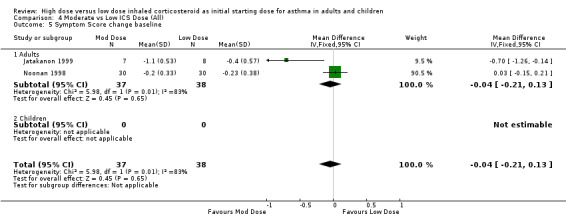
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 5 Symptom Score change baseline.
4.6. Analysis.
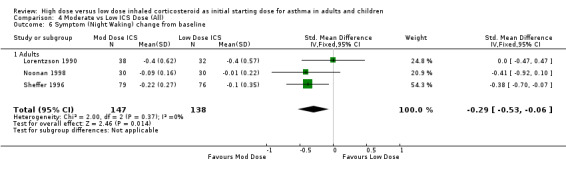
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 6 Symptom (Night Waking) change from baseline.
4.7. Analysis.
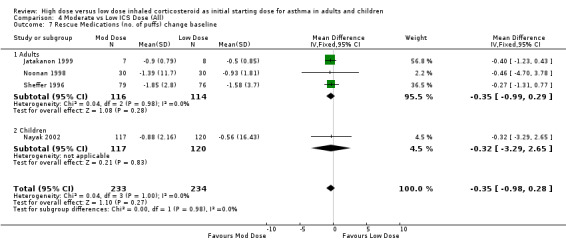
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 7 Rescue Medications (no. of puffs) change baseline.
4.8. Analysis.
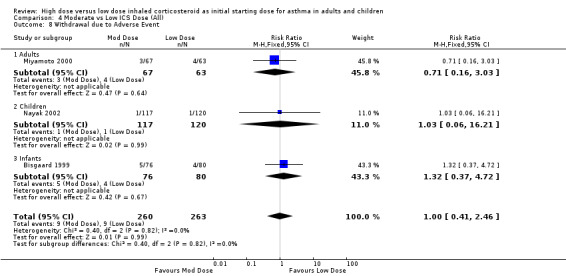
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 8 Withdrawal due to Adverse Event.
4.9. Analysis.
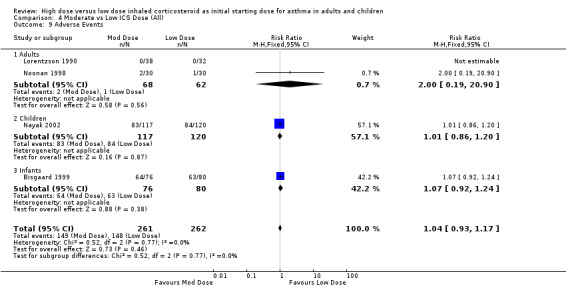
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 9 Adverse Events.
4.10. Analysis.
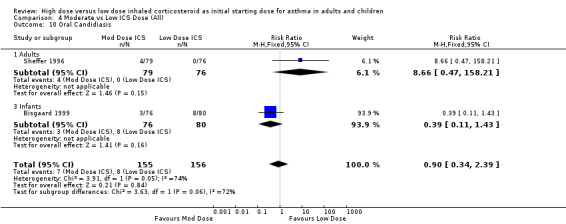
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 10 Oral Candidiasis.
4.11. Analysis.
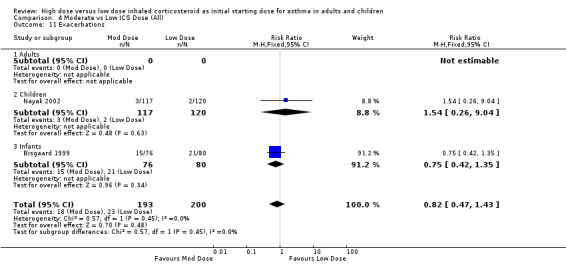
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 11 Exacerbations.
4.12. Analysis.
Comparison 4 Moderate vs Low ICS Dose (All), Outcome 12 Plasma Cortisol below reference range.
Comparison 5. Fold Difference in ICS Dose (All).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 | 5 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 > = 4 Fold Change | 4 | 217 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.04 [‐0.23, 0.31] |
| 1.2 2 Fold Change | 1 | 12 | Std. Mean Difference (IV, Fixed, 95% CI) | 1.13 [‐0.13, 2.39] |
| 2 FEV1 change from baseline | 6 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 >= 4 Fold Change | 3 | 240 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.18 [‐0.08, 0.43] |
| 2.2 2 Fold Change | 5 | 704 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.14 [‐0.01, 0.29] |
| 3 Morning PEF (L/min) | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 >= 4 Fold Change | 3 | 146 | Mean Difference (IV, Fixed, 95% CI) | 1.82 [‐18.70, 22.34] |
| 3.2 2 Fold Change | 1 | 12 | Mean Difference (IV, Fixed, 95% CI) | ‐75.0 [‐228.70, 78.70] |
| 4 Morning PEF (L/min) change from baseline | 10 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 >= 4 Fold Change | 5 | 621 | Mean Difference (IV, Fixed, 95% CI) | 10.21 [1.75, 18.67] |
| 4.2 2 Fold Change | 8 | 1406 | Mean Difference (IV, Fixed, 95% CI) | 6.80 [0.75, 12.84] |
| 5 Evening PEF (L/min) change from baseline | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 >= 4 Fold Change | 1 | 117 | Mean Difference (IV, Fixed, 95% CI) | 15.00 [‐6.39, 36.39] |
| 5.2 2 Fold Change | 4 | 424 | Mean Difference (IV, Fixed, 95% CI) | 2.37 [‐5.78, 10.53] |
| 6 PEF Diurnal Variation change from baseline | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 >= 4 Fold Change | 2 | 132 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.32 [‐0.66, 0.03] |
| 6.2 2 Fold Change | 1 | 111 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.21 [‐0.59, 0.16] |
| 7 Diary Card Symptom Score (Day) change from baseline | 3 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 >= 4 Fold Change | 1 | 169 | Mean Difference (IV, Fixed, 95% CI) | 0.11 [‐0.08, 0.30] |
| 7.2 2 Fold Change | 2 | 695 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [‐0.09, 0.09] |
| 8 Symptoms (Night Waking) change from baseline | 5 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 8.1 >= 4 Fold Change | 2 | 324 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.17 [‐0.38, 0.05] |
| 8.2 2 Fold Change | 4 | 916 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.02 [‐0.15, 0.11] |
| 9 Rescue Medications (puffs per day) change from baseline | 3 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 9.1 >= 4 Fold Change | 1 | 169 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.37, 0.57] |
| 9.2 2 Fold Change | 2 | 689 | Mean Difference (IV, Fixed, 95% CI) | ‐0.21 [‐0.55, 0.14] |
| 10 Rescue Medications (puffs per night) change from baseline | 3 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 10.1 >= 4 Fold Change | 1 | 169 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.42, 0.22] |
| 10.2 2 Fold Change | 2 | 689 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.11, 0.06] |
| 11 Rescue Medications (no. of puffs) change baseline | 5 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 11.1 >= 4 Fold Change | 2 | 170 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.42, 0.19] |
| 11.2 2 Fold Change | 4 | 635 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.01 [‐0.16, 0.15] |
| 12 Withdrawal due to adverse event | 5 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 12.1 >= 4 Fold Change | 2 | 295 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.14, 2.33] |
| 12.2 2 Fold Change | 5 | 868 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.09 [0.49, 2.42] |
| 13 Oral Candidiasis | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 13.1 >= 4 Fold Change | 2 | 320 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.30 [0.68, 7.83] |
| 13.2 2 Fold Change | 3 | 473 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.27, 1.64] |
| 14 Plasma Cortisol below reference range | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 14.1 >= 4 Fold Change | 1 | 165 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.18, 2.14] |
| 14.2 2 Fold Change | 2 | 399 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.30, 1.27] |
5.1. Analysis.
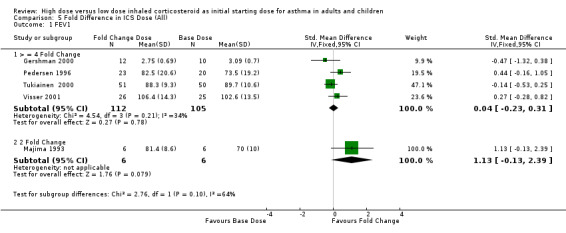
Comparison 5 Fold Difference in ICS Dose (All), Outcome 1 FEV1.
5.2. Analysis.
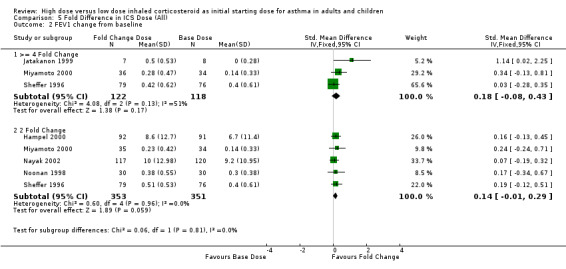
Comparison 5 Fold Difference in ICS Dose (All), Outcome 2 FEV1 change from baseline.
5.3. Analysis.
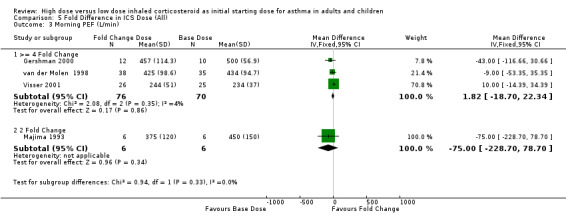
Comparison 5 Fold Difference in ICS Dose (All), Outcome 3 Morning PEF (L/min).
5.4. Analysis.
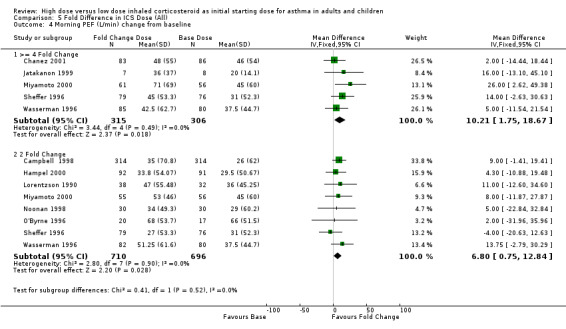
Comparison 5 Fold Difference in ICS Dose (All), Outcome 4 Morning PEF (L/min) change from baseline.
5.5. Analysis.
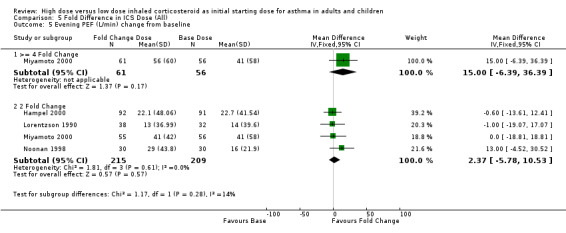
Comparison 5 Fold Difference in ICS Dose (All), Outcome 5 Evening PEF (L/min) change from baseline.
5.6. Analysis.
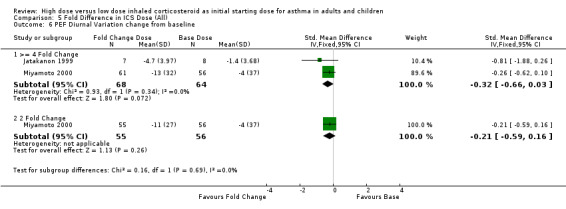
Comparison 5 Fold Difference in ICS Dose (All), Outcome 6 PEF Diurnal Variation change from baseline.
5.7. Analysis.
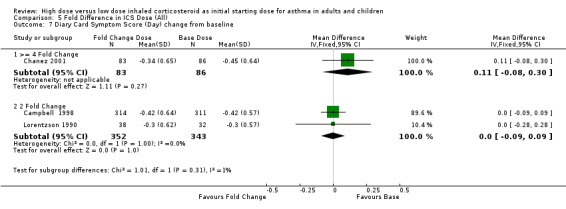
Comparison 5 Fold Difference in ICS Dose (All), Outcome 7 Diary Card Symptom Score (Day) change from baseline.
5.8. Analysis.
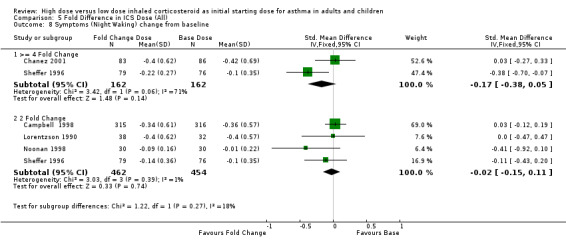
Comparison 5 Fold Difference in ICS Dose (All), Outcome 8 Symptoms (Night Waking) change from baseline.
5.9. Analysis.
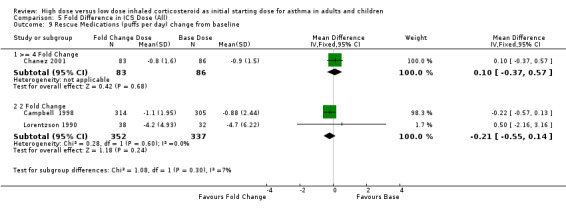
Comparison 5 Fold Difference in ICS Dose (All), Outcome 9 Rescue Medications (puffs per day) change from baseline.
5.10. Analysis.
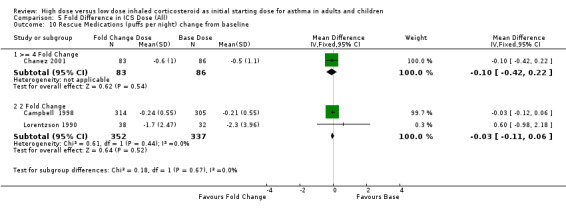
Comparison 5 Fold Difference in ICS Dose (All), Outcome 10 Rescue Medications (puffs per night) change from baseline.
5.11. Analysis.
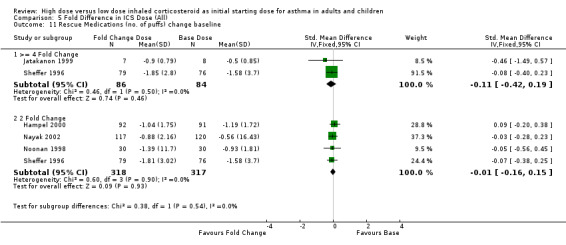
Comparison 5 Fold Difference in ICS Dose (All), Outcome 11 Rescue Medications (no. of puffs) change baseline.
5.12. Analysis.
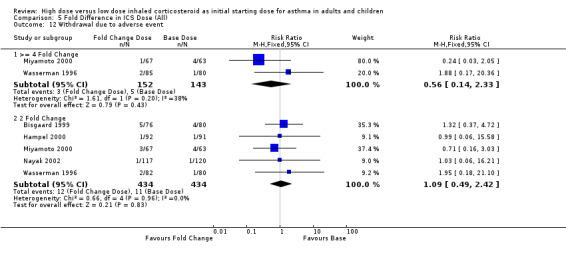
Comparison 5 Fold Difference in ICS Dose (All), Outcome 12 Withdrawal due to adverse event.
5.13. Analysis.
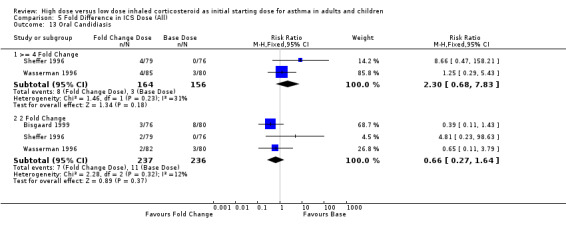
Comparison 5 Fold Difference in ICS Dose (All), Outcome 13 Oral Candidiasis.
5.14. Analysis.
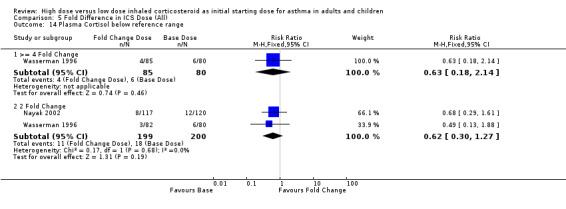
Comparison 5 Fold Difference in ICS Dose (All), Outcome 14 Plasma Cortisol below reference range.
Comparison 6. Step down vs Constant ICS Dose (ICS Type).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 BUD | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | ‐3.0 [‐48.76, 42.76] |
| 1.2 FP | 1 | 51 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [‐30.22, 30.22] |
| 2 Morning PEF (L/min) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 BUD | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | 0.83 [‐8.60, 10.26] |
| 2.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Diary Card Symptom Score (Day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 BUD | 2 | 647 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.16, 0.03] |
| 3.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Diary Card Symptom Score (Night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 BUD | 2 | 645 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [‐0.04, 0.15] |
| 4.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Rescue Medications (puffs per day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 BUD | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.45, 0.08] |
| 5.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 BUD | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.13, 0.05] |
| 6.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Withdrawal due to adverse event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 7.1 BUD | 2 | 850 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.73, 3.24] |
| 7.2 FP | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Proportion Achieving Asthma Control | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 BUD | 2 | 366 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.87, 1.16] |
| 8.2 FP | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
6.1. Analysis.
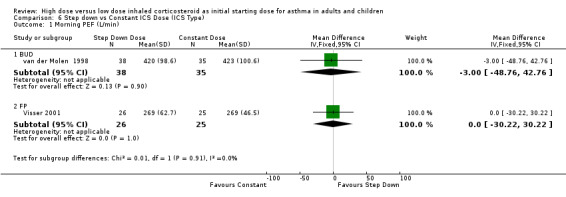
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 1 Morning PEF (L/min).
6.2. Analysis.
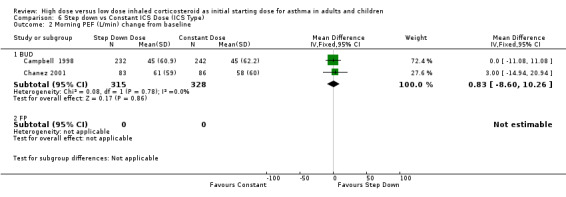
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 2 Morning PEF (L/min) change from baseline.
6.3. Analysis.
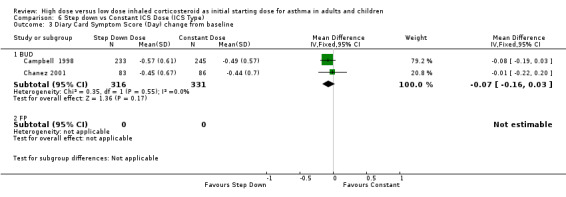
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 3 Diary Card Symptom Score (Day) change from baseline.
6.4. Analysis.
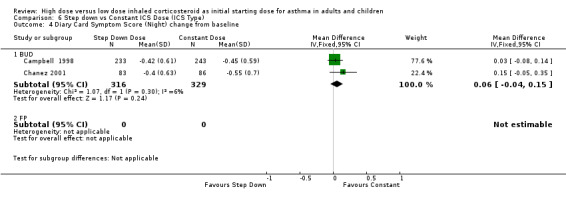
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 4 Diary Card Symptom Score (Night) change from baseline.
6.5. Analysis.
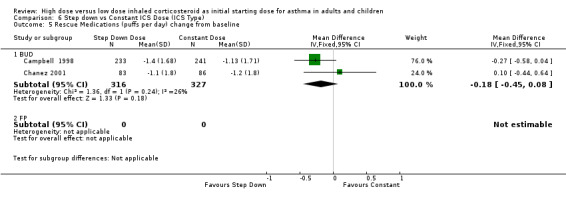
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 5 Rescue Medications (puffs per day) change from baseline.
6.6. Analysis.
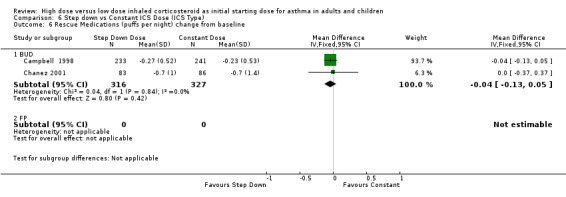
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 6 Rescue Medications (puffs per night) change from baseline.
6.7. Analysis.
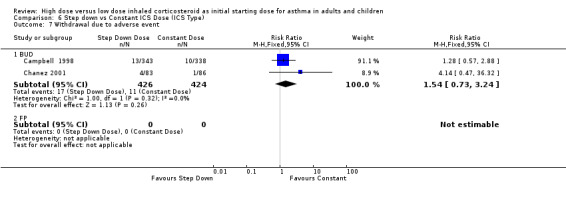
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 7 Withdrawal due to adverse event.
6.8. Analysis.
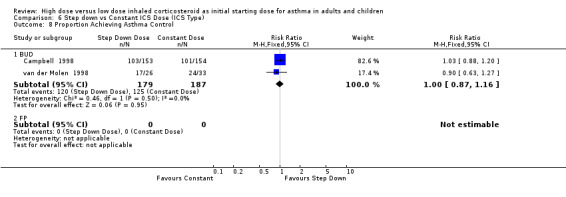
Comparison 6 Step down vs Constant ICS Dose (ICS Type), Outcome 8 Proportion Achieving Asthma Control.
Comparison 7. High vs Moderate ICS Dose (ICS Type).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 (% predicted) | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 BUD | 1 | 43 | Mean Difference (IV, Fixed, 95% CI) | 9.0 [‐2.90, 20.90] |
| 1.2 FP | 2 | 75 | Mean Difference (IV, Fixed, 95% CI) | 2.61 [‐3.18, 8.40] |
| 1.3 BDP | 1 | 12 | Mean Difference (IV, Fixed, 95% CI) | 11.40 [0.85, 21.95] |
| 2 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 BUD | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.2 FP | 1 | 51 | Mean Difference (IV, Fixed, 95% CI) | 10.0 [‐14.39, 34.39] |
| 2.3 BDP | 1 | 12 | Mean Difference (IV, Fixed, 95% CI) | ‐75.0 [‐228.70, 78.70] |
| 3 Morning PEF (L/min) change from baseline | 5 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 BUD | 4 | 950 | Mean Difference (IV, Fixed, 95% CI) | 8.26 [0.36, 16.16] |
| 3.2 FP | 1 | 167 | Mean Difference (IV, Fixed, 95% CI) | ‐8.75 [‐27.60, 10.10] |
| 4 Diary Card Symptom Score (day) change baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 BUD | 2 | 794 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.11] |
| 4.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Diary Card Symptom Score (night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 BUD | 2 | 800 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.10] |
| 5.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 BUD | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.39, 0.17] |
| 6.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Rescue Medications (puffs per night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 BUD | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.12, 0.05] |
| 7.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Withdrawal Due to Adverse Event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 BUD | 1 | 134 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.04, 3.12] |
| 8.2 FP | 1 | 167 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.14, 6.69] |
7.1. Analysis.
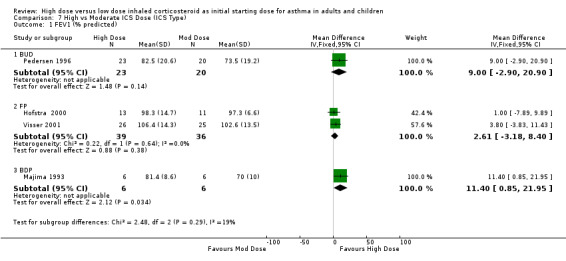
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 1 FEV1 (% predicted).
7.2. Analysis.
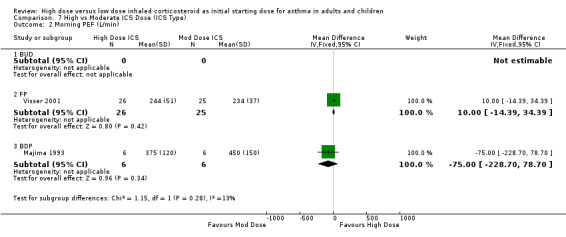
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 2 Morning PEF (L/min).
7.3. Analysis.
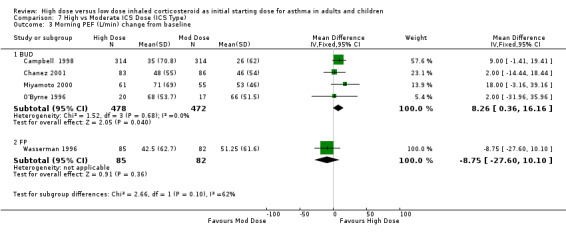
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 3 Morning PEF (L/min) change from baseline.
7.4. Analysis.
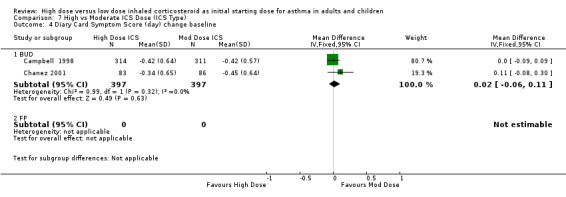
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 4 Diary Card Symptom Score (day) change baseline.
7.5. Analysis.
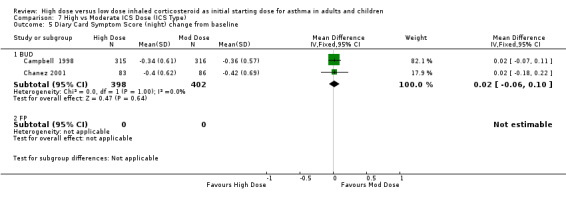
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 5 Diary Card Symptom Score (night) change from baseline.
7.6. Analysis.
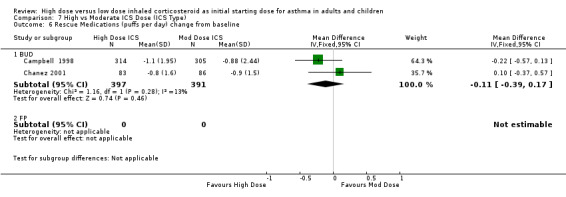
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 6 Rescue Medications (puffs per day) change from baseline.
7.7. Analysis.
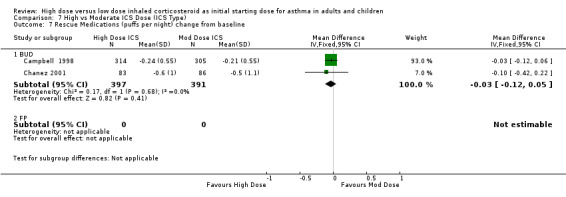
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 7 Rescue Medications (puffs per night) change from baseline.
7.8. Analysis.
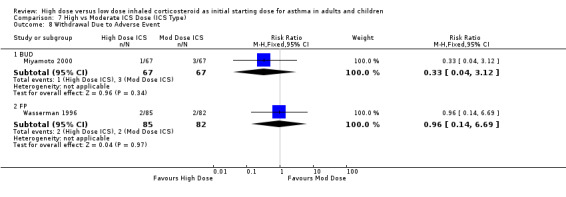
Comparison 7 High vs Moderate ICS Dose (ICS Type), Outcome 8 Withdrawal Due to Adverse Event.
Comparison 8. High vs Low ICS Dose (ICS Type).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 BUD | 1 | 101 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐0.53, 0.25] |
| 1.2 FP | 1 | 22 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.47 [‐1.32, 0.38] |
| 2 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 BUD | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | ‐9.0 [‐53.35, 35.35] |
| 2.2 FP | 1 | 22 | Mean Difference (IV, Fixed, 95% CI) | ‐43.0 [‐116.66, 30.66] |
| 3 Morning PEF (L/min) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 BUD | 2 | 282 | Mean Difference (IV, Fixed, 95% CI) | 12.00 [‐1.50, 25.51] |
| 3.2 FP | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Rescue Medications (no. of puffs) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 BUD | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.53, 0.73] |
| 4.2 FP | 1 | 22 | Mean Difference (IV, Fixed, 95% CI) | ‐1.8 [‐3.99, 0.39] |
| 5 Withdrawal Due to Adverse Event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 BUD | 1 | 130 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.24 [0.03, 2.05] |
| 5.2 FP | 1 | 165 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.88 [0.17, 20.36] |
8.1. Analysis.
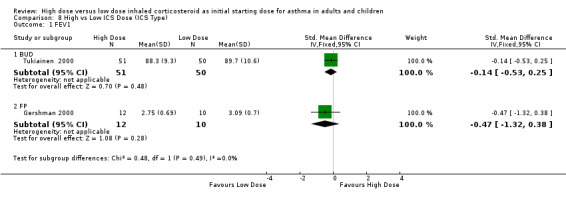
Comparison 8 High vs Low ICS Dose (ICS Type), Outcome 1 FEV1.
8.2. Analysis.
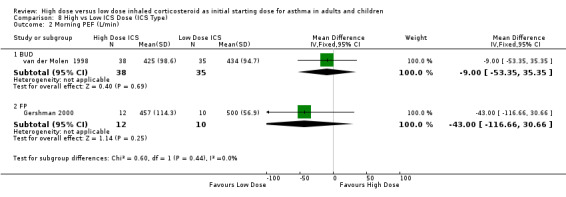
Comparison 8 High vs Low ICS Dose (ICS Type), Outcome 2 Morning PEF (L/min).
8.3. Analysis.
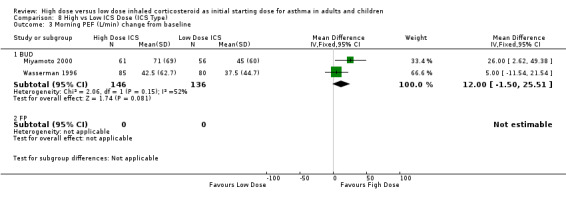
Comparison 8 High vs Low ICS Dose (ICS Type), Outcome 3 Morning PEF (L/min) change from baseline.
8.4. Analysis.
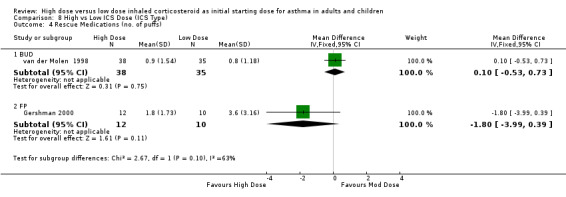
Comparison 8 High vs Low ICS Dose (ICS Type), Outcome 4 Rescue Medications (no. of puffs).
8.5. Analysis.
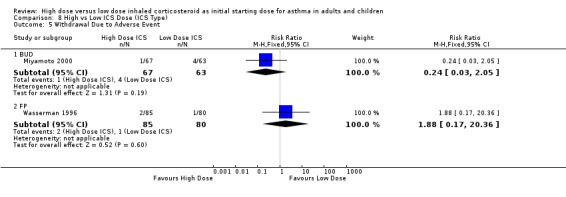
Comparison 8 High vs Low ICS Dose (ICS Type), Outcome 5 Withdrawal Due to Adverse Event.
Comparison 9. Moderate vs Low ICS Dose (ICS Type).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 change from baseline | 5 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 BUD | 2 | 84 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.37 [‐0.06, 0.81] |
| 1.2 FP | 2 | 215 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.07 [‐0.20, 0.34] |
| 1.3 BDP | 1 | 237 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.07 [‐0.19, 0.32] |
| 2 Morning PEF (L/min) change baseline | 5 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 BUD | 3 | 196 | Mean Difference (IV, Fixed, 95% CI) | 10.69 [‐2.78, 24.16] |
| 2.2 FP | 2 | 215 | Mean Difference (IV, Fixed, 95% CI) | 11.63 [‐2.64, 25.91] |
| 3 Evening PEF (L/min) change from baseline | 3 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 BUD | 2 | 181 | Mean Difference (IV, Fixed, 95% CI) | ‐0.52 [‐13.55, 12.51] |
| 3.2 FP | 1 | 60 | Mean Difference (IV, Fixed, 95% CI) | 13.00 [‐4.52, 30.52] |
| 4 PEF Diurnal Variation change from baseline | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 BUD | 2 | 126 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐0.63, 0.07] |
| 5 Symptom Score change baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 BUD | 1 | 15 | Mean Difference (IV, Fixed, 95% CI) | ‐0.70 [‐1.26, ‐0.14] |
| 5.2 FP | 1 | 60 | Mean Difference (IV, Fixed, 95% CI) | 0.03 [‐0.15, 0.21] |
| 6 Symptoms (night waking) change from baseline | 3 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 BUD | 1 | 70 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [‐0.47, 0.47] |
| 6.2 FP | 2 | 215 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.39 [‐0.66, ‐0.12] |
| 7 Rescue Medications (no. of puffs) change baseline | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 BUD | 1 | 15 | Mean Difference (IV, Fixed, 95% CI) | ‐0.4 [‐1.23, 0.43] |
| 7.2 FP | 2 | 215 | Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐1.29, 0.73] |
| 7.3 BDP | 1 | 237 | Mean Difference (IV, Fixed, 95% CI) | ‐0.32 [‐3.29, 2.65] |
| 8 Withdrawal due to Adverse Event | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 BUD | 1 | 130 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.16, 3.03] |
| 8.2 FP | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.32 [0.37, 4.72] |
| 8.3 BDP | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.06, 16.21] |
| 9 Adverse Events | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 9.1 BUD | 1 | 70 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.2 FP | 2 | 216 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [0.93, 1.27] |
| 9.3 BDP | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.86, 1.20] |
| 10 Oral Candidiasis | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 10.1 BUD | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10.2 FP | 2 | 311 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.34, 2.39] |
| 11 Exacerbations | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 11.1 BUD | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 FP | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.42, 1.35] |
| 11.3 BDP | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.26, 9.04] |
| 12 Plasma Cortisol below reference range | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 12.1 BUD | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.2 FP | 1 | 162 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.13, 1.88] |
| 12.3 BDP | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.29, 1.61] |
9.1. Analysis.
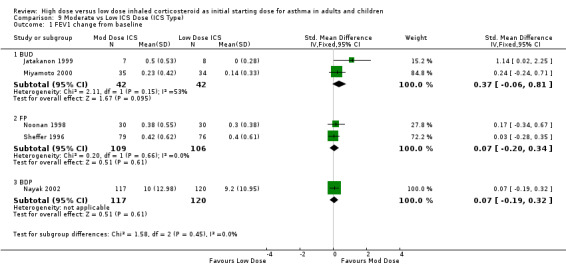
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 1 FEV1 change from baseline.
9.2. Analysis.
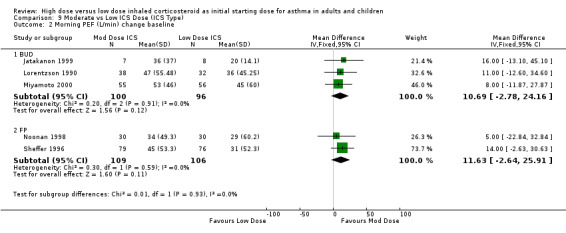
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 2 Morning PEF (L/min) change baseline.
9.3. Analysis.
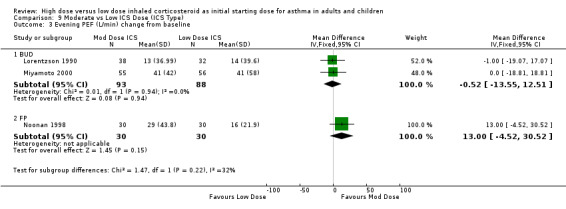
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 3 Evening PEF (L/min) change from baseline.
9.4. Analysis.
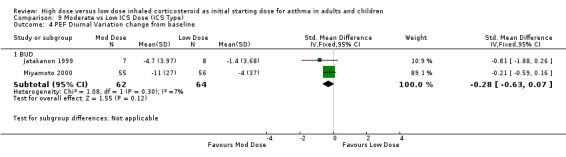
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 4 PEF Diurnal Variation change from baseline.
9.5. Analysis.
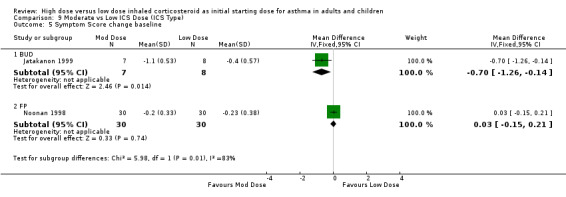
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 5 Symptom Score change baseline.
9.6. Analysis.
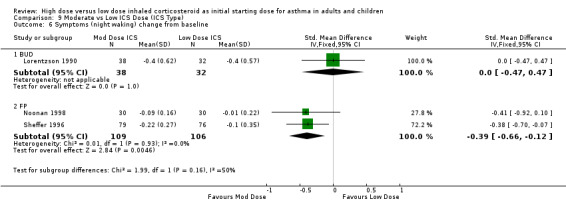
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 6 Symptoms (night waking) change from baseline.
9.7. Analysis.
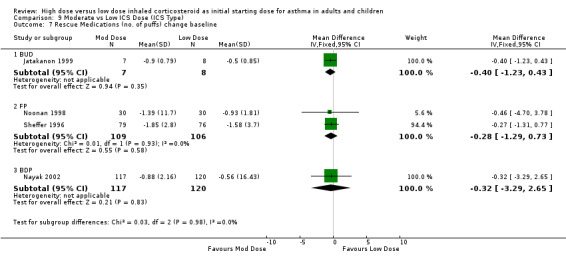
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 7 Rescue Medications (no. of puffs) change baseline.
9.8. Analysis.
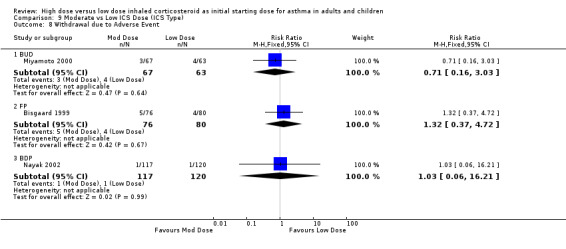
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 8 Withdrawal due to Adverse Event.
9.9. Analysis.
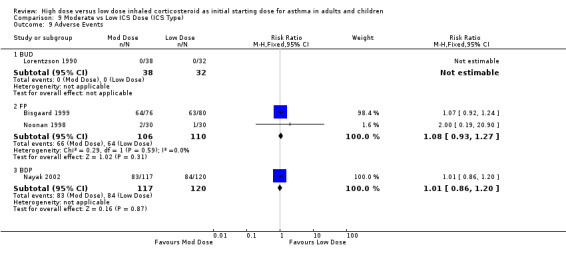
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 9 Adverse Events.
9.10. Analysis.
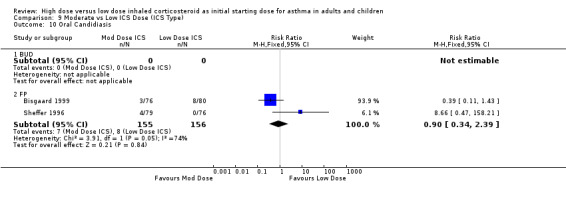
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 10 Oral Candidiasis.
9.11. Analysis.
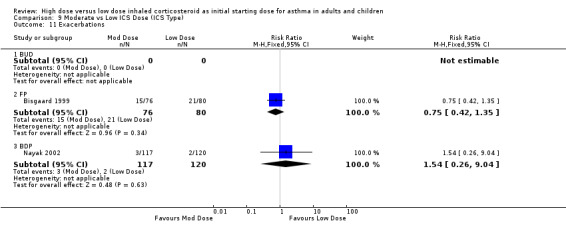
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 11 Exacerbations.
9.12. Analysis.
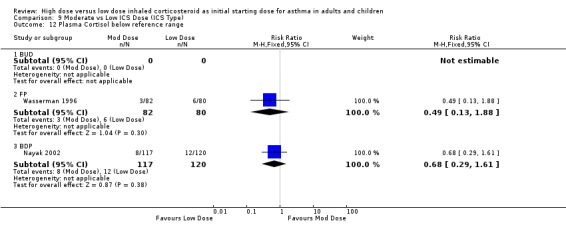
Comparison 9 Moderate vs Low ICS Dose (ICS Type), Outcome 12 Plasma Cortisol below reference range.
Comparison 10. Step down vs Constant ICS Dose (Device).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Morning PEF (L/min) | 1 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 DPI | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | ‐3.0 [‐48.76, 42.76] |
| 1.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Morning PEF (L/min) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 DPI | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | 0.83 [‐8.60, 10.26] |
| 2.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Diary Card Symptom Score (Day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 DPI | 2 | 647 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.16, 0.03] |
| 3.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Diary Card Symptom Score (Night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 DPI | 2 | 645 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [‐0.04, 0.15] |
| 4.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Rescue Medications (puffs per day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 DPI | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.45, 0.08] |
| 5.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 DPI | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.13, 0.05] |
| 6.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Withdrawal due to adverse event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 7.1 DPI | 2 | 850 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.73, 3.24] |
| 7.2 pMDI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Proportion Achieving Asthma Control | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 DPI | 2 | 366 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.87, 1.16] |
| 8.2 pMDI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
10.1. Analysis.
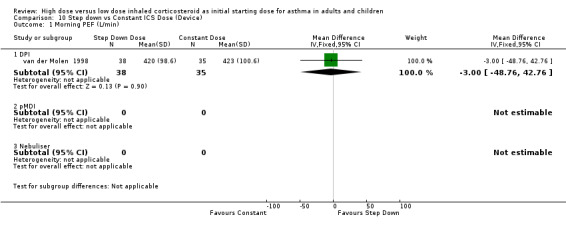
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 1 Morning PEF (L/min).
10.2. Analysis.
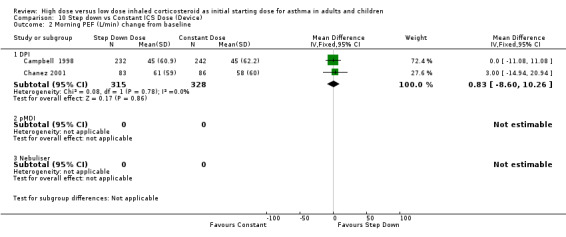
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 2 Morning PEF (L/min) change from baseline.
10.3. Analysis.
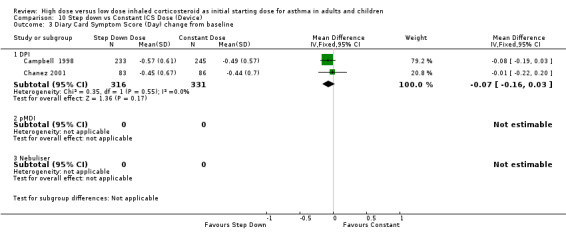
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 3 Diary Card Symptom Score (Day) change from baseline.
10.4. Analysis.
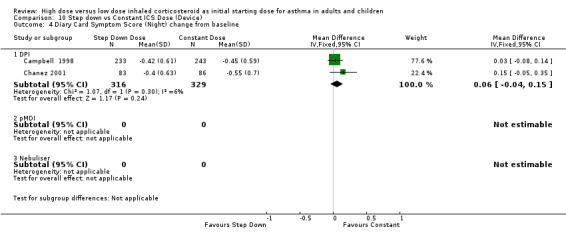
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 4 Diary Card Symptom Score (Night) change from baseline.
10.5. Analysis.
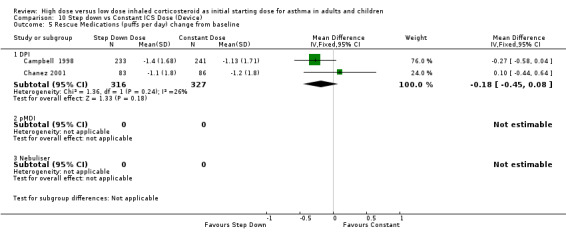
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 5 Rescue Medications (puffs per day) change from baseline.
10.6. Analysis.
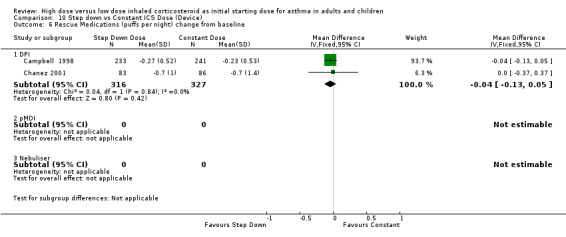
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 6 Rescue Medications (puffs per night) change from baseline.
10.7. Analysis.
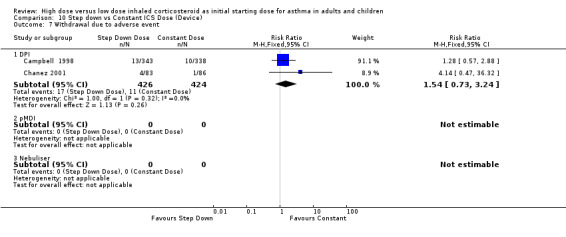
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 7 Withdrawal due to adverse event.
10.8. Analysis.
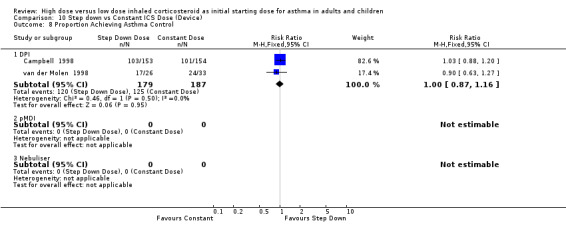
Comparison 10 Step down vs Constant ICS Dose (Device), Outcome 8 Proportion Achieving Asthma Control.
Comparison 11. High vs Moderate ICS Dose (Device).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 (% predicted) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 DPI | 1 | 43 | Mean Difference (IV, Fixed, 95% CI) | 9.0 [‐2.90, 20.90] |
| 1.2 pMDI | 1 | 24 | Mean Difference (IV, Fixed, 95% CI) | 1.0 [‐7.89, 9.89] |
| 1.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Morning PEF(L/min) change from baseline | 5 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 DPI | 4 | 1080 | Mean Difference (IV, Fixed, 95% CI) | 5.90 [‐1.56, 13.36] |
| 2.2 pMDI | 1 | 37 | Mean Difference (IV, Fixed, 95% CI) | 2.0 [‐31.96, 35.96] |
| 2.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Diary Card Symptom Score (day) change baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 DPI | 2 | 794 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.11] |
| 3.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Diary Card Symptom Score (night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 DPI | 2 | 800 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.10] |
| 4.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Rescue Medications (puffs per day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 DPI | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.39, 0.17] |
| 5.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 DPI | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.12, 0.05] |
| 6.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Withdrawal Due to Adverse Event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 7.1 DPI | 2 | 301 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.14, 2.43] |
| 7.2 pMDI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
11.1. Analysis.
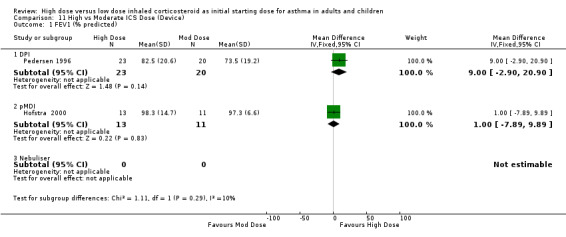
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 1 FEV1 (% predicted).
11.2. Analysis.
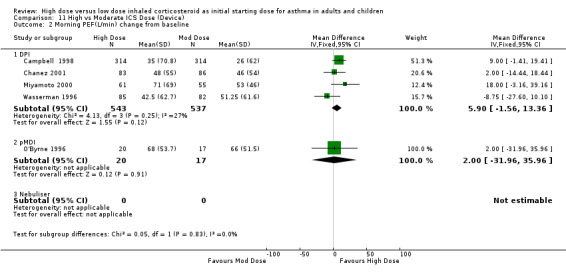
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 2 Morning PEF(L/min) change from baseline.
11.3. Analysis.
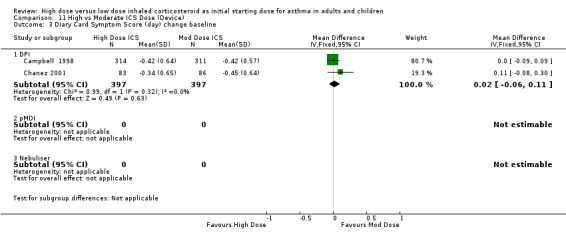
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 3 Diary Card Symptom Score (day) change baseline.
11.4. Analysis.
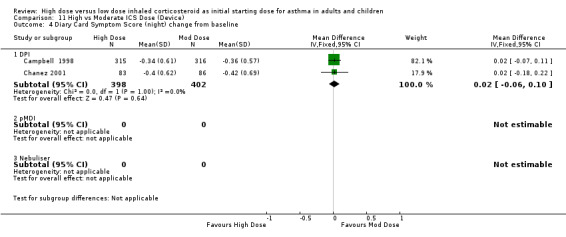
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 4 Diary Card Symptom Score (night) change from baseline.
11.5. Analysis.
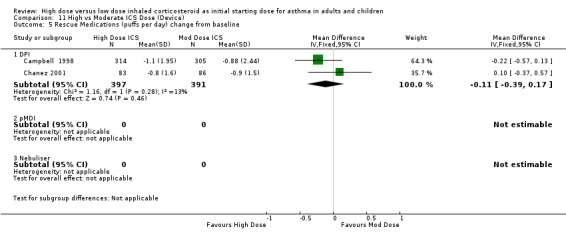
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 5 Rescue Medications (puffs per day) change from baseline.
11.6. Analysis.
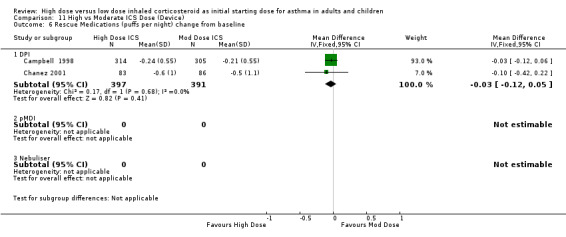
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 6 Rescue Medications (puffs per night) change from baseline.
11.7. Analysis.
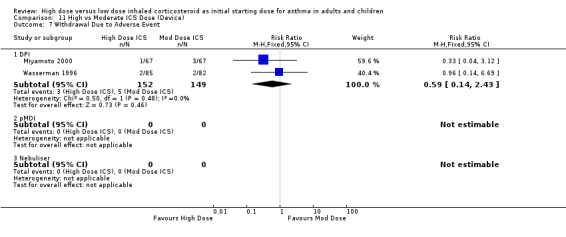
Comparison 11 High vs Moderate ICS Dose (Device), Outcome 7 Withdrawal Due to Adverse Event.
Comparison 12. High vs Low ICS Dose (Device).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 DPI | 1 | 101 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐0.53, 0.25] |
| 1.2 pMDI | 1 | 22 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.47 [‐1.32, 0.38] |
| 1.3 Nebuliser | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 DPI | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | ‐9.0 [‐53.35, 35.35] |
| 2.2 pMDI | 1 | 22 | Mean Difference (IV, Fixed, 95% CI) | ‐43.0 [‐116.66, 30.66] |
| 2.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Morning PEF (L/min) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 DPI | 2 | 282 | Mean Difference (IV, Fixed, 95% CI) | 12.00 [‐1.50, 25.51] |
| 3.2 pMDI | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Rescue Medications (no. of puffs) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 DPI | 1 | 73 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.53, 0.73] |
| 4.2 pMDI | 1 | 22 | Mean Difference (IV, Fixed, 95% CI) | ‐1.8 [‐3.99, 0.39] |
| 4.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Withdrawal Due to Adverse Event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 DPI | 2 | 295 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.14, 2.33] |
| 5.2 pMDI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
12.1. Analysis.
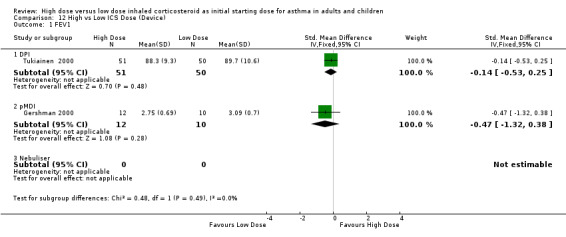
Comparison 12 High vs Low ICS Dose (Device), Outcome 1 FEV1.
12.2. Analysis.
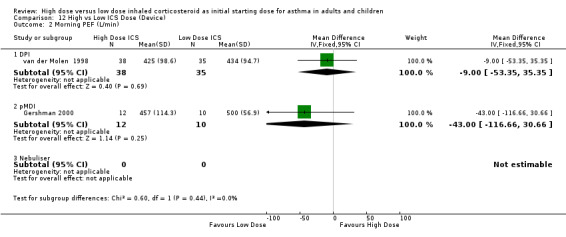
Comparison 12 High vs Low ICS Dose (Device), Outcome 2 Morning PEF (L/min).
12.3. Analysis.
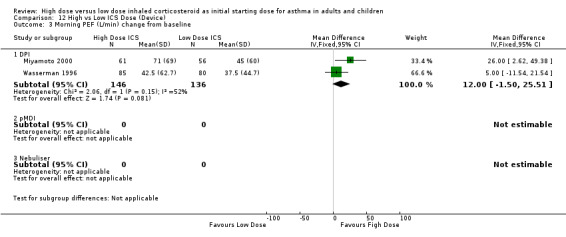
Comparison 12 High vs Low ICS Dose (Device), Outcome 3 Morning PEF (L/min) change from baseline.
12.4. Analysis.
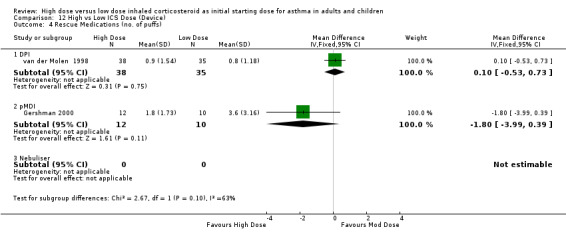
Comparison 12 High vs Low ICS Dose (Device), Outcome 4 Rescue Medications (no. of puffs).
12.5. Analysis.
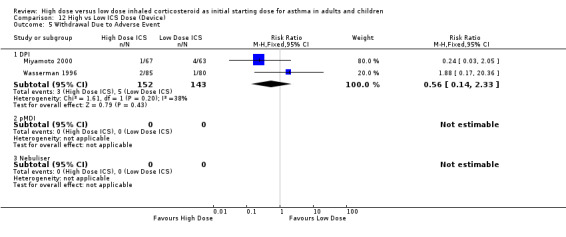
Comparison 12 High vs Low ICS Dose (Device), Outcome 5 Withdrawal Due to Adverse Event.
Comparison 13. Moderate vs Low ICS Dose (Device).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 change from baseline | 5 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 DPI | 2 | 84 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.37 [‐0.06, 0.81] |
| 1.2 pMDI | 3 | 452 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.07 [‐0.12, 0.25] |
| 1.3 Nebuliser | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Morning PEF (L/min) change baseline | 5 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 DPI | 2 | 126 | Mean Difference (IV, Fixed, 95% CI) | 10.54 [‐5.86, 26.95] |
| 2.2 pMDI | 3 | 285 | Mean Difference (IV, Fixed, 95% CI) | 11.46 [‐0.75, 23.68] |
| 2.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Evening PEF (L/min) change from baseline | 3 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 DPI | 1 | 111 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [‐18.81, 18.81] |
| 3.2 pMDI | 2 | 130 | Mean Difference (IV, Fixed, 95% CI) | 6.22 [‐6.36, 18.80] |
| 3.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 PEF Diurnal Variation change from baseline | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 DPI | 2 | 126 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐0.63, 0.07] |
| 4.2 pMDI | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Nebuliser | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Symptom Score change baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 DPI | 1 | 15 | Mean Difference (IV, Fixed, 95% CI) | ‐0.70 [‐1.26, ‐0.14] |
| 5.2 pMDI | 1 | 60 | Mean Difference (IV, Fixed, 95% CI) | 0.03 [‐0.15, 0.21] |
| 5.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Symptom (Night Waking) change from baseline | 3 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 DPI | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.2 pMDI | 3 | 285 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.29 [‐0.53, ‐0.06] |
| 6.3 Nebuliser | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Rescue Medications (no. of puffs) change baseline | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 DPI | 1 | 15 | Mean Difference (IV, Fixed, 95% CI) | ‐0.4 [‐1.23, 0.43] |
| 7.2 pMDI | 3 | 452 | Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐1.24, 0.67] |
| 7.3 Nebuliser | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Withdrawal due to Adverse Event | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 DPI | 1 | 130 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.16, 3.03] |
| 8.2 pMDI | 2 | 393 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.26 [0.40, 4.00] |
| 8.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Adverse Events | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 9.1 DPI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.2 pMDI | 4 | 523 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.93, 1.17] |
| 9.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10 Oral Candidiasis | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 10.1 DPI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10.2 pMDI | 2 | 311 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.34, 2.39] |
| 10.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11 Exacerbations | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 11.1 DPI | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 pMDI | 2 | 393 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.47, 1.43] |
| 11.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12 Plasma Cortisol below reference range | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 12.1 DPI | 1 | 162 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.13, 1.88] |
| 12.2 pMDI | 1 | 237 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.29, 1.61] |
| 12.3 Nebuliser | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
13.1. Analysis.
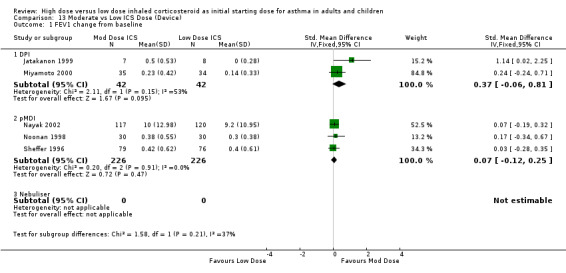
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 1 FEV1 change from baseline.
13.2. Analysis.
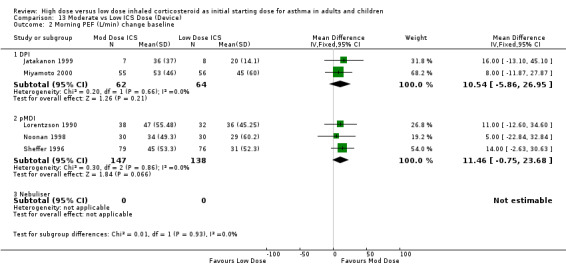
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 2 Morning PEF (L/min) change baseline.
13.3. Analysis.
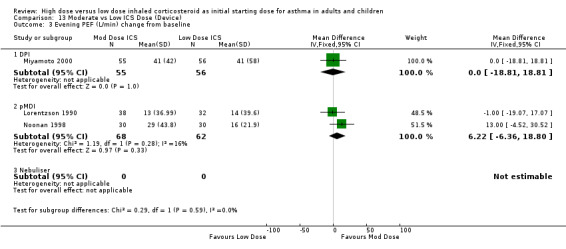
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 3 Evening PEF (L/min) change from baseline.
13.4. Analysis.
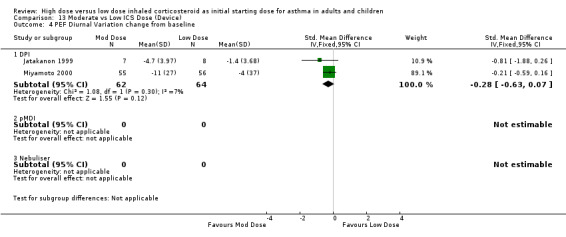
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 4 PEF Diurnal Variation change from baseline.
13.5. Analysis.
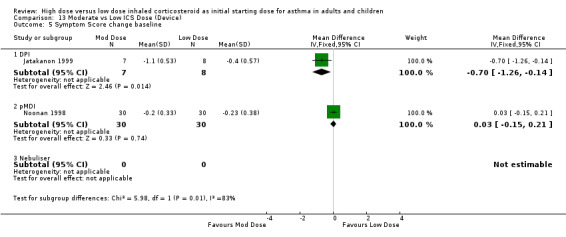
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 5 Symptom Score change baseline.
13.6. Analysis.
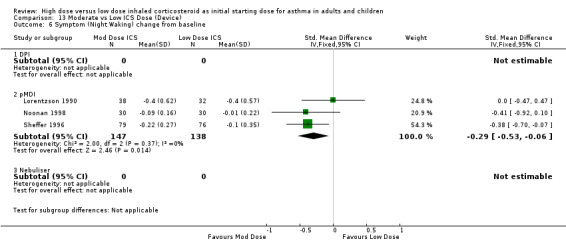
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 6 Symptom (Night Waking) change from baseline.
13.7. Analysis.
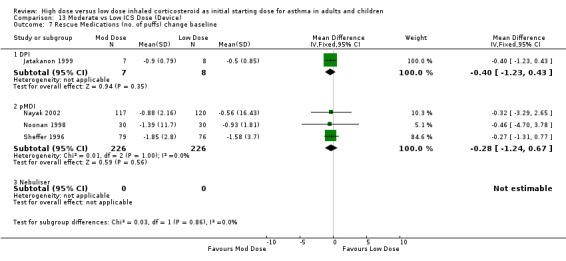
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 7 Rescue Medications (no. of puffs) change baseline.
13.8. Analysis.
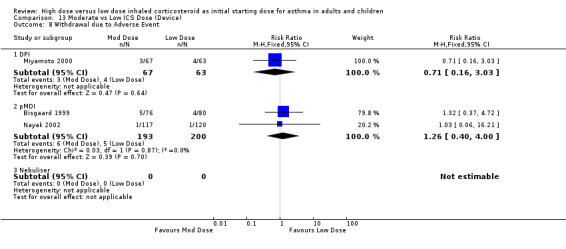
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 8 Withdrawal due to Adverse Event.
13.9. Analysis.
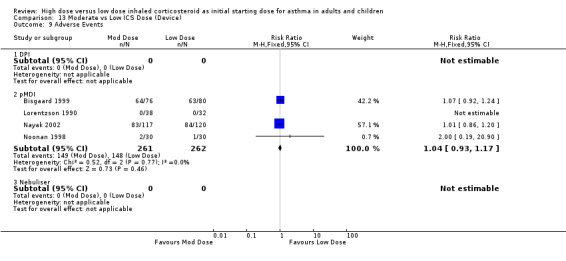
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 9 Adverse Events.
13.10. Analysis.
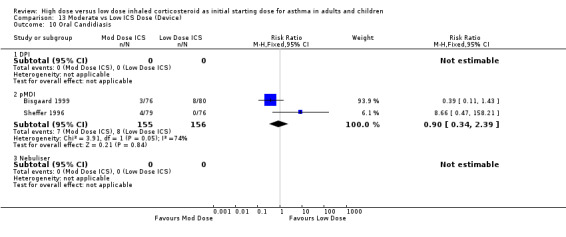
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 10 Oral Candidiasis.
13.11. Analysis.
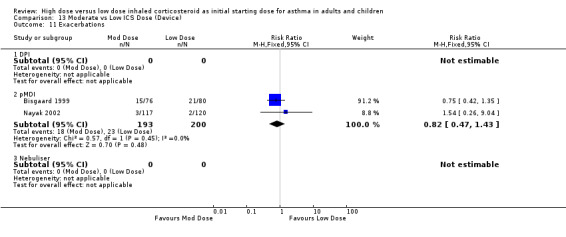
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 11 Exacerbations.
13.12. Analysis.
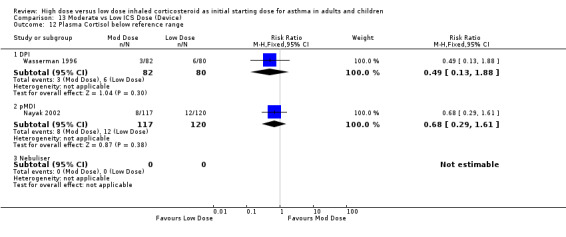
Comparison 13 Moderate vs Low ICS Dose (Device), Outcome 12 Plasma Cortisol below reference range.
Comparison 14. Step down vs constant (Duration).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 < = 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.2 > 12 weeks | 2 | 124 | Mean Difference (IV, Fixed, 95% CI) | ‐0.91 [‐26.13, 24.30] |
| 2 Morning PEF (L/min) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 < = 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.2 > 12 weeks | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | 0.83 [‐8.60, 10.26] |
| 3 Diary Card Symptom Score (Day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 < = 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 > 12 weeks | 2 | 647 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.16, 0.03] |
| 4 Diary Card Symptom Score (Night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 < = 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.2 > 12 weeks | 2 | 645 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [‐0.04, 0.15] |
| 5 Rescue Medications (puffs per day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 < = 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 > 12 weeks | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.45, 0.08] |
| 6 Rescue Medications (puffs per night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 < = 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.2 > 12 weeks | 2 | 643 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.13, 0.05] |
| 7 Withdrawal due to adverse event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 7.1 < = 12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.2 > 12 weeks | 2 | 850 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.73, 3.24] |
| 8 Proportion Achieving Asthma Control | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 < = 12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.2 > 12 weeks | 2 | 366 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.87, 1.16] |
14.1. Analysis.
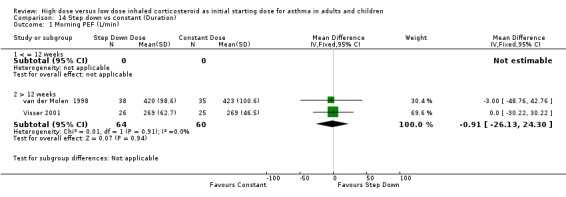
Comparison 14 Step down vs constant (Duration), Outcome 1 Morning PEF (L/min).
14.2. Analysis.
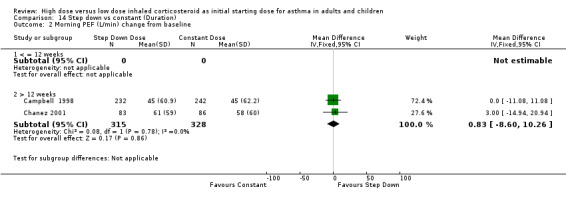
Comparison 14 Step down vs constant (Duration), Outcome 2 Morning PEF (L/min) change from baseline.
14.3. Analysis.
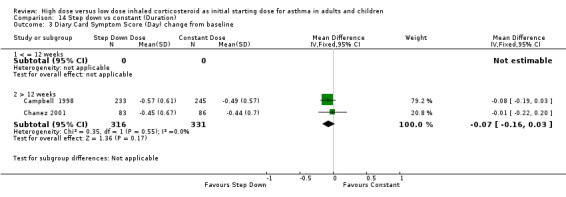
Comparison 14 Step down vs constant (Duration), Outcome 3 Diary Card Symptom Score (Day) change from baseline.
14.4. Analysis.
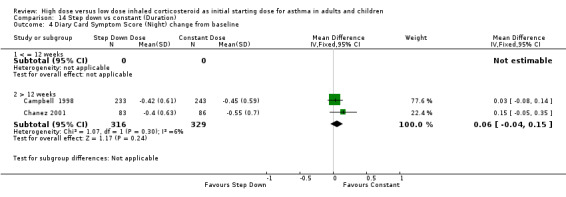
Comparison 14 Step down vs constant (Duration), Outcome 4 Diary Card Symptom Score (Night) change from baseline.
14.5. Analysis.
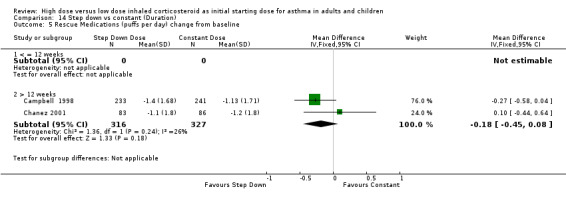
Comparison 14 Step down vs constant (Duration), Outcome 5 Rescue Medications (puffs per day) change from baseline.
14.6. Analysis.
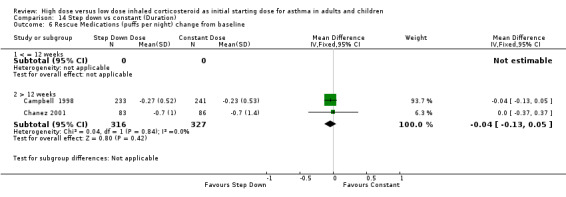
Comparison 14 Step down vs constant (Duration), Outcome 6 Rescue Medications (puffs per night) change from baseline.
14.7. Analysis.
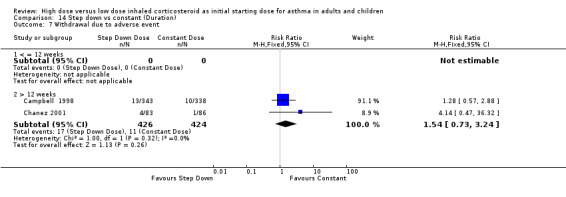
Comparison 14 Step down vs constant (Duration), Outcome 7 Withdrawal due to adverse event.
14.8. Analysis.
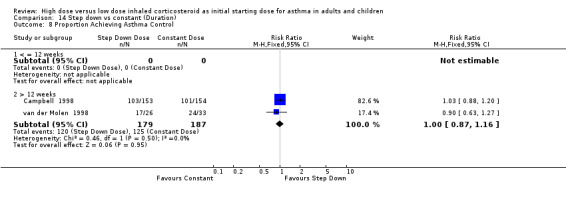
Comparison 14 Step down vs constant (Duration), Outcome 8 Proportion Achieving Asthma Control.
Comparison 15. High vs Moderate ICS Dose (Duration).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 (% predicted) | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 <= 12 weeks | 1 | 12 | Mean Difference (IV, Fixed, 95% CI) | 11.40 [0.85, 21.95] |
| 1.2 > 12 weeks | 3 | 118 | Mean Difference (IV, Fixed, 95% CI) | 3.83 [‐1.37, 9.04] |
| 2 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 < =12 weeks | 1 | 12 | Mean Difference (IV, Fixed, 95% CI) | ‐75.0 [‐228.70, 78.70] |
| 2.2 >12 weeks | 1 | 51 | Mean Difference (IV, Fixed, 95% CI) | 10.0 [‐14.39, 34.39] |
| 3 Morning PEF (L/min) change from baseline | 5 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 < =12 weeks | 3 | 320 | Mean Difference (IV, Fixed, 95% CI) | 2.93 [‐10.07, 15.93] |
| 3.2 >12 weeks | 2 | 797 | Mean Difference (IV, Fixed, 95% CI) | 7.00 [‐1.80, 15.79] |
| 4 Diary Card Symptom Score (day) change baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 < = 12 weeks | 2 | 794 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.11] |
| 4.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Diary Card Symptom Score (night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 < = 12 weeks | 2 | 800 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.06, 0.10] |
| 5.2 >12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Rescue Medications (puffs per day) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 < = 12 weeks | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.39, 0.17] |
| 6.2 >12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Rescue Medications (puffs per night) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 < = 12 weeks | 2 | 788 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.12, 0.05] |
| 7.2 >12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Withdrawal Due to Adverse Event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 < = 12 weeks | 2 | 301 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.14, 2.43] |
| 8.2 > 12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
15.1. Analysis.
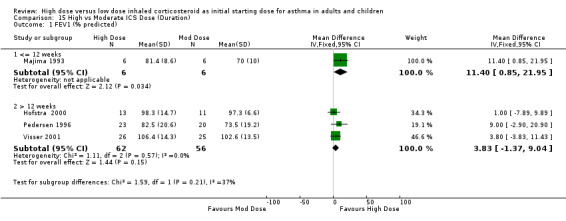
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 1 FEV1 (% predicted).
15.2. Analysis.
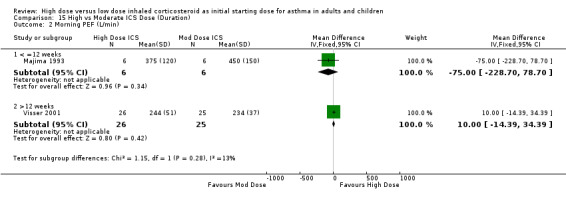
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 2 Morning PEF (L/min).
15.3. Analysis.
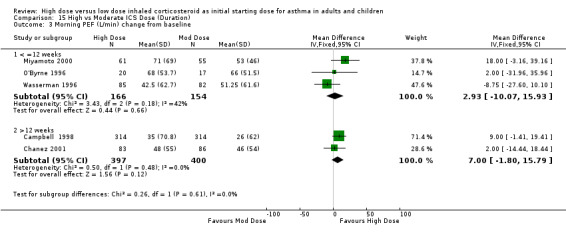
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 3 Morning PEF (L/min) change from baseline.
15.4. Analysis.
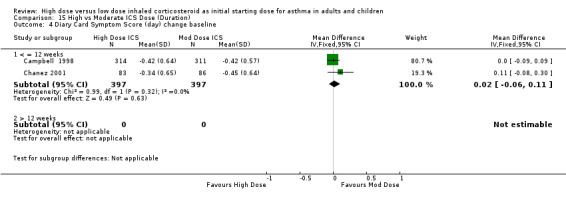
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 4 Diary Card Symptom Score (day) change baseline.
15.5. Analysis.
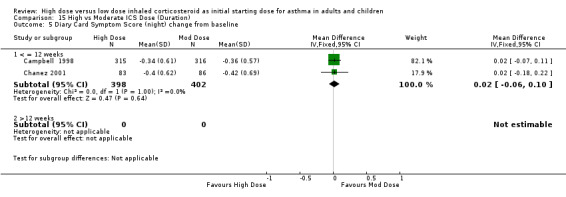
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 5 Diary Card Symptom Score (night) change from baseline.
15.6. Analysis.
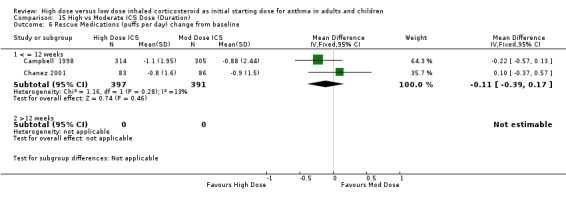
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 6 Rescue Medications (puffs per day) change from baseline.
15.7. Analysis.
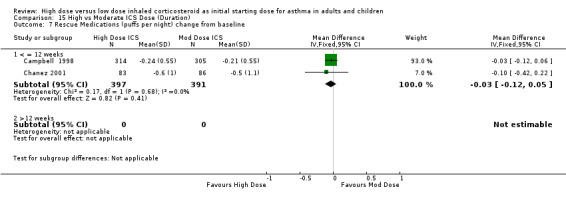
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 7 Rescue Medications (puffs per night) change from baseline.
15.8. Analysis.
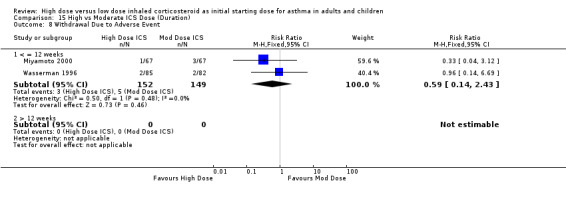
Comparison 15 High vs Moderate ICS Dose (Duration), Outcome 8 Withdrawal Due to Adverse Event.
Comparison 16. High vs Low ICS Dose (Duration).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 < = 12 weeks | 2 | 123 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.55, 0.16] |
| 1.2 > 12 weeks | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Morning PEF (L/min) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 < = 12 weeks | 2 | 95 | Mean Difference (IV, Fixed, 95% CI) | ‐18.05 [‐56.04, 19.95] |
| 2.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Morning PEF (L/min) change from baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 <= 12 weeks | 2 | 282 | Mean Difference (IV, Fixed, 95% CI) | 12.00 [‐1.50, 25.51] |
| 3.2 > 12 Weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Rescue Medications (no. of puffs) | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 < = 12 weeks | 2 | 95 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.65, 0.56] |
| 4.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Withdrawal Due to Adverse Event | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 < = 12 weeks | 2 | 295 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.14, 2.33] |
| 5.2 >12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
16.1. Analysis.
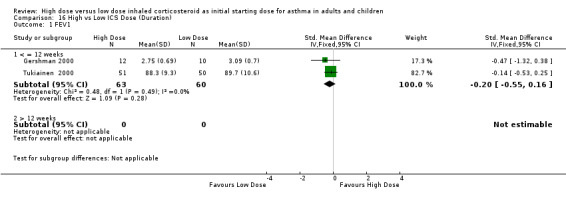
Comparison 16 High vs Low ICS Dose (Duration), Outcome 1 FEV1.
16.2. Analysis.
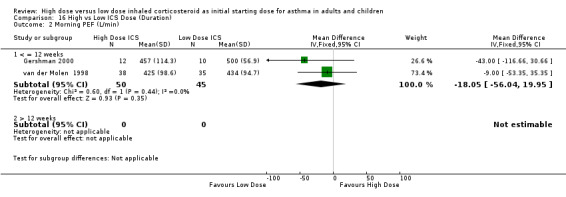
Comparison 16 High vs Low ICS Dose (Duration), Outcome 2 Morning PEF (L/min).
16.3. Analysis.
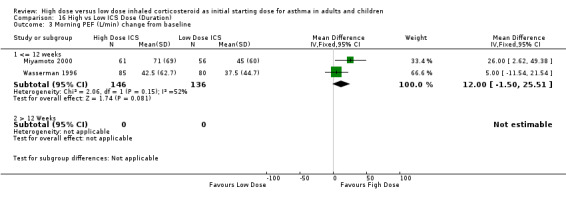
Comparison 16 High vs Low ICS Dose (Duration), Outcome 3 Morning PEF (L/min) change from baseline.
16.4. Analysis.
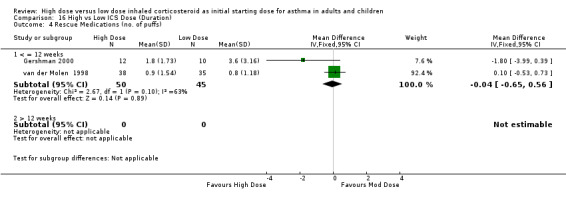
Comparison 16 High vs Low ICS Dose (Duration), Outcome 4 Rescue Medications (no. of puffs).
16.5. Analysis.
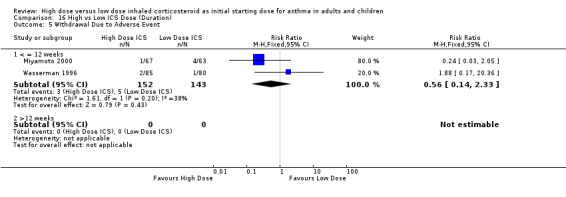
Comparison 16 High vs Low ICS Dose (Duration), Outcome 5 Withdrawal Due to Adverse Event.
Comparison 17. Moderate vs Low ICS Dose (Duration).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 FEV1 change from baseline | 5 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 < = 12 weeks | 5 | 536 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.11 [‐0.06, 0.28] |
| 1.2 > 12 weeks | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Morning PEF (L/min) change baseline | 5 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 < = 12 weeks | 5 | 411 | Mean Difference (IV, Fixed, 95% CI) | 11.14 [1.34, 20.93] |
| 2.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Evening PEF (L/min) change from baseline | 3 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.1 < = 12 weeks | 3 | 241 | Mean Difference (IV, Fixed, 95% CI) | 4.30 [‐6.16, 14.75] |
| 3.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 PEF Diurnal Variation change from baseline | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 < = 12 weeks | 2 | 126 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.28 [‐0.63, 0.07] |
| 4.2 > 12 weeks | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Symptom Score change baseline | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 < = 12 weeks | 2 | 75 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.21, 0.13] |
| 5.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Symptom (Night Waking) change from baseline | 3 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 < = 12 weeks | 3 | 285 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.29 [‐0.53, ‐0.06] |
| 6.2 > 12 weeks | 0 | 0 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Rescue Medications (n0. of puffs change baseline | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 < = 12 weeks | 4 | 467 | Mean Difference (IV, Fixed, 95% CI) | ‐0.35 [‐0.98, 0.28] |
| 7.2 > 12 weeks | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Withdrawal due to Adverse Event | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 < = 12 weeks | 3 | 523 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.41, 2.46] |
| 8.2 >12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Adverse Events | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 9.1 < = 12 weeks | 4 | 523 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.93, 1.17] |
| 9.2 > 12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10 Oral Candidiasis | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 10.1 < = 12 weeks | 2 | 311 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.34, 2.39] |
| 10.2 > 12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11 Exacerbations | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 11.1 < = 12 weeks | 2 | 393 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.47, 1.43] |
| 11.2 > 12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12 Plasma Cortisol below reference range | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 12.1 < = 12 weeks | 2 | 399 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.30, 1.27] |
| 12.2 >12 weeks | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
17.1. Analysis.
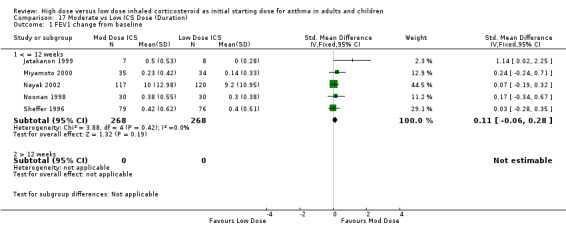
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 1 FEV1 change from baseline.
17.2. Analysis.
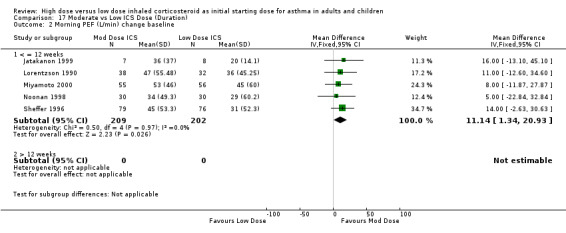
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 2 Morning PEF (L/min) change baseline.
17.3. Analysis.
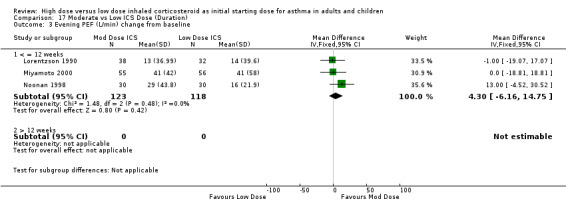
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 3 Evening PEF (L/min) change from baseline.
17.4. Analysis.
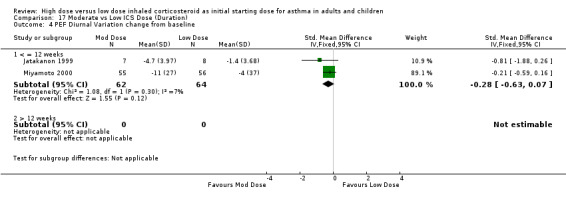
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 4 PEF Diurnal Variation change from baseline.
17.5. Analysis.
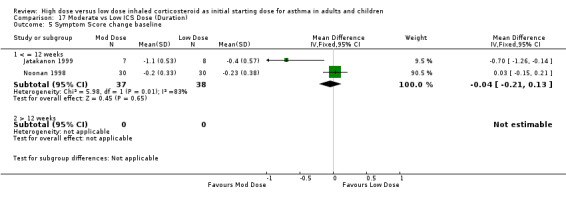
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 5 Symptom Score change baseline.
17.6. Analysis.
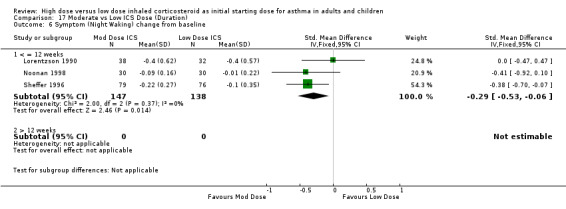
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 6 Symptom (Night Waking) change from baseline.
17.7. Analysis.
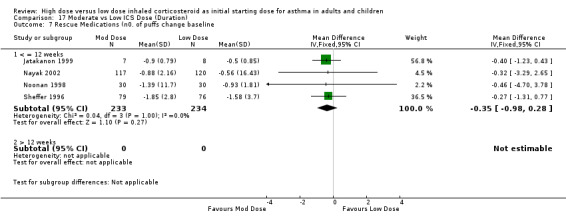
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 7 Rescue Medications (n0. of puffs change baseline.
17.8. Analysis.
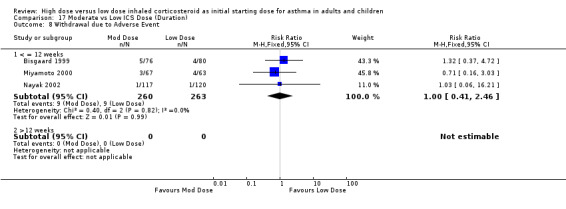
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 8 Withdrawal due to Adverse Event.
17.9. Analysis.
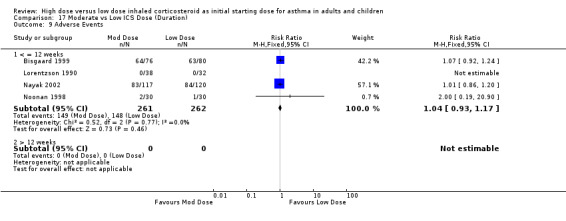
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 9 Adverse Events.
17.10. Analysis.
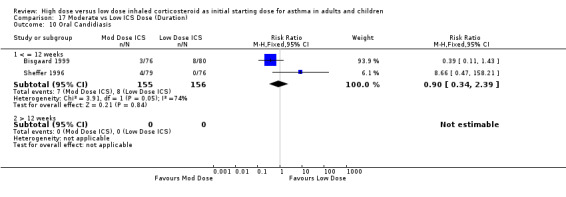
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 10 Oral Candidiasis.
17.11. Analysis.
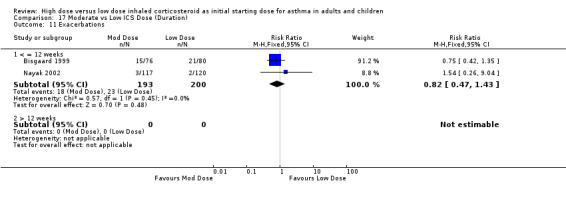
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 11 Exacerbations.
17.12. Analysis.
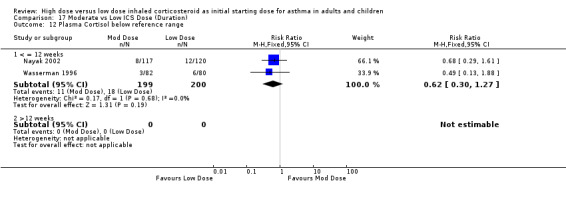
Comparison 17 Moderate vs Low ICS Dose (Duration), Outcome 12 Plasma Cortisol below reference range.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Bisgaard 1993.
| Methods | Study Design: Crossover RCT Randomisation: Random stated method not described. Concealment of Allocation: Not stated Double Blinding: No ‐ single blind. Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: Outpatients No eligible: Not stated. No randomised: 18 No completed: 14 Sex: Males 9 (64%); Females 5 (36%) Age: mean 27 months. Diagnostic criteria for asthma: Dr diagnosis Inclusion criteria: < 3 yrs age, recurrent wheeze, previous ICS but not in past month. Exclusion criteria: No OCS in past 3 months, exacerbations requiring > 4 puffs beta 2 agonist for > 4 days during study period, temp >38 degrees during study period. Baseline severity of asthma: Not stated | |
| Interventions | 1: BUD 800mcg /day
2: BUD 200mcg/day
3. Placebo
Delivery device: pMDI plus spacer
Duration of treatment: 4wks for each treatment ‐ 12 weeks total. Run in phase: not stated Wash out phase: no washout between treatments Other: rescue meds prn, no other meds allowed, no glucocorticoid ointments. |
|
| Outcomes | exacerbations, growth rate, symptoms, rescue meds | |
| Notes | No washout period between treatments | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Bisgaard 1999.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described. Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described ‐ Intention to treat Jadad Score: 3 | |
| Participants | Study site: Outpatients at 33 centres across 9 countries No eligible: 314 enrolled No randomised: 237 No completed: 212 Sex:34% female. Age: Mean 28 months; Range: 12‐47 months Diagnostic criteria for asthma: documented history of recurrent wheeze or asthma symptoms Inclusion criteria: Asthma symptoms or rescue Beta 2 use for 7 of the last 14 days run in. > 2weeks without ICS (plus 4 week run in without ICS) Exclusion criteria: ICS, systemic CS, methylxanthines or antibiotics for chest infection in the 2 weeks prior to run in. Ineffective use of the Babyhaler spacer device. Baseline severity of asthma: Moderate to severe asthma symptoms. Placebo group had fewer and milder symptoms than both treatment groups. 34% used asthma medications in the month prior to randomisation. 42% had a history of eczema. | |
| Interventions | 1: FP 200mcg/day (100mcg bd) 2: FP 100mcg/day (50mcg bd) 3: Placebo 2 puffs bd Delivery device: pMDI plus Babyhaler spacer device Duration of treatment: 12 weeks Run in phase: 4 weeks with placebo 2 puffs bd and salbutamol prn Other: salbutamol prn, continued regular medications including sodium cromoglycate, ketotifen & antihistamines providing the dose remained constant. ICS or OCS used for treatment of an exacerbation. Withdrawan if > one exacerbation. | |
| Outcomes | Symptoms, exacerbations, adverse events, control of asthma, treatment with corticosteroids, hoarseness/dysphonia, oral candidiasis. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Campbell 1998.
| Methods | Study Design: Parallel RCT Randomisation: computer generated randomisation code in block sizes of 4. Concealment of Allocation: not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: described Statistical analysis: described ‐ per protocol analysis Jadad Score: 4 | |
| Participants | Study site: 146 General Practices No eligible: 855 No randomised: 682 (343/338) ‐one subject withdrew before commencement of treatment. No completed: 454 (223/231) 18 weeks. 295 (146/149) 24 weeks Sex: Males 297 (43.6%); Females 384 (56.4%) Age: mean (SD) Intervention1: 33.5(13.8), Intervention 2: 33.3 (15.6) Diagnostic criteria for asthma: 'documented diagnosis of asthma.' Inclusion criteria: > 12 years, asthma symptoms 2 out of the past 7 days, bronchodilator prn for last 2 weeks, requiring ICS according to Dr. Exclusion criteria: PEF <60% predicted, pregnancy, breast feeding, significant concomitant disease, medication in last 3 months of beta 2 blockers, sodium cromoglycate or sodium nedocromil. Baseline severity of asthma: PEF mean (SD) ‐ Intervention1: 418(92), Intervention 2: 408 (89) | |
| Interventions | 1: Budesonide 800mcg /day (400mcg bd) for 6 weeks; Budesonide 400mg nocte for another 12 weeks; if met asthma controlled criteria budesonide 200mcg nocte for a further 6 weeks. 2: Budesonide 400mcg nocte/ placebo mane for 6 weeks; budesonide 400mcg nocte for further 12 weeks; if asthma controlled then budesonide 200mcg nocte for 6 weeks. Delivery device: DPI Duration of treatment: Initial treatment 6 weeks, 12 weeks phase 2 ‐ static dose for lntervention2 and step down for Intervention1; then for those subjects who achieved asthma control a further reduction to 200mcg for 6 weeks. Total duration 24 weeks. Run in phase: 4‐10 days Other: | |
| Outcomes | PEF, rescue medication, nocturnal asthma, asthma symptoms (diary and clinic), adverse events, compliance, asthma control at 18 weeks | |
| Notes | High dropout rate ‐ 50% non compliance. For morning PEF and day rescue medications it was assumed that the variance was SE as the measure was very small in comparison to the reported clinic PEF SD and night rescue meds SD mentioned in the text. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer generated randomisation code in block sizes of 4. |
Chanez 2001.
| Methods | Study Design: Parallel RCT Randomisation: Computer generated random list, randomised by centre in block size of four. Concealment of Allocation: Described Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described ‐ intention to treat analysis Jadad Score: 4 | |
| Participants | Study site: Multicentred ‐ outpatients No eligible: 214 enrolled No randomised: 169 (83/86) No completed: 137 (66/71) Sex: Males 84 (50%); Females 85 (50%) Age: mean 38 yrs Range: eligible 18‐70 yrs Diagnostic criteria for asthma: PEF diurnal variation > 20%, FEV1 >12% reversibility Inclusion criteria: uncontrolled asthma defined as daily nocturnal symptoms, wheezing and beta agonist use in last 7 days, not currently on ICS (includes beclomethasone up to 500mcg/day), use of long acting theophylline in past 2 weeks. Exclusion criteria: FEV1 <55 % predicted, use of nasal steroids or oral and parenteral steroids in past month. Use of leukotriene antagonists in past, exacerbation or airway infection during past month. Baseline severity of asthma: FEV1: mean 74% predicted. All subjects uncontrolled asthma at enrolment, allergy 83%, previous ICS use 50%. | |
| Interventions | 1: Budesonide 1600mcg/day (800mcg bid) reducing by half every 4 weeks if controlled to min dose of BUD 200mcg/day (100mcg bid).
2: BUD 400mcg/day (200mcg bid)
Delivery device: DPI
Duration of treatment: 16 weeks Run in phase: 2 weeks ‐ beta agonist as needed or long acting theophylline Other: No nebulised or parenteral beta agonist allowed during study. Withdrawn if not controlled i.e. an exacerbation |
|
| Outcomes | PEF, FEV1, rescue medication, symptoms, exacerbations, serum eosinophils, reversibility % | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer generated random list, randomised by centre in block size of four. |
Gershman 2000.
| Methods | Study Design: Parallel RCT Randomisation: stated, method not described Concealment of Allocation: numbered identical containers administered sequentially Double Blinding: Yes Withdrawals / dropouts: described Adverse events: not described Statistical analysis: described Jadad Score: 4 | |
| Participants | Study site: recruited by advertising in local newspapers and database of subjects from previous studies‐ site not stated No eligible: not stated No randomised: 24 (12/12) No completed: 22 (12/10) Sex: Males = 21 (87.5 %) Age: mean 38.7; range 22‐55 Diagnostic criteria for asthma: Objective lung function. Inclusion criteria: FEV1 <=80% predicted, PC20 (methacholine), FEF25‐75 <75% predicted Exclusion criteria: history of ICS or OCS use, URTI in 6 weeks prior to study, tobacco use in past year or total smoking history > 10 pack years Baseline severity of asthma: Intervention 1= FEV1 69% predicted (mean). Intervention 2 = FEV1 66% predicted (mean). | |
| Interventions | 1: FP 1000mcg/day (2x250mcg twice daily) 2: FP 100mcg / day (1x50mcg & 1x placebo twice daily) Delivery device: pMDI plus spacer Duration of treatment: 6 weeks Run in phase: 2 weeks Other: Salbutamol prn allowed throughout study, inhaler technique assessed at start. 3 weeks placebo run out. | |
| Outcomes | Lung function, airway hyperresponsiveness, rescue medication use, asthma symptoms, airway inflammation parameters | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Hampel 2000.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described. Concealment of Allocation: Not stated Double Blinding: Study was 'blinded'‐double blinded not stated Withdrawals / dropouts: described Adverse events: Described Statistical analysis: Descibed ‐ Intention to treat analysis Jadad Score:2 | |
| Participants | Study site: Multi‐centred study
No eligible: 270 eligible for randomisation
No randomised: 270 (92/91/87)
No completed: 253 (87/87/79)
Sex: Females: 153 (57%)
Age: Mean(SD) Intervention 1: 35.8(12.1); Intervention 2: 52 (57.1); Intervention 3: 45 (51.7)
Diagnostic criteria for asthma: Mild to mod asthma according to GINA guidelines, objective lung function
Inclusion criteria: Steroid naive(no ICS in past 3 months), mild‐to‐mod asthmatics, Beta 2 use only, >= 15% reversibility after Beta 2, non smoker, proper MDI inhaler technique. Exclusion criteria: Cardiac disease, pulmonary disease other than asthma, known hypersensitivity to BDP, history of alcohol or substance abuse, no other medications allowed except Beta 2 during study. Baseline severity of asthma: FEV1 mean(SD) percent predicted. Intervention 1: 75.5 (7.4); Intervention 2: 75.3 (8.6); Intervention 3: 77.1 (5.8) |
|
| Interventions | 1: Beclomethasone (BDP) 200mcg/day (100mcg bd)
2: BDP 100mcg/day (50mcg bd) Delivery device: pMDI Duration of treatment: 6 weeks Run in phase: 14 days. eligible if 65‐85% predicted normal FEV1 during this time Other: |
|
| Outcomes | FEV1, PEF, FEF25‐75, nocturnal asthma, symptoms, rescue meds, adverse events, withdrawals due to adverse events. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Hofstra 2000.
| Methods | Study Design: Parallel RCT Randomisation: Computer. Concealment of Allocation: Coded identical containers Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described Jadad Score: 5 | |
| Participants | Study site: Multicentre ‐ hospital No eligible: 39 No randomised: 37 (14/11/12) No completed: 36 (14/11/11) Sex:Male 23 (62%); Female 14(38%) Age: mean 10.3 years; Range 6‐14 yrs Diagnostic criteria for asthma: 'clinically diagnosed asthma' Inclusion criteria: intranasal and dermatological ICS allowed, and opthalmological cromones, provided at constant dose throughout study. Exclusion criteria: No ICS in past 4 months, no viral infection previous 2 weeks, no hospital admission or OCS in 4 weeks prior to screening. Baseline severity of asthma: FEV1 > 70% predicted, > 20% fall in FEV1 after exercise test, clinically stable | |
| Interventions | 1: Initially FP 500mcg/day (250mcg bd) for 24 weeks 2: FP 200mcg/day (100mcg bd) for 24 weeks 3: Placebo for 6 weeks then randomised to FP 500mcg/day or 200mcg/day groups for 18 weeks Delivery device: pMDI with volumatic spacer. Duration of treatment: 24 weeks Run in phase: 2 weeks, salbutamol prn Other: | |
| Outcomes | Exercise challenge, AHR, lung function, rescue medication, symptoms, airway inflammation, adverse events | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer generated random numbers |
Jatakanon 1999.
| Methods | Study Design: Parallel RCT Randomisation: random stated, method not described. Concealment of Allocation: Not stated. Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Not described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: Not stated No eligible: Not stated No randomised: 22 (8/8/6) No completed: 21 (7/8/6) Sex: All males (100%) Age: mean(SE) Intervention 1: 29 (2.4); Intervention 2:31 (1.2); Intervention 3: 31 (2.8) Range: Diagnostic criteria for asthma: Dr diagnosis, objective lung function ‐ PC20(methacholine) Inclusion criteria: stable asthma with no history ICS use, Beta2 agonist therapy only, positive allergy, no exacerbation in past 3 months Exclusion criteria: URTI in past 6 weeks, nasal steroids past 2 months Baseline severity of asthma: FEV1 >=80% predicted, Mean (se) Intervention 1: 91.5(4.2); Intervention 2: 92.3 (3.1);Intervention 3:97.2 (4.0). PEF Intervention 1:501 (19); Intervention 2: 512 (20); Intervention 3:552 (32) | |
| Interventions | 1: Budesonide 400mcg/day 2: Budesonide 100mcg/day 3: Placebo Delivery device: DPI ‐ Turbohaler Duration of treatment: 4 weeks Run in phase: 1 week Other: Also concurrently ran a 1600mcg /day vs placebo crossover trial | |
| Outcomes | PEF, FEV1, AHR, rescue medication, symptoms, sputum eosinophils, exhaled NO | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Jonasson 1998.
| Methods | Study Design: Parallel RCT Randomisation: Randomised in balanced blocks of four Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: outpatient clinics No eligible: 166 enrolled No randomised: 163 (42/40/41/40) No completed: 160 (42/40/38/40) Sex: Males 107 (66%); Females 56 (34%) Age: mean 9.9 yrs, range: 7‐16 Diagnostic criteria for asthma: defined by the definition in the International Consensus Report Inclusion criteria: 3 previous obstructive episodes or one episode with atopy, 1 episode in the past year. No previous ICS Exclusion criteria: No ICS in past 2 months, No cromglycate/nedocromil in past 4 weeks, no respiratory tract infection in past 4 weeks Baseline severity of asthma: FEV1 mean 103% predicted. Atopy 67.5% | |
| Interventions | 1: BUD 200mcg daily
2: BUD 100mcg bid (200/day)
3: BUD 100mcg daily
4:Placebo
Delivery device: DPI
Duration of treatment: 12 weeks initial and then continued for a further 24 months Run in phase: 2 weeks Other: Terbutaline as needed |
|
| Outcomes | PEF, FEV1, airway hyperresponsiveness, symptoms, exercise induced fall in FEV1, serum ECP/eosinophils, adherence, compliance | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomised in balanced blocks of four |
Lorentzson 1990.
| Methods | Study Design: Parallel RCT Randomisation: random stated, method not described Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: described Adverse events: Described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: multicentre No eligible: No randomised: 104 No completed: 103 (38/32/33) Sex: Males 58 (56%); Females 45 (44%) Age: mean 32 Diagnostic criteria for asthma: Not stated Inclusion criteria: >= 35 puffs beta agonist during run in, mean PEF variation 10‐30% over last 4 days of run in, mean PEF 75% predicted. Exclusion criteria: ICS or OCS use, use of spacer for beta agonist, concomitant disease, respiratory infection or acute asthma exacerbation in past month Baseline severity of asthma: mean duration of asthma 11 yrs. Mean(SEM) morning PEF (L/min): Intervention 1:429(16); intervention 2: 398 (16); Intervention 3: 397(16) | |
| Interventions | 1: BUD 400mcg/day (200mcg bd)
2: BUD 200mcg/day (100mcg bd)
3:Placebo
Delivery device: pMDI
Duration of treatment: 6 weeks Run in phase: 1 week, Beta agonist only Wash out phase: 1 week prior to run in for subjects who were on oral beta agonists Other: |
|
| Outcomes | PEF, rescue medications, symptoms, adverse events | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Majima 1993.
| Methods | Study Design: Parallel RCT Randomisation: random stated, method not described Concealment of Allocation: "envelope" method Double Blinding: No Withdrawals / dropouts: not described Adverse events: Not described. Statistical analysis: Described Jadad Score: 1 | |
| Participants | Study site: Respiratory outpatients clinic, university hospital No eligible: Not stated No randomised: 17 No completed: Not stated Sex: Males 11 (65%); Females 6 (35%) Age: mean Intervention 1: 45.3; Intervention 2: 43.3; Intervention 3: 48.6 Diagnostic criteria for asthma: Objective lung function according to Japanese Allergy Association Inclusion criteria: Progressive airway hypersensitivity and reversibility of bronchoconstriction identified by astrograph:Not on anti‐ allergy or steroid medication for more than 2 months prior to study Exclusion criteria: Baseline severity of asthma: mild to moderate asthmatics | |
| Interventions | 1: BDP 800mcg/day
2: BDP 400mcg/day
3: Salbutamol 800mcg/day Delivery device: Not stated Duration of treatment: 8 weeks Run in phase: not stated Other: |
|
| Outcomes | FEV1, PEF, airway sensitivity and reactivity, flow volume curve | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Miyamoto 2000.
| Methods | Study Design: Parallel RCT Randomisation: Computer generated sequence in blocks of four. Concealment of Allocation: sequentially administered coded containers. Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described Jadad Score: 5 | |
| Participants | Study site: multicentre trial in 62 medical centres No eligible: Not stated No randomised: 267 (67/67/63/70) No completed: 224 (61/55/56/52) Sex: Male 126/238 (53%), Female 112/238 (47%) Age: mean (SD) Intervention 1: 50.4(15); Intervention 2: 47.8(15.9); Intervention 3: 50.9 (15.5); Intervention 4: 50.6 (14.2) Diagnostic criteria for asthma: Not stated Inclusion criteria: PEF 50‐80% predicted, stable perennial symptoms of asthma > 3 days/week during run in Exclusion criteria: Glucocorticoid use within 1 month of study, respiratory tract infection in previous 4 weeks, other dominant respiratory disease, cardiovascular disease, hepatic or renal disease. Baseline severity of asthma: mild asthma 147/238 (62%); Mod asthma 88/238 (37%); Severe asthma 3/238 (1%) | |
| Interventions | 1:BUD 800mcg/day (400mcg bd) 2: BUD 400mcg/day (200mcg bd) 3: BUD 200mcg/day (100mcg bd) 4: Placebo Delivery device: DPI Duration of treatment: 6 weeks Run in phase: 2 weeks Other: oropharyngeal rinsing after each dose to minimise oral candidiasis | |
| Outcomes | PEF, FEV1, symptoms, safety, Doctor efficacy assessment, adverse events, hospitalisations, ER visits, lost production, symptoms, costs | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer generated sequence in blocks of four. |
Nayak 2002.
| Methods | Study Design: Parallel RCT Randomisation:Computer produced randomization schedule. Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described. Adverse events: Described. Statistical analysis: Intention to treat analysis Jadad Score: 4 | |
| Participants | Study site: 50 outpatient centres. No eligible: 684 screened No randomised: 353 (117/120/116) No completed: 310 (108/105/97) Sex: Males 224 (63%) Age: mean (SD) Intervention 1:8.9 (1.9); Intervention 2: 9.4 (2.0); Intervention 3: 9.3 (2.1) Diagnostic criteria for asthma: Objective lung function Inclusion criteria: Stable moderate symptomatic asthma > 6 months duration and on Beta agonists, FEV1 50‐80 % predicted,, >= 12 % reversibility, use of beta agonist on 50% of days in 2 week run in. Exclusion criteria: significant nonreversible pulmonary disease other than asthma, evidence of any clinically significant immnuologic, neoplastic, endocrine, hematologic, cardiac, hepatic, renal, GI, neurologic, or psychiatric abnormalities or illness. Upper or lower respiratory tract infection last 2‐4 weeks, use of OCS, ICS or injectable corticosteroids within 8 wks, 6 wks or 6 months respectively. Baseline severity of asthma: FEV1 (L): mean(SD) Intervention 1: 1.5 (0.4); Intervention 2: 1.6 (0.4; Intervention 3: 1.6(0.5) PEF L/min: mean(SD): Intervention 1: 217(65.4); Intervention 2: 232.5(62.9); Intervention 3: 235.4 (70.0). | |
| Interventions | 1: BDP ‐HFA 160mcg/day (80mcg bd)
2: BDP‐HFA 80mcg/day (40mcg bd)
Delivery device: pMDI
Duration of treatment: 12 weeks Run in phase: 2 weeks Other:No other asthma maintenance therapies were permitted |
|
| Outcomes | exacerbations, adverse events, clinical improvement, PEF, symptoms, rescue medications, plasma cortisol | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer produced randomization schedule |
Noonan 1998.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described Concealment of Allocation: Not stated Double Blinding: yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: multicentre No eligible: Not stated No randomised: 138 (33/34/35/36) No completed: 119 (30/30/31/28) Sex: Males: 84 (61%); Females: 54 (39%). Age: mean 23;29;30;26 Range: 12‐59 Diagnostic criteria for asthma: ATS Inclusion criteria: >= 12 yrs old, 6 month history asthma, documented history of allergy, FEV1 between 60 and 85% predicted, >= 15% reversability, positive PD20(methacholine) . Exclusion criteria: recent hospitalisation for asthma, corticosteroid use, oral theophylline, inhaled cromolyn, nedocromil,, smokers, significant concurrent disease,, pregnancy or lactation. Baseline severity of asthma: FEV1: 74;73;76;74 percent predicted | |
| Interventions | 1: FP (10% lecithin) 200mcg/day
2: FP (1% Lecithin) 200mcg/day
3: FP (1% Lecithin) 100mcg/day
4: Placebo (1% Lecithin) 2 puffs day
Delivery device: pMDI
Duration of treatment: 8 weeks
Run in phase: 2 weeks Other: Albuterol and oral terfanadine as needed |
|
| Outcomes | FEV1, PEF, AHR, rescue medications, nocturnal asthma, symptoms | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
O'Byrne 1996.
| Methods | Study Design: Parallel RCT
Randomisation: Computer generated code‐ stratified by allergy status and randomised in blocks of 3 at each centre
Concealment of Allocation: coded identical containers
Double Blinding: Yes Withdrawals / dropouts: Described. Adverse events: Described. Statistical analysis: Intention to treat Jadad Score: 4 |
|
| Participants | Study site: multicentre trial in 7 cities, at primary care physicians No eligible: Not stated No randomised: 57 (20/17/20) No completed: 39 (12/13/14) Sex: Male 25 Female 32 Age: mean 37/32/36 Diagnostic criteria for asthma: Objective lung function:> 10% PEF diurnal variation at least 3 of the last 7 days. Inclusion criteria: > 18 years, mild asthma and on clinical evaluation not requiring ICS. Exclusion criteria: No ICS in past 3 months, Requiring ICS Baseline severity of asthma: PEF mean (SD): Intervention 1: 364 (115); Intervention 2: 381 (112); Intervention 3 : 403 (116) | |
| Interventions | 1: BUD 800mcg/day (400mcg bid) 2: BUD 400mcg /day (200mcg bid) 3: Placebo Delivery device: nebuhaler Duration of treatment: 16 weeks Run in phase: Other: Beta agonists as needed, theophylline if remained constant through out study, allowed budesonide and/or prednisone for exacerbation during study. | |
| Outcomes | costs, hospitalisations, ER visits, PEF, symptoms, exacerbations, adverse events | |
| Notes | For morning PEF it was assumed the variance estimate was SE. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer generated code‐ stratified by allergy status and randomised in blocks of 3 at each centre |
Pedersen 1995.
| Methods | Study Design: Crossover RCT Randomisation:Random stated, method not described. Concealment of Allocation: Identical drug canisters administered in randomized order Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Not described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: multicentred No eligible: Not stated No randomised: 19 No completed: 18 Sex: Males:17(89%); Females 2(11%). Age: mean 11yrs Diagnostic criteria for asthma: Objective lung function Inclusion criteria: beta 2 use only, Symptoms> 3 days/week, >=20% bronchial reversibility post terbutaline or >= 20% fall in FEV1 post exercise test. Exclusion criteria: Bronchopulmonary disease other than asthma, oral prednisolone within 1 month before study Baseline severity of asthma: PC20 range: 22.5%‐76.5%; bronchial reversibility range: 26.5% ‐ 82.4%. mean duration of asthma: 7.5 yrs | |
| Interventions | 1: BUD 100mcg/day (50mcg bd)
2: BUD 200mcg/day (100mcg bd)
3: BUD 400mcg/day (200mcg bd) Delivery device: metered‐dose inhaler with spacer Duration of treatment: 4 weeks for each treatment period, no washout. Total treatment 12 weeks. Run in phase: 2 weeks Wash out phase: none Other: > 80% compliance |
|
| Outcomes | PEF & FEV1 post exercise testing, AHR (exercise), rescue medication, symptoms, urinary cortisol | |
| Notes | no washout period between dose changes | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Pedersen 1996.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described. Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Not fully described Adverse events: Not described Statistical analysis: Described Jadad Score: 2 | |
| Participants | Study site: Not stated No eligible: Not stated No randomised: ? 85(29;29;27) No completed: 53 (23;20;10) Sex: Male 48 (55%); Female 37 (45%) Age: mean(SD) Intervention 1: 46.1(11.2); Intervenmtion 2: 46.8 (12.5) Diagnostic criteria for asthma: ATS criteria Inclusion criteria: Beta 2 use only, in need of regular maintenance treatment due to attacks of dyspnoea, cough, wheezing and airflow variability Exclusion criteria: Not stated Baseline severity of asthma: FEV1 % predicted mean(SD): Intervention 1: 70.7%(14.2); Intervention 2: 68.8 % (19.8).Allergic: Intervention 1: 40%; Intervention 2 : 31% | |
| Interventions | 1: BUD 1600mcg/day 2: BUD 400mcg/day 3: Theophylline600mg/day Delivery device: Not stated. Duration of treatment: 9 months Run in phase: Not stated Other: subjects followed up for 3 months post intervention | |
| Outcomes | FEV1, rescue meds, PC20, Vital Capacity, Blood eosinophils, serum EPX, ECP, MPO, lactoferrin | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Pirozynski 1996.
| Methods | Study Design: Parallel RCT Randomisation: random stated, method not described Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Not described Statistical analysis: Not described Jadad Score: 3 | |
| Participants | Study site: Not stated
No eligible: Not stated
No randomised: 262
No completed: 23 withdrew due to exacerbation or adverse event, ? number completed.
Sex: Females: 143 (55%); Males: 119 (45%)
Age: Mean 36yrs
Diagnostic criteria for asthma: Not stated
Inclusion criteria: Not stated Exclusion criteria: Not stated Baseline severity of asthma: mild‐to‐moderate asthma. Mean FEV1 % predicted: 82.3%. Mean PEF: 380 L/min |
|
| Interventions | 1: BUD 800mcg/day (400mcg bd) for 4 weeks then reduced to 200mcg/day (100mcg bd) for a further 8 weeks
2: BUD 200mcg/day (100mcg bd) for 12 weeks Delivery device: DPI Duration of treatment: 12 weeks Run in phase: 2 weeks Other: |
|
| Outcomes | Withdrawal due to exacerbations or adverse events, PEF, FEV1, rescue medications, symptoms | |
| Notes | Abstract only | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Sheffer 1996.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis:Described Jadad Score: 3 | |
| Participants | Study site: Multicentred No eligible: Not stated. No randomised: 307 (79/79/76/73) No completed: 294(52/48/47/29) Sex: Male 185 (60%); Female 122(40%). Age: Mean (range) Intervention 1:28(12‐72); Intervention 2: 30 (12‐63); Intervention 3:30 (12‐69); Intervention 4 :30 (13‐54) Diagnostic criteria for asthma:Objective lung function Inclusion criteria: > 12 yrs, history of asthma requiring daily pharmacotherapy for at least 3 months, FEV1 45‐75% predicted, >= FEV1 reversibility. Exclusion criteria: Pregnancy or lactating women, patients who had taken long term OCS, Steroids in any form in past month, cromolyn sodium in month prior to study, history of life threatening asthma. Baseline severity of asthma: FEV1 mean (SEM): Intervention 1: 2.45 (0.06); Intervention 2: 2.38 (0.07); Intervention 3: 2.43 (0.06); Intervention 4: 2.36 (0.06) | |
| Interventions | 1: FP 200mcg bid (100mcg bid) 2: FP 100mcg/day (50mcg bid) 3:FP 50mcg/day (25mcg bid) 4: placebo 2 puffs bid Delivery device: pMDI Duration of treatment: 12 weeks Run in phase: 1 week placebo Other: albuterol as needed, no other medications allowed | |
| Outcomes | PEF, FEV1, rescue medication, asthma symptoms including night symptoms, adverse events, withdrawal due to worsening asthma | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Szefler 2002.
| Methods | Study Design: Parallel design of two ICS ‐ both increasing doses. Randomisation: Random stated, method not described. Concealment of Allocation: Not stated. Double Blinding: No ‐ open label Withdrawals / dropouts: Described Adverse events: Not stated Statistical analysis: Described Jadad Score: 2 | |
| Participants | Study site: Multicentred No eligible: Not stated No randomised: 30 (FP: 15; BDP :15) No completed: 26 Sex: Males 23 (77%) Age: mean (SD) FP: 29.6 (7.2); BDP: 30.3 (7.6) Diagnostic criteria for asthma: Objective lung function. Inclusion criteria: FEV1 55‐85% predicted, beta agonist response >=12%, FEV1 improvement of 200ml post beta agonist, PC20(methacholine) <=8mg/ml, AHR(exercise)>=12%, plasma cortisol >5mcg/dL, smoking history < 10 pack years . Exclusion criteria: Corticosteroids in past 6 months Baseline severity of asthma: FEV1 % predicted FP: 75.07(11.2); BDP: 73.3 (11.1) | |
| Interventions | 1: FP in increasing doses for 6 weeks each ‐ 88mcg/day; 352mcg/day;704mcg/day 2: BDP increasing doses for 6 weeks each: 168mcg/day;672mcg/day;1344mcg/day Delivery device: pMDI plus spacer Duration of treatment: 24 weeks total (3wks run in, 3x6wks treatment, 3wks washout) Run in phase: 3 weeks with placebo Other: 3 weeks with FP 2mg/day at completion of treatments. no washout in between increasing ICS doses | |
| Outcomes | FEV1, AHR (methacholine and exercise), rescue medications, symptoms, plasma cortisol, sputum eosinophils, sputum ECP, sputum neutrophils, eNO | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Tukiainen 2000.
| Methods | Study Design: Parallel RCT Randomisation: Computer randomisation in balanced blocks of four. Concealment of Allocation: computer randomisation after run in Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Nil Statistical analysis: APT (all patients treated) approach used for analysis. Jadad Score:4 | |
| Participants | Study site: Not stated No eligible: 105 No randomised: 101 (51/50) No completed: 97 (49/48). Sex:Males 36 (36%), Females 65 (64%) Age: mean (range): Intervention 1: 40.5 (19‐62); Intervention 2: 37.3 (18‐68) Diagnostic criteria for asthma: ATS Inclusion criteria: positive histamine challenge (PC20 FEV1 <= 8mgl), newly diagnosed asthma (in past 3 months), symptoms < 6 months Exclusion criteria: No ICS or previous regular pharmacological treatment for asthma Baseline severity of asthma: FEV1 mean (SEM) Intervention 1 : 3.05 (.1); Intervention 2: 3.29 (.1) PEF mean (SEM) intervention 1: 432 (12); intervention 2: 456 (10) | |
| Interventions | 1:Budesonide 800mcg/day (400mcg bid)
2: Budesonide 200mcg/day (100mcg bid)
Delivery device: DPI
Duration of treatment: 12 weeks
Run in phase: 1 week Other: Terbutaline via DPI prn throughout run in and study period. No other regular asthma meds allowed |
|
| Outcomes | PEF, FEV1, AHR, rescue medications, asthma symptoms (day & night), airway inflammation. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer randomisation in balanced blocks of four. |
van der Molen 1998.
| Methods | Study Design: Parallel RCT Randomisation: random stated, method not described Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described Jadad Score: 3 | |
| Participants | Study site: 25 General Practices No eligible: unknown, 91 selected. No randomised: 84 (44/40) No completed: 73 (38/35) Sex: Males = 37 (44%); Females = 47 (56%) Age: Intervention 1: mean (SD) 31.3 (10.8); Intervention 2: mean (SD) 32.0 (8.1) Diagnostic criteria for asthma: definition of the Dutch College for General Practitioners ‐ objective lung function. Inclusion criteria: PEF & FEV1 > 50% predicted, > 3 doses bronchodilator per week in month prior to trial. Exclusion criteria: Smoking history > 20 pack years, steroid treatment, exacerbation in previous 2 months Baseline severity of asthma: FEV1 L mean (SD): Intervention 1: 3.09(0.7); Intervention 2: 3.16 (0.79) PEF L/min mean (SD): Intervention1: 427 (89); Intervention 2: 437(99) | |
| Interventions | 1: Budesonide 800mcg /day (400x2) for 4 weeks then BUD 200mcg daily for 8 weeks
2: BUD 200mcg/day (100x2) for 4 weeks then BUD 200mcg daily for 8 weeks.
Delivery device: DPI
Duration of treatment: 4 weeks initial treatment followed by 8 weeks static treatment ‐ 12 weeks total treatment.
Run in phase: 1 week on bronchodilator therapy only Other: |
|
| Outcomes | Lung function, night waking, asthma symptoms, time to reach asthma control, adverse events,compliance. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Visser 2001.
| Methods | Study Design: Parallel RCT Randomisation: Computer randomisation. Concealment of Allocation: Coded identical containers. Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described. Statistical analysis: Described Jadad Score: 5 | |
| Participants | Study site: Multicentred No eligible: 55 No randomised: 55 (27/28) No completed: 51 (26/25) Sex: Males 29 (53%); Females 26 (47%) Age: mean (sem) Intervention 1: 8.1 (0.3), Intervention 2: 7.8 (0.2) Diagnostic criteria for asthma: Not stated Inclusion criteria: Chronic persistent asthma, age 6‐10 yrs, PD20 (methacholine ) <80mcg during washout Exclusion criteria: major illness, systemic corticosteroids during past 6 weeks, respiratory tract infection in past 4 weeks. Baseline severity of asthma: FEV1 %predicted mean (sem): Intervention 1: 92.1 (2.8); Intervention 2: 89.4(2.7). Pd20 (methacholine) geometric mean (range): Intervention 1: 36 (25‐53); Intervention 2: 30 (22‐43) | |
| Interventions | 1: FP 1000mcg/day for 2 months then step down every 2months to 500mcg /day for 2 months, 200mcg/day for 2months, then 100mcg/day for 6months.
2: FP 200mcg/day constant
Delivery device: Not stated
Duration of treatment: 12 months total Run in phase: 6 weeks on FP 200mcg/day Wash out phase: 2‐8 weeks inhaled salbutamol only prior to randomisation Other: |
|
| Outcomes | PEF, FEV1, airway hyperresponsiveness, symptoms, exacerbations, exhaled NO, adherence | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer randomisation |
Volovitz 1998.
| Methods | Study Design: Parallel RCT Randomisation: Randomised in blocks of eight Concealment of Allocation: patients given numbered identical looking drug packets containing four repsules each. Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: described Statistical analysis: Described Jadad Score: 5 | |
| Participants | Study site: Outpatient asthma clinics No eligible: 42 No randomised: 42 (21/21) No completed: 41 (42 used for analysis) Sex: Males 28(67%) Females 14 (33%) Age: mean (SD) Intervention 1: 21.5 (9.4) months; Intervention 2: 17.05 (8.9) months Diagnostic criteria for asthma: Symptoms over past 3 months Inclusion criteria: 3 wheezing episodes and asthma symptomatology > 40% of days in the past 3 months. Total symptom score of 21 during 3 week run in . Exclusion criteria: ICS, sodium cromoglycate, or ketotife in past 4 weeks. OCS use > 5 days, acute viral infection, pneumonia, chronic pulmonary disease or Tb. Family history of prematurity. Baseline severity of asthma: Percentage of time symptoms last 3 months: Intervention 1: 65%; Intervention 2:70% Exacerbations in past 3 months: Intervention 1: 6.8(8.2); Intervention 2: 5.5(4.6) | |
| Interventions | 1:Budesonide 2mg (1mg bd) decreasing 25% every 2nd day for one week then maintained on BUD 0.5mg(0.25 bd) for 9 weeks
2: Budesonide 0.5mg (0.25 bd)
Delivery device: nebuliser
Duration of treatment: 1 week initial treatment for intervention 1, 10 weeks total Run in phase: 3 weeks using previous anti asthma medication Other: Terbutaline prn, budesonide suspension (0.5mg qid) with 25% decrease every 4 days for exacerbations allowed during study |
|
| Outcomes | Symptoms, plasma cortisol, Time to clinical response | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Randomised in blocks of eight |
Wasserman 1996.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described. Concealment of Allocation: Not stated Double Blinding: Yes Withdrawals / dropouts: Described Adverse events: Described Statistical analysis: Described ‐ intention to treat analysis for safety variables, efficacy analysis on efficacy population. Jadad Score: 3 | |
| Participants | Study site: 20 sites
No eligible:
No randomised: 331 (85/82/80/84)
No completed: 256 (72/65/61/58)
Sex: Males % (71/60/63/71)
Age: Mean 29/27/29/29
Diagnostic criteria for asthma: ATS
Inclusion criteria: > 12 years, FEV1 50‐80% predicted, >= 15% reversibility oral or inhaled bronchodilators in 2 weeks prior to screening, negative pregnancy test. Exclusion criteria: Smokers, oral, intranasal, opthalmological, topical or inhaled corticosteroid within 1 month of screening or 2months for OCS Baseline severity of asthma: Mean FEV1: 2.66/2.57/2.68/2.6. Mean morning PEF (L/min): 436/418/434/432 |
|
| Interventions | 1: FP 500mcg/day (250mcg bd) 2: FP 200mcg/day (100mcg bd) 3: FP 100mcg/day (50mcg bd) 4: Placebo Delivery device: DPI Duration of treatment: 12 weeks Run in phase: 2 weeks. albuterol prn, no other asthma medications allowed. Had to meet criteria for stable asthma ‐ <= 12 puffs Beta2/day, <= 4 mornings with > 20% PEF variability, <= 2 nights with nocturnal asthma, FEV1 50‐80% predicted, reproducible lung function and adequate compliance. Other: regularly scheduled terfenadine for allergic rhinitis allowed if dose remained constant throughout study, cromolyn sodium & oxymetazoline allowed prn. Participants withdrawn due to lack of efficacy if: exacerbation requiring emergency treatment, hospitalisation, asthma meds needed, 20% decrease in morning FEV1 or PEF on 3 of 7 days, > 12 puffs beta2/day on 2 of 7 days or > 2 nights nocturnal asthma. | |
| Outcomes | Withrawals due to adverse events, FEV1, PEF, FEF25‐75, symptoms, rescue medications, nocturnal asthma, physician global assessment, plasma cortisol, adverse events | |
| Notes | For FEV1 and morning PEF it was assumed the variance estimate was SE. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Wennergren 1996.
| Methods | Study Design: Parallel RCT Randomisation: Random stated, method not described Concealment of Allocation: controlled by pharmacy Double Blinding: Yes ‐ Doctor and patient Withdrawals / dropouts: Described Adverse events: described Statistical analysis: Described Jadad Score: 4 | |
| Participants | Study site: Conducted throughout 12 paediatric units No eligible: Not stated No randomised: 102 (50/52) No completed: 88 (44/44) Sex: males: 83 (81%); Females: 19 (19%) Age:mean: Intervention 1: 20 months; Intervention 2: 23 months Diagnostic criteria for asthma: Not stated Inclusion criteria: > 6months <= 4 years, uncontrolled asthma without steroids, symptoms min 6 days/month in past 2 months, symptoms with exercise/laughter/crying 50 % of days in past month Exclusion criteria: No ICS, concomitant lung disease, previous inclusion in study Baseline severity of asthma: | |
| Interventions | 1:Budesonide 2mg/day ( 1mg bid) for 3 weeks then halved every 3 weeks if subject fitted criteria for dose reduction until on placebo. 2: Budesonide 0.5mg/day (0.25mgbid) throughout study. If fulfilled criteria for dose reduction 3 times then placed on placebo Delivery device: Nebulizer Duration of treatment: 18 weeks Run in phase: 2‐4 weeks Other:Allowed sodium cromoglycate during study, short courses of beclomethasone or prednisolone allowed for exacerbations | |
| Outcomes | Heath care utilisation, rescue medication, asthma symptoms, adverse events, overall well‐being score | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Described as randomised; other information not available |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Adachi 2001 | Unclear if ICS free ‐ no response from authors |
| Affrime 2000 | Not ICS free for > 28 days prior to study commencement |
| Agertoft 1997 | Treatment period 2 weeks |
| Baker 1999 | 30% of subjects on ICS |
| Boe 1989 | Subjects not ICS free in month prior to study |
| Brompton Hospital | Subjects on OCS |
| Busse 1999 | Not ICS free ‐ steroid washout period 1‐28 days |
| Carpentiere 1990 | Treatment period 3 weeks |
| Chervinsky 1994 | Subjects on ICS |
| Del Bufalo 1988 | Unclear if ICS free ‐ no response from authors |
| Ellul‐Micallef 1983 | Treatment period 4 days, also given single prednisolone dose |
| Falcoz 2000 | ICS use not stated |
| Gillissen 2002 | Did not compare different doses of ICS |
| Harrison 2002 | < 4 weeks treatment |
| Kudo 1995 | Not an RCT |
| Pearlman 1999 | Patients continued asthma medication throughout study ‐ ? ICS |
| Reddel 2000 | Subjects not ICS free in month prior to study |
| Sue‐Chu 1999 | Subjects not ICS free in month prior to study |
| Turktas 1995 | Did not measure asthma outcomes |
| Visser 2002 | Subjects not ICS free in month prior to study |
| Welch 1997 | Subjects on ICS at commencement of study |
| White 1999 | Some subjects on ICS |
| ZuWallack 2000 | Not ICS free for > 28 days prior to study commencement |
Contributions of authors
Powell H ‐ protocol design, inclusion/exclusion, quality assessment, data extraction, analysis, interpretation and writing Gibson PG ‐ conception, protocol design, inclusion/exclusion, quality assessment, data extraction, analysis and interpretation, writing and editing.
Sources of support
Internal sources
No sources of support supplied
External sources
Cooperative Research Centre for Asthma, Australia.
Garfield Weston Foundation, UK.
Declarations of interest
Nil
Edited (no change to conclusions)
References
References to studies included in this review
Bisgaard 1993 {published data only}
- Bisgaard H. Systemic activity of inhaled topical steroid in toddlers studied by knemometry. Acta Paediatr 1993;82:1066‐71. [DOI] [PubMed] [Google Scholar]
Bisgaard 1999 {published data only}
- Bisgaard H, Gillies J, Groenwald M, Maden C on behalf of an International Study Group. The effect of inhaled fluticasone propionate in the treatment of young asthmatic children: a dose comparison study. Am J Respir Crit Care Med 1999;160:126‐31. [DOI] [PubMed] [Google Scholar]
Campbell 1998 {published data only}
- Campbell LM, Gooding TN, Aitchison WR, Smith N, Powell JA. Initial loading (400mcg twice daily) versus static (400 mcg nocte) dose budesonide for asthma management. PLAN Research Group. Int J Clin Pract 1998;52:361‐370. [PubMed] [Google Scholar]
Chanez 2001 {published data only}
- Chanez P, Karlstrom R, Godard P. High or standard initial dose of budesonide to control mild‐to‐moderate asthma?. Eur Respir J 2001;17:856‐62. [DOI] [PubMed] [Google Scholar]
Gershman 2000 {published data only}
- Gershman NH, Wong HH, Liu JT, Fahy JV. Low‐ and high‐dose fluticasone propionate in asthma; effects during and after treatment. Eur Respir J 2000;15:11‐18. [DOI] [PubMed] [Google Scholar]
Hampel 2000 {published data only}
- Hampel F, Lisberg E, Guerin JC, and the Study Group. Effectiveness of low doses (50 and 100 mcg b.i.d) of beclomethasone dipropionate delivered as a CFC‐free extrafine aerosol in adults with mild to moderate asthma. Study Group. J Asthma 2000;37:389‐98. [DOI] [PubMed] [Google Scholar]
Hofstra 2000 {published data only}
- Hofstra WB, Neijens HJ, Duiverman EJ, Kouwenberg JM, Mulder PG, Kuethe MC, Sterk PJ. Dose‐responses over time to inhaled fluticasone propionate treatment of exercise‐and methacholine‐induced bronchoconstriction in children with asthma. Pediatric Pulmonology 2000;29:415‐23. [DOI] [PubMed] [Google Scholar]
Jatakanon 1999 {published data only}
- Jatakanon A, Kharitonov S, Lim S, Barnes PJ. Effect of differing doses of inhaled budesonide on markers of airway inflammation in patients with mild asthma. Thorax 1999;54:108‐14. [DOI] [PMC free article] [PubMed] [Google Scholar]
Jonasson 1998 {published data only}
- Jonasson G, Carlsen K‐ H, Hultquist C. Low‐dose budesonide improves exercise‐induced bronchospasm in school children. Pediatr Allergy Immunol 2000;11:120‐25. [DOI] [PubMed] [Google Scholar]
- Jonasson G, Carlsen K‐H, Blomqvist P. Clinical efficacy of low‐dose inhaled budesonide once or twice daily in children with mild asthma not previously treated with steroids. Eur Respir J 1998;12:1099‐104. [DOI] [PubMed] [Google Scholar]
- Jonasson G, Carlsen K‐H, Jonasson C, Mowinckel P. Low‐dose inhaled budesonide once or twice daily for 27 months in children with mild asthma. Allergy 2000;55:740‐48. [DOI] [PubMed] [Google Scholar]
- Jonasson G, Carlsen K‐H, Mowinckel P. Asthma drug adherence in a long term clinical trial. Arch Dis Child 2000;83:330‐33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonasson G, Carlsen K‐H, Sodal A, Jonasson C, Mowinckel P. Patient compliance in a clinical trial with inhaled budesonide in children with mild asthma. Eur Respir J 1999;14:150‐54. [DOI] [PubMed] [Google Scholar]
Lorentzson 1990 {published data only}
- Lorentzson S, Boe J, Eriksson G, Persson G. Use of inhaled corticosteroids in patients with mild asthma. Thorax 1990;45:733‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Majima 1993 {published data only}
- Majima T, Katoh H, Yoshida N, Akiyama Y, Hashimoto N, Yamaguchi M, Horie T. Effects of high doses of beclomethasone dipropionate in bronchial asthma. Arerugi 1993;42:534‐40. [PubMed] [Google Scholar]
Miyamoto 2000 {published data only}
- Miyamoto T, Fujino S, Nakajima S, Tollemar U, Liljas B. Cost‐effectiveness of budesonide turbuhaler in the treatment of mild‐to moderate asthma in Japan. Allergol Int 1999;48:275‐85. [Google Scholar]
- Miyamoto T, Takahashi T, Nakajima S, Makino S, Yamakido M, Mano K, Nakashima M, Tollemar U, Selroos O. [A double‐blind, placebo‐controlled dose‐response study with budesonide turbuhaler in Japanese asthma patients]. Respirology 2000;5:247‐56. [DOI] [PubMed] [Google Scholar]
Nayak 2002 {published data only}
- Nayak A, Lanier R, Weinstein S, Stampone P, Welch M. Efficacy and safety of beclomethasone dipropionate extrafine aerosol in childhood asthma: a 12 week, randomized, double‐blind, placebo‐controlled study. Chest 2002;122:1956‐65. [DOI] [PubMed] [Google Scholar]
Noonan 1998 {published data only}
- Noonan MJ, Chervisnky P, Wolfe J, Liddle R, Kellermen DJ, Crescenzi KL. Dose‐related response to inhaled fluticasone propionate in patients with methacholine‐induced bronchial hyperresponsiveness: a double‐blind, placebo‐controlled study. J Asthma 1998;35:153‐64. [DOI] [PubMed] [Google Scholar]
O'Byrne 1996 {published data only}
- O'Byrne P, Cuddy L, Taylor DW, Birch S, Morris J, Syrotuik J. Efficacy and cost benefit of inhaled corticosteroids in patients considered to have mild asthma in primary care practice. Canadian Respiratory Journal 1996;3:169‐75. [Google Scholar]
Pedersen 1995 {published data only}
- Pedersen S, Hansen OR. Budesonide treatment of moderate and severe asthma in children: a dose response study. J Allergy Clin Immunol 1995;95:29‐33. [DOI] [PubMed] [Google Scholar]
Pedersen 1996 {published data only}
- Pedersen B, Dahl R, Karlstrom R, Peterson CG, Venge P. Eosinophil and neutrophil activity in asthma in a one‐year trial with inhaled budesonide. The impact of smoking. Am J Respir Crit Care Med 1996;153:1519‐29. [DOI] [PubMed] [Google Scholar]
Pirozynski 1996 {published data only}
- Pirozynski M, Kulaga Z, Karstrom R. Pulmicort (budesonide) Turbuhaler in mild to moderate asthma: comparison of initial high dose, constant low dose and placebo. Am J Respir Crit Care Med 1996;153:A343. [Google Scholar]
Sheffer 1996 {published data only}
- Sheffer AL, LaForce C, Chervinsky P, Pearlman D, Schaberg A, and the Fluticasone Propionate Asthma Study Group. Fluticasone propionate aerosol: efficacy in patients with mild to moderate asthma. J Fam Pract 1996;42:369‐75. [PubMed] [Google Scholar]
Szefler 2002 {published data only}
- Szefler SJ, Martin RJ, King TS, Boushey HA, Cherniack RM, Vernon M, Chinchilli VM, Craig TJ et al. Asthma Clinical Research Netwirk of the National Heart Lung and Blood Institute. Significant variability in response to inhaled corticosteroids for persistent asthma. J Allergy Clin Immunol 2002;109:410‐8. [DOI] [PubMed] [Google Scholar]
Tukiainen 2000 {published data only}
- Tukiainen H, Taivainen A, Majander R, Poussa T, Svahn T, Puolijoki H, Terho EO. Comparison of high and low dose of the inhaled steroid, budesonide, as an initial treatment in newly detected asthma. Respir Med 2000;94:678‐83. [DOI] [PubMed] [Google Scholar]
van der Molen 1998 {published data only}
- Molen T, Meyboom‐de Jong B, Mulder HH, Postma DS. Starting with a higher dose of inhaled corticosteroids in primary care asthma treatment. Am J Respir Crit Care Med 1998;158:121‐5. [DOI] [PubMed] [Google Scholar]
Visser 2001 {published data only}
- Visser J, Postma DS, Arends LR, Vries TW, Duiverman EJ, Brand PLP. One‐year treatment with different dosing schedules of fluticasone propionate in childhood asthma. Am J Respir Crit Care Med 2001;164:2073‐2077. [DOI] [PubMed] [Google Scholar]
Volovitz 1998 {published data only}
- Volovitz B, Soferman R, Blau H, Nussinovitch M, Varsano I. Rapid induction of clinical response with a short‐term high‐dose starting schedule of budesonide nebulizing suspension in young children with recurrent wheezing episodes. Journal of Allergy and Clin immunology 1998;101:464‐9. [DOI] [PubMed] [Google Scholar]
Wasserman 1996 {published data only}
- Wasserman SI, Gross GN, Schoenwetter WF, Munk ZM, Kral KM, Schaberg A, Kellerman DJ. A 12 week dose‐ranging study of fluticasone propionate powder in the treatment of asthma. J Asthma 1996;33:265‐74. [DOI] [PubMed] [Google Scholar]
Wennergren 1996 {published data only}
- Wennergren G, Nordvall SL, Hedlin G, Moller C, Wille S, Nilsson EA. Nebulized budesonide for the treatment of moderate to severe asthma in infants and toddlers. Acta Paediatr 1996;85:183‐9. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Adachi 2001 {published data only}
- Adachi M, Kochno Y, Minoguchi K. Step‐down and step‐up therapy in moderate persistent asthma. Int Arch Allergy Immunol 2001;124:414‐6. [DOI] [PubMed] [Google Scholar]
Affrime 2000 {published data only}
- Affrime MB, Kosoglou T, Thonoor CM, Flannery B, Herron JM. Mometasone furoate has minimal effects on the hypothalamic‐pituitary‐adrenal axis when delivered at high doses. Chest 2000;118:1538‐46. [DOI] [PubMed] [Google Scholar]
Agertoft 1997 {published data only}
- Agertoft L, Pedersen S. Short‐term knemometry and urine cortisol excretion in children treated with fluticasone propionate and budesonide: a dose response study. Eur Respir J 1997;10:1507‐12. [DOI] [PubMed] [Google Scholar]
Baker 1999 {published data only}
- Baker JW, Mellon M, Wald M, Welch M, Cruz‐Rivera M, Walton‐Bowen K. A multiple‐dosing, placebo‐controlled study of budesonide inhalation suspension given once or twice daily for treatment of persistent asthma in young children and infants. Paediatrics 1999;103:414‐21. [DOI] [PubMed] [Google Scholar]
Boe 1989 {published data only}
- Boe J, Rosenhall L, Alton M, Carlsson LG, Carlsson U, Hermansson BA, Hetta L, Kiviloog J, Karlson BW, Lundback B, et al. Comparison of dose‐response effects of inhaled beclomethasone dipropionate and budesonide in the management of asthma. Allergy 1989;44:349‐55. [DOI] [PubMed] [Google Scholar]
Brompton Hospital {published data only}
- Brompton Hospital and Medical Research Council. Double‐blind trial comparing two dosage schedules of beclomethasone dipropionate aerosol with a placebo in chronic bronchial asthma.. Br J Dis Chest 1979;73:121‐32. [PubMed] [Google Scholar]
Busse 1999 {published data only}
- Busse WW, Brazinsky S, Jacobsen K, Stricker W, Schmitt K, Vanden BurgtJ, Donnell D, Hannon S, Colice GL. Efficacy response of inhaled beclomethasone dipropionate in asthma is proportional to dose and is improved by formulation with a new propellant. J Allergy Clin Immunol 1999;104:1215‐22. [DOI] [PubMed] [Google Scholar]
Carpentiere 1990 {published data only}
- Carpentiere G, Marino S, Castello F, Baldanza C, Bonanno CT. Dose‐related effect of beclomethasone dipropionate on airway responsiveness in asthma. Respiration 1990;57:100‐3. [DOI] [PubMed] [Google Scholar]
Chervinsky 1994 {published data only}
- Chervinsky P, As A, Bronsky EA, Dockhorn R, Noonan M, LaForce C, Pleskow W. Fluticasone propionate aerosol for the treatment of adults with mild to moderate asthma. The Fluticasone Propionate Study Group. J Allergy Clin Immunol 1994;94:676‐83. [DOI] [PubMed] [Google Scholar]
Del Bufalo 1988 {published data only}
- Bufalo C, Sturani C, Miniero R, Testi R, Gunella G. Beclomethasone dipropionate: four week treatment with high dose vs standard dose in asthmatic patients. Eur Respir J 1988;1 (Suppl 2):378s. [Google Scholar]
Ellul‐Micallef 1983 {published data only}
- Ellul‐Micallef R, Johansson SA. Acute dose‐response studies in bronchial asthma with a new corticosteroid, budesonide. British Journal of Clinical Pharmacology 1983;15:419‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Falcoz 2000 {published data only}
- Falcoz C, Horton J, Mackie AE, Harding SM, Daley‐Yates PT. Pharmacokinetics of fluticasone propionate inhaled via the DiskhalerTM and DiskusTM powder devices in patients with mild‐to‐moderate asthma. Clinical Pharmacokinetics 2000;39 (Suppl 1):31‐7. [DOI] [PubMed] [Google Scholar]
Gillissen 2002 {published data only}
- Gillissen A, Richter A, Oster H, Criee C‐P. Effectiveness of low doses (2x100mcg) of HFA beclomethasone (BDP) given once daily or twice in adults with mild to moderate asthma. Am J Respir Crit Care Med 2002;165 (Suppl 8):A768. [Google Scholar]
Harrison 2002 {published data only}
- Harrison LI, Kurup S, Wagner C, Ekholm BP, Larson JS, Kaiser HB. Pharmacokinetics of beclomethasone 17‐monopropionate from a beclomethasone dipropionate extrafine aerosol in adults with asthma. Eur J Clin Pharmacol 2002;58:197‐201. [DOI] [PubMed] [Google Scholar]
Kudo 1995 {published data only}
- Kudo K, Hojo M, Kabe J. Inhaled beclomethasone in long‐term management of asthma: Optimal dose and optimal duration of treatment. Japanese Journal of Thoracic Diseases 1995;33:956‐65. [PubMed] [Google Scholar]
Pearlman 1999 {published data only}
- Pearlman DS, Kane RE, Banerji D, for the Azmacort HFA‐134a Study Group. Comparative dose‐ranging study of triamcinolone acetonide inhalation aerosol using propellants hydrofluoroalkane 134a or P‐12 in children with chronic asthma. Current Therapeutic Research 1999;60:595‐606. [Google Scholar]
Reddel 2000 {published data only}
- Reddel HK, Jenkins CR, Marks GB, Ware SI, Xuan W, Salome CM, Badcock C‐A, Woolcock AJ. Optimal asthma control, starting with high doses of inhaled budesonide. Eur Respir J 2000;16:226‐35. [DOI] [PubMed] [Google Scholar]
Sue‐Chu 1999 {published data only}
- Sue‐Chu M, Wallin A, Wilson S, Ward J, Sandstrom T, Djukanovic R, Holgate ST, Bjermer L. Bronchial biopsy study in asthmatics treated with low and high dose fluticasone propionate (FP) compared to low dose FP combined with salmeterol. European Respiratory Society. 1999; Vol. Oct 9‐13:Madrid, Spain.
Turktas 1995 {published data only}
- Turktas I, Gokcora N, Yavuz O, Elbeck S, Cevik C, Demirsoy s. Effect of inhaled budesonide on lipid metabolism and hypothalamic‐pituitary‐adrenal axis function in patients with bronchial asthma. Turkish Journal of Medical Sciences 1995;25:183‐6. [Google Scholar]
Visser 2002 {published data only}
- Visser MJ, Postma DS, Brand PL, Arends LR, Duiverman EJ, Kauffman HF. Influence of different dosage schedules of inhaled fluticasone propionate on peripheral blood cytokine concentrations in childhood asthma. Clin Exp Allergy 2002;32:1497‐503. [DOI] [PubMed] [Google Scholar]
Welch 1997 {published data only}
- Welch MJ, Levy S, Smith JA, Feiss G, Farrar JR. Dose‐ranging study of the clinical efficacy of twice‐daily triamcinolone acetonide inhalation aerosol in moderately severe asthma. Chest 1997;112:597‐606. [DOI] [PubMed] [Google Scholar]
White 1999 {published data only}
- White MV, Cruz‐Rivera M, Walton‐Bowen K. The efficacy and safety of budesonide inhalation suspension: A nebulizable corticosteroid for persistent asthma in infants and young children. Family Medicine 1999;31:337‐45. [PubMed] [Google Scholar]
ZuWallack 2000 {published data only}
- ZuWallack R, Adelglass J, Clifford DP, Duke SP, Wire PD, Faris M, Harding SM. Long‐term efficacy and safety of fluticasone propionate powder administered once or twice daily via inhaler to patients with moderate asthma. Chest 2000;118:303‐12. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
Baraket 2012 {published data only}
- Baraket M, Oliver BGG, Burgess JK, Lim S, King GG, Black JL. Is low dose inhaled corticosteroid therapy as effective for inflammation and remodeling in asthma? A randomized, parallel group study. Respiratory Research 2012;13(1):11. [2988259; IL:1465‐9921] [DOI] [PMC free article] [PubMed] [Google Scholar]
Bateman 2012 {published data only}
Busse 2014 {published data only}
- Busse WW, Bateman ED, O'Byrne PM, Lötvall J, Woodcock A, Medley H, et al. Once‐daily fluticasone furoate 50 mcg in mild‐to‐moderate asthma: a 24‐week placebo‐controlled randomized trial. Allergy 2014;69(11):1522‐30. [DOI: 10.1111/all.12480] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pinnas 2005 {published data only}
- Pinnas JL, Noonan MJ, Weinstein SF, Chervinsky P, Scott CA, Herje NE, et al. Fluticasone propionate HFA‐134a pressurized metered‐dose inhaler in adolescents and adults with moderate to severe asthma. Journal of Asthma 2005;42(10):865‐71. [DOI] [PubMed] [Google Scholar]
Ruff 2003 {published data only}
- Ruff M E, Szefler SJ, Meltzer EO, Berger WE. Efficacy and safety of extrafine beclomethasone dipropionate aerosol therapy in children with asthma: a twelve‐week placebo‐controlled trial. Pediatric Asthma, Allergy & Immunology 2004;16(1):1‐13. [2988304] [Google Scholar]
Selroos 2008 {published data only}
- Selroos O. Effect of disease duration on dose–response of inhaled budesonide in asthma. Respiratory Medicine 2008;102(7):1065–72. [DOI: 10.1016/j.rmed.2007.12.029; 2988306] [DOI] [PubMed] [Google Scholar]
Teper 2004 {published data only}
- Teper AM, Colom AJ, Kofman CD, Maffey AF, Vidaurreta SM, Bergadá I. Effects of inhaled fluticasone propionate in children less than 2 years old with recurrent wheezing. Pediatric Pulmonology 2004;37(2):115‐5. [2988312] [DOI] [PubMed] [Google Scholar]
Additional references
Adams 2002
- Adams N, Bestall J, Jones PW. Inhaled fluticasone propionate for chronic asthma. Cochrane Library 2002, Issue 1. [DOI] [PubMed] [Google Scholar]
Adams 2002 (b)
- Adams N, Bestall J, Jones PW. Budesonide for chronic asthma in children and adults. The Cochrane Library 2002, Issue 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Adams 2002 (c)
- Adams N, Bestall J, Jones PW. Inhaled Beclomethasone versus placebo for chronic asthma. The Cochrane Library 2002, Issue 1. [DOI] [PubMed] [Google Scholar]
Barnes 1998
- Barnes PJ, Pedersen S, Busse WW. Efficacy and safety of inhaled corticosteroids. AM J Respir Crit Care Med 1998;157:S1‐S53. [DOI] [PubMed] [Google Scholar]
Black 2000
- Black PN, Lawrence BJ, Goh KH, Barry MS. Differences in the potencies of inhaled steroids are not reflected in the doses prescribed in primary care in New Zealand. Eur J Clin Pharmacol 2000;56:431‐5. [DOI] [PubMed] [Google Scholar]
BTS/SIGN 2003
- The British Thoracic Society / Scottish Intercollegiate Guidelines Network. British Guideline on the Management of Asthma. Thorax 2003;58:Suppl 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Holt 2001
- Holt S, Suder A, Weatherall M, Cheng S, Shirtcliffe P, Beasley R. Dose‐response relation of inhaled fluticasone propionate in adolescents and adults with asthma: meta‐analysis. BMJ 2001;323:253‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Jadad 1996
- Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ. Assessing the quality of reports of randomized clinical trials: is blinding necessary?. Controlled Clinical Trials 1996;17(1):1‐12. [DOI] [PubMed] [Google Scholar]
NAC 2002
- National Asthma Council. Asthma Management Handbook. 2002. [Google Scholar]
NIH 2002
- National Institutes of Health, NHLBI Workshop Report, 2002. Global strategy for asthma management and prevention. NIH publication No 02‐3659. [Google Scholar]
NZGG 2003
- New Zealand Guideline Group. Best practice evidence‐based guidelines: the diagnosis and treatment of adult asthma. Enigma Publishing Pyt Ltd 2003. [Google Scholar]
Powell 2003
- Powell H, Gibson PG. Inhaled corticosteroid doses in asthma: an evidence‐based approach. Med J Aus 2003;178:223‐225. [DOI] [PubMed] [Google Scholar]
Suissa 2001
- Suissa S, Ernst P. Inhaled corticosteroids: impact on asthma morbidity and mortality. J Allergy Clin Immunol 2001;107:937‐44. [DOI] [PubMed] [Google Scholar]
Todd 2002
- Todd GR, Acerini CL, Buck JJ, Murphy NP, Ross‐Russell R, Warner JT. Acute adrenal crisis in asthmatics treated with high‐dose fluticasone propionate. Eur Respir J 2002;19:1207‐9. [DOI] [PubMed] [Google Scholar]


